Nephrology
Every renal diagnosis, electrolyte disorder, acid-base disturbance, dialysis modality, transplant protocol, glomerular disease, classification system, and management strategy in one place.
01 Renal Anatomy & Physiology
The kidneys are paired retroperitoneal organs located at the T12–L3 vertebral levels, each weighing approximately 120–170 g in adults and measuring 10–12 cm in length. They receive ~20–25% of cardiac output (~1,000–1,200 mL/min) despite constituting only 0.5% of body weight, making them the most highly perfused organs per gram of tissue. The kidneys regulate fluid and electrolyte balance, acid-base homeostasis, blood pressure (via the renin-angiotensin-aldosterone system), erythropoiesis (EPO production), and vitamin D activation (1,25-dihydroxyvitamin D).
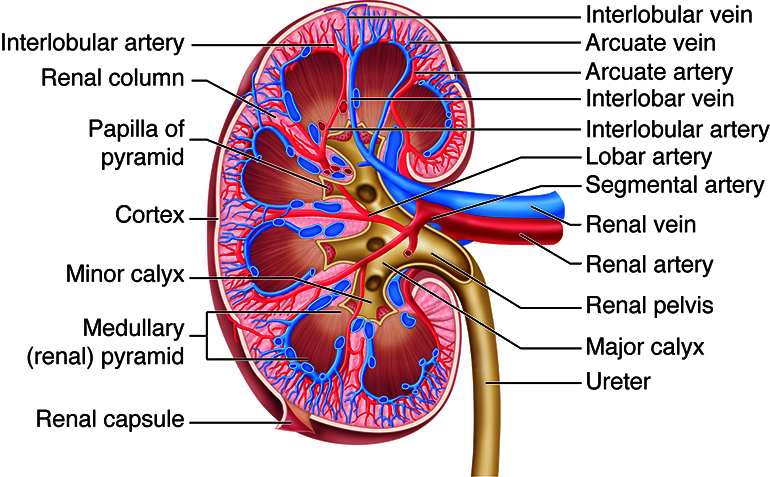
Gross Anatomy
Each kidney is surrounded by a fibrous renal capsule, perinephric fat, and Gerota's fascia. The parenchyma consists of an outer cortex (containing glomeruli, proximal and distal convoluted tubules) and an inner medulla (containing loops of Henle and collecting ducts arranged in 8–18 renal pyramids). The pyramids drain into minor calyces → major calyces → renal pelvis → ureter. The renal hilum transmits the renal artery, renal vein, ureter, lymphatics, and nerves. The left renal vein is longer and crosses anterior to the aorta (relevant for nutcracker syndrome and as a surgical landmark).
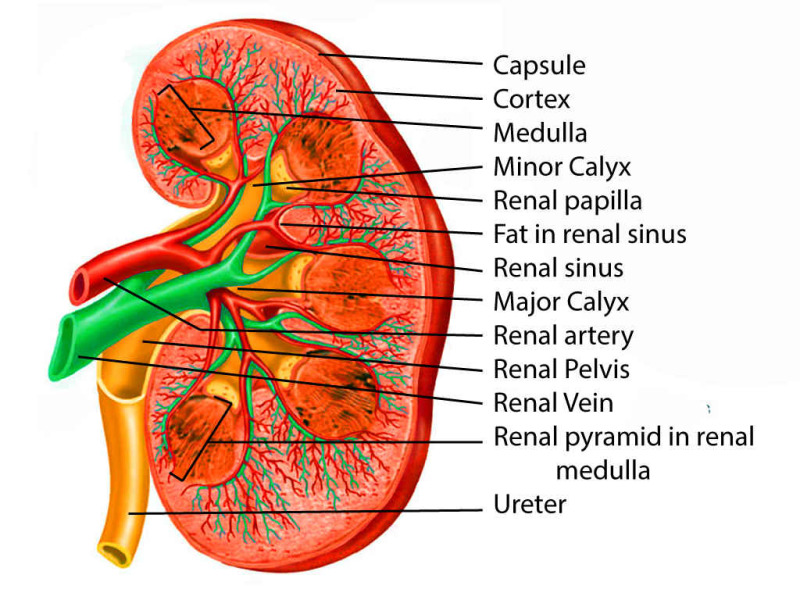
The Nephron
Each kidney contains approximately 1 million nephrons — the functional unit of the kidney. Each nephron consists of a glomerulus (capillary tuft within Bowman's capsule), proximal convoluted tubule (PCT), loop of Henle (thin descending limb, thin ascending limb, thick ascending limb), distal convoluted tubule (DCT), and collecting duct. Cortical nephrons (~85%) have short loops; juxtamedullary nephrons (~15%) have long loops that penetrate deep into the medulla, critical for the countercurrent concentrating mechanism.
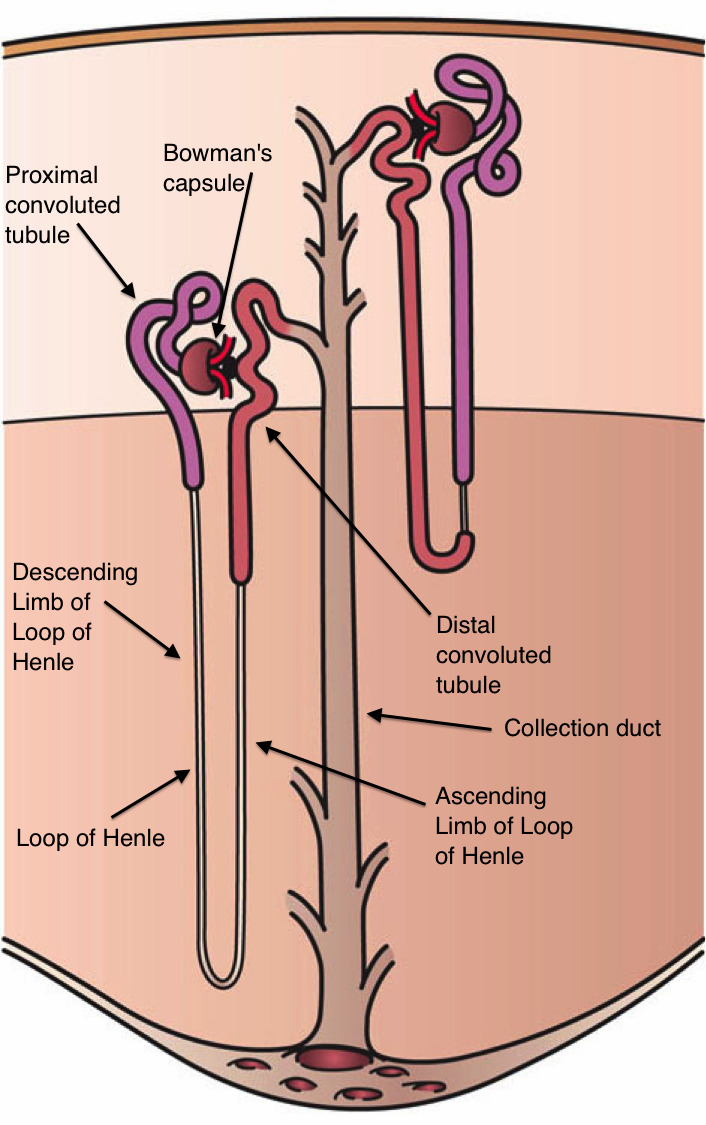
Nephron Segment Functions
| Segment | Key Functions | Transporters | Drugs Acting Here |
|---|---|---|---|
| Proximal tubule | Reabsorbs ~65% Na+, HCO3−, glucose, amino acids, phosphate, uric acid; secretes organic acids/bases | Na+/H+ exchanger (NHE3), SGLT2, Na-phosphate cotransporter | SGLT2 inhibitors, carbonic anhydrase inhibitors (acetazolamide) |
| Thin descending limb | Permeable to water, impermeable to solute → concentrates tubular fluid | Aquaporin-1 | — |
| Thick ascending limb (TAL) | Reabsorbs ~25% Na+, K+, Cl−; impermeable to water ("diluting segment"); generates medullary gradient | Na-K-2Cl cotransporter (NKCC2) | Loop diuretics (furosemide, bumetanide) |
| Distal convoluted tubule | Reabsorbs ~5% NaCl; Ca2+ reabsorption (PTH-sensitive) | Na-Cl cotransporter (NCC) | Thiazide diuretics |
| Collecting duct (principal cells) | Na+ reabsorption, K+ secretion (aldosterone-mediated); water reabsorption (ADH-mediated) | ENaC (epithelial Na channel), ROMK, Aquaporin-2 | K-sparing diuretics (amiloride, spironolactone) |
| Collecting duct (intercalated cells) | Type A: H+ secretion, HCO3− reabsorption; Type B: HCO3− secretion, H+ reabsorption | H+-ATPase, H+/K+-ATPase, pendrin | — |
Glomerular Filtration
The glomerular filtration barrier consists of three layers: fenestrated endothelium, glomerular basement membrane (GBM), and podocyte foot processes with slit diaphragms. Normal GFR is ~120 mL/min/1.73 m² in young adults. The GBM carries a negative charge that repels albumin (anionic); loss of this charge barrier (e.g., in minimal change disease) leads to proteinuria. Starling forces govern filtration: GFR = Kf × [(PGC − PBS) − (πGC − πBS)], where PGC = glomerular capillary hydrostatic pressure (~60 mmHg), PBS = Bowman's space hydrostatic pressure (~15 mmHg), and πGC = glomerular capillary oncotic pressure (~28 mmHg).
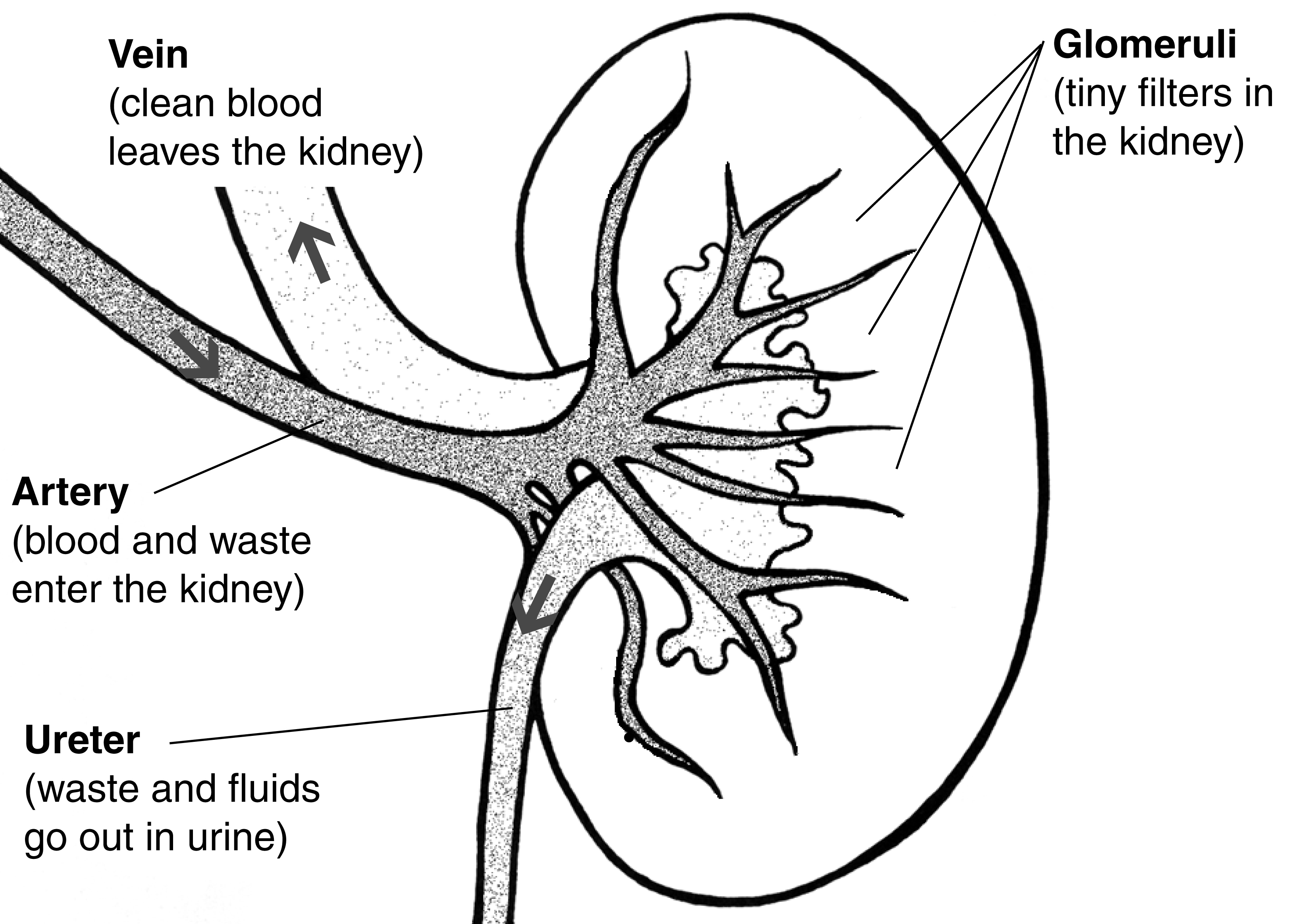
Renal Blood Flow & Autoregulation
Blood enters via the renal artery → segmental → interlobar → arcuate → interlobular arteries → afferent arteriole → glomerular capillaries → efferent arteriole → peritubular capillaries (cortex) or vasa recta (medulla). The kidney autoregulates RBF and GFR over MAP 80–180 mmHg via two mechanisms: myogenic reflex (afferent arteriolar constriction in response to stretch) and tubuloglomerular feedback (macula densa senses NaCl delivery; increased delivery causes afferent constriction via adenosine). ACE inhibitors/ARBs dilate the efferent arteriole, reducing intraglomerular pressure — this is renoprotective long-term but can acutely reduce GFR (especially in bilateral renal artery stenosis).
Countercurrent Mechanism
The countercurrent multiplier (loop of Henle) creates the medullary osmotic gradient (300 mOsm at the corticomedullary junction up to 1,200 mOsm at the papillary tip). The thick ascending limb actively transports NaCl without water, diluting tubular fluid and concentrating the interstitium. The countercurrent exchanger (vasa recta) maintains the gradient by passively exchanging solutes and water in a hairpin arrangement. ADH (vasopressin) inserts aquaporin-2 channels into the collecting duct, allowing water to be reabsorbed along the medullary gradient, producing concentrated urine (up to 1,200 mOsm/kg). Without ADH, dilute urine (<100 mOsm/kg) is excreted.
02 The Renal Physical Exam & Assessment
Volume Status Assessment
| Finding | Hypovolemia | Euvolemia | Hypervolemia |
|---|---|---|---|
| JVP | Flat (<5 cm H₂O) | 5–8 cm H₂O | Elevated (>8 cm H₂O) |
| Mucous membranes | Dry | Moist | Moist |
| Skin turgor | Decreased (tenting) | Normal | Normal or taut |
| Orthostatics | Positive (SBP drop ≥20 or HR rise ≥20) | Negative | Negative |
| Edema | Absent | Absent | Peripheral, sacral, pulmonary |
| Urine output | Oliguria (<400 mL/day) | Normal (800–2000 mL/day) | Variable |
| BUN/Cr ratio | >20:1 | 10–20:1 | Variable |
Edema Grading
| Grade | Depth | Rebound Time | Location |
|---|---|---|---|
| 1+ | 2 mm | Immediate | Feet/ankles |
| 2+ | 4 mm | 15 seconds | Below knee |
| 3+ | 6 mm | 30 seconds | Above knee |
| 4+ | 8+ mm | >30 seconds | Anasarca (generalized) |
Urinalysis Interpretation
| Parameter | Normal | Abnormal Significance |
|---|---|---|
| Color | Pale to dark yellow | Red/brown: hematuria or myoglobin; cola-colored: glomerulonephritis |
| Specific gravity | 1.005–1.030 | <1.005: dilute (DI, water excess); >1.030: concentrated (dehydration); fixed at 1.010: tubular damage |
| pH | 4.5–8.0 | Persistently alkaline: UTI (urease organisms), RTA type 1 |
| Protein | Negative to trace | Detects albumin primarily; false positive with concentrated/alkaline urine |
| Blood | Negative | Positive without RBCs on microscopy: myoglobinuria or hemoglobinuria |
| Glucose | Negative | Positive: DM (glucose >180 mg/dL), Fanconi syndrome, SGLT2 inhibitors |
| Leukocyte esterase | Negative | Suggests pyuria/UTI |
| Nitrites | Negative | Positive: gram-negative bacteria (Enterobacteriaceae) |
Urine Electrolytes & Fractional Excretion
| Test | Prerenal | Intrinsic Renal (ATN) | Interpretation |
|---|---|---|---|
| FENa | <1% | >2% | Most useful in oliguric AKI; unreliable with diuretics |
| FEUrea | <35% | >50% | Preferred when patient is on diuretics |
| Urine Na | <20 mEq/L | >40 mEq/L | Low = avid Na retention (prerenal) |
| Urine osmolality | >500 mOsm/kg | <350 mOsm/kg | High = concentrating ability intact (prerenal) |
| BUN/Cr ratio | >20:1 | 10–15:1 | Elevated in prerenal, GI bleed, steroids |
Urine Microscopy — Key Casts
| Cast Type | Appearance | Associated Condition |
|---|---|---|
| Hyaline | Transparent, low refractive index | Normal (concentrated urine, dehydration); nonspecific |
| Granular ("muddy brown") | Coarse or fine granules, dark | Acute tubular necrosis (ATN) |
| RBC casts | Red/orange, intact RBCs in cast matrix | Glomerulonephritis (pathognomonic) |
| WBC casts | White cells within cast | Pyelonephritis, acute interstitial nephritis |
| Waxy | Broad, waxy, sharply defined edges | Advanced CKD, severe stasis |
| Fatty (Maltese cross) | Fat globules, birefringent under polarized light | Nephrotic syndrome |
03 Key Terminology & Abbreviations
Nephrology employs a dense vocabulary of abbreviations spanning renal physiology, dialysis, transplant, and electrolyte disorders. Mastery of these terms is essential for interpreting renal panels, urinalyses, biopsy reports, and dialysis prescriptions.
| Abbreviation | Meaning |
|---|---|
| GFR / eGFR | Glomerular filtration rate / estimated GFR |
| CrCl | Creatinine clearance |
| BUN | Blood urea nitrogen |
| AKI / CKD / ESRD | Acute kidney injury / chronic kidney disease / end-stage renal disease |
| ATN / AIN | Acute tubular necrosis / acute interstitial nephritis |
| FENa / FEUrea | Fractional excretion of sodium / urea |
| UACR / UPCR | Urine albumin-to-creatinine ratio / urine protein-to-creatinine ratio |
| RRT / KRT | Renal replacement therapy / kidney replacement therapy |
| HD / PD / CRRT | Hemodialysis / peritoneal dialysis / continuous renal replacement therapy |
| AVF / AVG / CVC | Arteriovenous fistula / arteriovenous graft / central venous catheter |
| Kt/V | Dialysis adequacy measure (K=clearance, t=time, V=volume of distribution) |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| RAAS | Renin-angiotensin-aldosterone system |
| ACEi / ARB | ACE inhibitor / angiotensin receptor blocker |
| MRA | Mineralocorticoid receptor antagonist |
| EPO / ESA | Erythropoietin / erythropoiesis-stimulating agent |
| CKD-MBD | CKD-mineral and bone disorder |
| PTH / iPTH | Parathyroid hormone / intact PTH |
| FSGS / MCD / MN | Focal segmental glomerulosclerosis / minimal change disease / membranous nephropathy |
| RPGN | Rapidly progressive glomerulonephritis |
| ANCA / GBM | Anti-neutrophil cytoplasmic antibody / glomerular basement membrane |
| GPA / MPA / EGPA | Granulomatosis with polyangiitis / microscopic polyangiitis / eosinophilic GPA |
| TTP / HUS / aHUS | Thrombotic thrombocytopenic purpura / hemolytic uremic syndrome / atypical HUS |
| ADPKD | Autosomal dominant polycystic kidney disease |
| RTA | Renal tubular acidosis |
| SIADH / DI | Syndrome of inappropriate ADH / diabetes insipidus |
| ADH (AVP) | Antidiuretic hormone (arginine vasopressin) |
04 AKI: Classification & Etiologies Critical
Acute kidney injury (AKI) is defined as an abrupt decline in kidney function over hours to days. It is classified by the KDIGO criteria based on serum creatinine rise and/or urine output decline. AKI occurs in ~10–15% of hospitalized patients and up to 50% of ICU patients, and is independently associated with increased mortality, length of stay, and progression to CKD.
KDIGO AKI Staging
| Stage | Serum Creatinine | Urine Output |
|---|---|---|
| 1 | Rise ≥0.3 mg/dL within 48 h OR 1.5–1.9× baseline within 7 days | <0.5 mL/kg/h for 6–12 h |
| 2 | 2.0–2.9× baseline | <0.5 mL/kg/h for ≥12 h |
| 3 | ≥3.0× baseline OR ≥4.0 mg/dL OR initiation of RRT | <0.3 mL/kg/h for ≥24 h OR anuria for ≥12 h |
AKI Etiologies
| Category | Mechanism | Common Causes | Diagnostic Clues |
|---|---|---|---|
| Prerenal (~55–60%) | Decreased renal perfusion with intact tubular function | Hypovolemia (hemorrhage, dehydration, GI losses), heart failure (cardiorenal), sepsis (early), hepatorenal syndrome, NSAIDs, ACEi/ARB | FENa <1%, UOsm >500, BUN/Cr >20:1, bland sediment |
| Intrinsic (~35–40%) | Direct parenchymal damage to tubules, glomeruli, interstitium, or vessels | ATN (ischemic or nephrotoxic), AIN (drug-induced), glomerulonephritis, TMA, atheroembolic disease | FENa >2%, muddy brown casts (ATN), RBC casts (GN), WBC casts (AIN), eosinophiluria (AIN) |
| Postrenal (~5%) | Urinary tract obstruction | BPH, bilateral ureteral stones, pelvic malignancy, neurogenic bladder, retroperitoneal fibrosis | Hydronephrosis on US, high post-void residual, rapid improvement with decompression |
Type 1 (HRS-AKI): Rapid decline in GFR (Cr doubling to >2.5 mg/dL in <2 weeks) in advanced cirrhosis. Pathophysiology: splanchnic vasodilation → decreased effective circulating volume → intense renal vasoconstriction. Diagnostic criteria: cirrhosis + ascites, AKI without improvement after 48 h of volume expansion with albumin (1 g/kg/day, max 100 g/day) and withdrawal of diuretics, no nephrotoxins, no parenchymal disease. Treatment: midodrine + octreotide + albumin (or norepinephrine in ICU); terlipressin (approved outside US); definitive treatment is liver transplantation. Median survival without treatment: 2 weeks.
05 Acute Tubular Necrosis
ATN is the most common cause of intrinsic AKI, accounting for ~85% of intrinsic renal failure. It results from ischemic or nephrotoxic injury to tubular epithelial cells, particularly in the S3 segment of the proximal tubule and the medullary thick ascending limb — areas with high metabolic demand and borderline oxygen supply.
ATN Classification
| Type | Mechanism | Common Causes |
|---|---|---|
| Ischemic ATN | Prolonged renal hypoperfusion exceeding autoregulatory capacity | Shock (septic, cardiogenic, hemorrhagic), major surgery (especially cardiac with CPB), aortic cross-clamping |
| Nephrotoxic ATN | Direct tubular cell toxicity | Aminoglycosides, cisplatin, amphotericin B, radiocontrast, vancomycin, tenofovir, IV immunoglobulin (sucrose-based) |
Phases of ATN
| Phase | Duration | Features |
|---|---|---|
| Initiation | Hours to days | Insult occurs; rising creatinine, declining urine output |
| Extension | 1–2 days | Ongoing ischemia and inflammation; apoptosis and necrosis |
| Maintenance | 1–2 weeks | Established AKI; oliguric or nonoliguric; complications (hyperkalemia, acidosis, volume overload) |
| Recovery | Days to weeks | Tubular regeneration; polyuric phase (may lose large volumes of water and electrolytes); gradual GFR improvement |
Management
Treatment is primarily supportive: optimize volume status, discontinue nephrotoxins, avoid further hemodynamic insults, manage complications (hyperkalemia, acidosis, fluid overload). There is no proven pharmacologic therapy to accelerate recovery. Initiate RRT for refractory hyperkalemia, severe metabolic acidosis, volume overload unresponsive to diuretics, or uremic complications (encephalopathy, pericarditis, bleeding).
06 Acute Interstitial Nephritis
AIN is a hypersensitivity-mediated inflammation of the renal interstitium, most commonly drug-induced (~70–75% of cases). It accounts for 15–20% of all AKI. The classic triad of fever, rash, and eosinophilia is present in only ~10–15% of cases (more common with methicillin, now rarely used).
Etiologies
| Category | Common Agents / Causes |
|---|---|
| Antibiotics | Penicillins (classic), cephalosporins, sulfonamides (TMP-SMX), fluoroquinolones, rifampin |
| NSAIDs | All NSAIDs (onset may be delayed weeks to months; often with nephrotic-range proteinuria) |
| PPIs | Omeprazole, pantoprazole (increasingly recognized; may have insidious onset) |
| Other drugs | Allopurinol, phenytoin, checkpoint inhibitors (nivolumab, pembrolizumab) |
| Infections | Pyelonephritis, Legionella, CMV, EBV, Mycobacterium tuberculosis |
| Autoimmune | Sarcoidosis, SLE, Sjögren syndrome, IgG4-related disease, TINU syndrome |
Diagnosis & Management
Urinalysis: sterile pyuria, WBC casts, eosinophiluria (detected by Hansel stain; sensitivity only ~60–70%). Mild proteinuria is typical; nephrotic-range proteinuria suggests NSAID-associated AIN with concurrent MCD. Renal biopsy is the gold standard: interstitial edema and inflammatory infiltrate (lymphocytes, eosinophils, monocytes) with tubulitis. Treatment: withdraw offending agent (most important step). If no improvement in 5–7 days, consider prednisone 1 mg/kg/day for 2–4 weeks with taper. Earlier steroid initiation (within 7 days of diagnosis) is associated with better renal recovery.
07 Contrast-Induced & Pigment Nephropathy
Contrast-Induced Nephropathy (CIN)
CIN is defined as a rise in serum creatinine ≥0.5 mg/dL or ≥25% above baseline within 48–72 hours of contrast administration. Mechanism: renal vasoconstriction + direct tubular toxicity + oxidative stress. Risk factors: CKD (eGFR <30 highest risk), diabetes, volume depletion, heart failure, large contrast volumes, concurrent nephrotoxins.
| Prevention Strategy | Evidence |
|---|---|
| IV isotonic saline (1–1.5 mL/kg/h for 6–12 h pre and post) | Strong evidence; cornerstone of prevention |
| Use low-osmolar or iso-osmolar contrast; minimize volume | Contrast volume / eGFR ratio <3.7 associated with lower risk |
| Hold metformin (restart 48 h post if renal function stable) | Prevents lactic acidosis if AKI develops; does not prevent CIN itself |
| NAC (N-acetylcysteine) | PRESERVE trial showed no benefit; not recommended |
| Withhold NSAIDs, diuretics if possible | Reduces additive nephrotoxic/hemodynamic insult |
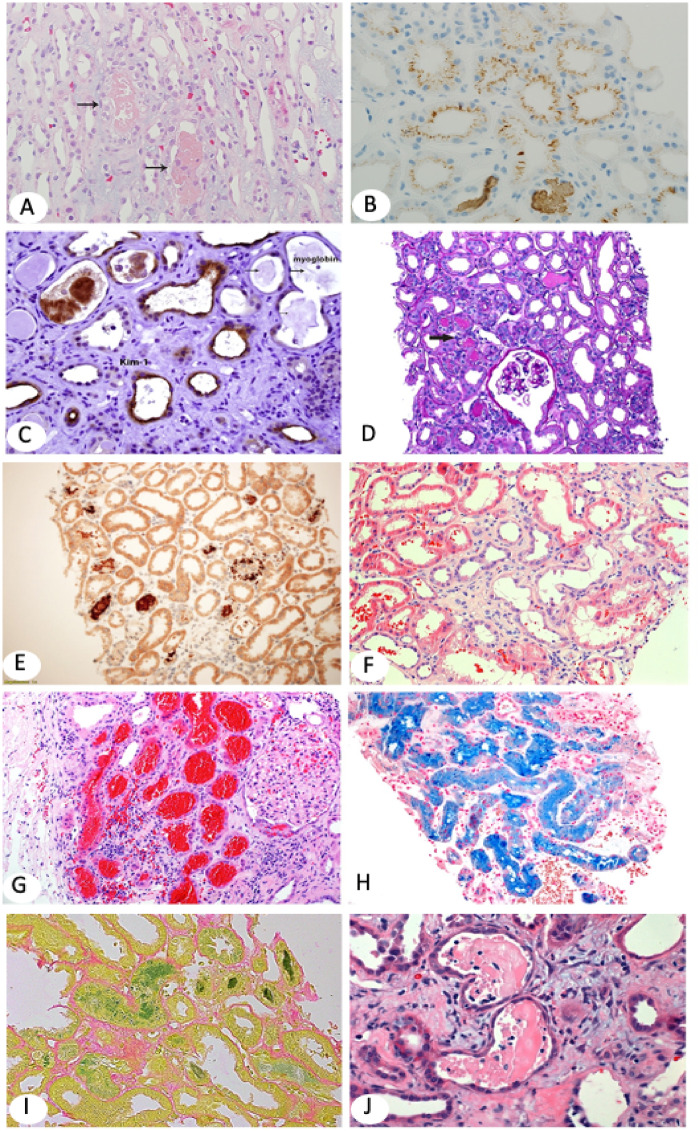
Pigment Nephropathy
Rhabdomyolysis: skeletal muscle breakdown releases myoglobin, which causes AKI via direct tubular toxicity, intratubular cast formation, and renal vasoconstriction. Labs: CK >5,000 U/L (often >10,000), hyperkalemia, hyperphosphatemia, hypocalcemia (early), hyperuricemia, elevated LDH. Urine dipstick positive for blood but no RBCs on microscopy (myoglobin cross-reacts). Treatment: aggressive IV NS (200–300 mL/h targeting UOP 200–300 mL/h); bicarbonate infusion is controversial (may alkalinize urine to prevent myoglobin cast formation, but no RCT evidence of benefit). Monitor for compartment syndrome.
In rhabdomyolysis, the most life-threatening early complication is hyperkalemia from massive intracellular K+ release. Check K+ immediately and prepare for emergency management. Calcium should be given cautiously — it may deposit in damaged muscle, worsening injury. The early hypocalcemia typically self-corrects; avoid aggressive calcium replacement unless symptomatic or severe hyperkalemia requires it.
08 CKD Staging & Progression
CKD is defined as abnormalities of kidney structure or function present for >3 months, with implications for health. The most common causes in the US are diabetes mellitus (~45%) and hypertension (~28%). KDIGO classifies CKD by both GFR category and albuminuria category to determine risk and management.
KDIGO GFR Categories
| Stage | GFR (mL/min/1.73 m²) | Description |
|---|---|---|
| G1 | ≥90 | Normal or high (kidney damage with normal GFR) |
| G2 | 60–89 | Mildly decreased |
| G3a | 45–59 | Mild-to-moderately decreased |
| G3b | 30–44 | Moderate-to-severely decreased |
| G4 | 15–29 | Severely decreased |
| G5 | <15 | Kidney failure (ESRD) |
KDIGO Albuminuria Categories
| Category | UACR (mg/g) | Description |
|---|---|---|
| A1 | <30 | Normal to mildly increased |
| A2 | 30–300 | Moderately increased (formerly "microalbuminuria") |
| A3 | >300 | Severely increased (formerly "macroalbuminuria") |
Slowing CKD Progression
| Intervention | Target / Details | Evidence |
|---|---|---|
| ACEi or ARB | Titrate to max tolerated dose; accept up to 30% Cr rise | RENAAL, IDNT, REIN trials |
| SGLT2 inhibitor | Dapagliflozin 10 mg or empagliflozin 10 mg daily; initiate if eGFR ≥20 | DAPA-CKD, EMPA-KIDNEY, CREDENCE |
| Finerenone (nsMRA) | 10–20 mg daily (requires K+ <5.0, eGFR ≥25) | FIDELIO-DKD, FIGARO-DKD |
| Blood pressure control | <130/80 mmHg (SPRINT); <120 systolic may confer additional benefit | SPRINT, ACCORD-BP |
| Glycemic control (DKD) | HbA1c <7%; avoid hypoglycemia in advanced CKD | DCCT, UKPDS, ADVANCE |
| Dietary protein restriction | 0.8 g/kg/day for CKD 3–5 (not on dialysis) | MDRD study (modest benefit) |
| Avoid nephrotoxins | NSAIDs, aminoglycosides, excess contrast | Universal recommendation |
09 CKD Complications Management
Anemia of CKD
Normocytic normochromic anemia develops primarily from decreased erythropoietin (EPO) production, typically when eGFR <30–45. Iron deficiency is common (reduced absorption, chronic blood loss from uremia-related platelet dysfunction, dialysis losses). Workup: CBC, reticulocytes, iron studies (TSAT and ferritin).
| Parameter | Target in CKD | Treatment |
|---|---|---|
| Iron stores | TSAT >20% and ferritin >100 ng/mL (non-dialysis); ferritin >200 (HD) | IV iron (ferric carboxymaltose, iron sucrose) preferred in HD patients; oral iron for CKD ND |
| Hemoglobin | 10–11.5 g/dL (do NOT target normalization) | ESAs: epoetin alfa 50–100 U/kg SC 3×/week or darbepoetin alfa 0.45 mcg/kg SC q2weeks |
| ESA response | Hb rise 1–2 g/dL over 4 weeks | ESA hyporesponsiveness: check iron stores, infection, inflammation, B12/folate, secondary hyperPTH |
Targeting Hb >13 g/dL with ESAs increases risk of stroke, cardiovascular events, and death (CHOIR, CREATE, TREAT trials). Always replete iron first before initiating ESAs. HIF-PHIs (e.g., roxadustat, daprodustat) are newer oral alternatives that stimulate endogenous EPO production.
CKD-Mineral Bone Disorder (CKD-MBD)
The pathophysiology begins with phosphate retention in CKD → stimulates FGF23 → decreased 1,25(OH)₂D production → hypocalcemia → secondary hyperparathyroidism → bone disease (osteitis fibrosa cystica from high-turnover). Over time, vascular calcification occurs.
| Abnormality | Target | Treatment |
|---|---|---|
| Hyperphosphatemia | Phosphorus 3.5–5.5 mg/dL (CKD 3–5); keep within normal range | Dietary restriction (<800 mg/day); phosphate binders: sevelamer, lanthanum (non-calcium); calcium acetate (limit if Ca elevated) |
| Hypocalcemia | Correct albumin-adjusted calcium; avoid aggressive supplementation | Calcitriol (0.25–0.5 mcg/day) or paricalcitol if PTH elevated |
| Secondary hyperPTH | PTH 2–9× upper limit of normal for CKD G5D | Calcitriol, active vitamin D analogs (paricalcitol, doxercalciferol), cinacalcet (calcimimetic 30–180 mg daily) |
| Vitamin D deficiency | 25(OH)D >30 ng/mL | Ergocalciferol or cholecalciferol supplementation |
Metabolic Acidosis in CKD
Impaired ammoniagenesis and reduced HCO3− regeneration lead to non-anion-gap metabolic acidosis in CKD (typically serum HCO3− 12–20 mEq/L). Acidosis accelerates muscle catabolism, bone demineralization, and CKD progression. Treatment: oral sodium bicarbonate 650–1300 mg TID (target serum HCO3− ≥22 mEq/L). Monitor for volume overload from sodium load.
Hyperkalemia in CKD
Reduced renal K+ excretion, metabolic acidosis, and medications (ACEi/ARB, spironolactone) predispose to hyperkalemia. Chronic management: dietary K+ restriction (<2 g/day), loop diuretics (enhance K+ excretion), potassium binders (patiromer 8.4 g daily or sodium zirconium cyclosilicate (SZC) 10 g TID × 48 h then 5–10 g daily). These newer binders allow continued RAAS inhibitor use.
10 Diabetic Kidney Disease
Diabetic kidney disease (DKD) is the leading cause of ESRD in the US (~45% of new dialysis initiations). It develops in ~30–40% of patients with type 1 or type 2 diabetes. Natural history: glomerular hyperfiltration → microalbuminuria (UACR 30–300) → overt proteinuria (>300) → declining GFR → ESRD (typically over 10–20 years in type 1 DM, often faster in type 2).
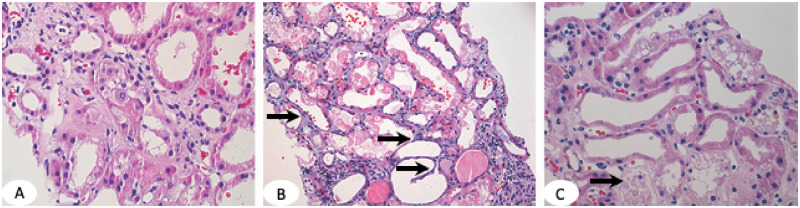
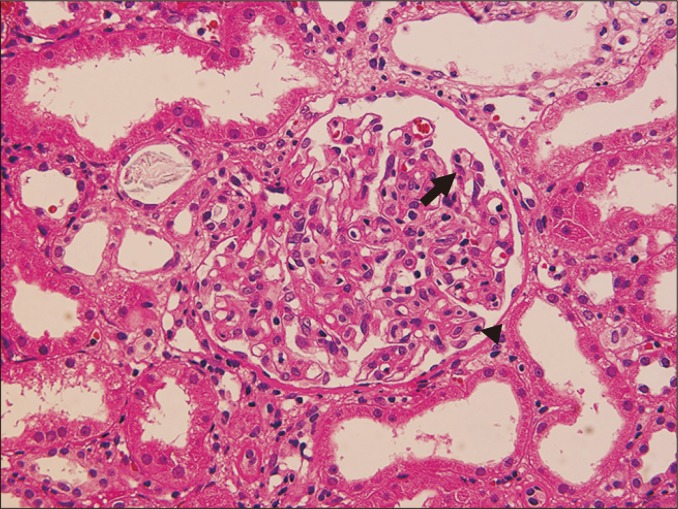
Pathology
Key histologic findings: mesangial expansion (earliest), GBM thickening, Kimmelstiel-Wilson nodules (nodular glomerulosclerosis, pathognomonic but not always present), and diffuse glomerulosclerosis. Afferent and efferent arteriolar hyalinosis is characteristic (afferent alone is seen in hypertension).
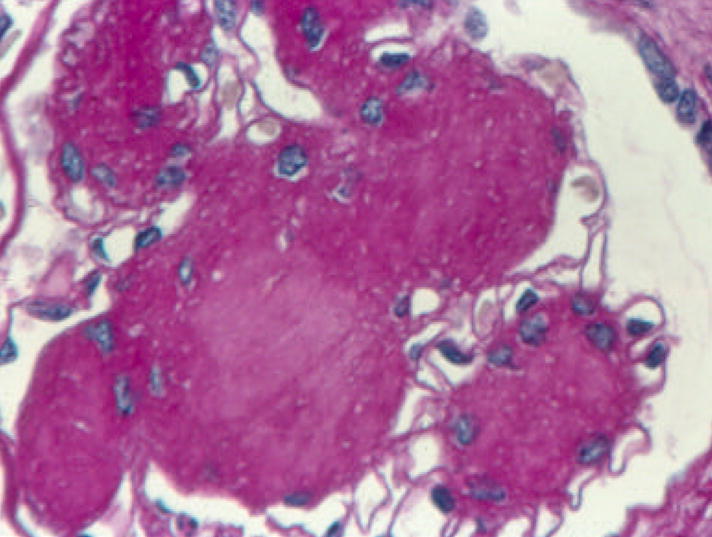
Management of DKD
| Intervention | Dosing / Target | Key Evidence |
|---|---|---|
| ACEi or ARB (first-line) | Titrate to max tolerated dose | RENAAL (losartan), IDNT (irbesartan) — 16–20% reduction in renal endpoints |
| SGLT2 inhibitor | Empagliflozin 10 mg, dapagliflozin 10 mg, canagliflozin 100 mg daily | CREDENCE: 30% reduction in renal composite; DAPA-CKD: 39% reduction; EMPA-KIDNEY: 28% reduction |
| Finerenone (nsMRA) | 10–20 mg daily (add to ACEi/ARB + SGLT2i) | FIDELIO-DKD: 18% reduction in kidney composite; FIGARO-DKD: 13% reduction in CV composite |
| GLP-1 receptor agonist | Semaglutide, liraglutide | FLOW trial (semaglutide): 24% reduction in kidney events |
| Blood pressure | <130/80 mmHg | UKPDS, ACCORD-BP |
| Glycemic control | HbA1c <7% (individualize; <8% in advanced CKD/elderly) | DCCT, UKPDS, ADVANCE |
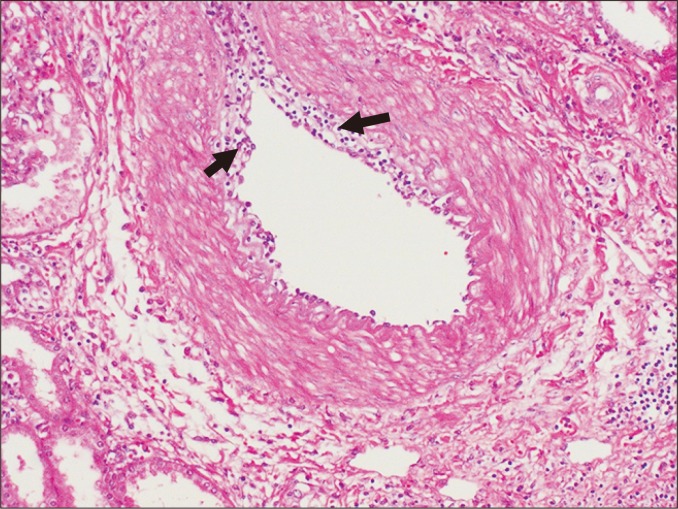
11 Hypertensive Nephrosclerosis
Hypertensive nephrosclerosis is the second leading cause of ESRD in the US. Two forms exist: benign nephrosclerosis (chronic, slowly progressive) and malignant nephrosclerosis (accelerated hypertension with rapid GFR decline).
Benign Nephrosclerosis
Chronic hypertension causes arteriolar hyalinosis and intimal fibrosis of small renal vessels → ischemic injury to glomeruli and tubulointerstitium → global glomerulosclerosis. Clinical features: slowly progressive CKD, mild proteinuria (usually <1 g/day), small kidneys on imaging (bilateral), bland urine sediment. More common and more aggressive in African Americans (APOL1 risk variants).
Malignant Hypertension / Hypertensive Emergency
BP typically >180/120 mmHg with end-organ damage: AKI (fibrinoid necrosis of arterioles, "onion-skinning"), microangiopathic hemolytic anemia (schistocytes), retinopathy (flame hemorrhages, papilledema), encephalopathy. Treatment: IV antihypertensives (nicardipine, nitroprusside, labetalol); reduce MAP by no more than 25% in first hour, then gradually to 160/100 over 24 h.
Variants in the APOL1 gene (G1 and G2 alleles) are carried by ~13% of African Americans and confer a 7–10× increased risk of hypertensive nephrosclerosis, FSGS, and HIV-associated nephropathy. These variants evolved as protection against Trypanosoma brucei (sleeping sickness). APOL1-mediated kidney disease tends to be more aggressive with faster progression to ESRD.
12 Nephrotic Syndrome
Nephrotic syndrome is defined by: proteinuria >3.5 g/day (or UPCR >3,500 mg/g), hypoalbuminemia (<3.0 g/dL), peripheral edema, hyperlipidemia, and lipiduria (fatty casts, oval fat bodies, Maltese crosses). Complications include venous thromboembolism (loss of antithrombin III, protein C, protein S), infections (loss of immunoglobulins), and accelerated atherosclerosis.

Major Causes of Nephrotic Syndrome
| Disease | Epidemiology | Pathology | Key Features | Treatment |
|---|---|---|---|---|
| Minimal Change Disease (MCD) | Most common in children (77%); 10–15% in adults | LM: normal; IF: negative; EM: podocyte foot process effacement | Abrupt nephrotic syndrome, often post-URI or atopy; highly steroid-responsive | Prednisone 1 mg/kg/day × 4–8 weeks then taper; 90% adults remit; relapsers: cyclophosphamide, rituximab, CNIs |
| FSGS | Most common primary GN causing ESRD in African Americans; increasing incidence | LM: segmental sclerosis in some glomeruli; IF: IgM/C3 trapping; EM: foot process effacement | Nephrotic syndrome + hypertension + hematuria; poor steroid response; APOL1-associated; can recur post-transplant (especially collapsing variant) | Prednisone × 4–6 months (only 30–50% respond); CNIs (cyclosporine, tacrolimus); rituximab for relapsing |
| Membranous Nephropathy (MN) | Most common primary nephrotic syndrome in white adults; M:F 2:1 | LM: GBM thickening ("spike and dome"); IF: granular IgG/C3; EM: subepithelial deposits | ~70% have anti-PLA2R antibodies; secondary causes: hepatitis B, malignancy (lung, colon, stomach), SLE, drugs (NSAIDs, gold, penicillamine) | Rule of thirds: 1/3 spontaneous remission, 1/3 stable, 1/3 progress. Immunosuppression for high risk: rituximab (now first-line), cyclophosphamide + steroids (Ponticelli), CNIs |
| Diabetic Nephropathy | Most common cause of nephrotic syndrome overall | Mesangial expansion, GBM thickening, Kimmelstiel-Wilson nodules | Progressive proteinuria in longstanding DM; usually bilateral enlarged kidneys (early); retinopathy in 90% of T1DM | ACEi/ARB + SGLT2i + finerenone + GLP-1 RA; glycemic control |
| Amyloidosis (AL or AA) | AL: plasma cell dyscrasia; AA: chronic inflammatory conditions | Congo red stain: apple-green birefringence under polarized light; amyloid deposits in mesangium and GBM | Nephrotic syndrome + cardiomyopathy + neuropathy (AL); hepatosplenomegaly (AA) | AL: treat underlying plasma cell clone (bortezomib, daratumumab); AA: treat underlying inflammation |
13 Nephritic Syndrome
Nephritic syndrome is characterized by: hematuria (often with RBC casts and dysmorphic RBCs), hypertension, edema, oliguria, and a mild-to-moderate rise in creatinine. Proteinuria is usually subnephrotic (<3.5 g/day) but can overlap. The hallmark on biopsy is inflammatory injury to the glomerulus.
Major Causes of Nephritic Syndrome
| Disease | Pathology | Key Features | Complement | Treatment |
|---|---|---|---|---|
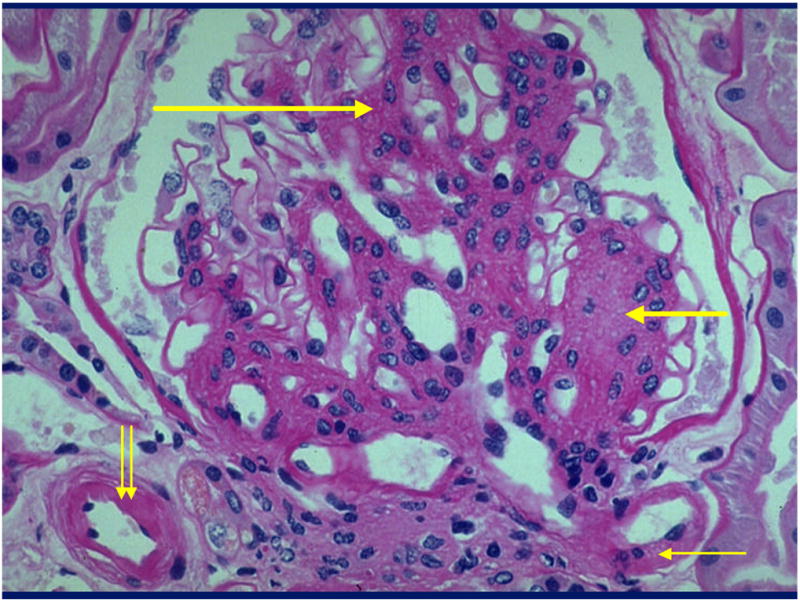
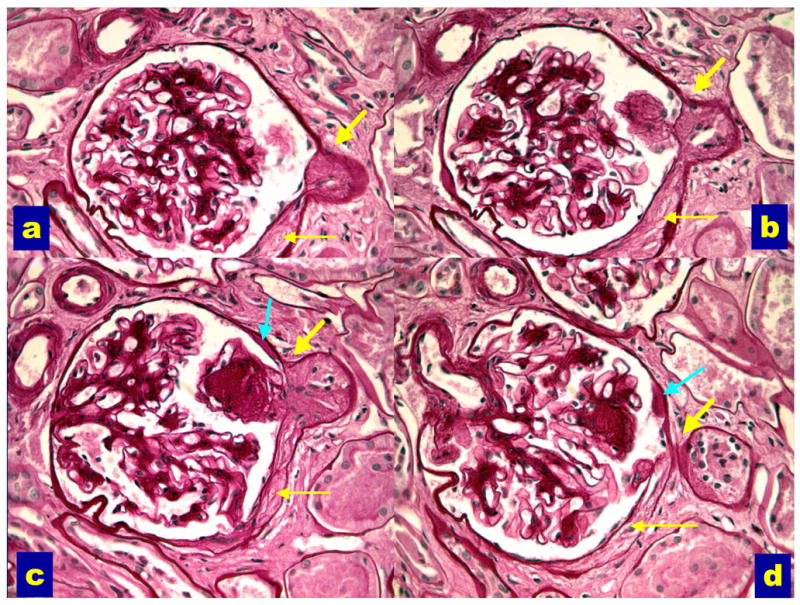
| IgA Nephropathy (Berger disease) | Mesangial IgA deposits (IF); mesangial proliferation (LM) | Most common GN worldwide; episodic gross hematuria 1–2 days after URI ("synpharyngitic"); persistent microscopic hematuria | Normal | ACEi/ARB + SGLT2i; steroids if persistent proteinuria >1 g/day despite 3–6 months RAAS blockade; budesonide (Tarpeyo); consider fish oil |
| Post-Streptococcal GN | Subepithelial "humps" (EM); "lumpy-bumpy" IgG/C3 (IF); diffuse proliferative (LM) | Children 6–10 y/o; 1–3 weeks after pharyngitis or 3–6 weeks after skin infection; cola-colored urine, periorbital edema | Low C3 (normal C4) | Supportive (self-limited in children; >95% recover); adults have worse prognosis |
| RPGN (crescentic GN) | Crescents (>50% of glomeruli) in Bowman's space | Rapidly progressive renal failure over days to weeks; nephritic sediment | Varies by type | Emergency: pulse methylprednisolone + cyclophosphamide ± plasmapheresis; type-specific therapy |
| Anti-GBM disease (Goodpasture) | Linear IgG staining along GBM (IF); crescentic GN (LM) | RPGN + pulmonary hemorrhage (Goodpasture syndrome); anti-GBM antibodies positive; young males (20–30) or elderly (60–70) | Normal | Plasmapheresis (remove circulating antibodies) + cyclophosphamide + steroids; poor prognosis if Cr >5.7 or requiring dialysis at presentation |
| MPGN | GBM double contour ("tram-tracking"); mesangial/endocapillary proliferation | Mixed nephritic-nephrotic features; secondary causes: HCV, cryoglobulinemia, monoclonal gammopathy, C3 glomerulopathy | Low C3 ± C4 | Treat underlying cause; immunosuppression for C3 glomerulopathy |
Type I (anti-GBM) ~10%: linear IgG on IF; anti-GBM antibodies. Type II (immune complex) ~45%: granular IF; includes lupus nephritis, IgA nephropathy, post-infectious GN. Type III (pauci-immune) ~45%: negative/minimal IF; ANCA-positive (GPA, MPA, EGPA). Treatment must be initiated empirically before biopsy results if RPGN is suspected clinically.
14 Lupus Nephritis
Lupus nephritis (LN) occurs in ~50–60% of SLE patients and is a major determinant of morbidity and mortality. Renal biopsy is essential for classification and treatment decisions. The ISN/RPS classification guides therapy.
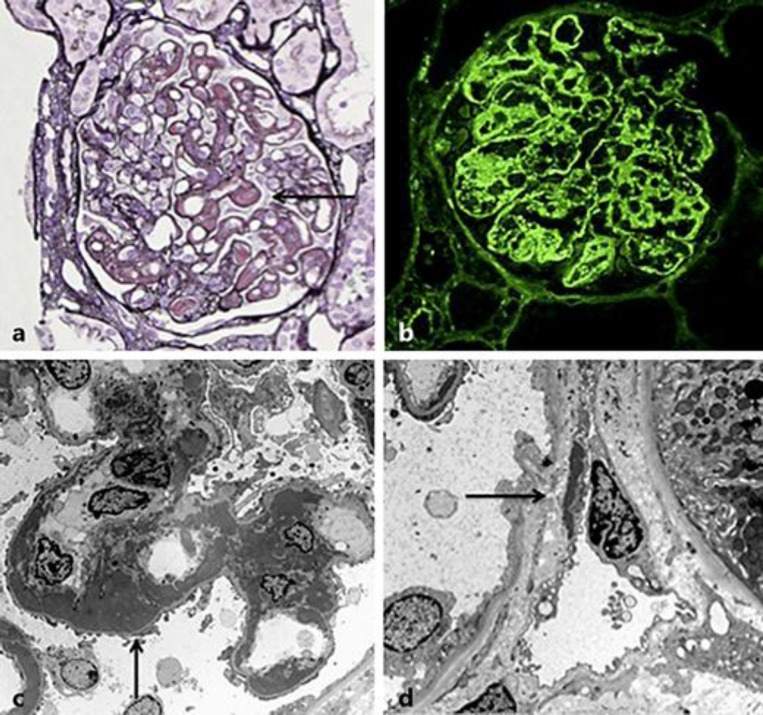
ISN/RPS Classification of Lupus Nephritis (2003/2018 Revised)
| Class | Name | Pathology | Clinical Features | Treatment |
|---|---|---|---|---|
| I | Minimal mesangial | Normal LM; mesangial immune deposits on IF/EM only | Normal UA, normal renal function | Treat extrarenal SLE only |
| II | Mesangial proliferative | Mesangial hypercellularity; mesangial deposits | Microscopic hematuria, mild proteinuria | Treat extrarenal SLE; ACEi/ARB if proteinuria |
| III | Focal (proliferative) | <50% glomeruli with endocapillary ± extracapillary proliferation; subendothelial deposits | Hematuria, proteinuria, possible AKI; active sediment | Induction: mycophenolate mofetil (MMF) or IV cyclophosphamide + steroids; maintenance: MMF or azathioprine |
| IV | Diffuse (proliferative) | ≥50% glomeruli affected; "full house" IF (IgG, IgA, IgM, C3, C1q); wire loop lesions | Nephritic syndrome, heavy proteinuria, hypertension, renal insufficiency; most severe form | Same as III; consider adding belimumab or voclosporin; plasmapheresis if concurrent anti-GBM or TMA |
| V | Membranous | GBM thickening with subepithelial deposits; may coexist with III or IV | Nephrotic syndrome; may have "pure" nephrotic picture | If combined with III/IV: treat as proliferative. Pure V: MMF + steroids; consider rituximab, CNIs |
| VI | Advanced sclerosing | ≥90% globally sclerosed glomeruli | CKD/ESRD; minimal active disease | No immunosuppression; RRT; LN rarely recurs in transplant |
MMF-based (preferred for most): MMF 2–3 g/day + methylprednisolone 500–1000 mg IV × 3 days then oral prednisone 0.5–1 mg/kg/day taper. Euro-Lupus cyclophosphamide: IV CYC 500 mg q2weeks × 6 doses (lower dose than NIH protocol; similar efficacy, less toxicity). Voclosporin (AURORA trial): add to MMF + steroids for improved complete remission rates. Belimumab (BLISS-LN trial): add to standard induction for improved renal response. Maintenance: MMF 1–2 g/day for ≥3–5 years.
15 ANCA-Associated Vasculitis
The ANCA-associated vasculitides (AAV) are small-vessel vasculitides that frequently involve the kidney as pauci-immune crescentic glomerulonephritis (type III RPGN). They include GPA (granulomatosis with polyangiitis), MPA (microscopic polyangiitis), and EGPA (eosinophilic granulomatosis with polyangiitis).
AAV Classification
| Disease | ANCA Pattern | Upper Airway | Lung | Kidney | Unique Features |
|---|---|---|---|---|---|
| GPA (Wegener's) | c-ANCA / anti-PR3 (~80%) | Sinusitis, nasal crusting, saddle nose deformity, subglottic stenosis | Nodules, cavitary lesions, hemorrhage | Crescentic GN (RPGN) | Granulomatous inflammation; "limited" (no renal) or "severe" (renal/lung) |
| MPA | p-ANCA / anti-MPO (~60%) | Rare | Pulmonary hemorrhage (no nodules/cavities) | Crescentic GN (most common AAV renal involvement) | No granulomas; higher proportion of renal-limited disease |
| EGPA (Churg-Strauss) | p-ANCA / anti-MPO (~40%); 60% ANCA-negative | Allergic rhinitis, nasal polyps | Asthma (precedes vasculitis by years), eosinophilic infiltrates | Less frequent (~25%); when present, usually less severe | Peripheral eosinophilia >1,500/μL; cardiac involvement (myocarditis); mononeuritis multiplex |
Renal Management of AAV
Induction: Pulse methylprednisolone (500–1000 mg IV × 3 days) + rituximab 375 mg/m² × 4 weekly doses (RAVE, RITUXVAS trials) OR IV cyclophosphamide. Rituximab is preferred for relapsing disease and PR3-ANCA positive disease. Plasmapheresis: Consider for severe renal disease (Cr >5.7 mg/dL) or concurrent pulmonary hemorrhage; PEXIVAS trial showed no benefit for renal recovery but remains standard for pulmonary hemorrhage. Maintenance: Rituximab 500 mg q6months for ≥2 years (MAINRITSAN); alternatively azathioprine 2 mg/kg/day. Avacopan (C5a receptor inhibitor) can be added to reduce steroid exposure (ADVOCATE trial).
16 Thrombotic Microangiopathies
The thrombotic microangiopathies (TMAs) are characterized by microangiopathic hemolytic anemia (MAHA), thrombocytopenia, and organ injury (especially kidney and brain). The hallmark pathology is endothelial injury with fibrin-platelet thrombi in arterioles and capillaries.
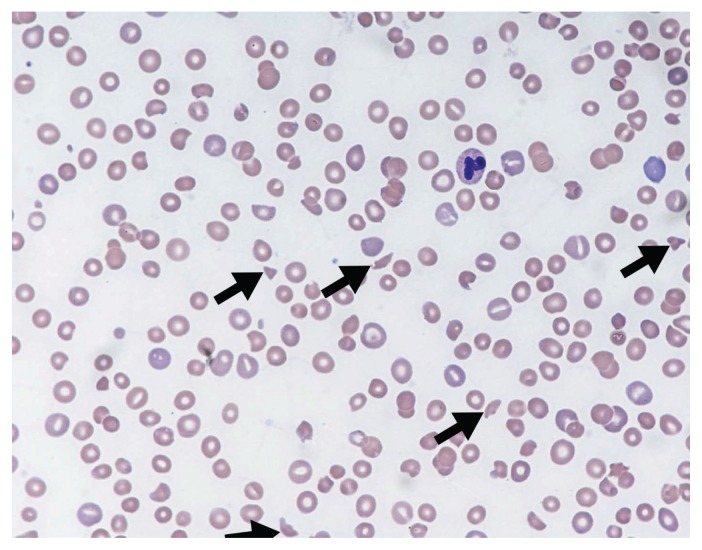
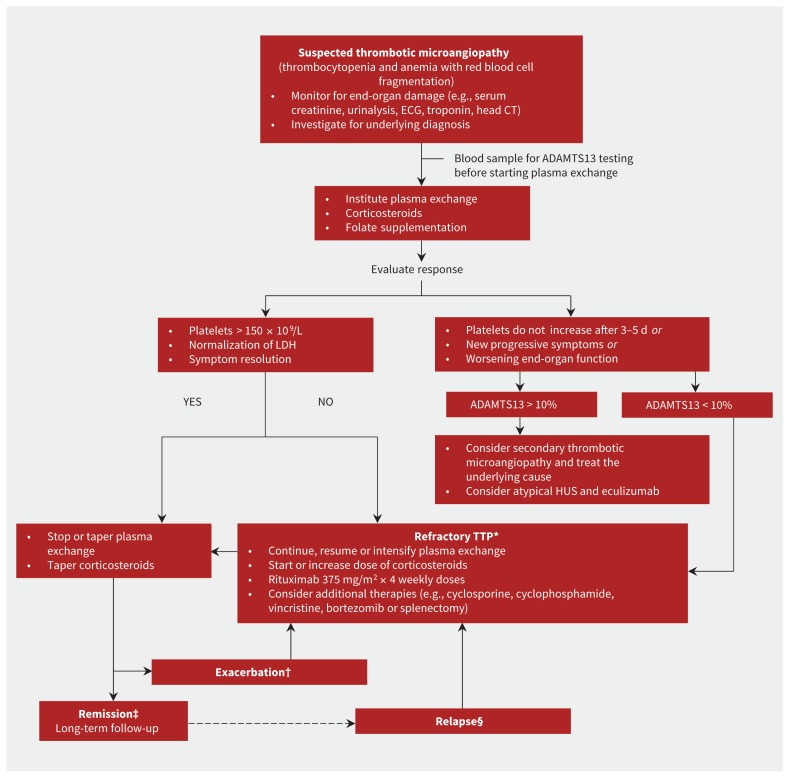
TMA Classification
| Disease | Mechanism | Key Labs | Clinical Features | Treatment |
|---|---|---|---|---|
| TTP | Severe ADAMTS13 deficiency (<10%) — acquired (autoantibody) or congenital | ADAMTS13 activity <10%; schistocytes on smear; LDH elevated; indirect bilirubin elevated; haptoglobin low | Classic pentad (rare): MAHA, thrombocytopenia, AKI, fever, neurologic symptoms; mortality >90% without treatment | Emergent plasma exchange (PEX); do NOT wait for ADAMTS13 results; caplacizumab (anti-vWF); steroids; rituximab for refractory |
| Typical HUS | Shiga toxin-producing E. coli (STEC) O157:H7; toxin causes endothelial injury | Stool culture, Shiga toxin PCR; ADAMTS13 normal | Children (peak 1–5 y/o); bloody diarrhea 5–7 days before MAHA/AKI; often severe AKI requiring dialysis | Supportive care (IV fluids, dialysis PRN); NO antibiotics (may increase toxin release); NO plasma exchange; >90% children recover |
| Atypical HUS (aHUS) | Complement dysregulation (mutations in complement regulators: CFH, CFI, MCP, C3, CFB) | Low C3 (sometimes); normal ADAMTS13; genetic testing for complement mutations | No preceding diarrhea; recurrent TMA episodes; AKI; can occur at any age; high recurrence in transplant | Eculizumab (anti-C5 monoclonal antibody) or ravulizumab (long-acting); meningococcal vaccination required before use |
If TTP is suspected (MAHA + thrombocytopenia + neurologic symptoms), initiate plasma exchange immediately — every hour of delay increases mortality. The PLASMIC score can help differentiate TTP from other TMAs (score ≥6 = high probability of ADAMTS13 <10%). Do NOT transfuse platelets — this can precipitate fatal thrombosis ("fuel on the fire").
17 Polycystic Kidney Disease
Autosomal dominant polycystic kidney disease (ADPKD) is the most common inherited kidney disease (1:400–1,000), caused by mutations in PKD1 (85%, chromosome 16, worse prognosis) or PKD2 (15%, chromosome 4, milder). It accounts for ~5% of ESRD. Bilateral renal cysts develop and enlarge progressively, destroying normal parenchyma.
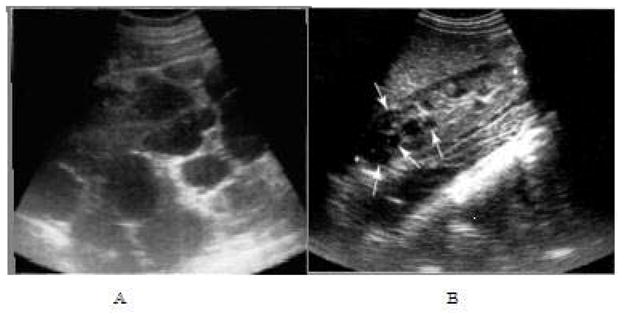
Clinical Features
| Manifestation | Details |
|---|---|
| Renal | Bilateral enlarged kidneys with innumerable cysts; flank/back pain; hematuria; nephrolithiasis (uric acid, calcium oxalate in 20–30%); UTIs and cyst infections; hypertension (onset 20–30s); progressive CKD → ESRD (median age: 54 for PKD1, 74 for PKD2) |
| Hepatic | Hepatic cysts (most common extrarenal manifestation, ~80%); rarely cause liver failure; can cause mass effect |
| Cardiovascular | Mitral valve prolapse (25%); intracranial aneurysms (~8%, screen if family history of SAH or prior to major surgery); aortic root dilation |
| Other | Pancreatic cysts; seminal vesicle cysts; diverticular disease; inguinal and abdominal wall hernias |
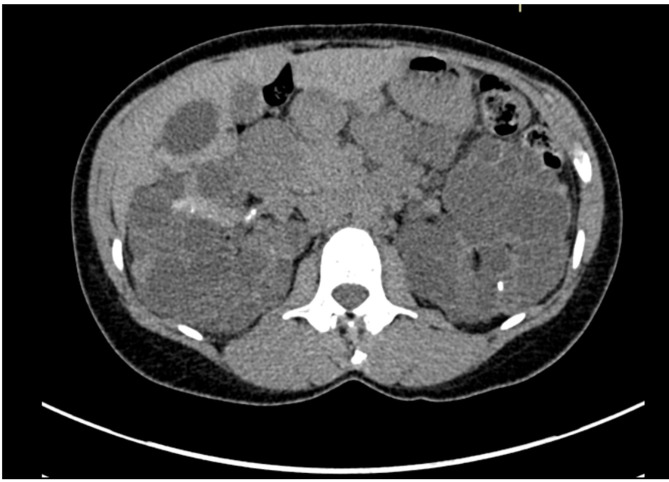
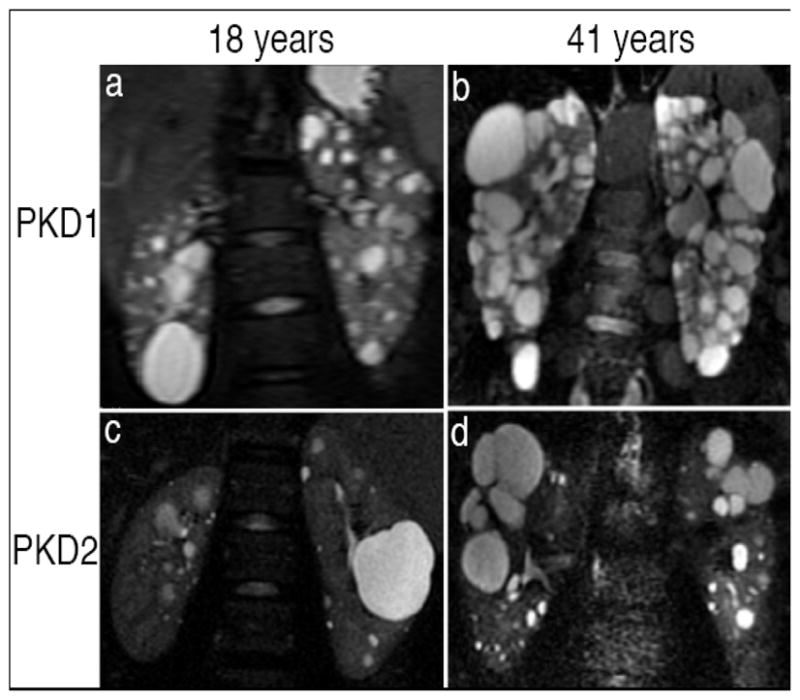
Diagnosis & Management
Diagnosis: Ultrasound criteria (Ravine criteria, age-dependent): age 15–39, ≥3 cysts total; age 40–59, ≥2 cysts per kidney; age ≥60, ≥4 cysts per kidney. MRI is more sensitive for cyst counting and total kidney volume (TKV) measurement, used for Mayo classification (1A–1E) to predict progression.
Tolvaptan (TEMPO 3:4, REPRISE trials): V2 receptor antagonist that slows TKV growth and GFR decline. Indicated for patients at risk of rapid progression (Mayo class 1C–1E, eGFR >25). Dosing: 45/15 mg → titrate to 90/30 mg daily (split AM/PM). Monitoring: liver function tests monthly × 18 months then q3months (risk of hepatotoxicity in ~5%); ensure adequate water intake (2–4 L/day) due to aquaresis. Avoid in pregnancy.
18 Renal Artery Stenosis
Renal artery stenosis (RAS) causes renovascular hypertension and ischemic nephropathy. Two main etiologies exist with distinct demographics and management approaches.
| Feature | Atherosclerotic RAS | Fibromuscular Dysplasia |
|---|---|---|
| Epidemiology | Older adults (>50); men; atherosclerosis risk factors | Young women (15–50 years) |
| Location | Proximal 1/3 of renal artery (ostial) | Mid-to-distal renal artery |
| Pathology | Atherosclerotic plaque | Medial fibroplasia ("string of beads" on angiography) |
| Bilateral | 30–40% | 35% (may also involve carotid, vertebral arteries) |
| Treatment | Medical therapy (ACEi/ARB + statin + antihypertensives); revascularization (stenting) only for refractory HTN, flash pulmonary edema, or progressive CKD | Percutaneous transluminal angioplasty (PTA) WITHOUT stenting; ~90% cure or improvement |
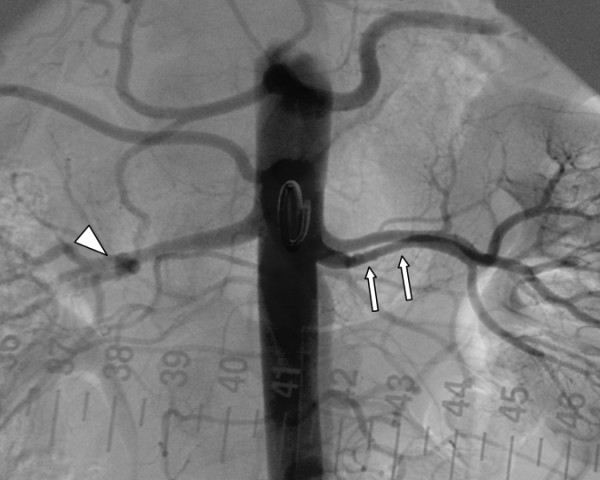
Diagnostic Clues for RAS
Suspect RAS when: resistant hypertension (uncontrolled on ≥3 drugs); flash (recurrent) pulmonary edema with bilateral RAS; AKI after starting ACEi/ARB (Cr rise >30%); asymmetric kidney size (>1.5 cm difference); abdominal bruit. Diagnosis: CTA or MRA (preferred noninvasive); duplex Doppler US (operator-dependent but useful for screening); catheter angiography is the gold standard.
19 Renal Tubular Acidosis
Renal tubular acidosis (RTA) refers to a group of disorders characterized by non-anion-gap metabolic acidosis due to impaired renal acid excretion or bicarbonate reabsorption, with a disproportionately preserved GFR.
RTA Classification
| Feature | Type 1 (Distal) | Type 2 (Proximal) | Type 4 (Hyperkalemic) |
|---|---|---|---|
| Defect | Impaired H+ secretion in collecting duct (alpha-intercalated cells) | Impaired HCO3− reabsorption in proximal tubule | Aldosterone deficiency or resistance |
| Urine pH | >5.5 (cannot acidify urine) | <5.5 (once serum HCO3− below reabsorptive threshold ~15 mEq/L) | <5.5 |
| Serum K+ | Low (hypokalemia) | Low (hypokalemia) | High (hyperkalemia) |
| Serum HCO3− | May be very low (<10 mEq/L) | Moderately low (12–18 mEq/L) | Mildly low (17–22 mEq/L) |
| Complications | Nephrolithiasis (calcium phosphate), nephrocalcinosis, osteomalacia | Osteomalacia, rickets (children), Fanconi syndrome | Hyperkalemia-related cardiac risk |
| Causes | Sjögren, SLE, amphotericin B, toluene, sickle cell, lithium, medullary sponge kidney | Fanconi syndrome (multiple myeloma, tenofovir, ifosfamide, Wilson disease), carbonic anhydrase inhibitors | Diabetic nephropathy (hyporeninemic hypoaldosteronism), ACEi/ARB, spironolactone, TMP, Addison disease, heparin |
| Treatment | Oral sodium bicarbonate or sodium citrate (1–2 mEq/kg/day); potassium citrate if hypokalemic | High-dose bicarbonate (10–15 mEq/kg/day); thiazides may help (enhance proximal reabsorption by volume contraction) | Fludrocortisone (if truly aldosterone deficient); dietary K+ restriction; loop diuretics; discontinue offending drugs; sodium bicarbonate |
20 Sodium Disorders Critical
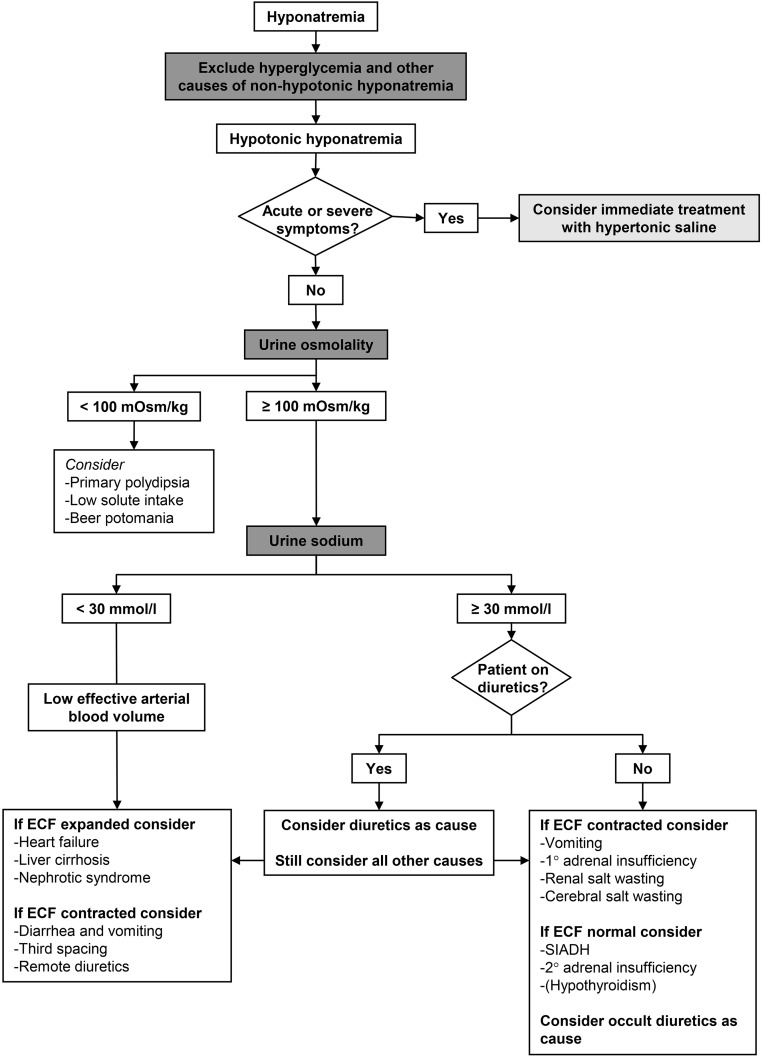
Hyponatremia (Na+ <135 mEq/L)
The most common electrolyte disorder in hospitalized patients (~15–30%). Always assess serum osmolality first to exclude pseudohyponatremia (isotonic: hyperlipidemia/hyperproteinemia) and hypertonic hyponatremia (hyperglycemia: corrected Na+ = measured Na+ + 1.6 × [(glucose − 100)/100]).
| Volume Status | Etiology | Urine Na+ | Treatment |
|---|---|---|---|
| Hypovolemic | GI losses, diuretics, cerebral salt wasting, burns, third-spacing | <20 (extrarenal loss); >20 (renal loss: diuretics, CSW) | Isotonic saline (NS); treats both volume and Na+ |
| Euvolemic | SIADH (most common), hypothyroidism, adrenal insufficiency, psychogenic polydipsia, beer potomania, tea-and-toast | >40 (in SIADH) | Fluid restriction (500–1000 mL/day); salt tablets + loop diuretic; tolvaptan (V2 receptor antagonist); urea |
| Hypervolemic | Heart failure, cirrhosis, nephrotic syndrome, advanced CKD | <20 (HF, cirrhosis); >20 (CKD) | Fluid + salt restriction; loop diuretics; treat underlying cause |
Critical safety limit: Correct Na+ by no more than 8 mEq/L in any 24-hour period (some experts say 6–8 for high-risk patients: alcoholism, malnutrition, liver disease, hypokalemia). Overcorrection causes osmotic demyelination syndrome (ODS) — central pontine myelinolysis: dysarthria, dysphagia, quadriparesis, "locked-in" syndrome. If overcorrecting: administer DDAVP 2 mcg IV q8h + D5W to re-lower Na+. Acute symptomatic hyponatremia (<48 h): can correct faster, give 3% hypertonic saline 100–150 mL bolus over 10–20 min (may repeat ×2); target 4–6 mEq/L rise in first 6 hours.
Hypernatremia (Na+ >145 mEq/L)
Always represents a water deficit. Patients either lost water or cannot access/drink water (ICU patients, elderly, infants).
| Etiology | Mechanism | Urine Osmolality |
|---|---|---|
| Insensible/GI losses | Fever, burns, diarrhea (osmotic: lactulose) | >700 mOsm/kg (appropriate ADH response) |
| Central DI | Decreased ADH production (post-pituitary surgery, trauma, tumors) | <300 mOsm/kg; responds to desmopressin |
| Nephrogenic DI | Kidney resistance to ADH (lithium, hypercalcemia, hypokalemia, demeclocycline) | <300 mOsm/kg; does NOT respond to desmopressin |
| Osmotic diuresis | Glucosuria, mannitol, urea | 300–600 mOsm/kg (inappropriately low for hypernatremia) |
Treatment: Calculate free water deficit = TBW × [(Na+/140) − 1], where TBW = 0.6 × weight (males) or 0.5 × weight (females). Replace with D5W or free water enterally. Correct no faster than 10–12 mEq/L per 24 h (risk of cerebral edema with rapid correction). Central DI: desmopressin (DDAVP) 1–4 mcg IV/SC q12h. Nephrogenic DI: treat underlying cause; thiazides (paradoxical effect), amiloride (for lithium-induced), low-sodium diet.
21 Potassium Disorders Critical
Hyperkalemia (K+ >5.0 mEq/L)
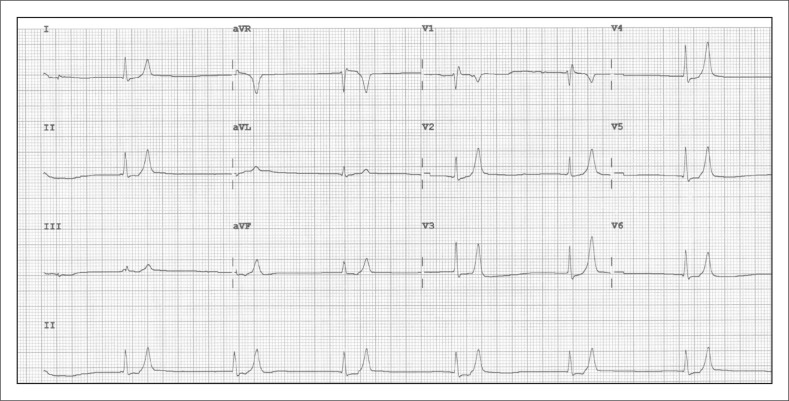
| K+ Level | Severity | EKG Changes (progressive) |
|---|---|---|
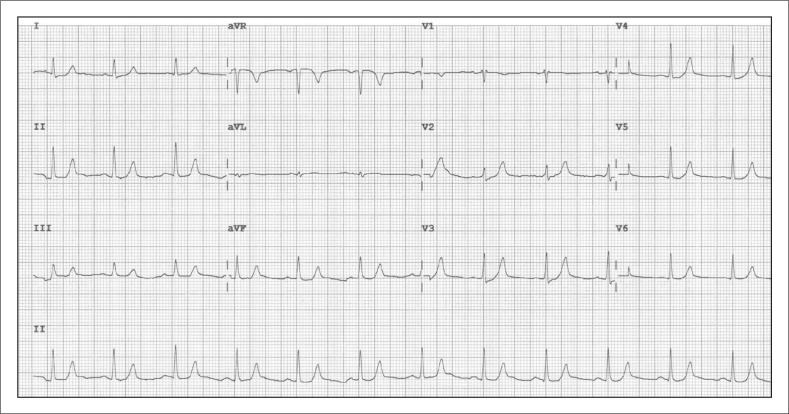
| 5.5–6.0 | Mild | Peaked T waves (earliest sign, best seen in precordial leads) |
| 6.0–7.0 | Moderate | Prolonged PR interval; flattened P waves; widened QRS |
| 7.0–8.0 | Severe | Loss of P waves; further QRS widening; sine wave pattern |
| >8.0 | Life-threatening | Sine wave → ventricular fibrillation → asystole |
Step 1 — Cardiac membrane stabilization: Calcium gluconate 10% 10 mL IV over 2–3 min (or calcium chloride via central line); onset 1–3 min, lasts 30–60 min. Does NOT lower K+ but protects the heart. Repeat if EKG changes persist. Step 2 — Shift K+ intracellularly: Regular insulin 10 units IV + D50W 25 g (to prevent hypoglycemia); onset 15–30 min, lasts 4–6 h. Albuterol 10–20 mg nebulized; onset 15–30 min. Sodium bicarbonate 50 mEq IV (only if acidotic; minimal effect alone). Step 3 — Remove K+ from body: Loop diuretics (furosemide 40–80 mg IV); SZC (sodium zirconium cyclosilicate) 10 g PO; patiromer 8.4 g PO; hemodialysis (definitive for severe/refractory cases). Kayexalate (sodium polystyrene sulfonate) is falling out of favor due to GI necrosis risk and slow onset.
Hypokalemia (K+ <3.5 mEq/L)
Causes: GI losses (diarrhea, vomiting/NG suction — via metabolic alkalosis and renal K+ wasting), renal losses (diuretics, hyperaldosteronism, RTA type 1 and 2, hypomagnesemia), transcellular shifts (insulin, beta-agonists, alkalosis). EKG changes: flattened T waves, ST depression, U waves, prolonged QT, predisposition to arrhythmias (especially with digoxin).
Treatment: Oral KCl 40–80 mEq/day (preferred; each 10 mEq raises K+ ~0.1 mEq/L). IV KCl for severe (<3.0) or symptomatic: max rate 10–20 mEq/h via peripheral line, 40 mEq/h via central line with continuous monitoring. Always check and correct magnesium — hypomagnesemia causes refractory hypokalemia by increasing ROMK-mediated K+ secretion in the collecting duct.
22 Calcium & Phosphorus Disorders
Hypercalcemia
The two most common causes account for ~90%: primary hyperparathyroidism (outpatient) and malignancy (inpatient — via PTHrP, osteolytic metastases, or 1,25(OH)₂D production by lymphoma). Symptoms: "stones, bones, groans, thrones, and psychiatric overtones" (nephrolithiasis, bone pain, abdominal pain/constipation, polyuria, confusion/depression).
| Severity | Corrected Ca2+ (mg/dL) | Management |
|---|---|---|
| Mild | 10.5–12.0 | Hydration; address cause; monitor |
| Moderate | 12.0–14.0 | IV NS (200–300 mL/h); loop diuretics (after volume repleted); calcitonin 4 IU/kg q12h (rapid but tachyphylaxis in 48 h) |
| Severe / symptomatic | >14.0 or symptomatic | Aggressive IV NS; calcitonin; zoledronic acid 4 mg IV (onset 2–4 days, duration weeks) or denosumab; hemodialysis for refractory/life-threatening |
Hypocalcemia
Symptoms: perioral/digital paresthesias, carpopedal spasm, Chvostek sign (facial twitching with tapping CN VII), Trousseau sign (carpal spasm with BP cuff inflation × 3 min), QT prolongation, seizures, laryngospasm. Common causes: hypoparathyroidism (post-surgical most common), vitamin D deficiency, CKD, acute pancreatitis, hyperphosphatemia, hypomagnesemia, hungry bone syndrome (post-parathyroidectomy).
Treatment: Symptomatic/severe: calcium gluconate 1–2 g IV over 10–20 min (10–20 mL of 10% solution) then continuous infusion. Always correct hypomagnesemia. Chronic: oral calcium carbonate/citrate + calcitriol.
Phosphorus Disorders
Hyperphosphatemia: CKD (most common), rhabdomyolysis, tumor lysis syndrome, hypoparathyroidism. Treatment: dietary restriction, phosphate binders (sevelamer, lanthanum, calcium acetate), dialysis. Hypophosphatemia: refeeding syndrome (most dangerous), respiratory alkalosis (shifts into cells), antacids (bind phosphorus), hyperparathyroidism, vitamin D deficiency. Severe (<1.0 mg/dL): IV sodium/potassium phosphate 0.08–0.16 mmol/kg over 6 h.
23 Magnesium Disorders
Hypomagnesemia (Mg2+ <1.8 mg/dL)
Common in hospitalized patients (~12%). Causes: GI losses (diarrhea, malabsorption, PPI use), renal losses (loop/thiazide diuretics, alcohol, aminoglycosides, cisplatin, amphotericin B, calcineurin inhibitors), hungry bone syndrome. Effects: refractory hypokalemia, refractory hypocalcemia (impairs PTH secretion and action), QT prolongation, torsades de pointes.
Treatment: Mild (1.2–1.8): oral magnesium oxide 400–800 mg BID (poorly absorbed; causes diarrhea) or magnesium glycinate/taurate (better tolerated). Severe (<1.2) or symptomatic: magnesium sulfate 1–2 g IV over 1 h; for torsades de pointes: 2 g IV push. Renal excretion is rapid, so sustained repletion requires multiple doses over 24–48 h.
Hypermagnesemia (Mg2+ >2.5 mg/dL)
Almost exclusively in CKD/ESRD patients receiving magnesium-containing medications (antacids, laxatives). Progressive symptoms: hyporeflexia (4–7 mg/dL) → somnolence (7–10) → respiratory depression, cardiac arrest (ECG: prolonged PR, widened QRS, heart block) (>10–12). Treatment: stop Mg-containing agents; IV calcium gluconate (antagonizes cardiac effects); IV saline + furosemide (enhance excretion); hemodialysis for severe cases.
24 Metabolic Acidosis
Metabolic acidosis is defined by low pH (<7.35) with low HCO3− (<22 mEq/L). The first step is to calculate the anion gap (AG) = Na+ − (Cl− + HCO3−); normal = 12 ± 4. Adjust for albumin: corrected AG = AG + 2.5 × (4.0 − albumin).
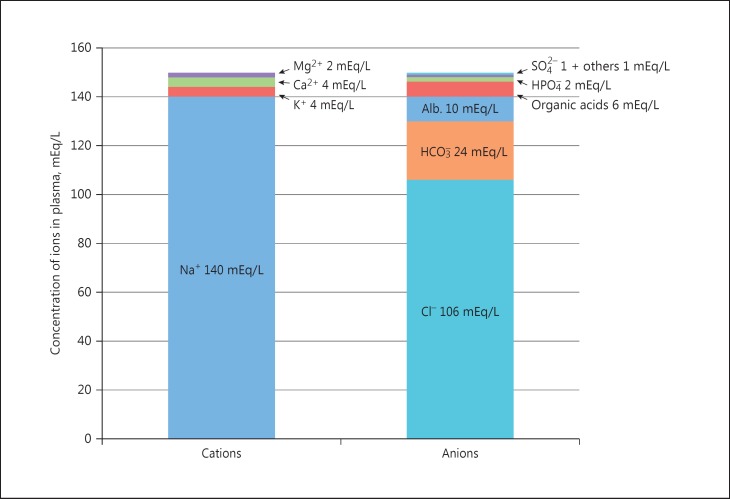
Anion Gap Metabolic Acidosis (AGMA)
Mnemonic: MUDPILES
| Letter | Etiology | Key Features |
|---|---|---|
| M | Methanol | Visual changes ("snowfield" vision), osmol gap elevated, formic acid |
| U | Uremia | Advanced CKD (eGFR <15); retained organic acids |
| D | Diabetic ketoacidosis | Hyperglycemia, ketonemia/ketonuria, AG often >20 |
| P | Propylene glycol / Paraldehyde | IV lorazepam infusion (propylene glycol vehicle), osmol gap elevated |
| I | Isoniazid / Iron | INH: seizures + lactic acidosis; treat with pyridoxine |
| L | Lactic acidosis | Type A: tissue hypoxia (shock, sepsis); Type B: metformin, liver failure, malignancy, thiamine deficiency |
| E | Ethylene glycol | Flank pain, calcium oxalate crystals in urine, osmol gap elevated, AKI |
| S | Salicylates | Mixed AG metabolic acidosis + respiratory alkalosis; tinnitus; ferric chloride test |
Non-Anion Gap Metabolic Acidosis (NAGMA)
AG is normal; the acidosis results from HCO3− loss or impaired acid excretion. Calculate the urine anion gap (UAG) = UNa + UK − UCl.
| UAG | Interpretation | Causes |
|---|---|---|
| Negative (UCl > UNa + UK) | Appropriate renal NH4+ excretion (GI HCO3− loss) | Diarrhea, external pancreatic/biliary fistula, ureteral diversion |
| Positive | Impaired renal acid excretion | RTA (types 1, 2, 4), CKD (early) |
25 Metabolic Alkalosis
Metabolic alkalosis (pH >7.45, HCO3− >28 mEq/L) requires both a generation phase (HCO3− gain or H+ loss) and a maintenance phase (impaired renal HCO3− excretion, usually from volume depletion, chloride depletion, or hypokalemia). Check urine chloride to classify.
| Category | Urine Cl− | Causes | Treatment |
|---|---|---|---|
| Saline-responsive (chloride-responsive) | <20 mEq/L | Vomiting/NG suction (loss of HCl), diuretics (after effect), post-hypercapnia, chloride-losing diarrhea | IV normal saline (provides Cl− for renal HCO3− excretion); KCl replacement; treat underlying cause |
| Saline-resistant (chloride-resistant) | >20 mEq/L | Primary hyperaldosteronism (Conn), Cushing syndrome, renal artery stenosis, Bartter syndrome, Gitelman syndrome, licorice ingestion, Liddle syndrome | Treat underlying cause; spironolactone/eplerenone for mineralocorticoid excess; amiloride for Liddle; acetazolamide (enhances HCO3− excretion) for refractory cases |
26 Hemodialysis
Indications for Dialysis (AEIOU)
| Letter | Indication | Details |
|---|---|---|
| A | Acidosis | Severe metabolic acidosis (pH <7.1) refractory to bicarbonate therapy |
| E | Electrolytes | Refractory hyperkalemia (K+ >6.5 with EKG changes, or resistant to medical therapy) |
| I | Intoxication | Dialyzable toxins: methanol, ethylene glycol, lithium, salicylates, metformin, isopropanol |
| O | Overload | Volume overload refractory to diuretics (pulmonary edema) |
| U | Uremia | Uremic symptoms: encephalopathy, pericarditis, neuropathy, bleeding diathesis |
Vascular Access Types
| Access | Description | Maturation | Advantages | Disadvantages |
|---|---|---|---|---|
| AVF (preferred) | Surgical anastomosis of artery to vein (radiocephalic > brachiocephalic > brachiobasilic) | 2–4 months ("Rule of 6s": flow >600 mL/min, diameter >6 mm, <6 mm deep, >6 cm length) | Lowest infection rate; best patency; best long-term outcomes | Long maturation time; primary failure rate 20–50% (especially radiocephalic) |
| AVG | Synthetic graft (usually PTFE) connecting artery to vein | 2–4 weeks | Faster maturation than AVF; usable in patients with poor native vessels | Higher infection and thrombosis rates than AVF; requires surgical revision |
| CVC (tunneled catheter) | Dual-lumen catheter (internal jugular preferred; avoid subclavian if possible) | Immediate | Immediate use; no maturation needed; bridge to permanent access | Highest infection rate (catheter-related bloodstream infection); central venous stenosis risk; lower blood flow rates |
Dialysis Adequacy
Kt/V is the standard measure: K = dialyzer clearance, t = treatment time, V = urea distribution volume. Target: Kt/V ≥1.4 per session (KDOQI); minimum adequate: Kt/V ≥1.2. URR (urea reduction ratio) = (pre-BUN − post-BUN) / pre-BUN × 100; target ≥65%. Standard prescription: 3–4 h sessions, 3×/week.
27 Peritoneal Dialysis
Peritoneal dialysis (PD) uses the peritoneal membrane as the dialysis membrane. Dialysate is instilled into the peritoneal cavity via a Tenckhoff catheter; solutes diffuse from blood to dialysate, and water is removed by osmotic ultrafiltration (using dextrose or icodextrin in dialysate).
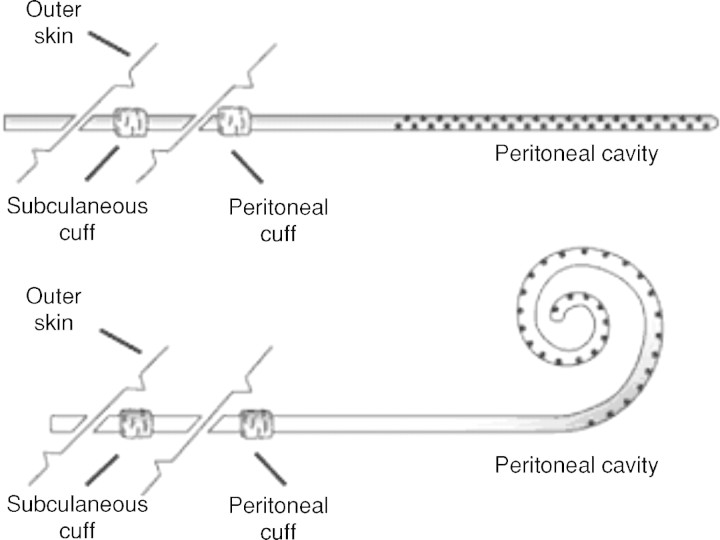
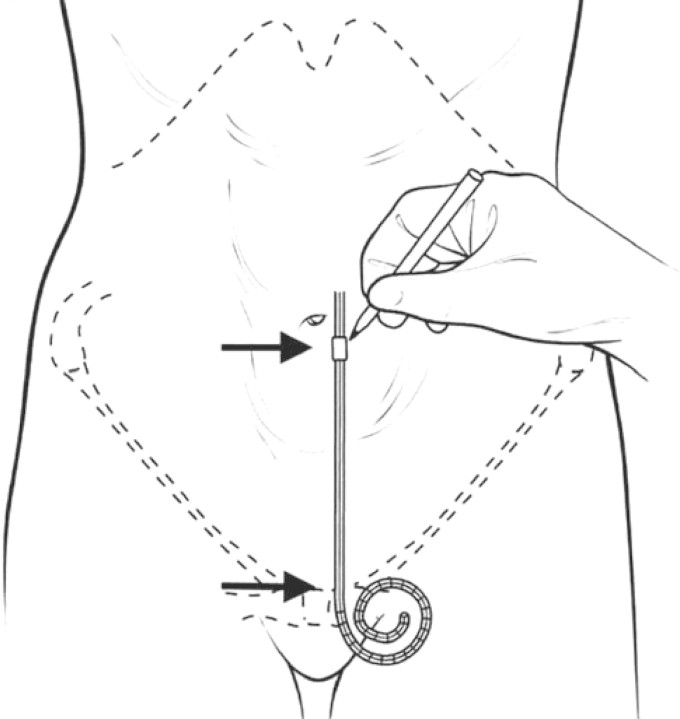
PD Modalities
| Modality | Description | Typical Prescription |
|---|---|---|
| CAPD (Continuous Ambulatory PD) | Manual exchanges, no machine; 4–5 exchanges/day with 4–6 h dwells | 2–2.5 L exchanges × 4/day |
| APD (Automated PD / CCPD) | Machine (cycler) performs exchanges overnight; patient free during day | 10–15 L total over 8–10 h overnight ± daytime dwell |
PD Adequacy
Weekly Kt/V target ≥1.7 (total: peritoneal + residual renal function). PET (peritoneal equilibration test) classifies membrane transport: high transporters (rapid solute equilibration, poor ultrafiltration — best for APD with short dwells), low transporters (slow solute clearance, excellent ultrafiltration — best for CAPD with long dwells).
PD Peritonitis
Diagnosed by ≥2 of 3: abdominal pain/cloudy effluent, WBC >100/μL in effluent (with >50% PMNs), positive effluent culture. Most common organisms: coagulase-negative Staphylococcus (~30%), S. aureus (~15%), gram-negatives (~20%). Treatment: empiric intraperitoneal antibiotics covering gram-positive (vancomycin or cefazolin) + gram-negative (ceftazidime or gentamicin). Catheter removal for: refractory peritonitis (>5 days without response), fungal peritonitis, relapsing peritonitis, or tunnel infection with peritonitis.
28 Continuous Renal Replacement Therapy
CRRT provides slow, continuous solute clearance and fluid removal over 24 hours, making it ideal for hemodynamically unstable ICU patients who cannot tolerate intermittent HD. It requires ICU-level monitoring and continuous anticoagulation of the circuit.
CRRT Modalities
| Modality | Mechanism | Primary Clearance | Replacement Fluid |
|---|---|---|---|
| CVVH (continuous veno-venous hemofiltration) | Convection (large-volume ultrafiltration with replacement fluid) | Middle molecules + small solutes | Pre- or post-filter replacement |
| CVVHD (continuous veno-venous hemodialysis) | Diffusion (dialysate flows countercurrent to blood) | Small solutes (urea, creatinine, K+) | Dialysate only |
| CVVHDF (continuous veno-venous hemodiafiltration) | Convection + diffusion (combined) | Both small and middle molecules | Replacement fluid + dialysate |
CRRT Prescribing
Recommended dose: 20–25 mL/kg/h of effluent (RENAL, ATN trials showed no benefit from higher doses). Anticoagulation: regional citrate anticoagulation (preferred; chelates calcium in the circuit; calcium infused back into the patient) or systemic heparin. Citrate contraindicated in severe liver failure (impaired citrate metabolism → metabolic alkalosis, ionized hypocalcemia). Monitor ionized calcium, acid-base, electrolytes q4–6h.
CRRT advantages: better hemodynamic stability, more precise fluid management, continuous drug clearance (important for antibiotics). Disadvantages: requires ICU stay, continuous anticoagulation, immobilization, higher cost, nurse-intensive. Major RCTs (RENAL, ATN) show no mortality difference between CRRT and intermittent HD in AKI, but CRRT is preferred for hemodynamically unstable patients.
29 Dialysis Emergencies
Dialysis Disequilibrium Syndrome
Occurs during or shortly after first HD sessions in severely uremic patients (BUN >150 mg/dL). Mechanism: rapid urea removal from blood creates an osmotic gradient between plasma and brain → cerebral edema. Symptoms: headache, nausea, confusion, seizures, rarely fatal herniation. Prevention: short initial sessions (2 h), slow blood flow rates (200 mL/min), use high-sodium dialysate, consider mannitol infusion. Treatment: stop dialysis, mannitol, supportive care.
Access-Related Emergencies
| Emergency | Presentation | Management |
|---|---|---|
| Access thrombosis | Loss of thrill/bruit in AVF/AVG; inability to dialyze | Urgent thrombectomy (surgical or pharmacomechanical); catheter placement for next HD |
| Access infection | Erythema, warmth, tenderness; fever, positive blood cultures | AVF: antibiotics (usually IV vancomycin); AVG: may need partial/complete graft excision if infected; CVC: exchange over wire vs removal + new site |
| Access hemorrhage | Bleeding from puncture sites or aneurysmal dilation | Direct pressure; protamine if post-heparin; surgical evaluation for pseudoaneurysm |
| Air embolism | Dyspnea, chest pain, "mill-wheel" murmur, cardiovascular collapse | Clamp catheter; left lateral decubitus + Trendelenburg position; aspirate air via catheter; 100% O2; hyperbaric O2 if available |
Flash pulmonary edema in a dialysis patient is a medical emergency. If the patient missed dialysis: emergent hemodialysis (or CRRT if unstable). Bridge therapy while arranging HD: high-dose IV nitroglycerin (reduces preload), BIPAP/CPAP, IV furosemide 200–400 mg (may have minimal effect in anuric patients). Consider continuous ultrafiltration. If bilateral renal artery stenosis is suspected (recurrent flash pulmonary edema between dialysis sessions in a patient with normal EF), evaluate with renal artery imaging.
30 Pre-Transplant Evaluation & Listing
Kidney transplantation is the optimal renal replacement therapy for eligible ESRD patients, offering superior survival, quality of life, and cost-effectiveness compared to dialysis. Pre-emptive transplantation (before dialysis initiation) has the best outcomes.
Evaluation Workup
| Domain | Assessment |
|---|---|
| Cardiovascular | EKG, echocardiogram; stress testing if ≥3 risk factors or prior cardiac history; coronary angiography if indicated |
| Malignancy screening | Age-appropriate cancer screening; 2–5 year cancer-free interval required for most malignancies |
| Infections | Hepatitis B/C, HIV, CMV, EBV, TB (quantiferon), syphilis, toxoplasma; vaccination update (live vaccines BEFORE transplant) |
| Immunologic | ABO blood typing, HLA typing, PRA (panel reactive antibody — measures degree of sensitization); crossmatch |
| Urologic | Voiding cystourethrogram if bladder dysfunction suspected; native nephrectomy if recurrent infections or massive PKD |
| Psychosocial | Adherence assessment, substance abuse screening, social support evaluation |
Allocation & Donor Types
Deceased donor: UNOS allocation based on EPTS (Estimated Post-Transplant Survival) score and KDPI (Kidney Donor Profile Index). Higher KDPI kidneys have shorter expected graft survival. Living donor: ABO-compatible or ABO-incompatible (with desensitization); paired kidney exchange for incompatible pairs. Living donor transplants have superior outcomes (graft half-life ~15–20 years vs 10–12 years for deceased donor).
31 Immunosuppression Protocols
Induction Therapy (perioperative)
| Agent | Mechanism | Indication | Key Side Effects |
|---|---|---|---|
| Basiliximab | Anti-IL-2R (CD25) monoclonal antibody; blocks T-cell proliferation | Low immunologic risk patients | Generally well-tolerated; low infection risk |
| Anti-thymocyte globulin (ATG) | Polyclonal antibody causing T-cell depletion | High immunologic risk (high PRA, repeat transplant, AA recipients) | Leukopenia, thrombocytopenia, serum sickness, cytokine release syndrome; increased infection and malignancy risk |
| Alemtuzumab | Anti-CD52; depletes T and B cells | Some steroid-avoidance protocols | Profound lymphopenia; increased infection risk |
Maintenance Immunosuppression (Triple Therapy Standard)
| Agent | Mechanism | Typical Dose | Monitoring | Key Toxicities |
|---|---|---|---|---|
| Tacrolimus (FK506) | Calcineurin inhibitor; blocks IL-2 transcription | 0.05–0.1 mg/kg BID; target trough 8–12 ng/mL (early), 5–8 (maintenance) | Trough levels; renal function; glucose; K+; Mg2+ | Nephrotoxicity, diabetes (NODAT), tremor, hyperkalemia, hypomagnesemia, posterior reversible encephalopathy |
| Mycophenolate mofetil (MMF) | IMPDH inhibitor; blocks purine synthesis in lymphocytes | 1000 mg BID (or mycophenolic acid 720 mg BID) | CBC (for leukopenia); GI symptoms | GI toxicity (diarrhea, nausea), leukopenia, teratogenic (pregnancy category X) |
| Prednisone | Anti-inflammatory; suppresses multiple immune pathways | Taper from 30 mg to 5 mg daily by 3–6 months; some protocols withdraw completely | Glucose; bone density; BP | Diabetes, osteoporosis, weight gain, cataracts, avascular necrosis, adrenal suppression |
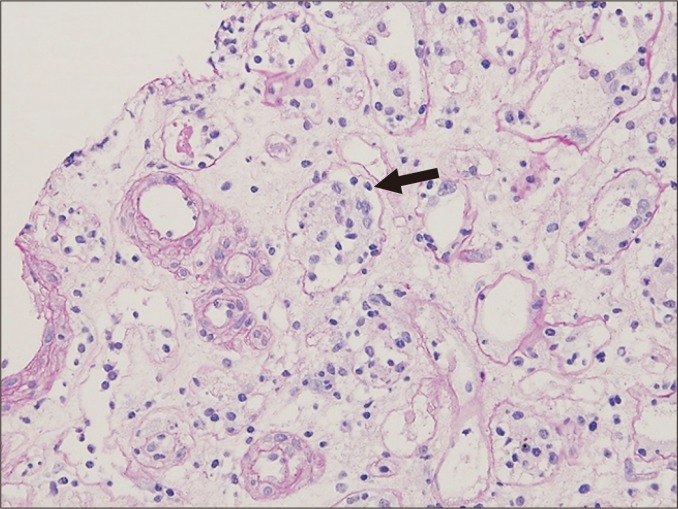
32 Transplant Complications
Rejection Types
| Type | Timing | Mechanism | Biopsy Findings | Treatment |
|---|---|---|---|---|
| Hyperacute | Minutes to hours | Preformed donor-specific antibodies (ABO or HLA); complement activation | Fibrin thrombi, neutrophilic infiltration, cortical necrosis | Nephrectomy (irreversible); prevented by crossmatch testing |
| Acute T-cell mediated (TCMR) | Days to months (usually first 6 months) | T-cell infiltration of tubules and interstitium (tubulitis) | Banff classification: tubulitis + interstitial inflammation; graded IA, IB, IIA, IIB, III | Pulse methylprednisolone 500 mg × 3 days; ATG for steroid-resistant rejection |
| Acute antibody-mediated (ABMR) | Days to years | Donor-specific antibodies (DSA) against HLA; complement-mediated endothelial injury | C4d staining of peritubular capillaries; microvascular inflammation; glomerulitis | Plasmapheresis + IVIG ± rituximab; increase baseline immunosuppression; eculizumab for refractory |
| Chronic (CAMR/IFTA) | Months to years | Both immune (chronic ABMR) and non-immune (CNI toxicity, recurrent disease) factors | Interstitial fibrosis and tubular atrophy (IF/TA); transplant glomerulopathy (double contours) | No proven treatment for chronic changes; optimize immunosuppression; minimize CNI; control BP and proteinuria |
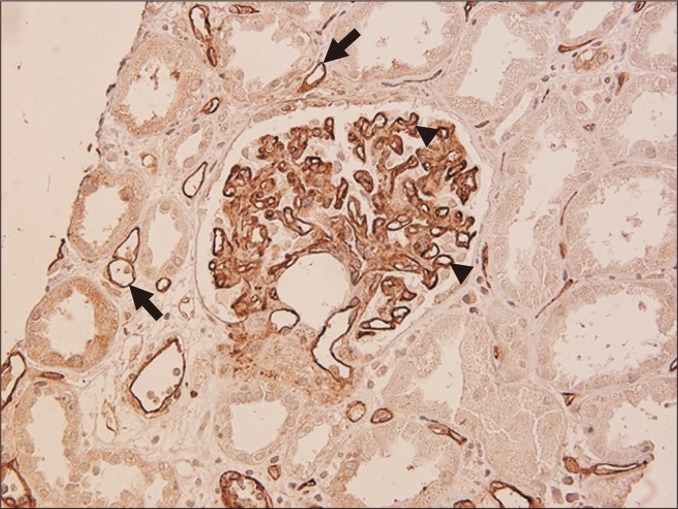
Infectious Complications (Timeline)
| Period | Infections | Prophylaxis |
|---|---|---|
| 0–1 month | Surgical site infections, UTI, C. difficile, donor-derived infections | Perioperative antibiotics |
| 1–6 months | CMV, BK virus (nephropathy), PJP (Pneumocystis), EBV, fungal | Valganciclovir (CMV) × 3–6 months; TMP-SMX (PJP) × 6–12 months |
| >6 months | Community-acquired infections; opportunistic infections if over-immunosuppressed; PTLD (EBV-driven) | Annual influenza vaccine; continued monitoring |
BK Virus Nephropathy
BK polyomavirus reactivation occurs in ~5–10% of transplant recipients (due to immunosuppression), causing BK nephropathy with graft dysfunction. Screen with BK viremia (PCR) monthly for first 6 months, then q3months through 2 years. Viremia >10,000 copies/mL suggests active nephropathy. Treatment: reduce immunosuppression (first step — typically decrease MMF by 50% or switch tacrolimus to lower target); cidofovir and IVIG have limited evidence. No antiviral with proven efficacy.
Post-Transplant Lymphoproliferative Disorder (PTLD)
EBV-driven lymphoproliferation occurring in ~1–3% of adult kidney transplant recipients (higher in EBV-seronegative recipients). Spectrum ranges from benign polyclonal B-cell proliferation to aggressive monomorphic lymphoma (usually diffuse large B-cell). Treatment: reduce immunosuppression (may be curative for early lesions); rituximab for CD20+ disease; chemotherapy (R-CHOP) for advanced/monomorphic PTLD.
33 Kidney Biopsy
Indications
Unexplained AKI, nephrotic syndrome in adults, nephritic syndrome, rapidly progressive GN, persistent proteinuria >1 g/day, unexplained hematuria with proteinuria, systemic disease with renal involvement (SLE, vasculitis, amyloid), transplant dysfunction (to differentiate rejection from other causes).
Contraindications
| Absolute | Relative |
|---|---|
| Uncontrolled bleeding diathesis (INR >1.5, platelets <50,000) | Uncontrolled hypertension (correct to <160/90 before biopsy) |
| Solitary functioning kidney (unless transplant biopsy) | Small kidneys (<9 cm) suggesting advanced CKD (findings unlikely to change management) |
| Active renal or urinary tract infection | Obesity (technical difficulty) |
| Uncooperative patient | Multiple bilateral cysts (ADPKD) |
Performed percutaneously under ultrasound guidance with the patient prone. Target the lower pole of the left kidney (or the transplant kidney in the iliac fossa). Use a 16- or 18-gauge automated spring-loaded biopsy needle. Obtain 2 cores (adequate if ≥10 glomeruli for LM). Specimens sent for: light microscopy (H&E, PAS, trichrome, silver stain), immunofluorescence (IgG, IgA, IgM, C3, C1q, kappa, lambda, fibrinogen), and electron microscopy. Post-procedure: bed rest × 6–8 h, serial vitals and Hb checks.
Complications
Gross hematuria (~3–5%); perinephric hematoma (~15% by imaging, most clinically insignificant); need for blood transfusion (~1–2%); need for angiographic intervention (<0.5%); nephrectomy (extremely rare, <0.01%); death (<0.02%).
34 Urinalysis & Urine Studies
Complete UA Interpretation
The urinalysis is the "poor man's kidney biopsy" and remains the most important initial test in nephrology. A complete UA includes: dipstick analysis (chemical), microscopy (cellular elements, casts, crystals), and quantitative studies (protein, albumin).
Urine Crystals
| Crystal | Appearance | Urine pH | Clinical Significance |
|---|---|---|---|
| Calcium oxalate (monohydrate) | Dumbbell or oval-shaped | Acidic | Ethylene glycol poisoning; hyperoxaluria; most common kidney stone type |
| Calcium oxalate (dihydrate) | Envelope or bipyramidal | Any | Often incidental; hypercalciuria |
| Uric acid | Rhomboid or rosette; yellow-brown | Acidic (<5.5) | Gout, tumor lysis syndrome, uric acid nephrolithiasis |
| Triple phosphate (struvite) | "Coffin lid" shape | Alkaline (>7.0) | UTI with urease-producing organisms (Proteus); staghorn calculi |
| Cystine | Hexagonal plates | Acidic | Cystinuria (autosomal recessive); recurrent stones |
| Calcium phosphate | Wedge-shaped prisms; amorphous | Alkaline | RTA type 1; hyperparathyroidism |
Quantifying Proteinuria
| Method | Normal | Significance |
|---|---|---|
| Dipstick protein | Negative to trace | Semi-quantitative; detects albumin only; misses light chains (Bence Jones) |
| UACR (spot) | <30 mg/g | Preferred screening for DKD; 30–300 = moderately increased; >300 = severely increased |
| UPCR (spot) | <150 mg/g | Approximates 24-h protein excretion (UPCR of 3,500 ≈ 3.5 g/day) |
| 24-h urine protein | <150 mg/day | Gold standard but cumbersome; >3,500 mg = nephrotic range |
| Sulfosalicylic acid (SSA) test | Negative | Detects all proteins including light chains; positive dipstick + negative SSA = false positive; negative dipstick + positive SSA = light chains |
35 Pregnancy & the Kidney
Normal Renal Changes in Pregnancy
GFR increases ~50% by second trimester (hyperfiltration) → serum creatinine normally decreases to 0.4–0.6 mg/dL. A "normal" Cr of 0.9 mg/dL may indicate renal impairment in pregnancy. Physiologic hydronephrosis (R > L, due to dextrorotated uterus compressing right ureter) is common and does not require treatment. Mild proteinuria up to 300 mg/day is normal.
Preeclampsia
Affects 5–8% of pregnancies; defined as new hypertension (≥140/90 after 20 weeks gestation) + proteinuria (≥300 mg/day) OR end-organ damage (thrombocytopenia, elevated LFTs, creatinine >1.1, cerebral/visual symptoms). Pathophysiology: abnormal placentation → placental ischemia → anti-angiogenic factors (sFlt-1) → endothelial dysfunction. Renal pathology: glomerular endotheliosis. HELLP syndrome: Hemolysis, Elevated Liver enzymes, Low Platelets — a severe variant. Treatment: delivery (definitive); magnesium sulfate (seizure prophylaxis); antihypertensives (labetalol, nifedipine, hydralazine). Prevention: low-dose aspirin (81 mg daily from 12–16 weeks) in high-risk women.
CKD in Pregnancy
| CKD Stage | Risk | Key Considerations |
|---|---|---|
| CKD 1–2 (eGFR >60) | Moderate | Generally favorable outcomes; increased monitoring; switch ACEi/ARB to labetalol/nifedipine (ACEi/ARB are teratogenic) |
| CKD 3–4 | High | 30–40% risk of preeclampsia; accelerated CKD progression; preterm delivery in ~50% |
| CKD 5 / Dialysis | Very high | Fertility reduced; if pregnant on dialysis: intensified HD (6×/week, target BUN <50); multidisciplinary management |
ACE inhibitors / ARBs: fetotoxic (renal dysgenesis, oligohydramnios, skull defects) — discontinue immediately upon confirmed pregnancy. MMF: teratogenic (pregnancy category X); switch to azathioprine ≥6 weeks before conception. Cyclophosphamide, methotrexate, mTOR inhibitors: all contraindicated. Safe immunosuppressants in pregnancy: azathioprine, calcineurin inhibitors (tacrolimus, cyclosporine), prednisone.
36 Imaging in Nephrology
| Modality | Indication | Advantages | Limitations |
|---|---|---|---|
| Renal Ultrasound | First-line for all renal pathology; AKI (rule out obstruction); CKD (kidney size); cysts; transplant evaluation | No radiation, no contrast; bedside availability; Doppler for RAS screening | Operator-dependent; limited by body habitus; cannot differentiate medical renal disease etiologies |
| CT Abdomen/Pelvis | Nephrolithiasis (non-contrast); renal masses (with contrast); renal artery evaluation (CTA) | High sensitivity for stones and masses; fast acquisition | Radiation; iodinated contrast risk in CKD; cannot characterize some cystic lesions |
| MRI / MRA | Renal artery stenosis; complex cystic lesions (Bosniak); RCC staging; ADPKD volume measurement | No radiation; excellent soft tissue contrast; gadolinium-enhanced MRA for RAS | Gadolinium contraindicated if eGFR <30 (risk of nephrogenic systemic fibrosis with group 1 agents); cost; availability |
| Nuclear Renal Scan | MAG3 (differential function, obstruction with furosemide washout); DMSA (cortical scarring); captopril renogram (RAS) | Functional assessment (split function); can diagnose obstruction vs dilation | Radiation; low spatial resolution; time-consuming |
| Renal Angiography | Gold standard for RAS; therapeutic (angioplasty/stenting); AV malformations; bleeding | Diagnostic and therapeutic; highest resolution | Invasive; contrast exposure; access site complications; atheroembolic risk |
37 Classification Systems (All)
KDIGO AKI Staging
| Stage | Creatinine Criteria | Urine Output Criteria |
|---|---|---|
| 1 | ≥0.3 mg/dL rise in 48 h OR 1.5–1.9× baseline | <0.5 mL/kg/h × 6–12 h |
| 2 | 2.0–2.9× baseline | <0.5 mL/kg/h × ≥12 h |
| 3 | ≥3.0× baseline OR Cr ≥4.0 OR RRT initiation | <0.3 mL/kg/h × ≥24 h OR anuria ≥12 h |
KDIGO CKD GFR & Albuminuria Classification
| GFR Category | GFR Range | Albuminuria A1 (<30) | Albuminuria A2 (30–300) | Albuminuria A3 (>300) |
|---|---|---|---|---|
| G1 (≥90) | Normal/high | Low risk | Moderate risk | High risk |
| G2 (60–89) | Mildly decreased | Low risk | Moderate risk | High risk |
| G3a (45–59) | Mild-mod decreased | Moderate risk | High risk | Very high risk |
| G3b (30–44) | Mod-sev decreased | High risk | Very high risk | Very high risk |
| G4 (15–29) | Severely decreased | Very high risk | Very high risk | Very high risk |
| G5 (<15) | Kidney failure | Very high risk | Very high risk | Very high risk |
ISN/RPS Lupus Nephritis Classification
| Class | Name | Key Features |
|---|---|---|
| I | Minimal mesangial | Normal LM; mesangial deposits on IF/EM |
| II | Mesangial proliferative | Mesangial hypercellularity |
| III | Focal proliferative | <50% glomeruli; endocapillary ± extracapillary |
| IV | Diffuse proliferative | ≥50% glomeruli; most severe; wire loops |
| V | Membranous | Subepithelial deposits; nephrotic picture |
| VI | Advanced sclerosing | ≥90% sclerosed glomeruli |
Oxford Classification for IgA Nephropathy (MEST-C)
| Score | Feature | Definition | Prognostic Significance |
|---|---|---|---|
| M0/M1 | Mesangial hypercellularity | ≤50% / >50% of glomeruli | M1 predicts worse outcome |
| E0/E1 | Endocapillary hypercellularity | Absent / Present | E1 may respond to immunosuppression |
| S0/S1 | Segmental sclerosis | Absent / Present | S1 predicts worse outcome |
| T0/T1/T2 | Tubular atrophy/interstitial fibrosis | ≤25% / 26–50% / >50% | Strongest predictor of progression |
| C0/C1/C2 | Crescents | Absent / <25% / ≥25% of glomeruli | C2 predicts rapid progression |
38 Medications Master Table
Immunosuppressants
| Drug | Mechanism | Dose | Key Toxicity | Monitoring |
|---|---|---|---|---|
| Cyclophosphamide | Alkylating agent; crosslinks DNA | IV: 0.5–1 g/m² monthly; Euro-Lupus: 500 mg q2wk × 6 | Hemorrhagic cystitis (give MESNA), leukopenia, infections, gonadal toxicity, bladder cancer | CBC q1–2 weeks; UA for hematuria |
| Mycophenolate mofetil | IMPDH inhibitor (purine synthesis) | 500–1500 mg BID | GI (diarrhea), leukopenia, teratogenic | CBC; MPA levels if available |
| Rituximab | Anti-CD20 (B-cell depletion) | 375 mg/m² weekly × 4 or 1 g × 2 (days 1, 15) | Infusion reactions, infections, PML (rare), HBV reactivation | CD19/CD20 counts; HBV screen before use |
| Tacrolimus | Calcineurin inhibitor | 0.05–0.1 mg/kg BID | Nephrotoxicity, NODAT, tremor, hyperkalemia, hypomagnesemia | Trough level (goal varies by indication) |
| Cyclosporine | Calcineurin inhibitor | 3–5 mg/kg/day divided BID | Nephrotoxicity, hypertension, hirsutism, gingival hyperplasia | Trough or C2 level |
Phosphate Binders
| Drug | Type | Dose | Advantages | Disadvantages |
|---|---|---|---|---|
| Sevelamer carbonate | Non-calcium, non-metal polymer | 800–1600 mg TID with meals | No calcium loading; may lower LDL; no aluminum toxicity | GI (bloating, constipation); large pill burden; cost |
| Lanthanum carbonate | Non-calcium metal binder | 500–1000 mg TID with meals | Effective; chewable | GI symptoms; long-term lanthanum tissue deposition (unclear significance) |
| Calcium acetate | Calcium-based | 667 mg (1–2 tabs) TID with meals | Inexpensive; effective | Hypercalcemia; vascular calcification risk; avoid if Ca >10.2 or PTH suppressed |
| Sucroferric oxyhydroxide | Iron-based | 500 mg TID with meals (chewable) | Low pill burden; provides some iron | Dark stools; GI side effects |
Diuretics in Nephrology
| Class | Drug | Site of Action | Typical Dose | Key Notes |
|---|---|---|---|---|
| Loop | Furosemide | TAL (NKCC2) | 20–400 mg IV/PO; ceiling effect in CKD | Most potent diuretic; ototoxic at high doses; hypoK, hypoMg, hypoCa |
| Loop | Bumetanide | TAL (NKCC2) | 0.5–10 mg; 1 mg bumetanide ≈ 40 mg furosemide | Better oral bioavailability than furosemide (80% vs 50%) |
| Loop | Torsemide | TAL (NKCC2) | 10–200 mg PO; 20 mg ≈ 40 mg furosemide | Longest acting loop; best oral absorption; may reduce HF readmissions |
| Thiazide | Chlorthalidone / HCTZ | DCT (NCC) | 12.5–50 mg daily | Ineffective if eGFR <30 (except metolazone); hypoK, hypoNa, hyperCa, hyperuricemia |
| Thiazide-like | Metolazone | DCT + proximal tubule | 2.5–10 mg (give 30 min before loop) | Sequential nephron blockade with loop; effective in advanced CKD; potent — monitor closely |
| K-sparing | Spironolactone / Eplerenone | Collecting duct (MR antagonist) | 25–100 mg daily | Hyperkalemia risk; gynecomastia (spironolactone); eplerenone more selective |
| K-sparing | Amiloride | Collecting duct (ENaC blocker) | 5–20 mg daily | Used for lithium-induced NDI; Liddle syndrome; hyperkalemia risk |
| Osmotic | Mannitol | Proximal tubule / loop | 0.25–1 g/kg IV | Used in cerebral edema; dialysis disequilibrium; check osmol gap |
| CA inhibitor | Acetazolamide | Proximal tubule | 250–500 mg BID | Metabolic acidosis (desired in alkalosis treatment); altitude sickness; glaucoma |
39 Abbreviations Master List
General Nephrology
Tubular & Electrolyte
Glomerular Disease
Dialysis & Transplant
Medications
40 Risk Scores & Guidelines
Kidney Failure Risk Equation (KFRE)
Predicts 2- and 5-year probability of kidney failure requiring dialysis or transplant, using four variables: age, sex, eGFR, and UACR. Validated in >30 countries and >700,000 patients. Used to guide referral timing and dialysis planning. An 8-variable version adds serum calcium, phosphorus, bicarbonate, and albumin for improved accuracy in CKD G4–5.
| KFRE 5-Year Risk | Clinical Action |
|---|---|
| <3% | Continue primary care management; annual nephrology review if CKD G3b+ |
| 3–5% | Nephrology referral; education about KRT modalities |
| 5–15% | Active KRT preparation; vascular access planning; transplant evaluation |
| >15% | Imminent dialysis planning; access creation; consider pre-emptive transplant listing |
UACR Thresholds & Management
| UACR (mg/g) | Category | Management Implications |
|---|---|---|
| <30 | A1 (normal) | Standard cardiovascular risk management |
| 30–300 | A2 (moderately increased) | ACEi/ARB indicated; add SGLT2i if DKD; screen annually |
| >300 | A3 (severely increased) | Aggressive RAAS blockade; SGLT2i; finerenone; nephrology referral; monitor q3–6 months |
Nephrology Referral Criteria (KDIGO)
| Indication | Details |
|---|---|
| eGFR <30 mL/min (CKD G4–5) | Mandatory referral for KRT planning |
| Persistent UACR >300 mg/g | Evaluation for underlying glomerular disease |
| Rapid GFR decline | >5 mL/min/year sustained decline |
| AKI of uncertain etiology | Especially with active sediment or systemic features |
| Resistant hypertension | Uncontrolled on ≥3 agents including a diuretic |
| Persistent hematuria with proteinuria | After urologic evaluation excludes structural causes |
| Hereditary kidney disease | ADPKD, Alport syndrome, Fabry disease |
| Recurrent nephrolithiasis | Metabolic evaluation and prevention |
Key Clinical Trial Summary
| Trial | Drug / Intervention | Population | Key Finding |
|---|---|---|---|
| CREDENCE (2019) | Canagliflozin | T2DM + CKD (eGFR 30–90, UACR >300) | 30% reduction in renal composite; stopped early for efficacy |
| DAPA-CKD (2020) | Dapagliflozin | CKD (eGFR 25–75, UACR 200–5000); 33% non-diabetic | 39% reduction in renal composite; benefit in non-diabetic CKD |
| EMPA-KIDNEY (2022) | Empagliflozin | CKD (eGFR 20–45, or 45–90 with UACR ≥200) | 28% reduction in progression; broadest CKD population studied |
| FIDELIO-DKD (2020) | Finerenone | T2DM + CKD (UACR 30–5000, on ACEi/ARB) | 18% reduction in kidney composite |
| FIGARO-DKD (2021) | Finerenone | T2DM + CKD (broader, earlier CKD) | 13% reduction in CV composite |
| FLOW (2024) | Semaglutide | T2DM + CKD | 24% reduction in kidney events; stopped early |
| RAVE (2010) | Rituximab vs cyclophosphamide | ANCA vasculitis | Non-inferior for remission induction; superior for relapsing disease |
| PEXIVAS (2020) | Plasma exchange in AAV | ANCA vasculitis with severe renal disease | No benefit for death or ESRD composite |
| SPRINT (2015) | Intensive BP control | High CV risk (no diabetes) | SBP <120 vs <140: 25% reduction in CV events; eGFR decline faster but less clinical kidney disease |
| PRESERVE (2018) | NAC + sodium bicarb for CIN prevention | CKD undergoing angiography | No benefit of NAC or IV sodium bicarbonate over IV saline |
References
Figures
- Figure 1 — Kidney Internal Anatomy. Source: StatPearls, NCBI Bookshelf.
- Figure 2 — The Nephron. Source: StatPearls, NCBI Bookshelf.
- Figure 3 — Kidney Cross-Section with Functional Labels. Source: NIDDK/NIH.
- Figure 4 — Renal Blood Supply. Source: StatPearls, NCBI Bookshelf.
- Figure 5 — AKI Pathology. Source: Okubo K et al. Clin J Am Soc Nephrol. 2021. PMC7886540.
- Figure 6 — ATN Histology. Source: Okubo K et al. Clin J Am Soc Nephrol. 2021. PMC7886540.
- Figure 7 — Early Diabetic Nephropathy. Source: Tervaert TW et al. PMC2746982.
- Figures 8–9 — Kimmelstiel-Wilson Nodules / Mesangial Expansion. Source: Tervaert TW et al. PMC2746982.
- Figure 10 — Glomerular Pathology in Nephrotic Syndrome. Source: Tervaert TW et al. PMC2746982.
- Figure 11 — Lupus Nephritis Biopsy. Source: Shimizu M et al. Case Rep Nephrol Dial. 2018. PMC5903134.
- Figures 12–13 — ANCA-Associated GN. Source: Lionaki S et al. Front Med. 2022. PMC9205443.
- Figures 14–15 — TMA Blood Smear / Algorithm. Source: Arnold DM et al. CMAJ. 2017. PMC5266569.
- Figures 16–18 — ADPKD Imaging (Ultrasound, CT, MRI). Source: Bae KT & Bhatt S. PMC3143364; Chebib FT et al. PMC6592088.
- Figure 19 — FMD String of Beads. Source: Defined N et al. J Med Case Rep. 2007. PMC1973079.
- Figure 20 — Hyponatremia Diagnostic Algorithm. Source: Hoorn EJ & Zietse R. J Am Soc Nephrol. 2017. PMC5407738.
- Figures 21–22 — ECG in Hyperkalemia. Source: Medford-Davis L & Rafique Z. Perm J. 2013. PMC3627796.
- Figure 23 — Anion Gap Gamblegram. Source: Rastegar A. Kidney Dis. 2017. PMC5757610.
- Figure 24 — Renal Ultrasound. Source: Chebib FT et al. PMC6592088.
- Figures 25–26 — PD Catheter Types / Site Selection. Source: Saran KA et al. Semin Nephrol. 2015. PMC4421142.
- Figures 27–30 — Transplant Rejection Histology. Source: Hara S. Kidney Res Clin Pract. 2020. PMC7105630.
Key Trials & Guidelines
- KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int Suppl. 2013;3(1):1–150.
- KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100(4S):S1–S276.
- KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of CKD. Kidney Int. 2024;105(4S):S117–S314.
- Perkovic V et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy (CREDENCE). N Engl J Med. 2019;380(24):2295–2306.
- Heerspink HJL et al. Dapagliflozin in Patients with Chronic Kidney Disease (DAPA-CKD). N Engl J Med. 2020;383(15):1436–1446.
- The EMPA-KIDNEY Collaborative Group. Empagliflozin in Patients with Chronic Kidney Disease (EMPA-KIDNEY). N Engl J Med. 2023;388(2):117–127.
- Bakris GL et al. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes (FIDELIO-DKD). N Engl J Med. 2020;383(23):2219–2229.
- Pitt B et al. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes (FIGARO-DKD). N Engl J Med. 2021;385(24):2252–2263.
- Perkovic V et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes (FLOW). N Engl J Med. 2024;391(2):109–121.
- Stone JH et al. Rituximab versus Cyclophosphamide for ANCA-Associated Vasculitis (RAVE). N Engl J Med. 2010;363(3):221–232.
- Walsh M et al. Plasma Exchange and Glucocorticoids in Severe ANCA-Associated Vasculitis (PEXIVAS). N Engl J Med. 2020;382(7):622–631.
- Weisbord SD et al. Outcomes after Angiography with Sodium Bicarbonate and Acetylcysteine (PRESERVE). N Engl J Med. 2018;378(7):603–614.
- SPRINT Research Group. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. N Engl J Med. 2015;373(22):2103–2116.
- Torres VE et al. Tolvaptan in Patients with Autosomal Dominant Polycystic Kidney Disease (TEMPO 3:4). N Engl J Med. 2012;367(25):2407–2418.
Textbooks & Core References
- Brenner BM, ed. Brenner & Rector's The Kidney. 11th ed. Elsevier; 2019.
- Johnson RJ, Feehally J, Floege J, eds. Comprehensive Clinical Nephrology. 7th ed. Elsevier; 2024.
- Rose BD, Post TW. Clinical Physiology of Acid-Base and Electrolyte Disorders. 5th ed. McGraw-Hill; 2001.
- Daugirdas JT, Blake PG, Ing TS, eds. Handbook of Dialysis. 5th ed. Wolters Kluwer; 2015.
- National Kidney Foundation. KDOQI Clinical Practice Guidelines. Available at: kidney.org.