PM&R
Every diagnosis, classification system, electrodiagnostic finding, functional assessment, rehabilitation protocol, procedure, device, medication, and management strategy in one place.
01 Neuroanatomy for Rehabilitation
A thorough understanding of motor and sensory neuroanatomy is the foundation of PM&R practice. Localizing lesions to upper motor neuron (UMN) versus lower motor neuron (LMN) pathways, understanding dermatomes and myotomes, and recognizing spinal cord tract organization are essential for accurate diagnosis, prognosis, and rehabilitation planning across stroke, spinal cord injury, traumatic brain injury, and peripheral nerve disorders.
Motor Pathways
The corticospinal tract (CST) is the primary motor pathway. It originates from the primary motor cortex (Brodmann area 4), premotor cortex (area 6), and supplementary motor area. Axons descend through the corona radiata, posterior limb of the internal capsule, cerebral peduncles, basis pontis, and medullary pyramids. At the cervicomedullary junction, ~85% of fibers decussate (pyramidal decussation) to form the lateral corticospinal tract, which controls voluntary distal limb movements contralaterally. The remaining ~15% continue ipsilaterally as the anterior corticospinal tract, controlling axial/proximal muscles bilaterally. The rubrospinal tract assists with upper extremity flexor tone. The reticulospinal tracts (pontine and medullary) modulate muscle tone, posture, and locomotion — pontine reticulospinal facilitates extensors; medullary reticulospinal facilitates flexors. The vestibulospinal tract maintains upright posture and balance via extensor facilitation.
Sensory Pathways
The dorsal column–medial lemniscus (DCML) pathway transmits proprioception, vibration, and fine (discriminative) touch. First-order neurons enter the spinal cord and ascend ipsilaterally in the fasciculus gracilis (lower body, below T6) and fasciculus cuneatus (upper body, above T6) to synapse in the medulla (nucleus gracilis and cuneatus). Second-order neurons decussate as the internal arcuate fibers and ascend in the medial lemniscus to the ventral posterolateral (VPL) nucleus of the thalamus. Third-order neurons project to the primary somatosensory cortex (S1, Brodmann areas 3, 1, 2). The spinothalamic tract (STT) transmits pain, temperature, and crude touch. First-order neurons synapse in the dorsal horn (laminae I, II, V); second-order neurons decussate within 1–2 spinal levels via the anterior white commissure and ascend contralaterally to the VPL thalamus.
UMN vs LMN Lesions
| Feature | UMN Lesion | LMN Lesion |
|---|---|---|
| Muscle tone | Increased (spasticity, clasp-knife) | Decreased (flaccidity) |
| Reflexes | Hyperreflexia, clonus | Hyporeflexia or areflexia |
| Babinski sign | Positive (upgoing toe) | Negative (downgoing toe) |
| Atrophy | Mild, disuse atrophy (late) | Significant, early denervation atrophy |
| Fasciculations | Absent | Present |
| Distribution | Pyramidal pattern (extensors in UE, flexors in LE weak) | Specific nerve root or peripheral nerve distribution |
| EMG findings | Normal or reduced recruitment; no fibrillations | Fibrillations, positive sharp waves, reduced recruitment |
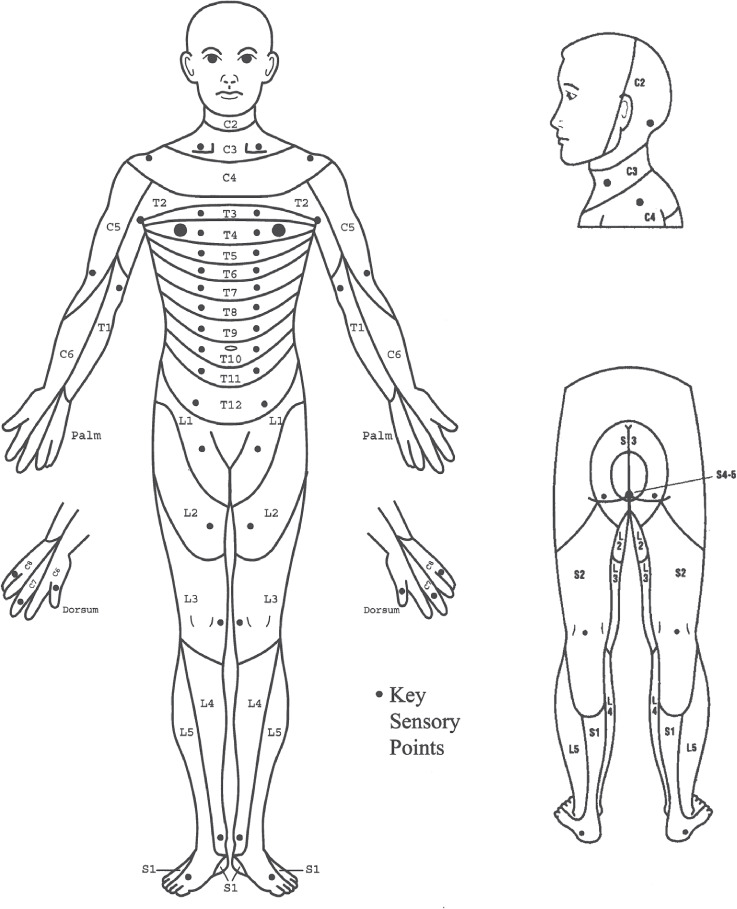
Dermatomes & Myotomes — Key Levels
| Level | Dermatome (Sensory) | Myotome (Motor) | Reflex |
|---|---|---|---|
| C5 | Lateral arm (regimental badge area) | Elbow flexors (biceps, brachialis) | Biceps (C5–C6) |
| C6 | Lateral forearm, thumb, index finger | Wrist extensors (ECRL, ECRB) | Brachioradialis (C5–C6) |
| C7 | Middle finger | Elbow extensors (triceps) | Triceps (C7–C8) |
| C8 | Medial forearm, ring & little finger | Finger flexors (FDP to middle finger) | — |
| T1 | Medial arm (axilla) | Small hand muscles (abductor digiti minimi) | — |
| T4 | Nipple line | — | — |
| T10 | Umbilicus | — | — |
| L2 | Anterior thigh | Hip flexors (iliopsoas) | — |
| L3 | Medial thigh, knee | Knee extensors (quadriceps) | Patellar (L3–L4) |
| L4 | Medial leg, medial malleolus | Ankle dorsiflexors (tibialis anterior) | Patellar (L3–L4) |
| L5 | Lateral leg, dorsum of foot, great toe | Great toe extensors (EHL) | — |
| S1 | Lateral foot, small toe, sole | Ankle plantarflexors (gastrocnemius, soleus) | Achilles (S1–S2) |
| S2–S4 | Perianal region (saddle area) | Bladder/bowel sphincters | Bulbocavernosus (S3–S4) |
02 Musculoskeletal Anatomy Essentials
PM&R physicians must understand musculoskeletal anatomy as it relates to functional impairment, biomechanics, and rehabilitation planning. Key areas include the shoulder complex, spine, hip, knee, and hand/wrist — the most common sites of musculoskeletal complaints encountered in rehabilitation practice.
Shoulder Complex
The shoulder has the greatest range of motion of any joint, achieved through four articulations: glenohumeral (primary), acromioclavicular, sternoclavicular, and scapulothoracic. The rotator cuff consists of four muscles: supraspinatus (abduction initiation, most commonly torn, suprascapular nerve C5–C6), infraspinatus (external rotation, suprascapular nerve C5–C6), teres minor (external rotation, axillary nerve C5–C6), and subscapularis (internal rotation, upper/lower subscapular nerves C5–C7). The mnemonic SITS captures the four muscles. The subacromial space is bounded superiorly by the acromion and coracoacromial ligament; narrowing causes impingement syndrome.
Spinal Anatomy
The vertebral column consists of 7 cervical, 12 thoracic, 5 lumbar, 5 fused sacral, and 3–4 coccygeal vertebrae. The intervertebral disc has an inner nucleus pulposus (gelatinous, high water content, shock absorption) and outer annulus fibrosus (concentric collagen lamellae). Disc herniations typically occur posterolaterally where the annulus is thinnest (posterior longitudinal ligament reinforces midline). In the lumbar spine, the L4–L5 and L5–S1 levels account for >90% of disc herniations. The spinal cord terminates as the conus medullaris at L1–L2 in adults; below this, the cauda equina (nerve roots L2–S5) traverses the thecal sac.
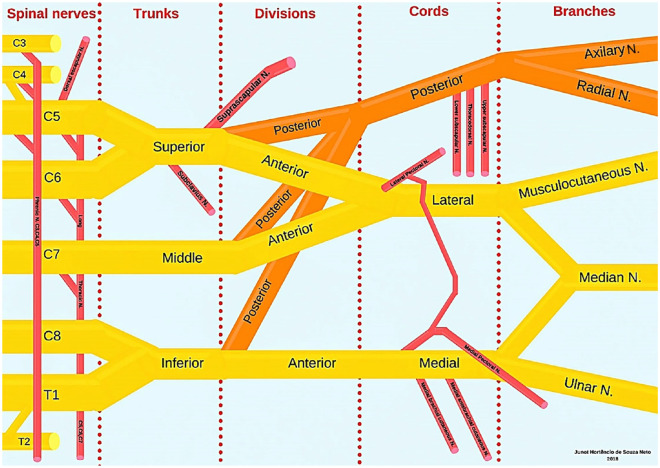
Brachial Plexus
The brachial plexus (C5–T1) is organized as: Roots → Trunks (upper C5–C6, middle C7, lower C8–T1) → Divisions (anterior and posterior) → Cords (lateral, posterior, medial — named by position relative to the axillary artery) → Branches (terminal nerves). Key terminal branches: musculocutaneous nerve (lateral cord → biceps, brachialis), axillary nerve (posterior cord → deltoid, teres minor), radial nerve (posterior cord → triceps, wrist/finger extensors), median nerve (lateral + medial cord → forearm flexors, thenar muscles), ulnar nerve (medial cord → intrinsic hand muscles, FDP to ring/small fingers).
Lumbosacral Plexus
The lumbar plexus (L1–L4) gives rise to the femoral nerve (L2–L4: quadriceps, hip flexion, knee extension, anterior thigh sensation) and obturator nerve (L2–L4: hip adductors, medial thigh sensation). The sacral plexus (L4–S3) gives rise to the sciatic nerve (the largest nerve in the body), which divides into the tibial nerve (plantarflexion, toe flexion, sole of foot sensation) and common peroneal (fibular) nerve (dorsiflexion, eversion, lateral leg and dorsum of foot sensation). The common peroneal nerve is highly vulnerable at the fibular head; injury causes foot drop.
03 The PM&R Evaluation & ICF Model
The PM&R evaluation is unique among medical specialties in its focus on function rather than solely on disease. The International Classification of Functioning, Disability, and Health (ICF), adopted by the World Health Organization in 2001, provides the conceptual framework that underlies all rehabilitation assessment and planning.
ICF Framework
| ICF Domain | Definition | Example (Stroke Patient) |
|---|---|---|
| Body Functions & Structures | Physiological functions and anatomical parts | Left hemiparesis, spasticity, hemianopia |
| Activity | Execution of a task by an individual | Cannot dress independently, difficulty walking |
| Participation | Involvement in life situations | Unable to return to work, cannot drive |
| Environmental Factors | Physical, social, attitudinal environment | Home accessibility, caregiver support, insurance |
| Personal Factors | Individual background features | Age, motivation, coping style, education |
Historical Terminology (Nagi Model / ICIDH)
The older Nagi model used related but distinct terms: Pathology (underlying disease) → Impairment (loss of function at organ level, e.g., weakness) → Functional Limitation (restriction in performing an activity, e.g., cannot walk) → Disability (inability to fulfill social role, e.g., cannot work). The WHO ICIDH (1980) used Impairment → Disability → Handicap. The ICF replaced this linear model with a biopsychosocial, interactive framework.
Functional Assessment
The PM&R evaluation systematically assesses: mobility (bed mobility, transfers, ambulation, wheelchair skills, community mobility), self-care (feeding, grooming, bathing, dressing upper/lower body, toileting), communication (expression, comprehension, reading, writing), cognition (orientation, attention, memory, problem-solving, safety awareness), psychosocial (adjustment, mood, motivation, social interaction), and vocational/avocational (work capacity, leisure, driving). Standardized outcome measures (FIM, Barthel Index, etc.) quantify these domains and track progress.
Rehabilitation Settings
| Setting | Requirements | Intensity | Typical Conditions |
|---|---|---|---|
| Inpatient Rehabilitation Facility (IRF) | Requires physician supervision, 3 hours/day of therapy (PT, OT, SLP), medical necessity, multidisciplinary team | Highest | Stroke, SCI, TBI, amputation, major joint replacement |
| Skilled Nursing Facility (SNF) | Skilled nursing/therapy needs; not able to tolerate IRF intensity | Moderate | Deconditioned patients, post-surgical, hip fracture |
| Long-Term Acute Care (LTAC) | Average LOS ≥25 days; complex medical needs | Variable | Ventilator weaning, complex wounds, multi-organ failure |
| Home Health | Homebound status; skilled therapy/nursing need | Low-moderate | Post-acute continued therapy |
| Outpatient | Community-dwelling; able to travel | Variable | Ongoing therapy, chronic conditions |
04 Key Terminology & Abbreviations
Core PM&R Terminology
| Term | Definition |
|---|---|
| Spasticity | Velocity-dependent increase in tonic stretch reflexes (UMN pattern); distinct from rigidity (velocity-independent) |
| Contracture | Fixed shortening of muscle, tendon, or joint capsule causing loss of ROM |
| Heterotopic Ossification (HO) | Abnormal formation of mature lamellar bone in soft tissues, most common after TBI, SCI, burns |
| Neurogenic Bladder | Bladder dysfunction due to neurological injury; classified as UMN (spastic/reflex) or LMN (areflexic/flaccid) |
| Orthosis | Externally applied device to support, align, prevent, or correct deformity and improve function |
| Prosthesis | Device replacing an absent body part (limb, digit) |
| Neuroplasticity | Ability of the nervous system to reorganize structurally and functionally in response to experience or injury |
| Functional Electrical Stimulation (FES) | Application of electrical current to produce functional muscle contraction in paralyzed muscles |
Essential Abbreviations
| Abbreviation | Meaning |
|---|---|
| ADL | Activities of Daily Living |
| IADL | Instrumental Activities of Daily Living |
| ROM | Range of Motion (AROM = active, PROM = passive) |
| MMT | Manual Muscle Testing (0–5 scale) |
| FIM | Functional Independence Measure |
| ASIA | American Spinal Injury Association |
| ISNCSCI | International Standards for Neurological Classification of SCI |
| GCS | Glasgow Coma Scale |
| PTA | Post-Traumatic Amnesia |
| NCS | Nerve Conduction Study |
| EMG | Electromyography |
| CMAP | Compound Motor Action Potential |
| SNAP | Sensory Nerve Action Potential |
| AFO | Ankle-Foot Orthosis |
| KAFO | Knee-Ankle-Foot Orthosis |
| TLSO | Thoracolumbosacral Orthosis |
| CIC | Clean Intermittent Catheterization |
| AD | Autonomic Dysreflexia |
| DVT | Deep Vein Thrombosis |
| HO | Heterotopic Ossification |
| ITB | Intrathecal Baclofen |
| TENS | Transcutaneous Electrical Nerve Stimulation |
| NMES | Neuromuscular Electrical Stimulation |
| CP | Cerebral Palsy |
| GMFCS | Gross Motor Function Classification System |
| TUG | Timed Up and Go |
| 6MWT | 6-Minute Walk Test |
Manual Muscle Testing (MMT) Scale
| Grade | Description | Clinical Finding |
|---|---|---|
| 0 | Zero | No visible or palpable contraction |
| 1 | Trace | Visible or palpable contraction, no joint movement |
| 2 | Poor | Full ROM with gravity eliminated |
| 3 | Fair | Full ROM against gravity, no added resistance |
| 4 | Good | Full ROM against gravity with moderate resistance |
| 5 | Normal | Full ROM against gravity with maximum resistance |
05 Stroke Syndromes & Deficits
Stroke is the leading cause of adult disability and the most common diagnosis in inpatient rehabilitation facilities. Recognizing the vascular territory involved predicts the pattern of deficits and guides rehabilitation planning. Each stroke syndrome produces a characteristic constellation of motor, sensory, visual, language, and cognitive impairments.
Major Stroke Syndromes
| Syndrome | Artery | Key Deficits |
|---|---|---|
| MCA (Middle Cerebral Artery) | MCA (most common stroke territory) | Contralateral hemiparesis (face & arm > leg), contralateral hemianesthesia, contralateral homonymous hemianopia; Dominant: Broca (inferior frontal) or Wernicke (superior temporal) aphasia; Non-dominant: hemispatial neglect, anosognosia, constructional apraxia |
| ACA (Anterior Cerebral Artery) | ACA | Contralateral leg > arm weakness, contralateral leg sensory loss, urinary incontinence (medial frontal), abulia (bilateral ACA), alien hand syndrome, transcortical motor aphasia (dominant) |
| PCA (Posterior Cerebral Artery) | PCA | Contralateral homonymous hemianopia with macular sparing, visual agnosia, alexia without agraphia (dominant), prosopagnosia (bilateral), memory impairment (hippocampal), thalamic pain syndrome |
| Basilar Artery | Basilar | Locked-in syndrome (ventral pons — quadriplegia, anarthria, preserved vertical eye movements and consciousness), coma, bilateral motor/sensory deficits, cranial nerve palsies |
| Lateral Medullary (Wallenberg) | PICA or vertebral artery | Ipsilateral: facial pain/temperature loss, Horner syndrome, ataxia, dysphagia, hoarseness; Contralateral: body pain/temperature loss. NO motor weakness |
| Medial Medullary | Anterior spinal artery branches | Contralateral hemiparesis (arm & leg), contralateral proprioception/vibration loss, ipsilateral tongue weakness (CN XII) |
| Weber Syndrome | Midbrain (PCA penetrators) | Ipsilateral CN III palsy with contralateral hemiparesis |
Lacunar Stroke Syndromes
Lacunar infarcts are small (<15 mm) subcortical strokes caused by lipohyalinosis of small penetrating arteries, most commonly in the basal ganglia, thalamus, internal capsule, and pons. They account for ~25% of ischemic strokes.
| Lacunar Syndrome | Location | Presentation |
|---|---|---|
| Pure motor hemiparesis | Posterior limb internal capsule or basis pontis | Contralateral face, arm, leg weakness; no sensory/visual/cognitive deficits |
| Pure sensory stroke | VPL thalamus | Contralateral numbness/paresthesias; no motor deficits |
| Ataxic hemiparesis | Posterior limb internal capsule or pons | Contralateral weakness with ipsilateral cerebellar-type ataxia |
| Sensorimotor stroke | Thalamocapsular | Combined motor and sensory deficits contralaterally |
| Dysarthria–clumsy hand | Basis pontis or genu of internal capsule | Dysarthria with ipsilateral hand clumsiness |
06 Acute Stroke Rehab & Motor Recovery
Acute Rehabilitation Criteria
Stroke patients are candidates for inpatient rehabilitation facility (IRF) admission when they are medically stable, can tolerate at least 3 hours/day of multidisciplinary therapy (PT, OT, and often SLP), have identifiable functional goals, and require physician supervision. The IRF-PAI (Patient Assessment Instrument) documents admission function using Section GG (self-care and mobility items). CMS requires that 60% of IRF patients have qualifying diagnoses (the "60% rule"), including stroke as one of the 13 qualifying conditions.
Brunnstrom Stages of Motor Recovery (1–7)
The Brunnstrom stages describe the stereotyped sequence of motor recovery following stroke, from flaccidity through synergy patterns to isolated voluntary movement:
| Stage | Description | Clinical Findings |
|---|---|---|
| 1 | Flaccidity | No voluntary movement; hypotonia; areflexia |
| 2 | Emerging spasticity; synergy patterns begin | Minimal voluntary movement; basic limb synergies may appear as associated reactions |
| 3 | Spasticity peaks; voluntary synergy patterns | Patient can voluntarily produce synergy patterns but cannot deviate from them. UE flexor synergy: shoulder abduction/external rotation, elbow flexion, forearm supination, wrist/finger flexion. LE extensor synergy: hip extension/adduction/IR, knee extension, ankle plantarflexion/inversion |
| 4 | Spasticity begins to decline; some out-of-synergy movements | Movement combinations deviating from synergy emerge: hand behind back, arm to 90° flexion with elbow extended, forearm pronation/supination with elbow at 90° |
| 5 | Spasticity further decreases; isolated movements | Movements increasingly independent of synergy patterns; more complex movement combinations possible |
| 6 | Spasticity essentially absent; near-normal coordination | Individual joint movements performed with good coordination and near-normal speed; synergy patterns no longer dominate |
| 7 | Normal motor function | Full speed, coordination, and agility restored (rarely achieved after significant stroke) |
Post-Stroke Shoulder Pain
Hemiplegic shoulder pain affects 30–70% of stroke patients and is a major barrier to rehabilitation. Common causes include: subluxation (due to flaccid deltoid/rotator cuff muscles, gravitational pull on the humerus — most common early post-stroke), spasticity (subscapularis and pectoralis spasticity causing internal rotation/adduction contracture), adhesive capsulitis (frozen shoulder from immobility), complex regional pain syndrome (CRPS) type I (previously shoulder-hand syndrome — pain, edema, autonomic changes), rotator cuff injury, and brachial plexopathy. Management includes positioning (support the arm on a lapboard or armrest), ROM exercises, shoulder slings (controversial — may limit function), suprascapular nerve block, and spasticity treatment.
Stroke Rehabilitation Evidence-Based Interventions
Key evidence-based approaches include: constraint-induced movement therapy (CIMT) — restraining the unaffected limb to force use of the paretic limb for 6+ hours/day (requires some wrist/finger extension); body-weight-supported treadmill training; functional electrical stimulation (FES) for dorsiflexion and wrist extension; robotic-assisted therapy; mirror therapy for improving motor function and reducing pain/neglect; and task-specific repetitive practice (highest level of evidence for motor learning).
AHA/ASA 2016 Guidelines for Adult Stroke Rehabilitation07 Spasticity Management
Spasticity is defined as a velocity-dependent increase in tonic stretch reflexes resulting from UMN lesion, manifesting as increased resistance to passive stretch. It is one of the most common and functionally limiting sequelae of stroke, SCI, TBI, MS, and cerebral palsy. Spasticity can be functionally helpful (aids standing/transfers in SCI) or harmful (causes pain, contracture, impaired hygiene, positioning difficulties).
Modified Ashworth Scale (MAS)
| Grade | Description |
|---|---|
| 0 | No increase in muscle tone |
| 1 | Slight increase in tone; catch and release OR minimal resistance at end of ROM when the affected part(s) moved in flexion or extension |
| 1+ | Slight increase in tone; catch followed by minimal resistance throughout the remainder (<50%) of the ROM |
| 2 | More marked increase in tone through most of the ROM, but affected part(s) easily moved |
| 3 | Considerable increase in tone; passive movement difficult |
| 4 | Affected part(s) rigid in flexion or extension |
Oral Antispasticity Medications
| Medication | Mechanism | Starting Dose | Max Dose | Key Side Effects |
|---|---|---|---|---|
| Baclofen | GABA-B agonist (spinal level) | 5 mg TID | 80–120 mg/day | Sedation, weakness, withdrawal seizures (taper!) |
| Tizanidine | Central alpha-2 agonist | 2 mg TID | 36 mg/day | Sedation, dry mouth, hepatotoxicity (monitor LFTs), hypotension |
| Dantrolene | Peripheral — blocks Ca++ release from sarcoplasmic reticulum | 25 mg daily | 400 mg/day | Hepatotoxicity (monitor LFTs q3 months), weakness, diarrhea |
| Diazepam | GABA-A agonist (enhances Cl− conductance) | 2 mg BID | 40–60 mg/day | Sedation, dependence, cognitive impairment, respiratory depression |
Botulinum Toxin for Spasticity
Botulinum toxin (onabotulinumtoxinA [Botox], abobotulinumtoxinA [Dysport], incobotulinumtoxinA [Xeomin]) blocks acetylcholine release at the neuromuscular junction, producing focal, reversible chemodenervation. Onset 3–7 days, peak effect 2–6 weeks, duration 3–6 months. Injection is guided by EMG, electrical stimulation, or ultrasound. Maximum total dose of onabotulinumtoxinA per treatment session is typically 400–600 U. Common upper extremity targets: biceps (100–200 U), brachialis (50–100 U), brachioradialis (50–100 U), flexor carpi radialis (25–100 U), flexor carpi ulnaris (25–75 U), flexor digitorum superficialis (25–75 U per head). Common lower extremity targets: gastrocnemius (100–200 U per head), soleus (75–100 U), tibialis posterior (50–100 U).
Intrathecal Baclofen (ITB)
ITB delivers baclofen directly to the intrathecal space via a programmable pump, achieving effective CSF concentrations at 1/100th of the oral dose, thereby reducing systemic side effects. Indications include severe spasticity refractory to oral medications and botulinum toxin, particularly in SCI and CP. A screening trial (50–100 mcg bolus via lumbar puncture) is performed first; a ≥1-point reduction on the Ashworth Scale constitutes a positive trial. Maintenance doses typically range from 100–900 mcg/day. Pump reservoir requires refills every 1–6 months.
Intrathecal baclofen withdrawal is a life-threatening emergency caused by abrupt cessation (pump malfunction, catheter kink/disconnect, missed refill). Signs: rebound spasticity, hyperthermia (up to 42°C), altered mental status, seizures, rhabdomyolysis, multi-organ failure. Can mimic malignant hyperthermia or neuroleptic malignant syndrome. Management: restore ITB delivery immediately; oral/enteral baclofen (high doses), IV benzodiazepines, cyproheptadine, dantrolene; ICU admission. Mortality is significant if untreated.
08 Dysphagia, Aphasia & Neglect
Dysphagia Assessment
Dysphagia occurs in 37–78% of acute stroke patients and is a major risk factor for aspiration pneumonia, malnutrition, and dehydration. Bedside assessment includes the water swallow test, oral motor exam, and cough/voice quality evaluation. Instrumental assessments are the gold standard:
| Test | Method | Advantages | Limitations |
|---|---|---|---|
| VFSS (Videofluoroscopic Swallow Study) | Modified barium swallow under fluoroscopy; evaluates all phases of swallowing | Gold standard; visualizes entire swallow mechanism; allows diet/posture trials | Radiation exposure; cannot be done bedside; brief assessment window |
| FEES (Fiberoptic Endoscopic Evaluation of Swallowing) | Flexible nasopharyngoscope passed transnasally; evaluates pharyngeal phase | Portable (bedside); no radiation; can assess secretion management; repeatable | Cannot visualize oral phase or moment of swallow ("white-out"); invasive |
The Penetration-Aspiration Scale (PAS) grades severity from 1 (material does not enter airway) to 8 (material enters airway, passes below vocal folds, and no effort made to eject — silent aspiration). Scores ≥6 indicate aspiration.
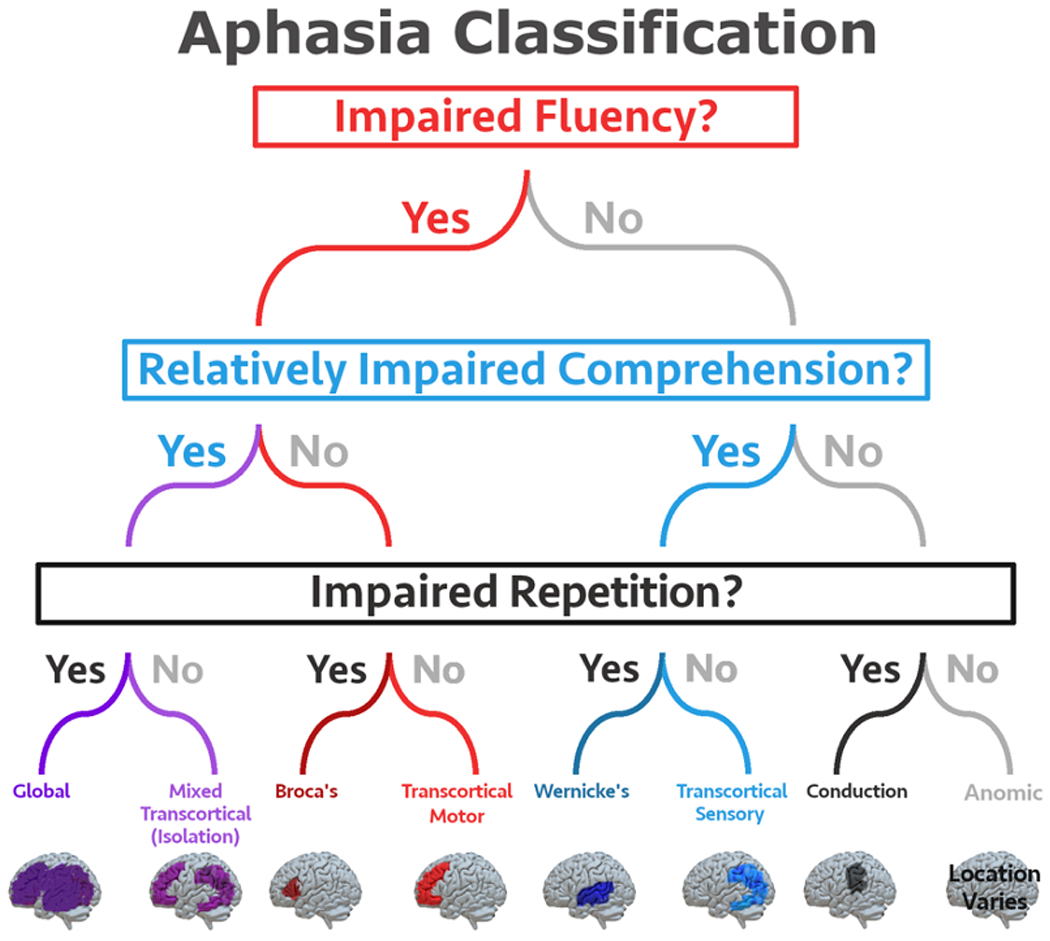
Aphasia Types
| Aphasia Type | Fluency | Comprehension | Repetition | Lesion Location |
|---|---|---|---|---|
| Broca (Expressive) | Non-fluent | Intact | Impaired | Inferior frontal gyrus (Brodmann area 44/45) |
| Wernicke (Receptive) | Fluent (but paraphasic, jargon) | Impaired | Impaired | Superior temporal gyrus (Brodmann area 22) |
| Global | Non-fluent | Impaired | Impaired | Large MCA territory (frontal + temporal) |
| Conduction | Fluent | Intact | Impaired (disproportionate) | Arcuate fasciculus |
| Transcortical Motor | Non-fluent | Intact | Intact | Anterior/superior to Broca area (ACA/MCA watershed) |
| Transcortical Sensory | Fluent | Impaired | Intact | Posterior to Wernicke area (PCA/MCA watershed) |
| Mixed Transcortical | Non-fluent | Impaired | Intact | Bilateral watershed zones |
| Anomic | Fluent | Intact | Intact | Various (angular gyrus, temporal); mildest form |
Hemispatial Neglect
Hemispatial neglect is failure to attend, perceive, or respond to stimuli on the contralesional side, most commonly left neglect from right (non-dominant) hemisphere lesions involving the right parietal lobe, temporoparietal junction, or frontal lobe. Types include: personal neglect (unawareness of contralesional body), peripersonal neglect (near space), extrapersonal neglect (far space). Associated findings: anosognosia (unawareness of deficit), anosodiaphoria (indifference to deficit), extinction (failure to detect contralesional stimulus during bilateral simultaneous stimulation). Assessment tools: line bisection, cancellation tasks, clock drawing. Treatment: visual scanning training, prism adaptation, limb activation strategies.
09 ASIA/ISNCSCI Classification
The International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI), developed by the American Spinal Injury Association (ASIA), is the universal standard for classifying SCI severity. It determines the neurological level of injury and the completeness of injury through systematic motor and sensory examination.
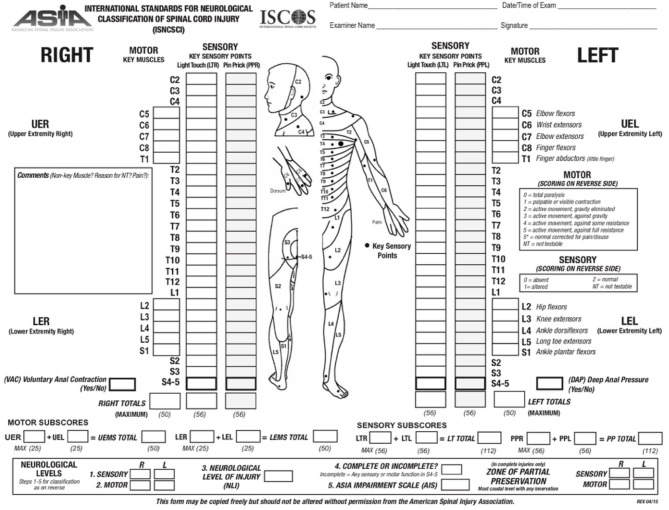
ASIA Impairment Scale (AIS)
| Grade | Classification | Definition |
|---|---|---|
| A | Complete | No sensory or motor function preserved in sacral segments S4–S5 (no sacral sparing) |
| B | Sensory Incomplete | Sensory but not motor function preserved below the neurological level AND includes sacral segments S4–S5 (light touch or pin prick at S4–S5, or deep anal pressure); no motor function >3 levels below motor level |
| C | Motor Incomplete | Motor function preserved below the neurological level AND more than half of key muscles below the neurological level have a muscle grade <3 (less than antigravity) |
| D | Motor Incomplete | Motor function preserved below the neurological level AND at least half of key muscles below the neurological level have a muscle grade ≥3 (antigravity or better) |
| E | Normal | Sensory and motor function normal in all segments (patient may still have abnormalities on exam — prior SCI with full recovery) |
Neurological Level Determination
The neurological level of injury (NLI) is the most caudal segment with normal sensory and motor function on both sides. Sensory level is determined by testing light touch and pin prick at 28 dermatome key points (C2–S4/5) on each side, scored 0 (absent), 1 (impaired), or 2 (normal). Motor level is determined by testing 10 key muscles bilaterally (C5–T1, L2–S1), scored 0–5 using the MMT scale. The motor level is the most caudal level with at least grade 3 strength, provided all muscles above are grade 5.
Key Muscles for Motor Level Testing
| Level | Key Muscle | Action Tested |
|---|---|---|
| C5 | Elbow flexors (biceps, brachialis) | Elbow flexion |
| C6 | Wrist extensors (ECRL, ECRB) | Wrist extension |
| C7 | Elbow extensors (triceps) | Elbow extension |
| C8 | Finger flexors (FDP to middle finger) | Finger flexion at DIP |
| T1 | Small finger abductor (ADM) | Small finger abduction |
| L2 | Hip flexors (iliopsoas) | Hip flexion |
| L3 | Knee extensors (quadriceps) | Knee extension |
| L4 | Ankle dorsiflexors (tibialis anterior) | Ankle dorsiflexion |
| L5 | Long toe extensors (EHL) | Great toe extension |
| S1 | Ankle plantarflexors (gastrocnemius, soleus) | Ankle plantarflexion |
Functional Expectations by SCI Level
| Level | Key Abilities | Mobility | Expected FIM |
|---|---|---|---|
| C4 | Neck control, scapular elevation; dependent for all ADLs | Power wheelchair with head/chin/sip-puff control | Dependent |
| C5 | Elbow flexion, shoulder abduction; feeding with setup using adaptive equipment | Power wheelchair with hand control; dependent transfers | Modified dependent |
| C6 | Wrist extension (tenodesis grasp); self-feeding, some dressing, driving with hand controls | Manual wheelchair on flat surfaces; sliding board transfers with assist | Modified independent for some ADLs |
| C7 | Elbow extension (triceps); independent transfers, most ADLs independent | Manual wheelchair; independent transfers | Modified independent |
| T1–T9 | Full UE function; trunk control varies; independent ADLs | Manual wheelchair; independent community mobility | Independent (wheelchair level) |
| T10–L2 | Good trunk stability; may ambulate with KAFOs + crutches (exercise only) | Manual wheelchair primary; limited therapeutic ambulation | Independent |
| L3–L4 | Hip flexion, knee extension; community ambulation with AFOs | AFOs + forearm crutches or canes | Independent |
| L5–S1 | Near-normal LE function; community ambulation | AFOs or shoe inserts; near-normal gait | Independent |
10 Incomplete SCI Syndromes
Incomplete SCI syndromes result from partial spinal cord damage affecting specific tracts while sparing others. Each produces a characteristic pattern of motor and sensory deficits based on the anatomy of the affected region of the spinal cord cross-section.
| Syndrome | Mechanism / Anatomy | Motor Deficits | Sensory Deficits | Prognosis |
|---|---|---|---|---|
| Central Cord Syndrome | Hyperextension injury in elderly with cervical spondylosis; damage to central gray matter and medial CST | UE > LE weakness (arms weaker than legs — because cervical fibers are medial in CST); hand function most impaired | Variable; "cape" distribution pain/temperature loss if anterior commissure involved | Best prognosis of incomplete syndromes; LE and bladder recover first, then UE; hand function recovers last |
| Brown-Séquard Syndrome | Hemisection of the cord (penetrating trauma, tumor, disc herniation) | Ipsilateral motor paralysis (CST) below lesion | Ipsilateral loss of proprioception/vibration (DCML); contralateral loss of pain/temperature (STT) 1–2 levels below | Second-best prognosis; >90% regain ambulatory function |
| Anterior Cord Syndrome | Anterior spinal artery occlusion, flexion injuries, disc herniation; damages anterior 2/3 of cord | Complete motor paralysis below lesion (bilateral CST) | Bilateral loss of pain/temperature (STT); PRESERVED proprioception/vibration (dorsal columns spared) | Worst prognosis; <10% achieve functional motor recovery |
| Posterior Cord Syndrome | Rare; posterior spinal artery occlusion, B12 deficiency (subacute combined degeneration), tabes dorsalis | Motor function preserved | Bilateral loss of proprioception/vibration (dorsal columns); pain/temperature intact | Good motor prognosis; sensory ataxia may be significant |
| Cauda Equina Syndrome | Compression of nerve roots below conus medullaris (L1–L2); disc herniation, tumor, abscess | LMN pattern: flaccid, areflexic lower extremities; asymmetric | Saddle anesthesia (S2–S4/5); radicular pain | Depends on timing of decompression; surgical emergency |
| Conus Medullaris Syndrome | Damage to the sacral cord segments (S2–S5) at the L1–L2 vertebral level | LMN bladder/bowel; symmetric LE weakness | Saddle anesthesia; early bladder/bowel/sexual dysfunction | Poor for bladder/bowel recovery |
Cauda equina syndrome is a surgical emergency requiring urgent decompression (ideally within 24–48 hours). Red flags: bilateral sciatica, saddle anesthesia, urinary retention or incontinence, fecal incontinence, progressive bilateral lower extremity weakness. MRI is the imaging modality of choice. Delayed decompression correlates with permanent neurological deficits, particularly bladder dysfunction.
11 Autonomic Dysreflexia & Cardiovascular
Autonomic dysreflexia (AD) is a potentially life-threatening medical emergency occurring in patients with SCI at T6 or above. A noxious stimulus below the level of injury triggers an uninhibited sympathetic response, causing severe hypertension (SBP may exceed 250–300 mmHg) that can lead to stroke, seizures, MI, or death. The parasympathetic response above the lesion causes bradycardia, flushing, sweating, and nasal congestion above the level of injury.
Most common triggers: bladder distension (most common — 75–85%), bowel impaction, pressure injuries, UTI, ingrown toenails, tight clothing, DVT, fractures, sexual activity.
Immediate management:
- Sit the patient upright (orthostatic drop reduces BP)
- Loosen all restrictive clothing, abdominal binders, stockings
- Check and drain the bladder (straight catheterize if Foley blocked; if Foley in place, irrigate or replace)
- If bladder not the cause, perform digital rectal exam (use lidocaine jelly first) and disimpact bowel
- Monitor BP every 2–5 minutes
- If BP remains elevated after removing trigger: nifedipine 10 mg bite-and-swallow or nitropaste 1–2 inches (remove when BP normalizes); IV hydralazine or labetalol for refractory cases
- Do NOT use beta-blockers alone (unopposed alpha-mediated vasoconstriction)
Cardiovascular Complications in SCI
Patients with SCI above T6 lose supraspinal sympathetic control, leading to: neurogenic shock (acute SCI — hypotension with bradycardia due to loss of sympathetic tone and unopposed vagal tone; treat with vasopressors and atropine), orthostatic hypotension (common in tetraplegia; manage with compression stockings, abdominal binder, midodrine, fludrocortisone, slow position changes), and loss of cardiac sympathetic innervation (T1–T4 — maximum HR limited to ~110–120 bpm, reduced cardiac output with exercise).
DVT/PE Prophylaxis in SCI
SCI patients have an extremely high DVT risk (40–80% without prophylaxis in the first 3 months). The highest risk period is 2–12 weeks post-injury. Standard prophylaxis: LMWH (enoxaparin 30 mg BID or 40 mg daily) initiated within 72 hours if no contraindication; combined with pneumatic compression devices and compression stockings. Duration: minimum 8 weeks for incomplete SCI, 12 weeks for complete SCI, or until mobilization. Screening with duplex ultrasound is recommended. IVC filter considered only if anticoagulation is contraindicated.
Pulmonary embolism is a leading cause of death in SCI patients during the first year. SCI patients may present atypically: dyspnea may be limited by intercostal paralysis, tachycardia may be absent (loss of cardiac sympathetics). Maintain a high index of suspicion for any SCI patient with new-onset dyspnea, oxygen desaturation, chest pain, or hemodynamic instability. CT pulmonary angiography is the diagnostic standard. Treat with systemic anticoagulation per standard PE protocols.
12 Neurogenic Bladder, Bowel & Skin
Neurogenic Bladder
| Feature | UMN (Spastic/Reflex) Bladder | LMN (Areflexic/Flaccid) Bladder |
|---|---|---|
| Lesion level | Above conus medullaris (suprasacral) | At or below conus/cauda equina |
| Detrusor function | Overactive (spastic, involuntary contractions) | Acontractile (no reflex contractions) |
| Sphincter | Dyssynergic (DSD — detrusor-sphincter dyssynergia: bladder contracts against closed sphincter) | Incompetent (denervated, low tone) |
| Bladder capacity | Small (reduced compliance) | Large (overdistended, high compliance) |
| Clinical pattern | Small volume, frequency, urgency, incontinence, high pressures | Overflow incontinence, large residuals, low pressures |
| Primary management | CIC + anticholinergics (oxybutynin 5 mg TID or tolterodine); botulinum toxin into detrusor | CIC on timed schedule; Valsalva/Credé maneuver (caution: can worsen reflux) |
| Monitoring | Annual urodynamics, annual renal ultrasound; keep bladder pressures <40 cm H₂O | Annual urodynamics, renal ultrasound |
Neurogenic Bowel
| Feature | UMN (Reflex) Bowel | LMN (Areflexic) Bowel |
|---|---|---|
| Lesion | Supraconal (above conus) | Conal/cauda equina |
| Anal tone | Intact or increased; intact bulbocavernosus reflex | Decreased or absent; absent bulbocavernosus reflex |
| Pattern | Stool retention; constipation; reflex evacuation possible | Stool incontinence; constipation; no reflex evacuation |
| Management | Timed bowel program with digital rectal stimulation + suppositories (bisacodyl or glycerin); high-fiber diet; stool softeners | Timed manual evacuation; bulk-forming agents; digital disimpaction; colostomy may be needed |
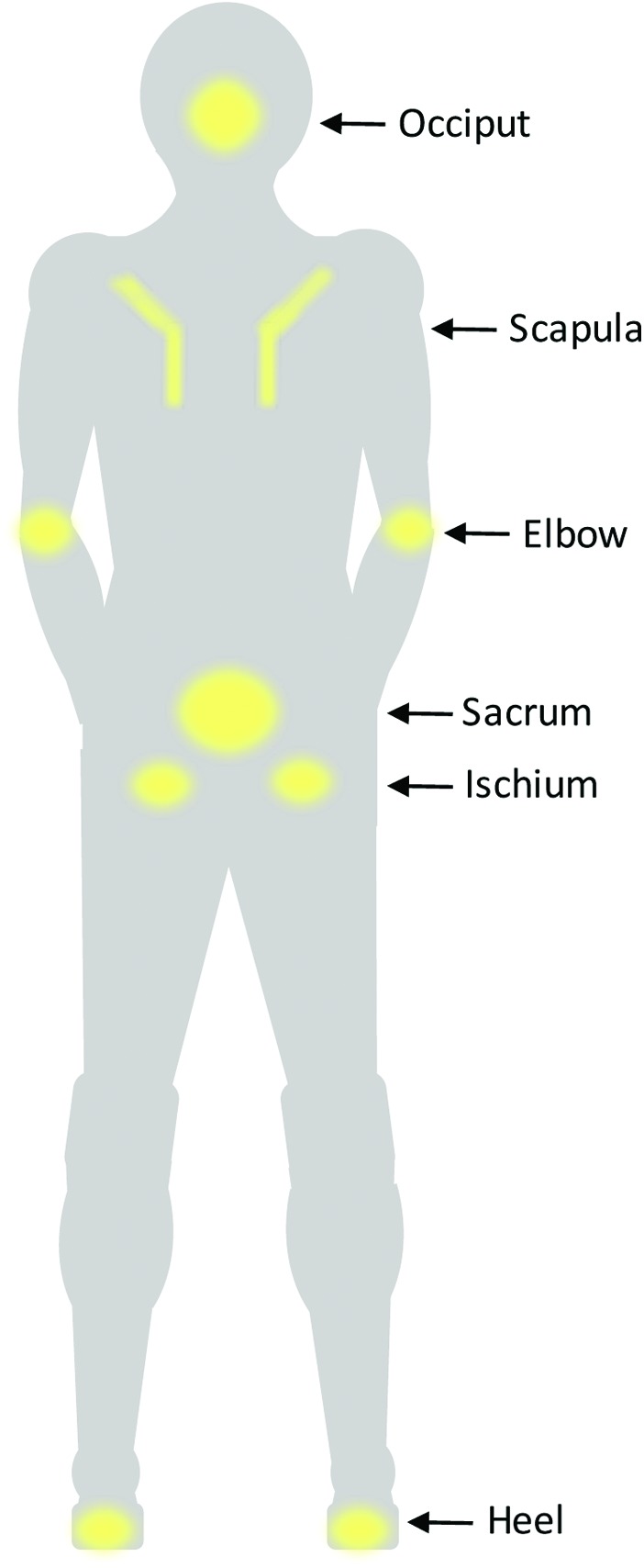
Pressure Injury Prevention
SCI patients are at extremely high risk for pressure injuries (formerly pressure ulcers/decubitus ulcers) due to sensory loss, immobility, moisture, and poor nutrition. Common sites: sacrum, ischial tuberosities (seated), trochanters, heels, occiput, and malleoli. Prevention strategies: pressure relief every 15–30 minutes in wheelchair (weight shifts), turning every 2 hours in bed, appropriate support surface (pressure-redistributing mattress), skin inspection daily, adequate nutrition (albumin, prealbumin), moisture management.
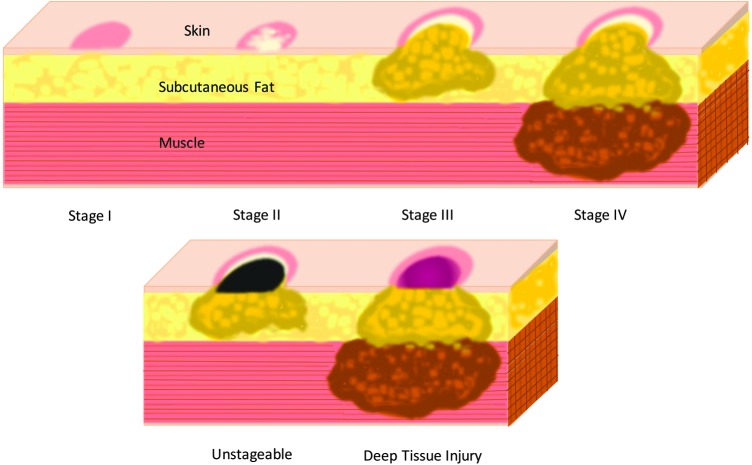
| Stage | Description |
|---|---|
| Stage 1 | Intact skin with non-blanchable erythema |
| Stage 2 | Partial-thickness skin loss (epidermis/dermis); blister or shallow open ulcer |
| Stage 3 | Full-thickness skin loss; subcutaneous fat visible; bone/tendon/muscle NOT exposed |
| Stage 4 | Full-thickness tissue loss with exposed bone, tendon, or muscle |
| Unstageable | Base covered by slough (yellow/tan/gray/green/brown) and/or eschar (tan/brown/black) |
| Deep Tissue Injury (DTI) | Intact or non-intact skin with localized area of persistent deep red, maroon, purple discoloration or blood-filled blister |
13 SCI Complications
Heterotopic Ossification (HO)
HO is the formation of mature lamellar bone in periarticular soft tissues, occurring in 10–53% of SCI patients, typically 1–4 months post-injury. Most common location: hip (most frequent), followed by knee, shoulder, elbow. Clinical presentation: localized swelling, warmth, decreased ROM, low-grade fever. Diagnosis: elevated alkaline phosphatase (earliest lab marker); triple-phase bone scan (most sensitive early imaging); X-rays show mature HO (delayed 2–4 weeks). Treatment: NSAIDS (indomethacin 75 mg/day for prophylaxis), etidronate disodium, ROM exercises (gentle — aggressive ROM may worsen), surgical resection (only after HO maturation, ~18 months; confirmed by normal alkaline phosphatase and "cold" bone scan). Radiation therapy is used for prophylaxis post-resection.
Syringomyelia (Post-Traumatic)
Post-traumatic syringomyelia occurs in 3–4% of SCI patients, may present months to years post-injury. A fluid-filled cavity (syrinx) develops within the spinal cord, typically extending rostrally from the injury site. Symptoms: ascending sensory level, new pain, increased spasticity, progressive weakness, loss of previously recovered function, new autonomic changes. Diagnosis: MRI (best imaging). Treatment: observation if asymptomatic; surgical shunting or untethering if progressive.
Respiratory Complications
Respiratory failure is the leading cause of mortality in acute SCI. The diaphragm is innervated by the phrenic nerve (C3, C4, C5 — "C3, 4, 5 keeps the diaphragm alive"). Injuries above C3 require mechanical ventilation. C3–C5 injuries may have partial diaphragm function. Intercostal muscles (T1–T12) and abdominal muscles (T6–T12) are needed for cough, forced expiration, and secretion clearance. SCI patients develop a restrictive ventilatory pattern with reduced vital capacity, reduced cough strength, and increased mucus retention. Management: assisted cough techniques (manual or mechanical insufflation-exsufflation), incentive spirometry, pulmonary toilet, possible phrenic nerve stimulation for high cervical SCI.
Thermoregulation
SCI patients with injuries above T6 have impaired thermoregulation (poikilothermia) below the level of injury due to loss of sympathetic control of cutaneous blood flow and sweating. They are susceptible to both hypothermia and hyperthermia. Environmental temperature management is essential.
Sexual Function & Fertility
UMN lesions (above S2–S4): reflex erections preserved, psychogenic erections lost; ejaculation impaired. LMN lesions (S2–S4): reflex erections lost, psychogenic may be preserved; ejaculation impaired. Fertility is significantly impaired in men with SCI due to ejaculatory dysfunction and poor semen quality. Treatment: PDE5 inhibitors (sildenafil) for erectile dysfunction; penile vibratory stimulation or electroejaculation for fertility.
Paralyzed Veterans of America Clinical Practice Guidelines for SCI14 TBI Classification & GCS
Traumatic brain injury (TBI) is a leading cause of death and disability worldwide, with approximately 2.87 million emergency department visits, hospitalizations, and deaths annually in the US. Classification is based on severity (GCS), mechanism (penetrating vs closed), pathology (focal vs diffuse), and associated complications.
Glasgow Coma Scale (GCS)
| Component | Response | Score |
|---|---|---|
| Eye Opening (E) | Spontaneous | 4 |
| To voice/command | 3 | |
| To pressure/pain | 2 | |
| None | 1 | |
| Verbal Response (V) | Oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Incomprehensible sounds | 2 | |
| None | 1 | |
| Motor Response (M) | Obeys commands | 6 |
| Localizes to pain | 5 | |
| Withdrawal (flexion) | 4 | |
| Abnormal flexion (decorticate) | 3 | |
| Extension (decerebrate) | 2 | |
| None | 1 |
TBI Severity Classification
| Severity | GCS | LOC Duration | PTA Duration |
|---|---|---|---|
| Mild (mTBI/Concussion) | 13–15 | <30 minutes | <24 hours |
| Moderate | 9–12 | 30 min – 24 hours | 1–7 days |
| Severe | 3–8 | >24 hours | >7 days |
Post-Traumatic Amnesia (PTA)
PTA is the period of impaired continuous memory formation following TBI, from the moment of injury until the patient demonstrates consistent orientation and ability to form new memories. It is the single best predictor of long-term outcome after TBI. Assessment: Galveston Orientation and Amnesia Test (GOAT) — score ≥75 on two consecutive days indicates PTA resolution.
| PTA Duration | Projected Outcome |
|---|---|
| <1 hour | Good recovery expected |
| 1–24 hours | Good recovery likely |
| 1–7 days | Good recovery possible; some residual deficits |
| 1–4 weeks | Fair recovery; significant cognitive deficits likely |
| >4 weeks | Poor prognosis; likely persistent severe disability |
15 Rancho Los Amigos Scale & Recovery
Rancho Los Amigos Levels of Cognitive Functioning (Revised Scale, Levels I–X)
| Level | Description | Behavioral Characteristics | Rehab Approach |
|---|---|---|---|
| I | No Response | Complete absence of observable change in behavior to any stimuli | Sensory stimulation program; prevent complications |
| II | Generalized Response | Inconsistent, non-purposeful, non-specific reactions to stimuli; responses may be physiologic (changes in HR, RR, diaphoresis) or gross body movements | Sensory stimulation; family education |
| III | Localized Response | Specific but inconsistent responses directly related to stimulus type (turns toward sound, pulls at tubes, follows simple commands inconsistently) | Sensory stimulation; structured environment |
| IV | Confused–Agitated | Heightened state of activity; severely confused; bizarre, non-purposeful behavior; agitation; combativeness; unable to process environmental information; no short-term memory; verbalizations often incoherent | Safety; low-stimulation environment; consistent routine; redirection; avoid restraints; medications if needed (see agitation management) |
| V | Confused–Inappropriate, Non-Agitated | Alert, not agitated; responds to simple commands fairly consistently; highly distractible; memory severely impaired; inappropriate verbalization; no new learning; requires maximum structure | Structured tasks; repetition; cueing; supervised ADLs |
| VI | Confused–Appropriate | Goal-directed behavior with cueing; follows simple directions consistently; memory improving; aware of self and family; emerging awareness of deficits; some carryover of new learning | Increase independence with structure; functional task training; community reintegration planning |
| VII | Automatic–Appropriate | Oriented to setting; performs daily routine automatically with minimal confusion; robot-like, poor insight; shallow recall; poor judgment/problem-solving; requires supervision for safety | Increase demands; metacognitive strategies; pre-vocational tasks |
| VIII | Purposeful–Appropriate (Stand-By Assistance) | Oriented; recalls and integrates past and recent events; new learning with stand-by assistance; acknowledges impairments; depression/irritability common; reduced stress tolerance | Community reintegration; vocational rehab; compensatory strategies |
| IX | Purposeful–Appropriate (Stand-By Assistance on Request) | Independently shifts between tasks; uses assistive memory devices; aware of and monitors impairments; depression/irritability may continue; low frustration tolerance in social situations | Advanced community skills; driving evaluation; return to work/school |
| X | Purposeful–Appropriate (Modified Independent) | Able to handle multiple tasks simultaneously; independently uses compensatory strategies; may require more time or periodic breaks; anticipates impact of impairments; social interactions appropriate | Vocational/academic reentry; ongoing counseling; long-term follow-up |
Agitation Management (Rancho Level IV)
Agitation in TBI (Rancho IV) is a transient phase of recovery, not a behavioral disorder. Environmental management is first-line: low stimulation, consistent staff, quiet room, minimize restraints (worsen agitation), establish predictable routines, redirect rather than confront. Pharmacological management when safety is at risk:
| Medication | Dose | Notes |
|---|---|---|
| Amantadine | 100 mg BID (morning and noon) | Dopaminergic; improves arousal and speed of recovery; strongest evidence in TBI |
| Beta-blockers (propranolol, pindolol) | Propranolol 10–60 mg TID | Reduce autonomic arousal; useful for aggression |
| Buspirone | 5–15 mg TID | Anxiolytic; minimal sedation |
| Trazodone | 25–100 mg HS | Useful for insomnia/agitation; serotonergic |
| Valproic acid | 250–500 mg BID | Mood stabilizer for aggression; monitor levels |
Avoid medications that impair neuroplasticity and cognitive recovery: benzodiazepines (impair memory, reduce neuroplasticity), typical antipsychotics (haloperidol — dopamine blockade impairs recovery; use only for imminent danger), phenytoin for chronic seizure prophylaxis (>7 days; impairs cognition), anticholinergics (impair memory). If antipsychotic needed, use atypical (quetiapine at lowest dose). First-line for agitation: amantadine, beta-blockers, buspirone.
16 TBI Complications & Management
Post-Traumatic Seizures
Seizures after TBI are classified by timing: immediate (<24 hours — impact seizures), early (1–7 days), and late (>7 days — post-traumatic epilepsy). Risk factors for late seizures: penetrating injury, intracranial hemorrhage, depressed skull fracture, cortical contusion, early seizures, prolonged PTA.
Prophylaxis with phenytoin or levetiracetam is indicated for the first 7 days after severe TBI (GCS ≤10) to prevent early seizures. Beyond 7 days, prophylactic anticonvulsants have NOT been shown to prevent late post-traumatic epilepsy and should be discontinued (phenytoin also impairs cognitive recovery). Levetiracetam is preferred over phenytoin for prophylaxis due to better cognitive side-effect profile. Late seizures require treatment with standard antiepileptic medications.
Hydrocephalus
Post-traumatic hydrocephalus (communicating type, from impaired CSF absorption) occurs in 1–8% of TBI patients. Suspect when recovery plateaus or declines. Triad (similar to NPH): gait disturbance, cognitive decline, urinary incontinence. Diagnosis: CT/MRI showing ventriculomegaly out of proportion to atrophy; confirm with CSF tap test (improvement after removing 30–50 mL CSF). Treatment: VP shunt.
Neuroendocrine Dysfunction
Pituitary dysfunction occurs in 25–50% of moderate-to-severe TBI patients due to the pituitary's vulnerable location and blood supply. Anterior pituitary: GH deficiency (most common chronic deficiency), hypogonadism, hypothyroidism, adrenal insufficiency (most dangerous acutely). Posterior pituitary: diabetes insipidus (DI — polyuria, hypernatremia; common acutely, usually transient) and SIADH (hyponatremia — most common cause of hyponatremia in TBI). Screen with morning cortisol, TSH, free T4, testosterone/estradiol, IGF-1 at 3–6 months and 12 months post-injury.
Heterotopic Ossification in TBI
HO occurs in 10–20% of TBI patients, most commonly at the hip (most common), followed by elbow, shoulder, and knee. More common in patients with prolonged coma. Detection and management are identical to SCI-associated HO (see Section 13). The hip is at particular risk for development of clinically significant HO limiting ROM for sitting and ambulation.
Paroxysmal Sympathetic Hyperactivity (PSH)
Previously termed "sympathetic storming," "dysautonomia," or "diencephalic seizures." Occurs in 15–33% of severe TBI patients. Characterized by paroxysmal episodes of tachycardia, hypertension, tachypnea, hyperthermia, diaphoresis, and posturing (extensor or flexor). Episodes may be triggered by stimulation (noxious or non-noxious). Treatment: minimize triggers, beta-blockers (propranolol), bromocriptine, gabapentin, benzodiazepines, morphine, dantrolene. Intrathecal baclofen may be considered for refractory cases.
17 Concussion & Return to Activity
Sport-Related Concussion (mTBI)
Concussion is a mild TBI (GCS 13–15) caused by biomechanical forces. It is a functional rather than structural injury — standard neuroimaging is typically normal. Diagnosis is clinical. Core features: headache (most common symptom), dizziness, confusion, amnesia, balance problems, visual disturbance, sensitivity to light/noise, cognitive slowing, emotional lability. Loss of consciousness occurs in <10% of concussions. Symptoms typically resolve within 7–10 days in adults and 2–4 weeks in children.
Graduated Return-to-Sport Protocol (Consensus Statement on Concussion in Sport, 2023)
| Stage | Activity | Objective | Minimum Duration |
|---|---|---|---|
| 1 | Symptom-limited activity | Relative rest; daily activities that do not provoke symptoms | 24–48 hours |
| 2 | Light aerobic exercise | Walking, swimming, stationary cycling at <70% max HR; no resistance training | 24 hours |
| 3 | Sport-specific exercise | Running drills, skating drills; no head impact activities | 24 hours |
| 4 | Non-contact training drills | Progression to complex drills; may add resistance training | 24 hours |
| 5 | Full-contact practice | Following medical clearance; normal training activities | 24 hours |
| 6 | Return to sport | Normal game play | — |
Minimum 24 hours at each step; if symptoms recur, drop back to previous asymptomatic stage. Children/adolescents: return to learn (school) before return to sport. No same-day return to play. Medical clearance required before full-contact practice.
Post-Concussion Syndrome
Persistent symptoms beyond expected recovery time (typically >2–4 weeks in adults). Symptoms: headache, dizziness, cognitive difficulties, fatigue, irritability, sleep disturbance, anxiety, depression. Risk factors: female sex, history of prior concussions, migraine history, psychiatric history, prolonged initial symptoms. Management: active rehabilitation (aerobic exercise below symptom threshold), vestibular therapy, vision therapy, cervicogenic headache treatment, cognitive behavioral therapy; avoid prolonged rest (>48 hours of strict rest is no longer recommended).
Amsterdam Consensus Statement on Concussion in Sport (2023)18 Low Back Pain & Spinal Disorders
Red Flags for Low Back Pain
- Cauda equina syndrome: saddle anesthesia, urinary retention/incontinence, fecal incontinence, bilateral LE weakness
- Cancer: history of cancer, unexplained weight loss, age >50 with new-onset LBP, pain at rest, night pain
- Infection: fever, IV drug use, recent procedure, immunosuppression, night pain
- Fracture: significant trauma, osteoporosis, prolonged corticosteroid use, age >70
- Abdominal aortic aneurysm: pulsatile abdominal mass, age >60, vascular risk factors
Imaging Indications
Imaging is NOT indicated for acute non-specific low back pain (<6 weeks) without red flags. Indications for early imaging include: red flag symptoms (above), progressive neurological deficit, failure to improve after 6 weeks of conservative care. MRI is the modality of choice for evaluating disc herniation, spinal stenosis, infection, and malignancy. X-rays for suspected fracture, spondylolisthesis, or instability. CT when MRI is contraindicated.
McKenzie Classification (Mechanical Diagnosis and Therapy)
| Syndrome | Characteristics | Treatment |
|---|---|---|
| Derangement | Most common (>70%); symptoms change with repeated movements or sustained positions; directional preference present | Repeated movements in the directional preference (often extension); centralization indicates favorable prognosis |
| Dysfunction | Pain occurs at end-range only due to shortened/scarred tissue; no change in pain pattern with repeated movements | Stretching/remodeling at end-range to restore normal tissue length |
| Postural | Pain from sustained postures only (sitting, slouching); no pain with movement; typically younger patients | Postural correction; ergonomic education |
Cervical Radiculopathy & Myelopathy
Cervical radiculopathy presents with dermatomal arm pain, numbness, and weakness in a nerve root distribution (most common: C6 — thumb/index, C7 — middle finger). Special tests: Spurling test (head extension + lateral flexion + axial compression reproduces radicular symptoms), upper limb tension test (ULTT). Most cases (75–90%) resolve with conservative management (NSAIDs, PT, cervical epidural steroid injection).
Cervical myelopathy is a surgical condition caused by spinal cord compression (spondylosis, disc herniation, OPLL). UMN findings: gait instability (earliest finding), hand clumsiness, hyperreflexia, Hoffman sign, Babinski sign, Lhermitte sign (electric sensation with neck flexion). Surgery (decompression) is indicated for moderate-to-severe or progressive myelopathy.
Lumbar Spinal Stenosis
Narrowing of the spinal canal (central) or neural foramina (lateral/foraminal). Most common cause: degenerative spondylosis (age >60). Classic presentation: neurogenic claudication — bilateral leg pain/heaviness/weakness with walking or prolonged standing, relieved by sitting or forward flexion (opening the canal). Distinguish from vascular claudication (pain with exercise, relieved by standing still, normal spine flexion). The "shopping cart sign" (relief with forward flexion while pushing a cart) suggests spinal stenosis. Treatment: PT (flexion-based exercises), epidural steroid injections, surgical decompression/laminectomy for refractory cases.
19 Peripheral Nerve Injuries
Seddon Classification
| Type | Pathology | Motor/Sensory Loss | EMG Findings | Recovery |
|---|---|---|---|---|
| Neurapraxia | Focal myelin damage; axon intact; no Wallerian degeneration | Conduction block at lesion site; motor/sensory loss distal to lesion | Conduction block on NCS; no denervation potentials on EMG | Complete; weeks to months (remyelination); best prognosis |
| Axonotmesis | Axon disrupted; endoneurium intact; Wallerian degeneration distal to lesion | Complete motor/sensory loss distal to lesion | Fibrillations/PSWs on EMG (after 2–3 weeks); reduced/absent CMAPs and SNAPs | Good; axon regenerates along intact endoneurial tubes at ~1 mm/day (1 inch/month) |
| Neurotmesis | Complete nerve disruption (axon + connective tissue); Wallerian degeneration | Complete motor/sensory loss distal to lesion | Same as axonotmesis acutely; no recovery on serial EMG | No spontaneous recovery; requires surgical repair |
Sunderland Classification (Expanded)
| Grade | Structures Damaged | Equivalent Seddon | Recovery |
|---|---|---|---|
| I | Myelin only | Neurapraxia | Complete; weeks to months |
| II | Axon + myelin; endoneurium intact | Axonotmesis | Good; regeneration along intact endoneurial tubes |
| III | Axon + myelin + endoneurium; perineurium intact | Axonotmesis (severe) | Variable; intrafascicular fibrosis may impede recovery |
| IV | Axon + myelin + endoneurium + perineurium; epineurium intact | Neurotmesis (partial) | Poor without surgery; neuroma-in-continuity |
| V | Complete transection of entire nerve trunk | Neurotmesis (complete) | No recovery without surgical repair |
Common Peripheral Nerve Injuries
| Nerve | Common Site of Injury | Motor Deficit | Sensory Deficit |
|---|---|---|---|
| Radial nerve | Spiral groove of humerus (Saturday night palsy, humeral fracture) | Wrist drop, finger drop (cannot extend wrist/fingers/thumb) | First dorsal web space |
| Ulnar nerve | Elbow (cubital tunnel); wrist (Guyon canal) | Claw hand (ring/small finger), weak grip, intrinsic hand weakness | Small finger, ulnar half of ring finger |
| Median nerve | Wrist (carpal tunnel); elbow (pronator syndrome) | Weak thumb opposition/abduction (thenar atrophy); weak forearm pronation (if at elbow) | Palmar thumb, index, middle, radial ring finger |
| Common peroneal | Fibular head (leg crossing, casts, knee surgery) | Foot drop (weak dorsiflexion, eversion) | Lateral leg, dorsum of foot |
| Axillary nerve | Shoulder dislocation, proximal humeral fracture | Weak deltoid (shoulder abduction) | Regimental badge area |
| Long thoracic nerve | Traction injury (carrying heavy loads) | Scapular winging (serratus anterior) | None |
20 Upper Extremity Rehabilitation
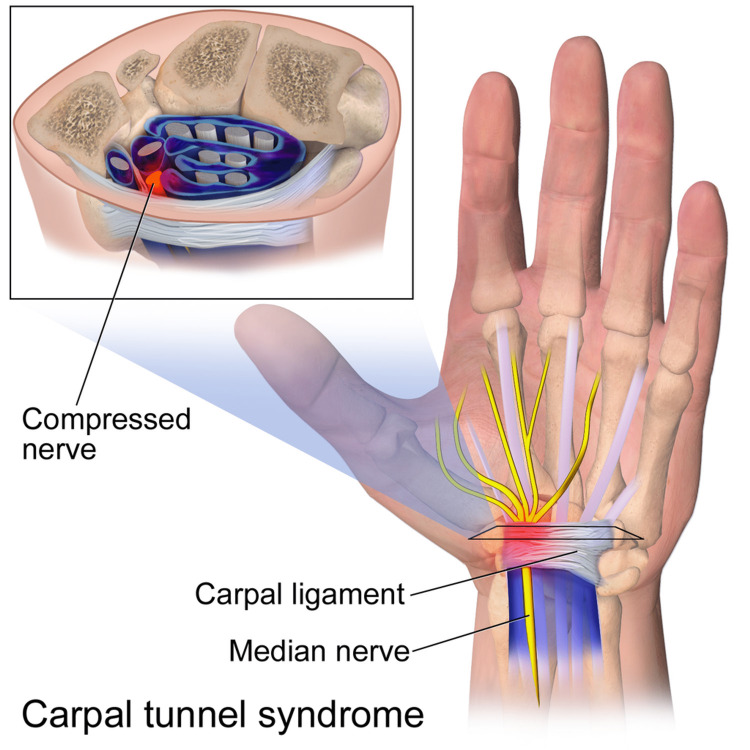
Carpal Tunnel Syndrome (CTS)
CTS is the most common entrapment neuropathy, caused by compression of the median nerve at the wrist within the carpal tunnel (bounded by carpal bones and transverse carpal ligament). Presentation: numbness/tingling in median nerve distribution (thumb, index, middle, radial ring finger), pain worse at night (wakes patient), weak grip, thenar atrophy (late finding). Provocative tests: Tinel sign (tapping over carpal tunnel reproduces symptoms), Phalen test (sustained wrist flexion for 60 seconds reproduces symptoms; most sensitive clinical test), Durkan test (direct compression over carpal tunnel). Risk factors: pregnancy, diabetes, hypothyroidism, RA, obesity, repetitive wrist use.
Electrodiagnostic confirmation: prolonged median sensory distal latency (>3.5 ms over 14 cm) is the most sensitive NCS finding. Prolonged median motor distal latency (>4.2 ms) indicates more severe involvement. EMG may show fibrillations in APB (abductor pollicis brevis) in severe cases. Treatment: wrist splint in neutral (especially at night), activity modification, corticosteroid injection, surgical carpal tunnel release for moderate-to-severe or refractory cases.
Rotator Cuff Rehabilitation
Rotator cuff disorders represent a continuum: tendinopathy → partial tear → full-thickness tear. Supraspinatus is the most commonly affected tendon. Assessment tests: Neer test (passive forward flexion with scapula stabilized — impingement), Hawkins test (90° forward flexion then internal rotation — impingement), empty can test (Jobe — supraspinatus), external rotation lag sign (infraspinatus), lift-off test and belly press test (subscapularis). Rehabilitation phases: (1) pain control + protected ROM, (2) progressive ROM + scapular stabilization, (3) rotator cuff strengthening (internal/external rotation), (4) sport/work-specific training. Surgical repair is indicated for acute full-thickness tears in active patients and failed conservative management (≥6 months).
Lateral Epicondylitis (Tennis Elbow)
Tendinopathy of the common extensor origin, primarily ECRB (extensor carpi radialis brevis). Presentation: lateral elbow pain with gripping, wrist extension against resistance. Treatment: activity modification, counterforce brace (forearm strap), eccentric strengthening (wrist extensors), corticosteroid injection (short-term benefit but may impair long-term healing), PRP injection, surgical debridement (refractory cases).
21 Lower Extremity & Joint Replacement Rehab
Total Hip Arthroplasty (THA) Rehabilitation
The surgical approach determines post-operative precautions:
| Approach | Precautions | Duration |
|---|---|---|
| Posterior (most common) | No hip flexion >90°, no adduction past midline, no internal rotation (avoid combined flexion + adduction + IR). No low chairs, use elevated toilet seat, hip abduction pillow in bed | 6–12 weeks |
| Anterior (direct anterior) | No hip extension past neutral, no external rotation, no combined extension + ER. Fewer restrictions overall | 2–6 weeks |
| Lateral (anterolateral) | Limited hip abduction against resistance; gluteus medius precautions | 6–8 weeks |
Rehabilitation milestones: weight-bearing as tolerated (typically), gait training with walker/crutches, stair training (up with good leg first, down with operated leg first — "up with the good, down with the bad"), hip strengthening (abductors, extensors), ROM exercises, ADL training with adaptive equipment (long-handled reacher, sock aid, shoe horn). DVT prophylaxis for 35 days post-operatively.
Total Knee Arthroplasty (TKA) Rehabilitation
Goals: achieve ≥0° extension and ≥120° flexion. Immediate WBAT with assistive device. Cryotherapy, CPM (continuous passive motion) use is controversial (recent evidence shows minimal benefit). Progressive rehabilitation: quad sets, SLR, AROM, progressive weight-bearing, stair training, stationary bike (when ≥90° flexion achieved). DVT prophylaxis for 14–35 days. Key complications: stiffness (may require manipulation under anesthesia if <90° flexion at 6–8 weeks), peroneal nerve palsy (from traction, positioning — foot drop), wound complications, instability.
Osteoarthritis Management (Non-Surgical)
Evidence-based approach: weight management (5% weight loss reduces knee OA symptoms significantly), aerobic and strengthening exercise, PT, NSAIDs (topical preferred for knee OA), acetaminophen, intra-articular corticosteroid injections (short-term relief), viscosupplementation (hyaluronic acid — modest benefit in knee OA), bracing (unloader knee brace for unicompartmental OA), assistive devices (cane in contralateral hand reduces hip joint loading by 25%).
22 Tendinopathy & Myofascial Pain
Tendinopathy
Modern understanding favors the term tendinopathy over "tendinitis" because histopathology typically shows degenerative changes (collagen disorganization, ground substance mucoid degeneration, neovascularization) rather than inflammatory infiltrate. Common tendinopathies: Achilles, patellar, rotator cuff (supraspinatus), lateral epicondyle (ECRB), de Quervain (APL/EPB at first dorsal compartment). Treatment principles: relative rest (not complete immobilization), eccentric exercise (strongest evidence — particularly for Achilles and patellar tendinopathy), load management, progressive tendon loading (heavy slow resistance training), NSAIDS (short-term only — may impair healing long-term), nitroglycerin patches (some evidence), PRP injection (mixed evidence), shockwave therapy (ESWT — evidence for calcific tendinopathy), avoid repeated corticosteroid injections (accelerate tendon degeneration).
Myofascial Pain Syndrome
Myofascial pain syndrome (MPS) is characterized by myofascial trigger points (MTrPs) — hyperirritable spots within a taut band of skeletal muscle that produce local and referred pain. Features: palpable taut band, exquisite spot tenderness, patient recognition of pain (jump sign), referred pain pattern, local twitch response with snapping palpation. Active trigger points cause spontaneous pain; latent trigger points cause pain only with compression. Common locations: upper trapezius (refers to temporal headache), levator scapulae, infraspinatus, quadratus lumborum, piriformis. Treatment: trigger point injection (lidocaine or dry needling), spray and stretch, manual therapy (ischemic compression, myofascial release), postural correction, stretching, addressing perpetuating factors (poor ergonomics, sleep disturbance, stress, vitamin D deficiency).
23 Nerve Conduction Studies
Nerve conduction studies (NCS) are the objective, quantitative component of the electrodiagnostic evaluation. They assess the integrity and function of peripheral nerves by applying electrical stimulation and recording the evoked responses. NCS evaluate both motor and sensory nerve fibers and can localize lesions to specific nerve segments.
Motor NCS
A compound motor action potential (CMAP) is recorded from a muscle after supramaximal stimulation of its motor nerve at two or more points along its course. Parameters measured: distal latency (time from distal stimulation to CMAP onset), amplitude (baseline to negative peak, reflects number of functioning motor axons), conduction velocity (distance between stimulation sites / difference in latencies; normal ≥50 m/s in upper extremity, ≥40 m/s in lower extremity), duration (temporal dispersion). A >50% drop in CMAP amplitude between distal and proximal stimulation sites indicates conduction block (focal demyelination — hallmark of neurapraxia).
Sensory NCS
A sensory nerve action potential (SNAP) is recorded after stimulating a sensory or mixed nerve. Can be performed orthodromic (stimulate distally, record proximally) or antidromic (stimulate proximally, record distally — larger amplitude, more commonly used). Parameters: onset latency, peak latency, amplitude (reflects number of functioning sensory axons), conduction velocity. SNAPs are critical for distinguishing pre-ganglionic (root) from post-ganglionic (plexus/nerve) lesions: in radiculopathy, the dorsal root ganglion (DRG) is proximal to the lesion, so the sensory axons distal to the DRG remain intact and SNAPs are preserved despite clinical sensory loss.
Late Responses
| Response | Pathway | Clinical Utility |
|---|---|---|
| F-wave | Motor fiber → anterior horn cell (antidromic activation) → back down motor fiber; assesses entire motor nerve length including proximal segments | Abnormal in proximal neuropathies (GBS, plexopathy), radiculopathy; variable latency (not a reflex); late, low-amplitude response |
| H-reflex | Afferent Ia sensory fiber → spinal cord (monosynaptic arc) → efferent motor fiber; equivalent of the electrically elicited ankle jerk | Most reliably obtained from soleus (tibial nerve, S1 root); absent or prolonged in S1 radiculopathy, peripheral neuropathy, GBS. Present bilaterally in normal adults; side-to-side latency difference >1.5 ms is abnormal |
24 Needle EMG
Needle electromyography (EMG) assesses the electrical activity of muscle fibers and motor units by inserting a needle electrode into the muscle. It provides information about denervation, reinnervation, myopathy, and motor unit morphology that NCS alone cannot provide.
Insertional Activity
Normal insertional activity: brief burst of electrical activity with needle insertion/movement, lasting <300 ms. Increased insertional activity: sustained activity beyond normal duration; earliest sign of denervation (before fibrillations appear). Decreased insertional activity: seen in fibrotic or fatty-replaced muscle (chronic denervation, severe myopathy).
Spontaneous Activity
| Finding | Morphology | Sound | Significance |
|---|---|---|---|
| Fibrillation potentials | Brief, biphasic or triphasic spikes; regular firing rate (1–30 Hz) | Rain on a tin roof / ticking clock | Denervated muscle fiber spontaneous discharge; indicates active denervation (appears 2–3 weeks post-injury); also in myopathy, NMJ disorders |
| Positive sharp waves (PSWs) | Initial positive deflection followed by slow negative phase; regular firing | Dull thud | Same significance as fibrillations; often seen together; denervation |
| Fasciculation potentials | Motor unit morphology; irregular firing rate | Popcorn popping | Spontaneous motor unit discharge; benign fasciculations (common, no associated weakness) vs pathologic (ALS — with other denervation signs) |
| Myotonic discharges | Waxing and waning amplitude and frequency | Dive bomber / revving motorcycle | Myotonic dystrophy, myotonia congenita, acid maltase deficiency, hyperkalemic periodic paralysis |
| Complex repetitive discharges (CRDs) | Polyphasic; regular firing; abrupt onset/cessation | Machine gun / helicopter | Chronic denervation or myopathy; non-specific; indicates chronicity |
| Myokymic discharges | Grouped, repetitive motor unit potentials firing semi-rhythmically | Marching soldiers | Radiation plexopathy (distinguishes from tumor recurrence), GBS, MS |
Motor Unit Analysis
Motor unit action potentials (MUAPs) are assessed during voluntary contraction for morphology (amplitude, duration, phases) and recruitment (firing pattern):
| Feature | Neurogenic (Denervation/Reinnervation) | Myopathic |
|---|---|---|
| MUAP amplitude | Increased (reinnervation: surviving motor units adopt orphaned fibers) | Decreased (fewer functional muscle fibers per motor unit) |
| MUAP duration | Increased (larger, more dispersed motor unit territory) | Decreased (smaller motor unit territory) |
| Phases | Polyphasic (reinnervation — nascent units) | Polyphasic (short duration) |
| Recruitment | Reduced / discrete (fewer motor units available; remaining units fire rapidly — fast firing rate with few units) | Early / full (all motor units activated at low force levels; each generates less force) |
| Interference pattern | Reduced (incomplete) | Full but low amplitude |
25 Electrodiagnostic Patterns by Condition
Characteristic EDX Patterns
| Condition | NCS Findings | EMG Findings |
|---|---|---|
| Carpal Tunnel Syndrome (CTS) | Prolonged median sensory distal latency (most sensitive); prolonged median motor distal latency; normal ulnar NCS; normal median SNAP amplitude (early) or reduced (severe) | APB: fibrillations/PSWs if severe; reduced recruitment; neurogenic MUAPs |
| Ulnar Neuropathy at Elbow | Slowed conduction velocity across elbow segment (>10 m/s drop); conduction block across elbow; reduced ulnar SNAP; reduced ulnar CMAP (ADM, FDI) | Denervation in ulnar-innervated hand intrinsics (FDI, ADM) and FDP to ring/small finger; FCU may be spared (branch proximal to cubital tunnel) |
| Radiculopathy | Normal NCS (SNAPs preserved because lesion is preganglionic); F-waves may be prolonged | Fibrillations/PSWs in a myotomal pattern (multiple muscles sharing the same root, different peripheral nerves); abnormal paraspinals at involved level |
| Polyneuropathy (Axonal) | Reduced SNAP/CMAP amplitudes (length-dependent: legs > arms); normal or mildly slow conduction velocities | Distal > proximal denervation; length-dependent pattern; fibs/PSWs in distal muscles |
| Polyneuropathy (Demyelinating) | Markedly slowed conduction velocities (<70% LLN); prolonged distal latencies; prolonged F-waves; temporal dispersion; conduction block | May be normal or show mild neurogenic changes |
| Myopathy | Normal NCS (or mildly reduced CMAPs in severe cases); SNAPs normal | Short-duration, low-amplitude, polyphasic MUAPs; early recruitment; fibrillations in inflammatory/necrotizing myopathies (dermatomyositis, polymyositis) |
| ALS | Normal SNAPs (pure motor); reduced CMAPs; normal conduction velocities | Widespread denervation (fibrillations, PSWs, fasciculations) in ≥3 body regions (cranial, cervical, thoracic, lumbosacral); large, long-duration MUAPs; reduced recruitment |
| GBS | Early: prolonged F-waves, prolonged distal latencies; Progressive: slow conduction velocities, conduction block, temporal dispersion (AIDP variant); Axonal variant (AMAN): reduced CMAPs with normal velocities | Denervation in severe/axonal cases |
| Myasthenia Gravis | Repetitive nerve stimulation (RNS): >10% decrement at 2–3 Hz (low-frequency); SNAP/CMAP normal | Normal routine EMG; single-fiber EMG (SFEMG) shows increased jitter (most sensitive test) |
Timing of Electrodiagnostic Studies
| Time Post-Injury | Findings Available |
|---|---|
| Immediately (0–3 days) | NCS can identify pre-existing pathology and conduction block (neurapraxia); EMG normal |
| 3–5 days | Motor CMAPs begin to decline distally (Wallerian degeneration of motor axons) |
| 7–10 days | SNAPs decline distally (sensory Wallerian degeneration); can distinguish neurapraxia from axonotmesis |
| 2–3 weeks | Fibrillations/PSWs appear in proximal muscles |
| 3–4 weeks (OPTIMAL) | Full EDX evaluation possible: NCS changes complete, EMG denervation present in proximal + distal muscles; can localize and grade severity |
| 4–6 weeks | Fibrillations in most distal muscles; complete picture |
| 3–6 months | Reinnervation potentials (nascent, polyphasic MUAPs) appear if recovery occurring |
26 Cerebral Palsy & GMFCS
Cerebral palsy (CP) is a group of permanent disorders of movement and posture causing activity limitation, attributed to non-progressive disturbances that occurred in the developing fetal or infant brain. It is the most common cause of childhood physical disability, affecting 2–3 per 1,000 live births. Although the brain lesion is static, its musculoskeletal manifestations change with growth.
CP Classification by Motor Type
| Type | Frequency | Lesion Location | Features |
|---|---|---|---|
| Spastic | 70–80% | Cortex or white matter (pyramidal) | Velocity-dependent hypertonia; UMN signs; diplegia (most common — PVL in preterm), hemiplegia, or quadriplegia |
| Dyskinetic | 10–15% | Basal ganglia (extrapyramidal) | Involuntary movements: dystonia (sustained postures), choreoathetosis (writhing); fluctuating tone; often full-term, kernicterus |
| Ataxic | 5% | Cerebellum | Poor coordination, balance, intention tremor; wide-based gait; hypotonia |
| Mixed | 10–15% | Multiple areas | Combination (most commonly spastic + dyskinetic) |

Gross Motor Function Classification System (GMFCS) — Levels I–V
| Level | Description (Age 6–12 years) | Mobility Aids |
|---|---|---|
| I | Walks without limitations; limitations in more advanced gross motor skills (running, jumping, speed, balance) | None needed for walking |
| II | Walks in most settings with limitations; difficulty with uneven surfaces, inclines, long distances, crowds; minimal ability to run/jump; may use handheld mobility device outdoors | May use assistive device for community distances |
| III | Walks with handheld mobility device (walker, crutches) indoors; wheelchair for long distances and outdoors; may climb stairs with railing | Walker/crutches indoors; manual or power wheelchair outdoors |
| IV | Self-mobility with limitations; transported in wheelchair in most settings; may use power mobility; may achieve assisted walking for short distances with adaptive equipment | Power wheelchair primary; may walk short distances with body support walker |
| V | Transported in manual wheelchair in all settings; severely limited in ability to maintain antigravity head and trunk postures; limited voluntary control of movement; requires extensive adaptations for seating | Manual wheelchair pushed by others; dependent for all mobility; adaptive seating |
Tone Management in CP
Treatment is goal-directed based on GMFCS level and specific functional limitations. Options: oral medications (baclofen, diazepam, dantrolene — limited by sedation/generalized weakness), botulinum toxin injections (focal spasticity, delayed need for surgery, commonly injected: gastrocnemius, hamstrings, hip adductors), intrathecal baclofen (generalized lower extremity spasticity, GMFCS III–V), selective dorsal rhizotomy (SDR) (sectioning posterior rootlets L1–S2 to reduce spasticity; best candidates: spastic diplegic CP, GMFCS II–III, age 4–8, good underlying strength), orthopedic surgery (muscle-tendon lengthening, tendon transfers, bony procedures for hip displacement/scoliosis).
Hip Surveillance in CP
Hip displacement is common in CP (1% in GMFCS I, up to 90% in GMFCS V). The migration percentage (MP) on AP pelvis X-ray quantifies femoral head uncovering. Screening protocol: AP pelvis X-ray at age 12–24 months, then annually for GMFCS III–V, every 1–2 years for GMFCS II. MP >30% warrants referral to orthopedics; MP >40–50% typically requires surgical intervention (soft tissue releases, varus derotation osteotomy, pelvic osteotomy).
27 Spina Bifida & Brachial Plexus Injury
Spina Bifida (Myelomeningocele)
Myelomeningocele is the most severe form of spina bifida, involving herniation of the spinal cord and meninges through a vertebral defect. It is the most common neural tube defect compatible with life. Associated conditions: Arnold-Chiari II malformation (virtually 100% — cerebellar tonsillar herniation through foramen magnum), hydrocephalus (80–90% require VP shunt), tethered cord (can present with new neurological decline during growth), and latex allergy (up to 65% sensitization — use latex-free precautions for ALL procedures).
Functional Expectations by Lesion Level
| Level | Key Muscles Innervated | Ambulatory Potential | Orthotic Needs |
|---|---|---|---|
| Thoracic | No LE function | Non-functional/exercise ambulation only | RGO (reciprocating gait orthosis) or parapodium; wheelchair primary |
| L1–L2 | Hip flexors (weak) | Household ambulation possible | KAFOs + walker/crutches; wheelchair for community |
| L3–L4 | Quadriceps, hip adductors | Community ambulation | AFOs + forearm crutches; wheelchair for long distances |
| L5 | + Hamstrings, tibialis anterior, gluteus medius | Community ambulation | AFOs; may use crutches |
| S1–S2 | + Gastrocnemius, gluteus maximus | Community ambulation without assistive device | May need shoe inserts or ankle supports |
Obstetric Brachial Plexus Injury
| Type | Roots | Presentation | Prognosis |
|---|---|---|---|
| Erb-Duchenne palsy | C5–C6 (upper trunk) | Most common (75–80%); "waiter's tip" position: shoulder adducted/IR, elbow extended, forearm pronated, wrist flexed | Good — 70–90% recover spontaneously by 3–6 months |
| Extended Erb palsy | C5–C7 | Erb pattern + loss of elbow/wrist/finger extension | Intermediate |
| Klumpke palsy | C8–T1 (lower trunk) | Rare (<1%); absent hand intrinsics and finger flexors; "claw hand"; may have Horner syndrome (T1 sympathetic involvement) | Variable; Horner syndrome worsens prognosis |
| Total plexus palsy | C5–T1 | Flail arm; worst prognosis | Poor; often requires surgical intervention |
If no biceps function by 3 months, microsurgical nerve repair (nerve grafting, nerve transfer) should be considered. Active ROM exercises, splinting, and positioning are mainstays of conservative management.
28 Muscular Dystrophies & Developmental Milestones
Muscular Dystrophies
| Type | Inheritance | Onset | Key Features | Prognosis |
|---|---|---|---|---|
| Duchenne (DMD) | X-linked recessive (dystrophin absent) | 2–5 years | Progressive proximal weakness; Gowers sign (climbing up legs to stand); pseudohypertrophy of calves; CK markedly elevated (10,000+); loss of ambulation by 12; scoliosis; cardiomyopathy; cognitive impairment (1 SD below mean) | Death in 20s–30s (respiratory/cardiac failure); corticosteroids (deflazacort, prednisone) prolong ambulation 2–5 years |
| Becker (BMD) | X-linked recessive (dystrophin reduced/abnormal) | 5–15 years | Similar to DMD but milder; ambulatory into adulthood; cardiomyopathy may be prominent | Variable; often survive into 40s–50s+ |
| Myotonic Dystrophy type 1 (DM1) | Autosomal dominant (CTG repeat) | Adolescence–adult | Myotonia (grip/percussion), distal > proximal weakness, facial weakness, ptosis, cataracts, cardiac conduction defects, insulin resistance, cognitive involvement; genetic anticipation (earlier/more severe in successive generations) | Variable; cardiac events are leading cause of death |
| Facioscapulohumeral (FSHD) | Autosomal dominant | Adolescence | Face (cannot whistle, close eyes tightly), scapular stabilizer (scapular winging), and humeral weakness; asymmetric; foot drop common; hearing loss; retinal vasculopathy | Normal lifespan in most; 20% become wheelchair-dependent |
Developmental Milestones
| Age | Gross Motor | Fine Motor | Language | Social |
|---|---|---|---|---|
| 2 months | Lifts head prone | Hands open frequently | Coos | Social smile |
| 4 months | Rolls front to back; holds head steady | Reaches for objects | Laughs | Enjoys social interaction |
| 6 months | Sits with support; rolls both ways | Raking grasp; transfers objects | Babbles (consonant sounds) | Stranger anxiety emerging |
| 9 months | Sits independently; pulls to stand; crawls | Pincer grasp | "Mama/dada" (non-specific) | Stranger anxiety peaks |
| 12 months | Walks with hand held or independently | Pincer grasp mature; releases on request | 1–3 words (specific) | Waves bye-bye; plays pat-a-cake |
| 18 months | Walks well; runs stiffly | Tower of 3–4 blocks; scribbles | 10–25 words; points to body parts | Parallel play |
| 24 months | Runs well; kicks ball; walks up stairs | Tower of 6 blocks; turns pages | 2-word phrases; 50+ words | Parallel play; imitates adults |
| 3 years | Pedals tricycle; broad jumps | Copies circle; tower of 9 blocks | 3-word sentences; 75% intelligible | Group play; knows name/age/gender |
| 4 years | Hops on one foot; climbs stairs alternating feet | Copies cross; draws person (3 parts) | Tells stories; 100% intelligible | Cooperative play; imaginary friends |
29 Amputation Levels & Pre-Prosthetic Management
Lower Extremity Amputation Levels
| Level | Description | Prosthetic Considerations | Energy Cost of Ambulation |
|---|---|---|---|
| Toe / Ray | Partial digit or metatarsal ray amputation | Shoe filler; toe prosthesis | Minimal increase |
| Transmetatarsal (TMA) | Through metatarsal shafts | Rigid ankle rocker-bottom shoe; AFO shoe insert | Slight increase |
| Syme | Through ankle joint; distal tibial/fibular flares removed; heel pad preserved (end-bearing) | Low-profile foot; excellent weight bearing; bulbous distal residual limb | ~15% increase |
| Transtibial (below-knee, BKA) | Through tibia/fibula; optimal length 12–17 cm from medial tibial plateau (mid-third ideal) | Preserved knee joint; best functional outcomes of major amputations; PTB or TSB socket | ~25–40% increase (vascular); ~10–20% (traumatic) |
| Knee Disarticulation | Through knee joint | End-bearing; long lever arm; bulbous distal end (fitting challenge); good for sitting balance | ~40% increase |
| Transfemoral (above-knee, AKA) | Through femur; optimal length 25–30 cm from greater trochanter | Requires prosthetic knee unit; hip musculature critical for prosthetic control | ~65–100% increase (vascular); ~25–40% (traumatic) |
| Hip Disarticulation | Through hip joint | Canadian-type socket; very high energy demands | >100% increase |
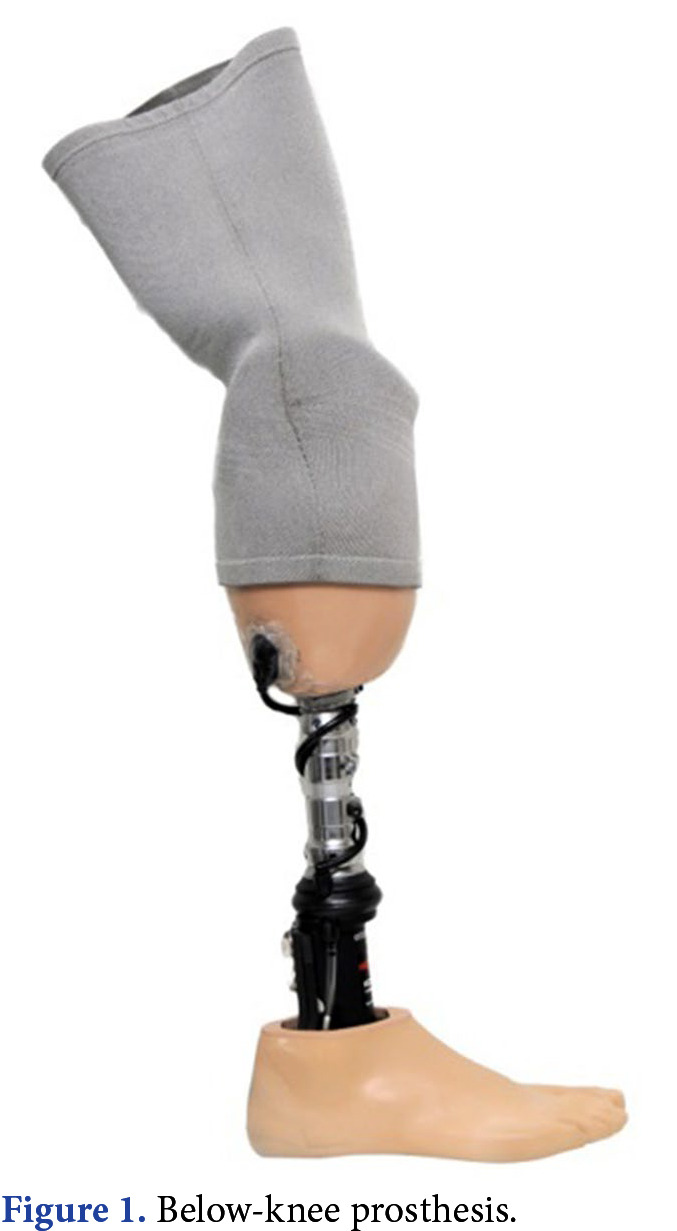
Pre-Prosthetic Management
Pre-prosthetic care begins immediately post-operatively: wound care and monitoring for healing (especially critical in vascular amputees with diabetes and peripheral vascular disease), edema control and limb shaping (figure-of-eight elastic wrapping, rigid removable dressings, or shrinker socks to shape the residual limb into a conical shape for optimal socket fit), desensitization (tapping, rubbing, weight bearing on residual limb to reduce hypersensitivity), ROM exercises (prevent hip flexion/abduction contracture in transfemoral, knee flexion contracture in transtibial — prone positioning helps), strengthening (emphasis on hip extensors/abductors in AKA, knee extensors in BKA), single-leg balance and transfers, and psychological support (grief, body image, adjustment).
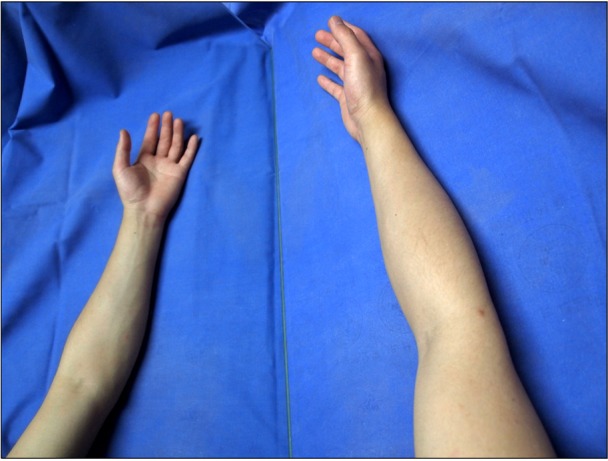
Phantom Limb Pain
Phantom limb pain occurs in 50–80% of amputees; distinguished from phantom limb sensation (non-painful awareness of the absent limb, nearly universal) and residual limb pain (pain in the remaining stump). Mechanisms include peripheral (neuroma formation), spinal (dorsal horn sensitization), and cortical (maladaptive neuroplasticity / cortical reorganization) factors. Management:
| Treatment | Mechanism/Notes |
|---|---|
| Mirror therapy | Visual feedback using a mirror to "see" the missing limb move; reduces cortical reorganization; first-line non-pharmacological therapy |
| Gabapentin (300–3600 mg/day) | Neuropathic pain; most commonly used medication |
| Pregabalin (75–300 mg BID) | Neuropathic pain; may be better tolerated than gabapentin |
| TCAs (amitriptyline, nortriptyline) | Neuropathic pain adjunct; anticholinergic side effects limit use |
| SNRIs (duloxetine, venlafaxine) | Dual mechanism; fewer side effects than TCAs |
| TENS | Applied to residual limb or contralateral limb |
| Desensitization techniques | Tapping, massage, weight-bearing on residual limb |
| Prosthetic use | Early prosthetic fitting reduces phantom pain incidence |
30 Prosthetic Components & Prescription
Transtibial Prosthetic Components
Socket: Patellar-tendon-bearing (PTB) — primary weight bearing on patellar tendon, medial tibial flare, anterior tibia protected; Total surface bearing (TSB) — distributes pressure over the entire residual limb using gel liner (modern standard). Suspension: Pin/shuttle lock (pin at end of gel liner locks into socket), suction (vacuum seal), sleeve suspension (neoprene or gel sleeve over socket and thigh), supracondylar cuff/strap. Foot/ankle units: SACH (Solid Ankle Cushion Heel) — simplest, durable, no moving parts, cushioned heel compresses to simulate plantarflexion; single-axis foot — allows plantarflexion/dorsiflexion, improves knee stability at heel strike; multi-axis foot — allows motion in all planes, good for uneven terrain; dynamic response/energy-storing feet (carbon fiber, Flex-Foot) — store energy during stance, release during push-off, best for active ambulators (K3–K4).
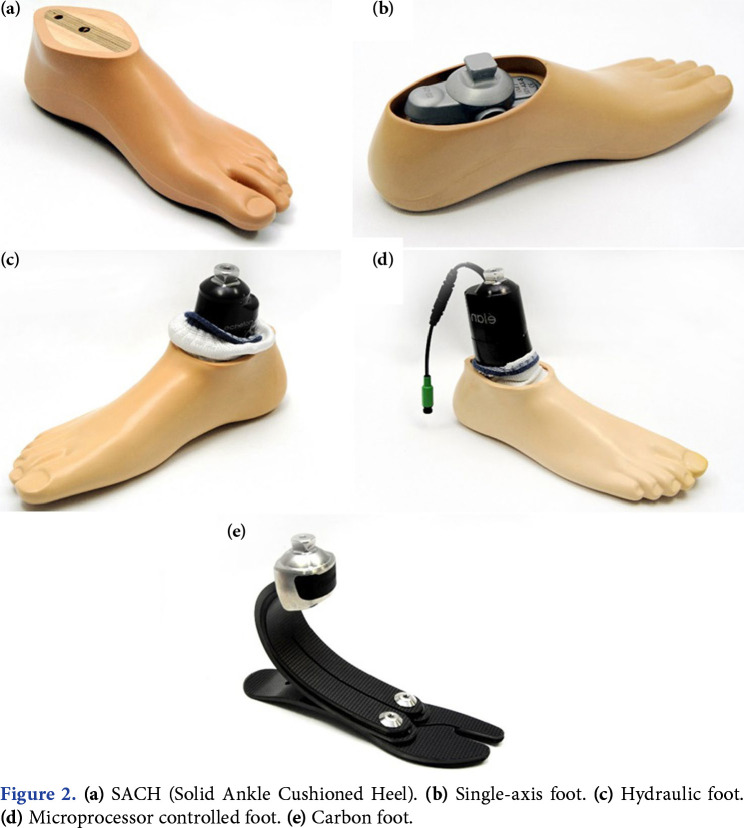
Transfemoral Prosthetic Components
Socket: Ischial containment (IC) — modern standard; ischial tuberosity inside the socket, narrow ML dimension, improved mediolateral control; Quadrilateral — older design, ischial tuberosity sits on posterior brim shelf. Knee units: Single-axis (manual lock) — most stable, for K1 patients; polycentric (multi-axis) — shortens in swing phase (cosmetic benefit), inherent stability; hydraulic/pneumatic — fluid resistance for smooth swing phase at variable cadences (K3+); microprocessor knee (C-Leg, Genium, Rheo Knee) — computer-controlled resistance, adapts to walking speed, improved stumble recovery, stair descent, ramp navigation; for K2–K4 patients.

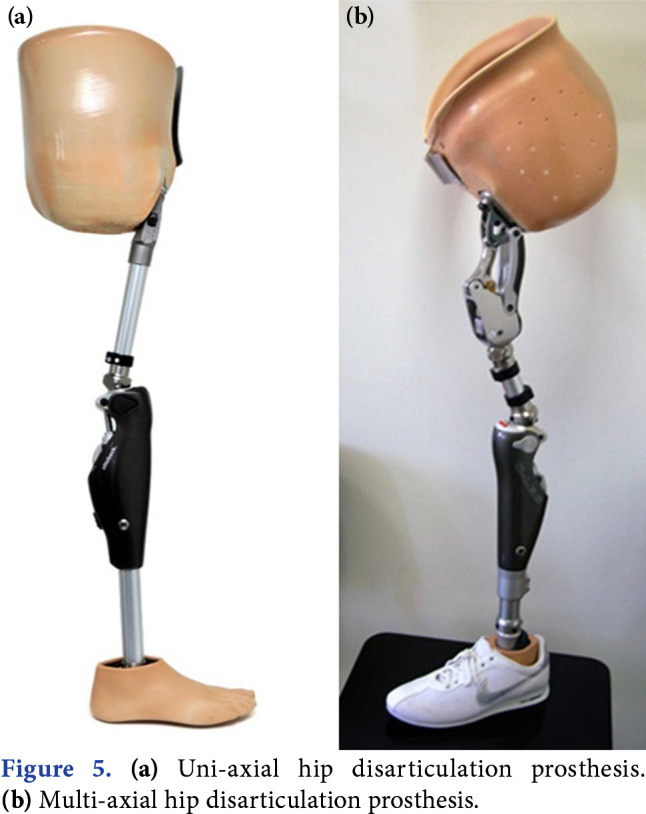
Upper Extremity Prosthetics
Types: body-powered (cable-operated; harness converts shoulder motion to terminal device operation; most durable, provides proprioceptive feedback), myoelectric (EMG signals from residual muscles control electric motors; more cosmetic, stronger grip, but heavier and less durable), passive/cosmetic (no active function; cosmesis and light tasks), activity-specific (adapted for sports, work). Terminal devices: hook (voluntary opening or closing; excellent prehension, most functional) and hand (cosmetic advantage; weaker grasp).
31 Gait Deviations & K-Levels
Common Prosthetic Gait Deviations
| Deviation | Prosthetic Cause | Patient/Residual Limb Cause |
|---|---|---|
| Lateral trunk lean | Prosthesis too short; wide ML socket | Weak hip abductors (Trendelenburg); short residual limb; hip pain |
| Circumduction | Prosthesis too long; excessive knee friction; inadequate suspension | Weak hip flexors; hip abduction contracture |
| Vaulting | Prosthesis too long; inadequate knee flexion in swing; poor socket fit | Fear of stubbing toe |
| Wide-based gait | Prosthesis too long; excessive outset of foot; wide socket | Abduction contracture; instability; poor balance |
| Excessive knee flexion at heel strike | Socket set too far anterior; too much dorsiflexion; soft heel cushion | Weak quadriceps; knee flexion contracture |
| Terminal swing impact | Insufficient knee friction (swing too fast) | Forceful hip flexion |
| Uneven step length | Socket discomfort; unstable knee unit | Hip flexion contracture; fear; poor prosthetic training |
| Medial/lateral whip | Rotational malalignment of knee bolt or foot | Residual limb rotational deformity |
Medicare K-Levels (Functional Classification)
| K-Level | Description | Prosthetic Components Justified |
|---|---|---|
| K0 | Does not have the ability or potential to ambulate or transfer with or without assistance; prosthesis does not enhance quality of life or mobility | No prosthetic limb (or cosmetic prosthesis only) |
| K1 | Has ability or potential to use a prosthesis for transfers or ambulation on level surfaces at a fixed cadence (limited or unlimited household ambulator) | Basic components: SACH foot, single-axis knee with manual lock; no energy-storing feet or microprocessor knees |
| K2 | Has ability or potential for ambulation with ability to traverse low-level environmental barriers (curbs, stairs, uneven surfaces); limited community ambulator | Single-axis or multi-axis foot; weight-activated stance control knee or polycentric knee; microprocessor knee may be justified |
| K3 | Has ability or potential for ambulation with variable cadence; community ambulator with ability to traverse most environmental barriers; may have vocational, therapeutic, or exercise activity demanding prosthetic use beyond simple locomotion | Dynamic response/energy-storing feet; hydraulic/pneumatic knee; microprocessor knee |
| K4 | Has ability or potential for prosthetic ambulation exceeding basic ambulation skills; high impact, stress, or energy levels (child, active adult, athlete) | Highest-level components; sport-specific prostheses; running blades |
32 Spasticity Injections & Neurolysis
Botulinum Toxin Injection — Dosing by Muscle
OnabotulinumtoxinA (Botox) dosing guidelines for common spasticity targets (doses are for onabotulinumtoxinA; conversion: 1 U Botox ≈ 3–5 U Dysport ≈ 1 U Xeomin):
| Muscle | Dose Range (Units) | Injection Sites | Functional Target |
|---|---|---|---|
| Biceps brachii | 100–200 | 2–4 | Elbow flexion spasticity |
| Brachioradialis | 50–100 | 1–2 | Elbow flexion spasticity |
| Brachialis | 50–100 | 1–2 | Elbow flexion spasticity |
| Pronator teres | 25–75 | 1 | Forearm pronation posturing |
| Flexor carpi radialis | 25–100 | 1–2 | Wrist flexion spasticity |
| Flexor carpi ulnaris | 25–75 | 1 | Wrist flexion spasticity |
| Flexor digitorum superficialis | 25–75 per head | 1–2 per head | Finger flexion / clenched fist |
| Flexor digitorum profundus | 25–50 | 1–2 | Finger flexion |
| Adductor pollicis / FPL | 10–30 | 1 | Thumb-in-palm deformity |
| Gastrocnemius (each head) | 75–200 | 2–4 per head | Equinovarus foot / ankle plantarflexion |
| Soleus | 75–100 | 2–3 | Ankle plantarflexion |
| Tibialis posterior | 50–100 | 1–2 | Foot inversion (equinovarus) |
| Adductor magnus / longus / brevis | 75–200 (total) | 2–4 | Hip adductor spasticity (scissoring gait, perineal care) |
| Hamstrings (medial / lateral) | 100–200 (total) | 2–4 | Knee flexion spasticity |
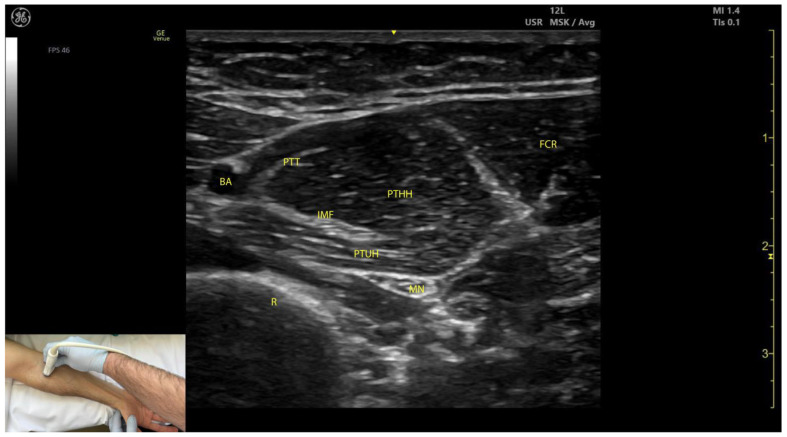
Maximum total dose per treatment session: typically 400–600 U onabotulinumtoxinA for adults (some experts use up to 800 U for large adults with diffuse spasticity). Injection localization methods: EMG guidance, electrical stimulation, ultrasound (increasingly preferred for superficial muscles). Re-injection interval: ≥12 weeks minimum.
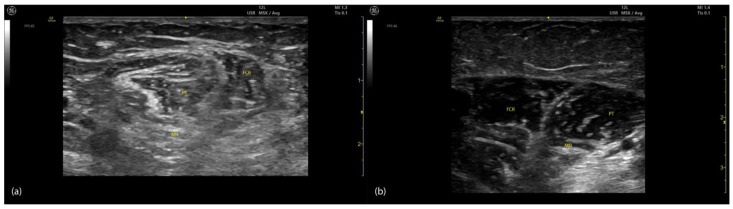
Phenol & Alcohol Neurolysis
Phenol (5–7% aqueous solution) and alcohol (45–100% ethanol) produce chemical neurolysis — protein denaturation and nerve destruction, reducing spasticity. Advantages: immediate effect, longer duration (3–12+ months), lower cost than botulinum toxin, no dose ceiling. Disadvantages: painful injection, risk of dysesthesias (especially with mixed sensorimotor nerves), fibrosis with repeated injections. Best targets: motor nerve branches with minimal sensory component — musculocutaneous nerve (elbow flexion spasticity), obturator nerve (hip adductor spasticity — most common target), tibial nerve branches to gastrocnemius (equinovarus). Phenol motor point blocks inject the motor point (NMJ entry zone) directly within the muscle.
33 Intrathecal Baclofen & Guided Procedures
Intrathecal Baclofen Pump
The ITB pump system consists of a programmable titanium pump (implanted subcutaneously in the abdominal wall) and an intrathecal catheter threaded into the spinal subarachnoid space (tip typically at T10–T12 for LE spasticity, C5–C7 for UE + LE spasticity). The pump delivers continuous baclofen to the CSF at a programmable rate.
ITB Trial Protocol
| Step | Dose | Assessment |
|---|---|---|
| First bolus | 50 mcg via lumbar puncture | Assess Ashworth scores at 1, 2, 4, 6, 8 hours post-injection |
| Positive trial | ≥1-point Ashworth reduction | Proceed to pump implant |
| Negative trial (first dose) | 75 mcg second trial (24+ hours later) | Repeat assessment |
| Negative trial (second dose) | 100 mcg third trial | If still negative, patient is not a candidate |
Post-implant: starting continuous dose is typically 2× the effective bolus trial dose per 24 hours (e.g., 50 mcg bolus → 100 mcg/day). Dose titrated over weeks to months. Pump refills required every 1–6 months depending on concentration and flow rate. MRI-conditional pumps are available.
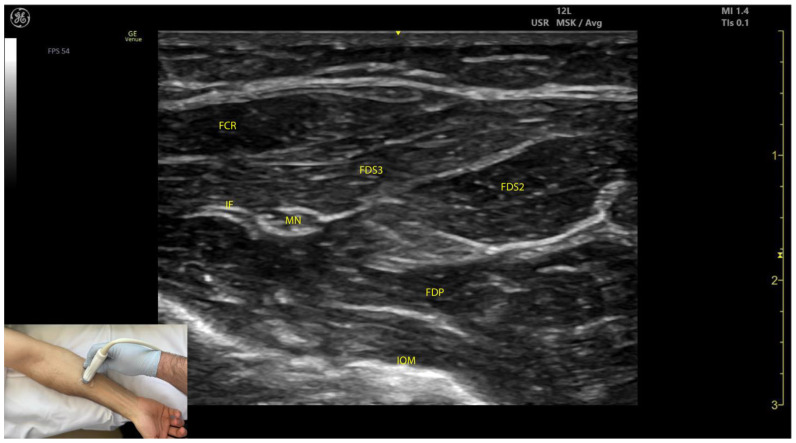
Fluoroscopic & Ultrasound-Guided Procedures
PM&R physicians commonly perform image-guided procedures:
| Procedure | Guidance | Indication |
|---|---|---|
| Epidural steroid injection (ESI) — interlaminar, transforaminal, caudal | Fluoroscopy (standard) or CT | Radiculopathy, spinal stenosis |
| Facet joint injection / medial branch block | Fluoroscopy | Facetogenic back/neck pain; diagnostic/therapeutic |
| Radiofrequency ablation (RFA) | Fluoroscopy | Confirmed facet-mediated pain after positive diagnostic blocks |
| Sacroiliac joint injection | Fluoroscopy or ultrasound | SI joint dysfunction/sacroiliitis |
| Peripheral nerve block | Ultrasound or nerve stimulator | Diagnostic/therapeutic for specific nerve pain or spasticity |
| Joint injection/aspiration | Ultrasound or landmark-based | Inflammatory/degenerative joint disease; joint effusion |
| Trigger point injection | Palpation-guided or ultrasound | Myofascial pain syndrome |
34 Therapeutic Modalities
Thermal Modalities
| Modality | Mechanism | Indications | Contraindications |
|---|---|---|---|
| Superficial Heat (hot packs, paraffin, fluidotherapy) | Conduction/convection heating; penetrates 1–2 cm; increases blood flow, extensibility, pain threshold | Muscle spasm, pain, joint stiffness, before stretching | Acute injury (<48 hours), hemorrhage, insensate skin, malignancy, DVT |
| Cryotherapy (ice packs, cold spray, ice massage) | Vasoconstriction, reduced nerve conduction velocity, decreased metabolic rate, reduced edema | Acute injury, post-surgical, edema, spasticity reduction | Raynaud disease, cold urticaria, cryoglobulinemia, insensate skin, PVD |
| Therapeutic Ultrasound (1–3 MHz) | Deep heating (3–5 cm); 1 MHz for deeper structures, 3 MHz for superficial; thermal (continuous) and non-thermal (pulsed) effects; increases collagen extensibility | Tendinopathy, joint contracture (combined with stretching), deep muscle spasm, delayed fracture healing (pulsed low-intensity) | Over growth plates, malignancy, CNS, pacemaker, pregnant uterus, eyes, testes, thrombophlebitis, cemented metal implants (thermal mode) |
| Diathermy (shortwave, microwave) | Deep heating via electromagnetic energy; greater depth and area than ultrasound | Deep joint/muscle heating, OA, muscle spasm | Metal implants (absolute for shortwave), pacemaker, pregnancy, malignancy |
Electrotherapy
| Modality | Mechanism | Clinical Use |
|---|---|---|
| TENS (Transcutaneous Electrical Nerve Stimulation) | Sensory-level stimulation; gate control theory (large-fiber afferent input closes pain gate); also stimulates endorphin release (low-frequency/acupuncture-like TENS) | Pain management (acute and chronic); non-invasive, patient-applied; conventional (high frequency 50–100 Hz, sensory level), acupuncture-like (low frequency 2–4 Hz, motor level) |
| NMES (Neuromuscular Electrical Stimulation) | Motor-level stimulation producing visible muscle contraction; prevents disuse atrophy, strengthens weak muscles | Muscle re-education post-surgery, strengthening (quadriceps post-TKA), prevention of disuse atrophy, edema reduction |
| FES (Functional Electrical Stimulation) | NMES applied during functional tasks to produce purposeful movement in paralyzed muscles | Foot drop (peroneal nerve stimulator for dorsiflexion during gait), hand grasp in SCI (C5–C6), FES cycling for SCI (cardiovascular conditioning, muscle preservation) |
| Iontophoresis | Uses direct current to drive charged medication molecules through the skin | Dexamethasone (anti-inflammatory, lateral epicondylitis), lidocaine (local analgesia), acetic acid (calcific tendinitis) |
Traction
Cervical traction: mechanical or manual; angle determines which disc space is opened (neutral = upper cervical; flexion = lower cervical). Sustained traction: 25–30 lb for 15–20 minutes (disc herniation). Intermittent traction: 5–10 seconds on/off cycles. Contraindications: ligamentous instability, RA (atlantoaxial instability), fracture, malignancy, acute disc herniation with progressive neurological deficit, spinal cord compression. Lumbar traction: force must exceed ~50% body weight to separate vertebral segments (typically 60–100 lb); limited evidence for efficacy.
35 Orthotic Prescribing
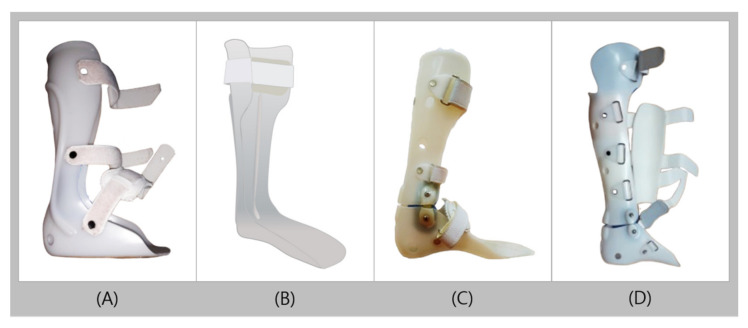
Lower Extremity Orthoses
| Orthosis | Design | Indications | Biomechanical Effect |
|---|---|---|---|
| Solid AFO | Rigid posterior leaf spring; fixed ankle in neutral or slight dorsiflexion | Severe foot drop, significant spasticity (ankle plantarflexors), mediolateral instability, weak quadriceps (blocks tibial advancement, creates knee extension moment) | Blocks all ankle motion; controls knee via ground reaction force |
| Hinged (Articulated) AFO | Solid AFO with ankle hinge; may have dorsiflexion assist and/or plantarflexion stop | Foot drop with preserved ankle dorsiflexion strength; allows more natural gait; patients needing dorsiflexion for sit-to-stand and stairs | Allows controlled ankle motion; dorsiflexion assist spring compensates for foot drop |
| Posterior Leaf Spring AFO | Thin, flexible posterior strut; trim lines narrow behind malleoli | Pure foot drop without significant spasticity or mediolateral instability (common peroneal palsy) | Flexible dorsiflexion assist; no mediolateral support; no spasticity control |
| CROW (Charcot Restraint Orthopedic Walker) | Total-contact clamshell AFO; bivalved, rigid, rocker-bottom sole | Charcot foot (neuropathic arthropathy); acute Charcot after initial casting phase; chronic Charcot deformity | Total contact; offloads deformed areas; protects insensate foot |
| KAFO | AFO extending above knee with knee joint (locked, drop-lock, or stance-control) | Quadriceps weakness (MMT <3), knee instability (recurvatum, valgus/varus), combined knee and ankle weakness | Controls knee and ankle; drop-lock allows locking for stance, unlocking for sitting |

Spinal Orthoses
| Orthosis | Indications | Motion Restricted |
|---|---|---|
| Soft cervical collar | Comfort/proprioceptive reminder; whiplash, minor cervical strain | Minimal motion restriction (~5–15%) |
| Philadelphia collar | Stable cervical fractures, post-surgical cervical support | Moderate flexion/extension restriction (~70%) |
| Miami J collar | Similar to Philadelphia; better fit, removable pads | Moderate to good cervical motion restriction |
| SOMI (Sternal-Occipital-Mandibular Immobilizer) | Upper/mid cervical fractures; limits flexion better than extension | Good flexion restriction; fair extension restriction |
| Cervicothoracic orthosis (CTO) / Minerva | Upper cervical fractures; post-surgical C-spine | Best cervical motion restriction short of halo |
| Halo vest | Unstable cervical fractures (C1–C2: odontoid, Hangman); gold standard for cervical immobilization | Best overall cervical motion restriction (~96%) |
| TLSO (Thoracolumbosacral Orthosis) | Thoracolumbar fractures (T6–L3); TLSO body jacket (Jewett hyperextension brace for compression fractures, custom molded TLSO for unstable fractures) | Limits thoracolumbar flexion/extension/rotation |
| LSO (Lumbosacral Orthosis) | Lumbar fractures, post-surgical lumbar support, chronic LBP (corset) | Limits lumbar flexion/extension |
36 Wheelchair Prescription & Seating
Manual vs Power Wheelchair
| Feature | Manual Wheelchair | Power Wheelchair |
|---|---|---|
| Best for | Adequate UE strength and endurance; paraplegic SCI (T1+); good cardiopulmonary fitness | Tetraplegia (C4–C6); progressive neuromuscular disease; severe deconditioning; limited UE function |
| Advantages | Lighter; easier transport; promotes UE fitness; lower cost; fewer mechanical failures | Independence for those unable to propel manual chair; less UE overuse injury; can accommodate tilt/recline |
| Disadvantages | UE overuse injuries (rotator cuff, CTS); limited for long distances if weak; inaccessible to those with limited UE function | Heavy (200–400 lb); requires van transport; expensive; battery maintenance; mechanical breakdowns |
Seating & Positioning
Proper seating is essential for posture, skin protection, and function. Key principles: seat width = widest point of hips + 1–2 inches; seat depth = posterior buttock to popliteal fossa − 2–3 inches (avoid popliteal pressure); seat height = allow proper footrest clearance and transfer height. Cushion selection for pressure redistribution: foam (basic, lightweight, inexpensive; limited lifespan), gel (good pressure distribution; heavy), air (ROHO — excellent pressure redistribution; requires maintenance, can bottom out if under-inflated), alternating pressure (powered; best for highest-risk patients). Tilt-in-space maintains hip/knee angles while tilting the entire seating system posteriorly for pressure relief and positioning (essential for patients unable to do weight shifts independently). Recline opens the seat-to-back angle for pressure relief, catheterization access, and spasm management, but creates shear forces on the skin.
Wheelchair Driving Interfaces
| Interface | SCI Level / Condition |
|---|---|
| Standard joystick | C6–C7 (with wrist extension / tenodesis) and below |
| Short-throw joystick | C5–C6 (limited hand function) |
| Head array | C4 (head/neck control intact) |
| Sip-and-puff | C3–C4 (diaphragmatic breathers); ventilator-dependent |
| Chin control | C4 (good head control) |
| Eye-tracking / switch scanning | Highest-level injuries; progressive neuromuscular diseases (ALS) |
37 Functional Outcome Measures
Functional Independence Measure (FIM)
The FIM is the most widely used functional assessment in rehabilitation, measuring the burden of care across 18 items in 6 domains. Each item is scored 1–7; total score ranges from 18 (complete dependence) to 126 (complete independence). FIM score change during rehabilitation admission is the primary measure of rehabilitation effectiveness.
FIM Scoring Scale
| Score | Level | Description |
|---|---|---|
| 7 | Complete Independence | Activity performed safely, without modification, assistive device, or helper; in reasonable time |
| 6 | Modified Independence | Activity requires assistive device, extra time, or safety concern; no helper needed |
| 5 | Supervision / Setup | Requires standby assist, cueing, coaxing, or setup (helper sets up items) only; no physical contact |
| 4 | Minimal Assist | Patient performs ≥75% of effort; helper provides <25% of effort (contact guard or light touch) |
| 3 | Moderate Assist | Patient performs 50–74% of effort; helper provides 25–50% |
| 2 | Maximal Assist | Patient performs 25–49% of effort; helper provides 50–75% |
| 1 | Total Assist | Patient performs <25% of effort; helper provides ≥75% |
FIM Items (18 Total)
| Domain | Items |
|---|---|
| Self-Care (6 items) | 1. Eating, 2. Grooming, 3. Bathing, 4. Dressing — Upper Body, 5. Dressing — Lower Body, 6. Toileting |
| Sphincter Control (2 items) | 7. Bladder Management, 8. Bowel Management |
| Transfers (3 items) | 9. Bed/Chair/Wheelchair, 10. Toilet, 11. Tub/Shower |
| Locomotion (2 items) | 12. Walk/Wheelchair, 13. Stairs |
| Communication (2 items) | 14. Comprehension (auditory/visual), 15. Expression (verbal/non-verbal) |
| Social Cognition (3 items) | 16. Social Interaction, 17. Problem Solving, 18. Memory |
Barthel Index
An older ADL measure, scored 0–100 (0 = complete dependence, 100 = independence). Ten items: feeding (10), bathing (5), grooming (5), dressing (10), bowels (10), bladder (10), toilet use (10), transfers (15), mobility/walking (15), stairs (10). A score ≥60 generally indicates potential for discharge home with some assistance. Simpler than FIM but less sensitive to change.
Other Key Outcome Measures
| Measure | What It Assesses | Scoring | Interpretation |
|---|---|---|---|
| Berg Balance Scale (BBS) | Static and dynamic balance; 14 tasks (sitting, standing, reaching, turning, single-leg stance) | Each item 0–4; total 0–56 | <36 = nearly 100% fall risk; 36–45 = moderate fall risk; >45 = low fall risk |
| Timed Up and Go (TUG) | Functional mobility: stand from chair, walk 3 meters, turn, walk back, sit down | Time in seconds | <10 s = freely mobile; 10–20 s = mostly independent; 20–30 s = variable; >30 s = impaired, requires assistive device |
| 6-Minute Walk Test (6MWT) | Submaximal exercise capacity; endurance; functional exercise tolerance | Distance walked (meters) in 6 minutes on a 30-meter course | Normal adults: 400–700 m; <300 m associated with increased mortality in heart/lung disease; change of 50–80 m considered clinically meaningful (MCID varies by condition) |
| 10-Meter Walk Test (10MWT) | Gait speed (comfortable and fast); strong predictor of community ambulation | Meters/second | <0.4 m/s = household ambulator; 0.4–0.8 m/s = limited community ambulator; >0.8 m/s = full community ambulator; >1.2 m/s = normal |
38 Classification Systems (All)
This section consolidates all major classification systems used in PM&R practice for rapid reference. Each is fully enumerated with all grades/levels.
ASIA Impairment Scale (AIS)
| Grade | Type | Definition |
|---|---|---|
| A | Complete | No sensory or motor function in S4–S5 |
| B | Sensory Incomplete | Sensory but not motor function below neurological level including S4–S5 |
| C | Motor Incomplete | Motor function below NLI; >50% of key muscles below NLI have grade <3 |
| D | Motor Incomplete | Motor function below NLI; ≥50% of key muscles below NLI have grade ≥3 |
| E | Normal | Normal sensory and motor function |
Modified Ashworth Scale
| Grade | Description |
|---|---|
| 0 | No increase in muscle tone |
| 1 | Slight increase; catch and release or minimal resistance at end ROM |
| 1+ | Slight increase; catch followed by minimal resistance through <50% ROM |
| 2 | More marked increase through most ROM; part easily moved |
| 3 | Considerable increase; passive movement difficult |
| 4 | Rigid in flexion or extension |
Brunnstrom Stages of Motor Recovery
| Stage | Description |
|---|---|
| 1 | Flaccidity; no voluntary movement |
| 2 | Spasticity developing; synergies beginning as associated reactions |
| 3 | Spasticity peaks; voluntary synergy patterns but no out-of-synergy movement |
| 4 | Spasticity declining; some movements deviating from synergy |
| 5 | Spasticity waning; isolated joint movements possible |
| 6 | Spasticity minimal; near-normal coordination |
| 7 | Normal motor function restored |
Rancho Los Amigos Levels of Cognitive Functioning
| Level | Description |
|---|---|
| I | No Response |
| II | Generalized Response |
| III | Localized Response |
| IV | Confused–Agitated |
| V | Confused–Inappropriate, Non-Agitated |
| VI | Confused–Appropriate |
| VII | Automatic–Appropriate |
| VIII | Purposeful–Appropriate (Stand-By Assistance) |
| IX | Purposeful–Appropriate (Stand-By Assistance on Request) |
| X | Purposeful–Appropriate (Modified Independent) |
Glasgow Coma Scale
| Component | Response | Score |
|---|---|---|
| Eye Opening | Spontaneous | 4 |
| To voice | 3 | |
| To pressure | 2 | |
| None | 1 | |
| Verbal | Oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Incomprehensible sounds | 2 | |
| None | 1 | |
| Motor | Obeys commands | 6 |
| Localizes pain | 5 | |
| Withdrawal | 4 | |
| Abnormal flexion | 3 | |
| Extension | 2 | |
| None | 1 |
GMFCS Levels (Cerebral Palsy)
| Level | Description |
|---|---|
| I | Walks without limitations |
| II | Walks with limitations; difficulty on uneven surfaces, inclines |
| III | Walks with handheld mobility device; wheelchair for long distances |
| IV | Self-mobility with limitations; wheelchair in most settings |
| V | Transported in wheelchair; severely limited voluntary control |
K-Levels (Medicare Functional Classification for Amputees)
| Level | Description |
|---|---|
| K0 | No ability/potential to ambulate or transfer |
| K1 | Household ambulator; transfers on level surfaces at fixed cadence |
| K2 | Limited community ambulator; traverses low-level barriers |
| K3 | Community ambulator with variable cadence; traverses most barriers |
| K4 | High-activity; exceeds basic ambulation skills (athletes, children) |
Seddon / Sunderland Classification of Nerve Injury
| Seddon | Sunderland | Pathology | Recovery |
|---|---|---|---|
| Neurapraxia | Grade I | Myelin damage only; no axonal loss | Complete; weeks to months |
| Axonotmesis | Grade II | Axon + myelin; endoneurium intact | Good; 1 mm/day regeneration |
| Axonotmesis | Grade III | Axon + myelin + endoneurium; perineurium intact | Variable |
| Neurotmesis | Grade IV | All internal structures; epineurium intact | Poor without surgery |
| Neurotmesis | Grade V | Complete nerve transection | None without surgical repair |
39 Medications Master Table
Antispasticity Agents
| Medication | Mechanism | Dosing | Key Side Effects | Monitoring |
|---|---|---|---|---|
| Baclofen (oral) | GABA-B agonist (spinal) | 5 mg TID → max 80–120 mg/day | Sedation, weakness, dizziness; withdrawal seizures (must taper) | Taper gradually; never abrupt discontinuation |
| Tizanidine | Alpha-2 agonist (central) | 2 mg TID → max 36 mg/day | Sedation, dry mouth, hypotension, hepatotoxicity | LFTs at baseline, 1, 3, 6 months |
| Dantrolene | Peripheral — blocks SR Ca++ release | 25 mg daily → max 400 mg/day | Weakness, diarrhea, hepatotoxicity (serious) | LFTs at baseline and q3 months |
| Diazepam | GABA-A enhancer | 2 mg BID → max 60 mg/day | Sedation, dependence, cognitive impairment | Avoid in TBI; limit duration |
| Baclofen (intrathecal) | GABA-B agonist (CSF delivery) | Trial 50–100 mcg bolus; maintenance 100–900 mcg/day | Overdose: sedation, respiratory depression; Withdrawal: life-threatening (see emergency box) | Pump refills; catheter integrity; withdrawal prevention |
| OnabotulinumtoxinA | Blocks ACh at NMJ | Total 400–600 U/session (see dosing table) | Weakness, injection site pain; rare: dysphagia, respiratory compromise if systemic spread | 12-week minimum between sessions |
Neuropathic Pain Medications
| Medication | Mechanism | Dosing | Key Side Effects |
|---|---|---|---|
| Gabapentin | Calcium channel alpha-2-delta ligand | 300 mg TID → max 3600 mg/day | Sedation, dizziness, peripheral edema, weight gain |
| Pregabalin | Same as gabapentin (more potent) | 75 mg BID → max 600 mg/day | Same as gabapentin; also euphoria (Schedule V) |
| Duloxetine | SNRI | 30 mg daily → max 120 mg/day | Nausea, dry mouth, constipation, hepatotoxicity |
| Amitriptyline | TCA (NE + 5-HT reuptake inhibition) | 10–25 mg HS → max 150 mg/day | Anticholinergic effects, sedation, cardiac arrhythmia, weight gain |
| Nortriptyline | TCA (less anticholinergic than amitriptyline) | 10–25 mg HS → max 150 mg/day | Similar to amitriptyline but better tolerated |
| Carbamazepine | Sodium channel blocker | 100 mg BID → max 1200 mg/day | Dizziness, diplopia, hyponatremia, aplastic anemia (rare), SJS (HLA-B*1502) |
| Capsaicin (topical) | TRPV1 agonist (substance P depletion) | Apply TID–QID to affected area | Burning sensation initially; improves with continued use |
| Lidocaine 5% patch | Sodium channel blocker (topical) | Up to 3 patches/day; 12 hours on, 12 hours off | Local skin irritation; minimal systemic absorption |
Neurogenic Bladder Agents
| Medication | Mechanism | Use | Key Side Effects |
|---|---|---|---|
| Oxybutynin | Anticholinergic/antimuscarinic | UMN (overactive) bladder; reduces detrusor contractions | Dry mouth, constipation, cognitive impairment (crosses BBB), blurred vision |
| Tolterodine | Antimuscarinic (more bladder-selective) | UMN bladder | Less dry mouth than oxybutynin; similar anticholinergic effects |
| Tamsulosin | Alpha-1 blocker | DSD (reduces sphincter resistance) | Orthostatic hypotension, dizziness, retrograde ejaculation |
| OnabotulinumtoxinA (intradetrusor) | Blocks ACh at detrusor NMJ | Refractory overactive/neurogenic bladder (200 U for neurogenic) | Urinary retention (may need CIC), UTI |
| Desmopressin (DDAVP) | Vasopressin analog | Nocturia; SCI patients with nocturnal polyuria | Hyponatremia (monitor sodium), fluid restriction needed |
Bowel Medications
| Medication | Type | Mechanism | Use in Neurogenic Bowel |
|---|---|---|---|
| Docusate sodium | Stool softener | Surfactant; increases water in stool | Daily maintenance; UMN and LMN bowel |
| Senna | Stimulant laxative | Stimulates colonic peristalsis | Daily or as needed; UMN bowel |
| Bisacodyl suppository | Stimulant | Direct rectal stimulation | Part of timed bowel program (UMN bowel) |
| Glycerin suppository | Osmotic/lubricant | Draws water into rectum | Bowel program adjunct |
| Polyethylene glycol (MiraLAX) | Osmotic laxative | Retains water in colon | Daily maintenance for constipation |
| Methylnaltrexone | Peripheral mu-opioid antagonist | Reverses opioid-induced constipation without crossing BBB | Opioid-induced constipation in palliative/SCI patients |
40 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AD | Autonomic Dysreflexia |
| ADL | Activities of Daily Living |
| AFO | Ankle-Foot Orthosis |
| AIS | ASIA Impairment Scale |
| AKA | Above-Knee Amputation (Transfemoral) |
| ASIA | American Spinal Injury Association |
| BBS | Berg Balance Scale |
| BKA | Below-Knee Amputation (Transtibial) |
| CIC | Clean Intermittent Catheterization |
| CIMT | Constraint-Induced Movement Therapy |
| CMAP | Compound Motor Action Potential |
| CP | Cerebral Palsy |
| CRPS | Complex Regional Pain Syndrome |
| CTS | Carpal Tunnel Syndrome |
| DI | Diabetes Insipidus |
| DSD | Detrusor-Sphincter Dyssynergia |
| DTI | Deep Tissue Injury |
| DVT | Deep Vein Thrombosis |
| EMG | Electromyography |
| ESI | Epidural Steroid Injection |
| FEES | Fiberoptic Endoscopic Evaluation of Swallowing |
| FES | Functional Electrical Stimulation |
| FIM | Functional Independence Measure |
| GCS | Glasgow Coma Scale |
| GMFCS | Gross Motor Function Classification System |
| GOAT | Galveston Orientation and Amnesia Test |
| HO | Heterotopic Ossification |
| IADL | Instrumental Activities of Daily Living |
| ICF | International Classification of Functioning, Disability, and Health |
| IRF | Inpatient Rehabilitation Facility |
| ISNCSCI | International Standards for Neurological Classification of SCI |
| ITB | Intrathecal Baclofen |
| KAFO | Knee-Ankle-Foot Orthosis |
| LMN | Lower Motor Neuron |
| LTAC | Long-Term Acute Care |
| MAS | Modified Ashworth Scale |
| MMT | Manual Muscle Testing |
| MUAP | Motor Unit Action Potential |
| NCS | Nerve Conduction Study |
| NLI | Neurological Level of Injury |
| NMES | Neuromuscular Electrical Stimulation |
| OT | Occupational Therapy |
| PE | Pulmonary Embolism |
| PSH | Paroxysmal Sympathetic Hyperactivity |
| PSW | Positive Sharp Wave |
| PT | Physical Therapy |
| PTA | Post-Traumatic Amnesia |
| RFA | Radiofrequency Ablation |
| ROM | Range of Motion |
| SCI | Spinal Cord Injury |
| SDR | Selective Dorsal Rhizotomy |
| SIADH | Syndrome of Inappropriate ADH Secretion |
| SLP | Speech-Language Pathology |
| SNAP | Sensory Nerve Action Potential |
| SNF | Skilled Nursing Facility |
| TBI | Traumatic Brain Injury |
| TENS | Transcutaneous Electrical Nerve Stimulation |
| THA | Total Hip Arthroplasty |
| TKA | Total Knee Arthroplasty |
| TLSO | Thoracolumbosacral Orthosis |
| TUG | Timed Up and Go |
| UMN | Upper Motor Neuron |
| VFSS | Videofluoroscopic Swallow Study |
| 6MWT | 6-Minute Walk Test |
41 Risk Factors & Comorbidities
Comorbidities Affecting Rehabilitation Outcomes
| Comorbidity | Impact on Rehabilitation | Management Considerations |
|---|---|---|
| Diabetes Mellitus | Impaired wound healing, increased infection risk, neuropathy (reduced balance/sensation), retinopathy (impaired vision), nephropathy, increased amputation risk, cardiovascular disease | Glycemic control (target HbA1c <7–8% in rehab); foot care education; neuropathic pain management; daily skin inspection |
| Depression | Reduced motivation, decreased participation in therapy, poorer functional outcomes (strongest psychosocial predictor of poor rehab outcomes), insomnia, fatigue, pain amplification | Screen with PHQ-9; SSRIs preferred (sertraline — fewest drug interactions); CBT; peer support; avoid TCAs in TBI (anticholinergic) |
| Cognitive Impairment | Impaired new learning, safety awareness deficits, difficulty following therapy instructions, reduced carryover, increased supervision needs | Simplified instructions; repetition; errorless learning; structured environment; caregiver training; compensatory strategies |
| Obesity | Increased joint loading, increased energy cost of ambulation, skin breakdown risk, difficult transfers, limited prosthetic/orthotic options, OSA | Weight management; bariatric equipment; appropriate weight limits for DME; nutritional counseling |
| Cardiovascular Disease | Exercise intolerance, limited therapy intensity, increased DVT/PE risk, medication interactions | Cardiac risk stratification; HR/BP monitoring during therapy; METs-based activity progression |
| Osteoporosis | Fracture risk (pathologic fractures with minimal trauma, especially in SCI below the level of injury), kyphotic posture, pain | Fall prevention; bone density screening; bisphosphonates/denosumab; vitamin D + calcium; weight-bearing when possible |
| Substance Use Disorder | Poor compliance, cognitive impairment, social instability, increased TBI/SCI risk, pain management challenges | Screening (CAGE, AUDIT); avoid opioids/benzodiazepines when possible; motivational interviewing; addiction medicine referral |
Risk Factors for Common Rehabilitation Conditions
| Condition | Key Risk Factors |
|---|---|
| Stroke | Hypertension (#1), AFib, diabetes, dyslipidemia, smoking, obesity, prior stroke/TIA, age, family history |
| SCI | Motor vehicle accidents (most common), falls (elderly), violence/GSW (urban), sports (diving), age peaks: 16–30 and >65 |
| TBI | Falls (most common overall, #1 in elderly), motor vehicle accidents (#1 in young adults), assault, sports, military/blast injury |
| Amputation | Peripheral vascular disease + diabetes (most common cause — 80% of amputations), trauma, malignancy, congenital |
| Pressure Injury | Immobility, sensory loss, incontinence, malnutrition (low albumin/prealbumin), shear/friction, age, moisture |
| DVT in Rehabilitation | Immobility, SCI, TBI, stroke, surgery (hip/knee), cancer, obesity, prior DVT, hypercoagulable state |
| Falls | Age >65, balance impairment (Berg <45), gait disorder, polypharmacy, visual impairment, cognitive impairment, orthostatic hypotension, environmental hazards |
Metabolic Equivalents (METs) for Activity Prescription
| METs | Activity Level | Examples |
|---|---|---|
| 1–2 | Very light | Eating, desk work, driving, standing |
| 2–3 | Light | Walking 2 mph, light housework, cooking |
| 3–5 | Moderate | Walking 3–4 mph, cycling (leisure), golf, gardening |
| 5–7 | Heavy | Jogging, swimming, singles tennis, mowing lawn |
| 7–10+ | Very heavy | Running, competitive sports, heavy manual labor, cross-country skiing |