Internal Medicine
Every organ system, diagnosis, classification, medication, risk score, and management strategy — cardiovascular through rheumatology — in one place.
01 Systems-Based Approach
Internal medicine is the parent specialty of adult non-surgical care — its scope spans every organ system, and its intellectual core is the integration of multi-system pathology in a single patient. The internist's daily work is diagnosis under uncertainty, multi-drug optimization, and the longitudinal management of chronic disease superimposed on acute illness. Unlike organ-based subspecialties, general internal medicine owns the whole patient, which means the differential diagnosis list is always broad before it is narrow.
The Organ-System Framework
| System | Core Diagnoses Managed by IM | Key Diagnostic Anchors |
|---|---|---|
| Cardiovascular | HTN, HF, CAD, AFib, VTE, valvular disease | ECG, troponin, BNP, echo |
| Pulmonary | COPD, asthma, pneumonia, PE, ILD, pleural effusion | PFTs, CXR, CT chest, ABG |
| GI / Hepatology | GERD, PUD, IBD, cirrhosis, pancreatitis, GI bleed | LFTs, lipase, endoscopy, imaging |
| Nephrology | AKI, CKD, electrolyte disorders, glomerulonephritis | Cr, BUN, UA, electrolytes, biopsy |
| Endocrinology | DM (types 1/2), thyroid, adrenal, calcium/bone | A1C, TSH, cortisol, calcium/PTH |
| Hematology | Anemia, coagulopathy, thrombocytopenia, VTE | CBC, smear, iron studies, coags |
| Infectious Disease | Sepsis, pneumonia, UTI, endocarditis, HIV, C. diff | Cultures, procalcitonin, imaging |
| Rheumatology | RA, SLE, gout, vasculitis, sarcoidosis | ANA, RF, CCP, uric acid, ESR/CRP |
02 The Internal Medicine Physical Exam
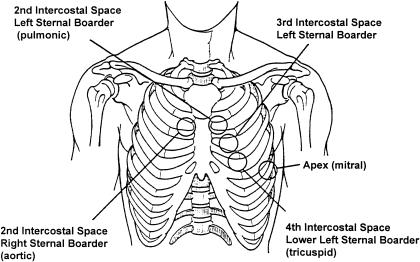
The IM exam is the most comprehensive in medicine — a full head-to-toe assessment on admission, then focused daily exams targeting active problems. Key elements that change management daily: volume status, cardiopulmonary findings, abdominal exam, and mental status.
Volume Status Assessment
| Finding | Hypovolemia | Euvolemia | Hypervolemia |
|---|---|---|---|
| JVP | Flat, < 6 cm H₂O | 6–8 cm H₂O | Elevated > 8 cm, hepatojugular reflux + |
| Mucous membranes | Dry, tacky | Moist | Moist |
| Skin turgor | Tenting (less reliable in elderly) | Normal | Taut, shiny |
| Edema | Absent | Absent | Pitting (grade 1–4+) |
| Orthostatics | SBP drop ≥ 20 or HR rise ≥ 20 on standing | Normal | Usually normal |
| Urine output | < 0.5 mL/kg/hr | 0.5–1.0 mL/kg/hr | Variable |
Cardiac Auscultation — Murmur Grading (Levine Scale)
| Grade | Description |
|---|---|
| I/VI | Barely audible, heard only in a quiet room with concentration |
| II/VI | Soft but readily audible |
| III/VI | Moderately loud, no thrill |
| IV/VI | Loud with palpable thrill |
| V/VI | Very loud, thrill present, audible with stethoscope partially off chest |
| VI/VI | Audible without stethoscope |
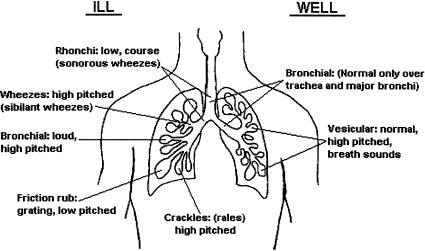
Lung Auscultation Findings
| Sound | Character | Implies |
|---|---|---|
| Crackles (rales) | Discontinuous, inspiratory, fine or coarse | Fine = fibrosis, early CHF; Coarse = pneumonia, pulmonary edema |
| Wheezes | High-pitched, musical, expiratory | Bronchospasm (asthma, COPD), CHF ("cardiac asthma") |
| Rhonchi | Low-pitched, rumbling, clears with cough | Secretions in large airways |
| Stridor | High-pitched, inspiratory, monophonic | Upper airway obstruction — emergency |
| Diminished/absent | Reduced air entry | Effusion, pneumothorax, severe COPD, obesity |
| Egophony ("E to A") | Spoken "E" sounds like "A" | Consolidation (pneumonia) above effusion |
03 Key Terminology
| Term | Definition / Clinical Relevance |
|---|---|
| Preload | Volume/pressure stretching the ventricle at end-diastole (approximated by LVEDP, CVP). Elevated in volume overload; reduced in hemorrhage/dehydration. |
| Afterload | Resistance the ventricle must overcome to eject blood (approximated by SVR, MAP). Elevated in HTN, aortic stenosis. Vasodilators reduce afterload. |
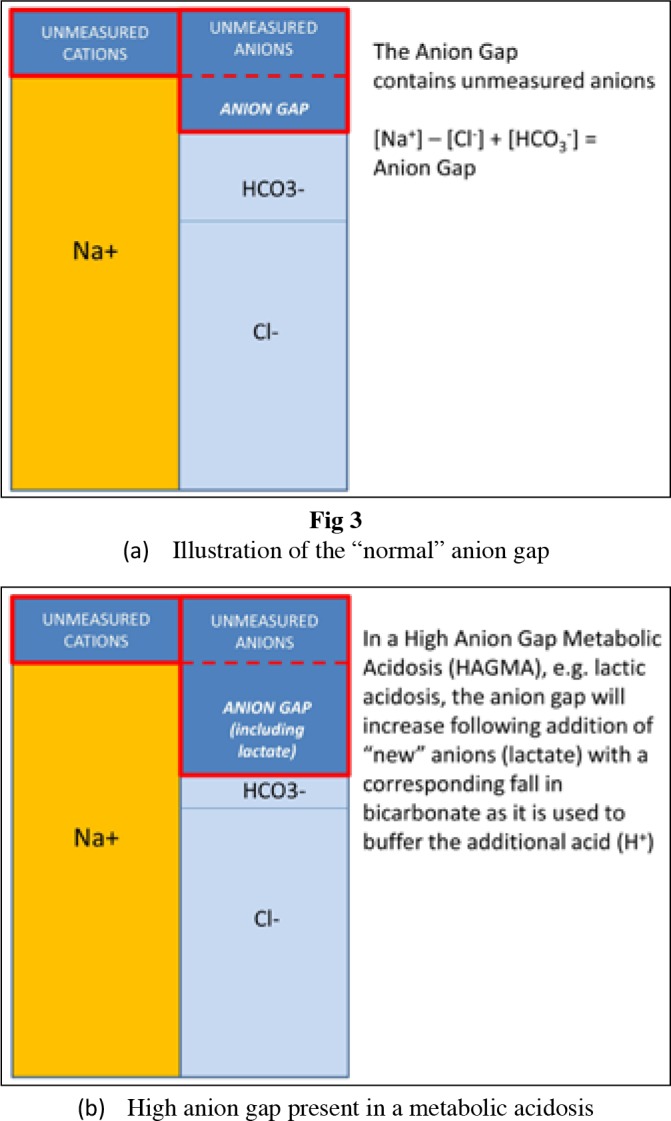
| Anion gap | Na − (Cl + HCO₃). Normal 8–12. Elevated = unmeasured acid accumulation (MUDPILES: Methanol, Uremia, DKA, Propylene glycol, INH/Iron, Lactic acidosis, Ethylene glycol, Salicylates). |
| GFR | Volume of plasma filtered by the glomeruli per minute. Gold standard for kidney function. Normal ~120 mL/min/1.73 m². Estimated by CKD-EPI equation (preferred over MDRD). |
| A-a gradient | PAO₂ − PaO₂. Normal = 2.5 + (0.21 × age). Elevated in V/Q mismatch, shunt, diffusion impairment. Normal in hypoventilation. |
| Sensitivity / Specificity | Sensitivity = true positives / (TP + FN) — rules OUT when negative (SnNOut). Specificity = TN / (TN + FP) — rules IN when positive (SpPIn). |
| NNT / NNH | Number needed to treat (benefit one patient) and number needed to harm. NNT = 1/ARR. Lower NNT = more effective intervention. |
04 Hypertension Cardiovascular
Hypertension affects ~47% of US adults under the 2017 ACC/AHA guideline thresholds. It is the single largest modifiable risk factor for stroke, MI, HF, CKD, and aortic dissection. Most cases (90–95%) are primary (essential) — no identifiable cause. Secondary causes must be considered in resistant HTN, onset < 30 years, or abrupt worsening.
Classification (ACC/AHA 2017)
| Category | SBP (mmHg) | DBP (mmHg) | Action |
|---|---|---|---|
| Normal | < 120 | < 80 | Reassess annually |
| Elevated | 120–129 | < 80 | Lifestyle modification |
| Stage 1 HTN | 130–139 | 80–89 | Lifestyle + meds if ASCVD risk ≥ 10% |
| Stage 2 HTN | ≥ 140 | ≥ 90 | Lifestyle + medication (usually 2 agents) |
| Hypertensive crisis | > 180 | > 120 | Urgency (no organ damage) vs emergency (end-organ damage — treat IV) |
Secondary Causes of HTN
| Cause | Clue | Workup |
|---|---|---|
| Renal artery stenosis | Refractory HTN, flash pulmonary edema, renal bruit, Cr rise with ACEI | Renal duplex, CTA/MRA |
| Primary aldosteronism | Hypokalemia, resistant HTN, adrenal incidentaloma | Aldosterone/renin ratio (> 30 with aldo > 15) |
| Pheochromocytoma | Paroxysmal HTN, headache, palpitations, diaphoresis (triad) | 24-hr urine metanephrines or plasma free metanephrines |
| Cushing syndrome | Central obesity, striae, moon facies, hyperglycemia | 24-hr urine cortisol, overnight dexamethasone suppression, late-night salivary cortisol |
| Coarctation of aorta | Young patient, upper > lower extremity BP, rib notching on CXR | Echo, CT/MRA |
| OSA | Snoring, daytime somnolence, obesity, resistant HTN | Polysomnography (AHI ≥ 5) |
First-Line Agents
| Class | Examples | Preferred Indication | Key Pearl |
|---|---|---|---|
| ACE inhibitors | Lisinopril 10–40 mg, enalapril 5–20 mg BID | DM with proteinuria, HFrEF, post-MI, CKD | Monitor K+ and Cr; cough in ~10% (switch to ARB) |
| ARBs | Losartan 50–100 mg, valsartan 80–320 mg | Same as ACEI (ACEI-intolerant) | Do NOT combine with ACEI (hyperkalemia, no benefit) |
| CCBs (dihydropyridine) | Amlodipine 5–10 mg, nifedipine ER | Elderly, black patients, isolated systolic HTN | Peripheral edema is dose-dependent, not allergic |
| Thiazide diuretics | Chlorthalidone 12.5–25 mg, HCTZ 25 mg | Elderly, black patients, osteoporosis | Chlorthalidone preferred (longer half-life, better outcomes data); hypoK+, hypoNa+ |
Target: reduce MAP by no more than 25% in the first hour, then to 160/100 over the next 2–6 hours. Avoid precipitous drops (risk of watershed stroke). IV agents: nicardipine (5–15 mg/hr, titratable), labetalol (20 mg IV bolus then drip), nitroprusside (0.25–10 mcg/kg/min — cyanide toxicity risk with prolonged use). Exception: aortic dissection — target HR < 60 and SBP < 120 rapidly with IV esmolol or labetalol before vasodilators.
05 Heart Failure Cardiovascular
Heart failure is a clinical syndrome of dyspnea, fatigue, and volume overload resulting from structural or functional cardiac impairment. Prevalence: ~6.7 million US adults. Five-year mortality: ~50%. HF is the #1 cause of hospitalization in adults > 65.
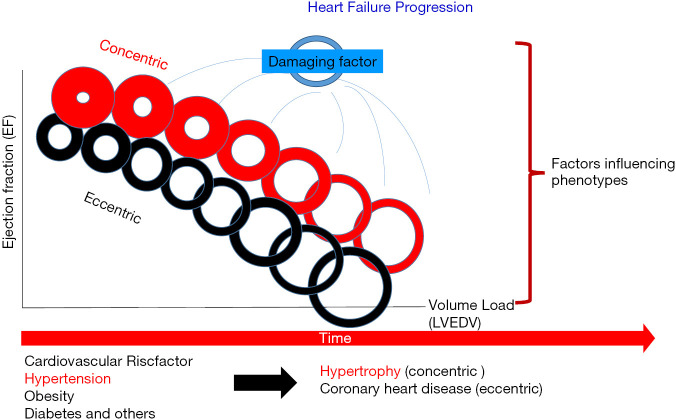
Classification by Ejection Fraction
| Category | EF | Key Feature | Evidence-Based Therapies |
|---|---|---|---|
| HFrEF (reduced) | ≤ 40% | Systolic dysfunction, dilated LV | GDMT: ACEI/ARB/ARNI + BB + MRA + SGLT2i (the "four pillars") |
| HFmrEF (mildly reduced) | 41–49% | Intermediate — may respond to HFrEF therapies | SGLT2i, consider ARNI/BB/MRA |
| HFpEF (preserved) | ≥ 50% | Diastolic dysfunction, stiff LV, normal size | SGLT2i (EMPEROR-Preserved, DELIVER), diuretics, BP/rate control |
NYHA Functional Classification
| Class | Symptoms |
|---|---|
| I | No limitation of physical activity; ordinary activity does not cause symptoms |
| II | Slight limitation; comfortable at rest, ordinary activity causes fatigue/dyspnea/palpitations |
| III | Marked limitation; comfortable at rest, less-than-ordinary activity causes symptoms |
| IV | Unable to carry on any physical activity without symptoms; symptoms at rest |
ACC/AHA Stages
| Stage | Description | Management |
|---|---|---|
| A | At risk (HTN, DM, CAD) but no structural disease or symptoms | Risk factor control, ACEI if appropriate |
| B | Structural heart disease (LVH, prior MI, low EF) but no symptoms | All Stage A + ACEI/ARB + beta-blocker |
| C | Structural disease with current or prior HF symptoms | Full GDMT, devices (ICD, CRT), diuretics |
| D | Refractory HF requiring advanced therapies | LVAD, transplant, inotropes, palliative care |
The Four Pillars of HFrEF (2022 AHA/ACC/HFSA Guideline)
All four should be initiated and uptitrated as tolerated — each independently reduces mortality (Heidenreich et al., 2022):
| Pillar | Drug | Target Dose | Mortality Reduction |
|---|---|---|---|
| ARNI (or ACEI/ARB) | Sacubitril/valsartan (Entresto) | 97/103 mg BID | ~20% (PARADIGM-HF) |
| Beta-blocker | Carvedilol, metoprolol succinate, bisoprolol | Carvedilol 25 mg BID (50 if >85 kg) | ~34% (MERIT-HF, COPERNICUS) |
| MRA | Spironolactone 25–50 mg or eplerenone 50 mg | Spironolactone 50 mg daily | ~30% (RALES) |
| SGLT2 inhibitor | Dapagliflozin 10 mg or empagliflozin 10 mg | 10 mg daily (no titration) | ~17% (DAPA-HF, EMPEROR-Reduced) |
Presentation: Acute dyspnea, orthopnea, PND, crackles, S3 gallop, elevated JVP, peripheral edema, weight gain. BNP > 400 pg/mL (or NT-proBNP > 900 age-adjusted) strongly supports diagnosis. Management: IV furosemide (double home dose or 40 mg if diuretic-naive, target UOP 100–150 mL/hr), upright positioning, supplemental O₂/NIPPV if SpO₂ < 90%, hold beta-blocker if hypotensive or in cardiogenic shock. Vasodilators (IV nitroglycerin) if SBP > 100. Daily weights and strict I&O. Transition to oral diuretics when euvolemic.
06 Coronary Artery Disease Cardiovascular
CAD is atherosclerotic narrowing of the coronary arteries — the #1 cause of death worldwide. Manifests as stable angina (supply-demand mismatch with exertion), unstable angina (rest pain, new-onset, crescendo), NSTEMI (troponin elevation without ST elevation), or STEMI (transmural ischemia with ST elevation — door-to-balloon < 90 minutes).
Acute Coronary Syndrome Spectrum
| Entity | Troponin | ECG | Pathology | Management |
|---|---|---|---|---|
| Stable angina | Normal | ST depression with exercise | Fixed stenosis ≥ 70% | Anti-anginals, risk factor modification, stress test |
| Unstable angina | Normal | ST depression, T-wave inversion, or normal | Plaque rupture, non-occlusive thrombus | Antiplatelet, anticoagulant, invasive strategy |
| NSTEMI | Elevated | ST depression, T-wave inversion | Partial occlusion with myocyte necrosis | Same as UA + urgent cath (< 24 hr if high risk) |
| STEMI | Elevated | ≥ 1 mm ST elevation in 2+ contiguous leads | Complete occlusion (transmural infarct) | Emergent PCI (door-to-balloon < 90 min) |
Post-MI GDMT
All patients without contraindication: DAPT (ASA 81 mg + ticagrelor 90 mg BID or clopidogrel 75 mg daily × 12 months), high-intensity statin (atorvastatin 80 mg), beta-blocker (metoprolol succinate, carvedilol), ACEI/ARB (especially if EF ≤ 40%, anterior MI, or DM). Cardiac rehab referral is a Class I recommendation (Virani et al., 2021).
07 Atrial Fibrillation Cardiovascular
AFib is the most common sustained arrhythmia — irregular, chaotic atrial activity replacing organized P waves, producing an "irregularly irregular" ventricular response. Prevalence: ~5 million US adults, rising with age (10% of those > 80). Key risks: 5× stroke risk, 2× mortality, HF progression.
Classification
| Type | Duration | Terminates Spontaneously? |
|---|---|---|
| Paroxysmal | ≤ 7 days (usually < 48 hr) | Yes |
| Persistent | > 7 days | No — requires cardioversion |
| Long-standing persistent | > 12 months | No, but rhythm control still considered |
| Permanent | Indefinite (accepted by patient/clinician) | Rate control strategy only |
| Valvular | Associated with moderate–severe mitral stenosis or mechanical heart valve | Requires warfarin (DOACs not approved) |
CHA₂DS₂-VASc Score (Stroke Risk)
| Factor | Points |
|---|---|
| CHF (or LVEF ≤ 40%) | 1 |
| Hypertension | 1 |
| Age ≥ 75 | 2 |
| Diabetes | 1 |
| Stroke/TIA/VTE history | 2 |
| Vascular disease (MI, PAD, aortic plaque) | 1 |
| Age 65–74 | 1 |
| Sex category (female) | 1 |
Score ≥ 2 in men or ≥ 3 in women: anticoagulate. Score 1 in men / 2 in women: consider anticoagulation. Score 0 in men / 1 in women: no anticoagulation needed.
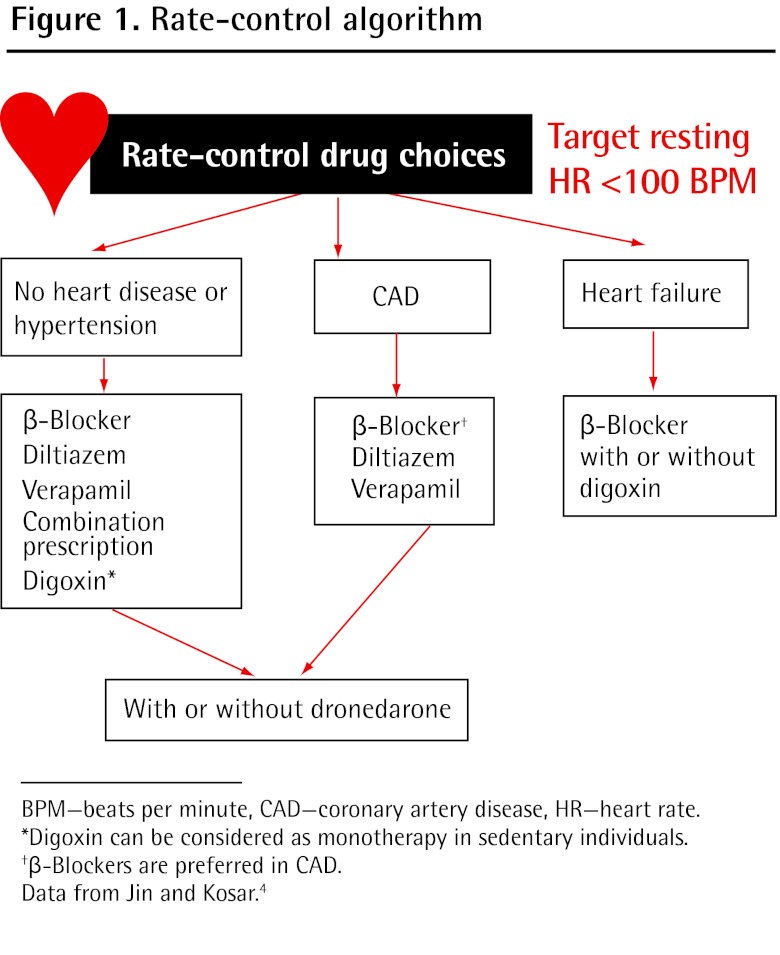
Rate vs Rhythm Control
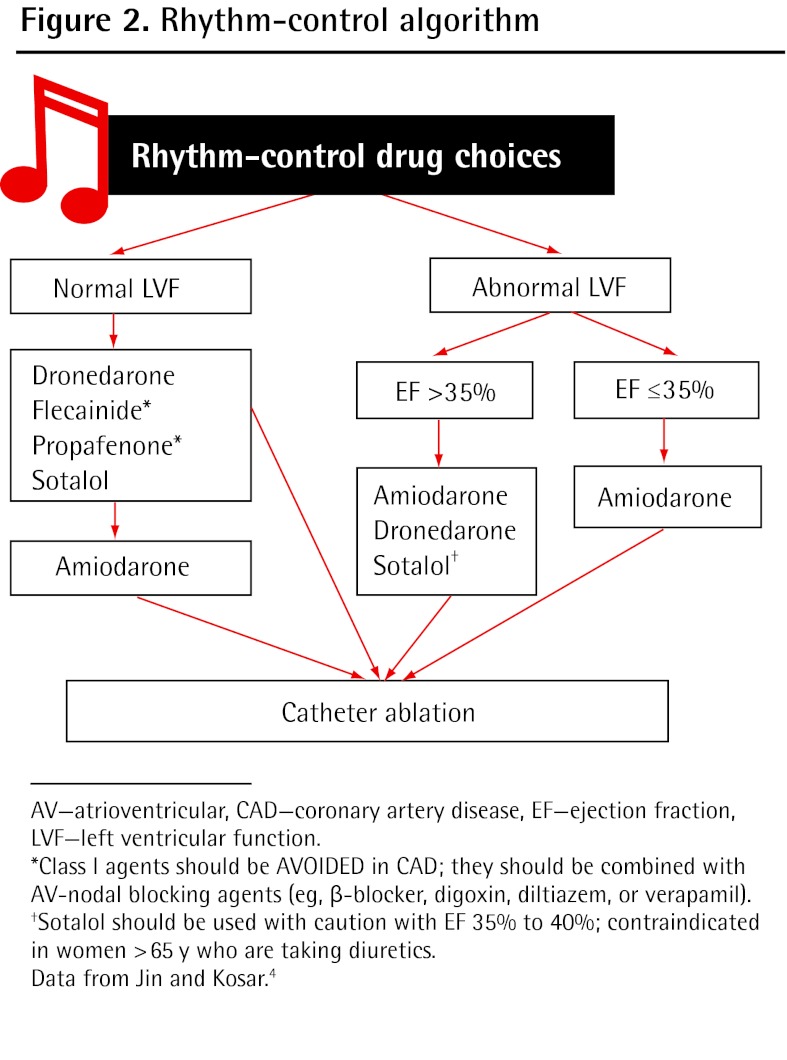
Rate control: Target resting HR < 110 bpm (lenient) or < 80 bpm (strict — no proven benefit over lenient per RACE II). Agents: beta-blockers (metoprolol, carvedilol), non-DHP CCBs (diltiazem, verapamil — avoid in HFrEF), digoxin (adjunct, narrow therapeutic window 0.5–0.9 ng/mL). Rhythm control: Favored in symptomatic patients, early AFib, younger patients — EAST-AFNET 4 showed early rhythm control reduces CV outcomes (Kirchhof et al., 2020). Agents: flecainide (no structural heart disease), amiodarone (any substrate, but toxicity profile), sotalol, dofetilide (requires inpatient initiation with QTc monitoring). Catheter ablation (pulmonary vein isolation) for symptomatic drug-refractory AFib.
08 Venous Thromboembolism (DVT/PE) Cardiovascular
VTE encompasses DVT and PE — a single disease continuum. DVT: most commonly in the deep veins of the lower extremities (iliac, femoral, popliteal). PE: thrombus embolizes to pulmonary vasculature — can be fatal. Virchow's triad: stasis, endothelial injury, hypercoagulability.
Wells Score — DVT
| Criterion | Points |
|---|---|
| Active cancer (treatment within 6 months) | 1 |
| Paralysis/paresis/recent immobilization of lower extremity | 1 |
| Bedridden > 3 days or major surgery within 12 weeks | 1 |
| Localized tenderness along deep venous system | 1 |
| Entire leg swollen | 1 |
| Calf swelling > 3 cm vs asymptomatic side | 1 |
| Pitting edema confined to symptomatic leg | 1 |
| Collateral superficial veins (non-varicose) | 1 |
| Alternative diagnosis as likely or more likely | −2 |
≥ 2 points = DVT likely → ultrasound. < 2 points = DVT unlikely → D-dimer first (if negative, DVT excluded).
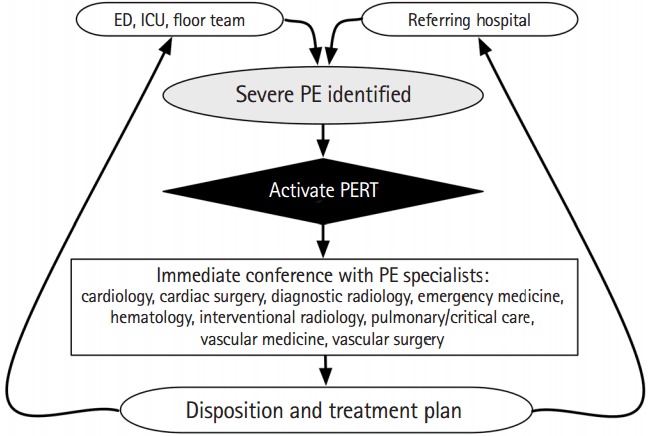
PE Severity & Management
| Category | Hemodynamics | RV Dysfunction | Troponin | Treatment |
|---|---|---|---|---|
| Low-risk | Stable | No | Normal | Anticoagulation alone (DOAC preferred) |
| Submassive | Stable | Yes (echo/CT) | Often elevated | Anticoagulation ± catheter-directed therapy; monitor in ICU |
| Massive | Hypotensive (SBP < 90) | Yes | Elevated | Systemic thrombolysis (tPA 100 mg over 2 hr), surgical/catheter embolectomy |
Anticoagulation for VTE
First-line: DOACs — apixaban (10 mg BID × 7 days, then 5 mg BID) or rivaroxaban (15 mg BID × 21 days, then 20 mg daily). Alternative: LMWH bridge to warfarin (target INR 2–3). Duration: provoked VTE = 3 months; unprovoked = at least 3 months then reassess (consider extended if low bleeding risk); cancer-associated = LMWH or DOAC long-term.
09 COPD Pulmonary
COPD is a chronic, progressive airflow limitation that is not fully reversible — caused by significant exposure to noxious particles (cigarette smoke in > 80% of cases). It encompasses chronic bronchitis (productive cough ≥ 3 months/year for 2 consecutive years) and emphysema (destruction of alveolar walls, loss of elastic recoil, air trapping). Third leading cause of death worldwide.
Diagnosis
Spirometry is required: post-bronchodilator FEV₁/FVC < 0.70 confirms airflow obstruction. Severity is graded by FEV₁:
| GOLD Stage | FEV₁ (% predicted) | Severity |
|---|---|---|
| 1 | ≥ 80% | Mild |
| 2 | 50–79% | Moderate |
| 3 | 30–49% | Severe |
| 4 | < 30% | Very severe |
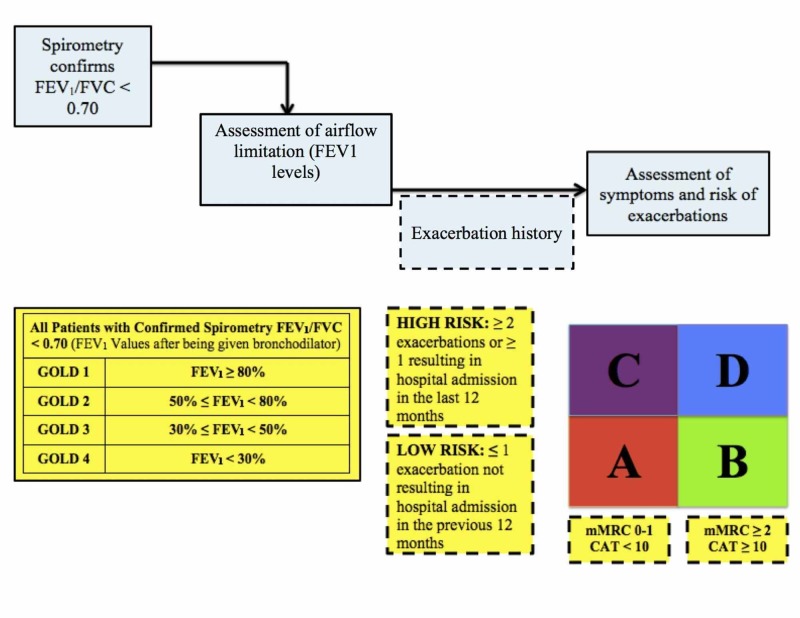
GOLD ABE Assessment (2023 Update)
| Group | Exacerbation History | Symptoms (mMRC/CAT) | Initial Therapy |
|---|---|---|---|
| A | 0–1 moderate (no hospitalization) | Low (mMRC 0–1, CAT < 10) | Bronchodilator (SABA or LABA or LAMA) |
| B | 0–1 moderate (no hospitalization) | High (mMRC ≥ 2, CAT ≥ 10) | LABA + LAMA |
| E | ≥ 2 moderate OR ≥ 1 hospitalization | Any | LABA + LAMA (add ICS if eos ≥ 300) |
Triggered by viral/bacterial infection, pollution, or non-adherence. Treatment: SABA (albuterol nebulizer q4–6h), systemic corticosteroids (prednisone 40 mg daily × 5 days — REDUCE trial showed 5 days = 14 days), antibiotics (if purulent sputum or severe exacerbation — azithromycin 500 mg then 250 mg × 4 days, or doxycycline, or amoxicillin-clavulanate). NIPPV (BiPAP) reduces intubation and mortality in acute hypercapnic respiratory failure (pH < 7.35, PaCO₂ > 45). Supplemental O₂ target: SpO₂ 88–92% — avoid hyperoxia (suppresses hypoxic drive, worsens CO₂ retention).
10 Asthma Pulmonary
Asthma is reversible airflow obstruction due to airway hyperresponsiveness, chronic inflammation, and mucus hypersecretion. Unlike COPD, spirometry shows improvement post-bronchodilator (≥ 12% and ≥ 200 mL increase in FEV₁). Peak incidence in childhood but can present at any age. Type 2 (eosinophilic) inflammation drives most cases — IL-4, IL-5, IL-13 pathway.
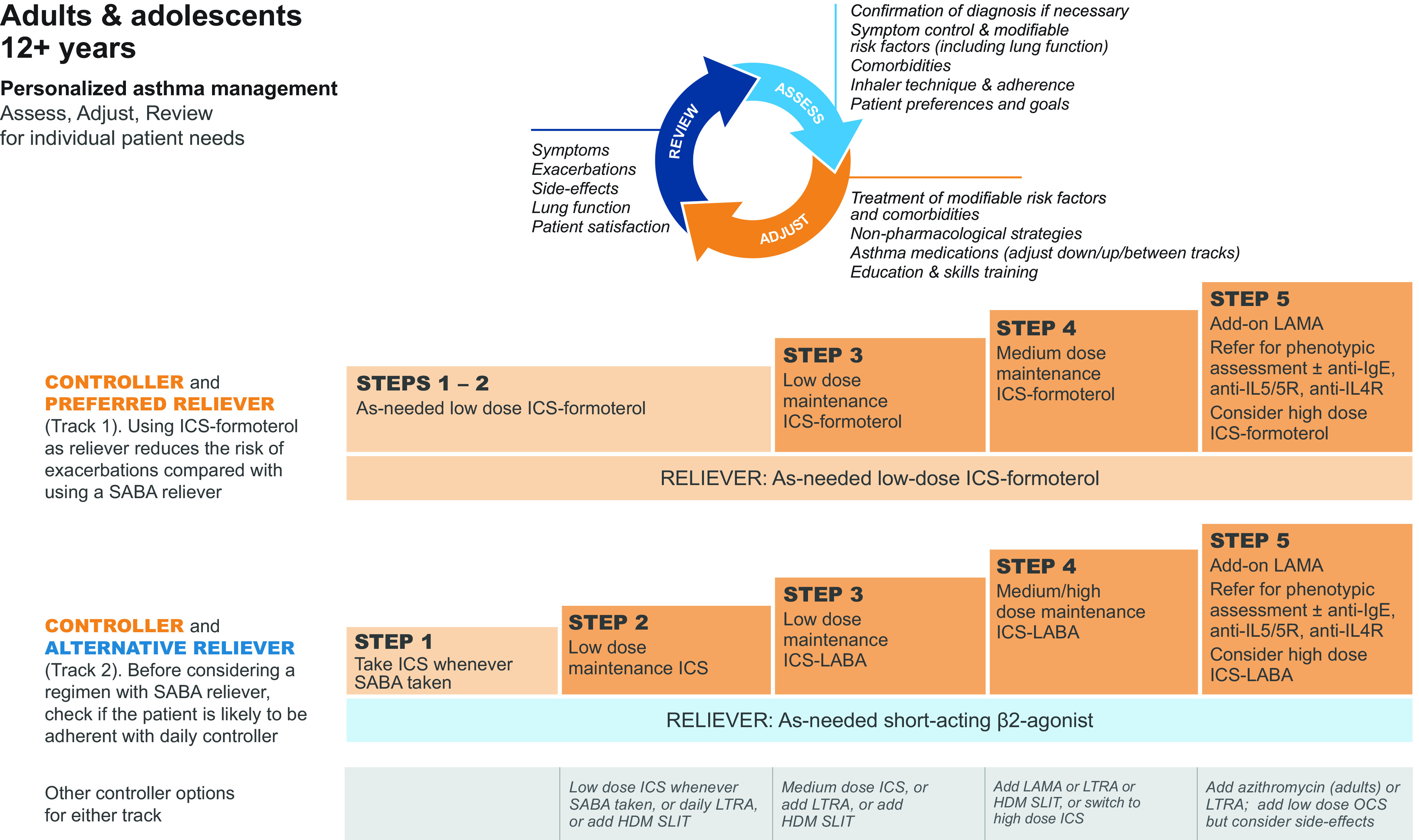
Stepwise Therapy (GINA 2023)
| Step | Preferred Controller | Reliever |
|---|---|---|
| 1 | Low-dose ICS-formoterol PRN | ICS-formoterol PRN |
| 2 | Low-dose ICS daily (or ICS-formoterol PRN) | ICS-formoterol PRN |
| 3 | Low-dose ICS-LABA (budesonide/formoterol) | ICS-formoterol PRN |
| 4 | Medium-dose ICS-LABA | ICS-formoterol PRN |
| 5 | High-dose ICS-LABA + add-on (tiotropium, anti-IL5, anti-IgE, anti-IL4R) | ICS-formoterol PRN |
Life-threatening asthma not responding to standard bronchodilators. Management: continuous albuterol nebulization, ipratropium bromide 0.5 mg q20min × 3, IV methylprednisolone 125 mg, IV magnesium sulfate 2 g over 20 min (smooth muscle relaxation). If impending respiratory failure (silent chest, declining mental status, PaCO₂ rising): intubation with ketamine (bronchodilatory) for induction, low tidal volume/low rate ventilation (permissive hypercapnia — avoid dynamic hyperinflation).
11 Pneumonia Pulmonary
Pneumonia is infection of the lung parenchyma causing consolidation. Leading infectious cause of death globally. Classification determines empiric antibiotic choice:
| Type | Common Organisms | Empiric Treatment |
|---|---|---|
| Community-acquired (CAP) — outpatient, no comorbidities | S. pneumoniae, M. pneumoniae, H. influenzae, respiratory viruses | Amoxicillin 1 g TID or doxycycline 100 mg BID |
| CAP — outpatient with comorbidities | Same + S. aureus, gram-negatives | Amoxicillin-clavulanate + macrolide, or respiratory fluoroquinolone (levofloxacin 750 mg) |
| CAP — inpatient (non-ICU) | Same spectrum | Beta-lactam (ceftriaxone 2 g IV, or ampicillin-sulbactam) + macrolide (azithromycin), or respiratory FQ alone |
| CAP — ICU | Add Legionella, S. aureus | Beta-lactam + macrolide (both required); add vancomycin if MRSA risk |
| Hospital-acquired (HAP) / VAP | Pseudomonas, MRSA, Acinetobacter, ESBL gram-negatives | Anti-pseudomonal beta-lactam (piperacillin-tazobactam, cefepime, or meropenem) ± vancomycin/linezolid if MRSA risk |
Severity Assessment — CURB-65
| Criterion | Points |
|---|---|
| Confusion (new) | 1 |
| Urea > 7 mmol/L (BUN > 19) | 1 |
| Respiratory rate ≥ 30 | 1 |
| Blood pressure (SBP < 90 or DBP ≤ 60) | 1 |
| Age ≥ 65 | 1 |
0–1: outpatient. 2: consider short inpatient stay. 3–5: inpatient, 4–5 consider ICU.
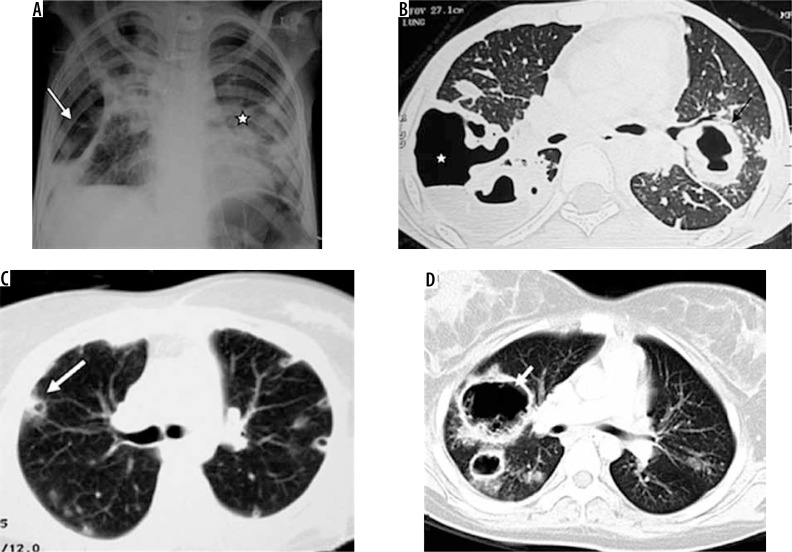
12 Interstitial Lung Disease & Pleural Disease Pulmonary
ILD is a group of disorders causing progressive fibrosis or inflammation of the lung interstitium. PFTs show a restrictive pattern: reduced FVC and TLC with preserved or elevated FEV₁/FVC ratio. DLCO is characteristically reduced (impaired gas diffusion across thickened alveolar membrane). HRCT is the key diagnostic imaging modality.
Major ILD Categories
| Category | Examples | Key Features |
|---|---|---|
| Idiopathic pulmonary fibrosis (IPF) | — | UIP pattern on HRCT (basal-predominant honeycombing, traction bronchiectasis). Median survival 3–5 yr. Antifibrotics: pirfenidone (Esbriet), nintedanib (Ofev) |
| Connective tissue disease-ILD | RA-ILD, SSc-ILD, myositis-ILD | Screen all CTD patients with PFTs + HRCT. Treatment: immunosuppression + antifibrotics |
| Hypersensitivity pneumonitis | Bird fancier's, farmer's lung | Antigen exposure history critical. Remove exposure. Acute: corticosteroids. Chronic: may progress to fibrosis |
| Sarcoidosis-related | — | Non-caseating granulomas. Bilateral hilar lymphadenopathy. See Section 36 |
| Drug-induced | Methotrexate, amiodarone, bleomycin, nitrofurantoin | Temporal relation to drug initiation. Withdraw offending agent |
Pleural Effusion — Light's Criteria
Thoracentesis fluid is exudative if any one criterion is met (sensitivity 98%, specificity 83%):
| Criterion | Exudative Threshold |
|---|---|
| Pleural protein / serum protein | > 0.5 |
| Pleural LDH / serum LDH | > 0.6 |
| Pleural LDH | > 2/3 upper limit of normal serum LDH |
Common transudates: CHF (most common overall), cirrhosis (hepatic hydrothorax), nephrotic syndrome. Common exudates: pneumonia (parapneumonic), malignancy, PE, TB, autoimmune.
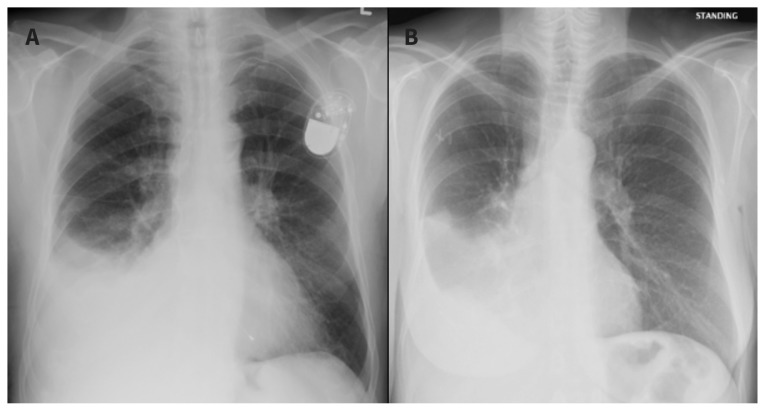
13 GERD & Peptic Ulcer Disease GI
GERD: reflux of gastric acid into the esophagus causing heartburn, regurgitation, and risk of erosive esophagitis, stricture, Barrett's esophagus (intestinal metaplasia — premalignant, requires surveillance EGD). Diagnosis: clinical for typical symptoms; pH monitoring or impedance testing for refractory cases. Treatment: lifestyle (elevate HOB, avoid eating 3 hr before bed, weight loss) + PPI (omeprazole 20 mg, pantoprazole 40 mg daily). Step down to H2RA if controlled. Long-term PPI risks (generally low but real): C. diff, hypomagnesemia, osteoporosis, B12 deficiency, AIN.
PUD: mucosal ulceration of the stomach or duodenum. Two dominant causes: H. pylori infection (~60% duodenal, ~40% gastric) and NSAIDs. Diagnosis: EGD (gold standard) — biopsy gastric ulcers to rule out malignancy. H. pylori testing: stool antigen (preferred), urea breath test, or biopsy-based testing. Treatment: PPI BID + triple therapy (clarithromycin 500 mg BID + amoxicillin 1 g BID × 14 days) or bismuth quadruple therapy if clarithromycin resistance is suspected (> 15% local rate).
14 Inflammatory Bowel Disease GI
IBD comprises two chronic idiopathic inflammatory disorders of the GI tract. Bimodal age onset: 15–30 and 50–70.
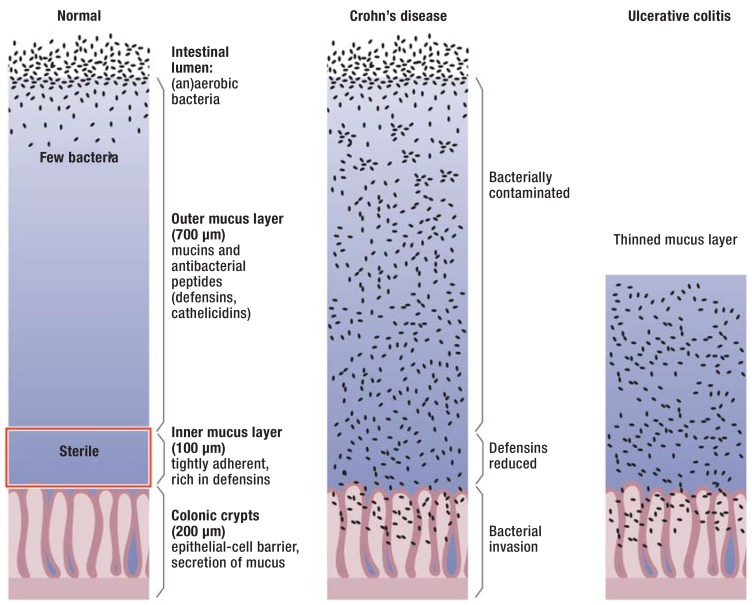
| Feature | Crohn's Disease | Ulcerative Colitis |
|---|---|---|
| Location | Mouth to anus (terminal ileum most common) | Rectum → proximal colon (continuous) |
| Depth | Transmural (full-thickness) | Mucosa/submucosa only |
| Pattern | Skip lesions, cobblestoning | Continuous, no skip lesions |
| Histology | Non-caseating granulomas (pathognomonic) | Crypt abscesses, pseudopolyps |
| Complications | Fistulae, strictures, abscesses, B12 deficiency (ileal disease) | Toxic megacolon, colorectal cancer risk |
| Surgery | Not curative (recurrence at anastomosis) | Total proctocolectomy is curative |
| Smoking | Worsens disease | Protective (paradoxically) |
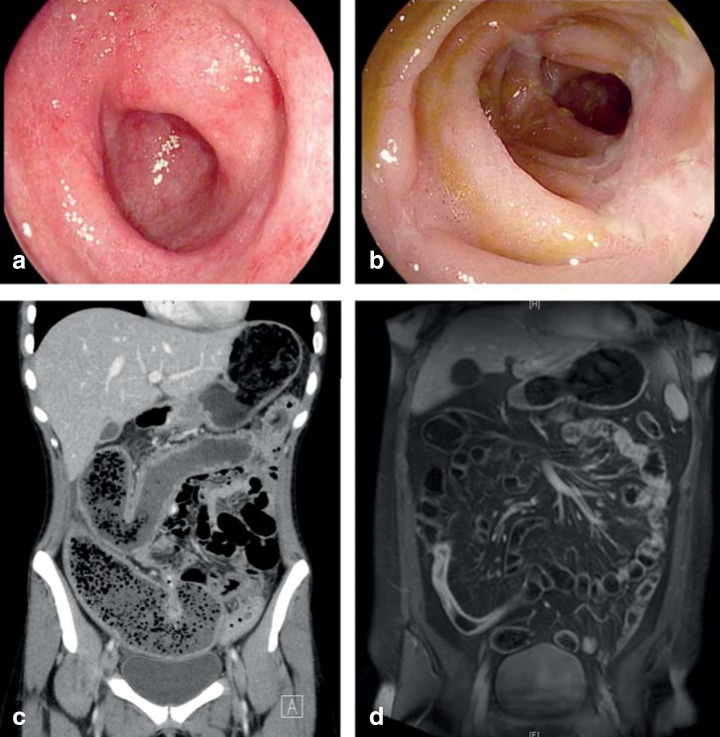
Treatment Ladder
Mild: 5-ASA (mesalamine — UC only, not effective in Crohn's), budesonide (ileal/right-sided Crohn's). Moderate-severe: corticosteroids for induction only (prednisone 40–60 mg taper over 8–12 weeks — never maintenance), immunomodulators (azathioprine/6-MP, methotrexate for Crohn's). Moderate-severe or steroid-refractory: biologics — anti-TNF (infliximab, adalimumab), anti-integrin (vedolizumab — gut-selective), anti-IL-12/23 (ustekinumab), JAK inhibitors (tofacitinib — UC only). Screen for TB and hepatitis B before starting biologics.
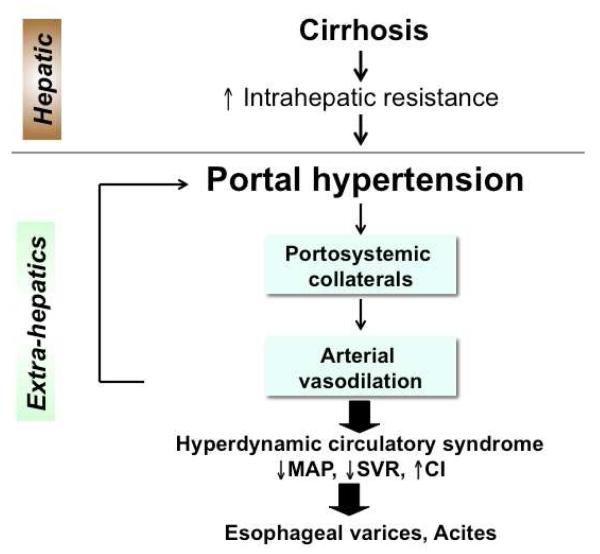
15 Liver Disease & Cirrhosis GI
Cirrhosis is the end stage of chronic liver injury — replacement of functional hepatocytes with fibrosis and regenerative nodules. Common causes: alcohol (60–70%), NAFLD/NASH (rising rapidly, now called MASLD/MASH), hepatitis C, hepatitis B, autoimmune hepatitis, hemochromatosis, Wilson disease, alpha-1 antitrypsin deficiency, PBC, PSC.
Child-Pugh Score
| Parameter | 1 Point | 2 Points | 3 Points |
|---|---|---|---|
| Bilirubin (mg/dL) | < 2 | 2–3 | > 3 |
| Albumin (g/dL) | > 3.5 | 2.8–3.5 | < 2.8 |
| INR | < 1.7 | 1.7–2.3 | > 2.3 |
| Ascites | None | Slight (controlled) | Moderate-severe (refractory) |
| Encephalopathy | None | Grade I–II | Grade III–IV |
Class A (5–6): well-compensated, 1-year survival ~100%. Class B (7–9): significant functional compromise, 1-year survival ~80%. Class C (10–15): decompensated, 1-year survival ~45%.
MELD-Na Score
Used for organ allocation in liver transplantation. Variables: bilirubin, INR, creatinine, sodium. Range 6–40. MELD ≥ 15 = consider transplant listing. MELD ≥ 30 = high 3-month mortality (> 50%).
Complications of Cirrhosis
| Complication | Pathophysiology | Management |
|---|---|---|
| Ascites | Portal HTN + low albumin + splanchnic vasodilation | Na restriction (< 2 g/day), spironolactone 100 mg + furosemide 40 mg (100:40 ratio), paracentesis, TIPS for refractory |
| SBP | Translocation of gut bacteria into ascitic fluid | Diagnostic paracentesis (PMN ≥ 250 = SBP). Treat: cefotaxime 2 g IV q8h. Prophylaxis: ciprofloxacin or TMP-SMX if prior SBP or GI bleed |
| Variceal bleeding | Portal HTN → portosystemic collaterals → esophageal/gastric varices | Primary prophylaxis: NSBB (propranolol, carvedilol) or EBL. Acute bleed: octreotide + ceftriaxone + emergent EGD with banding. TIPS if refractory |
| Hepatic encephalopathy | Ammonia (and other toxins) bypass hepatic clearance | Lactulose (titrate to 3–4 BMs/day) + rifaximin 550 mg BID. Identify precipitant (infection, GI bleed, constipation, meds, electrolytes) |
| Hepatorenal syndrome | Splanchnic vasodilation → renal vasoconstriction | Type 1 (rapid, Cr doubles in < 2 weeks): IV albumin + vasopressors (terlipressin preferred, or norepinephrine/midodrine-octreotide). Definitive: liver transplant |
| HCC screening | Cirrhosis of any cause = HCC risk | Ultrasound ± AFP every 6 months |
16 Pancreatitis GI
Acute pancreatitis: inflammatory injury to the pancreas. Diagnosis requires 2 of 3: (1) characteristic abdominal pain (epigastric, radiating to back), (2) lipase ≥ 3× ULN, (3) characteristic imaging findings on CT. Causes: gallstones (~40%) and alcohol (~30%) account for ~70%. Others: hypertriglyceridemia (> 1000 mg/dL), post-ERCP, drugs (azathioprine, valproic acid, didanosine), autoimmune (IgG4-related), trauma.
Severity — Revised Atlanta Classification
| Severity | Organ Failure | Local Complications | Mortality |
|---|---|---|---|
| Mild | None | None | < 1% |
| Moderately severe | Transient (< 48 hr) or local complications alone | Peripancreatic collections, necrosis | ~5% |
| Severe | Persistent (> 48 hr) | Usually present | 20–40% |
Management: Aggressive IV fluid resuscitation (LR preferred, goal-directed — target UOP ≥ 0.5 mL/kg/hr), pain control (IV opioids, multimodal), early oral feeding as tolerated (within 24 hr if mild — no need for NPO until "pain-free"). Antibiotics only if infected necrosis is suspected or confirmed (not for prophylaxis in sterile necrosis). Cholecystectomy before discharge for gallstone pancreatitis (index admission if mild). Necrotizing pancreatitis with infection: step-up approach — percutaneous drainage first, surgical necrosectomy if drainage fails (van Santvoort et al., 2010 — PANTER trial).
17 Celiac Disease & Malabsorption GI
Celiac disease: autoimmune enteropathy triggered by dietary gluten (wheat, barley, rye) in genetically susceptible individuals (HLA-DQ2 in ~95%, DQ8 in ~5%). Prevalence ~1%. Presents with chronic diarrhea, steatorrhea, weight loss, iron deficiency anemia (refractory to oral iron), and dermatitis herpetiformis (pruritic vesicular rash on extensor surfaces). Can be silent — screen high-risk groups (T1DM, Down syndrome, autoimmune thyroid, first-degree relatives).
Diagnosis: Serology — tissue transglutaminase IgA (tTG-IgA) is the preferred screening test (sensitivity/specificity > 95%). Check total IgA simultaneously (IgA deficiency in ~3% of celiacs causes false-negative). Confirm with duodenal biopsy: villous atrophy, crypt hyperplasia, intraepithelial lymphocytosis (Marsh classification). Patient must be on gluten-containing diet during workup. Treatment: strict lifelong gluten-free diet — clinical improvement in weeks, mucosal healing in months.
Malabsorption — Differential by Nutrient
| Deficiency | Absorption Site | Causes |
|---|---|---|
| Iron | Duodenum | Celiac, Crohn's (proximal), gastrectomy, chronic blood loss |
| B12 | Terminal ileum | Crohn's (ileal), pernicious anemia, ileal resection, bacterial overgrowth, metformin |
| Folate | Proximal jejunum | Celiac, tropical sprue, alcohol, phenytoin, methotrexate |
| Fat-soluble vitamins (A, D, E, K) | Proximal small bowel (bile-dependent) | Cholestatic liver disease, pancreatic insufficiency, short bowel, bile salt malabsorption |
| Calcium | Duodenum/proximal jejunum | Vitamin D deficiency, celiac, post-bariatric surgery |
18 Acute Kidney Injury Nephrology
AKI is defined by KDIGO as any of: Cr rise ≥ 0.3 mg/dL within 48 hr, Cr rise ≥ 1.5× baseline within 7 days, or UOP < 0.5 mL/kg/hr for 6 hours. Incidence: 10–15% of hospitalized patients, up to 50% in ICU.
KDIGO Staging
| Stage | Serum Creatinine | Urine Output |
|---|---|---|
| 1 | 1.5–1.9× baseline OR ≥ 0.3 mg/dL increase | < 0.5 mL/kg/hr for 6–12 hr |
| 2 | 2.0–2.9× baseline | < 0.5 mL/kg/hr for ≥ 12 hr |
| 3 | ≥ 3.0× baseline OR Cr ≥ 4.0 OR initiation of RRT | < 0.3 mL/kg/hr for ≥ 24 hr OR anuria ≥ 12 hr |
Etiology — Pre-Renal vs Intrinsic vs Post-Renal
| Category | Mechanism | FENa | BUN/Cr Ratio | Urine Findings | Common Causes |
|---|---|---|---|---|---|
| Pre-renal (~60%) | Decreased perfusion | < 1% | > 20:1 | Bland, concentrated | Dehydration, hemorrhage, HF, sepsis (early), hepatorenal |
| Intrinsic (~35%) | Tubular, glomerular, or interstitial injury | > 2% | ~10–15:1 | Muddy brown granular casts (ATN), RBC casts (GN), WBC casts/eosinophils (AIN) | ATN (ischemic/toxic), GN, AIN (drugs), TTP/HUS |
| Post-renal (~5%) | Obstruction | Variable | Variable | Variable | BPH, kidney stones, tumor, retroperitoneal fibrosis |
19 Chronic Kidney Disease Nephrology
CKD is defined as kidney damage or GFR < 60 mL/min/1.73 m² for ≥ 3 months. Most common causes: diabetes (40%), hypertension (28%), glomerulonephritis (10%). CKD is a cardiovascular risk multiplier — patients are more likely to die of CV disease than progress to ESRD.
CKD Staging (KDIGO)
| Stage | GFR (mL/min/1.73 m²) | Description | Action |
|---|---|---|---|
| G1 | ≥ 90 | Normal/high GFR with kidney damage (proteinuria, hematuria) | Identify cause, ACEI/ARB if proteinuria, CVD risk reduction |
| G2 | 60–89 | Mildly decreased | Estimate progression rate |
| G3a | 45–59 | Mild-moderately decreased | Monitor complications (anemia, bone disease, acidosis) |
| G3b | 30–44 | Moderate-severely decreased | Refer to nephrology, adjust medications |
| G4 | 15–29 | Severely decreased | Prepare for RRT (access planning, transplant evaluation) |
| G5 | < 15 | Kidney failure | RRT (dialysis or transplant) if symptomatic |
CKD Complications & Management
| Complication | Mechanism | Treatment |
|---|---|---|
| Anemia | Decreased EPO production | Iron repletion first (ferritin > 100, TSAT > 20%), then ESA (epoetin, darbepoetin) targeting Hgb 10–11.5 |
| Metabolic bone disease (CKD-MBD) | Decreased 1,25-vit D → hypocalcemia → secondary hyperPTH → bone resorption | Phosphate binders (calcium acetate, sevelamer, lanthanum), calcitriol, calcimimetics (cinacalcet) |
| Metabolic acidosis | Decreased acid excretion | Oral sodium bicarbonate (target HCO₃ ≥ 22) |
| Hyperkalemia | Decreased K excretion | Dietary K restriction, avoid K-sparing diuretics, patiromer or SZC for chronic management |
| Volume overload | Decreased Na/water excretion | Loop diuretics (dose escalation as GFR falls), Na restriction |
SGLT2 inhibitors (dapagliflozin, empagliflozin) reduce CKD progression and cardiovascular events in CKD patients with or without diabetes — now a pillar of CKD management (DAPA-CKD trial, Heerspink et al., 2020).
20 Electrolyte Disorders Nephrology
Sodium Disorders
| Disorder | Definition | Key Causes | Treatment Principles |
|---|---|---|---|
| Hyponatremia (< 135) | Most common electrolyte disorder in hospitalized patients | Hypovolemic: diuretics, vomiting. Euvolemic: SIADH (most common), hypothyroidism, adrenal insufficiency. Hypervolemic: CHF, cirrhosis, nephrotic syndrome | Correct no faster than 8 mEq/L per 24 hr (risk of osmotic demyelination syndrome). Severe/symptomatic: 3% hypertonic saline 100 mL bolus |
| Hypernatremia (> 145) | Always indicates water deficit | Inadequate water intake (elderly, intubated), diabetes insipidus (central vs nephrogenic), osmotic diuresis | Free water replacement. Correct no faster than 10 mEq/L per 24 hr (risk of cerebral edema). D5W or free water via NGT |
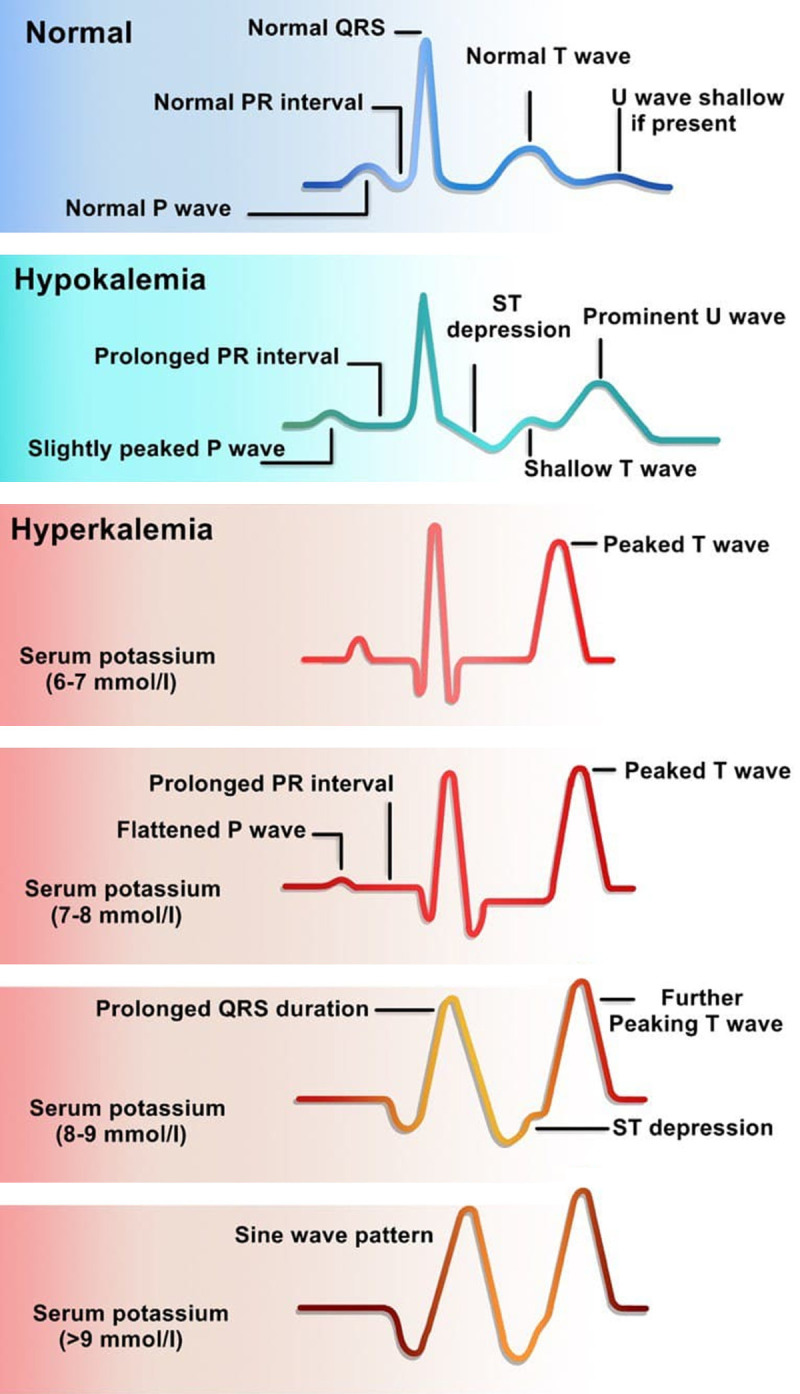
Potassium Disorders
| Disorder | ECG Changes | Causes | Treatment |
|---|---|---|---|
| Hypokalemia (< 3.5) | Flattened T waves, U waves, ST depression, prolonged QT | Diuretics, vomiting/NG suction (alkalosis shifts K intracellular), diarrhea, renal tubular acidosis, hypomagnesemia | Oral/IV KCl (10 mEq raises serum K by ~0.1). Always replete Mg concurrently (refractory hypoK if Mg low). Max IV rate 10 mEq/hr peripheral, 20 mEq/hr central |
| Hyperkalemia (> 5.0) | Peaked T waves → flattened P → widened QRS → sine wave → VFib/asystole | CKD, ACEI/ARB, K-sparing diuretics, acidosis, rhabdomyolysis, tumor lysis, hemolysis (pseudo) | K > 6.5 or ECG changes: calcium gluconate 1 g IV (cardioprotection, onset 1–3 min), insulin 10 units + D50 (shifts K intracellular), albuterol neb, kayexalate/patiromer (K removal), HD if refractory |
21 Acid-Base Disorders & Glomerulonephritis Nephrology
Acid-Base — Systematic Approach
Step 1: pH < 7.35 = acidemia, > 7.45 = alkalemia. Step 2: Primary disorder — same direction as pH? (low HCO₃ = metabolic acidosis, high PaCO₂ = respiratory acidosis). Step 3: Calculate expected compensation (Winter's formula for metabolic acidosis: expected PaCO₂ = 1.5 × HCO₃ + 8 ± 2). Step 4: Calculate anion gap = Na − (Cl + HCO₃). Step 5: If AG elevated, calculate delta-delta = (AG − 12) / (24 − HCO₃). Delta-delta > 2 = concurrent metabolic alkalosis; < 1 = concurrent non-AG metabolic acidosis.
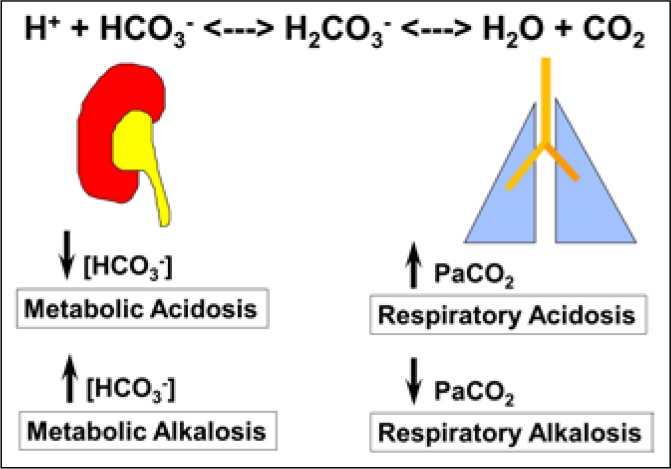
Common Acid-Base Patterns
| Disorder | pH | Primary Change | Compensation | Common Causes |
|---|---|---|---|---|
| AG metabolic acidosis | ↓ | ↓ HCO₃ | ↓ PaCO₂ | MUDPILES: Methanol, Uremia, DKA, Propylene glycol, INH/Iron, Lactic acidosis, Ethylene glycol, Salicylates |
| Non-AG metabolic acidosis | ↓ | ↓ HCO₃ | ↓ PaCO₂ | Diarrhea, RTA (types 1, 2, 4), normal saline infusion, acetazolamide |
| Metabolic alkalosis | ↑ | ↑ HCO₃ | ↑ PaCO₂ | Vomiting/NG suction (saline-responsive), diuretics, Cushing, Conn (saline-resistant) |
| Respiratory acidosis | ↓ | ↑ PaCO₂ | ↑ HCO₃ | COPD, obesity hypoventilation, neuromuscular disease, opioid overdose |
| Respiratory alkalosis | ↑ | ↓ PaCO₂ | ↓ HCO₃ | Anxiety, pain, PE, early sepsis, high altitude, pregnancy, salicylate toxicity (early) |
Glomerulonephritis — Nephritic vs Nephrotic
| Feature | Nephritic Syndrome | Nephrotic Syndrome |
|---|---|---|
| Proteinuria | Subnephrotic (< 3.5 g/day) | ≥ 3.5 g/day |
| Hematuria | Present (RBC casts = pathognomonic) | Usually absent |
| Edema | Mild (periorbital) | Severe (anasarca, periorbital, peripheral) |
| Hypertension | Common | Variable |
| Albumin | Normal or mildly low | Low (hypoalbuminemia) |
| Key diseases | IgA nephropathy (most common worldwide), post-infectious GN, anti-GBM, ANCA vasculitis, lupus nephritis | Minimal change (children), FSGS (adults, most common cause of nephrotic in Black adults), membranous nephropathy, diabetic nephropathy, amyloidosis |
22 Diabetes Mellitus Endocrinology
Diabetes affects ~37 million Americans (~11%). Type 2 accounts for 90–95% of cases. Diagnosis: fasting glucose ≥ 126 mg/dL, random glucose ≥ 200 with symptoms, OGTT 2-hr ≥ 200, or A1C ≥ 6.5% (confirmed on repeat testing unless unequivocal hyperglycemia). Prediabetes: A1C 5.7–6.4%, FG 100–125, OGTT 140–199.
Type 1 vs Type 2
| Feature | Type 1 | Type 2 |
|---|---|---|
| Mechanism | Autoimmune beta-cell destruction (anti-GAD, anti-IA2, anti-insulin Ab) | Insulin resistance + progressive beta-cell failure |
| Onset | Usually childhood/young adult; can occur at any age (LADA) | Usually adult; increasingly in adolescents |
| Body habitus | Often lean | Often obese (80%) |
| C-peptide | Low/absent | Normal/high (early), low (late) |
| Ketosis-prone | Yes (DKA is presenting feature in ~30%) | Less common (but can occur under stress) |
| Treatment | Insulin always (basal-bolus or pump) | Stepwise: lifestyle → metformin → add agents → insulin |
Type 2 DM — Pharmacotherapy (ADA Standards of Care 2023)
| Class | Example | Mechanism | A1C Reduction | Key Pearl |
|---|---|---|---|---|
| Biguanide | Metformin 500–2000 mg | Decreases hepatic glucose output, improves insulin sensitivity | 1.0–1.5% | First-line for all T2DM. Hold if eGFR < 30. GI side effects common, use XR formulation |
| SGLT2 inhibitor | Empagliflozin 10–25 mg, dapagliflozin 10 mg | Blocks glucose reabsorption in proximal tubule (glycosuria) | 0.5–0.9% | CV and renal benefits independent of A1C. Risk: euglycemic DKA, genital mycotic infections, Fournier gangrene (rare) |
| GLP-1 receptor agonist | Semaglutide (Ozempic) 0.5–2 mg/wk, liraglutide (Victoza) | Incretin mimetic — glucose-dependent insulin secretion, slows gastric emptying, reduces appetite | 1.0–1.8% | CV benefit (SUSTAIN-6, LEADER). Weight loss 5–15%. GI side effects (nausea). Avoid in MEN2/medullary thyroid cancer (animal data) |
| DPP-4 inhibitor | Sitagliptin (Januvia) 100 mg | Prevents incretin degradation | 0.5–0.8% | Weight-neutral, well-tolerated, but weaker A1C lowering. Do not combine with GLP-1 RA |
| Sulfonylurea | Glipizide 5–20 mg, glimepiride | Stimulates insulin secretion (K-ATP channel) | 1.0–1.5% | Cheap, effective, but hypoglycemia risk and weight gain. Avoid glyburide (highest hypo risk) |
| Thiazolidinedione | Pioglitazone 15–45 mg | PPAR-gamma agonist, improves insulin sensitivity | 0.5–1.4% | Fluid retention, CHF exacerbation, weight gain, bladder cancer risk (debated), fractures |
| Insulin | Basal: glargine (Lantus), detemir; Bolus: lispro (Humalog), aspart | Exogenous insulin replacement | No ceiling | Always needed in T1DM. In T2DM: start basal 10 units or 0.2 units/kg, titrate by 2–4 units q3 days to fasting goal |
| Feature | DKA | HHS |
|---|---|---|
| Typical patient | T1DM (or T2DM under stress) | T2DM, elderly |
| Glucose | > 250 mg/dL (can be lower — "euglycemic DKA") | > 600 mg/dL |
| pH | < 7.30 | > 7.30 |
| Bicarbonate | < 18 | > 18 |
| Ketones | Positive (serum beta-hydroxybutyrate > 3) | Absent or trace |
| Osmolality | Variable | > 320 mOsm/kg |
| Mental status | Alert to obtunded | Often obtunded/comatose |
| Mortality | 1–5% | 10–20% |
| Treatment | IVF (NS then switch to D5 1/2 NS when glucose < 200), insulin drip 0.1 units/kg/hr, K+ replacement (add K when < 5.3, hold insulin if K < 3.3), monitor AG closure | Aggressive IVF (often 6–9 L deficit), insulin drip (lower dose), gradual glucose correction |
23 Thyroid Disorders Endocrinology
Approach: TSH Is the Screening Test
| TSH | Free T4 | Interpretation | Common Cause |
|---|---|---|---|
| High | Low | Primary hypothyroidism | Hashimoto's (anti-TPO+), post-ablation, iodine deficiency |
| High | Normal | Subclinical hypothyroidism | Treat if TSH > 10 or symptomatic |
| Low | High | Hyperthyroidism | Graves' (TRAb+), toxic multinodular goiter, toxic adenoma |
| Low | Normal | Subclinical hyperthyroidism | Observe vs treat based on age, AFib risk, bone density |
| Low | Low | Central hypothyroidism | Pituitary disease (tumor, surgery, Sheehan's) |
Hypothyroidism treatment: levothyroxine (Synthroid) 1.6 mcg/kg/day, taken on empty stomach 30–60 min before food. Recheck TSH in 6–8 weeks. In elderly/cardiac patients, start low (25–50 mcg) and titrate slowly.
Hyperthyroidism treatment: Graves' — three options: (1) antithyroid drugs (methimazole preferred, start 10–30 mg daily — monitor for agranulocytosis, check WBC if sore throat/fever; PTU only in first trimester pregnancy or thyroid storm), (2) radioactive iodine ablation (I-131 — results in permanent hypothyroidism), (3) thyroidectomy. Thyroid storm: life-threatening decompensation — tachycardia, hyperthermia, AMS, HF. Treatment: PTU loading dose (500–1000 mg) + iodine (SSKI) 1 hr after PTU + beta-blocker (propranolol) + hydrocortisone 100 mg IV q8h + cooling.
24 Adrenal Disorders Endocrinology
Adrenal Insufficiency
Primary (Addison's): destruction of the adrenal cortex (autoimmune in developed countries, TB worldwide). Cortisol AND aldosterone deficient. Presentation: fatigue, weight loss, hypotension, hyperpigmentation (ACTH excess drives MSH), hyponatremia, hyperkalemia. Secondary: pituitary ACTH deficiency (most common cause = chronic exogenous steroid use then abrupt withdrawal). No hyperpigmentation, no hyperkalemia (aldosterone intact via RAAS). Diagnosis: morning cortisol < 3 = diagnostic, > 18 = excludes AI. 3–18 = cosyntropin stimulation test (cortisol should rise to ≥ 18 at 30 or 60 min). Treatment: hydrocortisone 15–25 mg/day in divided doses (2/3 AM, 1/3 PM) + fludrocortisone 0.05–0.2 mg daily (primary only). Adrenal crisis: IV hydrocortisone 100 mg bolus + aggressive IVF — do not wait for lab confirmation in shock.
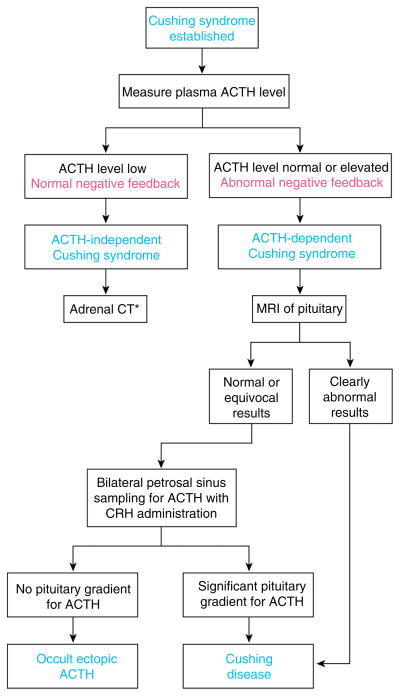
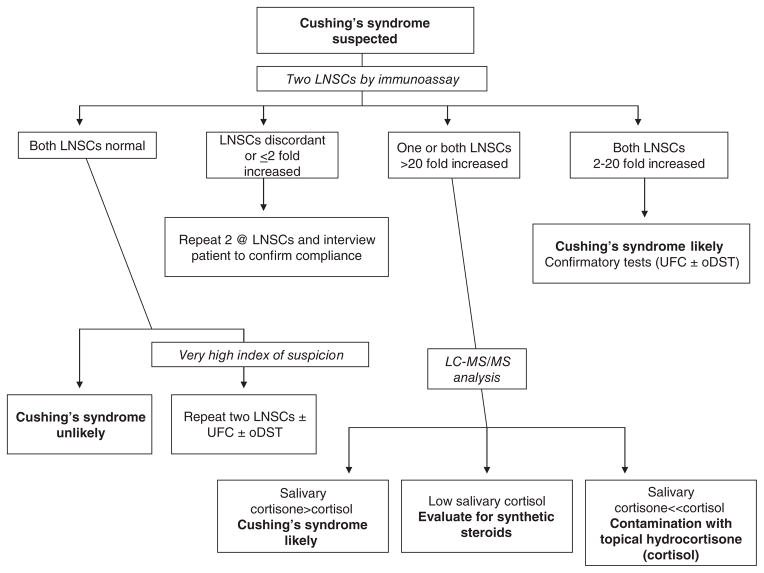
Cushing Syndrome
Excess cortisol. Most common cause overall: exogenous steroids. Most common endogenous cause: pituitary adenoma (Cushing's disease, ~70%). Features: central obesity, moon facies, dorsocervical fat pad, abdominal striae (purple, wide), proximal myopathy, hyperglycemia, hypertension, osteoporosis, easy bruising. Screening: 24-hr urine free cortisol, late-night salivary cortisol, 1 mg overnight dexamethasone suppression test (cortisol > 1.8 = positive). If confirmed, check ACTH: suppressed = adrenal source (CT adrenals); elevated = pituitary vs ectopic (high-dose dex suppression + pituitary MRI + IPSS if needed).
25 Calcium & Bone Disorders Endocrinology
Calcium Homeostasis
Always correct for albumin: corrected Ca = measured Ca + 0.8 × (4.0 − albumin). Or check ionized calcium directly. Two key hormones: PTH (raises Ca: bone resorption, renal reabsorption, activates vitamin D) and calcitonin (lowers Ca — clinically minor).
| Disorder | Calcium | PTH | Key Causes | Treatment |
|---|---|---|---|---|
| Primary hyperPTH | High | High (inappropriate) | Parathyroid adenoma (85%), hyperplasia (15%) | Surgery if symptomatic, Ca > 1 above normal, GFR < 60, T-score < −2.5, or age < 50 |
| Hypercalcemia of malignancy | High | Low (suppressed) | PTHrP (squamous cell, renal), bone mets (breast, lung), 1,25-vit D (lymphoma) | IVF (aggressive), calcitonin (rapid, transient), zoledronic acid (onset 2–4 days), denosumab |
| Vitamin D deficiency | Low-normal | High (secondary) | Inadequate sun/intake, malabsorption, CKD | Ergocalciferol 50,000 IU weekly × 8 weeks then 1000–2000 IU daily maintenance |
| Hypoparathyroidism | Low | Low | Post-thyroidectomy (#1 cause), autoimmune, DiGeorge | Calcium + calcitriol supplementation |
Symptoms: "stones, bones, groans, psychiatric overtones" — kidney stones, bone pain, abdominal pain/constipation, confusion/lethargy. ECG: shortened QT. Treatment: IV NS 200–300 mL/hr (volume repletion first), calcitonin 4 IU/kg q12h (onset 4–6 hr, tachyphylaxis after 48 hr), zoledronic acid 4 mg IV over 15 min (onset 2–4 days, lasts weeks). Loop diuretics only after volume resuscitation (forcing calciuresis). Dialysis for refractory cases.
26 Anemia Hematology
Anemia is defined as Hgb < 13.5 g/dL (men) or < 12.0 g/dL (women). The MCV-based approach is the diagnostic backbone:
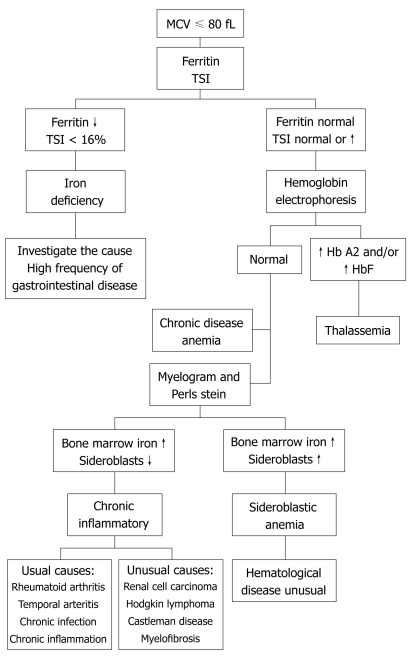
| MCV Category | MCV (fL) | Key Diagnoses | Diagnostic Tests |
|---|---|---|---|
| Microcytic (< 80) | < 80 | Iron deficiency (#1 worldwide), thalassemia, anemia of chronic disease, sideroblastic, lead poisoning | Iron studies: ferritin (< 30 = IDA), TIBC (high in IDA), Fe (low), TSAT (< 20%). Hgb electrophoresis for thalassemia |
| Normocytic (80–100) | 80–100 | Anemia of chronic disease/inflammation, CKD (EPO deficiency), acute blood loss, hemolytic anemia, bone marrow failure | Reticulocyte count (high = destruction/loss, low = underproduction). Hemolysis labs: LDH ↑, haptoglobin ↓, indirect bili ↑, smear (schistocytes = MAHA) |
| Macrocytic (> 100) | > 100 | B12 deficiency, folate deficiency, MDS, liver disease, hypothyroidism, reticulocytosis, drugs (MTX, hydroxyurea, AZT) | B12 and folate levels. If B12 borderline: methylmalonic acid (elevated in B12 def only) + homocysteine (elevated in both) |
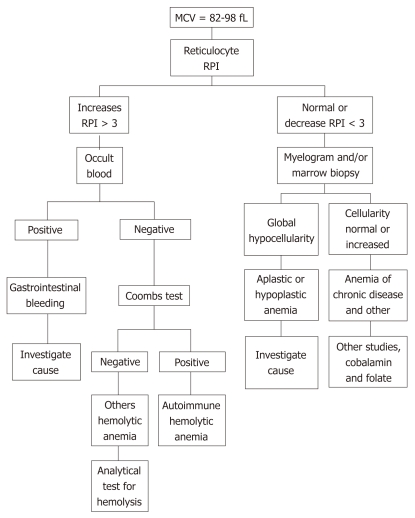
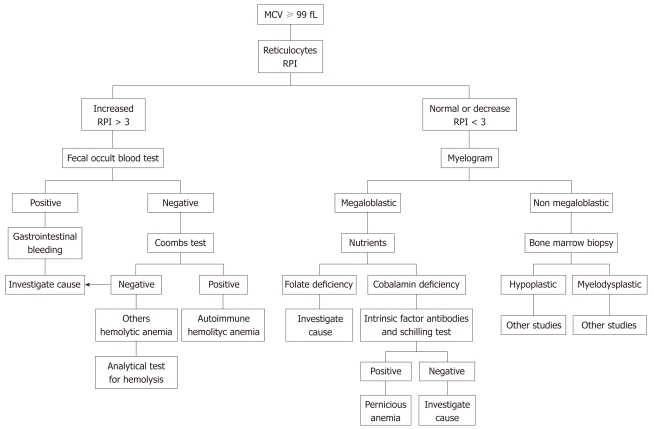
27 Coagulation Disorders Hematology
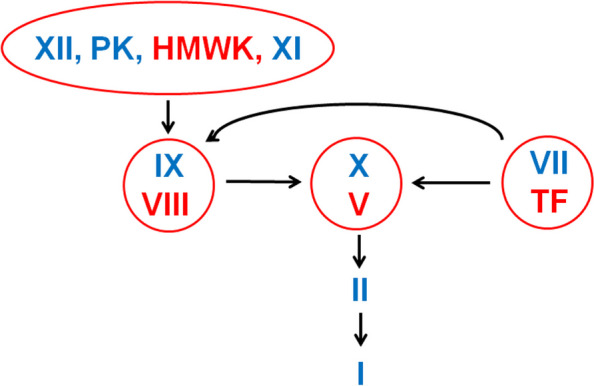
Coagulation Cascade — Key Lab Correlations
| Test | Pathway | Measures | Prolonged In |
|---|---|---|---|
| PT / INR | Extrinsic + common | Factors VII, X, V, II, fibrinogen | Warfarin, liver disease, vitamin K deficiency, DIC |
| aPTT | Intrinsic + common | Factors XII, XI, IX, VIII, X, V, II, fibrinogen | Heparin (UFH), hemophilia A (VIII) / B (IX), lupus anticoagulant, DIC |
| Thrombin time | Common (final) | Fibrinogen → fibrin conversion | Dabigatran, heparin contamination, low fibrinogen, FDPs |
| Mixing study | — | Corrects = factor deficiency; Doesn't correct = inhibitor | Factor inhibitors, lupus anticoagulant |
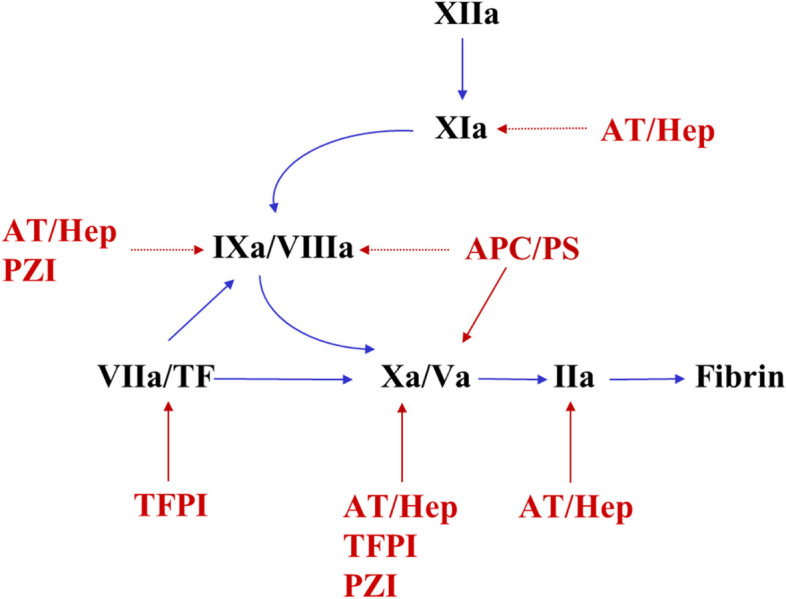
DIC (Disseminated Intravascular Coagulation)
Pathologic activation of coagulation with simultaneous consumption of clotting factors and platelets — producing both thrombosis and hemorrhage. Causes: sepsis (#1), trauma, malignancy (especially AML M3/APL), obstetric complications (abruption, amniotic fluid embolism). Labs: platelets ↓, PT/aPTT ↑, fibrinogen ↓, D-dimer ↑↑, schistocytes on smear. Treatment: treat the underlying cause. Supportive: platelets if < 10 (or < 50 with active bleeding), cryoprecipitate if fibrinogen < 100, FFP if bleeding with prolonged PT/aPTT.
HIT (Heparin-Induced Thrombocytopenia)
Type II HIT: immune-mediated (IgG against PF4-heparin complex). Platelet drop ≥ 50% typically 5–10 days after heparin exposure (or sooner if prior exposure). Paradoxically pro-thrombotic — venous and arterial thrombosis in 30–50%. Use 4T score to estimate probability. If HIT suspected: stop ALL heparin (including flushes), start alternative anticoagulant (argatroban, bivalirudin, fondaparinux). Do NOT give warfarin until platelets recover (causes skin necrosis/venous limb gangrene via protein C depletion). Confirm with anti-PF4 ELISA then serotonin release assay (gold standard).
28 Transfusion Medicine Hematology
| Product | Contents | Indication / Threshold | Volume / Details |
|---|---|---|---|
| Packed RBCs | Red cells in additive solution | Hgb < 7 (restrictive, most patients). Hgb < 8 (cardiac disease, symptomatic). Active hemorrhage | ~300 mL/unit. Expected rise: 1 g/dL per unit. Type and screen before transfusion |
| Platelets | Concentrated platelets | < 10K (prophylactic). < 50K (active bleeding or pre-procedure). < 100K (neurosurgery/ocular surgery) | 1 apheresis unit raises count ~30–50K. Irradiated if immunocompromised |
| FFP | All clotting factors | INR > 1.5–2.0 with active bleeding. Warfarin reversal (if PCC unavailable). DIC. TTP (plasma exchange) | 10–15 mL/kg. Takes ~30 min to thaw. Contains ~250 mL/unit |
| Cryoprecipitate | Fibrinogen, vWF, Factor VIII, XIII | Fibrinogen < 100–150 (DIC, massive transfusion). Hemophilia A (if factor concentrate unavailable) | 1 pool (10 units) raises fibrinogen ~60–100 mg/dL |
| PCC (4-factor) | Factors II, VII, IX, X, protein C & S | Warfarin reversal (urgent/emergent — preferred over FFP), factor deficiency | 25–50 units/kg IV. Onset 10–15 min. Much smaller volume than FFP |
Transfusion Reactions
| Reaction | Timing | Features | Management |
|---|---|---|---|
| Febrile non-hemolytic | During or within 4 hr | Fever, chills, rigors (no hemolysis) | Stop transfusion, acetaminophen. Resume if no hemolysis suspected. Leukoreduced products prevent recurrence |
| Allergic (mild) | During | Urticaria, pruritus | Diphenhydramine. May resume if symptoms resolve |
| Acute hemolytic | Within minutes | Fever, flank pain, dark urine, hypotension, DIC | Stop immediately. IVF, send direct Coombs, free Hgb, repeat type & screen. ABO mismatch is clerical error |
| TRALI | Within 6 hr | Acute bilateral pulmonary edema, hypoxia, normal cardiac pressures | Supportive (O₂, possible intubation). No diuretics (not volume overload) |
| TACO | Within 6 hr | Volume overload — dyspnea, HTN, elevated BNP, elevated JVP | Slow rate, diuretics, sit upright |
29 Sepsis & SIRS Infectious Disease
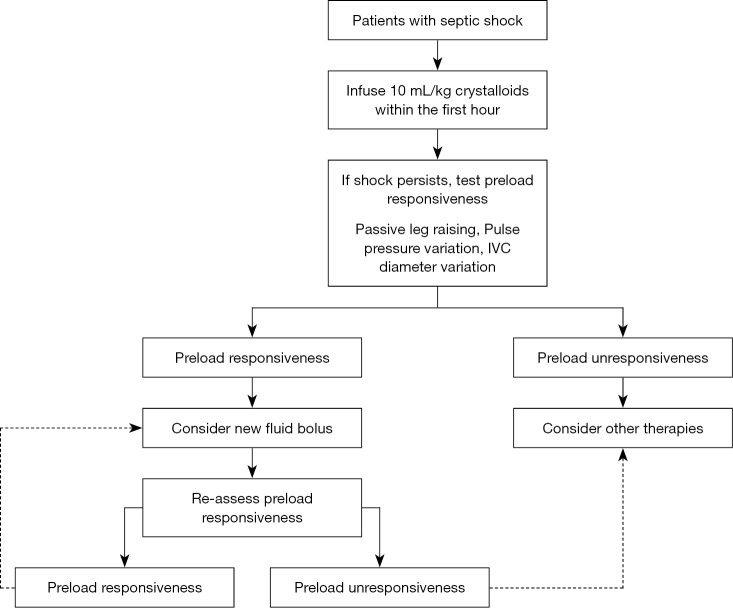
Sepsis (Sepsis-3, 2016): life-threatening organ dysfunction caused by a dysregulated host response to infection, operationally defined as suspected/documented infection + acute increase in SOFA score ≥ 2. Septic shock: sepsis + vasopressor requirement to maintain MAP ≥ 65 + lactate > 2 mmol/L despite adequate fluid resuscitation. Hospital mortality: sepsis ~10%, septic shock ~40% (Singer et al., 2016).
Surviving Sepsis Campaign — Hour-1 Bundle
| Action | Details |
|---|---|
| Measure lactate | Remeasure within 2–4 hr if initial > 2 mmol/L |
| Obtain blood cultures | Before antibiotics (2 sets from 2 sites). Do not delay antibiotics > 45 min for cultures |
| Administer broad-spectrum antibiotics | Within 1 hour of recognition. Common: vancomycin + piperacillin-tazobactam (or cefepime or meropenem) |
| Administer 30 mL/kg crystalloid | For hypotension or lactate ≥ 4. Balanced crystalloid (LR) preferred over NS |
| Start vasopressors | If MAP < 65 despite fluids. Norepinephrine first-line. Vasopressin (0.03 units/min) as second agent |
30 UTI & Pyelonephritis Infectious Disease
| Entity | Presentation | Diagnosis | Treatment |
|---|---|---|---|
| Uncomplicated cystitis (women) | Dysuria, frequency, urgency, suprapubic pain. No fever | UA (pyuria, nitrites, LE positive). Culture optional in uncomplicated | Nitrofurantoin 100 mg BID × 5 days (first-line), TMP-SMX DS BID × 3 days (if local resistance < 20%), fosfomycin 3 g single dose |
| Complicated UTI | UTI with structural abnormality, catheter, male sex, pregnancy, immunocompromised | UA + culture + sensitivities | Fluoroquinolone or beta-lactam; duration 7–14 days. Remove/replace catheter if present |
| Pyelonephritis | Fever, flank pain, CVA tenderness ± lower urinary symptoms, nausea/vomiting | UA + culture. Consider CT if no improvement in 48–72 hr (rule out abscess, obstruction) | Outpatient: ciprofloxacin 500 mg BID × 7 days or TMP-SMX × 14 days. Inpatient: ceftriaxone 1 g IV daily or fluoroquinolone IV. Blood cultures if systemic |
31 Infective Endocarditis Infectious Disease
Infection of the endocardial surface, usually a heart valve. Most common organisms: S. aureus (#1 overall and in acute IE/IVDU), viridans group streptococci (subacute, native valve), Enterococcus (elderly, GU source), HACEK organisms (subacute, culture-negative for days), coagulase-negative staph (prosthetic valves).
Modified Duke Criteria
Definite IE: 2 major, or 1 major + 3 minor, or 5 minor criteria.
| Major Criteria | Minor Criteria |
|---|---|
| Positive blood cultures (typical organisms from 2 separate cultures, or persistently positive) | Predisposing condition (IVDU, prosthetic valve, congenital heart disease) |
| Endocardial involvement on echo (vegetation, abscess, new dehiscence of prosthetic valve, new valvular regurgitation) | Fever ≥ 38°C |
| Vascular phenomena (Janeway lesions, arterial emboli, mycotic aneurysm, conjunctival hemorrhage) | |
| Immunologic phenomena (Osler nodes, Roth spots, glomerulonephritis, positive RF) | |
| Positive blood culture not meeting major criteria |
Treatment: Prolonged IV antibiotics (4–6 weeks). Native valve S. aureus: nafcillin/oxacillin (or vancomycin if MRSA) × 6 weeks. Viridans strep: penicillin G or ceftriaxone × 4 weeks (± gentamicin × 2 weeks for synergy). Prosthetic valve S. aureus: vancomycin + rifampin × ≥ 6 weeks + gentamicin × 2 weeks. Surgical indications: HF from valvular dysfunction, uncontrolled infection, large/mobile vegetations (> 10 mm with embolic event), abscess.
32 HIV & C. difficile Infectious Disease
HIV
Diagnosis: 4th-gen HIV Ag/Ab combo test (detects p24 antigen + HIV-1/2 antibodies). If positive, confirm with HIV-1/HIV-2 differentiation assay. Acute HIV: mononucleosis-like illness 2–4 weeks post-exposure (fever, pharyngitis, rash, lymphadenopathy) — high viral load, may be antibody-negative (Ag/Ab detects p24). Start antiretroviral therapy (ART) immediately upon diagnosis regardless of CD4 count (INSIGHT START trial, 2015). Preferred initial regimens: integrase inhibitor-based (bictegravir/emtricitabine/TAF = Biktarvy, or dolutegravir + emtricitabine/TAF).
Opportunistic infections by CD4 count: < 200: Pneumocystis jirovecii pneumonia (PJP — prophylaxis: TMP-SMX DS daily), oral candidiasis. < 100: Toxoplasma encephalitis, Cryptococcus meningitis. < 50: Mycobacterium avium complex (MAC), CMV retinitis.
Clostridioides difficile Infection
Toxin-producing anaerobic gram-positive bacillus — #1 cause of healthcare-associated infectious diarrhea. Risk factors: antibiotics (fluoroquinolones, clindamycin, cephalosporins), age > 65, hospitalization, PPI use. Diagnosis: stool PCR (toxin gene) — highly sensitive but may detect colonization; IDSA recommends testing only formed stools (never test formed stool).
| Severity | Criteria | Treatment (IDSA/SHEA 2021) |
|---|---|---|
| Non-severe | WBC ≤ 15K, Cr < 1.5 | Fidaxomicin 200 mg BID × 10 days (preferred) or vancomycin 125 mg PO QID × 10 days |
| Severe | WBC > 15K or Cr ≥ 1.5 | Vancomycin 125 mg PO QID × 10 days (or fidaxomicin) |
| Fulminant | Hypotension, ileus, megacolon, or organ failure | Vancomycin 500 mg PO/NGT QID + vancomycin rectal enema + IV metronidazole 500 mg q8h. Surgical consult for colectomy |
| Recurrence (1st) | Symptoms recur within 2–8 weeks | Fidaxomicin (pulsed-taper if prior vanco), or bezlotoxumab (anti-toxin B monoclonal) adjunct, or FMT for ≥ 2 recurrences |
33 Rheumatoid Arthritis & SLE Rheumatology
Rheumatoid Arthritis
Chronic systemic autoimmune disease causing symmetric inflammatory polyarthritis, predominantly affecting the small joints of the hands (MCP, PIP — spares DIP) and feet. Morning stiffness > 1 hour (improves with activity, unlike OA which worsens). Seropositive: RF (70–80%) and anti-CCP (more specific, ~95%). Extra-articular manifestations: rheumatoid nodules (subcutaneous, extensor surfaces), interstitial lung disease, pericarditis, Felty syndrome (RA + splenomegaly + neutropenia), secondary amyloidosis (AA type).
Treatment: Early aggressive DMARD therapy — methotrexate 15–25 mg/week (cornerstone, start within 3 months of diagnosis) + folic acid 1 mg daily. If inadequate response: add biologic — anti-TNF (adalimumab, etanercept, infliximab), IL-6 inhibitor (tocilizumab), B-cell depletion (rituximab), T-cell co-stimulation blocker (abatacept), or JAK inhibitor (tofacitinib, baricitinib). Treat-to-target: DAS28 remission or low disease activity.
Systemic Lupus Erythematosus
Systemic autoimmune disease with multi-organ involvement. Classic: young women (F:M = 9:1), photosensitive malar (butterfly) rash, arthritis, serositis, cytopenias, nephritis. Hallmark: ANA (positive in > 95% — sensitive but not specific). Specific antibodies: anti-dsDNA (lupus nephritis, disease activity), anti-Smith (most specific for SLE), anti-Ro/La (neonatal lupus, Sjogren overlap), antiphospholipid antibodies (thrombosis, pregnancy loss).
Lupus nephritis (occurs in ~50%): classified ISN/RPS Class I–VI. Class III (focal) and IV (diffuse) require aggressive immunosuppression — induction: mycophenolate mofetil (MMF) or cyclophosphamide + corticosteroids. Maintenance: MMF or azathioprine. Belimumab (anti-BLyS) added for active lupus. Voclosporin added for nephritis (AURORA trial, 2021). Hydroxychloroquine for ALL lupus patients (reduces flares, improves survival, renal protection) — annual eye exam for retinal toxicity.
34 Gout & Pseudogout Rheumatology
| Feature | Gout | Pseudogout (CPPD) |
|---|---|---|
| Crystal | Monosodium urate (MSU) | Calcium pyrophosphate dihydrate (CPPD) |
| Polarized microscopy | Needle-shaped, negatively birefringent (yellow parallel) | Rhomboid-shaped, positively birefringent (blue parallel) |
| Classic joint | 1st MTP (podagra, 50% of first attacks) | Knee (most common), wrist |
| X-ray | Erosions with overhanging edges ("rat bite"), tophi | Chondrocalcinosis (linear calcification of cartilage) |
| Risk factors | Hyperuricemia, alcohol (beer > liquor), purine-rich diet, CKD, diuretics | Elderly, hemochromatosis, hyperPTH, hypomagnesemia, hypothyroidism |
| Acute treatment | NSAIDs (indomethacin 50 mg TID), colchicine (1.2 mg then 0.6 mg in 1 hr), corticosteroids (if NSAID/colchicine CI) | Same acute management as gout |
| Chronic (urate-lowering) | Allopurinol (start 100 mg, titrate to urate < 6), febuxostat, pegloticase (refractory). Start with colchicine 0.6 mg daily prophylaxis | No urate-lowering therapy. Low-dose colchicine for prophylaxis if recurrent |
35 Vasculitis Rheumatology
Vasculitis = inflammation of blood vessel walls. Classified by vessel size (Chapel Hill Consensus):
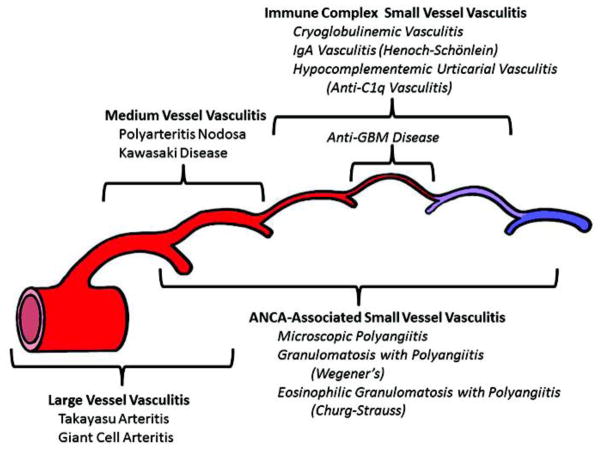
| Vessel Size | Disease | Key Features | Diagnosis / Treatment |
|---|---|---|---|
| Large | Giant Cell Arteritis (GCA) | Age > 50, new headache, jaw claudication, visual loss (ophthalmic artery — emergency), ESR/CRP markedly elevated, PMR overlap | Temporal artery biopsy (skip lesions — sample ≥ 2 cm). Start high-dose prednisone 60 mg immediately (do not wait for biopsy). Tocilizumab for steroid-sparing |
| Takayasu Arteritis | Young women (< 40), aortic arch/branch stenosis, "pulseless disease," limb claudication, BP discrepancy between arms | CTA/MRA showing wall thickening/stenosis. Steroids + methotrexate. Tocilizumab for refractory | |
| Medium | Polyarteritis Nodosa (PAN) | Hep B association. Skin (livedo, nodules), renal (microaneurysms, NOT glomerulonephritis), neuropathy (mononeuritis multiplex), GI (mesenteric ischemia). ANCA negative | Angiography (microaneurysms). Steroids + cyclophosphamide. Treat Hep B if associated |
| Kawasaki Disease | Children < 5, fever ≥ 5 days + 4 of 5: bilateral conjunctival injection, oral changes, rash, extremity changes, cervical LAD | IVIG + high-dose ASA. Risk of coronary artery aneurysms | |
| Small (ANCA-associated) | GPA (Wegener's) | Upper airway (sinusitis, saddle-nose deformity), lungs (cavitary lesions), kidneys (RPGN). c-ANCA / anti-PR3 | Steroids + rituximab (preferred over cyclophosphamide per RAVE trial). Maintenance: rituximab or azathioprine |
| MPA (Microscopic Polyangiitis) | Kidneys (RPGN, most common finding), lungs (alveolar hemorrhage). p-ANCA / anti-MPO | Same as GPA | |
| EGPA (Churg-Strauss) | Asthma, eosinophilia (> 1500), neuropathy, cardiac involvement. p-ANCA in ~40% | Steroids + mepolizumab (anti-IL5). Cyclophosphamide if organ-threatening | |
| Small (immune complex) | IgA Vasculitis (HSP) | Children. Palpable purpura (buttocks/legs), abdominal pain, arthritis, IgA nephropathy | Usually self-limited. NSAIDs for arthritis, steroids if renal or severe GI involvement |
36 Sarcoidosis Special
Systemic granulomatous disease of unknown etiology characterized by non-caseating granulomas in affected organs. Peak incidence: 25–35 years; 3× more common and more severe in Black Americans. Most common presentation: bilateral hilar lymphadenopathy (BHL) on CXR, often discovered incidentally.
Organ Involvement
| Organ | Frequency | Manifestations |
|---|---|---|
| Lungs | > 90% | BHL, parenchymal infiltrates, restrictive PFTs. Staging: I (BHL alone), II (BHL + parenchymal), III (parenchymal alone), IV (pulmonary fibrosis) |
| Skin | 25–35% | Erythema nodosum (NOT granulomatous — good prognosis), lupus pernio (violaceous facial plaques — poor prognosis), papules, plaques |
| Eyes | 25–50% | Anterior uveitis (most common ocular finding), lacrimal gland enlargement. All sarcoid patients need ophthalmologic exam |
| Liver | 60–80% (biopsy) | Granulomatous hepatitis, elevated ALP. Usually subclinical |
| Heart | 5–25% | Conduction defects (AV block), cardiomyopathy, sudden death. Screen with ECG; cardiac MRI if suspected |
| Neuro | 5–10% | CN VII palsy (most common), meningitis, hypothalamic/pituitary dysfunction, seizures |
| Calcium/endocrine | 10–20% | Hypercalcemia (granulomas produce 1,25-vit D via 1-alpha-hydroxylase), hypercalciuria, nephrolithiasis |
Diagnosis: Clinical + radiographic + histologic (non-caseating granulomas on biopsy of accessible tissue — skin, lymph node, lung via bronchoscopy with EBUS-TBNA). Elevated ACE level is supportive but neither sensitive nor specific. Must exclude TB and fungal infection (both cause granulomas). Treatment: Many cases resolve spontaneously (especially Lofgren syndrome: BHL + erythema nodosum + fever + polyarthralgia). Treat when symptomatic or organ-threatening: prednisone 20–40 mg/day, taper over 6–12 months. Steroid-sparing: methotrexate, azathioprine, mycophenolate. Refractory: infliximab.
37 Amyloidosis Special
Amyloidosis is the deposition of misfolded protein fibrils (amyloid) in tissues, disrupting organ function. Congo red stain shows apple-green birefringence under polarized light — pathognomonic.
| Type | Precursor Protein | Associations | Key Organ Involvement | Treatment |
|---|---|---|---|---|
| AL (primary) | Immunoglobulin light chain (lambda > kappa) | Plasma cell dyscrasia (myeloma, MGUS) | Heart (restrictive CMP — "thick walls, low voltage on ECG"), kidney (nephrotic syndrome), liver, neuropathy, macroglossia, periorbital purpura | Chemo targeting plasma cells: daratumumab + bortezomib/CyDex. Stem cell transplant if eligible |
| AA (secondary) | Serum amyloid A | Chronic inflammation (RA, IBD, FMF, chronic infections) | Kidneys (proteinuria, nephrotic syndrome) | Treat underlying inflammatory disease |
| ATTR (transthyretin) | Transthyretin (wild-type or mutant) | Wild-type: elderly men ("senile cardiac amyloidosis"). Hereditary: Val122Ile (cardiac, Black Americans), Val30Met (neuropathic) | Heart (HFpEF phenotype, often CTS), neuropathy | Tafamidis (Vyndamax) — TTR stabilizer, reduces mortality in ATTR-CM. Patisiran/inotersen for neuropathic |
38 Autoimmune Hepatitis Special
Chronic liver inflammation caused by autoimmune attack on hepatocytes. Bimodal: young women (Type 1, most common) and children (Type 2). Presents with fatigue, jaundice, elevated transaminases (often ALT > AST, sometimes dramatically elevated — can mimic acute viral hepatitis), hypergammaglobulinemia (elevated IgG), and autoantibodies. Can progress to cirrhosis if untreated.
Classification
| Type | Autoantibodies | Demographics |
|---|---|---|
| Type 1 | ANA, anti-smooth muscle antibody (ASMA) | Any age, most common in North America/Europe |
| Type 2 | Anti-LKM-1, anti-LC1 | Children and young adults, more severe |
Diagnosis: Simplified AIH scoring (IAIHG): autoantibodies + IgG elevation + liver histology (interface hepatitis, lymphoplasmacytic infiltrate, hepatocyte rosettes) + exclusion of viral hepatitis. Treatment: Induction: prednisone 30–60 mg/day tapered over weeks, plus azathioprine 50 mg then 1–2 mg/kg/day as steroid-sparing agent. Maintenance: azathioprine alone or low-dose prednisone + azathioprine. Remission goal: normal transaminases + IgG. Check TPMT before starting azathioprine (toxicity risk if deficient). Second-line: mycophenolate mofetil. Liver transplant for decompensated cirrhosis or fulminant presentation (can recur post-transplant in 20–30%).
39 Imaging & Diagnostics
| Modality | Primary Use in IM | Key Interpretation Points |
|---|---|---|
| Chest X-ray | Pneumonia, CHF, pleural effusion, pneumothorax, masses | CHF: cephalization, Kerley B lines, peribronchial cuffing, bilateral pleural effusions. Pneumonia: lobar consolidation (air bronchograms) vs interstitial pattern |
| ECG (12-lead) | Arrhythmia, ACS, PE, electrolyte abnormalities | STEMI criteria, AFib (absent P waves, irregular RR), PE (S1Q3T3, RV strain), hyperK (peaked T), hypoK (U waves) |
| Echocardiography (TTE) | EF, valvular disease, wall motion, pericardial effusion, diastolic function | HFrEF ≤ 40%, severe AS (AVA < 1.0 cm², mean gradient > 40 mmHg), vegetation in IE |
| CT chest (with contrast) | PE (CTPA), ILD (HRCT), aortic dissection, lung mass | PE: filling defect in pulmonary artery. UIP pattern (IPF): basal honeycombing. Dissection: intimal flap |
| CT abdomen/pelvis | Pancreatitis complications, abscess, obstruction, renal stones | Pancreatitis: peripancreatic fat stranding, necrosis (non-enhancing areas), fluid collections |
| Abdominal ultrasound | Gallstones, liver/renal morphology, ascites, hydronephrosis | Cirrhosis: nodular liver, splenomegaly, ascites. Hydronephrosis in obstructive AKI |
| Renal ultrasound | AKI workup (obstruction), CKD (kidney size) | Normal kidney 9–12 cm. Small kidneys = CKD (chronic). Large kidneys: DM nephropathy, amyloid, HIV, polycystic |
| PFTs (spirometry, lung volumes, DLCO) | Obstructive vs restrictive, severity, response to bronchodilator | Obstructive: FEV₁/FVC < 0.70 (COPD, asthma). Restrictive: FVC ↓, TLC ↓, FEV₁/FVC normal/↑. DLCO ↓ = ILD, emphysema, PAH |
| Paracentesis fluid analysis | SBP vs secondary peritonitis, portal HTN assessment | SAAG ≥ 1.1 = portal hypertension. PMN ≥ 250 = SBP. Total protein, glucose, LDH, culture |
| Bone marrow biopsy | Unexplained cytopenias, MDS, leukemia, myelofibrosis, amyloidosis | Cellularity, blast percentage, iron stores, flow cytometry, cytogenetics |
40 Classification Systems (All)
| System | Used For | Key Features |
|---|---|---|
| ACC/AHA HTN (2017) | Blood pressure classification | Normal/Elevated/Stage 1/Stage 2/Crisis. Stage 1 starts at 130/80 (lowered from 140/90) |
| NYHA (I–IV) | Heart failure functional status | I = no limitation, IV = symptoms at rest. Guides device therapy (ICD if EF ≤ 35% + NYHA II–III) |
| ACC/AHA HF Stages (A–D) | Heart failure progression | A = at risk, D = refractory (LVAD/transplant). Unlike NYHA, stages only advance (never regress) |
| CHA₂DS₂-VASc | Stroke risk in AFib | Score 0–9. ≥ 2 men / ≥ 3 women: anticoagulate |
| HAS-BLED | Bleeding risk on anticoagulation | Score 0–9. ≥ 3 = high risk — does NOT contraindicate anticoagulation, but mandates closer monitoring |
| Wells (DVT / PE) | Pre-test probability | DVT: ≥ 2 = likely. PE: > 4 = likely. Low probability + negative D-dimer = excludes VTE |
| GOLD (COPD) | Airflow obstruction severity | Stages 1–4 by FEV₁. ABE groups guide therapy |
| GINA Steps (1–5) | Asthma controller therapy | Step up if uncontrolled, step down after 3 months of control |
| CURB-65 | CAP severity | 0–1 outpatient, 2 short stay, 3–5 inpatient/ICU |
| Light's Criteria | Pleural effusion (exudate vs transudate) | Any 1 of 3 criteria met = exudate. Sensitivity 98% |
| Child-Pugh (A/B/C) | Cirrhosis severity | 5 parameters, each 1–3 points. A = compensated, C = decompensated |
| MELD-Na | Liver transplant allocation | Bilirubin, INR, Cr, Na. Range 6–40. ≥ 15 = consider listing |
| Revised Atlanta | Acute pancreatitis severity | Mild/Moderate/Severe by organ failure + local complications |
| KDIGO (AKI & CKD) | Kidney disease staging | AKI: stages 1–3 by Cr/UOP. CKD: G1–G5 by GFR + A1–A3 by albuminuria |
| Marsh (I–IIIc) | Celiac disease histology | I = intraepithelial lymphocytosis, IIIc = total villous atrophy |
| ISN/RPS (I–VI) | Lupus nephritis | III (focal) and IV (diffuse) = aggressive immunosuppression |
| Modified Duke Criteria | Infective endocarditis diagnosis | Major (blood cultures, echo) + minor criteria. 2 major = definite IE |
| Sepsis-3 / SOFA | Sepsis definition & organ dysfunction | Suspected infection + SOFA ≥ 2. qSOFA (bedside): RR ≥ 22, AMS, SBP ≤ 100 |
| Schofield/Sarcoid Staging (0–IV) | Pulmonary sarcoidosis | 0 = normal CXR, I = BHL, II = BHL + parenchymal, III = parenchymal alone, IV = fibrosis |
| Chapel Hill Consensus | Vasculitis classification | By vessel size: large, medium, small. Guides workup and treatment |
41 Medications Master Table
Cardiovascular
| Generic (Brand) | Class | Mechanism | Typical Dose | Indication | Critical Pearl |
|---|---|---|---|---|---|
| Lisinopril (Zestril) | ACEI | Blocks ACE → decreases angiotensin II → vasodilation + decreased aldosterone | 10–40 mg daily | HTN, HFrEF, DM nephropathy, post-MI | Monitor K+/Cr. Cough 10%. Angioedema rare but life-threatening. CI in pregnancy |
| Sacubitril/valsartan (Entresto) | ARNI | Neprilysin inhibitor + ARB | 97/103 mg BID (target) | HFrEF (replaces ACEI/ARB) | 36-hr washout from ACEI before starting (angioedema risk). Do not use with ACEI |
| Metoprolol succinate (Toprol-XL) | Beta-blocker (selective) | Beta-1 blockade → decreased HR, contractility, renin | 25–200 mg daily | HFrEF, HTN, rate control AFib, post-MI | Succinate (XR) for HF, not tartrate (IR). Do not stop abruptly (rebound tachycardia) |
| Amlodipine (Norvasc) | DHP-CCB | Blocks L-type Ca channels → arterial vasodilation | 5–10 mg daily | HTN, angina | Peripheral edema is dose-related, not allergic. Safe in HFrEF (unlike verapamil/diltiazem) |
| Spironolactone (Aldactone) | MRA | Aldosterone antagonist → K-sparing diuresis | 25–50 mg daily | HFrEF, resistant HTN, ascites, primary aldosteronism | Gynecomastia (switch to eplerenone). Hyperkalemia risk with ACEI/ARB + CKD |
| Furosemide (Lasix) | Loop diuretic | Blocks NKCC2 in thick ascending limb | 20–80 mg IV/PO (up to 600 mg/day in CKD) | CHF, edema, CKD fluid overload | IV:PO ratio is 1:2. Causes hypoK, hypoNa, hypoMg, hypoCa, ototoxicity (high dose) |
| Apixaban (Eliquis) | DOAC (Xa inhibitor) | Direct factor Xa inhibition | 5 mg BID (VTE, AFib). 2.5 mg BID if ≥ 2 of: age ≥ 80, weight ≤ 60 kg, Cr ≥ 1.5 | AFib stroke prevention, VTE treatment | No routine monitoring. Reversal: andexanet alfa. Avoid if CrCl < 25 (or < 15). Fewest GI bleeds among DOACs |
Pulmonary
| Generic (Brand) | Class | Dose | Use | Pearl |
|---|---|---|---|---|
| Albuterol (ProAir, Ventolin) | SABA | 2 puffs q4–6h PRN or nebulizer 2.5 mg | Acute bronchospasm (COPD, asthma) | Tachycardia, tremor, hypokalemia at high doses |
| Tiotropium (Spiriva) | LAMA | 18 mcg inhaled daily (HandiHaler) or 2.5 mcg (Respimat) | Maintenance COPD, step-up asthma | Dry mouth common. Do not use with short-acting antimuscarinics |
| Budesonide/formoterol (Symbicort) | ICS/LABA | 160/4.5 mcg 2 puffs BID + PRN (SMART therapy) | Asthma (Steps 3–5), COPD with eos ≥ 300 | Rinse mouth after ICS to prevent oral candidiasis. LABA monotherapy in asthma = increased death |
Endocrine & GI
| Generic (Brand) | Class | Dose | Use | Pearl |
|---|---|---|---|---|
| Metformin (Glucophage) | Biguanide | 500–2000 mg daily (XR preferred) | T2DM first-line | Hold if eGFR < 30 or before iodinated contrast (risk of lactic acidosis). GI side effects common — start low, go slow |
| Semaglutide (Ozempic) | GLP-1 RA | 0.25 mg → 0.5 mg → 1 mg → 2 mg SQ weekly | T2DM, obesity (Wegovy formulation) | Nausea common initially. CV benefit. Weight loss 10–15%. Pancreatitis risk (debated) |
| Empagliflozin (Jardiance) | SGLT2i | 10–25 mg daily | T2DM, HFrEF, HFpEF, CKD | Genital mycotic infections. Euglycemic DKA (rare). Hold before surgery |
| Levothyroxine (Synthroid) | Thyroid hormone | 1.6 mcg/kg/day | Hypothyroidism | Empty stomach, 30–60 min before food. Ca, Fe, PPIs reduce absorption (separate by 4 hr) |
| Omeprazole (Prilosec) | PPI | 20–40 mg daily | GERD, PUD, H. pylori (with abx) | Long-term risks: C. diff, hypoMg, B12 def, osteoporosis, AIN. Step down to H2RA when possible |
| Lactulose | Osmotic laxative | 15–45 mL PO q1–2h initially, then titrate to 3–4 BMs/day | Hepatic encephalopathy | Works by converting NH₃ to NH₄⁺ (non-absorbable) in the colon. Diarrhea and electrolyte depletion if over-dosed |
Infectious Disease & Rheumatology
| Generic (Brand) | Class | Dose | Use | Pearl |
|---|---|---|---|---|
| Vancomycin (Vancocin) | Glycopeptide | 15–20 mg/kg IV q8–12h (AUC/MIC target 400–600) | MRSA (bacteremia, endocarditis, pneumonia) | Nephrotoxicity, red man syndrome (infuse over ≥ 1 hr). Oral form for C. diff only (not absorbed) |
| Piperacillin-tazobactam (Zosyn) | Beta-lactam/BLI | 4.5 g IV q6h (extended infusion over 4 hr preferred) | Broad-spectrum empiric (HAP, intra-abdominal, febrile neutropenia) | Pseudomonal coverage. Increased AKI risk when combined with vancomycin (use cefepime instead) |
| Methotrexate | DMARD (antimetabolite) | 15–25 mg PO/SQ weekly | RA (cornerstone), psoriasis, SLE, sarcoidosis | Always give folic acid 1 mg daily. Hepatotoxicity, myelosuppression, ILD. CI in pregnancy (teratogen). Hold for surgery/infection |
| Hydroxychloroquine (Plaquenil) | Antimalarial/immunomodulator | 200–400 mg daily | SLE (all patients), RA | Retinal toxicity with long-term use — annual eye exam after 5 years. QTc prolongation at high doses |
| Allopurinol (Zyloprim) | Xanthine oxidase inhibitor | 100 mg → titrate to urate < 6 mg/dL (max 800 mg) | Chronic gout, tumor lysis prophylaxis | Start low, go slow. HLA-B*5801 testing in SE Asian/Black patients (DRESS/SJS risk). Never start during acute flare |
42 Abbreviations Master List
General & Cardiovascular
| Abbreviation | Meaning |
|---|---|
| ACEI / ARB | Angiotensin-converting enzyme inhibitor / angiotensin receptor blocker |
| AFib | Atrial fibrillation |
| ACS | Acute coronary syndrome |
| BNP / NT-proBNP | B-type natriuretic peptide / N-terminal pro-BNP |
| CAD | Coronary artery disease |
| CCB | Calcium channel blocker |
| CHA₂DS₂-VASc | CHF, HTN, Age, DM, Stroke, Vascular, Age, Sex category |
| DAPT | Dual antiplatelet therapy |
| DOAC | Direct oral anticoagulant |
| EF / LVEF | Ejection fraction / left ventricular EF |
| GDMT | Guideline-directed medical therapy |
| HFrEF / HFpEF | Heart failure with reduced / preserved ejection fraction |
| HTN | Hypertension |
| INR | International normalized ratio |
| MAP | Mean arterial pressure |
| MI / NSTEMI / STEMI | Myocardial infarction / non-ST-elevation MI / ST-elevation MI |
| MRA | Mineralocorticoid receptor antagonist |
| NYHA | New York Heart Association |
| PCI | Percutaneous coronary intervention |
| VTE / DVT / PE | Venous thromboembolism / deep vein thrombosis / pulmonary embolism |
Pulmonary & GI
| Abbreviation | Meaning |
|---|---|
| ABG | Arterial blood gas |
| ARDS | Acute respiratory distress syndrome |
| BiPAP / NIPPV | Bilevel positive airway pressure / non-invasive positive pressure ventilation |
| CAP / HAP / VAP | Community-acquired / hospital-acquired / ventilator-associated pneumonia |
| COPD | Chronic obstructive pulmonary disease |
| DLCO | Diffusing capacity of the lungs for carbon monoxide |
| FEV₁ / FVC | Forced expiratory volume in 1 second / forced vital capacity |
| GERD | Gastroesophageal reflux disease |
| GI | Gastrointestinal |
| IBD / UC / CD | Inflammatory bowel disease / ulcerative colitis / Crohn's disease |
| ICS / LABA / LAMA | Inhaled corticosteroid / long-acting beta-agonist / long-acting muscarinic antagonist |
| ILD | Interstitial lung disease |
| LFTs | Liver function tests (AST, ALT, ALP, bilirubin, albumin) |
| MELD | Model for End-Stage Liver Disease |
| PFTs | Pulmonary function tests |
| PPI | Proton pump inhibitor |
| PUD | Peptic ulcer disease |
| SBP | Spontaneous bacterial peritonitis (or systolic blood pressure, context-dependent) |
| TIPS | Transjugular intrahepatic portosystemic shunt |
Nephrology, Endocrine, Heme, Rheum, ID
| Abbreviation | Meaning |
|---|---|
| AKI | Acute kidney injury |
| ANA | Antinuclear antibody |
| ANCA | Anti-neutrophil cytoplasmic antibody (c-ANCA = PR3, p-ANCA = MPO) |
| ATN | Acute tubular necrosis |
| BUN / Cr | Blood urea nitrogen / creatinine |
| CBC | Complete blood count |
| CKD | Chronic kidney disease |
| DIC | Disseminated intravascular coagulation |
| DKA / HHS | Diabetic ketoacidosis / hyperosmolar hyperglycemic state |
| DMARD | Disease-modifying antirheumatic drug |
| EPO / ESA | Erythropoietin / erythropoiesis-stimulating agent |
| FENa | Fractional excretion of sodium |
| GFR / eGFR | Glomerular filtration rate / estimated GFR |
| GN / RPGN | Glomerulonephritis / rapidly progressive GN |
| A1C / HbA1c | Glycosylated hemoglobin |
| HIT | Heparin-induced thrombocytopenia |
| MAHA | Microangiopathic hemolytic anemia |
| MCV | Mean corpuscular volume |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| PF4 | Platelet factor 4 |
| PTH | Parathyroid hormone |
| RA / SLE | Rheumatoid arthritis / systemic lupus erythematosus |
| RRT | Renal replacement therapy (dialysis) |
| SGLT2i | Sodium-glucose cotransporter 2 inhibitor |
| SIRS | Systemic inflammatory response syndrome |
| SOFA / qSOFA | Sequential Organ Failure Assessment / quick SOFA |
| TSH | Thyroid-stimulating hormone |
| TTP / HUS | Thrombotic thrombocytopenic purpura / hemolytic uremic syndrome |
| UA | Urinalysis |
43 Risk Scores & Preventive Guidelines
Key Risk Scores
| Score | Purpose | Components | Action Threshold |
|---|---|---|---|
| ASCVD Pooled Cohort Equation | 10-year CV risk (primary prevention) | Age, sex, race, total/HDL cholesterol, SBP, BP treatment, DM, smoking | ≥ 7.5%: moderate-intensity statin. ≥ 20%: high-intensity statin + ASA discussion |
| HEART Score | Chest pain risk stratification in ED | History, ECG, Age, Risk factors, Troponin (each 0–2) | 0–3: low risk (discharge). 4–6: intermediate. 7–10: high (admit, early cath) |
| RCRI (Revised Cardiac Risk Index) | Perioperative cardiac risk | High-risk surgery, CAD, HF, CVA/TIA, DM on insulin, Cr > 2.0 | 0 = 3.9% MACE, 1 = 6%, 2 = 10%, ≥ 3 = 15% |
| MELD-Na | Liver disease mortality / transplant priority | Bilirubin, INR, Cr, Na | ≥ 15: transplant evaluation. ≥ 30: high 3-month mortality |
| qSOFA | Bedside sepsis screening | RR ≥ 22, AMS, SBP ≤ 100 (each 1 point) | ≥ 2: high risk, further evaluation |
| 4T Score | HIT probability | Thrombocytopenia, Timing, Thrombosis, oTher causes (each 0–2) | 0–3: low. 4–5: intermediate. 6–8: high probability |
| STOPBANG | OSA screening | Snoring, Tired, Observed apnea, Pressure (HTN), BMI > 35, Age > 50, Neck > 40 cm, Gender male | ≥ 3: high risk for OSA, refer for polysomnography |
USPSTF Screening Guidelines (High-Yield)
| Condition | Screen | Population | Interval |
|---|---|---|---|
| Colorectal cancer | Colonoscopy, FIT, or stool DNA | Age 45–75 | Colonoscopy q10yr, FIT annually, Cologuard q3yr |
| Breast cancer | Mammography | Women 40–74 | Every 2 years (biennial) |
| Cervical cancer | Pap smear ± HPV co-testing | Women 21–65 | Pap q3yr (21–29), Pap + HPV q5yr (30–65) |
| Lung cancer | Low-dose CT chest | Age 50–80, ≥ 20 pack-year smoking hx (current or quit < 15 yr) | Annually |
| AAA | Abdominal US | Men 65–75 who have ever smoked | Once |
| Osteoporosis | DEXA | Women ≥ 65 (or younger with risk factors) | Baseline, repeat based on T-score |
| Diabetes | Fasting glucose, A1C, or OGTT | Age 35–70 with BMI ≥ 25 (overweight/obese) | Every 3 years if normal |
| Hepatitis C | Anti-HCV antibody | All adults 18–79 (one-time) | Once |
| HIV | HIV Ag/Ab combo | All adults 15–65 | Once (more frequent if high-risk) |
| Lipids | Lipid panel | Men ≥ 35, women ≥ 45 (earlier if risk factors) | Every 5 years |