Hospitalist Medicine
Admission and discharge workflows, inpatient diabetes and anticoagulation management, DVT prophylaxis, hospital-acquired infections, transitions of care, observation status, and every protocol, scoring system, and management algorithm across the full scope of hospital medicine.
01 Admission & History Framework
Hospital medicine (hospitalist medicine) is the specialty devoted to the care of acutely ill patients from the moment of admission through discharge. The hospitalist performs the initial history and physical examination, writes admission orders, coordinates specialist consultations, manages day-to-day inpatient care, and orchestrates a safe transition back to the outpatient setting. A structured, systematic approach to admission prevents errors, ensures all safety measures are addressed, and satisfies documentation requirements for medical necessity.
The Complete Admission H&P
The admission history and physical examination (H&P) is the cornerstone document of the hospitalization. It establishes the chief complaint, constructs a differential diagnosis, documents the patient’s baseline status, and justifies the level of care. A thorough H&P includes: Chief Complaint (in the patient’s own words), History of Present Illness (onset, location, duration, character, aggravating/alleviating factors, radiation, timing, severity — OLDCARTS), Past Medical/Surgical History, Medications (with doses and frequency — critical for reconciliation), Allergies (with reaction type), Family History, Social History (tobacco, alcohol, drugs, living situation, functional status, advance directives), Review of Systems (10-system minimum for comprehensive), and the Physical Exam (vitals plus organ-system examination).
Admission Orders — ADC VANDALISM Mnemonic
The ADC VANDALISM mnemonic ensures no critical admission order is overlooked. Each letter maps to an essential order category:
| Letter | Category | Details |
|---|---|---|
| A | Admit to | Service (hospitalist, medicine, telemetry), attending physician name, level of care (floor, stepdown, ICU) |
| D | Diagnosis | Admitting diagnosis (drives medical necessity documentation) |
| C | Condition | Stable, guarded, serious, critical |
| V | Vitals | Frequency (q4h routine, q1h if unstable), call parameters (SBP <90 or >180, HR <50 or >120, RR >24, O₂ sat <92%, temp >38.3°C, UOP <30 mL/h) |
| A | Activity | Bedrest, bedrest with bathroom privileges, up ad lib, fall precautions, restraints |
| N | Nursing | Daily weights, I&Os, wound care, Foley care, neuro checks, fall risk protocol, skin assessment |
| D | Diet | Regular, cardiac (2 g Na), diabetic/carb-controlled, renal, NPO (for procedures), clear liquids, thickened liquids (dysphagia) |
| A | Allergies | Document all with reaction type (anaphylaxis, rash, GI upset) — enters allergy banner |
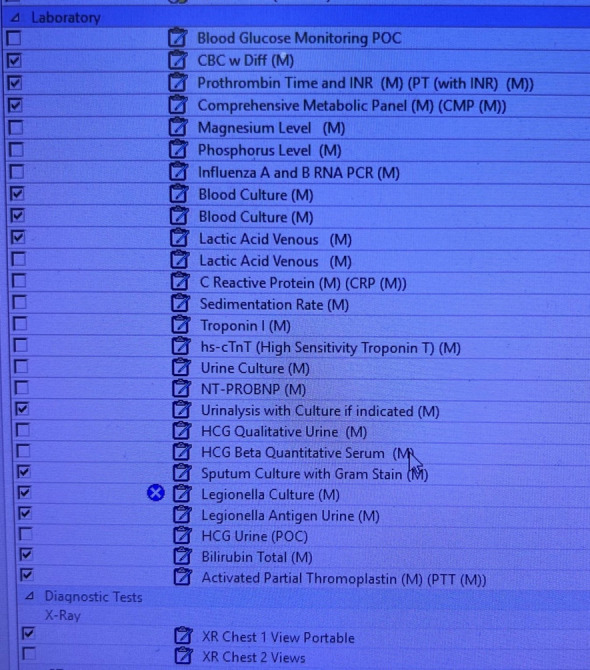
| L | Labs | Admission labs (CBC, CMP, Mg, Phos, coags, troponin, BNP, UA, blood cultures if febrile), AM labs, serial troponins |
| I | IV fluids | Type (NS, LR, D5W), rate, bolus vs maintenance; LR preferred in most situations (lower hyperchloremic acidosis risk vs NS) |
| S | Special studies | Imaging (CXR, CT, US), EKG, echocardiogram, telemetry monitoring, continuous pulse oximetry |
| M | Medications | Home medications (reconciled), new medications, VTE prophylaxis, DVT prophylaxis, stress ulcer prophylaxis if indicated, pain management, PRN medications (acetaminophen, ondansetron, melatonin) |
Level of Care Determination
Choosing the appropriate level of care is a critical admission decision. General medical floor is appropriate for stable patients requiring IV medications, monitoring, or workup. Telemetry/stepdown is needed for patients requiring continuous cardiac monitoring (new arrhythmia, chest pain with elevated troponin, heart failure, electrolyte abnormalities with ECG changes) or more frequent nursing assessments (q2h). Intensive care unit (ICU) is indicated for hemodynamic instability requiring vasopressors, respiratory failure requiring mechanical ventilation or high-flow oxygen >15 L/min, acute MI with complications, status epilepticus, or any condition requiring continuous 1:1 nursing.
Code Status Discussion
Code status must be addressed at every admission. Patients should be asked whether they have an advance directive, a healthcare power of attorney (HCPOA), or a POLST/MOLST form. Document the discussion and the patient’s stated preferences: Full Code (CPR, intubation, all interventions), DNR/DNI (do not resuscitate / do not intubate — clarify what interventions are desired: pressors, ICU transfer, BiPAP), or Comfort Measures Only (symptom management only, no life-prolonging treatments). Code status does not equal goals of care — a DNR patient may still desire aggressive treatment of reversible conditions.
02 Hospital Systems & Quality Metrics
Modern hospitalists operate within a complex quality and regulatory framework. Understanding these metrics is essential because they directly influence hospital reimbursement, public reporting, accreditation, and patient safety.
CMS Core Measures
The Centers for Medicare & Medicaid Services (CMS) mandates reporting on condition-specific quality measures. Key hospitalist-relevant core measures include: Pneumonia — blood cultures before first antibiotic dose, appropriate initial antibiotic selection, antibiotic administration within 6 hours (previously 4 hours for sepsis); Heart Failure — documentation of LVEF, ACEi/ARB/ARNI at discharge for HFrEF, discharge instructions including weight monitoring, activity, diet, medication list, follow-up; VTE — VTE prophylaxis within 24 hours of admission, anticoagulation overlap for warfarin initiation; Sepsis (SEP-1) — 3-hour bundle (lactate, blood cultures, broad-spectrum antibiotics) and 6-hour bundle (repeat lactate if initial >2.0 mmol/L, 30 mL/kg crystalloid for hypotension/lactate ≥4, vasopressors if refractory).
Value-Based Purchasing (VBP)
CMS withholds a percentage of DRG payments (currently 2%) and redistributes based on hospital performance across four domains: Clinical Outcomes (30-day mortality for AMI, HF, pneumonia, COPD, CABG — 25% weight), Safety (HAI measures, PSI-90 composite — 25%), Efficiency and Cost Reduction (Medicare spending per beneficiary — 25%), and Person and Community Engagement (HCAHPS — 25%). Hospitals scoring above the median earn back more than the 2%, while poor performers lose revenue.
Hospital Readmission Reduction Program (HRRP)
CMS penalizes hospitals with excess 30-day all-cause readmission rates for six conditions: AMI, heart failure, pneumonia, COPD, THA/TKA, and CABG. Penalties can reach up to 3% of total Medicare reimbursement. The LACE index (Length of stay, Acuity of admission, Comorbidities via Charlson index, Emergency department visits in prior 6 months) predicts 30-day readmission risk and helps target discharge interventions to high-risk patients.
HCAHPS (Hospital Consumer Assessment of Healthcare Providers and Systems)
HCAHPS is the standardized patient experience survey. Key domains include: communication with doctors, communication with nurses, responsiveness of hospital staff, pain management, communication about medications, discharge information, cleanliness and quietness of hospital environment, care transition (3 questions). Scores are publicly reported on Hospital Compare and directly affect VBP reimbursement.
Leapfrog Group
The Leapfrog Hospital Safety Grade (A through F) evaluates hospitals on 28 patient safety measures including falls, infections, medication errors, and ICU physician staffing. Leapfrog also surveys computerized physician order entry (CPOE) adoption, evidence-based hospital referral, and ICU staffing by board-certified intensivists.
Patient Safety Indicators (PSIs)
AHRQ Patient Safety Indicators are a set of measures using administrative/billing data to screen for potentially preventable complications. PSI-90 is the composite used in VBP. Key individual PSIs include: PSI-03 (pressure ulcer rate), PSI-06 (iatrogenic pneumothorax), PSI-08 (postoperative hip fracture from falls), PSI-09 (perioperative hemorrhage/hematoma), PSI-11 (postoperative respiratory failure), PSI-12 (perioperative PE/DVT), PSI-13 (postoperative sepsis), PSI-14 (postoperative wound dehiscence), PSI-15 (unrecognized abdominopelvic accidental puncture/laceration).
CMS does not reimburse hospitals for certain hospital-acquired conditions (HACs): CAUTI, CLABSI, surgical site infection after CABG/bariatric/orthopedic procedures, falls with injury, pressure injuries stage III/IV, air embolism, blood incompatibility, wrong-site/wrong-patient surgery, foreign object retained after surgery, and manifestations of poor glycemic control (DKA, hypoglycemia). Preventing these conditions is a core hospitalist responsibility.
03 Key Terminology & Abbreviations
Hospital medicine uses abbreviations drawn from every medical and surgical subspecialty, plus administrative and regulatory terminology unique to the inpatient setting. Mastery of this vocabulary is essential for efficient chart review, order writing, and communication.
| Term | Definition |
|---|---|
| LOS | Length of stay — number of midnights the patient occupies a hospital bed |
| ADT | Admission, discharge, transfer — the hospital tracking system |
| DRG | Diagnosis-Related Group — CMS payment classification based on principal diagnosis, procedures, complications, age, and discharge status |
| CMI | Case mix index — average DRG weight; reflects patient complexity and drives hospital revenue |
| CDI | Clinical documentation improvement — queries from coders to clarify diagnoses for accurate DRG assignment |
| UR / UM | Utilization review / utilization management — ensures patients meet inpatient criteria |
| InterQual / Milliman | Proprietary evidence-based criteria sets for determining admission status and level of care |
| 2-Midnight Rule | Medicare guideline: hospital stays expected to span at least 2 midnights qualify for inpatient; shorter stays are observation |
| RAC | Recovery Audit Contractor — audits Medicare claims for overpayment, frequently targets inpatient vs observation status |
| SNF / LTAC / IRF | Skilled nursing facility / long-term acute care hospital / inpatient rehabilitation facility |
| ALOS / GMLOS | Average length of stay / geometric mean length of stay (for a given DRG) |
| CC / MCC | Complication or comorbidity / major complication or comorbidity — diagnoses that increase DRG severity and reimbursement |
| POA | Present on admission — indicator distinguishing conditions present at admission from hospital-acquired conditions |
| HAC | Hospital-acquired condition — CMS does not provide additional payment for these |
| AMA | Against medical advice — patient-initiated discharge before the care team recommends |
| I&O | Intake and output — fluid balance monitoring |
| PRN | Pro re nata — as needed |
| NPO | Nil per os — nothing by mouth |
| BMP / CMP | Basic / comprehensive metabolic panel |
| VTE / DVT / PE | Venous thromboembolism / deep vein thrombosis / pulmonary embolism |
| SCD | Sequential compression device — mechanical VTE prophylaxis |
04 Admission Workflow & Order Sets
The admission process is a high-risk transition that demands a systematic approach. Errors in admission orders — missed VTE prophylaxis, incorrect medication doses, inappropriate diet — can cause preventable harm. A standardized workflow reduces variability and improves outcomes.
Admission Criteria
A patient requires inpatient admission when outpatient management is insufficient due to: severity of illness (hemodynamic instability, acute organ failure, active hemorrhage), need for intensive monitoring or nursing (telemetry, q1–2h vitals, 1:1 observation), need for treatments available only in the hospital (IV medications, blood products, emergent procedures), or inability to perform activities of daily living safely (altered mental status, inability to ambulate, no safe discharge plan). The clinical decision must be supported by documentation of medical necessity — why the patient cannot be safely managed at a lower level of care.
Observation vs Inpatient — Initial Determination
At admission, the hospitalist must determine whether the patient meets criteria for inpatient admission or should be placed in observation status. The 2-midnight rule (CMS, effective 2013) states: if the admitting physician expects the patient to require hospital care spanning at least 2 midnights, inpatient admission is generally appropriate. If the expected stay is <2 midnights, the patient should be placed in observation status unless a procedure is on the Inpatient-Only list. InterQual and Milliman criteria provide evidence-based clinical benchmarks for this determination.
Initial Order Set Components
A comprehensive admission order set addresses every element of ADC VANDALISM and includes condition-specific protocols. Common order sets include: General medicine admission, Chest pain / ACS (serial troponins q3h ×3, telemetry, aspirin, heparin), Heart failure (daily weights, 2L fluid restriction, I&Os, BNP, BMP, Mg), Pneumonia (blood cultures ×2, sputum culture, Legionella/pneumococcal urine antigens, appropriate antibiotics), Sepsis (3-hour bundle: lactate, cultures, antibiotics, 30 mL/kg crystalloid), DKA (insulin drip, q1h BG, q2h BMP, AG monitoring, fluid resuscitation protocol).
Telemetry should be ordered for: ACS/chest pain with elevated troponin, new or unstable arrhythmias, heart failure with active diuresis, syncope of suspected cardiac origin, electrolyte abnormalities with ECG changes (K <3.0 or >6.0, Mg <1.0, Ca <7.0), initiation of QT-prolonging medications, post-procedural monitoring (cardioversion, pacemaker placement, cardiac catheterization), acute stroke, and severe sepsis/septic shock. Telemetry is not indicated for low-risk chest pain (HEART score ≤3, serial troponins negative), stable chronic conditions, or as a substitute for nursing assessment.
Medication Reconciliation on Admission
The hospitalist must obtain the best possible medication history (BPMH) from multiple sources: patient/family interview, outpatient pharmacy records, EMR medication list, and pill bottles. Each home medication is continued, held, or modified with documentation of rationale. Common medications held on admission include: oral hypoglycemics (NPO status, insulin preferred inpatient), metformin (contrast studies, AKI risk), ACEi/ARBs (acute kidney injury, hypotension), diuretics (if volume depleted), anticoagulants (procedural planning), and anti-hypertensives (if hypotensive).
05 Discharge Planning & Transitions of Care
Discharge planning begins at admission. Effective transitions of care reduce 30-day readmission rates, prevent medication errors, and improve patient satisfaction. Approximately 20% of Medicare patients are readmitted within 30 days, costing the healthcare system billions annually.
Discharge Readiness Criteria
A patient is ready for discharge when: the acute condition is stabilized or resolving, the patient can tolerate oral medications and nutrition, vital signs are stable and trending toward normal for ≥24 hours, no pending critical test results, the patient (or caregiver) demonstrates understanding of the care plan, a safe discharge destination has been identified, and follow-up appointments are arranged. Specific criteria vary by condition — for heart failure: net negative on diuresis, weight at or near dry weight, stable on oral diuretics for ≥24 hours, oxygen saturation ≥92% on room air or baseline O₂ requirements.
IDEAL Discharge Framework
The AHRQ IDEAL framework guides patient-centered discharge education:
| Letter | Component | Implementation |
|---|---|---|
| I | Include the patient and family | Engage patient and caregiver in all discharge decisions and teach-back |
| D | Discuss with the patient | Explain diagnosis, prognosis, what to expect at home, activity restrictions |
| E | Educate the patient | Medication changes (new, stopped, changed), warning signs requiring return to ED |
| A | Assess how well clinicians educate | Use teach-back method: "Tell me in your own words what you should do if..." |
| L | Listen to and honor the patient | Address barriers (health literacy, language, transportation, cost of medications) |
Discharge Summary Elements
The Joint Commission requires the discharge summary to include: reason for hospitalization, significant findings (labs, imaging, procedures), procedures performed, hospital course, condition at discharge, discharge diagnosis (principal and secondary), discharge medications (with changes from admission highlighted), follow-up instructions (appointments, labs), pending results, and patient education. The summary must be completed within 30 days but ideally within 24–48 hours to be available for the outpatient follow-up visit.

LACE Index for Readmission Risk
| Component | Points |
|---|---|
| Length of stay: 1 d = 1, 2 d = 2, 3 d = 3, 4–6 d = 4, 7–13 d = 5, ≥14 d = 7 | 1–7 |
| Acuity of admission: emergency = 3, urgent = 2, elective = 0 | 0–3 |
| Comorbidities (Charlson index): 0 = 0, 1 = 1, 2 = 2, 3 = 3, ≥4 = 5 | 0–5 |
| Emergency department visits in prior 6 months: 0 = 0, 1 = 1, 2 = 2, 3 = 3, ≥4 = 4 | 0–4 |
Total score range: 0–19. Score ≥10 indicates high readmission risk; these patients should receive intensified discharge planning including post-discharge phone calls within 48–72 hours, early outpatient follow-up (within 7 days), and referral to transitional care programs.
Post-Acute Care Disposition
Home: independent with ADLs, safe environment, caregiver available if needed. Home with home health: needs skilled nursing visits (wound care, IV antibiotics, physical therapy) but is otherwise safe at home. Skilled nursing facility (SNF): requires 24-hour nursing supervision, skilled care (IV medications, wound care, PT/OT), and a qualifying 3-midnight inpatient stay for Medicare Part A coverage. Inpatient rehabilitation facility (IRF): requires intensive rehabilitation (minimum 3 hours/day, 5 days/week), must be able to participate actively; common for stroke, hip fracture, major joint replacement. Long-term acute care hospital (LTAC): for patients requiring prolonged hospital-level care (ventilator weaning, complex wound care, IV antibiotics >14 days) with average LOS ≥25 days.
06 Handoff Communication — I-PASS & SBAR
Communication failures during care transitions are the leading root cause of sentinel events. Standardized handoff tools reduce preventable adverse events by 23–30% (I-PASS study, NEJM 2014). Every patient handoff — shift change, service transfer, cross-cover — must be structured, verbal, and accompanied by a written or electronic signout.
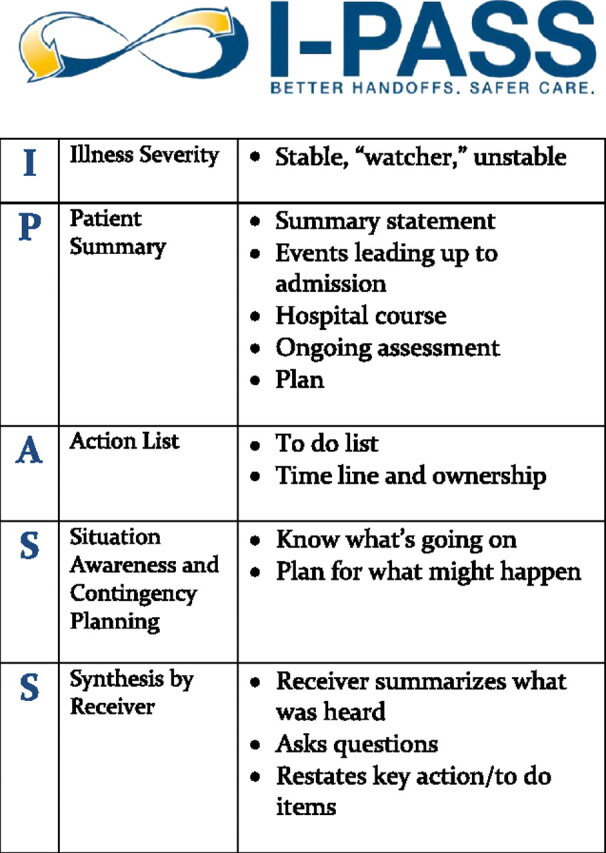
I-PASS Framework
| Component | Full Name | Content |
|---|---|---|
| I | Illness Severity | Stable, "watcher" (may decompensate), or unstable. Sets the recipient’s mental model for level of vigilance. |
| P | Patient Summary | One-liner: age, sex, chief complaint, relevant history, hospital course, current status. Example: "72M with HFrEF (EF 25%), admitted for acute decompensated heart failure, now net negative 3L on IV furosemide." |
| A | Action List | To-do items: pending labs (AM BMP, repeat lactate), tasks (titrate diuretic if UOP <30 mL/h), medications due (next dose of vancomycin at 2200), and scheduled events (CT at 0600). |
| S | Situation Awareness & Contingency Planning | What to watch for and what to do: "If K >5.5 on AM labs, hold spironolactone and give kayexalate. If SBP <90, hold carvedilol and give 250 mL NS bolus." This is the most critical component for overnight safety. |
| S | Synthesis by Receiver | The receiving physician reads back the key elements: illness severity, primary concern, action items. This closed-loop communication catches errors and omissions. |
SBAR Communication
SBAR is the standard framework for nurse-to-physician (or any inter-professional) communication about acute changes:
| Component | Content | Example |
|---|---|---|
| Situation | What is happening right now | "I’m calling about Mr. Smith in room 412 — his blood pressure has dropped to 78/50." |
| Background | Relevant clinical context | "He was admitted yesterday for pneumonia. He has a history of CHF. He received his evening dose of lisinopril 2 hours ago." |
| Assessment | What you think is going on | "I think he may be septic — he’s also tachycardic at 112 and his temp is 39.1°C." |
| Recommendation | What you think should be done | "I would like to give a 500 mL NS bolus and get a lactate and blood cultures. Would you like to come evaluate?" |

Signout Best Practices
Written signout should be updated daily and include: patient name, room, age, one-liner, code status, allergies, key medications (anticoagulants, insulin, pressors), active issues, anticipated problems overnight, and contact information for the primary team. Verbal signout should occur face-to-face in a quiet environment with minimal interruptions. The outgoing team should explicitly identify "watchers" — patients at highest risk for overnight decompensation — and provide specific contingency plans. Cross-cover physicians should round on watchers early in the shift.
07 Observation vs Inpatient Status
The distinction between observation and inpatient status is one of the most consequential administrative decisions in hospital medicine. It affects hospital reimbursement, patient cost-sharing, and eligibility for post-acute care benefits. Hospitalists must understand the regulatory framework and its clinical and financial implications.
Medicare 2-Midnight Rule
Under the 2-midnight rule (CMS-1599-F, effective October 2013), hospital stays are presumed appropriate for inpatient admission if the admitting physician expects the patient to require medically necessary hospital services spanning at least 2 midnights. The physician must document: (1) the reason the patient requires hospital care, (2) the expected length of stay, and (3) the plan of care. Stays expected to be <2 midnights are generally observation unless the patient undergoes a procedure on the Inpatient-Only (IPO) list or an exception applies (e.g., patient dies or is transferred before the second midnight, or the clinical documentation supports a rare, unforeseen circumstance).
Recovery Audit Contractor (RAC) Audits
RACs review Medicare claims to identify overpayments. Short inpatient stays (<2 midnights) are prime targets. If a RAC determines a stay did not meet inpatient criteria, the hospital must repay the full DRG payment and may re-bill under observation (at a significantly lower rate). Hospitalists can protect their institution by documenting medical necessity thoroughly, using clinical criteria (InterQual/Milliman) to support decisions, and converting to observation proactively when criteria are not met.
Financial Implications for Patients
The distinction carries major financial consequences: Inpatient: covered under Medicare Part A (hospital deductible of $1,632 in 2024, then $0 copay for days 1–60). Observation: covered under Medicare Part B (20% coinsurance for all services, self-administered drugs not covered — patients may be billed full price for home medications like atorvastatin, metformin, or lisinopril given during observation). Most critically, observation days do not count toward the 3-midnight qualifying inpatient stay required for Medicare Part A coverage of skilled nursing facility (SNF) care.
Self-Administered Drug List
Under observation status, Medicare Part B does not cover self-administered drugs — medications the patient could normally take themselves. This includes most oral medications (antihypertensives, statins, oral hypoglycemics, anticoagulants). The hospital may charge the patient directly. IV medications, nebulized treatments, and injections administered by nursing staff are generally covered under Part B. Some hospitals have policies absorbing these costs; others pass them to the patient.
Condition Code 44
Condition Code 44 allows a hospital to change a patient’s status from inpatient to outpatient/observation during the stay (before discharge) when utilization review determines that inpatient criteria are not met. Requirements: the physician must concur with the status change, UR must approve, and the patient must be notified (Medicare Outpatient Observation Notice — MOON). Condition Code 44 cannot be applied retroactively after discharge. When applied correctly, it allows re-billing under Part B observation, preserving at least partial reimbursement rather than risking a RAC denial and full repayment.
As of March 2017, hospitals must provide written notice (the MOON) to Medicare beneficiaries who have been in observation status for more than 24 hours. The notice explains their outpatient status, the reason, and the implications for cost-sharing and SNF eligibility. It must be delivered within 36 hours of the patient being placed in observation and signed by the patient or representative.
08 Community-Acquired Pneumonia
Community-acquired pneumonia (CAP) is the most common infectious cause of hospitalization and the leading infectious cause of death in the United States, with approximately 1.5 million hospitalizations and 50,000 deaths annually. The hospitalist must rapidly assess severity, initiate appropriate empiric therapy, and de-escalate based on culture data.
CURB-65 Severity Score
| Criterion | Points |
|---|---|
| Confusion (new-onset, AMT ≤8) | 1 |
| Urea (BUN >20 mg/dL or >7 mmol/L) | 1 |
| Respiratory rate ≥30/min | 1 |
| Blood pressure (SBP <90 or DBP ≤60 mmHg) | 1 |
| Age ≥65 | 1 |
Interpretation: 0–1 = low risk, consider outpatient treatment (30-day mortality ~1.5%); 2 = moderate risk, consider short hospitalization or closely supervised outpatient (mortality ~9%); 3–5 = high risk, hospitalize; score 4–5 consider ICU (mortality ~22%).
PSI/PORT Score
The Pneumonia Severity Index (PSI) uses 20 variables to stratify patients into Risk Classes I–V. Class I = age <50, no comorbidities, normal vitals (outpatient). Class II = ≤70 points (mortality 0.6%). Class III = 71–90 points (mortality 0.9%, brief observation). Class IV = 91–130 points (mortality 9.3%, inpatient). Class V = >130 points (mortality 27%, inpatient/ICU). Points are assigned for: age (years for men, years minus 10 for women), nursing home residence (+10), neoplastic disease (+30), liver disease (+20), CHF (+10), cerebrovascular disease (+10), renal disease (+10), altered mental status (+20), RR ≥30 (+20), SBP <90 (+20), temp <35 or ≥40 (+15), HR ≥125 (+10), pH <7.35 (+30), BUN ≥30 (+20), Na <130 (+20), glucose ≥250 (+10), hematocrit <30 (+10), PaO₂ <60 (+10), pleural effusion (+10).
Empiric Antibiotic Therapy
| Setting | Regimen | Notes |
|---|---|---|
| Outpatient, no comorbidities | Amoxicillin 1 g PO TID × 5 days or doxycycline 100 mg PO BID × 5 days | Azithromycin only if local resistance <25% |
| Outpatient, with comorbidities | Amoxicillin-clavulanate 875/125 PO BID + azithromycin 500 mg day 1 then 250 mg daily or respiratory FQ (levofloxacin 750 mg daily) | Comorbidities: COPD, DM, CKD, CHF, alcoholism, asplenia |
| Inpatient (non-ICU) | Ceftriaxone 1–2 g IV daily + azithromycin 500 mg IV/PO daily or respiratory FQ monotherapy (levofloxacin 750 mg IV daily) | Standard 5–7 day course; de-escalate to PO when improving and tolerating oral |
| ICU | Ceftriaxone 2 g IV daily + azithromycin 500 mg IV daily or ceftriaxone + respiratory FQ | If Pseudomonas risk: piperacillin-tazobactam 4.5 g IV q6h or cefepime 2 g IV q8h + FQ |
| MRSA risk factors | Add vancomycin 15–20 mg/kg IV q8–12h (target trough 15–20 mcg/mL or AUC/MIC 400–600) or linezolid 600 mg IV/PO q12h | Risk factors: prior MRSA, recent hospitalization, hemodialysis, injection drug use, necrotizing/cavitary pneumonia |
De-escalation & Duration
Switch from IV to PO antibiotics when the patient is: clinically improving (defervescing, decreasing WBC), hemodynamically stable, tolerating oral intake, and has a functioning GI tract. Minimum 5 days of therapy is standard per ATS/IDSA 2019 guidelines. Extend to 7 days for complicated infections (empyema, lung abscess, Pseudomonas, S. aureus bacteremia). Procalcitonin levels <0.25 ng/mL or decreasing by ≥80% from peak support antibiotic discontinuation.
09 Acute Heart Failure
Acute decompensated heart failure (ADHF) accounts for over 1 million hospitalizations annually in the US and is the leading cause of 30-day readmission. The hospitalist manages the acute episode, optimizes guideline-directed medical therapy (GDMT), and coordinates a transition designed to prevent readmission.
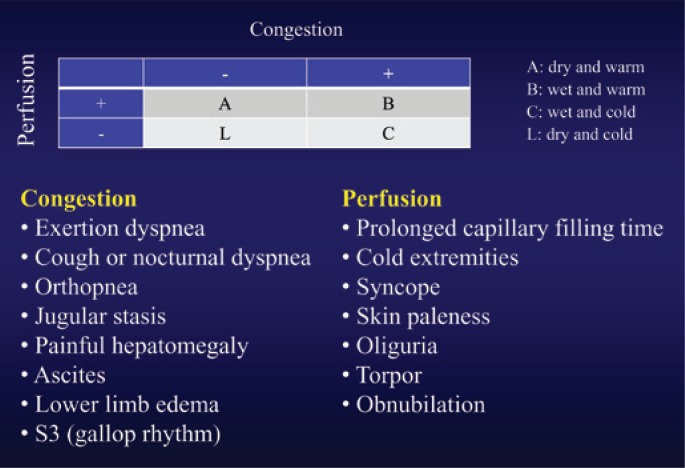
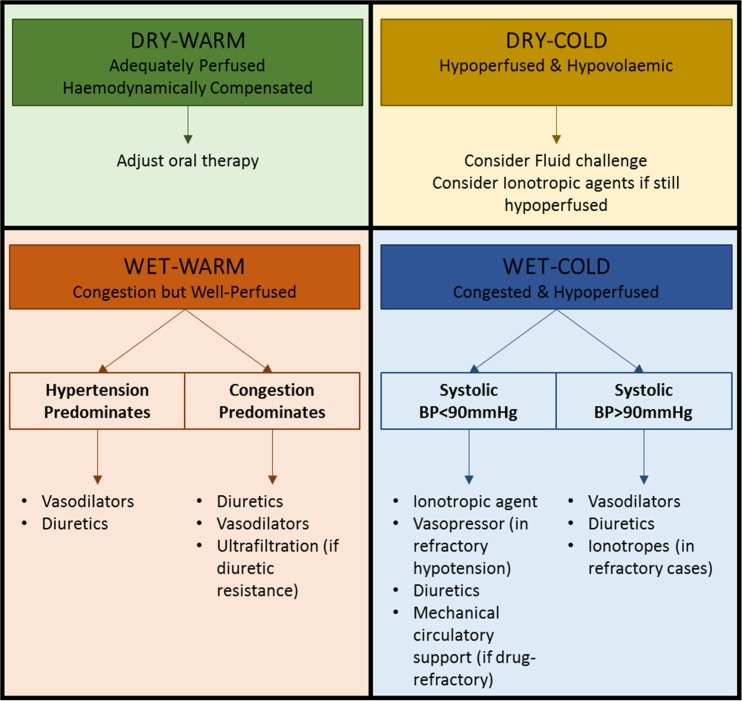
Stevenson Hemodynamic Profiles
The Stevenson classification uses bedside assessment of congestion (wet vs dry) and perfusion (warm vs cold) to guide therapy:
| Profile | Congestion | Perfusion | Clinical Features | Treatment |
|---|---|---|---|---|
| Warm & Dry (A) | No | Adequate | Compensated HF, no edema, adequate UOP, warm extremities | Optimize oral GDMT, discharge when stable |
| Warm & Wet (B) | Yes | Adequate | Most common presentation (~70%); JVD, rales, edema, adequate BP and UOP | IV diuretics (furosemide); may add vasodilators (nitroglycerin) |
| Cold & Dry (L) | No | Poor | Hypotension, narrow pulse pressure, cool extremities, poor UOP, no congestion | Cautious IV fluids (250 mL bolus); inotropes if no response (dobutamine, milrinone) |
| Cold & Wet (C) | Yes | Poor | Worst prognosis; cardiogenic shock with congestion | Inotropes + diuretics; may need vasopressors; consider mechanical circulatory support |
Diuretic Strategies
IV furosemide is first-line. Initial dose: give the patient’s total daily oral furosemide dose as IV (2.5:1 oral-to-IV conversion is approximately equivalent, but practice is to give the same number — e.g., 40 mg PO at home = start 40 mg IV). If diuretic-naive, start furosemide 20–40 mg IV. Administer as bolus q12h or continuous infusion (no mortality difference per DOSE trial, but infusion may cause less ototoxicity). Goal: net negative 1–2 L/day (urine output >100–150 mL/h in first 2 hours after dose). If inadequate response after first dose, double the dose (max single bolus 200 mg IV). If still refractory: add metolazone 2.5–5 mg PO 30 minutes before furosemide (sequential nephron blockade — metolazone blocks distal tubule sodium reabsorption). Alternatively, add chlorothiazide 500 mg IV or acetazolamide 500 mg IV (ADVOR trial showed benefit of acetazolamide added to loop diuretic for faster decongestion).
BNP/NT-proBNP Trending
BNP (>100 pg/mL suggests HF; >400 strongly supports) and NT-proBNP (>300 pg/mL for age <50; >900 for age 50–75; >1800 for age >75) are useful for diagnosis and monitoring. A ≥30% decrease in BNP from admission to discharge is associated with lower readmission risk. Persistently elevated or rising BNP despite treatment indicates inadequate decongestion or worsening HF.
GDMT Optimization Before Discharge
Hospitalization is an opportunity to initiate or uptitrate GDMT for HFrEF (EF ≤40%). The four pillars: ARNI (sacubitril-valsartan, target 97/103 mg BID) or ACEi/ARB, beta-blocker (carvedilol, metoprolol succinate, or bisoprolol — initiate only when euvolemic and stable, do not start or uptitrate in acute decompensation), MRA (spironolactone 25–50 mg daily or eplerenone 25–50 mg daily — check K <5.0, eGFR >30), and SGLT2 inhibitor (dapagliflozin 10 mg or empagliflozin 10 mg daily — can initiate in-hospital per EMPULSE trial). Additional therapies: hydralazine-isosorbide dinitrate (for Black patients or those intolerant of ACEi/ARB), ivabradine (HR ≥70 on max beta-blocker), and loop diuretic for persistent congestion.
10 COPD Exacerbation
Acute exacerbations of COPD (AECOPD) are defined by worsening dyspnea, increased sputum volume, and/or increased sputum purulence beyond normal day-to-day variation. AECOPD accounts for ~700,000 hospitalizations per year in the US and carries in-hospital mortality of 2–4%, with 1-year post-hospitalization mortality reaching 23%.
GOLD Classification of COPD Severity
| GOLD Stage | Post-Bronchodilator FEV1 | Severity |
|---|---|---|
| GOLD 1 | FEV1 ≥80% predicted | Mild |
| GOLD 2 | 50% ≤ FEV1 <80% predicted | Moderate |
| GOLD 3 | 30% ≤ FEV1 <50% predicted | Severe |
| GOLD 4 | FEV1 <30% predicted | Very severe |
ABG Interpretation in AECOPD
Baseline COPD patients are chronic CO₂ retainers with compensated respiratory acidosis (elevated PaCO₂ 50–60 mmHg with normal pH due to renal HCO₃ retention). During acute exacerbation, PaCO₂ rises further and pH drops (acute-on-chronic respiratory acidosis). A pH <7.35 with PaCO₂ >45 mmHg indicates acute ventilatory failure and is the threshold for initiating non-invasive ventilation. For every 10 mmHg acute rise in PaCO₂, pH drops by ~0.08. For every 10 mmHg chronic rise, HCO₃ increases by ~3.5 mEq/L. Comparing expected vs actual HCO₃ helps determine the chronicity of the respiratory acidosis.
Inpatient Management
Bronchodilators: Albuterol 2.5 mg nebulized q2–4h (or MDI 4–8 puffs q1–4h with spacer) plus ipratropium 0.5 mg nebulized q6h (or combination DuoNeb). Systemic corticosteroids: Prednisone 40 mg PO daily × 5 days (REDUCE trial showed 5-day course equivalent to 14-day; IV methylprednisolone 125 mg q6–12h if unable to take PO, then transition to oral). Antibiotics: indicated when 2 of 3 Anthonisen criteria are met (increased dyspnea, increased sputum volume, increased sputum purulence) or when mechanical ventilation is required. First-line: azithromycin 500 mg day 1, then 250 mg × 4 days; alternatives: doxycycline 100 mg BID × 5 days, amoxicillin-clavulanate 875/125 BID × 5–7 days, or levofloxacin 750 mg daily × 5 days (reserved for complicated cases due to resistance concerns).
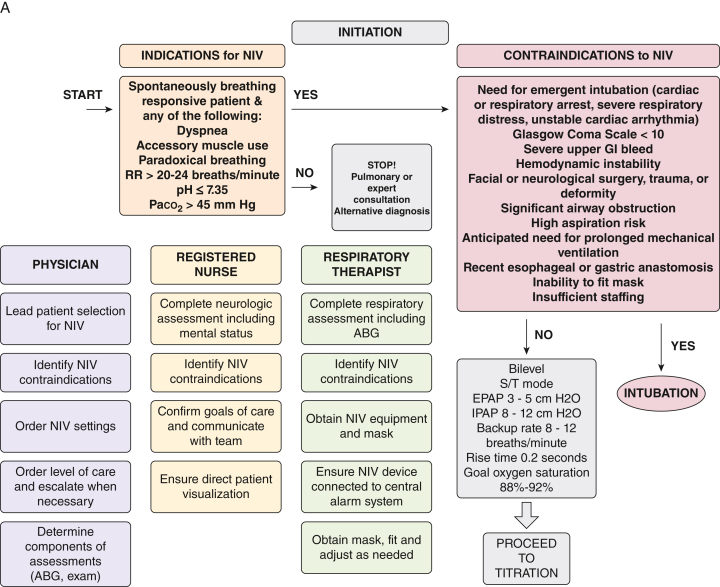
Non-Invasive Ventilation (NIV)
BiPAP is the cornerstone of management for acute respiratory acidosis in AECOPD. Indications: pH <7.35 and PaCO₂ >45 mmHg, severe dyspnea with accessory muscle use, or persistent hypoxemia despite supplemental O₂. Initial settings: IPAP 10–12 cmH₂O, EPAP 4–5 cmH₂O, titrate IPAP up by 2 cmH₂O every 15–30 minutes targeting tidal volume 6–8 mL/kg and improved pH/PaCO₂. Maximum IPAP typically 20–25 cmH₂O. FiO₂ titrated to SpO₂ 88–92% (avoid hyperoxia in CO₂ retainers — suppresses hypoxic respiratory drive). BiPAP reduces intubation rates by ~65% and mortality by ~50% compared to standard therapy alone (Cochrane 2017).
Oxygen Therapy
Target SpO₂ 88–92% in COPD patients (GOLD guidelines). The Haldane effect means that excessive supplemental O₂ in chronic CO₂ retainers can worsen hypercapnia by three mechanisms: (1) loss of hypoxic pulmonary vasoconstriction increasing V/Q mismatch, (2) reduced hypoxic ventilatory drive, and (3) the Haldane effect itself (increased O₂ displaces CO₂ from hemoglobin). Use Venturi masks for precise FiO₂ delivery (24–50%) rather than nasal cannula in the acute setting.
11 Acute Kidney Injury
Acute kidney injury (AKI) occurs in 10–15% of all hospitalized patients and up to 50% of ICU patients. AKI is independently associated with increased mortality, prolonged LOS, progression to CKD, and higher healthcare costs. The hospitalist must rapidly classify the injury, identify the cause, and initiate targeted management.
KDIGO Staging
| Stage | Serum Creatinine Criteria | Urine Output Criteria | Approximate Mortality |
|---|---|---|---|
| Stage 1 | Rise ≥0.3 mg/dL within 48 h or 1.5–1.9× baseline within 7 days | <0.5 mL/kg/h for 6–12 h | ~15% |
| Stage 2 | 2.0–2.9× baseline | <0.5 mL/kg/h for ≥12 h | ~25% |
| Stage 3 | ≥3.0× baseline or creatinine ≥4.0 mg/dL with acute rise ≥0.5 or initiation of RRT | <0.3 mL/kg/h for ≥24 h or anuria for ≥12 h | ~35% |
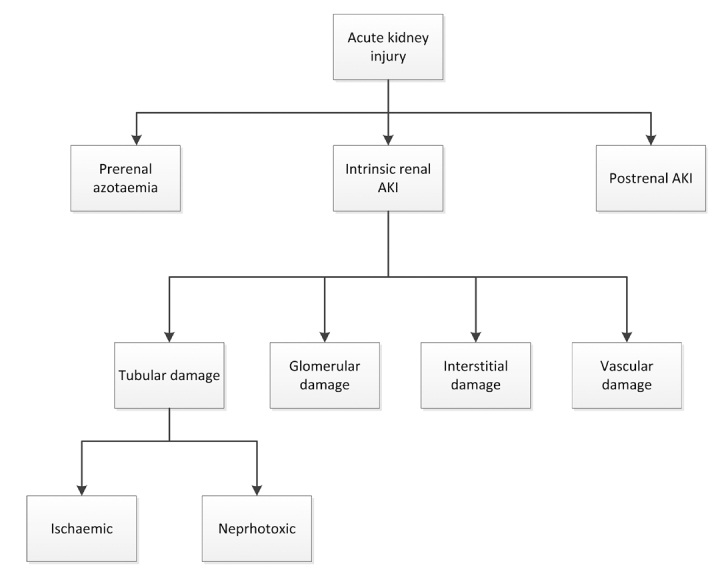
Prerenal vs Intrinsic vs Postrenal
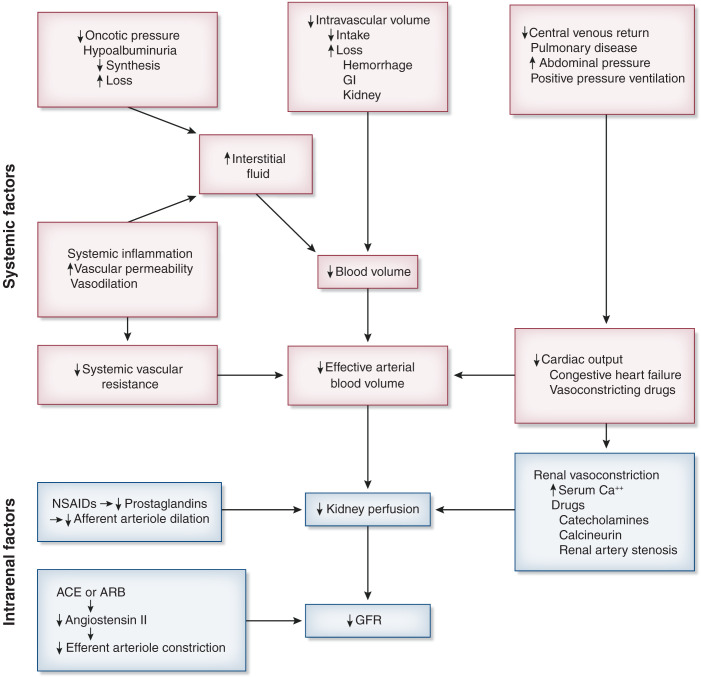
Prerenal (60–70% of hospital AKI): decreased renal perfusion from hypovolemia (hemorrhage, dehydration, GI losses), hypotension (sepsis, cardiogenic shock), or altered renal hemodynamics (NSAIDs constrict afferent arteriole; ACEi/ARBs dilate efferent arteriole). Labs: BUN:Cr ratio >20:1, FENa <1%, FEUrea <35%, urine osmolality >500, bland sediment. Treatment: volume repletion with isotonic crystalloid (LR preferred), hold offending agents.
Intrinsic (25–35%): damage to the tubules, interstitium, glomeruli, or vasculature. Acute tubular necrosis (ATN) is the most common intrinsic cause — from prolonged ischemia or nephrotoxins (aminoglycosides, contrast, myoglobin). Labs: FENa >2%, FEUrea >50%, muddy brown granular casts on UA. Acute interstitial nephritis (AIN): drug-induced (NSAIDs, PPIs, antibiotics — especially beta-lactams), presents with fever, rash, eosinophilia, WBC casts, eosinophiluria. Glomerulonephritis: RBC casts, proteinuria, dysmorphic RBCs; send complement levels, ANA, ANCA, anti-GBM, hepatitis panel.
Postrenal (5–10%): obstruction. Bilateral ureteral obstruction, bladder outlet obstruction (BPH most common in older men), or obstruction of a solitary kidney. Diagnosis: renal ultrasound showing hydronephrosis. Treatment: relieve obstruction (Foley catheter for bladder outlet, ureteral stent or nephrostomy tube for ureteral obstruction). Post-obstructive diuresis can be massive (UOP >200 mL/h) — replace half of urine output with 0.45% NS to prevent dehydration.
FENa and FEUrea Calculations
FENa = (UNa × PCr) / (PNa × UCr) × 100. Prerenal: <1%; ATN: >2%. FEUrea = (UUrea × PCr) / (PUrea × UCr) × 100. Prerenal: <35%; ATN: >50%. FEUrea is more reliable in patients on diuretics (diuretics increase sodium excretion, making FENa falsely elevated in prerenal AKI).
Renal Replacement Therapy Indications — AEIOU
| Letter | Indication | Details |
|---|---|---|
| A | Acidosis | Severe metabolic acidosis (pH <7.1) refractory to bicarbonate therapy |
| E | Electrolytes | Refractory hyperkalemia (K >6.5 with ECG changes despite medical therapy) |
| I | Intoxication | Dialyzable toxins: methanol, ethylene glycol, lithium, salicylates |
| O | Overload | Volume overload refractory to diuretics (pulmonary edema despite max diuresis) |
| U | Uremia | Uremic symptoms: pericarditis, encephalopathy, bleeding diathesis (typically BUN >80–100 mg/dL) |
12 Cellulitis & Skin/Soft Tissue Infections
Skin and soft tissue infections (SSTIs) are among the most common reasons for hospitalization. The hospitalist must distinguish purulent from non-purulent infections, classify severity, identify necrotizing infections early, and select appropriate antibiotic therapy.
Purulent vs Non-Purulent SSTIs
Purulent SSTIs (abscess, furuncle, carbuncle) are most commonly caused by Staphylococcus aureus (including MRSA in 50–60% of community-acquired cases). Management centers on incision and drainage (I&D) — antibiotics alone without I&D are often inadequate. Non-purulent SSTIs (cellulitis, erysipelas) are predominantly caused by beta-hemolytic streptococci (Group A Strep). Treatment is antibiotic therapy without I&D. Erysipelas involves the superficial dermis and lymphatics (raised, well-demarcated borders, "orange peel" texture); cellulitis extends deeper into the subcutaneous tissue (ill-defined borders, warm, tender, erythematous).
Severity Classification & Empiric Antibiotics
| Severity | Criteria | Non-Purulent (Cellulitis) | Purulent (Abscess) |
|---|---|---|---|
| Mild | No systemic toxicity, immunocompetent | Cephalexin 500 mg PO QID or dicloxacillin 500 mg PO QID × 5–7 days | I&D alone may suffice; if antibiotics added: TMP-SMX DS 1–2 tabs PO BID or doxycycline 100 mg PO BID |
| Moderate | Systemic signs (fever, tachycardia, WBC >12k) or immunocompromised or failed oral therapy | Cefazolin 1–2 g IV q8h or nafcillin 1–2 g IV q4h | I&D + vancomycin 15–20 mg/kg IV q8–12h or TMP-SMX IV + cephalexin |
| Severe | Sepsis, failed IV antibiotics, suspected deep tissue involvement, immunocompromised | Vancomycin + piperacillin-tazobactam 4.5 g IV q6h (covers MRSA + gram-negatives + anaerobes) | I&D + vancomycin + piperacillin-tazobactam; surgical consultation |
Necrotizing Fasciitis
Necrotizing fasciitis is a surgical emergency with mortality of 20–40% even with treatment. Clinical clues: pain out of proportion to exam findings, rapidly spreading erythema, crepitus (gas in tissues), bullae, skin necrosis, hemodynamic instability, and failure to improve on antibiotics. Type I (polymicrobial — diabetics, postoperative) and Type II (monomicrobial — Group A Strep, most common in healthy patients). Treatment: emergent surgical debridement (definitive therapy — every hour of delay increases mortality), broad-spectrum antibiotics (vancomycin + piperacillin-tazobactam + clindamycin 900 mg IV q8h for toxin inhibition), ICU admission, and serial debridements until clean margins.
LRINEC Score for Necrotizing Fasciitis
| Parameter | Value | Points |
|---|---|---|
| CRP | ≥150 mg/L | 4 |
| WBC (cells/mm³) | 15,000–25,000 / >25,000 | 1 / 2 |
| Hemoglobin (g/dL) | 11–13.5 / <11 | 1 / 2 |
| Sodium (mEq/L) | <135 | 2 |
| Creatinine (mg/dL) | >1.6 | 2 |
| Glucose (mg/dL) | >180 | 1 |
Score ≥6: suspicious for necrotizing fasciitis (PPV 92%); score ≥8: strongly predictive. However, a low score does not exclude necrotizing fasciitis — if clinical suspicion is high, proceed with surgical exploration regardless. The LRINEC score should never delay surgical consultation.
13 Venous Thromboembolism
Venous thromboembolism (VTE), encompassing deep vein thrombosis (DVT) and pulmonary embolism (PE), is a leading cause of preventable hospital death. The hospitalist must be proficient in risk stratification, diagnosis, and anticoagulation management.
Wells Score for DVT
| Criterion | Points |
|---|---|
| Active cancer (treatment within 6 months or palliative) | +1 |
| Paralysis, paresis, or recent cast of lower extremity | +1 |
| Recently bedridden >3 days or major surgery within 12 weeks | +1 |
| Localized tenderness along deep venous system | +1 |
| Entire leg swollen | +1 |
| Calf swelling >3 cm compared to asymptomatic leg | +1 |
| Pitting edema confined to symptomatic leg | +1 |
| Collateral superficial veins (non-varicose) | +1 |
| Previously documented DVT | +1 |
| Alternative diagnosis at least as likely as DVT | −2 |
Score ≤0: low probability (5% prevalence) — check D-dimer, if negative DVT excluded; ≥1–2: moderate (17%) — D-dimer or ultrasound; ≥3: high probability (53%) — proceed directly to compression ultrasound.
Wells Score for PE
| Criterion | Points |
|---|---|
| Clinical signs/symptoms of DVT | +3 |
| PE is the most likely diagnosis | +3 |
| Heart rate >100 | +1.5 |
| Immobilization ≥3 days or surgery in prior 4 weeks | +1.5 |
| Previous PE or DVT | +1.5 |
| Hemoptysis | +1 |
| Active malignancy (treatment within 6 months or palliative) | +1 |
Score ≤4 (unlikely PE): check D-dimer; if negative (<500 ng/mL, or age-adjusted cutoff: age × 10 for patients >50), PE excluded. Score >4 (likely PE): proceed to CT pulmonary angiography (CTPA). If CTPA contraindicated (contrast allergy, CKD), V/Q scan is an alternative.
Anticoagulation for VTE
| Agent | Dose for VTE Treatment | Monitoring | Pearls |
|---|---|---|---|
| Unfractionated heparin (UFH) | 80 units/kg bolus, then 18 units/kg/h infusion | aPTT q6h (target 1.5–2.5× normal, ~60–80 sec) | Use for massive PE, high bleeding risk (short half-life, reversible with protamine), renal failure |
| Enoxaparin (LMWH) | 1 mg/kg SC q12h or 1.5 mg/kg SC daily | Anti-Xa level (target 0.6–1.0 for q12h dosing); check in renal impairment, obesity, pregnancy | Avoid if CrCl <30 (or reduce to 1 mg/kg daily); predictable pharmacokinetics |
| Rivaroxaban | 15 mg PO BID × 21 days, then 20 mg daily | No routine monitoring | Take with food; single-drug approach (no heparin lead-in needed) |
| Apixaban | 10 mg PO BID × 7 days, then 5 mg BID | No routine monitoring | Preferred in renal impairment; single-drug approach; lowest major bleeding rate in trials |
| Edoxaban | 60 mg PO daily (30 mg if CrCl 15–50, wt ≤60 kg, or P-gp inhibitor) | No routine monitoring | Requires 5–10 days of initial heparin/LMWH lead-in |
| Dabigatran | 150 mg PO BID | No routine monitoring (dTT or ecarin time if needed) | Requires 5–10 days of initial heparin/LMWH lead-in; reversible with idarucizumab |
| Warfarin | Start 5 mg daily, adjust to INR 2.0–3.0 | INR (target 2.0–3.0) | Bridge with heparin/LMWH for ≥5 days AND until INR ≥2.0 for ≥24h |
Massive PE — Systemic Thrombolysis
Massive PE is defined by sustained hypotension (SBP <90 mmHg for ≥15 minutes), need for vasopressors, or cardiac arrest. Treatment: alteplase (tPA) 100 mg IV over 2 hours (or 50 mg IV bolus in cardiac arrest), plus heparin anticoagulation. Absolute contraindications: active internal bleeding, recent (<3 months) hemorrhagic stroke, intracranial neoplasm. Relative contraindications: recent surgery <3 weeks, active peptic ulcer, current anticoagulation. Catheter-directed therapy (half-dose tPA via pulmonary artery catheter or mechanical thrombectomy) is an alternative for patients with relative contraindications to systemic lysis.
Submassive PE — PESI Score
Submassive PE = hemodynamically stable but with evidence of RV strain (RV dilation on CT or echo, elevated troponin, elevated BNP). The Pulmonary Embolism Severity Index (PESI) stratifies risk:
| Variable | Points |
|---|---|
| Age (in years) | +age |
| Male sex | +10 |
| Cancer | +30 |
| Heart failure | +10 |
| Chronic lung disease | +10 |
| Heart rate ≥110 | +20 |
| SBP <100 | +30 |
| Respiratory rate ≥30 | +20 |
| Temperature <36°C | +20 |
| Altered mental status | +60 |
| SpO₂ <90% | +20 |
Class I (≤65): very low risk (30-day mortality ~1.6%). Class II (66–85): low risk (3.5%). Class III (86–105): intermediate (7.1%). Class IV (106–125): high (11.4%). Class V (>125): very high (23.9%). sPESI (simplified): 1 point each for age >80, cancer, heart failure/chronic lung disease, HR ≥110, SBP <100, SpO₂ <90%; score 0 = low risk (1.0% mortality), ≥1 = high risk (10.9%).
Duration of Anticoagulation
Provoked VTE (surgery, immobilization, estrogen, pregnancy): 3 months. Unprovoked VTE: at least 3 months, then reassess — extended anticoagulation if bleeding risk is low (apixaban 2.5 mg BID or rivaroxaban 10 mg daily for extended prophylaxis reduce recurrence by ~80%). Cancer-associated VTE: indefinite anticoagulation (LMWH or DOAC preferred over warfarin; edoxaban and rivaroxaban non-inferior to LMWH in trials, but avoid DOACs in GI/GU malignancies due to higher mucosal bleeding). Recurrent unprovoked VTE: indefinite.
14 Inpatient Diabetes Management
Hyperglycemia (BG >140 mg/dL) occurs in up to 40% of hospitalized patients and is associated with increased infection rates, impaired wound healing, longer LOS, and higher mortality. Insulin is the preferred agent for inpatient glycemic management; oral hypoglycemics are generally held during hospitalization due to unpredictable oral intake, variable renal function, and need for tighter control.
Basal-Bolus-Correction Insulin Protocol
The physiologic basal-bolus-correction regimen mimics normal insulin secretion. Total daily dose (TDD) calculation: 0.4–0.5 units/kg/day for most patients; start lower (0.2–0.3 units/kg) for elderly, renal impairment (CrCl <30), or type 1 DM. Divide TDD: 50% as basal insulin (glargine or detemir once daily, or NPH BID) and 50% as prandial insulin (lispro, aspart, or glulisine divided equally before meals). Correction insulin (same rapid-acting) is added to the prandial dose based on a sliding scale (low-dose: 1 unit per 50 mg/dL over target; medium: 1 per 40; high: 1 per 30). Target glucose: 140–180 mg/dL for most patients; 110–140 mg/dL for ICU patients per ADA guidelines.
A sliding scale alone (without basal insulin) is reactive, not proactive — it treats hyperglycemia after it occurs rather than preventing it. The RABBIT 2 trial demonstrated that basal-bolus insulin achieves target glucose in 66% of patients vs only 38% with sliding scale alone, with no increase in hypoglycemia. Sliding scale monotherapy should be avoided; always pair correction doses with scheduled basal (and prandial if eating) insulin.
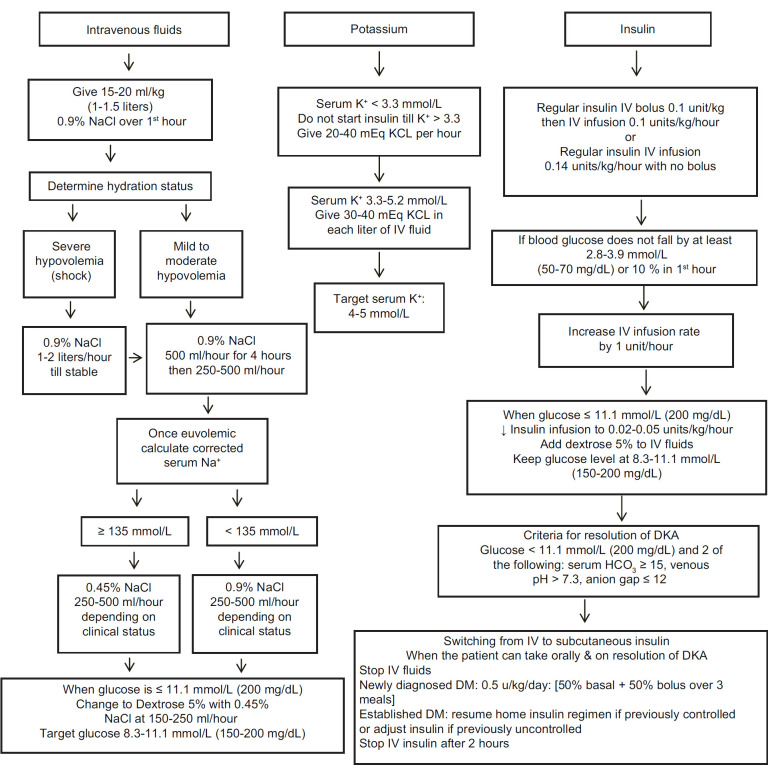
DKA Management
Diabetic ketoacidosis (DKA): glucose usually >250 mg/dL (can be euglycemic with SGLT2 inhibitors), pH <7.3, HCO₃ <18, positive serum ketones, anion gap >12.
Fluids: NS 1–1.5 L/h for the first 1–2 hours (15–20 mL/kg/h), then 250–500 mL/h of NS or 0.45% NS (based on corrected Na). When BG reaches 200–250 mg/dL, switch to D5 0.45% NS to prevent hypoglycemia while continuing insulin to close the anion gap.
Insulin: Regular insulin bolus 0.1 units/kg IV, then continuous infusion at 0.1 units/kg/h. Target BG decline of 50–75 mg/dL per hour. If BG does not drop by ≥10% in the first hour, give another 0.1 units/kg bolus. Transition to subcutaneous insulin when: AG closed (<12), BG <200, pH >7.3, HCO₃ ≥15, patient eating. Overlap IV and SC insulin by 2 hours before discontinuing the drip.
Potassium: Check K before starting insulin. If K <3.3 mEq/L: hold insulin, replace K aggressively (20–40 mEq/h IV). If K 3.3–5.3: add 20–40 mEq KCl to each liter of IV fluids. If K >5.3: hold potassium replacement, recheck in 2 hours. Insulin drives K intracellularly — starting insulin with a K <3.3 can cause fatal hypokalemia and cardiac arrest.
Hyperosmolar Hyperglycemic State (HHS)
HHS: glucose usually >600 mg/dL, serum osmolality >320 mOsm/kg, minimal ketosis, pH >7.3, profound dehydration (average fluid deficit 8–10 L). Treatment: aggressive fluid resuscitation (1–1.5 L NS/h initially, then titrate to urine output 0.5–1 mL/kg/h); low-dose insulin infusion (0.05–0.1 units/kg/h — glucose drops rapidly with fluids alone; avoid rapid osmolality shifts that can cause cerebral edema). Mortality is higher than DKA (10–20% vs 1–5%) due to older patient population and comorbidities.
Hypoglycemia Protocol
Hypoglycemia: BG <70 mg/dL. Mild (BG 54–70, patient alert and able to swallow): 15–20 g oral glucose (4 oz juice, glucose tablets), recheck in 15 minutes, repeat if still <70. Severe (BG <54 or altered mental status/unable to swallow): dextrose 50% (D50) 25 mL (12.5 g) IV push, or glucagon 1 mg IM if no IV access; recheck BG in 15 minutes. For all hypoglycemia events: identify the cause (excess insulin, missed meal, renal failure reducing insulin clearance), adjust regimen (reduce TDD by 20%), and ensure the patient receives a meal or snack.
Perioperative Glucose Management
NPO patients: hold prandial insulin, continue basal insulin at 75–80% of the usual dose (prevents fasting hyperglycemia while reducing hypoglycemia risk). Monitor BG q4–6h. If BG >180: use correction insulin. Resume prandial insulin when eating. Target perioperative glucose 140–180 mg/dL. Hold metformin 48 hours before procedures using IV contrast and for 48 hours after (lactic acidosis risk). Hold SGLT2 inhibitors 3–4 days before surgery (euglycemic DKA risk).
15 VTE Prophylaxis
Venous thromboembolism is the most common preventable cause of hospital death. Every hospitalized patient should undergo VTE risk assessment on admission and receive appropriate prophylaxis. Failure to provide prophylaxis is a never event for surgical patients and a key CMS quality measure.
Padua Prediction Score (Medical Patients)
| Risk Factor | Points |
|---|---|
| Active cancer | 3 |
| Previous VTE (excluding superficial) | 3 |
| Reduced mobility (≥3 days bed rest) | 3 |
| Known thrombophilia | 3 |
| Recent (≤1 month) trauma or surgery | 2 |
| Age ≥70 | 1 |
| Heart and/or respiratory failure | 1 |
| Acute MI or ischemic stroke | 1 |
| Acute infection and/or rheumatic disorder | 1 |
| Obesity (BMI ≥30) | 1 |
| Ongoing hormonal treatment | 1 |
Score ≥4 = high risk (11% VTE rate without prophylaxis) — pharmacologic prophylaxis indicated. Score <4 = low risk (0.3% VTE rate) — early ambulation, consider mechanical prophylaxis.
Caprini Score (Surgical Patients)
The Caprini RAM assigns 1–5 points per risk factor. 1 point each: age 41–60, minor surgery, BMI >25, swollen legs, varicose veins, pregnancy/postpartum, history of unexplained stillbirth, oral contraceptives/HRT, sepsis <1 month, serious lung disease (including pneumonia <1 month), COPD, abnormal pulmonary function, MI, CHF <1 month, inflammatory bowel disease, bed rest. 2 points each: age 61–74, major surgery (>45 min), laparoscopic surgery (>45 min), malignancy, bed rest >72 h, immobilizing cast, central venous access. 3 points each: age ≥75, history of VTE, family history of VTE, Factor V Leiden, prothrombin 20210A, lupus anticoagulant, anticardiolipin antibodies, elevated homocysteine, HIT. 5 points each: stroke <1 month, elective arthroplasty, hip/pelvis/leg fracture, acute spinal cord injury <1 month.
Score 0–1: very low risk (early ambulation). 2: low (mechanical prophylaxis). 3–4: moderate (pharmacologic + mechanical). ≥5: high (pharmacologic + mechanical + extended prophylaxis post-discharge for high-risk surgery).
Pharmacologic Prophylaxis Options
| Agent | Dose | Notes |
|---|---|---|
| Enoxaparin | 40 mg SC daily (prophylaxis); 30 mg SC q12h for trauma/orthopedic | Avoid if CrCl <30 (use UFH); for obesity (BMI >40 or wt >120 kg): 40 mg SC q12h |
| Heparin (UFH) | 5,000 units SC q8h (preferred) or q12h | Use when CrCl <30 or high bleeding risk (shorter half-life); q8h dosing superior to q12h |
| Fondaparinux | 2.5 mg SC daily | Alternative for HIT; avoid if CrCl <30; no HIT cross-reactivity |
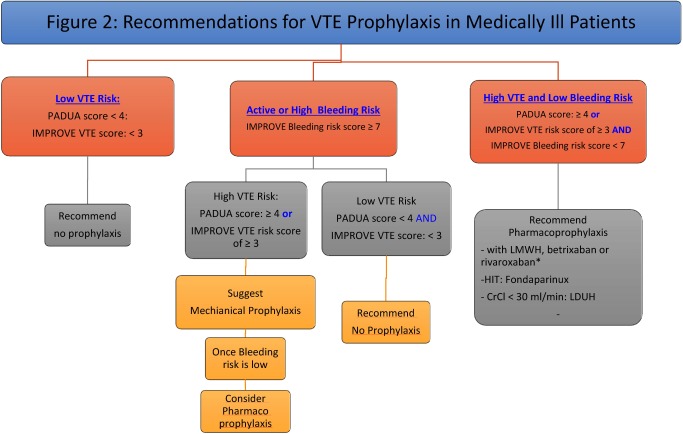
Mechanical Prophylaxis
Sequential compression devices (SCDs) are the primary mechanical option. Indications: all patients with contraindications to pharmacologic prophylaxis (active bleeding, severe thrombocytopenia <50,000, recent neurosurgery, epidural catheter in place) or as adjunct to pharmacologic prophylaxis in high-risk patients. Graduated compression stockings (TED hose) provide modest benefit but are less effective than SCDs. Mechanical prophylaxis should be applied on admission and worn continuously except during ambulation and bathing.
DOAC Perioperative Management
For elective procedures: apixaban/rivaroxaban — hold 48 hours before high-bleeding-risk procedures, 24 hours before low-risk. Dabigatran — hold 48–72 hours (longer if CrCl 30–50: hold 72–96 hours for high-risk). No bridging with heparin is generally needed for DOACs (PERIOP2 and PAUSE trials). Resume 48–72 hours postoperatively once hemostasis is confirmed. For warfarin: hold 5 days before procedure (INR target <1.5); bridge with LMWH only for high-risk patients (mechanical mitral valve, recent VTE <3 months, high-risk thrombophilia).
16 Anticoagulation Management
Anticoagulation is one of the highest-risk medication classes in the hospital, accounting for a disproportionate share of adverse drug events. The hospitalist must master heparin dosing, warfarin management, DOAC selection, reversal strategies, and the recognition and management of heparin-induced thrombocytopenia.
Heparin Nomogram (Weight-Based)
| aPTT (seconds) | Action |
|---|---|
| <35 (<1.2× control) | 80 units/kg bolus, increase rate by 4 units/kg/h |
| 35–45 (1.2–1.5×) | 40 units/kg bolus, increase rate by 2 units/kg/h |
| 46–70 (1.5–2.3×) — therapeutic | No change |
| 71–90 (2.3–3.0×) | Decrease rate by 2 units/kg/h |
| >90 (>3.0×) | Hold infusion for 1 hour, then decrease rate by 3 units/kg/h |
Check aPTT 6 hours after any rate change. Once two consecutive therapeutic values are achieved, extend monitoring to q12–24h. Anti-Xa monitoring (target 0.3–0.7 IU/mL) may be more reliable than aPTT in patients with lupus anticoagulant, elevated factor VIII, or liver disease.
Warfarin Initiation & INR Targets
Start warfarin 5 mg daily (lower starting dose of 2–3 mg for elderly, liver disease, malnourished, CYP2C9/VKORC1 polymorphisms, or concurrent interacting drugs). Bridge with heparin/LMWH for ≥5 days AND until INR ≥2.0 for ≥24 hours. INR targets: 2.0–3.0 for DVT/PE, atrial fibrillation, bioprosthetic valve; 2.5–3.5 for mechanical mitral valve or mechanical aortic valve with additional risk factors. Supratherapeutic INR management: INR 4.5–10 without bleeding — hold warfarin, no vitamin K needed (unless high bleeding risk); INR >10 without bleeding — hold warfarin + vitamin K 2.5–5 mg PO; serious/life-threatening bleeding — hold warfarin + 4-factor PCC (Kcentra) 25–50 IU/kg + vitamin K 10 mg IV.
DOAC Dosing by Indication
| Drug | Atrial Fibrillation | VTE Treatment | VTE Prevention (Extended) |
|---|---|---|---|
| Apixaban | 5 mg BID (2.5 mg if ≥2 of: age ≥80, wt ≤60 kg, Cr ≥1.5) | 10 mg BID × 7d, then 5 mg BID | 2.5 mg BID |
| Rivaroxaban | 20 mg daily with dinner (15 mg if CrCl 15–50) | 15 mg BID × 21d, then 20 mg daily | 10 mg daily |
| Edoxaban | 60 mg daily (30 mg if CrCl 15–50 or wt ≤60 kg) | 60 mg daily after 5–10d heparin | Not established |
| Dabigatran | 150 mg BID (110 mg BID if age >80 or CrCl 30–50 in some guidelines) | 150 mg BID after 5–10d heparin | 150 mg BID |
Reversal Agents
| Anticoagulant | Reversal Agent | Dose | Notes |
|---|---|---|---|
| UFH | Protamine sulfate | 1 mg per 100 units heparin given in last 2–3 h (max 50 mg) | Anaphylaxis risk in NPH insulin users, fish allergy; monitor for rebound |
| LMWH (enoxaparin) | Protamine sulfate | 1 mg per 1 mg enoxaparin (given <8 h); only ~60% reversal | Incomplete reversal; consider 4F-PCC if life-threatening bleeding |
| Warfarin | Vitamin K + 4F-PCC (Kcentra) | Vit K 10 mg IV; 4F-PCC 25–50 IU/kg based on INR | 4F-PCC onset <30 min (vs FFP 6–8 h); recheck INR in 30 min |
| Dabigatran | Idarucizumab (Praxbind) | 5 g IV (two 2.5 g vials) | Specific reversal agent; complete reversal in minutes; does not affect other DOACs |
| Apixaban/Rivaroxaban | Andexanet alfa (Andexxa) | Low dose: 400 mg bolus + 4 mg/min × 120 min; High dose: 800 mg bolus + 8 mg/min × 120 min | High dose for rivaroxaban >10 mg or apixaban >5 mg taken <8 h ago; very expensive (~$50k); 4F-PCC 50 IU/kg is a reasonable alternative |
Heparin-Induced Thrombocytopenia (HIT) — 4T Score
| Category | 2 Points | 1 Point | 0 Points |
|---|---|---|---|
| Thrombocytopenia | Platelet fall >50% and nadir ≥20,000 | Fall 30–50% or nadir 10–19,000 | Fall <30% or nadir <10,000 |
| Timing of platelet fall | Day 5–10 of heparin exposure or ≤1 day if prior heparin within 30 days | Consistent with day 5–10 but unclear; onset after day 10; or fall ≤1 day with prior heparin 30–100 days ago | Fall <4 days without recent heparin |
| Thrombosis or other sequelae | New thrombosis, skin necrosis, or acute systemic reaction after IV bolus | Progressive or recurrent thrombosis; erythematous skin lesions; suspected thrombosis not proven | None |
| OTher cause for thrombocytopenia | No other apparent cause | Possible other cause | Definite other cause present |
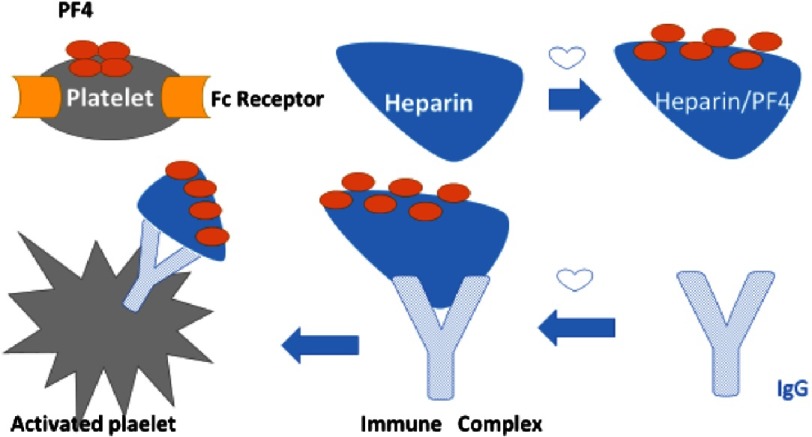
Score 0–3: low probability (<5% chance of HIT) — HIT unlikely, continue heparin. Score 4–5: intermediate (14%) — send HIT antibody (PF4/heparin ELISA); if positive, confirm with serotonin release assay (SRA). Score 6–8: high probability (64%) — stop all heparin immediately (including flushes), start alternative anticoagulation (argatroban for hepatic metabolism, bivalirudin for short procedures, fondaparinux off-label). Do NOT start warfarin until platelets recover >150,000 (warfarin depletes protein C faster than clotting factors, causing a transient hypercoagulable state that can cause venous limb gangrene).
17 Electrolyte Management
Electrolyte disorders are ubiquitous in hospitalized patients and can cause life-threatening arrhythmias, seizures, and neuromuscular dysfunction. The hospitalist must know correction rates, treatment protocols, and the critical ECG changes that demand emergent intervention.
Hyponatremia
Serum Na <135 mEq/L. Most common electrolyte abnormality in hospitalized patients. Classification by volume status: hypovolemic (GI losses, diuretics, adrenal insufficiency — treat with NS), euvolemic (SIADH, hypothyroidism, adrenal insufficiency, psychogenic polydipsia — fluid restrict to <1 L/day, consider salt tabs, demeclocycline, or tolvaptan for refractory SIADH), hypervolemic (CHF, cirrhosis, nephrotic syndrome — fluid restrict, diuretics). Correction rate: do not exceed 8 mEq/L in any 24-hour period (6 mEq/L in patients at high risk for osmotic demyelination syndrome: alcoholism, malnutrition, hypokalemia, liver disease). Overcorrection treatment: desmopressin (DDAVP) 2–4 mcg IV to halt free water excretion, plus D5W infusion to re-lower sodium.
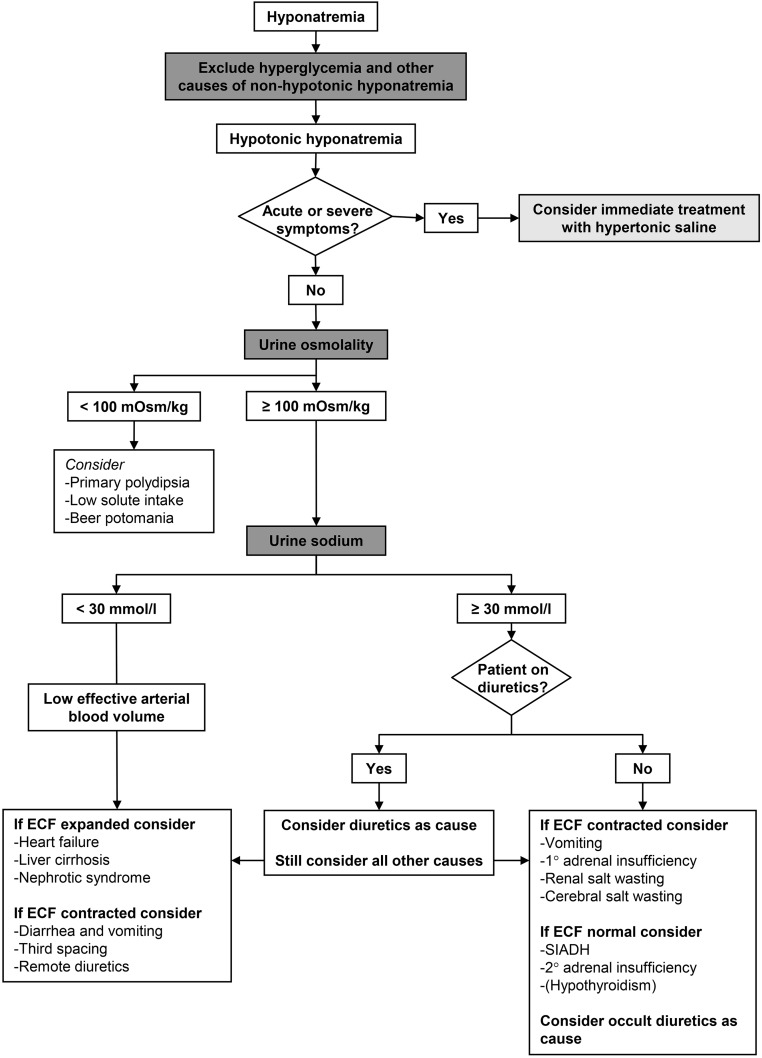
Severe Symptomatic Hyponatremia
Seizures or obtundation from hyponatremia: 3% hypertonic saline 100 mL IV bolus over 10 minutes, may repeat twice at 10-minute intervals (total 300 mL). Goal: raise Na by 4–6 mEq/L in the first few hours to stop symptoms, then slow correction to stay within 8 mEq/L in 24 hours. Check Na q2h during active correction.
Hypernatremia
Serum Na >145 mEq/L. Almost always due to free water deficit (impaired thirst, restricted access to water, diabetes insipidus, osmotic diuresis). Calculate free water deficit: FWD = TBW × [(Na/140) − 1], where TBW = 0.6 × weight (kg) for men, 0.5 for women. Replace half the deficit in the first 24 hours and the remainder over the next 24–48 hours using D5W or free water via feeding tube. Correction rate: do not exceed 10 mEq/L per 24 hours (faster correction risks cerebral edema).
Hypokalemia
K <3.5 mEq/L. Causes: GI losses, diuretics, alkalosis, refeeding syndrome, insulin, beta-agonists, hypomagnesemia. ECG changes: flattened/inverted T waves, prominent U waves, ST depression, prolonged QT, risk of torsades de pointes. Treatment: Mild (3.0–3.5): oral KCl 40–80 mEq/day in divided doses. Moderate (2.5–3.0): IV KCl 10–20 mEq/h via central line (max 40 mEq/h for emergencies via central line with continuous monitoring) + oral supplementation. Severe (<2.5 or symptomatic): IV KCl 20–40 mEq/h via central line with continuous cardiac monitoring. Always check and replace magnesium — hypomagnesemia makes hypokalemia refractory to treatment (Mg is required for ROMK channel function in the distal nephron). 10 mEq IV KCl raises serum K by approximately 0.1 mEq/L.
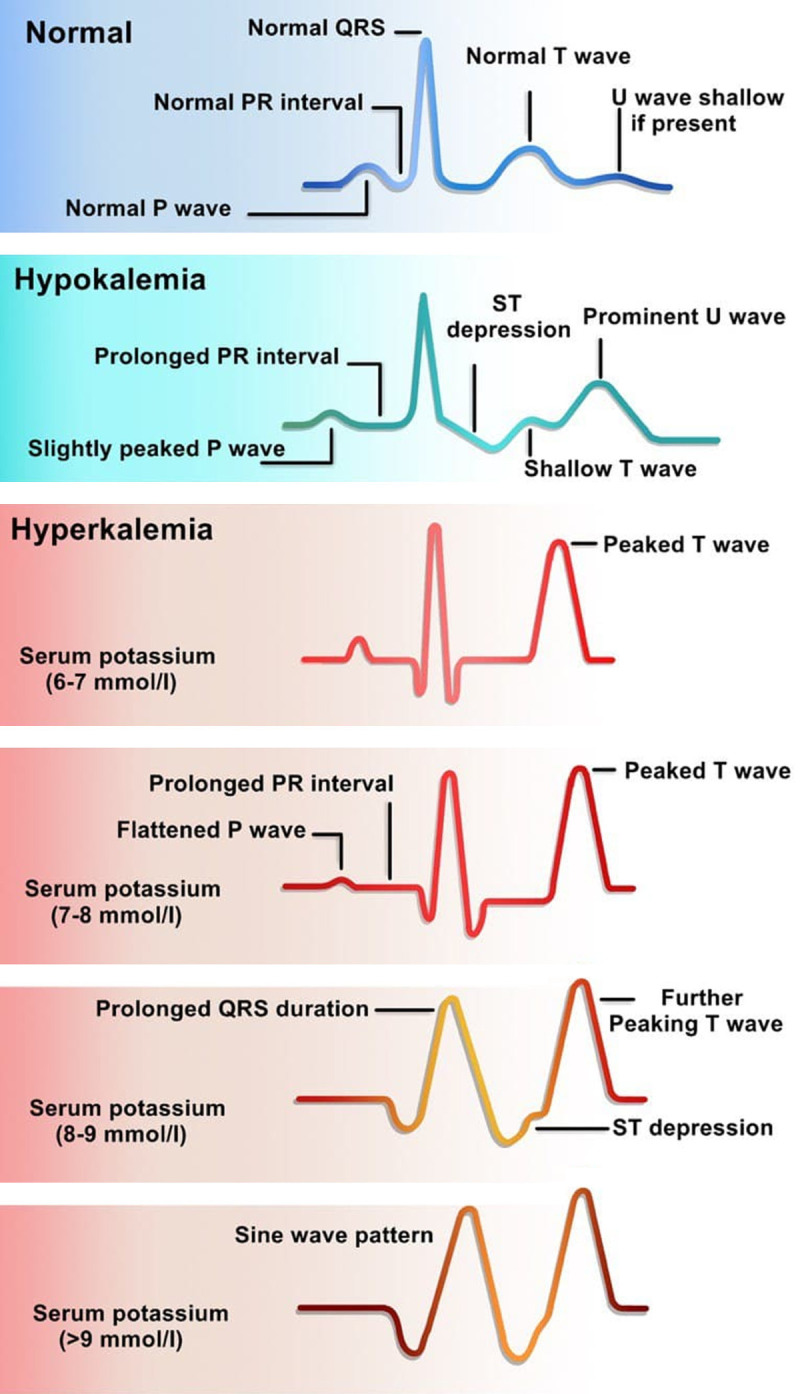
Hyperkalemia
K >5.0 mEq/L. Causes: AKI/CKD, acidosis, cell lysis (rhabdomyolysis, tumor lysis, hemolysis), medications (ACEi/ARB, spironolactone, TMP-SMX, NSAIDs, heparin), pseudohyperkalemia (hemolyzed sample, tourniquet, thrombocytosis). ECG progression: peaked T waves (earliest, K ~5.5–6.5) → PR prolongation → loss of P waves → widened QRS → sine wave → V-fib/asystole.
Treatment ladder:
| Intervention | Dose | Onset | Mechanism |
|---|---|---|---|
| Calcium gluconate | 1–2 g (10–20 mL of 10%) IV over 2–3 min | 1–3 min | Membrane stabilization (does not lower K) |
| Regular insulin + D50 | 10 units regular insulin IV + 25 g dextrose (1 amp D50) | 15–30 min | Drives K intracellularly; lowers K by 0.5–1.0 mEq/L; check BG at 1 and 2 h |
| Albuterol nebulizer | 10–20 mg nebulized over 15 min | 15–30 min | Beta-2 stimulation drives K into cells; lowers K by 0.5–1.0 mEq/L; additive with insulin |
| Sodium bicarbonate | 50–100 mEq IV | 30–60 min | Drives K into cells in setting of acidosis; minimal effect if pH normal |
| Sodium polystyrene sulfonate (Kayexalate) | 15–30 g PO or rectal | 1–6 h | GI cation exchange; unreliable; risk of intestinal necrosis (avoid post-op) |
| Patiromer (Veltassa) | 8.4 g PO daily | 7 h | GI K binder; better tolerated; for chronic management |
| Sodium zirconium cyclosilicate (Lokelma) | 10 g PO TID × 48 h | 1 h | GI K binder; faster onset than patiromer |
| Furosemide | 40–80 mg IV | 30–60 min | Renal K excretion (requires adequate renal function) |
| Hemodialysis | Emergent | Immediate | Definitive removal; for refractory hyperkalemia or anuric patients |
Hypocalcemia & Hypercalcemia
Hypocalcemia (corrected Ca <8.5 or ionized Ca <4.0): causes include hypoparathyroidism (post-thyroidectomy), vitamin D deficiency, CKD, hypomagnesemia, massive transfusion (citrate chelation), pancreatitis. Symptoms: perioral/extremity paresthesias, Chvostek sign, Trousseau sign, QT prolongation. Treatment: symptomatic or ionized Ca <3.2 — calcium gluconate 1–2 g IV over 10–20 min, then 0.5–1.5 mg/kg/h infusion; mild — calcium carbonate 1–2 g PO TID with vitamin D. Hypercalcemia (Ca >10.5): causes — primary hyperparathyroidism and malignancy account for >90%. Treatment: IV NS 200–300 mL/h (volume expansion and calciuresis), then furosemide 20–40 mg once euvolemic (not first-line), calcitonin 4 IU/kg SC/IM q12h (rapid onset, tachyphylaxis in 48 h), zoledronic acid 4 mg IV over 15 min (peak effect 2–4 days, lasts weeks; avoid if CrCl <35), denosumab 120 mg SC for refractory/renal failure.
Hypomagnesemia
Mg <1.8 mg/dL. Common in hospitalized patients (up to 65% of ICU patients). Causes: diuretics (loop and thiazide), alcoholism, PPIs (chronic use), diarrhea, refeeding syndrome. Makes hypokalemia and hypocalcemia refractory to correction. Treatment: mild (1.2–1.8) — magnesium oxide 400–800 mg PO daily; moderate/severe (<1.2 or symptomatic) — magnesium sulfate 1–2 g IV over 1 hour (can give up to 4–6 g/day); for torsades de pointes — magnesium sulfate 2 g IV push over 1–2 min.
Hypophosphatemia & Refeeding Syndrome
Phosphate <2.5 mg/dL. Refeeding syndrome occurs when malnourished patients (alcoholism, anorexia nervosa, prolonged NPO, cancer cachexia) are refed. Insulin surge drives phosphate, potassium, and magnesium intracellularly, causing severe hypophosphatemia (can cause respiratory failure from diaphragmatic weakness, cardiac dysfunction, rhabdomyolysis, seizures). Prevention: start nutrition at 10–15 kcal/kg/day, advance slowly over 5–7 days; supplement thiamine 200–300 mg IV before feeding (prevents Wernicke encephalopathy); check and replace PO₄, K, Mg BID. Treatment of hypophosphatemia: mild (2.0–2.5) — sodium or potassium phosphate 30–40 mmol PO daily; severe (<1.0 or symptomatic) — potassium phosphate or sodium phosphate 20–40 mmol IV over 4–6 hours; monitor Ca (IV phosphate can cause hypocalcemia and metastatic calcification).
18 Pain Management in Hospitalized Patients
Effective pain management improves patient outcomes, mobility, and satisfaction while reducing complications. The hospitalist must employ multimodal analgesia, understand opioid equivalencies, and anticipate opioid side effects.
Multimodal Analgesia Approach
The multimodal strategy uses agents from different drug classes to target multiple pain pathways, reducing opioid requirements by 30–50%. The foundation: scheduled acetaminophen (650–1000 mg PO/IV q6h, max 3 g/day in liver disease or elderly, 4 g/day otherwise) + NSAID/COX-2 inhibitor (ketorolac 15–30 mg IV q6h, max 5 days; or ibuprofen 400–600 mg PO q6h; avoid in AKI, GI bleed, heart failure, post-CABG) + adjuncts as appropriate (gabapentin 100–300 mg PO TID for neuropathic pain, lidocaine patch 5% topical for localized pain, muscle relaxants for musculoskeletal spasm). Add opioids only when non-opioid multimodal therapy is insufficient.
Opioid Equivalency Table
| Opioid | IV Dose (mg) | PO Dose (mg) | PO:IV Ratio | Duration |
|---|---|---|---|---|
| Morphine | 10 | 30 | 3:1 | 3–4 h |
| Hydromorphone (Dilaudid) | 1.5 | 7.5 | 5:1 | 3–4 h |
| Oxycodone | N/A (no IV form) | 20 | N/A | 3–4 h |
| Hydrocodone | N/A | 30 | N/A | 4–6 h |
| Fentanyl | 0.1 (100 mcg) | N/A (transdermal/buccal) | N/A | 30–60 min IV |
| Tramadol | N/A | 120 (weak; avoid — serotonin syndrome, seizure risk) | N/A | 4–6 h |
When converting between opioids, calculate the total 24-hour dose in morphine milligram equivalents (MME), then reduce by 25–50% for incomplete cross-tolerance.
IV-to-PO Conversion
When transitioning from IV to PO opioids: calculate the total IV dose in 24 hours, convert to oral morphine equivalents using the ratios above, reduce by 25% for safety, and divide into appropriate dosing intervals. Example: patient receiving morphine 2 mg IV q4h (12 mg IV/day) → 12 × 3 = 36 mg PO morphine/day → reduce by 25% = 27 mg → morphine IR 5 mg PO q4h PRN (30 mg/day available).
PCA Protocols
Patient-controlled analgesia (PCA) delivers small opioid doses on patient demand. Standard settings: demand dose (morphine 1–2 mg or hydromorphone 0.2–0.4 mg), lockout interval (6–10 minutes), 4-hour limit (to prevent excessive dosing), and optional continuous/basal rate (use with caution — increases sedation and respiratory depression risk; generally avoid in opioid-naive patients). PCA requires: patient ability to understand and operate the button, no cognitive impairment, continuous pulse oximetry monitoring, and a bowel regimen.
Bowel Regimen with Opioids
Opioid-induced constipation (OIC) occurs in 40–80% of patients on opioids. Prophylactic bowel regimen for all patients on scheduled opioids: senna 8.6–17.2 mg PO BID (stimulant) + docusate 100 mg PO BID (stool softener). If no BM in 48 hours: add bisacodyl 10 mg PO or PR. If refractory: polyethylene glycol (MiraLAX) 17 g PO daily, or lactulose 15–30 mL PO q6h. For severe OIC in patients on chronic opioids: methylnaltrexone (Relistor) 12 mg SC every other day (peripherally acting mu-opioid receptor antagonist, does not cross BBB, does not reverse analgesia) or naloxegol 25 mg PO daily.
19 Hospital-Acquired & Ventilator-Associated Pneumonia
Hospital-acquired pneumonia (HAP) develops ≥48 hours after admission in non-intubated patients. Ventilator-associated pneumonia (VAP) develops ≥48 hours after endotracheal intubation. Together they are the leading cause of death from hospital-acquired infections, with mortality rates of 20–50% for VAP.
Definitions & Diagnosis
HAP: new or progressive pulmonary infiltrate on imaging plus clinical signs of infection (fever >38°C, purulent sputum, leukocytosis >12,000 or leukopenia <4,000, declining oxygenation) developing ≥48 hours after admission, without preceding intubation. VAP: same criteria, developing ≥48 hours after endotracheal intubation. Diagnosis requires: chest imaging showing new infiltrate, plus at least 2 of 3 clinical criteria (fever/hypothermia, leukocytosis/leukopenia, purulent secretions). Send sputum or endotracheal aspirate (or BAL) for Gram stain and culture before starting antibiotics (but do not delay treatment while awaiting results).
Empiric Coverage
HAP/VAP empiric therapy must cover MRSA + Pseudomonas aeruginosa + gram-negative bacilli. Recommended empiric regimen:
| Target | Agent Options | Dose |
|---|---|---|
| MRSA coverage | Vancomycin or linezolid | Vancomycin 15–20 mg/kg IV q8–12h (AUC/MIC 400–600) or linezolid 600 mg IV q12h |
| Pseudomonas coverage (choose one) | Piperacillin-tazobactam or cefepime or meropenem or imipenem-cilastatin | Pip-tazo 4.5 g IV q6h; cefepime 2 g IV q8h; meropenem 1 g IV q8h |
Add a second antipseudomonal agent (aminoglycoside or fluoroquinolone) only if: high risk of multidrug-resistant organisms, prior IV antibiotics within 90 days, structural lung disease, or >25% local Pseudomonas resistance to the primary agent. Obtain nasal MRSA PCR: negative result has >95% NPV — supports de-escalation off MRSA coverage within 48–72 hours.
De-escalation & Duration
Narrow antibiotics based on culture and sensitivity results at 48–72 hours. Standard duration: 7 days for uncomplicated HAP/VAP (2016 IDSA guidelines). Procalcitonin-guided de-escalation (discontinue antibiotics when procalcitonin decreases by ≥80% from peak or to <0.5 ng/mL) reduces antibiotic exposure without increasing mortality.
VAP Prevention Bundle
Evidence-based interventions that reduce VAP incidence by 40–70%: head of bed elevation 30–45°, daily sedation interruption and spontaneous breathing trials (SAT/SBT pairing), oral care with chlorhexidine 0.12% q12h (controversial — some recent data suggests possible harm; institutional variation), subglottic suctioning endotracheal tubes, peptic ulcer prophylaxis only when indicated (not universal), VTE prophylaxis, and daily assessment of readiness for extubation.
20 Clostridioides difficile Infection
Clostridioides difficile (formerly Clostridium difficile) is the most common cause of healthcare-associated infectious diarrhea. CDI rates have increased dramatically, with approximately 500,000 infections and 29,000 deaths annually in the US. The hospitalist must recognize risk factors, select appropriate testing, and tailor treatment to severity.
Risk Factors
Major risk factors: antibiotics (fluoroquinolones, clindamycin, cephalosporins, and carbapenems are highest risk; any antibiotic can cause CDI), age ≥65, hospitalization ≥4 days, proton pump inhibitors (controversial but listed in IDSA guidelines as risk factor), immunosuppression (chemotherapy, organ transplant), and prior CDI (recurrence rate 20–25% after first episode, >40% after second).
Diagnostic Testing
Test only patients with ≥3 unformed stools in 24 hours. The optimal strategy is a two-step algorithm: (1) GDH (glutamate dehydrogenase) antigen screen — sensitive but not specific (detects toxigenic and non-toxigenic strains), then (2) toxin EIA (detects toxins A and B) or NAAT/PCR if GDH positive. Alternatively, a NAAT (nucleic acid amplification test / PCR) alone is very sensitive (sensitivity >95%) but may detect colonization rather than active disease (positive PCR without clinical disease = carrier, not CDI). Toxin EIA alone is specific (99%) but less sensitive (75–85%). Do not test formed stools. Do not repeat testing within 7 days if initially negative. Do not use test of cure after treatment.
Severity Classification & Treatment
| Severity | Criteria | First-Line Treatment | Alternative |
|---|---|---|---|
| Non-severe | WBC ≤15,000 AND Cr <1.5 mg/dL | Fidaxomicin 200 mg PO BID × 10 days (preferred per 2021 IDSA update) or vancomycin 125 mg PO QID × 10 days | Metronidazole 500 mg PO TID × 10 days (only if fidaxomicin/vancomycin unavailable) |
| Severe | WBC ≥15,000 OR Cr ≥1.5 mg/dL | Vancomycin 125 mg PO QID × 10 days or fidaxomicin 200 mg PO BID × 10 days | Vancomycin preferred for severe per many institutions |
| Fulminant | Hypotension, ileus, megacolon, ICU admission for CDI | Vancomycin 500 mg PO/NG QID + metronidazole 500 mg IV q8h; if ileus: vancomycin 500 mg in 100 mL NS per rectum q6h | Surgical consultation for colectomy if: lactate ≥5, WBC ≥50,000, megacolon, perforation, refractory shock |
Recurrence Management
First recurrence: fidaxomicin 200 mg PO BID × 10 days (preferred — lower recurrence rate than vancomycin), or vancomycin pulse-taper (125 mg QID × 14 days, then BID × 7 days, then daily × 7 days, then every 2–3 days × 2–4 weeks). Second or subsequent recurrence: consider fecal microbiota transplant (FMT) — efficacy 80–90% for recurrent CDI; FDA-approved options include fecal microbiota live-jslm (Rebyota, rectal) and fecal microbiota spores live-brpk (Vowst, oral). Bezlotoxumab (monoclonal antibody against toxin B) 10 mg/kg IV single dose can be given with standard therapy to reduce recurrence (NNT ~10).
CDI requires contact precautions (gown and gloves) for the duration of diarrhea. Handwashing with soap and water is mandatory (alcohol-based hand sanitizers do NOT kill C. difficile spores). Dedicated patient equipment (stethoscope, thermometer, BP cuff). Terminal room cleaning with sporicidal agent (bleach-based, UV light, or hydrogen peroxide vapor). Antibiotic stewardship — minimize use of fluoroquinolones, clindamycin, and unnecessary broad-spectrum antibiotics.
21 Catheter-Associated UTI & CLABSI
Catheter-associated urinary tract infections (CAUTI) and central line-associated bloodstream infections (CLABSI) are the two most common device-associated hospital-acquired infections and CMS-designated never events. Prevention is a core hospitalist responsibility.
CAUTI
Definition: patient with an indwelling urinary catheter (or catheter removed within 48 hours) who develops urinary symptoms (fever, suprapubic tenderness, costovertebral angle tenderness, urgency, frequency, dysuria) plus urine culture ≥10⁵ CFU/mL of ≤2 organisms. Critical distinction: asymptomatic bacteriuria (ASB) — positive urine culture without urinary symptoms — should NOT be treated in catheterized patients (except in pregnancy or before urologic procedures). Treating ASB drives antibiotic resistance and CDI without clinical benefit.
Prevention bundle: (1) Avoid unnecessary catheter insertion — appropriate indications: urinary retention/obstruction, accurate I&O measurement in critically ill patients, perioperative use for selected surgeries, open sacral/perineal wounds in incontinent patients, comfort care at end of life. (2) Daily assessment of catheter necessity with prompt removal (nurse-driven removal protocols reduce CAUTI by 50%). (3) Aseptic insertion technique. (4) Maintain closed drainage system. (5) Keep bag below bladder. (6) Consider alternatives: condom catheter, intermittent straight catheterization, bladder scanner.
CLABSI
Definition: laboratory-confirmed bloodstream infection in a patient with a central venous catheter in place for ≥2 calendar days (day 1 = insertion day), with no other identified source of infection. Common organisms: coagulase-negative Staphylococci (most common), S. aureus, Enterococcus, Candida, gram-negative bacilli.
Prevention bundle (central line insertion): hand hygiene, maximal sterile barrier precautions (cap, mask, sterile gown, sterile gloves, full-body sterile drape), chlorhexidine skin antisepsis (allow to dry), optimal site selection (subclavian preferred over IJ or femoral for lowest infection risk — but ultrasound-guided IJ is often preferred to avoid pneumothorax), sterile dressing. Maintenance bundle: daily assessment of line necessity, scrub the hub (alcohol swab for 15 seconds before access), chlorhexidine-impregnated dressing or sponge, standardized tubing changes.
Blood Culture Interpretation
Distinguishing true bacteremia from contamination is essential. Likely contaminants (single positive culture): coagulase-negative Staphylococci, Corynebacterium spp., Bacillus spp., Propionibacterium acnes. True pathogens (even single positive): S. aureus, Streptococcus pneumoniae, E. coli and other Enterobacteriaceae, Pseudomonas aeruginosa, Candida spp. When a potential contaminant is isolated, repeat cultures before initiating treatment. S. aureus bacteremia always requires a minimum 2 weeks of IV antibiotics (4–6 weeks if endocarditis, osteomyelitis, or metastatic infection) and a transthoracic or transesophageal echocardiogram to rule out endocarditis.
Line Removal Decision
Remove the central line for: S. aureus (always), Candida (always), tunnel infection or port pocket infection, septic shock, persistent bacteremia after 72 hours of appropriate antibiotics, and complicated infection (endocarditis, septic thrombosis). Line salvage (antibiotic lock therapy + systemic antibiotics) may be attempted for coagulase-negative Staphylococci in patients with limited venous access and uncomplicated infection, provided the patient defervesces within 72 hours.
22 Falls, Delirium & Pressure Injuries
Falls, delirium, and pressure injuries are the triad of preventable hospital-acquired conditions that disproportionately affect elderly and critically ill patients. All three are CMS-reportable quality measures and are tied to reimbursement penalties.
Morse Fall Scale
| Risk Factor | Scale | Points |
|---|---|---|
| History of falling (within 3 months) | No = 0, Yes = 25 | 0 or 25 |
| Secondary diagnosis (≥2 medical diagnoses) | No = 0, Yes = 15 | 0 or 15 |
| Ambulatory aid | None/bed rest/nurse assist = 0; Crutches/cane/walker = 15; Furniture = 30 | 0, 15, or 30 |
| IV therapy / heparin lock | No = 0, Yes = 20 | 0 or 20 |
| Gait | Normal/bedrest/immobile = 0; Weak = 10; Impaired = 20 | 0, 10, or 20 |
| Mental status | Oriented to own ability = 0; Overestimates/forgets limitations = 15 | 0 or 15 |
Score 0–24: no risk; 25–44: low risk (standard fall precautions); ≥45: high risk (intensive fall prevention — bed alarm, non-slip socks, low bed, sitter if needed, PT consult, medication review).
Delirium — CAM (Confusion Assessment Method)
The CAM is the gold standard bedside delirium screening tool. Diagnosis requires features 1 AND 2, plus either 3 OR 4:
| Feature | Description | Assessment |
|---|---|---|
| 1. Acute onset & fluctuating course | Change in mental status from baseline that waxes and wanes over the course of the day | Ask nursing staff: "Is the patient’s behavior different from baseline? Does it fluctuate?" |
| 2. Inattention | Difficulty focusing, easily distracted, difficulty following conversation | Ask patient to recite months backward from December to July; errors = positive |
| 3. Disorganized thinking | Rambling, irrelevant, or incoherent speech; illogical flow of ideas | Ask: "Will a stone float on water?" "Are there fish in the sea?" Errors = positive |
| 4. Altered level of consciousness | Anything other than alert: vigilant, lethargic, stuporous, comatose | Observe; use RASS (Richmond Agitation-Sedation Scale) |
CAM sensitivity 94–100%, specificity 90–95% when performed correctly. For ICU patients, use the CAM-ICU (modified for non-verbal patients on ventilators).
Delirium Prevention — HELP Protocol
The Hospital Elder Life Program (HELP) reduces delirium incidence by 40% (Inouye et al., NEJM 1999). Components: daily reorientation (calendar, clock, familiar objects, windows), cognitive stimulation (conversation, word games), early mobilization (PT/OT, ambulation 3×/day), sleep promotion (minimize nighttime vitals/medications, reduce noise, warm milk, relaxation techniques, avoid benzodiazepines for sleep — use melatonin 1–3 mg instead), nutritional support (assistance with meals, hydration), vision and hearing aids (ensure glasses and hearing aids are at the bedside). Non-pharmacologic prevention is far more effective than pharmacologic treatment.
Pressure Injuries — Braden Scale
The Braden Scale predicts pressure injury risk. Six subscales, each scored 1–4 (except friction/shear, scored 1–3):
| Subscale | 1 (Worst) | 2 | 3 | 4 (Best) |
|---|---|---|---|---|
| Sensory perception | Completely limited | Very limited | Slightly limited | No impairment |
| Moisture | Constantly moist | Very moist | Occasionally moist | Rarely moist |
| Activity | Bedfast | Chairfast | Walks occasionally | Walks frequently |
| Mobility | Completely immobile | Very limited | Slightly limited | No limitation |
| Nutrition | Very poor | Probably inadequate | Adequate | Excellent |
| Friction/Shear | Problem | Potential problem | No apparent problem | — |
Total score range 6–23. Score ≤12: high risk; 13–14: moderate risk; 15–18: mild risk; ≥19: no significant risk. High-risk patients should receive: repositioning q2h, pressure-redistribution mattress, moisture management, nutritional optimization (protein 1.25–1.5 g/kg/day, vitamin C, zinc), skin inspection every shift, and heel elevation off the bed surface.
Pressure Injury Staging (NPUAP/EPUAP)
| Stage | Description |
|---|---|
| Stage 1 | Non-blanchable erythema of intact skin; may be painful, warm, or cooler than surrounding tissue |
| Stage 2 | Partial-thickness skin loss involving epidermis and/or dermis; presents as shallow open ulcer with pink wound bed or intact/ruptured serum-filled blister |
| Stage 3 | Full-thickness skin loss extending into subcutaneous tissue; may include undermining/tunneling; bone/tendon/muscle not visible |
| Stage 4 | Full-thickness tissue loss with exposed bone, tendon, or muscle; often includes undermining and tunneling |
| Unstageable | Full-thickness tissue loss, base covered by slough (yellow/tan/gray/green/brown) and/or eschar (tan/brown/black); true depth cannot be determined until slough/eschar is removed |
| Deep Tissue Pressure Injury (DTPI) | Intact or non-intact skin with localized persistent non-blanchable deep red, maroon, or purple discoloration, or epidermal separation revealing a dark wound bed or blood-filled blister; indicates damage to underlying soft tissue from pressure/shear |
23 Bedside Procedures
Hospitalists perform and interpret several core bedside procedures. Proficiency in these procedures allows rapid diagnosis and therapeutic intervention without waiting for subspecialty availability.
Paracentesis
Indicated for: new-onset ascites (diagnostic), suspected spontaneous bacterial peritonitis (SBP), and symptomatic relief of tense ascites (therapeutic — remove up to 5 L; if >5 L, give albumin 6–8 g per liter removed to prevent post-paracentesis circulatory dysfunction). Site: left lower quadrant, lateral to rectus muscle (use ultrasound guidance to confirm fluid pocket and avoid inferior epigastric vessels). Send fluid for: cell count and differential (PMN ≥250 = SBP), albumin (calculate SAAG = serum albumin − ascites albumin), total protein, culture (inoculate blood culture bottles at bedside).
SAAG ≥1.1 g/dL = portal hypertension (97% accuracy): cirrhosis, heart failure, Budd-Chiari, portal vein thrombosis. SAAG <1.1 g/dL = non-portal hypertension: malignancy (peritoneal carcinomatosis), tuberculosis, nephrotic syndrome, pancreatitis. Ascites fluid total protein further distinguishes: protein <2.5 (cirrhosis, nephrotic) vs >2.5 (cardiac, Budd-Chiari, TB, malignancy).
Thoracentesis — Light’s Criteria
Indicated for: new pleural effusion of unknown etiology, suspected empyema/parapneumonic effusion, or symptomatic relief. Perform under ultrasound guidance (reduces pneumothorax risk to <2%). Therapeutic thoracentesis: remove up to 1–1.5 L (excessive removal risks re-expansion pulmonary edema). Send fluid for: cell count, protein, LDH, glucose, pH, Gram stain, culture, cytology (if malignancy suspected).
Light’s Criteria classify an effusion as exudative if ANY ONE of the following is met:
| Criterion | Exudate Threshold |
|---|---|
| Pleural fluid protein / serum protein | >0.5 |
| Pleural fluid LDH / serum LDH | >0.6 |
| Pleural fluid LDH | >2/3 the upper limit of normal serum LDH |
Transudates (meet none of the above): CHF (most common), cirrhosis (hepatic hydrothorax), nephrotic syndrome, atelectasis. Exudates: infection (pneumonia, empyema, TB), malignancy, PE, autoimmune (RA, lupus), pancreatitis, chylothorax. Note: Light’s criteria misclassify ~25% of CHF transudates as exudates (especially after diuresis) — if the fluid is near the cutoff and CHF is suspected, check serum-pleural fluid albumin gradient >1.2 to confirm transudate.
Lumbar Puncture
Indicated for: suspected meningitis/encephalitis, subarachnoid hemorrhage (after negative CT), normal-pressure hydrocephalus evaluation, CNS malignancy. Position: lateral decubitus (most accurate opening pressure) or seated. Opening pressure: normal 10–20 cmH₂O; elevated >25 cmH₂O (infection, idiopathic intracranial hypertension, mass lesion). CSF analysis: cell count (normal WBC <5; bacterial meningitis: 1,000–5,000+ with >80% neutrophils; viral: 50–1,000 with lymphocyte predominance; TB/fungal: 50–500 with lymphocyte predominance), protein (normal 15–45 mg/dL; elevated in bacterial >250, viral 50–200, TB 100–500), glucose (normal 60–70% of serum glucose; low <40 in bacterial, TB, fungal; normal in viral). Always get CT before LP if: papilledema, focal neurologic deficit, new seizure, altered consciousness, or immunocompromised (rule out mass lesion).
Central Line Placement
Site selection: Internal jugular (IJ) — most common for temporary access, lowest mechanical complication rate with ultrasound guidance, preferred for dialysis catheters. Subclavian — lowest infection rate, but highest pneumothorax risk (5%) and should be avoided in coagulopathy (non-compressible). Femoral — easiest landmark access, used in emergencies, but highest infection and DVT risk. All central lines should be placed with ultrasound guidance (real-time visualization reduces complications by ~70%), maximal sterile barrier precautions, and chlorhexidine skin prep. Post-placement CXR to confirm tip position (IJ/subclavian: at cavoatrial junction) and rule out pneumothorax.
Arthrocentesis
Indicated for: acute monoarthritis (mandatory to rule out septic arthritis), crystal arthropathy evaluation, and symptomatic relief of large effusions. Send fluid for: cell count (WBC >50,000 with >90% PMNs = septic until proven otherwise; 2,000–50,000 = inflammatory; <2,000 = non-inflammatory/OA), Gram stain and culture, crystal analysis under polarized microscopy (negatively birefringent = monosodium urate/gout; positively birefringent = calcium pyrophosphate/pseudogout). Anticoagulation is NOT a contraindication to arthrocentesis of an acutely inflamed joint — septic arthritis is an emergency that cannot be excluded without joint aspiration.
24 Consultation & Comanagement
Effective consultation is a core hospitalist skill. Knowing when to consult, how to frame the clinical question, and how to comanage complex patients with other specialties improves outcomes and efficiency.
When to Consult
Consult when: the patient has a condition outside the hospitalist’s scope of practice, a procedure is needed that the hospitalist does not perform, the diagnosis is uncertain despite thorough workup, the patient is not responding to standard therapy, or medicolegal risk is high and specialist input is warranted. Avoid "courtesy consults" with no clinical question — they waste specialist time and fragment care.
The 5 C’s of an Effective Consult Request
| C | Component | Example |
|---|---|---|
| Contact | Who is calling and how to reach them | "Dr. Smith, hospitalist team, pager 1234" |
| Communicate | Urgency level | "Urgent — please see within 4 hours" or "routine — within 24 hours" |
| Core question | Specific clinical question | "Is this patient a candidate for cholecystectomy?" NOT "please evaluate" |
| Clinical data | Relevant history, labs, imaging | "72F with RUQ pain, WBC 18k, lipase normal, US shows gallstones with wall thickening" |
| Collaborate | Express willingness to comanage | "Happy to manage the medical issues perioperatively" |
Surgical Comanagement
The hospitalist comanages surgical patients by handling: perioperative medical optimization (cardiac risk assessment — RCRI, medication management), postoperative medical complications (AKI, delirium, glycemic control, VTE prophylaxis, electrolytes, pain management), and discharge planning. Clear delineation of responsibilities between surgical and medical teams is essential. Common comanagement models: advisory (hospitalist provides recommendations), collaborative (shared order writing), and primary medical (hospitalist writes all medical orders while surgeon manages surgical issues).
Rapid Response & Code Blue
Rapid Response Teams (RRT) are activated for acute clinical deterioration before cardiac arrest. Common activation criteria: RR <8 or >28, HR <40 or >130, SBP <90, SpO₂ <90% despite O₂, acute change in mental status, new-onset seizure, or nursing concern ("worried" criterion). The hospitalist is typically part of the RRT and must rapidly assess, stabilize, and determine disposition (remain on floor, transfer to ICU). Code Blue (cardiac arrest): follow ACLS algorithms — high-quality CPR (rate 100–120, depth 2–2.4 inches, full recoil, minimize interruptions), early defibrillation for shockable rhythms (V-fib/pulseless V-tach: 200 J biphasic), epinephrine 1 mg IV q3–5 min, and identify/treat reversible causes (H’s and T’s: hypovolemia, hypoxia, hydrogen ion, hypo/hyperkalemia, hypothermia, tension pneumothorax, tamponade, toxins, thrombosis pulmonary, thrombosis coronary).
Palliative Care Consultation
Trigger criteria for palliative care consult: ICU stay >7 days with no improvement, advanced cancer with declining functional status, end-stage organ disease (HFrEF EF <20%, COPD on home O₂, ESRD on dialysis with declining function), frequent readmissions (≥3 in 6 months), surrogate decision-making conflicts, uncontrolled symptoms despite treatment, or patient/family requesting goals of care discussion. Early palliative care involvement reduces ICU LOS, healthcare costs, and improves quality of life without shortening survival (Temel et al., NEJM 2010).
25 Geriatric Hospital Medicine
Patients aged ≥65 account for over 35% of hospitalizations and are at disproportionate risk for medication adverse events, delirium, falls, functional decline, and iatrogenic complications. Geriatric-specific principles should guide every aspect of hospital care for this population.
Beers Criteria Highlights
The AGS Beers Criteria (updated 2023) identify potentially inappropriate medications in older adults. Key drugs to avoid:
| Drug / Class | Risk | Alternative |
|---|---|---|
| Benzodiazepines (lorazepam, diazepam, alprazolam) | Falls, fractures, delirium, cognitive impairment, oversedation | Melatonin, trazodone, non-pharmacologic sleep hygiene |
| First-generation antihistamines (diphenhydramine, hydroxyzine) | Anticholinergic: delirium, constipation, urinary retention, dry mouth | Cetirizine, loratadine (for allergy); melatonin for sleep |
| Long-acting sulfonylureas (glyburide) | Prolonged hypoglycemia (>24 hours) | Glipizide (shorter-acting), insulin, SGLT2i |
| Meperidine (Demerol) | Neurotoxic metabolite (normeperidine) causes seizures, especially in renal impairment | Morphine, hydromorphone, oxycodone |
| NSAIDs (chronic use) | GI bleeding, AKI, HTN exacerbation, heart failure | Acetaminophen, topical diclofenac, gabapentin for neuropathic pain |
| Metoclopramide | Extrapyramidal symptoms, tardive dyskinesia | Ondansetron |
| Skeletal muscle relaxants (cyclobenzaprine, methocarbamol) | Anticholinergic effects, sedation, falls | Physical therapy, acetaminophen, topical agents |
Polypharmacy & Deprescribing
Polypharmacy (≥5 medications) affects >40% of older adults and is the single strongest risk factor for adverse drug events. Hospitalization is an opportunity to deprescribe. Systematic approach: (1) list all medications, (2) identify drugs with no current indication, (3) identify drugs where risks outweigh benefits (Beers criteria, STOPP criteria), (4) identify drug interactions, (5) discuss with patient/family and taper/discontinue. Common targets: PPIs (taper after 8 weeks unless Barrett’s or chronic NSAID), bisphosphonates (drug holiday after 5 years oral / 3 years IV), statins (questionable benefit in limited life expectancy <1–2 years), and duplicative medications.
Functional Decline Prevention
Up to 35% of hospitalized older adults experience functional decline during hospitalization. Prevention strategies: early mobilization (PT/OT within 24 hours, ambulate in hallway TID), minimizing unnecessary bed rest orders, limiting tethers (remove Foley, telemetry, IVs as soon as possible), ensuring nutritional adequacy, and maintaining normal sleep-wake cycles. The ACE (Acute Care for Elders) unit model bundles these interventions and has been shown to reduce functional decline, LOS, and institutionalization.
26 Palliative Care & Goals of Care
Palliative care focuses on relief of suffering and improvement of quality of life for patients with serious illness, regardless of prognosis. It is not synonymous with hospice or end-of-life care. Every hospitalist should be competent in primary palliative care skills: symptom management, goals-of-care conversations, and advance care planning.
Symptom Management
| Symptom | First-Line | Second-Line | Notes |
|---|---|---|---|
| Dyspnea | Opioids: morphine 2–5 mg PO/IV q4h (most evidence-based); fan directed at face | Oxygen (if hypoxemic); benzodiazepines (lorazepam 0.5–1 mg) for anxiety component; scopolamine for secretions | Opioids do not hasten death at appropriate doses; supplemental O₂ has no benefit over fan/room air if SpO₂ >90% |
| Nausea | Ondansetron 4 mg IV/PO q6h; metoclopramide 10 mg PO/IV q6h (for gastroparesis/ileus) | Haloperidol 0.5–1 mg IV q6h (for opioid/chemotherapy-induced); dexamethasone 4–8 mg IV daily (for bowel obstruction, increased ICP) | Choose antiemetic based on mechanism: CTZ-mediated, vestibular, visceral, cortical |
| Pain | WHO pain ladder: non-opioid → weak opioid → strong opioid; multimodal as above | Adjuvants: dexamethasone for bone pain/inflammation, gabapentin/pregabalin for neuropathic, ketamine for refractory | Always assess pain with validated scale; reassess within 1 hour of intervention |
| Agitation / Terminal restlessness | Haloperidol 0.5–2 mg IV/PO q4h PRN | Lorazepam 0.5–2 mg IV q2h PRN (if refractory to haloperidol); chlorpromazine 25–50 mg IV/PO for refractory; palliative sedation with midazolam infusion for intractable cases | Rule out reversible causes: urinary retention, fecal impaction, pain, medication side effects |
Hospice Eligibility
Medicare hospice benefit requires: (1) two physicians certify a prognosis of ≤6 months if the disease follows its natural course, and (2) the patient elects comfort-focused care (forgoing curative treatment for the terminal diagnosis). General indicators of hospice-appropriate prognosis: recurrent hospitalizations, progressive functional decline (PPS ≤50%), unintentional weight loss >10% over 6 months, albumin <2.5, increasing dependence in ADLs. Disease-specific criteria exist for heart failure (EF <20%, NYHA IV despite optimal therapy), COPD (FEV1 <30%, O₂-dependent, cor pulmonale), dementia (FAST scale ≥7, unable to ambulate/dress/bathe/speak >6 words per day), and renal disease (not pursuing dialysis, CrCl <10 or Cr >8).
Advance Directive Documentation
Key documents: Advance directive / Living will — written instructions about desired medical treatment if the patient cannot communicate (varies by state). Healthcare Power of Attorney (HCPOA) / Healthcare proxy — designates a surrogate decision-maker. POLST/MOLST (Physician/Medical Orders for Life-Sustaining Treatment) — portable, actionable medical orders signed by both physician and patient; more specific than an advance directive (specifies code status, hospitalization preferences, artificial nutrition). Document the presence of these forms in the EMR, scan copies, and ensure orders match the patient’s expressed wishes.
Code Status Discussions — REMAP Framework
| Step | Component | Example Language |
|---|---|---|
| R | Reframe | "Given what’s happening with your illness, I want to make sure we are focused on what matters most to you." |
| E | Expect emotion | Pause. Acknowledge: "I can see this is difficult. It’s okay to feel that way." |
| M | Map out what matters | "If you were to get sicker, what would be most important to you — living as long as possible, or being comfortable?" |
| A | Align with values | "Based on what you’ve told me — that being at home with family is most important — I want to make sure our plan reflects that." |
| P | Plan treatments that match | "I would recommend that we focus on your comfort and avoid interventions like CPR that would not give you the quality of life you described." |
27 Medical-Legal & Documentation
Hospitalists carry substantial medicolegal risk due to the volume and acuity of patients they manage. Thorough documentation is the single most important defense against liability and ensures accurate coding, reimbursement, and quality reporting.
Medical Necessity Documentation
Every note should support the medical necessity for the hospitalization. Document: why the patient cannot be safely managed at a lower level of care, what services are being provided that require inpatient resources, and the expected length of stay. Use language such as "The patient requires inpatient hospitalization due to [specific clinical reasons]. Outpatient management is not appropriate because [specific barriers]." This documentation protects against RAC audits and denial of claims.
Clinical Decision-Making Levels (E&M 2021)
Under the 2021 CMS E&M guidelines, the level of service is determined by either total time OR medical decision-making (MDM) complexity. MDM has three components:
| MDM Level | Number/Complexity of Problems | Data Reviewed/Ordered | Risk of Complications/Management |
|---|---|---|---|
| Straightforward | 1 self-limited or minor problem | Minimal or no data | Minimal risk |
| Low | 2+ self-limited problems or 1 acute uncomplicated illness | Limited data (order/review labs, imaging, or records from external source) | Low risk (OTC drugs, minor surgery, PT) |
| Moderate | 1+ chronic illness with exacerbation or 1 acute illness with systemic symptoms | Moderate data (order/interpret tests, independent interpretation of imaging, discussion with external provider) | Moderate risk (prescription drug management, minor surgery with risk factors, IV fluids) |
| High | 1+ chronic illness with severe exacerbation or acute life-threatening illness | Extensive data (independent interpretation of tests, discussion of management with external physician, data from 3+ sources) | High risk (drug requiring intensive monitoring, hospitalization, decision regarding emergency surgery, DNR decision) |
Two of three components must meet the level to qualify. For hospital inpatient E&M: 99221 (straightforward/low), 99222 (moderate), 99223 (high) for initial; 99231 (low), 99232 (moderate), 99233 (high) for subsequent days.
Incident Reporting
Incidents requiring reporting: medication errors (wrong drug, wrong dose, wrong route, wrong patient), falls with injury, wrong-site/wrong-patient procedures, equipment malfunction causing harm, sentinel events (death or serious harm from a hospital-acquired cause), and near-misses. Incident reports are internal quality improvement documents. In the medical record, document the objective clinical facts (what happened, what was found, what was done) but do NOT reference the incident report itself (it is a privileged quality document in most jurisdictions).
AMA Discharge — Capacity Assessment & Documentation
When a patient wishes to leave against medical advice (AMA), the hospitalist must: (1) Assess decision-making capacity — the patient understands the diagnosis, the proposed treatment, the risks of leaving (including death), and the alternatives; this is a clinical assessment, not a legal determination (capacity is task-specific). (2) Clearly explain the risks of leaving. (3) Document the conversation thoroughly: "Patient was informed of [specific risks including death], understands and can articulate these risks, and still wishes to leave." (4) Offer alternatives: "You may return at any time. I would be happy to provide prescriptions for [medications] and arrange follow-up in [timeframe]." (5) Provide discharge medications, instructions, and follow-up information even if the patient leaves AMA. Do NOT withhold prescriptions or follow-up as "punishment." AMA discharge does NOT automatically void insurance coverage — this is a common myth.
Informed Consent
Required elements of valid informed consent: (1) nature of the procedure/treatment, (2) benefits, (3) risks (including common and serious/life-threatening), (4) alternatives (including no treatment), (5) opportunity to ask questions. The patient must have capacity, be free from coercion, and sign voluntarily. For patients lacking capacity, consent is obtained from the surrogate decision-maker (HCPOA, or next of kin per state hierarchy). In emergencies where consent cannot be obtained, the emergency exception doctrine allows treatment to prevent death or serious harm.
28 Scoring Systems Master Table
This section consolidates every major scoring system referenced in hospital medicine. Each score is fully enumerated with thresholds and interpretation.
CURB-65 (Pneumonia Severity)
| Score | 30-Day Mortality | Disposition |
|---|---|---|
| 0 | 0.6% | Outpatient |
| 1 | 2.7% | Outpatient or brief observation |
| 2 | 6.8% | Admission (short stay or observation) |
| 3 | 14.0% | Admission (consider ICU) |
| 4 | 27.8% | ICU |
| 5 | 57.0% | ICU |
Wells PE Score Interpretation
| Category | Score | PE Prevalence | Next Step |
|---|---|---|---|
| Unlikely | ≤4 | ~8% | D-dimer; if negative, PE excluded |
| Likely | >4 | ~35% | CTPA |
Wells DVT Score Interpretation
| Category | Score | DVT Prevalence | Next Step |
|---|---|---|---|
| Low | ≤0 | 5% | D-dimer; if negative, DVT excluded |
| Moderate | 1–2 | 17% | D-dimer or ultrasound |
| High | ≥3 | 53% | Compression ultrasound |
PESI Score Classes
| Class | Score | 30-Day Mortality | Risk |
|---|---|---|---|
| I | ≤65 | 1.6% | Very low — consider outpatient |
| II | 66–85 | 3.5% | Low |
| III | 86–105 | 7.1% | Intermediate |
| IV | 106–125 | 11.4% | High |
| V | >125 | 23.9% | Very high — ICU |
4T Score for HIT
| Score | Probability | HIT Likelihood | Action |
|---|---|---|---|
| 0–3 | Low | <5% | HIT unlikely; continue heparin |
| 4–5 | Intermediate | ~14% | Send PF4 ELISA; consider alternative anticoagulant |
| 6–8 | High | ~64% | Stop heparin; start argatroban or bivalirudin; send PF4 ELISA + SRA |
LACE Index (Readmission Risk)
| Score | 30-Day Readmission Risk |
|---|---|
| 0–4 | Low (~5%) |
| 5–9 | Moderate (~10–15%) |
| ≥10 | High (~25–30%) |
LRINEC Score (Necrotizing Fasciitis)
| Score | Interpretation |
|---|---|
| ≤5 | Low risk (<50% probability of NF) |
| 6–7 | Moderate risk (50–75%); consider MRI or surgical exploration |
| ≥8 | High risk (>75%); strong predictor of NF |
Braden Scale (Pressure Injury Risk)
| Score | Risk Level | Intervention |
|---|---|---|
| ≤9 | Very high risk | Specialty mattress, q1h repositioning, wound care consult |
| 10–12 | High risk | Pressure redistribution mattress, q2h repositioning, heel elevation |
| 13–14 | Moderate risk | Pressure redistribution, q2h repositioning, skin inspection q shift |
| 15–18 | Mild risk | Standard mattress with q2h repositioning |
| ≥19 | No significant risk | Standard care, encourage mobility |
Morse Fall Scale
| Score | Risk | Intervention |
|---|---|---|
| 0–24 | No risk | Standard precautions |
| 25–44 | Low risk | Standard fall precautions |
| ≥45 | High risk | Intensive fall prevention (alarm, sitter, PT, medication review) |
CAM (Confusion Assessment Method) — Summary
| Criteria Required | Features |
|---|---|
| Required (both) | 1. Acute onset and fluctuating course + 2. Inattention |
| Plus one of | 3. Disorganized thinking OR 4. Altered level of consciousness |
MELD Score (Model for End-Stage Liver Disease)
MELD = 3.78 × ln(bilirubin mg/dL) + 11.2 × ln(INR) + 9.57 × ln(creatinine mg/dL) + 6.43. MELD-Na incorporates serum sodium (improves prognostic accuracy). Used for: liver transplant organ allocation priority, predicting 90-day mortality in cirrhosis (MELD 10 = 6%, 20 = 20%, 30 = 53%, 40 = 80%), and guiding decisions about TIPS and other interventions. Minimum values are set to 1.0 for each variable; maximum creatinine is capped at 4.0 (and 4.0 if on dialysis).
29 Medications Master Table
This table lists the most commonly used inpatient medications by class, with dosing, indications, and clinical pearls relevant to hospitalist practice.
Antibiotics
| Drug | Class / Mechanism | Typical Dose | Indication | Pearls |
|---|---|---|---|---|
| Ceftriaxone | 3rd-gen cephalosporin; beta-lactam, PBP inhibitor | 1–2 g IV daily | CAP, UTI, meningitis, SBP prophylaxis | Biliary sludging; avoid with calcium-containing IV solutions in neonates |
| Piperacillin-tazobactam | Antipseudomonal penicillin + BLI | 4.5 g IV q6h (extended infusion over 4 h preferred) | HAP/VAP, intra-abdominal, diabetic foot, neutropenic fever | Monitor renal function when combined with vancomycin (AKI risk); extended infusion improves outcomes |
| Vancomycin | Glycopeptide; cell wall inhibitor | 15–20 mg/kg IV q8–12h; AUC/MIC 400–600 target | MRSA (bacteremia, pneumonia, endocarditis, bone/joint), C. diff (PO only) | AUC-guided dosing preferred over trough-based; nephrotoxicity with pip-tazo combo; Red Man syndrome (infuse over ≥1 h) |
| Azithromycin | Macrolide; 50S ribosomal inhibitor | 500 mg IV/PO daily (or 500 day 1, then 250 × 4d) | CAP (atypical coverage), COPD exacerbation | QT prolongation; drug interactions with statins, warfarin |
| Levofloxacin | Fluoroquinolone; DNA gyrase/topoisomerase IV inhibitor | 750 mg IV/PO daily | CAP, UTI (complicated), Legionella | Black box: tendon rupture, peripheral neuropathy, aortic dissection; avoid in elderly; C. diff risk |
| Metronidazole | Nitroimidazole; DNA disruption | 500 mg IV/PO q8h | Anaerobic infections, C. diff (fulminant — IV), intra-abdominal | Disulfiram reaction with alcohol; metallic taste; peripheral neuropathy with prolonged use |
Cardiovascular
| Drug | Mechanism | Typical Dose | Indication | Pearls |
|---|---|---|---|---|
| Furosemide | Loop diuretic; inhibits NKCC2 in thick ascending limb | 20–200 mg IV q6–12h | Acute HF, volume overload, edema | Monitor K, Mg, Cr; ototoxicity at high doses; 2.5:1 PO:IV conversion |
| Metoprolol tartrate | Selective beta-1 blocker | 5 mg IV q5min × 3 (acute); 25–50 mg PO q6h | Rate control (AFib, sinus tachycardia), ACS, HTN | Hold if HR <60, SBP <100; IV for acute rate control; switch to succinate for chronic HF |
| Amiodarone | Class III antiarrhythmic (K channel blocker + Na, Ca, beta-blocking properties) | 150 mg IV over 10 min, then 1 mg/min × 6 h, then 0.5 mg/min × 18 h | AFib, VT, post-arrest | QT prolongation; hepatotoxicity, pulmonary toxicity, thyroid dysfunction; many drug interactions (warfarin, digoxin) |
| Heparin (UFH) | Antithrombin III activator; inhibits factors IIa and Xa | See nomogram above | VTE treatment, ACS, bridging | Short half-life (1–2 h); protamine reversible; HIT risk |
| Nitroglycerin | Venodilator (reduces preload); coronary vasodilator at higher doses | 5–200 mcg/min IV infusion; 0.4 mg SL q5min × 3 | ACS, acute HF (warm-wet), hypertensive emergency | Contraindicated with PDE5 inhibitors (sildenafil — 24h; tadalafil — 48h); headache; tachyphylaxis |
Endocrine
| Drug | Mechanism | Typical Dose | Indication | Pearls |
|---|---|---|---|---|
| Insulin glargine | Long-acting basal insulin analog | 0.2–0.5 units/kg SC daily | Inpatient basal coverage for DM | No peak; 24 h duration; do not mix with other insulins; reduce by 20% if NPO |
| Insulin lispro/aspart | Rapid-acting prandial insulin analog | 0.1 units/kg SC before meals + correction scale | Prandial coverage, correction doses | Onset 15 min, peak 1 h, duration 3–4 h; hold if patient NPO |
| Regular insulin | Short-acting insulin | Drip: 0.1 units/kg/h (DKA); SC: per correction scale | DKA/HHS (IV drip), correction doses | Only insulin given IV; onset 30–60 min SC, immediate IV; overlap with SC 2 h before stopping drip |
| Dexamethasone | Glucocorticoid (anti-inflammatory, immunosuppressive) | 4–10 mg IV q6–12h | Cerebral edema, croup, anti-emetic, adrenal crisis (no mineralocorticoid effect to confound cortisol testing) | Hyperglycemia (often requires insulin); insomnia; GI upset; does not interfere with cortisol assay |
GI / Antiemetics
| Drug | Mechanism | Typical Dose | Indication | Pearls |
|---|---|---|---|---|
| Pantoprazole | Proton pump inhibitor; irreversible H+/K+ ATPase blockade | 40 mg IV/PO daily (80 mg IV bolus + 8 mg/h drip for GI bleed) | Stress ulcer prophylaxis (ventilated, coagulopathy), GI bleed, GERD | CDI risk with prolonged use; hypomagnesemia; B12/iron malabsorption; deprescribe at discharge if no clear indication |
| Ondansetron | 5-HT3 receptor antagonist | 4 mg IV/PO q6–8h | Nausea/vomiting (post-op, chemo, general) | QT prolongation (avoid if QTc >500 ms); constipation; max 16 mg IV/day per FDA |
| Lactulose | Osmotic laxative; converts NH3 to NH4+ in colon | 30 mL PO q1–2h until BM, then TID; retention enema 300 mL in 700 mL water | Hepatic encephalopathy, constipation | Target 2–3 soft BMs/day for HE; may cause hypernatremia and dehydration with excessive dosing |
Sedation / Psychiatry
| Drug | Mechanism | Typical Dose | Indication | Pearls |
|---|---|---|---|---|
| Haloperidol | Typical antipsychotic; D2 receptor antagonist | 0.5–5 mg IV/PO q4–6h PRN | Acute agitation, delirium, ICU agitation | QT prolongation (check ECG before and after); EPS risk; avoid in Parkinson disease and Lewy body dementia |
| Quetiapine | Atypical antipsychotic; D2/5-HT2 antagonist | 12.5–50 mg PO BID–TID | Delirium (hypoactive or hyperactive), insomnia in elderly | Sedating; less EPS than haloperidol; metabolic effects with chronic use; QT prolongation |
| Melatonin | MT1/MT2 receptor agonist | 1–5 mg PO at bedtime | Insomnia, delirium prevention (sleep-wake cycle restoration) | Preferred over benzodiazepines in elderly; minimal side effects; may reduce delirium incidence |
30 Abbreviations Master List
Comprehensive list of abbreviations used throughout hospital medicine documentation, orders, and communication.
| Abbreviation | Meaning |
|---|---|
| ABG | Arterial blood gas |
| ACLS | Advanced cardiovascular life support |
| ADL | Activities of daily living |
| ADHF | Acute decompensated heart failure |
| ADT | Admission, discharge, transfer |
| AECOPD | Acute exacerbation of COPD |
| AG | Anion gap |
| AIN | Acute interstitial nephritis |
| AKI | Acute kidney injury |
| AMA | Against medical advice |
| AMS | Altered mental status |
| ARNI | Angiotensin receptor-neprilysin inhibitor (sacubitril-valsartan) |
| ASB | Asymptomatic bacteriuria |
| ATN | Acute tubular necrosis |
| BiPAP | Bilevel positive airway pressure |
| BMP | Basic metabolic panel |
| BNP | B-type natriuretic peptide |
| BPH | Benign prostatic hyperplasia |
| BPMH | Best possible medication history |
| BUN | Blood urea nitrogen |
| CABG | Coronary artery bypass graft |
| CAM | Confusion Assessment Method |
| CAP | Community-acquired pneumonia |
| CAUTI | Catheter-associated urinary tract infection |
| CBC | Complete blood count |
| CC | Chief complaint; complication or comorbidity (DRG context) |
| CDI | Clinical documentation improvement; Clostridioides difficile infection |
| CHF | Congestive heart failure |
| CKD | Chronic kidney disease |
| CLABSI | Central line-associated bloodstream infection |
| CMI | Case mix index |
| CMP | Comprehensive metabolic panel |
| CMS | Centers for Medicare & Medicaid Services |
| COPD | Chronic obstructive pulmonary disease |
| CPOE | Computerized physician order entry |
| CPR | Cardiopulmonary resuscitation |
| CrCl | Creatinine clearance |
| CRP | C-reactive protein |
| CSF | Cerebrospinal fluid |
| CT | Computed tomography |
| CTPA | CT pulmonary angiography |
| CVP | Central venous pressure |
| CXR | Chest X-ray |
| D50 | Dextrose 50% (25 g/50 mL) |
| DKA | Diabetic ketoacidosis |
| DNR/DNI | Do not resuscitate / do not intubate |
| DOAC | Direct oral anticoagulant |
| DRG | Diagnosis-related group |
| DTPI | Deep tissue pressure injury |
| DVT | Deep vein thrombosis |
| ECG/EKG | Electrocardiogram |
| ED | Emergency department |
| EF | Ejection fraction |
| eGFR | Estimated glomerular filtration rate |
| EMR/EHR | Electronic medical/health record |
| FENa | Fractional excretion of sodium |
| FMT | Fecal microbiota transplant |
| FQ | Fluoroquinolone |
| GDMT | Guideline-directed medical therapy |
| GDH | Glutamate dehydrogenase |
| GI | Gastrointestinal |
| HAC | Hospital-acquired condition |
| HAP | Hospital-acquired pneumonia |
| HCAHPS | Hospital Consumer Assessment of Healthcare Providers and Systems |
| HCPOA | Healthcare power of attorney |
| HFrEF | Heart failure with reduced ejection fraction |
| HHS | Hyperosmolar hyperglycemic state |
| HIT | Heparin-induced thrombocytopenia |
| HRRP | Hospital Readmission Reduction Program |
| HTN | Hypertension |
| I&O | Intake and output |
| ICU | Intensive care unit |
| INR | International normalized ratio |
| IPO | Inpatient-only (procedure list) |
| IRF | Inpatient rehabilitation facility |
| IV | Intravenous |
| JVD | Jugular venous distension |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| LACE | Length of stay, Acuity, Comorbidities, ED visits (readmission risk index) |
| LDH | Lactate dehydrogenase |
| LMWH | Low-molecular-weight heparin |
| LOS | Length of stay |
| LP | Lumbar puncture |
| LTAC | Long-term acute care hospital |
| MCC | Major complication or comorbidity |
| MDM | Medical decision-making |
| MELD | Model for End-Stage Liver Disease |
| MME | Morphine milligram equivalents |
| MOON | Medicare Outpatient Observation Notice |
| MRA | Mineralocorticoid receptor antagonist |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| NAAT | Nucleic acid amplification test |
| NIV | Non-invasive ventilation |
| NPO | Nil per os (nothing by mouth) |
| NS | Normal saline (0.9% NaCl) |
| OIC | Opioid-induced constipation |
| PCA | Patient-controlled analgesia |
| PE | Pulmonary embolism |
| PESI | Pulmonary Embolism Severity Index |
| PF4 | Platelet factor 4 |
| PMN | Polymorphonuclear leukocyte (neutrophil) |
| POA | Present on admission |
| POLST | Physician Orders for Life-Sustaining Treatment |
| PPI | Proton pump inhibitor |
| PRN | Pro re nata (as needed) |
| PSI | Patient Safety Indicator; Pneumonia Severity Index |
| PT/OT | Physical therapy / occupational therapy |
| RAC | Recovery Audit Contractor |
| RASS | Richmond Agitation-Sedation Scale |
| RRT | Rapid Response Team; renal replacement therapy |
| SAAG | Serum-ascites albumin gradient |
| SBP | Systolic blood pressure; spontaneous bacterial peritonitis |
| SC | Subcutaneous |
| SCD | Sequential compression device |
| SGLT2i | Sodium-glucose cotransporter 2 inhibitor |
| SIADH | Syndrome of inappropriate antidiuretic hormone |
| SNF | Skilled nursing facility |
| SRA | Serotonin release assay |
| SSTI | Skin and soft tissue infection |
| TDD | Total daily dose (insulin) |
| TMP-SMX | Trimethoprim-sulfamethoxazole |
| tPA | Tissue plasminogen activator |
| UA | Urinalysis |
| UFH | Unfractionated heparin |
| UOP | Urine output |
| UR | Utilization review |
| UTI | Urinary tract infection |
| VAP | Ventilator-associated pneumonia |
| VBP | Value-based purchasing |
| VTE | Venous thromboembolism |
| WBC | White blood cell count |