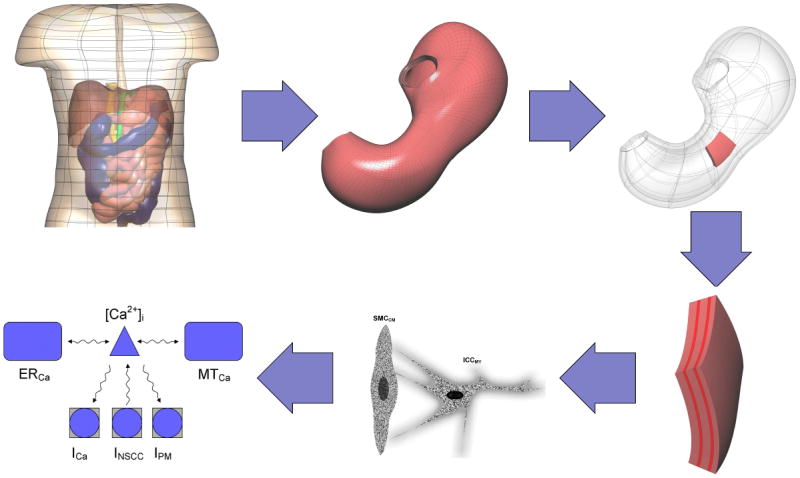
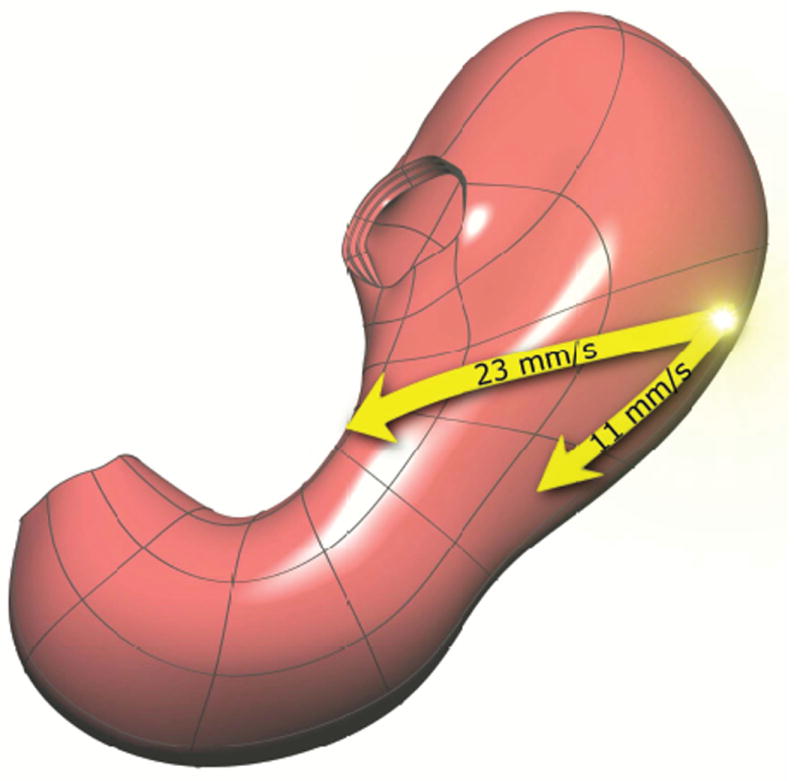
Gastroenterology
Every diagnosis, procedure, classification system, medication, endoscopic technique, hepatology detail, and management strategy in one place.
01 GI Anatomy — Esophagus Through Rectum
Esophagus
The esophagus is a 25 cm muscular tube extending from the pharynx (C6) to the gastroesophageal junction (GEJ, ~T11). Divided into thirds: upper third (skeletal muscle, innervated by recurrent laryngeal nerve), middle third (mixed), lower third (smooth muscle, controlled by vagus nerve and myenteric plexus). Three physiologic narrowings: cricopharyngeus (upper esophageal sphincter, UES) at 15 cm from incisors, aortic arch/left mainstem bronchus crossing at 23 cm, and diaphragmatic hiatus/lower esophageal sphincter (LES) at 40 cm. These narrowing points are where foreign bodies lodge and where strictures/tumors preferentially develop. The LES is not a true anatomic sphincter but a zone of tonic smooth muscle contraction 2-4 cm in length maintained at 10-30 mmHg above intragastric pressure.
Stomach
The stomach is divided into four anatomic regions: cardia (immediately distal to the GEJ, mucus-secreting glands), fundus (dome above the GEJ, contains parietal cells and chief cells), body/corpus (largest portion, majority of parietal cells producing HCl and intrinsic factor, plus chief cells producing pepsinogen), and antrum/pylorus (G cells producing gastrin, D cells producing somatostatin). The incisura angularis marks the border between body and antrum on the lesser curvature. Gastric pH is normally 1.5-3.5. The pylorus controls gastric emptying into the duodenum; pyloric stenosis (congenital or acquired from ulcer scarring) causes gastric outlet obstruction.
Duodenum
The duodenum is 25-30 cm, C-shaped, and divided into four parts: D1 (bulb/cap) — most common site for duodenal ulcers; D2 (descending) — contains the ampulla of Vater where the common bile duct (CBD) and main pancreatic duct (of Wirsung) empty, the target of ERCP; D3 (horizontal/transverse) — crosses the aorta and IVC, can be compressed by the SMA (SMA syndrome); D4 (ascending) — transitions to jejunum at the ligament of Treitz. The ligament of Treitz is the landmark dividing upper from lower GI bleeding.
Jejunum & Ileum
The jejunum (proximal 40% of small bowel) and ileum (distal 60%) together measure 4-6 meters. The jejunum has thicker walls, larger villi, more prominent plicae circulares, and a larger lumen. Iron and folate are absorbed in the proximal jejunum. The terminal ileum is critically important: it is the exclusive site of vitamin B12 absorption and bile salt reabsorption (enterohepatic circulation). Resection or disease of the terminal ileum (Crohn's disease) causes B12 deficiency and bile salt malabsorption leading to secretory diarrhea. The ileocecal valve marks the transition to the colon.
Colon & Rectum
The colon (~150 cm) is divided into: cecum (with appendix), ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon, and rectum (12-15 cm). The ascending and descending colon are retroperitoneal; the transverse and sigmoid colon are intraperitoneal on mesenteries. The splenic flexure is a watershed zone between SMA and IMA territories — most vulnerable to ischemic colitis. The colon absorbs water and electrolytes, converting ~1.5 L of ileal effluent to ~200 mL of stool daily. The rectum has three valves of Houston and transitions to the anal canal at the dentate (pectinate) line.
GI Blood Supply
The GI tract is supplied by three major branches of the abdominal aorta: the celiac trunk (T12 — supplies the foregut: distal esophagus, stomach, proximal duodenum, liver, gallbladder, spleen, pancreas via left gastric, common hepatic, and splenic arteries), the superior mesenteric artery (SMA) (L1 — supplies the midgut: distal duodenum, jejunum, ileum, cecum, ascending and transverse colon via inferior pancreaticoduodenal, jejunal, ileal, ileocolic, right colic, and middle colic arteries), and the inferior mesenteric artery (IMA) (L3 — supplies the hindgut: descending colon, sigmoid colon, and upper rectum via left colic, sigmoid, and superior rectal arteries). The marginal artery of Drummond and the arc of Riolan (meandering mesenteric artery) connect the SMA and IMA territories — critical collateral pathways in mesenteric ischemia.
Innervation
The GI tract has both extrinsic and intrinsic innervation. Extrinsic: parasympathetic (vagus nerve — CN X — to the proximal two-thirds of the colon; pelvic splanchnic nerves S2-S4 to the distal third) stimulates motility and secretion; sympathetic (greater/lesser/least splanchnic nerves from T5-L2) inhibits motility and causes vasoconstriction. Intrinsic: the enteric nervous system (ENS) — "the second brain" — contains ~100 million neurons organized into two plexuses: myenteric (Auerbach's) plexus (between circular and longitudinal muscle layers, controls motility) and submucosal (Meissner's) plexus (controls secretion and blood flow). Hirschsprung's disease results from absence of ganglion cells in the myenteric and submucosal plexuses. Achalasia results from degeneration of inhibitory neurons in the myenteric plexus of the LES.
02 Hepatobiliary & Pancreatic Anatomy
Liver
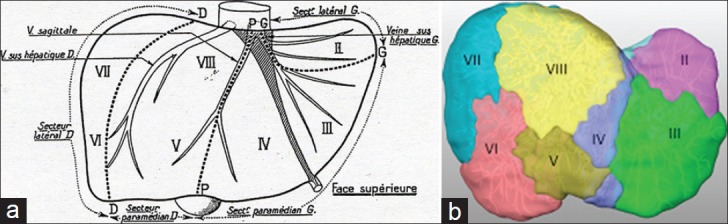
The liver (~1.5 kg) occupies the right upper quadrant. Functionally divided into right and left lobes by Cantlie's line (from the gallbladder fossa to the IVC), not the falciform ligament. The Couinaud classification divides the liver into 8 functionally independent segments, each with its own portal pedicle (portal vein branch, hepatic artery branch, bile duct) and hepatic venous drainage — this is the basis for anatomic hepatic resections. The portal triad (portal vein, hepatic artery, bile duct) enters at the hilum (porta hepatis). The liver receives a dual blood supply: ~75% from the portal vein (nutrient-rich, deoxygenated blood from the GI tract) and ~25% from the hepatic artery (oxygenated blood from the celiac trunk). Blood exits via 3 hepatic veins (right, middle, left) draining into the IVC.
Biliary System
Bile is produced by hepatocytes (~500-600 mL/day), flows through intrahepatic bile ducts into the right and left hepatic ducts, which merge at the hilum to form the common hepatic duct (CHD). The cystic duct from the gallbladder joins the CHD to form the common bile duct (CBD), which is normally ≤ 6 mm in diameter (up to 8-10 mm post-cholecystectomy or with age). The CBD passes through the head of the pancreas and enters the duodenum at the ampulla of Vater, controlled by the sphincter of Oddi. The gallbladder (fundus, body, infundibulum/Hartmann's pouch, neck) stores and concentrates bile, releasing it in response to cholecystokinin (CCK) stimulated by fat and amino acids in the duodenum.
Pancreas
The pancreas (12-15 cm) is a retroperitoneal organ with four parts: head (nestled in the C-curve of the duodenum, includes the uncinate process), neck (overlies the SMV/portal vein confluence — key in surgical resectability assessment), body, and tail (extends to the splenic hilum). The main pancreatic duct (duct of Wirsung) runs the length of the pancreas and joins the CBD at the ampulla. The accessory duct (of Santorini) drains through the minor papilla — relevant in pancreas divisum (present in ~7% of the population, the most common congenital pancreatic anomaly). Exocrine function: acinar cells produce digestive enzymes (lipase, amylase, trypsinogen, chymotrypsinogen). Endocrine function: islets of Langerhans (beta cells produce insulin, alpha cells produce glucagon, delta cells produce somatostatin).
03 The GI Physical Exam
Abdominal Exam — Systematic Approach
Inspection: Distension (ascites, obstruction, ileus), visible peristalsis (in thin patients with obstruction), scars (prior surgeries), caput medusae (periumbilical venous distension — portal hypertension), Grey Turner sign (flank ecchymosis — retroperitoneal hemorrhage/severe pancreatitis), Cullen sign (periumbilical ecchymosis — hemoperitoneum/pancreatitis), jaundice (scleral icterus first visible at bilirubin ~2.5 mg/dL), spider angiomata (estrogen excess in cirrhosis — distribution above the nipple line).
Auscultation: Bowel sounds in all four quadrants — hyperactive/high-pitched (early obstruction, diarrheal illness), hypoactive/absent (ileus, late obstruction, peritonitis). Auscultate before palpation. Bruits over the epigastrium may indicate celiac or SMA stenosis.
Percussion: Tympany (gas-filled loops — normal, or exaggerated in obstruction), dullness (fluid, mass, organomegaly). Shifting dullness and fluid wave for ascites. Liver span by percussion: normal 6-12 cm in the right midclavicular line.
Palpation: Start away from pain. Assess for guarding (voluntary = patient tenses; involuntary = peritoneal irritation), rigidity (board-like — diffuse peritonitis), rebound tenderness (pain worse on release — peritoneal inflammation). Murphy's sign (inspiratory arrest during palpation of the RUQ — acute cholecystitis, 97% specific when ultrasound-confirmed). McBurney's point (1/3 distance from ASIS to umbilicus — appendicitis). Rovsing's sign (LLQ palpation causing RLQ pain — appendicitis). Hepatomegaly: liver edge palpable > 2 cm below the right costal margin. Splenomegaly: spleen tip palpable below the left costal margin (must be 2-3x enlarged to be palpable).
Signs of Chronic Liver Disease
| Finding | Mechanism | Clinical Significance |
|---|---|---|
| Jaundice / scleral icterus | Elevated conjugated bilirubin | Visible at total bilirubin > 2.5 mg/dL |
| Spider angiomata | Estrogen excess (impaired hepatic clearance) | > 5 spider nevi is abnormal; distribution above nipple line |
| Palmar erythema | Estrogen-mediated vasodilation | Bilateral, thenar and hypothenar eminences |
| Gynecomastia | Estrogen excess, androgen deficiency | Common in alcoholic cirrhosis |
| Caput medusae | Portal hypertension; paraumbilical vein recanalization | Periumbilical venous distension radiating outward |
| Ascites | Portal HTN + hypoalbuminemia + renal Na retention | Shifting dullness detects ≥ 1500 mL |
| Asterixis | Hepatic encephalopathy — impaired ammonia clearance | "Liver flap" — negative myoclonus at the wrist |
| Terry's nails | Hypoalbuminemia, altered nail bed vasculature | White proximal nail beds with normal distal pink band |
| Dupuytren's contracture | Palmar fascia fibrosis; associated with alcoholic liver disease | Flexion contracture of 4th/5th digits |
Digital Rectal Exam (DRE)
Essential in GI bleeding evaluation: assess for melena (black, tarry — upper GI source), hematochezia (bright red — lower GI or brisk upper GI), masses, hemorrhoids, fissures, fistulae, sphincter tone (low in neurologic disease, high in acute fissure), and prostatic abnormalities. Stool guaiac testing (fecal occult blood) at the bedside is rapidly being replaced by fecal immunochemical testing (FIT) for screening purposes.
04 Key Terminology
Dysphagia: Difficulty swallowing. Oropharyngeal = difficulty initiating swallow (neurologic/muscular). Esophageal = food sticks after initiation; solids only = mechanical obstruction (stricture, ring, tumor); solids + liquids = motility disorder (achalasia, scleroderma).
Odynophagia: Pain with swallowing. Think esophageal ulceration — pill esophagitis, infectious esophagitis (Candida, HSV, CMV), caustic ingestion.
Hematemesis: Vomiting blood (red or coffee-ground). Upper GI source proximal to the ligament of Treitz.
Melena: Black, tarry, malodorous stool. Requires ~150 mL of blood. Usually upper GI source but can originate from small bowel or right colon.
Hematochezia: Bright red blood per rectum. Usually lower GI source but can be brisk upper GI bleed (> 1 L).
Steatorrhea: Fatty, greasy, foul-smelling stool that floats. Indicates fat malabsorption (pancreatic insufficiency, celiac disease, bile salt deficiency).
Tenesmus: Sensation of incomplete evacuation with straining. Think rectal pathology — proctitis (IBD, radiation), rectal mass, IBS.
Borborygmi: Audible rumbling stomach/bowel sounds from peristalsis of gas and fluid. Increased in diarrheal states, early obstruction.
05 GERD Esophageal
Gastroesophageal reflux disease is the most common outpatient GI diagnosis — affecting ~20% of the US adult population weekly. GERD occurs when reflux of gastric contents causes troublesome symptoms (heartburn, regurgitation) or mucosal complications (erosive esophagitis, stricture, Barrett's esophagus). Pathophysiology centers on failure of the antireflux barrier: transient lower esophageal sphincter relaxations (TLESRs) are the primary mechanism in most patients, compounded by low baseline LES pressure, hiatal hernia (which dissociates the LES from the crural diaphragm), impaired esophageal clearance, and delayed gastric emptying.
Diagnosis
GERD is a clinical diagnosis when typical symptoms (heartburn, regurgitation) respond to empiric PPI therapy. Endoscopy (EGD) is indicated for alarm features (dysphagia, odynophagia, weight loss, GI bleeding, anemia, age > 60 with new symptoms), PPI non-responders, and Barrett's screening. Erosive esophagitis is graded by the Los Angeles classification: Grade A (mucosal breaks < 5 mm, not extending between mucosal folds), Grade B (> 5 mm, not extending between folds), Grade C (extends between folds but < 75% of circumference), Grade D (≥ 75% of circumference). Ambulatory pH monitoring (96-hour wireless Bravo capsule or 24-hour impedance-pH catheter) is the gold standard for quantifying acid exposure — DeMeester score > 14.7 or acid exposure time > 6% is abnormal. Lyon Consensus 2.0 (2021) defines conclusive GERD: AET > 6%, or LA grade C/D esophagitis, or Barrett's ≥ 1 cm.
Management
Lifestyle: Weight loss (most evidence-backed), head-of-bed elevation 6-8 inches, avoid eating 2-3 hours before bedtime, avoid trigger foods (caffeine, chocolate, alcohol, fatty/spicy foods, mint). Pharmacologic: Step-up approach — antacids/alginates for intermittent symptoms → H2RAs (famotidine 20 mg BID) for mild symptoms → PPIs for moderate-severe or erosive disease. Standard PPI dosing: omeprazole 20 mg, lansoprazole 30 mg, esomeprazole 20 mg, pantoprazole 40 mg, rabeprazole 20 mg — all taken 30-60 minutes before first meal of the day. PPI therapy for 8 weeks, then attempt step-down to lowest effective dose. Surgical: Nissen fundoplication (360-degree wrap) or Toupet fundoplication (partial posterior wrap) for PPI-dependent, confirmed GERD with anatomy suitable for surgery. Magnetic sphincter augmentation (LINX device) is an alternative for patients without large hiatal hernias.
06 Barrett's Esophagus Esophageal
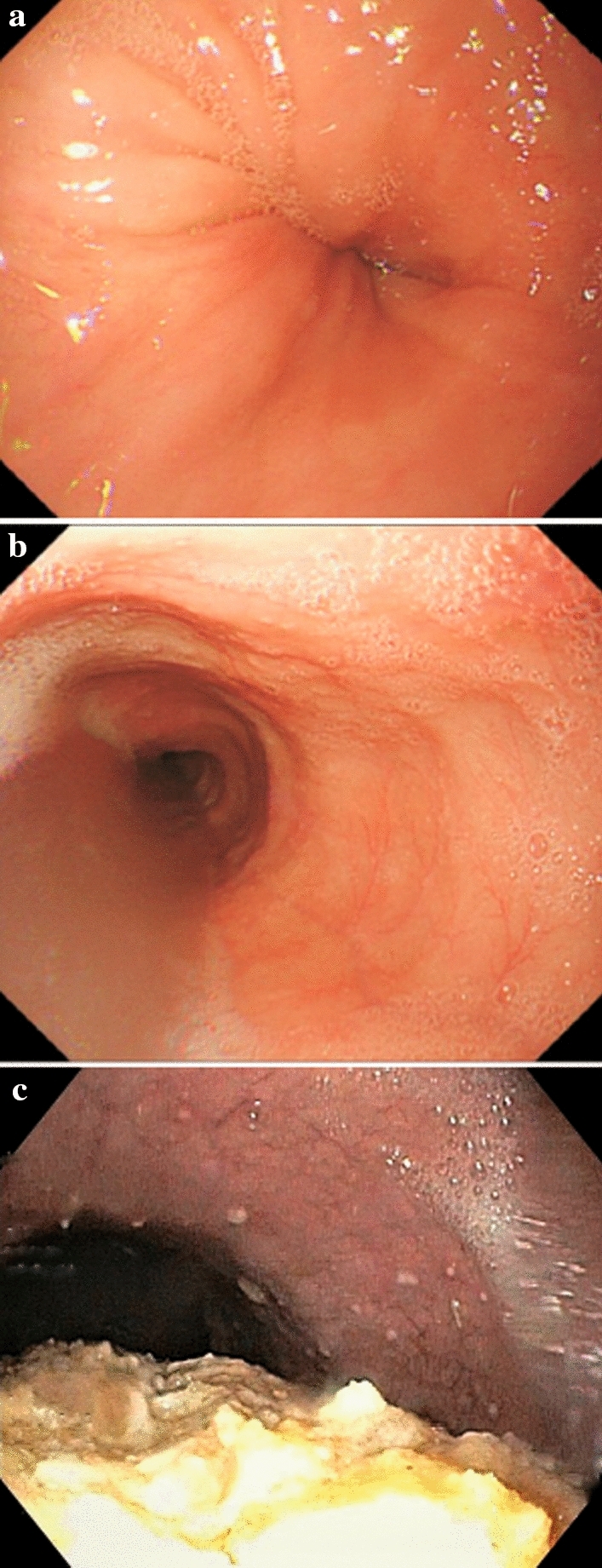
Barrett's esophagus is intestinal metaplasia of the esophageal squamous epithelium — replacement by columnar epithelium with goblet cells — caused by chronic GERD. It is the only known precursor to esophageal adenocarcinoma. Prevalence: 5-15% of GERD patients undergoing endoscopy. Risk factors: chronic GERD (> 5 years), male sex (8:1), white race, age > 50, central obesity, smoking, family history. Diagnosed by endoscopy showing salmon-colored mucosa extending above the GEJ (Prague C&M criteria: C = circumferential extent, M = maximum extent) with biopsy confirmation of intestinal metaplasia with goblet cells.
Surveillance & Management
| Histology | Surveillance Interval | Management |
|---|---|---|
| Non-dysplastic Barrett's | EGD every 3-5 years | PPI therapy, biopsy per Seattle protocol (4-quadrant q2cm) |
| Low-grade dysplasia (LGD) | EGD every 6-12 months OR endoscopic eradication | Confirm with expert GI pathologist; endoscopic eradication therapy (RFA) preferred |
| High-grade dysplasia (HGD) | Endoscopic eradication therapy | Radiofrequency ablation (RFA), endoscopic mucosal resection (EMR) for visible lesions, cryotherapy |
| Intramucosal carcinoma (T1a) | Endoscopic eradication therapy | EMR/ESD for nodular lesions + RFA for flat Barrett's; < 2% lymph node metastasis risk |
| Submucosal invasion (T1b) | Surgical evaluation | Esophagectomy generally recommended; 20% lymph node metastasis risk |
07 Esophageal Cancer Esophageal
Two main histologic types with distinct epidemiology, risk factors, and location:
| Feature | Squamous Cell Carcinoma (SCC) | Adenocarcinoma |
|---|---|---|
| Location | Upper and middle esophagus | Distal esophagus / GEJ |
| Epidemiology | Predominant worldwide; "Asian esophageal belt" | Rising rapidly in Western countries; now most common in US/Europe |
| Risk factors | Alcohol, tobacco (synergistic), caustic injury, achalasia, Plummer-Vinson syndrome, hot beverages, HPV (some) | GERD, Barrett's esophagus, obesity, male sex, white race, tobacco |
| Precursor | Squamous dysplasia | Barrett's with dysplasia |
| 5-year survival (all stages) | ~15-25% | ~15-25% |
Presentation: progressive dysphagia (solids then liquids), weight loss, odynophagia, chest pain. By the time dysphagia occurs, the lumen is typically narrowed to < 13 mm (normal ~20 mm). Staging: EGD with biopsy → EUS for T and N staging (most accurate for locoregional staging) → CT chest/abdomen/pelvis for distant metastases → PET-CT for M staging. Treatment: early (T1a) — endoscopic resection; locally advanced — neoadjuvant chemoradiation (CROSS regimen: carboplatin/paclitaxel + 41.4 Gy) then esophagectomy (van Hagen et al., NEJM 2012); unresectable/metastatic — palliative chemotherapy, immunotherapy (nivolumab + chemo per CheckMate 649), esophageal stenting for dysphagia palliation.
08 Achalasia Esophageal
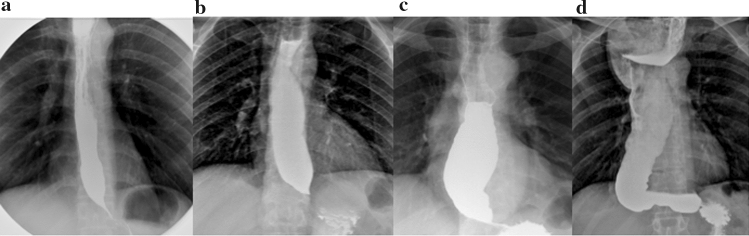
Achalasia is a primary esophageal motility disorder characterized by failure of LES relaxation and absent peristalsis of the esophageal body, caused by loss of inhibitory neurons (nitric oxide/VIP-secreting) in the myenteric (Auerbach's) plexus. Incidence: ~1/100,000 per year. Presentation: progressive dysphagia to both solids AND liquids (distinguishing it from mechanical obstruction), regurgitation of undigested food, chest pain, weight loss, aspiration pneumonia. "Bird's beak" appearance on barium swallow (dilated esophageal body with smooth tapering at the GEJ).
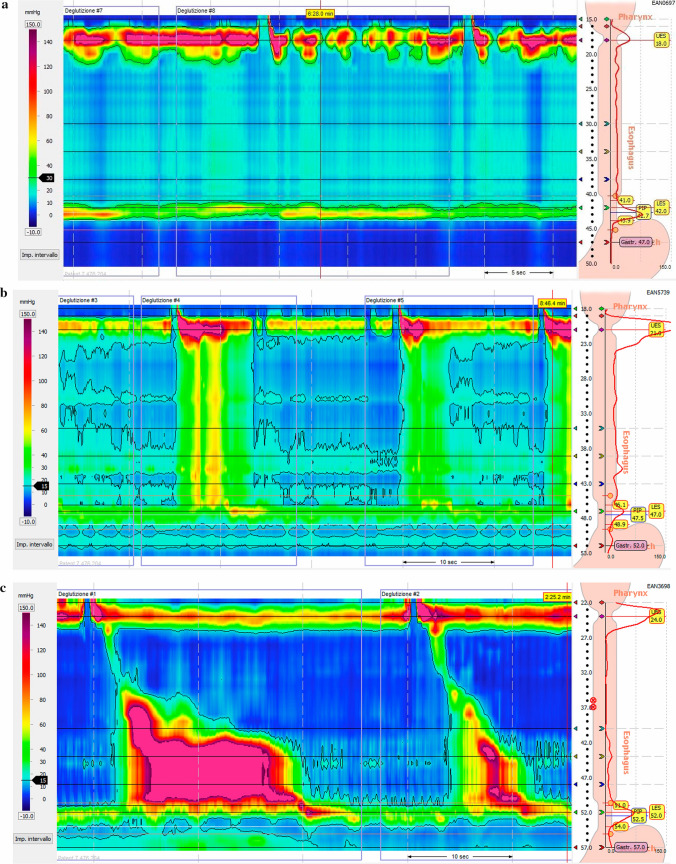
Chicago Classification v4.0 — High-Resolution Manometry
| Type | Manometric Pattern | Treatment Response |
|---|---|---|
| Type I (classic) | Absent peristalsis, elevated IRP (> 15 mmHg), no pressurization | Moderate response to all therapies |
| Type II (with compression) | Absent peristalsis, elevated IRP, panesophageal pressurization in ≥ 20% swallows | Best response to all therapies (~96% success with Heller myotomy or POEM) |
| Type III (spastic) | Absent normal peristalsis, elevated IRP, premature/spastic contractions | Worst response; POEM preferred (can extend myotomy length) |
Treatment: Definitive therapies target the LES. Heller myotomy (laparoscopic, with partial fundoplication to prevent reflux) — gold standard with 85-95% success. Per-oral endoscopic myotomy (POEM) — endoscopic alternative with equivalent short-term efficacy; higher post-procedure GERD rates. Pneumatic dilation (graded balloons: 30 → 35 → 40 mm Rigiflex) — effective but may require repeat sessions; 2-5% perforation risk. Botulinum toxin injection — temporary (3-6 months), reserved for poor surgical candidates. Medical therapy (nitrates, calcium channel blockers) is poorly effective and rarely used.
09 Eosinophilic Esophagitis (EoE) Esophageal
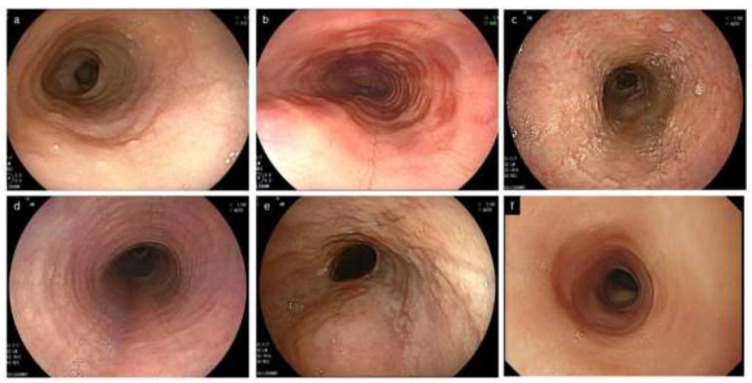
Chronic, immune/antigen-mediated esophageal disease characterized by eosinophilic infiltration of the esophageal mucosa (≥ 15 eosinophils per high-power field on biopsy). Prevalence is rising — now ~1/2,000 adults. Strongly associated with atopic conditions (asthma, eczema, food allergy in 50-70%). Presentation in adults: intermittent dysphagia to solids, food impaction (EoE is the #1 cause of food impaction and esophageal emergency in young adults), chest pain. Endoscopic findings: rings ("trachealization"), linear furrows, white exudates/plaques (eosinophilic microabscesses), strictures, narrow-caliber esophagus ("crepe paper" mucosa that tears easily).
Treatment: PPI therapy — first-line; ~50% respond (PPI-responsive esophageal eosinophilia is now considered part of the EoE spectrum). Swallowed topical corticosteroids — fluticasone MDI (880-1760 mcg/day, swallowed not inhaled) or budesonide oral suspension/slurry (1-2 mg BID). Dupilumab (Dupixent) — FDA-approved 2022, IL-4/IL-13 blocker, 300 mg SC weekly, first biologic for EoE (Dellon et al., NEJM 2022). Dietary elimination — empiric 6-food elimination diet (milk, wheat, egg, soy, nuts, seafood) then sequential reintroduction; 2-food elimination (dairy and wheat) is simpler and effective in ~40%. Endoscopic dilation — for symptomatic strictures; proceed cautiously (higher mucosal tear risk).
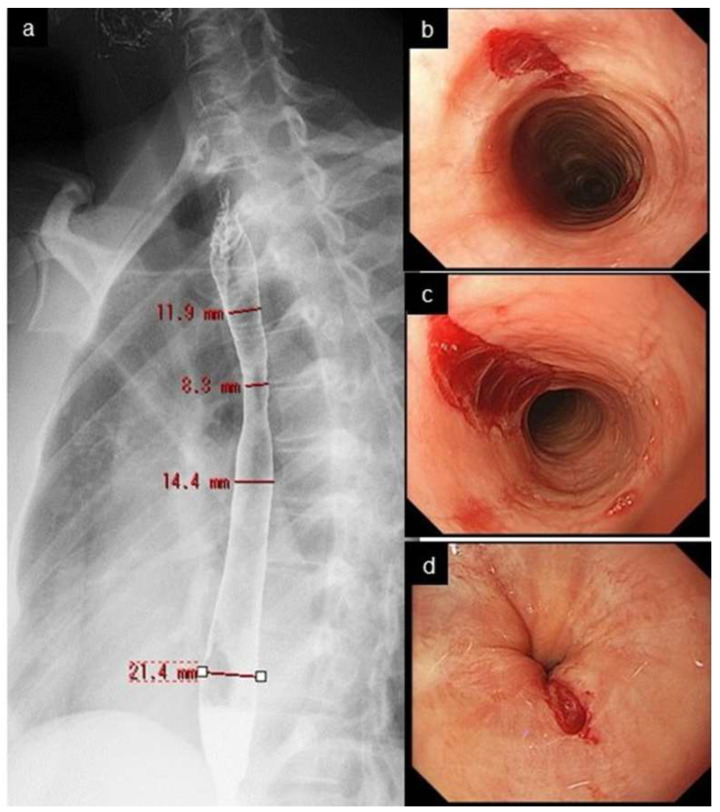
10 Esophageal Varices Esophageal
Dilated submucosal veins in the distal esophagus resulting from portal hypertension (hepatic venous pressure gradient [HVPG] ≥ 10 mmHg). Present in ~50% of cirrhotics; ~30% will bleed within 2 years if untreated. Variceal hemorrhage carries 15-20% mortality per episode. Varices form because portal hypertension diverts blood through portosystemic collaterals, including the left gastric (coronary) vein → esophageal submucosal plexus → azygos system.
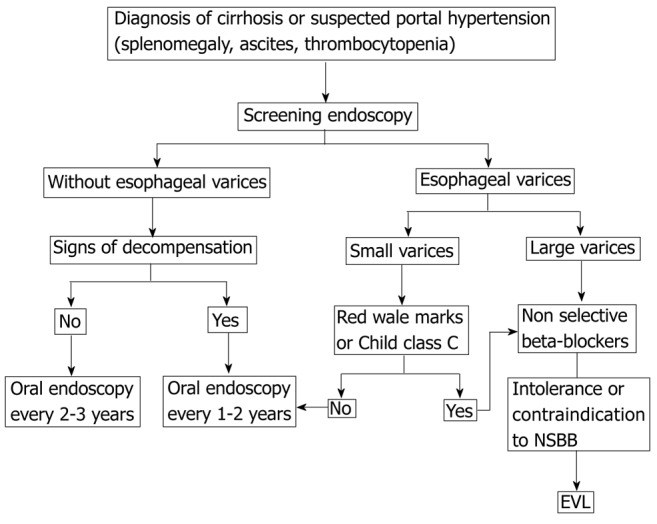
Primary prophylaxis (prevent first bleed): Screen all cirrhotics with EGD at diagnosis. Non-selective beta-blockers (NSBB) — propranolol (20-40 mg BID, titrate to resting HR 55-60) or nadolol (40-80 mg daily), or carvedilol (6.25-12.5 mg daily, most effective NSBB, also reduces intrahepatic resistance). Endoscopic variceal ligation (EVL, banding) — for medium/large varices or NSBB-intolerant patients; repeat every 2-4 weeks until eradication. Baveno VII consensus: patients with compensated cirrhosis and liver stiffness < 20 kPa + platelets > 150k can avoid screening EGD (de Franchis et al., J Hepatol 2022).
Acute variceal bleeding management is covered in Section 47.
11 Peptic Ulcer Disease Gastric
Peptic ulcers are mucosal defects extending through the muscularis mucosae in the stomach or duodenum. Two dominant etiologies account for > 90% of cases: Helicobacter pylori infection (~60-70% of duodenal ulcers, ~30-40% of gastric ulcers) and NSAID use (~20-25%). Other causes: physiologic stress ulcers (Curling's = burns, Cushing's = CNS injury with high acid), Zollinger-Ellison syndrome, Crohn's, CMV, crack cocaine.
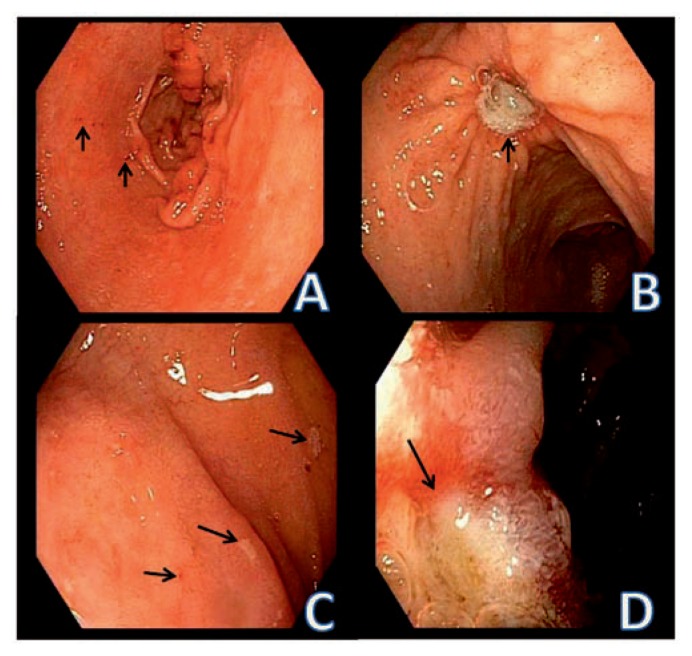
Duodenal vs Gastric Ulcers
| Feature | Duodenal Ulcer | Gastric Ulcer |
|---|---|---|
| Location | D1 (bulb), anterior or posterior wall | Lesser curvature most common; antrum |
| Pain pattern | Epigastric pain 2-5 hours after meals, relieved by food/antacids, nocturnal pain | Pain worsened by eating |
| H. pylori association | ~90% | ~70% |
| Malignancy risk | Essentially zero | 3-5% — always biopsy gastric ulcers |
| Complications | Posterior: GDA erosion → hemorrhage. Anterior: perforation | Hemorrhage, perforation, gastric outlet obstruction |
H. pylori — Diagnosis & Treatment
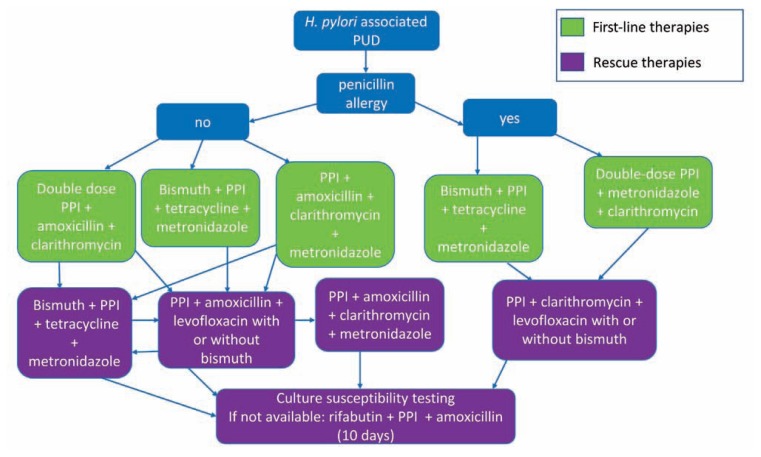
Testing: urea breath test (UBT) and fecal antigen test — preferred non-invasive tests (sensitivity/specificity > 95%). Hold PPIs for 2 weeks and antibiotics/bismuth for 4 weeks before testing to avoid false negatives. Serology (IgG) cannot distinguish active from prior infection — not useful for test of cure. Biopsy-based: rapid urease test (CLO test), histology, culture. Treatment: First-line: bismuth quadruple therapy (bismuth subsalicylate 524 mg QID + metronidazole 250 mg QID + tetracycline 500 mg QID + PPI BID × 14 days) OR concomitant therapy (PPI + amoxicillin 1g + clarithromycin 500 mg + metronidazole 500 mg, all BID × 14 days). Traditional clarithromycin triple therapy is no longer first-line due to rising resistance (> 15% in most US regions). Always confirm eradication ≥ 4 weeks after completing therapy (UBT or fecal antigen).
12 Gastric Cancer Gastric
Gastric adenocarcinoma accounts for ~95% of gastric malignancies. Two histologic types (Lauren classification): intestinal type (well-differentiated, gland-forming, associated with H. pylori → chronic atrophic gastritis → intestinal metaplasia → dysplasia → cancer sequence; declining incidence) and diffuse type (poorly differentiated, signet ring cells, infiltrates the wall producing linitis plastica/"leather bottle stomach"; not declining, worse prognosis, younger patients, CDH1 mutation in hereditary diffuse gastric cancer). Risk factors: H. pylori (strongest modifiable risk, WHO Class I carcinogen), smoking, high-salt diet, pernicious anemia, prior partial gastrectomy (15-20 year latency), family history, blood type A.
Presentation: often asymptomatic until advanced; dyspepsia, early satiety, weight loss, anemia, dysphagia (proximal tumors). Late signs: Virchow's node (left supraclavicular), Sister Mary Joseph nodule (periumbilical), Krukenberg tumor (ovarian metastasis, especially diffuse type), Blumer's shelf (rectal shelf on DRE). Staging: EGD with multiple biopsies → CT → EUS for T/N staging → PET-CT → staging laparoscopy for ≥ T3 or node-positive to exclude peritoneal carcinomatosis. Treatment: early gastric cancer (T1) — endoscopic submucosal dissection (ESD) if criteria met; locally advanced — perioperative chemotherapy (FLOT regimen: 5-FU, leucovorin, oxaliplatin, docetaxel) + gastrectomy with D2 lymphadenectomy (≥ 15 nodes); metastatic — palliative chemotherapy, HER2 testing (trastuzumab if HER2+), PD-L1/MSI testing for immunotherapy eligibility.
13 Gastroparesis Gastric
Delayed gastric emptying in the absence of mechanical obstruction. Three main etiologies: diabetic (~30%, from vagal neuropathy), idiopathic (~35%, often post-viral), and post-surgical (~15%, vagotomy, fundoplication, bariatric surgery). Symptoms: nausea/vomiting (often of partially digested food hours after eating), early satiety, bloating, abdominal pain, weight loss. Diagnosis: gastric emptying scintigraphy (GES) — gold standard. Standard meal (low-fat egg whites with toast and jam labeled with Tc-99m sulfur colloid). Abnormal: > 60% retention at 2 hours OR > 10% retention at 4 hours. Hold medications that affect motility for 48-72 hours before testing (opioids, anticholinergics, GLP-1 agonists).
Treatment: Dietary modification (small, frequent, low-fat, low-fiber meals; liquid calorie supplementation). Metoclopramide (Reglan) — dopamine antagonist/prokinetic, 5-10 mg AC and QHS; FDA black box warning for tardive dyskinesia with use > 12 weeks. Domperidone — peripheral dopamine antagonist, available via FDA compassionate use program; fewer CNS side effects but QTc prolongation risk. Erythromycin — motilin agonist, 50-100 mg IV or 250 mg PO AC; potent prokinetic but tachyphylaxis develops in 2-4 weeks. Gastric electrical stimulation (GES/Enterra) — surgically implanted neurostimulator, FDA humanitarian device exemption; primarily improves nausea/vomiting, less effect on gastric emptying. Refractory: pyloric interventions (G-POEM = gastric peroral endoscopic myotomy of the pylorus; pyloroplasty), jejunal feeding tube for nutritional support.
14 Zollinger-Ellison Syndrome (ZES) Gastric
Gastrin-secreting neuroendocrine tumor (gastrinoma) causing severe acid hypersecretion → refractory/multiple peptic ulcers, secretory diarrhea, and esophagitis. 80% sporadic, 20% associated with MEN1 (parathyroid hyperplasia + pituitary adenoma + pancreatic/duodenal NETs). Gastrinomas: 60-80% in the "gastrinoma triangle" (confluence of cystic/CBD, junction of D2-D3, junction of neck/body of pancreas); 60-90% are malignant with lymph node or liver metastases at diagnosis.
Diagnosis: Fasting serum gastrin > 1000 pg/mL with gastric pH < 2 is virtually diagnostic. For equivocal gastrin levels (110-1000 pg/mL), perform secretin stimulation test: IV secretin 0.4 mcg/kg bolus; gastrin rise > 120 pg/mL above baseline is positive (in normal patients, secretin suppresses gastrin). Must exclude other causes of hypergastrinemia: PPI use (most common cause of elevated gastrin — must stop for 1-2 weeks), chronic atrophic gastritis/pernicious anemia, renal failure, retained antrum post-Billroth II. Tumor localization: CT/MRI, somatostatin receptor scintigraphy (OctreoScan), EUS (most sensitive for small duodenal tumors), Ga-68 DOTATATE PET (most sensitive overall).
Treatment: High-dose PPI (omeprazole 60-120 mg/day, titrate to BAO < 10 mEq/hr). Surgical resection if localized and no MEN1 (duodenotomy with enucleation of duodenal gastrinomas, distal pancreatectomy for pancreatic body/tail tumors). MEN1-associated gastrinomas are usually multiple and small — surgery is controversial; medical acid suppression is the mainstay. Metastatic disease: somatostatin analogs (octreotide LAR, lanreotide), everolimus, sunitinib, PRRT (Lu-177 DOTATATE), hepatic-directed therapy.
15 Celiac Disease Small Bowel
Immune-mediated enteropathy triggered by gluten (gliadin fraction of wheat, barley, rye) in genetically susceptible individuals (HLA-DQ2 in ~95%, HLA-DQ8 in ~5%). Prevalence: ~1% of the general population (the majority undiagnosed). Pathophysiology: tissue transglutaminase (tTG) deaminates gliadin peptides → presentation by HLA-DQ2/DQ8 to CD4+ T cells → inflammatory response → villous atrophy, crypt hyperplasia, intraepithelial lymphocytosis in the proximal small bowel (duodenum/jejunum).
Presentation: Classic: chronic diarrhea, steatorrhea, weight loss, failure to thrive (children), iron deficiency anemia (most common lab abnormality in adults). Atypical/silent: osteoporosis, dermatitis herpetiformis (pathognomonic — IgA deposits in skin), elevated transaminases, infertility/recurrent miscarriage, peripheral neuropathy, dental enamel defects. Associated conditions: type 1 diabetes, autoimmune thyroiditis, Down syndrome, Turner syndrome, IgA deficiency (check total IgA — if deficient, tTG-IgA will be falsely negative; use deamidated gliadin peptide IgG).
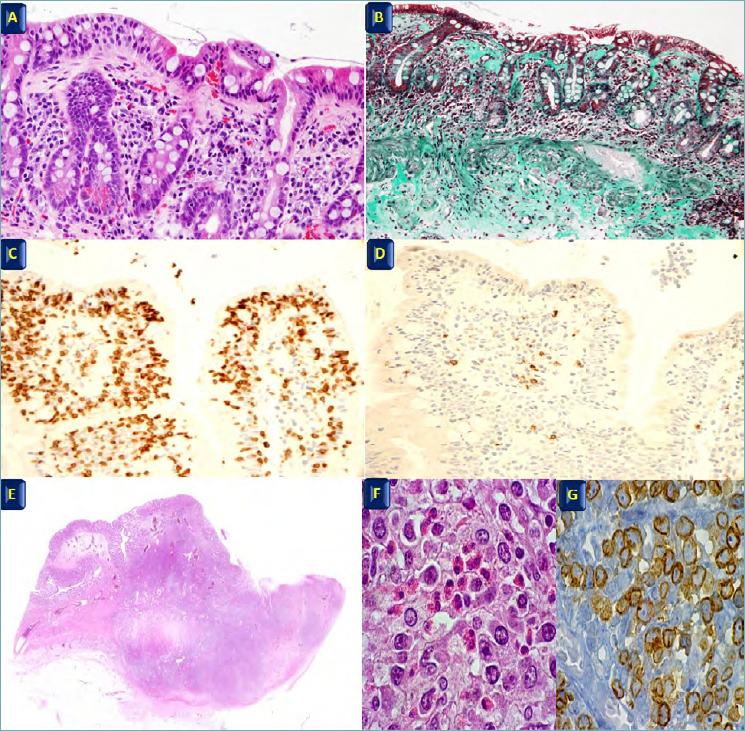
Diagnosis: Serologic screening while patient is on a gluten-containing diet: tTG-IgA (sensitivity 93-96%, specificity 96-98%) + total IgA level. Confirm with duodenal biopsy (minimum 4 biopsies from D2, 2 from D1): Marsh classification — Type 0 (normal), Type 1 (intraepithelial lymphocytosis > 25/100 enterocytes), Type 2 (+ crypt hyperplasia), Type 3a (partial villous atrophy), Type 3b (subtotal), Type 3c (total villous atrophy). Treatment: Lifelong strict gluten-free diet — the only proven therapy. Response: symptoms improve in weeks, serologies normalize in 6-12 months, histology may take 1-2 years. Non-responsive celiac: verify dietary compliance (most common cause of persistent symptoms), exclude refractory celiac disease (Type I — polyclonal T cells, responds to steroids; Type II — clonal T cells, high risk of enteropathy-associated T-cell lymphoma).
16 Small Bowel Obstruction (SBO) Small Bowel
Mechanical obstruction of the small intestine. Most common cause: postoperative adhesions (~65-75%), followed by hernias (~15%), malignancy (~10%), and Crohn's disease (~5%). Presentation: colicky abdominal pain, nausea/vomiting (bilious, earlier with more proximal obstruction), abdominal distension, obstipation (absence of flatus and stool — late sign). Strangulation (compromised blood supply) is the feared complication — occurs in 5-10% and is suggested by constant (not colicky) pain, fever, tachycardia, peritoneal signs, leukocytosis, and lactic acidosis.
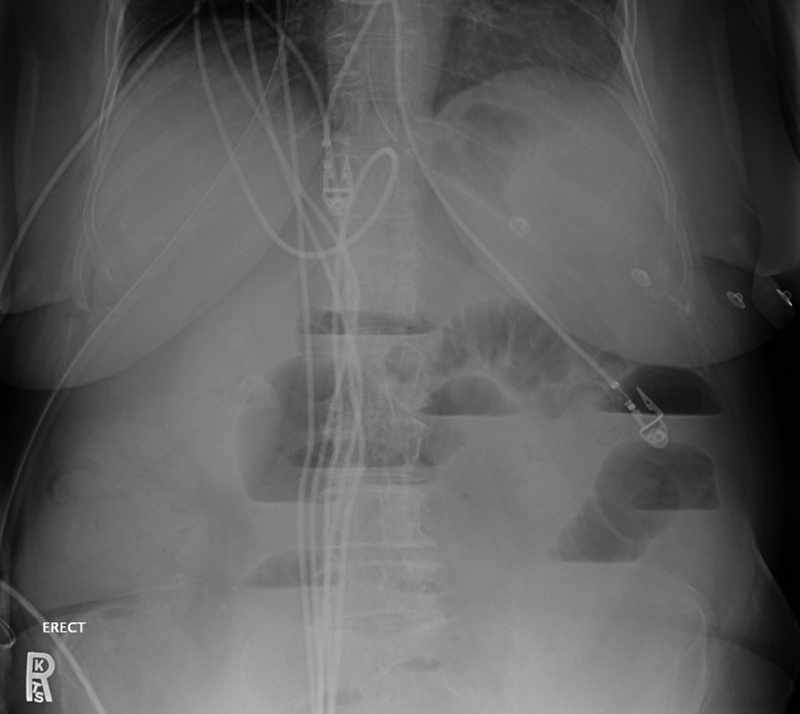
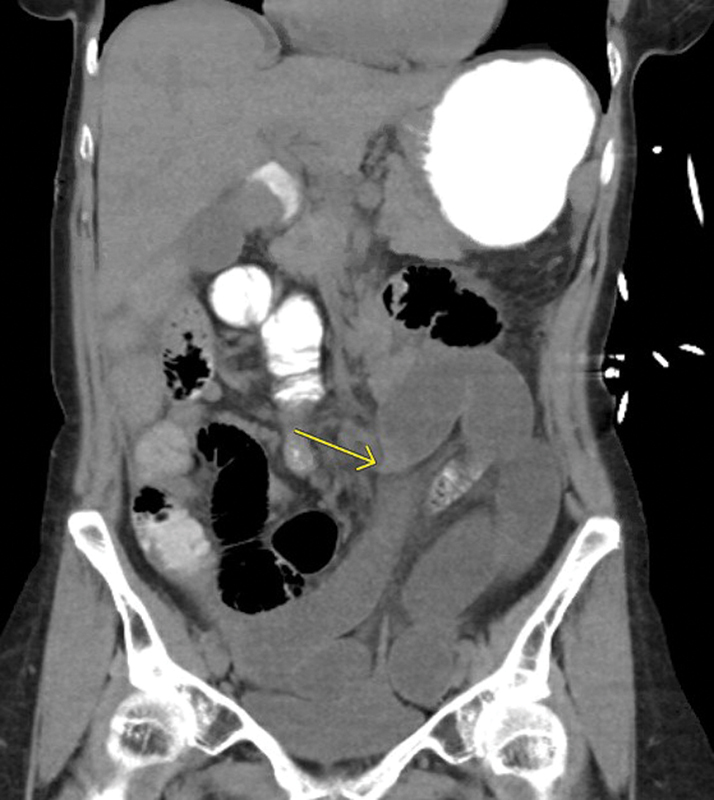
Imaging: CT abdomen/pelvis with IV contrast — sensitivity > 90% for SBO; identifies transition point, identifies cause (adhesive band, hernia, tumor), and detects signs of strangulation (mesenteric haziness, bowel wall thickening, reduced wall enhancement, portal venous gas). Abdominal X-ray: dilated small bowel loops (> 3 cm), air-fluid levels, absence of colonic gas (in complete obstruction). Management: NPO, NGT decompression, IV fluids, electrolyte correction. Partial SBO/adhesive: trial of conservative management (48-72 hours); water-soluble contrast (Gastrografin) challenge — appearance of contrast in the colon at 24 hours predicts resolution (sensitivity 97%) and is also therapeutic (Defined guidelines for adhesive SBO management). Indications for surgery: complete obstruction, closed-loop obstruction, signs of strangulation/ischemia, hernia as etiology, failure to resolve with conservative management.
17 Carcinoid Tumors (Neuroendocrine Tumors) Small Bowel
Small bowel NETs (carcinoid tumors) are the most common small bowel malignancy and the most common neuroendocrine tumor of the GI tract. Arise from enterochromaffin cells, predominantly in the ileum. Often multifocal (30%). Secrete serotonin (5-HT), which is normally cleared by the liver — so carcinoid syndrome (episodic flushing, watery diarrhea, wheezing, right-sided heart disease) occurs only when serotonin bypasses hepatic metabolism, meaning: liver metastases (most common trigger), retroperitoneal/ovarian tumors draining into systemic veins, or very high tumor burden. Carcinoid heart disease (Hedinger syndrome): serotonin causes fibrosis of right-sided cardiac valves → tricuspid regurgitation and pulmonary stenosis (left-sided valves are protected by MAO in the lungs).
Diagnosis: 24-hour urine 5-HIAA (5-hydroxyindoleacetic acid, serotonin metabolite) — sensitivity ~70%, specificity ~90%. Chromogranin A (CgA) — nonspecific but useful for tumor burden tracking (falsely elevated by PPIs). Imaging: CT/MRI, Ga-68 DOTATATE PET-CT (most sensitive, > 90%). WHO grading by Ki-67 index: G1 (< 3%), G2 (3-20%), G3 (> 20%). Treatment: Surgical resection with wide mesenteric lymphadenectomy (even in metastatic disease, debulking improves symptoms and survival). Somatostatin analogs — octreotide LAR 20-30 mg IM monthly or lanreotide 120 mg SC monthly; control symptoms and slow tumor growth (PROMID and CLARINET trials). PRRT — Lu-177 DOTATATE (Lutathera) for progressive somatostatin receptor-positive NETs (NETTER-1 trial: 79% reduction in progression risk). Carcinoid crisis prevention: pre-procedural octreotide (500 mcg IV bolus) before surgery/biopsy; avoid catecholamines and opioids during anesthesia.
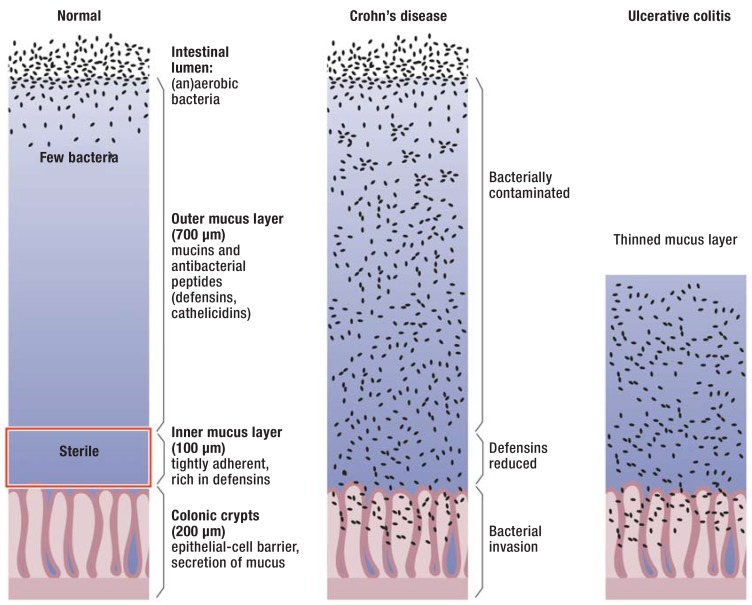
18 Small Bowel Crohn's Disease Small Bowel
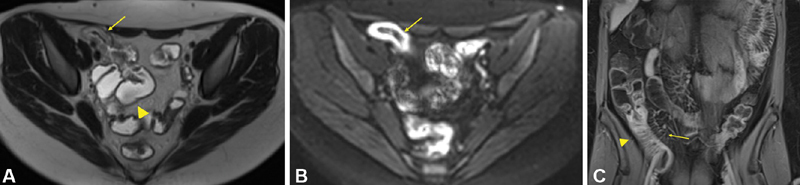
Crohn's disease is a transmural, chronic inflammatory condition that can affect any part of the GI tract from mouth to anus but involves the small bowel (especially terminal ileum) in ~70% of cases, with isolated ileal disease in ~30%. Unlike ulcerative colitis, Crohn's is characterized by skip lesions (discontinuous), transmural inflammation (leading to fistulae, abscesses, strictures), and granulomas on biopsy (present in only ~30% but pathognomonic when found). Terminal ileal disease may mimic appendicitis (RLQ pain, tenderness). Small bowel complications include strictures (causing partial SBO), fistulae (enteroenteric, enterocutaneous, enterovesical, enterovaginal), and abscesses. Full Crohn's disease discussion, including Montreal classification and management, is in Section 20.
19 Small Intestinal Bacterial Overgrowth (SIBO) Small Bowel
Excessive colonization of the small intestine by bacteria normally found in the colon, leading to malabsorption and GI symptoms. Normal small bowel bacterial count is < 103 CFU/mL; SIBO is defined as ≥ 105 CFU/mL (though this threshold is debated). Risk factors: anything that impairs small bowel motility or anatomy — post-surgical (Roux-en-Y, blind loops, strictures), gastroparesis, scleroderma, diabetes, achlorhydria (chronic PPI use), chronic opioid use, IBS (overlap), intestinal pseudo-obstruction, immunodeficiency.
Presentation: Bloating (most common), flatulence, diarrhea, abdominal discomfort, steatorrhea (bacterial deconjugation of bile salts → fat malabsorption), B12 deficiency (bacteria consume it — unlike terminal ileum disease), folate excess (bacteria produce it). Diagnosis: Glucose or lactulose hydrogen/methane breath test — non-invasive, widely available; sensitivity 55-80%, specificity 80-85%. Jejunal aspirate and culture is the gold standard but rarely performed. Treatment: Rifaximin (Xifaxan) 550 mg TID × 14 days — non-absorbable antibiotic, first-line; 65-70% response rate. Alternatives: ciprofloxacin, metronidazole, amoxicillin-clavulanate, or neomycin (especially for methane-predominant). Address underlying cause. Nutritional repletion: B12, fat-soluble vitamins (A, D, E, K), iron. Recurrence is common (45% within 9 months) — cycling antibiotics or low-dose prophylaxis may be needed.
20 Crohn's Disease IBD
Crohn's disease (CD) is a chronic, relapsing, transmural inflammatory condition that can affect any segment of the GI tract. Bimodal age distribution: peaks at 15-35 and 55-70. Pathogenesis involves dysregulated immune response to commensal gut microbiota in genetically susceptible individuals (NOD2/CARD15 is the most strongly associated gene). Smoking doubles the risk and worsens disease course. Unlike UC, CD is not cured by surgery — recurrence at anastomosis is the rule.
Montreal Classification
| Category | Classification | Description |
|---|---|---|
| Age at diagnosis (A) | A1 | ≤ 16 years |
| A2 | 17-40 years | |
| A3 | > 40 years | |
| Location (L) | L1 | Ileal (terminal ileum) |
| L2 | Colonic | |
| L3 | Ileocolonic (most common, ~50%) | |
| L4 | Upper GI (modifier, added to L1-L3) | |
| Behavior (B) | B1 | Non-stricturing, non-penetrating (inflammatory) |
| B2 | Stricturing | |
| B3 | Penetrating (fistulae, abscesses) | |
| Modifier | p | Perianal disease (added to B1-B3) |
Clinical Presentation
Depends on location and behavior. Ileal: RLQ pain, diarrhea (often non-bloody), B12 deficiency, bile salt malabsorption. Colonic: bloody diarrhea, urgency, perianal disease. Stricturing: obstructive symptoms (postprandial pain, N/V, distension). Penetrating: abscesses (fever, localized tenderness), fistulae (drainage, pneumaturia). Perianal disease in up to 30% — fistulae, abscesses, skin tags, fissures (often lateral, unlike idiopathic midline fissures). Extraintestinal manifestations: erythema nodosum, pyoderma gangrenosum, uveitis/episcleritis, peripheral arthritis (correlates with bowel activity), ankylosing spondylitis/sacroiliitis (independent of bowel activity), PSC (more common in UC but occurs in CD), aphthous stomatitis, cholelithiasis, nephrolithiasis (oxalate stones from fat malabsorption).
Medical Management
Induction: Corticosteroids (prednisone 40-60 mg daily, budesonide 9 mg daily for ileal/right colonic — less systemic toxicity) for moderate-severe flares — never for maintenance. Maintenance (step-up approach increasingly replaced by early biologic use): Immunomodulators — azathioprine 2-2.5 mg/kg/day or 6-mercaptopurine 1-1.5 mg/kg/day (check TPMT/NUDT15 before starting — metabolizer status affects dosing and toxicity), methotrexate 25 mg SC weekly (especially for patients who fail thiopurines). Biologics — see Section 22. Current practice increasingly favors top-down therapy: early biologic ± immunomodulator for moderate-severe disease rather than waiting for failure of conventional agents.
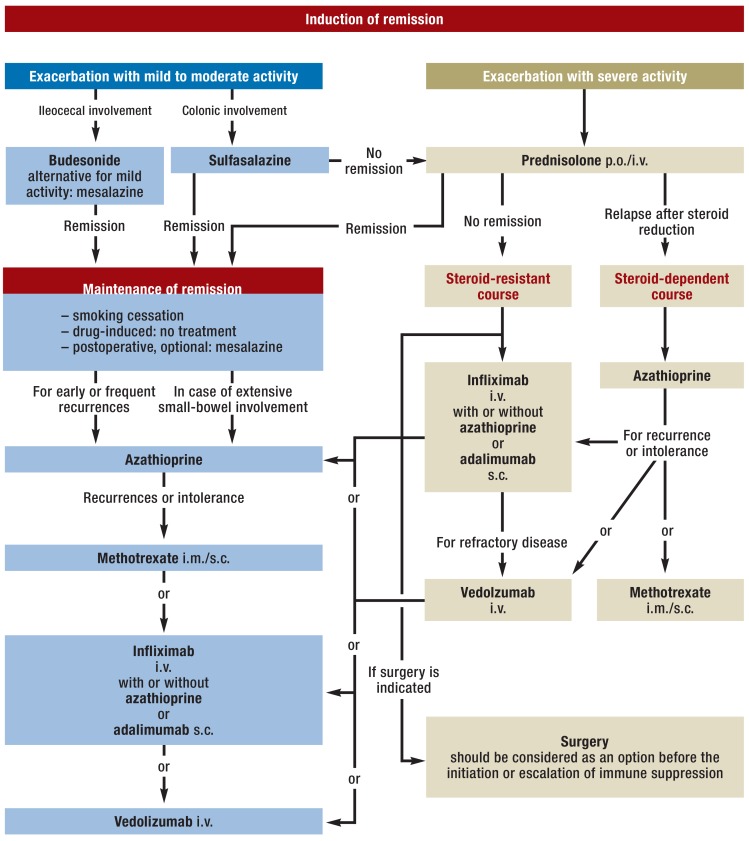
Surgical Indications
Surgery is needed in 70-80% of CD patients over their lifetime. Indications: medically refractory disease, fibrostenotic strictures (not responsive to anti-inflammatory therapy), abscess/fistula not amenable to medical management, perforation, hemorrhage, dysplasia/cancer. Principle: conserve bowel — strictureplasty (Heineke-Mikulicz for short strictures, Finney for longer) rather than resection when possible. Post-surgical endoscopic recurrence at the anastomosis is near-universal — prophylaxis with anti-TNF or thiopurine started within 2-4 weeks of surgery; colonoscopy at 6-12 months post-op to assess recurrence (Rutgeerts score: i0-i4).
21 Ulcerative Colitis IBD
Ulcerative colitis (UC) is a chronic inflammatory disease confined to the mucosa and submucosa of the colon and rectum, extending proximally in a continuous pattern from the rectum (always involved). Unlike CD, there are no skip lesions, no transmural disease, no granulomas, and no small bowel involvement (except "backwash ileitis" in severe pancolitis). UC is cured by proctocolectomy.
Extent Classification (Montreal)
| Classification | Extent | Frequency | Management Implications |
|---|---|---|---|
| E1 — Proctitis | Rectum only (below rectosigmoid junction) | ~30% | Topical therapy often sufficient (mesalamine suppositories/enemas) |
| E2 — Left-sided | To splenic flexure | ~40% | Combination oral + topical mesalamine; moderate disease risk |
| E3 — Pancolitis | Extends proximal to splenic flexure | ~30% | Highest cancer risk; higher biologic/immunomodulator need; highest colectomy rate |
Severity — Truelove and Witts Criteria (Modified)
| Parameter | Mild | Moderate | Severe |
|---|---|---|---|
| Bloody stools/day | < 4 | 4-6 | > 6 |
| Pulse | Normal | ≤ 90 | > 90 |
| Temperature | Normal | ≤ 37.8°C | > 37.8°C |
| Hemoglobin | > 11.5 g/dL | 10.5-11.5 | < 10.5 g/dL |
| ESR | < 20 | 20-30 | > 30 |
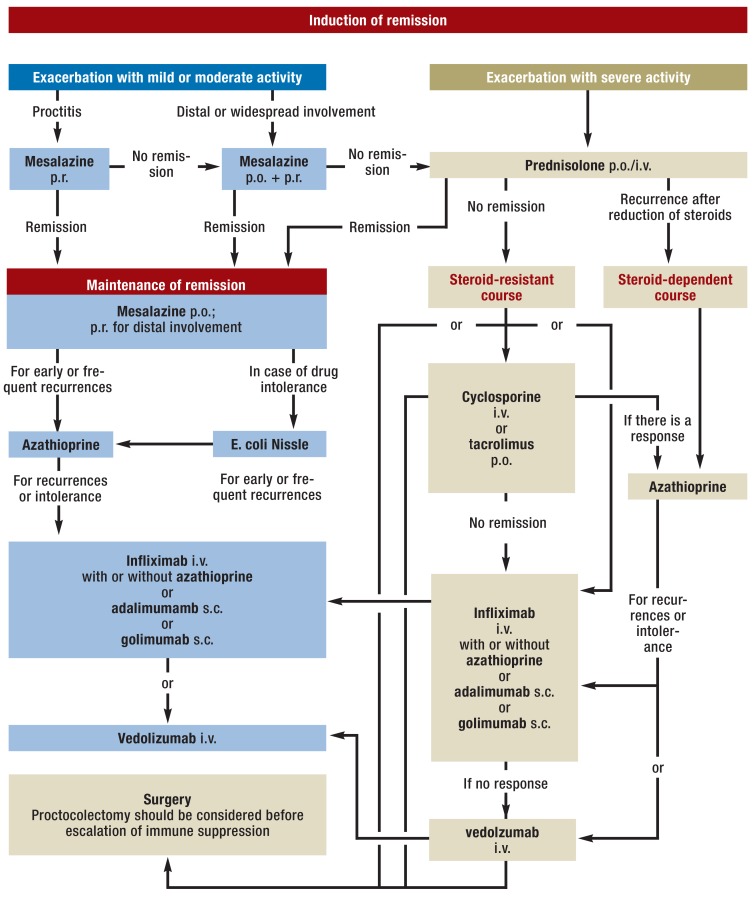
Medical Management
Mild-moderate: 5-ASA (mesalamine/5-aminosalicylic acid) is the backbone — oral (Asacol HD 1.6g TID, Lialda 4.8g daily, Pentasa 1g QID) + topical (Rowasa enema 4g QHS, Canasa suppository 1g daily for proctitis). Combination oral + rectal 5-ASA is superior to either alone. Moderate-severe: Corticosteroids for induction (prednisone, IV methylprednisolone for severe/hospitalized). Maintenance: Thiopurines (azathioprine, 6-MP), biologics (see Section 22), tofacitinib (Xeljanz), ozanimod (Zeposia). Acute severe UC (hospitalized): IV methylprednisolone 60 mg/day; if no response by day 3-5, rescue therapy with infliximab or cyclosporine, or proceed to colectomy. The Travis criteria (day 3 stool frequency > 8, or 3-8 plus CRP > 45) predict steroid failure and need for rescue therapy or surgery.
Colectomy Indications
Fulminant colitis/toxic megacolon (transverse colon > 6 cm + systemic toxicity) refractory to medical rescue, medically refractory chronic disease, dysplasia or cancer on surveillance. Standard procedure: total proctocolectomy with ileal pouch-anal anastomosis (IPAA / J-pouch) — restores intestinal continuity. Pouchitis (inflammation of the J-pouch) occurs in 40-50% and responds to ciprofloxacin or metronidazole. CRC surveillance: begin 8-10 years after diagnosis of pancolitis (or left-sided UC), with chromoendoscopy-guided biopsies every 1-3 years.
22 Biologic & Advanced Therapies for IBD IBD
| Drug (Brand) | Class / Target | Dosing | Indications | Key Pearls |
|---|---|---|---|---|
| Infliximab (Remicade) | Anti-TNF (chimeric mAb) | 5 mg/kg IV at 0, 2, 6 wks then q8wk | CD and UC (moderate-severe); fistulizing CD | Most studied biologic in IBD; combination with thiopurine (combo therapy) reduces immunogenicity and improves efficacy (SONIC trial); check for latent TB before starting |
| Adalimumab (Humira) | Anti-TNF (fully human mAb) | 160 mg SC → 80 mg wk 2 → 40 mg q2wk | CD and UC | SC self-administration; multiple biosimilars available |
| Certolizumab pegol (Cimzia) | Anti-TNF (PEGylated Fab') | 400 mg SC at 0, 2, 4 wks → q4wk | CD only (not FDA-approved for UC) | Does not cross placenta — preferred in pregnancy |
| Vedolizumab (Entyvio) | Anti-integrin (alpha4-beta7) | 300 mg IV at 0, 2, 6 wks → q8wk | CD and UC | Gut-selective — lower systemic immunosuppression; slower onset than anti-TNFs; better for UC than CD |
| Ustekinumab (Stelara) | Anti-IL-12/23 (p40 subunit) | Weight-based IV induction → 90 mg SC q8wk | CD and UC | Favorable safety profile; effective in anti-TNF failures |
| Risankizumab (Skyrizi) | Anti-IL-23 (p19 subunit) | 600 mg IV at 0, 4, 8 wks → 360 mg SC q8wk | CD (FDA 2023); UC (FDA 2024) | Selective IL-23 blockade; ADVANCE/MOTIVATE trials showed superiority over placebo |
| Tofacitinib (Xeljanz) | JAK inhibitor (JAK1/3) | 10 mg PO BID induction → 5 mg BID maintenance | UC only | Oral small molecule; VTE/cardiovascular risk at higher doses; herpes zoster reactivation risk; not for CD |
| Upadacitinib (Rinvoq) | JAK inhibitor (selective JAK1) | 45 mg PO daily induction → 15-30 mg daily maintenance | UC and CD | Selective JAK1 may have better safety profile than tofacitinib; rapid onset |
| Ozanimod (Zeposia) | S1P receptor modulator | 0.92 mg PO daily (after titration) | UC | Oral; first-dose cardiac monitoring (bradycardia); not for CD |
23 Colorectal Cancer Screening Colorectal
Colorectal cancer (CRC) is the 3rd most common cancer and 2nd leading cause of cancer death in the US. Lifetime risk: ~4.5%. The adenoma-carcinoma sequence (normal mucosa → adenomatous polyp → carcinoma) takes ~10-15 years, making CRC highly amenable to screening and prevention. The US Preventive Services Task Force (USPSTF) recommends screening from age 45 to 75 for average-risk adults (USPSTF, JAMA 2021).
Screening Modalities
| Test | Interval | Sensitivity for CRC | Notes |
|---|---|---|---|
| Colonoscopy | Every 10 years | ~95% | Gold standard; diagnostic + therapeutic; requires bowel prep and sedation |
| FIT (fecal immunochemical test) | Annual | 74% per test (95% over 3 rounds) | Detects human hemoglobin; no dietary restrictions; preferred stool test |
| FIT-DNA (Cologuard) | Every 3 years | 92% for CRC, 42% for advanced adenomas | Combines FIT + methylated DNA markers; higher false-positive rate |
| CT colonography | Every 5 years | ~90% for polyps ≥ 10 mm | Requires bowel prep; cannot biopsy; extracolonic findings |
| Flexible sigmoidoscopy | Every 5 years (or 10 with FIT) | ~70% (limited to left colon) | Declining use; misses right-sided lesions |
High-risk screening: Family history of CRC in first-degree relative < 60 years or ≥ 2 first-degree relatives at any age: begin screening at age 40 or 10 years before the youngest diagnosis, whichever is earlier; colonoscopy every 5 years. IBD: begin 8-10 years after pancolitis diagnosis (see Section 21). Lynch syndrome, FAP, and other hereditary syndromes have specific protocols (Section 49).
24 Polyp Classification & Management Colorectal
Paris Endoscopic Classification
| Type | Morphology | Description |
|---|---|---|
| 0-Ip | Polypoid, pedunculated | Attached by a stalk |
| 0-Isp | Polypoid, subpedunculated | Short or broad stalk |
| 0-Is | Polypoid, sessile | Broad-based, no stalk |
| 0-IIa | Non-polypoid, slightly elevated | Flat, elevated < 2.5 mm above mucosa |
| 0-IIb | Non-polypoid, flat | No elevation or depression |
| 0-IIc | Non-polypoid, slightly depressed | Depressed below mucosa — highest malignancy risk |
| 0-III | Non-polypoid, excavated | Ulcerated — almost always malignant |
Histologic Classification
| Type | Malignant Potential | Key Features |
|---|---|---|
| Tubular adenoma | Low (5% if < 1 cm) | Most common adenoma (~70-85%); villous component increases risk |
| Tubulovillous adenoma | Intermediate (20%) | Mixed glandular pattern |
| Villous adenoma | High (40%) | Finger-like projections; classically causes secretory diarrhea with mucus |
| Sessile serrated lesion (SSL) | Moderate | Serrated pathway to CRC (via BRAF mutation → CpG island methylation); typically right colon; flat, difficult to see |
| Traditional serrated adenoma | Moderate | Rare; left colon; eosinophilic cytoplasm |
| Hyperplastic polyp | Negligible | No malignant potential (unless > 1 cm, right colon, or in serrated polyposis syndrome) |
Post-Polypectomy Surveillance
| Findings at Baseline Colonoscopy | Surveillance Interval |
|---|---|
| 1-2 tubular adenomas < 10 mm | 7-10 years |
| 3-4 tubular adenomas < 10 mm | 3-5 years |
| ≥ 5 adenomas, or ≥ 10 mm, or tubulovillous/villous, or HGD | 3 years |
| Piecemeal resection of ≥ 20 mm | 6 months (to confirm complete resection), then 1 year, then 3 years |
| SSL < 10 mm without dysplasia | 5-10 years |
| SSL ≥ 10 mm or with dysplasia | 3 years |
| Hyperplastic polyps (rectosigmoid, < 10 mm) | 10 years (average risk) |
25 Diverticular Disease Colorectal
Diverticulosis — outpouchings of the colonic mucosa and submucosa through the muscularis propria at points of vascular penetration (vasa recta). Present in > 60% of adults over age 60. Predominantly left-sided (sigmoid) in Western populations; right-sided in Asian populations. Risk factors: low-fiber diet, obesity, physical inactivity, aging, connective tissue disorders (Marfan's, Ehlers-Danlos). Most patients are asymptomatic (95%).
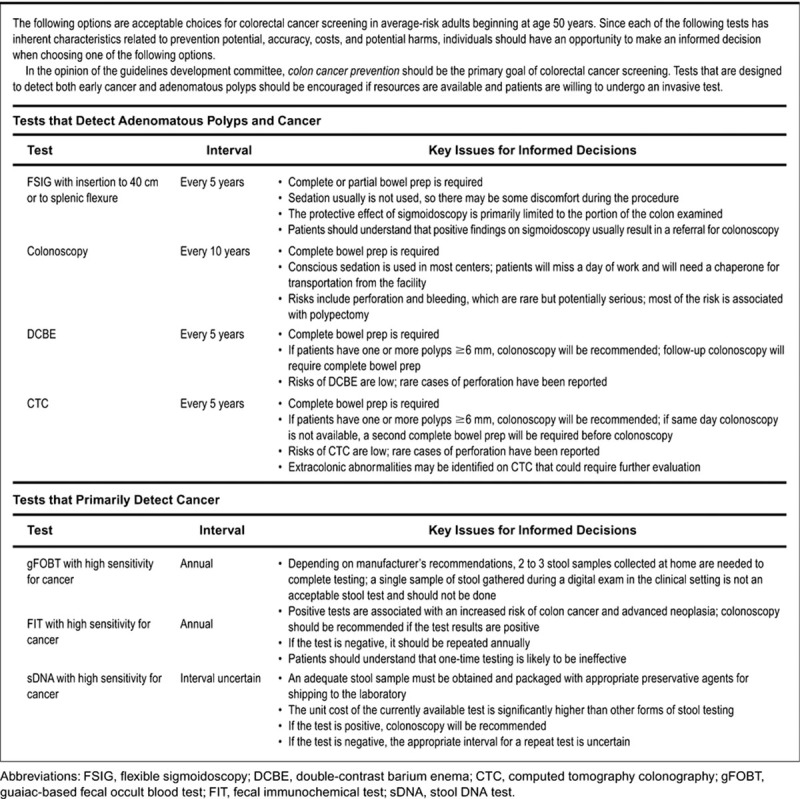
Complications: Acute diverticulitis (~4% lifetime risk in those with diverticulosis): microperforation of a diverticulum → localized inflammation/infection. Presentation: LLQ pain, fever, leukocytosis, ± peritoneal signs. CT with IV contrast (no oral contrast needed): pericolic fat stranding, wall thickening, ± abscess, ± free air. Hinchey classification: Stage I (pericolic/mesenteric abscess), Stage II (pelvic abscess), Stage III (purulent peritonitis), Stage IV (fecal peritonitis). Management: uncomplicated — antibiotics (ciprofloxacin + metronidazole or amoxicillin-clavulanate × 7-10 days); recent data support observation without antibiotics for mild cases. Complicated (abscess > 3-4 cm): CT-guided percutaneous drainage + antibiotics; < 3 cm: antibiotics alone. Stage III/IV: surgical emergency — Hartmann's procedure (sigmoid resection + end colostomy) or primary anastomosis with diverting ileostomy. Diverticular bleeding: painless, brisk hematochezia (diverticulosis is the most common cause of massive lower GI bleeding in adults > 60); right-sided diverticula bleed more often than left-sided despite left-sided predominance. 75-80% stop spontaneously; urgent colonoscopy after rapid prep can identify and treat the source.
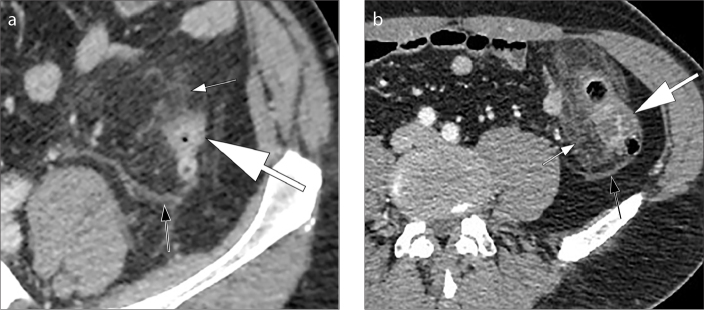
26 Clostridioides difficile Infection (CDI) Colorectal
C. difficile is the most common healthcare-associated infection and the most common cause of antibiotic-associated diarrhea. Toxin A (enterotoxin) and toxin B (cytotoxin) cause mucosal damage, inflammation, and pseudomembrane formation. Risk factors: antibiotic exposure (fluoroquinolones, clindamycin, cephalosporins are highest risk, but any antibiotic can cause it), age > 65, hospitalization, PPI use, immunosuppression. Hypervirulent strain NAP1/BI/027 (ribotype 027) produces more toxin and is associated with severe/recurrent disease.
Diagnosis
Preferred approach: two-step algorithm — GDH (glutamate dehydrogenase) antigen screen (sensitive, detects C. diff organism) + toxin EIA (specific, detects toxin production). If GDH+/toxin-: reflex to NAAT (PCR) for tcdB gene. PCR alone is highly sensitive but detects colonization (which does not require treatment) — should not be used as sole test. Do NOT test formed stool. Do NOT test for cure (stool remains positive for weeks).
Severity & Treatment (IDSA/SHEA 2021 Guidelines)
| Severity | Criteria | Treatment |
|---|---|---|
| Non-severe (initial) | WBC ≤ 15,000; Cr < 1.5 mg/dL | Fidaxomicin (Dificid) 200 mg BID × 10 days (preferred) OR vancomycin 125 mg PO QID × 10 days |
| Severe | WBC ≥ 15,000 OR Cr ≥ 1.5 | Vancomycin 125 mg PO QID × 10 days OR fidaxomicin 200 mg BID × 10 days |
| Fulminant | Hypotension, ileus, megacolon, organ failure | Vancomycin 500 mg PO/NG QID + vancomycin 500 mg rectal QID + metronidazole 500 mg IV q8h; surgical consult for subtotal colectomy |
| First recurrence | Symptoms recur within 2-8 weeks | Fidaxomicin (if initial was vanco) OR vancomycin pulse-taper (125 mg QID × 14d → BID × 7d → daily × 7d → q2-3d × 2-8 wks) |
| ≥ Second recurrence | Multiple recurrences | Fidaxomicin extended-pulse, vancomycin + bezlotoxumab (Zinplava, anti-toxin B mAb), OR fecal microbiota transplant (FMT) — 85-90% cure rate for recurrent CDI |
27 Microscopic Colitis Colorectal
Chronic, watery, non-bloody diarrhea with a grossly normal colonoscopy — diagnosis requires random colonic biopsies (especially right colon). Two subtypes: collagenous colitis (thickened subepithelial collagen band ≥ 10 mcm) and lymphocytic colitis (increased intraepithelial lymphocytes ≥ 20/100 surface epithelial cells). Affects predominantly women > 50 years. Strongly associated with medications: NSAIDs, PPIs, SSRIs, statins, and immune checkpoint inhibitors. Associated with celiac disease and autoimmune conditions.
Treatment: Discontinue offending medications. Budesonide 9 mg/day × 6-8 weeks then taper — first-line; induces remission in ~80%. Maintenance budesonide 3-6 mg daily often required (relapse rate ~60-80% after stopping). Refractory: cholestyramine, bismuth subsalicylate 9 tablets/day × 8 weeks, immunomodulators (azathioprine), or biologics (anti-TNFs, vedolizumab).
28 Irritable Bowel Syndrome (IBS) Colorectal
Functional GI disorder defined by recurrent abdominal pain associated with defecation or a change in bowel habits, in the absence of structural or biochemical abnormalities. Prevalence: 10-15% of the population. Rome IV criteria: recurrent abdominal pain on average ≥ 1 day/week in the last 3 months, associated with ≥ 2 of: related to defecation, associated with change in stool frequency, associated with change in stool form. Symptom onset ≥ 6 months before diagnosis. Subtypes: IBS-C (constipation-predominant), IBS-D (diarrhea-predominant), IBS-M (mixed), IBS-U (unsubtyped) — classified by Bristol Stool Form Scale.
Management: Dietary: Low-FODMAP diet (fermentable oligo-, di-, monosaccharides, and polyols) — effective in 50-80% for bloating/pain; implement with dietitian guidance (elimination then reintroduction). Fiber supplementation (soluble fiber: psyllium — beneficial; insoluble fiber like bran — may worsen bloating). Pharmacologic by subtype:
| IBS Subtype | Drug (Brand) | Mechanism | Key Details |
|---|---|---|---|
| IBS-C | Linaclotide (Linzess) | Guanylate cyclase-C agonist | 145-290 mcg daily before breakfast; also reduces visceral pain |
| Lubiprostone (Amitiza) | Chloride channel activator | 8 mcg BID with food; approved for women only | |
| Plecanatide (Trulance) | Guanylate cyclase-C agonist | 3 mg daily | |
| IBS-D | Eluxadoline (Viberzi) | Mixed opioid receptor modulator | 100 mg BID; contraindicated without gallbladder (pancreatitis risk) or with heavy alcohol use |
| Rifaximin (Xifaxan) | Non-absorbable antibiotic | 550 mg TID × 14 days; may repeat × 2 | |
| Alosetron (Lotronex) | 5-HT3 antagonist | 0.5-1 mg BID; restricted program (severe IBS-D in women); risk of ischemic colitis | |
| All subtypes | Antispasmodics (dicyclomine, hyoscyamine) | Anticholinergic | PRN for cramping; limited evidence |
| All (pain) | TCAs (amitriptyline 10-50 mg QHS) | Neuromodulator | Low-dose for visceral hypersensitivity; better for IBS-D (slows transit) |
29 Viral Hepatitis (A, B, C) Hepatology
Hepatitis A (HAV)
RNA picornavirus, fecal-oral transmission. Self-limited, does NOT cause chronic hepatitis. Incubation: 15-50 days. Diagnosis: anti-HAV IgM (acute infection), anti-HAV IgG (prior infection or vaccination/immunity). Treatment: supportive. Prevention: HAV vaccine (2-dose series — Havrix or Vaqta). Rarely causes fulminant hepatic failure (< 1%, higher risk in those with pre-existing liver disease).
Hepatitis B (HBV)
DNA hepadnavirus, transmitted by blood, sexual contact, vertical (perinatal). 95% of immunocompetent adults clear acute infection; < 5% develop chronic HBV (vs. 90% of neonates infected perinatally → chronic). Chronic HBV affects ~250 million people worldwide and is the leading cause of HCC globally.
HBV Serology Interpretation
| HBsAg | Anti-HBs | Anti-HBc (total) | Anti-HBc IgM | HBeAg | Interpretation |
|---|---|---|---|---|---|
| + | − | + | + | + | Acute infection |
| + | − | + | − | + | Chronic infection, high replicative (immune active) |
| + | − | + | − | − | Chronic infection, low replicative (HBeAg-negative; check HBV DNA) |
| − | + | + | − | − | Recovered (immune from natural infection) |
| − | + | − | − | − | Vaccinated (immune from vaccination) |
| − | − | + | − | − | Isolated anti-HBc (resolved infection with waning anti-HBs, occult HBV, or false positive) |
| − | − | − | − | − | Susceptible (not immune, not infected) |
Treatment indications (AASLD 2018): Immune-active phase (HBsAg+, elevated ALT > 2× ULN, HBV DNA > 2,000 IU/mL if HBeAg-negative or > 20,000 if HBeAg-positive). Also treat: cirrhosis (any DNA level), immunosuppression prophylaxis, HBV/HDV coinfection, pregnancy with high viral load (> 200,000 IU/mL, begin tenofovir at 28-32 weeks). First-line: entecavir (Baraclude) 0.5 mg daily or tenofovir disoproxil (Viread) 300 mg daily or tenofovir alafenamide (Vemlidy) 25 mg daily. Functional cure (HBsAg loss) occurs in < 5% with NUC therapy; pegylated interferon-alpha (48 weeks) achieves higher HBsAg loss rates but with significant side effects.
Hepatitis C (HCV)
RNA flavivirus, bloodborne (IVDU is the leading risk factor in the US, followed by blood transfusion before 1992, needlestick, tattoos, intranasal drug use, high-risk sexual contact). 75-85% develop chronic infection (unlike HBV). Chronic HCV → progressive fibrosis → cirrhosis (20-30% over 20-30 years) → HCC (1-4% annual risk once cirrhotic). Diagnosis: anti-HCV antibody (screening, positive in 1-2 months, remains positive for life even after cure) → confirm with HCV RNA (viral load, confirms active infection). Genotype testing guides treatment duration in some regimens (though pangenotypic regimens are now standard).
Treatment — Direct-Acting Antivirals (DAAs): All patients with chronic HCV should be treated. Pangenotypic regimens achieve > 95% SVR (sustained virologic response = cure, defined as undetectable HCV RNA 12 weeks post-treatment). Sofosbuvir/velpatasvir (Epclusa) 1 tablet daily × 12 weeks — preferred pangenotypic regimen. Glecaprevir/pibrentasvir (Mavyret) 3 tablets daily × 8 weeks (12 weeks if cirrhotic) — alternative pangenotypic regimen, shortest duration. Sofosbuvir/velpatasvir/voxilaprevir (Vosevi) — for DAA-experienced patients or retreatment. Universal screening: USPSTF recommends one-time HCV screening for all adults 18-79 years (USPSTF, JAMA 2020). SVR = cure but does NOT confer immunity — reinfection is possible. Patients with cirrhosis continue HCC surveillance even after SVR.
30 Cirrhosis & Portal Hypertension Hepatology
Cirrhosis is the end stage of progressive hepatic fibrosis — irreversible distortion of hepatic architecture with regenerative nodules replacing normal parenchyma. In the US, leading causes: alcohol-associated liver disease (~30%), NAFLD/NASH (~25%), chronic hepatitis C (~20%), chronic hepatitis B. Cirrhosis is classified as compensated (no complications; median survival > 12 years) or decompensated (ascites, variceal hemorrhage, hepatic encephalopathy, jaundice; median survival 2-4 years). The transition from compensated to decompensated is the pivotal event — once decompensation occurs, liver transplant evaluation should be initiated.
Child-Pugh Score
| Parameter | 1 Point | 2 Points | 3 Points |
|---|---|---|---|
| Total bilirubin (mg/dL) | < 2 | 2-3 | > 3 |
| Albumin (g/dL) | > 3.5 | 2.8-3.5 | < 2.8 |
| INR | < 1.7 | 1.7-2.3 | > 2.3 |
| Ascites | None | Mild (diuretic-responsive) | Moderate-severe (refractory) |
| Hepatic encephalopathy | None | Grade I-II | Grade III-IV |
Class A (5-6 points): well-compensated, 1-year survival ~100%. Class B (7-9): significant functional compromise, 1-year survival ~80%. Class C (10-15): decompensated, 1-year survival ~45%.
MELD Score
The Model for End-Stage Liver Disease (MELD) score uses bilirubin, INR, and creatinine to predict 90-day mortality — used for liver transplant allocation in the US. MELD 3.0 (updated 2022) adds sex and albumin for improved accuracy. MELD ≥ 15: refer for transplant evaluation. MELD ≥ 20-25: high transplant priority. The MELD-Na incorporates sodium (hyponatremia correlates with severity of portal hypertension and poor prognosis).
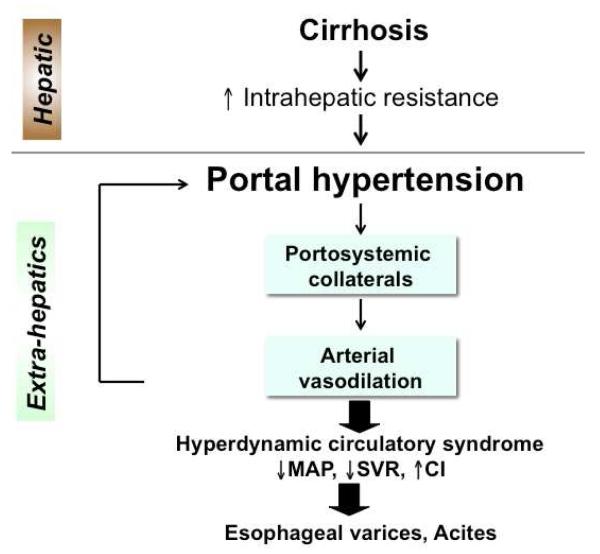
Portal Hypertension
Defined by hepatic venous pressure gradient (HVPG) ≥ 5 mmHg (normal 1-5). Clinically significant portal hypertension (CSPH): HVPG ≥ 10 mmHg — threshold for variceal formation and other complications. Portosystemic collaterals develop at sites connecting the portal and systemic venous systems: esophageal varices (left gastric vein → azygos), caput medusae (paraumbilical veins → epigastric), hemorrhoidal varices (superior rectal → middle/inferior rectal), splenorenal shunts. TIPS (transjugular intrahepatic portosystemic shunt) creates a direct communication between hepatic vein and portal vein branches, reducing portal pressure but increasing hepatic encephalopathy risk.
31 Ascites & Spontaneous Bacterial Peritonitis Hepatology
Ascites is the most common decompensating event in cirrhosis, developing in ~60% of compensated cirrhotics within 10 years. Pathogenesis: portal hypertension + splanchnic vasodilation → activation of RAAS and sympathetic nervous system → renal sodium and water retention + hypoalbuminemia → fluid transudation into the peritoneal cavity.
Diagnosis: Diagnostic paracentesis is mandatory for new-onset ascites and for all hospitalized patients with ascites (to exclude SBP). The serum-ascites albumin gradient (SAAG) is the single most useful test: SAAG ≥ 1.1 g/dL = portal hypertension (cirrhosis, CHF, Budd-Chiari) — 97% accuracy. SAAG < 1.1 = non-portal hypertensive (malignancy, TB, nephrotic, pancreatitis).
Management: Sodium restriction (2g/day), diuretics: spironolactone 100 mg + furosemide 40 mg daily (maintain the 100:40 ratio; target weight loss 0.5 kg/day without edema, 1 kg/day with edema). Maximum doses: spironolactone 400 mg, furosemide 160 mg. Refractory ascites (diuretic-intolerant or diuretic-resistant): serial large-volume paracentesis (LVP) with albumin replacement (6-8 g IV albumin per liter removed if > 5 L) or TIPS.
Spontaneous Bacterial Peritonitis (SBP)
Infection of ascitic fluid without an identifiable intra-abdominal source, occurring in 10-30% of hospitalized cirrhotics with ascites. Organisms: E. coli, Klebsiella, Streptococcus pneumoniae (monomicrobial gram-negatives most common). Presentation: may be subtle — fever, abdominal pain, altered mental status, worsening encephalopathy, renal dysfunction. Diagnosis: ascitic fluid PMN count ≥ 250 cells/mm3 — treat immediately, do not wait for culture. Treatment: ceftriaxone 2g IV daily × 5-7 days (or cefotaxime 2g IV q8h). IV albumin 1.5 g/kg on day 1, then 1 g/kg on day 3 reduces HRS and mortality (Sort et al., NEJM 1999). SBP prophylaxis: norfloxacin 400 mg daily (or ciprofloxacin or TMP-SMX) for: prior SBP (secondary prophylaxis — indefinite), ascitic fluid protein < 1.5 g/dL + Child-Pugh ≥ 9 or bilirubin ≥ 3 or Cr ≥ 1.2 (primary prophylaxis), and during GI bleeding.
32 Hepatic Encephalopathy Hepatology
Reversible neuropsychiatric dysfunction in patients with liver disease, caused primarily by failure to clear gut-derived neurotoxins — especially ammonia — from the portal circulation. Ammonia crosses the blood-brain barrier, is metabolized by astrocytes to glutamine (osmotically active) → astrocyte swelling → cerebral edema in severe cases. Precipitants (identify and correct): GI bleeding (protein load), infection/SBP, constipation, dehydration, electrolyte disturbances (hypokalemia, metabolic alkalosis), excessive diuresis, opioids/benzodiazepines, TIPS placement, dietary protein excess.
West Haven Grading
| Grade | Clinical Features |
|---|---|
| Covert (minimal) | No clinical signs; detected by psychometric testing (number connection test, Stroop test, critical flicker frequency) |
| Grade I | Trivial lack of awareness, euphoria/anxiety, shortened attention span, impaired addition/subtraction |
| Grade II | Lethargy, disorientation to time, obvious personality change, inappropriate behavior, asterixis |
| Grade III | Somnolence to semi-stupor, responsive to stimuli, confused, gross disorientation, bizarre behavior |
| Grade IV | Coma, unresponsive |
Treatment: Lactulose — osmotic laxative metabolized by colonic bacteria to lactic/acetic acid → acidifies colonic lumen → converts NH3 to non-absorbable NH4+ and promotes osmotic catharsis. Dose: 15-30 mL PO q1-2h until bowel movement, then titrate to 2-3 soft stools/day. For acute severe HE: lactulose enemas (300 mL in 700 mL water). Rifaximin (Xifaxan) 550 mg PO BID — added to lactulose for prevention of recurrent HE; reduces recurrence by 58% (Bass et al., NEJM 2010). Dietary protein: do NOT restrict protein (worsens sarcopenia and outcomes); target 1.2-1.5 g/kg/day, prefer vegetable and dairy protein. Zinc supplementation (zinc is a cofactor in the urea cycle).
33 Hepatocellular Carcinoma (HCC) Hepatology
The most common primary liver malignancy and the 3rd leading cause of cancer death worldwide. Arises almost exclusively in the setting of chronic liver disease/cirrhosis. Risk factors: cirrhosis (any etiology), chronic HBV (can cause HCC without cirrhosis), chronic HCV, alcohol, NAFLD/NASH (rising), aflatoxin B1 exposure. Surveillance: US abdomen ± AFP every 6 months in all cirrhotics and in non-cirrhotic HBV patients at risk (Asian males > 40, Asian females > 50, Africans > 20, family history of HCC).
BCLC (Barcelona Clinic Liver Cancer) Staging
| Stage | Definition | Treatment | Median Survival |
|---|---|---|---|
| 0 (Very early) | Single ≤ 2 cm, Child-Pugh A, PS 0 | Resection, ablation, or transplant | > 5 years |
| A (Early) | Single or up to 3 nodules ≤ 3 cm, Child-Pugh A-B, PS 0 | Resection, ablation, or transplant (Milan criteria: single ≤ 5 cm OR up to 3 ≤ 3 cm each, no vascular invasion, no extrahepatic disease) | > 5 years |
| B (Intermediate) | Multinodular, Child-Pugh A-B, PS 0 | TACE (transarterial chemoembolization) | ~20 months |
| C (Advanced) | Portal invasion, N1, M1, Child-Pugh A-B, PS 1-2 | Systemic therapy: atezolizumab + bevacizumab (IMbrave150, first-line); sorafenib or lenvatinib; second-line: regorafenib, cabozantinib, ramucirumab (if AFP ≥ 400) | ~12 months |
| D (Terminal) | Child-Pugh C (not transplant candidates), PS 3-4 | Best supportive care | < 3 months |
34 Autoimmune Hepatitis Hepatology
Chronic inflammatory liver disease caused by loss of immune tolerance to hepatocytes. Predominantly affects women (4:1). Bimodal age distribution: 10-20 and 45-70 years. Two types: Type 1 (80% — ANA and/or anti-smooth muscle antibody [ASMA] positive; any age) and Type 2 (anti-liver kidney microsomal antibody [anti-LKM-1] positive; primarily children/adolescents). Presentation ranges from asymptomatic transaminase elevation to acute hepatitis mimicking viral hepatitis to fulminant liver failure. Hallmark labs: markedly elevated transaminases (ALT/AST often > 10× ULN), elevated IgG, positive autoantibodies. Biopsy: interface hepatitis (lymphoplasmacytic infiltrate at the portal-parenchymal interface), emperipolesis, hepatic rosettes.
Treatment: Prednisone 40-60 mg/day (or budesonide 9 mg/day in non-cirrhotics) + azathioprine 50-150 mg/day (combination allows lower steroid doses). Prednisone tapered over weeks to months. Azathioprine maintained for at least 2-3 years. Treatment response: normalization of transaminases and IgG. Relapse rate after withdrawal: 50-80%, so many patients require lifelong maintenance. Second-line for refractory: mycophenolate mofetil, tacrolimus, 6-mercaptopurine. Liver transplant for decompensated disease refractory to immunosuppression.
35 NAFLD & NASH (MASLD / MASH) Hepatology
Non-alcoholic fatty liver disease is the most common chronic liver disease worldwide, affecting ~25% of the global population. Recently renamed: NAFLD → metabolic dysfunction-associated steatotic liver disease (MASLD); NASH → metabolic dysfunction-associated steatohepatitis (MASH). Spectrum: simple steatosis (≥ 5% hepatocytes with fat) → MASH (steatosis + inflammation + hepatocyte ballooning ± fibrosis) → cirrhosis → HCC. Only 10-20% of MASLD patients develop MASH, but MASH is now the fastest-growing indication for liver transplant and cause of HCC in the US.
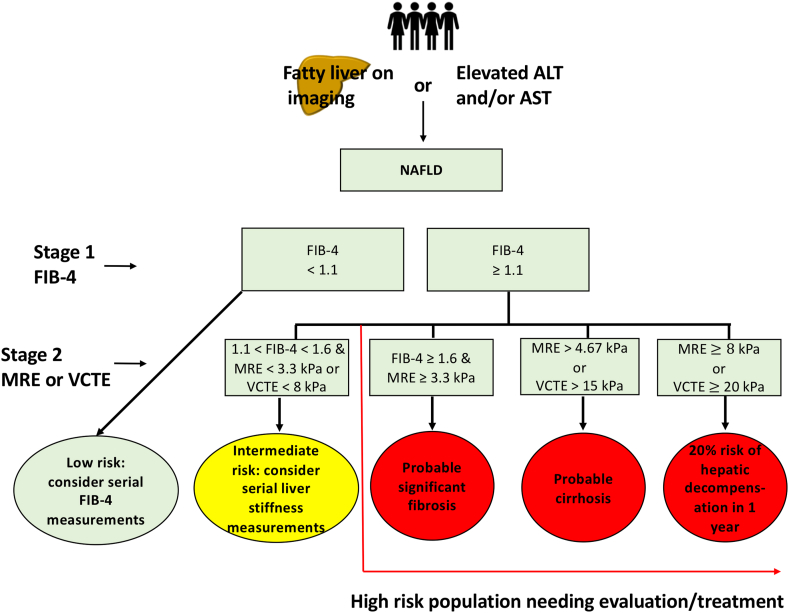
Diagnosis: Imaging (US, CT, or MRI showing hepatic steatosis) + exclusion of significant alcohol use (< 20g/day women, < 30g/day men) + at least one cardiometabolic risk factor. Fibrosis staging is the key prognostic determinant — non-invasive tools: FIB-4 index (age, AST, ALT, platelets), NAFLD Fibrosis Score (NFS), vibration-controlled transient elastography (FibroScan — liver stiffness < 8 kPa = low risk, > 12 kPa = advanced fibrosis). Liver biopsy remains the gold standard: NAS (NAFLD Activity Score) ≥ 5 with ballooning is diagnostic of MASH.
Treatment: Weight loss is the most effective therapy — 5% weight loss improves steatosis, 7-10% improves MASH and fibrosis. Resmetirom (Rezdiffra) — thyroid hormone receptor beta agonist, FDA-approved 2024, first drug specifically for MASH with moderate-to-advanced fibrosis (F2-F3). Pioglitazone — improves MASH histology even in non-diabetics (but weight gain, fluid retention, fracture risk). Vitamin E (800 IU/day) — modest benefit for MASH in non-diabetics without cirrhosis (PIVENS trial). GLP-1 receptor agonists (semaglutide) — improving steatosis and MASH, with phase 3 trials showing MASH resolution; major ancillary benefit of weight loss. Bariatric surgery: MASH resolution in > 80% of morbidly obese patients. Statins are safe in MASLD/MASH and should not be withheld.
36 Wilson's Disease & Hemochromatosis Hepatology
Wilson's Disease
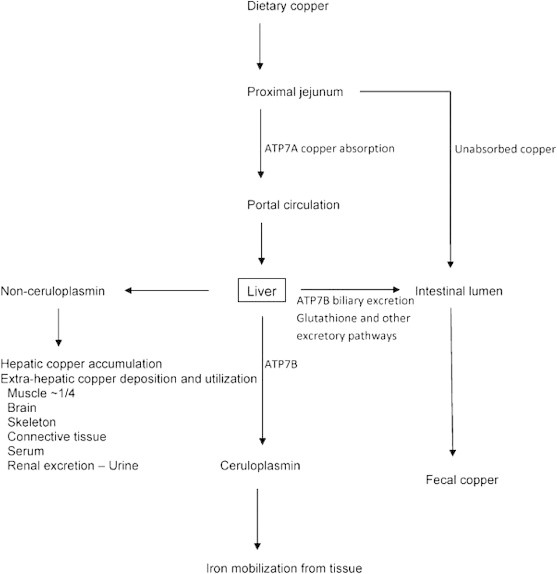
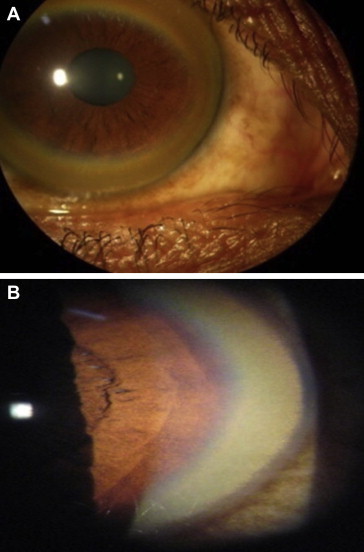
Autosomal recessive disorder of copper metabolism (ATP7B gene mutation) causing copper accumulation in the liver, brain, cornea, and kidneys. Prevalence: ~1/30,000. Presents between ages 5-40. Hepatic: fatty liver, chronic hepatitis, cirrhosis, fulminant hepatic failure (with Coombs-negative hemolytic anemia — classic!). Neuropsychiatric: tremor, dysarthria, dystonia, parkinsonism, psychiatric symptoms. Kayser-Fleischer rings (copper deposits in Descemet's membrane of the cornea — pathognomonic, present in 95% with neurologic disease, ~50% with hepatic-only disease; slit-lamp exam). Diagnosis: low serum ceruloplasmin (< 20 mg/dL), elevated 24-hour urine copper (> 40 mcg), elevated hepatic copper on biopsy (> 250 mcg/g dry weight), KF rings.
Treatment: D-penicillamine (copper chelator, 1-1.5 g/day) or trientine (better tolerated, 750-1500 mg/day) for active disease; zinc acetate 150 mg/day (blocks intestinal copper absorption) for maintenance and presymptomatic patients. Liver transplant for decompensated cirrhosis or fulminant failure — curative (transplanted liver has normal ATP7B).
Hereditary Hemochromatosis
Autosomal recessive iron overload disorder, most commonly from homozygous C282Y mutation in the HFE gene. Prevalence: 1/200-300 people of Northern European descent. Pathogenesis: defective hepcidin regulation → excessive intestinal iron absorption → iron deposition in liver (cirrhosis, HCC), pancreas (diabetes — "bronze diabetes"), heart (cardiomyopathy, arrhythmia), joints (arthropathy — 2nd/3rd MCP joints, chondrocalcinosis), skin (hyperpigmentation), pituitary (hypogonadism). Diagnosis: elevated transferrin saturation (> 45% — most sensitive screening test), elevated ferritin (> 200 women, > 300 men — but ferritin is an acute-phase reactant and may be elevated from inflammation). Confirm with HFE genotyping. MRI can quantify hepatic iron (R2* / FerriScan). Liver biopsy: indicated if ferritin > 1,000 or elevated LFTs to assess fibrosis (hepatic iron index > 1.9 is diagnostic). Treatment: phlebotomy — 500 mL (removes ~250 mg iron) weekly until ferritin 50-100 ng/mL, then maintenance q2-4 months. If phlebotomy-intolerant: deferasirox (oral chelator). Screen first-degree relatives.
37 Cholelithiasis & Choledocholithiasis Biliary
Cholelithiasis (gallstones): present in 10-15% of the US population. Types: cholesterol stones (~80%, from bile supersaturated with cholesterol, gallbladder hypomotility, and crystal nucleation) and pigment stones (black pigment — hemolysis, cirrhosis; brown pigment — biliary stasis/infection, common in Asian populations). Risk factors (the "4 F's" is a simplification): female sex, age > 40, obesity, rapid weight loss, TPN, pregnancy, fibrates, octreotide, Native American and Hispanic ethnicity. 80% of gallstones are asymptomatic — prophylactic cholecystectomy generally not indicated (exceptions: porcelain gallbladder, stones > 3 cm, gallbladder polyps > 1 cm).
Biliary colic: episodic, steady RUQ/epigastric pain lasting 30 min to several hours (NOT truly "colicky"), often postprandial (fatty meals trigger CCK → gallbladder contraction against stone in cystic duct). Diagnosis: RUQ ultrasound (sensitivity 95% for gallstones). Treatment: elective laparoscopic cholecystectomy.
Choledocholithiasis (CBD stone): 10-15% of patients with gallstones have concurrent CBD stones. Presentation: biliary colic + obstructive jaundice (conjugated hyperbilirubinemia, elevated ALP/GGT, dilated CBD > 6 mm on US). Risk stratification guides management: high probability (CBD stone on imaging, cholangitis, bilirubin > 4) → ERCP directly; intermediate probability → MRCP or EUS to confirm before ERCP; low probability → proceed with cholecystectomy ± intraoperative cholangiogram.
38 Cholangitis (Tokyo Guidelines) Biliary
Acute cholangitis is a bacterial infection of the biliary system caused by biliary obstruction (most commonly choledocholithiasis, but also strictures, tumors, stents). Classic Charcot's triad: fever/chills + jaundice + RUQ pain (present in ~50-70%). Reynolds' pentad adds hypotension + altered mental status — indicates suppurative cholangitis with sepsis.
Tokyo Guidelines (TG18) Severity Grading
| Grade | Severity | Criteria | Management |
|---|---|---|---|
| Grade I | Mild | Does not meet Grade II or III criteria; responds to initial medical treatment (antibiotics + IV fluids) | Antibiotics; early biliary drainage (ERCP within 24-48 hours) |
| Grade II | Moderate | Any 2 of: WBC > 12k or < 4k, fever ≥ 39°C, age ≥ 75, bilirubin ≥ 5 mg/dL, albumin < 0.7 × LLN | Antibiotics + urgent biliary drainage (ERCP within 24 hours) |
| Grade III | Severe | Organ dysfunction: cardiovascular (hypotension requiring vasopressors), neurologic (altered consciousness), respiratory (PaO2/FiO2 < 300), renal (Cr > 2.0), hepatic (INR > 1.5), hematologic (platelets < 100k) | Antibiotics + emergent biliary drainage (ERCP or percutaneous transhepatic biliary drainage within hours); ICU admission |
Antibiotics: Cover gram-negatives and anaerobes — piperacillin-tazobactam, ciprofloxacin + metronidazole, or meropenem (for severe/healthcare-associated). Biliary drainage: ERCP with sphincterotomy and stone extraction is the gold standard. If ERCP fails or is unavailable: percutaneous transhepatic biliary drainage (PTBD) or EUS-guided biliary drainage.
39 Primary Sclerosing Cholangitis & Primary Biliary Cholangitis Biliary
Primary Sclerosing Cholangitis (PSC)
Chronic, progressive disease characterized by inflammation and fibrosis of intrahepatic and extrahepatic bile ducts leading to multifocal strictures, cholestasis, and eventual biliary cirrhosis. Strong association with IBD — 70-80% of PSC patients have UC (but only 5% of UC patients develop PSC). More common in men (2:1), age 30-50. Diagnosis: cholestatic biochemistry (elevated ALP, often markedly; mildly elevated bilirubin) + MRCP showing "beaded" pattern of multifocal strictures and dilations + exclusion of secondary causes. Liver biopsy: periductal "onion-skin" fibrosis (classic but not always seen). No autoantibody is diagnostic (p-ANCA positive in ~80% but not specific). No medical therapy has been proven to alter disease progression. Ursodeoxycholic acid (UDCA) improves biochemistry but has not shown survival benefit in randomized trials — high-dose UDCA (> 28 mg/kg/day) may be harmful. Annual cancer surveillance: MRCP + CA 19-9 for cholangiocarcinoma (10-15% lifetime risk), colonoscopy every 1-2 years for CRC (PSC-IBD has the highest CRC risk of any IBD subtype). Liver transplant is the only definitive treatment — excellent outcomes but PSC can recur in the graft.
Primary Biliary Cholangitis (PBC)
Autoimmune destruction of small intrahepatic bile ducts. Predominantly affects women (9:1), age 40-60. Diagnosis: cholestatic biochemistry (elevated ALP) + anti-mitochondrial antibody (AMA) positive (95% sensitivity and specificity) — if AMA+, liver biopsy is not required for diagnosis. Presentation: fatigue (most common and most debilitating), pruritus (often severe), jaundice (late), hyperlipidemia (elevated HDL), xanthomas/xanthelasma. Associated with other autoimmune conditions (Sjogren's, thyroiditis, celiac, scleroderma). Treatment: UDCA 13-15 mg/kg/day — first-line; improves survival and delays transplant need; response assessed at 1 year by ALP, bilirubin, and biochemical response criteria (Paris, Barcelona). Obeticholic acid (Ocaliva) — FXR agonist, add-on for inadequate UDCA response; risk of pruritus, contraindicated in decompensated cirrhosis. Elafibranor (Iqirvo) — PPAR alpha/delta agonist, FDA-approved 2024, alternative add-on. Liver transplant for decompensated disease; PBC can recur in the graft (30% at 10 years).
40 Acute Pancreatitis Pancreatic
Inflammation of the pancreas caused by premature intracellular activation of digestive enzymes. Diagnosis requires 2 of 3 criteria: (1) characteristic abdominal pain (acute onset, severe, epigastric, radiating to the back, worse supine, better leaning forward), (2) serum lipase or amylase ≥ 3× ULN, (3) imaging findings. Lipase is more sensitive and specific than amylase. Etiology: gallstones (~40%) and alcohol (~30%) account for ~70%. Other: hypertriglyceridemia (> 1,000 mg/dL), post-ERCP (5-10%), drugs (azathioprine, valproic acid, didanosine, GLP-1 agonists — rare), autoimmune, trauma, pancreas divisum, ampullary tumors, scorpion stings.
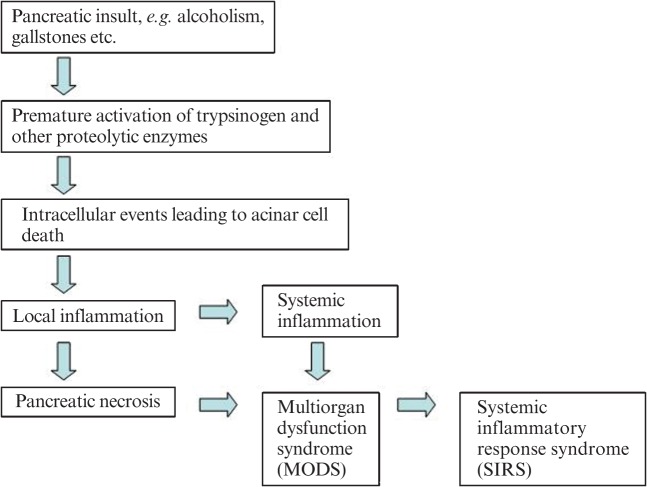
Ranson's Criteria
| At Admission | At 48 Hours |
|---|---|
| Age > 55 | Hematocrit drop > 10% |
| WBC > 16,000 | BUN rise > 5 mg/dL |
| Glucose > 200 mg/dL | Calcium < 8 mg/dL |
| AST > 250 IU/L | PaO2 < 60 mmHg |
| LDH > 350 IU/L | Base deficit > 4 mEq/L |
| Fluid sequestration > 6 L |
≥ 3 criteria = severe; ≥ 6 criteria = ~40% mortality. GA LAW (admission) and C HOBBS (48h).
BISAP Score (Bedside Index of Severity in Acute Pancreatitis)
| Criterion | Points |
|---|---|
| BUN > 25 mg/dL | 1 |
| Impaired mental status (GCS < 15) | 1 |
| SIRS (systemic inflammatory response syndrome) | 1 |
| Age > 60 | 1 |
| Pleural effusion on imaging | 1 |
BISAP ≥ 3: 5-10× increased mortality risk. Advantage: can be calculated within 24 hours of admission.
Revised Atlanta Classification (2012)
| Severity | Definition | Mortality |
|---|---|---|
| Mild | No organ failure, no local/systemic complications | < 1% |
| Moderately severe | Transient organ failure (< 48 hours) OR local complications (peripancreatic collections, necrosis) without persistent organ failure | ~5% |
| Severe | Persistent organ failure (> 48 hours) — single or multi-organ (modified Marshall score) | 20-40% |
Management: Aggressive IV fluid resuscitation (goal-directed, lactated Ringer's preferred — 1.5 mL/kg/hr initially, titrate to clinical targets), pain control (multimodal — NSAIDs, acetaminophen, opioids if needed), NPO → early oral feeding within 24 hours if tolerated (no need to wait for pain resolution or enzyme normalization — early feeding reduces complications). No prophylactic antibiotics — even in necrotizing pancreatitis. Gallstone pancreatitis: cholecystectomy during the same admission (after clinical improvement) — delays increase recurrence risk. ERCP within 24 hours only if concurrent cholangitis or persistent CBD obstruction. Necrotizing pancreatitis with infected necrosis (suspect if clinical deterioration after 7-10 days, gas in collection on CT): antibiotics (meropenem or imipenem preferred for pancreatic penetration) + drainage; prefer step-up approach (percutaneous/endoscopic drainage → video-assisted retroperitoneal debridement or endoscopic necrosectomy → open necrosectomy only as last resort).
41 Chronic Pancreatitis Pancreatic
Progressive inflammatory destruction of the pancreas leading to irreversible structural damage and loss of exocrine (maldigestion) and endocrine (diabetes) function. Causes: alcohol (~45% in Western countries, requires > 5 years of heavy use), idiopathic (~30%), genetic (PRSS1, SPINK1, CFTR mutations), autoimmune (IgG4-related — type 1; idiopathic duct-centric — type 2), obstructive (pancreas divisum, tumors), recurrent acute pancreatitis. The TIGAR-O classification organizes etiologies: Toxic-metabolic, Idiopathic, Genetic, Autoimmune, Recurrent/severe acute, Obstructive.
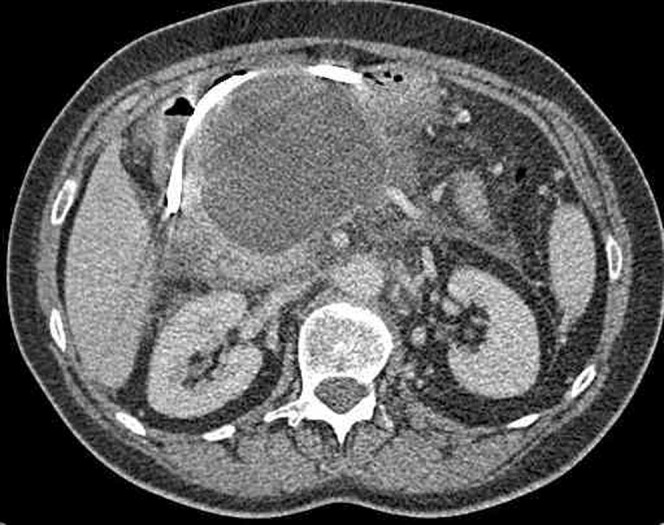
Presentation: chronic or recurrent epigastric pain (often debilitating, the primary cause of morbidity), steatorrhea (not until > 90% of exocrine function is lost — "too little too late" clinical threshold), weight loss, diabetes mellitus (type 3c — pancreatogenic), pancreatic calcifications on imaging (pathognomonic). Diagnosis: CT/MRI (calcifications, duct dilation, atrophy); MRCP with secretin stimulation for early disease; EUS (most sensitive for early changes — Rosemont criteria); fecal elastase-1 (< 200 mcg/g = exocrine insufficiency).
Treatment: Pain management: abstinence from alcohol and tobacco, analgesics (start with non-opioid, escalate per WHO ladder), pancreatic enzyme replacement (may reduce pain by negative feedback on CCK-mediated pancreatic secretion — evidence is mixed), antioxidants. Endoscopic therapy: ERCP with pancreatic duct stenting and/or stone extraction for dominant strictures/stones; ESWL (extracorporeal shockwave lithotripsy) for large pancreatic duct stones. Surgical: lateral pancreaticojejunostomy (Puestow/Frey procedure) for dilated duct (> 7 mm); total pancreatectomy with islet autotransplantation (TPIAT) for refractory pain with preserved islet function. Pancreatic enzyme replacement therapy (PERT): lipase 40,000-50,000 USP units per meal, 25,000 per snack (adjust based on response); take with meals, not before; concurrent PPI may improve efficacy by preventing acid degradation of enzymes in duodenum.
42 Pancreatic Cancer Pancreatic
Pancreatic ductal adenocarcinoma (PDAC) is the 4th leading cause of cancer death in the US with a 5-year survival of ~12%. Risk factors: smoking (2× risk), chronic pancreatitis, new-onset diabetes after age 50, obesity, family history (5-10% have familial predisposition — BRCA2, PALB2, CDKN2A, ATM, Lynch syndrome), hereditary pancreatitis (PRSS1 — 40% lifetime risk). 60-70% arise in the pancreatic head (causing painless obstructive jaundice — Courvoisier's sign: palpable, non-tender gallbladder); 20-30% in body/tail (usually present late with pain and weight loss).
Diagnosis & Staging: Pancreas-protocol CT (triple-phase with thin cuts) — sensitivity ~90% for pancreatic masses, assesses vascular involvement (SMA, celiac axis, SMV/portal vein) for resectability. EUS + FNA for tissue diagnosis (most sensitive for small lesions). CA 19-9: elevated in ~80% but not specific (elevated in any biliary obstruction; also falsely normal in Lewis-negative individuals ~10%). Staging: resectable (no arterial contact, < 180° venous contact without contour irregularity), borderline resectable (limited arterial contact ≤ 180°, venous contact > 180° but reconstructable), locally advanced (unreconstructable arterial/venous involvement), metastatic.
Treatment: Resectable: upfront surgery (pancreaticoduodenectomy / Whipple for head lesions, distal pancreatectomy + splenectomy for body/tail) followed by adjuvant modified FOLFIRINOX × 6 months (Conroy et al., NEJM 2018) OR gemcitabine/capecitabine. Borderline resectable/locally advanced: neoadjuvant chemotherapy (FOLFIRINOX or gemcitabine/nab-paclitaxel) ± radiation → reassess resectability. Metastatic: palliative chemotherapy — FOLFIRINOX (best survival, ~11 months, for good PS) or gemcitabine/nab-paclitaxel (~8.5 months); BRCA/PALB2-mutated: platinum-based chemo + olaparib maintenance (POLO trial). Biliary stenting for jaundice palliation; celiac plexus neurolysis for pain.
43 Pancreatic Cysts & IPMN Pancreatic
Pancreatic cysts are incidentally detected in 2-20% of abdominal imaging studies. Most are benign, but some harbor malignant potential. Key types:
| Cyst Type | Demographic | Location | Fluid Analysis | Malignant Potential | Management |
|---|---|---|---|---|---|
| Serous cystadenoma (SCA) | Women > 60 | Any | Low CEA, low amylase | Essentially zero | Observation unless symptomatic |
| Mucinous cystic neoplasm (MCN) | Women 40-60 | Body/tail (does not communicate with duct) | Elevated CEA (> 192), low amylase | 15-30% | Surgical resection recommended |
| IPMN — Main duct (MD) | Men = Women, 60-70 | Any (main pancreatic duct ≥ 5 mm) | Elevated CEA, elevated amylase | 40-70% | Surgical resection |
| IPMN — Branch duct (BD) | Men = Women, 60-70 | Any (communicates with duct) | Elevated CEA, elevated amylase | 15-25% | Surveillance vs surgery based on high-risk features |
| Solid pseudopapillary neoplasm | Young women (20-30) | Any | Variable | Low-grade malignant | Surgical resection |
| Pseudocyst | History of pancreatitis | Any | Low CEA, high amylase | None | Drainage if symptomatic (> 6 cm or persistent > 6 weeks) |
IPMN surveillance (AGA/ACG guidelines): Branch-duct IPMN without high-risk stigmata: MRI/MRCP-based surveillance. High-risk stigmata (absolute indications for surgery): obstructive jaundice, enhancing solid component ≥ 5 mm, main duct ≥ 10 mm. Worrisome features: cyst ≥ 3 cm, enhancing mural nodule < 5 mm, thickened/enhancing walls, main duct 5-9 mm, lymphadenopathy, elevated CA 19-9 → EUS for further evaluation.
44 Upper GI Bleeding GI Bleeding
Upper GI bleeding (UGIB) originates proximal to the ligament of Treitz. Annual incidence: ~100/100,000. Causes: peptic ulcer disease (~35-50%), variceal bleeding (~10-20%), Mallory-Weiss tear (~5-10%), erosive esophagitis/gastritis, Dieulafoy lesion (aberrant submucosal artery that erodes through normal mucosa — massive, intermittent hemorrhage), malignancy, angiodysplasia (AVM — more common in elderly with aortic stenosis = Heyde syndrome), aortoenteric fistula (graft-to-duodenum — must consider in any patient with aortic graft and GI bleed).
Glasgow-Blatchford Score (GBS)
| Parameter | Value | Score |
|---|---|---|
| BUN (mg/dL) | 18.2–22.3 | 2 |
| 22.4–27.9 | 3 | |
| 28.0–69.9 | 4 | |
| ≥ 70 | 6 | |
| Hemoglobin — Men (g/dL) | 12.0–12.9 | 1 |
| 10.0–11.9 | 3 | |
| < 10 | 6 | |
| Hemoglobin — Women | 10.0–11.9 | 1 |
| < 10 | 6 | |
| Systolic BP (mmHg) | 100–109 / 90–99 / < 90 | 1 / 2 / 3 |
| Heart rate ≥ 100 | 1 | |
| Melena | 1 | |
| Syncope | 2 | |
| Hepatic disease | 2 | |
| Heart failure | 2 |
GBS 0-1: low risk, may be considered for outpatient management. GBS ≥ 7: high risk, need ICU monitoring.
Forrest Classification (Endoscopic Ulcer Appearance)
| Forrest Class | Description | Rebleed Risk (no therapy) | Endoscopic Therapy |
|---|---|---|---|
| Ia | Spurting hemorrhage | ~90% | Required |
| Ib | Oozing hemorrhage | ~50% | Required |
| IIa | Non-bleeding visible vessel (NBVV) | ~50% | Required |
| IIb | Adherent clot | ~30% | Consider (remove clot, treat underlying lesion) |
| IIc | Flat pigmented spot | ~10% | Not required |
| III | Clean-base ulcer | ~5% | Not required |
Management: Resuscitation (crystalloid, pRBC if Hb < 7 or hemodynamically unstable — restrictive transfusion strategy is superior to liberal). IV PPI bolus (80 mg) then infusion (8 mg/hr) × 72 hours AFTER endoscopic therapy (not beneficial before EGD). EGD within 24 hours (within 12 hours if high-risk features). Endoscopic hemostasis for high-risk lesions (Forrest Ia-IIb): combination therapy preferred (injection + thermal or mechanical). Post-hemostasis: high-dose PPI × 72 hours, then oral PPI. Rebleeding (5-10%): repeat EGD; if fails → interventional radiology (angiographic embolization) or surgery.
45 Lower GI Bleeding GI Bleeding
Lower GI bleeding (LGIB) originates distal to the ligament of Treitz. Causes by age: Young adults — hemorrhoids, anal fissures, IBD, Meckel's diverticulum. Middle-aged/elderly — diverticular bleeding (#1 cause of massive LGIB in > 60), angiodysplasia (#2), ischemic colitis, colorectal cancer/polyps, post-polypectomy bleeding, radiation proctitis. Most LGIB (> 80%) stops spontaneously.
Management: Hemodynamic resuscitation, rectal exam, rule out upper source if hemodynamically significant (NGT lavage or EGD if concern). Colonoscopy after rapid bowel prep (4-6 L PEG within 4-6 hours) is both diagnostic and therapeutic — within 24 hours for most patients. If colonoscopy non-diagnostic and bleeding is ongoing: CT angiography (detects bleeding at rates ≥ 0.3-0.5 mL/min) → angiographic embolization if positive. Tc-99m tagged red blood cell scan (detects bleeding ≥ 0.1-0.4 mL/min) — more sensitive than CTA but less precise in localizing the source. Surgery (segmental or subtotal colectomy) reserved for massive, refractory hemorrhage.
46 Obscure & Occult GI Bleeding GI Bleeding
Occult GI bleeding: positive fecal occult blood test (FOBT/FIT) or iron deficiency anemia without visible blood loss. Obscure GI bleeding: bleeding of unknown origin that persists or recurs after negative EGD and colonoscopy (now termed "suspected small bowel bleeding"). Causes: angiodysplasia (most common in > 40 years), small bowel tumors (GIST, lymphoma, adenocarcinoma, NETs), Crohn's disease, Meckel's diverticulum (most common cause in < 40 years — contains ectopic gastric mucosa, diagnosed by Tc-99m pertechnetate scan), Dieulafoy lesion, NSAID enteropathy, portal hypertensive enteropathy.
Evaluation: Repeat EGD/colonoscopy (missed lesions found in 10-20%). Video capsule endoscopy (VCE) — first-line for suspected small bowel bleeding; diagnostic yield 40-60%; swallowed capsule with camera transmits images wirelessly. Contraindicated in known or suspected strictures (capsule retention risk — use patency capsule first in Crohn's). If VCE positive: deep enteroscopy (double-balloon, single-balloon, or spiral enteroscopy) for biopsy and therapy. If VCE negative: repeat during active bleeding, CT enterography, angiography during active bleeding, intraoperative enteroscopy (rare), or Meckel's scan in younger patients.
47 Variceal Bleeding Management GI Bleeding
Acute variceal hemorrhage is a life-threatening emergency with 15-20% mortality per episode. The approach combines resuscitation, pharmacologic therapy, and endoscopic intervention.
Acute Management Protocol:
1. Resuscitation: Restrictive transfusion (target Hb 7-8 g/dL — over-transfusion worsens portal pressure and rebleeding). Airway protection — early intubation if massive hematemesis or altered mental status. Correct coagulopathy only if needed for procedures (platelets > 50k, fibrinogen > 100).
2. Vasoactive drugs (start immediately, before EGD): Octreotide 50 mcg IV bolus then 50 mcg/hr infusion × 3-5 days (reduces splanchnic blood flow). OR terlipressin (vasopressin analog) where available.
3. Antibiotics: Ceftriaxone 1g IV daily × 7 days — reduces bacterial infections AND rebleeding AND mortality in cirrhotics with GI bleeding (Fernandez et al., Hepatology 2006).
4. Endoscopy (within 12 hours): Endoscopic variceal ligation (EVL/banding) — first-line; rubber bands placed on each varix. Repeat every 2-4 weeks until eradication. If banding fails or is not feasible: balloon tamponade (Sengstaken-Blakemore or Minnesota tube) as a temporary bridge (< 24 hours; high complication rate — esophageal rupture, aspiration).
5. Salvage for refractory bleeding: TIPS (transjugular intrahepatic portosystemic shunt) — highly effective at controlling refractory variceal bleeding; also consider "early TIPS" (preemptive within 72 hours) for high-risk patients (Child-Pugh C 10-13 or Child-Pugh B with active bleeding at EGD) — improves survival (Garcia-Pagan et al., NEJM 2010).
6. Secondary prophylaxis: NSBB + EVL (combination is superior to either alone); repeat EVL every 2-4 weeks until eradication, then surveillance EGD every 6-12 months.
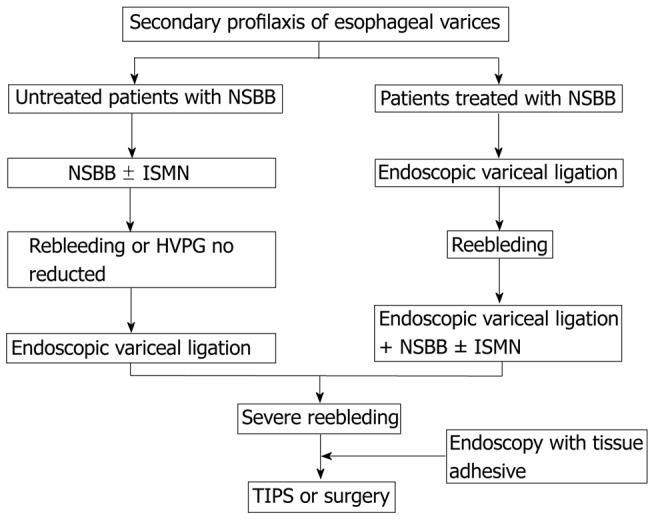
48 Ischemic Colitis Special
The most common form of intestinal ischemia. Caused by transient, non-occlusive hypoperfusion of the colon (unlike acute mesenteric ischemia, which involves arterial occlusion of the SMA). The splenic flexure (watershed between SMA and IMA) and the rectosigmoid junction (watershed between IMA and internal iliac) are the most vulnerable segments. Risk factors: age > 60, atherosclerosis, hypotension/shock, cardiac surgery (especially post-AAA repair — IMA ligation), cocaine, vasoconstrictors, hypercoagulable states, vasculitis. The rectum is typically spared (dual blood supply from IMA and internal iliac arteries) — rectal involvement suggests a different diagnosis (IBD, radiation proctitis).
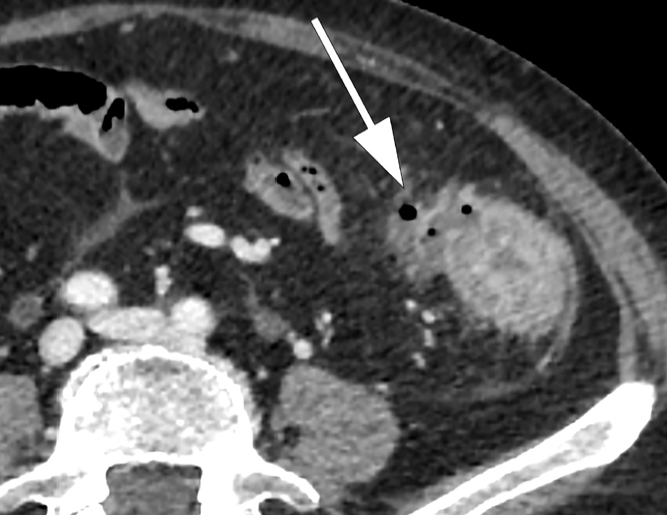
Presentation: acute onset of crampy LLQ pain, urgency, and bloody diarrhea. CT: colonic wall thickening with thumbprinting (mucosal edema), pericolonic fat stranding, in a segmental, watershed distribution. Colonoscopy (careful, low-pressure insufflation): edematous, friable, hemorrhagic mucosa with a sharp transition to normal bowel — single stripe sign (longitudinal ulcer along the antimesenteric border). Treatment: supportive (IV fluids, bowel rest, antibiotics for moderate-severe — to cover bacterial translocation). 85% resolve spontaneously within 1-2 weeks. Surgical indications: peritonitis, perforation, fulminant colitis, stricture with obstruction, gangrene. 10-20% develop chronic stricture requiring resection.
49 Hereditary GI Cancer Syndromes Special
Lynch Syndrome (Hereditary Nonpolyposis Colorectal Cancer / HNPCC)
Autosomal dominant, caused by germline mutations in DNA mismatch repair (MMR) genes: MLH1, MSH2, MSH6, PMS2, or EPCAM (EPCAM deletions silence MSH2). Most common hereditary CRC syndrome (~3% of all CRC). Tumors exhibit microsatellite instability (MSI-high) and loss of MMR protein expression on immunohistochemistry. CRC risk: 40-80% lifetime (mean age ~45, right-sided predominant). Also increased risk of endometrial cancer (40-60%), ovarian, gastric, urinary tract, small bowel, pancreatic, and sebaceous skin tumors. Screening: universal tumor testing (IHC for MMR proteins ± MSI testing) on all CRC and endometrial cancers. Surveillance for Lynch carriers: colonoscopy every 1-2 years starting at age 20-25 (or 2-5 years before youngest family diagnosis). Aspirin 600 mg daily reduces CRC incidence in Lynch (CAPP2 trial). Consider: prophylactic hysterectomy + BSO after childbearing for women.
Familial Adenomatous Polyposis (FAP)
Autosomal dominant mutation in APC (adenomatous polyposis coli) tumor suppressor gene. Hundreds to thousands of colorectal adenomatous polyps develop by teenage years. 100% CRC risk by age 40-50 if untreated. Attenuated FAP (AFAP): 10-100 adenomas, later onset, 70% CRC risk. Associated features: duodenal adenomas (4-12% progress to cancer — must surveil), desmoid tumors (most common cause of death in prophylactically colectomized FAP patients), fundic gland polyps, congenital hypertrophy of retinal pigment epithelium (CHRPE), osteomas, epidermoid cysts (Gardner syndrome = FAP + extracolonic manifestations). Surveillance: annual sigmoidoscopy/colonoscopy starting at age 10-12. Treatment: prophylactic colectomy (total proctocolectomy with IPAA or total colectomy with IRA) when polyp burden becomes unmanageable — typically late teens to early 20s.
Peutz-Jeghers Syndrome (PJS)
Autosomal dominant, STK11/LKB1 mutation. Characterized by mucocutaneous melanotic pigmentation (lips, buccal mucosa, fingers — characteristically fade after puberty except on buccal mucosa) and hamartomatous polyps throughout the GI tract (especially small bowel). Despite being hamartomas, there is a significantly elevated cancer risk: 39% GI cancers (colon, stomach, small bowel, pancreas), 32-54% breast cancer, and increased risk of ovarian, cervical, testicular (Sertoli cell), and lung cancers. Lifetime cancer risk: ~85%. Complications: intussusception from polyps (especially in children). Surveillance: start at age 8 with upper and lower endoscopy + capsule/MR enterography every 2-3 years; breast MRI annually from age 25.
50 GI Manifestations of Systemic Disease Special
| Systemic Disease | GI Manifestations | Key Details |
|---|---|---|
| Scleroderma (systemic sclerosis) | Esophageal dysmotility (aperistalsis of distal 2/3, patulous LES → severe GERD), watermelon stomach (GAVE), colonic pseudo-obstruction | Esophagus is the most commonly involved GI organ in scleroderma |
| Amyloidosis | GI dysmotility, malabsorption, GI bleeding, macroglossia (AL amyloidosis), hepatomegaly | Congo red staining → apple-green birefringence under polarized light |
| Diabetes mellitus | Gastroparesis, GERD, candidal esophagitis, constipation, diarrhea, NAFLD | Autonomic neuropathy drives motility disorders |
| Celiac disease | Malabsorption, dermatitis herpetiformis, elevated LFTs | See Section 15 |
| Cystic fibrosis | Pancreatic insufficiency, meconium ileus, DIOS (distal intestinal obstruction syndrome), biliary cirrhosis | CFTR mutation → thick secretions blocking pancreatic ducts |
| HIV/AIDS | Opportunistic esophagitis (Candida, CMV, HSV), chronic diarrhea (MAC, Cryptosporidium, microsporidiosis), Kaposi sarcoma, lymphoma | Approach depends on CD4 count; CMV = linear ulcers; HSV = small shallow ulcers; Candida = white plaques |
| Vasculitis (PAN, HSP/IgA) | Mesenteric ischemia, GI bleeding, bowel perforation | IgA vasculitis: abdominal pain + purpura + arthritis ± renal |
| Pregnancy | Hyperemesis gravidarum, GERD, cholestasis of pregnancy, HELLP syndrome, acute fatty liver of pregnancy | Intrahepatic cholestasis: pruritus + elevated bile acids; treat with UDCA |
51 EGD & Colonoscopy Procedures
Esophagogastroduodenoscopy (EGD)
Forward-viewing flexible endoscope passed through the mouth to examine the esophagus, stomach, and duodenum (to D2). Indications: dysphagia, odynophagia, refractory GERD, UGIB, unexplained anemia/weight loss, Barrett's surveillance, gastric ulcer follow-up, celiac biopsy, caustic ingestion assessment. Diagnostic and therapeutic: biopsy, hemostasis (injection, thermal, clips, banding), dilation (balloon or bougie for strictures), stent placement, foreign body removal, PEG tube placement, EMR/ESD. Sedation: moderate (midazolam + fentanyl) or monitored anesthesia care (propofol). Complications: perforation (0.03%), bleeding (0.5-1% when biopsying), aspiration, sedation-related. Hold anticoagulants per ASGE guidelines; continue aspirin for high-risk cardiac patients.
Colonoscopy
Examination of the entire colon and terminal ileum using a flexible endoscope. Bowel preparation: split-dose PEG (e.g., GoLYTELY 4L or MiraLAX + Gatorade, or low-volume preps: SUTAB tablets, SUPREP). Split dosing (half the evening before, half on the morning of procedure) produces superior prep quality. Adequacy assessed by Boston Bowel Preparation Scale (BBPS): 0-3 per segment (right, transverse, left), total score 0-9; ≥ 6 with all segments ≥ 2 = adequate. Key quality metrics: cecal intubation rate (≥ 95%), adenoma detection rate (ADR) ≥ 25% (minimum acceptable), withdrawal time ≥ 6 minutes. Therapeutic capabilities: polypectomy (cold snare for < 10 mm, hot snare for > 10 mm), EMR for large sessile polyps, hemostasis, dilation, stent placement, decompression (Ogilvie syndrome). Complications: perforation (0.05-0.1%), post-polypectomy bleeding (0.5-2%), post-polypectomy syndrome (transmural burn without perforation — focal pain, low-grade fever, managed conservatively).
52 ERCP & EUS Procedures
Endoscopic Retrograde Cholangiopancreatography (ERCP)
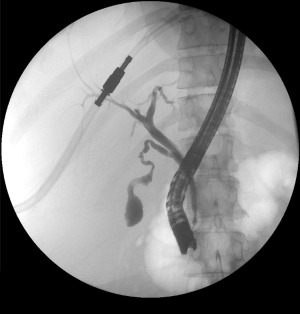
Side-viewing duodenoscope positioned at the ampulla of Vater to access the biliary and pancreatic ducts. Now almost exclusively therapeutic (diagnostic ERCP replaced by MRCP and EUS). Indications: CBD stone extraction, biliary sphincterotomy, biliary/pancreatic stenting (for malignant/benign strictures), ampullectomy, treatment of bile duct leaks, management of pancreatic duct disruption/strictures. Technique: cannulation of the desired duct via the ampulla, contrast injection under fluoroscopy, sphincterotomy with electrocautery wire, balloon sweep for stone extraction, dilation of strictures, stent placement (plastic for temporary, metal for malignant obstruction). Complications: post-ERCP pancreatitis (PEP) (3-10%, most common; risk factors: young female, normal bilirubin, sphincter of Oddi dysfunction, difficult cannulation); prevention with rectal indomethacin (100 mg suppository) + prophylactic pancreatic duct stent in high-risk patients. Other complications: bleeding post-sphincterotomy (1-2%), perforation (0.3-0.6%), cholangitis, air embolism (rare).
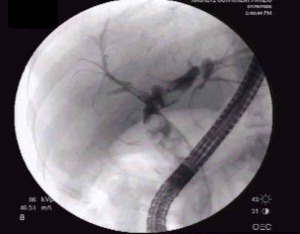
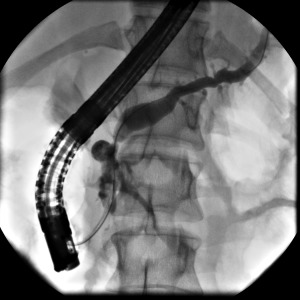
Endoscopic Ultrasound (EUS)
Echoendoscope (linear or radial) providing high-resolution ultrasound imaging from within the GI lumen. Two main functions: diagnostic imaging (highest resolution for pancreatic masses, subepithelial lesions, bile duct stones, mediastinal lymph nodes, locoregional staging of esophageal/gastric/rectal cancers) and tissue acquisition (FNA/FNB) — gold standard for sampling pancreatic masses, mediastinal/abdominal lymph nodes, liver lesions, and subepithelial GI tumors. Expanding therapeutic role: EUS-guided celiac plexus neurolysis (pain in pancreatic cancer), EUS-guided biliary drainage (when ERCP fails), EUS-guided pancreatic duct drainage, EUS-guided gallbladder drainage (LAMS = lumen-apposing metal stent), EUS-guided gastrojejunostomy for gastric outlet obstruction, EUS-guided pancreatic cyst ablation.
53 Capsule Endoscopy, Liver Biopsy, Paracentesis Procedures
Video Capsule Endoscopy (VCE)
Swallowed wireless capsule (PillCam SB3 is most common) with camera capturing 2-6 frames/second transmitted to a recorder belt. Battery life: ~12 hours. Primarily for small bowel evaluation: obscure GI bleeding (first-line), suspected small bowel Crohn's, small bowel tumors, celiac assessment, polyposis surveillance. Bowel prep (clear liquids + simethicone, some protocols add PEG) improves visualization. Contraindications: known or suspected stricture (capsule retention risk — use patency capsule first), swallowing disorders (can be placed via EGD), pacemakers (generally safe with newer capsules). Capsule retention (< 2% overall, higher in Crohn's) managed with deep enteroscopy retrieval, medical therapy, or surgery.
Percutaneous Liver Biopsy
Needle biopsy for histologic evaluation when non-invasive tests are insufficient. Common indications: MASLD/MASH staging, autoimmune hepatitis diagnosis, drug-induced liver injury, infiltrative diseases, unexplained LFT elevation, post-transplant rejection monitoring. Approaches: percutaneous (most common — right intercostal, US-guided, using cutting needle [16-18G Tru-Cut] or aspiration needle), transjugular (for coagulopathy or ascites — also allows HVPG measurement), and EUS-guided. Pre-procedure: check platelets (> 50-60k), INR (< 1.5), hold anticoagulants/antiplatelets per guidelines, type and screen. Complications: pain (most common, 20-30%), bleeding (0.5%, mostly within 2-4 hours — observe 4-6 hours post-procedure), pneumothorax, biliary peritonitis, and rarely tumor seeding.
Diagnostic & Therapeutic Paracentesis
Needle aspiration of ascitic fluid from the peritoneal cavity. Diagnostic paracentesis: every patient with new-onset ascites and every hospitalized patient with ascites. Technique: US-guided preferred (reduces complication risk); site: LLQ (left lower quadrant, lateral to the rectus abdominis, in the zone of deepest fluid), avoid surgical scars and the inferior epigastric vessels. Use Z-track technique. Send: cell count and differential (SBP if PMN ≥ 250), albumin (for SAAG), total protein, culture (inoculate blood culture bottles at bedside), glucose, LDH (if secondary peritonitis suspected). Therapeutic (large-volume paracentesis): for tense ascites causing respiratory compromise or refractory ascites. Remove as much fluid as needed. If > 5 L removed, give albumin 6-8 g/L removed to prevent post-paracentesis circulatory dysfunction (PPCD). Complications rare (< 1%): abdominal wall hematoma, bowel perforation, persistent leak (use a larger bore needle, not smaller; purse-string suture if persistent).
54 Endoscopic Hemostasis Techniques Procedures
| Technique | Mechanism | Indications | Details |
|---|---|---|---|
| Injection therapy (epinephrine) | Tamponade + vasoconstriction | Initial control, always combine with second modality | 1:10,000 epinephrine, inject in 0.5-2 mL aliquots around the bleeding site; monotherapy has high rebleed rate |
| Thermal coagulation (bipolar/heater probe) | Coaptive coagulation — compression + heat seals vessels | Bleeding ulcers (Forrest I-IIa), Dieulafoy, angiodysplasia | Bipolar (Gold Probe) 15-20W, firm apposition; heater probe 25-30J |
| Hemostatic clips (through-the-scope) | Mechanical compression of vessel | Bleeding ulcers, Dieulafoy, post-polypectomy bleeding, perforations | Resolution clip, QuickClip; can also mark lesions for surgical/angiographic localization |
| Over-the-scope clip (OTSC) | Large bear-claw mechanical compression | Large/deep ulcers, refractory bleeding (after standard clip failure), GI perforations, fistula closure | Padlock, OTSC — deployed like a variceal band, larger bite; 80-90% hemostasis in refractory bleeding |
| Endoscopic variceal ligation (EVL) | Band ligation — rubber bands applied to varix base | Esophageal variceal bleeding | Multi-band ligator; start at GEJ, work proximally; 4-6 bands per session |
| Argon plasma coagulation (APC) | Non-contact thermal coagulation via ionized argon gas | Angiodysplasia, GAVE (watermelon stomach), radiation proctitis, tumor ooze | 40-60W, 1-2 L/min flow; superficial — low perforation risk |
| Hemostatic spray/powder (TC-325 / Hemospray) | Mineral powder forms adherent mechanical barrier | Diffuse bleeding, cancer ooze, bridge to definitive therapy | Temporary (< 24 hours); does not allow tissue marking; must use in active bleeding (will not stick to dry surface) |
| Cyanoacrylate injection | Tissue glue polymerizes on contact with blood | Gastric variceal bleeding (fundal varices) | Mix with Lipiodol; risk of systemic embolization; preferred for GOV2 and IGV1 varices |
55 Imaging in Gastroenterology
| Modality | Common Indications | Key Interpretation Points |
|---|---|---|
| Abdominal X-ray (KUB) | Obstruction, perforation, foreign body, constipation | Free air under diaphragm (upright) = perforation; dilated loops with air-fluid levels = obstruction; "3/6/9 rule" (small bowel > 3 cm, colon > 6 cm, cecum > 9 cm) |
| Abdominal ultrasound | Gallstones (#1 test), cholecystitis, liver lesions, ascites, biliary dilation | Sonographic Murphy's sign = US-specific; CBD > 6 mm = dilated (adjust for age, post-cholecystectomy) |
| CT abdomen/pelvis | Acute abdomen, pancreatitis staging, diverticulitis, obstruction, malignancy staging, abscess | Pancreas-protocol triple-phase for pancreatic mass; portal venous phase for liver lesions; oral contrast rarely needed in acute setting |
| MRI / MRCP | CBD stones, PSC, IPMN surveillance, pancreatic cysts, liver lesion characterization (hemangioma, FNH, HCC) | MRCP: non-invasive biliary/pancreatic duct visualization; secretin-enhanced MRCP for chronic pancreatitis; MRI with hepatobiliary contrast (Eovist/gadoxetate) for HCC characterization |
| CT enterography | Small bowel Crohn's, small bowel tumors, obscure GI bleeding | Oral contrast (neutral/low density) + IV contrast; mural hyperenhancement, wall thickening, fat stranding, strictures, fistulae |
| FibroScan (VCTE) | Liver fibrosis assessment, steatosis quantification | Liver stiffness (kPa): < 7 = F0-F1; 7-9.5 = F2; 9.5-12.5 = F3; > 12.5 = F4 (cirrhosis). CAP score for steatosis. |
| Gastric emptying scintigraphy | Gastroparesis | Standard low-fat egg meal; > 60% retention at 2h or > 10% at 4h = delayed |
| HIDA scan (hepatobiliary iminodiacetic acid) | Acute cholecystitis (non-visualization of gallbladder), biliary leak, biliary atresia | Non-filling of gallbladder at 4 hours (even after morphine augmentation) = acute cholecystitis; sensitivity ~97% |
| Tagged RBC scan | Lower GI bleeding localization | Tc-99m labeled RBCs; detects bleeding ≥ 0.1 mL/min; more sensitive but less precise than CTA |
| Meckel's scan | Meckel's diverticulum | Tc-99m pertechnetate localizes ectopic gastric mucosa; sensitivity 85% in children, lower in adults |
56 Classification Systems (All)
| System | Used For | Key Features |
|---|---|---|
| Los Angeles Classification | Erosive esophagitis | Grade A (< 5 mm mucosal breaks); B (> 5 mm, not bridging folds); C (bridging, < 75%); D (≥ 75% circumference) |
| Prague C&M | Barrett's esophagus extent | C = circumferential extent; M = maximum tongue extent (cm from GEJ) |
| Chicago Classification v4.0 | Esophageal motility disorders | Types I-III achalasia (see Section 8); also classifies EGJ outflow obstruction, absent contractility, DES, jackhammer esophagus |
| Forrest Classification | Peptic ulcer bleeding | Ia (spurting), Ib (oozing), IIa (NBVV), IIb (clot), IIc (flat spot), III (clean base) |
| Lauren Classification | Gastric cancer histology | Intestinal type vs diffuse type (signet ring) |
| Paris Classification | GI polyp/lesion morphology | 0-Ip, Isp, Is (polypoid); 0-IIa, IIb, IIc (non-polypoid); 0-III (excavated) |
| Marsh Classification | Celiac disease biopsy | 0 (normal), 1 (IELs), 2 (+crypt hyperplasia), 3a/3b/3c (partial/subtotal/total villous atrophy) |
| Montreal Classification — CD | Crohn's disease phenotype | Age (A1-3), Location (L1-4), Behavior (B1-3+p) |
| Montreal Classification — UC | UC extent | E1 (proctitis), E2 (left-sided), E3 (pancolitis) |
| Truelove & Witts | UC severity | Mild/Moderate/Severe based on bloody stools, HR, temp, Hb, ESR |
| Rutgeerts Score | Post-surgical Crohn's recurrence | i0 (none), i1 (≤ 5 aphthous ulcers), i2 (> 5 aphthous or larger), i3 (diffuse aphthous ileitis), i4 (diffuse inflammation with nodules/strictures) |
| Child-Pugh Score | Cirrhosis severity | A (5-6), B (7-9), C (10-15) based on bilirubin, albumin, INR, ascites, encephalopathy |
| MELD / MELD-Na / MELD 3.0 | Liver transplant allocation / 90-day mortality | Bilirubin + INR + creatinine (± sodium ± sex/albumin in 3.0) |
| BCLC | HCC staging/treatment allocation | 0 (very early), A (early), B (intermediate), C (advanced), D (terminal) |
| Milan Criteria | HCC liver transplant eligibility | Single ≤ 5 cm OR up to 3 ≤ 3 cm each; no vascular invasion; no extrahepatic disease |
| West Haven | Hepatic encephalopathy grading | Covert (minimal), Grade I-IV |
| Hinchey Classification | Diverticulitis complications | I (pericolic abscess), II (pelvic abscess), III (purulent peritonitis), IV (fecal peritonitis) |
| Tokyo Guidelines (TG18) | Cholangitis severity | Grade I (mild), II (moderate), III (severe/organ dysfunction) |
| Ranson's Criteria | Acute pancreatitis severity | 11 criteria at admission and 48 hours; ≥ 3 = severe |
| BISAP | Acute pancreatitis severity | BUN, Impaired mental status, SIRS, Age > 60, Pleural effusion; ≥ 3 = high risk |
| Revised Atlanta | Acute pancreatitis classification | Mild, moderately severe, severe (persistent organ failure > 48h) |
| Glasgow-Blatchford | UGIB risk stratification | 0-1 = low risk (outpatient); uses BUN, Hb, SBP, HR, melena, syncope, liver/heart disease |
| Baveno VII | Portal hypertension / variceal risk | LSM < 20 kPa + platelets > 150k = can avoid screening EGD |
| Rome IV | Functional GI disorders (IBS) | Abdominal pain ≥ 1d/week × 3 months + ≥ 2 defecation-related criteria |
| Bristol Stool Scale | Stool form | Type 1 (separate hard lumps) to Type 7 (watery); 3-4 = normal |
| Boston Bowel Prep Scale | Colonoscopy prep quality | 0-3 per segment (R, T, L); total 0-9; ≥ 6 with all segments ≥ 2 = adequate |
| SAAG | Ascites etiology | ≥ 1.1 g/dL = portal hypertension (97% accuracy) |
57 Medications Master Table
Acid Suppression
| Drug (Brand) | Class / Mechanism | Dose | Indication | Pearl |
|---|---|---|---|---|
| Omeprazole (Prilosec) | PPI — irreversible H+/K+ ATPase inhibitor | 20-40 mg daily | GERD, PUD, ZES, H. pylori | Take 30-60 min before first meal; CYP2C19 substrate |
| Esomeprazole (Nexium) | PPI | 20-40 mg daily | GERD, erosive esophagitis | S-isomer of omeprazole; IV form available for GI bleeding |
| Pantoprazole (Protonix) | PPI | 40 mg daily (80 mg IV load for UGIB) | GERD, UGIB | Least CYP2C19 interaction; preferred in patients on clopidogrel |
| Lansoprazole (Prevacid) | PPI | 15-30 mg daily | GERD, PUD | Available as orally disintegrating tablet |
| Famotidine (Pepcid) | H2RA — reversible H2 receptor antagonist | 20-40 mg BID | Mild GERD, stress ulcer prophylaxis | Tachyphylaxis develops in days; no CYP interactions |
| Sucralfate (Carafate) | Mucosal protectant — binds ulcer base | 1g QID (1 hour before meals and QHS) | Duodenal ulcer, stress ulcers | Requires acidic environment to activate; do not give with PPI; binds many drugs — separate by 2 hours |
IBD Therapies
| Drug (Brand) | Mechanism | Dose | Indication | Pearl |
|---|---|---|---|---|
| Mesalamine (Asacol HD, Lialda, Pentasa, Rowasa, Canasa) | 5-ASA — topical anti-inflammatory (inhibits prostaglandin/leukotriene synthesis) | 2.4-4.8 g/day oral; 4g enema QHS; 1g supp daily | UC (mild-moderate); NOT effective for CD | Combination oral + rectal superior to either alone; nephrotoxicity rare but check Cr annually |
| Azathioprine / 6-MP (Imuran / Purinethol) | Thiopurine — purine analog immunosuppressant | AZA 2-2.5 mg/kg/day; 6-MP 1-1.5 mg/kg/day | IBD maintenance; steroid-sparing | Check TPMT/NUDT15 genotype before starting; monitor CBC q3 months; lymphoma risk (small absolute risk); pancreatitis (3%); avoid allopurinol (6× toxicity unless dose-reduced to 25%) |
| Methotrexate | Folate antagonist — immunosuppressant | 25 mg SC weekly (induction) → 15 mg SC weekly (maintenance) | Crohn's disease (thiopurine-intolerant) | Supplement folic acid 1 mg daily (except MTX day); teratogenic — absolute contraindication in pregnancy; monitor LFTs, CBC |
| Budesonide (Entocort EC, Uceris) | Locally active corticosteroid, 90% first-pass metabolism | 9 mg daily × 8-12 weeks, then taper | Mild-moderate ileal/right colonic CD; mild-moderate UC (Uceris formulation) | Fewer systemic steroid side effects than prednisone; still suppresses HPA at 9 mg |
Hepatitis Antivirals
| Drug (Brand) | Mechanism | Dose | Indication | Pearl |
|---|---|---|---|---|
| Entecavir (Baraclude) | NUC — HBV polymerase inhibitor | 0.5 mg daily (1 mg if lamivudine-resistant) | Chronic HBV | High barrier to resistance; renal dose adjust; take on empty stomach |
| Tenofovir disoproxil (Viread) | NUC — HBV polymerase inhibitor | 300 mg daily | Chronic HBV | Risk of nephrotoxicity and bone mineral density loss; check Cr/phosphorus |
| Tenofovir alafenamide (Vemlidy) | NUC — HBV polymerase inhibitor (prodrug) | 25 mg daily | Chronic HBV | Improved renal/bone safety vs TDF; not for decompensated cirrhosis |
| Sofosbuvir/velpatasvir (Epclusa) | NS5B + NS5A inhibitor (pangenotypic DAA) | 1 tablet daily × 12 weeks | Chronic HCV (all genotypes) | Check HBV before starting (risk of HBV reactivation); > 95% SVR |
| Glecaprevir/pibrentasvir (Mavyret) | NS3/4A protease + NS5A inhibitor | 3 tablets daily × 8-12 weeks | Chronic HCV (all genotypes) | Shortest treatment course; avoid in decompensated cirrhosis (protease inhibitor) |
Prokinetics & Antiemetics
| Drug (Brand) | Mechanism | Dose | Indication | Pearl |
|---|---|---|---|---|
| Metoclopramide (Reglan) | Dopamine antagonist / prokinetic | 5-10 mg PO/IV AC and QHS | Gastroparesis, GERD, nausea | Black box: tardive dyskinesia with > 12 weeks use; also causes QTc prolongation; avoid in Parkinson's |
| Ondansetron (Zofran) | 5-HT3 antagonist | 4-8 mg PO/IV q6-8h PRN | Nausea/vomiting | QTc prolongation at high doses; may worsen constipation |
| Prochlorperazine (Compazine) | Dopamine antagonist (phenothiazine) | 5-10 mg PO/IV q6h PRN | Nausea/vomiting | EPS risk (akathisia, dystonia); give diphenhydramine prophylactically |
| Erythromycin | Motilin agonist | 250 mg PO AC (50-100 mg IV) | Gastroparesis (prokinetic) | Tachyphylaxis within 2-4 weeks limits chronic use; QTc prolongation |
Laxatives & Antidiarrheals
| Drug (Brand) | Mechanism | Dose | Indication | Pearl |
|---|---|---|---|---|
| Polyethylene glycol (MiraLAX) | Osmotic laxative | 17g in 8oz water daily | Chronic constipation | Does not cause electrolyte shifts; safe long-term |
| Lactulose | Osmotic laxative / ammonia trap | 15-30 mL BID-QID (titrate to 2-3 stools/day) | Hepatic encephalopathy, constipation | Colonic bacteria convert to organic acids → traps NH3 as NH4+ |
| Psyllium (Metamucil) | Soluble fiber / bulk-forming | 1 teaspoon in 8oz water 1-3× daily | IBS, constipation, diarrhea (absorbs water) | Must take with adequate fluid; most evidence-based fiber for IBS |
| Loperamide (Imodium) | Mu-opioid agonist (peripheral) | 2 mg after each loose stool (max 16 mg/day) | Acute/chronic diarrhea | Does not cross BBB at standard doses; contraindicated in C. difficile and bloody diarrhea |
| Bismuth subsalicylate (Pepto-Bismol) | Antisecretory + antibacterial | 524 mg QID | Traveler's diarrhea, H. pylori (quad therapy), microscopic colitis | Causes black tongue/stool (not melena); contains salicylate — caution with ASA allergy/Reye's |
| Rifaximin (Xifaxan) | Non-absorbable antibiotic | 550 mg TID × 14d (IBS-D); 550 mg BID (HE prophylaxis) | IBS-D, HE prevention, SIBO | Minimal systemic absorption (< 0.4%); well-tolerated; expensive |
| Cholestyramine (Questran) | Bile acid sequestrant | 4g BID-TID | Bile acid diarrhea (post-cholecystectomy, ileal resection), pruritus in cholestasis | Binds many drugs — take other meds 1 hour before or 4 hours after; tastes gritty |
58 Abbreviations Master List
Anatomy
Diagnoses
Procedures & Imaging
Medications & Labs
References & Figure Sources
Figures
- Figure 1 — Overview of GI Tract Architecture. Cheng LK et al. PMC4221587. NIH/PMC.
- Figure 2 — Gastric Slow Wave Propagation. Cheng LK et al. PMC4221587. NIH/PMC.
- Figure 3 — Hepatic Portal Venous Territories. Abdel-Misih SR, Bloomston M. PMC3823389. NIH/PMC.
- Figure 4 — Couinaud Liver Segment Classification. Abdel-Misih SR, Bloomston M. PMC3823389. NIH/PMC.
- Figure 5 — Endoscopic Appearance in Esophageal Motility Disorders. Dumic I et al. PMC9174137. NIH/PMC.
- Figure 6 — Endoscopic Detection of Barrett Esophagus. Yamamoto K et al. PMC8252963. NIH/PMC.
- Figure 7 — Achalasia on Barium Swallow. Dumic I et al. PMC9174137. NIH/PMC.
- Figure 8 — Achalasia Subtypes on High-Resolution Manometry. Dumic I et al. PMC9174137. NIH/PMC.
- Figure 9 — Endoscopic Findings in Eosinophilic Esophagitis. Okimoto E et al. PMC9777529. NIH/PMC.
- Figure 10 — EoE Complications: Narrow-Caliber Esophagus. Okimoto E et al. PMC9777529. NIH/PMC.
- Figure 11 — Primary Prophylaxis of Variceal Bleeding. Gluud LL et al. PMC3399010. NIH/PMC.
- Figure 12 — Peptic Ulcer Endoscopic Appearance. Kavitt RT et al. PMC6140150. NIH/PMC.
- Figure 13 — H. pylori Treatment Regimens. Kavitt RT et al. PMC6140150. NIH/PMC.
- Figure 14 — Gastric Cancer Histology: Signet Ring Cell Carcinoma. Hu B et al. PMC3418539. NIH/PMC.
- Figure 15 — Gastric Cancer Histology: Tubular Adenocarcinoma. Hu B et al. PMC3418539. NIH/PMC.
- Figure 16 — Celiac Disease Histopathology: Marsh Classification. Giuffrida P et al. PMC7931573. NIH/PMC.
- Figure 17 — Small Bowel Obstruction on Abdominal X-ray. Defined. PMC8292005. NIH/PMC.
- Figure 18 — SBO Transition Point on CT. Defined. PMC8292005. NIH/PMC.
- Figure 19 — Crohn's Ileitis on MR Enterography. Defined. PMC8292005. NIH/PMC.
- Figure 20 — Mucosal Barrier in Health vs. IBD. Kucharzik T et al. PMC4782273. Dtsch Arztebl Int.
- Figure 21 — IBD Endoscopic and Imaging Findings. Kucharzik T et al. PMC4782273. Dtsch Arztebl Int.
- Figure 22 — Crohn's Disease Treatment Algorithm. Kucharzik T et al. PMC4782273. Dtsch Arztebl Int.
- Figure 23 — Ulcerative Colitis Treatment Algorithm. Kucharzik T et al. PMC4782273. Dtsch Arztebl Int.
- Figure 24 — Colorectal Cancer Screening Guidelines. Defined. PMC6878826. NIH/PMC.
- Figure 25 — Acute Diverticulitis on CT. Defined. PMC7664754. NIH/PMC.
- Figure 26 — Complicated Diverticulitis: Abscess on CT. Defined. PMC7664754. NIH/PMC.
- Figure 27 — Portal Hypertension Pathophysiology. Defined. PMC3971388. NIH/PMC.
- Figure 28 — Hepatic Stellate Cell Activation in Cirrhosis. Defined. PMC3971388. NIH/PMC.
- Figure 29 — NAFLD/MASLD Fibrosis Assessment Algorithm. Defined. PMC8324681. NIH/PMC.
- Figure 30 — Copper Homeostasis Pathway. Ala A et al. PMC3940372. NIH/PMC.
- Figure 31 — Kayser-Fleischer Ring in Wilson's Disease. Ala A et al. PMC3940372. NIH/PMC.
- Figure 32 — Pathophysiology of Severe Acute Pancreatitis. Defined. PMC3473535. BJR.
- Figure 33 — Mild Acute Pancreatitis on CT. Defined. PMC3473535. BJR.
- Figure 34 — Necrotizing Pancreatitis on CT. Defined. PMC3473535. BJR.
- Figure 35 — Infected Pancreatic Necrosis. Defined. PMC3473535. BJR.
- Figure 36 — Pancreatic Pseudocyst on CT. Defined. PMC3473535. BJR.
- Figure 37 — Secondary Prophylaxis of Variceal Bleeding. Gluud LL et al. PMC3399010. NIH/PMC.
- Figure 38 — Diverticular Microperforation on CT. Defined. PMC7664754. NIH/PMC.
- Figure 39 — Normal ERCP Cholangiogram. Defined. PMC6448759. NIH/PMC.
- Figure 40 — Choledocholithiasis on ERCP. Defined. PMC6448759. NIH/PMC.
- Figure 41 — Chronic Pancreatitis on ERCP Pancreatogram. Defined. PMC6448759. NIH/PMC.
- Figure 42 — Celiac Disease Complications: Histopathology. Giuffrida P et al. PMC7931573. NIH/PMC.
Key Trials & Guidelines
- Gyawali CP, Kahrilas PJ, Savarino E, et al. Modern diagnosis of GERD: the Lyon Consensus 2.0. Gut. 2024;73(2):361-371. PMID: 34099516
- van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer (CROSS trial). N Engl J Med. 2012;366(22):2074-2084. PMID: 22646630
- Dellon ES, Rothenberg ME, Collins MH, et al. Dupilumab in adults and adolescents with eosinophilic esophagitis. N Engl J Med. 2022;387(25):2317-2330. PMID: 35639667
- de Franchis R, Bosch J, Garcia-Tsao G, et al. Baveno VII — renewing consensus in portal hypertension. J Hepatol. 2022;76(4):959-974. PMID: 34129884
- Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med. 1999;341(6):403-409. PMID: 10468063
- Bass NM, Mullen KD, Sanyal A, et al. Rifaximin treatment in hepatic encephalopathy. N Engl J Med. 2010;362(12):1071-1081. PMID: 20200386
- Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer (PRODIGE 24). N Engl J Med. 2018;379(25):2395-2406. PMID: 29893969
- Garcia-Pagan JC, Caca K, Bureau C, et al. Early use of TIPS in patients with cirrhosis and variceal bleeding. N Engl J Med. 2010;362(25):2370-2379. PMID: 20554546
- Fernandez J, Ruiz del Arbol L, Gomez C, et al. Norfloxacin vs ceftriaxone in the prophylaxis of infections in patients with advanced cirrhosis and hemorrhage. Gastroenterology. 2006;131(4):1049-1056. PMID: 16931569
- US Preventive Services Task Force. Screening for colorectal cancer: US Preventive Services Task Force recommendation statement. JAMA. 2021;325(19):1965-1977. PMID: 34003218
- US Preventive Services Task Force. Screening for hepatitis C virus infection in adolescents and adults. JAMA. 2020;323(10):970-975. PMID: 32150748
Textbooks & Reference Works
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease. 11th ed. Feldman M, Friedman LS, Brandt LJ, eds. Elsevier; 2021.
- Schiff's Diseases of the Liver. 12th ed. Schiff ER, Maddrey WC, Reddy KR, eds. Wiley-Blackwell; 2018.
- Gray's Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. Standring S, ed. Elsevier; 2020.