Endocrinology
Every hormone axis, diabetes protocol, thyroid disorder, adrenal condition, pituitary tumor, calcium pathway, and metabolic emergency - with diagnostic algorithms, dosing tables, and dynamic testing protocols in one place.
01 Endocrine Anatomy & Physiology
The endocrine system comprises glands and specialized cells that produce hormones — chemical messengers released into the bloodstream to act on distant target organs. Unlike the nervous system's rapid, point-to-point signaling, endocrine signaling is slower but sustained, governing metabolism, growth, reproduction, electrolyte balance, and stress responses. The major endocrine organs include the hypothalamus, pituitary, thyroid, parathyroids, adrenal glands, pancreatic islets, and gonads.
Hypothalamic-Pituitary Axes
The hypothalamus integrates neural and hormonal signals and secretes releasing/inhibiting hormones into the hypophyseal portal system to regulate the anterior pituitary. Each axis follows a three-tier hierarchy: hypothalamic hormone → pituitary tropic hormone → peripheral gland hormone. Negative feedback by the peripheral hormone on both the hypothalamus and pituitary is the central regulatory mechanism.
| Axis | Hypothalamic Signal | Pituitary Hormone | Target Gland | End Hormone |
|---|---|---|---|---|
| Thyroid | TRH | TSH | Thyroid | T4, T3 |
| Adrenal | CRH | ACTH | Adrenal cortex | Cortisol |
| Gonadal | GnRH (pulsatile) | LH, FSH | Ovaries / Testes | Estradiol, Progesterone / Testosterone |
| Growth | GHRH (+) / Somatostatin (−) | GH | Liver (+ tissues) | IGF-1 |
| Prolactin | Dopamine (inhibitory) | Prolactin | Breast | — |
Hormone Classification
| Type | Examples | Receptor Location | Carrier Proteins | Half-Life |
|---|---|---|---|---|
| Peptide / Protein | Insulin, GH, PTH, ACTH | Cell surface | Usually free | Minutes |
| Steroid | Cortisol, aldosterone, estradiol, testosterone | Intracellular (nuclear) | CBG, SHBG, albumin | Hours |
| Thyroid hormones | T4, T3 | Nuclear | TBG, albumin, TTR | T4: 6–7 days; T3: 1 day |
| Amine | Catecholamines, serotonin | Cell surface | Free | Seconds to minutes |
Feedback Loops
Negative feedback is the dominant regulatory mechanism: rising peripheral hormone levels suppress the hypothalamic-pituitary drive. Example: elevated free T4 suppresses TSH and TRH secretion. Positive feedback is rare but critical: the mid-cycle estradiol surge triggers the LH surge to cause ovulation. Understanding feedback is essential for interpreting lab patterns. In primary gland failure, the peripheral hormone is low and the pituitary tropic hormone is high (loss of negative feedback). In secondary (pituitary) failure, both the tropic and peripheral hormones are low.
02 The Endocrine Physical Exam
Thyroid Examination
The thyroid gland sits in the anterior neck, overlying the 2nd–4th tracheal rings. Normal weight is 15–20 g. Examine from behind the patient: palpate both lobes and the isthmus while the patient swallows (the thyroid moves with swallowing because it is attached to the pretracheal fascia — this distinguishes thyroid masses from other neck masses). Assess for nodules (size, consistency, mobility, tenderness), diffuse enlargement (goiter), and thyroid bruit (increased vascularity in Graves disease). Pemberton sign: raise both arms above the head for 1 minute — facial plethora and JVD indicate substernal goiter compressing the thoracic inlet.
Signs of Hyperthyroidism
| System | Findings |
|---|---|
| Eyes | Lid lag, lid retraction, proptosis/exophthalmos (Graves-specific), chemosis, extraocular muscle restriction |
| Cardiovascular | Tachycardia, atrial fibrillation, widened pulse pressure, systolic flow murmur |
| Neurologic | Fine tremor (outstretched hands), hyperreflexia (brisk DTRs with short relaxation phase) |
| Skin | Warm, moist skin; pretibial myxedema (Graves-specific); onycholysis; palmar erythema |
| General | Weight loss despite increased appetite, heat intolerance, anxiety, insomnia |
Signs of Hypothyroidism
| System | Findings |
|---|---|
| Skin / Hair | Coarse, dry skin; non-pitting edema (myxedema); hair loss (lateral third of eyebrows), brittle nails |
| Cardiovascular | Bradycardia, diastolic hypertension, pericardial effusion |
| Neurologic | Delayed relaxation phase of DTRs (classic), carpal tunnel syndrome, cognitive slowing |
| General | Weight gain, cold intolerance, constipation, fatigue, menorrhagia |
Cushing Syndrome Habitus
Classic findings: central obesity with thin extremities, moon facies, dorsocervical fat pad (buffalo hump), supraclavicular fat pads, wide purple striae (>1 cm, violaceous — distinguish from common pink striae of obesity), proximal muscle weakness (difficulty rising from chair), easy bruising, thin skin, and facial plethora. Women may have hirsutism and acne from adrenal androgen excess.
Acromegaly Features
Insidious onset — compare old photographs. Look for: enlarged hands and feet (increasing ring/shoe size), coarsened facial features (frontal bossing, prognathism, macroglossia, widened nose), skin tags, deepened voice, carpal tunnel syndrome, hyperhidrosis, and visual field defects (bitemporal hemianopsia from pituitary macroadenoma compressing the optic chiasm).
Diabetic Foot Examination
Comprehensive foot exam at every diabetes visit: inspect for ulcers, calluses, deformities (Charcot foot), and nail pathology. Test sensation with 10 g Semmes-Weinstein monofilament at plantar sites (1st, 3rd, 5th metatarsal heads, plantar hallux) plus 128 Hz tuning fork at dorsal great toe for vibration sense. Assess dorsalis pedis and posterior tibial pulses. Loss of protective sensation is the primary risk factor for diabetic foot ulceration.
03 Key Terminology & Abbreviations
Endocrinology uses dense abbreviations across diabetes management, thyroid diagnostics, adrenal testing, and pituitary evaluation. Mastery of these terms is essential for interpreting labs, imaging reports, and clinical notes.
| Abbreviation | Meaning |
|---|---|
| A1C (HbA1c) | Glycosylated hemoglobin (reflects ~3-month average glucose) |
| DKA | Diabetic ketoacidosis |
| HHS | Hyperosmolar hyperglycemic state |
| GLP-1 RA | Glucagon-like peptide-1 receptor agonist |
| SGLT2i | Sodium-glucose co-transporter 2 inhibitor |
| DPP-4i | Dipeptidyl peptidase-4 inhibitor |
| TZD | Thiazolidinedione |
| SU | Sulfonylurea |
| CGM | Continuous glucose monitor |
| TSH | Thyroid-stimulating hormone (thyrotropin) |
| FT4 / FT3 | Free thyroxine / free triiodothyronine |
| TPO Ab | Thyroid peroxidase antibody |
| TRAb / TSI | TSH receptor antibody / thyroid-stimulating immunoglobulin |
| RAIU | Radioactive iodine uptake |
| FNA | Fine-needle aspiration |
| ACTH | Adrenocorticotropic hormone |
| CRH | Corticotropin-releasing hormone |
| DST | Dexamethasone suppression test |
| UFC | 24-hour urinary free cortisol |
| ARR | Aldosterone-to-renin ratio |
| PTH | Parathyroid hormone |
| PTHrP | Parathyroid hormone-related peptide |
| DEXA (DXA) | Dual-energy X-ray absorptiometry |
| MEN | Multiple endocrine neoplasia |
| IGF-1 | Insulin-like growth factor 1 |
| GH | Growth hormone |
| SHBG | Sex hormone-binding globulin |
| DHEA-S | Dehydroepiandrosterone sulfate |
| PCOS | Polycystic ovary syndrome |
| DI | Diabetes insipidus |
| SIADH | Syndrome of inappropriate antidiuretic hormone |
| ADH (AVP) | Antidiuretic hormone (arginine vasopressin) |
04 Type 1 Diabetes
Type 1 diabetes mellitus (T1DM) results from autoimmune destruction of pancreatic beta cells, leading to absolute insulin deficiency. It accounts for ~5–10% of all diabetes cases. Peak incidence is bimodal: ages 4–6 and 10–14, but can occur at any age (including adults — latent autoimmune diabetes in adults, LADA). The autoimmune process is triggered by environmental factors in genetically susceptible individuals (HLA-DR3, HLA-DR4).
Autoantibodies
| Antibody | Target | Clinical Significance |
|---|---|---|
| GAD65 | Glutamic acid decarboxylase | Most sensitive in adults; present in ~70–80% of T1DM |
| IA-2 | Islet antigen 2 (tyrosine phosphatase) | Present in ~60%; higher specificity |
| IAA | Insulin autoantibodies | Most useful in children <5 years; must measure before exogenous insulin |
| ZnT8 | Zinc transporter 8 | Present in ~60–80%; adds sensitivity when others negative |
Clinical Presentation
Classic triad: polyuria, polydipsia, polyphagia with weight loss. Onset is typically acute (days to weeks). ~30% of children present with DKA at diagnosis. C-peptide is low or undetectable (reflects endogenous insulin production). The honeymoon phase occurs in ~60% of patients after insulin initiation: residual beta cells temporarily recover, reducing insulin requirements for weeks to months. Insulin requirements eventually rise as beta cell destruction becomes complete.

Management Principles
All patients with T1DM require lifelong insulin therapy. The goal is to mimic physiologic insulin secretion with basal-bolus therapy or an insulin pump (CSII). Target A1C is generally <7% for most adults (ADA), with individualization. CGM use is strongly recommended — time in range (TIR) of 70–180 mg/dL should be ≥70% of the time. Carbohydrate counting and insulin-to-carb ratios are fundamental skills.
05 Type 2 Diabetes
Type 2 diabetes mellitus (T2DM) is characterized by insulin resistance and progressive beta cell dysfunction. It accounts for ~90–95% of all diabetes. Strong risk factors include obesity (especially visceral adiposity), family history, sedentary lifestyle, and ethnicity (higher prevalence in Black, Hispanic, Native American, and Asian populations). The pathophysiology involves the "ominous octet": reduced insulin secretion, increased glucagon, increased hepatic glucose production, decreased peripheral glucose uptake, increased lipolysis, decreased incretin effect, increased renal glucose reabsorption, and neurotransmitter dysfunction.
Diagnostic Criteria (ADA)
| Test | Diabetes | Prediabetes | Normal |
|---|---|---|---|
| Fasting plasma glucose | ≥126 mg/dL | 100–125 mg/dL (IFG) | <100 mg/dL |
| 2-hour OGTT (75 g) | ≥200 mg/dL | 140–199 mg/dL (IGT) | <140 mg/dL |
| A1C | ≥6.5% | 5.7–6.4% | <5.7% |
| Random glucose + symptoms | ≥200 mg/dL | — | — |
Two abnormal tests are required for diagnosis in asymptomatic patients. A single random glucose ≥200 mg/dL with classic symptoms (polyuria, polydipsia, weight loss) is sufficient.
Screening Recommendations
Screen all adults age ≥35 (ADA 2024). Screen earlier if BMI ≥25 kg/m² (or ≥23 in Asian Americans) with one or more risk factors: first-degree relative with diabetes, high-risk ethnicity, history of GDM, PCOS, hypertension, dyslipidemia (HDL <35 or TG >250), A1C ≥5.7%, or cardiovascular disease. Repeat testing every 3 years if normal; annually if prediabetes.
A1C Targets
| Population | A1C Target | Rationale |
|---|---|---|
| Most non-pregnant adults | <7.0% | Proven microvascular benefit (DCCT, UKPDS) |
| Selected motivated patients, short duration | <6.5% | If achievable without significant hypoglycemia |
| Older adults, comorbidities, limited life expectancy | <8.0% | Avoid hypoglycemia; harms of intensive control (ACCORD) |
| Pregnancy (pregestational) | <6.0–6.5% | Minimize teratogenicity and macrosomia |
06 Oral & Non-Insulin Agents
Pharmacotherapy for T2DM has evolved dramatically. Agent selection now considers not only glycemic efficacy but also cardiovascular benefit, renal protection, weight effects, and hypoglycemia risk. Metformin remains first-line for most patients, but GLP-1 RAs and SGLT2 inhibitors are now recommended as early therapy in patients with established ASCVD, heart failure, or CKD regardless of A1C.
| Drug Class | Mechanism | A1C Reduction | Key Benefits | Key Risks | Representative Agents |
|---|---|---|---|---|---|
| Biguanide | Decreases hepatic glucose production; improves insulin sensitivity | 1.0–1.5% | Weight neutral/mild loss, no hypoglycemia, low cost | GI side effects, lactic acidosis (rare), B12 deficiency | Metformin 500–2000 mg/day |
| SGLT2 inhibitor | Blocks renal glucose reabsorption in PCT | 0.5–0.9% | CV mortality benefit, HF benefit, renal protection | Genital mycotic infections, euglycemic DKA, volume depletion | Empagliflozin 10–25 mg, dapagliflozin 5–10 mg, canagliflozin 100–300 mg |
| GLP-1 RA | Incretin mimetic: enhances glucose-dependent insulin secretion, suppresses glucagon, slows gastric emptying, promotes satiety | 1.0–1.8% | CV benefit (MACE reduction), significant weight loss | GI (nausea, vomiting), pancreatitis risk (small), contraindicated in MTC/MEN2 | Semaglutide 0.25–2 mg/wk, liraglutide 0.6–1.8 mg/day, dulaglutide 0.75–4.5 mg/wk, tirzepatide 2.5–15 mg/wk |
| DPP-4 inhibitor | Prevents incretin degradation (raises endogenous GLP-1/GIP) | 0.5–0.8% | Weight neutral, well tolerated, no hypoglycemia | Possible HF risk (saxagliptin), joint pain | Sitagliptin 100 mg, linagliptin 5 mg, saxagliptin 5 mg |
| Sulfonylurea | Stimulates insulin secretion (closes K-ATP channels) | 1.0–1.5% | Rapid efficacy, low cost | Hypoglycemia, weight gain | Glipizide 5–20 mg, glimepiride 1–4 mg, glyburide 2.5–20 mg |
| TZD | PPAR-gamma agonist; improves insulin sensitivity in adipose/muscle | 1.0–1.5% | Durable effect, no hypoglycemia | Weight gain, fluid retention, HF exacerbation, fractures | Pioglitazone 15–45 mg |
| Alpha-glucosidase inhibitor | Delays carbohydrate absorption in small intestine | 0.5–0.8% | Reduces postprandial glucose | Flatulence, abdominal discomfort | Acarbose 25–100 mg TID |
Step 1: Metformin + lifestyle for all patients. Step 2: If ASCVD or high CV risk → add GLP-1 RA or SGLT2i with proven CV benefit. If heart failure → add SGLT2i. If CKD → add SGLT2i (or GLP-1 RA if SGLT2i not tolerated). If weight management is primary concern → GLP-1 RA (or tirzepatide). If cost is a barrier → SU or TZD. Do not combine GLP-1 RA with DPP-4i (redundant mechanism).
07 Insulin Therapy
Insulin is required for all patients with T1DM and many with T2DM when oral agents are insufficient or contraindicated. The goal of insulin therapy is to replicate the physiologic insulin profile: a continuous low-level basal secretion plus meal-time bolus surges.
Insulin Types
| Category | Insulin | Onset | Peak | Duration | Use |
|---|---|---|---|---|---|
| Rapid-acting | Lispro, Aspart, Glulisine | 10–15 min | 1–2 hr | 3–5 hr | Bolus (meals), correction doses, pumps |
| Short-acting | Regular (R) | 30–60 min | 2–4 hr | 6–8 hr | Meals (if cost issue), IV insulin drip (DKA/HHS) |
| Intermediate | NPH | 1–2 hr | 4–8 hr | 12–16 hr | Basal (BID dosing), low cost |
| Long-acting | Glargine (U-100, U-300), Detemir | 1–2 hr | Minimal | 20–24 hr | Basal (once daily) |
| Ultra-long | Degludec | 1–2 hr | None | >42 hr | Basal (once daily, flexible timing) |
| Premixed | 70/30 (NPH/Regular), 75/25 (lispro mix) | 30 min | Dual peak | 12–16 hr | BID dosing, fixed regimen |
Dosing Calculations
Total daily dose (TDD) for T1DM: 0.4–0.6 units/kg/day (higher in puberty, illness). For T2DM initiation: start basal insulin at 0.1–0.2 units/kg/day (or 10 units) and titrate. The TDD is split: ~50% basal, ~50% bolus (divided across meals). Insulin-to-carb ratio (ICR): 450–500 ÷ TDD = grams of carbohydrate covered by 1 unit (the "450 rule" for rapid-acting). Correction factor (CF): 1700 ÷ TDD = mg/dL drop per 1 unit of rapid-acting insulin (the "1700 rule").
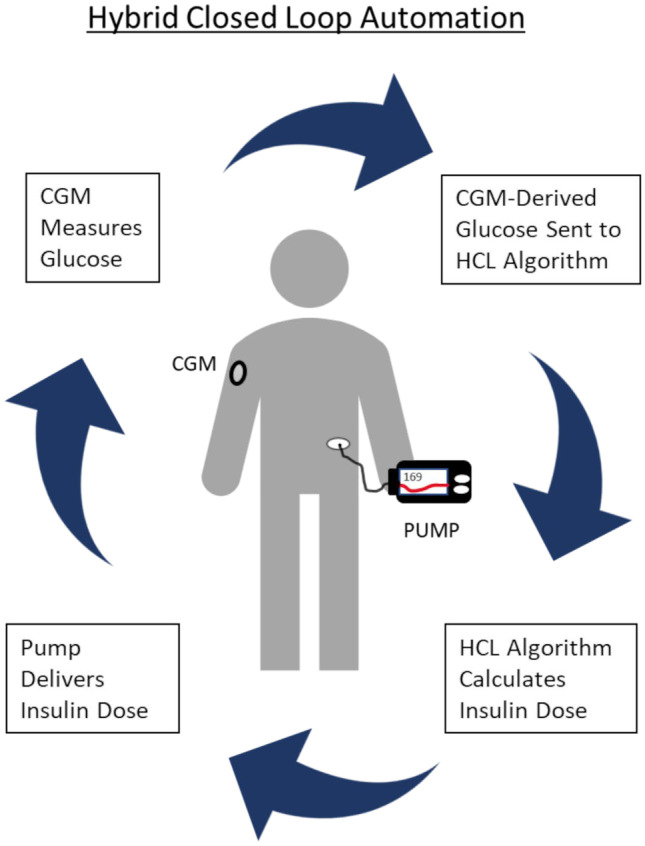
Insulin Pumps & CGM
Continuous subcutaneous insulin infusion (CSII) delivers rapid-acting insulin via a pump with programmable basal rates and on-demand boluses. Advantages: flexible basal profiles, precise dosing, reduced hypoglycemia. CGM measures interstitial glucose every 1–5 minutes. Automated insulin delivery (AID) systems ("closed loop" or "hybrid closed loop") combine a pump with CGM and an algorithm that auto-adjusts basal delivery. Key metrics from CGM: time in range (TIR) 70–180 mg/dL (goal ≥70%), time below range <70 (<4%), glucose management indicator (GMI) as an alternative A1C estimate.
08 Diabetic Ketoacidosis (DKA)
DKA is a life-threatening complication of absolute or relative insulin deficiency. Mortality is 1–5% in adults but higher with delayed treatment, cerebral edema (especially in children), or severe comorbidities. The most dangerous electrolyte abnormality in DKA is hypokalemia — always check K+ before starting insulin.
Diagnostic Criteria
| Parameter | Mild DKA | Moderate DKA | Severe DKA |
|---|---|---|---|
| Glucose | >250 mg/dL | >250 mg/dL | >250 mg/dL |
| Arterial pH | 7.25–7.30 | 7.00–7.24 | <7.00 |
| Serum bicarbonate | 15–18 mEq/L | 10–14 mEq/L | <10 mEq/L |
| Anion gap | >10 | >12 | >12 |
| Serum ketones | Positive | Positive | Positive |
| Mental status | Alert | Alert / Drowsy | Stupor / Coma |
Precipitants (The 5 I's)
Infection (most common, ~40%), Insulin omission/noncompliance, Infarction (MI, stroke, mesenteric ischemia), Intoxication (alcohol, cocaine), Iatrogenic (steroids, SGLT2 inhibitors — can cause euglycemic DKA).
DKA Management Protocol
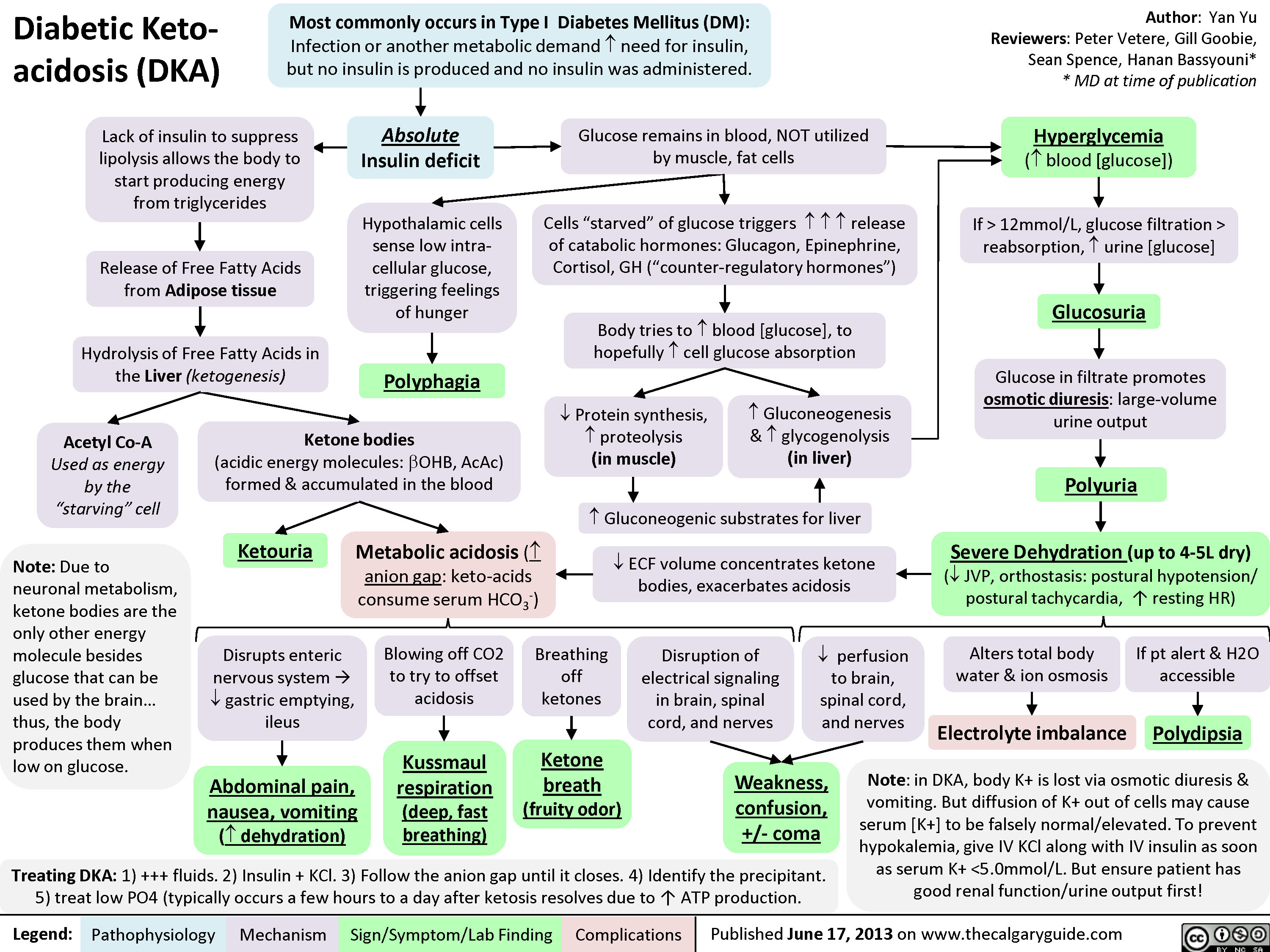
1. Fluids: Start 0.9% NS at 1–1.5 L/hr for the first hour (15–20 mL/kg). Then adjust based on corrected sodium: if Na+ is low, continue 0.9% NS at 250–500 mL/hr; if Na+ is normal or high, switch to 0.45% NS at 250–500 mL/hr. When glucose reaches 200–250 mg/dL, add D5 to the IV fluids (D5 0.45% NS) to prevent hypoglycemia while continuing insulin to close the anion gap.
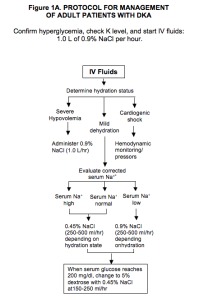
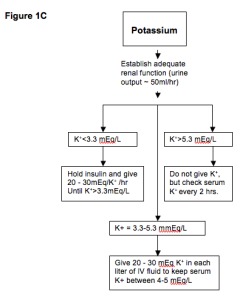
2. Potassium: Check K+ before insulin. If K+ <3.3 mEq/L: hold insulin, replete K+ aggressively (20–40 mEq/hr). If K+ 3.3–5.3: add 20–40 mEq KCl to each liter of IV fluids. If K+ >5.3: hold K+ replacement, recheck in 2 hours. Goal: maintain K+ at 4–5 mEq/L.
3. Insulin: Regular insulin bolus 0.1 units/kg IV, then continuous drip at 0.1 units/kg/hr (or skip bolus and start at 0.14 units/kg/hr). Target glucose decline: 50–75 mg/dL/hr. If glucose does not drop by 50–70 in the first hour, double the drip rate. When glucose reaches 200–250 mg/dL, reduce drip to 0.02–0.05 units/kg/hr and add dextrose to fluids. Continue insulin drip until anion gap closes (AG ≤12, bicarb ≥15, pH >7.30).
4. Bicarbonate: Only if pH <6.9. Give 100 mEq NaHCO3 in 400 mL H2O with 20 mEq KCl over 2 hours. Recheck ABG; repeat if pH remains <6.9. Do NOT give bicarbonate if pH ≥6.9 — it worsens intracellular acidosis and hypokalemia.
Monitoring
Check BMP (glucose, K+, bicarb, anion gap) every 1–2 hours until stable. Transition to subcutaneous insulin when: patient tolerating PO, anion gap closed, glucose <200 mg/dL, and at least 2 of: pH >7.30, bicarb ≥15, AG ≤12. Give subcutaneous basal insulin 2 hours before stopping drip.
09 Hyperosmolar Hyperglycemic State (HHS)
HHS is a life-threatening diabetic emergency characterized by severe hyperglycemia (>600 mg/dL), hyperosmolality (>320 mOsm/kg), and profound dehydration without significant ketoacidosis. It occurs almost exclusively in T2DM, typically in elderly patients with limited access to fluids. Mortality is higher than DKA (5–20%) due to the older, more comorbid population.
DKA vs HHS Comparison
| Feature | DKA | HHS |
|---|---|---|
| Diabetes type | Usually T1DM (can occur in T2DM) | T2DM |
| Glucose | >250 mg/dL | >600 mg/dL (often >1000) |
| pH | <7.30 | >7.30 |
| Bicarbonate | <18 mEq/L | >18 mEq/L |
| Ketones | Strongly positive | Absent or trace |
| Anion gap | Elevated | Normal or mildly elevated |
| Serum osmolality | Variable | >320 mOsm/kg |
| Fluid deficit | 3–6 L | 8–12 L |
| Mental status | Variable | Often altered (stupor, coma, seizures) |
| Onset | Hours to days | Days to weeks |
| Mortality | 1–5% | 5–20% |
Management
Aggressive fluid resuscitation is the cornerstone — patients may have 8–12 L deficits. Start 0.9% NS at 1–1.5 L/hr for the first 1–2 hours. Corrected Na+ determines subsequent fluids (same as DKA protocol). Insulin is started at 0.1 units/kg/hr IV (some protocols use lower initial rates of 0.05 units/kg/hr since these patients are insulin-sensitive and the primary problem is dehydration). Monitor closely for rapid osmolality shifts, which can precipitate cerebral edema. Thromboprophylaxis with heparin is recommended due to the severely dehydrated, hypercoagulable state.
10 Thyroid Function Testing & Interpretation
The TSH is the single best screening test for thyroid dysfunction. It is exquisitely sensitive to small changes in free thyroid hormone levels due to the log-linear relationship between free T4 and TSH. A 2-fold change in FT4 produces a 100-fold change in TSH.
Lab Patterns
| Condition | TSH | Free T4 | Free T3 | Notes |
|---|---|---|---|---|
| Primary hypothyroidism | ↑↑ | ↓ | ↓ or normal | Most common: Hashimoto thyroiditis |
| Subclinical hypothyroidism | ↑ (4.5–10) | Normal | Normal | Treat if TSH >10, symptoms, or pregnancy |
| Primary hyperthyroidism | ↓↓ (<0.1) | ↑ | ↑ | Graves, toxic MNG, toxic adenoma |
| Subclinical hyperthyroidism | ↓ (0.1–0.4) | Normal | Normal | Risk for AFib (especially age >65), osteoporosis |
| T3 thyrotoxicosis | ↓↓ | Normal | ↑ | Early Graves; check FT3 when TSH is suppressed but FT4 normal |
| Central hypothyroidism | Low or normal | ↓ | ↓ | Pituitary/hypothalamic disease; TSH unreliable |
| Sick euthyroid (NTIS) | Low, normal, or mildly ↑ | Low or normal | ↓ | Acute illness; do NOT treat. Recheck 6–8 weeks after recovery |
| TSH-secreting adenoma | ↑ or normal | ↑ | ↑ | Rare; inappropriate TSH for elevated T4 |
11 Hypothyroidism
Primary hypothyroidism accounts for >95% of cases. The most common cause in iodine-sufficient regions is Hashimoto thyroiditis (chronic lymphocytic thyroiditis), an autoimmune condition with anti-TPO antibodies (positive in ~95%) and anti-thyroglobulin antibodies. Other causes include post-radioactive iodine therapy, post-thyroidectomy, medications (amiodarone, lithium, immune checkpoint inhibitors), iodine deficiency (most common cause worldwide), and radiation.
Levothyroxine (T4) Replacement
| Population | Starting Dose | Titration | Notes |
|---|---|---|---|
| Young, healthy adults | 1.6 mcg/kg/day (full replacement) | Recheck TSH in 6–8 weeks | Goal TSH 0.5–2.5 mU/L for most |
| Elderly or cardiac disease | 25–50 mcg/day | Increase by 12.5–25 mcg every 6–8 weeks | Start low, go slow to avoid angina/arrhythmia |
| Pregnancy | Increase dose by ~30% once pregnancy confirmed | Check TSH every 4 weeks in 1st trimester | Goal TSH: 1st trimester <2.5; 2nd/3rd <3.0 |
| Post-thyroidectomy (cancer) | Full suppressive dose (TSH <0.1) | Per risk stratification | Higher doses for high-risk DTC |
Administration: take on an empty stomach, 30–60 minutes before breakfast (or at bedtime, 3+ hours after last meal). Separate from calcium, iron, PPIs, and antacids by at least 4 hours (these impair absorption).
Myxedema coma is the extreme, life-threatening form of hypothyroidism. Triggers: cold exposure, infection, sedatives, surgery. Features: hypothermia, altered mental status/coma, bradycardia, hypotension, hypoventilation, hyponatremia, and hypoglycemia. Mortality: 25–60%. Treatment: IV levothyroxine loading dose 200–400 mcg, then 50–100 mcg/day IV + IV liothyronine (T3) 5–20 mcg q8h. Give IV hydrocortisone 100 mg q8h empirically (coexistent adrenal insufficiency must be excluded before thyroid replacement, or adrenal crisis may be precipitated). Supportive: passive rewarming, mechanical ventilation if needed, vasopressors.
12 Hyperthyroidism
Hyperthyroidism refers to excess thyroid hormone production by the thyroid gland. Thyrotoxicosis is the broader term for any cause of excess thyroid hormone (including exogenous intake or destructive thyroiditis without glandular overproduction).
Etiology & RAIU Patterns
| Condition | Mechanism | RAIU | Thyroid Exam | Key Features |
|---|---|---|---|---|
| Graves disease | TSH receptor-stimulating antibodies (TRAb/TSI) | Diffusely increased | Diffuse goiter, bruit | Ophthalmopathy, pretibial myxedema, acropachy; most common cause |
| Toxic multinodular goiter | Autonomous nodules | Heterogeneous / patchy uptake | Multiple palpable nodules | Older patients; insidious onset |
| Toxic adenoma | Single autonomous nodule | Hot nodule with suppressed surrounding | Single nodule | Rarely malignant |
| Subacute thyroiditis (de Quervain) | Viral-triggered destruction | Very low (<5%) | Tender, firm thyroid | Neck pain, elevated ESR; self-limited (thyrotoxic → hypothyroid → recovery) |
| Painless (silent) thyroiditis | Autoimmune destruction | Low | Non-tender, small goiter | Postpartum variant common; self-limited |
| Factitious thyrotoxicosis | Exogenous T4/T3 ingestion | Low | Normal/small, no goiter | Low thyroglobulin (vs elevated in thyroiditis) |
| Iodine-induced (Jod-Basedow) | Excess iodine in autonomous tissue | Low | Variable | After iodinated contrast or amiodarone |
Treatment Options
| Modality | Indications | Details |
|---|---|---|
| Antithyroid drugs (ATDs) | Graves disease (first-line in many countries); bridge to RAI/surgery | Methimazole preferred: start 10–30 mg/day, taper to 5–10 mg maintenance. PTU reserved for: 1st trimester pregnancy, thyroid storm, ATD allergy. PTU dose: 100–200 mg TID. Side effects: agranulocytosis (0.2–0.5% — check WBC if fever/sore throat), hepatotoxicity (PTU > MMI), rash |
| Radioactive iodine (RAI / I-131) | Graves disease (definitive), toxic MNG, toxic adenoma | Single oral dose. Contraindicated in pregnancy/breastfeeding and active Graves ophthalmopathy (can worsen — use steroid prophylaxis). Most patients become hypothyroid within 6–12 months |
| Surgery (thyroidectomy) | Large goiter, compression symptoms, suspicious nodule, patient preference, contraindication to RAI | Near-total or total thyroidectomy. Risks: recurrent laryngeal nerve injury (hoarseness), hypoparathyroidism (transient ~20%, permanent ~2%), bleeding/hematoma |
| Beta-blockers | Symptomatic control in all forms | Propranolol 20–40 mg TID (also inhibits T4→T3 conversion). Atenolol 25–100 mg/day if propranolol contraindicated |
13 Thyroid Nodules & Cancer
Thyroid nodules are extremely common (palpable in 5% of the population, incidental on imaging in 20–70%). The clinical challenge is identifying the ~5–15% of nodules that harbor malignancy. All patients with a thyroid nodule should have a TSH measured. If TSH is low, obtain a radioactive iodine uptake scan — a "hot" (hyperfunctioning) nodule is almost never malignant and does not require FNA. If TSH is normal or high, obtain a thyroid ultrasound to assess nodule features.
Ultrasound Features Suggesting Malignancy
Suspicious features: solid hypoechoic, microcalcifications, irregular margins, taller-than-wide shape, extrathyroidal extension, and suspicious cervical lymphadenopathy. The ATA risk stratification system (TI-RADS) guides FNA decisions based on ultrasound pattern and nodule size.
Bethesda Classification of Thyroid FNA Cytology
| Category | Diagnosis | Malignancy Risk | Management |
|---|---|---|---|
| I | Nondiagnostic / Unsatisfactory | 5–10% | Repeat FNA in 6–12 weeks |
| II | Benign | 0–3% | Follow-up US in 12–24 months |
| III | Atypia of undetermined significance (AUS) / Follicular lesion | 10–30% | Repeat FNA, molecular testing, or diagnostic lobectomy |
| IV | Follicular neoplasm / Suspicious for follicular neoplasm | 25–40% | Molecular testing or diagnostic lobectomy |
| V | Suspicious for malignancy | 50–75% | Near-total thyroidectomy or lobectomy |
| VI | Malignant | 97–99% | Surgery (total thyroidectomy for most cancers >1 cm) |
Thyroid Cancer Types
| Type | % of Cases | Pathology | Prognosis | Key Features |
|---|---|---|---|---|
| Papillary | 80–85% | Psammoma bodies, Orphan Annie nuclei, nuclear grooves | Excellent (10-yr survival >95%) | Spreads via lymphatics; RAI-responsive |
| Follicular | 10–15% | Capsular/vascular invasion required for diagnosis | Good (slightly worse than papillary) | Hematogenous spread (lung, bone); RAI-responsive |
| Medullary (MTC) | 3–5% | Parafollicular C cells; calcitonin marker | Moderate (10-yr ~75%) | 25% familial (MEN2A, MEN2B, FMTC); RET mutation; amyloid stroma |
| Anaplastic | 1–2% | Undifferentiated, rapidly growing | Dismal (median survival 5 months) | Elderly patients, rapidly enlarging neck mass, often inoperable |
14 Thyroid Storm
Thyroid storm is a rare, life-threatening exacerbation of thyrotoxicosis with multisystem decompensation. Mortality is 10–30% even with treatment. Triggers include surgery, infection, trauma, iodine load, DKA, and abrupt ATD discontinuation. Clinical features: high fever (>104°F/40°C), tachycardia out of proportion to fever (HR often >140), agitation/delirium/psychosis, GI symptoms (nausea, vomiting, diarrhea, jaundice), and cardiovascular collapse (atrial fibrillation, heart failure, shock).
Burch-Wartofsky Point Scale (BWPS)
| Parameter | Points |
|---|---|
| Temperature: 99–99.9°F (5), 100–100.9 (10), 101–101.9 (15), 102–102.9 (20), 103–103.9 (25), ≥104 (30) | 5–30 |
| CNS: Mild agitation (10), Moderate (delirium, psychosis) (20), Severe (seizures, coma) (30) | 0–30 |
| GI-hepatic: Diarrhea/nausea (10), Jaundice (20) | 0–20 |
| HR: 100–109 (5), 110–119 (10), 120–129 (15), 130–139 (20), ≥140 (25) | 0–25 |
| Heart failure: Mild (5), Moderate (10), Severe/pulmonary edema (15) | 0–15 |
| Atrial fibrillation: Present (10) | 0–10 |
| Precipitant: Present (10) | 0–10 |
Score: ≥45 = thyroid storm; 25–44 = impending storm; <25 = storm unlikely.
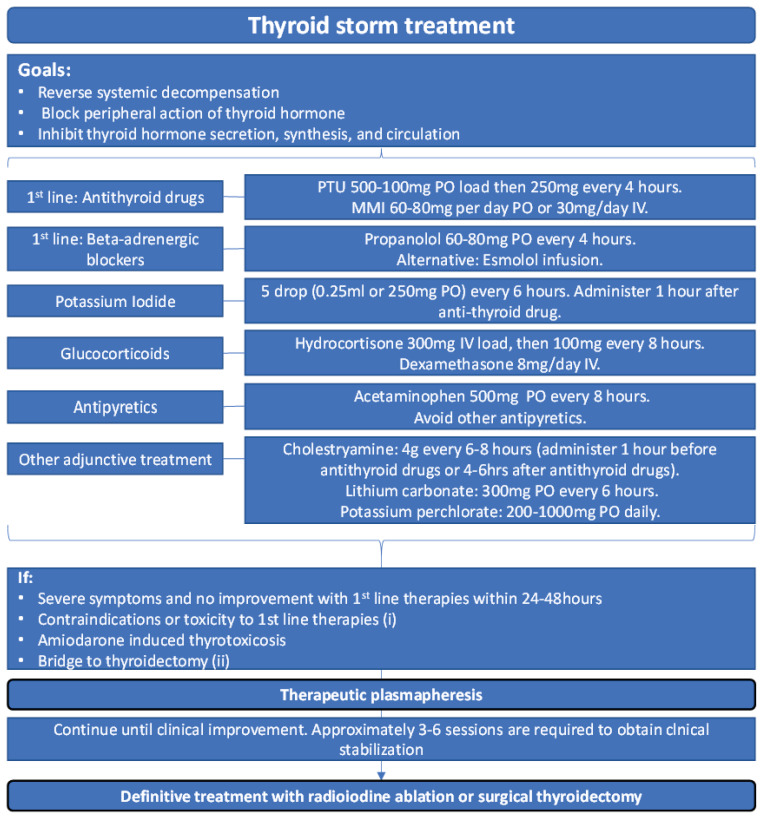
Thyroid Storm Treatment Protocol (Order Matters)
| Step | Agent | Dose | Purpose |
|---|---|---|---|
| 1 | Beta-blocker | Propranolol 60–80 mg PO q4h (or esmolol drip) | Blocks adrenergic symptoms + inhibits T4→T3 conversion |
| 2 | Thionamide (PTU preferred) | PTU 200–300 mg PO/NG q4–6h (or methimazole 20–25 mg q4h) | Blocks new hormone synthesis; PTU also blocks peripheral T4→T3 |
| 3 | Iodine (give 1 hour AFTER PTU) | SSKI 5 drops PO q6h or Lugol's solution 8–10 drops q8h | Blocks thyroid hormone release (Wolff-Chaikoff effect) |
| 4 | Glucocorticoid | Hydrocortisone 100 mg IV q8h (or dexamethasone 2 mg IV q6h) | Inhibits T4→T3 conversion; treats possible relative adrenal insufficiency |
| 5 | Supportive care | Cooling blankets (avoid ASA — displaces T4 from TBG), IV fluids, treat precipitant | Temperature control, hemodynamic support |
15 Adrenal Anatomy & Hormone Synthesis
The adrenal glands are paired retroperitoneal organs sitting atop each kidney, weighing ~4 g each. Each gland has two functionally distinct regions: the outer cortex (mesoderm-derived, produces steroid hormones) and the inner medulla (neural crest-derived, produces catecholamines).
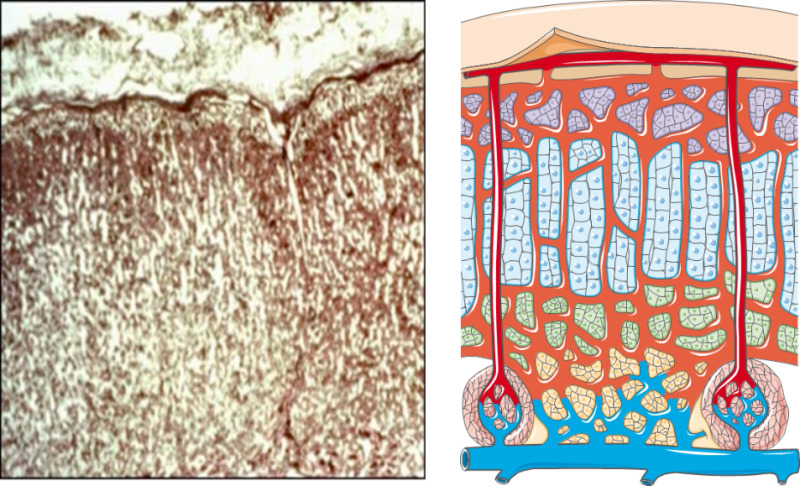
Adrenal Cortex Zones
| Zone | Mnemonic | Hormones | Regulation | Key Enzyme |
|---|---|---|---|---|
| Zona Glomerulosa | "Salt" | Aldosterone (mineralocorticoid) | Renin-angiotensin-aldosterone system (RAAS), K+ | Aldosterone synthase (CYP11B2) |
| Zona Fasciculata | "Sugar" | Cortisol (glucocorticoid) | ACTH (HPA axis) | 11β-hydroxylase (CYP11B1) |
| Zona Reticularis | "Sex" | DHEA, DHEA-S, androstenedione | ACTH | 17,20-lyase |
| Medulla | — | Epinephrine (80%), norepinephrine (20%) | Sympathetic preganglionic neurons | PNMT (converts NE to Epi) |

Cortisol Physiology
Cortisol follows a diurnal rhythm: peak at 6–8 AM, nadir at midnight. This is critical for interpreting cortisol levels and designing dynamic tests. Cortisol circulates 90% bound to cortisol-binding globulin (CBG/transcortin) and ~6% to albumin; only ~4% is free (biologically active). Conditions that alter CBG (estrogen/OCPs increase CBG; nephrotic syndrome/cirrhosis decrease CBG) affect total cortisol levels without changing the free cortisol.
16 Cushing Syndrome
Cushing syndrome refers to the clinical manifestations of chronic glucocorticoid excess from any cause. Cushing disease specifically refers to ACTH-secreting pituitary adenoma (the most common endogenous cause, ~70%). The most common overall cause is exogenous glucocorticoid use (iatrogenic Cushing).
Classification
| Category | ACTH Level | Cause | Frequency |
|---|---|---|---|
| ACTH-dependent (~80%) | Elevated or inappropriately normal | Cushing disease (pituitary adenoma) | ~70% |
| Ectopic ACTH (SCLC, carcinoid, MTC) | ~10% | ||
| ACTH-independent (~20%) | Suppressed (<5 pg/mL) | Adrenal adenoma | ~10% |
| Adrenal carcinoma, bilateral hyperplasia | ~10% |
Screening Tests (Need ≥2 Positive)
| Test | Protocol | Positive Result | Sensitivity |
|---|---|---|---|
| 24-hr urinary free cortisol (UFC) | Collect 24-hr urine; confirm adequate collection (Cr) | >3× upper limit of normal | ~95% |
| Late-night salivary cortisol | Collect saliva at 11 PM (2 samples) | Elevated (loss of diurnal nadir) | ~95% |
| 1 mg overnight DST | Dexamethasone 1 mg PO at 11 PM; measure AM cortisol at 8 AM | Cortisol >1.8 mcg/dL (failure to suppress) | ~98% |
Localization
After confirming hypercortisolism, check ACTH. If ACTH-dependent: pituitary MRI + high-dose (8 mg) dexamethasone suppression test or CRH stimulation test to distinguish pituitary (Cushing disease) from ectopic ACTH. If results are equivocal: bilateral inferior petrosal sinus sampling (BIPSS) is the gold standard — a central-to-peripheral ACTH gradient >2 (basal) or >3 (after CRH) confirms pituitary source. If ACTH-independent: adrenal CT to identify adenoma or carcinoma.
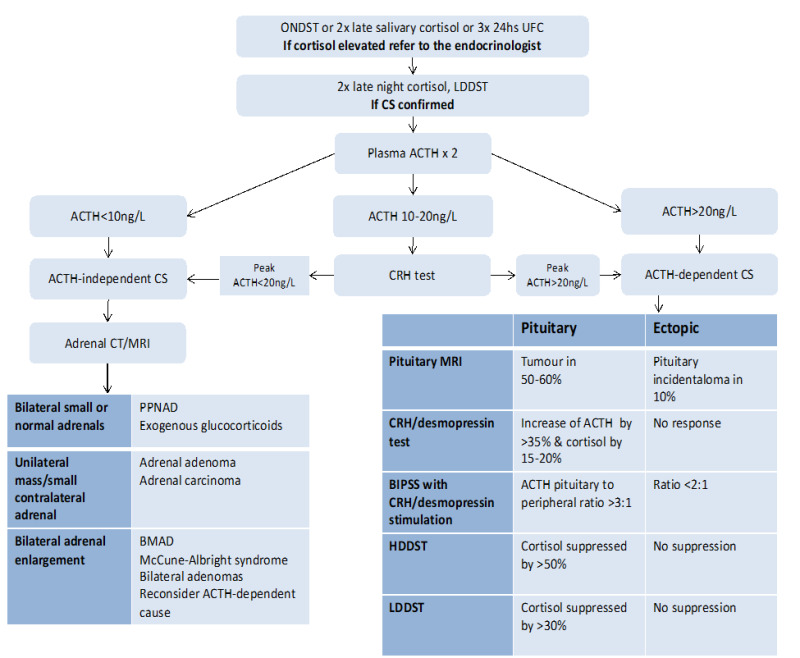
17 Adrenal Insufficiency
Adrenal crisis is a life-threatening emergency caused by acute cortisol deficiency. Triggers: abrupt steroid withdrawal, physiologic stress (illness, surgery, trauma) in patients with undiagnosed or undertreated adrenal insufficiency, pituitary apoplexy, or bilateral adrenal hemorrhage (Waterhouse-Friderichsen syndrome in meningococcemia). Presents with: refractory hypotension, shock, abdominal pain mimicking an acute abdomen, fever, hyponatremia, hyperkalemia (primary AI), and hypoglycemia. Treatment: IV hydrocortisone 100 mg stat, then 50 mg q6–8h + aggressive IV normal saline resuscitation. Do NOT delay treatment for confirmatory testing.
Primary vs Secondary vs Tertiary
| Type | Level | Common Causes | Cortisol | ACTH | Aldosterone | Key Features |
|---|---|---|---|---|---|---|
| Primary (Addison disease) | Adrenal gland | Autoimmune (80% in developed countries), TB, adrenal hemorrhage, infiltrative, drugs (ketoconazole, etomidate) | ↓ | ↑↑ | ↓ | Hyperpigmentation (elevated ACTH/MSH), hyperkalemia, salt craving, vitiligo |
| Secondary | Pituitary | Pituitary tumors, surgery, Sheehan syndrome, infiltrative | ↓ | ↓ or normal | Normal | No hyperpigmentation, no hyperkalemia (aldosterone intact via RAAS) |
| Tertiary | Hypothalamic / Iatrogenic | Chronic exogenous glucocorticoid use (most common cause overall) | ↓ | ↓ or normal | Normal | HPA axis suppression after ≥3 weeks of supraphysiologic steroids |
Diagnosis
Morning cortisol (8 AM): <3 mcg/dL is diagnostic of AI; >18 mcg/dL effectively rules it out. For values 3–18, perform a cosyntropin (ACTH) stimulation test: give cosyntropin 250 mcg IV/IM, measure cortisol at 30 and 60 minutes. A stimulated cortisol <18 mcg/dL confirms adrenal insufficiency. Note: this test may be falsely normal in recent-onset secondary AI (adrenals have not yet atrophied).
Replacement Therapy
| Hormone | Replacement | Dose | Monitoring |
|---|---|---|---|
| Glucocorticoid | Hydrocortisone | 15–25 mg/day in 2–3 divided doses (10 mg AM, 5 mg noon, 5 mg PM) | Clinical (energy, weight, BP); no reliable lab marker |
| Mineralocorticoid (primary AI only) | Fludrocortisone | 0.05–0.2 mg/day | Orthostatic BP, K+, renin (target high-normal PRA) |
| DHEA (optional, women with primary AI) | DHEA | 25–50 mg/day | DHEA-S levels |
Stress Dosing (Sick Day Rules)
| Stress Level | Example | Steroid Adjustment |
|---|---|---|
| Minor illness (cold, low-grade fever) | URI, mild GI illness | Double oral dose for 2–3 days |
| Moderate illness | Fever >101°F, influenza, dental extraction | Triple oral dose for 2–3 days |
| Major stress / surgery | Trauma, major surgery, critical illness | Hydrocortisone 100 mg IV, then 50 mg q6–8h; taper over days |
| Vomiting (cannot take PO) | — | IM hydrocortisone 100 mg; proceed to ED |
18 Primary Aldosteronism
Primary aldosteronism (PA) is autonomous aldosterone production independent of renin. It is now recognized as the most common cause of secondary hypertension, present in 5–10% of all hypertensives and ~20% of those with resistant hypertension. It causes hypertension plus hypokalemia (though ~50% of patients are normokalemic).
Screening
Screen with the aldosterone-to-renin ratio (ARR). Positive screening: aldosterone >15 ng/dL AND ARR >30. Potassium-sparing diuretics (spironolactone, eplerenone) and MRA must be held 4–6 weeks before testing. Beta-blockers suppress renin (false-positive ARR); ACE inhibitors/ARBs increase renin (false-negative).
Who to Screen
Resistant hypertension (3+ drugs including a diuretic), hypertension with spontaneous or diuretic-induced hypokalemia, hypertension with adrenal incidentaloma, hypertension onset <30 years, severe hypertension (≥150/100 on 2+ agents), and family history of early-onset hypertension or cerebrovascular accident <40.
Confirmatory Testing
| Test | Protocol | Positive Result |
|---|---|---|
| Oral sodium loading | High-sodium diet (>200 mEq/day) for 3 days, then 24-hr urine | Urinary aldosterone >12 mcg/24h despite sodium loading |
| IV saline infusion | 2 L NS over 4 hours | Plasma aldosterone >10 ng/dL post-infusion |
| Fludrocortisone suppression | Fludrocortisone 0.1 mg q6h × 4 days | Upright aldosterone >6 ng/dL on day 4 |
Subtype Differentiation
After confirmation, obtain adrenal CT. If bilateral disease or equivocal imaging: adrenal vein sampling (AVS) is the gold standard to lateralize (unilateral adenoma vs bilateral hyperplasia). Lateralization ratio >4:1 indicates unilateral disease. Unilateral adenoma → laparoscopic adrenalectomy (cure rate ~50–70% for hypertension, ~100% for hypokalemia). Bilateral hyperplasia → mineralocorticoid receptor antagonist (spironolactone 25–100 mg/day or eplerenone 50–200 mg/day).
19 Pheochromocytoma & Paraganglioma
Pheochromocytomas (pheos) are catecholamine-secreting tumors arising from chromaffin cells of the adrenal medulla. Paragangliomas arise from extra-adrenal sympathetic or parasympathetic paraganglia. Together they are called PPGLs. Classic presentation: episodic headache, sweating, palpitations, and hypertension (sustained or paroxysmal). The classic triad (headache + sweating + tachycardia) in a hypertensive patient has ~90% specificity.
The "Rule of 10s" (Now Outdated)
The traditional teaching was: 10% bilateral, 10% extra-adrenal, 10% malignant, 10% familial, 10% in children. Current evidence shows up to 40% are hereditary (genetic testing is now recommended for all patients). Key syndromes: MEN2A/2B (RET), von Hippel-Lindau (VHL), neurofibromatosis type 1 (NF1), and SDHx mutations (succinate dehydrogenase subunits — associated with paragangliomas and malignancy).
Biochemical Workup
| Test | Sensitivity | Specificity | Notes |
|---|---|---|---|
| Plasma free metanephrines | 96–99% | 85–89% | Best screening test; must draw supine after 20 min rest |
| 24-hr urine metanephrines + catecholamines | 90–95% | 95–98% | Alternative if plasma unavailable; higher specificity |
| Plasma catecholamines | Lower | Lower | Not recommended as sole screening (episodic secretion) |
Imaging & Localization
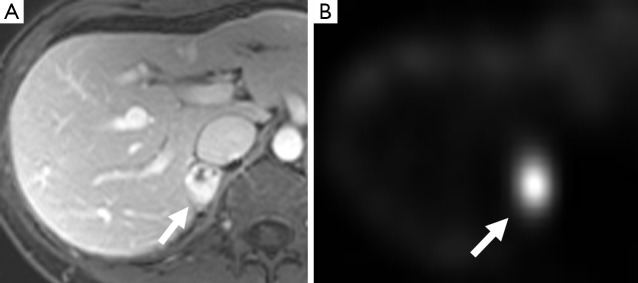
CT abdomen/pelvis with contrast (or MRI — pheos are classically "bright" on T2-weighted MRI) for initial localization. For extra-adrenal, metastatic, or recurrent disease: MIBG scan (I-123/I-131 metaiodobenzylguanidine) or Ga-68 DOTATATE PET/CT (superior sensitivity for SDHx-related tumors).
Preoperative Management
| Step | Agent | Details |
|---|---|---|
| 1. Alpha-blockade (start 10–14 days preop) | Phenoxybenzamine 10 mg BID, titrate to 20–30 mg BID (or doxazosin 2–16 mg/day) | Goal: seated BP <130/80, standing SBP >90, no orthostatic hypotension limiting function |
| 2. Volume expansion | Liberal salt diet + fluids | Counteract chronic vasoconstriction-induced volume depletion |
| 3. Beta-blockade (add AFTER alpha) | Propranolol or atenolol | NEVER give beta-blocker before adequate alpha-blockade (unopposed alpha stimulation → hypertensive crisis) |
| 4. Metyrosine (optional) | Metyrosine 250 mg QID | Inhibits catecholamine synthesis; used if alpha-blockade insufficient |
20 Pituitary Anatomy & Hormones
The pituitary gland (hypophysis) sits in the sella turcica of the sphenoid bone, connected to the hypothalamus by the pituitary stalk (infundibulum). It weighs ~0.6 g and has two lobes with distinct embryologic origins and functions.
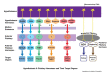
Anterior Pituitary (Adenohypophysis)
| Cell Type | Hormone | Hypothalamic Control | Target | % of Cells |
|---|---|---|---|---|
| Somatotroph | GH (growth hormone) | GHRH (+), Somatostatin (−) | Liver (IGF-1), bone, muscle | ~50% |
| Lactotroph | Prolactin | Dopamine (−), TRH (+) | Breast | ~15–25% |
| Corticotroph | ACTH | CRH (+) | Adrenal cortex | ~15–20% |
| Thyrotroph | TSH | TRH (+), Somatostatin (−) | Thyroid | ~5% |
| Gonadotroph | LH, FSH | GnRH (+) | Ovaries, testes | ~10% |
Posterior Pituitary (Neurohypophysis)
The posterior pituitary does not synthesize hormones. It stores and releases oxytocin and ADH (vasopressin), which are synthesized in hypothalamic nuclei (paraventricular and supraoptic, respectively) and transported down axons via the stalk. ADH acts on V2 receptors in the collecting duct to insert aquaporin-2 channels, promoting water reabsorption.
21 Pituitary Adenomas
Pituitary adenomas account for ~15% of intracranial neoplasms. They are classified by size (microadenoma <10 mm, macroadenoma ≥10 mm) and by functional status (secreting vs non-functioning). The most common incidental finding on brain MRI is a pituitary microadenoma (~10% prevalence in autopsy series).

Types of Functioning Pituitary Adenomas
| Type | Frequency | Hormone | Clinical Syndrome | First-Line Treatment |
|---|---|---|---|---|
| Prolactinoma | ~40% (most common) | Prolactin | Women: amenorrhea, galactorrhea, infertility. Men: decreased libido, erectile dysfunction, gynecomastia | Dopamine agonist (cabergoline preferred over bromocriptine — more effective, fewer side effects) |
| GH-secreting | ~20% | GH / IGF-1 | Acromegaly (adults), gigantism (children). Coarsened features, enlarged hands/feet, OSA, carpal tunnel, diabetes, cardiomegaly | Transsphenoidal surgery (first-line); somatostatin analogs (octreotide, lanreotide) if not cured |
| ACTH-secreting | ~5–10% | ACTH | Cushing disease | Transsphenoidal surgery; medical therapy (ketoconazole, osilodrostat, pasireotide) if not cured |
| TSH-secreting | <1% (rare) | TSH | Central hyperthyroidism (elevated FT4 with non-suppressed TSH) | Transsphenoidal surgery; somatostatin analogs |
| Non-functioning | ~30% | None (or clinically silent) | Mass effect: bitemporal hemianopsia, headache, hypopituitarism | Surgery for macroadenomas with mass effect; observation for incidental microadenomas |
Any mass compressing the pituitary stalk can cause mild hyperprolactinemia (typically PRL <100–200 ng/mL) by interrupting dopaminergic inhibition of prolactin. This "stalk effect" must be distinguished from a true prolactinoma: PRL >200 ng/mL generally indicates prolactinoma (PRL level correlates with tumor size). A macroadenoma with PRL of only 50–80 ng/mL is more likely a non-functioning adenoma with stalk effect than a prolactinoma. This distinction is critical because prolactinomas are treated medically (dopamine agonists), while non-functioning adenomas require surgery.
Acromegaly Diagnosis
Screen with IGF-1 (elevated, age- and sex-adjusted). Confirm with oral glucose tolerance test (OGTT): GH should suppress to <1 ng/mL (or <0.4 with ultrasensitive assay) after 75 g glucose load; failure to suppress confirms autonomous GH secretion. MRI pituitary for localization.
22 Hypopituitarism
Hypopituitarism is partial or complete loss of anterior pituitary hormone secretion. It can result from pituitary tumors (most common), surgery, radiation, infiltrative diseases (sarcoidosis, hemochromatosis, Langerhans cell histiocytosis), Sheehan syndrome (postpartum pituitary necrosis from hemorrhagic shock), pituitary apoplexy (hemorrhage into an adenoma), traumatic brain injury, or autoimmune hypophysitis (including from immune checkpoint inhibitors).
Order of Hormone Loss
When the pituitary is compressed by a gradually enlarging mass, hormones are typically lost in a predictable sequence: GH → LH/FSH → TSH → ACTH → Prolactin. GH is the most vulnerable (often first deficit); ACTH is the most critical (last to be lost but most dangerous if deficient). Prolactin deficiency is rare and only occurs with severe pituitary destruction.
Hormone Replacement Hierarchy
| Priority | Deficiency | Replacement | Critical Note |
|---|---|---|---|
| 1 (Most critical) | ACTH / Cortisol | Hydrocortisone 15–25 mg/day | Must replace BEFORE thyroid hormone (LT4 accelerates cortisol metabolism → adrenal crisis) |
| 2 | TSH / Thyroid | Levothyroxine (dose by FT4, NOT TSH) | Monitor with FT4 (TSH is unreliable in central hypothyroidism) |
| 3 | LH, FSH / Gonadal | Testosterone (men), Estrogen + progesterone (women), or gonadotropins if fertility desired | — |
| 4 | GH | rhGH 0.2–0.5 mg/day SQ; titrate by IGF-1 | Improves body composition, bone density, quality of life |
| 5 | ADH (if posterior involved) | Desmopressin (DDAVP) | Central DI from stalk/posterior pituitary involvement |
Sudden hemorrhage or infarction of a pituitary adenoma. Presents with: sudden severe headache ("thunderclap"), visual loss (chiasmal compression), ophthalmoplegia (cavernous sinus compression of CN III, IV, VI), and acute adrenal crisis. Treatment: immediate IV hydrocortisone 100 mg + urgent neurosurgical consultation for transsphenoidal decompression if visual loss or altered consciousness. Must distinguish from SAH (similar headache presentation).
23 Diabetes Insipidus & SIADH
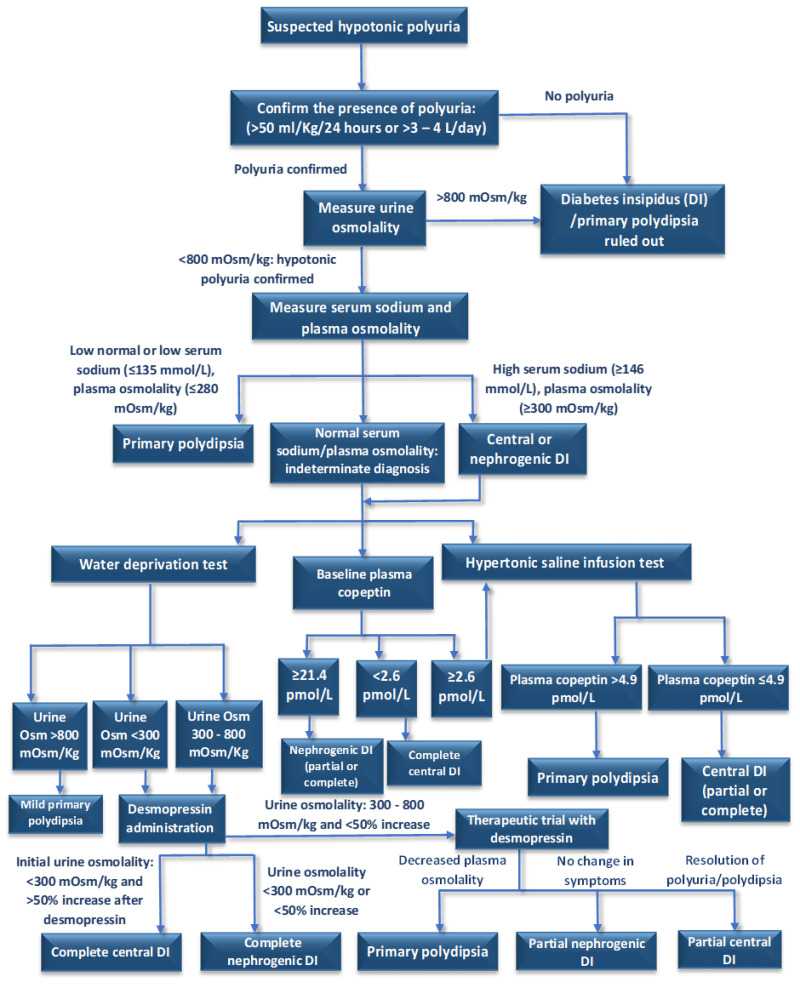
Diabetes Insipidus (DI)
DI is characterized by the excretion of large volumes of dilute urine (polyuria >3 L/day, urine osmolality <300 mOsm/kg) due to ADH deficiency or resistance. Patients present with polyuria, polydipsia, nocturia, and if water intake is inadequate, hypernatremia and dehydration.
| Type | Mechanism | Common Causes | ADH Level | Response to DDAVP |
|---|---|---|---|---|
| Central DI | Insufficient ADH production/secretion | Post-pituitary surgery (most common), tumors, infiltrative disease, trauma, idiopathic | Low | Urine concentrates (>50% increase in Uosm) |
| Nephrogenic DI | Kidney resistance to ADH | Lithium (most common drug cause), hypercalcemia, hypokalemia, chronic kidney disease, genetic (V2 receptor or AQP2 mutations) | High | No significant response |
| Primary polydipsia | Excessive water intake suppresses ADH | Psychiatric illness (psychogenic polydipsia), medications | Low (appropriate) | Not indicated |
Water Deprivation Test
Gold standard for diagnosing and differentiating DI. Patient is fluid-restricted under observation with serial measurements of body weight, urine osmolality, serum osmolality, and serum sodium. Urine osmolality is measured until either: urine concentrates normally (>600 mOsm/kg, ruling out DI), or serum osmolality exceeds 295–300 or Na+ >145 (confirming DI). Then administer DDAVP 2 mcg IV/SQ and measure urine osmolality after 1–2 hours. Central DI: urine concentrates >50%. Nephrogenic DI: minimal response (<50% increase or Uosm remains <300).
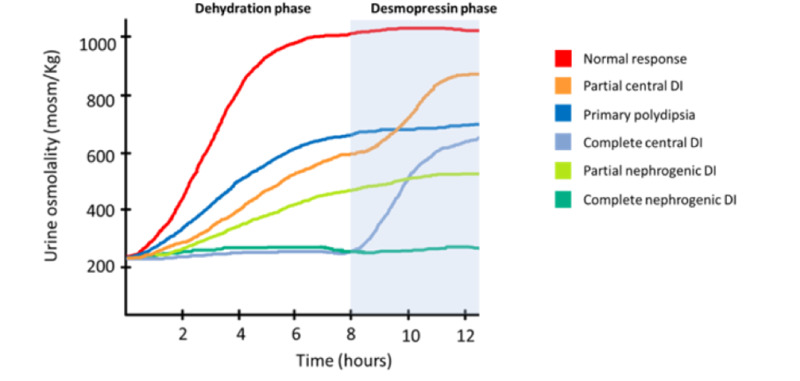
Treatment of DI
Central DI: Desmopressin (DDAVP) — intranasal 10–20 mcg BID, oral 0.1–0.4 mg BID–TID, or SQ 1–2 mcg BID. Titrate to control polyuria without hyponatremia. Nephrogenic DI: Remove offending agent if possible. Thiazide diuretics (paradoxically reduce urine output by promoting proximal reabsorption), amiloride (especially for lithium-induced — blocks lithium entry via ENaC), low-sodium diet, and NSAIDs (reduce renal prostaglandins).
SIADH
Syndrome of inappropriate ADH secretion causes euvolemic hypotonic hyponatremia. ADH is secreted despite low serum osmolality, leading to water retention, dilutional hyponatremia, and concentrated urine.
| Category | Causes |
|---|---|
| CNS | Stroke, meningitis, encephalitis, traumatic brain injury, SAH |
| Pulmonary | Pneumonia, TB, lung abscess, positive pressure ventilation |
| Malignancy | Small-cell lung cancer (most common malignant cause), head/neck cancers |
| Medications | SSRIs, carbamazepine, cyclophosphamide, vincristine, desmopressin, oxytocin |
| Other | Pain, nausea, postoperative state, HIV |
SIADH Diagnostic Criteria
All of the following: serum osmolality <275 mOsm/kg, urine osmolality >100 mOsm/kg (inappropriately concentrated), urine sodium >40 mEq/L, euvolemic, normal thyroid and adrenal function, no diuretic use.
SIADH Management
Mild/chronic: Fluid restriction (800–1000 mL/day) is first-line. Salt tablets + loop diuretic if fluid restriction insufficient. Vaptans (tolvaptan 15–60 mg PO daily) block V2 receptors — promote free water excretion. Severe/symptomatic (seizures, coma, Na+ <120): hypertonic saline (3% NaCl) with goal correction of Na+ by 4–6 mEq/L in first 6 hours, maximum 8 mEq/L in 24 hours to prevent osmotic demyelination syndrome (ODS, formerly central pontine myelinolysis).
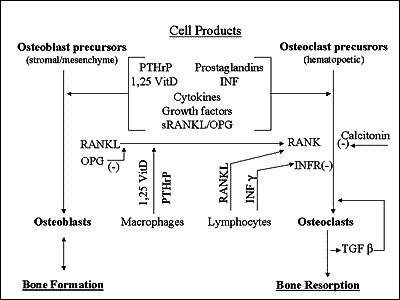
24 Calcium Homeostasis
Calcium is tightly regulated between 8.5–10.5 mg/dL (total) or 4.5–5.5 mg/dL (ionized). Approximately 40% of total calcium is protein-bound (mainly albumin), 10% is complexed with anions, and 50% is free/ionized (biologically active). The corrected calcium adjusts for hypoalbuminemia: add 0.8 mg/dL for each 1 g/dL albumin below 4.0.
Key Hormones
| Hormone | Source | Stimulus | Actions | Net Effect on Ca2+ |
|---|---|---|---|---|
| PTH | Parathyroid glands (chief cells) | Low ionized calcium | Increases bone resorption, increases renal Ca2+ reabsorption, stimulates 1-alpha-hydroxylase (activates vitamin D) | ↑ Calcium |
| Vitamin D (1,25-dihydroxy) | Kidneys (activated from 25-OH-D) | PTH, low phosphate | Increases intestinal Ca2+ and phosphate absorption, promotes bone mineralization | ↑ Calcium |
| Calcitonin | Thyroid C cells (parafollicular) | High calcium | Inhibits osteoclast activity, increases renal Ca2+ excretion | ↓ Calcium (clinically modest) |
| PTHrP | Various tumors | Paraneoplastic | Mimics PTH at PTH1 receptor | ↑ Calcium (humoral hypercalcemia of malignancy) |
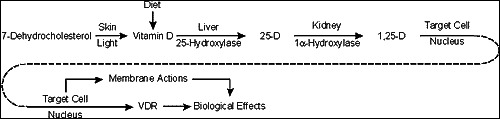
Vitamin D Metabolism
Vitamin D3 (cholecalciferol, from skin UV exposure or diet) and D2 (ergocalciferol, from plants) are hydroxylated in the liver to 25-hydroxyvitamin D (calcidiol) — the storage form and best marker of vitamin D status. This is then hydroxylated in the kidney by 1-alpha-hydroxylase (stimulated by PTH, low phosphate, and low calcium) to 1,25-dihydroxyvitamin D (calcitriol) — the active hormonal form.
25 Hyperparathyroidism
Primary Hyperparathyroidism (PHPT)
The most common cause of hypercalcemia in the outpatient setting. Etiology: single parathyroid adenoma (~85%), four-gland hyperplasia (~10–15%, often MEN1 or MEN2A), parathyroid carcinoma (<1%). Lab pattern: elevated calcium + elevated or inappropriately normal PTH. Additional findings: low phosphate (PTH is phosphaturic), elevated urine calcium, elevated 1,25-vitamin D.
Surgical Indications (2022 Guidelines)
| Criterion | Threshold for Surgery |
|---|---|
| Serum calcium | >1 mg/dL above upper limit of normal |
| Bone density | T-score ≤−2.5 at any site, or vertebral fracture |
| Renal | CrCl <60 mL/min, 24-hr urine Ca >400 mg/day, or nephrolithiasis/nephrocalcinosis |
| Age | <50 years |
| Monitoring not feasible | Patient unable to follow up |
Familial Hypocalciuric Hypercalcemia (FHH)
FHH is an autosomal dominant condition caused by inactivating mutations of the calcium-sensing receptor (CaSR). Lab pattern mimics PHPT (elevated calcium, non-suppressed PTH) but urine calcium is LOW. The key discriminating test: calcium-to-creatinine clearance ratio (CCCR) <0.01 suggests FHH; >0.02 suggests PHPT. FHH is benign and does NOT require parathyroidectomy.
Secondary & Tertiary Hyperparathyroidism
Secondary HPT: Physiologic PTH elevation in response to chronic hypocalcemia (most commonly CKD, also vitamin D deficiency). Calcium is low or normal; PTH is high. Treat the underlying cause (phosphate binders, calcitriol, calcimimetics in CKD). Tertiary HPT: Autonomous PTH secretion from prolonged secondary HPT (parathyroid hyperplasia becomes autonomous). Seen post-kidney transplant. Calcium becomes elevated despite correction of the original stimulus. May require parathyroidectomy.
26 Hypoparathyroidism & Hypocalcemia
Causes of Hypocalcemia
| Category | Causes | PTH | Notes |
|---|---|---|---|
| Hypoparathyroidism | Post-surgical (most common — after thyroidectomy/parathyroidectomy), autoimmune (APS type 1), DiGeorge syndrome, infiltrative | Low | Also causes hyperphosphatemia |
| Vitamin D deficiency | Malabsorption, CKD, inadequate intake/sunlight, liver disease | High (secondary HPT) | Low 25-OH vitamin D |
| Pseudohypoparathyroidism | PTH resistance (Gs-alpha mutation — Albright hereditary osteodystrophy) | High | Short stature, round facies, short 4th/5th metacarpals, subcutaneous calcifications |
| Acute pancreatitis | Saponification of calcium with fatty acids | High | Severity marker |
| Hyperphosphatemia | Rhabdomyolysis, tumor lysis syndrome, renal failure | Variable | Ca × PO4 product elevated |
| Hungry bone syndrome | After parathyroidectomy for severe HPT | Low | Rapid bone uptake of Ca2+; may last weeks |
Clinical Signs of Hypocalcemia
Chvostek sign: Tapping the facial nerve (anterior to ear, below zygomatic arch) causes ipsilateral facial muscle twitch. Sensitivity ~10–70% (also positive in ~10% of normals). Trousseau sign: Inflate BP cuff above systolic for 3 minutes → carpal spasm (wrist flexion, metacarpophalangeal flexion, thumb adduction). More specific (~94%) than Chvostek. Other symptoms: perioral and fingertip paresthesias, muscle cramps, tetany, laryngospasm, seizures, QTc prolongation.
Acute Severe Hypocalcemia Treatment
IV calcium gluconate 1–2 g (10–20 mL of 10% solution) over 10–20 minutes, followed by continuous infusion (calcium gluconate 60–80 mL of 10% in 1 L D5W at 0.5–2 mg/kg/hr elemental Ca). Monitor ionized calcium every 4–6 hours. Simultaneously correct hypomagnesemia (magnesium is required for PTH secretion — hypomagnesemia causes functional hypoparathyroidism). Use calcium gluconate peripherally; calcium chloride is reserved for central lines (causes severe tissue necrosis if extravasated).
27 Osteoporosis & Metabolic Bone Disease
Osteoporosis is a systemic skeletal disease characterized by low bone mass and microarchitectural deterioration, leading to increased fracture risk. It is defined by DEXA T-score or by the occurrence of a fragility fracture.
DEXA Interpretation
| T-Score | Classification | Definition |
|---|---|---|
| ≥−1.0 | Normal | — |
| −1.0 to −2.5 | Osteopenia (low bone mass) | — |
| ≤−2.5 | Osteoporosis | At lumbar spine, femoral neck, total hip, or 1/3 radius |
| ≤−2.5 + fragility fracture | Severe osteoporosis | — |
T-score compares to young adult peak bone mass (used for postmenopausal women and men ≥50). Z-score compares to age-matched controls (used for premenopausal women, men <50, and children). Z-score ≤−2.0 warrants workup for secondary causes.
FRAX (Fracture Risk Assessment Tool)
FRAX estimates 10-year probability of major osteoporotic fracture and hip fracture using clinical risk factors ± femoral neck BMD. Treatment thresholds (NOF/AACE): ≥20% major fracture risk or ≥3% hip fracture risk.
Pharmacotherapy
| Class | Agent | Mechanism | Route / Dosing | Key Considerations |
|---|---|---|---|---|
| Bisphosphonate | Alendronate | Antiresorptive (osteoclast apoptosis) | 70 mg PO weekly | Take fasting with water, remain upright 30 min (esophagitis). Drug holiday after 5 yr PO / 3 yr IV |
| Bisphosphonate | Zoledronic acid | Antiresorptive | 5 mg IV once yearly | Monitor renal function; contraindicated if CrCl <35 |
| RANKL inhibitor | Denosumab | Antiresorptive (blocks RANKL → inhibits osteoclasts) | 60 mg SQ every 6 months | Rebound vertebral fractures if discontinued — must transition to bisphosphonate |
| PTH analog | Teriparatide | Anabolic (stimulates osteoblasts) | 20 mcg SQ daily × 2 years max | Contraindicated in Paget disease, bone mets, prior radiation, hypercalcemia. Black box: osteosarcoma (in rats) |
| PTHrP analog | Abaloparatide | Anabolic | 80 mcg SQ daily × 2 years | Similar to teriparatide; possibly less hypercalcemia |
| Sclerostin inhibitor | Romosozumab | Dual action (anabolic + antiresorptive) | 210 mg SQ monthly × 12 months | Black box: CV risk (MI, stroke); avoid if recent MI/stroke |
Paget Disease of Bone
Paget disease is characterized by excessive, disorganized bone remodeling. Most patients are asymptomatic (elevated alkaline phosphatase found incidentally). Symptomatic disease: bone pain, deformity (bowing of long bones, skull enlargement), pathologic fractures, hearing loss (temporal bone involvement), and rarely high-output heart failure or osteosarcoma (<1%). Labs: markedly elevated ALP, normal calcium/phosphate. Treatment: bisphosphonates (zoledronic acid preferred) for symptomatic disease or elevated ALP.
28 Male Hypogonadism
Male hypogonadism is defined by low testosterone with associated symptoms. Symptoms include decreased libido, erectile dysfunction, fatigue, loss of muscle mass, increased body fat, decreased bone density, depressed mood, and reduced facial/body hair.
Classification
| Type | Level | Testosterone | LH/FSH | Common Causes |
|---|---|---|---|---|
| Primary (hypergonadotropic) | Testes | Low | High | Klinefelter syndrome (47,XXY — most common genetic cause), orchitis, trauma, chemotherapy, radiation |
| Secondary (hypogonadotropic) | Pituitary / Hypothalamus | Low | Low or normal | Pituitary adenoma, hyperprolactinemia, Kallmann syndrome (anosmia + hypogonadism), obesity, opioids, exogenous testosterone/anabolic steroids |
Diagnosis
Measure morning total testosterone (8–10 AM, fasting) on two separate occasions. Low total testosterone (<300 ng/dL) requires confirmation. If borderline, check free testosterone (calculated from total T, SHBG, and albumin). Obtain LH, FSH to classify primary vs secondary. In secondary hypogonadism: check prolactin, iron saturation (hemochromatosis), and pituitary MRI if indicated.
Testosterone Replacement
| Formulation | Dose | Route | Notes |
|---|---|---|---|
| Testosterone cypionate/enanthate | 100–200 mg every 1–2 weeks | IM | Most common; peaks and troughs |
| Testosterone gel | 50–100 mg/day | Topical | Steady levels; risk of transference to women/children |
| Testosterone patch | 2–6 mg/day | Transdermal | Skin irritation common |
| Testosterone undecanoate | 750 mg q10 weeks (after loading) | IM | Long-acting; REMS due to pulmonary oil microembolism risk |
| Nasal testosterone | 11 mg TID | Intranasal | No transference risk |
Monitoring on TRT
Check testosterone levels (trough for IM, any time for gel), hematocrit (target <54% — erythrocytosis is the most common adverse effect), PSA and DRE, lipid panel, and bone density (if osteoporosis). Contraindications: active desire for fertility (exogenous T suppresses gonadotropins and spermatogenesis), polycythemia vera, untreated severe OSA, severe heart failure, PSA >4 without urologic evaluation, breast or prostate cancer.
29 PCOS
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in women of reproductive age, affecting 6–12%. It is a leading cause of anovulatory infertility and is strongly associated with insulin resistance, metabolic syndrome, and type 2 diabetes.
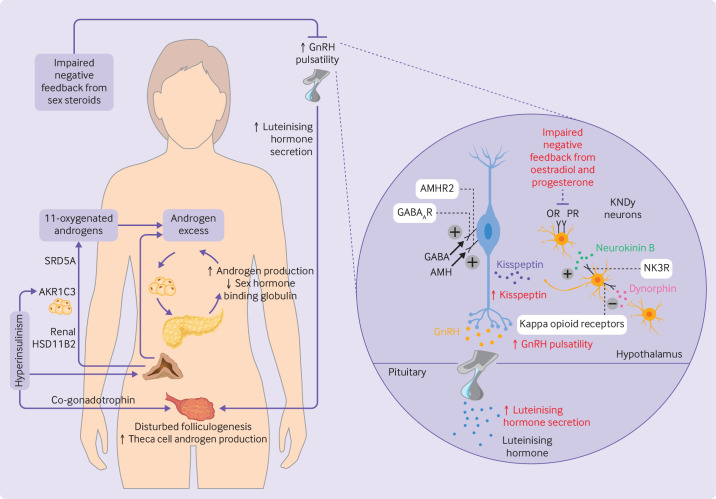
Rotterdam Diagnostic Criteria (Need 2 of 3)
| # | Criterion | Details |
|---|---|---|
| 1 | Oligo-ovulation or anovulation | Cycle length >35 days, <8 cycles/year, or amenorrhea |
| 2 | Clinical and/or biochemical hyperandrogenism | Clinical: hirsutism (Ferriman-Gallwey score ≥8), acne, androgenic alopecia. Biochemical: elevated total or free testosterone, DHEA-S |
| 3 | Polycystic ovarian morphology on US | ≥12 follicles (2–9 mm) per ovary or ovarian volume >10 mL |
Must exclude other causes: thyroid disease, congenital adrenal hyperplasia (17-hydroxyprogesterone), hyperprolactinemia, Cushing syndrome, androgen-secreting tumors.
Management
| Goal | Treatment | Details |
|---|---|---|
| Menstrual regulation | Combined OCP | Regulates cycles, reduces androgens (increases SHBG), protects endometrium from hyperplasia |
| Hirsutism/acne | Spironolactone 50–200 mg/day | Anti-androgen; must use contraception (feminizes male fetus). Alternatives: finasteride, eflornithine cream |
| Insulin resistance | Metformin 1500–2000 mg/day | Improves insulin sensitivity, may restore ovulation; weight loss |
| Ovulation induction | Letrozole (first-line) or clomiphene | Letrozole 2.5–7.5 mg/day × 5 days superior to clomiphene for live birth rate |
| Weight management | Lifestyle + pharmacotherapy if needed | 5–10% weight loss significantly improves ovulation, androgen levels, and insulin sensitivity |
30 Menopause & HRT
Menopause is defined as 12 consecutive months of amenorrhea without other cause, reflecting permanent cessation of ovarian function. Mean age: 51 years. The perimenopause transition begins 4–8 years before the final menstrual period. Diagnosis is clinical in women ≥45; FSH (>30–40 IU/L) can confirm in younger women or equivocal cases.
Symptoms
Vasomotor symptoms (hot flashes, night sweats) are the most common reason women seek treatment — affect ~75%, typically peak 1–2 years post-menopause, may persist 7+ years. Other symptoms: vaginal dryness/atrophy, dyspareunia, sleep disturbance, mood changes, urinary symptoms. Long-term consequences: accelerated bone loss (greatest in first 5–7 years), increased cardiovascular risk, and genitourinary syndrome of menopause (GSM).
Hormone Replacement Therapy (HRT)
| Aspect | Details |
|---|---|
| Indications | Moderate-to-severe vasomotor symptoms, GSM, prevention of osteoporosis (when other agents not suitable) |
| Formulations | Estrogen alone (for women without uterus) or estrogen + progesterone (for women with uterus — to prevent endometrial hyperplasia) |
| Timing | "Window of opportunity": initiate within 10 years of menopause or before age 60 for optimal benefit-risk ratio |
| Benefits | Effective relief of vasomotor symptoms, prevents bone loss, improves GSM, possible CV benefit if started early |
| Risks | VTE (oral > transdermal), stroke (small absolute risk), breast cancer (with combined E+P after ~5 years), gallbladder disease |
| Contraindications | History of breast cancer, active VTE/PE, active liver disease, unexplained vaginal bleeding, coronary heart disease, stroke |
31 MEN Syndromes
The multiple endocrine neoplasia (MEN) syndromes are autosomal dominant inherited disorders predisposing to tumors of multiple endocrine glands. Early genetic testing and surveillance are critical for affected families.
| Syndrome | Gene | Components | Key Clinical Features |
|---|---|---|---|
| MEN1 (Wermer syndrome) | MEN1 (menin) — tumor suppressor, chr 11q13 | 3 P's: Parathyroid hyperplasia (95%), Pancreatic NETs (gastrinoma 40%, insulinoma 10%), Pituitary adenomas (prolactinoma most common, 30–40%) | Also: adrenal cortical tumors, carcinoid (thymic/bronchial), facial angiofibromas, collagenomas. Primary HPT is usually the first manifestation |
| MEN2A (Sipple syndrome) | RET proto-oncogene (gain of function) — chr 10q11.2 | Medullary thyroid cancer (MTC, ~100%), Pheochromocytoma (50%), Primary hyperparathyroidism (20–30%) | Prophylactic thyroidectomy based on RET mutation risk category. Screen pheo BEFORE thyroid surgery |
| MEN2B | RET (most commonly M918T mutation) | MTC (earliest and most aggressive form), Pheochromocytoma (50%), Mucosal neuromas, Marfanoid habitus | NO hyperparathyroidism. Mucosal neuromas on tongue, lips, eyelids. Prophylactic thyroidectomy in infancy |
Screening & Surveillance
MEN1: Annual biochemistry (calcium, PTH, prolactin, IGF-1, fasting glucose, chromogranin A); periodic imaging (MRI pituitary, CT/MRI pancreas). MEN2: Genetic testing for RET mutations in all first-degree relatives. Calcitonin screening. Pheo screening (plasma metanephrines) annually starting age 8–11 (varies by mutation). Always rule out pheochromocytoma before any surgery in MEN2 (undiagnosed pheo during surgery can cause fatal hypertensive crisis).
32 Carcinoid Tumors & NETs
Neuroendocrine tumors (NETs) arise from enterochromaffin and other neuroendocrine cells throughout the body. The most common sites are the GI tract (small intestine > rectum > appendix) and lungs. They are graded by Ki-67 proliferation index: G1 (<3%), G2 (3–20%), G3 (>20%). High-grade neuroendocrine carcinomas (NECs) are aggressive and distinct from well-differentiated NETs.
Carcinoid Syndrome
Occurs when serotonin and other vasoactive substances reach the systemic circulation. Typically requires hepatic metastases (liver normally metabolizes serotonin via first-pass). Symptoms: episodic flushing (80%), secretory diarrhea (70%), bronchospasm/wheezing, and right-sided valvular heart disease (carcinoid heart disease — tricuspid regurgitation, pulmonic stenosis; left side protected by lung MAO metabolism).
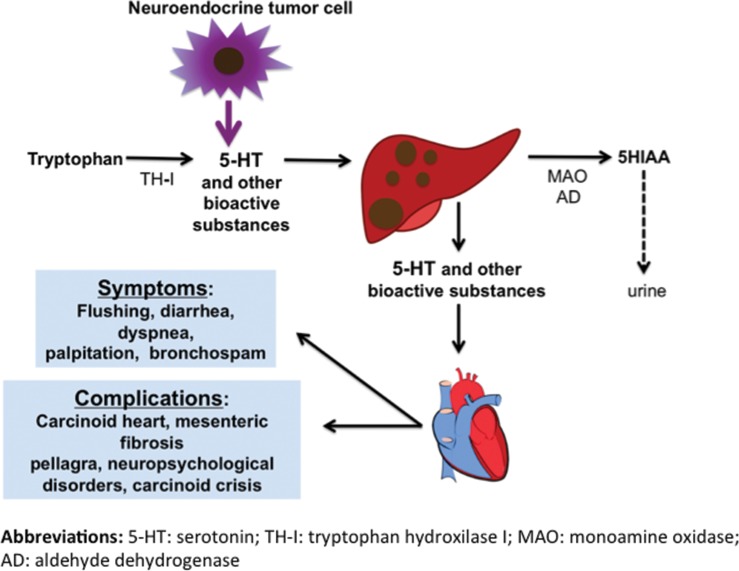
Diagnosis & Management
| Aspect | Details |
|---|---|
| Biochemical marker | 24-hour urine 5-HIAA (5-hydroxyindoleacetic acid, serotonin metabolite). Avoid serotonin-rich foods (bananas, avocados, walnuts) 48 hr before collection |
| Blood markers | Chromogranin A (non-specific but useful for monitoring), serotonin |
| Imaging | Ga-68 DOTATATE PET/CT (somatostatin receptor imaging — most sensitive), CT/MRI, octreotide scan |
| Medical therapy | Somatostatin analogs (octreotide LAR 20–30 mg IM monthly, lanreotide 120 mg SQ monthly) — control symptoms and have antiproliferative effect |
| Carcinoid crisis prevention | Octreotide 250–500 mcg IV before surgery or procedures (massive serotonin release can cause hypotension, bronchospasm) |
| PRRT | Lu-177 DOTATATE (Lutathera) for advanced somatostatin receptor-positive NETs (NETTER-1 trial) |
33 Hypoglycemia in Non-Diabetic Adults
Hypoglycemia in a non-diabetic adult requires the Whipple triad: (1) symptoms consistent with hypoglycemia, (2) documented low plasma glucose (<55 mg/dL), and (3) resolution of symptoms with glucose correction.
Differential Diagnosis
| Category | Conditions | Key Lab Features |
|---|---|---|
| Insulinoma | Pancreatic beta-cell tumor (most common cause of endogenous hyperinsulinemic hypoglycemia) | Elevated insulin, elevated C-peptide, elevated proinsulin, negative sulfonylurea screen |
| Factitious (exogenous insulin) | Surreptitious insulin injection | Elevated insulin, LOW C-peptide (exogenous insulin suppresses endogenous), no proinsulin elevation |
| Factitious (sulfonylurea) | Surreptitious SU use | Elevated insulin, elevated C-peptide, positive sulfonylurea screen |
| Non-islet cell tumor (NICTH) | Large mesenchymal tumors secreting IGF-2 (big IGF-2) | Low insulin, low C-peptide, elevated IGF-2/IGF-1 ratio |
| Adrenal insufficiency | Cortisol deficiency impairs gluconeogenesis | Low cortisol, elevated ACTH (primary) |
| Hepatic failure | Impaired gluconeogenesis and glycogenolysis | Abnormal LFTs, low albumin |
| Post-gastric bypass | Nesidioblastosis / dumping-related hyperinsulinemia | Postprandial pattern (1–3 hours after eating) |
| Autoimmune (insulin antibodies) | Autoimmune hypoglycemia (Hirata disease) | High insulin, anti-insulin antibodies positive |
72-Hour Supervised Fast
Gold standard for diagnosing insulinoma and other causes of fasting hypoglycemia. Patient fasts under observation with serial glucose, insulin, C-peptide, proinsulin, and beta-hydroxybutyrate every 6 hours (then every 1–2 hours as glucose approaches 60). End fast when glucose <45 mg/dL with symptoms, or at 72 hours. Insulinoma: insulin ≥3 μU/mL, C-peptide ≥0.6 ng/mL, proinsulin ≥5 pmol/L, BHB ≤2.7 mmol/L, negative SU screen. Most insulinomas (~90%) are benign, solitary, and <2 cm. Localize with CT, endoscopic US, or intraoperative US.
34 Dynamic Endocrine Testing
Dynamic testing is the cornerstone of endocrine diagnosis. Stimulation tests assess gland reserve (can it respond?); suppression tests assess autonomy (can it be turned off?).
| Test | Indication | Protocol | Interpretation |
|---|---|---|---|
| Cosyntropin (ACTH) stimulation | Adrenal insufficiency | Cosyntropin 250 mcg IV/IM; measure cortisol at 0, 30, 60 min | Peak cortisol ≥18 mcg/dL = normal adrenal function. <18 = AI. May miss recent-onset secondary AI |
| Low-dose (1 mcg) cosyntropin | Suspected secondary/partial AI | Cosyntropin 1 mcg IV; cortisol at 0 and 30 min | More sensitive for partial/secondary AI. Peak ≥18 = normal |
| 1 mg overnight DST | Screening for Cushing syndrome | Dexamethasone 1 mg PO at 11 PM; 8 AM cortisol | Cortisol >1.8 mcg/dL = positive screen (failure to suppress) |
| High-dose (8 mg) DST | Distinguish Cushing disease from ectopic ACTH | Dexamethasone 8 mg PO at 11 PM; 8 AM cortisol | Cushing disease: cortisol suppresses >50%. Ectopic ACTH: no suppression |
| CRH stimulation test | Distinguish Cushing disease from ectopic ACTH | CRH 1 mcg/kg IV; measure ACTH and cortisol at intervals | Cushing disease: ACTH and cortisol rise. Ectopic: no response |
| Oral glucose tolerance test (OGTT) for GH | Confirm acromegaly | 75 g oral glucose; GH measured at 0, 30, 60, 90, 120 min | Normal: GH suppresses to <1 ng/mL. Acromegaly: failure to suppress |
| Water deprivation test | Differentiate central DI, nephrogenic DI, primary polydipsia | Fluid restrict, monitor Uosm/Sosm; give DDAVP when criteria met | Central DI: Uosm rises >50% after DDAVP. Nephrogenic: no response |
| Insulin tolerance test (ITT) | GH and ACTH reserve (gold standard) | Regular insulin 0.1 units/kg IV to induce hypoglycemia (<40 mg/dL) | Normal: cortisol >18 and GH >5 ng/mL. Contraindicated in seizure disorders, CAD, elderly |
| GnRH stimulation test | Precocious puberty, gonadotropin deficiency | GnRH (or leuprolide) injection; measure LH, FSH | Pubertal response: LH rises >5 IU/L. Prepubertal: minimal rise |
35 Thyroid FNA & Endocrine Imaging
Thyroid Ultrasound
First-line imaging for thyroid nodules. Key features to report: nodule size, composition (solid, cystic, mixed), echogenicity (hypoechoic vs isoechoic vs hyperechoic), margins, calcifications (microcalcifications are suspicious), shape (taller-than-wide), and vascularity. ACR TI-RADS provides a standardized scoring system to guide FNA decisions.
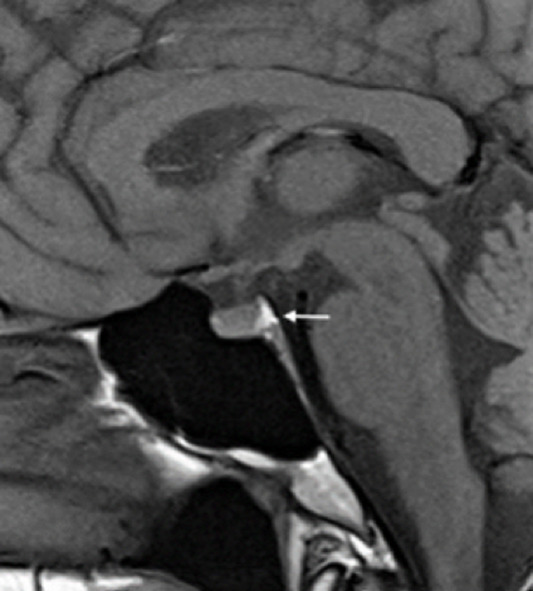
FNA Technique
Performed under ultrasound guidance with a 25–27 gauge needle. Typically 2–3 passes per nodule. On-site cytology evaluation ("rapid on-site evaluation" or ROSE) improves adequacy rates. Bethesda classification (see Section 13) guides further management. Molecular testing (Afirma, ThyroSeq) is increasingly used for Bethesda III/IV nodules to avoid diagnostic surgery.
Radioactive Iodine Uptake (RAIU) Scan
Uses I-123 or Tc-99m pertechnetate. Measures percentage of administered radioiodine taken up by the thyroid at 4 and 24 hours. Elevated uptake: Graves disease (diffuse), toxic MNG (patchy), toxic adenoma (focal hot nodule with suppressed background). Low uptake: thyroiditis (subacute, painless), factitious thyrotoxicosis, iodine-induced, struma ovarii.
Other Endocrine Imaging
| Modality | Indication | Key Points |
|---|---|---|
| Pituitary MRI (with gadolinium) | Pituitary adenoma, hypopituitarism, DI | Microadenomas: hypointense on post-contrast T1. Macroadenomas: assess suprasellar extension, cavernous sinus invasion, optic chiasm compression |
| Adrenal CT (non-contrast) | Adrenal incidentaloma, Cushing, pheo, PA | Lipid-rich adenoma: <10 HU on non-contrast CT (benign). >10 HU: consider washout CT or MRI (chemical shift) |
| MIBG scan (I-123/131) | Pheochromocytoma, paraganglioma, neuroblastoma | Chromaffin tissue-specific. Used for localization, staging, and as theranostic (I-131 MIBG therapy) |
| Ga-68 DOTATATE PET/CT | NETs, pheo/paraganglioma (especially SDHx) | Somatostatin receptor imaging; superior sensitivity to octreotide scan |
| Sestamibi scan (Tc-99m) | Parathyroid adenoma localization | Single adenoma uptake with delayed washout. Often combined with SPECT/CT. 4D CT also increasingly used |
36 Imaging in Endocrinology
| Clinical Scenario | First-Line Imaging | Second-Line / Advanced | Key Findings |
|---|---|---|---|
| Thyroid nodule | Thyroid ultrasound | FNA (US-guided), RAIU scan (if TSH low) | Microcalcifications, hypoechoic, irregular margins = suspicious |
| Hyperthyroidism etiology | RAIU scan (I-123) | TRAb/TSI, thyroid US | Diffuse high uptake = Graves; low uptake = thyroiditis |
| Pituitary lesion | MRI with gadolinium | CT (if MRI contraindicated) | Microadenomas enhance less than normal pituitary on dynamic MRI |
| Adrenal incidentaloma | Non-contrast adrenal CT | Adrenal washout CT, MRI (chemical shift), PET/CT | <10 HU = lipid-rich adenoma (benign). >10 HU needs further workup |
| Pheochromocytoma | CT abdomen/pelvis | MRI (T2 bright), MIBG scan, Ga-68 DOTATATE PET | Heterogeneous enhancing adrenal mass; "light bulb" on T2 MRI |
| Primary hyperparathyroidism | Sestamibi scan with SPECT/CT | 4D CT, thyroid US | Delayed sestamibi washout in adenoma |
| Osteoporosis | DEXA (central: hip + spine) | VFA (vertebral fracture assessment), TBS (trabecular bone score) | T-score ≤−2.5 = osteoporosis |
| Neuroendocrine tumors | Ga-68 DOTATATE PET/CT | CT/MRI, octreotide scan (if PET unavailable) | Somatostatin receptor-positive lesions |
| Insulinoma localization | CT pancreas (triphasic) | Endoscopic US, arterial calcium stimulation with hepatic venous sampling | Small hypervascular pancreatic lesion |
37 Classification Systems
WHO Diabetes Classification (2019)
| Type | Key Features |
|---|---|
| Type 1 | Autoimmune beta-cell destruction, absolute insulin deficiency |
| Type 2 | Insulin resistance + progressive beta-cell failure |
| Gestational (GDM) | First recognized during pregnancy (not preexisting) |
| Specific types | MODY (monogenic), pancreatic disease, drug-induced (steroids), endocrinopathies (Cushing, acromegaly, pheo) |
Bethesda Thyroid Cytology (See Section 13)
Categories I–VI with associated malignancy risk and recommended management. Molecular testing (Afirma GSC, ThyroSeq v3) now used for indeterminate nodules (Bethesda III/IV) to reclassify and potentially avoid surgery.
KDIGO CKD-MBD Guidelines
| CKD Stage | eGFR | Calcium/Phosphate Issues | PTH Target |
|---|---|---|---|
| 3a–3b | 30–59 | Begin monitoring Ca, PO4, PTH, 25-OH-D | Within normal range |
| 4 | 15–29 | Hyperphosphatemia, secondary HPT developing | Trend toward normal |
| 5 / 5D (dialysis) | <15 | Renal osteodystrophy, vascular calcification | 2–9× upper limit of normal |
Management: phosphate binders (calcium-based, sevelamer, lanthanum), calcitriol or active vitamin D analogs, calcimimetics (cinacalcet, etelcalcetide) for dialysis patients. Goal: avoid hypercalcemia, minimize Ca × PO4 product.
ATA Thyroid Nodule Risk Stratification (2015)
| Pattern | US Features | Malignancy Risk | FNA Size Threshold |
|---|---|---|---|
| High suspicion | Solid hypoechoic + microcalcifications or irregular margins or taller-than-wide or ETE | >70–90% | ≥1 cm |
| Intermediate suspicion | Solid hypoechoic without above features | 10–20% | ≥1 cm |
| Low suspicion | Isoechoic or hyperechoic solid, or partially cystic with eccentric solid component | 5–10% | ≥1.5 cm |
| Very low suspicion | Spongiform or partially cystic without suspicious features | <3% | ≥2 cm (observation also acceptable) |
| Benign | Purely cystic | <1% | No FNA needed |
38 Medications Master Table
Insulins
| Name | Type | Onset | Peak | Duration | Notes |
|---|---|---|---|---|---|
| Lispro (Humalog) | Rapid | 15 min | 1–2 hr | 3–5 hr | Bolus/pump use |
| Aspart (NovoLog) | Rapid | 15 min | 1–2 hr | 3–5 hr | Bolus/pump use |
| Regular (Humulin R) | Short | 30 min | 2–4 hr | 6–8 hr | IV insulin drip; meals |
| NPH (Humulin N) | Intermediate | 1–2 hr | 4–8 hr | 12–16 hr | Basal (BID); cloudy |
| Glargine U-100 (Lantus) | Long | 1–2 hr | Minimal | ~24 hr | Once daily basal; cannot mix |
| Glargine U-300 (Toujeo) | Long | 6 hr | None | >24 hr | Flatter profile; less hypoglycemia |
| Detemir (Levemir) | Long | 1–2 hr | Minimal | 16–24 hr | May need BID dosing |
| Degludec (Tresiba) | Ultra-long | 1 hr | None | >42 hr | Flexible dosing window |
Oral & Non-Insulin Diabetes Agents
| Drug | Class | Typical Dose | Key Side Effects |
|---|---|---|---|
| Metformin | Biguanide | 500–2000 mg/day (divided BID) | GI (diarrhea, nausea), B12 deficiency, lactic acidosis (rare) |
| Empagliflozin | SGLT2i | 10–25 mg daily | Genital infections, euDKA, volume depletion |
| Dapagliflozin | SGLT2i | 5–10 mg daily | Same as above; also approved for HF and CKD |
| Semaglutide (SQ) | GLP-1 RA | 0.25–2 mg weekly | Nausea, vomiting, pancreatitis (rare), MTC (contraindicated in MEN2) |
| Tirzepatide | GIP/GLP-1 dual agonist | 2.5–15 mg weekly | GI, significant weight loss, pancreatitis (rare) |
| Sitagliptin | DPP-4i | 100 mg daily | Nasopharyngitis, arthralgia; renal dose adjust |
| Glipizide | SU | 5–20 mg daily | Hypoglycemia, weight gain |
| Pioglitazone | TZD | 15–45 mg daily | Weight gain, edema, fractures, HF exacerbation, bladder cancer (debated) |
Thyroid Medications
| Drug | Indication | Dose Range | Key Notes |
|---|---|---|---|
| Levothyroxine (T4) | Hypothyroidism | 1.6 mcg/kg/day (full); 25–50 mcg start (elderly) | Empty stomach; separate from Ca, Fe, PPIs by 4 hr |
| Liothyronine (T3) | Myxedema coma, combination therapy | 5–25 mcg BID–TID | Short half-life; not routinely used as monotherapy |
| Methimazole (MMI) | Hyperthyroidism | 5–30 mg daily | First-line ATD; teratogenic in 1st trimester |
| Propylthiouracil (PTU) | Hyperthyroidism (1st trimester, thyroid storm) | 100–200 mg TID | Blocks T4→T3 conversion; hepatotoxicity risk |
Steroid Replacements
| Drug | Glucocorticoid Potency | Mineralocorticoid Potency | Equivalent Dose | Half-Life |
|---|---|---|---|---|
| Hydrocortisone | 1 | 1 | 20 mg | 8–12 hr |
| Prednisone | 4 | 0.8 | 5 mg | 12–36 hr |
| Methylprednisolone | 5 | 0.5 | 4 mg | 12–36 hr |
| Dexamethasone | 25 | 0 | 0.75 mg | 36–72 hr |
| Fludrocortisone | 10 | 125 | N/A | 18–36 hr |
Bone Agents
| Drug | Class | Route/Frequency | Key Consideration |
|---|---|---|---|
| Alendronate | Bisphosphonate | 70 mg PO weekly | Drug holiday after 5 yr; esophagitis; ONJ rare |
| Zoledronic acid | Bisphosphonate | 5 mg IV yearly | Drug holiday after 3 yr IV; contraindicated CrCl <35 |
| Denosumab | RANKL inhibitor | 60 mg SQ q6mo | Rebound fractures if stopped without transition |
| Teriparatide | PTH analog (anabolic) | 20 mcg SQ daily × 2 yr | Osteosarcoma risk (black box, rat data); contraindicated in Paget, bone mets |
| Romosozumab | Sclerostin inhibitor | 210 mg SQ monthly × 12 mo | Black box: CV events |
39 Abbreviations Master List
40 Risk Scores & Guidelines
Key Guidelines
| Organization | Guideline | Year | Key Recommendations |
|---|---|---|---|
| ADA | Standards of Care in Diabetes | 2024 | A1C <7% for most adults; SGLT2i/GLP-1 RA for ASCVD/HF/CKD regardless of A1C; CGM for all T1DM and insulin-using T2DM; comprehensive foot exam annually |
| ATA | Thyroid Nodules & Differentiated Thyroid Cancer | 2015 | US-based risk stratification for FNA; lobectomy acceptable for low-risk <4 cm unifocal papillary; TSH suppression by risk category |
| Endocrine Society | Cushing Syndrome | 2015 | Screen with 2+ tests (UFC, late-night salivary cortisol, 1 mg DST); BIPSS for equivocal ACTH-dependent cases |
| Endocrine Society | Primary Aldosteronism | 2016 | ARR screening for at-risk hypertensives; confirmatory testing required; AVS for subtype differentiation in surgical candidates |
| Endocrine Society | Pheochromocytoma / Paraganglioma | 2014 | Plasma free metanephrines for screening; alpha-blockade preoperatively; genetic testing for all |
| AACE / ACE | Adrenal Incidentaloma | 2009 | Biochemical workup: 1 mg DST, plasma metanephrines, ARR if hypertensive. Imaging follow-up at 3–6 mo, then 12–24 mo |
| NOF / AACE | Osteoporosis | 2020 | DEXA for women ≥65, men ≥70, younger with risk factors; treat if T-score ≤−2.5 or FRAX ≥20%/3% |
| Endocrine Society | Male Hypogonadism | 2018 | Confirm low AM testosterone on 2 occasions; individualize TRT; monitor hematocrit, PSA |
Adrenal Incidentaloma Workup
An adrenal mass found incidentally on imaging requires two assessments: (1) Is it functional? — 1 mg DST (Cushing), plasma metanephrines (pheo), and ARR if hypertensive (PA). (2) Is it malignant? — Size (>4 cm increases risk), non-contrast CT density (>10 HU concerning), interval growth, and irregular features. Surgery recommended for functional tumors or imaging characteristics concerning for malignancy.
Glycemic Targets Summary
| Metric | General Target | Notes |
|---|---|---|
| A1C | <7.0% | Individualize: <6.5% (young, new diagnosis, no hypoglycemia) to <8.0% (elderly, comorbid) |
| Fasting/pre-meal glucose | 80–130 mg/dL | ADA standard |
| Post-meal glucose (1–2 hr) | <180 mg/dL | ADA standard |
| Time in range (CGM) | ≥70% (70–180 mg/dL) | TIR correlates with A1C and microvascular outcomes |
| Time below range (<70) | <4% | Critical safety metric |
| Time below range (<54) | <1% | Clinically significant hypoglycemia |
| GMI | Parallels A1C target | Derived from CGM mean glucose |
References & Figure Sources
Figures
- Figure 1 — The Hypothalamic-Pituitary-Adrenal Axis. Adapted from Panagiotakopoulos L, Bhatt S. Front Behav Neurosci. 2021;14:601939. Licensed under CC BY 4.0.
- Figure 2 — Steroid Hormone Receptor Inputs to the HPA Axis. Adapted from Panagiotakopoulos L, Bhatt S. Front Behav Neurosci. 2021;14:601939. Licensed under CC BY 4.0.
- Figure 3 — Thyroid and Parathyroid Gland Anatomy. StatPearls [Internet]. Licensed under CC BY 4.0.
- Figure 4 — Insulin Pump and Continuous Glucose Monitor. Adapted from Ang L et al. Curr Diab Rep. 2022;22:587-597. Licensed under CC BY 4.0.
- Figure 5 — Hybrid Closed-Loop Insulin Delivery System. Adapted from Ang L et al. Curr Diab Rep. 2022;22:587-597. Licensed under CC BY 4.0.
- Figure 6 — Pathophysiology of Diabetic Ketoacidosis. Calgary Guide, University of Calgary. Used for educational purposes.
- Figure 7 — DKA Management: Fluid Replacement Protocol. Adapted from Kitabchi AE et al. Endotext [Internet]. Licensed under CC BY-NC-ND.
- Figure 8 — DKA and HHS: Insulin Infusion Protocol. Adapted from Kitabchi AE et al. Endotext [Internet]. Licensed under CC BY-NC-ND.
- Figure 9 — Immunopathogenesis of Graves Ophthalmopathy. Adapted from Bahn RS. N Engl J Med. 2010;362(8):726-738.
- Figure 10 — Treatment Algorithm for Graves Ophthalmopathy. Adapted from Bahn RS. N Engl J Med. 2010;362(8):726-738.
- Figure 11 — Thyroid Storm Management Algorithm. Adapted from Idrose AM. J Intensive Care Med. 2015;30(3):131-140.
- Figure 12 — Adrenal Gland Vascular Anatomy. Endotext [Internet]: Adrenal Cortex. Licensed under CC BY-NC-ND.
- Figure 13 — Adrenal Cortex Zonal Organization. Endotext [Internet]: Adrenal Cortex. Licensed under CC BY-NC-ND.
- Figure 14 — Adrenal Steroidogenesis Pathway. Endotext [Internet]: Adrenal Cortex. Licensed under CC BY-NC-ND.
- Figure 15 — Cushing Syndrome Diagnostic Algorithm. Endotext [Internet]: Cushing’s Disease. Licensed under CC BY-NC-ND.
- Figure 16 — Potassium Management in Diabetic Emergencies. Adapted from Kitabchi AE et al. Endotext [Internet]. Licensed under CC BY-NC-ND.
- Figure 17 — The Renin-Angiotensin-Aldosterone System. StatPearls [Internet]: Physiology, Renin Angiotensin System. Licensed under CC BY 4.0.
- Figure 18 — Pheochromocytoma on MRI. Adapted from Blake MA et al. Gland Surg. 2015;4(4):350-358. Licensed under CC BY 4.0.
- Figure 19 — Pheochromocytoma: MRI and MIBG Scan. Adapted from Blake MA et al. Gland Surg. 2015;4(4):350-358. Licensed under CC BY 4.0.
- Figure 20 — Hypothalamic and Pituitary Hormones. StatPearls [Internet]: Pituitary Gland Anatomy. Licensed under CC BY 4.0.
- Figure 21 — Microscopic Anatomy of the Pituitary Gland. StatPearls [Internet]: Pituitary Gland Anatomy. Licensed under CC BY 4.0.
- Figure 22 — Pituitary Macroadenoma on CT. StatPearls [Internet]: Pituitary Gland Anatomy. Licensed under CC BY 4.0.
- Figure 23 — Diagnostic Algorithm for Diabetes Insipidus. Endotext [Internet]: Diagnostic Testing for DI. Licensed under CC BY-NC-ND.
- Figure 24 — Water Deprivation Test Results. Endotext [Internet]: Diagnostic Testing for DI. Licensed under CC BY-NC-ND.
- Figure 25 — Calcium and Skeletal Metabolism. Endotext [Internet]: Calcium and Phosphate Homeostasis. Licensed under CC BY-NC-ND.
- Figure 26 — Metabolic Activation of Vitamin D. Endotext [Internet]: Calcium and Phosphate Homeostasis. Licensed under CC BY-NC-ND.
- Figure 27 — The Bone Remodeling Cycle. Endotext [Internet]: Epidemiology and Pathogenesis of Osteoporosis. Licensed under CC BY-NC-ND.
- Figure 28 — RANKL/OPG Signaling in Bone Metabolism. Endotext [Internet]: Calcium and Phosphate Homeostasis. Licensed under CC BY-NC-ND.
- Figure 29 — PCOS Pathophysiology. Adapted from Lim SS et al. BMJ Med. 2023;2(1):e000548. Licensed under CC BY 4.0.
- Figure 30 — Pathophysiology of Carcinoid Syndrome. Adapted from Pinto MP et al. Clinics. 2018;73(suppl 1):e490s. Licensed under CC BY.
- Figure 31 — Pituitary Gland MRI. Endotext [Internet]: Diagnostic Testing for DI. Licensed under CC BY-NC-ND.
Key Trials & Guidelines
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes — 2024. Diabetes Care. 2024;47(Suppl 1):S1-S321. PMID: 38078582
- Davies MJ, Aroda VR, Collins BS, et al. Management of hyperglycaemia in type 2 diabetes, 2022: ADA/EASD consensus report. Diabetologia. 2022;65(12):1925-1966. PMID: 36151309
- Haugen BR, Alexander EK, Bible KC, et al. 2015 ATA management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1-133. PMID: 26462967
- Nieman LK, Biller BM, Findling JW, et al. Treatment of Cushing's syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(8):2807-2831. PMID: 26222757
- Funder JW, Carey RM, Mantero F, et al. The management of primary aldosteronism: case detection, diagnosis, and treatment. J Clin Endocrinol Metab. 2016;101(5):1889-1916. PMID: 26934393
- Lenders JW, Duh QY, Eisenhofer G, et al. Pheochromocytoma and paraganglioma: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2014;99(6):1915-1942. PMID: 24893135
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes (EMPA-REG OUTCOME). N Engl J Med. 2015;373(22):2117-2128. PMID: 26378978
- Marso SP, Daniels GH, Tanaka K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes (LEADER). N Engl J Med. 2016;375(4):311-322. PMID: 27295427
- Ross DS, Burch HB, Cooper DS, et al. 2016 American Thyroid Association guidelines for diagnosis and management of hyperthyroidism and other causes of thyrotoxicosis. Thyroid. 2016;26(10):1343-1421. PMID: 27521067
- Bornstein SR, Allolio B, Arlt W, et al. Diagnosis and treatment of primary adrenal insufficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2016;101(2):364-389. PMID: 26760044
- Shoback D, Rosen CJ, Black DM, et al. Pharmacological management of osteoporosis in postmenopausal women: an Endocrine Society guideline update. J Clin Endocrinol Metab. 2020;105(3):dgaa048. PMID: 32068863
- Battelino T, Danne T, Bergenstal RM, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care. 2019;42(8):1593-1603. PMID: 31177185
Textbooks & Reference Works
- Williams Textbook of Endocrinology. 14th ed. Melmed S, Auchus RJ, Goldfine AB, et al., eds. Elsevier; 2020.
- Greenspan's Basic & Clinical Endocrinology. 10th ed. Gardner DG, Shoback DM, eds. McGraw-Hill; 2018.
- Endocrine Society Clinical Practice Guidelines. Available at: endocrine.org/clinical-practice-guidelines.
- ADA Standards of Care in Diabetes. Available at: diabetesjournals.org/care.