Pediatrics
Every age-specific diagnosis, milestone, vaccination, weight-based dosing, neonatal condition, and management strategy from birth through adolescence in one place.
01 Pediatric Anatomy & Physiology
Children are not small adults. Virtually every organ system differs from adult physiology in ways that have direct clinical consequences — from airway anatomy that predisposes to obstruction, to immature renal function that alters drug clearance, to a higher metabolic rate that demands proportionally greater caloric and fluid intake. Understanding these age-specific differences is the foundation of all pediatric care.
Airway & Respiratory Differences
The pediatric airway is narrower, more anterior, and more cephalad than the adult airway. The infant larynx sits at C3–C4 (versus C4–C5 in adults). The epiglottis is omega-shaped and floppy. The narrowest point is the cricoid ring (subglottic region) in children <8 years, unlike adults where the glottis is narrowest. Infants are obligate nasal breathers until approximately 4–6 months of age — nasal obstruction (e.g., choanal atresia, mucus from URI) can cause significant respiratory distress. The chest wall is highly compliant in neonates, leading to paradoxical breathing with subcostal and intercostal retractions as early signs of respiratory distress.
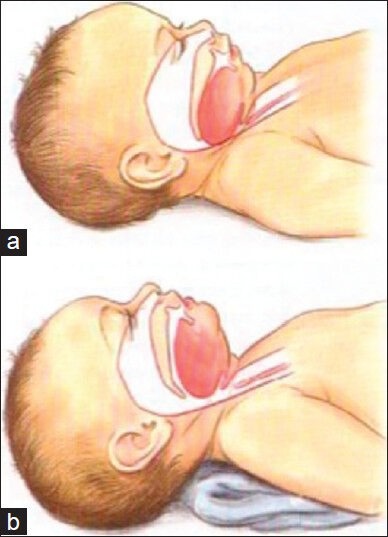
Cardiovascular Differences
The neonatal myocardium has fewer contractile elements and is less compliant than the adult heart. Cardiac output in infants is heart-rate dependent because stroke volume is relatively fixed — bradycardia is poorly tolerated and is the most common preterminal rhythm in children (unlike adults, where VF/VT predominates). Normal heart rates are much higher in infants (120–160 bpm in neonates) and decrease with age. Blood pressure is lower in young children; hypotension is a late sign of shock in pediatrics because children compensate with tachycardia and increased SVR until they decompensate precipitously.
Fluid & Caloric Requirements
Children have a higher body surface area-to-mass ratio and higher metabolic rate per kilogram than adults. Maintenance fluid is calculated using the Holliday-Segar formula:
| Weight | Fluid Rate | Caloric Need |
|---|---|---|
| First 10 kg | 100 mL/kg/day (4 mL/kg/hr) | 100 kcal/kg/day |
| Next 10 kg (11–20 kg) | 50 mL/kg/day (2 mL/kg/hr) | 50 kcal/kg/day |
| Each kg >20 kg | 20 mL/kg/day (1 mL/kg/hr) | 20 kcal/kg/day |
Thermoregulation
Neonates, especially preterm infants, are highly susceptible to hypothermia due to large surface area-to-volume ratio, thin subcutaneous fat, and inability to shiver (neonates generate heat via non-shivering thermogenesis using brown fat). Hypothermia increases oxygen consumption, causes metabolic acidosis, and worsens respiratory distress. Maintaining a neutral thermal environment (36.5–37.5°C axillary) is critical in neonatal care.
Renal & Hepatic Maturation
The GFR at birth is ~20 mL/min/1.73 m² in term neonates and even lower in preterm infants; it does not reach adult values (~120 mL/min/1.73 m²) until 1–2 years of age. This affects drug clearance for renally eliminated medications (aminoglycosides, vancomycin). Hepatic enzyme systems (particularly glucuronidation via UGT) are immature at birth — this explains physiologic jaundice and the risk of gray baby syndrome with chloramphenicol.
Age-Specific Vital Signs
| Age | Heart Rate (bpm) | Respiratory Rate | Systolic BP (mmHg) |
|---|---|---|---|
| Newborn | 120–160 | 30–60 | 60–76 |
| Infant (1–12 mo) | 100–150 | 25–50 | 72–104 |
| Toddler (1–3 yr) | 90–140 | 20–30 | 86–106 |
| Preschool (4–5 yr) | 80–120 | 20–25 | 89–112 |
| School age (6–12 yr) | 70–110 | 18–22 | 97–120 |
| Adolescent (13–18 yr) | 60–100 | 12–20 | 110–131 |
02 The Pediatric Assessment
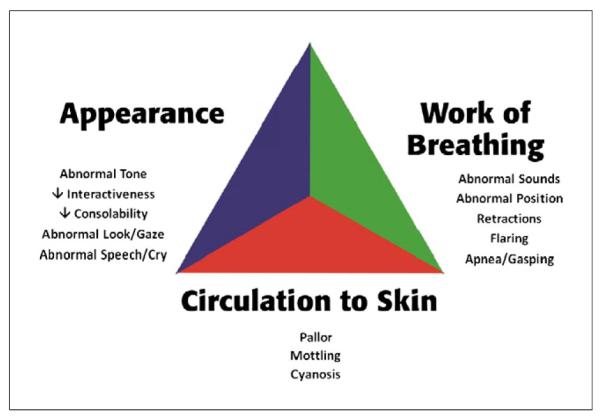
The pediatric assessment begins the moment you enter the room. The Pediatric Assessment Triangle (PAT) provides a rapid (<30 second) visual assessment of severity before any hands-on examination. It evaluates three components: Appearance (TICLS: Tone, Interactiveness, Consolability, Look/Gaze, Speech/Cry), Work of Breathing (retractions, nasal flaring, grunting, positioning), and Circulation to Skin (pallor, mottling, cyanosis).
ABCDE Approach
| Component | Pediatric-Specific Considerations |
|---|---|
| A — Airway | Head in sniffing position (not hyperextended); large occiput in infants may require shoulder roll; suction secretions; consider jaw thrust in trauma |
| B — Breathing | Count respiratory rate before disturbing child; look for retractions, nasal flaring, grunting, head bobbing; auscultate all fields |
| C — Circulation | Assess cap refill (<2 sec normal), pulses (brachial in infants, radial/femoral in older children), skin temperature/mottling; HR is the primary compensatory mechanism |
| D — Disability | AVPU scale (Alert, Voice, Pain, Unresponsive) or GCS; pupil reactivity; glucose check (neonates/infants prone to hypoglycemia) |
| E — Exposure | Full undressing with temperature; look for rashes, bruising patterns (NAT); keep warm — rapid heat loss in small children |
Growth Charts & Percentiles
Growth monitoring is the cornerstone of well-child care. The WHO growth standards are used for children 0–2 years (based on breastfed children), and CDC growth charts for children 2–20 years. Parameters tracked include weight-for-age, length/height-for-age, head circumference (until age 2–3), and BMI-for-age (starting at age 2). A child consistently tracking along a percentile is reassuring; crossing two or more major percentile lines (either up or down) warrants investigation.
Developmental Screening Tools
| Tool | Ages | What It Screens |
|---|---|---|
| ASQ-3 (Ages & Stages Questionnaire) | 1–66 months | Communication, gross motor, fine motor, problem solving, personal-social |
| M-CHAT-R/F | 16–30 months | Autism spectrum disorder |
| PEDS (Parents’ Evaluation of Developmental Status) | 0–8 years | General developmental concerns |
| Edinburgh Postnatal Depression Scale | Postpartum | Maternal depression (screened at well-child visits) |
| PHQ-A / PHQ-9 Modified for Adolescents | ≥12 years | Adolescent depression |
03 Key Terminology & Abbreviations
Pediatrics uses age-specific terminology and abbreviations that differ from adult medicine in important ways. Mastering this vocabulary is essential for interpreting growth data, immunization records, neonatal documentation, and pediatric emergency protocols.
| Abbreviation | Meaning |
|---|---|
| AGA / SGA / LGA | Appropriate / Small / Large for gestational age |
| APGAR | Appearance, Pulse, Grimace, Activity, Respiration (newborn scoring) |
| NEC | Necrotizing enterocolitis |
| RDS | Respiratory distress syndrome (surfactant deficiency) |
| TTN | Transient tachypnea of the newborn |
| GBS | Group B Streptococcus |
| NICU | Neonatal intensive care unit |
| VLBW / ELBW | Very low birth weight (<1,500 g) / Extremely low (<1,000 g) |
| GA | Gestational age |
| PMA | Postmenstrual age |
| BPD | Bronchopulmonary dysplasia |
| IVH | Intraventricular hemorrhage |
| PDA | Patent ductus arteriosus |
| VSD / ASD | Ventricular / Atrial septal defect |
| TOF | Tetralogy of Fallot |
| TGA | Transposition of the great arteries |
| RSV | Respiratory syncytial virus |
| PALS | Pediatric Advanced Life Support |
| IO | Intraosseous (vascular access) |
| FTT | Failure to thrive |
| NAT | Non-accidental trauma |
| VCUG | Voiding cystourethrogram |
| VUR | Vesicoureteral reflux |
| DKA | Diabetic ketoacidosis |
| ADHD | Attention-deficit/hyperactivity disorder |
| ASD (neuro) | Autism spectrum disorder |
04 Developmental Milestones
Developmental milestones represent the expected acquisition of skills across four domains: gross motor, fine motor, language, and social-emotional. While there is normal variation, consistent delay in reaching milestones may indicate developmental disability, neurologic disease, or environmental deprivation. The CDC/AAP updated milestone checklists in 2022 to reflect when most children (75th percentile) achieve a skill, rather than the 50th percentile used previously.
Milestones by Age
| Age | Gross Motor | Fine Motor | Language | Social |
|---|---|---|---|---|
| 2 months | Lifts head prone | Hands unfisted 50% | Coos, vowel sounds | Social smile |
| 4 months | Holds head steady upright; pushes up on elbows | Reaches for objects | Laughs, squeals | Smiles spontaneously |
| 6 months | Rolls both ways; sits with support | Transfers objects hand to hand; raking grasp | Babbles (consonant sounds) | Stranger anxiety begins |
| 9 months | Sits unsupported; pulls to stand | Pincer grasp (immature) | "Mama/dada" nonspecific | Object permanence; separation anxiety |
| 12 months | Walks with one hand held; cruises | Mature pincer grasp; releases objects | 1–3 words with meaning | Waves bye-bye; plays pat-a-cake |
| 15 months | Walks independently | Stacks 2 blocks; scribbles | 4–6 words | Points to desires; imitates activities |
| 18 months | Runs; kicks ball | Stacks 3–4 blocks; feeds self with spoon | 10–25 words; points to body parts | Parallel play; temper tantrums |
| 2 years | Walks up/down stairs (2 feet per step); jumps | Stacks 6 blocks; turns pages one at a time | 2-word phrases; 50+ words; 50% intelligible | Parallel play; follows 2-step commands |
| 3 years | Pedals tricycle; climbs stairs alternating feet | Copies circle; uses scissors | 3-word sentences; 75% intelligible; knows name/age/sex | Group play; shares; knows gender |
| 4 years | Hops on one foot; throws overhand | Copies cross (+); draws person with 4 parts | Tells stories; 100% intelligible; counts to 4 | Cooperative play; imaginary friends |
| 5 years | Skips; balances on one foot 10 sec | Copies triangle; ties shoes | Speaks in full sentences; names colors | Understands rules; plays organized games |
Primitive Reflexes
| Reflex | How to Elicit | Present At | Disappears By |
|---|---|---|---|
| Moro (startle) | Sudden head drop or loud noise | Birth | 4–6 months |
| Rooting | Stroke cheek → turns toward stimulus | Birth | 3–4 months |
| Palmar grasp | Place finger in palm | Birth | 4–6 months |
| Babinski | Stroke lateral sole → toe dorsiflexion & fanning | Birth | 12–24 months |
| Tonic neck (fencing) | Turn head to side → ipsilateral arm extends | Birth | 4–6 months |
| Stepping | Hold upright, feet touch surface | Birth | 2 months |
| Parachute | Suspend prone, lower toward surface → arms extend | 8–9 months | Persists for life |
05 Immunization Schedule
Vaccination is the single most effective preventive intervention in pediatrics. The CDC immunization schedule is updated annually and published by the Advisory Committee on Immunization Practices (ACIP). Understanding the schedule, minimum intervals, contraindications, and the distinction between live and inactivated vaccines is essential for every pediatric provider.
Routine Childhood Immunization Schedule (Birth–18 Years)
| Vaccine | Type | Schedule | Key Notes |
|---|---|---|---|
| Hepatitis B (HepB) | Inactivated | Birth, 1 mo, 6 mo | First dose within 24 hours of birth; monovalent only at birth |
| Rotavirus (RV) | Live oral | 2, 4 mo (RV1) or 2, 4, 6 mo (RV5) | Must start by 15 weeks; cannot give after 8 months 0 days |
| DTaP | Inactivated | 2, 4, 6, 15–18 mo, 4–6 yr | 5-dose series; Tdap booster at age 11–12 |
| Hib | Inactivated | 2, 4, (6), 12–15 mo | Number of doses depends on product; PRP-OMP (PedvaxHIB) needs no 6-month dose |
| IPV (Polio) | Inactivated | 2, 4, 6–18 mo, 4–6 yr | 4-dose series; OPV no longer used in U.S. |
| PCV15 or PCV20 | Inactivated | 2, 4, 6, 12–15 mo | PCV15 followed by PPSV23 at high risk; PCV20 alone is alternative |
| MMR | Live | 12–15 mo, 4–6 yr | CI in severe immunodeficiency and pregnancy |
| Varicella | Live | 12–15 mo, 4–6 yr | CI in immunocompromised; avoid aspirin for 6 weeks after |
| Hepatitis A | Inactivated | 12–23 mo (2 doses, 6 mo apart) | 2-dose series starting at age 1 |
| HPV | Inactivated | 11–12 yr (2 or 3 doses) | 2 doses if started before age 15; 3 doses if ≥15 yr |
| Meningococcal ACWY | Inactivated | 11–12 yr, booster 16 yr | High-risk groups start earlier |
| Meningococcal B | Inactivated | 16–23 yr (shared decision) | Recommended for asplenia, complement deficiency |
| Influenza | Inactivated or Live (LAIV) | Annually starting 6 mo | 2 doses first year if <9 years and first-time; LAIV not for <2 yr or immunocompromised |
Live vs. Inactivated Vaccines
MMR, Varicella, Rotavirus, LAIV (intranasal flu), BCG, Oral Polio (OPV), Yellow Fever. Live vaccines are contraindicated in severe immunodeficiency (e.g., SCID, high-dose steroids ≥2 mg/kg/day for ≥14 days, chemotherapy). If two live vaccines are not given on the same day, they must be separated by ≥28 days. Live vaccines can cause mild illness (vaccine-strain disease) and can rarely revert to virulence.
06 Growth & Nutrition
Expected Growth Patterns
| Age | Weight Gain | Length/Height Gain | Head Circumference |
|---|---|---|---|
| 0–3 months | 30 g/day (1 oz/day) | 3.5 cm/month | 2 cm/month |
| 3–6 months | 20 g/day | 2 cm/month | 1 cm/month |
| 6–12 months | 10 g/day | 1.5 cm/month | 0.5 cm/month |
| 1–2 years | ~2.5 kg/year | 12 cm/year | — |
| 2–puberty | ~2 kg/year | 5–7 cm/year | — |
Breastfeeding
Breast milk is the recommended sole source of nutrition for the first 6 months. It provides optimal nutrition, immunoglobulins (especially secretory IgA), and growth factors. The AAP recommends continued breastfeeding with complementary foods through at least 12 months. Breast milk is deficient in vitamin D (supplement 400 IU/day from birth) and iron after 4–6 months (supplement 1 mg/kg/day or introduce iron-rich foods).
Formula Feeding
Standard cow’s milk-based formula (20 kcal/oz) is the alternative when breastfeeding is not possible. Soy-based formula is for galactosemia or cow’s milk protein intolerance (but 10–14% cross-react). Extensively hydrolyzed (e.g., Alimentum, Nutramigen) or amino acid-based (EleCare) formulas are for true cow’s milk protein allergy. Whole cow’s milk should not be introduced before 12 months (low iron, excess protein/sodium, risk of GI blood loss).
Introduction of Solid Foods
Begin complementary foods at approximately 4–6 months when the infant shows developmental readiness (sits with support, good head control, loss of tongue thrust reflex). Introduce one new food every 3–5 days. Early introduction of allergenic foods (peanut, egg, milk) between 4–6 months is now recommended by the AAP and LEAP study data to reduce allergy risk, especially in high-risk infants (eczema, egg allergy).
Failure to Thrive (FTT)
Defined as weight <3rd percentile for age, weight-for-length <5th percentile, or crossing down 2 or more major percentile lines. Causes are classified as inadequate intake (most common — poverty, neglect, feeding difficulties, cleft palate), inadequate absorption (celiac, CF, cow’s milk protein allergy), or increased metabolic demand (CHD, chronic infection, hyperthyroidism). Workup includes a detailed feeding history, calorie count, CBC, CMP, urinalysis, and celiac screening. In FTT, weight is affected first, then length, then head circumference (this pattern distinguishes nutritional FTT from genetic/endocrine short stature).
Childhood Obesity
| Category | BMI Percentile (age 2–20) |
|---|---|
| Underweight | <5th percentile |
| Normal weight | 5th–84th percentile |
| Overweight | 85th–94th percentile |
| Obese | ≥95th percentile |
| Severe obesity | ≥120% of 95th percentile or BMI ≥35 |
07 Anticipatory Guidance & Screening
Preventive care in pediatrics involves both surveillance for disease and anticipatory guidance for safety, nutrition, and development at each well-child visit.
AAP Recommended Screening Schedule
| Screening | Ages | Method |
|---|---|---|
| Newborn metabolic screen | 24–48 hours of life | Heel-stick blood spot (PKU, hypothyroidism, sickle cell, CAH, CF, galactosemia, and ~30 others) |
| Hearing | Before hospital discharge; repeat 1, 3, 5 yr (risk-based) | OAE or ABR |
| Vision | Newborn (red reflex); 3–5 yr (visual acuity); annually school-age | Red reflex, Snellen chart, instrument-based screening |
| Lead | 12 and 24 months (universal in high-risk areas) | Venous lead level; action level ≥3.5 µg/dL (CDC 2021) |
| Iron deficiency | 12 months | CBC or hemoglobin; risk-based at other ages |
| Autism (M-CHAT-R/F) | 18 and 24 months | Parent questionnaire; follow-up interview if positive |
| Dyslipidemia | 9–11 years (universal); 2+ yr if family history | Fasting lipid panel or non-fasting non-HDL cholesterol |
| Depression (PHQ-A) | ≥12 years annually | Patient Health Questionnaire for Adolescents |
| STI screening | Sexually active adolescents | Chlamydia, gonorrhea, HIV; per USPSTF |
Anticipatory Guidance Highlights by Age
| Age | Key Topics |
|---|---|
| Newborn | Safe sleep (back to sleep, no co-sleeping, firm surface), skin-to-skin, breastfeeding support, car seat safety |
| 2–6 months | Tummy time, fall prevention, choking hazards, no honey before 1 year (botulism) |
| 6–12 months | Childproofing, poison control number, introduction of solids, dental care (first tooth/first birthday) |
| 1–4 years | Water safety/drowning prevention, car seat transitions (rear-facing until age 2+), limit screen time |
| 5–12 years | Bicycle helmets, seatbelt use, stranger safety, screen time limits, bullying |
| Adolescent | Substance use, safe sex, mental health, driving safety, sleep hygiene, social media |
08 Newborn Assessment
APGAR Score
Assessed at 1 and 5 minutes after birth (and every 5 minutes thereafter if score <7). It is a rapid assessment of the newborn’s transition to extrauterine life, not a predictor of long-term neurologic outcome.
| Sign | 0 Points | 1 Point | 2 Points |
|---|---|---|---|
| Appearance (color) | Blue/pale all over | Pink body, blue extremities (acrocyanosis) | Completely pink |
| Pulse (heart rate) | Absent | <100 bpm | ≥100 bpm |
| Grimace (reflex irritability) | No response | Grimace/weak cry | Cough, sneeze, vigorous cry |
| Activity (muscle tone) | Limp | Some flexion | Active motion, well-flexed |
| Respiration | Absent | Slow/irregular, weak cry | Good effort, strong cry |
Gestational Age Assessment
The New Ballard Score uses neuromuscular maturity (6 items: posture, square window, arm recoil, popliteal angle, scarf sign, heel-to-ear) and physical maturity (6 items: skin, lanugo, plantar surface, breast, eye/ear, genitals) to estimate GA from 20–44 weeks. Each item is scored −1 to +5; the sum maps to GA.
Common Birth Injuries
| Injury | Description | Prognosis |
|---|---|---|
| Caput succedaneum | Diffuse scalp edema crossing suture lines | Resolves spontaneously in days |
| Cephalohematoma | Subperiosteal hemorrhage; does NOT cross suture lines | Resolves in weeks–months; risk of jaundice |
| Subgaleal hemorrhage | Bleeding between periosteum and galea aponeurotica; can cross suture lines and expand rapidly | Life-threatening; can hold entire blood volume; emergent management |
| Erb palsy (C5–C6) | Upper trunk brachial plexus; "waiter’s tip" position | 80–90% recover by 3–6 months |
| Klumpke palsy (C8–T1) | Lower trunk brachial plexus; "claw hand" | Poorer prognosis than Erb |
| Clavicle fracture | Most common fracture during birth; asymmetric Moro | Heals spontaneously; palpable callus at 7–10 days |
A rapidly expanding, fluctuant scalp mass that crosses suture lines in a newborn (often after vacuum-assisted delivery) is a subgaleal hemorrhage until proven otherwise. The subgaleal space can hold 260 mL of blood — nearly the entire blood volume of a neonate. Manage with immediate volume resuscitation, serial hemoglobin/hematocrit, and blood product transfusion as needed.
09 Neonatal Jaundice
Jaundice (hyperbilirubinemia) affects ~60% of term and ~80% of preterm newborns. Most is benign physiologic jaundice, but severe unconjugated hyperbilirubinemia can cause kernicterus (bilirubin encephalopathy) — irreversible brain damage to the basal ganglia, hippocampus, and cranial nerve nuclei, presenting as choreoathetoid cerebral palsy, sensorineural hearing loss, and upward gaze palsy.
Physiologic vs. Pathologic Jaundice
| Feature | Physiologic | Pathologic |
|---|---|---|
| Onset | After 24 hours of life | Within first 24 hours |
| Peak | Day 3–5 (term); Day 5–7 (preterm) | Rises rapidly (>5 mg/dL/day) |
| Duration | Resolves by 1–2 weeks | Persists >2 weeks (term) or >3 weeks (preterm) |
| Level | Usually <12–15 mg/dL | >17–20 mg/dL (or threshold for age/risk on Bhutani nomogram) |
| Type | Unconjugated (indirect) | Unconjugated or conjugated (>2 mg/dL or >20% of total = always pathologic) |
Breastfeeding vs. Breast Milk Jaundice
| Feature | Breastfeeding Jaundice | Breast Milk Jaundice |
|---|---|---|
| Timing | First week (days 2–5) | After first week (peaks 2–3 weeks) |
| Cause | Inadequate intake → decreased stooling → increased enterohepatic circulation | Substance in breast milk (beta-glucuronidase) that increases bilirubin reabsorption |
| Management | Increase breastfeeding frequency (8–12 times/day); lactation support | Continue breastfeeding; resolves by 12 weeks; rarely exceeds phototherapy threshold |
Management
Use the Bhutani nomogram (hour-specific total serum bilirubin plotted against age in hours) to determine risk zone and guide management. Treatment includes phototherapy (blue-green light, 430–490 nm, converts unconjugated bilirubin to water-soluble photoisomers excreted without conjugation) and, in severe cases, exchange transfusion (double-volume exchange, ~160 mL/kg, to rapidly lower bilirubin and remove antibodies in hemolytic disease).
Consider exchange transfusion when total serum bilirubin rises to exchange level on AAP nomogram (typically >25 mg/dL in term healthy newborns, lower thresholds with risk factors), when intensive phototherapy fails to reduce bilirubin by 1–2 mg/dL within 4–6 hours, or when signs of acute bilirubin encephalopathy are present (hypertonia, retrocollis, opisthotonos, fever, high-pitched cry).
10 Neonatal Respiratory Distress
Respiratory distress is the most common reason for NICU admission. The differential is guided by gestational age, mode of delivery, and timing of symptom onset.
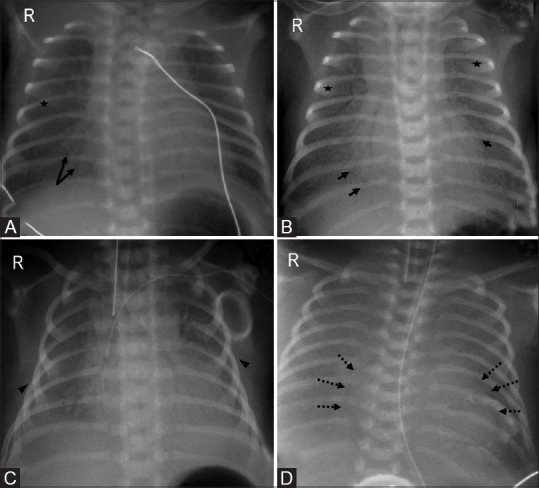
Causes of Neonatal Respiratory Distress
| Condition | Pathophysiology | Risk Factors | CXR Findings | Treatment |
|---|---|---|---|---|
| RDS (HMD) | Surfactant deficiency → alveolar collapse | Prematurity (<34 wk), maternal diabetes, C-section without labor | Diffuse bilateral ground-glass opacities, air bronchograms, low lung volumes | Surfactant replacement (via ETT or LISA); CPAP; supportive |
| TTN | Delayed resorption of fetal lung fluid | C-section without labor (no catecholamine surge), term/near-term | Perihilar streaky opacities, fluid in fissures, hyperinflation | Supportive (O2, CPAP); resolves 24–72 hours |
| MAS | Meconium aspiration → chemical pneumonitis, airway obstruction, surfactant inactivation | Post-term, fetal distress, meconium-stained amniotic fluid | Patchy bilateral infiltrates, hyperinflation, possible pneumothorax | Suction if not vigorous; surfactant; mechanical ventilation; iNO if PPHN |
| Pneumothorax | Air leak into pleural space | MAS, RDS, positive pressure ventilation, spontaneous | Hyperlucent hemithorax, mediastinal shift (tension) | Needle decompression (tension); chest tube; observation if small |
| PPHN | Failure of normal fall in pulmonary vascular resistance → right-to-left shunting via PDA/foramen ovale | MAS, RDS, sepsis, CDH, idiopathic | May be clear or show underlying cause; differential cyanosis (pre- vs post-ductal SpO2 >5% difference) | iNO (first-line pulmonary vasodilator); ECMO if refractory; treat underlying cause |
Grunting (auto-PEEP to maintain FRC), nasal flaring, intercostal/subcostal retractions, tachypnea (>60/min), and cyanosis are the cardinal signs. Grunting in a premature neonate suggests RDS until proven otherwise. A term baby with respiratory distress immediately after C-section is TTN until proven otherwise. Progressive respiratory failure with differential cyanosis suggests PPHN.
11 Neonatal Sepsis
Neonatal sepsis is a clinical syndrome of systemic infection in the first 28 days of life. It is the leading infectious cause of neonatal mortality. The immature neonatal immune system (poor opsonization, low complement, deficient neutrophil function) makes neonates uniquely susceptible to overwhelming bacterial infection.
Early-Onset vs. Late-Onset Sepsis
| Feature | Early-Onset (≤72 hours) | Late-Onset (>72 hours–28 days) |
|---|---|---|
| Timing | Birth to 72 hours (most within 24 hr) | 3–28 days |
| Source | Vertical transmission (maternal genital tract) | Horizontal/nosocomial or community |
| Common organisms | Group B Strep (GBS), E. coli (most common in VLBW), Listeria | Coagulase-negative Staphylococci (CoNS), S. aureus, GBS, gram-negatives, Candida (in VLBW) |
| Risk factors | Maternal GBS colonization, PROM >18 hr, chorioamnionitis, prematurity | Prematurity, central lines, prolonged hospitalization, TPN |
| Empiric antibiotics | Ampicillin + Gentamicin | Vancomycin + Gentamicin (or cefepime based on local resistance) |
Neonates with sepsis may present with nonspecific signs: temperature instability (hypothermia more common than fever in neonates), lethargy, poor feeding, apnea, respiratory distress, tachycardia or bradycardia, jaundice, abdominal distension. A "well-appearing" neonate can deteriorate within hours. When sepsis is suspected: obtain blood culture, CBC with differential, CRP/procalcitonin, urinalysis and urine culture (late-onset), and LP if clinically indicated. Start empiric antibiotics within 1 hour of suspicion — do not delay for LP.
GBS Prevention
Universal screening with vaginal-rectal culture at 36–37 weeks gestation. Intrapartum antibiotic prophylaxis (IAP) is indicated for: GBS-positive culture, GBS bacteriuria during current pregnancy, previous infant with GBS disease, or unknown GBS status with risk factors (preterm <37 wk, ROM ≥18 hr, intrapartum fever ≥38.0°C). First-line IAP: Penicillin G 5 million units IV then 2.5–3 million units q4h until delivery. Penicillin allergy: cefazolin if low risk; clindamycin or vancomycin if high-risk allergy.
12 Congenital Anomalies
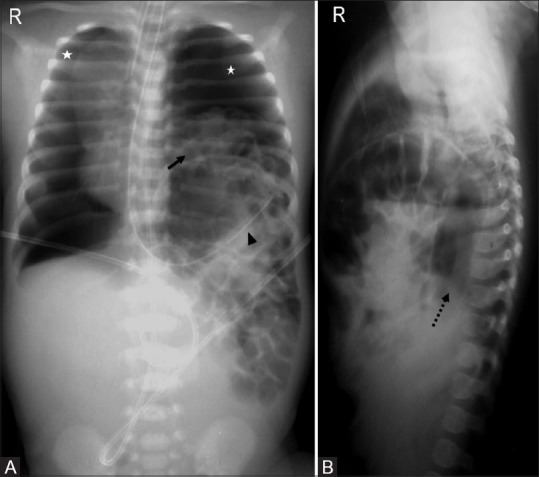
TORCH Infections
| Infection | Key Congenital Findings | Diagnosis |
|---|---|---|
| Toxoplasmosis | Intracranial calcifications (diffuse/scattered), hydrocephalus, chorioretinitis, hepatosplenomegaly | Toxoplasma IgM/IgA; PCR |
| Other (Syphilis) | Snuffles (rhinitis), rash, osteochondritis, Hutchinson teeth (late), saddle nose, saber shins | RPR/VDRL, FTA-ABS |
| Rubella | Cataracts, sensorineural deafness, PDA (most common cardiac lesion), "blueberry muffin" rash (extramedullary hematopoiesis) | Rubella IgM; viral culture |
| CMV | Periventricular calcifications, microcephaly, sensorineural hearing loss, petechiae/purpura, hepatosplenomegaly — most common congenital infection | Urine CMV PCR or culture within first 3 weeks of life |
| HSV | Skin vesicles, encephalitis, disseminated disease with liver failure, seizures; typically acquired during delivery | HSV PCR of vesicle/CSF; viral culture |
Newborn Metabolic Screening
All U.S. states screen for a core panel of conditions via heel-stick blood spot at 24–48 hours of life (and often a second screen at 1–2 weeks). The Recommended Uniform Screening Panel (RUSP) includes >35 core conditions:
| Category | Key Conditions |
|---|---|
| Amino acid disorders | PKU, maple syrup urine disease, homocystinuria, tyrosinemia |
| Fatty acid oxidation disorders | MCAD, VLCAD, LCHAD |
| Organic acid disorders | Methylmalonic acidemia, propionic acidemia, isovaleric acidemia |
| Endocrine | Congenital hypothyroidism, congenital adrenal hyperplasia (21-hydroxylase deficiency) |
| Hemoglobinopathies | Sickle cell disease (SS, SC, S-beta thal), other hemoglobinopathies |
| Other | Cystic fibrosis (IRT → sweat chloride), galactosemia, biotinidase deficiency, SCID (TREC assay) |
Congenital Heart Disease Screening
Pulse oximetry screening is performed on all newborns after 24 hours of life (or before discharge if earlier). SpO2 is measured on the right hand (preductal) and either foot (postductal). The screen fails if any reading is <90%, if both readings are <95% on three measurements separated by 1 hour, or if there is >3% difference between right hand and foot. A failed screen requires echocardiography.
13 Asthma in Children
Asthma is the most common chronic disease of childhood, affecting ~8% of U.S. children. It is characterized by reversible airway obstruction, bronchial hyperresponsiveness, and chronic airway inflammation. The NAEPP EPR-4 (2020) guidelines provide stepwise therapy recommendations.
Classification of Asthma Severity (Ages ≥12, similar for younger with modifications)
| Severity | Symptoms | Nighttime Awakenings | SABA Use | FEV1 |
|---|---|---|---|---|
| Intermittent | ≤2 days/week | ≤2×/month | ≤2 days/week | >80% |
| Mild Persistent | >2 days/week (not daily) | 3–4×/month | >2 days/week (not daily) | ≥80% |
| Moderate Persistent | Daily | >1×/week | Daily | 60–80% |
| Severe Persistent | Throughout the day | Often nightly | Several times/day | <60% |
Stepwise Therapy (Children 5–11 Years)
| Step | Preferred Controller | Alternative |
|---|---|---|
| Step 1 | SABA PRN (no daily controller) | — |
| Step 2 | Low-dose ICS daily | LTRA (montelukast) or cromolyn |
| Step 3 | Low-dose ICS + LABA, or medium-dose ICS | Low-dose ICS + LTRA |
| Step 4 | Medium-dose ICS + LABA | Medium-dose ICS + LTRA |
| Step 5 | High-dose ICS + LABA | Consider biologics (omalizumab, mepolizumab, dupilumab) |
| Step 6 | High-dose ICS + LABA + oral corticosteroids | Biologics; refer to specialist |
Acute Asthma Exacerbation Management
| Severity | Treatment |
|---|---|
| Mild-Moderate | Albuterol MDI 4–8 puffs (or nebulizer 2.5–5 mg) q20min ×3; oral prednisolone 1–2 mg/kg (max 60 mg) |
| Severe | Continuous albuterol nebulization; ipratropium 0.5 mg q20min ×3; IV/IM methylprednisolone 2 mg/kg; magnesium sulfate 25–75 mg/kg IV (max 2 g) |
| Impending respiratory failure | IV terbutaline; consider ketamine for intubation (bronchodilator properties); avoid histamine-releasing agents |
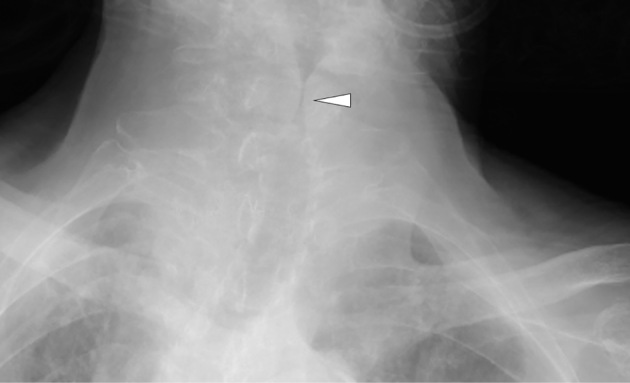
14 Croup & Epiglottitis
Croup (Laryngotracheobronchitis)
Croup is the most common cause of acute upper airway obstruction in children ages 6 months to 3 years (peak 1–2 years). It is caused by parainfluenza virus (types 1 and 3 most common) and produces subglottic edema with a characteristic "barking" or "seal-like" cough, inspiratory stridor, and hoarse voice. Symptoms are typically worse at night.
| Severity | Findings | Treatment |
|---|---|---|
| Mild | Occasional barky cough; no stridor at rest; no retractions | Single dose dexamethasone 0.6 mg/kg PO/IM (max 10 mg); cool mist; reassurance |
| Moderate | Frequent barky cough; stridor at rest; mild retractions; no agitation | Dexamethasone 0.6 mg/kg + racemic epinephrine 0.5 mL of 2.25% solution nebulized; observe ≥2–4 hours after epinephrine |
| Severe | Stridor at rest; significant retractions; agitation or lethargy; cyanosis | Dexamethasone + racemic epinephrine (may repeat); minimize agitation; prepare for airway management |
Epiglottitis
Epiglottitis is a life-threatening infection of the epiglottis and supraglottic structures, now rare due to Hib vaccination but still seen (now more often caused by Streptococcus spp. and Staphylococcus spp.). Classic presentation: acute onset of high fever, toxic-appearing, "tripod" or sniffing position, drooling, muffled "hot potato" voice, dysphagia, and minimal cough. Do NOT examine the throat, do NOT lay the child supine, do NOT agitate the child — any of these can precipitate complete airway obstruction. Lateral neck X-ray shows the "thumbprint sign" (enlarged epiglottis). Management: controlled intubation in the OR with a surgeon ready for tracheostomy; IV antibiotics (ceftriaxone or cefotaxime + vancomycin).
Differentiating Croup from Epiglottitis
| Feature | Croup | Epiglottitis |
|---|---|---|
| Age | 6 months–3 years | 2–7 years (now any age) |
| Onset | Gradual (1–2 days URI prodrome) | Rapid (hours) |
| Fever | Low-grade | High (>39°C) |
| Cough | Barking/seal-like | Absent or minimal |
| Voice | Hoarse | Muffled/"hot potato" |
| Drooling | Absent | Present |
| Position | Any | Tripod/sniffing |
| X-ray | Steeple sign | Thumbprint sign |
| Etiology | Parainfluenza | H. influenzae type b (historically); Strep/Staph now |
15 Bronchiolitis
Bronchiolitis is the most common lower respiratory tract infection in infants <12 months (peak 2–6 months). RSV causes ~70% of cases (other causes: rhinovirus, human metapneumovirus, parainfluenza, adenovirus). It produces inflammation, edema, and mucus plugging of the small airways (bronchioles), causing wheezing, crackles, and respiratory distress.
AAP Clinical Practice Guideline (2014, reaffirmed 2023)
| Recommendation | Details |
|---|---|
| Diagnosis | Clinical diagnosis based on history and physical exam; routine CXR and viral testing are NOT recommended |
| Treatment | Supportive care only: nasal suctioning, supplemental O2 if SpO2 persistently <90%, adequate hydration (oral or IV/NG) |
| Bronchodilators | Albuterol and epinephrine should NOT be routinely used; one trial of bronchodilator is acceptable but should be discontinued if no improvement |
| Corticosteroids | NOT recommended for bronchiolitis |
| Antibiotics | NOT indicated unless bacterial co-infection documented |
| Hypertonic saline (3%) | May be considered for hospitalized patients (not in ED); modest benefit in reducing length of stay |
| High-flow nasal cannula (HFNC) | Increasingly used for moderate-severe bronchiolitis; provides CPAP-like effect, heated humidification, and washout of dead space |
RSV Prevention
Nirsevimab (Beyfortus) — a long-acting monoclonal antibody — is now recommended for all infants <8 months entering their first RSV season and for high-risk infants 8–19 months entering their second season. It replaced palivizumab (Synagis), which required monthly injections and was limited to high-risk groups. Palivizumab is still available for eligible infants when nirsevimab is not available.
16 Pneumonia in Children
The most likely causative organisms vary significantly by age group, which determines empiric therapy.
Etiology by Age
| Age | Most Common Organisms | Empiric Therapy |
|---|---|---|
| Neonates (<28 days) | GBS, E. coli, Listeria, HSV | Ampicillin + gentamicin (± acyclovir if HSV suspected) |
| 1–3 months | Chlamydia trachomatis (afebrile pneumonitis), RSV, S. pneumoniae, B. pertussis | Macrolide (azithromycin) if afebrile; cephalosporin if febrile/toxic |
| 3 months–5 years | Viral (most common overall), S. pneumoniae (most common bacterial) | High-dose amoxicillin 90 mg/kg/day (outpatient); ampicillin IV (inpatient) |
| 5–18 years | Mycoplasma pneumoniae (most common), S. pneumoniae, Chlamydophila pneumoniae | Macrolide (azithromycin 10 mg/kg day 1, then 5 mg/kg days 2–5) for atypical; amoxicillin or ampicillin if typical bacterial |
Parapneumonic Effusion & Empyema
Complicated parapneumonic effusions occur in ~5% of pediatric pneumonias. Ultrasound is the initial imaging modality. Pleural fluid pH <7.2, glucose <40 mg/dL, or positive Gram stain/culture indicates empyema requiring drainage (chest tube or VATS). S. pneumoniae and S. aureus (including MRSA) are the most common organisms.
17 Fever in Children
Fever is the most common presenting complaint in pediatric emergency departments. The approach to fever varies dramatically by age because the risk of serious bacterial infection (SBI) — UTI, bacteremia, meningitis — is highest in the youngest infants who may appear well despite serious illness.
Age-Based Approach to Fever Without a Source
| Age | Risk Level | Workup | Management |
|---|---|---|---|
| <28 days | Highest risk; neonatal immune immaturity | Full sepsis workup: blood culture, UA/urine culture (cath), LP, CBC, CRP/procalcitonin; CXR if respiratory symptoms; stool culture if diarrhea | Admit and empiric IV antibiotics (ampicillin + gentamicin or cefotaxime) pending cultures; consider HSV coverage (acyclovir) if risk factors |
| 29–60 days | High risk; may use validated criteria to risk-stratify | Blood culture, UA/urine culture, CBC, CRP/procalcitonin; LP if ill-appearing or high inflammatory markers | Low-risk by validated criteria (Rochester, Step-by-Step, PECARN): may observe closely without antibiotics. High-risk: admit and empiric antibiotics |
| 61–90 days | Moderate risk | UA/urine culture; blood culture and CBC if ill-appearing; LP if warranted | Well-appearing with negative UA: consider close outpatient follow-up. Ill-appearing: admit |
| 3–36 months | Lower risk (post-PCV era) | UA/urine culture if female <24 mo or uncircumcised male <12 mo; blood work if T ≥39°C and ill-appearing | UTI is most common SBI; occult bacteremia rare (<1%) in post-PCV/Hib era; treat identified source |
Any fever (rectal temperature ≥38.0°C / 100.4°F) in an infant <28 days old requires a full sepsis workup and empiric IV antibiotics regardless of appearance. This is non-negotiable. These infants can harbor bacteremia, meningitis, or UTI while appearing well. HSV should also be considered in neonates with fever, vesicles, seizures, or elevated LFTs — add acyclovir 60 mg/kg/day IV divided q8h.
18 Otitis Media & Pharyngitis
Acute Otitis Media (AOM)
AOM is the most common reason for antibiotic prescriptions in children. Peak incidence is 6–24 months. Diagnosis requires: (1) acute onset of symptoms, (2) middle ear effusion (bulging TM, impaired mobility on pneumatic otoscopy, air-fluid level), and (3) signs of middle ear inflammation (erythematous TM, otalgia). Most common organisms: S. pneumoniae, nontypeable H. influenzae, and Moraxella catarrhalis.
| Scenario | Management |
|---|---|
| Age ≥6 months, unilateral, non-severe | Watchful waiting for 48–72 hours with follow-up (per AAP 2013); pain management with ibuprofen/acetaminophen |
| Age ≥6 months, bilateral or severe (T ≥39°C, severe otalgia, >48 hr symptoms) | Amoxicillin 80–90 mg/kg/day divided BID ×10 days (<2 yr) or 5–7 days (≥2 yr) |
| Treatment failure (no improvement at 48–72 hr) | High-dose amoxicillin-clavulanate (90 mg/kg/day of amoxicillin component); or ceftriaxone IM ×3 days |
| Penicillin allergy | Cefdinir, cefuroxime, or azithromycin (less effective) |
| Recurrent AOM (≥3 episodes in 6 mo or ≥4 in 12 mo) | Refer for tympanostomy tubes |
Pharyngitis — Group A Streptococcus (GAS)
GAS pharyngitis accounts for ~15–30% of pharyngitis in children ages 5–15 (rare <3 years). Treatment prevents rheumatic fever (ARF) but primarily targets symptom relief and reduced transmission.
| Feature | GAS Pharyngitis | Viral Pharyngitis |
|---|---|---|
| Age | 5–15 years | Any age |
| Symptoms | Sudden onset sore throat, fever, headache, abdominal pain, tonsillar exudates, palatal petechiae, tender anterior cervical lymphadenopathy | Cough, rhinorrhea, conjunctivitis, hoarseness, diarrhea, oral ulcers |
| Diagnosis | Rapid strep test (high specificity); if negative, confirm with throat culture (gold standard) in children | Clinical; viral features suggest non-GAS etiology |
| Treatment | Penicillin V 250 mg BID or 500 mg BID ×10 days; OR amoxicillin 50 mg/kg once daily (max 1 g) ×10 days; pen-allergy: cephalexin, azithromycin | Supportive care only |
19 Common Childhood Exanthems
| Disease | Agent | Rash Description | Key Features |
|---|---|---|---|
| Measles (Rubeola) | Paramyxovirus | Erythematous maculopapular rash starting at face/hairline → spreads cephalocaudal; confluent | 3 C’s: Cough, Coryza, Conjunctivitis; Koplik spots (white spots on buccal mucosa) before rash; high fever; highly contagious |
| Rubella (German measles) | Rubivirus | Pink maculopapular rash starting on face → spreads downward; clears in 3 days | Postauricular and suboccipital lymphadenopathy; mild illness in children; teratogenic in first trimester |
| Roseola (Exanthem subitum) | HHV-6 | Rose-pink macular/maculopapular rash on trunk → spreads peripherally | High fever for 3–5 days, then rash appears as fever breaks; age 6–24 months; febrile seizures common |
| Erythema infectiosum (Fifth disease) | Parvovirus B19 | "Slapped cheek" facial erythema → reticular (lace-like) rash on trunk/extremities | Child is no longer contagious when rash appears; aplastic crisis in sickle cell; hydrops fetalis if maternal infection |
| Varicella (Chickenpox) | VZV | Pruritic vesicles on erythematous base ("dewdrop on rose petal"); lesions in different stages (crops) | Centripetal distribution (trunk > extremities); highly contagious until all vesicles crusted |
| Hand-Foot-Mouth Disease | Coxsackievirus A16 (or Enterovirus 71) | Vesicles/ulcers on oral mucosa; maculopapular or vesicular lesions on palms and soles | Peak 1–4 years; summer/fall; usually self-limited; EV71 can cause severe neurologic disease |
| Scarlet Fever | GAS (erythrogenic toxin) | Diffuse erythematous "sandpaper" rash; spares face (circumoral pallor); Pastia lines (petechiae in skin folds) | Strawberry tongue; follows GAS pharyngitis; treat with penicillin/amoxicillin |
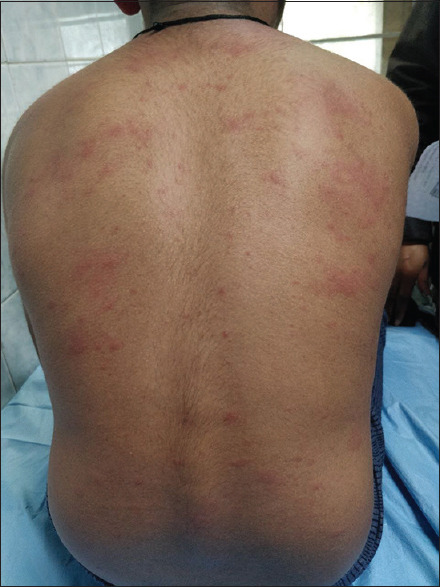
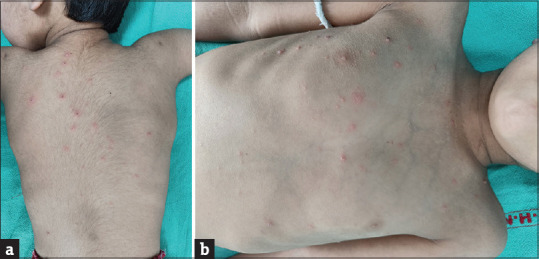
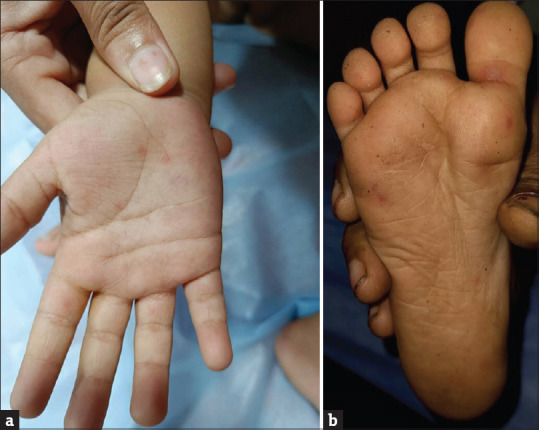
20 Meningitis in Children
Bacterial meningitis is a medical emergency in pediatrics. Prompt recognition and treatment reduce mortality and long-term neurologic sequelae (hearing loss, intellectual disability, seizures).
Age-Based Empiric Therapy
| Age | Common Organisms | Empiric Antibiotics |
|---|---|---|
| <1 month | GBS, E. coli, Listeria | Ampicillin + cefotaxime (or gentamicin) |
| 1–3 months | GBS, E. coli, Listeria, S. pneumoniae, N. meningitidis | Ampicillin + cefotaxime (or ceftriaxone if >28 days) + vancomycin |
| 3 months–18 years | S. pneumoniae, N. meningitidis | Ceftriaxone + vancomycin; add dexamethasone 0.15 mg/kg IV q6h ×4 days (give before or with first antibiotic dose for Hib and S. pneumoniae) |
CSF Interpretation
| Parameter | Normal | Bacterial | Viral (Aseptic) | TB/Fungal |
|---|---|---|---|---|
| Opening pressure | <20 cm H2O | Elevated | Normal/mildly elevated | Elevated |
| WBC | <5 cells/µL | >1,000 (neutrophil predominant) | 10–500 (lymphocyte predominant) | 10–500 (lymphocyte predominant) |
| Protein | <45 mg/dL | Very elevated (>100) | Normal/mildly elevated | Very elevated |
| Glucose | >40 mg/dL (or CSF:serum >0.6) | Low (<40) | Normal | Low |
| Gram stain | Negative | Positive in 60–90% | Negative | AFB/India ink |
Chemoprophylaxis
| Organism | Who Needs Prophylaxis | Regimen |
|---|---|---|
| N. meningitidis | Close contacts (household, daycare, direct exposure to secretions) | Rifampin 10 mg/kg (max 600 mg) q12h ×2 days; OR ciprofloxacin 500 mg ×1 (adults); OR ceftriaxone 250 mg IM ×1 |
| H. influenzae type b | Household contacts if any unvaccinated child <4 years in household | Rifampin 20 mg/kg (max 600 mg) daily ×4 days |
21 UTI in Children
UTI is the most common serious bacterial infection in febrile infants. Presentation varies dramatically with age: neonates present with nonspecific signs (fever, irritability, poor feeding, jaundice), while older children present with classic dysuria, frequency, and suprapubic/flank pain. E. coli causes 80–90% of first UTIs in children.
AAP Guidelines (2011, reaffirmed 2016) — Children 2–24 Months
| Step | Details |
|---|---|
| Diagnosis | Requires BOTH positive urinalysis (pyuria and/or bacteriuria) AND ≥50,000 CFU/mL on culture from catheterized or suprapubic specimen; bag specimens are for screening only (high false-positive rate) |
| Treatment | Oral antibiotics for non-toxic children: cephalexin 50–100 mg/kg/day, cefixime, or TMP-SMX; IV antibiotics if toxic-appearing, unable to tolerate PO, or <2 months old; 7–14 day course |
| Imaging | Renal/bladder ultrasound (RBUS) after first febrile UTI; VCUG indicated if RBUS is abnormal, atypical UTI, or recurrent febrile UTI |
Vesicoureteral Reflux (VUR) Grading
| Grade | Description |
|---|---|
| I | Reflux into non-dilated ureter only |
| II | Reflux into renal pelvis without dilation |
| III | Mild-moderate dilation of ureter, pelvis, and calyces |
| IV | Moderate dilation with blunting of calyceal fornices |
| V | Gross dilation and tortuosity of ureter with loss of papillary impressions |
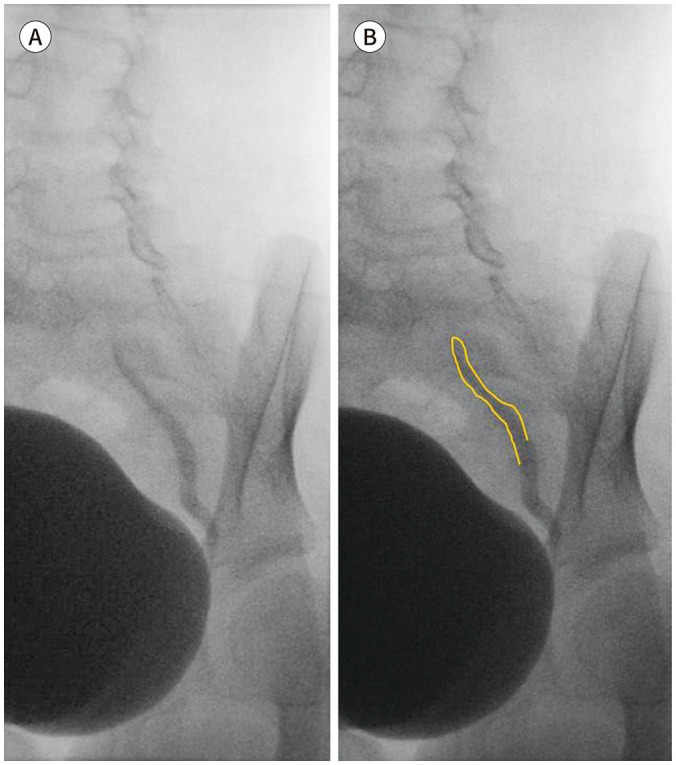
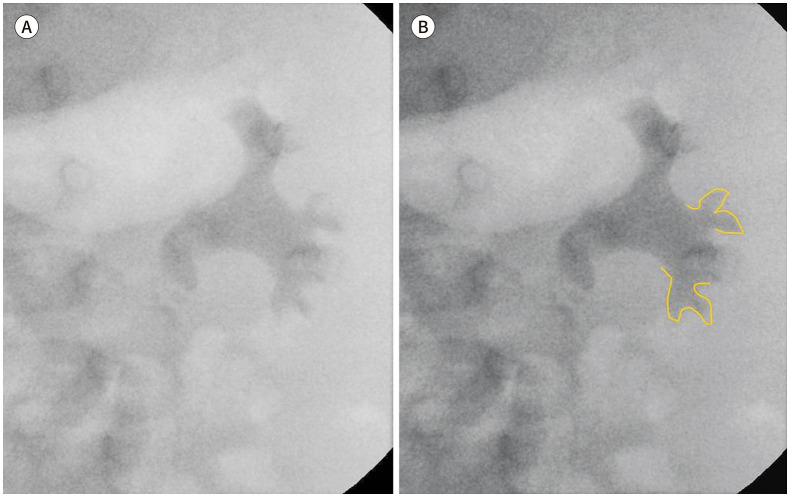
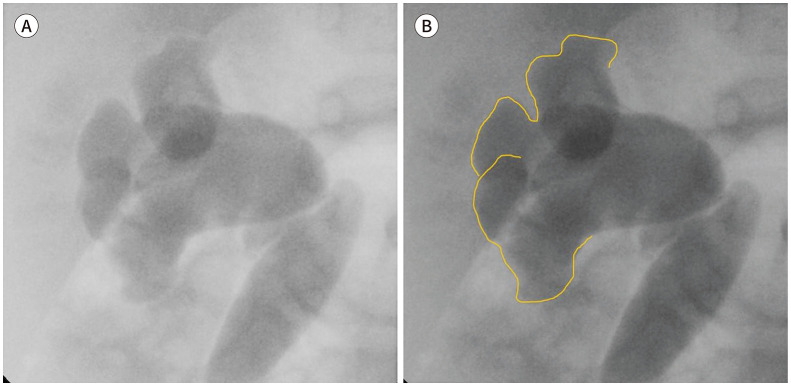
22 Vomiting & Dehydration
Assessment of Dehydration Severity
| Feature | Mild (3–5%) | Moderate (6–9%) | Severe (≥10%) |
|---|---|---|---|
| Mental status | Normal, thirsty | Irritable, restless | Lethargic, obtunded |
| Eyes | Normal | Slightly sunken | Deeply sunken |
| Tears | Present | Decreased | Absent |
| Mucous membranes | Slightly dry | Dry | Parched |
| Skin turgor | Normal | Decreased (slow recoil) | Tenting |
| Cap refill | <2 seconds | 2–3 seconds | >3 seconds |
| Urine output | Slightly decreased | Decreased (<1 mL/kg/hr) | Minimal/absent |
| Heart rate | Normal | Tachycardic | Tachycardic → bradycardic (preterminal) |
Rehydration
| Severity | Approach |
|---|---|
| Mild | ORS (oral rehydration solution) 50 mL/kg over 4 hours + replace ongoing losses |
| Moderate | ORS 100 mL/kg over 4 hours; ondansetron 0.15 mg/kg ODT may facilitate oral rehydration; IV if ORS fails |
| Severe | IV NS or LR bolus 20 mL/kg over 15–20 min (may repeat ×3); then replace deficit + maintenance; if signs of shock: immediate 20 mL/kg bolus |
Pyloric Stenosis
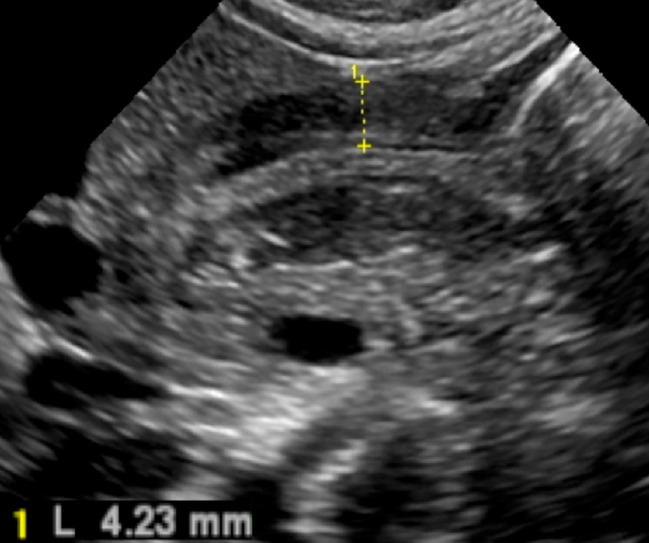
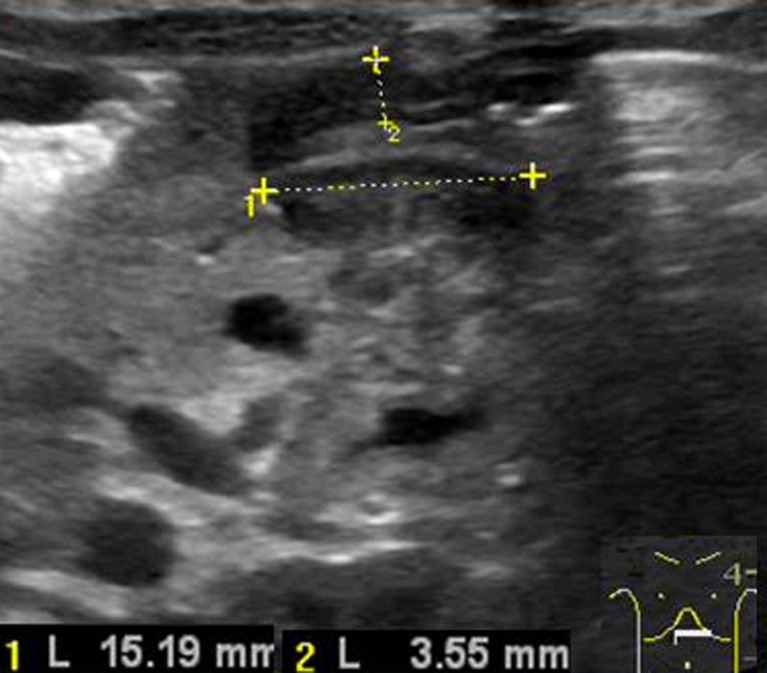
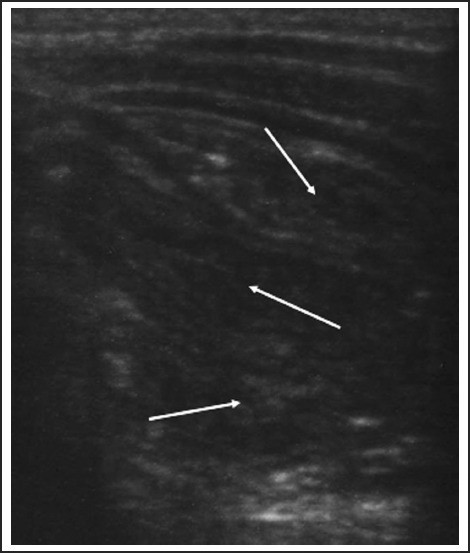
Hypertrophic pyloric stenosis presents at 2–6 weeks of age (classic: firstborn male) with projectile, nonbilious vomiting after feeds, "hungry vomiter" who wants to feed immediately after vomiting. Physical exam may reveal a palpable "olive" in the right upper quadrant. Labs show hypochloremic, hypokalemic metabolic alkalosis (from loss of HCl in vomitus). Ultrasound diagnostic criteria: pyloric muscle thickness >3 mm, pyloric length >15 mm. Treatment: correct electrolytes and dehydration first, then Ramstedt pyloromyotomy.
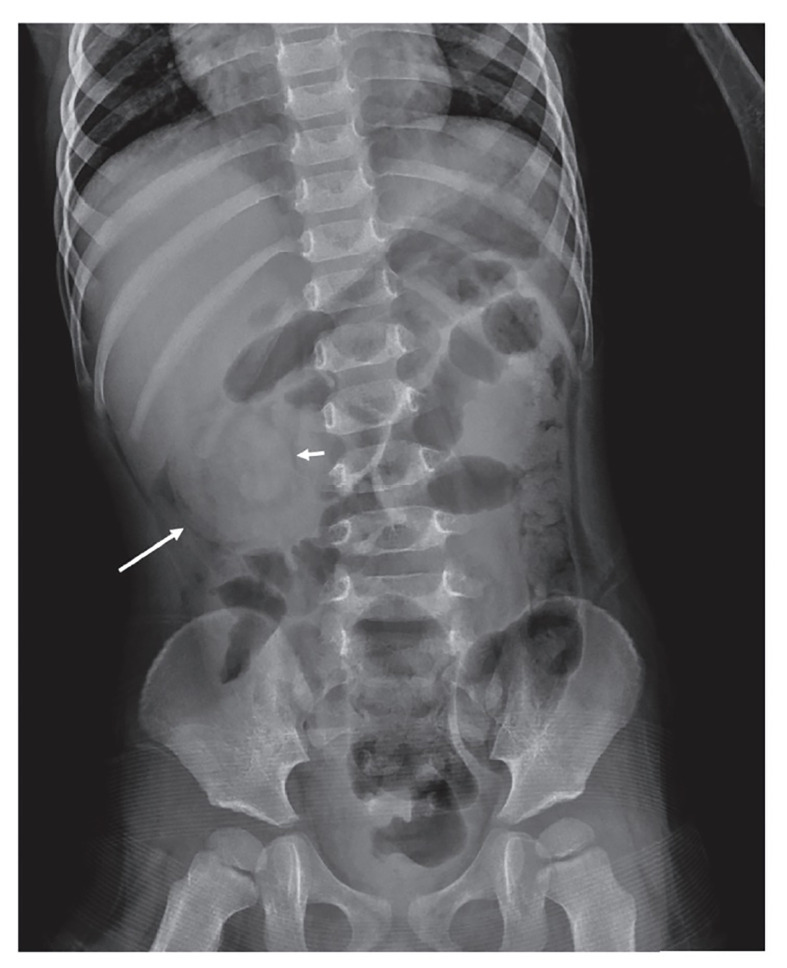
Intussusception
Most common cause of intestinal obstruction in children 6 months–3 years (peak 5–9 months). Most cases are ileocolic and idiopathic (hypertrophied Peyer patches serving as lead point after viral illness). Classic triad (present in <50%): colicky abdominal pain (episodic, drawing up legs), "currant jelly" stools (blood and mucus), and sausage-shaped abdominal mass. Diagnosis: ultrasound showing "target sign" (concentric rings). Treatment: air or contrast enema (both diagnostic and therapeutic) — ~90% success rate; surgery if enema fails, peritonitis, or perforation.
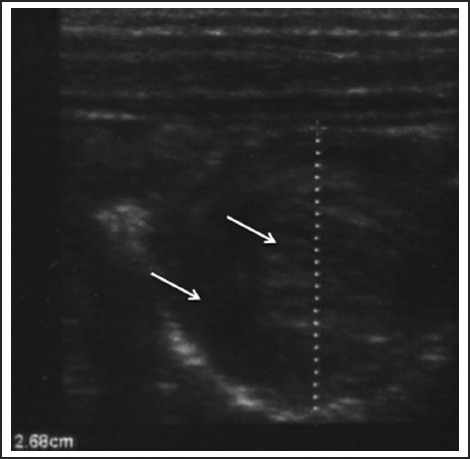
23 Abdominal Pain in Children
Appendicitis
Most common surgical emergency in children. Classic presentation: periumbilical pain migrating to right lower quadrant (McBurney point), anorexia, nausea/vomiting, low-grade fever. The Pediatric Appendicitis Score (PAS) and Alvarado score help risk-stratify. Diagnosis: ultrasound first in children (avoids radiation); CT if ultrasound equivocal. Nonperforated appendicitis: appendectomy (laparoscopic) within 24 hours; some centers now offer antibiotic-first approach for uncomplicated cases. Perforated: IV antibiotics (piperacillin-tazobactam or ceftriaxone + metronidazole) ± percutaneous drainage if abscess, with interval appendectomy 6–8 weeks later.
Meckel Diverticulum
The "rule of 2s": 2% of population, 2 feet from ileocecal valve, 2 inches long, presents by age 2, 2 types of ectopic tissue (gastric and pancreatic). Most common presenting symptom in children is painless rectal bleeding (dark red or brick-colored) due to acid secretion from ectopic gastric mucosa causing ulceration of adjacent ileal tissue. Diagnosis: Meckel scan (technetium-99m pertechnetate scintigraphy — tags ectopic gastric mucosa). Treatment: surgical resection.
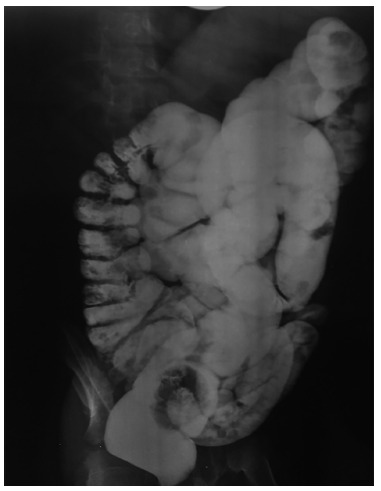
Malrotation with Midgut Volvulus
Malrotation results from incomplete rotation and fixation of the midgut during embryologic development. The narrow mesenteric pedicle predisposes to midgut volvulus — twisting of the bowel around the SMA — causing ischemia of the entire midgut. Presentation: bilious (green) vomiting in a neonate is volvulus until proven otherwise. Diagnosis: upper GI series (gold standard) showing abnormal position of duodenojejunal junction (ligament of Treitz). Emergency treatment: Ladd procedure (detorsion, lysis of Ladd bands, appendectomy, broadening of mesenteric base). Time is critical — delay leads to bowel necrosis and short gut syndrome.
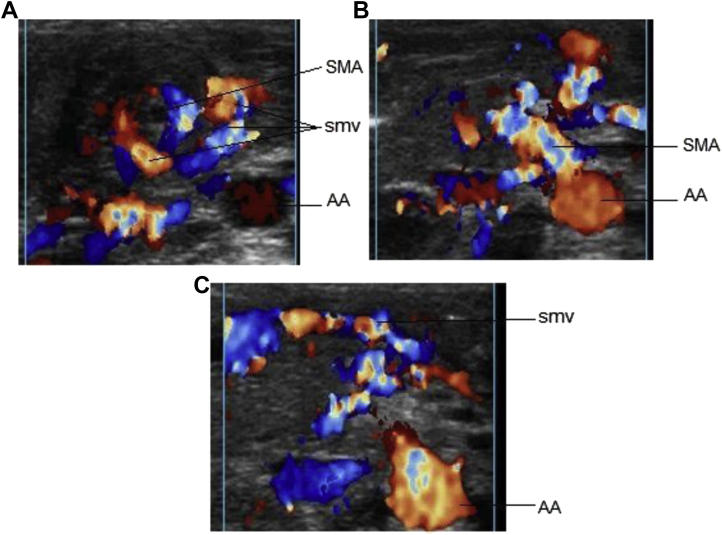
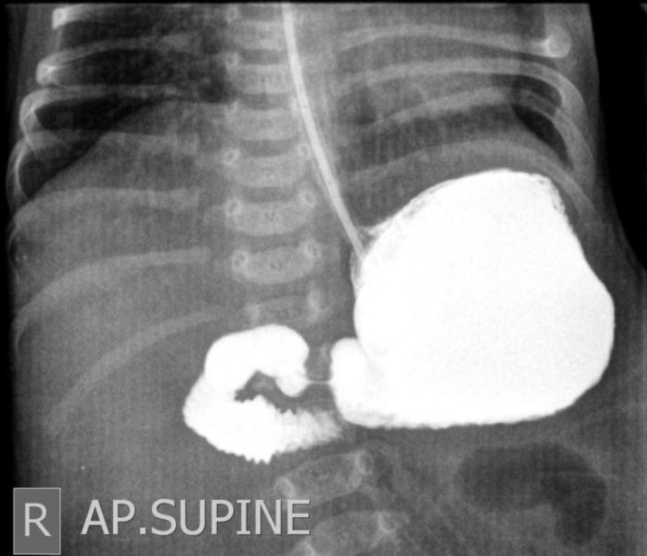
Hirschsprung Disease
Congenital absence of ganglion cells in the distal colon (aganglionosis), most commonly affecting the rectosigmoid. Presents with failure to pass meconium within 48 hours of birth, abdominal distension, and bilious vomiting. Can present later with chronic constipation (ribbon-like stools, failure to thrive). Diagnosis: barium enema (transition zone), rectal suction biopsy (gold standard — absent ganglion cells, hypertrophied nerve trunks). Treatment: pull-through procedure (Soave, Swenson, or Duhamel).
24 Pediatric GI Emergencies
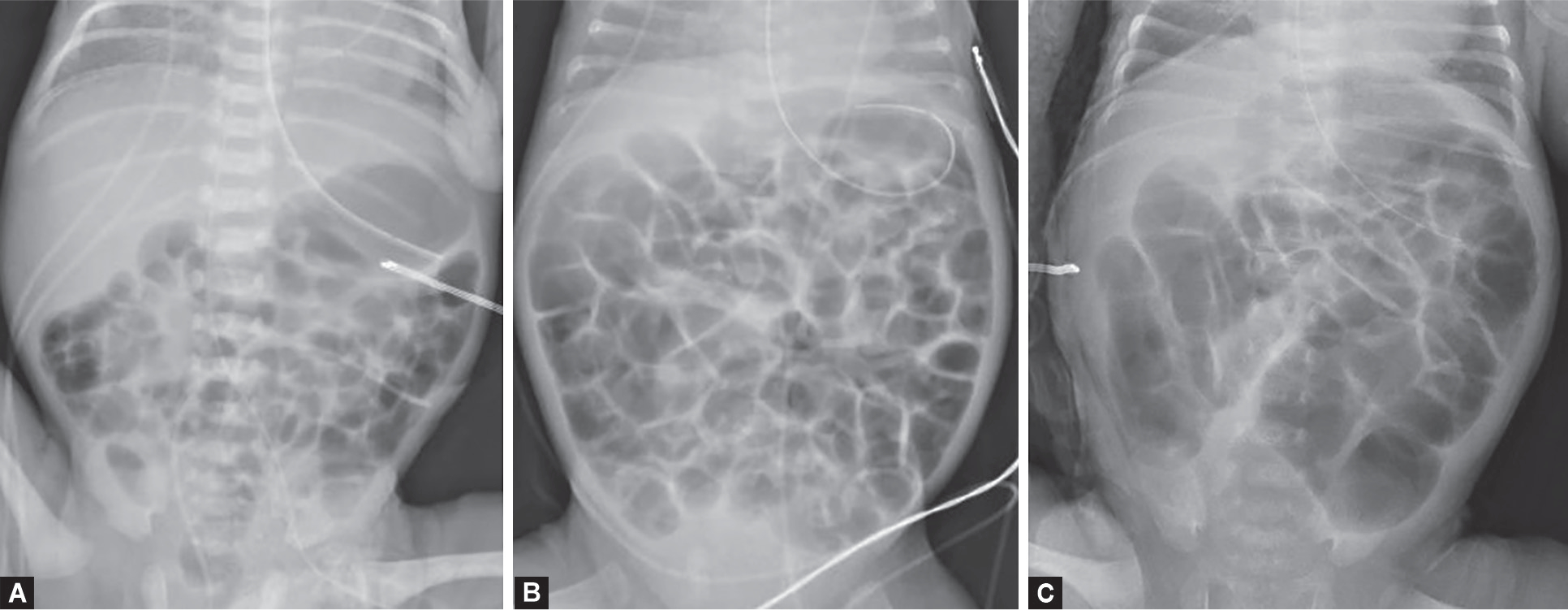
Necrotizing Enterocolitis (NEC)
NEC is the most common GI emergency in premature neonates (primarily <32 weeks GA and <1,500 g). Pathogenesis involves mucosal injury from ischemia, formula feeding, and bacterial invasion. Presentation: feeding intolerance, abdominal distension, bloody stools, bilious aspirates, and systemic signs (apnea, lethargy, temperature instability, DIC).
| Modified Bell Staging | Clinical | Radiographic | Treatment |
|---|---|---|---|
| Stage I (Suspected) | Nonspecific signs, feeding intolerance, mild distension | Normal or mildly dilated loops | NPO, NGT decompression, IV antibiotics, serial exams |
| Stage II (Definite) | Bloody stools, absent bowel sounds, abdominal wall erythema | Pneumatosis intestinalis (pathognomonic); portal venous gas | NPO 7–14 days, IV antibiotics (ampicillin + gentamicin + metronidazole), TPN |
| Stage III (Advanced) | Peritonitis, hemodynamic instability, DIC | Pneumoperitoneum (free air = perforation) | Surgical consultation; laparotomy or peritoneal drain; resuscitation |
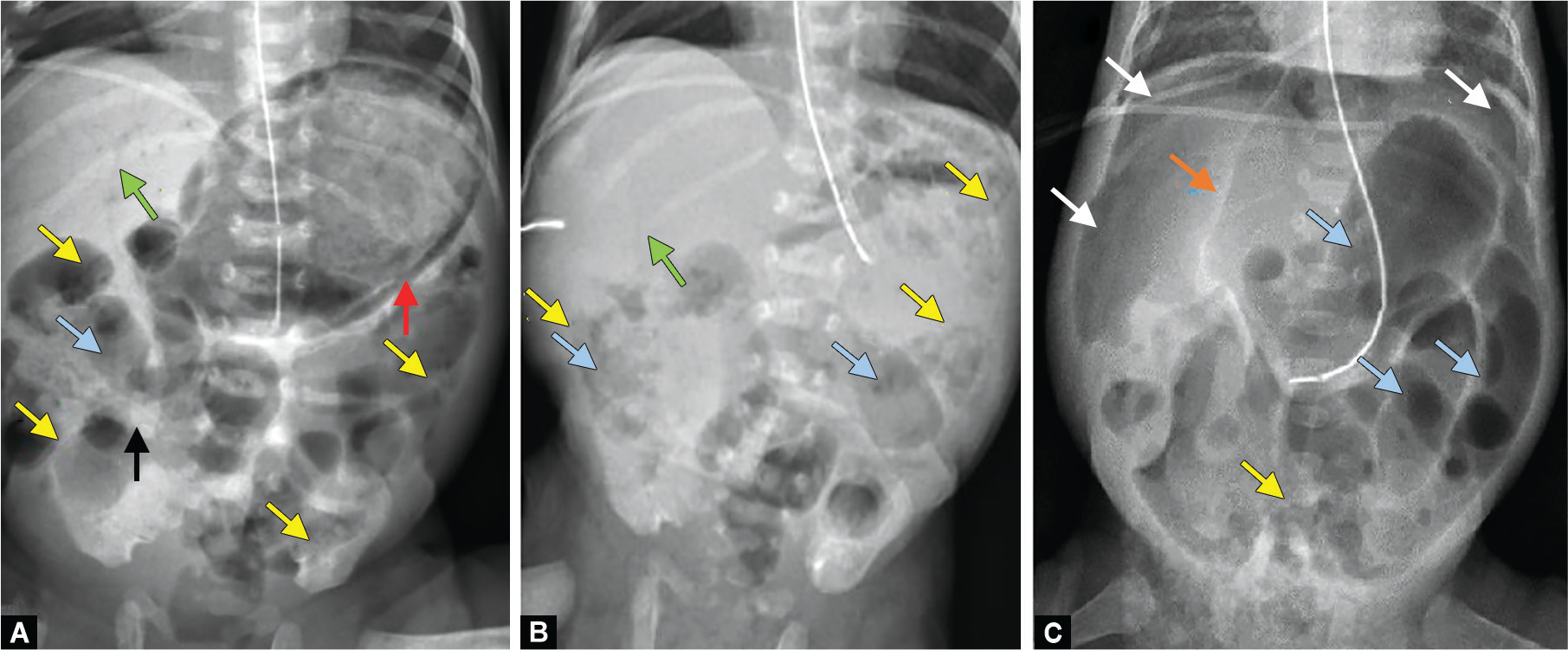
Pyloric Stenosis — Quick Reference
See Section 22 for full details. Key points: projectile nonbilious vomiting at 2–6 weeks; "olive" mass; hypochloremic hypokalemic metabolic alkalosis; US >3 mm thickness, >15 mm length; pyloromyotomy after correcting metabolic derangements.
Intussusception — Quick Reference
See Section 22 for full details. Key points: 6 months–3 years; episodic colicky pain; currant jelly stools; US target sign; air enema reduction 90% success; surgery if failed enema or perforation.
25 Celiac Disease & Food Allergies
Celiac Disease
An immune-mediated enteropathy triggered by gluten (wheat, barley, rye) in genetically predisposed individuals (HLA-DQ2 or DQ8). Prevalence ~1% in Western populations. Classic presentation in children: chronic diarrhea, abdominal distension, failure to thrive, irritability, and iron deficiency anemia beginning after introduction of gluten-containing foods (typically 6–24 months). Older children may present with constipation, short stature, delayed puberty, or dermatitis herpetiformis.
| Diagnostic Test | Details |
|---|---|
| Initial screen | TTG-IgA (tissue transglutaminase IgA) + total serum IgA (to rule out IgA deficiency, present in 2–3% of celiac patients causing false-negative TTG-IgA) |
| If IgA deficient | TTG-IgG or deamidated gliadin peptide (DGP) IgG |
| Confirmatory | Esophagogastroduodenoscopy with small bowel biopsy showing villous atrophy, crypt hyperplasia, and intraepithelial lymphocytosis (Marsh classification) |
| Treatment | Strict lifelong gluten-free diet; monitor with TTG-IgA; screen for associated conditions (Type 1 DM, thyroiditis, IgA nephropathy) |
Food Allergies
IgE-mediated food allergy affects ~6–8% of children <3 years. The most common allergens are cow’s milk, egg, peanut, tree nuts, soy, wheat, fish, and shellfish. Milk, egg, soy, and wheat allergies are usually outgrown by school age; peanut, tree nut, and shellfish allergies tend to persist.
Epinephrine IM (anterolateral thigh) is the first-line treatment: 0.01 mg/kg of 1:1,000 (0.1 mg/mL) solution, max 0.3 mg in children <30 kg, max 0.5 mg in adolescents. May repeat every 5–15 minutes. Position supine with legs elevated (unless vomiting or respiratory distress). After epinephrine: IV fluid bolus (20 mL/kg NS), albuterol for bronchospasm, H1/H2 blockers (diphenhydramine + ranitidine), methylprednisolone 1–2 mg/kg IV. Observe 4–6 hours for biphasic reaction. Prescribe epinephrine auto-injector at discharge.
26 Congenital Heart Disease — Acyanotic
Acyanotic (left-to-right shunt) lesions present with signs of pulmonary overcirculation: tachypnea, poor feeding, diaphoresis with feeds, recurrent respiratory infections, and failure to thrive. They do not cause cyanosis initially, but long-standing shunts can cause pulmonary hypertension and eventually Eisenmenger syndrome (shunt reversal → cyanosis).
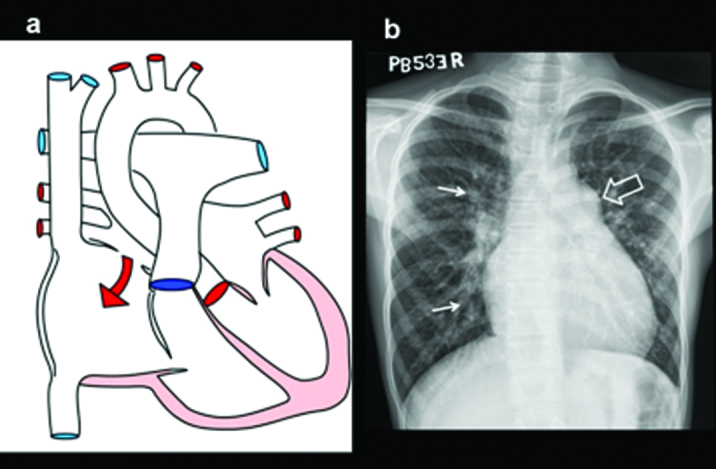
| Lesion | Pathology | Murmur | Management |
|---|---|---|---|
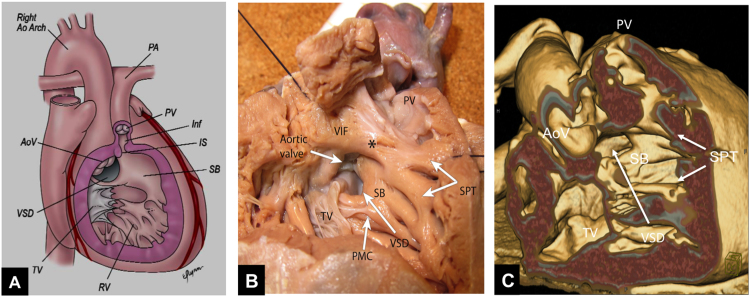
| VSD | Most common CHD (~30%); communication between ventricles; L→R shunt | Holosystolic murmur at LLSB; louder murmur = smaller VSD (more turbulence) | Small: observe (many close spontaneously by age 2). Large: diuretics, surgical closure if symptomatic HF |
| ASD | Communication between atria; L→R shunt; often asymptomatic in childhood | Systolic ejection murmur at LUSB (increased flow across pulmonic valve); fixed split S2 | Secundum type: transcatheter device closure if significant shunt (Qp:Qs >1.5:1); primum: surgical repair |
| PDA | Persistent patency of ductus arteriosus; L→R shunt from aorta to PA | Continuous "machinery" murmur at left infraclavicular area; bounding pulses, wide pulse pressure | Preterm: indomethacin or ibuprofen (prostaglandin inhibition). Term: transcatheter or surgical closure if hemodynamically significant |
| Coarctation of Aorta | Narrowing of aorta, typically at insertion of ductus arteriosus (juxtaductal) | Systolic murmur between scapulae; upper extremity hypertension + weak femoral pulses; BP gradient >20 mmHg arm-to-leg | Neonatal (critical): PGE1 to maintain ductal patency; surgical repair. Older: balloon angioplasty ± stent or surgical repair |
| Aortic Stenosis | Obstruction at valvular (most common), subvalvular, or supravalvular (Williams syndrome) level | Systolic ejection murmur at RUSB radiating to carotids; ejection click if valvular | Balloon valvuloplasty for severe valvular AS; surgical intervention for subvalvular/supravalvular |
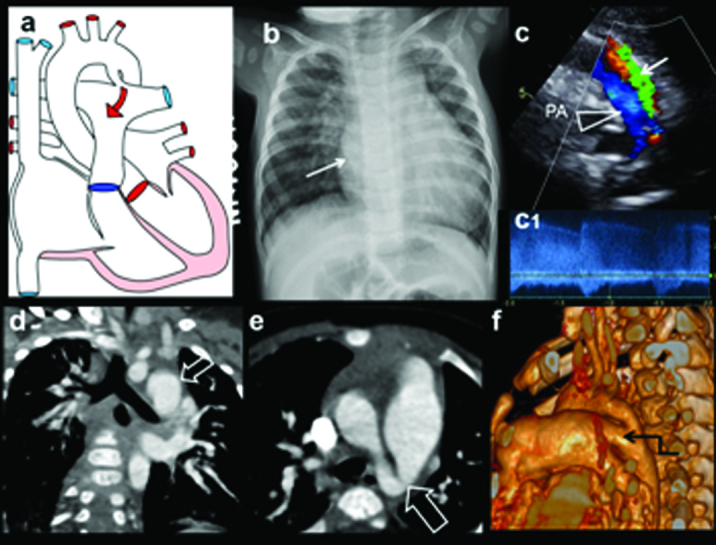
27 Congenital Heart Disease — Cyanotic
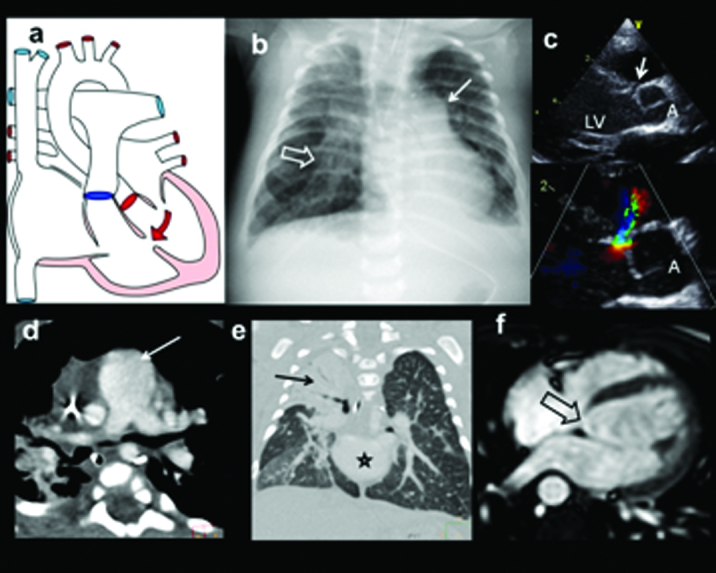
Cyanotic CHD presents with hypoxemia that does not significantly improve with supplemental oxygen (the "hyperoxia test": PaO2 remains <150 mmHg after 10 minutes of 100% FiO2, suggesting intracardiac right-to-left shunt rather than lung disease). The "5 T’s" of cyanotic CHD:
| Lesion | Pathology | Key Features | Management |
|---|---|---|---|
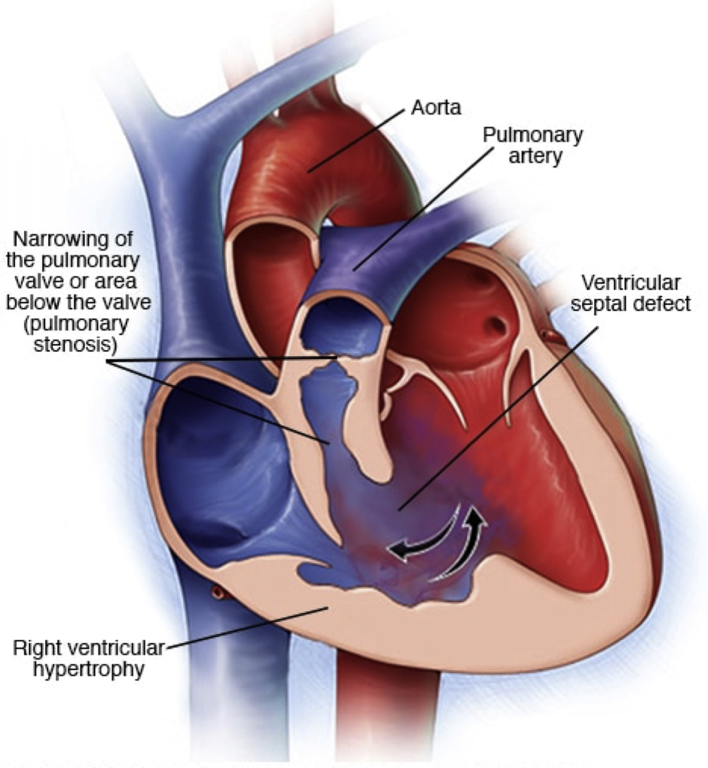
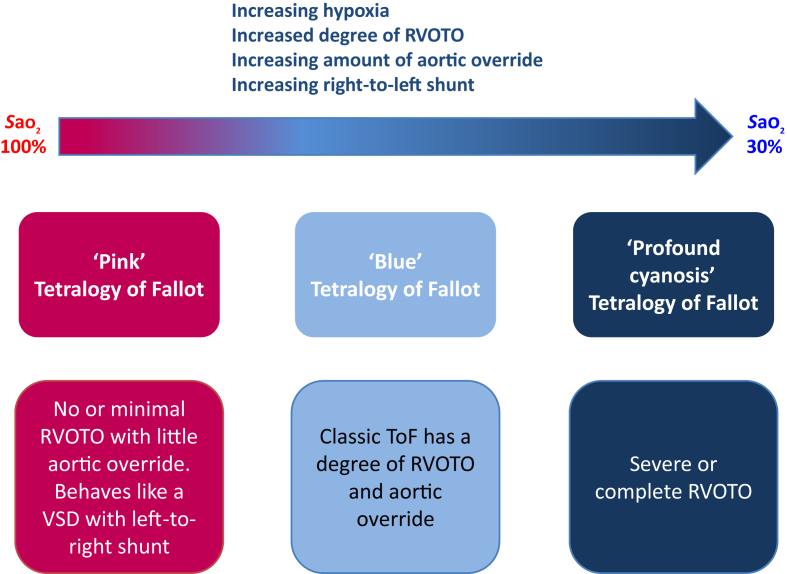
| Tetralogy of Fallot (TOF) | VSD + overriding aorta + RV outflow tract obstruction + RV hypertrophy | Most common cyanotic CHD beyond neonatal period; "boot-shaped" heart on CXR; "tet spells" (hypercyanotic episodes) | Tet spell: knee-chest position, oxygen, morphine, IV fluid, phenylephrine. Definitive: complete surgical repair at 3–6 months |
| Transposition of the Great Arteries (TGA) | Aorta arises from RV, PA arises from LV → parallel circuits | Most common cyanotic CHD at birth; cyanosis in first hours of life; "egg on a string" heart on CXR | PGE1 to maintain PDA (allows mixing); balloon atrial septostomy (Rashkind); definitive: arterial switch operation (Jatene) within first 2 weeks |
| Truncus Arteriosus | Single great vessel overriding a VSD giving rise to aorta, PA, and coronary arteries | Cyanosis + HF; single S2; associated with DiGeorge syndrome (22q11.2 deletion) | Surgical repair in neonatal period |
| TAPVR | All pulmonary veins drain to systemic venous system (SVC, coronary sinus, IVC) instead of LA | Cyanosis; survival requires ASD/PFO for mixing; obstructed TAPVR = emergency ("snowman" heart on CXR in supracardiac type) | Surgical redirection of pulmonary veins to LA |
| Tricuspid Atresia | Absent tricuspid valve; no connection between RA and RV | Cyanosis; requires ASD + VSD or PDA for blood flow; left axis deviation on ECG (unusual in CHD) | Staged palliation: BT shunt → Glenn → Fontan |
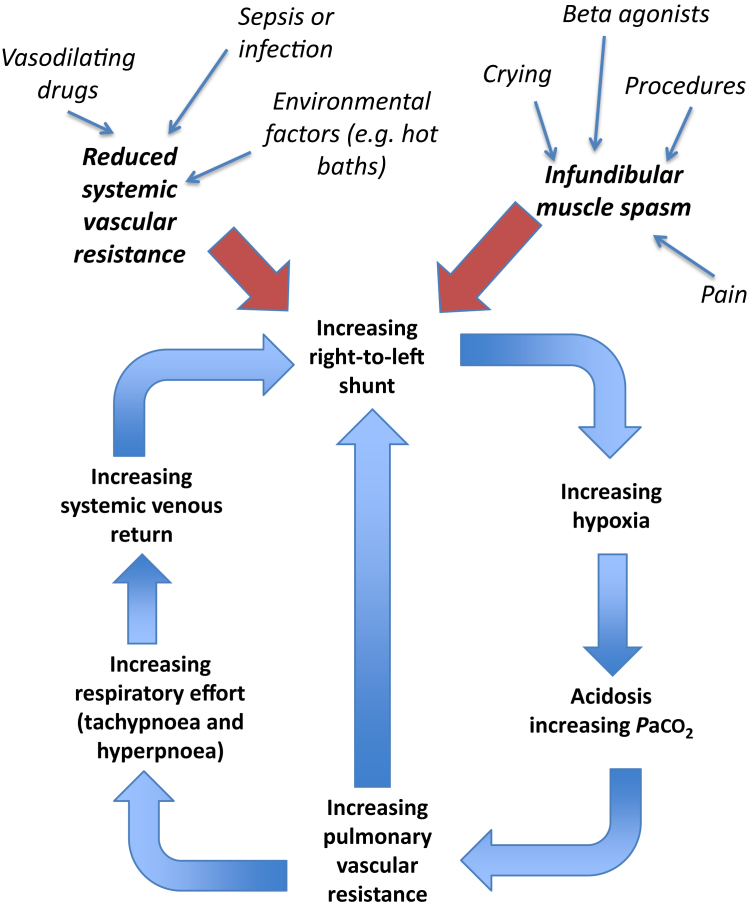
Occur in unrepaired TOF, typically in infants 2–4 months old, triggered by crying, feeding, or defecation. Mechanism: increased RV outflow tract obstruction → increased right-to-left shunt → profound cyanosis → metabolic acidosis → further RVOT spasm (vicious cycle). Management: (1) knee-chest position (increases SVR, decreases venous return), (2) supplemental O2, (3) IV fluid bolus, (4) morphine 0.1 mg/kg (reduces agitation), (5) phenylephrine 5–20 mcg/kg IV (increases SVR, reverses shunt), (6) emergent surgical consultation if refractory.
28 Kawasaki Disease
Kawasaki disease (KD) is an acute, self-limited vasculitis of medium-sized arteries with a predilection for the coronary arteries. It is the leading cause of acquired heart disease in children in developed countries. Peak incidence: 6 months to 5 years; male predominance (1.5:1); highest incidence in Asian/Pacific Islander populations.
Diagnostic Criteria (Classic KD)
Fever ≥5 days PLUS ≥4 of the following 5 criteria:
| # | Criterion | Details |
|---|---|---|
| 1 | Bilateral conjunctival injection | Non-exudative, limbal-sparing |
| 2 | Oral mucous membrane changes | Strawberry tongue, erythematous/cracked lips, oropharyngeal erythema |
| 3 | Extremity changes | Erythema/edema of hands and feet (acute); periungual desquamation (convalescent) |
| 4 | Polymorphous rash | Non-vesicular; often accentuated in groin; not specific |
| 5 | Cervical lymphadenopathy | ≥1.5 cm, usually unilateral |
Suspect in children with fever ≥5 days and only 2–3 criteria, especially if <12 months (who are at highest risk for coronary aneurysms and most likely to have incomplete presentation). If CRP ≥3 mg/dL and/or ESR ≥40 mm/hr with 3 or more supplementary lab criteria (albumin ≤3.0, anemia, elevated ALT, platelets >450K after day 7, WBC ≥15K, urine WBC ≥10/hpf), treat as KD. Echocardiogram should be obtained — coronary abnormalities on echo are diagnostic.
Treatment
| Therapy | Details |
|---|---|
| IVIG | 2 g/kg IV as single infusion over 10–12 hours; given within first 10 days of illness (ideally by day 7). Reduces coronary aneurysm risk from ~25% to <5% |
| Aspirin | High-dose (80–100 mg/kg/day divided QID) until afebrile ×48–72 hours, then low-dose (3–5 mg/kg/day) for 6–8 weeks (or indefinitely if coronary aneurysms present) |
| IVIG-resistant (≥36 hr persistent/recurrent fever after IVIG) | Second IVIG infusion; consider IV methylprednisolone 30 mg/kg/day ×3 days; infliximab in refractory cases |
Coronary Artery Follow-Up
Echocardiography at diagnosis, 1–2 weeks, and 6–8 weeks. Coronary artery aneurysms are classified by z-score: small (z 2.5–5), medium (z 5–10), giant (z ≥10 or absolute diameter ≥8 mm). Giant aneurysms require long-term anticoagulation (warfarin + aspirin) and carry risk of thrombosis, stenosis, and MI.
29 Short Stature & Growth Disorders
Short stature is defined as height <3rd percentile or >2 SD below the mean for age and sex. The differential is broad, but most children have either constitutional delay of growth and puberty or familial (genetic) short stature, both of which are normal variants.
Differential Diagnosis
| Category | Features | Bone Age |
|---|---|---|
| Constitutional delay | Short for age but normal growth velocity; delayed puberty; family history of "late bloomers"; will reach normal adult height | Delayed (predicts more growth remaining) |
| Familial short stature | Short parents; growing along low-but-consistent percentile; normal growth velocity; normal puberty timing | Normal (matches chronologic age) |
| GH deficiency | Severe short stature; declining growth velocity (<5 cm/yr after age 2); increased truncal adiposity; micropenis/midline defects; neonatal hypoglycemia | Delayed |
| Hypothyroidism | Growth deceleration, weight gain, fatigue, constipation, delayed puberty | Delayed |
| Turner syndrome (45,X) | Short stature, webbed neck, shield chest, coarctation, streak gonads, primary amenorrhea; affects girls only | Mildly delayed |
| Skeletal dysplasias | Disproportionate short stature (limb vs trunk); achondroplasia most common | Variable |
| Chronic disease | IBD, celiac, renal failure, cystic fibrosis, chronic steroid use | Delayed |
Workup
Initial evaluation: growth velocity calculation (plot serial heights), bone age X-ray (left hand and wrist, Greulich-Pyle atlas), CBC, CMP, ESR, TSH/free T4, celiac panel (TTG-IgA), IGF-1 and IGFBP-3 (GH axis screening). If IGF-1 low, GH stimulation testing (clonidine or arginine) is the gold standard for GH deficiency diagnosis. Karyotype in all short girls (to rule out Turner syndrome).
30 Precocious & Delayed Puberty
Tanner Staging
| Stage | Girls (Breast) | Girls (Pubic Hair) | Boys (Genitalia) | Boys (Pubic Hair) |
|---|---|---|---|---|
| I | Prepubertal | None | Prepubertal | None |
| II | Breast buds (thelarche) | Sparse, straight, medial labia | Testicular enlargement (>4 mL / >2.5 cm) | Sparse, base of penis |
| III | Breast elevation | Darker, curlier, over mons | Penile lengthening | Darker, curlier |
| IV | Areola forms secondary mound | Adult-type, not to thighs | Penile widening, glans development | Adult-type, not to thighs |
| V | Adult contour | Adult, extends to thighs | Adult | Adult, extends to thighs |
Precocious Puberty
Defined as onset of secondary sexual characteristics before age 8 in girls or 9 in boys.
| Type | Mechanism | Causes | Workup / Treatment |
|---|---|---|---|
| Central (GnRH-dependent) | Premature activation of HPG axis; follows normal pubertal sequence | Idiopathic (most common in girls); CNS tumors (hamartoma, astrocytoma), hydrocephalus, radiation | Bone age, LH/FSH (LH predominant), estradiol/testosterone, brain MRI; treat with GnRH agonist (leuprolide) |
| Peripheral (GnRH-independent) | Sex steroids from gonads or adrenals, independent of HPG axis | CAH, adrenal tumors, gonadal tumors, McCune-Albright syndrome, exogenous hormones | Suppressed LH/FSH with elevated sex steroids; adrenal/gonadal imaging; treat underlying cause |
Delayed Puberty
No breast development by age 13 in girls; no testicular enlargement by age 14 in boys. Most common cause in both sexes: constitutional delay. Pathologic causes include hypogonadotropic hypogonadism (Kallmann syndrome — associated with anosmia), hypergonadotropic hypogonadism (Turner syndrome in girls, Klinefelter syndrome in boys), chronic disease, and eating disorders.
31 Pediatric Diabetes
Type 1 Diabetes
Autoimmune destruction of pancreatic beta cells; accounts for ~95% of diabetes in children <10 years. Presentation: polyuria, polydipsia, polyphagia, weight loss over days to weeks. ~30–40% present in DKA at diagnosis. Autoantibodies: GAD65, IA-2, insulin autoantibodies (IAA), ZnT8.
DKA Management in Children
DKA is defined as glucose >200 mg/dL + pH <7.30 or bicarb <15 + ketonemia/ketonuria. The most feared complication of pediatric DKA treatment is cerebral edema (0.5–1% incidence, 21–24% mortality). Risk factors: young age, new-onset DM, severe DKA, rapid fluid administration, rapid glucose decline, failure of Na+ to rise with treatment. Management protocol: (1) IV NS bolus 10–20 mL/kg over 1 hour for hemodynamic instability (avoid rapid bolusing if hemodynamically stable); (2) Calculate fluid deficit and replace over 24–48 hours (not faster); (3) Start insulin infusion 0.05–0.1 units/kg/hr (no bolus) AFTER 1 hour of fluids; (4) Target glucose decline ≤50–100 mg/dL/hr; (5) Monitor neurologic status hourly.
| DKA Severity | pH | Bicarbonate | Clinical |
|---|---|---|---|
| Mild | 7.20–7.30 | 10–15 mEq/L | Alert, tolerating oral fluids |
| Moderate | 7.10–7.19 | 5–9 mEq/L | Kussmaul respirations, may be drowsy |
| Severe | <7.10 | <5 mEq/L | Obtunded, hemodynamically unstable |
Type 2 Diabetes in Adolescents
Rising incidence parallel to the obesity epidemic, particularly in minority populations (Native American, Black, Hispanic, Asian/Pacific Islander). Typically presents in overweight/obese adolescents (age ≥10 or pubertal) with acanthosis nigricans, family history, and insidious onset. C-peptide is normal or elevated (vs. low in T1DM). Treatment: lifestyle modification + metformin (FDA-approved ≥10 years); insulin if HbA1c >8.5% or symptomatic hyperglycemia at presentation.
32 Febrile Seizures
Febrile seizures are the most common seizure type in children, affecting 2–5% of children ages 6 months to 5 years. They occur in the setting of fever (≥38°C / 100.4°F) without evidence of CNS infection or other defined cause.
Simple vs. Complex
| Feature | Simple Febrile Seizure | Complex Febrile Seizure |
|---|---|---|
| Duration | <15 minutes | ≥15 minutes |
| Type | Generalized (tonic-clonic) | Focal features |
| Recurrence in 24 hr | Does not recur | Recurs within 24 hours |
| Postictal | Brief | May be prolonged; focal deficit (Todd paralysis) |
Management
| Scenario | Approach |
|---|---|
| Simple febrile seizure, well-appearing, recovered | Reassurance; identify source of fever; NO EEG, NO imaging, NO LP (in ≥12 months fully immunized); counsel on recurrence risk |
| Complex febrile seizure | Consider LP (especially if <12 months, partially immunized, or pretreated with antibiotics); consider EEG; neuroimaging if focal features |
| Febrile status epilepticus (>5 min) | Benzodiazepine (midazolam intranasal 0.2 mg/kg or diazepam rectal 0.5 mg/kg); ABCs; identify/treat infection |
33 ADHD
Attention-deficit/hyperactivity disorder is the most common neurodevelopmental disorder in children, affecting ~8–10% of school-age children. Boys are diagnosed 2–3× more often than girls (partly due to more externalizing behavior; girls more often present with inattentive type).
DSM-5 Diagnostic Criteria
≥6 symptoms (or ≥5 if age ≥17) of inattention and/or hyperactivity-impulsivity, present for ≥6 months, in ≥2 settings (school, home, work), with onset before age 12. Symptoms must cause functional impairment and not be better explained by another mental disorder.
| Presentation | Features |
|---|---|
| Predominantly Inattentive | Difficulty sustaining attention, careless mistakes, poor organization, loses things, easily distracted, forgetful |
| Predominantly Hyperactive-Impulsive | Fidgets, leaves seat, runs/climbs inappropriately, talks excessively, interrupts, difficulty waiting turn |
| Combined | Meets criteria for both inattention and hyperactivity-impulsivity (most common presentation) |
Treatment
| Age | First-Line | Details |
|---|---|---|
| 4–5 years | Behavioral therapy (parent training) | Medication only if behavioral therapy fails; methylphenidate if needed |
| 6–11 years | FDA-approved medication AND/OR behavioral therapy | Stimulants are first-line: methylphenidate (Ritalin, Concerta) or amphetamines (Adderall, Vyvanse) |
| ≥12 years | FDA-approved medication with or without behavioral therapy | Stimulants first-line; non-stimulants (atomoxetine, guanfacine, clonidine) for those who cannot tolerate stimulants |
Monitoring
Before starting stimulants: height, weight, blood pressure, heart rate, personal/family cardiac history. Monitor every 3–6 months: growth parameters (stimulants can suppress appetite/growth), BP, HR, symptom control, side effects (insomnia, appetite suppression, irritability, headache). No routine ECG required unless cardiac history or symptoms.
34 Autism Spectrum Disorder
ASD is a neurodevelopmental disorder characterized by persistent deficits in social communication/interaction and restricted, repetitive behaviors/interests. Prevalence: ~1 in 36 children (CDC 2023), with 4:1 male-to-female ratio.
DSM-5 Criteria (Both Required)
| Domain | Examples |
|---|---|
| A. Deficits in social communication and social interaction (all three must be present) | (1) Deficits in social-emotional reciprocity (back-and-forth conversation, shared interests, emotions). (2) Deficits in nonverbal communication (eye contact, gestures, facial expression). (3) Deficits in developing/maintaining relationships (adjusting behavior to context, sharing play, making friends). |
| B. Restricted, repetitive behaviors (at least 2 of 4) | (1) Stereotyped movements, speech, or object use (echolalia, hand flapping, lining up toys). (2) Insistence on sameness, inflexible routines. (3) Highly restricted, fixated interests (abnormal intensity). (4) Hyper- or hyporeactivity to sensory input. |
Screening & Diagnosis
AAP recommends universal screening with M-CHAT-R/F at 18 and 24 months. Early signs include lack of pointing by 12 months, no single words by 16 months, no 2-word phrases by 24 months, lack of joint attention, and regression of language or social skills. Diagnosis is made by comprehensive developmental evaluation (developmental pediatrician, child psychologist, or multidisciplinary team) using standardized tools (ADOS-2, ADI-R).
Management
| Intervention | Details |
|---|---|
| Applied Behavior Analysis (ABA) | Gold-standard behavioral intervention; intensive, early initiation (<3 years) yields best outcomes |
| Speech-language therapy | For communication deficits; augmentative/alternative communication (AAC) devices if needed |
| Occupational therapy | Sensory integration, fine motor skills, daily living activities |
| Medications | No medication treats core ASD symptoms. Risperidone and aripiprazole are FDA-approved for irritability/aggression in ASD (age ≥5–6). SSRIs for anxiety, stimulants for comorbid ADHD. |
| Educational support | IEP (Individualized Education Program) under IDEA; structured environment; social skills training |
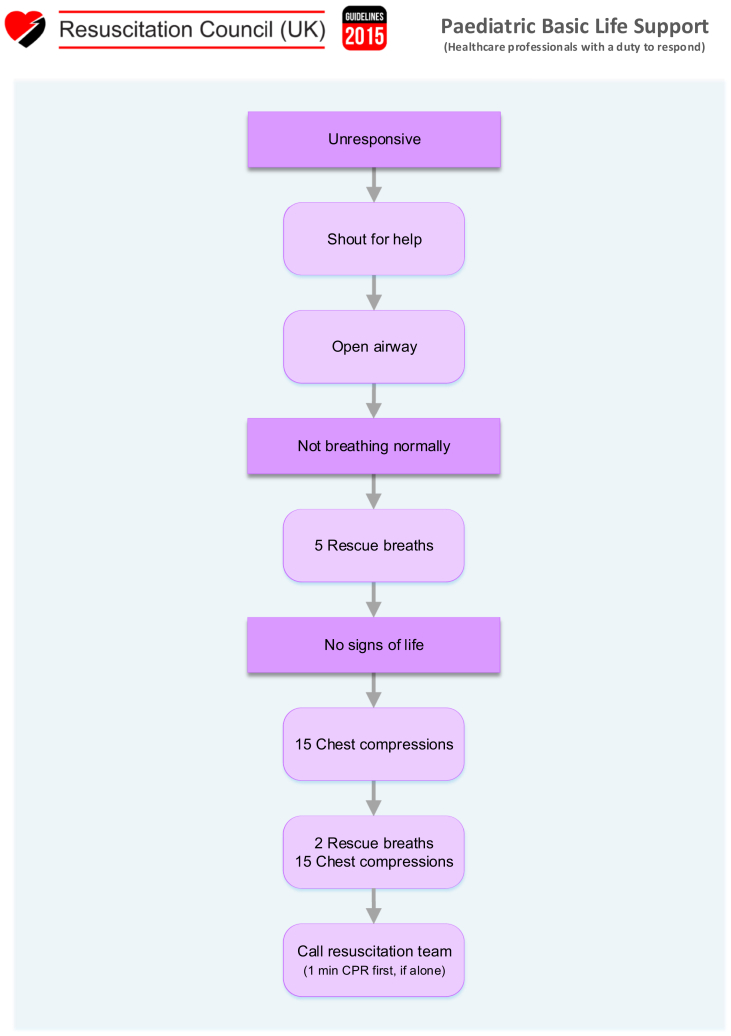
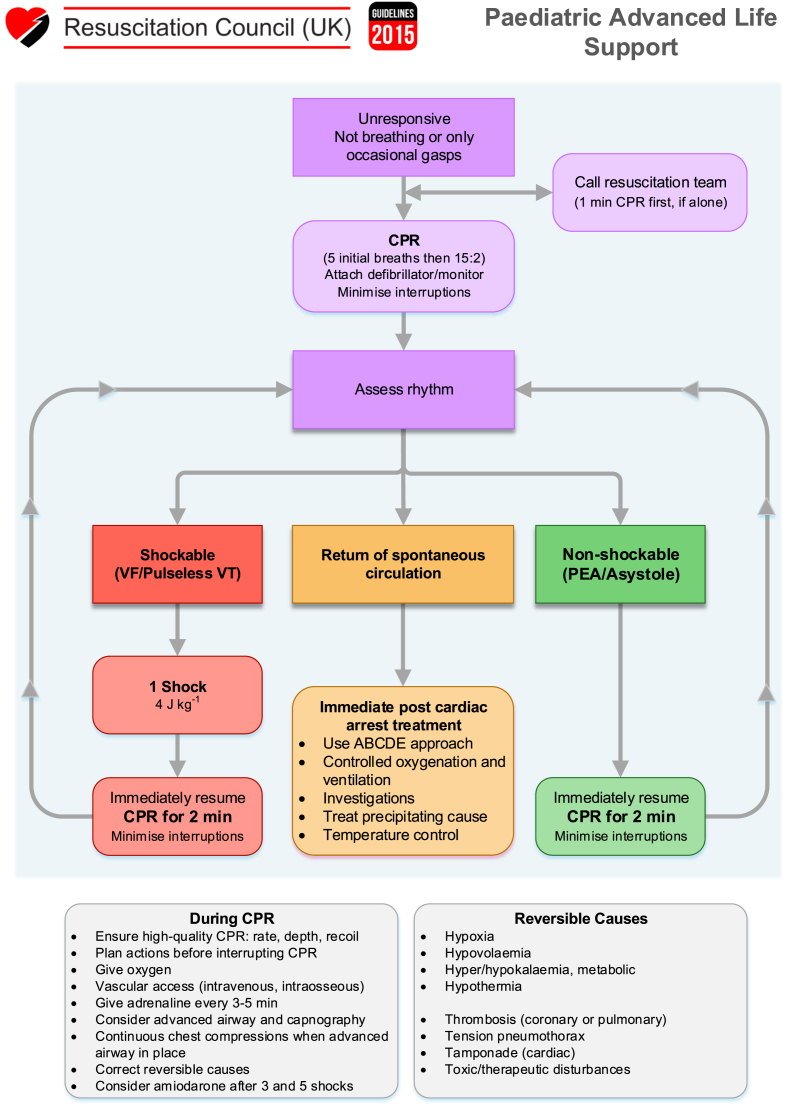
35 Pediatric Resuscitation
Pediatric cardiac arrest is most often due to respiratory failure or shock (asphyxia), unlike adult arrest which is usually primary cardiac (VF/VT). The most common preterminal rhythm in children is bradycardia progressing to PEA/asystole. Early recognition of respiratory distress and shock, before arrest occurs, is the most impactful intervention.
PALS Key Algorithms
| Rhythm | Management |
|---|---|
| Asystole / PEA | CPR (15:2 ratio with 2 rescuers); epinephrine 0.01 mg/kg (0.1 mL/kg of 1:10,000) IV/IO q3–5 min; identify and treat reversible causes (H’s and T’s) |
| VF / Pulseless VT | CPR + defibrillation 2 J/kg → 4 J/kg → max 10 J/kg; epinephrine q3–5 min; amiodarone 5 mg/kg (max 300 mg) or lidocaine 1 mg/kg |
| Bradycardia with poor perfusion | Oxygenation/ventilation first; if HR <60 despite adequate ventilation with O2: start CPR; epinephrine 0.01 mg/kg; atropine 0.02 mg/kg (min 0.1 mg) if vagal |
| SVT with pulses | Vagal maneuvers (ice to face in infants); adenosine 0.1 mg/kg rapid IV push (max 6 mg), may repeat at 0.2 mg/kg (max 12 mg); synchronized cardioversion 0.5–1 J/kg if unstable |
Weight-Based Equipment & Dosing
The Broselow tape (length-based resuscitation tape) provides rapid weight estimation and corresponding drug doses, equipment sizes, and defibrillation energy for children up to ~36 kg. Weight estimation formula: weight (kg) = (age in years × 2) + 8 for children 1–10 years.
Vascular Access
If IV access cannot be obtained within 60–90 seconds in a critically ill child, intraosseous (IO) access should be established. Preferred site: proximal tibia (1–2 cm below tibial tuberosity on medial flat surface). Alternative sites: distal tibia, distal femur, humeral head. Any medication or fluid that can be given IV can be given IO.
H’s: Hypovolemia (most common treatable cause), Hypoxia, Hydrogen ion (acidosis), Hypo/Hyperkalemia, Hypothermia, Hypoglycemia (especially in infants). T’s: Tension pneumothorax, Tamponade, Toxins/poisons, Thrombosis (pulmonary embolism), Trauma.
36 Non-Accidental Trauma
Child abuse is tragically common: ~700,000 confirmed cases annually in the U.S., with ~1,750 fatalities per year. Children <3 years are at highest risk. All healthcare providers are mandatory reporters in all 50 states — suspected abuse must be reported even without proof. Reporting in good faith is legally protected.
Red Flags for Non-Accidental Injury
| Finding | Significance |
|---|---|
| Injuries inconsistent with developmental stage | e.g., "fell off couch" in a pre-mobile infant |
| Changing/inconsistent history | Story changes between caregivers or over time |
| Delay in seeking care | Significant injury presented hours to days later |
| Patterned bruising | Belt marks, loop marks, bite marks, hand prints; bruises in various stages of healing |
| Bruising in non-mobile infants | "Those who don’t cruise rarely bruise" — any bruise in a pre-cruising infant is concerning |
| Specific fracture patterns | Classic metaphyseal lesions (corner/bucket-handle fractures), rib fractures (especially posterior), spiral fractures of long bones in non-ambulatory children, multiple fractures in different stages of healing |
| Retinal hemorrhages | Strongly associated with abusive head trauma (shaken baby syndrome); multi-layer, bilateral; rare in accidental injury |
| Subdural hematomas | Especially bilateral or interhemispheric; in combination with retinal hemorrhages = highly suspicious for abusive head trauma |
| Burns | Stocking/glove pattern (immersion), well-demarcated, circular (cigarette), pattern burns (iron, grill); sparing of flexion creases (child pulled limbs in) |
Workup
Skeletal survey (full body radiographic series) in all children <2 years with suspected abuse. Head CT for all children <6 months with suspected abuse (or any age with neurologic symptoms). Ophthalmologic exam for retinal hemorrhages. Labs: CBC, CMP, coagulation studies, lipase, urinalysis (to rule out bleeding disorders and assess for occult organ injury).
37 Foreign Body Ingestion & Poisoning
Foreign Body Ingestion
| Object | Urgency | Management |
|---|---|---|
| Button battery (disk/coin battery) | EMERGENT | If in esophagus: endoscopic removal within 2 hours (generates hydroxide at negative pole → liquefactive necrosis → esophageal perforation, aortoesophageal fistula, death). If past esophagus: may observe if asymptomatic, but close follow-up. Honey (sucralfate in some protocols) can mitigate injury pre-removal. |
| Magnets (≥2 or magnet + metal) | EMERGENT | Can attract through bowel walls → pressure necrosis, perforation, fistula. Endoscopic or surgical removal. |
| Coins | Moderate | Most common ingested foreign body. Esophageal: endoscopic removal if symptomatic or stuck >24 hours. Gastric/distal: observe, most pass spontaneously in 1–4 weeks. |
| Sharp objects (pins, bones) | High | Endoscopic removal if in esophagus or stomach (risk of perforation). If beyond reach of endoscope and asymptomatic, observe with serial radiographs. Surgical removal if signs of perforation. |
A button battery lodged in the esophagus can cause severe injury within 2 hours. It generates an electrical current and creates hydroxide at the negative pole, causing liquefactive necrosis. Complications include esophageal perforation, tracheoesophageal fistula, mediastinitis, and fatal aortoesophageal fistula. AP/lateral X-ray shows a "double ring" or "halo" sign (distinguishing it from a coin). Emergent endoscopic removal is mandatory. Do NOT induce vomiting.
Common Pediatric Poisonings
| Substance | Key Features | Management |
|---|---|---|
| Acetaminophen | Most common pediatric poisoning; toxic dose >150 mg/kg; liver failure if untreated; Rumack-Matthew nomogram guides treatment | N-acetylcysteine (NAC) within 8 hours of ingestion; 72-hour IV or 72-hour oral protocol |
| Iron | GI hemorrhage, metabolic acidosis, hepatotoxicity; abdominal X-ray may show pills; serum iron >500 mcg/dL = severe | Whole bowel irrigation; deferoxamine chelation if serum iron >500 or symptomatic |
| Lead | Chronic: developmental delay, behavior changes, abdominal pain, anemia, basophilic stippling; Acute: encephalopathy (>70 mcg/dL) | BLL 3.5–44: environmental investigation, follow-up. BLL 45–69: oral succimer (DMSA). BLL ≥70: IV CaNa2EDTA ± dimercaprol; hospitalize |
| Hydrocarbons | Aspiration pneumonitis is the main risk; do NOT induce emesis; CNS depression with high volatility | Supportive care; monitor for aspiration; do NOT give activated charcoal (risk of aspiration) |
| Caustics (alkali/acid) | Alkali: liquefactive necrosis (deeper injury). Acid: coagulative necrosis (self-limiting). No emesis, no charcoal, no NG tube | Endoscopy within 12–24 hours to assess injury grade; supportive care |
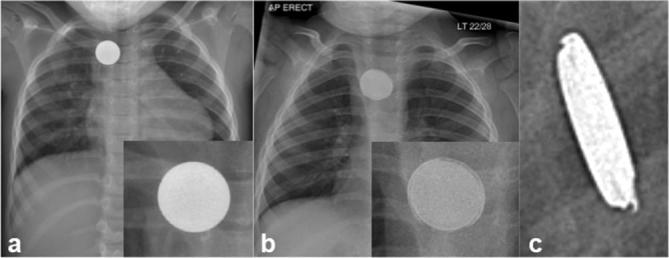
38 Pediatric Vital Signs by Age
| Age | Heart Rate (bpm) | Respiratory Rate | Systolic BP (mmHg) | Diastolic BP (mmHg) | Weight (kg, est.) |
|---|---|---|---|---|---|
| Premature | 120–170 | 40–70 | 55–75 | 35–45 | 1–2.5 |
| Newborn (0–28 days) | 120–160 | 30–60 | 60–76 | 30–45 | 2.5–4 |
| 1–6 months | 100–150 | 25–50 | 72–104 | 37–56 | 4–7 |
| 6–12 months | 90–140 | 25–40 | 72–104 | 37–56 | 7–10 |
| 1–3 years | 90–140 | 20–30 | 86–106 | 42–63 | 10–14 |
| 4–5 years | 80–120 | 20–25 | 89–112 | 46–72 | 14–20 |
| 6–9 years | 70–110 | 18–22 | 97–115 | 57–76 | 20–30 |
| 10–12 years | 60–100 | 16–20 | 102–120 | 61–80 | 30–45 |
| 13–18 years | 60–100 | 12–20 | 110–131 | 64–83 | 45–70 |
ETT Size by Age
| Age | Uncuffed ETT (mm ID) | Cuffed ETT (mm ID) |
|---|---|---|
| Premature | 2.5–3.0 | — |
| Newborn | 3.0–3.5 | 3.0 |
| 1–2 years | 4.0–4.5 | 3.5–4.0 |
| >2 years | (Age/4) + 4 | (Age/4) + 3.5 |
39 Classification Systems
APGAR Score
See Section 08 for full APGAR scoring table. Assessed at 1 and 5 minutes. Score 7–10 = reassuring; 4–6 = moderately depressed; 0–3 = severely depressed.
Tanner Staging
See Section 30 for full Tanner staging table. Stages I (prepubertal) through V (adult) for breast/genital and pubic hair development.
WHO Dehydration Classification
| Classification | Signs | Treatment Plan |
|---|---|---|
| No dehydration | Not enough signs to classify as some or severe dehydration | Plan A: ORS after each loose stool (10 mL/kg); continue feeding |
| Some dehydration (≥2 signs) | Restless/irritable, sunken eyes, drinks eagerly/thirsty, skin pinch goes back slowly | Plan B: ORS 75 mL/kg over 4 hours; reassess |
| Severe dehydration (≥2 signs) | Lethargic/unconscious, sunken eyes, unable to drink, skin pinch goes back very slowly (≥2 sec) | Plan C: IV Ringer’s lactate 100 mL/kg over 3–6 hours (age-dependent); NGT or ORS if IV unavailable |
Asthma Severity Classification
See Section 13 for full classification table (Intermittent, Mild Persistent, Moderate Persistent, Severe Persistent).
VUR Grading (International Reflux Study)
See Section 21 for full VUR grading table (Grades I–V based on VCUG findings).
Modified Bell Staging for NEC
See Section 24 for full NEC staging (Stage I = suspected, Stage II = definite with pneumatosis, Stage III = advanced with perforation).
Westley Croup Score
| Parameter | 0 | 1 | 2 | 3 | 5 |
|---|---|---|---|---|---|
| Chest wall retractions | None | Mild | Moderate | Severe | — |
| Stridor | None | With agitation | At rest | — | — |
| Cyanosis | None | — | — | — | With agitation / at rest (4/5) |
| Air entry | Normal | Decreased | Markedly decreased | — | — |
| Level of consciousness | Normal | — | — | — | Altered (5) |
40 Medications Master Table
Common Pediatric Antibiotics (Weight-Based Dosing)
| Drug | Dose | Route | Common Indication |
|---|---|---|---|
| Amoxicillin | 40–90 mg/kg/day ÷ BID-TID | PO | AOM (80–90 mg/kg/day), pharyngitis (50 mg/kg/day), pneumonia, UTI |
| Amoxicillin-clavulanate | 90 mg/kg/day of amoxicillin component ÷ BID | PO | Resistant AOM, sinusitis, bite wounds |
| Azithromycin | 10 mg/kg day 1 (max 500 mg), then 5 mg/kg (max 250 mg) days 2–5 | PO | Atypical pneumonia, pertussis, Chlamydia trachomatis |
| Cephalexin | 25–50 mg/kg/day ÷ BID-QID | PO | Skin/soft tissue, UTI, pharyngitis (penicillin-allergic) |
| Ceftriaxone | 50–100 mg/kg/day ÷ QD-BID (max 4 g/day) | IV/IM | Meningitis (100 mg/kg), pneumonia, UTI; avoid in neonates <28 days |
| Cefdinir | 14 mg/kg/day ÷ QD-BID (max 600 mg/day) | PO | AOM, sinusitis (penicillin-allergic); turns stools red (not blood) |
| TMP-SMX | 8–12 mg/kg/day of TMP component ÷ BID | PO | UTI, MRSA skin infections (in children >2 months) |
| Clindamycin | 30–40 mg/kg/day ÷ TID-QID | PO/IV | MRSA, bone/joint infections, dental infections |
Antipyretics & Analgesics
| Drug | Dose | Interval | Notes |
|---|---|---|---|
| Acetaminophen | 10–15 mg/kg/dose (max 75 mg/kg/day or 4 g/day) | q4–6h | Use from birth; no anti-inflammatory effect; hepatotoxic in overdose |
| Ibuprofen | 10 mg/kg/dose (max 40 mg/kg/day or 2.4 g/day) | q6–8h | Use from age ≥6 months; anti-inflammatory; avoid in dehydration (renal risk) |
Asthma Medications
| Drug | Dose | Notes |
|---|---|---|
| Albuterol (MDI) | 4–8 puffs via spacer q20min ×3 (acute); 2 puffs q4–6h PRN (maintenance) | Use spacer + mask in children <5 years |
| Albuterol (nebulizer) | 2.5 mg (<20 kg) or 5 mg (>20 kg) q20min ×3 (acute) | Continuous nebulization 0.5 mg/kg/hr for severe exacerbation |
| Ipratropium bromide | 0.25–0.5 mg nebulized q20min ×3 | Only for acute exacerbation; not for maintenance |
| Prednisolone/Prednisone | 1–2 mg/kg/day (max 60 mg) ×3–5 days | Oral; no taper needed for short course |
| Dexamethasone | 0.6 mg/kg ×1–2 doses (max 16 mg) | Alternative to 5-day prednisone course; better compliance |
| Montelukast | 4 mg (2–5 yr), 5 mg (6–14 yr), 10 mg (≥15 yr) QHS | LTRA; FDA black box warning for neuropsychiatric events |
PALS Resuscitation Drugs
| Drug | Dose | Indication |
|---|---|---|
| Epinephrine | 0.01 mg/kg (0.1 mL/kg of 1:10,000) IV/IO q3–5 min | Cardiac arrest, symptomatic bradycardia |
| Amiodarone | 5 mg/kg IV/IO (max 300 mg); may repeat ×2 | VF/pulseless VT refractory to defibrillation |
| Adenosine | 0.1 mg/kg (max 6 mg) rapid IV push; repeat: 0.2 mg/kg (max 12 mg) | SVT (with pulses) |
| Atropine | 0.02 mg/kg (min 0.1 mg, max 0.5 mg child / 1 mg adolescent) | Symptomatic bradycardia (vagal) |
| Dextrose | D10W: 5 mL/kg (neonates); D25W: 2–4 mL/kg (infants/children) | Hypoglycemia |
| Naloxone | 0.1 mg/kg IV/IM/IN (max 2 mg); may repeat | Opioid reversal |
| Calcium chloride 10% | 20 mg/kg (0.2 mL/kg) slow IV push | Hyperkalemia, calcium channel blocker overdose, hypocalcemia |
| Magnesium sulfate | 25–50 mg/kg IV (max 2 g) over 15–20 min | Torsades de pointes, severe refractory asthma |
41 Abbreviations Master List
42 Growth Charts & Screening Schedules
Well-Child Visit Schedule (AAP / Bright Futures)
| Visit | Age | Key Activities |
|---|---|---|
| Newborn | 3–5 days | Weight check, jaundice assessment, feeding evaluation, newborn screen results |
| 1 month | 1 month | Growth, HepB #2, feeding, development |
| 2 months | 2 months | DTaP #1, IPV #1, Hib #1, PCV #1, RV #1; developmental surveillance |
| 4 months | 4 months | DTaP #2, IPV #2, Hib #2, PCV #2, RV #2 |
| 6 months | 6 months | DTaP #3, Hib #3, PCV #3, HepB #3; start flu vaccine; introduce solids |
| 9 months | 9 months | Developmental screening (ASQ); developmental surveillance |
| 12 months | 12 months | MMR #1, Varicella #1, HepA #1, Hib booster, PCV booster; lead/Hb screen |
| 15 months | 15 months | DTaP #4; developmental surveillance |
| 18 months | 18 months | HepA #2; M-CHAT autism screen; developmental screening |
| 24 months | 24 months | M-CHAT autism screen; developmental screening; lead screen |
| 30 months | 30 months | Developmental screening |
| Annual visits | 3–21 years | Growth, BP (starting age 3), vision (3–5 yr), dental referral, anticipatory guidance, age-appropriate vaccines |
| 11–12 years | Preadolescent | Tdap, HPV, MenACWY; lipid screen (9–11 yr); depression screen starts at 12 |
Growth Chart Reference Points
| Milestone | Expected |
|---|---|
| Birth weight doubles | 4–5 months |
| Birth weight triples | 12 months |
| Birth weight quadruples | 24 months |
| Birth length increases 50% | 12 months |
| Birth length doubles | 4 years |
| Adult height = 2 × height at age 2 | Rough estimate |
| Head circumference at birth | ~35 cm (equals chest circumference) |
| Anterior fontanelle closes | 9–18 months (average 12–14 months) |
| Posterior fontanelle closes | 2–3 months |
References
- American Academy of Pediatrics. Bright Futures: Guidelines for Health Supervision of Infants, Children, and Adolescents, 4th ed. AAP, 2017.
- Kliegman RM, St Geme JW, et al. Nelson Textbook of Pediatrics, 21st ed. Elsevier, 2020.
- American Academy of Pediatrics, Subcommittee on Hyperbilirubinemia. "Management of Hyperbilirubinemia in the Newborn Infant 35 or More Weeks of Gestation." Pediatrics. 2004;114(1):297–316.
- Ralston SL, Lieberthal AS, et al. "Clinical Practice Guideline: The Diagnosis, Management, and Prevention of Bronchiolitis." Pediatrics. 2014;134(5):e1474–e1502.
- National Asthma Education and Prevention Program (NAEPP). "Expert Panel Report 4: Guidelines for the Diagnosis and Management of Asthma." NIH, 2020.
- McCrindle BW, Rowley AH, et al. "Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement from the American Heart Association." Circulation. 2017;135(17):e927–e999.
- Subcommittee on Urinary Tract Infection, AAP. "Reaffirmation of AAP Clinical Practice Guideline: The Diagnosis and Management of the Initial Urinary Tract Infection in Febrile Infants and Young Children 2–24 Months of Age." Pediatrics. 2016;138(6):e20163026.
- Du Pont-Thibodeau G, Bhatt DL, et al. "Pediatric Cardiac Arrest and Resuscitation." Pediatric Critical Care Medicine. 2021;22(1):e52–e66.
- Centers for Disease Control and Prevention. "Recommended Child and Adolescent Immunization Schedule, United States, 2024." MMWR. 2024.
- Wolfsdorf JI, Glaser N, et al. "ISPAD Clinical Practice Consensus Guidelines 2018: Diabetic Ketoacidosis and the Hyperglycemic Hyperosmolar State." Pediatric Diabetes. 2018;19(Suppl 27):155–177.
- Christian CW, Committee on Child Abuse and Neglect. "The Evaluation of Suspected Child Physical Abuse." Pediatrics. 2015;135(5):e1337–e1354.
- Du Toit G, Roberts G, et al. (LEAP Study). "Randomized Trial of Peanut Consumption in Infants at Risk for Peanut Allergy." N Engl J Med. 2015;372(9):803–813.
- Puopolo KM, Benitz WE, et al. "Management of Neonates Born at ≥35 0/7 Weeks’ Gestation with Suspected or Proven Early-Onset Bacterial Sepsis." Pediatrics. 2018;142(6):e20182894.
- Maenner MJ, Warren Z, et al. "Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years — ADDM Network, 2020." MMWR Surveill Summ. 2023;72(2):1–14.