Neonatology
Neonatal resuscitation, respiratory distress, prematurity management, congenital anomalies, neonatal sepsis, hyperbilirubinemia, and every scoring system, ventilator protocol, and management algorithm across the full scope of neonatal-perinatal medicine.
01 Neonatal Physiology & Transition
The transition from intrauterine to extrauterine life is the most complex physiologic adaptation that occurs in human biology. Within seconds to minutes of birth, the neonate must establish independent gas exchange, redirect circulatory pathways, maintain thermoregulation, and achieve metabolic homeostasis. Understanding fetal circulation and its postnatal transition is foundational to all neonatal care.
Fetal Circulation
Fetal circulation is a parallel circuit (unlike the postnatal series circuit) optimized to deliver the most oxygenated blood to the brain and myocardium. Three unique shunts characterize fetal hemodynamics:
| Shunt | Location | Function | Postnatal Closure |
|---|---|---|---|
| Ductus venosus | Liver — connects umbilical vein to IVC | Bypasses hepatic circulation; delivers oxygenated blood from placenta directly to IVC | Functional closure within hours; becomes ligamentum venosum |
| Foramen ovale | Interatrial septum | Shunts oxygenated blood from RA to LA, bypassing the pulmonary circuit | Functional closure within hours as LA pressure exceeds RA; anatomic closure by 3–12 months (patent in 25% of adults) |
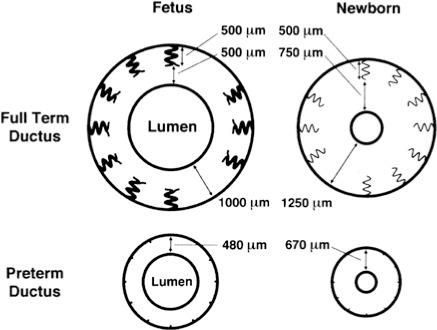
| Ductus arteriosus | Connects main pulmonary artery to descending aorta | Diverts blood away from high-resistance pulmonary vasculature to the systemic circulation | Functional closure 10–15 hours (rising PaO2 + falling prostaglandins); anatomic closure by 2–3 weeks → ligamentum arteriosum |
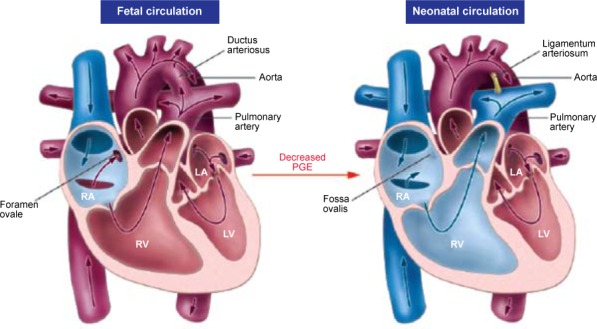
In utero, the placenta is the organ of gas exchange. The umbilical vein carries oxygenated blood (PaO2 ~30–35 mmHg) from the placenta to the fetus. Approximately 50% passes through the ductus venosus to the IVC; the remainder perfuses the hepatic sinusoids. In the IVC, this relatively oxygenated blood preferentially streams across the foramen ovale into the left atrium, then to the left ventricle and ascending aorta — supplying the coronary arteries and brain with the most oxygenated blood available. Deoxygenated blood returning via the SVC flows into the right ventricle and out the pulmonary artery, where 90% is diverted through the ductus arteriosus to the descending aorta (and back to the placenta via the umbilical arteries).
Transitional Circulation — The First Breath
Birth triggers a cascade of events that convert the parallel fetal circuit into the adult series circuit:
- Cord clamping → removes low-resistance placental circuit → systemic vascular resistance (SVR) rises → aortic pressure increases
- First breath → lung expansion → alveolar fluid clearance (driven by sodium channel activation, especially ENaC) → pulmonary vascular resistance (PVR) drops dramatically
- Rising PaO2 causes pulmonary vasodilation (via nitric oxide and prostacyclin) and ductal constriction
- Decreased PVR → increased pulmonary blood flow → increased LA return → LA pressure exceeds RA pressure → foramen ovale closes
- Rising O2 + falling prostaglandin E2 and I2 → ductus arteriosus constricts
Thermoregulation
Neonates, especially preterm infants, are extremely vulnerable to hypothermia due to:
- Large surface area-to-body mass ratio
- Thin skin with minimal subcutaneous fat insulation
- Inability to shiver (neonates rely on non-shivering thermogenesis)
- Wet skin at delivery (evaporative losses)
Brown adipose tissue (BAT) is the primary source of heat production in neonates. Located between the scapulae, around the kidneys, and along the great vessels, BAT contains high concentrations of mitochondria with uncoupling protein 1 (UCP-1, thermogenin) that dissipates the proton gradient as heat rather than ATP. BAT thermogenesis is triggered by norepinephrine via β3-adrenergic receptors. Hypothermia increases oxygen consumption and can lead to metabolic acidosis, hypoglycemia, and increased mortality — maintaining a neutral thermal environment (NTE, axillary temperature 36.5–37.5°C) is a core principle of neonatal care.
Glucose Homeostasis
The fetus receives a continuous glucose supply via the placenta. At birth, this supply is abruptly terminated, and the neonate must rapidly activate glycogenolysis and gluconeogenesis. Term infants normally experience a physiologic nadir in blood glucose (as low as 30 mg/dL) at 1–2 hours of life, which rises spontaneously by 3–4 hours with feeding. Preterm infants, SGA infants, and infants of diabetic mothers (IDM) are at high risk for hypoglycemia due to limited glycogen stores, increased metabolic demands, and (in IDM) hyperinsulinism from fetal exposure to maternal hyperglycemia.
Evaporation — wet skin at delivery; primary source of heat loss in the DR. Convection — air currents over exposed skin. Conduction — contact with cold surfaces (scales, mattresses). Radiation — heat transfer to cooler surrounding objects (walls, windows). Preventive measures: immediate drying, skin-to-skin contact, radiant warmers, polyethylene wraps for preterm infants.
02 APGAR Score & Neonatal Assessment
The APGAR score, introduced by Virginia Apgar in 1952, is the most widely used rapid assessment tool for neonatal transition. It is performed at 1 and 5 minutes of life (and at 5-minute intervals thereafter if the score remains <7). The APGAR does not determine the need for resuscitation — resuscitation should begin before the 1-minute score is assigned.
APGAR Score Components
| Sign | 0 Points | 1 Point | 2 Points |
|---|---|---|---|
| Appearance (color) | Blue or pale all over | Acrocyanosis (blue extremities, pink body) | Completely pink |
| Pulse (heart rate) | Absent | <100 bpm | ≥100 bpm |
| Grimace (reflex irritability) | No response to stimulation | Grimace or weak cry with stimulation | Vigorous cry, cough, or sneeze with stimulation |
| Activity (muscle tone) | Limp, flaccid | Some flexion of extremities | Active motion, good flexion |
| Respiration | Absent | Slow, irregular, weak cry | Good, strong cry |
7–10: Normal — routine care. 4–6: Moderately depressed — requires stimulation and possibly PPV. 0–3: Severely depressed — requires immediate resuscitation. A 5-minute APGAR of 0–3 is associated with increased neonatal mortality but is a poor predictor of long-term neurologic outcome in isolation. The APGAR should never be used to withhold resuscitation or to diagnose asphyxia.
New Ballard Score — Gestational Age Assessment
The New Ballard Score (NBS) estimates gestational age from 20 to 44 weeks based on six neuromuscular and six physical maturity criteria. It is accurate to ±2 weeks and is most reliable when performed within 48 hours of birth (up to 96 hours for premature infants). Each criterion is scored from −1 to 5 (neuromuscular) or −1 to 4 (physical); the total is mapped to gestational age.
| Neuromuscular Criteria | Physical Maturity Criteria |
|---|---|
| Posture | Skin (texture, opacity, peeling) |
| Square window (wrist flexion) | Lanugo |
| Arm recoil | Plantar surface (creases) |
| Popliteal angle | Breast tissue |
| Scarf sign | Eye/ear maturity (pinna, eyelid fusion) |
| Heel-to-ear | Genitalia (male: rugae, testes; female: labia, clitoris) |
Growth Classification
Birth weight is classified relative to gestational age using population-based growth curves (Fenton or Olsen charts):
- Small for gestational age (SGA): Birth weight <10th percentile for gestational age
- Appropriate for gestational age (AGA): Birth weight 10th–90th percentile
- Large for gestational age (LGA): Birth weight >90th percentile
SGA infants are at risk for hypoglycemia, polycythemia, hypothermia, and may have underlying IUGR (intrauterine growth restriction) from placental insufficiency, maternal hypertension, or congenital infection. LGA infants are commonly seen in maternal diabetes and are at risk for birth trauma (shoulder dystocia, brachial plexus injury), hypoglycemia, and polycythemia.
03 Terminology & Abbreviations
Neonatology uses a specialized vocabulary for gestational age classification, birth weight categories, and clinical conditions. Mastery of these terms is essential for documentation, communication, and interpreting the neonatal literature.
| Term | Definition |
|---|---|
| Preterm | Born <37 weeks’ gestation |
| Late preterm | Born 34 0/7 – 36 6/7 weeks |
| Early term | Born 37 0/7 – 38 6/7 weeks |
| Full term | Born 39 0/7 – 40 6/7 weeks |
| Late term | Born 41 0/7 – 41 6/7 weeks |
| Post-term | Born ≥42 0/7 weeks |
| LBW | Low birth weight: <2,500 g |
| VLBW | Very low birth weight: <1,500 g |
| ELBW | Extremely low birth weight: <1,000 g |
| Corrected gestational age (CGA) | Chronologic age minus weeks of prematurity; used for developmental milestones until age 2–3 |
| Postmenstrual age (PMA) | Gestational age + postnatal age in weeks |
| DOL | Day of life (DOL 0 = birth day) |
| GIR | Glucose infusion rate (mg/kg/min) |
| TPN | Total parenteral nutrition |
| NEC | Necrotizing enterocolitis |
| RDS | Respiratory distress syndrome |
| BPD | Bronchopulmonary dysplasia |
| IVH | Intraventricular hemorrhage |
| PVL | Periventricular leukomalacia |
| ROP | Retinopathy of prematurity |
| PDA | Patent ductus arteriosus |
| PPHN | Persistent pulmonary hypertension of the newborn |
04 NRP Algorithm
Approximately 10% of newborns require some assistance to begin breathing, and about 1% require extensive resuscitation. The Neonatal Resuscitation Program (NRP), updated in the 8th edition (2020), provides an evidence-based, algorithmic approach. Every delivery should have at least one person skilled in NRP present whose sole responsibility is the newborn.
Initial Assessment (Birth)
Three rapid questions at birth determine the initial approach:
- Term gestation?
- Good muscle tone?
- Breathing or crying?
If yes to all three → routine care: skin-to-skin, dry, clear airway as needed, ongoing assessment. If no to any → move to the radiant warmer and begin initial steps.
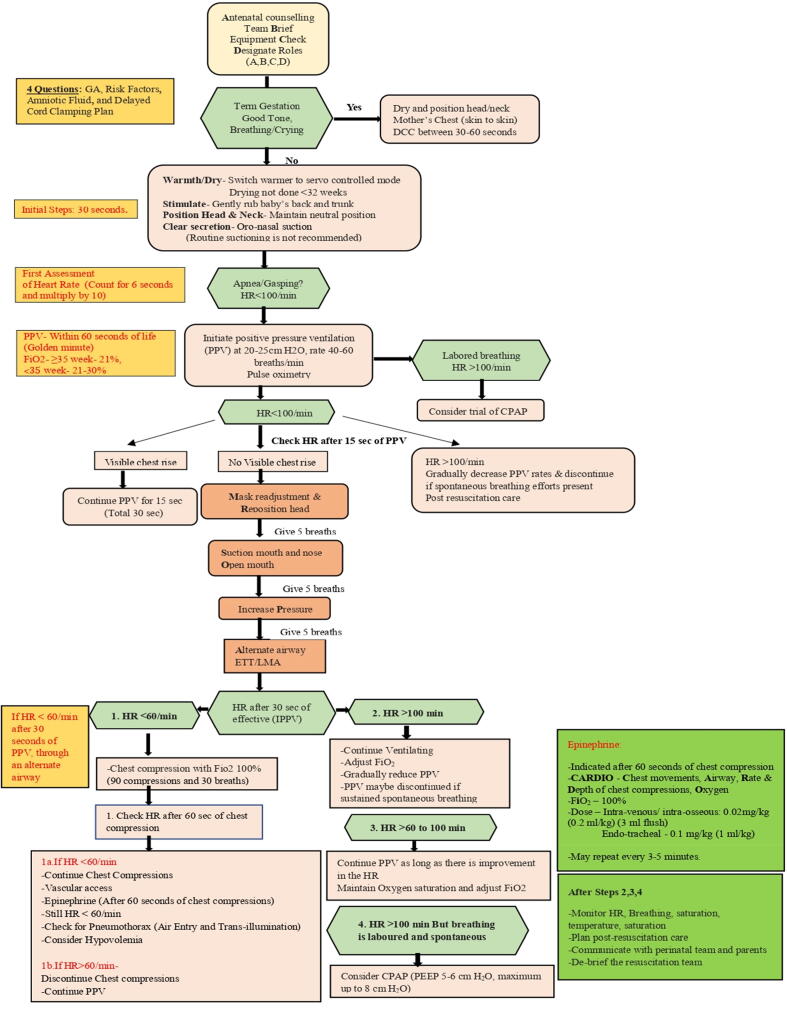
Initial Steps (within 30 seconds)
Warm (radiant warmer, dry, remove wet linen, polyethylene wrap for <32 weeks), position (sniffing position, shoulder roll if needed), clear airway (gentle suctioning only if secretions obstruct; mouth before nose), dry, and stimulate (flick soles, rub back).
The initial steps should be completed within 60 seconds of birth. By 60 seconds, the clinician should have assessed the infant’s response and, if the heart rate is <100 bpm or the infant is apneic/gasping, initiated positive pressure ventilation (PPV). Effective ventilation is the single most important and effective step in neonatal resuscitation.
Positive Pressure Ventilation (PPV)
If HR <100 bpm or apneic/gasping after initial steps → begin PPV at 40–60 breaths/min using a T-piece resuscitator or self-inflating bag with a correctly sized mask (covers mouth and nose but not eyes). Initial PIP: 20–25 cm H2O (up to 30–40 for the first few breaths if needed to establish FRC). Initial FiO2: 21% for ≥35 weeks; 21–30% for <35 weeks. Titrate by pulse oximetry.
MR SOPA — Corrective Steps for Ineffective PPV
| Letter | Corrective Action |
|---|---|
| M | Mask adjustment — ensure good seal |
| R | Reposition — sniffing position, shoulder roll |
| S | Suction mouth then nose |
| O | Open mouth — open the mouth slightly during PPV |
| P | Pressure increase — increase PIP incrementally |
| A | Alternative airway — endotracheal tube or laryngeal mask |
Chest Compressions
If HR <60 bpm despite 30 seconds of effective PPV (with adequate chest rise) → begin chest compressions. Use the two-thumb encircling technique (preferred): compress the lower third of the sternum to a depth of one-third the AP diameter. Ratio: 3 compressions : 1 ventilation (3:1), delivering 90 compressions + 30 ventilations = 120 events per minute. Increase FiO2 to 100% when compressions are initiated.
Epinephrine & Vascular Access
If HR remains <60 bpm despite effective PPV + compressions for 60 seconds → administer epinephrine:
- IV (UVC preferred): 0.01–0.03 mg/kg (0.1–0.3 mL/kg of 1:10,000) — may repeat every 3–5 minutes
- Endotracheal: 0.05–0.1 mg/kg (0.5–1 mL/kg of 1:10,000) — only while IV access is being established
An umbilical venous catheter (UVC) is the preferred emergency vascular access in the delivery room. Insert through the umbilical vein (thin-walled, single vessel) to a depth where blood can be freely aspirated (typically 2–4 cm in a term infant — just past the abdominal wall).
Meconium-Stained Amniotic Fluid
The 2020 NRP no longer recommends routine intubation and tracheal suctioning for non-vigorous infants born through meconium-stained fluid. Instead, follow the standard NRP algorithm: provide initial steps and PPV if needed. Intubation and tracheal suctioning should be considered only if the airway appears obstructed despite proper PPV technique.
05 Delivery Room Stabilization
Delayed Cord Clamping
Delayed cord clamping (DCC) is recommended for all vigorous newborns who do not require immediate resuscitation:
- Term infants: Delay clamping for at least 30–60 seconds; improves iron stores, reduces iron deficiency anemia at 3–6 months
- Preterm infants: Delay clamping for at least 30 seconds; reduces IVH, NEC, need for transfusion, and improves transitional circulation
If DCC is not possible (non-vigorous infant, placental abruption), umbilical cord milking (stroking the cord 3–4 times toward the infant over 2 seconds) is an alternative, though evidence is less robust and it is not recommended for infants <28 weeks due to a possible association with increased IVH.
Temperature Management
Target admission temperature: 36.5–37.5°C. Hypothermia on NICU admission is an independent predictor of mortality and morbidity in preterm infants. Strategies by gestational age:
| Population | Temperature Interventions |
|---|---|
| ≥32 weeks | Radiant warmer, immediate drying, skin-to-skin if vigorous, warm blankets, hat |
| <32 weeks | All of the above PLUS: polyethylene wrap/bag (place undried infant from shoulders down into bag), increase delivery room temperature to 23–25°C, heated mattress, humidified gases for respiratory support |
Target SpO2 by Minute of Life
Healthy term newborns do not achieve SpO2 >90% until approximately 10 minutes after birth. The NRP provides target preductal SpO2 ranges (right hand):
| Time After Birth | Target SpO2 |
|---|---|
| 1 minute | 60–65% |
| 2 minutes | 65–70% |
| 3 minutes | 70–75% |
| 4 minutes | 75–80% |
| 5 minutes | 80–85% |
| 10 minutes | 85–95% |
Initial FiO2 Recommendations
- ≥35 weeks GA: Start at 21% (room air); titrate by pulse oximetry
- <35 weeks GA: Start at 21–30%; titrate to target SpO2 ranges above
- Avoid hyperoxia (>95%) in preterm infants — increases ROP risk and oxidative stress
06 Perinatal Asphyxia & HIE
Hypoxic-ischemic encephalopathy (HIE) results from impaired cerebral blood flow and oxygen delivery around the time of birth, leading to a cascade of excitotoxicity, oxidative stress, inflammation, and apoptosis. It occurs in 1–3 per 1,000 term live births and remains a leading cause of neonatal death and long-term neurodisability.
Sarnat Staging of HIE
| Feature | Stage I (Mild) | Stage II (Moderate) | Stage III (Severe) |
|---|---|---|---|
| Level of consciousness | Hyperalert, irritable | Lethargic, obtunded | Stuporous, comatose |
| Muscle tone | Normal or mildly increased | Hypotonia | Flaccid |
| Posture | Mild distal flexion | Strong distal flexion | Intermittent decerebration |
| Tendon reflexes | Hyperactive | Hyperactive | Absent |
| Myoclonus | Present | Present | Absent |
| Suck reflex | Weak | Weak or absent | Absent |
| Moro reflex | Strong, low threshold | Weak, incomplete | Absent |
| Pupils | Mydriasis | Miosis | Unequal, poor light reflex |
| Seizures | None | Common (within 24 h) | Uncommon (decerebrate posturing may be mistaken for seizures) |
| EEG | Normal | Low-voltage, seizure activity | Burst suppression or isoelectric |
| Duration | <24 hours | 2–14 days | Hours to weeks |
| Outcome | Good — normal | Variable — 20–40% disability | Poor — >50% mortality; survivors have severe disability |
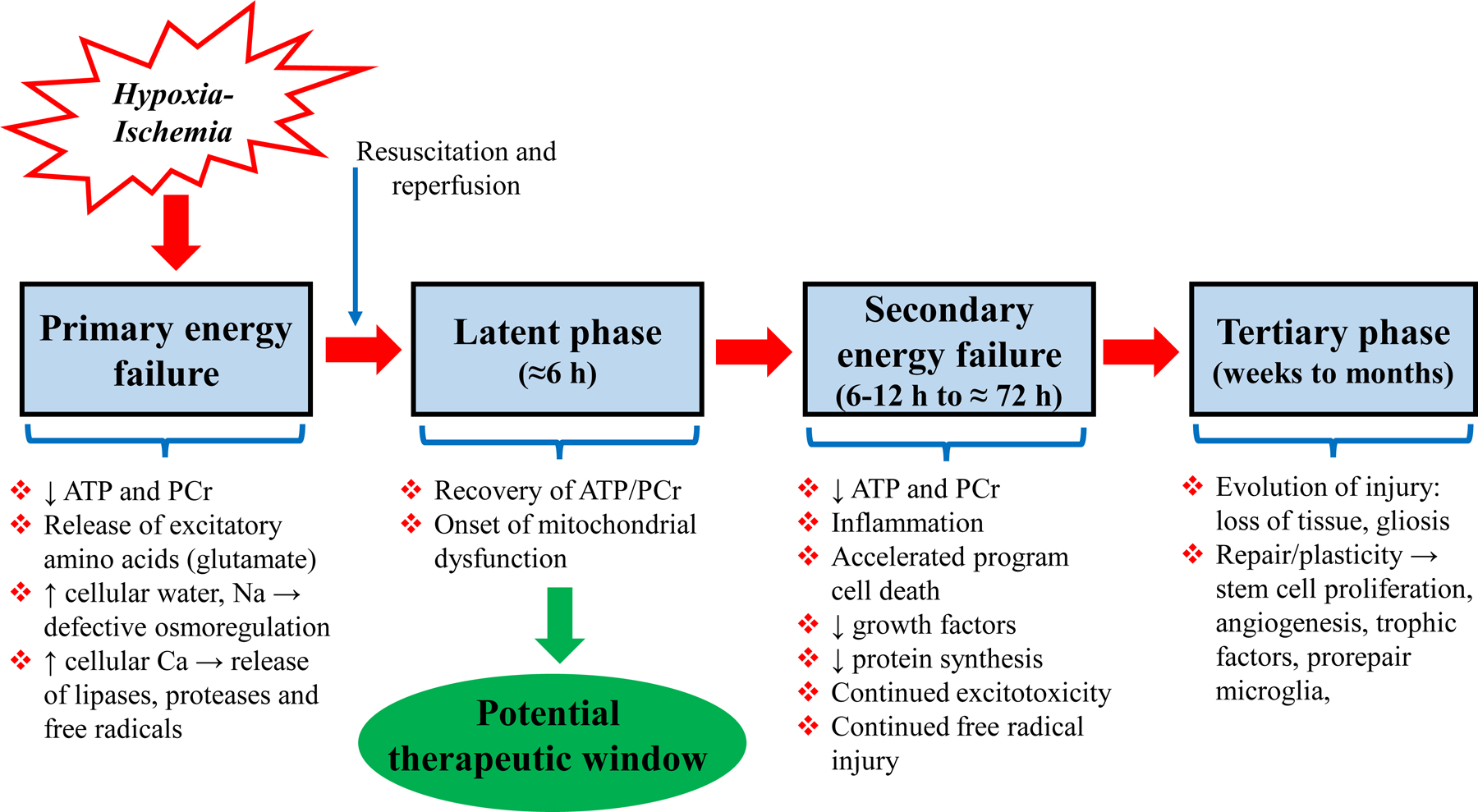
Therapeutic Hypothermia
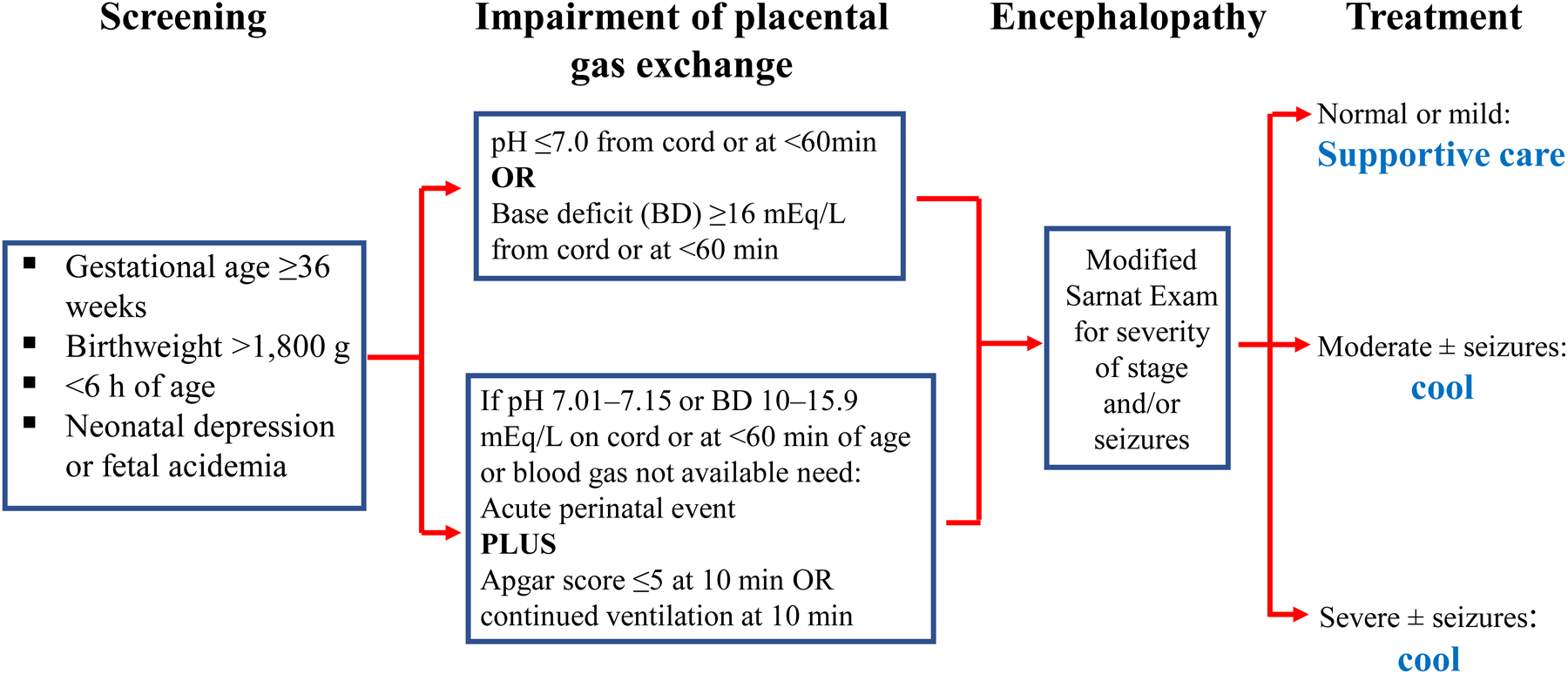
Therapeutic hypothermia (TH) is the only proven neuroprotective intervention for moderate-to-severe HIE. It reduces death and major neurodisability by approximately 25%.
Eligibility criteria: ≥36 weeks GA, ≤6 hours of age, evidence of perinatal asphyxia (any of: Apgar ≤5 at 10 min, continued need for resuscitation at 10 min, pH <7.0 or base deficit ≥16 on cord/arterial blood gas within 60 min of birth) PLUS moderate-to-severe encephalopathy on clinical exam or abnormal aEEG.
Target temperature: 33.5°C (±0.5°C) — whole body cooling (or 34.5°C for selective head cooling).
Duration: 72 hours of cooling followed by slow rewarming at 0.5°C per hour over 6–12 hours.
Monitoring during cooling: Continuous rectal/esophageal temperature, cardiac monitor (sinus bradycardia is expected), blood pressure, blood gases, electrolytes (watch for hypokalemia during rewarming), coagulation, liver/renal function, aEEG or continuous EEG.
Amplitude-Integrated EEG (aEEG)
aEEG provides continuous bedside monitoring of cerebral electrical activity. Background patterns classified as: continuous normal voltage (CNV, best prognosis), discontinuous normal voltage (DNV), burst suppression, continuous low voltage, and flat trace (worst prognosis). aEEG within 6 hours is used to help determine eligibility for TH and predict outcomes. Seizures appear as sudden rises in both upper and lower margins.
07 Respiratory Distress Syndrome
Respiratory distress syndrome (RDS), formerly known as hyaline membrane disease, is caused by deficiency of pulmonary surfactant, a complex mixture of phospholipids (primarily dipalmitoylphosphatidylcholine/DPPC) and surfactant proteins (SP-A, SP-B, SP-C, SP-D) produced by type II pneumocytes. Surfactant reduces alveolar surface tension, prevents end-expiratory atelectasis, and improves lung compliance. RDS is the leading cause of respiratory failure in premature infants.
Risk Factors
- Prematurity — the primary risk factor; surfactant production begins at ~24 weeks and is adequate by ~35 weeks
- Maternal diabetes (insulin inhibits surfactant production)
- Male sex (androgens delay surfactant maturation)
- Cesarean delivery without labor (lack of catecholamine surge)
- Perinatal asphyxia, hypothermia
- Multiple gestation (second twin)
Clinical Features & Diagnosis
Onset within minutes to hours of birth: tachypnea, nasal flaring, intercostal/subcostal retractions, expiratory grunting (attempts to maintain FRC by adducting vocal cords), cyanosis. Progressive worsening over 48–72 hours without treatment, then gradual improvement as endogenous surfactant production increases.
CXR findings: Diffuse, bilateral, reticulogranular (“ground-glass”) opacities with air bronchograms, decreased lung volumes. Severe cases may show complete “white-out” with indistinguishable cardiac silhouette.
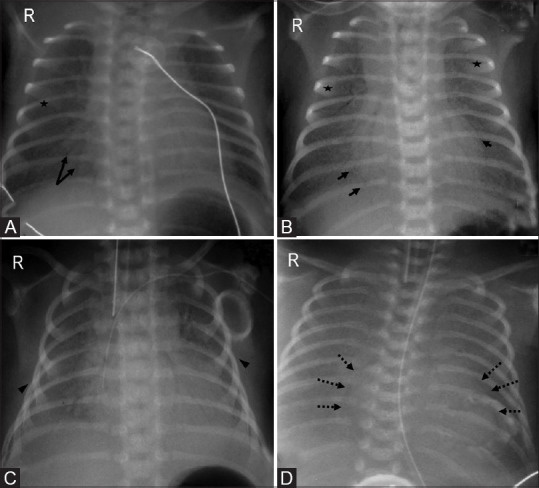
Management
| Intervention | Details |
|---|---|
| CPAP | First-line for mild-moderate RDS; 5–8 cm H2O via nasal prongs or mask; maintains FRC and reduces surfactant need |
| Surfactant replacement | Natural surfactants preferred (poractant alfa/Curosurf, beractant/Survanta, calfactant/Infasurf); dose varies by product (poractant: 200 mg/kg initial, 100 mg/kg repeat; beractant: 100 mg/kg × 4 doses) |
| INSURE technique | INtubate, SURfactant, Extubate to CPAP — reduces duration of mechanical ventilation |
| LISA/MIST technique | Less Invasive Surfactant Administration: thin catheter placed in trachea during spontaneous breathing on CPAP, surfactant instilled; avoids intubation; increasingly preferred method |
| Mechanical ventilation | For severe RDS failing CPAP; use gentle ventilation with lowest effective pressures |
Prevention
Antenatal corticosteroids are the most effective intervention for preventing RDS: betamethasone 12 mg IM × 2 doses 24 hours apart (or dexamethasone 6 mg IM × 4 doses 12 hours apart) given to mothers at 24–34 weeks gestation with threatened preterm delivery. Reduces RDS incidence by ~50%, IVH, NEC, and neonatal mortality.
08 Transient Tachypnea & Meconium Aspiration
Transient Tachypnea of the Newborn (TTN)
TTN (“wet lung”) is the most common cause of respiratory distress in term and late preterm newborns, caused by delayed clearance of fetal lung fluid. Normally, labor triggers catecholamine-mediated activation of epithelial sodium channels (ENaC) that reabsorb lung fluid. Cesarean delivery without labor bypasses this mechanism.
- Risk factors: Elective cesarean without labor, precipitous delivery, maternal diabetes, maternal sedation, late preterm birth
- Presentation: Tachypnea (RR 60–120), mild retractions, occasional grunting within the first 2 hours; usually self-resolving within 24–72 hours
- CXR: Perihilar streaking, fluid in fissures, hyperinflation, flattened diaphragms
- Management: Supportive — supplemental O2 (rarely >40%), CPAP if needed; NPO until RR <60–80; self-resolving
Meconium Aspiration Syndrome (MAS)
Meconium-stained amniotic fluid (MSAF) occurs in 8–15% of deliveries, but MAS develops in only 2–5% of those. Meconium passage is a sign of fetal maturity (rarely occurs before 34 weeks) or fetal distress (hypoxia triggers vagal stimulation → passage of meconium, followed by gasping → aspiration).
Pathophysiology: Meconium causes (1) mechanical airway obstruction with ball-valve effect → air trapping and pneumothorax, (2) chemical pneumonitis from bile acids and enzymes, (3) surfactant inactivation, and (4) pulmonary vasoconstriction → PPHN.
| Severity | Clinical Features | Management |
|---|---|---|
| Mild | Tachypnea, mild O2 need (<40%) | Supplemental O2, observation |
| Moderate | Significant respiratory distress, O2 40–70% | CPAP or mechanical ventilation; consider surfactant |
| Severe | Respiratory failure, PPHN | Mechanical ventilation (avoid high PEEP — air trapping risk), iNO, surfactant lavage (investigational), ECMO if refractory |
Persistent Pulmonary Hypertension of the Newborn (PPHN)
PPHN occurs when PVR remains elevated after birth, causing right-to-left shunting through the foramen ovale and ductus arteriosus. Etiologies: MAS (most common), RDS, pneumonia, CDH, idiopathic. Presents with labile hypoxemia, preductal-postductal SpO2 difference >5–10%, and poor response to supplemental O2.
- Inhaled nitric oxide (iNO): Selective pulmonary vasodilator; dose 20 ppm; reduces need for ECMO; wean by 5 ppm decrements when FiO2 <60%; do not discontinue abruptly (rebound PHN)
- Sildenafil: PDE-5 inhibitor; 0.5–2 mg/kg PO q6–8h; used as adjunct or when iNO is unavailable
- Milrinone: PDE-3 inhibitor/inodilator; improves RV function; load 50 mcg/kg then 0.33–0.99 mcg/kg/min
- ECMO: Last resort for refractory PPHN; criteria: oxygenation index (OI) >40, ≥34 weeks GA, weight >2 kg
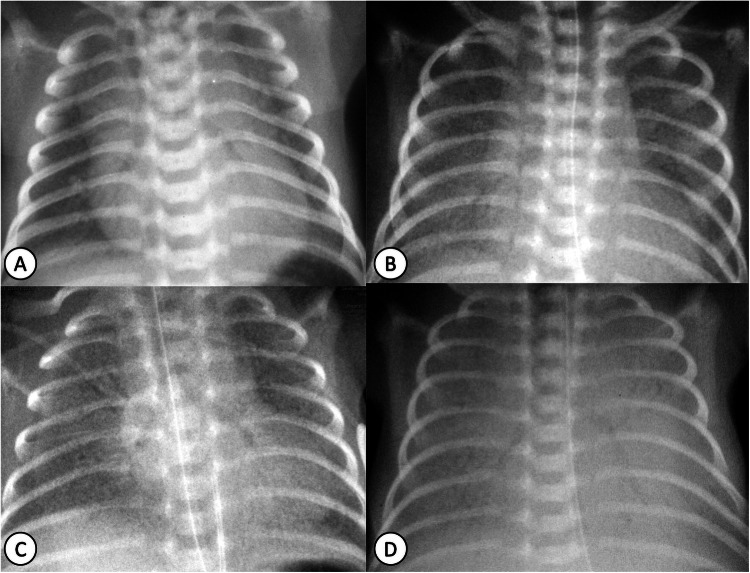
09 Bronchopulmonary Dysplasia
Bronchopulmonary dysplasia (BPD) is the most common chronic lung disease of infancy, resulting from arrested alveolar and vascular development in the immature lung, compounded by inflammation from mechanical ventilation, oxygen toxicity, and infection.
Definition & Grading
The 2001 NICHD definition classifies BPD in infants born <32 weeks based on respiratory support at 36 weeks PMA (or discharge, whichever comes first):
| Severity | Criteria at 36 Weeks PMA |
|---|---|
| Mild | Room air |
| Moderate | Need for <30% O2 |
| Severe | Need for ≥30% O2 and/or positive pressure (CPAP/ventilator) |
The Jensen 2019 evidence-based definition simplifies grading based solely on the mode of respiratory support at 36 weeks PMA, regardless of FiO2:
| Grade | Respiratory Support at 36 Weeks PMA |
|---|---|
| Grade 1 | Nasal cannula ≤2 L/min |
| Grade 2 | Nasal cannula >2 L/min, CPAP, or NIV |
| Grade 3 | Invasive mechanical ventilation |
Prevention
- Caffeine — the CAP trial demonstrated a significant reduction in BPD; start within 24 hours of birth
- Vitamin A — 5,000 IU IM 3 times/week for 4 weeks; modest BPD reduction in ELBW infants
- Gentle ventilation — target lowest effective pressures and tidal volumes (4–6 mL/kg); permissive hypercapnia (PaCO2 45–55 mmHg)
- Early CPAP — avoid intubation when possible; CPAP from delivery room
- Judicious oxygen use — target SpO2 91–95% in preterm infants
Management of Established BPD
- Diuretics: Furosemide 1–2 mg/kg/day or chlorothiazide 10–20 mg/kg/dose BID + spironolactone 1–2 mg/kg/day; improves lung compliance but no proven mortality benefit
- Inhaled corticosteroids: Budesonide nebulized; may reduce need for systemic steroids
- Postnatal dexamethasone: Facilitates extubation in ventilator-dependent infants; DART protocol (0.15 mg/kg/day tapered over 10 days); use cautiously due to concerns about neurodevelopmental adverse effects; reserve for infants unable to wean from mechanical ventilation
- Nutritional optimization: Caloric needs increased (120–150 kcal/kg/day); fortified breast milk or 24–27 kcal/oz formula
- Discharge on home O2: If SpO2 <92% in room air; with home pulse oximetry; wean as tolerated at follow-up
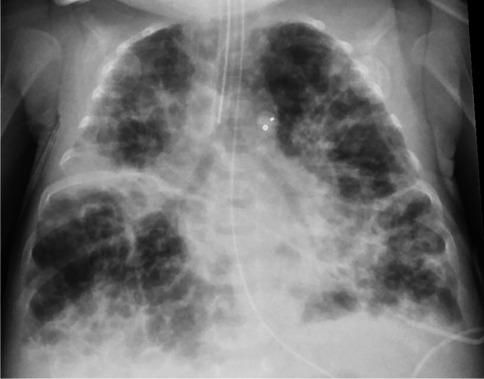
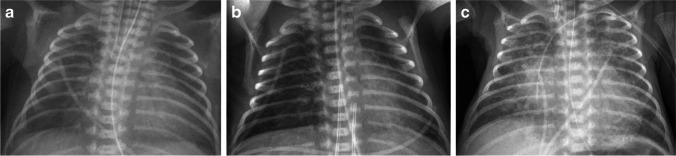
10 Neonatal Ventilation
Respiratory support in the NICU spans a spectrum from non-invasive strategies (CPAP, HFNC) to conventional mechanical ventilation and high-frequency oscillatory/jet ventilation. The principle of lung-protective ventilation — using the minimum support necessary to maintain acceptable gas exchange — is paramount to reducing ventilator-induced lung injury and BPD.
Non-Invasive Respiratory Support
| Mode | Mechanism & Settings | Indications |
|---|---|---|
| Bubble CPAP | Continuous distending pressure via nasal prongs; pressure determined by depth of tube submerged in water (typically 5–8 cm); oscillations may enhance gas exchange | First-line for RDS in very preterm infants; post-extubation support |
| Ventilator CPAP | Pressure-regulated CPAP delivered by ventilator circuit; more precise pressure control | Same as bubble CPAP; some centers prefer for consistency |
| NIPPV | Nasal intermittent positive pressure ventilation; delivers backup rate on top of CPAP | Apnea failing CPAP; post-extubation (reduces reintubation vs CPAP alone) |
| HFNC | High-flow nasal cannula; 2–8 L/min humidified; generates variable distending pressure | Mild RDS, post-extubation; possibly less nasal trauma than CPAP |
Conventional Mechanical Ventilation
When non-invasive support fails (persistent apnea, rising FiO2, worsening acidosis), intubation and mechanical ventilation are indicated.
| Mode | Description | Typical Initial Settings |
|---|---|---|
| Assist/Control (AC) | Patient-triggered; every breath receives full support; backup rate if apnea | PIP 15–25, PEEP 4–6, Rate 30–40, Ti 0.3–0.4 s, Vt target 4–6 mL/kg |
| SIMV | Synchronized intermittent mandatory ventilation; set number of supported breaths; spontaneous breaths unsupported or with PS | Rate 20–40, PEEP 4–6, PIP 15–25, PS 6–10 for spontaneous breaths |
| Volume guarantee (VG) | Targets a set tidal volume by adjusting PIP breath-to-breath; reduces volutrauma | Vt 4–6 mL/kg, PIP max 25–30, PEEP 4–6 |
High-Frequency Ventilation
| Mode | Mechanism | Indications |
|---|---|---|
| HFOV | High-frequency oscillatory ventilation; active inspiration and expiration; frequency 8–15 Hz; MAP determines lung recruitment; amplitude (delta P) determines ventilation | Air leak syndromes, pulmonary interstitial emphysema, refractory hypoxemia on conventional ventilation, severe RDS |
| HFJV | High-frequency jet ventilation; delivers short pulses of gas; passive expiration; rate 240–660/min; used with conventional ventilator providing PEEP and sigh breaths | Air leak syndromes, PIE, rescue from failing conventional ventilation |
Weaning Strategies
Wean FiO2 first to <30–40%, then reduce PIP/MAP as tolerated. Target permissive hypercapnia (PaCO2 45–55 mmHg, pH >7.22) to minimize volutrauma and allow lower ventilator settings. Extubation criteria: low ventilator settings (MAP <8, FiO2 <30%, rate <15–20), adequate spontaneous respiratory effort, caffeine on board. Extubate to CPAP or NIPPV.
<1 kg / <28 wk: 2.5 mm. 1–2 kg / 28–34 wk: 3.0 mm. 2–3 kg / 34–38 wk: 3.5 mm. >3 kg / >38 wk: 3.5–4.0 mm. Insert depth (cm at lip): 6 + weight (kg) for oral ETT. Confirm placement with CXR (tip at T1–T2), colorimetric CO2 detector, and clinical assessment.
11 Neonatal Sepsis
Neonatal sepsis is a clinical syndrome of systemic infection in the first 28 days of life. It is a leading cause of neonatal morbidity and mortality, particularly in preterm infants. Classification by timing of onset has critical implications for microbiology, evaluation, and empiric therapy.
Early-Onset Sepsis (EOS) vs. Late-Onset Sepsis (LOS)
| Feature | Early-Onset (≤72 hours) | Late-Onset (>72 hours) |
|---|---|---|
| Incidence | 0.5–1 per 1,000 live births (term); 10–15 per 1,000 (VLBW) | Up to 20–30% of VLBW infants |
| Source | Vertical (ascending from birth canal) | Nosocomial or community (central lines, hands, equipment) |
| Organisms | GBS (#1), E. coli (#2, especially in preterm), Listeria monocytogenes | CoNS (#1 in NICU), S. aureus, Gram-negatives (Klebsiella, Pseudomonas, Serratia), Candida |
| Risk factors | Prolonged ROM >18 h, maternal GBS+, chorioamnionitis, preterm birth, maternal fever | Prematurity, central venous catheters, prolonged TPN, prolonged antibiotics, NEC, surgery |
| Empiric antibiotics | Ampicillin + gentamicin | Vancomycin + cefotaxime (or cefepime/piperacillin-tazobactam if Pseudomonas suspected); add fluconazole if Candida suspected |
GBS Prophylaxis
Universal screening at 36 0/7–37 6/7 weeks with rectovaginal culture. Intrapartum antibiotic prophylaxis (IAP) with penicillin G (5 million units IV loading, then 2.5–3 million q4h) or ampicillin (2 g IV loading, then 1 g q4h) is given to GBS-positive mothers, those with GBS bacteriuria in current pregnancy, or prior infant with GBS disease. Adequate prophylaxis = ≥4 hours before delivery.
Evaluation & Workup
- Blood culture: Minimum 1 mL in a single aerobic bottle (sensitivity ~80%); the gold standard
- CBC with differential: WBC <5,000 or >30,000, I:T ratio >0.2 (immature:total neutrophils), thrombocytopenia, neutropenia
- CRP: Low sensitivity at onset; serial CRP at 24–48 hours has high NPV (>99%); useful for guiding antibiotic discontinuation
- Procalcitonin: Rises earlier than CRP; physiologic surge in first 24–48 hours limits utility in EOS
- Lumbar puncture: Indicated if blood culture positive, clinical signs of meningitis, or infant deteriorating; CSF: WBC >20–30 cells, protein >150 mg/dL (term) or >200 mg/dL (preterm), glucose <60% of serum
Duration of Therapy
- Bacteremia without focus: 7–10 days
- Meningitis (GBS): 14–21 days
- Gram-negative meningitis: 21 days (or 14 days after first sterile CSF culture)
- Negative cultures, well infant: Discontinue empiric antibiotics at 36–48 hours
12 TORCH Infections
TORCH is a mnemonic for congenital infections that share overlapping clinical features: intrauterine growth restriction, hepatosplenomegaly, jaundice, thrombocytopenia, rash, and central nervous system involvement. Each infection has unique characteristics that guide diagnosis and treatment.
Toxoplasmosis
| Feature | Details |
|---|---|
| Organism | Toxoplasma gondii (intracellular parasite) |
| Transmission | Transplacental; maternal primary infection (undercooked meat, cat feces); risk of fetal infection increases with GA (10% 1st trimester, 60% 3rd trimester) but severity decreases |
| Classic triad | Chorioretinitis, intracranial calcifications (diffuse/scattered), hydrocephalus |
| Diagnosis | IgM and IgA in infant serum; PCR of amniotic fluid or blood; serial IgG to confirm (persistence beyond 12 months) |
| Treatment | Pyrimethamine + sulfadiazine + folinic acid for 12 months; steroids if active chorioretinitis or CSF protein >1 g/dL |
Rubella
| Feature | Details |
|---|---|
| Organism | Rubella virus (togavirus) |
| Transmission | Transplacental; highest risk if maternal infection in first trimester (>80% risk of congenital rubella syndrome) |
| Classic triad | Sensorineural hearing loss (most common single finding), cataracts (or glaucoma, salt-and-pepper retinopathy), congenital heart disease (PDA, peripheral pulmonary stenosis) |
| Other features | “Blueberry muffin” rash (dermal erythropoiesis), hepatosplenomegaly, thrombocytopenia, radiolucent bone disease |
| Diagnosis | Rubella-specific IgM in infant; virus isolation from nasopharynx (contagious up to 1 year) |
| Treatment | No specific antiviral; supportive care; prevention via maternal MMR vaccination before pregnancy |
Cytomegalovirus (CMV)
| Feature | Details |
|---|---|
| Organism | CMV (herpesvirus); most common congenital infection (0.5–1% of all live births) |
| Transmission | Transplacental (primary infection most dangerous; 30–40% transmission rate); also perinatal (breast milk, birth canal) and postnatal |
| Symptomatic at birth (10–15%) | Petechiae, hepatosplenomegaly, jaundice, microcephaly, periventricular calcifications, SNHL, chorioretinitis, thrombocytopenia |
| Asymptomatic at birth (85–90%) | 10–15% develop late sequelae, primarily sensorineural hearing loss (most common infectious cause of SNHL) |
| Diagnosis | Urine or saliva CMV PCR or culture within first 21 days of life (after 21 days, cannot distinguish congenital from perinatal acquisition) |
| Treatment | Valganciclovir 16 mg/kg/dose PO BID × 6 months (or IV ganciclovir 6 mg/kg/dose BID if unable to take PO); for symptomatic congenital CMV with CNS involvement; monitor CBC (neutropenia) and LFTs |
Herpes Simplex Virus (HSV)
| Feature | Details |
|---|---|
| Organism | HSV-1 or HSV-2; HSV-2 accounts for ~75% of neonatal cases |
| Transmission | Intrapartum (85%) via contact with genital lesions; highest risk with primary maternal infection near delivery (30–50% transmission vs <2% with recurrent) |
| Presentations | SEM (skin, eye, mouth) — vesicular rash, keratoconjunctivitis; CNS — seizures, lethargy, CSF pleocytosis; Disseminated — hepatitis, DIC, pneumonitis, multi-organ failure (highest mortality ~30%) |
| Diagnosis | HSV PCR of CSF and blood (surface cultures of vesicles also helpful); LFTs, CBC, coagulation studies |
| Treatment | Acyclovir 60 mg/kg/day IV divided q8h × 14 days (SEM) or 21 days (CNS/disseminated); followed by oral acyclovir suppressive therapy 300 mg/m2/dose TID × 6 months |
Syphilis
| Feature | Details |
|---|---|
| Organism | Treponema pallidum |
| Transmission | Transplacental, any stage of pregnancy; risk highest with primary/secondary maternal syphilis (60–100%) |
| Early features (<2 yr) | Hepatosplenomegaly, mucocutaneous lesions (snuffles, maculopapular rash on palms/soles), osteochondritis/periostitis, hemolytic anemia, thrombocytopenia, nephrotic syndrome |
| Late features (>2 yr) | Hutchinson triad: Hutchinson teeth, interstitial keratitis, CN VIII deafness; also saddle nose, frontal bossing, saber shins, Clutton joints |
| Diagnosis | Maternal RPR/VDRL screening + FTA-ABS confirmation; infant: RPR/VDRL titer ≥4× maternal titer, dark-field microscopy, IgM FTA-ABS, treponema PCR |
| Treatment | Aqueous crystalline penicillin G 50,000 units/kg IV q12h (DOL 0–7) or q8h (DOL 8–28) × 10 days; OR procaine penicillin G 50,000 units/kg IM daily × 10 days |
Zika Virus
Congenital Zika syndrome: severe microcephaly with partially collapsed skull, subcortical calcifications, macular scarring, congenital contractures (arthrogryposis), and marked neurological impairment. Transmitted by Aedes mosquitoes or sexually. Diagnosis: Zika IgM, RT-PCR in maternal/infant serum and urine. No specific treatment; supportive care.
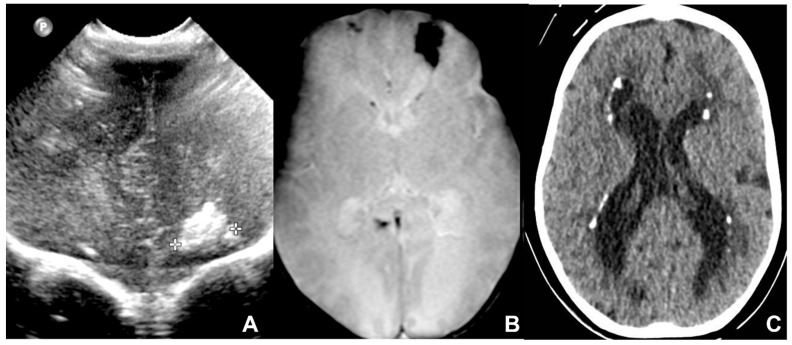
13 Necrotizing Enterocolitis
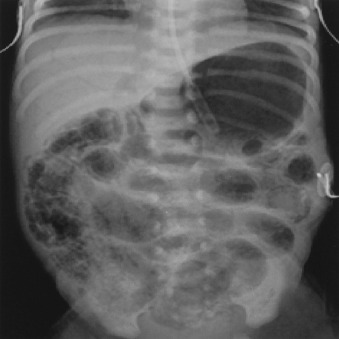
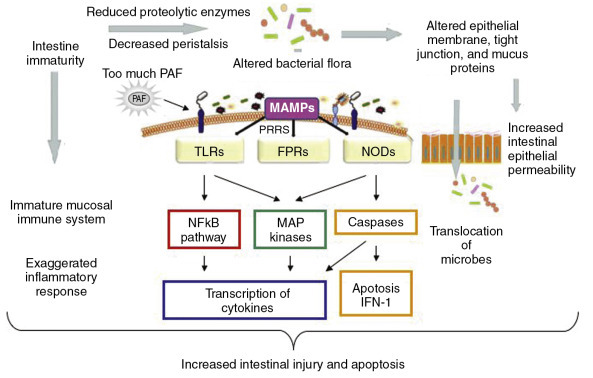
Necrotizing enterocolitis (NEC) is the most common life-threatening gastrointestinal emergency in neonates, affecting 5–10% of VLBW infants, with mortality of 20–30% (up to 50% in those requiring surgery). It is characterized by intestinal inflammation, mucosal necrosis, and bacterial invasion of the bowel wall.
Pathophysiology & Risk Factors
NEC results from a complex interplay of intestinal immaturity, abnormal bacterial colonization, ischemia-reperfusion injury, and inflammatory cascade activation. The terminal ileum and ascending colon are most commonly affected.
- Prematurity — the single greatest risk factor (90% of NEC occurs in preterm infants)
- Formula feeding — breast milk is strongly protective (up to 6–10× lower risk)
- Abnormal intestinal flora / dysbiosis
- Intestinal ischemia (PDA with diastolic steal, CHD, polycythemia)
- Rapid advancement of enteral feeds
- Prolonged empiric antibiotics (alters microbiome)
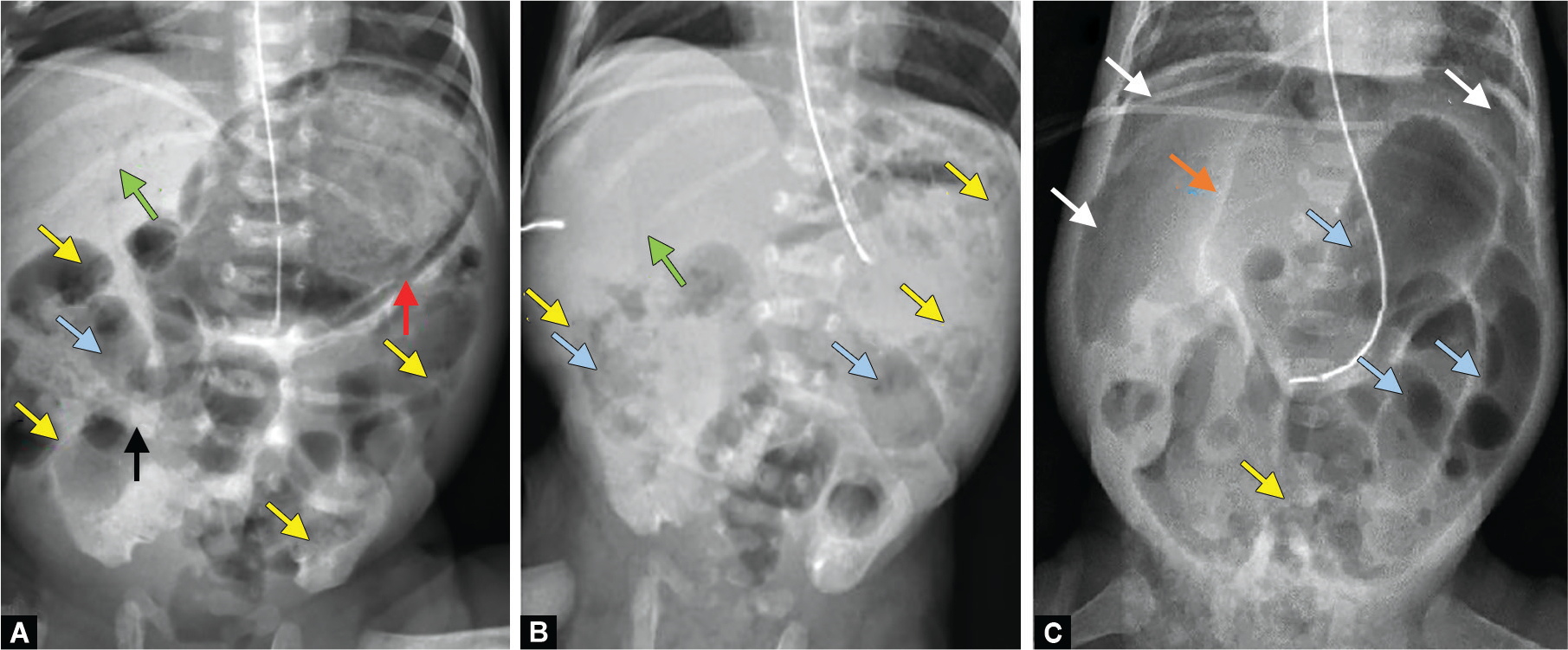
Modified Bell Staging Criteria
| Stage | Clinical Signs | Radiographic Findings | Treatment |
|---|---|---|---|
| IA — Suspected | Temperature instability, apnea, bradycardia, feeding intolerance, mild abdominal distension, occult blood in stool | Normal or mild ileus | NPO, gastric decompression, IV antibiotics × 3 days, serial exams and radiographs |
| IB — Suspected | Same as IA plus gross bloody stool | Normal or mild ileus | Same as IA |
| IIA — Definite, Mild | All of Stage I plus absent bowel sounds, abdominal tenderness | Pneumatosis intestinalis (pathognomonic — intramural gas), mild ileus | NPO, IV antibiotics (ampicillin + gentamicin + metronidazole or clindamycin) × 7–14 days, serial labs/imaging |
| IIB — Definite, Moderate | All of IIA plus metabolic acidosis, thrombocytopenia, abdominal wall erythema or mass | Extensive pneumatosis, portal venous gas, ascites | Same as IIA plus fluid resuscitation, inotropes if needed, surgical consultation |
| IIIA — Advanced, Intact Bowel | Generalized peritonitis, shock, DIC, severe acidosis, neutropenia | Prominent ascites, fixed or dilated loops, may have pneumatosis | Aggressive resuscitation, broad-spectrum antibiotics, surgical consultation, paracentesis if tense ascites |
| IIIB — Advanced, Perforation | Same as IIIA plus clinical deterioration despite maximal medical therapy | Pneumoperitoneum (free air) | Surgical intervention: primary peritoneal drain (ELBW) or laparotomy with resection and ostomy; fluid/blood product resuscitation |
Surgical Indications
- Absolute: Pneumoperitoneum (intestinal perforation)
- Relative: Fixed dilated loop on serial radiographs, portal venous gas, clinical deterioration despite maximal medical therapy, abdominal wall erythema/induration, positive paracentesis (bile-stained or bacteria on Gram stain)
Prevention
Human breast milk is the most effective preventive measure. The AAP recommends exclusive breast milk for all preterm infants; donor human milk if mother’s own milk is unavailable. Standardized feeding protocols (slow, consistent advancement at 20–30 mL/kg/day), avoidance of prolonged empiric antibiotics, and probiotics (evidence supports Lactobacillus and Bifidobacterium species, though specific products and dosing remain debated) all reduce NEC incidence.
14 Neonatal Hyperbilirubinemia
Jaundice is visible in ~60% of term and ~80% of preterm newborns in the first week of life. Most jaundice is benign (physiologic), but unchecked unconjugated hyperbilirubinemia can cause bilirubin encephalopathy and kernicterus, a devastating and preventable condition.
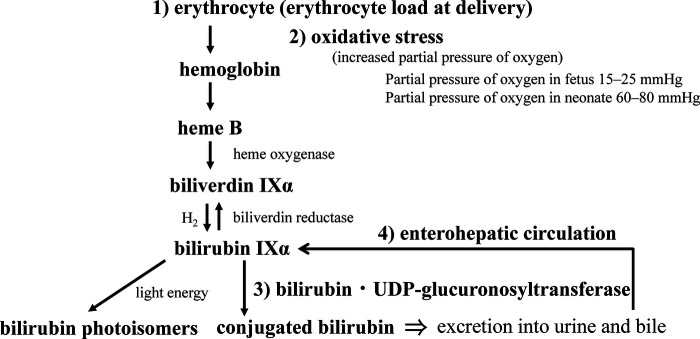
Bilirubin Metabolism in the Neonate
Neonates produce bilirubin at twice the adult rate (higher RBC mass, shorter RBC lifespan of 60–90 days vs. 120 days) and have immature hepatic conjugation (low UDP-glucuronosyltransferase/UGT1A1 activity). Unconjugated (indirect) bilirubin is lipid-soluble and crosses the blood-brain barrier; conjugated (direct) bilirubin is water-soluble and does not cause neurotoxicity.
Physiologic vs. Pathologic Jaundice
| Feature | Physiologic | Pathologic |
|---|---|---|
| Onset | ≥24 hours of life | <24 hours (always pathologic) |
| Peak | DOL 3–5 (term); DOL 5–7 (preterm) | Rapid rise >5 mg/dL/day |
| Level | <12 mg/dL (term) | >95th percentile on Bhutani nomogram |
| Duration | Resolves by DOL 10–14 | Persists >2 weeks (term) or >3 weeks (preterm) |
| Direct bilirubin | Normal | Direct >1.0 mg/dL or >20% of total → cholestatic jaundice (biliary atresia, hepatitis, metabolic disease) |
Bhutani Nomogram
The Bhutani hour-specific nomogram plots total serum bilirubin (TSB) against the infant’s age in hours and classifies risk as: low risk (<40th percentile), low-intermediate (40th–75th), high-intermediate (75th–95th), and high risk (>95th percentile). Risk zone at discharge guides follow-up timing and need for repeat TSB.
Phototherapy Thresholds (AAP 2022 Guidelines)
| Gestational Age | Age 24 h | Age 48 h | Age 72 h | Age 96 h+ |
|---|---|---|---|---|
| ≥38 wk, no risk factors | 12.0 | 15.0 | 18.0 | 20.0 |
| ≥38 wk, with risk factors* | 10.5 | 13.0 | 15.5 | 17.5 |
| 35–37 6/7 wk, no risk factors | 10.0 | 12.5 | 15.0 | 17.0 |
| 35–37 6/7 wk, with risk factors* | 8.0 | 10.0 | 12.0 | 14.0 |
*Risk factors (neurotoxicity risk): isoimmune hemolytic disease, G6PD deficiency, asphyxia, sepsis, acidosis, albumin <3.0 g/dL. Values in mg/dL. Thresholds are approximate; always refer to gestational age and hour-specific AAP charts.
Exchange Transfusion
Indicated when TSB rises to exchange transfusion threshold (generally 2–5 mg/dL above phototherapy threshold depending on age and risk factors) or if signs of acute bilirubin encephalopathy develop. Double-volume exchange (160–180 mL/kg) removes ~85% of circulating bilirubin and antibody-coated RBCs. Risks: electrolyte disturbances, thrombocytopenia, infection, NEC, portal vein thrombosis.
Bilirubin Encephalopathy & Kernicterus
- Acute bilirubin encephalopathy: Early — lethargy, hypotonia, poor feeding; Intermediate — hypertonia (opisthotonos, retrocollis), high-pitched cry, fever; Advanced — pronounced opisthotonos, apnea, seizures, coma
- Chronic bilirubin encephalopathy (kernicterus): Tetrad of choreoathetoid cerebral palsy, upward gaze palsy, sensorineural hearing loss, and dental enamel hypoplasia
Breastfeeding vs. Breast Milk Jaundice
| Feature | Breastfeeding Jaundice | Breast Milk Jaundice |
|---|---|---|
| Timing | First week (DOL 3–5) | After DOL 5–7; peaks at 2 weeks |
| Cause | Insufficient intake → dehydration → decreased bilirubin clearance | Substances in breast milk (β-glucuronidase, EGF) that increase enterohepatic circulation |
| Management | Increase breastfeeding frequency (8–12 times/day); supplement if needed; lactation support | Continue breastfeeding; may persist 4–12 weeks; benign; temporary cessation (24–48 h) confirms diagnosis but is rarely necessary |
15 Neonatal Hypoglycemia
Neonatal hypoglycemia is one of the most common metabolic problems in the nursery. While a precise definition remains debated, the AAP Committee on Fetus and Newborn uses an operational threshold of <45 mg/dL (2.5 mmol/L) as the level below which intervention is recommended in symptomatic and at-risk neonates.
At-Risk Populations
| Category | Mechanism | Examples |
|---|---|---|
| Decreased glycogen stores | Insufficient hepatic glycogen deposition | SGA, IUGR, preterm, post-term |
| Increased glucose utilization | Higher metabolic demand | Perinatal stress/asphyxia, sepsis, hypothermia, respiratory distress, polycythemia |
| Hyperinsulinism | Excessive insulin suppresses glycogenolysis and gluconeogenesis | IDM (infant of diabetic mother), LGA, Beckwith-Wiedemann syndrome, congenital hyperinsulinism, perinatal stress-induced hyperinsulinism |
Clinical Features
Symptoms are non-specific: jitteriness, tremors, poor feeding, lethargy, hypotonia, apnea, seizures, cyanosis, hypothermia. Many hypoglycemic neonates are asymptomatic, underscoring the importance of screening at-risk infants.
Management Algorithm
Step 1 — Asymptomatic, glucose 25–45 mg/dL: Feed (breast or formula); recheck in 30–60 minutes. Consider oral glucose gel 40% (0.5 mL/kg = 200 mg/kg massaged into buccal mucosa) followed by feeding.
Step 2 — Glucose <25 mg/dL or symptomatic at any level: IV D10W bolus 2 mL/kg (200 mg/kg) over 5–10 minutes, followed by continuous dextrose infusion.
Step 3 — Continuous infusion: Start GIR 4–6 mg/kg/min (D10W at 60–80 mL/kg/day provides ~4–6 mg/kg/min). Titrate up to 8–12 mg/kg/min as needed. If GIR >12 mg/kg/min required → evaluate for hyperinsulinism.
GIR formula: GIR (mg/kg/min) = [% dextrose × rate (mL/hr)] / [6 × weight (kg)]
Persistent Hypoglycemia Workup
If hypoglycemia persists beyond 48–72 hours or requires GIR >12 mg/kg/min, obtain a critical sample during hypoglycemia (glucose <50 mg/dL):
- Insulin — elevated insulin with low glucose = hyperinsulinism (insulin:glucose ratio >0.3)
- Cortisol — low cortisol = adrenal insufficiency
- Growth hormone — low GH = GH deficiency
- Free fatty acids & beta-hydroxybutyrate — suppressed in hyperinsulinism (insulin inhibits lipolysis and ketogenesis)
- Lactate, ammonia, acylcarnitine profile, urine organic acids — fatty acid oxidation defects, organic acidemias, glycogen storage diseases
16 Fluid, Electrolytes & Nutrition
Daily Fluid Requirements
Neonatal fluid management is guided by gestational age, postnatal age, body weight changes, urine output, and serum sodium. Insensible water loss (IWL) is highest in the most premature infants due to thin, permeable skin and high surface area-to-volume ratio.
| Day of Life | Term (mL/kg/day) | Preterm (mL/kg/day) |
|---|---|---|
| DOL 1 | 60–80 | 80–100 |
| DOL 2 | 80–100 | 100–120 |
| DOL 3 | 100–120 | 120–140 |
| DOL 4–7 | 120–150 | 140–160 |
| DOL >7 | 140–160 | 150–180 |
Expect a physiologic weight loss of 5–10% in term and 10–15% in ELBW infants over the first week, primarily from extracellular fluid contraction.
Electrolyte Requirements
| Electrolyte | Requirement | Timing |
|---|---|---|
| Sodium | 2–4 mEq/kg/day | After post-natal diuresis (DOL 2–3); do not add to Day 1 fluids |
| Potassium | 1–3 mEq/kg/day | After urine output established; monitor closely in ELBW (non-oliguric hyperkalemia common) |
| Calcium | 40–80 mg/kg/day elemental | Begin DOL 1 in TPN; preterm infants at high risk for hypocalcemia |
| Phosphorus | 30–45 mg/kg/day | Begin with TPN; essential for bone mineralization |
Total Parenteral Nutrition (TPN)
Initiate TPN within the first hours of life for preterm infants unable to tolerate full enteral feeds:
- Dextrose: Start D10W (GIR ~4–6 mg/kg/min); advance to D12.5–D15 as needed (max GIR ~11–12 mg/kg/min via peripheral IV)
- Amino acids: Start 2–3 g/kg/day on DOL 1; advance to 3.5–4 g/kg/day by DOL 3–5
- Lipids (intralipid 20%): Start 1–2 g/kg/day on DOL 1; advance to 3 g/kg/day; monitor triglycerides (keep <200–250 mg/dL)
- Vitamins & trace elements: Add MVI-Pediatric and trace element solution
Enteral Nutrition
- Trophic feeds: 10–20 mL/kg/day of breast milk starting DOL 1–2 (primes the gut, promotes motility, does not increase NEC risk)
- Advancement: Increase by 20–30 mL/kg/day in stable preterm infants using standardized feeding protocols
- Full feeds: 140–160 mL/kg/day; target caloric intake 110–130 kcal/kg/day
- Human milk fortification: Add HMF (human milk fortifier) once feeds reach 80–100 mL/kg/day to provide additional protein (to 3.5–4 g/kg/day), calcium, phosphorus, and calories for preterm infants
- Growth target: 15–20 g/kg/day (approximates intrauterine growth rate)
17 Neonatal Hematology
Anemia of Prematurity
Anemia of prematurity (AOP) is a normocytic, normochromic anemia that develops in preterm infants over the first weeks of life, caused by low erythropoietin (EPO) production, shortened RBC lifespan (40–60 days), rapid body growth, and iatrogenic blood loss from laboratory draws. It is the most common anemia in preterm infants.
| Transfusion Thresholds (ELBW/VLBW) | Hematocrit | Hemoglobin |
|---|---|---|
| Ventilated / critically ill | <35% | <12 g/dL |
| CPAP / supplemental O2 | <30% | <10 g/dL |
| Stable, growing | <20–25% | <7–8 g/dL |
Transfuse with pRBC 10–20 mL/kg over 3–4 hours; use CMV-negative, irradiated, leukoreduced units. EPO (erythropoietin) with iron supplementation can reduce transfusion needs in stable preterm infants, but routine use remains controversial; iron supplementation (2–4 mg/kg/day) is universally recommended for preterm infants once on full enteral feeds.
Polycythemia
Defined as venous hematocrit >65%. Causes: delayed cord clamping, twin-to-twin transfusion, IDM, SGA, maternal-fetal transfusion, chromosomal abnormalities. Most are asymptomatic; symptomatic infants (lethargy, poor feeding, hypoglycemia, respiratory distress, cyanosis, seizures) may require partial exchange transfusion with normal saline to reduce Hct to ~55%. Asymptomatic polycythemia is managed with hydration and monitoring.
Neonatal Thrombocytopenia
| Timing | Common Causes |
|---|---|
| Early (<72 h) | Placental insufficiency (preeclampsia, IUGR), congenital infection (TORCH), chromosomal abnormalities, NAIT |
| Late (>72 h) | Sepsis, NEC, DIC, medication-related, fungal infection |
Neonatal alloimmune thrombocytopenia (NAIT) is the platelet equivalent of hemolytic disease of the newborn. Maternal antibodies (most commonly anti-HPA-1a) cross the placenta and destroy fetal platelets. Unlike Rh disease, NAIT can occur in the first pregnancy. Treatment: transfusion with HPA-1a-negative platelets (or washed maternal platelets); IVIG; monitor for intracranial hemorrhage.
Hemolytic Disease of the Newborn
| Type | Rh Hemolytic Disease | ABO Incompatibility |
|---|---|---|
| Mechanism | Rh-negative mother + Rh-positive fetus; maternal anti-D IgG crosses placenta | Type O mother + Type A or B fetus; maternal anti-A/B IgG crosses placenta |
| Severity | Can be severe: hydrops fetalis, severe anemia, heart failure | Usually mild; rarely requires exchange transfusion |
| First pregnancy | Rarely affected (sensitization during delivery); subsequent pregnancies worsen | Can occur in first pregnancy (naturally occurring anti-A/B IgG) |
| DAT (Coombs) | Strongly positive | Weakly positive or negative |
| Smear | Nucleated RBCs, reticulocytosis | Spherocytes, reticulocytosis |
| Management | Phototherapy, IVIG (0.5–1 g/kg) if TSB rising despite intensive phototherapy, exchange transfusion for severe cases | Phototherapy usually sufficient |
| Prevention | Anti-D immunoglobulin (RhoGAM) at 28 weeks and within 72 hours of delivery to Rh-negative mothers | Not applicable |
18 Congenital Heart Disease
Congenital heart disease (CHD) affects approximately 8–10 per 1,000 live births and is the leading cause of death from congenital anomalies. Classification into cyanotic vs. acyanotic lesions guides the initial clinical approach.
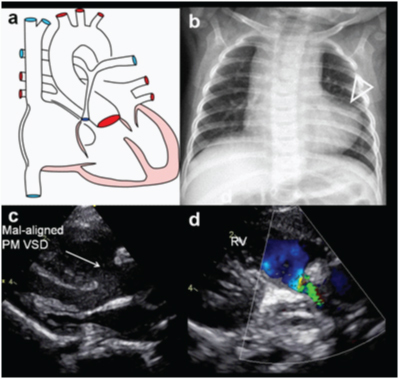
Cyanotic CHD — The 5 T’s
| Lesion | Key Features |
|---|---|
| Tetralogy of Fallot | Most common cyanotic CHD; 4 features: VSD, overriding aorta, RV outflow tract obstruction (RVOTO), RV hypertrophy; “tet spells” (hypercyanotic episodes) treated with knee-chest position, O2, morphine, phenylephrine; boot-shaped heart on CXR |
| Transposition of the Great Arteries (TGA) | Most common cyanotic CHD presenting in the first 24 hours; aorta arises from RV, PA from LV; parallel circuits → lethal without mixing (ASD, VSD, PDA); “egg on a string” CXR; Rx: PGE1 to maintain PDA, Rashkind balloon atrial septostomy, arterial switch operation (Jatene) |
| Tricuspid Atresia | Absent tricuspid valve; obligatory R-to-L shunt at atrial level (ASD); depends on VSD and PDA for pulmonary blood flow; hypoplastic RV |
| Total Anomalous Pulmonary Venous Return (TAPVR) | All 4 pulmonary veins drain to systemic venous system instead of LA; types: supracardiac (most common), cardiac, infracardiac (worst prognosis — obstruction), mixed; “snowman” sign on CXR (supracardiac type); surgical emergency if obstructed |
| Truncus Arteriosus | Single arterial trunk arising from both ventricles, overriding a large VSD; common trunk gives rise to aorta, pulmonary arteries, and coronary arteries; presents with CHF and mild cyanosis in first weeks; associated with DiGeorge syndrome (22q11.2 deletion) |
Acyanotic CHD
| Lesion | Key Features |
|---|---|
| Ventricular Septal Defect (VSD) | Most common CHD overall; holosystolic murmur at LLSB; small — loud murmur, large — soft murmur + CHF; many close spontaneously; surgical repair if large/symptomatic |
| Atrial Septal Defect (ASD) | L-to-R shunt at atrial level; fixed split S2; systolic ejection murmur at LUSB (from increased flow across pulmonic valve); often asymptomatic in infancy; secundum type most common |
| Patent Ductus Arteriosus (PDA) | Failure of ductus arteriosus to close; continuous “machinery” murmur; bounding pulses, wide pulse pressure; common in preterm infants (see section 19) |
| Coarctation of the Aorta | Narrowing of the aorta, typically at the isthmus (juxtaductal); upper extremity hypertension, weak femoral pulses, BP gradient >20 mmHg between upper and lower extremities; associated with Turner syndrome and bicuspid aortic valve; ductal-dependent in neonatal presentation |
Hyperoxia Test
Used to differentiate cyanotic CHD from pulmonary disease: administer 100% FiO2 for 10 minutes. In pulmonary disease, PaO2 typically rises to >150 mmHg. In cyanotic CHD with fixed right-to-left shunt, PaO2 remains <100 mmHg (often <70 mmHg) and SpO2 does not significantly improve. This test is not definitive and echocardiography is required for diagnosis.
Prostaglandin E1 (PGE1) for Ductal-Dependent Lesions
Any neonate with suspected ductal-dependent CHD should receive PGE1 to maintain ductal patency:
- Dose: 0.05–0.1 mcg/kg/min IV continuous infusion; may reduce to 0.01–0.025 mcg/kg/min once ductus is open
- Side effects: Apnea (10–12% — have intubation equipment ready), hypotension, fever, flushing, diarrhea
- Ductal-dependent for pulmonary blood flow: Pulmonary atresia, critical PS, tricuspid atresia, severe TOF
- Ductal-dependent for systemic blood flow: Critical coarctation, interrupted aortic arch, hypoplastic left heart syndrome
- Ductal-dependent for mixing: TGA
Critical CHD Screening
Pulse oximetry screening is performed on all newborns after 24 hours of life (or before discharge if earlier). Measure SpO2 on the right hand (preductal) and either foot (postductal). A positive screen requires echocardiography: SpO2 <90% in any extremity (immediate fail), SpO2 90–94% in both extremities or >3% difference between right hand and foot on 3 measures separated by 1 hour.
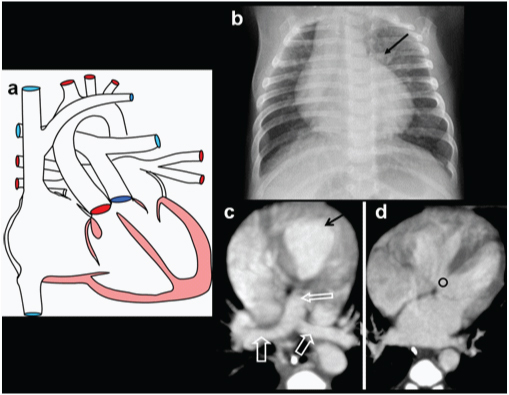
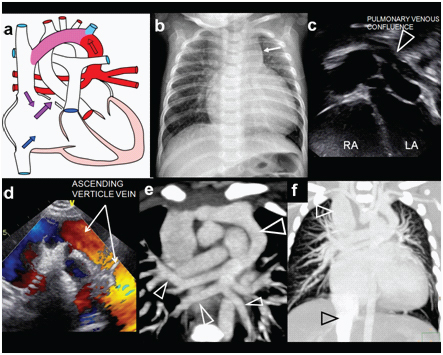
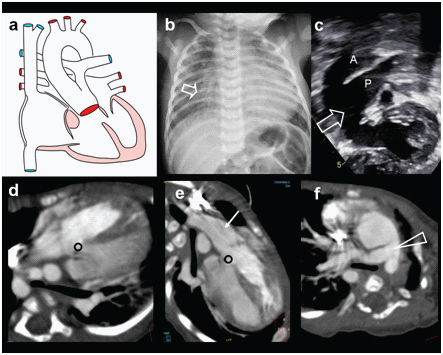
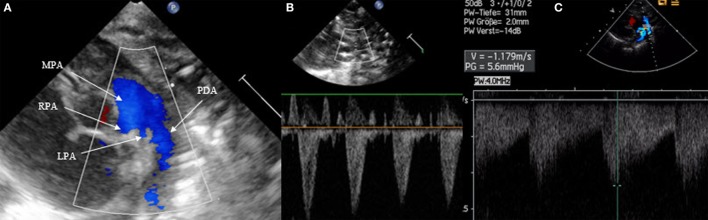
19 Patent Ductus Arteriosus
The patent ductus arteriosus (PDA) occurs in up to 70% of infants born <28 weeks gestation and is a major contributor to morbidity in preterm infants. As PVR falls postnatally, the shunt direction becomes left-to-right, causing pulmonary overcirculation and systemic hypoperfusion.
Clinical Signs of Hemodynamic Significance
- Continuous “machinery” murmur best heard at left upper sternal border (may be only systolic in preterm infants)
- Bounding peripheral pulses and wide pulse pressure (low diastolic BP)
- Hyperdynamic precordium
- Worsening respiratory status (increased ventilator requirements, inability to wean)
- Hepatomegaly, feeding intolerance
- Diastolic flow reversal in descending aorta on echo (sign of steal)
Echocardiographic Criteria
Hemodynamically significant PDA (hsPDA): duct diameter >1.5 mm (or >1.5 mm/kg), LA:Ao ratio >1.5, left ventricular dilation, diastolic flow reversal in the descending aorta, absent or reversed diastolic flow in the anterior cerebral or renal arteries.
Management
| Approach | Details |
|---|---|
| Conservative | Fluid restriction, diuretics, respiratory support optimization; many PDAs close spontaneously, especially in larger preterm and term infants; watchful waiting is increasingly favored |
| Ibuprofen | 10 mg/kg IV/PO × 1, then 5 mg/kg × 2 doses at 24 h intervals; success rate ~70–80%; fewer renal side effects than indomethacin; preferred first-line pharmacologic agent in many centers |
| Indomethacin | 0.1–0.25 mg/kg IV q12–24h × 3 doses; also effective; side effects include decreased renal blood flow, NEC risk, decreased platelet function |
| Acetaminophen | 15 mg/kg PO/IV q6h × 3–7 days; emerging alternative; inhibits prostaglandin synthesis at the peroxidase segment; fewer renal and GI side effects; growing evidence but not yet universally accepted as first-line |
| Surgical ligation | Via left thoracotomy; reserved for failed medical therapy or contraindications to pharmacologic closure; risks include LRLN injury, chylothorax, pneumothorax, post-ligation cardiac syndrome |
| Catheter-based closure | Increasingly performed even in small preterm infants (>700 g at some centers); occlusion devices or coils; avoids thoracotomy |
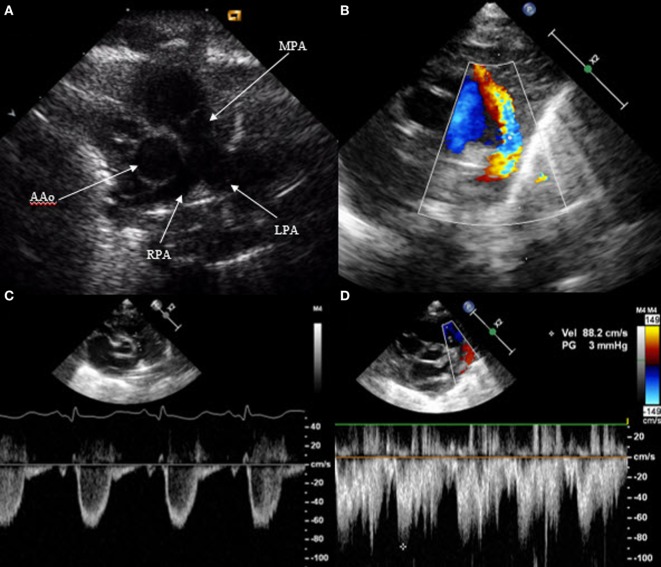
20 Neonatal Neurology
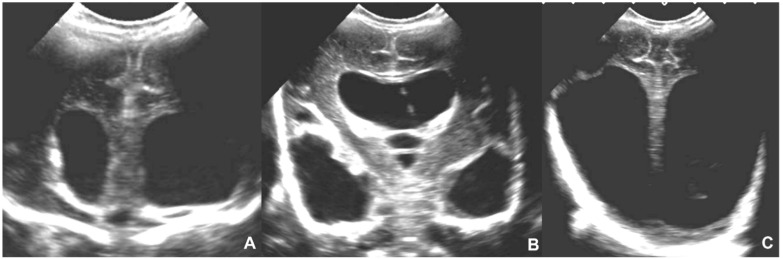
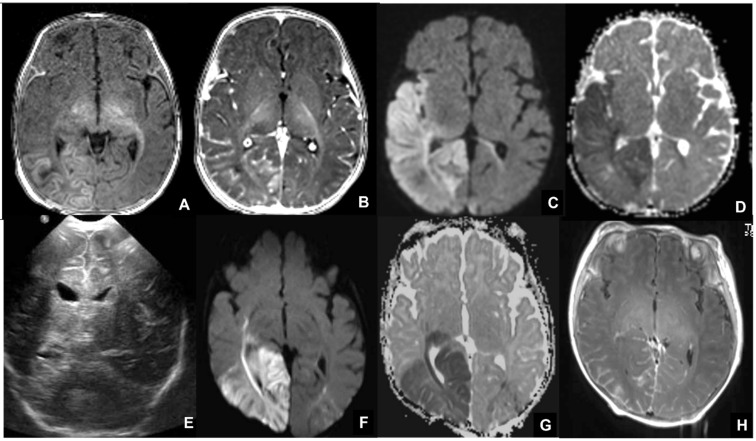
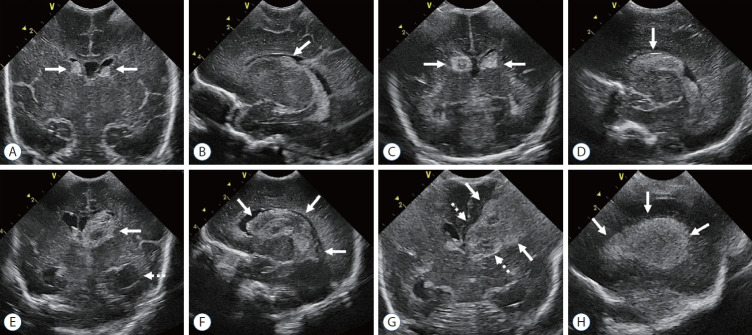
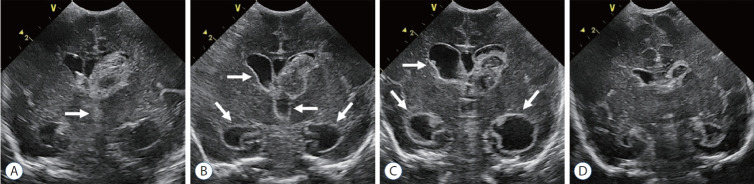
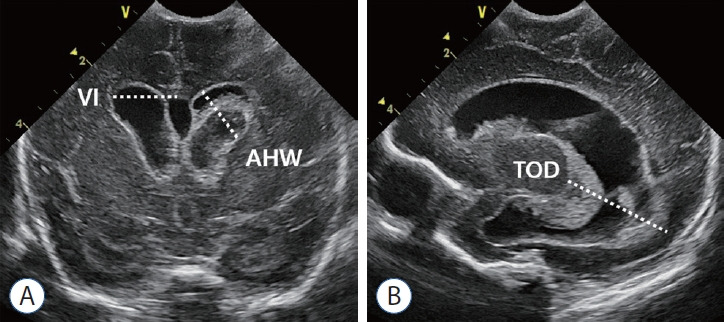
Intraventricular Hemorrhage (IVH)
IVH occurs in ~25–30% of VLBW infants, originating from the fragile germinal matrix vasculature. The germinal matrix is a highly vascularized region in the subependymal zone that involutes by 34–36 weeks; its vessels lack smooth muscle support and are vulnerable to fluctuations in cerebral blood flow.
Papile Grading System
| Grade | Description | Prognosis |
|---|---|---|
| Grade I | Germinal matrix hemorrhage only (subependymal) | Excellent; resolves spontaneously |
| Grade II | IVH without ventricular dilation (<50% of ventricle filled with blood) | Good; 5–10% risk of hydrocephalus |
| Grade III | IVH with ventricular dilation (>50% filled) | Moderate; 25–50% major disability; 20–30% post-hemorrhagic hydrocephalus |
| Grade IV | Periventricular hemorrhagic infarction (PVHI) — venous infarction of periventricular white matter, typically unilateral | Poor; >50% major disability; contralateral hemiplegia common |
Screening Protocol
Cranial ultrasound screening for all infants <32 weeks: DOL 3–7 (detect early IVH), DOL 10–14 (detect late IVH and early PVL), 36 weeks PMA or near discharge (detect PVL, post-hemorrhagic ventricular dilation). Repeat sooner if clinical deterioration (unexplained drop in Hct, bulging fontanelle, apnea/bradycardia).
Periventricular Leukomalacia (PVL)
PVL is ischemic injury to periventricular white matter, the most common brain injury associated with cerebral palsy in preterm infants. Cystic PVL (visible cysts on ultrasound) carries the worst prognosis but has decreased in incidence; non-cystic (diffuse) PVL is more common and may be detected only on MRI. Risk factors: chorioamnionitis, postnatal sepsis, hypotension, hypocapnia.
Neonatal Seizures
| Type | Description | Correlation with EEG |
|---|---|---|
| Subtle | Most common; tonic eye deviation, oral-buccal-lingual movements (sucking, lip smacking), pedaling, boxing, apnea | Often not electrographic (may be brainstem release phenomena) |
| Clonic | Rhythmic jerking; focal (one limb) or multifocal (migrating); cannot be stopped by restraint | Usually electrographic; most reliably epileptic |
| Tonic | Sustained posturing; focal (asymmetric) or generalized (extension or flexion) | Focal tonic — often electrographic; generalized tonic — often not |
| Myoclonic | Rapid, single or repetitive jerks; focal, multifocal, or generalized | Generalized myoclonic — often electrographic; may indicate severe brain injury |
Treatment: Correct underlying cause (hypoglycemia, hypocalcemia, meningitis). Phenobarbital is first-line: loading dose 20 mg/kg IV (may give additional 10 mg/kg boluses × 2 if seizures persist, up to 40 mg/kg total). Second-line: levetiracetam (40–60 mg/kg IV), fosphenytoin (20 mg/kg PE IV), or midazolam infusion. Monitor with aEEG or continuous EEG — >50% of neonatal seizures are electrographic only (no clinical correlate).
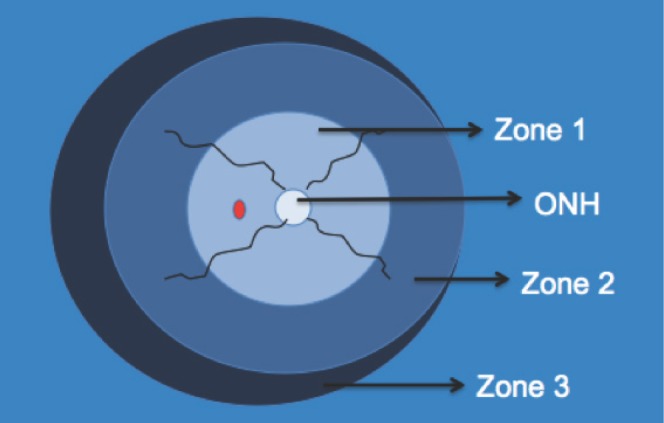
Retinopathy of Prematurity (ROP)
ROP is a vasoproliferative disorder of the immature retina. The retina is vascularized centrifugally from the optic disc, completing nasally by ~36 weeks and temporally by ~40 weeks. Premature birth interrupts vascularization; subsequent hyperoxia suppresses VEGF → vascular growth arrest → then, as retinal metabolic demand increases, relative hypoxia triggers VEGF upregulation → abnormal neovascularization.
| Stage | Description |
|---|---|
| Stage 1 | Demarcation line between vascularized and avascular retina |
| Stage 2 | Ridge (demarcation line with height and width) |
| Stage 3 | Ridge with extraretinal neovascularization (fibrovascular proliferation) |
| Stage 4 | Partial retinal detachment (4A: fovea attached; 4B: fovea detached) |
| Stage 5 | Total retinal detachment (funnel-shaped) |
Plus disease: Dilated, tortuous posterior pole vessels; indicates active, aggressive disease and lowers the threshold for treatment. Aggressive posterior ROP (AP-ROP): Rapidly progressive disease in zone I or posterior zone II with prominent plus disease; requires urgent treatment.
Screening Criteria
Screen all infants <30 weeks GA or <1,500 g (and selected infants 30–36 weeks with unstable course). Initial exam at 31 weeks PMA or 4 weeks postnatal age, whichever is later. Follow-up per AAP/AAO guidelines based on zone and stage.
Treatment
- Laser photocoagulation: Ablates avascular retina to reduce VEGF drive; standard of care for type 1 ROP (zone I any stage with plus, zone I stage 3, zone II stage 2 or 3 with plus)
- Anti-VEGF therapy (bevacizumab/ranibizumab): Intravitreal injection; increasingly used for zone I disease and AP-ROP; allows continued peripheral vascularization (advantage over laser); requires long-term follow-up (late recurrence possible)
21 Prematurity & NICU Organization
Classification of Preterm Birth
| Category | Gestational Age | Approximate Survival |
|---|---|---|
| Extreme preterm | <28 weeks | 22 wk: 30–40%; 24 wk: 60–70%; 26 wk: 80–85% |
| Very preterm | 28–31 6/7 weeks | >90–95% |
| Moderate preterm | 32–33 6/7 weeks | >98% |
| Late preterm | 34–36 6/7 weeks | >99% |
Periviability & Limits of Viability
The “gray zone” of viability is generally 22 0/7 to 25 6/7 weeks. Management should be guided by individualized assessment (GA, estimated fetal weight, sex, singleton/multiple, antenatal steroids exposure) and family counseling using tools such as the NICHD Neonatal Research Network’s Extremely Preterm Birth Outcome Data tool. At 22 weeks, active resuscitation is offered based on institutional guidelines and family preferences; at 25 weeks, active resuscitation is standard of care at most centers.
Antenatal Interventions
- Antenatal corticosteroids: Betamethasone 12 mg IM × 2 doses 24 hours apart (or dexamethasone 6 mg IM × 4 doses 12 hours apart); for threatened preterm delivery at 24 0/7 to 33 6/7 weeks (may consider at 23 weeks); reduces RDS, IVH, NEC, and mortality; optimal benefit 48 hours to 7 days after administration; a single “rescue” course may be given if ≥14 days since prior course and delivery remains imminent
- Magnesium sulfate for neuroprotection: 4–6 g IV loading over 20–30 min, then 1–2 g/hr until delivery or for 24 hours; for impending delivery at <32 weeks; reduces risk of cerebral palsy by ~30–40% (NNT ~63)
- GBS prophylaxis: As detailed in section 11
- Tocolysis: Short-term to allow steroid administration; agents include nifedipine, indomethacin (<32 weeks), atosiban (outside US)
NICU Levels of Care
| Level | Capabilities |
|---|---|
| Level I (Well Newborn Nursery) | Stabilize and provide care for healthy term newborns (35–37+ weeks); stabilize ill newborns until transfer |
| Level II (Special Care Nursery) | Care for infants ≥32 weeks and ≥1,500 g; provide CPAP and short-term ventilation (<24 h); IV fluids and tube feedings |
| Level III (NICU) | Sustained life support including prolonged mechanical ventilation; advanced imaging; subspecialty consultants; care for infants at any GA |
| Level IV (Regional NICU) | All Level III capabilities PLUS on-site pediatric surgical subspecialties (cardiac surgery, neurosurgery); ECMO; serves as regional referral center |
22 Congenital Anomalies
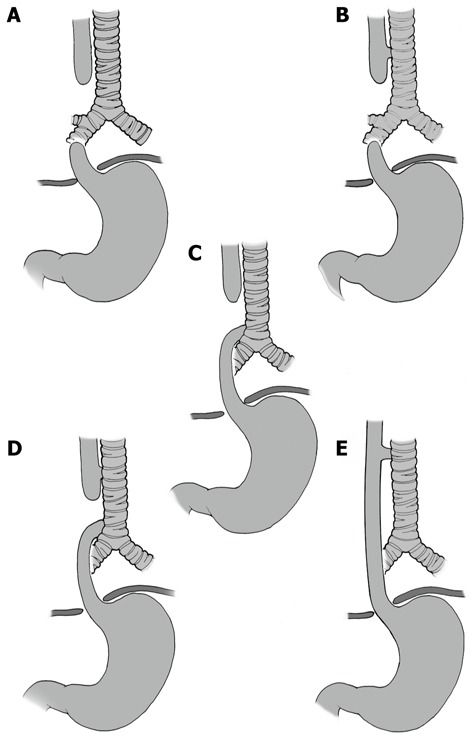
Tracheoesophageal Fistula / Esophageal Atresia
Incidence ~1:3,500. The most common type (85%, Type C) is esophageal atresia with a distal tracheoesophageal fistula (TEF). Presents with drooling, choking, cyanosis with first feed, inability to pass an orogastric tube (coils in proximal esophageal pouch on CXR). Gas in the stomach confirms a distal fistula; gasless abdomen suggests pure atresia (Type A).
| Type | Description | Frequency |
|---|---|---|
| A | Esophageal atresia without fistula (pure atresia) | 8% |
| B | Esophageal atresia with proximal TEF | 1% |
| C | Esophageal atresia with distal TEF | 85% |
| D | Esophageal atresia with proximal and distal TEF | 2% |
| E (H-type) | TEF without esophageal atresia; recurrent pneumonia, choking with feeds; delayed diagnosis | 4% |
Associated anomalies: VACTERL association (Vertebral, Anorectal, Cardiac, TE fistula, Renal, Limb). Management: suctioning of proximal pouch, upright positioning, IV fluids, antibiotics if pneumonia, surgical repair (primary anastomosis when possible).
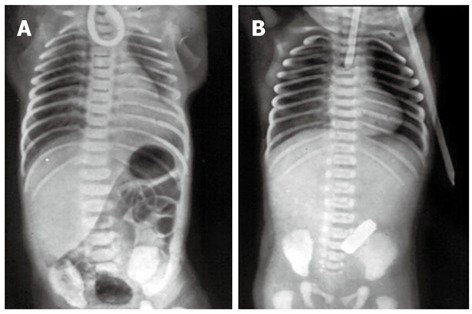
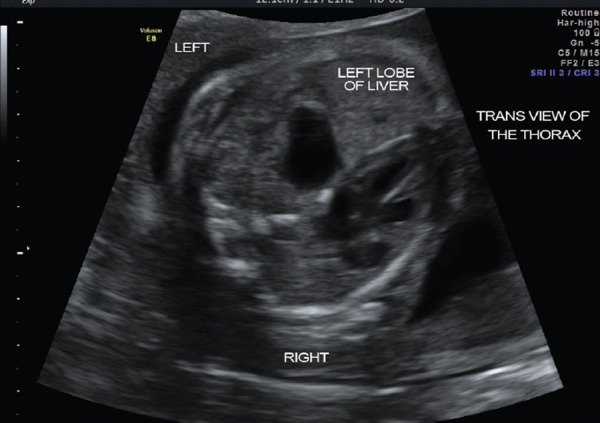
Congenital Diaphragmatic Hernia (CDH)
Incidence ~1:2,500. Left-sided in 85% (Bochdalek hernia through the posterolateral defect). Abdominal viscera herniate into the thorax → pulmonary hypoplasia + pulmonary hypertension. Presents with scaphoid abdomen, absent breath sounds on affected side, mediastinal shift to contralateral side, respiratory distress at birth.
- Stabilization: Immediate intubation (avoid bag-mask — gastric distension worsens lung compression), OG tube to continuous suction, gentle ventilation with permissive hypercapnia, target preductal SpO2 >85%
- PPHN management: iNO, sildenafil, milrinone as needed
- ECMO criteria: OI >40 or failure to maintain preductal SpO2 >85% despite maximal therapy; typically VA ECMO
- Surgical repair: After hemodynamic stabilization (typically 24–72 hours); primary closure or patch repair
Gastroschisis vs. Omphalocele
| Feature | Gastroschisis | Omphalocele |
|---|---|---|
| Defect location | Right of umbilicus (paraumbilical) | At umbilicus (through umbilical ring) |
| Covering membrane | None — bowel exposed | Peritoneal sac covers contents (unless ruptured) |
| Contents | Small bowel (primarily) | Bowel, liver, spleen |
| Associated anomalies | Uncommon (intestinal atresia in 10–15%) | Common (50–70%): cardiac, chromosomal (T18, T13, Beckwith-Wiedemann) |
| Management | Cover with sterile saline-soaked gauze + plastic wrap; IV fluids (massive fluid losses); primary closure or staged silo reduction | Protect sac; evaluate for associated anomalies; primary closure, staged reduction, or paint-and-wait (for giant omphaloceles) |
Neural Tube Defects
Myelomeningocele (spina bifida cystica): Failure of neural tube closure exposing spinal cord and meninges; most common at lumbosacral level. Motor/sensory deficits below the level of the lesion, neurogenic bladder/bowel, Chiari II malformation (hindbrain herniation → hydrocephalus in 80–90%). Management: Moist sterile dressing over defect, prone positioning, early surgical closure (within 24–48 hours), VP shunt for hydrocephalus, ongoing multidisciplinary follow-up. Prevention: folic acid 400 mcg/day before conception (4 mg/day if prior affected pregnancy).
Chromosomal Anomalies
| Condition | Key Neonatal Features |
|---|---|
| Trisomy 21 (Down) | Hypotonia, flat facies, upslanting palpebral fissures, single palmar crease, AV canal defect (40–50% have CHD), duodenal atresia (“double bubble” sign), Brushfield spots |
| Trisomy 18 (Edwards) | IUGR, clenched fists with overlapping fingers (2nd over 3rd, 5th over 4th), rocker-bottom feet, micrognathia, VSD/PDA; median survival 5–15 days |
| Trisomy 13 (Patau) | Holoprosencephaly, midline defects (cleft lip/palate), microcephaly, polydactyly, cutis aplasia (scalp defect), severe cardiac defects; median survival 7–10 days |
23 Neonatal Abstinence Syndrome
Neonatal abstinence syndrome (NAS), increasingly termed neonatal opioid withdrawal syndrome (NOWS), occurs in 55–94% of neonates exposed to opioids in utero. Incidence has risen dramatically with the opioid epidemic, from ~1.5 per 1,000 hospital births in 2004 to ~7 per 1,000 in 2014.
Clinical Features
Onset is typically 24–72 hours after birth for short-acting opioids (heroin) and 48–96 hours for long-acting opioids (methadone, buprenorphine). Symptoms span multiple systems:
- CNS: High-pitched cry, tremors (disturbed and undisturbed), irritability, seizures (rare), hypertonicity, poor sleep
- GI: Poor feeding, uncoordinated suck, vomiting, diarrhea, excessive weight loss
- Autonomic: Sneezing, yawning, nasal stuffiness, sweating, fever, mottling, tachypnea
Finnegan Neonatal Abstinence Scoring System
The modified Finnegan scoring tool assesses 21 items across CNS, metabolic, respiratory, and GI domains. Scored every 3–4 hours. Scores ≥8 on three consecutive assessments, or a single score ≥12, traditionally prompted pharmacologic treatment.
Eat-Sleep-Console (ESC) Approach
An increasingly adopted, function-based assessment that replaces the Finnegan score with three simple questions:
- Can the infant eat ≥1 oz per feed (or breastfeed well)?
- Can the infant sleep undisturbed for ≥1 hour?
- Can the infant be consoled within 10 minutes?
If yes to all three, no pharmacologic treatment is needed regardless of Finnegan score. ESC emphasizes non-pharmacologic care first and has reduced pharmacologic treatment rates by 50%, shortened hospital stays by 50%, and decreased NICU admissions.
Non-Pharmacologic Management (First-Line)
- Rooming-in with mother (reduces NAS severity and LOS)
- Swaddling, skin-to-skin contact, low-stimulation environment (dim lights, reduced noise)
- Frequent small feedings on demand
- Breastfeeding encouraged if mother is in stable medication-assisted treatment (MAT) program, HIV-negative, and not using illicit substances
Pharmacologic Treatment (When Non-Pharmacologic Fails)
| Medication | Dose | Notes |
|---|---|---|
| Morphine sulfate | 0.04–0.08 mg/kg PO q3–4h; titrate to symptoms; wean by 10–20% every 24–48 hours | Most commonly used first-line agent |
| Methadone | 0.05–0.1 mg/kg PO q6–12h | Longer half-life; may allow less frequent dosing; may prolong treatment duration |
| Buprenorphine | Sublingual; emerging evidence for shorter treatment duration | Not yet widely adopted; promising results in clinical trials |
| Clonidine | 0.5–1 mcg/kg PO q4–6h | Adjunct for opioid NAS; also for non-opioid withdrawal (benzodiazepines, SSRIs) |
| Phenobarbital | Load 20 mg/kg, then 5 mg/kg/day | Adjunct or for polydrug exposure; avoid as monotherapy for opioid NAS |
24 Discharge Planning
Discharge from the NICU represents a critical transition. The preterm infant must meet several physiologic milestones and the family must be prepared for home care, follow-up, and monitoring.
Physiologic Criteria for Discharge
- Thermal stability: Maintain normal temperature in an open crib for ≥24–48 hours
- Feeding competence: Adequate oral feeding to sustain growth (breast or bottle); coordinated suck-swallow-breathe; consistent weight gain (20–30 g/day)
- Cardiorespiratory stability: No significant apnea, bradycardia, or desaturation events for 5–8 days (event-free period varies by institution); off supplemental O2 or stable on home O2
Car Seat Challenge
Recommended for all infants born <37 weeks before discharge. The infant is placed in their own car seat and monitored for 90–120 minutes (or the duration of the planned car ride home, whichever is longer). A fail is defined as: sustained desaturation <90%, bradycardia <80 bpm, or apnea >20 seconds. Options for failure include a car bed or repeat testing after clinical improvement.
Immunizations
- Hepatitis B vaccine: First dose within 24 hours of birth for all medically stable infants ≥2 kg; for infants <2 kg born to HBsAg-negative mothers, delay until 1 month of age or hospital discharge
- All routine childhood immunizations given at chronologic age (not corrected age), using standard doses regardless of weight at time of vaccination
- Palivizumab (Synagis): RSV monoclonal antibody; recommended for infants <29 weeks GA during first RSV season, infants with CLD or hemodynamically significant CHD; 15 mg/kg IM monthly during RSV season (October–March)
Screening Before Discharge
| Screen | Details |
|---|---|
| Newborn metabolic screening | State-mandated panel (typically 30–50+ disorders); initial screen at 24–48 hours of life; repeat at 1–2 weeks; preterm infants may need multiple screens due to immaturity confounders (e.g., low T4 without hypothyroidism) |
| Hearing screen | OAE (otoacoustic emissions) or ABR (auditory brainstem response); all infants before discharge; NICU infants should have ABR (higher sensitivity for auditory neuropathy); refer for diagnostic ABR if failed |
| Critical CHD screen | Pulse oximetry (see section 18) |
| ROP screening | Ensure follow-up arranged if not yet completed (see section 20) |
| Head ultrasound | Near-discharge scan for all infants <32 weeks to assess for PVL and ventriculomegaly |
Follow-Up
- Pediatrician: Within 48–72 hours of discharge; weight check, feeding assessment, bilirubin if needed
- Developmental follow-up: High-risk infant follow-up clinic at 4, 8, 12, 18, 24 months corrected age; Bayley Scales of Infant Development at 18–24 months; early intervention referral as needed
- Ophthalmology: Continued ROP screening until retinal vascularization is complete
- Audiology: Repeat hearing assessment if risk factors (aminoglycosides, hyperbilirubinemia, congenital CMV)
- Home monitoring: Apnea monitors for selected infants (severe BRUE, siblings of SIDS, home O2); pulse oximetry for infants on supplemental O2
25 Scoring Systems
APGAR Score
| Sign | 0 | 1 | 2 |
|---|---|---|---|
| Appearance | Blue/pale | Acrocyanosis | Pink |
| Pulse | Absent | <100 | ≥100 |
| Grimace | No response | Grimace | Cry/cough/sneeze |
| Activity | Limp | Some flexion | Active motion |
| Respiration | Absent | Slow/irregular | Good cry |
New Ballard Score
Total score range: −10 to 50. Score mapped to gestational age: −10 = 20 weeks, 0 = 26 weeks, 10 = 30 weeks, 20 = 34 weeks, 30 = 38 weeks, 40 = 42 weeks, 50 = 44 weeks.
Sarnat Staging (HIE)
| Feature | Stage I | Stage II | Stage III |
|---|---|---|---|
| Consciousness | Hyperalert | Lethargic | Comatose |
| Tone | Normal/increased | Hypotonia | Flaccid |
| Reflexes | Hyperactive | Hyperactive | Absent |
| Seizures | None | Common | Uncommon |
| EEG | Normal | Low voltage/seizures | Burst suppression/flat |
| Outcome | Good | Variable (20–40% disability) | Poor (>50% mortality) |
Modified Bell Staging (NEC)
| Stage | Clinical | Radiograph | Treatment |
|---|---|---|---|
| I (Suspected) | Feeding intolerance, mild distension, guaiac+ stool | Normal or mild ileus | NPO, antibiotics × 3 days |
| IIA (Definite, mild) | Absent bowel sounds, tenderness | Pneumatosis intestinalis | NPO, antibiotics × 7–14 days |
| IIB (Definite, moderate) | Acidosis, thrombocytopenia, wall erythema | Portal venous gas, ascites | Medical + surgical consult |
| IIIA (Advanced, intact) | Shock, DIC, peritonitis | Prominent ascites, fixed loops | Aggressive resuscitation |
| IIIB (Advanced, perforation) | Cardiovascular collapse | Pneumoperitoneum | Emergent surgery |
Papile IVH Grading
| Grade | Description |
|---|---|
| I | Germinal matrix hemorrhage only |
| II | IVH without ventricular dilation |
| III | IVH with ventricular dilation |
| IV | Periventricular hemorrhagic infarction |
Finnegan Neonatal Abstinence Scoring (Abbreviated)
| Domain | Signs Scored |
|---|---|
| CNS | Cry (excessive/high-pitched/continuous), sleep (after feeding), Moro (hyperactive/markedly hyperactive), tremors (disturbed/undisturbed, mild/moderate-severe), muscle tone (increased), excoriation, myoclonic jerks, seizures |
| Metabolic/Vasomotor | Sweating, fever (37.2–38.3°C / >38.4°C), mottling, nasal stuffiness, sneezing (>3–4 times), nasal flaring, respiratory rate (>60 / >60 with retractions) |
| GI | Excessive sucking, poor feeding, regurgitation/projectile vomiting, loose/watery stools |
Score ≥8 on 3 consecutive assessments or single score ≥12 traditionally indicates need for pharmacologic treatment. Total possible score ~45.
ROP Staging
| Stage | Finding |
|---|---|
| 1 | Demarcation line |
| 2 | Ridge |
| 3 | Ridge + extraretinal neovascularization |
| 4A | Partial retinal detachment, fovea attached |
| 4B | Partial retinal detachment, fovea detached |
| 5 | Total retinal detachment |
26 Medications Master Table
| Medication | Category | Neonatal Dose | Key Notes |
|---|---|---|---|
| Poractant alfa (Curosurf) | Surfactant | 200 mg/kg initial; 100 mg/kg repeat × 2 | Natural porcine surfactant; intratracheal; fastest onset |
| Beractant (Survanta) | Surfactant | 100 mg/kg/dose × 4 doses q6h PRN | Natural bovine surfactant; intratracheal |
| Calfactant (Infasurf) | Surfactant | 105 mg/kg/dose; repeat × 3 q12h PRN | Natural calf surfactant |
| Caffeine citrate | Methylxanthine | Load: 20 mg/kg IV/PO; Maint: 5–10 mg/kg/day | Apnea of prematurity; reduces BPD; therapeutic level 5–20 mcg/mL |
| Prostaglandin E1 (alprostadil) | Vasodilator | 0.05–0.1 mcg/kg/min; maintenance 0.01–0.05 | Maintains ductal patency; apnea risk 10–12% |
| Ampicillin | Antibiotic | 25–50 mg/kg/dose IV; meningitis: 75–100 mg/kg/dose | Covers GBS, Listeria, some Gram-negatives; empiric EOS |
| Gentamicin | Aminoglycoside | 4–5 mg/kg IV q24–48h (varies by GA/PMA) | Gram-negative coverage; monitor trough <2 mcg/mL |
| Vancomycin | Glycopeptide | 10–15 mg/kg IV q8–12h (varies by PMA) | MRSA, CoNS; target trough 10–20 mcg/mL; AUC/MIC dosing emerging |
| Cefotaxime | 3rd-gen cephalosporin | 50 mg/kg/dose IV q8–12h | Late-onset sepsis/meningitis; good CNS penetration |
| Acyclovir | Antiviral | 60 mg/kg/day IV divided q8h | HSV; 14 days (SEM) or 21 days (CNS/disseminated) |
| Valganciclovir | Antiviral | 16 mg/kg/dose PO BID × 6 months | Congenital CMV; monitor CBC (neutropenia) |
| Ibuprofen | NSAID | 10 mg/kg, then 5 mg/kg × 2 at 24h intervals | PDA closure; fewer renal effects than indomethacin |
| Indomethacin | NSAID | 0.1–0.25 mg/kg IV q12–24h × 3 | PDA closure; decreases IVH if given prophylactically |
| Acetaminophen (IV/PO) | Analgesic/PDA | 15 mg/kg PO/IV q6h × 3–7 days (PDA) | Alternative PDA closure; fewer renal/GI side effects |
| Phenobarbital | Anticonvulsant | Load: 20 mg/kg IV; Maint: 3–5 mg/kg/day | First-line for neonatal seizures; target level 20–40 mcg/mL |
| Inhaled nitric oxide (iNO) | Pulmonary vasodilator | Start 20 ppm; wean by 5 ppm | PPHN; do not discontinue abruptly; monitor methemoglobin |
| Dopamine | Vasopressor | 2–20 mcg/kg/min | First-line pressor; low-dose (<5) = renal; mid (5–10) = cardiac; high (>10) = vasoconstrictive |
| Dobutamine | Inotrope | 5–20 mcg/kg/min | Cardiogenic shock; may cause hypotension at high doses (vasodilation) |
| Epinephrine | Vasopressor/inotrope | 0.01–0.03 mg/kg IV (resuscitation); infusion 0.05–1 mcg/kg/min | Refractory hypotension; cardiac arrest per NRP |
| Milrinone | PDE-3 inhibitor | Load 50 mcg/kg; infusion 0.33–0.99 mcg/kg/min | Inodilator; PPHN, low cardiac output; watch for hypotension |
| Furosemide | Loop diuretic | 1–2 mg/kg/dose IV/PO q12–24h | BPD, CHF; monitor electrolytes, nephrocalcinosis with chronic use |
| Morphine | Opioid analgesic | 0.05–0.1 mg/kg IV q4h; NAS: 0.04–0.08 mg/kg PO q3–4h | Analgesia; NAS treatment; respiratory depression risk |
| Vitamin A | Fat-soluble vitamin | 5,000 IU IM 3×/week × 4 weeks | BPD prevention in ELBW; modest benefit |
| Iron supplementation | Mineral | 2–4 mg/kg/day elemental iron PO | Start at 2–4 weeks or when on full feeds; preterm infants |
27 Normal Neonatal Values
Vital Signs by Gestational Age
| Parameter | 24–28 wk | 28–32 wk | 32–36 wk | Term (≥37 wk) |
|---|---|---|---|---|
| Heart rate (bpm) | 140–180 | 130–170 | 120–160 | 120–160 |
| Respiratory rate | 40–70 | 40–60 | 30–60 | 30–60 |
| Systolic BP (mmHg) | 35–55 | 40–60 | 50–70 | 60–80 |
| Mean BP (mmHg) | 24–40 | 30–45 | 35–55 | 40–60 |
| Temperature (°C) | 36.5–37.5 (axillary) for all gestational ages | |||
Rule of thumb: mean BP (mmHg) should approximate gestational age in weeks during the first days of life (e.g., 28 weeks GA → target MAP ~28 mmHg).
Laboratory Reference Ranges
| Test | Preterm | Term |
|---|---|---|
| Hemoglobin (g/dL) | 13–20 (cord) | 14–22 (cord) |
| Hematocrit (%) | 40–60 | 42–65 |
| WBC (×103/μL) | 5–30 | 9–30 |
| Platelets (×103/μL) | 100–300 | 150–400 |
| Glucose (mg/dL) | 40–80 | 40–100 |
| Calcium total (mg/dL) | 6.5–10.0 | 7.6–10.4 |
| Ionized Ca (mmol/L) | 1.0–1.3 | 1.1–1.4 |
| Sodium (mEq/L) | 133–146 | 133–146 |
| Potassium (mEq/L) | 4.5–7.0 (DOL 1–3) | 3.7–5.9 |
| Creatinine (mg/dL) | 0.3–1.0 (reflects maternal; falls over first week) | 0.3–1.0 (DOL 1); 0.2–0.4 (DOL 7) |
| Total bilirubin (mg/dL) | Variable; see nomogram | Peak 5–12 (DOL 3–5) |
| Direct bilirubin (mg/dL) | <1.0 | <1.0 |
| Total protein (g/dL) | 4.0–6.0 | 5.0–7.4 |
| Albumin (g/dL) | 2.0–3.6 | 2.5–3.8 |
CSF Values
| Parameter | Preterm | Term |
|---|---|---|
| WBC (cells/μL) | 0–30 (up to 60 normal) | 0–20 |
| Protein (mg/dL) | 65–200 | 40–150 |
| Glucose (mg/dL) | 30–100 | 40–80 |
| Glucose ratio (CSF:serum) | 0.55–1.05 | 0.55–1.05 |
Blood Gas Values
| Parameter | Arterial | Capillary | Venous |
|---|---|---|---|
| pH | 7.30–7.40 | 7.28–7.38 | 7.25–7.35 |
| PaCO2 (mmHg) | 35–45 | 38–50 | 40–55 |
| PaO2 (mmHg) | 50–80 | Not reliable | 30–50 |
| HCO3 (mEq/L) | 18–25 | 18–25 | 18–25 |
| Base deficit | −4 to +4 | −4 to +4 | −4 to +4 |
28 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| ABR | Auditory brainstem response |
| AC | Assist/control (ventilator mode) |
| aEEG | Amplitude-integrated electroencephalography |
| AGA | Appropriate for gestational age |
| AOP | Apnea of prematurity |
| AP-ROP | Aggressive posterior retinopathy of prematurity |
| BAT | Brown adipose tissue |
| BPD | Bronchopulmonary dysplasia |
| BRUE | Brief resolved unexplained event |
| CDH | Congenital diaphragmatic hernia |
| CGA | Corrected gestational age |
| CHD | Congenital heart disease |
| CMV | Cytomegalovirus |
| CNV | Continuous normal voltage (aEEG pattern) |
| CoNS | Coagulase-negative staphylococci |
| CPAP | Continuous positive airway pressure |
| CRP | C-reactive protein |
| DAT | Direct antiglobulin test (Coombs) |
| DCC | Delayed cord clamping |
| DIC | Disseminated intravascular coagulation |
| DOL | Day of life |
| DPPC | Dipalmitoylphosphatidylcholine |
| EA | Esophageal atresia |
| ECMO | Extracorporeal membrane oxygenation |
| ELBW | Extremely low birth weight (<1,000 g) |
| ENaC | Epithelial sodium channel |
| EOS | Early-onset sepsis |
| EPO | Erythropoietin |
| ESC | Eat-sleep-console |
| ETT | Endotracheal tube |
| FiO2 | Fraction of inspired oxygen |
| FRC | Functional residual capacity |
| GA | Gestational age |
| GBS | Group B Streptococcus |
| GH | Growth hormone |
| GIR | Glucose infusion rate |
| HFJV | High-frequency jet ventilation |
| HFNC | High-flow nasal cannula |
| HFOV | High-frequency oscillatory ventilation |
| HIE | Hypoxic-ischemic encephalopathy |
| HMF | Human milk fortifier |
| HPA | Human platelet antigen |
| HSV | Herpes simplex virus |
| hsPDA | Hemodynamically significant PDA |
| IAP | Intrapartum antibiotic prophylaxis |
| IDM | Infant of diabetic mother |
| iNO | Inhaled nitric oxide |
| INSURE | Intubate-surfactant-extubate |
| IUGR | Intrauterine growth restriction |
| IVH | Intraventricular hemorrhage |
| IWL | Insensible water loss |
| LBW | Low birth weight (<2,500 g) |
| LGA | Large for gestational age |
| LISA | Less invasive surfactant administration |
| LOS | Late-onset sepsis |
| MAP | Mean airway pressure |
| MAS | Meconium aspiration syndrome |
| MAT | Medication-assisted treatment |
| MIST | Minimally invasive surfactant therapy |
| MSAF | Meconium-stained amniotic fluid |
| NAIT | Neonatal alloimmune thrombocytopenia |
| NAS | Neonatal abstinence syndrome |
| NBS | New Ballard Score |
| NEC | Necrotizing enterocolitis |
| NICHD | National Institute of Child Health and Human Development |
| NICU | Neonatal intensive care unit |
| NIPPV | Nasal intermittent positive pressure ventilation |
| NOWS | Neonatal opioid withdrawal syndrome |
| NPO | Nil per os (nothing by mouth) |
| NRP | Neonatal Resuscitation Program |
| NTE | Neutral thermal environment |
| OAE | Otoacoustic emissions |
| OI | Oxygenation index |
| PDA | Patent ductus arteriosus |
| PEEP | Positive end-expiratory pressure |
| PGE1 | Prostaglandin E1 |
| PIE | Pulmonary interstitial emphysema |
| PIP | Peak inspiratory pressure |
| PMA | Postmenstrual age |
| PPHN | Persistent pulmonary hypertension of the newborn |
| PPV | Positive pressure ventilation |
| PS | Pressure support |
| PVL | Periventricular leukomalacia |
| PVHI | Periventricular hemorrhagic infarction |
| PVR | Pulmonary vascular resistance |
| RDS | Respiratory distress syndrome |
| ROM | Rupture of membranes |
| ROP | Retinopathy of prematurity |
| SGA | Small for gestational age |
| SIMV | Synchronized intermittent mandatory ventilation |
| SNHL | Sensorineural hearing loss |
| SVR | Systemic vascular resistance |
| TAPVR | Total anomalous pulmonary venous return |
| TEF | Tracheoesophageal fistula |
| TGA | Transposition of the great arteries |
| TH | Therapeutic hypothermia |
| TORCH | Toxoplasma, Other, Rubella, CMV, HSV |
| TPN | Total parenteral nutrition |
| TSB | Total serum bilirubin |
| TTN | Transient tachypnea of the newborn |
| UCP-1 | Uncoupling protein 1 (thermogenin) |
| UGT | UDP-glucuronosyltransferase |
| UVC | Umbilical venous catheter |
| VACTERL | Vertebral, Anorectal, Cardiac, TE fistula, Renal, Limb |
| VEGF | Vascular endothelial growth factor |
| VG | Volume guarantee (ventilator mode) |
| VLBW | Very low birth weight (<1,500 g) |