Geriatrics
Every syndrome, assessment tool, prescribing principle, classification system, functional measure, ethical framework, and management strategy in geriatric medicine in one place.
01 Physiology of Aging
Aging produces predictable physiological changes across every organ system. These changes reduce physiological reserve and narrow the margin between baseline function and clinical decompensation. Understanding normal aging is essential to distinguishing disease from senescence and to anticipating how older adults respond differently to illness, medications, and surgery.
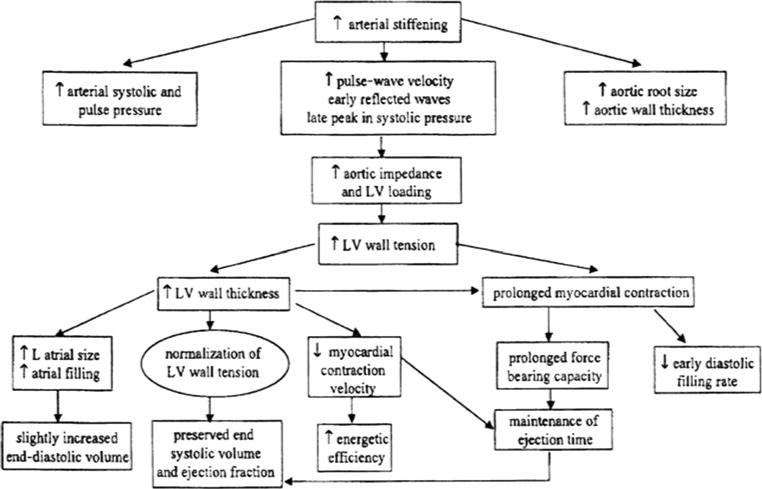
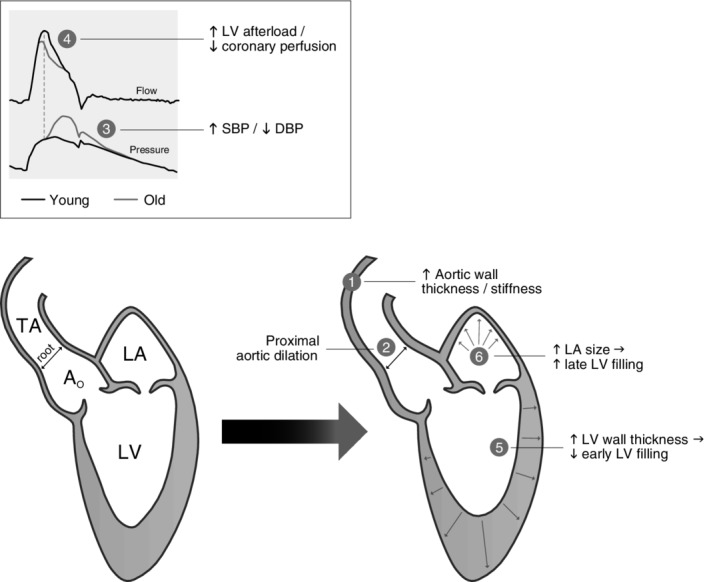
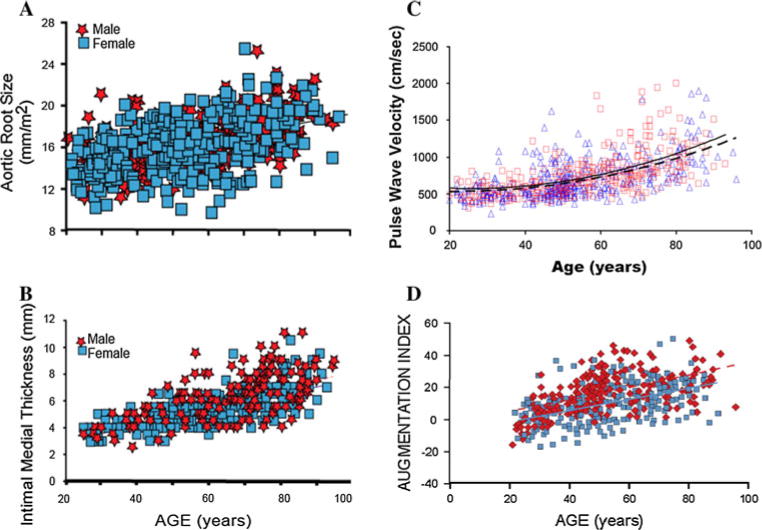
Cardiovascular Changes
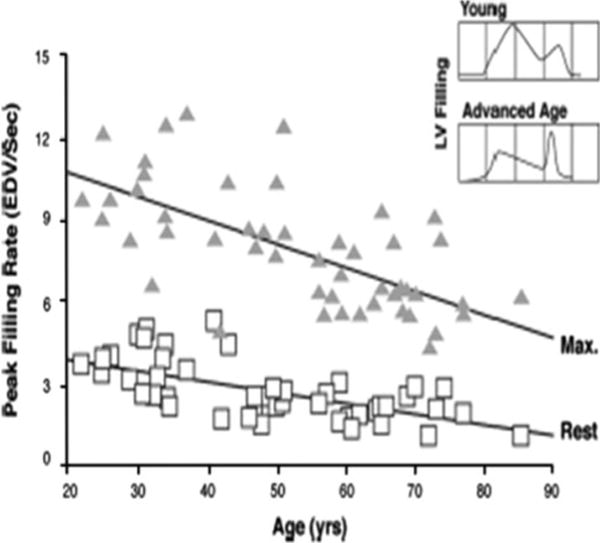
The aged heart demonstrates increased left ventricular wall thickness (up to 30% increase by age 80), decreased diastolic compliance, and reduced maximal heart rate (estimated max HR = 220 − age). Arterial stiffening raises systolic blood pressure while diastolic pressure may decrease after age 60, widening pulse pressure. Cardiac output at rest is generally maintained, but maximal cardiac output declines ~1% per year after age 30. Baroreceptor sensitivity diminishes, predisposing to orthostatic hypotension (present in 20–30% of community-dwelling elders ≥65). The conduction system accumulates fibrosis and fat, increasing the prevalence of atrial fibrillation (10% by age 80) and sick sinus syndrome.
Pulmonary Changes
Chest wall compliance decreases (costal cartilage calcification, kyphosis), while lung compliance increases (loss of elastic recoil). FEV1 declines ~30 mL/year after age 30. Residual volume (RV) increases; vital capacity (VC) decreases. FRC and closing capacity converge, leading to airway closure during tidal breathing (particularly in the supine position), which creates ventilation-perfusion mismatch and reduces PaO2. Expected PaO2 ≈ 104 − (0.27 × age) mmHg. Mucociliary clearance slows, and the cough reflex weakens, increasing aspiration pneumonia risk.
Renal Changes
Renal mass decreases ~25–30% by age 80, primarily from cortical nephron loss. GFR declines approximately 1 mL/min/1.73 m² per year after age 40, even without hypertension or diabetes. Serum creatinine may remain normal despite reduced GFR because of decreased muscle mass (reduced creatinine production). The Cockcroft-Gault equation [(140 − age) × weight / (72 × Cr) × 0.85 if female] or CKD-EPI is essential for dosing adjustments. Tubular concentrating ability declines, reducing the ability to conserve sodium and water — older adults are more susceptible to dehydration and hyponatremia.
Neurological Changes
Brain weight declines ~5% per decade after age 40. Cerebral blood flow decreases ~20% by age 80. Processing speed slows, and short-term memory mildly declines, but crystallized intelligence (vocabulary, general knowledge) is preserved. Deep tendon reflexes may diminish; ankle jerks may be absent. Vibratory sensation in the feet decreases. Sleep architecture shifts: total sleep time decreases, sleep latency increases, Stage N3 (slow-wave sleep) decreases, and nighttime awakenings increase.
Musculoskeletal Changes
Muscle mass declines ~3–8% per decade after age 30 (sarcopenia). Type II (fast-twitch) muscle fibers are preferentially lost, reducing power and increasing fall risk. Bone mineral density peaks at age 25–30 and then declines ~0.5–1% per year; in postmenopausal women, loss accelerates to 2–3% per year for the first 5–10 years. Joint cartilage thins, and intervertebral discs lose water content, contributing to height loss (~1 cm per decade after age 40).
Hepatic & GI Changes
Hepatic blood flow decreases ~40–45% by age 65, reducing first-pass metabolism. Phase I metabolism (CYP450 oxidation) declines, while Phase II (conjugation) is relatively preserved. Gastric acid production may decrease (achlorhydria in ~30% of those >80). Colonic transit time increases, contributing to constipation (reported in 30–50% of community-dwelling elders).
Immune System — Immunosenescence
Thymic involution begins in puberty; by age 60–70, the thymus is largely replaced by fat. Naïve T-cell production declines, and the T-cell repertoire narrows. Vaccine response is attenuated — influenza vaccine efficacy drops from 70–90% in young adults to 30–40% in those ≥65 (hence high-dose and adjuvanted formulations). Chronic low-grade inflammation ("inflammaging") contributes to atherosclerosis, sarcopenia, and frailty.
02 Comprehensive Geriatric Assessment
The Comprehensive Geriatric Assessment (CGA) is the cornerstone of geriatric medicine — a multidimensional, interdisciplinary diagnostic process to determine an older person's medical, psychological, social, and functional capabilities and to develop a coordinated care plan. Randomized trials show CGA improves survival, function, and rates of living at home versus usual care (meta-analysis: NNT ~12 to prevent one death or institutional placement at 12 months).
CGA Domains
| Domain | Assessment Tools | Key Elements |
|---|---|---|
| Functional status | Katz ADL, Lawton IADL, Barthel Index | Bathing, dressing, toileting, transfers, continence, feeding; shopping, cooking, finances, medications, transportation |
| Cognition | MMSE, MoCA, Mini-Cog, Clock Draw | Memory, orientation, executive function, language, visuospatial |
| Mood | GDS-15, PHQ-9, Cornell Scale | Depression, anxiety, apathy, suicidal ideation |
| Mobility & balance | Timed Up and Go (TUG), Berg Balance Scale, 4-Stage Balance, gait speed | Fall risk, assistive device use, lower-extremity strength |
| Nutrition | MNA (Mini Nutritional Assessment), BMI, albumin, prealbumin | Weight loss, appetite, dentition, dysphagia screening |
| Polypharmacy | Beers criteria, STOPP/START, medication reconciliation | Number of medications, high-risk drugs, adherence, OTC/supplements |
| Social | Social history, caregiver assessment | Living situation, caregiver burden (Zarit), financial resources, social isolation |
| Sensory | Whisper test, Snellen chart, otoscopy | Vision (cataracts, macular degeneration), hearing (presbycusis) |
| Advance care planning | Advance directive review | Healthcare proxy, POLST, code status, goals of care |
Screening Tools at a Glance
| Tool | Time | Cut-off | Use |
|---|---|---|---|
| Mini-Cog | 3 min | ≤2 of 5 (3-word recall + clock) = positive | Rapid dementia screen |
| MoCA | 10 min | <26/30 = cognitive impairment | Detects MCI (more sensitive than MMSE) |
| MMSE | 7–10 min | <24/30 = dementia likely | Classic cognitive screen; ceiling effect |
| GDS-15 | 5 min | ≥5 = depression likely | Yes/no format; avoids somatic symptoms |
| TUG | 1 min | ≥12 sec = high fall risk | Rise from chair, walk 3 m, turn, return, sit |
| Gait speed | 1 min | <0.8 m/s = mobility limitation | Single best predictor of disability and mortality in elders |
03 Key Terminology & Abbreviations
Geriatric medicine relies on specific terminology for syndromes, functional measures, prescribing frameworks, and care settings. Mastering these terms is essential for communicating across the multidisciplinary team.
| Abbreviation | Meaning |
|---|---|
| CGA | Comprehensive geriatric assessment |
| ADL / IADL | Activities of daily living / instrumental ADLs |
| MMSE / MoCA | Mini-Mental State Examination / Montreal Cognitive Assessment |
| GDS | Geriatric Depression Scale |
| CDR | Clinical Dementia Rating |
| CAM | Confusion Assessment Method (delirium) |
| TUG | Timed Up and Go test |
| BBS | Berg Balance Scale |
| MNA | Mini Nutritional Assessment |
| FRAX | Fracture Risk Assessment Tool |
| DXA | Dual-energy X-ray absorptiometry |
| AGS | American Geriatrics Society |
| STOPP/START | Screening Tool of Older Persons' Prescriptions / Screening Tool to Alert to Right Treatment |
| ACB | Anticholinergic Cognitive Burden scale |
| POLST / MOLST | Physician Orders for Life-Sustaining Treatment / Medical Orders for LST |
| SNF | Skilled nursing facility |
| ALF | Assisted living facility |
| FIM | Functional Independence Measure |
| LCD | Local Coverage Determination (hospice eligibility) |
| ACP | Advance care planning |
| HCP | Healthcare proxy |
| DNR / DNI | Do not resuscitate / do not intubate |
| BPSD | Behavioral and psychological symptoms of dementia |
| PIM | Potentially inappropriate medication |
| ADR | Adverse drug reaction |
| CFS | Clinical Frailty Scale |
| ACS-NSQIP | American College of Surgeons National Surgical Quality Improvement Program |
04 Dementia — Alzheimer Disease
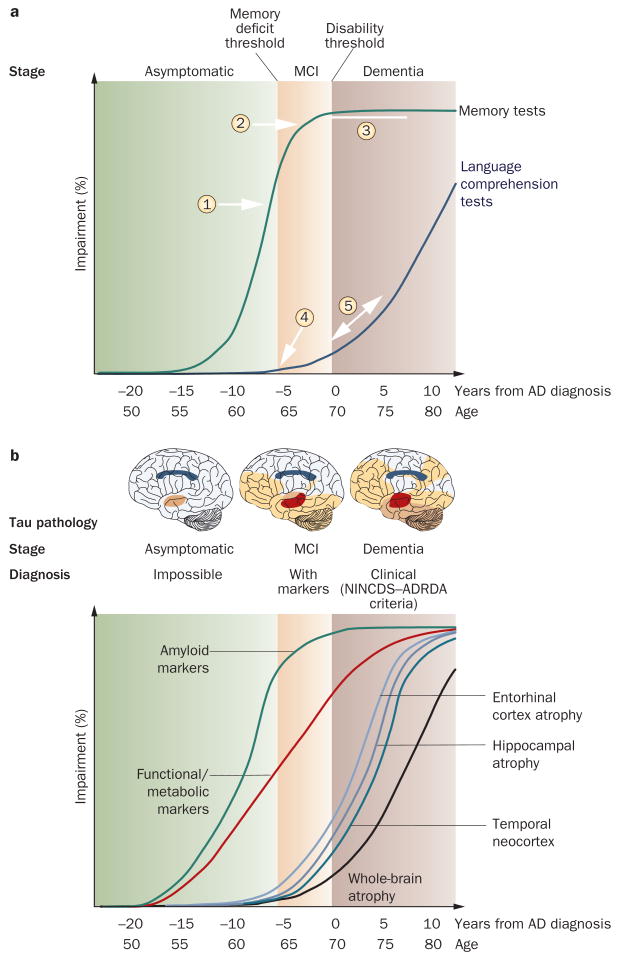
Alzheimer disease (AD) accounts for 60–80% of all dementias. Prevalence doubles every 5 years after age 65 (1% at 65, 3% at 70, 6% at 75, 12% at 80, >30% at 85). It is characterized pathologically by extracellular amyloid-beta plaques and intraneuronal neurofibrillary tangles (hyperphosphorylated tau), beginning in the entorhinal cortex and hippocampus before spreading to neocortical areas.
Clinical Presentation
The earliest feature is episodic memory impairment (inability to learn and retain new information) — patients repeat questions and misplace items. Language difficulty (word-finding), visuospatial dysfunction (getting lost in familiar places), and executive dysfunction (impaired planning, judgment) follow. Progression is insidious over 8–12 years. Anosognosia (lack of awareness of deficits) is common and increases with severity.
Diagnosis
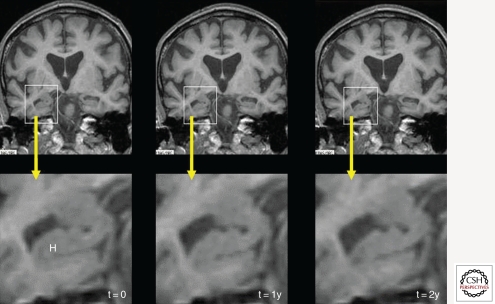
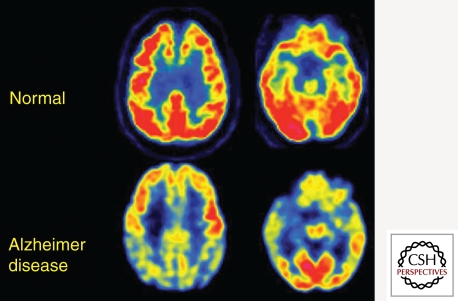
Diagnosis is clinical, supported by neuropsychological testing, structural imaging (MRI showing hippocampal & medial temporal lobe atrophy), and biomarkers. CSF biomarkers: decreased Aβ42 and elevated phospho-tau and total tau. Amyloid PET (florbetapir, flutemetamol) positive. FDG-PET shows temporoparietal hypometabolism. Labs to exclude reversible causes: TSH, B12, folate, CBC, CMP, RPR/VDRL, HIV (in appropriate populations).
Pharmacological Management
| Drug | Class | Dose | Stage | Key Points |
|---|---|---|---|---|
| Donepezil (Aricept) | AChE inhibitor | 5 mg daily × 4–6 wk, then 10 mg daily; 23 mg daily for moderate-severe | Mild-moderate-severe | GI side effects (nausea, diarrhea); bradycardia risk; vivid dreams |
| Rivastigmine (Exelon) | AChE inhibitor | Patch: 4.6 mg/24h → 9.5 mg/24h → 13.3 mg/24h | Mild-moderate | Patch preferred (less GI effects); also approved for Parkinson dementia |
| Galantamine (Razadyne) | AChE inhibitor | 4 mg BID → 8 mg BID → 12 mg BID | Mild-moderate | Also modulates nicotinic receptors |
| Memantine (Namenda) | NMDA antagonist | 5 mg daily → 10 mg BID (target 20 mg/day) | Moderate-severe | Can combine with AChE inhibitor; renal dose adjustment at CrCl <30 |
| Memantine/donepezil (Namzaric) | Combination | 28 mg/10 mg daily | Moderate-severe | Convenience; take at bedtime |
Non-Pharmacological Management
Structured daily routines, cognitive stimulation therapy, physical exercise (150 min/week moderate aerobic activity associated with slowed decline), caregiver education and support, adult day programs, and environmental safety modifications (remove throw rugs, install grab bars, door alarms for wandering). Music therapy and reminiscence therapy can reduce agitation.
Probable AD dementia: (1) Insidious onset; (2) Clear history of worsening cognition; (3) Initial and most prominent deficits in one of: amnestic presentation (most common), nonamnestic (language, visuospatial, or executive); (4) No evidence of other dementia etiology. Biomarker support: Positive amyloid (CSF Aβ42 low or amyloid PET positive) + positive neurodegeneration markers (CSF tau elevated, FDG-PET temporoparietal hypometabolism, MRI hippocampal atrophy).
05 Vascular, Lewy Body & Frontotemporal Dementias
Vascular Dementia (VaD)
Second most common cause (~15–20% of dementias). Caused by cerebrovascular disease — large-vessel strokes, small-vessel ischemia (lacunar infarcts, white matter disease), or hemorrhagic events. Classic features: stepwise decline (sudden worsening with new vascular events), executive dysfunction prominent (planning, organization, processing speed impaired more than memory), focal neurological signs (gait abnormality, pseudobulbar affect, urinary urgency). Imaging shows multi-infarct pattern or extensive white matter hyperintensities (Fazekas grade 2–3). Management: secondary stroke prevention (antihypertensives, statins, antiplatelets), control vascular risk factors. AChE inhibitors have limited evidence but are sometimes used.
Dementia with Lewy Bodies (DLB)
Third most common dementia (~5–10%). Core features (2 of 4 = probable DLB): (1) Fluctuating cognition with pronounced variations in attention and alertness; (2) Recurrent visual hallucinations (typically well-formed, detailed — people, animals); (3) REM sleep behavior disorder (acting out dreams, may precede cognitive symptoms by years); (4) Spontaneous parkinsonism (bradykinesia, rigidity, rest tremor). Supportive features: severe neuroleptic sensitivity (can cause NMS or severe EPS), postural instability, repeated falls, autonomic dysfunction.
Patients with DLB are at extreme risk for severe, potentially fatal reactions to antipsychotics — including marked worsening of parkinsonism, NMS-like syndrome, and death. Avoid first-generation antipsychotics (haloperidol). If absolutely necessary for severe psychosis or agitation, use low-dose quetiapine (12.5–25 mg) or pimavanserin (34 mg daily, FDA-approved for Parkinson disease psychosis). Monitor closely.
Frontotemporal Dementia (FTD)
Onset typically age 45–65 (younger than AD). Three clinical variants: (1) Behavioral variant FTD (bvFTD) — the most common; early personality change, disinhibition, apathy, loss of empathy, compulsive behaviors, dietary changes (sweet-food craving), executive dysfunction with relative preservation of memory and visuospatial skills; (2) Semantic variant PPA — loss of word and object meaning, fluent but empty speech; (3) Nonfluent/agrammatic variant PPA — effortful, halting speech with grammatical errors. Imaging: frontal and/or temporal atrophy. Pick bodies (intraneuronal tau inclusions) in some cases. No disease-modifying treatment; SSRIs for behavioral symptoms; AChE inhibitors are NOT effective and may worsen behavior.
06 Delirium
Delirium is an acute, fluctuating disturbance in attention and awareness, with additional cognitive disturbance (memory, orientation, language, visuospatial, perception), that develops over hours to days and is caused by a medical condition, substance, or medication. It occurs in 10–30% of hospitalized older adults and up to 50–80% of ICU patients. It is independently associated with increased mortality (3-fold), prolonged hospitalization, increased institutionalization, and accelerated cognitive decline.
Delirium is a medical emergency signaling acute brain failure. Every new case of delirium demands an urgent search for the underlying cause. Common etiologies (mnemonic DELIRIUM): Drugs (anticholinergics, benzodiazepines, opioids, steroids); Electrolyte abnormalities (Na, Ca, glucose); Lack of drugs (withdrawal from alcohol, benzodiazepines); Infection (UTI, pneumonia, skin/soft tissue); Reduced sensory input (missing glasses/hearing aids); Intracranial pathology (stroke, SDH, meningitis); Urinary/fecal retention; Myocardial/pulmonary (MI, PE, hypoxia).
Confusion Assessment Method (CAM)
The CAM is the most widely used bedside delirium screening tool (sensitivity 94%, specificity 89%). Diagnosis requires both of:
(1) Acute onset and fluctuating course — Is there evidence of an acute change in mental status from baseline? Does the abnormal behavior fluctuate during the day?
(2) Inattention — Does the patient have difficulty focusing attention (e.g., easily distracted, difficulty keeping track of what is being said)?
Plus one or more of:
(3) Disorganized thinking — Rambling or irrelevant conversation, illogical flow of ideas, unpredictable switching between subjects.
(4) Altered level of consciousness — Anything other than "alert" (vigilant, lethargic, stuporous, comatose).
Delirium Subtypes
| Subtype | Prevalence | Features | Prognosis |
|---|---|---|---|
| Hyperactive | ~25% | Agitation, restlessness, hallucinations, pulling at lines/tubes, combativeness | Better recognized; generally better prognosis |
| Hypoactive | ~50% | Lethargy, withdrawal, decreased responsiveness, apathy, reduced movement | Often missed (mistaken for depression or fatigue); worse prognosis |
| Mixed | ~25% | Features of both; fluctuates between hyperactive and hypoactive states | Intermediate prognosis |
Prevention — HELP Protocol
The Hospital Elder Life Program (HELP) reduces delirium incidence by ~40% (NNT = 6). Key components: (1) Frequent reorientation (calendar, clock, familiar objects); (2) Non-pharmacologic sleep protocol (warm milk, massage, noise reduction, avoid nighttime vitals); (3) Early mobilization; (4) Vision and hearing aids provided; (5) Hydration and nutrition; (6) Avoid deliriogenic medications (benzodiazepines, anticholinergics, meperidine).
Delirium Workup
Systematic evaluation for every new case: (1) Medications review — recent additions, dose changes, or discontinuations (especially opioids, benzodiazepines, anticholinergics, steroids, fluoroquinolones); (2) Infection — CBC, UA with culture, CXR, blood cultures if febrile; (3) Metabolic — BMP (Na, Ca, glucose, BUN/Cr), hepatic panel, TSH; (4) Cardiac/pulmonary — ECG, troponin (if indicated), pulse oximetry, ABG if hypoxic; (5) Urinary/fecal — assess for retention (bladder scan) and impaction; (6) CNS — CT head (if focal signs, on anticoagulation, or history of fall/trauma); lumbar puncture if meningitis suspected; (7) Substance — alcohol level, urine drug screen if appropriate.
Management
First line: Treat the underlying cause. Non-pharmacological interventions (reorientation, de-escalation, family presence, minimize tethers). Pharmacological (only for severe agitation with risk of harm): Haloperidol 0.5–1 mg PO/IM q4h PRN (avoid IV — QTc risk); alternative: olanzapine 2.5–5 mg PO/IM. Avoid benzodiazepines (worsen delirium) except in alcohol/benzodiazepine withdrawal and DLB/Parkinson patients (avoid antipsychotics). Dexmedetomidine for ICU delirium.
Delirium vs Dementia vs Depression
| Feature | Delirium | Dementia | Depression |
|---|---|---|---|
| Onset | Acute (hours to days) | Insidious (months to years) | Weeks to months |
| Course | Fluctuating | Progressive | Diurnal variation (worse AM) |
| Attention | Impaired (core feature) | Intact until late stages | Intact but may lack effort |
| Consciousness | Altered (clouded) | Clear until late | Clear |
| Hallucinations | Common (visual) | Less common (except DLB) | Rare |
| Reversibility | Usually reversible | Irreversible (mostly) | Treatable |
| Psychomotor | Hyper- or hypoactive | Normal until late | Retarded (or agitated) |
07 Depression & Behavioral Disturbances
Late-Life Depression
Major depression affects 1–5% of community-dwelling elders but 10–15% of hospitalized elderly and up to 25–50% of nursing home residents. Late-life depression often presents differently than in younger adults: somatic complaints predominate (fatigue, pain, GI symptoms, weight loss), cognitive dysfunction is prominent ("pseudodementia" — depressive cognitive impairment that improves with treatment), and patients may deny feeling "sad" but endorse hopelessness, worthlessness, or loss of interest. Suicide risk is highest in white men ≥85 years (rate 4× the national average).
Geriatric Depression Scale (GDS-15)
| Score | Interpretation |
|---|---|
| 0–4 | Normal, no depression |
| 5–8 | Mild depression |
| 9–11 | Moderate depression |
| 12–15 | Severe depression |
Treatment: SSRIs are first-line (sertraline 25–50 mg starting dose, target 100–200 mg; escitalopram 5 mg starting, target 10–20 mg). Avoid TCAs in elderly (anticholinergic burden, orthostatic hypotension, cardiac conduction delays). Mirtazapine (7.5–30 mg at bedtime) is useful when insomnia and weight loss are prominent. Bupropion (100–300 mg/day) avoids sexual side effects and sedation; avoid if seizure risk. SNRIs (duloxetine 30–60 mg, venlafaxine 75–225 mg) are helpful with comorbid chronic pain. Start low, go slow — but titrate to adequate dose. Response may take 6–12 weeks in elderly (longer than younger adults).
Behavioral & Psychological Symptoms of Dementia (BPSD)
BPSD affect up to 90% of dementia patients at some point. Includes agitation, aggression, wandering, sundowning, sleep-wake cycle disruption, psychosis (delusions, hallucinations), apathy, and disinhibition.
Management approach: (1) Rule out underlying cause (pain [use PAINAD scale in nonverbal patients], infection, constipation, urinary retention, medication side effect); (2) Non-pharmacological interventions first (music therapy, aromatherapy, structured activities, reduce environmental stimulation, caregiver training — DICE approach: Describe, Investigate, Create, Evaluate); (3) Pharmacological only if non-pharmacological fails and patient is danger to self/others.
All antipsychotics (typical and atypical) carry an FDA black box warning for increased mortality in elderly patients with dementia-related psychosis (1.6–1.7× increased risk of death, primarily from cardiovascular events and infections). Use only when benefits clearly outweigh risks, with informed consent, and frequent reassessment for discontinuation.
08 Falls Assessment & Prevention
Falls are the leading cause of injury, injury-related death, and emergency department visits in adults ≥65. One-third of community-dwelling elders and two-thirds of nursing home residents fall each year. Of those who fall, 20–30% suffer moderate to severe injury (hip fractures, subdural hematomas, lacerations). Fall-related costs in the US exceed $50 billion annually.
Risk Factors for Falls
| Intrinsic | Extrinsic | Situational |
|---|---|---|
| Gait/balance impairment | Throw rugs, clutter | Rushing to the bathroom |
| Lower-extremity weakness | Poor lighting | Reaching overhead |
| Visual impairment | Lack of grab bars/handrails | Climbing on chairs/ladders |
| Orthostatic hypotension | Slippery surfaces | Carrying heavy objects |
| Cognitive impairment | Ill-fitting shoes | Alcohol use |
| Medications (sedatives, antihypertensives, ≥4 meds) | Stairs without railings | Unfamiliar environments |
| Peripheral neuropathy | Uneven surfaces | Acute illness |
| Arthritis, foot disorders | Restraints (increase fall risk) | Post-prandial hypotension |
Falls Assessment Tools
| Test | Method | Interpretation |
|---|---|---|
| Timed Up and Go (TUG) | Rise from armchair, walk 3 meters, turn, walk back, sit | <10 sec = normal; 10–12 sec = borderline; ≥12 sec = high fall risk; ≥20 sec = significant mobility impairment |
| Berg Balance Scale (BBS) | 14-item test of static and dynamic balance (0–56 points) | <45 = increased fall risk; <36 = nearly 100% fall risk |
| 30-Second Chair Stand | Number of sit-to-stand repetitions in 30 seconds | Below age-sex norms = weakness |
| 4-Stage Balance Test | Feet together → semi-tandem → tandem → single-leg stand (10 sec each) | Inability to hold tandem for 10 sec = increased fall risk |
| Morse Fall Scale (hospital) | 6-item scale: history of falls, secondary diagnosis, ambulatory aid, IV/heparin lock, gait, mental status | 0–24 = low risk; 25–50 = moderate; ≥51 = high risk |
Multifactorial Fall Prevention
Exercise programs are the single most effective intervention (NNT ~10 to prevent 1 fall). Tai Chi reduces falls by 20–40%. The Otago Exercise Programme (strength + balance exercises, 3×/week + walking 2×/week) reduces falls by 35%. Additional interventions: medication review (withdraw sedatives, reduce antihypertensives if orthostatic), home safety assessment, vision correction, vitamin D supplementation (≥800 IU/day if deficient), management of orthostatic hypotension, footwear assessment, and physical therapy referral.
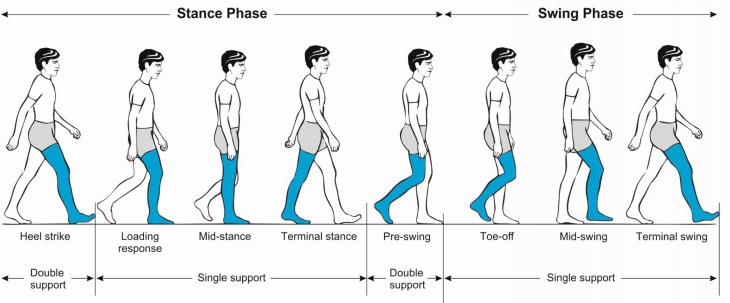
09 Gait Disorders & Assistive Devices
Common Gait Patterns in the Elderly

| Gait Type | Description | Common Causes |
|---|---|---|
| Antalgic gait | Short stance phase on affected side; limping to minimize pain | Arthritis, hip fracture, foot pathology |
| Parkinsonian gait | Short shuffling steps, reduced arm swing, flexed posture, festination, difficulty initiating movement, en bloc turning | Parkinson disease, vascular parkinsonism, DLB |
| Frontal/apraxic gait | Wide-based, magnetic (feet "glued to floor"), short steps, difficulty initiating gait, relatively preserved leg strength in bed | Normal pressure hydrocephalus (triad: gait, incontinence, dementia), frontal lobe lesions |
| Cerebellar ataxic gait | Wide-based, irregular steps, unsteady, difficulty with tandem gait | Cerebellar stroke/degeneration, alcohol, phenytoin toxicity |
| Sensory ataxic gait | Wide-based, high-stepping, positive Romberg sign (worsens with eyes closed) | B12 deficiency (subacute combined degeneration), peripheral neuropathy, posterior column disease |
| Steppage gait | High-stepping to avoid dragging foot; foot drop with foot slap | Peroneal nerve palsy, L5 radiculopathy, peripheral neuropathy |
| Waddling gait | Trunk sways side-to-side; Trendelenburg sign positive | Proximal myopathy, bilateral hip OA |
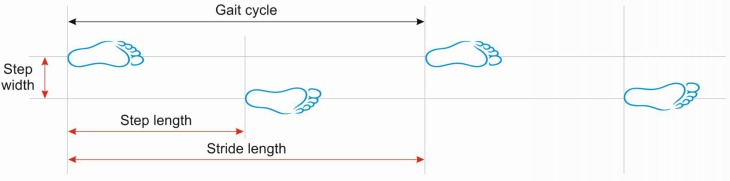
Assistive Device Selection
| Device | Support Level | Indications | Key Points |
|---|---|---|---|
| Standard cane | ~25% body weight offload | Mild balance impairment, unilateral weakness | Hold in contralateral hand; top should align with wrist crease |
| Quad cane (small/large base) | ~25% with greater stability | Moderate balance impairment, mild hemiparesis | More stable than standard cane; slower gait |
| Standard walker | ~50% body weight | Significant bilateral weakness or balance impairment | Must be lifted with each step; requires upper-extremity strength |
| Front-wheeled walker | ~50% with easier advancement | Patients who cannot lift standard walker; parkinsonian gait | Wheels allow continuous forward motion |
| Rollator (4-wheeled walker) | ~25% with seat for rest | Endurance limitations, need for seated rest | Brake locks essential; less stable than standard walker |
| Wheelchair | Full support | Non-ambulatory or severely limited endurance | Proper fit critical; pressure relief cushion; consider power wheelchair if upper extremity weakness |
10 Hip Fractures
Hip fractures are a geriatric emergency. Over 300,000 occur annually in the US, with 95% in patients ≥65. One-year mortality is 20–30% (higher in men: 37% vs 20% in women). Only 40–60% regain pre-fracture mobility, and 10–20% require long-term institutionalization. Median age at fracture is 80 years.
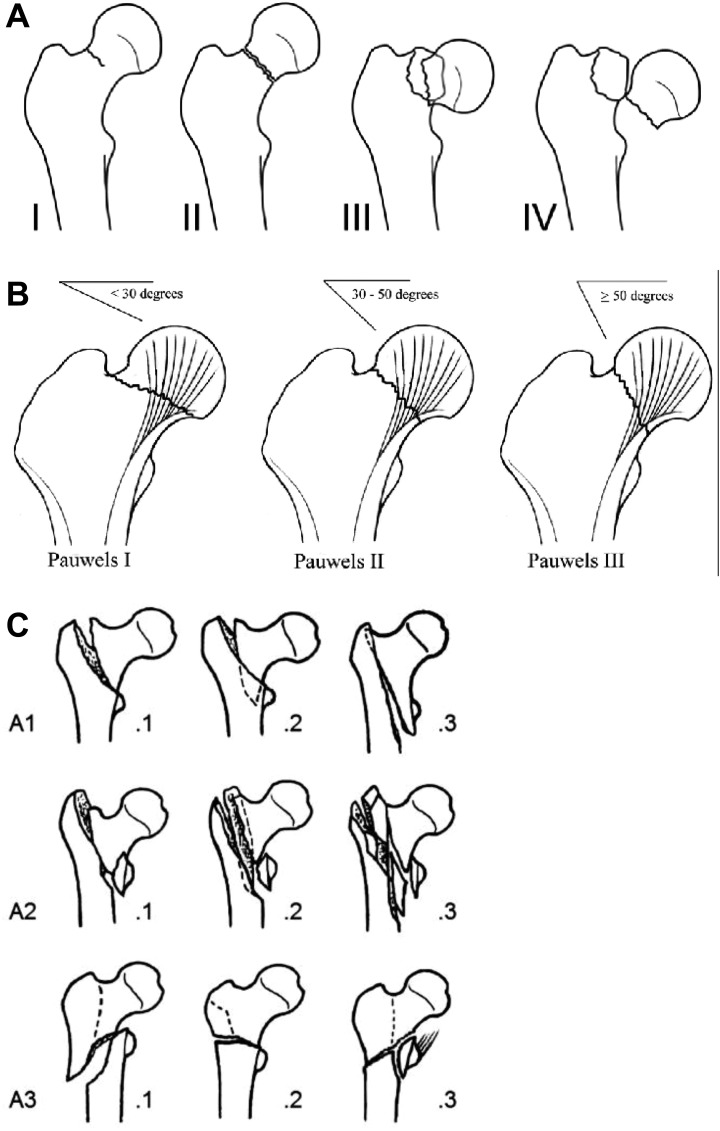
Classification
| Type | Location | Blood Supply | Treatment |
|---|---|---|---|
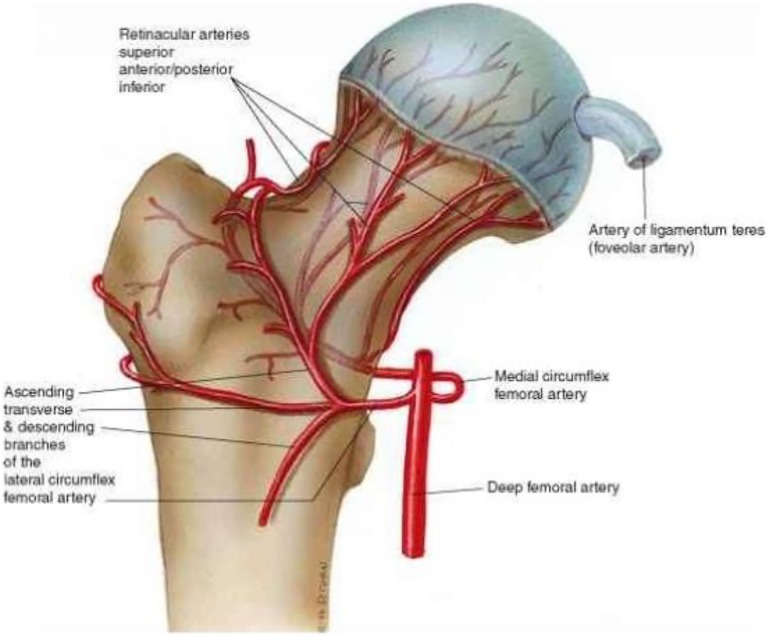
| Femoral neck (intracapsular) | Between femoral head and intertrochanteric line | Disrupted (medial femoral circumflex artery) — high AVN risk | Non-displaced: internal fixation with screws. Displaced: hemiarthroplasty (≥65 yr, low-demand) or total hip arthroplasty (active, independent) |
| Intertrochanteric (extracapsular) | Between greater and lesser trochanter | Preserved — low AVN risk | Sliding hip screw (stable patterns) or intramedullary nail (unstable patterns) |
| Subtrochanteric | Below lesser trochanter (within 5 cm) | Preserved | Intramedullary nail; consider atypical fracture (bisphosphonate-related) if lateral cortex thickening present |
Time to surgery: Operate within 24–48 hours of presentation (delays >48 hours associated with increased mortality, pulmonary complications, and pressure injuries). Preoperative: crossmatch, ECG, CXR, CBC, BMP, coagulation studies. Hold anticoagulation per protocol. DVT prophylaxis from admission (LMWH or fondaparinux if not on therapeutic anticoagulation). Pain control: fascia iliaca block preferred over systemic opioids; acetaminophen 1 g q6h scheduled. Early mobilization post-op (day 1). Orthogeriatric co-management reduces 30-day mortality by 5–7%.
11 Osteoporosis
Osteoporosis is defined by the WHO as a DXA T-score ≤−2.5 at the lumbar spine, femoral neck, or total hip. Prevalence: ~25% of women and ~5% of men ≥65. Osteoporosis is responsible for >2 million fractures annually in the US (vertebral compression fractures most common, followed by hip and distal radius fractures).
WHO Diagnostic Criteria (DXA T-score)
| T-score | Classification |
|---|---|
| ≥−1.0 | Normal |
| −1.0 to −2.5 | Osteopenia (low bone mass) |
| ≤−2.5 | Osteoporosis |
| ≤−2.5 with fragility fracture | Severe (established) osteoporosis |
FRAX Tool
The FRAX (Fracture Risk Assessment Tool) calculates 10-year probability of major osteoporotic fracture and hip fracture using age, sex, BMI, prior fracture, parental hip fracture, smoking, alcohol (≥3 units/day), glucocorticoids, rheumatoid arthritis, secondary osteoporosis, and femoral neck BMD. Treatment thresholds (US NOF): ≥20% 10-year risk of major osteoporotic fracture OR ≥3% 10-year risk of hip fracture.
Screening Recommendations
USPSTF: DXA screening for all women ≥65 and younger postmenopausal women with risk factors (FRAX equivalent of a 65-year-old white woman). Men: screen at ≥70 or earlier with risk factors (glucocorticoid use, low body weight, prior fracture, hypogonadism).
Pharmacological Treatment
| Drug | Class | Dose | Key Points |
|---|---|---|---|
| Alendronate (Fosamax) | Oral bisphosphonate | 70 mg weekly | Take fasting with 8 oz water, remain upright 30 min; esophagitis risk; drug holiday after 5 years if low-risk |
| Risedronate (Actonel) | Oral bisphosphonate | 35 mg weekly or 150 mg monthly | Similar to alendronate; delayed-release formulation taken after breakfast |
| Zoledronic acid (Reclast) | IV bisphosphonate | 5 mg IV once yearly | Preferred if GI intolerance to oral; hydrate before infusion; acute phase reaction (flu-like) in 30% |
| Denosumab (Prolia) | RANKL inhibitor | 60 mg SQ q6 months | No renal contraindication; rebound vertebral fractures if discontinued — must transition to bisphosphonate |
| Teriparatide (Forteo) | PTH analog (anabolic) | 20 mcg SQ daily × 2 years | For severe osteoporosis; black box: osteosarcoma in rats (avoid if Paget or radiation history); follow with antiresorptive |
| Romosozumab (Evenity) | Sclerostin inhibitor (anabolic) | 210 mg SQ monthly × 12 months | Dual action (builds and prevents resorption); black box: MI/stroke risk; avoid if recent CV event; follow with antiresorptive |
All patients should receive calcium 1,000–1,200 mg/day (diet + supplement) and vitamin D 800–1,000 IU/day (target 25(OH)D ≥30 ng/mL). Weight-bearing exercise and fall prevention are essential non-pharmacological measures.
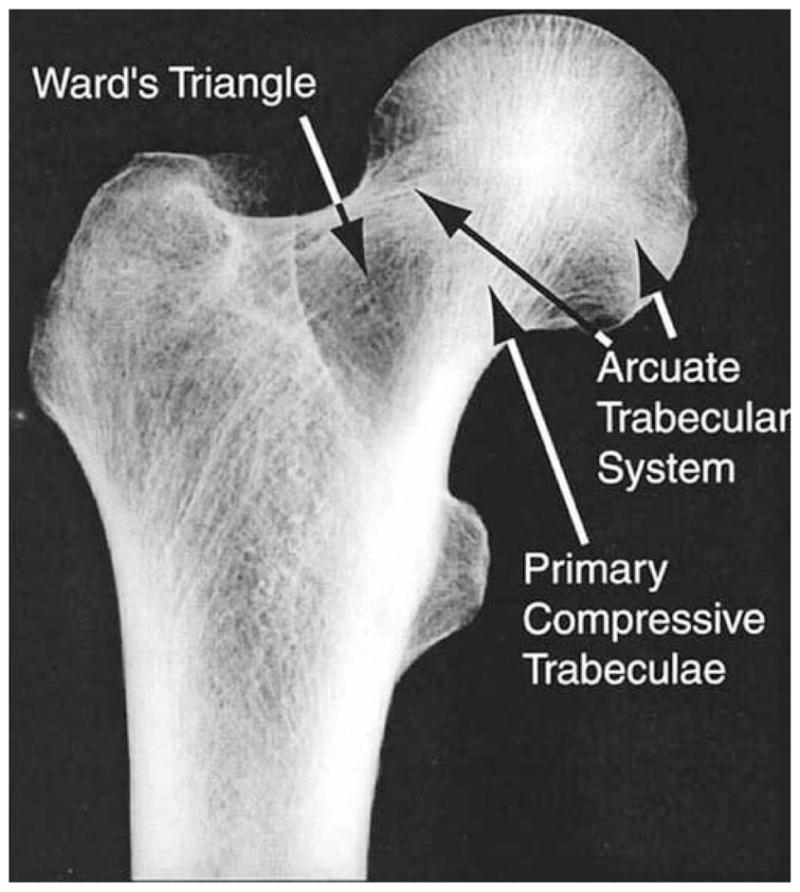
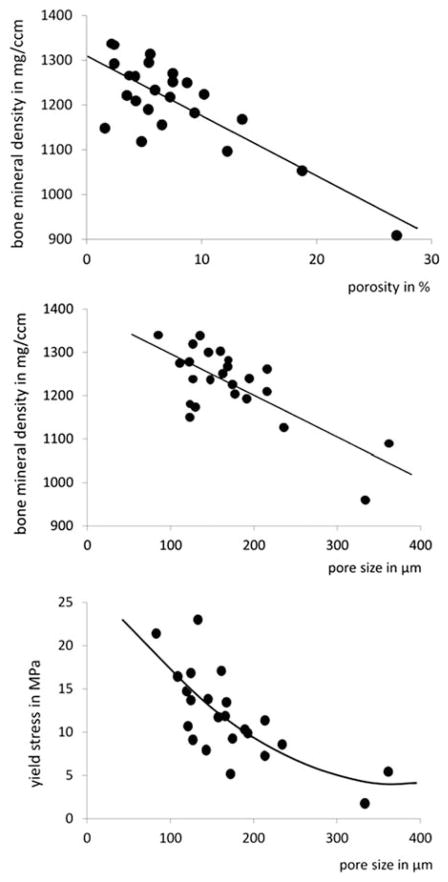
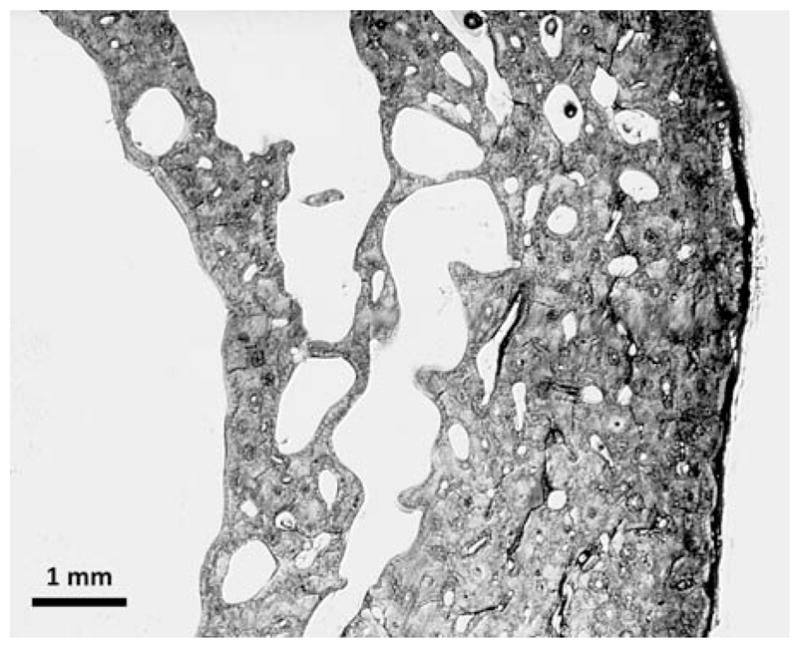
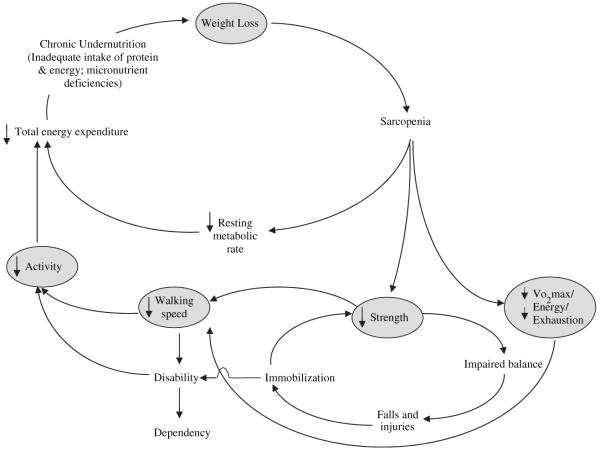
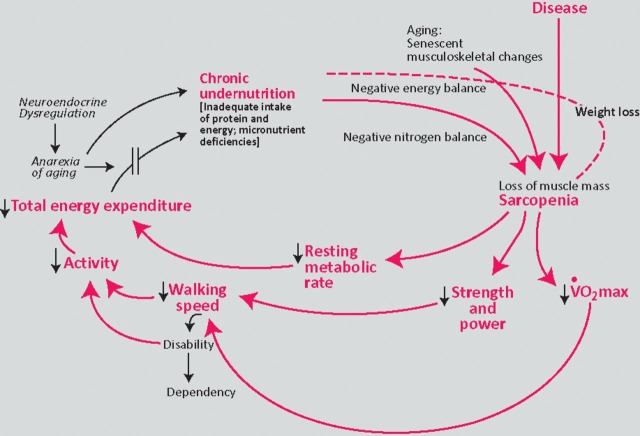
12 Sarcopenia & Frailty
Sarcopenia
Sarcopenia is the progressive, age-related loss of skeletal muscle mass and function. The EWGSOP2 (2019) definition requires: (1) Low muscle strength (grip strength <27 kg men, <16 kg women; or chair stand >15 sec for 5 rises) for probable sarcopenia; (2) Low muscle quantity (appendicular skeletal muscle mass index by DXA: <7.0 kg/m² men, <5.5 kg/m² women) to confirm; (3) Low physical performance (gait speed <0.8 m/s, SPPB ≤8, TUG ≥20 sec) indicates severe sarcopenia. Prevalence: 5–13% in 60–70 year-olds, 11–50% in those ≥80. Treatment: resistance exercise (most effective), adequate protein intake (1.0–1.2 g/kg/day, ideally 25–30 g per meal), vitamin D supplementation if deficient.
Frailty
Frailty is a clinical syndrome of decreased physiological reserve and increased vulnerability to stressors (illness, surgery, medications), leading to disproportionate adverse outcomes. Two major conceptualizations:
Fried Frailty Phenotype (Physical Frailty)
≥3 of 5 criteria = frail; 1–2 = pre-frail; 0 = robust:
| # | Criterion | Measurement |
|---|---|---|
| 1 | Unintentional weight loss | ≥10 lbs (4.5 kg) or ≥5% body weight in past year |
| 2 | Self-reported exhaustion | CES-D scale: "everything was an effort" or "could not get going" ≥3 days/week |
| 3 | Low physical activity | Lowest quintile of kcal/week expended (men <383 kcal/wk, women <270 kcal/wk) |
| 4 | Slow walking speed | Lowest quintile for height (<0.65–0.76 m/s depending on sex and height) |
| 5 | Weakness (grip strength) | Lowest quintile adjusted for sex and BMI |
Clinical Frailty Scale (Rockwood, 9-point)
| Score | Category | Description |
|---|---|---|
| 1 | Very fit | Robust, active, energetic, well-motivated, exercises regularly |
| 2 | Fit | No active disease symptoms but less fit than category 1 |
| 3 | Managing well | Medical problems well controlled; not regularly active beyond walking |
| 4 | Living with very mild frailty | Not dependent but slowed down; symptoms limit activities |
| 5 | Living with mild frailty | More evident slowing; needs help with IADLs (finances, transportation, heavy housework) |
| 6 | Living with moderate frailty | Needs help with all outside activities and housekeeping; problems with stairs; may need help with bathing |
| 7 | Living with severe frailty | Completely dependent for ADLs; stable, not at high risk of dying within 6 months |
| 8 | Very severely frail | Completely dependent; approaching end of life; could not recover from minor illness |
| 9 | Terminally ill | Approaching end of life; life expectancy <6 months; not otherwise evidently frail |
13 Beers Criteria & High-Risk Medications
The AGS Beers Criteria (updated 2023) is the most widely referenced list of potentially inappropriate medications (PIMs) in older adults (≥65). Polypharmacy (≥5 medications) affects >40% of community-dwelling elders and >90% of nursing home residents. The risk of adverse drug reactions (ADRs) increases roughly linearly with the number of medications: 5 drugs = ~50% risk of drug interaction; 8+ drugs = ~100% risk.
Key Beers Criteria Categories (Selected High-Yield)
| Drug/Class | Concern in Elderly | Recommendation |
|---|---|---|
| Benzodiazepines (all) | Cognitive impairment, delirium, falls, fractures, motor vehicle accidents | Avoid for insomnia, agitation, or delirium (use for seizures, alcohol withdrawal, anesthesia only) |
| Non-benzo hypnotics (zolpidem, eszopiclone) | Similar risks to benzodiazepines; minimal improvement in sleep latency | Avoid |
| Anticholinergics (1st-gen antihistamines: diphenhydramine, hydroxyzine; antispasmodics: oxybutynin, tolterodine) | Confusion, constipation, urinary retention, dry mouth, blurred vision, delirium, cognitive decline | Avoid; use 2nd-gen antihistamines; for OAB, use mirabegron or vibegron instead |
| NSAIDs (chronic use) | GI bleeding (risk 2–4× increased), renal failure, fluid retention, HTN exacerbation | Avoid chronic use; short course only with PPI; topical NSAIDs preferred |
| Sulfonylureas (glyburide, chlorpropamide) | Prolonged hypoglycemia (especially glyburide — long half-life, active metabolites) | Avoid glyburide; use glipizide or glimepiride if SU needed; target A1c 7.5–8.5% in frail elders |
| Meperidine (Demerol) | Neurotoxic metabolite (normeperidine) causes seizures; poor analgesic in elderly | Avoid completely |
| Metoclopramide | Extrapyramidal symptoms, tardive dyskinesia (risk increases with duration) | Avoid unless gastroparesis; limit to ≤12 weeks |
| Sliding scale insulin (sole therapy) | Higher risk of hypoglycemia without improved glycemic control | Avoid as sole strategy; use basal insulin with correction scale |
| Alpha-blockers for HTN (doxazosin, prazosin, terazosin) | Orthostatic hypotension, syncope, falls | Avoid for hypertension; acceptable for BPH |
| Skeletal muscle relaxants (cyclobenzaprine, methocarbamol, carisoprodol) | Sedation, anticholinergic effects, falls; poorly tolerated | Avoid |
14 STOPP/START Criteria
The STOPP/START criteria (version 3, 2023) complement the Beers list with a European evidence-based approach. STOPP (Screening Tool of Older Persons' Prescriptions) identifies PIMs to stop; START (Screening Tool to Alert to Right Treatment) identifies beneficial medications that are commonly omitted in older adults.
Selected STOPP Criteria (Medications to Discontinue)
| Criterion | Rationale |
|---|---|
| PPI beyond 8 weeks without indication (ulcer, GERD, Barrett) | C. difficile risk, hypomagnesemia, hip fracture, B12 deficiency |
| Long-acting benzodiazepine (diazepam, chlordiazepoxide, flurazepam) | Prolonged sedation, falls, cognitive impairment |
| Duplicate drug classes (e.g., 2 SSRIs, 2 ACE inhibitors) | No added benefit; increased side effects |
| Anticholinergic drugs in patients with dementia | Worsens cognitive function |
| Aspirin for primary prevention in ≥70 without established CVD | Bleeding risk exceeds benefit (ASPREE trial) |
| Beta-blockers with verapamil or diltiazem | Risk of heart block and severe bradycardia |
| Theophylline as monotherapy for COPD | Narrow therapeutic index; safer alternatives exist |
Selected START Criteria (Medications to Consider Starting)
| Criterion | Rationale |
|---|---|
| Statin in diabetes with major CV risk factor | Cardiovascular risk reduction |
| ACE-I/ARB in heart failure with reduced EF | Mortality reduction; frequently underused in elderly |
| Bone protection (bisphosphonate + calcium/vitamin D) if on chronic steroids | Glucocorticoid-induced osteoporosis prevention |
| Pneumococcal, influenza, herpes zoster vaccines | Preventable infections; frequently omitted |
| Topical prostaglandin for open-angle glaucoma | First-line treatment; prevents vision loss |
| Beta-blocker post-MI | Secondary prevention of MI; mortality benefit |
| SSRI/SNRI for moderate-severe depression | Untreated depression increases morbidity and mortality |
15 Deprescribing & Adverse Drug Reactions
Deprescribing Principles
Deprescribing is the planned, supervised process of dose reduction or stopping medications that are no longer beneficial or may be causing harm. It is not simply "stopping medications" — it requires a systematic approach:
(1) Ascertain all medications (prescription, OTC, supplements, herbals); (2) Identify potentially inappropriate medications using Beers/STOPP criteria and clinical judgment; (3) Determine whether each medication can be discontinued (consider indication, life expectancy, time-to-benefit, goals of care); (4) Plan withdrawal schedule (many drugs require gradual taper: benzodiazepines, SSRIs, beta-blockers, corticosteroids, opioids, gabapentinoids); (5) Monitor for withdrawal syndromes and disease recurrence.
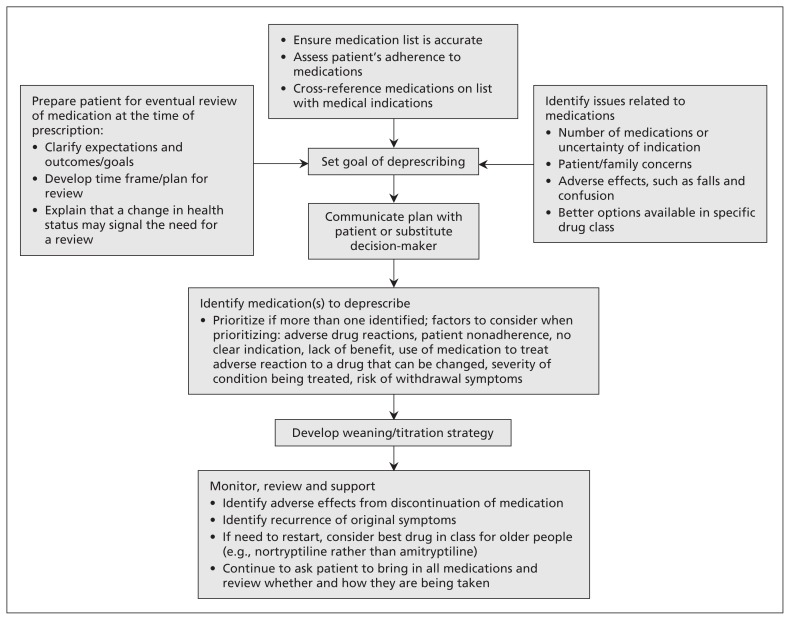
Medications Requiring Gradual Taper
| Drug Class | Withdrawal Risk | Taper Strategy |
|---|---|---|
| Benzodiazepines | Seizures, insomnia, anxiety, delirium | Reduce by 10–25% every 1–2 weeks; slower if long-term use |
| SSRIs/SNRIs | Discontinuation syndrome (dizziness, nausea, paresthesias, irritability) | Reduce by 25–50% every 2–4 weeks; paroxetine and venlafaxine highest risk |
| Opioids | Withdrawal syndrome (sweating, diarrhea, anxiety, pain flare) | Reduce by 10% every 1–2 weeks |
| Corticosteroids | Adrenal insufficiency if >3 weeks of use | Taper by 2.5–5 mg prednisone equivalent every 3–7 days |
| Beta-blockers | Rebound tachycardia, hypertension, angina | Reduce by 25–50% every 1–2 weeks |
| PPIs | Rebound acid hypersecretion | Step down to H2 blocker or half dose × 2–4 weeks, then stop |
Adverse Drug Reactions in the Elderly
ADRs account for 5–10% of hospital admissions in older adults and occur 2–7× more frequently than in younger adults. The most common culprits: anticoagulants (bleeding), diuretics (electrolyte disturbances, falls), NSAIDs (GI bleed, renal failure), digoxin (toxicity), hypoglycemic agents, and psychotropics. Presentation is often atypical: confusion (anticholinergics), falls (sedatives, antihypertensives), incontinence (diuretics, cholinesterase inhibitors), constipation (opioids, calcium channel blockers), anorexia (digoxin, SSRIs).
16 Renal Dosing & Anticholinergic Burden
Renal Dosing in the Elderly
Age-related GFR decline makes renal dose adjustment critical. Key drugs requiring adjustment in CKD:
| Drug | Normal Dose | CrCl 30–60 | CrCl 15–30 | CrCl <15 |
|---|---|---|---|---|
| Gabapentin | 300–600 mg TID | 200–700 mg BID | 100–300 mg daily | 100–300 mg post-HD |
| Metformin | 500–1000 mg BID | 500 mg BID (max) | Avoid (eGFR <30) | Contraindicated |
| Enoxaparin (treatment) | 1 mg/kg BID | Standard dose | 1 mg/kg daily | Avoid; use UFH |
| Dabigatran | 150 mg BID | 150 mg BID | 75 mg BID | Avoid |
| Memantine | 10 mg BID | Standard | 5 mg BID | 5 mg BID |
| Allopurinol | 300 mg daily | 200 mg daily | 100 mg daily | 100 mg QOD |
| Pregabalin | 150–300 mg BID | 75–150 mg BID | 25–75 mg daily | 25–75 mg post-HD |
Anticholinergic Cognitive Burden (ACB) Scale
The ACB scale assigns points (1–3) to medications based on anticholinergic activity. A total ACB score ≥3 is associated with increased risk of cognitive decline, delirium, falls, hospitalization, and mortality. Each 1-point increase is associated with a 26% increase in mortality risk.
| ACB Score | Level | Examples |
|---|---|---|
| 1 (possible anticholinergic) | Serum anticholinergic activity but no known clinical effect | Furosemide, metoprolol, ranitidine, trazodone, alprazolam, atenolol, prednisone |
| 2 (definite anticholinergic — clinically relevant) | Evidence of clinical anticholinergic effects | Amantadine, carbamazepine, cyproheptadine, loperamide, nortriptyline, pimozide |
| 3 (definite anticholinergic — high potency) | Strong anticholinergic effects; most dangerous in elderly | Oxybutynin, tolterodine, amitriptyline, paroxetine, chlorpheniramine, diphenhydramine, hydroxyzine, olanzapine, clozapine, thioridazine, doxepin (>6 mg) |
17 Urinary Incontinence
Urinary incontinence affects 30–50% of community-dwelling women ≥65 and 15–30% of men ≥65. Prevalence exceeds 50% in nursing home residents. Despite being common, it is frequently underreported and under-treated. It is a major contributor to social isolation, depression, skin breakdown, falls (rushing to bathroom), and institutionalization.
Types of Urinary Incontinence
| Type | Mechanism | Presentation | Common Causes | Treatment |
|---|---|---|---|---|
| Stress | Urethral sphincter weakness; loss of anatomic support | Leakage with cough, sneeze, laugh, exercise, lifting | Pelvic floor weakness, vaginal atrophy, post-prostatectomy | Pelvic floor exercises (Kegels), pessary, topical estrogen, duloxetine, surgical sling |
| Urge (overactive bladder) | Detrusor overactivity | Sudden urge followed by involuntary leakage; frequency, nocturia | Idiopathic, CNS disease (stroke, dementia, Parkinson), bladder irritation (UTI, stones) | Behavioral (bladder training, timed voiding), mirabegron 25–50 mg daily, vibegron 75 mg daily; avoid anticholinergics in elderly if possible |
| Overflow | Detrusor underactivity or outlet obstruction | Continuous dribbling, incomplete emptying, large PVR (>200 mL) | BPH, diabetic neuropathy, anticholinergic drugs, spinal cord lesions | Alpha-blocker (tamsulosin 0.4 mg), 5-alpha reductase inhibitor, intermittent catheterization, discontinue causative meds |
| Functional | Physical or cognitive inability to reach toilet in time | Normal urinary tract; incontinence due to external factors | Severe arthritis, dementia, restraints, environmental barriers | Prompted voiding, scheduled toileting, bedside commode, remove barriers |
| Mixed | Combination (usually stress + urge) | Features of both types | Multiple factors | Address predominant component first |
Transient Causes — DIAPERS Mnemonic
Delirium; Infection (UTI); Atrophic vaginitis; Pharmaceuticals (diuretics, anticholinergics, alpha-blockers, sedatives); Excessive urine output (hyperglycemia, CHF, hypercalcemia); Restricted mobility; Stool impaction (fecal impaction can cause urinary retention/overflow).
18 Pressure Injuries
Pressure injuries (formerly pressure ulcers or decubitus ulcers) affect ~2.5 million patients annually in the US. Prevalence in acute care: 5–15%; long-term care: 10–25%. They result from sustained pressure over bony prominences that exceeds capillary closing pressure (~32 mmHg), causing tissue ischemia and necrosis. Most common locations: sacrum (28–36%), heels (23–30%), ischial tuberosities (sitting), greater trochanters, lateral malleoli.
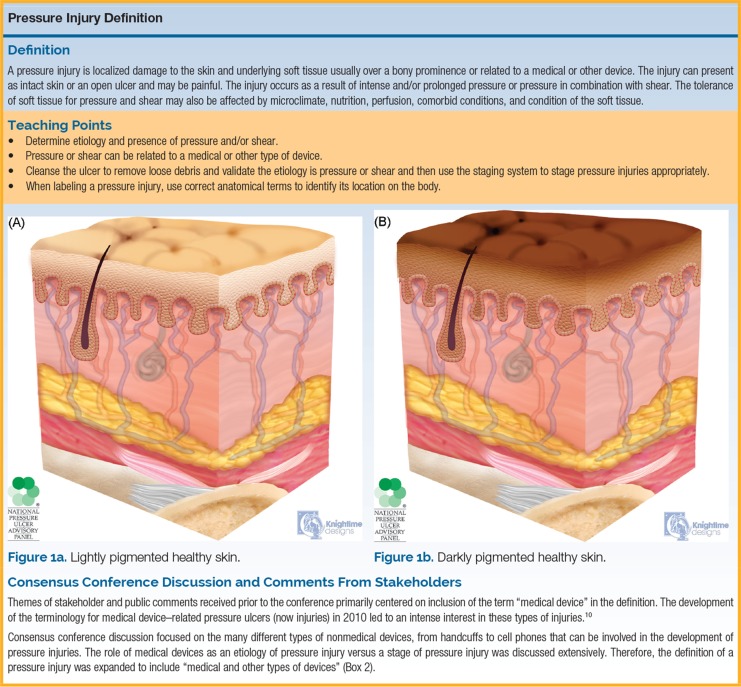
NPUAP/EPUAP Pressure Injury Staging (2016)
| Stage | Description | Features |
|---|---|---|
| Stage 1 | Non-blanchable erythema of intact skin | Skin intact; area may be painful, firm, soft, warmer or cooler than adjacent tissue. Darkly pigmented skin may not show visible blanching. |
| Stage 2 | Partial-thickness skin loss with exposed dermis | Wound bed is viable, pink or red, moist. May present as intact or ruptured serum-filled blister. No granulation tissue, slough, or eschar. |
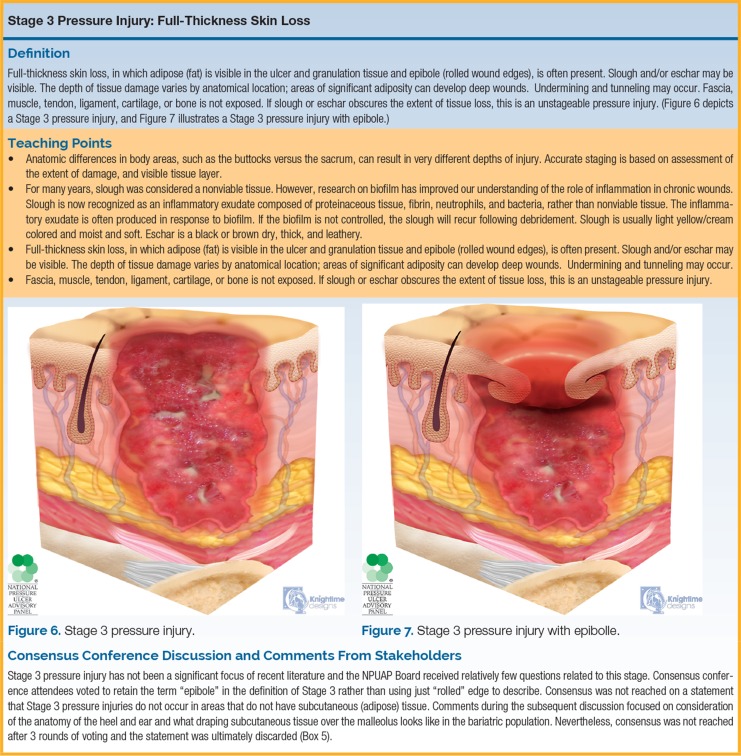
| Stage 3 | Full-thickness skin loss | Fat visible; granulation tissue and rolled wound edges may be present. Slough and/or eschar may be visible. Undermining and tunneling may occur. Fascia, muscle, tendon, ligament, cartilage, or bone NOT exposed. |
| Stage 4 | Full-thickness skin and tissue loss | Exposed or palpable fascia, muscle, tendon, ligament, cartilage, or bone. Slough and/or eschar may be present. Undermining and tunneling often present. Osteomyelitis risk. |
| Unstageable | Obscured full-thickness skin and tissue loss | Extent of tissue damage cannot be determined because it is covered by slough (yellow, tan, gray, green, brown) or eschar (tan, brown, black). True depth cannot be determined until slough/eschar is removed. Stable eschar on heel should NOT be removed. |
| Deep tissue pressure injury (DTPI) | Persistent non-blanchable deep red, maroon, or purple discoloration | Intact or non-intact skin with localized area of deep tissue damage. May evolve rapidly to reveal full extent of injury. Epidermal separation revealing dark wound bed or blood-filled blister. |
Prevention — Braden Scale
The Braden Scale assesses pressure injury risk using 6 subscales (each scored 1–4, except friction/shear scored 1–3; total range 6–23):
| Subscale | Range | Measures |
|---|---|---|
| Sensory perception | 1–4 | Ability to respond meaningfully to pressure-related discomfort |
| Moisture | 1–4 | Degree of skin exposure to moisture |
| Activity | 1–4 | Degree of physical activity |
| Mobility | 1–4 | Ability to change and control body position |
| Nutrition | 1–4 | Usual food intake pattern |
| Friction and shear | 1–3 | Amount of sliding in bed/chair |
Risk levels: 19–23 = no risk; 15–18 = at risk; 13–14 = moderate risk; 10–12 = high risk; ≤9 = very high risk.
Norton Scale
| Factor | Score Range |
|---|---|
| Physical condition | 1 (very bad) – 4 (good) |
| Mental condition | 1 (stuporous) – 4 (alert) |
| Activity | 1 (bedbound) – 4 (ambulant) |
| Mobility | 1 (immobile) – 4 (full) |
| Incontinence | 1 (doubly incontinent) – 4 (none) |
Total score range: 5–20. Score ≤14 = at risk; ≤12 = high risk.
Management Principles
Prevention: repositioning q2h, pressure-redistribution surfaces, moisture management, nutrition optimization (protein 1.25–1.5 g/kg/day, vitamin C 500 mg BID, zinc 220 mg daily). Treatment by stage: Stage 1–2: keep moist with appropriate dressings (hydrocolloid, foam, transparent film), relieve pressure. Stage 3–4: debridement (sharp, enzymatic [collagenase], or autolytic), negative-pressure wound therapy (wound VAC), infection management, surgical consultation for closure. Osteomyelitis: suspect if bone visible or probe-to-bone positive; MRI is gold standard; treat with 6–8 weeks IV antibiotics ± surgical debridement.
19 Malnutrition & Failure to Thrive
Malnutrition in the Elderly
Malnutrition affects 5–10% of community-dwelling elders, 30–60% of hospitalized elders, and 35–85% of nursing home residents. Causes include decreased appetite (anorexia of aging), dental problems, dysphagia, depression, dementia, medications (causing nausea, altered taste), social isolation, and poverty. Malnutrition increases mortality (2–3×), hospital length of stay, infection risk, pressure injury risk, and impairs wound healing.
Screening — Mini Nutritional Assessment (MNA)
The MNA-Short Form (MNA-SF) is a validated 6-item screening tool (max 14 points): ≥12 = normal nutritional status; 8–11 = at risk of malnutrition; 0–7 = malnourished. Items: food intake decline, weight loss, mobility, acute disease/stress, neuropsychological problems, BMI (or calf circumference if BMI unavailable).
Laboratory Markers
| Marker | Half-life | Normal | Clinical Significance |
|---|---|---|---|
| Albumin | ~20 days | 3.5–5.0 g/dL | Reflects chronic nutritional status AND inflammation (negative acute phase reactant); <3.0 associated with poor surgical outcomes |
| Prealbumin (transthyretin) | 2–3 days | 20–40 mg/dL | Better indicator of recent nutritional change; goal >15 mg/dL during repletion |
| Transferrin | 8–10 days | 200–360 mg/dL | Intermediate marker; affected by iron status |
| Total lymphocyte count | N/A | >1,500/mm³ | <800 = severe immune compromise from malnutrition |
Failure to Thrive (FTT)
Geriatric failure to thrive is a syndrome of weight loss (>5% in 1 month or >10% in 6 months), decreased appetite, poor nutrition, inactivity, dehydration, depression, and cognitive impairment — leading to progressive functional decline. It represents the convergence of multiple geriatric syndromes. Workup: exclude reversible causes (depression, thyroid disease, malignancy, medication effects, dental disease, dysphagia, social factors). Management is multidisciplinary: treat underlying causes, dietary supplements (oral nutritional supplements add 200–600 kcal/day), appetite stimulants (mirtazapine 7.5–15 mg, megestrol acetate 160–800 mg/day [risk of DVT, adrenal suppression — use cautiously]), physical therapy, and social services.
20 Constipation, Sleep Disorders & Dizziness
Constipation
Affects 30–50% of community-dwelling elders and up to 74% of nursing home residents. Defined as <3 bowel movements per week, straining, hard stools, or sense of incomplete evacuation. Causes: low fiber/fluid intake, immobility, medications (opioids, calcium channel blockers, anticholinergics, iron, aluminum antacids), hypothyroidism, hypercalcemia, colorectal cancer, and neurological disorders (Parkinson disease, spinal cord lesions).
Management ladder: (1) Increase fiber (25–30 g/day) and fluids (1.5–2 L/day); (2) Osmotic laxative: polyethylene glycol (MiraLAX) 17 g daily (safest long-term choice); (3) Stimulant laxative: senna 8.6–17.2 mg at bedtime (safe for chronic use despite old myths); (4) For opioid-induced: methylnaltrexone 8–12 mg SQ or naloxegol 25 mg PO daily. Avoid chronic use of mineral oil (aspiration risk, lipoid pneumonia) and phosphate enemas in CKD (hyperphosphatemia, cardiac arrest).
Sleep Disorders in the Elderly
Sleep complaints affect >50% of older adults. Normal age-related changes: decreased total sleep time (6–7 hours), increased sleep latency, more nighttime awakenings, decreased slow-wave sleep (Stage N3), and advanced sleep phase (earlier bedtime and wake time). Pathological sleep disorders: insomnia, obstructive sleep apnea (OSA; prevalence 20–60% in elderly), restless legs syndrome (RLS; 10–35%), REM sleep behavior disorder (screen for prodromal synucleinopathy), and periodic limb movement disorder.
Insomnia management: First-line: sleep hygiene (consistent bed/wake times, dark/cool/quiet room, avoid screens, limit caffeine after noon, avoid daytime naps >30 min) and CBT-I (cognitive behavioral therapy for insomnia — most effective long-term intervention). Pharmacological (short-term only if non-pharmacological fails): melatonin 0.5–3 mg, suvorexant 10 mg (dual orexin receptor antagonist), low-dose trazodone 25–50 mg. Avoid: benzodiazepines, zolpidem, diphenhydramine (Beers criteria).
Dizziness & Vertigo
Dizziness is reported by 30% of people ≥65 and 50% of those ≥85. Four categories: (1) Vertigo (illusion of movement — BPPV, vestibular neuritis, Meniere disease); (2) Presyncope (lightheadedness — orthostatic hypotension, cardiac arrhythmia, vasovagal); (3) Disequilibrium (unsteadiness — peripheral neuropathy, cerebellar disease, musculoskeletal problems); (4) Non-specific dizziness (multifactorial, anxiety, medications).
BPPV is the most common cause of vertigo in elderly (~50% of cases). Diagnosed by the Dix-Hallpike maneuver (positive = latent-onset, fatigable, upbeating/torsional nystagmus). Treated with the Epley maneuver (canalith repositioning; 80% effective in single session). Meclizine suppresses symptoms but delays central compensation — use only for acute severe episodes (≤3 days).
21 ADLs, IADLs & Functional Scales
Katz Index of Independence in ADLs
Assesses 6 basic activities of daily living. Each activity is scored as independent (1) or dependent (0). Total score: 6 = full independence; 4 = moderate impairment; ≤2 = severe functional impairment.
| ADL | Independent | Dependent |
|---|---|---|
| Bathing | Bathes self completely or needs help with only one body part | Needs help bathing more than one body part or getting in/out of tub |
| Dressing | Gets clothes and dresses self completely; may need help with shoes | Needs help dressing or needs to be completely dressed |
| Toileting | Goes to toilet, cleans self, arranges clothes | Needs help going to toilet, cleaning, or uses bedpan/commode |
| Transferring | Moves in/out of bed and chair independently (may use cane/walker) | Needs help moving in/out of bed or chair or is bedbound |
| Continence | Controls bladder and bowel completely | Partially or totally incontinent |
| Feeding | Gets food to mouth independently (food prep may be by others) | Needs partial or total help with feeding or uses parenteral feeding |
Lawton Instrumental ADL Scale
Assesses 8 more complex activities that allow independent community living. Each scored 0 (low function, dependent) or 1 (high function, independent). Total: 0 (completely dependent) to 8 (completely independent).
| IADL | Examples of Assessment |
|---|---|
| Using telephone | Looks up numbers, dials independently vs. cannot use telephone |
| Shopping | Shops independently for all needs vs. needs accompaniment or unable |
| Food preparation | Plans and prepares adequate meals vs. needs meals prepared |
| Housekeeping | Maintains house alone or with occasional help vs. needs help with all tasks |
| Laundry | Does personal laundry completely vs. all laundry done by others |
| Transportation | Drives or arranges travel independently vs. does not travel at all |
| Medications | Takes medications correctly and independently vs. unable to manage |
| Finances | Manages financial matters independently vs. unable to handle money |
Functional Independence Measure (FIM)
The FIM is an 18-item scale used in rehabilitation settings. Each item scored 1 (total assistance) to 7 (complete independence). Total score range: 18–126. Two subscales: motor FIM (13 items, max 91): self-care, sphincter control, transfers, locomotion; cognitive FIM (5 items, max 35): comprehension, expression, social interaction, problem-solving, memory. FIM score determines rehabilitation intensity needed and is used for Medicare reimbursement in inpatient rehabilitation facilities.
| FIM Level | Score | Description |
|---|---|---|
| Complete independence | 7 | All tasks performed safely, without modification, in reasonable time |
| Modified independence | 6 | Task requires assistive device, extra time, or safety concerns |
| Supervision/setup | 5 | No physical assistance but standby, cueing, or coaxing needed |
| Minimal assistance | 4 | Subject performs ≥75% of task |
| Moderate assistance | 3 | Subject performs 50–74% of task |
| Maximal assistance | 2 | Subject performs 25–49% of task |
| Total assistance | 1 | Subject performs <25% of task |
22 Rehabilitation Goals & Settings
Levels of Post-Acute Care
| Setting | Requirements | Therapy Intensity | Typical Stay |
|---|---|---|---|
| Inpatient Rehabilitation Facility (IRF) | Can tolerate 3 hours of therapy/day, 5–7 days/week; requires physician supervision; must have realistic functional goals | High (PT, OT, SLP as needed) | 12–16 days average |
| Skilled Nursing Facility (SNF) | Requires skilled nursing or therapy services; Medicare covers days 1–20 at 100% (after qualifying 3-day hospitalization); days 21–100 with copay | Moderate (1–2 hours/day) | 20–35 days average |
| Long-Term Acute Care Hospital (LTACH) | Average LOS ≥25 days; patients with complex medical needs (ventilator weaning, complex wounds, IV antibiotics) | Variable | ≥25 days |
| Home Health | Homebound status; requires skilled services (nursing, PT, OT, SLP); physician-ordered plan of care | Intermittent (2–5 visits/week) | 60-day episodes |
| Outpatient rehabilitation | Medically stable; can travel to facility | Variable (2–5 times/week) | Weeks to months |
Medicare Requirements for SNF Coverage
The "3-day rule": patient must have a qualifying inpatient hospital stay of ≥3 consecutive midnights (observation status does NOT count) within 30 days of SNF admission. Medicare Part A covers: days 1–20 fully; days 21–100 with daily copay ($204.50/day in 2024); days 101+ not covered. Patient must require daily skilled services (skilled nursing care, PT, OT, or SLP).
Short Physical Performance Battery (SPPB)
The SPPB is a composite measure of lower-extremity function used in geriatric rehabilitation research and clinical practice. Three timed tests, each scored 0–4 (total 0–12):
| Component | Scoring (0–4) |
|---|---|
| Balance (side-by-side, semi-tandem, tandem × 10 sec each) | 0 = unable; 1 = side-by-side only; 2 = semi-tandem; 3 = tandem 3–9 sec; 4 = tandem ≥10 sec |
| Gait speed (4-meter walk) | 0 = unable; 1 = >8.70 sec; 2 = 6.21–8.70; 3 = 4.82–6.20; 4 = <4.82 sec |
| Chair stands (×5) | 0 = unable; 1 = >16.70 sec; 2 = 13.70–16.69; 3 = 11.20–13.69; 4 = <11.19 sec |
Interpretation: 0–3 = severe limitation (disability, high mortality); 4–6 = moderate limitation; 7–9 = mild limitation; 10–12 = minimal limitation. A 1-point change is clinically meaningful. SPPB ≤9 is used as a diagnostic criterion for severe sarcopenia.
23 Elder Abuse — Types & Screening
Elder abuse affects an estimated 10% of community-dwelling older adults (≥60) annually, though only 1 in 14 cases is reported. It is associated with 300% increased risk of death within 3 years. Types of elder abuse:
| Type | Prevalence | Indicators |
|---|---|---|
| Physical abuse | ~2% | Unexplained bruises (in various stages of healing), fractures, burns (especially in patterns), welts; injuries inconsistent with stated history; fear of caregiver |
| Neglect (most common) | ~5% | Poor hygiene, malnutrition, dehydration, untreated medical conditions, pressure injuries, soiled clothing, unsafe living conditions |
| Emotional/psychological abuse | ~5% | Withdrawal, fearfulness, depression, anxiety, agitation; caregiver belittling, threatening, isolating the elder |
| Financial exploitation | ~5% | Unexplained changes in financial situation, missing belongings, unpaid bills despite adequate resources, new "best friends," sudden changes in will/power of attorney |
| Sexual abuse | ~0.6% | Genital bruising, STIs in unexpected populations, torn undergarments, behavioral changes |
| Self-neglect | ~10% | Inability or unwillingness to meet own basic needs (food, hygiene, medical care, safe living environment); hoarding |
| Abandonment | Rare | Desertion by caregiver; "granny dumping" at ED |
Risk Factors
Victim: cognitive impairment, functional dependence, social isolation, prior history of family violence. Perpetrator: caregiver stress/burnout, substance abuse, mental illness, financial dependence on the elder, history of violence.
Screening & Reporting
Screen with the Elder Abuse Suspicion Index (EASI) (6 yes/no questions, ≥1 positive answer warrants further investigation) or the Vulnerability to Abuse Screening Scale (VASS). All 50 US states have mandatory reporting laws for suspected elder abuse — healthcare professionals must report to Adult Protective Services (APS) even without proof. In most states, reporters are immune from liability for good-faith reports. Patient consent is NOT required to file a report.
Healthcare providers are mandatory reporters of suspected elder abuse in all 50 US states. You do NOT need proof — reasonable suspicion is sufficient. Report to Adult Protective Services (APS) or local law enforcement. Document findings objectively in the medical record (body maps for injuries, photographs with consent, verbatim quotes). If the patient is in immediate danger, involve security and social work. Do NOT confront the suspected abuser before ensuring patient safety.
24 Capacity, Guardianship & Advance Directives
Decision-Making Capacity Assessment
Capacity is a clinical determination (made by the treating physician); competency is a legal determination (made by a court). Capacity is decision-specific and may fluctuate. The four components of capacity (all must be present):
| Component | Assessment Question |
|---|---|
| Understanding | "Can you tell me what the doctor has told you about your condition and the proposed treatment?" |
| Appreciation | "How do you think this condition and treatment will affect you personally?" |
| Reasoning | "How did you arrive at your decision? What factors did you consider?" |
| Expressing a choice | "What is your decision about the proposed treatment?" (Must be able to clearly communicate a consistent choice) |
Guardianship & Conservatorship
Guardianship (of the person) grants authority over personal and healthcare decisions. Conservatorship (of the estate) grants authority over financial decisions. Both require court proceedings and a finding of incapacity. They are measures of last resort — less restrictive alternatives should be explored first (healthcare proxy, power of attorney, representative payee, supported decision-making).
Advance Directives
| Document | Purpose | Key Features |
|---|---|---|
| Living will | Written instructions about future medical care preferences | Typically addresses end-of-life scenarios (terminal illness, permanent unconsciousness); limited in scope (cannot anticipate every situation); requires capacity at time of execution |
| Durable power of attorney for healthcare (healthcare proxy) | Designates a surrogate decision-maker for healthcare decisions | Activated when patient loses capacity; proxy makes decisions based on patient's known wishes (substituted judgment) or best interests; most flexible advance directive |
| POLST / MOLST | Physician Orders for Life-Sustaining Treatment | Actionable medical orders (not just wishes); signed by physician and patient/surrogate; addresses CPR, intubation, antibiotics, artificial nutrition; portable across care settings; intended for patients with serious illness or frailty; bright pink form in most states |
| DNR / DNI order | Specifies no CPR and/or no intubation | Medical order written by physician; requires informed consent or surrogate consent; applies in current care setting; separate out-of-hospital DNR may be needed for community |
25 Goals of Care & Hospice Eligibility
Goals of Care Conversations
Goals of care discussions should occur at key transitions: new serious diagnosis, significant functional decline, hospitalization, or when prognosis is <1–2 years. The REMAP framework: Reframe (why the medical situation has changed); Expect emotion (respond with empathy); Map out patient values ("What matters most to you? What are you hoping for? What are you afraid of?"); Align with values; Plan next steps. Document goals clearly: full treatment, limited interventions (e.g., hospitalize but no ICU/intubation), or comfort-focused care.
Hospice Eligibility — General Criteria
Medicare Hospice Benefit requires: (1) Two physicians certify prognosis of ≤6 months if disease follows expected course; (2) Patient (or surrogate) chooses comfort over curative treatment; (3) Covered services: nursing, home health aide, medications for comfort, DME, counseling, spiritual care, respite care (up to 5 days), bereavement support for 13 months after death.
Disease-Specific LCD Guidelines for Hospice
| Disease | Key Eligibility Indicators |
|---|---|
| Dementia | FAST Stage 7c (loss of ambulation, meaningful speech ≤6 words, incontinence) PLUS one or more in past 12 months: aspiration pneumonia, pyelonephritis, septicemia, multiple Stage 3–4 pressure injuries, recurrent fevers, inability to maintain sufficient intake with 10% weight loss in 6 months or albumin <2.5 |
| Heart disease | NYHA Class IV despite optimal treatment; EF ≤20%; refractory angina or arrhythmia; optimal medical therapy already in place; history of cardiac arrest or syncope; concomitant HIV, renal failure, or other comorbidities |
| Pulmonary disease | FEV1 <30% predicted; resting O2 sat ≤88% on supplemental O2; cor pulmonale; unintentional weight loss >10% in 6 months; resting tachycardia >100; recurrent hospitalizations for exacerbations |
| Cancer | Metastatic or locally advanced with poor prognosis; declining functional status (PPS ≤50%); declining despite treatment or patient declines further treatment |
| Liver disease | PT >5 sec over control or INR >1.5; albumin <2.5; ascites refractory to treatment; hepatic encephalopathy; hepatorenal syndrome |
| Renal disease | Not seeking or discontinuing dialysis; CrCl <10 mL/min (<15 for diabetics); serum Cr >8.0 (>6.0 for diabetics); uremia symptoms with declining status |
| Stroke/coma | Comatose with poor prognosis on day 3; persistent vegetative state; Karnofsky ≤40% or PPS ≤40% |
26 Comfort Care & Symptom Management
Pain Management at End of Life
Pain affects 50–80% of patients at end of life. The WHO analgesic ladder (adapted for elderly): Step 1: non-opioids (acetaminophen 325–500 mg q6h, max 2 g/day in frail elderly; topical lidocaine, NSAIDs short-term); Step 2: weak opioids (tramadol 25–50 mg q6h — use cautiously due to seizure and serotonin syndrome risk); Step 3: strong opioids (morphine 2.5–5 mg PO q4h PRN initially, or oxycodone 2.5–5 mg q4–6h; hydromorphone 0.5–1 mg PO q4h for renal impairment). Adjuvants at any step: gabapentin, pregabalin (neuropathic pain); dexamethasone (bone metastases, visceral pain); lidocaine patch (localized pain).
Dyspnea Management
Opioids are first-line for dyspnea at end of life (morphine 2–5 mg PO q4h or 1–2 mg IV q2–4h). Supplemental oxygen only if hypoxic AND symptomatic benefit — a fan directed at the face can be equally effective for the sensation of breathlessness. Benzodiazepines (lorazepam 0.5–1 mg q4–6h) for anxiety-related dyspnea. Nebulized normal saline may help with secretions.
Terminal Secretions ("Death Rattle")
Occurs in 25–50% of dying patients. Caused by pooling of secretions in the hypopharynx. More distressing to family than patient (patient is typically unconscious). Treatment: reposition (lateral with head elevated), gentle oral suctioning (avoid deep suctioning), anticholinergic agents: glycopyrrolate 0.2 mg IV/SQ q4–6h (preferred — does not cross BBB), hyoscine (scopolamine) patch 1.5 mg/72h, or atropine 1% ophthalmic drops 1–2 drops SL q4h.
Nausea & Vomiting
Ondansetron 4 mg IV/PO q6–8h; haloperidol 0.5–1 mg PO/IV q6–8h (useful for opioid-induced and bowel obstruction nausea); prochlorperazine 5–10 mg PO/IV q6h. For bowel obstruction with no surgical option: octreotide 100–300 mcg SQ TID reduces GI secretions; dexamethasone 4–8 mg IV/SQ for partial obstruction.
Delirium at End of Life
Terminal delirium affects up to 88% of dying patients. First: address reversible causes if consistent with goals (medications, urinary retention, fecal impaction, pain). Haloperidol 0.5–2 mg PO/IM/IV q4–6h PRN for agitation. For refractory terminal agitation: palliative sedation with midazolam infusion (0.5–1 mg/hr IV) or phenobarbital (loading 200 mg IV, then 50–100 mg q4–6h) — a last resort for intractable symptoms near death.
Signs of Imminent Death
The following signs indicate death is likely within hours to days: (1) Progressive decrease in consciousness; (2) Mottling of extremities (livedo reticularis); (3) Cool, cyanotic extremities; (4) Decreased or absent urine output; (5) Cheyne-Stokes or agonal breathing; (6) Terminal secretions (death rattle); (7) Loss of ability to swallow; (8) Mandibular breathing (jaw movement with each breath). Educate family about these expected changes. Discontinue unnecessary medications, monitoring, and interventions. Focus on mouth care, positioning, skin care, and emotional/spiritual support.
Medications to Discontinue at End of Life
Statins, antihypertensives (unless symptomatic hypertension), diabetes medications (unless symptomatic hyperglycemia), osteoporosis medications, vitamins/supplements, anticoagulants (unless active DVT/PE causing symptoms), screening medications (aspirin for primary prevention). Continue: analgesics, anticonvulsants (prevent seizures), anxiolytics, anti-secretory agents, antiemetics, and medications providing immediate symptom relief.
27 Prognostication, Caregiver Support & Grief
Prognostication Tools
| Tool | Population | Key Features |
|---|---|---|
| Palliative Performance Scale (PPS) | All patients with serious illness | 11-level scale (0–100% in 10% increments); assesses ambulation, activity, self-care, intake, consciousness. PPS ≤50% associated with median survival ~6 months; PPS ≤20% associated with days to weeks |
| Karnofsky Performance Status (KPS) | Cancer patients | 0–100 scale; KPS ≤40 = disabled, needs specialized care; KPS ≤20 = very sick, hospitalization needed |
| ePrognosis (eprognosis.ucsf.edu) | Community-dwelling elderly | Online calculators for mortality risk at 1–10 years based on demographics, function, comorbidities |
| FAST (Functional Assessment Staging) | Alzheimer dementia | Stage 7c or beyond = hospice-eligible (see Section 25); 7 stages with substages |
| Surprise Question | All patients | "Would I be surprised if this patient died in the next 12 months?" If no, consider palliative care referral. Sensitivity ~70%, specificity ~70%. |
Caregiver Support
Caregivers of older adults (especially those with dementia) experience high rates of depression (40–70%), anxiety, social isolation, and physical illness. The Zarit Burden Interview (ZBI) is a 22-item tool (0–88 points): 0–20 = little or no burden; 21–40 = mild to moderate; 41–60 = moderate to severe; 61–88 = severe burden. Interventions: respite care (Medicare hospice benefit includes up to 5 days of inpatient respite), support groups (Alzheimer's Association 24/7 helpline: 800-272-3900), psychoeducation, caregiver training in behavioral management techniques, family counseling, adult day programs.
Grief & Bereavement
Normal grief: sadness, yearning, preoccupation with the deceased, sleep disturbance, appetite changes; typically diminishes over 6–12 months with fluctuating intensity ("waves of grief"). Prolonged grief disorder (DSM-5-TR): persistent, pervasive grief lasting ≥12 months in adults (6 months in children) after bereavement, with intense yearning/longing, preoccupation with the deceased, and clinically significant distress or impairment. Affects ~10% of bereaved individuals. Treatment: grief-focused CBT, support groups. Anticipatory grief: grief that occurs before death (during prolonged illness); can facilitate post-death adjustment. Medicare hospice benefit provides 13 months of bereavement support for families.
28 Common Bedside Procedures
Joint Injections & Aspirations
Corticosteroid injections for osteoarthritis in elderly: triamcinolone 40 mg (large joints: knee, shoulder) or 10–20 mg (small joints). Limit to 3–4 injections per joint per year. Contraindications: overlying cellulitis, prosthetic joint (refer to orthopedics), coagulopathy. Hyaluronic acid injections (viscosupplementation) for knee OA: modest evidence; may benefit patients who cannot tolerate NSAIDs or are not surgical candidates.
Urinary Catheterization
Indications for chronic indwelling catheter (Foley) in elderly: urinary retention not amenable to intermittent catheterization, Stage 3–4 sacral/perineal pressure injury with incontinence, comfort care in terminally ill patients, or acute urinary obstruction awaiting surgery. Catheter-associated UTI (CAUTI) risk increases ~5% per day of catheterization. Prevention: insert only for valid indications, remove as soon as possible, nurse-driven removal protocols, maintain closed drainage system. Avoid chronic catheter for convenience or nursing preference. Alternatives: condom catheter (male), intermittent catheterization, prompted voiding.
Paracentesis & Thoracentesis in the Elderly
Procedural considerations: higher bleeding risk with liver disease (but INR and platelet count do NOT reliably predict bleeding in cirrhosis — transfusion thresholds debated). Large-volume paracentesis (>5 L): give albumin 6–8 g per liter removed to prevent post-paracentesis circulatory dysfunction. Thoracentesis: limit to 1–1.5 L per session to reduce re-expansion pulmonary edema risk. Ultrasound guidance is standard of care for both procedures — reduces complications by 50%.
29 Pre-operative Assessment in the Elderly
Patients ≥65 account for ~40% of all surgical procedures but have 2–3× higher perioperative mortality and complications than younger patients. Standard cardiac risk assessment (Revised Cardiac Risk Index, ACC/AHA guidelines) is necessary but insufficient — geriatric-specific risks (frailty, cognitive impairment, functional dependence, polypharmacy, nutritional status) are equally important.
Geriatric-Specific Pre-operative Assessment
| Domain | Assessment | Impact |
|---|---|---|
| Frailty | Clinical Frailty Scale, Fried criteria, or Edmonton Frail Scale | CFS ≥5 associated with 2–3× increased 30-day mortality, longer LOS, discharge to facility |
| Cognition | Mini-Cog or MoCA pre-operatively | Pre-existing cognitive impairment is the strongest predictor of post-operative delirium (risk 3–5×) |
| Function | ADL/IADL assessment | Functional dependence predicts complications and inability to return home |
| Nutrition | Albumin, BMI, weight loss | Albumin <3.0 g/dL associated with 3–5× increased surgical complications and mortality |
| Polypharmacy | Medication reconciliation; Beers criteria review | Identify medications to hold (anticoagulants, antiplatelets, metformin, SGLT2i) and continue (beta-blockers, statins, levothyroxine) |
| Falls risk | TUG, fall history | Post-operative fall risk assessment and prevention protocols |
| Goals of care | Advance directive review; discuss surgical risks vs benefits in context of prognosis and values | May change decision to operate; ensures appropriate code status documentation |
ACS-NSQIP Surgical Risk Calculator
The ACS-NSQIP (American College of Surgeons National Surgical Quality Improvement Program) calculator estimates 30-day risk of major complications, mortality, and length of stay based on procedure type and 21 patient variables. Available online at riskcalculator.facs.org. Does not directly measure frailty, so should be combined with frailty assessment for comprehensive risk stratification in elderly surgical candidates.
Post-operative Delirium Prevention
Affects 15–53% of elderly surgical patients (highest after hip fracture and cardiac surgery). Prevention: proactive geriatrics consultation (reduces delirium by 36%), multicomponent non-pharmacological protocols (similar to HELP), avoid benzodiazepines and meperidine, optimize pain control (regional anesthesia when possible, scheduled acetaminophen, low-dose opioids), early mobilization (post-op day 1), sleep promotion, reorient frequently, ensure glasses/hearing aids in place.
30 Feeding Tube Decisions & Wound Care
Feeding Tube Decisions in Advanced Dementia
Dysphagia and aspiration are nearly universal in advanced dementia. Evidence strongly shows that tube feeding in advanced dementia does NOT: prevent aspiration pneumonia, prolong survival, improve nutrition, promote wound healing, improve function, or increase comfort. Tube feeding in advanced dementia is associated with increased agitation (restraints often needed to prevent self-removal), increased aspiration (continued aspiration of oral secretions and gastric reflux), and medical complications (tube site infections, diarrhea, refeeding syndrome).
The American Geriatrics Society states: "Feeding tubes are NOT recommended for older adults with advanced dementia. Careful hand feeding should be offered." The Choosing Wisely campaign lists PEG tubes in advanced dementia as one of the top interventions to avoid. Careful hand feeding (small amounts, texture-modified diet, upright positioning, unhurried meals) provides comfort and human connection and is the recommended approach.
Wound Care Principles in the Elderly
Aging skin is thinner, less elastic, and heals more slowly (50% slower epithelialization by age 80). Key principles: (1) Moisture balance — wounds heal faster in a moist environment (use appropriate dressings: hydrocolloid for low exudate, foam for moderate, alginate/hydrofiber for heavy); (2) Debridement — remove necrotic tissue (autolytic with moist dressings, enzymatic with collagenase, or sharp debridement); (3) Infection control — distinguish colonization from infection (increasing pain, erythema, warmth, purulence, delayed healing indicate infection); (4) Nutrition optimization — protein 1.25–1.5 g/kg/day, vitamin C 500 mg BID, zinc 220 mg daily; (5) Offloading — pressure redistribution for pressure injuries; therapeutic footwear for diabetic foot ulcers.
Skin Tears
Very common in elderly (fragile skin, anti-coagulant use). Classification (ISTAP): Type 1 — no skin loss (linear or flap tear, flap can be repositioned); Type 2 — partial flap loss; Type 3 — total flap loss. Management: approximate skin flap if present, apply non-adherent dressing (silicone-based dressings preferred), avoid adhesive tape directly on fragile skin (use tubular gauze or paper tape). Prevention: moisturize skin BID, padded bed rails, long sleeves, gentle handling during transfers.
31 Imaging & Diagnostics in the Elderly
Neuroimaging
Brain MRI is the preferred modality for evaluating cognitive decline: hippocampal atrophy (AD), frontal/temporal atrophy (FTD), white matter hyperintensities (vascular disease), normal pressure hydrocephalus (ventricular enlargement disproportionate to sulcal enlargement). CT head (non-contrast) for acute presentations: falls with altered mental status (r/o subdural hematoma — chronic SDH is common in elderly on anticoagulants), acute stroke. FDG-PET: temporoparietal hypometabolism (AD), frontal hypometabolism (FTD), occipital hypometabolism (DLB). DaTscan (I-123 ioflupane SPECT): differentiates DLB (abnormal) from AD (normal); evaluates nigrostriatal dopaminergic integrity.
Bone Densitometry (DXA)
Gold standard for osteoporosis diagnosis. Measures BMD at lumbar spine (L1–L4), femoral neck, and total hip. T-score = number of standard deviations from peak bone mass of a young adult reference. Z-score (compared to age-matched controls) is used in premenopausal women and men <50 — Z-score ≤−2.0 = "below expected range for age." Artifactual elevation of lumbar spine BMD can occur from degenerative changes, compression fractures, and aortic calcification — in these cases, hip BMD is more reliable.
Laboratory Testing Considerations
Age-related reference range changes: BNP increases with age (normal <100 pg/mL in <75 years, <300 pg/mL in ≥75 years); PSA cutoffs may differ; ESR is elevated with age (rule of thumb: upper normal = age/2 in men, (age + 10)/2 in women). D-dimer: age-adjusted threshold (age × 10 ng/mL for patients >50) improves specificity for VTE without reducing sensitivity. TSH: some experts suggest higher target TSH range (0.5–8.0 mIU/L) in very elderly (≥85) to avoid overtreatment of subclinical hypothyroidism.
Echocardiography in the Elderly
Age-related findings that should not be over-interpreted: mild aortic sclerosis (50% of those ≥85; does not necessarily progress to stenosis), mild mitral annular calcification (MAC), basal septal hypertrophy (sigmoid septum), and mild diastolic dysfunction (Grade I is near-universal after age 70). Aortic stenosis prevalence increases dramatically with age (2–5% of those ≥75 have severe AS) and is the most common valvular disease requiring intervention in the elderly.
Cancer Screening Cessation
| Screening | Reasonable to Stop When | Rationale |
|---|---|---|
| Colonoscopy | Age ≥75 (USPSTF: individualize 76–85; stop at 85) | Time-to-benefit for colon cancer screening is ~10 years; harms increase with age |
| Mammography | Life expectancy <10 years (USPSTF: biennial 50–74; insufficient evidence ≥75) | Time-to-benefit ~7–10 years; overdiagnosis increases with age |
| Pap smear | Age ≥65 with adequate prior screening (3 normal Paps or 2 normal HPV co-tests in 10 years) | Very low risk after adequate screening |
| PSA | Age ≥70 or life expectancy <10–15 years | Overdiagnosis and overtreatment; most prostate cancers in elderly are indolent |
| Lung CT (low-dose) | Age ≥80 or 15 years since smoking cessation | USPSTF: screen ages 50–80 with ≥20 pack-year history and currently smoke or quit <15 years ago |
32 Classification Systems (All)
This section consolidates all major classification and staging systems referenced throughout geriatric medicine.
Mini-Mental State Examination (MMSE) — Score Interpretation
| Score | Severity | Clinical Correlation |
|---|---|---|
| 27–30 | Normal | No significant cognitive impairment |
| 21–26 | Mild cognitive impairment | May manage ADLs; IADLs may be affected; subtle deficits |
| 11–20 | Moderate cognitive impairment | Requires assistance with ADLs; behavioral symptoms common; cannot live independently |
| 0–10 | Severe cognitive impairment | Dependent for all ADLs; limited verbal communication; requires 24-hour supervision |
Montreal Cognitive Assessment (MoCA) — Score Interpretation
| Score | Interpretation |
|---|---|
| 26–30 | Normal |
| 18–25 | Mild cognitive impairment |
| 10–17 | Moderate cognitive impairment |
| <10 | Severe cognitive impairment |
Note: add 1 point if education ≤12 years (maximum score remains 30). MoCA is more sensitive than MMSE for detecting mild cognitive impairment (90% vs 18% sensitivity for MCI).
Clinical Dementia Rating (CDR)
| CDR | Stage | Description |
|---|---|---|
| 0 | Normal | No cognitive impairment |
| 0.5 | Questionable/very mild dementia | Consistent slight forgetfulness; partial recollection of events; "benign" forgetfulness; fully independent |
| 1 | Mild dementia | Moderate memory loss affecting daily activities; unable to function independently at community affairs; mild difficulty with home activities; personal care intact |
| 2 | Moderate dementia | Severe memory loss; only highly learned material retained; oriented to person only; cannot make judgments; requires assistance with dressing, hygiene |
| 3 | Severe dementia | Only fragments of memory remain; oriented to person only; no pretense of independent function; requires much help with personal care; frequent incontinence |
FAST (Functional Assessment Staging — Reisberg)
| Stage | Description | Clinical Correlation |
|---|---|---|
| 1 | Normal adult | No functional decline |
| 2 | Normal aged adult | Subjective complaints of forgetfulness; no objective deficits |
| 3 | Early dementia | Decreased job functioning; difficulty with complex tasks; denial |
| 4 | Mild dementia | Needs assistance with complex tasks (finances, shopping, planning) |
| 5 | Moderate dementia | Needs help choosing appropriate clothing; may need coaxing to bathe |
| 6a | Moderately severe | Needs help dressing |
| 6b | Needs help bathing | |
| 6c | Needs help with mechanics of toileting | |
| 6d | Urinary incontinence | |
| 6e | Fecal incontinence | |
| 7a | Severe dementia | Speech ability limited to ~6 words per day |
| 7b | Speech limited to single intelligible word | |
| 7c | Loss of ambulation (hospice-eligible with comorbidities) | |
| 7d | Loss of ability to sit without support | |
| 7e | Loss of ability to smile | |
| 7f | Loss of ability to hold head up |
Fried Frailty Phenotype
See Section 12 for full criteria. Summary: 0 criteria = robust; 1–2 = pre-frail (intermediate risk); ≥3 = frail (poor outcomes).
Clinical Frailty Scale (Rockwood)
See Section 12 for complete 9-point enumeration.
Braden Scale
See Section 18 for full subscale enumeration. Total 6–23; ≤18 = at risk.
Norton Scale
See Section 18 for enumeration. Total 5–20; ≤14 = at risk.
NPUAP Pressure Injury Staging
See Section 18 for complete staging from Stage 1 through DTPI.
Berg Balance Scale (BBS)
14 items, each scored 0–4 (total 0–56): sitting balance, standing balance, sit-to-stand, standing with eyes closed, tandem standing, picking up object from floor, turning 360 degrees, alternating foot on step, standing on one leg, reaching forward, and more. Scores: 41–56 = low fall risk (independent); 21–40 = medium fall risk (walking with assistance); 0–20 = high fall risk (wheelchair-dependent).
Morse Fall Scale
| Item | Score |
|---|---|
| History of falling (this admission or within 3 months) | 0 = No; 25 = Yes |
| Secondary diagnosis | 0 = No; 15 = Yes |
| Ambulatory aid | 0 = None/bed rest/nurse assist; 15 = Crutches/cane/walker; 30 = Furniture for support |
| IV/heparin lock | 0 = No; 20 = Yes |
| Gait | 0 = Normal/immobile; 10 = Weak; 20 = Impaired |
| Mental status | 0 = Oriented to own ability; 15 = Overestimates or forgets limitations |
Total: 0–24 = low risk; 25–50 = moderate risk; ≥51 = high risk.
WHO DXA T-score Classification
See Section 11 for complete classification.
Zarit Burden Interview
See Section 27 for scoring: 0–20 little/none; 21–40 mild-moderate; 41–60 moderate-severe; 61–88 severe.
AGS 2023 Updated Beers Criteria STOPP/START Criteria v3 (2023) Alzheimer's Association Clinical Resources33 Medications Master Table
Cholinesterase Inhibitors & NMDA Antagonists
| Drug | Mechanism | Dose | Indication | Key Side Effects / Pearls |
|---|---|---|---|---|
| Donepezil (Aricept) | Reversible AChE inhibitor | 5 mg daily → 10 mg daily; 23 mg daily (mod-severe) | AD (all stages) | N/V/D, bradycardia, vivid dreams, muscle cramps; take at bedtime |
| Rivastigmine (Exelon) | Pseudo-irreversible AChE + BuChE inhibitor | Patch: 4.6 → 9.5 → 13.3 mg/24h | AD (mild-mod), Parkinson dementia | GI effects less with patch; rotate application site; only AChEI approved for PD dementia |
| Galantamine (Razadyne) | Reversible AChE inhibitor + nicotinic receptor modulator | 4 mg BID → 8 → 12 mg BID; ER: 8 → 16 → 24 mg daily | AD (mild-mod) | GI side effects; adjust dose in hepatic/renal impairment (avoid if severe) |
| Memantine (Namenda) | Non-competitive NMDA receptor antagonist | 5 mg daily → 5 mg BID → 10 mg BID (target 20 mg/day) | AD (mod-severe) | Dizziness, headache, constipation; reduce dose to 5 mg BID if CrCl <30; can combine with AChEI |
Antidepressants in the Elderly
| Drug | Class | Dose Range | Key Features in Elderly |
|---|---|---|---|
| Sertraline (Zoloft) | SSRI | 25–200 mg daily | First-line; well-tolerated; start 25 mg; GI side effects, hyponatremia (SIADH), bleeding risk with anticoagulants |
| Escitalopram (Lexapro) | SSRI | 5–20 mg daily | First-line; clean pharmacokinetic profile; max 20 mg (was 10 mg in ≥65 per FDA, but recent data supports 20 mg with QTc monitoring) |
| Mirtazapine (Remeron) | NaSSA | 7.5–45 mg at bedtime | Promotes appetite and sleep; weight gain desirable in malnourished elderly; more sedating at 7.5–15 mg than at 30–45 mg |
| Bupropion (Wellbutrin) | NDRI | 100–300 mg daily (SR/XL) | Activating; no sexual side effects; no weight gain; lower seizure threshold — avoid if seizure history |
| Duloxetine (Cymbalta) | SNRI | 30–60 mg daily | Useful for comorbid neuropathic pain; avoid if CrCl <30; discontinuation syndrome |
| Venlafaxine (Effexor XR) | SNRI | 37.5–225 mg daily | Dose-dependent HTN at >150 mg; worst discontinuation syndrome of all antidepressants; taper very slowly |
| Nortriptyline (Pamelor) | TCA | 10–75 mg daily | Least anticholinergic TCA; therapeutic window 50–150 ng/mL; ECG before starting (avoid if QTc >450); reserved for refractory cases |
Antipsychotics in the Elderly
| Drug | Dose Range | Key Features |
|---|---|---|
| Quetiapine (Seroquel) | 12.5–200 mg daily | Lowest EPS risk; preferred in DLB/Parkinson; sedating; metabolic effects with chronic use |
| Risperidone (Risperdal) | 0.25–2 mg daily | Best evidence for BPSD agitation; dose-related EPS; stroke risk in dementia |
| Olanzapine (Zyprexa) | 2.5–10 mg daily | IM formulation for acute agitation; weight gain, metabolic syndrome; anticholinergic |
| Haloperidol (Haldol) | 0.5–2 mg PO/IM q4–6h PRN | Gold standard for acute delirium agitation; high EPS/akathisia risk; QTc prolongation (especially IV); AVOID in DLB/Parkinson |
| Pimavanserin (Nuplazid) | 34 mg daily | Selective 5-HT2A inverse agonist; FDA-approved for Parkinson disease psychosis; no dopamine blockade; QTc prolongation |
Bisphosphonates & Bone Agents
| Drug | Mechanism | Dose | Key Pearls |
|---|---|---|---|
| Alendronate (Fosamax) | Osteoclast inhibitor (bisphosphonate) | 70 mg PO weekly | Esophagitis risk; take fasting, upright 30 min; drug holiday after 5 yr if low-risk; avoid if CrCl <35 |
| Zoledronic acid (Reclast) | Osteoclast inhibitor (IV bisphosphonate) | 5 mg IV yearly | Best adherence; acute phase reaction; hydrate before; check Ca/VitD before; avoid if CrCl <35 |
| Denosumab (Prolia) | RANKL monoclonal antibody | 60 mg SQ q6 months | No renal restriction; MUST NOT miss doses (rebound vertebral fractures); transition to bisphosphonate if stopping |
| Teriparatide (Forteo) | Recombinant PTH 1-34 (anabolic) | 20 mcg SQ daily × 2 yr | For severe osteoporosis, multiple fractures, or bisphosphonate failure; must follow with antiresorptive; osteosarcoma risk (box warning) |
| Romosozumab (Evenity) | Sclerostin monoclonal antibody | 210 mg SQ monthly × 12 mo | Greatest BMD increase of any agent; CV black box warning; avoid if MI/stroke within 1 year |
| Calcitonin (Miacalcin) | Inhibits osteoclast activity | 200 IU intranasal daily (alternate nostrils) | Weakest agent; useful for acute vertebral compression fracture pain; nasal irritation; cancer risk with long-term use |
OAB Medications (Non-Anticholinergic Preferred in Elderly)
| Drug | Mechanism | Dose | Pearls |
|---|---|---|---|
| Mirabegron (Myrbetriq) | Beta-3 adrenergic agonist | 25–50 mg daily | NO anticholinergic effects; HTN risk (monitor BP); preferred over oxybutynin in elderly |
| Vibegron (Gemtesa) | Beta-3 adrenergic agonist | 75 mg daily | Similar to mirabegron; less BP effect in studies; no CYP interactions |
| Oxybutynin (Ditropan) | Anticholinergic (M3 antagonist) | 5 mg BID-TID (IR); 5–30 mg daily (ER) | AVOID in elderly (Beers criteria): crosses BBB, cognitive impairment, delirium, dry mouth, constipation, blurred vision |
34 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| ACB | Anticholinergic Cognitive Burden |
| AChEI | Acetylcholinesterase inhibitor |
| ACP | Advance care planning |
| ACS-NSQIP | American College of Surgeons National Surgical Quality Improvement Program |
| AD | Alzheimer disease |
| ADL | Activities of daily living |
| ADR | Adverse drug reaction |
| AGS | American Geriatrics Society |
| ALF | Assisted living facility |
| APS | Adult Protective Services |
| BBS | Berg Balance Scale |
| BMD | Bone mineral density |
| BPPV | Benign paroxysmal positional vertigo |
| BPSD | Behavioral and psychological symptoms of dementia |
| bvFTD | Behavioral variant frontotemporal dementia |
| CAM | Confusion Assessment Method |
| CAUTI | Catheter-associated urinary tract infection |
| CBT-I | Cognitive behavioral therapy for insomnia |
| CDR | Clinical Dementia Rating |
| CFS | Clinical Frailty Scale |
| CGA | Comprehensive geriatric assessment |
| DaTscan | Dopamine transporter SPECT scan |
| DLB | Dementia with Lewy bodies |
| DNR / DNI | Do not resuscitate / do not intubate |
| DTPI | Deep tissue pressure injury |
| DXA | Dual-energy X-ray absorptiometry |
| EASI | Elder Abuse Suspicion Index |
| EWGSOP | European Working Group on Sarcopenia in Older People |
| FAST | Functional Assessment Staging Tool |
| FIM | Functional Independence Measure |
| FRAX | Fracture Risk Assessment Tool |
| FTD | Frontotemporal dementia |
| FTT | Failure to thrive |
| GDS | Geriatric Depression Scale |
| HCP | Healthcare proxy |
| HELP | Hospital Elder Life Program |
| IADL | Instrumental activities of daily living |
| IRF | Inpatient rehabilitation facility |
| KPS | Karnofsky Performance Status |
| LCD | Local Coverage Determination |
| LTACH | Long-term acute care hospital |
| MCI | Mild cognitive impairment |
| MMSE | Mini-Mental State Examination |
| MNA | Mini Nutritional Assessment |
| MoCA | Montreal Cognitive Assessment |
| MOLST | Medical Orders for Life-Sustaining Treatment |
| NIA-AA | National Institute on Aging–Alzheimer's Association |
| NMDA | N-methyl-D-aspartate |
| NMS | Neuroleptic malignant syndrome |
| NPUAP | National Pressure Ulcer Advisory Panel |
| OAB | Overactive bladder |
| PAINAD | Pain Assessment in Advanced Dementia |
| PEG | Percutaneous endoscopic gastrostomy |
| PIM | Potentially inappropriate medication |
| POLST | Physician Orders for Life-Sustaining Treatment |
| PPA | Primary progressive aphasia |
| PPS | Palliative Performance Scale |
| PVR | Post-void residual |
| RANKL | Receptor activator of nuclear factor kappa-B ligand |
| SDH | Subdural hematoma |
| SNF | Skilled nursing facility |
| SPPB | Short Physical Performance Battery |
| STOPP/START | Screening Tool of Older Persons' Prescriptions / Screening Tool to Alert to Right Treatment |
| TUG | Timed Up and Go test |
| VASS | Vulnerability to Abuse Screening Scale |
| ZBI | Zarit Burden Interview |
35 Risk Factors & Comorbidities
Multimorbidity in the Elderly
Approximately 67% of adults ≥65 have ≥2 chronic conditions and 36% have ≥4. The most prevalent chronic conditions in order: hypertension (70%), hyperlipidemia (55%), arthritis (50%), ischemic heart disease (30%), diabetes (27%), CKD (25%), heart failure (15%), COPD (12%), dementia (10%), cancer (various), depression (15–25%). Multimorbidity drives polypharmacy, functional decline, increased healthcare utilization, and mortality. Disease management must be prioritized based on patient values, prognosis, and treatment burden.
Key Risk Factors by Geriatric Syndrome
| Syndrome | Major Risk Factors |
|---|---|
| Falls | Prior falls, gait/balance impairment, medications (≥4 or psychotropics), visual impairment, orthostatic hypotension, cognitive impairment, lower-extremity weakness, neuropathy, environmental hazards, foot problems |
| Delirium | Dementia (single strongest risk factor), age ≥80, severe illness, sensory impairment (vision/hearing), dehydration, malnutrition, polypharmacy (especially anticholinergics, benzodiazepines), urinary catheter, restraints, surgery (especially hip fracture, cardiac) |
| Pressure injuries | Immobility, incontinence, malnutrition (albumin <3.0), sensory impairment, diabetes, PVD, edema, age ≥75, acute illness, cognitive impairment |
| Dementia | Age (strongest), family history (ApoE4 allele for AD), cardiovascular risk factors (HTN, DM, obesity, smoking, hyperlipidemia), low education, traumatic brain injury, depression, social isolation, hearing loss |
| Frailty | Age, sarcopenia, malnutrition, multimorbidity, polypharmacy, depression, social isolation, low physical activity, low income, cognitive impairment |
| Osteoporotic fracture | Prior fracture (2× risk), age, female sex, low BMI, parental hip fracture, smoking, alcohol ≥3 units/day, glucocorticoids, rheumatoid arthritis, falls, low BMD, vitamin D deficiency, early menopause |
| Elder abuse | Cognitive impairment, functional dependence, social isolation, shared living situation, caregiver stress/substance abuse/mental illness, financial dependence of caregiver on elder |
Preventive Care in the Elderly
| Intervention | Recommendation |
|---|---|
| Influenza vaccine | Annually; high-dose (Fluzone HD) or adjuvanted (FLUAD) for ≥65 |
| Pneumococcal vaccine | PCV20 alone OR PCV15 followed by PPSV23 (≥1 year later); for all adults ≥65 |
| Shingles vaccine | Recombinant zoster vaccine (Shingrix) 2 doses, 2–6 months apart; ≥50 years; >90% efficacy (vs 51% for live vaccine) |
| COVID-19 vaccine | Updated formulation annually per CDC guidance |
| Tdap/Td | Tdap once if not previously received; Td booster every 10 years |
| RSV vaccine | Single dose for adults ≥60 through shared clinical decision-making |
| Fall prevention | Screen annually; multifactorial intervention for high-risk |
| Osteoporosis screening | DXA for all women ≥65; men ≥70 or with risk factors |
| AAA screening | One-time US for men 65–75 who ever smoked |
| Statin therapy | Continue in established CVD; initiation in primary prevention after 75 is individualized |
| Aspirin | Do NOT initiate for primary prevention in ≥60 (USPSTF 2022); continue if established CVD indication |