Anesthesiology
Pre-operative evaluation, airway management, general and regional anesthesia, pharmacology of anesthetic agents, hemodynamic monitoring, pain management, and every classification system, drug dosing protocol, and management algorithm across the full scope of anesthesiology.
01 Airway Anatomy & Physiology
Upper Airway Anatomy
The upper airway extends from the nares and oral cavity to the laryngeal inlet. The nasal cavity is lined with highly vascular mucosa (Kiesselbach plexus on the anterior septum is the most common site of epistaxis) and warms, humidifies, and filters inspired gas. Three turbinates (inferior, middle, superior) increase mucosal surface area. The oral cavity serves as the primary route for airway instrumentation; the tongue is the most common cause of upper airway obstruction in the unconscious patient because loss of genioglossus tone allows it to fall posteriorly against the soft palate and posterior pharyngeal wall. The pharynx is divided into the nasopharynx (posterior to nasal cavity, contains adenoids), oropharynx (posterior to oral cavity, from soft palate to epiglottis tip), and hypopharynx/laryngopharynx (epiglottis tip to cricoid cartilage, continuous with the esophageal inlet posteriorly).
Laryngeal Anatomy
The larynx extends from C3 to C6 and comprises a cartilaginous skeleton, intrinsic and extrinsic muscles, and mucosal lining. The epiglottis is a leaf-shaped elastic cartilage attached to the inner surface of the thyroid cartilage; during swallowing it folds posteriorly to cover the glottis, preventing aspiration. The thyroid cartilage (the largest, forming the laryngeal prominence or "Adam's apple") shields the vocal folds anteriorly. The cricoid cartilage is the only complete cartilaginous ring in the airway and defines the narrowest point of the adult airway at the subglottic level (in children <8 years, the cricoid ring — not the glottis — is the narrowest point). The cricothyroid membrane connects the thyroid and cricoid cartilages anteriorly and is the target for emergency surgical airway access (cricothyrotomy); it is approximately 9 mm high and 30 mm wide and lies 2–3 cm below the laryngeal prominence.
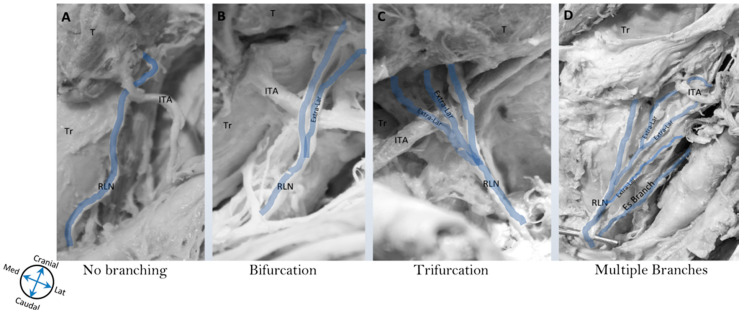
The vocal cords (true vocal folds) are paired mucosal folds stretched between the arytenoid cartilages posteriorly and the thyroid cartilage anteriorly. They are innervated by the recurrent laryngeal nerve (RLN) — a branch of the vagus — which controls all intrinsic laryngeal muscles except the cricothyroid (innervated by the external branch of the superior laryngeal nerve, which tenses the cords). Unilateral RLN injury causes ipsilateral vocal cord paralysis (cord fixed in paramedian position, hoarseness); bilateral RLN injury causes bilateral cord adduction and stridor — a potential airway emergency. The superior laryngeal nerve (SLN) has an internal branch providing sensation above the vocal cords and an external branch providing motor innervation to the cricothyroid muscle.
Trachea & Bronchial Anatomy
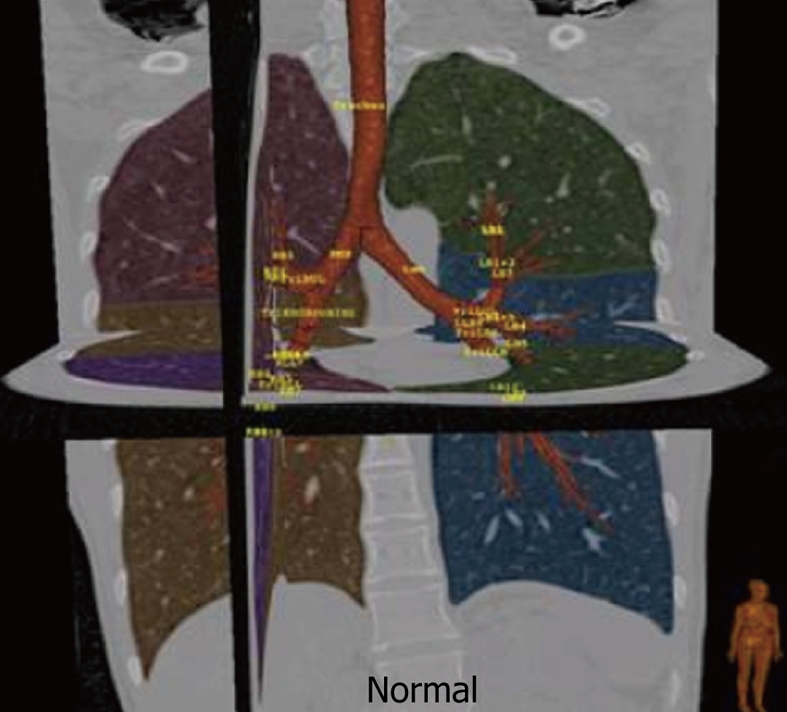
The trachea is approximately 10–12 cm long in adults, extending from the cricoid cartilage (C6) to the carina (T4–T5), supported by 16–20 C-shaped cartilaginous rings open posteriorly (the posterior membranous wall abuts the esophagus). The tracheal internal diameter averages 20 mm in men, 18 mm in women. At the carina, the trachea bifurcates into the right and left mainstem bronchi. The right mainstem bronchus is wider, shorter (~2.5 cm), and more vertically oriented (25° from midline) than the left (~5 cm, 45°) — this explains why aspiration, endobronchial intubation, and foreign body impaction occur preferentially on the right. The right upper lobe bronchus takeoff is only ~2.5 cm from the carina, which can be obstructed by an endobronchial tube or a deeply placed endotracheal tube.
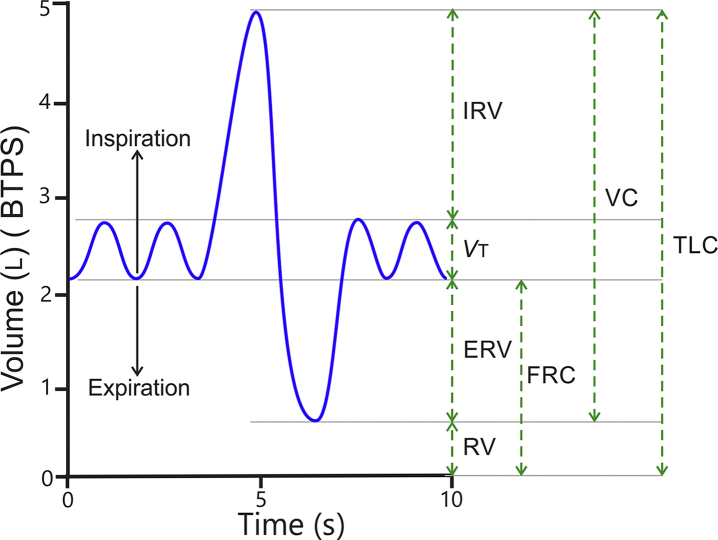
Lung Volumes & Capacities
Understanding lung volumes is essential for ventilator management, preoxygenation efficacy, and predicting desaturation time during apnea.
| Parameter | Abbreviation | Adult Normal (~70 kg) | Clinical Relevance |
|---|---|---|---|
| Tidal volume | TV (VT) | 500 mL (6–8 mL/kg IBW) | Normal breathing; lung-protective ventilation targets 6–8 mL/kg IBW |
| Inspiratory reserve volume | IRV | 3000 mL | Additional volume above TV on maximal inspiration |
| Expiratory reserve volume | ERV | 1100 mL | Additional volume exhaled beyond TV; decreased in obesity |
| Residual volume | RV | 1200 mL | Volume remaining after maximal exhalation; cannot be measured by spirometry |
| Functional residual capacity | FRC = ERV + RV | 2300 mL | Oxygen reservoir during apnea; decreased in obesity, pregnancy, supine position |
| Vital capacity | VC = TV + IRV + ERV | 4600 mL | Maximum volume exhaled after maximum inspiration |
| Total lung capacity | TLC = VC + RV | 5800 mL | Total volume at maximum inspiration |
| Closing capacity | CC | Increases with age | When CC > FRC, small airway closure occurs during tidal breathing → shunt |
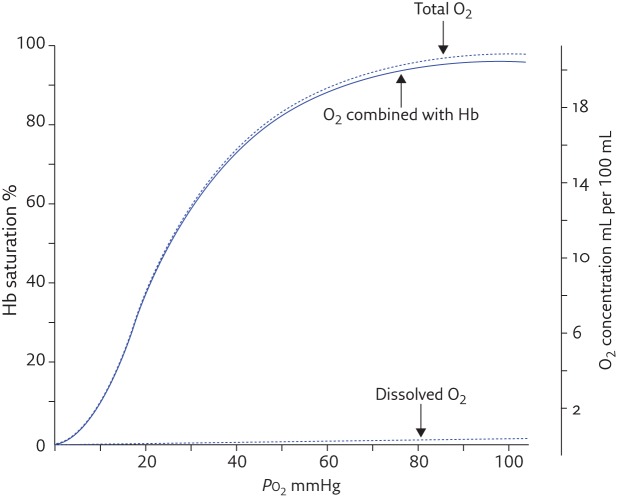
Oxygen-Hemoglobin Dissociation Curve
The oxygen-hemoglobin dissociation curve plots hemoglobin saturation (SpO₂) against partial pressure of oxygen (PaO₂). Its sigmoid shape reflects cooperative binding: as each O₂ molecule binds, affinity for the next increases. At a PaO₂ of 60 mmHg, saturation is approximately 90% — below this, small decreases in PaO₂ produce precipitous desaturation. The P50 is the PaO₂ at which hemoglobin is 50% saturated; normal P50 is 26.7 mmHg.
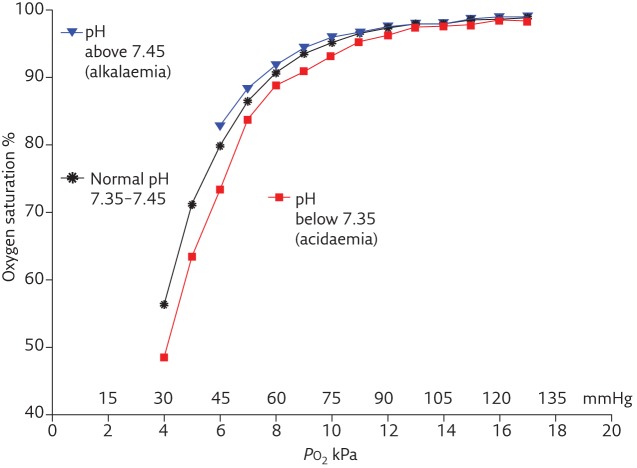
Right shift (increased P50, decreased affinity, favors O₂ unloading to tissues): acidosis (Bohr effect), hyperthermia, increased 2,3-DPG (chronic hypoxia, anemia), hypercarbia. Mnemonic: "Right = Release." Left shift (decreased P50, increased affinity, impaired O₂ release): alkalosis, hypothermia, decreased 2,3-DPG (stored blood), hypocarbia, carbon monoxide (CO) poisoning, fetal hemoglobin (HbF), methemoglobinemia. Left shift: saturation looks adequate but tissues are hypoxic because O₂ is not released.
Ventilation-Perfusion Matching & Shunt
Gas exchange efficiency depends on matching of ventilation (V) and perfusion (Q). The ideal V/Q ratio is 1.0. Shunt (V/Q = 0) occurs when blood passes through non-ventilated alveoli (atelectasis, pneumonia, pulmonary edema, endobronchial intubation) — cannot be corrected by increasing FiO₂ alone (the hallmark of true shunt). Dead space (V/Q = ∞) occurs when ventilated alveoli have no perfusion (PE, low CO states). Hypoxic pulmonary vasoconstriction (HPV) is a protective reflex that diverts blood away from poorly ventilated lung regions to better-ventilated areas, optimizing V/Q matching. All volatile anesthetics inhibit HPV in a dose-dependent manner (significant at >1 MAC), worsening V/Q mismatch and increasing shunt — this is one reason arterial oxygenation may be worse under general anesthesia. The lateral decubitus position creates V/Q mismatch because the dependent lung receives more perfusion (gravity) but less ventilation (compression); one-lung ventilation amplifies this significantly.
Dead Space
Anatomical dead space (~150 mL in adults, approximately 2 mL/kg) is the volume of the conducting airways (nose to terminal bronchioles) that does not participate in gas exchange. Physiological dead space = anatomical dead space + alveolar dead space (alveoli ventilated but not perfused). In healthy individuals, physiological ≈ anatomical dead space. Increased dead space (pulmonary embolism, hypovolemia, high PEEP, ARDS) widens the PaCO₂–ETCO₂ gradient and increases minute ventilation requirements. Dead space is quantified by the Bohr equation: VD/VT = (PaCO₂ − PECO₂) / PaCO₂; normal VD/VT ≈ 0.3.
02 Cardiovascular Physiology for Anesthesia
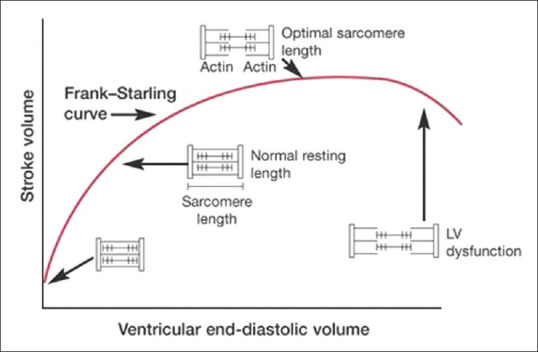
Frank-Starling Mechanism
The Frank-Starling law states that the force of ventricular contraction increases with increasing preload (end-diastolic volume) up to a physiologic limit, because stretching sarcomeres to optimal length improves actin-myosin overlap. On the Frank-Starling curve, the x-axis is preload (LVEDV or LVEDP) and the y-axis is stroke volume or cardiac output. On the ascending limb, volume administration increases output — the patient is "fluid responsive." On the flat portion, additional volume provides no hemodynamic benefit and risks pulmonary edema. Heart failure shifts the entire curve downward and rightward; inotropes shift it upward.
Cardiac Output & Its Determinants
Cardiac output (CO) = heart rate (HR) × stroke volume (SV). Normal resting CO is approximately 5 L/min. Stroke volume is determined by three factors: preload (ventricular end-diastolic volume, reflecting venous return), afterload (resistance the ventricle must overcome to eject — approximated by SVR for the LV), and contractility (intrinsic myocardial force independent of loading conditions). Cardiac index (CI) = CO / BSA; normal 2.5–4.0 L/min/m².
Mean Arterial Pressure
MAP = CO × SVR, or clinically estimated as (SBP + 2 × DBP) / 3. MAP must be maintained ≥65 mmHg to ensure adequate organ perfusion. Under anesthesia, MAP commonly falls due to decreased SVR (vasodilation from volatile agents and propofol), reduced CO (myocardial depression, decreased venous return), and loss of sympathetic tone. MAP <55 mmHg is associated with acute kidney injury and myocardial ischemia.
Myocardial Oxygen Supply & Demand
Myocardial oxygen supply depends on coronary blood flow and arterial oxygen content (CaO₂). The left ventricle is perfused primarily during diastole (unlike most organs, which are perfused during systole) because intramyocardial compressive forces during systole occlude subendocardial vessels. Coronary perfusion pressure (CPP) = aortic diastolic pressure (DBP) − left ventricular end-diastolic pressure (LVEDP). Tachycardia shortens diastole and reduces coronary perfusion time while simultaneously increasing demand. Myocardial oxygen demand is determined by: heart rate (single most important), wall tension (by Laplace: proportional to pressure × radius / wall thickness), and contractility. The anesthesiologist's goal in coronary patients is to maintain adequate CPP while minimizing demand: avoid tachycardia, maintain diastolic pressure, and prevent excessive increases in contractility or afterload.
Baroreceptor Reflex
The baroreceptors in the carotid sinus (glossopharyngeal nerve, CN IX) and aortic arch (vagus nerve, CN X) detect changes in arterial stretch. A drop in blood pressure decreases baroreceptor firing, which disinhibits the vasomotor center in the medulla, causing increased sympathetic outflow (tachycardia, vasoconstriction) and decreased parasympathetic tone. Volatile anesthetics, propofol, and neuraxial blockade blunt this reflex, contributing to hemodynamic instability. Chronic hypertension resets baroreceptors to a higher set point, making these patients more susceptible to hypotension under anesthesia.
Hemodynamic Profiles by Shock Type
Understanding hemodynamic profiles is essential for diagnosing and managing shock under anesthesia:
| Shock Type | CO | SVR | CVP/PCWP | SvO₂ | Example |
|---|---|---|---|---|---|
| Hypovolemic | ↓ | ↑ | ↓ | ↓ | Hemorrhage, dehydration |
| Cardiogenic | ↓ | ↑ | ↑ | ↓ | MI, cardiomyopathy, valvular failure |
| Distributive (septic, early) | ↑ or normal | ↓ | ↓ or normal | ↑ (early) | Sepsis, anaphylaxis, neurogenic |
| Obstructive | ↓ | ↑ | ↑ (right-sided) | ↓ | PE, tension pneumothorax, tamponade |
Autonomic Nervous System Effects on the Heart
| Parameter | Sympathetic (T1–T4) | Parasympathetic (Vagus, CN X) |
|---|---|---|
| Heart rate | ↑ (chronotropy via β1) | ↓ (negative chronotropy via M2 at SA node) |
| Contractility | ↑ (inotropy via β1) | Minimal effect on ventricles |
| Conduction velocity | ↑ (dromotropy via β1 at AV node) | ↓ (negative dromotropy — can cause AV block) |
| Vascular tone | Vasoconstriction (α1); skeletal muscle vasodilation (β2) | Minimal direct vascular effect |
| Coronary arteries | Vasoconstriction (α1), vasodilation (β2 — predominates) | Mild vasodilation |
03 Pharmacokinetics & Pharmacodynamics
Compartment Models
Anesthetic drug distribution is modeled using multi-compartment pharmacokinetics. In a one-compartment model, the drug distributes instantaneously throughout a single homogeneous volume; elimination follows first-order kinetics with a single half-life. Most IV anesthetics follow a two- or three-compartment model: the central compartment (plasma and highly perfused organs — brain, heart, kidneys), a rapidly equilibrating peripheral compartment (muscle, viscera), and a slowly equilibrating deep compartment (fat). After a bolus, plasma concentration falls in three phases: rapid distribution (α phase), slower redistribution (β phase), and terminal elimination (γ phase). For highly lipophilic drugs like propofol and thiopental, rapid redistribution from brain to muscle terminates clinical effect after a single bolus, even though elimination half-life is long.
Context-Sensitive Half-Time
The context-sensitive half-time (CSHT) is the time required for plasma concentration to fall by 50% after stopping a continuous infusion, where "context" refers to infusion duration. Unlike elimination half-life, CSHT accounts for tissue redistribution and is far more clinically relevant for predicting recovery. For remifentanil, CSHT remains constant (~3–4 minutes) regardless of infusion duration because of rapid ester hydrolysis — it is "context-insensitive." For fentanyl, CSHT increases dramatically with prolonged infusion (from ~20 min at 1 hour to >200 min at 6 hours) as peripheral compartments saturate. Propofol CSHT increases modestly (from ~10 min at 1 hour to ~40 min at 8 hours). This concept guides drug selection for cases of varying duration.
Minimum Alveolar Concentration (MAC)
MAC is the minimum alveolar concentration of an inhaled anesthetic at 1 atm that prevents purposeful movement in response to a standard surgical stimulus (skin incision) in 50% of patients. MAC is the ED50 for movement suppression and represents a single point on the dose-response curve. MAC values are additive: 0.5 MAC of sevoflurane + 0.5 MAC of nitrous oxide = 1.0 MAC total. Key MAC variants:
| MAC Variant | Definition | Typical Ratio to MAC |
|---|---|---|
| MAC | Prevents movement to skin incision in 50% | 1.0 |
| MAC-awake | Prevents voluntary response to command in 50% | 0.3–0.5 MAC |
| MAC-BAR | Blocks adrenergic response (tachycardia, hypertension) to incision in 50% | 1.5–1.7 MAC |
| MAC-intubation | Prevents coughing/movement during intubation in 50% | 1.3 MAC |
Decrease MAC (less anesthetic needed): advanced age (MAC decreases ~6% per decade after age 40), hypothermia, hypotension (MAP <40 mmHg), pregnancy (decreases ~30%), acute intoxication (alcohol, opioids), metabolic acidosis, hyponatremia, hypoxia (PaO₂ <40 mmHg), anemia (Hct <10%), concurrent sedatives/opioids/benzodiazepines/clonidine/dexmedetomidine/lithium. Increase MAC (more anesthetic needed): young age (MAC peaks at ~6 months of life), chronic alcohol/drug abuse, hyperthermia, hypernatremia, MAOIs (debated), chronic amphetamine or cocaine use (acute cocaine decreases MAC). No effect on MAC: gender, duration of anesthesia, PaCO₂ (within physiologic range), thyroid function.
Cp50 & Pharmacodynamic Principles
For IV anesthetics, Cp50 is the plasma concentration producing a specified effect in 50% of patients (analogous to MAC for volatiles). Target-controlled infusion (TCI) pumps use pharmacokinetic models (e.g., Marsh or Schnider for propofol, Minto for remifentanil) to achieve a specified plasma or effect-site concentration. The effect-site equilibration constant (ke0) describes the rate of drug equilibration between plasma and the brain; a higher ke0 means faster onset. The biophase (effect-site compartment) cannot be measured directly but is modeled based on the time course of clinical effect.
04 Key Terminology & Abbreviations
| Term | Definition |
|---|---|
| General anesthesia | Drug-induced loss of consciousness with loss of protective reflexes, including inability to maintain airway independently |
| MAC (monitored anesthesia care) | Anesthesia team provides sedation, analgesia, and monitoring while patient breathes spontaneously; ranges from minimal sedation to deep sedation |
| Balanced anesthesia | Combination of inhaled agents, IV drugs, and regional techniques to achieve amnesia, analgesia, akinesia, and autonomic stability |
| TIVA | Total intravenous anesthesia — maintenance using only IV agents (typically propofol + remifentanil), no volatile agents |
| RSI | Rapid sequence induction/intubation — simultaneous induction and paralysis without bag-mask ventilation to minimize aspiration risk |
| TOF | Train-of-four — peripheral nerve stimulation pattern used to assess degree of neuromuscular blockade |
| BIS | Bispectral index — processed EEG monitor for depth of anesthesia; target 40–60 for general anesthesia |
| ETCO₂ | End-tidal carbon dioxide — measured by capnography; correlates with PaCO₂ (normally 2–5 mmHg less) |
| FiO₂ | Fraction of inspired oxygen; room air = 0.21 |
| PEEP | Positive end-expiratory pressure — maintains alveolar patency at end-expiration, improves oxygenation |
| Emergence | Transition from general anesthesia to wakefulness |
| Extubation | Removal of the endotracheal tube after return of protective reflexes and adequate ventilation |
| Preoxygenation | Breathing 100% O₂ to denitrogenate the FRC, extending safe apnea time |
| LAST | Local anesthetic systemic toxicity — CNS and cardiac toxicity from excessive local anesthetic absorption |
| PDPH | Post-dural puncture headache — positional headache after dural puncture, worse upright, relieved supine |
05 ASA Physical Status Classification
The ASA Physical Status Classification System, developed by the American Society of Anesthesiologists, is a subjective assessment of a patient's overall health status before surgery. It is the single most widely used preoperative risk stratification tool and is documented for every anesthetic. It does not directly predict surgical risk but correlates with perioperative morbidity and mortality.
| Class | Definition | Examples | Perioperative Mortality |
|---|---|---|---|
| ASA I | Normal healthy patient | Healthy nonsmoker, no or minimal alcohol use, no significant medical history | <0.03% |
| ASA II | Mild systemic disease without functional limitation | Well-controlled DM or HTN, mild lung disease, BMI 30–40, current smoker, social alcohol use, pregnancy | 0.06% |
| ASA III | Severe systemic disease with substantive functional limitation | Poorly controlled DM or HTN, COPD, morbid obesity (BMI ≥40), active hepatitis, ESRD on dialysis, moderate reduction of EF, history of MI/CVA/TIA >3 months, stable angina, implanted pacemaker | 0.4% |
| ASA IV | Severe systemic disease that is a constant threat to life | Recent MI or CVA (<3 months), ongoing myocardial ischemia, severe valve disease, sepsis, DIC, ARDS, ESRD not on regular dialysis, severe reduction of EF | 4.0% |
| ASA V | Moribund patient not expected to survive without surgery | Ruptured abdominal/thoracic aneurysm, massive trauma, intracranial bleed with mass effect, ischemic bowel with cardiac decompensation, multiorgan failure | 25% |
| ASA VI | Brain-dead patient undergoing organ procurement | Declared brain-dead organ donor | N/A |
The ASA-E modifier is appended to any class (e.g., ASA IIIE) when the procedure is emergent. An emergent case is defined as one in which delay would lead to significant increase in the threat to life or body part. Emergency status independently increases perioperative risk.
06 Preoperative Assessment
Cardiovascular Risk Assessment — ACC/AHA Stepwise Approach
The 2014 ACC/AHA Guideline on Perioperative Cardiovascular Evaluation and Management provides a stepwise algorithm for non-cardiac surgery:
Step 1: Is the surgery emergent? If yes → proceed to surgery with intraoperative risk stratification. Step 2: Does the patient have an active cardiac condition (ACS, decompensated HF, significant arrhythmia, severe valvular disease)? If yes → delay surgery for evaluation and management. Step 3: Estimate surgical risk — low-risk (<1% cardiac risk, e.g., cataract, endoscopy, superficial procedures) → proceed without further testing. Step 4: Assess functional capacity. If ≥4 METs without symptoms → proceed to surgery. Step 5: If functional capacity is poor (<4 METs) or unknown, use the Revised Cardiac Risk Index (RCRI) to guide further testing.
Revised Cardiac Risk Index (RCRI / Lee Index)
Each factor scores 1 point. Total score predicts major adverse cardiac events (MACE):
| Risk Factor | Definition |
|---|---|
| 1. High-risk surgery | Intraperitoneal, intrathoracic, or suprainguinal vascular surgery |
| 2. History of ischemic heart disease | History of MI, positive stress test, current angina, use of nitrates, or pathologic Q waves on ECG |
| 3. History of congestive heart failure | History of CHF, pulmonary edema, PND, bilateral rales or S3, or CXR showing pulmonary vascular redistribution |
| 4. History of cerebrovascular disease | History of TIA or stroke |
| 5. Diabetes requiring insulin | Preoperative insulin use (not diet-controlled or oral agents only) |
| 6. Preoperative creatinine >2 mg/dL | Serum creatinine ≥2.0 mg/dL (or ≥177 µmol/L) |
| RCRI Score | Estimated Risk of MACE |
|---|---|
| 0 points | 3.9% |
| 1 point | 6.0% |
| 2 points | 10.1% |
| ≥3 points | 15% |
Functional Capacity — METs
Functional capacity is measured in metabolic equivalents (METs). 1 MET = resting oxygen consumption (~3.5 mL O₂/kg/min). Patients unable to achieve 4 METs of activity are at elevated perioperative cardiac risk.
| METs | Activity Level |
|---|---|
| 1 MET | Eating, dressing, walking around the house, using the toilet |
| 4 METs | Climbing a flight of stairs, walking uphill, walking on level ground at 4 mph, doing heavy housework (scrubbing floors) |
| 4–10 METs | Climbing >1 flight of stairs, walking uphill briskly, short-distance running, heavy yard work, moderate recreational sports (golf, bowling, dancing) |
| >10 METs | Strenuous sports (swimming, singles tennis, running), heavy physical labor |
Pulmonary Risk Assessment
The ARISCAT score (Assess Respiratory Risk in Surgical Patients in Catalonia) predicts postoperative pulmonary complications (PPCs). Seven independent risk factors are scored: age (≥51: 3 pts; ≥80: 16 pts), preoperative SpO₂ (91–95%: 8 pts; ≤90%: 24 pts), respiratory infection in past month (17 pts), preoperative anemia (Hb ≤10 g/dL: 11 pts), upper abdominal or intrathoracic incision (15 pts), surgery duration >2 hours (16 pts), emergency surgery (8 pts). Low risk: <26 pts (1.6%); intermediate: 26–44 pts (13.3%); high risk: ≥45 pts (42.1%).
Hepatic & Renal Considerations
MELD score (Model for End-Stage Liver Disease) = 3.78 × ln(bilirubin mg/dL) + 11.2 × ln(INR) + 9.57 × ln(creatinine mg/dL) + 6.43. MELD ≥15 carries significant perioperative mortality for non-hepatic surgery; MELD >20 is associated with >50% 90-day mortality. Active hepatitis and Child-Pugh class C cirrhosis are relative contraindications to elective surgery. For renal patients, assess dialysis schedule, potassium level, volume status, and acid-base balance preoperatively; schedule dialysis the day before surgery to optimize electrolytes while allowing post-dialysis volume reequilibration.
Preoperative Laboratory Testing
Routine preoperative laboratory testing in healthy patients undergoing low-risk surgery is no longer recommended (Choosing Wisely campaign). Testing should be guided by patient comorbidities and the planned procedure:
| Test | When to Order |
|---|---|
| CBC | Expected blood loss >500 mL, anemia symptoms, hematologic disease, chemotherapy, chronic kidney disease |
| BMP (electrolytes, BUN, Cr, glucose) | Renal disease, diabetes, diuretic use, ACE-I/ARB use, digoxin, known electrolyte abnormality |
| Coagulation (PT/INR, aPTT) | Anticoagulant use, liver disease, bleeding history, procedures with high bleeding risk |
| Type & screen | Any procedure with expected blood loss >500 mL or anticipated transfusion need |
| ECG | Known cardiovascular disease, diabetes, hypertension, age >65 (for intermediate/high-risk surgery), symptoms suggesting cardiac disease |
| Chest X-ray | New/unstable cardiopulmonary symptoms; NOT routine for age alone |
| Pregnancy test | All women of childbearing potential (recommended by ASA; institution-specific policies) |
| Hemoglobin A1c | Diabetic patients (poor glycemic control — HbA1c >8% associated with higher perioperative complications) |
| PFTs / ABG | Rarely indicated preoperatively; consider for new dyspnea, lung resection surgery, or optimization of severe COPD |
NPO Guidelines (ASA Fasting Recommendations)
| Substance | Minimum Fasting Period | Rationale |
|---|---|---|
| Clear liquids (water, black coffee, tea without milk, apple juice, sports drinks) | 2 hours | Gastric emptying of clear liquids is rapid (<2 hours) |
| Breast milk | 4 hours | Contains fat and protein but empties faster than formula |
| Infant formula, non-human milk | 6 hours | Higher casein content slows gastric emptying |
| Light meal (toast, crackers without meat/fat) | 6 hours | Low-fat meals empty by 6 hours |
| Full meal, fatty/fried foods, meat | 8 hours (or more) | Fat significantly delays gastric emptying |
07 Airway Assessment
Mallampati Classification
The modified Mallampati classification is assessed with the patient sitting upright, mouth open maximally, tongue protruded, without phonation. It predicts the relative size of the tongue to the pharynx and correlates with difficulty of laryngoscopy.
| Class | Structures Visible | Implication |
|---|---|---|
| I | Soft palate, fauces, uvula, tonsillar pillars | Easy intubation expected |
| II | Soft palate, fauces, uvula partially visible | Usually straightforward |
| III | Soft palate, base of uvula only | Moderate difficulty predicted |
| IV | Hard palate only (soft palate not visible) | Difficult intubation likely |
Additional Airway Assessment
Thyromental distance (TMD): measured from the thyroid notch to the mentum with the neck fully extended. Normal ≥6.5 cm (roughly three finger breadths). TMD <6 cm suggests anterior larynx and potential difficulty. Neck mobility: atlanto-occipital extension is essential for the "sniffing position." Decreased mobility (rheumatoid arthritis, ankylosing spondylitis, cervical spine fusion, morbid obesity) impairs alignment of oral, pharyngeal, and laryngeal axes. Inter-incisor distance: mouth opening <3 cm (two finger breadths) limits blade insertion. Upper lip bite test (ULBT): class I — lower incisors can bite above the vermilion of the upper lip (easy); class II — lower incisors can bite the upper lip below the vermilion (moderate); class III — lower incisors cannot bite the upper lip (difficult).
LEMON Mnemonic for Difficult Intubation
L — Look externally (facial trauma, large tongue, short neck, large teeth, obesity, facial hair). E — Evaluate 3-3-2 rule (3 finger breadths mouth opening, 3 finger breadths hyoid-to-chin, 2 finger breadths thyroid notch-to-floor of mouth). M — Mallampati score (III/IV = concern). O — Obstruction (epiglottitis, peritonsillar abscess, neck hematoma, Ludwig angina, tumor). N — Neck mobility (cervical collar, rheumatoid arthritis, ankylosing spondylitis, prior fusion).
Predictors of Difficult Mask Ventilation — MOANS
M — Mask seal difficulty (beard, facial trauma, blood/secretions on face). O — Obesity/obstruction (BMI >26 kg/m², OSA, pregnancy, epiglottitis, neck mass). A — Age >55 years (loss of pharyngeal tone). N — No teeth (edentulous patients — paradoxically difficult to seal the mask despite easier laryngoscopy). S — Stiff lungs/cervical spine (COPD, asthma, pulmonary fibrosis, ARDS, or reduced neck extension).
Predictors of Difficult Intubation — HEAVEN
H — Hypoxemia (SpO₂ <93% at baseline limits safe apnea time). E — Extremes of size (obesity, pediatric/neonatal). A — Anatomic challenge (Mallampati III–IV, short TMD, limited mouth opening, facial disruption). V — Vomit/blood/fluid in the airway (impairs visualization, aspiration risk). E — Exsanguination (hemodynamic collapse during intubation attempt). N — Neck immobility (cervical collar, ankylosing spondylitis, prior surgery).
Cannot-Intubate-Cannot-Oxygenate (CICO) Preparation
Every anesthetic plan must include a CICO contingency. Before any induction: confirm availability of a surgical airway kit (scalpel-bougie-tube or commercial cricothyrotomy set), identify the cricothyroid membrane by palpation or ultrasound, and brief the team. In CICO: perform front-of-neck access (FONA) via cricothyrotomy — vertical skin incision, horizontal stab through cricothyroid membrane, insert bougie, railroad a cuffed 6.0 ETT or tracheostomy tube. Jet ventilation through a needle cricothyrotomy is a temporizing measure but carries risk of barotrauma and is not definitive.
08 Medication Management — Perioperative
Anticoagulants — Hold Times & Bridging
| Drug | Hold Before Surgery | Bridging Consideration |
|---|---|---|
| Warfarin | 5 days (target INR ≤1.5) | Bridge with LMWH if high thrombotic risk (mechanical mitral valve, recent VTE <3 months, CHA₂DS₂-VASc ≥7); no bridging for low risk (AFib with CHA₂DS₂-VASc ≤4, bioprosthetic valve >3 months) |
| Apixaban (Eliquis) | 48 hours (72 h if CrCl <25 or high-bleed procedure) | No bridging needed due to rapid onset/offset |
| Rivaroxaban (Xarelto) | 48 hours (72 h if renal impairment) | No bridging needed |
| Dabigatran (Pradaxa) | 48–72 hours (longer if CrCl <50); idarucizumab available for emergent reversal | No bridging needed |
| Enoxaparin (therapeutic) | 24 hours | Restart 48–72 h postoperatively |
| Unfractionated heparin (IV) | 4–6 hours (check aPTT) | Protamine for reversal if needed |
Antiplatelet Agents
| Drug | Hold Before Surgery | Special Considerations |
|---|---|---|
| Aspirin | 7–10 days for procedures with high bleeding risk; continue for cardiac/vascular surgery and carotid endarterectomy | Do NOT stop within 6 weeks of bare-metal stent or 6 months of drug-eluting stent — risk of stent thrombosis |
| Clopidogrel (Plavix) | 5–7 days | Irreversible P2Y12 inhibitor; platelet recovery depends on new platelet generation |
| Ticagrelor (Brilinta) | 5 days | Reversible P2Y12 inhibitor; faster offset than clopidogrel |
| Prasugrel (Effient) | 7–10 days | Irreversible; more potent than clopidogrel; higher bleeding risk |
Other Medications
Beta-blockers: CONTINUE perioperatively — abrupt withdrawal causes rebound tachycardia/hypertension and increased risk of MI. Do not initiate high-dose beta-blockers on the day of surgery (POISE trial: increased stroke/death). Statins: Continue — perioperative withdrawal increases cardiac risk. ACE inhibitors / ARBs: HOLD on the morning of surgery — associated with refractory intraoperative hypotension. May continue if the indication is CHF with poor EF (individualized decision). Diuretics: Hold morning of surgery to avoid hypovolemia and hypokalemia. Insulin: Basal insulin (glargine, detemir): give 50–80% of usual dose the night before/morning of surgery. Hold prandial insulin while NPO. Hold oral hypoglycemics (especially metformin — lactic acidosis risk with contrast or hemodynamic instability; hold day of surgery, restart after confirmed renal function). MAOIs: Traditional irreversible MAOIs (phenelzine, tranylcypromine) — ideally taper and discontinue 2 weeks before elective surgery due to interaction with indirect sympathomimetics (ephedrine, meperidine → hypertensive crisis or serotonin syndrome). If cannot discontinue, avoid meperidine and indirect-acting agents; use phenylephrine for hypotension. Herbals: Discontinue 1–2 weeks before surgery — garlic (platelet inhibition), ginkgo (bleeding), St. John's wort (induces CYP3A4, reduces drug levels), ephedra (sympathomimetic), valerian (potentiates sedation), kava (hepatotoxic).
09 Induction Agents
| Agent | Mechanism | Induction Dose (IV) | Onset | Duration | Advantages | Adverse Effects / Pearls |
|---|---|---|---|---|---|---|
| Propofol | GABAA receptor potentiation | 1.5–2.5 mg/kg | 30–40 sec | 5–10 min | Rapid, smooth induction; antiemetic properties; decreases ICP; pleasant emergence | Hypotension (decreased SVR and myocardial depression), pain on injection (mitigated by lidocaine 20–40 mg pre-treatment), respiratory depression/apnea, no analgesic properties, green discoloration of urine (benign), egg/soy allergy historically cited but modern consensus suggests safe |
| Etomidate | GABAA receptor agonist at β2/β3 subunits | 0.2–0.3 mg/kg | 30–60 sec | 3–5 min | Hemodynamically stable (minimal change in HR, BP, SVR); cerebro-protective (decreases ICP/CMRO₂) | Adrenal suppression (inhibits 11β-hydroxylase → decreased cortisol for 24–48 h even after single dose — avoid in sepsis), myoclonus (not seizures — subcortical), pain on injection, no analgesic properties, postoperative nausea |
| Ketamine | NMDA receptor antagonist; also affects opioid, monoaminergic, and muscarinic receptors | 1–2 mg/kg IV; 4–6 mg/kg IM | 30–60 sec IV; 3–5 min IM | 10–20 min | Potent analgesic, bronchodilator, maintains airway reflexes and respiratory drive (relative), sympathomimetic (increases HR/BP via central catecholamine release) | Emergence delirium/dysphoria (mitigated by coadministered benzodiazepine), increases ICP (debated — may be safe with controlled ventilation), increases airway secretions (pretreat with glycopyrrolate), increases myocardial O₂ demand, avoid in CAD; drug of choice for induction in severe bronchospasm, hypovolemic/hemorrhagic shock (if not catecholamine-depleted), pericardial tamponade |
| Midazolam | Benzodiazepine; GABAA potentiation at α1 subunit | 0.01–0.05 mg/kg (co-induction); 0.1–0.3 mg/kg (sole induction — rarely used) | 1–3 min | 15–30 min | Anxiolysis, amnesia, anticonvulsant; useful as premedication (1–2 mg IV) | Minimal hemodynamic effect; respiratory depression (especially with opioids); paradoxical agitation in elderly; flumazenil for reversal (0.2 mg IV q1min, max 1 mg — risk of seizures in chronic benzodiazepine users) |
| Thiopental | Barbiturate; GABAA agonist (opens Cl− channel directly at high doses) | 3–5 mg/kg | 30 sec | 5–10 min | Rapid onset, cerebro-protective (decreases CMRO₂/ICP), anticonvulsant | Significant hypotension (venodilation + myocardial depression), respiratory depression, histamine release, tissue necrosis if extravasated (highly alkaline pH ~10.5), laryngospasm, porphyria trigger (absolute contraindication), no analgesia |
10 Volatile Anesthetics
Volatile (inhaled) anesthetics are halogenated ethers (except N₂O) that provide all components of general anesthesia: amnesia, unconsciousness, immobility, and some analgesia. They are administered via calibrated vaporizers (except desflurane, which requires a heated pressurized vaporizer due to its near-room-temperature boiling point of 22.8°C).
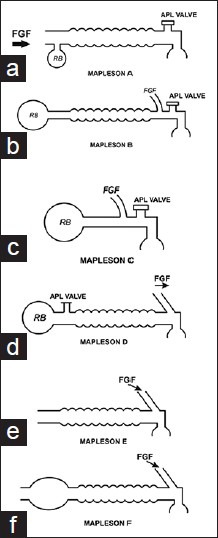
| Agent | MAC (%) | Blood:Gas Partition Coefficient | Onset/Offset | Key Properties |
|---|---|---|---|---|
| Sevoflurane | 2.0 | 0.65 (low) | Rapid | Non-pungent, ideal for inhalation induction (especially pediatrics); minimal airway irritation; metabolized to compound A by CO₂ absorbents (theoretical nephrotoxicity at low flows — clinically insignificant with modern absorbents); dose-dependent vasodilation; mild myocardial depression; minimal effect on HR |
| Desflurane | 6.0 | 0.42 (very low) | Very rapid | Fastest offset of volatile agents — ideal for obese patients and outpatient surgery; pungent — NOT suitable for inhalation induction (causes coughing, breath-holding, laryngospasm); rapid increase in concentration causes sympathetic stimulation (transient tachycardia and hypertension); requires heated vaporizer; higher greenhouse gas potential |
| Isoflurane | 1.15 | 1.46 | Moderate | Potent vasodilator (decreases SVR more than other agents); moderate myocardial depression; can cause coronary steal (theoretical — vasodilates normal coronaries, diverting flow from fixed stenotic vessels); mildly pungent; cost-effective |
| Nitrous oxide (N₂O) | 104 (cannot achieve 1 MAC without hypoxia) | 0.47 | Very rapid | Weak anesthetic but good analgesic; used as adjunct (50–70%) to reduce volatile MAC requirements by 50–60%; second gas effect (high-volume N₂O uptake concentrates co-administered volatile agent → faster induction) |
Nitrous Oxide — Special Concerns
N₂O is 34× more soluble in blood than nitrogen, so it diffuses into closed gas-filled spaces faster than nitrogen can diffuse out, causing expansion. Contraindicated in: pneumothorax, bowel obstruction, middle ear surgery, pneumocephalus, air embolism, recent intraocular gas injection (SF₂ bubble). Diffusion hypoxia: at emergence, rapid N₂O elimination from blood into alveoli dilutes alveolar O₂ — prevented by administering 100% O₂ for 5–10 minutes after discontinuing N₂O. N₂O irreversibly oxidizes the cobalt atom in vitamin B₂₁₂, inhibiting methionine synthase. Prolonged exposure (>6 hours) can cause megaloblastic anemia and subacute combined degeneration of the spinal cord (similar to B₂₁₂ deficiency). Avoid in B₂₁₂-deficient patients.
MAC Values Summary
| Agent | MAC in 100% O₂ (40 yr, 1 atm) | MAC in 70% N₂O | MAC-awake | Blood:Gas PC | Oil:Gas PC |
|---|---|---|---|---|---|
| Sevoflurane | 2.0% | 1.0% | 0.6% | 0.65 | 47 |
| Desflurane | 6.0% | 3.0% | 2.4% | 0.42 | 19 |
| Isoflurane | 1.15% | 0.56% | 0.4% | 1.46 | 98 |
| Nitrous oxide | 104% | N/A | 60–70% | 0.47 | 1.4 |
| Halothane (historical) | 0.75% | 0.3% | 0.3% | 2.5 | 224 |
Cardiovascular & Respiratory Effects (All Volatiles)
All volatile agents cause dose-dependent myocardial depression (decreased contractility and CO), systemic vasodilation (decreased SVR, decreased MAP), and respiratory depression (decreased tidal volume, increased respiratory rate, blunted ventilatory response to CO₂ and hypoxia). They increase cerebral blood flow (which can increase ICP — use cautiously in neurosurgery; sevoflurane has the least effect at <1 MAC). All volatiles are bronchodilators (useful in bronchospasm; sevoflurane preferred due to non-pungent nature). All trigger malignant hyperthermia in susceptible individuals. All potentiate non-depolarizing neuromuscular blockers in a dose-dependent manner.
11 Neuromuscular Blocking Agents
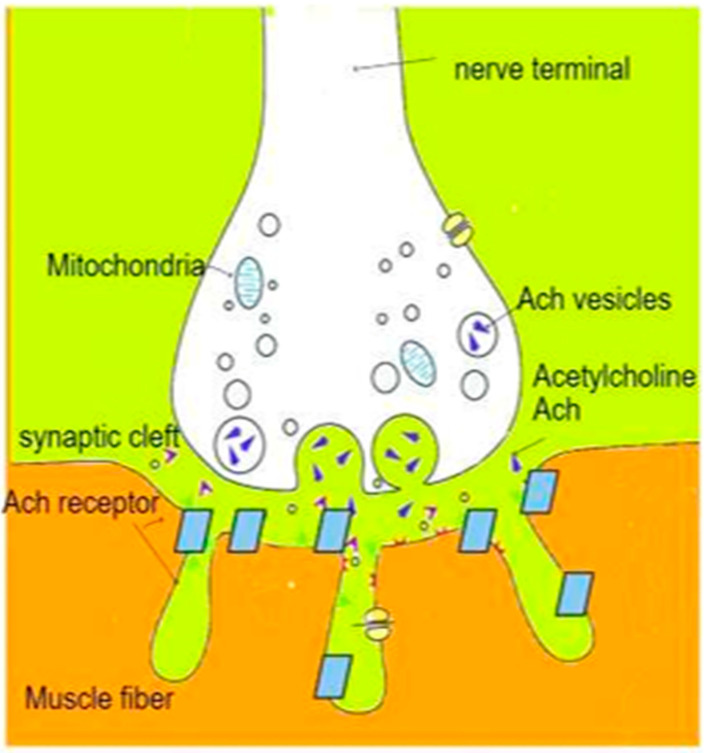
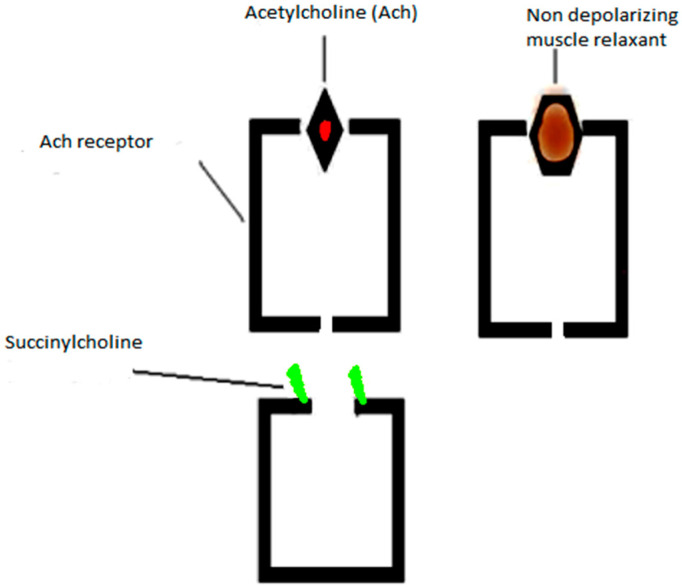
Depolarizing Agent — Succinylcholine
Succinylcholine (SCh) is the only depolarizing neuromuscular blocker in clinical use. It mimics acetylcholine at the nicotinic receptor, causing initial depolarization (fasciculations) followed by sustained depolarization and desensitization of the motor end plate, producing flaccid paralysis.
| Property | Detail |
|---|---|
| Dose | 1–1.5 mg/kg IV (IM: 3–4 mg/kg if no IV access) |
| Onset | 30–60 seconds — fastest onset of any NMBA |
| Duration | 5–10 minutes (ultra-short acting) |
| Metabolism | Rapid hydrolysis by plasma cholinesterase (pseudocholinesterase/butyrylcholinesterase) |
| Phase I block | Sustained depolarization → non-fade on TOF, no post-tetanic potentiation |
| Phase II block | After large/repeated doses (>7 mg/kg), block characteristics resemble non-depolarizing — TOF fade, post-tetanic potentiation |
Succinylcholine causes a normal increase in serum K+ of ~0.5 mEq/L. However, in the following conditions, massive K+ release can cause fatal cardiac arrest: burns (starting 24 h after injury, risk persists for months), denervation injuries (spinal cord injury, stroke — risk begins ~72 h to 1 week after onset), prolonged immobility/ICU myopathy, crush injury, neuromuscular disease (muscular dystrophies — especially Duchenne), severe intra-abdominal infection/sepsis. The mechanism is proliferation of extrajunctional (immature) acetylcholine receptors that remain open longer, releasing K+ from the entire muscle membrane. Contraindicated in these populations.
Other adverse effects: bradycardia (muscarinic stimulation — especially with repeat doses; pretreat children with atropine), increased IOP (transient, ~5–10 mmHg — relative concern in open globe injury, though evidence of actual extrusion is lacking), increased intragastric pressure (offset by increased lower esophageal sphincter tone), masseter muscle rigidity (may herald malignant hyperthermia), malignant hyperthermia trigger (the only NMBA that triggers MH), prolonged block with pseudocholinesterase deficiency (homozygous atypical: duration 4–8 hours; dibucaine number <30).
Non-Depolarizing Agents
| Agent | Intubation Dose | Onset | Duration | Metabolism / Elimination | Key Features |
|---|---|---|---|---|---|
| Rocuronium | 0.6 mg/kg (standard); 1.0–1.2 mg/kg (RSI — onset ~60 sec) | 60–90 sec (standard dose) | 30–45 min | Primarily hepatic uptake and biliary excretion; minor renal | Most commonly used NMBA; only non-depolarizer with rapid-enough onset for RSI at high dose; sugammadex enables immediate reversal |
| Vecuronium | 0.08–0.1 mg/kg | 2–3 min | 25–40 min | Hepatic metabolism (30% active metabolite 3-desacetylvecuronium — accumulates in renal failure) | Hemodynamically stable; no histamine release; packaged as powder requiring reconstitution |
| Cisatracurium | 0.15–0.2 mg/kg | 2–3 min | 25–40 min | Hofmann elimination (non-enzymatic degradation at physiologic pH and temperature) + ester hydrolysis | Organ-independent elimination — ideal for hepatic/renal failure; no histamine release; no vagolytic or sympathetic effects |
| Atracurium | 0.4–0.5 mg/kg | 2–3 min | 20–35 min | Hofmann elimination + ester hydrolysis | Similar organ-independent metabolism as cisatracurium but causes histamine release at higher doses → hypotension, bronchospasm, flushing |
Reversal of Neuromuscular Blockade
Neostigmine (0.03–0.07 mg/kg IV, max 5 mg): an acetylcholinesterase inhibitor that increases ACh at the neuromuscular junction, competing with residual non-depolarizing agent. Must be co-administered with an anticholinergic (glycopyrrolate 0.2 mg per 1 mg neostigmine, or atropine 0.4 mg per 1 mg neostigmine) to block muscarinic effects (bradycardia, salivation, bronchospasm). Neostigmine is ineffective at deep levels of block (requires at least 1–2 twitches on TOF).
Sugammadex is a modified γ-cyclodextrin that encapsulates rocuronium (and to a lesser extent vecuronium) in a 1:1 complex, rendering it inactive. It does not require anticholinergic co-administration and works at any depth of block:
| Depth of Block | TOF Finding | Sugammadex Dose |
|---|---|---|
| Moderate (reappearance of T2) | TOF count ≥2 | 2 mg/kg |
| Deep (no TOF response, PTC 1–2) | Post-tetanic count 1–2 | 4 mg/kg |
| Immediate reversal (RSI rescue — 3 min after rocuronium 1.2 mg/kg) | No response | 16 mg/kg |
TOF Monitoring
Train-of-four stimulation delivers four supramaximal stimuli at 2 Hz to a peripheral nerve (typically the ulnar nerve, measuring adductor pollicis response). The TOF ratio = T4/T1 amplitude. A TOF ratio ≥0.9 (by quantitative monitoring) is required before extubation to minimize residual neuromuscular blockade, which increases the risk of aspiration, hypoxia, and reintubation. Qualitative (subjective) assessment by tactile TOF is unreliable for detecting ratios between 0.4 and 0.9.
12 Opioids in Anesthesia
Opioids are the primary analgesic agents in anesthesia, acting at mu (μ), kappa (κ), and delta (δ) receptors in the CNS and periphery. Mu-receptor activation produces analgesia, euphoria, respiratory depression, miosis, decreased GI motility, and physical dependence.
| Agent | Relative Potency (Morphine = 1) | Typical Anesthetic Dose | Onset (IV) | Duration | Key Features |
|---|---|---|---|---|---|
| Fentanyl | 80–100× | 1–2 mcg/kg (induction adjunct); 50–100 mcg boluses intraop | 1–2 min | 30–60 min | Most commonly used intraoperative opioid; lipophilic with rapid redistribution; minimal histamine release; minimal hemodynamic effect at standard doses; chest wall rigidity with rapid high-dose bolus (>5 mcg/kg) |
| Sufentanil | 500–1000× | 0.1–0.5 mcg/kg (induction); 10–30 mcg boluses | 1–2 min | 20–45 min | 5–10× more potent than fentanyl; used in cardiac surgery; slightly longer duration; greater hemodynamic stability than fentanyl at equipotent doses |
| Remifentanil | 100–200× | Infusion: 0.05–0.2 mcg/kg/min; bolus: 0.5–1 mcg/kg | 1–1.5 min | 3–5 min (offset) | Ultra-short acting; metabolized by nonspecific plasma and tissue esterases — organ-independent; context-insensitive half-time ~3–4 min regardless of infusion duration; ideal for cases requiring rapid emergence; no accumulation; but causes hyperalgesia and acute tolerance — plan for transitional analgesia before stopping infusion |
| Morphine | 1× (reference) | 0.1–0.15 mg/kg | 5–10 min | 3–4 hours | Histamine release (hypotension, bronchospasm, pruritus); active metabolite M6G (potent, accumulates in renal failure); useful for prolonged postoperative analgesia; intrathecal morphine 0.1–0.3 mg for post-surgical pain (24 h duration) |
| Hydromorphone | 5–7× | 0.01–0.02 mg/kg (0.5–1 mg boluses) | 5 min | 3–4 hours | Less histamine release than morphine; no clinically significant active metabolites; preferred in renal insufficiency over morphine |
Equianalgesic Dosing Table (Approximate)
| Opioid | IV Dose (mg) | Oral Dose (mg) |
|---|---|---|
| Morphine | 10 | 30 |
| Hydromorphone | 1.5 | 7.5 |
| Fentanyl | 0.1 (100 mcg) | N/A (transdermal/transmucosal) |
| Oxycodone | N/A | 20 |
| Hydrocodone | N/A | 30 |
| Methadone | Variable (dose-ratio dependent) | Variable |
13 Airway Management Techniques
Mask Ventilation
Bag-mask ventilation (BMV) is the foundational airway skill. The jaw thrust (bilateral mandibular angles lifted anteriorly) and chin lift displace the tongue from the posterior pharynx. Oral airways (Guedel) are sized from the corner of the mouth to the angle of the mandible and are contraindicated in conscious patients (provoke gag/laryngospasm). Nasal airways (nasopharyngeal trumpet) are better tolerated in semi-conscious patients; size = diameter of the patient's little finger; caution with coagulopathy and basilar skull fracture. The C-E technique: the thumb and index finger form a "C" over the mask; the remaining three fingers form an "E" on the mandible. Two-person technique improves seal in difficult cases.
Direct Laryngoscopy
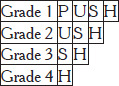
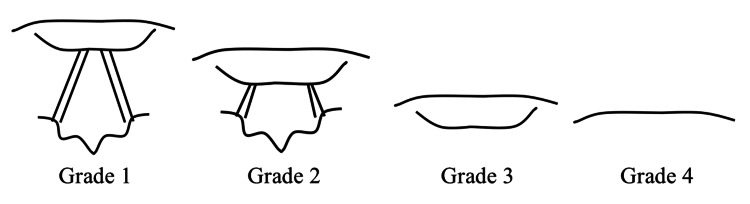
Macintosh blade (curved): inserted into the vallecula between the base of tongue and epiglottis; pressure on the hyoepiglottic ligament indirectly lifts the epiglottis to expose the glottis. Available in sizes 1–4 (most adults: size 3 or 4). Miller blade (straight): tip placed posterior to the epiglottis, directly lifting it. Preferred in neonates and infants (floppy epiglottis), anterior airways, and situations where direct epiglottic lift is needed. Macintosh is generally preferred in adults because it provides more room for tube passage. View is graded by the Cormack-Lehane classification:
| Grade | View | Intubation Difficulty |
|---|---|---|
| I | Full vocal cords visible | Easy |
| IIa | Partial cords visible | Usually straightforward |
| IIb | Only arytenoids or posterior commissure visible | May require adjuncts (bougie) |
| III | Only epiglottis visible, no glottic structures | Difficult — requires bougie or alternative technique |
| IV | Neither epiglottis nor glottis visible | Intubation by DL not possible — use VL, SGA, or surgical airway |
Video Laryngoscopy (VL)
Video laryngoscopes incorporate a camera near the blade tip, providing an indirect view of the glottis on a screen. They generally improve the Cormack-Lehane grade by 1–2 levels compared to DL. GlideScope: hyperangulated blade (60°), provides excellent view but requires a stylet with matching curvature for tube delivery. McGrath MAC: Macintosh-geometry disposable blade; can be used as standard DL or video-assisted. C-MAC (Storz): available in standard Macintosh and D-blade (hyperangulated) configurations. VL is recommended as the first-attempt strategy by many institutions, especially for anticipated difficult airways. Limitations: fogging, blood/secretions on camera, difficulty with tube delivery despite good view ("grade 1 view, grade 3 intubation").
Bougie (Tracheal Tube Introducer)
The bougie (gum elastic bougie, Eschmann introducer) is a critical rescue device for Cormack-Lehane grade IIb–III views. It is a 60–70 cm semi-rigid stylet with an angled (coupé) tip that is advanced blindly under the epiglottis toward the trachea during direct laryngoscopy. Confirmation of tracheal placement: (1) "clicks" felt as the tip slides over tracheal rings (positive in ~90%), and (2) "hold-up" at 30–40 cm as the tip reaches a distal bronchus (100% if present; in esophageal placement, the bougie slides freely without resistance). The ETT is then railroaded over the bougie into the trachea. Studies demonstrate that first-pass intubation success is significantly higher when the bougie is used routinely for DL compared to a standard stylet (BEAM trial). Many institutions now advocate routine bougie use for all DL intubations.
Supraglottic Airways (SGAs)
SGAs sit above the glottis and provide a hands-free airway without tracheal intubation. They are a key rescue device in the difficult airway algorithm. The laryngeal mask airway (LMA) is the prototypical SGA. Types include: LMA Classic (reusable, first-generation), LMA ProSeal (second-generation, with gastric drain tube and higher seal pressures up to 30 cmH₂O), LMA Supreme (disposable second-generation with gastric channel), i-gel (non-inflatable gel cuff, gastric channel), and LMA Fastrach/intubating LMA (designed for blind or fiberoptic-guided intubation through the SGA). Size selection: #3 for 30–50 kg, #4 for 50–70 kg, #5 for 70–100 kg.
Endotracheal Tube Selection & Positioning
Standard ETT sizing for adults: women 7.0–7.5 mm ID, men 7.5–8.0 mm ID. A smaller tube (6.0–6.5) should be selected for: anticipated difficult intubation (easier to pass), nasal intubation, subglottic stenosis, and pregnancy (airway edema). ETT depth at the lip: typically 21 cm for women, 23 cm for men (mnemonic: "21 for women, 23 for men"). Proper position places the tip 3–5 cm above the carina. Confirm placement by: (1) persistent ETCO₂ waveform (most reliable), (2) bilateral chest rise, (3) bilateral breath sounds, (4) absence of epigastric sounds, (5) chest X-ray (tip should project between T2 and T4). The cuff is inflated to achieve a seal at 20–30 cmH₂O (minimal leak technique) — pressures >30 cmH₂O risk tracheal mucosal ischemia and stenosis.
Fiberoptic Intubation
Awake fiberoptic intubation (AFOI) is the gold standard for anticipated difficult airway when mask ventilation is also expected to be difficult. The patient breathes spontaneously throughout. Preparation: antisialagogue (glycopyrrolate 0.2 mg IV), topicalization of the airway with lidocaine (nebulized 4%, oropharyngeal spray, transtracheal injection through cricothyroid membrane, or "spray-as-you-go" through the scope's working channel), and judicious sedation (dexmedetomidine or remifentanil infusion — maintain cooperation and airway reflexes). The flexible bronchoscope is advanced through the nose or mouth, the ETT is loaded on the scope, the scope is guided past the vocal cords, and the ETT is railroaded over the scope into the trachea. Confirm position by visualizing tracheal rings and carina through the scope.
Rapid Sequence Induction (RSI)
RSI is indicated when the patient is at risk for aspiration (full stomach, pregnancy, bowel obstruction, GERD, hiatal hernia, emergency surgery). Sequence: preoxygenation for 3–5 minutes (or 8 vital capacity breaths of 100% O₂), pre-treatment if indicated, induction agent + succinylcholine 1–1.5 mg/kg (or rocuronium 1.2 mg/kg) given simultaneously, cricoid pressure (Sellick maneuver — 30 N of posterior force on cricoid cartilage to occlude the esophagus; controversial, may impair laryngoscopic view), NO bag-mask ventilation between induction and intubation (to avoid gastric insufflation), and intubation within 60 seconds of apnea. Modified RSI: gentle bag-mask ventilation with low pressures (<20 cmH₂O) may be applied if desaturation occurs before intubation conditions are achieved.
ASA 2022 Difficult Airway Algorithm (Simplified)
1. Evaluate the airway: Is difficult airway anticipated? 2. If anticipated difficult airway: awake intubation (fiberoptic, VL) with spontaneous ventilation maintained. 3. If unanticipated difficulty after induction: call for help, attempt mask ventilation. If mask ventilation adequate: try alternative intubation techniques (VL, bougie, different blade, SGA as conduit). If mask ventilation inadequate: place SGA. If SGA ventilation adequate: decide whether to wake the patient or intubate through SGA. 4. Cannot intubate, cannot oxygenate (CICO): emergency surgical airway (cricothyrotomy). Limit intubation attempts to 3 (+1 by a more experienced provider). Between attempts, return to oxygenation via mask or SGA. Consider waking the patient at any point if possible.
14 Local Anesthetics
Mechanism of Action
Local anesthetics (LAs) reversibly block voltage-gated sodium channels from the intracellular side, preventing sodium influx, depolarization, and action potential propagation. They preferentially block small, myelinated (Aδ — pain, temperature) and unmyelinated (C — dull pain, postganglionic autonomic) fibers before large myelinated fibers (Aα — motor, proprioception). Clinical sequence of blockade: sympathetic → pain → temperature → touch → proprioception → motor.
Amides vs. Esters
| Property | Amides (contain "i" before "-caine") | Esters |
|---|---|---|
| Examples | Lidocaine, bupivacaine, ropivacaine, mepivacaine, prilocaine, etidocaine | Chloroprocaine, tetracaine, procaine, cocaine, benzocaine |
| Metabolism | Hepatic (CYP450 enzymes) | Plasma cholinesterase (pseudocholinesterase) hydrolysis |
| Allergic reactions | Exceedingly rare (true allergy) | More common (PABA metabolite is the allergen); cross-reactivity among esters |
| Stability | Heat stable | Less stable in solution |
Pharmacology of Key Agents
| Agent | pKa | Onset | Protein Binding | Duration | Max Dose (no epi / with epi) | Clinical Use |
|---|---|---|---|---|---|---|
| Lidocaine | 7.7 | Rapid | 65% | 60–120 min | 4.5 / 7 mg/kg | Infiltration, nerve block, epidural, topical, IV regional (Bier block), antiarrhythmic |
| Bupivacaine | 8.1 | Slow | 95% | 240–480 min | 2.5 / 3 mg/kg | Epidural, spinal, nerve block; most cardiotoxic LA (binds cardiac Na+ channels with slow dissociation) |
| Ropivacaine | 8.1 | Slow | 94% | 240–480 min | 3 / 3.5 mg/kg | Similar to bupivacaine but less cardiotoxic and less motor block; S-enantiomer only; preferred for epidural infusions and nerve blocks |
| Chloroprocaine | 8.7 | Very rapid | Low | 30–60 min | 11 / 14 mg/kg | Epidural (rapid onset, short duration, rapid metabolism — ideal for obstetric emergency epidural top-up); very high max dose due to rapid metabolism |
| Tetracaine | 8.5 | Slow | 85% | 180–600 min | 1.5 mg/kg (topical) | Spinal anesthesia; topical (ophthalmology); high potency |
Local Anesthetic Systemic Toxicity (LAST)
LAST occurs from inadvertent intravascular injection or excessive absorption. Plasma levels rise and produce a biphasic toxicity pattern:
CNS toxicity (occurs first, at lower plasma levels): perioral numbness, metallic taste, tinnitus, visual disturbances, dizziness → muscle twitching → seizures → CNS depression/coma. Cardiovascular toxicity (at higher levels): hypotension, bradycardia, conduction abnormalities, wide-complex arrhythmias, ventricular tachycardia/fibrillation, asystole. Bupivacaine is most cardiotoxic because it binds cardiac sodium channels tightly ("fast-in, slow-out").
Treatment — Intralipid 20% Protocol: Bolus: 1.5 mL/kg over 1 minute. Infusion: 0.25 mL/kg/min. Repeat bolus once or twice if cardiovascular instability persists. Increase infusion to 0.5 mL/kg/min if needed. Maximum recommended dose: ~12 mL/kg. Additional measures: stop LA injection, call for help, secure airway, benzodiazepines for seizures (avoid propofol in hemodynamically unstable patient), small-dose epinephrine (≤1 mcg/kg) for hemodynamic support (avoid large doses and vasopressin), consider cardiopulmonary bypass or ECMO for refractory arrest. Avoid: lidocaine for arrhythmias, calcium channel blockers, beta-blockers, large-dose epinephrine.
15 Neuraxial Anesthesia — Spinal & Epidural
Spinal Anesthesia
Spinal (subarachnoid) anesthesia involves a single injection of local anesthetic into the cerebrospinal fluid (CSF) in the lumbar subarachnoid space. The spinal cord terminates at L1–L2 in adults (the conus medullaris), so the needle is inserted at L3–L4 or L4–L5 interspace to avoid cord injury. The L4–L5 interspace is identified by a line connecting the iliac crests (Tuffier's line). Needle types include Quincke (cutting tip — higher PDPH risk) and Whitacre/Sprotte (pencil-point — lower PDPH risk, preferred). CSF confirmation (clear, free-flowing) confirms subarachnoid placement.
The most common agent is hyperbaric bupivacaine 0.75% (with 8.25% dextrose), which is denser than CSF and affected by gravity/patient position (baricity). Typical dose: 10–15 mg for lower abdominal/lower extremity surgery; 7.5–10 mg for C-section. Onset: 2–5 minutes. Duration: 90–150 minutes (depending on dose and adjuvants). Common adjuvants: fentanyl 10–25 mcg (faster onset, improved quality, minimal prolongation), morphine 0.1–0.2 mg (prolonged postoperative analgesia up to 24 hours but delayed respiratory depression risk up to 24 h).
Dermatome Levels by Procedure
| Procedure | Target Dermatome Level |
|---|---|
| Foot/ankle surgery | T12–L1 |
| Knee surgery (TKA, arthroscopy) | T10 |
| Hip surgery (THA, hip fracture repair) | T10 |
| TURP, vaginal delivery | T10 |
| Lower abdominal surgery (hernia, appendectomy) | T6–T8 |
| Cesarean section | T4 (nipple line) |
| Upper abdominal surgery | T4 |
Spinal Complications
Post-dural puncture headache (PDPH): incidence 1–3% with pencil-point needles, up to 30% with large-bore cutting needles. Frontal/occipital headache worsened by sitting/standing, relieved within 30 minutes of lying supine. Onset within 5 days of dural puncture. Mechanism: CSF leak through dural hole → decreased intracranial pressure → compensatory cerebral vasodilation and traction on meninges. Management: conservative (hydration, caffeine 300–500 mg PO/IV, analgesics) for 24–48 h; epidural blood patch (15–25 mL autologous blood injected into epidural space) for persistent symptoms — >90% effective. Hypotension: most common side effect of spinal anesthesia (sympathectomy → vasodilation + decreased venous return); treat with IV fluid, phenylephrine 50–100 mcg boluses (first-line), or ephedrine 5–10 mg (if bradycardia). High/total spinal: excessive cephalad spread → hypotension, bradycardia, respiratory insufficiency (diaphragmatic paralysis at C3–C5), loss of consciousness; treat with hemodynamic support, airway management, and intubation if needed. TNS (transient neurologic symptoms): pain/dysesthesia in buttocks/legs within 24 h, resolves in days; more common with lidocaine — a reason bupivacaine is preferred for spinals.
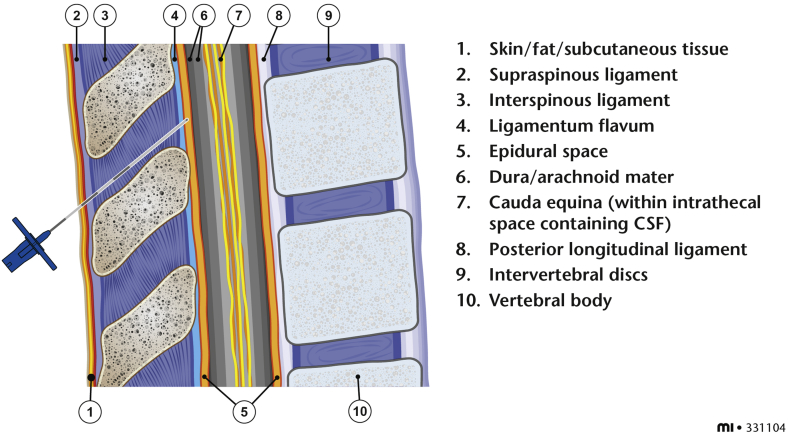
Epidural Anesthesia
The epidural space lies between the ligamentum flavum and the dura mater. Access is via the loss of resistance (LOR) technique: a syringe filled with saline (or air) is attached to the Tuohy needle; as the needle advances through the ligamentum flavum into the epidural space, resistance suddenly disappears. A catheter is then threaded 3–5 cm into the space for continuous infusion. Epidural anesthesia can be placed at any vertebral level (thoracic for chest/abdominal surgery, lumbar for lower extremity/obstetric).
Test dose: 3 mL of lidocaine 1.5% with 1:200,000 epinephrine (45 mg lidocaine + 15 mcg epinephrine). If intrathecal: rapid motor block within 3–5 minutes. If intravascular: heart rate increase ≥20 bpm within 60 seconds. Patient-controlled epidural analgesia (PCEA): typical regimen for labor — bupivacaine 0.0625–0.1% + fentanyl 2 mcg/mL; basal rate 6–10 mL/h, demand dose 5–8 mL, lockout 10–20 minutes.
Combined Spinal-Epidural (CSE)
The CSE technique provides the rapid onset and reliability of spinal anesthesia with the flexibility of an epidural catheter for prolonged procedures or postoperative analgesia. Needle-through-needle technique: the Tuohy needle is placed in the epidural space, a long spinal needle is advanced through it to puncture the dura, the intrathecal dose is injected, the spinal needle is withdrawn, and the epidural catheter is threaded. Commonly used for C-sections and labor analgesia.
16 Peripheral Nerve Blocks — Upper Extremity
Brachial Plexus Anatomy
The brachial plexus is formed by the ventral rami of C5–T1 and follows the mnemonic Robert Taylor Drinks Cold Beer — Roots, Trunks, Divisions, Cords, Branches:
| Level | Components | Location |
|---|---|---|
| Roots | C5, C6, C7, C8, T1 | Exit intervertebral foramina, between anterior and middle scalene muscles |
| Trunks | Superior (C5–C6), Middle (C7), Inferior (C8–T1) | Interscalene groove, above the clavicle |
| Divisions | Each trunk splits into anterior and posterior division (6 total) | Behind the clavicle |
| Cords | Lateral (anterior divisions of superior + middle trunks), Posterior (all posterior divisions), Medial (anterior division of inferior trunk) | Named by relationship to axillary artery, in the infraclavicular region |
| Branches | Musculocutaneous (lateral cord), axillary (posterior), radial (posterior), median (lateral + medial), ulnar (medial) | Axilla and distal |
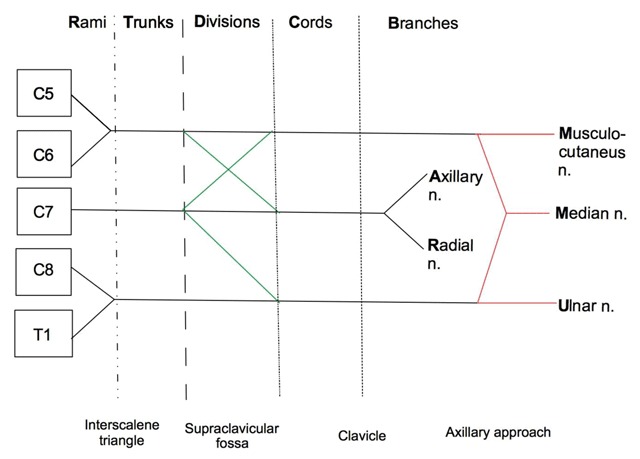
Interscalene Block
The interscalene block targets the roots/trunks of the brachial plexus (C5–C7) in the interscalene groove between the anterior and middle scalene muscles at the level of the cricoid cartilage (C6). It provides excellent anesthesia for shoulder and proximal humerus surgery. The inferior trunk (C8–T1) is commonly spared, resulting in incomplete ulnar nerve blockade — not ideal for hand/forearm procedures. Key complications: phrenic nerve paralysis (virtually 100% ipsilateral hemidiaphragm paresis — avoid bilaterally and in patients with contralateral phrenic nerve dysfunction or significant pulmonary disease), Horner syndrome (miosis, ptosis, anhidrosis), recurrent laryngeal nerve block (hoarseness), vertebral artery injection, epidural/intrathecal spread.
Supraclavicular Block
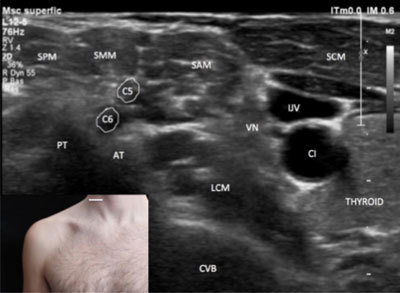
The supraclavicular block targets the trunks/divisions at the level of the first rib, lateral to the subclavian artery, behind the clavicle. Often called the "spinal of the arm" because it provides rapid, dense blockade of the entire upper extremity below the shoulder. Excellent for elbow, forearm, wrist, and hand surgery. Under ultrasound guidance, the plexus appears as a "cluster of grapes" lateral and posterior to the subclavian artery, above the hyperechoic first rib. Risk: pneumothorax (historically ~1% with landmark technique, now <0.5% with ultrasound), phrenic nerve paresis (~50%).
Infraclavicular Block
The infraclavicular block targets the cords beneath the clavicle, around the axillary artery. The needle is inserted medial to the coracoid process, directed posteriorly. Advantages: catheter-friendly location (less prone to dislodgment), complete coverage below the mid-humerus including the musculocutaneous nerve (which may be missed with axillary block). Lower risk of pneumothorax and phrenic nerve paresis than supraclavicular approach.
Axillary Block
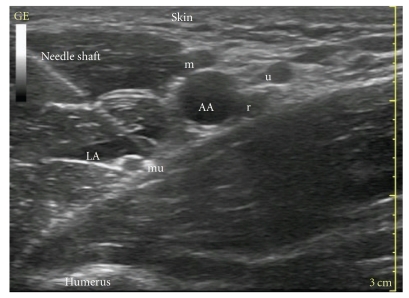
The axillary block targets the terminal branches (median, ulnar, radial) of the brachial plexus in the axilla around the axillary artery. Ideal for hand, wrist, and forearm surgery. Safest brachial plexus block (no pneumothorax, no phrenic nerve paresis). The musculocutaneous nerve leaves the brachial plexus sheath early and enters the coracobrachialis muscle — it must be blocked separately (injected within the coracobrachialis) for complete lateral forearm coverage. Under ultrasound, the nerves are identified relative to the axillary artery: median (superficial/lateral), ulnar (superficial/medial), radial (posterior/deep).
17 Peripheral Nerve Blocks — Lower Extremity & Truncal
Lower Extremity Blocks
| Block | Target Nerve(s) | Indication | Technique / Pearls |
|---|---|---|---|
| Femoral nerve block | Femoral nerve (L2–L4) | Anterior thigh, knee (TKA, ACL repair), femur fracture | Ultrasound: nerve is lateral to femoral artery below inguinal ligament. Provides quadriceps weakness — fall risk. Continuous catheter for TKA common but causes quad weakness. |
| Adductor canal block | Saphenous nerve (terminal sensory branch of femoral) + nerve to vastus medialis | TKA (motor-sparing alternative to femoral block), knee arthroscopy | Performed at mid-thigh where the saphenous nerve lies beneath the sartorius muscle alongside the superficial femoral artery. Preserves quadriceps strength → enables earlier ambulation. |
| Sciatic nerve block | Sciatic nerve (L4–S3) | Posterior thigh, leg below knee, foot/ankle | Multiple approaches: subgluteal (posterior), anterior, infragluteal-parabiceps. Provides complete anesthesia below knee when combined with saphenous block. |
| Popliteal sciatic block | Sciatic nerve at the popliteal fossa (before division into tibial and common peroneal) | Foot and ankle surgery | Performed 5–10 cm above the popliteal crease. Block both tibial and common peroneal components. Add saphenous nerve block for medial ankle (saphenous/great saphenous vein territory). |
| Lumbar plexus / Psoas compartment block | L2–L4 (femoral, lateral femoral cutaneous, obturator) | Hip surgery, proximal femur fracture | Deep block, landmark-based or ultrasound/nerve stimulator. Risks: epidural spread, retroperitoneal hematoma. Essentially a "3-in-1" block for the lumbar plexus. |
Truncal & Fascial Plane Blocks
| Block | Fascial Plane / Target | Indication | Pearls |
|---|---|---|---|
| TAP block (Transversus Abdominis Plane) | Between internal oblique and transversus abdominis muscles | Abdominal wall analgesia (laparoscopy, cesarean section, hernia, appendectomy) | Somatic (not visceral) analgesia only; injection of 15–20 mL per side; lateral approach covers T10–L1; subcostal approach extends to T6–T9 |
| Erector spinae plane (ESP) block | Deep to erector spinae muscle, superficial to transverse process | Thoracic and abdominal wall analgesia, rib fractures, thoracotomy, breast surgery | LA diffuses to paravertebral space providing both somatic and some visceral coverage; relatively safe (muscle target far from pleura/neuraxis); bilateral blocks for midline procedures |
| Quadratus lumborum (QL) block | Around the quadratus lumborum muscle (anterior, lateral, or posterior approach) | Abdominal wall analgesia, hip surgery adjunct, cesarean section | Provides broader dermatomal coverage than TAP (T7–L1), may include visceral component via spread to paravertebral/thoracolumbar fascia; posterior approach is most common |
| Paravertebral block (PVB) | Paravertebral space (bounded by parietal pleura laterally, vertebral body medially) | Unilateral thoracic/abdominal analgesia: thoracotomy, breast surgery, rib fractures, hernia | Provides ipsilateral somatic and sympathetic block; lower risk of hypotension than epidural; risk of pneumothorax; can place catheter for continuous infusion |
| Pectoralis (PECS) I & II blocks | PECS I: between pec major and minor; PECS II: between pec minor and serratus anterior | Breast surgery, port placement | PECS II provides broader coverage (lateral cutaneous branches of intercostal nerves, long thoracic, thoracodorsal); increasingly popular for breast surgery analgesia |
18 Standard ASA Monitors
The ASA Standards for Basic Anesthetic Monitoring (last affirmed 2020) mandate monitoring of oxygenation, ventilation, circulation, and temperature during all anesthetics. A qualified anesthesia provider must be continuously present.
Oxygenation
Pulse oximetry (SpO₂) is mandatory for every anesthetic. It measures the ratio of oxyhemoglobin to total hemoglobin using two wavelengths of light (660 nm red, 940 nm infrared) based on the Beer-Lambert law (light absorption is proportional to concentration of the absorbing substance and path length). Limitations: inaccurate with carboxyhemoglobin (CO-Hb reads falsely high ~100%), methemoglobinemia (SpO₂ trends toward 85% regardless of true saturation), severe peripheral vasoconstriction, nail polish (especially blue/green), motion artifact, and anemia (accurate until Hb <5 g/dL). An oxygen analyzer with a low-concentration alarm must be used in the breathing circuit to verify FiO₂.
Ventilation
Capnography (ETCO₂) is mandatory whenever an ETT or SGA is in use, and strongly encouraged during MAC/sedation. It is the most reliable method to confirm endotracheal tube placement (presence of CO₂ waveform) and is the earliest indicator of esophageal intubation (absent CO₂). Normal ETCO₂: 35–45 mmHg, typically 2–5 mmHg less than PaCO₂.
Phase I: Baseline (inspiration or early expiration — dead space gas with no CO₂). Phase II: Rapid upstroke (mixing of dead space and alveolar gas). Phase III: Alveolar plateau (uniform alveolar gas exhalation; the highest point at end-expiration is the ETCO₂ value). Phase IV (inspiratory downstroke): Rapid return to baseline as fresh gas is inspired. A Phase 0 (cleft on plateau) suggests spontaneous respiratory effort during mechanical ventilation. An upsloping Phase III ("shark fin") pattern indicates obstructive airway disease (bronchospasm, COPD). Sudden loss of ETCO₂ to zero: esophageal intubation, circuit disconnection, or cardiac arrest. Gradual rise in ETCO₂: malignant hyperthermia, hypoventilation, increased CO₂ production (fever, sepsis). Sudden decrease in ETCO₂ (not zero): pulmonary embolism, decreased cardiac output, air embolism.
Circulation
ECG is continuously displayed. Standard 5-lead monitoring allows ST-segment analysis in leads II (inferior ischemia, P-wave analysis, arrhythmia detection) and V5 (anterior/lateral ischemia). Combined leads II + V5 detect ~95% of intraoperative ST changes. Non-invasive blood pressure (NIBP) is measured at intervals of ≤5 minutes. Cuff size matters: too small overestimates, too large underestimates (bladder width should be 40% of arm circumference).
Temperature
Monitoring is indicated whenever clinically significant changes in body temperature are anticipated. Sites (in order of accuracy for core temperature): pulmonary artery catheter (gold standard), distal esophageal, nasopharyngeal, tympanic membrane, bladder. Hypothermia (<36°C) is common under general anesthesia (impaired thermoregulation, cold OR environment, cold IV fluids, heat loss from surgical exposure) and increases bleeding (impaired coagulation), infection risk, and cardiac events. Active warming (forced-air warming blanket, fluid warmer) is standard of care.
Neuromuscular Monitoring Sites & Patterns
Beyond TOF, several other stimulation patterns are used: single twitch (1 Hz) — simple but insensitive (cannot detect residual block <75% receptor occupancy). Tetanic stimulation (50 Hz for 5 sec) — sustained stimulus; fade indicates non-depolarizing block. Post-tetanic count (PTC) — after tetanic stimulus, count the number of post-tetanic twitches; used to assess deep block when TOF = 0 (PTC 1–2 = deep; PTC ≥10 = approaching moderate block, TOF will reappear soon). Double burst stimulation (DBS) — two bursts of 3 stimuli at 50 Hz separated by 750 ms; easier to detect fade manually than TOF (clinician compares two discrete responses rather than four). Monitoring site: the adductor pollicis (ulnar nerve at the wrist) is the reference muscle for neuromuscular monitoring. The corrugator supercilii (facial nerve) has faster onset and recovery than the adductor pollicis — useful for predicting intubating conditions but overestimates recovery at the diaphragm and peripheral muscles.
Depth of Anesthesia — BIS Monitor
The bispectral index (BIS) is a processed EEG parameter scaled from 0 (isoelectric) to 100 (fully awake). Target range for general anesthesia: 40–60. BIS >60: increased risk of awareness; BIS <40: deeper than necessary, may increase postoperative delirium. Indications: TIVA (no end-tidal volatile to titrate), high-risk patients for awareness (cardiac surgery, C-section under GA, trauma, history of awareness), neuromuscular blockade (clinical signs of light anesthesia are masked).
19 Invasive Monitoring
Goal-Directed Fluid Therapy (GDFT)
Rather than relying on fixed formulas, goal-directed fluid therapy uses dynamic hemodynamic parameters to guide individual fluid administration. Static parameters (CVP, PCWP) are poor predictors of fluid responsiveness. Dynamic parameters include: pulse pressure variation (PPV) and stroke volume variation (SVV) — PPV >13% or SVV >12% during mechanical ventilation (in sinus rhythm, TV ≥8 mL/kg, closed chest) predicts fluid responsiveness with ~85% sensitivity and specificity. Passive leg raise (PLR) test: raising the legs to 45° from supine autotransfuses ~300 mL from the lower extremities; a ≥10% increase in CO within 1 minute predicts fluid responsiveness — this works even with spontaneous breathing, arrhythmias, and open chest (unlike PPV/SVV). Point-of-care cardiac output monitors (FloTrac/Vigileo, LiDCO, ClearSight) and transesophageal echocardiography enable real-time GDFT implementation.
Arterial Line
An arterial catheter provides continuous, beat-to-beat blood pressure measurement and facilitates repeated arterial blood gas sampling. Common sites: radial (most common — perform modified Allen test to confirm dual blood supply via ulnar artery before cannulation), femoral, brachial, dorsalis pedis. Indications: hemodynamic instability anticipated, vasoactive infusions, frequent ABGs, deliberate hypotension, morbid obesity (NIBP unreliable).
Arterial waveform analysis: the waveform shows a systolic upstroke, systolic peak, dicrotic notch (aortic valve closure), and diastolic runoff. Overdamping (air bubble, clot, kink, soft tubing): underestimates systolic and overestimates diastolic pressure, loss of dicrotic notch, slurred upstroke. Underdamping/resonance (long tubing, tachycardia): overestimates systolic and underestimates diastolic, ringing artifact. The fast-flush (square wave) test assesses system dynamics: 1–2 oscillations before settling = optimally damped; >2 oscillations = underdamped; no oscillation = overdamped. MAP is least affected by damping and is the most reliable value.
Central Venous Catheter (CVC)
A CVC is placed in a large central vein for venous access (vasoactive infusions, caustic drugs, TPN), hemodynamic monitoring (CVP), and rapid volume resuscitation (large-bore introducer sheath). Common sites: internal jugular vein (IJV) — preferred, ultrasound-guided, right side preferred (straight path to SVC); subclavian vein — lower infection rate but higher pneumothorax risk, not compressible if hemorrhage occurs; femoral vein — easiest landmark, no pneumothorax risk, but higher infection rate and DVT risk. Confirm tip position in the SVC-RA junction by chest X-ray.
CVP waveform has three positive waves and two descents: a wave (atrial contraction — absent in atrial fibrillation, giant "cannon" a waves in complete heart block or junctional rhythm when atrium contracts against a closed tricuspid valve), c wave (tricuspid valve bulging into atrium during isovolumetric contraction), v wave (passive atrial filling during ventricular systole, with tricuspid closed — giant v waves in tricuspid regurgitation), x descent (atrial relaxation), y descent (early ventricular filling after tricuspid opening). Normal CVP: 2–8 mmHg. CVP reflects right atrial pressure but is a poor predictor of fluid responsiveness.
Pulmonary Artery Catheter (PAC / Swan-Ganz)
The PAC is a flow-directed, balloon-tipped catheter inserted through a central vein introducer, advanced through the RA, RV, and PA, and "wedged" in a branch of the PA. It measures: RA pressure (CVP), RV pressure, PA pressure (systolic/diastolic/mean), pulmonary capillary wedge pressure (PCWP) (reflects LA pressure and LVEDP when mitral valve is normal; normal 6–12 mmHg), cardiac output (thermodilution), and mixed venous oxygen saturation (SvO₂; normal 65–75%, low = increased extraction from inadequate DO₂).
| Parameter | Formula | Normal Value |
|---|---|---|
| Cardiac output (CO) | HR × SV (or thermodilution) | 4–8 L/min |
| Cardiac index (CI) | CO / BSA | 2.5–4.0 L/min/m² |
| SVR | (MAP − CVP) × 80 / CO | 800–1200 dynes·s/cm&sup5; |
| PVR | (mPAP − PCWP) × 80 / CO | 100–250 dynes·s/cm&sup5; |
| Stroke volume (SV) | CO / HR × 1000 | 60–100 mL/beat |
| Stroke volume index (SVI) | CI / HR × 1000 | 33–47 mL/beat/m² |
20 Fluid Management & Transfusion
Crystalloids vs. Colloids
Crystalloids are electrolyte solutions that distribute freely across the extracellular space. Approximately 25% of infused crystalloid remains intravascular after 30 minutes (the rest shifts to the interstitium). Balanced crystalloids — lactated Ringer's (LR: Na 130, K 4, Ca 3, Cl 109, lactate 28 mEq/L) and PlasmaLyte (Na 140, K 5, Mg 3, Cl 98, acetate 27, gluconate 23) — are preferred over normal saline (NS: Na 154, Cl 154) for large-volume resuscitation because NS causes hyperchloremic metabolic acidosis and may worsen AKI (SMART trial). Colloids (albumin 5%, hydroxyethyl starch) theoretically expand intravascular volume more efficiently (~75% remains intravascular), but HES increases renal injury and mortality in critically ill patients (6S and CHEST trials) and is no longer recommended in sepsis or ICU settings.
Maintenance Fluid Calculation — 4-2-1 Rule
| Body Weight | Rate |
|---|---|
| First 10 kg | 4 mL/kg/h |
| Second 10 kg (11–20 kg) | 2 mL/kg/h |
| Each additional kg (>20 kg) | 1 mL/kg/h |
Example: 70 kg adult = (4 × 10) + (2 × 10) + (1 × 50) = 110 mL/h. Intraoperative fluid management additionally accounts for: NPO deficit (hourly rate × hours fasted — replace 50% in first hour, 25% in second and third hours), ongoing losses (estimated blood loss, third-space losses — minimal for superficial surgery, 4–8 mL/kg/h for major abdominal), and urine output (target 0.5–1 mL/kg/h).
Transfusion
Massive transfusion protocol (MTP): activated for anticipated need of ≥10 units pRBCs in 24 hours (or ≥4 units in 1 hour). Goal-directed: transfuse pRBCs, FFP, and platelets in a 1:1:1 ratio (PROPPR trial). Adjuncts: tranexamic acid 1 g IV over 10 min then 1 g over 8 hours (CRASH-2 — give within 3 hours of injury), calcium replacement (citrate in blood products chelates calcium — monitor ionized calcium, give CaCl₂ 1 g per ~4 units), cryoprecipitate if fibrinogen <150 mg/dL.
| Component | Trigger (General) | Trigger (Cardiac/Ischemic) | Expected Effect per Unit |
|---|---|---|---|
| Packed RBCs (pRBCs) | Hb <7 g/dL | Hb <8 g/dL (or active bleeding/ischemia) | Increases Hb by ~1 g/dL, Hct by ~3% |
| Fresh frozen plasma (FFP) | INR >1.5 with active bleeding | Same | Increases factors by ~5%; one unit = 200–250 mL |
| Platelets | <50,000/µL with surgery/bleeding; <10,000 prophylactic | <100,000 for neurosurgery or ophthalmic surgery | Increases count by 30,000–50,000/µL per unit (apheresis) |
| Cryoprecipitate | Fibrinogen <150 mg/dL | Same | Increases fibrinogen by ~50 mg/dL per 5-unit pool |
Transfusion Reactions
TRALI (transfusion-related acute lung injury): non-cardiogenic pulmonary edema within 6 hours of transfusion, bilateral infiltrates, hypoxemia, no evidence of volume overload. Leading cause of transfusion-related death. Treatment: supportive (stop transfusion, oxygen, mechanical ventilation if needed). TACO (transfusion-associated circulatory overload): cardiogenic pulmonary edema from volume excess. Distinguishing feature: elevated BNP and JVP in TACO but not TRALI. Treatment: diuretics, slow transfusion rate.
Complications of Massive Transfusion
Beyond TRALI and TACO, massive transfusion carries several additional risks: hypothermia (stored blood is at 4°C — use fluid warmers for all rapid transfusions), hypocalcemia (citrate in blood products chelates ionized calcium — monitor iCa²+ and give CaCl₂ 1 g IV per approximately 4 units pRBCs; symptoms include hypotension, prolonged QT, and cardiac depression), hyperkalemia (potassium leaks from stored RBCs, especially older units — can reach 30–50 mEq/L in the supernatant of aged pRBCs; wash or use fresh units for neonates), dilutional coagulopathy (progressive depletion of factors and platelets with crystalloid and pRBC-only resuscitation — prevented by balanced ratio transfusion), metabolic alkalosis (citrate is metabolized to bicarbonate — late complication after massive transfusion), and 2,3-DPG depletion in stored blood (left-shifts the oxyhemoglobin curve, impairing O₂ release to tissues).
Viscoelastic Testing
TEG (thromboelastography) and ROTEM (rotational thromboelastometry) provide point-of-care assessment of the entire coagulation cascade from clot initiation through fibrinolysis.
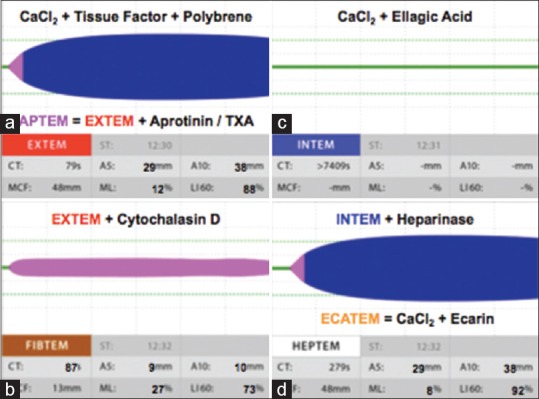
Key TEG parameters: R time (reaction time — time to initial clot formation; prolonged = factor deficiency → give FFP), K time (clot kinetics — time to reach 20 mm amplitude), α angle (rate of clot strengthening — low = fibrinogen deficiency → give cryoprecipitate), MA (maximum amplitude — reflects platelet function and fibrinogen; low = platelet deficiency/dysfunction → give platelets or DDAVP), LY30 (percent lysis at 30 minutes — >3% = fibrinolysis → give tranexamic acid).
21 Malignant Hyperthermia
Pathophysiology
Malignant hyperthermia (MH) is a pharmacogenetic syndrome triggered by exposure to volatile anesthetic agents (sevoflurane, desflurane, isoflurane, halothane) and/or succinylcholine in genetically susceptible individuals. The defect is most commonly an autosomal dominant mutation in the RYR1 gene (ryanodine receptor type 1 on skeletal muscle sarcoplasmic reticulum). Triggering agents cause uncontrolled release of calcium from the sarcoplasmic reticulum into the myoplasm, resulting in sustained muscle contraction, a hypermetabolic state (massively increased O₂ consumption and CO₂ production), heat generation, rhabdomyolysis, and ultimately cellular death if untreated. Mortality without treatment exceeds 70%; with early dantrolene, mortality is <5%.
Clinical Presentation
Earliest sign: unexplained, rapidly rising ETCO₂ (despite increasing minute ventilation) — this is often the first indicator. Tachycardia (out of proportion, refractory to treatment). Masseter muscle rigidity (especially after succinylcholine — jaw cannot be opened). Metabolic/respiratory acidosis (mixed). Generalized skeletal muscle rigidity. Hyperthermia (may rise 1–2°C every 5 minutes — this is a relatively LATE sign; do not wait for temperature elevation to begin treatment). Rhabdomyolysis (elevated CK, myoglobinuria, hyperkalemia). Late: DIC, cardiac arrest, renal failure.
Treatment — Dantrolene Protocol
1. Call for help — MH cart. Contact the MH Hotline: 1-800-644-9737 (MHAUS). 2. Immediately discontinue ALL volatile agents and succinylcholine. Do NOT delay for completion of surgery. 3. Hyperventilate with 100% O₂ at high fresh gas flows (≥10 L/min). Change the circuit and CO₂ absorber if time permits, but do not delay dantrolene. 4. Administer dantrolene 2.5 mg/kg IV as rapidly as possible. Repeat every 5 minutes until signs resolve (ETCO₂ decreasing, HR decreasing, temperature stabilizing). May require up to 10 mg/kg total (occasionally more). Each vial contains 20 mg — reconstitute with 60 mL sterile water (Ryanodex formulation: 250 mg/vial, reconstitute with 5 mL). 5. Active cooling: cold IV saline, ice packs to axillae/groin/neck, forced cold air, lavage if needed. Stop cooling at 38°C to avoid overshoot. 6. Treat hyperkalemia: insulin + glucose, calcium chloride/gluconate, sodium bicarbonate, hyperventilation. 7. Monitor: serial ABGs, electrolytes, CK (peaks at 12–24 h), myoglobin, coagulation, urine output. 8. Maintain urine output >2 mL/kg/h (IV fluids, mannitol/furosemide if needed) to prevent myoglobin-induced renal failure. 9. Convert to TIVA for remainder of case (propofol, opioids, non-depolarizing NMBA). 10. ICU admission for ≥24 hours. Continue dantrolene 1 mg/kg IV q4–6h for 24–48 hours (recrudescence occurs in ~25%).
Safe Anesthesia for MH-Susceptible Patients
For patients with known or suspected MH susceptibility: avoid all triggering agents (volatile anesthetics and succinylcholine). Use TIVA (propofol + remifentanil or fentanyl), non-depolarizing NMBAs (rocuronium/cisatracurium), benzodiazepines, opioids, and regional/neuraxial anesthesia as appropriate. N₂O is safe (not a trigger). Prepare the anesthesia machine: flush with high-flow O₂ (≥10 L/min) for ≥20 minutes with fresh circuit and soda lime (or use a "clean" machine with activated charcoal filters), remove or disable vaporizers. Ensure dantrolene is immediately available (36 vials of standard dantrolene or 3 vials of Ryanodex). Monitor temperature and ETCO₂ continuously. Amide local anesthetics are safe (they do not trigger MH despite historical concern). Post-anesthesia: monitor in PACU for at least 1 hour; extended observation is not strictly required if the procedure was short and the patient is stable.
Diagnosis & Genetic Testing
The gold standard diagnostic test is the caffeine-halothane contracture test (CHCT), performed on a fresh muscle biopsy (vastus lateralis). Muscle strips are exposed to halothane and caffeine; MH-susceptible muscle demonstrates enhanced contracture. Sensitivity ~97%, specificity ~78%. Genetic testing for RYR1 and CACNA1S mutations is available but a negative genetic test does not exclude MH susceptibility (not all causative mutations are known). First-degree relatives of confirmed MH patients should be considered susceptible until tested.
22 Anaphylaxis in the OR
Common Triggers
Perioperative anaphylaxis occurs in approximately 1:10,000–1:20,000 anesthetics. The most common triggers are: neuromuscular blocking agents (~60% of cases — particularly rocuronium and succinylcholine, due to quaternary ammonium cross-reactivity), antibiotics (especially beta-lactams/cephalosporins given at induction), latex (declining with latex-free environments but still relevant), chlorhexidine (skin prep, CVC impregnation), colloids (gelatin, dextran), blood products, dyes (methylene blue, patent blue, isosulfan blue), and protamine.
Clinical Grading
| Grade | Severity | Features |
|---|---|---|
| I | Cutaneous only | Urticaria, erythema, flushing, angioedema |
| II | Moderate multi-organ | Cutaneous signs + hypotension (responsive to fluids), tachycardia, bronchospasm (mild) |
| III | Severe / life-threatening | Cardiovascular collapse (severe hypotension, tachycardia or bradycardia), severe bronchospasm, cutaneous signs may be absent (blood diverted from skin) |
| IV | Cardiac arrest | Cardiac arrest (PEA, VF, asystole) |
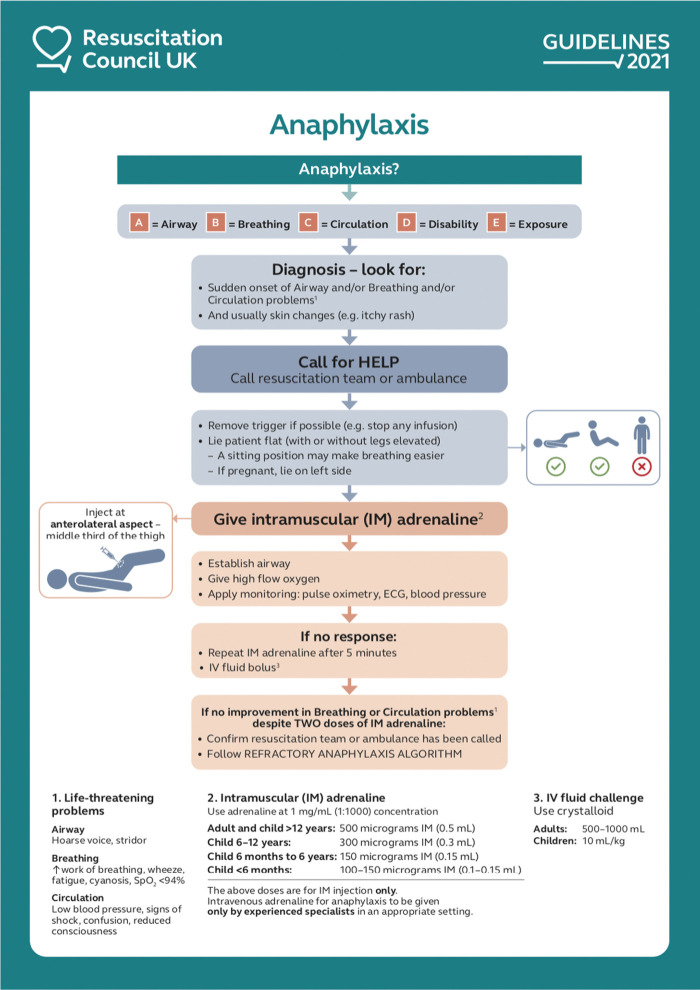
Immediate Management
1. Stop the suspected trigger (discontinue antibiotic infusion, remove latex gloves, etc.). 2. Call for help. 3. Administer 100% FiO₂. 4. Epinephrine — the cornerstone of treatment: Grade II: 10–20 mcg IV bolus (dilute to 10 mcg/mL); Grade III: 100–200 mcg IV bolus, repeat every 1–2 minutes; Grade IV: 1 mg IV (cardiac arrest dose) + ACLS. Start epinephrine infusion (0.05–0.5 mcg/kg/min) for refractory hypotension. 5. Volume resuscitation: rapid crystalloid bolus 20–30 mL/kg (massive vasodilation causes relative hypovolemia). 6. Bronchodilators: albuterol for bronchospasm refractory to epinephrine. 7. Secondary treatments: hydrocortisone 200 mg IV (prevents biphasic reaction — takes 4–6 h for effect), diphenhydramine 50 mg IV (H1 blocker), ranitidine or famotidine (H2 blocker). 8. Vasopressin 1–2 units IV bolus for epinephrine-refractory shock. Norepinephrine infusion as second-line vasopressor.

Differential Diagnosis of Intraoperative Anaphylaxis
Not all hemodynamic collapse during anesthesia is anaphylaxis. The differential includes: non-immune anaphylactoid reaction (direct mast cell degranulation without IgE — e.g., morphine, vancomycin "red man syndrome"), vasovagal response, high spinal, tension pneumothorax, air embolism, acute blood loss, cardiogenic shock, transfusion reaction, and propofol infusion-related hypotension. Bronchospasm without hypotension may represent reactive airway disease triggered by airway manipulation rather than anaphylaxis. The triad of hypotension + bronchospasm + cutaneous findings is most specific for anaphylaxis. In the anesthetized, draped patient, cutaneous signs (urticaria, flushing) may be hidden or absent, making tryptase levels essential for retrospective diagnosis.
Post-Event Workup
Draw serum tryptase levels at 3 time points: immediately when feasible, at 1–2 hours after symptom onset (peak), and at 24 hours (baseline). An elevated peak tryptase (>11.4 ng/mL or >2 + 1.2 × baseline) supports the diagnosis of mast cell degranulation. Refer the patient to an allergist for skin testing (intradermal testing with suspected agents, ideally at 4–6 weeks post-event) and specific IgE measurement. Results guide future anesthetic planning and drug avoidance.
23 Perioperative Cardiac Events
Intraoperative Myocardial Ischemia / Infarction
Intraoperative STEMI/NSTEMI may present as new ST elevation/depression ≥1 mm on ECG monitor, new regional wall motion abnormality on TEE, hemodynamic instability (hypotension, new arrhythmia), or elevated troponin postoperatively. Management: optimize hemodynamics (correct hypotension and tachycardia, maintain CPP), administer nitroglycerin for ischemia (avoid if hypotensive), heparin (if surgical bleeding is acceptable), consider emergent PCI if STEMI, and consult cardiology. Aspirin and dual antiplatelet therapy decisions are balanced against surgical bleeding risk.
Bradycardia
Intraoperative bradycardia (HR <50 bpm with hemodynamic compromise): common causes include high spinal/epidural, vagal stimulation (laryngoscopy, surgical manipulation of peritoneum/eye), opioids, beta-blockers, dexmedetomidine. Treatment ladder: atropine 0.5–1 mg IV (muscarinic antagonist — first-line), glycopyrrolate 0.2–0.4 mg IV (does not cross BBB), ephedrine 5–10 mg IV (indirect sympathomimetic, increases HR and BP), epinephrine 10–100 mcg IV for severe bradycardia, transcutaneous/transvenous pacing for refractory cases.
Intraoperative Hypertension
Intraoperative hypertension (SBP >20% above baseline) is common and has multiple causes: inadequate anesthesia depth (most common), pain/noxious stimulation, hypercarbia, hypoxemia, full bladder, medications (ketamine, vasopressors, ergot alkaloids), pheochromocytoma, aortic cross-clamping, malignant hyperthermia, and pre-existing essential hypertension. Management: first address the cause (deepen anesthesia, increase opioid, improve ventilation); pharmacologic options include increasing volatile agent concentration, labetalol 5–20 mg IV (combined alpha/beta blockade), esmolol 0.5–1 mg/kg bolus then 50–200 mcg/kg/min infusion (ultra-short-acting beta-1 selective), hydralazine 5–10 mg IV (direct vasodilator, slower onset 15 min), nicardipine infusion 2.5–15 mg/h (calcium channel blocker, smooth titratable vasodilation), or sodium nitroprusside 0.25–10 mcg/kg/min (potent, rapid, but risk of cyanide toxicity and rebound hypertension). Clevidipine (Cleviprex) is an ultra-short-acting IV calcium channel blocker with a 1-minute half-life, ideal for precise BP control.
Acute Pulmonary Embolism
Presents intraoperatively as sudden hemodynamic collapse (hypotension, tachycardia), acute right heart failure (elevated CVP, RV dilation on TEE), increased dead space (sudden drop in ETCO₂ with stable or increasing PaCO₂), and hypoxemia. Treatment: 100% FiO₂, volume resuscitation, vasopressors (norepinephrine, vasopressin), consider thrombolytics (alteplase 50–100 mg) if massive PE with hemodynamic collapse despite vasopressors (weigh against surgical bleeding), heparin bolus and infusion, inotropes (dobutamine/milrinone for RV failure), emergent surgical embolectomy or catheter-directed therapy in refractory cases.
Cardiac Arrest in the OR — ACLS Modifications
Advantages: the patient is already monitored, has IV access, and an airway is established. Modifications: Immediately switch to 100% FiO₂, turn off volatile agents, consider reversible causes specific to OR: anaphylaxis, MH, hyperkalemia (succinylcholine), LAST, tension pneumothorax (laparoscopic insufflation, central line), air embolism, massive hemorrhage, vagal reflex. Standard ACLS otherwise: epinephrine 1 mg q3–5min, defibrillation for VF/pVT, amiodarone 300 mg then 150 mg. If the patient is prone: begin chest compressions in prone position (counterpressure on sternum) while rapidly turning supine.
Tension Pneumothorax
Can occur from central line placement (especially subclavian), positive-pressure ventilation with barotrauma, or laparoscopic surgery. Signs: sudden hypotension, increased peak airway pressures, decreased breath sounds (unilateral), tracheal deviation (late). Treatment: immediate needle decompression (14G needle, 2nd intercostal space midclavicular line or 5th ICS anterior axillary line) followed by chest tube insertion.
Local Anesthetic Systemic Toxicity (LAST) in the OR
LAST can mimic other perioperative emergencies and must be in the differential for any seizure, arrhythmia, or cardiovascular collapse occurring within 60 minutes of local anesthetic administration (though delayed presentations up to 30 minutes after injection are well-documented). Under general anesthesia, the CNS prodromal symptoms (tinnitus, perioral numbness, metallic taste) are masked, and cardiac toxicity may be the first manifestation. Bupivacaine has the narrowest CC/CNS ratio (ratio of the dose causing cardiovascular collapse to the dose causing seizures) of any commonly used LA, meaning cardiac arrest may occur with little warning. Ropivacaine and levobupivacaine have wider safety margins. Always aspirate before injecting LA, use incremental dosing with epinephrine markers, observe for signs of intravascular injection (tachycardia with epinephrine-containing test dose), and keep intralipid 20% immediately available whenever performing regional anesthesia.
Venous Air Embolism
Risk during sitting craniotomy, posterior cervical spine surgery, central line placement/removal, laparoscopic surgery, and any procedure where the surgical site is above the heart (entrains air through open veins). The lethal volume in adults is approximately 3–5 mL/kg. Signs: sudden decrease in ETCO₂, hypotension, "mill wheel" murmur, desaturation. Treatment: flood surgical field with saline, lower the surgical site below the heart, aspirate air through CVC (if tip is at SVC-RA junction), Durant maneuver (left lateral decubitus and Trendelenburg position to trap air in the RA apex away from the RV outflow tract), hemodynamic support, consider hyperbaric oxygen for massive embolism.
24 Acute Postoperative Pain
Multimodal Analgesia
Modern postoperative pain management uses a multimodal approach — combining agents with different mechanisms to optimize analgesia while minimizing opioid consumption and side effects. This is a core principle of Enhanced Recovery After Surgery (ERAS) protocols.
| Agent / Class | Dose | Mechanism | Pearl |
|---|---|---|---|
| Acetaminophen | 1 g IV/PO q6h (max 4 g/day; 2 g/day if hepatic impairment) | Central COX inhibition, serotonergic pathways | Opioid-sparing effect 20–30%; schedule around the clock, not PRN; IV onset 5–10 min |
| NSAIDs (ketorolac, ibuprofen, celecoxib) | Ketorolac 15–30 mg IV q6h (max 5 days); celecoxib 200–400 mg PO | COX-1/COX-2 inhibition → decreased prostaglandin synthesis | Avoid in renal insufficiency, bleeding risk (ketorolac), GI ulcer history; COX-2 selective (celecoxib) has less bleeding risk; opioid-sparing 30–50% |
| Gabapentinoids (gabapentin, pregabalin) | Gabapentin 300–600 mg PO preoperatively; pregabalin 75–150 mg | α2δ calcium channel subunit binding → decreased excitatory neurotransmitter release | Reduces opioid consumption and neuropathic pain; sedation/dizziness risk, especially in elderly; avoid with OSA |
| Ketamine (sub-anesthetic) | 0.1–0.5 mg/kg IV bolus; infusion 0.1–0.2 mg/kg/h | NMDA receptor antagonist | Reduces central sensitization and opioid tolerance; particularly useful in opioid-tolerant patients and chronic pain; avoid in psychosis history |
| Dexmedetomidine | 0.2–0.7 mcg/kg/h IV infusion | α2-adrenergic agonist | Analgesic, anxiolytic, opioid-sparing, no respiratory depression; risk of bradycardia and hypotension |
| IV lidocaine | Bolus 1–1.5 mg/kg, infusion 1–2 mg/kg/h | Systemic Na+ channel blockade, anti-inflammatory | Particularly beneficial for abdominal surgery; monitor for LAST signs; stop before discharge or transition to oral mexiletine |
IV Patient-Controlled Analgesia (PCA)
| Opioid | Demand Dose | Lockout Interval | 4-Hour Limit | Basal Rate |
|---|---|---|---|---|
| Morphine | 1–2 mg | 6–10 min | 20–30 mg | 0–1 mg/h (optional, increases respiratory depression risk) |
| Hydromorphone | 0.2–0.4 mg | 6–10 min | 4–6 mg | 0–0.2 mg/h (optional) |
| Fentanyl | 10–25 mcg | 6–10 min | 200–400 mcg | 0–25 mcg/h (optional) |
Opioid-Sparing Strategies
The opioid epidemic has driven a paradigm shift toward minimizing perioperative opioid use. Key strategies include: preemptive analgesia (administering analgesics before surgical stimulus to prevent central sensitization), regional anesthesia (peripheral nerve blocks and neuraxial techniques as primary analgesic modality), scheduled non-opioid analgesics (acetaminophen + NSAID scheduled around the clock, not PRN), IV lidocaine infusion (particularly effective for abdominal surgery), dexmedetomidine (alpha-2 agonist with analgesic, sedative, and opioid-sparing properties without respiratory depression), magnesium sulfate (NMDA antagonist, 30–50 mg/kg loading dose followed by 8–15 mg/kg/h infusion — reduces postoperative opioid consumption by 25%), and ketamine low-dose infusion (0.1–0.3 mg/kg/h intraoperatively, continued 24–48 h postoperatively in opioid-tolerant patients). These strategies collectively can reduce opioid consumption by 40–60% and shorten recovery time.
ERAS (Enhanced Recovery After Surgery) Pain Principles
ERAS protocols emphasize: preoperative education, preoperative multimodal analgesia (acetaminophen + gabapentinoid + celecoxib), regional anesthesia whenever possible, minimize intraoperative opioids (use short-acting agents like remifentanil), scheduled postoperative non-opioid analgesics, early mobilization, and early enteral nutrition. ERAS programs have been shown to reduce opioid consumption by 30–50%, shorten hospital length of stay, reduce complications, and improve patient satisfaction.
25 Chronic Pain Fundamentals
Pain Classification
Nociceptive pain results from activation of peripheral nociceptors by tissue damage (somatic = well-localized, sharp/aching from skin, muscle, bone; visceral = diffuse, cramping from internal organs). Neuropathic pain arises from damage or dysfunction of the nervous system itself (peripheral neuropathy, post-herpetic neuralgia, phantom limb, CRPS). Characterized by burning, shooting, electric-shock quality, allodynia (pain from normally non-painful stimulus), and hyperalgesia (exaggerated pain response). Nociplastic pain (IASP 2017) arises from altered nociception without clear evidence of tissue or nerve damage (fibromyalgia, irritable bowel syndrome, chronic widespread pain, tension-type headache). Driven by central sensitization and altered descending modulation.
Complex Regional Pain Syndrome (CRPS)
CRPS is a chronic pain condition characterized by pain disproportionate to the inciting event, accompanied by sensory, motor, sudomotor, vasomotor, and trophic changes. Type I (formerly reflex sympathetic dystrophy): no identifiable nerve injury; typically follows trauma, fracture, or surgery. Type II (formerly causalgia): identifiable peripheral nerve injury. Diagnosis requires the Budapest criteria: (1) continuing pain disproportionate to inciting event, (2) at least one sign in 2+ categories AND one symptom in 3+ categories (sensory: hyperesthesia/allodynia; vasomotor: temperature/color asymmetry; sudomotor/edema: asymmetric sweating/edema; motor/trophic: decreased ROM, weakness, dystonia, skin/nail/hair changes), (3) no other diagnosis better explains findings. Treatment: physical therapy (cornerstone), pharmacotherapy (gabapentinoids, duloxetine, corticosteroids in acute phase), sympathetic nerve blocks (stellate ganglion for upper extremity, lumbar sympathetic for lower extremity), spinal cord stimulation, and psychological support.
Central Sensitization & Gate Control Theory
Central sensitization is increased excitability of central neurons in the dorsal horn and brain, leading to amplified pain signals (hyperalgesia, allodynia, expanded receptive fields). Mediated by NMDA receptor activation, wind-up phenomenon, and glial cell activation. Gate control theory (Melzack & Wall, 1965): pain perception is modulated by a "gate" in the dorsal horn of the spinal cord. Large-diameter Aβ fibers (touch, vibration) activate inhibitory interneurons that "close the gate," reducing pain transmission by small C and Aδ fibers. This is the theoretical basis for TENS units, spinal cord stimulators, and rubbing a painful area.
Pain Assessment Tools
| Tool | Description | Use |
|---|---|---|
| Numeric Rating Scale (NRS) | 0 (no pain) to 10 (worst imaginable) | Most widely used in adults; quick, reproducible |
| Visual Analog Scale (VAS) | 100 mm line; patient marks pain level | Research standard; more sensitive than NRS |
| Wong-Baker FACES | 6 faces from smiling to crying | Pediatric (≥3 years), cognitively impaired adults |
| FLACC (Face, Legs, Activity, Cry, Consolability) | Behavioral observation scale 0–10 | Preverbal children, non-communicative patients |
| McGill Pain Questionnaire | 78 descriptors in sensory, affective, evaluative categories | Comprehensive pain characterization; research |
WHO Cancer Pain Ladder (3-Step)
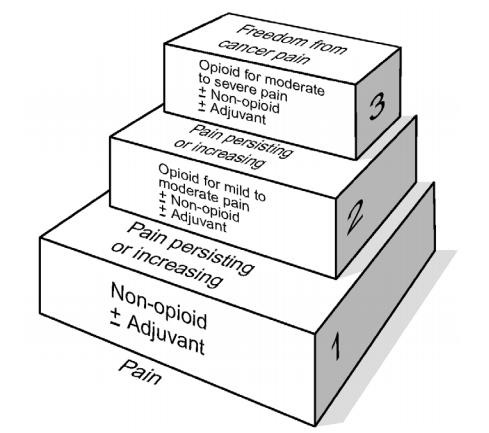
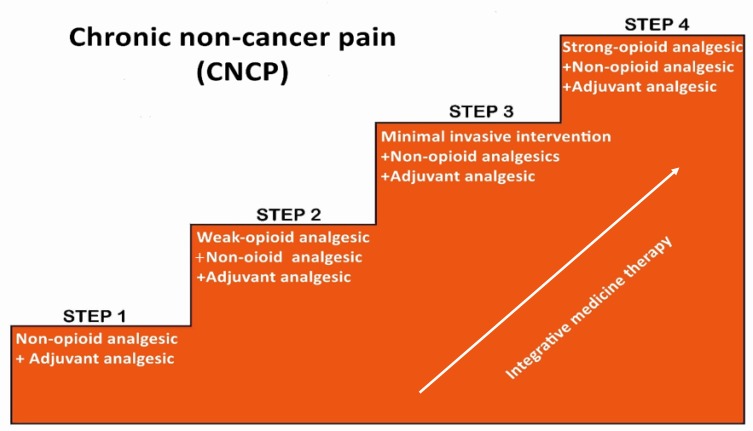
Step 1 (mild pain): non-opioid analgesics (acetaminophen, NSAIDs) ± adjuvants. Step 2 (moderate pain): weak opioid (codeine, tramadol) + non-opioid ± adjuvants. Step 3 (severe pain): strong opioid (morphine, oxycodone, fentanyl, hydromorphone, methadone) + non-opioid ± adjuvants. Adjuvants at any step: antidepressants (duloxetine, amitriptyline for neuropathic pain), anticonvulsants (gabapentin, pregabalin), corticosteroids (bone metastases, neural compression), bisphosphonates, ketamine.
Neuropathic Pain Pharmacotherapy
First-line agents for neuropathic pain include: gabapentin (start 300 mg QHS, titrate to 300–1200 mg TID; NNT ~6 for 50% pain relief), pregabalin (start 75 mg BID, titrate to 150–300 mg BID; NNT ~4; faster onset than gabapentin, linear pharmacokinetics), duloxetine (30–60 mg daily; SNRI, effective for diabetic neuropathy, fibromyalgia, musculoskeletal pain; NNT ~6), tricyclic antidepressants (amitriptyline/nortriptyline 10–25 mg QHS, titrate to 75–150 mg; NNT ~4 — most effective but limited by anticholinergic side effects, sedation, cardiac conduction risk). Second-line: topical lidocaine 5% patch (for localized neuropathic pain, minimal systemic absorption), capsaicin 8% patch (applied in clinic for 30–60 minutes, depletes substance P; effective for post-herpetic neuralgia; reapply every 3 months), tramadol, and opioids (reserved for refractory cases due to risks of tolerance, dependence, and hyperalgesia).
Opioid Conversion & Rotation
When switching between opioids (rotation), calculate the total 24-hour morphine equivalent dose, convert to the new agent using equianalgesic tables, then reduce the calculated dose by 25–50% to account for incomplete cross-tolerance. Methadone has unique pharmacology: dual mechanism (mu-agonist + NMDA antagonist), extremely variable and long half-life (8–59 hours, mean 22 h), high oral bioavailability (~80%), QTc prolongation risk, and unpredictable conversion ratios (the higher the morphine equivalent dose, the more potent methadone is relatively — for morphine >500 mg/day, the ratio may be 12:1 or higher). Methadone conversions should only be performed by experienced clinicians.
26 Interventional Pain Procedures
Anticoagulation & Neuraxial/Regional Anesthesia Timing
The ASRA (American Society of Regional Anesthesia and Pain Medicine) guidelines provide evidence-based timing recommendations for neuraxial and deep peripheral blocks relative to anticoagulant administration:
| Anticoagulant | Time Before Procedure | Time After Catheter Removal to Restart |
|---|---|---|
| Unfractionated heparin (SQ prophylactic) | 4–6 hours; check aPTT | 1 hour |
| LMWH (prophylactic) | 12 hours | 4 hours |
| LMWH (therapeutic) | 24 hours | 4 hours |
| Warfarin | 5 days; INR ≤1.4 | After catheter removal |
| Rivaroxaban | 72 hours | 6 hours |
| Apixaban | 72 hours | 6 hours |
| Dabigatran (CrCl >50) | 72 hours | 6 hours |
| Clopidogrel | 5–7 days | After catheter removal |
| Ticagrelor | 5–7 days | 6 hours after catheter removal |
| Aspirin (low-dose 81 mg) | No hold required for neuraxial | N/A |
Epidural Steroid Injection (ESI)
Used for radicular pain from disc herniation, spinal stenosis, or degenerative disease. Interlaminar ESI: needle enters the epidural space between laminae using loss of resistance — deposits medication in the posterior epidural space. Transforaminal ESI: fluoroscopy-guided needle enters the neural foramen, depositing medication in the anterior epidural space adjacent to the nerve root — more targeted, higher efficacy for unilateral radiculopathy, but carries risk of particulate steroid injection into the radicular artery (use dexamethasone, a non-particulate steroid, for cervical and some lumbar transforaminal injections to minimize stroke/paralysis risk). Typical agents: dexamethasone 8–10 mg or methylprednisolone 40–80 mg + preservative-free lidocaine or bupivacaine.
Facet Joint Procedures
Medial branch blocks (MBB): diagnostic blocks of the medial branches of the dorsal rami that innervate the facet (zygapophyseal) joints. Two concordant positive blocks (≥80% pain relief) confirm the facet joint as the pain generator. Each facet is innervated by the medial branch at that level and the level above (e.g., L4-5 facet is innervated by L3 and L4 medial branches). Radiofrequency ablation (RFA): thermal lesioning of the medial branch nerve using a radiofrequency probe heated to 80°C for 60–90 seconds. Provides 6–12 months of relief (nerve regenerates). Performed after two positive diagnostic MBBs.
Sacroiliac (SI) Joint Injection
Fluoroscopy- or ultrasound-guided injection of local anesthetic and corticosteroid into the SI joint for sacroiliac joint dysfunction (confirmed by provocation tests: FABER/Patrick, distraction, compression, thigh thrust, Gaenslen). If positive diagnostic block, options include intra-articular steroid, lateral branch RFA, or SI joint fusion for refractory cases.
Regenerative & Emerging Therapies
Platelet-rich plasma (PRP) injections concentrate autologous growth factors (PDGF, TGF-β, VEGF) for injection into tendons, ligaments, and joints. Evidence is strongest for lateral epicondylitis and knee osteoarthritis; evidence for spine pain is limited. Prolotherapy involves injection of dextrose (hyperosmolar solution) to provoke a localized inflammatory response and stimulate tissue repair at ligament/tendon insertions. Peripheral nerve stimulation (PNS) is gaining traction as an alternative to SCS for focal pain conditions (e.g., occipital neuralgia with occipital nerve stimulation, knee pain with genicular nerve stimulation). Advantages include less invasive implant and lower complication rates than SCS.
Spinal Cord Stimulation (SCS)
Spinal cord stimulation involves implanting epidural electrodes connected to a pulse generator that delivers electrical impulses to the dorsal columns. Mechanism: gate control theory — stimulation of large Aβ fibers in the dorsal columns inhibits pain transmission. Also modulates descending inhibitory pathways and neurochemical mediators. Indications: failed back surgery syndrome (most common), CRPS types I and II, peripheral neuropathy, refractory angina, peripheral vascular disease pain. Trial period (5–7 days with temporary percutaneous leads) required before permanent implant; success = ≥50% pain reduction. Modern waveforms: traditional tonic, burst, high-frequency (10 kHz — paresthesia-free), and closed-loop systems.
Intrathecal Drug Delivery Systems (IDDS)
Implanted pump delivers medication (morphine, ziconotide, bupivacaine, baclofen) directly into the intrathecal space. Provides potent analgesia at 1/300th of the systemic dose (intrathecal-to-oral morphine ratio is approximately 1:300). Indications: cancer pain refractory to systemic opioids, severe spasticity (baclofen), and selected non-cancer chronic pain. Requires MRI-compatible pump, regular refills (every 1–6 months), and monitoring for catheter-related complications (granuloma, migration, fracture).
Joint Injections & Viscosupplementation
Intra-articular corticosteroid injections are commonly performed for osteoarthritis and inflammatory arthritis. Typical agents: triamcinolone acetonide 40–80 mg (knee) or methylprednisolone 40–80 mg, mixed with 2–5 mL of 1% lidocaine. Fluoroscopy or ultrasound guidance improves accuracy, particularly for hip and shoulder injections. Limit frequency to 3–4 injections per joint per year (concern for accelerated cartilage degeneration with repeated injections). Patients scheduled for total joint arthroplasty should avoid corticosteroid injection within 3 months of surgery due to increased infection risk. Viscosupplementation (hyaluronic acid injections) provides lubrication and may offer 3–6 months of relief in knee OA. Evidence is mixed, with most guidelines providing a conditional recommendation.
Other Interventional Procedures
Trigger point injections (TPI): injection of local anesthetic (with or without corticosteroid) or dry needling into myofascial trigger points (taut bands of skeletal muscle). Stellate ganglion block: injection of local anesthetic at C6–C7 targeting the cervicothoracic sympathetic ganglion. Indications: CRPS type I of the upper extremity, phantom limb pain, vascular insufficiency of the upper extremity. Confirm by development of ipsilateral Horner syndrome. Complications: recurrent laryngeal nerve block, vertebral artery injection, pneumothorax, esophageal puncture. Celiac plexus block/neurolysis: injection of local anesthetic (block) or alcohol/phenol (neurolysis) at the celiac plexus (T12–L1, anterior to the aorta). Primary indication: upper abdominal cancer pain (pancreatic cancer). Neurolysis provides pain relief in 70–90% of pancreatic cancer patients. Side effects: orthostatic hypotension, diarrhea (sympatholysis).
27 Obstetric Anesthesia
Physiologic Changes of Pregnancy
Pregnancy produces profound physiologic changes relevant to anesthesia:
| System | Change | Anesthetic Implication |
|---|---|---|
| Respiratory | Decreased FRC (20%), increased oxygen consumption (20%), increased minute ventilation (50%), decreased PaCO₂ to 30 mmHg | Rapid desaturation during apnea; reduced safe apnea time (from ~6 min to ~3 min); difficult preoxygenation in morbid obesity |
| Cardiovascular | Increased CO (40%), increased blood volume (45%), decreased SVR, aortocaval compression by gravid uterus in supine position | Left uterine displacement essential after 20 weeks (prevents supine hypotension syndrome); increased sensitivity to vasopressors |
| GI | Decreased LES tone, increased intragastric pressure, delayed gastric emptying (especially in labor) | Full stomach precautions from 2nd trimester; RSI for GA; consider ranitidine + sodium citrate (Bicitra) preoperatively |
| Neuraxial | Engorged epidural veins (reduced epidural space volume), increased CSF sensitivity to local anesthetics | Decreased local anesthetic dose requirements (30% less for spinal); increased risk of epidural venous puncture and intravascular catheter |
| Airway | Mucosal edema, capillary engorgement, Mallampati score worsens during labor, weight gain | Use smaller ETT (6.0–6.5); anticipate more difficult intubation; 8× higher rate of failed intubation than general population |
| MAC | Decreased by ~30% | Reduced volatile anesthetic requirements |
Labor Epidural Analgesia
Initiation: typically at patient request when active labor is established. Lumbar epidural catheter at L2–L3 or L3–L4. Test dose to exclude intrathecal or intravascular placement. Loading dose: bupivacaine 0.0625–0.125% + fentanyl 50–100 mcg in 10–15 mL. PCEA maintenance: bupivacaine 0.0625–0.1% + fentanyl 2 mcg/mL; basal rate 6–10 mL/h, demand 5–8 mL, lockout 10–20 min. Target: T10–L1 for labor pain (uterine contractions); extend to S2–S4 for second stage (perineal pain). Monitor for hypotension (treat with phenylephrine or ephedrine + fluid), fetal heart rate changes, pruritis (fentanyl-related, treat with nalbuphine 2.5–5 mg), and motor block (reduce concentration if excessive).
Spinal Anesthesia for Cesarean Section
Standard regimen: hyperbaric bupivacaine 0.75% (10–12 mg) + fentanyl 10–25 mcg + preservative-free morphine 0.1–0.2 mg (for postoperative analgesia up to 24 hours). Target dermatome level: T4. Hypotension is the most common complication (up to 80% without prophylaxis); prevention: left uterine displacement, crystalloid/colloid co-loading, prophylactic phenylephrine infusion (0.25–0.5 mcg/kg/min, titrated to maintain BP within 20% of baseline).
General Anesthesia for Emergent C-Section
Indicated when neuraxial anesthesia is contraindicated or insufficient and delivery is urgent. RSI technique with preoxygenation, cricoid pressure, propofol 2–2.5 mg/kg + succinylcholine 1–1.5 mg/kg (or rocuronium 1.2 mg/kg with sugammadex available). Maintain with sevoflurane or desflurane 1.0–1.5 MAC + N₂O/O₂ (50:50) or 100% O₂. After delivery: deepen anesthesia, add opioid, reduce volatile to 0.5–0.7 MAC, administer oxytocin. Keep induction-to-delivery time <8 minutes to minimize fetal drug exposure.
Postpartum Hemorrhage & Anesthetic Considerations
PPH is the leading cause of maternal mortality worldwide. Defined as blood loss ≥1000 mL with signs/symptoms of hypovolemia. The anesthesiologist's role includes: ensuring large-bore IV access (14–16G × 2), type and screen (or crossmatch), activating MTP early, maintaining hemodynamic stability with vasopressors and blood products, converting from neuraxial to general anesthesia if surgical intervention is needed (hemorrhage worsening under epidural anesthesia), and monitoring for coagulopathy (TEG/ROTEM-guided transfusion if available). Tranexamic acid 1 g IV within 3 hours of delivery reduces death from bleeding (WOMAN trial). The RCOG guideline recommends a stepwise approach to uterotonic therapy:
Uterotonic Medications
| Agent | Dose/Route | Mechanism | Contraindication/Side Effect |
|---|---|---|---|
| Oxytocin (Pitocin) | 10–40 units in 1 L LR IV infusion; or 3–5 units IV bolus (slow) | Oxytocin receptor agonist | Rapid IV bolus → hypotension, tachycardia; water intoxication with high doses (ADH-like effect) |
| Methylergonovine (Methergine) | 0.2 mg IM | Ergot alkaloid — sustained uterine contraction | Contraindicated in hypertension, preeclampsia (causes severe vasoconstriction/hypertensive crisis) |
| Carboprost (Hemabate) | 0.25 mg IM q15min (max 8 doses) | PGF2α analog | Contraindicated in asthma (bronchospasm); causes diarrhea, fever, nausea |
| Misoprostol (Cytotec) | 600–1000 mcg PR/SL | PGE1 analog | Fever, diarrhea; no absolute contraindications; used when other agents fail |
28 Pediatric Anesthesia
Weight-Based Dosing & Equipment Sizing
Pediatric drug dosing is universally weight-based (mg/kg or mcg/kg). ETT sizing: cuffed ETT (now preferred even in young children): internal diameter (mm) = (age in years / 4) + 3.5 for cuffed tubes. For uncuffed ETT (historically used in children <8 years): (age / 4) + 4. ETT depth at the lip (cm): ETT ID × 3 (e.g., 4.0 tube inserted to 12 cm). Cuffed ETTs are now standard even in neonates because they reduce gas leak, improve ventilation, decrease reintubation rates, and reduce waste gas pollution; cuff pressure must be monitored (<20–25 cmH₂O).
| Age | Weight (approx) | ETT Size (cuffed) | LMA Size | Defibrillation (J/kg) |
|---|---|---|---|---|
| Premature | 1–2.5 kg | 2.5–3.0 uncuffed | 1 | 2 J/kg |
| Term neonate | 3–4 kg | 3.0–3.5 | 1 | 2 J/kg |
| 6 months | 7 kg | 3.0–3.5 | 1.5 | 2 J/kg |
| 1 year | 10 kg | 3.5 | 1.5–2 | 2 J/kg |
| 4 years | 16 kg | 4.0–4.5 | 2 | 2 J/kg |
| 8 years | 25 kg | 5.0–5.5 | 2.5 | 2 J/kg |
| 12 years | 40 kg | 5.5–6.0 | 3 | 2 J/kg |
IV Access Challenges in Pediatrics
Peripheral IV access is the most common technical challenge in pediatric anesthesia. For patients without IV access, inhalation induction with sevoflurane is the standard approach. Once unconscious, IV access is obtained. If peripheral IV is impossible: options include intraosseous (IO) access (proximal tibia, distal femur, or distal tibia — 10–15 mL flush to clear marrow), external jugular vein cannulation, or central venous access (femoral preferred in emergencies). Ultrasound guidance significantly improves first-attempt success rates for peripheral and central access in children. Topical anesthetic cream (EMLA or LMX 4%) applied 30–60 minutes before reduces pain of IV insertion in awake children and should be offered whenever time permits.
Inhalation Induction
Inhalation induction with sevoflurane (up to 8% in 70% N₂O/30% O₂ or 100% O₂) is the standard technique for children without IV access. Sevoflurane's low blood-gas partition coefficient (0.65) allows rapid induction (loss of consciousness in 1–2 minutes). Incremental technique (starting at 2% and increasing by 1% every few breaths) or single-breath technique (vital capacity breath of 8%) can be used. Once unconscious, establish IV access and proceed with intubation or SGA placement.
Laryngospasm
Laryngospasm is involuntary sustained closure of the vocal cords, causing complete airway obstruction. Most common in children (especially ages 1–5), particularly during light anesthesia, airway manipulation, secretions, blood, or irritation. Incidence: 1–3% in children. Management: immediate CPAP with 100% O₂ and jaw thrust (gentle positive pressure may open the cords), deepen anesthesia with propofol 0.5–1 mg/kg if IV available, and if refractory: succinylcholine 0.5–1 mg/kg IV (or 4 mg/kg IM if no IV access) to break the spasm. Larson's maneuver: firm bilateral digital pressure in the "laryngospasm notch" (behind the lobule of the ear, between the ascending ramus of the mandible and the mastoid process) — causes a pain-mediated jaw thrust that may relieve spasm.
Temperature Regulation
Infants and neonates are extremely vulnerable to hypothermia due to large body surface area-to-weight ratio, thin skin, minimal subcutaneous fat, and immature thermoregulatory mechanisms. Hypothermia causes increased oxygen consumption, metabolic acidosis, delayed drug metabolism, and coagulopathy. Prevention: warm OR (26°C for neonates), forced-air warming blanket, heated humidified circuit gases, fluid warmer, minimal skin exposure.
Emergence Delirium
Emergence delirium (ED) is a dissociated state of consciousness with inconsolable crying, thrashing, and disorientation during emergence from general anesthesia. Incidence: 10–50% in preschool-age children (peak age 2–5 years). Risk factors: sevoflurane or desflurane anesthesia, young age, preoperative anxiety, ENT/ophthalmologic procedures. Prevention: midazolam premedication, IV propofol at emergence (1 mg/kg), fentanyl 1–2 mcg/kg, dexmedetomidine 0.3–0.5 mcg/kg, or TIVA. Treatment: ensure pain is adequately treated (pain and ED are easily confused), low-dose propofol, parental presence, calm environment.
Common Pediatric Anesthetic Drug Doses
| Drug | Pediatric Dose | Notes |
|---|---|---|
| Propofol | 2.5–3.5 mg/kg IV (higher than adults) | Higher Vd in children; may cause pain on injection |
| Succinylcholine | 2 mg/kg IV; 4 mg/kg IM | Always pretreat with atropine 0.02 mg/kg (min 0.1 mg) in children <5 years |
| Rocuronium | 0.6–1.0 mg/kg | Faster onset in children due to higher CO relative to muscle mass |
| Fentanyl | 1–3 mcg/kg | Neonates more sensitive; reduce dose in <3 months |
| Atropine | 0.02 mg/kg IV (min 0.1 mg, max 0.5 mg) | Must have available before any pediatric induction |
| Dexamethasone | 0.15–0.5 mg/kg (max 10 mg) | Antiemetic and reduces airway edema post-intubation |
| Ondansetron | 0.1–0.15 mg/kg (max 4 mg) | PONV prophylaxis; children have higher PONV rates |
Neonatal Considerations
Neonates have transitional circulation (patent foramen ovale, ductus arteriosus may remain open, especially in premature infants) which can cause right-to-left shunting with hypoxia, hypercarbia, or acidosis — maintaining normal pH, temperature, and oxygenation is critical to prevent persistent pulmonary hypertension of the newborn (PPHN). Premature infants are at risk for post-anesthetic apnea (apnea/bradycardia for up to 24 hours after general anesthesia in infants <60 weeks post-conceptual age — must monitor postoperatively). Regional anesthesia (spinal, caudal) is preferred when feasible to avoid GA-related apnea.
29 Geriatric & High-Risk Patients
Pharmacokinetic Changes with Aging
The elderly demonstrate decreased hepatic blood flow (by ~40%) and reduced Phase I metabolism (CYP450), leading to prolonged elimination of midazolam, fentanyl, and propofol. Decreased renal function (GFR declines ~1 mL/min/year after age 40) prolongs the effect of renally cleared drugs (morphine M6G metabolite, vecuronium metabolites, gabapentin). Decreased lean body mass and increased body fat alter the volume of distribution: lipophilic drugs (benzodiazepines, fentanyl) have larger Vd and prolonged effect; hydrophilic drugs (NMBAs) have smaller Vd and relatively higher initial plasma concentrations. Decreased plasma albumin increases the free (active) fraction of highly protein-bound drugs. Decreased total body water concentrates water-soluble drugs. The net result: the elderly are more sensitive to virtually all anesthetic agents, and doses must be reduced.
Dose Reduction Recommendations
| Agent | Dose Modification in Elderly |
|---|---|
| Propofol | Reduce induction dose by 30–50% (1–1.5 mg/kg); slow injection; expect more profound hypotension |
| Etomidate | Reduce by 25% (0.15–0.2 mg/kg) |
| Volatile agents | MAC decreases ~6% per decade after 40 — an 80-year-old needs ~40% less than a 40-year-old |
| Opioids | Reduce by 30–50%; increase dosing interval; fentanyl preferred for titratability |
| Benzodiazepines | Reduce midazolam by 50% or more; prolonged effect; paradoxical agitation more common |
| NMBAs | Onset may be delayed (decreased CO); duration prolonged (especially vecuronium with renal impairment) |
| Neuraxial | Spinal dose reduced by 25–30% due to decreased CSF volume and increased spread |
Regional Anesthesia Preference in the Elderly
Regional anesthesia offers several theoretical advantages in geriatric patients: avoidance of airway manipulation and mechanical ventilation, reduced exposure to sedatives and volatile agents (which may contribute to delirium/POCD), better preservation of pulmonary function (especially thoracic epidural), superior postoperative analgesia with reduced opioid consumption, potential reduction in thromboembolic events (neuraxial sympathectomy improves blood flow), and earlier return of cognitive function. However, large randomized trials (RAGA trial for hip fracture) have not consistently shown mortality benefit of regional vs. general anesthesia. The decision should be individualized based on patient factors (anticoagulation status, spinal anatomy, patient preference) and surgical requirements. Peripheral nerve blocks (e.g., fascia iliaca block for hip fracture) provide excellent preoperative analgesia and reduce opioid requirements in the emergency department and perioperatively.
Frailty Assessment
Frailty is a clinical syndrome of decreased physiologic reserve. The Clinical Frailty Scale (CFS) ranges from 1 (very fit) to 9 (terminally ill). Frail patients (CFS ≥5) have 2–5× higher risk of postoperative complications, ICU admission, longer hospital stay, and 30-day mortality. Preoperative frailty screening should guide shared decision-making, choice of anesthetic technique (regional preferred), and postoperative disposition planning.
Postoperative Delirium & POCD
Postoperative delirium is an acute, fluctuating disturbance in attention and cognition. Incidence: 10–50% after major surgery in elderly patients. Risk factors: age >65, pre-existing cognitive impairment/dementia, sensory impairment (hearing/vision), major surgery, prolonged OR time, benzodiazepine use, opioid use, sleep deprivation, infection, electrolyte imbalance, urinary retention, pain, ICU admission. Prevention — the HELP protocol (Hospital Elder Life Program): orientation (reorientation boards, clock), early mobilization, sleep hygiene (minimize nighttime disruptions), hydration and nutrition, vision and hearing aids at bedside, avoid delirium-inducing medications (benzodiazepines, anticholinergics, meperidine). No pharmacologic agent reliably prevents delirium. Treatment: identify and correct underlying cause, non-pharmacologic interventions first; antipsychotics (haloperidol 0.5–1 mg) only for severe agitation with safety risk; avoid benzodiazepines (worsen delirium except for alcohol/benzodiazepine withdrawal).
Postoperative cognitive dysfunction (POCD) is a measurable decline in cognitive function (memory, attention, executive function) lasting weeks to months after surgery. More common after cardiac surgery and in the elderly. Risk factors overlap with delirium risk factors. No proven prevention or treatment; regional anesthesia may reduce POCD risk compared to general anesthesia (data conflicting). Distinguished from delirium by chronicity (weeks to months) and absence of fluctuating attention.
30 Drug Dosing Master Table
Induction Agents
| Drug | IV Induction Dose | Onset | Duration |
|---|---|---|---|
| Propofol | 1.5–2.5 mg/kg | 30–40 sec | 5–10 min |
| Etomidate | 0.2–0.3 mg/kg | 30–60 sec | 3–5 min |
| Ketamine | 1–2 mg/kg IV; 4–6 mg/kg IM | 30–60 sec IV | 10–20 min |
| Midazolam (co-induction) | 0.01–0.05 mg/kg | 1–3 min | 15–30 min |
| Thiopental | 3–5 mg/kg | 30 sec | 5–10 min |
Neuromuscular Blocking Agents
| Drug | Intubation Dose | Onset | Duration | Reversal |
|---|---|---|---|---|
| Succinylcholine | 1–1.5 mg/kg | 30–60 sec | 5–10 min | Spontaneous (pseudocholinesterase) |
| Rocuronium | 0.6–1.2 mg/kg | 60–90 sec | 30–45 min | Sugammadex 2–16 mg/kg; neostigmine 0.05 mg/kg |
| Vecuronium | 0.08–0.1 mg/kg | 2–3 min | 25–40 min | Sugammadex 2–4 mg/kg; neostigmine |
| Cisatracurium | 0.15–0.2 mg/kg | 2–3 min | 25–40 min | Neostigmine (sugammadex ineffective) |
Opioids
| Drug | Typical Dose | Onset | Duration | Potency (vs. Morphine) |
|---|---|---|---|---|
| Fentanyl | 1–2 mcg/kg bolus | 1–2 min | 30–60 min | 80–100× |
| Sufentanil | 0.1–0.5 mcg/kg | 1–2 min | 20–45 min | 500–1000× |
| Remifentanil | 0.05–0.2 mcg/kg/min infusion | 1–1.5 min | 3–5 min offset | 100–200× |
| Morphine | 0.1–0.15 mg/kg | 5–10 min | 3–4 h | 1× |
| Hydromorphone | 0.01–0.02 mg/kg | 5 min | 3–4 h | 5–7× |
Local Anesthetics — Maximum Doses
| Drug | Max Dose Without Epinephrine | Max Dose With Epinephrine |
|---|---|---|
| Lidocaine | 4.5 mg/kg | 7 mg/kg |
| Bupivacaine | 2.5 mg/kg | 3 mg/kg |
| Ropivacaine | 3 mg/kg | 3.5 mg/kg |
| Chloroprocaine | 11 mg/kg | 14 mg/kg |
| Mepivacaine | 4.4 mg/kg | 6.6 mg/kg |
Vasopressors & Emergency Drugs
| Drug | Dose | Mechanism | Indication |
|---|---|---|---|
| Phenylephrine | 50–200 mcg IV bolus; infusion 0.5–5 mcg/kg/min | Pure α1 agonist | Hypotension (first-line for spinal-induced), reflex bradycardia may occur |
| Ephedrine | 5–10 mg IV bolus | Indirect sympathomimetic (α + β) | Hypotension with bradycardia; less effective after repeated dosing (tachyphylaxis) |
| Epinephrine | 10–100 mcg IV bolus; infusion 0.01–0.5 mcg/kg/min; 1 mg for cardiac arrest | α1, β1, β2 agonist | Anaphylaxis, cardiac arrest, severe hypotension, bronchospasm |
| Norepinephrine | Infusion 0.05–0.5 mcg/kg/min | α1 >> β1 agonist | Septic shock, vasodilatory shock, refractory hypotension |
| Vasopressin | 1–2 units IV bolus; infusion 0.01–0.04 units/min | V1 receptor agonist | Vasodilatory shock refractory to catecholamines |
| Atropine | 0.5–1 mg IV (max 3 mg total) | Muscarinic antagonist | Symptomatic bradycardia |
| Glycopyrrolate | 0.2–0.4 mg IV | Muscarinic antagonist (does not cross BBB) | Bradycardia, antisialagogue, neostigmine co-administration |
| Dantrolene | 2.5 mg/kg IV, repeat q5min up to 10 mg/kg | Ryanodine receptor antagonist — inhibits Ca²+ release from SR | Malignant hyperthermia |
| Intralipid 20% | 1.5 mL/kg bolus, then 0.25 mL/kg/min infusion | Lipid sink — sequesters lipophilic LA | Local anesthetic systemic toxicity (LAST) |
| Naloxone | 0.04–0.4 mg IV q2–3min | Opioid (mu) antagonist | Opioid overdose / respiratory depression |
| Flumazenil | 0.2 mg IV q1min (max 1 mg) | Benzodiazepine antagonist (GABAA) | Benzodiazepine overdose (caution: seizure risk in chronic users) |
31 Classification Systems
ASA Physical Status Classification
| Class | Definition |
|---|---|
| ASA I | Normal healthy patient |
| ASA II | Mild systemic disease, no functional limitation |
| ASA III | Severe systemic disease, substantive functional limitation |
| ASA IV | Severe systemic disease, constant threat to life |
| ASA V | Moribund, not expected to survive without surgery |
| ASA VI | Brain-dead organ donor |
| ASA-E | Emergency modifier (appended to any class) |
Mallampati Classification
| Class | Visible Structures |
|---|---|
| I | Soft palate, fauces, uvula, tonsillar pillars |
| II | Soft palate, fauces, uvula partially visible |
| III | Soft palate, base of uvula |
| IV | Hard palate only |
Cormack-Lehane Laryngoscopic View
| Grade | View at Laryngoscopy | Expected Difficulty |
|---|---|---|
| I | Full vocal cords visible | Easy |
| IIa | Partial cords visible | Usually straightforward |
| IIb | Arytenoids or posterior commissure only | May require bougie |
| III | Epiglottis only, no glottic structure | Difficult — alternative technique needed |
| IV | Neither epiglottis nor glottis visible | Cannot intubate by direct laryngoscopy |
Revised Cardiac Risk Index (RCRI)
| Factor | Score |
|---|---|
| High-risk surgery (intraperitoneal, intrathoracic, suprainguinal vascular) | 1 |
| History of ischemic heart disease | 1 |
| History of congestive heart failure | 1 |
| History of cerebrovascular disease | 1 |
| Diabetes requiring insulin | 1 |
| Creatinine ≥2 mg/dL | 1 |
Risk of MACE: 0 points = 3.9%, 1 = 6.0%, 2 = 10.1%, ≥3 = 15%.
Ramsay Sedation Scale
| Level | Description |
|---|---|
| 1 | Anxious, agitated, restless |
| 2 | Cooperative, oriented, tranquil |
| 3 | Responds to commands only |
| 4 | Brisk response to light glabellar tap or loud auditory stimulus |
| 5 | Sluggish response to stimulus |
| 6 | No response |
Aldrete Post-Anesthesia Recovery Score
| Criterion | 2 Points | 1 Point | 0 Points |
|---|---|---|---|
| Activity | Moves all 4 extremities | Moves 2 extremities | No movement |
| Respiration | Breathes deeply, coughs | Dyspnea, limited breathing | Apnea |
| Circulation | BP ±20% of pre-anesthetic | BP ±20–50% | BP ±>50% |
| Consciousness | Fully awake | Arousable on calling | Not responding |
| SpO₂ | ≥92% on room air | Needs O₂ to maintain ≥90% | <90% even with O₂ |
Discharge from PACU requires score ≥9 out of 10.
Wilson Risk Sum Score for Difficult Intubation
| Factor | 0 Points | 1 Point | 2 Points |
|---|---|---|---|
| Weight | <90 kg | 90–110 kg | >110 kg |
| Head/neck movement | >90° | ~90° | <90° |
| Jaw movement | Inter-incisor gap ≥5 cm or SLux >0 | IG <5 cm and SLux = 0 | IG <5 cm and SLux <0 |
| Receding mandible | Normal | Moderate | Severe |
| Buck teeth | Normal | Moderate | Severe |
Score ≥2: ~75% sensitivity for difficult intubation. SLux = subluxation of the mandible (lower incisors anterior to upper incisors).
32 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| ABG | Arterial blood gas |
| ACLS | Advanced cardiac life support |
| AFOI | Awake fiberoptic intubation |
| AKI | Acute kidney injury |
| ARDS | Acute respiratory distress syndrome |
| ARISCAT | Assess Respiratory Risk in Surgical Patients in Catalonia |
| ASA | American Society of Anesthesiologists |
| BBB | Blood-brain barrier |
| BIS | Bispectral index |
| BMV | Bag-mask ventilation |
| BP | Blood pressure |
| BSA | Body surface area |
| CaO₂ | Arterial oxygen content |
| CHCT | Caffeine-halothane contracture test |
| CI | Cardiac index |
| CICO | Cannot intubate, cannot oxygenate |
| CO | Cardiac output |
| COPD | Chronic obstructive pulmonary disease |
| CPP | Coronary perfusion pressure |
| CRPS | Complex regional pain syndrome |
| CSE | Combined spinal-epidural |
| CSF | Cerebrospinal fluid |
| CSHT | Context-sensitive half-time |
| CVP | Central venous pressure |
| CXR | Chest X-ray |
| DBP | Diastolic blood pressure |
| DIC | Disseminated intravascular coagulation |
| DL | Direct laryngoscopy |
| DOAC | Direct oral anticoagulant |
| DO₂ | Oxygen delivery |
| ECG | Electrocardiogram |
| ECMO | Extracorporeal membrane oxygenation |
| ED | Emergence delirium |
| EF | Ejection fraction |
| ERAS | Enhanced Recovery After Surgery |
| ESRD | End-stage renal disease |
| ESP | Erector spinae plane |
| ETCO₂ | End-tidal carbon dioxide |
| ETT | Endotracheal tube |
| EVD | External ventricular drain |
| FiO₂ | Fraction of inspired oxygen |
| FONA | Front-of-neck access |
| FRC | Functional residual capacity |
| GA | General anesthesia |
| GFR | Glomerular filtration rate |
| HCM | Hypertrophic cardiomyopathy |
| HELP | Hospital Elder Life Program |
| HR | Heart rate |
| IBW | Ideal body weight |
| ICP | Intracranial pressure |
| IDDS | Intrathecal drug delivery system |
| IJV | Internal jugular vein |
| IOP | Intraocular pressure |
| IV | Intravenous |
| JVD | Jugular venous distension |
| LA | Local anesthetic |
| LAST | Local anesthetic systemic toxicity |
| LMA | Laryngeal mask airway |
| LMWH | Low-molecular-weight heparin |
| LOR | Loss of resistance |
| LR | Lactated Ringer's solution |
| LVEDP | Left ventricular end-diastolic pressure |
| LVEDV | Left ventricular end-diastolic volume |
| LVOT | Left ventricular outflow tract |
| MAC | Minimum alveolar concentration (or monitored anesthesia care, context-dependent) |
| MACE | Major adverse cardiac event |
| MAP | Mean arterial pressure |
| MAOI | Monoamine oxidase inhibitor |
| MBB | Medial branch block |
| MELD | Model for End-Stage Liver Disease |
| MET | Metabolic equivalent |
| MH | Malignant hyperthermia |
| MHAUS | Malignant Hyperthermia Association of the United States |
| mPAP | Mean pulmonary artery pressure |
| MTP | Massive transfusion protocol |
| NIBP | Non-invasive blood pressure |
| NMBA | Neuromuscular blocking agent |
| NMDA | N-methyl-D-aspartate |
| NPO | Nil per os (nothing by mouth) |
| NRS | Numeric rating scale |
| NS | Normal saline (0.9% NaCl) |
| NSAID | Non-steroidal anti-inflammatory drug |
| OSA | Obstructive sleep apnea |
| PAC | Pulmonary artery catheter |
| PACU | Post-anesthesia care unit |
| PCA | Patient-controlled analgesia |
| PCEA | Patient-controlled epidural analgesia |
| PCWP | Pulmonary capillary wedge pressure |
| PDA | Posterior descending artery (or patent ductus arteriosus) |
| PDPH | Post-dural puncture headache |
| PEEP | Positive end-expiratory pressure |
| POCD | Postoperative cognitive dysfunction |
| PONV | Postoperative nausea and vomiting |
| PPHN | Persistent pulmonary hypertension of the newborn |
| pRBC | Packed red blood cells |
| PRIS | Propofol infusion syndrome |
| PTC | Post-tetanic count |
| PVB | Paravertebral block |
| PVR | Pulmonary vascular resistance |
| QL | Quadratus lumborum |
| RA | Regional anesthesia (or right atrium) |
| RCRI | Revised Cardiac Risk Index |
| RFA | Radiofrequency ablation |
| RLN | Recurrent laryngeal nerve |
| ROTEM | Rotational thromboelastometry |
| RSI | Rapid sequence induction/intubation |
| RV | Right ventricle (or residual volume) |
| RYR1 | Ryanodine receptor type 1 |
| SBP | Systolic blood pressure |
| SCh | Succinylcholine |
| SCS | Spinal cord stimulation |
| SGA | Supraglottic airway |
| SI | Sacroiliac |
| SLN | Superior laryngeal nerve |
| SpO₂ | Peripheral oxygen saturation (pulse oximetry) |
| SV | Stroke volume |
| SVC | Superior vena cava |
| SvO₂ | Mixed venous oxygen saturation |
| SVR | Systemic vascular resistance |
| TACO | Transfusion-associated circulatory overload |
| TAP | Transversus abdominis plane |
| TCI | Target-controlled infusion |
| TEE | Transesophageal echocardiography |
| TEG | Thromboelastography |
| TENS | Transcutaneous electrical nerve stimulation |
| TIVA | Total intravenous anesthesia |
| TLC | Total lung capacity |
| TMD | Thyromental distance |
| TNS | Transient neurologic symptoms |
| TOF | Train-of-four |
| TRALI | Transfusion-related acute lung injury |
| TV | Tidal volume |
| TXA | Tranexamic acid |
| ULBT | Upper lip bite test |
| VAS | Visual analog scale |
| VC | Vital capacity |
| VL | Video laryngoscopy |