Dermatology
Every diagnosis, classification, procedure, medication, biopsy technique, staging system, and management strategy across the full scope of clinical dermatology in one place.
01 Skin Anatomy & Physiology
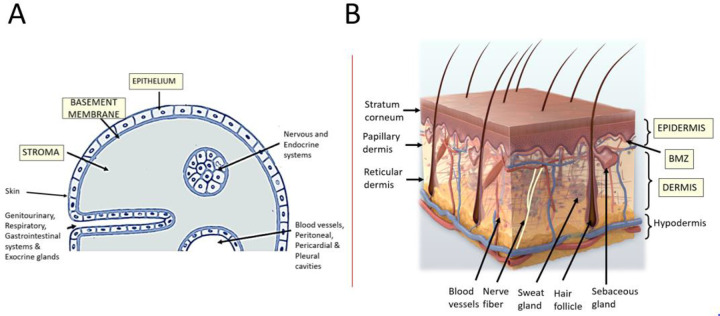
The skin is the largest organ in the body, weighing 3.5-10 kg in adults and covering approximately 1.5-2.0 m2 of surface area. It functions as a barrier against pathogens, UV radiation, chemical exposure, and mechanical injury; regulates temperature through vasodilation/vasoconstriction and eccrine sweat; synthesizes vitamin D3 via UV-B conversion of 7-dehydrocholesterol in the epidermis; and provides immune surveillance through resident Langerhans cells, dendritic cells, and dermal macrophages. Understanding its layered architecture is essential for interpreting biopsy reports, selecting surgical depth, and understanding disease pathogenesis.
Epidermis
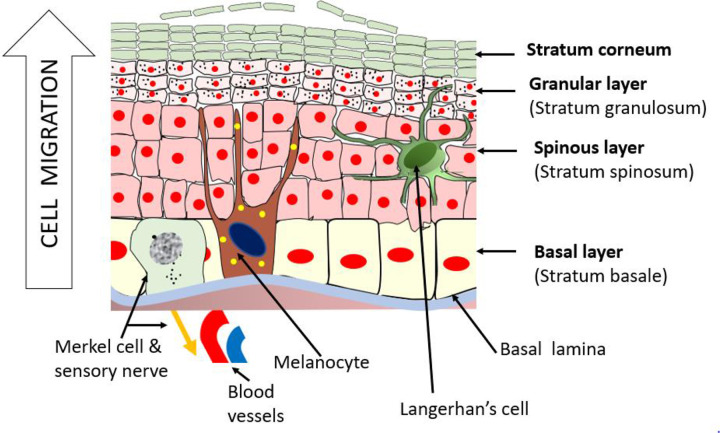
The epidermis is the outermost layer, avascular, and composed of stratified squamous epithelium. It ranges from 0.05 mm (eyelids) to 1.5 mm (palms and soles). It consists of five layers from deep to superficial:
| Layer | Key Features | Clinical Relevance |
|---|---|---|
| Stratum basale (basal layer) | Single row of columnar keratinocytes attached to basement membrane via hemidesmosomes; contains melanocytes (1 per ~36 keratinocytes) and Merkel cells (mechanoreceptors) | Site of origin for BCC; melanocytes produce melanin transferred to keratinocytes via melanosomes; target of autoimmune attack in vitiligo |
| Stratum spinosum (squamous/prickle cell layer) | Several layers of polyhedral keratinocytes connected by desmosomes; contains Langerhans cells (antigen-presenting cells from bone marrow) | Site of origin for SCC; desmosomes are the target in pemphigus vulgaris (anti-desmoglein 3 antibodies); "spiny" appearance is an artifact of fixation |
| Stratum granulosum (granular layer) | 3-5 layers of flattened cells containing keratohyalin granules (profilaggrin) and lamellar bodies that extrude lipid bilayers | The lipid bilayer forms the critical waterproof barrier; disrupted in atopic dermatitis (filaggrin mutations); site of target in pemphigus foliaceus (anti-desmoglein 1) |
| Stratum lucidum | Thin, clear layer; only present in thick skin (palms, soles) | Extra layer adds mechanical resistance to friction-heavy sites |
| Stratum corneum (horny layer) | 15-20 layers of anucleated, flattened, keratin-filled corneocytes embedded in lipid matrix ("bricks and mortar" model) | Primary physical barrier; abnormal in ichthyosis, xerosis; target of topical therapies — drug penetration depends on lipophilicity and molecular weight (< 500 Da ideal) |
Dermal-Epidermal Junction (DEJ)
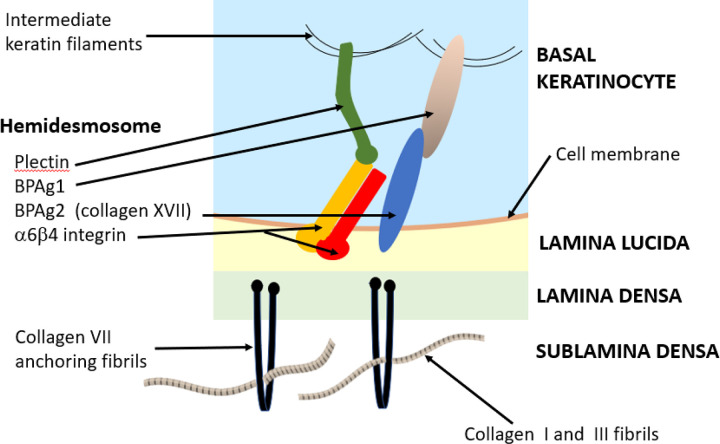
The basement membrane zone (BMZ) anchors the epidermis to the dermis. It consists of (superficial to deep): hemidesmosome attachment plaques in basal keratinocytes, the lamina lucida (containing laminin-332 and BP180/BP230 antigens — targets in bullous pemphigoid), the lamina densa (type IV collagen), and anchoring fibrils (type VII collagen — target in epidermolysis bullosa acquisita). Understanding the BMZ is critical because the level of the split determines the blister type: intraepidermal (pemphigus, flaccid blisters) vs subepidermal (bullous pemphigoid, tense blisters).
Dermis
The dermis lies deep to the epidermis, ranges from 0.3 mm (eyelids) to 3.0 mm (back), and is divided into two zones. The papillary dermis (superficial) consists of thin, loosely arranged type III collagen fibers, capillary loops that nourish the epidermis, and Meissner corpuscles (light touch). The reticular dermis (deep) contains thick, densely packed type I collagen bundles (80% of dermal collagen), elastic fibers (providing skin recoil — loss with aging produces wrinkles), and the cutaneous vascular plexus. The dermis also houses sensory nerve endings (Pacinian corpuscles for pressure/vibration, free nerve endings for pain/temperature), mast cells (central to urticaria and anaphylaxis), fibroblasts (collagen synthesis), and the skin's immune cell population.
Subcutaneous Tissue (Hypodermis)
The subcutis lies below the dermis and consists of lobules of adipocytes separated by fibrous septae containing blood vessels and nerves. It provides insulation, energy storage, mechanical cushioning, and anchors the skin to underlying fascia and muscle. Pathology here includes panniculitis (erythema nodosum involves the septae; erythema induratum involves the lobules), lipomas, and deep infections (necrotizing fasciitis tracks along fascial planes below this layer).
Skin Appendages
Eccrine glands: Distributed across the entire body (highest density on palms, soles, forehead); produce clear, odorless sweat for thermoregulation; innervated by sympathetic cholinergic fibers. Apocrine glands: Located in axillae, groin, areolae, periumbilical; become active at puberty; produce milky secretion that becomes malodorous when metabolized by skin bacteria. Sebaceous glands: Associated with hair follicles (the pilosebaceous unit); produce sebum (lipid mixture) under androgen regulation; hypertrophy leads to sebaceous hyperplasia; blockage leads to comedones and acne. Hair follicles: Cycle through anagen (active growth, 2-6 years on scalp), catagen (regression, 2-3 weeks), and telogen (rest, 2-3 months) phases. Approximately 85-90% of scalp hairs are in anagen at any time. The follicular bulge region contains stem cells critical for wound healing and hair regeneration — destruction of this region in cicatricial alopecia makes hair loss permanent.
02 Dermatologic Terminology — Lesion Morphology
Precise morphologic description is the foundation of dermatologic diagnosis. Every skin finding must be described using standard terminology for primary lesions (the initial morphology), secondary lesions (changes resulting from evolution, trauma, or manipulation), and configuration/distribution patterns. Imprecise descriptions ("rash," "skin lesion") are clinically useless.
Primary Lesions
| Term | Definition | Size | Classic Examples |
|---|---|---|---|
| Macule | Flat, non-palpable color change | ≤ 1 cm | Freckle (ephelid), petechiae, vitiligo patch edge |
| Patch | Flat, non-palpable color change | > 1 cm | Vitiligo, cafe-au-lait macule, tinea versicolor |
| Papule | Elevated, solid, palpable | ≤ 1 cm | Molluscum contagiosum, lichen planus, acne comedone |
| Plaque | Elevated, flat-topped, solid | > 1 cm | Psoriasis, mycosis fungoides, eczema |
| Nodule | Solid, palpable, extends into dermis/subcutis | > 1 cm (depth) | Cyst, lipoma, BCC nodular, erythema nodosum |
| Tumor | Large nodule | > 2 cm | Large BCC, SCC, melanoma, dermatofibrosarcoma |
| Vesicle | Fluid-filled blister | ≤ 1 cm | HSV, varicella, dyshidrotic eczema, contact dermatitis |
| Bulla | Fluid-filled blister | > 1 cm | Bullous pemphigoid, bullous impetigo, SJS/TEN |
| Pustule | Pus-filled lesion | Variable | Acne, folliculitis, pustular psoriasis, AGEP |
| Wheal (hive) | Edematous, transient, pruritic plaque | Variable | Urticaria (individual wheals last < 24 hrs) |
| Cyst | Encapsulated, fluid- or semi-solid-filled cavity | Variable | Epidermal inclusion cyst, pilar cyst |
| Comedo | Follicular plug of keratinized debris and sebum | 1-3 mm | Open (blackhead) and closed (whitehead) comedones in acne |
| Purpura | Non-blanching red-purple discoloration (extravasated RBCs) | Variable | Petechiae (< 3 mm), purpura (3 mm-1 cm), ecchymoses (> 1 cm) |
Secondary Lesions
| Term | Definition | Clinical Context |
|---|---|---|
| Scale | Flakes of stratum corneum | Psoriasis (silvery), seborrheic dermatitis (greasy), tinea (fine), ichthyosis (fish-like) |
| Crust | Dried serum, blood, or pus on skin surface | Honey-colored (impetigo), hemorrhagic (herpes), serous (eczema) |
| Erosion | Superficial loss of epidermis; heals without scarring | Ruptured vesicles/bullae (pemphigus, HSV) |
| Ulcer | Full-thickness loss through epidermis into dermis or deeper; scars | Venous stasis ulcer, pyoderma gangrenosum, SCC |
| Fissure | Linear crack in skin | Hand eczema, heel xerosis, angular cheilitis |
| Excoriation | Superficial abrasion from scratching | Atopic dermatitis, prurigo nodularis, neurotic excoriations |
| Lichenification | Thickened skin with accentuated skin lines from chronic rubbing | Chronic atopic dermatitis, lichen simplex chronicus |
| Atrophy | Thinning of epidermis and/or dermis | Chronic topical steroid use, morphea, lichen sclerosus |
| Scar | Fibrous tissue replacing normal skin after injury | Hypertrophic (stays within wound borders), keloid (extends beyond) |
Configuration & Distribution Terms
| Term | Meaning | Classic Association |
|---|---|---|
| Annular | Ring-shaped | Tinea corporis, granuloma annulare, secondary syphilis |
| Arcuate/Polycyclic | Arc-shaped or overlapping rings | Urticaria, erythema annulare centrifugum |
| Linear | In a line | Allergic contact dermatitis (poison ivy), lichen striatus, Koebner phenomenon |
| Grouped/Herpetiform | Clustered lesions | HSV, dermatitis herpetiformis |
| Reticular | Net-like or lace-like | Livedo reticularis, erythema ab igne |
| Dermatomal | Following a dermatome | Herpes zoster (does not cross midline) |
| Photodistributed | Sun-exposed areas | Drug photosensitivity, SLE, polymorphous light eruption |
| Koebnerization | Lesions arising in sites of trauma | Psoriasis, lichen planus, vitiligo, warts |
| Blaschkoid | Following lines of Blaschko (embryonic cell migration) | Lichen striatus, inflammatory linear verrucous epidermal nevus (ILVEN) |
03 The Dermatologic Exam
A complete skin exam (CSE) requires examination of the entire skin surface including scalp, ears, interdigital spaces, nails, oral mucosa, and genital skin. Adequate lighting is essential — a handheld magnifying lens or dermatoscope (10x polarized or non-polarized) transforms diagnostic accuracy for pigmented lesions. The exam follows a systematic top-to-bottom, front-to-back approach.
Describing a Lesion — The Standard Framework
Every dermatologic finding is described with a structured vocabulary: number (single vs multiple vs innumerable), color (erythematous, violaceous, hyperpigmented, hypopigmented, flesh-colored, pearly), morphology (using primary lesion terms), size (in mm or cm), shape/configuration (round, oval, annular, linear, irregular), distribution (localized, generalized, symmetric, photodistributed, dermatomal, acral), surface changes (smooth, verrucous, scaly, crusted, ulcerated), and palpation characteristics (firm, soft, fluctuant, tender, blanching vs non-blanching).
Dermoscopy (Dermatoscopy)
Dermoscopy is a non-invasive technique using a handheld device (dermatoscope) with magnification (typically 10x) and a light source (polarized or non-polarized) to examine subsurface skin structures invisible to the naked eye. It dramatically improves sensitivity for melanoma detection (from ~60% with naked eye to >90% with experienced dermoscopy). Key dermoscopic structures include: pigment network (typical = benign melanocytic nevus; atypical = melanoma), globules/dots, streaks/pseudopods (melanoma), blue-white veil (melanoma — dermal melanin + fibrosis), arborizing vessels (BCC), leaf-like structures (BCC), strawberry pattern (actinic keratosis), and comma vessels (dermatofibroma).
Distribution Patterns and Differential Diagnosis
| Distribution | Think Of |
|---|---|
| Extensor surfaces (elbows, knees) | Psoriasis, dermatitis herpetiformis |
| Flexural surfaces (antecubital, popliteal fossae) | Atopic dermatitis (adults/children), inverse psoriasis |
| Sun-exposed areas (face, neck, dorsal hands, V-chest) | Actinic keratosis, SCC, BCC, photodermatosis, lupus, rosacea |
| Dermatomal (unilateral, band-like) | Herpes zoster |
| Palms and soles | Secondary syphilis, hand-foot-mouth, erythema multiforme, psoriasis (palmoplantar), dyshidrotic eczema |
| Scalp | Seborrheic dermatitis, psoriasis, tinea capitis, alopecia areata, CCLE |
| Intertriginous (skin folds) | Candidiasis, tinea cruris, inverse psoriasis, intertrigo, Hailey-Hailey |
| Generalized/widespread | Drug eruption, viral exanthem, secondary syphilis, pityriasis rosea, erythroderma |
04 Atopic Dermatitis Inflammatory
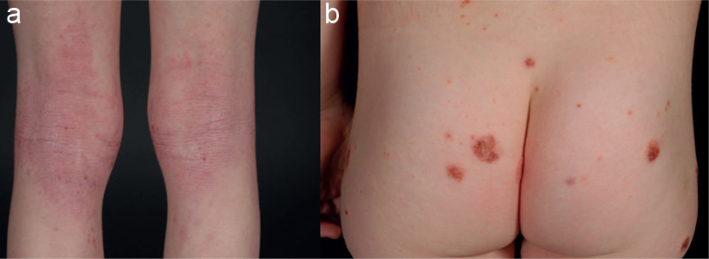
Atopic dermatitis (AD) is the most common inflammatory skin disease, affecting 15-20% of children and 2-10% of adults worldwide. It is a chronic, relapsing, intensely pruritic eczematous dermatosis driven by a combination of epidermal barrier dysfunction (loss-of-function mutations in the filaggrin gene FLG found in ~30% of patients), Th2-skewed immune dysregulation (IL-4, IL-13, IL-31 overexpression), and environmental triggers. AD is part of the "atopic triad" (AD, allergic rhinitis, asthma) — the "atopic march" describes the typical progression from AD in infancy to rhinitis and asthma in childhood (Weidinger & Novak, Lancet 2016).
Age-Dependent Distribution
| Age Group | Typical Distribution | Key Features |
|---|---|---|
| Infants (0-2 yr) | Face (cheeks, forehead), scalp, extensor surfaces | Acute weeping, crusted patches; diaper area typically spared |
| Children (2-12 yr) | Flexural surfaces (antecubital fossae, popliteal fossae, neck, wrists) | Lichenification from chronic scratching; periorbital darkening (Dennie-Morgan folds) |
| Adults | Hands, flexures, face/neck, nipples | Chronic lichenified plaques; may be localized (hand eczema) or generalized |
Severity Assessment — SCORAD and EASI
The SCORAD (Scoring Atopic Dermatitis) index combines affected BSA (using rule of nines: head 9%, each arm 9%, each leg 18%, anterior trunk 18%, posterior trunk 18%), intensity (erythema, edema/papulation, oozing/crusts, excoriation, lichenification, dryness — each scored 0-3), and subjective symptoms (pruritus and sleep loss, each 0-10 VAS). Total score range: 0-103. Mild < 25; moderate 25-50; severe > 50.
The EASI (Eczema Area and Severity Index) assesses four body regions (head/neck, trunk, upper extremities, lower extremities) for erythema, induration/papulation, excoriation, and lichenification (each 0-3), weighted by BSA involvement. Range: 0-72. Mild 1-7; moderate 7-21; severe 21-50; very severe >50. EASI-75 (75% reduction from baseline) is the primary endpoint in most clinical trials for AD biologics.
Treatment Ladder
| Severity | Treatment | Details |
|---|---|---|
| All severities | Emollients/moisturizers | Apply within 3 minutes of bathing ("soak and seal"); ceramide-containing preferred; fragrance-free; use liberally (250-500 g/week for children) |
| Mild | Low-potency topical corticosteroids (TCS) | Hydrocortisone 2.5% or desonide 0.05% for face/folds; mid-potency (triamcinolone 0.1%) for body |
| Moderate | Mid-to-high potency TCS + topical calcineurin inhibitors (TCI) | Tacrolimus 0.1% ointment (Protopic) or pimecrolimus 1% cream (Elidel) for steroid-sparing maintenance, especially face/folds; crisaborole 2% (Eucrisa, PDE4 inhibitor) for mild-moderate |
| Moderate-severe | Phototherapy (NB-UVB) or systemic agents | NB-UVB 2-3x/week; if insufficient: dupilumab (Dupixent, IL-4R-alpha blocker) is first-line systemic — 600 mg loading then 300 mg q2wk SC; tralokinumab (Adbry, anti-IL-13) |
| Severe/refractory | JAK inhibitors or traditional immunosuppressants | Abrocitinib (Cibinqo, JAK1) 100-200 mg daily; upadacitinib (Rinvoq, JAK1) 15-30 mg daily; baricitinib (Olumiant, JAK1/2); cyclosporine 3-5 mg/kg/day (bridge therapy); methotrexate 7.5-25 mg/week; azathioprine 1-3 mg/kg/day |
05 Contact Dermatitis (Allergic & Irritant) Inflammatory
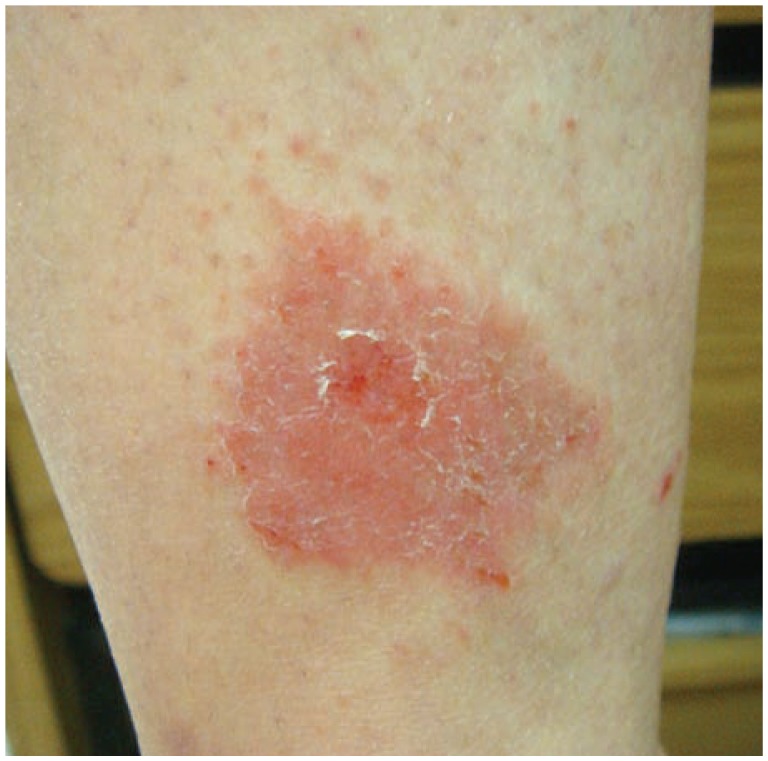
Contact dermatitis accounts for ~90% of occupational skin diseases. It presents as eczematous dermatitis at sites of exposure and is classified into two mechanistically distinct forms:
| Feature | Allergic Contact Dermatitis (ACD) | Irritant Contact Dermatitis (ICD) |
|---|---|---|
| Mechanism | Type IV delayed hypersensitivity (T-cell mediated); requires prior sensitization (5-21 days for first exposure; 12-72 hours on re-exposure) | Direct cytotoxic damage to keratinocytes; no prior sensitization required; dose-dependent |
| Prevalence | ~20% of contact dermatitis | ~80% of contact dermatitis |
| Borders | May extend beyond contact area (id reaction) | Sharply limited to exposure area |
| Common causes | Nickel (#1 allergen globally), poison ivy/oak/sumac (urushiol), fragrances, preservatives (methylisothiazolinone), neomycin, rubber accelerators, hair dyes (PPD) | Soaps/detergents, solvents, water (chronic wet work), acids/alkalis, fiberglass |
| Histology | Spongiotic dermatitis with eosinophils | Spongiotic dermatitis, often with necrotic keratinocytes |
Patch Testing
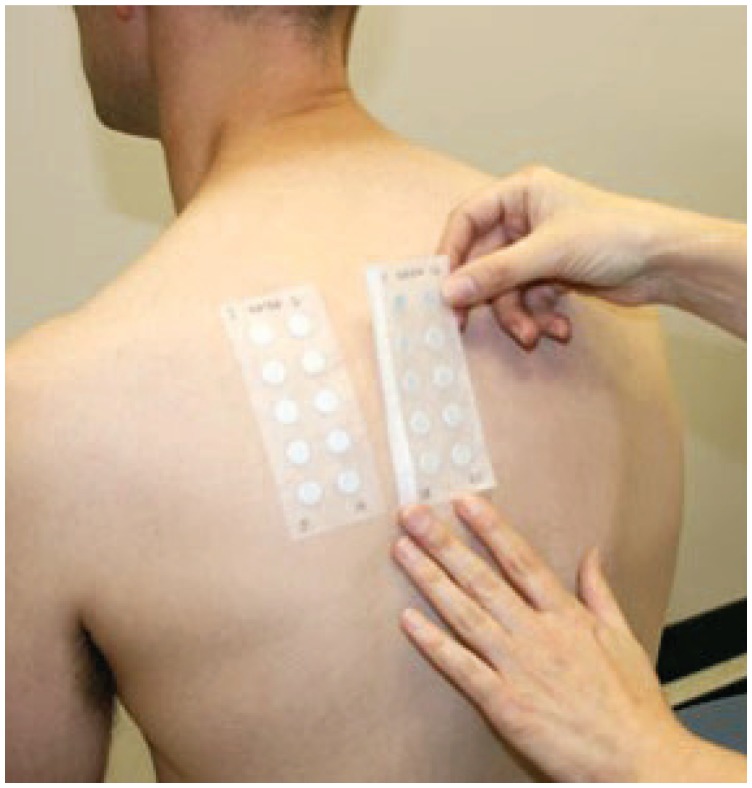
Patch testing is the gold standard for diagnosing ACD. Standardized allergen panels (T.R.U.E. Test has 36 allergens; extended panels from Chemotechnique/SmartPractice have 80+) are applied to the upper back on non-inflamed skin under occlusion. Readings are performed at 48 hours (removal) and 96 hours (delayed reactions). Grading: negative (-), irritant reaction (IR), doubtful/macular erythema (?), weak positive (1+, palpable erythema), strong positive (2+, vesicles), extreme positive (3+, bullous). A positive reaction must be clinically correlated with the patient's exposure history and distribution to confirm relevance.
06 Seborrheic Dermatitis Inflammatory
Seborrheic dermatitis (SD) is a chronic, relapsing inflammatory dermatosis affecting 1-3% of the general population and up to 83% of HIV/AIDS patients. It occurs in areas with high sebaceous gland density: scalp, eyebrows, nasolabial folds, ears, central chest, and intertriginous areas. The pathogenesis involves Malassezia yeast (M. globosa, M. restricta) colonization of sebaceous areas, lipase-mediated hydrolysis of triglycerides in sebum to oleic acid, and an inflammatory response in susceptible individuals. Neurologic associations are strong: SD is markedly more prevalent and severe in Parkinson disease, facial nerve palsy, spinal cord injury, and other conditions that increase sebum production or reduce facial movement.
Presentation: erythematous patches and thin plaques with greasy, yellowish, flaky scale. On the scalp, it ranges from mild dandruff (pityriasis capitis) to thick, adherent plaques. In infants, cradle cap is self-limited seborrheic dermatitis of the scalp. Treatment: topical antifungals (ketoconazole 2% shampoo or cream, ciclopirox 1% shampoo, selenium sulfide 2.5%, zinc pyrithione) are first-line. Low-potency TCS (hydrocortisone 1-2.5%) for acute flares. TCIs (tacrolimus, pimecrolimus) for steroid-sparing maintenance on the face. Refractory cases: oral itraconazole 200 mg/day for 1-2 weeks.
07 Nummular Dermatitis & Stasis Dermatitis Inflammatory
Nummular Dermatitis (Discoid Eczema)
Coin-shaped (nummular), well-demarcated, pruritic, erythematous plaques with fine scale and sometimes exudate/crusting — typically on extensor surfaces of extremities and trunk. More common in men over 50 and in winter (low humidity). Often confused with tinea corporis, but nummular eczema lacks central clearing and KOH is negative. Histology shows spongiotic dermatitis. Treatment: mid-to-high potency TCS (triamcinolone 0.1%, betamethasone valerate 0.1%), emollients, and addressing xerosis triggers. Refractory: phototherapy (NB-UVB).
Stasis Dermatitis
Stasis dermatitis is eczematous inflammation of the lower legs driven by chronic venous insufficiency (CVI). Venous hypertension causes capillary leakage, red blood cell extravasation, and hemosiderin deposition, producing the characteristic brown-red discoloration of the medial lower legs (gaiter area). Progressive changes include scaling, oozing, crusting, lichenification, and ultimately lipodermatosclerosis (fibrotic induration giving the classic "inverted champagne bottle" leg shape). The critical management is addressing the underlying venous disease: compression therapy (30-40 mmHg knee-high stockings — contraindicated if ABI < 0.8), leg elevation, and treatment of superficial venous reflux (endovenous ablation). Topical steroids manage acute flares. Stasis ulcers (venous leg ulcers) are a common complication — characteristically shallow, irregularly bordered, located at the medial malleolus, with a moist granulating base.
08 Psoriasis Papulosquamous
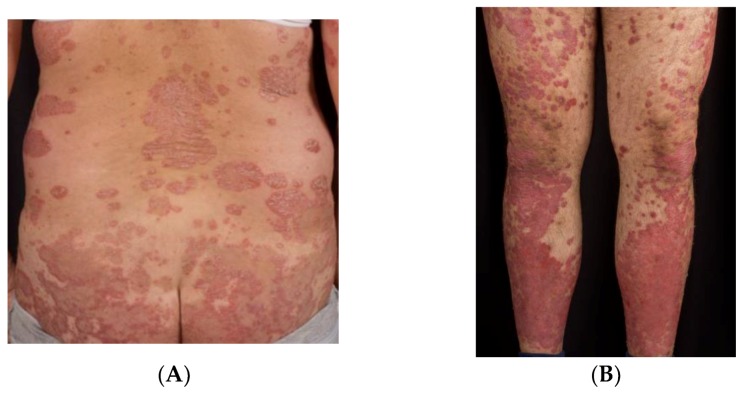
Psoriasis is a chronic, immune-mediated, systemic inflammatory disease affecting 2-3% of the world population. It is driven by the IL-23/Th17 axis: dendritic cells produce IL-23, which activates Th17 cells to secrete IL-17A, IL-17F, and IL-22, driving keratinocyte hyperproliferation, neutrophil recruitment, and angiogenesis. Psoriasis is associated with significant comorbidities: psoriatic arthritis (up to 30% of patients), cardiovascular disease (50% increased risk of MI), metabolic syndrome, depression, and inflammatory bowel disease. It is not merely a skin disease — it is a systemic inflammatory condition with cutaneous manifestations (Armstrong & Read, JAMA 2020).
Clinical Types
| Type | Description | Key Features |
|---|---|---|
| Plaque (vulgaris) | ~90% of cases; well-demarcated, erythematous plaques with thick silvery-white scale | Extensor surfaces (elbows, knees), scalp, lumbosacral area, intergluteal cleft; Auspitz sign (pinpoint bleeding on scale removal); Koebner phenomenon |
| Guttate | Acute onset of small (0.5-1.5 cm) drop-shaped papules and plaques | Classically triggered by streptococcal pharyngitis (2-3 weeks prior); more common in children/young adults; often self-limiting; check ASO titer/throat culture |
| Inverse (flexural) | Erythematous, shiny, well-demarcated plaques in skin folds | Axillae, groin, inframammary, intergluteal; minimal scale (macerated off by moisture); DDx: candidiasis, tinea, intertrigo |
| Pustular (generalized — von Zumbusch) | Acute, widespread sterile pustules on erythematous base | Dermatologic emergency: fever, leukocytosis, hypoalbuminemia, electrolyte abnormalities; triggered by rapid steroid withdrawal, infection, pregnancy; hospitalize |
| Pustular (localized — palmoplantar) | Sterile pustules on palms and soles | Chronic, relapsing; strongly associated with smoking; difficult to treat; acrodermatitis of Hallopeau (digit tips) |
| Erythrodermic | > 90% BSA involvement with erythema and scaling | Dermatologic emergency: impaired thermoregulation, high-output cardiac failure risk, protein loss, infection risk; requires hospitalization |
| Nail psoriasis | Pitting, oil-drop spots, onycholysis, subungual hyperkeratosis | Present in ~50% of psoriasis patients, ~80% of PsA patients; nail pitting distinguishes from onychomycosis (which has subungual debris/dystrophy without pitting) |
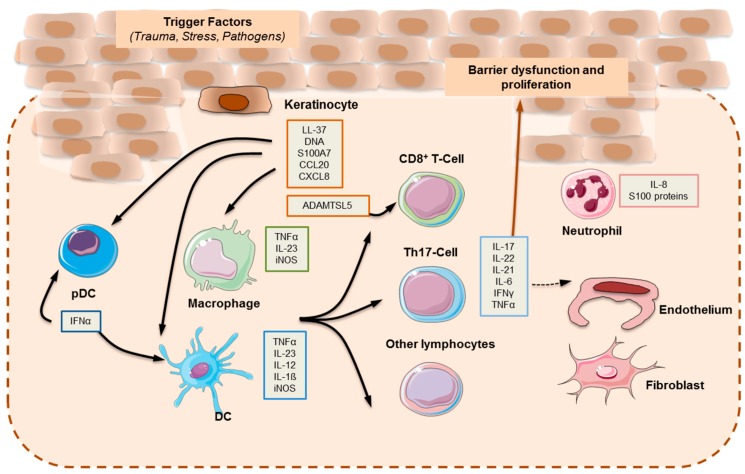
Psoriasis Area and Severity Index (PASI)
PASI is the standard clinical trial outcome measure. It evaluates four body regions (head 10%, upper extremities 20%, trunk 30%, lower extremities 40%) for erythema, induration, and desquamation (each scored 0-4) multiplied by area of involvement (0-6 scale). Total range: 0-72. Mild: PASI < 7 or BSA < 3%; moderate: PASI 7-12 or BSA 3-10%; severe: PASI > 12 or BSA > 10%. PASI 75 (75% improvement from baseline) is the standard efficacy benchmark for biologics; many modern agents achieve PASI 90 or PASI 100.
Treatment by Severity
| Severity | First-Line | Second-Line / Adjuncts |
|---|---|---|
| Mild (< 3% BSA) | Topical corticosteroids (mid-to-high potency for body, low-potency for face/folds); vitamin D analogs (calcipotriene 0.005%) | Combination calcipotriene/betamethasone (Enstilar foam, Taclonex); tazarotene 0.1% (retinoid); coal tar |
| Moderate (3-10% BSA) | Phototherapy (NB-UVB 311 nm, 2-3x/week) or topical therapy to multiple sites | Excimer laser (308 nm) for localized plaques; consider systemic if topical burden too high |
| Severe (> 10% BSA) or moderate with high impact | Biologics (see below) or oral systemics | Combination biologic + topical for residual plaques |
Systemic & Biologic Therapies for Psoriasis
| Drug (Brand) | Target / Mechanism | Dose | Key Monitoring / Pearls |
|---|---|---|---|
| Methotrexate | Dihydrofolate reductase inhibitor | 7.5-25 mg PO/SC weekly + folic acid 1 mg daily | CBC, LFTs, Cr q3 months; hepatotoxicity risk with cumulative dose; avoid alcohol; teratogenic (Category X) |
| Cyclosporine (Neoral) | Calcineurin inhibitor (IL-2 suppression) | 2.5-5 mg/kg/day PO | Limit to 1-2 years; nephrotoxicity (monitor Cr, BP); drug interactions (CYP3A4); bridge to biologic |
| Acitretin (Soriatane) | Systemic retinoid | 25-50 mg/day PO | Lipids, LFTs; teratogenic — women must avoid pregnancy for 3 YEARS after stopping (unlike isotretinoin's 1 month); no alcohol (converts to etretinate with 120-day half-life) |
| Apremilast (Otezla) | PDE4 inhibitor | 30 mg BID after titration | No lab monitoring required; GI side effects (nausea, diarrhea); weight loss; depression screening |
| Adalimumab (Humira) | Anti-TNF-alpha | 80 mg then 40 mg q2wk SC | TB screening (QuantiFERON-Gold) before starting; hepatitis B/C screening; increased infection risk; demyelination rare |
| Secukinumab (Cosentyx) | Anti-IL-17A | 300 mg SC at weeks 0,1,2,3,4 then q4wk | Candidiasis risk; avoid in active IBD (may worsen Crohn's); high PASI 90 rates (~70%) |
| Ixekizumab (Taltz) | Anti-IL-17A | 160 mg then 80 mg q2wk x12wk then q4wk | Similar profile to secukinumab; injection site reactions |
| Guselkumab (Tremfya) | Anti-IL-23 (p19 subunit) | 100 mg at weeks 0, 4, then q8wk SC | PASI 90 rates ~70-80%; favorable safety profile; less frequent dosing than IL-17 agents |
| Risankizumab (Skyrizi) | Anti-IL-23 (p19 subunit) | 150 mg at weeks 0, 4, then q12wk SC | Highest PASI 90/100 rates in head-to-head trials; 12-week dosing interval is longest among biologics (Gordon et al., Lancet 2021) |
| Deucravacitinib (Sotyktu) | TYK2 inhibitor (oral) | 6 mg PO daily | First oral selective TYK2 inhibitor; no lab monitoring requirement per label; superior to apremilast in trials |
09 Pityriasis Rosea Papulosquamous
Pityriasis rosea (PR) is an acute, self-limited papulosquamous eruption most common in adolescents and young adults (10-35 years). It is likely triggered by reactivation of HHV-6 and/or HHV-7. The classic presentation begins with a herald patch (2-10 cm oval, salmon-colored plaque with a collarette of scale) on the trunk, followed 1-2 weeks later by a secondary eruption of smaller, oval, scaly patches oriented along skin tension lines (Langer lines), producing a characteristic "Christmas tree" distribution on the back. The eruption is self-limited, typically resolving in 6-8 weeks. Pruritus is variable. Treatment is symptomatic: emollients, low-potency TCS, antihistamines, and NB-UVB for severe pruritus. Oral acyclovir (800 mg 5x/day for 1 week) may shorten disease course if started within the first week.
10 Lichen Planus Papulosquamous
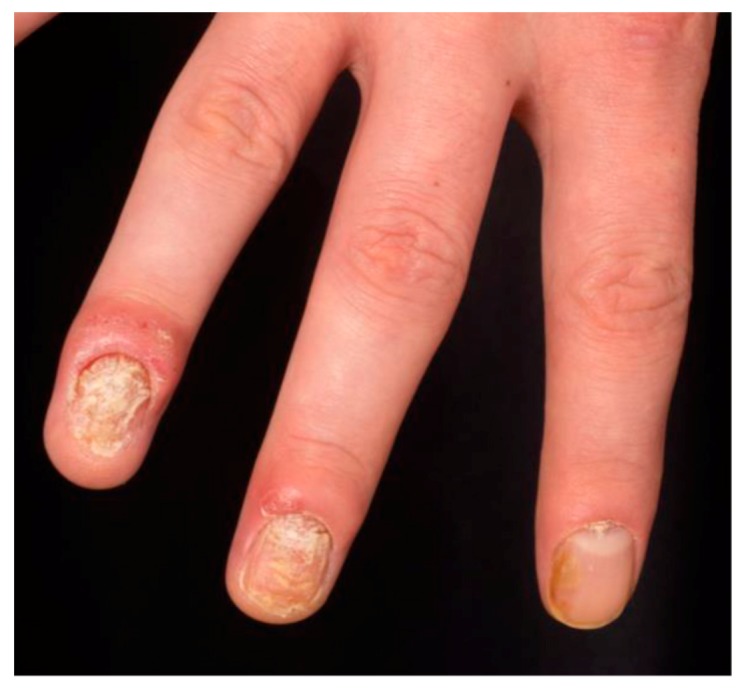
Lichen planus (LP) is a T-cell mediated inflammatory dermatosis characterized by the 5 P's: Pruritic, Purple (violaceous), Polygonal, Planar (flat-topped), Papules. Affects 0.5-1% of the population, peak onset 30-60 years. The pathogenesis involves cytotoxic CD8+ T-cell attack on basal keratinocytes, producing a band-like (lichenoid) lymphocytic infiltrate that obscures the dermal-epidermal junction. Histology shows: hyperkeratosis, irregular (sawtooth) acanthosis, wedge-shaped hypergranulosis, civatte bodies (apoptotic keratinocytes), and band-like lymphocytic infiltrate at the DEJ. Direct immunofluorescence shows fibrinogen deposition at the BMZ in a shaggy pattern.
Classic cutaneous LP: violaceous, flat-topped papules and plaques on the flexor wrists, forearms, ankles, and presacral area. Wickham striae — fine white lacy lines on the surface (corresponding to hypergranulosis) — are pathognomonic and best seen with dermoscopy. Koebner phenomenon is common. Oral LP affects 50-70% of cutaneous LP patients: reticular (white lacy pattern on buccal mucosa — most common, usually asymptomatic), erosive (painful red erosions — increased risk of oral SCC; requires monitoring), and plaque forms. Lichen planopilaris is LP of the scalp producing cicatricial (scarring) alopecia with perifollicular erythema and scale. Drug-induced lichenoid eruptions (beta-blockers, ACE inhibitors, thiazides, antimalarials, gold) can mimic LP — drug history is essential.
Treatment: potent topical corticosteroids (clobetasol 0.05% for cutaneous, triamcinolone acetonide oral paste for mucosal); oral corticosteroids for severe widespread disease; systemic retinoids (acitretin); calcineurin inhibitors for mucosal disease; phototherapy (NB-UVB or PUVA). Cutaneous LP typically self-resolves in 1-2 years; oral LP is chronic.
11 Secondary Syphilis & Other Papulosquamous Mimics Papulosquamous
Secondary syphilis is the "great imitator" and must be considered in the differential diagnosis of virtually any papulosquamous eruption. It occurs 6-8 weeks after the primary chancre (which may have been unnoticed) when Treponema pallidum disseminates hematogenously. Key features: diffuse, symmetric, copper-red to pink, maculopapular or papulosquamous rash that classically involves the palms and soles (a crucial discriminator — few conditions do this), condylomata lata (moist, flat, gray-white papules in intertriginous areas — highly infectious), mucous patches (painless gray-white oral erosions), moth-eaten alopecia (patchy, non-scarring hair loss), generalized lymphadenopathy, and constitutional symptoms (low-grade fever, malaise, weight loss). Diagnosis: RPR or VDRL (screening) + FTA-ABS or TP-PA (confirmatory); darkfield microscopy of condylomata lata. Treatment: benzathine penicillin G 2.4 million units IM x1 (same as primary syphilis). Jarisch-Herxheimer reaction (acute febrile reaction within 24 hours of treatment) is common and should be anticipated.
12 Actinic Keratosis & Field Cancerization Cancer
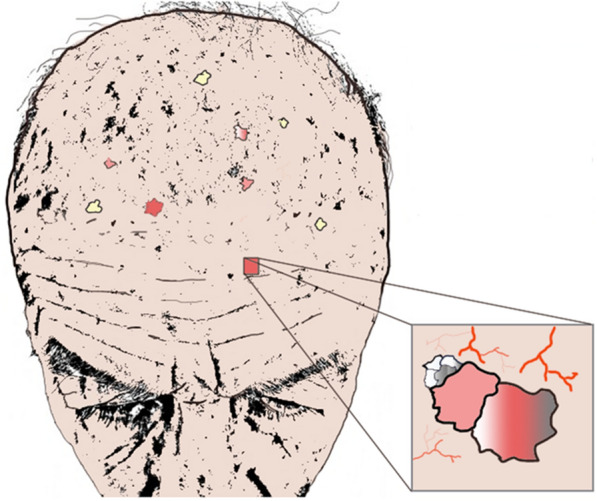
Actinic keratoses (AKs) are UV-induced dysplastic proliferations of epidermal keratinocytes — they represent squamous cell carcinoma in situ and are the most common pre-malignant skin lesion, affecting >58 million Americans. AKs present as rough, gritty, sandpaper-textured papules and patches on sun-exposed skin (face, scalp, dorsal hands, forearms). They are often easier to feel than see. Clinically, they range from flat, pink, scaly macules to hypertrophic, keratotic papules. A cutaneous horn (conical keratinous projection) should be biopsied — SCC is present at the base in ~20% of cases. The risk of any individual AK progressing to invasive SCC is ~0.025-16% per year (wide range in the literature); however, field cancerization — the concept that the entire sun-damaged area harbors subclinical dysplasia — means that patients with visible AKs have diffuse molecular damage in surrounding "normal" skin (Criscione et al., JAMA Dermatol 2009).
Treatment
| Modality | Mechanism | Best For | Notes |
|---|---|---|---|
| Cryotherapy (liquid nitrogen) | Rapid freeze/thaw cycle destroys dysplastic cells | Few isolated lesions | Most commonly used; 5-10 sec freeze; 75-99% clearance rate; hypopigmentation risk |
| 5-Fluorouracil (Efudex 5%, Carac 0.5%) | Antimetabolite — blocks thymidylate synthase in rapidly dividing cells | Field treatment (many AKs) | Apply BID x 2-4 weeks; expected intense erythema/erosion/crusting ("uglier before better"); reveals subclinical AKs |
| Imiquimod 5% (Aldara) or 3.75% (Zyclara) | TLR-7 agonist — activates innate/adaptive immunity | Field treatment | Apply 2-3x/week x 4-16 weeks; less intense reaction than 5-FU; local flu-like symptoms possible |
| Photodynamic therapy (PDT) | Aminolevulinic acid (ALA) or methyl aminolevulinate (MAL) applied → metabolized to protoporphyrin IX → activated by blue or red light → ROS destroy dysplastic cells | Field treatment, facial/scalp AKs | Excellent cosmetic outcome; pain during light exposure; strict sun avoidance 48 hrs post-treatment |
| Diclofenac 3% gel (Solaraze) | COX-2 inhibition in dysplastic cells | Mild, scattered AKs | Apply BID x 60-90 days; mild irritation; slower response |
| Tirbanibulin 1% (Klisyri) | Src kinase/tubulin polymerization inhibitor | Face/scalp AKs (up to 25 cm2) | Newest agent; apply daily x 5 days; short treatment course; minimal inflammation |
13 Basal Cell Carcinoma (BCC) Cancer
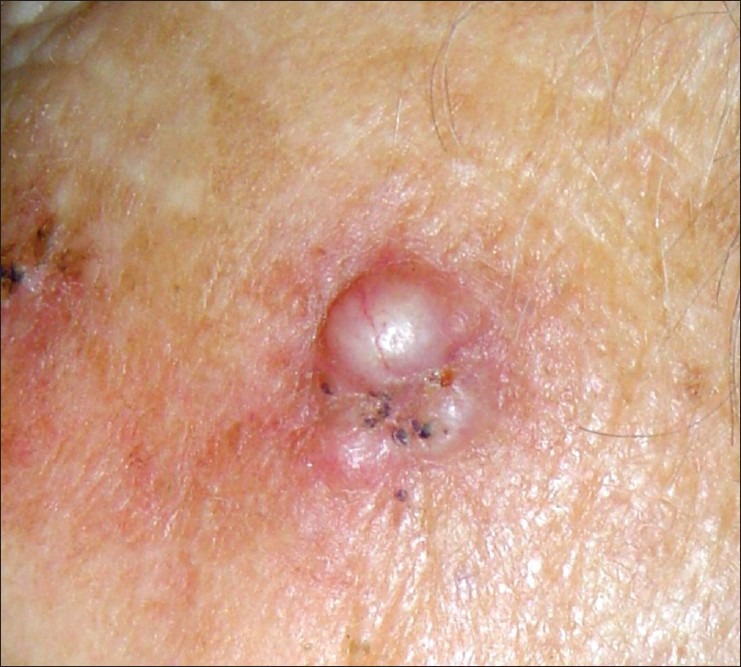
BCC is the most common human malignancy — approximately 3.6 million cases diagnosed annually in the US. It arises from basal cells of the epidermis and hair follicles, driven by aberrant activation of the Hedgehog signaling pathway (mutations in PTCH1 tumor suppressor gene in ~90% of sporadic BCCs). BCC is locally destructive but metastasizes in < 0.1% of cases (metastasis, when it occurs, usually involves regional lymph nodes or lungs). Risk factors: cumulative and intermittent UV exposure, fair skin (Fitzpatrick I-II), prior radiation therapy, immunosuppression, and basal cell nevus syndrome (Gorlin syndrome — AD mutation of PTCH1 producing multiple BCCs from a young age, odontogenic keratocysts, palmar pits, calcified falx cerebri, skeletal anomalies).
BCC Subtypes
| Subtype | Clinical | Histology | Behavior |
|---|---|---|---|
| Nodular (60-80%) | Pearly, translucent papule/nodule with telangiectasias and rolled borders; may ulcerate centrally ("rodent ulcer") | Well-circumscribed nests of basaloid cells with peripheral palisading and retraction artifact | Slow-growing; well-defined margins; favorable for excision |
| Superficial (10-15%) | Thin, erythematous, scaly plaque with pearly rim; trunk and extremities | Buds of basaloid cells attached to undersurface of epidermis | Multifocal; may be treated with topical therapies (imiquimod, 5-FU) or superficial destruction |
| Morpheaform/Infiltrative (5-10%) | Scar-like, indurated, ill-defined, white-yellow plaque; may lack the classic "pearly" appearance | Thin strands of basaloid cells in dense fibrous stroma | Aggressive local behavior; margins poorly defined clinically; high recurrence with standard excision; Mohs surgery indicated |
| Micronodular | Similar to nodular but poorly defined clinically | Small nests of basaloid cells without peripheral palisading | High recurrence rate; subclinical extension beyond clinical margins; Mohs preferred |
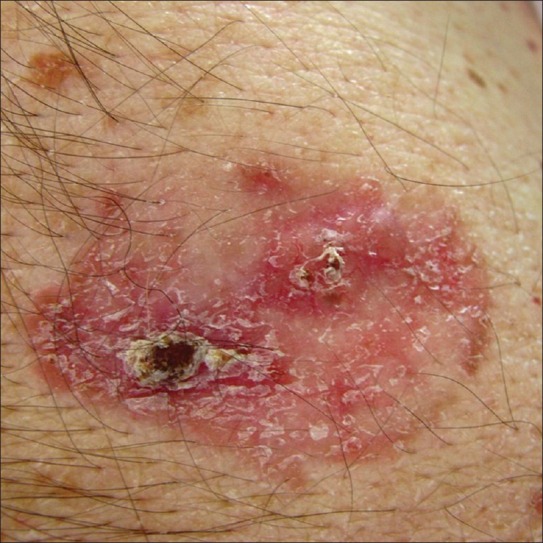
Management
Standard surgical excision with 4 mm clinical margins achieves >95% cure rate for low-risk nodular BCC. Mohs micrographic surgery is indicated for: high-risk anatomic sites (H-zone of face: central face, eyelids, nose, lips, ears, temple), recurrent BCC, morpheaform/infiltrative or micronodular subtypes, large tumors (> 2 cm), perineural invasion, and immunosuppressed patients. Mohs achieves 99% cure rate with maximal tissue conservation. Electrodesiccation and curettage (ED&C) is appropriate for small, low-risk nodular or superficial BCC on the trunk/extremities (not face) — scrape tumor with curette, electrodesiccate base, repeat 2-3 cycles; ~95% cure rate for appropriately selected lesions. Topical imiquimod 5% (5x/week x 6 weeks) and 5-FU (BID x 6-12 weeks) are options for superficial BCC only. For locally advanced or metastatic BCC: vismodegib (Erivedge) 150 mg PO daily or sonidegib (Odomzo) 200 mg PO daily — both are Hedgehog pathway inhibitors (smoothened inhibitors); common side effects include muscle spasms, dysgeusia, alopecia, weight loss; teratogenic (Category D).
14 Squamous Cell Carcinoma (SCC) Cancer
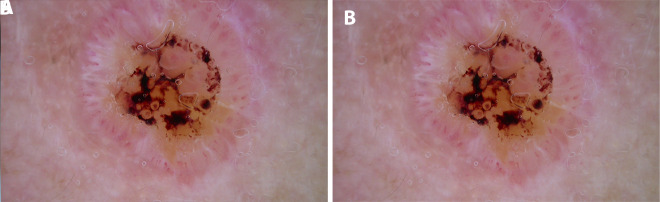
Cutaneous SCC is the second most common skin cancer (~1.8 million cases/year in the US) and the most common skin cancer to metastasize. It arises from malignant proliferation of epidermal keratinocytes, driven primarily by cumulative UV exposure (UVB induces CC→TT signature mutations in p53 tumor suppressor). SCC occurs on a spectrum from in situ disease (Bowen disease) to invasive SCC with potential for regional and distant metastasis. Immunosuppression (organ transplant recipients have 65-250x increased risk of SCC — it is the #1 malignancy in this population) and chronic inflammation (Marjolin ulcer — SCC arising in chronic wounds, burns, scars) are major risk factors (Que et al., JAAD 2018).
High-Risk Features (NCCN Criteria)
| Feature | Low Risk | High Risk |
|---|---|---|
| Location/Size | Trunk/extremities < 2 cm; head/neck < 1 cm | Trunk/extremities ≥ 2 cm; head/neck ≥ 1 cm; lip, ear, temple; arising in scar/chronic wound/radiation field |
| Borders | Well-defined | Poorly defined |
| Histologic subtype | Well-differentiated | Poorly differentiated, desmoplastic, acantholytic, spindle cell |
| Depth | < 2 mm or Clark level I-III | ≥ 2 mm or Clark level IV-V (beyond reticular dermis) |
| Perineural invasion | Absent | Present (especially named nerves ≥ 0.1 mm) |
| Immunosuppression | Immunocompetent | Immunosuppressed (organ transplant, CLL, HIV) |
AJCC 8th Edition Staging (Cutaneous SCC of Head & Neck)
| T Stage | Criteria |
|---|---|
| Tis | Carcinoma in situ (Bowen disease) |
| T1 | ≤ 2 cm in greatest dimension |
| T2 | > 2 cm but ≤ 4 cm |
| T3 | > 4 cm; or minor bone erosion; or perineural invasion of named nerve; or deep invasion beyond subcutaneous fat |
| T4a | Gross cortical bone/marrow invasion |
| T4b | Skull base/axial skeleton invasion |
Management: standard excision with 4-6 mm margins for low-risk SCC. Mohs surgery for high-risk features (same indications as high-risk BCC, plus immunosuppressed patients). Radiation therapy for non-surgical candidates or as adjuvant for perineural invasion/positive margins. Cemiplimab (Libtayo), an anti-PD-1 checkpoint inhibitor, is FDA-approved for locally advanced or metastatic cutaneous SCC not amenable to surgery or radiation — overall response rate ~47% in clinical trials (Migden et al., NEJM 2018).
15 Melanoma Cancer
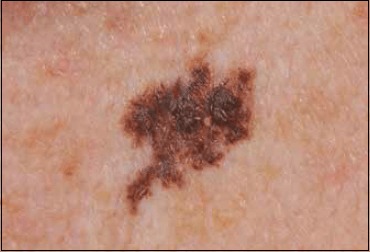
Melanoma accounts for only ~5% of skin cancers but ~75% of skin cancer deaths. Incidence: ~100,000 new invasive cases/year in the US. Arises from malignant transformation of melanocytes, with key driver mutations: BRAF V600E (~50% of cutaneous melanomas — target for vemurafenib/dabrafenib), NRAS (~20%), NF1 (~14%), and KIT (acral/mucosal melanoma). Risk factors: UV exposure (intermittent intense exposure/sunburns), fair skin (Fitzpatrick I-II), >50 melanocytic nevi, atypical (dysplastic) nevi, family history (CDKN2A mutations), personal history of melanoma (increased recurrence risk), and giant congenital melanocytic nevus.
ABCDE Criteria for Clinical Suspicion
| Letter | Feature | Description |
|---|---|---|
| A | Asymmetry | One half does not match the other |
| B | Border irregularity | Edges ragged, notched, blurred |
| C | Color variation | Multiple shades: brown, black, red, white, blue |
| D | Diameter | > 6 mm (though melanomas can be smaller) |
| E | Evolution | Change in size, shape, color, or symptoms (most important criterion) |
Melanoma Subtypes
| Subtype | % Cases | Key Features |
|---|---|---|
| Superficial spreading | ~70% | Irregular borders, color variation; horizontal (radial) growth phase first, then vertical invasion; any body site |
| Nodular | ~15-30% | Rapidly growing, dome-shaped, blue-black or amelanotic nodule; vertical growth from onset (worst prognosis for given depth); lacks ABCDE features early — use "EFG" (Elevated, Firm, Growing) |
| Lentigo maligna | ~5-15% | Slow-growing, irregular tan-brown patch on sun-damaged skin (face) of elderly; in situ (lentigo maligna) → invasive (lentigo maligna melanoma) |
| Acral lentiginous | ~5% | Palms, soles, subungual (nail bed); most common type in dark-skinned individuals and Asians; Hutchinson sign (periungual pigment extension) suggests subungual melanoma |
| Desmoplastic | ~1-4% | Amelanotic, firm, scar-like; high perineural tropism; often diagnosed late; high local recurrence but lower nodal metastasis rate |
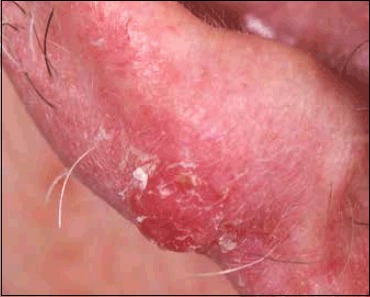
Breslow Depth & Clark Level
Breslow depth — measured in millimeters from the granular layer to the deepest identifiable melanoma cell — is the single most important prognostic factor. Clark level (I-V) describes the anatomic level of invasion but has been largely supplanted by Breslow depth (Clark level is no longer in AJCC 8th edition T-staging). Mitotic rate (mitoses/mm2) was removed from AJCC 8th edition staging criteria but remains prognostically relevant.
AJCC 8th Edition TNM Staging — Melanoma
| T Category | Thickness | Ulceration Status |
|---|---|---|
| Tis | In situ | N/A |
| T1a | < 0.8 mm | Without ulceration |
| T1b | < 0.8 mm with ulceration OR 0.8-1.0 mm +/- ulceration | — |
| T2a | > 1.0-2.0 mm | Without ulceration |
| T2b | > 1.0-2.0 mm | With ulceration |
| T3a | > 2.0-4.0 mm | Without ulceration |
| T3b | > 2.0-4.0 mm | With ulceration |
| T4a | > 4.0 mm | Without ulceration |
| T4b | > 4.0 mm | With ulceration |
Surgical Margins & Sentinel Lymph Node Biopsy
| Breslow Depth | Recommended Excision Margin | SLNB Recommendation |
|---|---|---|
| In situ | 0.5-1.0 cm | Not indicated |
| ≤ 1.0 mm | 1.0 cm | Consider if T1b (ulcerated or 0.8-1.0 mm), high mitotic rate, lymphovascular invasion, or young age |
| 1.01-2.0 mm | 1-2 cm | Recommended (discuss with patient) |
| 2.01-4.0 mm | 2.0 cm | Recommended |
| > 4.0 mm | 2.0 cm | Recommended |
Systemic Therapy for Advanced Melanoma
The treatment landscape for advanced (unresectable stage III/IV) melanoma has been revolutionized by immunotherapy and targeted therapy. Checkpoint inhibitors: nivolumab (Opdivo, anti-PD-1) + ipilimumab (Yervoy, anti-CTLA-4) combination produces 5-year OS ~52%; pembrolizumab (Keytruda, anti-PD-1) monotherapy 5-year OS ~43%. BRAF/MEK inhibitor combinations: for BRAF V600-mutant melanoma: dabrafenib (Tafinlar) + trametinib (Mekinist), or encorafenib (Braftovi) + binimetinib (Mektovi); rapid response rates (~70%) but median duration of response ~12 months before resistance develops. Adjuvant therapy: pembrolizumab or nivolumab for resected stage III melanoma reduces recurrence by ~35-40% (Luke et al., NEJM 2022).
16 Merkel Cell Carcinoma Cancer
Merkel cell carcinoma (MCC) is a rare, aggressive neuroendocrine carcinoma of the skin with approximately 3,000 cases/year in the US. Etiology: ~80% are associated with Merkel cell polyomavirus (MCPyV) integration; the remaining ~20% are UV-driven (higher mutation burden). Risk factors: age >65, immunosuppression (10-30x increased risk), fair skin, chronic UV exposure. Presentation: rapidly growing, painless, dome-shaped, firm, violaceous-to-red nodule — the mnemonic AEIOU (Asymptomatic, Expanding rapidly, Immunosuppression, Older than 50, UV-exposed/fair skin) captures the typical profile. Often misdiagnosed as cyst or BCC. Histology: small round blue cell tumor (DDx: lymphoma, small cell lung cancer, melanoma); immunohistochemistry: CK20 positive (perinuclear dot pattern, highly characteristic), TTF-1 negative (distinguishes from small cell lung cancer), chromogranin/synaptophysin positive (neuroendocrine markers).
Staging: AJCC 8th edition uses separate staging for MCC recognizing its aggressive behavior. 5-year OS: localized ~55%, regional ~36%, distant ~14%. Treatment: wide local excision (1-2 cm margins) + SLNB (high rate of occult nodal metastasis ~30%); adjuvant radiation to primary site and draining nodal basin is commonly performed. For advanced/metastatic MCC: avelumab (Bavencio), anti-PD-L1, was the first FDA-approved therapy for metastatic MCC (ORR ~33%); pembrolizumab also shows activity.
17 Bacterial Skin Infections Infections
Impetigo
Impetigo is the most common bacterial skin infection in children. Non-bullous impetigo (~70%): begins as vesicles/pustules that rupture leaving classic honey-colored crusted erosions, caused by Staphylococcus aureus or Group A Streptococcus (GAS). Bullous impetigo (~30%): flaccid, clear-fluid bullae caused specifically by S. aureus producing exfoliative toxins (same toxins responsible for staphylococcal scalded skin syndrome in neonates). Treatment: localized disease — topical mupirocin 2% (Bactroban) or retapamulin 1% (Altabax) TID x 5 days; widespread or refractory — oral dicloxacillin 250 mg QID or cephalexin 250-500 mg QID x 7 days. If MRSA suspected: TMP-SMX DS or doxycycline.
Cellulitis & Erysipelas
Cellulitis: acute bacterial infection of the deep dermis and subcutaneous tissue — erythema, warmth, swelling, tenderness with poorly defined borders. Most commonly caused by beta-hemolytic streptococci (GAS) and S. aureus. Risk factors: skin barrier disruption (tinea pedis is the most common portal of entry for lower extremity cellulitis — treat the tinea to prevent recurrence), lymphedema, venous insufficiency, obesity. Treatment: mild — oral cephalexin 500 mg QID or dicloxacillin 500 mg QID x 5-10 days; moderate/severe — IV cefazolin 1-2 g q8h or nafcillin 1-2 g q4-6h. Erysipelas: more superficial infection (upper dermis/lymphatics) with sharply demarcated, raised, erythematous plaque and prominent lymphatic involvement — classic "butterfly" distribution on the face or well-demarcated patch on the leg; almost always GAS; treat with penicillin VK or amoxicillin.
MRSA Skin Infections
Community-acquired MRSA (CA-MRSA, most commonly USA300/PVL-positive) presents primarily as skin and soft tissue infections — recurrent abscesses, furuncles, and carbuncles, often mistaken for "spider bites." Treatment of abscesses: incision and drainage (I&D) is the primary treatment — antibiotics alone without I&D are insufficient. For surrounding cellulitis or systemic symptoms, add oral TMP-SMX DS BID, doxycycline 100 mg BID, or clindamycin 300-450 mg QID x 7-10 days. Decolonization protocol for recurrent MRSA: mupirocin 2% ointment to anterior nares BID x 5 days + chlorhexidine body washes x 5 days; treat household contacts simultaneously.
Necrotizing Fasciitis
Necrotizing fasciitis is a rapidly progressive, life-threatening deep soft tissue infection that spreads along fascial planes with necrosis of subcutaneous tissue and fascia. Mortality: 20-40% even with treatment. Type I (polymicrobial): mixed aerobic/anaerobic organisms; typically in diabetics, post-surgical, or perianal (Fournier gangrene). Type II (monomicrobial): usually GAS (Streptococcus pyogenes), can occur in healthy individuals after minor trauma. Clinical clues that distinguish from simple cellulitis: pain out of proportion to exam findings, rapid spread, crepitus (subcutaneous gas), dusky/purple skin discoloration, bullae/hemorrhagic bullae, systemic toxicity (sepsis), and failure to respond to appropriate antibiotics. The LRINEC score (Laboratory Risk Indicator for Necrotizing Fasciitis) uses CRP, WBC, hemoglobin, sodium, creatinine, and glucose — score ≥6 suggests necrotizing fasciitis (but clinical suspicion should not wait for scoring). Treatment: emergent surgical debridement (the definitive treatment — delay increases mortality), broad-spectrum antibiotics (vancomycin + piperacillin-tazobactam + clindamycin — clindamycin inhibits toxin production), ICU-level care, serial debridements as needed.
18 Viral Skin Infections Infections
Herpes Simplex Virus (HSV)
HSV-1 and HSV-2 produce grouped vesicles on an erythematous base that ulcerate and crust. HSV-1 classically causes orolabial herpes (cold sores); HSV-2 classically causes genital herpes (though either type can infect either site). Primary infection is typically more severe and prolonged (7-14 days) than recurrences (5-7 days). Eczema herpeticum — disseminated HSV in patients with atopic dermatitis — is a dermatologic emergency: widespread, monomorphic, punched-out erosions/vesicles; requires IV acyclovir. Herpetic whitlow: HSV of the finger. Tzanck smear shows multinucleated giant cells (also seen in VZV — not specific to HSV); PCR is the gold standard. Treatment: acyclovir 400 mg TID x 7-10 days (primary) or 400 mg TID x 5 days (recurrence); valacyclovir 1 g BID x 7-10 days (primary) or 500 mg BID x 3 days (recurrence). Suppressive therapy: valacyclovir 500 mg-1 g daily for frequent recurrences (≥6/year).
Varicella-Zoster Virus (VZV)
Varicella (chickenpox): primary VZV infection producing generalized, pruritic vesicular rash in crops (lesions in various stages — macules, papules, vesicles, crusts simultaneously — "crops of lesions in different stages" is the classic teaching). Herpes zoster (shingles): reactivation of latent VZV in dorsal root ganglia producing painful, unilateral, dermatomal vesicular eruption (does not cross midline). Thoracic dermatomes are most common. Complications: postherpetic neuralgia (PHN) (persistent pain >90 days after rash — risk increases with age), herpes zoster ophthalmicus (HZO) (V1 dermatome — Hutchinson sign: vesicles on the nose tip suggests nasociliary nerve involvement and high risk of ocular complications — urgent ophthalmology referral), and disseminated zoster (immunosuppressed patients — >20 vesicles outside the primary dermatome). Treatment: valacyclovir 1 g TID x 7 days or acyclovir 800 mg 5x/day x 7 days, ideally started within 72 hours of rash onset. Shingrix (recombinant adjuvanted vaccine) recommended for adults ≥50 — 2-dose series; >90% efficacy in preventing zoster and PHN.
Human Papillomavirus (HPV) — Warts
HPV infects keratinocytes through microabrasions. Common warts (verruca vulgaris, HPV 2/4): dome-shaped papules with verrucous surface and thrombosed capillaries ("black dots"). Plantar warts (verruca plantaris, HPV 1): endophytic on soles, painful with lateral pressure (vs callus: painful with direct pressure), disrupt skin lines. Flat warts (verruca plana, HPV 3/10): small, flat-topped papules on face and extremities; numerous. Condylomata acuminata (genital warts, HPV 6/11): soft, flesh-colored, filiform or cauliflower-like papules; oncogenic strains HPV 16/18 cause cervical, anal, and oropharyngeal carcinoma (not the same strains that cause visible warts). Treatment: destructive (cryotherapy, electrosurgery, laser, salicylic acid 17-40%), immunologic (imiquimod 5%), chemical (cantharidin, podophyllotoxin for genital warts). No single treatment is universally effective; ~65% of warts resolve spontaneously within 2 years.
Molluscum Contagiosum
Caused by a poxvirus (Molluscum contagiosum virus). Presents as discrete, dome-shaped, flesh-colored to pearly papules with characteristic central umbilication (dell). Common in children (spread by skin-to-skin contact and fomites) and sexually active adults (genital distribution). Widespread/large molluscum in adults should prompt HIV testing. Self-limited in immunocompetent hosts (6-12 months), but treatment may be sought for cosmesis or to prevent spread: cantharidin (vesicant applied in office), cryotherapy, curettage.
19 Fungal Skin Infections Infections
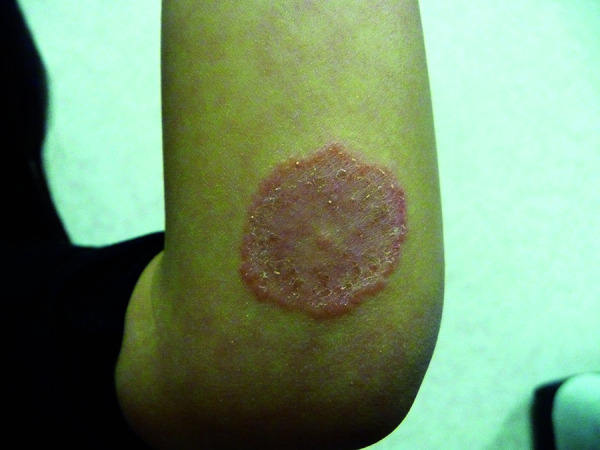
Dermatophytosis (Tinea)
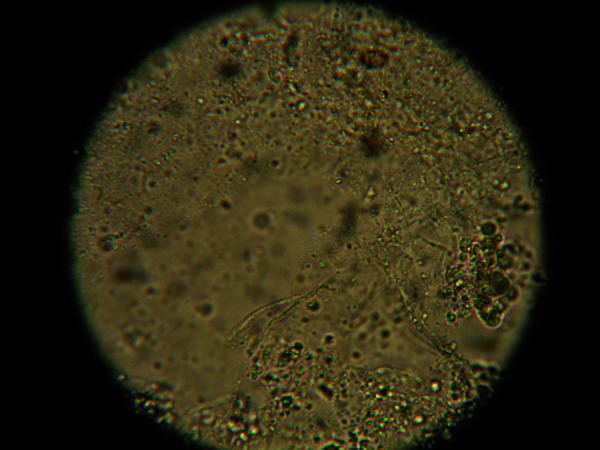
Dermatophytes (Trichophyton, Microsporum, Epidermophyton) infect keratinized tissue (stratum corneum, hair, nails). Diagnosis: KOH preparation — scrape scale from active advancing border, add 10-20% KOH, examine under microscopy for branching septate hyphae. Fungal culture (Sabouraud agar or dermatophyte test medium/DTM) confirms species.
| Type | Location | Clinical Features | Treatment |
|---|---|---|---|
| Tinea corporis | Body (trunk, extremities) | Annular, scaly, erythematous plaques with central clearing and advancing raised border | Topical: terbinafine 1% or clotrimazole 1% BID x 2-4 weeks |
| Tinea pedis | Feet | Interdigital (most common), moccasin (chronic scaling of soles — consider terbinafine PO), vesiculobullous (acute) | Topical antifungal x 4 weeks; moccasin type may need oral terbinafine 250 mg/day x 2-6 weeks |
| Tinea cruris | Groin/inner thighs | Erythematous, scaly plaques sparing the scrotum (vs candida which involves scrotum + satellite papules) | Topical antifungal x 2-4 weeks; keep area dry |
| Tinea capitis | Scalp | Scaly patches with hair breakage ("black dot" = broken hairs at surface), kerion (boggy, tender mass with pustules — not abscess, do not I&D) | MUST use oral antifungal — topicals do not penetrate hair follicle: griseofulvin 20-25 mg/kg/day x 6-8 weeks (first-line in children); terbinafine 250 mg/day x 4-6 weeks (adults/Trichophyton) |
| Tinea versicolor | Trunk, proximal extremities | Multiple hypopigmented or hyperpigmented macules/patches with fine scale; caused by Malassezia furfur (a yeast, not dermatophyte); KOH shows "spaghetti and meatballs" (short hyphae + round spores) | Topical: selenium sulfide 2.5% lotion or ketoconazole 2% shampoo left on 10 min before rinsing; oral: fluconazole 300 mg x 1 dose repeated in 2 weeks |
Cutaneous Candidiasis
Candida albicans (and increasingly non-albicans species) causes superficial infection in warm, moist, occluded areas. Intertriginous candidiasis: beefy-red, erythematous patches with characteristic satellite papules/pustules at the periphery (key distinguishing feature from tinea, which lacks satellites). Oral candidiasis (thrush): white pseudomembranes on buccal mucosa that can be scraped off (DDx: oral hairy leukoplakia — cannot be scraped off, EBV-associated, seen in HIV). Angular cheilitis: erythema/fissuring at oral commissures — often Candida +/- S. aureus. Treatment: topical nystatin cream or clotrimazole cream BID x 2-4 weeks; oral fluconazole 150 mg x 1 for vulvovaginal candidiasis; oral fluconazole 100-200 mg daily for extensive mucocutaneous disease.
Onychomycosis
Fungal nail infection affects ~10% of the general population. Distal lateral subungual onychomycosis (DLSO) is the most common pattern: distal thickening, subungual debris, yellow-brown discoloration, onycholysis. White superficial onychomycosis: white, crumbly patches on the nail plate surface. Proximal subungual onychomycosis: white discoloration at the proximal nail fold — in an immunocompetent patient, consider HIV testing. Always confirm with KOH, culture, or PAS-stained nail clipping before starting systemic therapy. Treatment: terbinafine 250 mg PO daily x 6 weeks (fingernails) or 12 weeks (toenails) — mycologic cure ~70%, complete cure ~40%; itraconazole 200 mg BID x 1 week/month for 2-3 months (pulse dosing); efinaconazole 10% topical solution (Jublia) or tavaborole 5% solution (Kerydin) daily x 48 weeks for mild-moderate disease or patients who cannot take oral therapy. Monitor LFTs with oral antifungals.
20 Parasitic Infestations Infections
Scabies
Caused by the mite Sarcoptes scabiei var. hominis burrowing into the stratum corneum. Hallmarks: intense, generalized pruritus (worse at night, due to mite activity and delayed-type hypersensitivity), burrows (serpiginous, 2-10 mm tracks, most commonly in interdigital web spaces, wrists, axillae, periumbilical, genitalia, buttocks), papules, vesicles, and excoriations. Spares the head in adults (but includes scalp and face in infants/elderly/immunosuppressed). Norwegian (crusted) scabies: massive mite burden (millions vs 10-15 mites in ordinary scabies) in immunosuppressed or neurologically impaired patients — thick, crusted, hyperkeratotic plaques, often misdiagnosed as psoriasis; highly contagious. Diagnosis: mineral oil prep — scrape burrow, examine under microscopy for mites, eggs, or fecal pellets (scybala). Treatment: permethrin 5% cream applied neck-down, left on 8-14 hours, repeated in 1-2 weeks (kills mites but not all eggs — hence repeat); oral ivermectin 200 mcg/kg x 1 dose, repeated in 2 weeks (first-line for crusted scabies; also preferred in institutional outbreaks); treat ALL close contacts and household members simultaneously; wash bedding/clothing in hot water.
Pediculosis (Lice)
Head lice (Pediculus humanus capitis): scalp pruritus, visible nits (eggs) cemented to hair shafts — nits within 6 mm of scalp are viable. Treatment: permethrin 1% lotion (OTC), malathion 0.5% lotion, spinosad 0.9% suspension (Natroba), ivermectin 0.5% lotion (Sklice — single application, no nit combing needed). Body lice (Pediculus humanus corporis): live in clothing seams (not on body); pruritus with excoriations on trunk; associated with homelessness; vector for epidemic typhus (Rickettsia prowazekii), trench fever (Bartonella quintana), relapsing fever (Borrelia recurrentis). Treatment: improved hygiene + laundering clothing at >130 degrees F. Pubic lice (Pthirus pubis): intensely pruritic; found in pubic hair, axillae, eyebrows, eyelashes; treatment: permethrin 1% or malathion; for eyelash involvement: petroleum jelly BID x 10 days (suffocates lice) or ivermectin PO.
Cutaneous Larva Migrans
Caused by penetration of hookworm larvae (Ancylostoma braziliense, A. caninum) through skin in contact with contaminated sand/soil (beaches, sandboxes). Larvae cannot penetrate the basement membrane in humans (accidental host) and migrate through the epidermis, producing characteristic intensely pruritic, serpiginous, erythematous, raised tracks that advance 1-2 cm/day. Usually on feet, buttocks, or hands. Self-limited but slow to resolve (weeks to months). Treatment: oral ivermectin 200 mcg/kg x 1 dose (most effective) or albendazole 400 mg daily x 3 days; topical thiabendazole is an alternative.
21 Pemphigus Vulgaris Autoimmune
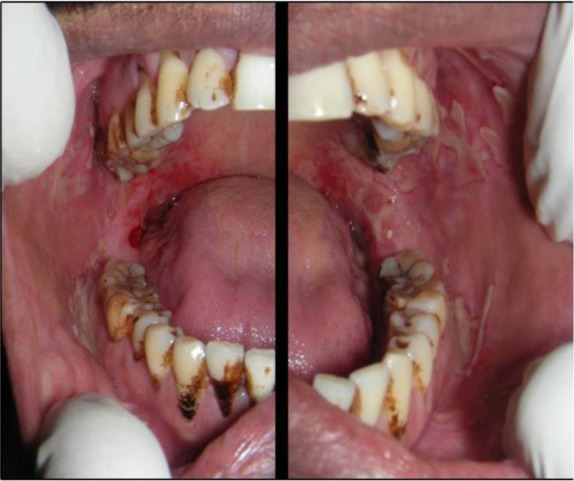
Pemphigus vulgaris (PV) is a potentially fatal autoimmune blistering disease caused by IgG autoantibodies against desmoglein 3 (mucosal predominant) and/or desmoglein 1 (mucocutaneous). These desmosomal cadherins are essential for keratinocyte-to-keratinocyte adhesion; antibody binding causes acantholysis (loss of intercellular adhesion) producing flaccid blisters and erosions. Before the steroid era, mortality exceeded 75%; with current therapy it is ~5-10%. Peak onset: 40-60 years.
Presentation: almost always begins with painful oral mucosal erosions (rarely intact blisters — they rupture immediately due to thin mucosal epithelium) that precede skin involvement by weeks to months. Cutaneous lesions: flaccid blisters that rupture easily leaving painful, non-healing erosions; Nikolsky sign positive (lateral pressure on normal-appearing skin adjacent to a lesion causes shearing/blister formation); Asboe-Hansen sign (direct pressure on a bulla causes lateral extension). Diagnosis: skin biopsy for histology (suprabasal acantholysis with "tombstone" pattern — basal cells still attached to basement membrane with detached suprabasal cells), direct immunofluorescence (DIF) of perilesional skin (intercellular IgG and C3 in a "chicken-wire" or "fishnet" pattern — gold standard), and serum anti-desmoglein 1/3 ELISA (antibody titers correlate with disease activity).
Treatment: systemic corticosteroids (prednisone 1-2 mg/kg/day) for acute control, tapered with steroid-sparing agents. Rituximab (Rituxan) — anti-CD20 B-cell depleting antibody — is now considered first-line steroid-sparing therapy based on the RITUX 3 trial (rituximab + low-dose prednisone superior to prednisone alone for complete remission) (Joly et al., Lancet 2017). Other steroid-sparing agents: mycophenolate mofetil (CellCept) 2-3 g/day, azathioprine 2-3 mg/kg/day (check TPMT activity before starting), IVIG (for refractory disease).
22 Bullous Pemphigoid Autoimmune
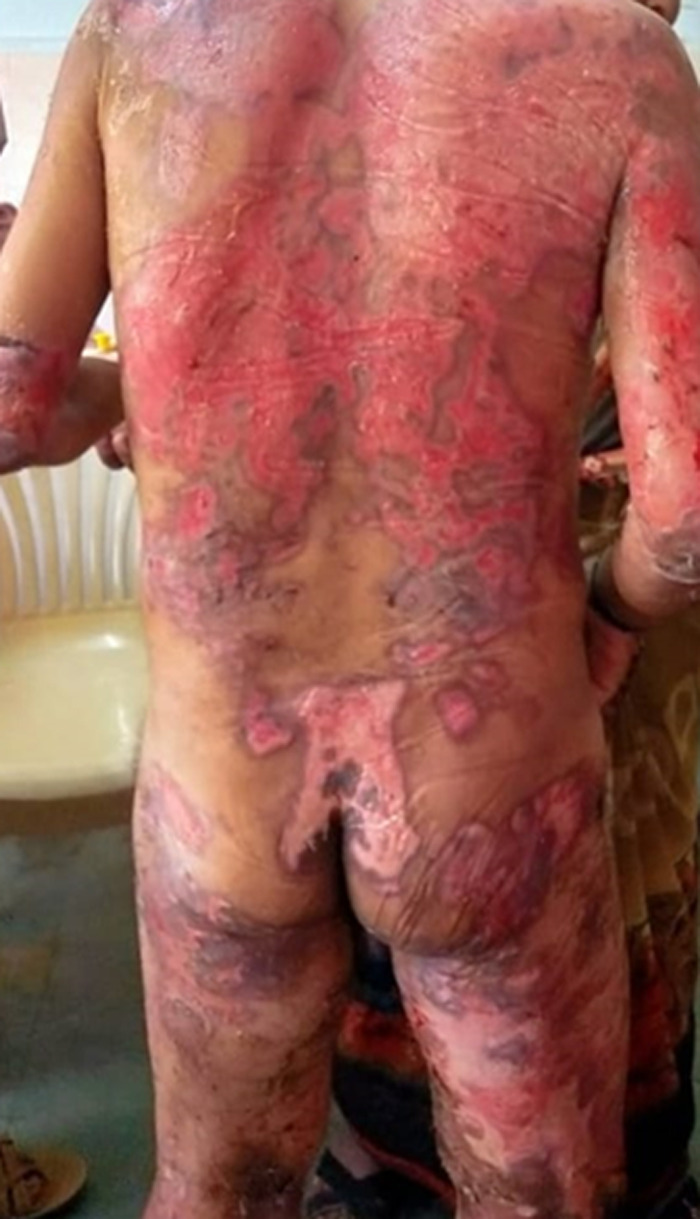
Bullous pemphigoid (BP) is the most common autoimmune blistering disease, primarily affecting elderly patients (>70 years). IgG autoantibodies target BP180 (collagen XVII) and BP230 at the dermal-epidermal junction (hemidesmosome components), causing subepidermal blistering. Unlike pemphigus, the split is below the epidermis, producing tense blisters that do not rupture as easily. Nikolsky sign is typically negative.
Presentation: prodromal urticarial or eczematous phase (weeks to months — often misdiagnosed as eczema) followed by tense bullae on erythematous or normal-appearing skin, predominantly on the trunk, proximal extremities, and flexural areas. Oral involvement occurs in ~10-30% (vs nearly 100% in PV). Pruritus is often severe. Diagnosis: biopsy shows subepidermal blister with eosinophil-rich infiltrate; DIF of perilesional skin shows linear IgG and C3 along the BMZ (linear, not intercellular — distinguishes from pemphigus); serum anti-BP180/BP230 ELISA.
Treatment: mild/localized — superpotent topical corticosteroid (clobetasol 0.05% to entire body) is as effective as oral prednisone with fewer side effects (landmark trial by Joly et al., 2002). Moderate/severe — oral prednisone 0.5 mg/kg/day (lower doses than PV). Steroid-sparing: doxycycline 200 mg/day (anti-inflammatory properties), methotrexate, mycophenolate mofetil, azathioprine, dapsone, or rituximab for refractory cases. Drug-induced BP (DPP-4 inhibitors/gliptins — notably vildagliptin, linagliptin — are an increasingly recognized trigger; PD-1/PD-L1 checkpoint inhibitors are another important cause).
23 Dermatitis Herpetiformis Autoimmune
Dermatitis herpetiformis (DH) is the cutaneous manifestation of celiac disease (gluten-sensitive enteropathy). IgA antibodies against epidermal transglutaminase (eTG) deposit in the dermal papillae, causing neutrophilic inflammation. ~90% of DH patients have underlying celiac disease on intestinal biopsy (often subclinical). Presentation: intensely pruritic, grouped (herpetiform) vesicles and papules on extensor surfaces — elbows (most common), knees, buttocks, posterior scalp, upper back. Vesicles are often excoriated (patients scratch them before they can be examined intact). Diagnosis: DIF of perilesional uninvolved skin (not the lesion itself) shows granular IgA deposits at the dermal papillae — pathognomonic. Serum: anti-tissue transglutaminase (tTG) IgA and anti-endomysial antibodies (same as celiac disease workup). Treatment: dapsone (diaminodiphenylsulfone) 25-150 mg/day produces dramatic improvement within 24-48 hours (so fast it is almost diagnostic); check G6PD before starting (risk of hemolytic anemia); monitor CBC every 2 weeks x 3 months then every 3 months. Long-term: strict gluten-free diet controls both the skin and intestinal disease and may allow discontinuation of dapsone after 1-2 years.
24 Dermatomyositis Autoimmune
Dermatomyositis (DM) is an autoimmune inflammatory myopathy with characteristic cutaneous findings. The pathognomonic skin findings are: heliotrope rash (violaceous erythema/edema of the upper eyelids), Gottron papules (violaceous, flat-topped papules over the MCP and IP joints, elbows, and knees — DDx: lupus affects the skin between the joints), Gottron sign (macular violaceous erythema over the same distribution), shawl sign (erythema over the posterior neck and upper back), V-sign (V-shaped erythema on the anterior chest/neck), mechanic's hands (hyperkeratotic, fissured skin on the lateral fingers — associated with anti-synthetase antibodies), and periungual telangiectasias with ragged cuticles (dilated nailfold capillary loops visible on dermoscopy/capillaroscopy).
Muscle involvement: symmetric proximal weakness (difficulty rising from chair, climbing stairs, lifting arms above head); elevated CK, aldolase, LDH; EMG shows myopathic changes; MRI shows muscle edema; muscle biopsy shows perifascicular atrophy (pathognomonic). Amyopathic/clinically amyopathic dermatomyositis (CADM): classic skin findings without clinically significant muscle weakness — carries significant malignancy and ILD risk. Autoantibodies: anti-Mi-2 (classic DM, good prognosis), anti-MDA5 (CADM, rapidly progressive ILD — can be fatal), anti-TIF1-gamma (strongly associated with malignancy), anti-NXP2 (calcinosis, malignancy). Malignancy screening is essential: DM carries a 3-8x increased risk of underlying malignancy (most commonly ovarian, lung, GI, breast, lymphoma); risk is highest in the first 3 years after diagnosis; age-appropriate cancer screening plus CT chest/abdomen/pelvis is standard.
Treatment: systemic corticosteroids (prednisone 1 mg/kg/day) + steroid-sparing immunosuppressant (methotrexate, azathioprine, mycophenolate mofetil); IVIG for refractory disease; rituximab. For skin: photoprotection (photosensitive), hydroxychloroquine, topical steroids/calcineurin inhibitors.
25 Cutaneous Lupus Erythematosus Autoimmune
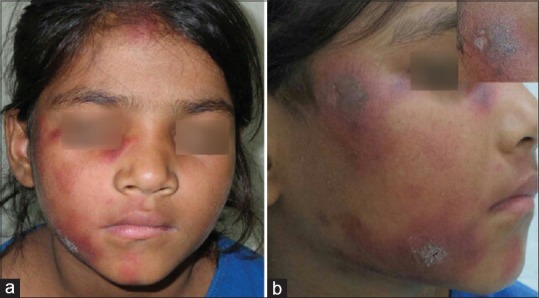
Cutaneous lupus erythematosus (CLE) encompasses three main forms, which may occur independently or in the context of systemic lupus erythematosus (SLE):
| Type | Clinical Features | Association with SLE | Key Points |
|---|---|---|---|
| Acute CLE (ACLE) | Malar ("butterfly") rash: erythema over cheeks and nose bridge, sparing nasolabial folds; photosensitive; may be generalized as widespread maculopapular eruption | ~100% (ACLE is essentially always part of SLE) | Sparing of nasolabial folds distinguishes from seborrheic dermatitis (which involves them) and rosacea |
| Subacute CLE (SCLE) | Annular-polycyclic or papulosquamous (psoriasiform) plaques on sun-exposed areas (upper trunk, arms); photosensitive; heals without scarring | ~50% meet SLE criteria; strong association with anti-Ro/SSA antibodies | Drug-induced SCLE: thiazides, terbinafine, PPIs, calcium channel blockers, anti-TNF agents — resolves with drug discontinuation |
| Chronic CLE — Discoid (DLE) | Well-defined, erythematous, scaly plaques with follicular plugging, atrophic scarring, dyspigmentation (central hypopigmentation with hyperpigmented rim); most common on scalp, ears, face | ~5-10% progress to SLE; but ~25% of SLE patients have DLE | Scarring alopecia when scalp is involved (hair loss is permanent); biopsy shows interface dermatitis with follicular plugging; DIF shows "lupus band" (granular IgG, IgM, C3 at DEJ) |
Workup for all CLE: ANA, anti-dsDNA, anti-Ro/La, complement (C3, C4), CBC, urinalysis, renal function. Treatment: strict photoprotection (broad-spectrum SPF 50+, sun avoidance — UV can trigger or worsen CLE), topical corticosteroids, topical calcineurin inhibitors, and hydroxychloroquine (Plaquenil) 200-400 mg/day — first-line systemic therapy (requires baseline and annual ophthalmologic screening after 5 years for retinal toxicity). Refractory: quinacrine 100 mg/day (may be added to HCQ), methotrexate, mycophenolate mofetil, thalidomide/lenalidomide (for severe refractory DLE).
26 Morphea & Systemic Sclerosis Autoimmune
Morphea (Localized Scleroderma)
Morphea is a localized fibrotic condition of the skin characterized by excessive collagen deposition in the dermis and subcutis. It is NOT systemic sclerosis — morphea does not involve internal organs, does not cause Raynaud phenomenon, and does not have sclerodactyly. Types: plaque morphea (most common — well-defined, indurated, ivory-white plaque with lilac/violaceous border indicating active inflammation), linear morphea (band-like — when on the face/scalp, called en coup de sabre; can involve underlying muscle and bone in children), generalized morphea (≥4 plaques involving ≥2 body regions), and deep morphea (involves subcutis and fascia). Treatment: active disease (lilac ring present) — topical tacrolimus, mid-to-high potency TCS, phototherapy (UVA1), or systemic methotrexate + corticosteroids for rapidly progressive or disabling disease. Inactive ("burnt-out") morphea may leave atrophic, hyperpigmented patches that do not require treatment.
Systemic Sclerosis (Scleroderma) — Cutaneous Features
Systemic sclerosis (SSc) is an autoimmune connective tissue disease characterized by vascular damage, immune activation, and progressive fibrosis of skin and internal organs. Cutaneous features the dermatologist recognizes: Raynaud phenomenon (often the first symptom — triphasic color changes: white→blue→red), sclerodactyly (skin tightening of the fingers), digital ulcers and pitting scars, calcinosis cutis (subcutaneous calcium deposits), telangiectasias (matted perioral and facial telangiectasias), and nailfold capillary changes (dilated loops, avascular areas — visible on dermoscopy/capillaroscopy; distinguishes SSc-associated Raynaud from primary Raynaud). Limited cutaneous SSc (formerly CREST): skin thickening distal to elbows/knees + face; anti-centromere antibodies; pulmonary arterial hypertension risk. Diffuse cutaneous SSc: widespread skin involvement; anti-Scl-70 (anti-topoisomerase I) antibodies; higher risk of ILD, renal crisis, cardiac involvement.
27 Acne Vulgaris Acne/Rosacea
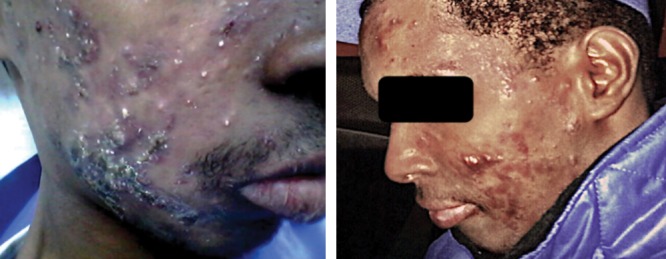
Acne vulgaris is the most common skin condition in the United States, affecting ~85% of adolescents and persisting into adulthood in ~50% of women and ~25% of men. Pathogenesis involves four key factors: (1) androgen-mediated excess sebum production, (2) follicular hyperkeratinization (keratin plugging of the follicular ostium), (3) Cutibacterium acnes (formerly Propionibacterium acnes) colonization and inflammation, and (4) innate immune activation (TLR-2 mediated). These produce the spectrum from non-inflammatory comedones (open/closed) to inflammatory papules, pustules, nodules, and cysts.
Grading
| Grade | Description | Lesion Types |
|---|---|---|
| Mild (comedonal) | Predominantly comedones with few inflammatory lesions | Open/closed comedones, scattered papules |
| Moderate (papulopustular) | More inflammatory lesions, comedones | Papules, pustules, comedones; moderate number of lesions |
| Severe (nodulocystic) | Numerous inflammatory lesions, nodules, cysts | Deep nodules (≥ 5 mm), cysts, widespread papulopustular; risk of scarring |
| Very severe (acne conglobata) | Interconnecting abscesses and sinus tracts | Double/triple comedones, draining sinuses, severe scarring; may be part of follicular occlusion triad (with HS and dissecting cellulitis of scalp) |
Treatment by Severity
| Severity | Topical | Systemic | Notes |
|---|---|---|---|
| Mild comedonal | Retinoid (adapalene 0.1% or tretinoin 0.025-0.1%) +/- benzoyl peroxide (BP) 2.5-10% | — | Retinoid is the cornerstone — normalizes follicular keratinization |
| Mild inflammatory | Retinoid + BP + topical antibiotic (clindamycin 1%) | — | Always combine topical antibiotic with BP to prevent resistance; never use topical antibiotic alone |
| Moderate | Retinoid + BP/antibiotic combination | Oral antibiotic: doxycycline 100 mg BID (first-line) or minocycline 100 mg BID x 3 months max, then reassess | Limit oral antibiotic duration; transition to topical maintenance |
| Severe / Refractory | Adjunctive topicals | Isotretinoin (Accutane/Absorica) 0.5-1 mg/kg/day x 5-6 months (cumulative dose: 120-150 mg/kg) | See isotretinoin details below |
| Hormonal (women) | As above | Oral contraceptive (norgestimate/EE, drospirenone/EE); spironolactone 50-200 mg/day (anti-androgen) | Spironolactone: monitor K+ (hyperkalemia risk), pregnancy test (feminization of male fetus) |
Isotretinoin — Detailed
Isotretinoin (13-cis-retinoic acid) is the only medication that addresses all four pathogenic factors of acne. Dose: 0.5-1.0 mg/kg/day (may start lower to reduce initial flare), total cumulative dose of 120-150 mg/kg to minimize relapse. Take with fatty food (absorption increased 2x). Monitoring: iPLEDGE program (mandatory FDA REMS) — all patients require monthly visits; females of childbearing potential require 2 forms of contraception, monthly pregnancy tests (urine/serum), 1-month negative pregnancy test before starting, and contraception continued 1 month after stopping. Labs: fasting lipid panel and LFTs at baseline, 1 month, and every 1-2 months. Side effects: mucocutaneous dryness (cheilitis ~100%, xerosis, epistaxis, dry eyes — all dose-dependent), myalgias/arthralgias, elevated triglycerides (rarely pancreatitis if TG >500), hepatotoxicity (rare), night vision changes, mood changes (depression/suicidality — contested but requires monitoring); teratogenic — Category X, absolute contraindication in pregnancy.
28 Rosacea Acne/Rosacea
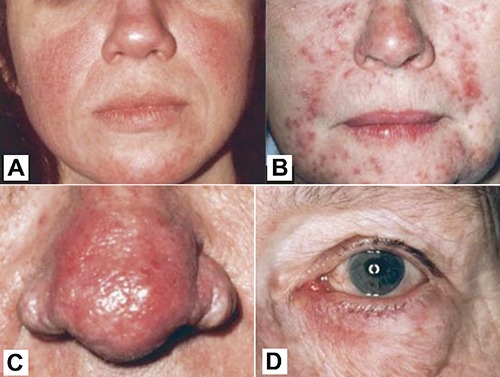
Rosacea is a chronic inflammatory facial dermatosis affecting ~16 million Americans, predominantly fair-skinned individuals aged 30-60. Pathogenesis involves neurovascular dysregulation, innate immune activation (cathelicidin/LL-37 pathway), Demodex folliculorum mite overcolonization, and vascular hyperreactivity. Triggers: heat, sun, alcohol, spicy food, hot beverages, emotional stress.
Rosacea Subtypes (Phenotype-Based Approach Now Preferred)
| Subtype | Features | Treatment |
|---|---|---|
| Erythematotelangiectatic (ETR) | Persistent central facial erythema, flushing, telangiectasias; stinging/burning | Topical brimonidine 0.33% gel (Mirvaso — alpha-2 agonist, reduces erythema within 30 min); oxymetazoline 1% cream (Rhofade); pulsed dye laser or IPL for telangiectasias |
| Papulopustular | Central facial papules and pustules on background erythema; resembles acne but NO comedones | Topical ivermectin 1% cream (Soolantra — anti-Demodex + anti-inflammatory, first-line), topical metronidazole 0.75-1%, topical azelaic acid 15%; oral doxycycline 40 mg MR (Oracea — anti-inflammatory subantimicrobial dose) for moderate-severe |
| Phymatous | Skin thickening, irregular surface, enlarged pores; rhinophyma (nose) most common | Procedural: CO2 laser, electrosurgery, surgical excision for rhinophyma; topicals for inflammatory component |
| Ocular | Eyelid/conjunctival involvement: blepharitis, meibomian gland dysfunction, conjunctivitis, recurrent chalazia; may occur without skin findings | Warm compresses, lid hygiene, topical cyclosporine 0.05% (Restasis), oral doxycycline; ophthalmology referral for severe cases |
29 Hidradenitis Suppurativa Acne/Rosacea
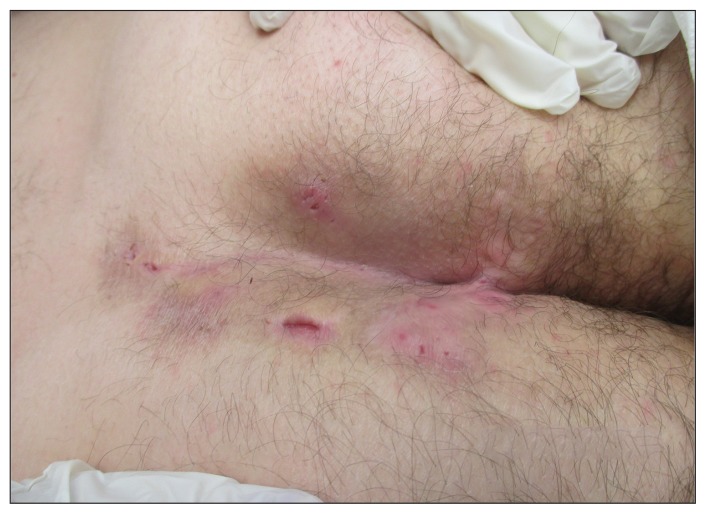
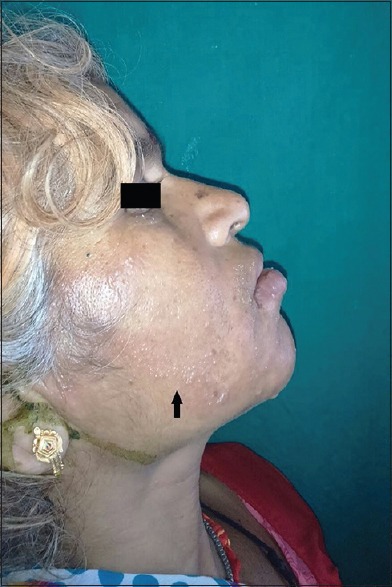
Hidradenitis suppurativa (HS) is a chronic, recurrent, debilitating inflammatory disease of the hair follicle (follicular occlusion disease, not a primary apocrine gland disorder as previously thought). Prevalence: ~1-4%; female predominance 3:1; onset after puberty; strong associations with smoking (75% of patients), obesity, metabolic syndrome, and family history (AD inheritance pattern identified in some families with gamma-secretase mutations). Pathogenesis: follicular hyperkeratinization → occlusion → follicular rupture → intense innate immune response (TNF-alpha, IL-1, IL-17 overexpression) → abscess formation → sinus tract development → fibrosis.
Presentation: painful, deep nodules and abscesses in apocrine gland-bearing skin — axillae, groin, inframammary folds, gluteal cleft, perineum. Recurrent abscesses in the same locations is pathognomonic. Chronic disease produces sinus tracts (interconnecting tunnels), scarring, and contractures. Double comedones (two blackheads connected by a subcutaneous tunnel) are characteristic.
Hurley Staging
| Stage | Description | Management |
|---|---|---|
| I | Abscess formation (single or multiple) without sinus tracts or scarring | Topical clindamycin 1% BID; warm compresses; intralesional triamcinolone (3-5 mg/mL) for acute nodules; combination oral antibiotics: clindamycin 300 mg BID + rifampin 300 mg BID x 10-12 weeks |
| II | Recurrent abscesses with sinus tracts and cicatricial scarring; single or multiple widely separated regions | Above + adalimumab (Humira — only FDA-approved biologic for HS; 160 mg then 80 mg then 40 mg weekly); deroofing of sinus tracts; consider hormonal therapy (spironolactone, finasteride, OCPs) |
| III | Diffuse involvement with interconnecting sinus tracts and abscesses across an entire region | Adalimumab; wide surgical excision of affected area (the only definitive treatment for stage III); secukinumab (recently FDA-approved for HS based on SUNSHINE/SUNRISE trials) |
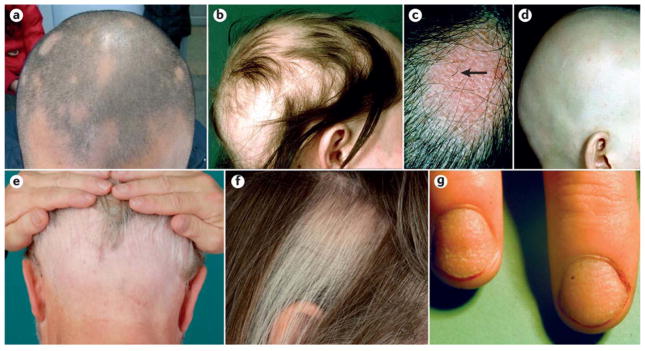
30 Alopecia — Non-Cicatricial & Cicatricial Hair/Nails
Non-Cicatricial (Non-Scarring) Alopecia
| Type | Pathogenesis | Clinical Features | Treatment |
|---|---|---|---|
| Androgenetic alopecia (AGA) | Androgen-mediated (DHT) miniaturization of hair follicles; genetic polygenic inheritance | Males: Hamilton-Norwood pattern (bitemporal recession → vertex thinning → confluence). Females: Ludwig pattern (diffuse thinning of crown with preserved frontal hairline) | Minoxidil 5% topical BID (M) or 2% BID (F); finasteride 1 mg/day PO (M only — 5-alpha-reductase inhibitor, decreases DHT; teratogenic); low-level laser therapy; hair transplant for stable disease; spironolactone 100-200 mg/day for women |
| Alopecia areata (AA) | Autoimmune T-cell attack on hair follicle bulb; associated with thyroid disease, vitiligo, atopy | Well-defined, smooth, round patches of complete hair loss; "exclamation point" hairs (tapered proximal shaft, 3 mm) at periphery; alopecia totalis (entire scalp), alopecia universalis (entire body) | Localized: intralesional triamcinolone 5-10 mg/mL q4-6 weeks; topical clobetasol. Extensive: baricitinib (Olumiant — first FDA-approved oral JAK inhibitor for severe AA, 2022); ritlecitinib (Litfulo, JAK3/TEC inhibitor); contact immunotherapy (DPCP/squaric acid); systemic steroids for acute onset |
| Telogen effluvium | Premature shift of follicles from anagen to telogen (resting phase); triggered by physiologic stress, surgery, high fever, crash dieting, postpartum, medication change | Diffuse shedding (100-300+ hairs/day) starting 2-3 months after trigger; positive hair pull test (>10% telogen hairs); no patches, no scarring | Identify and address trigger; reassurance (self-limited, 6-12 months); iron, vitamin D, thyroid optimization if deficient; no specific drug therapy needed |
| Trichotillomania | Hair-pulling disorder (OCD spectrum); self-inflicted | Irregular patches of broken hairs at varying lengths ("frying pan" pattern on dermoscopy); often frontotemporal; no inflammation or scarring | Behavioral therapy (habit reversal training); N-acetylcysteine 1200-2400 mg/day (modest evidence); SSRIs |
Cicatricial (Scarring) Alopecia
Cicatricial alopecias destroy the hair follicle stem cells in the bulge region, making hair loss permanent. Characterized by loss of follicular ostia (no visible pores in the bald area — distinguishes from non-scarring alopecia). Classification by predominant inflammatory infiltrate: lymphocytic (lichen planopilaris/LPP, frontal fibrosing alopecia/FFA, discoid lupus/DLE, central centrifugal cicatricial alopecia/CCCA) or neutrophilic (dissecting cellulitis, folliculitis decalvans). Diagnosis requires scalp biopsy (two specimens — one for H&E, one for DIF). Frontal fibrosing alopecia (FFA) — a variant of LPP — presents with progressive frontotemporal hairline recession and eyebrow loss, predominantly in postmenopausal women; increasing in prevalence. Treatment goal is to halt progression (regrowth unlikely): hydroxychloroquine, doxycycline, finasteride/dutasteride, topical/intralesional steroids.
31 Nail Disorders Hair/Nails
| Finding | Description | Associated Conditions |
|---|---|---|
| Onycholysis | Distal separation of nail plate from nail bed | Psoriasis, onychomycosis, trauma, thyroid disease, contact irritant |
| Pitting | Small depressions in nail plate (punctate) | Psoriasis (#1), alopecia areata, eczema, lichen planus |
| Oil drop sign | Yellow-brown discoloration of nail bed visible through nail plate | Psoriasis (salmon/oil-drop patches — highly specific) |
| Beau lines | Transverse grooves across nail plate | Any systemic illness, surgery, or stress that temporarily arrests nail matrix growth |
| Splinter hemorrhages | Longitudinal red-brown lines in nail bed | Trauma (most common), endocarditis, vasculitis, antiphospholipid syndrome |
| Koilonychia | Spoon-shaped concave nails | Iron deficiency anemia, hemochromatosis, normal in infants |
| Terry nails | White nail with distal pink-brown band (1-2 mm) | Cirrhosis, CHF, diabetes |
| Muehrcke lines | Paired horizontal white bands that do not move with nail growth | Hypoalbuminemia (nephrotic syndrome, liver disease) |
| Half-and-half nails (Lindsay) | Proximal white, distal brown (each ~50%) | Chronic kidney disease/uremia |
| Yellow nail syndrome | Yellow, thickened, slow-growing nails + lymphedema + pleural effusions | Classically a triad; associated with lymphatic dysfunction |
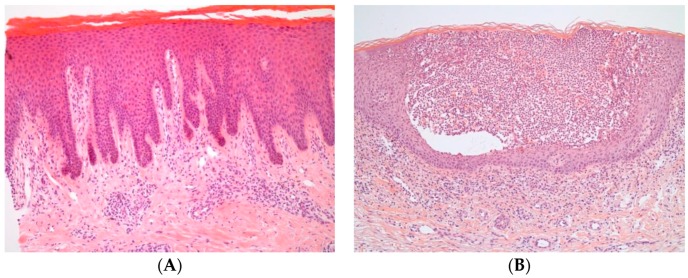
Melanonychia
Longitudinal melanonychia — a brown-black longitudinal streak in the nail — requires careful evaluation because it may represent benign nail matrix melanocyte activation (common in dark-skinned individuals, where longitudinal melanonychia is a normal variant), subungual melanocytic nevus, or subungual melanoma. Concerning features: new onset in a light-skinned adult, single digit (especially thumb or great toe), width >3 mm, irregular borders, pigment changes, Hutchinson sign (periungual pigmentation extending onto nail fold — concerning for melanoma), nail dystrophy, and personal history of melanoma. Nail matrix biopsy is required for suspicious lesions — a longitudinal excisional biopsy of the pigmented band from the nail matrix is the standard technique.
32 Vitiligo Pigmentary
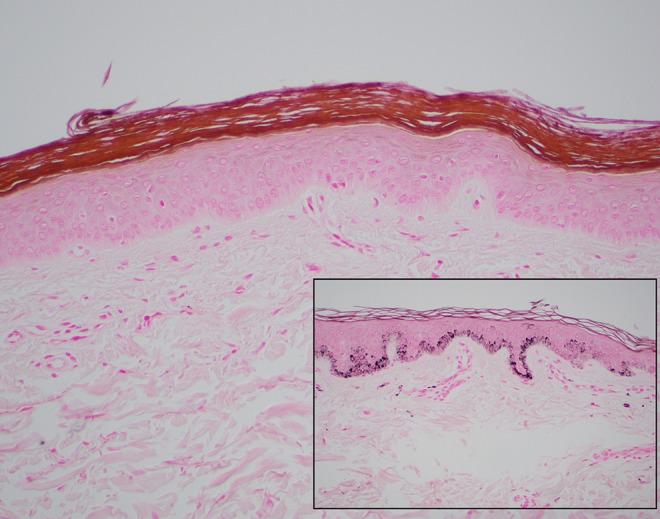
Vitiligo is an acquired autoimmune depigmenting disorder caused by CD8+ T-cell-mediated destruction of melanocytes, affecting ~1% of the population worldwide. It presents as well-demarcated, chalky-white macules and patches (total absence of melanin, unlike hypopigmentation where some melanin remains). Onset: typically before age 30; associated with other autoimmune diseases (thyroid — screen with TSH/free T4, type 1 DM, pernicious anemia, Addison disease, alopecia areata). Types: non-segmental (generalized) — most common (~90%), symmetric, progressive, often periorificial (around eyes, mouth, genitals), acral; segmental — unilateral, dermatomal, earlier onset, more stable, less associated with autoimmunity, responds better to surgical therapies.
Diagnosis is clinical — enhanced by Wood lamp examination (365 nm UV light): vitiligo fluoresces bright white/blue-white, making subtle lesions visible, especially in fair-skinned patients. DDx: pityriasis alba (ill-defined, hypopigmented, slightly scaly patches in children — associated with AD), tinea versicolor (fine scale, KOH positive), post-inflammatory hypopigmentation (history of prior inflammation), piebaldism (congenital, stable, white forelock). Treatment: ruxolitinib 1.5% cream (Opzelura) — first FDA-approved topical for vitiligo (2022); JAK1/2 inhibitor that blocks the IFN-gamma/CXCL10 signaling that drives melanocyte destruction. Other topicals: tacrolimus 0.1%, pimecrolimus 1% (preferred for face), high-potency TCS for body. NB-UVB phototherapy (311 nm) 2-3x/week is the most effective non-surgical therapy for widespread disease — produces repigmentation (from perifollicular melanocyte reservoirs) in ~50-70% of patients over 6-12 months. Surgical: autologous melanocyte-keratinocyte transplant or suction blister epidermal grafting for stable segmental or localized vitiligo refractory to medical therapy.
33 Melasma & Post-Inflammatory Hyperpigmentation Pigmentary
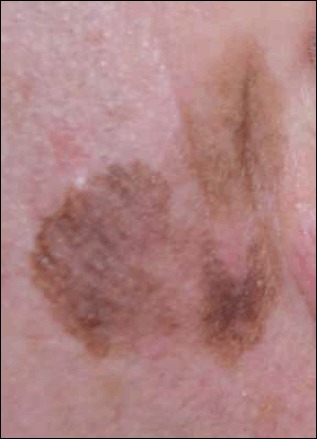
Melasma
Melasma is a common acquired disorder of hyperpigmentation characterized by symmetric, brown-to-gray patches on the face — centrofacial (forehead, cheeks, nose, upper lip, chin) is the most common pattern; malar and mandibular patterns also occur. Predominantly affects women (Fitzpatrick III-V), triggered by UV exposure, hormonal influences (pregnancy, oral contraceptives, HRT), and genetic predisposition. Three histologic types: epidermal (brown, well-defined, enhanced by Wood lamp — better treatment response), dermal (blue-gray, ill-defined, not enhanced by Wood lamp — more resistant), and mixed. Treatment: triple combination cream (hydroquinone 4% + tretinoin 0.05% + fluocinolone acetonide 0.01% — Tri-Luma) is the gold standard topical; apply nightly x 8-12 weeks. Strict photoprotection is essential (tinted sunscreen with iron oxide blocks visible light, which triggers melasma in dark skin types). Alternatives: azelaic acid 15-20%, tranexamic acid (oral 250 mg BID or topical — inhibits plasmin-mediated melanogenesis), vitamin C serum, cysteamine 5% cream, chemical peels (glycolic acid). Hydroquinone should be cycled (3-6 months on, 2-3 months off) to prevent ochronosis (paradoxical blue-gray discoloration with prolonged use).
Post-Inflammatory Hyperpigmentation (PIH)
PIH is acquired hyperpigmentation following cutaneous inflammation or injury (acne, eczema, trauma, procedures). More common and more persistent in darker skin types (Fitzpatrick III-VI). Epidermal PIH (brown): melanin increased in epidermis — responds to topical lightening agents. Dermal PIH (blue-gray): melanin dropped into dermis (melanin incontinence/melanophages) — slower to resolve, resistant to treatment. Management: treat the underlying inflammatory condition first (to prevent new PIH), photoprotection, topical retinoids, azelaic acid, vitamin C, hydroquinone 2-4%, chemical peels (caution in dark skin — aggressive peels can worsen PIH). Time is often the most effective treatment — most epidermal PIH resolves in 6-12 months; dermal PIH can take years.
34 Morbilliform Drug Eruptions & Urticaria Emergencies
Morbilliform (Exanthematous) Drug Eruption
The most common cutaneous drug reaction (~95% of drug rashes), occurring 7-14 days after drug initiation (or 1-3 days on re-exposure). Presents as widespread, symmetric, erythematous macules and papules that may coalesce — starts on the trunk and spreads peripherally. Pruritic. Mucous membranes spared. No systemic symptoms (distinguishes from DRESS and SJS/TEN). Most common culprits: antibiotics (amoxicillin, TMP-SMX, penicillins, cephalosporins), anticonvulsants (carbamazepine, phenytoin, lamotrigine), allopurinol, NSAIDs. The classic "ampicillin rash" in EBV mononucleosis is not a true drug allergy — it is an immune-mediated reaction that does not predict future penicillin allergy. Management: discontinue offending drug, supportive care (emollients, antihistamines, mild-to-mid potency TCS); resolves 1-2 weeks after drug withdrawal.
Urticaria (Hives)
Wheals (transient, pruritic, edematous papules and plaques) that individually last <24 hours and resolve without bruising. If an individual wheal lasts >24 hours or leaves bruising, consider urticarial vasculitis (biopsy needed). Acute urticaria (<6 weeks): usually identifiable trigger — drugs, foods, infections. Chronic urticaria (≥6 weeks): ~80% are chronic spontaneous urticaria (CSU) with no identifiable trigger; autoimmune etiology in ~30-50% (IgG anti-FceRI or anti-IgE antibodies). Treatment: second-generation H1 antihistamines (cetirizine, loratadine, fexofenadine) at standard then up to 4x standard dose → add H2 blocker (famotidine) or LTRA (montelukast) → omalizumab (Xolair), anti-IgE monoclonal antibody, 150-300 mg SC q4 weeks (FDA-approved for CSU refractory to antihistamines) → cyclosporine for refractory CSU. Avoid systemic corticosteroids for chronic urticaria — short courses only for severe flares.
35 Stevens-Johnson Syndrome / Toxic Epidermal Necrolysis Emergencies
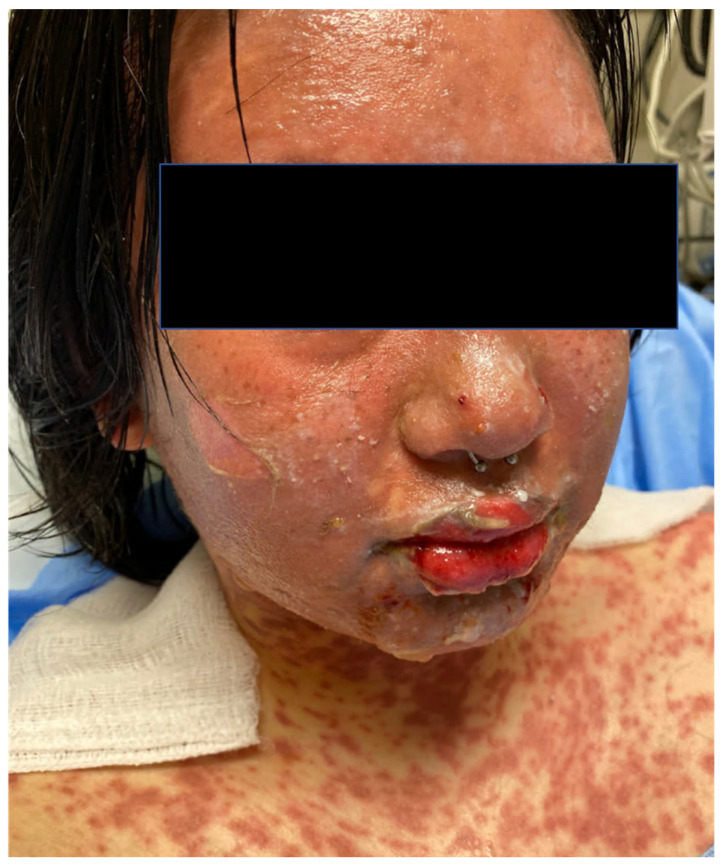
SJS and TEN are life-threatening mucocutaneous reactions characterized by extensive keratinocyte apoptosis mediated by cytotoxic T cells and FasL/granulysin. They exist on a spectrum: SJS = <10% BSA detachment; SJS/TEN overlap = 10-30% BSA; TEN = >30% BSA. Mortality: SJS ~5-10%; TEN ~25-35%. Nearly always drug-induced (onset 1-3 weeks after drug initiation). Highest-risk drugs: allopurinol (screen for HLA-B*5801 in high-risk populations — Southeast Asian, African American), carbamazepine (screen for HLA-B*1502 in Southeast Asian populations), lamotrigine, phenytoin, sulfonamides (TMP-SMX), nevirapine, piroxicam (Mockenhaupt et al., J Invest Dermatol 2008).
Presentation: prodromal fever and malaise → painful skin tenderness → erythematous-to-dusky macules, often with targetoid morphology (atypical targets), coalescing into patches → full-thickness epidermal necrosis with bullae → positive Nikolsky sign (epidermal detachment with lateral pressure). Mucous membrane involvement (≥2 sites: oral, ocular, genital, respiratory, GI) is nearly universal and is a key distinguishing feature from other drug eruptions. Ocular involvement can cause synechiae, corneal scarring, and permanent vision loss — urgent ophthalmology involvement is mandatory.
SCORTEN — Severity-of-Illness Score for TEN
| Parameter | Criterion (1 point each) |
|---|---|
| Age | ≥ 40 years |
| Malignancy | Present (active) |
| Heart rate | ≥ 120 bpm |
| BSA detached (day 1) | ≥ 10% |
| Serum BUN | > 28 mg/dL (10 mmol/L) |
| Serum glucose | > 252 mg/dL (14 mmol/L) |
| Serum bicarbonate | < 20 mEq/L |
| SCORTEN | Predicted Mortality |
|---|---|
| 0-1 | 3.2% |
| 2 | 12.1% |
| 3 | 35.3% |
| 4 | 58.3% |
| ≥ 5 | > 90% |
Management: immediate discontinuation of the causative drug (early withdrawal reduces mortality), transfer to burn unit or ICU, aggressive supportive care (wound care, fluid/electrolyte management, pain control, nutritional support, infection surveillance — do NOT give prophylactic antibiotics), ophthalmology and urology consults. Specific therapies: cyclosporine 3-5 mg/kg/day has the strongest emerging evidence for mortality benefit; IVIG is commonly used but evidence is mixed; systemic corticosteroids are controversial (some data suggest harm). Etanercept (anti-TNF) shows promise in recent trials (Wang et al., JAMA Dermatol 2021).
36 DRESS Syndrome, Erythema Multiforme & AGEP Emergencies
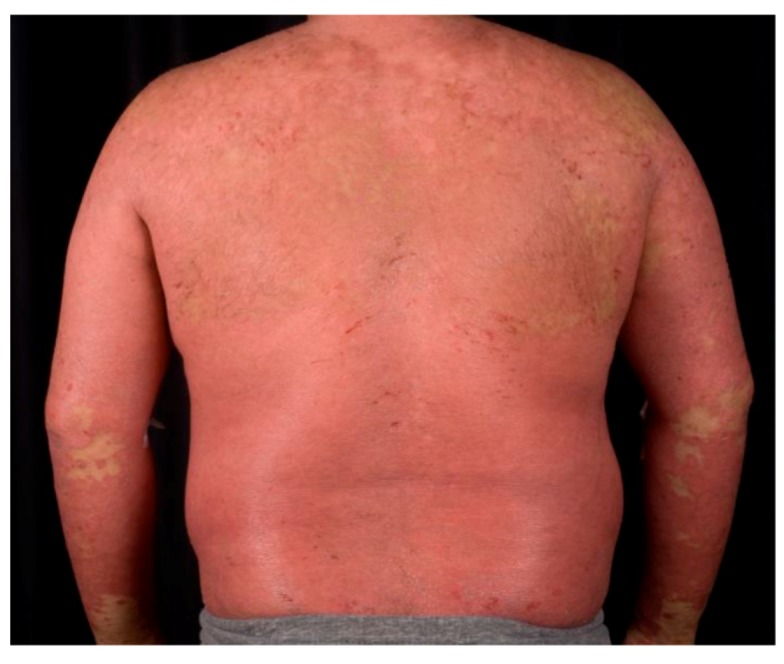
DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms)
Also known as drug-induced hypersensitivity syndrome (DIHS). Onset: 2-8 weeks after drug initiation (longer latency than morbilliform eruption). Hallmarks: widespread morbilliform or erythrodermic eruption + facial edema (especially periorbital) + fever + lymphadenopathy + hematologic abnormalities (eosinophilia ≥ 1,500/mcL and/or atypical lymphocytosis) + internal organ involvement (liver 60-80% — monitor AST/ALT, kidneys 10-30%, lungs, myocarditis, thyroiditis). Highest-risk drugs: anticonvulsants (carbamazepine, phenytoin, lamotrigine, phenobarbital — cross-reactivity among aromatic anticonvulsants ~40%), allopurinol, dapsone, minocycline, vancomycin, TMP-SMX. Strong association with HHV-6 reactivation. RegiSCAR scoring system validates diagnosis. Mortality: ~5-10%. Treatment: withdraw offending drug immediately; systemic corticosteroids (prednisone 1-1.5 mg/kg/day, taper slowly over 8-12 weeks — rapid taper risks rebound; steroid-associated HHV-6 reactivation must be monitored). Late sequelae: autoimmune thyroiditis (monitor TSH for 1 year) and type 1 DM can develop weeks to months after recovery.
Erythema Multiforme (EM)
EM is an acute, self-limited, immune-mediated reaction most commonly triggered by HSV infection (~90% of cases — not drugs). Classic lesion: target lesion (targetoid) — three concentric zones: dusky/necrotic center, pale edematous ring, erythematous outer ring — predominantly on the extremities (acral, symmetric), palms and soles. EM minor: skin only, limited mucous membrane involvement; EM major: more extensive mucous membrane involvement (oral, genital). EM is NOT on the SJS/TEN spectrum — different etiology (infection-triggered vs drug-induced), different target lesion morphology (true targets vs atypical targets in SJS), and different prognosis (EM recurs but has minimal mortality). Recurrent EM: triggered by recurrent HSV — suppressive valacyclovir 500 mg-1 g daily prevents recurrences.
AGEP (Acute Generalized Exanthematous Pustulosis)
Acute onset (within 48-96 hours of drug exposure, much faster than DRESS) of dozens to hundreds of small, non-follicular, sterile pustules on widespread erythematous skin, often starting in intertriginous areas. High fever, neutrophilia (not eosinophilia — distinguishes from DRESS). Most common triggers: aminopenicillins (ampicillin, amoxicillin), macrolides, quinolones, terbinafine, diltiazem, hydroxychloroquine. Histology: subcorneal/intraepidermal spongiform pustules with neutrophilic infiltrate and papillary dermal edema. Treatment: withdraw the causative drug; typically self-limited with spontaneous resolution in 1-2 weeks (pustules desquamate); supportive care; mortality <5% (usually in elderly with comorbidities).
37 Cutaneous T-Cell Lymphoma (Mycosis Fungoides & Sezary Syndrome) Special
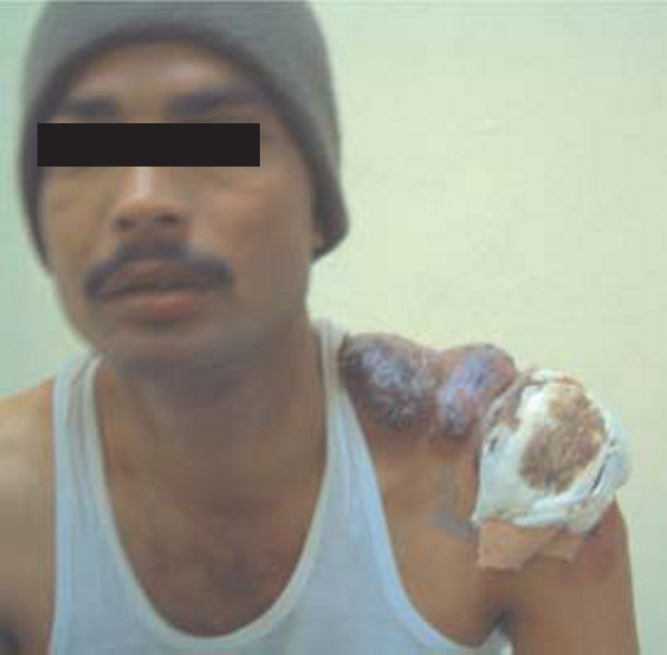
Cutaneous T-cell lymphoma (CTCL) is a group of non-Hodgkin lymphomas that primarily manifest in the skin. Mycosis fungoides (MF) is the most common type (~50% of all primary cutaneous lymphomas). It is a malignancy of skin-homing CD4+ T cells and progresses through characteristic clinical stages:
| Stage | Clinical Features | Key Points |
|---|---|---|
| Patch stage | Well-defined, erythematous, slightly scaly patches; often in "bathing trunk" (buttocks, trunk) or non-sun-exposed distribution | May mimic eczema, psoriasis, or tinea for years ("premycotic" phase); biopsy often non-diagnostic early — repeat if clinical suspicion persists; epidermotropism (atypical lymphocytes migrating into epidermis) is characteristic |
| Plaque stage | Raised, indurated, erythematous plaques; may be annular or horseshoe-shaped | Pautrier microabscesses (clusters of atypical lymphocytes within the epidermis) on histology; immunophenotype: CD3+, CD4+, CD8-, CD7 loss |
| Tumor stage | Large nodules and tumors, often ulcerated ("d'Emblee" variant: tumors without preceding patches/plaques) | Large cell transformation may occur (aggressive behavior, poor prognosis) |
| Erythrodermic MF / Sezary syndrome | Generalized erythroderma (>80% BSA); Sezary: erythroderma + circulating Sezary cells (cerebriform/convoluted nuclei) in blood ≥1,000/mcL + lymphadenopathy | Sezary syndrome is leukemic variant; intense pruritus; immunosuppression with infection susceptibility |
TNMB Staging (ISCL/EORTC)
MF/Sezary staging uses a modified TNMB system: T (skin involvement — T1 patches/plaques <10% BSA; T2 ≥10% BSA; T3 tumors; T4 erythroderma), N (lymph node involvement), M (visceral involvement), B (blood involvement — B0 <5% Sezary cells; B1 ≥5% but <1,000/mcL; B2 ≥1,000/mcL). Stage IA (T1N0M0B0) has excellent prognosis (median survival similar to age-matched controls); stage IVB (visceral involvement) has median survival ~1-2 years.
Treatment: early-stage (IA-IIA) — skin-directed therapies: topical corticosteroids, topical nitrogen mustard (mechlorethamine 0.02% gel — Valchlor), NB-UVB or PUVA phototherapy, localized radiation. Advanced-stage (IIB-IV) — systemic therapies: bexarotene (Targretin — RXR retinoid agonist), vorinostat (Zolinza — HDAC inhibitor), romidepsin (Istodax — HDAC inhibitor), brentuximab vedotin (Adcetris — anti-CD30), mogamulizumab (Poteligeo — anti-CCR4), extracorporeal photopheresis (ECP — particularly for Sezary syndrome), low-dose methotrexate, interferons (IFN-alpha). Allogeneic stem cell transplant is the only potentially curative therapy for advanced CTCL.
38 Granuloma Annulare & Pyoderma Gangrenosum Special
Granuloma Annulare (GA)
GA is a benign, self-limited granulomatous dermatosis of unclear etiology. Classic presentation: annular or arcuate, flesh-colored to erythematous, non-scaly, smooth plaques and papules on the dorsal hands and feet. The lack of scale distinguishes it from tinea corporis (KOH negative). Histology: palisading granuloma with central mucin deposition (Alcian blue positive). Subtypes: localized (most common, self-limited), generalized (widely distributed, may be associated with diabetes mellitus), subcutaneous (deep nodules, especially in children — DDx: rheumatoid nodule), perforating (papules with central crust from transepidermal elimination of degenerated collagen). Treatment: often unnecessary (localized resolves in 50% within 2 years); high-potency TCS, intralesional triamcinolone, cryotherapy; generalized GA: phototherapy (NB-UVB, PUVA), dapsone, hydroxychloroquine, methotrexate.
Pyoderma Gangrenosum (PG)
PG is a neutrophilic dermatosis presenting as a rapidly progressive, painful ulcer with violaceous, undermined borders and a purulent/necrotic base. Classic form begins as a sterile pustule or papule that rapidly expands into a deep ulcer. The key concept is pathergy — new lesions (or worsening of existing lesions) triggered by trauma, including surgical debridement, which is why debridement is contraindicated (a common pitfall). Diagnosis is clinical and histologic (neutrophilic infiltrate, ruling out infection and malignancy — PG is a diagnosis of exclusion). Associated with systemic diseases in 50-70%: inflammatory bowel disease (UC > Crohn), rheumatoid arthritis, hematologic malignancies (MDS, AML, myeloma), and monoclonal gammopathy (IgA). Treatment: high-dose systemic corticosteroids (prednisone 1 mg/kg/day), cyclosporine 3-5 mg/kg/day, TNF-alpha inhibitors (infliximab), topical tacrolimus for small/stable lesions; wound care with non-adherent dressings; AVOID debridement (pathergy risk).
39 Sweet Syndrome & Dermatologic Signs of Systemic Disease Special
Sweet Syndrome (Acute Febrile Neutrophilic Dermatosis)
Sweet syndrome presents with acute onset of painful, edematous, erythematous-to-violaceous papules, plaques, and nodules — classically on the face, neck, and upper extremities — accompanied by fever, neutrophilia, and elevated ESR/CRP. Histology: dense dermal neutrophilic infiltrate without vasculitis (distinguishes from leukocytoclastic vasculitis). Three categories: classical (triggered by URIs, GI infections, or associated with IBD, pregnancy), malignancy-associated (hematologic malignancies, especially AML and MDS — screen with CBC, peripheral smear, bone marrow biopsy if abnormal), and drug-induced (G-CSF, all-trans retinoic acid, TMP-SMX). Treatment: systemic corticosteroids produce dramatic rapid response (prednisone 0.5-1 mg/kg/day — improvement within 48 hours is almost diagnostic); steroid-sparing options include dapsone, colchicine, and potassium iodide.
Dermatologic Signs of Systemic Disease
| Skin Finding | Associated Systemic Disease |
|---|---|
| Acanthosis nigricans — velvety, hyperpigmented plaques in skin folds (neck, axillae) | Insulin resistance/type 2 DM (most common); malignant AN (rapid onset, extensive, involving palms/oral mucosa) → search for GI malignancy (especially gastric adenocarcinoma) |
| Xanthomas — eruptive, tendinous, tuberous, xanthelasma | Hyperlipidemia; eruptive xanthomas = hypertriglyceridemia (>1,000); tendinous xanthomas = familial hypercholesterolemia |
| Necrolytic migratory erythema — annular, erythematous, erosive plaques (groin, perineum, perioral) | Glucagonoma (pancreatic alpha-cell tumor); part of glucagonoma syndrome (DM, weight loss, DVT, anemia, glossitis) |
| Erythema nodosum — tender, red-purple nodules on shins (septal panniculitis) | Sarcoidosis, streptococcal infection, IBD, TB, medications (OCPs, sulfonamides); most common panniculitis |
| Palpable purpura — raised, non-blanching purpuric papules, typically on lower extremities | Leukocytoclastic vasculitis (small vessel); IgA vasculitis (Henoch-Schonlein), drug-induced, infection-related, connective tissue disease, cryoglobulinemia |
| Livedo reticularis — reticular (net-like) violaceous discoloration | Physiologic (cutis marmorata), antiphospholipid syndrome (livedo racemosa — irregular, broken net), cholesterol emboli, vasculitis, SLE |
| Leser-Trelat sign — sudden eruption of multiple seborrheic keratoses | Internal malignancy (GI, lymphoma); controversial — may be coincidental given high prevalence of SK in elderly |
| Dermatitis herpetiformis | Celiac disease (see Section 23) |
| Janeway lesions / Osler nodes | Infective endocarditis (Janeway: painless, palms/soles; Osler: painful, fingertips/toes) |
40 Skin Biopsy Techniques Procedures
Skin biopsy is the most fundamental diagnostic procedure in dermatology. Biopsy site selection is critical — biopsy the most representative, well-developed lesion (not the oldest or most excoriated). For vesiculobullous diseases, biopsy the edge of a fresh blister (not the blister roof or center); for DIF, biopsy perilesional uninvolved skin.
| Technique | Method | Best For | Limitations |
|---|---|---|---|
| Shave biopsy | Flexible blade or DermaBlade removes superficial tissue (epidermis + papillary dermis) tangentially; depth controlled by blade angle | Epidermal lesions: AK, superficial BCC, SK, skin tags; quick, minimal scarring, no sutures | Insufficient depth for melanoma (may transect base, limiting Breslow measurement), deep inflammatory diseases, panniculitis, or subcutaneous pathology |
| Punch biopsy | Cylindrical blade (2-8 mm; most commonly 4 mm) rotated through full skin thickness (epidermis → dermis → superficial subcutis) | Inflammatory dermatoses, deep dermal processes, melanocytic lesions requiring full-thickness assessment; standard diagnostic biopsy for most conditions | Small sample may not capture architecture of large tumors; not adequate for alopecia workup (need 2 punches — one horizontal, one for DIF) |
| Excisional biopsy | Elliptical excision of the entire lesion with margins (3:1 length-to-width ratio, oriented along skin tension lines/Langer lines) | Suspicious melanocytic lesions (preferred for melanoma — provides entire lesion with accurate Breslow depth); lipomas; cysts | Requires suturing; larger scar; more time-consuming |
| Incisional biopsy | Partial sampling of a large lesion (wedge or punch from representative area) | Large lesions where excision is impractical; panniculitis (must include subcutaneous fat — deep incisional or 6-8 mm punch) | Partial sampling may miss focal features |
41 Mohs Micrographic Surgery Procedures
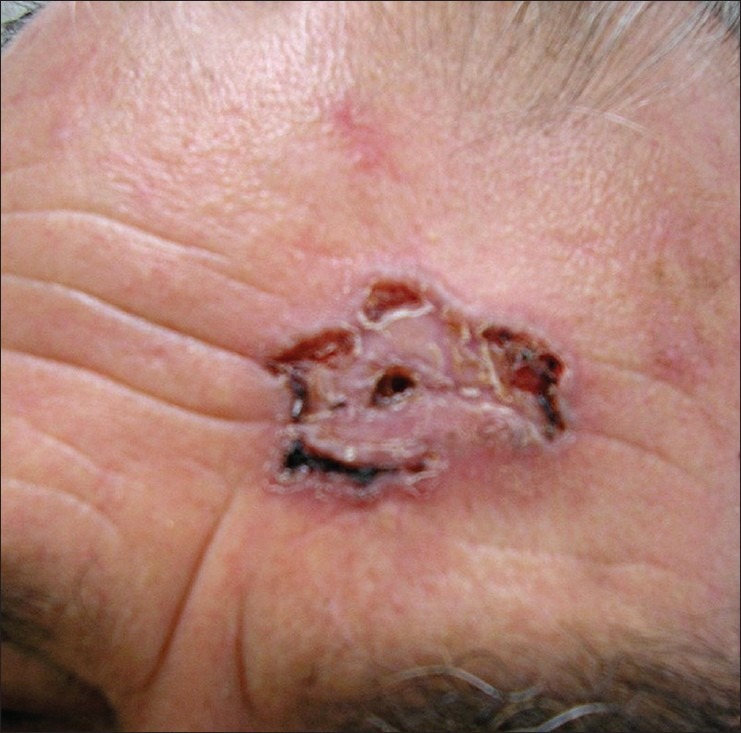
Mohs micrographic surgery (MMS) is the gold standard for skin cancer removal in high-risk settings. The technique, developed by Frederic Mohs, achieves the highest cure rate (99% for primary BCC, 95% for recurrent BCC) with maximal tissue conservation — critical on the face where every millimeter of tissue preservation matters.
Procedure
The visible tumor is debulked. A thin layer of tissue is excised around and beneath the defect. The tissue is mapped, color-coded, frozen sectioned, and examined under microscopy by the Mohs surgeon — 100% of the peripheral and deep margins are examined (vs <1% in standard "bread-loaf" histologic sectioning). If residual tumor is identified in any area, that specific area is re-excised and the process is repeated. The cycle continues until all margins are clear, then the defect is reconstructed (primary closure, flap, graft, or second-intention healing depending on size and location).
Appropriate Use Criteria (AUC) — Indications for Mohs
| Factor | Details |
|---|---|
| Location | H-zone (central face, eyelids, nose, lips, ears, temples, periauricular); genitalia; hands/feet; pretibial |
| Tumor features (BCC) | Aggressive subtypes (morpheaform, infiltrative, micronodular, basosquamous); recurrent; large (>2 cm on trunk, >1 cm on face); perineural invasion; poorly defined clinical margins |
| Tumor features (SCC) | High-risk per NCCN criteria (see Section 14); perineural invasion; poorly differentiated |
| Patient factors | Immunosuppression (organ transplant, CLL); genetic syndromes (Gorlin, xeroderma pigmentosum); prior radiation to site |
| Other tumors treated by Mohs | Dermatofibrosarcoma protuberans (DFSP), extramammary Paget disease, microcystic adnexal carcinoma, sebaceous carcinoma, lentigo maligna (melanoma in situ — modified Mohs with immunostains) |
42 Cryotherapy, ED&C & Phototherapy Procedures
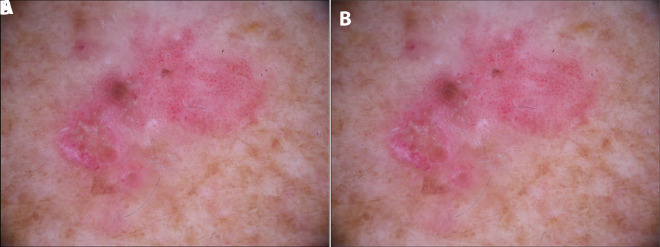
Cryotherapy (Liquid Nitrogen)
Liquid nitrogen (LN2, -196 degrees C) is the most commonly performed procedure in dermatology. Mechanism: rapid freeze → ice crystal formation in cells → thaw → cell membrane disruption → necrosis. Applied via spray gun (most common), cotton-tipped applicator, or cryoprobe. Freeze time varies by lesion: 5-10 seconds for AK, 10-15 seconds for verruca, 20-30 seconds for BCC (superficial). Double freeze-thaw cycle (freeze → thaw → freeze again) is more effective for malignant/premalignant lesions. Side effects: pain, blister formation (expected), dyspigmentation (hypopigmentation common — caution in dark skin), hair loss in treated area. Contraindications: cryoglobulinemia, cryofibrinogenemia, cold urticaria, Raynaud phenomenon in the area, poor circulation.
Electrodesiccation & Curettage (ED&C)
A destructive technique for low-risk BCC and SCC (see Sections 13-14 for appropriate selection criteria). Procedure: local anesthesia → curette the tumor (scrapes soft tumor tissue preferentially, healthy dermis resists curettage — the "feel" of resistance indicates adequate removal) → electrodesiccate the base → repeat 2-3 total cycles. Heals by secondary intention over 4-8 weeks. Produces a round, atrophic, hypopigmented scar. Not appropriate for high-risk tumors, recurrent tumors, or high-risk anatomic sites (face/H-zone — poor cosmetic outcome and limited margin assessment).
Phototherapy
| Modality | Wavelength | Mechanism | Indications | Key Points |
|---|---|---|---|---|
| NB-UVB | 311 nm | Induces T-cell apoptosis, suppresses local immune response, promotes melanocyte stimulation | Psoriasis, vitiligo, atopic dermatitis, MF (early), pruritus (uremic, polycythemia vera) | Most commonly used; 2-3x/week; cumulative dose tracked; no psoralen required; safer than PUVA for most indications |
| PUVA | UVA (320-400 nm) + oral psoralen (8-MOP) | Psoralen intercalates DNA → crosslinks with UVA → inhibits DNA synthesis → antiproliferative and immunosuppressive | Severe psoriasis, MF, severe atopic dermatitis, vitiligo (where NB-UVB fails) | Higher efficacy but greater photosensitivity (wear UVA-blocking glasses 24h post-treatment), nausea from psoralen, increased skin cancer risk with cumulative treatments (>200 sessions) |
| Excimer laser | 308 nm | Focused NB-UVB delivered to individual plaques | Localized psoriasis, vitiligo patches | Treats only affected skin, sparing normal tissue; fewer sessions needed; good for scalp, palmoplantar psoriasis |
| UVA1 | 340-400 nm (long UVA) | Penetrates deeper; induces fibroblast collagenase, T-cell apoptosis in dermis | Morphea/localized scleroderma, atopic dermatitis, mastocytosis | Best for sclerotic/fibrotic conditions; limited availability |
43 Intralesional Injections & Procedural Therapeutics Procedures

Intralesional Corticosteroid Injections
Triamcinolone acetonide (Kenalog) is the workhorse intralesional corticosteroid. Concentration varies by indication: 2.5-5 mg/mL for alopecia areata (scalp), 5-10 mg/mL for inflammatory acne nodules/cysts and keloids, 10-20 mg/mL for hypertrophic scars, 20-40 mg/mL for thick keloids. Injected directly into the lesion using a 30-gauge needle. Complications: dermal/subcutaneous atrophy (depressed scar — usually reversible over months), hypopigmentation (especially in dark skin), telangiectasia. Maximum dose per session is generally limited to avoid systemic absorption (typically <40-60 mg total). For HS nodules: intralesional triamcinolone 3-5 mg/mL provides rapid relief within 24-48 hours.
5-Fluorouracil (Intralesional)
Intralesional 5-FU (50 mg/mL, often mixed with triamcinolone) is used for hypertrophic scars and keloids — the antimetabolite inhibits fibroblast proliferation while the steroid suppresses inflammation. Inject q2-4 weeks; often combined with cryotherapy (triple therapy: cryo + intralesional steroid + 5-FU has best evidence for keloid reduction).
Bleomycin (Intralesional)
Intralesional bleomycin (1 U/mL) is used for recalcitrant warts. Injected directly into the wart or applied via bifurcated needle prick technique. Causes local necrosis of HPV-infected cells. Contraindicated in pregnancy and immunosuppression. Painful injection. Cure rates: 60-90% for common and plantar warts.
Chemical Peels
Chemical peeling agents by depth: superficial (glycolic acid 30-70%, salicylic acid 20-30%, Jessner solution, TCA 10-25% — epidermis only; good for acne, melasma, PIH), medium (TCA 35-50%, combination Jessner + TCA 35% — through papillary dermis; for actinic damage, moderate wrinkles), deep (phenol/Baker-Gordon formula — through reticular dermis; for severe photodamage, deep wrinkles; risk of cardiac arrhythmia from phenol absorption — requires cardiac monitoring). Post-peel: strict sun avoidance, gentle skin care, expect desquamation proportional to peel depth.
44 Classification Systems (All)
| System | Used For | Key Elements |
|---|---|---|
| Fitzpatrick Skin Phototype | Skin sun-sensitivity classification | I (always burns, never tans) → II (burns easily, tans minimally) → III (sometimes burns, tans uniformly) → IV (burns minimally, tans easily) → V (rarely burns, tans darkly) → VI (never burns, deeply pigmented) |
| ABCDE Criteria | Melanoma clinical screening | Asymmetry, Border irregularity, Color variation, Diameter >6 mm, Evolution |
| Breslow Depth | Melanoma prognosis / staging | Millimeter measurement from granular layer to deepest melanoma cell; single most important prognostic factor |
| Clark Level | Melanoma depth of invasion (historical) | I (epidermis/in situ), II (papillary dermis), III (fills papillary dermis), IV (reticular dermis), V (subcutis) |
| AJCC 8th Ed TNM — Melanoma | Melanoma staging | T1-T4 by Breslow depth and ulceration; N by regional LN involvement; M by distant metastasis and LDH |
| AJCC 8th Ed TNM — cSCC | Cutaneous SCC staging (H&N) | T1-T4 by size, bone invasion, PNI; see Section 14 |
| SCORAD | Atopic dermatitis severity | BSA + intensity (6 signs, each 0-3) + subjective symptoms (pruritus, sleep loss); 0-103; mild <25, moderate 25-50, severe >50 |
| EASI | Atopic dermatitis severity (trials) | 4 body regions × 4 signs (0-3) × BSA weighting; 0-72; EASI-75 = primary trial endpoint |
| PASI | Psoriasis severity (trials) | 4 body regions × 3 signs (0-4) × BSA (0-6); 0-72; PASI 75/90/100 standard endpoints |
| Hurley Staging | Hidradenitis suppurativa | I (abscesses, no tracts/scarring), II (recurrent abscesses, tracts, scarring), III (diffuse involvement, interconnecting tracts) |
| SCORTEN | SJS/TEN mortality prediction | 7 parameters (age, malignancy, HR, BSA, BUN, glucose, bicarbonate); score 0-7; mortality 3.2% to >90% |
| Hamilton-Norwood Scale | Male pattern hair loss | Types I-VII; I = minimal/no recession; VII = extensive loss with only band remaining |
| Ludwig Scale | Female pattern hair loss | I (thinning on crown), II (pronounced thinning), III (total loss on crown) |
| Wound Staging / Wagner | Diabetic foot ulcers | 0 (pre-ulcerative) through 5 (whole-foot gangrene); used in dermatology wound clinics |
| TNMB (ISCL/EORTC) | Mycosis fungoides / Sezary | T (skin: T1-T4), N (nodes), M (viscera), B (blood: B0-B2) |
| LRINEC Score | Necrotizing fasciitis likelihood | CRP, WBC, Hgb, Na, Cr, glucose; ≥6 suggestive; ≥8 strongly predictive |
| RegiSCAR Score | DRESS diagnosis validation | Hospitalization, drug reaction suspected, fever, eosinophilia, atypical lymphocytes, skin involvement, organ involvement, resolution >15 days; possible/probable/definite |
45 Medications Master Table Meds
Topical Corticosteroids by Potency Class (US System)
| Class (Potency) | Examples | Typical Use | Pearl |
|---|---|---|---|
| I (Superpotent) | Clobetasol propionate 0.05% (Temovate); betamethasone dipropionate 0.05% in optimized vehicle (Diprolene); halobetasol propionate 0.05% (Ultravate) | Thick plaques (psoriasis, LP), palms/soles, severe eczema; limit 2 weeks continuous on body, avoid face/folds | Highest risk of atrophy, striae, adrenal suppression; steroid rosacea on face with prolonged misuse |
| II (High) | Fluocinonide 0.05% (Lidex); desoximetasone 0.25% (Topicort); betamethasone dipropionate 0.05% cream | Moderate-severe dermatoses on body | Good balance of efficacy and safety for trunk/extremity use |
| III (Upper-mid) | Triamcinolone acetonide 0.5%; fluticasone propionate 0.005% ointment | Moderate eczema, psoriasis on non-facial skin | Ointment vehicle is more potent than cream of same concentration (better occlusion) |
| IV (Mid) | Triamcinolone acetonide 0.1% (Kenalog); mometasone furoate 0.1% (Elocon); fluocinolone acetonide 0.025% | Standard workhorse for most body dermatoses | Triamcinolone 0.1% cream/ointment is the most commonly prescribed mid-potency TCS |
| V (Lower-mid) | Fluticasone propionate 0.05% cream; betamethasone valerate 0.1% cream; hydrocortisone butyrate 0.1% | Moderate eczema; children (body); transitioning from higher potency | Fluticasone 0.05% cream is popular for pediatric AD on the body |
| VI (Low) | Desonide 0.05% (DesOwen); alclometasone dipropionate 0.05% | Face, eyelids, intertriginous areas; mild eczema; infants | Preferred for sensitive areas where atrophy risk must be minimized |
| VII (Lowest) | Hydrocortisone 1-2.5% | Mild dermatitis, face/folds in children; OTC availability | Low efficacy for most dermatoses but safest class; often inadequate for established eczema |
Topical Calcineurin Inhibitors & Non-Steroidal Anti-Inflammatories
| Drug (Brand) | Mechanism | Indication | Pearl |
|---|---|---|---|
| Tacrolimus 0.03%/0.1% ointment (Protopic) | Calcineurin inhibitor — blocks T-cell activation (IL-2, IFN-gamma) | AD (face, folds — steroid-sparing); vitiligo; oral LP | Burning/stinging on application (improves with continued use); no skin atrophy; FDA black box warning re: lymphoma risk (theoretical, not demonstrated in large studies) |
| Pimecrolimus 1% cream (Elidel) | Calcineurin inhibitor (less potent than tacrolimus) | Mild-moderate AD (face, folds); SD | Less burning than tacrolimus; cream vehicle may be preferred; less effective than mid-potency TCS |
| Crisaborole 2% ointment (Eucrisa) | PDE4 inhibitor | Mild-moderate AD (age ≥3 months) | Non-steroidal, non-calcineurin inhibitor; burning on application; modest efficacy |
| Ruxolitinib 1.5% cream (Opzelura) | JAK1/2 inhibitor | AD (non-immunocompromised, age ≥12); vitiligo (non-segmental) | First topical JAK inhibitor; limit to ≤20% BSA; application site reactions; do not use under occlusion |
| Roflumilast 0.3% cream (Zoryve) | PDE4 inhibitor | Plaque psoriasis (including intertriginous); AD | Once daily; can be used on sensitive areas including face, folds; approved for psoriasis in 2022 |
| Tapinarof 1% cream (Vtama) | Aryl hydrocarbon receptor (AhR) agonist | Plaque psoriasis | Novel non-steroidal mechanism; once daily; promotes skin barrier and reduces Th17 inflammation; no atrophy risk |
Retinoids
| Drug (Brand) | Route | Mechanism | Indication | Pearl |
|---|---|---|---|---|
| Tretinoin 0.025-0.1% (Retin-A) | Topical | RAR-alpha/gamma agonist — normalizes keratinization, promotes collagen | Acne (comedonal, inflammatory), photoaging | Start low, increase gradually (retinoid dermatitis); photosensitizing — apply at night; pregnancy Category X |
| Adapalene 0.1-0.3% (Differin) | Topical | RAR-beta/gamma selective — more stable, less irritating than tretinoin | Acne | 0.1% available OTC; less irritation, less photosensitivity than tretinoin; equally effective |
| Tazarotene 0.05-0.1% (Tazorac) | Topical | RAR-beta/gamma selective | Psoriasis (plaque), acne | Most potent topical retinoid; most irritating; pregnancy Category X; short-contact (20 min) application reduces irritation |
| Isotretinoin (Accutane, Absorica) | Oral | Pan-retinoic acid; reduces sebum, normalizes keratinization, anti-inflammatory, decreases C. acnes | Severe nodulocystic acne; refractory acne | See Section 27 for complete details; iPLEDGE REMS; Category X; monitor lipids/LFTs |
| Acitretin (Soriatane) | Oral | Systemic retinoid (active metabolite of etretinate) | Psoriasis (pustular, erythrodermic, severe plaque); lichen planus; ichthyosis | 3-year pregnancy avoidance (no alcohol — converts to etretinate with 120-day half-life); monitor lipids, LFTs |
Immunosuppressants & Biologics
| Drug (Brand) | Target | Indication | Key Monitoring / Pearl |
|---|---|---|---|
| Dupilumab (Dupixent) | Anti-IL-4R-alpha (blocks IL-4 and IL-13) | Moderate-severe AD, asthma, CRSwNP, prurigo nodularis | 600 mg load → 300 mg q2wk SC; conjunctivitis (10-25%); no lab monitoring; no immunosuppression signal |
| Tralokinumab (Adbry) | Anti-IL-13 | Moderate-severe AD | 600 mg load → 300 mg q2wk SC (may extend to q4wk after 16 weeks); conjunctivitis less common than dupilumab |
| Abrocitinib (Cibinqo) | JAK1 inhibitor (oral) | Moderate-severe AD | 100-200 mg daily; check CBC, lipids; increased VTE risk (class warning); herpes reactivation |
| Upadacitinib (Rinvoq) | JAK1 inhibitor (oral) | Moderate-severe AD | 15-30 mg daily; rapid onset; acne common side effect; same class warnings as other JAK inhibitors |
| Baricitinib (Olumiant) | JAK1/2 inhibitor (oral) | AD; severe alopecia areata (first approved systemic) | 2-4 mg daily; laboratory monitoring (CBC, lipids, LFTs) |
| Ritlecitinib (Litfulo) | JAK3/TEC inhibitor (oral) | Severe alopecia areata | 50 mg daily; approved 2023 |
| Omalizumab (Xolair) | Anti-IgE | Chronic spontaneous urticaria (refractory to antihistamines) | 150-300 mg SC q4wk; monitor for anaphylaxis (rare); no lab monitoring |
| Adalimumab (Humira) | Anti-TNF-alpha | Psoriasis, HS, pyoderma gangrenosum | TB/Hep B screening before starting; increased infection risk |
| Secukinumab (Cosentyx) | Anti-IL-17A | Psoriasis (plaque, PsA); HS (recently approved) | Candidiasis risk; avoid in IBD |
| Risankizumab (Skyrizi) | Anti-IL-23 p19 | Psoriasis (plaque); emerging in other indications | 150 mg q12wk (after loading); highest PASI 90/100 rates; favorable safety |
| Rituximab (Rituxan) | Anti-CD20 (B-cell depletion) | Pemphigus vulgaris/foliaceus; refractory BP; refractory dermatomyositis | 1,000 mg IV day 1 and 15; PML risk (rare); hepatitis B reactivation screening |
| Cyclosporine (Neoral) | Calcineurin inhibitor | Severe AD, psoriasis, PG; bridge therapy | 2.5-5 mg/kg/day; limit 1-2 years; monitor Cr, BP, lipids; drug interactions (CYP3A4) |
| Mycophenolate mofetil (CellCept) | IMPDH inhibitor (purine synthesis) | Pemphigus, pemphigoid, DM, CLE | 1-3 g/day; CBC, LFTs; GI side effects (diarrhea); teratogenic |
| Azathioprine (Imuran) | Purine analog (6-MP prodrug) | AD, pemphigus, BP, DM, CLE | Check TPMT/NUDT15 before starting (low activity = myelosuppression risk); 1-3 mg/kg/day; CBC q2wk then q3mo |
| Methotrexate | DHFR inhibitor | Psoriasis, AD, DM, PG, sarcoidosis, MF | 7.5-25 mg/week + folic acid 1 mg daily; CBC, LFTs, Cr q3 months; hepatotoxicity; Category X |
| Dapsone | Neutrophil/eosinophil inhibitor + antimicrobial | DH, linear IgA disease, BP, Sweet syndrome, LP | Check G6PD before starting; dose-related hemolysis; methemoglobinemia; CBC q2wk x3mo |
| Hydroxychloroquine (Plaquenil) | Antimalarial; immunomodulatory | CLE, DM, sarcoidosis, LP | 200-400 mg/day; baseline + annual eye exam after 5 years (retinal toxicity — risk increases with cumulative dose >1,000 g); weight-based dosing (≤5 mg/kg real body weight/day) |
Antifungals
| Drug (Brand) | Mechanism | Route | Indication | Pearl |
|---|---|---|---|---|
| Terbinafine (Lamisil) | Squalene epoxidase inhibitor | Oral / Topical | Dermatophytosis (tinea capitis, onychomycosis, moccasin tinea pedis) | Fungicidal; 250 mg/day oral; monitor LFTs; taste disturbance (rare, usually reversible); drug interactions (CYP2D6 inhibitor) |
| Itraconazole (Sporanox) | Azole — ergosterol synthesis inhibitor (14-alpha-demethylase) | Oral | Onychomycosis, tinea capitis (Microsporum), sporotrichosis, deep fungal infections | 200 mg BID pulse dosing (1 week on, 3 weeks off) for onychomycosis; requires acid for absorption (take with food); LFTs; numerous drug interactions (CYP3A4 inhibitor); CHF risk (negative inotrope) |
| Fluconazole (Diflucan) | Azole | Oral | Candidiasis (vaginal, mucocutaneous); tinea versicolor; tinea corporis (if topical fails) | Good safety profile; fewer drug interactions than itraconazole; 150 mg x 1 for VVC; 150-300 mg/week for onychomycosis (off-label) |
| Griseofulvin | Inhibits fungal mitosis (microtubule disruption) | Oral | Tinea capitis (first-line in children for Microsporum) | Microsize 20-25 mg/kg/day x 6-8 weeks; must take with fatty food; photosensitivity; pregnancy Category X; ineffective against Candida |
| Ketoconazole 2% (Nizoral) | Azole | Topical (shampoo, cream) | Seborrheic dermatitis, tinea versicolor | Oral ketoconazole largely abandoned (hepatotoxicity risk); topical is safe and effective |
| Nystatin | Polyene — binds ergosterol, disrupts membrane | Topical | Candidiasis (skin, oral) | Active only against Candida (not dermatophytes); available as cream, powder, oral suspension |
Antibiotics (Dermatologic Use)
| Drug | Route | Dermatologic Use | Pearl |
|---|---|---|---|
| Doxycycline 40-200 mg | Oral | Acne (100 mg BID); rosacea (40 mg MR/Oracea — subantimicrobial anti-inflammatory); bullous pemphigoid; perioral dermatitis; cellulitis | Photosensitivity (warn patients); esophagitis (take upright with full glass of water); not in children <8 or pregnancy |
| Minocycline 50-200 mg | Oral | Acne (100 mg BID) | Blue-gray skin discoloration with prolonged use; vestibular symptoms (dizziness); drug-induced lupus; less photosensitivity than doxycycline |
| TMP-SMX DS | Oral | MRSA skin infections; nocardiosis; acne (third-line) | Sulfonamide allergy cross-reactivity; photosensitivity; SJS/TEN risk (especially in HIV) |
| Mupirocin 2% (Bactroban) | Topical | Impetigo; MRSA nasal decolonization; wound infection prophylaxis | Not effective against gram-negatives; do not use >10 days (resistance) |
| Clindamycin 1% | Topical | Acne; HS | Always combine with benzoyl peroxide (prevents C. acnes resistance); available as solution, gel, lotion, foam, pledgets |
46 Abbreviations Master List
Anatomy & Histology
Diagnoses
Procedures & Therapeutics
Medications & Labs
Scoring Systems & Scales
References & Figure Sources
Figures
- Figure 1 — Skin Layers and Architecture. From: Skin Anatomy. Eplasty. 2023;23:QA8. PMC10373447. Licensed under CC BY 4.0.
- Figure 2 — Layers of the Epidermis. From: Skin Anatomy. Eplasty. 2023;23:QA8. PMC10373447. Licensed under CC BY 4.0.
- Figure 3 — Basement Membrane Zone Molecular Architecture. From: Skin Anatomy. Eplasty. 2023;23:QA8. PMC10373447. Licensed under CC BY 4.0.
- Figure 4 — Dermoscopic Evaluation of a Melanocytic Lesion. From: Lallas A, et al. Malignant Melanoma: A Pictorial Review. Ulster Med J. 2014;83(2):103-110. PMC4113154. Licensed under CC BY 4.0.
- Figure 5 — Atopic Dermatitis in Children. From: Wittbold KA, et al. Characterization of Classical Flexural and Nummular Forms of Atopic Dermatitis. Acta Derm Venereol. 2022;102:adv00979. PMC9631256. Licensed under CC BY 4.0.
- Figure 6 — Acute Allergic Contact Dermatitis. From: Litchman G, et al. Contact Dermatitis, Patch Testing, and Allergen Avoidance. Mo Med. 2015;112(4):296-300. PMC6170075.
- Figure 7 — Patch Testing for Allergic Contact Dermatitis. From: Litchman G, et al. Contact Dermatitis, Patch Testing, and Allergen Avoidance. Mo Med. 2015;112(4):296-300. PMC6170075.
- Figure 8 — Clinical Manifestations of Plaque Psoriasis. From: Rendon A, Schakel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20(6):1475. PMC6471628. Licensed under CC BY 4.0.
- Figure 9 — Psoriasis Pathogenesis: The IL-23/Th17 Axis. From: Rendon A, Schakel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20(6):1475. PMC6471628. Licensed under CC BY 4.0.
- Figure 10 — Nail Psoriasis. From: Rendon A, Schakel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20(6):1475. PMC6471628. Licensed under CC BY 4.0.
- Figure 11 — Actinic Keratosis and Field Cancerization. From: Dianzani C, et al. Management Pearls on the Treatment of Actinic Keratoses and Field Cancerization. Dermatol Ther. 2020;10:1015-1023. PMC7477025. Licensed under CC BY 4.0.
- Figure 12 — Nodular Basal Cell Carcinoma. From: Demirseren DD, et al. Clinical variants, stages, and management of basal cell carcinoma. Indian Dermatol Online J. 2014;5(Suppl 1):S12-S17. PMC3573444. Licensed under CC BY-NC-SA 3.0.
- Figure 13 — Superficial Basal Cell Carcinoma. From: Demirseren DD, et al. Clinical variants, stages, and management of basal cell carcinoma. Indian Dermatol Online J. 2014;5(Suppl 1):S12-S17. PMC3573444. Licensed under CC BY-NC-SA 3.0.
- Figure 14 — Cutaneous Squamous Cell Carcinoma. From: Combalia A, Carrera C. Squamous Cell Carcinoma: An Update on Diagnosis and Treatment. Dermatol Pract Concept. 2020;10(3):e2020066. PMC7319751. Licensed under CC BY 4.0.
- Figure 15 — Superficial Spreading Melanoma. From: Lallas A, et al. Malignant Melanoma: A Pictorial Review. Ulster Med J. 2014;83(2):103-110. PMC4113154. Licensed under CC BY 4.0.
- Figure 16 — Nodular Melanoma. From: Lallas A, et al. Malignant Melanoma: A Pictorial Review. Ulster Med J. 2014;83(2):103-110. PMC4113154. Licensed under CC BY 4.0.
- Figure 17 — Herpes Zoster (Shingles). From: Mittal N, et al. Herpes zoster: A clinicocytopathological insight. J Oral Maxillofac Pathol. 2016;20(3):547. PMC5051314. Licensed under CC BY-NC-SA 3.0.
- Figure 18 — Tinea Corporis (Ringworm). From: Leung AKC, et al. Tinea corporis: an updated review. Drugs Context. 2020;9:2020-5-6. PMC7375854. Licensed under CC BY 4.0.
- Figure 19 — KOH Preparation Showing Dermatophyte Hyphae. From: Leung AKC, et al. Tinea corporis: an updated review. Drugs Context. 2020;9:2020-5-6. PMC7375854. Licensed under CC BY 4.0.
- Figure 20 — Scabies Burrows. From: Chandler DJ, Fuller LC. Classic and Non-classic (Surrepticius) Scabies. Cureus. 2020;12(3):e7419. PMC7186094. Licensed under CC BY 3.0.
- Figure 21 — Oral Pemphigus Vulgaris. From: Dagistan S, et al. Oral Pemphigus Vulgaris: Case Report. Ethiop J Health Sci. 2015;25(4):367-372. PMC4762976. Licensed under CC BY 4.0.
- Figure 22 — Bullous Pemphigoid. From: Coulibaly B, et al. Bullous pemphigoid: a rare clinical image. Pan Afr Med J. 2024;47:203. PMC11380604. Licensed under CC BY 4.0.
- Figure 23 — Dermal-Epidermal Junction Histology. From: Skin Anatomy. Eplasty. 2023;23:QA8. PMC10373447. Licensed under CC BY 4.0.
- Figure 24 — Discoid Lupus Erythematosus. From: Unilateral Discoid Lupus Erythematosus Over the Face. Indian Dermatol Online J. 2018;9(4):268-270. PMC6042185. Licensed under CC BY-NC-SA 3.0.
- Figure 25 — Severe Nodulocystic Acne Vulgaris. From: Al Rabahi SS, Al Bimani AS. Severe Nodulocystic Acne not Responding to Isotretinoin Therapy Successfully Treated with Oral Dapsone. Oman Med J. 2018;33(5):433-436. PMC6131924. Licensed under CC BY 4.0.
- Figure 26 — Rosacea Subtypes. From: Two AM, et al. Rosacea: a Clinical Review. Dermatol Reports. 2015;7(1):6387. PMC5134688. Licensed under CC BY 4.0.
- Figure 27 — Hidradenitis Suppurativa. From: Zouboulis CC, et al. Update on hidradenitis suppurativa: connecting the tracts. F1000Prime Reports. 2015;7:15. PMC4627882. Licensed under CC BY 4.0.
- Figure 28 — Alopecia Areata: Clinical Manifestations. From: Pratt CH, et al. Alopecia areata. Nat Rev Dis Primers. 2017;3:17011. PMC5573125.
- Figure 29 — Vitiligo Histopathology. From: Educational Case: Vitiligo. Acad Pathol. 2019;6:2374289519888719. PMC6886271. Licensed under CC BY-NC 4.0.
- Figure 30 — Toxic Epidermal Necrolysis (TEN). From: Frantz R, et al. Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis: A Review of Diagnosis and Management. Medicina. 2021;57(9):895. PMC8472007. Licensed under CC BY 4.0.
- Figure 31 — Mycosis Fungoides (Cutaneous T-Cell Lymphoma). From: Cutaneous T-Cell Lymphoma (Mycosis Fungoides). J Cutan Med Surg. 2016;20(5):457-459. PMC4925357.
- Figure 32 — Punch Biopsy Technique. From: Nischal U, et al. Techniques of Skin Biopsy and Practical Considerations. J Cutan Aesthet Surg. 2008;1(2):107-111. PMC2840913. Licensed under CC BY-NC-SA 3.0.
- Figure 33 — Morpheaform Basal Cell Carcinoma. From: Demirseren DD, et al. Clinical variants, stages, and management of basal cell carcinoma. Indian Dermatol Online J. 2014;5(Suppl 1):S12-S17. PMC3573444. Licensed under CC BY-NC-SA 3.0.
- Figure 34 — Bowen Disease (SCC In Situ). From: Combalia A, Carrera C. Squamous Cell Carcinoma: An Update on Diagnosis and Treatment. Dermatol Pract Concept. 2020;10(3):e2020066. PMC7319751. Licensed under CC BY 4.0.
- Figure 35 — Psoriasis Histopathology. From: Rendon A, Schakel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20(6):1475. PMC6471628. Licensed under CC BY 4.0.
- Figure 36 — Lentigo Maligna Melanoma. From: Lallas A, et al. Malignant Melanoma: A Pictorial Review. Ulster Med J. 2014;83(2):103-110. PMC4113154. Licensed under CC BY 4.0.
- Figure 37 — Erythrodermic Psoriasis. From: Rendon A, Schakel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20(6):1475. PMC6471628. Licensed under CC BY 4.0.
- Figure 38 — Dermoscopic Progression: AK to SCC. From: Combalia A, Carrera C. Squamous Cell Carcinoma: An Update on Diagnosis and Treatment. Dermatol Pract Concept. 2020;10(3):e2020066. PMC7319751. Licensed under CC BY 4.0.
Inline Citations
- Weidinger S, Novak N. Atopic dermatitis. Lancet. 2016;387(10023):1109-1122. PubMed
- Wollenberg A, et al. Proactive treatment of atopic dermatitis in adults with 0.1% tacrolimus ointment. JEADV. 2011;25(9):1012-1020. PubMed
- Armstrong AW, Read C. Pathophysiology, clinical presentation, and treatment of psoriasis: a review. JAMA. 2020;323(19):1945-1960. PubMed
- Gordon KB, et al. Efficacy and safety of risankizumab: results from IMMvent. Lancet. 2021;397(10284):1564-1575. PubMed
- Criscione VD, et al. Actinic keratoses: natural history and risk of malignant transformation. JAMA Dermatol. 2009;145(11):1275-1280. PubMed
- Que SKT, et al. Cutaneous squamous cell carcinoma: incidence, risk factors, diagnosis, and staging. JAAD. 2018;78(2):237-247. PubMed
- Migden MR, et al. PD-1 blockade with cemiplimab in advanced cutaneous squamous-cell carcinoma. NEJM. 2018;379(4):341-351. PubMed
- Luke JJ, et al. Pembrolizumab versus placebo as adjuvant therapy in resected stage IIB or IIC melanoma. NEJM. 2022;386(6):573-576. PubMed
- Joly P, et al. First-line rituximab combined with short-term prednisone versus prednisone alone for treatment of pemphigus (RITUX 3). Lancet. 2017;389(10083):2031-2040. PubMed
- Mockenhaupt M, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks. J Invest Dermatol. 2008;128(1):35-44. PubMed
- Wang CW, et al. Randomized, controlled trial of TNF-alpha antagonist in CTL-mediated severe cutaneous adverse reactions. JAMA Dermatol. 2021;157(12):1–9. PubMed