Wound Care
Wound healing physiology, chronic wound management, pressure injury staging, diabetic foot ulcers, venous and arterial ulcers, negative pressure therapy, debridement techniques, and every classification system, dressing selection, and treatment algorithm across the full scope of wound care.
01 Wound Healing Physiology
Wound healing is a dynamic, highly orchestrated process involving the coordinated interaction of cells, growth factors, cytokines, and extracellular matrix components. Normal acute wound healing proceeds through four overlapping phases: hemostasis, inflammation, proliferation, and remodeling. Disruption at any phase leads to chronic non-healing wounds. Understanding this cascade is foundational to every clinical decision in wound care.
Phase 1 — Hemostasis (Seconds to Hours)
Immediately following tissue injury, vascular disruption triggers the coagulation cascade. Vasoconstriction occurs within seconds, mediated by thromboxane A2 and endothelin, reducing blood loss. Exposed subendothelial collagen activates platelets, which adhere via glycoprotein Ib-IX-V receptors and von Willebrand factor, then aggregate to form the initial platelet plug. The coagulation cascade (intrinsic and extrinsic pathways) converges on the common pathway producing thrombin, which converts fibrinogen to fibrin. The resulting fibrin clot serves as both a hemostatic barrier and a provisional matrix for migrating cells. Platelets degranulate, releasing platelet-derived growth factor (PDGF), transforming growth factor-beta (TGF-β), epidermal growth factor (EGF), and vascular endothelial growth factor (VEGF), which serve as chemotactic signals initiating the inflammatory phase.
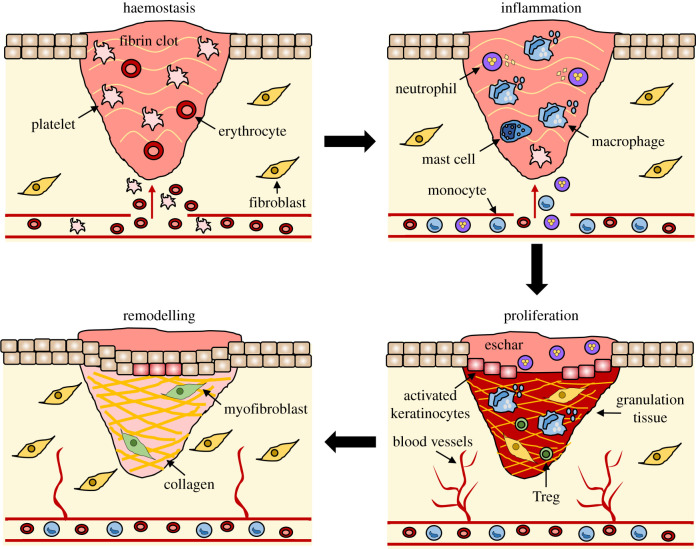
Phase 2 — Inflammation (Hours to Days 4-6)
Vasodilation follows the initial vasoconstriction, mediated by histamine, prostaglandins, and complement fragments (C3a, C5a). Increased vascular permeability allows plasma proteins and leukocytes to enter the wound. Neutrophils are the first inflammatory cells to arrive (within 6-12 hours), reaching peak numbers at 24-48 hours. They phagocytose bacteria and debris, release reactive oxygen species (ROS) and proteolytic enzymes, and undergo apoptosis after 24-48 hours. Monocytes/macrophages arrive at 48-72 hours and are the most critical cell in wound healing — they debride devitalized tissue, kill bacteria, release growth factors (PDGF, TGF-β, FGF, VEGF, IL-1, TNF-α), and transition the wound from inflammation to proliferation. Macrophage depletion experiments result in severely impaired healing.
Macrophage Phenotype Switching
Macrophages exhibit remarkable phenotypic plasticity, and the transition between phenotypes is critical for normal wound healing. M1 macrophages (classically activated; pro-inflammatory) predominate during the early inflammatory phase — they produce TNF-α, IL-1β, IL-6, reactive oxygen species, and nitric oxide, serving primarily bactericidal and debris-clearing functions. M2 macrophages (alternatively activated; anti-inflammatory/reparative) gradually replace M1 cells as the wound transitions to the proliferative phase. M2 macrophages produce TGF-β, VEGF, IL-10, and arginase, promoting angiogenesis, fibroblast recruitment, collagen synthesis, and resolution of inflammation. In chronic wounds, this M1-to-M2 transition fails — macrophages remain locked in the M1 pro-inflammatory phenotype, perpetuating tissue destruction and preventing progression to proliferation. This concept has implications for emerging therapies targeting macrophage polarization as a wound healing strategy.
Phase 3 — Proliferation (Days 4-21)
This phase is characterized by three concurrent processes: granulation tissue formation, wound contraction, and epithelialization.
Granulation tissue formation: Fibroblasts migrate into the wound along the fibrin scaffold, proliferate under the influence of PDGF and FGF, and begin synthesizing type III collagen (the initial collagen deposited in wounds), proteoglycans, and glycosaminoglycans. New capillary buds sprout from existing vessels in a process called angiogenesis, driven primarily by VEGF and FGF-2. The resulting tissue — a rich bed of new capillaries, fibroblasts, and loose extracellular matrix — is clinically visible as beefy red, moist, granular tissue.
Wound contraction: Myofibroblasts (fibroblasts that have differentiated to express α-smooth muscle actin) generate centripetal force, pulling wound edges together. Contraction reduces the wound surface area by 40-80% in open wounds, is most effective in loose-skinned areas, and is the primary mechanism of closure in secondary-intention wounds.
Epithelialization: Keratinocytes at the wound edge and from residual hair follicle remnants lose their desmosomal attachments, express integrin receptors, flatten, and migrate across the wound bed in a process called epiboly. They advance as a monolayer over viable granulation tissue (not over necrotic tissue or eschar). Migration is contact-inhibited — cells stop when they meet other advancing keratinocytes. Subsequent proliferation and differentiation restore the multilayered epidermis. In partial-thickness wounds, epithelialization occurs from both wound edges and adnexal structures (hair follicles, sweat glands), explaining faster healing.
Phase 4 — Remodeling (Day 21 to 1-2 Years)
The longest phase of wound healing involves the progressive replacement of type III collagen with type I collagen (the predominant collagen in mature skin). Collagen fibers are cross-linked and reorganized along lines of mechanical stress by matrix metalloproteinases (MMPs) balanced by tissue inhibitors of metalloproteinases (TIMPs). Net collagen content peaks at approximately 3 weeks. The collagen III:I ratio gradually shifts from 30:70 in early wounds toward the normal skin ratio of 10:90.
MMP regulation is critical during remodeling. Key enzymes include MMP-1 (interstitial collagenase, cleaves fibrillar collagen), MMP-2 and MMP-9 (gelatinases, degrade denatured collagen and basement membrane components), MMP-3 (stromelysin, broad substrate specificity including proteoglycans and laminin), and MMP-8 (neutrophil collagenase, predominant in acute wounds). In normal healing, MMPs are tightly regulated by TIMPs (TIMP-1 through TIMP-4) maintaining a balanced MMP:TIMP ratio. In chronic wounds, this ratio is pathologically elevated — excess MMP activity degrades newly deposited collagen, growth factors, and provisional matrix faster than they can accumulate, perpetuating the non-healing state.
Extracellular matrix (ECM) remodeling: Beyond collagen turnover, the ECM undergoes compositional changes during maturation. Fibronectin, which serves as a provisional scaffold in early healing, is progressively replaced by more organized collagen bundles. Proteoglycan content shifts from hyaluronic acid (predominant in early healing, promoting cell migration and hydration) to dermatan sulfate and chondroitin sulfate (characteristic of mature dermis, providing structural integrity). Elastic fiber regeneration is minimal in scar tissue, contributing to the lack of elasticity in healed wounds compared to normal skin.
1 week: ~3% of original strength (fibrin and early collagen). 3 weeks: ~20% (collagen accumulation accelerating). 6 weeks: ~40-50%. 3 months: ~80% of original tensile strength. Maximum: Scar tissue never regains more than 80% of the tensile strength of unwounded skin, regardless of how long remodeling continues. This has critical implications for surgical wound strength and recurrence risk.
Key Growth Factors in Wound Healing
| Growth Factor | Primary Source | Key Actions |
|---|---|---|
| PDGF | Platelets, macrophages, endothelial cells | Chemotaxis for neutrophils, macrophages, fibroblasts; fibroblast proliferation; collagen synthesis; angiogenesis stimulation |
| VEGF | Macrophages, keratinocytes, fibroblasts | Primary driver of angiogenesis; endothelial cell proliferation and migration; vascular permeability |
| TGF-β | Platelets, macrophages, fibroblasts | Fibroblast chemotaxis; collagen and fibronectin synthesis; MMP inhibition; immunomodulation; myofibroblast differentiation; excess leads to fibrosis/keloids |
| FGF (bFGF/FGF-2) | Macrophages, endothelial cells | Angiogenesis; fibroblast proliferation; keratinocyte migration; granulation tissue formation |
| EGF | Platelets, macrophages, saliva | Keratinocyte and fibroblast proliferation; epithelialization |
| IGF-1 | Liver, fibroblasts, macrophages | Fibroblast proliferation; collagen synthesis; cell metabolism |
| KGF (FGF-7) | Fibroblasts | Keratinocyte proliferation and migration (paracrine loop); epithelialization |
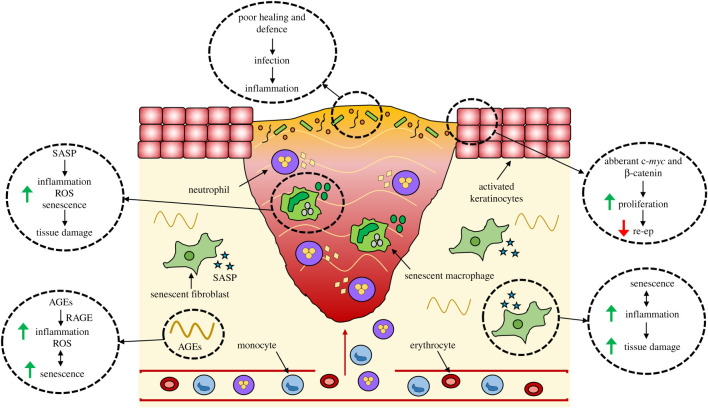
Chronic Wound Pathophysiology
In chronic wounds, the healing cascade is stalled — typically in a persistent inflammatory state. The chronic wound microenvironment is characterized by:
Elevated MMPs: MMP-2 and MMP-9 levels are 10-100x higher in chronic wound fluid compared to acute wound fluid. These proteases degrade not only necrotic tissue but also newly deposited collagen, fibronectin, growth factors (PDGF, VEGF, TGF-β), and their receptors, creating a self-perpetuating cycle of matrix destruction.
Deficient TIMPs: TIMP-1 and TIMP-2 levels are reduced, further tilting the protease-antiprotease balance toward degradation. This imbalance is a primary therapeutic target — collagen dressings act as "sacrificial substrates" to absorb excess MMP activity.
Senescent cells: Fibroblasts in chronic wounds exhibit a senescent phenotype with reduced proliferative capacity, diminished response to growth factor stimulation, and altered gene expression. These cells fail to synthesize adequate collagen and ECM components despite the presence of growth factor signals.
Biofilm: Present in 60-80% of chronic wounds, biofilms create a physical barrier to immune cell access, release enzymes that degrade host defenses, and maintain a persistent inflammatory stimulus that prevents transition to the proliferative phase. Biofilm bacteria are metabolically quiescent and protected by extracellular polymeric substance (EPS), rendering them 500-1000x more resistant to antibiotics than their planktonic counterparts.
Hypoxia: Chronic wounds are hypoxic (tissue pO2 often <20 mmHg vs 40-60 mmHg in normal tissue) due to impaired perfusion, edema, and increased metabolic demand. While transient hypoxia is a normal stimulus for angiogenesis (via HIF-1α and VEGF), persistent hypoxia impairs neutrophil oxidative killing, collagen synthesis (which requires O2 as a substrate for prolyl hydroxylase), and epithelialization.
02 Wound Assessment & Documentation
Accurate, reproducible wound assessment is the cornerstone of wound care practice. It drives treatment decisions, tracks healing trajectory, and provides medicolegal documentation. Every wound encounter requires systematic evaluation of wound dimensions, wound bed characteristics, exudate, periwound skin, and associated symptoms. The initial assessment must also include a comprehensive patient history: wound etiology and duration, prior treatments and their outcomes, relevant comorbidities (diabetes, PVD, venous disease, immunosuppression, malignancy), medications (especially steroids, anticoagulants, immunosuppressants), nutritional status, functional status, pain assessment, psychosocial factors (living situation, caregiver support, financial barriers), and patient goals of care (curative intent vs comfort-focused/palliative).
Wound-related pain is frequently underassessed and undertreated. Assess pain using a validated instrument (Numeric Rating Scale 0-10, Wong-Baker FACES for cognitive impairment, or behavioral pain scales for non-verbal patients). Document: background pain (constant baseline pain present even without manipulation), procedural pain (pain during dressing changes, debridement, or other interventions), and incident pain (pain triggered by movement, positioning, or weight-bearing). Pain character provides diagnostic information: burning pain suggests neuropathic etiology or infection, throbbing suggests vascular involvement, and pain disproportionate to wound appearance suggests atypical etiology (PG, vasculitis, calciphylaxis). Management is multimodal: topical anesthetics (lidocaine 2-4% applied 15-20 min before procedures), atraumatic dressing selection (silicone-based, non-adherent), moisture at dressing-wound interface (prevents adherence), adequate systemic analgesia for dressing changes, and treatment of underlying wound etiology.
Wound Measurement
Linear measurement: Length × width × depth in centimeters. Length is measured as the longest dimension in the head-to-toe axis; width is the longest dimension perpendicular to the length. Depth is measured by inserting a sterile cotton-tipped applicator at the deepest point and marking at the wound edge. All measurements use the "clock face" method with 12 o'clock toward the patient's head. Consistency in measurement technique across clinicians is essential for reliable longitudinal tracking — even small variations in orientation or measurement points can create artifact that mimics improvement or deterioration.
Digital planimetry: Computerized wound measurement systems (e.g., WoundZoom, eKare InSight, Silhouette) use digital photography with calibration markers or 3D imaging to calculate wound area and volume with greater accuracy and reproducibility than manual ruler measurements. These systems reduce inter-rater variability and provide photographic documentation simultaneously. While more expensive than manual measurement, they improve data quality for clinical decision-making and research.
Undermining: Tissue destruction extending under intact skin at the wound edges. Documented by clock position and distance (e.g., "undermining 2.0 cm from 2 o'clock to 5 o'clock"). Assessed by inserting a probe under the wound edge and measuring the distance from the wound edge to the extent of undermining.
Tunneling (sinus tract): A narrow channel extending from the wound in one direction. Documented by clock position and depth (e.g., "tunnel 3.5 cm at 9 o'clock"). Suggests abscess, foreign body, or fistula.
Area and volume: Surface area (L × W) is the most commonly tracked metric. Wound planimetry (tracing on transparent film or digital planimetry) provides more accurate area measurement. Volume can be estimated by instilling saline and measuring the volume required to fill the wound.
Wound Bed Assessment
| Tissue Type | Appearance | Significance |
|---|---|---|
| Granulation tissue | Beefy red, moist, granular, bleeds easily | Healthy proliferative phase; protect and maintain moist environment |
| Epithelial tissue | Pink/pearl, migrating from edges or islands | Active epithelialization; protect from trauma, avoid cytotoxic agents |
| Slough | Yellow, tan, or gray; stringy or mucinous; adherent or loosely attached | Devitalized tissue containing fibrin, dead cells, and debris; requires debridement for healing |
| Eschar | Black or brown; hard, leathery, firmly adherent | Full-thickness devitalized tissue; prevents wound assessment and healing; requires debridement (exception: stable heel eschar without signs of infection) |
| Hypergranulation | Dark red, raised above wound surface, friable | Excessive granulation tissue above the wound plane; impedes epithelialization; treat with silver nitrate, foam dressing, or brief topical steroid |
Document the percentage of each tissue type in the wound bed (e.g., "wound bed: 60% granulation, 30% slough, 10% eschar"). This quantification tracks debridement effectiveness and healing progress.
Exudate Assessment
| Type | Description | Significance |
|---|---|---|
| Serous | Clear, watery, straw-colored | Normal wound fluid; contains proteins and electrolytes |
| Sanguineous | Red, bloody | Vascular disruption; new blood vessel growth or trauma |
| Serosanguineous | Pink, watery-bloody | Most common type in healing wounds |
| Purulent | Opaque, thick, yellow/green/brown | Infection; may be malodorous |
Exudate amount is documented as none, scant, small, moderate, or large (copious). Excessive exudate in chronic wounds contains elevated levels of MMPs that degrade growth factors and extracellular matrix, impeding healing.
Periwound Skin Assessment
Evaluate the skin within 4 cm of the wound edge for: maceration (white, softened skin from excess moisture — indicates need for more absorptive dressing), erythema (redness extending >2 cm from wound edge suggests infection or cellulitis), induration (firmness indicating inflammation or infection), callus (hyperkeratosis common around DFUs — requires debridement), excoriation (denudation from adhesive or caustic drainage), edema, and temperature changes.
Validated Assessment Tools
Developed by the NPUAP. Scores three parameters: Surface area (0-10 points based on L×W), Exudate amount (0 = none, 1 = light, 2 = moderate, 3 = heavy), Tissue type (0 = closed, 1 = epithelial, 2 = granulation, 3 = slough, 4 = necrotic). Total score range 0-17. Decreasing scores over time indicate healing. Simple, quick, and valid for tracking pressure injury healing trajectory.
Comprehensive 13-item tool rating: size, depth, edges, undermining, necrotic tissue type, necrotic tissue amount, exudate type, exudate amount, skin color surrounding wound, peripheral tissue edema, peripheral tissue induration, granulation tissue, and epithelialization. Each item scored 1-5 (1 = best, 5 = worst). Total score range 13-65. Provides detailed longitudinal tracking of wound status. Requires training for inter-rater reliability.
Photography Standards
Wound photographs must include: patient identifier and date (label, not on patient), a disposable ruler or measuring guide in the field, consistent camera angle (perpendicular to wound surface), consistent distance and lighting, inclusion of an anatomic landmark for orientation, and the same background for serial photos. Photographs are part of the medical record and should be obtained at initial assessment and at least weekly or with significant changes.
Wound Healing Trajectory Monitoring
Calculating the percent area reduction (PAR) provides the most clinically meaningful measure of healing trajectory. PAR = [(initial area − current area) / initial area] × 100. A wound that does not achieve ≥40% area reduction by week 4 has a <10% probability of healing by week 12 with current therapy. This "4-week checkpoint" should trigger a comprehensive reassessment including: wound etiology confirmation (consider biopsy if atypical), vascular status, nutritional parameters, glycemic control, infection/biofilm evaluation, adherence to offloading/compression, medication review, and consideration of advanced therapies. Conversely, wounds achieving ≥50% PAR at 4 weeks have an ~80% probability of complete healing by 12 weeks, supporting continuation of the current treatment plan.
03 Terminology & Abbreviations
Wound care uses a specialized vocabulary shared across nursing, medicine, and surgical disciplines. Consistent terminology ensures clear communication, accurate documentation, and proper coding. Using standardized wound care terminology across all disciplines reduces ambiguity in treatment plans, facilitates care transitions between providers and settings (hospital to home health, wound clinic to primary care), and supports accurate data collection for quality improvement initiatives.
Essential Wound Care Terms
| Term | Definition |
|---|---|
| Acute wound | A wound proceeding through an orderly, timely healing process (typically <30 days); examples include surgical incisions and traumatic lacerations |
| Chronic wound | A wound that has failed to proceed through the normal healing phases in an orderly and timely manner; typically open >30 days or failing to show ≥40% area reduction at 4 weeks |
| Biofilm | Structured community of bacteria encased in a self-produced extracellular polymeric substance (EPS) matrix; adherent to the wound surface; resistant to antibiotics and host defenses; present in 60-80% of chronic wounds |
| Debridement | Removal of devitalized (necrotic) tissue, biofilm, and foreign material from a wound to promote healing |
| Granulation | The formation of new connective tissue and blood vessels on the wound surface during healing |
| Epithelialization | Migration of keratinocytes across the wound surface to restore the epidermal barrier |
| Contraction | Centripetal movement of wound edges mediated by myofibroblasts, reducing wound size |
| Maceration | Softening and whitening of skin due to prolonged moisture exposure; weakens tissue integrity |
| Desiccation | Drying of the wound bed; impedes cell migration and delays healing |
| Undermining | Tissue destruction beneath intact wound edges; extends in multiple directions |
| Tunneling | A narrow passageway extending from the wound in a single direction (sinus tract) |
| Periwound | The skin surrounding the wound (typically within 4 cm of the wound edge) |
| Exudate | Wound fluid containing water, electrolytes, proteins, MMPs, and growth factors |
| Induration | Abnormal firmness of tissue with palpable margins, often indicating inflammation |
| Erythema | Redness of surrounding skin; may indicate inflammation or infection |
| Sinus tract | A channel extending from an abscess or wound to the skin surface or between cavities |
| Fistula | An abnormal connection between two epithelialized surfaces (e.g., enterocutaneous fistula) |
04 Pressure Injuries
Pressure injuries (formerly pressure ulcers or decubitus ulcers) result from sustained pressure or pressure combined with shear, typically over bony prominences. They represent the most common chronic wound in acute and long-term care settings, affecting up to 3 million Americans annually with an estimated cost of $9.1-$11.6 billion per year.
NPUAP/EPUAP Pressure Injury Staging System (2016 Revision)
| Stage | Description | Key Features |
|---|---|---|
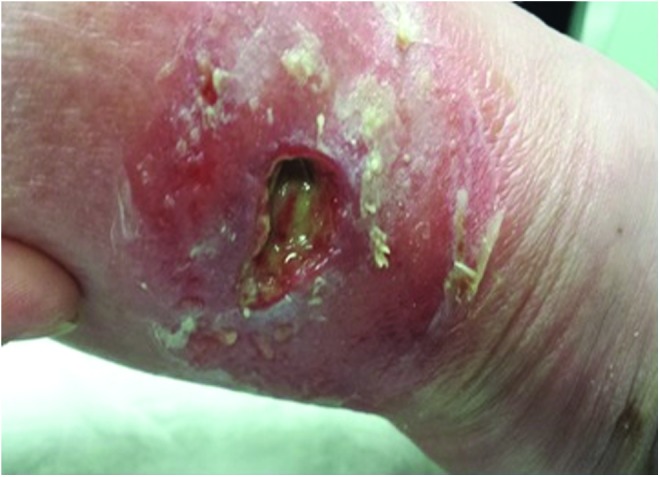
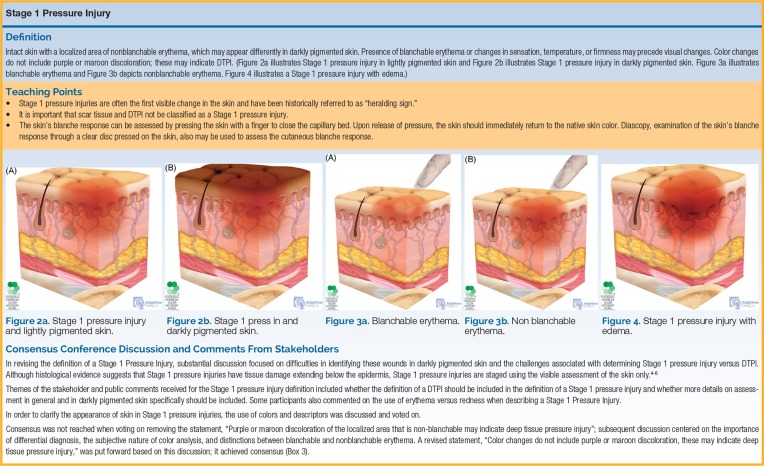
| Stage 1 | Non-blanchable erythema of intact skin | Intact skin with a localized area of non-blanchable erythema. May appear differently in darkly pigmented skin (color change, temperature difference, edema, or induration may be the only detectable sign). Does not include purple or maroon discoloration (which indicates DTPI). |
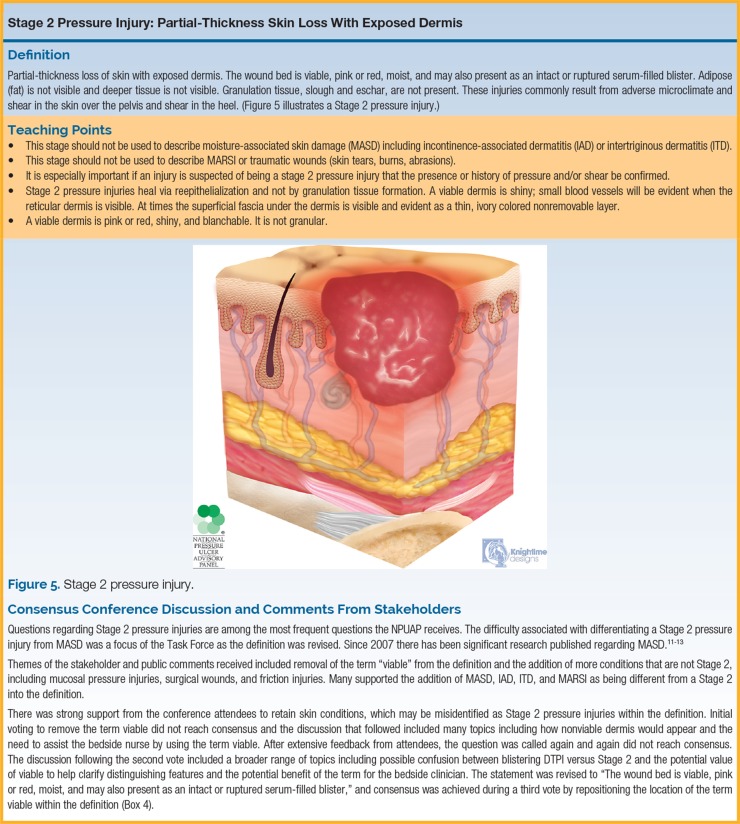
| Stage 2 | Partial-thickness skin loss with exposed dermis | The wound bed is viable, pink or red, moist, and may present as an intact or ruptured serum-filled blister. Adipose (fat) is NOT visible, and deeper tissues are not visible. Does NOT include moisture-associated skin damage (MASD), medical adhesive-related skin injury (MARSI), skin tears, tape burns, perineal dermatitis, or excoriation. |
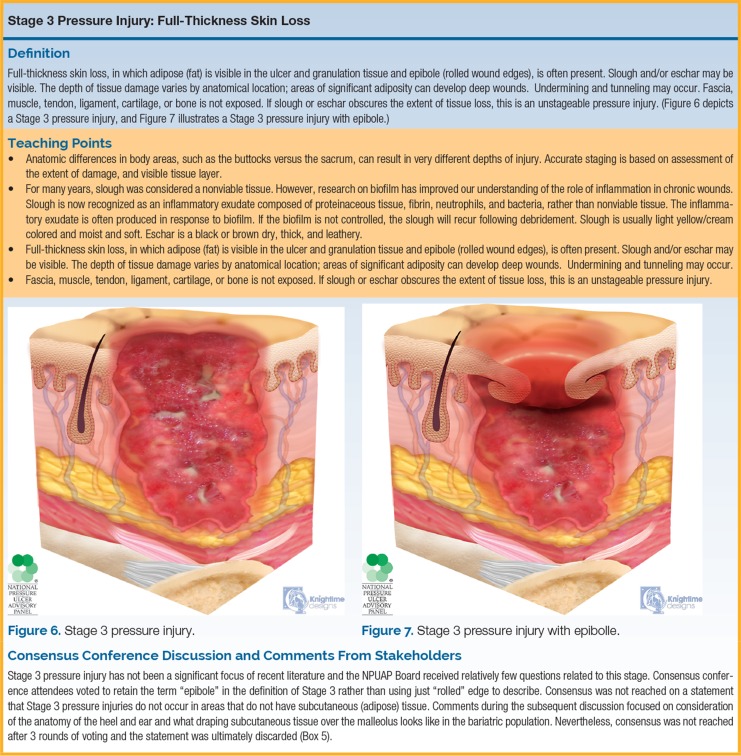
| Stage 3 | Full-thickness skin loss | Adipose (fat) is visible in the ulcer. Granulation tissue and rolled wound edges (epibole) often present. Slough and/or eschar may be visible. Depth varies by anatomic location: deep in areas with significant subcutaneous tissue (e.g., buttock) vs shallow over areas with little subcutaneous tissue (e.g., nose, ear, occiput, malleolus). Undermining and tunneling may occur. Fascia, muscle, tendon, ligament, cartilage, and bone are NOT visible or directly palpable. |
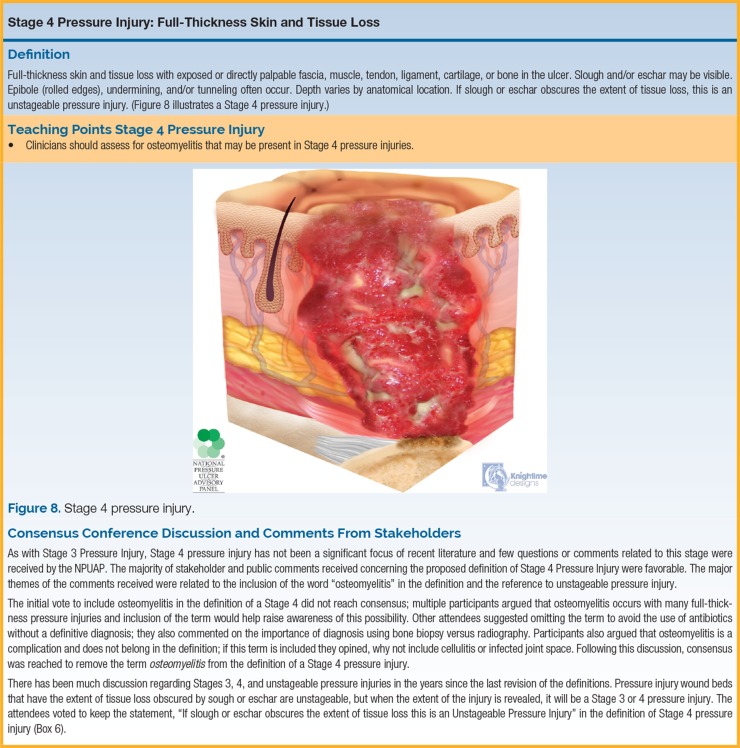
| Stage 4 | Full-thickness skin and tissue loss | Exposed or directly palpable fascia, muscle, tendon, ligament, cartilage, or bone in the ulcer. Slough and/or eschar may be visible. Rolled wound edges (epibole), undermining, and tunneling often occur. Depth varies by anatomic location. Osteomyelitis is common at this stage. |
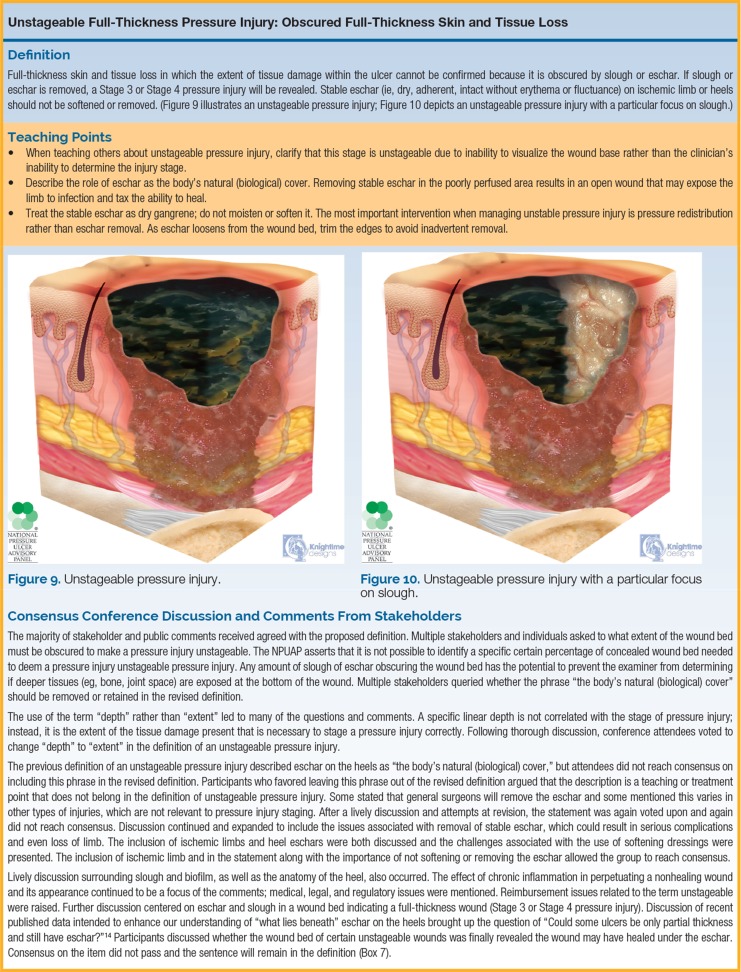
| Unstageable | Obscured full-thickness skin and tissue loss | The extent of tissue damage within the ulcer cannot be confirmed because it is obscured by slough or eschar. If slough or eschar is removed, a Stage 3 or Stage 4 pressure injury will be revealed. Stable eschar (dry, adherent, intact, without erythema or fluctuance) on the heel or ischemic limb should NOT be debrided. |
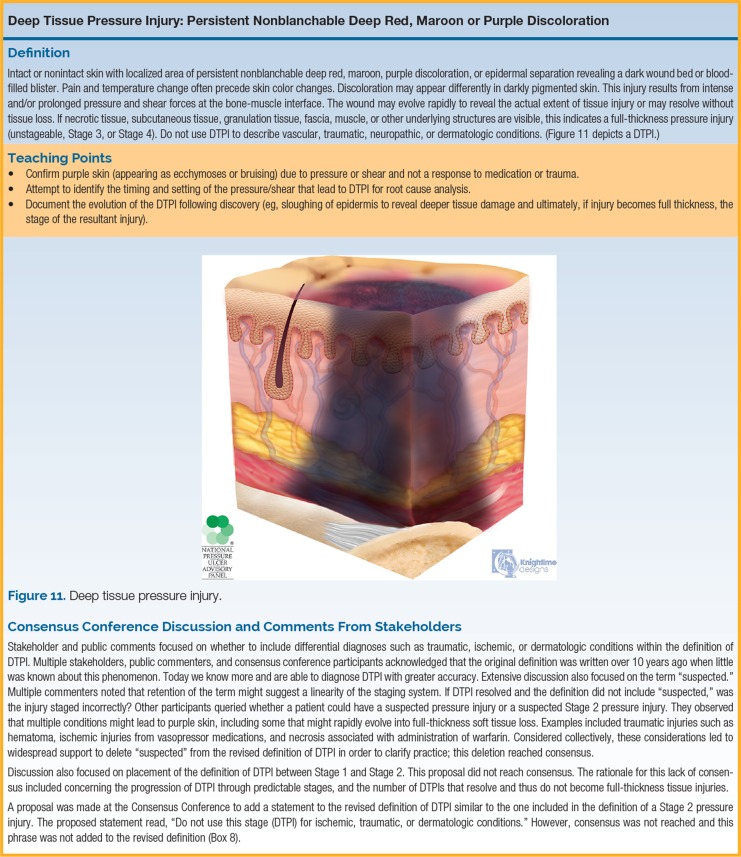
| Deep Tissue Pressure Injury (DTPI) | Persistent non-blanchable deep red, maroon, or purple discoloration | Intact or non-intact skin with localized area of persistent non-blanchable deep red, maroon, or purple discoloration or epidermal separation revealing a dark wound bed or blood-filled blister. Pain and temperature change often precede skin color changes. May evolve rapidly exposing additional layers of tissue even with optimal treatment. This injury results from intense and/or prolonged pressure and shear forces at the bone-muscle interface. |
Braden Scale for Predicting Pressure Injury Risk
The Braden Scale is the most widely validated risk assessment tool. It comprises six subscales, each scored 1-4 (except friction/shear scored 1-3). Lower scores indicate higher risk.
| Subscale | 1 (Worst) | 2 | 3 | 4 (Best) |
|---|---|---|---|---|
| Sensory Perception | Completely limited: unresponsive to painful stimuli | Very limited: responds only to painful stimuli | Slightly limited: responds to verbal commands but cannot always communicate discomfort | No impairment: responds to verbal commands, has no sensory deficit |
| Moisture | Constantly moist: skin is kept moist almost constantly | Very moist: skin is often but not always moist | Occasionally moist: skin is occasionally moist, linen change approximately once/day | Rarely moist: skin is usually dry, linen change at routine intervals |
| Activity | Bedfast: confined to bed | Chairfast: severely limited ability to walk | Walks occasionally: walks during day but very short distances | Walks frequently: walks outside room at least twice/day |
| Mobility | Completely immobile: does not make even slight changes in body position | Very limited: makes occasional slight position changes | Slightly limited: makes frequent though slight position changes independently | No limitations: makes major and frequent position changes without assistance |
| Nutrition | Very poor: never eats a complete meal; protein intake <adequate | Probably inadequate: rarely eats a complete meal; protein intake inadequate | Adequate: eats over half of most meals; protein intake adequate | Excellent: eats most of every meal; protein intake excellent |
| Friction & Shear | Problem: requires moderate-maximum assist in moving; complete lifting impossible | Potential problem: moves feebly or requires minimum assistance | No apparent problem: moves independently in bed and chair | — |
19-23: Not at risk (some authors use ≤18 as cutoff for "at risk"). 15-18: Mild risk. 13-14: Moderate risk. 10-12: High risk. ≤9: Very high risk. Total possible score range: 6-23.
Prevention Strategies
Repositioning: Reposition every 2 hours in bed; every 1 hour in chair. Use the 30-degree lateral tilt position to offload the sacrum. Avoid positioning directly on the trochanter. Elevate heels off the bed using pillows or heel-suspension devices. Use draw/lift sheets to reposition (reduces shear). Post repositioning schedule at the bedside and document each turn.
Support surfaces: Use pressure-redistribution mattresses based on risk level. Group 1 (reactive/constant low pressure): foam overlays, alternating pressure pads, gel overlays — appropriate for Stage 1-2 or at-risk patients who can be repositioned. Group 2 (powered pressure redistribution): alternating pressure mattresses, low-air-loss mattresses — for Stage 3-4, multiple turning surfaces impractical, or failure to heal on Group 1 surface. Group 3 (air-fluidized): fluidized silicone bead beds (e.g., Clinitron) — for large Stage 3-4 pressure injuries, myocutaneous flap patients, or burns >40% TBSA. Do not use ring cushions ("donut" cushions) — they concentrate pressure at the ring edges and worsen ischemia.
Moisture management: Use incontinence briefs with superabsorbent polymers; apply barrier creams (dimethicone-based) to protect perianal skin. Implement structured incontinence care protocols (assessment, scheduled toileting, perineal cleansing, barrier application). Differentiate moisture-associated skin damage (MASD) from pressure injury — MASD presents as diffuse erythema conforming to moisture exposure area, not localized over a bony prominence.
Nutrition: Ensure adequate caloric (30-35 kcal/kg/day) and protein (1.25-1.5 g/kg/day) intake; supplement zinc and vitamin C. Consult dietitian for all patients with existing pressure injuries or Braden nutrition subscale score ≤2. Provide oral nutritional supplements (ONS) between meals for patients unable to meet caloric requirements through diet alone.
Skin inspection: Perform head-to-toe skin assessment at admission and daily, with particular attention to bony prominences (sacrum, heels, ischial tuberosities, trochanters, occiput). In darkly pigmented skin, non-blanchable erythema may not be visually apparent — assess for localized warmth, edema, induration, or pain compared to adjacent tissue. Use good lighting and palpation. Document findings and interventions at each assessment.
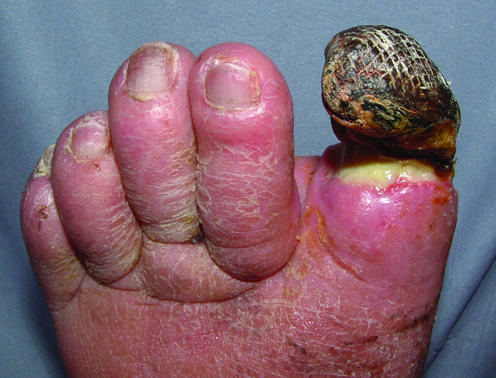
05 Diabetic Foot Ulcers
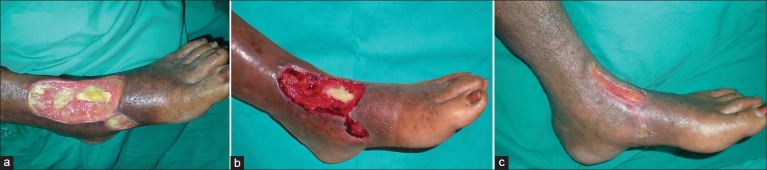
Diabetic foot ulcers (DFUs) affect approximately 15-25% of people with diabetes during their lifetime and precede 85% of diabetes-related lower-extremity amputations. The triad of peripheral neuropathy (present in 60-70% of DFU patients), peripheral arterial disease (present in 50%), and foot deformity creates the pathologic milieu for ulceration. Minor trauma or repetitive pressure on an insensate, ischemic, deformed foot leads to tissue breakdown.
Wagner Classification of Diabetic Foot Ulcers
| Grade | Description |
|---|---|
| Grade 0 | Pre-ulcerative lesion, healed ulcer, or presence of bony deformity (e.g., Charcot foot, bunion, hammer toe, callus) — at-risk foot with intact skin |
| Grade 1 | Superficial ulcer without penetration to deeper structures (partial or full-thickness through skin only) |
| Grade 2 | Deep ulcer penetrating to tendon, joint capsule, or bone without abscess or osteomyelitis |
| Grade 3 | Deep ulcer with abscess, osteomyelitis, or joint sepsis |
| Grade 4 | Localized gangrene (forefoot or heel) |
| Grade 5 | Extensive gangrene involving the entire foot, requiring major amputation |
University of Texas (UT) Classification
The UT system is a two-dimensional matrix combining wound depth (grade) with the presence of infection and/or ischemia (stage), providing superior prognostic value compared to Wagner alone.
| Grade/Stage | Stage A (No infection, no ischemia) | Stage B (Infection) | Stage C (Ischemia) | Stage D (Infection + Ischemia) |
|---|---|---|---|---|
| Grade 0: Pre- or post-ulcerative, epithelialized | 0A | 0B | 0C | 0D |
| Grade 1: Superficial wound, not involving tendon/capsule/bone | 1A | 1B | 1C | 1D |
| Grade 2: Wound penetrating to tendon or capsule | 2A | 2B | 2C | 2D |
| Grade 3: Wound penetrating to bone or joint | 3A | 3B | 3C | 3D |
IDSA/IWGDF Classification of Diabetic Foot Infections
| Severity | Clinical Findings | Management |
|---|---|---|
| Uninfected | No purulence, no signs of inflammation | Wound care only; no antibiotics |
| Mild | ≥2 signs of inflammation (purulence, erythema, pain, warmth, induration); erythema ≤2 cm from wound edge; infection limited to skin/superficial subcutaneous tissue | Outpatient oral antibiotics (target gram-positive cocci); narrow spectrum; 1-2 week course |
| Moderate | Infection in a metabolically stable patient with ≥1 of: cellulitis >2 cm, lymphangitis, spread beneath fascia, deep tissue abscess, gangrene, involvement of muscle/tendon/joint/bone | May require hospitalization; parenteral antibiotics initially; broad-spectrum (gram-positive, gram-negative, anaerobes); imaging for deep structures; surgical consultation |
| Severe | Infection in a patient with systemic toxicity or metabolic instability (fever, chills, tachycardia, hypotension, confusion, vomiting, leukocytosis, acidosis, severe hyperglycemia, azotemia) | Hospitalization required; IV broad-spectrum antibiotics (MRSA coverage + gram-negative + anaerobic); emergent surgical evaluation for drainage/debridement; consider ICU admission |
Neuropathy Screening
Semmes-Weinstein 10-g monofilament: The standard screening test. The monofilament is applied perpendicular to the skin at 10 sites on each foot (plantar aspects of hallux, 1st, 3rd, and 5th metatarsal heads, plantar midfoot, heel, and dorsal first web space). Loss of sensation at ≥4 sites indicates clinically significant neuropathy. Sensitivity 66-91%, specificity 34-86%. 128-Hz tuning fork: Tests vibratory perception at the hallux dorsal interphalangeal joint. Loss of vibratory sense correlates with large-fiber neuropathy. Vibration perception threshold (VPT): Biothesiometry provides quantitative measurement; VPT >25 volts indicates high ulceration risk (7-fold increased risk). Ankle reflexes: Absent Achilles reflex suggests peripheral neuropathy. Michigan Neuropathy Screening Instrument (MNSI): Validated questionnaire + physical exam scoring system.
Vascular Assessment
| Test | Normal | Interpretation |
|---|---|---|
| Ankle-Brachial Index (ABI) | 1.0-1.4 | >1.4: non-compressible (calcified vessels — common in diabetes/CKD; unreliable). 1.0-1.4: normal. 0.9-1.0: borderline. 0.5-0.9: moderate PAD (claudication). <0.5: severe PAD (rest pain, tissue loss). <0.4: critical limb ischemia. |
| Toe pressures | >60 mmHg | >60 mmHg: adequate for healing. 40-60 mmHg: borderline. <40 mmHg: impaired healing. <30 mmHg: critical ischemia. Toe vessels are less prone to calcification; more reliable in diabetes. |
| Toe-Brachial Index (TBI) | >0.7 | >0.7: normal. <0.64: significant PAD. Preferred over ABI in patients with medial arterial calcification. |
| TcPO2 (transcutaneous oxygen) | >40 mmHg | >40 mmHg: adequate for healing. 30-40 mmHg: may heal with optimal care. 20-30 mmHg: compromised healing. <20 mmHg: unlikely to heal without revascularization. Used for HBOT patient selection: periwound increase >10 mmHg with O2 challenge predicts benefit. |
Charcot Neuroarthropathy (Charcot Foot)
A progressive, destructive process affecting the bones, joints, and soft tissues of the foot and ankle in patients with peripheral neuropathy. The neurotraumatic and neurovascular theories explain the pathogenesis: repetitive unperceived trauma on an insensate foot with autonomic neuropathy-driven hyperemia leads to osteoclast activation (RANKL upregulation), microfractures, joint dislocation, and architectural collapse. The most commonly affected site is the tarsometatarsal (Lisfranc) joint complex (Sanders-Frykberg Type II, accounting for ~60% of cases), followed by the midfoot and hindfoot.
Stage 0 (Prodromal): Clinical suspicion with warm, swollen, erythematous foot; normal radiographs; MRI may show bone marrow edema. Stage 1 (Development/Fragmentation): Acute inflammation; radiographs show periarticular fragmentation, joint subluxation, debris; clinically red, hot, swollen foot with temperature ≥2°C warmer than contralateral side. Stage 2 (Coalescence): Decreased swelling and warmth; radiographic evidence of new bone formation, absorption of debris, coalescence of fragments. Stage 3 (Reconstruction/Consolidation): Resolution of inflammation; remodeling and consolidation of fractures; residual deformity (rocker-bottom foot) may create pressure points for ulceration.
Offloading
Total contact cast (TCC): The gold standard for offloading neuropathic plantar DFUs. Achieves healing rates of ~90% at 12 weeks. Distributes pressure across the entire plantar surface, reduces shear, and ensures adherence (non-removable). Contraindicated in active infection, significant PAD, and extreme edema fluctuation. Irremovable walkers: Removable cast walkers (e.g., CAM boot) rendered irremovable with a cohesive bandage wrap have equivalent efficacy to TCC and may be preferred for ease of application. Other options: Therapeutic footwear (depth shoes with custom insoles), half shoes, healing sandals, felted foam, and accommodative padding for lower-risk ulcers.
All patients with diabetes should receive a comprehensive foot examination at least annually, including: visual inspection (deformity, callus, nail pathology, skin integrity, interdigital maceration), monofilament testing (10-g at minimum 4 plantar sites per foot), vibratory sensation assessment, pedal pulse palpation, and risk stratification. Risk Category 0: No neuropathy, no PAD, no deformity → annual exam, patient education. Risk Category 1: Neuropathy present, no deformity or PAD → exam every 6 months, therapeutic footwear. Risk Category 2: Neuropathy + deformity or PAD → exam every 3 months, therapeutic footwear with custom insoles, podiatry referral. Risk Category 3: History of ulcer or amputation → exam every 1-3 months, therapeutic footwear, podiatric care, vascular follow-up. Patient education must include daily self-inspection, proper footwear, avoidance of walking barefoot, checking bath water temperature, and when to seek medical attention.
06 Vascular Ulcers
Vascular ulcers account for approximately 70-80% of chronic lower-extremity wounds. They are divided into venous (60-80% of leg ulcers), arterial (15-25%), and mixed (15-30%) etiologies. Accurate differentiation is critical because treatment strategies differ fundamentally — compression for venous ulcers is contraindicated in severe arterial disease.
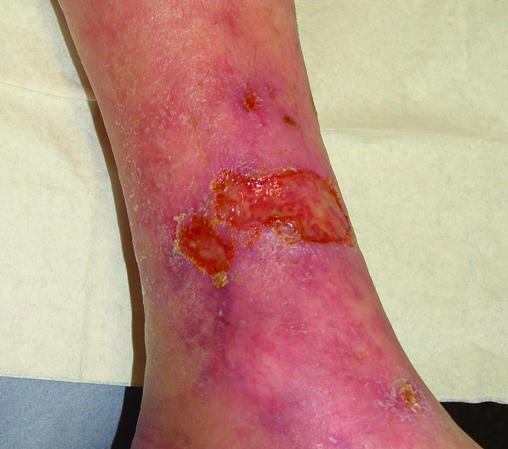
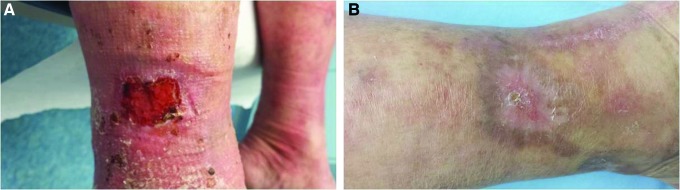
Venous Leg Ulcers
Venous ulcers result from sustained venous hypertension due to valvular incompetence in the superficial, deep, or perforator veins. Ambulatory venous pressure remains elevated (normally drops from ~100 mmHg at rest to ~20 mmHg with ambulation; in venous disease it remains >60 mmHg), causing capillary distension, fibrinogen leakage, pericapillary fibrin cuff formation, white blood cell trapping, and chronic inflammation.
Clinically they present as shallow, irregularly shaped ulcers in the gaiter area (medial lower leg between ankle and mid-calf), with surrounding hemosiderin staining (brown pigmentation from red blood cell extravasation and hemoglobin degradation), lipodermatosclerosis (woody induration of subcutaneous tissue from chronic fibrosis), varicose veins, stasis dermatitis (eczematous changes with pruritus), and edema. The wound bed is typically granular with heavy serous or serosanguineous exudate. Pain is variable — often described as a dull ache that improves with leg elevation (in contrast to arterial pain which worsens with elevation).
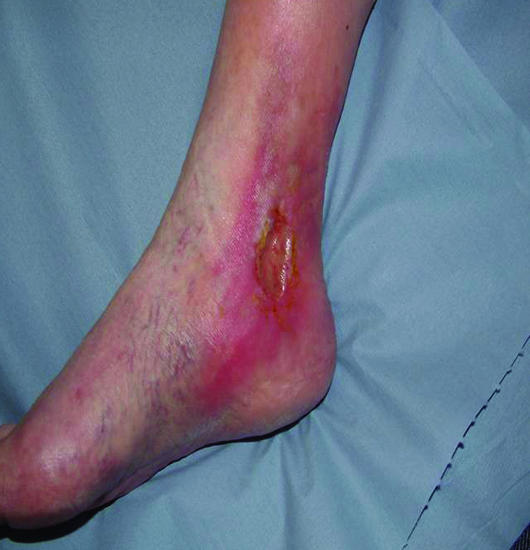
| Feature | Venous Ulcer | Arterial Ulcer |
|---|---|---|
| Location | Gaiter area (medial > lateral malleolus) | Toes, dorsal foot, lateral malleolus, pressure points |
| Shape | Irregular, shallow | Punched-out, well-demarcated, deep |
| Wound bed | Red granulation, fibrinous base, heavy exudate | Pale, necrotic, minimal granulation, dry |
| Pain | Aching, improves with elevation | Severe, worsens with elevation, rest pain (nocturnal) |
| Surrounding skin | Hemosiderin, lipodermatosclerosis, eczema, edema | Thin, shiny, hairless, cool, pale; dependent rubor |
| Pulses | Present (palpable pedal pulses) | Absent or diminished |
| ABI | ≥0.8 | <0.5 |
| Treatment priority | Compression therapy | Revascularization |
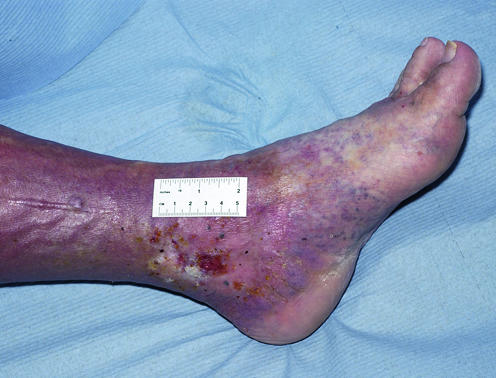
CEAP Classification of Chronic Venous Disease
| Class | Description |
|---|---|
| C0 | No visible or palpable signs of venous disease |
| C1 | Telangiectasias (<1 mm) or reticular veins (1-3 mm) |
| C2 | Varicose veins (>3 mm) |
| C3 | Edema without skin changes |
| C4a | Pigmentation (hemosiderin) or eczema (stasis dermatitis) |
| C4b | Lipodermatosclerosis or atrophie blanche |
| C5 | Healed venous ulcer |
| C6 | Active venous ulcer |
The full CEAP classification also includes Etiology (Ec = congenital, Ep = primary, Es = secondary, En = no cause identified), Anatomy (As = superficial, Ap = perforator, Ad = deep), and Pathophysiology (Pr = reflux, Po = obstruction, Pr,o = both). Clinical class is further qualified as symptomatic (s) or asymptomatic (a) — for example, C2s indicates symptomatic varicose veins.
Compression Therapy for Venous Ulcers
Compression is the cornerstone of venous ulcer management. Target compression is 30-40 mmHg at the ankle, graduated (highest at ankle, decreasing proximally). Options include:
Multilayer compression bandaging: Four-layer bandage system (e.g., Profore) providing sustained compression for up to 7 days. Considered the gold standard for active venous ulcers. Unna boot: Zinc oxide-impregnated gauze bandage providing semi-rigid compression. Changed every 1-2 weeks. Well-suited for ambulatory patients. Compression stockings: Used primarily for maintenance after ulcer healing. 30-40 mmHg knee-high stockings reduce recurrence by approximately 50%. Adjustable compression wraps: Velcro-type wraps (e.g., CircAid) allowing patient self-adjustment. Intermittent pneumatic compression (IPC): Mechanical pump devices for patients unable to tolerate sustained compression or with refractory ulcers.
Pentoxifylline 400 mg TID is the only medication with Level 1 evidence for improving venous ulcer healing when combined with compression (NNT ~6 for complete healing). Its mechanism includes reducing blood viscosity, improving red blood cell deformability, decreasing platelet aggregation, and anti-inflammatory effects (reduces TNF-α and leukotriene production). It may be used even in patients who cannot tolerate compression. Common side effects include gastrointestinal upset (nausea, dyspepsia). Aspirin 300 mg daily has shown modest evidence of accelerating venous ulcer healing in some studies, though results are inconsistent. Micronized purified flavonoid fraction (MPFF/Daflon) 500 mg BID has evidence in European guidelines for venous ulcer healing as adjunctive therapy, though it is not FDA-approved for this indication in the US.
Venous Ulcer Recurrence Prevention
Venous ulcer recurrence rates are 30-70% within 12 months without ongoing compression. Lifelong compression therapy is the cornerstone of recurrence prevention. Patients who have healed a venous ulcer should wear 30-40 mmHg graduated compression stockings daily for life. Compliance is the major challenge — stockings should be replaced every 3-6 months as elasticity degrades. Stocking donning aids (e.g., Medi Butler, Doff N' Donner) improve independence and compliance for patients with limited hand strength or mobility. Additional recurrence prevention measures include: regular exercise (calf muscle pump activation), leg elevation when sedentary, weight management, skin care (emollients to prevent dryness and cracking), treatment of underlying venous reflux (endovenous ablation, sclerotherapy), and prompt wound care at the first sign of skin breakdown.
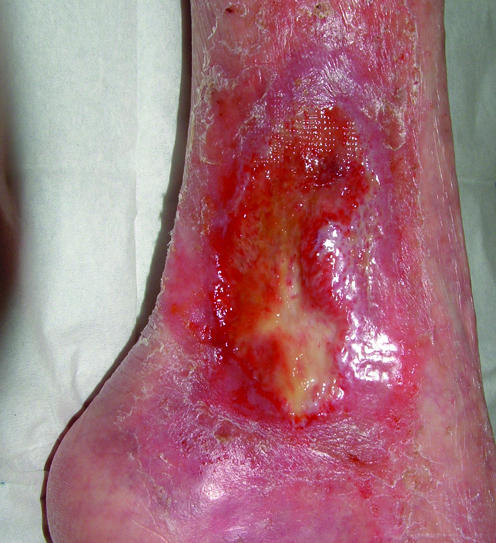
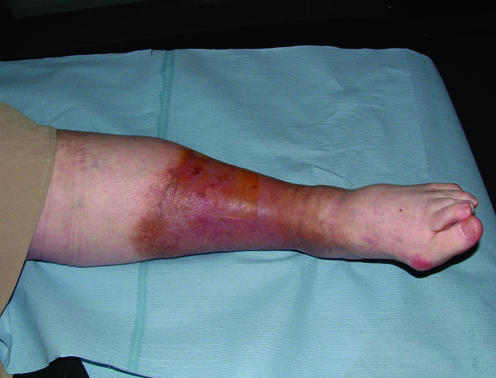
Arterial Ulcers
Arterial ulcers result from inadequate arterial perfusion (peripheral arterial disease). They present as well-demarcated, "punched-out" ulcers with pale or necrotic wound beds and minimal granulation tissue, located on the distal toes, dorsum of the foot, over the lateral malleolus, or at sites of trauma. Associated findings include absent pedal pulses, cool/pale extremity, dependent rubor (red-purple color with dependency that blanches on elevation), trophic changes (thin shiny skin, hair loss, thick dystrophic nails), and pain at rest (especially nocturnal, relieved by dependency).
Key diagnostic criteria: ABI <0.5 (severe PAD), rest pain, tissue loss. Treatment priority is revascularization first (endovascular angioplasty/stenting or surgical bypass), followed by wound care after adequate perfusion is established. Wound care without revascularization is futile in critical limb ischemia. Post-revascularization wound care follows standard moist wound healing principles. Post-procedure monitoring includes repeat ABI and clinical assessment to confirm improved perfusion before initiating definitive wound therapy.
Stage 0: Asymptomatic. Stage 1: Mild claudication. Stage 2: Moderate claudication. Stage 3: Severe claudication. Stage 4: Ischemic rest pain (critical limb ischemia begins). Stage 5: Minor tissue loss (non-healing ulcer, focal gangrene with diffuse pedal ischemia). Stage 6: Major tissue loss (extending above transmetatarsal level, functional foot no longer salvageable). Stages 4-6 constitute chronic limb-threatening ischemia (CLTI) — the current preferred terminology replacing "critical limb ischemia" — and require urgent vascular evaluation for revascularization to prevent limb loss.
Venous Ablation and Surgical Interventions
Correction of underlying venous reflux accelerates ulcer healing and reduces recurrence. Endovenous thermal ablation (radiofrequency or laser ablation of the great or small saphenous veins) and ultrasound-guided foam sclerotherapy are first-line interventions for superficial venous reflux. The EVRA trial demonstrated that early endovenous ablation (within 2 weeks of presentation) combined with compression resulted in faster ulcer healing compared to compression alone (median healing time 56 vs 82 days). Subfascial endoscopic perforator surgery (SEPS) addresses incompetent perforator veins. Deep venous reconstruction (valve repair, stenting for iliac vein obstruction) is reserved for refractory cases with documented deep venous pathology. All patients with venous ulcers should undergo duplex ultrasound assessment of the superficial and deep venous systems to identify correctable reflux.
Mixed Arterial-Venous Ulcers
Patients with concurrent venous insufficiency and moderate PAD (ABI 0.5-0.8). Management requires modified compression (reduced to 23-30 mmHg or short-stretch bandaging that provides high working pressure but low resting pressure) with vascular surgery consultation. Inelastic compression systems are preferred over elastic systems in mixed disease because they provide compression primarily during ambulation (muscle pump) with low resting pressure.
07 Moist Wound Healing & Dressing Categories
In 1962, George Winter demonstrated that wounds covered with an occlusive dressing epithelialized twice as fast as those left to dry in the air, establishing the principle of moist wound healing. A moist wound environment promotes cell migration (keratinocytes migrate faster over moist surfaces), growth factor activity, autolytic debridement, angiogenesis, and reduces pain. The goal is balanced moisture — enough to prevent desiccation but not so much as to cause maceration.
TIME Framework for Wound Bed Preparation
The TIME framework provides a systematic approach to removing barriers to healing:
| Component | Clinical Observation | Proposed Intervention |
|---|---|---|
| T — Tissue (non-viable) | Necrotic tissue (slough, eschar) present in wound bed | Debridement (sharp, autolytic, enzymatic, mechanical, or biological) to remove devitalized tissue and biofilm |
| I — Infection/Inflammation | Increased bacteria/biofilm; prolonged inflammation; elevated MMPs | Remove biofilm (debridement), topical antimicrobials for local infection, systemic antibiotics for spreading/systemic infection; anti-inflammatory strategies |
| M — Moisture imbalance | Wound too dry (desiccation) or too wet (maceration) | Select dressings to add moisture (hydrogels for dry wounds) or absorb excess (foams, alginates, hydrofibers for exudative wounds); protect periwound skin |
| E — Edge (non-advancing) | Non-migrating wound edges; rolled edges (epibole); hyperkeratosis | Reassess causative factors; debride wound edges; consider advanced therapies (skin substitutes, growth factors, NPWT) if stalled >4 weeks |
Comprehensive Dressing Category Guide
| Dressing Type | Composition | Absorption | Indications | Contraindications | Change Frequency |
|---|---|---|---|---|---|
| Transparent film | Thin polyurethane membrane with adhesive; semi-permeable (O2/moisture vapor passes, bacteria/water cannot) | None (moisture-retentive) | Superficial wounds, Stage 1 pressure injuries, skin tears (secondary dressing), IV site protection, autolytic debridement of thin slough | Infected wounds, moderate-heavy exudate, fragile periwound skin | Every 5-7 days or when seal is broken |
| Hydrocolloid | Inner layer of gelatin, pectin, carboxymethylcellulose (CMC) in adhesive matrix; outer polyurethane film | Low to moderate | Partial-thickness wounds, Stage 2 pressure injuries, minor burns, autolytic debridement, granulating wounds with low exudate | Heavily exudative wounds, infected wounds (traps bacteria), fragile periwound skin, arterial ulcers | Every 3-7 days (change when dressing swells to <1 cm from edge) |
| Foam | Polyurethane foam; available in adhesive/non-adhesive, with/without silicone border, various thicknesses | Moderate to high | Moderate-heavy exudate wounds, Stage 2-4 pressure injuries, venous ulcers, under compression, surgical wounds, peri-wound protection | Dry wounds, third-degree burns, wounds requiring packing | Every 3-7 days (change when saturated to within 1 cm of edge) |
| Alginate | Derived from brown seaweed (calcium/sodium salts of alginic acid); forms hydrophilic gel on contact with wound fluid | High (15-20x weight) | Moderate-heavy exudate, bleeding wounds (hemostatic), cavity/tunneling wounds (rope form), surgical wounds | Dry wounds (will desiccate wound bed), third-degree burns, implant exposure | Every 1-3 days depending on exudate |
| Hydrofiber | Sodium carboxymethylcellulose (CMC) fibers; forms cohesive gel when wet (vertical absorption with lateral wicking minimized) | High (25-30x weight) | Moderate-heavy exudate, cavity wounds, under NPWT, under compression, surgical wounds | Dry wounds, arterial ulcers with insufficient perfusion | Every 1-7 days; can remain in place up to 7 days under secondary dressing |
| Hydrogel | Water-based (70-90% water) polymer gels; available as amorphous gel, sheet, or impregnated gauze | None (donates moisture) | Dry wounds, slough-covered wounds (autolytic debridement), partial-thickness burns, radiation dermatitis, painful wounds (cooling effect) | Heavily exudative wounds, infected wounds (some formulations), macerated periwound | Every 1-3 days; amorphous gel reapplied at each dressing change |
| Collagen | Bovine, porcine, equine, or avian collagen; sheets, pads, powder, or gel | Moderate | Stalled wounds, chronic wounds with elevated MMP activity (collagen acts as MMP "sacrificial substrate"), partial and full-thickness wounds | Known collagen allergy, third-degree burns, heavy exudate | Daily to every 7 days depending on product |
| Silver dressings | Various substrates (foam, alginate, hydrofiber, CMC) impregnated with ionic or nanocrystalline silver | Depends on substrate | Infected or critically colonized wounds, wounds at high risk for infection, biofilm management | Silver allergy; wounds being treated with enzymatic debriders (silver inactivates collagenase); MRI-incompatible nanocrystalline silver | Every 1-7 days depending on substrate |
| Honey dressings | Medical-grade Manuka/Leptospermum honey (gamma-irradiated for sterility); sheets, gel, or paste | Low to moderate; osmotic action draws fluid | Infected and critically colonized wounds, sloughy wounds (promotes autolysis), odorous wounds, partial-thickness burns | Known honey allergy; pain intolerance (osmotic drawing may cause initial stinging); some formulations high in sugar — caution in diabetes (no systemic absorption) | Every 1-3 days |
| PHMB dressings | Polyhexamethylene biguanide (antiseptic) impregnated into gauze or foam | Depends on substrate | Infected or critically colonized wounds, biofilm disruption, wound irrigation/cleansing | Known PHMB sensitivity, concurrent use with anionic surfactants | Every 1-3 days |
Periwound Skin Protection
Protection of the periwound skin is as important as managing the wound bed itself. Periwound damage (maceration, excoriation, contact dermatitis) expands the wound margin and impairs healing. Strategies include:
Moisture barrier products: Dimethicone-based skin protectants (e.g., Cavilon, Critic-Aid) form a breathable, transparent barrier against wound exudate and incontinence. Apply to periwound skin at each dressing change. Petrolatum-based barriers (zinc oxide paste, A+D ointment) provide thicker protection but may interfere with adhesive dressing adherence. Cyanoacrylate-based skin protectants (e.g., Marathon liquid skin protectant) form a durable film lasting 48-72 hours that protects against adhesive stripping and moisture damage.
Window framing: Apply a hydrocolloid "frame" around the wound before placing the primary dressing. The hydrocolloid protects the periwound skin from adhesive trauma and exudate exposure while providing a secure adhesive surface for the secondary dressing.
Negative pressure considerations: When applying NPWT, ensure drape does not contact macerated skin directly. Apply skin prep or hydrocolloid under the drape margins to protect fragile periwound tissue.
Wound Cleansing
Proper wound cleansing removes surface contaminants, loose debris, and residual dressing material without damaging viable tissue. Normal saline (0.9% NaCl) is the traditional standard cleansing solution — isotonic, non-cytotoxic, and widely available. Tap water (potable) is equivalent to saline for wound cleansing in most settings (Cochrane review found no difference in infection rates). Antiseptic cleansers: Solutions containing PHMB with betaine surfactant (Prontosan) or hypochlorous acid (Vashe, Microcyn) provide antimicrobial wound cleansing with minimal cytotoxicity. These are preferred over traditional antiseptics (povidone-iodine, hydrogen peroxide, Dakin solution at full strength) which are cytotoxic to fibroblasts and keratinocytes at concentrations that kill bacteria.
Irrigation pressure: Optimal wound irrigation pressure is 4-15 psi (pounds per square inch). Pressures below 4 psi are insufficient to dislodge bacteria and debris. Pressures above 15 psi can drive bacteria into tissue and damage the wound bed. A 35 mL syringe with an 18-gauge angiocatheter delivers approximately 8 psi, which is the most commonly recommended bedside method. Commercially available irrigation systems (pulsed lavage devices) allow adjustable pressure settings for larger or deeper wounds.
08 Antimicrobial Dressings
Antimicrobial dressings play a critical role in managing the wound infection continuum, particularly at the level of critical colonization and local infection where systemic antibiotics are not indicated but bacterial burden impedes healing.
Wound Infection Continuum
| Level | Description | Management |
|---|---|---|
| Contamination | Presence of non-replicating organisms on wound surface (all open wounds are contaminated) | Standard wound care; no antimicrobial intervention needed |
| Colonization | Replicating organisms present without host immune response; no clinical signs of infection; healing not impaired | Standard wound care; routine cleansing |
| Critical colonization (local infection/biofilm) | Bacterial burden sufficient to impair healing but without classic infection signs; wound stalls; increased exudate, friable granulation, new slough formation | Topical antimicrobial dressings (silver, honey, PHMB, cadexomer iodine); biofilm disruption (debridement + antimicrobial); NO systemic antibiotics |
| Local infection | Classic signs: purulent drainage, erythema, warmth, pain, swelling, malodor, friable/discolored granulation tissue | Topical antimicrobials + consider systemic antibiotics if cellulitis; debridement; increased frequency of dressing changes |
| Spreading/systemic infection | Cellulitis, lymphangitis, fever, sepsis, bacteremia | Systemic antibiotics (empiric then culture-directed); debridement; possible hospitalization |
Silver-Based Dressings
Ionic silver (Ag+): Released from silver-containing compounds (silver sulfadiazine, silver nitrate, silver chloride). Disrupts bacterial cell membranes and enzyme systems. Broad-spectrum activity against gram-positive and gram-negative bacteria, fungi, and some viruses. Available in multiple substrates (foam, alginate, hydrofiber, CMC). Examples: Aquacel Ag, Mepilex Ag. Nanocrystalline silver: Provides sustained release of silver ions from ultra-small silver crystals. Higher antimicrobial potency at lower silver concentrations. Acticoat (nanocrystalline silver on rayon/polyethylene mesh) is the prototype. Must be moistened with sterile water (NOT saline, which precipitates silver chloride and inactivates it). Can cause transient gray-blue skin discoloration (argyria-like) that resolves when discontinued.
Other Antimicrobial Agents
Cadexomer iodine: Starch microspheres containing 0.9% iodine. As the microspheres absorb exudate, they swell and release iodine slowly into the wound. Provides sustained antimicrobial activity, absorbs exudate, and facilitates debridement. Effective against biofilm. Contraindicated in thyroid disorders (Hashimoto, Graves), iodine allergy, pregnancy/lactation, lithium use, and large wounds (>150 cm2). Example: Iodosorb.
PHMB (polyhexamethylene biguanide): A synthetic antiseptic with broad-spectrum activity against bacteria, fungi, and viruses. Low cytotoxicity relative to other antiseptics. Available as wound irrigation solution (e.g., Prontosan) and impregnated dressings. Effective for biofilm disruption when combined with a surfactant (betaine).
Medical-grade honey (Manuka/Leptospermum): Multiple mechanisms of action: high osmolarity draws fluid and creates hostile environment for bacteria; low pH (3.5-4.5) inhibits bacterial growth; continuous low-level hydrogen peroxide production from glucose oxidase; methylglyoxal (MGO) in Manuka honey provides non-peroxide antibacterial activity rated by Unique Manuka Factor (UMF). Effective against MRSA and biofilm. Must be medical-grade (gamma-irradiated, standardized). Do not use food-grade honey on wounds.
All topical antimicrobial dressings should be reassessed after 2 weeks of use. If the wound has improved (decreased bioburden, improved granulation, reduced exudate), the antimicrobial dressing may be continued or transitioned to a non-antimicrobial dressing. If no improvement is seen, reassess the treatment plan: consider different antimicrobial, re-debride biofilm, obtain wound culture, evaluate for systemic factors, or escalate to systemic antibiotics. Prolonged use without reassessment promotes resistance and adds unnecessary cost.
Biofilm Management
Biofilms are present in 60-80% of chronic wounds and are a primary cause of healing failure. Biofilm bacteria are 500-1000x more resistant to antibiotics than planktonic (free-floating) bacteria. Biofilm management requires a combined approach: (1) sharp/mechanical debridement to physically disrupt and remove the biofilm, followed immediately by (2) topical antimicrobial dressing application (within the "therapeutic window" before biofilm reconstitutes in 24-72 hours). This "wound hygiene" approach should be repeated at each dressing change for chronic wounds with suspected biofilm. Biofilm reformation occurs within 24 hours of debridement, reaching mature biofilm status again within 72 hours if not suppressed.
09 Advanced Wound Products
Advanced wound therapies are indicated for chronic wounds that have failed to demonstrate adequate healing progress (≥40-50% area reduction) after 4 weeks of standard care including debridement, moisture balance, offloading/compression, infection management, and nutritional optimization.
Skin Substitutes & Tissue-Engineered Products
| Product | Type | Composition | Indications |
|---|---|---|---|
| Apligraf | Living bilayered cellular construct | Bovine type I collagen matrix with neonatal fibroblasts (dermal layer) and neonatal keratinocytes (epidermal layer) | Venous leg ulcers >1 month duration; neuropathic DFUs >3 weeks duration. FDA-approved for both indications. |
| Dermagraft | Living dermal substitute | Bioabsorbable polyglactin mesh seeded with neonatal fibroblasts that secrete growth factors, collagen, and ECM proteins | Full-thickness DFUs >6 weeks duration; provides growth factor delivery rather than permanent structural scaffold |
| Integra | Acellular bilayer matrix | Cross-linked bovine collagen and chondroitin-6-sulfate (dermal layer) with silicone membrane (temporary epidermal layer); silicone layer removed after neodermis forms (2-3 weeks) and thin STSG applied | Deep/full-thickness wounds, burn reconstruction, complex soft tissue defects requiring dermal template |
| Oasis (SIS) | Acellular ECM | Porcine small intestinal submucosa (SIS); naturally occurring ECM containing collagen (types I, III, IV), fibronectin, glycosaminoglycans, and growth factors | Partial and full-thickness wounds, chronic wounds, DFUs, venous ulcers |
| EpiFix/AmnioFix | Dehydrated human amnion/chorion membrane (dHACM) | Processed human placental tissue containing collagen, growth factors (PDGF, VEGF, FGF, TGF-β), and anti-inflammatory cytokines (IL-1Ra, TIMP) | DFUs, venous ulcers, chronic wounds; applied to clean granulating wound bed |
| Grafix | Cryopreserved human placental membrane | Viable placental membrane containing living cells (MSCs, fibroblasts, epithelial cells), growth factors, and intact ECM | DFUs refractory to standard care |
Growth Factors
Becaplermin (Regranex): Recombinant human PDGF-BB (rhPDGF-BB) 0.01% gel. The only FDA-approved growth factor for wound healing. Applied as a thin layer to a clean, debrided DFU daily, covered with saline-moistened gauze. Indicated for neuropathic DFUs extending into subcutaneous tissue or deeper. Demonstrated a 43% complete healing rate vs 35% with placebo gel at 20 weeks. Carries a black box warning for increased mortality from malignancy with ≥3 tubes used (post-marketing data; causality debated). Contraindicated in known neoplasm at application site.
Platelet-Rich Plasma (PRP)
Autologous platelet-rich plasma is prepared by centrifuging the patient's own blood to concentrate platelets (typically 3-5x baseline concentration) and their associated growth factors (PDGF, TGF-β, VEGF, EGF, IGF-1). The concentrated platelet preparation is applied directly to the wound bed or injected into the wound edges. PRP provides a supraphysiologic concentration of autologous growth factors at the wound site. Evidence supports use in chronic DFUs and venous ulcers refractory to standard care. Advantages include autologous origin (no rejection risk), relatively low cost, and point-of-care preparation. Limitations include lack of standardized preparation protocols (significant variability between devices and techniques), variable platelet concentrations, and limited high-quality RCT data.
Extracellular Matrix (ECM) Scaffolds
ECM-based products provide a three-dimensional scaffold that supports cellular ingrowth and modulates the wound microenvironment. Unlike simple collagen dressings, intact ECM scaffolds retain the complex structural and signaling molecules of the native tissue, including basement membrane components (laminin, type IV collagen), growth factors bound to the matrix, and glycosaminoglycans. These products undergo "constructive remodeling" — host cells infiltrate the scaffold, degrade it over time, and replace it with site-appropriate tissue rather than scar. Key products include porcine SIS (Oasis), porcine urinary bladder matrix (MatriStem), bovine pericardium (PriMatrix), and ovine forestomach matrix (Endoform). Application requires a clean, granulating wound bed free of necrotic tissue and active infection.
Collagen Dressings & ECM Products
Collagen-based wound dressings (bovine, porcine, equine, or avian sourced) serve as "sacrificial substrates" for excess MMPs in chronic wounds. Elevated MMP-2 and MMP-9 in chronic wound fluid degrade endogenous collagen and growth factors. Exogenous collagen preferentially binds and is degraded by these MMPs, protecting the wound's native repair molecules. Collagen also provides a scaffold for fibroblast migration and proliferation. Products are available as sheets, particles, gels, and pads (e.g., Promogran, Puracol Plus, Endoform).
Cellular and/or Tissue-Based Products (CTPs) — Coverage Criteria
Medicare covers cellular and tissue-based products (CTPs) for DFUs and VLUs that meet specific criteria. DFU requirements: Full-thickness neuropathic ulcer (Wagner grade 1 or higher); present ≥30 days despite standard of care; adequate blood supply (TcPO2 ≥30 mmHg, ABI 0.7-1.2, or toe pressure ≥30 mmHg); free of infection; patient is compliant with offloading. VLU requirements: Present ≥30 days despite standard of care including compression therapy; ABI ≥0.8 (confirming adequate arterial supply for compression); no active infection. General requirements: Wound must be clean and granulating at time of application; maximum number of applications varies by product (typically 1-5 applications, some products limited to 1); documentation of wound measurements before and after each application; re-application only if wound shows measurable improvement (prevents continued application to non-responding wounds).
10 Negative Pressure Wound Therapy
Negative pressure wound therapy (NPWT), also known as vacuum-assisted closure (VAC), applies sub-atmospheric pressure to a wound through a sealed dressing system. Since its introduction in the 1990s, NPWT has become one of the most widely used adjunctive wound therapies.
Mechanisms of Action
Macrodeformation: The negative pressure mechanically draws wound edges together, reducing wound volume and surface area. Microdeformation: At the foam-wound interface, the negative pressure creates microstrain on cells, stimulating cell proliferation, angiogenesis, and granulation tissue formation via mechanotransduction pathways (similar to the distraction osteogenesis principle). Fluid removal: Active removal of excess wound exudate decreases interstitial edema, reduces bacterial burden in the fluid, and removes inhibitory factors (MMPs, pro-inflammatory cytokines). Stabilization of the wound environment: The sealed dressing maintains moist wound healing, insulates the wound, and provides a barrier to external contamination.
Settings and Application
| Parameter | Standard Setting | Details |
|---|---|---|
| Pressure | -75 to -125 mmHg | -125 mmHg is the most commonly used setting; -75 mmHg may be used for pain-sensitive patients, grafts, or flaps; pressures >-200 mmHg offer no additional benefit and increase pain |
| Mode | Continuous or intermittent | Continuous: standard for most wounds, initial therapy, and patient comfort. Intermittent (5 min on/2 min off): may stimulate more granulation tissue but is less well-tolerated due to cycling discomfort; generally used after initial 48 hours |
| Filler | Polyurethane (PU) foam or polyvinyl alcohol (PVA) foam | PU foam (black, reticulated): more aggressive granulation stimulation, larger pore size (400-600 μm); PVA foam (white, denser): gentler, used over grafts, tendons, or when granulation ingrowth must be minimized |
| Dressing change | Every 48-72 hours | Monday-Wednesday-Friday schedule is common; more frequent changes for infected wounds; silver-impregnated foam (Granufoam Silver) may allow extended wear |
Indications
Acute and traumatic wounds, dehisced surgical wounds, chronic wounds (pressure injuries, DFUs, venous ulcers) failing standard therapy, flap and graft management (bolster dressing), open abdominal wounds (abdominal NPWT/wound VAC), preparation of wound bed for definitive closure, and reduction of edema in complex wounds.
Contraindications
Malignancy in the wound: NPWT stimulates cell proliferation. Untreated osteomyelitis: Must be treated with antibiotics and/or surgical debridement before NPWT. Non-enteric and unexplored fistulae: Risk of damaging underlying structures. Necrotic tissue with eschar: Must debride first; NPWT does not debride. Exposed blood vessels or organs: Risk of hemorrhage (vessels) or damage (organs); must be covered with protective layer or contact layer. Active hemorrhage.
Troubleshooting Common NPWT Problems
| Problem | Likely Cause | Solution |
|---|---|---|
| Seal leak (alarm sounding) | Irregular skin surface, moisture under drape, wrinkles in drape, hair, undermining/tunneling creating air channel | Dry periwound skin; apply skin prep or ostomy paste to uneven areas before drape; shave hair if needed; add extra drape over leak sites; use hydrocolloid strips to bridge irregular contours |
| Excessive pain | Foam adhering to wound bed, pressure too high, granulation tissue ingrowth into foam | Reduce pressure to -75 mmHg; switch to PVA (white) foam; place non-adherent contact layer (Mepitel) between foam and wound bed; pre-medicate before dressing changes; instill saline 15-20 min before removal to loosen foam |
| Bleeding during dressing change | Granulation tissue ingrowth into PU foam; vascular friable tissue | Use PVA foam or contact layer; reduce time between changes; moisten foam before removal; hold pressure if bleeding occurs; assess for exposed vessels (contraindication) |
| Canister filling rapidly | High-output wound; fistula output; enteric communication | Check for unexpected enteric communication; increase canister size or frequency of canister changes; assess if wound is appropriate for continued NPWT |
| Wound bed not granulating | Inadequate perfusion, persistent biofilm, systemic factors, pressure insufficient | Reassess vascular supply (ABI/TcPO2); debride and re-evaluate for biofilm; address nutrition and systemic factors; consider increasing to -125 mmHg or switching to intermittent mode |
NPWT with Instillation (NPWTi-d)
NPWTi-d (negative pressure wound therapy with instillation and dwell time) combines standard NPWT with automated instillation of a topical wound solution (most commonly normal saline or dilute antiseptic such as 0.1% dakin solution or PHMB). The solution is instilled into the wound, held for a programmable dwell time (typically 10-20 minutes), then aspirated by the negative pressure. This cycle repeats every 2-4 hours. Indicated for infected wounds, wounds with heavy biofilm burden, and wounds requiring frequent irrigation. Evidence demonstrates reduced time to wound closure and decreased biofilm reformation compared to standard NPWT alone.
Portable and Disposable NPWT
Single-use, disposable NPWT systems (e.g., PICO, SNaP) provide a smaller, quieter, canister-free alternative to traditional NPWT. These battery-powered devices deliver continuous negative pressure through an absorbent multilayer dressing that manages exudate via evaporation rather than canister collection. Advantages include improved patient mobility and quality of life, ease of application in outpatient settings, and lower cost for smaller wounds. Typical settings are -80 mmHg continuous. Indicated for closed surgical incisions (prophylactic iNPWT), low-to-moderate exudate wounds, and wounds appropriate for transitioning from traditional NPWT to a lighter system as exudate decreases. Not appropriate for large, heavily exudating wounds or wounds requiring instillation therapy.
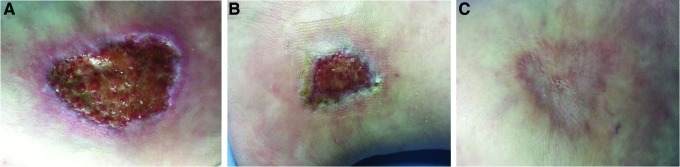
11 Debridement Methods
Debridement — the removal of devitalized tissue, debris, and biofilm from a wound — is the single most important intervention in chronic wound management. Necrotic tissue provides a medium for bacterial growth, obscures wound assessment, impedes granulation tissue formation, and prevents epithelialization. The method of debridement is selected based on wound characteristics, patient status, clinical setting, and clinician skill.
Debridement Methods Compared
| Method | Mechanism | Speed | Selectivity | Best Suited For | Limitations |
|---|---|---|---|---|---|
| Sharp (conservative) | Scalpel, scissors, curette used to remove devitalized tissue at bedside, stopping at viable tissue plane | Fast | Selective (experienced clinician) | Slough, thin eschar, callus, wound edge debridement; can be performed by trained nurses and podiatrists | Requires skill and training; risk of bleeding; may require topical anesthesia; not for large areas of thick eschar |
| Surgical | Excision in operating room, may include resection into viable tissue (margins) | Fastest | Non-selective (intentional margin of normal tissue) | Large areas of necrosis, deep wound infection/abscess, osteomyelitis requiring bone resection, necrotizing fasciitis | Requires OR, anesthesia; higher cost; greater tissue loss; bleeding risk; patient must tolerate surgery |
| Autolytic | Body's own enzymes and moisture within an occlusive or moisture-retentive dressing (hydrogel, hydrocolloid, film) soften and liquefy necrotic tissue | Slowest (days to weeks) | Highly selective (only dissolves devitalized tissue) | Patients who cannot tolerate sharp debridement; thin slough; small wounds; palliative care | Very slow; not for infected wounds; requires intact immune function; contraindicated with large amounts of necrotic tissue |
| Enzymatic | Topical application of exogenous enzymes that digest necrotic tissue; collagenase (Santyl/Clostridiopeptidase A) is the only FDA-approved enzymatic debrider in the US | Moderate (days) | Selective | Patients who cannot tolerate sharp debridement; long-term care setting; maintenance debridement between sharp sessions | Slow; expensive; inactivated by silver, heavy metals, acidic solutions, and detergents; must cross-hatch eschar for penetration; apply to moist wound only |
| Mechanical | Physical removal: wet-to-dry gauze (strongly discouraged — non-selective, painful, damages viable tissue), hydrotherapy/whirlpool, pulsed lavage with suction, ultrasonic debridement | Moderate | Non-selective (wet-to-dry); selective (pulsed lavage, ultrasonic) | Pulsed lavage for large wounds with loose debris; ultrasonic (low-frequency) for biofilm disruption | Wet-to-dry is outdated and causes pain and tissue damage; whirlpool risk of cross-contamination; pulsed lavage may cause aerosolization |
| Biological (maggot therapy) | Sterile larvae of Lucilia sericata (green bottle fly) secrete proteolytic enzymes that digest necrotic tissue, disinfect by killing bacteria (including MRSA), and stimulate granulation through growth factor release | Fast (2-3 days per application) | Highly selective (only digest dead tissue) | Wounds with significant necrotic tissue where sharp debridement is impractical; patients on anticoagulants; MRSA-colonized wounds | Patient/family acceptance (psychological barrier); may cause tingling/crawling sensation; contraindicated in wounds communicating with body cavities or large blood vessels; require containment (BioBag) |
Ultrasonic Debridement
Low-frequency ultrasonic debridement (LFUD) uses ultrasonic energy (typically 20-40 kHz) transmitted through a saline mist to the wound surface. The mechanical energy disrupts biofilm, loosens necrotic tissue, and promotes debridement while preserving viable tissue. Two modes of action are involved: (1) cavitation — formation and implosion of microscopic bubbles that mechanically disrupt cell membranes and biofilm matrix, and (2) acoustic microstreaming — fluid movement around the probe tip that facilitates debris removal and enhances drug penetration. Advantages over sharp debridement include reduced pain (can be performed without anesthesia in many patients), selective tissue removal, biofilm disruption at a depth sharp instruments cannot reach, and stimulation of growth factor release. Devices include contact (e.g., SonicOne) and non-contact (e.g., MIST Therapy) systems. LFUD is particularly useful for wounds with heavy biofilm burden, patients who cannot tolerate sharp debridement (anticoagulated, pain-sensitive), and as an adjunct to sharp debridement for biofilm management.
Hydrosurgical Debridement
Versajet is the primary hydrosurgical debridement device, delivering a high-pressure saline jet across a small window in a handheld tip. The Venturi effect created by the jet simultaneously cuts and aspirates tissue, allowing precise, controlled debridement of necrotic tissue while preserving viable structures. The power level is adjustable (1-10), allowing the operator to match the aggressiveness of debridement to the tissue type. Particularly useful for tangential debridement of burns (excision to viable dermis), removal of biofilm from large wound surfaces, and debridement around delicate structures (tendons, nerves). Requires training for safe and effective use.
Maintenance Debridement
The concept of maintenance debridement recognizes that a single debridement episode is insufficient for chronic wounds. Biofilm reconstitutes within 24-72 hours of disruption. Serial debridement at each wound visit (typically weekly) maintains a clean wound bed, disrupts biofilm reformation, removes senescent wound edge cells, and stimulates the acute wound healing response. Maintenance debridement is associated with improved healing rates in both DFUs and venous ulcers.
Wound Hygiene Protocol
The emerging concept of wound hygiene provides a structured approach to biofilm-based wound care at every dressing change. The four-step protocol includes:
Step 1 — Cleanse: Irrigate the wound and periwound skin with an antiseptic solution (PHMB with betaine surfactant or hypochlorous acid). Surfactant-based cleansers are more effective at disrupting biofilm EPS than saline alone. Extend cleansing to the periwound skin (at least 5 cm beyond the wound edge) to address the "biofilm penumbra" — bacteria extending into surrounding tissue beyond the visible wound margin.
Step 2 — Debride: Perform sharp debridement of the wound bed (remove slough, non-viable tissue, and biofilm) and wound edges (remove callus, rolled edges/epibole, and non-migrating epithelium). Debridement of the wound edge refreshes the epithelial front and stimulates keratinocyte migration.
Step 3 — Refashion wound edges: Sharp debridement of the wound perimeter removes the biofilm penumbra, senescent cells at the wound margin, and hyperkeratotic tissue. This step converts the non-advancing chronic wound edge back to an acute edge with renewed migratory potential.
Step 4 — Dress: Apply an antimicrobial dressing immediately after debridement to suppress biofilm reformation during the critical 24-72 hour window before biofilm reestablishes. Select the dressing based on wound bed characteristics, exudate level, and infection status.
12 Wound Infection Management
All chronic wounds contain bacteria; the clinical significance depends on the quantity, virulence, and host response. The wound infection continuum progresses from contamination through colonization, critical colonization (local infection), local infection, spreading infection, and systemic infection.
Clinical Signs of Wound Infection
N — Non-healing wound (despite appropriate care for ≥2 weeks). E — Exudate increasing. R — Red, friable granulation tissue (bleeds easily, dark beefy red, hypergranulation). D — Debris (new necrotic tissue or slough appearing in a previously clean wound). S — Smell (malodor). ≥3 of 5 NERDS criteria suggests superficial/critical colonization → treat with topical antimicrobials, NOT systemic antibiotics.
S — Size increasing. T — Temperature elevated (wound and periwound ≥3°F/1.7°C warmer than surrounding skin). O — Os (probe to bone). N — New areas of breakdown (satellite lesions or wound edge deterioration). E — Exudate, erythema, edema. E — Exudate purulent or spreading erythema. S — Smell. ≥3 of 7 STONEES criteria suggests deep infection → systemic antibiotics indicated plus topical antimicrobials and debridement.
Wound Culture Technique
Levine quantitative swab technique: The recommended method for wound culture. Cleanse the wound with saline, remove necrotic tissue. Rotate a sterile swab over a 1 cm2 area of clean granulation tissue with sufficient pressure to express fluid from the wound tissue (not superficial exudate). Bacteria count >105 CFU/g tissue (or per swab equivalent) indicates critical colonization/infection. This technique correlates well with tissue biopsy quantitative culture (the true gold standard).
Tissue biopsy: The gold standard for wound culture. Provides quantitative culture (CFU/g tissue) and identifies organisms within the tissue, not just surface contaminants. Reserved for wounds not responding to empiric therapy or when accurate speciation is critical.
"Z" swab technique: An older method where a swab is rotated across the wound bed in a zigzag pattern covering the entire wound surface. Less accurate than the Levine technique because it samples superficial exudate and contaminants rather than tissue-level organisms. Not recommended for clinical decision-making. Needle aspiration: Useful for sampling closed abscesses or deep fluid collections. A 10 mL syringe with 22-gauge needle is inserted through intact, prepped skin into the fluid collection; aspirated material is sent for aerobic and anaerobic culture. Avoid aspirating through the open wound surface.
Empiric Antibiotic Selection
| Severity | Route | Coverage Target | Empiric Regimen Options |
|---|---|---|---|
| Mild (cellulitis <2 cm) | Oral | Gram-positive (primarily S. aureus, Streptococcus) | Cephalexin 500 mg QID, or dicloxacillin 500 mg QID. If MRSA risk: TMP-SMX DS BID or doxycycline 100 mg BID (add cephalexin for streptococcal coverage as TMP-SMX and doxycycline have variable streptococcal activity) |
| Moderate (cellulitis >2 cm, lymphangitis, deep structure involvement) | Oral or IV | Broad-spectrum (gram-positive + gram-negative ± anaerobes) | Amoxicillin-clavulanate 875/125 mg BID, or levofloxacin 750 mg daily + metronidazole 500 mg TID, or IV ampicillin-sulbactam 3 g q6h |
| Severe/limb-threatening (systemic toxicity, sepsis) | IV | Broad-spectrum including MRSA + gram-negative + anaerobic | Vancomycin (15-20 mg/kg q8-12h, trough 15-20 μg/mL) + piperacillin-tazobactam 4.5 g q6h, or vancomycin + meropenem 1 g q8h; consider adding metronidazole for enhanced anaerobic coverage in gas-forming or putrid infections |
Always obtain cultures before starting empiric antibiotics and narrow coverage when sensitivities return. Duration of therapy depends on severity: mild infections 1-2 weeks; moderate 2-3 weeks; severe 3-4 weeks (longer if osteomyelitis). Reassess at 48-72 hours — if not improving, broaden coverage, obtain imaging for abscess/osteomyelitis, and reconsider wound etiology.
Wound Infection in Special Populations
Immunocompromised patients (HIV/AIDS, organ transplant recipients, chemotherapy, chronic corticosteroids) may not exhibit classic signs of infection due to impaired inflammatory response. In these patients, subtle findings such as increasing wound size, new friable tissue, delayed healing, or increasing pain may be the only indicators of infection. Maintain a low threshold for culture and empiric antibiotic initiation. Patients with diabetes often have blunted inflammatory responses and impaired leukocyte function; up to 50% of diabetic foot infections lack fever, leukocytosis, or elevated inflammatory markers. Bite wounds (human and animal) have unique microbiology: Pasteurella multocida (cat/dog bites), Capnocytophaga canimorsus (dog bites in asplenic patients), Eikenella corrodens (human bites); empiric treatment with amoxicillin-clavulanate covers typical bite wound pathogens. Cat bites carry higher infection rates than dog bites due to deep puncture inoculation.
13 Osteomyelitis in Chronic Wounds
Osteomyelitis is a serious complication of chronic wounds, particularly diabetic foot ulcers and pressure injuries overlying bony prominences. Contiguous-spread osteomyelitis (from adjacent soft tissue infection to bone) is the predominant mechanism in wound-associated osteomyelitis, as opposed to hematogenous osteomyelitis seen in children and IV drug users.
Diagnosis
Probe-to-bone (PTB) test: A sterile blunt metal probe is inserted into the wound. If bone is palpable (hard, gritty surface felt at the wound base), the test is positive. In the context of a diabetic foot ulcer, a positive PTB test has a positive predictive value (PPV) of 89% for osteomyelitis. In low-prevalence settings, the negative predictive value is more useful. Combined with clinical suspicion (wound area >2 cm2, wound depth >3 mm, ESR >70 mm/hr), the PTB test guides imaging decisions.
Laboratory markers: ESR >70 mm/hr has ~90% sensitivity for osteomyelitis in DFU. CRP is less specific but useful for monitoring treatment response. WBC count is often normal in chronic osteomyelitis. Procalcitonin is elevated in systemic infection but not reliable for localized osteomyelitis.
Imaging: Plain radiographs are the first-line study — findings include periosteal reaction, cortical destruction, and medullary lucency, but changes require 40-50% bone loss to become visible (2-4 week lag). MRI is the imaging gold standard for osteomyelitis (sensitivity 90%, specificity 83%) — shows bone marrow edema (low signal on T1, high signal on T2/STIR), cortical disruption, and soft tissue extent. Nuclear medicine scans (triple-phase bone scan, WBC-labeled scan) are used when MRI is contraindicated.
Bone biopsy and culture: The diagnostic gold standard. Provides definitive microbiologic identification and antibiotic sensitivity. Percutaneous bone biopsy or intraoperative sample obtained from a separate clean incision (avoid contamination from wound surface bacteria). Culture results guide targeted antibiotic therapy.
Cierny-Mader Classification of Osteomyelitis
| Category | Classification | Description |
|---|---|---|
| Anatomic Type | Type I: Medullary | Infection confined to the medullary canal (endosteal surface); most common in hematogenous osteomyelitis |
| Type II: Superficial | Infection of the outer cortex from contiguous soft tissue infection; most common type in wound-associated osteomyelitis | |
| Type III: Localized | Full-thickness cortical involvement (both cortices + medullary canal) in a circumscribed area; stable bone, segmental resection does not create instability | |
| Type IV: Diffuse | Full-thickness cortical and medullary involvement requiring segmental resection that creates mechanical instability; may require reconstruction/stabilization | |
| Host Classification | A Host: Normal | Normal immune system, normal vascular supply, good wound healing potential |
| B Host: Compromised | Systemic (Bs) or local (Bl) compromise. Bs: immune deficiency, diabetes, malnutrition, chronic steroid use, extremes of age. Bl: radiation, scarring, vascular insufficiency, lymphedema. Bls: both systemic and local compromise. | |
| C Host: Prohibitive | Treatment is worse than disease; expected morbidity of treatment exceeds that of living with the disease; includes patients with prohibitive surgical risk or terminal illness |
Treatment
Antibiotic therapy: Culture-directed antibiotics for 6 weeks (standard duration for osteomyelitis). IV antibiotics traditionally used for at least the first 2-4 weeks, with transition to oral agents with high bioavailability (fluoroquinolones, rifampin combinations, linezolid) if appropriate. The OVIVA trial demonstrated non-inferiority of early switch to oral antibiotics at 1 week in selected patients. Surgical management: Debridement of infected/necrotic bone, curettage of medullary canal, resection to bleeding bone ("paprika sign"). Dead space management with antibiotic beads/spacers or muscle flaps. In DFU osteomyelitis, partial ray amputation or metatarsal head resection may be limb-preserving. Conservative (antibiotic-only) management may be appropriate for small areas of osteomyelitis in poor surgical candidates.
MSSA: Nafcillin/oxacillin IV, transition to cephalexin or dicloxacillin oral. MRSA: Vancomycin IV (trough 15-20 μg/mL), transition to TMP-SMX DS + rifampin 300 mg BID or linezolid 600 mg BID oral. Pseudomonas: Ceftazidime or piperacillin-tazobactam IV, transition to ciprofloxacin 750 mg BID oral. Enterococcus: Ampicillin IV (if susceptible), transition to amoxicillin oral. Polymicrobial (common in DFU): Ampicillin-sulbactam or piperacillin-tazobactam IV, transition based on culture sensitivities. Rifampin is added for biofilm-associated infections (penetrates biofilm) but must always be used in combination (rapid resistance develops with monotherapy). Monitor CRP (not ESR) for treatment response — CRP normalizes faster and is a more reliable indicator of infection resolution during antibiotic therapy.
14 Surgical Wounds & Dehiscence
Surgical wounds are the most common acute wounds encountered in clinical practice. They are classified by the method of closure and the level of contamination.
Postoperative Wound Care Phases
Immediate postoperative (0-48 hours): Surgical incisions closed by primary intention should be covered with a sterile occlusive dressing for 24-48 hours. During this period, the wound is sealed by the fibrin clot and early epithelial cell migration begins. The dressing protects against external contamination during this vulnerable period. Gentle cleansing with saline or mild soap and water may begin after 48 hours for most clean surgical wounds. Subacute phase (48 hours to 2 weeks): Monitor for signs of infection (erythema >2 cm from incision, increasing pain, purulent drainage, fever). Sutures or staples are typically removed based on anatomic location: face 3-5 days, scalp 7-10 days, trunk/extremities 10-14 days, over joints 14 days. Steri-Strips may be applied after suture removal for additional support during the continued remodeling phase. Maturation phase (2 weeks to 1-2 years): Scar management includes silicone sheeting or gel (evidence-based for scar prevention and treatment), sun protection (UV exposure causes permanent hyperpigmentation of immature scars), and avoidance of excessive tension on the incision line.
Wound Closure Methods
| Intention | Description | Examples |
|---|---|---|
| Primary intention | Wound edges are approximated with sutures, staples, adhesive, or tape; minimal granulation tissue needed; fastest healing; lowest scar | Surgical incisions, repaired lacerations |
| Secondary intention | Wound is left open to heal by granulation, contraction, and epithelialization from the base and edges; used when closure is not possible or not advisable | Pressure injuries, open abscesses, chronic wounds |
| Tertiary intention (delayed primary closure) | Wound is initially left open (for drainage, infection clearance, or edema resolution) and closed surgically days later | Contaminated traumatic wounds, infected surgical wounds, fasciotomy closure |
CDC Surgical Site Infection (SSI) Classification
| Wound Class | Description | Expected SSI Rate |
|---|---|---|
| Class I (Clean) | Non-traumatic, no inflammation, no entry into respiratory/GI/GU/biliary tracts; closed primarily; no breaks in aseptic technique | 1-3% |
| Class II (Clean-contaminated) | Controlled entry into respiratory/GI/GU/biliary tract without unusual contamination | 5-8% |
| Class III (Contaminated) | Open, fresh accidental wounds; operations with major breaks in sterile technique; gross spillage from GI tract; entry into GU/biliary tract with infected urine/bile | 10-15% |
| Class IV (Dirty-infected) | Old traumatic wounds with devitalized tissue; existing clinical infection; perforated viscera | 25-40% |
SSI Depth Classification
Superficial incisional SSI: Within 30 days of surgery; involves only skin and subcutaneous tissue; purulent drainage, positive culture, or deliberate opening by surgeon with signs/symptoms of infection. Deep incisional SSI: Within 30 days (or 90 days if implant in place); involves fascia and muscle layers; purulent drainage from deep incision, spontaneous dehiscence or deliberate opening with fever >38°C, localized pain/tenderness, or abscess on imaging. Organ/space SSI: Any part of the body deeper than the fascia/muscle that was opened or manipulated; abscess or infection identified involving organ/space.
Wound Dehiscence
Dehiscence is the partial or complete separation of wound edges after primary closure. Risk factors include obesity, diabetes, malnutrition, smoking, chronic steroid use, infection, poor surgical technique, increased abdominal pressure (coughing, vomiting, ileus), and radiation. Evisceration (protrusion of abdominal contents through a dehisced abdominal wound) is a surgical emergency requiring immediate coverage with sterile saline-moistened gauze, patient positioning (supine, knees bent), and urgent surgical intervention.
Management of non-evisceration dehiscence depends on depth, infection status, and patient factors: small superficial dehiscence may heal by secondary intention; deep dehiscence may require NPWT for wound bed preparation followed by delayed primary closure or skin grafting; infected dehiscence requires antibiotic therapy and source control.
Incisional NPWT (iNPWT)
Prophylactic application of negative pressure to closed surgical incisions reduces SSI rates, seroma formation, and dehiscence in high-risk patients (obese, revisional surgery, vascular surgery groin incisions, cesarean section in obese patients, joint arthroplasty). Applied in the OR immediately after closure. Devices include Prevena (KCI/3M) and PICO (Smith+Nephew). Standard treatment duration is 5-7 days (matching the period of highest SSI vulnerability). The mechanism involves lateral tissue approximation, reduced dead space, increased lymphatic drainage, reduced lateral tension on incision edges, and maintenance of a clean closed environment.
Meta-analyses demonstrate that iNPWT reduces SSI rates by approximately 50% in high-risk surgical incisions (NNT ~10-15 depending on baseline SSI risk). The strongest evidence supports use in: groin incisions for vascular surgery (femoral artery exposure), cesarean sections in patients with BMI ≥30, lower extremity fracture fixation, perineal wounds after abdominoperineal resection, and sternal wounds in patients with multiple risk factors for mediastinitis. Cost-effectiveness improves with higher baseline SSI risk — routine use on all surgical incisions is not recommended.
The National Nosocomial Infections Surveillance (NNIS) risk index predicts SSI risk using three equally weighted factors (1 point each): (1) ASA score ≥3, (2) wound classification contaminated or dirty (Class III or IV), (3) operation duration exceeding the 75th percentile for that procedure. Scores range 0-3, with SSI rates increasing from ~1% (score 0) to ~13% (score 3). Laparoscopic approach reduces the score by 1 point.
15 Burn Wound Management
Burns are tissue injuries caused by heat (thermal), electricity, chemicals, radiation, or friction. Burn wound management involves rapid assessment of burn depth and total body surface area (TBSA) to guide resuscitation, determine need for burn center referral, and select appropriate wound care.
Burn Depth Classification
| Depth | Old Terminology | Layers Involved | Appearance | Sensation | Healing |
|---|---|---|---|---|---|
| Superficial | First-degree | Epidermis only | Red, dry, no blisters (sunburn-like) | Painful | 3-5 days; no scarring |
| Superficial partial-thickness | Superficial second-degree | Epidermis + superficial (papillary) dermis | Pink, moist, blisters; brisk capillary refill; weeping | Very painful (exposed nerve endings) | 7-14 days; minimal scarring if no infection |
| Deep partial-thickness | Deep second-degree | Epidermis + deep (reticular) dermis | Pale/white, mottled, may have blisters; sluggish capillary refill; less moist | Decreased (many nerve endings destroyed) | 14-35 days; significant scarring; may need grafting; indeterminate burns may convert to full-thickness |
| Full-thickness | Third-degree | Epidermis + entire dermis (into subcutaneous tissue) | White, waxy, leathery, charred, or translucent; no blanching; thrombosed vessels visible; dry | Insensate (nerve endings destroyed) | Cannot heal by epithelialization (no skin appendages remain); requires skin grafting; heals only from wound edges by contraction |
TBSA Estimation
Rule of 9s (adults): Head/neck = 9%, each upper extremity = 9%, anterior trunk = 18%, posterior trunk = 18%, each lower extremity = 18%, perineum = 1%. Lund-Browder chart: More accurate, especially for children; adjusts proportions for age (children have proportionally larger heads and smaller extremities). Palm method: Patient's palm (including fingers) represents approximately 1% TBSA; useful for small or scattered burns. Note: superficial (first-degree) burns are NOT included in TBSA calculations for fluid resuscitation.
Fluid Resuscitation
4 mL × body weight (kg) × %TBSA of lactated Ringer's solution in the first 24 hours. Give 50% in the first 8 hours (from time of burn, NOT time of presentation) and 50% over the subsequent 16 hours. Titrate to urine output: adults 0.5-1.0 mL/kg/hr; children 1.0 mL/kg/hr. The Parkland formula is a starting point — adjust based on clinical response. "Fluid creep" (over-resuscitation) is a recognized complication leading to abdominal compartment syndrome, pulmonary edema, and extremity compartment syndrome.
Burn Center Referral Criteria (ABA)
Partial-thickness burns >10% TBSA; any full-thickness burn; burns involving face, hands, feet, genitalia, perineum, or major joints; chemical or electrical burns (including lightning); inhalation injury; burns in patients with preexisting medical conditions that could complicate management; burns with associated trauma where burn is the greater risk; burns in children at facilities without qualified pediatric care; burns requiring special social, emotional, or rehabilitative intervention.
Topical Burn Agents
| Agent | Spectrum | Advantages | Disadvantages |
|---|---|---|---|
| Silver sulfadiazine (SSD) 1% | Broad gram-positive, gram-negative, yeast | Painless application; soothing; widely available | Pseudoeschar formation (gray-white residue mimicking infection); may delay epithelialization; contraindicated in sulfa allergy, pregnancy near term, neonates; leukopenia (transient, benign) |
| Mafenide acetate 11% cream | Broad gram-positive, gram-negative; penetrates eschar (unique advantage) | Penetrates eschar and cartilage; effective for ear burns (chondritis prevention) and heavily contaminated burns | Painful on application; carbonic anhydrase inhibitor causing metabolic acidosis (hyperventilation to compensate); hypersensitivity reactions |
| Silver-impregnated dressings | Broad-spectrum (ionic or nanocrystalline silver) | Less frequent dressing changes (every 3-7 days vs daily for creams); less painful; no pseudoeschar | Cost; may require secondary dressing; some not suitable for deep partial/full-thickness burns |
| Bacitracin/Polymyxin B | Gram-positive (bacitracin), gram-negative (polymyxin B) | Appropriate for superficial burns and facial burns; transparent; minimal adverse effects | No eschar penetration; limited spectrum; contact dermatitis risk |
Escharotomy
Full-thickness circumferential burns produce a rigid, inelastic eschar that cannot expand. As underlying tissue edema increases during resuscitation, compartment pressures rise, compromising distal perfusion and, in the case of thoracic burns, restricting ventilation. Escharotomy is an emergent bedside procedure involving longitudinal incision through the full thickness of the eschar (but not into subcutaneous fat) along the mid-lateral or mid-medial lines of the extremity, or along the anterior axillary lines for chest escharotomy. Because full-thickness burns are insensate, local anesthesia is typically not required. Immediate restoration of perfusion (palpable pulses, capillary refill, improved SpO2) or ventilatory compliance confirms adequate release. Fasciotomy may be required for deep electrical burns or when escharotomy alone fails to restore perfusion, as these injuries may involve muscle compartment pressure elevation.
Skin Grafting
Split-thickness skin graft (STSG): Harvested with a dermatome at 0.008-0.012 inches (epidermis + partial dermis). Can be meshed (1:1.5 to 1:6 expansion) for large wounds. Donor site heals by epithelialization in 10-14 days. Workhorse of burn surgery. Full-thickness skin graft (FTSG): Includes epidermis + full dermis. Better cosmetic result, less contracture; limited donor tissue availability; used for face, hands, and small defects. Donor site requires primary closure.
Graft take factors: Successful graft adherence ("take") requires: (1) well-vascularized recipient bed (clean granulation tissue), (2) close contact between graft and bed (no hematoma, seroma, or shear), (3) adequate immobilization (bolster dressings, NPWT, or splinting for 5-7 days), and (4) absence of infection. Causes of graft failure include hematoma/seroma beneath the graft (most common technical cause), infection (especially Pseudomonas and beta-hemolytic Streptococcus, which produce fibrinolysins that dissolve the fibrin attachment), shear/mechanical disruption, and inadequate recipient bed vascularity.
16 Skin Tears
Skin tears are traumatic wounds caused by shear, friction, or blunt force resulting in separation of the skin layers. They are the most common wound in elderly and neonatal populations, occurring primarily on the extremities (dorsal forearm and hands most common) due to age-related skin fragility (dermal thinning, decreased collagen, loss of rete ridges, decreased subcutaneous fat, increased capillary fragility).
ISTAP Classification (International Skin Tear Advisory Panel)
| Type | Description | Management Approach |
|---|---|---|
| Type 1: No skin loss | Linear or flap tear that can be repositioned to cover the wound bed | Gently cleanse, approximate the skin flap using moist cotton-tipped applicator, secure with skin adhesive strips (Steri-Strips) or tissue adhesive; cover with non-adherent contact layer + foam or silicone dressing |
| Type 2: Partial flap loss | Partial skin flap loss that cannot be repositioned to cover the entire wound bed | Cleanse, realign available flap to cover maximum wound bed, secure as above; cover exposed area with moisture-retentive dressing (hydrogel, silicone contact layer) |
| Type 3: Total flap loss | Complete loss of the skin flap exposing the entire wound bed | Cleanse, cover with moisture-retentive dressing (hydrogel sheet, foam, or silicone contact layer); may require skin substitute for large Type 3 tears |
Payne-Martin Classification (Historical)
Category I: Skin tear without tissue loss. Ia: linear type (epidermis and dermis pulled apart in a straight line). Ib: flap type (epidermal flap that can cover the dermis). Category II: Skin tear with partial tissue loss. IIa: scant tissue loss (≤25% of flap lost). IIb: moderate to large tissue loss (>25% of flap lost). Category III: Skin tear with complete tissue loss (entire epidermal flap absent).
Prevention in the Elderly
Moisturize skin BID with emollient creams (not lotions — lotions evaporate and may further dry skin). Protect extremities with long sleeves and shin guards. Pad bed rails and wheelchair arms. Use gentle adhesives (silicone-based tapes such as Mepitac, paper tape). Remove adhesives with adhesive remover rather than peeling. Maintain adequate nutrition and hydration. Use proper transfer and turning technique (lift, do not slide). Apply skin protectant (dimethicone or cyanoacrylate barrier) to vulnerable areas.
Risk Factors for Skin Tears
Intrinsic factors: Age ≥75 years, history of previous skin tears (strongest predictor of future tears), chronic corticosteroid use (thins dermis and impairs collagen synthesis), anticoagulant therapy, malnutrition and dehydration, cognitive impairment or agitation, impaired mobility requiring assistance with transfers, ecchymosis or senile purpura, peripheral neuropathy, and dry/flaky skin. Extrinsic factors: Mechanical forces during transfers, repositioning, and ADLs; adhesive product removal; wheelchair and bed rail contact; falls; ill-fitting clothing or shoes; fingernail scratching (patient or caregiver); and environmental hazards (sharp edges on furniture, equipment). Assessment of these risk factors allows targeted prevention strategies in high-risk patients.
17 Atypical Wounds
Atypical wounds are those whose etiology is not related to the common causes (pressure, neuropathy, venous/arterial insufficiency, or surgery). They should be suspected when a wound has an unusual appearance, location, or fails to respond to standard evidence-based treatment after 4-12 weeks. Biopsy of the wound edge is often required for diagnosis.
Pyoderma Gangrenosum (PG)
A neutrophilic dermatosis presenting as a rapidly progressive, painful ulcer with violaceous, undermined borders and a purulent/necrotic base. Key feature: pathergy — worsening with surgical debridement or trauma (new lesions at surgical sites). Associated with inflammatory bowel disease (ulcerative colitis > Crohn's), rheumatoid arthritis, hematologic malignancy (AML, myeloma), and monoclonal gammopathy. Diagnosis is clinical (no pathognomonic histology); must exclude mimics (infection, vascular, malignancy). Treatment: immunosuppression (systemic corticosteroids first-line, cyclosporine, infliximab, dapsone); local wound care (moist wound healing, NPWT with caution); avoid surgical debridement (pathergy risk).
Calciphylaxis (Calcific Uremic Arteriolopathy)
A life-threatening condition of systemic arteriolar calcification and skin necrosis, primarily affecting patients with end-stage renal disease (ESRD) on dialysis, though non-uremic calciphylaxis can occur in patients with normal renal function who have other risk factors. Presents as exquisitely painful, retiform (net-like) purpura progressing to necrotic ulcers, most commonly on the lower extremities, abdomen, and buttocks. Mortality is 60-80% at 1 year, primarily from sepsis secondary to wound infection.
Risk factors: ESRD (on hemodialysis or peritoneal dialysis), hyperparathyroidism (secondary or tertiary), elevated calcium-phosphorus product (>70 mg2/dL2), warfarin use (inhibits matrix Gla protein, a calcification inhibitor), obesity (BMI >30 — adipose tissue is preferentially affected), diabetes, female sex, hypoalbuminemia, and protein C or S deficiency.
Diagnosis: Skin biopsy shows pathognomonic findings of arteriolar medial calcification, intimal hyperplasia, thrombosis, and subcutaneous fat necrosis. Von Kossa staining highlights calcium deposits. However, biopsy carries risk of wound expansion and should be performed cautiously (use punch biopsy from the wound edge, avoid large excisional biopsies).
Treatment: Sodium thiosulfate (25 g IV three times weekly during dialysis — chelates calcium, antioxidant, vasodilator); intensify dialysis (daily or extended sessions to optimize phosphorus clearance); correct hyperparathyroidism (cinacalcet for medical management, parathyroidectomy for refractory cases); discontinue warfarin and calcium-based phosphate binders (switch to sevelamer or lanthanum); wound care with meticulous pain management (often requiring opioids); surgical debridement controversial (high morbidity, poor healing capacity in affected tissue); HBOT may be considered as adjunctive therapy. Multidisciplinary management with nephrology, wound care, pain management, and nutrition is essential.
Vasculitis Ulcers
Ulcers resulting from inflammation and destruction of blood vessel walls. Presentation varies by vessel size: small-vessel vasculitis (palpable purpura, petechiae, ulcers on lower legs), medium-vessel (livedo reticularis, nodules, deep ulcers), large-vessel (claudication, ischemic ulcers). Causes include autoimmune (SLE, RA, ANCA-associated vasculitis), infection (hepatitis B/C), drug-induced, and cryoglobulinemia. Diagnosis requires punch biopsy of the wound edge (including subcutaneous tissue for medium/large vessel) showing vessel wall inflammation and fibrinoid necrosis. Treatment: address underlying cause; immunosuppression (corticosteroids, cyclophosphamide, rituximab); local wound care.
Martorell Ulcer (Hypertensive Ischemic Ulcer)
A rare but frequently misdiagnosed ulcer caused by arteriolar narrowing and ischemia in patients with severe, often poorly controlled hypertension. Presents as an exquisitely painful ulcer on the lateral or posterolateral lower leg or Achilles tendon area — in contrast to the medial location of venous ulcers. The wound is typically well-demarcated with a necrotic base and violaceous borders. Arteriolar hyalinosis and subintimal fibrosis are seen on deep biopsy. Key differentiating feature from calciphylaxis: Martorell ulcers occur in patients with hypertension but normal renal function and normal calcium-phosphorus metabolism. Treatment includes aggressive blood pressure control, pain management (often requiring opioids), local wound care (moist wound healing, cautious debridement), and consideration of surgical closure with skin grafting once the wound bed is prepared.
When to Biopsy a Wound
Radiation Dermatitis & Radiation Ulcers
Acute radiation dermatitis occurs during radiation therapy and follows a predictable dose-dependent progression:
| Grade (CTCAE) | Dose Range (approximate) | Presentation | Management |
|---|---|---|---|
| Grade 1 | ~10-20 Gy | Faint erythema, dry desquamation, mild pruritus | Moisturizing cream, gentle cleansing, avoid irritants |
| Grade 2 | ~20-40 Gy | Moderate erythema, patchy moist desquamation (confined to skin folds), moderate edema | Hydrogel sheets, silicone foam dressings, silver sulfadiazine for moist areas, topical steroid for pruritus |
| Grade 3 | ~40+ Gy | Confluent moist desquamation beyond skin folds, bleeding induced by minor trauma | Antimicrobial dressings (silver, honey), gentle non-adherent dressings, may require treatment interruption |
| Grade 4 | High dose | Full-thickness skin necrosis, ulceration, spontaneous hemorrhage | Debridement, advanced wound care, HBOT, surgical consultation |
Late radiation injury (months to years post-treatment) presents as chronic non-healing ulcers in irradiated tissue with impaired vascularity, fibrosis, and poor healing potential. The underlying pathophysiology is obliterative endarteritis — progressive fibrosis and occlusion of small blood vessels in the radiation field, creating a hypoxic, hypocellular, hypovascular (the "3H" tissue) environment. Treatment: pentoxifylline + vitamin E (PENTOCLO protocol for radiation fibrosis); hyperbaric oxygen therapy (promotes angiogenesis in radiation-damaged tissue — approved indication; 40-60 sessions typically required); gentle wound care with moisture balance; surgical reconstruction with well-vascularized tissue (free flaps from outside the radiation field) for refractory ulcers.
Malignant Wounds (Fungating Tumors)
Wounds caused by primary skin cancer or metastatic cancer infiltrating the skin. Present as fungating (proliferative, cauliflower-like growth above the skin surface), ulcerating (crater-like erosion into tissue), or combined fungating-ulcerating lesions. Most common primary tumors causing fungating wounds include breast cancer (60-70%), head and neck cancers, and melanoma. Management is often palliative, focusing on:
Odor control: Malodor is caused by anaerobic bacteria colonizing necrotic tissue; treat with topical metronidazole gel 0.75% BID (directly targets anaerobes), activated charcoal dressings (adsorb volatile odor compounds), cadexomer iodine, or medical-grade honey. Environmental measures include room ventilation, charcoal filters, and scented products (used cautiously — some patients develop aversive associations with specific scents).
Bleeding management: Tumor neovascularization produces friable vessels prone to hemorrhage. Use atraumatic dressing changes with silicone contact layers (Mepitel), topical tranexamic acid (500 mg tablet crushed and dissolved in 5 mL saline, applied on gauze), alginate dressings for hemostasis, topical epinephrine (1:1000 on gauze), silver nitrate cautery sticks, or sucralfate paste. Avoid sharp debridement of actively vascularized tumor tissue. For catastrophic hemorrhage (erosion into major vessel), have dark-colored towels available to minimize visual distress, and ensure comfort measures are prioritized.
Exudate management: Superabsorbent dressings, hydrofiber or alginate primary layers, and frequent dressing changes. NPWT may be used cautiously if no exposed vessels. Pain management: Topical lidocaine 2-4% applied 15-20 minutes before dressing changes, topical morphine (mixed with hydrogel), systemic analgesics, and anxiolytics as needed. Psychosocial support: Fungating wounds cause significant body image distress, social isolation, and reduced quality of life; involve palliative care and psychosocial support services early.
18 Hyperbaric Oxygen Therapy
Hyperbaric oxygen therapy (HBOT) involves breathing 100% oxygen at pressures greater than 1 atmosphere absolute (ATA) in a pressurized chamber. The resulting supraphysiologic tissue oxygen tensions produce therapeutic effects that support wound healing in selected patients.
Mechanisms of Action
Hyperoxia: Tissue pO2 increases from normal ~40 mmHg to 200-400 mmHg during HBOT, creating an oxygen gradient that drives oxygen diffusion into hypoxic wound tissue. Angiogenesis: The oxygen gradient at the wound edge (high pO2 in healthy tissue, low pO2 in wound center) is the primary stimulus for VEGF release and new blood vessel formation. HBOT enhances this gradient. Bactericidal activity: Elevated oxygen tension directly kills obligate anaerobes and enhances oxidative killing by neutrophils (respiratory burst requires O2). Synergistic with aminoglycosides and fluoroquinolones (which require O2 for bacterial cell entry). Anti-inflammatory effects: Reduces neutrophil adhesion to endothelium, decreases edema, and modulates cytokine production. Stem cell mobilization: HBOT mobilizes bone marrow-derived stem/progenitor cells (CD34+) into the circulation, promoting vasculogenesis.
Approved Indications for HBOT (UHMS)
The Undersea and Hyperbaric Medical Society (UHMS) recognizes 14 approved indications for HBOT. Wound-related indications include:
| Indication | Protocol Details | Evidence Level |
|---|---|---|
| Diabetic foot ulcers | Wagner grade ≥3, failing ≥30 days of standard care, TcPO2 criteria met, adequate macrovascular supply; 30-40 sessions at 2.0-2.4 ATA | Level 1 (RCTs); Medicare-covered with documentation of standard care failure |
| Delayed radiation injury | Soft tissue radionecrosis, bony radionecrosis, radiation cystitis, proctitis, laryngeal necrosis; 40-60 sessions at 2.0-2.4 ATA | Level 2-3; strong clinical experience |
| Osteoradionecrosis prevention | Marx protocol: 20 pre-extraction + 10 post-extraction sessions for dental extraction in irradiated mandible (≥60 Gy); 2.4 ATA | Level 2; standard of care in radiation oncology |
| Compromised grafts and flaps | Failing flaps or grafts with evidence of ischemia; emergent initiation; 2.0-2.5 ATA; continue until graft/flap viability assured | Level 3; clinical series |
| Chronic refractory osteomyelitis | Adjunctive to surgery and antibiotics; 20-40 sessions at 2.0-2.4 ATA | Level 2-3 |
| Gas gangrene (clostridial myonecrosis) | Emergent; 3 sessions in first 24 hours at 3.0 ATA; adjunctive to surgery and antibiotics | Level 3; strong physiologic rationale |
| Necrotizing soft tissue infections | Adjunctive to aggressive surgical debridement and IV antibiotics; 2.0-2.5 ATA BID initially | Level 3; retrospective data suggest mortality reduction |
Treatment Protocol
Standard wound healing protocol: 2.0-2.4 ATA for 90 minutes of oxygen breathing time per session (with air breaks every 20-30 minutes to prevent oxygen toxicity). Sessions are daily, 5 days per week. Total course: 30-60 sessions depending on indication and response. DFUs typically receive 30-40 sessions. Radiation injury may require 40-60 sessions.
Air breaks: Patients breathe air (not 100% O2) for 5 minutes after every 20-30 minutes of oxygen breathing. Air breaks reduce the cumulative oxygen dose to the lungs and CNS, significantly decreasing the risk of pulmonary and CNS oxygen toxicity without diminishing wound healing benefit. A typical 90-minute treatment session includes three 30-minute oxygen periods separated by two 5-minute air breaks.
Chamber Types
Monoplace chambers: Single-patient acrylic chambers pressurized entirely with 100% oxygen. The patient lies supine inside the chamber. Advantages include lower cost, smaller footprint, and simpler operation. Limitations include inability for staff to directly access the patient during treatment, claustrophobia risk, and the entire chamber atmosphere is oxygen (higher fire risk — all materials inside must be approved for oxygen-enriched environments; no electronics, no petroleum-based products). Multiplace chambers: Room-sized steel chambers accommodating multiple patients and an inside attendant (nurse or technician). Chamber is pressurized with compressed air; patients breathe 100% oxygen through a hood, mask, or endotracheal tube. Advantages include direct bedside patient care, ability to treat critically ill or ventilated patients, reduced claustrophobia, and lower fire risk (chamber atmosphere is air). Disadvantages include higher construction and operating costs and larger space requirements.
Complications of HBOT
| Complication | Mechanism | Frequency | Management |
|---|---|---|---|
| Middle ear barotrauma | Inability to equalize middle ear pressure during compression (eustachian tube dysfunction) | Most common complication (2-5%) | Teach equalization techniques (Valsalva, jaw thrust, swallowing); slow compression rate; nasal decongestant pre-treatment; myringotomy tubes for refractory cases |
| Sinus barotrauma | Trapped air in sinus cavities unable to equalize | Uncommon | Nasal decongestant; avoid treatment during active URI; slow compression/decompression |
| CNS oxygen toxicity | Seizure from prolonged hyperoxia at high pressures (Paul Bert effect) | Rare (1-3 per 10,000 treatments) | Immediately remove O2 source (switch to air); seizures are self-limited after O2 removal; reduce treatment pressure or shorten O2 periods; air breaks are primary prevention |
| Pulmonary oxygen toxicity | Prolonged O2 exposure causing tracheobronchitis and decreased vital capacity | Rare with standard protocols | Monitor for cough, chest tightness, dyspnea; limit daily O2 exposure; air breaks; rest days (weekend breaks) |
| Myopia (reversible) | Oxidative changes to lens proteins causing increased refractive power | Common with prolonged courses (20-30% after 20+ sessions) | Reversible within 6-8 weeks of completing treatment; counsel patients before treatment course; avoid new corrective lens prescriptions during HBOT |
Contraindications
Absolute: Untreated pneumothorax (risk of tension pneumothorax during decompression). Relative: Uncontrolled seizure disorder (oxygen toxicity lowers seizure threshold), concurrent bleomycin or cisplatin use (enhanced pulmonary toxicity), severe COPD with CO2 retention (loss of hypoxic drive concern — largely theoretical), untreated malignancy in the treatment field (concern for tumor growth — evidence limited), claustrophobia (monoplace chambers), active upper respiratory infection or inability to equalize middle ear pressure, pregnancy (theoretical teratogenicity at high O2 tensions — not well studied), and high fever (lowers seizure threshold synergistically with hyperoxia).
Periwound TcPO2 is used to select patients most likely to benefit from HBOT. TcPO2 <40 mmHg on room air: indicates hypoxic wound — potential HBOT candidate. In-chamber TcPO2 challenge: if periwound TcPO2 rises >200 mmHg while breathing 100% O2 at 2.4 ATA, the patient is likely to respond to HBOT. If TcPO2 does not rise >200 mmHg in-chamber, vascular supply is likely insufficient and revascularization should be pursued before HBOT.
19 Offloading & Compression
Mechanical offloading (reducing pressure on the wound) and compression therapy (applying external pressure to the limb) are the foundational non-pharmacologic treatments for diabetic foot ulcers and venous leg ulcers, respectively. No wound product can overcome the continued mechanical insult of unaddressed pressure or venous hypertension.
Offloading for Diabetic Foot Ulcers
| Device | Efficacy | Advantages | Disadvantages |
|---|---|---|---|
| Total contact cast (TCC) | Gold standard; ~90% healing rate at 12 weeks; healing time 6-8 weeks average | Non-removable (ensures adherence); distributes pressure across entire plantar surface; reduces shear; reduces activity level | Requires trained applicator; cannot inspect wound daily; risk of cast-related skin breakdown; contraindicated with acute infection, deep abscess, significant ischemia, fluctuating edema |
| Irremovable walker | Equivalent to TCC when rendered non-removable (~85-90% healing rate) | Easier to apply than TCC; removable for wound inspection by clinician; rendered irremovable with cohesive wrap or strap | Heavier than TCC; bulky; requires patient to use assistive device for ambulation |
| Removable walker (CAM boot) | ~50-60% healing rate (reduced by non-adherence) | Easy application; allows daily wound inspection; patient can remove for hygiene and sleep | Adherence is the major limiting factor — patients wear removable devices only ~28% of the day; inferior healing rates unless made irremovable |
| Therapeutic footwear | Prevention-focused (depth shoes reduce reulceration by 50%) | Depth shoes accommodate custom-molded insoles and foot deformity; rocker-bottom soles reduce forefoot pressure; lifelong prescription after DFU | Not sufficient for acute DFU offloading; requires proper fitting; patient adherence variable |
| Felted foam/accommodative padding | Temporary/supplementary | Inexpensive; easy to apply; can be custom-cut to offload specific areas; used as adjunct to other modalities | Not adequate as sole offloading method for plantar DFU; needs frequent replacement |
Offloading Special Situations
Heel ulcers: Heel offloading requires complete elimination of pressure on the heel (unlike forefoot offloading which redistributes pressure). Options include heel suspension devices (Prevalon boot, DH Pressure Relief Walker), pillows placed lengthwise under the calf to "float" the heel, or wedge-shaped foam devices. Elevating the heel off the bed surface is critical — pillows alone may shift, re-exposing the heel. Purpose-built devices with straps maintain position during sleep. Do not use ring-shaped ("donut") cushions under the heel, as they concentrate pressure at the ring edge and worsen ischemia.
Bilateral DFUs: Present a significant challenge as the patient requires offloading on both feet simultaneously. Wheelchair use may be necessary for plantar ulcers during the acute healing phase. Bilateral irremovable walkers are generally impractical due to fall risk. A combination of removable walkers (accepted lower healing rate) with intensive patient education, or wheelchair with heel-free positioning, may be required.
Charcot foot with ulceration: The deformity creates abnormal pressure points (bony prominences from midfoot collapse). TCC can accommodate the deformity during the acute phase. Once consolidated, custom-molded shoes with accommodative insoles (total contact inserts) are required for lifelong ulcer prevention.
Compression Therapy for Venous Disease
| Modality | Pressure | Indications | Key Points |
|---|---|---|---|
| Elastic compression stockings | 20-30 or 30-40 mmHg graduated | Prevention of VLU recurrence; mild edema; varicose veins | Must be replaced every 3-6 months (elasticity loss); knee-high preferred; apply in morning before edema accumulates; requires ABI ≥0.8 |
| Multilayer compression bandaging | ~40 mmHg sustained | Active venous leg ulcers; significant edema; limb-shaping | Four-layer system (Profore); provides sustained compression for up to 7 days; applied by trained clinician; accommodates limb shape changes |
| Short-stretch bandaging | High working / low resting pressure | Active VLU; mixed arterial-venous disease (ABI 0.5-0.8); preferred for overnight wear | Provides compression mainly during ambulation (muscle pump); safe lower resting pressure reduces ischemia risk; requires skill to apply; must be reapplied daily |
| Unna boot | Semi-rigid ~30-40 mmHg | Active VLU; ambulatory patients; DFU offloading adjunct | Zinc-oxide impregnated gauze; provides compression and moist healing; changed every 1-2 weeks; inexpensive; cannot adjust once applied |
| Adjustable compression wraps | Patient-adjustable (goal 30-40 mmHg) | Active VLU; patient self-management; fluctuating edema | Velcro straps (CircAid, ReadyWrap); patient can adjust tension; built-in pressure indicators; launderable; promotes independence |
| Intermittent pneumatic compression (IPC) | Variable (30-60 mmHg cyclic) | Refractory VLU; lymphedema; immobile patients; adjunct to bandaging | Mechanical pump with sequential inflation; used 30-60 min 2-3x daily; enhances venous/lymphatic return; expensive; requires patient compliance |
20 Nutrition & Systemic Optimization
Nutritional deficiency is among the most modifiable risk factors for impaired wound healing. Wound healing is an energy-intensive, anabolic process requiring adequate calories, protein, vitamins, minerals, and hydration. Systemic conditions (hyperglycemia, smoking, edema, immunosuppression) must also be addressed for optimal healing.
Macronutrient Requirements
| Nutrient | Recommendation | Rationale |
|---|---|---|
| Protein | 1.25-1.5 g/kg/day (increase to 1.5-2.0 g/kg/day for Stage 3-4 pressure injuries or large wounds) | Amino acids are building blocks for collagen synthesis, immune function, and cell proliferation; protein deficiency leads to decreased fibroblast proliferation, impaired angiogenesis, reduced collagen synthesis, and immune compromise |
| Calories | 30-35 kcal/kg/day | Energy is required for all phases of wound healing; insufficient calories leads to protein catabolism for energy; excess calories (obesity) impairs perfusion and increases infection risk |
| Fluids | 1 mL/kcal/day or 30 mL/kg/day (minimum) | Adequate hydration supports cellular metabolism and nutrient transport; increase for high-output wounds, fever, or draining wounds |
Micronutrient Supplementation
| Nutrient | Dose | Role in Wound Healing |
|---|---|---|
| Vitamin C | 250 mg BID (or 500 mg daily) | Essential cofactor for collagen synthesis (hydroxylation of proline and lysine); antioxidant; enhances neutrophil function; severe deficiency (scurvy) causes wound dehiscence |
| Zinc | 40 mg elemental zinc daily (220 mg zinc sulfate = 50 mg elemental zinc) | Cofactor for over 300 enzymes involved in DNA synthesis, cell division, protein synthesis, and immune function; deficiency impairs all phases of healing; supplement for 2-4 weeks then reassess (prolonged zinc supplementation can cause copper deficiency) |
| Arginine | 4.5-9 g/day (often combined with HMB) | Precursor for nitric oxide (vasodilation, angiogenesis) and collagen synthesis (proline precursor); enhances T-cell function; commonly provided in commercial wound healing supplements (e.g., Juven: arginine + glutamine + HMB) |
| Vitamin A | 25,000 IU daily × 10 days (for steroid-impaired healing) | Reverses the anti-inflammatory and wound-healing impairment caused by corticosteroids; supports epithelialization and collagen synthesis; supplementation specifically indicated for patients on chronic steroids with wounds |
| Iron | Treat if deficient | Required for oxygen transport (hemoglobin) and oxidative metabolism; anemia (Hgb <10 g/dL) impairs wound oxygenation; supplement if iron-deficiency confirmed |
| Glutamine | 10-30 g/day (oral) | Conditionally essential amino acid during metabolic stress; primary fuel source for rapidly dividing cells (lymphocytes, macrophages, fibroblasts, enterocytes); supports immune function and collagen synthesis; depleted rapidly in catabolic states (burns, sepsis, major surgery) |
| Vitamin D | 1,000-2,000 IU daily (or treat documented deficiency) | Modulates immune function, antimicrobial peptide production (cathelicidin), and inflammatory response; deficiency (25-OH vitamin D <30 ng/mL) is prevalent in 60-80% of chronic wound patients; severe deficiency associated with impaired healing and increased infection risk |
| Copper | Treat if deficient (RDA 0.9 mg/day) | Cofactor for lysyl oxidase (collagen and elastin cross-linking), superoxide dismutase (antioxidant defense), and ceruloplasmin (iron metabolism); deficiency may result from prolonged zinc supplementation (>4 weeks of high-dose zinc competitively inhibits copper absorption) |
Juven (Abbott): Contains arginine (7 g), glutamine (7 g), and HMB (1.5 g) per packet; two packets daily. HMB (beta-hydroxy beta-methylbutyrate) is a leucine metabolite that reduces muscle protein breakdown and supports lean body mass preservation. Clinical studies demonstrate improved collagen deposition and healing rates in pressure injuries. Arginaid (Nestlé): Provides 4.5 g arginine per packet. ProSource Plus: Concentrated liquid protein supplement (15 g protein per 30 mL) for patients with fluid restrictions or poor oral intake. Impact Advanced Recovery: Immunonutrition formula with arginine, omega-3 fatty acids, and nucleotides. For patients unable to meet oral requirements, enteral nutrition with a wound-specific formula (e.g., Promote, Replete) should be considered.
Nutritional Screening Markers
Albumin (half-life 20 days): reflects long-term protein status; <3.5 g/dL indicates malnutrition; <2.5 g/dL is severe. However, albumin is a negative acute-phase reactant — it decreases in inflammation, infection, and fluid overload regardless of nutritional status. It is a prognostic marker for outcomes, not a direct treatment target. Prealbumin (transthyretin; half-life 2-3 days): more responsive to nutritional changes; <15 mg/dL suggests malnutrition; <11 mg/dL is severe. Useful for monitoring response to nutritional supplementation (check weekly). Also a negative acute-phase reactant.
Malnutrition Screening Tools
Malnutrition Universal Screening Tool (MUST): A five-step screening tool using BMI, unplanned weight loss, and acute disease effect. Score 0 = low risk, 1 = medium risk, ≥2 = high risk. Mini Nutritional Assessment (MNA): Validated for geriatric populations; includes dietary intake, weight loss, mobility, neuropsychological problems, and BMI. Score <17 = malnourished, 17-23.5 = at risk, ≥24 = well-nourished. Subjective Global Assessment (SGA): Clinician-administered tool combining history (weight change, dietary intake, GI symptoms, functional capacity) with physical examination (subcutaneous fat loss, muscle wasting, ankle/sacral edema). Rates patients as A (well-nourished), B (moderately malnourished or suspected malnutrition), or C (severely malnourished). All patients with chronic wounds should be screened for malnutrition on admission and at regular intervals; those identified as at-risk or malnourished require formal dietitian assessment and individualized nutrition care plans.
Systemic Optimization
Glucose control: Hyperglycemia impairs neutrophil function (chemotaxis, phagocytosis, and oxidative burst are all reduced at blood glucose >200 mg/dL), increases infection risk, glycosylates growth factors and their receptors reducing signaling efficacy, and impairs all phases of wound healing. Target HbA1C <8% for wound healing (some guidelines suggest <7%); avoid hypoglycemia. In the acute care setting, maintain blood glucose 140-180 mg/dL. Perioperative glucose >200 mg/dL doubles the risk of surgical site infection.
Smoking cessation: Nicotine causes vasoconstriction reducing tissue perfusion; carbon monoxide reduces oxygen-carrying capacity (shifts the oxyhemoglobin dissociation curve left); hydrogen cyanide inhibits oxidative metabolism. Smoking reduces wound oxygen tension by 30-40%. Cessation improves healing within 4 weeks. Even reduction in smoking frequency provides measurable benefit. Nicotine replacement therapy (patches, gum) is preferred over continued smoking, though nicotine itself has vasoconstrictive effects.
Edema management: Interstitial edema increases diffusion distance for oxygen and nutrients, compresses capillaries, and impairs cellular metabolism. Treat with compression, elevation, diuretics as indicated, and lymphedema therapy (complete decongestive therapy with manual lymphatic drainage and short-stretch bandaging for lymphedema component).
Medication review: Identify medications that impair healing and minimize or adjust when possible. Corticosteroids suppress inflammation, fibroblast proliferation, collagen synthesis, and immune function — vitamin A 25,000 IU daily for 10 days can partially reverse steroid-impaired healing. Immunosuppressants (methotrexate, mycophenolate, calcineurin inhibitors) impair cell proliferation and immune-mediated wound defense. Anticoagulants increase bleeding risk during debridement; warfarin specifically associated with calciphylaxis risk. Vasopressors reduce peripheral tissue perfusion. Chemotherapy suppresses bone marrow and cell proliferation. NSAIDs may impair the early inflammatory phase of healing, though evidence is mixed.
Obesity (BMI ≥30) independently impairs wound healing through multiple mechanisms: adipose tissue is poorly vascularized (relative hypoxia in subcutaneous fat), increased dead space in surgical wounds promotes seroma and hematoma formation, mechanical stress on wound edges increases dehiscence risk, adipose tissue produces pro-inflammatory adipokines (TNF-α, IL-6, leptin resistance) that perpetuate inflammation, and bariatric patients often have concurrent nutritional deficiencies despite caloric excess (protein, zinc, vitamin D, iron, B12 deficiencies are common after bariatric surgery). Surgical site infection rates increase by approximately 5% for each 5-unit increase in BMI above 25.
21 Wound Care Documentation
Complete and accurate wound care documentation serves clinical, legal, and reimbursement purposes. Every wound encounter must include a systematic assessment, treatment plan, and evidence of clinical decision-making.
Wound Assessment Documentation Template
Location: Anatomic site using standard terminology (e.g., "right medial malleolus," "sacral/coccygeal"). Etiology: Wound type (pressure injury, DFU, venous ulcer, surgical, traumatic). Duration: Time since wound onset; previous treatments attempted. Size: Length × width × depth in cm (clock-face method). Wound bed: Percentage of tissue types (granulation, slough, eschar, epithelial). Exudate: Type (serous, sanguineous, serosanguineous, purulent) and amount (none/scant/small/moderate/large). Wound edges: Attached, rolled (epibole), undermining (location and depth), tunneling (direction and depth). Periwound skin: Intact, macerated, erythematous, indurated, calloused, excoriated. Pain: Numeric rating scale, character, temporal pattern, and relation to dressing changes. Staging: Pressure injury stage, Wagner/UT grade, or other classification. Treatment plan: Debridement performed, dressing selected with rationale, offloading/compression, frequency of dressing changes, referrals.
Progress Tracking
Wound area reduction is the most important metric for predicting healing trajectory. Evidence demonstrates that wounds that achieve 40-50% area reduction at 4 weeks are highly likely to heal by 12 weeks, while those that do not are unlikely to heal with current therapy and require treatment reassessment. This "4-week rule" guides clinical decision-making about when to escalate therapy (advanced wound products, surgical intervention, HBOT, referral).
Additional metrics tracked longitudinally include: wound depth (decreasing = healing), exudate volume (decreasing = healing), wound bed tissue type progression (necrotic → slough → granulation → epithelial), periwound condition improvement, and pain trajectory.
CPT Coding for Wound Care
| CPT Code | Description | Key Requirements |
|---|---|---|
| 97597 | Debridement, open wound, selective; first 20 cm2 | Removal of devitalized tissue using sharp instruments (curette, scissors, scalpel) at the wound surface; without anesthesia; includes topical application and wound assessment |
| 97598 | Debridement, open wound, selective; each additional 20 cm2 | Add-on code to 97597; used for additional wound area or additional wounds debrided in the same session |
| 97602 | Removal of devitalized tissue, non-selective | Wet-to-dry, enzymatic, autolytic debridement; lower reimbursement; often bundled and not separately reportable |
| 97605 | NPWT (surface area ≤50 cm2) | Application of negative pressure wound therapy including wound assessment and instructions |
| 97606 | NPWT (surface area >50 cm2) | Same as 97605 for larger wound surface area |
Additional CPT Codes for Wound Care
| CPT Code | Description | Key Requirements |
|---|---|---|
| 11042 | Debridement, subcutaneous tissue; first 20 cm2 | Excisional debridement through dermis into subcutaneous tissue; performed by physician; higher reimbursement than 97597 |
| 11043 | Debridement, muscle and/or fascia; first 20 cm2 | Deep excisional debridement into muscle or fascia; typically performed in OR or procedural setting |
| 11044 | Debridement, bone; first 20 cm2 | Debridement extending to bone; often associated with osteomyelitis management |
| 15271-15278 | Skin substitute graft application | Codes vary by wound type (DFU/VLU vs other), location, and size; first 25 cm2 and each additional 25 cm2; requires documentation of wound type, standard care failure, and clean granulating bed |
| 99183 | Physician supervision of HBOT per session | Physician must be present and available during treatment; document indication, treatment pressure, duration, patient tolerance, and wound response |
Medical Necessity Documentation
For advanced wound care products and HBOT, payers require documentation of: wound duration (>30 days), failure of standard care (debridement, moisture management, offloading/compression, infection treatment, nutritional optimization), absence of adequate vascular supply exclusion (ABI/TcPO2), wound measurements showing lack of progress (<40% area reduction at 4 weeks), absence of untreated infection, and patient adherence to treatment plan. Failure to document these elements results in claim denial.
ICD-10 Coding for Wound Care
Accurate ICD-10 coding captures wound etiology, laterality, and specificity. Key code families include: L89.xxx (pressure ulcers by site and stage), L97.xxx (non-pressure chronic ulcer of lower extremity by site, laterality, severity, and tissue involvement), E11.621 (type 2 diabetes with foot ulcer), I87.0xx (postthrombotic syndrome with ulcer), I83.0xx (varicose veins with ulcer), and T81.31xA (disruption of wound, initial encounter). Code to the highest specificity available: laterality (right/left), severity (limited to breakdown of skin, with fat layer exposed, with necrosis of muscle, with necrosis of bone), and chronicity (initial encounter, subsequent encounter, sequela). Use combination codes when available (e.g., E11.621 + L97 for diabetic foot ulcer site specificity).
22 Quality Metrics & Prevention Programs
Wound care quality is measured through standardized metrics, regulatory requirements, and evidence-based prevention programs. Hospitals and long-term care facilities face financial penalties for hospital-acquired pressure injuries (HAPI) under CMS value-based purchasing programs. A comprehensive wound care quality program integrates prevention, evidence-based treatment, outcome tracking, and continuous improvement processes.
Present on Admission (POA) Documentation
CMS requires hospitals to report whether diagnoses were present on admission (POA). Stage 3, Stage 4, and unstageable pressure injuries classified as hospital-acquired (not POA) are designated as hospital-acquired conditions (HACs) and are no longer eligible for higher DRG payment — the hospital absorbs the additional cost of treatment. Accurate POA documentation requires: complete skin assessment within hours of admission (ideally during the emergency department visit or within the first nursing assessment), photographic documentation of any existing wounds, staging and measurement of all identified pressure injuries, and clear documentation that the wound was present before hospitalization. If a comprehensive skin assessment is not completed within the admission window, any subsequently discovered pressure injury may default to hospital-acquired status.
CMS Quality Measures
NQF #0201 (PSI 03): Pressure ulcer rate (hospital-acquired Stage 3, 4, or unstageable pressure injuries per 1000 discharges). Publicly reported and linked to hospital reimbursement. NQF #0678: Percent of residents with new or worsened pressure ulcers in long-term care (MDS-derived). HAC Reduction Program: Hospitals in the worst-performing quartile for hospital-acquired conditions (including Stage 3/4 pressure injuries) receive a 1% payment reduction on all Medicare discharges.
Wound Care Certification
| Credential | Certifying Body | Eligibility |
|---|---|---|
| WCC (Wound Care Certified) | National Alliance of Wound Care and Ostomy (NAWCO) | RN, LPN, PT, OT, MD, DPM, PA, NP with wound care experience |
| CWCN (Certified Wound Care Nurse) | Wound, Ostomy and Continence Nursing Certification Board (WOCNCB) | RN with BSN or higher; completion of accredited WOC nursing education program |
| CWSP (Certified Wound Specialist Physician) | American Board of Wound Management (ABWM) | MD or DO with wound care practice experience; examination-based |
| CWS (Certified Wound Specialist) | American Board of Wound Management (ABWM) | Healthcare professionals (non-physician); examination-based |
Skin Care Bundles for Prevention
Evidence-based prevention bundles reduce HAPI rates by 50-80% when implemented consistently. Components include: structured risk assessment on admission and daily (Braden Scale); skin inspection on admission and every shift; moisture management and incontinence care; pressure redistribution (support surfaces, repositioning schedule); nutritional assessment and supplementation; patient/family education; documentation compliance monitoring; and quality improvement data tracking with unit-level feedback.
SSKIN Bundle
A widely adopted pressure injury prevention framework:
| Component | Actions |
|---|---|
| S — Surface | Select appropriate support surface based on risk assessment; ensure proper inflation and function of alternating pressure devices; use heel elevation devices |
| S — Skin inspection | Complete skin assessment on admission, every shift, and with each repositioning; pay attention to bony prominences and under medical devices; document findings |
| K — Keep moving | Reposition every 2 hours in bed, every 1 hour in chair; use 30-degree lateral tilt; encourage mobility; use repositioning schedule and documentation tool |
| I — Incontinence | Implement structured continence care; cleanse perianal skin with pH-balanced cleanser; apply barrier cream; consider fecal management system for uncontrolled diarrhea |
| N — Nutrition | Nutritional screening on admission; dietitian referral for at-risk patients; provide oral nutritional supplements; monitor protein and caloric intake; supplement micronutrients |
Wound Care Team Structure
Comprehensive wound care programs utilize a multidisciplinary team approach: wound care nurse/CWOCN (direct wound assessment, treatment planning, product selection, education); physician/CWSP (medical management, debridement, HBOT supervision, surgical referral); podiatrist/DPM (DFU management, offloading, nail and callus care, Charcot management); vascular surgeon (revascularization assessment, ABI interpretation, compression clearance); physical/occupational therapy (mobility, offloading, edema management, functional optimization); registered dietitian (nutritional assessment, caloric and protein targets, supplement recommendations); social worker (insurance navigation, durable medical equipment procurement, adherence barriers, home care coordination); and orthotic/prosthetic specialist (custom footwear, offloading devices, prosthetics after amputation).
WOCN Society Guidelines
The Wound, Ostomy and Continence Nurses (WOCN) Society publishes evidence-based guidelines for pressure injury prevention and treatment, venous ulcer management, diabetic foot ulcer management, and ostomy care. These guidelines are updated periodically and serve as the standard of care in wound management. Key recommendations are graded by level of evidence (A = strong evidence, B = moderate evidence, C = expert opinion). The WOCN guidelines align with and complement international guidelines from EPUAP/NPIAP (pressure injuries), IWGDF (diabetic foot), and the Society for Vascular Surgery (venous disease), providing a comprehensive evidence framework for clinical practice and institutional policy development.
Key Performance Indicators (KPIs) for Wound Care Programs
Effective wound care programs track measurable outcomes including: HAPI incidence rate (new hospital-acquired pressure injuries per 1,000 patient-days), HAPI prevalence rate (point-prevalence survey data), average time to wound closure by wound type, percentage of wounds achieving ≥40% area reduction at 4 weeks, surgical site infection rates by procedure, debridement rates (percentage of wounds with necrotic tissue that receive timely debridement), compression therapy compliance rates for VLU patients, offloading compliance rates for DFU patients, amputation rates for diabetic foot patients, and patient-reported outcome measures (pain scores, quality of life). Benchmarking against national databases (NDNQI for pressure injuries, NSQIP for SSI) enables comparison with peer institutions.
Telehealth in Wound Care
Remote wound monitoring and telehealth consultations have expanded access to wound care expertise, particularly in rural and underserved settings. Store-and-forward (asynchronous) wound photography allows wound specialists to review images and provide treatment recommendations without synchronous visits. Real-time video consultations enable specialist guidance for bedside clinicians performing assessments and dressing changes. Requirements for effective telehealth wound care include: standardized photography protocols (consistent distance, lighting, angle, ruler in frame), calibrated digital measurement tools, structured assessment templates, and secure HIPAA-compliant platforms. Studies demonstrate comparable healing outcomes for telehealth-managed wounds versus in-person care, with improved access, reduced travel burden, and earlier specialist involvement.
Legal Considerations
Pressure injuries are the second most common claim in nursing malpractice litigation. Key documentation requirements for legal protection: evidence of risk assessment on admission, implementation of prevention interventions appropriate to risk level, regular reassessment and documentation of skin status, evidence that changes in wound status prompted treatment modifications, informed consent for debridement and advanced therapies, and documentation of patient/family education and adherence barriers. Facilities should maintain policies defining accountability for skin assessments, documentation timelines, escalation procedures when new pressure injuries are identified, root cause analysis processes, and staff competency validation in wound prevention and care.
23 Staging Systems Master Table
| System | Stage/Grade | Description |
|---|---|---|
| NPUAP Pressure Injury | Stage 1 | Non-blanchable erythema of intact skin |
| Stage 2 | Partial-thickness skin loss with exposed dermis; viable wound bed | |
| Stage 3 | Full-thickness skin loss; fat visible; no bone/tendon/muscle exposed | |
| Stage 4 | Full-thickness skin and tissue loss; bone/tendon/muscle visible | |
| Unstageable | Full-thickness loss obscured by slough/eschar | |
| DTPI | Persistent non-blanchable deep red/maroon/purple discoloration | |
| Wagner DFU | Grade 0 | Intact skin; at-risk foot with deformity |
| Grade 1 | Superficial ulcer (skin only) | |
| Grade 2 | Deep ulcer to tendon/joint/bone without abscess/osteomyelitis | |
| Grade 3 | Deep ulcer with abscess or osteomyelitis | |
| Grade 4 | Localized gangrene (forefoot/heel) | |
| Grade 5 | Extensive gangrene of entire foot | |
| UT DFU | Grade 0 | Pre- or post-ulcerative, epithelialized |
| Grade 1 | Superficial wound (not to tendon/capsule/bone) | |
| Grade 2 | Wound to tendon or capsule | |
| Grade 3 | Wound to bone or joint | |
| CEAP Venous | C0 | No visible/palpable venous disease |
| C1 | Telangiectasias or reticular veins | |
| C2 | Varicose veins | |
| C3 | Edema | |
| C4a | Pigmentation or eczema | |
| C4b | Lipodermatosclerosis or atrophie blanche | |
| C5 | Healed venous ulcer | |
| C6 | Active venous ulcer | |
| ISTAP Skin Tear | Type 1 | No skin loss; flap repositionable |
| Type 2 | Partial flap loss | |
| Type 3 | Total flap loss | |
| Cierny-Mader Osteomyelitis | Type I | Medullary osteomyelitis |
| Type II | Superficial osteomyelitis | |
| Type III | Localized osteomyelitis (stable bone after debridement) | |
| Type IV | Diffuse osteomyelitis (unstable bone, requires reconstruction) | |
| Burn Depth | Superficial | Epidermis only; red, dry; painful; heals 3-5 days |
| Superficial partial-thickness | Epidermis + papillary dermis; pink, moist, blisters; very painful; heals 7-14 days | |
| Deep partial-thickness | Epidermis + reticular dermis; pale/white, mottled; decreased sensation; heals 14-35 days with scarring | |
| Full-thickness | All skin layers destroyed; white/leathery/charred; insensate; requires grafting |
24 Dressing Selection Guide
This quick-reference table matches wound characteristics to the most appropriate dressing category with clinical rationale.
| Wound Characteristic | Recommended Dressing | Rationale |
|---|---|---|
| Dry wound, minimal exudate | Hydrogel (amorphous or sheet) | Donates moisture to maintain moist healing environment; promotes autolytic debridement of slough |
| Light exudate, granulating | Hydrocolloid or thin foam | Maintains moist environment; absorbs light exudate; protects granulation tissue; infrequent changes |
| Moderate exudate | Foam dressing | Excellent absorption with moisture vapor transmission; cushioning; available in adhesive and non-adhesive forms |
| Heavy exudate | Alginate or hydrofiber (primary) + foam (secondary) | High absorption capacity; gel formation maintains moisture balance; alginate is hemostatic |
| Cavity/tunneling wound | Alginate rope or hydrofiber ribbon | Conforms to irregular wound shape; gel formation fills dead space; easily removed in one piece |
| Slough-covered (non-infected) | Hydrogel + film or hydrocolloid | Moisture promotes autolytic debridement; softens slough for removal at next visit |
| Eschar-covered (non-infected, non-ischemic) | Enzymatic debrider (collagenase) + moist dressing; or hydrogel under film | Cross-hatch eschar, apply collagenase to promote enzymatic debridement; alternatively, hydrogel autolysis |
| Critically colonized/locally infected | Silver, honey, PHMB, or cadexomer iodine dressing | Topical antimicrobial reduces bioburden; reassess after 2 weeks; combine with debridement |
| Fragile periwound skin | Silicone-bordered foam or silicone contact layer | Silicone adhesive minimizes trauma on removal; prevents further skin damage |
| Over skin graft | Non-adherent contact layer (petrolatum gauze or silicone mesh) + bolster | Protects graft without disrupting attachment; allows exudate drainage |
| Superficial partial-thickness burn | Silver-impregnated foam/dressing or medical-grade honey | Antimicrobial protection; moist healing; reduced dressing change frequency vs SSD cream |
| Skin tear (flap present) | Silicone contact layer + foam or film | Holds flap in place without adhesive trauma; non-adherent; allows wound visualization |
| Under compression bandage | Foam (non-adhesive) or hydrofiber | Thin profile; absorbs exudate; does not interfere with compression gradient; non-adhesive to avoid skin trauma |
| Malodorous wound | Charcoal dressing or cadexomer iodine or medical-grade honey | Activated charcoal absorbs volatile odor compounds; iodine and honey have deodorizing properties |
25 ABI Interpretation Table
| ABI Value | Interpretation | Clinical Significance |
|---|---|---|
| >1.4 | Non-compressible (calcified vessels) | Common in diabetes, CKD, elderly; ABI unreliable; use toe pressures/TBI or TcPO2 instead; do NOT assume normal perfusion |
| 1.0-1.4 | Normal | Adequate arterial perfusion; full compression therapy appropriate; wound healing potential not limited by perfusion |
| 0.9-1.0 | Borderline/acceptable | May have early PAD; full compression generally safe; exercise ABI testing if symptomatic |
| 0.8-0.9 | Mild PAD | Full compression (30-40 mmHg) generally appropriate with monitoring; asymptomatic or mild claudication |
| 0.5-0.8 | Moderate PAD | Modified compression only (23-30 mmHg; inelastic/short-stretch preferred); claudication likely; vascular referral; wound healing may be compromised |
| <0.5 | Severe PAD | Compression contraindicated; rest pain/tissue loss likely; urgent vascular referral for revascularization assessment; wound healing unlikely without revascularization |
| <0.4 | Critical limb ischemia | Limb-threatening; emergent vascular consultation; revascularization required for limb salvage; high amputation risk |
26 TIME Framework Quick Reference
| Component | Clinical Question | Red Flags | Actions |
|---|---|---|---|
| T — Tissue | Is there non-viable tissue in the wound bed? | Slough >50%, eschar, necrotic tissue, non-granulating wound bed | Sharp debridement (preferred), enzymatic (collagenase), autolytic (hydrogel), biological (maggots); maintenance debridement at each visit |
| I — Infection/Inflammation | Is bacterial burden or persistent inflammation impairing healing? | NERDS criteria (superficial), STONEES criteria (deep), stalled wound, increased exudate, new slough in clean wound, malodor, biofilm | Debridement + topical antimicrobial (silver, honey, PHMB, cadexomer iodine); systemic antibiotics for spreading/deep infection; 2-week antimicrobial reassessment |
| M — Moisture | Is the wound too dry or too wet? | Desiccated wound bed (too dry); periwound maceration (too wet); excessive dressing saturation; dry adherent dressings | Too dry: hydrogel, moisture-donating dressings. Too wet: alginate, hydrofiber, foam, superabsorbent; barrier cream on periwound; adjust dressing change frequency |
| E — Edge | Is the wound edge advancing? | Non-migrating epithelium, rolled/epibolic edges, hyperkeratotic margins, wound stalled >4 weeks with <40% area reduction | Debride wound edges; treat epibole (silver nitrate, sharp debridement); reassess causative factors; escalate to advanced therapy (skin substitutes, growth factors, NPWT, HBOT) |
Apply the TIME framework at every wound assessment. Address each component sequentially: tissue management first (clean wound bed), then infection control (reduce bioburden), then moisture balance (appropriate dressing), then edge assessment (evaluate healing trajectory). If the wound edge is not advancing despite optimizing T, I, and M, systemic factors (nutrition, perfusion, glycemic control, medications) must be re-evaluated before escalating to advanced therapies.
27 Common Wound Care Products by Category
| Category | Brand Names (Examples) | Key Features |
|---|---|---|
| Transparent Film | Tegaderm, OpSite, Bioclusive, Mepore Film | Moisture-retentive, waterproof, allows wound visualization |
| Hydrocolloid | DuoDERM, Comfeel, Tegasorb, Replicare | Adhesive wafer, autolytic debridement, low-moderate absorption |
| Foam | Mepilex, Allevyn, Biatain, PolyMem, Optifoam | Moderate-high absorption, cushioning, various border options |
| Alginate | Kaltostat, Maxorb, AlgiSite, Sorbalgon, Melgisorb | High absorption, hemostatic, gel-forming, rope and sheet forms |
| Hydrofiber | Aquacel, Aquacel Extra, Aquacel Ag | Vertical absorption, cohesive gel, minimal lateral wicking |
| Hydrogel (amorphous) | IntraSite Gel, SAF-Gel, Woun'Dres, Derma-Gel | Moisture donation, cooling, autolytic debridement |
| Hydrogel (sheet) | Vigilon, Elasto-Gel, ClearSite | Cooling, moisture donation, transparent, for flat superficial wounds |
| Silver (ionic) | Aquacel Ag, Mepilex Ag, Biatain Ag, Silvercel | Antimicrobial, various substrates (foam, hydrofiber, alginate) |
| Silver (nanocrystalline) | Acticoat, Acticoat 7 | Sustained silver release, moisten with sterile water only (not saline) |
| Honey | Medihoney, TheraHoney, MelMax, Activon | Medical-grade Manuka honey, antimicrobial, debriding, deodorizing |
| Cadexomer Iodine | Iodosorb, Iodoflex | Sustained iodine release, absorbs exudate, debriding, anti-biofilm |
| PHMB | Prontosan (irrigation/gel), Kerlix AMD, Kendall AMD | Low cytotoxicity, broad-spectrum, biofilm disruption with surfactant |
| Collagen | Promogran, Puracol Plus, Endoform, Biopad | MMP modulation, scaffold for healing, various sources (bovine, porcine, equine, ovine) |
| Enzymatic Debrider | Santyl (collagenase) | Only FDA-approved enzymatic debrider; apply daily to moist wound; inactivated by silver and detergents |
| Skin Substitute (cellular) | Apligraf, Dermagraft, Grafix, TheraSkin | Living cells secreting growth factors; requires clean granulating wound bed; strict storage requirements |
| Skin Substitute (acellular) | Integra, Oasis, PriMatrix, EpiFix, AmnioExcel | Scaffold/matrix; no living cells; longer shelf life; various sources (human, porcine, bovine) |
| NPWT Systems | V.A.C. (KCI/3M), Prevena, PICO, SNaP, extriCARE | Sub-atmospheric pressure; foam or gauze filler; portable and hospital systems |
| Compression Systems | Profore, Coban 2, CircAid, Unna boot, Jobst stockings | Multilayer bandage, adjustable wrap, semi-rigid, and stocking options |
| Contact Layer | Mepitel, Adaptic Touch, Profore WCL, Silflex | Non-adherent, protects wound bed, allows exudate transfer to secondary dressing |
| Superabsorbent | DryMax, Vliwasorb, Zetuvit Plus, Flivasorb | For extremely high exudate wounds; locks fluid in polymer core; reduces dressing change frequency |
28 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ABI | Ankle-brachial index |
| ATA | Atmospheres absolute |
| BID | Twice daily |
| BMZ | Basement membrane zone |
| BWAT | Bates-Jensen Wound Assessment Tool |
| CAM | Controlled ankle movement (boot) |
| CDC | Centers for Disease Control and Prevention |
| CEAP | Clinical-Etiology-Anatomy-Pathophysiology (venous classification) |
| CFU | Colony-forming units |
| CKD | Chronic kidney disease |
| CMC | Carboxymethylcellulose |
| CMS | Centers for Medicare and Medicaid Services |
| CPT | Current Procedural Terminology |
| CRP | C-reactive protein |
| CWS | Certified Wound Specialist |
| CWCN | Certified Wound Care Nurse |
| CWSP | Certified Wound Specialist Physician |
| dHACM | Dehydrated human amnion/chorion membrane |
| DFU | Diabetic foot ulcer |
| DTPI | Deep tissue pressure injury |
| ECM | Extracellular matrix |
| EGF | Epidermal growth factor |
| EPUAP | European Pressure Ulcer Advisory Panel |
| ESR | Erythrocyte sedimentation rate |
| ESRD | End-stage renal disease |
| FGF | Fibroblast growth factor |
| FTSG | Full-thickness skin graft |
| HAC | Hospital-acquired condition |
| HAPI | Hospital-acquired pressure injury |
| HBOT | Hyperbaric oxygen therapy |
| HMB | Beta-hydroxy beta-methylbutyrate |
| IGF | Insulin-like growth factor |
| iNPWT | Incisional negative pressure wound therapy |
| IPC | Intermittent pneumatic compression |
| ISTAP | International Skin Tear Advisory Panel |
| IV | Intravenous |
| KGF | Keratinocyte growth factor |
| MARSI | Medical adhesive-related skin injury |
| MASD | Moisture-associated skin damage |
| MDS | Minimum Data Set |
| MGO | Methylglyoxal |
| MMP | Matrix metalloproteinase |
| MNSI | Michigan Neuropathy Screening Instrument |
| MRI | Magnetic resonance imaging |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| NAWCO | National Alliance of Wound Care and Ostomy |
| NNIS | National Nosocomial Infections Surveillance |
| NPUAP | National Pressure Ulcer Advisory Panel (now NPIAP) |
| NPIAP | National Pressure Injury Advisory Panel |
| NPWT | Negative pressure wound therapy |
| NPWTi-d | NPWT with instillation and dwell time |
| NQF | National Quality Forum |
| OR | Operating room |
| PAD | Peripheral arterial disease |
| PDGF | Platelet-derived growth factor |
| PG | Pyoderma gangrenosum |
| PHMB | Polyhexamethylene biguanide |
| PPV | Positive predictive value |
| PTB | Probe-to-bone |
| PU | Polyurethane |
| PUSH | Pressure Ulcer Scale for Healing |
| PVA | Polyvinyl alcohol |
| rhPDGF | Recombinant human platelet-derived growth factor |
| ROS | Reactive oxygen species |
| SIS | Small intestinal submucosa |
| SSD | Silver sulfadiazine |
| SSI | Surgical site infection |
| STSG | Split-thickness skin graft |
| TBI | Toe-brachial index |
| TBSA | Total body surface area |
| TCC | Total contact cast |
| TcPO2 | Transcutaneous oxygen pressure |
| TGF-β | Transforming growth factor-beta |
| TIMP | Tissue inhibitor of metalloproteinase |
| TIME | Tissue-Infection/Inflammation-Moisture-Edge |
| UMF | Unique Manuka Factor |
| UHMS | Undersea and Hyperbaric Medical Society |
| UT | University of Texas (classification) |
| VAC | Vacuum-assisted closure |
| VEGF | Vascular endothelial growth factor |
| VLU | Venous leg ulcer |
| VPT | Vibration perception threshold |
| WBC | White blood cell |
| WCC | Wound Care Certified |
| WOCN | Wound, Ostomy and Continence Nurses (Society) |
| WOCNCB | Wound, Ostomy and Continence Nursing Certification Board |
| XR | X-ray (plain radiograph) |