Palliative Care / Hospice
Every symptom management strategy, goals-of-care framework, opioid conversion, hospice eligibility criterion, prognostication tool, ethical consideration, and end-of-life protocol in one place.
01 Philosophy & Scope of Palliative Care
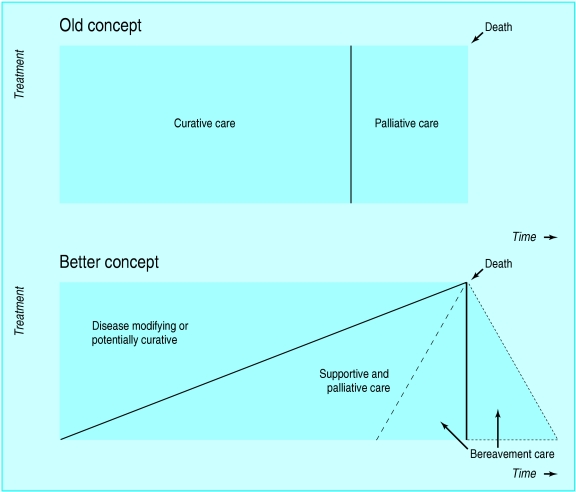
Palliative care is specialized medical care focused on providing relief from the symptoms, pain, and stress of serious illness, regardless of diagnosis or stage. The World Health Organization (WHO) defines it as “an approach that improves the quality of life of patients and their families facing the problems associated with life-threatening illness, through the prevention and relief of suffering by means of early identification and impeccable assessment and treatment of pain and other problems — physical, psychosocial, and spiritual.” Critically, palliative care is not limited to end-of-life care; it can and should be delivered concurrently with curative or disease-modifying treatments from the point of diagnosis of a serious illness.
Palliative Care vs Hospice vs Curative Care
| Feature | Curative / Disease-Directed | Palliative Care | Hospice |
|---|---|---|---|
| Goal | Cure or remission of disease | Relief of suffering; improve QOL | Comfort-focused care at end of life |
| Timing | From diagnosis | From diagnosis of serious illness (concurrent with curative Rx) | Prognosis ≤6 months if disease runs its usual course |
| Disease-modifying Rx | Primary focus | May continue alongside | Generally discontinued (some exceptions) |
| Setting | Hospital, clinic | Any setting (hospital, outpatient, home) | Home, nursing facility, hospice house, or hospital |
| Payment | Standard insurance | Standard insurance (inpatient consult, outpatient visit) | Medicare Hospice Benefit (per diem); Medicaid; private insurance |
| Team | Disease-specific specialists | Palliative MDT (physician, APN/PA, SW, chaplain) | IDT (physician, nurse, SW, chaplain, aide, volunteer, bereavement) |
Historical Foundations
The modern hospice and palliative care movement was founded by Dame Cicely Saunders (1918–2005), who established St. Christopher’s Hospice in London in 1967 — the first modern hospice. She pioneered the concept of total pain (see Section 02) and the use of regularly scheduled opioids rather than “as-needed” dosing. In the United States, the hospice movement was advanced by Elisabeth Kübler-Ross, whose 1969 book On Death and Dying introduced the five stages of grief (denial, anger, bargaining, depression, acceptance). The Medicare Hospice Benefit was established in 1982. Palliative medicine became a recognized ABMS subspecialty in 2006.
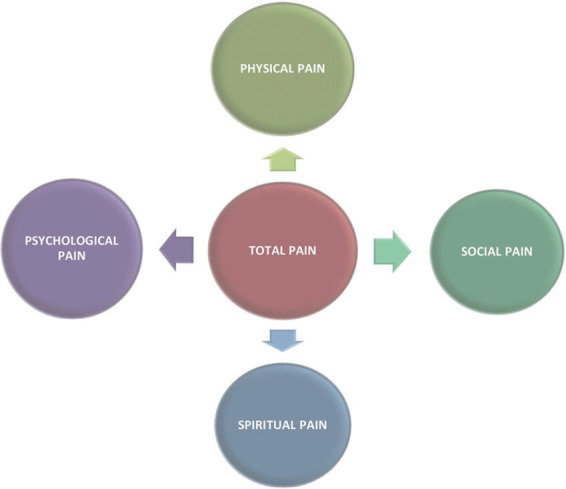
02 Total Pain & Suffering Framework
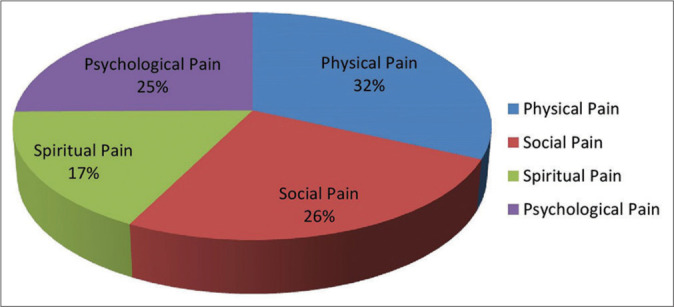
Cicely Saunders introduced the concept of total pain to describe the multidimensional nature of suffering in serious illness. Total pain encompasses four interrelated domains, all of which must be assessed and addressed for effective palliation.
The Four Domains of Total Pain
| Domain | Components | Assessment | Interventions |
|---|---|---|---|
| Physical | Pain, dyspnea, nausea, fatigue, insomnia, constipation, anorexia, wounds | Pain scales (NRS, FLACC, PAINAD), ESAS, symptom checklists | Pharmacologic (opioids, adjuvants), procedural, rehabilitation |
| Psychological | Anxiety, depression, fear of death, loss of control, anger, cognitive decline, delirium | PHQ-9, GAD-7, delirium screening (CAM), psychiatric evaluation | Counseling, psychotherapy (CBT, meaning-centered therapy), pharmacotherapy (SSRIs, anxiolytics) |
| Social | Family distress, caregiver burden, financial strain, loss of roles/identity, isolation, practical needs | Social work assessment, caregiver burden scales (Zarit), family meetings | Social work intervention, community resources, support groups, family meetings |
| Spiritual/Existential | Meaning-making, purpose, hope, guilt, forgiveness, afterlife concerns, religious/cultural needs | FICA tool, HOPE tool, chaplain assessment | Chaplaincy, spiritual counseling, life review, legacy projects, rituals |
Spiritual Assessment Tools
The FICA Spiritual History Tool (Puchalski, 2000) provides a structured framework:
- F — Faith, Belief, Meaning: “Do you consider yourself spiritual or religious?”
- I — Importance and Influence: “How important is faith in your life? How does it influence your healthcare decisions?”
- C — Community: “Are you part of a spiritual or religious community?”
- A — Address in Care: “How would you like healthcare providers to address spiritual needs?”
03 Palliative Care Triggers & Consultation Criteria
Early identification of patients who would benefit from palliative care consultation improves outcomes. Many institutions use screening tools or “trigger criteria” to prompt automatic palliative care referral.
Common Palliative Care Trigger Criteria
- The “Surprise Question”: “Would I be surprised if this patient died within the next 12 months?” If the answer is “No,” consider palliative care referral
- ≥2 hospital admissions for same illness in past 6 months
- ICU admission with multi-organ failure, age >80, or pre-existing severe functional impairment
- Advanced or metastatic cancer diagnosis
- Diagnosis of NYHA Class III–IV heart failure, GOLD Stage III–IV COPD, ESRD with decision against dialysis, advanced dementia (FAST ≥7), ALS, or advanced cirrhosis (Child-Pugh C)
- Difficult-to-control symptoms (pain, dyspnea, nausea) despite standard management
- Patient or family request for comfort-focused care
- Conflict between patient/family and medical team regarding goals of care
- Prolonged ICU stay (>7 days) without clear trajectory of improvement
- Post-cardiac arrest with poor neurologic prognosis
Primary vs Specialist Palliative Care
| Primary Palliative Care (all clinicians) | Specialist Palliative Care (consult team) |
|---|---|
| Basic symptom management | Refractory symptoms requiring complex interventions |
| Basic goals-of-care discussions | Complex family dynamics or conflict |
| Code status conversations | Existential/spiritual suffering |
| Simple advance care planning | Assistance with complex medical decision-making |
| Recognizing when specialist referral is needed | Guidance on withdrawal of life-sustaining treatments |
04 Key Terminology & Abbreviations
| Term | Definition |
|---|---|
| Palliative care | Specialized care focused on relief of suffering in serious illness at any stage |
| Hospice | Philosophy and program of comfort care for patients with prognosis ≤6 months |
| Total pain | Multidimensional suffering: physical, psychological, social, spiritual (Saunders) |
| Goals of care (GOC) | Patient-centered objectives guiding treatment decisions |
| Advance directive (AD) | Legal document specifying healthcare wishes if patient loses capacity |
| DPOA-HC | Durable power of attorney for healthcare — designated surrogate decision-maker |
| POLST/MOLST | Physician/Medical Orders for Life-Sustaining Treatment — portable medical order |
| DNR/DNAR | Do not resuscitate / do not attempt resuscitation |
| AND | Allow natural death — alternative framing of DNR |
| DNI | Do not intubate |
| Comfort measures only (CMO) | Care focused exclusively on comfort; no curative or life-prolonging measures |
| Terminal extubation | Removal of mechanical ventilation in a dying patient transitioning to comfort care |
| Palliative sedation | Use of sedatives to reduce consciousness to relieve refractory suffering at end of life |
| VSED | Voluntarily stopping eating and drinking to hasten death |
| MAID | Medical assistance in dying (physician-assisted death) |
| Breakthrough pain | Transient flare of pain on top of a controlled baseline |
| Equianalgesic dose | Dose of one opioid equivalent in analgesic effect to a reference dose of another |
| Opioid rotation | Switching from one opioid to another due to side effects or tolerance |
| Death rattle | Noisy breathing from retained pharyngeal secretions in the dying patient |
| Terminal agitation | Restlessness and agitation in the final hours to days of life |
| Cachexia | Involuntary weight loss with muscle wasting driven by inflammatory mediators |
| PPS | Palliative Performance Scale — 11-level functional assessment tool |
| ESAS | Edmonton Symptom Assessment Scale — 9-symptom visual analog tool |
| FAST | Functional Assessment Staging Tool for dementia (Reisberg) |
| PPI | Palliative Prognostic Index |
| PaP | Palliative Prognostic Score |
| GIP | General inpatient care (hospice level of care) |
| IDT | Interdisciplinary team |
| LCD | Local coverage determination (Medicare hospice eligibility guidelines) |
05 Pain Assessment Tools
Effective pain management requires systematic, repeated assessment using validated tools appropriate to the patient population. Pain is subjective — the patient’s self-report is the gold standard when obtainable.
Pain Assessment Scales
| Scale | Population | Components | Scoring |
|---|---|---|---|
| Numeric Rating Scale (NRS) | Verbal adults | Patient rates pain 0–10 | 0 = no pain; 1–3 = mild; 4–6 = moderate; 7–10 = severe |
| Visual Analog Scale (VAS) | Verbal adults | 100 mm line, patient marks point | Distance from left end in mm |
| Wong-Baker FACES | Children ≥3 years, cognitively impaired adults | 6 faces from smiling to crying | 0, 2, 4, 6, 8, 10 |
| FLACC Scale | Infants & nonverbal children (2 mo–7 yr) | Face, Legs, Activity, Cry, Consolability | Each 0–2; total 0–10 |
| PAINAD | Nonverbal adults with advanced dementia | Breathing, Negative vocalization, Facial expression, Body language, Consolability | Each 0–2; total 0–10 |
| CPOT | Intubated/nonverbal ICU patients | Facial expression, Body movements, Muscle tension, Compliance with ventilator (or vocalization) | Each 0–2; total 0–8 |
PAINAD Scale — Full Enumeration
| Item | 0 | 1 | 2 |
|---|---|---|---|
| Breathing (independent of vocalization) | Normal | Occasional labored breathing; short period of hyperventilation | Noisy labored breathing; long period of hyperventilation; Cheyne-Stokes |
| Negative vocalization | None | Occasional moan/groan; low-level speech with negative quality | Repeated troubled calling out; loud moaning/groaning; crying |
| Facial expression | Smiling or inexpressive | Sad; frightened; frown | Facial grimacing |
| Body language | Relaxed | Tense; distressed pacing; fidgeting | Rigid; fists clenched; knees pulled up; pulling/pushing away; striking out |
| Consolability | No need to console | Distracted or reassured by voice or touch | Unable to console, distract, or reassure |
Comprehensive Pain Assessment: OPQRSTUV
- Onset — When did the pain start? Sudden or gradual?
- Provocative/Palliative — What makes it worse? What makes it better?
- Quality — What does it feel like? (sharp, dull, burning, aching, stabbing, electric)
- Region/Radiation — Where is the pain? Does it radiate?
- Severity — Rate 0–10 (NRS)
- Temporal — Constant vs intermittent? Duration? Pattern?
- Understanding — What does the patient believe is causing the pain?
- Values — What is the patient’s goal for pain control? Acceptable pain level?
Nociceptive vs Neuropathic Pain
| Feature | Nociceptive (Somatic) | Nociceptive (Visceral) | Neuropathic |
|---|---|---|---|
| Mechanism | Tissue damage activating nociceptors | Stretch, distension, ischemia of viscera | Nerve injury or dysfunction |
| Quality | Sharp, aching, well-localized | Crampy, colicky, pressure, poorly localized | Burning, shooting, electric, tingling, numbness |
| Examples | Bone metastases, surgical wound, arthritis | Bowel obstruction, liver capsule distension, pancreatic cancer | Chemotherapy-induced neuropathy, post-herpetic neuralgia, spinal cord compression |
| First-line Rx | NSAIDs, acetaminophen, opioids | Opioids, anticholinergics, corticosteroids | Gabapentinoids, SNRIs, TCAs; opioids as adjunct |
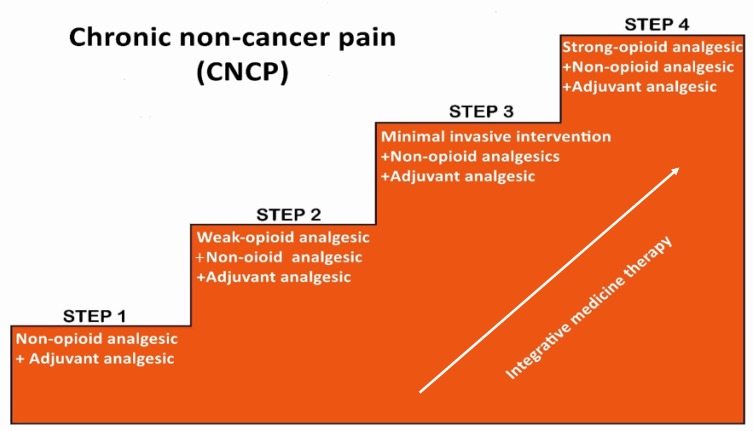
06 WHO Analgesic Ladder & Opioid Pharmacology
WHO Three-Step Analgesic Ladder
| Step | Pain Severity | Agents | Notes |
|---|---|---|---|
| Step 1 | Mild (NRS 1–3) | Non-opioid analgesics: acetaminophen (≤3 g/day; 2 g if hepatic impairment), NSAIDs (ibuprofen, naproxen, ketorolac) ± adjuvants | Ceiling effect for non-opioids; NSAIDs: GI, renal, CV risk |
| Step 2 | Moderate (NRS 4–6) | Weak opioids: tramadol, codeine, low-dose oxycodone or hydrocodone ± non-opioid ± adjuvants | Codeine requires CYP2D6 metabolism (poor metabolizers: ineffective; ultra-rapid: toxicity). Tramadol: seizure risk, serotonin syndrome |
| Step 3 | Severe (NRS 7–10) | Strong opioids: morphine, hydromorphone, oxycodone, fentanyl, methadone ± non-opioid ± adjuvants | No ceiling effect for pure mu-agonists; titrate to effect |
Opioid Pharmacology — Key Agents
| Opioid | Route | Onset | Duration | Key Features |
|---|---|---|---|---|
| Morphine | PO, IV, SC, PR, SL, IT | PO: 30 min; IV: 5–10 min | IR: 3–4 h; SR: 8–12 h | Gold standard; active metabolite M6G accumulates in renal failure → toxicity; histamine release |
| Hydromorphone | PO, IV, SC, PR | PO: 30 min; IV: 5 min | IR: 3–4 h | 5–7× more potent than morphine; preferred in renal impairment (no active metabolites); less histamine release |
| Oxycodone | PO, PR | PO: 15–30 min | IR: 3–4 h; CR: 12 h | Good oral bioavailability (60–87%); no parenteral form widely available in US |
| Fentanyl | IV, SC, TD (patch), TM, SL, buccal, IN | IV: 1–2 min; TD: 12–24 h | IV: 30–60 min; TD: 72 h | 75–100× more potent than morphine; lipophilic; no histamine release; not affected by renal failure; requires stable pain for transdermal |
| Methadone | PO, IV, SC, PR | PO: 30–60 min | 4–8 h (analgesic); t½ 15–60 h | NMDA-receptor antagonist (neuropathic pain benefit); long/variable t½ → accumulation risk; QTc prolongation; complex conversions; inexpensive |
| Codeine | PO | 30–60 min | 4–6 h | Prodrug (CYP2D6 → morphine); ceiling at ~60 mg/dose; avoid in ultra-rapid metabolizers and CYP2D6 poor metabolizers |
| Tramadol | PO | 60 min | 4–6 h | Weak mu-agonist + SNRI; seizure risk; serotonin syndrome; ceiling ~400 mg/day |
Opioid Side Effects & Management
| Side Effect | Incidence | Tolerance Develops? | Management |
|---|---|---|---|
| Constipation | >90% | No (never) | Prophylactic bowel regimen (stimulant + softener); methylnaltrexone for refractory OIC |
| Nausea/vomiting | 30–50% initially | Yes (3–5 days usually) | Ondansetron, haloperidol, metoclopramide, prochlorperazine |
| Sedation | Common initially | Yes (3–5 days) | Reduce dose if possible; methylphenidate for persistent sedation |
| Respiratory depression | Rare with proper titration | Yes (develops quickly) | Naloxone for emergency reversal; dose reduction; slower titration |
| Pruritus | 10–20% | Variable | Opioid rotation; antihistamines; ondansetron; nalbuphine |
| Myoclonus | Dose-related (high doses, renal failure) | No | Dose reduction; opioid rotation; benzodiazepines (clonazepam, lorazepam) |
| Urinary retention | 10–15% | Yes | Catheterization; opioid rotation |
| Hyperalgesia | Chronic high-dose use | No (paradoxical) | Dose reduction; opioid rotation (especially to methadone); NMDA antagonists (ketamine) |
Signs: Respiratory rate <8/min, pinpoint pupils, obtundation/unresponsiveness, cyanosis, hypotension.
Immediate management:
- Stimulate patient, maintain airway, provide bag-mask ventilation
- Naloxone (Narcan): In palliative care patients, use dilute, titrated doses to avoid precipitating pain crisis and withdrawal
- Dilute naloxone 0.4 mg in 10 mL NS → give 0.5–1 mL (0.02–0.04 mg) IV every 2–3 min until respiratory rate >8–10/min
- Goal: restore respiratory drive WITHOUT reversing analgesia
- Duration of naloxone (30–90 min) shorter than most opioids → observe for re-sedation; may need repeat dosing or infusion
In non-palliative emergency settings: Naloxone 0.4–2 mg IV/IM/IN; repeat every 2–3 min to max 10 mg. For IN: 4 mg intranasal spray.
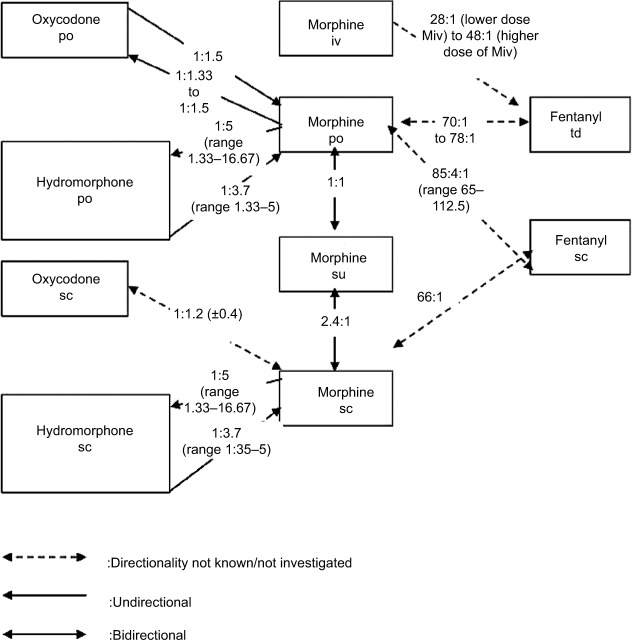
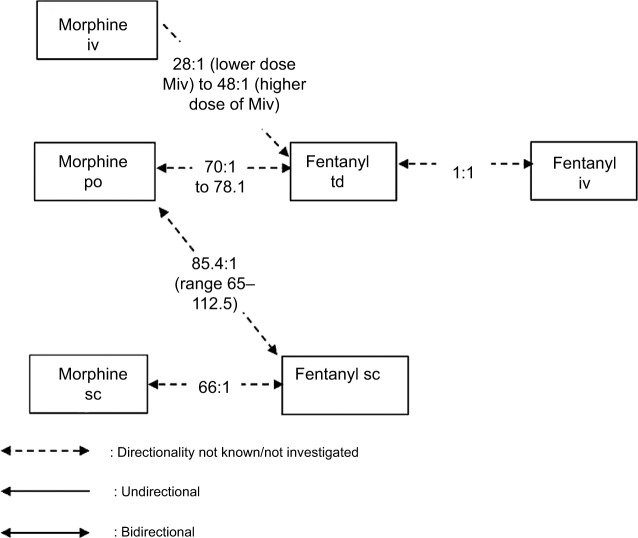
07 Opioid Rotation & Equianalgesic Dosing
Opioid rotation is the practice of switching from one opioid to another. Indications include: uncontrolled pain despite dose escalation, intolerable side effects (nausea, myoclonus, sedation, hallucinations), opioid-induced hyperalgesia, renal failure (avoid morphine), route change requirement, or cost/availability issues.
Equianalgesic Dose Table
| Opioid | PO Dose (mg) | IV/SC Dose (mg) | PO:IV Ratio | Relative Potency (to PO morphine) |
|---|---|---|---|---|
| Morphine | 30 | 10 | 3:1 | 1× (reference) |
| Hydromorphone (Dilaudid) | 6 | 1.5 | 4:1 | 5× |
| Oxycodone | 20 | N/A (US) | — | 1.5× |
| Hydrocodone | 30 | N/A | — | 1× |
| Codeine | 200 | N/A | — | 0.15× |
| Fentanyl (transdermal) | Fentanyl 25 mcg/h patch ≈ morphine 60–90 mg PO/24 h (use 2:1 or morphine mg PO/day ÷ 2–3 = fentanyl mcg/h) | |||
| Methadone | Variable ratio — see conversion table below | |||
Steps for Opioid Rotation
- Calculate total 24-hour dose of current opioid in oral morphine equivalents (OME)
- Using equianalgesic table, convert to new opioid dose
- Reduce the calculated dose by 25–50% for incomplete cross-tolerance (25% reduction if pain is well-controlled; 50% if rotating due to side effects or to methadone)
- Calculate new scheduled dose and breakthrough dose (10–15% of total 24-hour dose, given q1–2h PRN)
- Titrate over subsequent days based on number of breakthrough doses used
Methadone Conversion — Variable Ratio
Methadone conversion is not a fixed ratio. The equianalgesic ratio increases as the morphine dose increases, due to methadone’s long half-life and NMDA-receptor activity:
| Total Daily Oral Morphine Equivalent (mg) | Morphine:Methadone Ratio |
|---|---|
| <90 mg | 4:1 |
| 90–300 mg | 8:1 |
| 301–600 mg | 12:1 |
| 601–800 mg | 15:1 |
| 801–1000 mg | 20:1 |
| >1000 mg | 20:1 or higher (expert consultation recommended) |
- Methadone has a long, variable half-life (15–60 hours) — dose accumulation occurs over 5–7 days; do not increase dose more frequently than every 5–7 days
- Check baseline ECG and monitor QTc; hold if QTc >500 ms or increases >60 ms from baseline
- Many drug interactions (CYP3A4, CYP2B6 substrates) — check before prescribing
- Despite these challenges, methadone is an excellent palliative analgesic: effective for neuropathic pain, inexpensive, available in liquid form, and has no active metabolites
Breakthrough Pain Dosing
Breakthrough dose = 10–15% of the total 24-hour scheduled opioid dose, given as an immediate-release formulation every 1–2 hours PRN. Example: patient on morphine SR 60 mg q12h (120 mg/24h) → breakthrough = morphine IR 12–18 mg (round to 15 mg) q1–2h PRN. If patient uses ≥3 breakthrough doses per day consistently, incorporate breakthrough doses into the scheduled regimen by increasing the around-the-clock dose by the total daily breakthrough used.
08 Adjuvant Analgesics & Interventional Pain
Adjuvant analgesics are medications whose primary indication is not pain but which have analgesic properties for specific pain syndromes. They are essential in palliative care, particularly for neuropathic pain, bone pain, and pain refractory to opioids alone.
Adjuvant Analgesics by Class
| Class | Agents | Indication | Dosing | Key Side Effects |
|---|---|---|---|---|
| Anticonvulsants | Gabapentin, pregabalin | Neuropathic pain (burning, shooting, tingling) | Gabapentin: start 100–300 mg TID, titrate to 900–3600 mg/day; Pregabalin: start 25–75 mg BID, titrate to 150–600 mg/day | Sedation, dizziness, peripheral edema; renal dose adjustment |
| SNRIs | Duloxetine, venlafaxine | Neuropathic pain, musculoskeletal pain, CIPN | Duloxetine: 30 mg daily × 1 week then 60 mg daily; Venlafaxine: 37.5–225 mg daily | Nausea, dizziness, serotonin syndrome (with tramadol/SSRIs) |
| TCAs | Amitriptyline, nortriptyline, desipramine | Neuropathic pain, insomnia | Start 10–25 mg at bedtime, titrate to 75–150 mg | Anticholinergic effects, sedation, cardiac toxicity (avoid in elderly — Beers criteria); nortriptyline/desipramine better tolerated |
| Corticosteroids | Dexamethasone, prednisone | Bone pain, brain metastases, spinal cord compression, visceral pain (hepatic capsule stretch), bowel obstruction, nerve compression, appetite stimulation | Dexamethasone 4–16 mg/day (up to 96 mg for spinal cord compression); taper when possible | Hyperglycemia, insomnia, delirium, proximal myopathy, oral candidiasis, GI bleed |
| NMDA antagonists | Ketamine | Refractory pain, opioid-induced hyperalgesia, neuropathic pain | “Burst” ketamine: 0.1–0.5 mg/kg/h IV infusion × 24–72 h; oral: 10–25 mg TID–QID; intranasal | Dysphoria, hallucinations (co-administer midazolam or haloperidol), salivation, hypertension |
| Local anesthetics | Lidocaine (IV, topical patch) | Neuropathic pain, post-herpetic neuralgia, mucositis | Lidocaine 5% patch: up to 3 patches, 12 h on/12 h off; IV: 1–2 mg/kg bolus then 1–2 mg/kg/h | Local skin irritation (patch); arrhythmia, seizure (IV — monitor) |
| Bisphosphonates/Denosumab | Zoledronic acid, pamidronate, denosumab | Bone metastases pain, hypercalcemia of malignancy | Zoledronic acid 4 mg IV q3–4 weeks; denosumab 120 mg SC q4 weeks | ONJ (osteonecrosis of the jaw), nephrotoxicity (zoledronate), hypocalcemia |
| Muscle relaxants | Baclofen, tizanidine, cyclobenzaprine | Muscle spasm, spasticity | Baclofen 5–20 mg TID; tizanidine 2–8 mg TID | Sedation, weakness; baclofen withdrawal can be life-threatening |
Interventional Pain Management in Palliative Care
| Intervention | Indication | Mechanism | Key Considerations |
|---|---|---|---|
| Celiac plexus block/neurolysis | Pancreatic cancer pain, upper abdominal visceral pain | Chemical destruction (alcohol or phenol) of celiac plexus | 70–90% pain relief; complications: hypotension (sympathectomy), diarrhea |
| Superior hypogastric plexus block | Pelvic cancer pain | Chemical or thermal destruction of sympathetic plexus | Effective for cervical, rectal, bladder cancer pain |
| Intrathecal drug delivery (IDDS) | Refractory cancer pain despite high-dose systemic opioids | Direct delivery of opioids ± local anesthetics ± ziconotide to spinal cord | Implanted pump; 1/300 systemic dose; infection, pump malfunction |
| Epidural catheter | Localized pain in patients with limited prognosis | Opioids ± local anesthetics delivered to epidural space | Temporary; external catheter; infection risk with prolonged use |
| Vertebroplasty/kyphoplasty | Painful vertebral compression fractures (pathologic/osteoporotic) | Injection of PMMA bone cement into fractured vertebral body | Rapid pain relief; risk of cement leak, pulmonary embolism |
| Nerve blocks (peripheral) | Localized pain syndromes (intercostal, brachial plexus, paravertebral) | Local anesthetic ± steroid injection at specific nerve | Temporary relief; repeated injections may be needed |
| Palliative radiation | Bone metastases, brain metastases, tumor-related pain/compression | Tumor cytoreduction → reduced mass effect | Single fraction (8 Gy) effective for bone pain; response in 60–70% within 2–4 weeks |
09 Dyspnea
Dyspnea is the subjective sensation of breathing discomfort. It is one of the most distressing symptoms in advanced illness, affecting up to 70% of patients with advanced cancer and >90% with end-stage COPD or CHF. Like pain, dyspnea is subjective — the patient’s report is the gold standard; oxygen saturation does not correlate well with dyspnea severity.
Etiology in Palliative Patients
- Pulmonary: Pleural effusion, lung parenchymal disease (primary or metastatic), pneumonia, PE, COPD/asthma exacerbation, radiation pneumonitis, lymphangitic carcinomatosis, SVC syndrome
- Cardiac: Heart failure (pulmonary edema), pericardial effusion/tamponade
- Other: Anemia, ascites (diaphragmatic splinting), anxiety/panic, neuromuscular weakness (ALS), chest wall disease
Non-Pharmacologic Management
- Fan therapy: Directed air flow to the face stimulates V2/V3 trigeminal nerve branches — evidence-based, simple, and effective
- Upright positioning, energy conservation, relaxation/breathing techniques
- Oxygen: Only beneficial if hypoxemic (SpO2 <90%); in non-hypoxemic dyspnea, fan therapy is equivalent to supplemental O2
Pharmacologic Management
| Agent | Dose | Mechanism | Notes |
|---|---|---|---|
| Opioids (first-line) | Opioid-naïve: morphine 2.5–5 mg PO q4h or 1–2 mg IV/SC q2–4h; On opioids: increase current dose by 25% | Reduce ventilatory drive, decrease perception of breathlessness, reduce anxiety | Strong evidence for opioids in dyspnea; dose for dyspnea typically lower than for pain; nebulized morphine NOT supported by evidence |
| Benzodiazepines | Lorazepam 0.5–1 mg PO/SL/IV q6–8h; midazolam 2.5–5 mg SC q1h PRN | Anxiolysis; reduce air hunger perception | Second-line; most useful when anxiety is a major component; NOT first-line monotherapy for dyspnea |
| Corticosteroids | Dexamethasone 4–8 mg daily | Reduce inflammation, edema around tumors, lymphangitis | Useful for SVC syndrome, lymphangitic carcinomatosis, radiation pneumonitis, airway obstruction |
| Bronchodilators | Albuterol, ipratropium nebulizer | Bronchospasm relief | Primarily for bronchospastic component (COPD, asthma) |
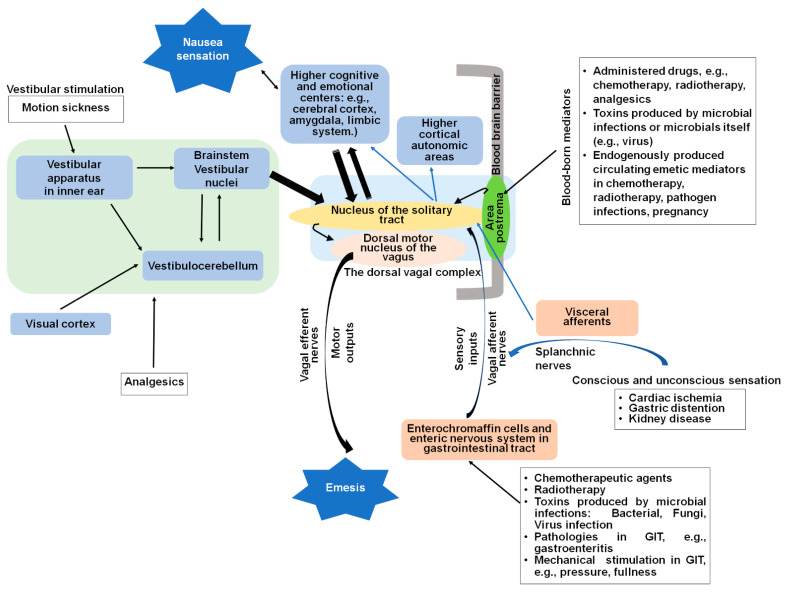
10 Nausea & Vomiting
Nausea and vomiting affect 40–70% of patients with advanced cancer and are common in many terminal illnesses. Effective management requires identifying the underlying mechanism to select targeted antiemetics. There are four main emetic pathways.
Etiology-Based Approach to Nausea/Vomiting
| Mechanism/Pathway | Receptors | Common Causes | Targeted Antiemetics |
|---|---|---|---|
| Chemoreceptor trigger zone (CTZ) | D2, 5-HT3, NK1 | Medications (opioids, chemo, digoxin), uremia, hypercalcemia, toxins | Haloperidol (D2), ondansetron (5-HT3), prochlorperazine (D2) |
| GI/peripheral | 5-HT3, 5-HT4, D2 | Gastroparesis, bowel obstruction, constipation, peritoneal disease | Metoclopramide (D2 + 5-HT4 prokinetic — AVOID in complete obstruction), ondansetron, erythromycin (motilin agonist) |
| Vestibular | H1, ACh (muscarinic) | Motion sickness, opioids (early), brain metastases (posterior fossa) | Meclizine (H1), scopolamine (ACh), promethazine (H1) |
| Central / cortical | Multiple (GABA, H1, ACh) | Raised ICP, anxiety, anticipatory nausea, meningeal disease | Dexamethasone, lorazepam, olanzapine, nabilone/dronabinol |
Key Antiemetics in Palliative Care
| Drug | Class | Dose | Notes |
|---|---|---|---|
| Haloperidol | D2 antagonist (butyrophenone) | 0.5–2 mg PO/SC/IV q6–8h | Excellent for opioid-induced and metabolic nausea; low sedation at low doses; QTc prolongation |
| Metoclopramide | D2 antagonist + 5-HT4 agonist | 10 mg PO/IV q6h (30 min before meals) | Prokinetic: best for gastroparesis; CONTRAINDICATED in complete bowel obstruction; EPS risk |
| Ondansetron | 5-HT3 antagonist | 4–8 mg PO/IV/ODT q6–8h | Chemotherapy-induced N/V; constipating; QTc (IV high doses) |
| Olanzapine | Multimodal (D2, 5-HT3, H1, ACh) | 2.5–5 mg PO daily–BID | Broad spectrum; excellent for refractory N/V and CINV; sedation, metabolic effects |
| Dexamethasone | Corticosteroid | 4–8 mg PO/IV daily | Raised ICP, bowel obstruction, CINV adjunct; appetite stimulant |
| Scopolamine | Anticholinergic | 1.5 mg transdermal patch q72h | Vestibular nausea; secretion reduction; delirium risk in elderly |
| Dronabinol/Nabilone | Cannabinoid | 2.5–5 mg PO BID–TID | Refractory N/V; appetite stimulation; sedation, dysphoria |
11 Constipation & Bowel Management
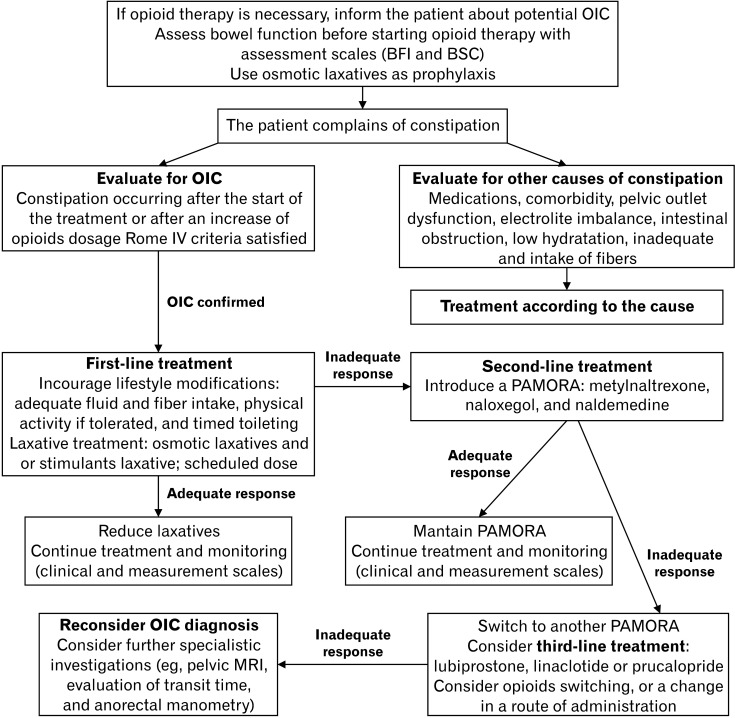
Constipation affects >90% of patients on chronic opioids and is the most common opioid side effect that does NOT develop tolerance. Prevention is essential: every patient starting an opioid should simultaneously start a bowel regimen.
Opioid-Induced Constipation (OIC) Pathophysiology
Opioids activate mu-receptors in the myenteric and submucosal plexus of the GI tract, causing: (1) decreased peristalsis (reduced propulsive contractions), (2) increased tone of non-propulsive circular muscle (spasm), (3) decreased secretions (fluid absorption increases), (4) increased anal sphincter tone. The net effect is hard, infrequent stools.
Laxative Ladder for OIC
| Step | Agents | Mechanism | Dose |
|---|---|---|---|
| Step 1 — Prevention (start with opioid) | Senna + docusate (Senokot-S) | Stimulant + stool softener | Senna 8.6–17.2 mg BID; docusate 100 mg BID; titrate senna to effect (up to 4 tabs BID) |
| Step 2 — Add osmotic | Polyethylene glycol (MiraLAX), lactulose | Osmotic laxative | PEG 17 g daily–BID; lactulose 15–30 mL BID–QID |
| Step 3 — Add or switch | Bisacodyl (suppository or oral), magnesium citrate | Stimulant, osmotic | Bisacodyl 10 mg PR or 5–15 mg PO; mag citrate 240 mL PO |
| Step 4 — Rectal measures | Glycerin suppository, mineral oil enema, warm water/soap suds enema, manual disimpaction | Local stimulation, lubrication | PRN; manual disimpaction for fecal impaction |
| Step 5 — Peripheral opioid antagonists | Methylnaltrexone (Relistor), naloxegol (Movantik), naldemedine (Symproic) | Peripheral mu-receptor antagonist (does not cross BBB — does NOT reverse analgesia) | Methylnaltrexone 8–12 mg SC every other day; naloxegol 25 mg PO daily |
Bowel Obstruction in Palliative Care
See Section 28 (Malignant Bowel Obstruction) for detailed management. Key principle: in inoperable malignant bowel obstruction, pharmacologic management with octreotide, dexamethasone, and antiemetics can achieve symptom control without surgery or NG tube in many patients.
12 Delirium & Terminal Agitation
Delirium is an acute, fluctuating disturbance of consciousness with inattention and cognitive dysfunction. It occurs in up to 88% of patients in the last weeks of life and is the most common neuropsychiatric complication in terminally ill patients. It is deeply distressing to patients, families, and clinicians.
Delirium Classification
| Subtype | Features | Frequency | Prognosis |
|---|---|---|---|
| Hyperactive | Agitation, restlessness, hallucinations, pulling at lines/tubes, combativeness | ~25% | More easily recognized; distressing to families |
| Hypoactive | Lethargy, somnolence, reduced awareness, psychomotor slowing, withdrawn | ~50% | Often unrecognized or mistaken for depression/sedation; associated with worse prognosis |
| Mixed | Alternating hyperactive and hypoactive features | ~25% | Variable |
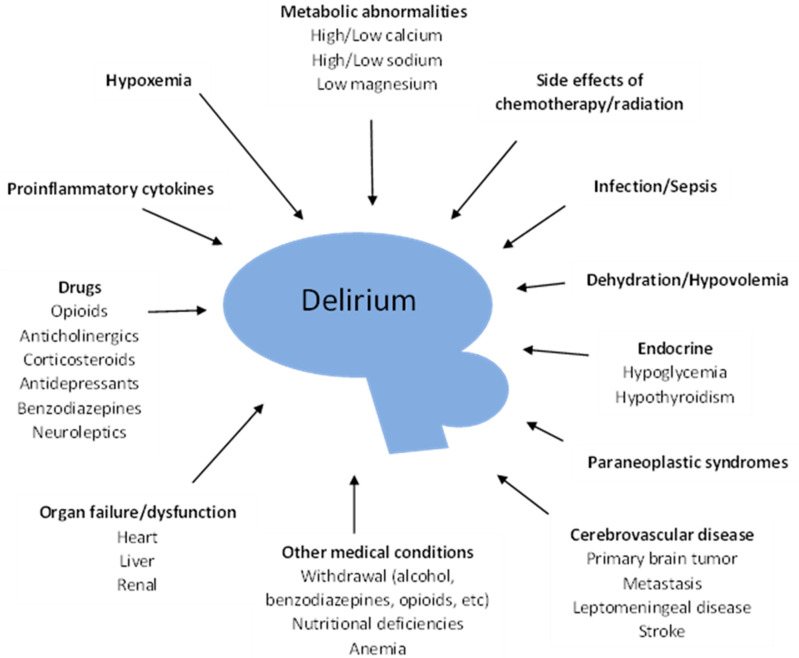
Reversible Causes (Mnemonic: DELIRIUM)
- D — Drugs (opioids, benzodiazepines, anticholinergics, corticosteroids, polypharmacy)
- E — Electrolyte abnormalities (hypercalcemia, hyponatremia, hypo/hyperglycemia)
- L — Liver/renal failure (uremia, hepatic encephalopathy)
- I — Infection (UTI, pneumonia, sepsis)
- R — Retention (urinary retention, fecal impaction)
- I — Intracranial pathology (brain metastases, leptomeningeal disease, stroke)
- U — Uncontrolled pain (inadequately treated pain can cause delirium)
- M — Metabolic (hypoxia, dehydration)
Management
Reversible delirium (~50% of episodes): Identify and correct underlying cause(s). Opioid rotation if opioid metabolite accumulation suspected. Adjust medications. Hydrate if dehydration likely contributes. Treat infection if consistent with goals of care.
Irreversible / terminal delirium: In the last days of life, delirium is often irreversible (“terminal agitation”) due to progressive multi-organ failure. Focus shifts to comfort and safety.
Pharmacologic Management
| Agent | Dose | Route | Notes |
|---|---|---|---|
| Haloperidol | 0.5–2 mg q4–6h; may increase to 5 mg q4h | PO, SC, IV | First-line antipsychotic for delirium in palliative care; EPS risk; QTc prolongation |
| Chlorpromazine | 12.5–50 mg q6–8h | PO, PR, IV | More sedating than haloperidol — preferred when sedation is desired; hypotension risk |
| Olanzapine | 2.5–5 mg PO/SL daily–BID | PO, SL (ODT wafer) | Alternative to haloperidol; more sedating; useful in mixed delirium with nausea |
| Midazolam | 1–5 mg SC/IV q1h PRN or infusion 0.5–2 mg/h | SC, IV | Reserved for refractory terminal agitation; can worsen delirium in non-terminal patients; used for palliative sedation |
| Phenobarbital | Loading: 200 mg IV/SC; infusion 0.5–1 mg/kg/h | SC, IV, PR | Last resort for refractory terminal agitation; used in palliative sedation protocols |
- Calm, quiet environment with familiar objects, photos, clock, calendar
- Reorient frequently; have familiar family members present
- Maintain sleep-wake cycle (lights on during day, minimize nighttime disturbances)
- Minimize restraints (chemical and physical) — restraints worsen agitation
- Ensure glasses and hearing aids are in place
- Educate family: delirium is a medical condition, not personal choice or “going crazy”
13 Fatigue, Cachexia & Anorexia
Cancer-related fatigue is the most common symptom in advanced cancer (60–90% prevalence) and is distinct from normal tiredness — it is not relieved by rest. Cancer cachexia is a multifactorial syndrome defined by ongoing muscle loss (with or without fat loss) that cannot be fully reversed by conventional nutritional support and leads to progressive functional impairment.
Cancer Cachexia Staging
| Stage | Features | Management Focus |
|---|---|---|
| Pre-cachexia | Weight loss ≤5%; anorexia, metabolic changes (impaired glucose tolerance, inflammation) | Nutritional counseling, exercise, monitoring |
| Cachexia | Weight loss >5% in 6 months (or BMI <20 + weight loss >2%, or sarcopenia + weight loss >2%); reduced food intake; systemic inflammation | Multimodal: nutrition, exercise, pharmacologic, psychosocial |
| Refractory cachexia | Advanced/rapidly progressive cancer; unresponsive to anticancer treatment; low performance status (ECOG 3–4); survival <3 months | Symptom relief only; avoid invasive nutritional interventions (TPN, feeding tubes); focus on comfort |
Pharmacologic Appetite Stimulants
| Agent | Dose | Evidence/Notes |
|---|---|---|
| Dexamethasone | 2–4 mg PO daily | Short-term appetite stimulation (2–4 weeks); limited weight gain; best for short prognosis; side effects limit long-term use |
| Megestrol acetate (Megace) | 400–800 mg PO daily | Appetite stimulation, some weight gain (mostly fat/water, not lean mass); thromboembolic risk; adrenal suppression; onset 2–4 weeks |
| Mirtazapine | 7.5–15 mg PO at bedtime | Dual benefit: appetite stimulation + antidepressant/anxiolytic; weight gain via H1 antagonism; sedation at lower doses (more sedating at 7.5 mg than 15 mg) |
| Dronabinol | 2.5 mg PO BID before meals | Appetite stimulation; modest evidence; sedation, dysphoria, dizziness |
| Olanzapine | 2.5–5 mg PO daily | Appetite stimulation + antiemetic; weight gain; emerging evidence in cachexia |
Fatigue Management
- Rule out reversible causes: anemia, hypothyroidism, depression, deconditioning, sleep disturbance, infection, electrolyte abnormalities, medication side effects (opioids, benzodiazepines)
- Non-pharmacologic: Exercise (moderate aerobic exercise is the most evidence-based intervention for cancer-related fatigue); energy conservation; sleep hygiene; psychosocial support
- Pharmacologic: Methylphenidate 5–10 mg PO morning and early afternoon (evidence supports use in cancer-related fatigue); dexamethasone 2–4 mg daily (short-term); modafinil (limited evidence)
14 Secretions, Pruritus, Hiccups & Wound Care
Death Rattle (Terminal Secretions)
The “death rattle” is noisy, gurgling breathing caused by accumulation of pharyngeal secretions in a patient too weak to clear them, typically in the last hours to days of life. It occurs in 23–92% of dying patients. It is generally more distressing to family members than to the patient (who is usually obtunded).
Management of Death Rattle
| Intervention | Details |
|---|---|
| Non-pharmacologic | Position patient on side or semi-prone (allows secretions to drain by gravity); elevate head of bed; gentle suctioning of oral secretions only (avoid deep suctioning — stimulates more secretions and is uncomfortable); educate family that rattling does not indicate distress/drowning |
| Glycopyrrolate (Robinul) | 0.2–0.4 mg SC/IV q4–6h or 1 mg PO TID; does NOT cross BBB (no CNS sedation/delirium — preferred in patients with delirium) |
| Hyoscine butylbromide (Buscopan) | 20 mg SC q4–6h; does NOT cross BBB |
| Atropine 1% ophthalmic drops | 1–2 drops sublingual q4–6h; readily available; crosses BBB (delirium risk) |
| Scopolamine (hyoscine hydrobromide) | 0.3–0.6 mg SC q4–6h or 1.5 mg transdermal patch q72h; crosses BBB (sedating — may be beneficial in agitated patients; delirium risk) |
Pruritus in Palliative Care
| Cause | Mechanism | Treatment |
|---|---|---|
| Opioid-induced | Histamine release (morphine > others); central mu-receptor activation | Opioid rotation; antihistamines (hydroxyzine 25–50 mg); ondansetron 4–8 mg; nalbuphine 2.5–5 mg IV; low-dose naloxone infusion |
| Cholestatic (biliary obstruction) | Bile salt deposition in skin; central opioidergic pathway | Cholestyramine 4 g PO TID; rifampin 150 mg BID; sertraline 75–100 mg; naltrexone (caution in patients on opioids) |
| Uremic | Multifactorial (uremic toxins, xerosis, peripheral neuropathy) | Emollients; gabapentin 100–300 mg post-dialysis (or adjusted in non-dialysis); UVB phototherapy; activated charcoal |
| Malignant / paraneoplastic | Cytokine-mediated (lymphoma, leukemia) | Treat underlying malignancy; mirtazapine 7.5–15 mg; paroxetine 10–20 mg; doxepin 10–25 mg |
Hiccups (Singultus)
Intractable hiccups can be severely debilitating. Common causes in palliative care include gastric distension, diaphragmatic irritation (hepatomegaly, ascites, phrenic nerve involvement), CNS lesions, and metabolic derangements (uremia, hyponatremia).
| Agent | Dose | Mechanism |
|---|---|---|
| Chlorpromazine | 25–50 mg PO TID–QID | D2 blockade; only FDA-approved drug for hiccups |
| Baclofen | 5–10 mg PO TID | GABA-B agonist; interrupts reflex arc |
| Gabapentin | 300 mg PO TID, titrate up | Central neuromodulation |
| Metoclopramide | 10 mg PO QID | Prokinetic; reduces gastric distension |
| Simethicone | 40–80 mg PO QID | Reduces gastric distension (gas) |
Malignant Wounds
Fungating wounds from cutaneous tumor involvement cause odor, exudate, bleeding, and pain. Management is palliative, not curative:
- Odor: Topical metronidazole 0.75% gel applied to wound BID (reduces anaerobic bacterial load); activated charcoal dressings; room deodorizers
- Bleeding: Hemostatic dressings (alginate); topical tranexamic acid (500 mg crushed in saline); topical sucralfate paste; adrenaline-soaked gauze (1:1000); gentle dressing changes; palliative radiation for recurrent bleeding
- Exudate: Absorptive dressings (foam, alginate, hydrofiber); gentle non-adherent primary layer
- Pain: Topical morphine gel (0.1% morphine in hydrogel); topical lidocaine before dressing changes; systemic analgesia
15 Goals of Care Conversations
Goals-of-care conversations are structured discussions that explore what matters most to the patient in the context of their illness, align treatment plans with patient values, and support shared decision-making. They are a core competency in palliative medicine.
SPIKES Protocol for Difficult Conversations
| Step | Description | Example Phrases |
|---|---|---|
| S — Setting | Ensure privacy, sit down, minimize interruptions, involve appropriate people | “Let’s find a quiet place to talk. Who would you like present for this conversation?” |
| P — Perception | Assess what the patient/family already knows about their illness | “What is your understanding of where things stand with your illness?” |
| I — Invitation | Ask permission to share information; determine how much detail the patient wants | “Would it be okay if I share what we have found?” “Are you someone who likes to know all the details, or prefer the big picture?” |
| K — Knowledge | Deliver the medical information clearly and in lay terms; use a warning shot | “I wish I had better news…” “Unfortunately, the scan shows that the cancer has spread…” |
| E — Emotions | Acknowledge and respond to emotional reactions with empathy | Use NURSE statements (see below); allow silence; “I can see this is really hard to hear.” |
| S — Strategy/Summary | Discuss next steps and plan; check understanding | “Given what matters most to you, here is what I recommend…” “What questions do you have?” |
NURSE Statements for Empathic Response
| Statement | Description | Example |
|---|---|---|
| N — Naming | Identify the emotion | “It sounds like you are feeling really frightened.” |
| U — Understanding | Express understanding | “I can understand why you would feel that way.” |
| R — Respecting | Praise coping or strength | “I can see how hard you have been fighting this.” |
| S — Supporting | Express commitment to continued care | “I want you to know that we will be with you every step of the way.” |
| E — Exploring | Invite the patient to share more | “Tell me more about what you are most worried about.” |
Ask-Tell-Ask Framework
An efficient communication structure for conveying medical information:
- Ask: “What do you already know about your condition?” (assess baseline understanding)
- Tell: Provide information concisely, in language the patient understands, addressing any gaps or misconceptions identified
- Ask: “What questions do you have about what I just shared?” (confirm understanding)

16 Breaking Bad News & Prognostic Disclosure
Prognostic Communication
Most patients with serious illness want some information about prognosis, though preferences vary. Key principles for prognostic disclosure:
- Ask before telling: “Would it be helpful if I shared what I think is likely to happen?”
- Frame as ranges: “I’m thinking in terms of weeks to months” rather than specific dates; use time frames: hours-to-days, days-to-weeks, weeks-to-months, months-to-years
- Hope for the best, plan for the worst: “I hope you will beat the odds, but I think it’s important for us to plan for the possibility that time may be limited.”
- Avoid false precision: Clinicians overestimate survival by a factor of 3–5× on average (Christakis & Lamont, BMJ 2000)
- Acknowledge uncertainty: “I wish I could tell you exactly how much time you have, but no one can predict that precisely.”
Cultural Considerations in Prognostic Disclosure
In many cultures, direct disclosure of terminal prognosis to the patient is considered harmful or disrespectful. Some families request that the diagnosis or prognosis be disclosed only to family members, not to the patient. Approach: ask the patient how they prefer to receive information and who should be involved in discussions. Respect cultural norms while fulfilling ethical duties of informed consent.
Common Physician Phrases to Use and Avoid
| Avoid | Use Instead | Rationale |
|---|---|---|
| “There is nothing more we can do” | “We are going to shift our focus to keeping you comfortable and making the most of your time” | There is always something that can be done; pivots away from abandonment |
| “Do you want us to do everything?” | “I want to understand what matters most to you so we can focus on that” | “Everything” is ambiguous and leads to misunderstanding |
| “Withdrawal of care” | “Transition to comfort-focused care” | Care is never withdrawn; only the goals of care change |
| “You’re a fighter” | Acknowledge their experience without implying that dying = giving up | Can make patients feel they “failed” if they choose comfort care |
17 Surrogate Decision-Making & Family Meetings
Surrogate Decision-Making Hierarchy
When a patient lacks decision-making capacity and has no advance directive or designated healthcare proxy, state laws define a hierarchy of surrogate decision-makers (varies by state, but commonly):
- Court-appointed guardian (if applicable)
- Designated healthcare proxy/DPOA-HC (if exists)
- Spouse or domestic partner
- Adult children
- Parents
- Adult siblings
- Other relatives or close friends
Standards for Surrogate Decision-Making
| Standard | Definition | When Used |
|---|---|---|
| Substituted judgment | What would the patient have wanted? Based on known values, prior statements, lifestyle | Preferred standard when patient’s wishes can be reasonably inferred |
| Best interest | What would a “reasonable person” want in this situation? Weighing benefits vs burdens | Used when patient’s wishes are unknown (never expressed preferences, never had capacity) |
Structured Family Meeting Framework
- Pre-meeting: Review medical facts; align medical team on prognosis and recommendations; identify participants; set agenda; designate meeting leader
- Introductions and ground rules: Introduce all participants; set expectation for length; clarify purpose
- Assess understanding: “What is your understanding of [patient’s] medical situation?”
- Provide medical update: Clear, jargon-free language; headline first (“I’m worried that…”)
- Respond to emotions: NURSE statements; allow silence and tears
- Explore values and goals: “If [patient] could speak for themselves, what would they say?” “What would a good day look like for [patient]?”
- Make a recommendation: “Based on what you have told me about [patient’s] values, I would recommend…” (clinicians should not ask families to “decide” but rather make a recommendation aligned with patient values)
- Establish plan and follow-up: Summarize decisions; identify next steps; schedule follow-up
18 Code Status Discussions (DNR/DNI/AND)
Definitions
| Order | Meaning | What It Covers | What It Does NOT Cover |
|---|---|---|---|
| Full Code | Attempt all resuscitative measures | CPR, defibrillation, intubation, vasopressors, ICU transfer | — |
| DNR (Do Not Resuscitate) | No CPR if cardiac/respiratory arrest occurs | No chest compressions, no defibrillation | Does NOT limit other treatments (antibiotics, transfusions, ICU, intubation for non-arrest indications, surgery) |
| DNAR (Do Not Attempt Resuscitation) | Same as DNR; preferred terminology (emphasizes that resuscitation is an “attempt,” not a guarantee) | Same as DNR | Same as DNR |
| DNI (Do Not Intubate) | No endotracheal intubation or mechanical ventilation | No intubation; may or may not include non-invasive ventilation (BiPAP/CPAP) | Does not address CPR (must specify separately) |
| AND (Allow Natural Death) | Allow death to occur naturally without intervention if arrest occurs | Same scope as DNR/DNI but framed positively | Same caveats — specify other limitations separately |
| CMO (Comfort Measures Only) | Focus exclusively on comfort; discontinue all non-comfort interventions | Analgesics, anxiolytics, secretion management, positioning, spiritual care; discontinue monitoring, labs, IV fluids, antibiotics, vasopressors | DNR/DNI is implicit in CMO |
Approach to Code Status Discussion
- Code status should be discussed in the context of overall goals of care, not in isolation
- Provide a medical recommendation based on prognosis and likely outcomes of CPR (not just “what do you want?”)
- Educate about realistic CPR outcomes: overall survival to hospital discharge after in-hospital cardiac arrest is ~25% for shockable rhythms and ~10% for non-shockable; for patients with metastatic cancer, survival to discharge is <6%; for patients with multi-organ failure, <2%
- For patients where CPR would not be medically beneficial, it is ethically appropriate to recommend DNR: “Based on your medical condition, I do not recommend CPR because it would not work and would cause suffering.”
19 Advance Directives & Living Wills
Types of Advance Directives
| Type | Description | Key Features |
|---|---|---|
| Living Will | Written document specifying treatment preferences if the patient becomes incapacitated | Takes effect only when patient lacks capacity; typically addresses life-sustaining treatments (mechanical ventilation, CPR, artificial nutrition/hydration, dialysis); varies by state law; may be limited by vague or overly specific language |
| Durable Power of Attorney for Healthcare (DPOA-HC) | Legal document designating a surrogate (proxy, healthcare agent) to make medical decisions when the patient lacks capacity | More flexible than living will (surrogate can respond to unforeseen situations); takes effect only when patient lacks capacity; surrogate should know patient’s values and wishes; most experts consider DPOA-HC more valuable than a living will alone |
| Combined Advance Directive | Single document combining living will and DPOA-HC | Most comprehensive; recommended approach |
Key Legal Principles
- Advance directives are governed by state law — requirements vary by jurisdiction (witnessing, notarization, specific forms)
- The Patient Self-Determination Act (1990) requires hospitals/healthcare facilities to inform patients of their right to create advance directives and to document whether they have one
- An advance directive from one state is generally honored in other states, but it is advisable to complete forms for the current state of residence
- Patients can revoke or modify advance directives at any time while they retain capacity
- Advance directives do not replace verbal expressions of current wishes — a patient with capacity can override their own advance directive at any time
Advance care planning is a process, not a form. The conversation is more important than the document. Key components:
- Explore patient’s understanding of their illness and prognosis
- Identify what matters most: values, goals, fears, acceptable/unacceptable outcomes
- Discuss specific scenarios: “If your heart stopped…” “If you could not eat on your own…” “If you could not recognize your family…”
- Designate a surrogate decision-maker who knows the patient’s wishes
- Document preferences and ensure accessibility in the medical record
- Revisit periodically and after major health changes
20 POLST/MOLST Paradigm
The POLST (Physician Orders for Life-Sustaining Treatment) is a brightly colored (usually pink or green) medical order form that translates patient wishes into actionable medical orders. Unlike advance directives (which are legal documents), POLST is a medical order signed by a clinician.
POLST vs Advance Directive
| Feature | Advance Directive | POLST |
|---|---|---|
| Type | Legal document | Medical order |
| Who completes | Patient (with witnesses/notary) | Clinician after conversation with patient/surrogate |
| Applicability | Any adult; recommended for all | Patients with serious illness or advanced frailty; NOT for healthy adults |
| When effective | Only when patient lacks capacity | Immediately; regardless of capacity status |
| EMS actionable? | No (EMS cannot interpret legal documents) | Yes — EMS must follow POLST orders |
| Portability | Must be retrieved from medical record | Travels with the patient (posted on refrigerator, in chart) |
| Content | General wishes, values, proxy designation | Specific orders: CPR yes/no, medical interventions level, artificial nutrition |
| Name variations | — | POLST, MOLST, POST, COLST, MOST, TPOST (varies by state) |
Standard POLST Sections
- Section A — Cardiopulmonary Resuscitation: Attempt CPR / Do Not Attempt Resuscitation (DNAR)
- Section B — Medical Interventions: Full treatment / Selective treatment (IV fluids, cardiac monitoring, but no intubation) / Comfort-focused treatment only
- Section C — Artificially Administered Nutrition: Long-term artificial nutrition (PEG tube) / Time-limited trial / No artificial nutrition
21 Capacity Assessment & Informed Consent
Decision-making capacity is a clinical determination (made by any physician, not just psychiatrists) that a patient can make a specific medical decision at a specific time. It is distinct from competence, which is a legal determination made by a court.
Four Elements of Decision-Making Capacity (Appelbaum Criteria)
| Element | Assessment Question | Explanation |
|---|---|---|
| Understanding | “Can you tell me in your own words what your medical condition is and what we are proposing?” | Patient can comprehend the disclosed information about diagnosis, treatment, risks, benefits, and alternatives |
| Appreciation | “Do you believe you have this condition? How do you think the proposed treatment might help you?” | Patient can apply the information to their own situation (not just recite facts abstractly); absence of pathologic denial |
| Reasoning | “How did you arrive at your decision? What factors are you considering?” | Patient can engage in a rational process of weighing options; does not require a “rational” decision — only a rational process |
| Expression of Choice | “What would you like us to do?” | Patient can clearly communicate a consistent decision (not rapidly vacillating or refusing to answer) |
Key Principles
- Capacity is decision-specific: a patient may have capacity for simple decisions (accepting food) but not complex ones (surgery consent)
- Capacity can fluctuate: reassess at different times of day (delirium often worse at night)
- Capacity is NOT determined by the outcome of the decision — a patient can refuse life-saving treatment if they have capacity; the threshold for capacity assessment may increase with the severity of consequences (sliding scale approach)
- Diagnosis alone does not determine capacity: patients with dementia, psychiatric illness, or intellectual disability may retain capacity for some decisions
- If capacity is unclear after bedside assessment, a formal psychiatric consultation may be obtained, but this is not mandatory
22 Medicare Hospice Benefit & Eligibility
The Medicare Hospice Benefit (MHB), established in 1982, is the primary funding mechanism for hospice care in the United States. To be eligible, the patient must meet two criteria: (1) a physician certifies a prognosis of ≤6 months if the disease runs its usual course, and (2) the patient elects comfort-focused care (forgoing curative treatment for the terminal diagnosis).
Medicare Hospice Benefit Structure
| Feature | Details |
|---|---|
| Certification periods | Two 90-day periods, then unlimited 60-day periods; face-to-face encounter required before recertification for 3rd+ benefit period |
| Covered services | Nursing visits, physician/NP visits, SW, chaplain, aide, medications related to terminal Dx, DME (hospital bed, O2, wheelchair), supplies, bereavement support (13 months), volunteer services |
| Patient cost | No deductible; 5% coinsurance for respite care; medication copays ≤$5 |
| What patient forfeits | Medicare Part A coverage for treatment of the terminal diagnosis; retains Medicare Part A/B for unrelated conditions |
| Revocation | Patient can revoke hospice at any time for any reason and return to standard Medicare; can re-elect later |
General Hospice Eligibility Guidelines
- Life-limiting condition with prognosis ≤6 months
- Patient has elected hospice level of care (understands comfort focus)
- Functional decline: PPS ≤70% (dependent in some activities), declining over time
- Nutritional decline: Unintentional weight loss >10% over 6 months, declining albumin, progressive difficulty eating
- Multiple comorbidities contributing to overall decline
- Frequent emergency or hospital visits despite optimal treatment
- Patient is not a candidate for or declines further disease-modifying treatment
23 Hospice Levels of Care
The Medicare Hospice Benefit defines four levels of care, each with different per-diem reimbursement rates and staffing requirements.
| Level | Setting | Indication | Staffing | Duration |
|---|---|---|---|---|
| Routine Home Care (RHC) | Patient’s home, nursing facility, ALF | Standard hospice care; patient is stable or slowly declining | Intermittent nursing visits (typically 1–3×/week), aide, SW, chaplain, volunteer as needed | Ongoing (majority of hospice days) |
| Continuous Home Care (CHC) | Patient’s home | Acute symptom crisis requiring ≥8 hours of predominantly nursing care in a 24-hour period to maintain patient at home | RN and/or aide providing a minimum of 8 hours, with >50% being skilled nursing | Short-term (during crisis only); must document resolution criteria; most expensive per-diem level |
| General Inpatient Care (GIP) | Hospice inpatient unit, hospital, or SNF (if contracted) | Acute symptom management that cannot be managed in the home setting (refractory pain, intractable vomiting, severe dyspnea, acute delirium/agitation) | 24/7 nursing care; physician available | Short-term; must document why home management has failed; convert to RHC once symptoms controlled |
| Inpatient Respite Care | Hospice inpatient unit, hospital, or SNF | Relief for the caregiver (caregiver burnout/need for temporary break) | Standard facility staffing | Maximum 5 consecutive days per admission; 5% coinsurance applies |
24 Disease-Specific Hospice Criteria
Medicare Local Coverage Determinations (LCDs) provide disease-specific guidelines for hospice eligibility. Below are the key criteria for major disease categories, based on the National Hospice and Palliative Care Organization (NHPCO) guidelines.
Cancer
Disease with distant metastases at presentation OR progression from an earlier stage despite curative therapy; declining performance status (PPS ≤70%); continued decline despite treatment. Certain cancers (pancreatic, hepatocellular, GBM, metastatic melanoma) have inherently poor prognosis. Declining nutritional status and functional decline support eligibility.
Heart Failure (CHF)
- NYHA Class IV symptoms (dyspnea at rest) despite optimal medical therapy (ACEi/ARB, beta-blocker, diuretics, aldosterone antagonist)
- EF ≤20% (or preserved EF with refractory symptoms)
- Resistant to maximum tolerated diuretics
- Recurrent hospitalizations (≥3 in past 12 months) for CHF exacerbation
- History of cardiac arrest, syncope, or CVA
- Supplemental documentation: renal insufficiency (BUN >25, Cr >1.5), refractory hyponatremia (Na <134), cardiac cachexia
COPD/Chronic Lung Disease
- Disabling dyspnea at rest, poorly responsive to bronchodilators
- FEV1 <30% predicted (after bronchodilator)
- Progressive disease with increasing ER visits/hospitalizations despite optimal therapy
- Cor pulmonale / right heart failure
- Resting hypoxemia (PaO2 ≤55 mmHg on supplemental O2 or SpO2 ≤88%)
- Resting tachycardia >100 bpm
- Unintentional weight loss >10% over 6 months
Dementia
- FAST Stage ≥7A (cannot speak >5 words, ambulatory ability lost, ability to sit up lost, ability to smile lost, cannot hold head up)
- At least one of the following complications in the past 12 months: aspiration pneumonia, pyelonephritis or UTI, septicemia, decubitus ulcers (Stage 3–4), recurrent fevers, inability to maintain sufficient caloric intake (weight loss >10% or albumin <2.5)
- PPS ≤40%
Renal Disease (ESRD)
- Patient is not seeking or has discontinued dialysis
- CrCl <10 mL/min (<15 if diabetic)
- Serum Cr >8.0 (>6.0 if diabetic)
- Uremia symptoms: nausea, vomiting, confusion, intractable pruritus, restless legs, fluid overload
- Oliguria (<400 mL/24h)
- GFR <10 mL/min
Liver Disease
- End-stage liver disease (Child-Pugh C score ≥10; MELD ≥21) not a transplant candidate
- PT >5 sec over control or INR >1.5
- Albumin <2.5 g/dL
- Refractory ascites or spontaneous bacterial peritonitis
- Hepatorenal syndrome
- Hepatic encephalopathy refractory to treatment
- Recurrent variceal bleeding despite treatment
Stroke & Coma
- Post-CVA in comatose state at onset: Coma or persistent vegetative state beyond 3 days
- Post-CVA in non-comatose patients: Dysphagia preventing adequate oral intake, PPS ≤40%, post-stroke dementia (FAST ≥7)
- Karnofsky ≤40% or PPS ≤40%
ALS / Neurodegenerative Disease
- Critically impaired breathing capacity: vital capacity <30% predicted, significant dyspnea at rest, refusal of artificial ventilation
- Rapid progression of disease with critical nutritional impairment (inability to maintain adequate oral intake, refusal of PEG tube, weight loss)
- Life-threatening complications: recurrent aspiration pneumonia, decubitus ulcers Stage 3–4, sepsis
25 Hospice Recertification, Revocation & Discharge
Recertification
- At the start of each new benefit period (two 90-day periods, then 60-day periods), the hospice physician and attending physician must recertify that the patient continues to have a prognosis of ≤6 months if the disease runs its usual course
- Beginning with the 3rd benefit period, a face-to-face encounter (by hospice physician or hospice NP) is required within 30 days prior to the start of the new period to confirm ongoing eligibility
- Document: continued decline, functional status, nutritional status, disease-specific criteria
Revocation
A patient may revoke the hospice benefit at any time for any reason, effective the date of revocation. Revocation does not require a reason. The patient returns to standard Medicare and may re-elect hospice at any time. Common reasons for revocation: desire to pursue curative/disease-directed treatment, feeling “better,” family pressure, need for a covered procedure or hospitalization not related to the terminal Dx but denied under hospice.
Live Discharge
The hospice may discharge a patient if: (1) the patient’s condition improves and they no longer meet eligibility criteria (extended prognosis, improved function); (2) the patient moves out of the service area; (3) the patient exhibits behavior that makes care delivery unsafe (rare, must document extensively and attempt to resolve). The hospice must provide a discharge plan and ensure continuity of care.
26 Interdisciplinary Team & Care Delivery
Hospice care is delivered by an interdisciplinary team (IDT), which is distinct from a multidisciplinary team. In an IDT, team members collaborate, share expertise across disciplines, and develop a unified care plan; in a multidisciplinary team, disciplines work in parallel.
Core IDT Members
| Role | Responsibilities | Required by Medicare? |
|---|---|---|
| Hospice Medical Director (Physician) | Certifies/recertifies eligibility, oversees plan of care, manages complex symptoms, leads IDT meetings, face-to-face encounters | Yes |
| Registered Nurse (Case Manager) | Assessment, care coordination, symptom management, medication management, patient/family education, on-call triage | Yes |
| Social Worker (MSW) | Psychosocial assessment, counseling, advance care planning, resource coordination, caregiver support, bereavement | Yes |
| Chaplain/Spiritual Counselor | Spiritual assessment, spiritual/existential support, rituals, grief counseling, meaning-making, legacy work | Yes (counseling services must be available) |
| Hospice Aide (CNA/HHA) | Personal care (bathing, dressing, grooming, toileting), light housekeeping, companionship | Yes (if aide services are part of the care plan) |
| Volunteer | Companionship, respite for caregiver, errands, vigil sitting, bereavement support, administrative | Yes (hospice must use volunteers for ≥5% of patient care hours) |
| Bereavement Coordinator | Bereavement assessment, counseling, support groups for up to 13 months after death | Yes (13 months of bereavement support required) |
IDT Care Conference
The IDT meets regularly (typically every 2 weeks) to review each patient’s plan of care, discuss symptom management, update goals of care, and coordinate services. The plan of care is updated based on IDT input and the patient’s changing needs.
27 Cancer-Related Palliative Emergencies
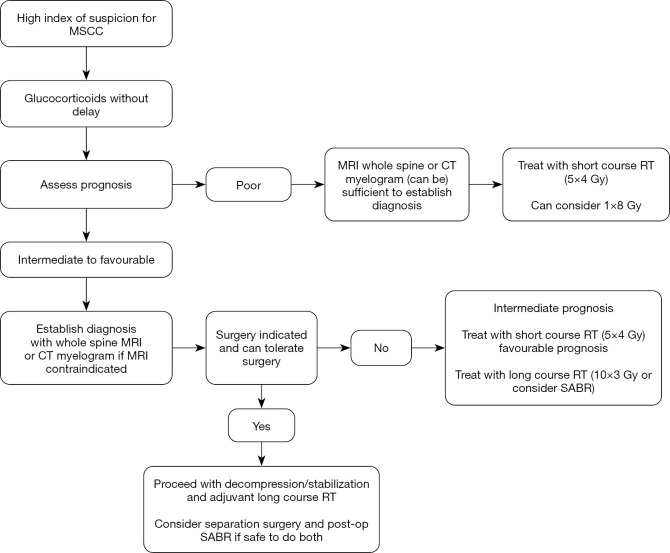
Presentation: Back pain (often worse when lying down), progressive lower extremity weakness, sensory level, bowel/bladder dysfunction (late). Most common: thoracic spine. Common primaries: lung, breast, prostate, myeloma, lymphoma, renal.
Diagnosis: Urgent MRI of entire spine (multiple levels in 30% of cases).
Treatment (prognosis-dependent):
- Dexamethasone 10–16 mg IV bolus immediately (then 4–16 mg q6h) — reduces vasogenic edema; give BEFORE MRI if suspicion is high
- Radiation therapy (standard) or surgical decompression (if prognosis >3 months, single level, and mechanical instability)
- In comfort-focused patients: dexamethasone for symptom relief + pain management
Prognosis: Neurologic status at time of treatment initiation is the strongest predictor of outcome — ambulatory patients have 70–90% chance of maintaining ambulation; paraplegia at presentation rarely reverses.
Presentation: Facial/upper extremity edema, venous distension of neck and chest wall, dyspnea, cough, headache (worse bending forward). Most common cause: lung cancer (especially SCLC), lymphoma, mediastinal tumors.
Treatment:
- Elevate head of bed; supplemental O2
- Dexamethasone 8–16 mg IV (especially if lymphoma or radiation-responsive tumor)
- SVC stenting (interventional radiology) for rapid relief
- Radiation therapy or systemic chemotherapy based on histology
- Diuretics may provide modest relief but are often ineffective
- In comfort-focused patients: dexamethasone, opioids for dyspnea, anxiolytics
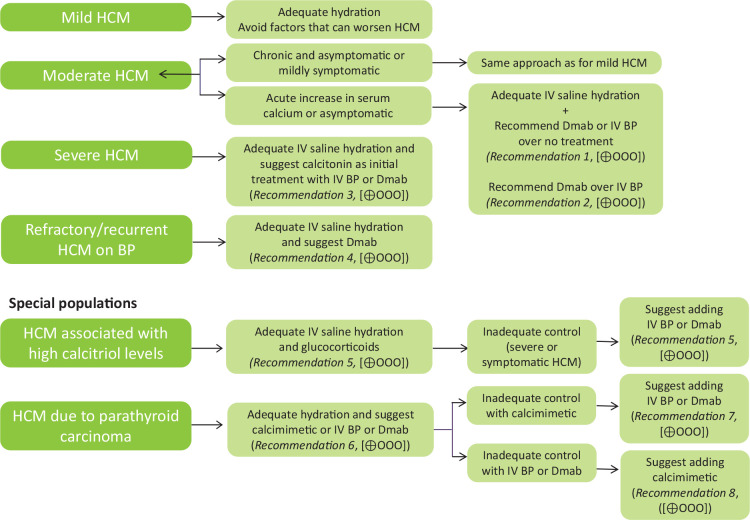
Hypercalcemia of Malignancy
Most common metabolic emergency of cancer (10–30% of cancer patients). Three mechanisms: (1) humoral (PTHrP-mediated) — squamous cell cancers, renal; (2) osteolytic — breast, myeloma; (3) calcitriol-mediated — lymphoma.
| Severity | Corrected Ca (mg/dL) | Symptoms | Treatment |
|---|---|---|---|
| Mild | 10.5–12 | Often asymptomatic, mild fatigue, constipation | Hydration, ambulation |
| Moderate | 12–14 | Polyuria, polydipsia, nausea, confusion, weakness | IV NS hydration (200–300 mL/h); zoledronic acid 4 mg IV or denosumab 120 mg SC; calcitonin 4 IU/kg SC q12h (rapid but temporary) |
| Severe | >14 | Stupor, coma, cardiac arrhythmias (short QT), renal failure | Aggressive IV hydration; zoledronic acid + calcitonin; consider dialysis if refractory; monitor cardiac |
28 Malignant Bowel Obstruction & Effusions
Malignant Bowel Obstruction (MBO)
MBO occurs in 5–42% of patients with advanced abdominal/pelvic cancers (ovarian, colorectal, gastric). In patients with peritoneal carcinomatosis, surgical intervention is often not feasible and carries high morbidity/mortality.
Medical Management of Inoperable MBO
| Agent | Role | Dose |
|---|---|---|
| Octreotide | Reduces GI secretions (antisecretory); decreases vomiting; most effective single agent | 100–300 mcg SC TID or 600–900 mcg/24h continuous SC/IV infusion |
| Dexamethasone | Reduces peritumoral edema and inflammation; may partially resolve obstruction | 8–16 mg IV/SC daily; trial for 5–7 days then taper |
| Haloperidol | Antiemetic (central D2 blockade) | 0.5–2 mg SC/IV q6–8h |
| Hyoscine butylbromide | Antisecretory, antispasmodic | 20 mg SC q4–6h or 60–120 mg/24h infusion |
| Opioids | Analgesia for colicky pain | Titrate to effect; consider fentanyl (less constipating than morphine) |
| Ranitidine/omeprazole | Reduce gastric acid secretion | Ranitidine 150–300 mg/day SC; omeprazole 40 mg IV daily |
Venting gastrostomy (PEG/G-tube): For patients with persistent high-volume vomiting despite maximal medical therapy, a percutaneous venting gastrostomy allows drainage of gastric contents and can dramatically improve quality of life by eliminating vomiting while allowing the patient to eat/drink for pleasure (with the understanding that intake will drain out).
NG tube: May be necessary short-term for acute decompression, but long-term NG tubes are uncomfortable and not appropriate for palliative management. Transition to medical management or venting gastrostomy.
Malignant Pleural Effusion
Common in advanced lung cancer, breast cancer, mesothelioma, lymphoma. Causes progressive dyspnea.
| Intervention | Indication | Notes |
|---|---|---|
| Therapeutic thoracentesis | Symptomatic relief; diagnostic | Rapid relief; recurs in 30 days in most; limit drainage to 1–1.5 L to prevent re-expansion pulmonary edema |
| Indwelling pleural catheter (IPC/PleurX) | Recurrent symptomatic effusion; prognosis >1 month | Tunneled catheter allowing outpatient drainage at home; can achieve spontaneous pleurodesis in ~50% over time |
| Chemical pleurodesis | Recurrent effusion with good lung re-expansion | Talc slurry via chest tube or talc poudrage via VATS; 70–90% success; requires 24–48h hospitalization |
Malignant Ascites
Common in ovarian, GI, and liver cancers. Causes abdominal distension, pain, dyspnea, early satiety, nausea.
- Paracentesis: Rapid symptom relief; can drain large volumes (up to 5 L or more with albumin replacement if >5 L); recurs rapidly (1–2 weeks)
- Indwelling peritoneal catheter: For recurrent drainage at home; reduces need for repeated paracentesis; infection risk (~5%)
- Diuretics (spironolactone + furosemide): Effective mainly for portal hypertension-related ascites; less effective for peritoneal carcinomatosis
29 Heart Failure & Device Deactivation
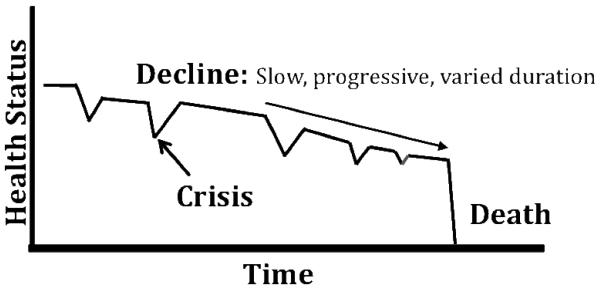
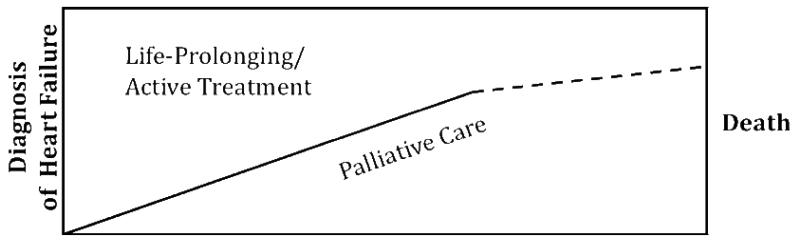
Palliative Care in Advanced Heart Failure
Heart failure follows an unpredictable trajectory with acute exacerbations and partial recovery, making prognostication difficult. Key palliative issues include refractory dyspnea, fatigue, depression, and device management.
Symptom Management in Advanced HF
| Symptom | Management |
|---|---|
| Dyspnea | Optimize diuresis; opioids (morphine 2.5–5 mg PO q4h or 1 mg IV PRN); fan therapy; oxygen if hypoxemic; positioning |
| Pain | Avoid NSAIDs (worsen fluid retention, renal function); acetaminophen first-line; opioids for moderate-severe pain; gabapentin for neuropathic pain |
| Depression | Sertraline (safest SSRI in HF); CBT; address spiritual/existential concerns |
| Fatigue | Energy conservation; exercise where feasible; treat anemia, hypothyroidism |
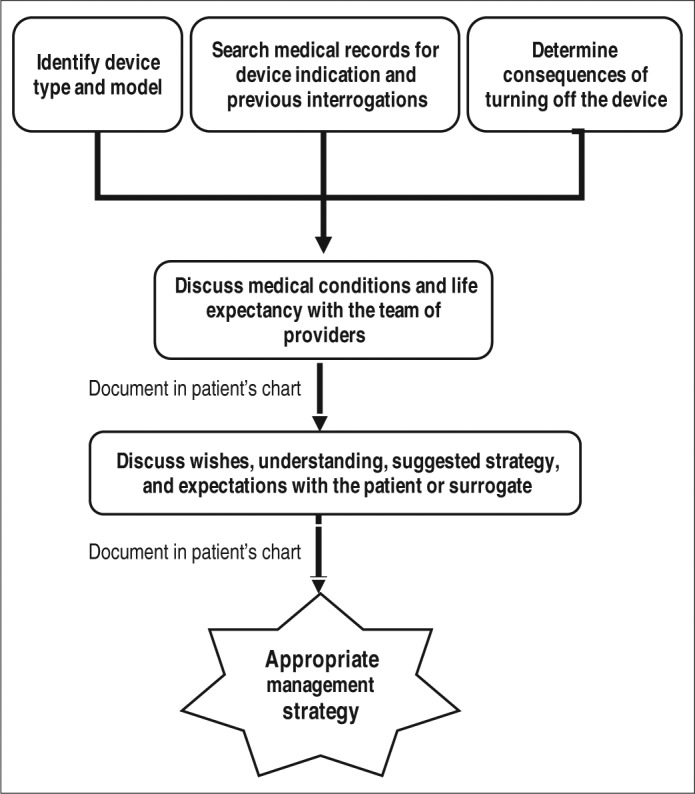
Cardiac Device Deactivation
Issue: An ICD can deliver painful shocks to a dying patient, preventing a peaceful death. The defibrillation function can be deactivated without affecting pacing function.
When to discuss: At time of goals-of-care transition; when patient is no longer a candidate for resuscitation; when patient is actively dying
Process:
- Goals-of-care conversation about the role of the ICD at this stage of illness
- Deactivation by device representative or cardiologist using a programmer
- If programmer not available urgently: place a magnet over the device to temporarily suspend shock therapy (does not deactivate permanently; must remain in place)
- Document discussion and consent
- Pacing function can be left on (does not cause discomfort) or deactivated separately if desired
Issue: Unlike an ICD, deactivating an LVAD will result in death within minutes to hours (depending on native cardiac function). LVAD deactivation is ethically and legally equivalent to withdrawing any other life-sustaining treatment.
Process:
- Extensive goals-of-care discussions with patient/family; ensure decision is informed and voluntary
- Ethics consultation recommended (but not required)
- Prepare comfort medications before deactivation: opioid infusion, benzodiazepine, anticholinergic for secretions
- Ensure patient is comfortable before and after LVAD is turned off
- Time to death after deactivation varies: minutes to hours
- Ideally performed in a controlled setting with palliative care involvement
30 End-Stage Pulmonary, Renal & Hepatic Disease
End-Stage COPD
COPD follows a trajectory of gradual decline punctuated by acute exacerbations, any of which may be fatal. Palliative care needs are often comparable to advanced cancer but receive less palliative attention.
- Dyspnea: Low-dose opioids (morphine 2.5–5 mg PO q4h) reduce dyspnea without clinically significant respiratory depression; fan therapy; pulmonary rehabilitation; optimize bronchodilators and oxygen
- Anxiety/panic: Low-dose benzodiazepines or buspirone; CBT for breathlessness anxiety
- Non-invasive ventilation (NIV): May be appropriate as a palliative measure for symptom relief even when intubation is not desired; establish time-limited trial with clear goals
- Decision-making: Discuss intubation/mechanical ventilation preferences in advance; many patients would decline intubation if fully informed of likely outcomes
End-Stage Renal Disease — Dialysis Withdrawal
Dialysis withdrawal is the second most common cause of death in dialysis patients in the US (~20% of deaths). It is ethically and legally appropriate when the patient decides the burdens of dialysis outweigh the benefits.
- Decision process: Ensure decision is voluntary, informed, and made with capacity; address depression/hopelessness
- Expected time to death: Typically 7–14 days after last dialysis (range: 1–30+ days depending on residual renal function)
- Symptom management: Pruritus (gabapentin, hydroxyzine); uremic nausea (ondansetron, haloperidol); dyspnea (opioids, oxygen); fluid overload (positioning); restless legs (gabapentin, opioids); myoclonus (clonazepam)
- Hospice enrollment: Transition to hospice at time of dialysis withdrawal
End-Stage Liver Disease
Key palliative issues in decompensated cirrhosis:
- Ascites: Serial paracentesis for refractory ascites; indwelling drain consideration; salt restriction; diuretics (spironolactone/furosemide)
- Hepatic encephalopathy: Lactulose (titrate to 3–4 soft BMs/day); rifaximin 550 mg BID; avoid sedatives
- Pain management: Avoid NSAIDs (renal failure, GI bleeding); acetaminophen ≤2 g/day is safe in stable cirrhosis; opioids: prefer hydromorphone (renally cleared), fentanyl; AVOID morphine (prolonged half-life, M6G accumulation)
- Variceal bleeding: If comfort-focused, may elect not to treat actively; dark towels, anxiolytics, maintain comfort
- Pruritus: Cholestyramine, rifampin, sertraline, naltrexone (caution with opioids)
31 Neurodegenerative Disease (ALS, Dementia)
Amyotrophic Lateral Sclerosis (ALS)
ALS is a progressive motor neuron disease with median survival of 3–5 years from diagnosis. Palliative care should be integrated from diagnosis.
Key Palliative Issues in ALS
| Issue | Management |
|---|---|
| Respiratory failure | Discuss NIV (BiPAP) early — improves survival and QOL; discuss tracheostomy/long-term ventilation preferences; when respiratory failure progresses, opioids + benzodiazepines for dyspnea; vital capacity <50% predicted triggers discussion |
| Dysphagia/Nutrition | Speech pathology; diet modification; PEG placement (ideally when FVC >50% — higher procedural risk if FVC <50%); discuss goals of artificial nutrition |
| Sialorrhea (excessive saliva) | Glycopyrrolate 1–2 mg PO TID; amitriptyline 25–50 mg; atropine drops SL; botulinum toxin injection to salivary glands; scopolamine patch |
| Pseudobulbar affect (pathologic laughing/crying) | Dextromethorphan/quinidine (Nuedexta) 20/10 mg BID; SSRIs; TCAs |
| Spasticity | Baclofen 5–20 mg TID; tizanidine; stretching; intrathecal baclofen for refractory |
| Communication loss | Eye-tracking devices, communication boards, speech-generating devices early in disease course |
| Advance care planning | Discuss early while patient can communicate: ventilation preferences, code status, feeding preferences, DPOA-HC designation |
Advanced Dementia
Advanced dementia (FAST Stage 7) is a terminal illness with median survival of 18 months. Care should focus on comfort, not prolonging life.
Key Palliative Issues in Advanced Dementia
| Issue | Approach |
|---|---|
| Feeding difficulties | Hand feeding is preferred over tube feeding; PEG tubes in advanced dementia do NOT reduce aspiration risk, do NOT prolong survival, do NOT prevent malnutrition, and are NOT recommended (AGA, AMDA, Choosing Wisely); careful hand feeding with pleasure-based approach |
| Pain assessment | Use PAINAD or ABBEY pain scale (nonverbal); trial of analgesics if behavioral signs of distress; observe for grimacing, guarding, vocalization, withdrawal |
| Infections | Pneumonia and UTI are common terminal events; antibiotics may be appropriate for comfort (treating symptoms of infection) but do not prolong survival in FAST Stage 7; discuss goals of antibiotic use with surrogate |
| Behavioral symptoms | Non-pharmacologic first (music, Montessori-based activities, familiar voices, reducing stimulation); avoid antipsychotics if possible (black box warning; increased mortality in elderly with dementia); low-dose haloperidol or risperidone for severe agitation if non-pharm measures fail |
| Hospitalization | Hospitalizations in advanced dementia cause harm (delirium, falls, pressure injuries, restraints) with little benefit; avoid transfers to ED/hospital when possible; treat in place |
32 Signs of Imminent Death
Recognizing the signs of imminent death (hours to days) is critical for preparing families, discontinuing non-beneficial interventions, and intensifying comfort measures.
Signs of Approaching Death
| Timeframe | Signs |
|---|---|
| Weeks before death | Progressive weakness and fatigue; increasing sleep; decreased appetite and fluid intake; progressive withdrawal from social interaction; difficulty swallowing medications; progressive cognitive decline |
| Days before death | Profound weakness (bed-bound); minimal to no oral intake; disorientation, confusion, or delirium; incontinence of urine and stool; decreased urine output (oliguria); “death rattle” (retained secretions); facial pallor, waxen appearance; cooling and mottling of extremities (begins distally) |
| Hours before death | Cheyne-Stokes respirations (cyclic crescendo-decrescendo breathing with apneic periods); mandibular breathing (“fish-out-of-water” jaw movements); cyanosis (central); anuria; obtundation/unconsciousness; loss of ability to swallow; cold, mottled, cyanotic extremities progressing proximally; pooling of blood (lividity); pulse thready and irregular; blood pressure unobtainable |
Predictive Signs with High Specificity for Death within 3 Days
- PPS ≤20% (totally bed-bound, unable to do any activity, confused or obtunded, minimal or sips-only intake)
- Respiration with mandibular movement (jaw drop breathing) — specificity >95% for death within days
- Cheyne-Stokes breathing
- Peripheral cyanosis (nail beds, lips)
- Decreased urine output (<100 mL in 12 hours)
- Non-reactive pupils
- Pulselessness of the radial artery
- “Death rattle” (audible rattling respirations)
- Explain that these changes are a normal part of the dying process, not signs of suffering
- Hearing may be the last sense to be lost — encourage family to continue speaking to the patient, say what they need to say
- The patient is not in pain from the breathing pattern changes; the rattle is not distressing to the patient (who is unconscious)
- No food or fluids are needed and may actually cause harm (aspiration, fluid overload); moistening lips with a sponge is appropriate
- Encourage family to touch, hold hands, play meaningful music, pray, or simply be present
- Prepare families for what death will look like: cessation of breathing, change in color, relaxation of muscles
33 Comfort Care & Medication Rationalization
Medication Rationalization (Deprescribing) in the Last Days
As a patient approaches death, many medications become unnecessary, burdensome, or impossible to administer. Systematic deprescribing is an active process of discontinuing medications that no longer serve the patient’s comfort goals.
Medication Assessment at End of Life
| Continue | Consider Discontinuing | Definitely Discontinue |
|---|---|---|
| Opioids (pain, dyspnea) | Antiepileptics (if no seizure risk) | Statins (no benefit at EOL) |
| Anxiolytics (anxiety, agitation) | Antidepressants (unless withdrawal risk) | Vitamins and supplements |
| Antiemetics (nausea) | Antihypertensives (risk of hypotension) | Osteoporosis medications |
| Anticholinergics (secretions) | Insulin (simplify to PRN for symptomatic hyperglycemia) | Preventive medications (aspirin for primary prevention) |
| Antipsychotics (delirium) | Antibiotics (comfort indication only) | Iron supplements |
| Laxatives (until oral intake stops) | Diuretics (if dyspnea relief) | Anticoagulants (unless symptomatic DVT/PE) |
| Corticosteroids (multiple symptom roles) | Bronchodilators (if still on nebulizer) | Diabetes medications (metformin, etc.) |
Route Conversion at End of Life
When the patient can no longer swallow, convert essential medications to alternative routes:
| Route | Appropriate Medications | Notes |
|---|---|---|
| Sublingual (SL) | Morphine/hydromorphone concentrated solution, lorazepam (Intensol), atropine drops, haloperidol | Small volumes only (≤1 mL); absorbed through oral mucosa; most useful route in home hospice |
| Subcutaneous (SC) | Morphine, hydromorphone, midazolam, haloperidol, glycopyrrolate, octreotide, dexamethasone | Equivalent bioavailability to IV for most drugs; can use continuous subcutaneous infusion (CSCI) via syringe driver; max volume ~2–3 mL/h |
| Rectal (PR) | Acetaminophen, morphine, hydromorphone, diazepam, prochlorperazine, bisacodyl, dexamethasone | Alternative when SL/SC not available; absorption variable; may be culturally unacceptable |
| Transdermal (TD) | Fentanyl patch, scopolamine patch | Not for acute dosing (slow onset); requires adequate subcutaneous tissue and circulation; unreliable in cachectic/diaphoretic patients |
| Intravenous (IV) | All injectable medications | Fastest onset; requires IV access; more common in inpatient settings |
34 Palliative Sedation & VSED
Palliative Sedation
Palliative sedation is the monitored use of medications intended to reduce consciousness to relieve refractory suffering at the end of life. A symptom is refractory when all available interventions have failed or no methods are available within the acceptable time frame or risk-benefit ratio.
Types of Palliative Sedation
| Type | Description | Indication |
|---|---|---|
| Proportionate palliative sedation | Titrate sedation to the minimum level needed to relieve symptoms; may include intermittent or mild sedation | Refractory symptoms (dyspnea, pain, agitation, seizures) at end of life |
| Continuous deep palliative sedation (CDS) | Continuous sedation to unconsciousness maintained until death | Imminently dying patient (hours to days) with refractory suffering unresponsive to all other measures |
Ethical Requirements for Palliative Sedation
- Patient is terminally ill with death expected within hours to days (for CDS)
- Suffering is refractory (all reasonable alternatives have been exhausted)
- Informed consent from patient (if capable) or surrogate
- Intent is to relieve suffering, not to hasten death (principle of double effect)
- Treatment should be proportionate (minimum dose to achieve comfort)
- Team consensus and documentation; ethics consultation recommended
Common Agents for Palliative Sedation
| Agent | Dose | Notes |
|---|---|---|
| Midazolam | 0.5–1 mg/h SC/IV infusion; titrate q15–30 min by 0.5–1 mg/h increments | First-line; rapid onset; easily titrated; may develop tachyphylaxis |
| Propofol | 10–70 mg/h (0.5–3 mg/kg/h) IV infusion | Very rapid onset; requires IV access; typically inpatient only; excellent efficacy |
| Phenobarbital | Loading: 200 mg IV/SC; infusion 0.5–2 mg/kg/h SC/IV | Useful when midazolam fails; very long half-life; can be given SC; used when midazolam tachyphylaxis develops |
| Levomepromazine | 12.5–25 mg SC q6–8h or infusion | Available outside US; combined neuroleptic and sedative properties |
Voluntarily Stopping Eating and Drinking (VSED)
VSED is a decision by a patient with decision-making capacity to refuse all food and fluids, with the understanding that this will lead to death. It is legally and ethically considered a patient’s right to refuse treatment (including nutrition/hydration).
- Time to death: Typically 7–14 days (range 1–21 days)
- Symptoms: Thirst and hunger (usually diminish after 1–3 days), dry mouth (treat with oral care, ice chips for comfort), delirium (later stages)
- Eligibility: Patient must have decision-making capacity at the time of initiation; support from palliative care/hospice team is essential
- Management: Meticulous mouth care; symptom management (opioids for discomfort, benzodiazepines for agitation); hospice enrollment; do NOT force or provide fluids if the patient has clearly expressed and documented their wishes
- Ethical considerations: If the patient loses capacity and asks for food/water during delirium, the pre-stated wishes (made with capacity) should generally be honored, per advance directive. This is an area of ethical complexity — advance planning and clear documentation are essential.
35 Pronouncement & Post-Mortem Care
Pronouncement of Death
When a hospice or palliative care patient dies, a healthcare provider must formally pronounce death and document the time, date, and findings.
Steps for Pronouncement
- Confirm identity of the patient
- Assess for absence of responsiveness (no response to verbal or tactile stimulation)
- Assess for absence of respirations (observe for at least 1 minute)
- Assess for absence of heart sounds (auscultate for at least 1 minute)
- Assess for absence of pulse (carotid for at least 1 minute)
- Assess for fixed, dilated pupils
- Note the time of death
- Document findings and notify attending physician, family, and hospice
Post-Mortem Care
- Allow family time with the body; respect cultural and religious practices
- Remove medical devices (IV lines, catheters) unless autopsy is planned or medical examiner case
- Clean the body, position with dignity, close eyes and mouth
- For patients with pacemakers: Must be removed before cremation (risk of explosion); document in death notification
- Notify the funeral home (per family arrangements)
- Complete death certificate (see Section 39)
- Initiate bereavement support for the family (hospice provides 13 months)
Complicated Grief
Normal grief involves waves of intense emotion that gradually diminish over time. Complicated grief (persistent complex bereavement disorder) involves prolonged, intense grief (>12 months in adults, >6 months in children) with persistent yearning, preoccupation with the deceased, difficulty accepting the death, and functional impairment. Risk factors include: sudden/unexpected death, death of a child, history of psychiatric illness, ambivalent or dependent relationship with the deceased, lack of social support, multiple concurrent losses.
36 Principle of Double Effect & Medical Futility
Principle of Double Effect
The principle of double effect (PDE) is an ethical framework that justifies an action with both a good effect and a foreseeable (but unintended) bad effect. It is widely invoked in palliative care to justify the use of opioids for symptom relief even when they may theoretically hasten death (though evidence shows this rarely occurs at appropriate doses).
Four Conditions for PDE
| Condition | Application to Palliative Care |
|---|---|
| 1. The action itself is morally good or neutral | Administering opioids for pain/dyspnea relief is a morally good medical act |
| 2. The good effect is intended; the bad effect is merely foreseen | Intent is to relieve suffering; possible hastening of death is foreseen but not intended |
| 3. The bad effect is not the means to the good effect | Symptom relief occurs through opioid receptor binding, not through death |
| 4. Proportionate reason exists | The severity of refractory suffering justifies the potential (small) risk |
Medical Futility
Medical futility refers to interventions that are unlikely to produce meaningful benefit. Two types:
| Type | Definition | Example |
|---|---|---|
| Quantitative (physiologic) futility | The intervention has virtually no chance of achieving its physiologic goal | CPR in a patient with multi-organ failure and refractory shock (estimated survival <1%) |
| Qualitative futility | The intervention may achieve a physiologic effect but will not benefit the patient in any meaningful way | ICU care for a patient in persistent vegetative state who will never regain consciousness |
Futility is controversial because it involves value judgments about what constitutes “meaningful benefit.” Most guidelines recommend: (1) transparent communication with patient/family, (2) ethics committee consultation, (3) institutional policy, (4) attempt to transfer care if disagreement persists. Unilateral withdrawal of life-sustaining treatment over patient/family objection remains legally risky in most US jurisdictions, though Texas has a formal futility process (Texas Advance Directives Act, §166.046).
37 Withholding vs Withdrawing Treatment
Ethically and legally, there is no moral distinction between withholding (not starting) and withdrawing (stopping) a life-sustaining treatment. Both are legally permissible when the patient (or surrogate) makes an informed decision or the treatment is determined to be futile. This is established in US common law (Cruzan v. Director, 1990) and endorsed by virtually all major medical and ethical organizations.
Withdrawal of Specific Life-Sustaining Treatments
| Treatment | Process | Anticipated Events | Comfort Measures |
|---|---|---|---|
| Mechanical ventilation | Two approaches: (1) Terminal extubation (remove ETT); (2) Terminal wean (gradually reduce FiO2 and rate). Extubation is preferred by most experts (faster, more definitive) | Dyspnea, tachypnea initially, then progressive respiratory failure over minutes to hours; some patients may breathe independently for hours to days | Pre-medicate: opioid bolus (morphine 2–4 mg IV) + midazolam 1–2 mg IV before extubation; continue infusions titrated to comfort; glycopyrrolate for secretions; suction before (not after) extubation |
| Vasopressors | Typically weaned simultaneously with other treatments or sequentially | Progressive hypotension; death usually within minutes to hours | Ensure adequate analgesia and sedation are in place before discontinuation |
| Artificial nutrition/hydration (ANH) | Discontinue TPN, tube feeds; if PEG tube in place, can stop feeds and either leave tube in situ or remove | Death over days to weeks; typically painless (dehydration at end of life may reduce edema, secretions, and dyspnea) | Meticulous mouth care; ice chips or swabs for dry mouth; reassure family that patient is not “starving” |
| Dialysis | Discontinue hemodialysis or peritoneal dialysis treatments | Uremia, somnolence, death over 7–14 days typically | Manage pruritus, nausea, dyspnea, myoclonus (see Section 30) |
| ICD / LVAD | See Section 29 | See Section 29 | See Section 29 |
38 Medical Assistance in Dying (MAID)
Medical Assistance in Dying (MAID), also called physician-assisted death (PAD) or physician-assisted suicide (PAS), refers to the practice of a physician prescribing (or in some jurisdictions, administering) a lethal medication to a terminally ill patient who requests it.
Legal Status (United States, as of 2025)
MAID is legal in: Oregon (1997 — Death with Dignity Act, the first), Washington, Vermont, California, Colorado, Hawai’i, Maine, New Jersey, New Mexico, and Washington D.C. The specific name and details of the law vary by jurisdiction. It is also legal in Canada (MAiD, including for non-terminal conditions under certain circumstances), Belgium, Netherlands, Luxembourg, Colombia, and several other countries.
Typical Safeguards (Oregon Model)
| Requirement | Details |
|---|---|
| Terminal illness | Prognosis ≤6 months, confirmed by two physicians |
| Capacity | Patient must have decision-making capacity at time of each request |
| Voluntariness | Request must be voluntary, without coercion |
| Requests | Two oral requests separated by ≥15 days (some states have reduced this), plus one written request with two witnesses |
| Waiting period | 15 days between first oral request and prescription (varies by state; some allow waiver if patient is expected to die sooner) |
| Self-administration | Patient must self-administer the medication (physician does not administer — distinguishes from euthanasia) |
| Psychiatric evaluation | Required if either physician suspects impaired judgment due to psychiatric disorder |
| Residency | Patient must be a resident of the state (some states do not require this) |
Distinctions
| Term | Definition | Legal Status (US) |
|---|---|---|
| Physician-assisted death (PAD) | Physician prescribes lethal medication; patient self-administers | Legal in select states (see above) |
| Euthanasia | Physician directly administers lethal medication to the patient | Illegal in all US states; legal in Belgium, Netherlands, Canada, Colombia |
| VSED | Patient voluntarily refuses food and fluid (see Section 34) | Legal everywhere (patient’s right to refuse treatment) |
| Palliative sedation | Sedation to unconsciousness for refractory suffering (see Section 34) | Legal and ethical when guidelines are followed |
39 Organ Donation, Autopsy & Death Certification
Organ Donation in Palliative Care
- Hospice and palliative care patients may be eligible for organ and tissue donation
- Tissue donation (corneas, skin, bone, heart valves) is possible for most patients, including those with cancer (with some exclusions); age limits are more flexible than for solid organ donation
- The organ procurement organization (OPO) should be contacted at time of death (required by CMS) to evaluate eligibility; do not pre-screen and exclude patients
- In some cases, donation after circulatory death (DCD) may be an option for hospice patients who die in the hospital, though this is uncommon
Autopsy
- Autopsy is generally not required for hospice patients who die of their terminal illness
- Cases that may require referral to the medical examiner/coroner: death within 24 hours of admission, death of unknown cause, death potentially related to injury or suspected foul play, death during or immediately after surgery
- State laws vary regarding which deaths must be reported to the medical examiner
- Autopsy can be offered to families for educational purposes or closure; should be presented sensitively
Death Certification
The death certificate is a legal document completed by the certifying physician (attending physician or hospice medical director).
- Cause of death: List the chain of events leading to death in order (immediate cause of death → due to/consequence of → underlying cause). Example: Immediate cause: respiratory failure; Due to: metastatic lung cancer; Due to: adenocarcinoma of the lung
- Manner of death: Natural (virtually always the case for hospice/palliative patients)
- Do NOT list “cardiac arrest” or “cardiopulmonary arrest” as a cause of death (this is the mechanism of all death, not the cause)
- Timeliness: Most states require completion within 24–72 hours of death
40 Performance & Prognostication Scales
Palliative Performance Scale (PPS) — All 11 Levels
| PPS % | Ambulation | Activity & Evidence of Disease | Self-Care | Intake | Conscious Level |
|---|---|---|---|---|---|
| 100% | Full | Normal activity & work; no evidence of disease | Full | Normal | Full |
| 90% | Full | Normal activity & work; some evidence of disease | Full | Normal | Full |
| 80% | Full | Normal activity with effort; some evidence of disease | Full | Normal or reduced | Full |
| 70% | Reduced | Unable normal job/work; significant disease | Full | Normal or reduced | Full |
| 60% | Reduced | Unable hobby/housework; significant disease | Occasional assistance necessary | Normal or reduced | Full or confusion |
| 50% | Mainly sit/lie | Unable to do any work; extensive disease | Considerable assistance required | Normal or reduced | Full or confusion |
| 40% | Mainly in bed | Unable to do most activity; extensive disease | Mainly assistance | Normal or reduced | Full or drowsy ± confusion |
| 30% | Totally bed bound | Unable to do any activity; extensive disease | Total care | Normal or reduced | Full or drowsy ± confusion |
| 20% | Totally bed bound | Unable to do any activity; extensive disease | Total care | Minimal to sips | Full or drowsy ± confusion |
| 10% | Totally bed bound | Unable to do any activity; extensive disease | Total care | Mouth care only | Drowsy or coma ± confusion |
| 0% | Death | ||||
ECOG Performance Status
| Grade | Description |
|---|---|
| 0 | Fully active, able to carry on all pre-disease performance without restriction |
| 1 | Restricted in physically strenuous activity but ambulatory and able to carry out work of a light or sedentary nature |
| 2 | Ambulatory and capable of all self-care but unable to carry out any work activities; up and about >50% of waking hours |
| 3 | Capable of only limited self-care; confined to bed or chair >50% of waking hours |
| 4 | Completely disabled; cannot carry on any self-care; totally confined to bed or chair |
| 5 | Dead |
Karnofsky Performance Status (KPS)
| KPS % | Description |
|---|---|
| 100 | Normal; no complaints; no evidence of disease |
| 90 | Able to carry on normal activity; minor signs/symptoms of disease |
| 80 | Normal activity with effort; some signs/symptoms of disease |
| 70 | Cares for self but unable to carry on normal activity or do active work |
| 60 | Requires occasional assistance but is able to care for most personal needs |
| 50 | Requires considerable assistance and frequent medical care |
| 40 | Disabled; requires special care and assistance |
| 30 | Severely disabled; hospitalization indicated though death not imminent |
| 20 | Very sick; hospitalization and active supportive care necessary |
| 10 | Moribund; fatal processes progressing rapidly |
| 0 | Dead |
FAST Scale (Functional Assessment Staging Tool) — Full Enumeration
| Stage | Clinical Characteristics | Approximate MMSE |
|---|---|---|
| 1 | No difficulty either subjectively or objectively | 29–30 |
| 2 | Complains of forgetting location of objects; subjective word-finding difficulty | 28–29 |
| 3 | Decreased job functioning evident to co-workers; difficulty traveling to new locations | 24–28 |
| 4 | Decreased ability to perform complex tasks (finances, planning dinner party, marketing) | 19–24 |
| 5 | Requires assistance choosing proper clothing to wear for the season or occasion | 15–19 |
| 6a | Difficulty putting on clothing properly without assistance | 10–15 |
| 6b | Unable to bathe properly (adjust water temperature) without assistance | — |
| 6c | Inability to handle mechanics of toileting (forgets to flush, does not wipe properly) | — |
| 6d | Urinary incontinence (occasional or more frequent) | — |
| 6e | Fecal incontinence (occasional or more frequent) | 0–5 |
| 7a | Ability to speak limited to approximately 6 intelligible words in the course of a day | 0 |
| 7b | Speech ability limited to a single intelligible word in the course of a day | 0 |
| 7c | Ambulatory ability lost (cannot walk without assistance) | 0 |
| 7d | Cannot sit up without assistance (falls over) | 0 |
| 7e | Loss of ability to smile | 0 |
| 7f | Loss of ability to hold head up independently | 0 |
Edmonton Symptom Assessment Scale (ESAS)
A validated 9-item patient self-report tool measuring symptom severity on a 0–10 NRS (0 = no symptom; 10 = worst possible):
| Symptom | Scale |
|---|---|
| Pain | 0 (no pain) — 10 (worst pain) |
| Tiredness/Fatigue | 0 (not tired) — 10 (worst tiredness) |
| Drowsiness | 0 (not drowsy) — 10 (worst drowsiness) |
| Nausea | 0 (not nauseated) — 10 (worst nausea) |
| Lack of appetite | 0 (best appetite) — 10 (worst appetite) |
| Shortness of breath | 0 (not short of breath) — 10 (worst shortness of breath) |
| Depression | 0 (not depressed) — 10 (worst depression) |
| Anxiety | 0 (not anxious) — 10 (worst anxiety) |
| Well-being | 0 (best well-being) — 10 (worst well-being) |
The ESAS-revised (ESAS-r) is the updated version. A 10th line is available for “other symptom” specified by the patient. The total ESAS distress score (sum of all items, 0–90) can track symptom burden over time.
Braden Scale for Predicting Pressure Sore Risk
| Subscale | 1 (Worst) | 2 | 3 | 4 (Best) |
|---|---|---|---|---|
| Sensory perception | Completely limited | Very limited | Slightly limited | No impairment |
| Moisture | Constantly moist | Very moist | Occasionally moist | Rarely moist |
| Activity | Bedfast | Chairfast | Walks occasionally | Walks frequently |
| Mobility | Completely immobile | Very limited | Slightly limited | No limitations |
| Nutrition | Very poor | Probably inadequate | Adequate | Excellent |
| Friction & shear | Problem | Potential problem | No apparent problem | — (3-point scale) |
Total score 6–23. Risk levels: ≤9 = very high risk; 10–12 = high risk; 13–14 = moderate risk; 15–18 = mild risk; 19–23 = no risk.
Palliative Prognostic Index (PPI)
| Factor | Criteria | Score |
|---|---|---|
| Palliative Performance Scale | PPS 10–20% | 4.0 |
| PPS 30–50% | 2.5 | |
| PPS ≥60% | 0 | |
| Oral intake | Mouthfuls or less | 2.5 |
| Reduced but more than mouthfuls | 1.0 | |
| Normal | 0 | |
| Edema | Present | 1.0 |
| Absent | 0 | |
| Dyspnea at rest | Present | 3.5 |
| Absent | 0 | |
| Delirium | Present | 4.0 |
| Absent | 0 |
Interpretation: PPI >6 = survival <3 weeks (sensitivity 80%, specificity 85%); PPI ≤4 = survival >6 weeks (sensitivity 80%, specificity 77%).
Palliative Prognostic (PaP) Score
| Factor | Criteria | Score |
|---|---|---|
| Dyspnea | Present | 1.0 |
| Absent | 0 | |
| Anorexia | Present | 1.5 |
| Absent | 0 | |
| KPS | ≥30 | 0 |
| 10–20 | 2.5 | |
| Clinical prediction of survival | >12 weeks | 0 |
| 11–12 weeks | 2.0 | |
| 7–10 weeks | 2.5 | |
| 5–6 weeks | 4.5 | |
| 3–4 weeks | 6.0 | |
| 1–2 weeks | 8.5 | |
| Total WBC | Normal (4,800–8,500) | 0 |
| High (8,501–11,000) | 0.5 | |
| Very high (>11,000) | 1.5 | |
| Lymphocyte % | Normal (20–40%) | 0 |
| Low (12–19.9%) | 1.0 | |
| Very low (0–11.9%) | 2.5 |
Total score 0–17.5. Risk groups: Group A (0–5.5) = 30-day survival >70%; Group B (5.6–11.0) = 30-day survival 30–70%; Group C (11.1–17.5) = 30-day survival <30%.
CAPC Prognostication Toolkit41 Opioid Equianalgesic Conversion Table
This comprehensive reference table provides equianalgesic doses, conversion ratios, and key clinical notes for all commonly used opioids in palliative care.
| Opioid | Equianalgesic PO Dose (mg) | Equianalgesic Parenteral Dose (mg) | PO:Parenteral Ratio | Duration (h) | Clinical Notes |
|---|---|---|---|---|---|
| Morphine | 30 | 10 | 3:1 | 3–4 (IR); 8–12 (SR) | Reference standard; M6G active metabolite accumulates in renal failure; histamine release |
| Hydromorphone | 6 | 1.5 | 4:1 | 3–4 | Preferred in renal impairment; no active metabolites; 5× more potent than morphine PO |
| Oxycodone | 20 | N/A | — | 3–4 (IR); 12 (CR) | 1.5× more potent than morphine PO; good oral bioavailability |
| Hydrocodone | 30 | N/A | — | 4–6 | Equivalent to morphine PO; available as combination products or extended-release |
| Codeine | 200 | 130 | 1.5:1 | 4–6 | Prodrug (CYP2D6); ceiling ~60 mg/dose; 0.15× morphine potency |
| Tramadol | 300 | N/A | — | 4–6 | Weak mu-agonist + SNRI; ceiling 400 mg/day; seizure, serotonin syndrome risk |
| Fentanyl (IV) | — | 0.1 (100 mcg) | — | 0.5–1 | 100× more potent than morphine; lipophilic; no histamine; safe in renal failure |
| Fentanyl (TD patch) | 25 mcg/h patch ≈ 60–90 mg oral morphine/24h; Conversion: daily oral morphine mg ÷ 2–3 = fentanyl mcg/h; 72-h patch duration; 12–24 h to onset, 12–24 h depot after removal | ||||
| Methadone | Variable ratio (see Section 07): <90 mg OME = 4:1; 90–300 = 8:1; 301–600 = 12:1; >600 = 15–20:1. NMDA antagonist; long t½ (15–60 h); QTc risk; expert consultation recommended | ||||
| Oxymorphone | 10 | 1 | 10:1 | 3–4 (IR); 12 (ER) | 3× more potent than morphine PO; no active metabolites |
- Morphine PO → IV: Divide by 3 (e.g., 30 mg PO = 10 mg IV)
- Morphine PO → Hydromorphone PO: Divide by 5 (e.g., 30 mg morphine PO = 6 mg hydromorphone PO)
- Morphine PO → Oxycodone PO: Divide by 1.5 (e.g., 30 mg morphine PO = 20 mg oxycodone PO)
- Morphine PO → Fentanyl patch: Total daily morphine PO (mg) ÷ 2–3 = fentanyl patch (mcg/h) (e.g., 90 mg morphine/day ≈ fentanyl 25–37 mcg/h patch)
- Always reduce by 25–50% for incomplete cross-tolerance when rotating opioids
42 Medications Master Table
Opioid Analgesics
| Drug | Route(s) | Starting Dose (opioid-naïve) | Max/Notes |
|---|---|---|---|
| Morphine IR | PO, SL, PR | 5–15 mg q4h | No ceiling; titrate to effect; reduce in renal failure |
| Morphine SR | PO | 15 mg q8–12h | Do not crush or chew; not for opioid-naïve PRN use |
| Morphine concentrated solution | SL | 5–10 mg (0.25–0.5 mL of 20 mg/mL) q2–4h | Hospice comfort kit staple; for patients unable to swallow |
| Hydromorphone (Dilaudid) | PO, IV, SC | PO: 1–2 mg q4h; IV: 0.2–0.5 mg q2–4h | Preferred in renal failure |
| Oxycodone IR | PO | 5–10 mg q4–6h | Available as single agent or combination |
| Fentanyl patch | TD | 12–25 mcg/h q72h | NOT for opioid-naïve patients; requires stable pain and opioid tolerance |
| Methadone | PO, IV, SC | 2.5–5 mg q8–12h | Titrate no faster than q5–7 days; ECG monitoring; expert prescribing recommended |
Adjuvant Analgesics
| Drug | Route | Dose Range | Indication |
|---|---|---|---|
| Gabapentin | PO | 100–1200 mg TID | Neuropathic pain, pruritus, hiccups |
| Pregabalin | PO | 25–300 mg BID | Neuropathic pain |
| Duloxetine | PO | 30–60 mg daily | Neuropathic pain, CIPN, depression |
| Dexamethasone | PO, IV, SC | 2–16 mg daily | Bone pain, brain/spinal cord edema, bowel obstruction, appetite, nausea |
| Ketamine | IV, SC, PO, IN | IV: 0.1–0.5 mg/kg/h; PO: 10–25 mg TID | Refractory pain, opioid-induced hyperalgesia |
| Lidocaine 5% patch | Topical | Up to 3 patches, 12 h on/12 h off | Localized neuropathic pain |
Antiemetics
| Drug | Route | Dose | Mechanism / Notes |
|---|---|---|---|
| Haloperidol | PO, SC, IV | 0.5–2 mg q6–8h | D2; opioid/metabolic nausea; also antipsychotic for delirium |
| Ondansetron | PO, IV, ODT | 4–8 mg q6–8h | 5-HT3; CINV, post-op; constipating |
| Metoclopramide | PO, IV, SC | 10 mg q6h (ac) | D2 + prokinetic; gastroparesis; avoid in complete obstruction |
| Olanzapine | PO, SL (ODT) | 2.5–5 mg daily | Broad-spectrum; CINV; appetite; sedation |
| Dexamethasone | PO, IV, SC | 4–8 mg daily | CINV adjunct; raised ICP; appetite |
| Scopolamine | TD patch | 1.5 mg q72h | Vestibular nausea; also for secretions |
| Prochlorperazine | PO, PR, IV | 5–10 mg q6–8h | D2; general-purpose antiemetic; EPS risk |
Laxatives
| Drug | Class | Dose | Notes |
|---|---|---|---|
| Senna | Stimulant | 8.6–34.4 mg BID | Foundation of OIC regimen; titrate to effect |
| Docusate | Stool softener | 100 mg BID | Alone insufficient for OIC; optional with senna |
| Polyethylene glycol (MiraLAX) | Osmotic | 17 g daily–BID | Well tolerated; no electrolyte shifts |
| Lactulose | Osmotic | 15–30 mL BID–QID | Also for hepatic encephalopathy; bloating/gas |
| Bisacodyl | Stimulant | 5–15 mg PO or 10 mg PR | Suppository for rectal stimulation |
| Methylnaltrexone (Relistor) | Peripheral opioid antagonist | 8–12 mg SC qod | Does NOT cross BBB; does not reverse analgesia; for OIC refractory to conventional laxatives |
| Naloxegol (Movantik) | Peripheral opioid antagonist | 25 mg PO daily | Oral option for OIC; avoid with strong CYP3A4 inhibitors |
Anxiolytics & Sedatives
| Drug | Route | Dose | Indication |
|---|---|---|---|
| Lorazepam (Ativan) | PO, SL, IV, SC | 0.5–2 mg q4–8h | Anxiety, insomnia, seizures, dyspnea adjunct; no active metabolites |
| Midazolam (Versed) | IV, SC, IN, buccal | 1–5 mg q1–2h PRN or 0.5–2 mg/h infusion | Terminal agitation, palliative sedation, seizures; short-acting |
| Clonazepam | PO | 0.25–0.5 mg BID–TID | Anxiety, myoclonus, neuropathic pain, seizures |
| Phenobarbital | IV, SC, PR | Loading: 200 mg; 0.5–2 mg/kg/h infusion | Refractory seizures, palliative sedation when benzodiazepines fail |
Anticholinergics (Secretion Management)
| Drug | Route | Dose | Notes |
|---|---|---|---|
| Glycopyrrolate (Robinul) | IV, SC, PO | 0.2–0.4 mg q4–6h SC/IV; 1 mg TID PO | Does NOT cross BBB — preferred (less delirium risk) |
| Atropine 1% ophthalmic | SL | 1–2 drops SL q4–6h | Readily available; crosses BBB (delirium risk) |
| Scopolamine (hyoscine HBr) | TD, SC | 1.5 mg patch q72h; 0.3–0.6 mg SC q4–6h | Crosses BBB; sedating (may be beneficial in agitated patients) |
| Hyoscine butylbromide (Buscopan) | SC | 20 mg q4–6h | Does NOT cross BBB; also antispasmodic for colic; not available in US |
43 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AAHPM | American Academy of Hospice and Palliative Medicine |
| ACP | Advance care planning |
| AD | Advance directive |
| AND | Allow natural death |
| BID | Twice daily |
| BTP | Breakthrough pain |
| CBT | Cognitive behavioral therapy |
| CHC | Continuous home care |
| CIPN | Chemotherapy-induced peripheral neuropathy |
| CMO | Comfort measures only |
| CMS | Centers for Medicare & Medicaid Services |
| CSCI | Continuous subcutaneous infusion |
| DCD | Donation after circulatory death |
| DNAR | Do not attempt resuscitation |
| DNI | Do not intubate |
| DNR | Do not resuscitate |
| DPOA-HC | Durable power of attorney for healthcare |
| ECOG | Eastern Cooperative Oncology Group |
| EOL | End of life |
| ESAS | Edmonton Symptom Assessment Scale |
| FAST | Functional Assessment Staging Tool |
| GIP | General inpatient care |
| GOC | Goals of care |
| HHA | Home health aide |
| ICD | Implantable cardioverter-defibrillator |
| IDDS | Intrathecal drug delivery system |
| IDT | Interdisciplinary team |
| IR | Immediate release |
| IV | Intravenous |
| KPS | Karnofsky Performance Status |
| LCD | Local coverage determination |
| LVAD | Left ventricular assist device |
| M6G | Morphine-6-glucuronide |
| MAID | Medical assistance in dying |
| MBO | Malignant bowel obstruction |
| MHB | Medicare Hospice Benefit |
| MOLST | Medical Orders for Life-Sustaining Treatment |
| NHPCO | National Hospice and Palliative Care Organization |
| NIV | Non-invasive ventilation |
| NMDA | N-methyl-D-aspartate |
| NRS | Numeric rating scale |
| OIC | Opioid-induced constipation |
| OME | Oral morphine equivalent |
| OPO | Organ procurement organization |
| PAINAD | Pain Assessment in Advanced Dementia |
| PaP | Palliative Prognostic Score |
| PDE | Principle of double effect |
| POLST | Physician Orders for Life-Sustaining Treatment |
| PPI | Palliative Prognostic Index |
| PPS | Palliative Performance Scale |
| PRN | As needed (pro re nata) |
| QOL | Quality of life |
| RHC | Routine home care |
| SC | Subcutaneous |
| SL | Sublingual |
| SR / CR / ER | Sustained/controlled/extended release |
| SVC | Superior vena cava |
| SW | Social worker |
| TD | Transdermal |
| TID | Three times daily |
| VSED | Voluntarily stopping eating and drinking |
| WHO | World Health Organization |