Allergy & Immunology
Every allergic condition, immunodeficiency, diagnostic test, immunotherapy protocol, biologic agent, classification system, and management strategy in one place.
01 Immune System Anatomy & Physiology
The immune system is a distributed network of organs, cells, and soluble mediators that protects the host from pathogens, toxins, and aberrant cells. It is conventionally divided into innate immunity (rapid, non-specific, no memory) and adaptive immunity (slower onset, antigen-specific, generates immunological memory). Allergy and immunodeficiency arise from dysregulation or deficiency of these components.
Innate Immunity
The innate immune system provides immediate defense within hours. Key components include physical barriers (skin, mucosal epithelium, ciliary clearance), soluble factors (complement, lysozyme, defensins, interferons), and cellular effectors. Neutrophils are the most abundant circulating leukocytes (60–70% of WBCs) and the first responders to bacterial infection via phagocytosis and degranulation. Macrophages (tissue-resident monocyte derivatives) perform phagocytosis, antigen presentation, and cytokine secretion. Natural killer (NK) cells are large granular lymphocytes that kill virus-infected and tumor cells without prior sensitization via perforin/granzyme release. Dendritic cells (DCs) are the most potent antigen-presenting cells (APCs), bridging innate and adaptive immunity by processing antigens and presenting them via MHC molecules to T cells.
Mast Cells, Basophils, & Eosinophils
Mast cells are tissue-resident effectors found in skin, airways, and gut mucosa. They bear high-affinity IgE receptors (FcεRI) and are the central effectors of type I hypersensitivity. Cross-linking of surface-bound IgE by multivalent allergen triggers degranulation, releasing histamine, tryptase, heparin, proteases, and lipid mediators (prostaglandin D2, leukotriene C4). Basophils (<1% of circulating WBCs) also express FcεRI and release histamine and IL-4/IL-13, amplifying Th2 responses. Eosinophils (1–3% of WBCs) contain cytotoxic granules (major basic protein, eosinophil peroxidase, eosinophil cationic protein) and are key effectors in helminth defense, allergic inflammation, and eosinophilic disorders. Eosinophil survival is driven by IL-5 (the target of mepolizumab and benralizumab).
Adaptive Immunity — T Cells
T lymphocytes mature in the thymus and mediate cell-mediated immunity. CD4+ T helper (Th) cells recognize antigen presented via MHC class II on APCs and differentiate into functional subsets: Th1 (secrete IFN-γ, drive cell-mediated immunity against intracellular pathogens), Th2 (secrete IL-4, IL-5, IL-13; drive allergic inflammation, IgE class switching, eosinophilia), Th17 (secrete IL-17; mucosal defense, neutrophilic inflammation, autoimmunity), Treg (express FoxP3; suppress immune responses, maintain tolerance), and Tfh (follicular helper; support B-cell germinal center reactions). CD8+ cytotoxic T cells (CTLs) recognize antigen via MHC class I and directly kill infected or neoplastic cells via perforin/granzyme.
Adaptive Immunity — B Cells & Immunoglobulins
B lymphocytes mature in the bone marrow and produce antibodies (immunoglobulins). Upon antigen encounter and T-cell help, naïve B cells undergo class-switch recombination and somatic hypermutation in germinal centers, generating high-affinity antibody-secreting plasma cells and memory B cells. There are five immunoglobulin classes:
| Class | % of Serum Ig | Structure | Key Functions |
|---|---|---|---|
| IgG | 75–80% | Monomer; 4 subclasses (IgG1–4) | Opsonization, complement activation (IgG1, IgG3), placental transfer (neonatal immunity), ADCC |
| IgA | 10–15% | Monomer (serum) or dimer (secretory) | Mucosal defense (respiratory, GI, GU tracts); secretory IgA resists proteolysis |
| IgM | 5–10% | Pentamer | Primary immune response, strongest complement activator per molecule, does not cross placenta |
| IgE | <0.01% | Monomer | Allergic reactions (mast cell/basophil degranulation via FcεRI), helminth defense; elevated in atopy |
| IgD | <1% | Monomer | B-cell surface receptor (co-expressed with IgM on naïve B cells); function incompletely understood |
The Complement System
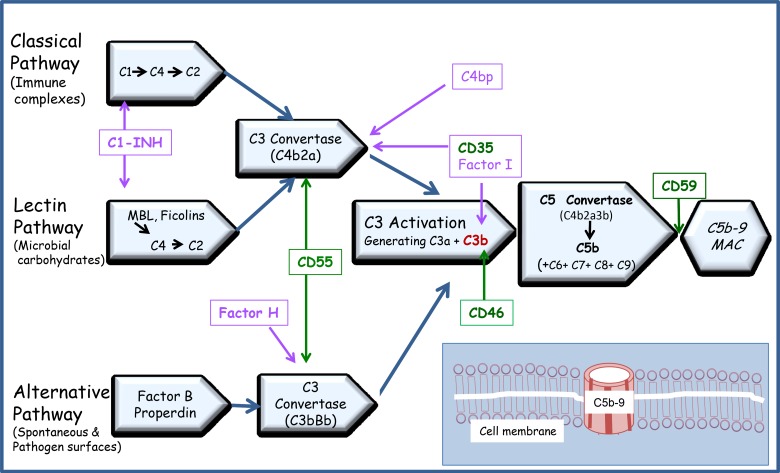
The complement system comprises >30 serum proteins activated via three pathways: classical (C1q binds antigen-antibody complexes — IgM or IgG), lectin (mannose-binding lectin binds pathogen surfaces), and alternative (spontaneous C3 hydrolysis, amplified on pathogen surfaces lacking complement regulatory proteins). All three converge on C3 convertase, which cleaves C3 to C3a (anaphylatoxin) and C3b (opsonin). C5 convertase generates C5a (most potent anaphylatoxin, neutrophil chemotaxis) and C5b, which initiates the membrane attack complex (MAC, C5b-9) that lyses target cells. Complement deficiencies predispose to specific infections: C1q/C2/C4 deficiency → SLE-like disease and encapsulated bacterial infections; C3 deficiency → severe pyogenic infections; C5–C9 (terminal) deficiency → recurrent Neisseria infections.
02 The Allergy & Immunology Evaluation
History
The allergy/immunology history is the single most important diagnostic tool. Key elements include: nature and timing of symptoms (seasonal vs perennial, immediate vs delayed), suspected triggers (foods, medications, stinging insects, environmental exposures), reproducibility of reactions, prior testing results, family history of atopy (allergic rhinitis, asthma, atopic dermatitis — the atopic triad), occupational exposures, and medication history (including OTC antihistamines, which must be held before skin testing). For immunodeficiency evaluation, document frequency and severity of infections, unusual organisms, failure to thrive, autoimmune manifestations, and family history of early death or immunodeficiency.
Physical Examination
Allergic signs include allergic shiners (infraorbital darkening from venous congestion), Dennie-Morgan lines (infraorbital skin folds), allergic salute (upward nose rubbing causing a transverse nasal crease), cobblestoning of posterior pharynx (lymphoid hyperplasia), pale/boggy nasal turbinates with clear rhinorrhea, and conjunctival injection with papillary reaction. Skin exam assesses for eczematous changes (flexural distribution in atopic dermatitis), urticaria (wheals), angioedema, and contact dermatitis patterns. Pulmonary exam assesses for wheezing, prolonged expiratory phase, and accessory muscle use. Lymphadenopathy, hepatosplenomegaly, and absence of tonsils/lymphoid tissue may suggest immunodeficiency.
Initial Laboratory Evaluation
| Test | Purpose | Key Findings |
|---|---|---|
| CBC with differential | Screen for eosinophilia, lymphopenia, neutropenia | Eosinophils >500/μL suggests allergic/parasitic; lymphocyte count <1500 in adults suggests T-cell deficiency |
| Total IgE | Assess atopic status | Normal <100 IU/mL in adults; markedly elevated (>2000) in ABPA, hyper-IgE syndrome, parasitic infection |
| Quantitative immunoglobulins (IgG, IgA, IgM) | Screen for antibody deficiency | Low IgG <600 mg/dL; absent IgA <7 mg/dL (selective IgA deficiency is most common PID, ~1:500) |
| Specific IgE (sIgE) panels | Identify allergen sensitization | ≥0.35 kU/L is positive; higher values correlate with (but do not confirm) clinical allergy |
| Tryptase | Mast cell activation/anaphylaxis | Normal <11.5 ng/mL; elevated >11.5 during acute event (draw 15 min–3 h after onset); persistently elevated suggests mastocytosis |
| Complement (CH50, C3, C4) | Screen for complement deficiency | CH50 = 0 suggests complete pathway deficiency; low C4 with normal C3 suggests C2 or C4 deficiency or hereditary angioedema |
03 Key Terminology & Abbreviations
Allergy and immunology uses a dense vocabulary of abbreviations across clinical documentation, research, and guidelines.
| Term | Meaning |
|---|---|
| Atopy | Genetic predisposition to produce IgE against common environmental allergens; manifests as allergic rhinitis, asthma, and/or atopic dermatitis |
| Sensitization | Presence of allergen-specific IgE (positive skin test or sIgE) — does NOT equal clinical allergy |
| Allergy | Clinical symptoms upon exposure to a sensitized allergen (sensitization + clinical reactivity) |
| Anaphylaxis | Acute, potentially fatal systemic hypersensitivity reaction involving ≥2 organ systems |
| Tolerance | Active immunologic non-responsiveness to a specific antigen (e.g., oral tolerance to foods) |
| Desensitization | Temporary induction of tolerance to a drug or allergen by graded dose escalation |
| Abbreviation | Meaning |
|---|---|
| SPT | Skin prick test |
| IDT | Intradermal test |
| sIgE | Allergen-specific immunoglobulin E |
| CRD | Component-resolved diagnostics |
| SCIT | Subcutaneous immunotherapy ("allergy shots") |
| SLIT | Sublingual immunotherapy (tablets or drops) |
| OFC | Oral food challenge |
| OIT | Oral immunotherapy |
| ARIA | Allergic Rhinitis and Its Impact on Asthma |
| GINA | Global Initiative for Asthma |
| NAEPP | National Asthma Education and Prevention Program |
| FeNO | Fractional exhaled nitric oxide |
| PFT | Pulmonary function test |
| FEV1 | Forced expiratory volume in 1 second |
| FVC | Forced vital capacity |
| PID | Primary immunodeficiency disease |
| CVID | Common variable immunodeficiency |
| XLA | X-linked agammaglobulinemia (Bruton's) |
| SCID | Severe combined immunodeficiency |
| CGD | Chronic granulomatous disease |
| HAE | Hereditary angioedema |
| CSU | Chronic spontaneous urticaria |
| EoE | Eosinophilic esophagitis |
| FPIES | Food protein–induced enterocolitis syndrome |
| ACD | Allergic contact dermatitis |
| AD | Atopic dermatitis |
| ABPA | Allergic bronchopulmonary aspergillosis |
| AERD | Aspirin-exacerbated respiratory disease (Samter's triad) |
| DRESS | Drug reaction with eosinophilia and systemic symptoms |
| SJS/TEN | Stevens-Johnson syndrome / toxic epidermal necrolysis |
| IVIG / SCIG | Intravenous / subcutaneous immunoglobulin |
| MAC | Membrane attack complex (C5b-9) |
| ALPS | Autoimmune lymphoproliferative syndrome |
| IPEX | Immune dysregulation, polyendocrinopathy, enteropathy, X-linked |
04 Allergic Rhinitis
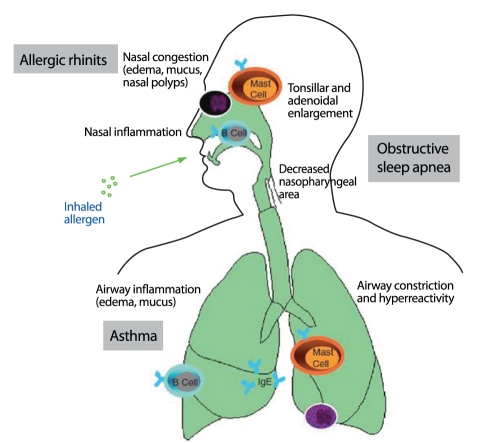
Allergic rhinitis (AR) affects 10–30% of adults and up to 40% of children worldwide. It is an IgE-mediated inflammatory condition of the nasal mucosa triggered by aeroallergens, causing sneezing, rhinorrhea, nasal congestion, and pruritus. AR significantly impairs quality of life, sleep, and work/school productivity and is the strongest risk factor for developing asthma.
Pathophysiology
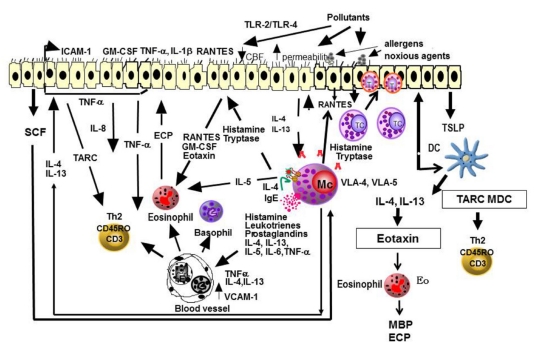
Inhaled allergens are processed by mucosal dendritic cells, which present peptides to Th2 cells. This drives IgE production by B cells, with IgE binding to FcεRI on nasal mast cells. Re-exposure causes allergen cross-linking of IgE, triggering the early-phase response (minutes: histamine, tryptase, prostaglandins, leukotrienes → sneezing, rhinorrhea, pruritus) and the late-phase response (4–8 hours: eosinophil and basophil recruitment via IL-5, RANTES, eotaxin → persistent nasal congestion). This late-phase inflammation underlies the concept of "priming" — repeated allergen exposure lowers the threshold for symptom triggering.
ARIA Classification
| Parameter | Intermittent | Persistent |
|---|---|---|
| Duration | <4 days/week OR <4 consecutive weeks | ≥4 days/week AND ≥4 consecutive weeks |
| Parameter | Mild | Moderate-Severe |
|---|---|---|
| Impact | None of the following impaired | ≥1 of: sleep disturbance, impairment of daily activities/sport/leisure, impairment of school/work, troublesome symptoms |
Management
Allergen avoidance is foundational but often insufficient alone. Intranasal corticosteroids (INCS) are first-line for moderate-severe AR (fluticasone propionate 1–2 sprays/nostril daily, mometasone, budesonide) — most effective single agent for all nasal symptoms including congestion. Second-generation antihistamines (cetirizine 10 mg, loratadine 10 mg, fexofenadine 180 mg daily) are first-line for mild AR and add-on for moderate-severe. Intranasal antihistamines (azelastine 1–2 sprays/nostril BID) have faster onset than INCS (15 min vs days). The combination of INCS + intranasal antihistamine (e.g., fluticasone/azelastine [Dymista]) is superior to either alone for moderate-severe AR. Montelukast 10 mg daily is modestly effective but carries an FDA black box warning for neuropsychiatric events (2020). Intranasal cromolyn is safe but less effective; useful in pregnancy. Allergen immunotherapy (SCIT or SLIT) is disease-modifying and indicated for moderate-severe AR inadequately controlled with pharmacotherapy.
05 Non-Allergic Rhinitis
Non-allergic rhinitis (NAR) accounts for ~25% of rhinitis cases and is a diagnosis of exclusion (negative skin testing/sIgE). It presents with nasal congestion and rhinorrhea but typically lacks sneezing and pruritus. Recognizing NAR subtypes prevents unnecessary allergen avoidance and immunotherapy.
Subtypes
| Subtype | Mechanism | Key Features | Treatment |
|---|---|---|---|
| Vasomotor rhinitis (idiopathic NAR) | Autonomic dysfunction, neural hyperreactivity | Triggered by temperature changes, strong odors, humidity, spicy foods; predominant congestion/rhinorrhea | Ipratropium nasal 0.03% (rhinorrhea), INCS (congestion), azelastine nasal |
| NARES (non-allergic rhinitis with eosinophilia) | Eosinophilic inflammation without IgE sensitization | Nasal eosinophilia >20% on smear, negative allergy testing; may progress to nasal polyps and AERD | INCS (first-line), responds well to topical steroids |
| Gustatory rhinitis | Vagally mediated | Profuse watery rhinorrhea triggered by eating (especially hot/spicy foods) | Ipratropium nasal 0.03% before meals |
| Rhinitis medicamentosa | Rebound congestion from topical decongestant overuse | History of oxymetazoline/phenylephrine use >3–5 days; severe rebound congestion upon discontinuation | Discontinue offending agent, INCS taper, short oral steroid burst if severe |
| Drug-induced rhinitis | Various mechanisms | ACE inhibitors, beta-blockers, NSAIDs, oral contraceptives, PDE-5 inhibitors | Discontinue/switch offending medication |
| Hormonal rhinitis | Estrogen/progesterone effects on nasal vasculature | Pregnancy rhinitis (affects ~20–30% of pregnant women, especially 2nd/3rd trimester), hypothyroidism | Saline, INCS if needed (budesonide preferred in pregnancy) |
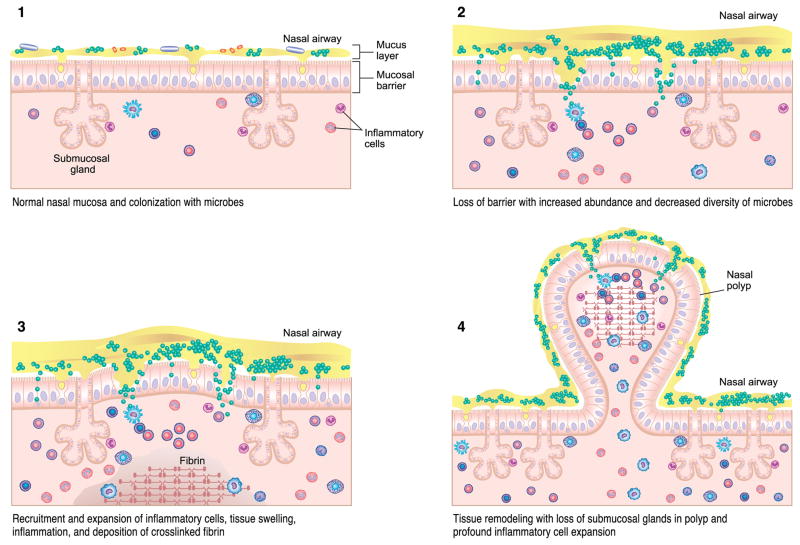
06 Chronic Rhinosinusitis & Nasal Polyps
Chronic rhinosinusitis (CRS) is defined as sinonasal inflammation lasting ≥12 weeks with at least 2 of: nasal obstruction/congestion, mucopurulent drainage, facial pain/pressure, and hyposmia/anosmia — confirmed by objective evidence (endoscopic findings or CT changes). CRS affects ~12% of adults and is divided into CRS without nasal polyps (CRSsNP) and CRS with nasal polyps (CRSwNP), which have distinct pathophysiology and treatment approaches.
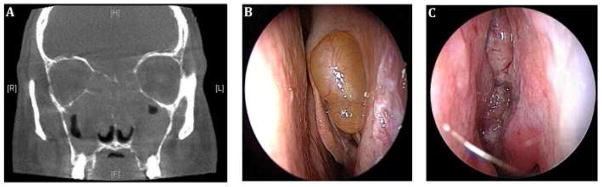
CRS Without Nasal Polyps (CRSsNP)
Predominantly Th1/neutrophilic inflammation. Often associated with recurrent bacterial sinusitis, anatomic obstruction (deviated septum, concha bullosa), and biofilm formation. Management: saline irrigations, INCS (mometasone or budesonide irrigations), short-course antibiotics for acute exacerbations, and functional endoscopic sinus surgery (FESS) for refractory cases.
CRS With Nasal Polyps (CRSwNP)
Predominantly Th2/eosinophilic inflammation with elevated tissue IL-4, IL-5, IL-13, and local IgE. Polyps arise from edematous mucosa in the middle meatus and ethmoid sinuses. Strongly associated with asthma (especially severe/eosinophilic), AERD (Samter's triad: nasal polyps + asthma + NSAID sensitivity), and allergic fungal sinusitis (AFS). Management: INCS (high-volume budesonide irrigations), short oral steroid courses (prednisone 0.5 mg/kg × 5–7 days for flares), FESS for refractory obstruction, and biologics for recurrent polyps post-surgery.
Biologics for CRSwNP
| Biologic | Target | Dose | Key Findings |
|---|---|---|---|
| Dupilumab | IL-4Rα (blocks IL-4 and IL-13) | 300 mg SC q2w | FDA-approved for CRSwNP; reduces polyp score, improves congestion, smell, and reduces need for surgery and oral steroids (SINUS-24/52 trials) |
| Omalizumab | IgE | 75–375 mg SC q2–4w (weight/IgE-based) | FDA-approved for CRSwNP; reduces nasal polyp score and congestion (POLYP 1/2 trials) |
| Mepolizumab | IL-5 | 100 mg SC q4w | FDA-approved for CRSwNP; reduces polyp recurrence post-surgery (SYNAPSE trial) |
Allergic Fungal Sinusitis (AFS)
AFS is a subset of CRSwNP caused by a hypersensitivity response to colonizing fungi (most commonly Aspergillus, Alternaria, Bipolaris). Diagnostic criteria (Bent-Kuhn): (1) nasal polyposis, (2) eosinophilic mucin with fungal hyphae on histology, (3) positive fungal stain or culture, (4) type I hypersensitivity to fungi (positive SPT or sIgE), and (5) characteristic CT findings (unilateral or asymmetric opacification with areas of hyperattenuation/"double density" sign). Treatment requires FESS for debridement, followed by INCS irrigations, systemic steroids for recurrence, and possibly antifungal therapy (itraconazole) in refractory cases.
07 Asthma Pathophysiology & Phenotypes
Asthma is a chronic inflammatory airway disease characterized by variable airflow obstruction, bronchial hyperresponsiveness, and airway remodeling. It affects ~300 million people worldwide with prevalence of 5–10% in adults and up to 15% in children.
Pathophysiology
Allergen exposure in genetically predisposed individuals drives Th2 inflammation: IL-4 and IL-13 stimulate goblet cell hyperplasia, mucus hypersecretion, and smooth muscle contraction; IL-5 recruits eosinophils; IgE sensitizes airway mast cells. Chronic inflammation leads to airway remodeling: subepithelial fibrosis, smooth muscle hypertrophy, neovascularization, and goblet cell metaplasia, causing fixed airflow obstruction. Airway hyperresponsiveness (AHR) — exaggerated bronchoconstriction to stimuli (cold air, exercise, methacholine) — is a hallmark feature.
Asthma Phenotypes
| Phenotype | Age of Onset | Key Features | Biomarkers |
|---|---|---|---|
| Allergic (atopic) asthma | Childhood | Most common phenotype (~60%); associated with AR, AD, food allergy; allergen-triggered; good response to ICS and biologics | Elevated IgE, blood/sputum eosinophilia, positive SPT, elevated FeNO |
| Eosinophilic (non-allergic) asthma | Adult-onset | Severe, often steroid-dependent; associated with CRSwNP/AERD; not IgE-driven | Blood eos ≥300/μL, sputum eos ≥3%, elevated FeNO, normal IgE |
| Exercise-induced bronchoconstriction (EIB) | Any | Bronchoconstriction 5–15 min after vigorous exercise; worse in cold/dry air; ≥10% FEV1 drop on exercise challenge | Often normal between episodes |
| Aspirin-exacerbated respiratory disease (AERD) | Adult (20s–40s) | Samter's triad: asthma + nasal polyps + NSAID sensitivity; severe, often requires oral steroids | Elevated urinary LTE4, blood eosinophilia |
| Obesity-related asthma | Adult | BMI ≥30; often non-eosinophilic; less responsive to ICS; mechanical restriction + systemic inflammation | Often low FeNO, low eosinophils; neutrophilic sputum |
| Neutrophilic (paucigranulocytic) asthma | Adult | Poor response to ICS; associated with smoking, occupational exposures, infection | Sputum neutrophilia ≥61%, low eosinophils, low FeNO |
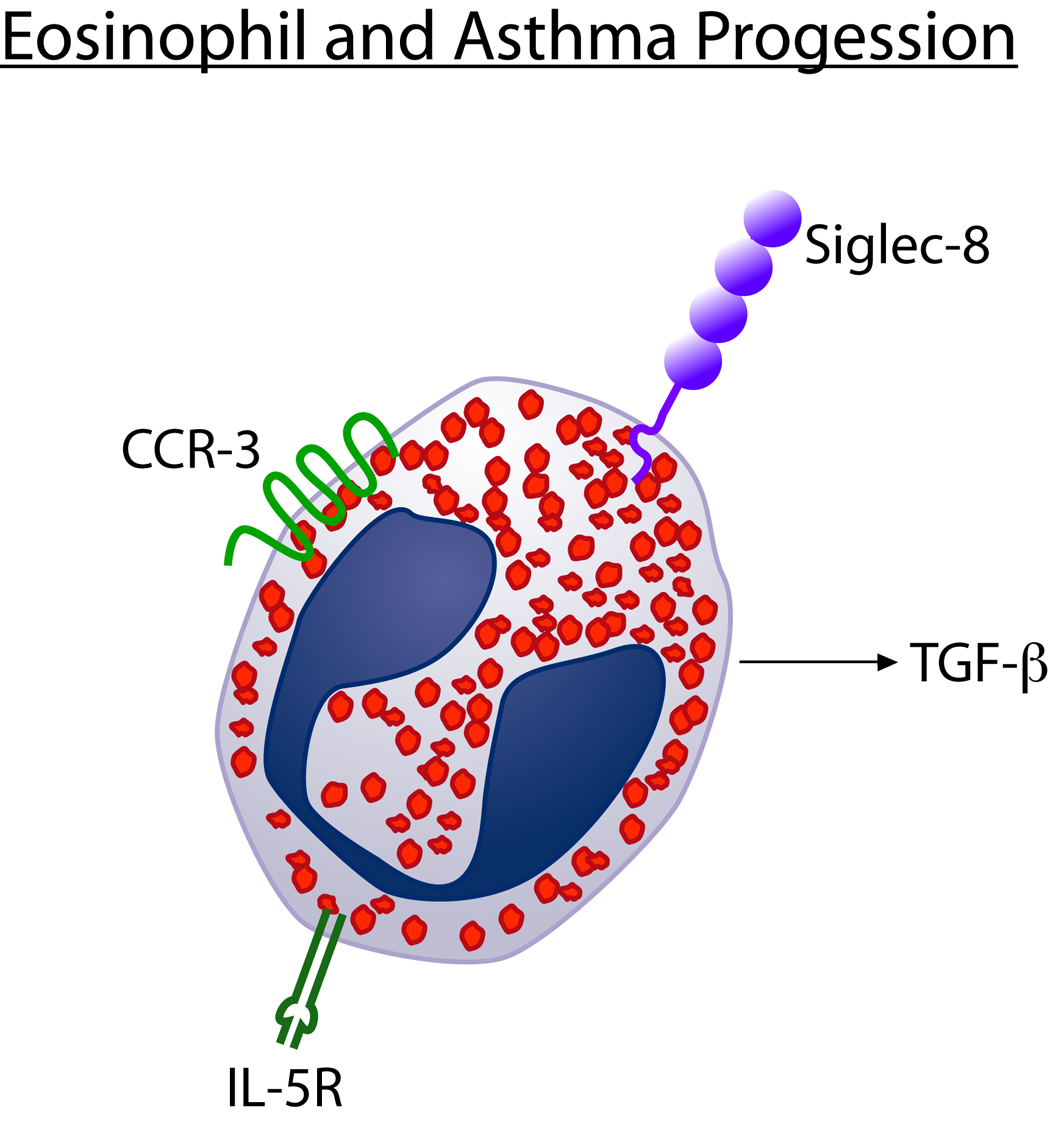
08 Asthma Severity & Stepwise Management
GINA/NAEPP Severity Classification (Treatment-Naïve Patients)
| Parameter | Intermittent | Mild Persistent | Moderate Persistent | Severe Persistent |
|---|---|---|---|---|
| Symptom frequency | ≤2 days/week | >2 days/week but not daily | Daily | Throughout the day |
| Nighttime awakenings | ≤2×/month | 3–4×/month | >1×/week but not nightly | Often 7×/week |
| SABA use | ≤2 days/week | >2 days/week but not daily | Daily | Several times/day |
| Activity limitation | None | Minor | Some limitation | Extremely limited |
| FEV1 (% predicted) | >80% | ≥80% | 60–80% | <60% |
| FEV1/FVC | Normal | Normal | Reduced 5% | Reduced >5% |
| Exacerbations | 0–1/year | ≥2/year | ≥2/year | ≥2/year |
GINA Stepwise Approach (Adults/Adolescents ≥12 years, 2023 Update)
| Step | Preferred Controller | Alternative Controller | Reliever |
|---|---|---|---|
| Step 1 | Low-dose ICS-formoterol PRN | Low-dose ICS whenever SABA taken | As-needed low-dose ICS-formoterol |
| Step 2 | Low-dose ICS-formoterol PRN (or daily low-dose ICS) | LTRA or low-dose ICS when SABA taken | As-needed low-dose ICS-formoterol |
| Step 3 | Low-dose ICS-LABA (maintenance) | Medium-dose ICS, or low-dose ICS + LTRA | As-needed low-dose ICS-formoterol |
| Step 4 | Medium-dose ICS-LABA | Add tiotropium or high-dose ICS-LABA | As-needed low-dose ICS-formoterol |
| Step 5 | High-dose ICS-LABA + add-on (LAMA, biologic, or low-dose OCS) | Phenotype-guided biologic therapy | As-needed low-dose ICS-formoterol |
Asthma Control Assessment (GINA)
| In the Past 4 Weeks | Well-Controlled | Partly Controlled | Uncontrolled |
|---|---|---|---|
| Daytime symptoms >2×/week? | None of these | 1–2 of these | 3–4 of these |
| Night waking due to asthma? | |||
| SABA reliever >2×/week? | |||
| Activity limitation due to asthma? |
09 Acute Asthma Exacerbation
Signs of severity: Cannot speak in full sentences, accessory muscle use, RR >30, HR >120, SpO2 <90%, FEV1 or PEF <25% predicted, silent chest, altered mental status, cyanosis, bradycardia (impending arrest).
Immediate management:
- Oxygen: Target SpO2 93–95% (adults), 94–98% (children)
- Inhaled SABA: Albuterol nebulizer 2.5–5 mg q20min × 3 or continuous nebulization 10–15 mg/hr; or MDI 4–8 puffs q20min × 3
- Ipratropium: 0.5 mg nebulized q20min × 3 (added to SABA for severe exacerbations)
- Systemic corticosteroids: Methylprednisolone 125 mg IV or prednisone 40–60 mg PO (onset 4–6 hours); give within 1 hour of presentation
- Magnesium sulfate: 2 g IV over 20 min for severe exacerbation unresponsive to initial treatment (FEV1 <25%)
- If impending respiratory arrest: Prepare for intubation (use ketamine for induction — bronchodilator properties; avoid histamine-releasing agents), consider IV terbutaline (loading dose 10 μg/kg), heliox
Exacerbation Severity Classification
| Feature | Mild | Moderate | Severe | Life-Threatening |
|---|---|---|---|---|
| Breathlessness | Walking | Talking (prefers sitting) | At rest (hunched forward) | Drowsy/confused |
| Talks in | Sentences | Phrases | Words only | Unable to speak |
| RR | Increased | Increased | >30/min | Paradoxical breathing |
| HR | <100 | 100–120 | >120 | Bradycardia |
| PEF (% predicted) | >70% | 40–69% | <40% | <25% or unable |
| SpO2 | >95% | 90–95% | <90% | <90% |
| PaCO2 | <42 mmHg | <42 mmHg | ≥42 mmHg | >45 mmHg |
Disposition Criteria
Discharge criteria: PEF ≥70% predicted, SpO2 ≥94% on room air, able to speak in full sentences, adequate medication understanding, and access to follow-up within 1–2 days. Discharge with: oral prednisone 40–50 mg daily × 5–7 days, SABA PRN, and controller therapy (initiate or step up ICS). Admission criteria: PEF <40% after initial treatment, persistent hypoxemia, prior ICU admission for asthma, multiple ED visits, or concerning social factors. ICU criteria: PEF <25%, PaCO2 ≥42 mmHg, altered mental status, hemodynamic instability, or need for non-invasive/invasive ventilation.
10 Asthma Biologics & Advanced Therapies
Biologic therapies target specific inflammatory pathways in severe asthma (GINA Step 5) and have transformed management of patients with T2-high disease. Selection is guided by phenotyping (blood eosinophils, FeNO, IgE, comorbidities).
| Biologic | Target | Mechanism | Indication / Criteria | Dose | Key Trial(s) |
|---|---|---|---|---|---|
| Omalizumab (Xolair) | Free IgE | Binds circulating IgE, preventing binding to FcεRI on mast cells/basophils | Moderate-severe allergic asthma; total IgE 30–1500 IU/mL + positive perennial aeroallergen SPT/sIgE; age ≥6 | 75–375 mg SC q2–4w (dosed by weight + IgE level) | INNOVATE, EXTRA |
| Mepolizumab (Nucala) | IL-5 | Anti-IL-5 monoclonal Ab; reduces eosinophil maturation and survival | Severe eosinophilic asthma; blood eos ≥150/μL at initiation or ≥300 in past 12 months; age ≥6 | 100 mg SC q4w (adults); 40 mg SC q4w (6–11 y) | MENSA, DREAM, MUSCA |
| Benralizumab (Fasenra) | IL-5Rα | Binds IL-5 receptor α-subunit; induces near-complete eosinophil depletion via ADCC (antibody-dependent cell-mediated cytotoxicity) | Severe eosinophilic asthma; blood eos ≥300/μL; age ≥12 | 30 mg SC q4w × 3, then q8w | SIROCCO, CALIMA, ZONDA |
| Dupilumab (Dupixent) | IL-4Rα | Blocks IL-4 and IL-13 signaling (shared receptor subunit); reduces eosinophilic inflammation, mucus, and IgE | Moderate-severe eosinophilic asthma or OCS-dependent asthma; blood eos ≥150 or FeNO ≥25 ppb; age ≥6 | Loading 400 mg (or 600 mg), then 200 mg (or 300 mg) SC q2w | LIBERTY ASTHMA QUEST, VENTURE |
| Tezepelumab (Tezspire) | TSLP (thymic stromal lymphopoietin) | Blocks upstream epithelial alarmin TSLP; reduces multiple inflammatory pathways (T2 and non-T2) | Severe asthma regardless of phenotype; NO eosinophil or IgE requirement; age ≥12 | 210 mg SC q4w | NAVIGATOR, SOURCE, CASCADE |
Other Advanced Therapies
Long-acting muscarinic antagonists (LAMA): Tiotropium Respimat 2.5 μg/day is add-on at Step 4–5; reduces exacerbations independent of eosinophil status. Azithromycin 250–500 mg 3×/week may reduce exacerbations in non-eosinophilic asthma (AMAZES trial). Bronchial thermoplasty delivers radiofrequency energy to reduce airway smooth muscle mass; considered for severe refractory asthma not responsive to biologics (AIR2 trial showed reduction in severe exacerbations and ED visits).
11 Asthma-COPD Overlap & Special Phenotypes
Asthma-COPD Overlap (ACO)
ACO describes patients with persistent airflow limitation (post-bronchodilator FEV1/FVC <0.70) and features of both asthma (reversibility, eosinophilia, atopy, variable symptoms) and COPD (smoking history, age >40, progressive decline). ACO affects ~15–25% of patients with obstructive lung disease and carries worse outcomes than either condition alone (more exacerbations, faster FEV1 decline, poorer quality of life). Treatment: ICS-LABA is cornerstone (unlike pure COPD, where ICS are not first-line); add LAMA for persistent symptoms; avoid SABA-only or LABA-only; consider blood eosinophil levels to guide ICS use.
Occupational Asthma
Accounts for 10–25% of adult-onset asthma. Two forms: sensitizer-induced (IgE-mediated or T-cell-mediated; requires latency period; >400 known agents including isocyanates, flour dust, latex, animal dander) and irritant-induced (reactive airways dysfunction syndrome [RADS]; no latency, follows massive irritant exposure). Diagnosis requires demonstrating temporal relationship to workplace (serial PEF monitoring over 2–4 weeks), specific inhalation challenge, or positive sIgE to occupational allergen. Complete removal from exposure is the ideal treatment; continued exposure leads to irreversible obstruction.
Exercise-Induced Bronchoconstriction (EIB)
Occurs in up to 90% of asthmatics and 10–15% of the general population (especially elite athletes). Diagnosis: ≥10% fall in FEV1 from baseline within 30 minutes of a standardized exercise challenge (6–8 min of vigorous exercise at 85–90% max HR). Management: SABA 15 min before exercise (most effective single agent), daily ICS for athletes with frequent EIB, LTRA as alternative or add-on, adequate warm-up (induces refractory period of ~2 hours).
12 Anaphylaxis
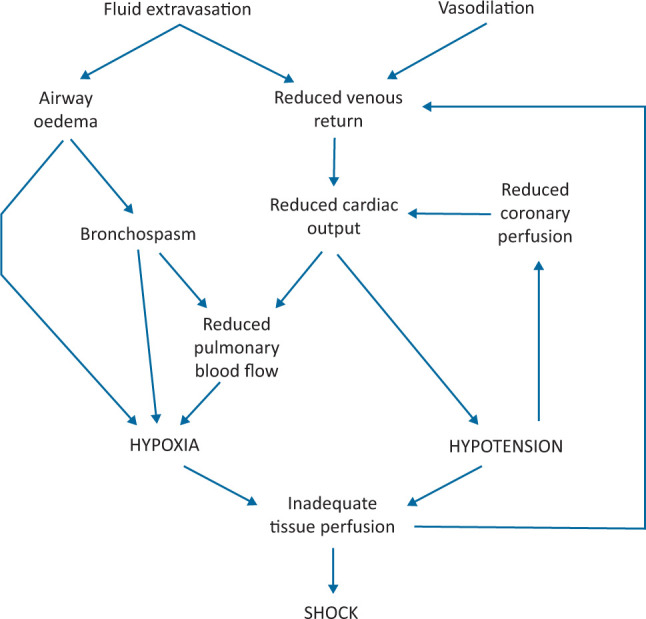
Anaphylaxis is an acute, potentially fatal, multi-organ system hypersensitivity reaction. Incidence is ~50–112 episodes per 100,000 person-years; lifetime prevalence is 0.5–2%. Most common triggers are foods (children), medications (adults), and stinging insects. Mortality rate is 0.5–1% of anaphylaxis cases, with death occurring most commonly from airway compromise (food-triggered) or cardiovascular collapse (drug/venom-triggered).
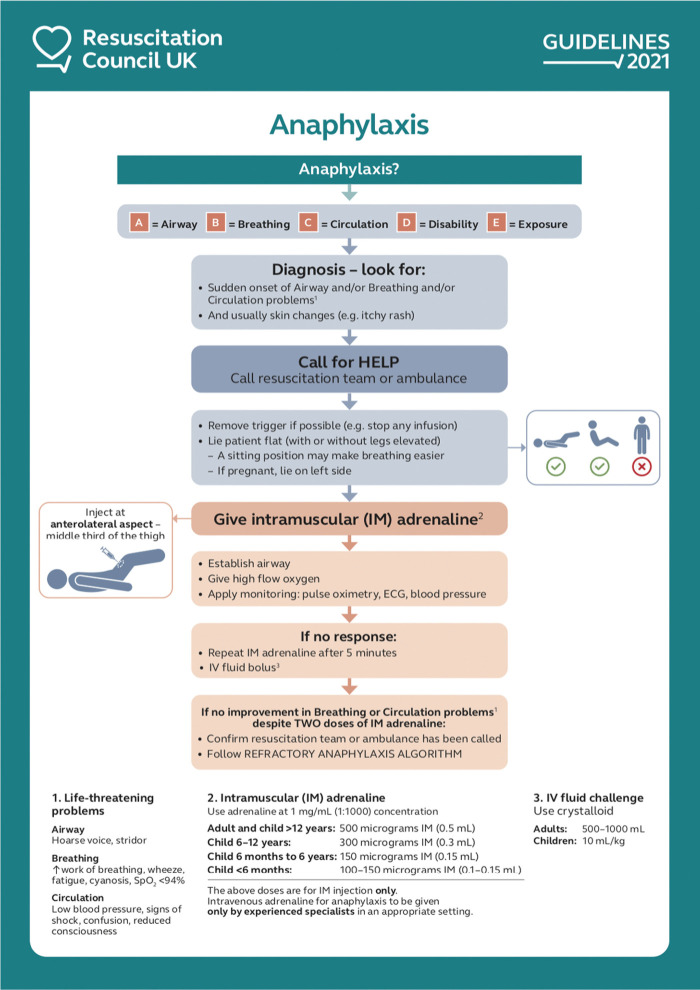
Step 1: Remove trigger if possible (stop infusion, remove stinger)
Step 2: EPINEPHRINE IM (anterolateral thigh) — this is the ONLY first-line treatment:
- Adults and children >30 kg: 0.3–0.5 mg (1:1000 = 1 mg/mL) IM
- Children 15–30 kg: 0.15 mg IM (EpiPen Jr)
- Infants <15 kg: 0.01 mg/kg IM
- Repeat q5–15 min as needed — most fatalities involve delayed or withheld epinephrine
Step 3: Position supine with legs elevated (Trendelenburg) unless respiratory distress (sit upright) or vomiting (recovery position). NEVER sit up or stand a hypotensive anaphylaxis patient — can cause cardiac arrest ("empty ventricle syndrome").
Step 4: Adjunctive measures:
- Oxygen: High-flow via non-rebreather mask
- IV access: Two large-bore IVs; NS bolus 1–2 L (adults) or 20 mL/kg (children)
- Albuterol nebulizer for bronchospasm refractory to epinephrine
- H1 blocker: Diphenhydramine 25–50 mg IV/IM (adjunct only — does NOT treat hypotension or airway edema)
- H2 blocker: Famotidine 20 mg IV (optional adjunct)
- Glucocorticoid: Methylprednisolone 125 mg IV (may reduce biphasic reactions, no proven acute benefit)
- Glucagon 1–5 mg IV for patients on beta-blockers (refractory to epinephrine)
Refractory anaphylaxis: Epinephrine IV infusion 0.1–1 μg/kg/min (NOT IM 1:1000 IV push), vasopressin, cardiac monitoring, ICU admission.
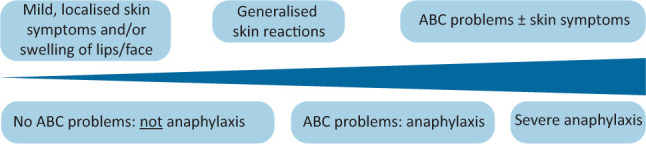
NIAID/FAAN Diagnostic Criteria for Anaphylaxis
Anaphylaxis is highly likely when any 1 of 3 criteria is met:
Acute onset (minutes to hours) of illness with skin/mucosal involvement (urticaria, pruritus, flushing, lip/tongue/uvula swelling) PLUS at least one of: (a) respiratory compromise (dyspnea, wheeze, stridor, hypoxemia), (b) hypotension or end-organ dysfunction (collapse, syncope, incontinence).
Two or more of the following occurring rapidly after exposure to a likely allergen: (a) skin/mucosal involvement, (b) respiratory compromise, (c) hypotension or end-organ dysfunction, (d) persistent GI symptoms (crampy abdominal pain, vomiting).
Hypotension after exposure to a known allergen for that patient: SBP <90 mmHg (adults) or >30% decrease from baseline; age-specific low SBP in children.

Biphasic Anaphylaxis
A second phase of symptoms occurs in 1–20% of cases (most estimates 5–10%), typically 1–72 hours after resolution of the initial episode (median 8–10 hours). Risk factors: severe initial reaction, delayed epinephrine, unknown trigger, and previous biphasic reactions. All patients should be observed for at least 4–6 hours post-treatment (some guidelines recommend 12–24 hours for severe reactions). Prescribe epinephrine auto-injectors (minimum 2) and develop an anaphylaxis action plan at discharge.
13 Urticaria (Acute & Chronic)
Urticaria presents as pruritic, erythematous, raised wheals (hives) that are transient (<24 hours per individual lesion) and migratory. Classified as acute (<6 weeks duration) or chronic (≥6 weeks).
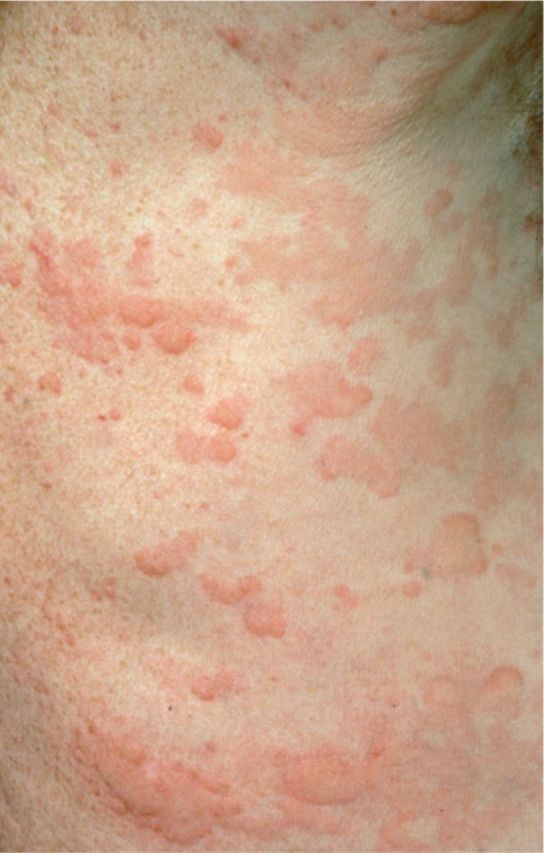
Acute Urticaria
Affects ~20% of people at some point in life. Most common causes: viral infections (especially in children), foods (nuts, shellfish, eggs, milk), medications (antibiotics, NSAIDs, opioids), insect stings, and latex. Often self-limited. Treatment: second-generation H1 antihistamine (cetirizine, loratadine, fexofenadine); if inadequate, add first-generation H1 antihistamine at night (hydroxyzine 25 mg, diphenhydramine 25–50 mg); short oral steroid course for severe episodes (prednisone 40 mg × 3–5 days).
Chronic Spontaneous Urticaria (CSU)
CSU occurs without a consistent external trigger. Most cases (80–90%) are idiopathic; autoimmune mechanisms (IgG anti-FcεRI or anti-IgE autoantibodies) are found in ~30–50% of cases. Associated with thyroid autoimmunity (check anti-TPO). Workup: CBC, ESR/CRP, TSH, anti-TPO; further testing only if clinical suspicion (e.g., complement levels, cryoglobulins, hepatitis serologies). Routine allergy testing is NOT indicated for CSU.
CSU Stepwise Management (EAACI/GA2LEN/WAO Guidelines)
| Step | Therapy | Details |
|---|---|---|
| Step 1 | Standard-dose second-gen H1 antihistamine | Cetirizine 10 mg, loratadine 10 mg, or fexofenadine 180 mg daily |
| Step 2 | Updose antihistamine (up to 4× standard) | e.g., cetirizine 10 mg QID (40 mg/day); safe and FDA-approved off-label; re-evaluate at 2–4 weeks |
| Step 3 | Add omalizumab | 150–300 mg SC q4w; onset 1–4 weeks; FDA-approved for CSU age ≥12 (ASTERIA I/II, GLACIAL trials); ~65–70% respond |
| Step 4 | Add cyclosporine | 3–5 mg/kg/day; monitor renal function, BP, drug levels; reserved for omalizumab failures |
14 Angioedema
Angioedema is localized, non-pitting swelling of deep dermal, subcutaneous, or submucosal tissue. The critical distinction is between histaminergic (mast cell–mediated) and bradykinin-mediated angioedema, as treatment differs fundamentally.
Histaminergic vs Bradykinin-Mediated Angioedema
| Feature | Histaminergic | Bradykinin-Mediated |
|---|---|---|
| Urticaria | Often present | Absent |
| Pruritus | Common | Absent (may have tingling/burning) |
| Onset | Minutes to hours | Hours (gradual) |
| Duration | 24–48 hours | 48–72 hours (longer) |
| Response to epinephrine | Yes | Minimal/none |
| Response to antihistamines | Yes | No |
| Response to corticosteroids | Yes (adjunctive) | No |
| Causes | Allergic reactions, NSAIDs, idiopathic | ACE inhibitors, HAE |
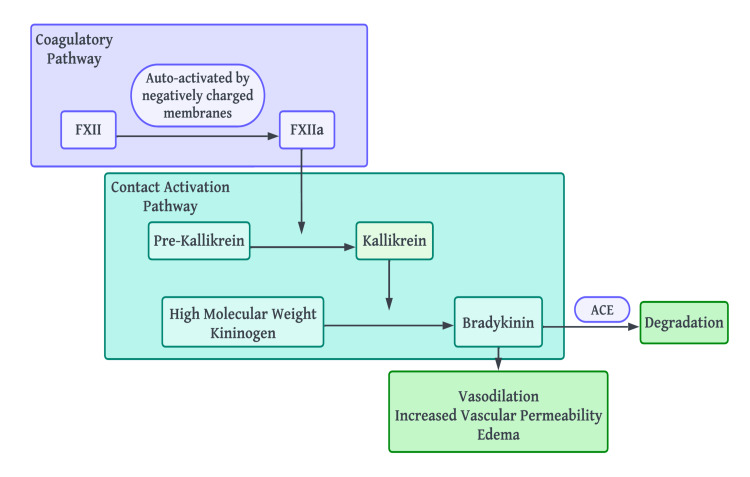
ACE Inhibitor–Induced Angioedema
Occurs in 0.1–0.7% of ACE inhibitor users; 3–5× more common in Black patients. Can occur at any point during treatment (even after years of use). Mechanism: ACE normally degrades bradykinin; inhibition leads to bradykinin accumulation. Treatment: discontinue ACE inhibitor permanently (do NOT substitute another ACE inhibitor; ARBs are generally safe with ~1–2% cross-reactivity). For acute episodes: supportive care, airway management, consider icatibant (off-label) or fresh frozen plasma. Antihistamines, epinephrine, and steroids are ineffective.
Hereditary Angioedema (HAE)
| Type | C4 | C1-INH Level | C1-INH Function | Mechanism |
|---|---|---|---|---|
| HAE Type I (~85%) | Low | Low (<50% normal) | Low | Decreased C1-INH production (quantitative deficiency) |
| HAE Type II (~15%) | Low | Normal or elevated | Low (<50%) | Dysfunctional C1-INH (qualitative deficiency) |
| HAE with normal C1-INH | Normal | Normal | Normal | Factor XII mutations, plasminogen mutations, or unknown; estrogen-sensitive |
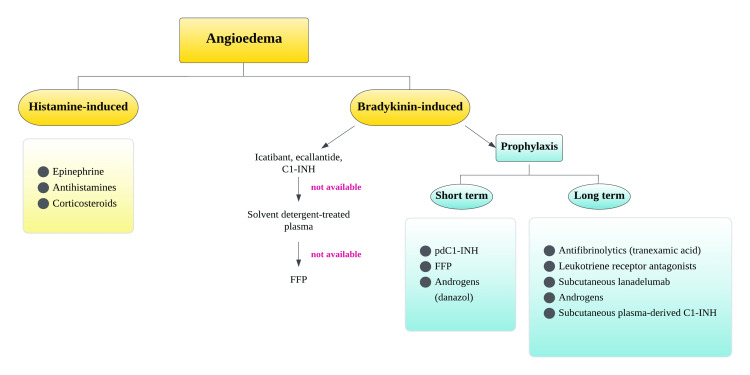
HAE Treatment
Acute attack treatment: C1-INH concentrate (Berinert 20 IU/kg IV, or Cinryze 1000 IU IV), icatibant (bradykinin B2 receptor antagonist, 30 mg SC), ecallantide (kallikrein inhibitor, 30 mg SC), or recombinant C1-INH (Ruconest 50 IU/kg IV). FFP is an alternative if specific therapies are unavailable. Long-term prophylaxis: lanadelumab (anti-kallikrein monoclonal Ab, 300 mg SC q2w; reduced attacks by 87% in HELP trial), berotralstat (oral kallikrein inhibitor, 150 mg PO daily; APEX trial), C1-INH concentrate prophylaxis (Cinryze 1000 IU IV q3–4 days, or Haegarda 60 IU/kg SC 2×/week). Short-term prophylaxis (before dental/surgical procedures): C1-INH concentrate 1–2 hours before procedure.
15 Drug Hypersensitivity & Desensitization
Gell-Coombs Classification of Hypersensitivity
| Type | Name | Mechanism | Timing | Examples |
|---|---|---|---|---|
| Type I | Immediate / IgE-mediated | IgE cross-linking on mast cells/basophils → degranulation | Minutes to hours | Penicillin anaphylaxis, food allergy, allergic rhinitis, allergic asthma |
| Type II | Cytotoxic / antibody-mediated | IgG/IgM bind cell-surface antigens → complement activation, ADCC, opsonization | Hours to days | Drug-induced hemolytic anemia (penicillin, methyldopa), drug-induced thrombocytopenia (heparin-HIT, quinine), Goodpasture's |
| Type III | Immune complex | Antigen-antibody complexes deposit in tissues → complement activation → inflammation | Days to weeks | Serum sickness, drug-induced vasculitis, SLE nephritis, Arthus reaction |
| Type IV | Delayed / T-cell-mediated | Sensitized T cells release cytokines or directly kill target cells | 24–72 hours (up to weeks) | Contact dermatitis, DRESS, SJS/TEN, drug-induced hepatitis, tuberculin (PPD) skin test |
Penicillin Allergy
~10% of patients report penicillin allergy, but >90% are NOT truly allergic upon evaluation. Penicillin allergy evaluation reduces unnecessary use of broad-spectrum antibiotics (vancomycin, fluoroquinolones, carbapenems) and decreases healthcare costs, C. difficile, and antimicrobial resistance. Evaluation: History risk-stratify into low-risk (remote, non-severe reactions >10 years ago), moderate-risk (urticaria/mild symptoms), and high-risk (anaphylaxis, DRESS, SJS/TEN). Low-risk: direct oral amoxicillin challenge (500 mg, observe 1 hour). Moderate-risk: skin testing with penicilloyl polylysine (major determinant), penicillin G (minor determinant), ampicillin, amoxicillin; negative predictive value ~97–99%; if negative, followed by oral amoxicillin challenge. High-risk (SJS/TEN, DRESS): do NOT skin test or challenge — permanently avoid penicillins and carbapenems.
Drug Desensitization
Drug desensitization is a temporary induction of tolerance to a medication to which a patient has a type I (IgE-mediated) hypersensitivity. It works by gradual, incrementally increasing sub-threshold doses that do not trigger mast cell degranulation. It is indicated when there is no adequate alternative (e.g., penicillin for neurosyphilis, aspirin for AERD, chemotherapy hypersensitivity, insulin allergy). Desensitization is performed in a monitored setting (ICU or step-down unit) with epinephrine at bedside. Typical protocols involve 12–16 steps doubling the dose every 15–30 minutes. The desensitized state is temporary — if the drug is interrupted for >2 half-lives, the patient reverts to the allergic state and requires re-desensitization.
Severe Cutaneous Adverse Drug Reactions (SCARs)
| Reaction | Timing | Key Features | Common Culprits | Management |
|---|---|---|---|---|
| SJS | 1–3 weeks | Mucosal involvement + skin detachment <10% BSA; targetoid lesions; Nikolsky sign + | Allopurinol, sulfonamides, anticonvulsants (carbamazepine, phenytoin, lamotrigine), NSAIDs | Discontinue culprit, supportive care (burn unit), IVIG or cyclosporine considered |
| TEN | 1–3 weeks | Same as SJS but skin detachment >30% BSA; mortality 25–35% (SCORTEN predicts mortality) | Same as SJS | Same as SJS; burn unit care, strict avoid re-exposure |
| DRESS | 2–8 weeks | Diffuse morbilliform rash, facial edema, fever, eosinophilia >1500, lymphocytosis, internal organ involvement (hepatitis 80%, nephritis, pneumonitis); HHV-6 reactivation | Anticonvulsants (carbamazepine, phenytoin), allopurinol, sulfonamides, dapsone, minocycline | Discontinue culprit, systemic steroids (prednisone 1 mg/kg with slow taper over 6–8 weeks); monitor for delayed autoimmune sequelae (thyroiditis, diabetes) |
16 Serum Sickness & Immune Complex Reactions
Serum sickness is a type III hypersensitivity reaction caused by circulating immune complexes that deposit in blood vessels and tissues, activating complement and causing inflammation. Classic serum sickness follows administration of heterologous serum (e.g., antithymocyte globulin, equine-derived antivenoms) or, less commonly, certain drugs.
Clinical Features
Onset 7–21 days after initial exposure (shorter with re-exposure). Classic triad: fever, urticaria/rash (often at hands/feet borders), and polyarthralgia/arthritis (multiple joints, migratory). Other features: lymphadenopathy, splenomegaly, glomerulonephritis, vasculitis. Laboratory findings: low C3 and C4, elevated ESR, proteinuria, positive circulating immune complexes. Treatment is usually self-limited after trigger removal; NSAIDs or prednisone 0.5–1 mg/kg/day tapered over 2–3 weeks for severe cases.
Serum Sickness–Like Reaction (SSLR)
SSLR is a clinically similar but immunologically distinct entity (no immune complexes, complement levels normal). Most common in children after cefaclor, but also seen with other beta-lactams, bupropion, and infliximab. Presents with fever, rash (urticarial or morbilliform), and arthralgias 1–3 weeks after drug exposure. Self-limited upon drug discontinuation; treated symptomatically with antihistamines and short steroid course if needed.
17 IgE-Mediated Food Allergy
IgE-mediated food allergy affects ~6–8% of children and ~3–4% of adults. It causes reproducible symptoms within minutes to 2 hours of ingestion. Nine foods account for >90% of food allergies in the US: milk, egg, peanut, tree nuts, wheat, soy, fish, shellfish, and sesame (added to major allergen list in 2023).
Clinical Manifestations
| System | Symptoms |
|---|---|
| Skin (most common, ~80%) | Urticaria, angioedema, flushing, pruritus, eczema flare |
| GI | Oral pruritus, nausea, vomiting, abdominal pain, diarrhea |
| Respiratory | Nasal congestion, rhinorrhea, sneezing, cough, wheeze, stridor, dyspnea |
| Cardiovascular | Hypotension, tachycardia, dizziness, syncope, cardiac arrest |
Diagnosis
Diagnosis requires clinical history of reproducible symptoms + evidence of sensitization (positive SPT or sIgE). Neither SPT nor sIgE alone confirms clinical allergy (sensitization ≠ allergy). Component-resolved diagnostics (CRD) improves specificity: e.g., peanut Ara h 2 sIgE ≥0.35 kU/L has >95% PPV for clinical peanut allergy; Ara h 8 (PR-10 protein) positivity with negative Ara h 2 suggests oral allergy syndrome only (low anaphylaxis risk). The oral food challenge (OFC) is the gold standard — open or double-blind, placebo-controlled food challenge (DBPCFC) in a supervised setting with graded doses over 2–4 hours, with observation period of 2 hours post-last dose.
Natural History
Most childhood milk, egg, wheat, and soy allergies resolve by age 5–10 (milk ~80% by age 16, egg ~70%). Peanut, tree nut, fish, and shellfish allergies are more persistent (~80% lifelong for peanut; ~90% for tree nut). Declining sIgE levels over time predict clinical tolerance; periodic re-evaluation with OFC is appropriate.
Food Allergy Prevention
The LEAP trial (2015) demonstrated that early introduction of peanut (at 4–6 months) in high-risk infants (those with severe eczema and/or egg allergy) reduced peanut allergy by 81% compared to avoidance. Current guidelines (AAP, NIAID): introduce peanut protein early (~4–6 months) in high-risk infants, after SPT or sIgE screening; no restriction of allergenic foods during pregnancy or lactation.
Oral Immunotherapy (OIT)
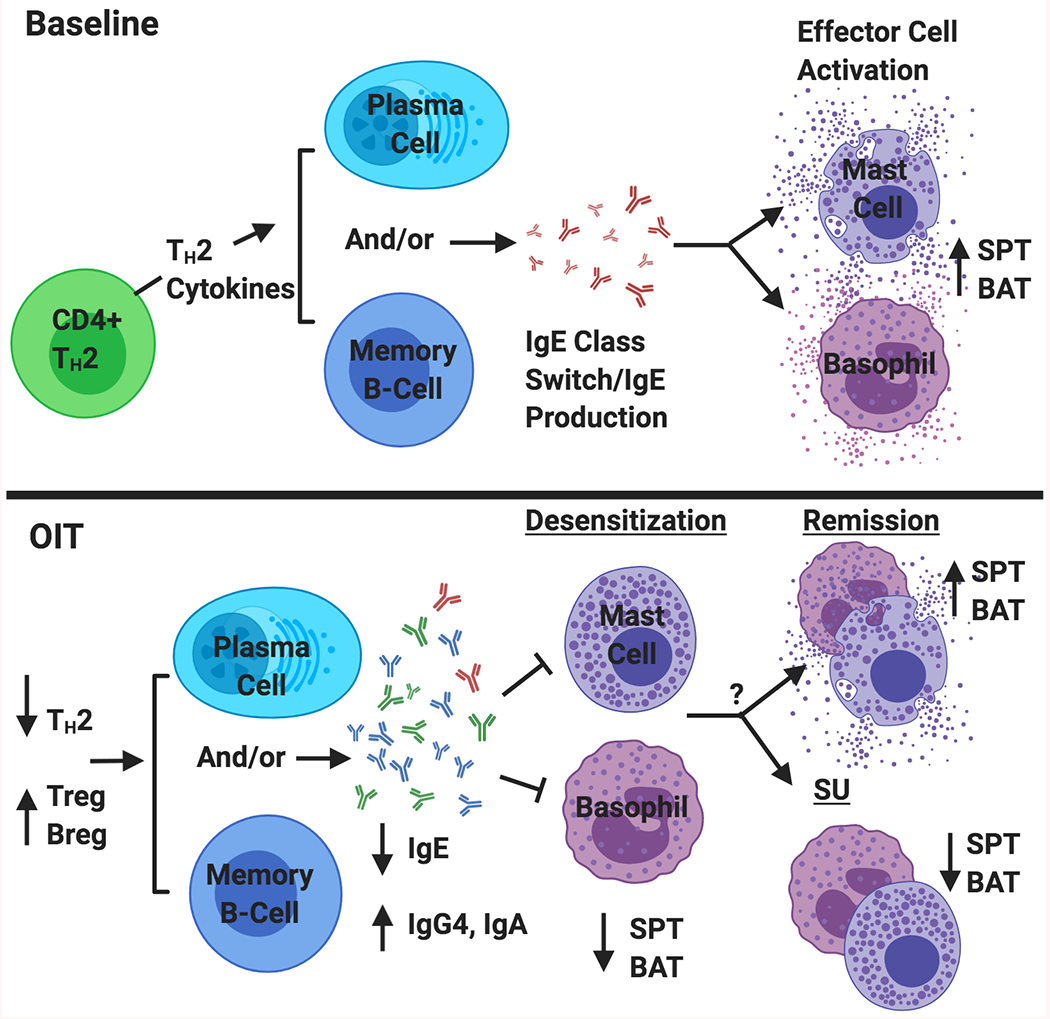
Palforzia (peanut allergen powder) is FDA-approved for peanut allergy ages 4–17 years. Protocol: initial dose escalation (day 1, observed, starting at 0.5 mg), up-dosing (biweekly escalation over ~6 months to 300 mg maintenance), then daily maintenance (300 mg indefinitely). PALISADE trial showed 67% of treated patients tolerated 600 mg peanut protein at exit OFC vs 4% placebo. Risks: GI side effects (~90%), anaphylaxis (~14% during up-dosing), eosinophilic esophagitis (~3%). Patients must continue to carry epinephrine and avoid exercise 2 hours after dosing.
18 Non-IgE Food Allergy (FPIES) & Mixed Disorders
Food Protein–Induced Enterocolitis Syndrome (FPIES)
FPIES is a non-IgE-mediated food allergy presenting in infancy (typically <9 months) with profuse vomiting 1–4 hours after ingestion of a trigger food, often followed by diarrhea, lethargy, and pallor. Severe episodes can cause dehydration, hypothermia, methemoglobinemia, and shock-like presentation (15–20% of acute episodes). Most common triggers: cow's milk, soy, rice, oats. Diagnosis is clinical (no reliable biomarker; sIgE and SPT are usually negative). OFC is confirmatory but not always needed if history is classic. Treatment of acute episodes: IV fluids, ondansetron 0.15 mg/kg IV/IM (shown to stop vomiting in acute FPIES). Most children outgrow FPIES by age 3–5 (milk/soy by 1–3 years; rice/oat by 3–5 years).
Food Protein–Induced Allergic Proctocolitis (FPIAP)
Benign condition in breast-fed infants presenting with mucousy, blood-streaked stools in an otherwise healthy, thriving infant. Most commonly triggered by cow's milk protein in maternal diet. Diagnosis is clinical. Management: maternal elimination of cow's milk (and sometimes soy, egg); if formula-fed, switch to extensively hydrolyzed formula or amino acid formula. Resolves by 12 months in most cases.
Food Protein–Induced Enteropathy (FPE)
Non-IgE-mediated small bowel inflammation causing chronic diarrhea, malabsorption, failure to thrive, and hypoalbuminemia in infants. Most common trigger: cow's milk. Villous atrophy on biopsy (mimics celiac disease but no anti-tTG/anti-DGP antibodies). Treatment: avoidance of trigger; most outgrow by age 2–3.
19 Eosinophilic Esophagitis
Eosinophilic esophagitis (EoE) is a chronic, immune/antigen-mediated esophageal disease characterized by symptoms of esophageal dysfunction and eosinophilic infiltration of the esophageal mucosa. Prevalence has increased dramatically (~1 in 2000); predominantly affects males (3:1), with peak incidence in the 30s–40s.
Clinical Presentation
Adults: dysphagia (most common, ~70%), food impaction (15–20% present as emergency food bolus), chest pain, heartburn refractory to PPI. Children: feeding difficulties, vomiting, failure to thrive, abdominal pain. Strongly associated with atopy (~70% have concomitant allergic diseases: allergic rhinitis, asthma, AD, food allergy, or peripheral eosinophilia).
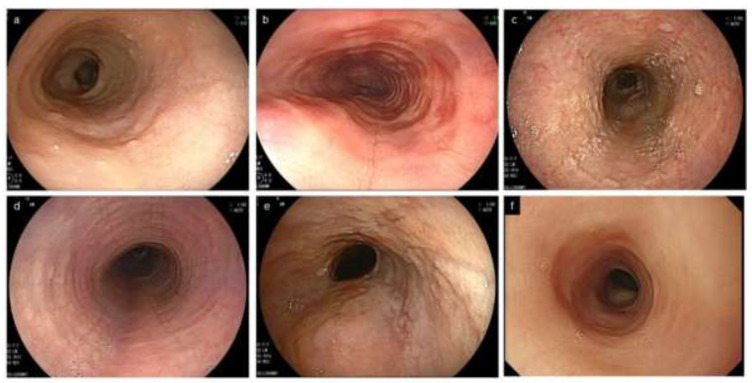
Diagnosis
Requires both: (1) symptoms of esophageal dysfunction AND (2) esophageal biopsy showing ≥15 eosinophils per high-power field (eos/hpf) in ≥1 biopsy specimen. Take biopsies from proximal AND distal esophagus (2–4 biopsies from each location). PPI-responsive esophageal eosinophilia is now considered part of the EoE spectrum (not an exclusion). Endoscopic findings: linear furrows, white plaques/exudates, concentric rings ("trachealization"), edema, stricture, narrow-caliber esophagus (crepe paper mucosa).
Management
| Therapy | Details | Efficacy |
|---|---|---|
| PPI therapy | Omeprazole 20–40 mg BID or equivalent × 8 weeks | ~50% histologic response; considered first-line by some guidelines |
| Swallowed topical corticosteroids | Fluticasone MDI 440–880 μg BID (swallowed, not inhaled; no spacer; no eating/drinking 30 min after) or budesonide viscous slurry 1–2 mg BID | ~65–70% histologic response; mainstay of pharmacologic therapy |
| Dietary elimination | Six-food elimination diet (remove milk, wheat, egg, soy, fish/shellfish, nuts) or 2–4 food elimination (milk and wheat most common triggers); systematic reintroduction with biopsy | 6-FED: ~70–75% response; 2-FED (milk + wheat): ~40–50% |
| Dupilumab | 300 mg SC weekly; first FDA-approved biologic for EoE (2022); ages ≥12 and ≥40 kg | 60% histologic response (<6 eos/hpf) at 24 weeks (Part A of TREET trial) |
| Esophageal dilation | For symptomatic strictures; gradual dilation; does not treat underlying inflammation | Immediate symptom relief; combine with pharmacologic/dietary therapy |
20 Oral Allergy Syndrome & Alpha-Gal Syndrome
Oral Allergy Syndrome (Pollen-Food Allergy Syndrome)
OAS results from cross-reactivity between aeroallergen pollen proteins and homologous proteins in raw fruits, vegetables, and nuts. Symptoms are typically limited to the oropharynx: oral/pharyngeal pruritus, tingling, and mild swelling of lips/tongue/palate within minutes of eating raw trigger foods. Symptoms resolve spontaneously within 30 minutes. Systemic reactions are rare (<2%). Cooking denatures the cross-reactive proteins and is usually tolerated.
| Pollen | Cross-Reactive Foods | Protein Family |
|---|---|---|
| Birch | Apple, peach, cherry, pear, plum, carrot, celery, hazelnut, almond, soy | PR-10 (Bet v 1 homologues) |
| Ragweed | Melon (cantaloupe, watermelon, honeydew), banana, zucchini, cucumber | Profilins |
| Grass | Tomato, potato, melon, orange, peach | Profilins |
| Mugwort | Celery, carrot, parsley, coriander, sunflower seeds, mustard | Lipid transfer proteins (LTPs) |
Alpha-Gal Syndrome (Mammalian Meat Allergy)
Alpha-gal syndrome is an IgE-mediated allergy to galactose-α-1,3-galactose (alpha-gal), an oligosaccharide present on non-primate mammalian tissues. Sensitization occurs through Lone Star tick (Amblyomma americanum) bites, which introduce alpha-gal from tick saliva. Unique features: delayed reaction onset (2–6 hours after eating mammalian meat — beef, pork, lamb), which is unusual for IgE-mediated allergy (explained by time required for fat digestion and alpha-gal liberation). Symptoms range from urticaria/GI symptoms to anaphylaxis. Diagnosis: positive sIgE to alpha-gal (≥0.35 kU/L) + clinical history. Management: avoidance of mammalian meat, dairy (some patients tolerate dairy), gelatin, and certain medications derived from mammalian sources (cetuximab, gelatin-containing vaccines, heparin). Patients must carry epinephrine. Some patients lose sensitivity over 1–5 years if tick re-exposure is avoided.
21 Allergic Contact Dermatitis
Allergic contact dermatitis (ACD) is a type IV (delayed, T-cell-mediated) hypersensitivity reaction to an external substance (hapten) that contacts the skin. It requires prior sensitization (1–2 week induction period) and manifests 24–72 hours after re-exposure. ACD affects ~15–20% of the general population and is a leading cause of occupational skin disease.
Common Contact Allergens
| Allergen | Sources | Distribution Pattern |
|---|---|---|
| Nickel sulfate (#1 worldwide) | Jewelry (earrings, belt buckles), watches, snaps, cell phones, coins | Earlobes, wrists, periumbilical, areas of metal contact |
| Fragrance mix | Perfumes, cosmetics, soaps, detergents, topical products | Face, neck, hands, axillae |
| Balsam of Peru (Myroxylon pereirae) | Cosmetics, flavorings, topical medications, dental products | Perioral, hands |
| Urushiol | Poison ivy, poison oak, poison sumac (Toxicodendron spp.) | Linear streaks on exposed areas (hands, arms, legs, face) |
| Neomycin | Topical antibiotics (Neosporin), ear/eye drops | Application sites; often co-sensitized with bacitracin |
| Formaldehyde / releasers | Cosmetics, nail products, fabric finishes, building materials | Eyelids, face, hands |
| Methylisothiazolinone (MI) | Preservative in cosmetics, paints, wet wipes, household products | Face, hands; epidemic increase since 2010 |
| Paraphenylenediamine (PPD) | Hair dye, black henna tattoos, rubber | Scalp line, ears, neck; can cause severe dermatitis |
| Cobalt chloride | Metals (often co-sensitized with nickel), cement, paints | Similar distribution to nickel |
| Potassium dichromate | Cement, leather, paints; occupational in construction workers | Hands, feet (leather shoes) |
Diagnosis — Patch Testing
Patch testing is the gold standard for diagnosing ACD. The standard series (T.R.U.E. Test or expanded North American Contact Dermatitis Group series, 70–80+ allergens) is applied to the upper back under occlusion for 48 hours. Readings at 48 hours (upon removal) and 72–96 hours (delayed reading, most important). Results graded: negative, irritant reaction, 1+ (erythema + papules), 2+ (erythema + papules + vesicles), 3+ (bullous reaction). Clinical relevance must be established (does the positive patch test explain the patient's dermatitis distribution and exposure history?).
Management
Allergen avoidance is the definitive treatment. Provide patients with specific avoidance lists (e.g., CAMP database for nickel-free products). Acute ACD: mid- to high-potency topical corticosteroids (triamcinolone 0.1% for body, hydrocortisone or desonide for face/intertriginous areas). Widespread/severe ACD: systemic prednisone 0.5–1 mg/kg/day tapered over 2–3 weeks (shorter courses risk rebound, especially with poison ivy). Chronic ACD: calcineurin inhibitors (tacrolimus 0.1%, pimecrolimus 1%) as steroid-sparing alternatives. Barrier creams for occupational prevention.
22 Atopic Dermatitis
Atopic dermatitis (AD) is a chronic, relapsing, intensely pruritic inflammatory skin disease affecting up to 20% of children and 7–10% of adults. It is the first manifestation of the "atopic march" (AD → food allergy → allergic rhinitis → asthma) and results from genetic (filaggrin mutations in ~30%), immunologic (Th2-skewed skin inflammation), and barrier dysfunction (transepidermal water loss).
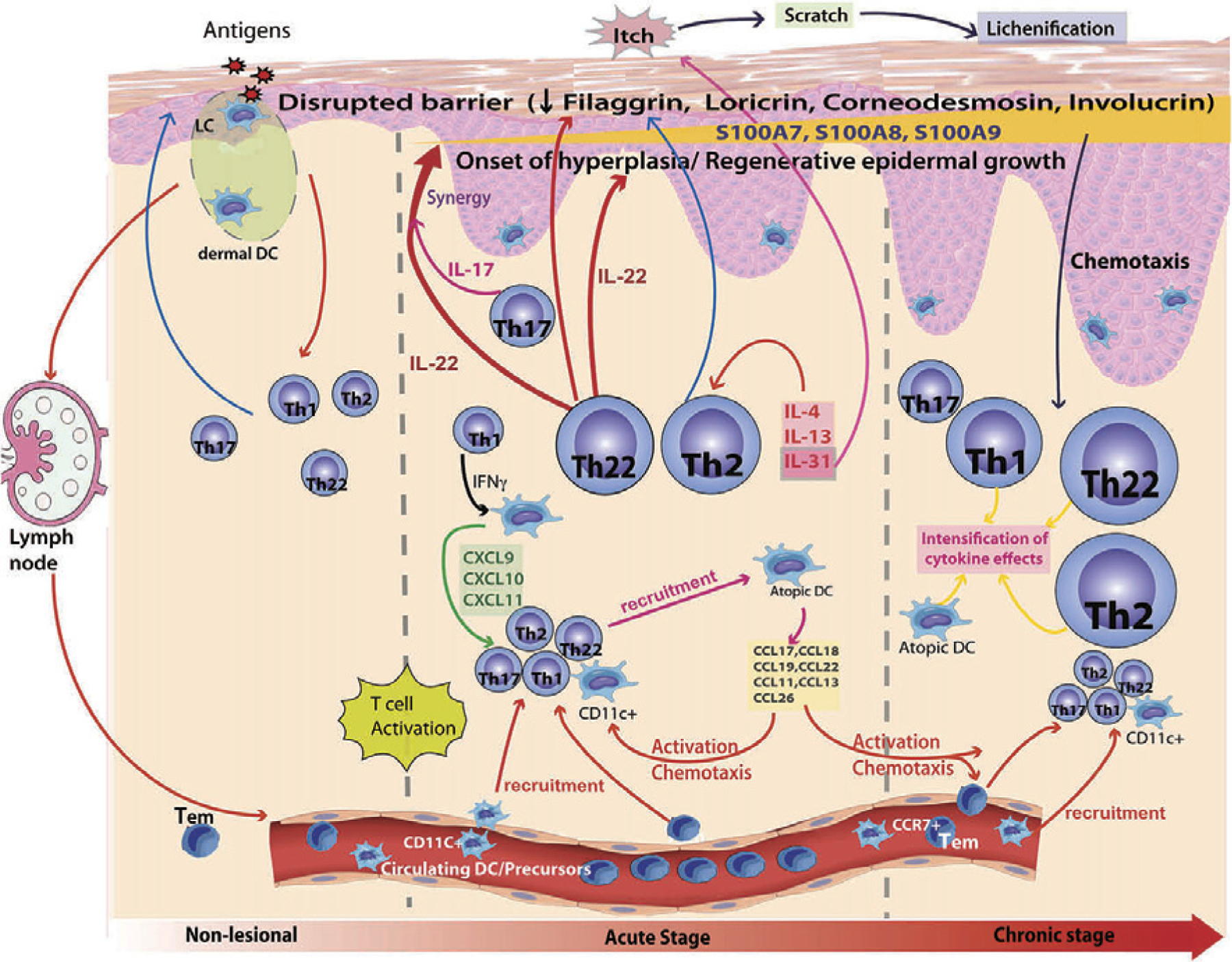
Diagnostic Criteria (Hanifin-Rajka, Simplified)
Must have pruritus plus ≥3 of: (1) typical morphology and distribution (flexural in older children/adults; facial/extensor in infants), (2) chronic or relapsing course, (3) personal or family history of atopy, (4) xerosis (dry skin). No definitive laboratory test; diagnosis is clinical. Elevated total IgE and peripheral eosinophilia are common but non-specific.
Severity Assessment
| Severity | SCORAD | EASI | Features |
|---|---|---|---|
| Mild | <25 | 1–7 | Dry skin, infrequent pruritus, minimal impact on daily life |
| Moderate | 25–50 | 7–21 | Frequent pruritus, lichenification, sleep disturbance, moderate QoL impact |
| Severe | >50 | >21 | Widespread eczema, intense pruritus, significant sleep and psychosocial impact, secondary infection |
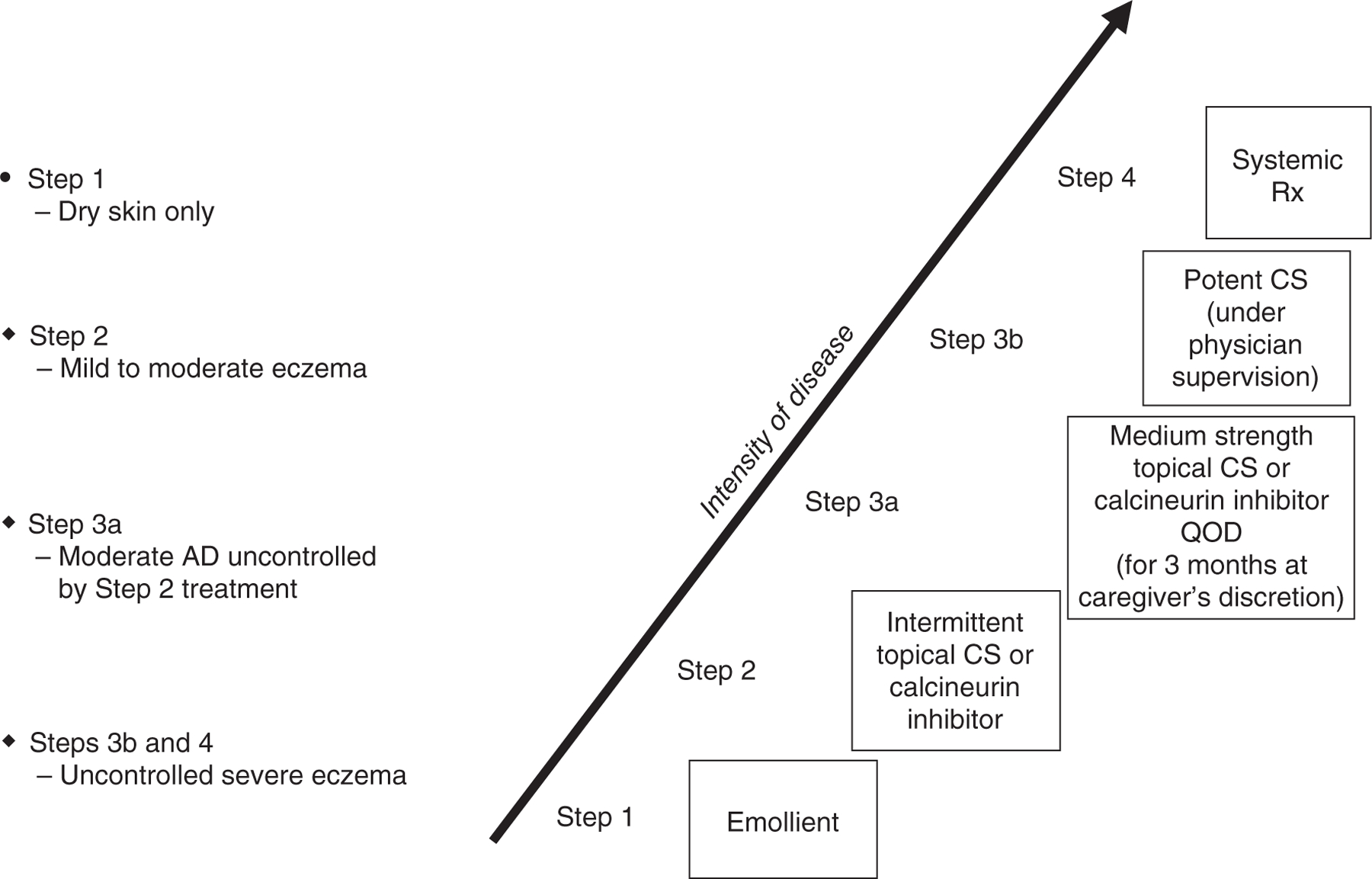
Management Ladder
| Severity | Treatment |
|---|---|
| All severities (baseline) | Daily emollient therapy (ceramide-based preferred, apply within 3 min of bathing); gentle cleanser; avoid triggers (irritants, allergens, stress, heat); "soak and seal" technique |
| Mild | Low-potency TCS (hydrocortisone 2.5%) for face/intertriginous; medium-potency TCS (triamcinolone 0.1%) for body; topical calcineurin inhibitors (tacrolimus 0.03%/0.1%, pimecrolimus 1%) for steroid-sparing or sensitive areas |
| Moderate | Medium- to high-potency TCS (fluocinonide 0.05%, betamethasone valerate 0.1%); tacrolimus 0.1%; crisaborole 2% (PDE4 inhibitor); ruxolitinib 1.5% cream (JAK inhibitor, FDA-approved for mild-moderate AD) |
| Moderate-severe (refractory) | Wet wrap therapy; phototherapy (narrow-band UVB 2–3×/week); systemic agents |
| Severe / systemic therapy | Dupilumab (first-line systemic: 600 mg loading, then 300 mg SC q2w; age ≥6 months); tralokinumab (IL-13 inhibitor, 600 mg loading, 300 mg SC q2w); abrocitinib (oral JAK1 inhibitor, 100–200 mg PO daily); upadacitinib (oral JAK inhibitor, 15–30 mg PO daily). Older agents: cyclosporine 3–5 mg/kg/day (short-term), methotrexate 7.5–25 mg/week, mycophenolate 1–3 g/day, azathioprine |
23 Latex Allergy
Latex allergy is an IgE-mediated (type I) hypersensitivity to natural rubber latex proteins (Hevea brasiliensis). Prevalence: 1–6% in the general population, 10–17% in healthcare workers, up to 73% in children with spina bifida (repeated surgical exposures). Can cause contact urticaria, rhinitis, asthma, and anaphylaxis. Cross-reactivity with plant proteins produces latex-fruit syndrome: banana, avocado, chestnut, kiwi (highest cross-reactivity).
Diagnosis
SPT with commercial latex extract (where available); sIgE to latex (k82); CRD: Hev b 5 and Hev b 6.01 (most associated with latex anaphylaxis), Hev b 8 (profilin — cross-reactive, lower risk). Patch testing diagnoses type IV (delayed) latex allergy (contact dermatitis from rubber accelerators, not latex protein).
Management
Complete latex avoidance: non-latex gloves (nitrile, vinyl), latex-free medical supplies, MedicAlert identification. Healthcare facilities should maintain latex-free operating rooms. Premedication (antihistamines + steroids) does NOT prevent IgE-mediated anaphylaxis and should NOT replace avoidance. Patients must carry epinephrine auto-injectors.
24 B-Cell (Antibody) Deficiencies
Antibody deficiencies are the most common primary immunodeficiencies, accounting for ~50% of all PIDs. They present with recurrent sinopulmonary infections (pneumonia, sinusitis, otitis media) from encapsulated organisms (S. pneumoniae, H. influenzae), GI infections (Giardia), and bronchiectasis from chronic infection.
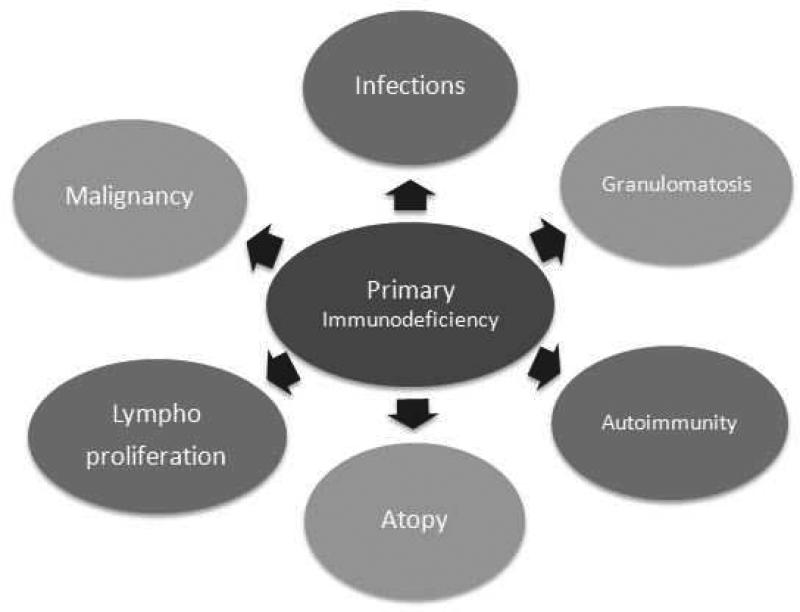
Major B-Cell Deficiencies
| Disorder | Inheritance | Defect | Ig Levels | B Cells | Key Features |
|---|---|---|---|---|---|
| X-Linked Agammaglobulinemia (XLA, Bruton's) | X-linked | BTK (Bruton tyrosine kinase) mutation → B-cell maturation arrest at pre-B stage | All Ig classes absent or severely reduced | Absent (<2% CD19+) | Males; infections begin 6–9 months (after maternal IgG wanes); absent tonsils/lymph nodes; NO autoimmunity (no B cells to make autoantibodies) |
| Common Variable Immunodeficiency (CVID) | Most sporadic; some AD (TACI, ICOS, BAFFR mutations) | Defective B-cell differentiation into plasma cells | IgG <500 mg/dL + low IgA and/or IgM | Normal or low | Most common symptomatic PID (~1:25,000); diagnosis age 20–40; sinopulmonary infections + autoimmune cytopenias (ITP, AIHA in ~25%) + granulomatous disease + increased lymphoma risk (50×) |
| Selective IgA Deficiency | Variable | Failure of B-cell class switch to IgA | IgA <7 mg/dL; IgG and IgM normal | Normal | Most common PID (~1:500 in Caucasians); most patients asymptomatic; increased sinopulmonary and GI infections; associated with celiac disease (10–15× risk), autoimmune diseases; risk of anaphylaxis to blood products containing IgA (rare) |
| Transient Hypogammaglobulinemia of Infancy | N/A | Delayed IgG production | Low IgG (physiologic nadir 3–6 months); normalizes by age 2–4 | Normal | Exaggerated physiologic nadir; usually benign; distinguish from more severe PIDs by monitoring |
| Specific Antibody Deficiency | Variable | Inability to mount antibody response to polysaccharide antigens despite normal Ig levels | Normal quantitative Ig | Normal | Recurrent sinopulmonary infections; diagnosed by absent response to Pneumovax (pre/post-titers); responds to IVIG in severe cases |
25 T-Cell & Combined Immunodeficiencies

Severe Combined Immunodeficiency (SCID)
SCID is a pediatric emergency — untreated, it is fatal within the first 1–2 years of life. SCID presents with severe, recurrent infections from bacteria, viruses, fungi, and opportunistic organisms (PJP, CMV, Candida) beginning in the first months of life, chronic diarrhea, failure to thrive, and absent thymic shadow on chest X-ray. SCID is detected by newborn screening using T-cell receptor excision circles (TRECs) — absent or very low TRECs indicates T-cell lymphopenia. Definitive treatment is hematopoietic stem cell transplant (HSCT) — survival >90% if performed before 3.5 months of age (before infections occur). Gene therapy is available for ADA-SCID and X-linked SCID.
| SCID Type | Inheritance | Defect | T Cells | B Cells | NK Cells |
|---|---|---|---|---|---|
| X-linked SCID (most common, ~45%) | X-linked | γc chain (IL2RG) — shared by IL-2, IL-4, IL-7, IL-9, IL-15, IL-21 receptors | Absent | Present (non-functional) | Absent |
| ADA deficiency (~15%) | AR | Adenosine deaminase → toxic purine metabolite accumulation | Absent | Absent | Absent |
| JAK3 deficiency | AR | JAK3 kinase (downstream of γc chain) | Absent | Present | Absent |
| IL-7Rα deficiency | AR | IL-7 receptor α-chain | Absent | Present | Present |
| RAG1/RAG2 deficiency | AR | Recombination-activating genes (V(D)J recombination) | Absent | Absent | Present |
DiGeorge Syndrome (22q11.2 Deletion)
DiGeorge syndrome results from 22q11.2 microdeletion causing defective development of the 3rd and 4th pharyngeal pouches. Classic features (mnemonic: CATCH-22): Cardiac defects (conotruncal: tetralogy of Fallot, interrupted aortic arch, truncus arteriosus, VSD), Abnormal facies, Thymic hypoplasia/aplasia, Cleft palate, Hypocalcemia (absent parathyroids). Immunodeficiency ranges from mild T-cell lymphopenia (partial DiGeorge, ~75% of cases, often improves with age) to complete DiGeorge (absent T cells, SCID-like, requires thymus transplant). Diagnosis: FISH or chromosomal microarray for 22q11.2 deletion.
Other Combined Immunodeficiencies
| Disorder | Defect | Key Features |
|---|---|---|
| Wiskott-Aldrich Syndrome | WAS gene (X-linked); defective WASp protein → abnormal cytoskeleton in hematopoietic cells | Classic triad: eczema, thrombocytopenia (small platelets), recurrent infections; elevated IgA and IgE; increased lymphoma risk; treat with HSCT |
| Hyper-IgE Syndrome (Job's) | STAT3 (AD) or DOCK8 (AR) mutations | AD-HIES: markedly elevated IgE (>2000), recurrent "cold" staphylococcal abscesses (no inflammation), eczema, pneumonia with pneumatoceles, retained primary teeth, coarse facies, scoliosis. AR (DOCK8): severe viral infections, malignancy |
| Ataxia-Telangiectasia | ATM gene (AR); defective DNA repair | Cerebellar ataxia (progressive), oculocutaneous telangiectasias, combined immunodeficiency (low IgA, IgG2), elevated AFP, radiation sensitivity, increased malignancy (lymphoma, leukemia) |
| Hyper-IgM Syndrome | CD40L (X-linked, most common) or AID/UNG (AR) | Elevated or normal IgM, absent IgG/IgA/IgE (failure of class-switch recombination); recurrent sinopulmonary infections + Pneumocystis + Cryptosporidium (sclerosing cholangitis) |
26 Phagocytic & Complement Deficiencies
Phagocytic Defects
| Disorder | Defect | Inheritance | Key Features | Diagnosis |
|---|---|---|---|---|
| Chronic Granulomatous Disease (CGD) | NADPH oxidase deficiency → absent respiratory burst → cannot kill catalase-positive organisms | X-linked (65–70%) or AR | Recurrent infections with catalase-positive organisms: S. aureus, Aspergillus, Serratia, Nocardia, Burkholderia cepacia; granuloma formation in lungs, liver, GI tract; colitis mimicking Crohn's | Dihydrorhodamine (DHR) flow cytometry (gold standard) or nitroblue tetrazolium (NBT) test |
| Leukocyte Adhesion Deficiency Type I (LAD-I) | CD18 (β2-integrin) deficiency → neutrophils cannot migrate to infection sites | AR | Delayed umbilical cord separation (>30 days), severe bacterial infections WITHOUT pus formation, markedly elevated WBC (>100,000 during infection), poor wound healing | Flow cytometry: absent CD18/CD11b on neutrophils |
| Chédiak-Higashi Syndrome | LYST gene → defective lysosomal trafficking → giant granules in neutrophils | AR | Partial oculocutaneous albinism, recurrent pyogenic infections, peripheral neuropathy, giant granules on peripheral smear; risk of "accelerated phase" (HLH-like) | Peripheral smear: giant azurophilic granules in neutrophils/all WBCs |
| Cyclic Neutropenia | ELANE gene mutation | AD | Periodic neutropenia (ANC <200) every ~21 days lasting 3–7 days; recurrent oral ulcers, fever, infections during nadirs | Serial CBCs 2–3×/week × 6 weeks documenting cyclical pattern |
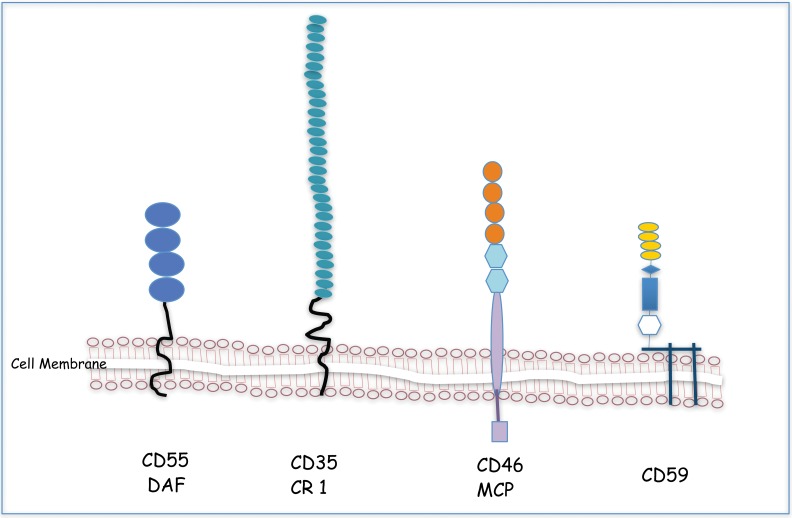
Complement Deficiencies
| Deficiency | Associated Conditions | Lab Findings |
|---|---|---|
| C1q, C1r/s, C2, C4 (early classical) | SLE-like disease (~90% for C1q deficiency; decreasing association down the pathway); encapsulated bacterial infections | CH50 = 0; low C3 and C4 (via classical pathway) |
| C2 deficiency (most common complement deficiency) | SLE, recurrent sinopulmonary infections with encapsulated bacteria | CH50 = 0; normal AH50; low C2 level |
| C3 deficiency | Severe recurrent pyogenic infections (all encapsulated organisms); glomerulonephritis | CH50 = 0; AH50 = 0; undetectable C3 |
| C5–C9 (terminal/MAC) | Recurrent Neisseria infections (meningococcal meningitis, gonococcal bacteremia) — up to 5000× increased risk | CH50 = 0; AH50 = 0; normal C3 and C4 |
| C1-INH deficiency | Hereditary angioedema (see Section 14) | Low C4 (screening); low C1-INH level or function |
| MBL deficiency | Recurrent infections in early childhood; usually clinically mild in immunocompetent adults | Normal CH50 and AH50; specific MBL level low |
27 Immunoglobulin Replacement Therapy
Immunoglobulin replacement is the cornerstone of treatment for antibody deficiencies (XLA, CVID, specific antibody deficiency, secondary hypogammaglobulinemia). The goal is to maintain trough IgG levels ≥500 mg/dL (some experts target ≥700–800 mg/dL, especially if infections continue).
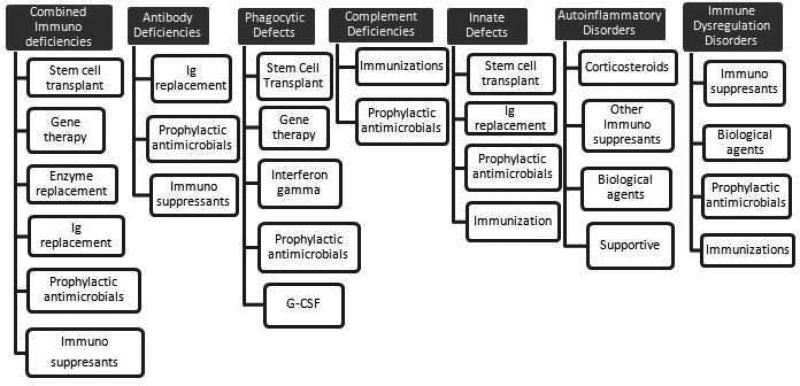
IVIG vs SCIG
| Parameter | IVIG | SCIG |
|---|---|---|
| Route | Intravenous | Subcutaneous (abdomen, thighs, upper arms) |
| Frequency | Every 3–4 weeks | Weekly (or biweekly with hyaluronidase-facilitated SCIG [HyQvia]) |
| Typical dose | 400–600 mg/kg/month | 100–200 mg/kg/week (equivalent monthly dose) |
| Peak/trough variation | Large peaks and troughs | Steady-state levels (fewer systemic side effects) |
| Administration | Requires IV access, 2–4 hour infusions in clinic or home | Self-administered at home via infusion pump; shorter training period |
| Systemic reactions | More common: headache (most common), fever, chills, myalgia, nausea; rare: aseptic meningitis, thrombosis, hemolysis, anaphylaxis (especially IgA-deficient patients) | Less common; mainly local site reactions (swelling, erythema) |
| Premedication | Acetaminophen, diphenhydramine, and/or hydrocortisone for patients with prior reactions | Usually not needed |
Monitoring
Check trough IgG level before each IVIG dose (or periodically for SCIG) until steady state is reached. Adjust dose to maintain trough ≥500–800 mg/dL. Monitor renal function (risk of osmotic nephrosis with sucrose-containing products), CBC (hemolysis screening), LFTs, and clinical infection frequency. Annual pulmonary function testing and CT chest to monitor for bronchiectasis progression.
28 Secondary Immunodeficiency
Secondary (acquired) immunodeficiencies are far more common than PIDs and result from external factors that impair immune function.
Major Causes
| Category | Examples | Immune Defect | Key Infections |
|---|---|---|---|
| HIV/AIDS | CD4+ T-cell depletion by HIV | Progressive cellular immunodeficiency; CD4 <200 = AIDS | PJP, toxoplasmosis, CMV, MAC, cryptococcosis, candidiasis, Kaposi sarcoma (HHV-8) |
| Iatrogenic (medications) | Corticosteroids (prednisone ≥20 mg/day × ≥2 weeks), rituximab (anti-CD20), cyclophosphamide, calcineurin inhibitors, mycophenolate, JAK inhibitors | Varies by agent: rituximab → B-cell depletion and hypogammaglobulinemia; steroids → broad immunosuppression; JAK inhibitors → impaired lymphocyte signaling | Opportunistic infections proportional to degree/duration of immunosuppression |
| Hematologic malignancy | CLL, multiple myeloma, lymphoma | CLL: hypogammaglobulinemia (50–70%); myeloma: immunoparesis (decreased uninvolved Ig) | Encapsulated bacteria, viral reactivation (VZV, HBV) |
| Solid organ transplant | Chronic immunosuppressive regimens | Combined T-cell and humoral suppression | CMV, BK virus, EBV (PTLD), PJP, Aspergillus |
| Asplenia | Splenectomy, functional asplenia (sickle cell) | Loss of splenic filtration of encapsulated bacteria and IgM memory B cells | Overwhelming post-splenectomy infection (OPSI): S. pneumoniae, H. influenzae, N. meningitidis; also Babesia, Capnocytophaga |
| Malnutrition | Protein-calorie malnutrition, zinc deficiency | Impaired T-cell function, reduced secretory IgA, thymic atrophy | Measles, tuberculosis, diarrheal diseases |
| Physiologic | Extremes of age (neonatal, elderly), pregnancy | Immature immunity (neonatal); immunosenescence (elderly: decreased naïve T cells, impaired vaccine responses) | Variable |
Rituximab-Induced Hypogammaglobulinemia
Rituximab (anti-CD20) causes B-cell depletion lasting 6–12 months per course. Repeated cycles lead to cumulative hypogammaglobulinemia in 20–60% of patients, with risk increasing with each cycle. Monitor quantitative immunoglobulins before each cycle and at least every 6–12 months. IgG replacement is indicated for IgG <400 mg/dL with recurrent infections.
29 Immune Dysregulation Syndromes
These rare disorders involve immune system overactivity or dysregulation rather than simple deficiency, leading to autoimmunity, lymphoproliferation, and/or hyperinflammation.
Key Immune Dysregulation Syndromes
| Syndrome | Defect | Key Features | Treatment |
|---|---|---|---|
| ALPS (Autoimmune Lymphoproliferative Syndrome) | Defective Fas-mediated lymphocyte apoptosis (FAS, FASL, or caspase-10 mutations) | Chronic non-malignant lymphadenopathy, hepatosplenomegaly, autoimmune cytopenias (multilineage); elevated double-negative T cells (CD3+CD4−CD8− α/β TCR+ >1.5%); elevated IL-10, vitamin B12, soluble FasL. Increased lymphoma risk (50×) | Sirolimus (mTOR inhibitor; first-line for autoimmune cytopenias); mycophenolate; rituximab for refractory cytopenias |
| IPEX (Immune dysregulation, Polyendocrinopathy, Enteropathy, X-linked) | FOXP3 mutation → absent/dysfunctional Treg cells | Early-onset (neonatal) severe watery diarrhea with villous atrophy, type 1 diabetes, eczematous dermatitis, thyroiditis, cytopenias, elevated IgE; usually fatal without treatment | HSCT (curative); immunosuppression (sirolimus, tacrolimus) as bridge |
| CTLA-4 Haploinsufficiency | Heterozygous CTLA4 mutations → defective T-cell inhibitory checkpoint | Autoimmune cytopenias, lymphocytic organ infiltration (lungs, GI, brain), hypogammaglobulinemia, lymphoproliferation | Abatacept (CTLA-4-Ig fusion protein; replaces missing CTLA-4 function); sirolimus |
| STAT3 GOF (Gain-of-Function) | Activating STAT3 mutations → excessive cytokine signaling | Early-onset multi-organ autoimmunity (diabetes, thyroiditis, cytopenias, enteropathy, interstitial lung disease), lymphoproliferation, short stature | JAK inhibitors (ruxolitinib, tofacitinib); HSCT in severe cases |
| HLH (Hemophagocytic Lymphohistiocytosis) | Primary: perforin/UNC13D/STX11 mutations (familial HLH); Secondary: infection (EBV), malignancy, autoimmune triggers | Fever, cytopenias, hepatosplenomegaly, hyperferritinemia (>500, often >10,000), hypertriglyceridemia, hypofibrinogenemia, elevated sIL-2R, hemophagocytosis on marrow biopsy (HLH-2004 criteria: ≥5 of 8) | HLH-94/2004 protocol: dexamethasone + etoposide + cyclosporine; HSCT for familial HLH; treat underlying trigger in secondary HLH |
30 Skin Testing & In Vitro Diagnostics
Skin Prick Testing (SPT)
SPT is the primary diagnostic tool for IgE-mediated sensitization. A drop of allergen extract is placed on the volar forearm or back, and the skin is pricked through the drop with a lancet. A positive result is a wheal ≥3 mm greater than the negative control (saline) at 15–20 minutes. Positive control: histamine (should produce wheal ≥3 mm; if negative, consider antihistamine interference). Sensitivity: ~85–90%; specificity: ~80–85%. SPT can test dozens of allergens simultaneously and provides results in 15 minutes.

Medications That Suppress SPT (Must Be Held)
| Medication | Withdrawal Period |
|---|---|
| First-generation H1 antihistamines (diphenhydramine, hydroxyzine) | 3–5 days |
| Second-generation H1 antihistamines (cetirizine, loratadine, fexofenadine) | 3–7 days |
| H2 blockers (famotidine, ranitidine) | 1–2 days |
| Tricyclic antidepressants (amitriptyline, doxepin) | 7–14 days |
| Omalizumab | 6 months (may suppress SPT) |
| Systemic corticosteroids, ICS, INCS, LTRAs, theophylline | Not needed (do NOT suppress SPT) |
Intradermal Testing (IDT)
More sensitive but less specific than SPT. A small volume (0.02–0.05 mL) of dilute allergen is injected intradermally, creating a 2–3 mm bleb. Positive: wheal ≥3 mm increase over initial bleb at 15 minutes. Used primarily for venom allergy and drug allergy (penicillin) testing when SPT is negative but clinical suspicion remains high.
In Vitro Testing — Specific IgE (ImmunoCAP)
Serum allergen-specific IgE measurement (ImmunoCAP platform). Advantages: not affected by antihistamines, no risk of systemic reaction, useful in patients with extensive dermatitis or dermatographism. Disadvantages: lower sensitivity than SPT for some allergens, more expensive, results take days. Values ≥0.35 kU/L are considered positive (Class 1); clinical significance increases with higher levels but varies by allergen.
Component-Resolved Diagnostics (CRD)
CRD measures sIgE to individual allergenic proteins rather than whole allergen extracts, improving diagnostic precision. Key applications:
| Allergen | Components | Clinical Significance |
|---|---|---|
| Peanut | Ara h 1, 2, 3 (storage proteins); Ara h 8 (PR-10); Ara h 9 (LTP) | Ara h 2 positive → high risk of systemic reaction; Ara h 8 only → OAS, low systemic risk |
| Milk | Bos d 4 (α-lactalbumin); Bos d 5 (β-lactoglobulin); Bos d 8 (casein) | Casein (Bos d 8) positive → likely reacts to all forms of milk; whey-only → may tolerate baked milk |
| Egg | Gal d 1 (ovomucoid); Gal d 2 (ovalbumin) | Ovomucoid (heat-stable) positive → reacts to baked egg; ovalbumin only → may tolerate baked egg |
| Bee/wasp venom | Api m 1 (phospholipase A2); Ves v 5 (antigen 5) | Distinguishes true bee vs wasp allergy from CCD (cross-reactive carbohydrate determinant) cross-reactivity |
31 Pulmonary Function & Bronchial Provocation
Spirometry
Spirometry is essential for asthma diagnosis, severity assessment, and monitoring. Key measurements: FEV1 (forced expiratory volume in 1 second), FVC (forced vital capacity), and FEV1/FVC ratio. Asthma shows an obstructive pattern: reduced FEV1/FVC ratio (<0.70 or below the lower limit of normal). Bronchodilator reversibility confirms asthma: ≥12% AND ≥200 mL improvement in FEV1 after SABA (albuterol 400 μg). Normal spirometry does not exclude asthma (variable airflow obstruction).
Fractional Exhaled Nitric Oxide (FeNO)
FeNO measures airway eosinophilic inflammation. Normal <25 ppb in adults, <20 ppb in children. Elevated FeNO (≥25 ppb adults, ≥35 ppb high) supports eosinophilic/allergic asthma diagnosis and predicts response to ICS and anti-IL-4/IL-13 biologics. FeNO >50 ppb strongly suggests eosinophilic airway inflammation and likely ICS responsiveness. False elevations: viral URI, allergen exposure, dietary nitrate. False low: smoking, bronchoconstriction at time of testing.
Bronchial Provocation Testing
Methacholine challenge is the most common bronchoprovocation test. Inhaled methacholine (a direct muscarinic agonist) is administered in increasing concentrations. A positive test is a ≥20% decline in FEV1 (PC20 or PD20). Interpretation: PC20 <4 mg/mL = positive (significant AHR); 4–16 mg/mL = borderline; >16 mg/mL = negative. Methacholine challenge has high sensitivity (~95%) but moderate specificity (~80%) for asthma — best used to EXCLUDE asthma (high negative predictive value). Contraindications: severe obstruction (FEV1 <60% predicted), recent MI/stroke, uncontrolled hypertension, aortic aneurysm.
Other Provocation Tests
Exercise challenge: Standardized exercise at 85–90% max HR for 6–8 min with serial FEV1 measurements for 30 min post-exercise; positive = ≥10% FEV1 decline. Eucapnic voluntary hyperventilation (EVH): Most sensitive for exercise-induced bronchoconstriction; used in Olympic athlete screening. Mannitol challenge: Indirect challenge via osmotic stimulus; FDA-approved (Aridol); positive = ≥15% FEV1 decline; higher specificity than methacholine.
32 Allergen Immunotherapy (SCIT & SLIT)
Allergen immunotherapy (AIT) is the ONLY disease-modifying treatment for allergic rhinitis, allergic asthma, and stinging insect allergy. It induces immune tolerance by shifting from Th2 to Th1/Treg responses, increasing blocking IgG4 antibodies, and reducing allergen-specific IgE over time.
Subcutaneous Immunotherapy (SCIT)
| Phase | Duration | Schedule | Details |
|---|---|---|---|
| Build-up | 3–6 months (conventional) or 1 day (rush/cluster) | Weekly or biweekly injections with gradually increasing doses | Start at 1:10,000 or 1:1,000 v/v dilution; escalate to maintenance concentration |
| Maintenance | 3–5 years | Monthly injections (every 4 weeks) | Maintenance dose typically 1,000–4,000 BAU or 3–15 μg major allergen per injection; continue for 3–5 years for sustained benefit |
Safety: Observe 30 minutes after each injection. Risk of systemic reaction: ~0.1–0.2% per injection (local reactions are common and expected). Fatal reactions: ~1 per 2.5 million injections. Contraindications: uncontrolled/severe asthma (FEV1 <70%), beta-blocker use (relative — increases risk of refractory anaphylaxis), medical conditions where epinephrine is contraindicated.
Sublingual Immunotherapy (SLIT)
FDA-approved SLIT tablets in the US:
| Product | Allergen | Dose | Start | Duration |
|---|---|---|---|---|
| Grastek | Timothy grass pollen | 2800 BAU daily SL | 12 weeks before grass season (or year-round) | 3 years (pre-seasonal or perennial) |
| Ragwitek | Short ragweed pollen | 12 Amb a 1-U daily SL | 12 weeks before ragweed season | 3 years (pre-seasonal) |
| Odactra | House dust mite | 12 SQ-HDM daily SL | Year-round (perennial allergen) | 3+ years continuous |
| Palforzia | Peanut | 300 mg daily (maintenance) | After supervised updosing | Ongoing |
First dose of any SLIT tablet must be administered under medical supervision (30 min observation). Subsequent doses are self-administered at home. Most common side effect: local oropharyngeal pruritus/swelling (resolves within 1–2 weeks). Anaphylaxis rate: ~1 per 100 million SLIT doses (much lower than SCIT).
33 Venom Immunotherapy
Venom immunotherapy (VIT) is indicated for patients with a history of systemic allergic reaction to Hymenoptera stings (bees, wasps, hornets, yellow jackets, fire ants) AND positive venom-specific IgE (SPT or sIgE). VIT is the most effective form of immunotherapy: reduces risk of systemic reaction from ~60% to <5% on re-sting.
Indications & Contraindications
Indicated: Systemic reaction (urticaria/angioedema remote from sting site, respiratory symptoms, cardiovascular symptoms, anaphylaxis) + positive venom testing. NOT indicated: Large local reactions only (normal physiologic response), cutaneous-only systemic reactions in children <16 years (low risk of progression to anaphylaxis, ~1–3%), or negative venom testing. Elevated baseline tryptase (>11.5 ng/mL) or known mastocytosis is an indication for lifelong VIT (higher risk of fatal sting anaphylaxis).
Protocol
Build-up: weekly injections over 8–12 weeks (conventional) or 1–2 days (rush protocol; higher systemic reaction rate ~10–15% but acceptable in supervised settings). Maintenance dose: 100 μg of each relevant venom per injection (300 μg for mixed vespid). Maintenance interval: every 4 weeks for year 1, then extend to every 6–8 weeks for years 2–5. Duration: minimum 5 years; consider lifelong in patients with prior near-fatal anaphylaxis, elevated baseline tryptase, or mastocytosis.
34 Drug Challenge & Desensitization Protocols
Drug Provocation Testing (Drug Challenge)
A graded drug challenge is used to exclude drug allergy in patients with a low-risk history or negative skin testing. The suspected drug is administered in incrementally increasing doses (typically 2–3 doses over 1–2 hours) with observation. It is a diagnostic test (different from desensitization, which is therapeutic). Common scenarios: amoxicillin challenge after negative penicillin skin test, NSAID challenge in patients with unclear NSAID reaction history, local anesthetic challenge (true allergy is extremely rare — <1%).
Aspirin Desensitization for AERD
Patients with AERD (Samter's triad) who require aspirin for cardiovascular indications or who would benefit from aspirin's anti-inflammatory effect on nasal polyps undergo aspirin desensitization in a monitored setting. Protocol: begin with aspirin 20–40 mg, escalate every 90–180 minutes through 40, 81, 162, 325, 650 mg. Watch for bronchospasm, naso-ocular symptoms, and GI reactions. Once 325–650 mg is tolerated, maintain BID dosing indefinitely (interruption >48 hours requires re-desensitization). Benefits: reduced nasal polyp recurrence, improved smell, decreased oral steroid requirements, better asthma control.
Chemotherapy Desensitization
Indicated for patients with confirmed type I hypersensitivity to carboplatin, oxaliplatin, paclitaxel, or monoclonal antibodies who have no alternative agent. Standard 12-step protocol (Castells protocol): three sequential solutions of increasing concentration (1/100, 1/10, full strength), each administered at escalating rates (2×, 5×, 10×, 25×, etc.), achieving full therapeutic dose over ~5.8 hours. Success rate >95%. Requires ICU or step-down unit setting. Must be repeated for each subsequent cycle.
35 Imaging & Diagnostics Table
| Test / Modality | Indication | Key Findings |
|---|---|---|
| Chest X-ray | Asthma (exclude other diagnoses), PID evaluation | Hyperinflation in acute asthma; absent thymic shadow in SCID/DiGeorge; bronchiectasis in PID |
| CT Sinuses (non-contrast) | Chronic rhinosinusitis evaluation | Mucosal thickening, opacification, ostiomeatal complex obstruction, polyps, double-density sign (AFS) |
| High-resolution CT (HRCT) Chest | Bronchiectasis screening in PID, hypersensitivity pneumonitis | Tram lines, signet ring sign (bronchiectasis); ground-glass opacities (HP); tree-in-bud (infection) |
| Spirometry (PFT) | Asthma diagnosis and monitoring | Obstructive pattern (low FEV1/FVC); bronchodilator reversibility ≥12% + 200 mL |
| FeNO | Eosinophilic airway inflammation, ICS adherence | ≥25 ppb suggests eosinophilic inflammation; ≥50 ppb strongly predictive |
| Methacholine challenge | Diagnose asthma with normal spirometry | PC20 <4 mg/mL = positive for AHR |
| Skin prick testing | IgE-mediated allergy (aeroallergens, foods, venoms) | Wheal ≥3 mm > negative control at 15 min |
| Patch testing | Allergic contact dermatitis | Positive reaction (erythema, papules, vesicles) at 48–96 hours |
| Serum tryptase | Anaphylaxis confirmation, mastocytosis screening | Elevated >11.5 ng/mL; acute: draw 15 min–3 h after symptom onset |
| Specific IgE (ImmunoCAP) | When SPT not possible or to supplement SPT | ≥0.35 kU/L positive; quantitative values aid risk stratification |
| CRD panels | Risk stratification for food allergy, venom cross-reactivity | Component-specific results improve diagnostic precision (see Section 30) |
| DHR flow cytometry | Chronic granulomatous disease | Absent oxidative burst in neutrophils |
| Flow cytometry (lymphocyte subsets) | PID evaluation (SCID, CVID, DiGeorge) | CD3/CD4/CD8/CD19/CD16+56 quantification; absent T/B/NK populations in specific PIDs |
| TREC assay (newborn screening) | SCID newborn screening | Absent or very low TRECs = T-cell lymphopenia → urgent confirmatory testing |
| Vaccine response titers | Functional antibody assessment | Pre/post Pneumovax titers: ≥1.3 μg/mL to ≥70% of serotypes = adequate response |
36 Classification Systems (All)
ARIA Classification of Allergic Rhinitis
| Dimension | Category | Definition |
|---|---|---|
| Duration | Intermittent | <4 days/week OR <4 consecutive weeks |
| Persistent | ≥4 days/week AND ≥4 consecutive weeks | |
| Severity | Mild | Normal sleep, no impairment of daily activities/work/school, no troublesome symptoms |
| Moderate-Severe | ≥1 of: sleep disturbance, impairment of activities, troublesome symptoms |
GINA Asthma Severity (Treatment-Naïve)
| Severity | Symptoms | Night Symptoms | FEV1 |
|---|---|---|---|
| Intermittent | ≤2 days/week | ≤2×/month | >80% predicted |
| Mild persistent | >2 days/week, not daily | 3–4×/month | ≥80% |
| Moderate persistent | Daily | >1×/week | 60–80% |
| Severe persistent | Throughout the day | Nightly | <60% |
GINA Asthma Control Assessment
| Level | Criteria (Past 4 Weeks) |
|---|---|
| Well-controlled | None of: daytime symptoms >2×/wk, night waking, SABA >2×/wk, activity limitation |
| Partly controlled | 1–2 of the above |
| Uncontrolled | 3–4 of the above |
Sampson Severity Grading of Anaphylaxis
| Grade | Skin | GI | Respiratory | Cardiovascular | Neurologic |
|---|---|---|---|---|---|
| 1 | Localized urticaria/pruritus, flushing, tingling | — | — | — | — |
| 2 | Generalized urticaria/pruritus | Nausea, cramping | Nasal congestion, sneeze | — | — |
| 3 | Generalized urticaria/pruritus | Vomiting, diarrhea | Rhinorrhea, mild wheeze, throat tightness | Tachycardia (ΔHR ≥15) | Anxiety |
| 4 | Generalized urticaria/pruritus | Vomiting, diarrhea | Hoarseness, stridor, dyspnea, wheeze, cyanosis | Hypotension | Lightheadedness, confusion |
| 5 | Generalized urticaria/pruritus | Loss of bowel control | Respiratory arrest | Severe bradycardia/arrest | Loss of consciousness |
Ring & Messmer Anaphylaxis Grading
| Grade | Clinical Features |
|---|---|
| I | Cutaneous signs only: erythema, urticaria, angioedema |
| II | Moderate multisystem: cutaneous + GI (nausea, vomiting) + mild respiratory (cough, dyspnea) + mild hemodynamic (tachycardia, hypotension responding to fluids) |
| III | Life-threatening: severe bronchospasm, laryngeal edema, cardiovascular collapse (systolic BP <70 mmHg), tachycardia or bradycardia |
| IV | Cardiac and/or respiratory arrest |
EAACI Food Allergy Severity Grading
| Grade | Severity | Description |
|---|---|---|
| 1 | Mild (localized) | Oral pruritus, lip swelling, few hives, mild abdominal discomfort |
| 2 | Mild (generalized) | Generalized urticaria, abdominal pain, nausea/vomiting (1–2 episodes) |
| 3 | Moderate | Above + any of: hoarseness, barky cough, difficulty swallowing, wheeze, persistent vomiting, uterine cramping |
| 4 | Severe (anaphylaxis) | Above + any of: laryngeal edema, severe bronchospasm, hypotension (SBP <90 or >30% drop), LOC, incontinence |
| 5 | Fatal | Death |
HAE Severity Classification
| Severity | Attack Frequency | Attack Characteristics | Impact |
|---|---|---|---|
| Mild | <6 attacks/year | Primarily peripheral (extremities), short duration, no airway involvement | Minimal impact on daily life |
| Moderate | 6–12 attacks/year | Some facial/abdominal attacks, longer duration | Moderate impairment; missed work/school days |
| Severe | >12 attacks/year or any laryngeal attack | Frequent abdominal/facial/laryngeal attacks, prolonged duration (>3 days) | Significant disability; requires long-term prophylaxis |
EoE Endoscopic Reference Score (EREFS)
| Feature | Grading |
|---|---|
| Edema (loss of vascular markings) | 0 = absent, 1 = mild, 2 = severe |
| Rings (concentric, "trachealization") | 0 = none, 1 = mild, 2 = moderate, 3 = severe |
| Exudates (white plaques) | 0 = none, 1 = mild, 2 = severe |
| Furrows (linear) | 0 = absent, 1 = present |
| Stricture | 0 = absent, 1 = present |
37 Medications Master Table
Antihistamines
| Drug | Generation | Dose (Adult) | Key Features |
|---|---|---|---|
| Cetirizine (Zyrtec) | 2nd | 10 mg PO daily | Fastest onset of 2nd-gen (~1 hr); mild sedation in ~10% |
| Loratadine (Claritin) | 2nd | 10 mg PO daily | Least sedating; available OTC; fewer drug interactions |
| Fexofenadine (Allegra) | 2nd | 180 mg PO daily | Truly non-sedating; avoid with fruit juice (reduces absorption) |
| Levocetirizine (Xyzal) | 2nd | 5 mg PO daily | Active enantiomer of cetirizine; may be slightly more potent |
| Diphenhydramine (Benadryl) | 1st | 25–50 mg PO/IV q4–6h | Significant sedation and anticholinergic effects; used for acute allergic reactions |
| Hydroxyzine (Atarax/Vistaril) | 1st | 25–50 mg PO q6h | Also anxiolytic; more sedating; useful for nocturnal pruritus |
| Azelastine nasal (Astelin) | 2nd (topical) | 1–2 sprays/nostril BID | Intranasal antihistamine; onset 15 min; bitter taste; effective for NAR and AR |
Intranasal Corticosteroids
| Drug | Dose | Notes |
|---|---|---|
| Fluticasone propionate (Flonase) | 1–2 sprays/nostril daily | OTC; most widely used; low systemic bioavailability (<1%) |
| Mometasone (Nasonex) | 2 sprays/nostril daily | Minimal systemic absorption; FDA-approved for ages ≥2 |
| Budesonide (Rhinocort) | 1–2 sprays/nostril daily | OTC; preferred in pregnancy (Category B equivalent) |
| Fluticasone furoate (Veramyst) | 1–2 sprays/nostril daily | Once-daily dosing; enhanced receptor affinity |
| Fluticasone/azelastine (Dymista) | 1 spray/nostril BID | Combination INCS + antihistamine; superior to either alone for moderate-severe AR |
Inhaled Corticosteroids (ICS) for Asthma
| Drug | Low Dose (μg/day) | Medium Dose | High Dose |
|---|---|---|---|
| Budesonide (Pulmicort) | 200–400 | 400–800 | >800 |
| Fluticasone propionate (Flovent) | 100–250 | 250–500 | >500 |
| Beclomethasone (QVAR) | 200–500 | 500–1000 | >1000 |
| Mometasone (Asmanex) | 200 | 400 | >400 |
| Ciclesonide (Alvesco) | 80–160 | 160–320 | >320 |
Leukotriene Modifiers
| Drug | Mechanism | Dose | Indications / Notes |
|---|---|---|---|
| Montelukast (Singulair) | Cysteinyl leukotriene receptor-1 antagonist | 10 mg PO daily (adults); 4–5 mg (children) | AR, asthma, EIB; FDA BLACK BOX (2020): neuropsychiatric events (agitation, depression, suicidal ideation) |
| Zafirlukast (Accolate) | Cysteinyl leukotriene receptor-1 antagonist | 20 mg PO BID | Take on empty stomach; hepatotoxicity risk (monitor LFTs); Churg-Strauss association (rare) |
| Zileuton (Zyflo) | 5-lipoxygenase inhibitor | 600 mg PO QID (or CR 1200 mg BID) | Only agent blocking LT synthesis; useful in AERD; hepatotoxicity (monitor LFTs monthly × 3 months) |
Epinephrine
| Formulation | Dose | Route | Notes |
|---|---|---|---|
| Epinephrine auto-injector (EpiPen) | 0.3 mg | IM (anterolateral thigh) | Adults and children >30 kg; carry 2 at all times |
| EpiPen Jr | 0.15 mg | IM | Children 15–30 kg |
| Epinephrine 1:1000 (1 mg/mL) | 0.01 mg/kg (max 0.5 mg) | IM | Drawn from vial; use for weight-based dosing in children <15 kg |
| Epinephrine IV infusion | 0.1–1 μg/kg/min | IV | ONLY for refractory anaphylaxis in monitored setting; NEVER give 1:1000 IM formulation IV push |
Biologics Summary
| Drug | Target | Approved Indications | Dose |
|---|---|---|---|
| Omalizumab (Xolair) | IgE | Allergic asthma, CSU, CRSwNP | 75–375 mg SC q2–4w (weight/IgE-based) |
| Mepolizumab (Nucala) | IL-5 | Severe eosinophilic asthma, EGPA, HES, CRSwNP | 100 mg SC q4w |
| Benralizumab (Fasenra) | IL-5Rα | Severe eosinophilic asthma | 30 mg SC q4w × 3, then q8w |
| Dupilumab (Dupixent) | IL-4Rα | Moderate-severe AD, asthma, CRSwNP, EoE, prurigo nodularis | 300 mg SC q2w (loading varies) |
| Tezepelumab (Tezspire) | TSLP | Severe asthma (all phenotypes) | 210 mg SC q4w |
| Lanadelumab (Takhzyro) | Kallikrein | HAE prophylaxis | 300 mg SC q2w (may extend to q4w) |
| Tralokinumab (Adbry) | IL-13 | Moderate-severe AD | 300 mg SC q2w (after loading) |
Immunosuppressants Used in Allergy/Immunology
| Drug | Mechanism | Dose Range | Key Monitoring |
|---|---|---|---|
| Cyclosporine | Calcineurin inhibitor (blocks IL-2 transcription) | 3–5 mg/kg/day PO | Renal function, BP, drug level (trough 100–200 ng/mL), lipids, K+, Mg2+ |
| Mycophenolate mofetil | IMPDH inhibitor (blocks purine synthesis in lymphocytes) | 1–3 g/day PO | CBC (leukopenia), GI toxicity, pregnancy category X |
| Azathioprine | Purine synthesis inhibitor | 1–3 mg/kg/day PO | TPMT genotype before starting; CBC, LFTs; pancreatitis risk |
| Methotrexate | Dihydrofolate reductase inhibitor | 7.5–25 mg/week PO/SC | CBC, LFTs, renal function, pulmonary toxicity; supplement folic acid 1 mg/day |
38 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ABPA | Allergic bronchopulmonary aspergillosis |
| ACD | Allergic contact dermatitis |
| ACO | Asthma-COPD overlap |
| AD | Atopic dermatitis |
| ADCC | Antibody-dependent cell-mediated cytotoxicity |
| AERD | Aspirin-exacerbated respiratory disease |
| AFS | Allergic fungal sinusitis |
| AHR | Airway hyperresponsiveness |
| AIT | Allergen immunotherapy |
| ALPS | Autoimmune lymphoproliferative syndrome |
| APC | Antigen-presenting cell |
| AR | Allergic rhinitis |
| ARIA | Allergic Rhinitis and Its Impact on Asthma |
| BAU | Bioequivalent allergy unit |
| BTK | Bruton tyrosine kinase |
| CCD | Cross-reactive carbohydrate determinant |
| CGD | Chronic granulomatous disease |
| CRD | Component-resolved diagnostics |
| CRS | Chronic rhinosinusitis |
| CRSsNP | Chronic rhinosinusitis without nasal polyps |
| CRSwNP | Chronic rhinosinusitis with nasal polyps |
| CSU | Chronic spontaneous urticaria |
| CTL | Cytotoxic T lymphocyte |
| CVID | Common variable immunodeficiency |
| DC | Dendritic cell |
| DHR | Dihydrorhodamine |
| DRESS | Drug reaction with eosinophilia and systemic symptoms |
| EAACI | European Academy of Allergy and Clinical Immunology |
| EIB | Exercise-induced bronchoconstriction |
| EoE | Eosinophilic esophagitis |
| EREFS | EoE endoscopic reference score |
| EVH | Eucapnic voluntary hyperventilation |
| FDEIA | Food-dependent exercise-induced anaphylaxis |
| FeNO | Fractional exhaled nitric oxide |
| FEV1 | Forced expiratory volume in 1 second |
| FESS | Functional endoscopic sinus surgery |
| FPIAP | Food protein-induced allergic proctocolitis |
| FPIES | Food protein-induced enterocolitis syndrome |
| FVC | Forced vital capacity |
| GINA | Global Initiative for Asthma |
| HAE | Hereditary angioedema |
| HLH | Hemophagocytic lymphohistiocytosis |
| HSCT | Hematopoietic stem cell transplant |
| ICS | Inhaled corticosteroid |
| IDT | Intradermal test |
| INCS | Intranasal corticosteroid |
| IPEX | Immune dysregulation, polyendocrinopathy, enteropathy, X-linked |
| IVIG | Intravenous immunoglobulin |
| LABA | Long-acting beta-agonist |
| LAD | Leukocyte adhesion deficiency |
| LAMA | Long-acting muscarinic antagonist |
| LTP | Lipid transfer protein |
| LTRA | Leukotriene receptor antagonist |
| MAC | Membrane attack complex |
| MBL | Mannose-binding lectin |
| NAEPP | National Asthma Education and Prevention Program |
| NAR | Non-allergic rhinitis |
| NARES | Non-allergic rhinitis with eosinophilia syndrome |
| NBT | Nitroblue tetrazolium |
| NIAID | National Institute of Allergy and Infectious Diseases |
| NK | Natural killer (cell) |
| OAS | Oral allergy syndrome |
| OFC | Oral food challenge |
| OIT | Oral immunotherapy |
| PEF | Peak expiratory flow |
| PFT | Pulmonary function test |
| PID | Primary immunodeficiency disease |
| RADS | Reactive airways dysfunction syndrome |
| SABA | Short-acting beta-agonist |
| SCAR | Severe cutaneous adverse reaction |
| SCID | Severe combined immunodeficiency |
| SCIG | Subcutaneous immunoglobulin |
| SCIT | Subcutaneous immunotherapy |
| sIgE | Specific immunoglobulin E |
| SJS | Stevens-Johnson syndrome |
| SLIT | Sublingual immunotherapy |
| SPT | Skin prick test |
| SSLR | Serum sickness-like reaction |
| TCS | Topical corticosteroid |
| TEN | Toxic epidermal necrolysis |
| TREC | T-cell receptor excision circle |
| TSLP | Thymic stromal lymphopoietin |
| VIT | Venom immunotherapy |
| WAS | Wiskott-Aldrich syndrome |
| XLA | X-linked agammaglobulinemia |
39 Risk Factors & Comorbidities
Risk Factors for Atopic Disease
| Factor | Association |
|---|---|
| Family history of atopy | One atopic parent: 40% risk in child; both atopic parents: 60–80% risk; strongest risk factor |
| Filaggrin (FLG) mutations | Loss-of-function mutations in ~10% of population; 3× risk of AD, increased risk of asthma and peanut allergy |
| Elevated cord blood IgE | Predicts atopic disease development (low sensitivity, high specificity) |
| Cesarean delivery | Modest increase in asthma and atopic disease (altered microbiome colonization) |
| Antibiotic use in infancy | Associated with increased asthma risk (microbiome disruption) |
| Tobacco smoke exposure | Prenatal and postnatal exposure increases asthma risk 1.5–2× |
| Air pollution (PM2.5, NO2, ozone) | Increases asthma incidence and exacerbations; emerging evidence for allergic sensitization |
| Obesity | Increases asthma risk 1.5× (especially non-eosinophilic phenotype); worsens asthma severity |
| Reduced microbial diversity (hygiene hypothesis) | Reduced early microbial exposure associated with increased allergic disease; farm exposure is protective |
The Atopic March
The atopic march describes the sequential development of atopic diseases: atopic dermatitis (infancy, often first manifestation) → food allergy (infancy/early childhood) → allergic rhinitis (school age) → asthma (school age/adolescence). Not all atopic children follow this progression, but early severe AD is a strong predictor of subsequent respiratory allergy. Epicutaneous sensitization through a defective skin barrier (FLG mutations) may drive subsequent food and aeroallergen sensitization.
Comorbidity Associations
| Condition | Key Comorbidities |
|---|---|
| Allergic rhinitis | Asthma (80% of asthmatics have AR), sinusitis, otitis media with effusion, sleep-disordered breathing, nasal polyps |
| Asthma | AR (90% of allergic asthma), GERD, OSA, obesity, anxiety/depression, vocal cord dysfunction, CRSwNP |
| Atopic dermatitis | Food allergy (30–40% of children with moderate-severe AD), AR, asthma, allergic conjunctivitis, S. aureus skin infections, eczema herpeticum, depression/anxiety, ADHD |
| CVID | Autoimmune cytopenias (ITP, AIHA), granulomatous disease, bronchiectasis, lymphoid interstitial pneumonia, lymphoma, GI disease (nodular lymphoid hyperplasia, villous atrophy) |
| Chronic urticaria | Autoimmune thyroid disease, anxiety/depression, angioedema |
| AERD | CRSwNP (nearly universal), severe asthma, NSAID intolerance |