Sleep Medicine
Obstructive sleep apnea, insomnia, narcolepsy, circadian rhythm disorders, parasomnias, polysomnography interpretation, CPAP management, and every scoring system, diagnostic criterion, and treatment algorithm across the full scope of sleep medicine.
01 Sleep Neurophysiology
Sleep is an active, highly regulated neurobiological process governed by the interplay of circadian and homeostatic drives. Understanding the neuroanatomical centers, neurotransmitter systems, and electrophysiologic signatures of each sleep stage is the foundation for interpreting polysomnography and diagnosing sleep disorders.
Sleep-Wake Neurotransmitter Systems
Wakefulness is maintained by a redundant network of ascending arousal pathways, while sleep is initiated by a discrete inhibitory center. The key neurotransmitters and their sources are as follows:
| Neurotransmitter | Source | Role in Sleep-Wake Regulation |
|---|---|---|
| Orexin (Hypocretin) | Lateral hypothalamus (~70,000 neurons) | Stabilizes wakefulness; prevents inappropriate transitions into sleep. Loss causes narcolepsy type 1. Target of dual orexin receptor antagonists (DORAs) |
| GABA | Ventrolateral preoptic area (VLPO), cortical interneurons | Primary inhibitory neurotransmitter; VLPO GABAergic neurons silence arousal centers to initiate and maintain sleep. Target of benzodiazepines and Z-drugs |
| Adenosine | Accumulates in basal forebrain during waking | Homeostatic sleep pressure marker (Process S). Inhibits cholinergic arousal neurons. Caffeine is a competitive adenosine receptor antagonist |
| Melatonin | Pineal gland (via SCN-superior cervical ganglion pathway) | Circadian timing signal; promotes sleep onset. Suppressed by light (especially 460-480 nm blue wavelength). Exogenous melatonin shifts circadian phase |
| Acetylcholine | Pedunculopontine (PPT) and laterodorsal tegmental (LDT) nuclei, basal forebrain | Promotes cortical activation in wake and REM. "REM-on" neurons. Suppressed during NREM |
| Norepinephrine | Locus coeruleus (LC) | Wake-promoting; silent during REM (allows REM atonia). LC firing rate: high in wake, low in NREM, off in REM |
| Serotonin (5-HT) | Dorsal raphe nucleus | Wake-promoting; suppresses REM. SSRIs increase serotonergic tone and suppress REM sleep |
| Histamine | Tuberomammillary nucleus (TMN) | Wake-promoting. Antihistamines (diphenhydramine, doxepin) cause sedation by blocking H1 receptors. Pitolisant is an H3 inverse agonist that increases histamine release |
| Dopamine | Ventral tegmental area (VTA), ventral periaqueductal gray | Wake-promoting. Modafinil and amphetamines increase dopaminergic tone |
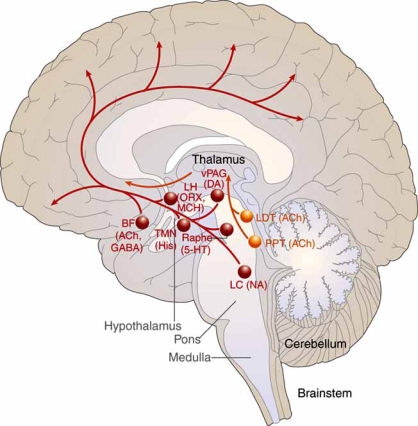
Key Neuroanatomical Centers
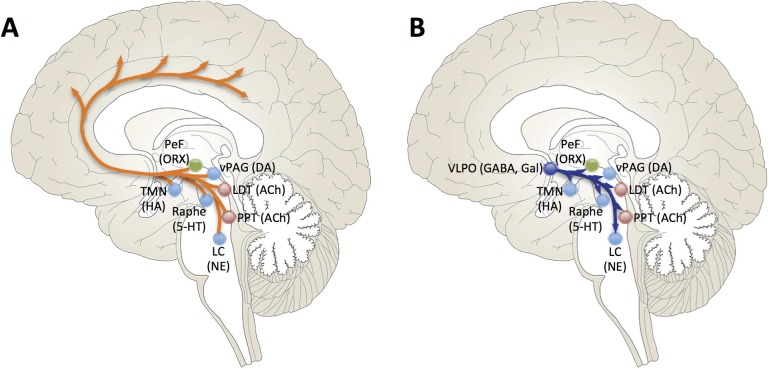
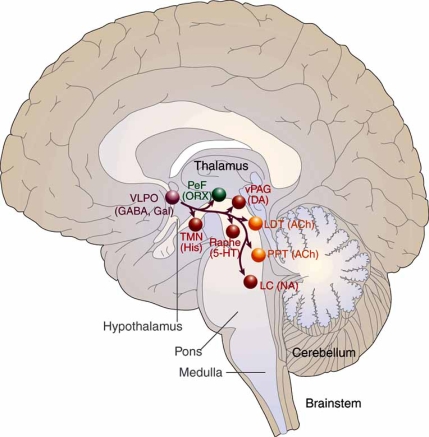
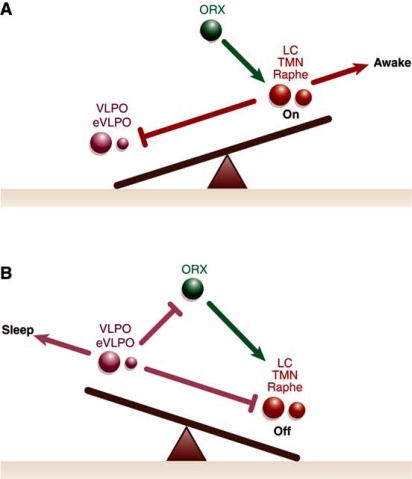
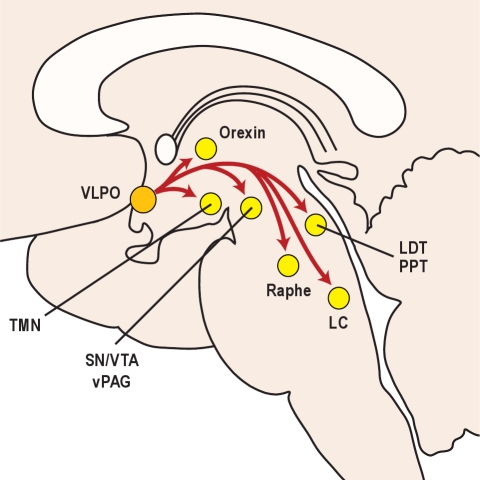
The ventrolateral preoptic area (VLPO) in the anterior hypothalamus is the master sleep-promoting center. It contains GABAergic and galaninergic neurons that, when active, inhibit all major arousal centers simultaneously — the LC, TMN, raphe, PPT/LDT, and orexin neurons. The suprachiasmatic nucleus (SCN) in the anterior hypothalamus is the master circadian pacemaker. It receives direct retinal input via the retinohypothalamic tract (melanopsin-containing retinal ganglion cells) and generates a near-24-hour endogenous rhythm that entrains to the light-dark cycle. The SCN drives circadian variation in alertness, core body temperature, cortisol secretion, and melatonin release.
The ascending arousal system consists of two branches: (1) a dorsal pathway from PPT/LDT cholinergic neurons projecting to the thalamus, gating sensory information to the cortex; and (2) a ventral pathway from monoaminergic nuclei (LC, raphe, TMN) plus orexin neurons projecting directly to the cortex and basal forebrain. These pathways are redundant — lesioning any single center rarely produces profound hypersomnia, but loss of orexin destabilizes the entire system, producing the state-switching instability of narcolepsy.
NREM Sleep — Stages N1, N2, N3
Stage N1 (light sleep, 2-5% of total sleep time): Transition from wakefulness. EEG shows attenuation of alpha rhythm (8-13 Hz) and appearance of low-amplitude, mixed-frequency theta activity (4-7 Hz). Slow rolling eye movements on EOG. Vertex sharp waves may appear. Easily arousable; patients may deny they were sleeping. Hypnic jerks (sleep starts) are normal N1 phenomena.
Stage N2 (stable sleep, 45-55% of total sleep time): Defined by the appearance of K-complexes — high-amplitude biphasic waves ≥0.5 seconds duration, maximal over frontal derivations, representing cortical responses to stimuli — and sleep spindles — bursts of 11-16 Hz (sigma frequency) activity lasting ≥0.5 seconds, generated by thalamocortical circuits involving the reticular nucleus of the thalamus. Sleep spindles reflect thalamic gating of sensory input, protecting sleep continuity. Higher spindle density is associated with better sleep maintenance and possibly memory consolidation.
Stage N3 (slow-wave sleep / deep sleep, 15-25% of total sleep time): EEG dominated by high-amplitude (≥75 microvolts), low-frequency delta waves (0.5-2 Hz) present in ≥20% of the epoch. Generated by synchronized cortical neuronal oscillations. Highest arousal threshold — most difficult stage from which to awaken. Predominates in the first third of the night. Decreases with age (may virtually disappear in the elderly). Growth hormone secretion is coupled to N3. NREM parasomnias (sleepwalking, sleep terrors) arise from N3.
REM Sleep Physiology
REM sleep (20-25% of total sleep time) is characterized by three cardinal features: (1) low-amplitude, mixed-frequency EEG resembling wakefulness (hence "paradoxical sleep"); (2) rapid eye movements on EOG — conjugate, irregular, sharply peaked; and (3) skeletal muscle atonia — active inhibition of spinal motor neurons via glycinergic and GABAergic pathways from the sublaterodorsal (SLD) nucleus in the pons, with the exception of the diaphragm and extraocular muscles. Loss of REM atonia is the pathophysiologic basis of REM sleep behavior disorder.
PGO waves (ponto-geniculo-occipital) are phasic bursts of activity originating in the pons, propagating through the lateral geniculate nucleus to the occipital cortex, occurring just before and during REM periods. They are thought to underlie the visual imagery of dreams. Phasic REM (with eye movement bursts and muscle twitches) alternates with tonic REM (without these phasic events).
Normal Sleep Architecture & the Hypnogram
A normal night consists of 4-6 ultradian cycles, each approximately 90 minutes in duration. Each cycle progresses through N1 → N2 → N3 → N2 → REM. The composition shifts across the night: early cycles are rich in N3 (slow-wave sleep dominates the first third), while later cycles are rich in REM (REM episodes lengthen across the night, with the longest REM period typically in the last cycle before awakening). Normal sleep latency (time from lights out to first epoch of sleep) is 10-20 minutes. REM latency (time from sleep onset to first REM epoch) is normally 70-120 minutes — shortened REM latency (<15 minutes) suggests narcolepsy, sleep deprivation, or REM-rebound from substance withdrawal.
Circadian Rhythm — Process S & Process C
The two-process model of sleep regulation (Borbely, 1982) describes the interaction of homeostatic and circadian drives. Process S (homeostatic sleep pressure) accumulates exponentially during waking, driven primarily by adenosine accumulation in the basal forebrain, and dissipates exponentially during sleep (especially slow-wave sleep). Process C (circadian alerting signal) is generated by the SCN and oscillates with near-24-hour periodicity independent of prior sleep. The SCN promotes wakefulness during the day and facilitates sleep during the biological night. The interaction of these two processes determines sleep propensity at any given moment: when Process S is high and Process C is low (nighttime), sleep pressure is maximal.
Core body temperature follows a circadian rhythm with a nadir (Tmin) approximately 2-3 hours before habitual wake time (typically ~4:00-5:00 AM). The temperature minimum is the critical anchor point for chronotherapy: light exposure before Tmin delays circadian phase, while light after Tmin advances it. Melatonin onset (DLMO) occurs approximately 2-3 hours before habitual bedtime and marks the beginning of the biological night.
Age-Related Changes in Sleep Architecture
Sleep architecture changes dramatically across the lifespan. Newborns spend ~50% of sleep in active sleep (REM equivalent), with a polyphasic pattern and no circadian rhythm. By 6 months, circadian rhythm consolidates and REM percentage begins to decline. Children have abundant N3 (slow-wave sleep), high sleep efficiency (>95%), and short sleep latency. Adolescents experience a physiologic circadian delay (later DLMO) and reduced homeostatic sleep pressure, creating a "perfect storm" of late bedtimes and early school start times. In older adults, N3 declines markedly (may be absent after age 60-70 in some individuals), sleep efficiency decreases (<80%), WASO increases, sleep latency lengthens, and the circadian amplitude dampens with phase advance (earlier bedtime and wake time). The arousal threshold decreases, making the elderly more susceptible to environmental disturbances.
Homeostatic vs. Circadian Regulation — Clinical Implications
The two-process model explains several clinical phenomena. Recovery sleep after sleep deprivation is disproportionately rich in N3 (SWS rebound — reflecting homeostatic debt). REM rebound occurs after REM deprivation (from medications, substances, or sleep disorders) and manifests as increased REM percentage, shorter REM latency, and more intense dreaming — this is why SSRI discontinuation or alcohol withdrawal produces vivid nightmares. The circadian nadir (2:00-6:00 AM) and the post-lunch dip (1:00-3:00 PM, a true circadian phenomenon independent of meal intake) are periods of maximal sleepiness and accident risk. Shift workers and long-haul drivers are most vulnerable during these windows.
02 Polysomnography & Scoring
Polysomnography (PSG) is the gold-standard diagnostic study in sleep medicine. It provides continuous, simultaneous recording of multiple physiologic parameters during sleep. Interpretation requires systematic application of AASM (American Academy of Sleep Medicine) scoring rules.
Standard PSG Montage — Channels
| Channel Type | Derivations | Purpose |
|---|---|---|
| EEG | F3-M2, F4-M1, C3-M2, C4-M1, O1-M2, O2-M1 (recommended: frontal for slow waves, central for spindles/K-complexes, occipital for alpha rhythm) | Sleep staging, arousal detection, seizure identification |
| EOG | E1-M2, E2-M2 (electrodes placed 1 cm above and below outer canthi) | Detect eye movements — slow rolling (N1), rapid conjugate (REM) |
| Chin EMG | Mentalis / submentalis muscles | Muscle tone assessment — high in wake, reduced in NREM, atonia in REM. Elevated REM tone suggests RBD |
| Airflow | Oronasal thermal sensor (thermistor) + nasal pressure transducer | Thermistor: detect apneas (cessation of airflow). Nasal pressure: detect hypopneas (flow limitation). Nasal pressure is more sensitive |
| Respiratory effort | Thoracic and abdominal respiratory inductance plethysmography (RIP) belts | Distinguish obstructive (paradoxical chest/abdomen movement) from central (absent movement) events |
| Pulse oximetry | Finger probe, sampling rate ≥25 Hz with ≤3 sec averaging | Oxygen desaturation events, baseline SpO2, time below 90%/88% |
| ECG | Modified lead II | Heart rate, arrhythmia detection (bradycardia/tachycardia with apneas, AF) |
| Leg EMG | Bilateral anterior tibialis | Periodic limb movements of sleep (PLMS) |
| Body position | Position sensor | Supine vs lateral — positional OSA determination |
| Snore sensor | Microphone or piezo sensor | Document snoring events |
AASM Epoch Scoring — General Rules
PSG records are divided into sequential 30-second epochs. Each epoch is assigned a single sleep stage based on the predominant pattern. When two stages are present in one epoch, the stage composing the majority (>15 seconds) is assigned. Scoring proceeds from lights out to lights on. The AASM Manual for the Scoring of Sleep and Associated Events (current version) provides standardized rules.
Sleep Stage Scoring Criteria (AASM)
| Stage | EEG Criteria | EOG | Chin EMG |
|---|---|---|---|
| Wake (W) | Alpha rhythm (8-13 Hz) over occipital region with eyes closed; or low-amplitude mixed frequency with eyes open | Rapid eye movements, reading eye movements, eye blinks (0.5-2 Hz) | High tonic activity |
| N1 | Low-amplitude, mixed frequency (predominantly 4-7 Hz theta). Alpha attenuation (<50% of epoch). Vertex sharp waves | Slow rolling eye movements (sinusoidal, <0.5 Hz) | Variable, may decrease from wake |
| N2 | K-complexes (biphasic wave, ≥0.5 sec duration, maximal frontal) and/or sleep spindles (11-16 Hz, ≥0.5 sec) on a background of relatively low-amplitude, mixed-frequency activity | No eye movements typically | Low tonic activity |
| N3 | Slow-wave activity: ≥20% of epoch contains high-amplitude (≥75 microvolts peak-to-peak) delta waves (0.5-2 Hz), measured over frontal derivations | None typically | Low tonic activity |
| REM (R) | Low-amplitude, mixed-frequency EEG (similar to N1 but without vertex sharp waves). Sawtooth waves (2-6 Hz, triangular morphology, maximal central) may appear | Rapid eye movements — conjugate, irregular, sharply peaked | Atonia — lowest tonic EMG of any stage |
Arousal Scoring
An arousal is scored when there is an abrupt shift in EEG frequency (to alpha, theta, or frequencies >16 Hz, but NOT spindles) lasting ≥3 seconds, with at least 10 seconds of stable sleep preceding the event. In REM, there must be a concurrent increase in chin EMG lasting ≥1 second. The arousal index (arousals per hour of sleep) is reported; normal is <10 in young adults, increasing with age. An elevated arousal index fragments sleep architecture and contributes to daytime sleepiness even when the AHI is not severely elevated — this is the basis of the upper airway resistance syndrome (UARS), where flow limitation causes arousals without meeting apnea or hypopnea criteria.
Respiratory Event Scoring (AASM)
Obstructive apnea: ≥90% reduction in airflow (thermistor) from baseline, lasting ≥10 seconds, with continued or increased respiratory effort (RIP belts).
Central apnea: ≥90% reduction in airflow lasting ≥10 seconds with absent respiratory effort throughout the event.
Mixed apnea: Begins as central (no effort) then develops obstructive effort in the latter portion; total duration ≥10 seconds.
Hypopnea (recommended rule): ≥30% reduction in nasal pressure signal amplitude from baseline, lasting ≥10 seconds, associated with ≥3% oxygen desaturation OR an arousal.
Hypopnea (acceptable rule): ≥30% reduction in nasal pressure signal amplitude from baseline, lasting ≥10 seconds, associated with ≥4% oxygen desaturation (this stricter definition is required by CMS for CPAP coverage).
RERA (Respiratory Effort-Related Arousal): Sequence of breaths lasting ≥10 seconds with increasing respiratory effort or flattening of nasal pressure waveform, terminated by arousal, but not meeting apnea or hypopnea criteria.
The AHI (Apnea-Hypopnea Index) = (total apneas + hypopneas) / total sleep time in hours. The RDI (Respiratory Disturbance Index) = (apneas + hypopneas + RERAs) / total sleep time in hours. The RDI is always ≥ AHI. CMS uses AHI for coverage determinations.
PLMS Scoring Rules
Periodic limb movements are scored from the anterior tibialis EMG. An individual limb movement must last 0.5-10 seconds in duration with amplitude ≥8 microvolts above resting EMG. A series of PLMS requires ≥4 consecutive movements with inter-movement intervals of 5-90 seconds (measured onset to onset). Movements associated with respiratory events (within 0.5 seconds of a respiratory event termination) are scored separately and not counted toward the PLMI. The PLMS arousal index (PLMS associated with EEG arousals, per hour of sleep) may be more clinically relevant than the total PLMI, as it reflects the sleep-disrupting impact of the movements.
Split-Night Study
A split-night PSG combines diagnostic and therapeutic components in a single night. The first portion (typically ≥2 hours) is diagnostic; if the AHI meets criteria (usually ≥40 events/hr, or ≥20 with clinical judgment), CPAP titration begins in the second half. Advantages: time-efficient, cost-effective, reduces patient burden. Limitations: potentially inadequate diagnostic data (limited REM in the first half), and shortened titration time. AASM guidelines require ≥3 hours of CPAP titration for adequate titration. Not appropriate for patients with suspected CSA, hypoventilation, or parasomnias requiring full-night observation.
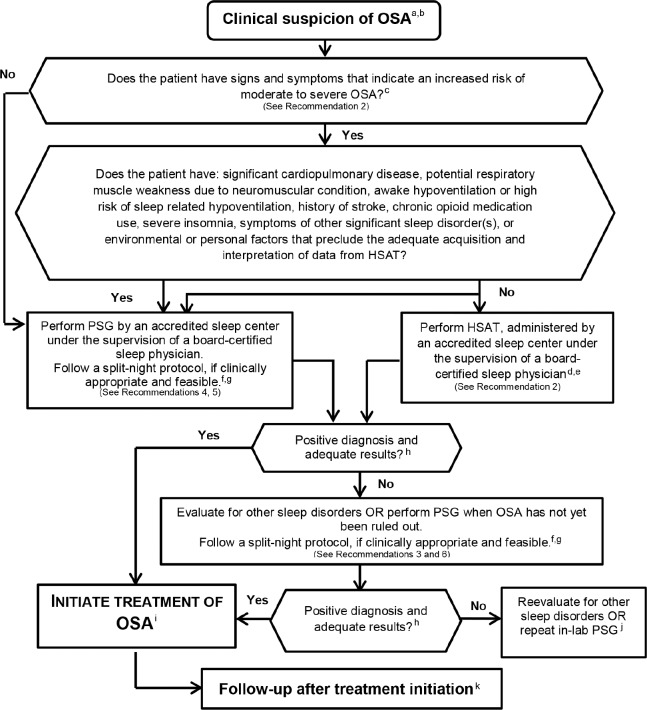
Home Sleep Apnea Testing (HSAT) — Type III Study
HSAT devices (Type III) record a minimum of 4 channels: airflow (nasal pressure ± thermistor), respiratory effort (RIP belts), and oxygen saturation (pulse oximetry), plus heart rate or ECG. They lack EEG, EOG, and EMG — they cannot determine sleep stages, arousals, or total sleep time. Appropriate for: adults with high pretest probability of moderate-to-severe OSA, without significant comorbid conditions (CHF, COPD, neuromuscular disease, chronic opioid use, suspected CSA, prior stroke, or severe insomnia). A negative HSAT does not rule out OSA — sensitivity for mild OSA is poor; in-lab PSG should follow if clinical suspicion persists.
03 Terminology & Abbreviations
| Term | Definition |
|---|---|
| Sleep latency | Time from lights out to the first epoch of any sleep stage |
| REM latency | Time from sleep onset to the first epoch of REM. Normal: 70-120 min. Shortened in narcolepsy, depression, sleep deprivation, alcohol/drug withdrawal |
| Sleep efficiency | Total sleep time / time in bed x 100%. Normal: >85%. Reduced in insomnia |
| WASO | Wake after sleep onset — total minutes of wakefulness from sleep onset to final awakening |
| Sleep fragmentation | Frequent brief arousals disrupting sleep continuity; measured by arousal index |
| AHI | Apnea-Hypopnea Index: events per hour of sleep |
| ODI | Oxygen Desaturation Index: number of ≥3% (or ≥4%) desaturations per hour of sleep |
| T90 | Percentage of total sleep time spent with SpO2 <90% |
| MSLT | Multiple Sleep Latency Test: 5 nap opportunities at 2-hour intervals; measures physiologic sleepiness and SOREMP count |
| SOREMP | Sleep-Onset REM Period: REM occurring within 15 minutes of sleep onset |
| MWT | Maintenance of Wakefulness Test: 4 trials measuring ability to stay awake in a dark, quiet room; used for fitness-for-duty |
| PLMS | Periodic Limb Movements of Sleep: repetitive dorsiflexion movements of the foot during sleep, 0.5-10 sec duration, 5-90 sec intervals, in sequences of ≥4 |
| PLMI | Periodic Limb Movement Index: PLMS events per hour of sleep. Clinically significant when ≥15/hr |
| Pcrit | Critical closing pressure of the upper airway; higher (less negative) values indicate greater collapsibility — central to OSA pathophysiology |
| DLMO | Dim Light Melatonin Onset: salivary/plasma melatonin rise in dim light conditions; gold standard circadian phase marker |
04 Obstructive Sleep Apnea
Obstructive sleep apnea (OSA) is characterized by repetitive upper airway collapse during sleep, leading to intermittent hypoxemia, hypercapnia, sleep fragmentation, and sympathetic surges. It is the most common sleep-related breathing disorder, affecting an estimated 10-30% of adults, with the majority undiagnosed.
Pathophysiology — Upper Airway Collapsibility
The pharynx is the only segment of the airway lacking rigid structural support. Its patency depends on a balance between collapsing forces (negative intraluminal pressure during inspiration, tissue weight, surface adhesion forces) and dilating forces (pharyngeal dilator muscle activity, particularly the genioglossus). The critical closing pressure (Pcrit) quantifies airway collapsibility: Pcrit in normal subjects is approximately -15 cm H2O (airway resists collapse), while in severe OSA it may be +5 cm H2O (airway collapses at atmospheric pressure). Four key pathophysiologic traits contribute to OSA (the PALM model): anatomic compromise (Pcrit), inadequate muscle responsiveness (pharyngeal dilator Activation), Low arousal threshold, and loop gain instability (Mixed ventilatory control).
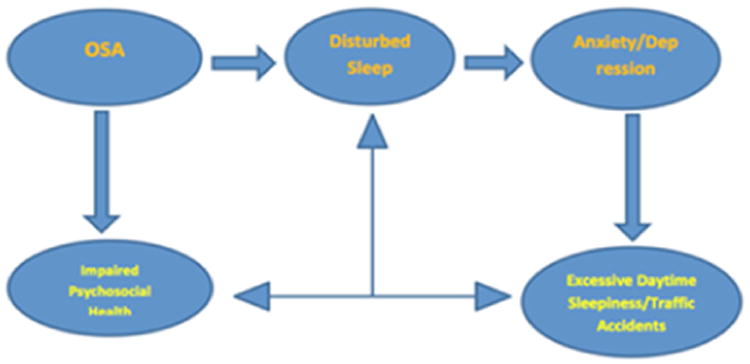
Risk Factors
Obesity (BMI ≥30 — strongest modifiable risk factor; each 10% weight gain increases AHI risk 6-fold), male sex (2-3:1 ratio, which narrows post-menopause), age (prevalence increases through 7th decade), neck circumference >17 inches (men) or >16 inches (women), craniofacial anatomy (retrognathia, micrognathia, macroglossia, enlarged tonsils, Mallampati III-IV), nasal obstruction, family history (first-degree relatives have 2-4x increased risk), alcohol/sedative use (reduce pharyngeal muscle tone), smoking, and endocrine conditions (hypothyroidism, acromegaly).
Clinical Presentation
Nocturnal symptoms: Loud, habitual snoring (often the presenting complaint — reported by bed partner), witnessed apneas (gasping, choking, snorting), restless sleep, frequent awakenings, nocturia (≥2 times per night — mediated by atrial natriuretic peptide release from cardiac distension during obstructive events), dry mouth upon awakening, nocturnal diaphoresis, and sleep-related gastroesophageal reflux.
Daytime symptoms: Excessive daytime sleepiness (the most functionally impactful symptom), unrefreshing sleep, morning headaches (CO2-mediated vasodilation), impaired concentration, memory difficulties, mood disturbance (irritability, depression), reduced libido, and impaired driving performance. Notably, ~50% of patients with moderate-to-severe OSA do not report subjective sleepiness (ESS <10) — the absence of sleepiness does not exclude clinically significant OSA.
Physical examination findings: Obesity (particularly central), large neck circumference, crowded oropharynx (Mallampati III-IV), enlarged tonsils (Friedman scale III-IV), retrognathia, macroglossia, high-arched palate, nasal septal deviation, inferior turbinate hypertrophy, and signs of pulmonary hypertension or right heart failure in severe, untreated cases.
STOP-BANG Screening Questionnaire
S — Snoring: Do you snore loudly (louder than talking or can be heard through closed doors)?
T — Tired: Do you often feel tired, fatigued, or sleepy during daytime?
O — Observed: Has anyone observed you stop breathing during your sleep?
P — Pressure: Are you being treated for high blood pressure?
B — BMI: BMI >35 kg/m2?
A — Age: Age >50 years?
N — Neck: Neck circumference >40 cm (16 inches)?
G — Gender: Male?
Scoring: 0-2 = low risk, 3-4 = intermediate risk, 5-8 = high risk for OSA. Sensitivity for moderate-severe OSA: ~93% at cutoff ≥3.
Epworth Sleepiness Scale (ESS)
Rate likelihood of dozing (0 = never, 1 = slight, 2 = moderate, 3 = high) in each situation:
1. Sitting and reading
2. Watching television
3. Sitting inactive in a public place (e.g., theater, meeting)
4. As a passenger in a car for an hour without a break
5. Lying down to rest in the afternoon when circumstances permit
6. Sitting and talking to someone
7. Sitting quietly after a lunch without alcohol
8. In a car, while stopped for a few minutes in traffic
Scoring: Total 0-24. Normal: 0-10. Mild sleepiness: 11-14. Moderate: 15-17. Severe: 18-24. ESS ≥10 suggests excessive daytime sleepiness warranting further evaluation.
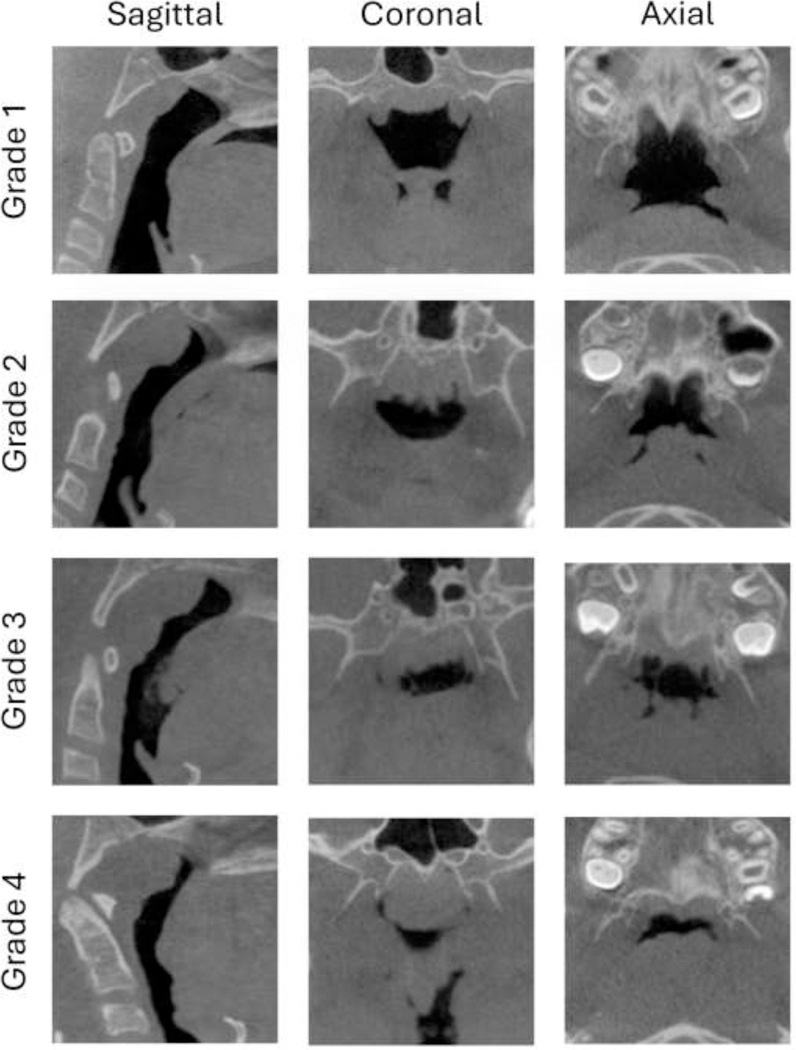
Diagnostic Criteria — AHI Severity Classification
OSA is diagnosed when the AHI is ≥5 events/hour with associated symptoms (sleepiness, witnessed apneas, choking/gasping) OR AHI ≥15 events/hour regardless of symptoms (ICSD-3). Severity grading:
| Severity | AHI (events/hour) | Clinical Significance |
|---|---|---|
| Mild | 5-14 | May be asymptomatic; treat if symptomatic, cardiovascular risk factors, or occupational concerns |
| Moderate | 15-29 | Usually symptomatic; treatment recommended |
| Severe | ≥30 | Strongly associated with cardiovascular morbidity; treatment is imperative |
05 CPAP & PAP Therapy
Continuous positive airway pressure (CPAP) is the first-line treatment for moderate-to-severe OSA. It acts as a pneumatic splint, maintaining positive intraluminal pressure throughout the respiratory cycle to prevent upper airway collapse. Optimal management requires proper titration, interface selection, troubleshooting, and adherence monitoring.
CPAP Titration
In-laboratory CPAP titration is performed during attended PSG. The technician starts at 4 cm H2O and increases pressure in 1 cm H2O increments (waiting ≥5 minutes at each level) to eliminate obstructive apneas, hypopneas, RERAs, and snoring in all body positions and sleep stages, including supine REM (the most vulnerable state). The optimal pressure is the single pressure that eliminates all respiratory events; typical range is 6-14 cm H2O for most adults. An AHI <5 during the titration with ≥15 minutes of supine REM at the selected pressure defines an optimal titration (AASM guidelines).
PAP Modalities
| Modality | Mechanism | Indications |
|---|---|---|
| CPAP | Single fixed pressure throughout inspiration and expiration | First-line for OSA |
| Auto-CPAP (APAP) | Automatically adjusts pressure (typically 4-20 cm H2O range) based on flow limitation, snoring, and apnea/hypopnea detection algorithms | Alternative to in-lab titration for uncomplicated OSA; allows home titration. Not for CSA, OHS, CHF, or chronic opioid use |
| BiPAP (Bilevel PAP) | Higher inspiratory (IPAP) and lower expiratory (EPAP) pressures; pressure support = IPAP - EPAP | CPAP intolerance (need >15 cm H2O), comorbid hypoventilation, neuromuscular disease, OHS, central apneas unresponsive to CPAP |
| BiPAP-ST | Bilevel with backup rate — delivers mandatory breath if patient does not trigger within set interval | Central sleep apnea, OHS, hypoventilation syndromes, neuromuscular disease |
| ASV | Adaptive servo-ventilation: auto-adjusts pressure support to stabilize ventilation; provides variable IPAP based on recent minute ventilation | Treatment-emergent CSA, complex sleep apnea. CONTRAINDICATED in HFrEF with EF ≤45% (SERVE-HF) |
Compliance Definition
CMS defines CPAP adherence as usage of ≥4 hours per night on ≥70% of nights (≥21 of 30 consecutive days) during the initial 90-day compliance period. The device must be downloaded and data reviewed at 30 and 90 days. If compliance criteria are not met, CMS may not cover continued therapy. Research consistently shows that the greatest clinical benefit occurs with ≥6 hours nightly use, and a dose-response relationship exists between hours of use and improvement in sleepiness, blood pressure, and quality of life.
Interface Selection
| Interface | Pros | Cons |
|---|---|---|
| Nasal mask | Good seal, lower pressures needed, allows speaking/oral intake | Ineffective with chronic mouth breathing; mouth leak |
| Nasal pillows | Minimal contact, least claustrophobic, good for active sleepers | May cause nasal irritation at higher pressures; less effective >15 cm H2O |
| Full-face mask | Covers nose and mouth; essential for obligate mouth breathers | More leak-prone, claustrophobic, higher pressure requirements, increased aerophagia, contraindicated in high aspiration risk |
| Oral mask | Alternative for nasal obstruction | Limited availability, niche use |
Troubleshooting Common CPAP Problems
Aerophagia (air swallowing): Lower pressure if possible; switch to BiPAP (lower EPAP); EPR/pressure relief feature; avoid eating close to bedtime. If persistent, evaluate for high-pressure requirement and consider surgical adjuncts to reduce Pcrit.
Mask leak: Refit mask, try different size/type, adjust headgear tension, ensure proper facial hair management, consider chinstrap for mouth leak with nasal masks.
Claustrophobia: Desensitization — wear mask during daytime while awake; start with nasal pillows (least claustrophobic); PAP-NAP (short supervised daytime nap with PAP for acclimatization); consider cognitive-behavioral techniques.
Nasal dryness/congestion: Heated humidification (standard of care); heated tubing to prevent rainout; nasal saline; intranasal corticosteroids; evaluate for septal deviation/polyps.
Pressure intolerance: EPR (expiratory pressure relief) or C-Flex; ramp feature; switch to APAP or BiPAP; PAP-NAP desensitization; ensure titration was not done exclusively in supine REM.
06 Surgical & Alternative Therapies for OSA
Non-PAP therapies are indicated when patients cannot tolerate or refuse CPAP, or as adjuncts to reduce the required CPAP pressure. Selection depends on the anatomic site of obstruction, OSA severity, and patient comorbidities.
Uvulopalatopharyngoplasty (UPPP)
The most commonly performed surgical procedure for OSA. Removes the uvula, redundant soft palate tissue, and tonsils (if present). Success rate: approximately 40-50% (defined as ≥50% reduction in AHI to <20). Best candidates: tonsil hypertrophy, Friedman stage I (large tonsils, low Mallampati), and retropalatal obstruction confirmed by drug-induced sleep endoscopy (DISE). Post-operative pain is significant (7-14 days). Does not address tongue-base or lateral wall collapse.
Maxillomandibular Advancement (MMA)
Advances the maxilla and mandible forward by 10-12 mm via Le Fort I osteotomy and bilateral sagittal split osteotomy, enlarging the entire posterior airway space. Success rate: 75-100% — the highest of any single surgical procedure for OSA. Indicated for moderate-to-severe OSA that has failed other treatments or in patients with retrognathia. Significant surgery with 4-6 week recovery, potential complications include malocclusion, numbness (inferior alveolar nerve), and cosmetic changes.
Hypoglossal Nerve Stimulation (Inspire)
1. Age ≥18 years
2. Moderate-to-severe OSA (AHI 15-65)
3. Unable to tolerate or benefit from CPAP
4. BMI ≤35 kg/m2 (some newer data support up to 40)
5. No complete concentric collapse at the velum on DISE (CCC is a contraindication — the stimulator advances the tongue but cannot open a circumferentially collapsed palate)
6. Central + mixed apneas must be <25% of total events
Mechanism: Implanted pulse generator stimulates the hypoglossal nerve during inspiration, protruding the tongue and stiffening the airway. Activated by the patient at bedtime with a remote. STAR trial showed median AHI reduction from 29.3 to 9.0 at 12 months.
Oral Appliance Therapy — Mandibular Advancement Device (MAD)
Custom-fabricated oral appliances protrude the mandible 6-10 mm during sleep, increasing posterior airway space. Indicated for mild-moderate OSA or severe OSA with CPAP intolerance. Custom devices (fitted by a dentist with sleep training) are preferred over over-the-counter models. Effectiveness: reduces AHI by 50-70% on average. Side effects: TMJ discomfort, excessive salivation, dental changes (bite shift) with long-term use. Follow-up PSG or HSAT to confirm efficacy is essential.
Positional Therapy
For patients with positional OSA (supine AHI ≥2x lateral AHI), devices that prevent supine sleep include positional belts, vibrotactile devices (Night Shift), and specialized pillows. Most effective in mild-moderate, non-obese, position-dependent OSA. Long-term adherence is the main limitation.
Weight Loss & Bariatric Surgery
A 10% weight loss can reduce AHI by approximately 26-50%. Bariatric surgery achieves dramatic AHI reductions (mean AHI decrease ~35-40 events/hr) but does not cure OSA in the majority — residual OSA requiring treatment persists in 50-60% post-bariatric surgery. PSG should be repeated post-weight loss to reassess PAP requirements.
Myofunctional Therapy
Oropharyngeal exercises targeting tongue, soft palate, and pharyngeal muscles. Meta-analyses show a ~50% reduction in AHI in mild-moderate OSA. Adjunctive role; adherence to daily exercises is the main barrier.
Drug-Induced Sleep Endoscopy (DISE)
DISE is a dynamic evaluation of upper airway collapse under sedation (typically propofol or midazolam), simulating the conditions of natural sleep. A flexible nasopharyngoscope is passed while the patient is sedated to directly visualize the site(s), pattern, and severity of airway obstruction. The VOTE classification grades collapse at four levels: Velum (palate), Oropharyngeal lateral walls (tonsils), Tongue base, and Epiglottis. Each level is graded for degree (no obstruction, partial, complete) and pattern (anterior-posterior, lateral, concentric). DISE is used to guide surgical site selection and to determine eligibility for hypoglossal nerve stimulation (complete concentric collapse at the velum is a contraindication for Inspire therapy). Limitations: sedation may not perfectly replicate natural sleep, and inter-rater reliability varies.
07 Central Sleep Apnea
Central sleep apnea (CSA) is characterized by repetitive cessation of airflow due to transient loss of respiratory drive from the brainstem, without upper airway obstruction. Respiratory effort is absent during events (flat thoracic and abdominal RIP belts on PSG).
Cheyne-Stokes Respiration (CSR)
A distinctive crescendo-decrescendo pattern of tidal volume alternating with central apneas or hypopneas. The cycle length is typically 45-90 seconds. Strongly associated with heart failure with reduced ejection fraction (HFrEF) — present in 30-50% of patients with LVEF ≤40%. Pathophysiology: prolonged circulation time delays feedback from PaCO2 changes reaching central chemoreceptors, causing overshoot and undershoot of ventilation. The apneic threshold for CO2 is narrowed. CSR carries independent prognostic significance in heart failure — its presence is associated with increased mortality.
Subtypes of Central Sleep Apnea
| Subtype | Mechanism | Key Features |
|---|---|---|
| CSA with CSR | High loop gain from prolonged circulation time and enhanced chemosensitivity | HFrEF, stroke. Crescendo-decrescendo pattern. ≥3 consecutive cycles, cycle length ≥40 sec |
| Treatment-emergent CSA | Central apneas emerge or persist after obstructive events are eliminated by CPAP (formerly "complex sleep apnea") | Occurs in ~5-15% of OSA patients on CPAP. Often resolves spontaneously within weeks to months. If persistent, consider ASV (if EF >45%) |
| High-altitude CSA | Hyperventilation-induced hypocapnia at altitude (>2500 m) drives PaCO2 below apneic threshold | Periodic breathing pattern. Resolves with descent or acclimatization. Acetazolamide prophylaxis |
| Opioid-induced CSA | Opioids depress medullary respiratory centers; reduce chemosensitivity to CO2; also cause ataxic/Biot's breathing | Dose-dependent; morphine equivalent dose ≥200 mg/day strongly associated. Irregular breathing pattern rather than typical crescendo-decrescendo. ASV or BiPAP-ST may be needed |
| Idiopathic CSA | Elevated hypercapnic ventilatory response with low waking PaCO2 | Rare; PaCO2 during wakefulness is typically ≤40 mmHg |
Treatment of CSA
Optimize underlying condition: Heart failure optimization (guideline-directed medical therapy, CRT if indicated) is the most important intervention for CSR-CSA. Opioid dose reduction for opioid-induced CSA.
CPAP: First-line for CSA associated with CHF; can reduce AHI by ~50%. The CANPAP trial showed no mortality benefit but improved EF and 6MWD in CPAP-responsive patients.
ASV: Highly effective at eliminating CSA events. However, the SERVE-HF trial demonstrated increased cardiovascular mortality (HR 1.34) and all-cause mortality (HR 1.28) with ASV in patients with HFrEF (LVEF ≤45%) and predominant CSA. ASV is absolutely contraindicated in symptomatic HFrEF with EF ≤45%. ASV remains appropriate for treatment-emergent CSA and CSA without HFrEF (EF >45%).
Phrenic nerve stimulation (remede System): Transvenous implantable device that stimulates the phrenic nerve to maintain diaphragmatic contraction during sleep, preventing central apneas. FDA-approved for moderate-to-severe CSA in adults. Implanted like a pacemaker. Alternative for patients ineligible for ASV.
Supplemental oxygen: Low-flow oxygen (2-4 L/min) can reduce CSA severity, particularly CSR in heart failure, by attenuating peripheral chemoreceptor sensitivity.
Acetazolamide: Induces mild metabolic acidosis, lowering the apneic threshold. Useful for high-altitude CSA (250 mg BID) and idiopathic CSA.
Theophylline: Stimulates central respiratory drive and reduces CSA severity in heart failure (level of evidence lower than other therapies). Narrow therapeutic window requires monitoring of serum levels. Rarely used as primary therapy but may be adjunctive.
Step 1: Optimize underlying condition (GDMT for heart failure, opioid dose reduction, descent from altitude)
Step 2: Determine EF — if ≤45%, ASV is contraindicated
Step 3: Trial of CPAP (first-line for CSA-CHF). Reassess AHI after 1-3 months
Step 4: If CPAP fails and EF >45%: consider ASV. If EF ≤45%: consider BiPAP-ST, supplemental O2, or phrenic nerve stimulation
Step 5: For treatment-emergent CSA on CPAP: observe for 3 months (many resolve spontaneously). If persistent: ASV (if EF >45%) or BiPAP-ST
08 Obesity Hypoventilation Syndrome
Obesity hypoventilation syndrome (OHS), also known as Pickwickian syndrome, is defined by the triad of: (1) BMI ≥30 kg/m2, (2) awake daytime hypercapnia (PaCO2 ≥45 mmHg), and (3) no alternative explanation for hypoventilation (excluding neuromuscular disease, severe obstructive lung disease, chest wall disorders, hypothyroidism, or central hypoventilation syndromes).
Overlap with OSA
Approximately 90% of OHS patients have coexistent OSA (AHI ≥5). Roughly 10-20% of patients with severe OSA (AHI ≥30) have OHS. OHS should be suspected in any obese patient with severe OSA, especially with BMI ≥40, awake SpO2 <94%, or serum bicarbonate ≥27 mEq/L. A useful bedside screening approach: check a venous basic metabolic panel — an elevated bicarbonate in an obese, sleepy patient with known or suspected OSA should prompt an ABG.
Epidemiology & Outcomes
OHS prevalence is 0.3-0.4% in the general population but rises to 8-20% among patients referred to sleep laboratories and up to 31% among hospitalized patients with BMI ≥35. Compared to eucapnic obese patients with OSA, OHS patients have significantly higher rates of pulmonary hypertension (50-88% vs. 10-15%), cor pulmonale, polycythemia, and healthcare utilization. Untreated OHS has a 5-year mortality rate estimated at 23-46%, which drops dramatically with PAP therapy and weight management. Most OHS patients are diagnosed during hospitalization for acute hypercapnic respiratory failure rather than through outpatient evaluation, highlighting the need for better screening in primary care and sleep clinics.
Screening & Diagnosis
Serum bicarbonate is the most practical screening test: a venous bicarbonate ≥27 mEq/L in an obese patient with sleep-disordered breathing has a sensitivity of ~92% for OHS. An arterial blood gas (ABG) is required for definitive diagnosis — PaCO2 ≥45 mmHg while awake on room air. The ABG should be obtained in the morning after the patient has been awake and seated for ≥30 minutes. Nocturnal oximetry often shows a sawtooth pattern (cyclic desaturations from OSA) superimposed on a sustained low baseline SpO2 (from hypoventilation).
Treatment
PAP therapy is the cornerstone. For OHS with coexistent severe OSA: start with CPAP — approximately 50% of OHS patients will normalize daytime PaCO2 with CPAP alone. If CPAP fails to correct hypercapnia or the patient has predominantly hypoventilation without significant OSA: BiPAP-ST (bilevel with backup rate) targeting a tidal volume of 8-10 mL/kg ideal body weight is preferred, with typical settings of IPAP 16-24, EPAP 8-12, backup rate 12-16. Volume-assured pressure support (VAPS/AVAPS) modes auto-titrate IPAP to achieve target tidal volume.
Weight loss is the only curative treatment. Bariatric surgery should be considered in all eligible OHS patients (BMI ≥35 with comorbidity or ≥40). Bariatric surgery resolves OHS in the majority, with normalization of PaCO2 and reduction/elimination of PAP need.
Pathophysiology of OHS
Three mechanisms converge: (1) Increased mechanical load — obesity increases chest wall and abdominal mass, reducing chest wall compliance, functional residual capacity (FRC), and expiratory reserve volume (ERV). The diaphragm is splinted upward, reducing its excursion. (2) Blunted central respiratory drive — a subset of obese individuals have reduced ventilatory response to hypercapnia and hypoxemia, possibly related to leptin resistance (leptin normally stimulates ventilation). (3) Repetitive nocturnal hypoxemia and hypercapnia from coexistent OSA — leads to renal bicarbonate retention (metabolic compensation), which raises the apneic threshold and perpetuates daytime hypercapnia. The elevated serum bicarbonate blunts the CO2-mediated respiratory drive even further.
Distinguishing OHS from COPD-Obesity Overlap
Both present with hypercapnia in obese patients, but the distinction matters for treatment. OHS patients have normal or near-normal spirometry (FEV1/FVC ratio preserved); COPD patients have airflow obstruction (FEV1/FVC <0.70). Some patients have both (overlap syndrome). In pure OHS, the A-a gradient may be mildly elevated but diffusion capacity (DLCO) is preserved — unlike COPD where DLCO is reduced (emphysema) or normal (chronic bronchitis). Spirometry and DLCO should be obtained in all suspected OHS patients to exclude significant obstructive lung disease.
09 Insomnia Disorder
Insomnia disorder is a subjective complaint of difficulty initiating sleep, maintaining sleep, or early-morning awakening, associated with daytime impairment, occurring despite adequate opportunity and circumstances for sleep. It is the most common sleep complaint, affecting 10-15% of the general population chronically.
Diagnostic Criteria (ICSD-3 / DSM-5)
A. Difficulty initiating sleep, difficulty maintaining sleep, and/or waking up earlier than desired
B. Adequate opportunity and circumstances for sleep
C. Associated daytime impairment: fatigue, concentration difficulty, mood disturbance, social/vocational/academic dysfunction, daytime sleepiness, reduced energy/motivation, proneness to errors/accidents, concern about sleep
D. Sleep difficulty and daytime symptoms occur ≥3 times per week
E. Duration ≥3 months (chronic). Short-term insomnia: <3 months
F. Not better explained by another sleep disorder, mental disorder, medical condition, or substance/medication
Spielman 3P Model
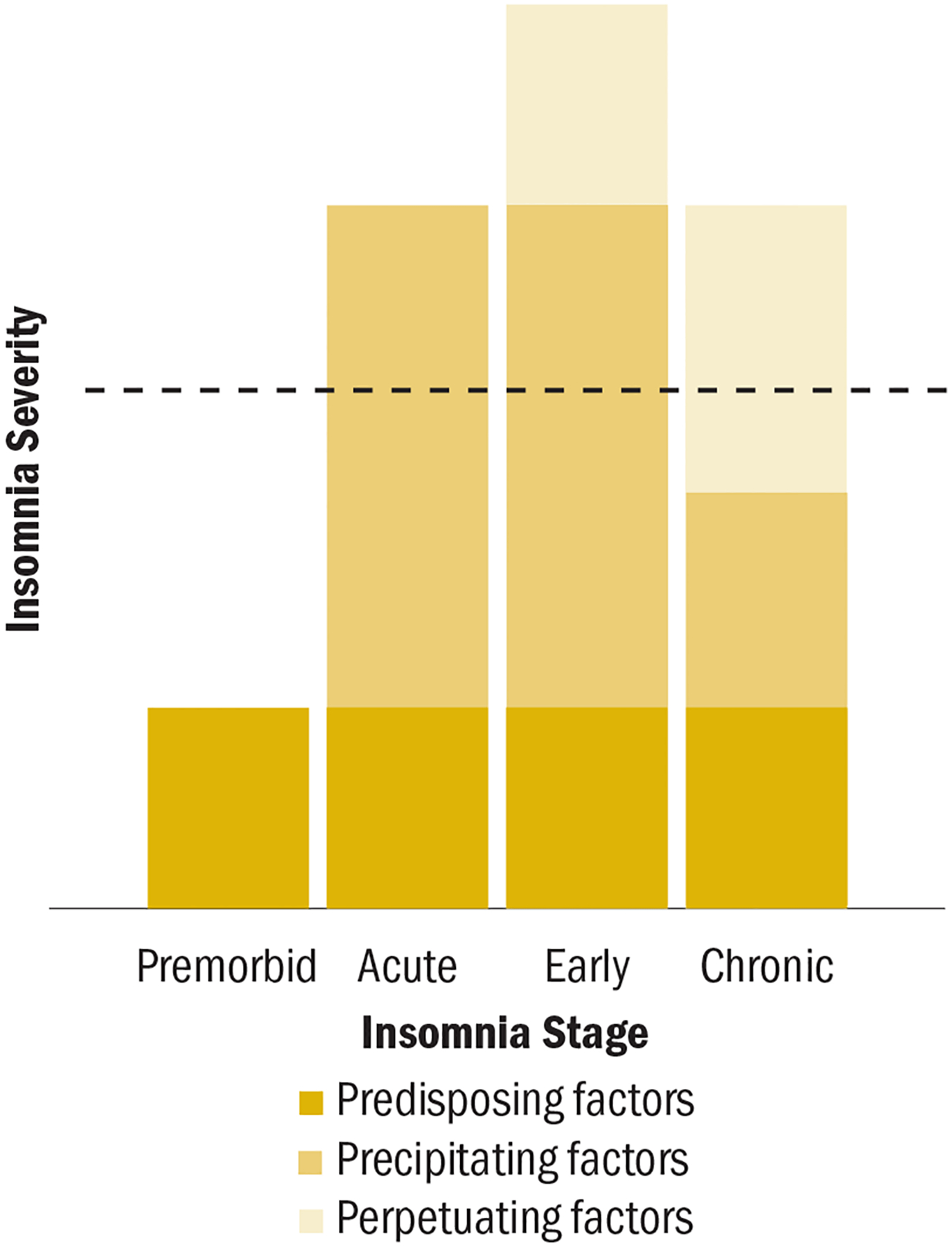
The 3P model is the dominant conceptual framework for understanding chronic insomnia:
Predisposing factors: Trait characteristics that lower the threshold for insomnia — genetic predisposition (hyperarousal phenotype, family history), personality traits (perfectionism, neuroticism, rumination tendency), female sex, older age.
Precipitating factors: Acute stressors that trigger insomnia onset — medical illness, pain, psychiatric episode, bereavement, job loss, life transition, schedule change, medication initiation.
Perpetuating factors: Maladaptive behaviors and cognitions that maintain insomnia after the precipitating event resolves — excessive time in bed, irregular schedule, daytime napping, clock-watching, catastrophizing about sleep consequences, conditioned arousal in the bedroom, compensatory caffeine or alcohol use. CBT-I specifically targets these perpetuating factors.
Comorbid Insomnia
Insomnia frequently coexists with and is bidirectional with psychiatric disorders (depression — present in 80% of MDD; anxiety; PTSD), chronic pain conditions, cardiovascular disease, and neurologic disorders. The term "comorbid insomnia" has replaced "secondary insomnia" — insomnia should be treated as a disorder in its own right even when a comorbid condition is identified, as it independently worsens outcomes and treating insomnia improves the comorbid condition.
Insomnia Severity Index (ISI)
Rate the severity of each item over the past 2 weeks (0 = none, 1 = mild, 2 = moderate, 3 = severe, 4 = very severe):
1. Difficulty falling asleep
2. Difficulty staying asleep
3. Problem waking up too early
4. How satisfied/dissatisfied are you with your current sleep pattern? (0 = very satisfied to 4 = very dissatisfied)
5. How noticeable to others do you think your sleep problem is in terms of impairing the quality of your life?
6. How worried/distressed are you about your current sleep problem?
7. To what extent do you consider your sleep problem to interfere with your daily functioning (fatigue, mood, concentration, memory, mood)?
Scoring (0-28): 0-7 = no clinically significant insomnia; 8-14 = subthreshold insomnia; 15-21 = clinical insomnia (moderate); 22-28 = clinical insomnia (severe). Clinically meaningful change: ≥6-point reduction.
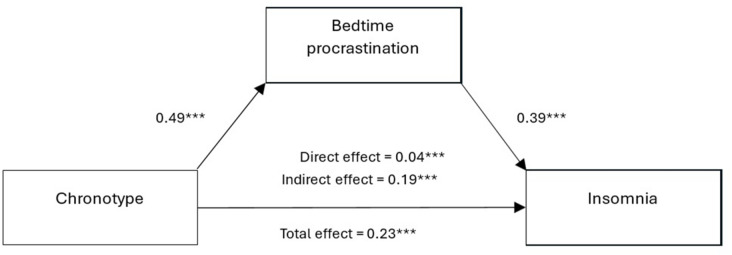
Sleep Diary
A 2-week sleep diary is the minimum recommended before initiating treatment. Key variables recorded nightly: bedtime, time of attempted sleep onset, estimated sleep latency, number and duration of awakenings, final wake time, rise time, naps (time and duration), caffeine/alcohol/medication use, and subjective sleep quality rating. The diary provides data for sleep efficiency calculation and is essential for sleep restriction therapy prescriptions in CBT-I.
10 CBT-I — Cognitive Behavioral Therapy for Insomnia
CBT-I is the first-line treatment for chronic insomnia disorder, recommended before pharmacotherapy by the AASM, ACP, and AAFP. It is a structured, typically 6-8 session intervention with durable effects that persist after treatment ends — unlike medications, which work only while being taken and often lead to rebound insomnia upon discontinuation.
Component 1: Sleep Restriction Therapy (SRT)
The most potent behavioral component. Rationale: insomnia patients spend excessive time in bed, creating a mismatch between time in bed and actual sleep time (poor sleep efficiency). SRT reduces time in bed to match actual total sleep time, consolidating sleep and increasing homeostatic sleep drive.
Protocol: Calculate average total sleep time from 2-week sleep diary (e.g., 5.5 hours). Set time in bed (TIB) = total sleep time, with a minimum floor of 5 hours (never prescribe <5 hours). Fix the wake time (anchored to schedule obligations) and calculate bedtime (e.g., wake at 6:00 AM → bedtime 12:30 AM). Each week, calculate sleep efficiency (TST/TIB x 100%): if SE ≥90%, advance bedtime by 15-20 minutes; if SE 85-90%, maintain; if SE <85%, delay bedtime by 15-20 minutes. Continue weekly adjustments until target sleep duration is achieved with SE ≥85%.
Component 2: Stimulus Control Therapy
Aims to re-associate the bed/bedroom with sleep rather than wakefulness. The 8 rules of stimulus control:
1. Go to bed only when sleepy.
2. Use the bed only for sleep and sex — no reading, watching TV, eating, or worrying in bed.
3. If unable to fall asleep within approximately 15-20 minutes, get up and go to another room.
4. Engage in a quiet, non-stimulating activity in dim light until sleepy, then return to bed.
5. Repeat step 3-4 as many times as necessary throughout the night.
6. Set an alarm and get up at the same time every morning, regardless of how much sleep was obtained.
7. Do not nap during the day.
8. Avoid clock-watching (turn clocks away from view).
Component 3: Cognitive Restructuring
Addresses maladaptive beliefs and catastrophic thinking about sleep. Common cognitive distortions in insomnia: "I must get 8 hours or I cannot function," "If I don't sleep tonight, tomorrow will be terrible," "My insomnia is going to cause serious health problems." Techniques: Socratic questioning, thought records, behavioral experiments (testing the accuracy of catastrophic predictions), and psychoeducation about normal sleep variability and individual sleep needs.
Component 4: Sleep Hygiene Education
All components of comprehensive sleep hygiene:
1. Maintain a consistent sleep-wake schedule (including weekends)
2. Create an optimal sleep environment: dark, quiet, cool (65-68 degrees F / 18-20 degrees C)
3. Limit caffeine — avoid within 6-8 hours of bedtime (caffeine half-life ~5 hours)
4. Limit alcohol — disrupts second-half sleep architecture, increases awakenings
5. Avoid nicotine close to bedtime (stimulant)
6. Exercise regularly (≥30 min moderate activity), but complete vigorous exercise ≥4 hours before bedtime
7. Avoid large meals within 2-3 hours of bedtime
8. Limit fluid intake in the evening to reduce nocturia
9. Create a relaxing pre-sleep routine (30-60 minutes wind-down)
10. Minimize blue light exposure in the evening (screens, LED lighting)
11. Reserve the bedroom for sleep and intimacy only
12. Manage stress/worry before bedtime — use a "worry journal" to offload concerns before entering the bedroom
Component 5: Relaxation Training
Progressive muscle relaxation (PMR), diaphragmatic breathing, guided imagery, and autogenic training. PMR involves systematic tensing and releasing of muscle groups from feet to head. These techniques reduce physiologic hyperarousal and are particularly useful for patients with somatic tension.
Component 6: Paradoxical Intention
The patient is instructed to lie in bed and try to stay awake with eyes open, rather than trying to fall asleep. This removes the performance anxiety and effort associated with trying to sleep, paradoxically facilitating sleep onset. Most useful for sleep-onset insomnia with prominent sleep anxiety.
Digital CBT-I
Somryst (Pear Therapeutics) was the first FDA-authorized prescription digital therapeutic for chronic insomnia, delivering CBT-I through a smartphone app over 9 weeks. Other digital CBT-I platforms include Insomnia Coach (VA), Sleepio, and CBT-i Coach. Digital CBT-I improves access to this evidence-based treatment, given the shortage of trained CBT-I therapists. Meta-analyses show moderate-to-large effect sizes, though generally smaller than in-person CBT-I.

Efficacy Data — CBT-I vs. Pharmacotherapy
Multiple RCTs and meta-analyses demonstrate that CBT-I produces comparable short-term improvement in sleep onset latency and sleep efficiency compared to hypnotic medications (Z-drugs, benzodiazepines). However, the long-term durability of CBT-I is markedly superior: benefits persist for ≥12-24 months after treatment completion, whereas medication effects cease upon discontinuation and rebound insomnia commonly occurs. Combined therapy (CBT-I + medication) may be useful in the acute phase, followed by medication taper as behavioral changes consolidate. The ACP 2016 clinical practice guideline recommends CBT-I as the initial treatment for all adults with chronic insomnia, with pharmacotherapy reserved for patients who do not respond to CBT-I alone.
11 Pharmacotherapy for Insomnia
Pharmacotherapy is second-line to CBT-I and should be considered when CBT-I is unavailable, inadequate, or while awaiting its effects. The choice of agent depends on the insomnia phenotype (sleep-onset vs. maintenance vs. both), comorbidities, abuse potential, and next-day impairment risk.
Drug Classes & Key Agents
| Drug | Class | Dose | Onset | Duration | Abuse Potential | Next-Day Impairment |
|---|---|---|---|---|---|---|
| Zolpidem IR | BzRA (Z-drug) | 5 mg (women), 5-10 mg (men) | 15-30 min | Short (2-3 h) | Moderate (Schedule IV) | Yes — FDA boxed warning for women at 10 mg; complex sleep behaviors |
| Zolpidem ER | BzRA | 6.25 mg (women), 6.25-12.5 mg (men) | 15-30 min | Intermediate (6-8 h) | Moderate | Higher than IR |
| Zaleplon | BzRA | 5-10 mg (reduce to 5 mg in elderly) | 15-30 min | Ultra-short (1 h) | Moderate | Minimal — can be dosed for middle-of-night awakening if ≥4 h before rising |
| Eszopiclone | BzRA | 1 mg (elderly), 2-3 mg (adults) | 30 min | Intermediate (6-8 h) | Moderate | Moderate; metallic taste common |
| Suvorexant | DORA | 10-20 mg | 30 min | Intermediate (6-7 h) | Low (Schedule IV) | Low-moderate; may cause next-day somnolence at 20 mg |
| Lemborexant | DORA | 5-10 mg | 30 min | Intermediate (6-8 h) | Low (Schedule IV) | Low; no dose reduction required in elderly |
| Ramelteon | MT1/MT2 agonist | 8 mg | 30-45 min | Short (1-2.5 h) | None (unscheduled) | Minimal |
| Doxepin | H1 antagonist (low-dose TCA) | 3 mg or 6 mg | 30-60 min | Intermediate (selective H1 at this dose) | None (unscheduled) | Minimal at 3-6 mg; excellent for sleep maintenance insomnia in elderly |
| Trazodone | 5-HT2A antagonist (off-label) | 25-100 mg | 30-60 min | Variable (3-6 h) | None | Variable; morning sedation, orthostatic hypotension, priapism (rare) |
| Gabapentin | Alpha-2-delta ligand (off-label) | 100-600 mg | 60 min | Variable | Low | Variable; useful when comorbid pain, RLS, or alcohol use disorder |
Selection by Insomnia Phenotype
Sleep-onset insomnia: Ramelteon (non-habit-forming, safe in elderly), zaleplon, zolpidem IR, suvorexant.
Sleep-maintenance insomnia: Low-dose doxepin (3-6 mg — FDA-approved for maintenance insomnia), suvorexant, lemborexant, eszopiclone, zolpidem ER.
Both onset and maintenance: DORAs (suvorexant, lemborexant), eszopiclone, zolpidem ER.
Elderly patients: Doxepin 3-6 mg (Beers list-safe at low dose), ramelteon, or DORAs preferred. Avoid benzodiazepines (falls, cognitive impairment, delirium) and high-dose Z-drugs.
Special Populations
Pregnancy: Insomnia affects 50-80% of pregnant women. Non-pharmacologic approaches (CBT-I, sleep hygiene) are first-line. Pharmacotherapy data is limited. Doxepin is Category C. Diphenhydramine and doxylamine (Category B) are commonly used though evidence for efficacy in insomnia is weak. Benzodiazepines and Z-drugs should generally be avoided (particularly in the first trimester — possible association with cleft palate, and neonatal withdrawal/respiratory depression if used near delivery). Suvorexant and lemborexant lack adequate pregnancy safety data.
Hepatic impairment: Most hypnotics are hepatically metabolized. Dose reduction required for eszopiclone, suvorexant, and lemborexant. Ramelteon is contraindicated in severe hepatic impairment (high first-pass metabolism). Zolpidem should be used at reduced doses.
Renal impairment: Gabapentin requires renal dose adjustment. Most Z-drugs and DORAs do not require significant adjustment. Solriamfetol requires dose reduction in renal impairment (avoid if eGFR <15).
Discontinuation Strategies
For patients on chronic hypnotic therapy who wish to taper: (1) stabilize with CBT-I first; (2) gradual dose reduction over 4-8 weeks (reduce dose by ~25% every 1-2 weeks); (3) intermittent dosing schedule (3-5 nights/week) as an intermediate step; (4) switch to a short-acting agent if on a long-acting benzodiazepine; (5) ramelteon can be used as a bridging agent during taper. Rebound insomnia (1-3 nights of worsened sleep) is expected and self-limited.
12 Narcolepsy Type 1 & 2
Narcolepsy is a chronic neurological disorder of central hypersomnolence caused by deficient orexin (hypocretin) signaling. It is classified into Type 1 (with cataplexy and/or low CSF orexin) and Type 2 (without cataplexy, normal CSF orexin).
Narcolepsy Type 1 — Orexin/Hypocretin Deficiency
Caused by autoimmune-mediated destruction of orexin-producing neurons in the lateral hypothalamus. Only ~70,000 orexin neurons exist, and >90% must be lost before clinical narcolepsy manifests. CSF orexin-1 (hypocretin-1) levels <110 pg/mL (or <1/3 of normal mean) are diagnostic and highly specific (>99%). HLA DQB1*06:02 is present in 98% of narcolepsy type 1 patients (vs. 25% of the general population) — it is necessary but not sufficient (low positive predictive value). Onset typically in the second decade (peak ages 10-25).
The Narcolepsy Tetrad
1. Excessive daytime sleepiness (EDS): The cardinal and earliest symptom, present in 100%. Irresistible sleep attacks, often at inappropriate times. Brief naps are refreshing (in contrast to idiopathic hypersomnia).
2. Cataplexy: Pathognomonic for narcolepsy type 1. Sudden, transient loss of skeletal muscle tone triggered by strong emotions (laughter, surprise, anger). Consciousness is preserved. Duration: seconds to minutes. Ranges from subtle (jaw drop, head nod, knee buckling) to complete postural collapse. Represents intrusion of REM atonia into wakefulness. Present in ~70% of type 1 at diagnosis.
3. Sleep paralysis: Inability to move or speak during sleep-wake transitions (hypnagogic or hypnopompic). Lasts seconds to minutes. Occurs in ~25-50% of narcolepsy patients, but also in 5-8% of the general population.
4. Hypnagogic/hypnopompic hallucinations: Vivid, often frightening, dream-like visual or auditory experiences at sleep onset (hypnagogic) or upon awakening (hypnopompic). Represent intrusion of REM-associated dreaming into wakefulness.
MSLT Diagnostic Criteria
The Multiple Sleep Latency Test (MSLT) is performed the day after a diagnostic PSG (which must show ≥6 hours of sleep and exclude other causes of sleepiness). The patient undergoes 5 nap opportunities at 2-hour intervals (e.g., 9, 11, 1, 3, 5). Each trial lasts 20 minutes; if sleep occurs, the trial continues for 15 additional minutes to assess for REM onset. MSLT criteria for narcolepsy:
1. Mean sleep latency ≤8 minutes (average across all naps)
2. ≥2 sleep-onset REM periods (SOREMPs) — REM occurring within 15 minutes of sleep onset
Note: A SOREMP on the preceding night's PSG can replace one of the MSLT SOREMPs. Medications that affect REM (antidepressants, stimulants) must be withdrawn for ≥2 weeks (5 half-lives for fluoxetine) before MSLT. Insufficient sleep must be excluded (sleep diary and/or actigraphy for ≥1 week before testing).
Narcolepsy Type 2
Meets MSLT criteria (mean latency ≤8 min + ≥2 SOREMPs) but cataplexy is absent and CSF orexin-1 levels are normal (>110 pg/mL) or not measured. Some type 2 patients may eventually develop cataplexy and be reclassified as type 1.
Narcolepsy — Associated Conditions & Comorbidities
Narcolepsy carries substantial psychiatric comorbidity: depression (25-50%), anxiety (15-30%), and ADHD-like symptoms. Obesity is common in narcolepsy type 1 (BMI ~2-3 kg/m2 higher than matched controls) and appears related to orexin deficiency rather than inactivity — orexin regulates appetite and energy metabolism. OSA coexists in approximately 25-30% of narcolepsy patients, partly related to obesity, and can confound the MSLT if not treated beforehand. RBD is also more prevalent in narcolepsy type 1 (~30-60%), though in this context it does not carry the same neurodegenerative risk as isolated RBD in the elderly.
Narcolepsy — Practical Considerations
Driving: Patients with untreated narcolepsy have a 3-7x higher rate of motor vehicle accidents. Most states do not mandate reporting, but physicians should counsel patients about driving risk and document the discussion. Treatment that achieves adequate alertness (ideally confirmed by MWT mean latency ≥40 min) is needed before resuming driving.
Perioperative management: Continue stimulant and anticataplectic medications perioperatively when possible. Abrupt discontinuation of anticataplectic agents (SSRIs, SNRIs, sodium oxybate) can precipitate status cataplecticus. Inform the anesthesia team of narcolepsy diagnosis and current medications. Sodium oxybate should be held the night before morning surgery (short half-life allows this). Postoperative sedation from anesthesia may be prolonged or atypical.
13 Treatment of Narcolepsy
Treatment targets the two core symptom domains: excessive daytime sleepiness (wake-promoting agents) and cataplexy/REM-related phenomena (anticataplectic agents). Behavioral measures — scheduled naps, regular sleep-wake schedules, driving safety — are essential adjuncts.
Sodium Oxybate (Xyrem) & Low-Sodium Oxybate (Xywav)
Sodium oxybate is the only FDA-approved treatment that addresses both EDS and cataplexy. It is the sodium salt of gamma-hydroxybutyrate (GHB). Mechanism: GABA-B receptor agonist that consolidates nocturnal sleep, increases slow-wave sleep, and suppresses cataplexy.
Dosing: Start 4.5 g/night (2.25 g at bedtime, 2.25 g 2.5-4 hours later). Titrate by 1.5 g/night at weekly intervals to max 9 g/night. Taken in 2 divided nightly doses (liquid, mixed in water). Must be taken in bed — onset of action 5-15 minutes.
REMS (Risk Evaluation and Mitigation Strategy): Required due to abuse potential (Schedule III). Central pharmacy distribution only. Prescriber, pharmacy, and patient must be enrolled in the REMS program.
Xywav (low-sodium oxybate) — same active compound but with 92% less sodium (calcium, magnesium, potassium oxybates). Important for patients with sodium-sensitive conditions (hypertension, heart failure). Same dosing and titration as Xyrem.
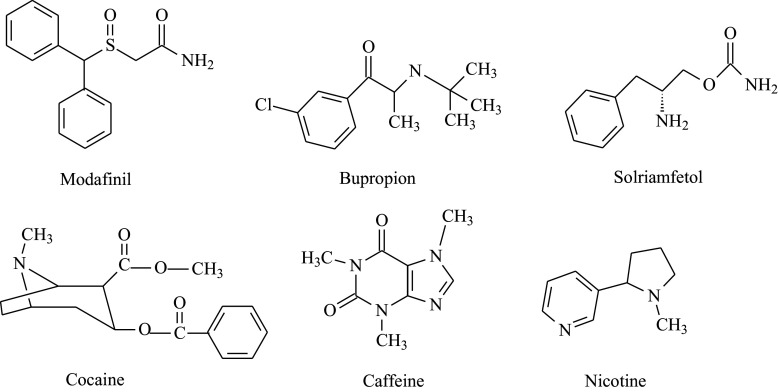
Wake-Promoting Agents
| Drug | Mechanism | Dose | Notes |
|---|---|---|---|
| Solriamfetol | Dopamine/norepinephrine reuptake inhibitor | 75-150 mg QAM | FDA-approved for EDS in narcolepsy and OSA. No abuse potential concerns comparable to amphetamines. Avoid in uncontrolled hypertension |
| Pitolisant | Histamine H3 receptor inverse agonist/antagonist — increases histamine release in the brain | 8.9-35.6 mg QAM | First-in-class, non-scheduled. Also reduces cataplexy. Unique mechanism. QT prolongation risk |
| Modafinil | Uncertain; involves dopamine reuptake inhibition | 100-200 mg QAM (or split QAM + noon) | Schedule IV. First-line for many clinicians. Does not treat cataplexy. Risk of SJS with HLA-B*15:02. Reduces OCP efficacy |
| Armodafinil | R-enantiomer of modafinil; longer half-life | 150-250 mg QAM | Schedule IV. Longer duration than modafinil. Same caveats |
| Methylphenidate | Dopamine/NE reuptake inhibitor | 10-60 mg/day in divided doses | Schedule II. Older agent; effective but higher abuse potential |
| Amphetamine/dextroamphetamine | Dopamine/NE releaser and reuptake inhibitor | 5-60 mg/day in divided doses | Schedule II. Most potent wake-promoting agent. Reserve for refractory cases |
Anticataplectic Agents
Sodium oxybate/Xywav: Most effective — reduces cataplexy attacks by 65-90%.
Venlafaxine: Most commonly used antidepressant for cataplexy (37.5-300 mg/day). Potent NE and serotonin reuptake inhibition suppresses REM and its manifestations. Not FDA-approved for this indication but widely used.
Fluoxetine: 20-60 mg/day. Also suppresses REM and cataplexy. Other SSRIs/SNRIs are used similarly off-label.
Pitolisant: FDA-approved for cataplexy. Reduces attacks by ~60-75%.
14 Idiopathic Hypersomnia & Kleine-Levin Syndrome
Idiopathic Hypersomnia (IH)
Idiopathic hypersomnia is a central disorder of hypersomnolence characterized by excessive daytime sleepiness despite normal or prolonged total sleep time, without cataplexy or the MSLT criteria for narcolepsy. Key distinguishing features:
Prolonged nocturnal sleep (often ≥10 hours, sometimes up to 14-16 hours) — yet the patient remains unrefreshed.
Sleep drunkenness (severe sleep inertia): Prolonged, disabling difficulty transitioning from sleep to wake. Patients may require multiple alarms, are confused and cognitively impaired upon awakening for 30-120+ minutes, and may exhibit automatic behaviors during this period.
Naps are long and unrefreshing — in contrast to narcolepsy, where brief naps are characteristically refreshing.
MSLT Criteria for IH
Mean sleep latency ≤8 minutes with <2 SOREMPs (including the preceding night's PSG). If the mean latency is >8 minutes but ≤14 minutes, the diagnosis can still be made if nocturnal PSG shows total sleep time ≥660 minutes (11 hours). Actigraphy over ≥7 days may demonstrate consistently prolonged sleep periods.
Treatment of IH
Same wake-promoting agents used in narcolepsy: modafinil, armodafinil, solriamfetol, methylphenidate, amphetamines. Response is generally less robust than in narcolepsy. Low-sodium oxybate (Xywav) received FDA approval for IH in 2021 — the first specifically approved treatment. Clarithromycin (off-label) has shown benefit in some studies, possibly through negative allosteric modulation of GABA-A receptors. Flumazenil (off-label) has also been investigated.
The hallmark sleep inertia of IH is particularly treatment-resistant. Strategies include multiple sequential alarms, bright light alarm clocks, scheduled morning activities that demand engagement, and in some cases stimulant dosing before full awakening (a family member administers the medication and the patient returns to sleep briefly before it takes effect). Unlike narcolepsy, IH patients do not benefit from scheduled naps — naps are typically prolonged and unrefreshing.
Kleine-Levin Syndrome (KLS)
Kleine-Levin syndrome is a rare (1-5 per million) recurrent hypersomnia characterized by episodes lasting days to weeks of:
1. Hypersomnolence: Sleeping 16-20+ hours per day
2. Cognitive disturbance: Confusion, derealization, apathy, amnestic episodes
3. Behavioral changes: Hyperphagia (compulsive eating), hypersexuality (inappropriate sexual behavior), irritability, childlike behavior
Between episodes, patients are entirely normal — neurologically, cognitively, and behaviorally. Predominantly affects adolescent males (4:1 male:female). Episodes recur every few weeks to months, with a median of 20 episodes over 8 years before spontaneous resolution. MRI is normal; interictal EEG may show diffuse slowing during episodes.
Treatment: Lithium is the best-studied prophylactic agent, reducing episode frequency and duration in approximately 40% of patients. Stimulants (modafinil, amphetamines) have limited efficacy during episodes for wakefulness. Valproate may also be considered for prophylaxis.
15 Circadian Rhythm Sleep-Wake Disorders
Circadian rhythm sleep-wake disorders (CRSWDs) arise from a mismatch between the endogenous circadian pacemaker (SCN) and the desired or required sleep-wake schedule, or from dysfunction of the circadian system itself. All subtypes require documentation by sleep diary and/or actigraphy for ≥7 days (preferably 14 days) and clinically significant distress or impairment.
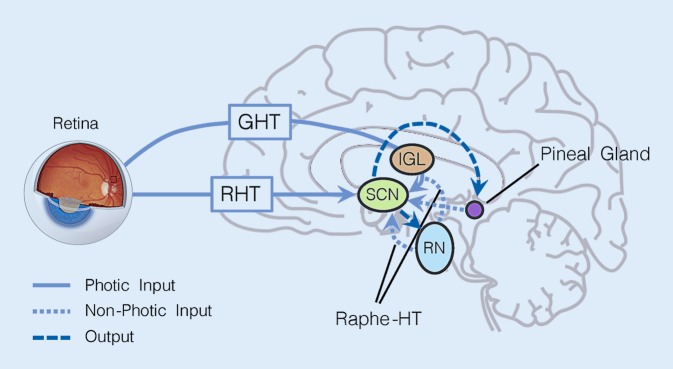
Delayed Sleep-Wake Phase Disorder (DSWPD)
The most common CRSWD, particularly in adolescents and young adults (prevalence ~7-16% in this age group). The endogenous circadian phase is delayed relative to the conventional sleep period, typically by 2-6 hours. Patients ("night owls") have difficulty falling asleep before 2:00-6:00 AM and difficulty waking before 10:00 AM-2:00 PM. When allowed to sleep on their preferred schedule, sleep quality and duration are normal. The problem arises when social or occupational obligations demand an earlier schedule, causing chronic sleep deprivation, excessive daytime sleepiness, and difficulty with morning functioning. DLMO is delayed. Treatment: strategic evening light avoidance, morning bright light therapy, and low-dose melatonin (0.5-3 mg) administered 3-6 hours before DLMO (typically early-to-mid evening).
Advanced Sleep-Wake Phase Disorder (ASWPD)
The converse of DSWPD — the circadian phase is advanced, with sleep onset at 6:00-9:00 PM and spontaneous awakening at 2:00-5:00 AM. More common in the elderly. Often familial — associated with mutations in clock genes (PER2, CK1-delta). Treatment: evening bright light therapy (7:00-9:00 PM) to delay the phase; avoid morning light exposure. Melatonin upon early morning awakening (to promote continued sleep) is sometimes used but less well-studied.
Non-24-Hour Sleep-Wake Rhythm Disorder
The endogenous circadian period runs slightly longer than 24 hours (typically 24.2-24.5 hours) and fails to entrain to the 24-hour light-dark cycle. Most commonly seen in totally blind individuals who lack light perception (50-80% prevalence in this population) — the absence of photic input to the SCN prevents entrainment. Sleep-wake times progressively drift later each day, cyclically moving in and out of alignment with the 24-hour day. When out of phase: insomnia, EDS. When in phase: normal sleep. Tasimelteon (Hetlioz), a melatonin MT1/MT2 receptor agonist, is FDA-approved for non-24 in totally blind adults, taken at the same time each night before bedtime.
Shift Work Disorder
Affects 10-38% of shift workers (particularly night and rotating shifts). Insomnia during the daytime sleep period and/or excessive sleepiness during the nighttime work period, with total sleep time reduction of 1-4 hours per 24-hour period. Associated with increased accident risk, cardiovascular disease, GI disorders, metabolic syndrome, and possibly cancer (IARC classified night shift work as "probably carcinogenic" — Group 2A). Diagnosis requires the symptoms to be temporally associated with a work schedule that overlaps the usual sleep period for ≥3 months.
Jet Lag Disorder
Transient circadian misalignment following rapid transmeridian travel (≥2 time zones). Eastward travel (phase advance required) is generally more difficult than westward (phase delay). Symptoms include insomnia, daytime sleepiness, GI upset, malaise, and impaired concentration. Recovery rate: approximately 1 day per time zone crossed eastward and 1 day per 1.5 time zones crossed westward.
Irregular Sleep-Wake Rhythm Disorder
Absence of a discernible circadian sleep-wake pattern — sleep is fragmented into ≥3 periods across the 24-hour day with no major consolidated nocturnal sleep period. Total sleep time over 24 hours may be normal, but no single sleep period exceeds 4 hours. Most commonly seen in patients with neurodegenerative disease (Alzheimer's, other dementias) due to SCN degeneration, and in institutionalized patients with reduced light exposure and social zeitgebers. Treatment: structured light exposure, scheduled activities, melatonin at bedtime, minimizing nighttime disruptions.
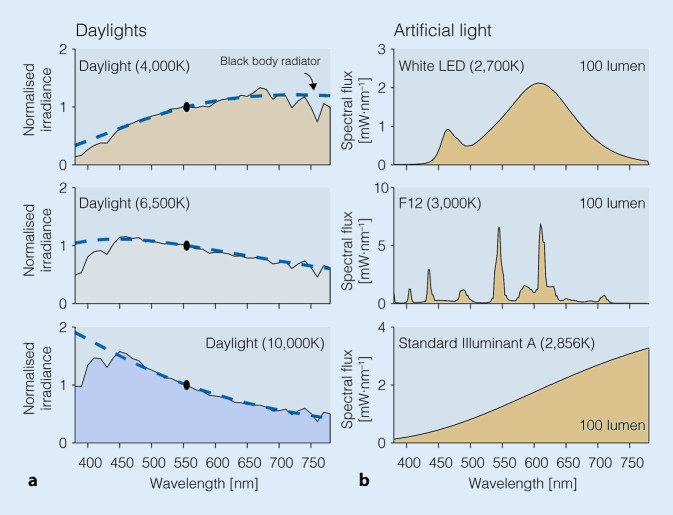
Circadian Rhythm Disorders — Summary Table
| Disorder | Phase Shift | Typical Population | Light Therapy Timing | Melatonin Timing |
|---|---|---|---|---|
| DSWPD | Delayed 2-6 hours | Adolescents, young adults | Morning (upon awakening) | Evening, 3-6 h before DLMO |
| ASWPD | Advanced 2-4 hours | Elderly | Evening (7-9 PM) | Morning (upon early awakening) |
| Non-24 | Free-running (>24h period) | Totally blind | Not applicable (no light perception) | Fixed time nightly (tasimelteon) |
| Shift work | Misaligned with work schedule | Night/rotating shift workers | First half of night shift | Before daytime sleep |
| Jet lag | Transient misalignment | Transmeridian travelers | AM (eastward), PM (westward) | Destination bedtime |
| Irregular rhythm | Absent rhythm | Dementia, institutionalized | Structured daytime exposure | Bedtime |
16 Chronotherapy & Light Therapy
Phase Response Curve (PRC)
The PRC describes the direction and magnitude of circadian phase shifts in response to a stimulus (light or melatonin) administered at different circadian times, using the core body temperature minimum (Tmin, approximately 4:00-5:00 AM in most adults) as the reference point:
Light before Tmin (late evening / early night) → phase delay (shifts the clock later)
Light after Tmin (early morning) → phase advance (shifts the clock earlier)
Melatonin has the opposite PRC to light: Melatonin in the evening → phase advance; melatonin in the morning → phase delay.
Bright Light Therapy
10,000 lux at the cornea is the standard therapeutic intensity, administered for 20-30 minutes (or 2,500 lux for 1-2 hours). Light boxes should be positioned at eye level, 12-18 inches from the face. The patient should face the light but not stare directly into it. Timing is critical and depends on the disorder:
DSWPD (phase advance desired): Morning light immediately upon awakening, combined with evening light avoidance (blue-light-blocking glasses after sunset).
ASWPD (phase delay desired): Evening light (7:00-9:00 PM), combined with morning light avoidance (dark sunglasses upon early awakening).
Shift work (night shift): Bright light during the first half of the night shift; dark sunglasses on the commute home.
The most effective wavelength is blue light (460-480 nm), which maximally stimulates melanopsin-containing intrinsically photosensitive retinal ganglion cells (ipRGCs) — the primary photic input to the SCN. This is why blue-light-emitting screens are particularly disruptive to circadian rhythms in the evening.
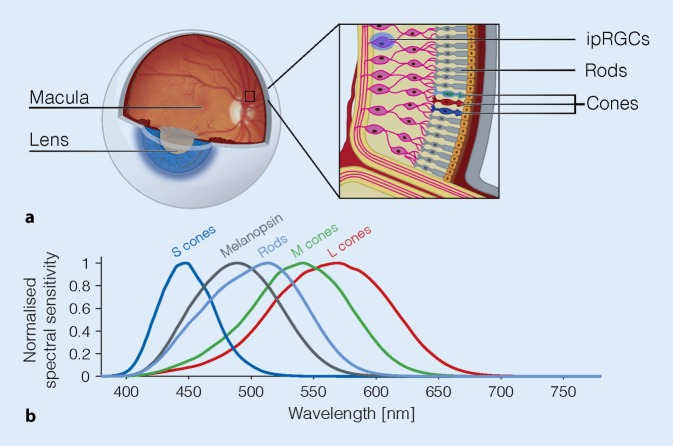
Exogenous Melatonin for Circadian Shifting
Low doses (0.5-3 mg) are preferred for chronobiotic (phase-shifting) effects — higher doses are sedating but do not provide additional phase-shifting benefit. Timing for DSWPD: administer 3-6 hours before DLMO (typically 5:00-7:00 PM for a patient whose DLMO is around 10:00 PM-12:00 AM). Timing for ASWPD: upon early morning awakening (phase delay). For non-24: fixed time nightly. Start low (0.5 mg) and titrate to effect.
DLMO Measurement
Dim Light Melatonin Onset (DLMO) is the gold-standard biomarker for circadian phase. Measured by serial salivary or plasma melatonin samples every 30-60 minutes beginning 6 hours before expected sleep onset, performed in dim light (<30 lux). DLMO is defined as the time at which melatonin rises above a threshold (typically 3 pg/mL for saliva or 10 pg/mL for plasma). Normal DLMO: approximately 8:00-10:00 PM for a conventional sleeper. Delayed DLMO (>11:00 PM) supports DSWPD.
17 Shift Work & Jet Lag Management
Shift Work — Alertness & Safety Strategies
Strategic napping: A 20-30 minute nap before the night shift ("prophylactic nap") and/or during a scheduled break significantly improves alertness. Naps >30 minutes risk sleep inertia upon awakening. The "anchor sleep" strategy recommends maintaining at least 4 hours of sleep at the same circadian time every day, even on days off, to provide a partial anchor point for circadian entrainment.
Caffeine: 150-200 mg at the start of the shift; avoid within 4-6 hours of the intended sleep time after the shift. Sustained low-dose caffeine (e.g., 100 mg every 2-3 hours) is more effective for sustained alertness than a single large dose.
Blackout curtains and sleep environment optimization: Essential for daytime sleep. Room should be dark (blackout curtains/eye mask), quiet (white noise machine/earplugs), cool, and phone-free.
Pharmacologic alertness: Modafinil 200 mg or armodafinil 150 mg before the shift is FDA-approved for shift work disorder. Solriamfetol is also FDA-approved for EDS in shift work disorder.
Sleep-promoting agents: Melatonin 0.5-3 mg before daytime sleep may improve sleep duration. Short-acting hypnotics (zaleplon, zolpidem) may be used cautiously for severe shift-work-related insomnia.
Jet Lag Management
Eastward travel (phase advance needed): Pre-trip: advance sleep schedule by 1 hour/day for 3 days before travel. On arrival: seek morning light, avoid afternoon/evening light. Melatonin 0.5-3 mg at target destination bedtime for 2-4 days.
Westward travel (phase delay needed): Pre-trip: delay sleep schedule by 1 hour/day for 3 days. On arrival: seek evening light, avoid morning light. Melatonin is generally less necessary for westward travel, but can be used at local bedtime if needed.
General principles: Hydration, avoid alcohol on the flight (worsens sleep disruption and dehydration), minimize sedative use to avoid masking circadian misalignment, adapt to local meal times and social schedule immediately. For trips ≤2 days, it may be more practical to remain on the home time zone.
Fatigue Risk Management Systems (FRMS)
Organizational approach to managing fatigue in safety-sensitive industries (aviation, healthcare, transportation). Components: education, scheduling practices (limit consecutive night shifts, ensure adequate recovery time, rotate shifts clockwise — delay direction), workload management, fatigue reporting systems, and monitoring/evaluation. Healthcare accreditation bodies (ACGME) limit resident duty hours to 80 hours/week with at least 1 day off per 7.
Employer Accommodation & Legal Considerations
Circadian rhythm sleep-wake disorders may qualify for reasonable accommodation under the Americans with Disabilities Act (ADA) when they substantially limit a major life activity (sleep). Accommodations may include shift reassignment, flexible scheduling, and permission for strategic napping during breaks. Employers in safety-sensitive industries (transportation, healthcare, nuclear power) must balance individual accommodation with public safety obligations. Documentation from a board-certified sleep medicine physician strengthens accommodation requests. The employer is not required to eliminate essential job functions or create undue hardship.
18 NREM Parasomnias
NREM parasomnias are disorders of arousal — they arise from partial arousal from slow-wave sleep (N3), typically during the first third of the night when N3 is most abundant. They share common features: impaired consciousness, amnesia for the event, autonomic activation, and difficulty being fully awakened.
Confusional Arousals
Episodes of mental confusion or confusional behavior during or following arousal from sleep, without the terror or ambulation of other NREM parasomnias. The individual appears disoriented, with slow mentation, slurred speech, and inappropriate behavior. Common in children under age 5 (prevalence ~17%). In adults, associated with sleep deprivation, shift work, and hypersomnia disorders.
Sleepwalking (Somnambulism)
Prevalence: 1-4% of adults, up to 17% of children (peak 8-12 years). Episodes: ambulation with a blank stare, reduced responsiveness, and impaired judgment. Complexity ranges from sitting up and walking to complex behaviors (leaving the house, eating, urinating in inappropriate locations). Duration: minutes to 30+ minutes. Amnesia for the event is typical. Triggers: sleep deprivation (most potent), fever, alcohol, sedatives (especially zolpidem — can cause "Ambien sleepwalking"), stress, sleep apnea (forced arousals from N3), full bladder.
Safety measures are the primary intervention: lock doors and windows, install alarms, remove hazardous objects, sleep on the ground floor, block stairways.
Sleep Terrors (Night Terrors)
Sudden arousal from N3 with a piercing scream or cry, intense autonomic activation (tachycardia, diaphoresis, mydriasis), and an appearance of extreme fear. The individual is inconsolable, unresponsive to reassurance, and confused if awakened. Duration: 1-10 minutes, followed by return to sleep. Complete amnesia. Most common in children ages 3-8. Key distinction from nightmares: sleep terrors arise from NREM (first third of night), have amnesia, and the individual appears terrified but has no dream recall; nightmares arise from REM (last third of night), the individual awakens fully with vivid dream recall and delayed return to sleep.
Sleep-Related Eating Disorder (SRED)
Recurrent episodes of involuntary eating during partial arousals from NREM sleep. The individual prepares and consumes food with partial or no awareness. May eat unusual or inedible substances. Associated with zolpidem use, sleepwalking history, RLS, and other sleep disorders. Weight gain and risk of injury (cooking while impaired) are complications. Treatment: address triggers, safety measures; topiramate and SSRIs have shown benefit.
Exploding Head Syndrome
Episodes of perceiving a sudden, loud noise (explosion, gunshot, cymbal crash) or flash of light at the wake-sleep transition, without actual external stimulus. There is no pain or headache. The experience can be frightening and may cause significant anxiety about falling asleep. Prevalence: up to 10-18% of the general population report at least one episode. The etiology is uncertain, possibly related to transient dysfunction of brainstem reticular formation neurons during the transition from wake to sleep. Treatment: reassurance that the condition is benign is usually sufficient. In severe cases, clomipramine or calcium channel blockers have been reported beneficial in case series.
Sexsomnia (Sleep-Related Abnormal Sexual Behaviors)
Sexual behaviors during partial arousals from NREM sleep, ranging from moaning to complex sexual acts, with amnesia for the events. Forensic implications are significant. Triggers mirror other NREM parasomnias: sleep deprivation, alcohol, comorbid sleep disorders.
Treatment of NREM Parasomnias
Safety measures: Priority in all cases. Secure the environment.
Address precipitants: Treat comorbid OSA (CPAP may eliminate parasomnias if forced arousals from N3 are the trigger), avoid sleep deprivation, discontinue offending medications, limit alcohol.
Scheduled (anticipatory) awakenings: Particularly effective in children. Wake the child 15-30 minutes before the typical time of the parasomnia event (identified by diary) for 2-4 weeks.
Clonazepam: 0.25-2 mg at bedtime — suppresses N3 and reduces arousal frequency. Reserve for severe, frequent, or dangerous events. Use lowest effective dose.
19 REM Sleep Behavior Disorder
REM sleep behavior disorder (RBD) is characterized by loss of the normal skeletal muscle atonia during REM sleep, allowing patients to physically act out their dreams — often with violent or vigorous motor behaviors (punching, kicking, jumping out of bed, shouting). The dream content is typically vivid, action-filled, and often involves being chased or attacked.
Pathophysiology
Normal REM atonia is generated by the sublaterodorsal (SLD) nucleus in the pons, which activates glycinergic and GABAergic interneurons in the ventral medulla and spinal cord to inhibit motor neurons. In RBD, degeneration or dysfunction of this atonia pathway allows motor commands from dream-generating circuits to be transmitted to skeletal muscles. On PSG, this manifests as sustained or excessive phasic chin EMG activity during REM sleep (REM sleep without atonia, or RSWA).
Association with Synucleinopathies
Idiopathic/isolated RBD is one of the strongest prodromal markers for alpha-synucleinopathies: Parkinson disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA). Longitudinal studies demonstrate that up to 90% of patients with isolated RBD will eventually convert to a defined synucleinopathy, at a rate of approximately 6-7% per year, with median conversion time of 12-14 years. This has led to RBD being incorporated into prodromal PD research criteria (MDS). Patients with isolated RBD should be counseled about this risk and offered longitudinal follow-up with screening for motor symptoms, cognitive changes, and autonomic dysfunction.
Biomarkers for Phenoconversion Risk in Isolated RBD
Several markers help estimate phenoconversion risk in patients with isolated RBD. Higher-risk features include: abnormal dopamine transporter (DAT) imaging (DaTscan), olfactory dysfunction (hyposmia), color vision impairment, subtle motor changes (quantitative motor testing), autonomic dysfunction (constipation, orthostatic hypotension, erectile dysfunction), and cognitive decline on serial neuropsychological testing. The combination of DAT deficit + hyposmia identifies patients at highest short-term conversion risk. Current research is exploring neuroprotective trial enrollment of these high-risk individuals, as the prodromal window offers a therapeutic opportunity before irreversible neurodegeneration.
Diagnosis
Requires: (1) Repeated episodes of dream enactment behavior (vocalizations and/or complex motor behaviors during REM), AND (2) PSG demonstration of REM sleep without atonia (RSWA) — excessive sustained or phasic EMG activity in the chin and/or limbs during REM epochs, AND (3) behaviors are not better explained by another sleep disorder, mental disorder, medication, or substance.
Secondary/medication-induced RBD: SSRIs, SNRIs, TCAs, and MAOIs can cause or worsen RBD by disinhibiting REM motor activity. Beta-blockers and bisoprolol have also been implicated.
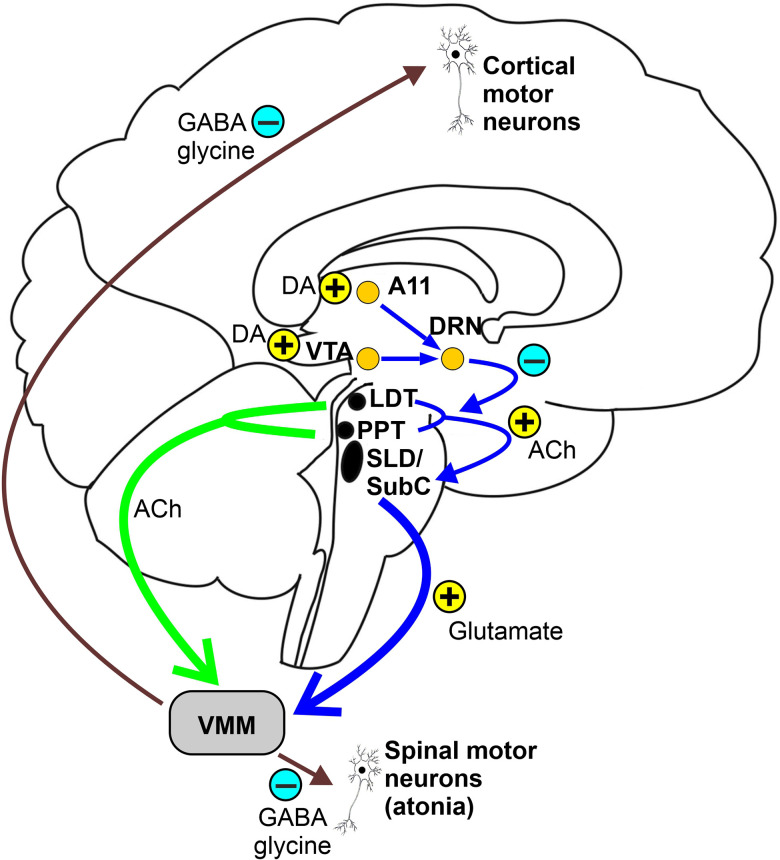
Treatment
Clonazepam 0.5-2 mg at bedtime: First-line pharmacotherapy. Reduces dream-enactment behaviors in ~90% of patients. Does not normalize REM atonia on PSG but suppresses phasic motor activity. Caution in patients with OSA (may worsen), cognitive impairment, gait instability, or fall risk.
Melatonin 3-12 mg at bedtime: Alternative or adjunct to clonazepam. May partially restore REM atonia. Better tolerated in elderly and patients with comorbid dementia or OSA. Often used first-line in these populations.
Bed safety: Critical. Remove sharp objects and furniture near the bed. Place mattress on the floor or use bed rails. Separate sleep surfaces for bed partners if injury risk is significant. Pad sharp corners.
20 Restless Legs Syndrome
Restless legs syndrome (RLS), also called Willis-Ekbom disease, is a sensorimotor disorder characterized by an irresistible urge to move the legs, usually accompanied by uncomfortable sensations, occurring primarily at rest and in the evening/nighttime, and relieved by movement.
IRLSD Essential Diagnostic Criteria (All 5 Required)
1. An urge to move the legs, usually accompanied by or felt to be caused by uncomfortable and unpleasant sensations in the legs
2. The urge begins or worsens during periods of rest or inactivity (lying down, sitting)
3. The urge is partially or totally relieved by movement (walking, stretching) — at least as long as the activity continues
4. The urge occurs exclusively or predominantly in the evening or night (circadian pattern, not merely a consequence of inactivity)
5. The above features are not solely accounted for by another condition (positional discomfort, leg cramps, myalgia, venous stasis, arthritis, habitual foot tapping, leg edema)
Supportive Features
1. Family history of RLS (first-degree relative) — present in ~50% of primary RLS
2. Response to dopaminergic therapy
3. Periodic limb movements during sleep (PLMS) — present in >80% of RLS patients
Iron Studies — The Critical Assessment
Brain iron deficiency is central to RLS pathophysiology (impaired dopamine synthesis in the substantia nigra). Serum ferritin <75 ng/mL (not the standard <20-30 ng/mL used for anemia) warrants iron supplementation in RLS patients. Check ferritin and transferrin saturation (TSAT) in all RLS patients. Oral iron: ferrous sulfate 325 mg with vitamin C on empty stomach, every other day (optimizes absorption). Reassess ferritin at 3-4 months. IV iron (ferric carboxymaltose 750 mg x2 doses, 1 week apart; or iron sucrose 200 mg x5 doses) is indicated when ferritin <100 with TSAT <20%, oral iron intolerance/failure, or moderate-severe RLS requiring rapid relief.
Pharmacotherapy
First-line: Alpha-2-delta calcium channel ligands
Gabapentin enacarbil (Horizant): 600 mg at 5:00 PM (FDA-approved for RLS). Prodrug of gabapentin with improved bioavailability. Effective for both sensory symptoms and PLMS.
Pregabalin: 150-450 mg at bedtime. RCT data support non-inferiority or superiority to pramipexole without augmentation risk.
Gabapentin: 300-2400 mg in divided doses with largest dose in the evening. Off-label but widely used.
Second-line: Dopamine agonists — now second-line due to augmentation risk
Pramipexole: 0.125-0.5 mg 2-3 hours before symptom onset. Ropinirole: 0.25-4 mg. Rotigotine patch: 1-3 mg/24h.
Augmentation — The Major Complication of Dopaminergic Therapy
Augmentation is a paradoxical worsening of RLS symptoms caused by the dopaminergic medication itself. Features: earlier onset of symptoms (afternoon instead of evening), faster onset at rest, spread to arms or trunk, increased intensity, shorter duration of relief from medication. Occurs in 30-70% of patients on long-term dopamine agonists (especially pramipexole). Risk factors for augmentation: higher dopaminergic dose, longer treatment duration, low ferritin, and use of short-acting agents. Management: taper and discontinue the dopamine agonist (gradually over 1-2 weeks — acute withdrawal can cause severe rebound lasting 2-4 weeks), check and optimize iron stores (IV iron if ferritin <75), switch to an alpha-2-delta ligand. During the withdrawal/transition period, low-dose opioids (oxycodone 5-15 mg, tramadol 50-100 mg) may be needed as rescue therapy for severe symptoms.
Periodic Limb Movements of Sleep (PLMS)
Repetitive, stereotyped dorsiflexion of the great toe and ankle (triple-flexion response) during sleep. Duration: 0.5-10 seconds. Interval: 5-90 seconds between movements. Series: ≥4 consecutive movements. The PLMS index (PLMI) = PLMS per hour of sleep. Clinically significant when PLMI ≥15/hr in adults (≥5/hr in children). PLMS are present in >80% of RLS patients but are also common in the elderly without RLS. PLMS alone without symptoms (RLS, insomnia, or EDS) is generally not treated.
21 Other Sleep Movement Disorders
Sleep Bruxism
Rhythmic or sustained jaw clenching and tooth grinding during sleep. Prevalence: 8-13% of adults. Manifests as tooth wear (attrition), jaw pain, temporal headache, and TMJ dysfunction. PSG shows rhythmic masticatory muscle activity (RMMA) — episodes of masseter EMG bursts. Treatment: dental occlusal guard (night guard/splint) is first-line for dental protection. Botulinum toxin injection into the masseter and/or temporalis muscles reduces bruxism severity and pain. Other measures: stress management, sleep hygiene, treatment of coexisting OSA (which may trigger bruxism through arousal-related jaw clenching).
Rhythmic Movement Disorder (RMD)
Repetitive, stereotyped, rhythmic movements (head banging [jactatio capitis nocturna], body rocking, head rolling) occurring at sleep-wake transition or during light sleep. Almost universal in infants (prevalence ~60% at 9 months), declining sharply with age (<5% by age 5). Persistent RMD in adults is rare and may be associated with intellectual disability, autism spectrum disorder, or anxiety. Generally benign; intervention (padded crib, helmet) only if injury risk.
Sleep-Related Leg Cramps
Sudden, involuntary, painful contractions of the calf (or foot/thigh) muscles during sleep or at rest, lasting seconds to minutes, with residual soreness. Extremely common (up to 50-60% of adults report occasional nocturnal cramps). Predisposing factors: dehydration, electrolyte abnormalities (magnesium, potassium), peripheral vascular disease, neuropathy, diuretic use, pregnancy, prolonged standing. Management: stretching exercises (calf stretches before bed), adequate hydration, magnesium supplementation (400-800 mg), correction of electrolyte abnormalities. Quinine is effective but FDA-warned against off-label use for leg cramps due to serious adverse effects (thrombocytopenia, arrhythmias).
Propriospinal Myoclonus at Sleep Onset
Sudden, repetitive jerks of the trunk and limbs occurring at the wake-sleep transition, preventing sleep onset. The myoclonus originates in the spinal cord and propagates rostrally and caudally along the propriospinal pathways. Distinguished from hypnic jerks by their repetitive nature and generation from the trunk. May be psychogenic in some cases. Treatment: clonazepam, valproate; address underlying anxiety or spinal pathology.
Hypnic Jerks (Sleep Starts)
Brief, sudden, involuntary muscle contractions occurring at the transition from wakefulness to sleep (N1). Extremely common — experienced by 60-70% of the general population at least occasionally. Typically involve a single whole-body jerk, often accompanied by a sensation of falling, a visual flash (hypnagogic imagery), or a brief vocalization. They are benign and require no treatment. Predisposing factors include sleep deprivation, caffeine, stress, and vigorous evening exercise. When frequent and disruptive to sleep onset, they may be termed excessive fragmentary myoclonus and occasionally warrant low-dose clonazepam.
Differential Diagnosis Table — Sleep-Related Movement Disorders
| Disorder | Timing | Character | Duration | Key Feature |
|---|---|---|---|---|
| PLMS | Throughout NREM (less in REM) | Stereotyped dorsiflexion of toe/ankle | 0.5-10 sec per movement | Series of ≥4, interval 5-90 sec; PLMI ≥15 |
| RLS | Evening/night at rest (wake) | Urge to move legs + sensory discomfort | Continuous while at rest | Relieved by movement; circadian pattern |
| Sleep bruxism | N1/N2, arousals | Rhythmic jaw clenching/grinding | Seconds per burst | Tooth wear, jaw pain, masseter RMMA on EMG |
| Hypnic jerks | Wake-N1 transition | Whole-body jerk, sensation of falling | Single event, <1 sec | Normal phenomenon; no treatment needed |
| RMD | Wake-sleep transition, light sleep | Rhythmic head banging or body rocking | Minutes | Common in infants; rare in adults |
| Propriospinal myoclonus | Wake-sleep transition | Repetitive trunk/limb jerks | Recurrent over minutes | Spinal cord origin; may be psychogenic |
| Nocturnal leg cramps | During sleep or rest | Painful involuntary calf contraction | Seconds to minutes | Residual soreness; common in elderly |
22 Sleep in Cardiovascular Disease
OSA & Hypertension
OSA is an independent risk factor for hypertension. The intermittent hypoxemia-reoxygenation cycles trigger sympathetic activation, oxidative stress, endothelial dysfunction, and RAAS activation. OSA is present in ~30-50% of patients with hypertension and in ~80% of patients with resistant hypertension (BP uncontrolled on ≥3 agents including a diuretic). The AHA and JNC guidelines recommend screening for OSA in all patients with resistant hypertension. CPAP has a modest but significant effect on BP reduction (mean ~2-3 mmHg systolic in meta-analyses), with greater benefit in adherent users (≥4 h/night) and those with resistant hypertension (mean ~5-7 mmHg).
Atrial Fibrillation
OSA is present in ~50% of patients with atrial fibrillation. Mechanisms: intermittent hypoxemia causes atrial remodeling, autonomic fluctuations (vagal surges followed by sympathetic activation), intrathoracic pressure swings, and inflammation. CPAP therapy reduces AF recurrence after cardioversion and ablation — untreated OSA is associated with ~25% higher AF recurrence rate after ablation. Current guidelines recommend OSA screening and treatment in all AF patients.
Heart Failure
Central sleep apnea with Cheyne-Stokes respiration occurs in 30-50% of HFrEF patients (see Section 7). OSA prevalence in heart failure is 12-53%. Both OSA and CSA independently predict worse outcomes. CPAP for OSA in heart failure improves EF and symptoms. ASV is contraindicated for CSA in HFrEF with EF ≤45% (SERVE-HF trial).
Stroke
Sleep-disordered breathing (predominantly OSA) is present in 50-70% of acute stroke patients. OSA is both a risk factor for stroke (2-3x risk) and a consequence of stroke (upper airway dysfunction from brainstem lesions). Screening for sleep apnea is recommended in all stroke patients. CPAP therapy in stroke rehabilitation may improve neurological recovery, though evidence is evolving.
Pulmonary Hypertension
Chronic intermittent and sustained hypoxemia from OSA can cause pulmonary vasoconstriction and eventual vascular remodeling, leading to Group 3 pulmonary hypertension. Severe OSA may elevate PA pressures even without daytime hypoxemia. CPAP treatment can reduce or reverse mild-moderate pulmonary hypertension when OSA is the sole cause.
Coronary Artery Disease & Nocturnal Ischemia
OSA is an independent risk factor for coronary artery disease (CAD) with a 1.5-2x adjusted risk. The mechanism involves intermittent hypoxemia triggering endothelial dysfunction, systemic inflammation (elevated CRP, IL-6, TNF-alpha), accelerated atherosclerosis, and platelet activation. Nocturnal ischemia is common: repetitive oxygen desaturations combined with sympathetic surges during apnea termination increase myocardial oxygen demand while simultaneously reducing supply. ST-segment depression on nocturnal ECG correlates with AHI severity. OSA patients have higher rates of nocturnal angina, non-dipping blood pressure pattern, and morning acute coronary syndrome events. CPAP therapy in patients with moderate-severe OSA and established CAD reduces nocturnal ischemia episodes and may improve endothelial function, though the SAVE trial did not demonstrate a reduction in major cardiovascular events with CPAP in secondary prevention.
Driving & OSA — Commercial Driver Regulations
The FMCSA does not mandate OSA testing but provides guidance: commercial motor vehicle (CMV) drivers with BMI ≥40, BMI ≥33 with risk factors, or who have had sleepiness-related accidents should be evaluated. Drivers with OSA must demonstrate PAP compliance (≥4 h/night on ≥70% of nights) to maintain medical certification. Annual recertification with PAP data download review is required. Non-adherent drivers should have driving privileges restricted until compliance is achieved.
Metabolic Consequences of OSA
OSA is independently associated with insulin resistance, glucose intolerance, and type 2 diabetes — mediated by intermittent hypoxemia, sympathetic activation, sleep fragmentation, and HPA axis dysregulation. The relationship is dose-dependent (worse with higher AHI and greater hypoxemic burden). OSA also independently increases dyslipidemia risk. CPAP therapy has modest but significant effects on insulin sensitivity in adherent users. All patients with OSA should be screened for metabolic syndrome. Conversely, patients with type 2 diabetes have a 50-70% prevalence of OSA.
Hypertension: 2-3x risk; dominant modifiable cause of resistant HTN
Coronary artery disease: 1.5-2x risk; nocturnal ischemia from hypoxemia + sympathetic surges
Atrial fibrillation: 2-4x risk; CPAP reduces recurrence post-ablation
Stroke: 2-3x risk; screen all stroke patients for SDB
Heart failure: Bidirectional relationship; CHF causes CSA, OSA worsens CHF
Sudden cardiac death: Peak occurrence during sleep hours in OSA (vs. early morning in the general population) — a distinctive epidemiologic pattern
Aortic aneurysm: Emerging association with severe OSA and intrathoracic pressure swings
23 Pediatric Sleep Medicine
Normal Sleep Duration by Age
| Age | Recommended Sleep (24h, including naps) | Notes |
|---|---|---|
| Newborn (0-3 months) | 14-17 hours | Irregular pattern; no circadian rhythm established |
| Infant (4-11 months) | 12-15 hours | Consolidated nocturnal sleep develops; 2-3 naps |
| Toddler (1-2 years) | 11-14 hours | Transition to 1 nap per day by 15-18 months |
| Preschool (3-5 years) | 10-13 hours | Napping diminishes by age 5 in most children |
| School-age (6-12 years) | 9-12 hours | No naps; increasing academic/social demands on sleep time |
| Teenager (13-17 years) | 8-10 hours | Physiologic circadian delay ("night owls"); early school start times conflict |
Behavioral Insomnia of Childhood
Sleep-onset association type: The child requires specific conditions to fall asleep (rocking, feeding, parental presence) and cannot self-soothe. When the association is absent (e.g., middle-of-night awakening without the parent), the child cannot return to sleep independently. Treatment: graduated extinction (letting the child self-soothe with timed check-ins at increasing intervals) or complete extinction (less parental involvement; faster but harder to implement). Teaching the child to fall asleep independently at bedtime transfers to middle-of-night awakenings.
Limit-setting type: The child stalls or refuses bedtime (requests for water, another story, bathroom). The parent fails to enforce consistent bedtime limits. Treatment: firm, consistent bedtime routine and limits; "bedtime pass" (one allowed out-of-bed request per night); positive reinforcement for compliance.
Pediatric Obstructive Sleep Apnea
Prevalence: 1-5% of children. Peak age: 2-8 years (adenotonsillar hypertrophy peak). Symptoms: snoring, mouth breathing, restless sleep, enuresis, behavioral problems (ADHD-like symptoms), failure to thrive, morning headaches. Adenotonsillectomy (AT) is first-line treatment and resolves OSA in 75-80% of otherwise healthy children. PSG is recommended before AT in children <2 years or with high surgical risk, and after AT if symptoms persist.
Pediatric AHI thresholds differ from adults: AHI ≥1 event/hour is abnormal in children (vs. ≥5 in adults). Mild: 1-5; moderate: 5-10; severe: >10. The obstructive apnea index (OAI, obstructive apneas only) ≥1 is also diagnostic.
PSG Indications in Children
Pre-AT in high-risk children (under 3, obesity, craniofacial anomalies, neuromuscular disease, Down syndrome), suspected OSA when clinical assessment is equivocal, evaluation of residual symptoms post-AT, suspected hypoventilation, and assessment for PAP titration in children who failed AT or are not surgical candidates.
Down Syndrome Screening
Children with Down syndrome have a 50-80% prevalence of OSA due to midface hypoplasia, macroglossia, hypotonia, and obesity. The AAP recommends PSG in all children with Down syndrome by age 4. Many require ongoing screening as OSA may develop or worsen with age.
Pediatric Insomnia
Insomnia in children can be categorized by developmental stage. In infants and toddlers, nighttime awakenings are the most common sleep complaint; signaling (crying upon awakening) is the problem rather than the awakening itself, which is developmentally normal. In school-age children, bedtime resistance and sleep-onset delay predominate, often related to anxiety, screen use, or inconsistent routines. In adolescents, a combination of circadian delay, social pressures, and electronic device use creates chronic sleep curtailment; mean sleep duration in U.S. teenagers is 7-7.5 hours, well below the recommended 8-10 hours. Pharmacotherapy for pediatric insomnia has minimal evidence; behavioral interventions are first-line at all ages. Melatonin (0.5-3 mg) may be considered for sleep-onset delay, particularly in children with autism spectrum disorder or ADHD, where evidence is strongest.
ALTE / BRUE
Brief Resolved Unexplained Event (BRUE) (replacing the older term ALTE — Apparent Life-Threatening Event): An event in an infant <1 year characterized by ≥1 of cyanosis/pallor, absent/decreased/irregular breathing, marked change in tone, and altered responsiveness. Lower-risk BRUE (age >60 days, gestational age ≥32 weeks, no CPR by trained provider, event duration <1 min, first event) can be managed with observation and parental education. Higher-risk BRUE may warrant further workup including consideration of PSG to evaluate for obstructive events or central apnea of infancy.
Congenital Central Hypoventilation Syndrome (CCHS)
CCHS (Ondine's curse) is a rare genetic disorder caused by PHOX2B gene mutations, resulting in failure of automatic (autonomic) respiratory drive, particularly during sleep. Affected infants hypoventilate during sleep (and in severe cases, during wakefulness) without perceiving dyspnea. Diagnosis requires genetic testing for PHOX2B polyalanine repeat expansion mutations (most common) or non-polyalanine repeat mutations. Management requires lifelong ventilatory support during sleep — typically via tracheostomy with mechanical ventilation in infants, with transition to diaphragm pacing or non-invasive ventilation in older children. Associated conditions include Hirschsprung disease (in ~20%), neural crest tumors (neuroblastoma, ganglioneuroblastoma), and autonomic nervous system dysregulation (cardiac rhythm abnormalities, pupillary dysfunction, temperature instability).
24 Sleep & Psychiatric Disorders
Insomnia and Depression — Bidirectional Relationship
Insomnia is both a symptom and risk factor for major depressive disorder (MDD). Insomnia is present in ~80% of depressive episodes. Persistent insomnia after MDD remission is the strongest predictor of relapse. Conversely, chronic insomnia increases the risk of developing new-onset MDD by 2-4x. Treating insomnia (CBT-I) in patients with comorbid depression improves both sleep and depressive symptoms — CBT-I augments antidepressant efficacy.
Sleep architecture abnormalities in depression include: shortened REM latency, increased REM density (number of eye movements per REM minute), decreased slow-wave sleep, increased sleep latency, and reduced sleep efficiency. These findings were historically proposed as biological markers for depression. The REM sleep changes reflect cholinergic-aminergic imbalance. SSRIs suppress REM sleep, which partly correlates with their antidepressant effect. Total sleep deprivation produces rapid (next-day) antidepressant effects in ~50-60% of depressed patients, though relapse occurs after recovery sleep — this effect is mediated through adenosine accumulation and monoaminergic modulation, and has led to research on wake therapy protocols combined with light therapy and sleep phase advance as augmentation strategies for treatment-resistant depression.
PTSD & Sleep
Sleep disturbance is a core feature of PTSD. Trauma-related nightmares occur in 50-70% of PTSD patients. Prazosin (alpha-1 adrenergic antagonist) was the standard pharmacotherapy for PTSD nightmares, dosed 1-15 mg at bedtime, though the RASKIND PTSD nightmare trial (VA study, 2018) showed mixed results and led to revised guidelines. Image rehearsal therapy (IRT) is an evidence-based behavioral approach: the patient rehearses a modified, non-threatening version of the nightmare during waking hours. OSA is highly prevalent in PTSD (up to 60-90% in some veteran cohorts) and should be actively screened for — untreated OSA worsens PTSD symptoms and nightmare frequency.
Bipolar Disorder
Sleep deprivation is a potent trigger for manic episodes. Even one night of total sleep loss can precipitate mania in vulnerable individuals. Maintaining a regular sleep-wake schedule (social rhythm therapy/interpersonal social rhythm therapy) is a cornerstone of bipolar management. Insomnia and hypersomnia are common in both manic and depressive phases. Sleep restriction therapy in CBT-I should be used with extreme caution in bipolar patients — a modified protocol with a higher time-in-bed floor (≥6.5 hours) is recommended.
During manic episodes, patients may sleep 2-4 hours per night (or not at all) without subjective sleepiness — this decreased need for sleep is a core DSM-5 criterion for mania and is distinct from insomnia, where the patient wants to sleep but cannot. During bipolar depressive episodes, hypersomnia (sleeping 10-14+ hours) occurs in 40-80% of patients and is a distinguishing feature from unipolar depression, where insomnia predominates. Medications used in bipolar disorder have varied sleep effects: lithium increases slow-wave sleep and total sleep time; quetiapine is strongly sedating and is often used specifically for its sleep-promoting effects; lamotrigine is relatively sleep-neutral; valproate may increase N3.
Substance Use & Sleep
Alcohol: Initial sedation (shortened sleep latency, increased N3 in the first half), followed by fragmented second-half sleep (REM rebound, increased awakenings). Chronic alcohol use suppresses REM and slow-wave sleep. Withdrawal: severe insomnia, anxiety, and REM rebound (vivid nightmares) — can persist for months to years in recovery.
Cannabis: Reduces REM sleep and dream recall; may reduce sleep latency acutely. Withdrawal: insomnia, vivid dreams for days to weeks.
Opioids: Reduce REM and N3; cause central sleep apnea; ataxic breathing patterns. Chronic opioid use (≥30 morphine milligram equivalents/day) is associated with sleep-disordered breathing in up to 75% of patients — both central and obstructive events. Ataxic (Biot's) breathing is characteristic of opioid effects on the brainstem and is dose-dependent.
Stimulants (cocaine, methamphetamine): Profound sleep deprivation during use; rebound hypersomnia with prolonged sleep during withdrawal ("crash"). Chronic use fragments sleep architecture and reduces total sleep time. REM rebound with vivid, often disturbing dreams occurs during early abstinence.
Anxiety Disorders & Sleep
Generalized anxiety disorder (GAD), panic disorder, and social anxiety disorder are all strongly associated with insomnia. In GAD, the hallmark pre-sleep cognitive hyperarousal — worry, rumination, and catastrophizing — directly interferes with sleep onset. Nocturnal panic attacks occur in ~45-70% of patients with panic disorder and typically arise from NREM stage N2 or N3 (not REM), distinguishing them from nightmares. The patient awakens abruptly with intense fear, tachycardia, dyspnea, and chest tightness. Unlike sleep terrors, the patient is fully alert upon awakening with complete recall. Treatment: address the underlying panic disorder (SSRIs, CBT for panic); benzodiazepines may be used short-term. CBT-I should be offered concurrently for comorbid insomnia.
Schizophrenia & Sleep
Sleep disturbance is nearly universal in schizophrenia. Features include prolonged sleep latency, reduced sleep efficiency, decreased slow-wave sleep, and variable REM abnormalities. Sleep architecture disruption often precedes psychotic relapse and can serve as an early warning sign. Antipsychotics have variable effects on sleep: sedating agents (olanzapine, quetiapine) may improve insomnia but cause excessive sedation and weight gain; activating agents (aripiprazole) may worsen insomnia initially.
Medication Effects on Sleep Architecture
| Medication Class | REM Sleep | N3/SWS | Sleep Latency | Other Notable Effects |
|---|---|---|---|---|
| SSRIs | Suppress (decreased REM %, increased REM latency) | Minimal effect | May increase | Increased PLMS; can trigger/worsen RBD; vivid dreams on withdrawal |
| TCAs | Suppress (potent REM suppression) | Variable (amitriptyline may increase) | Decrease (sedating TCAs) | Anticholinergic effects; weight gain; sedation varies by agent |
| Benzodiazepines | Suppress | Suppress | Decrease | Increase sleep spindles (N2); reduce arousal threshold; tolerance develops |
| Alcohol | Suppress (first half), rebound (second half) | Increase (first half), decrease (chronic use) | Decrease | Worsens OSA; fragments sleep; potentiates snoring |
| Beta-blockers | Suppress | Minimal | May increase | Suppress endogenous melatonin; vivid dreams/nightmares; insomnia |
| Corticosteroids | Suppress | Suppress | Increase | Profound insomnia at high doses; fragmented sleep |
| Stimulants | Suppress | May suppress | Increase | Dose-dependent insomnia; shorter-acting formulations preferred |
| Antipsychotics (sedating) | Variable | May increase (quetiapine) | Decrease | Quetiapine widely used off-label for insomnia; weight gain, metabolic effects |
| DORAs | Minimal effect | Minimal effect | Decrease | Preserve normal sleep architecture; no SWS/REM suppression |
| Opioids | Suppress | Suppress | Decrease | Central apneas; ataxic breathing; dose-dependent SDB; fragmented sleep |
| Lithium | Minimal | May increase | Variable | Increases total sleep time; polyuria may fragment sleep |
25 Scoring Systems
STOP-BANG Questionnaire
See Section 4 for full enumeration. Scoring: 0-2 low risk, 3-4 intermediate, 5-8 high risk for moderate-severe OSA.
Epworth Sleepiness Scale (ESS)
See Section 4 for full enumeration. 8 items, each 0-3. Total: 0-24. Abnormal: ≥10.
Insomnia Severity Index (ISI)
See Section 9 for full enumeration. 7 items, each 0-4. Total: 0-28. Clinical insomnia: ≥15.
Stanford Sleepiness Scale (SSS)
Rate your current level of sleepiness:
1. Feeling active, vital, alert, or wide awake
2. Functioning at high levels, but not fully alert
3. Awake, but relaxed; responsive but not fully alert
4. Somewhat foggy, let down
5. Foggy; losing interest in remaining awake; slowed down
6. Sleepy, woozy, fighting sleep; prefer to lie down
7. No longer fighting sleep, sleep onset soon; having dream-like thoughts
Interpretation: A single-point-in-time measure of subjective alertness. Scores ≥4 indicate significant sleepiness. Useful for repeated measurements across the day or shift.
Berlin Questionnaire
Category 1 (Snoring):
1. Do you snore? (Yes/No/Don't know)
2. Your snoring is: slightly louder than breathing / as loud as talking / louder than talking / very loud — can be heard in adjacent rooms
3. How often do you snore? (nearly every day / 3-4x/week / 1-2x/week / 1-2x/month / never or nearly never)
4. Has your snoring ever bothered other people? (Yes/No)
5. Has anyone noticed you quit breathing during your sleep? (nearly every day / 3-4x/week / 1-2x/week / 1-2x/month / never or nearly never)
Category 2 (Sleepiness):
6. How often do you feel tired or fatigued after your sleep? (nearly every day / 3-4x/week / 1-2x/week / 1-2x/month / never or nearly never)
7. During your waking time, do you feel tired, fatigued, or not up to par? (nearly every day / 3-4x/week / 1-2x/week / 1-2x/month / never or nearly never)
8. Have you ever nodded off or fallen asleep while driving a vehicle? (Yes/No)
If Yes: How often does this occur? (nearly every day / 3-4x/week / 1-2x/week / 1-2x/month / never or nearly never)
Category 3 (Hypertension/BMI):
9. Do you have high blood pressure? (Yes/No/Don't know)
10. BMI >30 kg/m2?
Scoring: Each category is positive or negative. Category 1 positive if ≥2 positive responses (questions 1-5). Category 2 positive if ≥2 positive responses (questions 6-8) and/or ESS >10. Category 3 positive if hypertension or BMI >30. High risk for OSA = ≥2 positive categories.
Maintenance of Wakefulness Test (MWT)
Protocol: 4 trials at 2-hour intervals (typically 10:00 AM, 12:00 PM, 2:00 PM, 4:00 PM). The patient sits upright in a dimly lit room in a comfortable chair, instructed to stay awake. Each trial is terminated at sleep onset or after 40 minutes if no sleep occurs. The PSG montage monitors for sleep onset.
Interpretation: Mean sleep latency ≥40 min (no sleep in any trial) = normal ability to maintain wakefulness. Mean latency <8 min = definitively impaired. Values between 8-40 min are in a "gray zone" with uncertain significance. The clinically important threshold for fitness-for-duty determinations is mean latency ≥40 minutes (all trials awake for the full 40 minutes), though no absolute consensus threshold exists.
Key distinction from MSLT: The MWT measures the ability to stay awake (capacity), while the MSLT measures the tendency to fall asleep (propensity). The MWT is used for fitness-for-duty evaluations (commercial driving, aviation, military, occupational safety), while the MSLT is a diagnostic tool for hypersomnolence disorders.
MSLT Criteria
See Section 12. Mean sleep latency ≤8 min + ≥2 SOREMPs = narcolepsy. Mean latency ≤8 min + <2 SOREMPs = idiopathic hypersomnia. Mean latency >8 min with normal number of SOREMPs = normal. Prerequisites: adequate prior night sleep on PSG (≥6 h), medication washout, sleep diary/actigraphy for ≥1 week.
Fatigue Severity Scale (FSS)
Rate each statement from 1 (strongly disagree) to 7 (strongly agree) based on the past week:
1. My motivation is lower when I am fatigued
2. Exercise brings on my fatigue
3. I am easily fatigued
4. Fatigue interferes with my physical functioning
5. Fatigue causes frequent problems for me
6. My fatigue prevents sustained physical functioning
7. Fatigue interferes with carrying out certain duties and responsibilities
8. Fatigue is among my three most disabling symptoms
9. Fatigue interferes with my work, family, or social life
Scoring: Mean score across 9 items. Mean ≥4 indicates clinically significant fatigue. Useful for distinguishing fatigue from sleepiness — an important distinction in sleep medicine, as many patients report "tired" when they mean sleepy, and management differs.
26 PAP Settings & Troubleshooting Quick Reference
| Issue | Likely Cause | Solution |
|---|---|---|
| Residual AHI >5 on CPAP | Inadequate pressure, weight gain, mask leak, positional component, treatment-emergent CSA | Check mask fit/leak data; consider pressure increase or repeat titration; evaluate for CSA; reassess weight |
| High mask leak (>24 L/min 95th percentile) | Ill-fitting mask, mouth leak, facial hair, skin breakdown | Refit mask, try different style/size, chinstrap for mouth leak, ensure clean and uncracked cushion |
| Aerophagia | High pressure, air swallowing | Lower pressure if possible, switch to BiPAP (lower EPAP), EPR/C-Flex, avoid eating before bed, elevate head of bed |
| Claustrophobia | Anxiety about mask/confinement | Nasal pillows (least contact), desensitization (wear while awake, TV), PAP-NAP, consider anxiolytic short-term |
| Dry mouth/nose | Mouth leak, low humidity | Heated humidifier + heated tubing, chinstrap, nasal saline, intranasal steroid, check for mouth leak |
| Nasal congestion | Pressure-induced rhinitis, allergic rhinitis | Heated humidity, intranasal steroid (fluticasone), intranasal antihistamine, evaluate for septal deviation/polyps |
| Skin breakdown / pressure sores | Overtightened headgear, ill-fitting mask | Loosen straps (mask should seal with minimal tension), try mask liner, alternate mask styles, wound care |
| Central apneas on CPAP | Treatment-emergent CSA | Often resolves over weeks; if persistent (>3 months), consider ASV (if EF >45%) or BiPAP-ST; lower CPAP pressure if overtitrated |
| Rainout (water in tubing) | Temperature differential between heated humidifier and cooler room/tubing | Heated tubing (climate control), insulate tubing, adjust humidity level, position device below bed level |
| Poor adherence (<4 h/night) | Multifactorial: discomfort, lack of perceived benefit, psychosocial | Address specific barrier, motivational interviewing, PAP-NAP, consider combination therapy (PAP + oral appliance at lower pressure), troubleshoot as above |
1. Usage hours: Mean, median, and distribution; % nights with ≥4 hours
2. Residual AHI: Should be <5; report obstructive vs. central events separately
3. Mask leak: 95th percentile; target <24 L/min (varies by device/manufacturer)
4. Pressure: 90th/95th percentile pressure on APAP (guides fixed-pressure prescription); pressure distribution
5. Cheyne-Stokes respiration: Some devices flag CSR episodes (percentage of night)
6. APAP 90th/95th percentile pressure: Guides conversion from APAP to fixed CPAP; set fixed pressure at 90th percentile + 1-2 cm H2O
PAP Pressure Modes & Comfort Features
| Feature | Description | Clinical Use |
|---|---|---|
| Ramp | Starts at a low pressure (4 cm H2O) and gradually increases to therapeutic pressure over 5-45 minutes | Improves comfort at sleep onset; patient falls asleep before high pressure is reached |
| EPR / C-Flex / A-Flex | Reduces expiratory pressure by 1-3 cm H2O relative to set CPAP pressure (manufacturer-specific terminology) | Reduces expiratory discomfort; improves adherence without compromising AHI control |
| AutoRamp | Device detects sleep onset and begins ramping from that point, maintaining low pressure during wake | More sophisticated than timed ramp; avoids pressure escalation while patient is still awake |
| Heated humidification | Integrated or attached heated water chamber adds moisture to delivered air | Standard of care; reduces nasal dryness, congestion, and mouth leak |
| Heated tubing | Tubing with embedded heating element prevents condensation (rainout) | Eliminates water accumulation in tubing from temperature differential |
27 Medications Master Table
Hypnotics
| Drug | Class | Dose Range | Half-Life | Schedule | Key Points |
|---|---|---|---|---|---|
| Zolpidem IR | BzRA | 5-10 mg | 2.5 h | IV | FDA lower dose for women (5 mg); complex sleep behaviors; take on empty stomach |
| Zolpidem ER | BzRA | 6.25-12.5 mg | 2.8 h | IV | Extended release for maintenance; same warnings |
| Zaleplon | BzRA | 5-20 mg | 1 h | IV | Ultra-short; can dose middle-of-night (≥4 h before rising) |
| Eszopiclone | BzRA | 1-3 mg | 6 h | IV | Longest half-life Z-drug; metallic taste; good for maintenance |
| Suvorexant | DORA | 10-20 mg | 12 h | IV | Orexin antagonist; onset/maintenance insomnia; no complex sleep behaviors warning |
| Lemborexant | DORA | 5-10 mg | 17-19 h | IV | Longer half-life DORA; no dose adjustment in elderly |
| Ramelteon | MT1/MT2 agonist | 8 mg | 1-2.6 h | Unscheduled | Sleep-onset insomnia; no abuse potential; avoid with fluvoxamine (CYP1A2 inhibitor) |
| Doxepin (low-dose) | Antihistamine (TCA) | 3-6 mg | 15 h | Unscheduled | Selective H1 antagonist at low dose; FDA-approved for maintenance insomnia; excellent in elderly |
| Trazodone | 5-HT2A antagonist | 25-100 mg | 7-8 h | Unscheduled | Off-label; widely used; orthostatic hypotension; priapism (rare) |
Wake-Promoting Agents
| Drug | Mechanism | Dose Range | Half-Life | Schedule | Key Points |
|---|---|---|---|---|---|
| Modafinil | DA reuptake inhibition (uncertain) | 100-400 mg | 15 h | IV | First-line wake-promoter; reduces OCP efficacy; SJS risk (HLA-B*15:02) |
| Armodafinil | R-enantiomer of modafinil | 150-250 mg | 15 h | IV | Longer effective duration than modafinil |
| Solriamfetol | DNRI | 75-150 mg (narcolepsy), 37.5-150 mg (OSA) | 7 h | IV | FDA-approved for narcolepsy and OSA; avoid in uncontrolled HTN; renal dose adjust |
| Pitolisant | H3 inverse agonist | 8.9-35.6 mg | 7-8 h | Unscheduled | Unique mechanism; also anti-cataplectic; QT prolongation; hepatic dose adjust |
| Methylphenidate | DA/NE reuptake inhibitor | 10-60 mg | 2-4 h | II | Short-acting; multiple daily doses needed; higher abuse potential |
| Dextroamphetamine | DA/NE releaser + reuptake inhibitor | 5-60 mg | 10-12 h | II | Most potent; highest abuse potential; reserve for refractory |
| Sodium oxybate | GABA-B agonist | 4.5-9 g/night (divided) | 0.5-1 h | III (REMS) | Addresses EDS + cataplexy; divided nocturnal dosing; REMS program required |
RLS Agents
| Drug | Class | Dose Range | Key Points |
|---|---|---|---|
| Gabapentin enacarbil | Alpha-2-delta ligand | 600 mg at 5 PM | FDA-approved for RLS; first-line; no augmentation |
| Pregabalin | Alpha-2-delta ligand | 150-450 mg qhs | Non-inferior to pramipexole; first-line; may be Schedule V in some states |
| Gabapentin | Alpha-2-delta ligand | 300-2400 mg/day | Off-label; largest dose in evening; dose-limit for tolerability |
| Pramipexole | Dopamine agonist (D2/D3) | 0.125-0.5 mg | Second-line; augmentation risk 30-70% long-term |
| Ropinirole | Dopamine agonist | 0.25-4 mg | Second-line; similar augmentation risk |
| Rotigotine patch | Dopamine agonist (transdermal) | 1-3 mg/24h | Steady-state levels; lower augmentation than oral DAs |
| Ferric carboxymaltose | IV iron | 750 mg x2 (1 week apart) | When ferritin <100 + TSAT <20%; rapid improvement |
28 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AASM | American Academy of Sleep Medicine |
| AHI | Apnea-Hypopnea Index |
| ALTE | Apparent Life-Threatening Event |
| APAP | Auto-titrating Positive Airway Pressure |
| ASV | Adaptive Servo-Ventilation |
| AT | Adenotonsillectomy |
| AVAPS | Average Volume-Assured Pressure Support |
| BiPAP | Bilevel Positive Airway Pressure |
| BMI | Body Mass Index |
| BRUE | Brief Resolved Unexplained Event |
| BzRA | Benzodiazepine Receptor Agonist |
| CBT-I | Cognitive Behavioral Therapy for Insomnia |
| CCC | Complete Concentric Collapse |
| CHF | Congestive Heart Failure |
| CMS | Centers for Medicare and Medicaid Services |
| CPAP | Continuous Positive Airway Pressure |
| CRT | Cardiac Resynchronization Therapy |
| CSA | Central Sleep Apnea |
| CSF | Cerebrospinal Fluid |
| CSR | Cheyne-Stokes Respiration |
| DA | Dopamine Agonist |
| DISE | Drug-Induced Sleep Endoscopy |
| DLB | Dementia with Lewy Bodies |
| DLMO | Dim Light Melatonin Onset |
| DNRI | Dopamine-Norepinephrine Reuptake Inhibitor |
| DORA | Dual Orexin Receptor Antagonist |
| DSWPD | Delayed Sleep-Wake Phase Disorder |
| ECG | Electrocardiogram |
| EDS | Excessive Daytime Sleepiness |
| EEG | Electroencephalogram |
| EF | Ejection Fraction |
| EMG | Electromyogram |
| EOG | Electrooculogram |
| EPAP | Expiratory Positive Airway Pressure |
| EPR | Expiratory Pressure Relief |
| ESS | Epworth Sleepiness Scale |
| FMCSA | Federal Motor Carrier Safety Administration |
| FRMS | Fatigue Risk Management System |
| GABA | Gamma-Aminobutyric Acid |
| GHB | Gamma-Hydroxybutyrate |
| HFrEF | Heart Failure with Reduced Ejection Fraction |
| HLA | Human Leukocyte Antigen |
| HSAT | Home Sleep Apnea Test |
| Hz | Hertz |
| ICSD-3 | International Classification of Sleep Disorders, 3rd Edition |
| IH | Idiopathic Hypersomnia |
| IPAP | Inspiratory Positive Airway Pressure |
| ipRGC | Intrinsically Photosensitive Retinal Ganglion Cell |
| IRLSD | International RLS Study Group Diagnostic Criteria |
| IRT | Image Rehearsal Therapy |
| ISI | Insomnia Severity Index |
| IV | Intravenous |
| KLS | Kleine-Levin Syndrome |
| LC | Locus Coeruleus |
| LDT | Laterodorsal Tegmental Nucleus |
| LVEF | Left Ventricular Ejection Fraction |
| MAD | Mandibular Advancement Device |
| MDD | Major Depressive Disorder |
| MMA | Maxillomandibular Advancement |
| MSA | Multiple System Atrophy |
| MSLT | Multiple Sleep Latency Test |
| MT1/MT2 | Melatonin Receptor Type 1 / Type 2 |
| MWT | Maintenance of Wakefulness Test |
| NE | Norepinephrine |
| NREM | Non-Rapid Eye Movement |
| OAI | Obstructive Apnea Index |
| ODI | Oxygen Desaturation Index |
| OHS | Obesity Hypoventilation Syndrome |
| OSA | Obstructive Sleep Apnea |
| PaCO2 | Partial Pressure of Arterial Carbon Dioxide |
| PALM | Pcrit / Arousal threshold / Loop gain / Muscle responsiveness |
| PAP | Positive Airway Pressure |
| Pcrit | Critical Closing Pressure |
| PD | Parkinson Disease |
| PGO | Ponto-Geniculo-Occipital (waves) |
| PLMI | Periodic Limb Movement Index |
| PLMS | Periodic Limb Movements of Sleep |
| PMR | Progressive Muscle Relaxation |
| PPT | Pedunculopontine Tegmental Nucleus |
| PRC | Phase Response Curve |
| PSG | Polysomnography |
| PTSD | Post-Traumatic Stress Disorder |
| RAAS | Renin-Angiotensin-Aldosterone System |
| RBD | REM Sleep Behavior Disorder |
| RDI | Respiratory Disturbance Index |
| REI | Respiratory Event Index |
| REM | Rapid Eye Movement |
| REMS | Risk Evaluation and Mitigation Strategy |
| RERA | Respiratory Effort-Related Arousal |
| RIP | Respiratory Inductance Plethysmography |
| RLS | Restless Legs Syndrome |
| RMD | Rhythmic Movement Disorder |
| RMMA | Rhythmic Masticatory Muscle Activity |
| RSWA | REM Sleep Without Atonia |
| SCN | Suprachiasmatic Nucleus |
| SE | Sleep Efficiency |
| SLD | Sublaterodorsal Nucleus |
| SOREMP | Sleep-Onset REM Period |
| SpO2 | Peripheral Oxygen Saturation |
| SRED | Sleep-Related Eating Disorder |
| SRT | Sleep Restriction Therapy |
| SSS | Stanford Sleepiness Scale |
| SWS | Slow-Wave Sleep (N3) |
| T90 | Percentage of Sleep Time with SpO2 <90% |
| TIB | Time in Bed |
| Tmin | Core Body Temperature Minimum |
| TMN | Tuberomammillary Nucleus |
| TSAT | Transferrin Saturation |
| TST | Total Sleep Time |
| UARS | Upper Airway Resistance Syndrome |
| UPPP | Uvulopalatopharyngoplasty |
| VAPS | Volume-Assured Pressure Support |
| VLPO | Ventrolateral Preoptic Area |
| VTA | Ventral Tegmental Area |
| WASO | Wake After Sleep Onset |
| CCHS | Congenital Central Hypoventilation Syndrome |
| CAD | Coronary Artery Disease |
| CRP | C-Reactive Protein |
| FSS | Fatigue Severity Scale |
| GAD | Generalized Anxiety Disorder |
| PHOX2B | Paired-like Homeobox 2B Gene |
| SDB | Sleep-Disordered Breathing |
| SOL | Sleep Onset Latency |
| VNS | Vagus Nerve Stimulation |
| VOTE | Velum, Oropharyngeal Lateral Walls, Tongue Base, Epiglottis (DISE classification) |
| GDMT | Guideline-Directed Medical Therapy |
| 6MWD | Six-Minute Walk Distance |
| AF | Atrial Fibrillation |