Toxicology
Toxidrome recognition, acetaminophen and salicylate poisoning, opioid and sedative overdose, toxic alcohols, envenomation, heavy metals, antidote therapy, decontamination, and every scoring system, antidote protocol, and management algorithm across the full scope of medical toxicology.
01 Toxicology Principles
Medical toxicology applies pharmacokinetic and pharmacodynamic principles to the unique context of overdose, where drug concentrations far exceed therapeutic ranges. Dose-response relationships in poisoning are often non-linear: at supratherapeutic doses, metabolic pathways saturate, protein binding sites are overwhelmed, and elimination shifts from first-order to zero-order (saturation) kinetics, meaning the body eliminates a fixed amount per unit time rather than a fixed fraction.
Pharmacokinetics in Overdose
| Parameter | Therapeutic Range | Overdose Behavior | Clinical Significance |
|---|---|---|---|
| Absorption | Predictable Tmax | Delayed by anticholinergic effects, bezoar formation, extended-release formulations | Levels may continue to rise hours after ingestion; repeat levels essential |
| Volume of Distribution (Vd) | Drug-specific constant | Low Vd (< 1 L/kg) — drug remains in plasma, amenable to dialysis; High Vd (> 2 L/kg) — drug distributes into tissues, dialysis ineffective | Lithium (Vd 0.7 L/kg) is dialyzable; digoxin (Vd 7 L/kg) is not |
| Protein Binding | High binding reduces free fraction | Binding sites saturate → free (active) fraction increases disproportionately | At therapeutic levels valproic acid is 90% bound; at toxic levels may be only 50% bound, doubling free drug |
| Metabolism | First-order kinetics (constant half-life) | Enzyme saturation → zero-order kinetics → half-life becomes unpredictable and prolonged | Phenytoin and salicylates classically switch to zero-order in overdose |
| Elimination | Renal/hepatic at predictable rate | Impaired if organ damage occurs (acetaminophen → hepatic failure, ethylene glycol → renal failure) | Enhanced elimination techniques may be life-saving when endogenous clearance fails |
Enhanced Elimination — Overview
When endogenous clearance is overwhelmed, extracorporeal or pharmacological methods can accelerate drug removal. The EXTRIP (Extracorporeal Treatments in Poisoning) Workgroup provides evidence-based recommendations for when to initiate dialysis in poisoning.
Drug properties: low molecular weight (< 500 Da), low Vd (< 1 L/kg), low protein binding (< 80%), water solubility. Clinical criteria: progressive clinical deterioration despite supportive care, serum level in lethal range, impaired endogenous clearance (renal or hepatic failure), prolonged or worsening clinical course, ingestion of substance with delayed toxicity (methanol, ethylene glycol).
Key Dose-Response Concepts
The toxic dose varies by substance and patient factors (age, hepatic/renal function, co-ingestants, tolerance). LD50 (lethal dose for 50% of a population) is a research metric with limited bedside utility. MLD (minimum lethal dose) is more clinically relevant. For acetaminophen, the toxic threshold is generally ≥150 mg/kg in a single ingestion. For many substances, the dose-response curve becomes sigmoidal — once a threshold is exceeded, toxicity escalates rapidly. Co-ingestants frequently shift dose-response curves leftward (lower threshold) through pharmacokinetic or pharmacodynamic interactions.
Initial Assessment of the Poisoned Patient
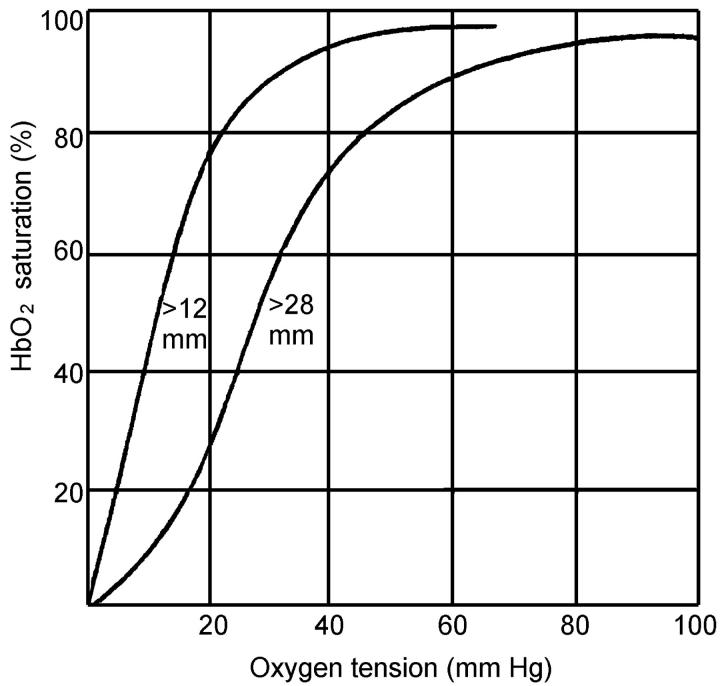
The approach to the undifferentiated poisoned patient follows a structured framework: 1. Stabilize: ABCs, IV access, cardiac monitoring, pulse oximetry. 2. Identify the toxidrome: vital signs, pupil size, skin moisture, bowel sounds, mental status, neuromuscular exam (tone, reflexes, clonus). 3. Obtain history: substance, dose, time, route, intent (accidental vs intentional), co-ingestants, medication list, past psychiatric history. 4. Targeted labs: BMP (AG, glucose, renal function), LFTs, APAP level, salicylate level, ethanol level, VBG/ABG, lactate, osmolality (for OG), pregnancy test, ECG (QRS/QTc), urinalysis (crystals, fluorescence under Wood lamp for EG). Urine drug screens (UDS) are of limited clinical utility — they have a high false-positive and false-negative rate, detect drug classes rather than specific agents, and should never delay empiric treatment.
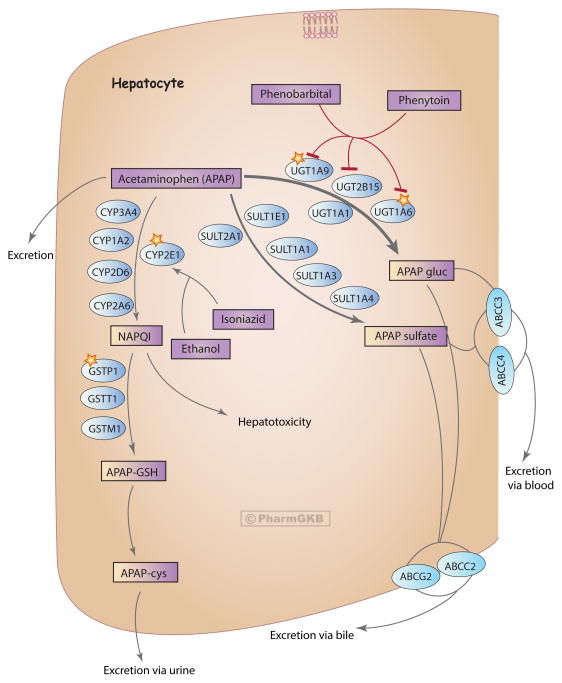
Anion Gap & Osmolar Gap
Anion gap = Na − (Cl + HCO3). Normal: 8–12 mEq/L. Elevated AG differential in toxicology (mnemonic CAT MUDPILES): Carbon monoxide/Cyanide, Aminoglycosides (rare), Toluene, Methanol, Uremia, DKA, Propylene glycol/Paraldehyde, Iron/INH, Lactic acidosis, Ethylene glycol, Salicylates.
Osmolar gap = Measured Osm − Calculated Osm. Calculated Osm = 2(Na) + BUN/2.8 + Glucose/18 + EtOH/4.6. Normal OG < 10. Elevated OG suggests: methanol, ethylene glycol, isopropanol, propylene glycol, ethanol, or mannitol. The OG and AG are inversely related in toxic alcohol poisoning — early: elevated OG with normal AG (parent compound present); late: normal OG with elevated AG (metabolites formed).
02 Toxidrome Recognition
A toxidrome is a constellation of signs and symptoms that suggests a specific class of poisoning. Rapid toxidrome identification narrows the differential and directs empiric therapy before laboratory confirmation. The five classic toxidromes plus serotonin syndrome and neuroleptic malignant syndrome form the foundation of clinical toxicology assessment.
Classic Toxidromes
| Toxidrome | Heart Rate | Blood Pressure | Temperature | Pupils | Skin | Mental Status | Other Key Features |
|---|---|---|---|---|---|---|---|
| Sympathomimetic | ↑↑ | ↑↑ | ↑ | Mydriasis | Diaphoretic | Agitation, psychosis | Seizures, tremor, hyperreflexia |
| Anticholinergic | ↑↑ | ↑ | ↑ | Mydriasis | Dry, flushed, hot | Delirium, hallucinations, mumbling | Urinary retention, decreased bowel sounds, "picking" |
| Cholinergic | ↓ (muscarinic) or ↑ (nicotinic) | Variable | Normal | Miosis | Diaphoretic | Confusion, coma | SLUDGE/DUMBBELSS, fasciculations, bronchorrhea |
| Opioid | ↓ | ↓ | ↓ | Miosis (pinpoint) | Normal | CNS depression, coma | Respiratory depression, hyporeflexia |
| Sedative-Hypnotic | ↓ | ↓ | ↓ | Normal or miosis | Normal | CNS depression, coma | Respiratory depression, hyporeflexia, no miosis distinguishes from opioid |
Anticholinergic Mnemonic
Dry as a bone (anhidrosis), Red as a beet (flushed), Hot as a hare (hyperthermia), Blind as a bat (mydriasis, cycloplegia), Mad as a hatter (delirium), Full as a flask (urinary retention), Stuffed as a sausage (ileus). Common agents: diphenhydramine, atropine, scopolamine, jimsonweed, TCAs, first-generation antihistamines, cyclobenzaprine.
Cholinergic Mnemonics
SLUDGE: Salivation, Lacrimation, Urination, Defecation, GI cramping, Emesis.
DUMBBELSS: Defecation, Urination, Miosis, Bradycardia, Bronchorrhea/Bronchospasm, Emesis, Lacrimation, Salivation, Sweating. DUMBBELSS is preferred because it includes bradycardia and bronchospasm, the two features most likely to be lethal.
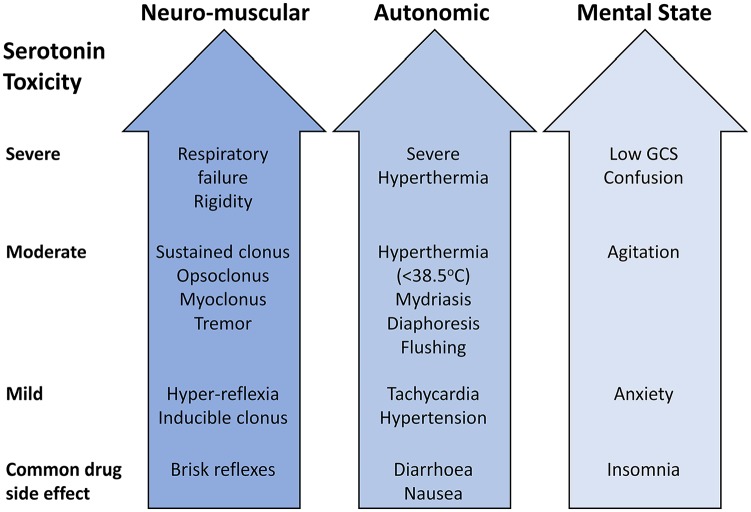
Serotonin Syndrome — Hunter Criteria
Serotonin syndrome requires exposure to a serotonergic agent PLUS at least one of the following:
In the presence of a serotonergic agent, serotonin syndrome is diagnosed if ANY of the following is present:
1. Spontaneous clonus
2. Inducible clonus + agitation OR diaphoresis
3. Ocular clonus + agitation OR diaphoresis
4. Tremor + hyperreflexia
5. Hypertonia + temperature > 38°C + ocular clonus OR inducible clonus
Treatment: Discontinue serotonergic agents. Benzodiazepines for agitation. Cyproheptadine 12 mg load PO, then 4 mg PO q6h (serotonin antagonist). Active cooling for hyperthermia. Avoid antipyretics (hyperthermia is muscular, not hypothalamic). Severe cases may require intubation and paralysis with non-depolarizing agents.
Neuroleptic Malignant Syndrome (NMS)
NMS results from dopaminergic blockade (antipsychotics, metoclopramide, or abrupt withdrawal of dopaminergic agents in Parkinson disease). Cardinal features: hyperthermia (often > 40°C), lead-pipe rigidity, altered mental status, autonomic instability (tachycardia, labile BP, diaphoresis). Labs: CK markedly elevated (often > 1000 U/L), leukocytosis, metabolic acidosis, elevated LDH and transaminases. Develops over days (slower onset than serotonin syndrome).
Treatment: Stop offending agent, aggressive IV fluids, active cooling, dantrolene 1–2.5 mg/kg IV q6h (skeletal muscle relaxant), bromocriptine 2.5 mg PO TID (dopamine agonist). Monitor CK and renal function for rhabdomyolysis.
Differentiating Serotonin Syndrome vs NMS vs Anticholinergic
| Feature | Serotonin Syndrome | NMS | Anticholinergic Toxicity |
|---|---|---|---|
| Onset | Hours (rapid) | Days to weeks | Hours |
| Causative agent | Serotonergic drugs | Dopamine antagonists | Anticholinergic drugs |
| Muscle tone | Clonus, hyperreflexia | Lead-pipe rigidity | Normal or mildly increased |
| Pupils | Mydriasis | Normal | Mydriasis |
| Skin | Diaphoretic | Diaphoretic ("pallid") | Dry, flushed |
| Bowel sounds | Hyperactive | Normal or decreased | Absent |
| CK | Mildly elevated | Markedly elevated (> 1000) | Normal |
| Key finding | Clonus (especially lower extremity) | Lead-pipe rigidity | Dry mucous membranes, urinary retention |
03 Terminology & Abbreviations
Foundational toxicology abbreviations used throughout this reference. A comprehensive master list is provided in Section 30.
04 Decontamination
GI decontamination aims to reduce absorption of ingested toxins. The window for efficacy is narrow, and interventions must be weighed against aspiration risk, patient cooperation, and substance identity. No decontamination method has been shown in randomized trials to improve clinical outcomes, so decisions are made based on the toxicity of the ingested substance and the time since ingestion.
Activated Charcoal (AC)
Dose: 1 g/kg (adult typical dose 50–100 g) as a single dose. Most effective when given within 1–2 hours of ingestion, though may be considered later for substances with delayed absorption (extended-release formulations, anticholinergic agents that slow GI motility).
Absolute: unprotected airway with depressed consciousness (aspiration risk), caustic ingestion (obscures endoscopy, no binding), GI perforation or obstruction.
Relative: substances not adsorbed by charcoal — metals (iron, lithium, potassium), alcohols (methanol, ethanol, ethylene glycol, isopropanol), hydrocarbons, and corrosives (acids, alkalis). Remember: "PHAILS" — Pesticides (some), Hydrocarbons, Acids/Alkalis/Alcohols, Iron, Lithium, Solvents.
Multiple-Dose Activated Charcoal (MDAC)
MDAC (25–50 g q2–4h) enhances elimination of substances that undergo enterohepatic recirculation or have prolonged GI absorption. Proven benefit for: carbamazepine, dapsone, phenobarbital, quinine, and theophylline. Requires a protected airway and active bowel sounds. Sorbitol should be given with only the first dose to avoid electrolyte disturbances.
Whole Bowel Irrigation (WBI)
GoLYTELY (polyethylene glycol electrolyte solution) at 1.5–2 L/hr via NG tube in adults (500 mL/hr in children) until rectal effluent is clear. Indications: body packers (drug-filled packets), sustained-release or enteric-coated preparations, iron or lithium ingestion (not adsorbed by charcoal), late presentations where charcoal is no longer effective. Contraindications: bowel obstruction, perforation, ileus, hemodynamic instability, unprotected airway.
Gastric Lavage
Rarely indicated in modern practice. Considered only within 1 hour of a life-threatening ingestion when charcoal alone is insufficient. Requires intubation for airway protection if mental status is depressed. Large-bore orogastric tube (36–40 Fr in adults). Complications: aspiration, esophageal perforation, laryngospasm, electrolyte disturbances. Not recommended routinely — American Academy of Clinical Toxicology/European Association of Poisons Centres position statement (2004).
Syrup of Ipecac
No longer recommended in any setting. Removed from OTC shelves in 2010. Induces vomiting via local gastric irritation and CTZ stimulation but offers no proven outcome benefit, delays definitive care, and poses aspiration risk. Not recommended by AAP, AACT, or EAPCCT.
05 Enhanced Elimination
Urinary Alkalinization
Sodium bicarbonate infusion (150 mEq NaHCO3 in 1 L D5W at 200–250 mL/hr) targeting urine pH 7.5–8.0. Ion trapping of weak acids in the renal tubule prevents reabsorption and enhances excretion. Effective for:
| Substance | Mechanism | Target Urine pH | Monitoring |
|---|---|---|---|
| Salicylates | Weak acid (pKa 3.5) — alkaline urine traps ionized form | 7.5–8.0 | Serum pH (do not exceed 7.55), urine pH q1h, potassium (must be repleted for alkalinization to work) |
| Methotrexate | Prevents crystallization in renal tubules | ≥7.0 | Serum methotrexate levels, creatinine, urine output |
| Phenobarbital | Weak acid trapping | 7.5–8.0 | Serum levels, respiratory status |
| Chlorpropamide | Weak acid trapping | 7.5–8.0 | Glucose, electrolytes |
Hemodialysis
Hemodialysis removes toxins by diffusion across a semipermeable membrane. Ideal properties for dialyzable substances: MW < 500 Da, Vd < 1 L/kg, protein binding < 80%, water soluble. EXTRIP-recommended indications:
| Substance | Dialysis Indications | Notes |
|---|---|---|
| Lithium | Level > 4 mEq/L (acute), > 2.5 mEq/L with renal impairment, or AMS/seizures at any level | Vd 0.7 L/kg; rebound after dialysis common — check levels 6h post-HD |
| Methanol | Metabolic acidosis, visual symptoms, level > 50 mg/dL, renal failure | Continue fomepizole during dialysis (dose adjustment needed as fomepizole is dialyzed) |
| Ethylene glycol | Metabolic acidosis, renal failure, level > 50 mg/dL | Dialysis removes both parent compound and toxic metabolites |
| Salicylates | Level > 100 mg/dL (acute), altered mental status, pulmonary edema, renal failure, pH < 7.2 despite treatment | Removes salicylate and corrects acidosis simultaneously |
| Theophylline | Level > 100 mcg/mL (acute), > 60 mcg/mL (chronic), seizures, arrhythmias | Charcoal hemoperfusion historically used; intermittent HD now preferred |
| Valproic acid | Level > 1300 mcg/mL, cerebral edema, hemodynamic instability | High protein binding at therapeutic levels but saturated in overdose — free fraction rises |
Other Modalities
CRRT (continuous renal replacement therapy): used when intermittent HD is not tolerated hemodynamically. Lower clearance rates but continuous removal; useful for lithium (prevents rebound). Exchange transfusion: rarely used; may be considered for severe methemoglobinemia in infants or massive hemolysis. ECMO: bridge therapy for refractory cardiotoxicity from beta-blockers, calcium channel blockers, or TCA poisoning when conventional vasopressors and antidotes fail.
Charcoal Hemoperfusion
Blood is passed over a charcoal cartridge that directly adsorbs toxin. Historically used for theophylline and carbamazepine, but largely replaced by standard intermittent HD due to availability, cost, and fewer complications (charcoal hemoperfusion causes thrombocytopenia, hypocalcemia, and cartridge saturation). May still be considered when HD is insufficient for large molecular weight or highly protein-bound substances.
Practical Considerations for Dialysis in Poisoning
| Factor | Standard Intermittent HD | CRRT | Charcoal Hemoperfusion |
|---|---|---|---|
| Clearance rate | Highest (200–300 mL/min) | Moderate (30–50 mL/min) | Variable (depends on cartridge) |
| Duration | 3–6 hours per session | Continuous (24+ hours) | 2–4 hours (cartridge saturation) |
| Hemodynamic tolerance | Requires stable hemodynamics | Tolerated in hypotension | Requires stable hemodynamics |
| Rebound prevention | Poor (drug redistributes after session) | Good (continuous removal) | Poor |
| Best for | Lithium, toxic alcohols, salicylates | Lithium (rebound), hemodynamically unstable patients | Theophylline, carbamazepine (historical) |
| Availability | Widely available | Most ICUs | Limited; rarely stocked |
| Complications | Hypotension, electrolyte shifts, air embolism | Filter clotting, citrate toxicity (if regional anticoagulation), hypothermia | Thrombocytopenia, hypocalcemia, cartridge saturation |
Step 1: Is the substance amenable to enhanced elimination? (Check MW, Vd, protein binding). Step 2: Is the clinical severity sufficient to warrant the intervention? (Deterioration despite supportive care, expected lethal ingestion, end-organ damage). Step 3: Can the procedure achieve clinically meaningful clearance? (Drug clearance by HD should exceed endogenous clearance by at least 30%). Step 4: Are there practical barriers? (Access to dialysis, patient hemodynamic stability, availability of alternative treatments such as antidotes).
06 Acetaminophen Toxicity
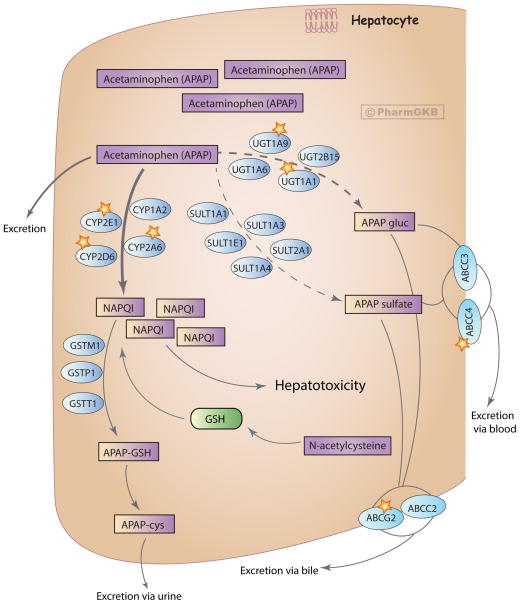
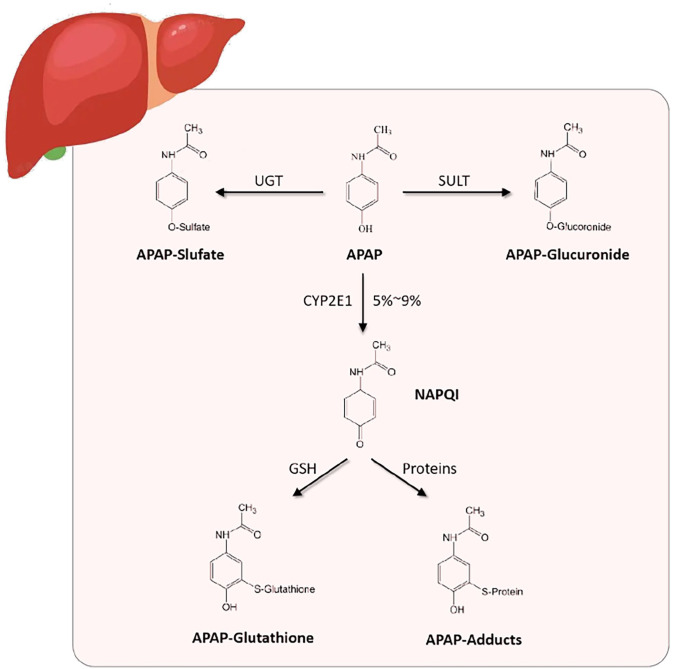
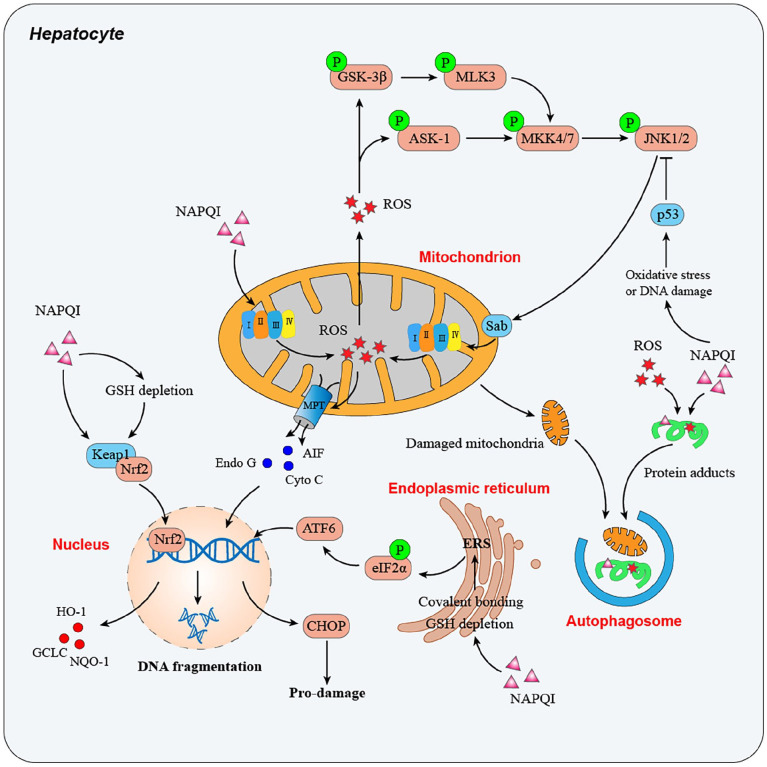
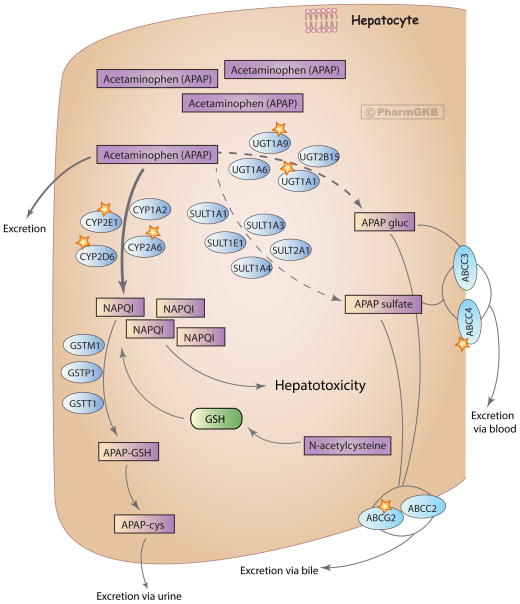
Acetaminophen (APAP) is the most common cause of acute liver failure in the United States and the most frequent drug involved in intentional overdose. At therapeutic doses, APAP is metabolized primarily by glucuronidation (40%) and sulfation (20–40%). A small fraction (< 5%) is oxidized by CYP2E1 to the reactive metabolite NAPQI (N-acetyl-p-benzoquinone imine), which is rapidly detoxified by glutathione. In overdose, glucuronidation and sulfation pathways saturate, shunting more drug through CYP2E1. Glutathione stores deplete when < 30% of normal, and unconjugated NAPQI binds hepatocyte proteins, causing centrilobular necrosis.
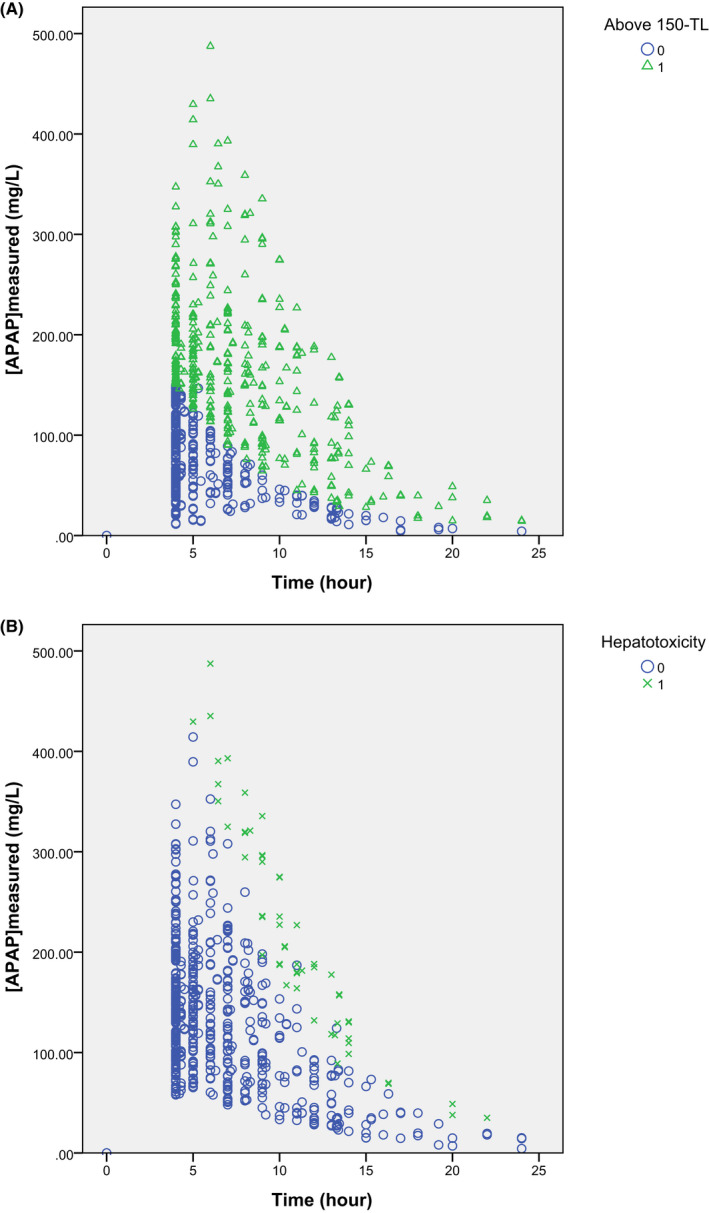
Rumack-Matthew Nomogram
Plot a single acute ingestion APAP level against time since ingestion (valid only from 4–24 hours). The original Rumack-Matthew line starts at 200 mcg/mL at 4 hours and declines to 50 mcg/mL at 12 hours and 6.25 mcg/mL at 24 hours (following the drug's half-life). In the US, treatment is initiated at the 150 mcg/mL at 4 hours treatment line (25% safety margin below the probable toxicity line). Any level above this line warrants NAC. The nomogram is not valid for: staggered ingestions, unknown time of ingestion, chronic/repeated supratherapeutic ingestion, or extended-release formulations.
Phases of Acetaminophen Toxicity
| Phase | Time Post-Ingestion | Clinical Features | Lab Findings |
|---|---|---|---|
| I | 0.5–24 hours | Asymptomatic, or nonspecific: nausea, vomiting, malaise, diaphoresis | Normal LFTs; APAP level may be elevated |
| II | 24–72 hours | Right upper quadrant pain; symptoms may initially improve ("latent period") | Rising AST/ALT (AST often rises first), PT/INR prolongation, possible rising creatinine |
| III | 72–96 hours | Peak hepatotoxicity: jaundice, coagulopathy, encephalopathy, renal failure, lactic acidosis, multi-organ failure | AST/ALT may exceed 10,000 IU/L, PT/INR markedly prolonged, metabolic acidosis, hypoglycemia |
| IV | 4 days–3 weeks | Recovery phase in survivors; liver regeneration begins | Gradual normalization of LFTs and coagulation parameters |
N-Acetylcysteine (NAC) Protocols
| Route | Protocol | Advantages | Disadvantages |
|---|---|---|---|
| IV (21-hour) | 150 mg/kg in 200 mL D5W over 1 hour, then 50 mg/kg in 500 mL D5W over 4 hours, then 100 mg/kg in 1000 mL D5W over 16 hours | Faster completion, can be used if vomiting, preferred for ALF | Anaphylactoid reactions (flushing, urticaria, bronchospasm) in 10–20%, especially with first bag |
| PO (72-hour) | 140 mg/kg loading dose, then 70 mg/kg q4h × 17 additional doses (total 18 doses over 72 hours) | Lower anaphylactoid risk, less costly | Prolonged course, vomiting may prevent administration (give with cola or ondansetron) |
NAC is nearly 100% effective at preventing hepatotoxicity when started within 8 hours of ingestion. Efficacy decreases with later initiation but NAC still provides benefit even in established hepatotoxicity (improved outcomes in ALF). Two-bag modified IV protocols (300 mg/kg over 20–21 hours) are increasingly used to reduce anaphylactoid reactions associated with the rapid first infusion.
NAC Anaphylactoid Reaction Management
Anaphylactoid reactions to IV NAC occur in 10–20% of patients, most commonly during the first (loading) infusion. These are non-IgE-mediated histamine release reactions, NOT true anaphylaxis. Risk factors: low initial APAP level (drug not present to bind NAC), history of asthma or atopy, rapid infusion rate. Management: stop the infusion temporarily, administer diphenhydramine 25–50 mg IV and/or ondansetron 4 mg IV for nausea. For bronchospasm: inhaled albuterol. For severe reactions: epinephrine 0.3 mg IM. Restart the infusion at a slower rate once symptoms resolve. NAC should NOT be permanently discontinued for anaphylactoid reactions in patients who need it — the risk of untreated APAP toxicity far outweighs the risk of the infusion reaction.
Repeated Supratherapeutic Ingestion (RSI)
Chronic or repeated supratherapeutic APAP ingestion cannot be plotted on the Rumack-Matthew nomogram. Risk assessment relies on: APAP level (any detectable level with symptoms warrants NAC), AST/ALT (if elevated, start NAC regardless of APAP level), and clinical risk factors (chronic alcohol use, CYP2E1 inducers, malnutrition/glutathione depletion, fasting). Start NAC if APAP level > 20 mcg/mL or if AST is elevated. Continue NAC until APAP is undetectable and AST is trending down or normal.
King's College Criteria — Liver Transplant Referral
Arterial pH < 7.30 after adequate fluid resuscitation (single strongest predictor), OR all three of: Grade III/IV hepatic encephalopathy + PT > 100 seconds (INR > 6.5) + Creatinine > 3.4 mg/dL. Additional poor prognostic markers: arterial lactate > 3.5 mmol/L after early resuscitation or > 3.0 mmol/L after full resuscitation.
Massive Ingestion (> 500 mg/kg)
Very large ingestions may overwhelm standard NAC dosing. Features suggesting massive ingestion: very early (< 4 hours) metabolic acidosis, markedly elevated lactate, early coma, initial APAP level > 500 mcg/mL. Consider doubling the NAC infusion rate and early nephrology consultation for possible hemodialysis (APAP is dialyzable, MW 151 Da, Vd 0.9 L/kg, low protein binding). Extended-release APAP may have a second peak; check a second level at 8–10 hours.
07 Salicylate Toxicity
Salicylate poisoning produces a characteristic mixed acid-base disturbance: early respiratory alkalosis (direct stimulation of the medullary respiratory center) followed by anion gap metabolic acidosis (uncoupling of oxidative phosphorylation, accumulation of organic acids). The combination of respiratory alkalosis + AG metabolic acidosis on ABG is nearly pathognomonic for salicylate toxicity.
Clinical Features by Severity
| Severity | Serum Level | Symptoms | Key Features |
|---|---|---|---|
| Mild | 30–50 mg/dL | Tinnitus, nausea, vomiting, hyperventilation | Respiratory alkalosis predominates |
| Moderate | 50–80 mg/dL | Agitation, diaphoresis, tachycardia, fever | Mixed respiratory alkalosis + metabolic acidosis |
| Severe | > 80–100 mg/dL | AMS, seizures, pulmonary edema, coma, cardiovascular collapse | Metabolic acidosis predominates, cerebral/pulmonary edema |
The Done nomogram is historically referenced but has significant limitations and is no longer recommended for clinical decision-making. Unlike the Rumack-Matthew nomogram, it does not reliably predict toxicity, particularly for chronic ingestions. Serial levels and clinical status are more useful.
Treatment
1. Volume resuscitation: IV NS bolus (salicylate patients are volume depleted from vomiting, insensible losses, and diuresis). 2. Urinary alkalinization: NaHCO3 infusion (150 mEq in 1 L D5W) targeting urine pH 7.5–8.0 and serum pH ≤7.55. Replete potassium aggressively (K+ 4.0–5.0 mEq/L). 3. Glucose: Administer D5W even with normal serum glucose — CNS glucose may be depleted despite normal serum levels. 4. Activated charcoal: 1 g/kg if within 1–2 hours (salicylate is well-adsorbed). 5. Hemodialysis for: altered mental status, pulmonary edema, renal failure, level > 100 mg/dL (acute) or > 60 mg/dL (chronic), seizures, clinical deterioration despite treatment, pH < 7.2.
Chronic Salicylate Toxicity
Chronic salicylate poisoning occurs in elderly patients on long-term aspirin therapy with declining renal function, or those inadvertently taking multiple salicylate-containing products. It is far more dangerous than acute ingestion because: the drug has already distributed into the CNS, serum levels may be only moderately elevated (30–60 mg/dL) despite severe clinical toxicity, and diagnosis is frequently delayed (average 72 hours from symptom onset to correct diagnosis). Presentation mimics sepsis or encephalopathy: confusion, tachypnea, tachycardia, fever, and metabolic acidosis. Mortality in chronic salicylism is 25% compared to < 2% in acute ingestion, largely due to diagnostic delay. Maintain a high index of suspicion in elderly patients with unexplained AG metabolic acidosis and altered mental status. Treatment is the same as for acute toxicity, but thresholds for hemodialysis are lower: consider dialysis for any chronic poisoning with neurologic symptoms regardless of level.
08 NSAID & COX-2 Overdose
NSAID overdose is generally benign in most cases. The majority of patients develop only mild GI symptoms (nausea, vomiting, epigastric pain) and recover with supportive care alone. Serious toxicity is rare but may include seizures, renal failure, and metabolic acidosis with massive ingestion.
Specific NSAID Considerations
| Agent | Toxic Dose | Unique Features | Management |
|---|---|---|---|
| Ibuprofen | > 400 mg/kg (serious) | Most common; GI symptoms predominate; seizures and metabolic acidosis with massive ingestion | Supportive; activated charcoal within 1–2 hours if > 400 mg/kg |
| Naproxen | > 20 g | Similar to ibuprofen; longer duration; seizures rare | Supportive |
| Mefenamic acid | > 5–10× therapeutic | Seizures are more common than with other NSAIDs — occurs in up to 20% of overdoses | Benzodiazepines for seizures; supportive care |
| Piroxicam | > 600 mg | Long half-life (50 hours); prolonged toxicity | Extended observation; supportive |
| COX-2 inhibitors | Limited data | Generally benign in overdose; cardiovascular risk at chronic therapeutic doses | Supportive; monitor renal function |
Treatment is supportive: IV fluids, GI decontamination with activated charcoal if presenting within 1–2 hours of a potentially significant ingestion, benzodiazepines for seizures, and monitoring renal function. Most patients can be observed for 4–6 hours and discharged if asymptomatic with normal labs.
Renal Complications of NSAID Overdose
NSAIDs inhibit prostaglandin-mediated afferent arteriolar vasodilation, reducing renal blood flow. In overdose, acute kidney injury may develop within 24–72 hours, particularly in patients with pre-existing renal impairment, volume depletion, or concurrent ACE inhibitor/ARB use. Monitor BUN, creatinine, and urine output in all significant NSAID ingestions. Interstitial nephritis (typically with chronic use rather than acute overdose) presents with eosinophiluria, pyuria, and renal failure; may require corticosteroids. Renal papillary necrosis is a complication of chronic NSAID abuse rather than acute overdose. Treatment of NSAID-induced AKI: discontinue the NSAID, aggressive IV hydration with isotonic crystalloid, avoid further nephrotoxins, and monitor electrolytes (hyperkalemia from reduced potassium secretion is a risk).
Observation and Disposition
| Ingestion Amount | Observation Period | Labs Required | Disposition |
|---|---|---|---|
| < 100 mg/kg ibuprofen | 2–4 hours | None if asymptomatic | Discharge if asymptomatic |
| 100–400 mg/kg ibuprofen | 4–6 hours | BMP, CBC | Discharge if asymptomatic with normal labs |
| > 400 mg/kg ibuprofen | 12–24 hours | BMP, CBC, LFTs, VBG q4–6h | Admit; seizure precautions; serial metabolic panels |
| Any mefenamic acid OD | 6–8 hours minimum | BMP, CBC | Observe with seizure precautions; IV access maintained |
09 Opioid Overdose
Opioid overdose presents with the classic triad: CNS depression, respiratory depression, and miosis (pinpoint pupils). It remains the leading cause of poisoning death in the United States. The rise of illicitly manufactured fentanyl has dramatically altered the epidemiology and clinical course of opioid overdose.
Naloxone (Narcan)
Initial dose: 0.04–0.4 mg IV/IM/IN for suspected opioid-dependent patients (start low to avoid precipitating withdrawal). Full reversal: 0.4–2 mg IV; may repeat q2–3 minutes to a maximum of 10 mg. Intranasal: 4 mg per nostril (Narcan nasal spray). Goal: restore adequate spontaneous respirations (RR ≥12), NOT full alertness. Titrate to respiratory effort, not consciousness, to minimize acute withdrawal.
Duration Mismatch & Renarcotization
Naloxone has a shorter duration of action (30–90 minutes) than most opioids. Renarcotization occurs when naloxone wears off while the opioid remains active. This is particularly dangerous with:
| Opioid | Duration | Required Observation | Special Considerations |
|---|---|---|---|
| Methadone | 24–36 hours | Minimum 24 hours | May require naloxone infusion (2/3 of initial effective dose per hour) |
| Extended-release morphine/oxycodone | 12–24 hours | Minimum 12–24 hours | Consider WBI if intact tablets suspected |
| Fentanyl (illicit) | Variable | Minimum 4 hours after last naloxone dose | May require higher naloxone doses (up to 10–12 mg); rapidly absorbed transdermally |
| Buprenorphine | 24–72 hours | Extended observation | High receptor affinity; partial agonist; may require very high naloxone doses |
| Heroin/immediate-release | 3–6 hours | Minimum 4–6 hours after last naloxone dose | Observation in ED usually sufficient |
Chest Wall Rigidity — Fentanyl Analogues
Rapid IV administration of potent synthetic opioids (fentanyl, sufentanil, carfentanil) can cause wooden chest syndrome — severe truncal and chest wall rigidity making bag-mask ventilation impossible. Treatment: naloxone (high dose, 2–10 mg), and if ventilation cannot be achieved, succinylcholine 1–2 mg/kg IV or rocuronium 1 mg/kg IV followed by endotracheal intubation.
Complications of Opioid Overdose
| Complication | Mechanism | Incidence / Risk Factors | Management |
|---|---|---|---|
| Noncardiogenic pulmonary edema (NCPE) | Increased alveolar-capillary permeability; may be related to naloxone-induced catecholamine surge | Up to 2% of heroin OD; may develop within hours of resuscitation | Supplemental O2, CPAP/BiPAP, intubation and PEEP for severe cases; usually resolves within 24–48 hours |
| Rhabdomyolysis | Prolonged immobility during stupor → muscle compression and ischemia | Patients found "down" for hours; concurrent stimulant use increases risk | Aggressive IV NS (200–300 mL/hr targeting UOP 200–300 mL/hr); monitor CK, K+, creatinine; consider NaHCO3 infusion for urine alkalinization if CK > 5000 |
| Compartment syndrome | Prolonged compression of extremity | Any limb trapped beneath body; concurrent tourniquet application | Measure compartment pressures if suspected; emergent fasciotomy for pressures > 30 mmHg or within 30 mmHg of diastolic BP |
| Aspiration pneumonia | Loss of airway protective reflexes during obtundation | Vomiting during obtundation; concurrent alcohol use | Antibiotics (ampicillin-sulbactam or clindamycin); suctioning; supportive care |
| Wound botulism | Clostridium botulinum colonization of injection sites | Black tar heroin subcutaneous injection ("skin popping"); California/Pacific NW | Heptavalent botulinum antitoxin; wound debridement; ICU monitoring for respiratory failure |
Naloxone Infusion
For opioids with long duration of action (methadone, extended-release preparations), a continuous naloxone infusion may be necessary. Protocol: calculate 2/3 of the initial bolus dose that achieved adequate respirations and infuse that amount per hour. Example: if 2 mg IV was the effective bolus, infuse ~1.3 mg/hr (2/3 of 2 mg). Titrate to maintain RR ≥12 and adequate tidal volume. Monitor for renarcotization if infusion rate is too low, and for withdrawal symptoms (agitation, vomiting, diarrhea, tachycardia) if rate is too high.
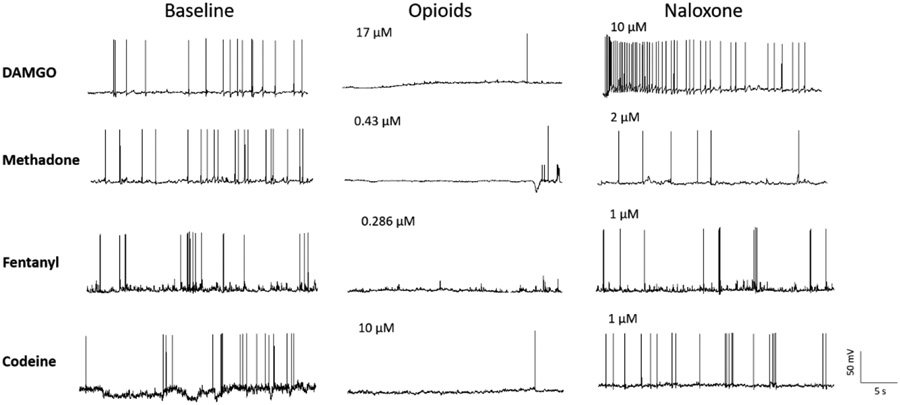
10 Tricyclic Antidepressant Poisoning
TCAs (amitriptyline, nortriptyline, imipramine, desipramine) remain among the most lethal medications in overdose. Toxicity results from three primary mechanisms: sodium channel blockade (cardiac conduction delays), anticholinergic effects, and alpha-1 adrenergic blockade (hypotension). Death typically occurs from refractory ventricular arrhythmias or cardiovascular collapse.
ECG Findings — Critical for Risk Stratification
| ECG Finding | Mechanism | Clinical Significance |
|---|---|---|
| QRS > 100 ms | Sodium channel blockade slowing phase 0 depolarization | Predicts seizures (QRS > 100 ms) and ventricular arrhythmias (QRS > 160 ms) |
| Right axis deviation of terminal 40 ms | Preferential slowing of right bundle branch | Terminal R wave in aVR > 3 mm is highly specific for TCA toxicity |
| Brugada pattern | Sodium channel blockade mimicking Brugada syndrome | ST elevation in V1–V3 with RBBB pattern; resolves with treatment |
| Sinus tachycardia | Anticholinergic effect + catecholamine reuptake inhibition | Often earliest finding; HR 120–160 common |
| QTc prolongation | Potassium channel blockade | Increases risk of torsades de pointes |
Treatment
Indication: QRS > 100 ms, ventricular arrhythmias, hypotension. Dose: 1–2 mEq/kg IV bolus, repeat q3–5 min until QRS narrows. Follow with NaHCO3 infusion (150 mEq in 1 L D5W) targeting serum pH 7.45–7.55. Mechanism: sodium loading overcomes sodium channel blockade, and alkalemia decreases free (active) TCA fraction by increasing protein binding. Do not exceed pH 7.55 (risk of alkalemia-induced arrhythmias).
Additional TCA Management
| Complication | First-Line Treatment | Second-Line / Rescue | Avoid |
|---|---|---|---|
| QRS widening | NaHCO3 1–2 mEq/kg IV bolus; repeat q3–5 min until QRS narrows | Hypertonic saline (3%) if refractory to bicarbonate; target serum Na 150–155 mEq/L | Class IA/IC antiarrhythmics (procainamide, flecainide) |
| Ventricular arrhythmias | NaHCO3; lidocaine 1–1.5 mg/kg IV (safe sodium channel blocker that does not prolong QRS) | Intralipid emulsion 20% 1.5 mL/kg bolus + infusion | Amiodarone (QTc prolongation); procainamide |
| Seizures | Benzodiazepines (lorazepam 4 mg IV, diazepam 10 mg IV) | Propofol or barbiturates for refractory seizures | Phenytoin (sodium channel blocker; worsens cardiac toxicity) |
| Hypotension | IV NS bolus + NaHCO3 bolus | Norepinephrine infusion (alpha-1 agonism counteracts TCA alpha blockade); intralipid emulsion | Pure vasodilators; physostigmine (risk of asystole) |
| Refractory cardiovascular collapse | Intralipid emulsion 20% (1.5 mL/kg bolus + 0.25 mL/kg/min) | VA-ECMO for bridge to recovery | Physostigmine at any point in TCA toxicity |
Observation: patients with TCA overdose require continuous cardiac monitoring for a minimum of 6 hours after the last abnormal ECG finding. If ECG is normal at 6 hours post-ingestion and patient is asymptomatic, the risk of subsequent cardiac toxicity is very low. Mental status changes alone (without ECG abnormalities) may persist longer due to anticholinergic effects and do not necessarily mandate prolonged cardiac monitoring.
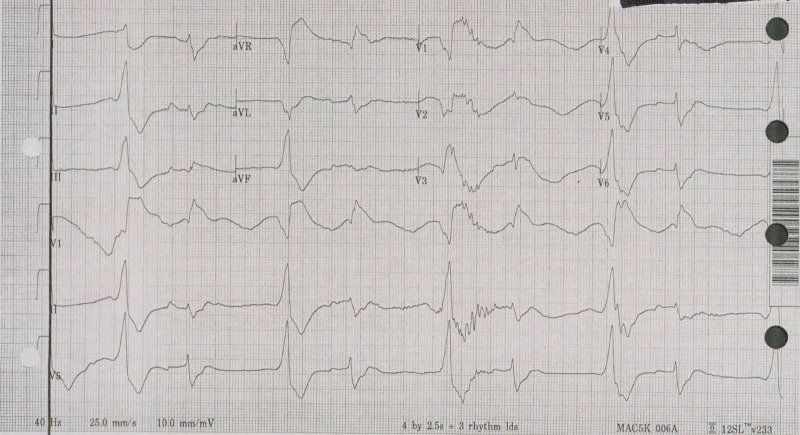
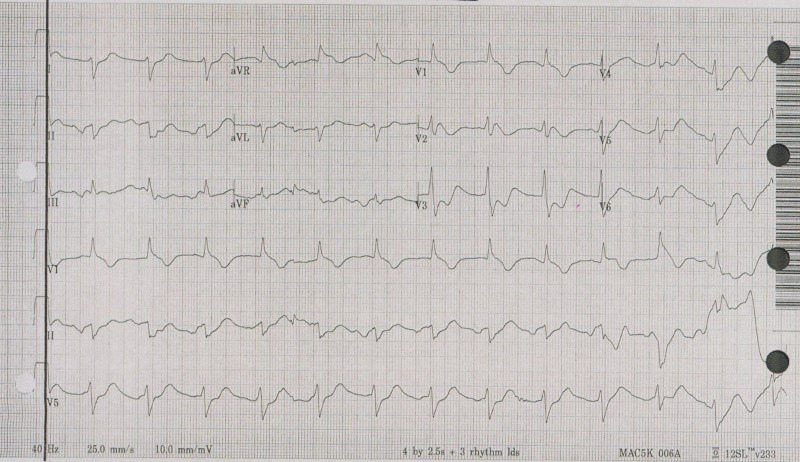
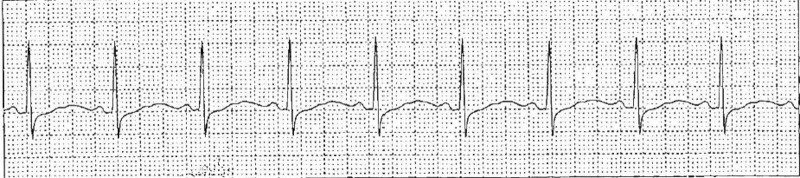
11 Lithium Toxicity
Lithium has a narrow therapeutic index (0.6–1.2 mEq/L). Toxicity patterns differ significantly between acute ingestion (lithium-naive patient or single large ingestion) and chronic toxicity (gradual accumulation in a patient on lithium therapy). Chronic toxicity is more dangerous because lithium has already equilibrated into the CNS.
Acute vs Chronic Lithium Toxicity
| Feature | Acute Ingestion | Chronic Toxicity |
|---|---|---|
| Serum level correlation | Poor — high levels may be asymptomatic initially | Better correlation with symptoms |
| Predominant symptoms | GI (nausea, vomiting, diarrhea) | Neurologic (tremor, ataxia, AMS, seizures) |
| Risk of permanent damage | Lower (drug not yet distributed into CNS) | Higher (CNS already saturated) |
| Response to dialysis | Levels drop rapidly; rebound common | Lower rebound; neurologic deficits may persist |
Severity Classification
| Severity | Level (mEq/L) | Clinical Features |
|---|---|---|
| Mild | 1.5–2.5 | GI symptoms, fine tremor, fatigue, mild confusion |
| Moderate | 2.5–3.5 | Coarse tremor, ataxia, dysarthria, nystagmus, agitation, myoclonic jerks |
| Severe | > 3.5 | Seizures, coma, cardiovascular collapse, nephrogenic DI, permanent cerebellar damage |
ECG Findings in Lithium Toxicity
Lithium affects cardiac conduction at toxic levels. Common ECG findings include: T wave flattening or inversion (most common, present in up to 30% of patients on therapeutic lithium), ST segment depression, QTc prolongation, sinus node dysfunction (sinus bradycardia, sinoatrial block — lithium concentrates in the sinus node), and rarely first-degree AV block. Severe toxicity can cause complete heart block and ventricular arrhythmias. The Brugada-like pattern (ST elevation in V1–V3) has been reported with lithium toxicity and resolves with treatment. All patients with suspected lithium toxicity should receive continuous cardiac monitoring. Pre-existing cardiac disease and electrolyte abnormalities (hyponatremia, hypokalemia) increase the risk of lithium-induced arrhythmias.
Drug Interactions Precipitating Lithium Toxicity
Thiazide diuretics: reduce sodium reabsorption in distal tubule → compensatory proximal tubule sodium (and lithium) reabsorption increases → lithium levels rise 25–40%. ACE inhibitors/ARBs: reduce GFR and increase proximal reabsorption of lithium. NSAIDs: inhibit prostaglandin-mediated renal blood flow → reduce lithium clearance by 15–20%. Metronidazole: reduces lithium renal clearance by unclear mechanism. Volume depletion from any cause (vomiting, diarrhea, poor intake, excessive sweating) concentrates lithium. Loop diuretics have a lesser effect than thiazides and are generally considered safer, though monitoring is still required.
SILENT Syndrome
The Syndrome of Irreversible Lithium-Effectuated Neurotoxicity (SILENT) describes persistent cerebellar dysfunction (ataxia, dysmetria, nystagmus, cognitive impairment) that persists after lithium levels normalize. Risk factors: prolonged exposure to toxic levels, chronic toxicity, delayed dialysis initiation, concurrent neuroleptic use. There is no specific treatment; deficits may be permanent.
Treatment
Fluid resuscitation: aggressive NS (lithium is handled like sodium by the kidney; volume depletion increases reabsorption). Hemodialysis indications: level > 4 mEq/L, significant neurologic symptoms regardless of level, renal impairment (cannot eliminate lithium), level > 2.5 mEq/L with symptoms and renal impairment. Check levels 6 hours post-dialysis — rebound is common and may necessitate repeat dialysis. Avoid: NSAIDs (reduce lithium clearance by 15–20%), ACE inhibitors, thiazide diuretics — all increase lithium reabsorption and worsen toxicity. Activated charcoal does NOT bind lithium.
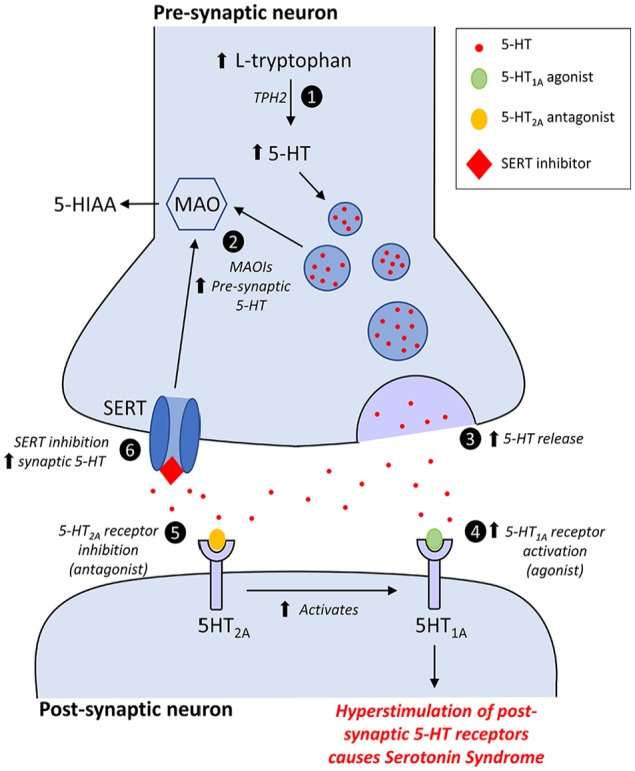
12 SSRI/SNRI & Serotonin Syndrome
SSRIs (fluoxetine, sertraline, citalopram, escitalopram, paroxetine, fluvoxamine) and SNRIs (venlafaxine, duloxetine, desvenlafaxine) in isolated overdose are generally less toxic than TCAs, but they can cause serotonin syndrome, especially in combination with other serotonergic agents. Citalopram is notable for dose-dependent QTc prolongation.
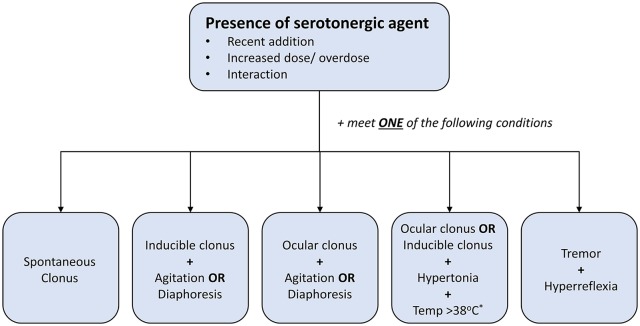
Serotonin Syndrome — Hunter Criteria (Full Enumeration)
Prerequisite: exposure to a serotonergic agent within the past 5 weeks (includes MAOIs, SSRIs, SNRIs, TCAs, tramadol, fentanyl, dextromethorphan, linezolid, St. John's wort, tryptophan, MDMA, lithium, ondansetron, metoclopramide). Diagnosis requires meeting at least ONE of the following criteria:
| Criterion | Details |
|---|---|
| 1 | Spontaneous clonus (rhythmic involuntary muscular contractions) |
| 2 | Inducible clonus + (agitation OR diaphoresis) |
| 3 | Ocular clonus (slow continuous horizontal eye movements) + (agitation OR diaphoresis) |
| 4 | Tremor + hyperreflexia |
| 5 | Hypertonia + temperature > 38°C + (ocular clonus OR inducible clonus) |
Treatment of Serotonin Syndrome
Mild: Discontinue serotonergic agents, observation, benzodiazepines for agitation. Moderate: Cyproheptadine 12 mg PO load, then 4 mg PO q6h (up to 32 mg/day); benzodiazepines; active cooling for T > 38.5°C. Severe: ICU admission, intubation and sedation with benzodiazepines, neuromuscular paralysis with non-depolarizing agent (vecuronium/rocuronium) for refractory hyperthermia, aggressive IV cooling, cyproheptadine (may need NG tube). Avoid succinylcholine (hyperkalemia risk from rhabdomyolysis). Antipyretics are ineffective because hyperthermia is muscular in origin, not hypothalamic.
Specific SSRI/SNRI Agent Toxicity Profiles
| Agent | Unique Overdose Features | Management Considerations |
|---|---|---|
| Citalopram/Escitalopram | Dose-dependent QTc prolongation (most cardiotoxic SSRIs); seizures with large ingestion (> 600 mg citalopram); delayed cardiac events up to 24 hours post-ingestion | Continuous cardiac monitoring for minimum 24 hours; serial ECGs; magnesium for QTc > 500 ms; avoid other QTc-prolonging agents |
| Fluoxetine | Very long half-life (1–3 days; active metabolite norfluoxetine: 4–16 days); serotonin syndrome risk persists for weeks after discontinuation | MAOIs must be withheld for 5 weeks after fluoxetine discontinuation; prolonged observation may be needed |
| Venlafaxine | Most toxic SNRI in overdose; seizures common (> 8% at toxic doses); serotonin syndrome; QRS widening and QTc prolongation at massive doses (> 8 g); hypertension followed by cardiovascular collapse | Extended-release formulations may have delayed peak toxicity; NaHCO3 for QRS widening; benzodiazepines for seizures |
| Sertraline | Generally benign in isolated overdose; serotonin syndrome risk primarily with co-ingestants; QTc prolongation less prominent than citalopram | Observation 6–8 hours; supportive care usually sufficient |
| Bupropion | Technically not an SSRI (NDRI); high seizure risk (dose-related, especially > 450 mg/day therapeutic, lower in overdose); delayed seizures up to 24 hours; tachycardia; QRS widening at massive doses | Observation minimum 24 hours (delayed seizures); seizure precautions; NaHCO3 if QRS widened; benzodiazepines for seizures; avoid MDAC (seizure risk during administration) |
NMS vs Serotonin Syndrome vs Anticholinergic Toxicity
See the differential table in Section 2. Key distinguishing features: serotonin syndrome has clonus (especially lower extremity) with hyperreflexia; NMS has lead-pipe rigidity with bradyreflexia; anticholinergic toxicity has dry skin and urinary retention. Serotonin syndrome develops over hours; NMS develops over days.
13 Antipsychotic Overdose
Antipsychotic toxicity differs between first-generation (typical) and second-generation (atypical) agents. All antipsychotics share dopamine D2 receptor antagonism; toxicity profiles diverge based on additional receptor pharmacology.
First vs Second Generation
| Feature | First-Generation (Haloperidol, Chlorpromazine) | Second-Generation (Olanzapine, Quetiapine, Risperidone, Ziprasidone) |
|---|---|---|
| Primary toxicity | EPS, NMS, QTc prolongation (especially droperidol, haloperidol IV) | Sedation, orthostatic hypotension, metabolic (hyperglycemia, DKA with olanzapine) |
| Seizure risk | Moderate (chlorpromazine, loxapine) | Higher with clozapine; moderate with quetiapine |
| QTc prolongation | Moderate | Most significant with ziprasidone — can cause TdP and metabolic acidosis in overdose |
| Anticholinergic effects | Significant (chlorpromazine, thioridazine) | Significant with olanzapine, quetiapine, clozapine |
QTc Prolongation & Torsades de Pointes Management
1. Magnesium sulfate 2 g IV over 1–2 minutes (first-line, even if serum Mg is normal). 2. Isoproterenol infusion 2–10 mcg/min (shortens QT by increasing HR). 3. Overdrive pacing (transvenous or transcutaneous) at 90–120 bpm if refractory. 4. Correct electrolytes (K+ to 4.5–5.0, Mg2+ > 2.0). Avoid: Class IA (procainamide), Class III (amiodarone) antiarrhythmics — they further prolong QT.
Neuroleptic Malignant Syndrome (NMS)
See Section 2 for full discussion. Treatment: stop offending agent, dantrolene 1–2.5 mg/kg IV q6h (maximum 10 mg/kg/day), bromocriptine 2.5 mg PO/NG TID, aggressive cooling, IV fluids, monitor CK and renal function. Mortality 5–20% even with treatment.
Extrapyramidal Symptoms (EPS)
| EPS Type | Onset | Features | Treatment |
|---|---|---|---|
| Acute dystonia | Hours to days after starting agent | Oculogyric crisis (forced upward eye deviation), torticollis, opisthotonos, laryngospasm (emergency), trismus | Diphenhydramine 25–50 mg IV or benztropine 1–2 mg IV; response within minutes; follow with 48 hours of oral anticholinergic therapy to prevent recurrence |
| Akathisia | Days to weeks | Subjective restlessness, inability to sit still, pacing, rocking; often misdiagnosed as agitation (increasing antipsychotic dose worsens it) | Reduce/change antipsychotic; propranolol 20–40 mg PO BID-TID; benzodiazepines; benztropine; mirtazapine |
| Drug-induced parkinsonism | Weeks to months | Bradykinesia, rigidity, tremor (less prominent than idiopathic PD), shuffling gait, masked facies | Benztropine 0.5–2 mg PO BID or trihexyphenidyl; dose reduction or switch to quetiapine/clozapine (lowest EPS risk) |
| Tardive dyskinesia | Months to years of use | Involuntary choreiform movements: lip smacking, tongue protrusion, grimacing, limb/trunk movements; may be irreversible | Stop or reduce offending agent; valbenazine 40–80 mg/day or deutetrabenazine 6–24 mg BID (VMAT2 inhibitors, FDA-approved) |
Specific Agent Considerations
| Agent | Unique Toxicity Profile | Management Notes |
|---|---|---|
| Quetiapine | Profound sedation, tachycardia, orthostatic hypotension; seizures with massive ingestion; less QTc prolongation than ziprasidone | Supportive; may require intubation for airway protection; most patients recover with supportive care |
| Olanzapine | Anticholinergic effects, sedation, metabolic derangements (hyperglycemia, DKA reported) | Monitor glucose; avoid concurrent IM benzodiazepines (risk of respiratory depression, hypotension) |
| Clozapine | Seizures (dose-related, most epileptogenic antipsychotic), myocarditis, agranulocytosis (chronic), profound sedation, sialorrhea | Seizure precautions; obtain troponin if chest pain/tachycardia; CBC for neutrophil count |
| Ziprasidone | Most significant QTc prolongation of atypical antipsychotics; metabolic acidosis in overdose | Continuous cardiac monitoring; magnesium prophylaxis; avoid other QTc-prolonging agents |
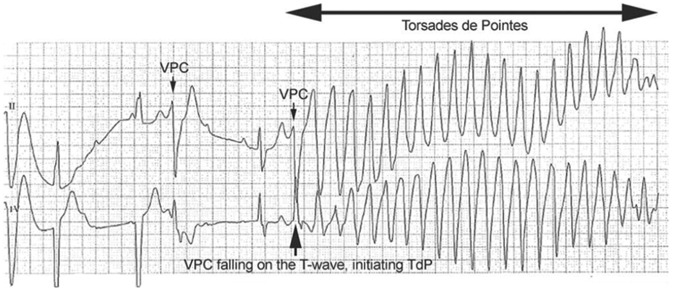
14 Methanol & Ethylene Glycol
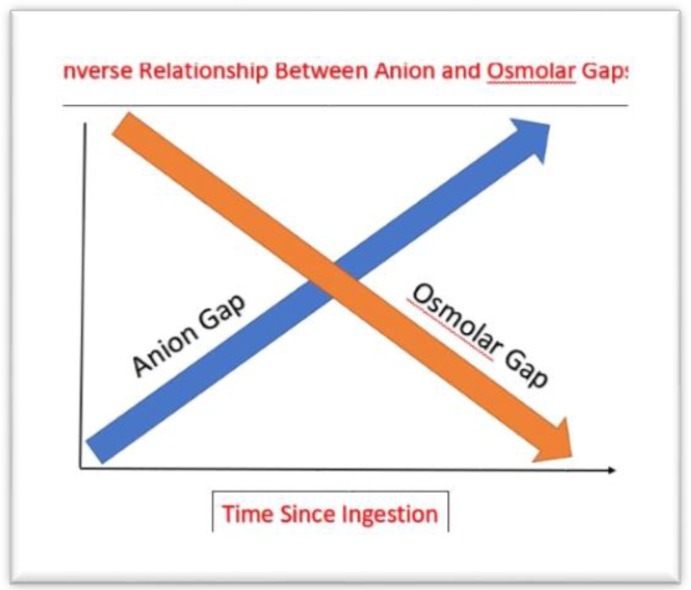
The toxic alcohols (methanol, ethylene glycol) are relatively non-toxic as parent compounds — their danger lies in their metabolites, produced by alcohol dehydrogenase (ADH). The diagnostic hallmark is a combination of elevated osmolar gap (early, before metabolism) and elevated anion gap metabolic acidosis (later, as metabolites accumulate). The osmolar gap and anion gap are inversely related over time.
Osmolar Gap Calculation
Calculated Osm = 2(Na) + BUN/2.8 + Glucose/18 + EtOH/4.6
Osmolar gap = Measured Osm − Calculated Osm. Normal < 10. Elevated OG (> 10) suggests unmeasured osmotically active substance: methanol, ethylene glycol, isopropanol, propylene glycol, or other low molecular weight solutes. A normal OG does NOT exclude toxic alcohol ingestion if metabolism has already occurred (parent compound already converted to toxic metabolites).
Methanol
Sources: windshield washer fluid, industrial solvents, illicit spirits (moonshine). Methanol is metabolized by ADH to formaldehyde, then by aldehyde dehydrogenase to formic acid. Formic acid causes: optic nerve toxicity (visual blurring, scotomata, blindness — "snowfield vision"), basal ganglia necrosis (putaminal hemorrhage on CT), and profound AG metabolic acidosis. Latency period of 12–24 hours before symptom onset (longer if ethanol co-ingested). The minimum lethal dose is approximately 1 mL/kg of pure methanol (15–30 mL can be fatal without treatment).
Methanol Clinical Staging
| Stage | Time | Features | Key Findings |
|---|---|---|---|
| I — Inebriation | 0–12 hours | CNS depression similar to ethanol intoxication; may be asymptomatic | Elevated osmolar gap; normal anion gap; normal pH |
| II — Latent period | 12–24 hours | Deceptive improvement or apparent sobriety | OG decreasing; AG beginning to rise as metabolites form |
| III — Metabolic acidosis | 24–72 hours | Visual changes (blurred vision, scotomata, blindness), headache, nausea, vomiting, abdominal pain, Kussmaul breathing | Profound AG metabolic acidosis; elevated lactate; elevated formate level; OG may be normal (parent compound metabolized) |
| IV — Multi-organ failure | > 72 hours | Seizures, coma, cardiovascular collapse, respiratory failure, death | Putaminal hemorrhage on CT; optic disc hyperemia on fundoscopy; renal failure |
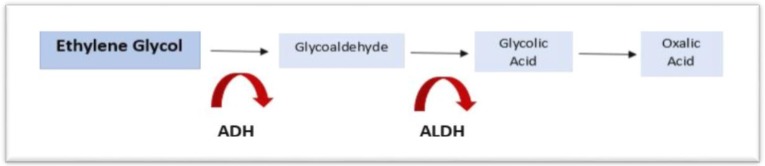
Ethylene Glycol
Sources: antifreeze (sweet taste → accidental pediatric ingestion), coolants. Metabolized by ADH to glycolaldehyde → glycolic acid (major contributor to acidosis) → glyoxylic acid → oxalic acid. Oxalic acid binds calcium → calcium oxalate crystals (needle-shaped or envelope-shaped on urinalysis, deposited in renal tubules → acute renal failure). Also causes hypocalcemia (ionized calcium drop from oxalate binding), cranial nerve palsies, and myocardial depression.
Treatment
| Intervention | Dose/Target | Mechanism |
|---|---|---|
| Fomepizole (4-MP) | 15 mg/kg IV load, then 10 mg/kg q12h × 4 doses, then 15 mg/kg q12h until level < 20 mg/dL and symptoms resolve | Competitive ADH inhibitor — blocks metabolism to toxic metabolites |
| Ethanol (alternative) | Loading dose: 0.8 g/kg IV (10% solution) or PO; maintenance to target serum ethanol 100–150 mg/dL | Competitive ADH substrate — preferentially metabolized over methanol/EG |
| Hemodialysis | Indicated for: metabolic acidosis, visual symptoms (methanol), renal failure (EG), level > 50 mg/dL, clinical deterioration | Removes parent compound and toxic metabolites; corrects acidosis |
| Co-factor therapy | Methanol: folic acid (leucovorin 50 mg IV q6h) — enhances formate metabolism. Ethylene glycol: thiamine 100 mg IV + pyridoxine 50 mg IV — shunt glyoxylate to non-toxic metabolites | Enhances alternative metabolic pathways |

15 Ethanol & Isopropanol
Acute Ethanol Intoxication
Management is primarily supportive: airway protection, glucose (check fingerstick — hypoglycemia is common, especially in children and malnourished adults), volume resuscitation, electrolyte correction, and observation until clinically sober. Serum ethanol levels do not reliably predict clinical status in chronic drinkers (tolerance). Life-threatening levels in non-tolerant adults generally > 400 mg/dL.
Wernicke Encephalopathy
Thiamine 500 mg IV TID for 3 days (high-dose regimen, per EFNS/Royal College guidelines), then 250 mg IV daily for 3–5 additional days. Classic triad (present in only ~16% of cases): encephalopathy (confusion), oculomotor dysfunction (nystagmus, ophthalmoplegia, conjugate gaze palsy), cerebellar ataxia. Thiamine must be given BEFORE or concurrent with glucose administration — glucose loading in a thiamine-depleted patient can precipitate or worsen Wernicke encephalopathy by exhausting remaining thiamine stores in pyruvate metabolism.
Ethanol Toxicity by Blood Alcohol Concentration
| BAC (mg/dL) | Clinical Effects (Non-Tolerant) | Management |
|---|---|---|
| 50–100 | Euphoria, impaired judgment, decreased coordination | Observation; assess for co-ingestants |
| 100–200 | Slurred speech, ataxia, nystagmus, impaired reaction time | Supportive; glucose check; fall precautions |
| 200–300 | Vomiting, stupor, marked ataxia, diplopia | Aspiration precautions; lateral recumbent position; IV fluids |
| 300–400 | Coma, respiratory depression, hypothermia | Airway protection; consider intubation; thiamine 500 mg IV; glucose; warming |
| > 400 | Respiratory arrest, cardiovascular collapse, death | ICU admission; intubation; aggressive resuscitation; hemodialysis for levels > 500–600 mg/dL with hemodynamic instability |
Alcoholic Ketoacidosis (AKA)
Occurs in chronic alcoholics after a binge followed by vomiting and poor intake. Presents with AG metabolic acidosis, low or normal glucose (distinguishes from DKA), elevated beta-hydroxybutyrate, positive ketones, and volume depletion. The nitroprusside test for ketones may be negative because AKA produces predominantly beta-hydroxybutyrate (not acetoacetate, which the test detects). Treatment: D5NS (dextrose stimulates insulin, shifting metabolism from ketogenesis to glucose utilization; saline repletes volume), thiamine, electrolyte correction. Resolves within 12–24 hours with fluid and dextrose. Insulin is NOT indicated.
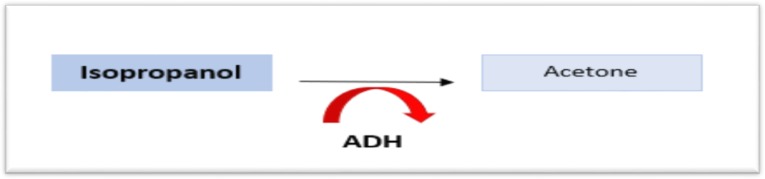
Isopropanol (Isopropyl Alcohol)
Sources: rubbing alcohol, hand sanitizer, windshield de-icer. Isopropanol is metabolized by ADH to acetone (NOT to an acid). Key features: ketosis without acidosis (fruity breath, positive serum ketones, but normal anion gap and normal pH), CNS depression proportional to dose, GI hemorrhagic gastritis. Treatment is supportive: airway protection, fluids, GI protection. Hemodialysis for severe intoxication (level > 400 mg/dL) or refractory hypotension, though rarely needed.
16 Sympathomimetics — Cocaine, Methamphetamine, MDMA
Cocaine
Cocaine blocks reuptake of norepinephrine, dopamine, and serotonin and also directly blocks sodium channels (local anesthetic effect). Toxicity includes: hypertensive emergency, acute coronary syndrome (even in young patients with normal coronaries — vasospasm), aortic dissection, stroke, seizures, and hyperthermia. Routes of use affect onset and duration: smoked/IV (onset seconds, duration 15–30 min, highest acute risk), insufflated (onset 5–10 min, duration 60–90 min), and oral/mucosal (onset 30–60 min, slowest). Body packers ("swallowers") ingest drug-filled packets for smuggling — packet rupture can cause massive cocaine toxicity with sudden death; management includes WBI with GoLYTELY and surgical consultation for obstructing or leaking packets.
Cocaine-Associated Rhabdomyolysis
Cocaine causes rhabdomyolysis through multiple mechanisms: direct myotoxicity, excessive muscular activity from agitation/seizures, ischemia from vasoconstriction, and hyperthermia. CK levels often exceed 10,000–100,000 IU/L. Management: aggressive IV crystalloid resuscitation targeting urine output 200–300 mL/hr, monitor for hyperkalemia and acute kidney injury, and consider NaHCO3 for urine alkalinization (target urine pH > 6.5) to prevent myoglobin precipitation in renal tubules.
First-line: benzodiazepines (diazepam 5–10 mg IV, lorazepam 2–4 mg IV) for both pain and hypertension. Nitroglycerin for persistent chest pain/hypertension. Aspirin 325 mg. Phentolamine 5 mg IV for refractory hypertension (alpha-blocker). AVOID beta-blockers (including labetalol) — unopposed alpha stimulation can worsen coronary vasospasm, hypertension, and cause end-organ damage. If ACS confirmed, PCI is preferred over thrombolytics. Calcium channel blockers (verapamil, diltiazem) may be used for vasospasm.
Methamphetamine
Sympathomimetic toxidrome plus serotonergic effects at high doses. Longer duration than cocaine (half-life 10–12 hours vs 1 hour). Unique complications: severe dental decay ("meth mouth"), skin excoriations from parasitosis delusions ("crank bugs"), pulmonary hypertension with chronic use, cardiomyopathy. Treatment: benzodiazepines for agitation and sympathomimetic symptoms, active cooling for hyperthermia, antihypertensives as needed. Serotonin syndrome may occur with massive doses.
MDMA (Ecstasy/Molly)
MDMA causes massive serotonin release plus sympathomimetic effects. Three life-threatening complications: hyperthermia (serotonin syndrome mechanism — can reach 42°C, often in setting of exertion + warm environment), hyponatremia (SIADH + excessive free water intake — can cause cerebral edema, seizures, death), and serotonin syndrome. Treatment: aggressive cooling (evaporative, ice packs; dantrolene if rigid), benzodiazepines, cyproheptadine for serotonin syndrome, hypertonic saline (3% NaCl 100–150 mL bolus) for symptomatic hyponatremia with seizures.
Bath Salts (Synthetic Cathinones)
Mephedrone, methylone, MDPV — produce a combined sympathomimetic/serotonergic toxidrome. Excited delirium is a hallmark: extreme agitation, hyperthermia, diaphoresis, paranoia, superhuman strength, and sudden cardiac arrest. Treatment: aggressive sedation with benzodiazepines (large doses often required — diazepam 40–80 mg or midazolam 20–40 mg total), active cooling, IV fluids, ketamine 4 mg/kg IM for refractory agitation.
17 Cannabis, Hallucinogens & Novel Substances
Cannabinoid Hyperemesis Syndrome (CHS)
Cyclic vomiting in chronic cannabis users (typically daily use for ≥1 year). Classic features: severe episodic nausea/vomiting, relief with hot showers/baths (pathognomonic behavior), diffuse abdominal pain. Diagnosis of exclusion. Treatment: cessation of cannabis (only definitive therapy), topical capsaicin 0.075% cream applied to the abdomen (activates TRPV1 receptors, paradoxically relieves symptoms), haloperidol 5 mg IV (superior to ondansetron in studies), IV fluids.
Synthetic Cannabinoids (K2/Spice)
Full agonists at CB1/CB2 receptors (unlike THC, which is a partial agonist) — much more potent and dangerous than natural cannabis. Effects: severe agitation, psychosis, seizures, tachycardia, hypertension, acute kidney injury (some formulations), coagulopathy (brodifacoum-contaminated batches — treat with vitamin K). Treatment: benzodiazepines, supportive care.
PCP (Phencyclidine)
NMDA receptor antagonist. Produces dissociative state with: vertical nystagmus (nearly pathognomonic), agitation, violence, analgesia (may not respond to pain compliance), rhabdomyolysis, seizures, hyperthermia. Treatment: benzodiazepines for agitation and seizures, physical restraint with caution (risk of death from rhabdomyolysis with prolonged struggle), active cooling, IV fluids for rhabdomyolysis, monitor CK and renal function.
Other Substances
| Substance | Key Features | Treatment |
|---|---|---|
| LSD/Psilocybin | Sympathomimetic signs, hallucinations, synesthesia; rarely life-threatening; risk of dangerous behavior during "bad trip" | Reassurance, benzodiazepines for agitation; supportive |
| GHB | CNS depression, bradycardia, respiratory depression; rapid onset and recovery (hours); combined with other CNS depressants → life-threatening | Airway management; supportive; most patients awaken within 2–6 hours |
| Kratom | Low-dose stimulant, high-dose opioid-like effects; seizures, liver toxicity reported; withdrawal syndrome | Supportive; naloxone for opioid-like effects; benzodiazepines for seizures |
| Fentanyl-laced substances | Any drug of abuse may contain illicit fentanyl; presents as unexpected opioid toxidrome in stimulant users | Naloxone; extended observation; multiple doses often needed |
18 Carbon Monoxide Poisoning
Carbon monoxide (CO) binds hemoglobin with 240× the affinity of oxygen, forming carboxyhemoglobin (COHb) and shifting the oxyhemoglobin dissociation curve leftward (impaired O2 unloading to tissues). CO also binds myoglobin and mitochondrial cytochrome oxidase, directly impairing cellular respiration. Sources: house fires (most common), space heaters, car exhaust in enclosed garages, charcoal grills indoors, gas-powered generators, propane-powered forklifts, and methylene chloride (paint stripper — metabolized to CO in the liver, causing delayed and prolonged COHb elevation).
COHb Levels and Symptoms
| COHb Level | Symptoms | Notes |
|---|---|---|
| < 10% | Often asymptomatic; headache in some | Smokers may have baseline 5–10% |
| 10–20% | Headache, dizziness, nausea, exertional dyspnea | Often misdiagnosed as viral illness or migraine |
| 20–30% | Severe headache, confusion, visual disturbances, tachycardia | Significant impairment |
| 30–40% | Syncope, tachypnea, chest pain, cognitive dysfunction | Risk of myocardial ischemia |
| 40–60% | Seizures, coma, cardiovascular collapse | High mortality |
| > 60% | Rapidly fatal | Rare to survive |
Cherry-red skin is classically described but is an unreliable and late finding, more often seen postmortem. Pulse oximetry is unreliable in CO poisoning — COHb is read as oxyhemoglobin by standard pulse oximeters, showing falsely normal SpO2. Co-oximetry (ABG with co-oximetry) is required.
CO Elimination Half-Lives
Room air (FiO2 21%): COHb half-life ~320 minutes. 100% O2 via NRB mask: half-life ~80 minutes. Hyperbaric oxygen (HBO) at 2.5–3 ATA: half-life ~23 minutes. All patients with suspected CO poisoning should receive 100% O2 immediately; do not wait for lab confirmation.
HBO Indications
Evidence is mixed, but generally accepted indications include: loss of consciousness (at any time during exposure), COHb > 25% (some use > 20%), pregnancy (fetal hemoglobin has higher CO affinity), cardiac ischemia or arrhythmia, persistent neurologic symptoms despite 100% O2, and severe metabolic acidosis. HBO may reduce delayed neuropsychiatric sequelae (DNS — cognitive impairment, personality changes, parkinsonian features appearing 2–40 days after exposure).
Delayed Neuropsychiatric Sequelae (DNS)
DNS affects 10–30% of CO-poisoned patients, typically presenting 2–40 days after apparent recovery. Features include: cognitive impairment (memory deficits, difficulty concentrating), personality changes (irritability, depression), parkinsonian features (gait disturbance, masked facies), and demyelination visible on MRI (periventricular white matter changes, globus pallidus lesions). Risk factors for DNS: loss of consciousness during exposure, COHb > 25%, age > 36, prolonged exposure duration. HBO therapy may reduce DNS incidence, though evidence remains debated. There is no specific treatment once DNS develops; symptoms may partially resolve over months but can be permanent in up to 25% of affected patients.
Special Populations in CO Poisoning
| Population | Key Considerations | Management Differences |
|---|---|---|
| Pregnant women | Fetal hemoglobin binds CO with 60% greater affinity than adult Hb; fetal COHb levels may be 10–15% higher than maternal levels; fetal elimination half-life is 2–3 times longer | Lower threshold for HBO (some recommend for any symptomatic exposure); treat until maternal COHb < 5%; fetal monitoring throughout |
| Children | Higher metabolic rate increases susceptibility; nonspecific symptoms (irritability, poor feeding) may delay diagnosis; higher surface area to body mass ratio | HBO if any neurologic symptoms or LOC; lower threshold for treatment than adults |
| Cardiac patients | Myocardial ischemia may occur at lower COHb levels (10–15%) due to reduced cardiac reserve; CO directly toxic to myocardium | Troponin and ECG for all cardiac patients with CO exposure; lower threshold for HBO |
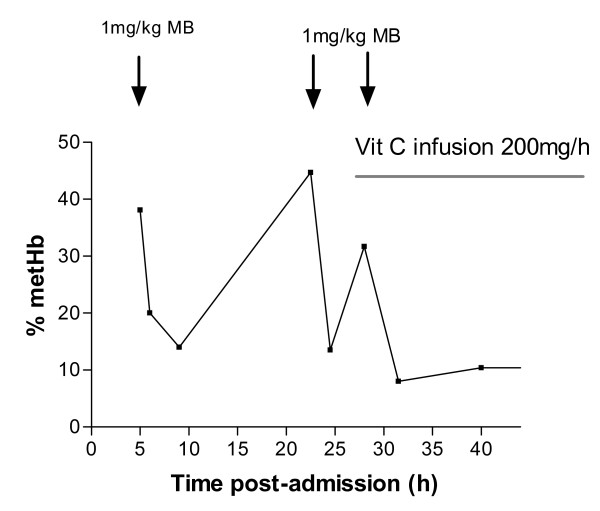
Methemoglobinemia
While not strictly CO poisoning, methemoglobinemia is an important environmental/drug-induced hemoglobinopathy. Methemoglobin (Fe3+) cannot bind oxygen. Causes: dapsone, benzocaine/lidocaine (topical anesthetics), nitrites, aniline dyes, nitrobenzene. Symptoms correlate with MetHb level: <15% typically asymptomatic; 15–20% chocolate-brown blood (does not turn red with O2 exposure), cyanosis unresponsive to supplemental O2; 20–50% dyspnea, headache, tachycardia; >50% seizures, coma, cardiovascular collapse; >70% lethal. Treatment: methylene blue 1–2 mg/kg IV over 5 minutes (acts as electron carrier, reducing MetHb to Hb via NADPH-methemoglobin reductase). Contraindicated in G6PD deficiency (causes hemolytic crisis). Pulse oximetry is unreliable — tends to read ~85% regardless of actual saturation at high MetHb levels.
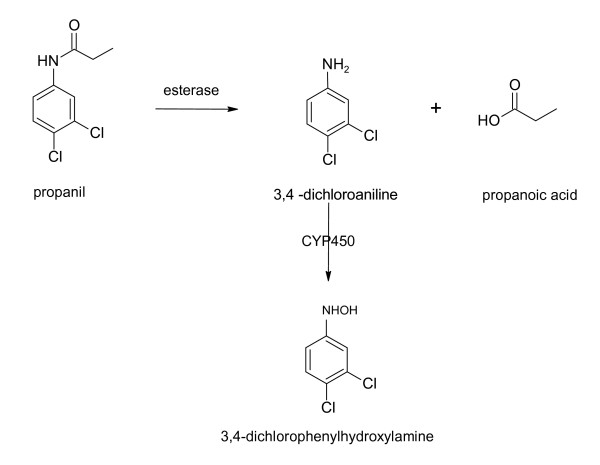
19 Organophosphate & Nerve Agent Poisoning
Organophosphates (OPs) irreversibly inhibit acetylcholinesterase (AChE), causing accumulation of acetylcholine at muscarinic, nicotinic, and CNS synapses. Sources: agricultural pesticides (malathion, parathion, chlorpyrifos), nerve agents (sarin, VX, novichok). The OP-AChE bond "ages" over time (hours to days depending on agent), becoming irreversible and resistant to oxime reactivation.
Clinical Presentation — Cholinergic Crisis
Muscarinic effects (SLUDGE/DUMBBELSS — see Section 2): salivation, lacrimation, urination, defecation, GI cramping, emesis, bronchospasm, bronchorrhea, miosis, bradycardia. Nicotinic effects: fasciculations, muscle weakness, paralysis (including respiratory muscles), tachycardia, hypertension. CNS effects: anxiety, seizures, coma, central apnea.
Intermediate Syndrome
Occurs 24–96 hours after acute exposure, after initial cholinergic crisis has resolved. Characterized by weakness of proximal muscles, neck flexors, respiratory muscles, and cranial nerve-innervated muscles. Mechanism: persistent AChE inhibition at nicotinic junctions. May require prolonged ventilatory support. Not prevented by atropine or pralidoxime.
Organophosphate-Induced Delayed Neuropathy (OPIDN)
Occurs 1–3 weeks after exposure. Distal sensorimotor polyneuropathy from inhibition of neuropathy target esterase (NTE). Presents as stocking-glove numbness progressing to weakness and ataxia. May be permanent. Not prevented by antidotes.
Treatment
Decontamination: Remove clothing, wash skin with soap and water; PROTECT responders with PPE (skin absorption risk). Atropine: 2–4 mg IV (pediatric: 0.05 mg/kg, minimum 0.1 mg) — no dose ceiling; double dose every 5 minutes until secretions dry. May require 10–100+ mg total. Titrate to drying of bronchial secretions (NOT pupil size or heart rate). Pralidoxime (2-PAM): 1–2 g IV over 15–30 minutes, then 500 mg/hr infusion. Most effective within 24–48 hours before AChE-OP bond ages. Reactivates AChE at nicotinic junctions (atropine alone does not reverse nicotinic effects). Benzodiazepines: Diazepam 5–10 mg IV for seizures. Mark I kit (military autoinjector): contains atropine 2 mg + pralidoxime 600 mg for field use by first responders.
Carbamate vs Organophosphate Poisoning
| Feature | Organophosphate | Carbamate |
|---|---|---|
| AChE binding | Irreversible (undergoes "aging") | Reversible (spontaneous regeneration in hours) |
| Duration of toxicity | Days to weeks; may require prolonged atropine | Hours to 1–2 days; shorter clinical course |
| CNS penetration | Most penetrate CNS → seizures, coma | Most do not penetrate CNS (quaternary ammonium) |
| Pralidoxime | Indicated (especially within 24–48 hours before aging) | Generally not indicated (spontaneous AChE recovery); not harmful if given empirically |
| Clinical significance | Higher mortality; prolonged ICU stay common | Generally better prognosis with supportive care |
20 Heavy Metals
Lead Poisoning
| Blood Lead Level (BLL) | Age Group | Clinical Effects | Management |
|---|---|---|---|
| ≥3.5 mcg/dL | Children | CDC reference value (previously 5, revised 2021); triggers investigation | Environmental assessment, source removal, nutritional counseling |
| 20–44 mcg/dL | Children | Cognitive impairment, behavioral changes, anemia, abdominal pain | Environmental remediation, close follow-up; consider chelation if 20–44 with symptoms |
| 45–69 mcg/dL | Children | Above + peripheral neuropathy, nephropathy, wrist/foot drop | Succimer (DMSA) 10 mg/kg PO TID × 5 days, then BID × 14 days |
| ≥70 mcg/dL | Children | Encephalopathy: seizures, coma, cerebral edema, death | CaEDTA 1000–1500 mg/m2/day IV + BAL (dimercaprol) 75 mg/m2 IM q4h — start BAL first (BAL prevents CaEDTA from redistributing lead to brain) |
| > 50 mcg/dL | Adults | Abdominal colic, peripheral motor neuropathy, gout, nephropathy, basophilic stippling on smear | Succimer or CaEDTA; remove from exposure |
| > 100 mcg/dL | Adults | Encephalopathy | CaEDTA + BAL as above |
Arsenic Poisoning
Acute: profuse watery diarrhea ("rice water"), abdominal pain, hypotension, multi-organ failure, QTc prolongation. Chronic: Mees lines (transverse white lines on nails), peripheral neuropathy, skin changes (rain-drop pattern hyperpigmentation, keratoses), increased cancer risk (lung, bladder, skin). Diagnosis: 24-hour urine arsenic (> 50 mcg/L is elevated; specify inorganic arsenic — seafood causes false elevation). Treatment: BAL (dimercaprol) 3–5 mg/kg IM q4h for severe acute poisoning, then transition to succimer (DMSA) PO for continued chelation. Unithiol (DMPS) is an alternative chelator.
Mercury
| Form | Source | Toxicity | Chelation |
|---|---|---|---|
| Elemental | Thermometers, dental amalgam | Inhalation: pneumonitis, CNS (erethism: tremor, insomnia, personality change); ingestion: benign (not absorbed) | Succimer or unithiol if symptomatic |
| Inorganic | Batteries, disinfectants | Corrosive GI injury, renal tubular necrosis | Succimer or unithiol; BAL for severe cases |
| Organic (methylmercury) | Fish consumption, industrial | CNS: paresthesias, ataxia, visual field constriction, hearing loss (Minamata disease); crosses placenta | Succimer (limited efficacy for organic forms) |
Iron Poisoning
Ingestion of ≥20 mg/kg elemental iron is potentially toxic; ≥60 mg/kg is potentially lethal. Five stages:
| Stage | Time | Features |
|---|---|---|
| I | 0–6 hours | GI: vomiting, diarrhea (often bloody), abdominal pain |
| II | 6–24 hours | Apparent improvement ("quiescent phase") — deceiving |
| III | 12–48 hours | Systemic toxicity: shock, metabolic acidosis, hepatic failure, coagulopathy |
| IV | 2–5 days | Hepatic necrosis (hepatotoxicity peaks) |
| V | 2–8 weeks | GI scarring and strictures (pyloric obstruction) |
Treatment: WBI for large ingestions (iron not adsorbed by charcoal). Deferoxamine 15 mg/kg/hr IV (maximum 6–8 g/day) for serum iron > 500 mcg/dL, significant symptoms, or clinical deterioration. Classic finding: "vin rosé" urine (pinkish-orange discoloration from ferrioxamine complex). Stop deferoxamine when urine color normalizes, symptoms resolve, and AG closes.
Thallium
Colorless, odorless, tasteless — historically used as rat poison and in homicides. Presents with GI symptoms followed by painful ascending neuropathy and alopecia (7–14 days post-ingestion, pathognomonic). Treatment: Prussian blue (ferric hexacyanoferrate) 250 mg/kg/day PO in divided doses — binds thallium in the GI tract and interrupts enterohepatic recirculation. MDAC may augment elimination.
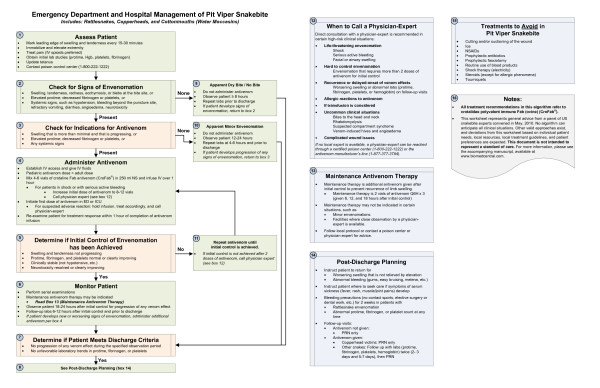
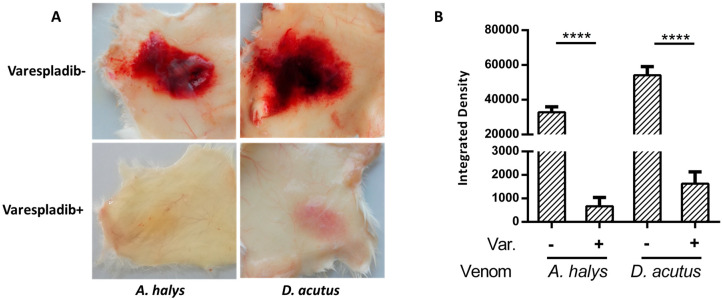
21 Envenomation
Pit Viper (Crotalid) Envenomation
Rattlesnakes, copperheads, and cottonmouths account for >95% of venomous snakebites in the US. Venom contains hemotoxins, cytotoxins, and myotoxins causing local tissue destruction, coagulopathy (thrombocytopenia, hypofibrinogenemia, elevated PT/INR), and compartment syndrome.
| Severity | Features | Antivenom |
|---|---|---|
| Minimal | Local pain/swelling < 1 joint space, no systemic symptoms, normal labs | Observation × 8–12 hours; antivenom if progression |
| Moderate | Progressive swelling crossing ≥1 joint, mild coagulopathy, no life-threatening signs | CroFab 4–6 vials IV over 1 hour; recheck labs at 1 hour; redose if not controlled. OR Anavip 10 vials IV. |
| Severe | Rapid swelling, severe coagulopathy (INR > 3, platelets < 50K, fibrinogen < 100), hypotension, respiratory compromise | CroFab 8–12 vials initially; additional 4–6 vials q1h until control; maintenance dosing. Anavip 10 vials, repeat as needed. |
Coral Snake
Elapid envenomation with neurotoxic venom (postsynaptic nicotinic acetylcholine receptor blockade). Delayed toxicity is the hallmark — symptoms may not appear for 6–18 hours but then progress rapidly to bulbar weakness, diplopia, dysphagia, and respiratory paralysis. Once paralysis develops it is NOT reversible with antivenom. Antivenom (Pfizer Coral Snake Antivenin) must be given prophylactically if a confirmed coral snake bite with fang marks is identified, even if asymptomatic. 4–6 vials IV. Red-on-yellow rule (North America only): "red on yellow, kill a fellow; red on black, venom lack."
Spider & Scorpion Envenomation
| Species | Syndrome | Treatment |
|---|---|---|
| Black Widow (Latrodectus) | Latrodectism: severe muscle cramping, abdominal rigidity (mimics acute abdomen), hypertension, diaphoresis, tachycardia | Benzodiazepines, IV calcium gluconate (limited evidence), opioids for pain; antivenom (Latrodectus mactans antivenin) for severe cases (hypertensive emergency, intractable pain, pregnancy) |
| Brown Recluse (Loxosceles) | Local necrotic wound with "red, white, and blue" sign (central pallor, surrounding ischemia, erythema); rarely systemic hemolysis, DIC, renal failure | Supportive: wound care, elevation, tetanus prophylaxis; dapsone controversial; no antivenom available in US; surgery for necrotic ulcers only after demarcation (weeks) |
| Bark Scorpion (Centruroides) | Neurotoxicity: opsoclonus (roving eye movements), muscle fasciculations, hypersalivation, respiratory distress (especially children) | Centruroides antivenom (Anascorp) 3 vials IV for significant neurotoxicity; benzodiazepines for muscle spasm; atropine for hypersalivation |
Marine Envenomation
Box jellyfish (Chironex fleckeri): tentacle contact causes excruciating pain, cardiovascular collapse, and death within minutes in severe cases. Remove tentacles with vinegar rinse (inactivates undischarged nematocysts). Antivenom available in Australia. Stonefish: intense pain from dorsal spine envenomation; hot water immersion (45°C for 30–90 minutes) denatures heat-labile toxin; stonefish antivenom for severe cases. General principle: hot water immersion for most marine stings (denatures thermolabile proteins in venom).
Snakebite Management Pearls
Do NOT: apply tourniquet, ice, electrical shock, incision/suction, or pressure-immobilization for pit viper bites (only for elapid bites). Do: remove jewelry from affected extremity (anticipate swelling), immobilize limb at level of heart, mark advancing edge of swelling with time stamp, obtain baseline labs (CBC, PT/INR, fibrinogen, BMP, CK, type and screen), and initiate antivenom early for progressive local effects or any systemic signs. Fasciotomy is rarely indicated — elevated compartment pressures are usually from tissue edema (responds to antivenom), not true compartment syndrome. Measure compartment pressures before considering surgical intervention.
22 Caustic Ingestions
Caustic ingestion causes direct chemical injury to the GI mucosa. Alkalis (pH > 12: NaOH, KOH, bleach, drain cleaners, button batteries) cause liquefactive necrosis — deep penetrating injury through esophageal wall. Acids (pH < 2: HCl, H2SO4, battery acid) cause coagulative necrosis — eschar formation limits depth of penetration; preferentially injure the stomach (acid passes quickly through the esophagus due to taste). Alkalis are generally more dangerous to the esophagus; acids to the stomach.
Critical Management Principles
Do NOT induce emesis (re-exposes mucosa to caustic agent). Do NOT administer activated charcoal (does not bind caustics, obscures endoscopic visualization). Do NOT attempt neutralization (exothermic reaction generates heat → additional thermal injury). Do NOT insert NG tube blindly (perforation risk). Do NOT delay endoscopy for imaging — proceed to EGD.
Zargar Classification (Endoscopic)
| Grade | Endoscopic Findings | Prognosis |
|---|---|---|
| 0 | Normal mucosa | No sequelae |
| I | Edema and erythema | Full recovery |
| IIa | Superficial ulceration, exudates, friability | Good; low stricture risk |
| IIb | Deep focal or circumferential ulceration | ~70% stricture rate; consider stent/serial dilation |
| IIIa | Focal necrosis (small scattered areas of black/grey discoloration) | High stricture and perforation risk |
| IIIb | Extensive necrosis (large areas of necrosis through esophageal wall) | Perforation, mediastinitis, death; may require esophagectomy |
Endoscopy timing: within 12–24 hours of ingestion (ideally within 12 hours). Avoid between 5–15 days post-ingestion (highest perforation risk due to weakened tissue). Grade IIb and higher require ICU admission, surgical consultation, and close monitoring for perforation. Long-term complications include esophageal stricture (may require repeated dilation or esophageal replacement), increased lifetime risk of esophageal squamous cell carcinoma (1000-fold increased risk with alkali injury), and pyloric stenosis after acid ingestion.
Specific Caustic Agents
| Agent | pH / Type | Common Source | Unique Features |
|---|---|---|---|
| Sodium hydroxide (NaOH) | pH 14 / Alkali | Drain cleaners (Drano), oven cleaners | Most severe esophageal injury; liquefactive necrosis penetrates full thickness within seconds |
| Hydrofluoric acid (HF) | pH ~1 / Acid | Rust removers, glass etching, industrial | Fluoride ion penetrates tissues causing deep burns beyond visible area; binds calcium → hypocalcemia → cardiac arrest. Treat with topical calcium gluconate gel 2.5%, IV calcium, intra-arterial calcium for digit exposure |
| Sulfuric acid (H2SO4) | pH < 1 / Acid | Car batteries, drain cleaners | Severe exothermic reaction with water; primarily gastric injury; coagulative necrosis |
| Bleach (NaOCl) | pH 11–13 / Alkali | Household cleaners | Dilute household bleach (3–6%): mild irritation only, no endoscopy needed. Concentrated industrial bleach (> 10%): significant injury possible |
| Ammonia | pH 11–12 / Alkali | Household cleaners | Primarily inhalation injury (pulmonary edema, airway burns); ingestion causes esophageal injury similar to other alkalis |
Button Battery Ingestion
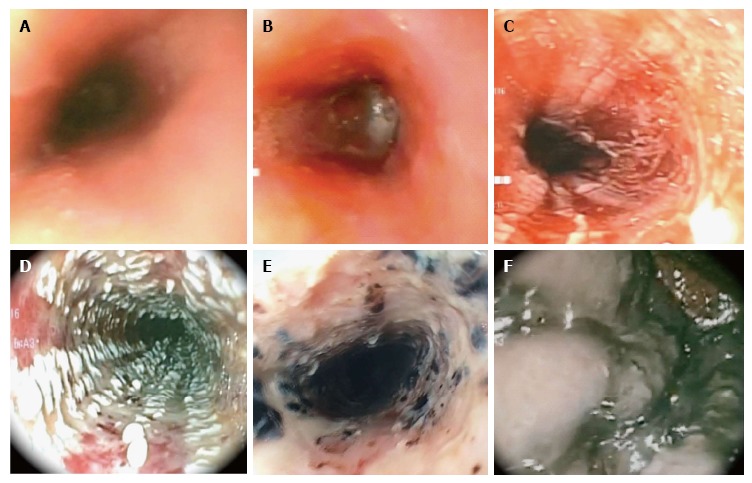
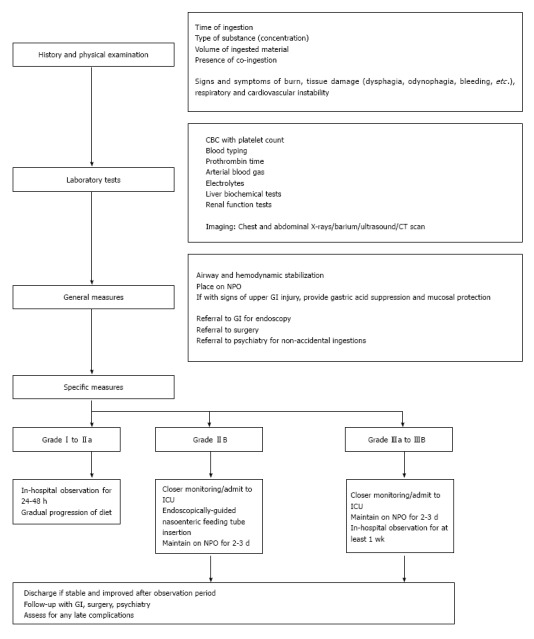
Button batteries lodged in the esophagus cause severe injury within 2 hours via hydroxide generation from electrical current (alkaline necrosis). Management: AP/lateral chest X-ray to confirm location; if esophageal, emergent endoscopic removal < 2 hours. Pre-removal temporizing measure: honey 10 mL PO q10 min (children > 12 months) or sucralfate may reduce injury progression while awaiting endoscopy (2018 NCPC guidance). If in the stomach and the child is ≥5 years old and the battery is < 20 mm, observation with repeat imaging in 48 hours is acceptable. If battery > 20 mm or child < 5 years, removal is recommended. Post-removal: monitor for delayed complications including tracheoesophageal fistula, aortoesophageal fistula (fatal hemorrhage — may present as sentinel bleed days to weeks later), and mediastinitis.
23 Cardiac Drug Toxicity
Beta-Blocker Overdose
Presents with bradycardia, hypotension, heart block, and hypoglycemia (especially propranolol). Propranolol also causes sodium channel blockade (QRS widening) and seizures. CNS-penetrant agents (propranolol, labetalol) are most lethal.
1. Atropine 0.5–1 mg IV for bradycardia (often ineffective). 2. Glucagon 3–5 mg IV bolus (up to 10 mg), then infusion 2–5 mg/hr — bypasses beta-receptor blockade via activation of adenylyl cyclase (Gs coupling). 3. High-dose insulin euglycemia (HIE) therapy: regular insulin 1 unit/kg IV bolus, then 1–10 units/kg/hr infusion + dextrose (D10W or D25W) to maintain glucose 100–200 mg/dL + K+ supplementation. Mechanism: shifts myocardial metabolism from fatty acid to glucose oxidation, improving contractility. 4. Vasopressors (norepinephrine/epinephrine). 5. Intralipid emulsion 20% (for lipophilic agents). 6. ECMO/mechanical support for refractory shock.
Calcium Channel Blocker Overdose
Dihydropyridine CCBs (nifedipine, amlodipine) cause vasodilation with reflex tachycardia. Non-dihydropyridine CCBs (verapamil, diltiazem) cause bradycardia, heart block, and negative inotropy. Both cause severe hypotension and hyperglycemia (blocked pancreatic insulin release) in overdose.
1. Calcium chloride 1–2 g IV (via central line; calcium gluconate 3–6 g if peripheral access only) q15–20 min up to 3–4 doses; monitor ionized calcium (target 2× normal). 2. HIE therapy — most effective intervention for CCB overdose: insulin 1 U/kg bolus then 1–10 U/kg/hr infusion; D10W or D25W to maintain glucose 100–200; monitor glucose q15–30 min initially then q1h; replace potassium aggressively. 3. Vasopressin 0.04 units/min infusion (acts via V1 receptors independent of calcium channels). 4. Norepinephrine infusion (may require high doses, 0.5–2 mcg/kg/min). 5. Intralipid emulsion 20% for lipophilic CCBs (verapamil, diltiazem). 6. Transcutaneous or transvenous pacing for refractory bradycardia. 7. ECMO or mechanical circulatory support for refractory shock.
Digoxin Toxicity
Classic features: GI (nausea, vomiting, anorexia), visual disturbances (yellow-green halos — xanthopsia), and nearly any arrhythmia (classically atrial tachycardia with AV block or bidirectional VT). Hyperkalemia is a marker of severity (Na/K-ATPase inhibition prevents cellular K+ uptake).
Indications: Life-threatening arrhythmias, hyperkalemia (K+ > 5.5 mEq/L), hemodynamic instability, serum digoxin level > 10 ng/mL (acute) or > 6 ng/mL (chronic) with symptoms. Vial calculation: Number of vials = (serum digoxin [ng/mL] × body weight [kg]) / 100. For known ingestion amount: number of vials = total digitalis body load (mg) / 0.5. Empiric dosing for cardiac arrest: 10–20 vials. Each vial binds ~0.5 mg digoxin. Note: after DigiFab, total digoxin levels are meaningless (measures bound + free) — monitor free digoxin if available.
Sodium Channel Blocker Toxicity (General)
Drugs with sodium channel blocking properties: TCAs, cocaine, class IA/IC antiarrhythmics (flecainide, procainamide), propranolol, diphenhydramine, carbamazepine, lamotrigine. ECG hallmark: QRS widening. Treatment: NaHCO3 1–2 mEq/kg IV bolus (sodium loading + alkalemia), targeting QRS < 120 ms and serum pH 7.45–7.55.
Intralipid Emulsion (ILE) Therapy
Indication: cardiovascular collapse from lipophilic drug toxicity (local anesthetics, TCAs, calcium channel blockers, beta-blockers) refractory to standard resuscitation. Dose: 20% lipid emulsion 1.5 mL/kg IV bolus over 1 minute, followed by 0.25 mL/kg/min infusion for 30–60 minutes. May repeat bolus 1–2 times for persistent cardiovascular collapse. Maximum total dose: 12.5 mL/kg in the first 30 minutes. Mechanism: "lipid sink" theory — lipid phase in plasma sequesters lipophilic drug from cardiac tissue; also provides metabolic substrate to myocardium and increases intracellular calcium. Agents with strongest evidence: bupivacaine (most data), TCAs, verapamil/diltiazem, beta-blockers (propranolol, atenolol). Monitor for pancreatitis and lipemia-induced laboratory interference with prolonged infusions.
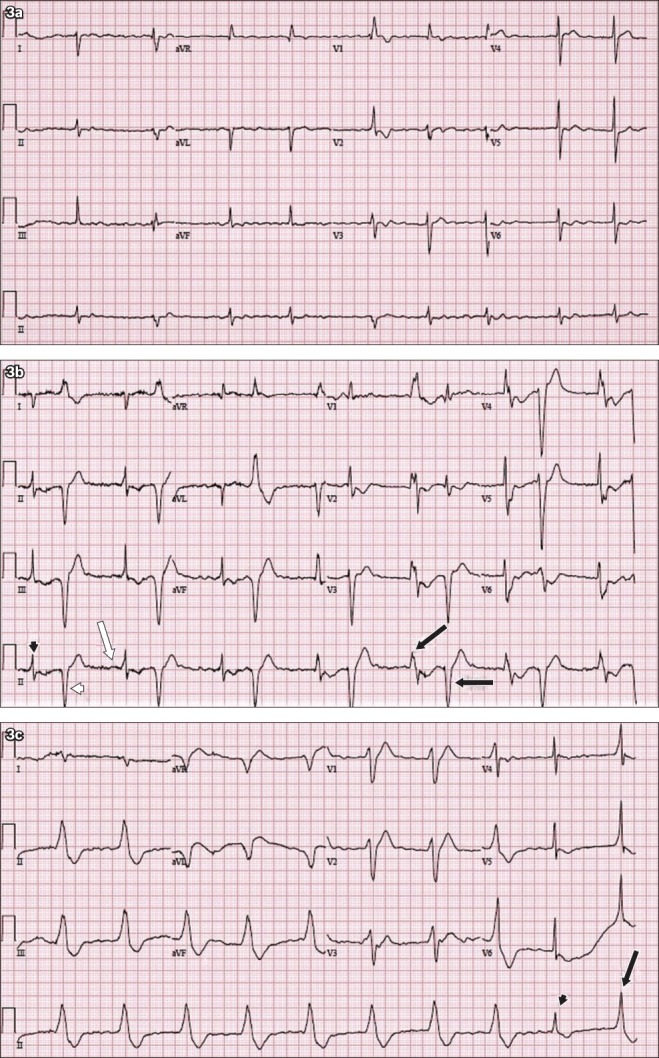
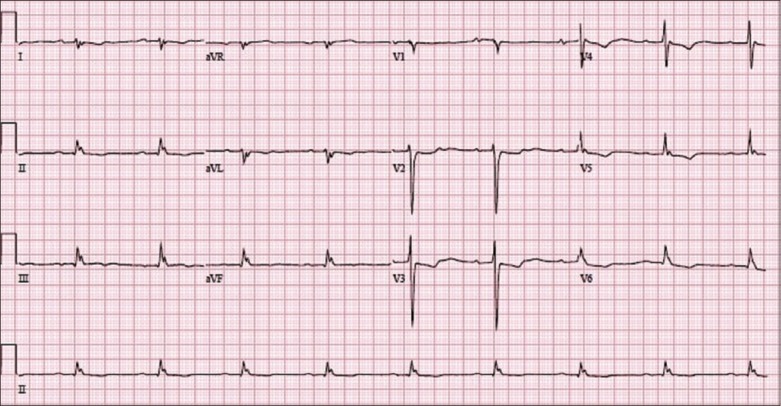
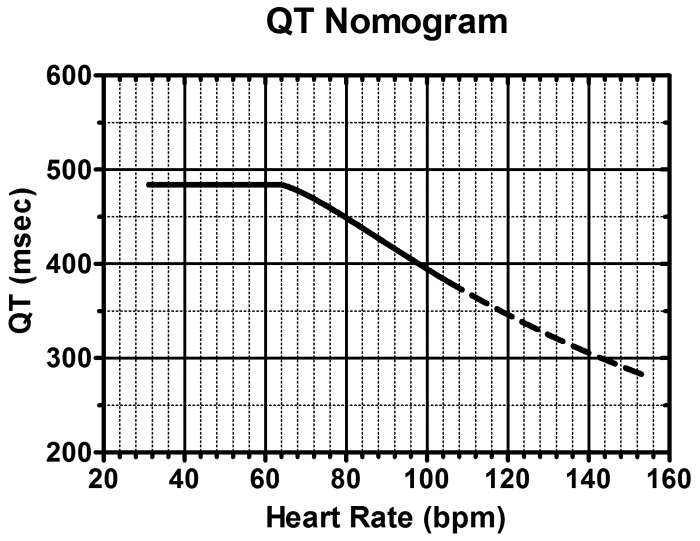
24 Cyanide Poisoning
Cyanide binds mitochondrial cytochrome oxidase a3, blocking the electron transport chain and halting aerobic metabolism. Cells switch to anaerobic glycolysis, producing profound lactic acidosis despite adequate oxygen delivery (PaO2 and SaO2 are normal). Sources: house fire smoke inhalation (most common — combustion of synthetic materials), industrial (electroplating, mining, laboratories), sodium nitroprusside infusion (releases cyanide), ingestion of cyanogenic plants (bitter almonds, cassava, apricot pits).
Clinical Features
Rapid onset: headache, confusion, seizures, cardiovascular collapse, death. Classic finding: bright red venous blood (cells cannot extract oxygen → high venous PO2). Bitter almond odor (only ~50% of population can detect due to genetic variation). In fire victims, suspect cyanide when: persistent lactic acidosis despite adequate oxygen delivery, COHb-corrected lactate > 8 mmol/L, or altered mental status disproportionate to COHb level.
Cyanide Toxicity by Exposure Route
| Route | Onset | Duration | Key Considerations |
|---|---|---|---|
| Inhalation (HCN gas) | Seconds to minutes | Rapid progression to cardiovascular collapse | Immediate removal from exposure; highest lethality; common in house fires |
| Ingestion (cyanide salts) | Minutes to hours | Delayed by food in stomach; vomiting may limit absorption | Activated charcoal if very early (< 1 hour); GI decontamination of limited utility once absorbed |
| Nitroprusside infusion | Hours to days | Progressive accumulation during prolonged infusion | Monitor for rising lactate, tachyphylaxis to nitroprusside; co-administer sodium thiosulfate prophylactically; risk increases with infusion > 2 mcg/kg/min for > 24–48 hours |
| Dermal absorption | Minutes to hours | Depends on concentration and surface area | Decontaminate skin immediately; responder PPE critical |
Laboratory Findings
Hallmark: severe lactic acidosis with elevated mixed venous oxygen saturation (SvO2 > 90%) — cells cannot utilize delivered oxygen. Additional findings: narrowed arteriovenous oxygen difference (A-V O2 gradient < 10 mmHg), elevated lactate (> 8 mmol/L strongly suggestive in fire victims), anion gap metabolic acidosis. Whole blood cyanide levels > 0.5 mg/L are toxic, > 3.0 mg/L often lethal — but results are rarely available in time to guide acute management. Point-of-care lactate is the most useful surrogate marker: lactate > 10 mmol/L in a smoke inhalation victim has high sensitivity for concurrent cyanide poisoning.
Treatment
| Antidote | Dose | Mechanism | Notes |
|---|---|---|---|
| Hydroxocobalamin (Cyanokit) | 5 g IV over 15 minutes (adults); may repeat once | Binds cyanide to form cyanocobalamin (vitamin B12), excreted renally | First-line in fire victims; safe in CO co-poisoning (does not reduce O2 carrying capacity); turns skin/urine red; interferes with colorimetric lab assays for 24–48 hours |
| Sodium thiosulfate | 12.5 g IV over 10–20 minutes | Provides sulfur substrate for rhodanese enzyme to convert cyanide to thiocyanate | Slower onset; often given with hydroxocobalamin; safe adjunct |
| Amyl nitrite | Inhale crushed ampule for 30 seconds per minute | Induces methemoglobinemia; methemoglobin binds cyanide (cyanmethemoglobin) | Field use only (bridge until IV access); contraindicated in fire victims (worsens O2 carrying capacity when combined with CO) |
| Sodium nitrite | 300 mg (10 mL of 3% solution) IV over 5 minutes | Methemoglobin formation (as above) | Risk of hypotension and excessive methemoglobinemia; avoid in fire victims; older Lilly cyanide kit component |
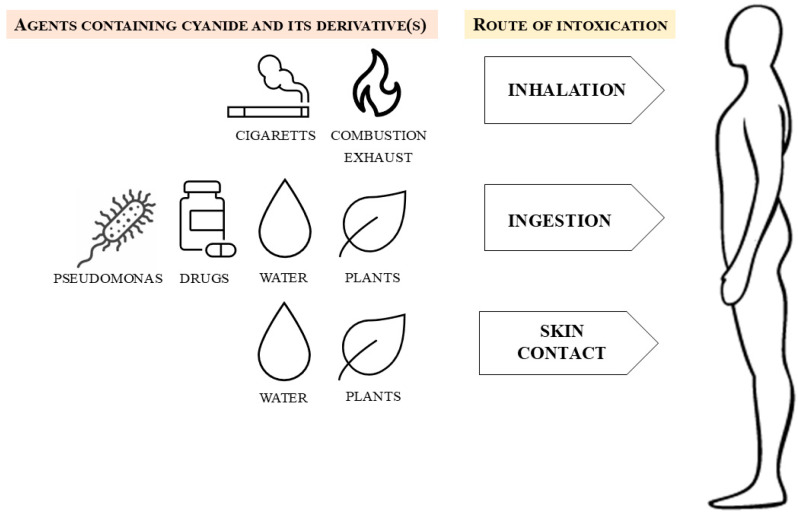
25 Withdrawal Syndromes
Alcohol Withdrawal
Onset 6–24 hours after last drink. Spectrum: tremulousness (6–24h) → withdrawal seizures (12–48h) → alcoholic hallucinosis (12–48h) → delirium tremens (48–96h, mortality 5–15% if untreated). Monitor with CIWA-Ar (Clinical Institute Withdrawal Assessment — Alcohol, revised) — scores nausea, tremor, paroxysmal sweats, anxiety, agitation, tactile/auditory/visual disturbances, headache, orientation. CIWA ≥8 triggers treatment; ≥20 indicates severe withdrawal.
Benzodiazepines are the mainstay. Symptom-triggered dosing (CIWA-based) is preferred over fixed-schedule dosing — reduces total benzodiazepine dose and ICU length of stay. Diazepam 10–20 mg IV q5–15 min (long-acting, self-tapering) or lorazepam 2–4 mg IV q15–30 min (preferred in hepatic dysfunction — no active metabolites). For refractory DT: phenobarbital 130–260 mg IV q15–30 min as adjunct, or continuous propofol/midazolam infusion in ICU. Thiamine 500 mg IV before glucose. Folate 1 mg IV. Magnesium 2 g IV (chronic alcoholics are depleted).
Benzodiazepine Withdrawal
Similar to alcohol withdrawal (both are GABA-A agonists) but more protracted. Onset depends on half-life of the specific agent: short-acting (alprazolam, lorazepam) → 1–2 days; long-acting (diazepam, clonazepam) → 3–7 days. Features: anxiety, insomnia, tremor, perceptual disturbances, seizures. Treatment: prolonged taper using a long-acting benzodiazepine (diazepam or chlordiazepoxide), reducing by 10–25% per week. Rapid cessation can be fatal.
Opioid Withdrawal
Extremely uncomfortable but rarely life-threatening (unlike alcohol/benzodiazepine withdrawal). Onset: short-acting (heroin) 8–24 hours; long-acting (methadone) 24–72 hours. Features: mydriasis (opposite of intoxication), lacrimation, rhinorrhea, yawning, diarrhea, piloerection ("goosebumps"), myalgias, abdominal cramps, insomnia. Monitor with COWS (Clinical Opiate Withdrawal Scale).
| Treatment | Dose | Mechanism |
|---|---|---|
| Buprenorphine (Subutex/Suboxone) | 4–8 mg SL initially, titrate to 16–24 mg/day | Partial mu-agonist; treats withdrawal and provides maintenance therapy |
| Clonidine | 0.1–0.3 mg PO q6–8h | Alpha-2 agonist; reduces sympathetic outflow; treats autonomic symptoms |
| Loperamide | 4 mg PO initially, then 2 mg after each loose stool | Peripheral mu-agonist; treats diarrhea |
| Ondansetron | 4–8 mg IV/PO q8h | 5-HT3 antagonist; treats nausea/vomiting |
GHB Withdrawal
GHB withdrawal can be life-threatening and uniquely resistant to benzodiazepines. Onset: 1–6 hours after last use (very rapid). Features: tremor, tachycardia, hypertension, diaphoresis, psychosis, delirium, seizures. Treatment: high-dose benzodiazepines (may require enormous amounts), barbiturates (pentobarbital/phenobarbital) if refractory, propofol infusion for refractory cases. GHB withdrawal may last 5–15 days.
Baclofen Withdrawal
Abrupt discontinuation of baclofen (especially intrathecal pump failure) causes: seizures, spasticity rebound, psychosis, hyperthermia, rhabdomyolysis, and multi-organ failure mimicking NMS or malignant hyperthermia. Treatment: restart baclofen immediately (oral or via pump); benzodiazepines for seizures; cyproheptadine and dantrolene as adjuncts; ICU admission for severe cases.
Stimulant Withdrawal
Withdrawal from cocaine, amphetamines, and other stimulants is not life-threatening but causes significant dysphoria. The "crash" phase (hours to days) involves hypersomnia, increased appetite, psychomotor retardation, depression, and vivid dreams. The withdrawal phase (days to weeks) involves anhedonia, fatigue, intense cravings, and depression that may persist for months. Treatment is entirely supportive: safe environment, psychiatric evaluation for suicidal ideation, sleep hygiene, and outpatient substance use treatment referral. No pharmacotherapy has demonstrated consistent benefit for stimulant withdrawal, though modafinil and bupropion are sometimes used off-label for persistent fatigue and anhedonia.
Nicotine Withdrawal
Onset within 2–12 hours of last nicotine use. Features: irritability, anxiety, difficulty concentrating, increased appetite, insomnia, restlessness, depressed mood. Peak severity at 24–72 hours; most symptoms resolve within 2–4 weeks, but cravings may persist for months. Treatment: nicotine replacement therapy (patch, gum, lozenge, inhaler, nasal spray), varenicline (partial nicotinic agonist, most effective single agent), bupropion SR, or combination therapy. Acute nicotine poisoning (e-cigarette liquid ingestion in children) presents with cholinergic symptoms: vomiting, diarrhea, diaphoresis, followed by seizures and respiratory failure in severe cases.
Withdrawal Syndrome Comparison
| Feature | Alcohol | Benzodiazepine | Opioid | GHB |
|---|---|---|---|---|
| Onset | 6–24 hours | 1–7 days | 8–72 hours | 1–6 hours |
| Seizure risk | High (12–48h) | High | Rare | Moderate |
| Mortality risk | High (DT 5–15%) | High | Very low | High |
| First-line treatment | Benzodiazepines | Slow benzodiazepine taper | Buprenorphine | Benzodiazepines + barbiturates |
| Duration | 3–7 days | Weeks to months | 5–10 days | 5–15 days |
| Hallmark feature | Delirium tremens | Protracted insomnia/anxiety | Piloerection, mydriasis | Benzodiazepine resistance |
26 Antidotes Master Table
| Poison/Toxin | Antidote | Dose | Route |
|---|---|---|---|
| Acetaminophen | N-acetylcysteine (NAC) | 150 mg/kg load (IV) or 140 mg/kg load (PO) | IV or PO |
| Anticholinesterase/OP | Atropine + Pralidoxime | Atropine 2–4 mg; 2-PAM 1–2 g | IV |
| Benzodiazepines | Flumazenil | 0.2 mg IV q1min (max 3–5 mg) | IV |
| Beta-blockers | Glucagon | 3–5 mg IV bolus, infusion 2–5 mg/hr | IV |
| Calcium channel blockers | Calcium chloride + HIE | CaCl2 1–2 g; insulin 1 U/kg bolus | IV |
| Carbon monoxide | 100% oxygen / HBO | NRB mask or hyperbaric chamber | Inhalation |
| Cyanide | Hydroxocobalamin | 5 g IV over 15 min | IV |
| Digoxin | DigiFab | Vials = (level × weight) / 100 | IV |
| Ethylene glycol / Methanol | Fomepizole | 15 mg/kg IV load, then 10 mg/kg q12h | IV |
| Heparin | Protamine | 1 mg per 100 U heparin (last dose) | IV |
| Iron | Deferoxamine | 15 mg/kg/hr IV (max 6–8 g/day) | IV |
| Isoniazid | Pyridoxine (B6) | Gram-for-gram of INH ingested (or 5 g empiric) | IV |
| Lead (BLL ≥70) | BAL + CaEDTA | BAL 75 mg/m2 q4h; CaEDTA 1500 mg/m2/day | IM/IV |
| Lead (BLL 45–69) | Succimer (DMSA) | 10 mg/kg PO TID ×5d, then BID ×14d | PO |
| Methemoglobinemia | Methylene blue | 1–2 mg/kg IV over 5 min | IV |
| Opioids | Naloxone | 0.04–2 mg, titrate to respirations | IV/IM/IN |
| Salicylates | NaHCO3 (urinary alkalinization) | 150 mEq in 1 L D5W | IV |
| Serotonin syndrome | Cyproheptadine | 12 mg load, then 4 mg q6h | PO |
| Sulfonylureas | Octreotide | 50–100 mcg SQ/IV q6–8h | SQ/IV |
| TCAs (wide QRS) | NaHCO3 | 1–2 mEq/kg IV bolus | IV |
| Thallium | Prussian blue | 250 mg/kg/day PO in divided doses | PO |
| Warfarin (acute) | Vitamin K + 4F-PCC | Vit K 10 mg IV; PCC per INR | IV |
| Crotalid envenomation | CroFab or Anavip | CroFab 4–12 vials; Anavip 10 vials | IV |
| NMS | Dantrolene + Bromocriptine | Dantrolene 1–2.5 mg/kg; Bromocriptine 2.5 mg TID | IV/PO |
| Arsenic | BAL (dimercaprol) | 3–5 mg/kg IM q4h, then transition to succimer | IM/PO |
| Coral snake | Coral Snake Antivenin (Pfizer) | 4–6 vials IV; give prophylactically for confirmed bite | IV |
| Fluoride / Hydrofluoric acid | Calcium gluconate | Topical gel 2.5%, IV 10 mL 10% solution, intra-arterial for digits | Topical/IV/IA |
| Local anesthetic toxicity (LAST) | Intralipid emulsion 20% | 1.5 mL/kg bolus, then 0.25 mL/kg/min infusion | IV |
| Malignant hyperthermia | Dantrolene | 2.5 mg/kg IV q5 min until symptoms resolve (max 10 mg/kg) | IV |
| Mercury (inorganic) | Succimer (DMSA) or BAL | Succimer 10 mg/kg PO TID × 5d then BID × 14d | PO/IM |
27 Toxidrome Summary Table
| Toxidrome | HR | BP | Temp | Pupils | Skin | Mental Status | Bowel Sounds | Treatment |
|---|---|---|---|---|---|---|---|---|
| Sympathomimetic | ↑ | ↑ | ↑ | Mydriasis | Diaphoretic | Agitated | ↑ | Benzodiazepines, cooling |
| Anticholinergic | ↑ | ↑ | ↑ | Mydriasis | Dry, flushed | Delirium | ↓ | Physostigmine (if no TCA); benzodiazepines |
| Cholinergic | ↓ | Variable | Normal | Miosis | Diaphoretic | Confusion/coma | ↑↑ | Atropine + pralidoxime |
| Opioid | ↓ | ↓ | ↓ | Pinpoint | Normal | Obtunded/coma | ↓ | Naloxone |
| Sedative-Hypnotic | ↓ | ↓ | ↓ | Normal | Normal | Sedated/coma | ↓ | Supportive; flumazenil (limited) |
| Serotonin Syndrome | ↑ | ↑ | ↑↑ | Mydriasis | Diaphoretic | Agitation/clonus | ↑ | Cyproheptadine; benzodiazepines |
| NMS | ↑ | Labile | ↑↑ | Normal | Diaphoretic | AMS/rigidity | Normal/↓ | Dantrolene; bromocriptine |
| Sympatholytic | ↓ | ↓ | ↓ | Miosis (clonidine) | Normal | Sedated | ↓ | Supportive; glucagon (beta-blockers) |
28 Dialyzable Substances
Hemodialysis effectively removes substances that meet specific pharmacokinetic criteria. The ideal dialyzable toxin has: low molecular weight (< 500 Da, passes membrane pores easily), low volume of distribution (< 1 L/kg, drug remains in blood compartment), low protein binding (< 80%, free drug available for clearance), and high water solubility.
| Substance | MW (Da) | Vd (L/kg) | Protein Binding | Dialyzable? | EXTRIP Recommendation |
|---|---|---|---|---|---|
| Lithium | 7 | 0.7 | 0% | Yes (excellent) | Strong recommendation for severe toxicity |
| Methanol | 32 | 0.7 | 0% | Yes (excellent) | Strong recommendation: acidosis, visual symptoms |
| Ethylene glycol | 62 | 0.7 | 0% | Yes (excellent) | Strong recommendation: acidosis, renal failure |
| Salicylates | 138 | 0.2 | 50–90%* | Yes (good) | Strong recommendation for level > 100 mg/dL or severe symptoms |
| Theophylline | 180 | 0.5 | 40% | Yes (good) | Conditional recommendation for severe toxicity |
| Acetaminophen | 151 | 0.9 | < 25% | Yes (massive ingestion) | Suggested for massive overdose with mitochondrial dysfunction |
| Valproic acid | 144 | 0.1–0.5 | 80–90%* | Yes (in overdose) | Conditional recommendation: free fraction rises in OD |
| Phenobarbital | 232 | 0.5–0.6 | 45–50% | Yes (moderate) | Suggested for prolonged coma, level > 150 mcg/mL |
| Metformin | 129 | 1–5 | 0% | Yes (variable) | Suggested for severe lactic acidosis |
| Carbamazepine | 236 | 0.8–1.4 | 75% | Limited | Suggested only for life-threatening toxicity |
| Digoxin | 781 | 7 | 25% | No | Not recommended (high Vd, use DigiFab instead) |
| TCAs | ~280 | 10–50 | 90–95% | No | Not recommended (very high Vd and protein binding) |
*Protein binding for salicylates and valproic acid is concentration-dependent; binding decreases (and free fraction increases) in overdose, making dialysis more effective than predicted from therapeutic-level pharmacokinetics.
Substances NOT Effectively Dialyzed
The following commonly encountered toxins are not removed by hemodialysis due to unfavorable pharmacokinetic properties (high Vd, high protein binding, or large molecular weight): TCAs (Vd 10–50 L/kg, 90–95% protein bound), digoxin (Vd 7 L/kg; use DigiFab instead), benzodiazepines (high Vd, 85–99% protein bound), opioids (high Vd, variable protein binding; use naloxone), phenytoin (90% protein bound at therapeutic levels; MDAC is preferred), calcium channel blockers (high Vd, > 90% protein bound), beta-blockers (variable; propranolol is highly protein-bound, but atenolol may be dialyzable), and warfarin (99% protein bound; use vitamin K and PCC). Requesting dialysis for these substances wastes resources and delays appropriate antidotal therapy.
29 Scoring Systems
Rumack-Matthew Nomogram
Plots serum acetaminophen concentration versus time after a single, acute ingestion. The treatment line begins at 150 mcg/mL at 4 hours, 75 mcg/mL at 8 hours, 37.5 mcg/mL at 12 hours, and 4.7 mcg/mL at 24 hours (log-linear decline). Levels above the line indicate potential toxicity and warrant NAC therapy. The nomogram is valid only for: known single acute ingestion, level drawn between 4–24 hours post-ingestion, and non-extended-release formulations. It is NOT valid for chronic ingestions, staggered ingestions, unknown ingestion time, or repeated supratherapeutic ingestions (RSI) — for these, use clinical judgment, ALT/AST trends, and APAP concentration.
CIWA-Ar (Clinical Institute Withdrawal Assessment for Alcohol, Revised)
Ten domains, each scored 0–7 (except orientation, 0–4):
1. Nausea/vomiting (0–7). 2. Tremor (0–7). 3. Paroxysmal sweats (0–7). 4. Anxiety (0–7). 5. Agitation (0–7). 6. Tactile disturbances (0–7). 7. Auditory disturbances (0–7). 8. Visual disturbances (0–7). 9. Headache/fullness (0–7). 10. Orientation/clouding (0–4).
Interpretation: < 8: mild withdrawal, may not need pharmacotherapy. 8–15: moderate, symptom-triggered benzodiazepines. 16–20: severe, aggressive treatment. > 20: very severe, high risk for delirium tremens and seizures.
COWS (Clinical Opiate Withdrawal Scale)
Eleven items: resting pulse rate (0–4), diaphoresis (0–4), restlessness (0–5), pupil size (0–5), bone/joint aches (0–4), runny nose/tearing (0–4), GI upset (0–5), tremor (0–4), yawning (0–4), anxiety/irritability (0–4), gooseflesh skin (0–5).
Interpretation: 5–12: mild withdrawal. 13–24: moderate. 25–36: moderately severe. > 36: severe. COWS ≥8–12 is generally the threshold to initiate buprenorphine to avoid precipitated withdrawal.
Poisoning Severity Score (PSS)
| Grade | Severity | Description |
|---|---|---|
| 0 | None | No symptoms or signs |
| 1 | Minor | Mild, transient, spontaneously resolving symptoms |
| 2 | Moderate | Pronounced or prolonged symptoms requiring treatment |
| 3 | Severe | Life-threatening symptoms, significant residual disability |
| 4 | Fatal | Death |
The PSS is applied to each organ system independently (GI, respiratory, nervous, cardiovascular, metabolic, hepatic, renal, hematologic, muscular, dermal, eyes, ears) and the overall grade is the highest grade in any system. Used primarily in clinical research, poison center data reporting, and for standardized comparison of poisoning outcomes across studies. The PSS should be scored at the time of worst clinical severity during the acute episode. It is validated for acute exposures and is endorsed by the EAPCCT and the International Programme on Chemical Safety (IPCS).
Poison Control Resources
US Poison Control: 1-800-222-1222 (connects to regional Poison Control Center 24/7; available for both public and healthcare providers). TOXBASE (UK): online clinical toxicology database used by National Poisons Information Service. Poisindex (Micromedex): comprehensive toxicology database with product identification, substance-specific management, and ingredient information — standard reference in US emergency departments. EXTRIP (www.extrip-workgroup.org): evidence-based recommendations for extracorporeal treatment in poisoning. Medical Toxicology Consultation: available through Poison Control and at certified Poison Centers; board-certified medical toxicologists provide real-time phone consultation for complex cases.
King's College Criteria (Non-Acetaminophen ALF)
For reference, the non-acetaminophen criteria differ from acetaminophen-specific criteria (Section 6): INR > 6.5 (PT > 100 seconds) regardless of encephalopathy grade, OR any three of: age < 10 or > 40 years, non-A/non-B hepatitis or drug-induced etiology, jaundice > 7 days before encephalopathy onset, INR > 3.5, bilirubin > 17.5 mg/dL. These criteria help identify patients who should be urgently referred for liver transplant evaluation.
Glasgow Coma Scale (GCS) in Poisoning
While the GCS was developed for trauma, it is frequently used in toxicology to track mental status. A GCS ≤8 generally indicates the need for airway protection (intubation). However, GCS has limitations in poisoning: some substances cause dissociative states that confound scoring, and rapidly reversible causes (opioids with naloxone) may not require intubation despite low GCS.
Prescott Criteria for NAC Efficacy
Historically used to assess NAC treatment outcomes in APAP toxicity: AST/ALT peak values, time to peak transaminases, development of coagulopathy (INR > 1.3), and clinical encephalopathy. Patients treated with NAC within 8 hours of ingestion have < 5% risk of hepatotoxicity (AST > 1000 IU/L) and essentially 0% mortality from hepatic failure. Patients treated 16–24 hours post-ingestion still benefit from NAC, with reduced mortality and improved transplant-free survival compared to no treatment.
Severity Assessment in Common Poisonings
| Poisoning | Mild | Moderate | Severe/Life-Threatening |
|---|---|---|---|
| Acetaminophen | APAP level above treatment line, normal LFTs, asymptomatic | Rising AST/ALT, RUQ pain, INR 1.3–2.0 | AST > 10,000, INR > 6.5, encephalopathy, pH < 7.3, lactate > 3.5 |
| Salicylates | Tinnitus, mild hyperventilation, level 30–50 mg/dL | Agitation, diaphoresis, level 50–80 mg/dL | AMS, seizures, pulmonary edema, level > 80 mg/dL, pH < 7.2 |
| TCA | Sinus tachycardia, dry mouth, QRS < 100 ms | QRS 100–160 ms, hypotension responsive to fluids | QRS > 160 ms, ventricular arrhythmias, refractory hypotension, seizures |
| Beta-blocker | Mild bradycardia (HR 50–60), normotensive | HR < 50, hypotension responsive to fluids/glucagon | Cardiogenic shock, high-grade AV block, refractory hypotension, seizures |
| Toxic alcohols | Inebriation, elevated OG, normal pH | Mild acidosis, early symptoms (visual changes, flank pain) | pH < 7.1, blindness (methanol), renal failure (EG), coma |