Nuclear Medicine
PET/CT, SPECT, thyroid scintigraphy, cardiac stress testing, bone scans, radiopharmaceuticals, theranostics, radiation safety, and every imaging protocol, uptake pattern, radiopharmaceutical agent, and interpretive framework across the full scope of nuclear medicine.
01 Nuclear Physics & Radioactive Decay
Nuclear medicine exploits the spontaneous transformation of unstable atomic nuclei to produce radiation that can be detected externally for imaging or deposited locally for therapy. Understanding the basic physics of radioactive decay, the properties of emitted particles and photons, and the concept of half-life is essential to selecting the right radiopharmaceutical, interpreting images, and ensuring patient and staff safety.
Modes of Radioactive Decay
Alpha (α) decay emits a helium-4 nucleus (2 protons + 2 neutrons) with high linear energy transfer (LET) and very short range in tissue (~50–100 µm). Alpha emitters (e.g., Ra-223, Ac-225) are ideal for targeted radionuclide therapy because they deposit massive energy within a few cell diameters, causing irreparable double-strand DNA breaks while sparing surrounding tissue. Beta-minus (β−) decay emits an electron and an antineutrino when a neutron converts to a proton; the emitted electron has a range of millimeters in tissue (e.g., Y-90 mean range ~2.5 mm, Lu-177 ~0.3 mm), making beta emitters suitable for therapy of larger tumor deposits. Beta-plus (β+) decay (positron emission) emits a positron that travels a short distance before annihilating with an electron, producing two 511-keV gamma photons emitted at ~180° — this is the physical basis of PET imaging. Gamma (γ) emission occurs when a nucleus in an excited state releases energy as electromagnetic radiation without changing atomic number or mass; gamma photons are highly penetrating and are the primary emissions detected in SPECT and planar scintigraphy. Isomeric transition (IT) is gamma emission from a metastable nuclear state (e.g., Tc-99m → Tc-99 + 140 keV gamma). Electron capture (EC) occurs when an inner orbital electron is captured by the nucleus, converting a proton to a neutron (e.g., I-123, Tl-201, In-111); characteristic X-rays and Auger electrons are emitted secondarily.
Half-Life Concepts
The physical half-life (T½p) is the time required for half the atoms in a sample to decay — an intrinsic property of each radionuclide (Tc-99m = 6.02 hours, F-18 = 109.8 minutes, I-131 = 8.02 days, Tl-201 = 73.1 hours). The biological half-life (T½b) is the time for the body to eliminate half the administered radiopharmaceutical through metabolic and excretory pathways. The effective half-life (T½eff) combines both: 1/T½eff = 1/T½p + 1/T½b. The effective half-life is always shorter than either the physical or biological half-life alone and determines the actual radiation dose to the patient.
Specific Activity & Radioactive Equilibrium
Specific activity is the radioactivity per unit mass of a radionuclide (Bq/g or Ci/g). High specific activity is critical for receptor-targeted radiopharmaceuticals (e.g., Lu-177 DOTATATE) because only a small mass of peptide is administered — if non-radioactive ("cold") atoms compete for receptor binding, imaging sensitivity and therapeutic efficacy decrease. Carrier-free preparations have the highest specific activity. Secular equilibrium occurs when the parent half-life is vastly longer than the daughter (T½parent >> T½daughter); the daughter activity eventually equals the parent activity and both decay at the parent's rate (e.g., Mo-99 [T½ = 66 hr] / Tc-99m [T½ = 6 hr] approaches transient equilibrium rather than true secular equilibrium because the parent half-life is only ~11× longer). Transient equilibrium is the correct description for the Mo-99/Tc-99m generator system: daughter activity rises, exceeds the parent activity, then decays in parallel with the parent. The Tc-99m activity in the generator reaches maximum ~23 hours after elution.
The Tc-99m Generator
The Mo-99/Tc-99m generator (informally the "moly cow") consists of Mo-99 adsorbed onto an alumina (Al₂O₃) column. When saline is passed through the column, Tc-99m (as pertechnetate, TcO₄−) is selectively eluted while Mo-99 (as molybdate) remains bound. The generator is eluted daily (or more frequently) in the radiopharmacy. Quality control includes testing for Mo-99 breakthrough (limit ≤0.15 µCi Mo-99 per mCi Tc-99m at administration, tested with a lead pig that attenuates Tc-99m 140 keV but allows Mo-99 740/780 keV gammas through) and Al breakthrough (≤10 µg/mL, tested colorimetrically, because excess aluminum can cause aggregation of sulfur colloid particles). After elution, the Tc-99m pertechnetate is used directly for thyroid/Meckel imaging or complexed with various kits (MDP, sestamibi, MAG3, HMPAO, mebrofenin, sulfur colloid, MAA) to produce specific radiopharmaceuticals. Each kit contains a lyophilized reagent with a reducing agent (stannous chloride) that reduces Tc-99m from the +7 oxidation state (pertechnetate) to a lower state (+3 to +5) that binds the chelating ligand. Radiochemical purity must be verified (typically ≥90–95%) using thin-layer chromatography to ensure adequate labeling.
| Test | Purpose | Limit | Method |
|---|---|---|---|
| Mo-99 breakthrough | Ensure parent radionuclide does not contaminate eluate | ≤0.15 µCi Mo-99 / mCi Tc-99m | Lead pig assay (attenuates 140 keV, passes 740 keV) |
| Al breakthrough | Prevent aluminum contamination from column | ≤10 µg/mL | Colorimetric test (aurin tricarboxylic acid) |
| Eluate volume | Ensure proper saline volume | Per manufacturer specifications | Visual inspection |
| Eluate clarity | Detect particulate matter | Clear, colorless solution | Visual inspection against light |
F-18 Production via Cyclotron
F-18 is produced in a cyclotron by bombarding O-18-enriched water with protons: ¹⁸O(p,n)¹⁸F. The F-18 is then incorporated into deoxyglucose to form F-18 FDG (fluorodeoxyglucose) via nucleophilic substitution. Because F-18 has a 110-minute half-life, FDG must be produced at a nearby cyclotron facility or regional radiopharmacy and transported quickly. The positron emitted by F-18 travels ~1 mm in tissue before annihilation, yielding two 511-keV photons detected by the PET ring detector in coincidence.
Tc-99m: IT, 140 keV gamma, T½ 6.02 hr. F-18: β+ decay, 511 keV annihilation photons, T½ 110 min. I-131: β− (for therapy) + 364 keV gamma (for imaging), T½ 8.02 days. I-123: EC, 159 keV gamma, T½ 13.2 hr. Tl-201: EC, 69–83 keV X-rays, T½ 73 hr. In-111: EC, 171 + 245 keV gammas, T½ 67.3 hr. Ga-67: EC, 93/185/300 keV gammas, T½ 78.3 hr. Ga-68: β+, 511 keV, T½ 68 min. Lu-177: β−, 208 keV gamma, T½ 6.7 days. Ra-223: α, T½ 11.4 days. Rb-82: β+, T½ 75 sec (from Sr-82 generator).
02 Radiation Detection & Imaging Systems
The Gamma Camera (Anger Camera)
The gamma camera, invented by Hal Anger in 1958, remains the primary imaging device for single-photon emitting radionuclides. Its components, in order from patient to output, are: (1) collimator — a lead plate with holes that selects photons traveling in a specific direction, rejecting scattered photons; (2) NaI(Tl) scintillation crystal — typically 9.5 mm thick, converts gamma photons to visible light (scintillation); (3) photomultiplier tubes (PMTs) — an array of 37–91 PMTs converts light to electrical signals; (4) position logic circuitry computes the X-Y coordinates of each scintillation event; (5) pulse height analyzer (PHA) accepts only events within a specified energy window (e.g., 140 keV ± 10% for Tc-99m) to reject scatter.
Collimator Types
The collimator is the most important determinant of spatial resolution and sensitivity in planar/SPECT imaging. Low-energy all-purpose (LEAP) and low-energy high-resolution (LEHR) parallel-hole collimators are used for Tc-99m (140 keV) and similar low-energy emitters; LEHR has longer, narrower holes providing better resolution but lower sensitivity. Medium-energy (ME) collimators have thicker septa for In-111 (171/245 keV) and Ga-67. High-energy (HE) collimators are required for I-131 (364 keV). Pinhole collimators provide magnified images of small organs (thyroid, pediatric joints) with excellent resolution at short distances but rapidly decreasing sensitivity with distance. Converging collimators (fan-beam, cone-beam) magnify the image and increase sensitivity for brain SPECT. Diverging collimators are rarely used but can image a large field of view (e.g., lungs) with a small crystal.
SPECT — Single-Photon Emission Computed Tomography
SPECT acquires planar projections at multiple angles (typically 60–128 stops over 180° or 360°) as the gamma camera rotates around the patient. These projections are reconstructed into transaxial slices using either filtered back projection (FBP) or, more commonly, iterative reconstruction algorithms (ordered subset expectation maximization — OSEM) that model the imaging physics and reduce noise. Attenuation correction is critical: photons from deep structures are attenuated more than those from the periphery, creating artifacts (e.g., apparent inferior wall defects in cardiac SPECT from diaphragmatic attenuation). Modern SPECT/CT systems use the CT for attenuation correction (AC) and anatomic localization. Resolution recovery algorithms model the depth-dependent collimator response to improve resolution.
SPECT/CT & CZT Cameras
SPECT/CT integrates a SPECT camera with a diagnostic or low-dose CT scanner, providing simultaneous functional and anatomic data. The CT component serves three purposes: attenuation correction, anatomic localization of radiotracer uptake (critical for differentiating benign from malignant lesions on bone SPECT, localizing parathyroid adenomas, and identifying surgical landmarks), and in some cases diagnostic CT interpretation. Cadmium-zinc-telluride (CZT) cameras represent a significant advancement: CZT is a solid-state semiconductor detector that converts gamma photons directly to electrical signals (without the intermediate light conversion step in NaI/PMT systems). CZT cameras offer: 2× better energy resolution (enabling cleaner dual-isotope acquisition), improved spatial resolution, and 5–10× higher sensitivity (allowing lower injected doses or faster acquisitions). CZT-based cardiac cameras (e.g., D-SPECT, Discovery NM 530c) have transformed cardiac SPECT with 5-minute acquisitions and half-dose protocols, reducing patient radiation exposure to ~3–5 mSv (comparable to cardiac PET).
Quality Control in Nuclear Medicine
Daily gamma camera quality control includes: uniformity flood (point source or sheet source to verify uniform response across the detector — integral uniformity should be <5%, differential uniformity <3%), energy peak check (verify photopeak is centered within the energy window), and visual inspection for artifacts. Weekly/monthly QC includes: spatial resolution and linearity (bar phantom or quadrant bar phantom), center of rotation (COR) calibration for SPECT (misalignment causes image blurring), and sensitivity checks. For PET scanners: daily blank and normalization scans, quarterly well-counter cross-calibration (ensures quantitative accuracy of SUV measurements), and routine phantom imaging for resolution verification. For dose calibrators: daily constancy testing, quarterly linearity testing (across the clinical activity range), and annual geometry testing. All QC results must be documented and available for NRC or state regulatory inspection.
PET — Positron Emission Tomography
In PET, a positron emitted from the nucleus travels a short distance (positron range depends on energy — ~1 mm for F-18, ~2.6 mm for Rb-82) before encountering an electron. The resulting annihilation produces two 511-keV photons traveling ~180° apart. The PET detector ring identifies both photons within a narrow timing window (coincidence detection, typically 4–12 ns) — this electronic collimation eliminates the need for physical collimators and provides superior sensitivity and resolution compared to SPECT. Time-of-flight (TOF) PET measures the tiny difference in arrival time of the two photons to localize the annihilation event along the line of response, improving signal-to-noise ratio, particularly in large patients. Modern TOF timing resolution is ~200–400 ps.
PET/CT & PET/MRI Fusion
PET/CT combines a PET scanner with a multi-detector CT in a single gantry. The CT provides: (1) rapid attenuation correction (replacing time-consuming Ge-68 transmission scans), (2) anatomic localization of metabolic findings, and (3) diagnostic-quality CT when performed with IV contrast. Patients are positioned on a single table that moves sequentially through the CT and PET portions. PET/MRI combines PET with MRI, offering superior soft-tissue contrast (particularly for brain, liver, pelvis, and pediatric imaging) and reduced radiation dose (no CT component). Attenuation correction in PET/MRI is more challenging because MRI does not directly measure photon attenuation; Dixon-based sequences classify tissue into air, lung, fat, and soft tissue for an approximate attenuation map. PET/MRI is increasingly used in neuroimaging, pediatric oncology, and hepatic assessment.
| Feature | SPECT | PET |
|---|---|---|
| Collimation | Physical (lead collimator) | Electronic (coincidence detection) |
| Spatial resolution | 8–12 mm | 4–5 mm |
| Sensitivity | Lower (collimator rejects >99.9% of photons) | 10–100× higher |
| Attenuation correction | CT-based or calculated | CT-based (PET/CT) or MRI-based |
| Quantification | Semi-quantitative | Quantitative (SUV) |
| Common tracers | Tc-99m agents, Tl-201, I-123, In-111 | F-18 FDG, Ga-68, Rb-82, F-18 NaF |
| Cost | Lower | Higher (cyclotron needed for most tracers) |
03 Radiation Safety & Regulatory
Regulatory Framework
In the United States, the Nuclear Regulatory Commission (NRC) regulates the medical use of byproduct material under 10 CFR Part 35. Individual states may become "Agreement States" and assume regulatory authority. The NRC defines categories of authorized users: physicians authorized under 35.100 (uptake/dilution/excretion), 35.200 (imaging and localization), 35.300 (written directive required — therapeutic administrations including I-131 >33 µCi), 35.400 (manual brachytherapy), 35.600 (sealed sources for diagnosis), and 35.1000 (emerging technologies). A Radiation Safety Officer (RSO) oversees the institutional radiation safety program, and a Radiation Safety Committee (RSC) governs policy at facilities with multiple authorized users.
ALARA Principle
The fundamental principle of radiation protection is ALARA — As Low As Reasonably Achievable. Dose reduction is achieved through three strategies: (1) time — minimize time spent near radioactive sources; (2) distance — radiation exposure decreases with the inverse square of distance (doubling distance reduces exposure to 25%); (3) shielding — lead aprons, syringe shields, L-blocks, and leaded glass reduce exposure from gamma emitters. For beta emitters (e.g., Y-90, P-32), low-Z shielding (acrylic/Plexiglass) is used first to absorb beta particles without producing bremsstrahlung radiation, followed by lead if significant gamma component exists.
Dose Limits
| Category | Annual Effective Dose Limit | Details |
|---|---|---|
| Occupational — whole body | 50 mSv/year (5 rem) | TEDE; also limited to cumulative 10 × age (mSv) |
| Occupational — lens of eye | 150 mSv/year (15 rem) | ICRP now recommends 20 mSv/yr averaged over 5 years |
| Occupational — extremities/skin | 500 mSv/year (50 rem) | Shallow dose equivalent |
| Declared pregnant worker | 5 mSv over gestation (0.5 rem) | Monthly monitoring; reassign duties if needed |
| Embryo/fetus | 5 mSv total (0.5 rem) | Should not exceed 0.5 mSv/month |
| Public (individual member) | 1 mSv/year (0.1 rem) | From licensed operations |
| Minor (student/trainee <18 yr) | 1 mSv/year (0.1 rem) | 10% of adult occupational limit |
Written Directives & Misadministration
A written directive is required before administering: (1) I-131 sodium iodide >30 µCi (1.11 MBq), (2) any therapeutic dose of an unsealed byproduct material, or (3) any dose requiring 10 CFR 35.300 authorization. The directive must specify the radiopharmaceutical, dosage, and route. A medical event (formerly "misadministration") must be reported to the NRC if the administered dose differs from the written directive by >20%, the wrong radiopharmaceutical is given, or dose is delivered to the wrong patient or wrong treatment site. Reporting must occur within 24 hours of discovery to the NRC, and the referring physician and patient must be notified within 24 hours unless the referring physician determines notification would be harmful.
Patient Release Criteria
Patients may be released from the facility after therapeutic radiopharmaceutical administration when the measured dose rate is ≤7 mR/hr at 1 meter from the patient, or when a patient-specific dose calculation shows that no individual will receive >5 mSv (0.5 rem) total effective dose equivalent. For I-131 therapy, patients receiving ≤33 mCi (1.22 GBq) generally meet release criteria immediately. Higher doses (e.g., 100–200 mCi for thyroid cancer) require patient-specific calculations considering occupancy factors and may necessitate brief hospitalization or detailed home radiation safety instructions (sleep alone, separate utensils, maintain distance from children/pregnant individuals, flush toilet twice).
Breastfeeding Cessation
| Radiopharmaceutical | Recommended Cessation |
|---|---|
| Tc-99m pertechnetate | 24 hours (pump and discard) |
| Tc-99m MAA | 12 hours |
| Tc-99m MDP | 24 hours |
| Tc-99m sestamibi | 24 hours |
| I-123 (diagnostic) | Cessation depends on dose; consult medical physicist |
| I-131 (any dose) | Complete cessation (permanently for that child) |
| Ga-67 citrate | 1 month |
| Tl-201 | 2 weeks |
| F-18 FDG | 24 hours (pump and discard) |
| In-111 WBC | 1 week |
Contamination & Waste
Wipe tests (swab surveys) are performed to detect removable contamination on surfaces; the threshold for acceptable contamination is ≤200 dpm/100 cm² for most beta/gamma emitters. Survey meters (Geiger-Mueller counters, ionization chambers) measure ambient exposure rates. Radioactive waste with half-lives ≤120 days may be held for decay in storage until activity is indistinguishable from background (≥10 half-lives), then disposed of as non-radioactive waste after removing all radiation labels. Longer-lived waste must be transferred to a licensed waste broker.
Personnel Monitoring
All individuals likely to receive ≥10% of any occupational dose limit must wear a personnel dosimeter. Common types include: optically stimulated luminescence (OSL) dosimeters (most common, aluminum oxide detector, can be re-read), thermoluminescent dosimeters (TLDs) (lithium fluoride, single-read), and electronic personal dosimeters (EPDs) (real-time readout). Dosimeters are worn at the collar (outside the lead apron if worn) to estimate head/neck dose, with a second dosimeter under the apron at waist level to estimate body dose in interventional settings. Ring dosimeters (TLD rings) are required when handling high-activity radiopharmaceuticals for dose to the extremities. Declared pregnant workers receive a fetal dosimeter worn at waist level under the lead apron, monitored monthly with a cumulative limit of 5 mSv for the gestation.
Spill Management
Minor spills (<1 mCi) are managed by: (1) notifying personnel in the area, (2) blotting (not wiping) with absorbent material, (3) surveying with a Geiger counter, and (4) wipe-testing to confirm decontamination. Major spills (>1 mCi or any alpha emitter) additionally require: clearing the area, contacting the RSO, sealing the area, and formal decontamination by trained personnel. Personnel who are contaminated should remove clothing, wash affected skin thoroughly, and be surveyed. Contamination control mats ("sticky mats") at room exits help prevent spread.
04 Key Terminology & Abbreviations
Nuclear medicine uses a specialized vocabulary drawn from physics, radiochemistry, and clinical medicine. This section provides the essential terms needed to navigate the discipline.
| Term | Definition |
|---|---|
| Activity | Rate of nuclear disintegration; SI unit = becquerel (Bq, 1 decay/sec); traditional unit = curie (Ci, 3.7 × 10¹⁰ dps). 1 mCi = 37 MBq. |
| Absorbed dose | Energy deposited per unit mass; SI = gray (Gy); traditional = rad. 1 Gy = 100 rad. |
| Equivalent dose | Absorbed dose × radiation weighting factor (wR); SI = sievert (Sv); traditional = rem. wR = 1 for beta/gamma, 20 for alpha. |
| Effective dose | Sum of equivalent doses to all organs × tissue weighting factors; reflects whole-body stochastic risk. |
| SUV | Standardized Uptake Value — ratio of tissue radioactivity concentration to injected dose per body weight. |
| LET | Linear energy transfer — energy deposited per unit path length (keV/µm). High LET = alpha > beta > gamma. |
| Photopeak | The full-energy peak in the gamma spectrum corresponding to complete absorption of the gamma photon. |
| Dead time | Time after detecting an event during which the system cannot register another event; causes count rate loss at high activities. |
| Collimator resolution | Ability to distinguish two point sources; degrades with increasing source-to-collimator distance. |
| Annihilation | Positron-electron interaction producing two 511-keV photons at ~180°. |
| Coincidence window | Timing window in PET within which two 511-keV photons must be detected to be registered as a true event. |
| Attenuation | Reduction of photon intensity as it passes through tissue; major source of artifacts in SPECT and PET. |
| Scatter | Compton scatter changes photon direction and energy; energy windowing and scatter correction algorithms reduce its effect. |
| Biodistribution | Normal pattern of radiotracer accumulation in organs/tissues; varies by radiopharmaceutical mechanism. |
| Target-to-background ratio | Ratio of radiotracer uptake in the target lesion to surrounding normal tissue; higher = better lesion detection. |
| Radiochemical purity (RCP) | Percentage of total activity in the desired chemical form; tested by thin-layer chromatography. |
| Radionuclidic purity | Percentage of total activity attributable to the desired radionuclide (e.g., absence of Mo-99 in Tc-99m eluate). |
| Carrier-free | Preparation containing only radioactive atoms of the element, with no stable (non-radioactive) isotopic carriers. |
| Kit preparation | Adding Tc-99m pertechnetate to a lyophilized vial containing ligand and reducing agent to form a specific radiopharmaceutical. |
| Written directive | NRC-required document specifying the radiopharmaceutical, dose, route, and patient for therapeutic administrations. |
| Medical event | Administration error (wrong patient, wrong drug, dose >20% deviation from directive) that must be reported to the NRC. |
| Dosimetry | Calculation of absorbed radiation dose to organs and tumors; guides therapy planning and safety assessment. |
| MIRD | Medical Internal Radiation Dose — committee and schema for internal dose calculation using source and target organ models. |
| Theranostics | Diagnostic-therapeutic pairing where the same molecular target is imaged (for selection) and treated (for therapy) using companion radiopharmaceuticals. |
Activity: 1 Ci = 37 GBq; 1 mCi = 37 MBq; 1 µCi = 37 kBq. Absorbed dose: 1 Gy = 100 rad; 1 cGy = 1 rad. Equivalent/effective dose: 1 Sv = 100 rem; 1 mSv = 100 mrem. Radiation weighting factors (wR): gamma/beta/X-ray = 1; alpha = 20; neutrons = 5–20 (energy dependent). Remember: for gamma/beta emitters, 1 Gy = 1 Sv (because wR = 1); for alpha emitters, 1 Gy = 20 Sv.
05 F-18 FDG PET/CT — Physics & Protocol
FDG Mechanism
F-18 fluorodeoxyglucose (FDG) is a glucose analog in which the 2′-hydroxyl group is replaced by F-18. FDG enters cells via glucose transporters (GLUT-1, GLUT-3 — upregulated in most cancers) and is phosphorylated by hexokinase to FDG-6-phosphate. Unlike glucose-6-phosphate, FDG-6-phosphate cannot be further metabolized by glucose-6-phosphate isomerase and is trapped intracellularly (metabolic trapping). The rate of FDG accumulation therefore reflects the rate of glucose metabolism. Malignant cells typically exhibit increased glycolysis even in the presence of oxygen (the Warburg effect), leading to preferential FDG uptake in tumors relative to most normal tissues.
Patient Preparation
Patients must fast for 4–6 hours before FDG injection to minimize insulin-mediated FDG uptake by skeletal muscle and to reduce competitive inhibition of FDG uptake by elevated serum glucose. Blood glucose must be <200 mg/dL at the time of injection (ideally <150 mg/dL); hyperglycemia competitively inhibits FDG uptake in tumors, reducing sensitivity. Diabetic patients should be scheduled in the early morning; insulin should not be given within 4 hours of injection (insulin drives FDG into muscle). Patients should avoid strenuous exercise for 24 hours to minimize muscular FDG uptake. They remain in a warm, quiet room during the 60-minute uptake period (some protocols use 90 minutes for improved tumor-to-background ratio) to minimize brown fat activation and muscle uptake. Oral hydration is encouraged to promote renal clearance.
SUV — Standardized Uptake Value
The SUV quantifies FDG accumulation: SUV = (tissue activity concentration [kBq/mL]) / (injected dose [kBq] / patient body weight [g]). An SUV of 1.0 means uniform distribution. Most malignancies have SUVmax >2.5, though this threshold is not absolute. SUVmax (maximum voxel value in a volume of interest) is the most commonly reported metric because it is reproducible and less dependent on ROI delineation. SUVmean averages across a defined volume. SUVpeak averages within a 1-cm³ sphere centered on the hottest voxel. SUL (SUV corrected for lean body mass) is used in PERCIST criteria and is less affected by body habitus than weight-based SUV. Metabolic tumor volume (MTV) and total lesion glycolysis (TLG) = MTV × SUVmean are volumetric PET metrics gaining prognostic importance in lymphoma, lung cancer, and other malignancies. Pitfalls affecting SUV include: patient weight changes, paravenous injection (always check injection site), uptake time variation (standardize at 60 ± 10 min), blood glucose level (high glucose competitively inhibits FDG uptake, falsely lowering SUV), scanner calibration drift, reconstruction algorithm and parameters, and respiratory motion (causes blurring and SUV underestimation in lung bases/liver dome).
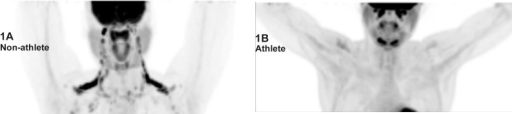
Brown Fat Activation
Brown adipose tissue (BAT) is a metabolically active fat that generates heat through uncoupling protein-1 (UCP-1). On FDG PET/CT, activated BAT appears as bilateral, symmetric areas of increased FDG uptake in characteristic locations: cervical, supraclavicular, axillary, mediastinal (paravertebral/paraspinal), and perirenal fat. The critical distinguishing feature is that the increased FDG uptake co-localizes with fat-density tissue on CT (Hounsfield units < −50). BAT activation is more common in women, young patients, low BMI individuals, and cold environments. Prevention strategies: keep the patient warm during the uptake period (blankets, warm room at 72–75°F), and consider propranolol (20 mg PO 60 min before injection) or diazepam in cases with recurrent prominent BAT that limits interpretation. BAT can mimic or obscure cervical/supraclavicular lymphadenopathy, which is a particular concern in head and neck cancer and lymphoma staging.
1. Confirm fasting ≥4 hr, check blood glucose (<200 mg/dL). 2. Inject 10–15 mCi (370–555 MBq) F-18 FDG IV. 3. Patient rests quietly in warm, dim room for 60 min. 4. Void bladder immediately before scanning. 5. Low-dose CT scout → CT acquisition (for attenuation correction ± diagnostic) → PET acquisition (2–3 min/bed position, skull base to mid-thigh standard; vertex to toes if melanoma or lower extremity concern). 6. Total scan time ~20–30 min. 7. Review fused PET/CT and MIP (maximum intensity projection) images.
06 FDG PET/CT — Oncologic Applications
Initial Staging & Restaging
FDG PET/CT is a cornerstone of oncologic staging. It detects unsuspected distant metastases in 10–30% of patients with various cancers, upstaging disease and avoiding futile surgery. Its principal value lies in whole-body assessment in a single examination. PET/CT is superior to CT alone for nodal staging (size criteria miss micrometastases but FDG detects metabolically active nodes) and for detection of osseous metastases (lytic > blastic). For restaging after therapy, PET/CT distinguishes viable tumor from post-treatment fibrosis or necrosis, which may appear identical on CT.
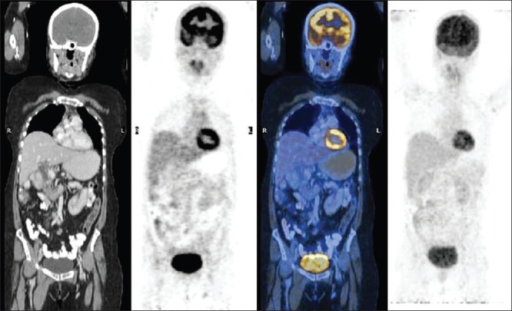
Response Assessment — PERCIST Criteria
The PET Response Criteria in Solid Tumors (PERCIST) uses SUL (SUV corrected for lean body mass) to classify treatment response: Complete metabolic response (CMR) — FDG uptake in all target lesions ≤ mean liver activity + 2 SD, no new FDG-avid lesions. Partial metabolic response (PMR) — ≥30% decrease in SULpeak of target lesion. Stable metabolic disease (SMD) — neither PMR nor PMD criteria met. Progressive metabolic disease (PMD) — ≥30% increase in SULpeak, or new FDG-avid lesions.
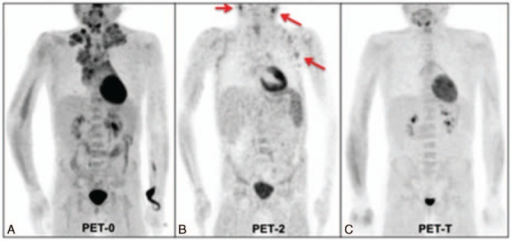
Deauville 5-Point Scale (Lymphoma)
The Deauville score is the standard for interim and end-of-treatment PET response assessment in FDG-avid lymphomas (Hodgkin, DLBCL, and other aggressive NHL). Scores are assigned by visual comparison of residual lesion uptake to reference structures:
| Score | FDG Uptake | Interpretation |
|---|---|---|
| 1 | No uptake above background | Complete metabolic response |
| 2 | Uptake ≤ mediastinal blood pool | Complete metabolic response |
| 3 | Uptake > mediastinal but ≤ liver | Complete metabolic response (most protocols) |
| 4 | Uptake moderately > liver | Residual metabolically active disease |
| 5 | Uptake markedly > liver and/or new lesions | Residual metabolically active disease |
| X | New areas unlikely related to lymphoma | Requires clinical correlation |
Common Malignancies — FDG PET/CT Role
| Malignancy | Primary PET/CT Indications | Key Considerations |
|---|---|---|
| NSCLC | Initial staging (mediastinum, distant mets); restaging; SPN characterization | PET-positive mediastinal nodes still require tissue confirmation; 80% sensitivity, 90% specificity for N2/N3 disease |
| Hodgkin lymphoma | Staging (Lugano); interim response (Deauville after 2 cycles); end-of-treatment | Replaces bone marrow biopsy if PET shows multifocal osseous involvement; Deauville 1–3 = CMR |
| DLBCL | Staging; end-of-treatment response; suspected transformation from low-grade | Deauville scoring; high SUV in follicular lymphoma suggests transformation to DLBCL |
| Melanoma | Stage III–IV staging; restaging; detection of in-transit/distant metastases | High sensitivity for soft tissue, nodal, and visceral mets; limited for brain mets (use MRI) |
| Colorectal cancer | Hepatic metastases detection; rising CEA with negative CT; recurrence | Mucinous histology may be FDG-negative (false negative) |
| Head & neck SCC | Staging; unknown primary detection (PET-guided biopsy); post-chemoRT response (at 12 weeks) | Negative PET at 12 weeks post-treatment has >95% NPV for residual disease |
| Esophageal cancer | Initial staging; response assessment after neoadjuvant therapy | Detects unsuspected distant disease in ~15% of patients, changing management |
| Breast cancer | Staging locally advanced/metastatic; treatment response monitoring | Not for primary detection or screening; lobular carcinoma may be less FDG-avid |
| Cervical cancer | Staging ≥IB2; detecting para-aortic nodal involvement; radiation planning | Sensitivity for para-aortic nodes ~75%; helps guide extended-field radiation |
| Pancreatic cancer | Staging; distinguishing malignancy from chronic pancreatitis (limited) | Chronic pancreatitis can be FDG-avid (false positive); autoimmune pancreatitis mimics cancer |
| Testicular cancer (seminoma) | Post-chemotherapy residual mass assessment | Residual mass >3 cm: PET differentiates viable tumor from necrosis/fibrosis |
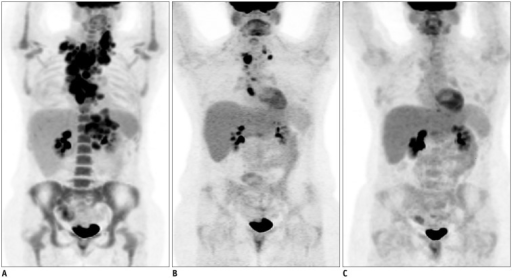
Important limitations: FDG PET is generally not recommended for: primary prostate cancer staging (use PSMA PET), well-differentiated NET staging (use Ga-68 DOTATATE), hepatocellular carcinoma initial staging (variable FDG avidity), multiple myeloma initial staging (variable, use whole-body low-dose CT or MRI), or brain metastases (high background cortical FDG uptake limits detection; MRI with contrast is superior).
False Positives & False Negatives
False Positives & False Negatives
False positives (FDG uptake mimicking malignancy):
| Cause | Mechanism | How to Distinguish |
|---|---|---|
| Infection / abscess | Activated neutrophils and macrophages are highly glycolytic | Clinical history, inflammatory markers, CT morphology |
| Granulomatous disease (sarcoid, TB, fungal) | Activated macrophages in granulomas | Distribution pattern, calcification, endemic exposure |
| Post-surgical inflammation | Healing and immune response | Wait ≥4–6 weeks post-surgery |
| Post-radiation inflammation | Radiation pneumonitis/mucositis | Wait ≥8–12 weeks post-RT; linear field margins suggest RT change |
| Brown fat activation | UCP-1 thermogenesis in BAT | Bilateral/symmetric; co-localizes with fat density on CT |
| Muscle uptake | Tension, recent exercise, anxiety (neck/shoulder) | Linear/symmetric pattern matching muscle anatomy |
| Thymic rebound | Post-chemotherapy immune reconstitution | Triangular anterior mediastinal shape; young patients |
| G-CSF effect | Marrow stimulation | Diffuse marrow and splenic uptake; history of recent G-CSF |
| Talc pleurodesis | Foreign body granulomatous reaction | Pleural distribution; history of pleurodesis (may persist years) |
False negatives (tumors with low FDG avidity): bronchoalveolar carcinoma (lepidic-predominant adenocarcinoma), mucinous adenocarcinomas (ovarian, appendiceal, gastric — mucin-producing tumors have low cellularity), well-differentiated neuroendocrine tumors (use Ga-68 DOTATATE instead), renal cell carcinoma (variable FDG avidity), low-grade lymphomas (follicular grade 1–2, marginal zone, SLL/CLL), hepatocellular carcinoma (variable, ~50% FDG-avid — glucose-6-phosphatase in hepatocytes dephosphorylates FDG-6-P), prostate cancer (low glycolytic rate in most tumors — use PSMA PET instead), and small lesions (<7–10 mm may be below the PET spatial resolution limit, producing partial volume effect with underestimation of true activity).
After chemotherapy: wait ≥2–3 weeks (ideally after completing a full cycle). After radiation therapy: wait ≥8–12 weeks to allow inflammation to subside (some guidelines recommend 12 weeks for head/neck cancers). After surgery: wait ≥4–6 weeks. After G-CSF administration: wait ≥2 weeks (G-CSF causes diffuse marrow FDG uptake and splenic enlargement). After talc pleurodesis: intense granulomatous FDG uptake may persist for years.
07 FDG PET/CT — Non-Oncologic Applications
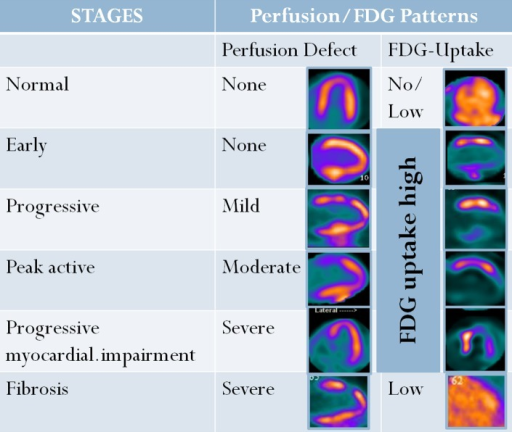
Cardiac Viability Assessment
FDG PET assesses myocardial viability by detecting preserved glucose metabolism in dysfunctional myocardium. The paradigm compares perfusion (using Rb-82 or N-13 ammonia PET, or Tc-99m SPECT) with FDG metabolism:
| Pattern | Perfusion | FDG Metabolism | Interpretation | Clinical Action |
|---|---|---|---|---|
| Normal | Normal | Normal | Normal myocardium | No intervention needed in that territory |
| Mismatch | Decreased | Preserved or increased | Hibernating myocardium (viable) | Revascularization likely to improve function |
| Match | Decreased | Proportionally decreased | Scar / infarction (nonviable) | Revascularization unlikely to recover function |
| Reverse mismatch | Normal or mildly reduced | Decreased | Stunned or repetitively ischemic myocardium | Consider revascularization |
For the viability protocol, patients fast, then receive an oral glucose load (25–50 g) followed by insulin if needed to shift cardiac metabolism toward glucose; alternatively, a glucose-insulin clamp may be used. Diabetic patients may require IV insulin titration to achieve adequate myocardial glucose utilization. The nicotinic acid derivative (Acipimox) protocol is an alternative: Acipimox suppresses free fatty acids, forcing the myocardium to use glucose, and may be useful in diabetic patients. Clinical significance: the presence of ≥7% hibernating myocardium (by PET viability) identifies patients with ischemic cardiomyopathy who are most likely to benefit from revascularization with improvement in LVEF and clinical outcomes.
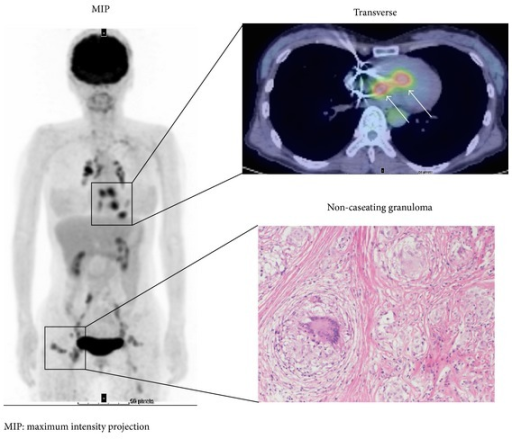
Cardiac Sarcoidosis
FDG PET detects active myocardial inflammation in cardiac sarcoidosis. The critical preparation is a prolonged fast (≥18 hours) with a preceding day of high-fat, very-low-carbohydrate diet (<5 g carbs) and optional IV heparin (50 units/kg 15 min pre-injection) to completely suppress normal myocardial FDG uptake. Without adequate suppression, physiologic myocardial uptake obscures pathologic inflammation. Focal or focal-on-diffuse FDG uptake in the myocardium, particularly when co-localized with perfusion defects, is highly suggestive of active cardiac sarcoidosis. FDG PET is used for diagnosis, monitoring treatment response (steroid therapy), and guiding decisions about ICD implantation. Sensitivity is approximately 89% and specificity 78% when adequate myocardial suppression is achieved.
Large-Vessel Vasculitis
FDG PET/CT detects active inflammation in the aortic wall and major branches in giant cell arteritis (GCA) and Takayasu arteritis. Smooth, linear, circumferential FDG uptake in the arterial wall (grade ≥2 vs liver) is characteristic. PET/CT is particularly valuable for detecting extracranial GCA involvement (aortitis, subclavian/axillary involvement) and for monitoring treatment response. A meta-analysis shows pooled sensitivity of 90% and specificity of 98% for large-vessel vasculitis. Notably, atherosclerotic plaques can cause patchy, focal arterial uptake, which must be distinguished from the smooth pattern of vasculitis. A vascular grading system compares arterial wall uptake to liver: grade 0 = none; grade 1 = < liver; grade 2 = similar to liver; grade 3 = > liver. Grades ≥2 are considered consistent with active vasculitis. The timing of PET relative to steroid initiation is important: sensitivity decreases after 3–10 days of glucocorticoid therapy, so PET should ideally be performed before or within the first 72 hours of treatment.
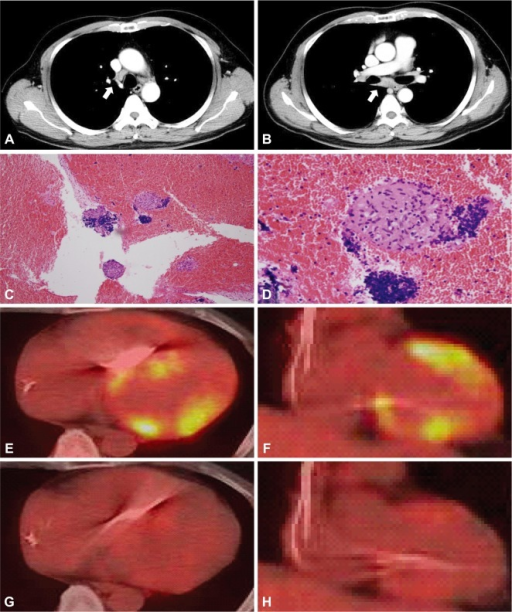
Sarcoidosis Assessment
Beyond cardiac sarcoidosis (section 7), FDG PET/CT is valuable for whole-body assessment of sarcoidosis activity. Active sarcoid granulomas are metabolically active and FDG-avid, producing the characteristic findings: bilateral hilar and mediastinal lymphadenopathy, pulmonary parenchymal nodules, extrapulmonary involvement (liver, spleen, parotid/lacrimal glands, skin, bones, CNS). The classic patterns on whole-body PET include the "lambda sign" (bilateral hilar + right paratracheal lymphadenopathy forming a lambda shape, analogous to the Ga-67 finding) and the "panda sign" (bilateral lacrimal and parotid gland uptake). FDG PET is used to: (1) identify active disease requiring treatment versus burned-out fibrotic disease; (2) guide biopsy to the most active site; (3) monitor treatment response (decreasing FDG uptake indicates successful immunosuppression); and (4) identify cardiac, CNS, or other occult organ involvement.
Infection Imaging
FDG PET/CT is increasingly used for: prosthetic valve endocarditis (modified Duke criteria include abnormal PET activity around a prosthetic valve as a major criterion — focal or heterogeneous FDG uptake around a prosthetic valve is considered abnormal, while homogeneous mild uptake may be nonspecific post-surgical change in the first 3 months), prosthetic vascular graft infection (focal intense uptake vs normal mild diffuse uptake of a healing graft — timing matters: low-grade diffuse graft uptake is common and normal in Dacron grafts but may persist for years; focal intense uptake suggests infection), prosthetic joint infection (though specificity is limited by post-surgical inflammation in the first 3–6 months), spondylodiscitis (FDG PET is the nuclear study of choice for spinal infections because WBC scans are unreliable in the axial skeleton), and fever of unknown origin (FUO) — PET/CT identifies the source in 30–50% of FUO cases after conventional workup has failed. The diagnostic yield of FDG PET in FUO is highest when inflammatory markers (CRP, ESR) are elevated. FDG PET/CT is also used to diagnose mycotic aneurysms (infected arterial aneurysms), infected central lines and device leads (pacemaker/ICD lead endocarditis), and peripancreatic collections in the setting of post-surgical or post-inflammatory complications.
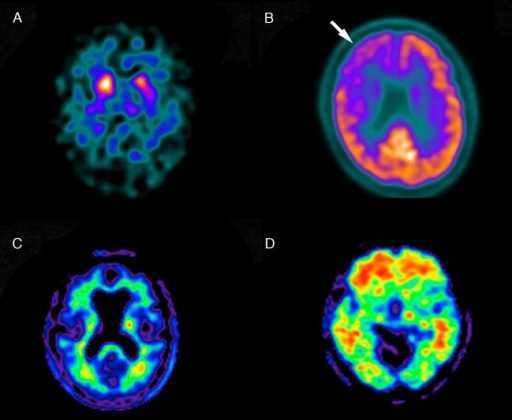
Neurologic Applications
Epilepsy: Interictal FDG PET shows hypometabolism in the epileptogenic zone, aiding pre-surgical lateralization and localization in temporal lobe epilepsy. The hypometabolic region is typically larger than the structural MRI abnormality. Concordance between MRI (hippocampal sclerosis), EEG, and FDG PET hypometabolism provides the strongest evidence for surgical resection. In MRI-negative temporal lobe epilepsy, FDG PET correctly lateralizes the seizure focus in ~70–80% of cases. For extratemporal epilepsy, FDG PET is less sensitive (~50–60%), and ictal SPECT/SISCOM may be more informative.
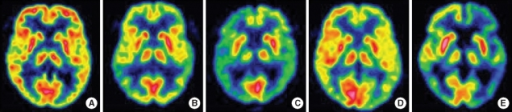
Dementia: Characteristic FDG PET patterns include bilateral temporoparietal hypometabolism with posterior cingulate involvement (early and prominent) in Alzheimer disease, frontal and/or anterior temporal hypometabolism in frontotemporal dementia, and occipital hypometabolism (including primary visual cortex, which is characteristically spared in AD) in dementia with Lewy bodies. FDG PET has ~90% sensitivity and ~70% specificity for distinguishing AD from normal aging. The posterior cingulate cortex is one of the earliest regions affected in AD and can show hypometabolism even in the mild cognitive impairment (MCI) stage. In corticobasal degeneration, asymmetric frontoparietal hypometabolism is seen. In primary progressive aphasia, left perisylvian hypometabolism predominates.
08 Non-FDG PET Tracers
F-18 Sodium Fluoride (NaF)
F-18 NaF is a bone-seeking PET tracer that localizes to areas of active bone remodeling by exchanging with hydroxyl ions on the surface of hydroxyapatite crystals. It offers superior sensitivity and spatial resolution compared to Tc-99m MDP bone scan for detecting osseous metastases, particularly lytic and mixed lesions. Uptake reflects regional blood flow and osteoblastic activity. The combination of F-18 NaF with PET/CT allows precise anatomic localization of foci, distinguishing degenerative changes from metastatic disease with greater confidence than planar bone scintigraphy. Unlike FDG, F-18 NaF has minimal soft-tissue uptake and no brain uptake, providing excellent bone-to-background contrast. Dose: typically 5–10 mCi (185–370 MBq) IV, imaging 45–60 min post-injection. F-18 NaF PET/CT detects 2–3× more bone metastases than conventional bone scintigraphy in head-to-head comparisons, with particular advantage for lytic lesions, spine metastases, and small metastases in the pelvis and ribs.
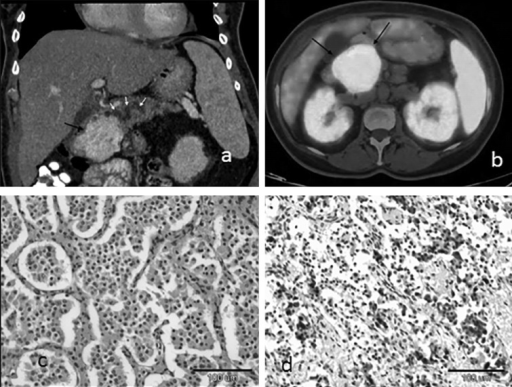
Ga-68 DOTATATE (Neuroendocrine Tumors)
Ga-68 DOTATATE PET/CT targets somatostatin receptor subtype 2 (SSTR2), which is overexpressed on well-differentiated neuroendocrine tumors (NETs). It has largely replaced In-111 octreotide (OctreoScan) SPECT due to superior resolution, sensitivity (>95% vs ~70%), and convenience (1–2 hour scan vs 24–48 hour protocol). The Krenning score grades uptake: 0 = no uptake; 1 = very low (below normal liver); 2 = ≤ normal liver; 3 = > normal liver; 4 = > normal spleen (highest physiologic uptake). Krenning ≥2 is generally required for selecting patients for peptide receptor radionuclide therapy (PRRT) with Lu-177 DOTATATE. Normal intense uptake occurs in spleen (highest), adrenals, pituitary, liver, kidneys, and the uncinate process of the pancreas (an important pitfall — do not mistake for a pancreatic NET).
Amyloid PET Tracers
Three FDA-approved amyloid PET tracers detect beta-amyloid neuritic plaques in the brain: F-18 florbetapir (Amyvid), F-18 florbetaben (Neuraceq), and F-18 flutemetamol (Vizamyl). A positive scan shows loss of the normal gray-white matter contrast as cortical binding increases. A negative amyloid PET essentially excludes Alzheimer disease as the cause of cognitive impairment (high negative predictive value). However, a positive scan does not diagnose AD — amyloid deposition increases with age (10–30% of cognitively normal elderly are amyloid-positive). Amyloid PET is most useful in patients with atypical presentations, early-onset dementia, or persistent diagnostic uncertainty despite standard workup.
Appropriate indications: (1) Persistent or progressive unexplained MCI after comprehensive evaluation. (2) Patients satisfying core clinical criteria for possible AD but with an atypical clinical course or etiologically mixed presentation. (3) Progressive dementia with atypically early age of onset (<65 years). Inappropriate indications: (1) Patients who meet core clinical criteria for probable AD with typical age of onset (high pretest probability). (2) To determine dementia severity (amyloid burden does not correlate well with severity). (3) Asymptomatic patients or those with only subjective cognitive complaints without objective deficits. (4) Non-medical purposes (insurance, legal). (5) Based solely on family history or APOE4 genotype. A positive amyloid PET in the context of anti-amyloid immunotherapy (lecanemab, donanemab, aducanumab) confirms the presence of the therapeutic target and is increasingly used for treatment eligibility.
Brain Tumor Tracers
F-18 FET (fluoroethyltyrosine) and F-18 FDOPA are amino acid PET tracers used for brain tumor imaging. They have low uptake in normal brain cortex (unlike FDG, which has high physiologic cortical uptake), providing superior tumor-to-background contrast. Applications include differentiating tumor recurrence from radiation necrosis, delineating tumor extent for surgical/radiation planning, and grading gliomas. FDOPA is also used for evaluating dopaminergic function in Parkinson disease and for localizing focal cortical dysplasia in epilepsy.
Prostate Cancer PET Tracers
The evolution of prostate cancer PET tracers: C-11 choline (11-min half-life, limited to centers with on-site cyclotron) detects phospholipid membrane synthesis. F-18 fluciclovine (Axumin), a synthetic amino acid, was widely used for biochemical recurrence but has been largely supplanted by PSMA agents. PSMA (prostate-specific membrane antigen) PET is now the dominant modality: Ga-68 PSMA-11 and F-18 piflufolastat (Pylarify, DCFPyL) target the extracellular domain of PSMA, which is overexpressed in >90% of prostate cancers. PSMA PET detects recurrence at very low PSA levels (even <0.5 ng/mL), identifies oligometastatic disease amenable to focal therapy, and guides theranostic selection for Lu-177 PSMA therapy. Normal PSMA uptake occurs in lacrimal glands, salivary glands (parotid > submandibular), liver, spleen, small bowel, kidneys, and proximal ureters.
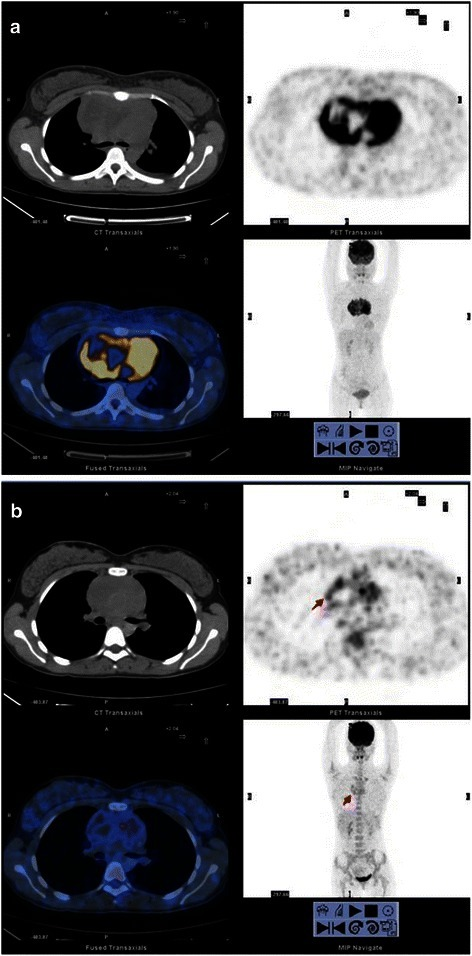
PSMA PET Interpretation Pitfalls
Important false positives and pitfalls on PSMA PET: Ganglia (celiac, stellate, sacral) show moderate PSMA uptake and may mimic lymph node metastases; correlation with CT morphology is essential. Benign osseous lesions (Paget disease, healing fractures, fibrous dysplasia) may show mild PSMA uptake. Other malignancies expressing PSMA: renal cell carcinoma neovasculature, hepatocellular carcinoma, some high-grade gliomas, and follicular thyroid cancer — awareness is important when PSMA PET is performed in patients with a history of non-prostate malignancy. Ureter activity can obscure pelvic or retroperitoneal lymph nodes; delayed images or furosemide-enhanced images can improve interpretation. F-18 DCFPyL has the advantage of a longer half-life than Ga-68 PSMA-11, allowing centralized production and wider distribution, and produces slightly higher-quality images due to the shorter positron range of F-18.
Cu-64 DOTATATE
Cu-64 DOTATATE (Detectnet) is an FDA-approved PET tracer for NET imaging with a longer half-life (12.7 hours) than Ga-68 (68 min), enabling centralized production and wider distribution. It provides comparable diagnostic performance to Ga-68 DOTATATE for localization and staging of somatostatin receptor–positive NETs.
Tau PET Tracers
F-18 flortaucipir (Tauvid) is the first FDA-approved tau PET tracer, binding to aggregated tau neurofibrillary tangles in the brain. Unlike amyloid PET (which detects plaques common to both AD and normal aging), tau PET more closely correlates with clinical symptoms and cognitive decline in Alzheimer disease. The pattern of tau deposition follows Braak staging: earliest in the medial temporal lobe (entorhinal cortex, hippocampus), then spreading to the lateral temporal and parietal cortices, and finally to frontal regions. Tau PET is particularly useful for distinguishing AD from other tauopathies and for predicting the trajectory of cognitive decline. The combination of amyloid PET (for diagnosis) and tau PET (for staging/prognosis) provides a comprehensive in vivo assessment of AD pathology.
Emerging PET Tracers
The field of PET tracer development is rapidly expanding: F-18 flotufolastat (Posluma) is another PSMA PET agent with high sensitivity for prostate cancer. Ga-68 FAPI (fibroblast activation protein inhibitor) targets cancer-associated fibroblasts present in >90% of epithelial cancers; it shows minimal background uptake in the brain, liver, and bone marrow, providing exceptional contrast for many tumor types where FDG has limitations (e.g., peritoneal carcinomatosis, gastric cancer, cholangiocarcinoma). FAPI PET also shows promise in non-oncologic fibrotic conditions (cardiac fibrosis, pulmonary fibrosis, Crohn disease). Zr-89 labeled antibodies (immuno-PET) image specific antigens in vivo (e.g., Zr-89 trastuzumab for HER2 imaging) but require long wait times (3–7 days post-injection) due to slow antibody pharmacokinetics. Additional emerging agents include C-11 methionine (amino acid PET for brain tumors, particularly useful for distinguishing recurrence from radiation necrosis), F-18 FACBC (fluciclovine) for amino acid transport imaging, and Cu-64 trastuzumab for HER2-positive breast cancer imaging. The trend in radiopharmaceutical development is toward increasingly specific molecular targets, enabling the theranostic paradigm to extend beyond NETs and prostate cancer to breast cancer (HER2-targeting), colorectal cancer (CEA-targeting), and beyond.
09 Myocardial Perfusion Imaging — Stress Protocols
Indications for Stress MPI
The principal indications for myocardial perfusion imaging include: (1) diagnosis of obstructive CAD in patients with intermediate pretest probability and uninterpretable or equivocal ECG stress test; (2) risk stratification in known or suspected CAD; (3) evaluation of chest pain syndromes; (4) preoperative cardiac risk assessment before high-risk non-cardiac surgery in patients with clinical risk factors and poor functional capacity (<4 METs); (5) assessment of myocardial viability in ischemic cardiomyopathy to guide revascularization decisions; (6) evaluation of new or worsening symptoms in patients with previously documented CAD. MPI is generally not indicated for screening asymptomatic, low-risk patients without known CAD (appropriate use criteria).
Exercise Stress
Exercise stress (treadmill or bicycle) is the preferred modality when the patient can exercise adequately, because it provides prognostic information beyond perfusion (exercise capacity, hemodynamic response, symptoms, ECG changes). The Bruce protocol is most commonly used, with stages increasing in speed and grade every 3 minutes: Stage 1 = 1.7 mph / 10% grade; Stage 2 = 2.5 mph / 12%; Stage 3 = 3.4 mph / 14%; Stage 4 = 4.2 mph / 16%; Stage 5 = 5.0 mph / 18%. Target heart rate is 85% of age-predicted maximum (220 − age). The radiotracer is injected at peak stress, and the patient continues exercising for 1–2 additional minutes to allow myocardial extraction. Exercise capacity itself is a powerful prognostic indicator: achieving ≥10 METs (Stage 4 Bruce) indicates excellent functional capacity and low cardiac risk regardless of perfusion results. Failure to reach Stage 2 (<5 METs) is a marker of poor prognosis. The Duke treadmill score = exercise time (minutes) − (5 × max ST deviation in mm) − (4 × angina index [0=none, 1=non-limiting, 2=limiting]). Score ≥5 = low risk; −10 to +4 = intermediate risk; ≤ −11 = high risk.
Pharmacologic Stress — Vasodilators
Pharmacologic stress is indicated when patients cannot exercise adequately (unable to reach 85% MPHR, orthopedic limitations, deconditioning, LBBB, ventricular pacing). Adenosine directly activates A2A receptors on coronary smooth muscle, causing maximal coronary vasodilation. Dose: 140 µg/kg/min IV over 6 minutes. Contraindications: second- or third-degree AV block without pacemaker, sinus node disease, reactive airway disease (asthma/COPD with active wheezing), SBP <90 mmHg, recent (<12 hr) dipyridamole/caffeine use. Side effects: flushing, chest tightness, dyspnea, AV block, hypotension. Reversal: aminophylline 50–250 mg IV.
Regadenoson (Lexiscan) is a selective A2A receptor agonist given as a single fixed-dose bolus (0.4 mg/5 mL IV over 10 seconds), followed by saline flush and radiotracer injection 10–20 seconds later. Its selectivity for A2A over A1 (AV node), A2B (bronchial), and A3 receptors reduces side effects compared to adenosine. It is the most widely used pharmacologic stress agent in the US. While considered safer in mild asthma/COPD than adenosine, it is still contraindicated in severe reactive airway disease. Reversal: aminophylline 75–250 mg IV (though effects are transient, ~2–4 minutes).
Dipyridamole (Persantine) inhibits adenosine deaminase, increasing endogenous adenosine levels. Dose: 0.56 mg/kg IV over 4 minutes. Same contraindications as adenosine. Longer duration of action (~15–30 min) means side effects persist longer. Reversal: aminophylline. Dipyridamole has been largely replaced by regadenoson in clinical practice.
Dobutamine Stress
Dobutamine is a synthetic catecholamine (β1 agonist) that increases heart rate, contractility, and myocardial oxygen demand, simulating exercise. Used when both exercise and vasodilator stress are contraindicated (e.g., severe reactive airway disease plus inability to exercise). Dose: start at 5–10 µg/kg/min, increase by 10 µg/kg/min every 3 minutes to maximum 40–50 µg/kg/min ± atropine 0.25–1.0 mg to achieve target heart rate. Contraindications: severe hypertension, unstable arrhythmias, aortic dissection, HOCM with significant obstruction. Side effects: tachyarrhythmias, hypertension, nausea. Reversal: short-acting beta-blocker (esmolol).
Stress Agent Selection Guide
| Clinical Scenario | Preferred Stress Agent | Rationale |
|---|---|---|
| Patient can exercise adequately | Exercise (Bruce protocol) | Best prognostic data; physiologic; ECG and hemodynamic data |
| Cannot exercise, no contraindications to vasodilators | Regadenoson (preferred) or adenosine | Selective A2A agonism; convenient fixed-dose bolus |
| LBBB or ventricular pacing | Vasodilator (regadenoson/adenosine) | Avoids false-positive septal defect from exercise-induced septal ischemia artifact |
| Severe reactive airway disease, cannot exercise | Dobutamine | Does not activate adenosine receptors in bronchial smooth muscle |
| Second- or third-degree AV block (no pacemaker) | Dobutamine | Vasodilators can worsen AV block via A1 receptor activation |
| Recent caffeine intake (<12 hr) | Exercise or dobutamine | Caffeine antagonizes vasodilator effect |
| Theophylline/aminophylline use | Exercise or dobutamine | Methylxanthines are adenosine receptor antagonists |
| Aortic stenosis (severe) | Vasodilator (with caution) | Avoid exercise (hemodynamic compromise); dobutamine relatively contraindicated |
Endpoints for Terminating Stress
Absolute endpoints for terminating exercise stress: (1) drop in SBP >10 mmHg from baseline with other evidence of ischemia, (2) moderate-to-severe angina, (3) increasing neurologic symptoms (ataxia, dizziness), (4) signs of poor perfusion (cyanosis, pallor), (5) sustained VT, (6) ST elevation ≥1 mm in leads without diagnostic Q waves, (7) patient request. Relative endpoints include: ST depression >2 mm, arrhythmias other than sustained VT, fatigue/shortness of breath/wheezing, SBP >250 mmHg or DBP >115 mmHg. For pharmacologic stress, the agent is stopped and reversed (aminophylline for vasodilators, esmolol for dobutamine) if severe symptoms, marked hemodynamic changes, or significant arrhythmias occur.
In patients with LBBB or ventricular-paced rhythm, exercise or dobutamine stress can cause a false-positive septal perfusion defect due to asynchronous septal contraction and altered septal blood flow — not true ischemia. Vasodilator stress (regadenoson, adenosine, dipyridamole) should be used in these patients to avoid this artifact. This is one of the highest-yield facts in cardiac nuclear medicine.
10 MPI — Radiopharmaceuticals & Acquisition
Tc-99m Sestamibi (Cardiolite) & Tc-99m Tetrofosmin (Myoview)
Both are lipophilic cationic complexes that enter myocardial cells passively via the mitochondrial membrane potential gradient. Uptake is proportional to regional myocardial blood flow and cell viability. Unlike Tl-201, these agents exhibit minimal redistribution (fixed distribution after extraction), so separate injections are needed for rest and stress images. One-day protocol: Low dose at rest (8–12 mCi) followed by high dose at stress (24–36 mCi) on the same day; the higher stress dose overcomes residual rest-dose activity ("shine-through"). Two-day protocol: Full dose (20–30 mCi) on each day, rest and stress on separate days; superior image quality but less convenient. Stress-first protocol: If stress images are completely normal, rest images can be omitted, saving time and reducing dose. Imaging is typically performed 30–60 minutes post-injection (allows hepatobiliary clearance, improving inferior wall visualization).
Tl-201 (Thallous Chloride)
Tl-201 is a potassium analog taken up by viable myocardial cells via the Na+/K+-ATPase pump. Its key property is redistribution: initial distribution reflects blood flow, but over 3–4 hours, Tl-201 washes out of normal myocardium and washes into ischemic (but viable) zones, "filling in" initial defects. A defect that is present on stress but fills in at 4 hours (or 24-hour delayed imaging) represents ischemia. A fixed defect (present on both stress and rest/redistribution) suggests infarction/scar, but some fixed defects improve with reinjection or 24-hour delayed imaging (indicating severely ischemic but viable tissue). Tl-201 has largely been replaced by Tc-99m agents for perfusion due to its suboptimal imaging characteristics (low-energy X-rays at 69–83 keV, longer half-life = higher radiation dose, poorer image quality), but it retains a niche role in viability assessment.
Gated SPECT
Gated SPECT acquires data synchronized to the ECG, dividing the cardiac cycle into 8 or 16 frames. This enables assessment of wall motion, wall thickening, and calculation of left ventricular ejection fraction (LVEF), end-diastolic volume (EDV), and end-systolic volume (ESV) from the same perfusion dataset. Normal LVEF by gated SPECT is ≥50%. Gating improves specificity by distinguishing true perfusion defects (associated with wall motion/thickening abnormalities) from attenuation artifacts (normal wall motion in the affected territory).
Dosing Protocols Summary
| Protocol | Rest Dose | Stress Dose | Imaging Timing | Advantage |
|---|---|---|---|---|
| 1-day rest-stress (Tc-99m) | 8–12 mCi | 24–36 mCi | 30–60 min post each injection | Convenient; completed same day |
| 1-day stress-rest (Tc-99m) | 24–36 mCi | 8–12 mCi | 15–45 min post stress; 3–4 hr later for rest | Can skip rest if stress normal |
| 2-day (Tc-99m) | 20–30 mCi (day 1) | 20–30 mCi (day 2) | 30–60 min post each injection | Best image quality; equal counts |
| Tl-201 stress-redistribution | N/A (redistribution) | 3–4 mCi at peak stress | Immediate stress + 4 hr redistribution | Viability assessment with redistribution |
| Tl-201 reinjection | 1 mCi reinjection at 4 hr | 3 mCi at peak stress | Immediate + 4 hr + post-reinjection | Detects additional viable tissue |
Pharmacokinetics of Tc-99m Agents
After IV injection, Tc-99m sestamibi achieves ~1.2% myocardial uptake (first-pass extraction ~65%) and Tc-99m tetrofosmin achieves ~1.0% uptake (first-pass extraction ~54%). Both agents show significant hepatobiliary excretion in the early post-injection period, which can overlap with the inferior wall of the heart on images. Waiting 30–60 minutes post-injection (or having the patient drink water or eat a small fatty snack to promote gallbladder contraction) improves inferior wall visualization by reducing subdiaphragmatic activity. For stress images, the higher myocardial blood flow during stress increases cardiac uptake, while exercise itself promotes hepatobiliary clearance, so imaging can proceed earlier (15–30 minutes post-injection) compared to rest (45–60 minutes).
11 MPI — Interpretation & Reporting
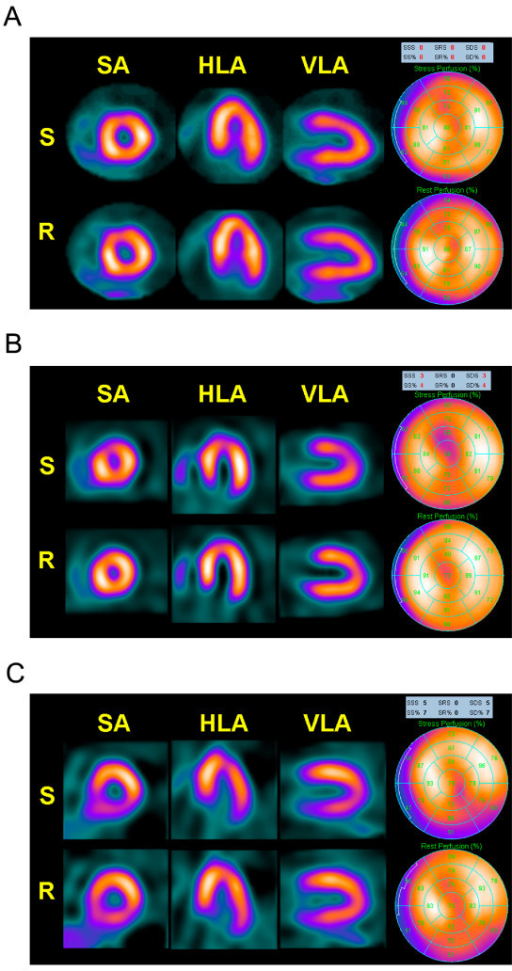
The 17-Segment Model
The AHA/ACC standardized 17-segment model divides the LV into 6 basal segments, 6 mid segments, 4 apical segments, and the apex (segment 17). Each segment is assigned to a coronary territory: the LAD supplies the anterior wall, anterior septum, and apex; the LCx supplies the lateral wall and inferolateral segments; the RCA supplies the inferior wall, inferior septum, and basal inferolateral segment (in right-dominant circulation). This mapping enables localization of perfusion defects to a culprit coronary artery.
5-Point Scoring System
| Score | Perfusion | Description |
|---|---|---|
| 0 | Normal | Homogeneous radiotracer uptake |
| 1 | Mildly reduced | Equivocal, possible mild decrease |
| 2 | Moderately reduced | Definite decrease, still visible uptake |
| 3 | Severely reduced | Markedly decreased, minimal uptake |
| 4 | Absent | No detectable radiotracer uptake (transmural defect) |
Summed Scores — SSS, SRS, SDS
The Summed Stress Score (SSS) is the sum of scores in all 17 segments on the stress image. The Summed Rest Score (SRS) is the same for rest images. The Summed Difference Score (SDS) = SSS − SRS, reflecting the amount of ischemia (reversibility). Clinical thresholds: SSS <4 = normal; SSS 4–8 = mildly abnormal; SSS 9–13 = moderately abnormal (intermediate risk); SSS ≥14 = severely abnormal (high risk). An SDS ≥7 (representing ≥10% ischemic myocardium) identifies patients who benefit most from revascularization over medical therapy. SRS reflects the burden of scar.
Transient Ischemic Dilation (TID)
TID is an apparent increase in LV cavity size on stress images compared to rest. A TID ratio >1.22 (for Tc-99m agents with exercise) is considered abnormal. TID suggests severe and extensive CAD (balanced ischemia or diffuse subendocardial ischemia) that causes post-stress LV stunning and dilation. TID is a high-risk finding associated with multivessel or left main disease, even when the perfusion images themselves appear near-normal.
Ancillary Findings
Increased RV uptake on stress images suggests RV pressure overload (pulmonary hypertension) or RV ischemia. Increased lung uptake on Tl-201 stress images (lung/heart ratio >0.5) indicates elevated pulmonary capillary wedge pressure during stress — a marker of severe, extensive CAD and poor prognosis. This finding is less reliable with Tc-99m agents. LV dilation on rest images with reduced EF indicates pre-existing cardiomyopathy.
Common Attenuation Artifacts
Breast attenuation in women produces an apparent anterior wall defect (fixed, present on both stress and rest). Diaphragmatic attenuation in men (particularly obese) produces an apparent inferior wall defect (fixed). These artifacts are distinguished from true perfusion defects by: (1) normal wall motion on gated images, (2) correction with CT-based attenuation correction, (3) prone imaging (shifts the heart and changes the attenuation pattern — an artifact will change while a true defect persists).
Attenuation artifact: Fixed defect, normal wall motion on gating, resolves with attenuation correction or changes with prone imaging. Infarction (scar): Fixed defect with corresponding wall motion abnormality on gating, persists with attenuation correction. Ischemia: Reversible defect (present on stress, improved/resolved on rest), with stress-induced wall motion abnormality on gated stress images.
Coronary Territory Mapping
| Territory | Segments (17-Segment Model) | Typical Findings |
|---|---|---|
| LAD | Basal anterior, basal anteroseptal, mid anterior, mid anteroseptal, apical anterior, apical septal, apex | Anterior and septal wall defects; apical involvement in proximal LAD lesions |
| LCx | Basal anterolateral, basal inferolateral, mid anterolateral, mid inferolateral, apical lateral | Lateral wall defects; may extend inferolaterally |
| RCA | Basal inferior, basal inferoseptal, mid inferior, mid inferoseptal, apical inferior | Inferior wall defects; RV involvement in proximal RCA lesions |
Prognostic Value of MPI
A normal stress MPI study confers an excellent prognosis with an annual cardiac event rate (MI or cardiac death) of <1% per year — this "warranty period" lasts approximately 2 years for most patients (shorter in diabetics and patients with known CAD). The warranty supports the guideline recommendation against repeat testing within 2 years if the initial study is normal without a change in clinical status. Conversely, a severely abnormal study (SSS ≥14 or SDS ≥7 with ≥10% ischemic myocardium) confers an annual event rate of ≥5%, identifying patients who benefit from aggressive intervention including coronary angiography and revascularization over optimal medical therapy alone.
12 MUGA Scan & Cardiac PET
MUGA (Multigated Acquisition) Scan
The MUGA scan (radionuclide ventriculography, RVG) measures LVEF by labeling red blood cells with Tc-99m and acquiring gated images of the cardiac blood pool. The ECG-gated acquisition divides the cardiac cycle into 16–24 frames, producing a cine loop. LVEF is calculated from the change in LV counts between end-diastole and end-systole: EF = (EDcounts − EScounts) / EDcounts. Normal LVEF ≥50%. The MUGA scan is highly reproducible (coefficient of variation ~3–5%) and is the gold standard for serial monitoring of LVEF in patients receiving cardiotoxic chemotherapy (anthracyclines — doxorubicin, daunorubicin; trastuzumab). A decline in LVEF of ≥10 percentage points to below 50% is generally the threshold for holding or modifying cardiotoxic therapy. RBC labeling can be in vivo (inject stannous pyrophosphate, wait 20 min, inject Tc-99m pertechnetate), modified in vivo, or in vitro (most accurate, labeling efficiency >97%).
Cardiac PET Perfusion
Rb-82 (rubidium-82) is a potassium analog produced by an on-site Sr-82/Rb-82 generator (no cyclotron needed). Its ultra-short half-life (75 seconds) requires pharmacologic stress only (no time for exercise protocol) and allows rest and stress imaging within 30 minutes total. N-13 ammonia (T½ 10 min, cyclotron-produced) provides superior image quality and can be used with exercise stress but requires an on-site cyclotron. Both enable absolute quantification of myocardial blood flow (MBF) in mL/g/min and calculation of coronary flow reserve (CFR) = stress MBF / rest MBF. Normal CFR is ≥2.0; reduced CFR (<2.0) identifies patients with hemodynamically significant CAD, microvascular disease, or balanced ischemia (which may appear normal on relative perfusion images).
F-18 Flurpiridaz
F-18 flurpiridaz is an FDA-approved PET myocardial perfusion agent that binds to mitochondrial complex I. Its advantages include: the 110-minute half-life of F-18 (allows exercise stress, unlike Rb-82), no need for an on-site cyclotron or generator, high myocardial extraction fraction (~94%, superior to Rb-82 and Tc-99m agents), enabling accurate absolute flow quantification even at high flow rates. This agent is expected to significantly expand access to cardiac PET perfusion imaging.
PET advantages: Higher spatial resolution (4–5 mm vs 12–15 mm), robust attenuation correction, absolute flow quantification, higher sensitivity for multivessel CAD, lower radiation dose for Rb-82 protocols. SPECT advantages: Lower cost, wider availability, exercise stress capability, established prognostic databases, can use existing gamma camera infrastructure.
13 Thyroid Scintigraphy & Uptake
Radiopharmaceuticals for Thyroid Imaging
I-123 is the preferred agent for thyroid imaging: it is organified (trapped and incorporated into thyroglobulin like stable iodine), emits a 159-keV gamma ideal for gamma camera imaging, and has a 13.2-hour half-life limiting radiation dose to the thyroid. Tc-99m pertechnetate is trapped by the sodium-iodide symporter (NIS) like iodide but is not organified; it provides images of trapping only. Tc-99m pertechnetate has the advantage of lower cost and immediate availability (from the Tc-99m generator) with imaging at 20 minutes post-injection. A key difference: a nodule that appears "warm" on Tc-99m pertechnetate may be "cold" on I-123 (trapping without organification — seen in ~5% of cases, some of which are malignant). I-131 is generally not used for diagnostic thyroid imaging due to its high radiation dose (beta emission) and suboptimal 364-keV gamma for camera imaging; however, post-therapy I-131 whole-body scans are standard.
Radioactive Iodine Uptake (RAIU)
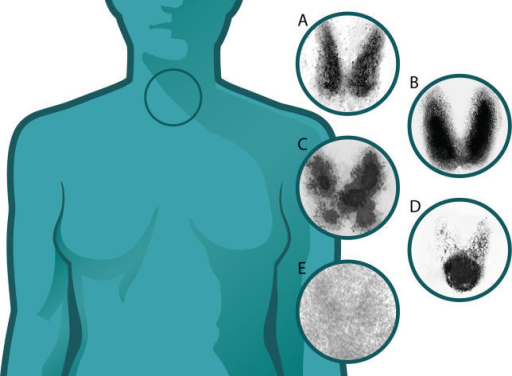
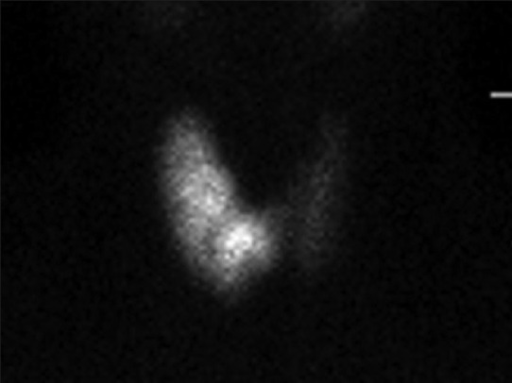
The RAIU test measures the percentage of an administered oral dose of I-123 (or I-131) taken up by the thyroid at 4–6 hours and 24 hours. Normal 24-hour uptake is 10–35% (varies regionally with dietary iodine intake). Uptake patterns in hyperthyroidism:
| Condition | RAIU | Scan Pattern |
|---|---|---|
| Graves disease | Diffusely increased (40–80%+) | Homogeneous, enlarged gland, pyramidal lobe often visible |
| Toxic multinodular goiter | Normal to increased | Heterogeneous, multiple hot and cold areas |
| Toxic adenoma (Plummer disease) | Normal to increased | Single hot nodule with suppressed surrounding tissue |
| Subacute thyroiditis (de Quervain) | Very low (<5%) | Faint or absent uptake; preformed hormone release |
| Painless/postpartum thyroiditis | Very low | Faint or absent uptake |
| Exogenous thyroid hormone (factitious) | Very low | Suppressed gland |
| Iodine excess (Jod-Basedow or contamination) | Very low | Suppressed by iodine load |
| Struma ovarii | Low (thyroid), uptake in pelvis | Ectopic thyroid tissue in ovarian teratoma |
Thyroid Nodule Assessment
On thyroid scintigraphy, nodules are classified by their uptake relative to surrounding normal thyroid tissue: Hot (hyperfunctioning) nodules concentrate radiotracer more intensely and often suppress the surrounding parenchyma; they are almost always benign (<1% malignancy risk) and do not require FNA. Warm (isofunctioning) nodules have uptake similar to surrounding tissue; low malignancy risk (~5%). Cold (hypofunctioning) nodules have decreased or absent uptake relative to surrounding tissue; these carry the highest malignancy risk (~5–15%) and typically warrant further evaluation with ultrasound and FNA biopsy per Bethesda/ACR TI-RADS criteria.
Medications Affecting Thyroid Uptake
Several medications and substances interfere with thyroid scintigraphy and RAIU: Iodinated contrast (CT contrast) saturates iodine pools and suppresses uptake for 4–8 weeks. Amiodarone contains 37% iodine by weight and has a half-life of 40–100 days; it may suppress uptake for 3–6 months or longer after discontinuation. Levothyroxine suppresses TSH and reduces RAIU (stop 4–6 weeks before if uptake measurement is needed). Antithyroid drugs (methimazole, propylthiouracil) reduce organification but increase trapping — I-123 images may appear normal but Tc-99m pertechnetate images remain hot in Graves (demonstrating the trapping-organification discordance). Kelp, seaweed, betadine, and other iodine-containing supplements can suppress uptake. A careful medication and supplement history is essential before thyroid scintigraphy.
Thyroid Cancer Follow-Up Strategy
After initial surgery and RAI ablation, surveillance for differentiated thyroid cancer includes serial thyroglobulin (Tg) levels (tumor marker — should be undetectable after total thyroidectomy and ablation), Tg antibodies (interfere with Tg assays; if present, trending the antibody level serves as a surrogate marker), neck ultrasound, and diagnostic whole-body I-131 or I-123 scan in selected patients. A rising Tg with negative anatomic imaging is an indication for FDG PET/CT, which can localize recurrent or metastatic disease that has lost the ability to concentrate iodine (dedifferentiated, iodine-refractory disease — a poor prognostic sign). The phenomenon of a "flip-flop" where tumors become FDG-avid as they lose iodine avidity reflects dedifferentiation and guides the transition from RAI therapy to external beam radiation or systemic therapy (tyrosine kinase inhibitors).
14 Radioiodine Therapy — I-131
I-131 for Graves Disease
Radioactive iodine (RAI) with I-131 is a definitive treatment for Graves disease. The typical dose is 10–15 mCi (370–555 MBq) administered as a single oral capsule. The I-131 is trapped and organified by the hyperactive thyroid, delivering a high local radiation dose (target 10,000–15,000 rad to the gland) that destroys follicular cells over weeks to months. Most patients become hypothyroid within 2–6 months and require lifelong levothyroxine replacement. Contraindications: pregnancy (absolute — I-131 crosses the placenta and ablates fetal thyroid after 10–12 weeks gestation), breastfeeding, inability to comply with radiation safety precautions. Graves ophthalmopathy may worsen after RAI, particularly in smokers; glucocorticoid prophylaxis (prednisone 0.3–0.5 mg/kg/day) should be considered in patients with moderate-to-severe active ophthalmopathy.
I-131 for Differentiated Thyroid Cancer (DTC)
After total thyroidectomy, I-131 is used for remnant ablation, adjuvant therapy, or treatment of metastatic disease, guided by ATA risk stratification:
| ATA Risk Category | Features | Typical I-131 Dose |
|---|---|---|
| Low risk | Intrathyroidal, ≤4 cm, N0, M0, no aggressive histology, no vascular invasion | 30 mCi (some guidelines: no remnant ablation needed for very low risk) |
| Intermediate risk | Minor extrathyroidal extension, vascular invasion, N1 (clinical or >5 nodes), aggressive histology | 30–150 mCi |
| High risk | Gross extrathyroidal extension, incomplete resection, distant metastases, Tg markedly elevated post-op | 100–200 mCi (dosimetry-guided for high doses) |
Preparation for RAI Therapy
TSH must be elevated (≥30 mIU/L) to stimulate iodine uptake. This is achieved by either: (1) thyroid hormone withdrawal (THW) — stop levothyroxine for 3–4 weeks (or switch to T3 for 2 weeks then stop for 2 weeks) producing clinical hypothyroidism; or (2) recombinant human TSH (rhTSH, Thyrogen) — 0.9 mg IM on two consecutive days before I-131, avoiding hypothyroidism symptoms. A low-iodine diet (<50 µg/day) for 1–2 weeks before therapy maximizes thyroid iodine avidity by depleting the body's stable iodine pool. Avoid iodinated contrast (wait 4–8 weeks after CT with contrast) and iodine-containing medications (amiodarone — may require 3–6 months washout due to long half-life).
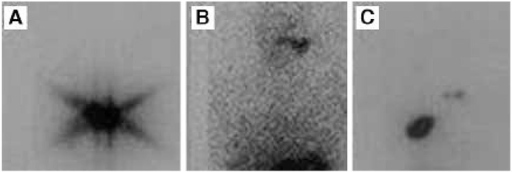
Stunning Phenomenon
Thyroid stunning refers to reduced I-131 uptake in thyroid remnant/tumor when a diagnostic dose of I-131 precedes the therapeutic dose. The diagnostic I-131 may partially damage or stun thyroid cells, reducing their ability to trap the subsequent therapeutic dose. To avoid stunning: use I-123 for diagnostic whole-body scanning (no stunning), or proceed directly to empiric I-131 therapy without a prior diagnostic scan, or use the diagnostic scan dose as the therapy dose (one-step approach). Stunning is dose-dependent: low diagnostic doses (1–3 mCi I-131) cause less stunning than higher doses (5–10 mCi), though controversy remains about the clinical significance of stunning at any dose level.
Iodine-Refractory Thyroid Cancer
Differentiated thyroid cancer that loses the ability to concentrate iodine (radioiodine-refractory DTC) is defined by: (1) absence of I-131 uptake on a post-therapy whole-body scan in all lesions; (2) loss of iodine avidity in some but not all lesions; (3) progression despite I-131 therapy within 12 months of the last treatment. These tumors typically show increased FDG avidity (the "flip-flop" phenomenon) reflecting dedifferentiation. Management shifts from RAI therapy to systemic therapy with tyrosine kinase inhibitors (lenvatinib, sorafenib — per SELECT and DECISION trials) or combination immunotherapy approaches. External beam radiation therapy may be used for focal symptomatic disease. Redifferentiation therapy with MEK inhibitors (selumetinib) or BRAF/MEK combination (dabrafenib/trametinib) in BRAF-mutant tumors can restore iodine avidity in some patients, enabling renewed RAI therapy.
Post-Therapy Whole-Body Scan
A post-therapy I-131 whole-body scan is obtained 5–7 days after therapeutic I-131 administration. The high therapeutic dose acts as its own diagnostic dose, sometimes revealing metastatic foci not seen on the pre-therapy diagnostic scan (detected in ~10–25% of cases). Common sites of uptake: thyroid bed remnant, cervical lymph nodes, lung metastases (diffuse pulmonary uptake in miliary disease), bone metastases. Physiologic uptake/false positives include: nasal secretions, salivary glands, mouth (contamination), esophageal activity, gastric mucosa, bladder (renal excretion), breast (lactating), liver (metabolism of iodinated thyroglobulin), and skin contamination.
I-131 Side Effects and Complications
Acute side effects (within days): radiation thyroiditis (neck tenderness and swelling, especially with large remnants), sialadenitis (salivary gland pain and swelling — sialogogues, hydration, and warm compresses can help; sour candy during therapy promotes salivary flow and may reduce dose), nausea (from gastric mucosal irradiation), transient bone marrow suppression (nadir at 4–6 weeks, usually mild). Chronic complications: permanent xerostomia (chronic dry mouth from salivary gland damage, especially with cumulative doses >200 mCi), dental caries (secondary to xerostomia), lacrimal gland dysfunction (dry eyes, nasolacrimal duct obstruction with tearing), pulmonary fibrosis (rare, with diffuse pulmonary metastases receiving high cumulative lung doses — lung dosimetry limits to ≤80 mCi retained at 48 hours), secondary malignancies (leukemia risk increased ~0.5–1.0% with cumulative doses >500–600 mCi; bladder cancer and salivary gland cancer also mildly elevated). Thyroid storm is a rare but life-threatening complication in patients with Graves disease with very large goiters or severe hyperthyroidism; pre-treatment with antithyroid drugs to achieve euthyroidism before RAI reduces this risk.
| Indication | Typical Dose | Special Considerations |
|---|---|---|
| Graves disease | 10–15 mCi (may calculate per gram of thyroid tissue) | Goal: ablation → hypothyroidism; consider steroid cover for ophthalmopathy |
| Toxic nodular goiter | 15–29 mCi (or calculated dose) | May need higher dose due to lower uptake per gram |
| Low-risk DTC remnant ablation | 30 mCi | Some guidelines suggest omitting RAI for very-low-risk tumors |
| Intermediate-risk DTC | 30–150 mCi | Higher doses for adverse features (vascular invasion, N1) |
| High-risk DTC / metastatic | 100–200 mCi | Dosimetry-guided for high doses; consider blood and lung dose limits |
| Retreatment for recurrence | 100–200 mCi | Cumulative lifetime dose monitoring (leukemia risk rises >600 mCi) |
15 Parathyroid Imaging & Other Endocrine
Parathyroid Scintigraphy
Tc-99m sestamibi is the primary agent for parathyroid adenoma localization. Sestamibi is taken up by both thyroid and parathyroid tissue, but it washes out of normal thyroid faster than from hyperfunctioning parathyroid tissue (which has high mitochondrial density). Dual-phase technique: early images (15–20 min) show both thyroid and parathyroid; delayed images (2–3 hr) show retained parathyroid activity with thyroid washout. Subtraction technique: Tc-99m pertechnetate (thyroid only) is subtracted from Tc-99m sestamibi (thyroid + parathyroid), revealing the parathyroid adenoma. SPECT/CT adds three-dimensional localization essential for surgical planning, distinguishing eutopic from ectopic glands (mediastinal, intrathyroidal, retroesophageal, carotid sheath). Sensitivity is ~80–90% for single adenomas but only ~30–45% for multigland hyperplasia.
4D-CT (four-dimensional CT = 3D CT + perfusion timing) has emerged as a complementary or alternative modality with sensitivity of ~80–90%, particularly useful when sestamibi is negative, for multigland disease, or in reoperative cases. It identifies parathyroid adenomas by their characteristic rapid enhancement pattern on arterial phase with washout on delayed phase. The typical 4D-CT protocol includes non-contrast, arterial (~25 sec), and delayed (~70 sec) phases, allowing differentiation of parathyroid adenoma (hypervascular with rapid wash-in) from lymph nodes (slow, progressive enhancement) and thyroid tissue. 4D-CT is superior to sestamibi for multigland disease and for detecting ectopic glands in previously operated necks, and some centers now use it as the primary preoperative localization study.
Parathyroid Imaging in Secondary and Tertiary Hyperparathyroidism
In secondary hyperparathyroidism (renal failure) and tertiary hyperparathyroidism, all four glands are hyperplastic rather than a single adenoma. Sestamibi sensitivity drops to ~30–45% for multigland disease because the technique relies on differential washout between thyroid and parathyroid, which is less pronounced when all glands are symmetrically enlarged. Subtraction scintigraphy may improve detection, and SPECT/CT localization assists the surgeon. In tertiary hyperparathyroidism (persistent HPT after renal transplant), surgical planning may require the combination of sestamibi SPECT/CT and 4D-CT for optimal preoperative mapping. Intraoperative PTH monitoring (Miami criterion: ≥50% drop from baseline at 10 minutes post-excision) confirms successful surgery regardless of imaging modality used.
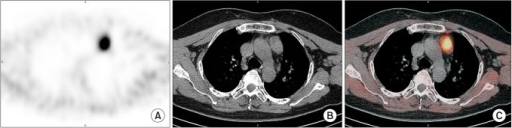
Adrenal Imaging
I-123 MIBG (metaiodobenzylguanidine) is a norepinephrine analog taken up by the norepinephrine transporter in sympathomedullary tissue. Primary indication: localization of pheochromocytoma and paraganglioma (sensitivity ~85–90%), especially for extra-adrenal, metastatic, or recurrent disease. Patient preparation: discontinue drugs that interfere with MIBG uptake (tricyclic antidepressants, labetalol, calcium channel blockers, sympathomimetics) 24–72 hours before; administer SSKI or potassium iodide to block thyroid uptake. I-131 MIBG is used for therapy of unresectable/metastatic pheochromocytoma and neuroblastoma. I-131 NP-59 (6-beta-iodomethyl-19-norcholesterol) is a cholesterol analog used for adrenal cortical imaging (Conn syndrome, subclinical Cushing) but is rarely used today due to limited availability.
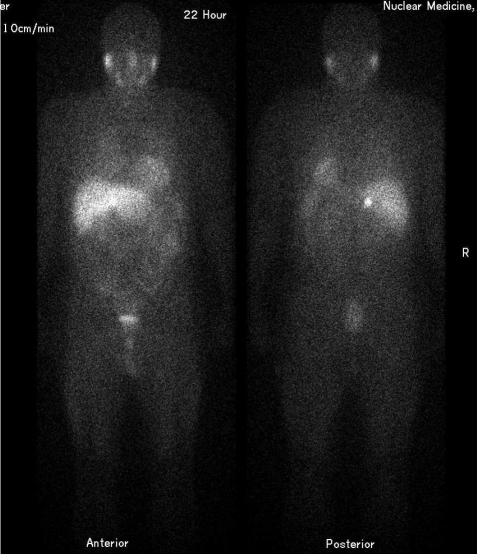
Ga-68 DOTATATE for Endocrine Tumors
Ga-68 DOTATATE PET/CT has become the imaging standard for neuroendocrine tumors (NETs), largely replacing In-111 octreotide SPECT. It is also used for pheochromocytoma/paraganglioma (especially SDH-mutated tumors, which strongly express SSTR2) and medullary thyroid cancer (variable sensitivity). Ga-68 DOTATATE PET has higher sensitivity than I-123 MIBG for pheochromocytoma (>95% vs ~85%), but MIBG retains theranostic utility (I-131 MIBG therapy).
Meningioma and Non-NET SSTR-Expressing Tumors
Ga-68 DOTATATE PET/CT also has utility beyond classical NETs. Meningiomas frequently express SSTR2 and demonstrate intense DOTATATE uptake, which can be useful for distinguishing meningioma from other dural-based lesions, delineating tumor extent for radiation planning, and detecting residual or recurrent disease post-surgery. Other SSTR-expressing pathologies include paragangliomas, pheochromocytomas, medullary thyroid cancer, small cell lung cancer (variable), and Merkel cell carcinoma.
Suspected pheochromocytoma/paraganglioma: First-line biochemical confirmation (plasma metanephrines), then CT/MRI for localization. Functional imaging with Ga-68 DOTATATE PET/CT (highest sensitivity) or I-123 MIBG (if PRRT/MIBG therapy is being considered) for: extra-adrenal disease, metastatic workup, and SDH-mutation carriers. Suspected NET: Ga-68 DOTATATE PET/CT for staging, restaging, and PRRT selection. FDG PET/CT complements DOTATATE — high-grade NETs (G3, Ki-67 >20%) lose SSTR expression and become FDG-avid (the "flip-flop phenomenon," analogous to differentiated vs dedifferentiated thyroid cancer). Insulinoma: Ga-68 exendin-4 PET/CT targets GLP-1 receptors (investigational) because insulinomas often lack SSTR2.
16 Bone Scintigraphy
Tc-99m MDP Mechanism
Tc-99m methylene diphosphonate (MDP) localizes to bone by chemisorption onto the surface of hydroxyapatite crystals in areas of active osteoblastic activity and increased regional blood flow. The degree of uptake is proportional to bone turnover and perfusion. Imaging is typically performed 2–4 hours after IV injection (20–25 mCi / 740–925 MBq) to allow soft-tissue clearance. Approximately 50% of the injected dose localizes to bone; the rest is excreted renally. Patients should be well hydrated and void frequently to reduce radiation dose to the bladder.
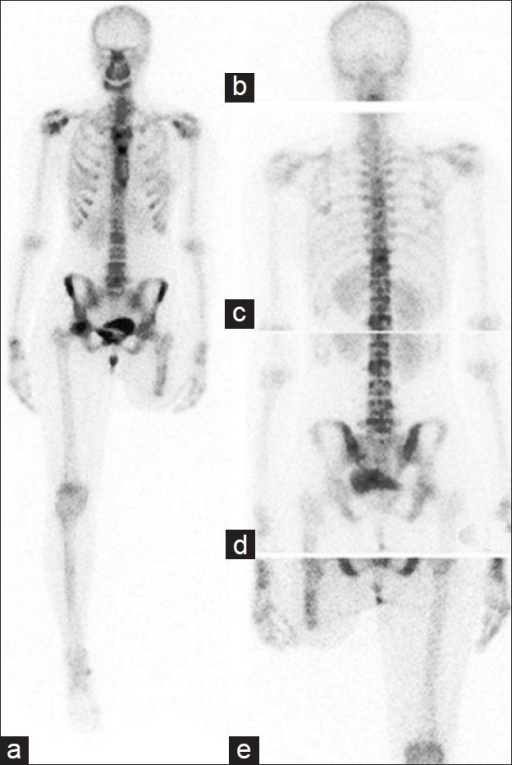
Three-Phase Bone Scan
The three-phase bone scan adds dynamic and blood pool phases to the standard delayed images:
| Phase | Timing | Reflects | Clinical Significance |
|---|---|---|---|
| Phase 1 — Flow (angiographic) | First 60 seconds, 2–5 sec/frame | Arterial perfusion | Increased flow = hyperemia (infection, acute fracture, tumor) |
| Phase 2 — Blood pool (soft tissue) | Immediately after flow, static at 5 min | Blood pool, capillary permeability | Increased = soft tissue inflammation/cellulitis |
| Phase 3 — Delayed (bone) | 2–4 hours post-injection | Osteoblastic activity, bone turnover | Increased = bone pathology (fracture, infection, tumor, Paget) |
In osteomyelitis: all three phases are positive (increased flow, blood pool, and delayed uptake) in a focal distribution. In cellulitis: phases 1 and 2 are positive but phase 3 is normal or only mildly diffusely increased (no focal bone uptake). A fourth phase (24-hour delayed) can be added to improve specificity in distinguishing osteomyelitis from soft-tissue infection.
Superscan
A superscan demonstrates diffusely increased skeletal uptake with absent or markedly diminished renal and soft-tissue activity. It occurs in: (1) widespread osteoblastic metastases (prostate cancer, breast cancer) — the "metastatic superscan"; (2) metabolic bone disease (renal osteodystrophy, hyperparathyroidism) — the "metabolic superscan," which shows uniformly increased uptake without focal lesions, often with prominent calvarium, mandible, sternum, and increased periarticular uptake ("beading"). The superscan can be mistaken for a normal scan if the reader does not notice absent kidney visualization.
| Feature | Metastatic Superscan | Metabolic Superscan |
|---|---|---|
| Distribution | Heterogeneous, irregular; may have focal "hot spots" within diffusely increased uptake | Homogeneous, uniform; smooth, symmetric uptake throughout skeleton |
| Axial vs appendicular | Often predominantly axial skeleton (spine, pelvis, ribs, proximal long bones) | Both axial and appendicular skeleton equally involved |
| Calvarium | Variable; may have focal lesions | Prominently and uniformly increased ("hot skull") |
| Mandible | Usually not prominent | Characteristically prominent (metabolic signal) |
| Periarticular activity | Not typically prominent | "Beading" pattern at joints (increased periarticular uptake) |
| Kidneys | Absent or faint | Absent or faint |
| Clinical context | Known cancer (prostate, breast, lung) | Renal failure, hyperparathyroidism, Paget disease (polyostotic) |
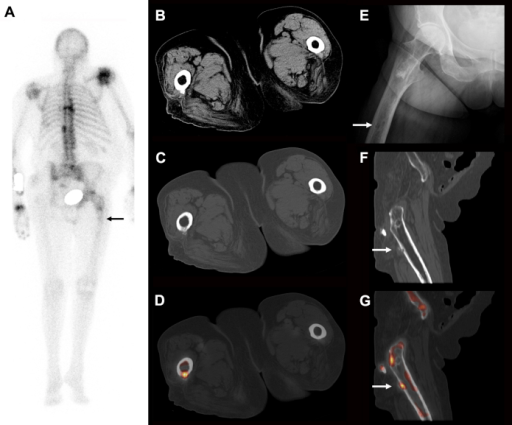
Flare Phenomenon
The flare phenomenon is a transient increase in radiotracer uptake at known metastatic sites on a bone scan obtained 2–12 weeks after initiating effective systemic therapy (chemotherapy, hormonal therapy). It represents an osteoblastic healing response and must not be misinterpreted as disease progression. Subsequent scans will show improvement if the flare is genuine. New lesions not present on baseline are more likely true progression. The flare response is also seen with F-18 NaF PET and can be seen with FDG PET (though less commonly).
Other Bone Scan Findings
Photopenic (cold) lesions: Areas of decreased uptake indicate aggressive, rapidly destructive processes that outpace osteoblastic repair — purely lytic metastases (renal cell, myeloma, thyroid), radiation therapy fields, avascular necrosis (early), metal prostheses, and artifacts. Paget disease: Markedly increased uptake with cortical thickening and bone expansion; monostotic or polyostotic; characteristic locations include pelvis, femur, tibia, skull, and spine. Stress fractures: Focal linear uptake, positive on all three phases; common in metatarsals (march fracture), tibia, femoral neck, and sacrum. AVN: Early = cold (ischemia); later = hot (revascularization/repair); classically described in the femoral head.
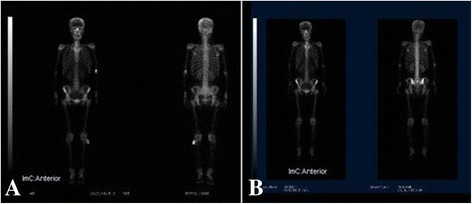
Bone Scan in Specific Clinical Scenarios
Prostate cancer: Bone scan remains the standard initial staging study for high-risk prostate cancer (PSA >20, Gleason ≥8, T3+). The majority of prostate metastases are osteoblastic and MDP-avid. However, PSMA PET is increasingly used and detects bone metastases earlier. Breast cancer: Bone scan is used for staging in locally advanced or symptomatic disease; mixed lytic-blastic metastases are common. Lung cancer: Bone scan or PET/CT; PET is generally preferred due to its ability to detect both lytic and blastic metastases plus soft-tissue staging in one study. Reflex sympathetic dystrophy (CRPS): Three-phase bone scan shows periarticular uptake on blood pool and delayed phases; however, sensitivity decreases in chronic CRPS. Shin splints vs stress fracture: Shin splints show longitudinal, linear uptake along the tibial cortex on delayed images (typically blood pool negative); stress fractures show focal, fusiform uptake positive on all three phases.
Bone Scan Artifacts and Pitfalls
Urinary contamination is the most common artifact on bone scans and can mimic pelvic osseous pathology; correlation with patient history and repeat imaging after voiding and cleaning resolves the issue. Injection site infiltration (paravenous injection) causes intense activity at the injection site and may produce ipsilateral axillary lymph node visualization. Renal activity is normal but asymmetric renal uptake (absent kidney) may indicate renal pathology. Free pertechnetate (from inadequate labeling) concentrates in thyroid, salivary glands, and stomach — these should not normally be visible on an MDP bone scan. The presence of thyroid or gastric activity signals poor radiochemical purity.
17 Infection & Inflammation Imaging
Labeled WBC Scans
In-111 oxine–labeled WBCs and Tc-99m HMPAO–labeled WBCs are used for localization of infection. Patient's own WBCs are drawn, labeled ex vivo with the radiotracer, and reinjected. Normal biodistribution includes liver, spleen (most intense), and bone marrow. In-111 WBCs: imaged at 18–24 hours post-injection; dual-energy windows (171 + 245 keV); longer half-life (67.3 hr) allows delayed imaging; no normal bowel activity (unlike Tc-99m HMPAO). Tc-99m HMPAO WBCs: imaged at 1–4 hours; better image quality due to higher count rates; normal GI and GU excretion limits interpretation after 4 hours. In-111 WBCs are preferred for chronic/abdominal infections; Tc-99m HMPAO WBCs are preferred when faster results are needed or for combined bone marrow scan protocols.
Osteomyelitis Workup Protocol
For suspected osteomyelitis in normal bone (without prior surgery/hardware/neuropathy): the three-phase bone scan is the initial study (sensitivity ~95%). If all three phases are focally positive, osteomyelitis is likely. For the complicated skeleton (diabetic foot, post-surgical, prosthetic joint): a combined approach is needed. Diabetic foot protocol: three-phase bone scan (sensitive but not specific in the setting of Charcot neuroarthropathy) followed by In-111 or Tc-99m HMPAO labeled WBC scan. Osteomyelitis shows concordant bone scan and WBC activity. Charcot foot shows positive bone scan but negative or discordant WBC scan.
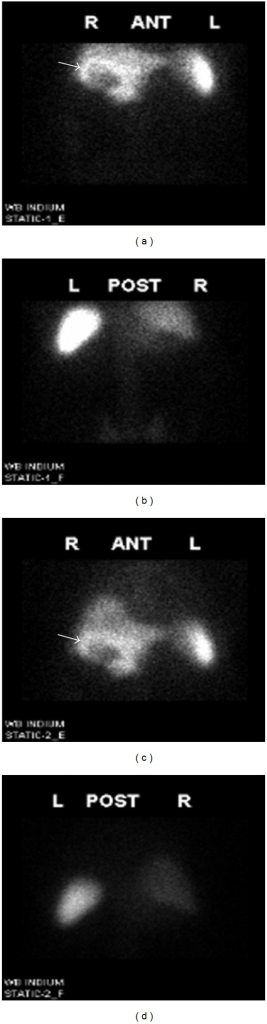
WBC Scan Interpretation Pearls
Key interpretation points for labeled WBC scans: (1) Temporal pattern matters: In-111 WBC images obtained at 3–4 hours (early) and 18–24 hours (delayed) — infection shows increasing intensity over time, while normal marrow distribution remains stable. If a focus is more intense at 24 hours than at 4 hours, it is more likely infection. (2) Spatial pattern matters: Focal WBC uptake that is more intense than normal marrow (iliac crests as reference) suggests infection. (3) Spine limitations: WBC scans are unreliable for spinal infection because vertebral marrow uptake is variable and infections may appear as photopenic defects rather than increased uptake (due to marrow replacement by infection). FDG PET or MRI is preferred for spondylodiscitis. (4) Chronic infection: WBC scans are less sensitive for chronic, low-grade infections (labeled WBCs accumulate best in acute pyogenic infections with high neutrophil recruitment). FDG PET is more sensitive for chronic osteomyelitis and low-virulence infections.
Prosthetic Joint Infection
The combination of labeled WBC scan + Tc-99m sulfur colloid bone marrow scan is the most accurate nuclear medicine approach for prosthetic joint infection (accuracy >90%). The principle: labeled WBCs accumulate in marrow as well as at sites of infection. If WBC uptake matches the sulfur colloid marrow distribution, the WBCs are in displaced marrow (not infection). If WBC uptake occurs in a location without corresponding sulfur colloid uptake (spatial incongruence), infection is diagnosed. This approach corrects for the altered marrow distribution that occurs around prostheses due to marrow remodeling.
Step 1: Three-phase bone scan — if normal, infection is effectively excluded (high sensitivity, low specificity). Step 2: If bone scan is abnormal (nearly always is around prostheses due to remodeling), proceed to labeled WBC scan (In-111 oxine or Tc-99m HMPAO). Step 3: If WBC scan shows periprosthetic uptake, perform sulfur colloid bone marrow scan. Interpretation: WBC and marrow scan concordant (matching pattern) = marrow activity, no infection. WBC uptake in area WITHOUT marrow uptake (incongruent) = infection. This dual-scan technique achieves ~90–95% accuracy for periprosthetic joint infection. Alternative: FDG PET/CT has high sensitivity (~95%) but variable specificity (~75–85%) for prosthetic joint infection because low-grade periprosthetic FDG uptake is common in asymptomatic prostheses; the pattern of uptake matters more than the presence — focal, intense uptake at the bone-prosthesis interface suggests loosening or infection, while mild diffuse uptake may be nonspecific.
FDG PET for Infection
FDG PET/CT has high sensitivity for infection due to the high glycolytic activity of activated neutrophils and macrophages. It is particularly useful for: chronic osteomyelitis (sensitivity >95%), spinal infections/discitis (especially when MRI is equivocal or contraindicated), vascular graft infections, prosthetic valve endocarditis, and FUO workup. Limitations: nonspecific (inflammation and infection are indistinguishable), post-surgical inflammation causes false positives, and it does not differentiate low-grade from high-grade infection.
Ga-67 Citrate
Ga-67 citrate was historically used for infection and tumor imaging but has been largely replaced by FDG PET/CT. It binds to transferrin and lactoferrin in the blood, localizing to sites of infection (via lactoferrin in neutrophils), inflammation, and certain tumors (lymphoma, hepatocellular carcinoma). Its current niche role includes evaluation of chronic opportunistic infections in immunocompromised patients and monitoring treatment response in sarcoidosis (panda sign = bilateral parotid/lacrimal uptake; lambda sign = bilateral hilar/right paratracheal uptake).
Uncomplicated osteomyelitis (normal bone): Three-phase bone scan → if positive on all 3 phases, high sensitivity (~95%). Complicated osteomyelitis (diabetic foot, post-surgical, hardware): Three-phase bone scan (sensitive but nonspecific) + labeled WBC scan (adds specificity). If WBC localizes to bone, osteomyelitis is likely. If discordant (bone scan hot, WBC cold), consider Charcot neuroarthropathy. Prosthetic joint infection: WBC scan + sulfur colloid marrow scan (most accurate combination, ~90%). Spatial incongruence (WBC uptake without matching marrow uptake) = infection. Spondylodiscitis: FDG PET/CT preferred (WBC scans are unreliable in the spine because of normal marrow activity). FUO: FDG PET/CT as first-line nuclear study (detects occult infection, vasculitis, and malignancy in 30–50% of cases). Prosthetic valve endocarditis: FDG PET/CT (included in modified Duke criteria).
18 Pediatric Nuclear Medicine
Renal Imaging
Tc-99m DMSA cortical scintigraphy is the gold standard for detecting acute pyelonephritis (photopenic cortical defects) and renal scarring in children. Split renal function calculation (normal: each kidney within 45–55% of total) is essential for surgical decision-making. Tc-99m MAG3 diuretic renography evaluates suspected urinary obstruction (e.g., ureteropelvic junction obstruction). After radiotracer injection, furosemide (1 mg/kg IV) is administered; a T½ drainage time <10 minutes is normal, 10–20 minutes is indeterminate, and >20 minutes suggests obstruction. The direct radionuclide cystogram (DRC) using Tc-99m pertechnetate instilled into the bladder via catheter is an alternative to fluoroscopic VCUG for detecting vesicoureteral reflux with significantly lower radiation dose (100× less gonadal dose), though it provides less anatomic detail.
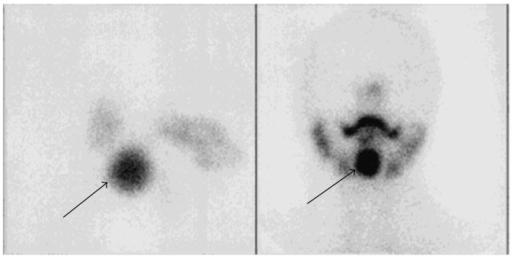
Thyroid Imaging in Congenital Hypothyroidism
Thyroid scintigraphy (I-123 or Tc-99m pertechnetate) in neonates with congenital hypothyroidism identifies: thyroid agenesis (no uptake anywhere), ectopic thyroid (lingual or sublingual uptake — the most common cause of congenital hypothyroidism with visible tissue), dyshormonogenesis (enlarged gland with increased uptake due to organification defect — confirmed with perchlorate discharge test), and thyroid hypoplasia. This information helps guide counseling: ectopic and absent thyroid require lifelong replacement, while some dyshormonogenetic cases may eventually produce adequate hormone.
MIBG for Neuroblastoma
I-123 MIBG scintigraphy is essential for staging and response assessment of neuroblastoma. Neuroblastoma cells express the norepinephrine transporter and avidly take up MIBG (~90% of tumors are MIBG-avid). The Curie scoring system semi-quantitatively grades MIBG uptake across 10 body regions (score 0–3 per region) for standardized response assessment. I-131 MIBG therapy is FDA-approved for relapsed/refractory neuroblastoma (Azedra). I-123 MIBG has also been supplemented by FDG PET/CT, which may be more sensitive in MIBG-non-avid tumors and for detecting disease in the setting of therapy response assessment.
Bone Scan in Pediatrics
In suspected child abuse, a skeletal survey (radiographs) is the primary imaging modality, but bone scintigraphy is more sensitive for rib fractures, subtle long bone fractures, and spinal injuries. The two modalities are complementary. For Langerhans cell histiocytosis (LCH), bone scan has moderate sensitivity (~70%) because LCH can be predominantly lytic with minimal osteoblastic response; FDG PET/CT is increasingly used and may be more sensitive. For osteoid osteoma, three-phase bone scan shows intense focal uptake (the "double-density" sign on SPECT).
Pediatric patients receive weight-based (or BSA-based) radiotracer doses per EANM/SNMMI guidelines (the "North American Consensus Guidelines" or European "Dosage Card"). Minimum acceptable doses are maintained to ensure diagnostic image quality. Additional strategies: shorter scan times using newer camera technology, reduced CT dose for attenuation correction (low-dose CT, iterative reconstruction), and careful justification (ALARA is especially important in children due to longer remaining lifespan for stochastic risk expression).
Hepatoblastoma and Other Pediatric Tumors
FDG PET/CT is used in the staging and monitoring of various pediatric malignancies including lymphoma, Ewing sarcoma, osteosarcoma, rhabdomyosarcoma, and Wilms tumor. For Langerhans cell histiocytosis, FDG PET is more sensitive than bone scan for detecting skeletal and extraskeletal disease and is increasingly used as the primary staging modality. For neuroblastoma, a dual-tracer approach (I-123 MIBG for MIBG-avid disease + FDG PET for MIBG-non-avid or undifferentiated disease) provides the most comprehensive assessment. For Ewing sarcoma and osteosarcoma, FDG PET provides superior restaging after neoadjuvant chemotherapy compared to conventional imaging; a decrease in SUVmax of ≥50% or post-treatment SUVmax <2.5 correlates with favorable histologic response (≥90% necrosis). For lymphoma in children, the same Deauville criteria used in adults apply, with the caveat that thymic rebound (diffuse, symmetric anterior mediastinal FDG uptake) is extremely common in the post-chemotherapy period and must not be mistaken for residual disease.
Pediatric Dose Calculations
Pediatric radiopharmaceutical doses are calculated using the EANM Dosage Card or North American Consensus Guidelines, which provide weight-based multipliers for each agent. The general principle: administered activity scales with body weight (or BSA), with specified minimum doses to ensure diagnostic image quality. For example, the minimum FDG dose for a pediatric whole-body PET/CT is typically 37 MBq (1 mCi), with a weight-based formula of approximately 5.2 MBq/kg (0.14 mCi/kg). For Tc-99m DMSA renal cortical imaging, the minimum dose is approximately 18.5 MBq (0.5 mCi). Newer CZT-based cameras and silicon photomultiplier PET/CT scanners with improved sensitivity allow further dose reduction (up to 50%) while maintaining diagnostic quality, which is particularly important in pediatric patients who have the greatest lifetime radiation risk per unit dose.
Biliary Atresia in Neonates
The Tc-99m mebrofenin (HIDA) scan helps differentiate biliary atresia from neonatal hepatitis in jaundiced neonates. In biliary atresia, the liver shows normal or good tracer uptake but no excretion into the bowel even on 24-hour delayed images (complete biliary obstruction). In neonatal hepatitis, hepatocyte function is impaired (delayed/poor uptake), but some tracer eventually reaches the bowel. Pre-treatment with phenobarbital (5 mg/kg/day for 5 days) enhances hepatocyte function and biliary excretion, improving the accuracy of the study by reducing false-positive results (hepatitis mimicking atresia due to poor excretion). Sensitivity for biliary atresia is ~97% with phenobarbital pretreatment.
19 Renal Scintigraphy
Tc-99m MAG3 (Mercaptoacetyltriglycine)
Tc-99m MAG3 is cleared almost entirely by tubular secretion (effective renal plasma flow, ERPF) with minimal glomerular filtration, providing excellent images even in patients with impaired renal function. Applications: (1) diuretic renography for obstruction (see pediatric section); (2) split renal function determination (relative percentage of each kidney); (3) renal transplant evaluation — ATN (preserved perfusion with delayed cortical transit and excretion), acute rejection (decreased perfusion and function), obstruction, urine leak (extravasation of tracer). Time-activity curves are generated over each kidney: normal = rapid uptake, peak at 3–5 min, prompt clearance. Obstructed pattern: rising curve without clearance (unresponsive to furosemide).
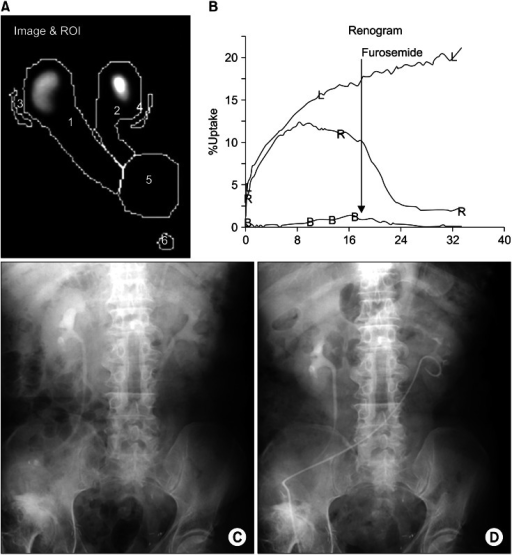
Tc-99m DMSA (Dimercaptosuccinic Acid)
Tc-99m DMSA binds to proximal tubular cells and remains in the renal cortex for hours, providing high-resolution cortical imaging. It is the gold standard for: detection of cortical scarring (chronic pyelonephritis), acute pyelonephritis (photopenic defects), ectopic or horseshoe kidney, split renal function (most accurate method), and assessment of functioning renal tissue in multicystic dysplastic kidney. Normal split function is 45–55% for each kidney.
Captopril Renogram
The captopril (ACE inhibitor) renogram detects renovascular hypertension caused by renal artery stenosis. In a kidney with hemodynamically significant renal artery stenosis, GFR is maintained by angiotensin II–mediated efferent arteriolar constriction. Administration of captopril (25–50 mg PO 1 hour before the scan) removes this compensatory mechanism, causing a drop in GFR and prolonged cortical transit time in the affected kidney. A positive study shows: delayed time-to-peak, decreased uptake, and prolonged cortical retention on the post-captopril MAG3 renogram compared to baseline. Sensitivity ~90% and specificity ~95% for functionally significant renal artery stenosis.
| Finding | Description | Significance |
|---|---|---|
| Normal baseline + abnormal captopril | Delayed TTP, prolonged cortical retention, decreased uptake on affected side after captopril | Positive for hemodynamically significant RAS; predicts BP improvement after revascularization |
| Abnormal baseline + more abnormal captopril | Baseline already shows functional asymmetry; worsens after captopril | Significant RAS with already-compromised function |
| Abnormal baseline + no change with captopril | Fixed functional abnormality | Chronic damage (nephrosclerosis); unlikely to improve with revascularization |
| Normal baseline + normal captopril | No change | No hemodynamically significant RAS; negative study |
Preparation: Hold ACE inhibitors and ARBs for 3–5 days before the study. Ensure adequate hydration. Monitor blood pressure before and after captopril (risk of hypotension, especially in bilateral RAS). The study is most useful in patients with moderate pretest probability of renovascular hypertension.
GFR Measurement
Tc-99m DTPA (diethylenetriaminepentaacetic acid) is cleared almost exclusively by glomerular filtration (no tubular secretion or reabsorption), making it suitable for GFR measurement. The Gates method calculates GFR from the renal uptake of DTPA in the 1–3 minute post-injection period, corrected for body surface area and camera-to-kidney depth. This nuclear medicine GFR is an alternative to plasma clearance methods and creatinine-based estimates, particularly useful when the latter are unreliable (extremes of body habitus, rapidly changing renal function).
Renal Transplant Evaluation
Tc-99m MAG3 renography is the primary nuclear medicine study for transplant kidney assessment. The transplanted kidney, located in the iliac fossa, is imaged with posterior or anterior views (depending on surgical placement). Key findings: Acute tubular necrosis (ATN) — preserved perfusion (good flow phase) with delayed cortical transit and excretion, reflecting tubular dysfunction; usually improves over days to weeks. Acute rejection — decreased perfusion AND decreased function; more ominous than ATN and requires urgent biopsy and immunosuppression adjustment. Cyclosporine/tacrolimus toxicity — may mimic rejection scintigraphically; clinical correlation and biopsy required. Urine leak — progressive accumulation of tracer outside the collecting system into the peritoneal or retroperitoneal space. Renal artery stenosis — captopril renogram can be performed on transplant kidneys using the same principles as native kidneys.
MAG3: Best for renal function and obstruction assessment; works even in impaired renal function (tubular secretion). DMSA: Best for cortical imaging, scarring, and ectopia; gold standard for split function. DTPA: Best for GFR measurement; also used for diuretic renography but inferior to MAG3 in renal insufficiency. Captopril + MAG3: Best for renovascular hypertension evaluation.
20 Pulmonary Scintigraphy
V/Q Scan Protocol
The ventilation/perfusion (V/Q) scan evaluates for pulmonary embolism by comparing the distribution of ventilation and perfusion in the lungs. Perfusion: Tc-99m macroaggregated albumin (MAA), 2–4 mCi IV, lodges in ~0.1% of pulmonary capillaries proportional to blood flow; images acquired in 8 views (anterior, posterior, RPO, LPO, RAO, LAO, right lateral, left lateral). The number of particles injected is typically 200,000–400,000 (reduced to 100,000–150,000 in pregnancy, pulmonary hypertension, and right-to-left shunt). Ventilation: Xe-133 gas (single-breath, equilibrium, and washout phases — disadvantage: performed before perfusion due to low 81 keV energy), Tc-99m DTPA aerosol (convenient, done after perfusion with Tc-99m MAA in a standard protocol, central deposition in airways in obstructive disease), or Kr-81m gas (190 keV, allows simultaneous imaging with Tc-99m perfusion, very short T½ = 13 sec, expensive).
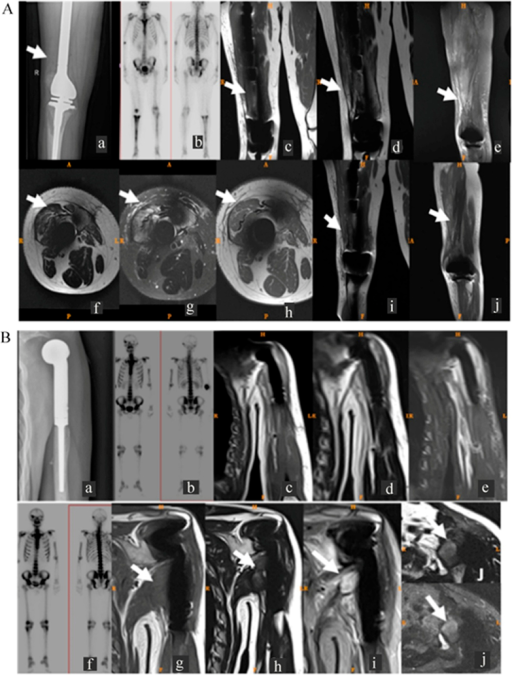
PIOPED II Criteria (Modified)
| Category | Criteria | PE Probability |
|---|---|---|
| Normal | No perfusion defects; perfusion outlines lung shape | ~0% |
| Very low probability | Non-segmental perfusion defects (e.g., cardiomegaly, elevated diaphragm, costophrenic effusion); perfusion defects smaller than corresponding CXR abnormality; 1–3 small segmental defects with normal ventilation | <5% |
| Low probability | Perfusion defects matched by ventilation abnormality or CXR abnormality (matched defects); single moderate V/Q mismatch | ~15–20% |
| Intermediate probability | Does not fit other categories; 1 moderate or 2 large mismatched defects with lower-lobe CXR abnormality; borderline or difficult-to-categorize findings | ~20–50% |
| High probability | ≥2 large (>75% segment) mismatched perfusion defects or equivalent (1 large + ≥2 moderate); defects substantially larger than CXR abnormalities | >80% |
Quantitative Lung Perfusion
Quantitative perfusion lung scan measures the relative perfusion to each lung (and individual lobes), expressed as a percentage of total pulmonary blood flow. This is essential for: (1) pre-pneumonectomy assessment — predicted postoperative FEV1 = preoperative FEV1 × (1 − percent perfusion to the resected lung). If predicted postoperative FEV1 is <40% of normal or <800 mL, the patient is at high risk for respiratory failure post-resection. (2) Pre-lung volume reduction surgery planning.
Right-to-Left Shunt Quantification
After injection of Tc-99m MAA, particles that bypass the pulmonary capillary bed through a right-to-left shunt (e.g., intracardiac shunt, pulmonary AVM) lodge in systemic capillary beds, particularly the brain and kidneys. The shunt fraction is calculated as: (systemic counts) / (systemic + pulmonary counts) × 100%. Normal shunt fraction is <5%. This quantification is important for evaluating hereditary hemorrhagic telangiectasia (Osler-Weber-Rendu) and other causes of pulmonary AVMs.
V/Q SPECT
V/Q SPECT is increasingly replacing planar V/Q imaging, offering improved sensitivity (~97%) and specificity (~91%) for PE, with significantly fewer indeterminate results compared to planar V/Q (reduced from ~30–40% to <5%). V/Q SPECT acquires three-dimensional perfusion and ventilation data, which can be fused with low-dose CT for anatomic correlation. The mismatch criterion on SPECT is conceptually the same as planar: a perfusion defect without a corresponding ventilation defect indicates PE. Quantitative V/Q SPECT can calculate ventilation-perfusion quotient maps, enabling automated detection of mismatches. V/Q SPECT is particularly advantageous for evaluating peripheral/subsegmental emboli and for follow-up studies to quantify resolution.
21 GI & Hepatobiliary Scintigraphy
Hepatobiliary (HIDA) Scan
Tc-99m mebrofenin (Choletec) or Tc-99m disofenin (Hepatolite) are IDA (iminodiacetic acid) analogs cleared by hepatocytes into bile, following the bilirubin pathway. The study evaluates biliary patency, gallbladder function, and bile leaks. Normal sequence: hepatic uptake within 5 minutes, gallbladder visualization by 30–60 minutes, bowel activity by 60 minutes. In acute cholecystitis, the cystic duct is obstructed → gallbladder fails to visualize by 60 minutes (with adequate hepatocyte function). Morphine (0.04 mg/kg IV) can be administered at 60 minutes to contract the sphincter of Oddi and increase biliary pressure, forcing tracer into the gallbladder if the cystic duct is patent (if the gallbladder still does not fill by 30 minutes after morphine, the study is positive for acute cholecystitis). The rim sign (pericholecystic hepatic uptake adjacent to the gallbladder fossa) indicates severe inflammation with ischemia or gangrene — a high-risk finding. Sensitivity and specificity for acute cholecystitis are ~95% and ~90%, respectively.
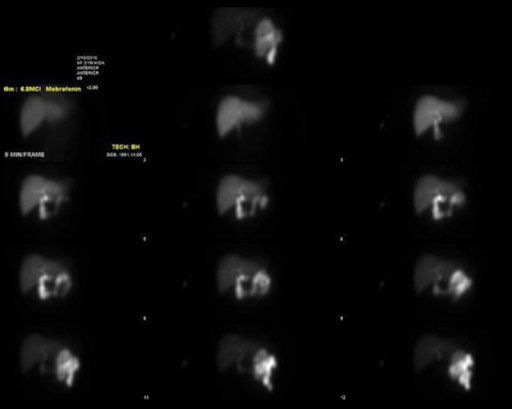
CCK-Stimulated Ejection Fraction
For suspected chronic acalculous cholecystitis or biliary dyskinesia, CCK (cholecystokinin, sincalide) is infused IV (0.02 µg/kg over 60 minutes — slow infusion is critical to avoid false-positive results from rapid infusion causing spasm) after gallbladder filling. The gallbladder ejection fraction (GBEF) is calculated from the change in gallbladder counts before and after CCK stimulation. Normal GBEF is >35% (some references use ≥38%). A low GBEF with reproduction of the patient's symptoms supports biliary dyskinesia and may predict symptom relief after cholecystectomy.
Bile Leak Detection
HIDA scan is highly sensitive for bile leak after cholecystectomy, hepatic surgery, or trauma. Any activity outside the biliary tree and bowel lumen (e.g., in the peritoneal cavity, subhepatic space, or around a drain site) confirms a bile leak. Delayed images (4–24 hours) may be needed for slow leaks.
Chronic Cholecystitis vs Acute Cholecystitis
In chronic cholecystitis, the gallbladder may visualize on the HIDA scan (patent cystic duct) but with delayed filling or a reduced ejection fraction on CCK stimulation. The key differentiator: in acute cholecystitis, the gallbladder never fills (cystic duct obstruction) even after morphine augmentation. In chronic cholecystitis, the gallbladder fills (sometimes delayed) but functions poorly. The HIDA scan with morphine augmentation has sensitivity >95% and specificity ~90% for acute cholecystitis, making it the nuclear medicine study with one of the highest diagnostic accuracies in all of abdominal imaging. False positives (non-visualization without acute cholecystitis) occur in: prolonged fasting (>24 hr — distended GB with thick bile), chronic cholecystitis with severely diseased cystic duct, severe hepatocellular disease (no excretion), and critically ill patients on TPN.
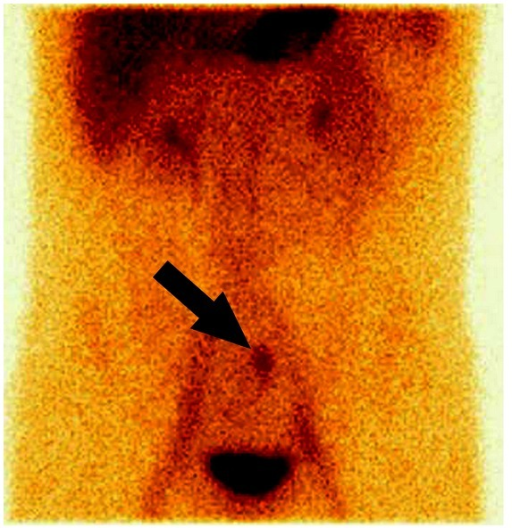
Meckel Diverticulum Scan
Tc-99m pertechnetate is taken up by ectopic gastric mucosa in a Meckel diverticulum (present in ~50% of symptomatic Meckel diverticula). The scan shows focal uptake in the right lower quadrant that appears simultaneously with or shortly after gastric uptake (because the ectopic gastric mucosa accumulates pertechnetate at the same rate as normal stomach). Sensitivity is ~85% in children but lower (~60%) in adults. Pre-treatment with cimetidine (H2 blocker) for 48 hours inhibits pertechnetate secretion by ectopic mucosa, increasing detection by retaining activity. Pentagastrin can enhance uptake, and glucagon can reduce peristaltic blurring.
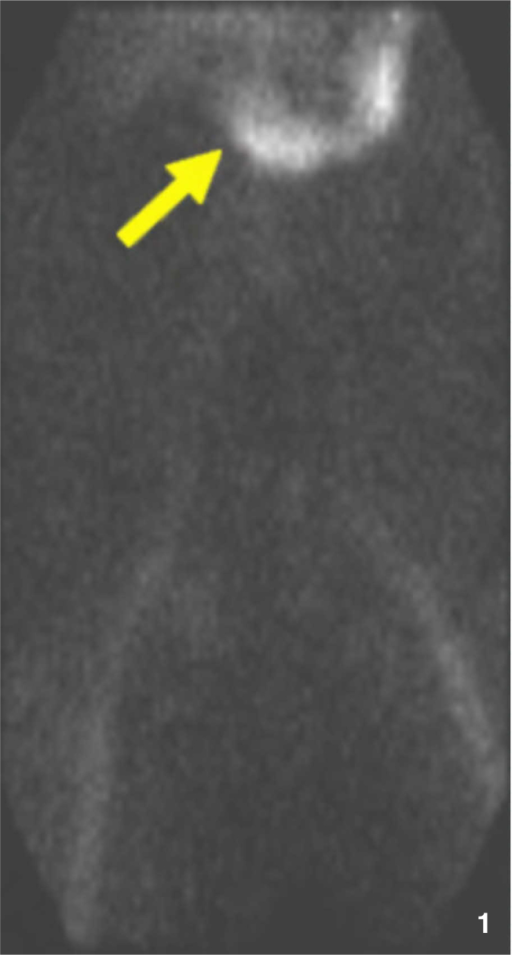
GI Bleeding Scan
Tc-99m labeled RBC scan detects active GI bleeding at rates as low as 0.1–0.4 mL/min (vs 0.5–1.0 mL/min for angiography and 0.3–0.5 mL/min for CTA). RBCs are labeled in vivo or in vitro with Tc-99m, and serial images are acquired over 90 minutes (with delayed images up to 24 hours). A positive study shows focal tracer accumulation that moves in an antegrade or retrograde pattern within the bowel lumen over time. The timing and location of the first blush help guide angiographic intervention or surgery. CT angiography has increasingly replaced nuclear RBC scans in the acute setting due to faster acquisition and precise anatomic localization, but the nuclear scan's ability to detect intermittent bleeding over extended periods (24-hour imaging window) remains a unique advantage for patients with slow, intermittent GI hemorrhage.
Liver-Spleen Scintigraphy
Tc-99m sulfur colloid liver-spleen scan evaluates reticuloendothelial system (RES) function. Normal distribution: liver (85%), spleen (10%), bone marrow (5%). In cirrhosis, Kupffer cell dysfunction leads to decreased hepatic uptake with a characteristic shift of colloid to the spleen and bone marrow ("colloid shift"). In Budd-Chiari syndrome, the caudate lobe (which drains directly into the IVC) is spared and shows relatively increased uptake compared to the rest of the liver. Focal lesions: most hepatic masses (metastases, HCC, cysts, abscesses) appear as photopenic defects; the notable exception is focal nodular hyperplasia (FNH), which contains functional Kupffer cells and may show normal or even increased sulfur colloid uptake (— a finding virtually pathognomonic for FNH). While this study has been largely replaced by cross-sectional imaging, the sulfur colloid scan retains niche utility for confirming FNH, evaluating diffuse hepatic disease, and as part of the bone marrow/WBC scan protocol for prosthetic joint infection.
Gastric Emptying Study
The 4-hour solid-phase gastric emptying study is the standard for diagnosing gastroparesis. The patient ingests a standardized meal of Tc-99m sulfur colloid-labeled egg whites with toast, jam, and water. Anterior and posterior images are obtained at 0, 1, 2, and 4 hours, and the geometric mean of counts is calculated to correct for depth attenuation. Normal gastric retention values:
| Time Point | Normal Gastric Retention | Abnormal (Delayed) If |
|---|---|---|
| 1 hour | ≤90% | >90% |
| 2 hours | ≤60% | >60% |
| 4 hours | ≤10% | >10% |
The 4-hour value is the most important and most reproducible. Patients should be off prokinetics (metoclopramide, erythromycin) for 48 hours and off opioids/anticholinergics for as long as clinically feasible. Diabetic patients should have blood glucose <275 mg/dL (hyperglycemia delays gastric emptying).
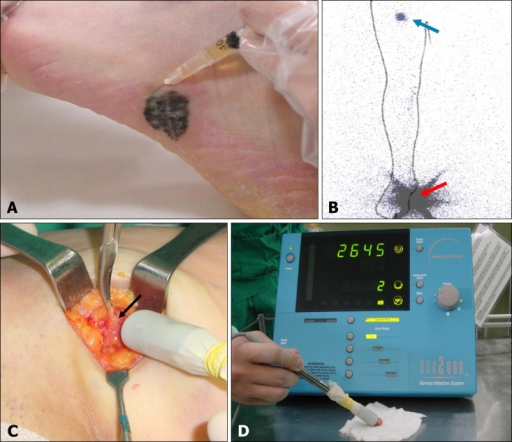
Sentinel Lymph Node Mapping
Tc-99m sulfur colloid (filtered) or Tc-99m tilmanocept (Lymphoseek) is injected peritumorally (e.g., breast cancer, melanoma) for sentinel lymph node (SLN) identification. The radiotracer drains through lymphatic channels to the first echelon node(s), which are identified intraoperatively using a handheld gamma probe. The SLN is excised and examined histopathologically; if negative for metastatic disease, formal lymph node dissection is avoided. Lymphoscintigraphy (preoperative imaging) maps the drainage pattern, which can be unpredictable, especially for truncal melanomas and head/neck lesions where drainage may be bilateral or to unexpected basins (internal mammary, epitrochlear, popliteal). Tc-99m tilmanocept specifically binds CD206 receptors on dendritic cells and macrophages in lymph nodes, providing more specific targeting and less shine-through than filtered sulfur colloid.
Salivary Gland Scintigraphy
Tc-99m pertechnetate salivary gland scan evaluates glandular function by measuring uptake and excretion by the parotid and submandibular glands. After IV injection, sequential images are obtained for 30 minutes. A sialagogue (lemon juice) is administered at 15 minutes to stimulate secretion. Normal: symmetric uptake followed by rapid washout after stimulation. In Sjogren syndrome, there is globally decreased uptake and poor or absent response to sialagogue. In Warthin tumor, there is focal increased uptake (Warthin tumor and oncocytoma are the only salivary tumors that are "hot" on pertechnetate and sestamibi scans). Obstructing calculi show preserved uptake but delayed or absent excretion.
Lymphoscintigraphy for Lymphedema
Tc-99m sulfur colloid (filtered) or Tc-99m tilmanocept is injected intradermally into the web spaces of the affected limb. Sequential images track lymphatic flow from the injection site through lymphatic channels to regional lymph nodes. In lymphedema, findings include: delayed transport (prolonged transit time), dermal backflow (retrograde flow into dermal lymphatic plexus — the hallmark finding), asymmetric or absent visualization of lymph nodes, and collateral lymphatic channels. Quantitative analysis (transport index) grades severity. Lymphoscintigraphy is useful for distinguishing primary from secondary lymphedema, assessing lymphedema severity, and planning surgical interventions (lymphovenous anastomosis or vascularized lymph node transfer). The study can also differentiate true lymphedema from venous edema (normal lymphatic flow in pure venous insufficiency) and lipedema (normal lymphatic flow).
Hepatic Hemangioma Imaging
Tc-99m labeled RBC SPECT was historically the gold standard for confirming hepatic hemangiomas. The characteristic finding is a peripheral enhancement pattern on dynamic images with progressive fill-in on delayed blood pool images, matching the pattern seen on multiphasic CT/MRI. Sensitivity is highest for lesions >2 cm; smaller lesions may not be resolved. This study has been largely supplanted by contrast-enhanced MRI (which has superior spatial resolution and does not involve radiation), but it retains utility when MRI is contraindicated or when ultrasound and CT findings are equivocal and MRI is unavailable.
Esophageal Transit Study & GI Motility
A Tc-99m sulfur colloid esophageal transit study evaluates esophageal motility by having the patient swallow a bolus of Tc-99m labeled liquid or semisolid while supine. Sequential images track the bolus through the esophagus. Normal transit time from oropharynx to stomach is <15 seconds. Delayed transit, stasis at specific levels, or retrograde activity (reflux) can be quantified. This study is useful for evaluating achalasia, diffuse esophageal spasm, scleroderma-related dysmotility, and post-fundoplication dysphagia. A related study, the gastroesophageal reflux scan (milk scan) in pediatrics, uses Tc-99m sulfur colloid in formula to detect GE reflux and assess aspiration risk (delayed images of the lungs may show aspiration of refluxed material). This is a more physiologic assessment than the barium fluoroscopy GI series and involves less radiation.
Gallium-68 Emerging Generator Applications
The Ge-68/Ga-68 generator provides Ga-68 (T½ 68 min) from the long-lived parent Ge-68 (T½ 271 days), enabling PET tracer production without a cyclotron. The generator has a useful life of approximately 9–12 months and can be eluted up to 2–3 times daily. Ga-68 is chelated to DOTA-peptides (DOTATATE, DOTATOC) for NET imaging and to PSMA ligands for prostate cancer imaging. The availability of the Ga-68 generator has democratized PET imaging of NETs and prostate cancer, enabling even smaller nuclear medicine departments without cyclotron access to offer these studies. The FDA approval of Ga-68 DOTATATE (NETSPOT) and Ga-68 PSMA-11 (LOCAMETZ/ILLUCCIX) from generator-produced Ga-68 has made these agents widely available across community practice settings.
22 Brain & CSF Imaging
Brain Death Study
A nuclear medicine brain perfusion study confirms brain death when clinical examination is equivocal or confounded (sedation, hypothermia, metabolic derangement). Tc-99m HMPAO (Ceretec) or Tc-99m ECD (Neurolite) are lipophilic agents that cross the intact blood-brain barrier and are retained in viable brain tissue proportional to cerebral blood flow. In brain death, there is complete absence of intracranial uptake on planar or SPECT images (the "hollow skull" or "empty light bulb" sign), with preserved external carotid artery supply to the scalp ("hot nose" sign from preserved external carotid perfusion). Tc-99m DTPA and Tc-99m pertechnetate (which do not cross the intact BBB) can also be used in an angiographic (flow) study: absence of anterior and middle cerebral artery flow bilaterally confirms brain death, but these agents cannot evaluate posterior fossa blood flow and have been largely replaced by HMPAO/ECD.
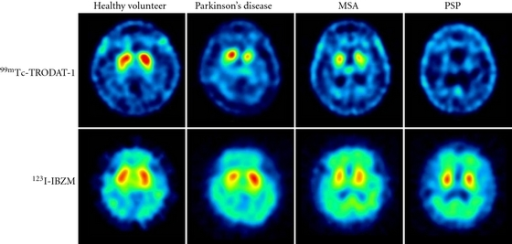
DaTscan (I-123 Ioflupane)
DaTscan uses I-123 ioflupane, which binds to the dopamine transporter (DAT) in the presynaptic terminals of nigrostriatal dopaminergic neurons. Normal images show symmetric comma-shaped or crescent-shaped uptake in both caudate nuclei and putamina. In Parkinsonian syndromes (Parkinson disease, multiple system atrophy, progressive supranuclear palsy, corticobasal degeneration), there is asymmetrically decreased putaminal uptake (the "period sign" or "dot" pattern) reflecting nigrostriatal dopaminergic degeneration. In essential tremor, DaTscan is normal. This distinction is the primary clinical indication: differentiating degenerative Parkinsonism (abnormal DaTscan) from essential tremor (normal DaTscan). DaTscan does not distinguish between the various Parkinsonian syndromes. FDA-approved thyroid blockade with Lugol solution or SSKI is given 1 hour before injection.
CSF Shunt Patency
In-111 DTPA or Tc-99m DTPA is injected into the shunt reservoir (accessed by the neurosurgeon or trained clinician through the scalp). Images are obtained at intervals (immediately, 15 min, 30 min, 1 hr, and delayed as needed). Normal: tracer flows promptly from the reservoir through the distal catheter to the peritoneal cavity (VP shunt), pleural space, or atrium (VA shunt). Obstruction is indicated by failure of tracer to clear the reservoir or failure to reach the distal end. Proximal catheter patency is assessed by briefly occluding the distal tubing and observing reflux of tracer into the ventricles.
LeVeen Shunt Patency
Peritoneovenous (LeVeen or Denver) shunt patency is assessed by injecting Tc-99m MAA or Tc-99m sulfur colloid into the peritoneal cavity. If the shunt is patent, radiotracer is detected in the lungs (MAA lodging in pulmonary capillaries) or liver/spleen (sulfur colloid RES uptake) within minutes. Failure of pulmonary or hepatic activity to appear indicates shunt obstruction. This study is straightforward and has high accuracy for confirming shunt dysfunction in patients with recurrent ascites.
Cisternography for CSF Leak
In-111 DTPA cisternography evaluates CSF rhinorrhea or otorrhea. The tracer is injected intrathecally via lumbar puncture and images are obtained at 2–4 hours, 24 hours, and 48 hours. Cotton pledgets placed in the nares are counted for radioactivity; a pledget-to-serum ratio >1.5 indicates a CSF leak. On imaging, early accumulation of tracer in the nasal cavity or middle ear (before it normally reaches the basal cisterns at 2–4 hours and convexities at 12–24 hours) confirms the leak and helps localize it. In normal pressure hydrocephalus (NPH), cisternography shows ventricular reflux of tracer at 24 hours with delayed clearance over the convexities at 48–72 hours, but this test has been largely supplanted by clinical criteria (triad of gait disturbance, urinary incontinence, dementia) and CSF drainage trials.
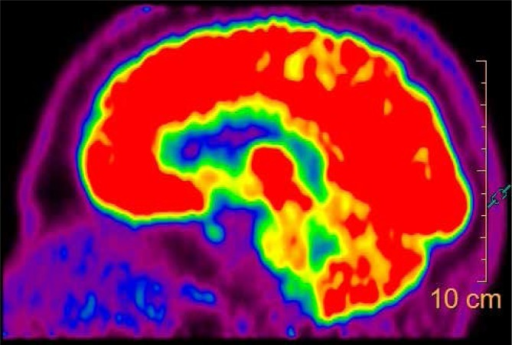
Ictal vs Interictal SPECT for Epilepsy
Ictal SPECT (injection of Tc-99m HMPAO or ECD during a seizure) shows hyperperfusion at the seizure focus. Interictal SPECT shows hypoperfusion at the same region. SISCOM (Subtraction Ictal SPECT Co-registered to MRI) digitally subtracts interictal from ictal SPECT, coregisters the difference map onto MRI, and highlights the seizure focus with high localizing value. This technique is invaluable for surgical planning in medically refractory epilepsy when MRI is non-lesional. The logistic challenge is having the radiotracer drawn up and ready for injection within 20–30 seconds of seizure onset during video-EEG monitoring, since HMPAO uptake is fixed within 30–60 seconds of injection.
I-123 MIBG Cardiac Imaging
I-123 MIBG cardiac imaging assesses sympathetic innervation of the heart. The heart-to-mediastinum (H/M) ratio on delayed images (4 hours) and the washout rate between early (15 min) and delayed images quantify cardiac sympathetic function. An H/M ratio <1.6 on delayed images is associated with increased risk of heart failure progression, sudden cardiac death, and ventricular arrhythmias. The ADMIRE-HF trial demonstrated that I-123 MIBG imaging independently predicts cardiac events in heart failure patients and can guide ICD implantation decisions. This study is FDA-approved for risk stratification in heart failure (NYHA class II–III with LVEF ≤35%). In Lewy body dementia, cardiac MIBG shows reduced H/M ratio (cardiac sympathetic denervation), helping distinguish it from Alzheimer disease (normal H/M), though this application is more established in Japan and Europe than in the US.
| Dementia Type | FDG PET Pattern | Additional Nuclear Studies |
|---|---|---|
| Alzheimer disease | Bilateral temporoparietal and posterior cingulate hypometabolism; frontal involvement later | Amyloid PET positive; tau PET (F-18 flortaucipir) positive |
| Frontotemporal dementia | Frontal and/or anterior temporal hypometabolism (asymmetric) | Amyloid PET typically negative |
| Lewy body dementia | Occipital hypometabolism (including visual cortex); temporoparietal also involved | DaTscan abnormal; cardiac MIBG low H/M ratio |
| Vascular dementia | Scattered, asymmetric cortical and subcortical defects; pattern follows vascular territories | Amyloid PET typically negative (unless mixed) |
| Normal aging | Mild symmetric frontal hypometabolism | Amyloid PET may be positive in 10–30% of elderly |
23 Theranostics Concept & PSMA
The Theranostic Paradigm
Theranostics pairs a diagnostic imaging agent with a therapeutic counterpart targeting the same molecular pathway. The diagnostic scan identifies patients whose tumors express the target (patient selection), and the therapeutic agent delivers targeted radiation to those tumors. This "see it, treat it" paradigm represents a shift from empiric treatment to precision medicine. Key theranostic pairs: Ga-68 DOTATATE (diagnostic) / Lu-177 DOTATATE (therapeutic) for NETs; Ga-68 PSMA-11 or F-18 DCFPyL (diagnostic) / Lu-177 PSMA-617 (therapeutic) for prostate cancer; I-123 MIBG (diagnostic) / I-131 MIBG (therapeutic) for pheochromocytoma/neuroblastoma; I-123 (diagnostic) / I-131 (therapeutic) for thyroid cancer.
Lu-177 PSMA-617 (Pluvicto)
Lu-177 PSMA-617 (Pluvicto) is FDA-approved for PSMA-positive metastatic castration-resistant prostate cancer (mCRPC) that has progressed after androgen receptor pathway inhibition (ARPI) and taxane-based chemotherapy. Approval was based on the VISION trial (2021), which demonstrated: median overall survival 15.3 months vs 11.3 months (HR 0.62, p < 0.001) and median radiographic progression-free survival 8.7 vs 3.4 months when Lu-177 PSMA was added to standard of care. The TheraP trial showed superiority over cabazitaxel in the second-line setting.
Patient Selection
Patients must have a positive Ga-68 PSMA-11 or F-18 DCFPyL PET/CT demonstrating: (1) PSMA-positive disease — at least one metastatic lesion with PSMA uptake greater than liver; (2) absence of PSMA-negative/FDG-positive disease (— FDG PET/CT is obtained to exclude discordant lesions that lack PSMA expression but are metabolically active, indicating dedifferentiated disease that would not respond to PSMA-targeted therapy). This dual-tracer selection strategy (PSMA-positive, FDG-negative/concordant) optimizes therapeutic benefit.
Treatment Protocol & Side Effects
Lu-177 PSMA-617 is administered as 7.4 GBq (200 mCi) IV every 6 weeks for up to 6 cycles. Post-therapy SPECT/CT at 24–72 hours confirms tracer localization to known disease sites. Side effects: xerostomia (dry mouth, 39% — PSMA is expressed in salivary glands; sialagogues and salivary gland cooling may mitigate), bone marrow suppression (anemia 32%, thrombocytopenia 17%, leukopenia 12% — monitor CBC before each cycle; hold if platelets <50K or ANC <1.0K), nausea (35%), fatigue (29%), and renal toxicity (rare, monitor creatinine). Ac-225 PSMA (alpha emitter) is being investigated for patients who progress on Lu-177 PSMA, with promising preliminary results but higher rates of xerostomia.
Emerging PSMA Applications
The PSMAfore trial demonstrated benefit of Lu-177 PSMA-617 earlier in the treatment sequence (pre-chemotherapy mCRPC after ARPI failure), expanding the eligible population. PSMA-targeted agents are also being explored in combination with immunotherapy, radiosensitizers, and DNA damage repair inhibitors (PARP inhibitors). PSMA-targeted fluorescence-guided surgery using PSMA-directed fluorescent agents is an emerging intraoperative application for identifying positive margins during radical prostatectomy. The broader concept of PSMA theranostics serves as a paradigm for precision oncology: molecular imaging selects patients, therapy targets the same molecular pathway, and post-therapy imaging confirms delivery.
Dosimetry in Theranostics
Dosimetry — the calculation of absorbed radiation dose to tumors and normal organs — is increasingly incorporated into theranostic treatment planning. The MIRD (Medical Internal Radiation Dose) schema calculates absorbed dose from: the cumulated activity in source organs (determined from serial imaging at multiple time points), the physical characteristics of the radionuclide, and the geometric relationship between source and target organs (S-factors). For Lu-177 therapies, post-therapy SPECT/CT images at 24, 72, and optionally 168 hours allow construction of time-activity curves for dose estimation. Key dose limits: kidney cumulative dose <23 Gy (to avoid nephrotoxicity), bone marrow dose <2 Gy per cycle (to limit hematotoxicity). Personalized dosimetry may allow dose escalation in patients with favorable biodistribution (improving efficacy) or dose reduction in patients at risk for toxicity (improving safety). While standardized fixed-dose regimens remain the norm for most theranostics, dosimetry-guided treatment is being evaluated in clinical trials and may represent the next frontier in precision radionuclide therapy.
24 Peptide Receptor Radionuclide Therapy
Lu-177 DOTATATE (Lutathera)
Lu-177 DOTATATE (Lutathera) is FDA-approved for SSTR-positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs). Approval was based on the NETTER-1 trial (2017), which showed: 65.2% estimated progression-free survival at 20 months vs 10.8% with high-dose octreotide LAR (HR 0.21, p < 0.0001) in midgut NETs. Objective response rate was 18% vs 3%, with clinically meaningful improvement in symptoms (diarrhea, flushing) and quality of life.
Patient Selection
Patients must have a positive Ga-68 DOTATATE PET/CT with a Krenning score ≥2 (uptake ≥ normal liver) in all target lesions. Tumors must be well-differentiated (grade 1–2; Ki-67 <20%); poorly differentiated/high-grade NETs typically lose SSTR expression and are better treated with cytotoxic chemotherapy. Additional eligibility: adequate renal function (GFR ≥30 mL/min), adequate bone marrow (Hgb ≥8, WBC ≥2K, platelets ≥75K), and ECOG performance status 0–2.
Treatment Protocol
Lu-177 DOTATATE is administered as 7.4 GBq (200 mCi) IV every 8 weeks for 4 cycles. An IV amino acid solution (lysine/arginine) is co-infused starting 30 minutes before and continuing for 4 hours after the radiopharmaceutical to provide renal protection: the positively charged amino acids competitively inhibit tubular reabsorption of the radiolabeled peptide, reducing renal radiation dose by 30–50%. Post-therapy SPECT/CT or whole-body planar imaging at 24 hours confirms tumor targeting. Anti-emetics (ondansetron) are administered prophylactically because the amino acid infusion commonly causes nausea.
Toxicity Monitoring
The primary toxicity is hematologic: grade 3–4 thrombocytopenia (~2%), lymphopenia (~9%), and rarely myelodysplastic syndrome/acute leukemia (~2% at 5 years). CBC is monitored every 4 weeks between cycles. Renal toxicity is uncommon with amino acid protection but can be dose-limiting in patients with pre-existing renal insufficiency. Hepatotoxicity may occur with extensive liver metastases (tumor lysis effect). Rare carcinoid crisis can be triggered by the treatment; octreotide rescue should be available.
PRRT Response Assessment
Response to PRRT is assessed using a combination of: (1) anatomic imaging (CT/MRI) with RECIST 1.1 criteria — noting that NET responses are often slow and stable disease may represent treatment success; (2) Ga-68 DOTATATE PET/CT for molecular response (decreased tumor uptake); (3) biochemical markers (chromogranin A, 5-HIAA for midgut NETs, neuron-specific enolase); and (4) symptom assessment. Disease control rate (CR + PR + SD) in NETTER-1 was 82.5% vs 67.3% for high-dose octreotide. In clinical practice, many patients treated with PRRT achieve prolonged stable disease rather than objective size reduction, which still represents meaningful clinical benefit in slow-growing NETs. Retreatment with additional PRRT cycles beyond the standard 4 is being evaluated in patients who initially responded and subsequently progressed, with several retrospective series showing acceptable safety and efficacy.
Y-90 DOTATOC
Y-90 DOTATOC is an alternative PRRT agent with a pure beta emitter (Y-90, higher energy, longer range ~2.5 mm vs 0.3 mm for Lu-177). Theoretical advantage for larger tumors (better cross-fire effect) but higher renal toxicity (no gamma for imaging — bremsstrahlung or PET imaging is suboptimal). Lu-177 is preferred in current practice due to its favorable therapeutic index and the ability to perform post-therapy imaging with its 208-keV gamma emission.
25 Other Radionuclide Therapies
Ra-223 Dichloride (Xofigo)
Ra-223 is an alpha-emitting calcium mimetic that targets areas of increased bone turnover in osteoblastic metastases. FDA-approved for symptomatic bone metastases in metastatic castration-resistant prostate cancer (mCRPC) without known visceral metastases. The ALSYMPCA trial (2013) demonstrated: median OS 14.9 months vs 11.3 months (HR 0.70, p < 0.001), with reduced skeletal events and improved quality of life. Dose: 55 kBq/kg IV every 4 weeks for 6 cycles. The alpha particles deposit their energy within ~50–100 µm of the bone surface, causing tumor cell death while sparing bone marrow (limited myelosuppression compared to beta emitters). Contraindication: combination with abiraterone/prednisone increased fracture risk and was harmful in the ERA-223 trial. Ra-223 must not be used with concurrent abiraterone.
I-131 MIBG Therapy
High-specific-activity I-131 MIBG (Azedra, iobenguane I-131) is FDA-approved for unresectable, locally advanced, or metastatic MIBG-avid pheochromocytoma/paraganglioma. Dose: 500 mCi (18.5 GBq) for the first cycle, 500 mCi for the second cycle at ≥90-day interval. Patients require inpatient admission in a radiation isolation room. Side effects: myelosuppression (up to 50% grade 3–4 thrombocytopenia), hypothyroidism (thyroid blockade is mandatory), nausea, and hypertension crisis (catecholamine release from tumor; alpha-blockade should be optimized beforehand). For neuroblastoma, I-131 MIBG therapy (12–18 mCi/kg) is used for relapsed/refractory disease, often combined with chemosensitization or myeloablative regimens with autologous stem cell rescue.
Y-90 Microsphere Radioembolization (TARE)
Y-90 microsphere transarterial radioembolization (TARE) delivers high-dose internal radiation to hepatic tumors. Resin microspheres (SIR-Spheres, 20–60 µm) or glass microspheres (TheraSphere, 20–30 µm) loaded with Y-90 are infused via a catheter into the hepatic artery. Indications: unresectable hepatocellular carcinoma (particularly as a bridge to transplant or downstaging), hepatic metastases (colorectal, NETs), and intrahepatic cholangiocarcinoma. Pre-treatment planning includes: (1) hepatic angiography to map hepatic arterial anatomy and embolize extrahepatic vessels (GDA, right gastric artery) to prevent non-target embolization; (2) Tc-99m MAA injection via the hepatic artery to estimate the lung shunt fraction (LSF) — if LSF >20%, treatment is contraindicated due to risk of radiation pneumonitis. Y-90 is a pure beta emitter (T½ 64.1 hours, mean range in tissue 2.5 mm); post-treatment imaging uses bremsstrahlung SPECT or Y-90 PET/CT (the small positron branching ratio of Y-90 allows PET imaging).
Bone Pain Palliation
Sm-153 EDTMP (Quadramet) and Sr-89 chloride (Metastron) are beta-emitting bone-seeking agents used for palliation of painful osteoblastic metastases. Sm-153 (T½ 1.9 days) also emits a 103-keV gamma allowing post-therapy imaging and has faster onset of pain relief (1–2 weeks). Sr-89 (T½ 50.5 days) has a longer duration of effect but prolonged myelosuppression. Both have been largely supplanted by Ra-223 (which improves survival) and external beam radiation, but remain options for diffuse, multifocal bone pain not amenable to focal radiation.
| Agent | Emission | T½ | Tissue Range | Indication | Key Trial |
|---|---|---|---|---|---|
| Ra-223 | Alpha | 11.4 days | ~50–100 µm | Bone mets CRPC | ALSYMPCA |
| Lu-177 PSMA-617 | Beta | 6.7 days | ~0.3 mm | PSMA+ mCRPC | VISION |
| Lu-177 DOTATATE | Beta | 6.7 days | ~0.3 mm | SSTR+ GEP-NETs | NETTER-1 |
| I-131 MIBG | Beta + gamma | 8 days | ~0.8 mm | Pheo/neuroblastoma | Various phase II |
| Y-90 microspheres | Beta (pure) | 64.1 hr | ~2.5 mm | Hepatic tumors (TARE) | SARAH, SIRveNIB |
| Sm-153 | Beta + gamma | 1.9 days | ~0.6 mm | Bone pain palliation | Phase III RCT |
| Sr-89 | Beta (pure) | 50.5 days | ~2.4 mm | Bone pain palliation | Phase III RCT |
Radiation Safety for Radionuclide Therapy
All therapeutic administrations requiring a written directive follow strict safety protocols. The administering physician must be an authorized user under 10 CFR 35.300 or 35.1000. Key considerations by agent: I-131 therapy (oral) — patients may be treated as outpatient if ≤33 mCi; higher doses may require hospitalization in a dedicated room with private bathroom, lead-lined walls, and continuous exposure monitoring; patients receive written instructions on contact restrictions (sleep alone for 1–3 weeks, minimize time with children/pregnant women, flush toilet twice, wash hands frequently). Lu-177 therapy — administered IV; lower gamma energy (208 keV) and lower dose rates allow same-day discharge in most cases; standard precautions for 48–72 hours post-treatment. Y-90 TARE — pure beta emitter with no external exposure risk; standard interventional radiology precautions during administration.
Y-90 TARE Planning and Post-Treatment
Detailed planning for Y-90 radioembolization includes: (1) diagnostic angiography to map hepatic arterial anatomy, identify variant anatomy (replaced or accessory hepatic arteries), and coil-embolize extrahepatic vessels (GDA, right gastric, cystic artery) to prevent non-target microsphere deposition; (2) Tc-99m MAA test injection via the planned treatment catheter position to simulate microsphere distribution and quantify lung shunt fraction; (3) Lung shunt fraction (LSF) calculation: LSF = lung counts / (lung + liver counts) × 100%. If LSF >20% (for resin microspheres) or >30 Gy to the lungs (for glass microspheres), treatment is contraindicated due to risk of radiation pneumonitis. Dose reduction is required if LSF is 10–20%. (4) Dose calculation using the body surface area method (resin), partition model, or MIRD-based approach (glass). Post-treatment, Y-90 PET/CT (exploiting the small positron branching fraction of Y-90) or bremsstrahlung SPECT confirms microsphere distribution and allows dosimetry verification.
26 Radiopharmaceutical Master Table
| Agent | Mechanism | T½ | Energy | Key Organ Dose | Primary Clinical Use |
|---|---|---|---|---|---|
| Tc-99m pertechnetate | Trapped by NIS (thyroid, salivary, gastric) | 6.02 hr | 140 keV γ | Thyroid, GI | Thyroid imaging, Meckel scan, salivary gland |
| Tc-99m MDP | Chemisorption to hydroxyapatite | 6.02 hr | 140 keV γ | Bladder | Bone scintigraphy |
| Tc-99m sestamibi | Mitochondrial membrane potential | 6.02 hr | 140 keV γ | Gallbladder, GI | Myocardial perfusion, parathyroid |
| Tc-99m tetrofosmin | Mitochondrial membrane potential | 6.02 hr | 140 keV γ | Gallbladder, GI | Myocardial perfusion |
| Tc-99m MAG3 | Tubular secretion | 6.02 hr | 140 keV γ | Bladder | Renal function, diuretic renogram |
| Tc-99m DMSA | Proximal tubular binding | 6.02 hr | 140 keV γ | Kidneys | Renal cortical imaging |
| Tc-99m DTPA | Glomerular filtration | 6.02 hr | 140 keV γ | Bladder | GFR measurement, ventilation aerosol |
| Tc-99m MAA | Capillary blockade | 6.02 hr | 140 keV γ | Lungs | Lung perfusion, shunt quantification |
| Tc-99m HMPAO | Lipophilic brain agent (BBB crossing) | 6.02 hr | 140 keV γ | Kidneys, GI | Brain perfusion (death, epilepsy), WBC labeling |
| Tc-99m sulfur colloid | RES phagocytosis | 6.02 hr | 140 keV γ | Liver, spleen | GI bleeding, liver/spleen, marrow, SLN, GES |
| Tc-99m mebrofenin | Hepatocyte uptake (bilirubin analog) | 6.02 hr | 140 keV γ | Gallbladder, GI | Hepatobiliary (HIDA) scan |
| Tc-99m RBCs (labeled) | Intravascular blood pool | 6.02 hr | 140 keV γ | Heart, blood | MUGA, GI bleeding |
| F-18 FDG | Glucose analog — metabolic trapping | 110 min | 511 keV (annihilation) | Bladder, brain | Oncology, cardiac, neuro, infection |
| F-18 NaF | Hydroxyapatite exchange | 110 min | 511 keV | Bone, bladder | Bone PET |
| Ga-68 DOTATATE | SSTR2 binding | 68 min | 511 keV | Spleen, kidneys | NET imaging |
| Ga-68 PSMA-11 | PSMA binding | 68 min | 511 keV | Kidneys, salivary | Prostate cancer imaging |
| Rb-82 | K+ analog (Na/K-ATPase) | 75 sec | 511 keV | Kidneys | Cardiac PET perfusion |
| I-123 | NIS trapping & organification | 13.2 hr | 159 keV γ | Thyroid | Thyroid imaging, RAIU |
| I-123 ioflupane | Dopamine transporter binding | 13.2 hr | 159 keV γ | Bladder | DaTscan (Parkinsonian syndromes) |
| I-123 MIBG | Norepinephrine transporter | 13.2 hr | 159 keV γ | Liver, spleen | Pheochromocytoma, neuroblastoma, cardiac |
| I-131 | NIS trapping & organification | 8.02 days | 364 keV γ + β− | Thyroid (therapeutic) | Thyroid therapy, post-therapy WBS |
| Tl-201 | K+ analog (Na/K-ATPase) | 73 hr | 69–83 keV X-rays | Kidneys, testes | Myocardial viability (historical) |
| In-111 oxine | WBC labeling (lipophilic chelate) | 67 hr | 171 + 245 keV γ | Spleen | Infection imaging (WBC scan) |
| In-111 DTPA | CSF tracer (intrathecal) | 67 hr | 171 + 245 keV γ | Spinal cord | Cisternography, shunt patency |
| Ga-67 citrate | Transferrin/lactoferrin binding | 78 hr | 93/185/300 keV γ | Colon, bone | Infection (historical), sarcoidosis |
| Lu-177 DOTATATE | SSTR2 binding (β− therapy) | 6.7 days | 208 keV γ + β− | Kidneys, marrow | PRRT for NETs |
| Lu-177 PSMA-617 | PSMA binding (β− therapy) | 6.7 days | 208 keV γ + β− | Kidneys, salivary | mCRPC therapy |
| Ra-223 | Calcium mimetic (α therapy) | 11.4 days | α particles | Bone surface | Bone-met CRPC therapy |
| Y-90 | Pure β− emitter (microspheres) | 64.1 hr | β− (no primary γ) | Liver | TARE/radioembolization |
27 Classification & Scoring Systems
Deauville 5-Point Scale (Lymphoma)
See section 6 for complete description. In summary: Scores 1–3 at end-of-treatment = complete metabolic response for most lymphoma subtypes. Score 4–5 = residual metabolically active disease. The reference structures are the mediastinal blood pool (upper threshold for score 2) and the liver (upper threshold for score 3).
Lugano Classification (Lymphoma Staging)
| Stage | Involvement |
|---|---|
| I | Single nodal region or single extranodal site (IE) |
| II | Two or more nodal regions on the same side of the diaphragm, or contiguous extranodal extension (IIE) |
| III | Nodal regions on both sides of the diaphragm; spleen = nodal |
| IV | Diffuse or disseminated extranodal involvement (liver, bone marrow, lungs — non-contiguous) |
The Lugano classification incorporates PET/CT for FDG-avid lymphomas: PET/CT is the standard for staging (replaces bone marrow biopsy in Hodgkin lymphoma and DLBCL if PET shows multifocal osseous involvement) and response assessment (using Deauville). For non-FDG-avid lymphomas (e.g., marginal zone), CT-based criteria (Cheson criteria) remain the standard for response assessment.
RECIST 1.1 vs PERCIST
RECIST 1.1 (Response Evaluation Criteria in Solid Tumors) uses CT-based measurements of target lesion diameters for response assessment: complete response (disappearance of all target lesions), partial response (≥30% decrease in sum of longest diameters), progressive disease (≥20% increase in sum + ≥5 mm absolute increase, or new lesions), stable disease (neither PR nor PD). PERCIST and RECIST may give discordant results because metabolic changes often precede anatomic changes. In general, PERCIST detects response (or progression) earlier than RECIST, which is particularly relevant for targeted therapies and immunotherapy. For immunotherapy response assessment, iPERCIST and LYRIC criteria account for pseudoprogression (initial apparent increase in tumor size/uptake due to immune cell infiltration before actual response).
Hopkins Criteria (Prostate Cancer PSMA PET)
The Hopkins molecular imaging criteria (miPSMA score) standardize interpretation of PSMA PET/CT:
| Score | Uptake Level | Description |
|---|---|---|
| 1 | Below blood pool | Benign or physiologic uptake |
| 2 | Equal to or above blood pool but below liver | Likely benign in most contexts |
| 3 | Above liver | Equivocal; further evaluation or follow-up needed |
| 4 | Markedly above liver (high confidence) | Consistent with prostate cancer |
Scores 1–2 in soft tissue or bone are generally considered negative; scores 3–4 are considered positive for prostate cancer. The liver serves as the reference organ, analogous to the Deauville score in lymphoma. miPSMA criteria improve interobserver agreement and standardize reporting for clinical trials and routine practice.
PROMISE/EANM Criteria (PSMA PET)
The PROMISE criteria provide a structured molecular imaging TNM classification for PSMA PET, mapping PSMA-positive lesions to anatomic compartments (prostate/bed, regional LN, distant LN, bone, visceral) and scoring uptake intensity (visual assessment relative to blood pool, liver, and parotid glands). This system enables standardized staging and response assessment in prostate cancer, facilitating multicenter trials and clinical communication.
PERCIST 1.0 Criteria — Full Detail
PERCIST uses SULpeak (lean body mass corrected, measured in a 1-cm³ sphere) of the hottest single target lesion. Baseline requirements: the target lesion must have SULpeak ≥1.5 × mean liver SUL + 2 SD. Response categories:
| Category | Criteria |
|---|---|
| CMR | Disappearance of all metabolically active disease; target lesion SUL ≤ mean liver SUL + 2 SD; no new FDG-avid lesions |
| PMR | ≥30% decrease in SULpeak of the target lesion AND ≥0.8 SUL unit decrease (absolute); no increase >30% in any other lesion; no new lesions |
| SMD | Not qualifying for CMR, PMR, or PMD |
| PMD | ≥30% increase in SULpeak of the target lesion AND ≥0.8 SUL unit increase (absolute); or visible increase in extent; or new FDG-avid lesions (not inflammatory/infectious) |
PIOPED Criteria
See section 20. Classifies V/Q scan probability for PE as normal, very low, low, intermediate, or high. A normal scan effectively excludes PE. A high-probability scan with high clinical pretest probability is diagnostic (~96% PPV). Intermediate probability is the clinical challenge, often requiring CTA for definitive diagnosis. Key PIOPED data: a high-probability V/Q scan with a high clinical pretest probability (Wells score >6) has a ~96% positive predictive value for PE. A normal/near-normal scan with low clinical pretest probability has a <4% chance of PE. The combination of clinical pretest probability (Wells or Geneva score) with V/Q scan results is essential for clinical decision-making — the V/Q result should never be interpreted in isolation.
Krenning Score (Somatostatin Receptor Imaging)
| Score | Uptake Level | Reference Structure | Clinical Significance |
|---|---|---|---|
| 0 | No uptake | Background | SSTR-negative; PRRT unlikely to benefit |
| 1 | Very low | Less than normal liver | Low SSTR expression; PRRT unlikely |
| 2 | Moderate | Equal to normal liver | Moderate expression; PRRT may benefit |
| 3 | High | Greater than normal liver | High expression; PRRT candidate |
| 4 | Very high | Greater than normal spleen | Very high expression; excellent PRRT candidate |
28 Patient Preparation Protocols
| Study | Preparation |
|---|---|
| FDG PET/CT (oncology) | Fast 4–6 hr; blood glucose <200 mg/dL; avoid strenuous exercise 24 hr; IV hydration; void before scan; warm/quiet uptake room 60 min |
| FDG PET/CT (cardiac viability) | Glucose load (50 g PO) + insulin as needed to shift metabolism to glucose; or glucose-insulin clamp |
| FDG PET/CT (cardiac sarcoidosis) | Fast ≥18 hr; high-fat/very-low-carb diet day before (<5 g carbs); optional IV heparin 50 U/kg 15 min before FDG |
| Thyroid scan/RAIU | Stop antithyroid drugs per clinical protocol; low-iodine diet if preceding I-131 therapy; avoid iodinated contrast ≥4–8 weeks before; stop amiodarone months before |
| I-131 therapy (DTC) | TSH ≥30 (THW or Thyrogen); low-iodine diet × 1–2 weeks; avoid contrast ≥6–8 weeks; pregnancy test; review radiation safety instructions |
| MPI (exercise stress) | NPO 4 hr; hold caffeine 12–24 hr; hold beta-blockers/CCBs if tolerated (to allow adequate HR response); comfortable clothes/shoes |
| MPI (vasodilator stress) | No caffeine 12–24 hr (including decaf coffee, tea, chocolate, sodas, energy drinks); hold dipyridamole 48 hr; hold aminophylline/theophylline 48 hr |
| Parathyroid sestamibi | No specific preparation; may need to hold calcium channel blockers in some protocols |
| Bone scan | Well hydrated; void frequently; remove metallic objects; imaging 2–4 hr post-injection |
| HIDA scan | Fast 4–6 hr (but not >24 hr — prolonged fasting causes GB distension and non-filling from cystic duct sludge); if fasting >24 hr, give sincalide to empty GB before study |
| Gastric emptying study | NPO overnight; stop prokinetics 48 hr; stop opioids/anticholinergics if clinically feasible; blood glucose <275 in diabetics; no smoking morning of test |
| MIBG scan | Thyroid blockade (SSKI or Lugol solution); stop interfering medications (TCAs 6 weeks, labetalol 72 hr, pseudoephedrine 48 hr); list of interfering drugs should be provided to patient |
| DaTscan | Thyroid blockade (SSKI 1 hr before injection); no need to stop dopaminergic medications (they do not affect DAT density) |
| Brain death study | No specific patient preparation; ensure clinical brain death criteria have been attempted first |
| Ga-68 DOTATATE PET | Short-acting octreotide: hold 24 hr before; long-acting octreotide LAR: ideally image just before next dose (4–6 weeks); adequate hydration; void before scan |
| Captopril renogram | Hold ACE inhibitors/ARBs 3–5 days before; adequate hydration; captopril 25–50 mg PO 1 hr before scan; monitor BP |
| Lu-177 DOTATATE (Lutathera) | Hold short-acting octreotide 24 hr; long-acting LAR: time scan before next injection; adequate hydration; antiemetic prophylaxis; amino acid infusion for renal protection; CBC and renal function within 2 weeks; ECOG ≤2 |
| Lu-177 PSMA-617 (Pluvicto) | PSMA PET confirming PSMA-positive disease; FDG PET to exclude PSMA-negative/FDG-positive discordant disease; adequate CBC and renal function; ECOG ≤2; no myelosuppressive therapy within 4 weeks |
| Ra-223 (Xofigo) | No concurrent abiraterone/prednisone (ERA-223 safety concern); adequate CBC; no visceral metastases; confirm osteoblastic bone disease on bone scan or CT |
| Y-90 TARE | Pre-treatment angiography and MAA mapping study; LSF <20%; adequate liver function (bilirubin <2, no hepatic encephalopathy); portal vein patent (relative); ECOG ≤2 |
29 Normal Biodistribution Patterns
| Radiopharmaceutical | Normal Biodistribution |
|---|---|
| F-18 FDG | Brain (intense, most avid organ), liver (moderate, reference structure), myocardium (variable — depends on fasting/insulin state), GI (mild variable), kidneys/bladder (renal excretion), bone marrow (mild diffuse), tonsils/Waldeyer ring (lymphoid), salivary glands (mild); brown fat activation: symmetric cervical/supraclavicular/paravertebral |
| Tc-99m MDP | Skeleton (diffuse, proportional to osteoblastic activity), kidneys and bladder (renal excretion), mild soft tissue background; absent in thyroid and liver. Sternum may show focal uptake at manubrium-body junction (normal variant) |
| Tc-99m pertechnetate | Thyroid, salivary glands (parotid, submandibular), gastric mucosa, choroid plexus; excreted by kidneys |
| Tc-99m sestamibi | Heart (target organ), liver, gallbladder (early), GI, thyroid, salivary glands; renal excretion |
| Tc-99m MAG3 | Kidneys (rapid tubular secretion → renal pelvis → ureters → bladder); liver uptake if renal failure |
| Tc-99m DMSA | Renal cortex (fixed binding, ~40% at 6 hr); minimal soft tissue and bladder activity |
| Tc-99m MAA | Lungs (uniform, proportional to pulmonary blood flow); small fraction may reach kidneys/brain in right-to-left shunt |
| Tc-99m sulfur colloid | Liver (85%), spleen (10%), bone marrow (5%) — reticuloendothelial system phagocytosis |
| Tc-99m mebrofenin | Liver (hepatocyte uptake, 5 min) → biliary ducts → gallbladder (30–60 min) → bowel (60 min) |
| Tc-99m HMPAO | Brain (proportional to cerebral blood flow); highest in gray matter (cortex, basal ganglia, thalami, cerebellum); also kidneys, GI (excretion) |
| Ga-68 DOTATATE | Spleen (most intense), adrenals, pituitary, liver, kidneys, uncinate process of pancreas, small bowel, urinary bladder; low lung and bone marrow |
| PSMA PET (Ga-68/F-18) | Lacrimal glands, parotid and submandibular salivary glands (intense), liver, spleen, small bowel, kidneys and ureters, mild bowel; ganglia (sympathetic chain) |
| I-123/I-131 | Thyroid (NIS mediated), salivary glands, gastric mucosa, nasal mucosa, bladder (renal excretion), small bowel; breast (if lactating) |
| I-123 MIBG | Liver (most intense), salivary glands, heart (adrenergic innervation), adrenal medulla (mild), bladder (renal excretion), nasal mucosa |
| In-111/Tc-99m WBC | Liver, spleen (most intense), bone marrow; NO normal bowel activity for In-111 (bowel activity = abnormal); Tc-99m HMPAO WBC: bowel and kidney activity appears by 4 hr (normal excretion) |
| Ga-67 citrate | Liver, spleen, bone marrow, salivary glands, lacrimal glands, nasopharynx, breast (lactating); bowel excretion by 48–72 hr |
| Tl-201 | Heart (target), kidneys, liver, skeletal muscle, thyroid, salivary glands; redistribution occurs over hours |
Key Biodistribution Pearls
FDG brain uptake: The brain is the most FDG-avid organ (~6–8% of injected dose), with cortical gray matter showing intense uniform uptake. Focal cortical hypometabolism indicates neurodegeneration, stroke, or post-ictal changes. The caudate and putamen are normally FDG-avid. Renal activity on bone scan: Normal kidneys should be visible on Tc-99m MDP bone scan; absence of renal activity (with excellent skeletal contrast) is the hallmark of the superscan. Thyroid activity on Tc-99m sestamibi: Mild symmetric thyroid uptake is normal; focal increased thyroid uptake warrants ultrasound evaluation (incidental thyroid incidentaloma on sestamibi scan, which may represent thyroid cancer in ~2% of cases). Bowel activity on In-111 WBC scan: Any bowel activity on In-111 labeled WBC scan is abnormal (unlike Tc-99m HMPAO WBC, which normally shows bowel and kidney activity after 4 hours). In-111 WBC bowel activity suggests inflammatory bowel disease, pseudomembranous colitis, or GI bleeding. Diffuse hepatic FDG uptake: The liver serves as the reference organ for many PET interpretations; diffusely increased hepatic FDG uptake above the blood pool may indicate steatohepatitis, drug-induced hepatitis, or hepatic infiltration.
30 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| AC | Attenuation correction |
| ACR | American College of Radiology |
| ALARA | As Low As Reasonably Achievable |
| ATA | American Thyroid Association |
| BBB | Blood-brain barrier |
| Bq | Becquerel (SI unit of activity; 1 decay/sec) |
| BSA | Body surface area |
| CCK | Cholecystokinin (sincalide) |
| CFR | Coronary flow reserve |
| Ci | Curie (traditional unit of activity; 3.7 × 10¹⁰ dps) |
| CMR | Complete metabolic response |
| CRPC | Castration-resistant prostate cancer |
| CT | Computed tomography |
| CTA | CT angiography |
| DAT | Dopamine transporter |
| DMSA | Dimercaptosuccinic acid |
| DPM | Disintegrations per minute |
| DTC | Differentiated thyroid cancer |
| DTPA | Diethylenetriaminepentaacetic acid |
| EANM | European Association of Nuclear Medicine |
| EC | Electron capture |
| ECD | Ethyl cysteinate dimer (Neurolite) |
| EDV | End-diastolic volume |
| EF | Ejection fraction |
| ERPF | Effective renal plasma flow |
| ESV | End-systolic volume |
| FBP | Filtered back projection |
| FDG | Fluorodeoxyglucose |
| FET | Fluoroethyltyrosine |
| FNA | Fine-needle aspiration |
| FUO | Fever of unknown origin |
| GBEF | Gallbladder ejection fraction |
| GBq | Gigabecquerel (10⁹ Bq) |
| GDA | Gastroduodenal artery |
| GEP-NET | Gastroenteropancreatic neuroendocrine tumor |
| GES | Gastric emptying study |
| GFR | Glomerular filtration rate |
| GLUT | Glucose transporter |
| Gy | Gray (SI unit of absorbed dose) |
| HE | High-energy (collimator) |
| HIDA | Hepatobiliary iminodiacetic acid |
| HMPAO | Hexamethylpropyleneamine oxime (Ceretec) |
| IDA | Iminodiacetic acid |
| IT | Isomeric transition |
| keV | Kilo-electron volt |
| LBBB | Left bundle branch block |
| LEAP | Low-energy all-purpose (collimator) |
| LEHR | Low-energy high-resolution (collimator) |
| LET | Linear energy transfer |
| LSF | Lung shunt fraction |
| LVEF | Left ventricular ejection fraction |
| MAA | Macroaggregated albumin |
| MAG3 | Mercaptoacetyltriglycine |
| MBF | Myocardial blood flow |
| MBq | Megabecquerel (10⁶ Bq) |
| mCi | Millicurie |
| mCRPC | Metastatic castration-resistant prostate cancer |
| MDP | Methylene diphosphonate |
| ME | Medium-energy (collimator) |
| MIBG | Metaiodobenzylguanidine |
| MIP | Maximum intensity projection |
| MPI | Myocardial perfusion imaging |
| MPHR | Maximum predicted heart rate |
| MRI | Magnetic resonance imaging |
| MUGA | Multigated acquisition (radionuclide ventriculography) |
| NaI(Tl) | Sodium iodide (thallium-doped) crystal |
| NET | Neuroendocrine tumor |
| NIS | Sodium-iodide symporter |
| NPH | Normal pressure hydrocephalus |
| NRC | Nuclear Regulatory Commission |
| OSEM | Ordered subset expectation maximization |
| PE | Pulmonary embolism |
| PERCIST | PET Response Criteria in Solid Tumors |
| PET | Positron emission tomography |
| PHA | Pulse height analyzer |
| PIOPED | Prospective Investigation of Pulmonary Embolism Diagnosis |
| PMD | Progressive metabolic disease |
| PMR | Partial metabolic response |
| PMT | Photomultiplier tube |
| PRRT | Peptide receptor radionuclide therapy |
| PSMA | Prostate-specific membrane antigen |
| RAIU | Radioactive iodine uptake |
| RBC | Red blood cell |
| RES | Reticuloendothelial system |
| rhTSH | Recombinant human thyroid-stimulating hormone (Thyrogen) |
| ROI | Region of interest |
| RSO | Radiation Safety Officer |
| RVG | Radionuclide ventriculography |
| SD | Standard deviation |
| SDS | Summed difference score |
| SISCOM | Subtraction ictal SPECT coregistered to MRI |
| SLN | Sentinel lymph node |
| SMD | Stable metabolic disease |
| SNMMI | Society of Nuclear Medicine and Molecular Imaging |
| SPECT | Single-photon emission computed tomography |
| SRS | Summed rest score |
| SSS | Summed stress score |
| SSTR | Somatostatin receptor |
| SUL | SUV corrected for lean body mass |
| SUV | Standardized uptake value |
| Sv | Sievert (SI unit of equivalent/effective dose) |
| TARE | Transarterial radioembolization |
| TEDE | Total effective dose equivalent |
| THW | Thyroid hormone withdrawal |
| TID | Transient ischemic dilation |
| TOF | Time-of-flight (PET) |
| V/Q | Ventilation/perfusion |
| VOI | Volume of interest |
| WBC | White blood cell |
| WBS | Whole-body scan |
1. Tc-99m is the most widely used radionuclide in diagnostic nuclear medicine — ideal 140 keV gamma, 6 hr half-life, generator-produced. 2. FDG PET sensitivity depends on blood glucose (<200 mg/dL required), fasting duration (≥4 hr), and uptake time (~60 min). 3. In LBBB/paced rhythm, use vasodilator (not exercise) stress to avoid false-positive septal defects. 4. Caffeine must be withheld ≥12–24 hr before vasodilator stress testing. 5. I-131 requires permanent breastfeeding cessation for that child. 6. A hot thyroid nodule is almost never malignant; a cold nodule has 5–15% malignancy risk. 7. The superscan mimics a normal bone scan — look for absent kidney activity. 8. Multiple myeloma is poorly detected by bone scan — use FDG PET or CT. 9. In-111 WBC scan should never show bowel activity (unlike Tc-99m HMPAO WBC). 10. Normal GBEF on CCK-HIDA is >35%. 11. Gastric emptying requires a full 4-hour study for accuracy. 12. DaTscan differentiates Parkinsonism (abnormal) from essential tremor (normal). 13. Brain death shows the "hollow skull" sign with absent intracranial tracer uptake. 14. Cardiac sarcoidosis FDG PET requires ≥18 hr fast + high-fat/low-carb diet for myocardial suppression. 15. Krenning score ≥2 is required for PRRT eligibility. 16. PSMA PET has largely replaced conventional imaging for prostate cancer biochemical recurrence. 17. Ra-223 must not be combined with abiraterone/prednisone (ERA-223 safety signal). 18. Y-90 TARE requires pre-treatment MAA lung shunt fraction <20%. 19. The flare phenomenon on bone scan after therapy represents healing, not progression. 20. Effective half-life is always shorter than both physical and biological half-life.