Radiology
Chest X-ray, CT, MRI, ultrasound, fluoroscopy, contrast agents, radiation safety, structured reporting, and every imaging protocol, classification system, critical finding, and interpretive framework across the full scope of diagnostic radiology.
01 Physics of Medical Imaging
All diagnostic imaging begins with the interaction between energy and tissue. Understanding the physics of image formation — from X-ray production to digital detection — is fundamental to selecting appropriate protocols, recognizing artifacts, and optimizing image quality while minimizing patient dose.
X-Ray Production
X-rays are generated when high-speed electrons strike a metal target (usually tungsten) in a vacuum tube. The cathode (negative electrode) emits electrons via thermionic emission from a heated tungsten filament. These electrons are accelerated across a potential difference (kVp) toward the anode (positive electrode). Upon striking the anode, two types of X-ray production occur: Bremsstrahlung radiation (braking radiation, ~80% of X-ray beam) when electrons decelerate near tungsten nuclei, producing a continuous spectrum; and characteristic radiation (~20%) when incident electrons eject inner-shell electrons from tungsten atoms, and outer-shell electrons fill the vacancy, emitting photons at discrete energies specific to the target element.
Photon Interactions with Matter
When X-ray photons enter patient tissue, five interactions are possible, but two dominate the diagnostic energy range (25-150 keV):
Photoelectric effect: the incident photon is completely absorbed by an inner-shell electron, which is ejected from the atom. The probability is proportional to Z3/E3 (atomic number cubed divided by energy cubed). This means the photoelectric effect is strongly dependent on tissue composition — bone (high Z due to calcium) absorbs far more than soft tissue. The photoelectric effect dominates at lower energies and in high-Z materials. It produces excellent subject contrast with no scatter but delivers higher patient dose.
Compton scattering: the incident photon interacts with an outer-shell electron, ejecting it and continuing in a different direction with reduced energy. Compton scattering depends on electron density (roughly proportional to physical density) but is independent of atomic number. It dominates at higher diagnostic energies and is the primary source of scatter radiation, which degrades image contrast and contributes to occupational exposure.
kVp vs mAs
kVp (kilovoltage peak) controls the maximum energy of the X-ray beam and determines penetrating ability. Increasing kVp increases the quantity and quality (energy) of X-rays, reduces contrast, and decreases patient dose per unit of image information. mAs (milliampere-seconds) = tube current x exposure time; it controls the quantity of X-rays produced without altering their energy. Doubling mAs doubles the number of photons and dose, reducing quantum noise. The 15% rule: increasing kVp by 15% has approximately the same effect on detector exposure as doubling mAs, but with lower patient dose.
Attenuation & CT Hounsfield Units
Attenuation refers to the reduction in X-ray beam intensity as it passes through tissue. The linear attenuation coefficient (mu) describes the fraction of photons removed per unit thickness for a given material. CT assigns numerical values — Hounsfield Units (HU) — based on attenuation relative to water: HU = 1000 x (mu_tissue - mu_water) / mu_water.
| Material | HU (typical) | Clinical Notes |
|---|---|---|
| Air | -1000 | Pneumothorax, pneumoperitoneum, pneumomediastinum |
| Fat | -100 to -50 | Lipomas, adrenal myelolipomas, dermoid cysts contain macroscopic fat |
| Water | 0 | Reference standard; simple cysts measure 0-20 HU |
| Soft tissue / muscle | +40 to +60 | Most solid organs measure in this range |
| Acute blood (unclotted) | +40 to +60 | Fresh hemorrhage is hyperdense on non-contrast CT |
| Clotted blood | +60 to +80 | Acute subdural or epidural hematoma |
| Calcium / cortical bone | +700 to +3000 | Calcified structures appear bright white |
| Metal | >+3000 | Streak artifact from hardware |
Digital Radiography — CR vs DR
Computed Radiography (CR) uses a photostimulable phosphor imaging plate that stores the X-ray energy pattern as a latent image. The plate is read by a laser scanner, which stimulates luminescence proportional to absorbed X-ray energy. CR is portable and retrofits existing X-ray equipment but has lower detective quantum efficiency (DQE) and slower workflow. Digital Radiography (DR) uses flat-panel detectors with either indirect conversion (scintillator + photodiode array) or direct conversion (amorphous selenium). DR provides higher DQE, faster image acquisition, lower dose potential, and real-time preview. DR has largely replaced CR in modern departments.
CT Technology
Modern CT scanners use helical (spiral) acquisition with multidetector arrays (MDCT), enabling volumetric data acquisition in a single breath hold. Current scanners employ 64 to 320 detector rows. Dual-energy CT (DECT) acquires data at two different kVp settings simultaneously, enabling material decomposition (e.g., distinguishing uric acid from calcium stones, virtual non-contrast images, iodine maps for perfusion assessment). CT reconstruction has evolved from filtered back projection (FBP) to iterative reconstruction (IR) algorithms that reduce noise, enabling dose reduction of 30-50% without sacrificing image quality.
MRI Physics — Brief Overview
MRI exploits the magnetic properties of hydrogen protons (abundant in water and fat). Placed in a strong external magnetic field (B0, typically 1.5T or 3T), protons align and precess at the Larmor frequency (42.58 MHz/T for hydrogen). Radiofrequency (RF) pulses tip protons into the transverse plane; as they relax back to equilibrium, they emit RF signals detected by receiver coils. T1 relaxation (spin-lattice): recovery of longitudinal magnetization — fast in fat (bright on T1), slow in water (dark on T1). T2 relaxation (spin-spin): decay of transverse magnetization — fast in solids (dark on T2), slow in water (bright on T2). Image contrast is manipulated by adjusting TR (repetition time) and TE (echo time).
Ultrasound Physics
Diagnostic ultrasound uses sound waves at frequencies of 2-18 MHz, produced by piezoelectric crystals that convert electrical energy to mechanical (sound) waves and vice versa. Sound waves travel through tissue and are reflected at interfaces between tissues of different acoustic impedance (Z = density x speed of sound). Greater impedance mismatch = stronger reflection. Ultrasound cannot penetrate air or bone effectively, which limits its applications but also defines its strengths — it excels for fluid-filled structures, soft tissue, and real-time dynamic assessment. Doppler ultrasound detects frequency shifts in reflected sound waves caused by moving structures (red blood cells), enabling assessment of blood flow direction, velocity, and volume.
Transducer selection determines frequency and imaging depth. Linear array (7-15 MHz): high resolution, shallow depth — thyroid, breast, vascular, musculoskeletal, superficial soft tissues. Curvilinear (convex) array (2-6 MHz): lower resolution, greater depth — abdomen, pelvis, obstetric. Phased array (1-5 MHz): small footprint for intercostal access — echocardiography, focused abdominal assessment. Endocavitary (5-10 MHz): transvaginal (gynecology, early pregnancy) and transrectal (prostate). Higher frequency = better resolution but less penetration; lower frequency = greater penetration but coarser resolution.
Common Ultrasound Artifacts
Posterior acoustic shadowing: signal void deep to a highly reflective or absorptive interface — gallstones, renal calculi, calcifications. Posterior acoustic enhancement: increased brightness deep to a fluid-filled structure — confirms cystic nature. Reverberation: repeated reflections between two highly reflective interfaces producing parallel echogenic lines — A-lines in normal lung. Comet-tail artifact: a type of reverberation from small reflective interfaces — B-lines in lung (interstitial edema), cholesterol crystals in gallbladder. Mirror artifact: duplication of a structure across a strong reflector (diaphragm — liver lesions may appear to be in the lung). Twinkle artifact: color Doppler signal behind a rough, irregular surface — useful for detecting small calculi that may not cast a classic shadow.
02 Radiation Safety & ALARA
Ionizing radiation carries both deterministic and stochastic risks. The principle of ALARA (As Low As Reasonably Achievable) mandates that every imaging study using ionizing radiation must be justified (the benefit outweighs the risk) and optimized (the lowest dose consistent with diagnostic quality).
Effective Dose by Common Examination
| Examination | Effective Dose (mSv) | Equivalent CXRs |
|---|---|---|
| Chest X-ray (PA) | 0.02 | 1 |
| Extremity radiograph | 0.001 | 0.05 |
| Mammogram (bilateral) | 0.4 | 20 |
| Lumbar spine X-ray | 1.5 | 75 |
| CT head | 2 | 100 |
| CT chest | 7 | 350 |
| CT abdomen/pelvis | 10 | 500 |
| CT coronary angiography | 12 | 600 |
| Barium enema (fluoroscopy) | 8 | 400 |
| Annual background radiation (US) | ~3 | 150 |
Deterministic vs Stochastic Effects
Deterministic effects have a threshold dose below which the effect does not occur; severity increases with dose above the threshold. Examples: skin erythema (threshold ~2 Gy), epilation (threshold ~3 Gy), cataract formation (threshold ~0.5 Gy cumulative), radiation burns. These are relevant during prolonged fluoroscopic procedures. Stochastic effects have no threshold; the probability (not severity) of the effect increases with dose. The primary stochastic effect is cancer induction. The linear no-threshold (LNT) model assumes any radiation dose, no matter how small, carries some cancer risk — this model underlies all radiation protection standards, though it remains debated at very low doses.
Dose Metrics in CT
CTDIvol (CT Dose Index — volume): the average absorbed dose within the scan volume for a single rotation, measured in mGy. Displayed on the scanner console before each acquisition. DLP (Dose-Length Product): CTDIvol x scan length (cm), measured in mGy-cm. Reflects the total dose burden for the entire scan. Effective dose: DLP x conversion factor (k, organ-specific), measured in mSv. Allows comparison of radiation burden across different examination types and body regions. For chest CT, k ≈ 0.014 mSv/(mGy-cm); for abdomen/pelvis CT, k ≈ 0.015.
Automatic tube current modulation (ATCM) adjusts mA in real-time based on patient attenuation — reduces dose in thinner body sections. Iterative reconstruction allows 30-50% dose reduction vs filtered back projection. Reduced kVp (e.g., 100 kVp for CT angiography in smaller patients) decreases dose and paradoxically increases iodine contrast enhancement. Scan length restriction: limit coverage to the clinical question — do not scan from skull base to pelvis when only the chest is indicated. Shielding: bismuth breast shields in chest CT are falling out of favor; tube current modulation is preferred.
Image Gently & Image Wisely Campaigns
The Image Gently campaign (Alliance for Radiation Safety in Pediatric Imaging) promotes pediatric dose reduction through awareness, education, and protocol optimization. Its four pillars: child-size the kVp and mAs, scan only the indicated region, scan only when indicated, and scan once (avoid multiphase studies in children). The Image Wisely campaign targets adult imaging with similar principles and also addresses referring clinicians through the "Choosing Wisely" initiative — discouraging imaging that does not add value (e.g., lumbar spine MRI for acute low back pain without red flags).
Radiation in Pregnancy
The fetus is most sensitive to radiation during organogenesis (weeks 2-8) and the early fetal period (weeks 8-15). The 50 mGy threshold is the commonly cited level below which deterministic effects on the fetus (microcephaly, intellectual disability) have not been demonstrated. Most diagnostic imaging studies deliver far less than this to the fetus — a chest CT delivers approximately 0.01-0.66 mGy fetal dose; even a CT abdomen/pelvis delivers approximately 25 mGy. A single diagnostic study should never be withheld solely due to pregnancy if clinically indicated. For PE in pregnancy, CTPA is preferred over V/Q scan because the fetal dose is lower (0.003-0.131 mGy vs 0.1-0.37 mGy), though V/Q delivers lower maternal breast dose.
Occupational Radiation Safety
Annual occupational dose limits: whole-body 50 mSv (with 5-year average ≤20 mSv), lens of eye 20 mSv/year, extremities 500 mSv. Protection principles — the three pillars: Time (minimize time in radiation field), Distance (inverse square law — doubling distance reduces exposure by 75%), Shielding (lead aprons 0.5 mm Pb reduce scatter by ~90%, thyroid shields, leaded eyewear). Dosimetry badges (TLD or OSL) must be worn by all personnel who may receive ≥10% of the annual limit; for fluoroscopists, a collar badge (worn outside lead apron at thyroid level) estimates lens/thyroid dose, and a waist badge (under apron) estimates effective whole-body dose. Pregnant radiation workers should declare pregnancy; the fetal dose limit is 1 mSv for the entire pregnancy (5 mSv per NCRP, though 1 mSv is widely adopted).
03 Contrast Agents — Iodinated & Gadolinium
Iodinated Contrast Media
Iodinated contrast agents are used in CT, fluoroscopy, and conventional angiography. Iodine attenuates X-rays by the photoelectric effect (Z=53). Agents are classified by osmolality and ionicity:
| Class | Osmolality | Examples | Clinical Notes |
|---|---|---|---|
| High-osmolality contrast media (HOCM) | 1500-2000 mOsm/kg (5-8x plasma) | Diatrizoate (Gastrografin) | Ionic; higher reaction rate; rarely used IV; used orally/rectally for GI opacification |
| Low-osmolality contrast media (LOCM) | 500-850 mOsm/kg (2-3x plasma) | Iohexol (Omnipaque), Iopamidol (Isovue), Ioversol (Optiray) | Non-ionic; current standard for IV use; significantly lower reaction rate than HOCM |
| Iso-osmolality contrast media (IOCM) | 290 mOsm/kg (~plasma) | Iodixanol (Visipaque) | Non-ionic dimer; lowest osmolality; theoretical advantage in high-risk patients |
Contrast Reactions — Classification & Management
Acute reactions occur within 1 hour of injection and are classified by severity:
Mild: Urticaria (limited), pruritus, nasal congestion, sneezing, nausea, mild emesis, anxiety. Self-limited; observation only; no treatment required unless symptoms worsen. Moderate: Diffuse urticaria/erythema, facial edema, throat tightness (without dyspnea), mild bronchospasm/wheezing, mild hypotension with tachycardia. Requires treatment and close monitoring — epinephrine IM may be needed; IV fluids for hypotension. Severe: Laryngeal edema with stridor, significant bronchospasm with hypoxia, anaphylaxis (hypotension + tachycardia + urticaria), pulmonary edema, cardiac arrest, convulsions. Life-threatening — requires immediate epinephrine (0.3-0.5 mg of 1:1000 IM in the anterolateral thigh), airway management, IV fluids, vasopressors, and activation of emergency response.
Premedication Protocol for Prior Contrast Reaction
The standard ACR premedication regimen for patients with a prior moderate or severe contrast reaction who require repeat iodinated contrast:
Prednisone 50 mg PO at 13 hours, 7 hours, and 1 hour before contrast injection, plus diphenhydramine 50 mg PO/IV/IM 1 hour before. An accelerated regimen for urgent cases: methylprednisolone 40 mg IV or hydrocortisone 200 mg IV at 5 hours and 1 hour before, plus diphenhydramine 50 mg IV 1 hour before. Premedication reduces but does not eliminate the risk of breakthrough reactions (recurrence rate ~10%). The most important consideration is whether contrast is truly needed — use an alternative modality (US, MRI without gadolinium, non-contrast CT) when feasible.
Contrast-Induced Nephropathy (CIN) / Post-Contrast Acute Kidney Injury (PC-AKI)
Defined as an increase in serum creatinine of ≥0.3 mg/dL or ≥50% above baseline within 48-72 hours after contrast administration. Risk factors include pre-existing renal impairment (eGFR <30 mL/min is highest risk), diabetes mellitus, heart failure, dehydration, concurrent nephrotoxins (NSAIDs, aminoglycosides), and large contrast volumes. The eGFR threshold for IV iodinated contrast: the ACR Manual on Contrast Media states that for IV contrast, withholding contrast or taking precautions is generally recommended when eGFR <30 mL/min. For eGFR 30-44, risk is low but hydration is recommended. For eGFR ≥45, no special precautions are needed. Prevention: IV normal saline at 1-1.5 mL/kg/hour for 6-12 hours before and after; minimize contrast volume; avoid repeated contrast within 48 hours. Bicarbonate infusion and N-acetylcysteine have not shown consistent benefit in modern trials.
Gadolinium-Based Contrast Agents (GBCAs)
Gadolinium (Gd3+) is paramagnetic and shortens T1 relaxation, causing signal enhancement on T1-weighted MRI sequences. Free gadolinium is highly toxic, so it is chelated to organic ligands:
| Group | Stability | Examples | NSF Risk |
|---|---|---|---|
| Group I (linear, non-ionic) | Least stable; highest free Gd release | Gadodiamide (Omniscan), Gadoversetamide (OptiMARK) | Highest risk — contraindicated in eGFR <30 |
| Group II (linear, ionic) | Intermediate stability | Gadopentetate dimeglumine (Magnevist), Gadobenate dimeglumine (MultiHance) | Intermediate risk — caution in eGFR <30 |
| Group III (macrocyclic) | Most stable; cage structure traps Gd | Gadoterate meglumine (Dotarem), Gadobutrol (Gadavist), Gadoteridol (ProHance) | Lowest risk — can be used with caution even in eGFR <30 |
Nephrogenic Systemic Fibrosis (NSF)
NSF is a potentially fatal fibrosing condition affecting the skin, joints, and internal organs in patients with severe renal impairment (eGFR <30) or acute kidney injury exposed to gadolinium. Onset: days to months after exposure. Presentation: symmetric skin thickening and hardening (often starting on the extremities), joint contractures, fibrosis of internal organs (lungs, liver, heart). No effective treatment exists once established. Since the introduction of Group III macrocyclic agents and screening protocols (eGFR measurement before GBCA administration), no unconfounded cases of NSF have been reported with Group III agents. Current ACR guideline: screen for renal dysfunction before GBCA; if eGFR <30, use Group III agents only if MRI with contrast is essential; consider alternative imaging.
Even with normal renal function, gadolinium deposits have been found in the brain (dentate nucleus, globus pallidus), bone, and skin after repeated GBCA exposure. Group I linear agents cause more deposition than Group III macrocyclic agents. Clinical significance remains uncertain — no definitive neurological effects have been proven. The FDA recommends considering GBCA retention when choosing agents, limiting GBCA use to situations where essential diagnostic information cannot be obtained otherwise, and using macrocyclic agents when possible.
04 Key Terminology & Abbreviations
Radiographic Descriptors
Radiolucent: allows X-rays to pass through, appearing dark/black on radiographs (air, fat). Radiopaque: absorbs X-rays, appearing white/bright (bone, metal, barium). Opacity: any white/bright area on radiograph — a general term avoiding presumption of etiology. Lucency: any dark area on radiograph. Consolidation: opacification of lung parenchyma with obscuration of underlying vessels and bronchi, implying airspace filling. Ground-glass opacity (GGO): hazy increased attenuation in lung that does not obscure underlying vessels — implies partial airspace filling or interstitial thickening. Atelectasis: volume loss — identified by shift of fissures, mediastinum, or hemidiaphragm toward the opacified region.
CT Density Descriptors
Hypodense / hypodensity: lower density (darker) than reference tissue. Hyperdense / hyperdensity: higher density (brighter) than reference tissue. Isodense: same density as reference. Enhancement: increase in density after IV contrast — implies vascularity. Rim enhancement: peripheral enhancement with central hypoattenuation — classic for abscess. Washout: decrease in enhancement on delayed phase — seen in HCC and adrenal adenomas.
MRI Signal Descriptors
Hyperintense: bright signal (high signal intensity). Hypointense: dark signal (low signal intensity). Isointense: same signal as reference tissue. Signal characteristics depend entirely on the sequence — always specify "T1 hyperintense" or "T2 hypointense" rather than just "bright" or "dark."
Ultrasound Descriptors
Anechoic: no internal echoes, appearing black — simple fluid (cyst, bile, urine). Hypoechoic: fewer echoes, darker than reference tissue. Hyperechoic: more echoes, brighter than reference. Isoechoic: same echogenicity as reference. Echogenic: producing echoes (a general descriptor). Posterior acoustic shadowing: signal void deep to a highly reflective/absorptive structure (gallstone, calcification). Posterior acoustic enhancement: increased echogenicity deep to a fluid-filled structure (cyst, gallbladder).
Fluoroscopy Terminology
Real-time imaging: continuous X-ray beam producing live video. Spot image: static radiograph captured during fluoroscopy for documentation. Last-image-hold: retains the last fluoroscopic frame, reducing dose. Collimation: narrowing the X-ray beam to the region of interest — reduces scatter and patient dose. Pulsed fluoroscopy: X-ray beam pulsed at defined rates (e.g., 7.5, 15 pulses/second) rather than continuous — can reduce dose by 50-75% with minimal impact on image quality for most applications.
Imaging Modality Selection Principles
The ACR Appropriateness Criteria provide evidence-based guidelines for imaging selection. General principles: use the least invasive, lowest-radiation modality that answers the clinical question. Ultrasound first for: right upper quadrant pain (gallstones), pregnancy, pediatric appendicitis, thyroid nodules, testicular/scrotal pathology, DVT, breast masses (as adjunct). CT first for: trauma, acute abdomen in adults, PE, stroke, renal colic. MRI first for: brain (non-emergent), spine, joints/soft tissue, liver lesion characterization, cardiac assessment, pelvic pathology. Radiograph first for: fractures, chest evaluation, bowel obstruction (followed by CT if positive), foreign body.
05 Chest X-Ray — Systematic Approach
Projections & Technical Adequacy
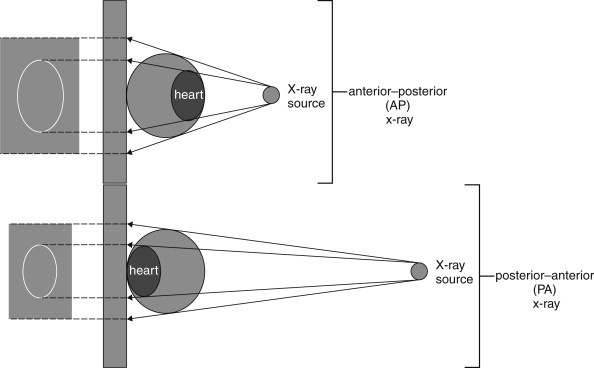
The standard chest radiograph is a PA (posteroanterior) upright projection taken at 72 inches (6 feet) source-to-image distance, with the anterior chest against the detector. The AP (anteroposterior) projection (portable/bedside) positions the detector behind the patient; it magnifies anterior structures (heart, mediastinum) and should not be used for cardiothoracic ratio assessment. The lateral projection (left side against detector by convention) allows visualization of retrosternal and retrocardiac space, posterior costophrenic angles, and spine.
Rotation: Medial clavicular heads should be equidistant from spinous processes; rotation causes spurious mediastinal widening and asymmetric lung density. Inspiration: Adequate when the right hemidiaphragm is at or below the 10th posterior rib (or 6th anterior rib); underinspiration makes the heart appear enlarged and the lung bases appear opacified. Penetration: Thoracic vertebral bodies should be faintly visible through the cardiac silhouette on PA view; overpenetration causes loss of vascular markings, underpenetration obscures mediastinal structures. Artifacts: Check for patient rotation, overlying leads/lines/gown snaps, motion blur.
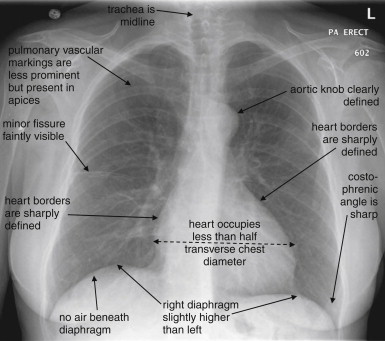
Systematic Interpretation — ABCDE Method
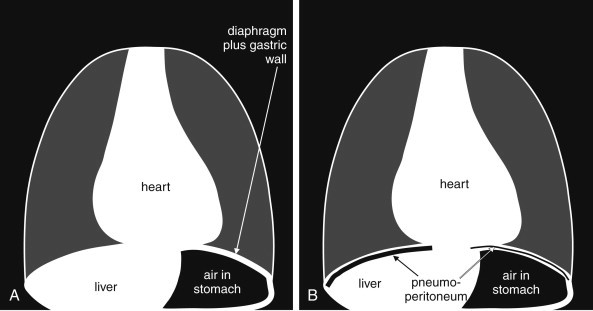
A — Airway: Trachea midline or slightly right of midline; carina at T5-T7; mainstem bronchi. B — Bones: Ribs (count and check for fractures, lytic/blastic lesions), clavicles, scapulae, spine, sternum (lateral view). C — Cardiac: Cardiothoracic ratio <0.5 on PA (greater suggests cardiomegaly); cardiac contour — right border = RA, left border = LV/LA appendage; aortic knob. D — Diaphragm: Right usually 1-2 cm higher than left (liver); costophrenic angles sharp (blunting suggests ≥200 mL effusion); free air under diaphragm (pneumoperitoneum). E — Everything else (soft tissues, edges, extras): Subcutaneous emphysema, breast shadows, tubes/lines/devices, lung apices (Pancoast tumor), behind the heart (retrocardiac opacity on lateral).
Tubes & Lines — Correct Positioning
| Device | Ideal Position | Malposition Risks |
|---|---|---|
| Endotracheal tube (ETT) | Tip 3-5 cm above carina (T2-T4 with head neutral) | Too low: right mainstem intubation (left lung atelectasis). Too high: above cords (ineffective, aspiration) |
| Central venous catheter (CVC) | Tip at cavoatrial junction (junction of SVC and RA) | Too deep: arrhythmia, cardiac perforation. Too shallow: unreliable CVP. Arterial placement: pulsatile bright-red blood |
| Swan-Ganz (PA catheter) | Tip in right or left main pulmonary artery, no further than 2 cm from hilum | Too peripheral: pulmonary infarction, PA rupture. Coiled in RV: arrhythmia |
| Nasogastric tube (NGT) | Tip below diaphragm, in the stomach (not coiled in esophagus or in the lung) | Bronchial placement: pneumothorax on feeding. Esophageal: aspiration, ineffective decompression |
| Chest tube | Anterior/apical for pneumothorax; posterior/basilar for effusion. All holes inside pleural space | Intraparenchymal or subfissural: ineffective drainage, lung laceration |
| Intra-aortic balloon pump (IABP) | Tip at aortic knob (just distal to left subclavian artery origin) | Too proximal: occlusion of carotid/subclavian. Too distal: renal/mesenteric ischemia |
06 Chest X-Ray Pathology
Pneumonia Patterns
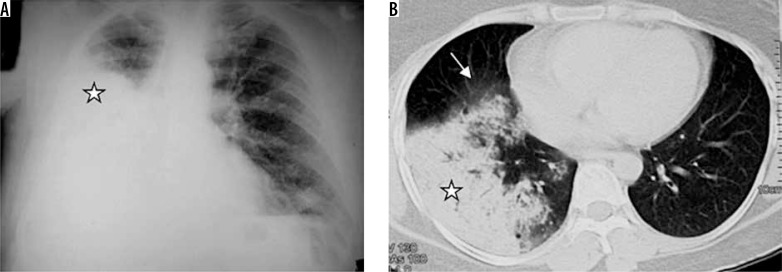
Lobar pneumonia: homogeneous consolidation confined to one lobe with air bronchograms (air-filled bronchi visible within opacified lung). Classic organism: Streptococcus pneumoniae. The consolidation respects fissural boundaries. Bronchopneumonia (lobular): patchy, multifocal, bilateral opacities centered on airways — often in dependent regions. Organisms: Staphylococcus aureus, gram-negatives, aspiration. Interstitial pneumonia: diffuse reticular or reticulonodular pattern, peribronchial cuffing, without air bronchograms. Organisms: viral (influenza, RSV, COVID-19 early), Mycoplasma, Pneumocystis jirovecii. Atypical patterns: round pneumonia (children, mimics mass), cavitary pneumonia (Klebsiella, TB, anaerobes, Staphylococcus), miliary pattern (hematogenous spread — TB, fungal).
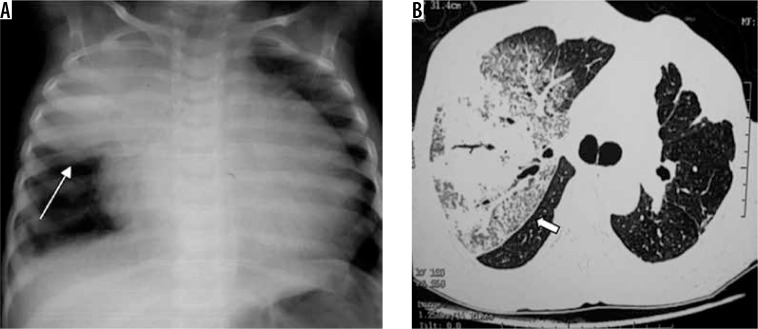
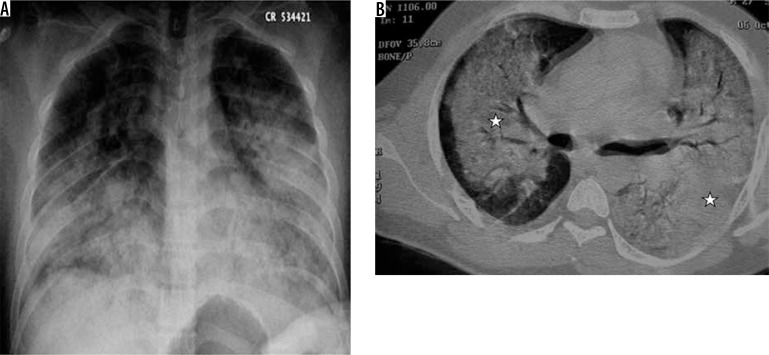
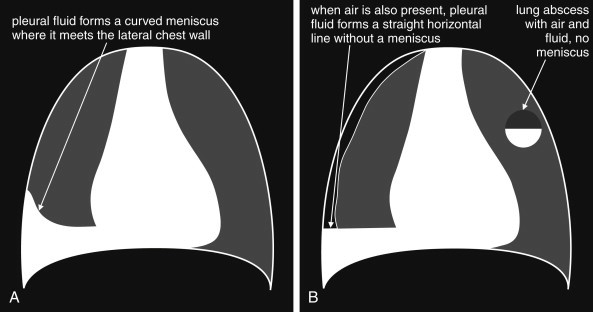
Pleural Effusion
Free-flowing fluid layers dependently. On upright PA, as little as 200-300 mL produces blunting of the costophrenic angle — the meniscus sign (concave upward opacity). A lateral decubitus film is the most sensitive plain radiograph for detecting small effusions (≥50 mL). Subpulmonic effusion mimics an elevated hemidiaphragm — clues include lateralization of the diaphragmatic peak and blunting of the lateral costophrenic angle. A loculated effusion does not layer on decubitus view; it may appear as a lens-shaped opacity against the chest wall (D-sign on US). Massive effusion causes complete opacification of the hemithorax with contralateral mediastinal shift — if the mediastinum shifts toward the opacified side, suspect associated atelectasis (e.g., central obstructing tumor).
Pneumothorax
The hallmark is a visible visceral pleural line separated from the chest wall by a lucent space devoid of lung markings. On upright films, air rises to the apex. On supine films (ICU patients), air collects anteriorly and basally — the deep sulcus sign is the key finding: an abnormally deep, lucent costophrenic angle extending inferiorly. Additional supine signs include a sharp cardiac border, lucency over the upper abdomen, and a visible anterior junction line. Tension pneumothorax is a clinical diagnosis — radiographic signs include mediastinal shift away from the affected side, flattening/inversion of the ipsilateral hemidiaphragm, and cardiovascular collapse.
Pulmonary Edema
Cardiogenic edema follows a predictable progression based on pulmonary capillary wedge pressure (PCWP): Stage 1 (PCWP 12-18 mmHg): cephalization (redistribution) — upper lobe pulmonary veins become equal to or larger than lower lobe veins (normally 2:1 lower:upper ratio). Stage 2 (PCWP 18-25 mmHg): interstitial edema — Kerley B lines (1-2 cm horizontal lines at lung periphery, perpendicular to pleural surface, representing thickened interlobular septa), peribronchial cuffing, septal lines. Stage 3 (PCWP >25 mmHg): alveolar edema — bilateral perihilar consolidation in a bat-wing (butterfly) distribution, air bronchograms, pleural effusions (bilateral, often right greater than left). Cardiomegaly is typically present. Non-cardiogenic edema (ARDS): bilateral opacities without cephalization, without cardiomegaly, with peripheral and dependent distribution.
The Silhouette Sign
The silhouette sign occurs when a density adjacent to a structure of similar density obliterates the visible border of that structure. It localizes pathology by identifying which border is lost:
| Lost Border | Anatomic Correlate | Pathology Location |
|---|---|---|
| Right heart border | Right middle lobe | RML pneumonia/atelectasis |
| Left heart border | Lingula | Lingular pneumonia/atelectasis |
| Right hemidiaphragm | Right lower lobe | RLL consolidation/effusion |
| Left hemidiaphragm | Left lower lobe | LLL consolidation/effusion |
| Aortic knob | Left upper lobe (apicoposterior) | LUL mass/consolidation |
| Ascending aorta | Right upper mediastinum/RUL | Anterior mediastinal mass, RUL pathology |
Mediastinal Widening
Mediastinal width >8 cm on PA upright CXR is considered widened (note: AP portable films routinely magnify the mediastinum). Differential diagnosis: traumatic aortic injury (widened mediastinum + loss of aortic knob contour + left apical cap + left pleural effusion + depression of left mainstem bronchus), aortic dissection, aortic aneurysm, lymphadenopathy (lymphoma, sarcoidosis, metastatic disease), mediastinal mass (thymoma, goiter), mediastinal hemorrhage (post-procedural, coagulopathy), and technical factors (AP projection, poor inspiration, supine positioning).
07 Chest CT — Protocols & Interpretation
CT Chest Protocols
Non-contrast CT chest: screening for lung nodules (low-dose CT — LDCT), evaluation of interstitial lung disease, assessment of emphysema severity, characterization of calcified structures. CT chest with IV contrast (standard): soft tissue enhancement for mass characterization, lymphadenopathy, pleural disease, mediastinal evaluation. CT pulmonary angiography (CTPA): bolus-tracked contrast timed to peak pulmonary artery opacification (typically 15-20 second delay); the primary study for suspected PE. CT aortography (CTA): timed for aortic opacification (20-25 second delay); used for aortic dissection, aneurysm, traumatic aortic injury. High-resolution CT (HRCT): thin-section (1-1.25 mm) imaging with sharp reconstruction kernel for interstitial lung disease evaluation — may be inspiratory and expiratory (air trapping).
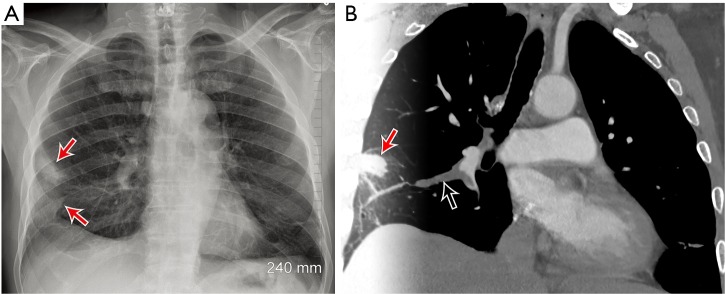
Pulmonary Embolism on CTPA
The diagnostic finding is an intraluminal filling defect within a contrast-opacified pulmonary artery — the clot appears as a dark area within a bright vessel. A saddle embolus straddles the main pulmonary artery bifurcation. Signs of right heart strain indicating massive/submassive PE: RV/LV diameter ratio >1.0 (measured on axial images at widest dimension), interventricular septal bowing toward the LV, reflux of contrast into the IVC and hepatic veins, and straightening or leftward deviation of the interventricular septum. Chronic PE: eccentric mural thrombus, calcified thrombus, webs/bands, mosaic attenuation pattern, enlarged bronchial arteries.
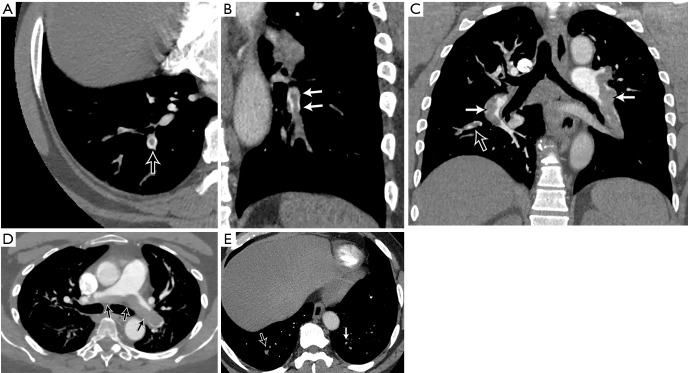
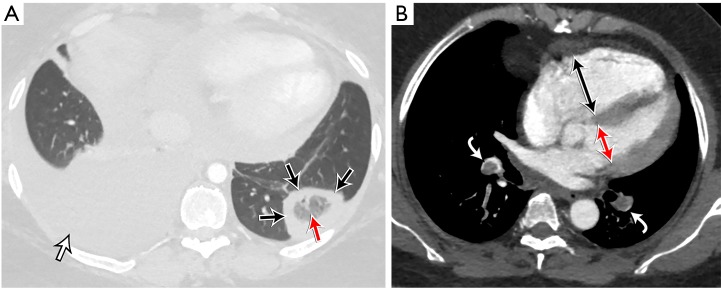
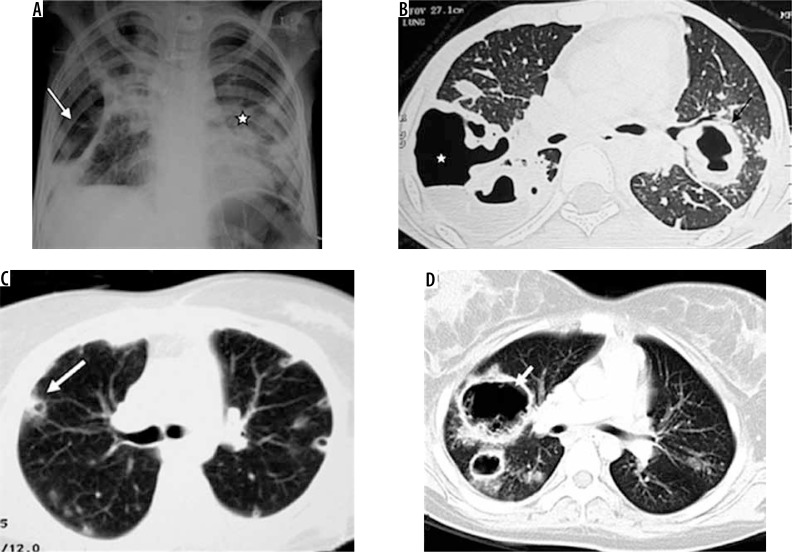
Fleischner Society Guidelines for Incidental Pulmonary Nodules (2017)
These guidelines apply to incidentally detected solid and subsolid nodules in adults ≥35 years without known cancer or immunosuppression:
<6 mm: No routine follow-up. 6-8 mm: CT at 6-12 months, then consider CT at 18-24 months. >8 mm: CT at 3 months, PET/CT, or tissue sampling.
<6 mm: Optional CT at 12 months. 6-8 mm: CT at 6-12 months, then CT at 18-24 months. >8 mm: CT at 3 months, PET/CT, or tissue sampling.
Solitary ground-glass <6 mm: No routine follow-up. Solitary ground-glass ≥6 mm: CT at 6-12 months to confirm persistence, then CT every 2 years until 5 years. Solitary part-solid: CT at 3-6 months; if persistent with solid component <6 mm, annual CT for 5 years; if solid component ≥6 mm, consider PET/CT or biopsy. Multiple subsolid: CT at 3-6 months; if stable, consider CT at 2 and 4 years. Management based on the most suspicious nodule.
Lung-RADS (Lung CT Screening Reporting & Data System)
| Category | Definition | Risk of Malignancy | Management |
|---|---|---|---|
| 1 — Negative | No nodules; definitely benign (complete calcification, fat) | <1% | Continue annual screening |
| 2 — Benign Appearance | Solid nodule <6 mm; new <4 mm; perifissural nodule <10 mm | <1% | Continue annual screening |
| 3 — Probably Benign | Solid nodule ≥6-<8 mm; new 4-<6 mm; part-solid <6 mm solid; non-solid ≥30 mm or new | 1-2% | 6-month LDCT |
| 4A — Suspicious | Solid ≥8-<15 mm; new 6-<8 mm; part-solid ≥6 mm solid; endobronchial nodule | 5-15% | 3-month LDCT; PET/CT may be used |
| 4B — Very Suspicious | Solid ≥15 mm; new ≥8 mm; growing solid nodule | >15% | Chest CT with/without contrast, PET/CT, tissue sampling |
| 4X | Category 3 or 4 with additional features raising suspicion (spiculation, upper lobe, lymphadenopathy) | Variable, ≥4A | As per highest applicable 4A/4B category |
Lung Parenchymal Patterns on CT
Ground-glass opacity (GGO): hazy increased attenuation that does not obscure underlying bronchi or vessels. Differential: infection (viral, PJP, early COVID-19), hemorrhage, edema, organizing pneumonia, adenocarcinoma in situ. Consolidation: dense opacification obscuring vessels and bronchi; air bronchograms often present. Differential: pneumonia, organizing pneumonia, lymphoma, alveolar proteinosis. Tree-in-bud: branching centrilobular nodular opacities resembling a budding tree; represents mucus/pus/fluid-filled bronchioles. Highly suggestive of endobronchial infection — TB, atypical mycobacteria, bacterial bronchiolitis, aspiration. Crazy paving: GGO with superimposed interlobular septal thickening, resembling irregular paving stones. Differential: pulmonary alveolar proteinosis (classic), ARDS, hemorrhage, PJP, lipoid pneumonia.
Interstitial Lung Disease Patterns on HRCT
Usual interstitial pneumonia (UIP): the radiologic pattern of IPF — basal-predominant, subpleural, heterogeneous reticulation with honeycombing and traction bronchiectasis. A definite UIP pattern on HRCT is diagnostic and precludes the need for surgical lung biopsy. Nonspecific interstitial pneumonia (NSIP): bilateral, symmetric, basal-predominant ground-glass opacity with fine reticulation; subpleural sparing is characteristic (helps distinguish from UIP). Organizing pneumonia (OP): peripheral, patchy consolidation, often migratory; may show the "reversed halo" (atoll) sign — a central GGO with a rim of consolidation. Lymphangitic carcinomatosis: smooth or nodular interlobular septal thickening, often asymmetric, with thickening of bronchovascular bundles; the pulmonary architecture is preserved (distinguishes from edema). Sarcoidosis: bilateral hilar lymphadenopathy (stage I), upper and mid-zone predominant perilymphatic nodules (along bronchovascular bundles, fissures, and subpleural surfaces), and fibrocystic change in advanced disease.
08 Thoracic MRI & Special Applications
Indications for Thoracic MRI
MRI of the chest is reserved for specific clinical scenarios where its superior soft tissue contrast and multiplanar capability provide information beyond CT: superior sulcus (Pancoast) tumors — assessment of brachial plexus invasion, subclavian vessel involvement, and chest wall extension; chest wall and mediastinal invasion by lung cancer or mesothelioma; posterior mediastinal masses (neurogenic tumors — schwannoma, neurofibroma) to assess intraspinal extension; cardiac and pericardial masses; and patients with iodinated contrast allergy or pregnancy where contrast-enhanced CT is not ideal.
Cardiac MRI (CMR)
Cardiac MRI is the reference standard for assessment of ventricular volumes, ejection fraction, and myocardial mass. Key indications include: myocardial viability (late gadolinium enhancement — LGE — identifies irreversible scarring; subendocardial or transmural enhancement in a coronary distribution = ischemic; mid-wall or epicardial enhancement = non-ischemic — myocarditis, sarcoidosis, HCM); arrhythmogenic right ventricular cardiomyopathy (fatty infiltration, wall motion abnormalities); cardiac masses (thrombus vs tumor); congenital heart disease (complex anatomy); pericardial disease (constrictive vs restrictive); and iron overload (T2* mapping in hemochromatosis/thalassemia).
Mediastinal Masses — The 4 T's of the Anterior Mediastinum
Mediastinal masses are classified by compartment. The anterior mediastinum (prevascular space) contains the "4 T's":
Thymoma: Most common anterior mediastinal mass in adults. Rounded, well-defined, may contain calcification. Associated with myasthenia gravis (30-50%), pure red cell aplasia, hypogammaglobulinemia. Masaoka staging determines resectability. Teratoma / Germ cell tumors: Mature teratomas contain fat, fluid, calcification, and/or teeth (pathognomonic on CT). Seminomas and non-seminomatous GCTs are solid, aggressive, and occur in young men. Terrible lymphoma: Both Hodgkin (most common mediastinal lymphoma in young adults) and non-Hodgkin. Presents as bulky anterior mediastinal lymphadenopathy. Hodgkin: lobulated contiguous mass; NHL: may encase vessels. Thyroid (retrosternal goiter): Continuity with the cervical thyroid on coronal/sagittal imaging. Often contains calcification, cystic change, and heterogeneous enhancement. May cause tracheal deviation and compression.
Middle mediastinum masses: lymphadenopathy (sarcoidosis — bilateral symmetric hilar + right paratracheal = "1-2-3 sign" or "lambda sign"), bronchogenic cyst (thin-walled, unilocular, near carina), pericardial cyst (right cardiophrenic angle, water density). Posterior mediastinum masses: neurogenic tumors (schwannoma, neurofibroma, ganglioneuroma — paravertebral location, may cause rib erosion or neural foraminal widening), lateral meningocele, extramedullary hematopoiesis, esophageal duplication cyst, descending aortic aneurysm.
Aortic Imaging
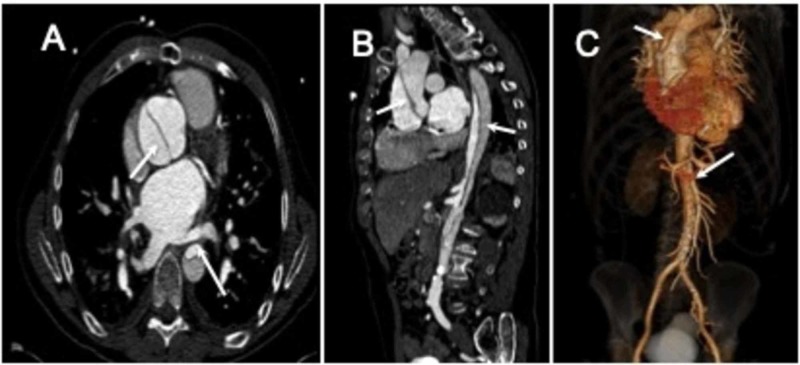
Aortic dissection: CTA is the primary diagnostic modality (sensitivity >95%). The hallmark finding is an intimal flap separating the true lumen (usually smaller, with denser contrast enhancement and faster flow) from the false lumen (usually larger, may contain thrombus). The Stanford classification divides dissection into Type A (involves the ascending aorta regardless of entry tear location — surgical emergency) and Type B (limited to the descending aorta distal to the left subclavian — medical management unless complicated). The DeBakey classification: Type I (ascending + descending), Type II (ascending only), Type III (descending only). Complications to evaluate on CTA: branch vessel involvement (renal, mesenteric, iliac — malperfusion syndrome), pericardial effusion (rupture into pericardium), aortic regurgitation, and mediastinal hematoma.
Aortic aneurysm: CT and CTA define the extent, maximum diameter, morphology (fusiform vs saccular), mural thrombus, and relationship to branch vessels. For abdominal aortic aneurysm (AAA): ≥3.0 cm is aneurysmal; surveillance intervals depend on size (3.0-3.9 cm: US every 3 years; 4.0-4.9 cm: US every 12 months; 5.0-5.4 cm: US/CT every 6 months). Surgical/endovascular repair threshold: ≥5.5 cm in men, ≥5.0 cm in women, or ≥0.5 cm growth in 6 months. The crescent sign (high-attenuation crescent within mural thrombus on non-contrast CT) suggests impending rupture.
09 Abdominal CT — Protocols
CT Abdomen/Pelvis — Contrast Phases
The timing of IV contrast injection relative to image acquisition is the central concept in abdominal CT protocol design. The CT abdomen/pelvis with IV contrast in the portal venous phase is the workhorse examination for most abdominal pathology.
| Phase | Delay (seconds post-injection) | What Enhances Best | Clinical Indications |
|---|---|---|---|
| Non-contrast | N/A (before contrast) | Nothing; baseline density | Renal stones, hemorrhage detection (hyperdense blood), adrenal adenoma washout calculation (baseline HU), calcium vs iodine differentiation |
| Arterial (late arterial) | 25-35 | Arteries, hypervascular tumors (HCC, RCC, NETs, carcinoid) | CT angiography (CTA), liver lesion characterization, active hemorrhage, trauma (active extravasation), pancreatic tumors |
| Portal venous | 60-70 | Liver parenchyma (peak), spleen, kidneys, mesenteric vessels, most solid organs | Standard phase for most abdominal indications — appendicitis, diverticulitis, abscess, bowel obstruction, cancer staging, organ assessment |
| Nephrographic | 80-100 | Renal parenchyma (homogeneous) | Renal mass detection and characterization — the entire kidney enhances uniformly, maximizing lesion-to-parenchyma contrast |
| Delayed / excretory | 5-15 minutes | Collecting system, ureters, bladder (opacified by excreted contrast) | CT urogram for hematuria workup, urothelial tumors, urinary leak, ureteral assessment |
Oral Contrast
Positive oral contrast (dilute barium or iodinated Gastrografin): opacifies the bowel lumen, making it bright white. Used primarily in CT for bowel obstruction evaluation, post-operative leak assessment, and differentiation of bowel from adjacent fluid collections. Neutral oral contrast (water, VoLumen): makes the bowel lumen dark (water density), allowing mucosal enhancement to be seen against the dark lumen. Preferred for CT enterography (Crohn disease), mesenteric CTA, and any study requiring assessment of bowel wall enhancement patterns. Many institutions now prefer water or no oral contrast for routine CT abdomen/pelvis, as IV contrast alone is sufficient for most indications and oral contrast slows workflow.
CT in Trauma — Solid Organ Injury Grading
The AAST (American Association for the Surgery of Trauma) organ injury scale guides management of solid organ injuries detected on CT. Key principles: Active extravasation (contrast blush — high-attenuation focus that increases from arterial to portal venous phase) indicates ongoing hemorrhage and may require angiographic embolization or surgery. Contained vascular injury (pseudoaneurysm, arteriovenous fistula) may be managed with observation or embolization.
| Organ | Key CT Findings by Grade | Management Trend |
|---|---|---|
| Spleen | Grade I: subcapsular hematoma <10% surface area. Grade II: parenchymal laceration 1-3 cm. Grade III: >3 cm laceration or subcapsular hematoma >50%. Grade IV: laceration involving segmental vessels. Grade V: shattered spleen or hilar vascular injury | Non-operative for Grades I-III if hemodynamically stable; angioembolization for contrast blush; splenectomy for Grade V or hemodynamic instability |
| Liver | Grade I: subcapsular hematoma <10%. Grade II: parenchymal laceration 1-3 cm. Grade III: laceration >3 cm. Grade IV: 25-75% of lobe destroyed. Grade V: >75% of lobe destroyed or juxtahepatic venous injury | Non-operative management successful in 80-90% with CT monitoring; angioembolization for blush; surgery for hemodynamic instability |
| Kidney | Grade I: contusion or subcapsular hematoma. Grade II: <1 cm laceration. Grade III: >1 cm laceration without collecting system injury. Grade IV: laceration into collecting system or segmental vessel injury. Grade V: shattered kidney or renal pedicle avulsion | Non-operative for I-III; Grade IV with urinoma may need stent/drain; Grade V often requires nephrectomy |
10 Hepatobiliary Imaging
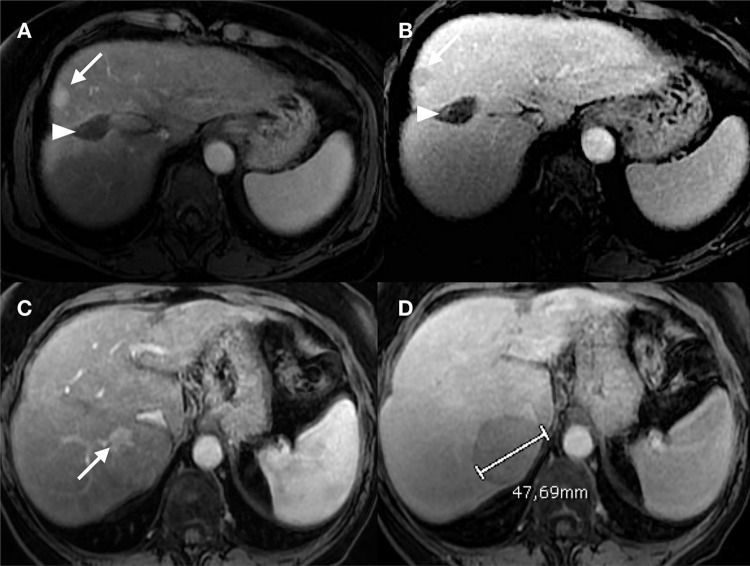
Liver Lesion Characterization — Enhancement Patterns
The key to liver lesion diagnosis is the enhancement pattern across multiple contrast phases:
| Lesion | Non-contrast | Arterial Phase | Portal Venous Phase | Delayed Phase | Key Features |
|---|---|---|---|---|---|
| Hemangioma | Hypodense | Peripheral, discontinuous nodular enhancement | Progressive centripetal fill-in | Persistent enhancement (isodense to blood pool) | Most common benign liver lesion; "light bulb bright" on T2 MRI |
| Focal Nodular Hyperplasia (FNH) | Isodense or slightly hypodense | Intense homogeneous enhancement (except central scar) | Isodense to liver | Central scar enhances on delayed phase (late scar enhancement on MRI) | Central scar with radiating septa; no malignant potential; no resection needed |
| Hepatocellular Carcinoma (HCC) | Variable | Intense arterial enhancement (APHE) | Washout (hypodense to liver) | Washout + capsule enhancement | Arterial phase hyperenhancement + portal venous washout = hallmark; occurs in cirrhotic livers |
| Hepatic adenoma | Iso/hypodense; may contain fat or hemorrhage | Moderate arterial enhancement | Iso/slightly hypodense | Iso/hypodense | Associated with OCP use; risk of hemorrhage and malignant transformation |
| Metastases (hypovascular) | Hypodense | Minimal enhancement (rim) | Hypodense (target/bull's-eye) | Variable | GI primaries (colon, stomach, pancreas); typically multiple |
| Metastases (hypervascular) | Hypodense | Intense arterial enhancement | Washout (may mimic HCC) | Hypodense | RCC, thyroid, carcinoid, melanoma, breast; must distinguish from HCC |
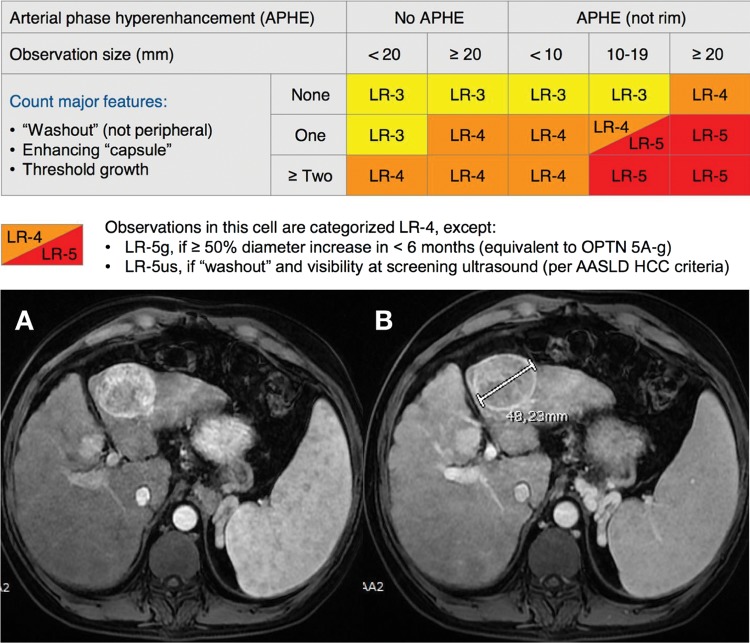
LI-RADS (Liver Imaging Reporting & Data System)
LI-RADS is used for liver observations in patients at risk for HCC (cirrhosis, chronic HBV, prior HCC). It applies to CT and MRI with extracellular contrast or hepatobiliary agents.
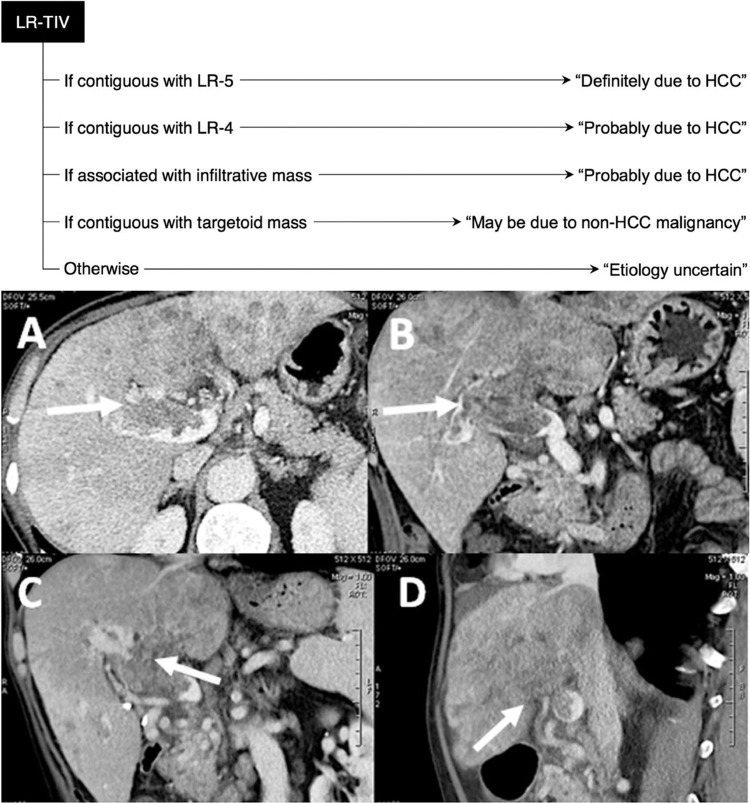
LR-NC (Non-categorizable): Image quality or coverage inadequate for categorization. LR-1 (Definitely Benign): Definite benign entity — cyst, hemangioma (with characteristic features), focal fat deposition/sparing, vascular anomaly. LR-2 (Probably Benign): Probably benign — distinctive nodule with benign features not meeting strict LR-1 criteria. LR-3 (Intermediate Probability): Moderate probability of malignancy — does not meet criteria for other categories; mass with arterial phase hyperenhancement (APHE) <20 mm without washout or capsule; mass without APHE 20+ mm with one or more ancillary features of HCC. LR-4 (Probably HCC): High probability of HCC — mass with APHE, <10 mm with washout or capsule; mass with APHE, 10-19 mm with washout but no capsule; non-APHE mass ≥20 mm with ≥2 additional major features. LR-5 (Definitely HCC): Definite HCC — mass with APHE, ≥10 mm with washout AND capsule; mass with APHE, ≥20 mm with washout OR capsule; allows treatment without biopsy per AASLD guidelines. LR-M (Probably or Definitely Malignant, Not Specific for HCC): Features suggest malignancy but not specific for HCC — targetoid appearance (rim APHE, peripheral washout, delayed central enhancement), infiltrative appearance without clear mass, marked diffusion restriction. Differential includes cholangiocarcinoma, combined HCC-CCA, metastases. Biopsy recommended. LR-TIV (Tumor in Vein): Definite tumor within a vein — enhancing soft tissue in portal vein, hepatic vein, or IVC contiguous with a parenchymal mass. Distinguished from bland thrombus by enhancement and expansion of the vein.
MRCP (Magnetic Resonance Cholangiopancreatography)
MRCP is a non-invasive, non-contrast technique using heavily T2-weighted sequences to visualize fluid-filled structures (bile ducts, pancreatic duct, gallbladder) as bright signal against a dark background. Indications: choledocholithiasis (sensitivity 85-100%), biliary stricture evaluation, primary sclerosing cholangitis (beaded appearance of intrahepatic and extrahepatic ducts), pancreatic duct assessment, congenital biliary anomalies (choledochal cyst, pancreas divisum). MRCP has largely replaced diagnostic ERCP, which is now reserved for therapeutic intervention.
HIDA Scan (Hepatobiliary Iminodiacetic Acid)
A nuclear medicine study using Tc-99m-labeled IDA derivatives taken up by hepatocytes and excreted into bile. Normal: radiotracer appears in the gallbladder, CBD, and duodenum within 60 minutes. Acute cholecystitis: non-visualization of the gallbladder at 60 minutes (and after morphine augmentation at 4 hours) indicates cystic duct obstruction — sensitivity 97%, specificity 90%. Biliary leak: tracer activity outside the biliary tree (post-cholecystectomy). Biliary atresia (neonatal): non-excretion of tracer into the bowel after 24 hours despite adequate hepatocyte uptake.
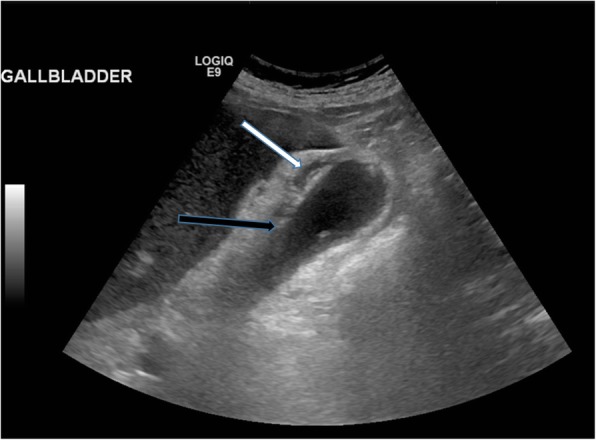
Gallbladder Ultrasound
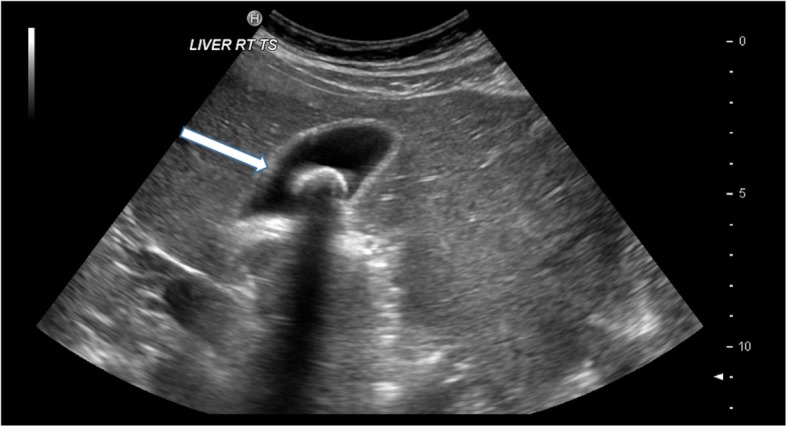
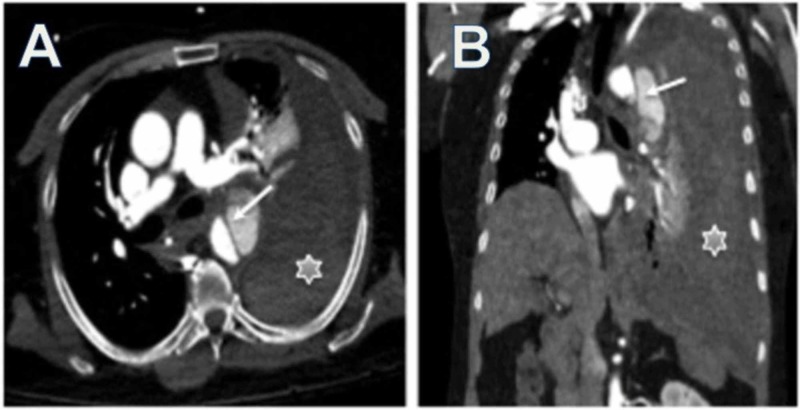
Ultrasound is the first-line study for right upper quadrant pain. Cholelithiasis: echogenic focus within the gallbladder with posterior acoustic shadowing and gravitational dependence (moves with patient repositioning). Acute cholecystitis — US findings (Murphy sign has highest specificity): sonographic Murphy sign (focal tenderness when the transducer is pressed over the gallbladder), gallstones, gallbladder wall thickening (>3 mm), pericholecystic fluid, gallbladder distension (>10 cm long or >5 cm transverse). Gangrenous cholecystitis: asymmetric wall thickening, intraluminal membranes, absence of Murphy sign (denervated wall), and striated wall enhancement on CT. Emphysematous cholecystitis: gas within the gallbladder wall (diabetic patients, Clostridium infection) — surgical emergency. Porcelain gallbladder: calcification of the gallbladder wall — associated with gallbladder carcinoma risk; prophylactic cholecystectomy is controversial (recent data suggest lower risk than historically reported).
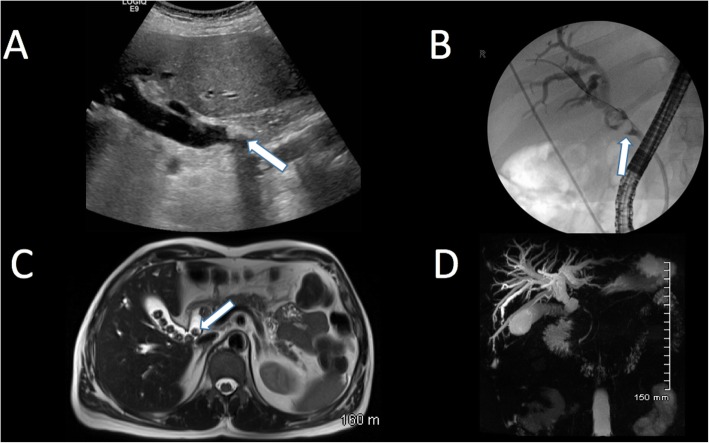
11 GI & Mesenteric Imaging
Pancreatitis Imaging
Acute pancreatitis: CT is not needed for diagnosis (clinical + lipase), but is indicated for assessing severity, complications, and alternative diagnoses. Initial CT is best performed at 72-96 hours after onset (earlier CT may underestimate necrosis). The CT Severity Index (CTSI / Balthazar score) grades pancreatitis from A (normal pancreas, 0 points) through E (two or more fluid collections or gas in/adjacent to pancreas, 4 points), with additional points for necrosis extent (0% = 0, <30% = 2, 30-50% = 4, >50% = 6). Total score ≥7 correlates with high morbidity/mortality. Key complications to identify: pancreatic necrosis (non-enhancing pancreatic parenchyma after IV contrast), peripancreatic fluid collections (acute: no wall, <4 weeks; walled-off necrosis: encapsulated, >4 weeks — may require drainage), pseudocyst (encapsulated fluid without solid debris, >4 weeks), hemorrhage (high-density fluid on non-contrast, pseudoaneurysm of splenic/gastroduodenal artery), and venous thrombosis (splenic vein thrombosis → left-sided portal hypertension → gastric varices).
Chronic pancreatitis: CT findings include pancreatic calcifications (pathognomonic), pancreatic duct dilation (>3 mm) and irregularity ("chain of lakes" appearance on MRCP), parenchymal atrophy, and pseudocyst. MRCP is the preferred modality for ductal assessment and detection of IPMN (intraductal papillary mucinous neoplasm). Pancreatic cancer: CT pancreas protocol (thin-section, pancreatic phase + portal venous) is the primary study. Ductal adenocarcinoma is hypovascular — appears hypodense (dark) relative to normally enhancing pancreatic parenchyma on the pancreatic (arterial) phase. Key staging findings: SMA/celiac trunk encasement (>180 degrees of circumferential involvement = unresectable), SMV/portal vein invasion, and metastatic disease (liver, peritoneum). The "double duct sign" (simultaneous dilation of both the common bile duct and pancreatic duct) on CT or MRCP suggests a pancreatic head mass compressing both ducts.
Acute Appendicitis — CT Criteria
CT is the primary imaging modality in adults with suspected appendicitis (sensitivity 98%, specificity 95%). Diagnostic criteria: dilated appendix >6 mm outer diameter (most specific finding), appendiceal wall thickening and enhancement, periappendiceal fat stranding (inflammatory change in surrounding fat), appendicolith (calcified fecalith within the appendix — present in ~25%), and periappendiceal fluid. Complications: phlegmon (inflammatory mass without drainable collection), abscess (rim-enhancing fluid collection), perforation (defect in wall, extraluminal air, fecalith outside lumen), and peritonitis (free fluid, diffuse mesenteric fat stranding).
Bowel Obstruction — SBO vs LBO
Small bowel obstruction (SBO): dilated small bowel (>3 cm) proximal to a transition point with decompressed bowel distally. The "small bowel feces sign" (particulate matter in dilated loops) occurs at or near the transition point. CT findings of strangulation (bowel ischemia requiring emergency surgery): bowel wall thickening, decreased/absent wall enhancement, mesenteric haziness, mesenteric fluid, pneumatosis intestinalis (air in bowel wall), portal venous gas. Closed-loop obstruction: a segment of bowel obstructed at two points (e.g., adhesive band or hernia), forming a C- or U-shaped loop; high risk of strangulation and perforation. Most common cause of SBO: adhesions (60-75%), then hernia (10-15%), then malignancy.
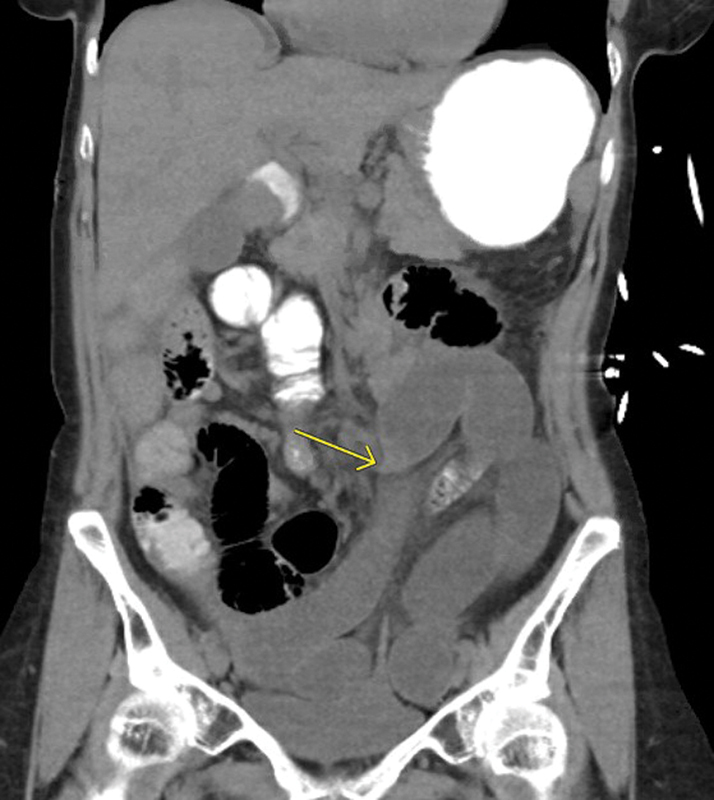
Large bowel obstruction (LBO): dilated colon (>6 cm, cecum >9 cm — risk of perforation if cecum >12 cm). Most common cause: colorectal carcinoma (60%), followed by volvulus (15%). The key distinction is LBO vs pseudo-obstruction (Ogilvie syndrome): in true LBO, there is a transition point with distal decompression; in pseudo-obstruction, the entire colon is dilated without a transition point. Sigmoid volvulus: "coffee bean sign" on plain film; "whirl sign" at the point of torsion on CT. Cecal volvulus: ectopic, dilated cecum in the left upper quadrant or midline with associated SBO.
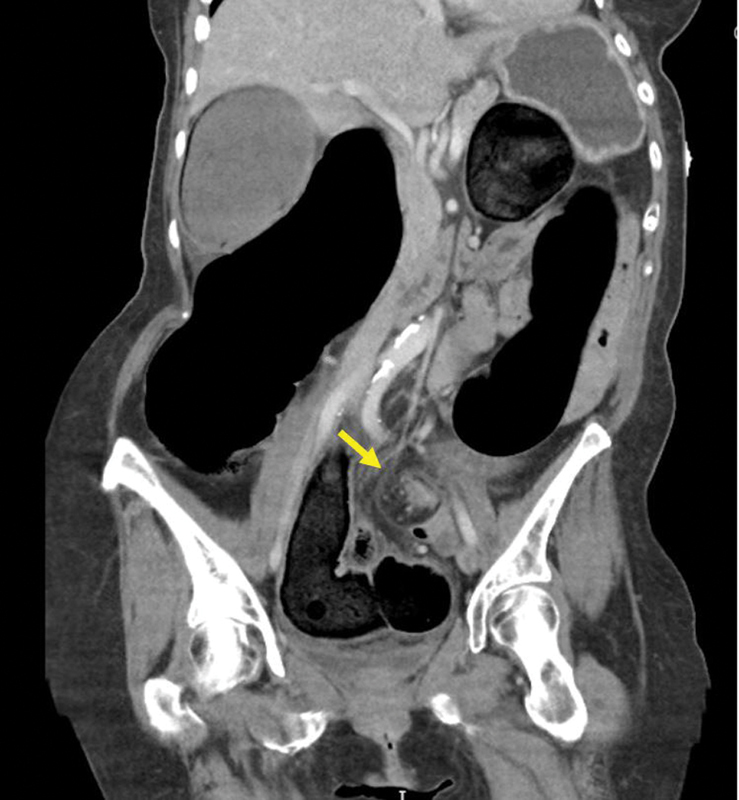
Epiploic Appendagitis & Omental Infarction
Epiploic appendagitis: a self-limiting cause of acute abdominal pain caused by torsion or venous thrombosis of an epiploic appendage (small fat-filled outpouching on the serosal surface of the colon). CT: oval fat-density lesion (<5 cm) with surrounding inflammatory stranding, often with a hyperdense ring ("hyperattenuating ring sign") adjacent to the anterior colon (sigmoid and cecum most common). Treatment: conservative (NSAIDs); no surgery needed. Omental infarction: spontaneous torsion or venous thrombosis of omental fat. CT: heterogeneous fat stranding in the greater omentum, typically right-sided, larger than epiploic appendagitis. Both entities are important to recognize on CT to avoid unnecessary surgery — they mimic appendicitis and diverticulitis.
Abdominal Aortic Pathology
Ruptured AAA: CT findings include retroperitoneal hematoma (hyperdense blood around the aorta, most commonly on the left), active contrast extravasation (bright blush that increases between arterial and delayed phases), the "draped aorta sign" (posterior aortic wall draping over the vertebral body, indicating contained rupture with loss of posterior adventitial integrity), and discontinuity of the aortic wall calcification ring. An unstable patient with known AAA goes directly to surgery — CTA is performed only if the patient is hemodynamically stable enough to be scanned. Aortoenteric fistula: a rare but often fatal complication of aortic graft surgery — CT shows gas around the graft, adjacent bowel wall thickening, and loss of the normal fat plane between the graft and duodenum. Active contrast extravasation into the bowel lumen is the definitive finding but is rarely captured on imaging.
Mesenteric Ischemia
CTA is the study of choice. Acute mesenteric ischemia: arterial occlusion (SMA embolus in 50% — look for filling defect in SMA, often at 3-8 cm from origin beyond the middle colic artery take-off), SMA thrombosis (at the SMA origin, often with pre-existing atherosclerotic disease), mesenteric venous thrombosis (SMV thrombosis with bowel wall thickening and mesenteric edema), or non-occlusive mesenteric ischemia (NOMI — diffuse arterial spasm in low-flow states). CT findings of bowel ischemia: bowel wall thickening, decreased enhancement, pneumatosis intestinalis, portal venous gas, mesenteric fat stranding, and free fluid. Pneumatosis + portal venous gas = advanced ischemia with high mortality.
Diverticulitis — Hinchey Classification
CT is the primary diagnostic study for acute diverticulitis. The Hinchey classification guides management:
| Stage | Description | CT Findings | Management |
|---|---|---|---|
| 0 | Mild clinical diverticulitis | Diverticula with pericolonic fat stranding, colonic wall thickening | Antibiotics, outpatient management |
| Ia | Confined pericolic inflammation or phlegmon | Pericolic fat stranding ± small pericolic phlegmon (<4 cm) | Antibiotics |
| Ib | Pericolic or mesocolic abscess | Pericolic or mesocolic abscess (≥4 cm) | Percutaneous drainage + antibiotics |
| II | Pelvic, distant intra-abdominal, or retroperitoneal abscess | Abscess distant from the sigmoid, often in pelvis | Percutaneous drainage + antibiotics |
| III | Generalized purulent peritonitis | Free intraperitoneal fluid without extraluminal air locules; no communication with bowel lumen | Emergency surgery (lavage or resection) |
| IV | Generalized fecal peritonitis | Extraluminal air, free fluid, fecal contamination | Emergency surgery (Hartmann procedure) |
12 Renal & Urologic Imaging
CT Urogram
The CT urogram is a multiphase study for evaluation of hematuria: non-contrast phase (stones, baseline density), nephrographic phase (renal mass detection), and excretory/delayed phase (urothelial lesions, collecting system filling defects). It has replaced IV pyelography (IVP) as the standard for hematuria workup. For renal colic, non-contrast CT (CT KUB) alone is the study of choice — sensitivity >95% for ureteral calculi. Signs of obstructing ureteral stone: hydroureter to the level of the stone, perinephric fat stranding, periureteral edema ("rim sign"), and unilateral nephromegaly.
Bosniak Classification of Renal Cysts (2019 Update)
The Bosniak system classifies cystic renal masses by imaging features to predict malignancy risk and guide management:
Bosniak I (Simple Cyst): Hairline-thin wall, no septa, no calcification, no solid component, water density (-9 to 20 HU), no enhancement. Malignancy risk: negligible. Management: benign, no follow-up. Bosniak II (Minimally Complex): Few hairline-thin septa, fine calcification in wall or septa, uniformly high-attenuation lesion ≤3 cm (hyperdense cyst), no enhancement. Malignancy risk: negligible. Management: benign, no follow-up. Bosniak IIF ("F" for Follow-up): Multiple hairline-thin septa, minimally thickened smooth wall or septa, thick or nodular calcification, high-attenuation lesion >3 cm, no enhancing soft tissue component. Malignancy risk: ~5-10%. Management: follow-up imaging at 6, 12, 24, and 48 months to assess for progression; if stable, no further follow-up needed. Bosniak III (Indeterminate): Thickened irregular wall or septa with measurable enhancement, without clearly solid enhancing components. Malignancy risk: ~50%. Management: surgery (partial nephrectomy) or active surveillance in select patients. Bosniak IV (Clearly Malignant): Distinct enhancing soft tissue component independent of wall or septa. Malignancy risk: >80%. Management: surgery (partial or radical nephrectomy).
Renal Mass Characterization
The first step in evaluating a renal mass is determining if it is cystic or solid. Simple cysts (Bosniak I) are extremely common and benign. A solid enhancing renal mass (>20 HU enhancement on post-contrast CT) is renal cell carcinoma (RCC) until proven otherwise. Key subtypes: clear cell RCC (70% — hypervascular, heterogeneous enhancement), papillary RCC (10-15% — hypovascular, homogeneous, slow enhancement), chromophobe RCC (5% — homogeneous, moderate enhancement). Angiomyolipoma (AML): benign tumor containing fat, smooth muscle, and blood vessels — macroscopic fat (<-10 HU on CT) is diagnostic and distinguishes AML from RCC without biopsy. Fat-poor AMLs lack macroscopic fat and may require MRI with chemical shift imaging or biopsy.
Ureteral Jets on Ultrasound
On color or pulsed-wave Doppler US of the bladder, ureteral jets are periodic bursts of urine entering the bladder from each ureteral orifice. The presence of bilateral symmetric jets excludes complete ureteral obstruction. Absent jets on the affected side supports the diagnosis of high-grade obstruction. This is a useful non-radiation technique for evaluating pregnant patients with suspected obstructing ureteral stone.
Adrenal Imaging
Incidental adrenal masses (adrenal incidentalomas) are found on approximately 4-5% of abdominal CT scans. The primary imaging question is whether the mass is a benign adenoma or a potentially malignant lesion (metastasis, adrenocortical carcinoma, pheochromocytoma). Adrenal adenoma: lipid-rich adenomas measure ≤10 HU on unenhanced CT (98% sensitivity for adenoma). For indeterminate nodules (11-30 HU), an adrenal washout study is performed: absolute washout = (enhanced HU - delayed HU) / (enhanced HU - unenhanced HU) x 100; ≥60% = adenoma. Relative washout (when no unenhanced phase is available) = (enhanced HU - delayed HU) / enhanced HU x 100; ≥40% = adenoma. On MRI, adenomas show signal drop on out-of-phase (opposed-phase) compared to in-phase images due to intracellular lipid (chemical shift imaging). Adrenal masses >4 cm, irregular margins, heterogeneous enhancement, or >10 HU without washout characteristics warrant further workup (biochemical testing, PET/CT, or biopsy).
13 Pelvic Imaging — Male & Female
Ovarian Mass — O-RADS (Ovarian-Adnexal Reporting & Data System)
O-RADS for MRI standardizes risk stratification of adnexal masses detected on ultrasound that require further characterization:
| O-RADS MRI Score | Description | Risk of Malignancy | Management |
|---|---|---|---|
| 1 | Normal ovaries, no adnexal lesion | Negligible | Routine care |
| 2 | Almost certainly benign (simple cyst, endometrioma, mature teratoma, typical fibroma) | <0.5% | Conservative management or follow-up as clinically indicated |
| 3 | Low risk (smooth wall/septa ≤3 mm enhancing, no solid tissue) | ~5% | Follow-up MRI in 3-6 months or referral to gynecology |
| 4 | Intermediate risk (smooth solid tissue enhancing, peritoneal disease absent) | ~50% | Referral to gynecologic oncology |
| 5 | High risk (irregular solid tissue enhancing, ± peritoneal disease/ascites) | >90% | Referral to gynecologic oncology for surgical management |
Ovarian Torsion
Ultrasound with Doppler is the primary imaging modality. Findings: enlarged ovary (often >5 cm, due to edema), peripheral follicles displaced by central edema ("string of pearls" sign), whirlpool sign (twisted vascular pedicle on Doppler), and free pelvic fluid. Absent or decreased arterial/venous Doppler flow is highly suggestive but not always present (intermittent torsion-detorsion, dual blood supply). Normal Doppler flow does NOT exclude torsion. An underlying ovarian mass (dermoid, cystadenoma) is the lead point in adults; in children/adolescents, the ovary may torse on an elongated fallopian tube without a mass. Surgical emergency requiring detorsion to salvage the ovary; viability correlates with duration of symptoms.
Ectopic Pregnancy — Ultrasound Findings
Transvaginal ultrasound is the primary imaging modality. Definitive diagnosis: extrauterine gestational sac with yolk sac or embryo (with or without cardiac activity). More commonly, indirect signs are present: adnexal mass separate from the ovary (tubal ring sign — echogenic ring surrounding an extrauterine sac), free pelvic fluid (hemorrhage), empty uterus with beta-hCG above the discriminatory level (1500-2000 mIU/mL for TVUS). The pseudogestational sac (decidual reaction within the endometrium) can mimic an intrauterine pregnancy — it is central in the cavity, lacks the "double decidual sign" of a true IUP, and lacks a yolk sac or embryo.
Uterine Fibroids
MRI is the most accurate modality for fibroid mapping before myomectomy or uterine artery embolization. Fibroids are T1 hypointense and T2 hypointense (dark on both sequences) relative to myometrium. Degenerating fibroids may have variable T1/T2 signal depending on the type of degeneration (hyaline, cystic, red/hemorrhagic, fatty). Location classification: submucosal (distorts endometrial cavity — highest impact on fertility), intramural, and subserosal. The FIGO classification system uses numbers 0-8 based on position relative to endometrium and serosa.
PI-RADS v2.1 (Prostate Imaging Reporting & Data System)
PI-RADS applies to multiparametric MRI (mpMRI) of the prostate for detection of clinically significant prostate cancer. Sequences required: T2-weighted, DWI/ADC map, and dynamic contrast-enhanced (DCE). Scoring differs by zone:
PI-RADS 1 (Very Low): Normal appearance on all sequences. Clinically significant cancer highly unlikely. No further workup. PI-RADS 2 (Low): Abnormality likely benign (e.g., BPH nodule, prostatitis). Clinically significant cancer unlikely. No biopsy. PI-RADS 3 (Intermediate): Equivocal — clinically significant cancer uncertain. In the peripheral zone: DWI is the dominant sequence; mild-moderate DWI abnormality without clear ADC restriction. In the transition zone: T2 is dominant; heterogeneous signal without definite focal lesion. Decision on biopsy depends on clinical factors (PSA density, family history). PI-RADS 4 (High): Clinically significant cancer likely. PZ: focal markedly hypointense ADC + hyperintense DWI, <1.5 cm. TZ: lenticular/non-circumscribed T2 hypointense lesion, <1.5 cm. MRI-targeted biopsy recommended. PI-RADS 5 (Very High): Clinically significant cancer highly likely. Same criteria as PI-RADS 4 but ≥1.5 cm, OR definite extraprostatic extension or invasion. MRI-targeted biopsy strongly recommended.
Testicular Ultrasound
Ultrasound is the first-line study for scrotal pathology. Testicular torsion: absent or decreased blood flow on color Doppler is the key finding; the testis may appear enlarged and hypoechoic (edema) or heterogeneous (infarction); the "whirlpool sign" (twisting of the spermatic cord) may be seen. Time-sensitive: >90% salvage if detorsion within 6 hours; <10% after 24 hours. Orchitis/epididymitis: enlarged, heterogeneous, hyperemic (increased Doppler flow) epididymis and/or testis. Testicular microlithiasis: ≥5 echogenic foci of 1-3 mm without acoustic shadowing per ultrasound field; associated with increased risk of testicular germ cell tumors — annual self-examination recommended; screening US is controversial. Testicular mass: any solid hypoechoic intratesticular mass is cancer until proven otherwise — further staging with CT chest/abdomen/pelvis and serum tumor markers (AFP, beta-hCG, LDH).
14 Brain CT & Emergencies
Acute Stroke — CT Findings
Non-contrast CT head is the initial study in acute stroke to exclude hemorrhage before thrombolysis. Early ischemic signs (within 6 hours): hyperdense MCA sign (thrombus in the MCA appearing bright white — 30-40% sensitivity), insular ribbon sign (loss of gray-white differentiation in the insular cortex), loss of basal ganglia definition (obscuration of lentiform nucleus), and cortical sulcal effacement. Frank hypodensity (established infarct) takes 12-24 hours to become obvious.
ASPECTS (Alberta Stroke Program Early CT Score)
A 10-point scoring system for MCA territory infarcts, where 1 point is subtracted for each region showing early ischemic change on non-contrast CT:
Basal ganglia level (4 regions): C = caudate head, L = lentiform nucleus, IC = internal capsule, I = insular ribbon. Supraganglionic level (6 regions): M1 = anterior MCA cortex (frontal operculum), M2 = MCA cortex lateral to insular ribbon, M3 = posterior MCA cortex (temporal lobe), M4 = anterior MCA territory above M1, M5 = lateral MCA territory above M2, M6 = posterior MCA territory above M3. Score 10 = normal; score 0 = complete MCA territory involvement. ASPECTS ≥6 generally favors thrombolysis/thrombectomy; ASPECTS <6 suggests large established infarct core with higher hemorrhagic transformation risk and poorer outcome.
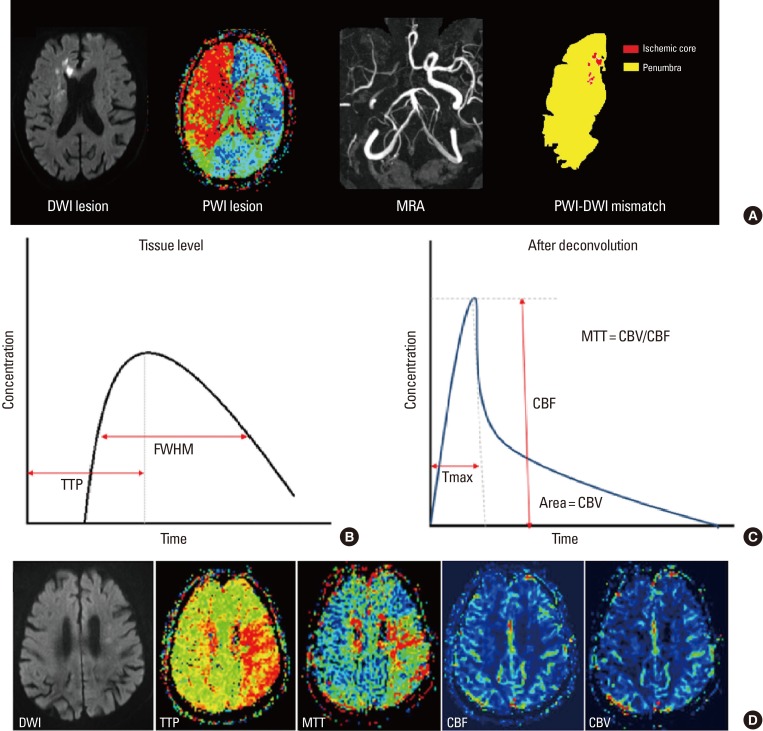
CT Perfusion (CTP)
CT perfusion maps the hemodynamic status of brain tissue using rapid sequential imaging during contrast bolus. Key parameters: CBF (cerebral blood flow): volume of blood flowing through a given brain volume per unit time. CBV (cerebral blood volume): total blood volume within a given brain volume. MTT (mean transit time): average time for blood to traverse the capillary bed. Tmax (time to maximum): delay in contrast arrival. The ischemic core has severely reduced CBF AND reduced CBV (irreversible injury). The penumbra (tissue at risk) has reduced CBF but preserved or elevated CBV (autoregulatory vasodilation maintains blood volume). A significant mismatch between core and penumbra (penumbra much larger than core) identifies patients who may benefit from thrombectomy even in extended time windows (6-24 hours per DAWN and DEFUSE 3 trials).
Intracranial Hemorrhage — CT Patterns
| Type | CT Appearance | Location / Shape | Etiology |
|---|---|---|---|
| Epidural hematoma | Hyperdense, biconvex (lens-shaped) | Between skull and dura; does not cross suture lines | Temporal bone fracture → middle meningeal artery rupture (85%); may have lucid interval |
| Subdural hematoma (acute) | Hyperdense, crescent-shaped | Between dura and arachnoid; crosses suture lines but not the midline falx | Tearing of bridging veins; trauma (elderly, anticoagulated); can be bilateral |
| Subdural hematoma (chronic) | Hypodense (isodense at 1-3 weeks — may be missed) | Same crescent shape | Prior trauma; elderly, alcoholics; can present with insidious confusion |
| Subarachnoid hemorrhage (SAH) | Hyperdense blood in sulci, cisterns, fissures | Basal cisterns (ruptured aneurysm), convexity (trauma) | Aneurysm rupture (80%); CTA to identify aneurysm; if CT negative, LP for xanthochromia |
| Intraparenchymal hemorrhage | Hyperdense mass within brain parenchyma | Basal ganglia, thalamus (hypertensive); lobar (amyloid angiopathy, tumor, AVM) | Hypertension (most common), amyloid angiopathy, coagulopathy, tumor, AVM |
| Intraventricular hemorrhage | Hyperdense blood layering in ventricles | Occipital horns (gravity-dependent) | Extension from intraparenchymal or SAH; risk of hydrocephalus |
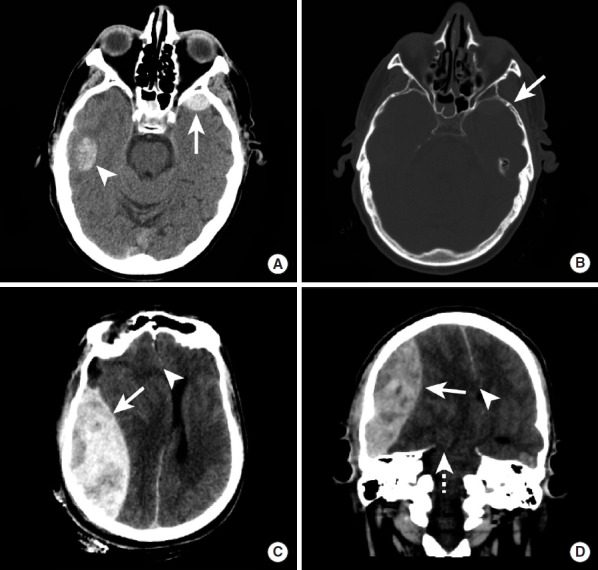
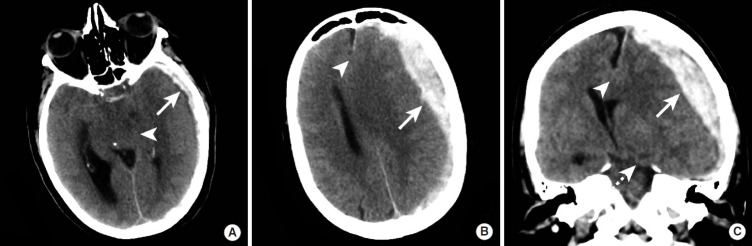
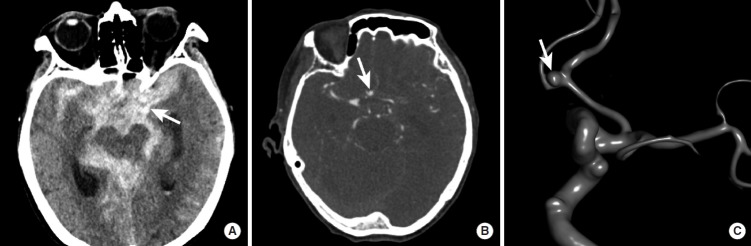
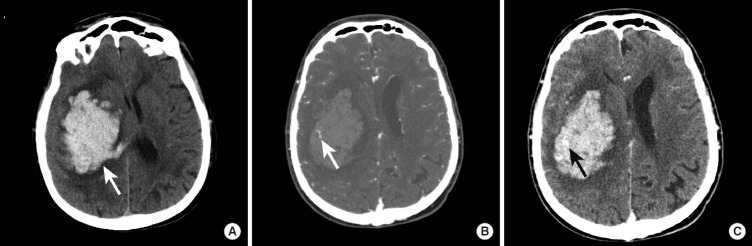
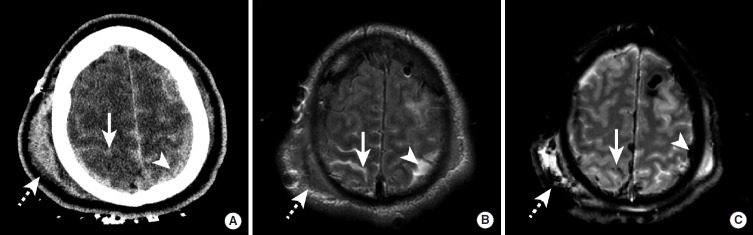
Herniation Syndromes
Subfalcine (cingulate): most common; cingulate gyrus herniates under the falx cerebri; can compress the ACA causing infarction. Transtentorial (uncal): uncus of temporal lobe herniates through the tentorial notch; compresses CN III (ipsilateral fixed dilated pupil) and PCA (occipital infarction); compresses the cerebral peduncle (contralateral hemiparesis, or ipsilateral hemiparesis if the opposite peduncle is compressed against the tentorial edge — Kernohan notch phenomenon). Central (downward): bilateral downward herniation through the tentorium; progressive rostral-to-caudal brainstem compression. Tonsillar: cerebellar tonsils herniate through the foramen magnum; compresses the medulla causing respiratory arrest — this is why lumbar puncture is contraindicated with posterior fossa mass lesion.
Hydrocephalus
Obstructive (non-communicating): obstruction within the ventricular system — tumor in the posterior fossa blocking the fourth ventricle, aqueductal stenosis blocking the cerebral aqueduct, colloid cyst at the foramen of Monro. CT/MRI shows dilation of ventricles proximal to the obstruction with normal-sized ventricles distally. Communicating hydrocephalus: impaired CSF absorption at the arachnoid granulations (post-meningitis, post-SAH) or overproduction (rare — choroid plexus papilloma). All ventricles are dilated. Normal pressure hydrocephalus (NPH): clinical triad of dementia, gait apraxia, and urinary incontinence ("wet, wacky, wobbly"). CT/MRI: ventriculomegaly out of proportion to sulcal prominence (distinguishes from ex-vacuo hydrocephalus of brain atrophy, where ventricles AND sulci enlarge proportionally). Evans index ≥0.3 and DESH (disproportionately enlarged subarachnoid space hydrocephalus) pattern on MRI support the diagnosis.
15 Brain MRI — Sequences & Pathology
MRI Sequences — What Each Shows
| Sequence | Key Principle | Bright Signal (Hyperintense) | Dark Signal (Hypointense) | Primary Use |
|---|---|---|---|---|
| T1-weighted | Short TR, short TE; reflects T1 relaxation | Fat, subacute blood (methemoglobin), gadolinium, melanin, proteinaceous fluid | Water/CSF, most pathology | Anatomy, post-contrast enhancement, fat-containing lesions |
| T2-weighted | Long TR, long TE; reflects T2 relaxation | Water/CSF, edema, most pathology | Cortical bone, fibrous tissue, air, deoxyhemoglobin, hemosiderin | Detecting pathology (edema, inflammation, tumors) |
| FLAIR (Fluid-Attenuated Inversion Recovery) | T2 with CSF signal suppressed | Edema, gliosis, demyelination, subarachnoid blood/infection | CSF (nulled), normal brain | Periventricular lesions (MS), cortical infarcts, SAH, meningitis |
| DWI (Diffusion-Weighted Imaging) | Detects restricted Brownian motion of water | Restricted diffusion (cytotoxic edema, abscess, highly cellular tumor) | Free water diffusion | Acute stroke (<30 min sensitivity), abscess, epidermoid, tumor cellularity |
| ADC (Apparent Diffusion Coefficient) map | Quantitative map derived from DWI; confirms true restriction | Free diffusion (vasogenic edema) | True restricted diffusion (acute infarct, abscess) | Distinguishing true restriction (DWI bright + ADC dark) from T2 shine-through (DWI bright + ADC bright) |
| SWI (Susceptibility-Weighted Imaging) | Exploits magnetic susceptibility differences | N/A (signal void = "blooming") | Blood products (hemosiderin, deoxyhemoglobin), calcium, iron | Microhemorrhages (amyloid angiopathy, DAI), cavernous malformations, calcification |
| MRA (MR Angiography) | TOF or contrast-enhanced; flow-sensitive | Flowing blood (appears bright) | Stationary tissue | Circle of Willis aneurysm screening, stenosis, dissection, AVM |
Acute Ischemic Stroke on MRI
DWI is the most sensitive and specific sequence for acute stroke, detecting cytotoxic edema within minutes of onset. The classic pattern: DWI bright + ADC dark = true restricted diffusion = acute infarct. The FLAIR sequence takes 4-6 hours to become positive — therefore, a DWI-FLAIR mismatch (DWI positive, FLAIR negative) suggests the stroke is <4.5 hours old and may be within the thrombolysis window even if the exact onset time is unknown (wake-up stroke — per the WAKE-UP trial).
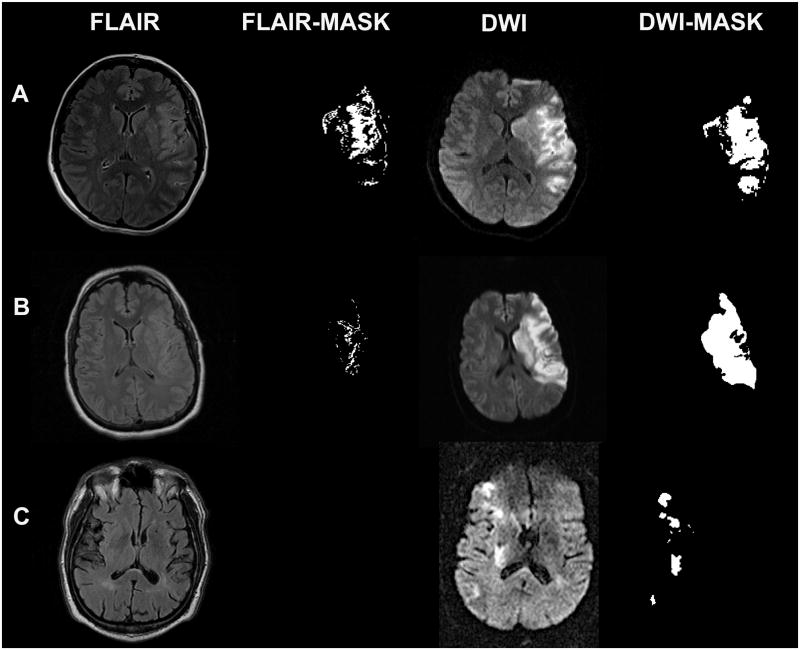
Multiple Sclerosis — MRI Findings
MRI is central to the diagnosis and monitoring of MS. The McDonald criteria (2017 revision) use MRI to demonstrate dissemination in space (DIS) and dissemination in time (DIT). DIS requires ≥1 T2 hyperintense lesion in ≥2 of 4 characteristic locations: periventricular (≥3 lesions, perpendicular to ventricles — Dawson fingers), cortical/juxtacortical, infratentorial (brainstem, cerebellum), and spinal cord. DIT requires simultaneous enhancing and non-enhancing lesions on a single MRI, OR a new T2/enhancing lesion on follow-up. Dawson fingers are the hallmark: perivenular demyelinating plaques radiating perpendicularly from the corpus callosum, best seen on sagittal FLAIR.
Brain Tumors — Enhancement Patterns
Ring enhancement: the differential includes GBM (glioblastoma multiforme — irregular thick ring, central necrosis, surrounding edema), metastasis (often at gray-white junction, multiple, variable ring thickness), and brain abscess (smooth thin ring with restricted diffusion centrally on DWI — a key distinguishing feature from necrotic tumor). Homogeneous enhancement: meningioma (dural-based, dural tail sign, extra-axial), lymphoma (periventricular, homogeneous, restricted diffusion, often in immunocompromised). Non-enhancing mass: low-grade glioma (T2/FLAIR hyperintense, no enhancement, may have calcification — oligodendroglioma).
MR Spectroscopy (MRS) — Basics
MRS measures the concentration of metabolites within a voxel of brain tissue. Key metabolites: NAA (N-acetylaspartate): neuronal marker — decreased in anything that destroys neurons (infarction, tumor, neurodegeneration). Choline (Cho): cell membrane turnover marker — elevated in tumors (high cellularity), demyelination. Creatine (Cr): energy metabolism — relatively stable; used as internal reference. Lactate: anaerobic metabolism — elevated in ischemia, abscess, high-grade tumors. Lipid: elevated in necrosis (high-grade tumors, abscess). High-grade tumors: elevated Cho/Cr ratio, decreased NAA, lactate/lipid peaks. Abscess: amino acid peaks (valine, leucine), lactate, acetate, succinate.
CNS Infections on MRI
Brain abscess: ring-enhancing lesion with smooth thin wall, surrounding vasogenic edema, and the critical distinguishing feature — restricted diffusion centrally (bright DWI, dark ADC), representing purulent material with high viscosity and cellularity. This differentiates abscess from necrotic tumor, which typically shows facilitated diffusion (dark DWI or bright ADC) centrally. Meningitis: leptomeningeal enhancement on post-contrast T1, FLAIR hyperintensity in the subarachnoid spaces, restricted diffusion in empyema. Encephalitis (HSV): T2/FLAIR hyperintensity with restricted diffusion involving the temporal lobes, insular cortex, and cingulate gyri bilaterally (often asymmetric); hemorrhagic transformation is common. Toxoplasmosis (in HIV/AIDS): ring-enhancing lesions in the basal ganglia, often multiple; eccentric "target sign" (eccentric nodule within the ring); responds to empiric treatment — if no response in 2 weeks, biopsy to exclude CNS lymphoma.
16 Spine Imaging
Disc Herniation — Levels & Nerve Roots
In the lumbar spine, disc herniations typically compress the traversing nerve root (the one exiting at the level below). A posterolateral (paracentral) L4-L5 disc herniation compresses the L5 nerve root. A far lateral (foraminal) L4-L5 disc herniation compresses the L4 nerve root (the exiting root at that level). In the cervical spine, C5-C6 disc herniation compresses the C6 nerve root (numbered by the lower vertebra in the cervical spine).
| Disc Level | Nerve Root (Posterolateral) | Motor Deficit | Sensory Distribution | Reflex |
|---|---|---|---|---|
| L3-L4 | L4 | Knee extension (quadriceps) | Medial leg / knee | Patellar (knee jerk) |
| L4-L5 | L5 | Great toe dorsiflexion (EHL), foot dorsiflexion | Lateral leg, dorsum of foot, great toe | None (or medial hamstring) |
| L5-S1 | S1 | Foot plantarflexion (gastrocnemius), foot eversion | Lateral foot, sole | Achilles (ankle jerk) |
| C5-C6 | C6 | Wrist extension, biceps | Lateral forearm, thumb, index finger | Biceps, brachioradialis |
| C6-C7 | C7 | Wrist flexion, finger extension, triceps | Middle finger | Triceps |
Cauda Equina Syndrome
A surgical emergency. MRI findings: large central disc herniation (usually L4-L5 or L5-S1) compressing multiple nerve roots of the cauda equina, or mass (tumor, abscess) within the spinal canal. The cauda equina nerve roots appear crowded, displaced, or effaced. Clinical correlation is essential: saddle anesthesia, urinary retention, bilateral lower extremity weakness, and loss of anal sphincter tone. MRI should be performed urgently (within hours) in suspected cases.
Disc Herniation Nomenclature
Standardized terminology per the combined task forces of the NASS, ASSR, and ASNR: Bulge: generalized extension (>180 degrees) of disc tissue beyond the vertebral body margin — a normal age-related finding, not a herniation. Herniation: localized displacement (<180 degrees) of disc tissue beyond the normal disc margin. Subtypes: protrusion (base of displaced material wider than its apex), extrusion (apex wider than base, or disc material extends above or below the disc space), and sequestration (free fragment — extruded material has lost continuity with the parent disc). Location: central, paracentral (most common — compresses traversing nerve root), foraminal (compresses exiting nerve root), and far lateral (extra-foraminal). On MRI, acute herniations are typically T2 hyperintense (hydrated), while chronic herniations may be T2 hypointense (desiccated).
Spinal Cord Compression
MRI with gadolinium is the study of choice. Etiologies: metastatic epidural disease (most common — prostate, breast, lung, kidney, myeloma; identified by vertebral body marrow signal abnormality with epidural soft tissue component), disc herniation, epidural abscess (rim-enhancing epidural collection with restricted diffusion), epidural hematoma, and primary tumors. The cord may show T2 signal abnormality (myelopathy) — a concerning finding indicating edema or gliosis. T2 hyperintensity with cord expansion suggests acute edema (potentially reversible); T1 hypointensity suggests chronic gliosis/myelomalacia (irreversible).
Spinal Infections & Inflammatory Disease
Discitis-osteomyelitis (spondylodiscitis): MRI with gadolinium is the study of choice. Classic findings: T1 hypointense and T2 hyperintense signal in the disc space AND adjacent vertebral endplates (two contiguous vertebral bodies + intervening disc), with enhancement of the disc space and endplates. The disc space is typically narrowed. Paraspinal/epidural abscess may be present as a rim-enhancing collection. In pyogenic infection, the disc is T2 hyperintense (edema/pus); in TB (Pott disease), the disc may be relatively preserved while multiple vertebral bodies are involved with large paraspinal abscesses (often calcified in chronic TB). CT-guided biopsy is often needed for organism identification.
Vertebral Fractures — TLICS (Thoracolumbar Injury Classification & Severity Score)
TLICS guides surgical vs non-operative management of thoracolumbar fractures based on three parameters:
Morphology: Compression fracture = 1, Burst fracture = 2, Translation/rotation = 3, Distraction = 4. Posterior ligamentous complex (PLC) integrity: Intact = 0, Indeterminate (MRI equivocal) = 2, Injured (disrupted — widened interspinous space, facet diastasis) = 3. Neurological status: Intact = 0, Nerve root injury = 2, Complete cord injury = 2, Incomplete cord injury = 3, Cauda equina = 3. Total score: ≤3 = non-operative management; 4 = indeterminate (consider either); ≥5 = surgical stabilization recommended.
17 Head & Neck Imaging
Vascular Imaging of the Head & Neck
CTA head and neck is the primary modality for evaluating the intracranial and extracranial vasculature in the acute setting. Indications: acute stroke (identify large vessel occlusion for thrombectomy), carotid stenosis (NASCET criteria: % stenosis = [1 - narrowest diameter / normal distal ICA diameter] x 100; ≥70% = symptomatic indication for endarterectomy), vertebral artery dissection (intimal flap, string sign, flame-shaped tapering), and intracranial aneurysm detection and characterization (location, size, morphology). MRA can be performed without contrast (TOF technique) or with gadolinium — used for screening (e.g., unruptured aneurysm surveillance in polycystic kidney disease or family history), carotid stenosis (may overestimate degree), and vertebrobasilar insufficiency.
Sinusitis
CT (non-contrast, coronal reformats) is the imaging standard for sinusitis when surgery is being considered or complications are suspected. Findings: mucosal thickening (>3 mm), air-fluid levels, complete opacification of a sinus. The ostiomeatal complex (OMC) is the key drainage pathway for the maxillary, anterior ethmoid, and frontal sinuses — obstruction of the OMC (by mucosal edema, polyps, or anatomic variants like concha bullosa) is the primary mechanism for recurrent sinusitis. Complications of sinusitis requiring urgent imaging: orbital cellulitis/abscess (subperiosteal abscess — rim-enhancing collection along the medial orbit from ethmoid sinusitis), cavernous sinus thrombosis (filling defect in cavernous sinus, bilateral orbital findings), intracranial abscess (frontal sinusitis → frontal epidural/subdural abscess — Pott puffy tumor involves frontal bone osteomyelitis with subperiosteal abscess).
Orbits
CT and MRI are complementary for orbital pathology. CT excels for bony detail (fractures, calcifications), while MRI provides superior soft tissue contrast. Orbital cellulitis vs preseptal cellulitis: the orbital septum divides the two; preseptal involves eyelid swelling with normal globe mobility (CT not always needed), while orbital (postseptal) cellulitis shows proptosis, restricted eye movement, and vision changes — CT with contrast reveals fat stranding posterior to the septum, subperiosteal abscess (most commonly medial, from ethmoid sinusitis), or intraconal abscess. Thyroid eye disease (Graves ophthalmopathy): CT/MRI shows enlargement of the extraocular muscles with tendon sparing (distinguishes from orbital myositis where tendons are involved). The inferior and medial recti are most commonly affected. Severe proptosis can cause optic nerve compression (compressive optic neuropathy) — orbital decompression may be needed. Optic neuritis: MRI with fat-suppressed post-contrast sequences shows enhancement and thickening of the optic nerve; associated with MS — brain MRI is indicated. Retinoblastoma: the most common intraocular malignancy of childhood; CT shows calcified intraocular mass; MRI defines extent and evaluates for trilateral disease (bilateral retinoblastoma + pinealoblastoma).
Temporal Bone — Cholesteatoma
A cholesteatoma is a keratinized squamous epithelial collection (not a true neoplasm) that erodes bone progressively. Acquired cholesteatoma (98%): typically arises from a retraction pocket of the pars flaccida (Prussak space) and extends into the epitympanum and mastoid. CT findings: soft tissue mass in the middle ear with adjacent bony erosion (scutum erosion is an early and specific finding), ossicular chain erosion (incus long process most commonly), and tegmen tympani erosion (risk of intracranial extension). MRI with DWI (non-EPI technique) is used to differentiate cholesteatoma (restricted diffusion — bright on DWI) from granulation tissue or cholesterol granuloma (no restriction), and to detect recurrence post-operatively.
Neck Masses — Nodal Stations
The neck is divided into 7 nodal levels for staging head and neck cancer:
| Level | Name / Location | Primary Drainage From |
|---|---|---|
| Ia | Submental — midline between anterior bellies of digastric | Floor of mouth, anterior tongue, lower lip |
| Ib | Submandibular — around submandibular gland | Oral cavity, submandibular gland, cheek |
| II (IIa/IIb) | Upper jugular — skull base to hyoid bone | Oral cavity, oropharynx, nasopharynx, parotid |
| III | Middle jugular — hyoid to cricoid | Oropharynx, hypopharynx, larynx |
| IV | Lower jugular — cricoid to clavicle | Hypopharynx, thyroid, cervical esophagus, larynx |
| V (Va/Vb) | Posterior triangle — behind sternocleidomastoid | Nasopharynx, scalp, skin (melanoma) |
| VI | Anterior compartment — between carotid sheaths, hyoid to manubrium | Thyroid, larynx, hypopharynx, cervical esophagus |
| VII | Superior mediastinal — below manubrium | Thyroid, esophagus |
Metastatic lymph nodes are characterized by size (>1 cm short axis, >1.5 cm for jugulodigastric), morphology (round rather than oval), central necrosis (the most specific CT finding for metastatic squamous cell carcinoma), extranodal extension (irregular margins, infiltration of surrounding fat), and heterogeneous enhancement. The Virchow node (left supraclavicular, level IV/V) is associated with abdominal malignancy (gastric, pancreatic). PET/CT is used for staging, treatment response assessment, and surveillance in head and neck SCC.
TI-RADS (Thyroid Imaging Reporting & Data System) — ACR TI-RADS
ACR TI-RADS assigns points based on ultrasound features in five categories and sums them to determine the TI-RADS level:
Point system: Composition (cystic/spongiform = 0, mixed = 1, solid = 2); Echogenicity (anechoic = 0, hyper/isoechoic = 1, hypoechoic = 2, very hypoechoic = 3); Shape (wider-than-tall = 0, taller-than-wide = 3); Margin (smooth = 0, ill-defined = 0, lobulated/irregular = 2, extra-thyroidal extension = 3); Echogenic foci (none/large comet-tail = 0, macrocalcification = 1, peripheral rim calc = 2, punctate echogenic foci = 3).
TR1 (Benign, 0 points): No FNA needed. Simple cyst, purely cystic nodule. TR2 (Not Suspicious, 2 points): No FNA needed. Spongiform, mixed cystic-solid with comet-tail artifacts. TR3 (Mildly Suspicious, 3 points): FNA if ≥2.5 cm; follow-up if ≥1.5 cm. TR4 (Moderately Suspicious, 4-6 points): FNA if ≥1.5 cm; follow-up if ≥1.0 cm. TR5 (Highly Suspicious, ≥7 points): FNA if ≥1.0 cm; follow-up if ≥0.5 cm. Features with highest malignancy risk: taller-than-wide shape, punctate echogenic foci (microcalcifications), very hypoechoic, irregular margins.
18 Fracture Imaging & Classifications
Radiography is the primary imaging study for suspected fractures. Key principles: obtain at least two orthogonal views (AP and lateral); include the joint above and below the fracture site; describe fracture location (diaphysis, metaphysis, epiphysis), type (transverse, oblique, spiral, comminuted), displacement, angulation, and associated findings (joint effusion, dislocation, soft tissue swelling). CT is used when radiographs are equivocal or for surgical planning. MRI is the gold standard for occult fractures — marrow edema appears as T1 hypointense, T2/STIR hyperintense signal.
Ottawa Ankle & Knee Rules
Evidence-based clinical decision rules that determine the need for radiography, reducing unnecessary imaging by 30-40%:
Bone tenderness at the posterior edge or tip of either malleolus (distal 6 cm), OR inability to bear weight immediately after injury and in the ED (4 steps). Ottawa Foot Rules: Radiograph indicated if bone tenderness at the base of the 5th metatarsal, OR bone tenderness at the navicular, OR inability to bear weight. Ottawa Knee Rules: Radiograph indicated if age ≥55, isolated patellar tenderness, tenderness at the fibular head, inability to flex knee to 90 degrees, OR inability to bear weight (4 steps). Sensitivity approaches 100% for clinically significant fractures; applies to adults (age ≥18 for ankle/foot; ≥2 for knee).
Salter-Harris Classification — Pediatric Physeal Fractures
The mnemonic SALTR describes the path of the fracture line relative to the growth plate (physis):
| Type | Fracture Pattern | Radiographic Finding | Frequency / Prognosis |
|---|---|---|---|
| I — Slipped (S) | Through the physis only (separation of epiphysis from metaphysis) | May appear normal; widened physis on comparison view; diagnosed clinically | 6%; excellent prognosis; no growth disturbance |
| II — Above (A) | Through physis and up into the metaphysis (Thurston-Holland fragment) | Triangular metaphyseal fragment; widened physis | 75% (most common); excellent prognosis |
| III — Lower (L) | Through physis and down into the epiphysis (intra-articular) | Epiphyseal fracture line; joint surface involved | 8%; requires anatomic reduction; risk of growth arrest |
| IV — Through (T) | Through metaphysis, physis, and epiphysis (crosses all three) | Fracture line crosses growth plate obliquely | 10%; requires ORIF; highest risk of growth arrest and angular deformity |
| V — Rammed (R) | Crush injury to the physis (compression) | Often normal initially; diagnosed retrospectively when growth arrest occurs | 1%; worst prognosis; almost always causes growth arrest |
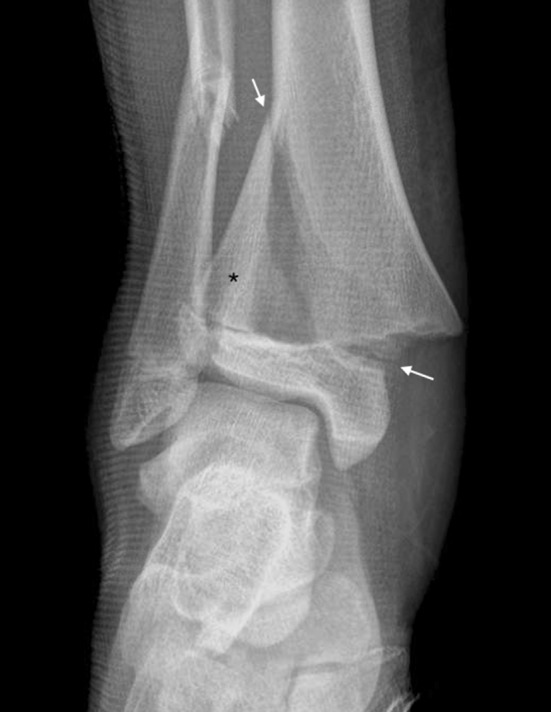
Garden Classification — Femoral Neck Fractures
| Grade | Description | Trabecular Alignment | Management |
|---|---|---|---|
| I — Incomplete / valgus impacted | Incomplete fracture; inferior cortex intact | Trabeculae angulated into valgus | Cannulated screws (internal fixation) |
| II — Complete, non-displaced | Complete fracture without displacement | Trabeculae interrupted but aligned | Cannulated screws |
| III — Complete, partially displaced | Complete with partial displacement; femoral head rotated | Trabeculae of head do not align with acetabulum | Hemiarthroplasty or THA (depending on age/activity) |
| IV — Complete, fully displaced | Complete with full displacement; no contact between fragments | Trabeculae of head re-align with acetabulum (paradoxical) | Hemiarthroplasty or THA; highest AVN risk (~30-40%) |
Weber Classification — Ankle Fractures
Based on the level of the fibular fracture relative to the ankle syndesmosis (tibiotalar joint line):
| Type | Fibular Fracture Level | Syndesmosis | Deltoid Ligament | Stability / Management |
|---|---|---|---|---|
| A — Below syndesmosis | At or below the level of the tibial plafond | Intact | Intact | Stable; usually non-operative (walking boot) |
| B — At the syndesmosis | At the level of the syndesmosis (spiral/oblique) | May be partially torn | May be torn | Variable stability; stress views may be needed; ORIF if unstable |
| C — Above the syndesmosis | Above the syndesmosis (proximal fibula — Maisonneuve) | Disrupted | Disrupted | Unstable; requires ORIF with syndesmotic fixation |
Stress Fractures & Pathologic Fractures
Stress fractures result from repetitive loading on normal bone (fatigue fractures) or normal loading on weakened bone (insufficiency fractures). Radiographs are often negative initially; periosteal reaction or sclerotic line may appear at 2-3 weeks. MRI is the most sensitive modality — periosteal edema (T2 hyperintense), marrow edema, and eventually a fracture line. Common locations: metatarsals (march fracture), tibial shaft, femoral neck, navicular. Pathologic fractures occur through abnormal bone — metastatic disease (look for lytic/blastic lesion at fracture site), osteoporosis, Paget disease, bone cysts. A fracture through a lytic lesion in an adult requires workup for underlying malignancy (primary or metastatic).
Additional Important Fracture Patterns
Maisonneuve fracture: proximal fibula fracture with disruption of the interosseous membrane and deltoid ligament/medial malleolus injury — the ankle radiograph may show only subtle medial clear space widening; always examine the proximal fibula on knee radiographs. Lisfranc injury: disruption of the tarsometatarsal joint — weight-bearing AP view shows widening between the base of the 1st and 2nd metatarsals, or a fleck fracture at the base of the 2nd metatarsal (Lisfranc ligament avulsion); missed in up to 20% of cases; CT is definitive. Scaphoid fracture: the most commonly fractured carpal bone; initial radiographs may be negative; MRI at 24-48 hours is the gold standard for occult fracture detection. Proximal pole fractures carry the highest risk of avascular necrosis due to the retrograde blood supply. Jefferson fracture: burst fracture of the C1 ring from axial loading; lateral mass offset of C1 on the open-mouth odontoid view. Hangman fracture: bilateral pars interarticularis fractures of C2 — traumatic spondylolisthesis of the axis. Chance fracture: horizontal fracture through the vertebral body extending through the pedicles and posterior elements — "seatbelt fracture"; associated with intra-abdominal injuries (bowel, mesenteric, splenic) in 50% of cases.
19 Joint & Soft Tissue Imaging
MRI is the primary modality for evaluating internal derangement of joints, soft tissue masses, and inflammatory arthropathies. The combination of multiplanar capability, superior soft tissue contrast, and absence of ionizing radiation makes MRI indispensable in musculoskeletal imaging.
MRI of the Knee
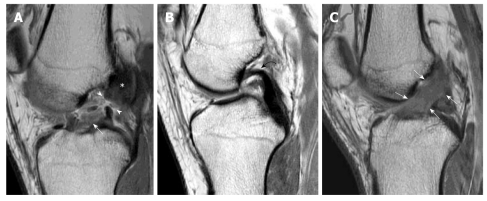
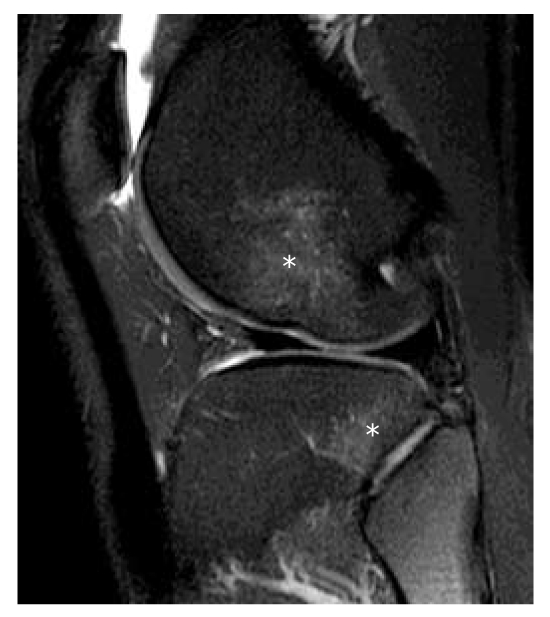
Meniscal tears: on MRI, a tear is diagnosed when high signal (grade 3) on proton density or T2-weighted images extends to the articular surface of the meniscus. Types: horizontal, vertical/longitudinal (including bucket-handle — a displaced fragment flipped into the intercondylar notch, producing the "double PCL sign" on sagittal images), radial, complex, and root tears. ACL tear: direct signs include discontinuity, abnormal signal, and abnormal orientation (the ACL normally parallels the Blumensaat line on sagittal images). Indirect signs: bone contusions of the lateral femoral condyle and posterolateral tibial plateau (pivot-shift mechanism), anterior tibial translation, posterior horn lateral meniscus tear (associated in 50-70%), deep lateral femoral notch sign. MCL tear: thickening and high T2 signal in the MCL along the medial joint line; associated with meniscal tears (O'Donoghue's unhappy triad: ACL + MCL + medial meniscus, though lateral meniscus injury is actually more common with ACL tears).
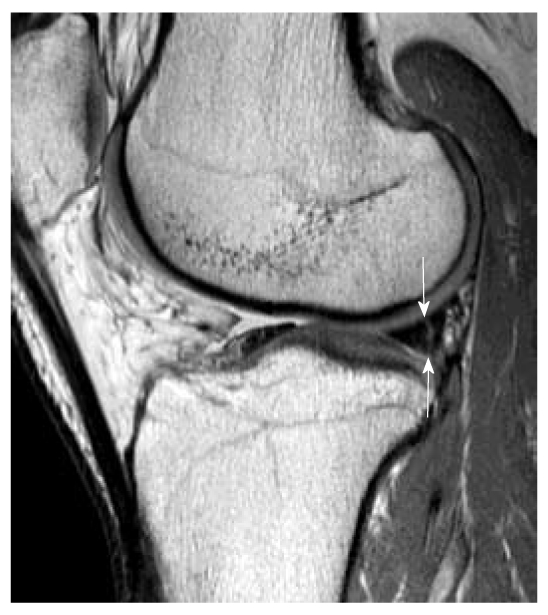
MRI of the Shoulder
Rotator cuff tears: supraspinatus is the most commonly torn tendon. On MRI: full-thickness tear shows tendon discontinuity with fluid-filled gap extending from bursal to articular surface (T2 bright). Partial-thickness tears may be articular-sided (more common) or bursal-sided. MR arthrography improves detection of partial tears and labral pathology. Labral tears: the glenoid labrum is a fibrocartilaginous ring that deepens the glenoid fossa. The Bankart lesion (anteroinferior labral tear) is the most common labral injury, associated with anterior shoulder dislocation. The SLAP tear (Superior Labrum Anterior to Posterior) involves the superior labrum at the biceps anchor — best seen on coronal oblique images with MR arthrography.
Arthritis Patterns on Imaging
| Feature | Osteoarthritis (OA) | Rheumatoid Arthritis (RA) | Gout | CPPD / Pseudogout |
|---|---|---|---|---|
| Distribution | DIP, PIP, 1st CMC, hip, knee, spine | MCP, PIP, wrist, MTP (bilateral symmetric) | 1st MTP (podagra), ankle, knee | Knee (most common), wrist, pubic symphysis |
| Joint space | Non-uniform narrowing | Uniform narrowing | Preserved until late | Preserved or narrowed |
| Erosions | Absent (osteophytes instead) | Marginal erosions (periarticular, juxta-articular osteopenia) | "Rat-bite" erosions with overhanging edges, sclerotic margins | Variable |
| Bone density | Subchondral sclerosis, osteophytes | Periarticular osteopenia | Preserved density | Chondrocalcinosis (cartilage calcification) |
| Soft tissue | No significant swelling | Symmetric soft tissue swelling, synovial pannus on MRI | Tophi (dense soft tissue masses, may calcify) | Meniscal/labral calcification, TFCC calcification |
| Key finding | Osteophytes | Marginal erosions + osteopenia | Erosion with overhanging edge | Chondrocalcinosis |
Soft Tissue Masses
MRI is the primary modality for soft tissue mass characterization. Lipoma: follows fat signal on all sequences (T1 bright, T2 intermediate, signal suppresses on fat-saturated sequences); no enhancement; well-encapsulated. Liposarcoma: contains fat but also non-fatty enhancing components, thick septa (>2 mm), nodularity. Ganglion cyst: well-defined, T2 hyperintense, arises from a joint capsule or tendon sheath. Giant cell tumor of tendon sheath (PVNS): T1 and T2 hypointense (hemosiderin — "blooming" on GRE/SWI), enhancing. Hemangioma: T2 hyperintense, serpentine vascular channels, may contain phleboliths. Any indeterminate enhancing soft tissue mass >5 cm or deep to fascia requires biopsy.
Hip Imaging
Avascular necrosis (AVN) of the femoral head: MRI is the most sensitive modality for early detection. Classic finding: serpiginous low T1 and low T2 signal line in the femoral head (reactive interface between viable and necrotic bone) with the "double line sign" on T2 (inner bright line of granulation tissue, outer dark line of sclerosis) — pathognomonic. Risk factors: corticosteroid use, alcohol, sickle cell disease, SLE, radiation, Gaucher disease. Staging: Ficat classification (Stage 0: normal radiograph and MRI; Stage I: normal radiograph, abnormal MRI; Stage II: sclerosis/cystic changes on radiograph; Stage III: subchondral fracture/"crescent sign"; Stage IV: collapse with secondary OA).
Femoroacetabular impingement (FAI): Cam-type: aspherical femoral head-neck junction ("bump" on the anterolateral femoral head-neck junction) causing labral and cartilage damage; alpha angle >55 degrees on MRI. Pincer-type: overcoverage of the femoral head by the acetabulum (crossover sign on AP pelvis radiograph — the anterior wall line crosses the posterior wall line, indicating acetabular retroversion); causes labral crush injury. Most cases are mixed cam-pincer. MR arthrography (intra-articular gadolinium) is the gold standard for detecting labral tears and cartilage delamination.
20 Bone Tumors & Metabolic Bone Disease
Bone Tumors — Age-Location Matrix
The combination of patient age and tumor location within bone narrows the differential diagnosis significantly:
| Tumor | Age Peak | Location in Bone | Preferred Bone | Key Imaging Feature |
|---|---|---|---|---|
| Osteosarcoma | 10-20 (primary); >60 (Paget-associated) | Metaphysis | Distal femur, proximal tibia, proximal humerus | Aggressive periosteal reaction (sunburst, Codman triangle), osteoid matrix |
| Ewing sarcoma | 5-15 | Diaphysis or metadiaphysis | Femur, pelvis, flat bones | Permeative "moth-eaten" destruction, onion-skin periosteal reaction, large soft tissue mass |
| Chondrosarcoma | 40-70 | Metaphysis or diaphysis | Pelvis, proximal femur, proximal humerus | Chondroid (rings-and-arcs) matrix calcification, endosteal scalloping |
| Giant cell tumor (GCT) | 20-40 | Epiphysis (extends to subchondral bone) | Distal femur, proximal tibia, distal radius | Eccentric, lytic, well-defined, no matrix, abuts articular surface |
| Osteochondroma | 10-30 | Metaphysis (surface) | Distal femur, proximal tibia, proximal humerus | Cortex and medullary canal continuous with parent bone; cartilage cap <2 cm (benign) |
| Enchondroma | 20-50 | Diaphysis or metaphysis (central) | Hands/feet (phalanges), proximal humerus, distal femur | Lobulated lytic lesion with chondroid calcifications; no periosteal reaction |
| Simple (unicameral) bone cyst | 5-15 | Metaphysis (central) | Proximal humerus, proximal femur | "Fallen fragment sign" (fracture fragment falls to dependent portion of cyst) |
Periosteal Reaction — Aggressive vs Non-Aggressive
Non-aggressive (benign) patterns: solid/thick periosteal reaction (dense, smooth, uniform — chronic osteomyelitis, healed fracture, slow-growing benign tumor). Aggressive (malignant/infection) patterns: lamellated (onion-skin) — multiple concentric layers (Ewing sarcoma, osteomyelitis), spiculated (sunburst/hair-on-end) — perpendicular spicules radiating from cortex (osteosarcoma), Codman triangle — triangular elevation of periosteum at the margin of a rapidly growing lesion (osteosarcoma, Ewing, aggressive infection), and interrupted/irregular periosteal reaction.
Osteoporosis — DEXA Scoring
Dual-energy X-ray absorptiometry (DEXA) measures bone mineral density (BMD). WHO classification based on T-score (standard deviations below mean BMD of healthy young adult): T-score ≥-1.0 = normal; T-score -1.0 to -2.5 = osteopenia; T-score ≤-2.5 = osteoporosis; T-score ≤-2.5 with fragility fracture = severe osteoporosis. Z-score compares to age-matched population — Z-score <-2.0 suggests secondary cause of bone loss. DEXA is measured at the lumbar spine (L1-L4) and proximal femur (femoral neck and total hip); the lowest T-score at any site determines the diagnosis. Screening: USPSTF recommends DEXA for all women ≥65 and postmenopausal women <65 with risk factors.
Paget Disease
Radiographic appearance depends on the disease phase: lytic phase — "osteoporosis circumscripta" (geographic lytic lesion in skull), "flame-shaped" / "blade of grass" advancing lytic front in long bones. Mixed phase — coarsened trabeculae, cortical thickening, bone expansion. Sclerotic (blastic) phase — dense, expanded bone ("cotton wool" appearance in skull, "picture frame" vertebra — sclerotic endplates with lucent center, "ivory vertebra" if entirely sclerotic). Complications: pathologic fracture, nerve compression (hearing loss from temporal bone involvement), high-output cardiac failure (hypervascular bone), sarcomatous degeneration (<1% — suspect if new pain or rapidly growing mass in previously stable Paget bone).
Multiple Myeloma
Imaging: skeletal survey (radiographs) has been the traditional screening tool — "punched-out" lytic lesions without reactive sclerosis (no osteoblastic response), diffuse osteopenia, vertebral compression fractures. Low-dose whole-body CT is more sensitive than skeletal survey and is now preferred. "Rain-drop skull" = multiple small lytic lesions throughout the calvarium. MRI is most sensitive for marrow infiltration (diffuse or focal T1 hypointense, T2 hyperintense marrow signal). PET/CT is used for treatment response assessment. Bone scintigraphy (bone scan) has limited sensitivity in myeloma because the lesions are purely lytic with no osteoblastic activity — a normal bone scan does NOT exclude myeloma.
Metastatic Bone Disease — Lytic vs Blastic
Bone metastases are the most common malignant bone tumors. The pattern helps identify the primary:
| Pattern | Common Primaries | Imaging Characteristics |
|---|---|---|
| Lytic (osteolytic) | Lung, renal, thyroid, melanoma, myeloma ("Kidney Loves Thyroid, Lung, Melanoma, Myeloma") | Destructive, dark on CT, risk of pathologic fracture, often permeative pattern |
| Blastic (osteosclerotic) | Prostate (most common), breast, carcinoid, medulloblastoma, Hodgkin lymphoma | Sclerotic (bright white on CT), dense on radiograph, may be diffuse |
| Mixed lytic/blastic | Breast (most common), lung, GI, GU | Combination of lytic and blastic lesions, variable appearance |
Bone scan (Tc-99m MDP) detects osteoblastic response and is sensitive for blastic metastases; it may miss purely lytic lesions (renal, thyroid, myeloma). PET/CT (FDG) detects metabolic activity in both lytic and blastic metastases and is increasingly used for staging. Whole-body MRI with diffusion-weighted imaging is the most sensitive modality for detecting bone marrow involvement and is increasingly adopted for myeloma and lymphoma staging.
This is a common and clinically important question in patients with known or suspected malignancy: Benign (osteoporotic) compression fracture: T1 hypointense band at endplate with preserved normal fatty marrow signal elsewhere, retropulsion of posterior fragment (not soft tissue mass), absence of pedicle signal abnormality, horizontal linear low-T1 band, normal adjacent disc. Malignant (pathologic) compression fracture: Complete T1 signal replacement of vertebral body (diffuse marrow infiltration), convex posterior body contour (epidural soft tissue mass), pedicle involvement (T1 hypointense signal in pedicle — highly specific), enhancement of entire vertebral body, associated paraspinal soft tissue mass. DWI can help: Acute benign fractures may show restricted diffusion (edema) but the ADC values are typically higher than in tumor; chronic benign fractures show no restriction. Tumor shows persistent restricted diffusion. Chemical shift imaging (in-phase/out-of-phase) can also help — benign fracture edema retains microscopic fat (signal drop on out-of-phase), while tumor replaces marrow fat (no signal drop).
21 Mammography & Screening
BI-RADS (Breast Imaging Reporting & Data System)
The ACR BI-RADS system standardizes mammographic reporting and provides management recommendations:
BI-RADS 0 (Incomplete): Additional imaging needed — spot compression, magnification views, ultrasound, or comparison with prior mammograms. Cannot render final assessment. BI-RADS 1 (Negative): No findings. Symmetric, no masses, calcifications, or architectural distortion. Routine screening. BI-RADS 2 (Benign): Definitely benign findings — involuting fibroadenomas with coarse calcifications, oil cysts (fat necrosis), lipomas, galactoceles, hamartomas, intramammary lymph nodes, vascular calcifications. Routine screening. BI-RADS 3 (Probably Benign): <2% probability of malignancy. Findings: solid mass with circumscribed margins, focal asymmetry, grouped round/punctate calcifications. Management: short-interval follow-up at 6 months, then 6 months, then 12 months (total 2 years of stability). If stable at 2 years, downgrade to BI-RADS 2. BI-RADS 4A (Low Suspicion): 2-10% malignancy risk. Palpable solid mass with circumscribed margins (not all features of fibroadenoma). Tissue diagnosis. BI-RADS 4B (Moderate Suspicion): 10-50% malignancy risk. Partially circumscribed mass, fine amorphous calcifications. Tissue diagnosis. BI-RADS 4C (High Suspicion): 50-95% malignancy risk. Irregular mass, fine linear/branching calcifications, new cluster of fine pleomorphic calcifications. Tissue diagnosis. BI-RADS 5 (Highly Suggestive of Malignancy): ≥95% probability of malignancy. Spiculated mass, irregular mass with fine linear/branching (casting) calcifications, spiculated mass with associated calcifications. Tissue diagnosis (biopsy). BI-RADS 6 (Known Malignancy): Biopsy-proven malignancy before definitive therapy. Used for staging or monitoring neoadjuvant chemotherapy response.
Screening Guidelines
USPSTF (2024): biennial screening mammography for women ages 40-74. ACR: annual screening mammography starting at age 40; risk assessment at age 25 to identify women who may benefit from earlier screening or supplemental screening with MRI. ACS: annual mammography ages 45-54, then biennial from 55 onward (option to continue annual); start at 40 based on patient preference. For women at high risk (≥20% lifetime risk by models such as Tyrer-Cuzick): annual mammography plus annual breast MRI starting at age 25-30.
Digital Breast Tomosynthesis (DBT / 3D Mammography)
DBT acquires multiple low-dose projections as the X-ray tube sweeps in an arc, reconstructing thin (1 mm) slices through the breast. Advantages over 2D mammography: reduces tissue overlap artifact, improves detection of masses and architectural distortion (especially in dense breasts), and reduces callback rates (false positives) by 15-40%. DBT is now the standard of care at most breast imaging centers, often combined with a synthesized 2D image (C-view) to avoid the additional dose of a separate 2D acquisition.
Calcification Morphology
Typically benign calcifications: skin (lucent center), vascular (tram-track parallel lines), coarse/popcorn (involuting fibroadenoma), large rod-like (ductal ectasia), round (lobular acini), rim/eggshell (fat necrosis, oil cyst), milk of calcium (teacup on 90-degree lateral), suture calcification. Suspicious calcifications: amorphous (BI-RADS 4B), coarse heterogeneous (BI-RADS 4B), fine pleomorphic (BI-RADS 4B-4C), fine linear/branching (BI-RADS 4C-5) — these fill the duct lumen and suggest DCIS (ductal carcinoma in situ). Distribution also matters: grouped or segmental distribution raises more concern than diffuse/scattered.
Breast Density Classification (ACR)
| Category | Description | Percentage of Fibroglandular Tissue | Clinical Impact |
|---|---|---|---|
| A — Almost entirely fatty | Breasts are almost entirely composed of fat | ~25% or less | High mammographic sensitivity; low masking risk |
| B — Scattered fibroglandular | Scattered areas of fibroglandular density | ~25-50% | Most common category; generally adequate sensitivity |
| C — Heterogeneously dense | Heterogeneously dense tissue that may obscure small masses | ~50-75% | Reduced sensitivity; supplemental screening may be considered |
| D — Extremely dense | Extremely dense tissue markedly reducing sensitivity | >75% | Markedly reduced sensitivity; supplemental screening recommended |
Multiple US states have enacted breast density notification laws requiring that patients be informed of their breast density and its implications for screening. The FDA's 2023 final rule mandates that all mammography facilities report breast density information to patients. Contrast-enhanced mammography (CEM) is an emerging alternative to breast MRI for supplemental screening — it involves IV iodinated contrast injection followed by dual-energy mammographic acquisition, producing a subtraction image that highlights areas of abnormal enhancement. CEM offers similar sensitivity to MRI at lower cost, shorter examination time, and with the familiar mammographic image format that breast imagers can interpret with minimal additional training.
Supplemental Screening Modalities
Whole-breast ultrasound: detects 2-4 additional cancers per 1000 women with dense breasts screened; however, it has a high false-positive rate (leading to unnecessary biopsies) and is operator-dependent. Automated breast ultrasound (ABUS) standardizes the examination and enables batch reading. Breast MRI: highest sensitivity for invasive cancer (~95-100%) but lower specificity (~70%), resulting in false positives and MRI-guided biopsies for benign findings. The abbreviated MRI (AB-MRI) protocol uses only pre- and post-contrast T1 sequences with subtraction and MIP, reducing scan time from 30-45 minutes to under 10 minutes while maintaining high sensitivity — this protocol is being studied as a potential mass-screening tool for women with dense breasts.
22 Breast MRI & Interventional
Breast MRI Indications
ACR-recommended indications for breast MRI: high-risk screening (≥20% lifetime risk — BRCA carriers, first-degree relatives of BRCA carriers, prior chest radiation ages 10-30, Li-Fraumeni syndrome); extent of disease assessment in newly diagnosed breast cancer (contralateral breast screening, assessment of multifocal/multicentric disease, chest wall invasion); neoadjuvant chemotherapy response monitoring; occult primary (axillary nodal metastasis with negative mammogram and ultrasound); implant evaluation (silicone implant integrity — MRI is the most sensitive modality for intracapsular and extracapsular rupture); and problem solving when mammography and US are inconclusive.
Enhancement Kinetics — Curve Types
Dynamic contrast-enhanced (DCE) breast MRI evaluates the time-intensity curve of enhancing lesions:
Type I (Persistent / Progressive): Signal intensity continues to increase over time. Most commonly seen in benign lesions (fibroadenoma, fibrocystic change). Malignancy rate: ~6%. Type II (Plateau): Initial rapid enhancement followed by leveling off. Indeterminate — seen in both benign and malignant lesions. Malignancy rate: ~30-60%. Type III (Washout): Initial rapid enhancement followed by signal decrease in the delayed phase. Highly suggestive of malignancy — reflects tumor neovascular leak. Malignancy rate: ~80-90%. Note: kinetics alone are not sufficient for diagnosis — morphology (mass shape, margins, internal enhancement) is equally important.
Image-Guided Breast Biopsy
Ultrasound-guided core needle biopsy: first-line for US-visible masses; real-time visualization of needle; 14-gauge spring-loaded or vacuum-assisted. Stereotactic biopsy: for mammographically visible calcifications or masses not seen on US; patient prone on dedicated table; paired stereo images localize the target in 3D; vacuum-assisted biopsy (9-11 gauge) preferred for calcifications. MRI-guided biopsy: for MRI-only lesions (enhancing foci not visible on US or mammography after targeted "second-look" US); performed with patient prone in MRI scanner; grid-and-pillar or computer-aided targeting. After any percutaneous biopsy, a radiopaque marker clip is placed at the biopsy site for future localization if surgery is needed. Concordance between imaging findings and pathology must be assessed — discordant results (e.g., suspicious imaging with benign pathology) require repeat biopsy or excision.
Pre-Operative Localization
When a non-palpable breast lesion requires surgical excision, pre-operative localization guides the surgeon to the target. Wire localization: a hooked wire is placed through the lesion under mammographic, US, or MRI guidance on the day of surgery; the wire tip should be within 1 cm of the target. Radioactive seed localization (RSL): a small I-125 seed is placed into the lesion up to 5 days before surgery; the surgeon uses a gamma probe to locate it intraoperatively. Magnetic seed and radar reflector localization (e.g., Magseed, SAVI SCOUT): non-radioactive alternatives that can be placed weeks before surgery, decoupling the localization from the surgical date. All methods require post-procedure imaging to confirm correct positioning relative to the target.
23 Pediatric-Specific Imaging
Image Gently — Principles
Children are more radiosensitive than adults (rapidly dividing cells, longer remaining life expectancy for stochastic effects to manifest). Pediatric CT dose should be reduced by 25-50% compared to adult protocols. The four pillars: child-size the technique (reduce kVp and mAs based on weight/size), scan only the indicated region, scan only when indicated (US and MRI first), scan once (avoid multiphase). The US-first approach is fundamental in pediatric imaging — many pediatric conditions (appendicitis, pyloric stenosis, intussusception, hip effusion, scrotal pathology) can be diagnosed with ultrasound, avoiding radiation entirely.
Hypertrophic Pyloric Stenosis (HPS)
Ultrasound is the definitive diagnostic study. Criteria (the "3s and 4s"): pyloric muscle thickness ≥3 mm (measured from outer wall to submucosa), pyloric channel length ≥15-17 mm (some use ≥14 mm). The thickened pylorus appears as a "donut" or "target" sign in cross-section and a "cervix sign" on longitudinal view. Real-time observation may show failure of the pyloric channel to open during gastric peristalsis. On upper GI series (if US is equivocal): "string sign" (elongated, narrow pyloric channel), "shoulder sign" (impression of the hypertrophied pylorus on the antrum), "double track sign" (compressed mucosa).
Intussusception
Ultrasound is the primary diagnostic modality — the target sign (concentric rings of bowel-within-bowel on transverse view) and pseudokidney sign (on longitudinal view). The most common type is ileocolic. US also identifies lead points in atypical presentations (age >3, outside typical 6-month to 3-year range). Air enema reduction (pneumatic reduction) is both diagnostic and therapeutic: air is insufflated rectally under fluoroscopic guidance; successful reduction is confirmed by free reflux of air into the ileum. Contraindications to enema reduction: peritonitis, perforation, hemodynamic instability. Recurrence rate after enema reduction: ~10%.
Necrotizing Enterocolitis (NEC)
Abdominal radiograph is the primary imaging modality in premature neonates with suspected NEC. Findings: pneumatosis intestinalis (intramural gas — linear or bubbly lucencies in the bowel wall; the pathognomonic radiographic finding), portal venous gas (branching lucencies extending to the liver periphery — ominous sign), pneumoperitoneum (free intraperitoneal air indicating perforation — surgical emergency; the "football sign" on supine radiograph is a large oval lucency with visible falciform ligament), and fixed dilated loop (a persistently dilated loop on serial radiographs suggesting necrotic bowel).
Non-Accidental Trauma (NAT) — Skeletal Survey
A skeletal survey is mandatory in suspected child abuse (<2 years old). High-specificity fractures for NAT: classic metaphyseal lesions (corner fractures / bucket-handle fractures — shearing forces at the metaphysis), posterior rib fractures (squeezing mechanism), scapular fractures, spinous process fractures, and sternal fractures. High-specificity but not pathognomonic: multiple fractures at different stages of healing (most characteristic overall finding of NAT), skull fractures (complex, bilateral, crossing suture lines), and long bone fractures in non-ambulatory infants. Follow-up skeletal survey at 2 weeks increases sensitivity for subtle fractures (healing periosteal reaction becomes visible).
Cranial Ultrasound — Neonates
Performed through the open anterior fontanelle in premature neonates. Primary indications: screening for germinal matrix hemorrhage / intraventricular hemorrhage (GMH-IVH) in premature infants. Grading: Grade I = germinal matrix hemorrhage only; Grade II = IVH filling <50% of ventricle without ventricular dilation; Grade III = IVH filling >50% of ventricle with ventricular dilation; Grade IV = parenchymal hemorrhagic infarction (not simply extension of IVH, but venous infarction of periventricular white matter). Also detects periventricular leukomalacia (PVL) — cystic changes in periventricular white matter appearing 2-3 weeks after ischemic insult.
24 Pediatric Chest & Airway
Croup (Laryngotracheobronchitis)
AP neck radiograph shows the steeple sign — symmetric subglottic narrowing of the trachea creating a pointed configuration (normally the subglottis has squared-off "shoulders" due to the cricoid cartilage). The lateral view shows a distended hypopharynx. Croup is a clinical diagnosis; imaging is reserved for atypical presentations or to exclude other causes of stridor (foreign body, epiglottitis, retropharyngeal abscess).
Epiglottitis
Lateral neck radiograph shows the thumb sign — a swollen, rounded epiglottis (normally thin and curved like a finger). Additional findings: thickened aryepiglottic folds, obliterated valleculae, and hypopharyngeal distension. Epiglottitis is a medical emergency; do not delay airway management for imaging. Incidence has decreased dramatically since routine Haemophilus influenzae type B (Hib) vaccination.
Foreign Body Aspiration
Most aspirated foreign bodies are radiolucent (food, plastic). Inspiratory/expiratory radiographs (or bilateral decubitus films in young children who cannot cooperate with expiratory films): air trapping on the affected side — the obstructed lung remains hyperinflated on expiration (acts as a ball-valve mechanism) while the normal lung deflates. On decubitus films, the dependent lung normally deflates; failure to deflate = air trapping. CT may be required when plain radiographs are equivocal. The right mainstem bronchus is the most common site for aspirated foreign bodies in children (as in adults), due to its wider caliber and more vertical orientation.
Neonatal Respiratory Distress — Radiographic Patterns
| Condition | Typical Timing | Chest X-Ray Findings | Key Feature |
|---|---|---|---|
| Respiratory Distress Syndrome (RDS / HMD) | Hours after birth in premature infants | Bilateral diffuse ground-glass opacities, air bronchograms, low lung volumes | Surfactant deficiency; bilateral and symmetric |
| Transient Tachypnea of the Newborn (TTN) | First hours, resolves within 24-72 hours | Perihilar streaky opacities, fluid in fissures, mild hyperinflation, small pleural effusions | "Wet lung" — retained fetal lung fluid; common after C-section |
| Meconium Aspiration Syndrome (MAS) | First hours in term/post-term infants | Coarse, patchy bilateral opacities, hyperinflation, pneumothorax (common complication) | Meconium-stained amniotic fluid; air trapping alternating with atelectasis |
| Neonatal Pneumonia (GBS) | Within 48 hours | Can mimic RDS — bilateral opacities, may have effusion | Indistinguishable from RDS on imaging; clinical context essential |
| Congenital Diaphragmatic Hernia (CDH) | Immediate | Bowel loops in the thorax (usually left-sided — Bochdalek hernia, 80%), mediastinal shift to contralateral side | Absent/partially absent hemidiaphragm; scaphoid abdomen |
Pediatric Musculoskeletal Imaging Pearls
In children, the growth plates (physes) are radiolucent and should not be confused with fractures. The comparison view of the contralateral extremity can help distinguish normal developmental variants from fractures in equivocal cases (though routine comparison views are discouraged by ACR if the diagnosis is clear). Toddler fracture: non-displaced spiral fracture of the tibial shaft in a child aged 1-3 years from twisting mechanism during walking/running; may be occult on initial radiographs — periosteal reaction appears at 10-14 days. Supracondylar fracture of the humerus: the most common elbow fracture in children (ages 4-8); look for the posterior fat pad sign ("sail sign") on lateral radiograph — posterior fat pad elevation is always abnormal and indicates joint effusion (occult fracture even if fracture line not visible). The anterior humeral line should intersect the middle third of the capitellum; anterior displacement of the line indicates extension-type fracture.
Congenital Pulmonary Anomalies
Congenital pulmonary airway malformation (CPAM), formerly CCAM: cystic or solid pulmonary mass; may present prenatally on US or with postnatal respiratory distress; classified by cyst size (type 1 = large cysts >2 cm, most common and best prognosis; type 2 = small cysts <2 cm; type 3 = microcystic/solid). Pulmonary sequestration: non-functioning lung tissue with systemic arterial blood supply and no connection to the tracheobronchial tree. Intralobar (75% — within the visceral pleura, venous drainage to pulmonary veins, presents in adolescence/adulthood with recurrent infections) vs extralobar (25% — separate pleural investment, systemic venous drainage, presents in infancy). CTA identifies the aberrant systemic feeding artery (typically from the thoracic or abdominal aorta), which is the key diagnostic finding.
Pediatric Abdominal Emergencies
Midgut volvulus: a surgical emergency resulting from intestinal malrotation with twisting of the small bowel around the superior mesenteric artery. Upper GI series is the gold standard: abnormal position of the duodenojejunal junction (ligament of Treitz — should be to the left of the spine at the level of the duodenal bulb); the "corkscrew" appearance of the twisted jejunum is pathognomonic. On US: the "whirlpool sign" (clockwise wrapping of the SMV and mesentery around the SMA). Bilious vomiting in a neonate is malrotation with volvulus until proven otherwise — this is among the most urgent diagnoses in pediatric radiology. Hirschsprung disease: contrast enema shows a transition zone between the narrow aganglionic distal segment and the dilated, normal proximal bowel (the "rectosigmoid ratio" is reversed — normally the rectum is larger than the sigmoid). Delayed 24-hour film showing retained contrast is supportive. Definitive diagnosis requires rectal biopsy (absence of ganglion cells).
25 Radiology Report Structure
Structured Reporting — Essential Components
The radiology report is the primary product of the radiologist. ACR Practice Parameters specify the following components:
Clinical Information: Relevant history, indication for the study, and specific clinical question. Drives the interpretation focus. Comparison: Identify prior studies used for comparison by date and modality. Comparison is essential for assessing interval change. Technique: Modality, body part, contrast administration (type, route, volume), any special protocols or modifications. Documents what was performed. Findings: Systematic description of all relevant observations, organized by anatomic region or pathologic significance. Use precise, standardized terminology. Report both positive and pertinent negative findings relative to the clinical question. Impression: Concise summary of the most important findings, diagnosis or differential diagnosis, and actionable recommendations. The impression should directly answer the clinical question. Number findings in order of clinical significance. Include standardized category assessments when applicable (BI-RADS, LI-RADS, Lung-RADS, etc.).
Critical Results Communication
The ACR Practice Parameter for Communication of Diagnostic Imaging Findings mandates direct (non-routine) communication with the ordering provider for critical or unexpected findings that may require immediate or urgent intervention. This communication must occur in a timely manner — minutes for emergent findings, hours for urgent findings.
Emergent (immediate): Tension pneumothorax, aortic dissection/rupture, massive PE with right heart strain, acute intracranial hemorrhage with midline shift, ectopic pregnancy with free fluid, testicular torsion, bowel perforation with pneumoperitoneum, malpositioned critical devices (ETT in right mainstem, NGT in bronchus). Urgent (within hours): New pulmonary embolism, new stroke, unexpected malignancy (e.g., pancreatic mass), critical incidental findings (large aneurysm, adrenal mass concerning for pheochromocytoma), worsening condition on follow-up. Non-urgent but important: Incidental findings requiring follow-up (lung nodule, thyroid nodule), discrepancy with prior report, recommendations for additional imaging. Documentation should include: who was contacted, by whom, when (date and time), how (phone, page, in-person), and what was communicated. This documentation is a medicolegal requirement.
RADPEER
RADPEER is an ACR peer review program where radiologists rate the accuracy of prior interpretations when reviewing current studies with priors. Scoring: 1 = concur with prior interpretation; 2 = discrepancy, not clinically significant; 3 = discrepancy, clinically significant but unlikely to be made in most practices; 4 = discrepancy, clinically significant, should have been made. RADPEER provides feedback for quality improvement and is used by accreditation bodies.
Artificial Intelligence in Radiology
AI/machine learning algorithms are increasingly integrated into radiology workflow. FDA-cleared applications include: triage (flagging critical findings such as large vessel occlusion on CTA head, pneumothorax on CXR, and PE on CTPA — prioritizing worklist order so emergent findings are read first), detection (computer-aided detection of lung nodules, rib fractures, breast lesions on mammography), quantification (automated calcium scoring, liver fat fraction measurement, bone density estimation from CT), and workflow optimization (automated hanging protocols, structured report generation, critical finding alerts). AI does not replace radiologist judgment — it augments it. Current FDA regulatory framework classifies most radiology AI as "computer-aided" (requiring radiologist oversight, not autonomous). Challenges include algorithm bias from non-representative training data, "black box" interpretability, and validation across diverse patient populations and scanner platforms.
Actionable Recommendations
When the report includes a recommendation for follow-up imaging or intervention, it should specify the recommended modality, timeframe, and clinical rationale. Vague recommendations ("clinical correlation recommended," "follow-up as clinically indicated") should be avoided in favor of specific, evidence-based guidance (e.g., "6-month follow-up chest CT recommended per Fleischner Society guidelines for 7 mm solid nodule in a high-risk patient"). Tracking systems for incidental findings (lung nodules, adrenal nodules, thyroid nodules, liver lesions) help ensure follow-up compliance.
Common Incidental Findings & Management
| Incidental Finding | Prevalence | Key Threshold | Recommended Follow-up |
|---|---|---|---|
| Pulmonary nodule | ~25% of chest CTs | <6 mm in low-risk: no follow-up | Fleischner Society 2017 guidelines |
| Adrenal nodule | ~4% of abdominal CTs | ≤10 HU = adenoma (no follow-up needed) | ACR Incidental Findings Committee white paper; ≥4 cm → further workup |
| Thyroid nodule on CT | ~15-25% of neck CTs | ≥1 cm (age <35) or ≥1.5 cm (age ≥35) | Dedicated thyroid US + TI-RADS assessment |
| Renal cyst | Extremely common | Bosniak I-II: no follow-up | Bosniak IIF: serial imaging; III-IV: urology referral |
| Hepatic cyst | ~15-18% of adults | Simple cyst with thin wall, no septations | No follow-up for simple cysts regardless of size |
| Pancreatic cyst | ~3% of abdominal CTs | <1.5 cm without concerning features | ACR/AGA guidelines; MRI with MRCP for characterization; ≥3 cm or mural nodule → EUS/surgery |
| Liver lesion in non-cirrhotic | Common | Hypodense lesion <1 cm: usually too small to characterize | If <1 cm in low-risk patient: likely benign cyst/hemangioma; follow-up or MRI if uncertain |
26 Image-Guided Procedures
Fluoroscopic Procedures
Upper GI series: barium swallow evaluates the esophagus (strictures, webs, Schatzki ring, achalasia — "bird beak" tapering, Zenker diverticulum) and stomach (ulcers, masses, hiatal hernia). Small bowel follow-through: serial images of barium transit through the small bowel — largely replaced by CT enterography for Crohn disease and small bowel tumors but still used for motility evaluation. Barium enema: retrograde filling of the colon — largely replaced by colonoscopy and CT colonography but may show "apple core" lesion (annular carcinoma), "lead pipe" sign (ulcerative colitis — loss of haustrations), "string sign" (Crohn stricture), and "thumbprinting" (mucosal edema from ischemia or infection). Voiding cystourethrogram (VCUG): fluoroscopic study of bladder filling and voiding — the gold standard for diagnosing vesicoureteral reflux (VUR) in children; graded I-V by the extent of retrograde contrast flow.
Hysterosalpingography (HSG): fluoroscopic injection of contrast through the cervix to evaluate uterine cavity morphology and fallopian tube patency in the infertility workup. Normal: smooth endometrial cavity with bilateral tubal filling and free spillage of contrast into the peritoneal cavity (confirms tubal patency). Abnormal: filling defects (polyps, fibroids, synechiae), uterine anomalies (septate, bicornuate), tubal occlusion (proximal or distal — hydrosalpinx appears as a distended, fluid-filled tube without spillage).
CT-Guided Biopsy
CT guidance provides precise targeting for deep or small lesions. Common targets: lung nodules/masses, mediastinal lymph nodes, retroperitoneal masses, bone lesions, and liver lesions not accessible by US. Technique: plan trajectory on pre-procedure CT, anesthetize skin and deeper tissues, advance coaxial needle under intermittent CT imaging, confirm needle tip position within the target, obtain core biopsy (18-gauge) or FNA (20-22 gauge). Pneumothorax is the most common complication of CT-guided lung biopsy (~20% overall, ~5% requiring chest tube). Risk factors: small lesion, deep lesion, emphysema, traversal of a fissure, multiple passes.
Ultrasound-Guided Biopsy & Drainage
US guidance is preferred when the target is visible sonographically — real-time visualization, no radiation, bedside availability. Applications: thyroid FNA, liver biopsy, kidney biopsy, lymph node biopsy, soft tissue mass biopsy, breast biopsy, paracentesis, thoracentesis, and abscess drainage. Abscess drainage: under US or CT guidance, an 8-14 French catheter is placed into the collection using the Seldinger or trocar technique; catheter is left in place for ongoing drainage until output decreases and the cavity collapses.
Paracentesis & Thoracentesis
Paracentesis: US identifies the largest pocket of ascites and guides needle insertion, avoiding the inferior epigastric vessels (map with color Doppler). Preferred site: left lower quadrant. US guidance reduces the risk of bowel or vessel injury. Thoracentesis: US confirms the presence and location of the pleural effusion, identifies the diaphragm, and guides needle insertion. Insert just above the rib to avoid the intercostal neurovascular bundle (which runs along the inferior rib margin). US-guided thoracentesis reduces pneumothorax rate from ~10% (blind) to <1%.
Lumbar Puncture Under Fluoroscopy
Fluoroscopic guidance is used when bedside LP is unsuccessful (obesity, prior lumbar surgery, severe degenerative disease). The patient is positioned prone; the interspinous space (typically L3-L4 or L4-L5) is identified under fluoroscopy; a spinal needle is advanced under real-time imaging to the subarachnoid space. CSF flow confirms correct positioning. Myelography can be performed simultaneously by injecting intrathecal iodinated contrast (non-ionic, preservative-free — e.g., iohexol), followed by CT for detailed evaluation of spinal canal, nerve roots, and cord.
Myelography
CT myelography (intrathecal contrast + CT) remains the gold standard when MRI is contraindicated (pacemaker, severe claustrophobia, MRI-incompatible implants) or provides insufficient detail. Indications: spinal stenosis evaluation, CSF leak localization (positional headache, low-pressure syndrome), post-surgical spine with hardware artifact obscuring MRI, and assessment of nerve root sleeve integrity. Contrast: non-ionic iodinated contrast (iohexol, iopamidol) injected intrathecally, followed by CT acquisition. Complications: post-dural-puncture headache (most common), seizure (rare — avoid metrizamide, which is no longer used), meningitis, subdural hematoma.
Vascular Access Procedures
Interventional radiologists perform a wide range of vascular and non-vascular procedures under image guidance. These procedures are increasingly performed as minimally invasive alternatives to surgery.
Vascular Access Procedures
PICC line placement: peripherally inserted central catheter, typically placed via basilic or brachial vein under US guidance with fluoroscopic confirmation of tip position at the cavoatrial junction. Tunneled central venous catheter (Hickman, Broviac): placed via the internal jugular vein under US and fluoroscopic guidance; subcutaneous tunnel provides infection resistance; tip at cavoatrial junction. Implantable port (Port-a-Cath): subcutaneous reservoir with attached catheter; placed under fluoroscopic guidance via the internal jugular or subclavian vein; used for long-term chemotherapy access. Inferior vena cava (IVC) filter: placed under fluoroscopic guidance via the femoral or jugular vein for patients with venous thromboembolism who have contraindications to anticoagulation; retrievable filters should be removed as soon as the indication resolves (complication rates increase with longer dwell time — strut penetration, filter fracture, IVC thrombosis, migration).
Embolization
Transcatheter embolization is performed under fluoroscopic/angiographic guidance to occlude blood vessels therapeutically. Applications: trauma (splenic, hepatic, pelvic hemorrhage — coil or gelfoam embolization may avoid surgery), GI hemorrhage (when endoscopy fails — super-selective embolization of bleeding vessel), uterine artery embolization (UAE) (treatment for symptomatic fibroids — particulate embolization of bilateral uterine arteries), bronchial artery embolization (massive hemoptysis), hepatic chemoembolization (TACE) (HCC treatment — targeted delivery of chemotherapy + embolic material to tumor-feeding arteries), and varicocele embolization. Embolic agents include coils (permanent mechanical occlusion), gelfoam (temporary — resorbs in 2-6 weeks), polyvinyl alcohol particles (PVA), microspheres, and liquid agents (Onyx, glue — for AVMs).
Standardized reporting systems accomplish three goals: they ensure consistent language across radiologists, provide evidence-based risk stratification, and include built-in management recommendations. Always include the system name and version (e.g., "BI-RADS 4C per ACR 5th edition"), the specific category with its descriptor, and the recommended next step. If findings straddle two categories, assign the higher category (err on the side of caution). Familiarity with these systems is expected of all radiologists and is increasingly expected of referring clinicians.
27 Classification Systems Master Table
A consolidated reference of the major imaging classification and reporting systems used across radiology. These standardized systems reduce ambiguity, provide evidence-based management recommendations, and enable quality benchmarking across practices.
| System | Organ | Categories | Key Decision Point |
|---|---|---|---|
| BI-RADS | Breast | 0 (Incomplete), 1 (Negative), 2 (Benign), 3 (Probably Benign), 4A/4B/4C (Suspicious), 5 (Highly Suggestive), 6 (Known Cancer) | BI-RADS 4+ → biopsy; BI-RADS 3 → 6-month follow-up |
| LI-RADS | Liver (cirrhosis/HBV) | LR-1 (Benign), LR-2 (Probably Benign), LR-3 (Intermediate), LR-4 (Probably HCC), LR-5 (Definitely HCC), LR-M (Malignant, not HCC-specific), LR-TIV (Tumor in Vein) | LR-5 → treat as HCC without biopsy; LR-M → biopsy |
| Lung-RADS | Lung (screening) | 1 (Negative), 2 (Benign), 3 (Probably Benign), 4A (Suspicious), 4B (Very Suspicious), 4X (additional suspicious features) | 3 → 6-month LDCT; 4A → 3-month LDCT; 4B → diagnostic CT/PET/biopsy |
| TI-RADS (ACR) | Thyroid | TR1 (Benign), TR2 (Not Suspicious), TR3 (Mildly Suspicious), TR4 (Moderately Suspicious), TR5 (Highly Suspicious) | TR3 → FNA if ≥2.5 cm; TR4 → FNA if ≥1.5 cm; TR5 → FNA if ≥1.0 cm |
| PI-RADS (v2.1) | Prostate | 1 (Very Low), 2 (Low), 3 (Intermediate), 4 (High), 5 (Very High) | PI-RADS 4-5 → MRI-targeted biopsy; PI-RADS 3 → clinical factors guide decision |
| Bosniak | Renal cyst | I (Simple), II (Minimally Complex), IIF (Follow-up), III (Indeterminate), IV (Clearly Malignant) | IIF → serial imaging; III → surgery or surveillance; IV → surgery |
| O-RADS (MRI) | Ovary | 1 (Normal), 2 (Almost Certainly Benign), 3 (Low Risk), 4 (Intermediate), 5 (High Risk) | O-RADS 4-5 → gynecologic oncology referral |
| Fleischner (2017) | Lung nodule (incidental) | Based on size, risk, and solid vs subsolid morphology | <6 mm low-risk solid → no follow-up; ≥8 mm → CT at 3 months or PET/biopsy |
28 CT Protocols Quick Reference
| Clinical Indication | Protocol | Contrast | Phase / Timing | Key Notes |
|---|---|---|---|---|
| Pulmonary embolism | CTPA | IV — bolus tracking in PA | Pulmonary arterial (~15-20 s) | ECG gating not required; look for filling defects + RV strain |
| Aortic dissection / traumatic aortic injury | CTA chest | IV — bolus tracking in aorta | Arterial (~20-25 s) + delayed | ECG gating preferred; identify intimal flap, true/false lumen |
| Lung cancer staging | CT chest/abdomen/pelvis with contrast | IV | Portal venous (60-70 s) | Include adrenals and liver for metastases |
| Lung nodule screening | Low-dose CT (LDCT) | None | N/A | 100 kVp, reduced mAs; Lung-RADS reporting |
| Interstitial lung disease | HRCT chest | None (sometimes IV for limited indications) | N/A | Thin sections, inspiration + expiration (air trapping) |
| Acute abdomen (appendicitis, diverticulitis) | CT abdomen/pelvis with IV contrast | IV (± oral) | Portal venous | Oral contrast may delay study; many centers use IV only |
| Liver mass characterization | Multiphase liver CT | IV | Arterial (25-35 s) + portal venous (60-70 s) + delayed (3-5 min) | LI-RADS in cirrhosis/HBV; assess enhancement and washout |
| Pancreatic mass / pancreatitis | Pancreas protocol CT | IV + neutral oral (water) | Pancreatic (arterial, 40 s) + portal venous | Pancreatic tumors are hypovascular (hypodense in pancreatic phase) |
| Hematuria | CT urogram (3-phase) | IV | Non-contrast + nephrographic (100 s) + excretory (10-15 min) | Evaluate stones, renal mass, urothelial lesions |
| Renal colic | CT KUB (non-contrast) | None | N/A | Low-dose protocol adequate; >95% sensitivity for stones |
| Acute stroke | CT head + CTA head/neck + CTP | IV for CTA/CTP | Non-contrast head → CTA → CTP | ASPECTS scoring; core vs penumbra on CTP; time is brain |
| Trauma (polytrauma) | CT head + C-spine + chest/abdomen/pelvis ("pan-scan") | IV | Arterial (for CTA) + portal venous | Active extravasation = arterial-phase blush; solid organ injury grading (AAST) |
| Mesenteric ischemia | CTA abdomen | IV — bolus tracking in aorta | Arterial + portal venous | SMA/SMV occlusion, bowel wall changes, pneumatosis |
| Bowel obstruction | CT abdomen/pelvis | IV; oral controversial | Portal venous | Identify transition point, closed loop, strangulation signs |
| Liver lesion (non-cirrhotic) | MRI with extracellular agent or hepatobiliary agent | IV gadolinium | Arterial + portal venous + delayed (± hepatobiliary phase if using Eovist) | MRI preferred over CT for characterization; Eovist adds 20-minute hepatobiliary phase |
| Adrenal mass characterization | CT adrenal washout protocol | IV | Non-contrast + enhanced (60 s) + delayed (15 min) | Calculate absolute/relative washout; ≤10 HU non-contrast = adenoma |
| Suspected AAA rupture | CTA abdomen/pelvis | IV — rapid bolus | Arterial | Retroperitoneal hematoma, active extravasation, "draped aorta" sign |
| Cervical spine trauma | CT cervical spine | None (CTA if vascular injury suspected) | N/A | Thin-section axial with sagittal/coronal reformats; 3-column assessment |
29 Normal Measurements & Values
Knowledge of normal measurements and thresholds is essential for identifying pathology on imaging. The following tables provide reference values for the most commonly measured structures. Measurements should be made using standardized techniques (e.g., outer wall to outer wall for the aorta, short axis for lymph nodes) and interpreted in clinical context — a structure at the upper limit of normal may be abnormal for a particular patient if prior studies show interval increase.
Vascular
| Structure | Normal Measurement | Abnormal Threshold |
|---|---|---|
| Ascending aorta | <4.0 cm | ≥4.0 cm = dilated; ≥4.5 cm = aneurysmal (some use ≥5.0 cm) |
| Descending thoracic aorta | <3.0 cm | ≥3.5 cm = aneurysmal |
| Abdominal aorta (infrarenal) | <2.0 cm | ≥3.0 cm = AAA; ≥5.5 cm = surgical threshold (men) |
| Main pulmonary artery | <2.9 cm | ≥2.9 cm suggests pulmonary hypertension; MPA:aorta ratio >1.0 is suggestive |
| IVC | 1.5-2.5 cm diameter | >2.5 cm with <50% respiratory variation suggests elevated RA pressure |
| Portal vein | <13 mm | ≥13 mm suggests portal hypertension |
Abdominal Organs
| Structure | Normal Measurement | Clinical Significance of Abnormality |
|---|---|---|
| Liver (craniocaudal, MCL) | <15.5 cm | Hepatomegaly; evaluate for cirrhosis, infiltrative disease, congestion, mass |
| Spleen (craniocaudal) | <13 cm | Splenomegaly; portal hypertension, hematologic malignancy, infection |
| Common bile duct (CBD) | <6 mm (add 1 mm per decade after age 60; <10 mm post-cholecystectomy) | CBD dilatation: choledocholithiasis, stricture, mass (pancreatic head, cholangiocarcinoma) |
| Pancreatic duct | <3 mm (body); <1.5 mm (tail) | Dilatation: chronic pancreatitis, obstructing mass, IPMN |
| Kidney (long axis) | 9-12 cm (adults) | <9 cm bilateral = chronic kidney disease; asymmetry >1.5 cm → renal artery stenosis |
| Appendix diameter | <6 mm (outer wall to outer wall) | ≥6 mm with wall thickening/fat stranding = appendicitis |
| Small bowel diameter | <3 cm | ≥3 cm proximal to transition point = small bowel obstruction |
| Colon diameter | <6 cm (cecum <9 cm) | Cecum >9 cm = risk of perforation; >12 cm = imminent perforation |
| Adrenal gland (limb thickness) | <10 mm (body); limb should not exceed ipsilateral crus | Thickening: hyperplasia, adenoma, metastasis, pheochromocytoma |
Neuroradiology
| Structure | Normal Measurement | Clinical Significance |
|---|---|---|
| Lateral ventricles (Evans index) | <0.3 (frontal horn width / biparietal diameter) | ≥0.3 = ventriculomegaly; evaluate for hydrocephalus vs atrophy |
| Third ventricle | <7 mm (adults) | Dilation suggests obstructive or communicating hydrocephalus |
| Optic nerve sheath diameter | <5 mm (measured 3 mm behind the globe on US or MRI) | ≥5 mm suggests elevated intracranial pressure |
| Midline shift | 0 mm | ≥5 mm = clinically significant; may warrant surgical intervention |
| Cervical spinal canal (AP diameter) | ≥13 mm | <13 mm = relative stenosis; <10 mm = absolute stenosis |
| Lumbar spinal canal (AP diameter) | ≥12 mm | <12 mm = relative stenosis; <10 mm = absolute stenosis |
Musculoskeletal
| Structure | Normal Measurement | Clinical Significance |
|---|---|---|
| Achilles tendon (AP thickness) | <6 mm | ≥6 mm = tendinopathy or partial tear |
| Plantar fascia (insertion thickness) | <4 mm | ≥4 mm = plantar fasciitis |
| Rotator cuff (supraspinatus tendon) | Homogeneous, low signal; ~5-6 mm thick | Thinning, signal abnormality, or gap = tear |
| Atlantodental interval (ADI) | <3 mm (adults); <5 mm (children) | ≥3 mm (adults) = atlantoaxial instability (RA, Down syndrome, trauma) |
Obstetric & Gynecologic
| Structure / Parameter | Normal Measurement | Clinical Significance |
|---|---|---|
| Gestational sac (mean sac diameter) | Visible at ~5 weeks (beta-hCG ~1500-2000) | Empty sac >25 mm (TVUS) without embryo = anembryonic pregnancy |
| Crown-rump length (CRL) | Embryo visible at CRL ~2-4 mm (~6 weeks) | CRL ≥7 mm without cardiac activity = embryonic demise |
| Nuchal translucency | <3 mm at 11-14 weeks | ≥3 mm = increased risk of chromosomal abnormality (trisomy 21, 18, 13) and structural heart defects |
| Cervical length (transvaginal) | ≥25 mm at 16-24 weeks | <25 mm = short cervix; increased risk of preterm delivery |
| Endometrial thickness (postmenopausal) | <4-5 mm | ≥4-5 mm in postmenopausal bleeding → endometrial biopsy to exclude carcinoma |
| Ovarian volume | <20 cm3 (premenopausal); <10 cm3 (postmenopausal) | Enlarged ovary raises concern for neoplasm; any solid component in postmenopausal ovary is concerning |
Chest
| Structure | Normal Measurement | Clinical Significance |
|---|---|---|
| Cardiothoracic ratio (PA CXR) | <0.5 | ≥0.5 = cardiomegaly (not valid on AP films) |
| Trachea (coronal diameter) | 15-25 mm (men); 10-21 mm (women) | Narrowing: stenosis, goiter, external compression; widening: tracheobronchomegaly (Mounier-Kuhn) |
| Mediastinal width (PA CXR) | <8 cm | ≥8 cm = mediastinal widening; evaluate for aortic injury, lymphadenopathy, mass |
| Lymph node short axis (mediastinal) | <10 mm | ≥10 mm short axis = pathologically enlarged; differential includes malignancy, sarcoidosis, infection |
| Aortic valve area (CT/echo) | 3-4 cm2 | <1.0 cm2 = severe aortic stenosis |
30 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AAA | Abdominal aortic aneurysm |
| ACR | American College of Radiology |
| ADC | Apparent diffusion coefficient |
| ALARA | As low as reasonably achievable |
| AML | Angiomyolipoma |
| AP | Anteroposterior |
| APHE | Arterial phase hyperenhancement |
| ASPECTS | Alberta Stroke Program Early CT Score |
| ATCM | Automatic tube current modulation |
| AVM | Arteriovenous malformation |
| BI-RADS | Breast Imaging Reporting and Data System |
| BMD | Bone mineral density |
| CBD | Common bile duct |
| CMR | Cardiac magnetic resonance |
| CPAM | Congenital pulmonary airway malformation |
| CR | Computed radiography |
| CTA | CT angiography |
| CTDIvol | CT dose index (volume) |
| CTP | CT perfusion |
| CTPA | CT pulmonary angiography |
| CXR | Chest X-ray |
| DBT | Digital breast tomosynthesis |
| DCE | Dynamic contrast-enhanced |
| DCIS | Ductal carcinoma in situ |
| DECT | Dual-energy CT |
| DEXA | Dual-energy X-ray absorptiometry |
| DIS | Dissemination in space |
| DIT | Dissemination in time |
| DLP | Dose-length product |
| DR | Digital radiography |
| DQE | Detective quantum efficiency |
| DWI | Diffusion-weighted imaging |
| EBUS | Endobronchial ultrasound |
| ERCP | Endoscopic retrograde cholangiopancreatography |
| ETT | Endotracheal tube |
| FBP | Filtered back projection |
| FLAIR | Fluid-attenuated inversion recovery |
| FNA | Fine needle aspiration |
| FNH | Focal nodular hyperplasia |
| GBCA | Gadolinium-based contrast agent |
| GBM | Glioblastoma multiforme |
| GCT | Germ cell tumor |
| GGO | Ground-glass opacity |
| GMH | Germinal matrix hemorrhage |
| GRE | Gradient recalled echo |
| HCC | Hepatocellular carcinoma |
| HIDA | Hepatobiliary iminodiacetic acid (scan) |
| HOCM | High-osmolality contrast media |
| HPS | Hypertrophic pyloric stenosis |
| HRCT | High-resolution CT |
| HU | Hounsfield units |
| IABP | Intra-aortic balloon pump |
| IOCM | Iso-osmolality contrast media |
| IPMN | Intraductal papillary mucinous neoplasm |
| IR | Iterative reconstruction |
| IVC | Inferior vena cava |
| IVH | Intraventricular hemorrhage |
| kVp | Kilovoltage peak |
| LBO | Large bowel obstruction |
| LDCT | Low-dose CT |
| LGE | Late gadolinium enhancement |
| LI-RADS | Liver Imaging Reporting and Data System |
| LNT | Linear no-threshold (model) |
| LOCM | Low-osmolality contrast media |
| LP | Lumbar puncture |
| Lung-RADS | Lung CT Screening Reporting and Data System |
| mAs | Milliampere-seconds |
| MCA | Middle cerebral artery |
| MDCT | Multidetector CT |
| mpMRI | Multiparametric MRI |
| MRA | MR angiography |
| MRCP | MR cholangiopancreatography |
| MRS | MR spectroscopy |
| mSv | Millisievert |
| NAA | N-acetylaspartate |
| NAT | Non-accidental trauma |
| NEC | Necrotizing enterocolitis |
| NET | Neuroendocrine tumor |
| NGT | Nasogastric tube |
| NOMI | Non-occlusive mesenteric ischemia |
| NSF | Nephrogenic systemic fibrosis |
| O-RADS | Ovarian-Adnexal Reporting and Data System |
| OMC | Ostiomeatal complex |
| ORIF | Open reduction internal fixation |
| PA | Posteroanterior |
| PC-AKI | Post-contrast acute kidney injury |
| PCWP | Pulmonary capillary wedge pressure |
| PE | Pulmonary embolism |
| PET | Positron emission tomography |
| PI-RADS | Prostate Imaging Reporting and Data System |
| PLC | Posterior ligamentous complex |
| PVL | Periventricular leukomalacia |
| PVNS | Pigmented villonodular synovitis |
| RCC | Renal cell carcinoma |
| RF | Radiofrequency |
| RV | Right ventricle |
| SAH | Subarachnoid hemorrhage |
| SBO | Small bowel obstruction |
| SMA | Superior mesenteric artery |
| SMV | Superior mesenteric vein |
| SVC | Superior vena cava |
| SWI | Susceptibility-weighted imaging |
| TI-RADS | Thyroid Imaging Reporting and Data System |
| TLICS | Thoracolumbar Injury Classification and Severity Score |
| TOF | Time of flight (MRA) |
| TR | Repetition time |
| TE | Echo time |
| TVUS | Transvaginal ultrasound |
| US | Ultrasound |
| UIP | Usual interstitial pneumonia |
| V/Q | Ventilation/perfusion (scan) |
| WHO | World Health Organization |
Radiology Report Abbreviation Standards
While abbreviations improve workflow efficiency, the ACR recommends that the final impression section of radiology reports use full terminology rather than abbreviations to avoid miscommunication. In the findings section, widely understood abbreviations (CT, MRI, US, IV) are acceptable, but less common ones should be spelled out on first use. The Joint Commission maintains a "Do Not Use" list of dangerous abbreviations (e.g., "U" for units, "QD" for daily) that must not appear in any medical documentation. Radiology-specific abbreviations should be understood in context — "PE" means pulmonary embolism in chest imaging but pericardial effusion in echocardiography context.
ABCDE for CXR: Airway, Bones, Cardiac, Diaphragm, Everything else. 4 T's of anterior mediastinum: Thymoma, Teratoma, Terrible lymphoma, Thyroid. SALTR for Salter-Harris: Slipped, Above, Lower, Through, Rammed. Aunt Minnie: A classic appearance so pathognomonic that the diagnosis is made instantly (e.g., "coffee bean" sigmoid volvulus, "string of pearls" SBO, "popcorn calcification" hamartoma, "sunburst" osteosarcoma, "double bubble" duodenal atresia). VINDICATE for differential diagnosis: Vascular, Infectious/Inflammatory, Neoplastic, Degenerative, Iatrogenic, Congenital, Autoimmune, Traumatic, Endocrine/metabolic. Mnemonic for contrast reaction treatment — severe anaphylaxis: Epinephrine IM first (0.3-0.5 mg of 1:1000), then ABC — Airway, Breathing, Circulation.