Cardiology
Every diagnosis, condition, procedure, device, medication, classification system, EKG finding, hemodynamic parameter, and management strategy in one place.
01 Cardiac Anatomy & Physiology
The heart is a four-chambered muscular pump weighing approximately 250–350 g in adults, located in the mediastinum behind the sternum and between the lungs. It receives deoxygenated blood from the systemic venous circulation, sends it through the pulmonary circuit for gas exchange, and then pumps oxygenated blood to the entire body. Understanding normal cardiac anatomy, the coronary arterial supply, the conduction system, and the cardiac cycle is prerequisite to every disease and procedure in cardiology.
Chambers & Great Vessels
The right atrium (RA) receives venous return via the superior vena cava (SVC), inferior vena cava (IVC), and coronary sinus. Blood passes through the tricuspid valve into the right ventricle (RV), which ejects through the pulmonic valve into the pulmonary arteries. Oxygenated blood returns via four pulmonary veins to the left atrium (LA), passes through the mitral valve (bicuspid, with anterior and posterior leaflets) into the left ventricle (LV) — the dominant pump — which ejects through the aortic valve (tricuspid semilunar) into the ascending aorta. The LV wall is 3× thicker than the RV (~12 mm vs ~4 mm) because it generates systemic pressures (120/80 mmHg) versus pulmonary pressures (25/10 mmHg).
Cardiac Valves
The four valves ensure unidirectional flow. The atrioventricular (AV) valves — tricuspid (right) and mitral (left) — have leaflets tethered by chordae tendineae to papillary muscles in the ventricular wall; papillary muscle dysfunction (e.g., from ischemia) can cause acute valvular regurgitation. The semilunar valves — pulmonic and aortic — have three cusps each and no chordae. The aortic valve cusps are named left coronary, right coronary, and non-coronary (the coronary arteries originate from the sinuses of Valsalva above the left and right cusps). Valve area and gradients are the core measurements for assessing stenosis severity.
Coronary Artery Anatomy
The left main coronary artery (LMCA) arises from the left sinus of Valsalva and bifurcates (within ~1 cm) into the left anterior descending (LAD) and the left circumflex (LCx). The LAD travels in the anterior interventricular groove and supplies the anterior wall, anterior septum, and apex of the LV via septal perforators and diagonal branches. The LCx travels in the left AV groove and supplies the lateral and posterolateral LV wall via obtuse marginal (OM) branches. The right coronary artery (RCA) arises from the right sinus of Valsalva, travels in the right AV groove, and supplies the RA, RV, inferior LV wall, and posterior septum. The posterior descending artery (PDA) determines dominance: in ~85% of people, the PDA arises from the RCA (right-dominant); in ~8%, from the LCx (left-dominant); in ~7%, both contribute (co-dominant). The RCA also typically supplies the SA node (60%) and AV node (90%) — so inferior MI can cause sinus bradycardia or heart block.
The Conduction System
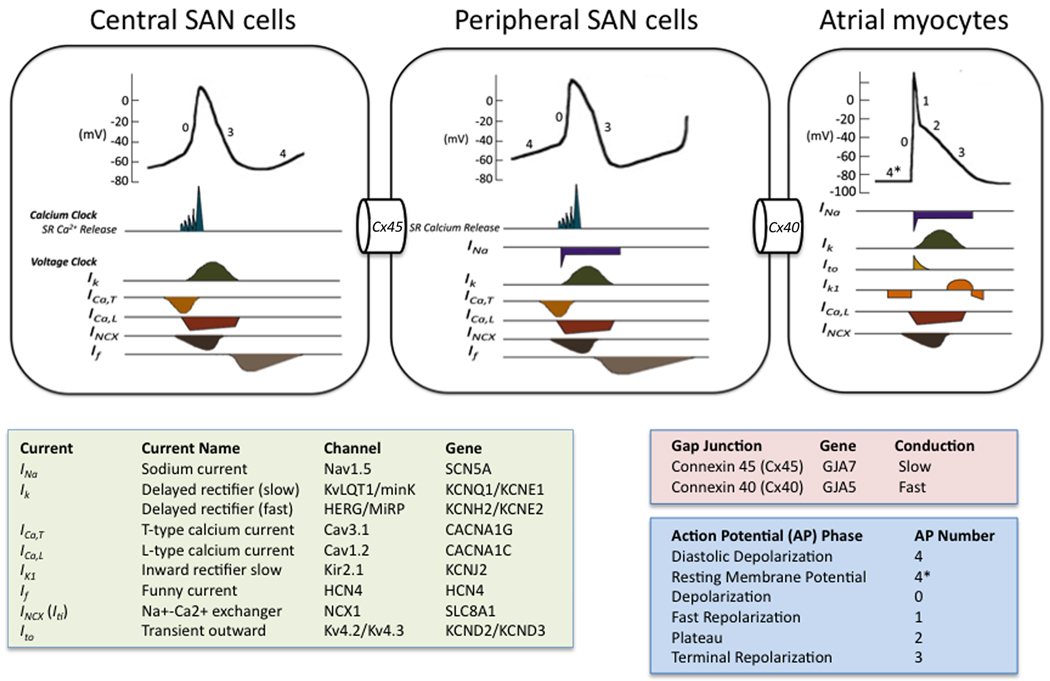
The sinoatrial (SA) node is the heart's primary pacemaker, located at the junction of the SVC and the RA, with an intrinsic rate of 60–100 bpm. The impulse spreads through atrial myocardium (and internodal pathways) to the atrioventricular (AV) node, located at the base of the interatrial septum near the coronary sinus ostium, where it is delayed ~120 ms (this delay allows atrial contraction to fill the ventricles before ventricular systole — the "atrial kick," which contributes ~15–25% of cardiac output). The impulse then travels through the bundle of His in the membranous interventricular septum, splits into the right bundle branch (RBB) and left bundle branch (LBB) (which further divides into the left anterior fascicle and left posterior fascicle), and finally through the Purkinje fibers to rapidly activate the ventricular myocardium from apex to base.
The Cardiac Cycle & Hemodynamics
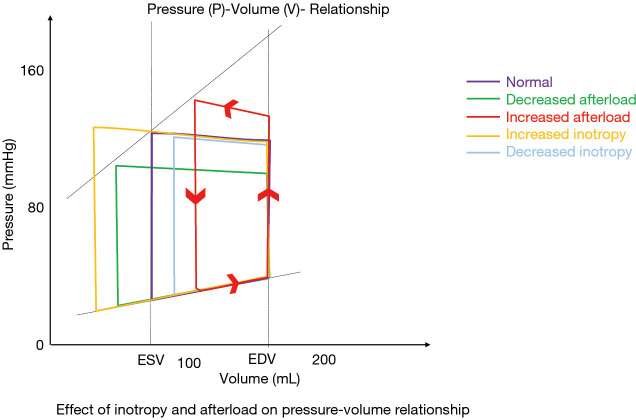
The cardiac cycle has two phases: systole (ventricular contraction and ejection, ~300 ms) and diastole (ventricular relaxation and filling, ~500 ms at rest). During isovolumetric contraction, ventricular pressure rises with all valves closed until it exceeds aortic/pulmonary pressure, opening the semilunar valves (ejection phase). During isovolumetric relaxation, ventricular pressure drops with all valves closed until it falls below atrial pressure, opening the AV valves (filling phase). Normal resting cardiac output (CO) = stroke volume (SV) × heart rate (HR) ≈ 70 mL × 70 bpm = ~5 L/min. Ejection fraction (EF) = SV / end-diastolic volume (EDV) × 100; normal LV EF = 55–70%.
02 The Cardiac Physical Exam
Inspection & Palpation
Assess for jugular venous distension (JVD) — the internal jugular vein is used; with the patient at 45°, the top of the venous pulsation measured vertically above the sternal angle + 5 cm (the estimated distance from the sternal angle to the RA) gives the estimated CVP. Normal JVP is ≤8 cm H₂O. Elevated JVP indicates right heart failure, volume overload, cardiac tamponade, or constrictive pericarditis. The hepatojugular reflux (apply sustained pressure over the liver for 10 seconds — a sustained rise in JVP ≥3 cm is positive) suggests elevated LVEDP or right heart failure. Palpate for the point of maximal impulse (PMI) at the 5th intercostal space, midclavicular line — a displaced, laterally shifted, or sustained PMI suggests LV dilation or hypertrophy.
Auscultation — Heart Sounds
S1 = mitral and tricuspid valve closure (beginning of systole). S2 = aortic and pulmonic valve closure (end of systole); physiologic splitting of S2 widens during inspiration (increased venous return delays pulmonic closure). Fixed splitting = ASD. Paradoxical splitting (splitting on expiration, single on inspiration) = LBBB or severe AS (delayed aortic closure). S3 (ventricular gallop) = rapid early diastolic filling into a volume-overloaded ventricle — pathologic in adults >40 years, associated with HF (sensitivity ~50% but specificity >90% for elevated LVEDP). S4 (atrial gallop) = atrial contraction against a stiff ventricle — LVH, diastolic dysfunction, acute ischemia. Absent in AFib (no organized atrial contraction).
Murmur Grading (Levine Scale)
| Grade | Intensity | Description |
|---|---|---|
| I/VI | Very faint | Heard only with careful auscultation in a quiet room |
| II/VI | Soft | Readily audible but quiet |
| III/VI | Moderate | Easily heard, no thrill |
| IV/VI | Loud | Loud with palpable thrill |
| V/VI | Very loud | Heard with stethoscope barely touching chest; thrill present |
| VI/VI | Audible without stethoscope | Heard with stethoscope lifted off chest; thrill present |
Classic Murmur Characteristics
| Valve Lesion | Timing | Best Heard | Character | Maneuvers |
|---|---|---|---|---|
| Aortic stenosis | Systolic (crescendo-decrescendo) | RUSB, radiates to carotids | Harsh, "diamond-shaped" | Decreases with Valsalva, increases with squatting |
| Mitral regurgitation | Holosystolic (pansystolic) | Apex, radiates to axilla | Blowing | Increases with squatting, hand grip |
| Aortic regurgitation | Early diastolic (decrescendo) | LUSB/LLSB, patient sitting up leaning forward | Blowing, high-pitched | Increases with squatting, hand grip |
| Mitral stenosis | Mid-diastolic (low-frequency rumble) | Apex, left lateral decubitus | Rumbling, opening snap | Increases with exercise |
| Tricuspid regurgitation | Holosystolic | LLSB | Blowing | Increases with inspiration (Carvallo's sign) |
| MVP (mitral valve prolapse) | Mid-systolic click ± late systolic murmur | Apex | Click earlier with Valsalva/standing | Click later with squatting |
| HCM (LVOT obstruction) | Systolic (crescendo-decrescendo) | LLSB | Harsh | Increases with Valsalva/standing; decreases with squatting |
Peripheral Edema Grading
| Grade | Depth | Rebound | Clinical |
|---|---|---|---|
| 1+ | 2 mm | Rapid (immediate) | Barely perceptible, ankle/foot |
| 2+ | 4 mm | 15 seconds | Visible depression, lower leg |
| 3+ | 6 mm | 30 seconds | Obvious swelling, above knee |
| 4+ | 8+ mm | > 30 seconds | Severe, anasarca, tight/weeping skin |
03 Key Terminology & Abbreviations
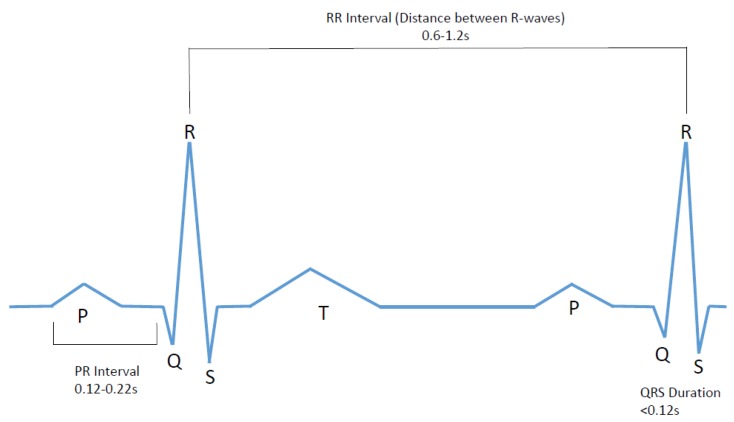
Cardiology has one of the densest abbreviation vocabularies in medicine. Mastering these terms is essential for reading EKG reports, catheterization data, echocardiograms, and clinical notes.
| Abbreviation | Meaning |
|---|---|
| EF | Ejection fraction |
| CO / CI | Cardiac output / cardiac index (CO adjusted for BSA) |
| SVR / PVR | Systemic / pulmonary vascular resistance |
| PCWP (or PAWP) | Pulmonary capillary wedge pressure (surrogate for LA pressure / LVEDP) |
| mPAP | Mean pulmonary artery pressure |
| LVEDP / LVEDV | Left ventricular end-diastolic pressure / volume |
| STEMI / NSTEMI | ST-elevation MI / non-ST-elevation MI |
| ACS | Acute coronary syndrome |
| PCI | Percutaneous coronary intervention |
| CABG | Coronary artery bypass grafting ("cabbage") |
| DAPT | Dual antiplatelet therapy (typically ASA + P2Y12 inhibitor) |
| BNP / NT-proBNP | B-type natriuretic peptide / N-terminal proBNP |
| GDMT | Guideline-directed medical therapy |
| HFrEF / HFpEF / HFmrEF | Heart failure with reduced / preserved / mildly reduced EF |
| TAVR (or TAVI) | Transcatheter aortic valve replacement (or implantation) |
| ICD / CRT / CRT-D | Implantable cardioverter-defibrillator / cardiac resynchronization therapy / CRT with defib |
| EP | Electrophysiology |
| AF / AFib / AFL | Atrial fibrillation / atrial flutter |
| SVT / VT / VF | Supraventricular tachycardia / ventricular tachycardia / ventricular fibrillation |
| RBBB / LBBB | Right / left bundle branch block |
| AVB | Atrioventricular block |
| NSR | Normal sinus rhythm |
| TEE / TTE | Transesophageal / transthoracic echocardiogram |
| LVAD | Left ventricular assist device |
04 Stable Angina / Chronic Coronary Syndrome CAD
Chronic coronary syndrome (CCS), formerly "stable angina," is fixed atherosclerotic narrowing of coronary arteries causing a supply-demand mismatch during exertion. Coronary artery disease remains the leading cause of death worldwide, responsible for ~18 million deaths per year globally. Approximately 20 million Americans have CAD, and half of all men and one-third of women will develop symptomatic CAD during their lifetime.
Pathophysiology
Atherosclerotic plaque (lipid core, fibrous cap, inflammatory infiltrate, calcium) progressively narrows the coronary lumen. At rest, coronary autoregulation (arteriolar dilation) compensates for up to ~70% stenosis. During exercise or stress, increased myocardial oxygen demand outstrips the fixed supply, producing reversible ischemia — manifesting as angina. Angina is typically substernal chest pressure, radiating to the left arm, jaw, or back, provoked by exertion or emotional stress, and relieved by rest or sublingual nitroglycerin within 1–5 minutes. Duration is usually 2–10 minutes. Symptoms lasting >20 minutes suggest ACS.
CCS Angina Grading
| CCS Class | Description | Example |
|---|---|---|
| I | Angina only with strenuous activity | Running, heavy lifting |
| II | Slight limitation — angina with moderate exertion | Walking >2 blocks, climbing >1 flight of stairs |
| III | Marked limitation — angina with mild exertion | Walking 1–2 blocks, climbing 1 flight |
| IV | Angina at rest or with any activity | Cannot perform ADLs without angina |
Diagnostic Workup
First-line: Resting 12-lead EKG (often normal in stable angina; may show old Q waves, ST-T changes, or LVH). Stress testing is the cornerstone. Exercise stress test (Bruce protocol treadmill) is preferred if the patient can exercise and the resting EKG is interpretable. Positive test: ≥1 mm horizontal or downsloping ST depression. High-risk findings: ≥2 mm ST depression, ST depression in ≥5 leads, ST depression at <6 METs, hypotension during exercise, or Duke Treadmill Score ≤−11. If the patient cannot exercise, use pharmacologic stress (dobutamine for echocardiography, adenosine/regadenoson for nuclear perfusion). If resting EKG is uninterpretable (LBBB, paced rhythm, WPW, >1mm ST depression at baseline), use imaging stress (nuclear or echo).
CT coronary angiography (CCTA): Excellent negative predictive value for ruling out obstructive CAD in low-to-intermediate risk patients. Provides anatomic detail of plaque burden. Endorsed by the 2021 AHA/ACC Chest Pain Guideline.
Invasive coronary angiography: Gold standard for defining coronary anatomy. Reserved for patients with high-risk non-invasive findings, refractory symptoms despite medical therapy, or when intervention (PCI or CABG) is being planned.
Management
Optimal medical therapy (OMT) is the foundation: antiplatelet (ASA 81 mg daily), high-intensity statin (atorvastatin 40–80 mg or rosuvastatin 20–40 mg — target LDL <70 mg/dL), beta-blocker (metoprolol succinate 25–200 mg daily or atenolol — reduces HR and myocardial oxygen demand), sublingual nitroglycerin 0.4 mg PRN for acute episodes. Second-line anti-anginals: long-acting nitrates (isosorbide mononitrate 30–120 mg daily — must allow a nitrate-free interval of 10–14 hours to avoid tolerance), calcium channel blockers (amlodipine 5–10 mg or diltiazem ER 120–360 mg for patients intolerant of beta-blockers), ranolazine (Ranexa) 500–1000 mg BID (late sodium current inhibitor, no hemodynamic effect). BP target <130/80; diabetes A1C <7%; smoking cessation.
Revascularization: PCI is considered for angina refractory to OMT or high-risk anatomy. The ISCHEMIA trial (2020) demonstrated that an initial invasive strategy did not reduce death or MI compared to OMT alone in patients with stable CAD and moderate-to-severe ischemia, but did improve angina. CABG is preferred over PCI for left main disease, three-vessel disease (especially with diabetes), and complex multivessel disease (guided by the SYNTAX score).
05 Acute Coronary Syndromes (STEMI, NSTEMI, UA) CAD
ACS encompasses the spectrum of acute myocardial ischemia from unstable angina (UA) to NSTEMI to STEMI — all caused by acute disruption of an atherosclerotic plaque with superimposed thrombus formation. ACS accounts for >1.5 million hospital admissions per year in the US. In-hospital STEMI mortality has decreased to ~5–7% with modern primary PCI, but 1-year mortality remains ~10–12%.
Pathophysiology & Spectrum
Plaque rupture or erosion exposes the thrombogenic lipid core and subendothelial collagen, triggering platelet aggregation and the coagulation cascade. UA = partial/intermittent occlusion with ischemia but no myocyte death (troponin negative). NSTEMI = partial occlusion or complete occlusion with collateral supply, causing subendocardial necrosis (troponin positive, no ST elevation on EKG). STEMI = complete occlusion causing transmural ischemia/necrosis (troponin positive, ST elevation on EKG).
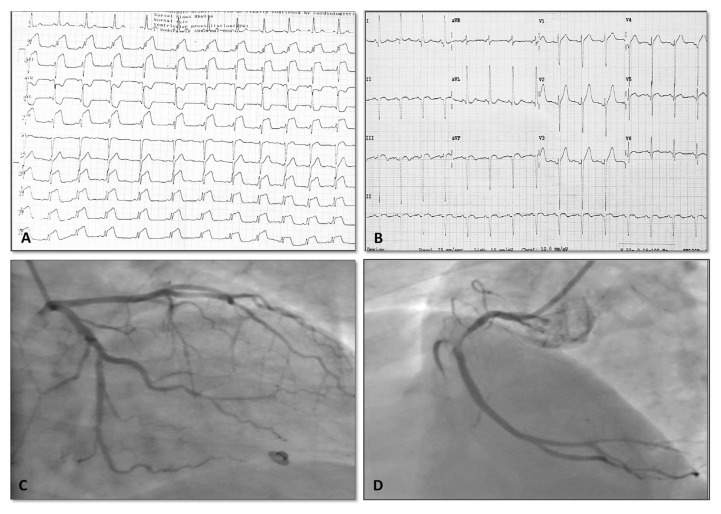
EKG Localization of STEMI
| EKG Leads with ST Elevation | Territory | Culprit Artery | Key Considerations |
|---|---|---|---|
| V1–V4 | Anterior wall | LAD | Highest mortality; large infarct area; risk of cardiogenic shock, VT/VF |
| V5–V6, I, aVL | Lateral wall | LCx or diagonal | May be subtle; check for reciprocal changes in III, aVF |
| II, III, aVF | Inferior wall | RCA (85%) or LCx (15%) | Check right-sided leads (V4R) for RV infarct; risk of bradycardia/heart block |
| V7–V9 (posterior leads) | Posterior wall | RCA or LCx | Look for ST depression V1–V3 (reciprocal changes); tall R waves in V1–V2 |
| V4R | Right ventricle | Proximal RCA | Hypotension with clear lungs; avoid nitrates and diuretics (preload-dependent) |
STEMI is a surgical emergency. Door-to-balloon time target: <90 minutes for primary PCI (or <120 min if transfer required). Fibrinolysis (alteplase, tenecteplase, or reteplase) is the alternative when PCI cannot be performed within 120 minutes of first medical contact. Symptom-onset-to-reperfusion time is the strongest determinant of myocardial salvage and survival. Every 30-minute delay in reperfusion increases relative mortality by ~7.5%.
STEMI Management
Immediate: ASA 325 mg chewed, P2Y12 inhibitor loading (ticagrelor 180 mg or prasugrel 60 mg preferred over clopidogrel 600 mg for PCI), anticoagulation (UFH bolus or bivalirudin), supplemental O₂ only if SpO₂ <90%, morphine for refractory pain (use cautiously — may reduce P2Y12 inhibitor absorption), nitroglycerin SL or IV (avoid in inferior/RV MI and if SBP <90). Primary PCI is the preferred reperfusion strategy: radial artery access preferred (lower bleeding/vascular complications vs femoral — per MATRIX trial), aspiration thrombectomy + drug-eluting stent (DES) to the culprit lesion. Post-PCI DAPT: ASA indefinitely + P2Y12 inhibitor for at least 12 months (can shorten to 6 months if high bleeding risk, or extend beyond 12 months if high ischemic risk and tolerated — per PEGASUS-TIMI 54).
NSTEMI / UA Management
Risk stratify using the TIMI score (0–7 points: age ≥65, ≥3 CAD risk factors, known CAD ≥50% stenosis, ASA use in past 7 days, ≥2 anginal events in 24h, ST deviation ≥0.5 mm, elevated troponin) or GRACE score (more accurate for in-hospital mortality). Early invasive strategy (angiography within 24–72 hours) is recommended for high-risk patients: TIMI ≥3, recurrent angina, hemodynamic instability, new HF, elevated troponin, or dynamic ST changes. Very high-risk features (ongoing chest pain, hemodynamic instability, VT/VF, acute HF) warrant emergent catheterization (<2 hours). An initial conservative (ischemia-guided) strategy is reasonable for low-risk patients.
06 Post-MI Complications CAD
Post-MI complications are time-dependent and must be anticipated. They are a frequent cause of in-hospital deterioration and a board exam staple.
| Timing | Complication | Mechanism / Presentation | Management |
|---|---|---|---|
| Hours | Arrhythmias (VT/VF, AFib, bradycardia) | VT/VF most common cause of early death; reperfusion arrhythmias (AIVR) usually benign | Defibrillation for VF; amiodarone/lidocaine for VT; atropine/pacing for symptomatic bradycardia |
| Hours–days | Cardiogenic shock | Extensive LV damage (>40% of myocardium); hypotension + pulmonary edema + end-organ hypoperfusion | Emergent revascularization; vasopressors; mechanical support (IABP, Impella, ECMO) |
| Days 1–3 | Pericarditis (early post-MI) | Transmural inflammation; pleuritic chest pain, friction rub, diffuse ST elevation | ASA high-dose; avoid NSAIDs/steroids (impair scar healing) |
| Days 3–5 | Papillary muscle rupture | Necrosis of posteromedial papillary muscle (single blood supply from PDA) → acute severe MR → flash pulmonary edema + new holosystolic murmur | Surgical emergency: emergent MV repair/replacement; IABP to reduce afterload as bridge |
| Days 3–5 | Ventricular septal rupture | Septal necrosis → new VSD → left-to-right shunt → acute HF + new harsh holosystolic murmur + oxygen step-up on right heart catheterization | Surgical emergency: emergent surgical repair; IABP as bridge |
| Days 5–14 | Free wall rupture | LV free wall tears → hemopericardium → tamponade → PEA/death. Risk factors: first MI, anterior wall, large transmural infarct, age >65, female | Immediately lethal without surgery: emergent pericardiocentesis + surgical repair |
| Weeks | LV aneurysm | Dyskinetic thin-walled segment; persistent ST elevation on EKG; risk of thrombus, VT, HF | Anticoagulation if thrombus; surgical aneurysmectomy if refractory VT or HF |
| Weeks 2–12 | Dressler syndrome | Autoimmune pericarditis; fever, pleuritic pain, pericardial effusion, elevated ESR | ASA or NSAIDs + colchicine |
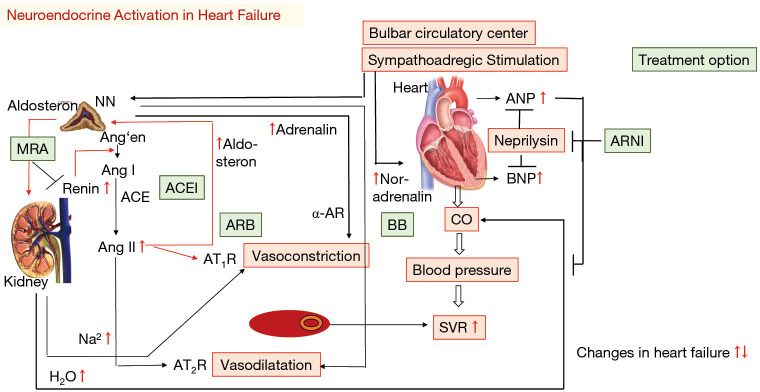
07 Heart Failure with Reduced EF (HFrEF) HF
HFrEF is defined as clinical heart failure with LVEF ≤40%. It affects ~6.7 million Americans, with 1 million new diagnoses per year. Five-year mortality remains ~50%. Ischemic cardiomyopathy (post-MI LV dysfunction) is the most common cause, followed by non-ischemic dilated cardiomyopathy (idiopathic, alcohol, viral, familial, toxins like anthracyclines).
ACC/AHA Heart Failure Stages
| Stage | Description | Examples | Treatment Focus |
|---|---|---|---|
| A | At risk — no structural heart disease, no symptoms | HTN, DM, CAD, obesity, FHx of CMP, cardiotoxin exposure | Risk factor modification; SGLT2i in DM |
| B | Pre-HF — structural heart disease but no symptoms | Prior MI, LVH, reduced EF, valvular disease (asymptomatic) | ACEi/ARB, beta-blocker if reduced EF or prior MI |
| C | Symptomatic HF | Current or prior HF symptoms with structural disease | Full GDMT (see below) |
| D | Advanced HF — refractory symptoms despite maximal therapy | Recurrent hospitalizations, inotrope dependence, LVAD/transplant candidate | Palliative care, inotropes, MCS, transplant evaluation |
NYHA Functional Classification
| NYHA Class | Symptoms | Functional Limitation |
|---|---|---|
| I | No symptoms with ordinary physical activity | None |
| II | Symptoms with ordinary activity (walking, climbing stairs) | Slight |
| III | Symptoms with less-than-ordinary activity | Marked |
| IV | Symptoms at rest | Unable to carry on any physical activity without discomfort |
GDMT — The Four Pillars of HFrEF Therapy
The 2022 AHA/ACC/HFSA Guideline emphasizes simultaneous initiation of all four drug classes (not sequential titration) for Stage C HFrEF:

| Pillar | Drug Class | Example | Key Benefit | Critical Pearl |
|---|---|---|---|---|
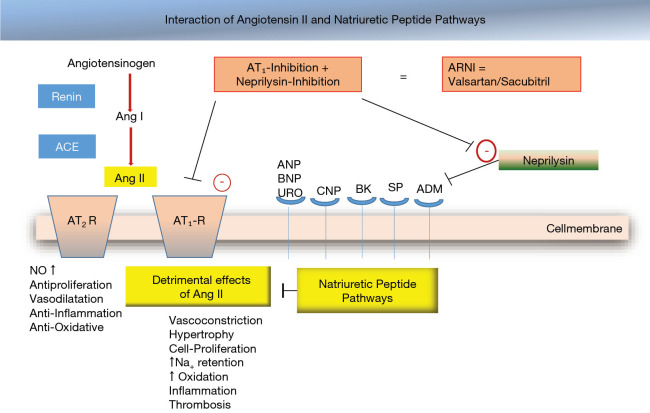
| 1 | ARNI (or ACEi/ARB) | Sacubitril/valsartan (Entresto) 24/26 → 97/103 mg BID | 20% mortality reduction vs enalapril (PARADIGM-HF) | 36-hour washout from ACEi before starting ARNI (angioedema risk); do not use with ACEi |
| 2 | Beta-blocker | Carvedilol 3.125 → 25 mg BID; metoprolol succinate 12.5 → 200 mg daily; bisoprolol 1.25 → 10 mg daily | ~35% mortality reduction | Only these 3 beta-blockers are evidence-based for HFrEF; do not start during acute decompensation |
| 3 | MRA | Spironolactone 12.5–50 mg daily; eplerenone 25–50 mg daily | 30% mortality reduction (RALES) | Monitor K+ and Cr; contraindicated if K+ >5.0 or eGFR <30 |
| 4 | SGLT2 inhibitor | Dapagliflozin (Farxiga) 10 mg daily; empagliflozin (Jardiance) 10 mg daily | ~25% reduction in HF hospitalization + CV death (DAPA-HF, EMPEROR-Reduced) | Benefit independent of diabetes status; monitor for genital mycotic infections, ketoacidosis risk in T1DM |
Additional therapies: Hydralazine/isosorbide dinitrate (BiDil) — recommended for Black patients already on standard GDMT or those intolerant of ACEi/ARB/ARNI (A-HeFT trial showed 43% mortality reduction in Black patients). Ivabradine (Corlanor) — funny-channel (If) inhibitor; for patients in sinus rhythm with HR ≥70 despite max beta-blocker. Diuretics (furosemide, bumetanide, torsemide) — for volume management (symptom relief) but no mortality benefit. Digoxin — reduces HF hospitalizations but not mortality; narrow therapeutic index (target 0.5–0.9 ng/mL).
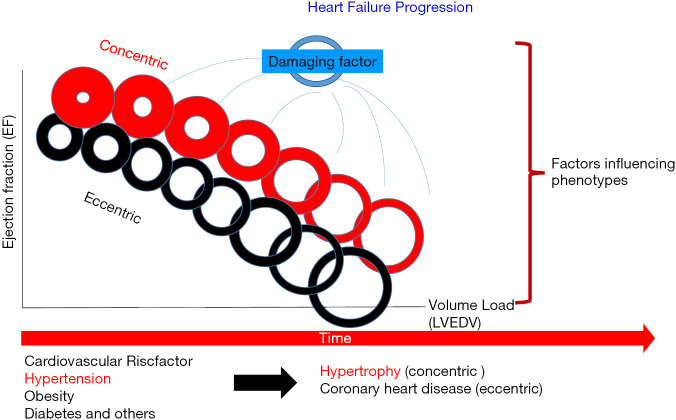
08 Heart Failure with Preserved EF (HFpEF) HF
HFpEF is defined as clinical heart failure with LVEF ≥50% (HFmrEF = EF 41–49%). It accounts for ~50% of all HF and is increasing in prevalence, driven by the aging population, obesity, hypertension, and diabetes. Unlike HFrEF, most traditional HF therapies failed to show mortality benefit in HFpEF — until SGLT2 inhibitors.
Pathophysiology
Diastolic dysfunction is the hallmark — impaired LV relaxation and increased LV stiffness lead to elevated filling pressures (LVEDP/PCWP) and pulmonary congestion, despite normal systolic function. Common phenotypes: elderly hypertensive with concentric LVH, obese/metabolic syndrome, atrial fibrillation-related, and pulmonary hypertension-predominant. The H2FPEF score (Heavy/BMI >30, Hypertensive, AFib, Pulmonary hypertension, Elder >60, Filling pressure/E/e' >9) helps diagnose HFpEF when noninvasive data is equivocal.
Diagnosis
Elevated BNP/NT-proBNP with LVEF ≥50% plus evidence of diastolic dysfunction on echocardiography: elevated E/e' ratio (>14 suggests elevated filling pressures), LA dilation (LA volume index >34 mL/m²), elevated estimated RVSP (>35 mmHg), LV hypertrophy. If borderline, exercise diastolic stress testing or invasive hemodynamics (exercise right heart catheterization showing PCWP >25 mmHg with exercise) may confirm the diagnosis.
Management
SGLT2 inhibitors are the first class with proven benefit: empagliflozin reduced the composite of CV death or HF hospitalization by 21% in EMPEROR-Preserved, and dapagliflozin showed similar results in DELIVER — both regardless of diabetes status. Diuretics for volume management remain essential. Treat comorbidities aggressively: HTN (target <130/80), AFib (rate/rhythm control), obesity (GLP-1 agonists showing HF benefit in STEP-HFpEF), diabetes, sleep apnea. MRAs (spironolactone) showed mixed results (TOPCAT — benefit in Americas cohort but not Russia/Georgia cohort); considered reasonable in selected patients.
09 Acute Decompensated Heart Failure HF
ADHF is the most common cause of hospital admission in patients >65 years. Presentation: worsening dyspnea (exertional → orthopnea → PND → dyspnea at rest), weight gain, peripheral edema, JVD, pulmonary crackles/rales, S3 gallop. Precipitants: medication/dietary non-adherence (most common), ischemia, arrhythmia (new AFib), infection, anemia, thyroid disease, PE, uncontrolled HTN.
Classification — Warm/Cold, Wet/Dry
| Dry (not congested) | Wet (congested) | |
|---|---|---|
| Warm (adequate perfusion) | Warm & Dry — compensated; optimize oral meds | Warm & Wet (most common ADHF) — IV diuretics, vasodilators |
| Cold (hypoperfused) | Cold & Dry — may need fluid challenge; consider inotropes | Cold & Wet — worst prognosis; inotropes + diuretics; consider MCS |
Management
IV loop diuretics are the mainstay: furosemide IV bolus (1–2.5× home oral dose) or continuous infusion; bumetanide 1–2 mg IV; torsemide. Target urine output >0.5 mL/kg/hr or net negative 1–2 L/day. If diuretic-resistant: add metolazone 2.5–5 mg PO 30 min before loop diuretic ("sequential nephron blockade"), or add chlorothiazide 250–500 mg IV, or add acetazolamide 500 mg IV (per ADVOR trial). Vasodilators: IV nitroglycerin (reduces preload, improves congestion) or nitroprusside (reduces afterload; caution with renal insufficiency — cyanide toxicity). Do not start or uptitrate beta-blockers during acute decompensation; hold if hypotensive. Continue ACEi/ARB/ARNI if BP tolerates.
Killip class predicts in-hospital mortality post-MI. Class I: No HF signs (mortality ~6%). Class II: Rales in lower lung fields, S3, elevated JVP (mortality ~17%). Class III: Acute pulmonary edema (mortality ~38%). Class IV: Cardiogenic shock (mortality ~60–80% without intervention).
10 Cardiogenic Shock HF
Cardiogenic shock is the most severe form of heart failure — inadequate cardiac output resulting in end-organ hypoperfusion despite adequate intravascular volume. Mortality remains 40–50% even with aggressive treatment. Most commonly caused by acute MI (especially large anterior STEMI), but also acute valvular catastrophe, myocarditis, acute on chronic HF, and takotsubo.
SCAI Shock Classification
| Stage | Name | Definition | Key Features |
|---|---|---|---|
| A | At Risk | Not currently in shock but at risk | Large acute MI, prior severe HF |
| B | Beginning | Clinical hypotension or tachycardia without hypoperfusion | SBP <90 or MAP <60; HR >100; still warm extremities |
| C | Classic | Hypoperfusion requiring intervention | Cold extremities, altered mental status, elevated lactate >2, oliguria; CI <2.2, PCWP >15 |
| D | Deteriorating | Not responding to initial interventions | Escalating vasopressors/inotropes; worsening end-organ function |
| E | Extremis | Cardiac arrest or refractory circulatory collapse | PEA, VF, refractory hypotension on multiple pressors; consider MCS or palliative care |
Hemodynamic Profile
Swan-Ganz catheter findings: elevated PCWP (>15–18 mmHg), low cardiac index (<2.2 L/min/m²), elevated SVR (>1200 dynes·s/cm&sup5;), low mixed venous O₂ saturation (<60%). Distinguish from other shock states: septic shock has low SVR and often elevated CI; hypovolemic shock has low PCWP; obstructive shock (PE, tamponade) has specific hemodynamic signatures.
Management
Vasopressors/inotropes: Norepinephrine is first-line vasopressor (alpha + beta; preferred over dopamine per SOAP II trial). Dobutamine (beta-1 agonist, inotrope) added if CI remains low. Milrinone (PDE3 inhibitor, inodilator) — increases contractility and reduces afterload; useful in RV failure and pulmonary hypertension. Mechanical circulatory support (MCS): IABP (intra-aortic balloon pump) — inflates during diastole (augments coronary perfusion), deflates during systole (reduces afterload); provides ~0.5 L/min augmentation. Impella (axial flow pump, percutaneous LV-to-aorta) — provides 2.5–5.5 L/min depending on model (Impella CP, 5.0, 5.5). VA-ECMO (venoarterial extracorporeal membrane oxygenation) — provides full cardiopulmonary support for refractory shock. Emergent revascularization for MI-related cardiogenic shock.
11 Atrial Fibrillation & Flutter Arrhythmia
Atrial fibrillation (AF) is the most common sustained arrhythmia, affecting ~6 million Americans and ~33 million people globally. AF is characterized by disorganized atrial electrical activity (no discrete P waves, irregularly irregular ventricular response) originating predominantly from the pulmonary vein ostia. AF increases stroke risk 5-fold and is an independent predictor of mortality. Atrial flutter (AFL) is a macro-reentrant circuit, usually in the right atrium around the tricuspid annulus (typical flutter), producing "sawtooth" flutter waves at ~300 bpm with a ventricular rate often ~150 bpm (2:1 block).
AF Classification
| Type | Duration | Treatment Implication |
|---|---|---|
| Paroxysmal | Self-terminating within 7 days | Rhythm control often pursued; antiarrhythmics or ablation |
| Persistent | >7 days, requires intervention to terminate | Cardioversion + antiarrhythmics; ablation if drugs fail |
| Long-standing persistent | >12 months | Ablation success lower; rate control may be preferred |
| Permanent | Decision made to accept AF | Rate control only; no further rhythm control attempts |
CHA₂DS₂-VASc Score (Stroke Risk)
| Factor | Points |
|---|---|
| C — CHF / LV dysfunction (EF ≤40%) | 1 |
| H — Hypertension | 1 |
| A₂ — Age ≥75 | 2 |
| D — Diabetes mellitus | 1 |
| S₂ — Stroke / TIA / thromboembolism | 2 |
| V — Vascular disease (prior MI, PAD, aortic plaque) | 1 |
| A — Age 65–74 | 1 |
| Sc — Sex category (female) | 1 |
Anticoagulation: Score ≥2 in men or ≥3 in women → OAC recommended (DOAC preferred over warfarin: apixaban 5 mg BID, rivaroxaban 20 mg daily, dabigatran 150 mg BID, or edoxaban 60 mg daily). Score of 1 in men or 2 in women → OAC should be considered. Score 0 → no OAC. DOACs are preferred over warfarin based on superior safety profiles (lower intracranial bleeding) in RE-LY, ROCKET-AF, ARISTOTLE, and ENGAGE AF-TIMI 48 trials.
HAS-BLED Score (Bleeding Risk)
| Factor | Points |
|---|---|
| H — Hypertension (uncontrolled, SBP >160) | 1 |
| A — Abnormal renal/liver function (1 each) | 1–2 |
| S — Stroke history | 1 |
| B — Bleeding history or predisposition | 1 |
| L — Labile INR (if on warfarin; TTR <60%) | 1 |
| E — Elderly (>65) | 1 |
| D — Drugs (antiplatelets, NSAIDs) or alcohol (1 each) | 1–2 |
Score ≥3 = high bleeding risk. This does not mean withhold anticoagulation — it means address modifiable risk factors (control BP, stop NSAIDs, limit alcohol, improve INR stability). High CHA₂DS₂-VASc scores typically outweigh high HAS-BLED scores.
Rate vs Rhythm Control
Rate control: Target resting HR <110 bpm (lenient) is adequate per RACE II trial; <80 bpm (strict) if symptomatic. Agents: beta-blockers (metoprolol, atenolol), non-dihydropyridine CCBs (diltiazem, verapamil — avoid in HFrEF), digoxin (adjunct for rate control in HF). Rhythm control: The EAST-AFNET 4 trial (2020) showed early rhythm control (within 1 year of diagnosis) reduced cardiovascular outcomes by 21%, shifting practice toward earlier rhythm control. Antiarrhythmics: flecainide or propafenone (Class IC — only in structurally normal hearts), sotalol (Class III), amiodarone (Class III — most effective but toxic), dofetilide (Class III — requires inpatient QTc monitoring for initiation), dronedarone (Class III — contraindicated in permanent AF and NYHA III/IV HF).
12 Supraventricular Tachycardias (SVT) Arrhythmia
SVT is a generic term for tachyarrhythmias (HR >100) originating above the ventricles. The most common mechanism is AVNRT (AV nodal reentrant tachycardia, ~60% of SVTs) — a reentrant circuit within or near the AV node using slow and fast pathways. Second most common is AVRT (AV reciprocating tachycardia, ~30%) — uses an accessory pathway (e.g., Bundle of Kent in WPW). Atrial tachycardia (~10%) originates from an ectopic atrial focus.
Acute Management
First: Vagal maneuvers (carotid sinus massage, Valsalva with modified Valsalva technique — strain for 15 sec then lie supine with legs raised; converts ~25–45%). If failed: Adenosine 6 mg rapid IV push (must be rapid with flush; if no effect, repeat 12 mg) — transiently blocks AV node conduction, terminating reentrant SVTs (success rate ~90% for AVNRT/AVRT). Adenosine is diagnostic and therapeutic. If refractory: IV diltiazem, verapamil, or beta-blocker. Synchronized cardioversion for hemodynamically unstable SVT.
WPW syndrome = accessory pathway (Bundle of Kent) + delta wave on baseline EKG + SVT episodes. If a WPW patient develops AF, the accessory pathway can conduct very rapidly to the ventricles (bypassing AV node), producing a wide-complex irregularly irregular tachycardia that can degenerate into VF. AV nodal blockers (adenosine, diltiazem, verapamil, digoxin) are CONTRAINDICATED in pre-excited AF — they block the AV node, forcing all conduction down the accessory pathway. Treatment: IV procainamide or ibutilide; or emergent cardioversion if unstable.
13 Ventricular Tachycardia & Fibrillation Arrhythmia
Ventricular tachycardia (VT) = ≥3 consecutive ventricular beats at >100 bpm. Non-sustained VT (NSVT) = lasts <30 seconds and self-terminates. Sustained VT = lasts ≥30 seconds or requires intervention. Monomorphic VT (uniform QRS) usually arises from scar-related reentry (prior MI) or idiopathic VT (structurally normal heart — RVOT VT, fascicular VT). Polymorphic VT (varying QRS morphology) with normal QT interval is usually ischemia-related. Torsades de pointes = polymorphic VT with prolonged QT — sine-wave "twisting of the points" pattern.
Management
Pulseless VT/VF: ACLS protocol — immediate defibrillation (120–200J biphasic), CPR, epinephrine 1 mg IV q3–5 min, amiodarone 300 mg IV (then 150 mg). Stable monomorphic VT: IV amiodarone (150 mg over 10 min), IV procainamide, or IV lidocaine. Synchronized cardioversion if drug-refractory. Torsades de pointes: IV magnesium 2g bolus, overdrive pacing (increases HR, shortens QT), isoproterenol; discontinue all QT-prolonging drugs. VT storm (≥3 sustained VT episodes in 24 hours): IV amiodarone + beta-blocker (esmolol or propranolol), sedation, emergent catheter ablation if refractory, stellate ganglion block or epidural for sympatholysis.
14 Bradyarrhythmias & Heart Block Arrhythmia
Bradycardia is HR <60 bpm. Symptomatic bradycardia (syncope, pre-syncope, HF, fatigue, exercise intolerance) requires treatment. Causes: intrinsic conduction system disease (fibrosis/degeneration — most common cause for permanent pacemaker), medications (beta-blockers, CCBs, digoxin, amiodarone), increased vagal tone (athletes, sleep), hypothyroidism, hyperkalemia, inferior MI (AV node ischemia).
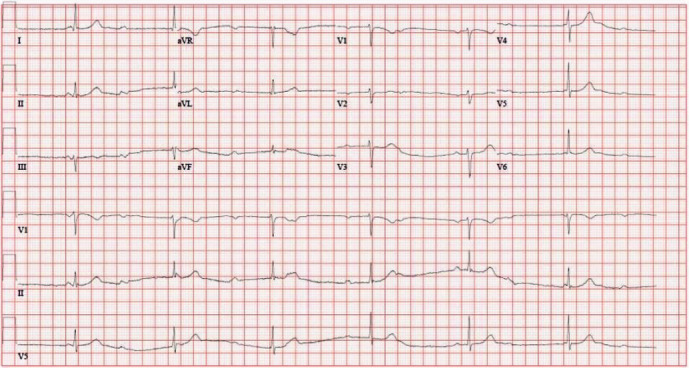
AV Block Classification
| Type | EKG Finding | Clinical Significance | Pacemaker? |
|---|---|---|---|
| 1st degree | PR >200 ms, all P waves conducted | Usually benign; prolonged AV conduction | No (unless symptomatic) |
| 2nd degree, Mobitz I (Wenckebach) | Progressive PR prolongation → dropped QRS | Usually AV nodal level; benign in most cases; common in inferior MI | Rarely needed |
| 2nd degree, Mobitz II | Fixed PR intervals with sudden dropped QRS | Below AV node (His-Purkinje); high risk of progression to complete block | Yes — pacemaker indicated |
| 3rd degree (complete) | No relationship between P waves and QRS; independent atrial and ventricular rates | No AV conduction; escape rhythm determines stability (junctional 40–60 bpm, ventricular 20–40 bpm) | Yes — pacemaker indicated |
Acute Management
Symptomatic bradycardia: Atropine 0.5–1 mg IV (may repeat q3–5 min, max 3 mg; less effective for infranodal block). Transcutaneous pacing as bridge. Dopamine or epinephrine infusion if atropine/pacing inadequate. Transvenous pacing for refractory cases. Medication-induced: Glucagon for beta-blocker overdose; calcium for CCB overdose; digoxin-specific antibody fragments (Digibind/DigiFab) for digoxin toxicity.
15 Sudden Cardiac Death & Channelopathies Arrhythmia
Sudden cardiac death (SCD) claims ~350,000 lives per year in the US. The most common cause in adults >35 is CAD/ischemic cardiomyopathy. In adults <35, the leading causes are HCM, arrhythmogenic right ventricular cardiomyopathy (ARVC), coronary artery anomalies, and channelopathies.
Key Channelopathies
| Condition | Mechanism | EKG Features | Management |
|---|---|---|---|
| Long QT Syndrome (LQTS) | Delayed cardiac repolarization (K+ or Na+ channel mutations) | QTc >470 ms (male) or >480 ms (female); risk of torsades | Beta-blockers (nadolol preferred); avoid QT-prolonging drugs; ICD if high-risk (LQT3, cardiac arrest survivor) |
| Brugada Syndrome | Na+ channel (SCN5A) dysfunction | Coved ST elevation ≥2 mm in V1–V3 (Type 1 pattern) | ICD for cardiac arrest survivors or syncope with Type 1 EKG; avoid Na+ channel blockers, fever (unmasks pattern) |
| Catecholaminergic Polymorphic VT (CPVT) | Ryanodine receptor (RyR2) mutation → abnormal calcium handling | Normal baseline EKG; bidirectional VT with exercise/catecholamines | Beta-blockers (nadolol); flecainide as add-on; ICD if arrest survivor; restrict strenuous exercise |
| Short QT Syndrome | Gain-of-function K+ channel mutations | QTc <340 ms; tall peaked T waves | ICD; quinidine may prolong QT |
ICD Indications for Primary Prevention of SCD
Per 2017 AHA/ACC/HRS guidelines: ICD is recommended for patients with LVEF ≤35% despite ≥3 months of GDMT and ≥40 days post-MI, with NYHA Class II–III symptoms and expected survival >1 year. Also for HCM with major risk factors, ARVC with sustained VT or significant RV dysfunction, and channelopathy survivors of cardiac arrest.
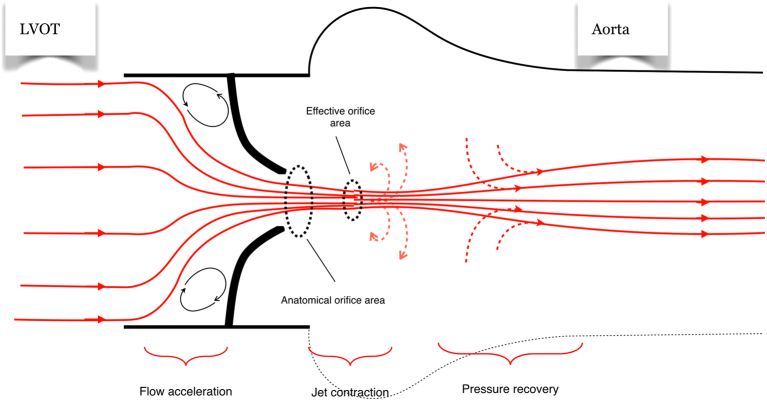
16 Aortic Stenosis Valvular
Aortic stenosis (AS) is the most common valvular disease requiring intervention in developed countries. The most common etiology in adults >70 is calcific/degenerative disease of a trileaflet valve. In patients 50–70, bicuspid aortic valve (BAV, prevalence ~1–2%) is the most common cause. Rheumatic AS is rare in the developed world but remains common globally. Once symptomatic, prognosis is poor without intervention: mean survival after angina onset = ~5 years, after syncope = ~3 years, after heart failure = ~2 years.
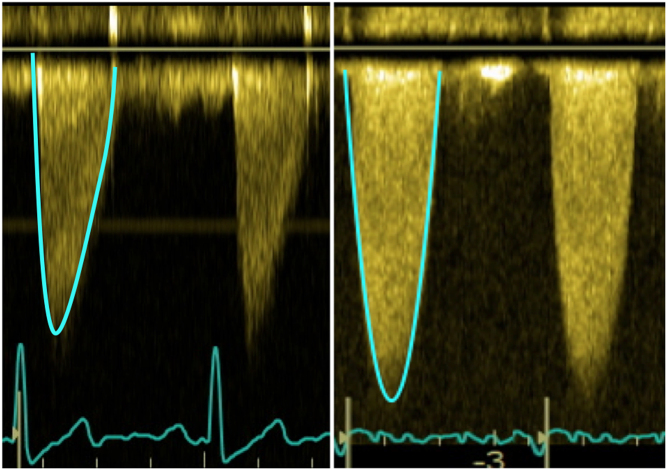
Severity Grading
| Parameter | Mild | Moderate | Severe |
|---|---|---|---|
| Aortic valve area (AVA) | >1.5 cm² | 1.0–1.5 cm² | <1.0 cm² |
| Mean gradient | <20 mmHg | 20–40 mmHg | >40 mmHg |
| Peak jet velocity | <3.0 m/s | 3.0–4.0 m/s | >4.0 m/s |
Low-flow, low-gradient severe AS: AVA <1.0 cm² but mean gradient <40 mmHg due to reduced LV function (EF <50%). Dobutamine stress echo helps differentiate true severe AS (AVA remains <1.0 with increased flow) from pseudo-severe AS (AVA increases to >1.0 — the valve opens more with higher flow).
Treatment
No medical therapy alters the natural history of AS. Intervention is indicated for: symptomatic severe AS (dyspnea, angina, syncope), asymptomatic severe AS with LVEF <50%, asymptomatic severe AS with very severe disease (Vmax ≥5 m/s), or asymptomatic severe AS with rapid progression or exercise-induced symptoms. SAVR (surgical aortic valve replacement) is the standard for low/intermediate surgical risk patients. TAVR (transcatheter aortic valve replacement) is approved for all surgical risk categories and is now the dominant approach for patients ≥65 years. Major TAVR trials: PARTNER series (balloon-expandable Edwards SAPIEN valve) and CoreValve/Evolut trials (self-expanding Medtronic CoreValve/Evolut). TAVR carries higher rates of paravalvular leak and pacemaker need; SAVR carries higher rates of bleeding and renal injury.
17 Aortic Regurgitation Valvular
Aortic regurgitation (AR) is backflow from the aorta into the LV during diastole. Chronic AR causes volume overload with progressive LV dilation (eccentric hypertrophy) — the LV compensates for years before decompensating. Common causes: bicuspid aortic valve, aortic root dilation (HTN, Marfan, bicuspid aortopathy), rheumatic disease, degenerative disease. Acute AR is a surgical emergency — the LV cannot accommodate the sudden volume load, causing acute pulmonary edema and cardiogenic shock. Causes: aortic dissection (flap disrupts valve coaptation), endocarditis (leaflet perforation/destruction), trauma.
Clinical Findings
Chronic severe AR produces a wide pulse pressure (e.g., 180/40) and numerous eponymous signs reflecting bounding pulses: Corrigan's pulse (water-hammer pulse), de Musset's sign (head bobbing), Quincke's sign (nail bed pulsations), Duroziez's sign (to-and-fro femoral murmur), Hill's sign (leg SBP >20 mmHg higher than arm). The murmur is an early diastolic decrescendo blowing murmur best heard at the LUSB with the patient sitting up and leaning forward. A mid-diastolic rumble at the apex (Austin Flint murmur) can occur from the regurgitant jet impinging on the anterior mitral leaflet.
Intervention Criteria
Surgical AVR is indicated for: symptomatic severe AR, asymptomatic severe AR with LVEF <55% or LVESD >50 mm (LV is dilating beyond compensation). Acute severe AR requires emergent surgery — IABP is contraindicated (diastolic augmentation worsens regurgitation).
18 Mitral Regurgitation Valvular
Mitral regurgitation (MR) is the most common valvular lesion in the US. Two pathologic types: Primary (organic) MR — intrinsic valve pathology (MVP with flail leaflet, rheumatic disease, endocarditis, annular calcification). Secondary (functional) MR — normal valve leaflets with regurgitation due to LV dilation/dysfunction or annular dilation (ischemic or non-ischemic cardiomyopathy).
Severity Assessment (by Echo)
| Parameter | Mild | Moderate | Severe |
|---|---|---|---|
| Regurgitant volume | <30 mL | 30–59 mL | ≥60 mL |
| Regurgitant fraction | <30% | 30–49% | ≥50% |
| EROA | <0.20 cm² | 0.20–0.39 cm² | ≥0.40 cm² |
| Vena contracta | <0.3 cm | 0.3–0.69 cm | ≥0.7 cm |
Management
Primary severe MR: Surgery (MV repair preferred over replacement; repair has lower operative mortality and better long-term outcomes) is indicated when symptomatic or when asymptomatic with LVEF ≤60% or LVESD ≥40 mm. Secondary severe MR: Optimize GDMT first (HF therapy often reduces MR). MitraClip (Abbott) — transcatheter edge-to-edge repair — was shown to reduce HF hospitalization and death in functional MR by 47% in the COAPT trial (in patients with disproportionate MR relative to LV dilation and on maximized GDMT).
19 Mitral Stenosis Valvular
Mitral stenosis (MS) is overwhelmingly caused by rheumatic heart disease (chronic inflammatory scarring fuses the commissures, thickens leaflets, and shortens chordae). Normal mitral valve area is 4–6 cm². Symptoms typically develop when MVA <2.0 cm². Severe MS = MVA <1.0 cm². MS causes LA pressure elevation → LA dilation → pulmonary congestion → pulmonary hypertension → eventual RV failure. The dilated LA is a substrate for AF (present in ~40% of symptomatic MS patients) and LA thrombus formation.
Clinical Presentation
Dyspnea (exertional → orthopnea → PND), hemoptysis (bronchial vein rupture from elevated LA pressure), hoarseness (Ortner syndrome — enlarged LA compresses recurrent laryngeal nerve), and malar flush (mitral facies — pinkish-purple discoloration of cheeks from chronic low CO). New-onset AF often precipitates acute decompensation (loss of atrial kick + shortened diastolic filling time at rapid rates).
Treatment
Medical: rate control (beta-blockers or CCBs to prolong diastolic filling time), diuretics for congestion, anticoagulation if AF or LA thrombus. Percutaneous mitral balloon commissurotomy (PMBC) is the treatment of choice for symptomatic moderate-to-severe MS with favorable valve morphology (Wilkins score ≤8, no more than mild MR, no LA thrombus). Surgical MV replacement for unfavorable anatomy or failed PMBC.
20 Tricuspid & Pulmonic Valve Disease Valvular
Tricuspid regurgitation (TR) is the most common right-sided valve lesion. Primary causes: endocarditis (especially IV drug users), carcinoid syndrome, Ebstein's anomaly, rheumatic disease. Secondary (functional) TR is far more common — caused by RV/annular dilation from pulmonary hypertension, left-sided heart disease, or chronic AF. Severe TR causes hepatic congestion (pulsatile liver, elevated LFTs), ascites, peripheral edema, and JVD with prominent V waves.
Pulmonic stenosis (PS) is almost always congenital. Mild PS is well-tolerated lifelong. Severe PS (peak gradient >60 mmHg) is treated with balloon valvuloplasty. Pulmonic regurgitation is usually physiologic (trace PR is normal) or secondary to pulmonary hypertension.
TR management: Treat underlying cause (PH therapy, left-sided valve correction). Diuretics for congestion. Surgical TV repair/replacement for severe TR during left-sided valve surgery. Emerging transcatheter options include the TriClip (edge-to-edge repair) and EVOQUE valve replacement systems.
21 Prosthetic Valve Management Valvular
Valve Types
| Type | Examples | Durability | Anticoagulation | Key Issue |
|---|---|---|---|---|
| Mechanical | St. Jude (bileaflet), Medtronic Hall (tilting disc), ball-and-cage (Starr-Edwards, historical) | Lifelong (>20–30 years) | Lifelong warfarin required (DOACs contraindicated) | Thrombosis risk if subtherapeutic INR; hemolysis |
| Bioprosthetic | Edwards Perimount, Medtronic Hancock, porcine or bovine pericardial | 10–20 years (degenerate faster in younger patients) | Warfarin × 3–6 months (aortic) or warfarin × 3–6 months (mitral), then ASA alone | Structural valve degeneration over time; valve-in-valve TAVR possible |
| TAVR valves | Edwards SAPIEN (balloon-expandable), Medtronic Evolut (self-expanding) | Data emerging; >5–10 year durability demonstrated | DAPT × 3–6 months, then ASA alone (or OAC if AF) | Paravalvular leak; conduction disturbances (pacemaker need ~10–20%) |
INR Targets for Mechanical Valves
| Position | INR Target | Notes |
|---|---|---|
| Aortic (bileaflet or tilting disc, no risk factors) | 2.5 (range 2.0–3.0) | Lower risk position; most blood flows through valve |
| Mitral (any mechanical) | 3.0 (range 2.5–3.5) | Higher thrombotic risk due to lower flow velocity |
| Aortic + risk factors (AF, prior thromboembolism, hypercoagulable) | 3.0 (range 2.5–3.5) | Add ASA 81 mg for very high-risk patients |
Suspect in any patient with a mechanical valve and new symptoms (dyspnea, embolic event) or new hemodynamic findings (increased gradient on echo). TEE is the diagnostic study of choice. Treatment: IV heparin, fibrinolysis (tPA) for non-obstructive or high surgical risk; emergent surgical thrombectomy/valve replacement for obstructive thrombosis in operative candidates.
22 Infective Endocarditis Valvular
Infective endocarditis (IE) is infection of the endocardial surface, typically involving heart valves. In-hospital mortality is 15–25%. Risk factors: prosthetic valves, structural heart disease, IV drug use (IVDU), poor dentition, central venous catheters, immunosuppression. IVDU-associated IE classically affects the tricuspid valve (Staph aureus >70%).
Modified Duke Criteria
Definite IE: 2 major, or 1 major + 3 minor, or 5 minor criteria.
| Major Criteria | Minor Criteria |
|---|---|
| 1. Blood cultures: typical IE organism from 2 separate cultures (viridans streptococci, S. bovis, HACEK, community-acquired S. aureus or enterococci without primary focus), or persistently positive cultures | 1. Predisposing condition (valve disease, IVDU) |
| 2. Endocardial involvement: vegetation, abscess, or new prosthetic valve dehiscence on echo; or new valvular regurgitation | 2. Fever ≥38°C |
| 3. Vascular phenomena: Janeway lesions, septic pulmonary emboli, mycotic aneurysm, conjunctival hemorrhage | |
| 4. Immunologic phenomena: Osler nodes, Roth spots, glomerulonephritis, positive rheumatoid factor | |
| 5. Positive blood cultures not meeting major criteria |
Treatment
Prolonged IV antibiotics (4–6 weeks): native valve strep — penicillin G or ceftriaxone ± gentamicin; staph — nafcillin/oxacillin (MSSA) or vancomycin (MRSA) ± gentamicin; enterococcal — ampicillin + gentamicin or ampicillin + ceftriaxone. Surgical indications: severe HF from valvular destruction, uncontrolled infection (abscess, persistent bacteremia >5–7 days), recurrent emboli, large mobile vegetations (>10 mm especially on the anterior mitral leaflet), prosthetic valve endocarditis with valve dysfunction.
23 Pericarditis (Acute & Constrictive) Peri/Myo
Acute Pericarditis
Inflammation of the pericardium, most commonly idiopathic/viral (Coxsackievirus, echovirus, CMV, EBV) in developed countries. Other causes: post-MI (early or Dressler syndrome), uremia, autoimmune (SLE, RA), malignancy, tuberculosis (most common infectious cause worldwide), post-cardiac surgery (post-pericardiotomy syndrome). Diagnosis requires 2 of 4: (1) sharp pleuritic chest pain (improved by sitting up and leaning forward), (2) pericardial friction rub, (3) diffuse ST elevation with PR depression on EKG, (4) new or worsening pericardial effusion.
Treatment: NSAIDs (ibuprofen 600 mg q8h or ASA 650 mg q8h) × 1–2 weeks + colchicine 0.5 mg BID × 3 months (reduces recurrence by ~50% — COPE/CORE/CORP/ICAP trials). Corticosteroids are second-line only (increase recurrence risk). Avoid NSAIDs in post-MI pericarditis — use ASA instead.
Constrictive Pericarditis
Chronic pericardial inflammation leads to a thick, fibrotic, sometimes calcified pericardium that restricts diastolic filling. Causes: prior cardiac surgery (most common in developed countries), radiation, TB, idiopathic. Presents like right heart failure: JVD (with Kussmaul's sign — paradoxical rise in JVP with inspiration), hepatomegaly, ascites, edema, but no pulmonary edema. EKG may show low voltage. Echo/CT/MRI shows pericardial thickening (>4 mm). Catheterization reveals equalization of diastolic pressures across all four chambers and the "square root" sign (rapid early diastolic filling followed by plateau). Treatment: pericardiectomy (surgical stripping of the pericardium).
24 Cardiac Tamponade Peri/Myo
Tamponade occurs when pericardial fluid accumulates fast enough to compress cardiac chambers, impairing diastolic filling and reducing cardiac output. Even small effusions (100–200 mL) can cause tamponade if they accumulate rapidly (e.g., hemorrhagic post-PCI or post-cardiac surgery). Slow-accumulating effusions (e.g., malignant, uremic) may reach >1 L before causing tamponade because the pericardium stretches over time.
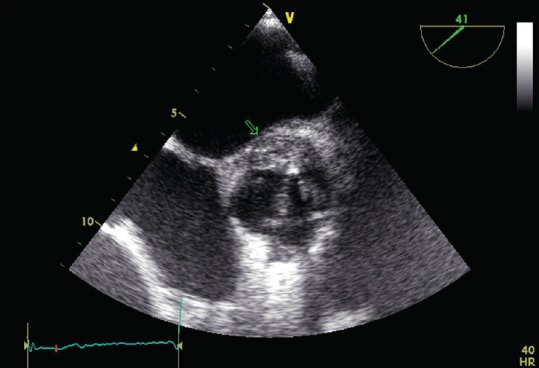
Beck's Triad
Hypotension + muffled heart sounds + JVD. Also: pulsus paradoxus (>10 mmHg drop in SBP during inspiration — exaggerated ventricular interdependence). Echo shows RA collapse in late diastole, RV collapse in early diastole, IVC plethora (>2.1 cm without respiratory variation), and respiratory variation of mitral/tricuspid inflow velocities.
Treatment
Emergent pericardiocentesis (subxiphoid approach, echo-guided). IV fluid resuscitation to maintain preload. Avoid positive pressure ventilation (reduces venous return). For recurrent effusions: pericardial window (surgical or percutaneous) or pericardiectomy. Treat underlying cause (malignancy, infection, autoimmune).
25 Myocarditis Peri/Myo
Inflammation of the myocardium, most commonly caused by viral infection (Coxsackievirus B, adenovirus, parvovirus B19, SARS-CoV-2, HHV-6). Also: autoimmune (giant cell myocarditis, eosinophilic myocarditis), drug-induced (immune checkpoint inhibitors — nivolumab, pembrolizumab — can cause fulminant myocarditis with mortality up to 40% if untreated), and toxic (cocaine, alcohol). Presentation ranges from subclinical to fulminant cardiogenic shock. Typical: young patient with recent viral prodrome presenting with chest pain (can mimic ACS), dyspnea, new HF symptoms, and arrhythmias.
Diagnosis
Elevated troponin, elevated inflammatory markers (CRP, ESR), EKG changes (diffuse ST elevation, PR depression — mimics pericarditis; or focal ST/T changes mimicking MI). Cardiac MRI is the noninvasive test of choice — shows myocardial edema (T2-weighted) and late gadolinium enhancement in a non-coronary distribution (mid-wall or epicardial, unlike ischemic patterns which are subendocardial). Endomyocardial biopsy (EMB) is the gold standard but reserved for cases of unexplained new-onset HF <2 weeks, hemodynamic compromise, or suspected giant cell myocarditis.
Management
Supportive: standard HF therapy (ACEi/ARB, beta-blocker once stable, diuretics), restrict exercise for 3–6 months. Immunosuppression for specific subtypes: IV pulse methylprednisolone for checkpoint inhibitor myocarditis (permanently discontinue the offending agent), corticosteroids + immunosuppressants for giant cell myocarditis. Mechanical circulatory support for fulminant cases. Most viral myocarditis resolves with supportive care, but ~10–20% progress to dilated cardiomyopathy.
26 Hypertrophic Cardiomyopathy (HCM) Peri/Myo
HCM is the most common inherited cardiac disorder (prevalence ~1:500), caused by mutations in sarcomere protein genes (most commonly beta-myosin heavy chain and myosin-binding protein C). Characterized by asymmetric septal hypertrophy (≥15 mm or ≥13 mm with family history) without another cause (e.g., HTN, AS). HCM is the most common cause of sudden cardiac death in young athletes.
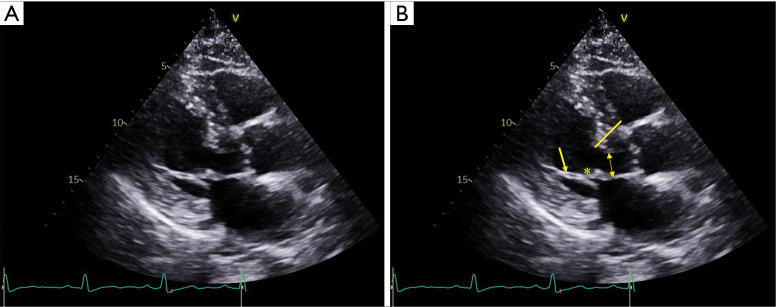
Pathophysiology
LVOT obstruction occurs in ~70% (obstructive HCM) — the thickened septum and systolic anterior motion (SAM) of the mitral valve create a dynamic gradient that worsens with decreased preload (dehydration, Valsalva, standing), decreased afterload, or increased contractility. This is why the murmur increases with Valsalva/standing and decreases with squatting/handgrip. Diastolic dysfunction from myocardial stiffness causes elevated filling pressures despite hyperdynamic systolic function. Myocardial fiber disarray creates a substrate for ventricular arrhythmias and SCD.
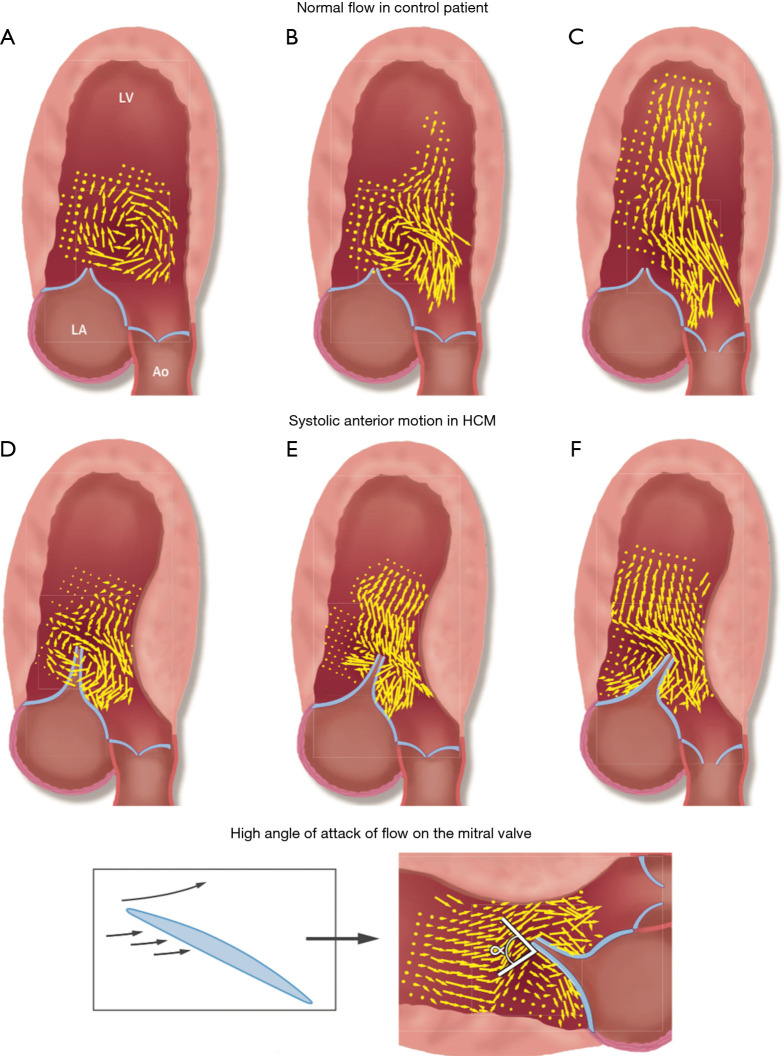
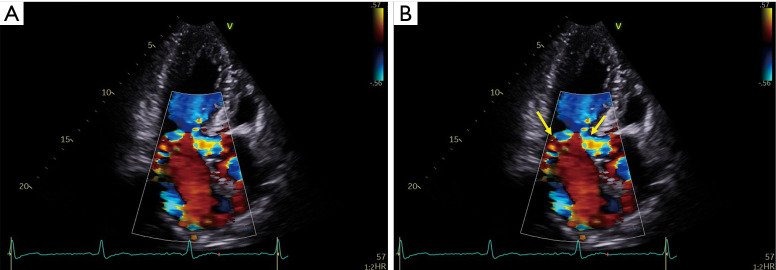
SCD Risk Stratification
Major risk factors: prior cardiac arrest or sustained VT, family history of SCD, unexplained syncope, massive LV hypertrophy (≥30 mm), NSVT on Holter, abnormal BP response to exercise. Patients with ≥1 major risk factor should be considered for ICD implantation.
Management
Non-obstructive HCM: Beta-blockers or verapamil for symptoms (dyspnea, angina). Obstructive HCM: Beta-blockers (first-line — reduce HR, prolong diastolic filling, reduce gradient), verapamil (alternative), disopyramide (Class IA antiarrhythmic with negative inotropic effect; added to beta-blocker for refractory obstruction). Mavacamten (Camzyos) — first-in-class cardiac myosin inhibitor, FDA-approved 2022 — directly reduces sarcomere contractility and LVOT gradient; 74% of patients achieved gradient <30 mmHg in the EXPLORER-HCM trial. Aficamten is a next-generation cardiac myosin inhibitor. Avoid: dihydropyridine CCBs (afterload reduction worsens obstruction), diuretics (reduce preload), digoxin (increases contractility), vasodilators, high-dose ACEi/ARBs. For refractory symptoms: septal myectomy (surgical — gold standard, Morrow procedure) or alcohol septal ablation (catheter-based; injects ethanol into septal perforator branch → controlled septal infarction → thins septum).
27 Dilated Cardiomyopathy Peri/Myo
Dilated cardiomyopathy (DCM) is characterized by LV (or biventricular) dilation with systolic dysfunction (EF <40%) not explained by coronary artery disease, valvular disease, or hypertension. It is the most common indication for heart transplantation. Causes: idiopathic (~50%), familial/genetic (~30% — TTN truncating variants most common), prior viral myocarditis, alcohol (typically >7–10 drinks/day for >5 years; partially reversible with abstinence), peripartum, tachycardia-mediated (prolonged rapid rates → reversible with rate control), chemotherapy-related (anthracyclines — dose-dependent, cumulative >400 mg/m² doxorubicin), and thyroid disease.
Management
Standard HFrEF GDMT (four pillars). Anticoagulation if LV thrombus, AF, or severely reduced EF with regional wall motion abnormalities. Genetic testing and family screening recommended (first-degree relatives with echo and EKG). Consider cardiac resynchronization therapy (CRT) if LBBB with QRS ≥150 ms and LVEF ≤35% despite GDMT (see Device Therapy section). Heart transplantation or LVAD for Stage D refractory HF.
28 Restrictive Cardiomyopathy Peri/Myo
Restrictive cardiomyopathy (RCM) is characterized by impaired ventricular filling due to stiff, non-compliant myocardium with preserved (or only mildly reduced) systolic function and normal or near-normal ventricular dimensions. The atria are typically severely dilated. Causes: amyloidosis (most common cause in developed countries — see dedicated section), sarcoidosis, hemochromatosis (iron deposition), radiation, eosinophilic myocarditis/Loeffler endocarditis, idiopathic. Hemodynamics mimic constrictive pericarditis (elevated filling pressures, diastolic HF) but pericardium is normal. Treatment targets the underlying cause; diuretics cautiously for congestion (overly aggressive diuresis drops preload and worsens output).
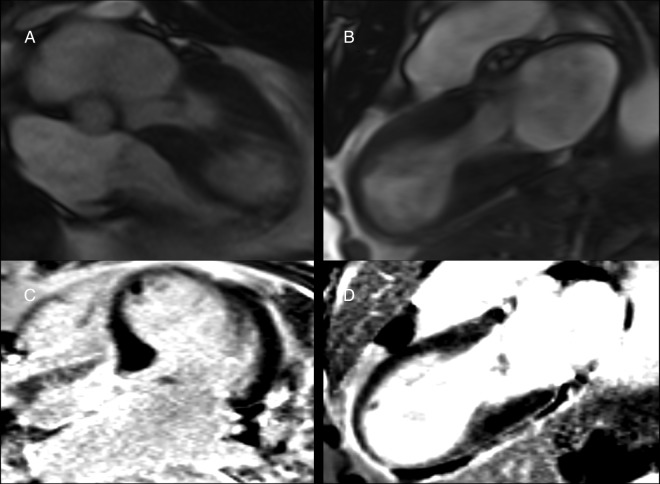
29 Takotsubo (Stress) Cardiomyopathy Peri/Myo
Transient LV dysfunction triggered by intense emotional or physical stress ("broken heart syndrome"). Classically occurs in postmenopausal women after a stressful event. Pathophysiology: catecholamine surge causes direct myocardial stunning. Classic pattern: apical ballooning with hyperkinetic base (the LV looks like a Japanese octopus pot — "takotsubo"). Presents identically to ACS: chest pain, troponin elevation, ST elevation or T-wave inversions on EKG. Coronary angiography shows no obstructive CAD (defining feature). Diagnosis uses the InterTAK criteria (emotional/physical trigger, characteristic wall motion, absence of obstructive CAD, new EKG changes, elevated troponin). Management: supportive care; avoid catecholamines/inotropes; beta-blockers may help. Most recover fully within 4–8 weeks, but acute complications include cardiogenic shock (~10%), LVOT obstruction, LV thrombus, and arrhythmias. In-hospital mortality is ~4–5%.
30 Hypertension Management Structural/Vasc
Hypertension affects ~1.3 billion adults globally and is the leading modifiable risk factor for cardiovascular death. The 2017 ACC/AHA Guideline redefined HTN categories:
| Category | SBP | DBP |
|---|---|---|
| Normal | <120 | <80 |
| Elevated | 120–129 | <80 |
| Stage 1 HTN | 130–139 | 80–89 |
| Stage 2 HTN | ≥140 | ≥90 |
| Hypertensive crisis | >180 | >120 |
Treatment Thresholds & Targets
Pharmacotherapy indicated at: Stage 1 with 10-year ASCVD risk ≥10% or with clinical CVD/DM/CKD (target <130/80); Stage 2 for all patients (target <130/80). The SPRINT trial demonstrated that intensive BP control (target SBP <120) reduced major CV events by 25% and all-cause mortality by 27% in high-risk patients without diabetes.
First-Line Drug Classes
| Class | Examples | Mechanism | Preferred When | Key Pearl |
|---|---|---|---|---|
| ACEi | Lisinopril 10–40 mg, enalapril, ramipril | Blocks ACE → reduces angiotensin II | DM, CKD (proteinuria), HFrEF, post-MI | Dry cough (~10%); angioedema risk; check K+/Cr at 1–2 weeks |
| ARB | Losartan 50–100 mg, valsartan, olmesartan | Blocks AT1 receptor | ACEi-intolerant (cough); same indications | No cough; do not combine with ACEi |
| CCB (DHP) | Amlodipine 5–10 mg, nifedipine ER | Blocks L-type Ca++ channels → vasodilation | Black patients, elderly, CAD, Raynaud's | Peripheral edema common; not first-line in HFrEF |
| Thiazide diuretic | Chlorthalidone 12.5–25 mg, HCTZ 25 mg, indapamide | Inhibits Na-Cl cotransporter in DCT | Black patients, elderly, osteoporosis, kidney stones | Chlorthalidone preferred (longer-acting, better outcome data); monitor Na+, K+, glucose, uric acid |
Hypertensive emergency: SBP >180 and/or DBP >120 WITH acute target organ damage (encephalopathy, ICH, aortic dissection, acute MI, pulmonary edema, eclampsia, acute renal failure). Requires ICU, IV antihypertensives (nicardipine, clevidipine, labetalol, nitroprusside), goal: reduce MAP by ~25% in the first hour. Hypertensive urgency: SBP >180 and/or DBP >120 WITHOUT organ damage. Oral agents, reduce over 24–48 hours. Do not drop BP too rapidly (risk of watershed stroke).
Resistant Hypertension
Defined as BP above goal despite 3 optimally dosed antihypertensives (including a diuretic) or requiring ≥4 drugs. Rule out pseudo-resistance (non-adherence, white-coat effect, improper technique). Screen for secondary causes: primary aldosteronism (aldosterone:renin ratio ≥30 with aldosterone ≥15), renal artery stenosis, pheochromocytoma (24-hour urine catecholamines/metanephrines), Cushing syndrome, OSA, thyroid disease, coarctation of aorta. Add spironolactone 25–50 mg as fourth agent (per PATHWAY-2 trial).
31 Pulmonary Hypertension Structural/Vasc
Pulmonary hypertension (PH) is defined hemodynamically as mean pulmonary artery pressure (mPAP) >20 mmHg at rest by right heart catheterization. The WHO classifies PH into 5 groups:
| Group | Category | Common Causes | Treatment Approach |
|---|---|---|---|
| 1 | Pulmonary arterial hypertension (PAH) | Idiopathic, heritable (BMPR2 mutation), CTD-associated (scleroderma), HIV, portal HTN, drugs | PAH-specific vasodilators (PDE5i, ERA, prostacyclin pathway) |
| 2 | PH due to left heart disease | HFrEF, HFpEF, valvular disease (most common cause of PH overall) | Treat underlying left heart disease; PAH drugs generally NOT indicated |
| 3 | PH due to lung disease/hypoxia | COPD, ILD, OSA | Treat underlying lung disease; supplemental O₂; consider PH drugs in severe cases |
| 4 | Chronic thromboembolic PH (CTEPH) | Unresolved PE organizing into chronic obstruction | Pulmonary thromboendarterectomy (PTE — potentially curative); balloon pulmonary angioplasty; riociguat |
| 5 | Multifactorial / unclear mechanisms | Sarcoidosis, hematologic disorders, metabolic | Treat underlying cause |
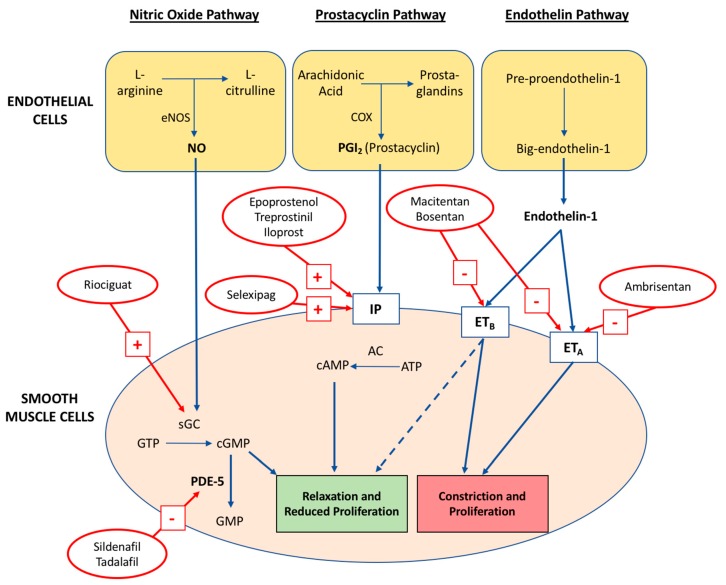
Group 1 PAH Medications
| Pathway | Drug | Route | Pearl |
|---|---|---|---|
| Nitric oxide / cGMP (PDE5 inhibitors) | Sildenafil (Revatio) 20 mg TID; tadalafil (Adcirca) 40 mg daily | Oral | Contraindicated with nitrates (severe hypotension) |
| Nitric oxide / cGMP (sGC stimulator) | Riociguat (Adempas) 0.5–2.5 mg TID | Oral | Only PH drug approved for both PAH and CTEPH; do NOT combine with PDE5i |
| Endothelin pathway (ERA) | Ambrisentan (Letairis) 5–10 mg; bosentan (Tracleer) 62.5–125 mg BID; macitentan (Opsumit) 10 mg | Oral | Hepatotoxicity (bosentan — monthly LFTs); teratogenic (REMS program) |
| Prostacyclin pathway | Epoprostenol (Flolan) IV continuous; treprostinil (Remodulin) IV/SQ/inhaled; iloprost (Ventavis) inhaled; selexipag (Uptravi) oral | Various | Epoprostenol: most potent but requires continuous IV infusion with no interruption (abrupt stop → rebound PH/death); half-life 3–5 minutes |
32 Aortic Disease (Cardiology Perspective) Structural/Vasc
Aortic Dissection
Tear in the aortic intima allowing blood to enter the media, creating a false lumen. Stanford Type A = involves the ascending aorta (regardless of extent) — surgical emergency (mortality increases ~1–2% per hour without surgery). Stanford Type B = descending aorta only (distal to left subclavian) — managed medically unless complicated (malperfusion, rupture, refractory pain, rapid expansion). Risk factors: HTN (most common), Marfan syndrome, bicuspid aortic valve, Ehlers-Danlos type IV, Loeys-Dietz, cocaine use, prior aortic surgery.
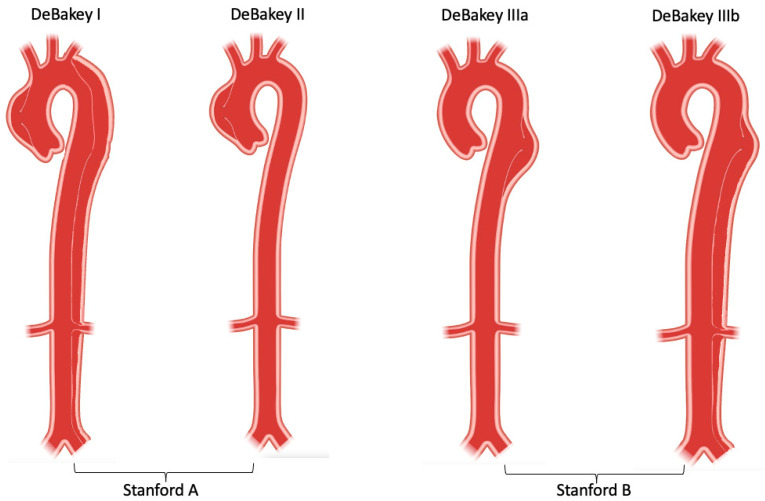
Presentation: tearing/ripping chest pain radiating to the back (classically "between the shoulder blades"), sudden onset, maximal at onset. BP differential >20 mmHg between arms, pulse deficits, aortic regurgitation murmur (Type A), stroke symptoms (carotid involvement), mesenteric ischemia, limb ischemia. Diagnosis: CTA chest/abdomen/pelvis (sensitivity >95%); TEE in unstable patients who cannot leave bedside.
Acute management: IV esmolol or labetalol — goal HR <60 and SBP 100–120 mmHg (reduce aortic wall shear stress). Add IV nicardipine or nitroprusside if BP not controlled with beta-blocker alone. Always start beta-blocker before vasodilators (vasodilator-induced reflex tachycardia increases shear stress). Type A → emergent surgical repair (ascending aorta replacement ± aortic valve ± arch). Complicated Type B → TEVAR (thoracic endovascular aortic repair).
Thoracic Aortic Aneurysm (TAA)
Ascending aorta aneurysm threshold for intervention: ≥5.5 cm in sporadic disease, ≥5.0 cm in bicuspid aortic valve, ≥4.5–5.0 cm in Marfan syndrome. Growth rate ≥0.5 cm/year is also an indication. Surveillance with serial imaging (CT or MRI) every 6–12 months for aneurysms approaching repair threshold.
33 Adult Congenital Heart Disease Structural/Vasc
The ACHD population is growing rapidly as surgical advances allow >90% of children with CHD to survive into adulthood. Common lesions encountered in adult cardiology practice:
| Lesion | Pathophysiology | Presentation in Adults | Management |
|---|---|---|---|
| Atrial Septal Defect (ASD) | Left-to-right shunt at atrial level; RV volume overload | Dyspnea, AF, paradoxical embolism (stroke), fixed splitting of S2; pulmonary overcirculation → eventual PH | Closure if Qp:Qs ≥1.5 and no irreversible PH; percutaneous device closure (Amplatzer) for secundum ASD; surgical for primum/sinus venosus |
| Patent Foramen Ovale (PFO) | Persistent flap-like opening between atria (present in ~25% of population) | Usually incidental; associated with cryptogenic stroke (paradoxical embolism) and migraine with aura | PFO closure (Amplatzer PFO Occluder, GORE CARDIOFORM) recommended for cryptogenic stroke in patients 18–60 per RESPECT/CLOSE/DEFENSE trials |
| Ventricular Septal Defect (VSD) | Left-to-right shunt at ventricular level | Small: asymptomatic with holosystolic murmur at LLSB. Large: HF, PH, Eisenmenger syndrome | Close if symptomatic or Qp:Qs ≥2.0; small restrictive VSDs may be observed; percutaneous closure for muscular VSDs |
| Coarctation of the Aorta | Discrete narrowing of the aorta, typically just distal to left subclavian origin (juxtaductal) | Upper extremity HTN, weak femoral pulses, rib notching on CXR (collateral intercostals), BP differential upper > lower extremities | Balloon angioplasty ± stenting or surgical repair; lifelong surveillance for recoarctation, associated BAV, and ascending aortic aneurysm |
| Tetralogy of Fallot (repaired) | Most common cyanotic CHD; repaired in infancy | Residual pulmonary regurgitation (most common late problem), RV dilation, VT risk | Pulmonary valve replacement when RV dilates significantly (RVEDV >150 mL/m²); ICD for sustained VT; lifelong follow-up |
34 Cardiac Tumors Special
Primary cardiac tumors are rare (~0.02% of autopsies). ~75% are benign, ~25% malignant. Metastatic tumors to the heart are 20–40× more common than primary tumors (lung, breast, melanoma, lymphoma). Myxoma is the most common primary cardiac tumor (~50%). Typically originates from the interatrial septum in the LA (~75%), pedunculated, gelatinous. Presents with the classic triad: obstruction (mimics mitral stenosis — dyspnea, positional symptoms, "tumor plop"), embolization (stroke, peripheral emboli), and constitutional symptoms (fever, weight loss, elevated ESR — produces IL-6). Diagnosis: echocardiography. Treatment: surgical excision (curative; ~3% recurrence, higher in Carney complex). Papillary fibroelastoma is the second most common benign tumor, typically found on the aortic valve, risk of embolism. Malignant: angiosarcoma (most common primary malignant cardiac tumor, typically RA, poor prognosis).
35 Cardiac Amyloidosis Special
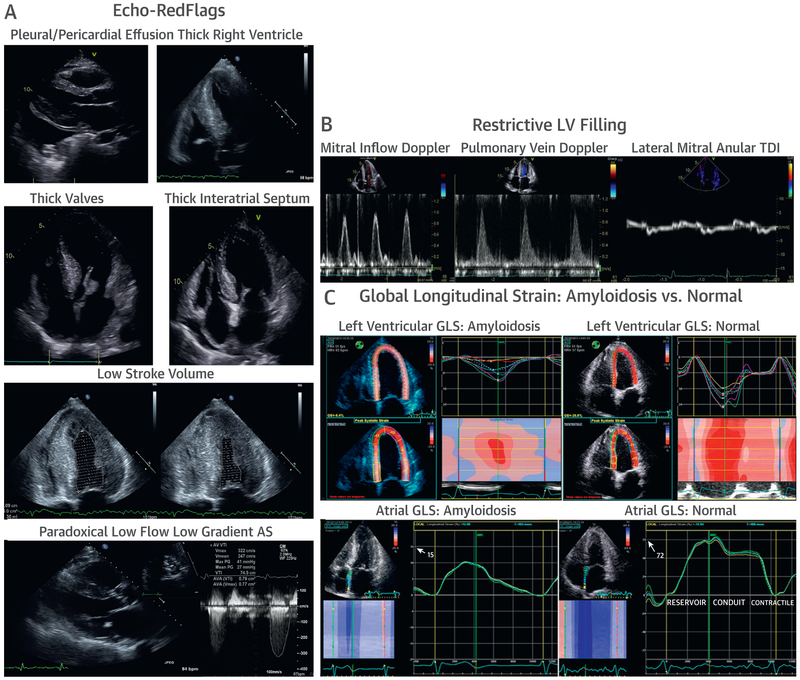
Amyloid deposition in the myocardium causes progressive restrictive cardiomyopathy with diastolic (and eventually systolic) heart failure. Two main types: AL amyloidosis (light-chain — from plasma cell dyscrasia/myeloma; rapidly progressive, median survival ~6 months without treatment) and ATTR amyloidosis (transthyretin — either hereditary/hATTR from TTR gene mutation, or wild-type/wtATTR, formerly "senile cardiac amyloidosis," increasingly recognized in elderly men with HFpEF). wtATTR is found in up to 13% of elderly patients hospitalized with HFpEF.
Diagnosis
Suspect when: HF with wall thickening on echo but low voltage on EKG (amyloid infiltration thickens walls but is electrically inert), granular "sparkling" myocardial texture, diastolic dysfunction disproportionate to hypertrophy, elevated BNP/troponin. Cardiac MRI shows diffuse subendocardial or transmural late gadolinium enhancement with abnormal nulling kinetics. Technetium pyrophosphate (Tc-PYP) scan is the game-changer for ATTR — Grade 2–3 uptake in the absence of a monoclonal protein (by serum free light chains and SPEP/UPEP/immunofixation) confirms ATTR amyloidosis without the need for biopsy.
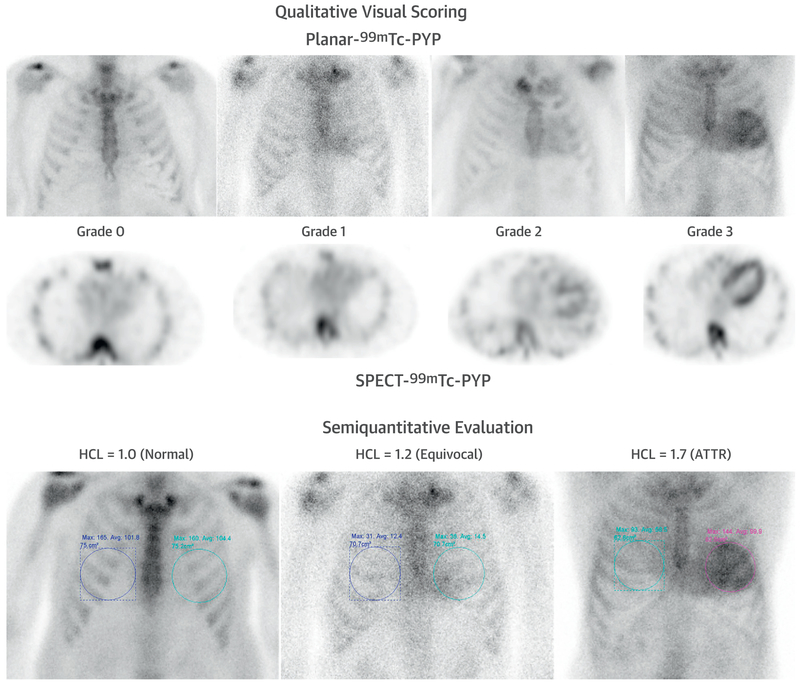
Treatment
AL amyloidosis: Chemotherapy targeting the plasma cell clone (bortezomib-based regimens; daratumumab-VCd per ANDROMEDA trial). ATTR amyloidosis: Tafamidis (Vyndamax/Vyndaqel) — TTR stabilizer, FDA-approved; reduced mortality by 30% and hospitalizations by 32% in the ATTR-ACT trial. TTR gene silencers: patisiran (Onpattro, siRNA), inotersen (Tegsedi, ASO) — primarily for hATTR with polyneuropathy; cardiac benefit emerging. Supportive: Diuretics cautiously; digoxin is generally avoided (binds to amyloid fibrils, increasing toxicity risk). ACEi/ARB/ARNI often poorly tolerated (hypotension).
36 Cardiac Sarcoidosis Special
Sarcoidosis involves the heart in ~25% of patients with systemic disease (often subclinical). Granulomatous infiltration of the myocardium causes conduction disease (AV block is the most common cardiac manifestation), ventricular arrhythmias (VT, SCD risk), and cardiomyopathy (regional wall motion abnormalities that do not follow coronary territories). Suspect cardiac sarcoidosis in young patients with unexplained high-degree AV block, VT, or non-ischemic cardiomyopathy — especially if pulmonary/systemic sarcoidosis is known.
Diagnosis
Cardiac MRI (patchy mid-wall or epicardial late gadolinium enhancement, often in the basal septum) and FDG-PET (focal myocardial uptake after prolonged fasting/high-fat diet to suppress normal myocardial glucose uptake — the PET protocol requires 12–18 hours of fasting or a high-fat/low-carb prep). EMB has low sensitivity (~20%) due to patchy involvement but is specific when positive.
Treatment
Immunosuppression: corticosteroids (prednisone 0.5–1 mg/kg) are first-line; methotrexate or mycophenolate as steroid-sparing agents. Pacemaker for high-grade AV block. ICD for sustained VT, EF ≤35%, or significant LGE on MRI with other risk factors. Catheter ablation for refractory VT. Serial FDG-PET to monitor treatment response.
37 Peripartum Cardiomyopathy Special
New-onset systolic HF (EF <45%) developing in the last month of pregnancy through 5 months postpartum, with no other identifiable cause. Incidence ~1:1,000 to 1:4,000 deliveries; higher in Black women, multiparity, preeclampsia, multiple gestations. Pathophysiology involves a cleaved prolactin fragment (16-kDa prolactin) with antiangiogenic and cardiotoxic properties. Presentation identical to HF: dyspnea, orthopnea, edema, fatigue. Treatment: standard HF therapy with pregnancy-specific modifications — ACEi/ARB/ARNI are teratogenic and contraindicated during pregnancy (use hydralazine/nitrates instead); beta-blockers safe; diuretics cautiously. Bromocriptine (dopamine agonist that inhibits prolactin secretion) may be beneficial (small trials). ~50% recover EF within 6 months; ~25% develop chronic severe cardiomyopathy. Subsequent pregnancies carry recurrence risk, especially if EF has not fully recovered.
38 Athlete's Heart vs Pathologic Hypertrophy Special
Chronic exercise produces physiologic cardiac remodeling — "athlete's heart" — that can mimic pathologic hypertrophy (HCM, DCM). Key distinctions:
| Feature | Athlete's Heart | HCM |
|---|---|---|
| Wall thickness | Usually ≤12 mm (gray zone 13–15 mm) | Usually ≥15 mm, asymmetric |
| LV cavity size | Dilated (LVEDD >54 mm) | Normal or small |
| Diastolic function | Normal or supranormal (E/e' normal) | Impaired (reduced e', elevated E/e') |
| Response to detraining | LVH regresses after 3 months of detraining | Does not regress |
| Family history | Negative | Often positive |
| EKG | Sinus bradycardia, early repolarization, LVH voltage criteria | Deep T-wave inversions, pathologic Q waves, LA enlargement |
| Cardiac MRI | No fibrosis (no LGE) | LGE at RV insertion points or within hypertrophied segments |
The "gray zone" (wall thickness 13–15 mm) requires careful multimodal assessment with clinical history, family screening, genetic testing, cardiac MRI with LGE, and sometimes detraining trials. Misdiagnosis in either direction has serious consequences: restricting a healthy athlete from competition, or clearing an athlete with HCM to participate (SCD risk).
39 Cardiac Catheterization & PCI Procedures
Diagnostic Catheterization
Invasive coronary angiography remains the gold standard for defining coronary anatomy. Access: radial artery (preferred — lower bleeding, lower access-site complications, earlier ambulation) or femoral artery (larger sheath capacity, used for complex interventions). Catheters advanced to coronary ostia under fluoroscopy; iodinated contrast injected to visualize coronary anatomy. Left ventriculography assesses LV function and MR. Right heart catheterization (Swan-Ganz catheter) measures RA, RV, PA, and PCWP pressures, CO by thermodilution or Fick method, calculates SVR/PVR, and assesses shunt presence (oxygen saturation step-up).
Percutaneous Coronary Intervention (PCI)
Balloon angioplasty + stent deployment to treat coronary stenosis. Modern PCI uses drug-eluting stents (DES) almost exclusively — coated with antiproliferative agents (everolimus, zotarolimus) to inhibit neointimal hyperplasia and reduce restenosis. Bare metal stents (BMS) are now rarely used. Lesion assessment adjuncts: fractional flow reserve (FFR) — pressure wire measures pressure drop across stenosis during hyperemia; FFR ≤0.80 = hemodynamically significant, benefits from PCI (FAME trial). Instantaneous wave-free ratio (iFR) — resting pressure assessment without adenosine; iFR ≤0.89 correlates with FFR ≤0.80. IVUS and OCT provide intravascular imaging to optimize stent deployment, sizing, and apposition.
Stent Types & DAPT Duration
| Stent Type | Drug Coating | Minimum DAPT | Notes |
|---|---|---|---|
| DES (current generation) | Everolimus (Xience, Synergy), Zotarolimus (Resolute Onyx) | 6 months (stable CAD); 12 months (ACS) | Can shorten to 1–3 months if high bleeding risk (per MASTER DAPT, STOPDAPT-2) |
| BMS | None | 1 month minimum | Rarely used; considered if impending surgery or high bleeding risk |
| Bioresorbable scaffold (BRS) | Everolimus (Absorb — withdrawn) | 12 months | Largely abandoned due to increased scaffold thrombosis; research ongoing |
Coronary Artery Bypass Grafting (CABG)
Preferred over PCI for: left main disease, three-vessel disease (especially with DM — per FREEDOM trial), SYNTAX score >32 (high anatomic complexity). Conduits: LIMA-to-LAD (left internal mammary artery — gold standard graft, 90%+ patency at 10 years); saphenous vein grafts (SVG) for other territories (50–60% patency at 10 years); radial artery (better patency than SVG, increasingly used as second arterial graft). Complete arterial revascularization (bilateral IMA + radial) is associated with superior long-term outcomes.
40 Electrophysiology Studies & Ablation Procedures
EP study: Diagnostic procedure using multipolar catheters placed in the heart (via femoral/jugular vein access) to map electrical activity, measure conduction intervals (AH and HV intervals), and induce arrhythmias with programmed stimulation to characterize their mechanism and identify ablation targets.
Catheter Ablation
| Arrhythmia | Ablation Target | Success Rate | Key Details |
|---|---|---|---|
| AVNRT | Slow pathway (near the coronary sinus ostium) | >95% | ~0.5–1% risk of complete heart block requiring pacemaker |
| WPW / AVRT | Accessory pathway | >95% | Left-sided pathways accessed via transseptal or retrograde aortic approach |
| Atrial flutter (typical) | Cavotricuspid isthmus (CTI) | >95% | One of the most straightforward ablations; bidirectional block confirmed |
| Atrial fibrillation | Pulmonary vein isolation (PVI) | 70–80% (single procedure, paroxysmal AF); 50–70% (persistent AF) | Pulsed field ablation (PFA) is newest energy source — tissue-selective (spares esophagus, phrenic nerve); cryoballoon and radiofrequency are established |
| Ventricular tachycardia | Scar border zone / re-entry circuit | 60–80% | 3D electroanatomic mapping (CARTO, EnSite); substrate ablation in ischemic VT; epicardial access sometimes needed |
41 Device Therapy (Pacemakers, ICDs, CRT) Procedures
Pacemaker Indications
Class I indications: symptomatic sinus node dysfunction, symptomatic second-degree Mobitz II AV block, third-degree (complete) AV block, alternating bundle branch block. Leads are placed transvenously (subclavian or axillary vein approach) into the RA appendage and/or RV apex/septum; generator implanted in a subcutaneous pocket below the clavicle.
Pacing Modes
| Mode | Chambers Paced/Sensed | Common Use |
|---|---|---|
| AAI | Atrium paced/sensed | Sinus node dysfunction with intact AV conduction (rare now) |
| VVI | Ventricle paced/sensed | Chronic AF with slow ventricular rate; backup pacing |
| DDD | Dual (atrium + ventricle) paced/sensed | Most common mode; maintains AV synchrony; used for AV block |
| CRT (BiV) | RA + RV + LV (via coronary sinus lead) | HFrEF with LBBB and QRS ≥150 ms (see below) |
ICD (Implantable Cardioverter-Defibrillator)
Detects and terminates VT/VF via anti-tachycardia pacing (ATP) or shock. Secondary prevention: survivors of VT/VF arrest or hemodynamically significant sustained VT. Primary prevention: LVEF ≤35% despite ≥3 months of GDMT, ≥40 days post-MI (if ischemic), NYHA II–III, and expected survival >1 year (based on MADIT-II, SCD-HeFT trials). Subcutaneous ICD (S-ICD) is an alternative that avoids transvenous leads (lower infection/lead complications, no pacing capability).
Cardiac Resynchronization Therapy (CRT)
Biventricular pacing coordinates LV and RV contraction in patients with dyssynchrony (LBBB). Class I indication: LVEF ≤35%, LBBB with QRS ≥150 ms, NYHA II–IV, on optimal GDMT. CRT reduces mortality by ~35% and HF hospitalizations by ~40%. CRT-D combines CRT with defibrillator. Conduction system pacing (CSP) — His bundle pacing or left bundle branch area pacing (LBBAP) — is emerging as a physiologic alternative to traditional CRT.
Leadless Pacemakers
The Micra (Medtronic) is a self-contained leadless pacemaker implanted directly into the RV via femoral venous access. Eliminates lead-related complications (lead fracture, infection, tricuspid regurgitation). Currently available for VVI and VDD modes. The AVEIR DR system uses two leadless devices (one atrial, one ventricular) communicating wirelessly for dual-chamber pacing.
42 Structural Interventions (TAVR, MitraClip, WATCHMAN) Procedures
TAVR (Transcatheter Aortic Valve Replacement)
Catheter-delivered bioprosthetic valve deployed within the diseased native aortic valve. Access: transfemoral (preferred, >90% of cases), transapical, transaortic, subclavian, or transcaval. Valve types: balloon-expandable (Edwards SAPIEN 3, SAPIEN 3 Ultra) — delivered on a balloon catheter, precise deployment; self-expanding (Medtronic Evolut R/PRO/FX) — nitinol frame self-deploys, partially repositionable. Pre-procedure planning: CT aortic annulus sizing (critical for valve selection), iliofemoral access assessment, coronary height assessment (risk of coronary obstruction). Complications: stroke (~2%), new pacemaker (~10–20% for self-expanding, ~5–10% for balloon-expandable), paravalvular leak, vascular access complications, coronary obstruction (rare but life-threatening).
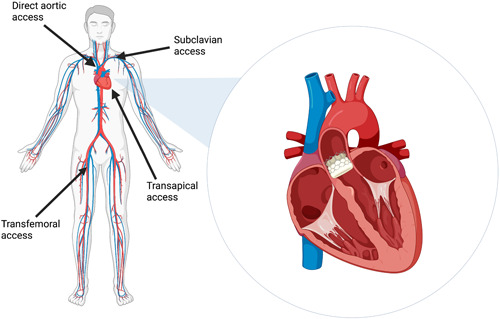
MitraClip / Edge-to-Edge Repair
The MitraClip (Abbott) and PASCAL (Edwards) devices perform transcatheter edge-to-edge repair of the mitral valve. Via transseptal puncture (femoral vein → RA → across septum into LA), a clip is delivered to grasp the anterior and posterior mitral leaflets, creating a double-orifice valve and reducing MR. Indicated for primary MR (high surgical risk, per EVEREST II) and secondary/functional MR on maximized GDMT (per COAPT criteria: EROA ≥0.30, moderate-severe MR disproportionate to LV dilation, LVEDV <200 mL).
Left Atrial Appendage Closure (WATCHMAN)
The WATCHMAN FLX (Boston Scientific) and Amulet (Abbott) devices occlude the LA appendage (where >90% of AF-related thrombi form) to reduce stroke risk in AF patients with contraindications to long-term anticoagulation. Implanted via transseptal approach. Post-implant: DAPT × 6 weeks, then ASA + clopidogrel or ASA alone. TEE at 45 days to confirm device seal.
PFO Closure
The Amplatzer PFO Occluder (Abbott) and GORE CARDIOFORM Septal Occluder are used for percutaneous PFO closure in patients 18–60 with cryptogenic stroke attributed to paradoxical embolism. Delivered via femoral venous access, deployed across the interatrial septum. Post-procedure: DAPT × 1–6 months, then ASA alone × 5 years.
43 Cardioversion & Defibrillation Procedures
Synchronized cardioversion delivers a shock timed to the R wave (avoids the vulnerable T-wave period, which could trigger VF). Used for: AFib/flutter, SVT, and hemodynamically stable monomorphic VT. Energy: AFib typically 120–200J biphasic; AFL 50–100J; SVT 50–100J; stable VT 100J. Defibrillation is unsynchronized (asynchronous shock) for VF and pulseless VT — 120–200J biphasic.
Pre-Cardioversion for AF
If AF duration >48 hours or unknown, there is risk of LA thrombus that could embolize with restoration of organized atrial contraction. Two strategies: (1) therapeutic anticoagulation for ≥3 weeks before and ≥4 weeks after cardioversion, or (2) TEE to exclude LA/LAA thrombus → if no thrombus, proceed with cardioversion + anticoagulation × ≥4 weeks post. Long-term OAC decision is based on CHA₂DS₂-VASc score regardless of cardioversion success.
44 Imaging & Diagnostics
| Modality | What It Shows | Clinical Interpretation Notes |
|---|---|---|
| 12-Lead EKG | Cardiac rhythm, rate, intervals, axis, ST/T changes, hypertrophy, conduction | PR interval (normal 120–200 ms), QRS (normal <120 ms), QTc (normal <470 ms male, <480 ms female). ST elevation: STEMI, pericarditis, early repolarization, Brugada, LV aneurysm |
| TTE (transthoracic echo) | LV/RV size and function, EF, valvular structure/function, pericardium, chamber pressures, wall motion | First-line cardiac imaging; EF by biplane Simpson's method; diastolic function graded I–III; E/e' >14 = elevated filling pressures |
| TEE (transesophageal echo) | Superior valvular detail, LA appendage thrombus, endocarditis vegetations, prosthetic valves, aortic dissection | Requires sedation; preferred for IE evaluation, pre-cardioversion, intraoperative guidance, TAVR planning |
| Stress testing (exercise) | Functional capacity, exercise-induced ischemia, arrhythmias, BP response | ≥1 mm ST depression = positive; Duke Treadmill Score risk-stratifies; <6 METs = poor functional capacity |
| Nuclear perfusion (SPECT/PET) | Myocardial perfusion defects, viability | Fixed defect = scar/infarct; reversible defect = ischemia (viable, may benefit from revascularization). PET is gold standard for viability |
| CT coronary angiography (CCTA) | Coronary anatomy, plaque burden, calcium score, anomalous coronaries | Excellent NPV for ruling out CAD; CAC score: 0 = very low risk, >400 = very high risk. TAVR planning: aortic annulus sizing, access |
| Cardiac MRI | Myocardial tissue characterization (fibrosis, edema, infiltration, iron), volumes, EF, congenital anatomy | LGE patterns: subendocardial = ischemic; mid-wall = non-ischemic CMP, myocarditis; epicardial = pericarditis/myocarditis. T1/T2 mapping for diffuse disease |
| Right heart catheterization (Swan-Ganz) | RA, RV, PA, PCWP pressures; CO/CI; SVR/PVR; shunt assessment | Normal PCWP 6–12 mmHg; mPAP >20 = PH; CI <2.2 = low output. Gold standard for PH diagnosis and hemodynamic assessment |
| Ambulatory EKG monitoring | Arrhythmia detection over extended periods | Holter (24–48h), event monitor (2–4 weeks), Zio patch (up to 14 days), implantable loop recorder (ILR — up to 3 years, e.g., Reveal LINQ) |
| Coronary calcium score (CAC) | Coronary artery calcification burden | Agatston score: 0 = no calcified plaque; 1–99 = mild; 100–399 = moderate; ≥400 = severe. Helps reclassify borderline ASCVD risk |
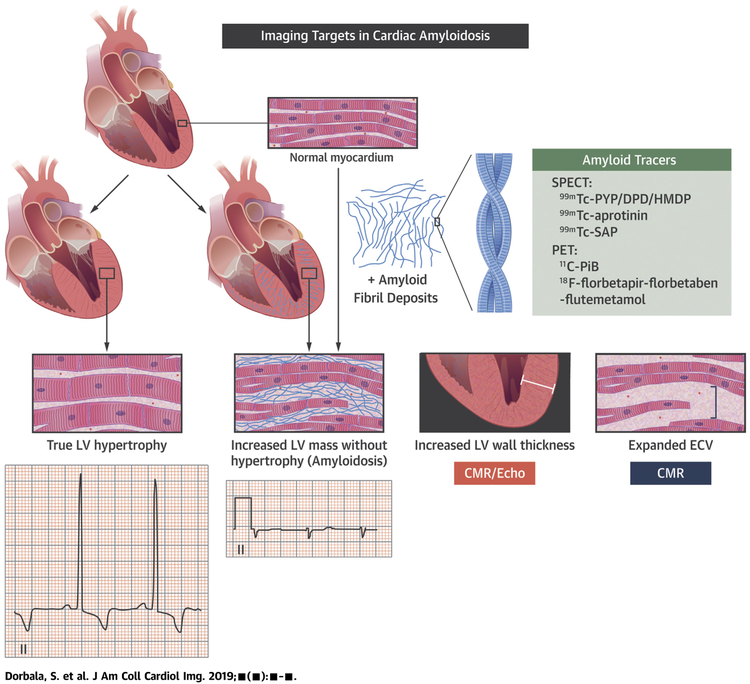
45 Classification Systems — All in One Place
| System | Classifies | Key Features |
|---|---|---|
| NYHA (I–IV) | HF functional class | I (no limitation) → IV (symptoms at rest); guides therapy and device decisions |
| ACC/AHA HF Stages (A–D) | HF disease progression | A (at risk) → D (advanced/refractory); stages only progress forward, never backward |
| Killip (I–IV) | ADHF in acute MI | I (no HF) → IV (cardiogenic shock); predicts in-hospital mortality |
| CCS Angina (I–IV) | Stable angina severity | I (strenuous) → IV (at rest); Canadian Cardiovascular Society scale |
| TIMI Score (0–7) | NSTEMI/UA risk | 7 variables; score ≥5 = high risk (~40% 14-day event rate) |
| GRACE Score | ACS mortality risk | More accurate than TIMI; validated for in-hospital and 6-month mortality |
| CHA₂DS₂-VASc (0–9) | Stroke risk in AF | Score ≥2 (men) or ≥3 (women) = OAC recommended |
| HAS-BLED (0–9) | Bleeding risk on anticoagulation | ≥3 = high risk; address modifiable factors, do not withhold OAC |
| EHRA Score (I–IV) | AF symptom severity | I (no symptoms) → IV (disabling symptoms); guides rhythm vs rate control |
| SCAI Shock (A–E) | Cardiogenic shock severity | A (at risk) → E (extremis); standardizes communication and escalation |
| SYNTAX Score | Coronary lesion complexity | ≤22 (low), 23–32 (intermediate), >32 (high); guides PCI vs CABG decision |
| Duke Criteria (Modified) | Infective endocarditis | Major + minor criteria; definite, possible, or rejected IE |
| Stanford (A/B) | Aortic dissection | A = ascending involved (surgical); B = descending only (medical unless complicated) |
| Carpentier (I–III) | Mitral regurgitation mechanism | I (annular dilation, normal motion); II (excessive motion/prolapse); III (restricted motion — IIIa rheumatic, IIIb functional) |
| Wilkins Score (4–16) | Mitral stenosis valve suitability for PMBC | Scores leaflet mobility, thickening, calcification, subvalvular disease (each 1–4); ≤8 = favorable for balloon commissurotomy |
| WHO PH Groups (1–5) | Pulmonary hypertension etiology | Group 1 = PAH; Group 2 = left heart; Group 3 = lung; Group 4 = CTEPH; Group 5 = multifactorial |
| Vaughan-Williams (I–IV) | Antiarrhythmic drug classification | I (Na+ channel); II (beta-blocker); III (K+ channel); IV (Ca++ channel) |
46 Medications Master Table Meds
Antiplatelet Agents
| Drug (Brand) | Mechanism | Dose | Indication | Critical Pearl |
|---|---|---|---|---|
| Aspirin | Irreversible COX-1 inhibitor | 81 mg daily (chronic); 325 mg loading | All ACS, post-PCI, chronic CAD, stroke prevention | Continue perioperatively in most cardiac patients |
| Clopidogrel (Plavix) | Irreversible P2Y12 blocker (prodrug) | 75 mg daily; 300–600 mg load | DAPT post-stent, ACS, stroke/PAD | CYP2C19 polymorphism → poor metabolizers have reduced efficacy; consider genotype-guided therapy |
| Ticagrelor (Brilinta) | Reversible P2Y12 blocker (no prodrug) | 90 mg BID (ACS); 60 mg BID (long-term) | ACS, post-PCI; preferred over clopidogrel in ACS | Dyspnea side effect (~14%); not a prodrug — no CYP2C19 issue; hold 5 days pre-surgery |
| Prasugrel (Effient) | Irreversible P2Y12 blocker (prodrug) | 10 mg daily; 60 mg load | ACS going to PCI | Contraindicated in prior stroke/TIA; not recommended if ≥75 or <60 kg; hold 7 days pre-surgery |
| Cangrelor (Kengreal) | Reversible P2Y12 blocker (IV) | 30 mcg/kg bolus then 4 mcg/kg/min infusion | Bridge during PCI when oral P2Y12 not possible | Half-life 3–6 min; effect off within 1 hour |
| GP IIb/IIIa inhibitors (abciximab, eptifibatide, tirofiban) | Block platelet aggregation at final common pathway | Variable (IV infusion) | High-risk PCI, large thrombus burden | Use declining with potent oral P2Y12 agents; high bleeding risk |
Anticoagulants
| Drug (Brand) | Mechanism | Monitoring | Reversal | Key Pearl |
|---|---|---|---|---|
| Heparin (UFH) | Antithrombin III activator → anti-IIa + anti-Xa | aPTT or anti-Xa | Protamine sulfate (1 mg per 100 units) | Short half-life (~1 hr); adjustable; HIT risk (~1–3%) |
| Enoxaparin (Lovenox) | LMWH — primarily anti-Xa | Anti-Xa levels if renal impairment/obesity | Protamine (partial, ~60%) | Dose-adjust for CrCl <30 (1 mg/kg daily instead of BID) |
| Warfarin (Coumadin) | Vitamin K antagonist (factors II, VII, IX, X, protein C&S) | INR (target 2.0–3.0 or 2.5–3.5) | Vitamin K (slow), FFP (moderate), PCC/Kcentra (rapid) | Required for mechanical valves (DOACs contraindicated); numerous drug/food interactions |
| Apixaban (Eliquis) | Direct factor Xa inhibitor | None routine | Andexanet alfa (Andexxa) | Preferred DOAC in AF (lowest bleeding in ARISTOTLE); reduce to 2.5 mg BID if ≥2 of: age ≥80, weight ≤60 kg, Cr ≥1.5 |
| Rivaroxaban (Xarelto) | Direct factor Xa inhibitor | None routine | Andexanet alfa | Take with food (20 mg dose); once-daily dosing for AF; low-dose (2.5 mg BID) + ASA for stable CVD (COMPASS) |
| Dabigatran (Pradaxa) | Direct thrombin (IIa) inhibitor | None routine | Idarucizumab (Praxbind) | Only DOAC with specific complete reversal agent; avoid in CrCl <30; GI side effects common |
| Edoxaban (Savaysa) | Direct factor Xa inhibitor | None routine | Andexanet alfa (off-label) | Once daily; less effective at CrCl >95 (contraindicated in AF with high CrCl) |
Heart Failure Medications
| Drug (Brand) | Class | Dose Range | Indication | Critical Pearl |
|---|---|---|---|---|
| Sacubitril/valsartan (Entresto) | ARNI | 24/26 → 97/103 mg BID | HFrEF (EF ≤40%); HFmrEF | 36-hour washout from ACEi; do not combine with ACEi; can cause hypotension |
| Carvedilol (Coreg) | Beta-blocker (alpha+beta) | 3.125 → 25 mg BID | HFrEF | One of only 3 evidence-based HF beta-blockers; do not initiate during decompensation |
| Metoprolol succinate (Toprol-XL) | Beta-1 selective blocker | 12.5 → 200 mg daily | HFrEF | Must be succinate (XL), NOT tartrate (IR) for HF mortality benefit |
| Spironolactone (Aldactone) | MRA | 12.5–50 mg daily | HFrEF (EF ≤35%); resistant HTN | Monitor K+ and Cr; gynecomastia risk; eplerenone is alternative (fewer endocrine SE) |
| Dapagliflozin (Farxiga) | SGLT2 inhibitor | 10 mg daily | HFrEF, HFpEF (regardless of DM) | UTI/genital mycotic infections; hold before surgery; avoid in eGFR <20 |
| Empagliflozin (Jardiance) | SGLT2 inhibitor | 10 mg daily | HFrEF, HFpEF (regardless of DM) | Same class as dapagliflozin; benefits in DM with CVD (EMPA-REG OUTCOME) |
| Hydralazine/ISDN (BiDil) | Vasodilator combo | 37.5/20 → 75/40 mg TID | Black patients with HFrEF; ACEi/ARB intolerant | A-HeFT: 43% mortality reduction in Black patients |
| Ivabradine (Corlanor) | If channel inhibitor | 2.5–7.5 mg BID | HFrEF in NSR with HR ≥70 on max BB | Only works in sinus rhythm; does not reduce BP |
| Digoxin (Lanoxin) | Na+/K+ ATPase inhibitor | 0.125–0.25 mg daily | HF (reduces hospitalizations); AF rate control | Target level 0.5–0.9 ng/mL; toxicity: visual changes, GI, arrhythmias; reverse with DigiFab |
| Furosemide (Lasix) | Loop diuretic | 20–600+ mg daily (oral or IV) | Volume management in HF | No mortality benefit; essential for decongestion; 2:1 IV:PO conversion |
Antiarrhythmic Drugs (Vaughan-Williams Classification)
| Class | Drug (Brand) | Mechanism | Primary Use | Critical Pearl |
|---|---|---|---|---|
| IA | Procainamide; quinidine; disopyramide | Na+ channel block (intermediate kinetics) | WPW with AF (procainamide); AF (quinidine); HCM (disopyramide) | Procainamide: lupus-like syndrome with chronic use; QT prolongation |
| IB | Lidocaine; mexiletine | Na+ channel block (fast kinetics) | VT (lidocaine IV); VT adjunct (mexiletine PO) | Minimal effect on atrial tissue; CNS toxicity (seizures, confusion) |
| IC | Flecainide (Tambocor); propafenone (Rythmol) | Na+ channel block (slow kinetics) | AF/SVT in structurally normal hearts ("pill-in-the-pocket") | Contraindicated in structural heart disease and CAD (CAST trial: increased mortality) |
| II | Beta-blockers (metoprolol, atenolol, esmolol) | Beta-adrenergic blockade | Rate control (AF, SVT), VT suppression | Esmolol: ultra-short acting (half-life 9 min); ideal for acute titration |
| III | Amiodarone (Cordarone); sotalol (Betapace); dofetilide (Tikosyn); dronedarone (Multaq); ibutilide (Corvert) | K+ channel block (prolong repolarization) | AF, VT, various arrhythmias | Amiodarone: most effective but extensive toxicity. Sotalol: also has Class II effect. Dofetilide: must initiate inpatient (QTc monitoring). Ibutilide: IV for acute AF/AFL conversion (torsades risk ~4%) |
| IV | Verapamil; diltiazem | L-type Ca++ channel block | SVT, AF rate control | Avoid in HFrEF and WPW; negative inotropic and chronotropic |
Lipid-Lowering Agents
| Drug (Brand) | Mechanism | LDL Reduction | Key Pearl |
|---|---|---|---|
| Atorvastatin 40–80 mg (Lipitor) | HMG-CoA reductase inhibitor | ~50–60% | High-intensity statin; standard for ASCVD/ACS |
| Rosuvastatin 20–40 mg (Crestor) | HMG-CoA reductase inhibitor | ~50–63% | Most potent statin per mg; renally excreted |
| Ezetimibe 10 mg (Zetia) | NPC1L1 inhibitor (cholesterol absorption) | ~18–25% (add-on) | Added when statin alone insufficient; IMPROVE-IT showed benefit in ACS |
| Evolocumab (Repatha) | PCSK9 inhibitor (monoclonal Ab) | ~60% (add-on to statin) | SQ injection q2 weeks; FOURIER trial: 15% reduction in CV events |
| Alirocumab (Praluent) | PCSK9 inhibitor (monoclonal Ab) | ~50–60% (add-on) | SQ injection; ODYSSEY OUTCOMES: reduced MACE post-ACS |
| Inclisiran (Leqvio) | siRNA targeting PCSK9 | ~50% (add-on) | SQ injection q6 months after initial loading; given in-office |
| Bempedoic acid (Nexletol) | ACL inhibitor (upstream of HMG-CoA) | ~18% (add-on) | Oral; no myalgia (not active in muscle); CLEAR trial: 13% MACE reduction in statin-intolerant |
| Icosapent ethyl (Vascepa) | Purified EPA omega-3 | Reduces TG ~20% | REDUCE-IT: 25% relative reduction in CV events in patients with elevated TG on statin |
47 Abbreviations Master List
Anatomy & Physiology
Diagnoses
Procedures & Devices
Labs & Medications
48 Risk Factors & Comorbidities
Hypertension: Current meds, BP control, adherence. Target <130/80 for most cardiac patients. Uncontrolled HTN is the leading risk factor for HF, stroke, AF, and aortic disease.
Diabetes mellitus: Type, A1C, current regimen, microvascular complications (nephropathy, retinopathy, neuropathy). SGLT2 inhibitors and GLP-1 RAs have demonstrated cardiovascular benefit independent of glucose control. DM patients have 2–4× increased CV mortality.
Hyperlipidemia: LDL level, statin type/dose, statin intolerance history. Target LDL <70 mg/dL for established ASCVD; <55 considered for very high-risk patients on maximal therapy.
Smoking: Pack-years, current status, quit date. Active smoking doubles CV risk. Smoking cessation reduces MI risk by ~50% within 1 year. Offer pharmacotherapy (varenicline, NRT, bupropion) at every visit.
Obesity: BMI, waist circumference. Independent risk factor for AF, HFpEF, CAD, HTN. GLP-1 RAs (semaglutide) show CV benefit with weight loss. Bariatric surgery reduces long-term CV events in severe obesity.
CKD: eGFR, albuminuria. CKD stage ≥3 confers CV risk equivalent to prior MI. Affects DOAC dosing, contrast use, MRA safety, and SGLT2i initiation. Finerenone (non-steroidal MRA) reduces CV events in DM + CKD.
Obstructive sleep apnea: AHI, CPAP adherence. OSA is an independent risk factor for AF, HTN, pulmonary hypertension, and HFpEF. Screen all HF and AF patients.
Family history: Premature ASCVD (male <55, female <65 in first-degree relative). Family history of SCD — screen for HCM, channelopathies, ARVC.
Substance use: Cocaine (coronary vasospasm, acute MI, aortic dissection — treat with benzodiazepines + NTG, avoid beta-blockers). Alcohol excess (>14 drinks/week — AF trigger, alcoholic cardiomyopathy). Methamphetamine (cardiomyopathy, pulmonary hypertension).
References & Figure Sources
Figures
- Figures 1, 5, 10–12 — Heart Failure Pathophysiology. Schwinger RHG. Pathophysiology of heart failure. Cardiovasc Diagn Ther. 2021;11(1):263-276. PMC 7944197. Licensed under CC BY-NC-ND 4.0.
- Figures 2–3 — Coronary Artery Anatomy. Defined in relation to cardiac anatomy, from Banchi anatomical drawings. PMC 8200569. Licensed under CC BY 4.0.
- Figure 4 — Cardiac Conduction System. Park DS, Bhatt I, Bhatt DL. Basic science for clinicians: the cardiac conduction system. Circulation. 2011. PMC 3064561.
- Figures 6, 13 — ECG Patterns. Hammad M et al. Early detection of atrial fibrillation based on ECG signals. Bioengineering. 2020;7(1):16. PMC 7148541. Licensed under CC BY 4.0.
- Figures 7, 15–16 — Aortic Stenosis Assessment. Defined echocardiographic approach. PMC 8115410. Licensed under CC BY-NC 4.0.
- Figure 8 — STEMI ECG and Angiography. ECG localization in STEMI. PMC 8051330.
- Figures 9, 31 — HFrEF GDMT Strategy. Optimizing foundational therapies in HFrEF. PMC 9156437. Licensed under CC BY-NC-ND 4.0.
- Figure 14 — AV Block ECG. Heart block hat-trick case report. PMC 10284657. Licensed under CC BY 4.0.
- Figures 17–18 — Infective Endocarditis. Endocarditis echocardiographic imaging. PMC 7011492.
- Figure 19, 37 — Pericarditis ECG. ECG diagnosis: acute pericarditis. PMC 3854820.
- Figure 20 — Cardiac Tamponade. Cardiac tamponade case report. PMC 3628467.
- Figures 21–23 — Hypertrophic Cardiomyopathy. SAM in HCM: a narrative review. PMC 9264047. Licensed under CC BY-NC-ND 4.0.
- Figures 24–25 — Takotsubo Cardiomyopathy. Imaging features in takotsubo. PMC 3822220. Licensed under CC BY-NC 3.0.
- Figures 26–27 — Aortic Dissection. Current understanding of aortic dissection. PMC 9605578. Licensed under CC BY 4.0.
- Figure 28 — PAH Treatment Pathways. PAH pathophysiology and treatment. PMC 6023499. Licensed under CC BY 4.0.
- Figures 29–30, 35 — Cardiac Amyloidosis Imaging. How to image cardiac amyloidosis. PMC 7148180.
- Figures 32–33 — ICD and CRT-D Radiographs. Overview of ICD and CRT in HF. PMC 4958710.
- Figure 34 — TAVR Access Routes. TAVR: past, present, and future. PMC 10788655. Licensed under CC BY 4.0.
- Figure 36 — ICD vs Pacemaker. Evolution of pacemakers and ICDs. PMC 10620620. Licensed under CC BY 4.0.
Key Trials & Guidelines
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. Circulation. 2022;145(18):e895-e1032. PMID: 35363499
- Maron DJ, Hochman JS, Reynolds HR, et al. Initial invasive or conservative strategy for stable coronary disease (ISCHEMIA trial). N Engl J Med. 2020;382(15):1395-1407. PMID: 31475799
- Kirchhof P, Camm AJ, Goette A, et al. Early rhythm-control therapy in patients with atrial fibrillation (EAST-AFNET 4). N Engl J Med. 2020;383(14):1305-1316. PMID: 32865375
- Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter mitral-valve repair in patients with heart failure (COAPT trial). N Engl J Med. 2018;379(24):2307-2318. PMID: 30280640
- Maurer MS, Schwartz JH, Gundapaneni B, et al. Tafamidis treatment for patients with transthyretin amyloid cardiomyopathy (ATTR-ACT). N Engl J Med. 2018;379(11):1007-1016. PMID: 30145929
- Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation (RE-LY). N Engl J Med. 2009;361(12):1139-1151. PMID: 19717844
- De Backer D, Biston P, Devriendt J, et al. Comparison of dopamine and norepinephrine in the treatment of shock (SOAP II). N Engl J Med. 2010;362(9):779-789. PMID: 20200382
- SPRINT Research Group. A randomized trial of intensive versus standard blood-pressure control (SPRINT). N Engl J Med. 2015;373(22):2103-2116. PMID: 26551272
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. J Am Coll Cardiol. 2018;71(19):e127-e248. PMID: 29133356
- Tonelli AR, Arelli V, Minai OA, et al. Causes and circumstances of death in pulmonary arterial hypertension. Am J Respir Crit Care Med. 2013;188(3):365-369. PMID: 23600452
- Gulati M, Levy PD, Mukherjee D, et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain. Circulation. 2021;144(22):e368-e454. PMID: 34756653
- Farkouh ME, Domanski M, Sleeper LA, et al. Strategies for multivessel revascularization in patients with diabetes (FREEDOM trial). N Engl J Med. 2012;367(25):2375-2384. PMID: 30145934
- Imazio M, Brucato A, Cemin R, et al. Colchicine for recurrent pericarditis (CORP). Ann Intern Med. 2011;155(7):409-414. PMID: 24040484
Textbooks & Reference Works
- Braunwald's Heart Disease: A Textbook of Cardiovascular Medicine. 12th ed. Libby P, Bonow RO, Mann DL, et al., eds. Elsevier; 2022.
- Hurst's The Heart. 14th ed. Fuster V, Harrington RA, Narula J, et al., eds. McGraw-Hill; 2022.
- ACC/AHA Clinical Practice Guidelines. Available at: acc.org/guidelines.
- Gray's Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. Standring S, ed. Elsevier; 2020.