Pulmonology / Critical Care
Every diagnosis, classification, procedure, ventilator mode, medication, scoring system, and management strategy in pulmonary and critical care medicine in one place.
01 Pulmonary Anatomy
The respiratory system extends from the nose and mouth to the alveolar surface area of approximately 70 m2 — roughly the size of a tennis court. Understanding its branching architecture, lobar anatomy, vascular supply, and lymphatic drainage is prerequisite to interpreting imaging, localizing disease, and performing procedures.
Airways — Conducting & Respiratory Zones
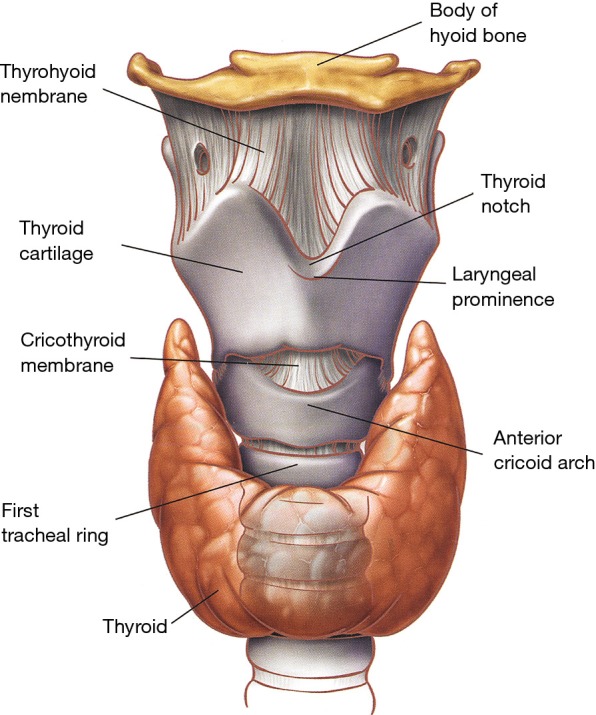
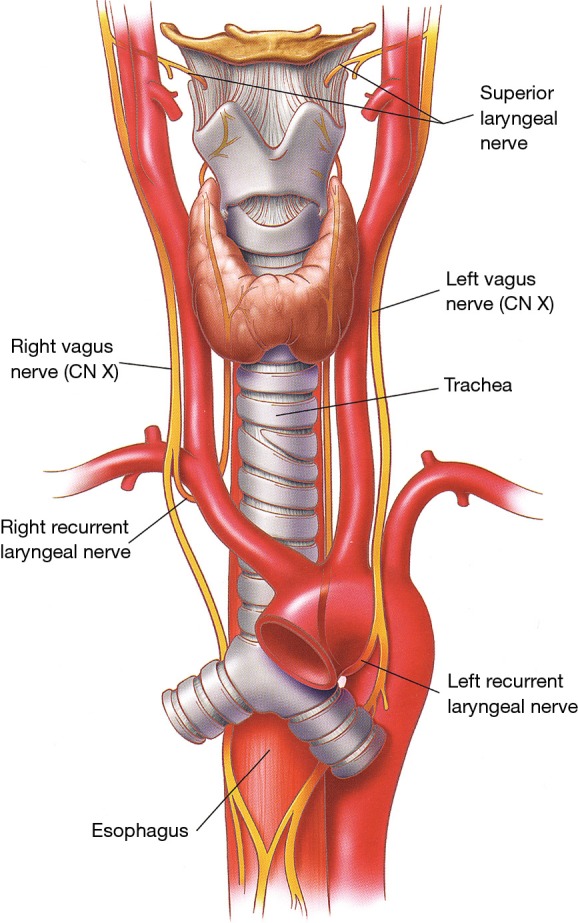
The trachea begins at the cricoid cartilage (C6), measures 10-12 cm in length and approximately 2 cm in diameter, and is supported by 16-20 C-shaped cartilaginous rings with a posterior membranous wall. It bifurcates at the carina (T4-T5 vertebral level) into the right and left mainstem bronchi. The right mainstem bronchus is wider, shorter (2.5 cm), and more vertical than the left (5 cm) — this is why aspirated material preferentially enters the right lung, and specifically the right lower lobe when the patient is upright, or the right upper lobe posterior segment when supine.
The bronchi divide through approximately 23 generations. The first 16 form the conducting zone (trachea, bronchi, bronchioles, terminal bronchioles) — no gas exchange occurs here; this is anatomic dead space (~150 mL). Generations 17-23 constitute the respiratory zone (respiratory bronchioles, alveolar ducts, alveolar sacs) where gas exchange occurs. The transition occurs at the terminal bronchiole, the smallest airway lacking alveoli. Cartilage disappears from the airway wall at the bronchiolar level; smooth muscle persists to the level of the alveolar ducts — this smooth muscle is the target of bronchodilators.
Lobes & Segments
The right lung has three lobes (upper, middle, lower) separated by the oblique (major) and horizontal (minor) fissures, containing 10 bronchopulmonary segments. The left lung has two lobes (upper, lower) separated by the oblique fissure, containing 8-10 segments; the left upper lobe includes the lingula (the anatomic equivalent of the right middle lobe).
| Lobe | Segments | Clinical Relevance |
|---|---|---|
| Right Upper (RUL) | Apical, Posterior, Anterior | Reactivation TB favors apical/posterior segments; Pancoast tumors arise in the apex |
| Right Middle (RML) | Lateral, Medial | RML syndrome (recurrent atelectasis/pneumonia from extrinsic lymph node compression) |
| Right Lower (RLL) | Superior, Medial basal, Anterior basal, Lateral basal, Posterior basal | Aspiration pneumonia (upright); most common location for pulmonary sequestration |
| Left Upper (LUL) | Apicoposterior, Anterior, Superior lingula, Inferior lingula | Lingula: analog of RML; lingular pneumonia can mimic LLL consolidation on CXR |
| Left Lower (LLL) | Superior, Anteromedial basal, Lateral basal, Posterior basal | Aspiration (supine — posterior segments bilaterally) |
Pulmonary Vasculature
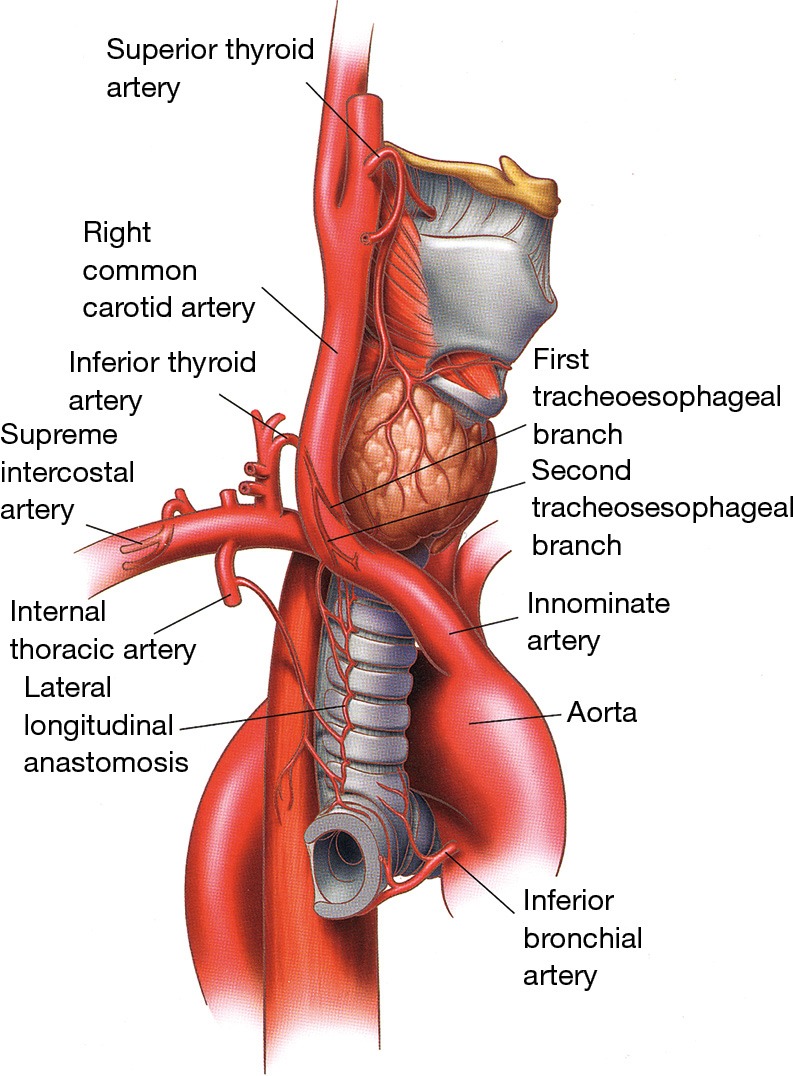
The lungs have a dual blood supply. The pulmonary arteries carry deoxygenated blood from the right ventricle and follow the bronchial tree, branching alongside each airway division to the alveolar capillary bed where gas exchange occurs. The pulmonary veins (4 total — 2 from each lung) drain oxygenated blood to the left atrium; they run between the lobes (intersegmental), not alongside the bronchi. The bronchial arteries (typically 1 left, 2 right, arising from the thoracic aorta or intercostal arteries) supply the airway walls, visceral pleura, and supporting structures with oxygenated blood. The bronchial circulation is the source of hemoptysis in bronchiectasis and is the target of bronchial artery embolization (BAE) for massive hemoptysis.
Lymphatics
Pulmonary lymph drains from the subpleural and peribronchovascular plexuses through intrapulmonary nodes → hilar (N1) nodes → mediastinal (N2) nodes → into the right lymphatic duct or thoracic duct. The lymph node stations are numbered 1-14 (IASLC map) and are critical for lung cancer staging. Station 7 (subcarinal) is accessible by EBUS-TBNA and is a key target for mediastinal staging.
The Diaphragm & Chest Wall
The diaphragm is the principal muscle of respiration, innervated by the phrenic nerve (C3-C5) — "C3, 4, 5 keeps the diaphragm alive." At rest, it sits at approximately T8-T10. During quiet inspiration it descends 1-2 cm; during forced inspiration, up to 10 cm. Diaphragmatic paralysis (phrenic nerve injury from thoracic surgery, trauma, neuralgic amyotrophy, or tumor infiltration) presents with orthopnea and a paradoxical rise of the affected hemidiaphragm on sniff test (fluoroscopy). Accessory muscles of inspiration include the external intercostals, sternocleidomastoid, and scalenes — their use at rest signals respiratory distress.
02 Pulmonary Physiology
Gas Exchange — The Alveolar-Capillary Interface
The alveolar-capillary membrane consists of surfactant → alveolar epithelium (type I pneumocyte) → basement membrane → capillary endothelium. Total thickness: 0.2-0.5 micrometers. Oxygen and CO2 exchange by passive diffusion along concentration gradients (Fick's law). Red blood cells transit the pulmonary capillary in approximately 0.75 seconds; gas exchange is normally complete within 0.25 seconds, providing a reserve for exercise or disease. This reserve is lost in diseases that thicken the membrane (fibrosis, edema) or reduce the capillary bed (emphysema, PE) — manifesting as exercise-induced desaturation before resting hypoxemia.
The alveolar gas equation: PAO2 = FiO2(Patm - PH2O) - (PaCO2 / R), where R (respiratory quotient) = 0.8 on a normal diet. At sea level on room air: PAO2 = 0.21(760 - 47) - (40/0.8) = ~100 mmHg. The A-a gradient = PAO2 - PaO2. Normal A-a gradient = 2.5 + (0.21 x age). An elevated A-a gradient indicates V/Q mismatch, shunt, or diffusion impairment — the three pathological causes of hypoxemia. A normal A-a gradient with hypoxemia means hypoventilation or low FiO2 (altitude).
Ventilation-Perfusion (V/Q) Matching
Ideal gas exchange requires matching of ventilation (V) and perfusion (Q). The normal V/Q ratio is approximately 0.8 (4 L/min alveolar ventilation / 5 L/min cardiac output). Gravity creates a gradient: in the upright lung, the apex is relatively over-ventilated and under-perfused (V/Q ~3.3), while the base is under-ventilated and over-perfused (V/Q ~0.6). Dead space (V/Q = infinity): ventilation without perfusion — as in PE, where blood flow to ventilated alveoli is blocked. Shunt (V/Q = 0): perfusion without ventilation — as in atelectasis, consolidated pneumonia, or ARDS. V/Q mismatch is the most common mechanism of hypoxemia and responds to supplemental O2; true shunt does not correct with O2 because blood bypasses ventilated alveoli entirely.
Compliance & Resistance
Compliance = change in volume / change in pressure. High compliance = easily expandable lung (emphysema — loss of elastic recoil). Low compliance = stiff lung (fibrosis, ARDS, pulmonary edema). Static compliance (measured during breath-hold) reflects lung and chest wall elastance. Dynamic compliance (measured during tidal breathing) also includes airway resistance. A drop in dynamic compliance with preserved static compliance = increased airway resistance (bronchospasm, mucus plugging). A drop in both = decreased lung compliance (worsening ARDS, tension pneumothorax, abdominal compartment syndrome).
Airway resistance is determined by Poiseuille's law: resistance is inversely proportional to the radius4. Halving the airway radius increases resistance 16-fold. This is why even modest bronchospasm, mucosal edema, or mucus plugging causes dramatic increases in the work of breathing. Medium-sized bronchi (generations 4-8) contribute the most to total airway resistance — not the smallest airways, which collectively have enormous cross-sectional area. The small airways are the "silent zone" — disease can be extensive before it is symptomatic or detectable on standard spirometry.
Oxygen Transport & the Oxyhemoglobin Dissociation Curve
Oxygen is carried in blood in two forms: dissolved (PaO2, only ~2% of total) and bound to hemoglobin (SaO2, ~98%). Factors that right-shift the curve (decreased O2 affinity, enhanced O2 delivery to tissues): increased temperature, increased 2,3-DPG, acidosis (Bohr effect), increased CO2. Factors that left-shift the curve (increased O2 affinity, impaired tissue delivery): hypothermia, decreased 2,3-DPG, alkalosis, CO poisoning, fetal hemoglobin, methemoglobinemia.
03 The Pulmonary Exam
Inspection
Respiratory rate (normal 12-20/min); pattern of breathing (Cheyne-Stokes, Kussmaul, Biot's); use of accessory muscles (sternocleidomastoid, intercostals, abdominal muscles); pursed-lip breathing (COPD — creates auto-PEEP to stent open airways); paradoxical abdominal movement (diaphragm fatigue — the abdomen moves inward during inspiration instead of outward); chest wall deformity (kyphoscoliosis, pectus excavatum/carinatum — both can cause restrictive physiology); barrel chest (hyperinflation in COPD — increased AP diameter, horizontal ribs); digital clubbing (lung cancer, IPF, bronchiectasis, cystic fibrosis — NOT seen in COPD or asthma); cyanosis (central = tongue/lips, requires deoxyhemoglobin > 5 g/dL).
Palpation & Percussion
Tactile fremitus: palpable vibration transmitted through the chest wall during phonation ("ninety-nine"). Increased by consolidation (solid tissue transmits sound better). Decreased by pleural effusion, pneumothorax, or thick chest wall. Percussion: resonant = normal lung; hyperresonant = pneumothorax or hyperinflation; dull = effusion, consolidation, or mass; flat/stony dull = large effusion. Tracheal shift: toward atelectasis (volume loss pulling mediastinum); away from tension pneumothorax or massive effusion (pressure pushing mediastinum).
Auscultation
| Finding | Sound | Mechanism | Seen In |
|---|---|---|---|
| Crackles (rales) | Discontinuous, popping | Opening of collapsed small airways / fluid in alveoli | Fine: IPF, early CHF. Coarse: pneumonia, bronchiectasis, late CHF |
| Wheezes | Continuous, high-pitched, musical | Airflow through narrowed airways | Asthma, COPD exacerbation, CHF ("cardiac asthma") |
| Rhonchi | Continuous, low-pitched, snoring | Secretions in large airways | Bronchitis, COPD; often clears with coughing |
| Stridor | Inspiratory, high-pitched, monophonic | Upper airway obstruction | Epiglottitis, tracheal tumor, post-extubation laryngeal edema |
| Pleural rub | Grating, "creaking leather" | Inflamed pleural surfaces rubbing | Pleurisy, PE with infarction, early effusion |
| Diminished / absent | Quiet or no breath sounds | Air or fluid barrier; no airflow | Effusion, pneumothorax, severe COPD, mainstem intubation |
| Bronchial breath sounds (over periphery) | Loud, tubular, expiratory > inspiratory | Consolidated lung transmitting bronchial sounds to periphery | Lobar pneumonia |
| Egophony ("E to A") | Spoken "E" heard as "A" | Sound filtering through consolidation or effusion above fluid level | Pneumonia, pleural effusion |
04 Pulmonary Function Tests
Spirometry
The cornerstone of pulmonary diagnostics. Measures FEV1 (forced expiratory volume in 1 second), FVC (forced vital capacity), and their ratio (FEV1/FVC). The patient takes a maximal inspiration then exhales as forcefully and completely as possible.
| Pattern | FEV1/FVC | FEV1 | FVC | Interpretation |
|---|---|---|---|---|
| Normal | ≥ 0.70 (or ≥ LLN) | Normal | Normal | No significant airflow limitation |
| Obstructive | < 0.70 | Decreased | Normal or decreased | COPD, asthma, bronchiectasis |
| Restrictive | Normal or increased | Decreased | Decreased | ILD, chest wall disease, neuromuscular (confirm with TLC) |
| Mixed | < 0.70 | Decreased | Decreased | Both obstruction and restriction; requires lung volumes for confirmation |
Lung Volumes
Measured by body plethysmography or helium dilution/nitrogen washout. TLC (total lung capacity): increased in hyperinflation (COPD/emphysema), decreased in restriction (ILD, chest wall, neuromuscular). RV (residual volume): the volume remaining after maximal exhalation — elevated in air trapping (COPD). FRC (functional residual capacity): the lung volume at end-normal exhalation — the balance point between lung elastic recoil (inward) and chest wall tendency to spring outward.
DLCO — Diffusing Capacity
Measures the ability of gas to transfer from alveoli to pulmonary capillary blood (using carbon monoxide as a tracer). Decreased in: emphysema (alveolar destruction), ILD/fibrosis (membrane thickening), pulmonary vascular disease (reduced capillary bed), anemia (less hemoglobin to bind CO). Increased in: pulmonary hemorrhage (free hemoglobin in alveoli binds CO), polycythemia, left-to-right shunt, asthma (increased capillary blood volume). DLCO is the most sensitive PFT for early emphysema and early ILD. An isolated low DLCO with otherwise normal PFTs should raise concern for pulmonary vascular disease or early emphysema.
Bronchodilator Response
Post-bronchodilator improvement of ≥ 12% and ≥ 200 mL in FEV1 or FVC indicates significant reversibility — supports asthma over COPD. However, partial reversibility can occur in COPD, and some asthma may not reverse acutely. The absence of reversibility does not exclude asthma; the presence of reversibility does not exclude COPD.
Methacholine Challenge
Provocation test for airway hyperresponsiveness. Methacholine (a muscarinic agonist) is inhaled in increasing concentrations; a ≥ 20% fall in FEV1 at a concentration ≤ 4 mg/mL = positive (PC20 ≤ 4). Excellent negative predictive value: a negative test essentially rules out asthma. Positive predictive value is lower — airway hyperresponsiveness occurs in COPD, post-viral cough, allergic rhinitis, and other conditions.
05 COPD Obstructive
Chronic obstructive pulmonary disease is defined by persistent airflow limitation (post-bronchodilator FEV1/FVC < 0.70) that is usually progressive and associated with an enhanced chronic inflammatory response in the airways and lungs to noxious particles or gases. COPD is the third leading cause of death worldwide. The two phenotypes — chronic bronchitis (airway inflammation, mucus hypersecretion, "blue bloater") and emphysema (alveolar destruction, loss of elastic recoil, "pink puffer") — usually coexist, though one may predominate.
GOLD Spirometric Classification
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2024 classifies airflow limitation severity as follows (all require post-bronchodilator FEV1/FVC < 0.70):
| GOLD Grade | FEV1 (% predicted) | Severity |
|---|---|---|
| GOLD 1 | ≥ 80% | Mild |
| GOLD 2 | 50-79% | Moderate |
| GOLD 3 | 30-49% | Severe |
| GOLD 4 | < 30% | Very severe |
GOLD ABE Assessment & Treatment Groups
GOLD 2024 replaced the ABCD groups with the ABE assessment tool, combining symptom burden and exacerbation history:
| Group | Exacerbation History | Symptoms (mMRC / CAT) | Initial Pharmacotherapy |
|---|---|---|---|
| A | 0-1 moderate (no hospitalization) | mMRC 0-1 / CAT < 10 | A bronchodilator (SABA, SAMA, LABA, or LAMA) |
| B | 0-1 moderate (no hospitalization) | mMRC ≥ 2 / CAT ≥ 10 | LABA + LAMA combination |
| E | ≥ 2 moderate OR ≥ 1 leading to hospitalization | Any | LABA + LAMA; consider LABA + LAMA + ICS if eosinophils ≥ 300 |
COPD Exacerbation Management
An exacerbation is an acute worsening of respiratory symptoms requiring additional therapy. Triggered by viral/bacterial infection (~70%), pollution, or unknown causes. Management: short-acting bronchodilators (albuterol + ipratropium nebulizers), systemic corticosteroids (prednisone 40 mg daily x 5 days — the REDUCE trial showed 5 days is equivalent to 14), antibiotics (if purulent sputum, need for mechanical ventilation, or severe exacerbation — azithromycin, doxycycline, or amoxicillin-clavulanate for 5-7 days), supplemental O2 targeting SpO2 88-92% (avoid hyperoxia in CO2 retainers — suppresses hypoxic drive and worsens hypercapnia via the Haldane effect), and noninvasive ventilation (NIV) for acute hypercapnic respiratory failure (pH < 7.35, PaCO2 > 45) — BiPAP reduces intubation rates and mortality.
Non-Pharmacologic Management
Smoking cessation is the single most effective intervention to slow FEV1 decline. Pulmonary rehabilitation (supervised exercise + education, 6-12 weeks) improves exercise capacity, dyspnea, and quality of life — indicated for GOLD B and E groups. Supplemental O2: long-term oxygen therapy (LTOT) ≥ 15 hr/day improves survival in COPD with resting PaO2 ≤ 55 mmHg or SpO2 ≤ 88% (or PaO2 56-59 with cor pulmonale/polycythemia). Lung volume reduction surgery (LVRS): improves survival in selected patients with upper-lobe-predominant emphysema and low exercise capacity (NETT trial). Endobronchial valves (Zephyr): bronchoscopic alternative to LVRS for heterogeneous emphysema without collateral ventilation.
06 Asthma Obstructive
Asthma is a chronic inflammatory disorder of the airways characterized by variable airflow obstruction, airway hyperresponsiveness, and airway remodeling. Unlike COPD, airflow limitation in asthma is typically reversible (spontaneously or with treatment). Affects ~300 million people worldwide with increasing prevalence in industrialized nations. The hallmark pathology is eosinophilic airway inflammation (type 2 inflammation), though neutrophilic and pauci-granulocytic phenotypes exist.
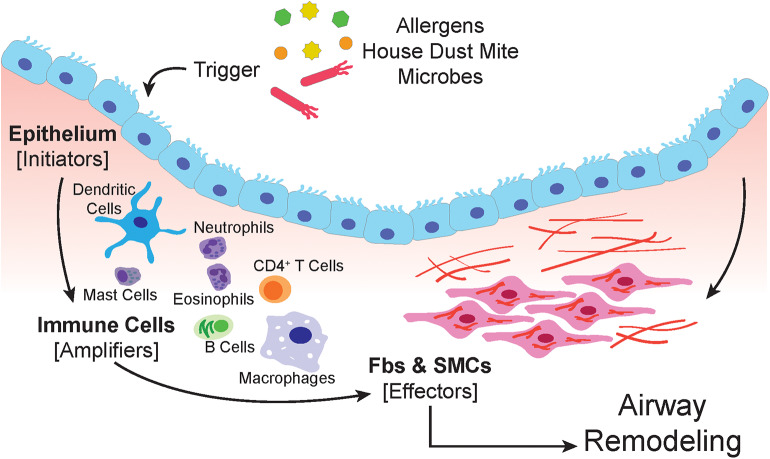
GINA Stepwise Treatment (2024)
The Global Initiative for Asthma (GINA) 2024 recommends ICS-containing therapy at ALL steps — SABA-only treatment is no longer recommended as a preferred option at any step:
| Step | Preferred Controller | Reliever | Patient Profile |
|---|---|---|---|
| 1 | As-needed low-dose ICS-formoterol | As-needed low-dose ICS-formoterol | Symptoms < 2x/month |
| 2 | Low-dose ICS daily, OR as-needed low-dose ICS-formoterol | As-needed low-dose ICS-formoterol | Symptoms ≥ 2x/month but not daily |
| 3 | Low-dose ICS-LABA (maintenance) | As-needed low-dose ICS-formoterol | Symptoms most days, or waking ≥ 1x/week |
| 4 | Medium-dose ICS-LABA | As-needed low-dose ICS-formoterol | Persistent symptoms despite Step 3 |
| 5 | High-dose ICS-LABA + add-on (LAMA, biologic, or low-dose OCS) | As-needed low-dose ICS-formoterol | Severe/refractory asthma, refer for phenotyping |
Asthma Severity Classification
| Severity | Daytime Symptoms | Nighttime Symptoms | FEV1 | Exacerbations |
|---|---|---|---|---|
| Intermittent | ≤ 2 days/week | ≤ 2x/month | ≥ 80% | 0-1/year |
| Mild persistent | > 2 days/week, not daily | 3-4x/month | ≥ 80% | ≥ 2/year |
| Moderate persistent | Daily | > 1x/week | 60-80% | ≥ 2/year |
| Severe persistent | Throughout the day | Often 7x/week | < 60% | Frequent |
Biologics for Severe Asthma
| Drug (Brand) | Target | Phenotype | Dose/Route | Key Pearl |
|---|---|---|---|---|
| Omalizumab (Xolair) | Anti-IgE | Allergic asthma, IgE 30-1500 | SC q2-4wk (dose by IgE/weight) | First biologic approved; anaphylaxis risk (~0.1%) |
| Mepolizumab (Nucala) | Anti-IL-5 | Eosinophilic (eos ≥ 150) | 100 mg SC q4wk | Also approved for EGPA |
| Benralizumab (Fasenra) | Anti-IL-5Rα | Eosinophilic (eos ≥ 150) | 30 mg SC q4wk x3, then q8wk | Depletes eosinophils via ADCC; fastest onset |
| Dupilumab (Dupixent) | Anti-IL-4Rα (blocks IL-4 & IL-13) | Eosinophilic or OCS-dependent | 200/300 mg SC q2wk | Also for atopic dermatitis, CRSwNP; may transiently increase eos |
| Tezepelumab (Tezspire) | Anti-TSLP | Severe asthma (all phenotypes) | 210 mg SC q4wk | First biologic effective regardless of eosinophil count or allergic status |
Acute Severe Asthma (Status Asthmaticus)
Life-threatening emergency. Management: continuous nebulized albuterol, ipratropium q20min x 3 then q4hr, systemic corticosteroids (methylprednisolone 125 mg IV or prednisone 60 mg PO), magnesium sulfate 2g IV over 20 min (smooth muscle relaxation), supplemental O2. Intubation if deteriorating despite maximal medical therapy — use ketamine for induction (bronchodilator properties), large ETT (minimize resistance), low RR with prolonged expiratory time (prevent dynamic hyperinflation/auto-PEEP/breath stacking). Heliox (helium-oxygen mixture) reduces airway resistance by decreasing gas density.
07 Bronchiectasis Obstructive
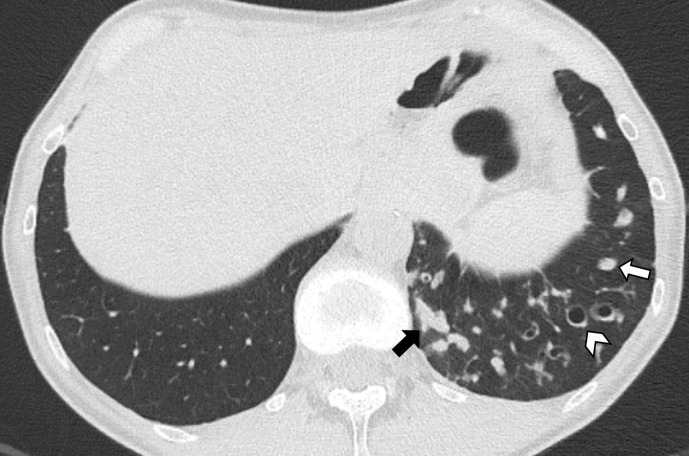
Permanent, abnormal dilation of bronchi caused by a cycle of infection, inflammation, and airway wall destruction. Presents with chronic productive cough with large volumes of purulent sputum (often "cupfuls"), recurrent respiratory infections, and hemoptysis (sometimes massive — bronchial artery erosion). CT chest is diagnostic: dilated airways with "signet ring" sign (airway lumen larger than its accompanying pulmonary artery), lack of normal airway tapering, and tree-in-bud opacities (mucus impaction in small airways).
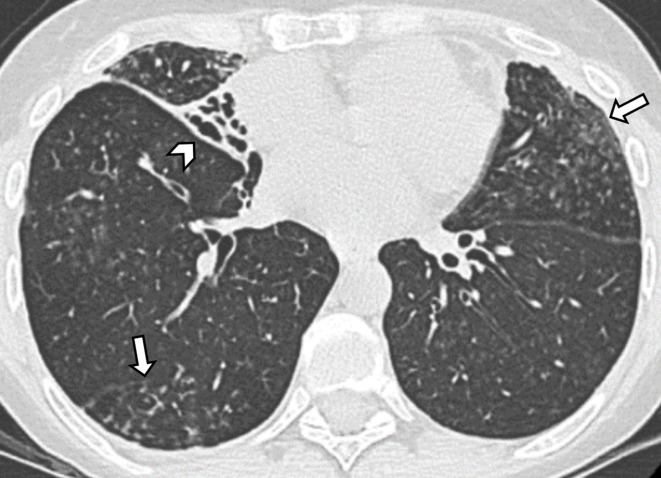
Etiologies: post-infectious (childhood pneumonia, TB, NTM), cystic fibrosis, primary ciliary dyskinesia (Kartagener syndrome = situs inversus + bronchiectasis + sinusitis), allergic bronchopulmonary aspergillosis (ABPA), immunodeficiency (CVID, HIV), autoimmune (RA, Sjogren's), alpha-1 antitrypsin deficiency. Workup: sputum cultures (including AFB and fungal), IgG/IgA/IgM, CF testing (sweat chloride, CFTR mutations), ABPA panel (total IgE, Aspergillus-specific IgE/IgG), alpha-1 antitrypsin level.
Management: airway clearance (chest physiotherapy, oscillatory PEP devices like Acapella/Flutter, high-frequency chest wall oscillation vest), chronic macrolide therapy (azithromycin 250 mg 3x/week — anti-inflammatory and reduces exacerbations; monitor QTc and hearing), inhaled antibiotics for chronic Pseudomonas (tobramycin nebulized 300 mg BID, alternating 28 days on/off), targeted antibiotics for acute exacerbations (culture-guided; empiric Pseudomonas coverage if colonized), bronchodilators for coexisting obstruction, and surgical resection for localized disease refractory to medical therapy. Massive hemoptysis (> 300-600 mL/24hr) requires bronchial artery embolization (BAE) and/or surgical resection.
08 Alpha-1 Antitrypsin Deficiency Obstructive
The most common genetic cause of COPD. Alpha-1 antitrypsin (AAT) is a serine protease inhibitor (SERPINA1 gene, chromosome 14) produced by the liver that protects lung tissue from neutrophil elastase. Deficiency allows unopposed elastase activity, leading to early-onset panacinar emphysema predominantly in the lower lobes — in contrast to smoking-related centrilobular emphysema which favors the upper lobes. Also causes liver disease (AAT polymers accumulate in hepatocytes — neonatal hepatitis, cirrhosis, hepatocellular carcinoma). Inherited in codominant fashion: normal allele = M; common deficiency alleles = S, Z. Pi*ZZ (homozygous Z) = < 15% of normal AAT, highest risk; Pi*MZ = heterozygous carrier, mild reduction, usually clinically insignificant unless smoking.
Screen all COPD patients, especially with early onset (< 45 yrs), lower-lobe-predominant emphysema, family history, or associated liver disease. Serum AAT level < 11 microM (57 mg/dL) = severe deficiency. Genotyping confirms phenotype. Treatment: smoking cessation (mandatory — smoking accelerates lung destruction dramatically), standard COPD therapy, and IV augmentation therapy (pooled human AAT — Prolastin-C, Zemaira, Glassia; 60 mg/kg IV weekly; slows emphysema progression in Pi*ZZ with FEV1 25-80% predicted). Lung transplantation for end-stage disease.
09 Idiopathic Pulmonary Fibrosis (IPF) ILD
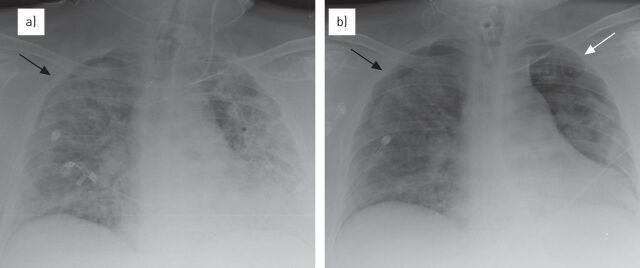
The most common and most lethal idiopathic interstitial pneumonia. Median survival 3-5 years from diagnosis — worse than many cancers. Affects predominantly older males (> 60 years), often with a history of smoking. Presents with progressive exertional dyspnea, dry cough, and bilateral inspiratory "Velcro" crackles at the lung bases. Digital clubbing is present in up to 50% of patients.
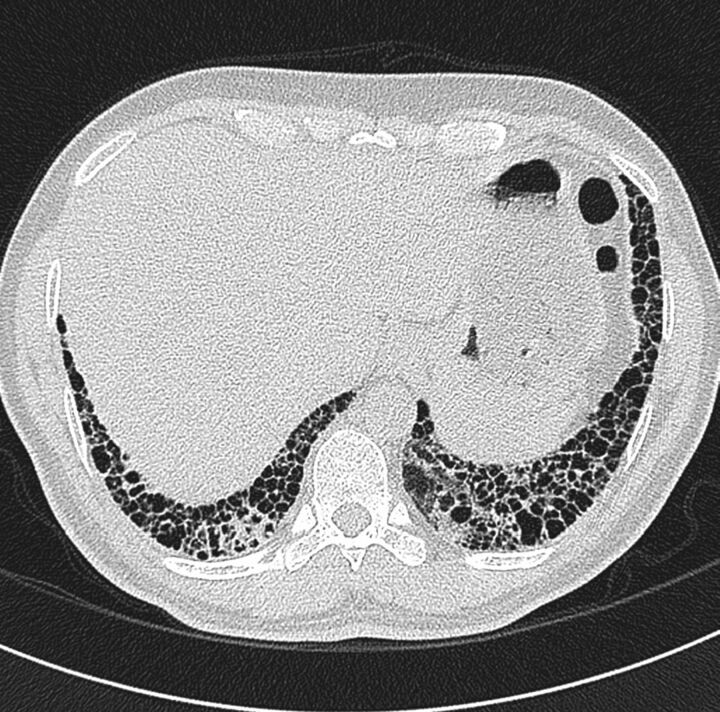
UIP Pattern on HRCT
The radiological hallmark is the usual interstitial pneumonia (UIP) pattern: bilateral, basal, peripheral predominant honeycombing (clustered thick-walled cystic airspaces), traction bronchiectasis, and reticulation with minimal ground-glass opacities. When a definite UIP pattern is present on HRCT in the appropriate clinical context, surgical lung biopsy is not required for diagnosis. A probable UIP pattern (reticulation and traction bronchiectasis but no honeycombing) may also be sufficient for diagnosis in the right clinical setting per the ATS/ERS/JRS/ALAT 2018 IPF guidelines.
Treatment
Two antifibrotic agents slow FVC decline by approximately 50% but neither reverses fibrosis or improves survival definitively: Pirfenidone (Esbriet) — anti-fibrotic, anti-inflammatory, and antioxidant; 801 mg TID with food; side effects include photosensitivity (strict sun avoidance), GI upset, and elevated LFTs. Nintedanib (Ofev) — tyrosine kinase inhibitor (VEGFR, FGFR, PDGFR); 150 mg BID with food; main side effect is diarrhea (often severe — loperamide as first-line management; dose reduce to 100 mg BID if needed); also monitor LFTs. Immunosuppression (prednisone, azathioprine, NAC) was proven harmful in IPF by the PANTHER-IPF trial — increased mortality and hospitalizations. Lung transplant referral for all appropriate patients at diagnosis.
Acute worsening of dyspnea within 30 days, with new bilateral ground-glass opacities on HRCT superimposed on the underlying UIP pattern, not fully explained by heart failure or fluid overload. Mortality exceeds 50%. No proven effective treatment — high-dose corticosteroids are commonly used empirically but evidence is weak. Rule out infection, PE, heart failure, and drug toxicity.
10 Nonspecific Interstitial Pneumonia (NSIP) ILD
The second most common idiopathic interstitial pneumonia, with significantly better prognosis than IPF (5-year survival ~80% for cellular NSIP, ~60% for fibrotic NSIP). Affects younger patients (40-50s), more women, and is often associated with connective tissue disease (scleroderma, polymyositis/dermatomyositis, RA, Sjogren's). HRCT shows bilateral, symmetric ground-glass opacities with subpleural sparing (a key distinguishing feature from UIP) and lower-lobe predominance. Unlike UIP, honeycombing is absent or minimal. Treatment: corticosteroids (prednisone 0.5-1 mg/kg/day tapered over months) plus steroid-sparing agents (mycophenolate, azathioprine) for fibrotic NSIP or recurrence. Response to immunosuppression is far better than in IPF.
11 Hypersensitivity Pneumonitis ILD
An immune-mediated lung disease caused by repeated inhalation of organic antigens in sensitized individuals. Common culprits: bird proteins (pigeon/parrot fancier's lung), Thermophilic actinomycetes in moldy hay (farmer's lung), hot tub mist (Mycobacterium avium complex), isocyanates, and mold/fungal antigens. Classified as non-fibrotic (formerly acute/subacute — ground-glass opacities, centrilobular nodules, air trapping on expiratory CT, lymphocytic BAL) and fibrotic (formerly chronic — honeycombing, traction bronchiectasis, fibrosis often in upper/mid lung zones). Antigen identification and avoidance is the most critical step. For non-fibrotic HP: antigen avoidance alone may suffice; corticosteroids for symptomatic disease. For fibrotic HP: prednisone plus mycophenolate or azathioprine; nintedanib has shown benefit in progressive fibrosing HP (INBUILD trial). Prognosis of fibrotic HP is intermediate between IPF and NSIP.
12 Sarcoidosis ILD
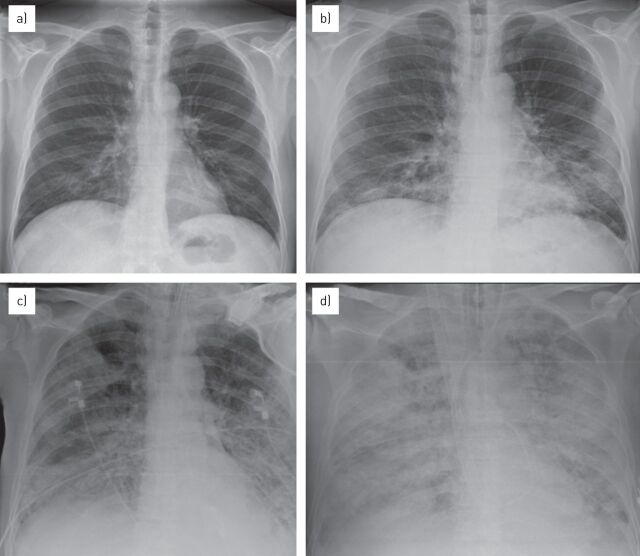
A systemic granulomatous disease of unknown etiology characterized by noncaseating granulomas in affected organs — lungs (90%+), lymph nodes, skin, eyes, liver, nervous system, heart. Most common in African Americans (3:1) and Northern Europeans, peak onset 25-40 years. The pulmonary presentation is bilateral hilar lymphadenopathy with or without parenchymal infiltrates. Extrapulmonary manifestations that every clinician must screen for: anterior uveitis (slit-lamp exam), skin lesions (erythema nodosum — good prognosis; lupus pernio — chronic disease marker), hypercalcemia (granulomas produce 1,25-dihydroxyvitamin D), cardiac sarcoidosis (conduction abnormalities, cardiomyopathy — get ECG at baseline), and neurosarcoidosis (CN VII palsy most common, diabetes insipidus).
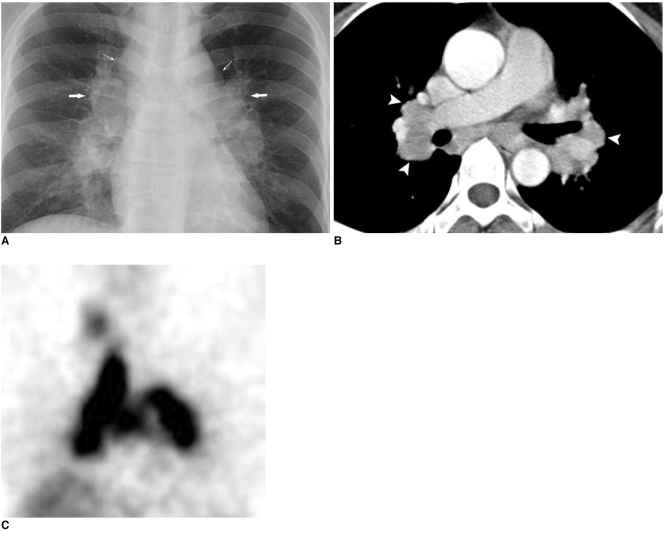
Scadding Radiographic Staging
| Stage | CXR Finding | Spontaneous Resolution Rate | Clinical Notes |
|---|---|---|---|
| 0 | Normal | N/A (extrapulmonary sarcoid) | Disease may be active in other organs |
| I | Bilateral hilar lymphadenopathy (BHL) alone | 55-90% | Most patients present here; often asymptomatic |
| II | BHL + pulmonary infiltrates | 40-70% | Infiltrates usually upper/mid zone predominant |
| III | Pulmonary infiltrates without BHL | 10-20% | Lymphadenopathy has regressed but parenchymal disease persists |
| IV | Pulmonary fibrosis (honeycombing, volume loss, bullae) | 0% | Irreversible; associated with pulmonary hypertension |
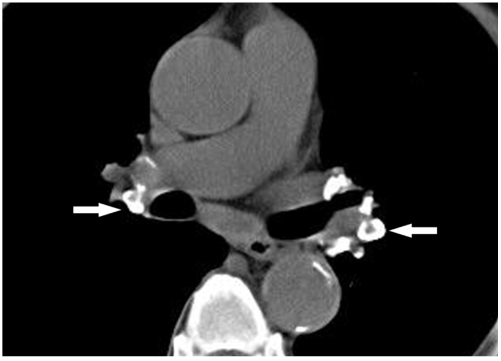
Diagnosis: tissue biopsy showing noncaseating granulomas + compatible clinical/radiographic picture + exclusion of other granulomatous diseases (TB, fungal, foreign body). Transbronchial biopsy via bronchoscopy is first-line; BAL showing CD4/CD8 ratio > 3.5 is supportive. Serum ACE level is elevated in ~60% but lacks specificity.
Treatment: many patients require no treatment (observation for Stage I). Indications for systemic therapy: significant pulmonary symptoms with declining PFTs, cardiac/neurologic/ocular involvement, hypercalcemia, disfiguring skin disease. First-line: prednisone 20-40 mg daily x 4-6 weeks then taper over 6-12 months. Steroid-sparing: methotrexate (preferred), azathioprine, mycophenolate. Refractory: infliximab (anti-TNF-alpha). Hydroxychloroquine for skin and joint disease.
13 Occupational Lung Disease ILD
Asbestosis
Diffuse interstitial fibrosis from asbestos fiber inhalation (shipyard workers, insulation, construction, automotive brakes). Latency period 15-20 years after exposure. HRCT shows bilateral lower-lobe predominant reticular opacities and honeycombing (UIP-like pattern). Pleural plaques (calcified, often bilateral, diaphragmatic) are the hallmark radiographic finding of asbestos exposure — they indicate exposure but not disease severity. Associated malignancies: mesothelioma (strongly linked, long latency ~30-40 years) and lung cancer (risk is multiplicative with smoking — asbestos exposure x smoking = ~50-fold increased lung cancer risk vs. 10-fold for smoking alone).
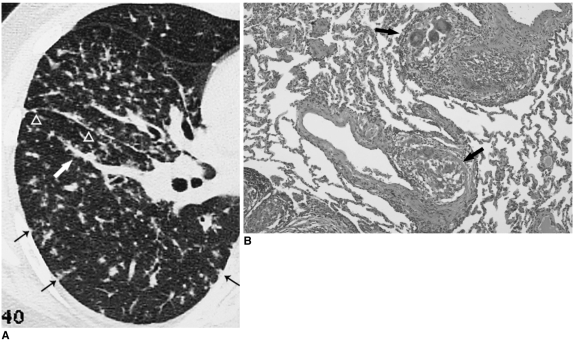
Silicosis
Caused by inhalation of crystalline silica (quartz) — sandblasting, mining, stone cutting, foundry work. Three forms: chronic/simple (10-30 years exposure; small round upper-lobe nodules; "eggshell" calcification of hilar lymph nodes is pathognomonic), accelerated (5-10 years, more aggressive), and acute/silicoproteinosis (< 5 years of massive exposure; alveolar filling pattern resembling pulmonary alveolar proteinosis; rapidly fatal). Complicated silicosis shows progressive massive fibrosis (PMF) — large conglomerate masses in the upper lobes. Associated with increased risk of TB (silica impairs macrophage function).
Coal Workers' Pneumoconiosis
Coal dust inhalation produces coal macules (collections of carbon-laden macrophages around respiratory bronchioles). Simple CWP: small round opacities on CXR, upper-lobe predominant, usually asymptomatic. Complicated CWP / PMF: large opacities (> 1 cm), often with background emphysema, progressive respiratory impairment. Caplan syndrome: CWP + rheumatoid arthritis + large peripheral lung nodules (rheumatoid pneumoconiosis). No specific treatment exists for any pneumoconiosis — management is exposure elimination, supportive care, and treatment of complications.
14 Community-Acquired Pneumonia (CAP) Infectious
The most common infectious cause of death in the developed world. Common pathogens: Streptococcus pneumoniae (most common identifiable cause), Mycoplasma pneumoniae (atypical; younger patients, walking pneumonia), Haemophilus influenzae (COPD patients), Legionella pneumophila (contaminated water systems; hyponatremia, diarrhea, liver dysfunction), Staphylococcus aureus (post-influenza superinfection, cavitary), respiratory viruses (influenza, SARS-CoV-2, RSV).
Severity Scoring — CURB-65
| Criterion | Points | Definition |
|---|---|---|
| Confusion | 1 | New disorientation to person, place, or time |
| Urea | 1 | BUN > 20 mg/dL (> 7 mmol/L) |
| Respiratory rate | 1 | ≥ 30/min |
| Blood pressure | 1 | SBP < 90 or DBP ≤ 60 mmHg |
| Age | 1 | ≥ 65 years |
Score 0-1: outpatient. Score 2: consider admission. Score 3-5: hospital admission; score ≥ 4 consider ICU.
PSI/PORT Score
The Pneumonia Severity Index uses 20 variables (age, comorbidities, physical exam findings, labs) to classify patients into Risk Classes I-V. Classes I-II: outpatient. Class III: brief observation. Classes IV-V: inpatient. PSI is better validated for identifying low-risk patients who can be treated as outpatients.
Empiric Treatment (ATS/IDSA 2019)
| Setting | Regimen | Duration |
|---|---|---|
| Outpatient, no comorbidities | Amoxicillin 1g TID, OR doxycycline 100mg BID, OR azithromycin 500mg x1 then 250mg daily (if local resistance < 25%) | 5 days minimum (treat until afebrile ≥ 48hr and ≤ 1 sign of instability) |
| Outpatient, with comorbidities | Amoxicillin-clavulanate 875/125 BID + azithromycin, OR respiratory fluoroquinolone (levofloxacin 750mg daily or moxifloxacin 400mg daily) | 5 days |
| Inpatient, non-ICU | Beta-lactam (ceftriaxone 1-2g IV daily, or ampicillin-sulbactam 3g IV q6h) + azithromycin 500mg IV daily, OR respiratory FQ monotherapy | 5 days |
| Inpatient, ICU | Beta-lactam (ceftriaxone or ampicillin-sulbactam) + azithromycin, OR beta-lactam + respiratory FQ. If MRSA risk: add vancomycin or linezolid. If Pseudomonas risk: piperacillin-tazobactam, cefepime, or meropenem + azithromycin or FQ | 5-7 days |
15 Hospital- & Ventilator-Associated Pneumonia Infectious
Hospital-acquired pneumonia (HAP): develops ≥ 48 hours after hospital admission, not intubated. Ventilator-associated pneumonia (VAP): develops ≥ 48 hours after endotracheal intubation. Both are caused by a different pathogen spectrum than CAP: MRSA, Pseudomonas aeruginosa, Klebsiella, Acinetobacter, Enterobacter, and other MDR gram-negatives. VAP incidence: 5-20 per 1,000 ventilator-days. Diagnosis is clinical: new/worsening infiltrate on CXR + at least 2 of: fever > 38C, leukocytosis (> 12,000) or leukopenia (< 4,000), purulent secretions. Obtain lower respiratory tract cultures before starting antibiotics (endotracheal aspirate or BAL).
Empiric therapy: antipseudomonal beta-lactam (piperacillin-tazobactam, cefepime, or meropenem) ± additional gram-negative agent (aminoglycoside or antipseudomonal FQ) if MDR risk + MRSA coverage (vancomycin or linezolid) if MRSA risk. De-escalate at 48-72 hours based on culture results. Duration: 7 days for uncomplicated VAP (short course reduces resistance emergence without worsening outcomes).
VAP prevention bundle: head-of-bed elevation 30-45 degrees, oral care with chlorhexidine, daily sedation interruption and spontaneous breathing trial assessment, DVT and stress ulcer prophylaxis, subglottic secretion drainage (ETT with suction port above cuff), minimize duration of mechanical ventilation.
16 Lung Abscess Infectious
A circumscribed collection of pus within the lung parenchyma, usually resulting from aspiration of oropharyngeal contents. Classic patient: poor dentition, alcoholism, impaired consciousness (seizures, overdose, anesthesia), dysphagia. Location: dependent lung segments — posterior segment of right upper lobe and superior segment of both lower lobes (aspiration while supine). CXR/CT shows a thick-walled cavity with an air-fluid level. Anaerobes are the predominant organisms (Fusobacterium, Peptostreptococcus, Prevotella, Bacteroides); polymicrobial is common.
Treatment: prolonged antibiotics — clindamycin 600mg IV q8h (or ampicillin-sulbactam, or metronidazole + amoxicillin) transitioned to oral when improving, total duration 4-6 weeks or until cavity has resolved or stabilized on imaging. CT-guided percutaneous drainage for large abscesses (> 6 cm) failing antibiotics. Surgical resection (lobectomy) for massive hemoptysis, failure to respond, or suspected malignancy.
17 Tuberculosis Infectious
Caused by Mycobacterium tuberculosis, an acid-fast bacillus transmitted by airborne droplet nuclei. One-quarter of the world's population has latent TB infection. In the US, TB disproportionately affects immigrants from endemic areas, homeless populations, incarcerated individuals, and immunocompromised patients (especially HIV).
Latent TB Infection (LTBI) vs. Active TB
| Feature | LTBI | Active TB |
|---|---|---|
| Symptoms | None | Cough > 2-3 weeks, hemoptysis, night sweats, weight loss, fevers |
| CXR | Normal (or Ghon complex/calcified granuloma) | Upper-lobe cavitary infiltrate (reactivation), hilar LAD ± effusion (primary) |
| AFB smear/culture | Negative | Positive (3 sputum specimens, early morning) |
| Infectiousness | Not infectious | Infectious (especially if cavitary, smear-positive) |
| TST/IGRA | Positive | Positive (but can be falsely negative in severe/miliary TB) |
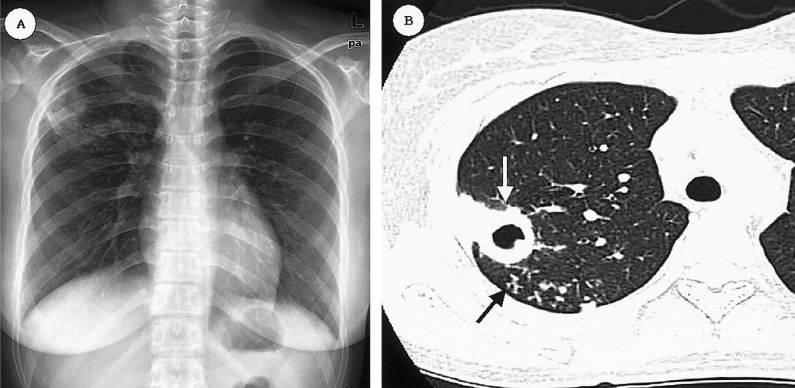
Treatment Regimens
LTBI (treat to prevent reactivation — 5-10% lifetime risk, higher with immunosuppression): preferred regimens include 3HP (isoniazid + rifapentine weekly x 12 weeks — directly observed), 4R (rifampin daily x 4 months), or 9H (isoniazid daily x 9 months; oldest regimen, worst completion rates). Pyridoxine (vitamin B6) 25-50 mg daily prevents INH-associated peripheral neuropathy.
Active TB: RIPE = Rifampin + Isoniazid + Pyrazinamide + Ethambutol for the intensive phase (2 months), then Rifampin + Isoniazid for the continuation phase (4 months) — total 6 months for drug-susceptible pulmonary TB. Monitor LFTs (INH/RIF hepatotoxicity), visual acuity and color vision (ethambutol optic neuritis), uric acid (pyrazinamide). Airborne isolation until 3 consecutive negative AFB sputum smears collected 8-24 hours apart.
18 Fungal Pulmonary Infections Infectious
Histoplasmosis
Endemic to the Ohio and Mississippi River valleys; Histoplasma capsulatum lives in soil contaminated with bat/bird droppings (caves, chicken coops). Most infections asymptomatic or mild flu-like illness. Severe acute pneumonia with diffuse reticulonodular infiltrates occurs in high-inoculum exposure. Chronic pulmonary histoplasmosis mimics TB (upper-lobe cavities in COPD patients). Disseminated histoplasmosis occurs in immunocompromised (pancytopenia, hepatosplenomegaly, mucosal ulcers). Diagnosis: Histoplasma urine/serum antigen (best for acute/disseminated), serology (complement fixation, immunodiffusion), culture. Treatment: mild = observation; moderate-severe = itraconazole 200 mg BID x 6-12 weeks; severe/disseminated = amphotericin B followed by itraconazole.
Coccidioidomycosis (Valley Fever)
Coccidioides immitis/posadasii, endemic to southwestern US, Mexico, Central/South America. Arthroconidia inhaled from disturbed soil. 60% asymptomatic; 40% develop pneumonia with desert bumps (erythema nodosum), arthralgias ("desert rheumatism"), and eosinophilia. Complications: cavitary disease (thin-walled cavities, often resolve spontaneously), dissemination to meninges (chronic basilar meningitis — lifelong fluconazole) and bone/joints. Risk of dissemination: Filipino and African American men, pregnant women (3rd trimester), immunosuppressed. Diagnosis: serology (IgM by EIA, IgG complement fixation — rising titers = active disease). Treatment: most primary infections resolve without therapy; fluconazole or itraconazole for symptomatic/prolonged disease; amphotericin B for severe or disseminated disease.
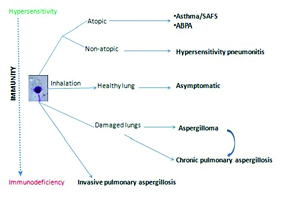
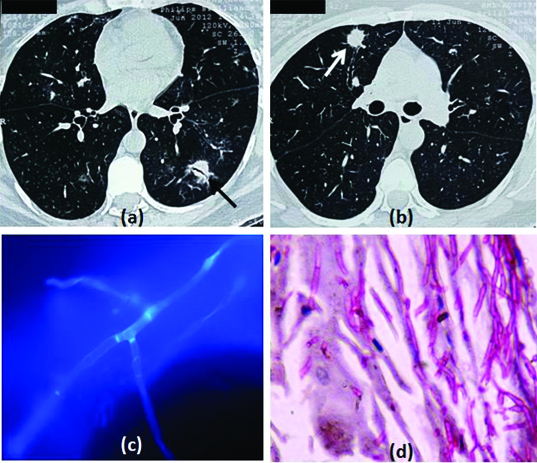
Aspergillosis — Spectrum of Disease
| Type | Host | Pathology | Diagnosis | Treatment |
|---|---|---|---|---|
| Allergic Bronchopulmonary Aspergillosis (ABPA) | Asthma/CF | Hypersensitivity to Aspergillus; central bronchiectasis, mucoid impaction | Total IgE > 1000, Aspergillus IgE/IgG positive, eosinophilia, central bronchiectasis on CT | Systemic corticosteroids ± itraconazole |
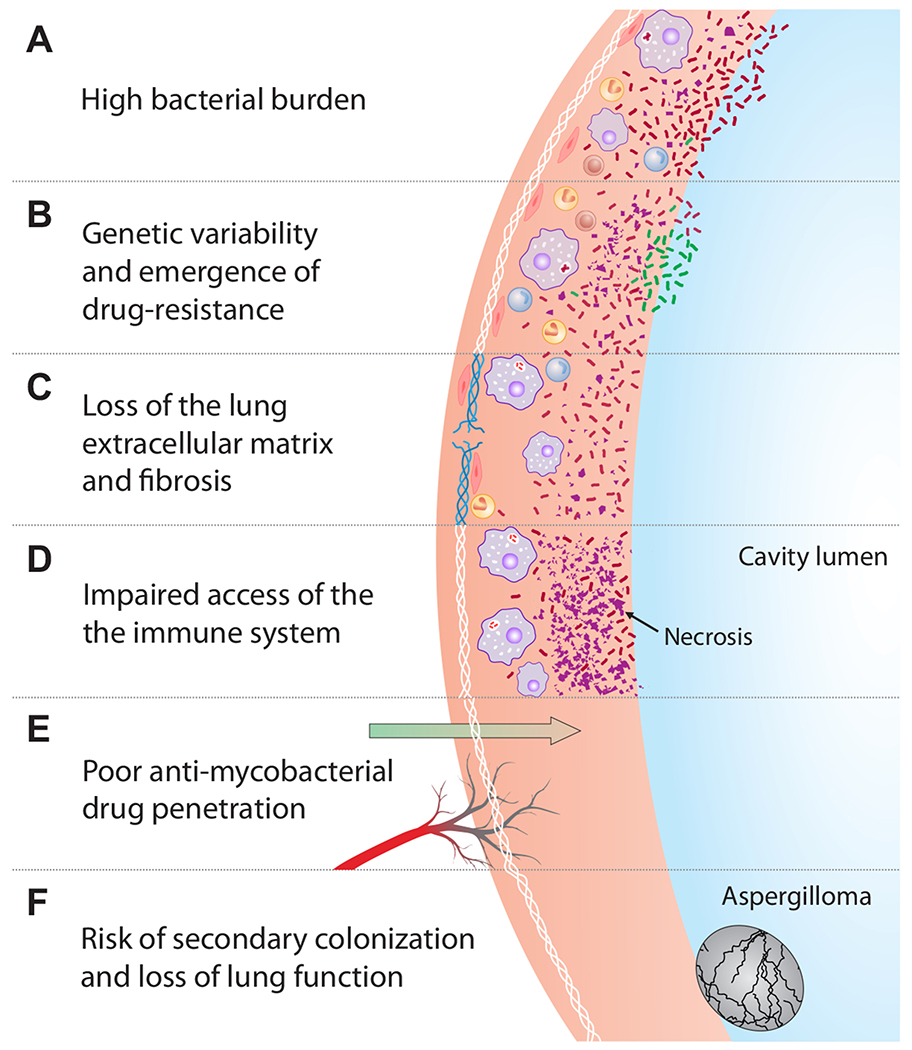
| Aspergilloma (fungus ball) | Pre-existing cavity (TB, sarcoid) | Saprophytic colonization of a cavity; no tissue invasion | CT: solid mass within a cavity with "air crescent" sign; Aspergillus IgG positive | Observation if asymptomatic; surgical resection for hemoptysis; BAE for massive hemoptysis |
| Invasive Pulmonary Aspergillosis (IPA) | Severely immunocompromised (neutropenic, transplant, high-dose steroids) | Angioinvasive — hyphae invade blood vessels → hemorrhagic infarction | CT: "halo sign" (early), "air crescent" sign (late); serum galactomannan; BAL galactomannan; biopsy with septate hyphae at 45° angles | Voriconazole (first-line); isavuconazole alternative; amphotericin B if azole-resistant |
| Chronic Pulmonary Aspergillosis (CPA) | Mildly immunocompromised or structural lung disease | Slowly progressive cavitary disease without overt immunosuppression | Elevated Aspergillus IgG, progressive cavitary changes over months | Long-term itraconazole or voriconazole (6+ months) |
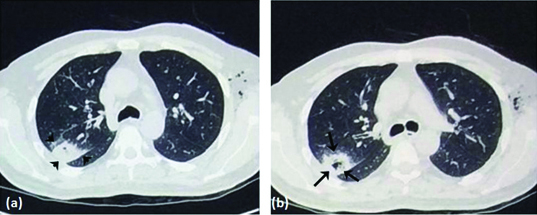
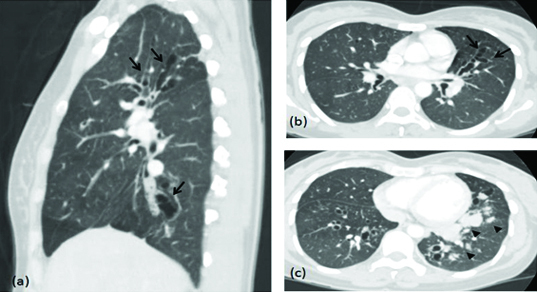
19 Pneumocystis Pneumonia (PJP) Infectious
Caused by Pneumocystis jirovecii (formerly P. carinii — hence the persistent abbreviation PCP/PJP). Occurs in severely immunocompromised patients: HIV with CD4 < 200, solid organ transplant, prolonged high-dose corticosteroids (≥ 20 mg prednisone daily for ≥ 3 weeks), hematologic malignancy on chemotherapy. In HIV, onset is insidious (weeks of progressive dyspnea, dry cough, low-grade fever). In non-HIV immunosuppressed, onset is often more acute and fulminant with higher mortality.
Diagnosis: bilateral diffuse ground-glass opacities on CT (classically diffuse and symmetric), markedly elevated serum LDH (> 500), elevated beta-D-glucan, SpO2 drops with ambulation. Definitive diagnosis: induced sputum or BAL with silver stain (cysts) or DFA (direct fluorescent antibody). PCR on BAL is highly sensitive. Treatment: TMP-SMX (trimethoprim 15-20 mg/kg/day divided q6-8h x 21 days in HIV, 14 days in non-HIV). If PaO2 < 70 mmHg or A-a gradient > 35: add adjunctive prednisone (40 mg BID x 5 days, then 40 mg daily x 5, then 20 mg daily x 11 — reduces mortality). Alternatives for TMP-SMX intolerance: pentamidine IV, atovaquone PO, dapsone + trimethoprim.
Prophylaxis: TMP-SMX DS 1 tablet daily (or 3x/week). Indications: HIV with CD4 < 200, solid organ transplant recipients (6-12 months minimum, often lifelong if on heavy immunosuppression), patients on ≥ 20 mg prednisone equivalent for ≥ 1 month.
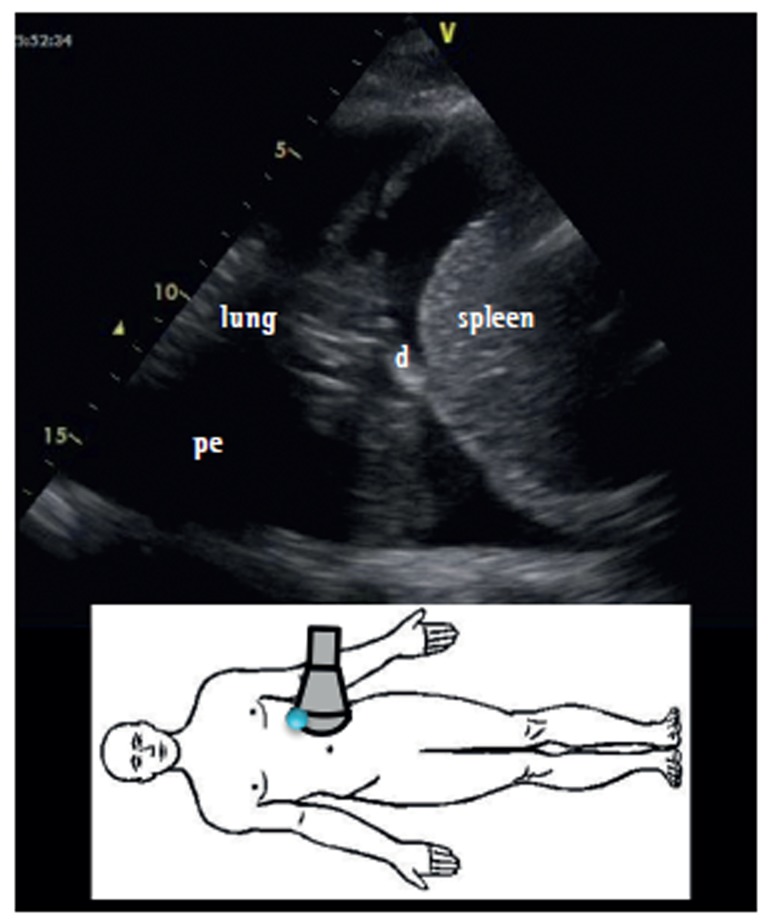
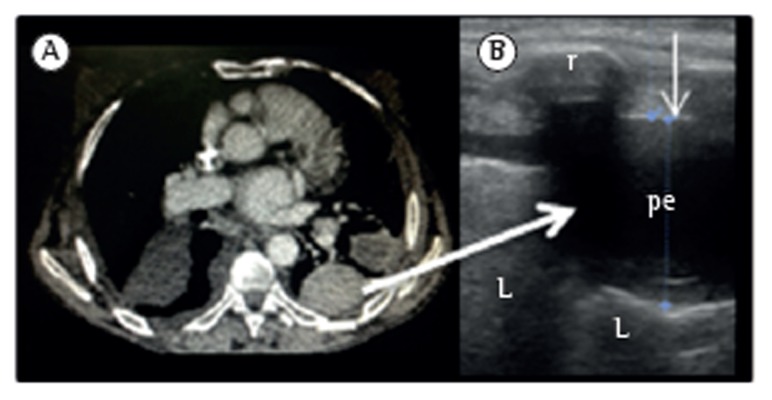
20 Pleural Effusion Pleural
Abnormal accumulation of fluid in the pleural space. Detected on CXR when ≥ 200-300 mL (blunting of the costophrenic angle on upright PA film); lateral decubitus can detect as little as 50 mL. Ultrasound is the best bedside tool for detecting, characterizing, and guiding drainage of effusions.
Light's Criteria — Exudate vs. Transudate
Pleural fluid is an exudate if ANY of the following are met (Light, 1972):
| Criterion | Exudate Threshold |
|---|---|
| Pleural fluid protein / serum protein | > 0.5 |
| Pleural fluid LDH / serum LDH | > 0.6 |
| Pleural fluid LDH | > 2/3 the upper limit of normal for serum LDH |
Common Causes by Category
| Transudates | Exudates |
|---|---|
| CHF (most common overall) | Parapneumonic / empyema |
| Hepatic hydrothorax (cirrhosis) | Malignancy (lung, breast, lymphoma) |
| Nephrotic syndrome | Tuberculosis |
| Peritoneal dialysis | Pulmonary embolism |
| Myxedema | Rheumatoid / lupus pleuritis |
| Constrictive pericarditis | Chylothorax (triglycerides > 110 mg/dL) |
Pleural Fluid Analysis — The Diagnostic Panel
Send on every thoracentesis: cell count with differential, protein, LDH, glucose, pH, Gram stain and culture, cytology. Additional studies as indicated: adenosine deaminase (ADA > 40 = TB pleuritis in endemic settings), triglycerides (chylothorax), amylase (pancreatitis, esophageal rupture), hematocrit (hemothorax if pleural Hct > 50% of blood Hct).
21 Empyema Pleural
Infected pleural effusion — the result of uncontrolled parapneumonic effusion progression. Stages: simple parapneumonic (exudative; free-flowing, sterile, pH > 7.2, glucose > 60, LDH < 1000; treat underlying pneumonia), complicated parapneumonic (fibrinopurulent; pH < 7.2, glucose < 60, LDH > 1000, positive Gram stain/culture, or loculated; requires chest tube drainage), empyema (frank pus in pleural space; requires drainage). Chest tube is first-line drainage. If loculated: intrapleural fibrinolytics (tPA 10 mg + DNase 5 mg instilled BID x 3 days via chest tube — the MIST2 trial showed this combination reduces surgical referral and hospital stay). If fibrinolytics fail: VATS (video-assisted thoracoscopic surgery) decortication. Open decortication for chronic organized empyema with trapped lung.
22 Pneumothorax Pleural
Types
| Type | Mechanism | Risk Factors | Management |
|---|---|---|---|
| Primary spontaneous (PSP) | Rupture of apical subpleural bleb in otherwise healthy lung | Tall, thin young males; smoking | Small (< 2 cm at apex, stable): observation + supplemental O2. Large or symptomatic: aspiration or chest tube |
| Secondary spontaneous (SSP) | Underlying lung disease (COPD, CF, LAM, Marfan's) | COPD most common cause | More dangerous — less pulmonary reserve. Chest tube for all > 1 cm or symptomatic. Admit all SSP. |
| Traumatic | Penetrating or blunt chest trauma, iatrogenic (central line, thoracentesis, lung biopsy, mechanical ventilation) | Procedure-related, trauma | Chest tube; observation for small iatrogenic in stable patient |
| Tension | One-way valve effect — air enters but cannot exit the pleural space; progressive ipsilateral lung collapse, mediastinal shift to contralateral side, compressed contralateral lung and IVC/SVC → cardiovascular collapse | Trauma, mechanical ventilation | Clinical diagnosis — do NOT wait for CXR. Immediate needle decompression (14-16G, 2nd ICS midclavicular line or 5th ICS anterior axillary line) → chest tube |
Recurrence prevention: after first PSP, ~30% recurrence. After second episode, recurrence exceeds 50% — pleurodesis (chemical: talc, doxycycline; or mechanical via VATS with apical bleb resection and pleural abrasion) is recommended after second ipsilateral PSP or first SSP.
23 Mesothelioma Pleural
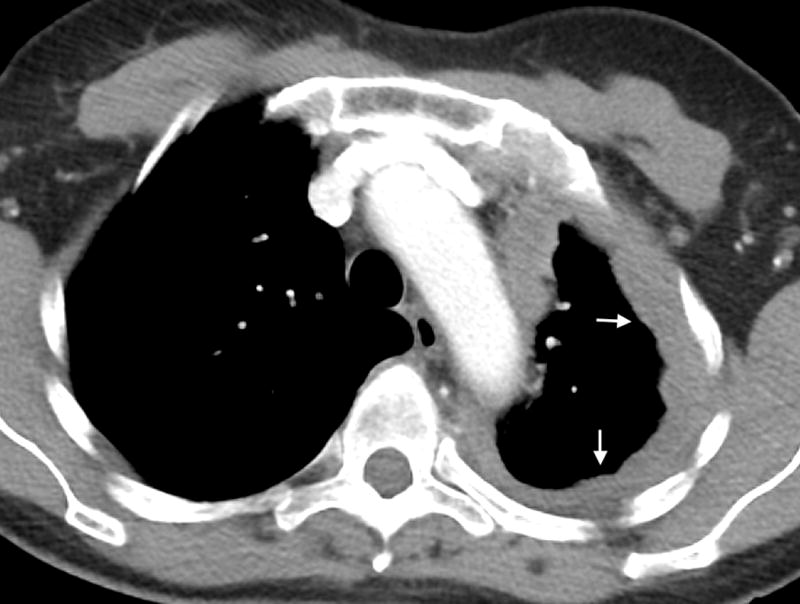
Malignant pleural mesothelioma is an aggressive malignancy of the pleural mesothelium with a strong association to asbestos exposure (latency 20-50 years). Median survival: 12-18 months. Histologic types: epithelioid (best prognosis), sarcomatoid (worst), biphasic (mixed). Presents with dyspnea, non-pleuritic chest pain, unilateral pleural effusion with nodular pleural thickening on CT. Diagnosis requires tissue biopsy (thoracoscopic preferred) — cytology alone is insufficient in most cases. Immunohistochemistry: calretinin+, CK5/6+, WT-1+ (mesothelioma markers) vs. TTF-1+, Napsin-A+ (adenocarcinoma). Treatment: multimodal for early stage (chemotherapy + surgery ± radiation) — pemetrexed/cisplatin is first-line chemotherapy. Nivolumab + ipilimumab (CheckMate 743) improves survival in unresectable disease. Palliative care and PleurX catheter for symptomatic effusion management.
24 Pulmonary Embolism (PE) Vascular
Obstruction of the pulmonary arterial vasculature by thrombus (usually from lower extremity DVT), resulting in V/Q mismatch, increased dead space, and in massive PE, right ventricular failure. PE is the third most common cause of cardiovascular death after MI and stroke. Mortality ranges from < 1% (low-risk) to > 50% (massive with cardiac arrest).
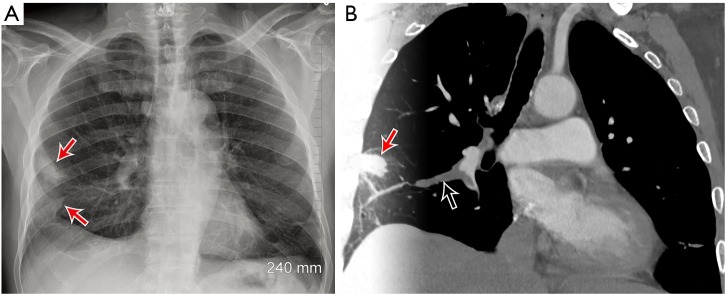
Wells Score for PE
| Criterion | Points |
|---|---|
| Clinical signs/symptoms of DVT | 3.0 |
| PE is #1 diagnosis OR equally likely | 3.0 |
| Heart rate > 100 | 1.5 |
| Immobilization (≥ 3 days) or surgery in past 4 weeks | 1.5 |
| Previous DVT/PE | 1.5 |
| Hemoptysis | 1.0 |
| Active cancer | 1.0 |
Score ≤ 4 (PE unlikely): D-dimer → if negative, PE excluded. Score > 4 (PE likely): proceed directly to CTPA. Age-adjusted D-dimer threshold (age x 10 ng/mL for patients > 50 years) improves specificity without compromising sensitivity.
Revised Geneva Score
| Criterion | Points |
|---|---|
| Age > 65 | 1 |
| Previous DVT or PE | 3 |
| Surgery or fracture within 1 month | 2 |
| Active malignancy | 2 |
| Unilateral lower limb pain | 3 |
| Hemoptysis | 2 |
| Heart rate 75-94 | 3 |
| Heart rate ≥ 95 | 5 |
| Pain on lower limb DVP and unilateral edema | 4 |
Score 0-3: low probability. 4-10: intermediate. ≥ 11: high.
Risk Stratification & Treatment
| Category | Hemodynamics | RV Dysfunction | Troponin | Treatment |
|---|---|---|---|---|
| Massive (high-risk) | SBP < 90 or requiring pressors | Yes | Yes | Systemic thrombolysis (alteplase 100 mg IV over 2hr) or catheter-directed therapy or surgical embolectomy; heparin |
| Submassive (intermediate-high) | Stable | Yes (CT/echo) | Yes | Anticoagulation; consider catheter-directed therapy if clinical deterioration |
| Submassive (intermediate-low) | Stable | Yes OR elevated troponin (not both) | — | Anticoagulation; monitor closely |
| Low-risk | Stable | No | No | Anticoagulation; consider early discharge or outpatient treatment if sPESI = 0 |
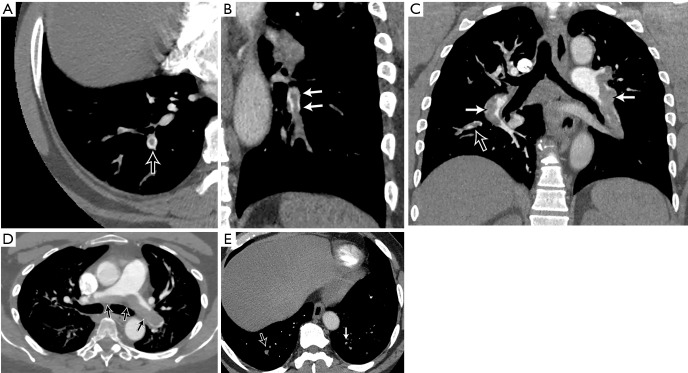
Anticoagulation: DOAC preferred for most patients (rivaroxaban 15 mg BID x 21 days then 20 mg daily, or apixaban 10 mg BID x 7 days then 5 mg BID). For cancer-associated PE: LMWH or DOAC (edoxaban, rivaroxaban — avoid in GI/GU malignancy due to bleeding risk). Duration: provoked PE = 3 months; unprovoked PE = consider indefinite if bleeding risk acceptable; cancer = while cancer is active.
25 Pulmonary Hypertension Vascular
Defined hemodynamically as mean pulmonary artery pressure (mPAP) > 20 mmHg at rest on right heart catheterization (updated from > 25 at the 6th World Symposium on PH, 2018). Pulmonary vascular resistance (PVR) > 2 Wood units further confirms pre-capillary PH. Symptoms are nonspecific: progressive dyspnea, fatigue, exertional presyncope/syncope, peripheral edema, chest pain. Physical exam: loud P2, right ventricular heave, tricuspid regurgitation murmur, JVD, hepatomegaly, peripheral edema.
WHO Classification of Pulmonary Hypertension
| Group | Category | Examples | Hemodynamics | Treatment Approach |
|---|---|---|---|---|
| 1 | Pulmonary Arterial Hypertension (PAH) | Idiopathic, heritable (BMPR2), drug-induced, CTD-associated (scleroderma), HIV, portal HTN, CHD | Pre-capillary: mPAP > 20, PAWP ≤ 15, PVR > 2 WU | PAH-specific therapies: PDE5i, ERA, prostacyclins, sGC stimulator |
| 2 | PH due to left heart disease | HFrEF, HFpEF, valvular disease | Post-capillary: PAWP > 15 | Treat underlying cardiac disease; PAH drugs generally NOT indicated |
| 3 | PH due to lung disease / hypoxia | COPD, ILD, sleep-disordered breathing, chronic high altitude | Pre-capillary | Treat underlying lung disease; supplemental O2; inhaled treprostinil for PH-ILD |
| 4 | Chronic thromboembolic PH (CTEPH) | Organized thrombus in pulmonary arteries after PE | Pre-capillary | Pulmonary thromboendarterectomy (PTE) — potentially curative; balloon pulmonary angioplasty; riociguat if inoperable |
| 5 | PH with unclear/multifactorial mechanisms | Sarcoidosis, myeloproliferative, glycogen storage, chronic hemolytic anemia (SCD), fibrosing mediastinitis | Variable | Treat underlying condition |
PAH-Specific Therapies (Group 1)
| Class | Drug (Brand) | Mechanism | Route | Key Pearls |
|---|---|---|---|---|
| PDE5 inhibitor | Sildenafil (Revatio), Tadalafil (Adcirca) | Inhibits PDE5 → increased cGMP → vasodilation | PO | Contraindicated with nitrates; sildenafil 20mg TID; tadalafil 40mg daily |
| Endothelin receptor antagonist (ERA) | Ambrisentan (Letairis), Bosentan (Tracleer), Macitentan (Opsumit) | Blocks endothelin-1 → vasodilation, anti-proliferative | PO | Hepatotoxicity (bosentan — monthly LFTs); teratogenic (REMS); macitentan reduced morbidity/mortality (SERAPHIN trial) |
| Prostacyclin analogs | Epoprostenol (Flolan), Treprostinil (Remodulin, Tyvaso), Iloprost (Ventavis), Selexipag (Uptravi) | Prostacyclin pathway activation → vasodilation, antiproliferative, antiplatelet | IV (epo), SC/IV/inhaled (trep), inhaled (ilo), PO (selex) | Epoprostenol IV = most potent; requires continuous central line infusion; T1/2 3-5 min — never abruptly discontinue (rebound PH crisis) |
| sGC stimulator | Riociguat (Adempas) | Stimulates soluble guanylate cyclase → increased cGMP | PO | Contraindicated with PDE5i; approved for PAH and CTEPH |
26 Pulmonary Arteriovenous Malformations Vascular
Abnormal direct connections between pulmonary arteries and veins bypassing the capillary bed, creating a right-to-left shunt. 80-90% are associated with hereditary hemorrhagic telangiectasia (HHT/Osler-Weber-Rendu syndrome) — autosomal dominant; mutations in ENG (endoglin) or ACVRL1 (ALK1). Presentation: hypoxemia refractory to supplemental O2 (true shunt physiology), paradoxical embolism (stroke, brain abscess — venous thrombi bypass the pulmonary filter), epistaxis and mucocutaneous telangiectasias (HHT), and hemoptysis. Diagnosis: contrast echocardiography (agitated saline — bubbles appearing in the left atrium after 3-5 cardiac cycles indicates intrapulmonary shunt, vs. < 3 cycles for intracardiac). CT chest confirms anatomy. Treatment: transcatheter embolization for feeding arteries ≥ 3 mm (prevents stroke/abscess). Screen all HHT patients for PAVMs. Antibiotic prophylaxis before dental procedures (prevention of brain abscess due to loss of pulmonary capillary filtration).
27 Non-Small Cell Lung Cancer (NSCLC) Cancer
NSCLC accounts for ~85% of all lung cancers. Three major subtypes: adenocarcinoma (40-50% — most common; peripheral location; associated with non-smokers and women; driver mutations common), squamous cell carcinoma (25-30% — central/endobronchial; strongly linked to smoking; can cavitate; associated with hypercalcemia via PTHrP), and large cell carcinoma (5-10% — undifferentiated; peripheral; poor prognosis). Lung cancer is the leading cause of cancer death in both men and women.
TNM Staging (8th Edition)
| T | Description |
|---|---|
| Tis | Carcinoma in situ |
| T1a/b/c | ≤ 1 cm / ≤ 2 cm / ≤ 3 cm; surrounded by lung or visceral pleura |
| T2a/b | > 3-4 cm / > 4-5 cm, or involves mainstem bronchus (not carina), visceral pleura invasion, or causes atelectasis to hilum |
| T3 | > 5-7 cm; or invades chest wall, pericardium, phrenic nerve; or separate nodule in same lobe |
| T4 | > 7 cm; or invades mediastinum, diaphragm, heart, great vessels, carina, trachea, esophagus, vertebra, RLN; or separate nodule in different ipsilateral lobe |
| N | Description |
|---|---|
| N0 | No regional lymph node metastasis |
| N1 | Ipsilateral peribronchial / hilar nodes |
| N2 | Ipsilateral mediastinal / subcarinal nodes |
| N3 | Contralateral mediastinal/hilar, or any scalene/supraclavicular nodes |
Molecular Markers & Targeted Therapy
| Marker | Frequency in Adenocarcinoma | Targeted Agent |
|---|---|---|
| EGFR mutation (exon 19 del, L858R) | 10-15% (Caucasian), 40-50% (Asian) | Osimertinib (Tagrisso) — 3rd gen TKI; first-line for EGFR-mutant NSCLC |
| ALK rearrangement | 3-7% | Alectinib (Alecensa), lorlatinib (Lorbrena) |
| ROS1 rearrangement | 1-2% | Crizotinib (Xalkori), entrectinib (Rozlytrek) |
| BRAF V600E | 1-3% | Dabrafenib + trametinib |
| KRAS G12C | ~13% | Sotorasib (Lumakras), adagrasib (Krazati) |
| PD-L1 expression (≥ 50%) | 25-30% | Pembrolizumab (Keytruda) monotherapy; combination chemo-IO for PD-L1 < 50% |
Stage-Based Treatment Summary
Stage I-II (localized): Surgical resection (lobectomy preferred; segmentectomy for small tumors or poor candidates); adjuvant osimertinib for EGFR-mutant resected stage IB-IIIA (ADAURA trial). Stage III (locally advanced): Concurrent chemoradiation followed by durvalumab (Imfinzi) consolidation x 12 months (PACIFIC trial). Stage IV (metastatic): Molecular testing guides therapy — driver mutation positive → targeted TKI; driver mutation negative → pembrolizumab ± chemotherapy based on PD-L1 status.
Screening
Low-dose CT chest annually for adults 50-80 years with ≥ 20 pack-year smoking history who currently smoke or quit within the past 15 years (USPSTF 2021, based on the National Lung Screening Trial showing 20% relative reduction in lung cancer mortality with LDCT vs. CXR).
28 Small Cell Lung Cancer (SCLC) Cancer
Aggressive neuroendocrine tumor accounting for ~15% of lung cancers. Almost exclusively in smokers. Characterized by rapid doubling time, early metastasis, and initial dramatic chemosensitivity followed by near-universal relapse. Central location; frequently associated with bulky mediastinal lymphadenopathy and paraneoplastic syndromes: SIADH (hyponatremia), ectopic ACTH (Cushing syndrome), Lambert-Eaton myasthenic syndrome (proximal muscle weakness improving with use), cerebellar degeneration (anti-Hu antibodies).
| Stage | Definition | Treatment | Median Survival |
|---|---|---|---|
| Limited-stage (LS) | Confined to one hemithorax and regional nodes (can be encompassed in one radiation field) | Concurrent chemoradiation: cisplatin/etoposide + thoracic RT; prophylactic cranial irradiation (PCI) if response. Atezolizumab + chemo now being explored. | 15-20 months; 5-yr survival ~20-25% |
| Extensive-stage (ES) | Disease beyond one hemithorax (distant mets, malignant effusion, contralateral lung) | Carboplatin/etoposide + atezolizumab (IMpower133) or durvalumab (CASPIAN trial) x 4 cycles then IO maintenance; PCI if response | 10-12 months; 5-yr survival < 5% |
29 Solitary Pulmonary Nodule Cancer
A round opacity ≤ 3 cm in the lung parenchyma (lesions > 3 cm are masses, assumed malignant until proven otherwise). Most are incidental findings. Assessment of malignancy risk: patient factors (age, smoking history, cancer history, COPD) and nodule factors (size, morphology, location, growth rate).
Fleischner Society Guidelines (2017) — Solid Nodules
| Size | Low Risk (< 5%) | High Risk or Upper Lobe/Spiculated |
|---|---|---|
| < 6 mm | No routine follow-up needed | Optional CT at 12 months |
| 6-8 mm | CT at 6-12 months, then consider CT at 18-24 months | CT at 6-12 months, then CT at 18-24 months |
| > 8 mm | CT at 3 months, PET/CT, and/or tissue sampling | |
Lung-RADS (Lung Imaging Reporting & Data System)
| Category | Finding | Malignancy Risk | Recommendation |
|---|---|---|---|
| 1 | Negative — no nodules or benign | < 1% | Continue annual screening |
| 2 | Benign appearance or behavior (solid < 6mm, new solid < 4mm) | < 1% | Continue annual screening |
| 3 | Probably benign (solid 6-8mm, new solid 4-6mm) | 1-2% | 6-month follow-up |
| 4A | Suspicious (solid 8-15mm, growing < 8mm) | 5-15% | 3-month follow-up; PET/CT may be indicated |
| 4B | Very suspicious (solid ≥ 15mm, new or growing ≥ 8mm) | > 15% | Chest CT with/without contrast, PET/CT, tissue sampling |
30 ARDS Critical Care
Acute respiratory distress syndrome is diffuse, non-cardiogenic pulmonary edema caused by inflammatory damage to the alveolar-capillary membrane. In-hospital mortality remains 35-45%. The most common causes are pneumonia and non-pulmonary sepsis (together accounting for ~60% of cases); other causes include aspiration, pancreatitis, trauma, transfusion (TRALI), and drug overdose.
Berlin Definition (2012)
| Criterion | Requirement |
|---|---|
| Timing | Within 1 week of known clinical insult or new/worsening respiratory symptoms |
| Imaging | Bilateral opacities not fully explained by effusions, atelectasis, or nodules |
| Origin | Respiratory failure not fully explained by cardiac failure or fluid overload (echocardiography to exclude if no risk factor) |
| Oxygenation (severity) | Mild: P/F 200-300 with PEEP/CPAP ≥ 5 Moderate: P/F 100-200 with PEEP ≥ 5 Severe: P/F ≤ 100 with PEEP ≥ 5 |
P/F = PaO2/FiO2 ratio (normal ~500 on room air). PEEP = positive end-expiratory pressure.
ARDSNet Lung-Protective Ventilation
The landmark ARDSNet trial (2000) demonstrated a 22% relative reduction in mortality with low tidal volume ventilation. Protocol:
Tidal volume: 6 mL/kg ideal body weight (IBW), NOT actual weight. IBW: males = 50 + 2.3(height in inches - 60); females = 45.5 + 2.3(height in inches - 60).
Plateau pressure: Target ≤ 30 cmH2O. If > 30, reduce Vt to as low as 4 mL/kg IBW.
PEEP: Use ARDSNet PEEP/FiO2 tables (low or high PEEP strategy). Titrate to maintain PaO2 55-80 mmHg or SpO2 88-95%.
Respiratory rate: Up to 35/min to maintain adequate minute ventilation; permissive hypercapnia acceptable (pH target ≥ 7.20).
Driving pressure: Plateau - PEEP; target ≤ 15 cmH2O. Driving pressure may be a better predictor of mortality than plateau pressure alone.
Prone Positioning
The PROSEVA trial (2013) showed that early prone positioning (≥ 16 hours/day) in moderate-severe ARDS (P/F < 150) reduces 28-day mortality from 32.8% to 16.0%. Mechanism: improves V/Q matching by recruiting dorsal lung regions compressed by the heart and abdominal contents in supine position, and creating more uniform pleural pressure distribution. Contraindications: hemodynamic instability requiring immediate intervention, spinal instability, open abdomen, raised ICP.
Other ARDS Interventions
Neuromuscular blockade: Cisatracurium for 48 hours in early severe ARDS — the ACURASYS trial showed mortality benefit, though ROSE (2019) did not confirm in a lighter sedation strategy. Reserved for severe refractory hypoxemia, ventilator dyssynchrony, and prone positioning. Conservative fluid management: FACTT trial showed improved oxygenation and ventilator-free days with conservative vs. liberal fluid strategy. Corticosteroids: Dexamethasone 20mg x 5 days then 10mg x 5 days in moderate-severe ARDS may reduce ventilator days and mortality (DEXA-ARDS trial). Inhaled pulmonary vasodilators: Inhaled nitric oxide (iNO) or inhaled epoprostenol for refractory hypoxemia — improve oxygenation transiently but no survival benefit; rescue bridge. ECMO: Venovenous ECMO for severe refractory ARDS (P/F < 80 for > 6 hours or pH < 7.25 with PaCO2 > 60 for > 6 hours despite optimized ventilation) — EOLIA trial showed trend toward survival benefit.
31 Mechanical Ventilation Critical Care
Modes of Ventilation
| Mode | How It Works | Patient Trigger | When Used |
|---|---|---|---|
| Volume Control (VC-CMV / AC) | Delivers a set tidal volume at a set rate. Pressure varies with compliance/resistance. | Patient can trigger additional breaths at set Vt | Most common initial mode; reliable minute ventilation |
| Pressure Control (PC-CMV) | Delivers a set inspiratory pressure for a set time. Vt varies with compliance/resistance. | Patient can trigger additional breaths at set pressure | ARDS (better inspiratory flow pattern), high airway pressures in VC |
| SIMV (+ PS) | Set number of mandatory breaths (volume or pressure) + patient can breathe spontaneously with pressure support between mandatory breaths | Spontaneous breaths above set rate | Weaning (now less favored); transitional mode |
| Pressure Support (PSV) | Patient triggers every breath; ventilator augments with set pressure support. No set rate — entirely patient-driven. | All breaths are patient-triggered | Spontaneous breathing trials (SBT), weaning, awake/cooperative patients |
| APRV (Airway Pressure Release) | High CPAP level (P-high) with intermittent brief releases to lower pressure (P-low) for CO2 clearance. Spontaneous breathing maintained throughout. | Spontaneous breaths at P-high | ARDS (promotes recruitment); specialized — requires experienced team |
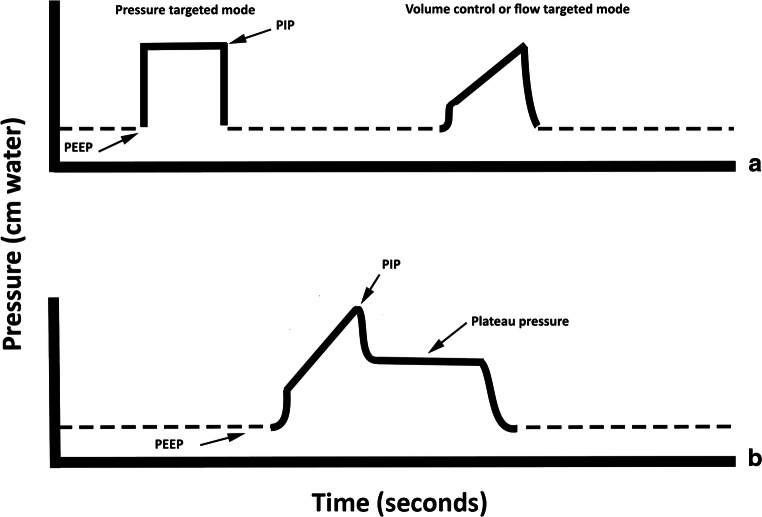
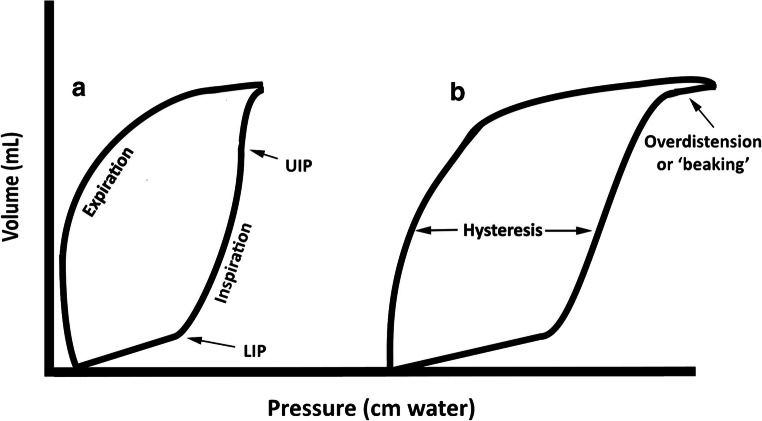
Key Ventilator Settings & Alarms
FiO2: Start at 100%, wean to ≤ 60% as quickly as possible (O2 toxicity). PEEP: Prevents alveolar collapse at end-expiration; improves oxygenation; typically 5-20 cmH2O. I:E ratio: normally 1:2-1:3; inverse ratio ventilation (I:E > 1:1) used in severe ARDS but requires sedation/paralysis. Peak airway pressure: reflects both compliance and resistance; elevated in bronchospasm, secretions, tube kinking, AND stiff lungs. Plateau pressure: measured during inspiratory hold (no flow); reflects compliance alone — if peak is high but plateau is normal, the problem is airway resistance (bronchospasm, mucus plug). If both are high, the problem is compliance (ARDS, pneumothorax, auto-PEEP, abdominal distension).
Occurs when expiration is incomplete before the next breath — air traps in the lungs, creating positive end-expiratory pressure above the set PEEP. Common in COPD, asthma, and with high respiratory rates or short expiratory times. Consequences: increased intrathoracic pressure → decreased venous return → hypotension; increased work of breathing; inaccurate plateau pressure readings. Detection: expiratory hold maneuver shows total PEEP greater than set PEEP. Management: reduce respiratory rate, increase expiratory time, treat underlying cause (bronchodilators), consider external PEEP at ~80% of measured auto-PEEP (reduces trigger work).
Weaning & Liberation
Assess daily for readiness: FiO2 ≤ 40%, PEEP ≤ 8, adequate mental status, no vasopressors or minimal dose, spontaneous respiratory effort. Spontaneous breathing trial (SBT): T-piece trial or low-level pressure support (5-8 cmH2O) for 30-120 minutes. Failure criteria: RR > 35, SpO2 < 90%, HR change > 20%, new arrhythmia, SBP < 90 or > 180, agitation/diaphoresis. Rapid shallow breathing index (RSBI) = RR / Vt (in liters); RSBI < 105 predicts successful extubation. The daily SBT protocol with sedation interruption reduces time on the ventilator.
Post-extubation: Prophylactic NIV (BiPAP) for high-risk patients (age > 65, CHF, COPD, hypercapnic during SBT) reduces reintubation rates. High-flow nasal cannula (HFNC) at 40-60 L/min is an alternative for post-extubation support.
32 Shock Critical Care
Circulatory failure resulting in inadequate tissue oxygen delivery. The four hemodynamic profiles:
| Type | CO | SVR | PCWP/CVP | Common Causes | Key Treatment |
|---|---|---|---|---|---|
| Distributive (septic, anaphylactic, neurogenic) | ↑ | ↓↓ | ↓ or normal | Sepsis (most common), anaphylaxis, spinal cord injury | Fluids + norepinephrine (first-line vasopressor for septic shock) |
| Cardiogenic | ↓↓ | ↑ | ↑↑ | MI (LV failure), acute valvular, myocarditis, arrhythmia | Dobutamine or milrinone; revascularization for MI; mechanical support (IABP, Impella) |
| Hypovolemic | ↓ | ↑ | ↓↓ | Hemorrhage, dehydration, burns | Volume resuscitation; blood products for hemorrhagic; source control |
| Obstructive | ↓ | ↑ | ↑ (R-sided) | Tension PTX, massive PE, cardiac tamponade | Treat underlying cause: needle decompression, thrombolysis, pericardiocentesis |
Vasopressors & Inotropes
| Drug | Primary Receptors | Main Effect | Typical Dose Range | Pearl |
|---|---|---|---|---|
| Norepinephrine (Levophed) | α1 >> β1 | Vasoconstriction + mild inotropy | 0.01-3 mcg/kg/min | First-line for septic shock; preferred over dopamine (SOAP II trial) |
| Vasopressin (Pitressin) | V1 receptors | Vasoconstriction (non-adrenergic) | 0.03-0.04 units/min (fixed dose) | Adjunct to NE in septic shock; catecholamine-sparing; not titrated |
| Epinephrine (Adrenalin) | β1, β2, α1 | Inotropy + vasoconstriction (dose-dependent) | 0.01-0.5 mcg/kg/min | Second-line for septic shock; first-line for anaphylaxis; raises lactate (aerobic glycolysis) |
| Phenylephrine (Neo-Synephrine) | Pure α1 | Vasoconstriction only | 0.5-5 mcg/kg/min | Reflex bradycardia; avoid in cardiogenic shock (increases afterload) |
| Dobutamine | β1 > β2 | Inotropy + mild vasodilation | 2-20 mcg/kg/min | Cardiogenic shock; can cause hypotension (vasodilation); tachyarrhythmias |
| Milrinone (Primacor) | PDE3 inhibitor | Inotropy + vasodilation ("inodilator") | 0.125-0.75 mcg/kg/min | RV failure, pulmonary HTN; longer half-life (2-3hr) — harder to titrate; dose adjust in renal failure |
| Dopamine | D1, β1, α1 (dose-dependent) | Low dose: renal vasodilation; Mod: inotropy; High: vasoconstriction | 2-20 mcg/kg/min | Largely replaced by NE for septic shock; still used for symptomatic bradycardia |
| Angiotensin II (Giapreza) | AT1 receptor | Vasoconstriction (RAAS pathway) | Starting 20 ng/kg/min, titrate | Refractory vasodilatory shock; ATHOS-3 trial |
33 Sepsis & Septic Shock Critical Care
Sepsis (Sepsis-3 definition): life-threatening organ dysfunction caused by a dysregulated host response to infection. Operationally: suspected/confirmed infection + acute change in SOFA score ≥ 2. Septic shock: sepsis with persistent hypotension requiring vasopressors to maintain MAP ≥ 65 AND serum lactate > 2 mmol/L despite adequate fluid resuscitation. Septic shock mortality: 40-50%.
The 1-Hour Bundle (Surviving Sepsis Campaign 2021)
1. Measure lactate — if > 2 mmol/L, remeasure within 2-4 hours to guide resuscitation.
2. Obtain blood cultures (2 sets from different sites) BEFORE antibiotics — but do NOT delay antibiotics for cultures.
3. Administer broad-spectrum antibiotics — each hour of delay increases mortality by ~4% in septic shock.
4. Begin rapid IV fluid resuscitation — 30 mL/kg crystalloid (balanced solutions preferred: LR or PlasmaLyte) for hypotension or lactate ≥ 4. Reassess volume status frequently (passive leg raise, stroke volume variation, IVC distensibility).
5. Start vasopressors if hypotensive during or after fluid resuscitation — norepinephrine first-line. Target MAP ≥ 65 mmHg.
Corticosteroids in septic shock: Hydrocortisone 200 mg/day (50 mg IV q6h) for patients with septic shock refractory to fluids and vasopressors. The ADRENAL and APROCCHSS trials showed conflicting mortality results, but hydrocortisone consistently accelerates shock reversal. Reasonable to start if the patient requires high-dose or escalating vasopressors.
Source control: Identify and drain/debride the source of infection as rapidly as possible — abscess drainage, infected device removal, necrotizing fasciitis debridement, perforated viscus repair. Delay in source control is an independent predictor of mortality.
34 Sedation, Analgesia & Delirium Critical Care
The PADIS Guidelines (Pain, Agitation/Sedation, Delirium, Immobility, Sleep)
The mantra: analgesia first, then sedation. Pain is the primary driver of agitation in the ICU; treating pain reduces sedative requirements. Target the lightest level of sedation that keeps the patient safe and comfortable.
Richmond Agitation-Sedation Scale (RASS)
| Score | Term | Description |
|---|---|---|
| +4 | Combative | Overtly combative, violent, immediate danger to staff |
| +3 | Very agitated | Pulls or removes tubes/catheters; aggressive |
| +2 | Agitated | Frequent non-purposeful movement, fights ventilator |
| +1 | Restless | Anxious, apprehensive but movements not aggressive |
| 0 | Alert & calm | Spontaneously pays attention to caregiver |
| -1 | Drowsy | Not fully alert; sustained awakening (eye opening/contact) to voice (> 10 sec) |
| -2 | Light sedation | Briefly awakens to voice (eye opening/contact < 10 sec) |
| -3 | Moderate sedation | Movement or eye opening to voice, no eye contact |
| -4 | Deep sedation | No response to voice; movement or eye opening to physical stimulation |
| -5 | Unarousable | No response to voice or physical stimulation |
Target RASS 0 to -2 for most ICU patients. Deep sedation (RASS -4/-5) is associated with longer ventilator time, increased delirium, and higher mortality.
CAM-ICU (Confusion Assessment Method for ICU)
The standard bedside tool for detecting ICU delirium. Four features: (1) Acute onset or fluctuating mental status, (2) Inattention (squeeze hand on letter A test), (3) Altered level of consciousness (RASS != 0), (4) Disorganized thinking (simple yes/no questions). Delirium = Features 1 + 2 + either 3 or 4. ICU delirium affects 60-80% of mechanically ventilated patients and is associated with prolonged hospitalization, cognitive impairment, and mortality.
Sedation & Analgesia Agents
| Drug | Class | Onset/Duration | Key Properties | Pearl |
|---|---|---|---|---|
| Propofol (Diprivan) | GABA agonist | Rapid on/off (seconds) | Sedation, amnesia, antiemetic; no analgesic properties; dose-dependent hypotension | Monitor triglycerides, CK for propofol infusion syndrome (> 48hr at high doses: metabolic acidosis, rhabdomyolysis, cardiac failure, hyperkalemia) |
| Dexmedetomidine (Precedex) | α2-agonist | Moderate onset; titrate over 15-30 min | Sedation WITHOUT respiratory depression; patient remains arousable; anxiolysis; mild analgesia | Bradycardia and hypotension; ideal for light sedation during weaning and extubation; does not suppress respiratory drive |
| Midazolam (Versed) | Benzodiazepine | 2-5 min onset; accumulates with prolonged use | Sedation, amnesia, anxiolysis, anticonvulsant | Prolonged sedation after extended infusion (lipophilic accumulation); associated with increased delirium — avoid as first-line |
| Fentanyl | Opioid (synthetic) | 1-2 min IV; short-acting bolus | Potent analgesia; 100x morphine potency; no histamine release | Accumulates with prolonged infusion (lipophilic); chest wall rigidity at high bolus doses |
| Hydromorphone (Dilaudid) | Opioid | 5-15 min IV | Potent analgesia; less histamine release than morphine | Preferred over morphine in renal failure (less active metabolite accumulation) |
| Ketamine | NMDA antagonist | 1-2 min IV | Dissociative sedation + analgesia; bronchodilation; preserves respiratory drive and hemodynamics | Emergence reactions (give benzodiazepine); raises ICP (relative contraindication); excellent for intubation in bronchospasm/hypotension |
35 Obstructive Sleep Apnea Sleep
Recurrent partial (hypopnea) or complete (apnea) upper airway collapse during sleep, causing intermittent hypoxemia, sleep fragmentation, and sympathetic activation. Prevalence: 10-20% of adults; severely underdiagnosed. Risk factors: obesity (BMI > 30 — strongest risk factor), male sex, age > 50, neck circumference > 17 inches (men) / > 16 inches (women), retrognathia/micrognathia, tonsillar hypertrophy, nasal obstruction. The STOP-BANG questionnaire (Snoring, Tired, Observed apnea, Pressure [HTN], BMI > 35, Age > 50, Neck > 40 cm, Gender male) screens for OSA risk; score ≥ 5 = high probability.
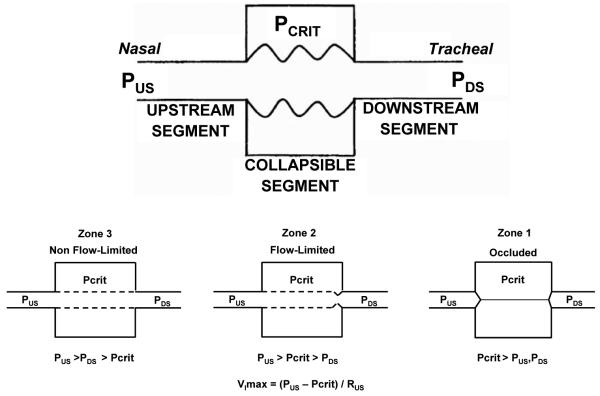
AHI Classification
| AHI (events/hour) | Severity | Treatment |
|---|---|---|
| < 5 | Normal | No treatment required |
| 5-14 | Mild | CPAP if symptomatic; weight loss; positional therapy; mandibular advancement device (MAD) |
| 15-29 | Moderate | CPAP recommended; alternatives: MAD, hypoglossal nerve stimulation (Inspire) |
| ≥ 30 | Severe | CPAP strongly recommended; surgical options if CPAP intolerant |
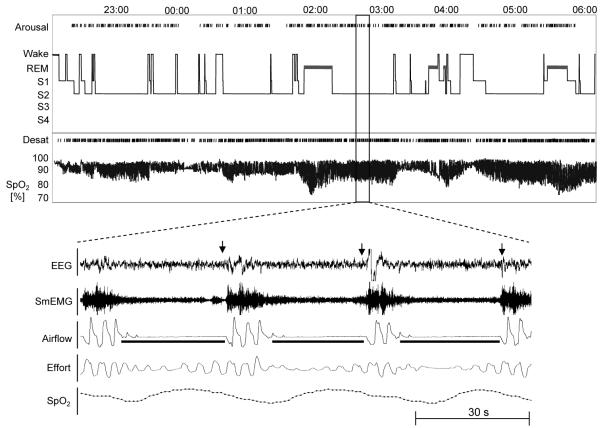
Diagnosis: polysomnography (PSG) is the gold standard — measures airflow, respiratory effort, SpO2, EEG (sleep staging), EMG, and ECG. Home sleep apnea testing (HSAT) is acceptable for uncomplicated patients with high pretest probability (measures airflow, effort, SpO2 — no EEG; underestimates AHI; negative HSAT does not rule out OSA).
Consequences of untreated OSA: hypertension (resistant HTN — always screen), atrial fibrillation, heart failure, stroke, MI, type 2 diabetes, motor vehicle accidents (2-3x risk), cognitive impairment, pulmonary hypertension. CPAP compliance (≥ 4 hours/night on ≥ 70% of nights) is the major challenge — 30-50% of patients are nonadherent.
36 Central Sleep Apnea Sleep
Cessation of airflow due to absent respiratory effort (no signal from the brainstem respiratory center), unlike OSA where effort continues against a collapsed airway. Most commonly associated with heart failure (Cheyne-Stokes respiration — crescendo-decrescendo breathing pattern with central apneas). Other causes: opioid use (dose-dependent respiratory depression), brainstem lesions (stroke, tumor), high-altitude periodic breathing, idiopathic, and treatment-emergent CSA (complex sleep apnea — CSA appearing or persisting when OSA is treated with CPAP).
Treatment: address underlying cause (optimize heart failure therapy — GDMT, CRT; taper opioids). Device therapy: adaptive servo-ventilation (ASV) for most CSA — EXCEPT in heart failure with EF ≤ 45% where ASV is contraindicated (SERVE-HF trial showed increased cardiovascular mortality). For HFrEF-related CSA: CPAP or supplemental O2; phrenic nerve stimulation (Remede System) for moderate-severe CSA.
37 Obesity Hypoventilation Syndrome Sleep
The triad of: (1) obesity (BMI ≥ 30, typically ≥ 40), (2) chronic daytime hypercapnia (PaCO2 > 45 mmHg while awake), and (3) exclusion of other causes of hypoventilation (neuromuscular, chest wall, COPD, hypothyroidism). Coexists with OSA in ~90% of cases. Pathophysiology: combination of reduced respiratory drive (leptin resistance), increased work of breathing (chest wall mass loading), and repetitive upper airway obstruction. Untreated OHS has significantly worse outcomes than OSA alone: higher rates of pulmonary hypertension, cor pulmonale, and mortality. Treatment: PAP therapy (CPAP if coexisting OSA is the dominant component; BiPAP with backup rate if CPAP fails to correct hypercapnia or if OSA is not predominant), weight loss (bariatric surgery produces best long-term results), and supplemental O2 if persistent hypoxemia despite PAP.
38 Neuromuscular Respiratory Failure Sleep
Respiratory failure from weakness of the respiratory muscles (diaphragm, intercostals, accessory muscles, bulbar muscles) in neuromuscular disease. Conditions: amyotrophic lateral sclerosis (ALS), myasthenia gravis, Guillain-Barre syndrome (GBS), muscular dystrophies (Duchenne, myotonic dystrophy), critical illness myopathy/polyneuropathy.
Key monitoring: forced vital capacity (FVC) — most useful bedside measure. In GBS/myasthenic crisis: the "20/30/40 rule" for intubation — FVC < 20 mL/kg, NIF (negative inspiratory force) weaker than -30 cmH2O, or > 30% decline in FVC from baseline. In ALS: NIV (BiPAP) when FVC < 50% predicted or symptomatic respiratory insufficiency — improves survival and quality of life. FVC < 25% or daytime PaCO2 > 50 mmHg = late-stage; discuss goals of care (invasive ventilation via tracheostomy vs. palliative approach). Cough assist devices (mechanical insufflation-exsufflation — CoughAssist) for patients with impaired cough (< 270 L/min peak cough flow) to prevent mucus plugging and pneumonia.
39 Pulmonary Eosinophilia Special
A group of disorders characterized by eosinophilic infiltration of the lung parenchyma ± peripheral blood eosinophilia. Acute eosinophilic pneumonia (AEP): acute febrile illness (days) with bilateral infiltrates, hypoxemic respiratory failure, BAL eosinophils > 25%, peripheral eosinophilia may be absent early. Classic trigger: new smoking onset or dust exposure in military personnel. Responds dramatically to corticosteroids. Chronic eosinophilic pneumonia (CEP): subacute (weeks to months), peripheral and migratory infiltrates classically in the "photographic negative of pulmonary edema" pattern (peripheral distribution sparing the perihilar region), peripheral eosinophilia > 6%, responds to prednisone but relapses are common upon taper. Eosinophilic granulomatosis with polyangiitis (EGPA, Churg-Strauss): asthma + eosinophilia + small-vessel vasculitis; p-ANCA positive in ~40%; cardiac involvement (eosinophilic myocarditis) is the leading cause of death.
40 Pulmonary Alveolar Proteinosis Special
Accumulation of surfactant-like lipoproteinaceous material in alveoli due to impaired macrophage clearance. Most common form (90%) is autoimmune PAP caused by antibodies against granulocyte-macrophage colony-stimulating factor (GM-CSF) — GM-CSF is essential for alveolar macrophage maturation. Classic CT finding: "crazy-paving" pattern (ground-glass opacities with superimposed interlobular septal thickening). BAL shows milky, PAS-positive fluid. Treatment: whole-lung lavage (WLL) — sequential single-lung ventilation while lavaging the contralateral lung with 10-15 liters of warm saline until the return is clear. Often needs to be repeated. Inhaled or subcutaneous GM-CSF therapy is an alternative or adjunct.
41 Lymphangioleiomyomatosis (LAM) Special
A rare, progressive cystic lung disease occurring almost exclusively in women of childbearing age. Characterized by proliferation of abnormal smooth muscle-like cells (LAM cells) that infiltrate airways, lymphatics, and blood vessels. Associated with tuberous sclerosis complex (TSC-LAM) or sporadic (S-LAM). Mutations in TSC1 or TSC2 genes → mTOR pathway activation. Presents with dyspnea, recurrent pneumothoraces (in ~50%), chylous pleural effusions, and renal angiomyolipomas. CT shows thin-walled, round cysts diffusely distributed throughout both lungs (unlike emphysema which spares the bases, or Langerhans cell histiocytosis which spares the costophrenic angles). Serum VEGF-D ≥ 800 pg/mL is diagnostic in the right clinical context. Treatment: sirolimus (rapamycin) — mTOR inhibitor; stabilizes lung function, reduces chylous effusions, and shrinks angiomyolipomas (MILES trial). Lung transplantation for end-stage disease.
42 Cystic Fibrosis — Adult Special
Autosomal recessive; CFTR gene (chromosome 7) mutations cause dysfunctional chloride/bicarbonate channels → thick, dehydrated secretions in lungs, pancreas, GI tract, reproductive system, and sweat glands. The most common mutation is F508del (~70% of alleles). Median survival has improved to > 50 years with modern care. Adult pulmonary manifestations: progressive bronchiectasis, chronic airway infection (Pseudomonas aeruginosa is the dominant organism by adulthood; also Staphylococcus aureus, Burkholderia cepacia complex — poor prognostic marker), hemoptysis, and respiratory failure.
CFTR modulator therapy has transformed CF care: Elexacaftor/tezacaftor/ivacaftor (Trikafta) — a triple combination corrector/potentiator effective in patients with at least one F508del allele (~90% of CF patients). Improves FEV1 by ~14%, reduces exacerbations by 63%, and improves sweat chloride, BMI, and quality of life. Other modulators: ivacaftor (Kalydeco — for gating mutations like G551D), lumacaftor/ivacaftor (Orkambi), tezacaftor/ivacaftor (Symdeko).
Chronic management: aggressive airway clearance (chest PT, oscillatory PEP, HFCWO vest), inhaled mucolytics (dornase alfa/Pulmozyme — DNase that cleaves neutrophil DNA in sputum; hypertonic saline 7% nebulized BID), chronic antibiotics (inhaled tobramycin alternating months, azithromycin 3x/week), pancreatic enzyme replacement (lipase with meals), CFRD (CF-related diabetes) screening and insulin therapy, nutritional optimization (high-calorie, high-fat diet; fat-soluble vitamin supplementation A, D, E, K), and lung transplantation for end-stage disease.
43 Bronchoscopy Procedures
Flexible Bronchoscopy
The workhorse diagnostic and therapeutic procedure of pulmonology. Performed via the nose or mouth (or via ETT in intubated patients) under moderate sedation or general anesthesia. The bronchoscopist sequentially examines the vocal cords, trachea, carina, and each lobar and segmental bronchus. Diagnostic capabilities: visual inspection, bronchoalveolar lavage (BAL — 100-300 mL sterile saline instilled and aspirated from a subsegmental bronchus; analysis for cell count/differential, cultures, cytology), brushings, endobronchial biopsy (forceps biopsy of visible lesions), transbronchial biopsy (TBBx — forceps advanced through bronchial wall into lung parenchyma; used for diffuse lung disease, transplant rejection surveillance), and transbronchial needle aspiration (TBNA — sampling mediastinal lymph nodes). Therapeutic uses: foreign body removal, hemoptysis management (iced saline lavage, epinephrine instillation, balloon tamponade), whole-lung lavage (PAP), stent placement for malignant airway obstruction, tumor debulking (laser, electrocautery, cryotherapy, argon plasma coagulation), and endobronchial valve placement.
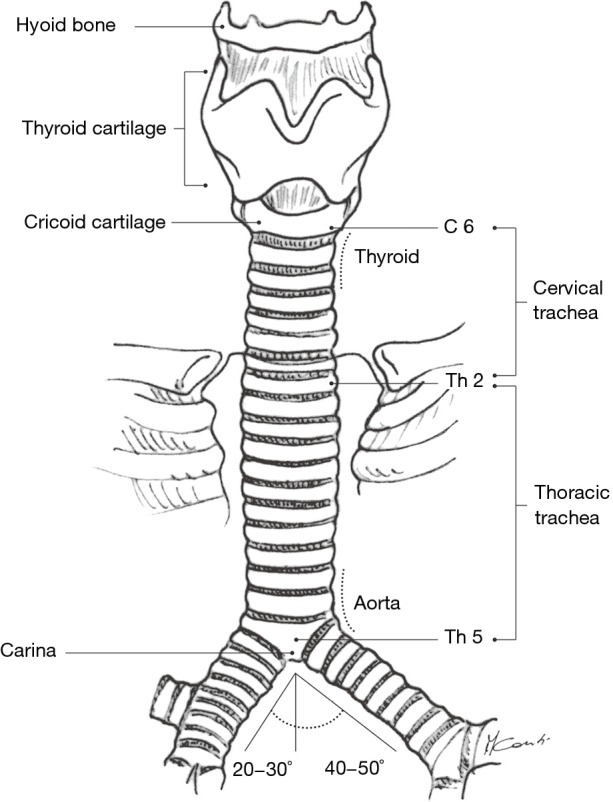
EBUS (Endobronchial Ultrasound)
A bronchoscope with a convex ultrasound probe at the tip allows real-time visualization of mediastinal and hilar structures adjacent to the airway. EBUS-TBNA (transbronchial needle aspiration under ultrasound guidance) is the first-line procedure for mediastinal lymph node sampling in lung cancer staging and sarcoidosis diagnosis. Accessible stations: 2R, 2L, 4R, 4L, 7 (subcarinal), 10R, 10L, 11R, 11L. Combined with EUS-FNA (esophageal ultrasound — accesses stations 7, 8, 9), achieves near-complete mediastinal staging. Sensitivity for mediastinal staging in NSCLC: 85-95%.
Navigational Bronchoscopy
Electromagnetic navigation bronchoscopy (ENB) uses a GPS-like system (superDimension, Ion by Intuitive, Monarch by Ethicon) to guide instruments to peripheral lung lesions beyond the reach of standard bronchoscopy. CT images are loaded pre-procedure to create a virtual roadmap. Used for biopsy of peripheral nodules too small or deep for CT-guided transthoracic biopsy. Diagnostic yield: 60-80% (improves with radial EBUS confirmation and robotic platforms). Complications: pneumothorax (~4%), lower than CT-guided TTNA (~15-25%).
44 Thoracentesis Procedures
Percutaneous drainage of pleural fluid for diagnostic and/or therapeutic purposes. Performed under ultrasound guidance (standard of care — reduces pneumothorax rate from ~10% to < 1%). Patient seated upright, leaning forward. Entry site: 1-2 intercostal spaces below the upper border of the effusion, posterior axillary line, needle inserted just above the rib (to avoid the neurovascular bundle running along the inferior rib margin). Diagnostic thoracentesis: 50-100 mL. Therapeutic thoracentesis: drain until symptoms improve or up to 1-1.5 L per session (risk of re-expansion pulmonary edema if > 1.5 L removed rapidly — manifests as cough, dyspnea, chest tightness during drainage; stop if manometry shows pleural pressure dropping below -20 cmH2O).
Contraindications (relative): coagulopathy (INR > 2, platelets < 25,000 — correct first), very small effusion, skin infection at insertion site, uncooperative patient. Complications: pneumothorax (< 1% with US guidance), hemothorax (intercostal vessel laceration), re-expansion pulmonary edema, vasovagal reaction, infection.
45 Chest Tube & PleurX Catheter Procedures
Chest Tube (Tube Thoracostomy)
Indications: pneumothorax (large, symptomatic, tension, or on mechanical ventilation), large or complicated pleural effusion, empyema, hemothorax, post-thoracic surgery. Insertion: 4th-5th intercostal space, anterior to mid-axillary line (the "safe triangle" — bounded by anterior border of latissimus dorsi, lateral border of pectoralis major, and a horizontal line at the nipple level). Blunt dissection through chest wall, finger sweep to confirm pleural space, tube directed apically for pneumothorax, posteriorly and basally for fluid. Connected to underwater seal (Pleur-Evac) with -20 cmH2O suction. Monitor output, air leak, and lung re-expansion on CXR. Removal: when output < 150-200 mL/24hr (for fluid) or no air leak for 24-48hr + lung fully expanded (for pneumothorax).
PleurX (Tunneled Pleural Catheter)
An indwelling tunneled silicone catheter with a one-way valve placed in the pleural space for long-term outpatient drainage of recurrent malignant or non-malignant effusions. The patient or caregiver drains the fluid at home (typically 500-1000 mL every other day). Achieves spontaneous pleurodesis in 40-70% of patients (pleural surfaces adhere as fluid is chronically drained). Avoids repeated thoracenteses and hospitalization. Complications: empyema (~5%), catheter tract metastases, catheter malfunction, cellulitis.
46 Medical Thoracoscopy & Tracheostomy Procedures
Medical Thoracoscopy (Pleuroscopy)
Performed by pulmonologists under moderate sedation and local anesthesia (contrast with surgical VATS which requires general anesthesia and lung isolation). A single port is used to introduce a rigid or semi-rigid pleuroscope into the pleural space. Indications: undiagnosed exudative pleural effusion (diagnostic yield 92-97% for malignancy — far superior to blind pleural biopsy), talc pleurodesis (talc insufflation under direct visualization for recurrent malignant effusion — 80-90% success rate), and pleural biopsy for suspected TB/mesothelioma. Complications: empyema (~2%), persistent air leak, subcutaneous emphysema.
Tracheostomy
Formation of an opening in the anterior trachea (usually between the 2nd-3rd tracheal rings) for insertion of a tracheostomy tube. Indications: prolonged mechanical ventilation (typically considered after 10-14 days if liberation from the ventilator is not expected within 7-10 days), upper airway obstruction (tumor, trauma, bilateral vocal cord paralysis), airway protection (severe bulbar dysfunction), and facilitation of pulmonary toilet. Percutaneous dilatational tracheostomy (PDT) is most commonly performed by intensivists at the bedside using bronchoscopic guidance and Seldinger technique. Open surgical tracheostomy by surgeons/ENT is preferred for difficult anatomy (short neck, morbid obesity, prior neck surgery, neck mass/radiation, pediatric patients).
Tracheostomy complications: early — hemorrhage, posterior tracheal wall injury, pneumothorax, subcutaneous emphysema, tube displacement (high risk in first 7 days before tract matures). Late — tracheal stenosis (cuff pressure should be maintained 20-25 cmH2O to prevent ischemic mucosal damage), tracheo-innominate fistula (rare but catastrophic — erosion into the innominate artery, typically 1-3 weeks post-tracheostomy; presents as sentinel bleed then massive hemorrhage; management: overinflate cuff, finger compression of innominate artery through the stoma, emergency OR), and tracheoesophageal fistula.
47 Imaging & Diagnostics
| Modality | What It Shows | Interpretation Notes |
|---|---|---|
| Chest X-ray (CXR) | Lung parenchyma, pleura, mediastinum, bones, cardiac silhouette | First-line for most pulmonary complaints; AP vs PA affects cardiac size; decubitus for layering effusion |
| CT Chest (HRCT) | High-resolution parenchymal detail, mediastinal structures, pleural disease | Gold standard for ILD (honeycombing, GGO, reticulation); 1-2 mm slices; expiratory images for air trapping |
| CT Pulmonary Angiography (CTPA) | Pulmonary arterial filling defects (PE) | Sensitivity/specificity > 95% for PE; also shows RV enlargement (RV/LV ratio > 1 = RV strain) |
| V/Q Scan | Ventilation and perfusion match/mismatch | Used when CTPA contraindicated (CKD, contrast allergy); normal = rules out PE; high probability = diagnostic; intermediate = inconclusive |
| PET/CT | Metabolic activity (FDG uptake, SUV) | Lung cancer staging; nodule evaluation (> 8mm); false positives: infection, inflammation, granulomatous disease |
| Chest Ultrasound | Pleural effusion (size, complexity, septations), pneumothorax (absent lung sliding), lung consolidation (hepatization, air bronchograms), B-lines (interstitial edema) | Bedside, no radiation; superior to CXR for effusion detection; guides thoracentesis and chest tube placement |
| Pulmonary Function Tests | Spirometry, lung volumes, DLCO | See Section 04; obstructive vs restrictive vs mixed; bronchodilator response; methacholine challenge |
| Polysomnography (PSG) | Sleep architecture, airflow, effort, SpO2, EMG, ECG | Gold standard for sleep-disordered breathing; AHI, RDI, oxygen nadir, sleep staging |
| Right Heart Catheterization | mPAP, PAWP, PVR, CO, SvO2 | Required for definitive PH diagnosis; distinguishes pre- vs post-capillary PH; vasodilator testing for Group 1 PAH |
| Cardiopulmonary Exercise Test (CPET) | VO2max, anaerobic threshold, ventilatory efficiency | Unexplained dyspnea evaluation; preoperative risk assessment for lung resection (VO2max > 20 mL/kg/min = low risk) |
48 Classification Systems — All in One Place
| System | Classifies | Key Features |
|---|---|---|
| GOLD (spirometric) | COPD airflow severity | 1 (≥ 80%), 2 (50-79%), 3 (30-49%), 4 (< 30%) based on FEV1 % predicted |
| GOLD ABE | COPD treatment groups | A (low symptoms, low exacerbations), B (high symptoms, low exacerbations), E (frequent exacerbations) |
| GINA Steps | Asthma treatment intensity | Steps 1-5; ICS-containing therapy at all steps; biologics at Step 5 |
| Berlin Definition | ARDS severity | Mild (P/F 200-300), Moderate (100-200), Severe (≤ 100) with PEEP ≥ 5 |
| CURB-65 | CAP severity | 0-1 outpatient, 2 consider admission, 3-5 inpatient/ICU |
| PSI/PORT | CAP mortality risk | Class I-V; I-II outpatient, III observe, IV-V inpatient |
| Wells Score | PE probability | ≤ 4 unlikely (D-dimer), > 4 likely (CTPA) |
| Geneva Score | PE probability | 0-3 low, 4-10 intermediate, ≥ 11 high |
| Light's Criteria | Exudate vs transudate | Protein ratio > 0.5, LDH ratio > 0.6, or fluid LDH > 2/3 upper normal = exudate |
| Scadding | Sarcoidosis CXR staging | 0-IV; I = BHL alone (best prognosis), IV = fibrosis (worst) |
| TNM 8th ed | Lung cancer staging | T1-4, N0-3, M0-1; stages IA-IVB |
| Fleischner | Pulmonary nodule management | Size- and risk-based follow-up intervals for solid and subsolid nodules |
| Lung-RADS | Lung cancer screening findings | Categories 1-4B with management recommendations |
| AHI | Sleep apnea severity | < 5 normal, 5-14 mild, 15-29 moderate, ≥ 30 severe |
| RASS | ICU sedation depth | -5 (unarousable) to +4 (combative); target 0 to -2 |
| CAM-ICU | ICU delirium | Features 1+2 + (3 or 4) = delirium |
| WHO PH Groups | Pulmonary hypertension etiology | Groups 1-5 with distinct hemodynamics and treatment approaches |
| SOFA | Organ dysfunction (sepsis) | 6 organ systems scored 0-4; ≥ 2 point acute increase + infection = sepsis |
49 Medications Master Table Meds
Inhaled Bronchodilators
| Drug (Brand) | Class | Mechanism | Dose/Device | Pearl |
|---|---|---|---|---|
| Albuterol (ProAir, Ventolin) | SABA | β2 agonist | 2 puffs PRN q4-6h; neb 2.5mg PRN | Tachycardia, tremor, hypokalemia at high doses |
| Ipratropium (Atrovent) | SAMA | Muscarinic antagonist (M1, M3) | 2 puffs QID; neb 0.5mg TID-QID | Often combined with albuterol (DuoNeb, Combivent) |
| Tiotropium (Spiriva) | LAMA | Selective M3 antagonist | 1 cap (18mcg) via HandiHaler daily; 2 puffs Respimat daily | Cornerstone of COPD maintenance; once daily dosing |
| Umeclidinium (Incruse) | LAMA | M3 antagonist | 1 inhalation (62.5mcg) daily via Ellipta | Commonly in combination products (Anoro, Trelegy) |
| Salmeterol (Serevent) | LABA | β2 agonist (long-acting) | 1 inhalation (50mcg) BID | Never as monotherapy in asthma (boxed warning) — always with ICS |
| Formoterol (Foradil, Perforomist) | LABA | β2 agonist (long-acting, fast onset) | 12mcg BID (DPI); 20mcg neb BID | Fast onset allows use as reliever in ICS-formoterol combination (SMART therapy) |
| Vilanterol | LABA | β2 agonist (ultra-long-acting) | 25mcg daily (always in combination) | Once daily; found in Breo, Anoro, Trelegy |
Inhaled Corticosteroids
| Drug (Brand) | Dose Range | Device | Pearl |
|---|---|---|---|
| Fluticasone propionate (Flovent) | Low: 88-264mcg/d; Med: 264-440; High: > 440 | MDI | Most commonly prescribed ICS in the US |
| Fluticasone furoate (Arnuity) | 100-200mcg daily | Ellipta DPI | Once daily; in combination: Breo (+ vilanterol), Trelegy (+ umec/vil) |
| Budesonide (Pulmicort) | Low: 180-540mcg/d; Med: 540-1080; High: > 1080 | Flexhaler DPI; neb suspension | Best studied ICS in pregnancy (Category B equivalent) |
| Mometasone (Asmanex) | 110-440mcg daily | Twisthaler DPI | Once or twice daily |
| Beclomethasone (QVAR) | Low: 80-160mcg/d; Med: 160-320; High: > 320 | MDI (extrafine particle) | HFA formulation delivers smaller particles → better small airway deposition |
Antifibrotics (IPF)
| Drug (Brand) | Mechanism | Dose | Side Effects | Pearl |
|---|---|---|---|---|
| Pirfenidone (Esbriet) | Anti-fibrotic, anti-inflammatory, antioxidant | 801mg TID with food | Photosensitivity (strict sun avoidance), GI (nausea, anorexia), elevated LFTs | Take with food to reduce GI effects; avoid direct sun exposure |
| Nintedanib (Ofev) | Triple tyrosine kinase inhibitor (VEGFR, FGFR, PDGFR) | 150mg BID with food | Diarrhea (often severe), elevated LFTs, bleeding risk | Also approved for progressive fibrosing ILD and SSc-ILD (INBUILD, SENSCIS trials) |
Antibiotics (Pulmonary)
| Drug (Brand) | Class | Spectrum/Use | Key Pearl |
|---|---|---|---|
| Azithromycin (Zithromax) | Macrolide | Atypicals; anti-inflammatory in bronchiectasis/COPD | QTc prolongation; 250mg 3x/week for exacerbation prevention |
| Levofloxacin (Levaquin) | Respiratory FQ | S. pneumoniae, atypicals, gram-negatives | Tendon rupture, QTc prolongation, C. diff; use only when no alternative |
| Piperacillin-tazobactam (Zosyn) | Beta-lactam/BLI | Pseudomonas, anaerobes, broad gram-negative | First-line empiric for HAP/VAP; dose 4.5g q6h (extended infusion improves PK) |
| Meropenem (Merrem) | Carbapenem | Broadest gram-negative; MDR organisms | Reserve for MDR infections; lowers seizure threshold less than imipenem |
| Vancomycin (Vancocin) | Glycopeptide | MRSA | Trough-based or AUC/MIC-guided dosing (target AUC/MIC 400-600); nephrotoxicity |
| Linezolid (Zyvox) | Oxazolidinone | MRSA, VRE | Serotonin syndrome risk with SSRIs; thrombocytopenia > 14 days; no renal adjustment |
| TMP-SMX (Bactrim) | Folate inhibitor | PJP, Nocardia, Stenotrophomonas | PJP treatment dose: 15-20 mg/kg/day TMP component; hyperkalemia, cytopenias |
| Voriconazole (Vfend) | Triazole antifungal | Invasive aspergillosis (first-line) | Trough monitoring (target 1-5.5 mcg/mL); visual disturbances; photosensitivity (skin cancer risk) |
| Isoniazid (INH) | Mycolic acid synthesis inhibitor | TB (active and latent) | Hepatotoxicity (monitor LFTs); peripheral neuropathy (give pyridoxine); CYP inhibitor |
| Rifampin (Rifadin) | RNA polymerase inhibitor | TB, NTM | Potent CYP3A4 inducer — reduces levels of warfarin, OCPs, azoles, many drugs; orange body fluids |
Vasopressors & ICU Medications
| Drug | Class | Primary Use in Pulm/CC | Pearl |
|---|---|---|---|
| Norepinephrine (Levophed) | α1 + β1 agonist | First-line vasopressor for septic shock | Central line preferred; extravasation → tissue necrosis (phentolamine rescue) |
| Vasopressin | V1 agonist | Adjunct to NE in septic shock | Fixed dose 0.03-0.04 U/min; not titrated; catecholamine-sparing |
| Epoprostenol (Flolan) | Prostacyclin analog | Inhaled: rescue for refractory hypoxemia (ARDS); IV: PAH | Inhaled: improves V/Q matching; IV: never stop abruptly (rebound PH crisis) |
| Inhaled nitric oxide (iNO) | Pulmonary vasodilator | Refractory hypoxemia, RV failure | Selective pulmonary vasodilation; methemoglobinemia at high doses; wean slowly |
| Propofol (Diprivan) | GABA agonist | ICU sedation | PRIS risk > 48hr at > 5 mg/kg/hr; monitor triglycerides, CK, pH |
| Dexmedetomidine (Precedex) | α2 agonist | Light sedation, extubation facilitation | No respiratory depression; bradycardia/hypotension; ideal for weaning |
| Fentanyl | Opioid | ICU analgesia | 100x morphine potency; accumulates with prolonged infusion; chest wall rigidity |
| Cisatracurium (Nimbex) | Neuromuscular blocker | ARDS paralysis, intubation | Organ-independent Hofmann degradation; does not release histamine; train-of-four monitoring |
| Alteplase (Activase) | tPA | Massive PE thrombolysis | 100mg IV over 2hr; also 10mg bolus + 90mg over 2hr; major bleeding risk ~10% |
Sedatives — Quick Reference
| Agent | Route | Onset | Key Advantage | Key Risk |
|---|---|---|---|---|
| Propofol | IV infusion | Seconds | Rapid on/off, easily titratable | PRIS, hypotension, no analgesia |
| Dexmedetomidine | IV infusion | 15-30 min | No respiratory depression, arousable | Bradycardia, hypotension, expensive |
| Midazolam | IV bolus/infusion | 2-5 min | Anxiolysis, anticonvulsant | Accumulation, delirium, prolonged sedation |
| Ketamine | IV/IM | 1-2 min (IV) | Hemodynamic stability, bronchodilation, analgesia | Emergence reactions, secretions |
50 ABG Interpretation
Systematic Approach
Step 1: Assess oxygenation (PaO2 — normal 80-100 mmHg on room air). Step 2: Assess pH (normal 7.35-7.45; < 7.35 = acidemia, > 7.45 = alkalemia). Step 3: Identify the primary disorder (respiratory: PaCO2 abnormal; metabolic: HCO3 abnormal). Step 4: Check for compensation. Step 5: Calculate anion gap (AG = Na - Cl - HCO3; normal 12 ± 4). Step 6: If AG elevated, calculate delta-delta (delta AG / delta HCO3).
Compensation Rules
| Primary Disorder | Expected Compensation | Formula |
|---|---|---|
| Metabolic acidosis | Respiratory (hyperventilate → decrease CO2) | Expected PaCO2 = 1.5(HCO3) + 8 ± 2 (Winter's formula) |
| Metabolic alkalosis | Respiratory (hypoventilate → increase CO2) | Expected PaCO2 = 0.7(HCO3) + 21 ± 2 |
| Acute respiratory acidosis | Metabolic (increase HCO3) | HCO3 increases 1 mEq/L per 10 mmHg rise in CO2 |
| Chronic respiratory acidosis | Metabolic (renal HCO3 retention) | HCO3 increases 3.5 mEq/L per 10 mmHg rise in CO2 |
| Acute respiratory alkalosis | Metabolic (decrease HCO3) | HCO3 decreases 2 mEq/L per 10 mmHg fall in CO2 |
| Chronic respiratory alkalosis | Metabolic (renal HCO3 excretion) | HCO3 decreases 5 mEq/L per 10 mmHg fall in CO2 |
Anion Gap Metabolic Acidosis — MUDPILES
Methanol, Uremia, Diabetic ketoacidosis, Propylene glycol, Isoniazid/Iron, Lactic acidosis (most common — shock, sepsis, mesenteric ischemia, seizures, liver failure), Ethylene glycol, Salicylates.
Non-Anion Gap Metabolic Acidosis — HARDUPS
Hyperalimentation, Acetazolamide / Addison's, Renal tubular acidosis, Diarrhea (most common), Ureteral diversion, Pancreatic fistula, Saline (dilutional from large-volume NS resuscitation).
51 Abbreviations Master List
Anatomy & Physiology
Diagnoses
Procedures & Imaging
Ventilator & ICU
Medications & Labs
References & Figure Sources
Figures
- Figure 1 — Tracheobronchial Tree Anatomy. Adapted from: Ge R, et al. Surgical anatomy of the tracheobronchial tree. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 2 — Lobar and Segmental Lung Anatomy. Adapted from: Ge R, et al. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 3 — Pulmonary Vascular Anatomy. Adapted from: Ge R, et al. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 4 — Mediastinal Lymph Node Stations. Adapted from: Ge R, et al. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 5 — Diaphragm and Chest Wall Anatomy. Adapted from: Ge R, et al. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 6 — Normal Chest Radiograph Anatomy. Adapted from: Ge R, et al. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 7 — Lung Volumes and Spirometric Patterns. Adapted from: Blanchard JD, et al. Pathogenesis, imaging and clinical characteristics of CF and non-CF bronchiectasis. BMC Pulm Med. 2018;18:83. PMC5964733. Licensed under CC BY 4.0.
- Figure 8 — CT Imaging in Obstructive Lung Disease. Adapted from: Defined imaging in non-cystic fibrosis bronchiectasis. BJR. 2022;95:20210026. PMC8328081. Licensed under CC BY 4.0.
- Figure 9 — Asthma Airway Remodeling. Adapted from: Hough KP, et al. Airway Remodeling in Asthma. Front Med. 2020;7:191. PMC7253669. Licensed under CC BY 4.0.
- Figure 10 — Bronchiectasis on HRCT. Adapted from: Defined imaging in non-cystic fibrosis bronchiectasis. BJR. 2022;95:20210026. PMC8328081. Licensed under CC BY 4.0.
- Figure 11 — UIP Pattern on HRCT in IPF. Adapted from: Walsh SLF. Imaging: how to recognise idiopathic pulmonary fibrosis. Eur Respir Rev. 2014;23:215. PMC9487568. Licensed under CC BY-NC 4.0.
- Figures 12-13 — Sarcoidosis Imaging. Adapted from: Criado E, et al. Typical and atypical manifestations of intrathoracic sarcoidosis. Korean J Radiol. 2010;10:623. PMC2770817. Licensed under CC BY-NC 3.0.
- Figure 14 — Occupational Lung Disease on CT. Adapted from: Criado E, et al. Korean J Radiol. 2010;10:623. PMC2770817. Licensed under CC BY-NC 3.0.
- Figure 15 — Cavitary Pulmonary Tuberculosis. Adapted from: Yoo H, et al. Clinical investigation of cavitary tuberculosis. Korean J Intern Med. 2006;21:230. PMC3891027. Licensed under CC BY-NC 3.0.
- Figure 16 — TB Pathogenesis. Adapted from: Urbanowski ME, et al. Cavitary tuberculosis: The gateway of disease transmission. Lancet Infect Dis. 2020;20(6):e117. PMC7357333.
- Figures 17-19 — Pulmonary Aspergillosis. Adapted from: Jhun BW, et al. Pulmonary aspergillosis: What CT can offer before it is too late! JCDR. 2016;10:TE01. PMC4866217. Licensed under CC BY-NC 4.0.
- Figure 20 — PJP on CT. Adapted from: Jhun BW, et al. JCDR. 2016;10:TE01. PMC4866217. Licensed under CC BY-NC 4.0.
- Figures 21-22 — Pleural Effusion. Adapted from: Volpicelli G, et al. Lung ultrasound in the evaluation of pleural effusion. J Bras Pneumol. 2014;40:1. PMC4075927. Licensed under CC BY-NC 3.0.
- Figures 23-24 — Pneumothorax. Adapted from: Ocon-Brito A. Radiological review of pneumothorax. Postgrad Med J. 2005;81:210. PMC558461. Licensed under CC BY-NC 2.0.
- Figure 25 — Mesothelioma on CT. Adapted from: Tsim S, et al. Malignant pleural mesothelioma. J Thorac Imaging. 2018;33:411. PMC6053321.
- Figures 26-27 — Pulmonary Embolism. Adapted from: Defined CT imaging of acute pulmonary embolism. Cardiovasc Diagn Ther. 2018;8:225. PMC6039809. Licensed under CC BY-NC-ND 4.0.
- Figures 28-29 — Pulmonary Hypertension Hemodynamics. Adapted from: Hoeper MM, et al. Hemodynamic assessment of pulmonary hypertension. World J Cardiol. 2011;3:10. PMC3030732. Licensed under CC BY-NC 3.0.
- Figures 30-32 — Lung Cancer Imaging. Adapted from: Carter BW, et al. Lung cancer and radiological imaging. Curr Respir Care Rep. 2014;13:238. PMC8206195.
- Figures 33-34 — ARDS Imaging. Adapted from: Ball L, et al. Overview of current lung imaging in ARDS. Eur Respir Rev. 2022;31:134. PMC9487404. Licensed under CC BY-NC 4.0.
- Figures 35-36 — Ventilator Waveforms. Adapted from: Lian JX. The basics of ventilator waveforms. Curr Res Nurs J. 2021. PMC7782574. Licensed under CC BY 4.0.
- Figures 37-38 — OSA Pathophysiology. Adapted from: Eckert DJ, et al. Adult obstructive sleep apnea: pathophysiology and diagnosis. Clin Chest Med. 2009;30:455. PMC2813513.
- Figure 39 — Chronic Pulmonary Aspergillosis. Adapted from: Jhun BW, et al. JCDR. 2016;10:TE01. PMC4866217. Licensed under CC BY-NC 4.0.
- Figure 40 — Bronchoscopic Anatomy. Adapted from: Ge R, et al. J Thorac Dis. 2016;8(Suppl 2):S121. PMC4775257. Licensed under CC BY-NC-SA 4.0.
- Figure 41 — Chest Tube Placement. Adapted from: Ocon-Brito A. Radiological review of pneumothorax. Postgrad Med J. 2005;81:210. PMC558461. Licensed under CC BY-NC 2.0.
Key Trials & Guidelines
- GOLD Science Committee. Global Strategy for the Diagnosis, Management, and Prevention of COPD: 2024 Report. Am J Respir Crit Care Med. 2024. PMID: 36535226
- Leuppi JD, Schuetz P, Bingisser R, et al. Short-term vs conventional glucocorticoid therapy in acute exacerbations of COPD: the REDUCE randomized clinical trial. JAMA. 2013;309(21):2223-2231. PMID: 23695200
- Reddel HK, Bacharier LB, Bateman ED, et al. Global Initiative for Asthma Strategy 2021: executive summary and rationale for key changes. Eur Respir J. 2022;59(1):2102730. PMID: 31611859
- Raghu G, Remy-Jardin M, Myers JL, et al. Diagnosis of idiopathic pulmonary fibrosis. An official ATS/ERS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care Med. 2018;198(5):e44-e68. PMID: 29461346
- Idiopathic Pulmonary Fibrosis Clinical Research Network. Prednisone, azathioprine, and N-acetylcysteine for pulmonary fibrosis (PANTHER-IPF). N Engl J Med. 2012;366(21):1968-1977. PMID: 22607134
- Light RW, MacGregor MI, Luchsinger PC, Ball WC Jr. Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med. 1972;77(4):507-513. PMID: 4927027
- Rahman NM, Maskell NA, West A, et al. Intrapleural use of tissue plasminogen activator and DNase in pleural infection (MIST2). N Engl J Med. 2011;365(6):518-526. PMID: 21507925
- ARDS Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for ALI and ARDS. N Engl J Med. 2000;342(18):1301-1308. PMID: 10793162
- Guerin C, Reignier J, Richard JC, et al. Prone positioning in severe acute respiratory distress syndrome (PROSEVA). N Engl J Med. 2013;368(23):2159-2168. PMID: 23688302
- National Lung Screening Trial Research Team. Reduced lung-cancer mortality with low-dose CT screening. N Engl J Med. 2011;365(5):395-409. PMID: 21714641
- Evans L, Rhodes A, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47(11):1181-1247. PMID: 34599691
Textbooks & Reference Works
- Murray and Nadel's Textbook of Respiratory Medicine. 7th ed. Broaddus VC, Ernst JD, King TE, et al., eds. Elsevier; 2022.
- Fishman's Pulmonary Diseases and Disorders. 6th ed. Grippi MA, Elias JA, Fishman JA, et al., eds. McGraw-Hill; 2023.
- Marino's The ICU Book. 4th ed. Marino PL. Wolters Kluwer; 2014.
- West's Respiratory Physiology: The Essentials. 11th ed. West JB, Luks AM. Wolters Kluwer; 2021.