Pain Management
Acute and chronic pain, nociceptive and neuropathic mechanisms, opioid pharmacology, interventional procedures, neuromodulation, multimodal analgesia, and every scoring tool, drug conversion, and treatment algorithm across the full scope of pain medicine.
01 Pain Neuroanatomy & Physiology
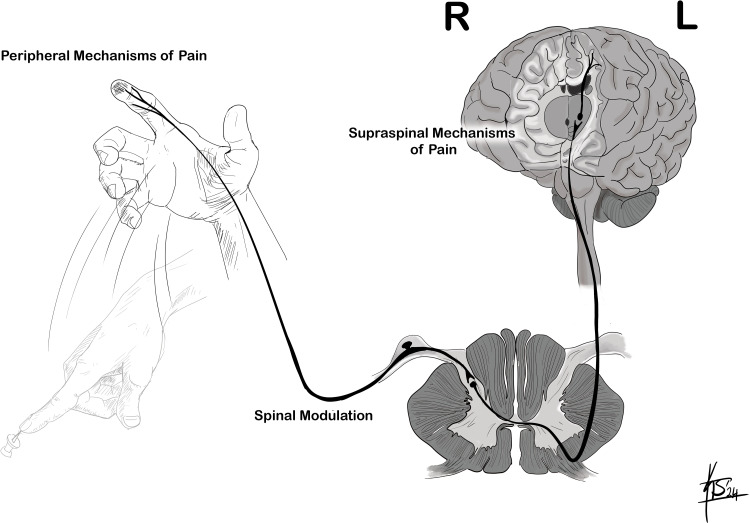
Pain perception is a complex process involving peripheral transduction, spinal cord transmission, supraspinal processing, and descending modulation. Understanding these mechanisms is essential for selecting targeted interventions at every level of the pain pathway.
Peripheral Nociceptors & Primary Afferents
Nociceptors are free nerve endings of primary afferent neurons whose cell bodies reside in the dorsal root ganglia (DRG) or trigeminal ganglion. They respond to noxious mechanical, thermal, or chemical stimuli. Two principal fiber types carry nociceptive information:
| Feature | A-delta Fibers | C Fibers |
|---|---|---|
| Diameter | 2–5 µm (thinly myelinated) | 0.4–1.2 µm (unmyelinated) |
| Conduction velocity | 5–30 m/s | 0.5–2 m/s |
| Pain quality | Sharp, well-localized “first pain” | Dull, burning, aching “second pain” |
| Modality | Mechanical and mechanothermal nociceptors | Polymodal nociceptors (mechanical, thermal, chemical) |
| Neurotransmitters | Glutamate | Glutamate, substance P, CGRP |
| Laminar termination | Laminae I and V of dorsal horn | Laminae I and II (substantia gelatinosa) |
A-beta fibers (large, myelinated, 30–70 m/s) normally carry innocuous touch but may transmit pain signals after peripheral or central sensitization, contributing to allodynia (pain from normally non-painful stimuli).
Dorsal Horn Processing & Gate Control Theory
The dorsal horn of the spinal cord is the first major relay and processing center for nociceptive input. The Rexed laminae organize sensory processing: Lamina I (marginal zone) receives A-delta and C fibers; Lamina II (substantia gelatinosa) is rich in inhibitory interneurons; Lamina V contains wide dynamic range (WDR) neurons that receive both nociceptive and non-nociceptive input and are critical to central sensitization.
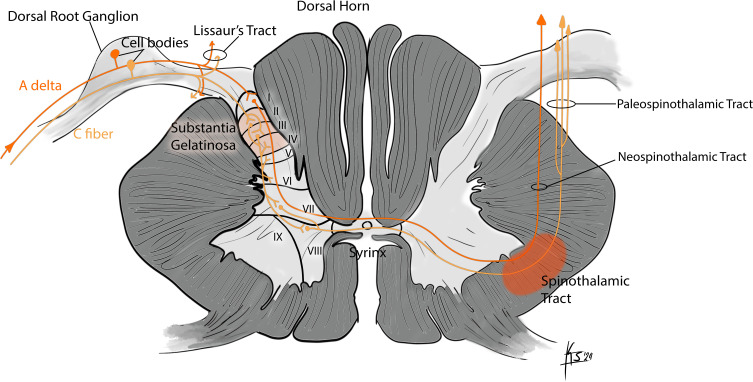
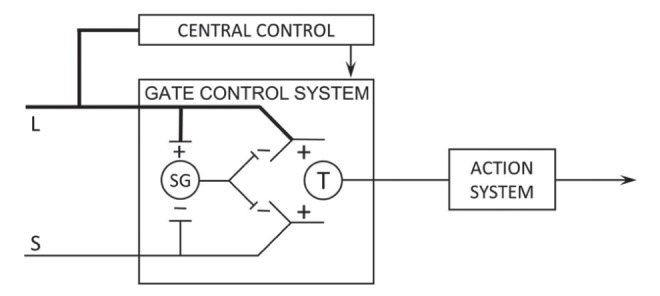
Non-nociceptive input via large-diameter A-beta fibers activates inhibitory interneurons in the substantia gelatinosa, which “close the gate” on nociceptive C-fiber transmission to projection neurons. This explains why rubbing an injured area reduces pain, and provides the theoretical basis for transcutaneous electrical nerve stimulation (TENS) and spinal cord stimulation (SCS). Conversely, loss of A-beta input (as in peripheral neuropathy) “opens the gate,” facilitating pain transmission.
Ascending Pain Pathways
| Tract | Origin | Decussation | Destination | Function |
|---|---|---|---|---|
| Spinothalamic (lateral) | Laminae I, V | Ventral white commissure (1–2 levels above entry) | VPL thalamus → S1/S2 cortex | Pain localization, intensity (sensory-discriminative) |
| Spinoreticular | Laminae VII, VIII | Bilateral | Reticular formation → medial thalamus | Arousal, autonomic responses, affective-motivational |
| Spinomesencephalic | Laminae I, V | Contralateral | PAG, superior colliculus | Activates descending modulation |
| Spinohypothalamic | Laminae I, V, X | Bilateral | Hypothalamus | Neuroendocrine and autonomic responses to pain |
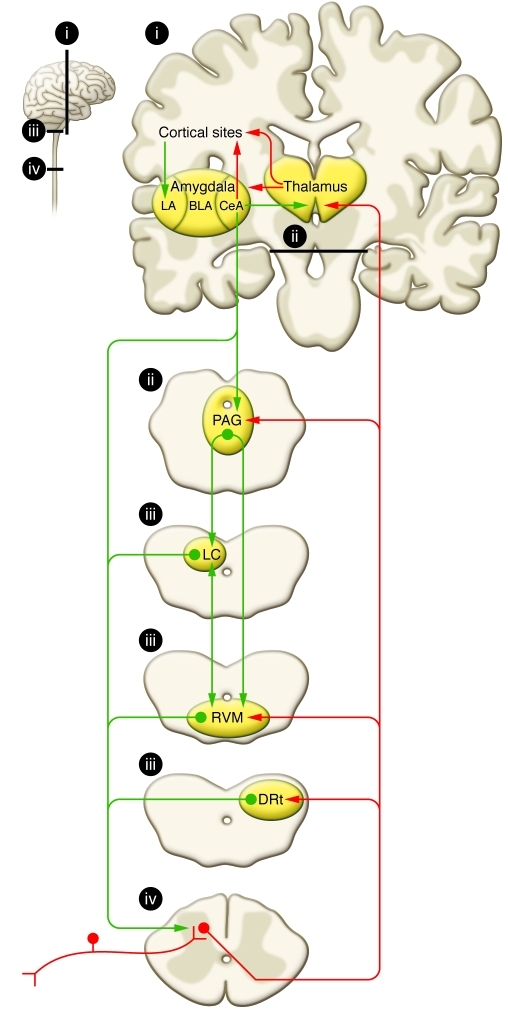
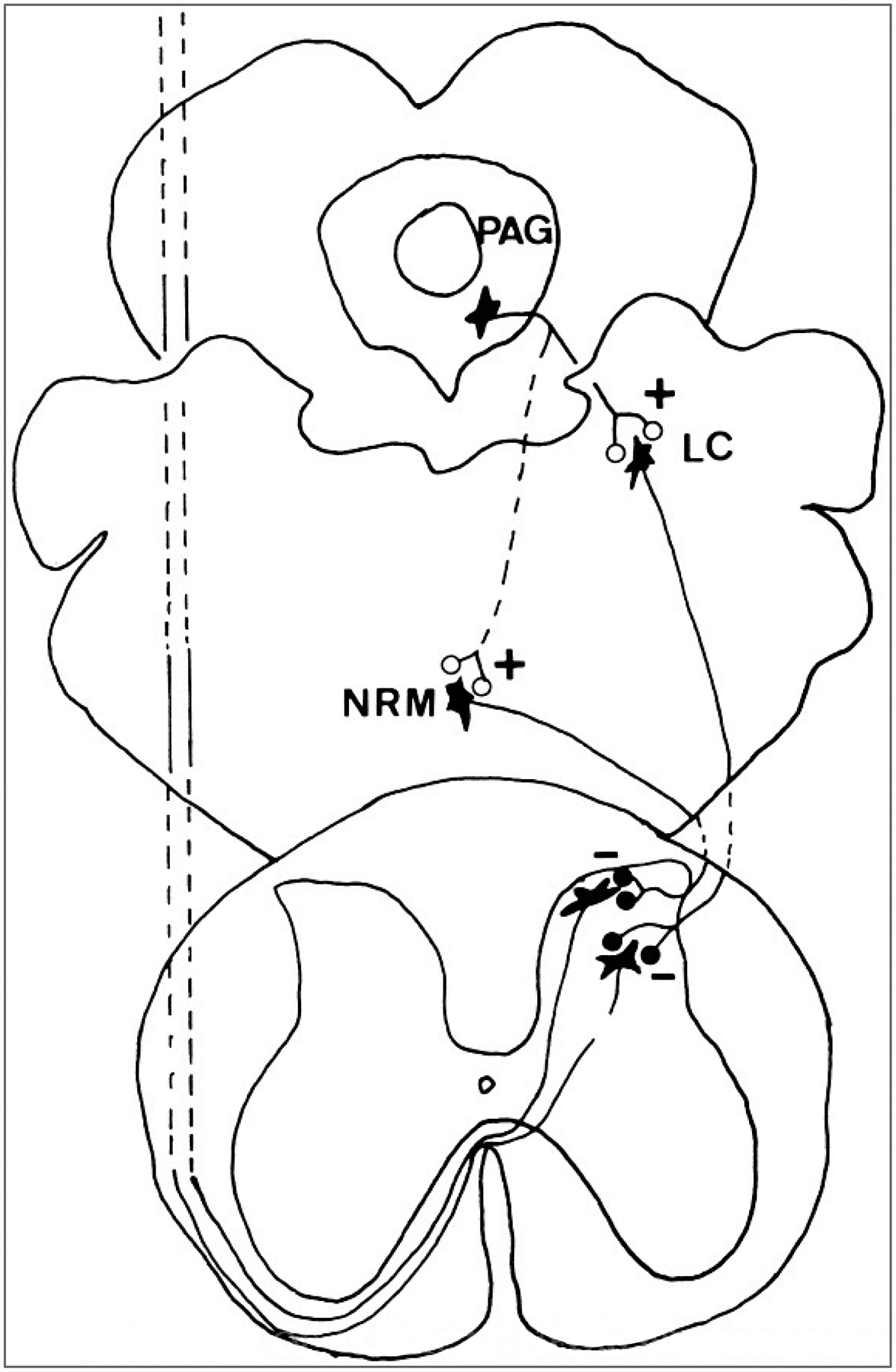
Descending Modulation
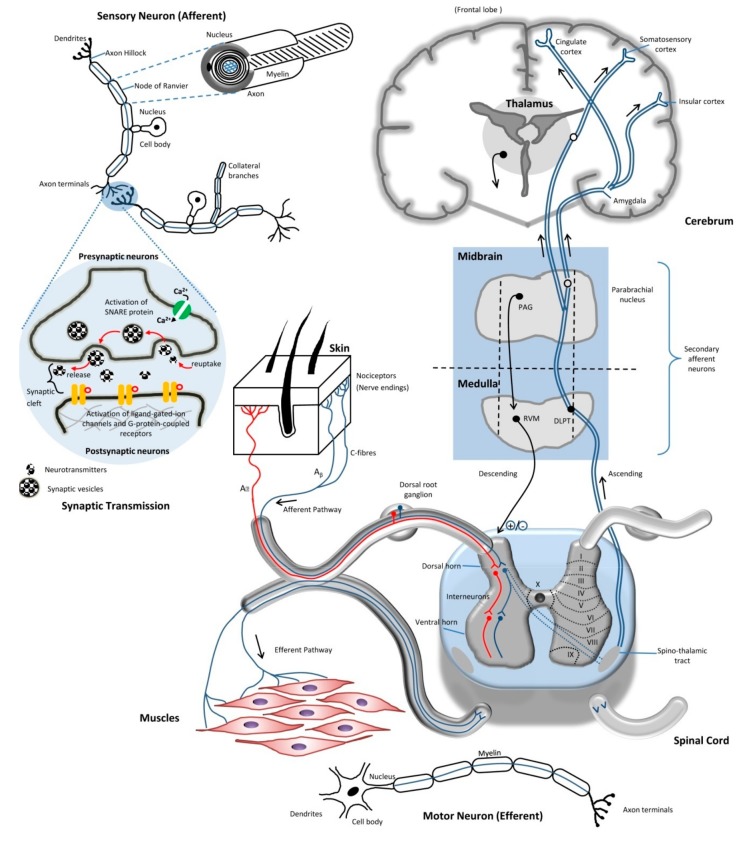
The periaqueductal gray (PAG) receives input from the cortex, hypothalamus, and amygdala, and projects to the rostral ventromedial medulla (RVM). The RVM contains ON cells (facilitate pain) and OFF cells (inhibit pain). Descending pathways travel in the dorsolateral funiculus and release serotonin (5-HT) and norepinephrine (NE) at the dorsal horn, activating enkephalinergic interneurons. This is the mechanism of action of SNRIs (duloxetine, venlafaxine) and TCAs in pain management — they augment descending inhibition by blocking reuptake of 5-HT and NE.
Peripheral & Central Sensitization
Peripheral sensitization occurs when tissue injury releases an “inflammatory soup” (bradykinin, prostaglandins, histamine, serotonin, nerve growth factor, H+, ATP) that lowers the activation threshold of nociceptors via TRPV1 and sodium channels. This manifests as primary hyperalgesia at the site of injury. NSAIDs and corticosteroids target this mechanism.
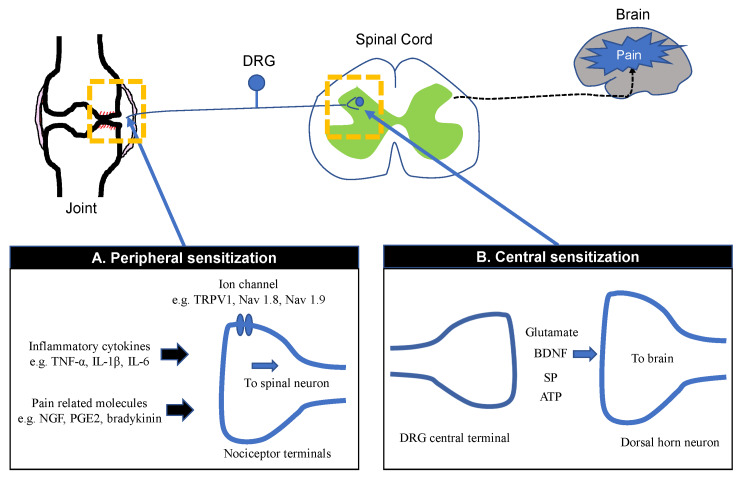
Central sensitization is increased excitability of dorsal horn neurons driven by sustained C-fiber input. Key mechanisms include: (1) Wind-up — progressive increase in action potential output of dorsal horn neurons with repetitive C-fiber stimulation at constant intensity, mediated by temporal summation at NMDA receptors; (2) NMDA receptor activation by glutamate/substance P removes the Mg2+ block, allowing Ca2+ influx and long-term potentiation (LTP); (3) Microglial activation releasing proinflammatory cytokines (TNF-alpha, IL-1beta, IL-6); (4) Loss of inhibitory interneuron function (GABAergic/glycinergic disinhibition).
- Allodynia — pain from a stimulus that does not normally cause pain (e.g., light touch)
- Hyperalgesia — increased pain from a stimulus that normally causes pain
- Secondary hyperalgesia — increased pain sensitivity in uninjured tissue surrounding the injury (central mechanism)
- Wind-up — progressive increase in pain with repetitive stimulation at identical intensity
- Temporal summation — increased pain perception when stimuli are repeated at ≥0.3 Hz
- Referred pain — pain perceived at a location distant from the source, due to convergence of visceral and somatic afferents on shared WDR neurons
Neuroplasticity in Chronic Pain
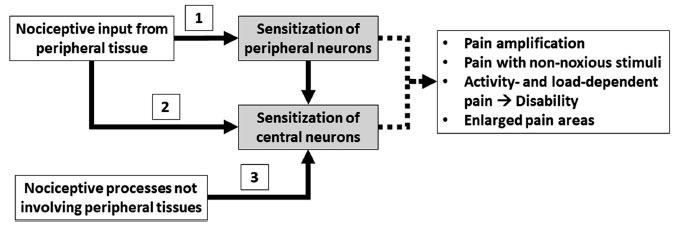
Persistent nociceptive input drives structural and functional changes in the CNS: dorsal horn neuron phenotypic switching (A-beta fibers expressing substance P), cortical reorganization (shrinkage of dorsal lateral prefrontal cortex, expansion of pain-processing areas on fMRI), hippocampal volume loss, and alterations in the default mode network. These changes explain why chronic pain behaves as a disease of the nervous system rather than merely a symptom of ongoing tissue damage.
Key Neurotransmitters & Receptors in Pain Processing
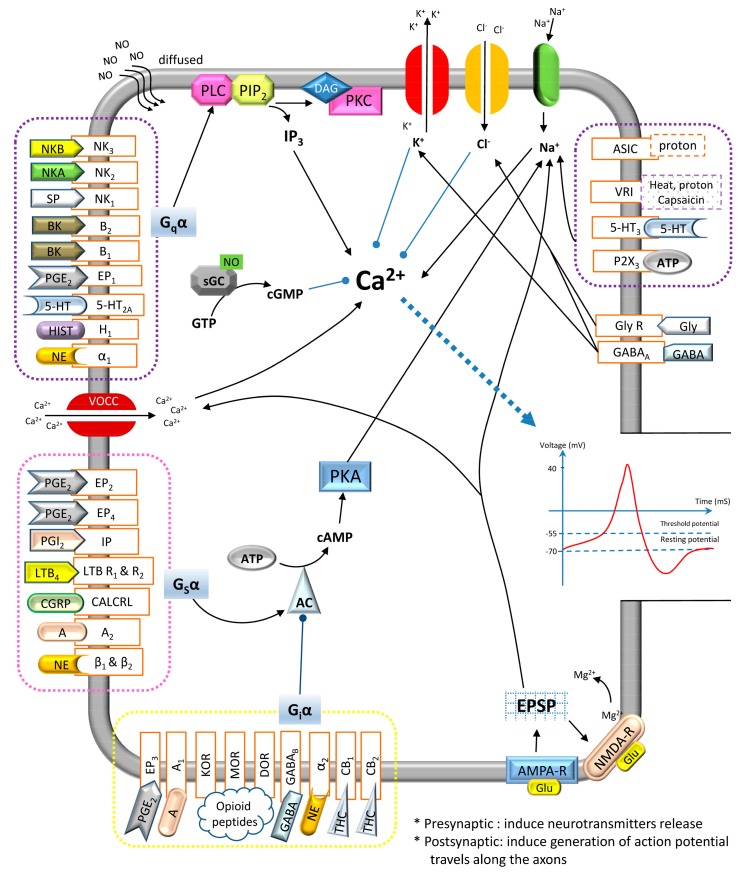
| Molecule / Receptor | Role in Pain | Pharmacologic Target |
|---|---|---|
| Glutamate / NMDA receptor | Primary excitatory NT; NMDA activation critical for wind-up and central sensitization | Ketamine, methadone, memantine, magnesium |
| Substance P / NK-1 receptor | Released by C fibers; facilitates nociceptive transmission in dorsal horn | Capsaicin depletes substance P; NK-1 antagonists (aprepitant) for CINV |
| GABA / GABA-A, GABA-B | Primary inhibitory NT in dorsal horn; loss of GABAergic inhibition promotes central sensitization | Baclofen (GABA-B), benzodiazepines (GABA-A); gabapentinoids act on Ca2+ channels, not GABA receptors |
| Serotonin (5-HT) | Descending modulation from RVM; both facilitatory and inhibitory depending on receptor subtype | SNRIs, TCAs, triptans (5-HT1B/1D for migraine) |
| Norepinephrine (NE) | Descending inhibition from locus coeruleus via alpha-2 receptors in dorsal horn | SNRIs, TCAs, clonidine, dexmedetomidine |
| Prostaglandin E2 (PGE2) | Peripheral sensitization; lowers nociceptor threshold via EP receptors | NSAIDs block COX-mediated PGE2 synthesis |
| TRPV1 (vanilloid receptor) | Heat/capsaicin-activated ion channel on C fibers; mediates thermal hyperalgesia | Capsaicin 8% patch (agonist → desensitization/defunctionalization) |
| Voltage-gated Na+ channels (Nav1.7, 1.8, 1.9) | Generate and propagate action potentials in nociceptors; gain-of-function mutations cause erythromelalgia | Carbamazepine, oxcarbazepine, lidocaine, lacosamide |
02 Pain Classification & Assessment
Mechanistic Classification
| Type | Mechanism | Character | Examples | Treatment Targets |
|---|---|---|---|---|
| Nociceptive — Somatic | Activation of nociceptors in skin, bone, muscle, joints | Well-localized, sharp, aching, throbbing | Fractures, OA, surgical incisions, muscle strain | NSAIDs, acetaminophen, opioids, local anesthetics |
| Nociceptive — Visceral | Activation of nociceptors in hollow/solid organs | Diffuse, crampy, pressure, referred | Pancreatitis, bowel obstruction, renal colic, angina | Opioids, antispasmodics, nerve blocks (celiac plexus) |
| Neuropathic | Lesion or disease of somatosensory nervous system | Burning, shooting, electric, tingling, numbness | DPN, PHN, trigeminal neuralgia, radiculopathy, CRPS-II | Gabapentinoids, SNRIs, TCAs, topical lidocaine, SCS |
| Nociplastic | Altered nociception without tissue damage or nerve lesion (central sensitization) | Widespread, fluctuating, fatigue, cognitive fog | Fibromyalgia, IBS, tension-type HA, interstitial cystitis | SNRIs, gabapentinoids, exercise, CBT, no opioids |
Temporal Classification
Acute pain (<3 months) is a protective mechanism signaling tissue injury; it correlates with identifiable pathology and is expected to resolve with healing. Chronic pain (≥3 months or beyond expected healing time) is a disease state involving maladaptive neuroplasticity and is often dissociated from ongoing tissue damage. Subacute pain (6 weeks–3 months) represents the transitional period where aggressive intervention may prevent chronification.
Unidimensional Pain Scales
| Scale | Type | Range | Population | Interpretation |
|---|---|---|---|---|
| NRS (Numeric Rating Scale) | Patient self-report | 0–10 | Adults, cognitively intact | 0 = no pain; 1–3 mild; 4–6 moderate; 7–10 severe |
| VAS (Visual Analog Scale) | Patient self-report | 0–100 mm line | Adults, research settings | Patient marks a point on a 100 mm line; measured in mm |
| Wong-Baker FACES | Patient self-report (visual) | 0–10 (6 faces) | Children ≥3 years, cognitively impaired adults | Patient selects the face matching their pain level |
| FLACC | Behavioral observation | 0–10 | Children 2 months–7 years, nonverbal | Face, Legs, Activity, Cry, Consolability — each 0–2 |
| CPOT | Behavioral observation | 0–8 | ICU patients, intubated/sedated | Facial expression, body movements, muscle tension, ventilator compliance — each 0–2 |
Multidimensional Assessment Tools
The McGill Pain Questionnaire (MPQ) assesses four dimensions: sensory (e.g., throbbing, shooting, stabbing), affective (e.g., tiring, sickening, fearful), evaluative (overall intensity), and miscellaneous. The short form (SF-MPQ-2) includes 22 descriptors rated 0–10. The Brief Pain Inventory (BPI) measures pain intensity (4 items: worst, least, average, current pain) and pain interference (7 items: general activity, mood, walking, work, relations, sleep, enjoyment) on 0–10 scales.
A rapid, validated screening tool for chronic pain assessment in primary care:
- P — What number best describes your Pain on average in the past week? (0–10)
- E — What number best describes how pain has interfered with your Enjoyment of life? (0–10)
- G — What number best describes how pain has interfered with your General activity? (0–10)
Mean score ≥4 suggests clinically significant pain requiring further assessment. Track over time to monitor treatment response (clinically meaningful change ≥1.0 point).
Functional Assessment
Pain intensity scores alone are insufficient. A comprehensive pain evaluation must include: functional status (ADLs, IADLs, employment, mobility), psychological screening (PHQ-9 for depression, GAD-7 for anxiety, PCS for pain catastrophizing), sleep quality, substance use history, and social determinants. Document the “5 A’s” of pain management at each visit: Analgesia, Activity, Adverse effects, Aberrant behavior, and Affect.
03 Terminology & Abbreviations
| Term | Definition |
|---|---|
| Allodynia | Pain from a stimulus that does not normally cause pain |
| Hyperalgesia | Increased pain from a normally painful stimulus |
| Hyperpathia | Exaggerated pain response, especially to repetitive stimuli, with increased threshold |
| Dysesthesia | Unpleasant abnormal sensation, spontaneous or evoked |
| Paresthesia | Abnormal sensation (tingling, prickling) that is not unpleasant |
| Analgesia | Absence of pain in response to normally painful stimulation |
| Anesthesia dolorosa | Pain in an area that is numb or has no sensation |
| Causalgia | Burning pain after peripheral nerve injury (CRPS Type II) |
| Deafferentation pain | Pain due to loss of afferent sensory input (e.g., phantom limb, brachial plexus avulsion) |
| Nociception | Neural process of encoding noxious stimuli (not synonymous with pain) |
| Central sensitization | Increased excitability of central neurons, leading to amplified pain processing |
| Wind-up | Progressive increase in dorsal horn neuron output to repetitive C-fiber stimulation |
| Tolerance | Decreased effect of a drug with repeated use, requiring dose escalation for same effect |
| Physical dependence | Adaptation producing withdrawal symptoms upon abrupt discontinuation or dose reduction |
| Addiction (OUD) | Compulsive use despite harm; loss of control; craving; continued use despite consequences |
| Pseudoaddiction | Drug-seeking behavior driven by undertreated pain (controversial concept) |
| Breakthrough pain | Transient flare of pain exceeding controlled baseline — spontaneous or incident-related |
| Equianalgesic dose | Dose of one opioid that provides analgesia equivalent to a reference dose of another |
| MME | Morphine milligram equivalents — standardized unit for comparing opioid doses |
| Opioid-induced hyperalgesia (OIH) | Paradoxical increased pain sensitivity caused by opioid exposure |
Common Abbreviations in Pain Medicine
| Abbreviation | Meaning | Abbreviation | Meaning |
|---|---|---|---|
| NRS | Numeric Rating Scale | VAS | Visual Analog Scale |
| ESI | Epidural Steroid Injection | MBB | Medial Branch Block |
| RFA | Radiofrequency Ablation | SCS | Spinal Cord Stimulation |
| DRG | Dorsal Root Ganglion | CRPS | Complex Regional Pain Syndrome |
| MME | Morphine Milligram Equivalent | ORT | Opioid Risk Tool |
| PDMP | Prescription Drug Monitoring Program | UDS | Urine Drug Screen |
| NMDA | N-Methyl-D-Aspartate | SNRI | Serotonin-Norepinephrine Reuptake Inhibitor |
| TCA | Tricyclic Antidepressant | COX | Cyclooxygenase |
| PAG | Periaqueductal Gray | RVM | Rostral Ventromedial Medulla |
| TENS | Transcutaneous Electrical Nerve Stimulation | FBSS | Failed Back Surgery Syndrome |
| OIH | Opioid-Induced Hyperalgesia | OIAD | Opioid-Induced Androgen Deficiency |
| ERAS | Enhanced Recovery After Surgery | IDDS | Intrathecal Drug Delivery System |
04 Acetaminophen & NSAIDs
Acetaminophen
Mechanism: Centrally acting; proposed mechanisms include inhibition of COX-3 (a splice variant of COX-1 in the CNS), augmentation of descending serotonergic inhibition, and activation of the endocannabinoid system via AM404 metabolite acting on TRPV1 and CB1 receptors. Lacks peripheral anti-inflammatory activity.
| Parameter | Detail |
|---|---|
| Standard dose | 650–1000 mg PO/PR q4–6h; IV 1000 mg q6h (650 mg if <50 kg) |
| Max daily dose (healthy adult) | 4000 mg/day (FDA recommends ≤3000 mg/day for OTC use) |
| Max daily dose (hepatic impairment/alcohol) | ≤2000 mg/day |
| Hepatotoxicity threshold | ≥150 mg/kg or ≥7.5 g single ingestion → NAC protocol |
| Onset / Duration | PO: 30–60 min onset, 4–6 h duration; IV: 5–10 min onset |
| Advantages | No GI/renal/platelet toxicity; safe in renal failure; pregnancy Category B |
NSAIDs — Mechanism & Classification
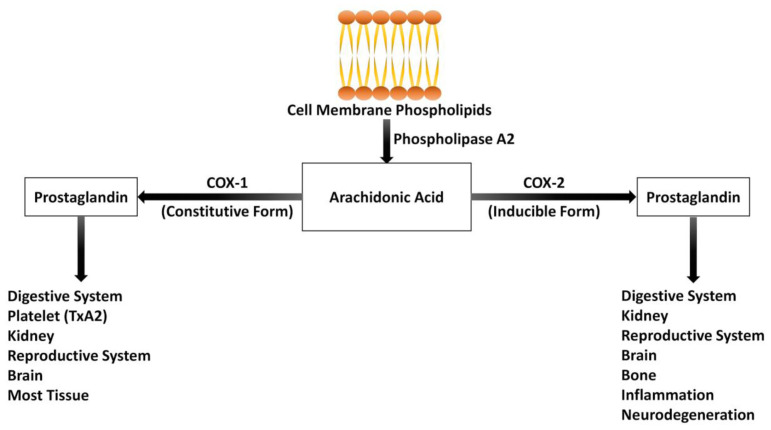
NSAIDs inhibit cyclooxygenase (COX), blocking conversion of arachidonic acid to prostaglandins (PGE2, PGI2) and thromboxane A2. COX-1 is constitutive (gastric mucosal protection, platelet aggregation, renal blood flow). COX-2 is inducible at sites of inflammation (also constitutive in kidney and vascular endothelium).
| Drug | COX Selectivity | Dose | Half-life | Key Notes |
|---|---|---|---|---|
| Ibuprofen | Non-selective | 200–800 mg PO q6–8h (max 3200 mg/day) | 2–4 h | OTC; lowest CV risk among non-selective NSAIDs |
| Naproxen | Non-selective | 250–500 mg PO BID (max 1500 mg/day) | 12–17 h | Lowest CV risk of all NSAIDs; preferred in CV patients |
| Diclofenac | Slightly COX-2 selective | 50 mg PO TID or 75 mg BID (max 150 mg/day) | 1–2 h | Also available topical (1% gel, 1.5% solution, patch) |
| Ketorolac | Non-selective (potent) | 15–30 mg IV/IM q6h; 10 mg PO q4–6h | 5–6 h | Max 5 days total; most potent parenteral NSAID |
| Meloxicam | Preferential COX-2 | 7.5–15 mg PO daily | 15–20 h | Once daily dosing; intermediate COX-2 selectivity |
| Celecoxib | Selective COX-2 | 100–200 mg PO BID (max 400 mg/day) | 11 h | Reduced GI risk; sulfonamide allergy caution; PRECISION trial showed non-inferior CV safety to naproxen/ibuprofen |
| Indomethacin | Non-selective (potent) | 25–50 mg PO TID (max 200 mg/day) | 4.5 h | Potent; used for gout, hemicrania continua; high GI risk |
- GI: All NSAIDs ↑ risk of ulcers/bleeding (3–5x baseline). Risk factors: age >65, H. pylori, concurrent anticoagulants/corticosteroids, prior GI bleed. Mitigate with PPI co-therapy or COX-2 selective agent
- Renal: Prostaglandin-mediated afferent arteriolar dilation is critical in low-flow states. Avoid in CKD stage ≥4, HF, cirrhosis, hypovolemia. Risk of AIN, papillary necrosis, hyperkalemia, sodium/water retention
- Cardiovascular: All NSAIDs (except naproxen) increase risk of MI and stroke. COX-2 inhibitors shift prostacyclin/thromboxane balance toward prothrombotic state. Contraindicated post-CABG (black box warning)
- Platelet: Aspirin irreversibly inhibits COX-1 in platelets; all other NSAIDs reversibly inhibit — hold 5 half-lives before surgery
Topical NSAIDs
Diclofenac gel 1% (Voltaren): 4g applied QID to affected joint (max 16g/day per joint for lower extremities, 8g/day for upper). Achieves therapeutic tissue concentrations with <5% systemic absorption. NNT of 6.4 for OA knee pain. Preferred in elderly to minimize systemic adverse effects. Diclofenac 1.5% topical solution: 40 drops applied QID; used with dimethyl sulfoxide carrier. Diclofenac epolamine patch (Flector): Applied BID for acute strains/sprains; 180 mg per patch with 10–15 mg delivered over 12 hours.
- No CV/GI/renal risk: Any NSAID (ibuprofen or naproxen preferred for cost and safety data)
- High GI risk, low CV risk: Celecoxib OR non-selective NSAID + PPI
- High CV risk, low GI risk: Naproxen (lowest CV risk); avoid all NSAIDs if possible post-MI/stroke
- High GI + high CV risk: Avoid NSAIDs if possible; if necessary, naproxen + PPI or celecoxib + PPI at lowest dose for shortest duration
- CKD stage ≥4 or acute kidney injury: Avoid ALL NSAIDs including topical (minimal but non-zero systemic absorption)
- Pregnancy: Avoid after 20 weeks (oligohydramnios risk); contraindicated after 30 weeks (premature ductus arteriosus closure)
- Elderly (>65): Topical NSAIDs first-line; if systemic needed, lowest dose, shortest duration, with PPI; Beers criteria caution
05 Neuropathic Pain Agents
First-line agents for neuropathic pain include gabapentinoids, SNRIs, and TCAs. Selection is guided by pain type, comorbidities, side-effect profile, and cost. NNT for ≥50% pain reduction ranges from 3.6 to 7.7 for first-line agents.
| Drug | Mechanism | Starting Dose | Titration | Target/Max | Key Considerations |
|---|---|---|---|---|---|
| Gabapentin | Binds alpha-2-delta subunit of voltage-gated Ca2+ channels; reduces excitatory neurotransmitter release | 100–300 mg QHS | Increase by 300 mg q3–7 days; TID dosing | 1800–3600 mg/day in 3 divided doses | Nonlinear absorption (bioavailability decreases at higher doses); renal dosing required; sedation, dizziness, edema; schedule V in some states |
| Pregabalin | Same as gabapentin; linear pharmacokinetics | 75 mg BID or 50 mg TID | Increase to 150 mg BID after 1 week; then 300 mg BID after 1 week | 300 mg BID (600 mg/day) | Schedule V (DEA); faster onset than gabapentin; approved for DPN, PHN, fibromyalgia, SCI pain |
| Duloxetine | SNRI; augments descending 5-HT/NE inhibition | 30 mg daily × 1 week | Increase to 60 mg daily | 60–120 mg daily | Approved DPN, fibromyalgia, chronic MSK pain, OA; avoid in hepatic impairment, CrCl <30; nausea most common SE; discontinuation syndrome |
| Venlafaxine | SNRI (NE effect ≥150 mg) | 37.5 mg daily | Increase by 75 mg q1–2 weeks | 150–225 mg daily | NE reuptake inhibition requires ≥150 mg; hypertension at higher doses; less evidence than duloxetine for pain |
| Amitriptyline | TCA; blocks 5-HT/NE reuptake, Na+ channels, NMDA receptors | 10–25 mg QHS | Increase by 10–25 mg q1–2 weeks | 75 mg QHS (analgesic ceiling) | Strong evidence for neuropathic pain; anticholinergic SEs limit use in elderly; ECG before starting (>50 years); QTc prolongation; weight gain, sedation |
| Nortriptyline | TCA; less anticholinergic than amitriptyline | 10–25 mg QHS | Same as amitriptyline | 75 mg QHS | Preferred TCA in elderly due to fewer anticholinergic effects; same efficacy as amitriptyline |
| Carbamazepine | Na+ channel blockade | 100 mg BID | Increase by 200 mg q3–7 days | 400–1200 mg/day in divided doses | First-line for trigeminal neuralgia (NNT 1.7); HLA-B*1502 testing in Asian patients (SJS risk); autoinduction; drug interactions; CBC/LFT monitoring |
| Oxcarbazepine | Na+ channel blockade | 150 mg BID | Increase by 300 mg q1 week | 1200–2400 mg/day | Alternative to carbamazepine for trigeminal neuralgia; fewer drug interactions; risk of hyponatremia |
Topical Agents for Neuropathic Pain
Lidocaine 5% patch: Apply up to 3 patches for 12 hours on, 12 hours off. Approved for PHN. Minimal systemic absorption. Can be used as first-line in localized neuropathic pain, especially in elderly. Capsaicin 8% patch (Qutenza): Applied for 60 minutes by a healthcare professional; depletes substance P from C fibers via TRPV1 agonism. Approved for PHN; can provide 3 months of relief per application. Pre-treat area with topical lidocaine. Capsaicin 0.025–0.075% cream: OTC; apply TID–QID; requires 2–4 weeks of consistent use for efficacy; burning on initial application is common and often limits adherence.
Neuropathic Pain Treatment Algorithm
- First-line (choose based on comorbidities):
- Gabapentin or pregabalin — preferred if anxiety, insomnia, or no depression
- Duloxetine or venlafaxine — preferred if concurrent depression, anxiety, fibromyalgia, or chronic MSK pain
- TCA (amitriptyline/nortriptyline) — effective and cheap; avoid in elderly, cardiac disease, or overdose risk; nortriptyline preferred in elderly for fewer anticholinergic effects
- Topical lidocaine 5% — first-line for localized neuropathic pain, especially PHN in elderly (no systemic effects)
- Second-line (if monotherapy fails after adequate trial 4–8 weeks at therapeutic dose):
- Combination therapy: gabapentinoid + SNRI or gabapentinoid + TCA (avoid SNRI + TCA due to serotonin syndrome risk)
- Capsaicin 8% patch (Qutenza) for localized neuropathic pain
- Tramadol (dual mechanism; weak mu agonism + NRI)
- Third-line: Strong opioids (morphine, oxycodone) in carefully selected patients; botulinum toxin type A (subcutaneous injection for PHN/DPN — NNT 1.9); lacosamide, lamotrigine (limited evidence)
- Fourth-line / Refractory: SCS or DRG stimulation; intrathecal drug delivery; NMDA antagonists (ketamine infusion)
Condition-Specific First-Line Agents
| Condition | Preferred First-Line | Notes |
|---|---|---|
| Diabetic peripheral neuropathy | Duloxetine, pregabalin, gabapentin | All three FDA-approved for DPN; duloxetine preferred if concurrent depression |
| Postherpetic neuralgia | Gabapentin, pregabalin, lidocaine 5% patch, capsaicin 8% | Topicals preferred in elderly; prevention with zoster vaccine more effective than treatment |
| Trigeminal neuralgia | Carbamazepine (NNT 1.7) | Oxcarbazepine as alternative; if refractory → MVD (microvascular decompression) or percutaneous procedures |
| Central post-stroke pain | Amitriptyline, lamotrigine, pregabalin | Gabapentin less effective for central pain; opioids generally ineffective |
| Spinal cord injury pain | Pregabalin (FDA-approved), gabapentin, amitriptyline | Pregabalin only gabapentinoid FDA-approved for SCI neuropathic pain |
| HIV neuropathy | Capsaicin 8% patch, lamotrigine | Gabapentinoids and TCAs show limited efficacy in HIV neuropathy specifically |
06 Muscle Relaxants & Adjuvants
Skeletal Muscle Relaxants
| Drug | Mechanism | Dose | Key Notes |
|---|---|---|---|
| Cyclobenzaprine | Centrally acting; structurally related to TCAs; reduces tonic somatic motor activity at brainstem | 5–10 mg PO TID (or 5 mg QHS for tolerability); ER 15–30 mg daily | Most studied; efficacy best in acute MSK spasm; sedation most common SE; avoid in elderly (Beers list); short-term use (≤2–3 weeks) |
| Tizanidine | Alpha-2 adrenergic agonist; reduces excitatory input at spinal interneurons | 2–4 mg PO TID (max 36 mg/day) | Less sedating than cyclobenzaprine at low doses; monitor LFTs (hepatotoxicity); CYP1A2 substrate — avoid with fluvoxamine, ciprofloxacin; hypotension, dry mouth |
| Baclofen | GABA-B agonist; inhibits monosynaptic and polysynaptic reflexes at spinal level | 5 mg PO TID, titrate by 5 mg q3 days (max 80 mg/day); intrathecal 50–100 mcg test dose | Best evidence for spasticity (MS, SCI, CP); intrathecal baclofen for severe spasticity; abrupt withdrawal → life-threatening syndrome (seizures, hyperthermia, rhabdomyolysis); renal dosing |
| Methocarbamol | Central sedative effect; poorly understood mechanism | 1500 mg PO QID × 2–3 days, then 750–1000 mg TID–QID | Available IV; less sedating; relatively safe in elderly compared to other muscle relaxants |
| Carisoprodol | Metabolized to meprobamate (barbiturate-like); sedative, anxiolytic | 250–350 mg PO TID + QHS | Schedule IV; high abuse potential; produces euphoria; avoid — no evidence of superiority over safer alternatives; withdrawal can be life-threatening |
| Dantrolene | Direct-acting; blocks Ca2+ release from sarcoplasmic reticulum (ryanodine receptor) | 25 mg daily, titrate to 100 mg QID | Only direct-acting relaxant; used for spasticity, malignant hyperthermia, NMS; hepatotoxicity — monitor LFTs |
NMDA Receptor Antagonists
Ketamine is a non-competitive NMDA receptor antagonist with additional activity at opioid, monoaminergic, and cholinergic receptors. In sub-anesthetic doses (0.1–0.5 mg/kg/h IV infusion), it reduces central sensitization, opioid tolerance, and opioid-induced hyperalgesia without causing dissociative anesthesia.
- Acute pain (perioperative): 0.5 mg/kg IV bolus, then 0.1–0.3 mg/kg/h infusion (no need for intubation at these doses)
- CRPS: Outpatient infusions 0.5 mg/kg over 4 hours, daily × 5 days; evidence supports short-term benefit
- Treatment-resistant chronic pain: 0.1–0.5 mg/kg/h × 4–8 hours; may provide weeks of relief
- Opioid-tolerant patients: Perioperative adjunct reduces opioid consumption by 30–50%
- Side effects: Dysphoria, vivid dreams, nystagmus, hypertension, tachycardia; co-administer midazolam 1–2 mg for psychomimetic effects
Other Adjuvants
Magnesium sulfate: NMDA receptor antagonist (Mg2+ blocks the channel pore); 30–50 mg/kg IV bolus then 10–15 mg/kg/h infusion; perioperative adjunct reduces opioid consumption; monitor for hypotension, loss of deep tendon reflexes. Clonidine: Alpha-2 agonist; 0.1–0.3 mg PO BID or transdermal patch; useful adjunct for neuropathic pain and opioid withdrawal; side effects include hypotension, bradycardia, sedation. Dexmedetomidine: Highly selective alpha-2 agonist; 0.2–0.7 mcg/kg/h IV; used as ICU sedation adjunct with analgesic-sparing properties; procedural sedation for interventional pain procedures.
07 Corticosteroids & Anti-Inflammatory Agents
Systemic Corticosteroids
| Agent | Relative Potency | Half-life | Dose / Protocol | Indication |
|---|---|---|---|---|
| Prednisone | 1 (reference) | 18–36 h | Taper: 60 mg × 3d → 40 mg × 3d → 20 mg × 3d → 10 mg × 3d; or 1 mg/kg × 5d then taper over 1–2 weeks | Acute radiculopathy, CRPS flares, cluster headache |
| Methylprednisolone dose pack | 1.25 | 18–36 h | 21 tablets: Day 1: 24 mg; Day 2: 20 mg; Day 3: 16 mg; Day 4: 12 mg; Day 5: 8 mg; Day 6: 4 mg | Acute radiculopathy, inflammatory flares; short-course convenience |
| Dexamethasone | 6.67 | 36–54 h | 4–10 mg PO/IV/IM daily × 3–5 days | Severe radiculopathy, cancer pain, spinal cord compression, cerebral edema |
Injectable Corticosteroids for Pain Procedures
| Agent | Type | Concentration | Common Doses | Notes |
|---|---|---|---|---|
| Triamcinolone acetonide | Particulate | 40 mg/mL | Epidural: 40–80 mg; joint: 20–40 mg (large), 10–20 mg (small) | Most commonly used; long-acting; risk of embolic infarct if injected intra-arterially (transforaminal) |
| Methylprednisolone acetate | Particulate | 40 or 80 mg/mL | Epidural: 40–80 mg; joint: 20–80 mg | Similar to triamcinolone; risk of arachnoiditis with intrathecal injection (contains PEG) |
| Betamethasone sodium phosphate/acetate | Mixed (particulate + soluble) | 6 mg/mL | Epidural: 6–12 mg; joint: 3–6 mg | Smaller particles than triamcinolone; intermediate risk profile |
| Dexamethasone sodium phosphate | Non-particulate (soluble) | 4 or 10 mg/mL | Epidural: 8–10 mg; joint: 4–8 mg | Non-particulate — REQUIRED for cervical transforaminal ESI to avoid embolic stroke; may have shorter duration of effect |
- Maximum 3 epidural steroid injections per spinal region per 12-month period
- Minimum 2-week interval between injections (some guidelines recommend 4–6 weeks)
- No more than 4–6 total epidural steroid injections per year across all regions
- Rationale: Cumulative exogenous corticosteroid burden → HPA axis suppression, bone loss, hyperglycemia, immunosuppression
- Diabetic patients: Warn of blood glucose elevation for 1–2 weeks post-injection; may need temporary insulin adjustment
08 Opioid Receptor Pharmacology
Opioid Receptor Types & Effects
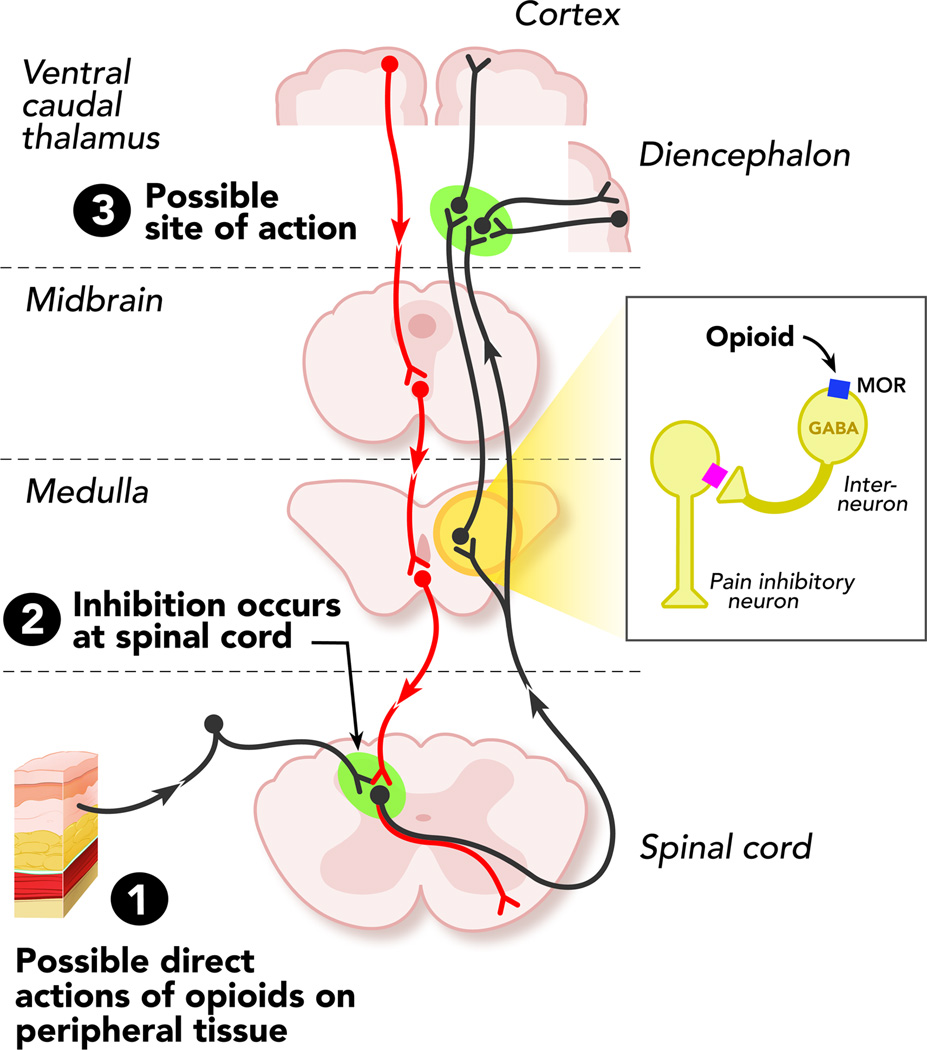
| Receptor | Endogenous Ligand | Location | Effects of Activation | Clinical Relevance |
|---|---|---|---|---|
| Mu (μ) (MOR) | Beta-endorphin, endomorphins | PAG, RVM, dorsal horn (lamina II), GI tract, nucleus accumbens | Analgesia (supraspinal & spinal), euphoria, respiratory depression, miosis, constipation, pruritus, physical dependence, bradycardia | Primary target of most clinical opioids; mu-1 (analgesia) vs mu-2 (respiratory depression, constipation) subtypes proposed |
| Kappa (κ) (KOR) | Dynorphin | Dorsal horn, hypothalamus, limbic system | Analgesia (spinal & visceral), sedation, dysphoria, diuresis, miosis | Butorphanol, nalbuphine, pentazocine act here; ceiling effect for respiratory depression; dysphoria limits abuse potential |
| Delta (δ) (DOR) | Enkephalins (met-, leu-) | Cortex, olfactory bulb, dorsal horn | Analgesia (modulates mu), anxiolysis, antidepressant effects, respiratory depression (minor) | Research target; delta agonists may provide analgesia without dependence; limited clinical agents |
| NOP/ORL1 | Nociceptin/orphanin FQ | Cortex, amygdala, spinal cord | Complex: anti-analgesic supraspinally, analgesic spinally; anxiolysis | Emerging target; nociceptin receptor agonists in development |
Opioid Drug Classifications
| Classification | Definition | Examples |
|---|---|---|
| Full agonist | Binds and fully activates mu receptor; no ceiling for analgesia (ceiling for respiratory depression is death) | Morphine, hydromorphone, fentanyl, oxycodone, hydrocodone, methadone |
| Partial agonist | Binds mu receptor but produces submaximal activation even at full receptor occupancy; ceiling effect | Buprenorphine (partial mu agonist / kappa antagonist) |
| Mixed agonist-antagonist | Agonist at one receptor, antagonist at another | Butorphanol (kappa agonist / mu antagonist), nalbuphine (kappa agonist / mu antagonist), pentazocine (kappa agonist / weak mu antagonist) |
| Full antagonist | Binds receptor without activation; competitively displaces agonists | Naloxone (IV, t½ 30–90 min), naltrexone (PO, t½ 4–12 h) |
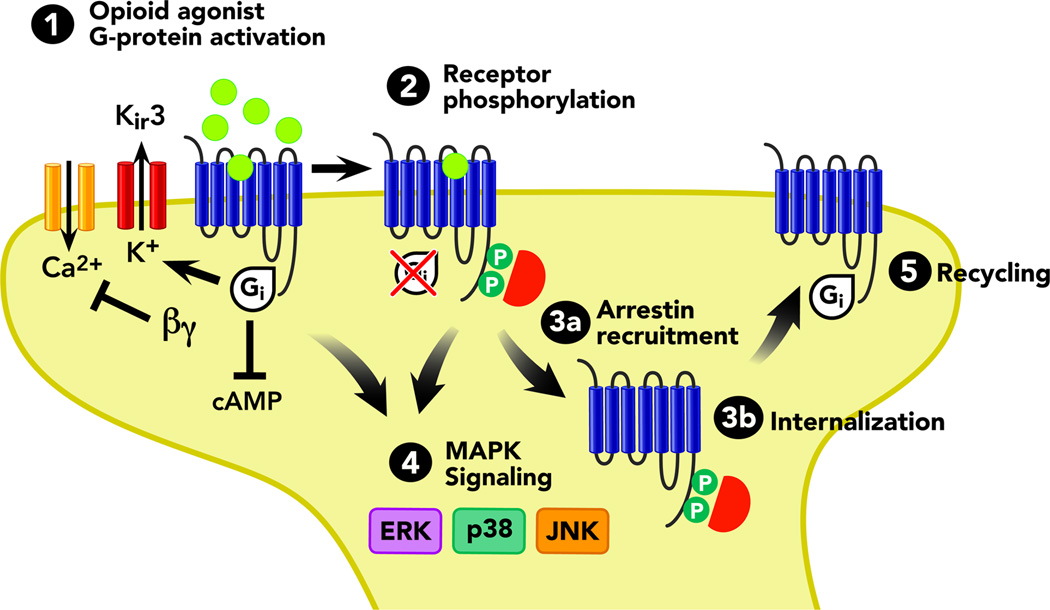
Signaling & Tolerance Mechanisms
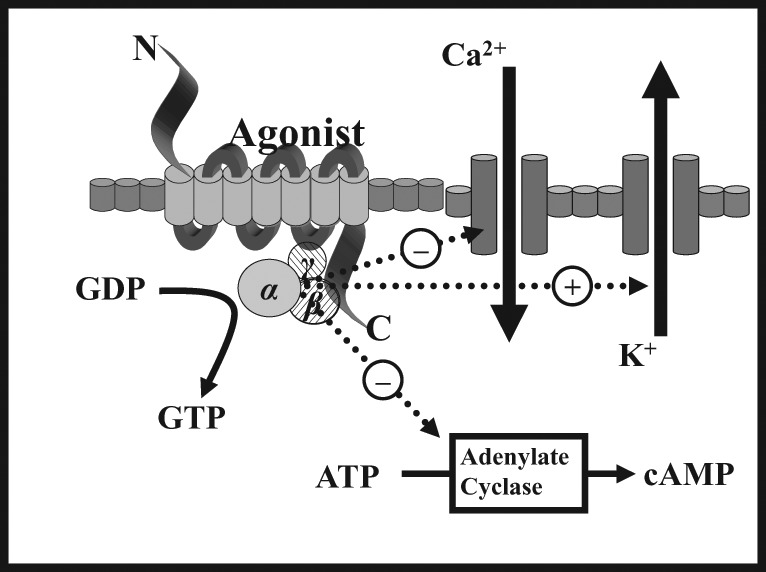
Opioids act via G-protein coupled receptors (GPCRs): Gi/Go activation → (1) inhibition of adenylyl cyclase → decreased cAMP; (2) opening of inwardly rectifying K+ channels (GIRK) → hyperpolarization; (3) closing of voltage-gated Ca2+ channels → decreased neurotransmitter release. Tolerance develops through receptor phosphorylation by GRK, beta-arrestin recruitment leading to receptor internalization, and NMDA receptor-mediated neuroplasticity. Tolerance develops at different rates for different effects: rapid for euphoria and nausea, moderate for analgesia and sedation, slow for constipation and miosis.
OIH is a paradoxical state in which opioid exposure increases pain sensitivity. Distinguished from tolerance: in tolerance, the same stimulus hurts the same but the drug works less well (dose escalation helps); in OIH, pain spreads and worsens diffusely despite dose escalation (dose escalation worsens it). Mechanisms include NMDA receptor upregulation, dynorphin release, enhanced descending facilitation from RVM ON cells, and spinal glial activation. Treatment: opioid dose reduction or rotation, NMDA antagonists (ketamine, methadone), and non-opioid multimodal analgesia.
09 Oral Opioids
| Drug | Starting Dose (Opioid-Naive) | Onset / Duration | Potency vs Morphine PO | Metabolism | Key Considerations |
|---|---|---|---|---|---|
| Morphine IR | 15 mg PO q4h (5–10 mg in elderly/renal) | 30 min / 3–4 h | 1 (reference) | Hepatic glucuronidation: M3G (neuroexcitatory, no analgesia) + M6G (active, 10x potency, renally cleared) | Avoid in renal failure (M6G accumulation → respiratory depression); histamine release; reference standard for equianalgesic dosing |
| Oxycodone IR | 5–10 mg PO q4–6h | 15–30 min / 3–6 h | 1.5x (30 mg morphine = 20 mg oxycodone) | CYP3A4 (major), CYP2D6 (minor → oxymorphone) | Less histamine release and nausea than morphine; available as single agent or with acetaminophen (Percocet) or ibuprofen |
| Hydrocodone | 5–10 mg PO q4–6h | 20–30 min / 4–6 h | 1x (equivalent to morphine PO) | CYP2D6 → hydromorphone (active); CYP3A4 | Schedule II (reclassified 2014); most prescribed opioid in US; always combined with acetaminophen or ibuprofen until single-entity ER (Zohydro) approved |
| Hydromorphone IR | 2–4 mg PO q4–6h; 0.2–1 mg IV q2–3h | 15–30 min PO / 3–4 h | 4–5x PO (30 mg morphine = 6–8 mg hydromorphone) | Hepatic glucuronidation → H3G (neuroexcitatory, renally cleared) | Preferred over morphine in renal impairment (no active metabolite, but H3G still accumulates); 5x potency — careful dose calculation |
| Codeine | 15–60 mg PO q4–6h (max 360 mg/day) | 30–60 min / 4–6 h | 0.15x (200 mg codeine = 30 mg morphine) | CYP2D6 → morphine (active); prodrug | CYP2D6 polymorphisms: ultra-rapid metabolizers (1–7%) → toxic morphine levels; poor metabolizers (5–10%) → no analgesia; BLACK BOX: contraindicated in children <12 and post-tonsillectomy <18 |
| Tramadol | 25–50 mg PO q4–6h (max 400 mg/day) | 60 min / 4–6 h | 0.1x (weak mu agonist) | CYP2D6 → O-desmethyltramadol (active mu agonist, 200x affinity) | Dual mechanism: mu agonism + NE/5-HT reuptake inhibition; seizure risk (especially with SSRIs, TCAs); serotonin syndrome risk; lower abuse potential but dependence occurs; CYP2D6 dependent like codeine |
| Tapentadol | 50–100 mg PO q4–6h (max 600 mg/day) | 60 min / 4–6 h | ~0.4x | Glucuronidation (no CYP involvement) | Dual mechanism: mu agonism + NE reuptake inhibition (no serotonergic activity — lower serotonin syndrome risk than tramadol); Schedule II; no CYP interactions; less GI side effects |
10 Extended-Release & Long-Acting Opioids
ER/LA opioids are reserved for patients requiring continuous around-the-clock analgesia who have already been on stable opioid therapy. The FDA REMS requires prescriber education. Never initiate in opioid-naive patients (exception: specific ER formulations at lowest dose with close monitoring).
| Drug | Formulation | Dosing Interval | Abuse-Deterrent | Key Notes |
|---|---|---|---|---|
| Morphine ER (MS Contin, Kadian) | Tablet (MS Contin); capsule with extended-release beads (Kadian) | q8–12h (MS Contin); q12–24h (Kadian) | No (standard formulations) | Reference LA opioid; convert from total daily IR morphine dose; Kadian capsules can be sprinkled on applesauce (not crushed) |
| Oxycodone ER (OxyContin) | Abuse-deterrent tablet | q12h | Yes (polyethylene oxide matrix — resists crushing, dissolving, extraction) | Abuse-deterrent reformulation (2010); must be swallowed whole; convert at 1.5:1 morphine:oxycodone ratio |
| Oxymorphone ER (Opana ER) | Crush-resistant tablet | q12h | Reformulated | 3x morphine potency PO; take on empty stomach (food increases levels 50%); original formulation withdrawn (IV abuse) |
| Hydromorphone ER (Exalgo) | Osmotic tablet (OROS) | q24h | No | OROS delivery system; must swallow whole; ghost tablet in stool (normal); 4–5x morphine potency |
| Fentanyl transdermal | Patch (12, 25, 37.5, 50, 75, 100 mcg/h) | q72h | No | Only for opioid-tolerant patients (≥60 mg oral morphine/day); 12–24h onset; depot in SC fat; fever/heat ↑ absorption (potentially fatal); conversion: morphine 60 mg/day PO ≈ fentanyl 25 mcg/h patch |
Methadone — Unique Pharmacology
Methadone is a full mu agonist with unique properties that set it apart from all other opioids:
- NMDA receptor antagonism — may be particularly effective for neuropathic pain and opioid-induced hyperalgesia
- Serotonin/NE reuptake inhibition — additional analgesic mechanism
- Variable half-life: 8–59 hours (mean ~24 hours) — steady state requires 5–7 days; risk of accumulation and delayed respiratory depression days after dose increase
- Non-linear equianalgesic conversion: Ratio to morphine is NOT fixed — increases at higher doses (at ≤90 mg morphine, ratio is 4:1; at 91–300 mg, ratio is 8:1; at >300 mg, ratio is 12:1 or higher)
- QTc prolongation: Blocks hERG potassium channels → risk of Torsades de Pointes; ECG at baseline, 30 days, annually, and after dose increase >30–40 mg/day; hold if QTc >500 ms
- Drug interactions: CYP3A4, 2B6, 2D6 substrate; many interactions (rifampin, antiretrovirals, benzodiazepines)
- No active metabolites — safe in renal failure (not dialyzable)
- Inexpensive: ~$30/month vs >$300 for many ER opioids
Levorphanol
Levorphanol is a long-acting mu agonist with NMDA antagonism, NE/5-HT reuptake inhibition, and kappa/delta agonism — similar to methadone but with more predictable pharmacokinetics (t½ 11–16 h). Dose: 2–4 mg PO q6–8h. No QTc prolongation. Useful alternative when methadone is not tolerated or ECG-contraindicated. Limited availability and prescriber familiarity.
11 Opioid Conversion & Rotation
Equianalgesic Dose Table
| Opioid | PO Dose (mg) | IV/IM/SC Dose (mg) | PO:IV Ratio | MME Factor (per mg PO) |
|---|---|---|---|---|
| Morphine | 30 | 10 | 3:1 | 1.0 |
| Oxycodone | 20 | N/A (no IV in US) | — | 1.5 |
| Hydrocodone | 30 | N/A | — | 1.0 |
| Hydromorphone | 6 | 1.5 | 4:1 | 4.0 |
| Oxymorphone | 10 | 1 | 10:1 | 3.0 |
| Codeine | 200 | 120 | 1.7:1 | 0.15 |
| Tramadol | 300 | N/A | — | 0.1 |
| Tapentadol | 75 | N/A | — | 0.4 |
| Fentanyl | N/A | 0.1 (100 mcg) | — | Patch: 25 mcg/h ≈ 60–90 mg morphine PO/day |
| Methadone | Variable (see text) | Variable | 2:1 | Variable: 4–12 depending on total daily morphine dose |
Opioid Rotation Methodology
- Calculate total 24-hour dose of current opioid (include all IR breakthrough doses)
- Convert to oral morphine equivalents using equianalgesic table
- Convert from oral morphine equivalents to new opioid using equianalgesic table
- Reduce calculated dose by 25–50% for incomplete cross-tolerance (receptor polymorphisms and differential receptor binding mean prior opioid exposure does not confer full tolerance to the new agent)
- Adjust further for clinical variables: age, renal/hepatic function, pain severity, drug interactions
- Provide IR breakthrough medication at 10–20% of total 24-hour dose of new opioid, available q1–2h PRN
- Reassess in 24–48 hours and titrate as needed
CDC Morphine Milligram Equivalent (MME) Thresholds
The 2022 CDC Clinical Practice Guideline (updated from 2016) recommends:
- Before starting opioids, optimize non-opioid therapy and use the lowest effective dose
- ≥50 MME/day: Exercise caution; reassess evidence of individual benefits and risks; co-prescribe naloxone
- ≥90 MME/day: Avoid or carefully justify; involves substantial risk of overdose and death
- Epidemiologic data: overdose risk at 50–99 MME/day is 3.7x baseline; at ≥100 MME/day, risk is 8.9x baseline
12 Opioid Risk Management
Risk Assessment Tools
| Tool | Format | Scoring | Use |
|---|---|---|---|
| ORT (Opioid Risk Tool) | 5-item self-report | 0–3 low; 4–7 moderate; ≥8 high risk | Pre-prescribing screen for aberrant behavior risk; assesses family/personal history of substance abuse, age, sexual abuse, psychological disease |
| SOAPP-R (Screener and Opioid Assessment for Patients with Pain — Revised) | 24-item self-report | ≥18 high risk (sensitivity 0.81, specificity 0.68) | Pre-prescribing; more comprehensive than ORT; validated in chronic pain populations |
| DIRE (Diagnosis, Intractability, Risk, Efficacy) | Clinician-rated, 4 domains | 7–21; higher = better candidate | Clinician assessment of suitability for long-term opioids; factors: diagnosis, intractability, psychological risk, chemical health risk, reliability, social support, efficacy |
| COMM (Current Opioid Misuse Measure) | 17-item self-report | ≥9 positive screen | Ongoing monitoring of patients already on opioids for current aberrant behavior |
Urine Drug Screening (UDS)
- Immunoassay (screening): Detects drug classes; cross-reactivity common; cannot distinguish specific opioids; confirms/denies presence of broad categories
- LC-MS/MS (confirmatory): Identifies specific drugs and metabolites; quantitative; required to resolve unexpected immunoassay results
- Frequency: Baseline before starting opioids; at least annually for stable patients; more frequently (quarterly) for higher-risk patients; random testing preferred
- Synthetic opioids NOT detected by standard immunoassay: fentanyl, methadone, buprenorphine, tramadol, tapentadol — require specific testing
- Common pitfalls: Hydromorphone is a metabolite of hydrocodone (positive for hydromorphone in patient prescribed hydrocodone is expected); oxymorphone is a metabolite of oxycodone; poppy seeds can cause morphine/codeine positive
- Expected results: Prescribed medication PRESENT; non-prescribed controlled substances ABSENT; illicit drugs ABSENT
PDMP Checking & Treatment Agreements
Prescription Drug Monitoring Programs (PDMP): Check at initiation and every 1–3 months (mandated by most states). Red flags: multiple prescribers, overlapping prescriptions, early refills, concurrent benzodiazepines. Treatment agreements (opioid contracts): Document informed consent, expectations, monitoring plan, and consequences of non-adherence. Include: single prescriber/pharmacy, UDS consent, pill counts, safe storage, naloxone provision, reasons for taper/discontinuation.
Naloxone Co-Prescribing
Co-prescribe naloxone for patients receiving ≥50 MME/day, concurrent benzodiazepines, history of overdose, history of substance use disorder, or any patient/family requesting it. Intranasal naloxone (Narcan): 4 mg/spray; may repeat in 2–3 minutes. IM auto-injector (Evzio): 2 mg; may repeat. Educate patient and household contacts on recognition of overdose (unresponsive, slow/stopped breathing, blue lips) and naloxone administration.
Opioid Taper Protocols
For chronic opioids when taper is indicated (lack of benefit, adverse effects, aberrant behavior, patient preference):
- Gradual taper: Reduce by 10% of total daily dose per month (slowest, best tolerated)
- Moderate taper: Reduce by 10–20% every 2–4 weeks
- Faster taper (when safety requires): 20–25% reduction every 1–2 weeks; closer monitoring needed
- Pause taper if withdrawal symptoms are intolerable; restart non-opioid analgesics before/during taper
- The final 20–30% of taper is the hardest — slow down further
Opioid-Induced Adverse Effects
| Effect | Mechanism | Tolerance? | Management |
|---|---|---|---|
| Constipation | Mu receptors in enteric plexus → decreased peristalsis, increased water absorption | Minimal/none | Prophylactic senna + docusate; PEG; methylnaltrexone (SC) or naloxegol (PO) for opioid-induced constipation refractory to laxatives (peripheral mu antagonists) |
| Nausea/vomiting | CTZ stimulation, vestibular sensitization, GI stasis | Yes (days to weeks) | Ondansetron, prochlorperazine, metoclopramide; usually resolves within 1 week |
| Sedation | Central mu activation | Yes (days) | Reduce dose; opioid rotation; methylphenidate (controversial) |
| Respiratory depression | Mu-2 receptors in brainstem (pre-Bötzinger complex) | Yes (parallel to analgesia) | Naloxone 0.04–0.4 mg IV q2–3 min; ventilatory support; lowest risk when titrated to effect |
| Pruritus | Central mu activation (NOT histamine for most opioids) | Yes | Opioid rotation; nalbuphine 2.5–5 mg IV; antihistamines less effective (mechanism is not histamine-mediated) |
| Endocrinopathy (OIAD) | Hypothalamic suppression of GnRH → hypogonadism; decreased ACTH, GH | No | Testosterone replacement in hypogonadal men on chronic opioids; check testosterone, DHEA-S, cortisol; dose reduction |
| Sleep-disordered breathing | Central apnea from depressed respiratory drive | Partial | Sleep study; CPAP/ASV; dose reduction |
13 Epidural Steroid Injections
Approaches
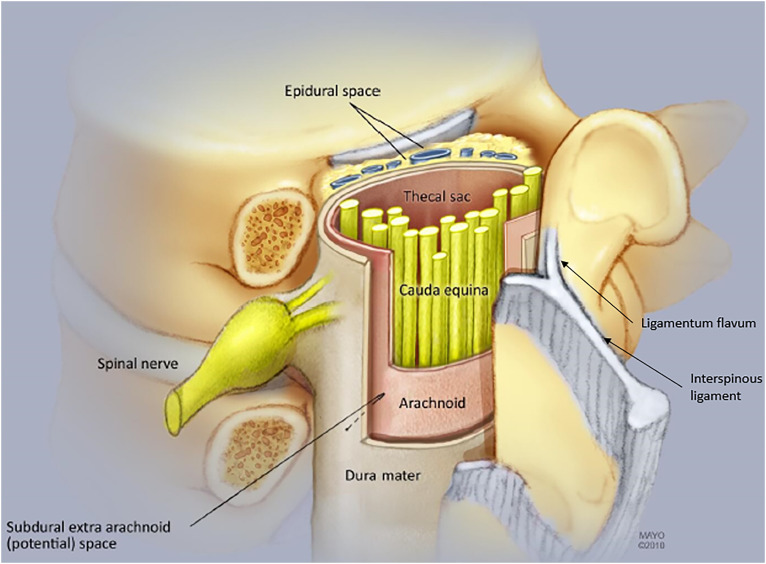
| Approach | Technique | Advantages | Disadvantages / Risks | Best For |
|---|---|---|---|---|
| Interlaminar (IL) | Midline or paramedian approach through ligamentum flavum into posterior epidural space; loss-of-resistance technique | Bilateral spread; technically simpler; larger epidural volume | No ventral spread; risk of dural puncture; steroid may not reach ventral pathology | Bilateral symptoms; central stenosis; non-focal radiculopathy |
| Transforaminal (TF) | Oblique approach into neural foramen under fluoroscopy; targets ventral epidural space near the affected nerve root | Targeted delivery; ventral spread; lower steroid volume needed; diagnostic value | Higher risk of vascular uptake (radicular artery); MUST use non-particulate steroid for cervical TF; risk of nerve injury | Unilateral radiculopathy; specific nerve root pathology; post-surgical anatomy |
| Caudal | Through sacral hiatus into epidural space; uses higher volumes (10–20 mL) | Low risk of dural puncture; easy in post-surgical patients; no midline scarring concern | Less targeted; requires higher volume; may not reach upper lumbar levels | Multi-level lumbar pathology; post-laminectomy; S1 radiculopathy; patients with difficult anatomy |
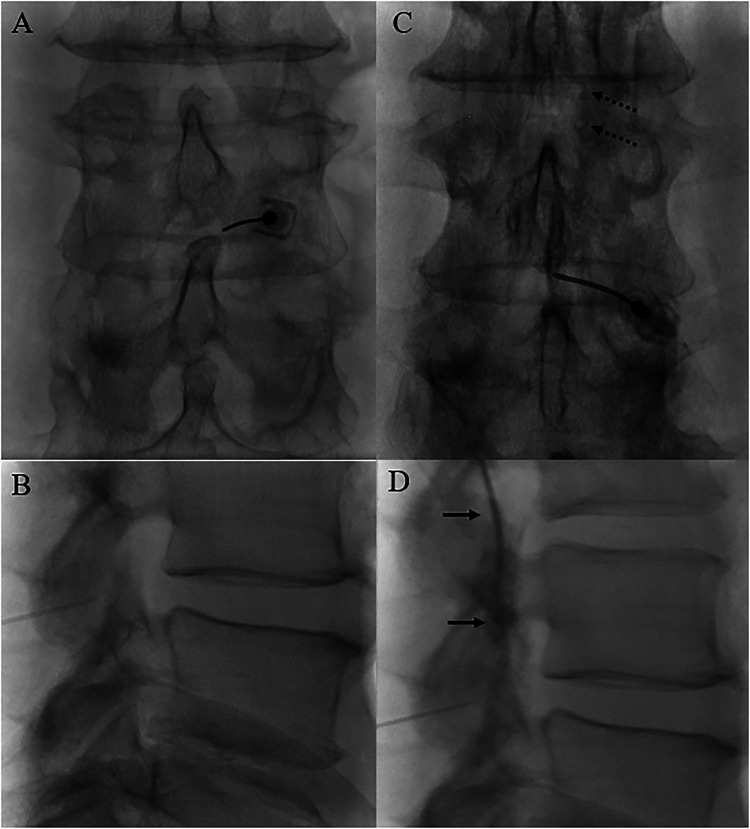
Evidence by Indication
- Lumbar radiculopathy (disc herniation): Best evidence; NNT ~3 for short-term relief; TF approach preferred for focal radiculopathy; greatest benefit within 3 months of symptom onset
- Lumbar spinal stenosis: Moderate evidence; IL approach with large volume for bilateral spread; benefit is modest and short-lived; SPORT and MILD trials showed limited long-term benefit over conservative care
- Axial low back pain (without radiculopathy): Weakest evidence; not recommended as sole intervention; pain generators are likely facet joints, SI joint, or discogenic
- Cervical radiculopathy: Good evidence for IL and TF approaches; TF MUST use non-particulate dexamethasone; IL approach preferred when possible to minimize vascular risk
- Dural puncture (1–5%): Post-dural puncture headache (PDPH); positional (worse upright, better supine); treat with caffeine, hydration, epidural blood patch if persistent >24–48 hours
- Epidural hematoma (rare, ~1:150,000): Risk increased with anticoagulation; ASRA guidelines for anticoagulant management; emergency MRI and surgical decompression within 8–12 hours if motor deficit
- Epidural abscess: Risk factors include immunosuppression, diabetes, recent bacteremia; presents with fever, back pain, progressive neurologic deficit; emergency MRI; IV antibiotics + surgical decompression
- Vascular injection (TF approach): Radicular artery penetration with particulate steroid → spinal cord infarction (anterior spinal artery syndrome); use live fluoroscopy with contrast to detect vascular uptake
- Steroid side effects: Facial flushing, hyperglycemia (1–2 weeks), HPA suppression, menstrual irregularity, insomnia, weight gain
14 Facet Joint & Medial Branch Interventions
Facet-Mediated Pain Referral Patterns
| Level | Referral Pattern |
|---|---|
| C2–3 | Occipital headache, upper cervical pain |
| C3–4 | Posterolateral neck, does not radiate past shoulder |
| C5–6 | Shoulder, suprascapular region (mimics rotator cuff) |
| C6–7 | Interscapular region |
| L1–2 | Groin, lateral hip |
| L3–4 | Posterior thigh (mimics radiculopathy) |
| L4–5 | Gluteal, posterior thigh to knee |
| L5–S1 | Gluteal, posterior thigh, may reach calf (does NOT go below knee typically) |
Diagnostic Medial Branch Blocks
Each facet joint is innervated by the medial branch of the dorsal ramus at the same level and one level above (e.g., L4–5 facet requires L3 and L4 medial branch blocks). The dual comparative block paradigm is the gold standard: two separate diagnostic blocks with different local anesthetics (e.g., lidocaine then bupivacaine) on different days. A block is positive if the patient reports ≥80% pain relief with duration consistent with the agent used. Dual blocks reduce false-positive rates from ~40% (single block) to ~15%.
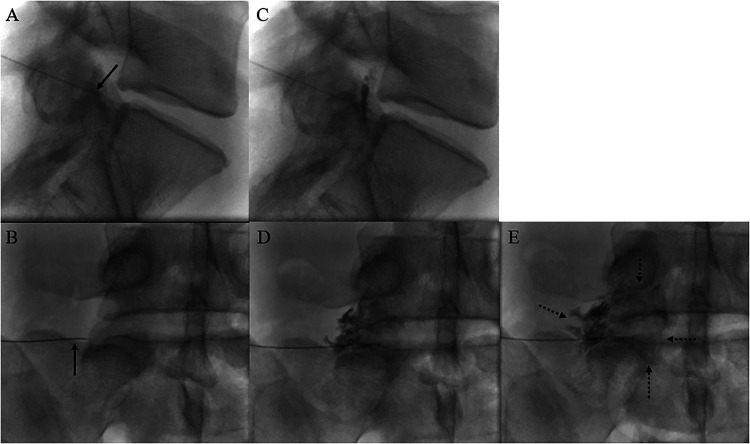
Radiofrequency Ablation (RFA)
Following positive dual diagnostic blocks, conventional radiofrequency ablation denervates the medial branch nerve. Technique: 18–22 gauge RF cannula placed parallel to the nerve course; sensory stimulation at 50 Hz (concordant pain at ≤0.5 V); motor stimulation at 2 Hz (no multifidus contraction below 2.0 V ensures safe distance from ventral ramus). Lesion parameters: 80°C for 60–90 seconds per lesion site. Typically 2–3 lesions per level for adequate coverage.
- Success rate: 50–80% of patients achieve ≥50% pain relief after properly selected dual blocks
- Duration of relief: 6–12 months (nerves regenerate); can be repeated
- Cervical RFA: Similar efficacy; higher technical precision required due to proximity to vertebral artery and spinal cord
- Post-procedure: Soreness for 1–2 weeks; neuritis (neuropathic pain at ablation site) in 5–10%; transient worsening before improvement
- Contraindications: Active infection, coagulopathy, negative diagnostic blocks, pregnancy, implanted cardiac device (relative)
15 Sacroiliac Joint Interventions
Clinical Diagnosis
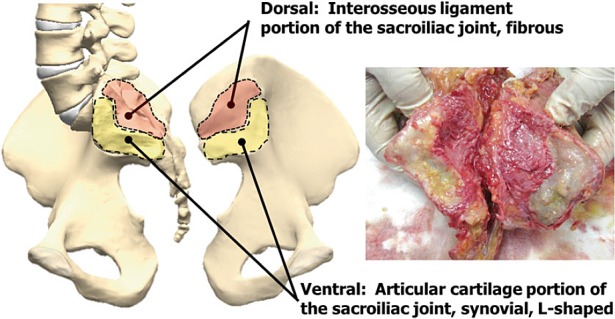
SI joint pain accounts for 15–30% of chronic low back pain. Pain is typically unilateral, below L5, maximal at the posterior superior iliac spine (PSIS), and may radiate to the buttock, groin, or posterior thigh (rarely below the knee). At least 3 of 5 provocation tests must be positive for clinical diagnosis:
| Test | Technique | Positive Finding |
|---|---|---|
| FABER (Patrick) test | Flex, abduct, externally rotate hip; press down on contralateral ASIS and ipsilateral knee | Pain at ipsilateral SI joint |
| Gaenslen test | Patient supine at table edge; hyperextend one hip off table while flexing contralateral hip to chest | Pain at SI joint of hyperextended side |
| Compression test | Patient side-lying; downward pressure on iliac crest | Pain at SI joint |
| Distraction (gapping) test | Patient supine; outward pressure on bilateral ASIS | Pain at SI joint |
| Thigh thrust (posterior shear) | Patient supine; hip flexed 90°; axial force through femur toward table | Pain at SI joint |
SI Joint Injection
Fluoroscopically guided intra-articular injection with local anesthetic ± corticosteroid. Diagnostic value: ≥75% pain relief confirms SI joint as pain generator. Ultrasound-guided injection is an alternative. Therapeutic relief with corticosteroid is typically short-lived (weeks to months).
Lateral Branch Blocks & RFA
The SI joint is innervated by the lateral branches of the S1–S3 dorsal rami and the L5 dorsal ramus. Lateral branch radiofrequency ablation (using conventional, cooled, or pulsed RF techniques) can provide 6–12 months of relief. Cooled RF creates larger lesions (spherical rather than cylindrical) which improves capture of the variable lateral branch anatomy.
SI Joint Fusion
Minimally invasive SI joint fusion (e.g., iFuse implant system) is indicated for SI joint pain that has failed conservative management and at least two positive diagnostic injections. The INSITE and iMIA RCTs demonstrated superiority over non-surgical management at 6 months. Three triangular titanium implants are placed across the SI joint under fluoroscopic guidance.
16 Joint & Soft Tissue Injections
Major Joint Injections
| Joint | Approach | Agent | Key Considerations |
|---|---|---|---|
| Knee | Suprapatellar (preferred — larger target, allows aspiration), inferolateral (seated, knee flexed 90°, lateral to patellar tendon), medial midpatellar | Triamcinolone 40 mg + lidocaine 5 mL; or hyaluronic acid (viscosupplementation) | Most common joint injected; limit to 3–4 corticosteroid injections/year; evidence of long-term cartilage loss with repeated steroid (NEJM 2017 trial) |
| Shoulder — Subacromial | Posterior approach, 2 cm inferior and medial to posterolateral acromion; needle directed anterosuperiorly | Triamcinolone 40 mg + lidocaine 4–5 mL | Impingement, bursitis, rotator cuff tendinopathy; landmark-guided often sufficient; avoid tendon injection |
| Shoulder — Glenohumeral | Posterior approach (preferred): 2 cm inferior and medial to posterolateral acromion, directed toward coracoid process; anterior approach also used | Triamcinolone 40 mg + lidocaine 3–5 mL | OA, adhesive capsulitis, inflammatory arthritis; accuracy improved with US guidance (68% landmark vs 93% US-guided) |
| Shoulder — AC joint | Direct superior approach into the AC joint space | Betamethasone 3 mg or triamcinolone 10–20 mg + lidocaine 0.5–1 mL | Small joint; small volume; palpate joint with cross-body adduction to identify |
| Hip | Anterior approach under fluoroscopy or US guidance (femoral head-neck junction) | Triamcinolone 40–80 mg + lidocaine/bupivacaine + contrast | Always use image guidance (deep joint, proximity to femoral neurovascular bundle); OA, labral tears, AVN |
Trigger Point Injections & Dry Needling
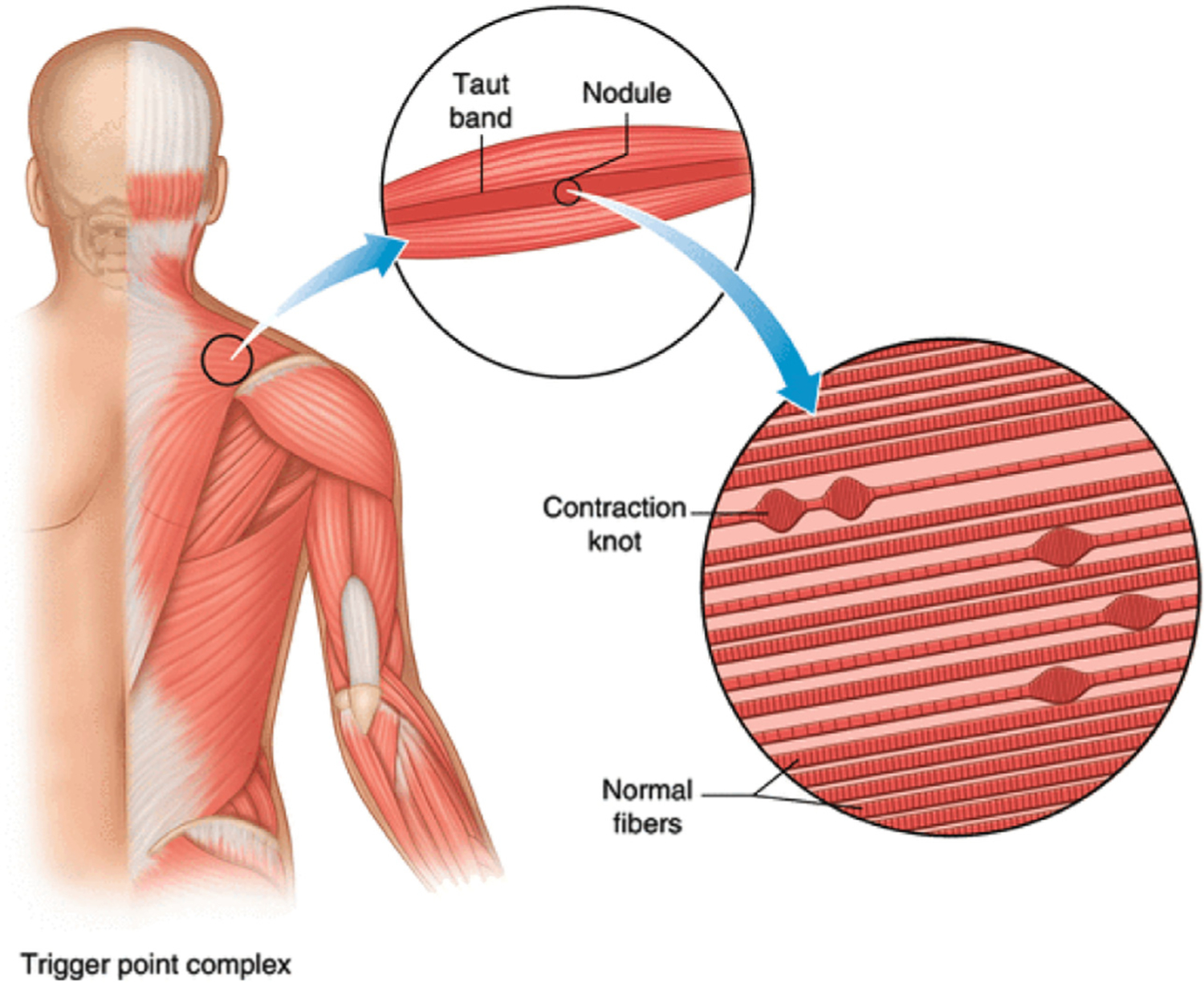
Trigger points are hyperirritable taut bands within skeletal muscle that produce local and referred pain. Injection technique: 25–27 gauge needle inserted into the taut band; elicit local twitch response; inject 0.5–1 mL 1% lidocaine or 0.25% bupivacaine. Dry needling uses acupuncture needles without injectate to disrupt the trigger point. Systematic reviews suggest equivalent efficacy between wet and dry needling; the mechanical disruption of the taut band is the key therapeutic element.
Viscosupplementation
Hyaluronic acid (HA) injections: Available as single-injection (Synvisc-One, Gel-One) or 3–5 weekly injection series (Euflexxa, Supartz, Hyalgan). Mechanism: restores synovial fluid viscoelasticity, anti-inflammatory and analgesic properties. Evidence is mixed — AAOS gives a “limited” recommendation; most benefit in mild-moderate knee OA (KL grade 2–3). Contraindicated in avian-derived products if egg allergy.
Platelet-Rich Plasma (PRP)
PRP is autologous blood concentrated to 3–5x baseline platelet count, providing supraphysiologic growth factors (PDGF, TGF-beta, VEGF). Evidence is strongest for knee OA (meta-analyses show superiority to HA at 12 months) and lateral epicondylitis. Weaker evidence for rotator cuff tendinopathy, plantar fasciitis, and Achilles tendinopathy. Not covered by most insurers. Leukocyte-poor PRP preferred for intra-articular use; leukocyte-rich PRP for tendon/soft tissue.
17 Neuromodulation
Spinal Cord Stimulation (SCS)
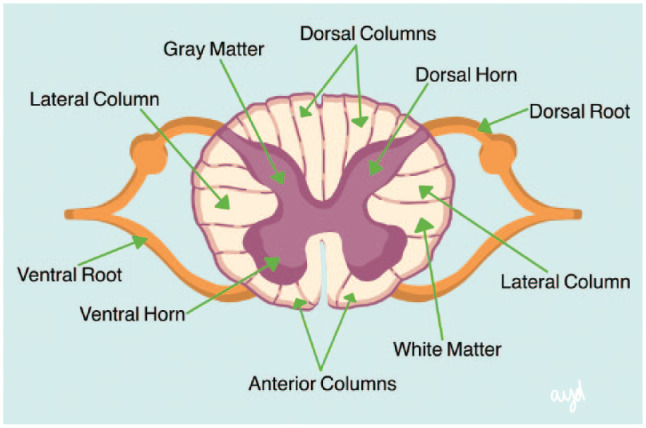
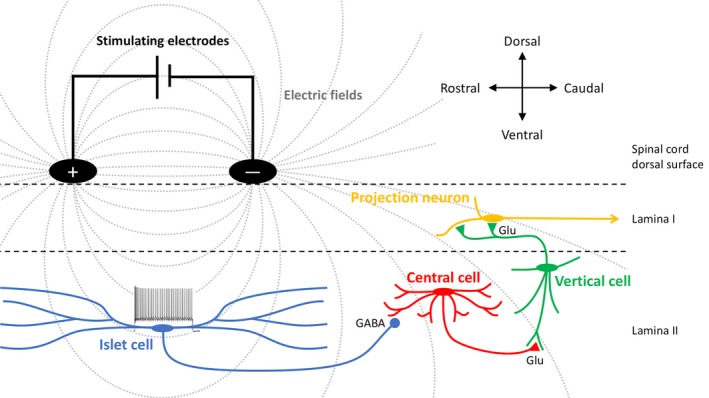
SCS involves placement of epidural electrodes over the dorsal columns to modulate pain signaling. The original theoretical basis is the gate control theory — electrical stimulation of large-diameter dorsal column fibers “closes the gate” on nociceptive input. Modern mechanisms also involve supraspinal modulation, GABA release in the dorsal horn, and activation of descending inhibition.
| Modality | Frequency | Mechanism | Paresthesia | Key Features |
|---|---|---|---|---|
| Conventional (tonic) | 40–100 Hz | Gate control; dorsal column activation | Yes (paresthesia must overlap pain area) | Oldest modality; requires paresthesia mapping during trial; positional variability |
| High-frequency (HF10) | 10,000 Hz (10 kHz) | Uncertain; may involve glial modulation, wide dynamic range neuron inhibition | No (paresthesia-free) | SENZA-RCT: superior to conventional at 24 months; no paresthesia mapping needed; simpler programming |
| Burst | 500 Hz bursts at 40 Hz interburst frequency | Modulates both lateral (sensory-discriminative) and medial (affective) pain pathways | No (paresthesia-free) | SUNBURST trial: non-inferior to tonic; may preferentially target affective pain component |
| DRG stimulation | 20 Hz | Direct stimulation of dorsal root ganglion; modulates primary afferent signaling | Minimal | ACCURATE study: superior to conventional SCS for CRPS and focal neuropathy; precise dermatomal targeting; lead placed in epidural space at target foramen |
SCS Trial & Implant Process
Trial phase (4–7 days): Percutaneous leads placed under fluoroscopy; external generator; patient assesses pain relief in daily activities. Trial is successful if ≥50% pain relief with functional improvement and reduced medication use. If successful, proceed to permanent implant with IPG (implantable pulse generator) placed in subcutaneous pocket (buttock or abdomen).
- Failed back surgery syndrome (FBSS): Strongest evidence (PROCESS trial: SCS + CMM superior to CMM alone)
- Complex regional pain syndrome (CRPS): Strong evidence (Kemler RCT); DRG stimulation preferred
- Painful diabetic peripheral neuropathy: SENZA-PDN trial; HF10 SCS demonstrated superiority to CMM
- Chronic angina refractory to medical therapy: European evidence; less used in US
- Peripheral vascular disease / critical limb ischemia: Improves microcirculation; European evidence
Intrathecal Drug Delivery (IDDS)
An intrathecal pump delivers medication directly into the CSF, achieving effective concentrations at 1/300th of the systemic dose. Reduces systemic side effects. Agents: morphine (first-line, FDA-approved), ziconotide (N-type Ca2+ channel blocker; FDA-approved; non-opioid; NNT 2.6 for cancer pain; side effects: dizziness, nausea, cognitive changes, psychiatric symptoms), baclofen (for spasticity — MS, SCI, CP). The Polyanalgesic Consensus Conference (PACC) algorithm guides intrathecal drug selection. Pump refills every 1–6 months; battery replacement surgery every 5–7 years.
18 Advanced & Emerging Procedures
Sympathetic Nerve Blocks
| Block | Target | Technique | Indications | Risks |
|---|---|---|---|---|
| Stellate ganglion block | Cervicothoracic sympathetic chain (C6–T1) | Anterolateral approach at C6 (Chassaignac tubercle) under fluoroscopy/US; 5–10 mL local anesthetic | Upper extremity CRPS, phantom limb pain, refractory angina, PTSD (investigational), hyperhidrosis | Horner syndrome (expected — confirms successful block), recurrent laryngeal nerve palsy, vertebral artery injection (seizure), pneumothorax, esophageal perforation |
| Lumbar sympathetic block | L2–L4 sympathetic chain (anterolateral to vertebral body) | Fluoroscopy-guided; needle at anterolateral L2 or L3 body; 15–20 mL local anesthetic | Lower extremity CRPS, peripheral vascular disease, phantom limb pain, hyperhidrosis | Intravascular injection, ureteral injury, genitofemoral neuralgia, hypotension |
| Celiac plexus block/neurolysis | Celiac plexus at T12–L1 (anterior to aorta) | Posterior bilateral approach or anterior EUS-guided; diagnostic block with local anesthetic; neurolysis with phenol 6–10% or absolute alcohol | Pancreatic cancer pain (NNT 1.6 for neurolysis), chronic pancreatitis, upper abdominal visceral malignancy | Hypotension (sympathectomy), diarrhea (unopposed parasympathetic), pneumothorax, aortic/IVC puncture, paraplegia (artery of Adamkiewicz) |
| Superior hypogastric plexus block | Superior hypogastric plexus at L5–S1 (anterior to vertebral body) | Bilateral fluoroscopy-guided; 6–8 mL local anesthetic per side | Pelvic visceral pain: endometriosis, pelvic malignancy, interstitial cystitis, chronic pelvic pain | Intravascular injection, discitis, ureteral injury |
| Ganglion impar block | Ganglion impar (unpaired ganglion at sacrococcygeal junction) | Trans-sacrococcygeal approach under fluoroscopy; 4–6 mL | Perineal pain: coccydynia, vulvodynia, rectal cancer pain | Rectal perforation, infection |
Vertebral Augmentation
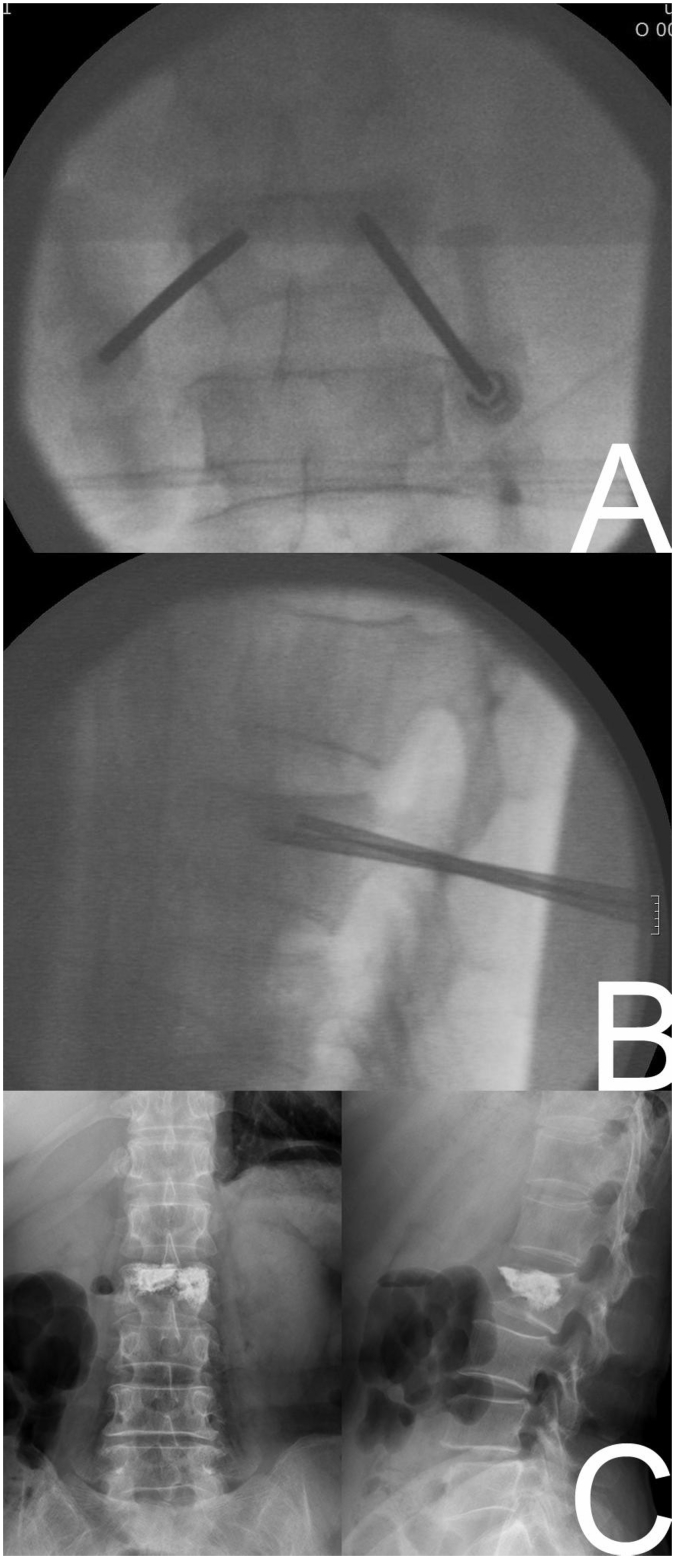
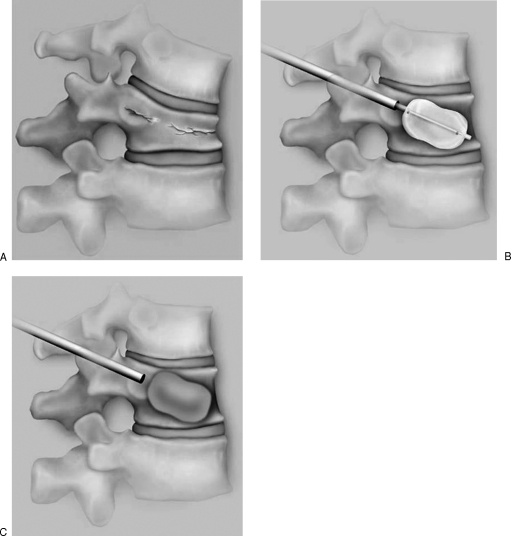
Vertebroplasty: Percutaneous injection of PMMA bone cement into the fractured vertebral body under fluoroscopy. Kyphoplasty: Balloon tamp inflated first to create a cavity and partially restore height, then PMMA injected. The VERTOS II trial showed vertebroplasty superior to sham for acute (<6 weeks) painful VCFs, while INVEST showed no difference (sham-controlled). FREE trial showed kyphoplasty superior to conservative care. Current consensus: vertebral augmentation is appropriate for painful osteoporotic VCFs refractory to 2–4 weeks of conservative management.
MILD Procedure (Minimally Invasive Lumbar Decompression)
Percutaneous removal of hypertrophied ligamentum flavum and small bone to create space in the lumbar spinal canal for patients with lumbar spinal stenosis and neurogenic claudication. Performed through a 5.1 mm portal under fluoroscopy. The MILD RCT demonstrated statistically significant improvement in ODI and VAS at 12 months vs epidural steroids.
Peripheral Nerve Stimulation (PNS)
Percutaneous placement of stimulating electrodes adjacent to peripheral nerves for localized neuropathic pain. Targets include occipital nerves (chronic migraine), suprascapular nerve (shoulder pain), medial branch of genicular nerves (knee OA), and tibial nerve (foot pain). Sprint PNS system allows 60-day percutaneous stimulation without permanent implant.
Regenerative Medicine
Bone marrow aspirate concentrate (BMAC) and adipose-derived stem cells are under investigation for OA, degenerative disc disease, and tendinopathy. FDA regulations classify minimally manipulated autologous tissue as exempt from premarket approval but restrict “more than minimally manipulated” products. Evidence remains limited; no FDA-approved stem cell therapy for musculoskeletal pain currently exists.
Intradiscal Procedures
Intradiscal electrothermal therapy (IDET): Thermal catheter placed in the annulus fibrosus; heats collagen to 90°C to denature annular nociceptors and seal annular fissures. Evidence is mixed; largely fallen out of favor. Biacuplasty: Bipolar radiofrequency heating of the posterior annulus via two cooled probes; IDEAL study showed modest benefit at 6 months. Intradiscal biologics: PRP and mesenchymal stem cell injections into degenerative discs are investigational; small RCTs show signal of benefit but lack Level I evidence.
19 Low Back Pain
Epidemiology & Natural History
Low back pain (LBP) is the leading cause of disability worldwide (Global Burden of Disease). Lifetime prevalence 80%. Most acute LBP resolves within 6 weeks regardless of treatment. However, 10–15% develop chronic LBP (>12 weeks). Risk factors for chronification: psychosocial (catastrophizing, fear-avoidance, depression, job dissatisfaction), yellow flags, workers’ compensation involvement, baseline disability level.
Red Flags
- Cauda equina syndrome: Saddle anesthesia, urinary retention/incontinence, fecal incontinence, bilateral leg weakness → emergent MRI and surgical referral
- Cancer: History of malignancy, unexplained weight loss, age >50, pain at rest/night, failure to improve after 4–6 weeks
- Infection: Fever, IV drug use, immunosuppression, recent spinal procedure, UTI/skin infection
- Fracture: Significant trauma, osteoporosis, chronic corticosteroid use, age >70
- Vascular: Pulsatile abdominal mass (AAA), tearing/ripping pain
Imaging Criteria
No imaging within the first 6 weeks of acute LBP in the absence of red flags (ACP/ACR Choosing Wisely). Imaging in the first 6 weeks is appropriate only with: progressive neurologic deficit, cauda equina symptoms, suspected cancer/infection/fracture, or failure to improve after adequate conservative trial. MRI is modality of choice; CT myelography if MRI contraindicated.
Disc Herniation — Nerve Root Correlation
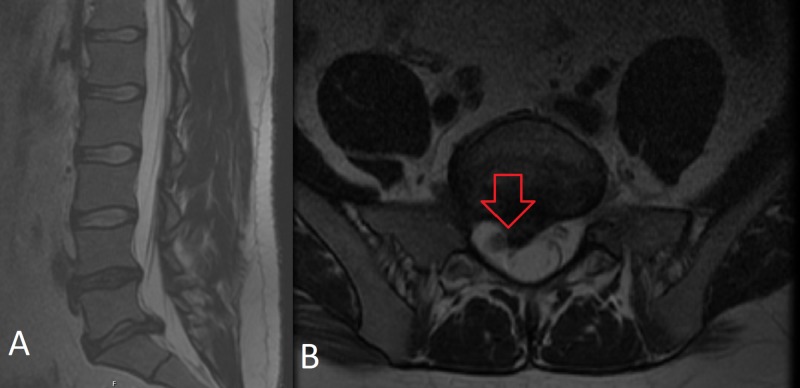
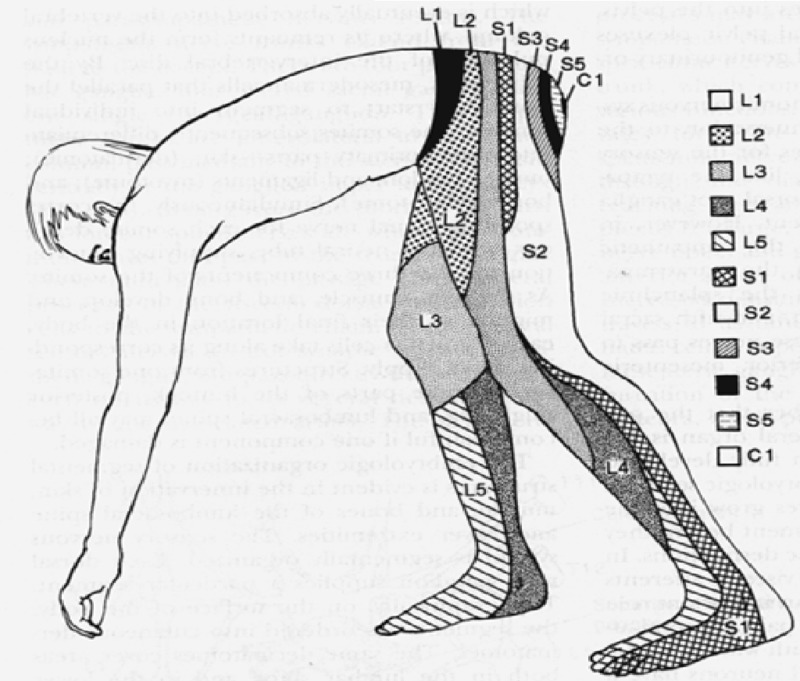
| Disc Level | Root Affected | Motor Deficit | Sensory Deficit | Reflex Loss | Exam Finding |
|---|---|---|---|---|---|
| L1–2 | L2 | Hip flexion (iliopsoas) | Anterior thigh | None reliably | Reverse SLR positive |
| L2–3 | L3 | Knee extension (quadriceps) | Anterior thigh, medial knee | Patellar (L3-4) | Reverse SLR positive |
| L3–4 | L4 | Knee extension, ankle dorsiflexion (tibialis anterior) | Medial leg, medial malleolus | Patellar | SLR may be positive |
| L4–5 | L5 | Great toe extension (EHL), hip abduction, ankle dorsiflexion | Lateral leg, dorsum of foot, 1st web space | None (medial hamstring) | SLR positive; foot drop |
| L5–S1 | S1 | Ankle plantarflexion (gastrocnemius), hip extension, toe walking | Posterior leg, lateral foot, sole | Achilles | SLR positive; cannot toe walk |
Pain Generators in Chronic LBP
Discogenic (39%): Internal disc disruption; pain worse with flexion, sitting; diagnosis via provocation discography (controversial). Facet joint (15–30%): Pain with extension, rotation; referral pattern follows affected level; diagnosed by medial branch blocks. Sacroiliac joint (15–30%): See Section 15. Myofascial: Trigger points in paraspinals, quadratus lumborum, piriformis. Failed back surgery syndrome (FBSS): Persistent or recurrent pain after lumbar spine surgery; multifactorial (recurrent herniation, epidural fibrosis, foraminal stenosis, adjacent segment disease, myofascial, psychological); SCS is first-line interventional option (PROCESS trial).
Treatment Algorithm for Chronic Low Back Pain
- Active self-management: Patient education, reassurance, remain active, physical therapy (core stabilization, McKenzie method), weight management, yoga/tai chi (ACP 2017 guideline first-line)
- Non-pharmacologic adjuncts: CBT for pain catastrophizing/fear-avoidance, mindfulness-based stress reduction (MBSR), spinal manipulation, acupuncture, massage therapy
- Pharmacologic first-line: NSAIDs (ibuprofen 400–600 mg TID or naproxen 500 mg BID × 2–4 weeks); duloxetine if NSAID-contraindicated or neuropathic component; muscle relaxants short-term for acute flares only
- Interventional (targeted): Based on identified pain generator — ESI for radiculopathy, MBB/RFA for facet pain, SI joint injection for SI joint pain; diagnostic blocks must precede ablative procedures
- Advanced: SCS for FBSS/refractory radiculopathy; intrathecal pump for cancer-related or refractory non-cancer pain; surgical referral for progressive neurologic deficit or structural pathology amenable to correction
20 Cervical & Headache Pain
Cervicogenic Headache
Cervicogenic headache (CGH) originates from cervical structures and is referred to the head. The trigeminocervical nucleus (convergence of trigeminal afferents with C1–C3 dorsal horn neurons) is the anatomic basis. Most common sources: C2–3 facet joint (innervated by the third occipital nerve, the superficial medial branch of C3 dorsal ramus), atlantoaxial joint, and upper cervical myofascial structures. Diagnostic criteria (ICHD-3): unilateral headache without sideshift, provoked by neck movement or sustained posture, reduced ROM, responds to diagnostic cervical nerve block.
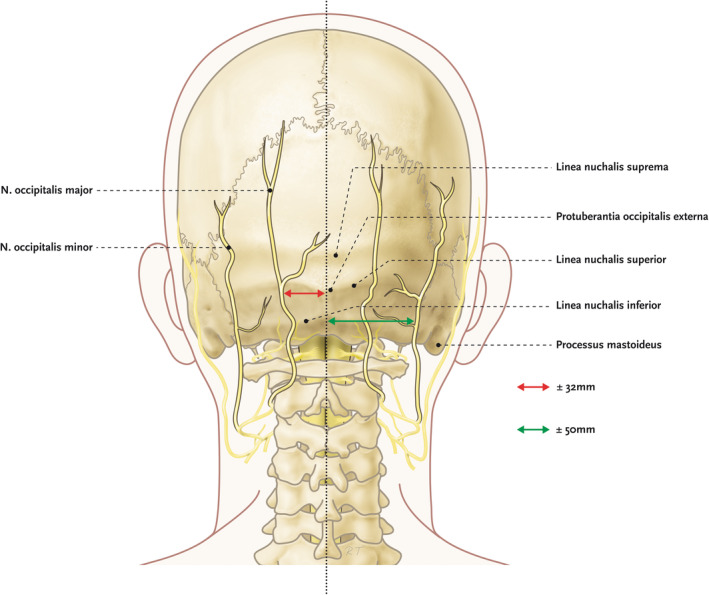
Occipital Neuralgia
Paroxysmal shooting or stabbing pain in the distribution of the greater occipital nerve (GON) (C2 dorsal ramus — medial branch) or lesser occipital nerve (LON) (C2–C3 ventral rami). Tender over the nerve at the nuchal line, 2–3 cm lateral to the occipital protuberance. Treatment: diagnostic/therapeutic GON block (2–3 mL 0.5% bupivacaine + triamcinolone 20–40 mg); if temporary relief, consider pulsed RF of GON or occipital nerve stimulation for refractory cases.
Cervical Radiculopathy
Spurling test: Cervical extension + lateral flexion + axial compression to affected side reproduces radicular arm pain (sensitivity 50%, specificity 86%). Most common levels: C5–6 (C6 root, 25%), C6–7 (C7 root, 60%). Natural history is generally favorable; 75–90% improve with conservative management. Cervical ESI (interlaminar preferred; TF with non-particulate dexamethasone ONLY) for persistent radicular pain. Surgical indications: progressive motor deficit, myelopathy, failure of 6–12 weeks of conservative treatment with persistent radicular symptoms.
Cervical Myelopathy
Compression of the spinal cord by degenerative cervical spondylosis — the most common cause of spinal cord dysfunction in adults >55. Clinical features: gait abnormality (broad-based, spastic), hand clumsiness (difficulty with buttons, handwriting — “myelopathy hand”), Hoffman sign (flicking the middle finger DIP produces thumb/index flexion), Lhermitte sign (electric shock sensation down the spine/limbs with neck flexion), hyperreflexia, Babinski sign, clonus. MRI shows cord compression with possible T2 signal change (myelomalacia). Treatment is primarily surgical decompression (anterior or posterior approach) — this is NOT an interventional pain condition; referral to spine surgery is mandatory for progressive myelopathy.
21 Complex Regional Pain Syndrome
Budapest Clinical Diagnostic Criteria (2003, IASP-adopted 2012)
Must meet ALL four of the following:
- Continuing pain disproportionate to any inciting event
- Must report at least one symptom in 3 of 4 categories:
- Sensory: Hyperesthesia and/or allodynia
- Vasomotor: Temperature asymmetry and/or skin color changes and/or skin color asymmetry
- Sudomotor/Edema: Edema and/or sweating changes and/or sweating asymmetry
- Motor/Trophic: Decreased ROM and/or motor dysfunction (weakness, tremor, dystonia) and/or trophic changes (hair, nail, skin)
- Must display at least one sign at examination in 2 or more categories:
- Sensory: Hyperalgesia to pinprick and/or allodynia to light touch/pressure/joint movement
- Vasomotor: Temperature asymmetry (>1°C) and/or skin color changes/asymmetry
- Sudomotor/Edema: Edema and/or sweating changes/asymmetry
- Motor/Trophic: Decreased ROM and/or motor dysfunction and/or trophic changes
- No other diagnosis better explains the signs and symptoms
Type I: No identifiable nerve lesion (formerly reflex sympathetic dystrophy). Type II: Identifiable nerve lesion present (formerly causalgia).
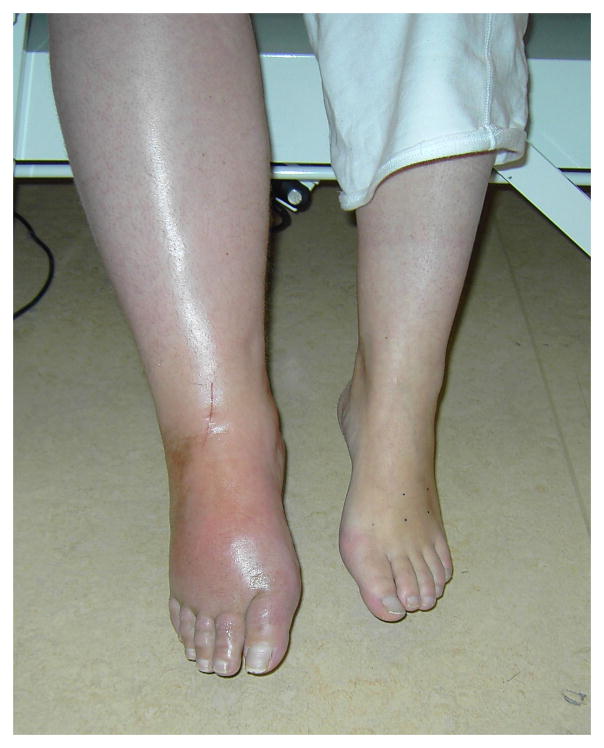
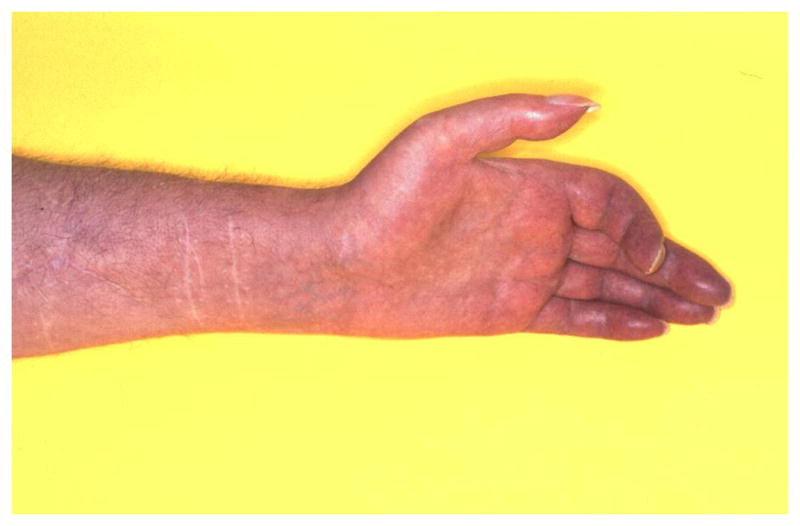
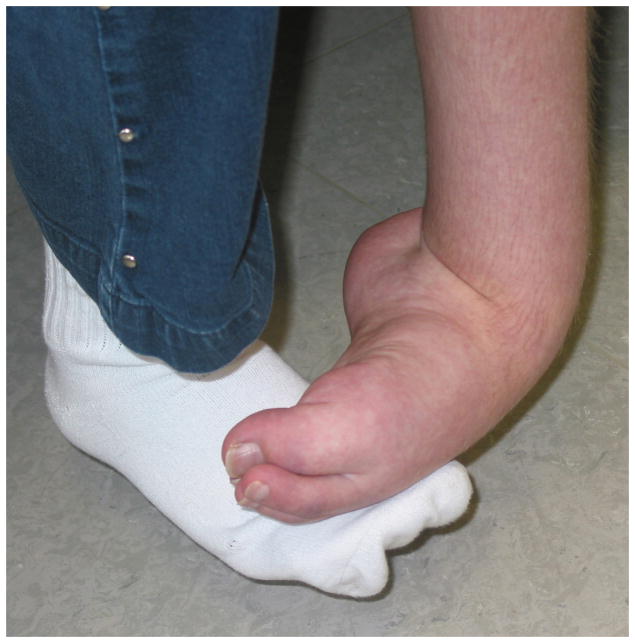
Diagnostic Studies
Three-phase bone scan: Increased uptake in all three phases (flow, blood pool, delayed) on the affected side. Sensitivity is highest in the first 3–6 months. Thermography: >1°C asymmetry. Quantitative sudomotor axon reflex testing (QSART): asymmetric sweat output. Plain radiographs: patchy osteoporosis (Sudeck atrophy) in later stages.
Treatment Ladder
| Stage | Interventions | Details |
|---|---|---|
| First-line | Physical/occupational therapy, mirror therapy, graded motor imagery (GMI) | PT is the cornerstone; active ROM, desensitization, edema management; mirror therapy (visual feedback of unaffected limb); GMI sequence: laterality recognition → imagined movements → mirror therapy |
| Pharmacologic | Neuropathic pain agents, corticosteroids, bisphosphonates | Gabapentin/pregabalin, duloxetine, nortriptyline; early corticosteroid burst (prednisone 30 mg/day × 2–4 weeks in acute CRPS); IV bisphosphonates (alendronate, pamidronate) reduce bone pain |
| Interventional | Sympathetic nerve blocks, SCS/DRG stimulation | Stellate ganglion block (upper limb) or lumbar sympathetic block (lower limb); series of 3–6 blocks; if response to sympathetic blocks → sympathetically maintained pain (SMP); SCS or DRG stimulation for refractory cases |
| Advanced | Ketamine infusion, intrathecal drug delivery, amputation (last resort) | Sub-anesthetic ketamine infusion (NMDA antagonism addresses central sensitization); intrathecal baclofen for CRPS dystonia; amputation controversial and may trigger CRPS in residual limb |
22 Cancer Pain
WHO Analgesic Ladder (1986, Modified)
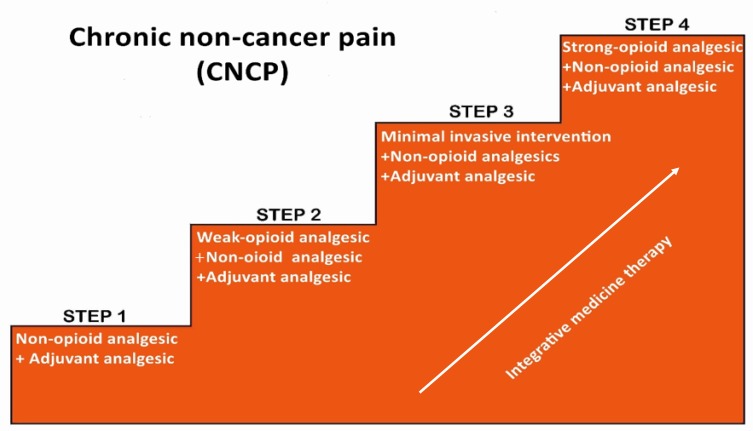
| Step | Pain Severity | Agents | Adjuvants |
|---|---|---|---|
| Step 1 | Mild (NRS 1–3) | Non-opioids: acetaminophen, NSAIDs | ± adjuvant (gabapentinoids, antidepressants, corticosteroids) |
| Step 2 | Moderate (NRS 4–6) | Weak opioids: tramadol, codeine, hydrocodone low-dose; OR low-dose strong opioid | ± non-opioids ± adjuvant |
| Step 3 | Severe (NRS 7–10) | Strong opioids: morphine, oxycodone, hydromorphone, fentanyl, methadone | ± non-opioids ± adjuvant |
| Step 4 (modified) | Refractory | Interventional: neuraxial analgesia, nerve blocks, neurolysis, SCS, intrathecal pumps | All appropriate adjuvants |
Breakthrough Cancer Pain (BTcP)
Dose: 10–20% of total 24-hour opioid dose as IR opioid q1–2h PRN. Available q1h for oral, q15–30 min for parenteral. Rapid-onset fentanyl products (TIRF): transmucosal fentanyl citrate (Actiq), fentanyl buccal tablet (Fentora), fentanyl nasal spray (Lazanda) — all restricted to opioid-tolerant patients (≥60 mg oral morphine/day) via TIRF REMS. If >3–4 breakthrough doses per day, increase the around-the-clock dose by 25–50%.
Opioid Rotation for Cancer Pain
Indications: uncontrolled pain despite dose escalation, intolerable side effects (especially cognitive changes, myoclonus from morphine metabolites), suspected OIH, end-organ changes (renal failure requiring switch from morphine). Rotate using equianalgesic table with 25–50% dose reduction for incomplete cross-tolerance; less reduction (25%) for poorly controlled pain.
Interventional Approaches for Cancer Pain
- Celiac plexus neurolysis: Pancreatic cancer pain; superior to medical management (NNT 1.6); perform early when pain is difficult to control; see Section 18
- Intrathecal drug delivery: Randomized evidence (Smith 2002) showed improved pain control, reduced toxicity, and improved survival with intrathecal morphine vs comprehensive medical management
- Palliative radiation: Single fraction (8 Gy) for painful bone metastases; response rate 60–70%; stereotactic body radiation for oligometastases
Bone Metastases Management
Bisphosphonates (zoledronic acid 4 mg IV q3–4 weeks) and denosumab (120 mg SC monthly) reduce skeletal-related events. Radionuclides: Strontium-89, samarium-153, radium-223 (for castration-resistant prostate cancer with bone metastases — improved survival). Radiation: single-fraction 8 Gy equivalent to multi-fraction for pain palliation (meta-analyses). Corticosteroids (dexamethasone 4–8 mg daily) for bone pain with periosteal stretching or perineural edema.
Cancer Pain Emergencies
| Emergency | Presentation | Immediate Management |
|---|---|---|
| Spinal cord compression | Back pain (90%), progressive weakness, sensory level, bowel/bladder dysfunction | Dexamethasone 10–16 mg IV bolus then 4 mg q6h; emergent MRI entire spine; radiation ± surgical decompression within 24–48 hours |
| Pathologic fracture | Acute severe bone pain at metastatic site; deformity; inability to bear weight | Immediate imaging; orthopedic consultation for stabilization; IV opioids for acute pain; radiation after fixation |
| Acute pain crisis | Sudden severe escalation of pain (NRS 8–10); often with visceral or neuropathic features | Rapid opioid titration (IV morphine 2–4 mg q15 min until controlled); identify cause (obstruction, hemorrhage, fracture, infection); consider ketamine adjunct for refractory pain |
| Malignant bowel obstruction | Nausea/vomiting, colicky abdominal pain, distension | Octreotide 100–600 mcg/day SC to reduce secretions; dexamethasone 8–16 mg IV; hyoscine butylbromide for colic; opioids for continuous pain; nasogastric decompression or venting gastrostomy if refractory; surgery only if single point of obstruction with good functional status |
23 Fibromyalgia & Central Sensitization
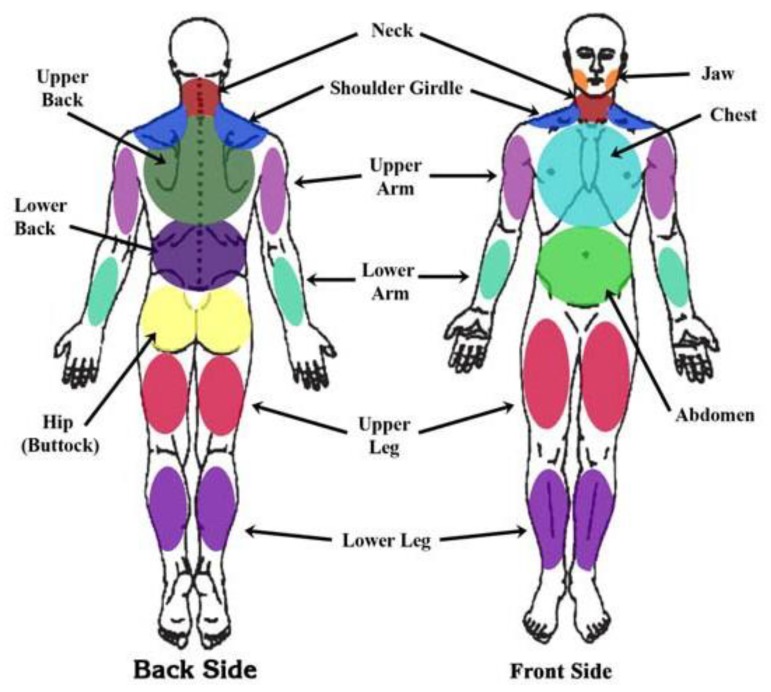
2016 ACR Revised Diagnostic Criteria
- Widespread Pain Index (WPI) ≥7 AND Symptom Severity Scale (SSS) ≥5, OR WPI 4–6 AND SSS ≥9
- Generalized pain in at least 4 of 5 regions (left upper, right upper, left lower, right lower, axial)
- Symptoms present at a similar level for at least 3 months
- Diagnosis is valid regardless of other diagnoses (fibromyalgia can coexist with other conditions)
WPI: Count of 19 specified body areas where patient has had pain in the past week (0–19). SSS: Sum of severity scores (0–3 each) for fatigue, waking unrefreshed, and cognitive symptoms, plus number of somatic symptoms (0–3) = total 0–12.
FDA-Approved Pharmacotherapy
| Drug | Class | Dose | Mechanism in Fibromyalgia | NNT |
|---|---|---|---|---|
| Duloxetine | SNRI | 60 mg daily (start 30 mg × 1 week) | Augments descending NE/5-HT inhibition | 7.2 for ≥50% pain reduction |
| Pregabalin | Gabapentinoid | 150–450 mg/day in divided doses | Alpha-2-delta Ca2+ channel modulation; reduces excitatory neurotransmitter release | 8.6 |
| Milnacipran | SNRI | 50 mg BID (titrate: 12.5 mg ×1d, 25 mg BID ×2d, 50 mg BID) | Stronger NE than 5-HT reuptake inhibition (3:1 ratio); analgesic and fatigue benefits | 7.7 |
Non-Pharmacologic Management
Exercise is the single most effective intervention (stronger evidence than any medication). Aerobic exercise (walking, swimming, cycling) 30 minutes, 3–5 times per week at moderate intensity. Strength training 2 times per week. Start low, progress slowly (“start low, go slow”). Cognitive behavioral therapy (CBT): Addresses catastrophizing, fear-avoidance beliefs, activity pacing; NNT ~4 for clinically meaningful improvement. Sleep optimization: Sleep hygiene, treat comorbid sleep disorders (sleep apnea is common); low-dose amitriptyline 10–25 mg QHS or cyclobenzaprine 5–10 mg QHS for non-restorative sleep.
Overlap Syndromes
Fibromyalgia frequently co-occurs with other central sensitization syndromes: irritable bowel syndrome (32–77%), tension-type/migraine headache (48–76%), temporomandibular disorder (18–75%), interstitial cystitis (12–21%), chronic fatigue syndrome (21–80%), and vulvodynia. The shared pathophysiology of augmented central pain processing suggests a common neurobiologic mechanism.
Fibromyalgia vs Other Chronic Pain Conditions
| Feature | Fibromyalgia | Polymyalgia Rheumatica | Hypothyroidism | Inflammatory Arthritis |
|---|---|---|---|---|
| Age | 20–50 (peak) | >50 (always >50) | Any age | Variable |
| Lab findings | Normal ESR/CRP, normal CK | Markedly elevated ESR (>40), CRP | Elevated TSH | Elevated ESR/CRP, positive RF/anti-CCP or HLA-B27 |
| Morning stiffness | Variable, non-specific | Severe, >45 min, shoulder/hip girdles | Generalized | >30–60 min, improves with activity |
| Tender points | Widespread (WPI) | Proximal girdle muscles | Generalized myalgia | Joint-specific |
| Response to steroids | No improvement | Dramatic response to prednisone 15–20 mg | No (responds to levothyroxine) | Partial response |
- Step 1 (All patients): Patient education, aerobic exercise (30 min 3–5x/week), sleep hygiene, CBT referral
- Step 2 (Persistent symptoms): Add duloxetine 60 mg daily OR pregabalin 75–225 mg BID OR milnacipran 50 mg BID; low-dose amitriptyline 10–25 mg QHS for sleep
- Step 3 (Partial response): Combination therapy (SNRI + gabapentinoid); aquatic therapy; acupuncture (EULAR conditional recommendation)
- Step 4 (Refractory): Reassess diagnosis; treat comorbid psychiatric conditions; consider low-dose naltrexone (4.5 mg QHS — off-label, emerging evidence); multidisciplinary pain rehabilitation program
24 Perioperative Pain & ERAS
Preemptive & Preventive Analgesia
Preemptive analgesia is administration of analgesics before surgical incision to prevent central sensitization. Preventive analgesia (broader concept) encompasses any perioperative intervention that reduces post-surgical pain beyond the expected duration of the drug. Evidence supports multimodal preventive strategies, not single-agent preemptive dosing.
Multimodal Opioid-Sparing Protocol
| Timing | Agent | Dose | Rationale |
|---|---|---|---|
| Preoperative | Acetaminophen | 1000 mg PO | Central analgesic (COX-3 inhibition) |
| Preoperative | Gabapentin | 300–600 mg PO | Alpha-2-delta modulation; reduces postoperative opioid consumption by 20–30%; anxiolysis |
| Preoperative | Celecoxib | 200–400 mg PO | COX-2 inhibition; no platelet effect (safe preoperatively) |
| Intraoperative | Ketorolac | 15–30 mg IV at wound closure | Potent parenteral NSAID; synergistic with acetaminophen |
| Intraoperative | Dexamethasone | 4–8 mg IV | Anti-inflammatory; antiemetic; extends block duration |
| Intra/Postop | Regional anesthesia | Procedure-specific (see below) | Site-specific analgesia; blocks afferent nociceptive transmission |
| Postoperative | Acetaminophen | 1000 mg PO q6h (scheduled) | Maintain central analgesia; avoid PRN dosing |
| Postoperative | Ibuprofen or celecoxib | 400 mg PO q6h or 200 mg PO BID | Anti-inflammatory synergy with acetaminophen |
| Postoperative (rescue) | Oxycodone | 5 mg PO q4h PRN | Reserved for breakthrough pain not controlled by multimodal regimen |
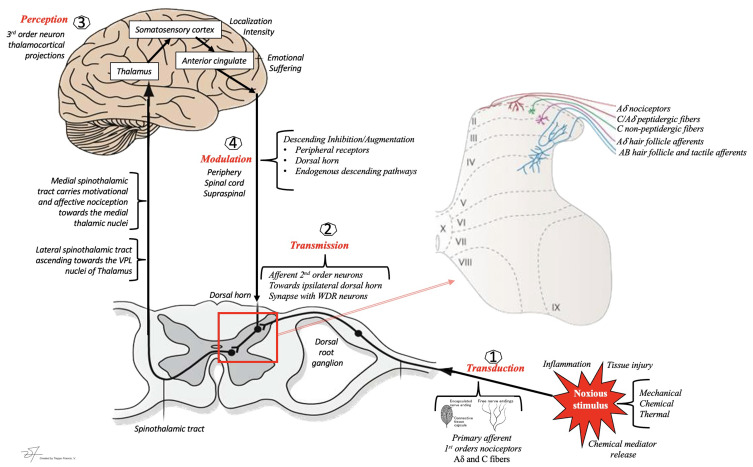
Ketamine & Lidocaine Infusions
Ketamine infusion: Bolus 0.5 mg/kg IV at induction, then 0.1–0.25 mg/kg/h intraoperatively and 0.05–0.15 mg/kg/h for 24–48h postoperatively. Indicated for opioid-tolerant patients, major surgery, or chronic pain patients. Reduces opioid consumption by 30–50%. IV lidocaine infusion: Bolus 1–2 mg/kg IV, then 1–2 mg/min (or 1.5 mg/kg/h) intraoperatively and 24–48h postoperatively. Strongest evidence for abdominal surgery (reduces ileus, pain scores, opioid use, length of stay). Monitor for LAST (local anesthetic systemic toxicity); do not exceed 4.5 mg/kg/h.
Regional Blocks by Surgery Type
| Surgery | Block | Coverage |
|---|---|---|
| Total knee arthroplasty | Adductor canal (saphenous) block + periarticular injection (PAI) | Anterior knee; motor-sparing (preserves quad function for early ambulation) |
| Total hip arthroplasty | Fascia iliaca block or pericapsular nerve group (PENG) block | Hip joint innervation (femoral, obturator, lateral femoral cutaneous) |
| Shoulder surgery | Interscalene brachial plexus block | C5–C7 distribution; risk of phrenic nerve palsy (avoid in contralateral diaphragm paresis) |
| Breast surgery | Pectoral nerve blocks (PECS I/II) or erector spinae plane (ESP) block | Anterior chest wall, axilla |
| Abdominal surgery (open) | Transversus abdominis plane (TAP) block or rectus sheath block or thoracic epidural | Abdominal wall (T6–L1) |
| Thoracotomy | Thoracic epidural or paravertebral block or erector spinae plane block | Unilateral or bilateral thoracic dermatomes; epidural is gold standard for thoracotomy |
| Rib fractures | Serratus anterior plane block or thoracic epidural or paravertebral block | Lateral chest wall; epidural for bilateral fractures |
Enhanced Recovery After Surgery (ERAS)
ERAS pathways integrate multimodal analgesia with other perioperative optimizations: preoperative carbohydrate loading, avoidance of prolonged fasting, early ambulation, early oral intake, minimally invasive technique, goal-directed fluid therapy, and early Foley removal. Pain management is a central pillar — opioid-sparing multimodal regimens reduce opioid-related complications (ileus, PONV, urinary retention, sedation) and accelerate recovery.
Opioid-Free Anesthesia (OFA)
An emerging paradigm using combinations of non-opioid analgesics to provide complete intraoperative and postoperative analgesia without any opioid. Typical OFA protocol includes: dexmedetomidine infusion (0.5–1.0 mcg/kg/h), ketamine infusion (0.1–0.3 mg/kg/h), lidocaine infusion (1.5 mg/kg/h), magnesium sulfate (30–50 mg/kg bolus then 10 mg/kg/h), and regional anesthesia. Evidence supports reduced PONV, faster GI recovery, and potentially less chronic postsurgical pain development. Not universally applicable — requires appropriate patient selection and anesthesia team experience.
Prevention of Chronic Postsurgical Pain (CPSP)
CPSP affects 10–50% of surgical patients (highest after thoracotomy, mastectomy, amputation, hernia repair). Risk factors: preoperative pain >1 month, younger age, female sex, psychological vulnerability (anxiety, catastrophizing), genetic predisposition (COMT polymorphisms), intraoperative nerve injury, and severity of acute postoperative pain. Prevention strategies: aggressive multimodal perioperative analgesia, nerve-sparing surgical technique, regional anesthesia, perioperative ketamine (NMDA antagonism reduces central sensitization), gabapentinoid pre-treatment, and early psychological intervention for high-risk patients.
25 Regulatory & Ethical Considerations
DEA Scheduling of Controlled Substances
| Schedule | Definition | Pain-Relevant Examples | Prescribing Rules |
|---|---|---|---|
| Schedule I | No accepted medical use, high abuse potential | Heroin, MDMA, psilocybin (cannabis per federal law) | Cannot prescribe; research only |
| Schedule II | High abuse potential; accepted medical use with severe restrictions | Morphine, oxycodone, hydromorphone, fentanyl, methadone, hydrocodone, amphetamines | Written/e-prescribed Rx; no refills; max 90-day supply (varies by state) |
| Schedule III | Moderate abuse potential | Buprenorphine, acetaminophen/codeine, ketamine (for compounding) | 5 refills in 6 months; phone-in Rx permitted |
| Schedule IV | Low abuse potential | Tramadol, carisoprodol, benzodiazepines, zolpidem | 5 refills in 6 months |
| Schedule V | Lowest abuse potential | Pregabalin, gabapentin (in some states), cough syrups with small amounts of codeine | Varies by state; OTC in some states |
State Prescribing Laws
Significant state-to-state variation in: initial opioid prescription limits (many states limit to 3–7 days for acute pain), PDMP checking requirements (most states mandate at initiation and periodically), continuing education mandates, prescriber-patient relationship requirements, and telemedicine prescribing rules for controlled substances (Ryan Haight Act requires in-person exam for most controlled substance prescriptions, with COVID-era flexibilities under review).
Medical Marijuana
As of 2026, medical marijuana is legal in 38+ states and DC. Common qualifying conditions include chronic pain, cancer, PTSD, epilepsy, and MS. Federal Schedule I status creates tensions: DEA registrants risk licensure; cannot prescribe (can only “certify” or “recommend”); VA physicians cannot recommend; crosses state lines is trafficking. Evidence is strongest for chronic neuropathic pain (NNT 5.6) and chemotherapy-induced nausea. Limited evidence for fibromyalgia, arthritis, headache. Risks: CUD (9% lifetime), impaired driving, psychosis in predisposed, respiratory effects with smoking.
Ethical Considerations
Informed consent for chronic opioids: Must include realistic discussion of benefits (modest, 30% responder rate for chronic non-cancer pain), risks (addiction 8–12% in chronic pain patients, overdose, endocrinopathy, falls, OIH), alternatives, monitoring requirements, and conditions for discontinuation. Palliative exception: In end-of-life care, adequate pain treatment takes priority over the risk of dependence; the principle of double effect permits medications that may hasten death if the primary intent is symptom relief. Equitable pain treatment: Systematic disparities exist — Black and Hispanic patients receive fewer opioid prescriptions and lower doses than White patients for the same conditions; women are more likely to have pain dismissed as psychogenic. Address implicit bias through standardized assessment tools, clinical decision support, and institutional audit.
Documentation Standards for Chronic Opioid Therapy
- Pain assessment: NRS intensity, functional status (PEG score), quality of life impact
- 5 A’s: Analgesia (pain relief), Activity (functional improvement), Adverse effects, Aberrant behaviors, Affect (mood)
- PDMP review: Date checked, findings, any concerns
- UDS results: Date obtained, findings, concordance with prescribed medications
- Treatment agreement: Confirm in effect; review if any concerns
- Naloxone: Prescribed and education provided (document for ≥50 MME or risk factors)
- Risk reassessment: Ongoing evaluation of risk-benefit ratio; document clinical rationale for continuation
- Non-pharmacologic therapies: Active engagement in PT, psychology, self-management strategies
- Exit strategy: Document plan for eventual taper or clearly articulate ongoing need
26 Equianalgesic & MME Conversion Tables
Comprehensive Equianalgesic Table
| Opioid | PO (mg) | IV/IM/SC (mg) | PO:IV Ratio | MME Factor | Duration |
|---|---|---|---|---|---|
| Morphine | 30 | 10 | 3:1 | 1.0 | 3–4 h (IR) |
| Oxycodone | 20 | — | — | 1.5 | 3–6 h (IR) |
| Hydrocodone | 30 | — | — | 1.0 | 4–6 h |
| Hydromorphone | 6 | 1.5 | 4:1 | 4.0 | 3–4 h |
| Oxymorphone | 10 | 1 | 10:1 | 3.0 | 3–6 h |
| Codeine | 200 | 120 | 1.7:1 | 0.15 | 4–6 h |
| Tramadol | 300 (approximate) | — | — | 0.1 | 4–6 h |
| Tapentadol | 75 | — | — | 0.4 | 4–6 h |
| Meperidine | 300 | 75 | 4:1 | 0.1 | 2–3 h (avoid — neurotoxic metabolite normeperidine) |
| Fentanyl IV | — | 0.1 (100 mcg) | — | — | 0.5–1 h IV; patch 72 h |
| Fentanyl Transdermal | 25 mcg/h patch ≈ 60–90 mg oral morphine/day (use 60 mg for conservative conversion, 90 mg per manufacturer) | ||||
| Methadone | See variable ratio: ≤30 mg morphine → 2:1; 31–90 mg → 4:1; 91–300 mg → 8:1; >300 mg → 12:1 | ||||
| Levorphanol | 4 | 2 | 2:1 | 11.0 | 6–8 h |
Methadone Conversion Table (Morphine → Methadone)
| Total Daily Oral Morphine Equivalent (mg) | Morphine:Methadone Ratio | Example (Methadone Daily Dose) |
|---|---|---|
| ≤30 | 2:1 | 30 mg morphine → 15 mg methadone |
| 31–90 | 4:1 | 60 mg morphine → 15 mg methadone |
| 91–300 | 8:1 | 240 mg morphine → 30 mg methadone |
| 301–600 | 12:1 | 480 mg morphine → 40 mg methadone |
| 601–1000 | 15:1 | 750 mg morphine → 50 mg methadone |
| >1000 | 20:1 | 1200 mg morphine → 60 mg methadone |
27 Scoring Tools
Numeric Rating Scale (NRS)
Patient rates pain intensity on a scale from 0 (no pain) to 10 (worst imaginable pain). Clinically meaningful change: ≥2 points or ≥30% reduction. Mild: 1–3; Moderate: 4–6; Severe: 7–10. Most widely used pain scale in clinical practice. Limitation: unidimensional (intensity only).
Visual Analog Scale (VAS)
100 mm horizontal line anchored at “no pain” (0 mm) and “worst pain imaginable” (100 mm). Patient marks a point. Distance measured in mm. Clinically meaningful change: ≥13 mm. More sensitive than NRS for detecting small changes (preferred in research).
FLACC Scale (Pediatric/Nonverbal)
| Category | 0 | 1 | 2 |
|---|---|---|---|
| Face | No particular expression; smile | Occasional grimace/frown; withdrawn | Frequent/constant frown; clenched jaw; quivering chin |
| Legs | Normal position; relaxed | Uneasy, restless, tense | Kicking or legs drawn up |
| Activity | Lying quietly; normal position; moves easily | Squirming, shifting, tense | Arched, rigid, or jerking |
| Cry | No cry (awake or asleep) | Moans, whimpers; occasional complaint | Crying steadily; screams; frequent complaints |
| Consolability | Content; relaxed | Reassured by occasional touching/hugging/talking; distractible | Difficult to console or comfort |
Total score 0–10. For ages 2 months–7 years and nonverbal patients. Score ≥4 indicates moderate pain requiring intervention.
CPOT (Critical-Care Pain Observation Tool)
| Indicator | 0 | 1 | 2 |
|---|---|---|---|
| Facial expression | Relaxed, neutral | Tense (brow lowering, orbit tightening) | Grimacing (all above + eyelid tightly closed) |
| Body movements | Absence of movements | Protection (slow, cautious, touching pain site) | Restlessness (pulling at tubes, attempting to sit up, thrashing) |
| Muscle tension (upper extremity) | Relaxed | Tense, rigid | Very tense or rigid |
| Compliance with ventilator (intubated) OR vocalization (extubated) | Tolerating / talking in normal tone | Coughing but tolerating / sighing, moaning | Fighting ventilator / crying out, sobbing |
Total score 0–8. Score ≥3 suggests clinically significant pain. Validated for ICU patients who cannot self-report.
Opioid Risk Tool (ORT)
| Risk Factor | Female Score | Male Score |
|---|---|---|
| Family history of substance abuse — Alcohol | 1 | 3 |
| Family history of substance abuse — Illegal drugs | 2 | 3 |
| Family history of substance abuse — Prescription drugs | 4 | 4 |
| Personal history of substance abuse — Alcohol | 3 | 3 |
| Personal history of substance abuse — Illegal drugs | 4 | 4 |
| Personal history of substance abuse — Prescription drugs | 5 | 5 |
| Age 16–45 | 1 | 1 |
| History of preadolescent sexual abuse | 3 | 0 |
| Psychological disease (ADD, OCD, bipolar, schizophrenia, depression) | 2 | 2 |
Total: 0–3 low risk; 4–7 moderate risk; ≥8 high risk for aberrant opioid behavior.
SOAPP-R (Screener and Opioid Assessment for Patients with Pain — Revised)
24-item self-report questionnaire; each item scored 0–4 (never to very often). Sample items: “How often do you have mood swings?”; “How often have you felt a need for higher doses of medication?”; “How often have others expressed concern over your use of medication?” Total score ≥18 indicates high risk for aberrant drug behavior. Sensitivity 0.81, specificity 0.68. Administer before initiating chronic opioid therapy. Recheck with COMM (Current Opioid Misuse Measure) during ongoing treatment.
Budapest Criteria for CRPS
See Section 21 for the complete enumeration of the Budapest Clinical Diagnostic Criteria (symptoms in 3 of 4 categories, signs in 2 of 4 categories, no alternative diagnosis).
PEG Scale
See Section 2 for the 3-item PEG rapid screening tool (Pain, Enjoyment, General activity). Score ≥4 suggests clinically significant pain. Minimum clinically important difference is 1.0 point.
DN4 (Douleur Neuropathique en 4 Questions)
| Interview Items (patient-reported) | Examination Items (clinician) |
|---|---|
| 1. Does the pain have burning characteristics? | 5. Is there hypoesthesia to touch? |
| 2. Does the pain have cold characteristics? | 6. Is there hypoesthesia to pinprick? |
| 3. Is the pain associated with electric shocks? | 7. Is the pain provoked or increased by brushing (allodynia)? |
| 4. Is the pain associated with tingling, pins and needles, numbness, or itching? |
Score ≥4/7 suggests neuropathic pain (sensitivity 83%, specificity 90%). Useful for screening in primary care to determine need for neuropathic pain agents.
28 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AAPM | American Academy of Pain Medicine |
| ACD | Abuse-Deterrent Formulation |
| APS | American Pain Society |
| ASRA | American Society of Regional Anesthesia and Pain Medicine |
| BPI | Brief Pain Inventory |
| BTcP | Breakthrough Cancer Pain |
| CGRP | Calcitonin Gene-Related Peptide |
| CMM | Comprehensive Medical Management |
| CNB | Central Neuraxial Block |
| COMM | Current Opioid Misuse Measure |
| COX | Cyclooxygenase |
| CPOT | Critical-Care Pain Observation Tool |
| CRPS | Complex Regional Pain Syndrome |
| CSE | Combined Spinal-Epidural |
| CT | Computed Tomography |
| DEA | Drug Enforcement Administration |
| DIRE | Diagnosis, Intractability, Risk, Efficacy |
| DPN | Diabetic Peripheral Neuropathy |
| DRG | Dorsal Root Ganglion |
| EMG | Electromyography |
| ERAS | Enhanced Recovery After Surgery |
| ER/LA | Extended-Release / Long-Acting |
| ESI | Epidural Steroid Injection |
| ESP | Erector Spinae Plane (block) |
| EUS | Endoscopic Ultrasound |
| FBSS | Failed Back Surgery Syndrome |
| FDA | Food and Drug Administration |
| FLACC | Face, Legs, Activity, Cry, Consolability |
| GABA | Gamma-Aminobutyric Acid |
| GIRK | G-protein Inwardly Rectifying Potassium (channel) |
| GMI | Graded Motor Imagery |
| GON | Greater Occipital Nerve |
| GPCR | G-Protein Coupled Receptor |
| GRK | G-Protein-Coupled Receptor Kinase |
| HA | Hyaluronic Acid |
| HPA | Hypothalamic-Pituitary-Adrenal (axis) |
| IASP | International Association for the Study of Pain |
| ICHD | International Classification of Headache Disorders |
| IDDS | Intrathecal Drug Delivery System |
| IL | Interlaminar |
| IPG | Implantable Pulse Generator |
| IR | Immediate-Release |
| IV | Intravenous |
| KL | Kellgren-Lawrence (OA grading) |
| LA | Local Anesthetic |
| LAST | Local Anesthetic Systemic Toxicity |
| LBP | Low Back Pain |
| LON | Lesser Occipital Nerve |
| LSS | Lumbar Spinal Stenosis |
| LTP | Long-Term Potentiation |
| MAID | Medical Assistance in Dying |
| MBB | Medial Branch Block |
| MILD | Minimally Invasive Lumbar Decompression |
| MME | Morphine Milligram Equivalent |
| MPQ | McGill Pain Questionnaire |
| MRI | Magnetic Resonance Imaging |
| NCS | Nerve Conduction Study |
| NE | Norepinephrine |
| NMDA | N-Methyl-D-Aspartate |
| NNT | Number Needed to Treat |
| NRS | Numeric Rating Scale |
| NSAID | Nonsteroidal Anti-Inflammatory Drug |
| OA | Osteoarthritis |
| OIAD | Opioid-Induced Androgen Deficiency |
| OIH | Opioid-Induced Hyperalgesia |
| ORT | Opioid Risk Tool |
| OUD | Opioid Use Disorder |
| PACC | Polyanalgesic Consensus Conference |
| PAG | Periaqueductal Gray |
| PAI | Periarticular Injection |
| PDMP | Prescription Drug Monitoring Program |
| PDPH | Post-Dural Puncture Headache |
| PEG | Pain, Enjoyment, General Activity (scale) |
| PENG | Pericapsular Nerve Group (block) |
| PHN | Postherpetic Neuralgia |
| PMMA | Polymethyl Methacrylate |
| PNS | Peripheral Nerve Stimulation |
| PRP | Platelet-Rich Plasma |
| PSIS | Posterior Superior Iliac Spine |
| QSART | Quantitative Sudomotor Axon Reflex Test |
| REMS | Risk Evaluation and Mitigation Strategy |
| RF / RFA | Radiofrequency / Radiofrequency Ablation |
| RVM | Rostral Ventromedial Medulla |
| SCI | Spinal Cord Injury |
| SCS | Spinal Cord Stimulation |
| SF-MPQ | Short Form McGill Pain Questionnaire |
| SI | Sacroiliac |
| SLR | Straight Leg Raise |
| SMP | Sympathetically Maintained Pain |
| SNRI | Serotonin-Norepinephrine Reuptake Inhibitor |
| SOAPP-R | Screener and Opioid Assessment for Patients with Pain — Revised |
| TAP | Transversus Abdominis Plane (block) |
| TCA | Tricyclic Antidepressant |
| TENS | Transcutaneous Electrical Nerve Stimulation |
| TF | Transforaminal |
| TIRF | Transmucosal Immediate-Release Fentanyl |
| TRPV1 | Transient Receptor Potential Vanilloid 1 |
| UDS | Urine Drug Screen |
| US | Ultrasound |
| VAS | Visual Analog Scale |
| VCF | Vertebral Compression Fracture |
| WDR | Wide Dynamic Range (neuron) |
| WHO | World Health Organization |
| WPI | Widespread Pain Index |