Critical Care / ICU
Mechanical ventilation, sepsis management, hemodynamic monitoring, vasopressor therapy, ARDS, multi-organ dysfunction, sedation protocols, and every scoring system, ventilator setting, and management algorithm across the full scope of intensive care medicine.
01 ICU Physiology
Critical care medicine is grounded in applied cardiopulmonary physiology. The intensivist must understand oxygen delivery, ventilation-perfusion relationships, and acid-base chemistry at a quantitative level to make rational decisions about ventilator settings, fluid resuscitation, and vasoactive therapy.
Oxygen Delivery (DO2)
The fundamental equation of critical care is the oxygen delivery equation:
DO2 = CO × CaO2, where CO = HR × SV (cardiac output), and CaO2 = (1.34 × Hb × SaO2) + (0.003 × PaO2). Normal DO2 is approximately 1000 mL O2/min. Oxygen consumption (VO2) is normally 250 mL/min. The oxygen extraction ratio (O2ER) = VO2/DO2, normally ~25%. When DO2 falls below a critical threshold (~330 mL/min), VO2 becomes supply-dependent and anaerobic metabolism with lactate production begins.
Determinants of DO2 and therapeutic targets: hemoglobin (transfuse if Hb < 7 g/dL in most ICU patients per the TRICC trial; threshold 8–10 in ACS), SaO2 (target ≥ 92%; dissolved O2 contributes minimally), cardiac output (optimize preload, contractility, afterload). Mixed venous oxygen saturation (SvO2) from a PA catheter or central venous oxygen saturation (ScvO2) from a central line reflects the balance between DO2 and VO2; ScvO2 < 70% suggests inadequate delivery or excessive demand.
V/Q Mismatch, Shunt & Dead Space
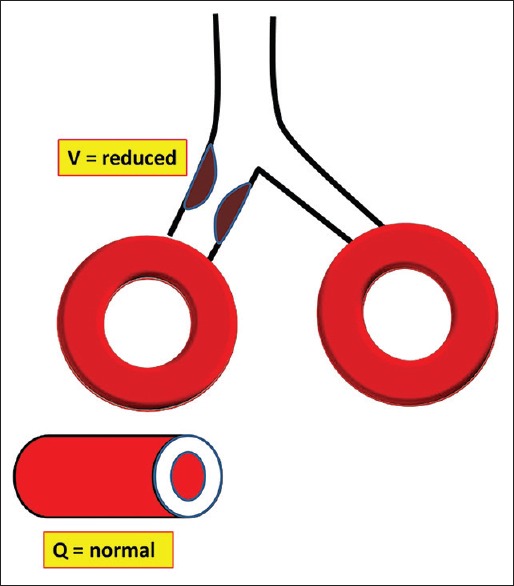
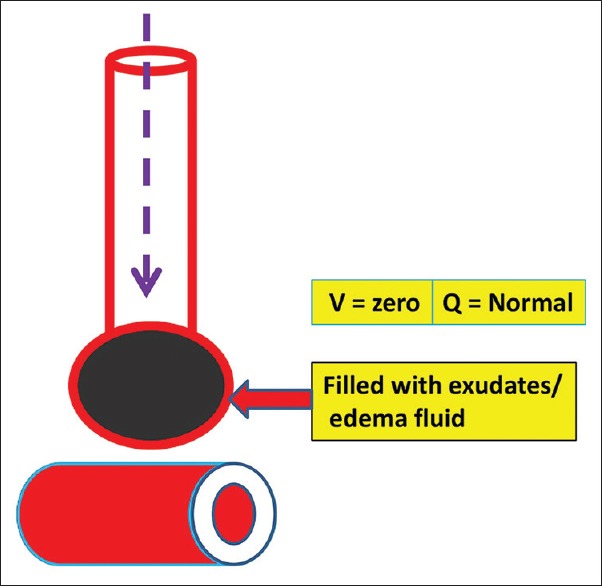
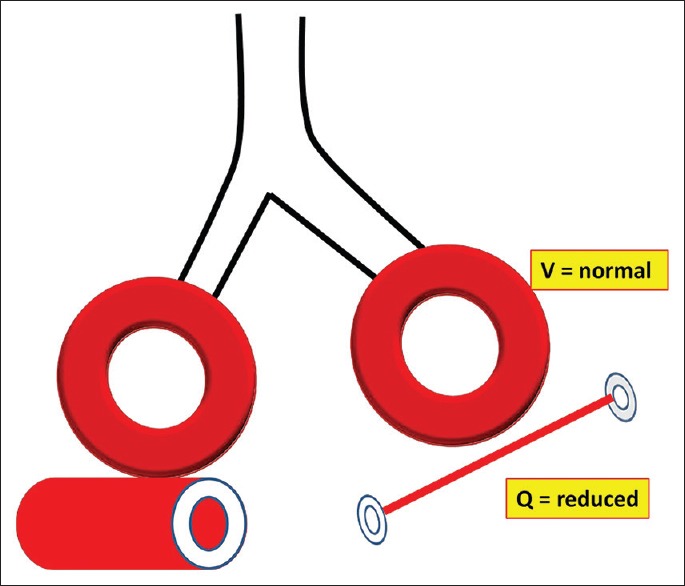
Gas exchange depends on matching ventilation (V) to perfusion (Q) across 300 million alveoli. The normal V/Q ratio is approximately 0.8.
| Concept | Definition | V/Q Ratio | Clinical Examples | Response to Supplemental O2 |
|---|---|---|---|---|
| Shunt (V/Q = 0) | Perfusion without ventilation; blood passes through non-ventilated alveoli | 0 | ARDS, pneumonia, atelectasis, intracardiac R-to-L shunt | Refractory — does not correct with O2 alone; requires PEEP/recruitment |
| Dead space (V/Q = ∞) | Ventilation without perfusion; ventilated alveoli receive no blood flow | ∞ | PE, low cardiac output, overdistension from high PEEP | Does not improve oxygenation; increases CO2 (increased Vd/Vt ratio) |
| V/Q mismatch | Spectrum between shunt and dead space; imperfect matching | Variable | COPD, asthma, pneumonia (early), PE (moderate) | Responds well to supplemental O2 |
Acid-Base Physiology — Stewart Approach
The traditional Henderson-Hasselbalch approach (pH = 6.1 + log[HCO3−]/(0.03 × PaCO2)) identifies respiratory vs. metabolic derangements, but the Stewart physicochemical approach identifies three independent determinants of pH:
| Independent Variable | Definition | Clinical Relevance |
|---|---|---|
| SID (Strong Ion Difference) | (Na+ + K+ + Ca2+ + Mg2+) − (Cl− + lactate−); normal ~40 mEq/L | Decreased SID (hyperchloremia, lactic acidosis) → acidosis; increased SID → alkalosis |
| ATOT | Total concentration of weak acids (albumin, phosphate) | Hypoalbuminemia causes alkalosis; corrected AG = AG + 2.5 × (4.0 − albumin) |
| PaCO2 | Partial pressure of CO2 | Respiratory component; increased PaCO2 → acidosis |
Anion Gap & Delta-Delta
Anion gap (AG) = Na+ − (Cl− + HCO3−); normal 12 ± 2 (or 8–10 if albumin-corrected). Always correct for albumin: corrected AG = AG + 2.5 × (4.0 − measured albumin). AGMA causes: MUDPILES — Methanol, Uremia, DKA, Propylene glycol, Isoniazid/Iron, Lactic acidosis, Ethylene glycol, Salicylates.
The delta-delta (Δ/Δ) = ΔAG / ΔHCO3− = (AG − 12) / (24 − HCO3−). If Δ/Δ < 1, there is a concurrent non-AG metabolic acidosis. If Δ/Δ > 2, there is a concurrent metabolic alkalosis. If 1–2, pure AG metabolic acidosis.
Winter's formula for expected respiratory compensation: expected PaCO2 = 1.5 × HCO3− + 8 (± 2). If measured PaCO2 is higher than expected, a concurrent respiratory acidosis is present; if lower, a concurrent respiratory alkalosis.
02 ICU Monitoring
Arterial Line & Waveform Analysis
An arterial line provides continuous beat-to-beat blood pressure monitoring and facilitates frequent blood gas sampling. Preferred sites: radial (most common), femoral, dorsalis pedis, brachial (avoid — end-artery risk). The Allen test assesses collateral ulnar circulation before radial artery cannulation.
The arterial waveform has a systolic upstroke (correlates with LV contractility), a dicrotic notch (aortic valve closure), and a diastolic runoff. A dampened waveform (loss of dicrotic notch, narrowed pulse pressure) suggests catheter kink, clot, air bubble, or arterial spasm — flush the system and reposition. An overdampened waveform underestimates systolic and overestimates diastolic; an underdampened (resonant) waveform overestimates systolic.
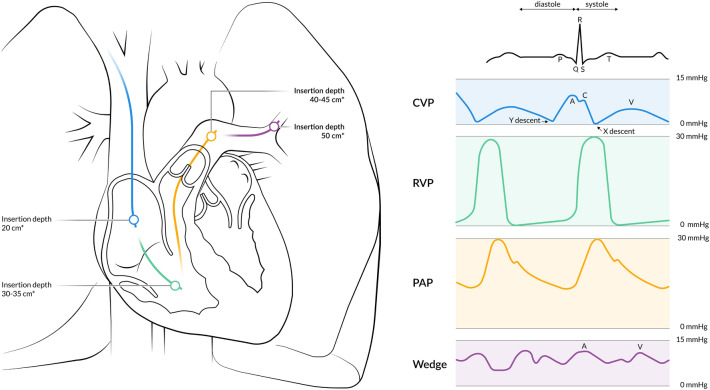
Central Venous Pressure (CVP)
CVP is measured via a central venous catheter with the transducer zeroed at the phlebostatic axis (4th intercostal space, midaxillary line). Normal CVP: 2–8 mmHg. CVP reflects right atrial pressure and is used as a surrogate for RV preload. However, CVP is a poor predictor of fluid responsiveness — systematic reviews show no correlation between CVP and preload or fluid response. Dynamic parameters (pulse pressure variation, stroke volume variation, passive leg raise) are far superior.
Pulmonary Artery (PA) Catheter
The Swan-Ganz catheter is a flow-directed, balloon-tipped catheter inserted via the IJ, subclavian, or femoral vein and advanced through the RA → RV → PA. It measures RA pressure, RV pressure, PA pressure, and pulmonary capillary wedge pressure (PCWP, a surrogate for LA pressure and LV preload).
| Parameter | Normal Range | Formula / Method | Clinical Interpretation |
|---|---|---|---|
| CVP / RA pressure | 2–8 mmHg | Direct measurement | RV preload; elevated in RV failure, tamponade, volume overload |
| PA pressure (systolic/diastolic) | 15–30 / 4–12 mmHg | Direct measurement | Elevated in pulmonary HTN, LV failure, ARDS, PE |
| PCWP | 6–12 mmHg | Balloon occlusion of PA branch | LV preload; > 18 = cardiogenic pulmonary edema; < 18 = non-cardiogenic (ARDS) |
| Cardiac output (CO) | 4–8 L/min | Thermodilution or Fick: CO = VO2 / (CaO2 − CvO2) | Low in cardiogenic shock; high in sepsis (early) |
| Cardiac index (CI) | 2.5–4.0 L/min/m2 | CO / BSA | CI < 2.2 with PCWP > 15 = cardiogenic shock |
| SVR | 800–1200 dynes·s/cm5 | 80 × (MAP − CVP) / CO | Low in sepsis/distributive shock; high in cardiogenic/hypovolemic shock |
| PVR | 20–120 dynes·s/cm5 | 80 × (mPAP − PCWP) / CO | Elevated in pulmonary HTN, ARDS, PE |
| SvO2 | 65–75% | PA blood sample | < 65% = inadequate DO2 or increased VO2 |
ScvO2 vs. SvO2
SvO2 (mixed venous) is measured from the PA catheter and represents true mixed venous blood. ScvO2 (central venous) is measured from the SVC via a central line and is a surrogate; it runs approximately 5% higher than SvO2 under normal conditions. Both track the balance between DO2 and VO2. Target ScvO2 ≥ 70% in sepsis resuscitation (Rivers early goal-directed therapy, although subsequent trials ProCESS/ARISE/ProMISe did not show benefit of protocolized ScvO2-guided resuscitation over usual care).
Lactate Trending
Lactate is a marker of tissue hypoperfusion and anaerobic metabolism. Normal < 2 mmol/L. Elevated lactate in sepsis independently predicts mortality. The Surviving Sepsis Campaign recommends lactate-guided resuscitation: remeasure lactate every 2–4 hours and target normalization or a decrease of ≥ 20% within 2 hours. Lactate clearance = (initial − delayed)/initial × 100%. Causes of elevated lactate beyond tissue hypoperfusion: epinephrine/albuterol use, hepatic dysfunction, seizures, mesenteric ischemia, thiamine deficiency, malignancy, and metformin.
Point-of-Care Ultrasound — RUSH Exam
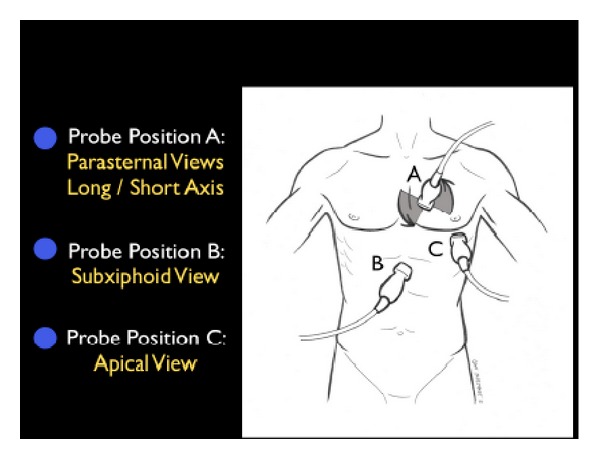
The RUSH (Rapid Ultrasound for Shock and Hypotension) exam is a systematic bedside ultrasound protocol for undifferentiated shock:
| Component | Views | Assessing |
|---|---|---|
| Pump | Parasternal long/short axis, apical 4-chamber, subxiphoid | LV function (EF eyeball), RV dilation (RV:LV > 1:1 = RV strain), pericardial effusion/tamponade |
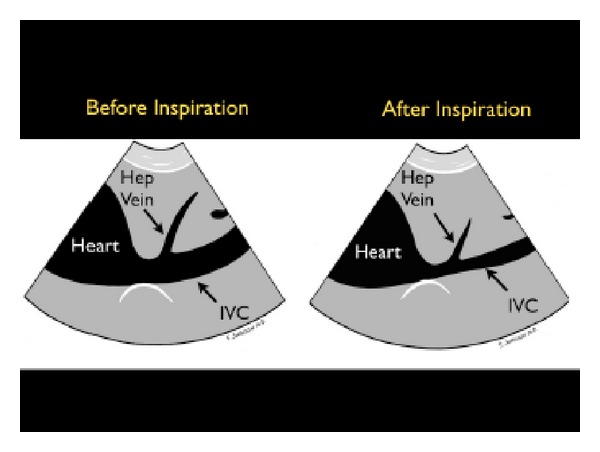
| Tank | IVC (subxiphoid), lung (anterior bilateral), FAST (RUQ, LUQ, pelvis) | IVC diameter/collapsibility (< 2.1 cm with > 50% collapse suggests low CVP/fluid responsiveness), B-lines (pulmonary edema), free fluid (hemorrhage) |
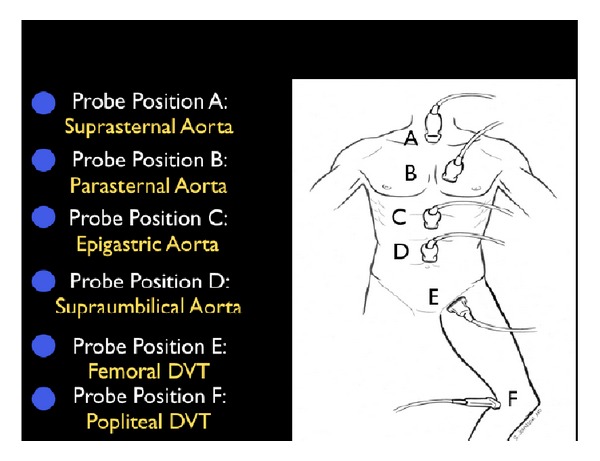
| Pipes | Abdominal aorta (epigastric, longitudinal + transverse), femoral/popliteal veins | AAA (> 3 cm), aortic dissection flap, DVT (non-compressible vein) |
Dynamic Assessment of Fluid Responsiveness
| Method | Threshold for Fluid Responsiveness | Requirements / Limitations |
|---|---|---|
| Pulse Pressure Variation (PPV) | > 13% variation over respiratory cycle | Requires mechanical ventilation with TV ≥ 8 mL/kg, no spontaneous breathing, sinus rhythm; unreliable with RV failure, low compliance, or open chest |
| Stroke Volume Variation (SVV) | > 12% variation | Same requirements as PPV; measured by arterial waveform analysis (FloTrac, PiCCO) or echocardiographic VTI variation |
| Passive Leg Raise (PLR) | ≥ 10% increase in CO or SV | Valid in spontaneous breathing, arrhythmias; must measure CO (not just BP); effects are transient (~1 min) — use real-time CO monitor |
| IVC Distensibility Index | > 18% variation (mechanically ventilated); > 40–50% collapse (spontaneous breathing) | Operator-dependent; unreliable with high PEEP, RV dysfunction, intra-abdominal hypertension, or non-intubated patients (poor specificity) |
| End-Expiratory Occlusion Test | ≥ 5% increase in CO during 15-sec expiratory hold | Simple bedside test for ventilated patients; increases venous return by eliminating PEEP effect |
Non-Invasive Cardiac Output Monitoring
Modern ICU practice increasingly uses non-invasive or minimally invasive CO monitors instead of PA catheters. Arterial pulse contour analysis (FloTrac/Vigileo, PiCCO, LiDCO): estimates SV from arterial waveform morphology; PiCCO requires transpulmonary thermodilution calibration and also provides extravascular lung water (EVLW) and global end-diastolic volume (GEDV). Echocardiographic CO: CO = VTI × LVOT area × HR (measure LVOT diameter in parasternal long axis, VTI in apical 5-chamber with pulsed-wave Doppler). Bioreactance (NICOM): non-invasive thoracic electrodes measure changes in phase shift of applied current to estimate SV; useful for trending. The PA catheter remains indicated when precise hemodynamic profiling is essential (cardiogenic shock, RV failure, pulmonary hypertension, complex mixed shock states).
03 Terminology & Abbreviations
A working knowledge of ICU abbreviations is essential for reading notes, interpreting orders, and communicating efficiently during rounds. A comprehensive master list is provided in Section 30. Key foundational terms:
04 Sepsis & Septic Shock
Sepsis-3 Definitions (2016)
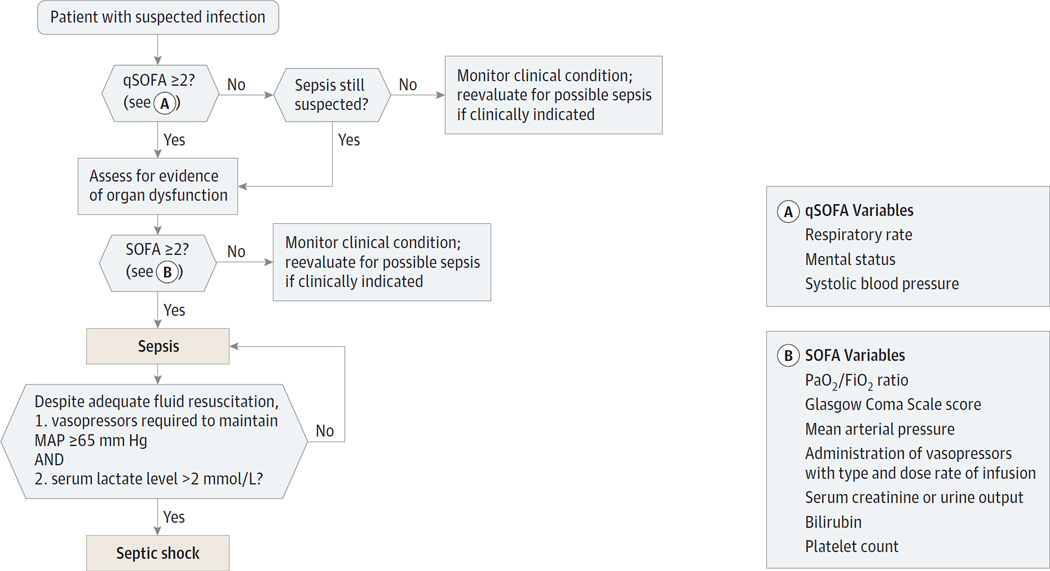
Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection. Organ dysfunction is identified as an acute change in total SOFA score ≥ 2 points consequent to the infection. Septic shock is a subset of sepsis with circulatory, cellular, and metabolic dysfunction associated with a higher mortality risk — clinically identified by a vasopressor requirement to maintain MAP ≥ 65 mmHg AND serum lactate > 2 mmol/L despite adequate volume resuscitation.
SOFA Score (Sequential Organ Failure Assessment)
| Organ System | 0 | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
| Respiration (PaO2/FiO2) | ≥ 400 | < 400 | < 300 | < 200 with respiratory support | < 100 with respiratory support |
| Coagulation (Platelets ×103/μL) | ≥ 150 | < 150 | < 100 | < 50 | < 20 |
| Liver (Bilirubin mg/dL) | < 1.2 | 1.2–1.9 | 2.0–5.9 | 6.0–11.9 | > 12.0 |
| Cardiovascular | MAP ≥ 70 | MAP < 70 | Dopamine < 5 or dobutamine (any) | Dopamine 5.1–15 or epi ≤ 0.1 or norepi ≤ 0.1 | Dopamine > 15 or epi > 0.1 or norepi > 0.1 |
| CNS (GCS) | 15 | 13–14 | 10–12 | 6–9 | < 6 |
| Renal (Creatinine mg/dL or UOP) | < 1.2 | 1.2–1.9 | 2.0–3.4 | 3.5–4.9 or UOP < 500 mL/d | > 5.0 or UOP < 200 mL/d |
qSOFA (Quick SOFA)
A bedside screening tool (not a definition of sepsis) to identify patients outside the ICU who may have sepsis. Score 1 point each for: RR ≥ 22, altered mentation (GCS < 15), SBP ≤ 100 mmHg. A qSOFA ≥ 2 should prompt further evaluation for organ dysfunction (SOFA) and consideration of sepsis.
Surviving Sepsis Campaign 2021 — Hour-1 Bundle
1. Measure lactate — remeasure within 2–4 hours if initial lactate > 2 mmol/L
2. Obtain blood cultures — before administering antibiotics (do not delay antibiotics to obtain cultures)
3. Administer broad-spectrum antibiotics — within 1 hour of recognition; each hour of delay increases mortality by ~4%
4. Begin rapid crystalloid infusion — 30 mL/kg for hypotension or lactate ≥ 4 mmol/L (use ideal or adjusted body weight; balanced crystalloid preferred)
5. Apply vasopressors — if hypotensive during or after fluid resuscitation, target MAP ≥ 65 mmHg; norepinephrine first-line
2021 guideline changes vs. 2016: Weak recommendation against using qSOFA as sole screening tool (sensitivity too low); suggestion to use balanced crystalloids (LR, Plasmalyte) over normal saline; recommendation against albumin for initial resuscitation; suggestion of capillary refill time-guided resuscitation as an adjunct to lactate; corticosteroids (IV hydrocortisone 200 mg/day) suggested for patients with ongoing vasopressor requirement after adequate fluid resuscitation.
Source Control
Source control is essential in sepsis and should be achieved as soon as medically feasible. Examples: drainage of abscess (percutaneous or surgical), debridement of necrotizing fasciitis, removal of infected device (central line, hardware), cholecystectomy for gangrenous cholecystitis, bowel resection for perforated viscus. Delay in source control independently increases mortality.
Corticosteroids in Septic Shock
The ADRENAL trial (2018) found that hydrocortisone did not reduce 90-day mortality but shortened the duration of shock and mechanical ventilation. The APROCCHSS trial (2018) found that hydrocortisone + fludrocortisone reduced 90-day mortality in septic shock. SSC 2021 suggests IV hydrocortisone 200 mg/day (50 mg q6h or continuous infusion) for patients who remain on escalating vasopressors despite adequate fluid resuscitation. Do NOT perform cosyntropin stimulation testing to select patients — relative adrenal insufficiency is common in critical illness and the test does not reliably guide therapy. Taper over 3–5 days once vasopressors are weaned. Monitor for hyperglycemia (insulin requirements increase) and hypernatremia.
Fluid Resuscitation in Sepsis
Crystalloid type: Balanced crystalloids (LR, Plasmalyte) are suggested over normal saline based on the SMART and BaSICS trials (reduced composite of death, new RRT, or persistent renal dysfunction). Avoid starch-based colloids (HES) — associated with increased AKI and mortality (6S, CHEST trials). Albumin: May be considered as a resuscitation adjunct in patients who have received large volumes of crystalloid (SSC weak recommendation); the SAFE trial showed albumin was safe but not superior to saline in general ICU patients; in the sepsis subgroup, there was a non-significant trend toward benefit. Volume assessment: After the initial 30 mL/kg bolus, further fluid administration should be guided by dynamic measures of fluid responsiveness (PPV, SVV, PLR, echocardiographic assessment) rather than static measures (CVP, MAP alone). The concept of "fluid stewardship" emphasizes that excessive fluid is harmful — the FACTT trial showed conservative fluid management improved outcomes in ARDS.
05 Vasopressors & Inotropes
Vasoactive medications are the pharmacologic backbone of shock management. They are classified by receptor activity (α1 = vasoconstriction, β1 = inotropy/chronotropy, β2 = vasodilation/bronchodilation, V1 = vasoconstriction, D1 = renal/splanchnic vasodilation) and clinical effect. The MAP target is 65 mmHg in most patients (higher in chronic hypertension, lower in young patients with hemorrhagic shock is not recommended).
| Agent | Receptor Profile | Dose Range | Primary Indication | Hemodynamic Effects | Key Considerations |
|---|---|---|---|---|---|
| Norepinephrine | α1 +++ , β1 + | 0.01–3.0 mcg/kg/min | First-line in septic shock, most shock states | ↑SVR, ↑MAP, mild ↑CO; less tachycardia than dopamine | SSC 2021 strong recommendation as first-line vasopressor; peripheral administration acceptable short-term via proximal PIV |
| Vasopressin | V1 (vascular smooth muscle) | 0.01–0.04 units/min (fixed, not titrated) | Second-line adjunct to norepinephrine in septic shock | ↑SVR, ↑MAP; no direct cardiac stimulation; may decrease CO in heart failure | Catecholamine-sparing; add when norepi ≥ 0.25–0.5 mcg/kg/min; VASST trial: benefit in less severe shock |
| Epinephrine | α1 ++ , β1 +++ , β2 ++ | 0.01–0.5 mcg/kg/min | Anaphylaxis (IM 0.3–0.5 mg); refractory septic shock; cardiogenic shock with hypotension | ↑SVR, ↑CO, ↑HR; potent inotrope + vasopressor | Increases lactate (aerobic glycolysis — not tissue hypoperfusion); arrhythmogenic; increases myocardial O2 demand |
| Phenylephrine | α1 +++ (pure) | 0.5–10 mcg/kg/min | Hypotension with tachyarrhythmia (avoid β-stimulation); neurogenic shock; adjunct | ↑SVR, ↑MAP; may reflexively ↓HR; can ↓CO | Avoid in cardiogenic shock (increased afterload); useful when tachycardia limits norepi |
| Dobutamine | β1 +++ , β2 + | 2.5–20 mcg/kg/min | Cardiogenic shock (inotropic support); low CO after adequate volume resuscitation | ↑CO, ↑HR; may ↓SVR (via β2), can ↓BP | Do not use without concurrent vasopressor if hypotensive; tachyarrhythmia common; increases myocardial O2 demand |
| Milrinone | PDE-3 inhibitor (not receptor-mediated) | 0.125–0.75 mcg/kg/min (load: 50 mcg/kg over 10 min, often omitted) | Acute decompensated HF; RV failure; pulmonary HTN | ↑CO, ↓SVR, ↓PVR; "inodilator" | Causes hypotension (loading dose especially); renally cleared (reduce in AKI); does not desensitize β-receptors (useful in β-blocker patients) |
Step 1: Norepinephrine (first-line) → titrate to MAP ≥ 65
Step 2: Add vasopressin 0.03–0.04 units/min (catecholamine-sparing) when norepi ≥ 0.25 mcg/kg/min
Step 3: Add epinephrine as third-line vasopressor if refractory
Step 4: Consider IV hydrocortisone 50 mg q6h (stress-dose steroids) if vasopressor doses remain high
Step 5: Angiotensin II (Giapreza) — rescue vasopressor for refractory vasodilatory shock (ATHOS-3 trial)
Hemodynamic Profiles by Shock Type
| Parameter | Septic (early/warm) | Cardiogenic | Hypovolemic | Obstructive (PE) |
|---|---|---|---|---|
| CO/CI | ↑ or normal | ↓↓ | ↓ | ↓ |
| SVR | ↓↓ | ↑↑ | ↑↑ | ↑ |
| CVP/PCWP | ↓ or normal | ↑↑ | ↓↓ | ↑ CVP, normal/↓ PCWP |
| SvO2 | ↑ (early) or ↓ (late) | ↓↓ | ↓ | ↓ |
| Extremities | Warm, flushed (early); cool (late) | Cool, clammy | Cool, clammy | Cool |
| First-line vasopressor | Norepinephrine | Norepinephrine + inotrope | Fluids/blood first | Treat obstruction |
06 Cardiogenic Shock
Cardiogenic shock is defined hemodynamically as CI < 2.2 L/min/m2 with an elevated PCWP > 15 mmHg (evidence of congestion) in the presence of adequate or elevated intravascular volume. It is the most lethal form of shock, with in-hospital mortality 40–60%.
Etiology
Acute MI (most common, especially large anterior STEMI — > 40% LV mass infarcted), acute-on-chronic cardiomyopathy, fulminant myocarditis, acute valvular catastrophe (mitral regurgitation from papillary muscle rupture, acute aortic regurgitation), ventricular septal rupture, free wall rupture with tamponade, stress cardiomyopathy (Takotsubo), and right ventricular infarction.
SCAI Shock Classification
| Stage | Description | Clinical Features | Hemodynamics | Lactate |
|---|---|---|---|---|
| A — At Risk | Not currently in shock but at risk | Large MI, acute HF without hypoperfusion | Normal CI, normal or mildly elevated filling pressures | Normal |
| B — Beginning | Clinical evidence of relative hypotension or tachycardia without hypoperfusion | SBP < 90 or MAP < 60 OR > 30 mmHg drop from baseline; heart rate ≥ 100 | CI ≥ 2.2, adequate UOP | Normal |
| C — Classic | Manifest hypoperfusion requiring intervention (vasopressors/inotropes/MCS) | Cold/clammy, altered mentation, oliguria, elevated JVP | CI < 2.2, PCWP > 15, CPO < 0.6 W | Elevated (≥ 2.0) |
| D — Deteriorating | Failure to stabilize on initial interventions | Worsening or failure to improve on vasopressors/inotropes | Worsening CI, escalating support | Rising |
| E — Extremis | Cardiovascular collapse, refractory cardiac arrest | PEA, refractory VT/VF, ongoing CPR with mechanical support | Near-zero CO, PEA/asystole | Severely elevated |
Management
Emergent revascularization: PCI for AMI-related cardiogenic shock is the most important intervention; the SHOCK trial demonstrated mortality benefit of early revascularization. Pharmacologic support: norepinephrine first-line vasopressor (SOAP II trial showed less arrhythmia than dopamine); dobutamine for inotropic support if adequate MAP; avoid milrinone loading dose (hypotension). Avoid excessive fluids — elevated PCWP means the patient is already volume-overloaded; further fluid worsens pulmonary edema.
Mechanical Circulatory Support (MCS)
| Device | Mechanism | CO Support | Indications | Key Limitations |
|---|---|---|---|---|
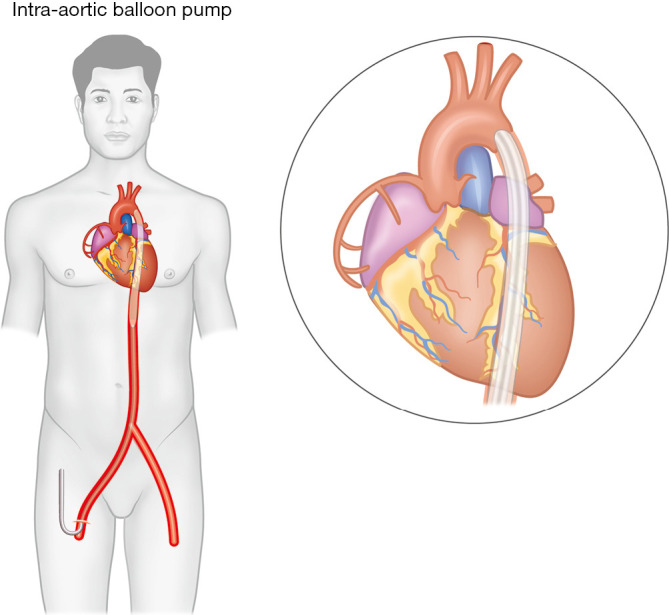
| IABP (Intra-Aortic Balloon Pump) | Inflates in diastole (augments coronary perfusion), deflates in systole (reduces afterload) | ~0.5 L/min | Limited current role; may stabilize before PCI; mechanical complications of MI | IABP-SHOCK II showed no mortality benefit in AMI cardiogenic shock; contraindicated in AI |
| Impella (CP, 5.0, 5.5) | Axial flow pump across aortic valve; aspirates LV blood into aorta | CP: 3.7 L/min; 5.0: 5 L/min; 5.5: 6.2 L/min | Cardiogenic shock (SCAI C–D), high-risk PCI, bridge to decision/recovery/transplant | Hemolysis, limb ischemia, aortic valve injury; requires large-bore arterial access |
| VA-ECMO | Femoral venous drainage → oxygenator → femoral arterial return | 3–7 L/min | Refractory cardiogenic shock, cardiac arrest (ECPR), bridge to transplant/LVAD | Increases LV afterload (may require LV vent/Impella); limb ischemia; bleeding/thrombosis |
07 Distributive, Obstructive & Hypovolemic Shock
Anaphylaxis
Anaphylaxis is a severe, potentially fatal allergic reaction causing distributive shock via massive histamine-mediated vasodilation and capillary leak. Treatment: Epinephrine IM 0.3–0.5 mg (1:1000) into anterolateral thigh — repeat every 5–15 min if no response. IV epinephrine drip (1–10 mcg/min) for refractory hypotension. Adjuncts: aggressive IVF (up to 5–10 L for capillary leak), diphenhydramine 50 mg IV (H1-blocker), ranitidine/famotidine (H2-blocker), methylprednisolone 125 mg IV (prevents biphasic reaction), albuterol nebulized (for bronchospasm). Remove trigger, maintain airway (intubate early if angioedema threatens airway).
Neurogenic Shock
Results from disruption of sympathetic outflow (spinal cord injury at T6 or above). Classic triad: hypotension + bradycardia + warm/flushed skin (loss of sympathetic tone with unopposed vagal activity). Treatment: IV fluids first, then vasopressors — norepinephrine or phenylephrine. Atropine for symptomatic bradycardia. Target MAP ≥ 85–90 mmHg for 5–7 days post-SCI to optimize spinal cord perfusion. Distinguish from spinal shock (loss of all reflexes below injury level, which is a neurological phenomenon, not a hemodynamic one).
Obstructive Shock — Tension Pneumothorax
Tension pneumothorax causes mediastinal shift, compression of great vessels, and obstructive shock. Clinical: tracheal deviation away from affected side, absent breath sounds ipsilaterally, distended neck veins, hypotension, tachycardia. Treatment is immediate needle decompression (14–16G needle, 2nd intercostal space midclavicular line or 4th/5th ICS anterior axillary line) followed by chest tube placement. This is a clinical diagnosis — do not delay treatment for imaging.
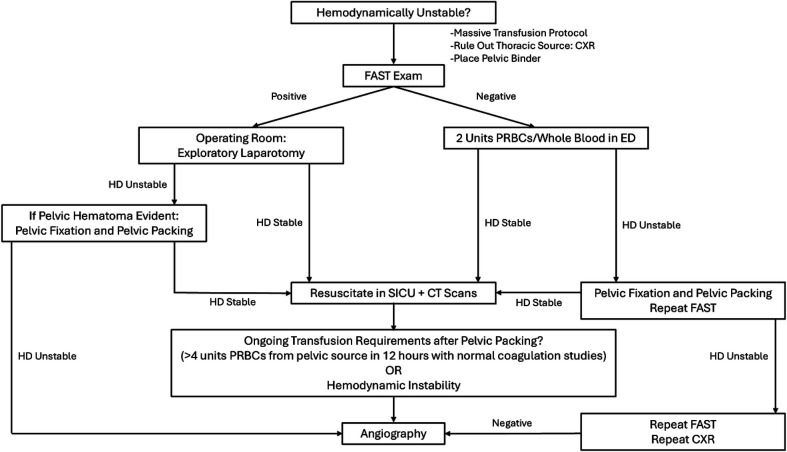
Obstructive Shock — Cardiac Tamponade
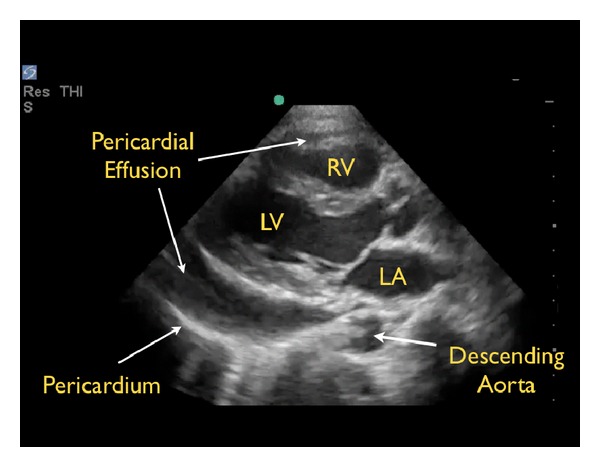
Beck's triad: hypotension, distended neck veins (JVD), muffled heart sounds. Pulsus paradoxus: > 10 mmHg drop in SBP during inspiration (exaggerated normal physiologic variation). ECG may show low voltage and electrical alternans. Bedside echo is diagnostic (pericardial effusion with RA/RV diastolic collapse). Treatment: pericardiocentesis (subxiphoid approach, ideally echo-guided) or surgical pericardial window. Bridge with IV fluids (increase preload) — avoid diuretics and positive pressure ventilation (both decrease preload and worsen tamponade physiology).
Obstructive Shock — Massive PE
Massive PE (> 50% pulmonary vascular occlusion) causes acute RV failure and obstructive shock. Signs: acute hypotension, tachycardia, elevated JVP, RV heave. Echo: RV dilation (RV:LV > 1:1), septal bowing, McConnell sign (RV free wall akinesis with apical sparing). Treatment: systemic thrombolysis with alteplase 100 mg IV over 2 hours (or 50 mg bolus in cardiac arrest). Alternatives: catheter-directed therapy, surgical embolectomy. Anticoagulate with heparin. Consider ECMO as bridge in refractory cases.
Hemorrhagic Shock Classification (ATLS)
| Class | Blood Loss | % Blood Volume | Heart Rate | Blood Pressure | Respiratory Rate | Mental Status | Treatment |
|---|---|---|---|---|---|---|---|
| I | < 750 mL | < 15% | < 100 | Normal | 14–20 | Slightly anxious | Crystalloid |
| II | 750–1500 mL | 15–30% | 100–120 | Normal (narrowed pulse pressure) | 20–30 | Anxious | Crystalloid, consider blood |
| III | 1500–2000 mL | 30–40% | 120–140 | Decreased | 30–40 | Confused | Crystalloid + blood products |
| IV | > 2000 mL | > 40% | > 140 | Profoundly low | > 35 | Lethargic/obtunded | Massive transfusion protocol (1:1:1 ratio pRBC:FFP:Platelets); TXA within 3 hours |
08 Ventilator Modes
Understanding ventilator modes requires mastery of three concepts: the trigger (what initiates the breath — patient effort or time), the target/limit (what controls gas delivery during inspiration — volume or pressure), and the cycle (what terminates inspiration — volume delivered, time elapsed, or flow decay).
Common Ventilator Modes
| Mode | Trigger | Target | Cycle | Settings | Advantages | Disadvantages |
|---|---|---|---|---|---|---|
| AC-VC (Assist-Control Volume Control) | Patient or time | Volume | Volume delivered | TV, RR, FiO2, PEEP, flow rate, flow pattern (square vs. decelerating) | Guaranteed tidal volume; predictable minute ventilation; easy to monitor compliance (plateau pressure) | Peak pressure varies with compliance/resistance; risk of barotrauma if compliance worsens; cannot limit peak pressure |
| AC-PC (Assist-Control Pressure Control) | Patient or time | Pressure | Time (set inspiratory time) | Inspiratory pressure, I-time, RR, FiO2, PEEP | Limits peak airway pressure; decelerating flow improves distribution; comfortable for patients | Tidal volume varies with compliance (must monitor closely); risk of hypoventilation if compliance drops |
| SIMV (Synchronized Intermittent Mandatory Ventilation) | Patient or time (mandatory); patient (spontaneous) | Volume or Pressure (mandatory); Pressure support (spontaneous) | Per mode | Mandatory RR + TV/pressure, PS for spontaneous breaths | Allows spontaneous breathing between mandatory breaths; historically used for weaning | Inferior for weaning vs. PSV or T-piece (longer weaning times); increased work of breathing on spontaneous breaths if PS inadequate |
| PSV (Pressure Support Ventilation) | Patient only | Pressure | Flow (terminates when flow drops to 25% of peak) | PS level, FiO2, PEEP | Patient controls RR, I-time, TV; most comfortable; excellent for weaning/SBT | No backup rate (apnea risk); tidal volume varies; not for patients with unreliable respiratory drive |
| PRVC (Pressure-Regulated Volume Control) | Patient or time | Pressure (auto-adjusted to achieve target volume) | Time | Target TV, RR, FiO2, PEEP; ventilator adjusts pressure breath-to-breath | Combines guaranteed TV with pressure-limited delivery; adapts to changing compliance | May deliver variable pressures; auto-adjustment can mask worsening compliance; not standardized across ventilators |
Initial Ventilator Settings
Mode: AC-VC (most common starting mode)
Tidal volume: 6–8 mL/kg ideal body weight (IBW: males = 50 + 2.3 × [height in inches − 60]; females = 45.5 + 2.3 × [height in inches − 60])
Respiratory rate: 14–18 breaths/min (adjust to target pH/PaCO2)
PEEP: 5 cmH2O (increase in ARDS per protocol)
FiO2: 100% initially, then rapidly wean to target SpO2 92–96% (88–92% in COPD)
Flow rate: 60 L/min (square or decelerating waveform)
I:E ratio: Target 1:2 to 1:3 (allow adequate expiratory time)
09 ARDS
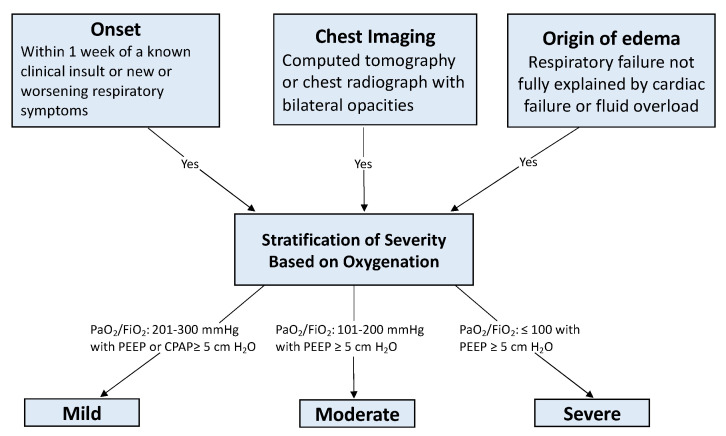
Berlin Definition of ARDS (2012)
| Criterion | Requirement |
|---|---|
| Timing | Within 1 week of known clinical insult or new/worsening respiratory symptoms |
| Imaging | Bilateral opacities not fully explained by effusions, lobar/lung collapse, or nodules (on CXR or CT) |
| Origin of edema | Respiratory failure not fully explained by cardiac failure or fluid overload (objective assessment needed, e.g., echocardiography) |
| Oxygenation | Classified by PaO2/FiO2 ratio on PEEP ≥ 5 cmH2O |
| Severity | PaO2/FiO2 | Mortality |
|---|---|---|
| Mild | 201–300 mmHg | ~27% |
| Moderate | 101–200 mmHg | ~32% |
| Severe | ≤ 100 mmHg | ~45% |
Low-Tidal-Volume Ventilation (ARDSNet Protocol)
Tidal volume: 6 mL/kg IBW (range 4–8 mL/kg; start at 6, decrease to 4 if plateau pressure > 30)
Plateau pressure: Target ≤ 30 cmH2O (check with inspiratory hold)
Driving pressure: ΔP = Plateau − PEEP; target < 15 cmH2O (strongest predictor of survival in ARDS per Amato meta-analysis)
PEEP: Set per ARDSNet PEEP/FiO2 table
pH target: 7.30–7.45; permissive hypercapnia acceptable (tolerate PaCO2 up to 60–80 if pH ≥ 7.20)
SpO2 target: 88–95%
ARDSNet PEEP/FiO2 Tables
Lower PEEP table:
| FiO2 | 0.3 | 0.4 | 0.4 | 0.5 | 0.5 | 0.6 | 0.7 | 0.7 | 0.7 | 0.8 | 0.9 | 0.9 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PEEP | 5 | 5 | 8 | 8 | 10 | 10 | 10 | 12 | 14 | 14 | 14 | 16 | 18 | 18–24 |
Higher PEEP table:
| FiO2 | 0.3 | 0.3 | 0.3 | 0.3 | 0.4 | 0.4 | 0.5 | 0.5 | 0.5–0.8 | 0.8 | 0.9 | 1.0 | 1.0 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PEEP | 5 | 8 | 10 | 12 | 14 | 16 | 16 | 18 | 20 | 22 | 22 | 22 | 24 |
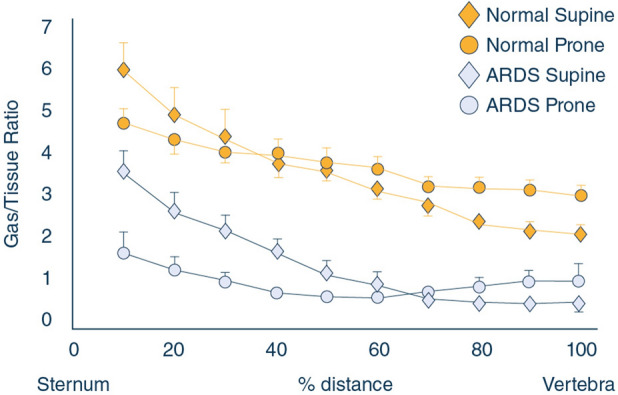
Prone Positioning
The PROSEVA trial demonstrated a significant mortality benefit (16% vs. 32.8%) for early prone positioning (≥ 16 hours/day) in severe ARDS (P/F < 150). Mechanism: improves V/Q matching by redistributing perfusion to ventral lung, recruits dorsal atelectasis, decreases ventral overdistension, improves secretion drainage. Contraindications: spinal instability, open abdomen, hemodynamic instability (relative), increased ICP (relative). Continue prone sessions until P/F > 150 on PEEP ≤ 10 and FiO2 ≤ 0.6 in supine for at least 4 consecutive hours.
Neuromuscular Blockade in ARDS
The ACURASYS trial (2010) showed mortality benefit with 48 hours of cisatracurium in early severe ARDS. However, the ROSE trial (2019) using lighter sedation found no benefit of early NMB over a strategy of light sedation with PRN NMB. Current practice: use NMB (cisatracurium 1–3 mcg/kg/min) for severe ventilator dyssynchrony, refractory hypoxemia, or high plateau pressures despite optimization, not routinely. Ensure adequate sedation before and during paralysis (patient has no ability to signal distress). Monitor with train-of-four (target 1–2/4 twitches).
Recruitment Maneuvers
Sustained inflation (30–40 cmH2O for 30–40 seconds) or stepwise PEEP increases to recruit collapsed alveoli. The ART trial (2017) showed that aggressive recruitment + high PEEP titration actually increased mortality. Use cautiously, if at all; stepwise increases with monitoring for hemodynamic compromise preferred. A brief recruitment maneuver after a disconnection or suction event is more widely accepted.
Adjunctive Therapies in Severe ARDS
| Therapy | Evidence | Recommendation |
|---|---|---|
| Inhaled pulmonary vasodilators (iNO 5–40 ppm, epoprostenol 10–50 ng/kg/min) | Improve oxygenation transiently; no mortality benefit in RCTs; iNO may increase renal failure | Rescue therapy for refractory hypoxemia as a bridge; not routine |
| ECMO (VV-ECMO) | EOLIA trial (2018): no significant mortality benefit in ITT analysis, but significant crossover; Bayesian analysis and post-hoc analyses suggest benefit in experienced centers | Consider for severe ARDS (P/F < 80 for ≥ 6 h, or P/F < 50 for ≥ 3 h, or pH < 7.25 with PaCO2 ≥ 60 for ≥ 6 h) after optimizing conventional therapy including prone; refer early to ECMO center |
| Corticosteroids | DEXA-ARDS (2020): dexamethasone 20 mg × 5 days then 10 mg × 5 days reduced ventilator days and mortality in moderate-severe ARDS; confirmed by meta-analyses | Consider dexamethasone or methylprednisolone in early moderate-severe ARDS (< 14 days); avoid initiation after day 14 (may increase mortality) |
| Conservative fluid strategy | FACTT trial: conservative fluid management improved oxygenation and shortened ventilator days without increasing organ failure | After initial resuscitation, target even to negative fluid balance; use diuretics to achieve; furosemide infusion or bolus PRN |
10 Ventilator Management & Troubleshooting
Auto-PEEP (Intrinsic PEEP)
Auto-PEEP occurs when expiratory flow does not reach zero before the next breath, causing gas trapping and increased end-expiratory pressure above the set PEEP. Common in obstructive disease (COPD, asthma) and when RR is too high or I:E ratio is too short. Detection: perform an expiratory hold maneuver — total PEEP (measured) minus set PEEP = auto-PEEP. On the flow-time waveform, expiratory flow does not return to zero baseline before the next inspiration.
Management: decrease RR (reduce minute ventilation), decrease I:E ratio (shorten inspiratory time, increase expiratory time), decrease TV (if minute ventilation is fixed), tolerate permissive hypercapnia, bronchodilators (albuterol, ipratropium), and in severe cases add external PEEP (set PEEP at ~80% of auto-PEEP to stent open airways and reduce triggering effort without further increasing total PEEP).
High Peak vs. High Plateau Pressure
| Finding | Peak Pressure | Plateau Pressure | Interpretation | Causes | Management |
|---|---|---|---|---|---|
| High Peak, Normal Plateau | ↑ | Normal (≤ 30) | Increased airway resistance (pressure dissipated across airways, not alveoli) | Bronchospasm, mucus plugging, kinked ETT, biting on tube, narrow ETT | Bronchodilators, suction, check ETT position, bite block, consider larger ETT |
| High Peak, High Plateau | ↑ | ↑ (> 30) | Decreased lung/chest wall compliance (pressure at alveolar level is elevated) | ARDS, pneumothorax, pleural effusion, pulmonary edema, abdominal compartment syndrome, auto-PEEP, mainstem intubation | Treat underlying cause; reduce TV; check for tension PTX; decompress abdomen; check ETT depth |
Ventilator Alarms
High-pressure alarm: Investigate high peak/plateau as above. Low-pressure alarm: Circuit disconnection, cuff leak, inadequate seal around tracheostomy. Low tidal volume alarm: Cuff leak, circuit leak, patient not triggering (over-sedation, neuromuscular weakness). High RR/minute ventilation alarm: Pain, anxiety, metabolic acidosis (compensatory tachypnea), PE, sepsis. Apnea alarm: Patient not triggering — check sedation depth, neuromuscular blockade, brainstem function.
Patient-Ventilator Asynchrony
| Type | Mechanism | Waveform Finding | Solution |
|---|---|---|---|
| Trigger asynchrony (missed triggers) | Patient effort insufficient to meet trigger threshold; auto-PEEP creates triggering burden | Patient effort visible on flow/pressure waveform without delivered breath | Increase trigger sensitivity; add applied PEEP (~80% of auto-PEEP); reduce sedation |
| Flow asynchrony (flow starvation) | Delivered flow rate insufficient for patient demand (AC-VC with square waveform) | Scooping/concavity in inspiratory pressure waveform | Increase flow rate; switch to decelerating flow; switch to pressure-targeted mode |
| Cycle asynchrony (premature/delayed cycling) | Ventilator cycles off before patient effort ends (premature) or continues after effort ends (delayed) | Active exhalation effort during inspiration (delayed); double-triggering (premature) | Adjust inspiratory time (PC) or cycle-off threshold (PSV); adjust flow termination criteria |
| Double-triggering | Two consecutive ventilator breaths from a single patient effort — results in stacked breaths with excessive TV | Two breaths in rapid succession without expiratory pause | Increase TV, increase inspiratory time, decrease RR; consider sedation adjustment |
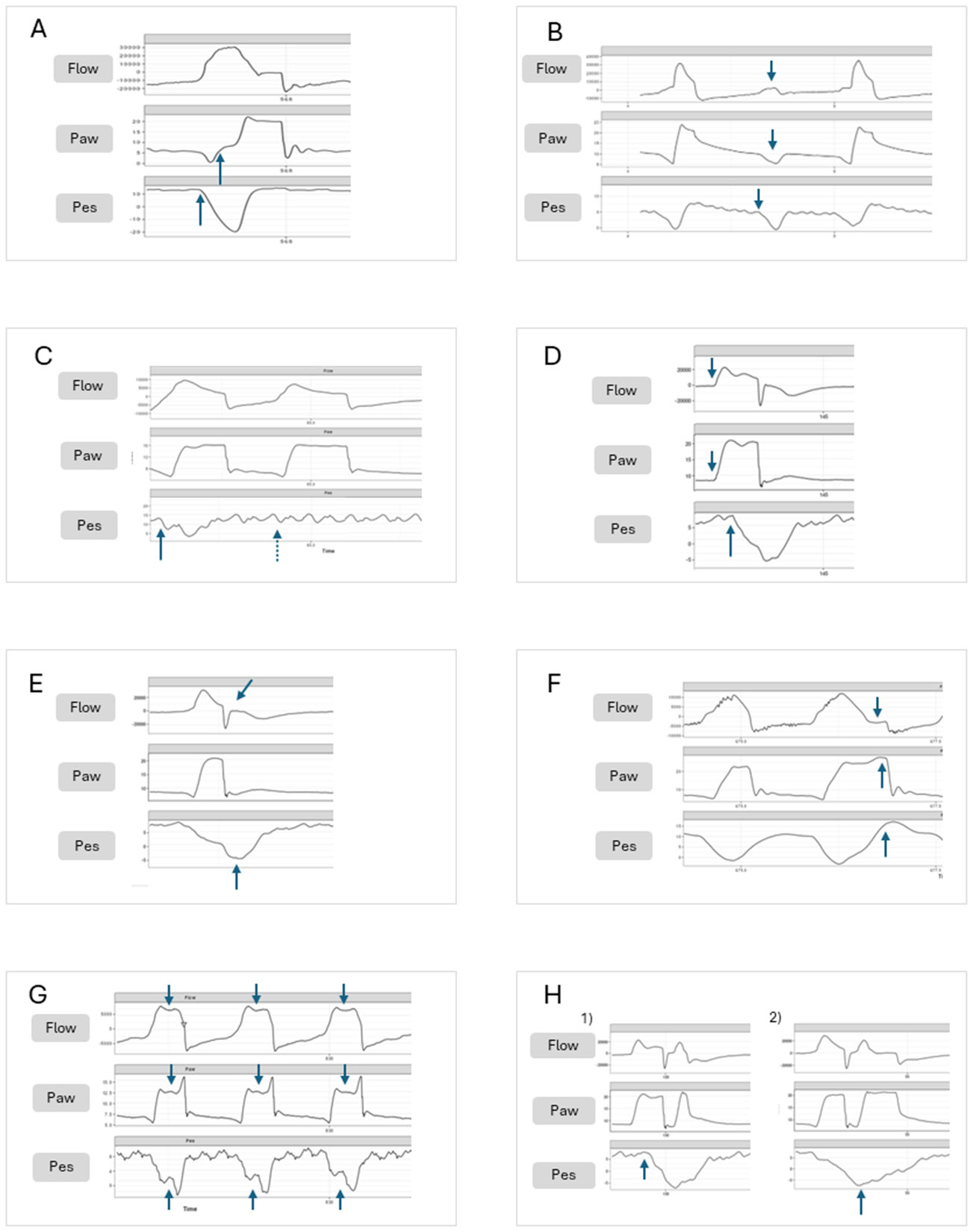
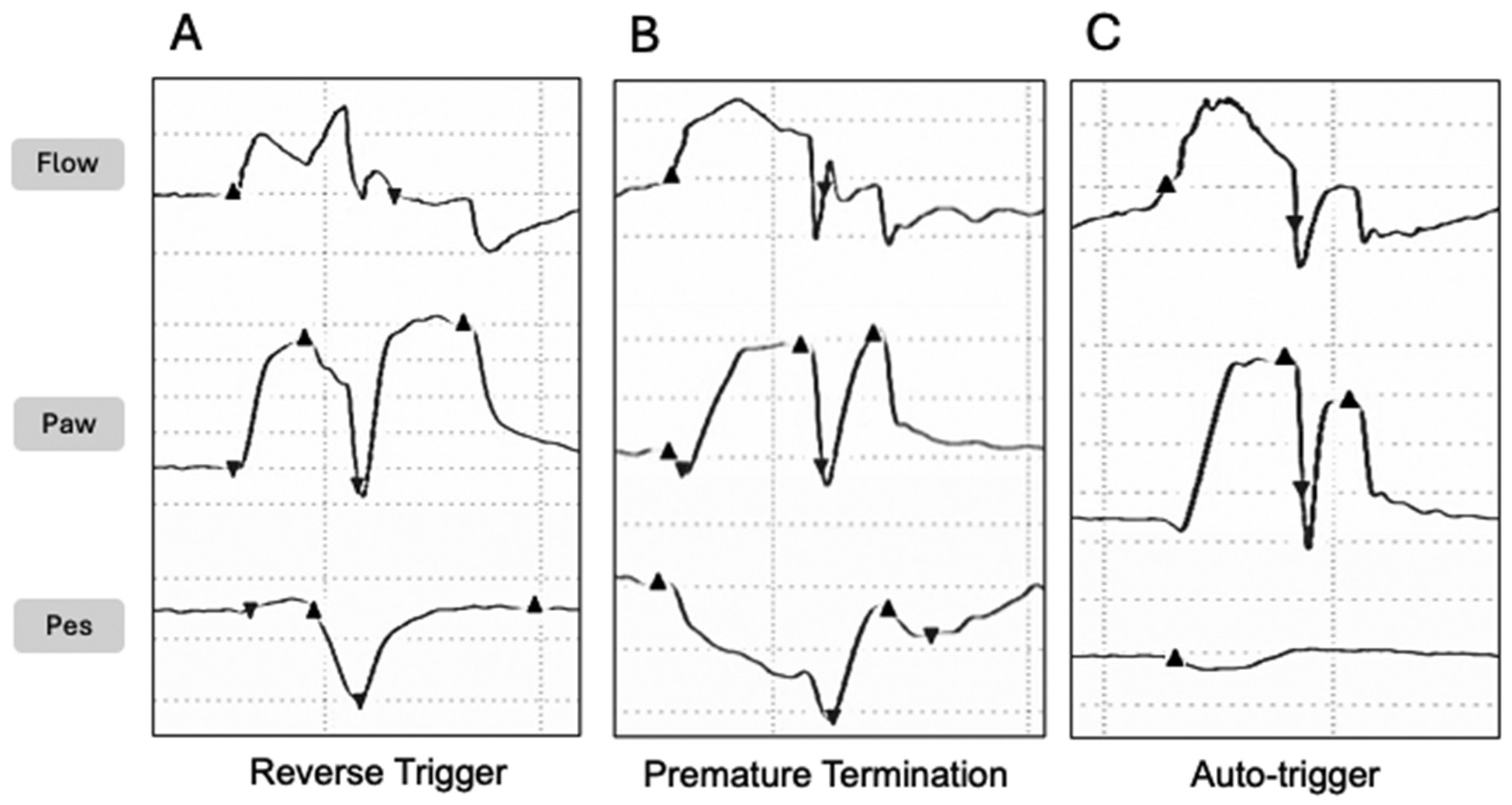
Permissive Hypercapnia
In ARDS, maintaining protective TV (6 mL/kg IBW) may result in elevated PaCO2. Permissive hypercapnia allows PaCO2 to rise to 60–80 mmHg (or even higher) as long as pH remains ≥ 7.20 (some protocols tolerate ≥ 7.15). Compensatory metabolic alkalosis develops over 24–48 hours. Contraindications to permissive hypercapnia: elevated ICP (CO2 causes cerebral vasodilation and increases ICP), severe pulmonary hypertension (hypercapnia increases PVR), significant right heart failure, and severe metabolic acidosis already present. Sodium bicarbonate infusion may be considered to buffer pH if severely acidotic, but evidence is limited and it generates CO2 that must be ventilated off.
Ventilator-Associated Pneumonia (VAP) Diagnosis
Suspect VAP when a ventilated patient (≥ 48 h on ventilator) develops new or progressive infiltrate on CXR plus at least 2 of: fever > 38°C, leukocytosis (> 12,000) or leukopenia (< 4,000), purulent secretions. Obtain lower respiratory tract cultures before starting antibiotics: endotracheal aspirate (quantitative: ≥ 106 CFU/mL) or BAL (≥ 104 CFU/mL). The Clinical Pulmonary Infection Score (CPIS) ≥ 6 suggests VAP but has limited sensitivity/specificity. Start empiric antibiotics targeting MRSA and Pseudomonas (see Section 22); narrow at 48–72 h based on culture results; total duration 7 days (procalcitonin-guided discontinuation may shorten further).
11 Liberation from Mechanical Ventilation
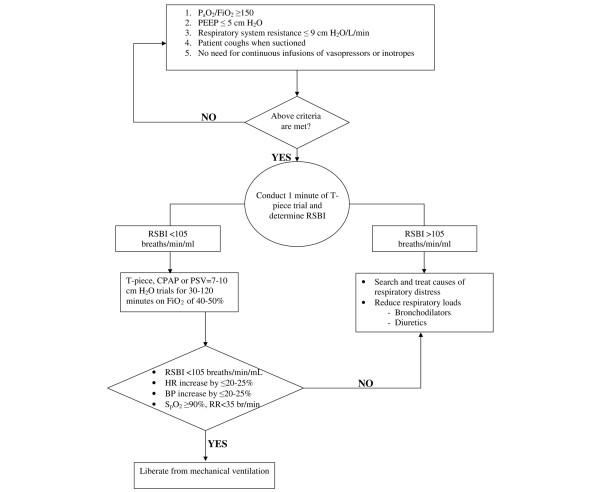
Readiness for SBT (Spontaneous Breathing Trial)
Assess daily once the patient meets the following criteria: underlying cause of respiratory failure improving/resolved, adequate oxygenation (PaO2/FiO2 ≥ 150 or SpO2 ≥ 90% on FiO2 ≤ 0.4 and PEEP ≤ 8), hemodynamically stable (no or low-dose vasopressors), able to initiate inspiratory effort, no significant acidosis (pH ≥ 7.25).
Rapid Shallow Breathing Index (RSBI)
RSBI = f / VT (respiratory rate in breaths/min divided by tidal volume in liters, measured during 1 minute of unassisted breathing or minimal support). RSBI < 105 predicts successful extubation (sensitivity ~97%, specificity ~64%). RSBI ≥ 105 predicts weaning failure. The RSBI is measured before or at the start of the SBT.
SBT Techniques
| Method | Description | Duration | Notes |
|---|---|---|---|
| T-piece | Patient breathes through ETT with humidified O2 without ventilator support | 30–120 min | Most demanding test; no PEEP to stent small airways; increased work of breathing through ETT |
| PSV 5–8 cmH2O | Low-level pressure support to overcome ETT resistance | 30–120 min | Most commonly used; may overestimate readiness (more support than after extubation); PEEP 0–5 |
| ATC (Automatic Tube Compensation) | Ventilator compensates exactly for ETT resistance based on tube diameter | 30–120 min | Most physiologically accurate simulation of post-extubation breathing |
SBT Pass/Fail Criteria
Fail criteria (terminate SBT): RR > 35, SpO2 < 90%, HR > 140 or change > 20%, SBP > 180 or < 90 mmHg, significant diaphoresis, increased anxiety/agitation, accessory muscle use, paradoxical breathing pattern. Pass criteria: Tolerated 30–120 min SBT without meeting fail criteria; RSBI < 105 throughout; adequate gas exchange maintained.
Daily Awakening + Breathing Trial (ABC Trial)
The Girard et al. (2008) ABC trial demonstrated that pairing daily sedation interruption (spontaneous awakening trial, SAT) with daily SBT reduced ventilator days, ICU LOS, and 1-year mortality. Protocol: (1) Hold sedation each morning (SAT), (2) Once patient awakens and passes safety screen, perform SBT, (3) If SBT passed, evaluate for extubation.
Post-Extubation Stridor & Cuff Leak Test
The cuff leak test assesses risk of post-extubation laryngeal edema/stridor. Method: deflate the ETT cuff and measure the difference between inspiratory and expiratory tidal volumes (the "leak"). A cuff leak volume < 110 mL (or < 24% of delivered TV) suggests significant laryngeal edema and higher risk of post-extubation stridor. For patients at risk (prolonged intubation > 7 days, traumatic intubation, prior failed extubation), administer methylprednisolone 20–40 mg IV q4–6h starting 12–24 hours before planned extubation. Post-extubation stridor treatment: nebulized racemic epinephrine 2.25% (0.5 mL in 3 mL NS), IV dexamethasone, heliox; reintubate if severe respiratory distress.
12 Non-Invasive Ventilation & High-Flow
CPAP vs. BiPAP
| Modality | Mechanism | Settings | Primary Indications |
|---|---|---|---|
| CPAP (Continuous Positive Airway Pressure) | Single level of positive pressure throughout respiratory cycle; splints open alveoli and airways, improves FRC, decreases preload/afterload | CPAP 5–15 cmH2O; FiO2 as needed | Acute cardiogenic pulmonary edema (strong evidence — 3CPO trial); OSA; post-extubation support |
| BiPAP (Bilevel Positive Airway Pressure) | Two levels: IPAP (during inspiration, augments tidal volume) and EPAP (during expiration, equivalent to PEEP); pressure support = IPAP − EPAP | IPAP 10–20, EPAP 5–10 cmH2O; FiO2; backup rate optional | COPD exacerbation (first-line NIV — reduces intubation, mortality); acute cardiogenic pulmonary edema; obesity hypoventilation; immunocompromised with respiratory failure |
High-Flow Nasal Cannula (HFNC)
Delivers heated, humidified oxygen at flows up to 60 L/min through large-bore nasal prongs. Mechanisms: washout of nasopharyngeal dead space (improves CO2 clearance), provides low-level PEEP (~1 cmH2O per 10 L/min flow, approximately 3–5 cmH2O at 50–60 L/min with mouth closed), delivers precise FiO2 (0.21–1.0), improved mucociliary clearance and comfort. The FLORALI trial demonstrated reduced intubation rates with HFNC vs. standard O2 or NIV in acute hypoxemic respiratory failure (P/F ≤ 300). Settings: start at 40–60 L/min flow, FiO2 titrated to SpO2 target.

Contraindications to NIV
Absolute: respiratory arrest, inability to protect airway, uncooperative/agitated patient, facial surgery/trauma precluding mask fit, undrained pneumothorax. Relative: hemodynamic instability, excessive secretions with poor cough, severely impaired consciousness (GCS < 10), high aspiration risk, inability to clear mask for emesis. Monitor closely for NIV failure: if no improvement in RR, SpO2, pH, or work of breathing within 1–2 hours, proceed to intubation. Delayed intubation after NIV failure is associated with worse outcomes.
Strong evidence (first-line): COPD exacerbation with respiratory acidosis (pH 7.25–7.35, PaCO2 > 45); cardiogenic pulmonary edema
Moderate evidence: Immunocompromised patients with acute respiratory failure; post-extubation respiratory failure (preventive use); facilitation of weaning in COPD
Weak/emerging evidence: Post-operative respiratory failure; acute asthma (adjunct); palliative dyspnea; do-not-intubate patients
13 Acute Kidney Injury & RRT
KDIGO Staging of AKI
| Stage | Serum Creatinine Criteria | Urine Output Criteria |
|---|---|---|
| 1 | 1.5–1.9 × baseline OR increase ≥ 0.3 mg/dL within 48 h | < 0.5 mL/kg/h for 6–12 h |
| 2 | 2.0–2.9 × baseline | < 0.5 mL/kg/h for ≥ 12 h |
| 3 | 3.0 × baseline OR increase to ≥ 4.0 mg/dL OR initiation of RRT | < 0.3 mL/kg/h for ≥ 24 h OR anuria for ≥ 12 h |
ICU AKI causes: prerenal (hypovolemia, low CO, hepatorenal syndrome — 40–60%), intrinsic (ATN from ischemia or nephrotoxins — 30–40%), and postrenal (obstruction — < 5%). FENa < 1% suggests prerenal; FENa > 2% suggests intrinsic (ATN). In patients on diuretics, use FEUrea (< 35% = prerenal). Nephrotoxin avoidance (aminoglycosides, contrast, NSAIDs), hemodynamic optimization, and avoidance of fluid overload are the cornerstones of AKI prevention.
Renal Replacement Therapy (RRT)
| Modality | Mechanism | Rate | Advantages | Disadvantages |
|---|---|---|---|---|
| IHD (Intermittent Hemodialysis) | High-efficiency solute removal over 3–4 hours | Standard dialysis session | Rapid correction of electrolytes/acidosis; less anticoagulation time | Hemodynamic instability (rapid fluid/solute shifts); intermittent — rebound between sessions |
| CRRT (Continuous Renal Replacement Therapy) | Slow, continuous solute and fluid removal over 24 h | Effluent dose 20–25 mL/kg/h | Hemodynamically better tolerated; precise fluid management; continuous clearance | Requires ICU monitoring; continuous anticoagulation; filter clotting; immobility |
| SLED/PIRRT (Prolonged Intermittent) | Hybrid: lower blood/dialysate flow rates over 6–12 hours | Variable | Compromise between IHD and CRRT; less hemodynamic impact than IHD | Less studied; not universally available |
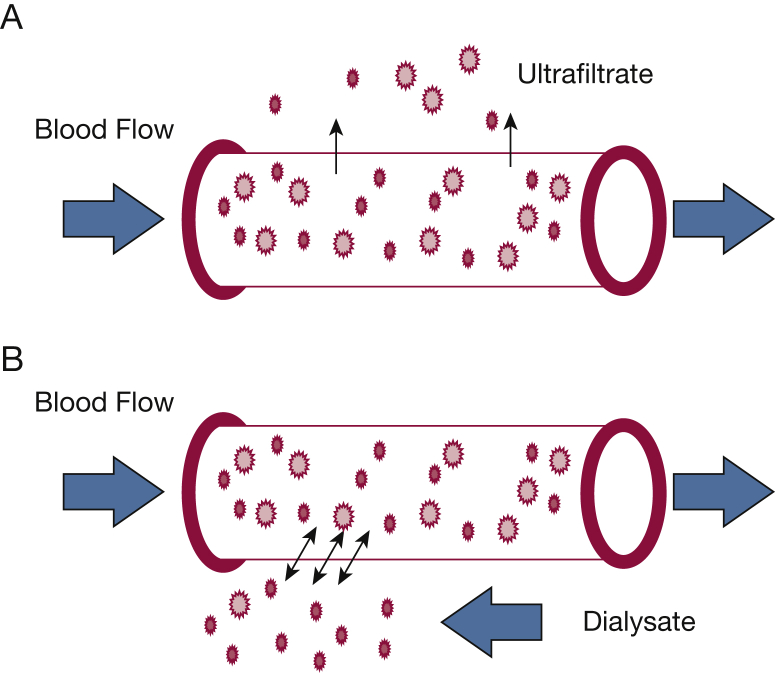
CRRT Modes
CVVH (Continuous Venovenous Hemofiltration): convective clearance only (replacement fluid pre- or post-filter). CVVHD (Continuous Venovenous Hemodialysis): diffusive clearance only (dialysate runs countercurrent to blood). CVVHDF (Continuous Venovenous Hemodiafiltration): both convective + diffusive clearance. The RENAL and ATN trials showed that higher-intensity CRRT dosing (> 25 mL/kg/h) provides no benefit over standard dosing (20–25 mL/kg/h).
Anticoagulation for CRRT
Regional citrate anticoagulation is preferred (KDIGO recommendation): citrate chelates calcium in the circuit, preventing clotting; calcium is then infused systemically to restore normal ionized calcium. Advantages: longer filter life, no systemic anticoagulation, lower bleeding risk. Contraindications: severe liver failure (citrate metabolized to bicarbonate by liver — accumulation causes citrate toxicity: low ionized calcium with elevated total calcium, metabolic acidosis). Heparin anticoagulation: systemic unfractionated heparin via circuit; simpler but increases bleeding risk; monitor aPTT.
Timing of RRT Initiation
The STARRT-AKI trial (2020) demonstrated no benefit of accelerated (early) RRT initiation compared with a standard strategy (wait for conventional indications) in critically ill patients with severe AKI. Conventional (urgent) indications for RRT: refractory hyperkalemia (K+ > 6.5 with ECG changes), severe metabolic acidosis (pH < 7.1), refractory fluid overload (pulmonary edema unresponsive to diuretics), uremic complications (pericarditis, encephalopathy, bleeding), and certain toxic ingestions (methanol, ethylene glycol, lithium, salicylates).
14 Hepatic Failure & Encephalopathy
Acute Liver Failure (ALF)
Defined as coagulopathy (INR ≥ 1.5) and hepatic encephalopathy in a patient without preexisting liver disease, with illness duration < 26 weeks. Causes: acetaminophen (most common in US/UK), viral hepatitis (A, B, E), drug-induced liver injury (idiosyncratic), autoimmune hepatitis, Wilson disease, Budd-Chiari, ischemic hepatitis ("shock liver"), HELLP syndrome, and acute fatty liver of pregnancy.
King's College Criteria (Acetaminophen)
Acetaminophen-induced ALF: pH < 7.3 (after resuscitation) OR all three of: INR > 6.5, creatinine > 3.4 mg/dL, Grade III/IV encephalopathy
Non-acetaminophen ALF: INR > 6.5 (irrespective of grade) OR any 3 of: age < 10 or > 40, non-A non-B hepatitis or drug reaction, jaundice-to-encephalopathy interval > 7 days, INR > 3.5, bilirubin > 17.5 mg/dL
West Haven Classification of Hepatic Encephalopathy
| Grade | Clinical Features | Asterixis |
|---|---|---|
| I | Trivial lack of awareness, shortened attention span, impaired arithmetic, euphoria/anxiety, altered sleep rhythm | May be present |
| II | Lethargy or apathy, disorientation to time, obvious personality change, inappropriate behavior | Present (classic) |
| III | Somnolence to semi-stupor, responsive to stimuli, confused, gross disorientation, bizarre behavior | Usually present if patient can cooperate |
| IV | Coma, unresponsive to verbal or painful stimuli | Absent (cannot test) |
Treatment of hepatic encephalopathy: Lactulose (first-line): 30–45 mL PO/NG q1–2h until bowel movement, then titrate to 2–3 soft stools/day; lactulose enemas (300 mL in 700 mL water) for patients who cannot take PO. Rifaximin (550 mg PO BID): non-absorbable antibiotic added to lactulose for secondary prophylaxis or refractory encephalopathy. Identify and treat precipitants: GI bleeding, infection/sepsis, constipation, hypokalemia, dehydration, sedating medications, dietary protein excess.
Acute-on-Chronic Liver Failure (ACLF)
ACLF is an acute deterioration of liver function in a patient with chronic liver disease, associated with organ failures and high 28-day mortality. Defined by the EASL-CLIF consortium (CLIF-SOFA score): Grade 1 = single organ failure (renal: Cr ≥ 2.0; or non-renal organ failure + renal dysfunction Cr 1.5–1.9 or HE grade 1–2); Grade 2 = two organ failures; Grade 3 = three or more organ failures. 28-day mortality: Grade 1 ~22%, Grade 2 ~32%, Grade 3 > 75%. Precipitants: infection (most common), alcoholic hepatitis, GI bleeding, drug-induced injury. Management: treat the precipitant, organ support as in any ICU patient, early transplant evaluation. Liver transplantation is the only definitive treatment for ACLF Grade 3 with failing medical therapy.
Cerebral Edema in ALF
Cerebral edema occurs in 75–80% of Grade IV encephalopathy and is the leading cause of death in ALF. Monitor: serial neurological exams, pupil reactivity; consider ICP monitoring (EVD or bolt) if Grade III–IV. Target ICP < 20 mmHg, CPP > 60 mmHg. Treatment: head of bed 30°, minimize stimulation, hypertonic saline (23.4% bolus 30 mL via CVC for acute herniation) or mannitol (0.5–1 g/kg IV — ensure serum osmolality < 320), avoid hyperthermia (target 35–36°C), hyperventilation (only as temporizing measure for acute herniation), correct hyperammonemia.
MELD-Na Score
Used for liver transplant allocation. MELD-Na = MELD + 1.32 × (137 − Na) − [0.033 × MELD × (137 − Na)], where MELD = 3.78 × ln(bilirubin) + 11.2 × ln(INR) + 9.57 × ln(creatinine) + 6.43. Na is bounded at 125–137 mEq/L. Higher MELD-Na = higher mortality = higher transplant priority.
15 GI Bleeding in the ICU
Upper GI Bleeding
Risk stratification: The Glasgow-Blatchford Score (GBS) identifies patients at very low risk (GBS = 0) who may not need endoscopy; components include BUN, hemoglobin, SBP, pulse, melena, syncope, hepatic disease, heart failure. The Rockall Score (pre-endoscopy and full) predicts rebleeding and mortality using age, shock status, comorbidities, diagnosis, and stigmata of recent hemorrhage.
Management: Resuscitate (2 large-bore IVs, type and crossmatch, transfuse pRBC if Hb < 7 g/dL, or < 9 in ACS). PPI drip: pantoprazole 80 mg IV bolus then 8 mg/h continuous infusion (initiate pre-endoscopy; continue 72 h post-endoscopy if high-risk stigmata). IV erythromycin 250 mg (prokinetic) 30–60 min before endoscopy to improve visualization. Endoscopy within 24 hours for all patients with significant UGIB; within 12 hours for high-risk (hemodynamic instability, bloody NG aspirate). Hold anticoagulants; reverse with appropriate agents as indicated by severity.
Variceal Hemorrhage
Variceal bleeding in cirrhotic patients is a medical emergency with 20% 6-week mortality. Triple therapy: (1) Octreotide 50 mcg IV bolus then 50 mcg/h infusion (reduces portal pressure via splanchnic vasoconstriction), (2) IV ceftriaxone 1 g daily (reduces bacterial translocation and rebleeding — give for 7 days), (3) Endoscopic variceal band ligation (preferred over sclerotherapy). If endoscopy fails, consider balloon tamponade (Blakemore tube — temporizing only, max 24 h) or TIPS (transjugular intrahepatic portosystemic shunt).
Lower GI Bleeding
Causes: diverticular bleeding (most common), angiodysplasia, colonic malignancy, ischemic colitis, hemorrhoids. Workup: CTA (identifies active extravasation if bleeding rate > 0.3–0.5 mL/min), colonoscopy after bowel prep (can diagnose and treat), angiography with embolization (for hemodynamically significant bleeding identified on CTA). Transfusion targets as for UGIB; restrictive transfusion strategy (Hb < 7) in hemodynamically stable patients.
16 Coagulopathy & DIC
ISTH DIC Score
| Parameter | 0 Points | 1 Point | 2 Points | 3 Points |
|---|---|---|---|---|
| Platelet count (×109/L) | > 100 | 50–100 | < 50 | — |
| D-dimer / FDP | No increase | Moderate increase | Strong increase | — |
| Prolonged PT (seconds above normal) | < 3 | 3–6 | > 6 | — |
| Fibrinogen (g/L) | > 1.0 | ≤ 1.0 | — | — |
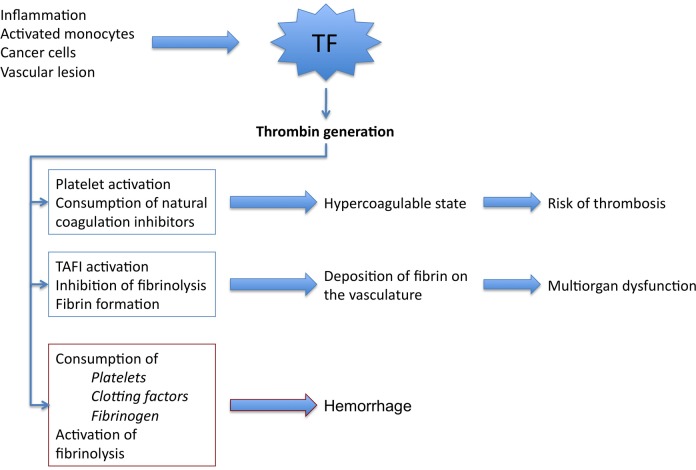
Score ≥ 5 = compatible with overt DIC. Score < 5 = suggestive, not affirmative; repeat in 1–2 days. Treat the underlying cause (sepsis, malignancy, trauma, obstetric emergency). Transfuse: FFP (15–20 mL/kg) for INR > 1.5 with active bleeding, cryoprecipitate (10 units) for fibrinogen < 150 mg/dL, platelets (1 apheresis unit) for count < 50 × 109/L with bleeding or < 20 × 109/L prophylactically. PCC (Prothrombin Complex Concentrate): 4-factor PCC for warfarin reversal or when FFP volume is prohibitive.
pRBC: Hb < 7 g/dL in most ICU patients (TRICC trial); Hb < 8 g/dL in ACS or post-cardiac surgery; Hb < 10 g/dL in severe hypoxemia or active hemorrhage
Platelets: < 10,000 prophylactically; < 50,000 before invasive procedures; < 100,000 before neurosurgery or intracranial procedures
FFP: INR > 1.5 with active bleeding or before urgent invasive procedure; not indicated to "correct" mildly elevated INR without bleeding/procedure
Cryoprecipitate: Fibrinogen < 150 mg/dL with active bleeding or DIC; < 200 mg/dL in massive hemorrhage
Transfusion reactions: Febrile non-hemolytic (most common — stop transfusion, antipyretics, rule out hemolytic reaction); acute hemolytic (ABO incompatibility — stop immediately, IVF, send DAT and clerical check); TRALI (non-cardiogenic pulmonary edema within 6 h — supportive care, avoid implicated donor); TACO (volume overload — diuretics, slow transfusion rate)
Heparin-Induced Thrombocytopenia (HIT)
Immune-mediated platelet activation via anti-PF4/heparin antibodies, causing thrombocytopenia AND paradoxical thrombosis (50% develop DVT/PE/arterial thrombosis). The 4T Score predicts pretest probability:
| Category | 0 Points | 1 Point | 2 Points |
|---|---|---|---|
| Thrombocytopenia | Fall < 30% or nadir < 10 | Fall 30–50% or nadir 10–19 | Fall > 50% AND nadir ≥ 20 |
| Timing | Fall < day 4 without recent heparin exposure | Consistent with day 5–10 but not clear; or fall after day 10; or ≤ 1 day with prior heparin 30–100 days ago | Clear onset days 5–10; or ≤ 1 day with prior heparin within 30 days |
| Thrombosis | None | Progressive, recurrent, or suspected | Confirmed new thrombosis, skin necrosis, or acute systemic reaction after heparin bolus |
| Other causes | Definite other cause present | Possible other cause | No other cause evident |
Score 0–3 = low probability; 4–5 = intermediate; 6–8 = high. If intermediate/high probability: stop all heparin immediately (including flushes), send PF4/heparin ELISA and SRA, start non-heparin anticoagulant (argatroban IV for hepatic metabolism, bivalirudin for renal/hepatic). Do NOT give warfarin until platelets recover > 150 (risk of venous limb gangrene).
Massive Transfusion Protocol & TEG/ROTEM
MTP: Activated for expected need of ≥ 10 units pRBC/24h. Target 1:1:1 ratio (pRBC:FFP:platelets). Give TXA 1 g IV within 3 hours of injury (then 1 g over 8 hours). TEG (Thromboelastography) / ROTEM (Rotational Thromboelastometry): viscoelastic assays that measure clot formation, strength, and lysis in real time. Guide targeted component therapy: prolonged R-time/CT = factor deficiency (give FFP); low MA/MCF = low fibrinogen or platelets (give cryo/platelets); increased LY30/ML = hyperfibrinolysis (give TXA). Goal-directed TEG/ROTEM-guided transfusion reduces blood product usage and improves outcomes in trauma and cardiac surgery.
17 Nutrition in the ICU
Enteral Nutrition (EN) — Preferred Route
Early enteral nutrition (within 24–48 hours of ICU admission) is recommended for all hemodynamically stable critically ill patients who cannot maintain voluntary intake. Enteral feeding maintains gut mucosal integrity, reduces bacterial translocation, modulates the immune response, and is associated with lower infection rates and shorter ICU stay compared with parenteral nutrition. Start via gastric feeding (NG/OG tube); advance to post-pyloric (NJ tube) if recurrent aspiration or gastroparesis.
Trophic vs. Full Feeds
The EDEN trial (ARDS Network) showed that trophic feeds (10–20 mL/h, ~400 kcal/day) for the first 6 days were non-inferior to full caloric feeding in early ARDS, with fewer GI complications. Current practice: trophic feeds in early critical illness (first 48–72 h), then advance to goal rate. The TARGET trial showed no benefit of energy-dense (1.5 kcal/mL) vs. standard (1 kcal/mL) formula for achieving higher caloric targets.
Caloric & Protein Targets
Caloric targets: 25–30 kcal/kg/day (use IBW for obese patients; some guidelines suggest 11–14 kcal/kg actual weight for BMI 30–50, or 22–25 kcal/kg IBW for BMI > 50). Protein targets: 1.2–2.0 g/kg/day (higher end for burns, multitrauma, surgical patients; lower for hepatic encephalopathy). Protein delivery is likely more important than meeting full caloric goals in the ICU.
Gastric Residual Volume (GRV) Debate
Routine GRV monitoring (checking residuals q4–6h, holding feeds for GRV > 250–500 mL) was standard practice but is now debated. The NUTRIREA2 sub-study and other data suggest that routine GRV monitoring does not reduce aspiration pneumonia and leads to unnecessary feed interruptions. SCCM/ASPEN 2016 guidelines suggest NOT routinely monitoring GRV. If monitored, threshold for holding feeds is GRV > 500 mL (not 200–250 mL). Prokinetics (metoclopramide 10 mg IV q6h or erythromycin 250 mg IV q12h) for gastroparesis/high residuals.
Parenteral Nutrition (PN)
Reserve PN for patients in whom EN is contraindicated (bowel obstruction, mesenteric ischemia, high-output fistula, short bowel syndrome) or when EN alone is insufficient after 7–10 days. Early PN (< 48 h) is associated with increased infections (EPaNIC trial). When PN is used, start at ~80% of target and advance; monitor glucose closely (hyperglycemia is a major complication); add supplemental PN to EN only after day 7 if EN alone is insufficient.
Refeeding Syndrome
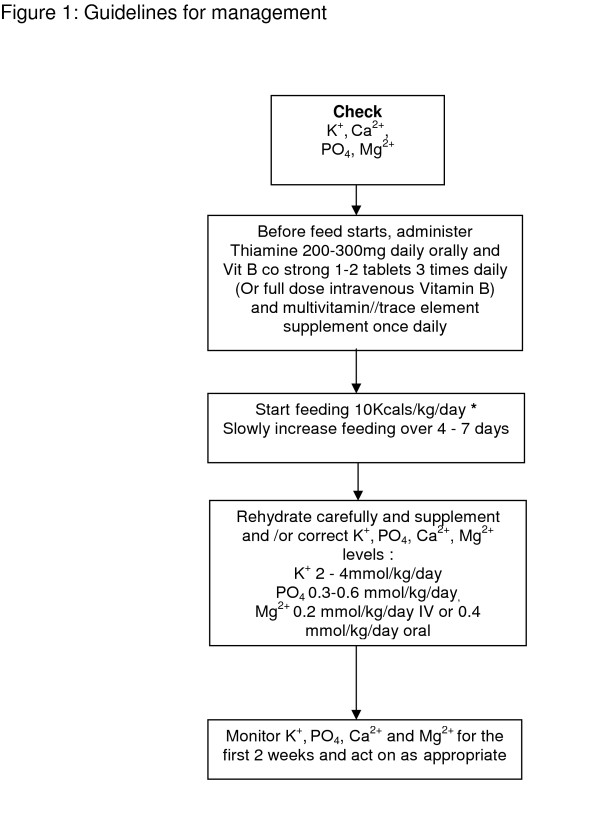
Occurs when malnourished patients (NPO > 5–7 days, BMI < 18.5, recent weight loss > 10%, chronic alcoholism, anorexia nervosa) are re-fed too rapidly. Insulin surge drives phosphate, potassium, and magnesium intracellularly. Hallmark: severe hypophosphatemia (can cause respiratory failure, cardiac arrhythmias, rhabdomyolysis, seizures, death). Prevention: check electrolytes before feeding; supplement thiamine 100–300 mg IV before starting feeds (prevents Wernicke encephalopathy); replete phosphate, potassium, magnesium aggressively; start feeds at 10–20 kcal/kg/day and advance slowly over 4–7 days; monitor electrolytes q12h for first 3 days.
18 Status Epilepticus
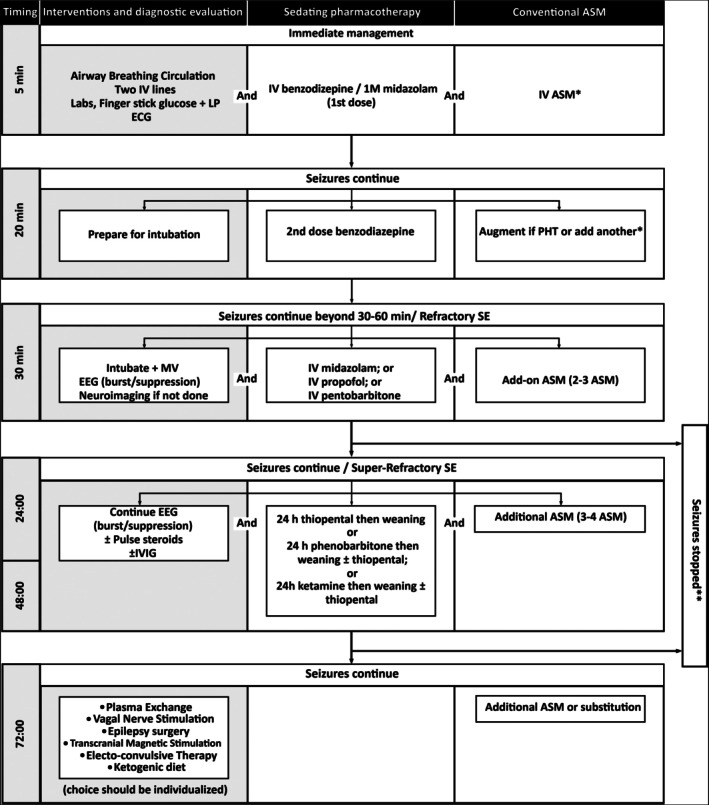
Status epilepticus (SE) is defined as a seizure lasting > 5 minutes or two or more seizures without full recovery of consciousness between them. Convulsive SE has a mortality of 10–20%; the longer the seizure, the harder it is to terminate and the greater the risk of neuronal injury.
Management Algorithm
| Phase | Time | Treatment | Details |
|---|---|---|---|
| Stabilization | 0–5 min | ABCs, glucose, vital signs | Protect airway, place patient on side, IV access, fingerstick glucose (give dextrose if low), check electrolytes, thiamine 100 mg IV if suspected alcohol use |
| First-line | 5–20 min | Benzodiazepines | Lorazepam 0.1 mg/kg IV (max 4 mg/dose, may repeat once) OR midazolam 10 mg IM (if no IV access) OR diazepam 0.15 mg/kg IV (max 10 mg). Lorazepam preferred for longer duration of action |
| Second-line | 20–40 min | Antiepileptic drugs | Fosphenytoin 20 mg PE/kg IV at 150 mg PE/min (or phenytoin 20 mg/kg IV at 50 mg/min) OR levetiracetam 60 mg/kg IV (max 4500 mg) over 10 min OR valproate 40 mg/kg IV (max 3000 mg) over 10 min. The ESETT trial showed equivalent efficacy of all three second-line agents |
| Refractory SE | > 40 min | Continuous infusions | Intubate for airway protection; then one of: propofol 1–2 mg/kg bolus then 20–80 mcg/kg/min, midazolam 0.2 mg/kg bolus then 0.05–2 mg/kg/h, OR pentobarbital 5 mg/kg bolus then 1–5 mg/kg/h. Titrate to seizure suppression or burst-suppression on continuous EEG |
| Super-refractory SE | > 24 h | Continues despite adequate anesthetic treatment | Consider ketamine 1–3 mg/kg bolus then 1–5 mg/kg/h; inhaled anesthetics (isoflurane); ketogenic diet; immunotherapy if autoimmune etiology suspected |
Continuous EEG monitoring is essential for all patients with refractory SE (clinical seizures may stop while electrographic seizures persist) and should be applied within 1 hour of onset. Non-convulsive SE accounts for up to 20% of ICU patients with unexplained altered mental status — a high index of suspicion is required.
19 Traumatic Brain Injury
Glasgow Coma Scale (GCS)
| Component | Response | Score |
|---|---|---|
| Eye Opening (E) | Spontaneous | 4 |
| To voice | 3 | |
| To pain | 2 | |
| None | 1 | |
| Verbal (V) | Oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Incomprehensible sounds | 2 | |
| None | 1 | |
| Motor (M) | Obeys commands | 6 |
| Localizes pain | 5 | |
| Withdraws from pain (flexion) | 4 | |
| Abnormal flexion (decorticate) | 3 | |
| Extension (decerebrate) | 2 | |
| None | 1 |
GCS 13–15 = mild TBI; 9–12 = moderate TBI; 3–8 = severe TBI (intubate for airway protection). The motor component is the most prognostically valuable individual component.
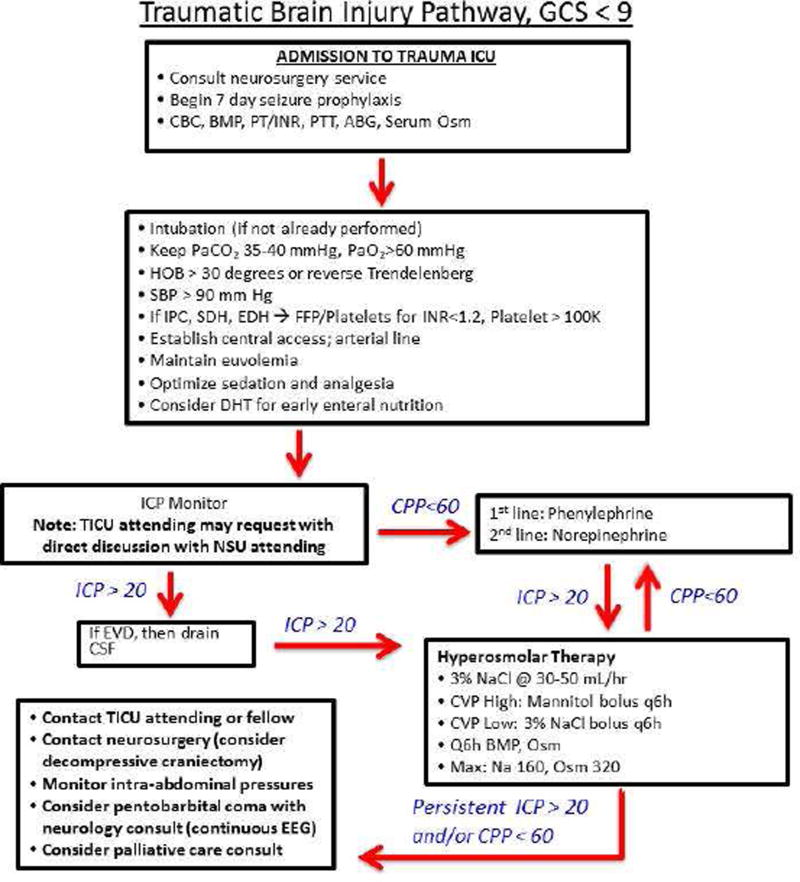
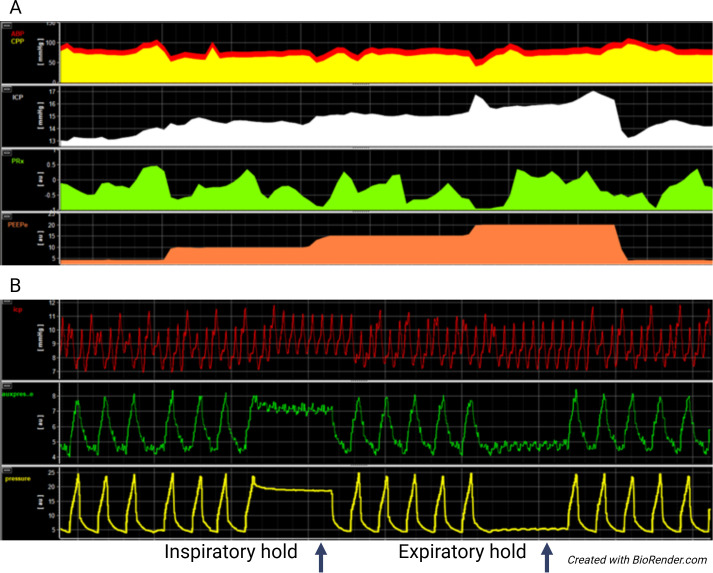
ICP Monitoring
Indications: severe TBI (GCS 3–8) with abnormal CT, or GCS 3–8 with normal CT plus 2 of: age > 40, unilateral or bilateral motor posturing, SBP < 90. Devices: External Ventricular Drain (EVD) — gold standard, allows both monitoring and therapeutic CSF drainage; placed into the lateral ventricle. Intraparenchymal bolt — fiber-optic or strain-gauge sensor; easier to place, cannot drain CSF, prone to drift. Target: ICP < 22 mmHg (BTF 2016 guidelines, revised from < 20). CPP targets: 60–70 mmHg (CPP = MAP − ICP; avoid CPP > 70 to reduce risk of ARDS from aggressive fluid/vasopressor therapy).
ICP Management Ladder
Tier 0 — General measures: Head of bed 30°, head midline (avoid jugular venous compression), avoid hyperthermia, treat pain/agitation, avoid hyponatremia (target Na 140–150 in severe TBI), maintain normoglycemia
Tier 1 — Sedation & CSF drainage: Propofol or midazolam infusion; drain CSF via EVD (5–10 mL boluses or continuous drainage with ICP set point)
Tier 2 — Osmotherapy: Mannitol 0.5–1 g/kg IV bolus (20% solution); maintain serum osmolality < 320 mOsm/kg; OR hypertonic saline (23.4% 30 mL via CVC for acute herniation, or 3% NaCl infusion targeting Na 145–155). Mannitol requires intact BBB and adequate renal function; HTS may be used even with renal insufficiency
Tier 3 — Hyperventilation: Brief, targeted hyperventilation (PaCO2 30–35 mmHg) as temporizing measure ONLY for acute herniation; avoid prolonged hyperventilation (causes cerebral vasoconstriction and ischemia)
Tier 4 — Decompressive craniectomy: Large unilateral or bifrontal craniectomy for refractory ICP elevation. The DECRA and RESCUEicp trials showed reduced mortality but increased severe disability; decision requires careful risk-benefit discussion
Mannitol vs. Hypertonic Saline
| Feature | Mannitol (20%) | Hypertonic Saline (3% or 23.4%) |
|---|---|---|
| Mechanism | Osmotic diuresis; draws water from brain into intravascular space | Osmotic shift; draws water from brain; may also reduce brain edema via anti-inflammatory effects |
| Dose | 0.5–1.0 g/kg IV bolus; repeat q4–6h PRN | 3%: 150–500 mL IV bolus or infusion (target Na 145–155); 23.4%: 30 mL via CVC for acute herniation |
| Onset | 15–30 min; peak at 60–90 min | 15–30 min |
| Monitoring | Serum osmolality (hold if > 320 mOsm/kg); serum Na; I/O (significant diuresis) | Serum Na q4–6h (hold if Na > 160); central line required for 23.4% |
| Advantages | Long track record; can give peripherally | Does not cause diuresis (may be advantageous in hypovolemia); may be more effective in refractory ICP elevation; usable in renal failure |
| Disadvantages | Causes hypovolemia (osmotic diuresis); contraindicated in renal failure (cannot excrete); rebound ICP elevation possible | Requires central access for 23.4%; risk of central pontine myelinolysis if Na rises too rapidly |
Multimodal Neuromonitoring
Beyond ICP monitoring, advanced neuromonitoring in severe TBI includes: brain tissue oxygen (PbtO2) monitoring via Licox probe (target PbtO2 > 20 mmHg; BOOST-3 trial evaluating PbtO2-guided therapy); cerebral microdialysis (lactate/pyruvate ratio > 40 suggests cerebral metabolic crisis); continuous EEG (detect non-convulsive seizures, which occur in 20–30% of severe TBI); transcranial Doppler (evaluate for vasospasm, assess autoregulation via pulsatility index and mean flow velocities). Integrated multimodal monitoring provides a more comprehensive picture of brain physiology than ICP alone.
20 Stroke in the ICU
Acute Ischemic Stroke (AIS)
IV alteplase (tPA): 0.9 mg/kg (max 90 mg), 10% as bolus then remainder over 60 min. Eligible within 4.5 hours of symptom onset (or last known well). Exclusions for 3–4.5 hour window: age > 80, NIHSS > 25, oral anticoagulant use, history of both DM and prior stroke. Tenecteplase (0.25 mg/kg, max 25 mg single IV bolus) is emerging as an alternative with simpler administration and non-inferior efficacy (AcT trial 2024). BP must be < 185/110 before and < 180/105 for 24 hours after tPA.
Mechanical thrombectomy: for large vessel occlusion (ICA, M1, proximal M2) with NIHSS ≥ 6, ASPECTS ≥ 6, up to 24 hours from onset (DAWN and DEFUSE 3 trials using perfusion imaging to select patients with salvageable penumbra). Combined with IV tPA if within tPA window.
Blood Pressure Management
| Clinical Scenario | BP Target | Rationale |
|---|---|---|
| AIS, not receiving tPA or thrombectomy | Permissive hypertension up to 220/120 | Maintain perfusion to ischemic penumbra; lower only if > 220/120 or end-organ damage |
| AIS, receiving tPA | < 180/105 for 24 hours | Reduce hemorrhagic transformation risk |
| AIS, post-thrombectomy with reperfusion | < 140–160 systolic (varies by protocol) | Reduce reperfusion injury and hemorrhagic conversion |
| ICH (intracerebral hemorrhage) | Target SBP 130–150 (INTERACT2/ATACH-2) | Reduce hematoma expansion; avoid excessive lowering (< 130) which may worsen outcomes |
Malignant MCA Infarction
Large MCA territory infarcts may develop massive edema peaking at 48–96 hours with midline shift, herniation, and death (80% mortality with medical management alone). Decompressive hemicraniectomy within 48 hours reduces mortality from ~80% to ~20% in patients age < 60 (DECIMAL, DESTINY, HAMLET trials pooled analysis). However, many survivors have significant disability (mRS 4). Discuss goals of care and acceptable functional outcomes with family before proceeding.
Hemorrhagic Stroke — Reversal Agents
| Anticoagulant | Reversal Agent | Dose | Notes |
|---|---|---|---|
| Warfarin | 4-factor PCC (Kcentra) | INR-based: INR 2–4 → 25 units/kg; INR 4–6 → 35 units/kg; INR > 6 → 50 units/kg | Preferred over FFP (faster, lower volume); give with vitamin K 10 mg IV |
| Dabigatran | Idarucizumab (Praxbind) | 5 g IV (two 2.5 g vials) | Monoclonal antibody fragment; complete reversal within minutes |
| Rivaroxaban, Apixaban, Edoxaban | Andexanet alfa (Andexxa) | Low dose: 400 mg bolus + 4 mg/min × 120 min; High dose: 800 mg bolus + 8 mg/min × 120 min | Recombinant modified factor Xa decoy; thrombotic risk ~10%; 4-factor PCC (50 units/kg) is alternative |
| Heparin (UFH) | Protamine | 1 mg per 100 units heparin given in prior 2–3 h (max 50 mg) | Anaphylaxis risk (especially in NPH insulin users, fish allergy) |
21 Sedation & Analgesia
PADIS Guidelines 2018 (Pain, Agitation/sedation, Delirium, Immobility, Sleep)
Key principles: Analgesia-first approach (treat pain before treating agitation), target light sedation (RASS 0 to −2) unless deeper sedation required (ARDS prone positioning, NMB, ICP management), conduct daily sedation interruption paired with SBT, use non-benzodiazepine sedatives (propofol or dexmedetomidine preferred over midazolam/lorazepam to reduce delirium), and implement early mobility.
Richmond Agitation-Sedation Scale (RASS)
| Score | Term | Description |
|---|---|---|
| +4 | Combative | Overtly combative, violent, immediate danger to staff |
| +3 | Very agitated | Pulls or removes tubes or catheters; aggressive |
| +2 | Agitated | Frequent non-purposeful movement, fights ventilator |
| +1 | Restless | Anxious but movements not aggressive or vigorous |
| 0 | Alert & calm | Spontaneously pays attention to caregiver |
| −1 | Drowsy | Not fully alert but has sustained (> 10 sec) awakening to voice (eye-opening/contact) |
| −2 | Light sedation | Briefly (< 10 sec) awakens with eye contact to voice |
| −3 | Moderate sedation | Movement or eye opening to voice (no eye contact) |
| −4 | Deep sedation | No response to voice, movement or eye opening to physical stimulation |
| −5 | Unarousable | No response to voice or physical stimulation |
Sedation & Analgesia Agents
| Agent | Class | Dose Range | Onset | Advantages | Disadvantages |
|---|---|---|---|---|---|
| Fentanyl | Opioid | 25–200 mcg/h IV infusion | 1–2 min | No histamine release; no active metabolites (preferred in renal failure); short-acting | Chest wall rigidity at high doses; accumulates in hepatic failure; respiratory depression |
| Hydromorphone | Opioid | 0.2–2 mg IV q2–4h or 0.2–0.8 mg/h infusion | 5 min | Longer duration than fentanyl; good for moderate-severe pain | Active metabolite hydromorphone-3-glucuronide (neuroexcitatory, accumulates in renal failure) |
| Propofol | GABA agonist | 5–80 mcg/kg/min | 30–60 sec | Rapid onset/offset; easy titration; anticonvulsant; reduces ICP | Hypotension (vasodilation + negative inotropy); hypertriglyceridemia; propofol infusion syndrome (PRIS) with prolonged high-dose use (> 5 mg/kg/h > 48h): rhabdo, metabolic acidosis, cardiac failure, death; count lipid calories (1.1 kcal/mL) |
| Dexmedetomidine | α2 agonist | 0.2–1.5 mcg/kg/h (no bolus) | 5–10 min | Cooperative sedation (arousable); less delirium; no respiratory depression; facilitates extubation | Bradycardia; hypotension; no analgesic ceiling; expensive; not appropriate for deep sedation |
| Midazolam | Benzodiazepine | 0.02–0.1 mg/kg/h | 2–5 min | Anxiolytic; anticonvulsant; amnestic | Prolonged sedation (active metabolite alpha-hydroxymidazolam, accumulates in renal failure/obesity); increases delirium; not first-line per PADIS |
ICU Delirium — CAM-ICU
The Confusion Assessment Method for the ICU (CAM-ICU) is the validated screening tool. Positive CAM-ICU requires: (1) acute onset or fluctuating mental status AND (2) inattention (squeeze hand on letter "A" in "SAVEAHAART" — ≥ 2 errors) PLUS EITHER (3) altered level of consciousness (RASS ≠ 0) OR (4) disorganized thinking (ask standardized questions + command: "hold up this many fingers"). ICU delirium affects 60–80% of mechanically ventilated patients and is independently associated with increased mortality, longer ICU stay, and long-term cognitive impairment. Prevention: ABCDEF bundle (Assess/prevent/manage pain; Both SAT + SBT; Choice of sedation — avoid benzodiazepines; Delirium assessment; Early mobility; Family engagement). Pharmacologic treatment of hyperactive delirium: haloperidol 2–5 mg IV q4–6h PRN (monitor QTc); quetiapine 50 mg PO BID as scheduled (MIND-USA trial showed no benefit of antipsychotics for delirium duration).
22 Antibiotics in the ICU
Empiric Antibiotic Regimens by Suspected Source
| Suspected Source | Empiric Regimen | Duration | Key Notes |
|---|---|---|---|
| Community-acquired pneumonia (CAP), ICU admission | β-lactam (ceftriaxone 2 g or ampicillin-sulbactam 3 g) + azithromycin 500 mg IV; OR β-lactam + respiratory FQ (levofloxacin 750 mg) | 5–7 days | If risk factors for MRSA/Pseudomonas (prior cultures, structural lung disease): add vancomycin + antipseudomonal β-lactam |
| Hospital-acquired / ventilator-associated pneumonia (HAP/VAP) | Antipseudomonal β-lactam (piperacillin-tazobactam 4.5 g q6h or meropenem 1 g q8h or cefepime 2 g q8h) + vancomycin or linezolid (for MRSA coverage) | 7 days | Two antipseudomonals if risk for resistant organisms; narrow based on sputum culture |
| Intra-abdominal sepsis | Piperacillin-tazobactam 4.5 g q6h OR meropenem 1 g q8h; OR ceftriaxone + metronidazole if lower severity | 4–5 days post source control | Source control is paramount; anaerobic coverage essential |
| Urinary tract (complicated UTI / urosepsis) | Ceftriaxone 2 g or FQ (if local resistance < 10%); add vancomycin if Enterococcus suspected | 7–14 days | Drain obstruction if present (nephrostomy/stent); ESBL coverage with meropenem if risk factors |
| Skin/soft tissue (necrotizing fasciitis) | Meropenem + vancomycin + clindamycin (for toxin suppression) | Until source controlled + clinical improvement | Emergent surgical debridement is the definitive treatment; antibiotics alone are insufficient |
| CNS infection (meningitis) | Vancomycin + ceftriaxone 2 g q12h + ampicillin 2 g q4h (if age > 50 or immunocompromised for Listeria) + dexamethasone 0.15 mg/kg q6h | 10–14 days (varies by organism) | Give dexamethasone before or with first antibiotic dose (reduces mortality in pneumococcal meningitis) |
| Bloodstream infection (unknown source) | Vancomycin + antipseudomonal β-lactam (pip-tazo or meropenem) | Depends on organism and source | Remove/exchange central lines; repeat cultures at 48–72 h; evaluate for endocarditis if S. aureus |
Extended / Prolonged Infusions
Time-dependent antibiotics (β-lactams) are more effective when free drug concentration remains above the MIC for a greater percentage of the dosing interval (%fT > MIC). Extended infusions (3–4 hour infusion): piperacillin-tazobactam 4.5 g q8h infused over 4 hours; meropenem 1–2 g q8h infused over 3 hours; cefepime 2 g q8h infused over 4 hours. Continuous infusion: some centers use 24-hour continuous infusion of meropenem or pip-tazo. Evidence suggests improved clinical cure rates and reduced mortality, especially in critically ill patients with augmented renal clearance.
MRSA Coverage — Vancomycin Dosing
Traditional trough-based monitoring (target trough 15–20 mcg/mL) has been replaced by AUC/MIC-guided dosing (target AUC/MIC 400–600) per 2020 consensus guidelines. AUC-guided dosing reduces nephrotoxicity while maintaining efficacy. Loading dose: 25–30 mg/kg IV (based on actual body weight). Maintenance: 15–20 mg/kg IV q8–12h, adjusted by AUC calculation (Bayesian software or two-level strategy).
Antifungal Coverage
Consider empiric antifungal in: prolonged ICU stay (> 7 days) on broad-spectrum antibiotics, TPN, recent abdominal surgery, immunocompromised, multifocal Candida colonization. Micafungin 100 mg IV daily (echinocandin — first-line for invasive candidiasis per IDSA guidelines). Fluconazole 400–800 mg IV daily (non-critically ill, susceptible Candida species — not for C. glabrata or C. krusei). De-escalate from echinocandin to fluconazole once species identified and susceptible, patient clinically improved.
De-Escalation & Procalcitonin
De-escalation: Narrow antibiotics at 48–72 hours based on culture results and clinical improvement. Broad empiric coverage is a bridge to directed therapy, not a destination. Procalcitonin (PCT): PCT-guided antibiotic discontinuation is supported by multiple RCTs (ProACT, PRORATA, SAPS) and reduces antibiotic exposure without increasing mortality. Algorithm: PCT < 0.25 ng/mL or ≥ 80% decrease from peak suggests bacterial infection resolved — consider stopping antibiotics. Do not use PCT to decide whether to START antibiotics in a critically ill patient (empiric antibiotics should not be withheld while awaiting PCT results).
23 Electrolyte & Acid-Base Emergencies
Hyperkalemia
ECG changes (in order of severity): peaked T waves → prolonged PR → wide QRS → sine wave → VF/asystole
Step 1 — Cardiac membrane stabilization: Calcium gluconate 1–3 g IV over 2–5 min (or calcium chloride 1 g via CVC — 3× more elemental calcium); onset 1–3 min; repeat if ECG changes persist
Step 2 — Intracellular K+ shift: Regular insulin 10 units IV + dextrose 25–50 g IV (50 mL D50W); onset 15–30 min; duration 4–6 h. Also: albuterol 10–20 mg nebulized (additive effect); sodium bicarbonate 50–100 mEq IV (if concurrent acidosis, limited K+ shifting alone)
Step 3 — K+ elimination: Sodium polystyrene sulfonate (Kayexalate) 15–30 g PO/PR (onset 1–6 h, limited evidence, risk of bowel necrosis); patiromer or sodium zirconium cyclosilicate (newer, safer GI binders); loop diuretics (furosemide 40–80 mg IV); hemodialysis (definitive, for refractory/severe hyperkalemia or K+ > 6.5 with ECG changes)
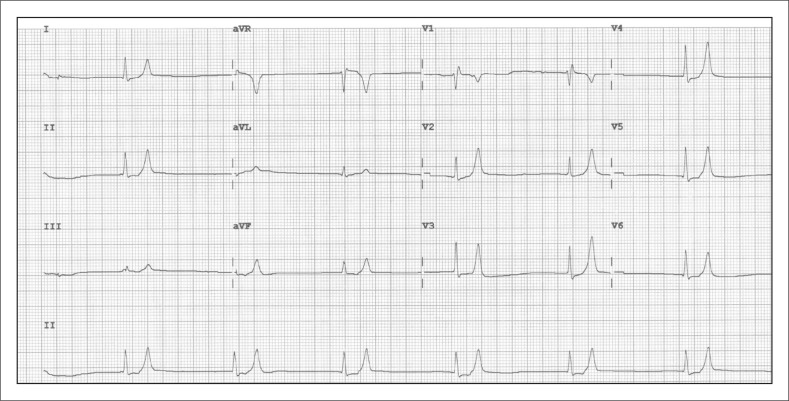
Severe Hyponatremia
Symptomatic hyponatremia (seizures, obtundation, respiratory arrest) requires urgent correction with 3% hypertonic saline: 100–150 mL IV bolus over 10–20 min; may repeat up to 3 times if symptoms persist. Target: increase Na by 4–6 mEq/L in first 1–2 hours (enough to stop symptoms). Total correction must not exceed 10 mEq/L in 24 hours (8 mEq/L in patients at high risk for osmotic demyelination syndrome: hyponatremia > 48 h, chronic alcoholism, malnutrition, hypokalemia, liver disease). If over-corrected: administer DDAVP 2 mcg IV q8h + D5W infusion to re-lower sodium ("rescue therapy").
Metabolic Acidosis — Systematic Approach
| Step | Calculation | Interpretation |
|---|---|---|
| 1. Identify acidosis | pH < 7.35 with low HCO3− | Metabolic acidosis present |
| 2. Calculate AG | AG = Na+ − (Cl− + HCO3−); correct for albumin | AG > 12 = AGMA; AG normal = non-AG (hyperchloremic) |
| 3. Delta-delta | ΔAG / ΔHCO3− | < 1 = concurrent non-AG acidosis; > 2 = concurrent metabolic alkalosis |
| 4. Winter's formula | Expected PaCO2 = 1.5 × HCO3− + 8 (± 2) | Compare to actual PaCO2: higher = concurrent respiratory acidosis; lower = concurrent respiratory alkalosis |
| 5. Osmolar gap (if AGMA) | Measured Osm − (2Na + Glu/18 + BUN/2.8) | > 10 suggests toxic alcohols (methanol, ethylene glycol) |
Non-AG metabolic acidosis causes (HARDUPS): Hyperalimentation/Hyperchloremia, Acetazolamide/Addison disease, RTA (types 1, 2, 4), Diarrhea, Uretero-enterostomy, Pancreatic fistula/drainage, Saline (large-volume NS resuscitation).
Severe Hypocalcemia
Ionized calcium < 0.8 mmol/L (symptomatic: perioral tingling, carpopedal spasm, Chvostek sign, Trousseau sign, QT prolongation, seizures, laryngospasm, cardiac arrest). Treatment: calcium gluconate 1–2 g IV over 10–20 min (via CVC: calcium chloride 1 g), followed by calcium infusion. Common ICU causes: massive transfusion (citrate chelation), pancreatitis, renal failure, post-parathyroidectomy (hungry bone syndrome), sepsis. Always check and correct hypomagnesemia concurrently (magnesium is required for PTH secretion and action).
Severe Hypomagnesemia
Mg < 1.0 mg/dL: refractory hypokalemia (Mg is required for renal K+ retention via Na-K-ATPase), refractory hypocalcemia, arrhythmias (torsades de pointes), seizures. Treatment: magnesium sulfate 2–4 g IV over 15–60 min (for torsades: 2 g IV push); maintenance 1–2 g IV q4–6h. Oral repletion for asymptomatic mild deficiency: magnesium oxide 400–800 mg PO daily. In ICU patients, target Mg ≥ 2.0 mg/dL.
Severe Hypernatremia
Na > 160 mEq/L: lethargy, irritability, seizures, intracranial hemorrhage (brain shrinkage). Correct slowly: lower Na by no more than 10 mEq/L per 24 hours (risk of cerebral edema with rapid correction). Use free water (D5W IV or free water via NG tube). Calculate free water deficit: 0.6 × weight (kg) × [(Na/140) − 1]. Address underlying cause: diabetes insipidus (central — DDAVP; nephrogenic — remove offending drug, thiazide + amiloride), insensible losses, osmotic diuresis.
Severe Metabolic Alkalosis
pH > 7.55 with HCO3− > 40: lethargy, arrhythmias, seizures, impaired oxygen unloading (left-shifted oxyhemoglobin curve). Classify as chloride-responsive (urine Cl < 25 mEq/L: vomiting, NG suction, diuretics — treat with NS and KCl) vs. chloride-resistant (urine Cl > 40 mEq/L: hyperaldosteronism, Cushing, severe hypokalemia — treat underlying cause). For severe refractory alkalosis: acetazolamide 250–500 mg IV (promotes bicarbonaturia); very rarely, HCl infusion via CVC (0.1–0.2 N, calculate deficit and infuse slowly); dialysis with low-bicarbonate bath.
24 Central Lines & Arterial Access
Central Venous Catheter (CVC) Placement
Site selection:
| Site | Advantages | Disadvantages | Key Anatomy |
|---|---|---|---|
| Internal Jugular (IJ) | Most common; reliable US landmarks; compressible; lower PTX risk than subclavian | Uncomfortable (neck); higher infection rate than subclavian; limits neck mobility | Within SCM triangle; lateral to carotid artery; needle entry at apex of SCM heads aimed toward ipsilateral nipple |
| Subclavian | Lowest infection rate (CLABSI); most comfortable; stable position | Highest PTX risk; non-compressible (cannot apply direct pressure for bleeding); contraindicated in coagulopathy; risk of subclavian stenosis (avoid in dialysis patients) | Needle enters inferior to clavicle at junction of medial and middle third, aimed toward sternal notch |
| Femoral | Easiest landmark identification; no PTX risk; compressible; useful during CPR | Highest infection rate; DVT risk; limits mobility; cannot measure CVP accurately | Medial to femoral artery (VAN mnemonic: vein, artery, nerve lateral to medial); below inguinal ligament |
Seldinger Technique
(1) Identify vein with ultrasound (real-time guidance is standard of care — reduces complications and improves first-pass success); (2) puncture vein with introducer needle under US visualization; (3) advance guidewire through needle (J-tip first, monitor for arrhythmia); (4) remove needle, keeping wire in place; (5) make skin nick with scalpel; (6) advance dilator over wire, then remove; (7) thread catheter over wire to appropriate depth; (8) remove wire; (9) aspirate and flush all ports; (10) secure catheter and apply sterile dressing.
Catheter depth: Right IJ/subclavian: 15–17 cm (tip at cavoatrial junction); Left IJ/subclavian: 17–20 cm; Femoral: 20–25 cm. Confirmation: CXR (tip should be at or just above the cavoatrial junction, approximately at the carina level; rule out PTX). Venous blood gas from the distal port (confirms venous placement if PaO2 is venous range).
Arterial Line Placement
Preferred site: radial artery (non-dominant hand, perform Allen test). Alternative: femoral artery (larger, easier in shock/vasoconstriction, but higher infection risk). Technique: US-guided or palpation-guided, 20G catheter, Seldinger (wire-through-needle) or over-the-needle. After placement, connect to pressure transducer zeroed at phlebostatic axis, confirm pulsatile waveform, perform a fast-flush (square wave) test to assess system damping.
Complications
CVC: pneumothorax (subclavian > IJ), arterial puncture, air embolism, arrhythmia (wire in RV), CLABSI, DVT/thrombosis, catheter malposition. Arterial line: thrombosis, infection, hemorrhage (accidental disconnection), pseudoaneurysm, distal ischemia (rare with Allen test), nerve injury.
Ultrasound-Guided Vascular Access
Real-time ultrasound guidance for CVC insertion is the standard of care and is strongly recommended by multiple professional societies (ASA, AIUM, SIR). Benefits: higher first-pass success rate, fewer needle passes, reduced arterial puncture rate, reduced pneumothorax, faster insertion time. Technique: use a high-frequency linear transducer (5–12 MHz) with sterile sheath and gel. Identify the vein (compressible, non-pulsatile, thin-walled) vs. artery (non-compressible, pulsatile, thick-walled). Short-axis (out-of-plane): transverse view, needle appears as a bright dot — easier to center on vessel but cannot see needle tip depth. Long-axis (in-plane): longitudinal view, entire needle shaft and tip visible — better depth control but harder to maintain alignment. Many operators use a combined approach: short-axis for initial puncture, confirm wire in vessel with long-axis or contrast flush. Always confirm venous placement (blood color, pressure waveform, ultrasound visualization of wire in vein) before dilation.
Intraosseous (IO) Access
IO access provides rapid vascular access when IV access cannot be established within 90 seconds or after 2 failed attempts in critical situations. Sites: proximal tibia (most common — 2 cm below tibial tuberosity, flat medial surface), humeral head (faster flow rates, preferred in adults), distal tibia (medial malleolus), distal femur (pediatric). Devices: EZ-IO power driver (most widely used), manual IO needles. All IV medications can be administered IO, including vasopressors, blood products, and anesthetics. Flush with 10 mL NS bolus to prime the marrow space (may cause brief pain in conscious patients — lidocaine 40 mg IO can reduce this). Remove within 24 hours; complications: extravasation, compartment syndrome (rare), osteomyelitis (rare), fracture, fat embolism.
25 Chest Tubes & Thoracic Procedures
Indications
Pneumothorax: symptomatic, large (> 2 cm apex-to-cupola), tension, or requiring positive pressure ventilation. Hemothorax: initial output > 1500 mL (consider thoracotomy) or ongoing output > 200 mL/h for 2–4 h. Empyema: infected pleural fluid (pH < 7.2, glucose < 60, LDH > 1000, positive Gram stain/culture). Malignant effusion: symptomatic, recurrent. Post-operative: cardiac/thoracic surgery.
Tube Size & Insertion
| Indication | Tube Size (French) | Insertion Site |
|---|---|---|
| Pneumothorax (simple) | 14–22 Fr (pigtail catheter often sufficient) | 4th–5th ICS, anterior axillary line (safe triangle) |
| Hemothorax | 28–36 Fr (large bore for blood evacuation) | 4th–5th ICS, anterior to mid-axillary line |
| Empyema | 28–36 Fr (need to drain thick fluid) | Image-guided placement preferred |
| Effusion (serous) | 12–20 Fr (pigtail adequate) | Image-guided, posterior approach for dependent fluid |
Insertion technique (safe triangle method): Patient upright or lateral decubitus; identify the safe triangle (bordered by lateral edge of pectoralis major, lateral edge of latissimus dorsi, 5th ICS). Prep and drape, local anesthetic to skin, periosteum, and pleura. Incision at the intercostal space, blunt dissection with a clamp over the TOP of the rib (avoid the intercostal neurovascular bundle on the inferior rib margin), puncture the parietal pleura, finger sweep to confirm pleural space, insert tube and direct posteriorly (effusion) or anteriorly/apically (pneumothorax). Secure with suture, connect to drainage system, obtain CXR.
Management
Water seal: Chamber with 2 cm of water acts as one-way valve. Air bubbling = air leak (persistent > 5–7 days may indicate bronchopleural fistula). Suction: −20 cmH2O is standard; used for hemothorax, empyema, or persistent air leak. Removal criteria: lung re-expanded on CXR, no air leak for 24 h (clamp trial optional), output < 200 mL/24 h (for effusions/hemothorax), clinical stability. Remove at end-expiration or during Valsalva to prevent air entry; apply occlusive dressing immediately.
Needle Decompression
For tension pneumothorax: 14–16G angiocatheter, 2nd intercostal space midclavicular line (traditional) or 4th–5th ICS anterior axillary line (preferred in obese/muscular patients — shorter chest wall distance). A rush of air confirms tension. Follow immediately with chest tube placement. Note: commercial decompression needles (3.25–5 inch) are more reliable than standard 1.5-inch angiocatheters, which may be too short in obese patients.
26 ICU Bundles & Prevention
VAP Prevention Bundle
(1) Elevate head of bed 30–45°; (2) Daily sedation interruption + assessment of readiness to extubate; (3) DVT prophylaxis; (4) Stress ulcer prophylaxis; (5) Oral care with chlorhexidine 0.12% q12h (some guidelines have moved away from CHG oral care due to concerns about increased mortality in certain studies — institution-specific); (6) Subglottic secretion drainage (specialized ETT); (7) Minimize sedation depth; (8) Early mobilization.
CLABSI Prevention Bundle
(1) Hand hygiene; (2) Full barrier precautions during insertion (cap, mask, sterile gown, sterile gloves, large sterile drape); (3) Chlorhexidine skin antisepsis; (4) Optimal site selection (subclavian preferred for lowest infection rate); (5) Daily review of line necessity with prompt removal when no longer needed. Additional: chlorhexidine-impregnated dressings, antimicrobial-coated catheters if institutional CLABSI rate remains elevated despite bundle compliance.
CAUTI Prevention Bundle
(1) Avoid unnecessary urinary catheters (nurse-driven insertion criteria); (2) Insert using aseptic technique; (3) Maintain closed drainage system; (4) Daily assessment of continued need with prompt removal; (5) Use alternatives when possible (condom catheter, intermittent catheterization). Catheter-associated UTI is the most common HAI; most are preventable with bundle adherence.
Stress Ulcer Prophylaxis (SUP)
Indicated in ICU patients with risk factors: mechanical ventilation > 48 hours, coagulopathy (INR > 1.5, platelets < 50,000), history of GI bleed within 1 year, TBI, burns > 35% TBSA, high-dose corticosteroids, 2+ minor risk factors (sepsis, ICU stay > 1 week, occult GI bleed). Agent: pantoprazole 40 mg IV daily (PPIs preferred over H2RAs based on lower bleeding rates; however, the SUP-ICU trial showed no benefit of pantoprazole vs. placebo in overall ICU mortality). PPI risks: C. difficile infection, pneumonia (gastric acid suppression and bacterial overgrowth). Discontinue SUP when risk factors resolve (e.g., extubation).
DVT Prophylaxis
All ICU patients should receive DVT prophylaxis unless contraindicated: Low-molecular-weight heparin (enoxaparin 40 mg SQ daily) or unfractionated heparin (5000 units SQ q8–12h). LMWH is preferred (slightly more effective). Mechanical prophylaxis (intermittent pneumatic compression devices) for patients with contraindications to pharmacologic prophylaxis (active bleeding, severe thrombocytopenia, recent intracranial surgery). Combine pharmacologic + mechanical in highest-risk patients (major trauma, spinal cord injury).
Glycemic Control
Target blood glucose 140–180 mg/dL in most critically ill patients. The NICE-SUGAR trial demonstrated that intensive glucose control (81–108 mg/dL) increased mortality compared with conventional control (140–180) due to severe hypoglycemia. Use continuous IV insulin infusion with standardized protocol and frequent glucose monitoring (q1–2h). Avoid hypoglycemia (< 70 mg/dL) aggressively — associated with increased mortality, arrhythmias, and neurological injury.
Early Mobility
ICU-acquired weakness (ICUAW) affects up to 50% of patients ventilated ≥ 7 days. Early mobility (physical and occupational therapy starting within 48–72 hours of ICU admission) reduces delirium, ventilator days, ICU and hospital length of stay, and improves functional outcomes at discharge. Safety criteria for mobilization: hemodynamically stable (MAP > 60, no active vasopressor titration), adequate oxygenation (SpO2 > 88%, FiO2 ≤ 0.6), no active procedures planned. Progressive mobility: passive ROM → active ROM in bed → seated edge of bed → standing → ambulation.
Temperature Management
Targeted temperature management (TTM) after cardiac arrest: the TTM2 trial (2021) showed no benefit of hypothermia (33°C) vs. normothermia (37.5°C target with active fever prevention) in out-of-hospital cardiac arrest. Current AHA 2023 guidelines recommend actively preventing fever (target ≤ 37.5°C) for at least 72 hours in comatose patients after ROSC. Avoid hyperthermia (> 37.7°C) aggressively in all neurologically injured ICU patients (TBI, stroke, post-cardiac arrest). Fever in the ICU: Evaluate for infection (cultures, imaging) but also consider non-infectious causes: drug fever, transfusion reaction, DVT/PE, adrenal insufficiency, post-operative/post-procedural, CNS injury, malignant hyperthermia, neuroleptic malignant syndrome. Antipyretics: acetaminophen 1 g IV/PO q6h; external cooling (cooling blankets, intravascular cooling devices) for refractory fever or when targeting specific temperature ranges.
Family Engagement & Open Visitation
The ABCDEF bundle includes Family engagement (F) as a core element. Open visitation policies (flexible hours, family presence during rounds, family participation in care) improve patient and family satisfaction, reduce delirium, and do not increase infection rates. Family members should be invited to participate in rounding, goal-setting, and shared decision-making. Structured communication at each shift change (nurse-to-family update) reduces family anxiety. Provide written materials about the ICU environment, common equipment, and what to expect.
27 Withdrawal of Care & Brain Death
Brain Death Determination
Brain death is the irreversible cessation of all functions of the entire brain, including the brainstem. It is a clinical diagnosis. Legal death is declared at the time of the brain death examination completion. Requirements vary by state/institution but generally follow the AAN 2010 guidelines (updated 2023):
Prerequisites (must be met before testing):
(1) Known etiology sufficient to cause brain death
(2) Exclusion of confounders: no residual sedatives/paralytics (5 half-lives or drug levels below therapeutic), core temperature ≥ 36°C, SBP ≥ 100 mmHg, no severe electrolyte/endocrine/acid-base derangements
(3) Neuroimaging confirms catastrophic brain injury consistent with clinical exam
Clinical Examination (all required):
(1) Coma (no eye opening or motor response to central pain — nail bed pressure, sternal rub, supraorbital pressure; spinal reflexes may persist)
(2) Absent brainstem reflexes: no pupillary light reflex (mid-position or dilated, fixed), no corneal reflex, no oculocephalic reflex (doll's eyes), no oculovestibular reflex (cold calorics — 50 mL ice water, wait 1 min each ear), no gag reflex, no cough reflex to tracheal suctioning
(3) Absent respiratory drive (apnea test)
Apnea Test Protocol:
Pre-oxygenate with 100% FiO2 for 10 min. Disconnect ventilator. Deliver O2 via tracheal catheter at 6 L/min (passive oxygenation). Observe for respiratory effort for 8–10 minutes. Draw ABG at end. Test is positive (supports brain death) if: PaCO2 ≥ 60 mmHg AND ≥ 20 mmHg above baseline with NO respiratory effort. Abort if SBP < 90, SpO2 < 85%, or cardiac arrhythmia.
Ancillary tests (used when clinical exam or apnea test cannot be completed): cerebral angiography (gold standard — absent intracranial filling), radionuclide cerebral perfusion scan (absent uptake — "hollow skull sign"), EEG (electrocerebral silence), transcranial Doppler (reverberating/oscillating flow or absent diastolic flow). Most institutions require 1 examination by an attending physician (some require 2 exams separated by a defined observation period, especially in pediatrics).
Comfort Care / Terminal Extubation
When a decision is made to withdraw life-sustaining treatment: (1) Discontinue vasopressors, (2) Reduce FiO2 and PEEP, (3) Administer morphine 2–10 mg IV bolus then infusion (titrate to comfort, not respiratory rate — the goal is symptom management, not hastening death), (4) Glycopyrrolate 0.2 mg IV for secretions ("death rattle"), (5) Remove the ETT or transition to T-piece, (6) Ensure family at bedside if desired, (7) Pronounce death when cardiac activity ceases. Time from withdrawal to death is variable (minutes to days); prepare family for this uncertainty.
Organ Donation
Contact the organ procurement organization (OPO) for every imminent death per federal law (CMS Conditions of Participation). The OPO determines medical suitability and approaches the family. Donation after brain death (DBD) and donation after circulatory death (DCD) are both established pathways. The treating physician should NOT be the one to discuss donation with the family — trained OPO coordinators are most effective. Maintain hemodynamic and hormonal support (vasopressin, thyroid hormone, methylprednisolone) in brain-dead donors to preserve organ function.
28 Prognostication & Family Communication
APACHE II Score
The Acute Physiology and Chronic Health Evaluation II (APACHE II) score is calculated from 12 physiologic variables (worst values in first 24 hours of ICU admission: temperature, MAP, HR, RR, oxygenation, arterial pH, sodium, potassium, creatinine, hematocrit, WBC, GCS) + age points + chronic health points. Score range: 0–71. Higher score = higher predicted hospital mortality. APACHE II is used for ICU benchmarking and research stratification; it should NOT be used to make individual patient care decisions. APACHE IV is the current version with better calibration.
SOFA Trending for Prognostication
Serial SOFA scores are more prognostically useful than a single admission score. A rising SOFA score over the first 48–96 hours predicts mortality > 50%. A declining SOFA score suggests improving organ function and better prognosis. Delta SOFA (change from admission to 48 hours) is an independent predictor of ICU mortality. Each 1-point increase in SOFA is associated with ~10% increase in mortality.
Family Meetings — Structured Approach
Before the meeting: Review chart thoroughly; identify decision-maker (healthcare proxy/POA); invite key team members (attending, nurse, social work, chaplain, palliative care if appropriate); reserve a private, quiet room
During the meeting (VALUE framework):
V — Value family statements (acknowledge emotions, validate concerns)
A — Acknowledge emotions ("I can see this is very difficult")
L — Listen (let family speak; clinicians should listen more than talk; aim for ≥ 50% family speaking time)
U — Understand the patient as a person ("What would your loved one say if they could speak for themselves?")
E — Elicit questions ("What questions do you have for us?")
Key principles: Avoid medical jargon; use consistent, honest language; provide a clear clinical picture and trajectory; frame goals in terms of the patient's values and wishes; offer a recommendation when appropriate ("based on what you've told us about your mother's wishes, I would recommend..."); schedule follow-up meeting
Shared Decision-Making
Shared decision-making is the preferred model for ICU decision-making. The physician provides medical information (prognosis, treatment options, burdens/benefits), and the surrogate provides information about the patient's values, preferences, and goals. Together they reach a decision that is consistent with the patient's wishes. This is distinct from paternalistic decision-making (physician decides) and pure autonomy (family decides without guidance). Most families want a physician recommendation — withholding one when asked is not patient-centered.
Palliative Care Integration
Palliative care should be integrated early in the ICU stay, not reserved for end-of-life. Triggers for palliative care consultation: ICU stay > 7–10 days, multisystem organ failure, metastatic malignancy, recurrent ICU admissions, disagreement about goals of care, patient/family request, predicted mortality > 50%. Early palliative care consultation is associated with shorter ICU length of stay, reduced non-beneficial treatments, and improved family satisfaction without increasing mortality.
29 Scoring Systems Master Table
SOFA Score
See Section 4 for the fully enumerated SOFA score table with all six organ systems and scoring criteria (0–4 points each, total 0–24).
qSOFA
| Criterion | Points |
|---|---|
| Respiratory rate ≥ 22/min | 1 |
| Altered mentation (GCS < 15) | 1 |
| Systolic BP ≤ 100 mmHg | 1 |
Score ≥ 2 → evaluate for sepsis (SOFA); associated with in-hospital mortality ≥ 10% in infection.
APACHE II
12 acute physiology variables (worst in first 24 h) + age points (0–6) + chronic health points (0–5). Total 0–71. Score ≥ 25 predicts > 50% hospital mortality. See Section 28 for full description.
GCS (Glasgow Coma Scale)
See Section 19 for fully enumerated table. Range 3–15 (Eye 1–4 + Verbal 1–5 + Motor 1–6).
RASS (Richmond Agitation-Sedation Scale)
See Section 21 for fully enumerated table. Range +4 (combative) to −5 (unarousable). Target RASS 0 to −2 for most ICU patients.
CAM-ICU (Confusion Assessment Method for the ICU)
| Feature | Assessment | Positive If |
|---|---|---|
| 1. Acute onset or fluctuating course | Is there an acute change in mental status from baseline? Does mental status fluctuate during the shift? | Yes to either |
| 2. Inattention | Squeeze my hand when I say the letter "A" — read "S-A-V-E-A-H-A-A-R-T" | ≥ 2 errors |
| 3. Altered level of consciousness | Current RASS score | RASS ≠ 0 |
| 4. Disorganized thinking | Standardized questions (e.g., "Will a stone float on water?") + command ("Hold up this many fingers") | ≥ 1 error in questions OR unable to follow command |
CAM-ICU positive = Feature 1 + Feature 2 + (Feature 3 OR Feature 4) = Delirium present.
KDIGO AKI Staging
See Section 13. Stages 1–3 based on creatinine rise and/or urine output criteria.
Berlin ARDS Classification
See Section 9. Mild (P/F 201–300), Moderate (P/F 101–200), Severe (P/F ≤ 100), all on PEEP ≥ 5.
SCAI Cardiogenic Shock Classification
See Section 6. Stages A (At Risk) through E (Extremis).
West Haven Hepatic Encephalopathy
See Section 14. Grades I–IV from mild cognitive changes to coma.
4T Score (HIT)
See Section 16. Categories: Thrombocytopenia, Timing, Thrombosis, oTher causes. Score 0–8; ≤ 3 = low, 4–5 = intermediate, 6–8 = high probability.
ISTH DIC Score
See Section 16. Components: platelet count, D-dimer, PT prolongation, fibrinogen. Score ≥ 5 = overt DIC.