Histology
Tissue architecture, microscopic anatomy, epithelial types, connective tissue, muscle, nervous tissue, organ-specific histology, staining techniques, and every cell type, tissue feature, and pathologic correlate across the full scope of medical histology.
01 Overview & Significance
Histology is the study of microscopic anatomy—the structure and organization of cells and tissues that compose every organ in the human body. Understanding tissue architecture is foundational to pathology, since nearly every disease process produces identifiable changes at the cellular or tissue level. The ability to recognize normal histologic patterns is the prerequisite for identifying pathologic deviations in biopsy specimens, surgical resections, and autopsy material.
The four fundamental tissue types—epithelial, connective, muscle, and nervous—combine in characteristic patterns to form every organ. Each tissue type has a distinct embryologic origin, structural organization, and functional role. Organs are defined by their unique combination of these tissue types arranged in reproducible architectural patterns.
Histology bridges gross anatomy and molecular biology. Clinicians rely on tissue-level understanding to interpret biopsy results, understand drug targets, predict disease progression, and communicate effectively with pathologists. Virtually every clinical specialty encounters histologic findings in diagnosis and treatment planning.
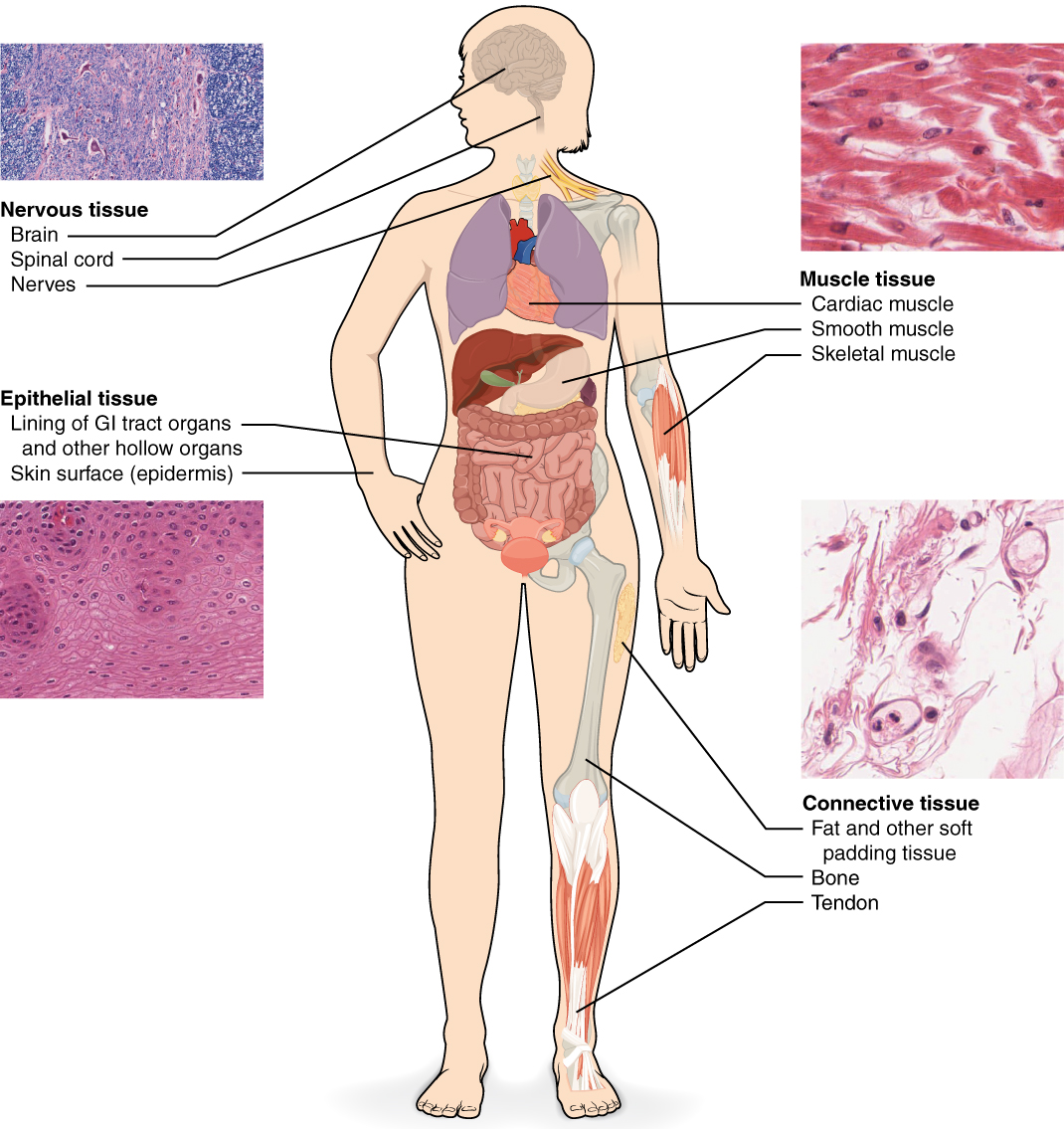
The Four Fundamental Tissue Types
| Tissue Type | Primary Origin | Key Function | Defining Feature |
|---|---|---|---|
| Epithelial | All three germ layers | Covering, lining, secretion | Cells on basement membrane; avascular |
| Connective | Mesoderm (mesenchyme) | Support, binding, transport | Cells in extracellular matrix |
| Muscle | Mesoderm | Contraction, movement | Contractile filaments (actin/myosin) |
| Nervous | Ectoderm (neuroectoderm) | Signal transmission | Electrically excitable cells with processes |
Pathologists classify tumors based on their tissue of origin: carcinomas (epithelial), sarcomas (connective/muscle), lymphomas (lymphoid), and gliomas (glial). Accurate classification determines treatment protocols and prognosis.
02 Tissue Processing & Preparation
Proper tissue processing is essential for producing high-quality histologic sections. The standard workflow involves fixation, dehydration, clearing, embedding, sectioning, and staining. Each step introduces potential artifacts that the histologist must recognize and distinguish from genuine pathology.
Fixation
10% neutral buffered formalin (NBF) is the universal fixative in clinical pathology. It cross-links proteins (primarily through lysine residues), preserving tissue architecture and preventing autolysis. Fixation should begin within minutes of excision; delayed fixation causes autolytic artifacts—loss of nuclear detail, cytoplasmic pallor, and detachment of epithelial lining.
| Fixative | Mechanism | Best Use | Limitation |
|---|---|---|---|
| 10% NBF | Protein cross-linking | Routine surgical specimens | Slow penetration (~1 mm/hr) |
| Glutaraldehyde | Strong cross-linking | Electron microscopy | Excessive hardening for LM |
| Bouin solution | Picric acid + formalin | Testicular biopsies, GI biopsies | Damages DNA for molecular tests |
| Carnoy fixative | Alcohol-based | Glycogen, lymph node biopsies | Causes tissue shrinkage |
| Zamboni fixative | Paraformaldehyde + picric acid | Immunohistochemistry | Limited availability |
Embedding & Sectioning
After dehydration through graded alcohols and clearing in xylene, tissue is infiltrated with paraffin wax (melting point 56–58°C). A microtome cuts 4–6 μm sections that are floated onto glass slides. For rapid intraoperative diagnosis, frozen sections bypass embedding: tissue is snap-frozen in OCT compound and cut on a cryostat at −20°C, yielding results within 15–20 minutes but with inferior morphologic detail.
Common Processing Artifacts
| Artifact | Cause | Appearance |
|---|---|---|
| Formalin pigment | Acidic formalin on bloody tissue | Brown-black birefringent granules |
| Tissue retraction | Dehydration/clearing | Clear spaces around structures |
| Freeze artifact | Ice crystal formation | "Swiss cheese" vacuolation |
| Crush artifact | Forceps handling | Distorted, dark-staining cells |
| Cautery artifact | Electrosurgery at margins | Elongated, hyperchromatic nuclei |
03 Staining Techniques
Stains transform colorless tissue sections into interpretable images by selectively binding to cellular and extracellular components. The hematoxylin and eosin (H&E) stain is the cornerstone of histologic diagnosis, but hundreds of special stains and immunohistochemical markers exist for specific diagnostic questions.
H&E Stain
Hematoxylin is a basic (cationic) dye that binds acidic structures—primarily DNA and RNA—staining nuclei blue-purple (basophilic). Eosin is an acidic (anionic) dye that binds basic structures—most cytoplasmic proteins—staining them pink-red (eosinophilic/acidophilic). The combination reveals nuclear-to-cytoplasmic ratio, chromatin pattern, cell boundaries, and tissue architecture.
Key Special Stains
| Stain | Target | Color Result | Clinical Use |
|---|---|---|---|
| PAS (Periodic Acid–Schiff) | Glycogen, glycoproteins, mucins | Magenta | Fungal walls, basement membranes, glycogen storage diseases |
| PAS-diastase | Non-glycogen PAS+ material | Magenta (glycogen digested) | Distinguishes glycogen from mucin |
| Masson trichrome | Collagen vs. muscle | Blue/green = collagen; red = muscle | Fibrosis assessment (liver, heart, kidney) |
| Reticulin (silver) | Reticular fibers (type III collagen) | Black fibers on gold background | Liver architecture, lymphoma classification |
| Congo red | Amyloid | Orange-red; apple-green birefringence | Amyloidosis diagnosis |
| Prussian blue (Perls) | Ferric iron (hemosiderin) | Blue granules | Iron overload, hemochromatosis, sideroblasts |
| Oil Red O | Neutral lipids | Red droplets (frozen sections only) | Fatty liver, lipid storage diseases |
| Alcian blue | Acid mucopolysaccharides | Blue | Mucin-secreting tumors, cartilage matrix |
| Von Kossa | Calcium deposits | Black | Calcification in tissues |
| Elastic van Gieson (EVG) | Elastic fibers | Black = elastic; red = collagen | Vascular pathology, emphysema |
| Gomori methenamine silver (GMS) | Fungal cell walls | Black organisms | Fungal infections (Pneumocystis, Aspergillus) |
| Ziehl–Neelsen | Acid-fast bacilli | Red bacilli on blue background | Mycobacterium tuberculosis |
| Gram stain | Bacteria | Purple (Gram+) or pink (Gram−) | Bacterial identification in tissue |
Immunohistochemistry (IHC)
IHC uses antibodies conjugated to chromogens (typically DAB, producing brown staining) to detect specific antigens in tissue sections. It is indispensable for tumor classification, prognostic markers, and identifying cell lineage. Key principles include antigen retrieval (heat-induced or enzymatic) to unmask formalin-crosslinked epitopes, and the use of appropriate positive and negative controls.
| Marker | Target Cell/Tissue | Diagnostic Use |
|---|---|---|
| Cytokeratins (AE1/AE3, CK7, CK20) | Epithelial cells | Carcinoma identification; CK7/CK20 pattern for site of origin |
| Vimentin | Mesenchymal cells | Sarcoma, melanoma, lymphoma |
| Desmin | Muscle cells | Rhabdomyosarcoma, leiomyosarcoma |
| S-100 | Neural crest derivatives | Melanoma, schwannoma, chondrocytes |
| CD45 (LCA) | All leukocytes | Lymphoma vs. carcinoma |
| CD20 | B lymphocytes | B-cell lymphoma |
| CD3 | T lymphocytes | T-cell lymphoma |
| Ki-67 | Proliferating cells | Proliferation index / tumor grade |
| p53 | Mutant p53 protein | High-grade dysplasia, many carcinomas |
| ER / PR | Estrogen/progesterone receptors | Breast cancer treatment planning |
| HER2 | ErbB2 receptor | Breast/gastric cancer targeted therapy |
| Chromogranin / Synaptophysin | Neuroendocrine cells | Neuroendocrine tumors |
04 Simple Epithelia
Epithelial tissue lines body surfaces, cavities, and tubes, and forms glands. All epithelia rest on a basement membrane (basal lamina + reticular lamina), are avascular (nourished by diffusion from underlying connective tissue), and exhibit polarity (apical, lateral, and basal domains). Classification is based on the number of cell layers (simple vs. stratified) and cell shape (squamous, cuboidal, columnar).
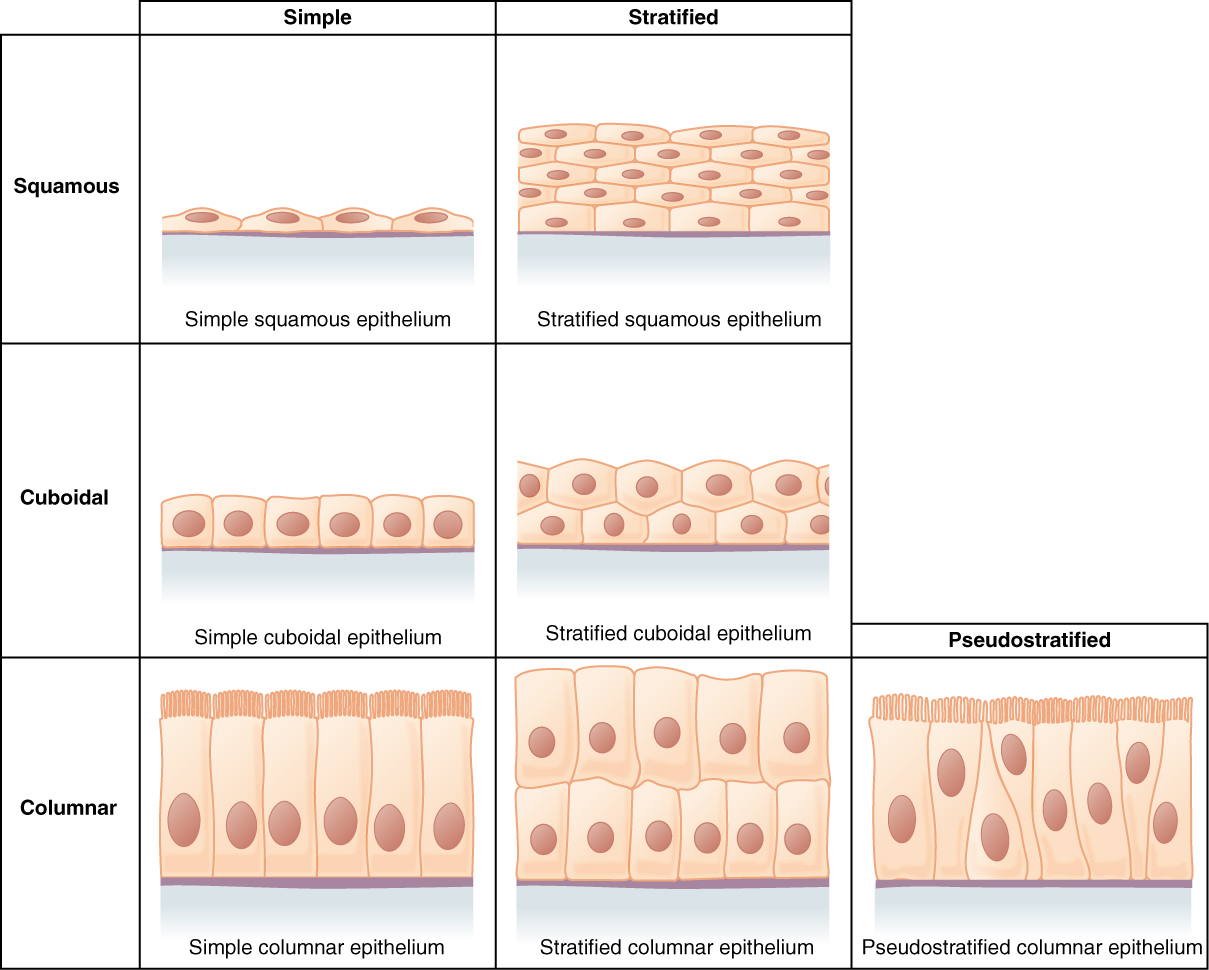
Simple Squamous Epithelium
A single layer of flat cells with disc-shaped nuclei. Optimized for diffusion and filtration due to minimal cytoplasmic thickness. Special names: endothelium (lines blood vessels), mesothelium (lines serous cavities—pleura, pericardium, peritoneum). Found also in Bowman capsule, thin loop of Henle, and alveolar type I cells.
Simple Cuboidal Epithelium
A single layer of cube-shaped cells with round, central nuclei. Specialized for secretion and absorption. Found in kidney tubules (proximal and distal convoluted tubules), thyroid follicles, small collecting ducts, and covering the ovary (germinal epithelium). Proximal tubule cells have a prominent brush border (microvilli) that dramatically increases absorptive surface area.
Simple Columnar Epithelium
A single layer of tall cells with oval, basally located nuclei. The primary lining of the GI tract from the stomach to the rectum. May be ciliated (fallopian tubes, small bronchi) or non-ciliated with microvilli (intestinal absorptive cells). Goblet cells are interspersed throughout, secreting mucus for lubrication and protection.
Pseudostratified Columnar Epithelium
Appears stratified but all cells contact the basement membrane—not all reach the apical surface. Nuclei at varying heights create the false impression of multiple layers. The ciliated form lines the respiratory tract (trachea, bronchi) and contains goblet cells; together they form the mucociliary escalator. The non-ciliated form with stereocilia (long microvilli) lines the epididymis and vas deferens.
| Structure | Core | Function | Location |
|---|---|---|---|
| Microvilli | Actin filaments | Increase absorptive surface | Small intestine, proximal tubule |
| Stereocilia | Actin filaments (long) | Absorption, concentration | Epididymis, hair cells of ear |
| Cilia | Microtubules (9+2 axoneme) | Propel mucus/fluid | Trachea, oviduct, ependyma |
| Flagella | Microtubules (9+2) | Cell locomotion | Spermatozoa |
Lateral Domain Specializations
The lateral domain of epithelial cells mediates cell-cell adhesion and communication. In addition to the junctional complex (tight junctions, adherens junctions, desmosomes), the lateral membrane contains lateral intercellular spaces that widen during active fluid transport (as seen in gallbladder epithelium absorbing water). Cadherins are calcium-dependent transmembrane adhesion molecules: E-cadherin predominates in epithelia, N-cadherin in neural and mesenchymal cells, P-cadherin in placenta. Loss of E-cadherin expression is a hallmark of lobular carcinoma of the breast and diffuse gastric carcinoma, and is used diagnostically on IHC.
Basal Domain Specializations
The basal domain anchors epithelial cells to the underlying basement membrane through hemidesmosomes (integrin α6β4 + BP antigens connecting to keratin intermediate filaments) and focal adhesions (integrin-mediated connections to the actin cytoskeleton). Basal infoldings are deep membrane invaginations at the basal surface that increase surface area for ion transport; they are packed with mitochondria and prominent in the distal convoluted tubule, striated ducts of salivary glands, and choroid plexus epithelium. These appear as basal striations on light microscopy.
Epithelial polarity is disrupted in carcinogenesis. Loss of tight junction integrity contributes to increased paracellular permeability in inflammatory bowel disease. Loss of E-cadherin (CDH1 gene) promotes epithelial-mesenchymal transition (EMT), a key step in cancer metastasis. Hereditary diffuse gastric cancer syndrome involves germline CDH1 mutations.
05 Stratified & Transitional Epithelia
Stratified epithelia have multiple cell layers, providing protection against mechanical abrasion, chemical insult, and desiccation. Classification is based on the shape of the surface (apical) cells, regardless of the shape of deeper layers.
Stratified Squamous Epithelium
The most widespread protective epithelium. The keratinized form covers the skin (epidermis)—surface cells are dead, anucleate, and filled with keratin. The non-keratinized form lines the oral cavity, esophagus, vagina, and anal canal—surface cells retain their nuclei and remain moist. Basal cells are mitotically active and continuously replace cells lost from the surface.
Stratified Cuboidal & Columnar
Rare epithelia with limited distribution. Stratified cuboidal (2 layers): lines sweat gland ducts and large excretory ducts of salivary glands. Stratified columnar (2+ layers): found in the male urethra, large excretory ducts, and conjunctiva of the eye. Neither type is commonly tested but may appear as distractors.
Transitional Epithelium (Urothelium)
Unique to the urinary tract (renal pelvis, ureters, bladder, proximal urethra). Surface cells are large, dome-shaped "umbrella cells" with specialized plaques of uroplakin proteins on the apical surface. When the bladder is relaxed, epithelium appears 5–6 layers thick with rounded surface cells; when distended, it thins to 2–3 layers with flattened surface cells. This is the only epithelium that changes appearance with mechanical stretch.
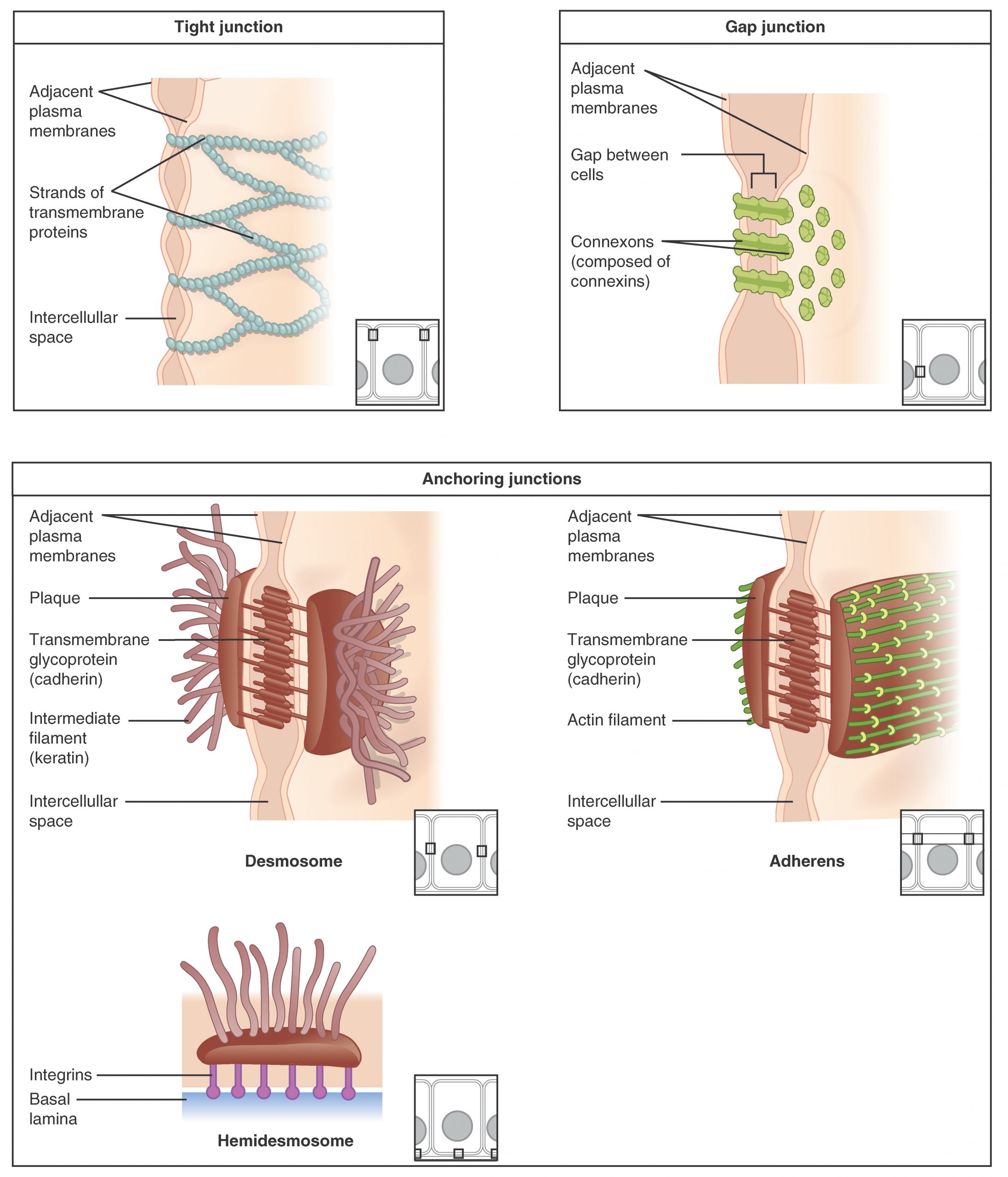
Epithelial Cell Junctions
| Junction | Location | Function | Key Proteins |
|---|---|---|---|
| Tight junction (zonula occludens) | Most apical | Seal; regulate paracellular permeability | Claudins, occludin, ZO-1 |
| Adherens junction (zonula adherens) | Below tight junctions | Cell-cell adhesion; link to actin | E-cadherin, catenins |
| Desmosome (macula adherens) | Below adherens | Strong spot adhesion; resist shear | Desmoglein, desmocollin, desmoplakin |
| Gap junction | Variable | Direct cell-cell communication | Connexins → connexons |
| Hemidesmosome | Basal surface | Attach cell to basement membrane | Integrin α6β4, BP antigens |
Pemphigus vulgaris: autoantibodies against desmoglein 3 (and 1) destroy desmosomes → intraepidermal acantholysis → flaccid blisters with + Nikolsky sign. Bullous pemphigoid: autoantibodies against BP180/BP230 at hemidesmosomes → subepidermal blisters → tense blisters, − Nikolsky sign.
Basement Membrane
The basement membrane underlies all epithelia and surrounds muscle cells, Schwann cells, and adipocytes. It consists of the basal lamina (lamina lucida + lamina densa, primarily type IV collagen, laminin, entactin/nidogen, and heparan sulfate proteoglycans) and the reticular lamina (type III collagen fibers from underlying connective tissue). It acts as a structural scaffold, molecular filter, and barrier to cell migration—cancer invasion through the basement membrane defines the transition from carcinoma in situ to invasive carcinoma.
06 Glandular Epithelium
Glands are epithelial structures specialized for secretion. They arise embryologically as epithelial invaginations into underlying connective tissue. Glands are classified as exocrine (secrete via ducts to a surface) or endocrine (secrete hormones directly into blood; ductless).
Exocrine Gland Classification
| Feature | Types | Examples |
|---|---|---|
| Duct structure | Simple (unbranched) vs. Compound (branched) | Simple: sweat glands; Compound: pancreas |
| Secretory unit shape | Tubular, acinar (alveolar), tubuloacinar | Tubular: intestinal glands; Acinar: pancreas; Tubuloacinar: submandibular gland |
| Secretion type | Serous (watery, enzyme-rich), Mucous (viscous, mucin), Mixed | Serous: parotid; Mucous: sublingual; Mixed: submandibular |
Modes of Secretion
| Mode | Mechanism | Example |
|---|---|---|
| Merocrine (eccrine) | Exocytosis; cell intact | Pancreatic acinar cells, eccrine sweat glands, salivary glands |
| Apocrine | Apical cytoplasm pinched off with product | Mammary gland (lipid secretion), apocrine sweat glands |
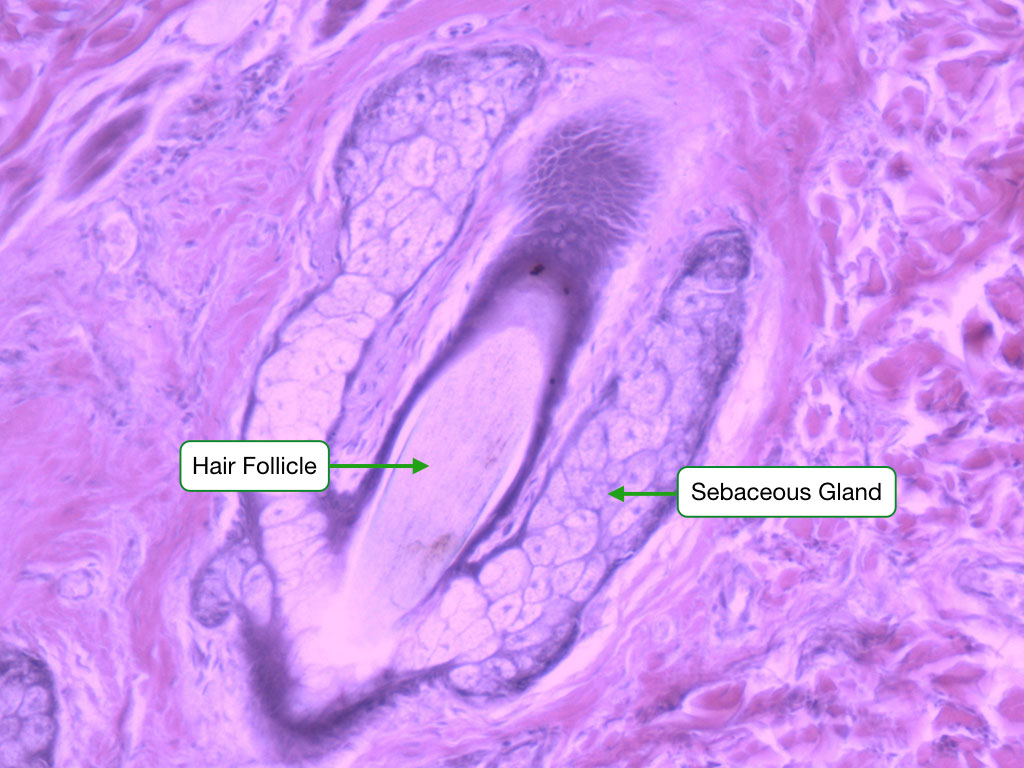
| Holocrine | Entire cell disintegrates to release product | Sebaceous glands |
Endocrine Glands
Endocrine glands lack ducts and release hormones into fenestrated capillaries. They are organized as cords and clusters (adrenal cortex, anterior pituitary, parathyroid) or follicles (thyroid—the only endocrine gland that stores its product extracellularly as colloid). Endocrine cells may also be scattered individually within other epithelia as the diffuse neuroendocrine system (DNES/APUD cells): enteroendocrine cells of the gut, pulmonary neuroendocrine cells, and C cells of the thyroid.
Myoepithelial Cells
Myoepithelial cells are contractile epithelial cells located between the secretory/ductal epithelium and the basement membrane of many exocrine glands (salivary glands, mammary gland, sweat glands, lacrimal gland). They have features of both epithelial (cytokeratin+) and smooth muscle (SMA+, calponin+) cells. Their contraction assists in expelling secretory products into ducts. Diagnostically, the presence of myoepithelial cells on IHC (p63+, SMA+, calponin+) distinguishes benign breast lesions and carcinoma in situ from invasive carcinoma, where the myoepithelial layer is lost.
Salivary Gland Summary
| Gland | Type | Secretion | Duct Features |
|---|---|---|---|
| Parotid | Purely serous | Watery, amylase-rich | Stensen duct; striated ducts prominent |
| Submandibular | Mixed (serous predominant) | Mixed; serous demilunes | Wharton duct; most common sialolith site |
| Sublingual | Mixed (mucous predominant) | Viscous, mucin-rich | Multiple small ducts (of Rivinus) |
The standard duct hierarchy in compound glands: intercalated ducts (low cuboidal; modify secretion) → striated ducts (columnar with basal striations; electrolyte/water modification) → interlobular (excretory) ducts (larger; carry secretion to surface). Striated ducts are so named because of basal membrane infoldings packed with mitochondria that create a striated appearance on LM.
07 Connective Tissue Proper
Connective tissue is the most abundant and widely distributed tissue type. Unlike epithelium, its cells are separated by abundant extracellular matrix (ECM) consisting of ground substance and fibers. All connective tissue derives from embryonic mesenchyme (mesoderm), except in the head region where neural crest contributes.
Connective Tissue Cells
| Cell Type | Function | Key Features |
|---|---|---|
| Fibroblast | Synthesizes ECM (collagen, elastin, GAGs) | Most common CT cell; spindle-shaped; active = euchromatic nucleus |
| Macrophage (histiocyte) | Phagocytosis, antigen presentation | Derived from blood monocytes; kidney-shaped nucleus |
| Mast cell | Releases histamine, heparin, proteases | Metachromatic granules with toluidine blue; IgE receptor (FcεRI) |
| Plasma cell | Antibody secretion | "Clock-face" nucleus; perinuclear hof (Golgi); abundant RER |
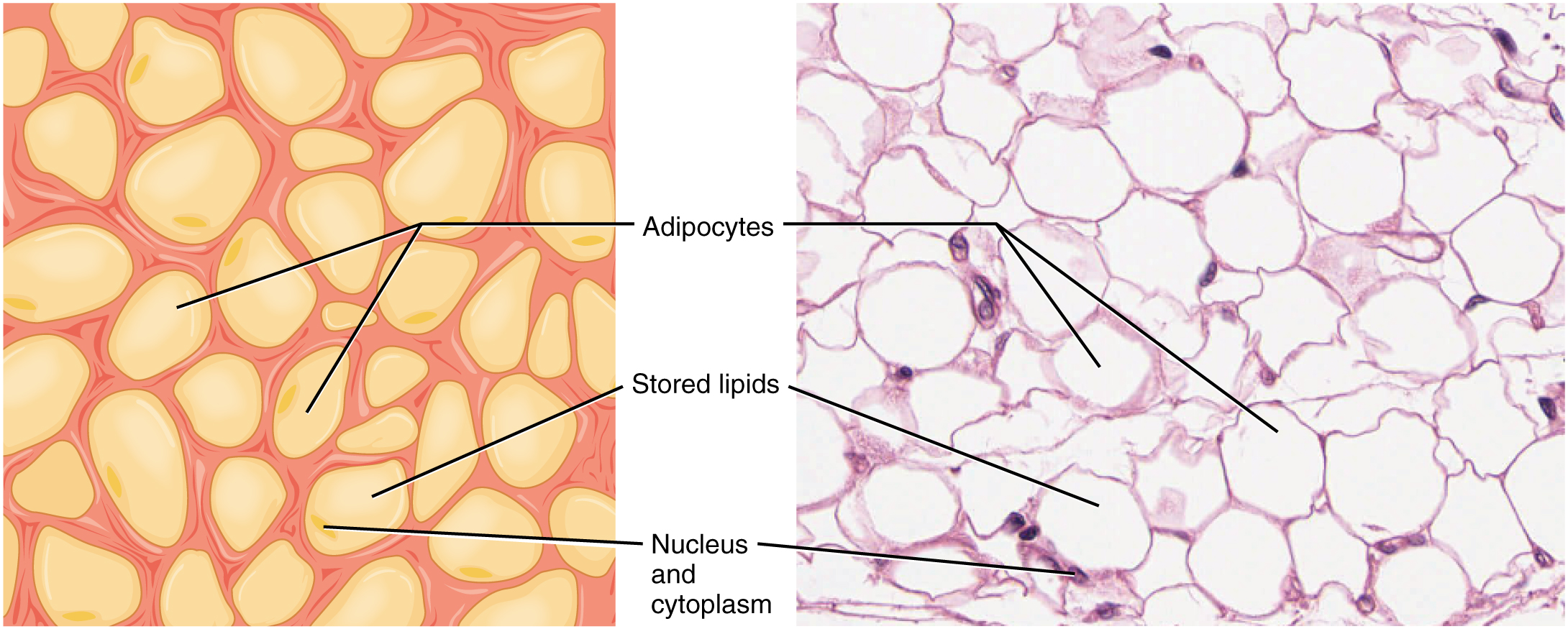
| Adipocyte | Energy storage, insulation, endocrine | White: single large lipid droplet; Brown: multilocular, many mitochondria |
| Mesenchymal stem cell | Multipotent progenitor | Can differentiate into fibroblast, chondrocyte, osteoblast, adipocyte |
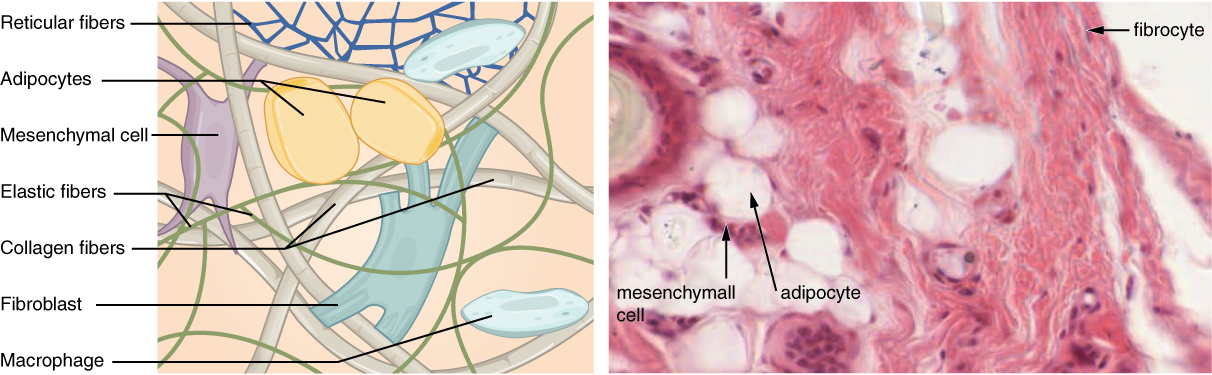
Connective Tissue Fibers
| Fiber Type | Composition | Properties | Stain |
|---|---|---|---|
| Collagen (type I) | Triple helix; 67-nm banding | High tensile strength | Eosinophilic on H&E; blue with Masson trichrome |
| Reticular (type III collagen) | Thin, branching fibers | Scaffolding for organs | Silver stains (reticulin); PAS+ |
| Elastic | Elastin core + fibrillin microfibrils | Stretch and recoil | Orcein, Verhoeff, EVG stains |
Collagen Types (High-Yield)
| Type | Location | Associated Disease |
|---|---|---|
| I | Bone, skin, tendon, dentin, cornea | Osteogenesis imperfecta (type I) |
| II | Cartilage (hyaline, elastic), vitreous humor | Achondrogenesis type II |
| III | Reticular fibers, blood vessels, uterus, skin | Ehlers-Danlos syndrome (vascular type) |
| IV | Basement membrane | Alport syndrome (COL4A5); Goodpasture syndrome (anti-GBM) |
| V | Placenta, skin, cornea | Classic Ehlers-Danlos |
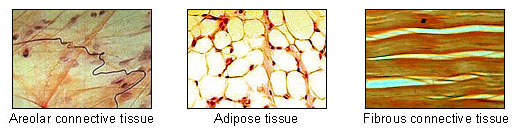
Types of Connective Tissue Proper
Types of Connective Tissue Proper
| Type | Fiber Arrangement | Location | Key Feature |
|---|---|---|---|
| Loose (areolar) | Loosely arranged; all fiber types | Beneath epithelia, around vessels/nerves, mesentery | Flexible; well-vascularized; all CT cell types present |
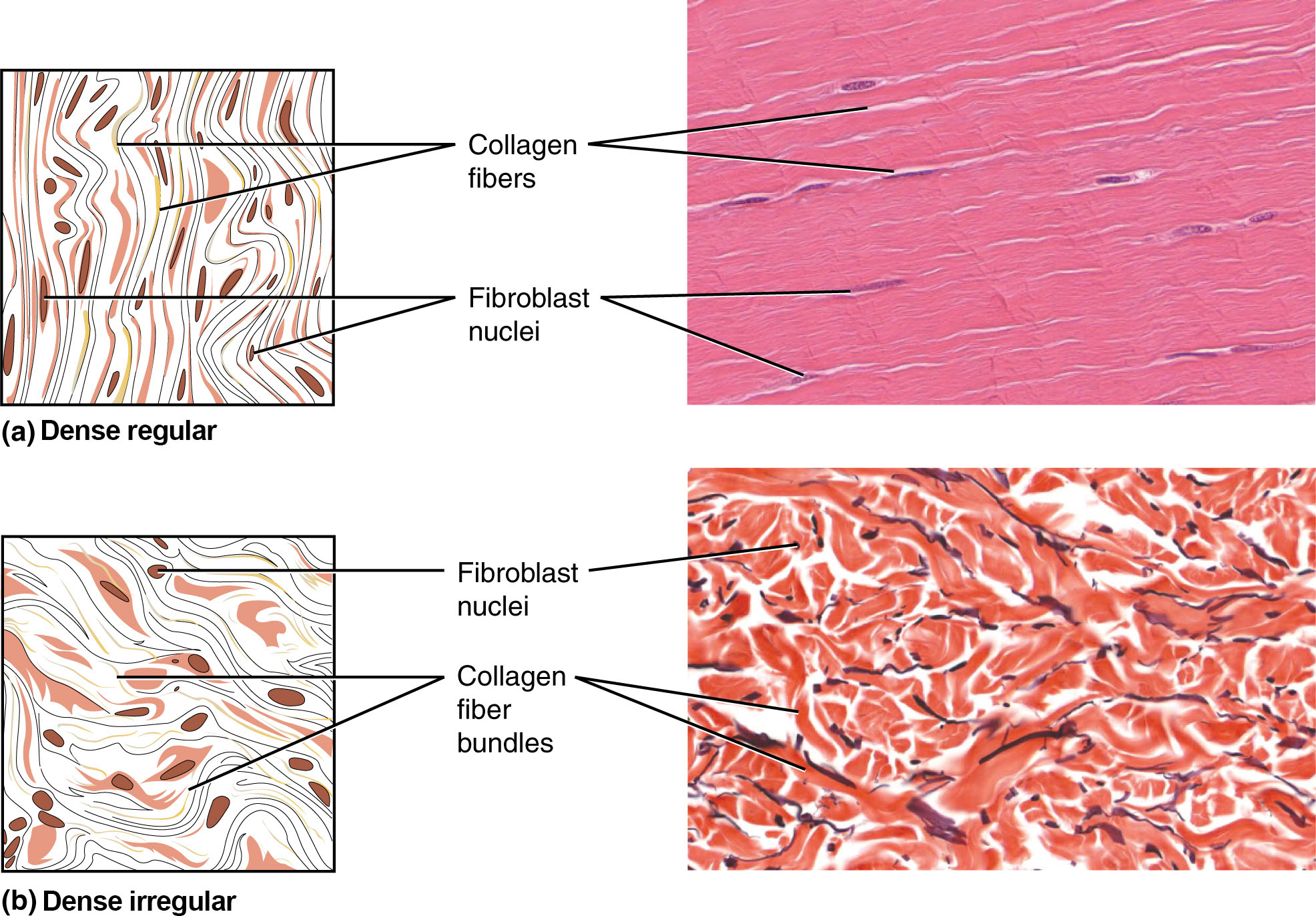
| Dense regular | Parallel collagen bundles | Tendons, ligaments, aponeuroses | High tensile strength in one direction; fibroblasts between bundles |
| Dense irregular | Interwoven collagen in multiple planes | Dermis, organ capsules, periosteum, pericardium | Tensile strength in multiple directions |
| Elastic | Predominantly elastic fibers | Aortic wall, ligamenta flava, vocal cords | Stretch and recoil; Verhoeff/EVG stains |
| Reticular | Type III collagen network | Stroma of lymph nodes, spleen, bone marrow | Scaffold for cellular organs; silver stain |
| Adipose (white) | Minimal fibers | Subcutaneous, mesentery, retroperitoneum | Single large lipid droplet; signet-ring appearance; endocrine (leptin, adiponectin) |
| Adipose (brown) | Minimal fibers | Neonates (interscapular); small amounts in adults | Multilocular lipid droplets; abundant mitochondria (UCP1); thermogenesis |
Ground Substance
The amorphous, gel-like component of ECM composed of glycosaminoglycans (GAGs), proteoglycans, and glycoproteins. GAGs are long, unbranched polysaccharide chains (hyaluronic acid, chondroitin sulfate, heparan sulfate, keratan sulfate, dermatan sulfate) that attract water through their negative charges, creating a hydrated gel that resists compression. Proteoglycans consist of a core protein with covalently attached GAG side chains; aggrecan (the major proteoglycan of cartilage) contains ~100 chondroitin sulfate and keratan sulfate chains. Adhesive glycoproteins (fibronectin, laminin) mediate cell-matrix interactions through integrin receptors.
Lysosomal storage diseases caused by deficiency of enzymes that degrade GAGs. Accumulated GAGs are deposited in connective tissue throughout the body.
| Disease | Enzyme Deficiency | Accumulated GAG | Key Features |
|---|---|---|---|
| Hurler (MPS I) | α-L-iduronidase | Heparan/dermatan sulfate | Corneal clouding, intellectual disability, gargoylism |
| Hunter (MPS II) | Iduronate sulfatase | Heparan/dermatan sulfate | X-linked; no corneal clouding; milder than Hurler |
| Sanfilippo (MPS III) | Various (A–D) | Heparan sulfate | Severe CNS involvement; mild somatic features |
08 Cartilage
Cartilage is an avascular, aneural connective tissue with a firm but flexible ECM. Chondrocytes reside in lacunae and receive nutrition by diffusion through the matrix. Cartilage grows by interstitial growth (mitosis of existing chondrocytes) and appositional growth (new cartilage deposited from perichondrium).
Types of Cartilage
| Type | Matrix | Location | Key Features |
|---|---|---|---|
| Hyaline | Type II collagen + aggrecan | Tracheal rings, nasal septum, articular surfaces, fetal skeleton | Most common; "glassy" matrix; isogenous groups; calcifies with age |
| Elastic | Type II collagen + abundant elastic fibers | External ear (pinna), epiglottis, auditory tube | Flexible; resists repeated bending; never calcifies |
| Fibrocartilage | Type I collagen (predominantly) | Intervertebral discs, pubic symphysis, menisci, TMJ disc | No perichondrium; rows of chondrocytes between dense collagen bundles; resists compression and tension |
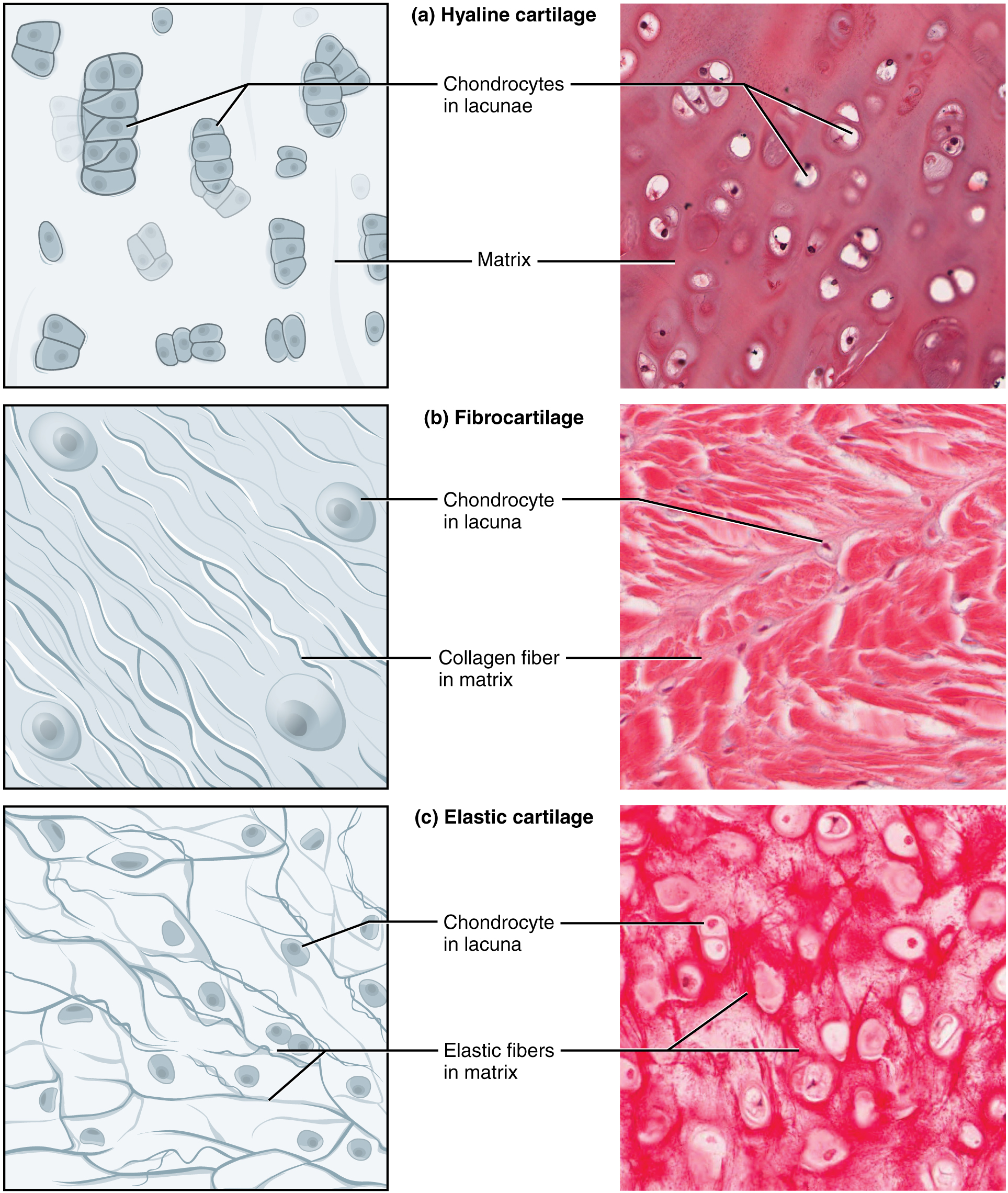
The perichondrium surrounds hyaline and elastic cartilage (but not fibrocartilage or articular cartilage). Its outer fibrous layer contains fibroblasts; the inner chondrogenic layer contains chondroblasts responsible for appositional growth.
Relapsing polychondritis: autoimmune destruction of cartilage (ears, nose, trachea). Osteoarthritis: progressive degradation of articular hyaline cartilage by metalloproteinases (MMP-13). Cartilage has very limited repair capacity due to its avascularity.
Cartilage Matrix Composition
Hyaline cartilage matrix contains type II collagen (providing tensile strength) and aggrecan (a large proteoglycan with chondroitin sulfate and keratan sulfate GAG chains that forms massive aggregates with hyaluronic acid). These aggrecan aggregates trap water, giving cartilage its resilience to compression—a property essential for weight-bearing at articular surfaces. The territorial matrix (pericellular, around lacunae) is rich in proteoglycans and stains darker with basic dyes than the interterritorial matrix between cell clusters. Cartilage matrix is PAS+ and stains with Alcian blue due to its high GAG content.
09 Bone & Ossification
Bone is a mineralized connective tissue providing structural support, mineral homeostasis (calcium/phosphate reservoir), and hematopoiesis. Its matrix is ~35% organic (type I collagen + osteocalcin, osteopontin) and ~65% inorganic (hydroxyapatite crystals, Ca10(PO4)6(OH)2).
Bone Cells
| Cell | Origin | Function | Key Features |
|---|---|---|---|
| Osteoblast | Mesenchymal stem cell | Bone formation; secretes osteoid | Cuboidal; lines bone surfaces; alkaline phosphatase+ |
| Osteocyte | Trapped osteoblast | Mechanotransduction; matrix maintenance | In lacunae; canaliculi connect via gap junctions |
| Osteoclast | Monocyte-macrophage lineage | Bone resorption | Large, multinucleated; ruffled border; Howship lacuna; TRAP+ |
| Bone lining cell | Quiescent osteoblast | Covers inactive surfaces | Flat; can be reactivated |
Compact (Cortical) Bone
The osteon (Haversian system) is the structural unit: concentric lamellae of mineralized matrix surrounding a central Haversian canal containing blood vessels and nerves. Osteocytes in lacunae communicate through canaliculi. Volkmann canals connect adjacent Haversian canals and the periosteal/endosteal surfaces. Interstitial lamellae are remnants of remodeled osteons.
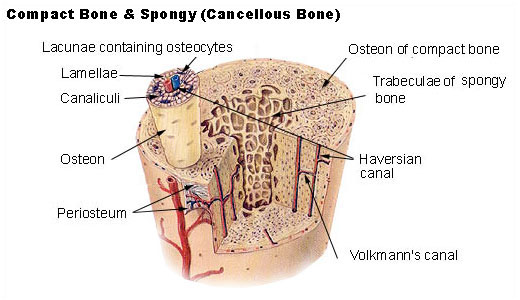
Types of Ossification
| Type | Mechanism | Location |
|---|---|---|
| Intramembranous | Direct bone formation from mesenchyme (no cartilage template) | Flat bones of skull, clavicle, mandible |
| Endochondral | Bone replaces a hyaline cartilage model | Long bones, vertebrae, pelvis, base of skull |
In endochondral ossification, the epiphyseal growth plate is organized into zones (from epiphysis to diaphysis): reserve (resting chondrocytes) → proliferative (mitotic columns) → hypertrophic (enlarged chondrocytes, type X collagen, VEGF secretion) → calcification (matrix mineralizes, chondrocytes undergo apoptosis) → ossification (osteoblasts deposit bone on calcified cartilage scaffolding).
Bone Remodeling & Metabolic Bone Disease
Bone is continuously remodeled by coordinated osteoclast resorption and osteoblast formation. The RANK/RANKL/OPG axis is the master regulator: osteoblasts express RANKL, which binds RANK on osteoclast precursors to promote differentiation and activation. Osteoprotegerin (OPG) is a decoy receptor that inhibits RANKL. PTH increases RANKL expression (stimulates resorption); estrogen increases OPG (inhibits resorption).
| Disease | Mechanism | Histologic Features |
|---|---|---|
| Osteoporosis | Decreased bone mass (resorption > formation) | Thinned trabeculae, widened marrow spaces; normal mineralization |
| Osteomalacia / Rickets | Defective mineralization (vitamin D deficiency) | Thickened osteoid seams on trabecular surfaces (unmineralized matrix) |
| Paget disease | Excessive disordered remodeling | "Mosaic" (jigsaw puzzle) pattern of cement lines; thick trabeculae |
| Osteogenesis imperfecta | Defective type I collagen | Thin cortices, thin trabeculae; woven bone may persist |
10 Blood & Hematopoiesis
Blood is a specialized liquid connective tissue with a fluid ECM called plasma. Formed elements include erythrocytes (RBCs), leukocytes (WBCs), and thrombocytes (platelets). Blood smears stained with Wright-Giemsa (Romanowsky-type) stain are the standard for peripheral blood evaluation.
Formed Elements
| Cell | Size | Nucleus | Key Features | Function |
|---|---|---|---|---|
| Erythrocyte | 7–8 μm | Anucleate | Biconcave disc; central pallor | O2/CO2 transport |
| Neutrophil | 12–15 μm | Multilobed (3–5) | Fine pink granules; Barr body = drumstick | Phagocytosis of bacteria |
| Eosinophil | 12–15 μm | Bilobed | Large red-orange (eosinophilic) granules | Parasites, allergic responses |
| Basophil | 10–12 μm | S-shaped (obscured) | Large dark purple (metachromatic) granules | Histamine, heparin release |
| Lymphocyte | 6–15 μm | Round, dark | Thin rim of blue cytoplasm | Adaptive immunity (B, T, NK) |
| Monocyte | 15–20 μm | Kidney/horseshoe | Largest WBC; grey-blue cytoplasm | Becomes tissue macrophage |
| Platelet | 2–4 μm | Anucleate | Cytoplasmic fragments from megakaryocytes | Hemostasis |
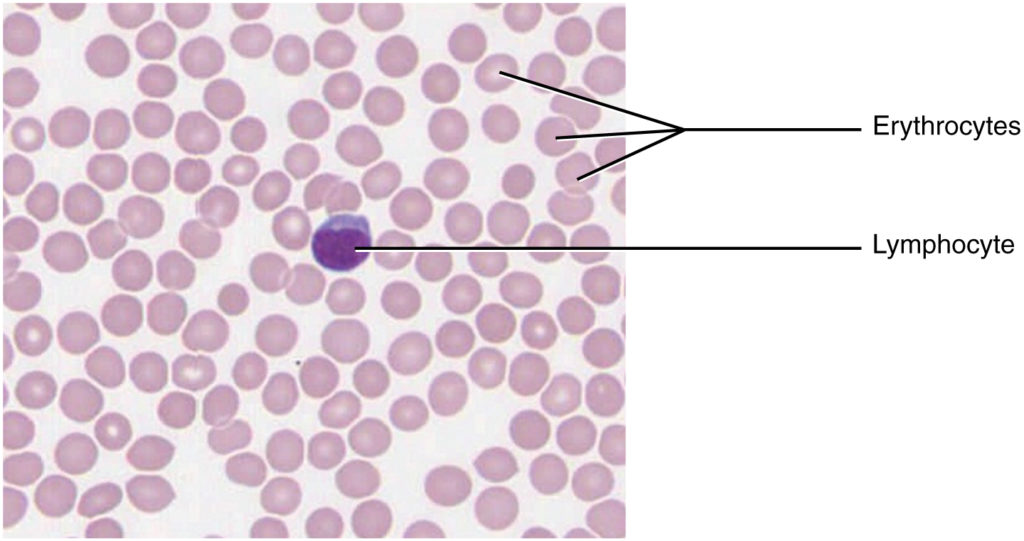
Hematopoiesis
All blood cells derive from hematopoietic stem cells (HSCs) in the bone marrow. HSCs give rise to common myeloid progenitors (erythrocytes, granulocytes, monocytes, megakaryocytes) and common lymphoid progenitors (B cells, T cells, NK cells). Sites of hematopoiesis shift during development: yolk sac (weeks 3–8) → liver/spleen (weeks 6–30) → bone marrow (week 18 onward, exclusive site after birth).
| Location | Name |
|---|---|
| Liver | Kupffer cells |
| Brain | Microglia |
| Bone | Osteoclasts |
| Lung | Alveolar macrophages (dust cells) |
| Skin | Langerhans cells (dendritic cells) |
| Kidney | Mesangial cells (partly) |
| Connective tissue | Histiocytes |
| Spleen / Lymph node | Sinus macrophages |
11 Skeletal Muscle
Skeletal muscle constitutes ~40% of body mass and is responsible for voluntary movement. Fibers are multinucleated syncytia formed by myoblast fusion during development, with peripheral nuclei pushed against the sarcolemma. Fibers are arranged in fascicles, each surrounded by connective tissue sheaths.
Connective Tissue Organization
| Layer | Surrounds | Composition |
|---|---|---|
| Epimysium | Entire muscle | Dense irregular CT |
| Perimysium | Fascicle (bundle of fibers) | CT with vessels and nerves |
| Endomysium | Individual muscle fiber | Reticular fibers + capillaries + satellite cells |
Sarcomere Ultrastructure
The sarcomere (Z-line to Z-line) is the functional contractile unit. Key bands and zones:
| Structure | Composition | Behavior During Contraction |
|---|---|---|
| A band | Full length of thick (myosin) filaments | Constant length |
| I band | Thin (actin) filaments only | Shortens |
| H zone | Thick filaments only (center of A band) | Shortens |
| M line | Myosin-binding proteins at center of H zone | Constant |
| Z disc | α-Actinin anchors thin filaments | Move closer together |
Titin (the largest known protein) spans from Z-disc to M-line, providing passive elasticity and centering thick filaments. Nebulin runs along thin filaments, regulating their length. The triad consists of one T-tubule flanked by two terminal cisternae of sarcoplasmic reticulum, coupling electrical excitation to calcium release.
Fiber Types
| Feature | Type I (Slow Oxidative) | Type IIa (Fast Oxidative) | Type IIb (Fast Glycolytic) |
|---|---|---|---|
| Color | Red (myoglobin-rich) | Red/intermediate | White (pale) |
| Metabolism | Oxidative (aerobic) | Oxidative/glycolytic | Glycolytic (anaerobic) |
| Fatigue resistance | High | Moderate | Low |
| Example | Postural muscles (soleus) | Walking muscles | Sprinting muscles |
Neuromuscular Junction
The motor end plate is the specialized synapse between a motor neuron axon terminal and the skeletal muscle fiber. The presynaptic terminal contains acetylcholine (ACh) vesicles. The synaptic cleft contains acetylcholinesterase (AChE) anchored in the basal lamina. The postsynaptic membrane has junctional folds that concentrate nicotinic ACh receptors (nAChRs) at their crests. Depolarization spreads via T-tubules to trigger calcium release from the sarcoplasmic reticulum.
| Disease | Target | Mechanism | Clinical Features |
|---|---|---|---|
| Myasthenia gravis | Postsynaptic nAChR | Autoantibodies block/destroy receptors | Fatigable weakness; ptosis, diplopia; thymic pathology (75%) |
| Lambert-Eaton | Presynaptic voltage-gated Ca2+ channels | Autoantibodies reduce ACh release | Proximal weakness that improves with use; associated with SCLC |
| Botulism | Presynaptic SNARE proteins | Toxin prevents vesicle fusion/ACh release | Descending flaccid paralysis |
Satellite Cells
Satellite cells are resident muscle stem cells located between the sarcolemma and the basal lamina of skeletal muscle fibers. They are normally quiescent but can be activated by injury to proliferate, differentiate into myoblasts, and fuse with damaged fibers or form new fibers. This is the basis of skeletal muscle regeneration. However, regenerative capacity is limited, and extensive damage leads to fibrosis and fat replacement rather than full restoration. Satellite cells are identified by PAX7 expression.
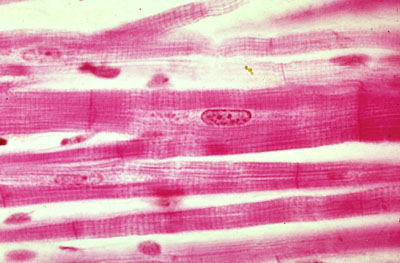
12 Cardiac Muscle
Cardiac muscle shares the striated pattern of skeletal muscle but has critical structural and functional differences. Cardiomyocytes are mononucleated (occasionally binucleated) with centrally located nuclei, branching fibers, and intercalated discs at cell junctions.
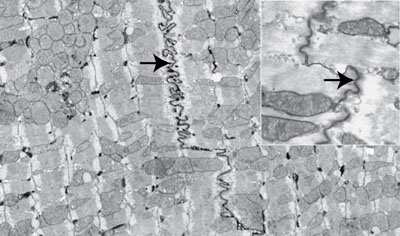
Intercalated Discs
These specialized junctions between cardiomyocytes contain three components:
| Component | Location in Disc | Function |
|---|---|---|
| Fascia adherens | Transverse portion | Anchors actin filaments; like zonula adherens (N-cadherin) |
| Desmosomes | Transverse portion | Resist separation during contraction |
| Gap junctions | Lateral portion | Electrical coupling → functional syncytium (connexin 43) |
The sarcoplasmic reticulum in cardiac muscle is less extensive than in skeletal muscle. T-tubules are wider and located at Z-lines (not A-I junctions as in skeletal muscle), forming dyads (one T-tubule + one SR cisterna) rather than triads. Approximately 20% of calcium for contraction enters from extracellular space through L-type Ca2+ channels (calcium-induced calcium release, CICR), making cardiac contraction dependent on extracellular calcium concentration.
Atrial cardiomyocytes contain atrial natriuretic peptide (ANP) granules visible by electron microscopy. Purkinje fibers are modified cardiomyocytes of the conduction system: they are larger, paler (glycogen-rich), have fewer myofibrils, and conduct impulses rapidly through the ventricular myocardium.
Cardiac Histopathology Timeline
| Time After MI | Gross Appearance | Histologic Finding |
|---|---|---|
| 0–4 hours | Normal or dark mottling | Wavy fibers; early coagulative necrosis |
| 4–24 hours | Dark mottling | Coagulative necrosis; contraction bands; neutrophilic infiltrate begins |
| 1–3 days | Yellow-tan center | Dense neutrophilic infiltrate; karyolysis |
| 3–7 days | Yellow-tan with hyperemic border | Macrophages phagocytose debris; early granulation tissue at margins |
| 1–3 weeks | Red-grey margins | Granulation tissue (capillaries, fibroblasts) replacing necrotic tissue |
| >3 weeks–months | White scar | Dense collagenous scar; no cardiomyocyte regeneration |
Days 3–7 post-MI carry the highest risk of myocardial wall rupture (free wall rupture → cardiac tamponade; papillary muscle rupture → acute mitral regurgitation; ventricular septal rupture → VSD) because this is when necrotic tissue has been softened by neutrophilic and macrophage digestion but granulation tissue has not yet provided structural support.
13 Smooth Muscle
Smooth muscle is the involuntary, non-striated muscle of visceral organs, blood vessels, and the iris. Cells are fusiform (spindle-shaped) with a single central, cigar-shaped nucleus that may appear corkscrew-shaped in contracted cells on longitudinal section.
Key Features
Smooth muscle lacks sarcomeres, troponin, and T-tubules. Contraction is regulated by calmodulin/MLCK (myosin light chain kinase) rather than troponin. Calcium binds calmodulin → activates MLCK → phosphorylates myosin light chain → cross-bridge cycling. Relaxation requires myosin light chain phosphatase (MLCP). Dense bodies (analogous to Z-discs) anchor actin filaments to the cytoskeleton and cell membrane. Caveolae serve as rudimentary T-tubule equivalents.
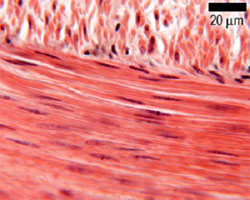
Comparison of Muscle Types
| Feature | Skeletal | Cardiac | Smooth |
|---|---|---|---|
| Striations | Yes | Yes | No |
| Nuclei | Multiple, peripheral | 1–2, central | 1, central |
| Control | Voluntary (somatic) | Involuntary (autonomic) | Involuntary (autonomic, hormonal) |
| Intercalated discs | Absent | Present | Absent |
| T-tubules | At A-I junction (triads) | At Z-line (dyads) | Absent (caveolae) |
| Ca2+ regulation | Troponin C | Troponin C + CICR | Calmodulin/MLCK |
| Regeneration | Limited (satellite cells) | Very limited | Good (mitotic capacity) |
Leiomyomas (fibroids) are benign smooth muscle tumors; the uterine leiomyoma is the most common tumor in women. Leiomyosarcoma is the malignant counterpart. Histologically, smooth muscle tumors are desmin+ and smooth muscle actin (SMA)+.
14 Neurons & Synapses
Neurons are electrically excitable cells that transmit information through electrical impulses and chemical synapses. They are post-mitotic in the adult (with limited exceptions in the hippocampus and olfactory bulb). Each neuron has a cell body (soma/perikaryon), dendrites (receive input), and an axon (conducts output).
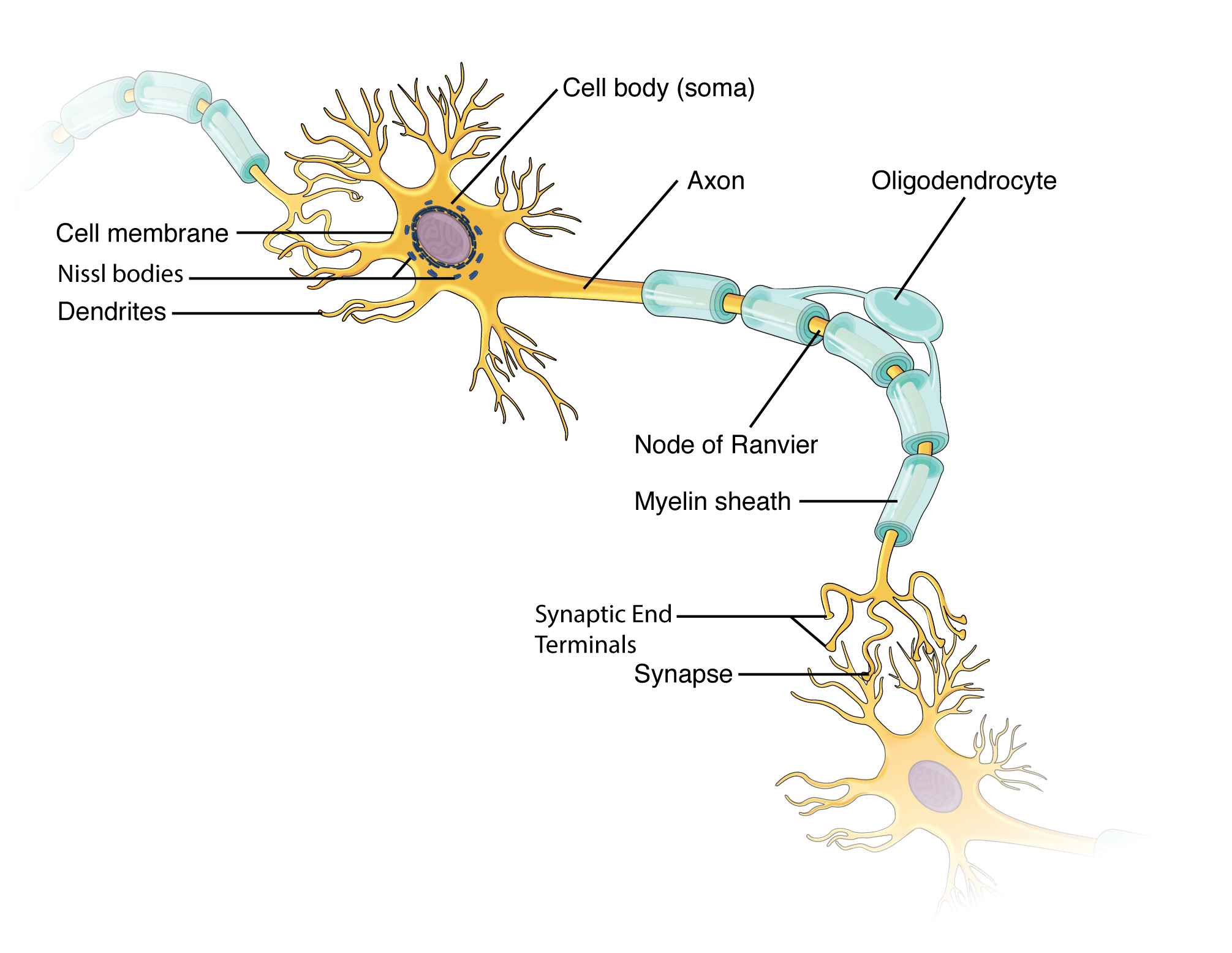
Neuronal Classification
| By Morphology | Description | Example |
|---|---|---|
| Multipolar | Multiple dendrites, one axon | Motor neurons, most CNS interneurons |
| Bipolar | One dendrite, one axon | Retinal bipolar cells, olfactory neurons |
| Pseudounipolar | Single process that bifurcates | Dorsal root ganglion sensory neurons |
| Anaxonic | No identifiable axon | Retinal amacrine cells, some CNS interneurons |
Cellular Features
The neuronal soma contains a large, pale, euchromatic nucleus with a prominent nucleolus. Nissl substance (rough ER + polyribosomes) appears as basophilic clumps in the soma and dendrites but is absent from the axon hillock and axon. Neurofilaments (intermediate filaments specific to neurons) provide structural support and are stained by silver impregnation. Lipofuscin pigment accumulates with age as "wear-and-tear" pigment from lysosomal activity.
Synapses
Chemical synapses contain a presynaptic terminal (bouton) with neurotransmitter-filled synaptic vesicles, a synaptic cleft (~20 nm), and a postsynaptic membrane with receptor proteins. Axodendritic synapses are most common; axosomatic and axoaxonic types also occur. Electrical synapses (gap junctions) allow direct ion flow between neurons and are found in cardiac and smooth muscle for synchronization.
Axonal Transport
Axonal transport is essential for moving organelles, vesicles, and proteins between the soma and axon terminals. Anterograde transport (soma → terminal) uses kinesin motors on microtubules; fast anterograde (~400 mm/day) carries membrane-bound organelles and vesicles, while slow anterograde (~1–5 mm/day) carries cytoskeletal components and soluble enzymes. Retrograde transport (terminal → soma) uses dynein motors (~200 mm/day); carries endosomes, growth factors (e.g., NGF), and also transports viruses (rabies, herpes) and toxins (tetanus) to the cell body.
Neurotransmitter Types & Vesicle Morphology
| Vesicle Type (EM) | Size | Contents | Examples |
|---|---|---|---|
| Small, clear | 40–60 nm | Classic neurotransmitters | ACh, glutamate, GABA, glycine |
| Small, dense-core | 40–60 nm | Catecholamines | Norepinephrine, dopamine, serotonin |
| Large, dense-core | 90–250 nm | Neuropeptides | Substance P, enkephalins, VIP |
| Inclusion | Composition | Disease |
|---|---|---|
| Lewy body | α-Synuclein | Parkinson disease, Lewy body dementia |
| Neurofibrillary tangle | Hyperphosphorylated tau | Alzheimer disease |
| Pick body | Tau (3-repeat) | Pick disease (frontotemporal dementia) |
| Bunina body | Cystatin C | ALS |
| Hirano body | Actin, actin-associated proteins | Alzheimer disease |
15 Glial Cells & Myelination
Glial cells (neuroglia) outnumber neurons and provide structural support, metabolic maintenance, and insulation. Unlike neurons, most glial cells retain mitotic capacity, making them the most common source of primary CNS tumors.
CNS Glial Cells
| Cell Type | Function | Key Features | Marker |
|---|---|---|---|
| Astrocyte | BBB maintenance, ion/NT homeostasis, scar formation | Star-shaped; foot processes on capillaries; most abundant glia | GFAP |
| Oligodendrocyte | CNS myelination (one cell myelinates multiple axons) | Round nucleus; perineuronal satellites | MBP, PLP |
| Microglia | Phagocytosis, immune defense | Mesoderm (monocyte) origin; small, dark, elongated nuclei | CD68, Iba-1 |
| Ependymal cells | Line ventricles; CSF circulation | Simple cuboidal/columnar; cilia on apical surface | — |
PNS Glial Cells
| Cell Type | Function | Key Features |
|---|---|---|
| Schwann cell | PNS myelination (one cell per internode) | Neural crest origin; supports regeneration |
| Satellite cell | Surrounds neuronal cell bodies in ganglia | Analogous to astrocytes in CNS |
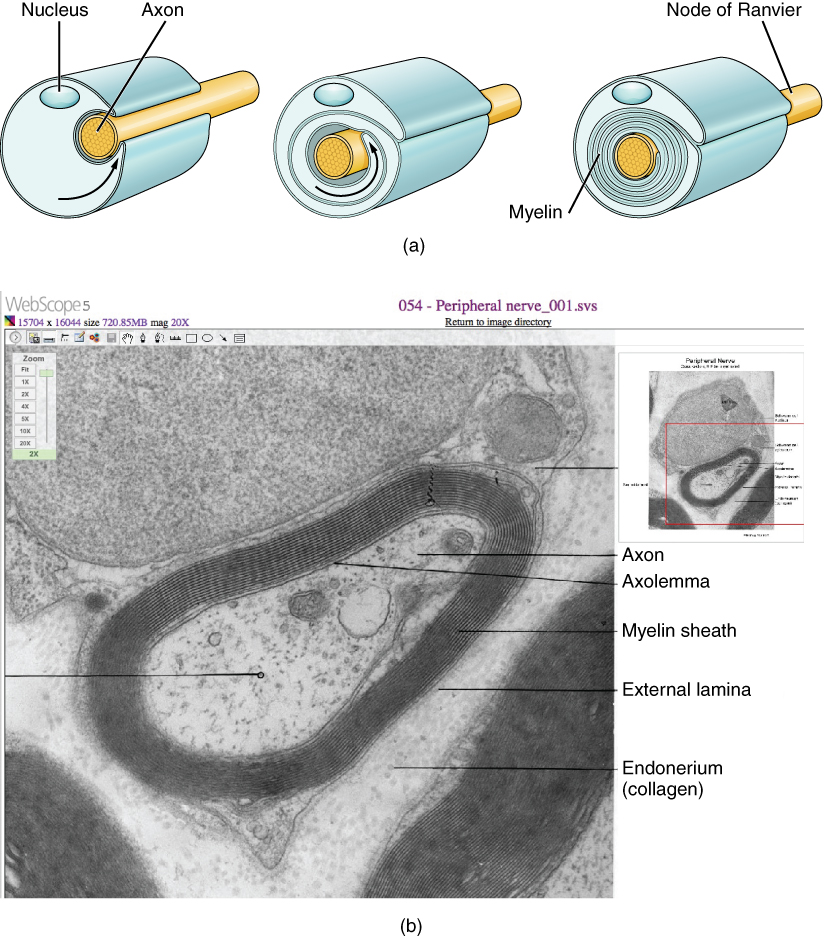
Myelination
Myelin is a lipid-rich membrane that wraps around axons, increasing conduction velocity through saltatory conduction (action potentials jump between nodes of Ranvier). In the CNS, each oligodendrocyte myelinates segments of up to 30–60 axons. In the PNS, each Schwann cell myelinates a single internode (~1 mm) of one axon. Unmyelinated axons in the PNS are enclosed in Schwann cell cytoplasm (Remak bundles) without wrapping.
| Feature | CNS Myelination | PNS Myelination |
|---|---|---|
| Cell | Oligodendrocyte | Schwann cell |
| Axons per cell | Up to 30–60 | 1 internode of 1 axon |
| Regeneration | Poor | Good (Schwann cell guides regrowth) |
| Demyelinating disease | Multiple sclerosis | Guillain-Barré syndrome |
| Tumor | Oligodendroglioma | Schwannoma |
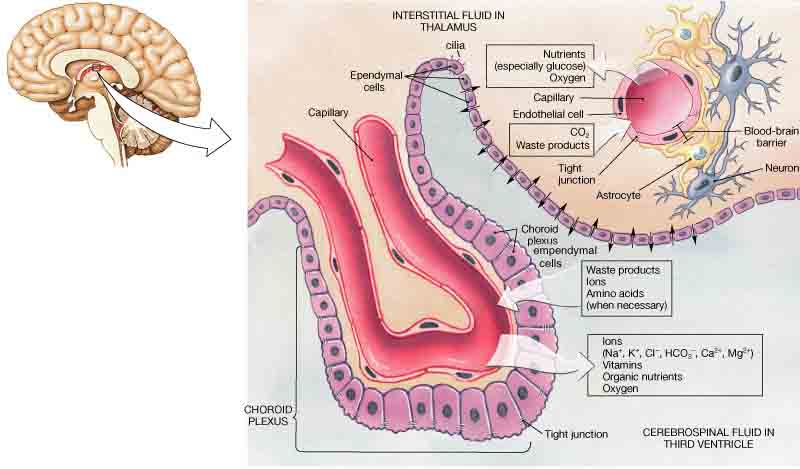
Blood-Brain Barrier
The blood-brain barrier (BBB) is formed by three components: (1) continuous capillary endothelium with tight junctions (claudin-5, occludin), (2) pericytes embedded in the capillary basement membrane, and (3) astrocyte foot processes (perivascular end-feet) covering >99% of the capillary surface. The BBB restricts paracellular diffusion and requires specific transporters for glucose (GLUT1), amino acids, and other essential molecules. Circumventricular organs (area postrema, median eminence, posterior pituitary, pineal gland) lack a BBB, featuring fenestrated capillaries that allow direct hormonal sensing and secretion.
CNS Reactions to Injury
| Response | Cell Involved | Histologic Finding | Significance |
|---|---|---|---|
| Gliosis (astrocytic scar) | Reactive astrocytes | Increased GFAP+ processes; gemistocytic astrocytes | CNS equivalent of fibrosis; permanent scar |
| Microglial activation | Microglia | Rod cells, microglial nodules, neuronophagia | Viral encephalitis, neurodegeneration |
| Demyelination | Oligodendrocyte loss | Loss of myelin staining (Luxol fast blue); preserved axons | MS, PML, ADEM |
| Wallerian degeneration | Axon + myelin distal to injury | Myelin ovoids, macrophage phagocytosis, Schwann cell proliferation (PNS) | Peripheral nerve injury; basis for regeneration in PNS |
16 Peripheral Nerve & Ganglia
Peripheral nerves have a connective tissue organization analogous to skeletal muscle:
| Layer | Surrounds | Special Features |
|---|---|---|
| Epineurium | Entire nerve | Dense irregular CT; outermost layer |
| Perineurium | Fascicle | Concentric layers of flat epithelioid cells; forms blood-nerve barrier; most important for surgical repair |
| Endoneurium | Individual nerve fiber | Loose CT with capillaries; contains Schwann cells |
Ganglia
Sensory (dorsal root / cranial) ganglia: contain pseudounipolar neurons with large, round, pale cell bodies surrounded by satellite cells. No synapses occur here. Autonomic ganglia: contain multipolar neurons; synapses occur between pre- and postganglionic neurons. Sympathetic chain ganglia and parasympathetic terminal ganglia (e.g., myenteric plexus, submucosal plexus) are clinically important.
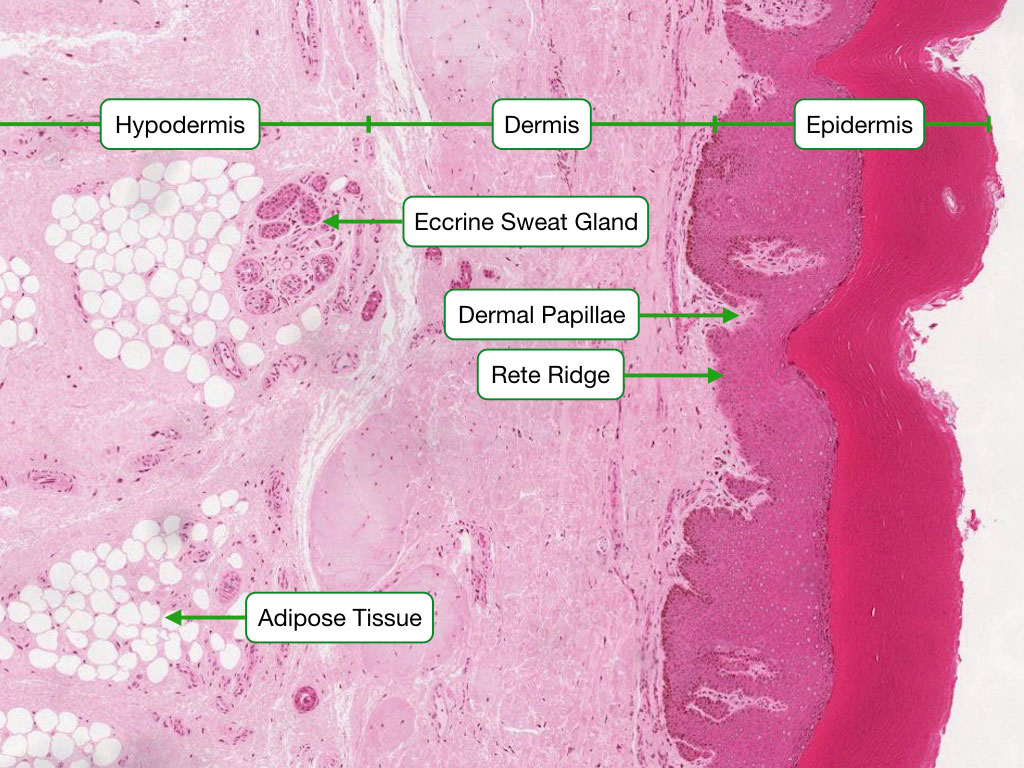
17 Skin & Appendages
The skin is the largest organ, composed of epidermis (keratinized stratified squamous epithelium) and dermis (connective tissue), overlying the hypodermis (subcutaneous fat, not technically part of the skin).
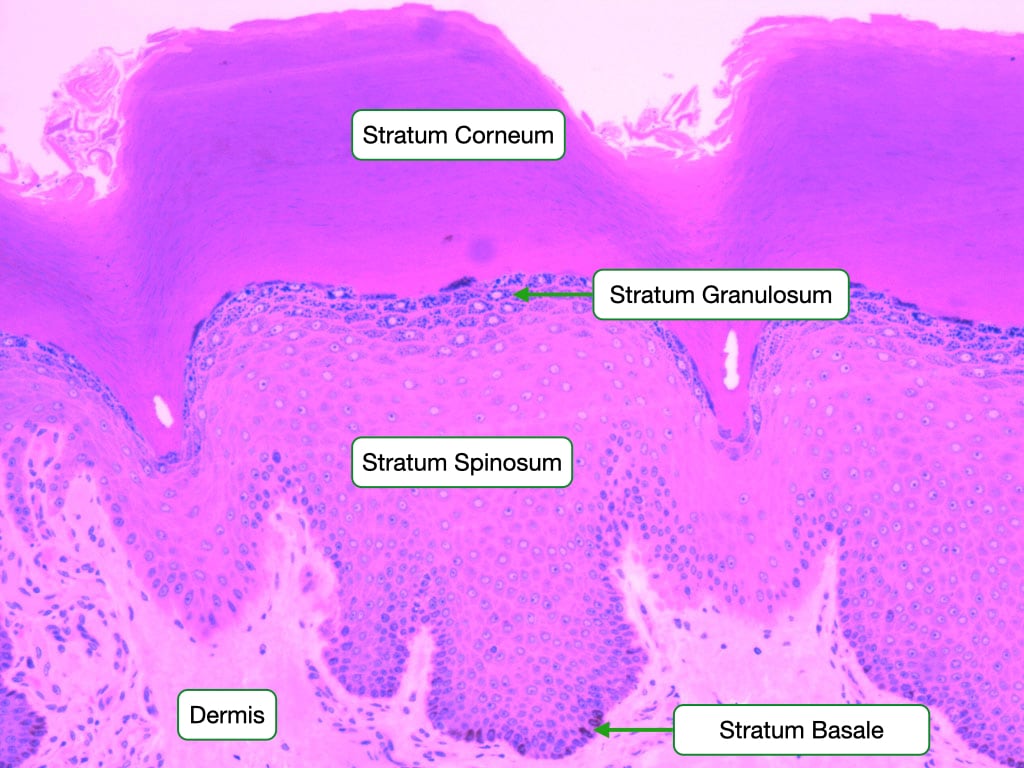
Epidermal Layers (Thick Skin, Deep to Superficial)
| Layer | Key Features |
|---|---|
| Stratum basale (germinativum) | Single layer of columnar cells on basement membrane; stem cells, melanocytes, Merkel cells; mitotically active |
| Stratum spinosum | Multiple layers; "spiny" appearance (desmosomes); Langerhans cells; keratinocytes begin keratin synthesis |
| Stratum granulosum | 2–5 layers; keratohyalin granules (profilaggrin); lamellar bodies release lipids (water barrier) |
| Stratum lucidum | Thin, clear layer; only in thick skin (palms, soles); eleidin |
| Stratum corneum | 15–20+ layers of dead, anucleate, keratin-filled squames; major barrier |
Epidermal Cell Types
| Cell | Origin | Location | Function |
|---|---|---|---|
| Keratinocyte | Ectoderm | All layers (95% of cells) | Keratin production; barrier |
| Melanocyte | Neural crest | Stratum basale | Melanin synthesis; UV protection |
| Langerhans cell | Bone marrow | Stratum spinosum | Antigen presentation (dendritic cell) |
| Merkel cell | Neural crest | Stratum basale | Mechanoreceptor (fine touch) |
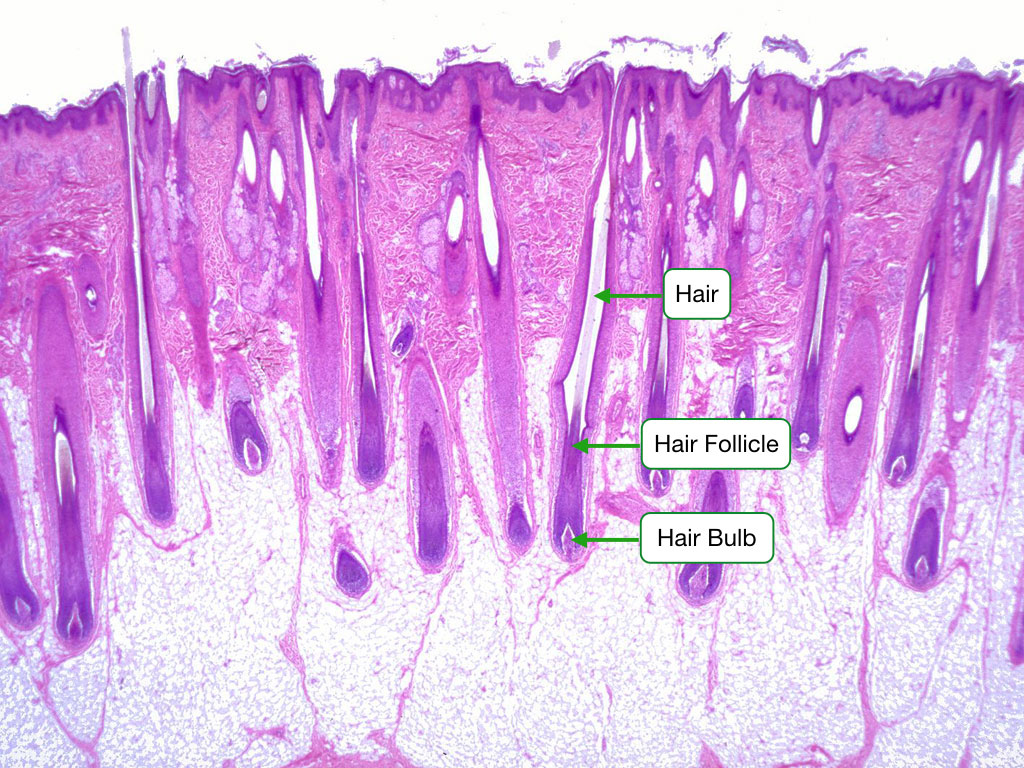
Skin Appendages
Hair follicles: dermal papilla at base provides inductive signals; matrix cells proliferate and keratinize. Sebaceous glands: holocrine secretion of sebum; associated with hair follicles. Eccrine sweat glands: merocrine secretion; coiled gland in dermis with duct to skin surface; thermoregulation; most of body. Apocrine sweat glands: axilla, groin; open into hair follicle; odorless secretion metabolized by bacteria to produce odor.
Dermis
The dermis has two layers. The papillary dermis is superficial loose connective tissue that interdigitates with the epidermis via dermal papillae, containing capillary loops and sensory receptors (Meissner corpuscles for fine touch). The reticular dermis is deep dense irregular connective tissue (type I collagen and elastic fibers) that provides tensile strength. Langer lines (cleavage lines) reflect the predominant collagen fiber orientation; surgical incisions along Langer lines produce less scarring.
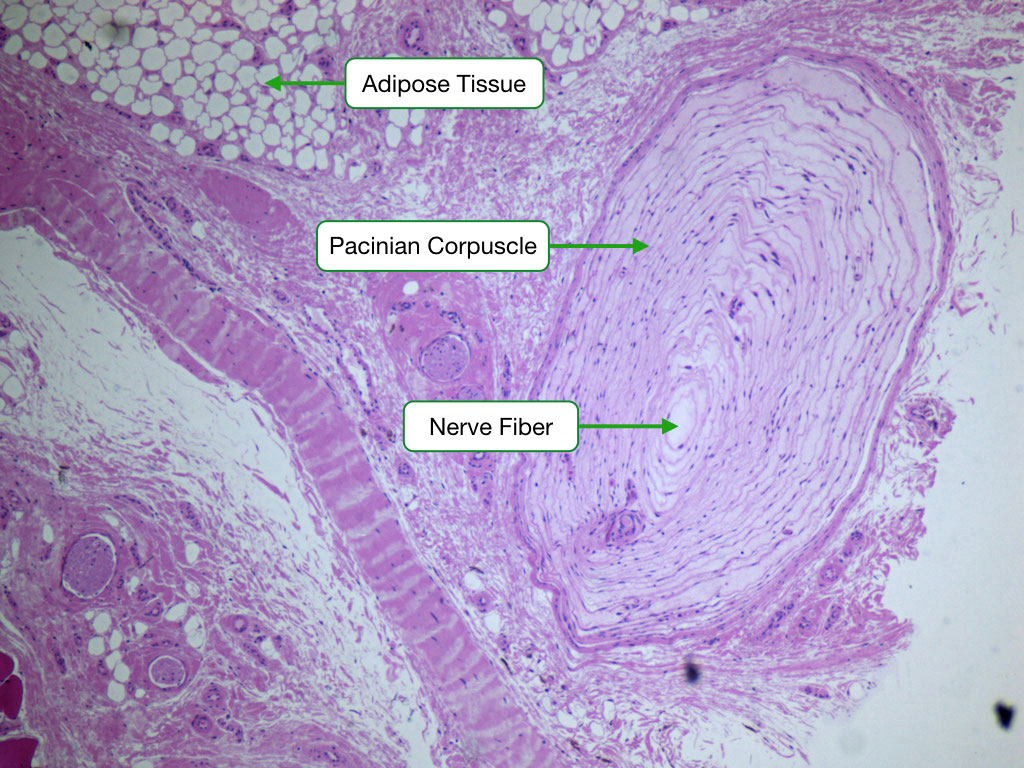
Sensory Receptors in Skin
| Receptor | Type | Location | Modality |
|---|---|---|---|
| Free nerve endings | Unencapsulated | Epidermis, dermis | Pain, temperature, crude touch |
| Meissner corpuscle | Encapsulated | Dermal papillae (glabrous skin) | Fine/discriminative touch; rapidly adapting |
| Pacinian corpuscle | Encapsulated (onion-like lamellae) | Deep dermis, subcutis, mesentery | Pressure, vibration; rapidly adapting |
| Ruffini corpuscle | Encapsulated | Deep dermis, joint capsules | Stretch, pressure; slowly adapting |
| Merkel disc | Unencapsulated | Stratum basale | Sustained pressure, texture; slowly adapting |
Melanocytes transfer melanin-containing melanosomes to surrounding keratinocytes (each melanocyte supplies ~36 keratinocytes, forming the epidermal melanin unit). Melanin production occurs in melanosomes via tyrosinase (tyrosine → DOPA → dopaquinone → melanin). Differences in skin color are due to melanosome size, number, distribution, and degradation rate—not melanocyte number. Albinism results from defective melanin synthesis (tyrosinase mutations in oculocutaneous albinism type 1) despite normal melanocyte numbers.
18 Gastrointestinal Tract
The GI tract follows a consistent four-layer plan from esophagus to anal canal: mucosa (epithelium + lamina propria + muscularis mucosae), submucosa, muscularis externa (inner circular + outer longitudinal smooth muscle), and serosa/adventitia.
Regional Epithelial Variation
| Region | Epithelium | Distinguishing Features |
|---|---|---|
| Esophagus | Non-keratinized stratified squamous | Submucosal glands (mucous); muscularis: upper 1/3 skeletal, middle mixed, lower 1/3 smooth |
| Stomach | Simple columnar (surface mucous cells) | Gastric pits + glands; rugae; no villi, no goblet cells |
| Small intestine | Simple columnar with goblet cells | Villi (tallest in duodenum); crypts of Lieberkuhn; Paneth cells at crypt base |
| Large intestine | Simple columnar with abundant goblet cells | No villi; straight tubular crypts; goblet cells increase distally |
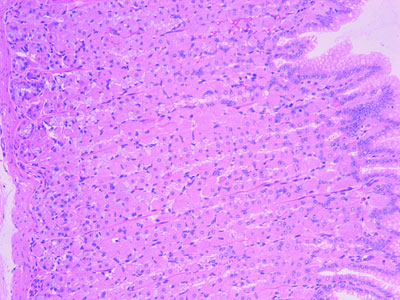
Gastric Gland Cells
| Cell | Location | Product | Key Feature |
|---|---|---|---|
| Mucous neck cell | Neck of gland | Soluble mucus | Pale, granular |
| Parietal (oxyntic) cell | Upper gland | HCl, intrinsic factor | Large, eosinophilic; intracellular canaliculi; tubulovesicular system; H+/K+-ATPase |
| Chief (zymogenic) cell | Base of gland | Pepsinogen, lipase | Basophilic basal (RER), apical granules |
| Enteroendocrine cells | Throughout | Gastrin (G cell), somatostatin (D cell), histamine (ECL cell) | Small; clear cytoplasm on H&E |
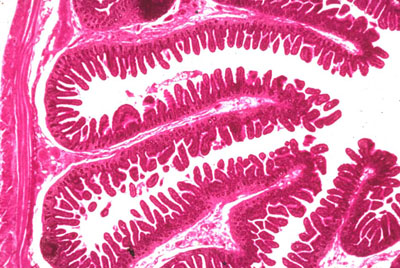
Small Intestine Specializations
Duodenum: Brunner glands in submucosa (alkaline mucus, bicarbonate). Jejunum: tallest plicae circulares, longest villi. Ileum: Peyer patches (aggregated lymphoid follicles) in submucosa; M cells overlie follicles for antigen sampling. Paneth cells at crypt bases secrete defensins and lysozyme (innate immunity); they contain large eosinophilic apical granules.
Submucosal (Meissner) plexus: in submucosa; regulates glandular secretion and mucosal blood flow. Myenteric (Auerbach) plexus: between inner circular and outer longitudinal muscle; regulates motility. Hirschsprung disease results from failure of neural crest cell migration → absent ganglia in affected segment → functional obstruction.
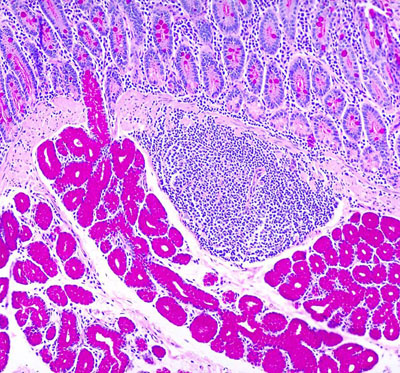
Appendix
The appendix shares the four-layer GI wall plan but is notable for abundant lymphoid tissue in the lamina propria and submucosa, often with prominent germinal centers. The lumen is small and irregular. Obliteration of the lumen by lymphoid tissue or a fecalith predisposes to acute appendicitis. Histologically, transmural neutrophilic infiltration and necrosis are the hallmarks of acute appendicitis. Carcinoid tumors are the most common appendiceal neoplasm.
Anal Canal Transition
The dentate (pectinate) line marks the transition from simple columnar epithelium (above, endoderm-derived) to stratified squamous epithelium (below, ectoderm-derived). Above the line: internal hemorrhoidal plexus (portal drainage), visceral innervation (insensitive to pain). Below the line: external hemorrhoidal plexus (caval drainage), somatic innervation (sensitive to pain). This is a high-yield anatomic-histologic correlation.
GI Endocrine Cells
| Cell | Location | Hormone | Function |
|---|---|---|---|
| G cell | Gastric antrum | Gastrin | Stimulates parietal cell HCl secretion |
| ECL cell | Gastric fundus/body | Histamine | Stimulates parietal cells (paracrine) |
| D cell | Throughout GI | Somatostatin | Inhibits acid and hormone secretion |
| I cell | Duodenum, jejunum | CCK | Gallbladder contraction, pancreatic enzyme secretion |
| S cell | Duodenum | Secretin | Bicarbonate secretion from pancreatic ducts |
| K cell | Duodenum, jejunum | GIP (glucose-dependent insulinotropic peptide) | Stimulates insulin release; incretin effect |
| L cell | Ileum, colon | GLP-1, PYY | Incretin effect; satiety |
| EC cell | Throughout GI | Serotonin (5-HT) | Motility regulation; most common GI endocrine cell |
19 Liver, Gallbladder & Pancreas
Liver
The liver is organized into hepatic lobules: hexagonal units centered on a central vein with portal triads (hepatic artery, portal vein, bile duct) at the periphery. Hepatocytes are arranged in anastomosing plates separated by sinusoids (fenestrated capillaries lined by endothelial cells lacking a continuous basement membrane). The space of Disse lies between hepatocytes and sinusoidal endothelium, containing hepatic stellate cells (Ito cells) that store vitamin A and, when activated, produce collagen (cirrhosis).
The portal lobule (centered on portal triad, draining bile) and hepatic acinus (of Rappaport; functional unit based on blood flow zones 1–3) are alternative organizational models. Zone 1 (periportal) receives blood first (most oxygenated); zone 3 (pericentral) is most susceptible to ischemic injury and is the site of CYP450 drug metabolism.
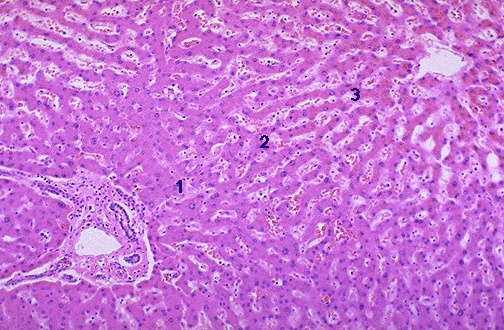
Hepatic Zonation & Pathology
| Zone | Blood Supply | Metabolic Activity | Pathologic Susceptibility |
|---|---|---|---|
| Zone 1 (periportal) | First to receive O2 and nutrients | Gluconeogenesis, beta-oxidation, bile acid secretion, urea synthesis | Viral hepatitis (earliest injury); phosphorus poisoning; eclampsia |
| Zone 2 (intermediate) | Intermediate | Intermediate functions | Yellow fever (midzonal necrosis) |
| Zone 3 (pericentral/centrilobular) | Last to receive O2 | Glycolysis, lipogenesis, CYP450 metabolism, glycogen synthesis | Ischemia (shock liver), acetaminophen toxicity, alcohol (steatosis), CCl4 poisoning, CHF (nutmeg liver) |
Gallbladder
Lined by simple columnar epithelium with prominent mucosal folds (rugae). It concentrates bile by absorbing water and ions. Key features: no submucosa, no muscularis mucosae; the wall has mucosa, muscularis (smooth muscle), and serosa/adventitia. Rokitansky-Aschoff sinuses are mucosal herniations into the muscular wall, prominent in chronic cholecystitis.
Pancreas
The exocrine pancreas (~98% of mass) consists of serous acini with centroacinar cells (unique feature; pale cells within the acinar lumen that begin the duct system and secrete bicarbonate). The islets of Langerhans (endocrine, ~2%) are pale clusters of cells with fenestrated capillaries: alpha cells (glucagon, peripheral), beta cells (insulin, central, ~70%), delta cells (somatostatin), and PP cells (pancreatic polypeptide).
Bile Canaliculi & Bile Flow
Bile is secreted into bile canaliculi—tiny channels formed by grooves in adjacent hepatocyte membranes sealed by tight junctions. Bile flows in the opposite direction to blood: from zone 3 (pericentral) toward zone 1 (periportal), draining into canals of Hering (lined partly by hepatocytes, partly by cholangiocytes), then interlobular bile ducts in the portal triads, and ultimately the common bile duct. Cholangiocytes (bile duct epithelial cells) modify bile composition by secreting bicarbonate and absorbing water. They are the target of autoimmune destruction in primary biliary cholangitis (PBC; anti-mitochondrial antibodies) and primary sclerosing cholangitis (PSC; concentric "onion-skin" periductal fibrosis).
Kupffer Cells & Sinusoidal Endothelium
Kupffer cells are liver-resident macrophages within sinusoidal lumens that filter portal blood, removing bacteria, endotoxin, and senescent red blood cells. They are CD68+ and PAS-diastase+. The sinusoidal endothelium is fenestrated without a conventional basement membrane, allowing direct plasma contact with hepatocytes through the space of Disse. This unique configuration facilitates the liver's metabolic, synthetic, and detoxification functions.
20 Kidney & Urinary Tract
Each kidney contains ~1 million nephrons, the functional units responsible for filtration, reabsorption, secretion, and concentration. The nephron comprises the renal corpuscle (glomerulus + Bowman capsule) and a tubular system.
Nephron Segments
| Segment | Epithelium | Key Features | Function |
|---|---|---|---|
| Bowman capsule | Simple squamous (parietal); podocytes (visceral) | Filtration slits with nephrin | Ultrafiltration |
| Proximal convoluted tubule | Simple cuboidal with brush border | Eosinophilic; prominent microvilli; basal striations (mitochondria) | Reabsorbs ~65% filtrate |
| Thin limb of Henle | Simple squamous | Resembles capillary | Water (descending) or solute (ascending) movement |
| Thick ascending limb | Simple cuboidal (no brush border) | Macula densa at distal end | NaCl reabsorption (NKCC2) |
| Distal convoluted tubule | Simple cuboidal (no brush border) | Shorter, paler than PCT; fewer cells per cross-section | NaCl reabsorption (NCC); Ca2+ (PTH) |
| Collecting duct | Simple cuboidal to columnar | Principal cells (pale) and intercalated cells (dark) | Water (ADH); acid-base balance |
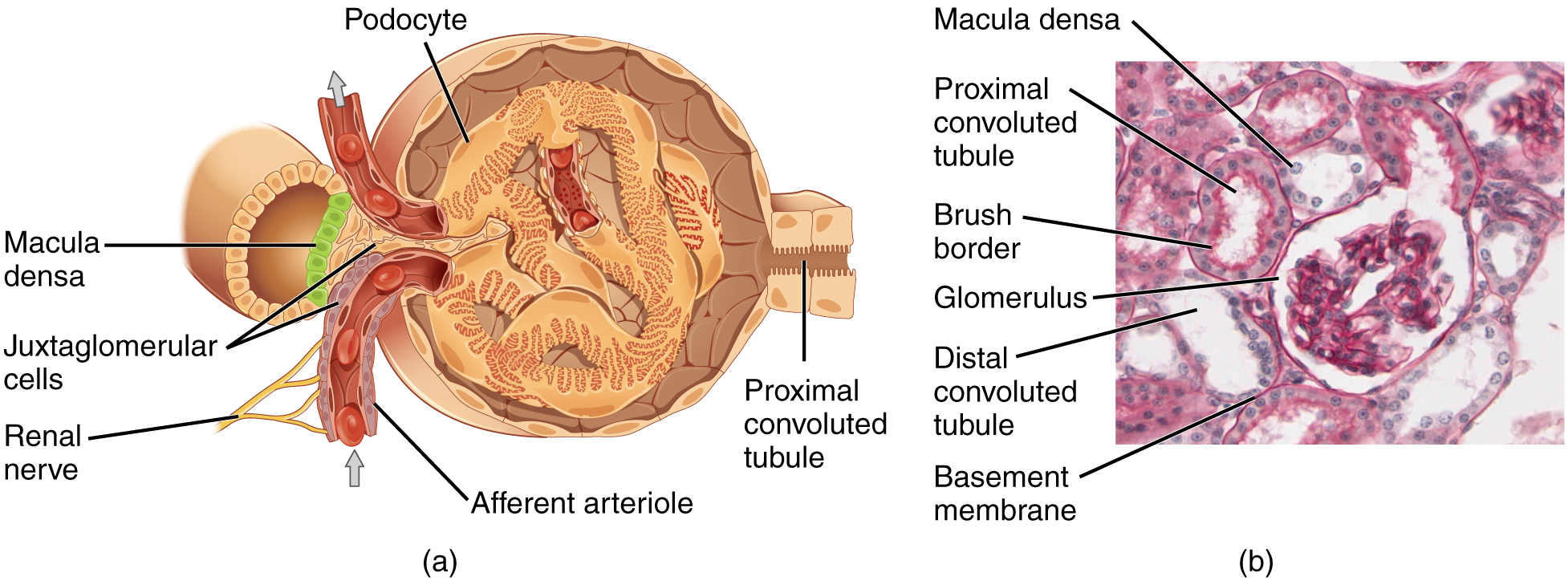
Juxtaglomerular Apparatus
The JGA comprises juxtaglomerular (JG) cells (modified smooth muscle in afferent arteriole; secrete renin), macula densa (specialized cells in thick ascending limb/early DCT; sense NaCl concentration), and extraglomerular mesangial cells (lacis cells). This apparatus regulates glomerular filtration rate and blood pressure via the renin-angiotensin-aldosterone system.
Minimal change disease: effacement of podocyte foot processes (visible only on electron microscopy); most common nephrotic syndrome in children. Diabetic nephropathy: thickening of glomerular basement membrane, mesangial expansion, Kimmelstiel-Wilson nodules (nodular glomerulosclerosis).
Renal Interstitium & Vasculature
The renal interstitium in the medulla contains interstitial cells that produce prostaglandins and erythropoietin. The vasa recta are thin-walled vessels that descend into the medulla alongside the loops of Henle, functioning as countercurrent exchangers to maintain the medullary osmotic gradient. The peritubular capillaries in the cortex arise from efferent arterioles and surround the tubules, enabling tubular reabsorption and secretion.
Ureter & Bladder
The ureter is lined by transitional epithelium (urothelium) and has two smooth muscle layers (inner longitudinal, outer circular) that reverse in the distal third to three layers (inner longitudinal, middle circular, outer longitudinal). The bladder has urothelium with umbrella cells, a thick lamina propria, and three smooth muscle layers (detrusor muscle) arranged in an interlacing pattern. Staging of bladder cancer depends on depth of invasion through these layers: CIS (urothelium only) → T1 (lamina propria) → T2 (muscularis propria/detrusor) → T3 (perivesical fat) → T4 (adjacent organs).
21 Respiratory System
The respiratory system transitions from the conducting zone (nasal cavity to terminal bronchioles) to the respiratory zone (respiratory bronchioles, alveolar ducts, alveolar sacs, alveoli) where gas exchange occurs.
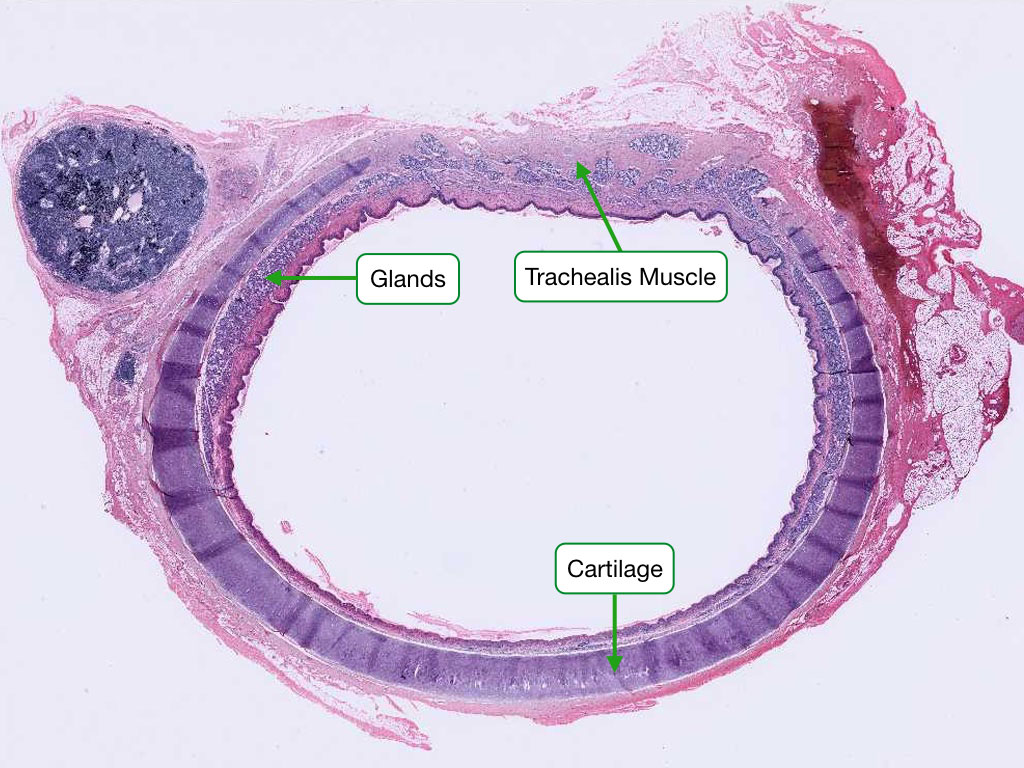
Airway Epithelial Changes
| Region | Epithelium | Support |
|---|---|---|
| Trachea / Primary bronchi | Pseudostratified ciliated columnar with goblet cells | C-shaped hyaline cartilage rings |
| Secondary/tertiary bronchi | Pseudostratified ciliated columnar | Irregular cartilage plates; smooth muscle prominent |
| Bronchioles | Simple ciliated columnar/cuboidal; no goblet cells | No cartilage; smooth muscle, elastic fibers |
| Terminal bronchioles | Simple cuboidal; Clara (club) cells | Smooth muscle; no cartilage |
| Respiratory bronchioles | Simple cuboidal; scattered alveoli | Transition zone |
| Alveoli | Type I (95% surface) + Type II pneumocytes | Elastic fibers |
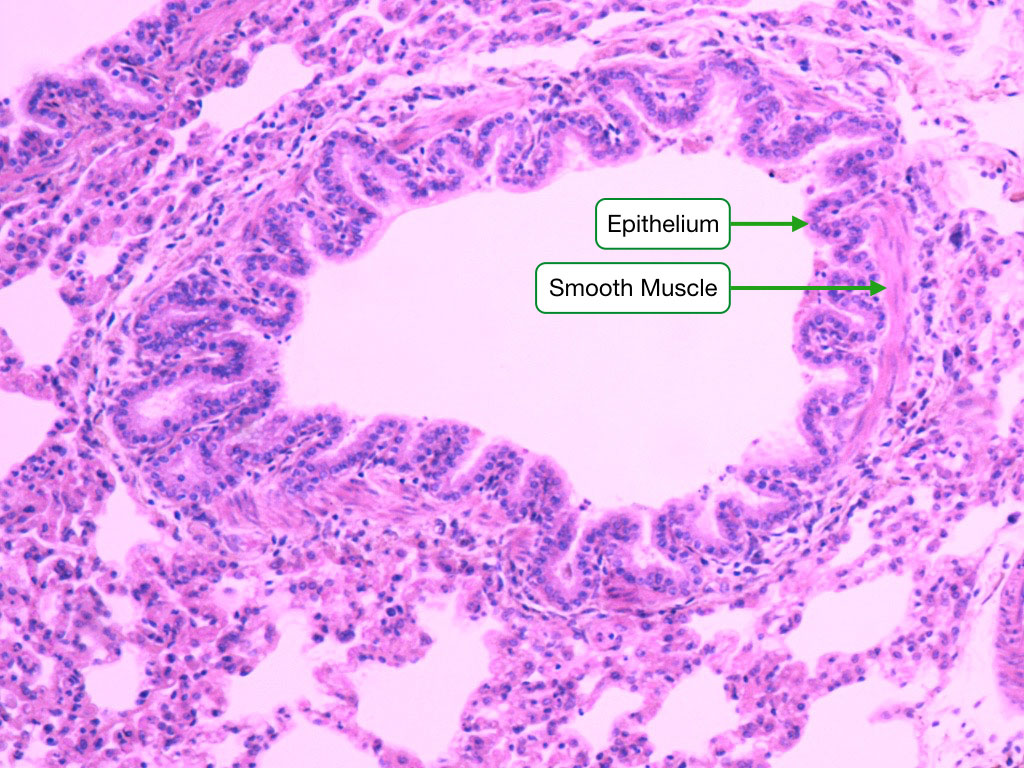
Alveolar Cell Types
Type I pneumocytes: extremely thin squamous cells covering ~95% of alveolar surface; form the gas exchange barrier. Type II pneumocytes: cuboidal cells with lamellar bodies that produce surfactant (dipalmitoylphosphatidylcholine, DPPC); serve as progenitor cells for both type I and type II cells. Alveolar macrophages (dust cells): phagocytose inhaled particles and pathogens.
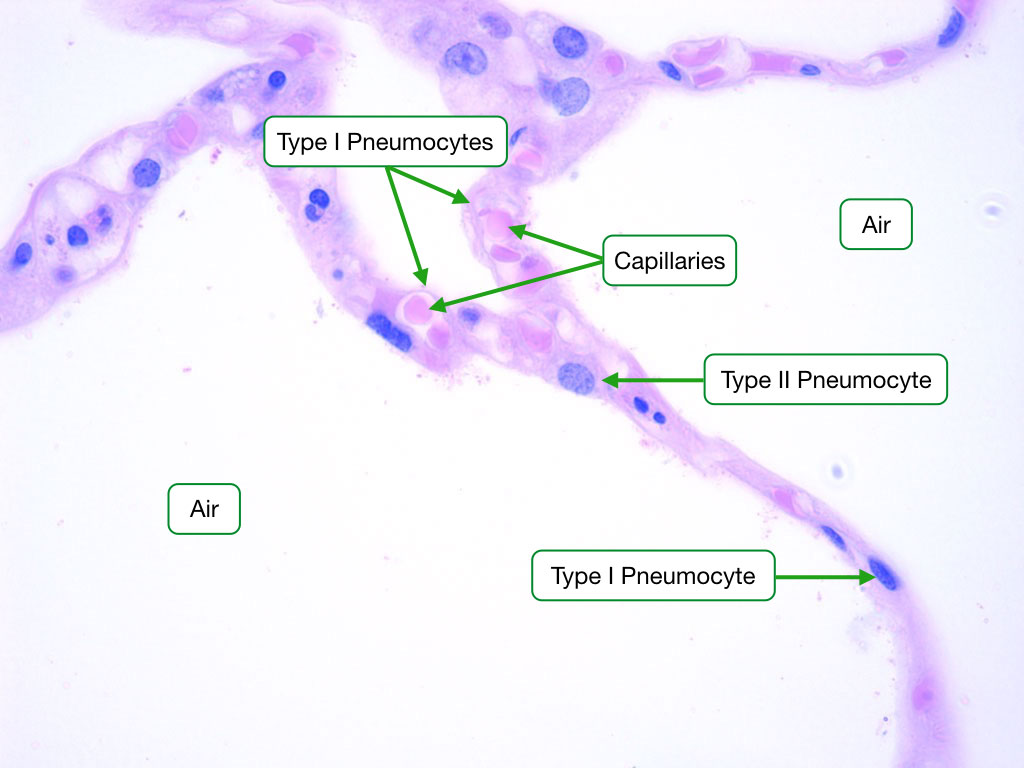
The blood-air barrier consists of (thin to thick): surfactant → type I pneumocyte → fused basal laminae → capillary endothelium. Total thickness: ~0.5 μm. This is the thinnest diffusion barrier in the body.
Club (Clara) Cells
Club cells (formerly Clara cells) are non-ciliated secretory cells found in terminal and respiratory bronchioles. They secrete club cell secretory protein (CC16), a component of the bronchiolar surfactant, and also function as progenitor cells for bronchiolar epithelial regeneration. Club cells contain abundant smooth ER and participate in xenobiotic metabolism (cytochrome P450 enzymes). They have a dome-shaped apical surface that protrudes into the airway lumen.
Pulmonary Vasculature
The lung has a dual blood supply: pulmonary arteries (deoxygenated blood from the right ventricle; travel alongside bronchi in bronchovascular bundles) and bronchial arteries (oxygenated blood from aorta; supply airway walls, visceral pleura). Pulmonary arteries are thin-walled elastic arteries that become muscular arteries at the level of bronchioles. Chronic hypoxia causes pulmonary arterial hypertension through medial hypertrophy and intimal fibrosis of pulmonary arterioles—histologically visible as thickened vascular walls and, in severe cases, plexiform lesions.
22 Lymphoid Organs
Lymphoid organs are classified as primary (where lymphocytes mature: bone marrow for B cells, thymus for T cells) or secondary (where immune responses are initiated: lymph nodes, spleen, MALT).
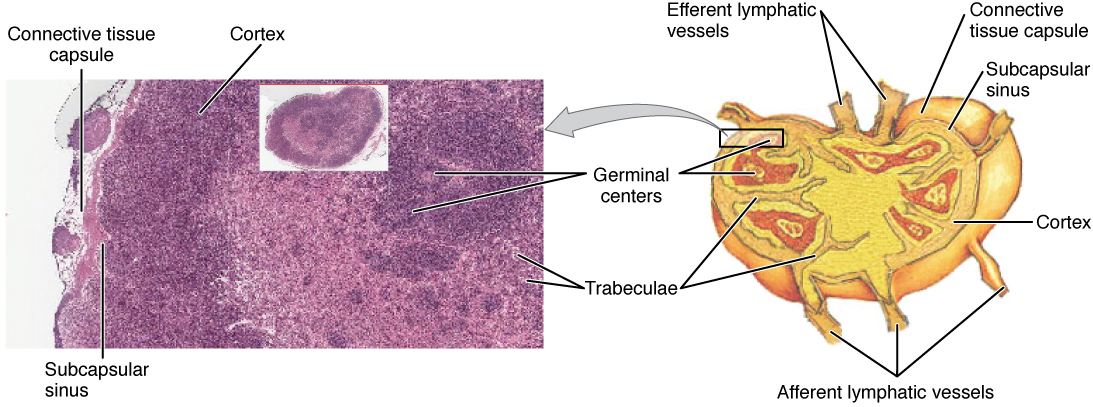
Lymph Node
Bean-shaped; surrounded by a capsule. Cortex: lymphoid follicles—primary (inactive, dense, small lymphocytes) and secondary (active, with germinal centers containing centroblasts, centrocytes, follicular dendritic cells, and tingible body macrophages). Paracortex: T-cell zone; high endothelial venules (HEVs) for lymphocyte entry. Medulla: medullary cords (plasma cells, macrophages) and medullary sinuses.
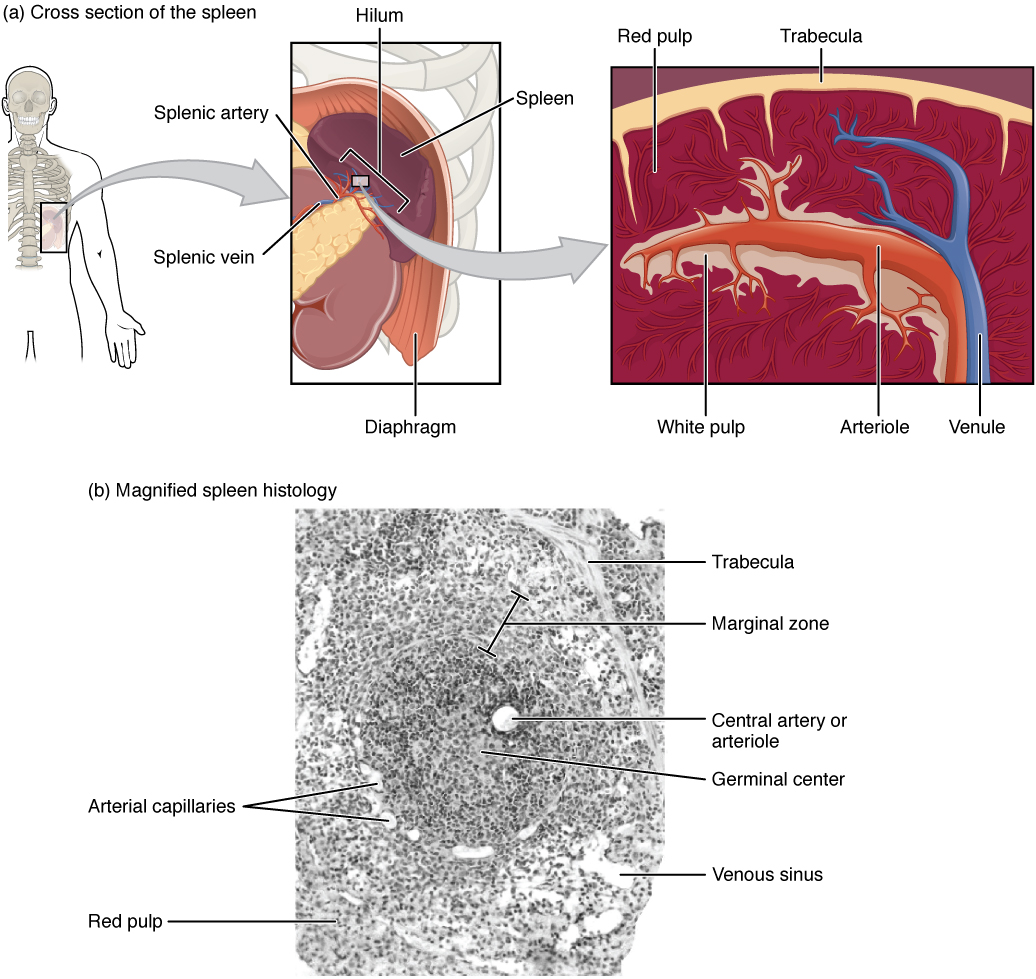
Spleen
The largest lymphoid organ. White pulp: periarterial lymphoid sheath (PALS, T cells around central arterioles) + lymphoid follicles (B cells). Marginal zone: between white and red pulp; APCs, marginal zone B cells. Red pulp: splenic cords (of Billroth) and sinusoids; functions as a blood filter, removing old/damaged RBCs (culling) and inclusions (pitting, e.g., Howell-Jolly bodies).
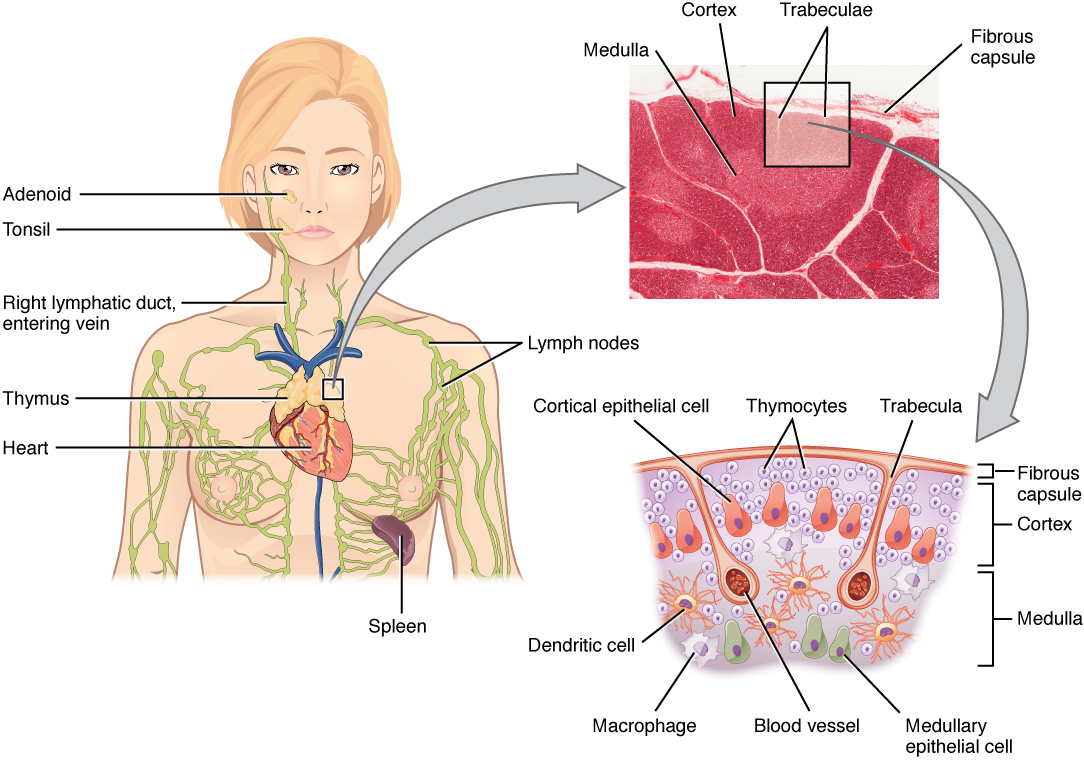
Thymus
Bilobed organ with lobules containing darker cortex (densely packed immature T cells/thymocytes, cortical epithelial cells, macrophages) and paler medulla (mature T cells, medullary epithelial cells, Hassall corpuscles—whorled keratinized epithelial cell remnants unique to thymus). The blood-thymus barrier in the cortex prevents premature antigen exposure. The thymus involutes with age (replaced by adipose tissue).
Mucosa-Associated Lymphoid Tissue (MALT)
MALT includes unencapsulated lymphoid aggregates in mucosal sites: Peyer patches (ileum), tonsils (palatine, pharyngeal/adenoid, lingual), appendix, and bronchus-associated lymphoid tissue (BALT). These sites sample antigens from mucosal surfaces via M cells (microfold cells) in the follicle-associated epithelium. M cells lack microvilli and a glycocalyx, facilitating antigen uptake by transcytosis to underlying dendritic cells and lymphocytes. MALT is the site of origin for MALT lymphoma (extranodal marginal zone B-cell lymphoma), which is associated with chronic H. pylori infection in the stomach.
| Feature | Lymph Node | Spleen | Thymus |
|---|---|---|---|
| Capsule | Yes + trabeculae | Yes + trabeculae | Yes + septa |
| Afferent lymphatics | Yes | No | No |
| Germinal centers | Yes (cortex) | Yes (white pulp) | No |
| Unique feature | HEVs in paracortex | Red pulp sinusoids | Hassall corpuscles |
| Filters | Lymph | Blood | Neither (T-cell education) |
23 Endocrine Glands
Pituitary Gland (Hypophysis)
The anterior pituitary (adenohypophysis) is organized as cords and clusters of cells. Three histologic types based on H&E staining: acidophils (GH, prolactin), basophils (FSH, LH, TSH, ACTH), and chromophobes (degranulated or inactive cells). The posterior pituitary (neurohypophysis) contains unmyelinated axons from hypothalamic neurons (supraoptic → ADH; paraventricular → oxytocin), Herring bodies (axonal swellings containing neurosecretory granules), and pituicytes (modified glial cells).
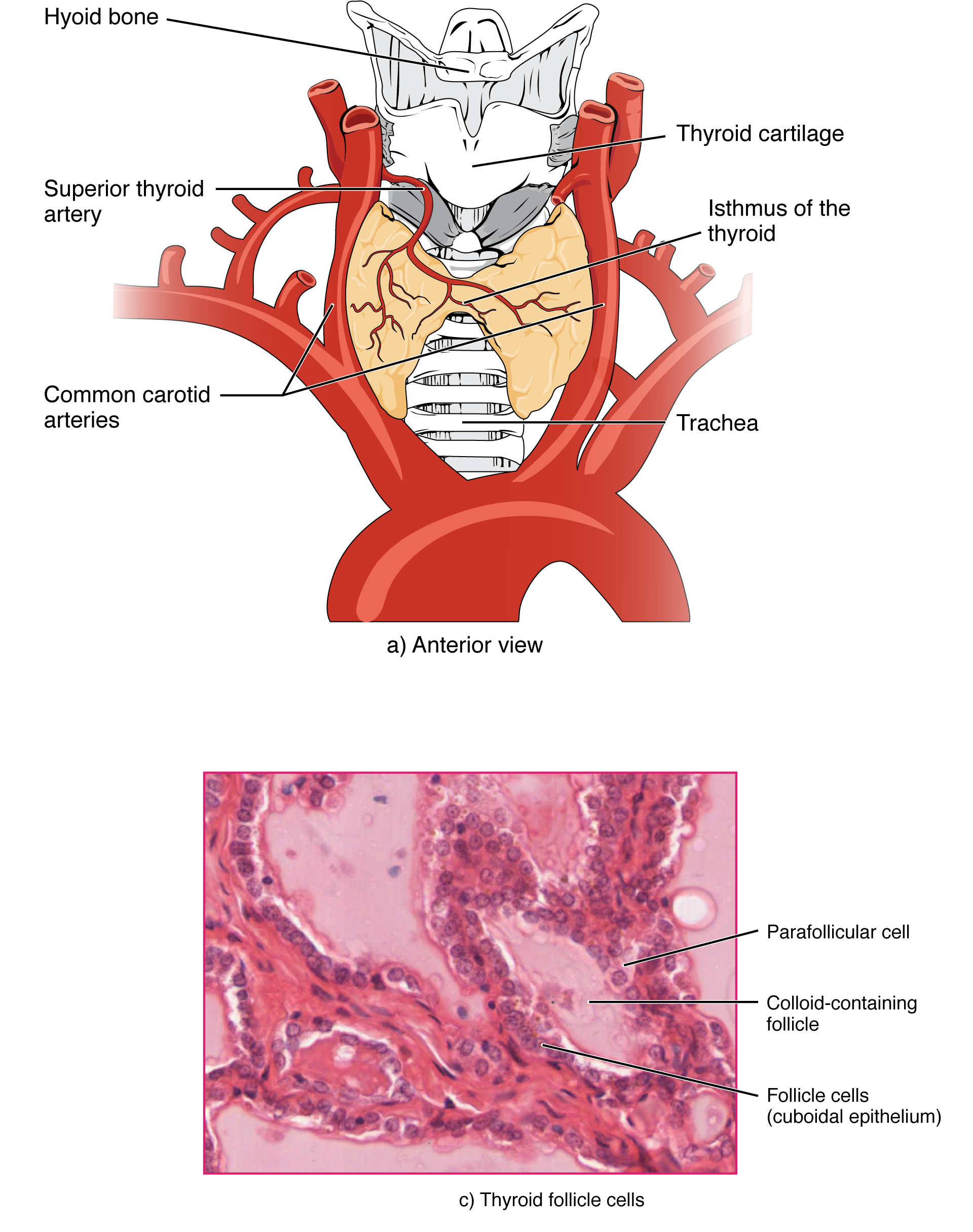
Thyroid Gland
The thyroid is the only endocrine gland that stores its product extracellularly. Follicles lined by simple cuboidal epithelium surround colloid (thyroglobulin). Active follicles have tall columnar cells with scalloped colloid; inactive follicles are flat with dense colloid. Parafollicular (C) cells are found between follicles or within the follicular epithelium; they secrete calcitonin and are the cells of origin for medullary thyroid carcinoma.
Adrenal Gland
The adrenal cortex (mesoderm origin) has three zones: zona glomerulosa (mineralocorticoids, aldosterone), zona fasciculata (glucocorticoids, cortisol; "spongiocytes" with lipid droplets), and zona reticularis (androgens, DHEA). Mnemonic: GFR = Salt, Sugar, Sex. The adrenal medulla (neural crest origin) contains chromaffin cells that secrete epinephrine and norepinephrine; stains brown with chromium salts (chromaffin reaction).
Parathyroid Glands
Composed of chief (principal) cells (produce PTH; small cells with central round nuclei) and oxyphil cells (larger, eosinophilic, abundant mitochondria; function uncertain; increase with age). Adipose tissue increases with age, comprising up to 50–70% of the gland in elderly individuals.
Pineal Gland
The pineal gland contains pinealocytes (produce melatonin from serotonin; regulate circadian rhythm) and glial cells (astrocyte-like interstitial cells). Corpora arenacea ("brain sand") are calcified concretions that increase with age and are visible on skull X-rays; they serve as a useful midline marker in radiology. Pineal tumors can cause Parinaud syndrome (paralysis of upward gaze) by compressing the superior colliculus.
Endocrine Pancreas — Additional Detail
| Cell Type | Hormone | % of Islet | Location in Islet | Stain Characteristic |
|---|---|---|---|---|
| Alpha (α) | Glucagon | ~15–20% | Peripheral | Argyrophilic (silver stains) |
| Beta (β) | Insulin, C-peptide, amylin | ~65–70% | Central core | Aldehyde fuchsin+ |
| Delta (δ) | Somatostatin | ~5–10% | Scattered | Immunostain only reliable method |
| PP (F) | Pancreatic polypeptide | ~2% | Scattered | Immunostain only reliable method |
| Epsilon (ε) | Ghrelin | <1% | Scattered | Rare; immunostain |
Type 1 DM: autoimmune destruction of beta cells; islets show lymphocytic infiltration (insulitis) early, then atrophy with near-total beta cell loss. Type 2 DM: amyloid deposition in islets (islet amyloid polypeptide/amylin derived from beta cells; Congo red+); beta cell mass reduced ~40–60% but not absent. These histologic patterns are diagnostically distinct.
24 Reproductive Organs
Male Reproductive System
The seminiferous tubule is lined by Sertoli cells (supporting, forming the blood-testis barrier with tight junctions) and spermatogenic cells in various stages: spermatogonia (basal) → primary spermatocytes → secondary spermatocytes → spermatids → spermatozoa (luminal). Leydig (interstitial) cells lie between tubules; they are large, eosinophilic cells with lipid droplets and Reinke crystals; they produce testosterone under LH stimulation.
The epididymis is lined by pseudostratified columnar epithelium with stereocilia (for sperm maturation and concentration). The vas deferens has thick muscular walls (inner longitudinal, middle circular, outer longitudinal) for peristaltic propulsion of sperm. The prostate gland is a tubuloalveolar gland surrounding the prostatic urethra; it produces prostatic fluid rich in PSA, citric acid, and zinc. Corpora amylacea (prostatic concretions) increase with age.
Female Reproductive System
The ovary has a cortex containing follicles at various stages of development (primordial → primary → secondary → Graafian/antral → corpus luteum → corpus albicans) and a vascular medulla. The uterus has three layers: endometrium (functional + basal layers), myometrium (thick smooth muscle), and serosa/adventitia. The endometrium undergoes cyclic changes: proliferative phase (estrogen; straight glands, mitotic activity), secretory phase (progesterone; coiled glands, glycogen-rich secretions, spiral arteries), and menstrual phase (spiral artery constriction, ischemic necrosis, shedding).
The cervix has an ectocervix lined by stratified squamous epithelium and an endocervix lined by simple columnar mucus-secreting epithelium. The squamocolumnar junction (transformation zone) is the site of cervical dysplasia and squamous cell carcinoma; it is the target of the Pap smear.
Placenta
The mature placenta is organized into chorionic villi, which are the functional exchange units. Each villus contains a core of fetal mesenchyme with fetal blood vessels, covered by an outer layer of syncytiotrophoblast (continuous multinucleated layer in direct contact with maternal blood; produces hCG, hPL, progesterone) and an inner layer of cytotrophoblast (discrete cells; mitotically active; serve as stem cells for syncytiotrophoblast). In early pregnancy, both layers are prominent; by term, cytotrophoblast cells become sparse and the syncytiotrophoblast thins, forming the vasculosyncytial membrane for efficient gas exchange.
Breast
The mammary gland is a modified apocrine sweat gland. The functional unit is the terminal duct lobular unit (TDLU), consisting of extralobular and intralobular ducts opening into acini (lobules). During lactation, acinar cells enlarge with lipid droplets (apocrine secretion of fat) and protein-rich secretions (merocrine). The resting breast is primarily interlobular connective tissue and fat, with small inactive TDLUs. Most breast carcinomas (both ductal and lobular) originate in the TDLU. Myoepithelial cells surround ducts and acini; their loss on IHC (p63, SMA, calponin negative) confirms invasive carcinoma.
25 Eye & Ear
Eye
The eye has three tunics: fibrous (sclera + cornea), vascular (uvea) (choroid, ciliary body, iris), and neural (retina). The cornea is avascular and has 5 layers: corneal epithelium (stratified squamous, non-keratinized), Bowman layer (acellular collagen), stroma (90% of thickness; type I collagen lamellae with keratocytes), Descemet membrane (thick basement membrane of endothelium), and corneal endothelium (simple squamous; maintains corneal clarity via Na+/K+-ATPase pump).
The retina has 10 layers. Key cell types: rods (dim-light vision; rhodopsin) and cones (color/acuity; iodopsin) are photoreceptors whose outer segments are phagocytosed daily by the retinal pigment epithelium (RPE). The fovea centralis has only cones with a 1:1 cone-to-ganglion-cell ratio for maximum acuity. The optic disc (blind spot) is where ganglion cell axons exit; no photoreceptors.
Ear
The organ of Corti in the cochlea is the receptor organ for hearing. It sits on the basilar membrane and contains inner hair cells (primary sensory transducers; ~3,500) and outer hair cells (amplifiers; ~12,000). Hair cell stereocilia are deflected by endolymph movement, opening mechanotransduction channels. Tip links connecting stereocilia are critical for channel gating.
The vestibular system includes the maculae of utricle (horizontal acceleration) and saccule (vertical acceleration), which contain hair cells with stereocilia embedded in a gelatinous otolithic membrane with calcium carbonate crystals (otoconia). The cristae ampullares in semicircular canals detect angular acceleration; hair cells project into a gelatinous cupula.
Cochlear Histology Details
The cochlea is divided into three chambers: scala vestibuli (perilymph), scala media / cochlear duct (endolymph, high K+), and scala tympani (perilymph). The stria vascularis on the lateral wall of the cochlear duct is the only vascularized epithelium in the body; it produces endolymph and maintains the endocochlear potential (+80 mV) essential for hair cell transduction. The Reissner membrane separates scala vestibuli from scala media. The basilar membrane (narrow and stiff at base → wide and floppy at apex) supports the organ of Corti and provides the tonotopic gradient (base = high frequency, apex = low frequency).
Retinal Layers (High-Yield)
The 10 layers of the retina from outermost (closest to choroid) to innermost (closest to vitreous): (1) Retinal pigment epithelium (RPE), (2) Photoreceptor layer (rods and cones outer/inner segments), (3) External limiting membrane, (4) Outer nuclear layer (photoreceptor nuclei), (5) Outer plexiform layer (synapses), (6) Inner nuclear layer (bipolar, amacrine, horizontal, Muller cell nuclei), (7) Inner plexiform layer (synapses), (8) Ganglion cell layer, (9) Nerve fiber layer (ganglion cell axons → optic nerve), (10) Internal limiting membrane (Muller cell end-feet). Light must pass through all inner layers to reach photoreceptors. The RPE phagocytoses shed photoreceptor outer segment discs daily.
Age-related macular degeneration: drusen deposits between RPE and Bruch membrane; dry (atrophic) vs. wet (neovascular) forms. Benign paroxysmal positional vertigo (BPPV): displaced otoconia from the utricle enter a semicircular canal (usually posterior), causing inappropriate cupula deflection with head movements.
Lens
The lens is a transparent, biconvex, avascular structure suspended by zonular fibers (zonules of Zinn) from the ciliary body. It is enclosed in a thick elastic lens capsule (the thickest basement membrane in the body, type IV collagen). The anterior surface is lined by a single layer of lens epithelial cells that proliferate at the equator and differentiate into lens fibers—elongated, anucleate cells packed with crystallin proteins. Cataracts result from opacification of crystallins due to age, UV exposure, diabetes (sorbitol accumulation via aldose reductase), or corticosteroids.
Blood Vessel Histology
Blood vessels have three tunics: tunica intima (endothelium + subendothelial CT + internal elastic lamina), tunica media (smooth muscle +/− elastic lamellae), and tunica adventitia (CT with vasa vasorum and nervi vasorum). Large arteries (aorta, pulmonary) are elastic arteries with abundant elastic lamellae in the media. Medium arteries (coronary, renal, femoral) are muscular arteries with prominent smooth muscle media and well-defined internal and external elastic laminae. Arterioles (1–2 layers of smooth muscle) are the primary regulators of vascular resistance and blood pressure.
| Vessel Type | Key Histologic Features | Clinical Significance |
|---|---|---|
| Elastic artery | Multiple elastic lamellae in media; vasa vasorum in adventitia | Atherosclerosis; aortic aneurysm/dissection |
| Muscular artery | Thick smooth muscle media; distinct IEL/EEL | Atherosclerosis; vasculitis (e.g., PAN) |
| Arteriole | 1–2 layers smooth muscle; no EEL | Hypertensive arteriosclerosis (hyaline, hyperplastic) |
| Capillary (continuous) | Endothelium + BM; tight junctions | BBB; muscle, lung, skin |
| Capillary (fenestrated) | Endothelial pores (60–80 nm) with diaphragms | GI villi, renal glomeruli, endocrine glands |
| Sinusoid (discontinuous) | Large gaps; discontinuous BM | Liver, spleen, bone marrow |
| Vein | Thin media; thick adventitia; valves (in limbs) | Varicose veins; DVT |
26 Pathologic Histology
Understanding normal tissue architecture enables recognition of pathologic patterns. Key categories include cellular adaptations, cell injury/death, inflammation, neoplasia, and deposition diseases.
Cellular Adaptations
| Adaptation | Definition | Example |
|---|---|---|
| Hypertrophy | Increase in cell size | Left ventricular hypertrophy (pressure overload) |
| Hyperplasia | Increase in cell number | Endometrial hyperplasia (excess estrogen) |
| Atrophy | Decrease in cell size | Disuse atrophy of skeletal muscle |
| Metaplasia | Reversible change of one differentiated cell type to another | Barrett esophagus: squamous → columnar (intestinal) |
| Dysplasia | Disordered growth with abnormal cell morphology | Cervical intraepithelial neoplasia (CIN) |
Cell Death
Necrosis (pathologic): coagulative (most organs; ischemia), liquefactive (brain, abscess), caseous (TB), fat necrosis (pancreas), fibrinoid (vessels). Nuclear changes: pyknosis (condensation) → karyorrhexis (fragmentation) → karyolysis (dissolution). Apoptosis (programmed): cell shrinkage, chromatin condensation, membrane blebbing, apoptotic bodies; no inflammatory reaction; mediated by caspases (intrinsic/mitochondrial and extrinsic/death receptor pathways).
Inflammation Patterns
| Pattern | Histologic Features | Example |
|---|---|---|
| Acute (suppurative) | Neutrophilic infiltrate; edema; tissue damage | Bacterial abscess, acute appendicitis |
| Chronic | Lymphocytes, macrophages, plasma cells; fibrosis | Chronic hepatitis, rheumatoid arthritis |
| Granulomatous | Epithelioid macrophages, multinucleated giant cells, lymphocytes | TB (caseating), sarcoidosis (non-caseating) |
| Fibrinous | Fibrin deposits on serosal surfaces | Fibrinous pericarditis (Dressler syndrome) |
Neoplastic Patterns
| Pattern | Description | Example |
|---|---|---|
| Glandular (adenocarcinoma) | Malignant glands invading stroma | Colon, prostate, lung adenocarcinoma |
| Squamous | Keratinization, intercellular bridges | Lung, esophageal, cervical SCC |
| Papillary | Finger-like projections with fibrovascular cores | Papillary thyroid carcinoma |
| Small round blue cell | Sheets of small cells with scant cytoplasm | Ewing sarcoma, neuroblastoma, lymphoma |
| Spindle cell | Elongated cells in fascicles or whorls | Fibrosarcoma, leiomyosarcoma, schwannoma |
| Signet ring | Intracellular mucin displaces nucleus | Diffuse gastric carcinoma (linitis plastica) |
Fibrosis & Wound Healing
Tissue repair proceeds through overlapping phases: hemostasis (platelet plug, fibrin clot) → inflammation (neutrophils, then macrophages) → proliferation (granulation tissue: new capillaries + fibroblasts + loose ECM) → remodeling (collagen maturation, type III replaced by type I, wound contraction by myofibroblasts). Granulation tissue is the hallmark of healing: it appears as beefy red, granular tissue rich in thin-walled capillaries and plump fibroblasts.
| Healing Type | Mechanism | Example |
|---|---|---|
| Primary intention | Clean, approximated wound edges; minimal granulation tissue | Surgical incision closed with sutures |
| Secondary intention | Open wound; heals from base up with granulation tissue; more contraction | Large ulcer, abscess cavity |
| Tertiary intention | Wound left open initially, then closed after infection control | Contaminated wound closed delayed |
Deposition Diseases
| Deposit | Composition | Stain | Disease |
|---|---|---|---|
| Amyloid | Misfolded protein fibrils (beta-pleated sheets) | Congo red (apple-green birefringence) | AL amyloidosis (light chain), AA amyloidosis (serum amyloid A) |
| Hemosiderin | Iron-protein complex | Prussian blue | Hemochromatosis, hemosiderosis, chronic congestion |
| Calcium | Calcium phosphate | Von Kossa (black); alizarin red | Dystrophic (damaged tissue, normal Ca) vs. metastatic (normal tissue, high Ca) calcification |
| Lipofuscin | Oxidized lipid/protein (wear-and-tear) | Autofluorescent; PAS+, acid-fast | Aging; brown atrophy of heart/liver |
| Melanin | Tyrosine-derived pigment | Fontana-Masson (silver) | Melanoma, dermal melanocytosis |
| Urate crystals | Monosodium urate | Negative birefringence; dissolved by formalin (need alcohol fixation) | Gout; tophaceous deposits |
Dysplasia vs. Neoplasia
Dysplasia is a precancerous condition characterized by disordered cellular maturation with architectural and cytologic abnormalities (nuclear enlargement, hyperchromasia, increased mitoses, loss of polarity) but without invasion through the basement membrane. Dysplasia can be classified as low-grade or high-grade. Carcinoma in situ (CIS) represents full-thickness dysplasia (high-grade) that has not breached the basement membrane. Once tumor cells penetrate the basement membrane into underlying stroma, the lesion is classified as invasive carcinoma. This transition is the critical event that enables metastatic potential.
| Feature | Benign | Malignant |
|---|---|---|
| Growth rate | Slow | Rapid (often) |
| Border | Well-circumscribed, encapsulated | Irregular, infiltrative |
| Differentiation | Well-differentiated | Variable (well to poorly differentiated) |
| Mitoses | Few, normal | Numerous, atypical |
| Necrosis | Rare | Common (outgrows blood supply) |
| Metastasis | Never | Yes (defines malignancy) |
| Nuclear features | Uniform, normal N:C ratio | Pleomorphic, high N:C ratio, hyperchromatic |
27 Clinical Correlates & Diagnostic Stains
Histologic findings directly guide clinical decision-making. This section summarizes high-yield diagnostic patterns and the stains used to identify them.
Diagnostic Stain Applications by Organ System
| Clinical Scenario | Stain | Expected Finding |
|---|---|---|
| Liver fibrosis staging | Masson trichrome | Blue collagen bridging portal triads |
| Liver iron overload | Prussian blue | Blue granules in hepatocytes |
| Liver copper (Wilson disease) | Rhodanine | Red-brown granules in hepatocytes |
| Alpha-1 antitrypsin deficiency | PAS-diastase | PAS+ globules (diastase-resistant) in periportal hepatocytes |
| Amyloidosis (any organ) | Congo red | Apple-green birefringence under polarized light |
| Fungal infection | GMS (silver) | Black fungal elements |
| Mycobacterial infection | Ziehl–Neelsen / Fite | Red acid-fast bacilli |
| H. pylori gastritis | Warthin-Starry (silver) or Giemsa | Curved organisms in mucus layer |
| Renal basement membrane disease | Electron microscopy + IF | Subepithelial deposits (membranous); linear IgG (Goodpasture) |
| Mast cell disease | Toluidine blue / Giemsa | Metachromatic granules |
Immunofluorescence Patterns in Renal Disease
| Pattern | Appearance | Disease |
|---|---|---|
| Linear | Smooth line along GBM | Goodpasture syndrome (anti-GBM) |
| Granular | Bumpy, irregular deposits | Membranous nephropathy, post-streptococcal GN, lupus nephritis |
| Negative (pauci-immune) | No significant Ig or complement | ANCA-associated vasculitis (GPA, MPA) |
Electron Microscopy in Diagnosis
EM remains essential for specific diagnoses where light microscopy and IHC are insufficient:
| Finding | Diagnosis |
|---|---|
| Foot process effacement | Minimal change disease |
| Subepithelial "humps" | Post-streptococcal glomerulonephritis |
| Subepithelial "spike and dome" | Membranous nephropathy |
| Birbeck granules (tennis racket) | Langerhans cell histiocytosis |
| Weibel-Palade bodies | Endothelial cell identification (vWF storage) |
| Dense-core neurosecretory granules | Neuroendocrine tumors |
| Reinke crystals | Leydig cell tumor |
The standard diagnostic approach: (1) H&E for overall architecture and cytology → (2) Special stains for specific components (collagen, mucin, organisms, deposits) → (3) IHC for cell lineage and markers → (4) Molecular studies (FISH, PCR, NGS) for genetic alterations → (5) EM for ultrastructural details when needed. Each step narrows the differential and guides treatment.
CK7/CK20 Pattern for Tumor Site of Origin
| Pattern | CK7 | CK20 | Primary Site |
|---|---|---|---|
| CK7+/CK20− | + | − | Lung, breast, ovary, endometrium, thyroid, pancreas (non-mucinous) |
| CK7−/CK20+ | − | + | Colorectal carcinoma, Merkel cell carcinoma |
| CK7+/CK20+ | + | + | Urothelial carcinoma, pancreatic, mucinous ovarian |
| CK7−/CK20− | − | − | Hepatocellular carcinoma, renal cell carcinoma, prostate, squamous cell carcinoma |
Molecular Techniques in Histopathology
| Technique | Principle | Clinical Application |
|---|---|---|
| FISH (fluorescence in situ hybridization) | Fluorescent DNA probes detect specific sequences | HER2 amplification (breast), BCR-ABL (CML), ALK rearrangement (lung) |
| PCR / RT-PCR | Amplify specific DNA/RNA sequences | Clonality assessment (IgH/TCR rearrangement), viral detection |
| Next-generation sequencing (NGS) | Massively parallel sequencing of targeted panels or whole exome | Targeted therapy selection (EGFR, BRAF, KRAS mutations) |
| In situ hybridization (ISH) | Labeled RNA/DNA probes on tissue sections | EBV (EBER), HPV, kappa/lambda light chain restriction |
| Flow cytometry | Fluorescent antibodies on cell suspensions | Lymphoma/leukemia immunophenotyping |
28 High-Yield Review & Reference Tables
Pathognomonic Histologic Findings
| Finding | Diagnosis |
|---|---|
| Auer rods (pink, needle-like cytoplasmic inclusions) | Acute myeloid leukemia (especially APL, M3) |
| Owl-eye nuclear inclusions | CMV infection |
| Cowdry type A inclusions (eosinophilic nuclear) | Herpes simplex/varicella-zoster |
| Reed-Sternberg cells (bilobed nuclei, "owl eyes") | Hodgkin lymphoma (classic) |
| Psammoma bodies (concentric calcifications) | Papillary thyroid carcinoma, meningioma, serous ovarian carcinoma |
| Call-Exner bodies | Granulosa cell tumor of ovary |
| Orphan Annie eye nuclei (clear, empty-appearing) | Papillary thyroid carcinoma |
| Schiller-Duval bodies (glomeruloid structures) | Yolk sac (endodermal sinus) tumor |
| Homer Wright rosettes | Neuroblastoma, medulloblastoma |
| Flexner-Wintersteiner rosettes | Retinoblastoma |
| Negri bodies (eosinophilic cytoplasmic inclusions) | Rabies |
| Verocay bodies (palisading nuclei) | Schwannoma (Antoni A pattern) |
| Councilman bodies (apoptotic hepatocytes) | Yellow fever, viral hepatitis |
| Mallory-Denk bodies (damaged cytokeratin) | Alcoholic hepatitis, steatohepatitis |
| Lewy bodies (eosinophilic cytoplasmic inclusions) | Parkinson disease, Lewy body dementia |
| Hirano bodies (eosinophilic rod-like) | Alzheimer disease |
| Neurofibrillary tangles (hyperphosphorylated tau) | Alzheimer disease |
| Schaumann bodies (laminated calcium concretions) | Sarcoidosis |
| Asteroid bodies (star-shaped inclusions in giant cells) | Sarcoidosis |
| Zebra bodies (lamellar lipid inclusions) | Fabry disease, Niemann-Pick disease |
| Birbeck granules (pentalaminar "tennis rackets") | Langerhans cell histiocytosis |
IHC Tumor Panels Quick Reference
| Tumor Type | Key Positive Markers | Key Negative Markers |
|---|---|---|
| Carcinoma (general) | Cytokeratins (AE1/AE3), EMA | Vimentin (usually) |
| Melanoma | S-100, HMB-45, Melan-A, SOX10 | Cytokeratins |
| Lymphoma | CD45 (LCA), CD20 (B), CD3 (T) | Cytokeratins |
| Sarcoma | Vimentin; lineage markers (desmin, SMA) | Cytokeratins (usually) |
| Neuroendocrine tumor | Chromogranin, synaptophysin, CD56 | Variable |
| GIST | CD117 (c-KIT), DOG1 | Desmin, S-100 |
| Mesothelioma | Calretinin, CK5/6, WT-1, D2-40 | CEA, TTF-1, BerEP4 |
| Prostate carcinoma | PSA, PSAP, NKX3.1 | High MW CK (p63 absent in cancer) |
| Hepatocellular carcinoma | HepPar-1, arginase-1, glypican-3 | CK7 (usually) |
Tissue Type Identification Quick Reference
| Clue on H&E | Think |
|---|---|
| Peripheral nuclei, striations | Skeletal muscle |
| Central nuclei, striations, intercalated discs | Cardiac muscle |
| Central cigar-shaped nuclei, no striations | Smooth muscle |
| Lacunae in glassy matrix | Hyaline cartilage |
| Concentric lamellae with central canal | Compact bone (osteon) |
| Cells on basement membrane, no vessels in tissue | Epithelium |
| Nissl substance, large nucleolus, axon hillock | Neuron |
| Whorled keratinized structure in thymic medulla | Hassall corpuscle |
| Dome-shaped surface cells, appears layered | Transitional epithelium (urothelium) |
| Central pallor in biconcave disc, 7–8 μm | Erythrocyte |
Metaplasia Examples (High-Yield)
| Site | Normal Epithelium | Metaplastic Epithelium | Stimulus | Premalignant Risk |
|---|---|---|---|---|
| Esophagus (Barrett) | Stratified squamous | Intestinal-type columnar (goblet cells) | Chronic GERD / acid reflux | Esophageal adenocarcinoma |
| Bronchi | Pseudostratified ciliated columnar | Stratified squamous | Smoking | Squamous cell carcinoma |
| Cervix | Columnar (endocervix) | Stratified squamous | Chronic irritation, low pH | Normal adaptive (not premalignant itself) |
| Bladder | Transitional (urothelium) | Squamous | Chronic infection, Schistosoma | Squamous cell carcinoma |
| Stomach | Gastric columnar | Intestinal-type (goblet cells) | Chronic H. pylori gastritis | Gastric adenocarcinoma (intestinal type) |
Collagen types: "Be (I) Seeing (II) Three (III) Floors (IV) — Bone/Skin, Cartilage, Reticular/Vessels, Basement membrane."
Epidermal layers (deep to superficial): "Californians (basale) Like (spinosum) Girls (granulosum) in (lucidum) String (corneum) Bikinis" — or "Come, Let's Get Sun Burned."
Adrenal cortex zones: "GFR — Salt (glomerulosa/aldosterone), Sugar (fasciculata/cortisol), Sex (reticularis/androgens)."
Growth plate zones: "Really (reserve) Please (proliferative) Help (hypertrophic) Children (calcification) Ossify (ossification)."
Organ Identification Quick Reference
| Histologic Clue | Organ |
|---|---|
| Sinusoids + hepatocyte plates + portal triads + central vein | Liver |
| Follicles with colloid, parafollicular C cells | Thyroid |
| Chief cells + oxyphil cells + adipose tissue | Parathyroid gland |
| Cortex (3 zones) + medulla (chromaffin cells) | Adrenal gland |
| Acidophils + basophils + chromophobes in cords | Anterior pituitary |
| Herring bodies + pituicytes + unmyelinated axons | Posterior pituitary |
| Glomeruli + PCT (brush border) + DCT + collecting ducts | Kidney |
| Villi + crypts + Paneth cells + goblet cells | Small intestine |
| Brunner glands in submucosa + villi | Duodenum |
| Peyer patches in submucosa + shorter villi | Ileum |
| No villi + abundant goblet cells + straight crypts | Large intestine |
| Gastric pits + parietal cells (eosinophilic) + chief cells | Stomach (body/fundus) |
| Centroacinar cells + islets of Langerhans | Pancreas |
| White pulp (PALS) + red pulp (cords + sinusoids) | Spleen |
| Cortex (follicles) + paracortex (HEVs) + medulla | Lymph node |
| Hassall corpuscles + cortex-medulla arrangement | Thymus |
| Seminiferous tubules + Leydig cells | Testis |
| Pseudostratified + stereocilia | Epididymis |
| Follicles at various stages + corpus luteum | Ovary |
| Endometrial glands + spiral arteries + myometrium | Uterus |
Commonly Tested Histology Comparisons
| Feature | PCT vs. DCT |
|---|---|
| Brush border | PCT: prominent brush border (microvilli) | DCT: absent |
| Cell height | PCT: taller, more eosinophilic | DCT: shorter, paler |
| Lumen | PCT: narrow, irregular (brush border fills lumen) | DCT: wider, round |
| Nuclei per cross-section | PCT: fewer (larger cells) | DCT: more (smaller cells) |
| Feature | Trachea vs. Esophagus (Cross-Section) |
|---|---|
| Epithelium | Trachea: pseudostratified ciliated columnar | Esophagus: stratified squamous |
| Support | Trachea: C-shaped hyaline cartilage rings | Esophagus: no cartilage |
| Glands | Trachea: seromucinous glands in submucosa | Esophagus: mucous glands in submucosa |
| Muscularis | Trachea: trachealis muscle (smooth) posteriorly | Esophagus: inner circular + outer longitudinal |
Step 1: Determine if it is a hollow organ (tubular with a lumen—GI, respiratory, urogenital) or a solid organ (parenchymal with capsule—liver, kidney, endocrine glands, lymphoid). Step 2: For hollow organs, identify the epithelium (squamous vs. columnar vs. transitional) and wall layers. Step 3: For solid organs, look for unique architectural features (follicles, sinusoids, lobules, cortex/medulla). Step 4: Identify any unique cell types or structures (Hassall corpuscles, Brunner glands, Leydig cells, islets). Step 5: Correlate with staining pattern if special stains are used.