Electrophysiology
Every arrhythmia, device, ablation technique, classification, complication, medication, and management algorithm across the full scope of cardiac electrophysiology in one place.
01 Cardiac Conduction Anatomy
Sinoatrial (SA) Node
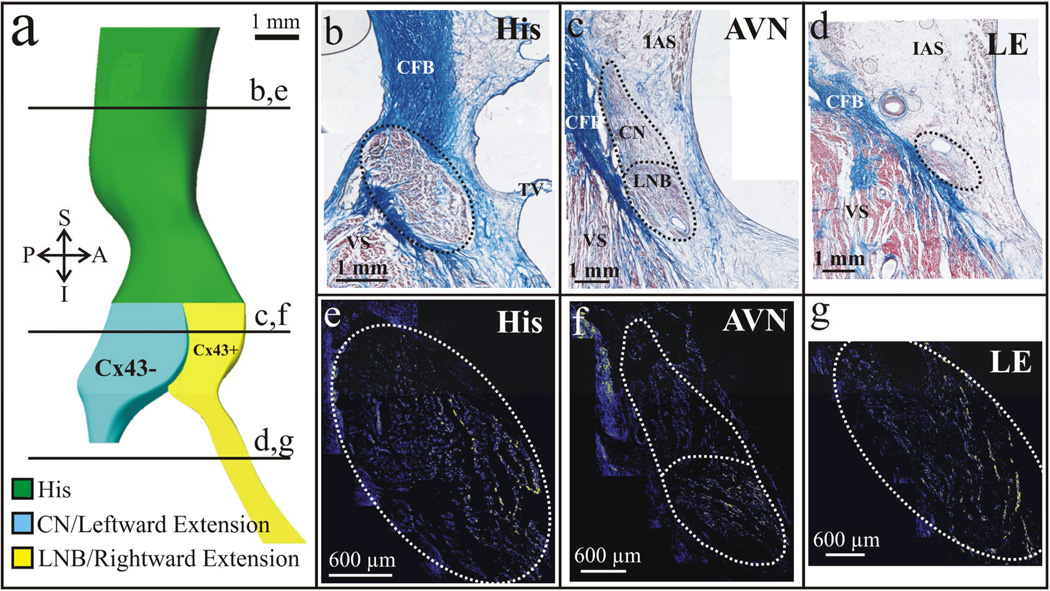
The SA node is the primary cardiac pacemaker, located at the junction of the superior vena cava (SVC) and the right atrium, in the sulcus terminalis. It is a crescent-shaped structure approximately 13-20 mm long. The SA node is supplied by the SA nodal artery — a branch of the RCA in ~55% of patients and of the LCx in ~45%. The SA node contains P cells (pacemaker cells) with the highest rate of spontaneous depolarization (60-100 bpm), which drives normal sinus rhythm. Autonomic innervation: parasympathetic (vagus nerve) slows the rate; sympathetic stimulation accelerates it.
Atrial Conduction
From the SA node, impulses propagate across both atria via preferential internodal pathways: the anterior (Bachmann bundle), middle (Wenckebach), and posterior (Thorel) pathways connect the SA node to the AV node. Bachmann bundle is the major interatrial conduction pathway, carrying the impulse from the right atrium to the left atrium. Disruption of Bachmann bundle conduction produces a biphasic P wave in the inferior leads (interatrial block) and is associated with atrial arrhythmias (Bayes syndrome).
Atrioventricular (AV) Node
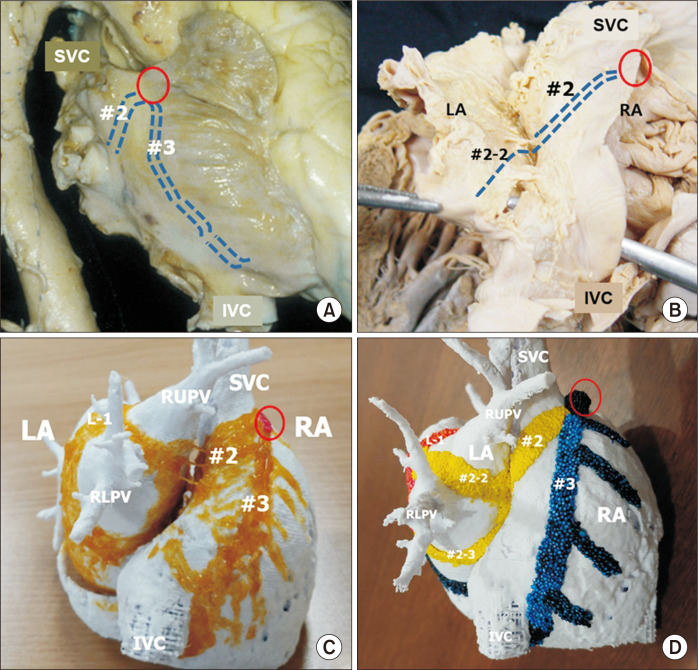
The AV node is located in the triangle of Koch, bounded by the coronary sinus ostium posteriorly, the tendon of Todaro superiorly, and the septal leaflet of the tricuspid valve inferiorly. The AV node is the only normal electrical connection between the atria and ventricles (the fibrous skeleton insulates them otherwise). It introduces a physiologic conduction delay (120-200 ms, reflected in the PR interval) that allows atrial contraction to complete before ventricular activation. The AV node is supplied by the AV nodal artery, typically a branch of the PDA (from the RCA in ~85% of patients). The AV node exhibits decremental conduction — faster atrial rates produce progressively slower conduction through the AV node, protecting the ventricles from excessively rapid atrial rhythms.
His Bundle, Bundle Branches & Purkinje Network
The bundle of His emerges from the AV node, penetrates the central fibrous body (membranous septum), and courses along the crest of the muscular interventricular septum. The His bundle is only ~20 mm long and divides into the right bundle branch (RBB) and left bundle branch (LBB). The RBB is a long, thin, cord-like structure that descends along the right side of the interventricular septum (travels in the moderator band) — its thin structure makes it vulnerable to injury. The LBB is broader and fans out into the left anterior fascicle (LAF) and left posterior fascicle (LPF). The LAF is thin and has a single blood supply (LAD), making it more susceptible to block than the LPF, which is thick and has dual blood supply (LAD and PDA).
The Purkinje network is a system of rapidly conducting fibers (conduction velocity ~4 m/s, the fastest in the heart) that distributes the impulse across the ventricular endocardium, ensuring near-simultaneous activation of both ventricles. The Purkinje system is the substrate for bundle branch reentrant VT and fascicular VT.
Coronary Sinus
The coronary sinus (CS) is the main venous drainage structure of the heart, running in the posterior atrioventricular groove and emptying into the right atrium. The CS ostium is guarded by the Thebesian valve. In EP, the CS is critically important: a CS catheter is routinely placed during EP studies to record left atrial activation. The CS is also the route for placing left ventricular leads during CRT implantation. CS tributaries include the great cardiac vein, middle cardiac vein, and posterolateral veins — target vessels for CRT lead placement.
Accessory Pathways
Accessory pathways (APs) are anomalous muscular connections between the atria and ventricles that bypass the AV node. They can occur anywhere around the mitral or tricuspid valve annuli. The most common location is the left free wall (~50-60%), followed by posteroseptal (~20-30%), right free wall (~10%), and anteroseptal (~5%). When an AP conducts anterogradely (atrium to ventricle), it produces pre-excitation (delta wave on ECG) — this is Wolff-Parkinson-White (WPW) pattern. Some APs only conduct retrogradely (concealed pathways) and do not produce a delta wave on baseline ECG.
02 Cellular Electrophysiology
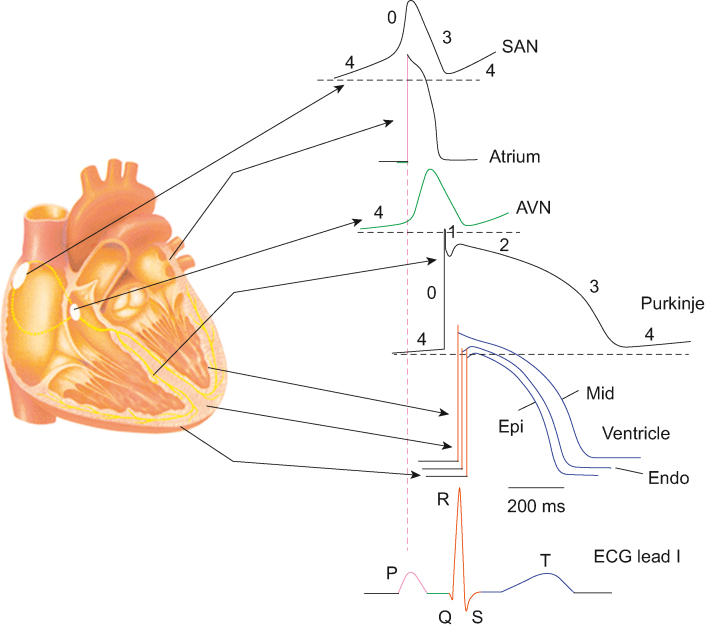
Cardiac Action Potential — Working Myocytes
The cardiac action potential in ventricular and atrial myocytes has five phases:
| Phase | Name | Ion Current | Mechanism |
|---|---|---|---|
| Phase 0 | Rapid depolarization | Na+ influx (INa) | Fast sodium channels open at threshold (~-70 mV); rapid upstroke; determines conduction velocity |
| Phase 1 | Early repolarization | K+ efflux (Ito) | Transient outward potassium current; produces the "notch" in the action potential |
| Phase 2 | Plateau | Ca2+ influx (ICa,L) balanced by K+ efflux | L-type calcium channels open; calcium entry triggers excitation-contraction coupling; this phase is responsible for the long duration of the cardiac AP |
| Phase 3 | Repolarization | K+ efflux (IKr, IKs, IK1) | Potassium channels dominate; membrane returns to resting potential; determines the QT interval |
| Phase 4 | Resting membrane potential | K+ (IK1) | Stable resting potential (~-90 mV) maintained by inward rectifier potassium current; no spontaneous depolarization in working myocytes |
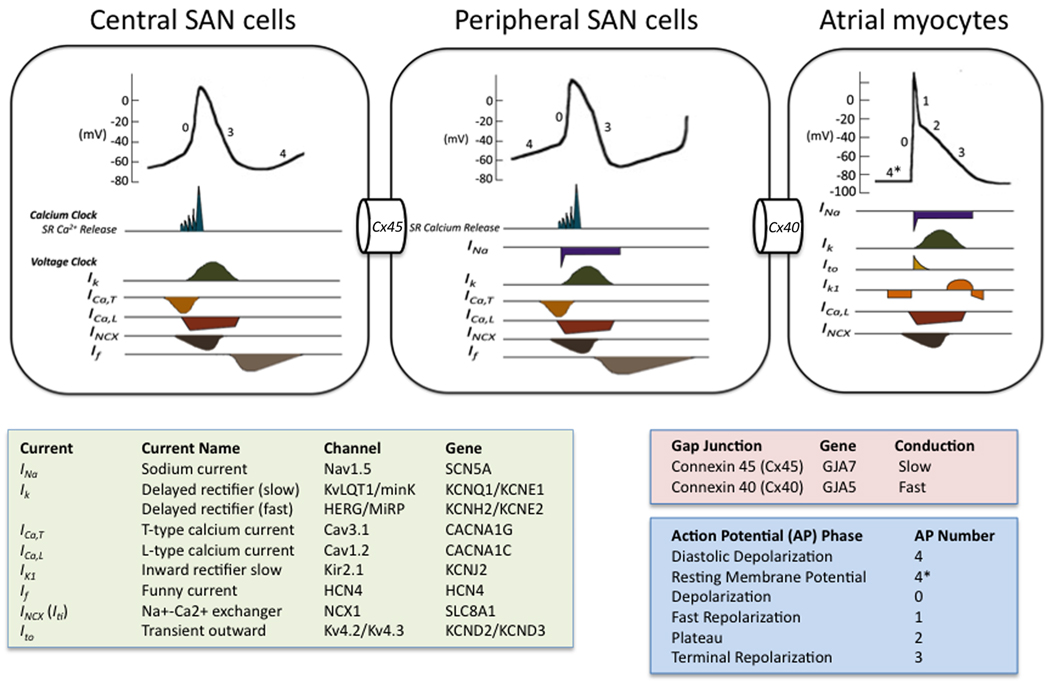
Pacemaker Cell Action Potential
SA and AV node cells lack fast sodium channels and have a distinctly different action potential. Phase 0 is mediated by L-type calcium channels (ICa,L) — producing a slow upstroke and slow conduction velocity (~0.05 m/s in the AV node vs ~0.5 m/s in atrial myocytes vs ~4 m/s in Purkinje fibers). There is no true Phase 1 or Phase 2. Phase 4 shows spontaneous diastolic depolarization driven by the funny current (If) — a mixed Na+/K+ inward current activated by hyperpolarization. This spontaneous Phase 4 depolarization is the basis of automaticity. The rate of Phase 4 depolarization determines the intrinsic firing rate of each pacemaker tissue: SA node 60-100 bpm (fastest, dominant pacemaker), AV node 40-60 bpm, His-Purkinje 20-40 bpm. This hierarchy ensures that the SA node normally drives the rhythm, with lower pacemakers serving as backup (escape rhythms). Ivabradine blocks If to reduce heart rate without affecting contractility or blood pressure — used for IST and as an adjunctive HF therapy.
The "calcium clock" mechanism also contributes to automaticity: spontaneous rhythmic calcium release from the sarcoplasmic reticulum via RyR2 channels activates the Na+/Ca2+ exchanger (NCX), generating an inward current that contributes to Phase 4 depolarization. This calcium clock mechanism is particularly relevant in CPVT, where abnormal RyR2 function leads to excessive calcium release and triggered DADs during adrenergic stimulation.
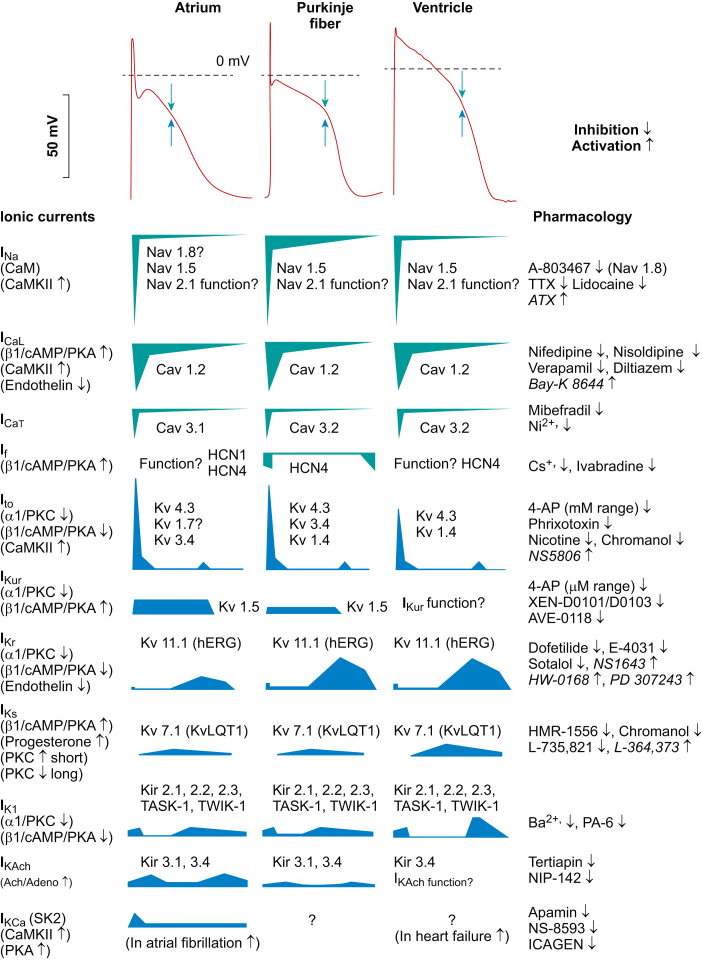
Ion Channels — Clinical Correlations
Sodium channels (INa, SCN5A gene): Mutations cause Brugada syndrome, LQT3, progressive cardiac conduction disease (Lenegre disease). Class I antiarrhythmics (flecainide, procainamide, lidocaine) block these channels. L-type calcium channels (ICa,L): Blocked by non-dihydropyridine CCBs (verapamil, diltiazem) — slows AV nodal conduction. Potassium channels: IKr (hERG, KCNH2) — blocked by Class III agents (sotalol, dofetilide, amiodarone); mutations cause LQT2. IKs (KCNQ1) — mutations cause LQT1 (most common LQTS). IK1 — maintains resting potential. Ito — prominent in epicardium; imbalance contributes to Brugada ECG pattern.
Refractoriness
Absolute refractory period (ARP): The cell cannot be re-excited regardless of stimulus strength (corresponds to Phases 0-2 and early Phase 3). Effective refractory period (ERP): The longest coupling interval at which a premature stimulus fails to propagate — this is what is measured during EP studies. Relative refractory period (RRP): A stronger-than-normal stimulus can trigger a propagated response, but conduction is slow and the action potential amplitude is reduced. Supernormal period: A brief period at the end of Phase 3 when a weaker-than-normal stimulus can trigger a response — rare but can cause unexpected conduction.
Mechanisms of Arrhythmias
All arrhythmias arise from one (or a combination) of three mechanisms:
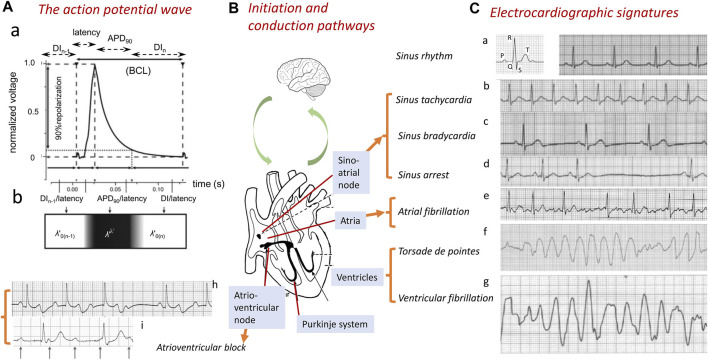
Requires three conditions: (1) two functionally distinct pathways, (2) unidirectional block in one pathway, and (3) slow conduction in the alternative pathway allowing recovery of excitability ahead of the advancing wavefront. Reentry can be anatomic (fixed circuit — AVNRT, AVRT, typical atrial flutter, scar-related VT) or functional (no fixed anatomic circuit — AF, VF). Reentrant arrhythmias are initiated and terminated by premature beats and respond to programmed stimulation — they are amenable to ablation.
Enhanced spontaneous Phase 4 depolarization in cells that normally have automaticity (SA node, AV node, His-Purkinje) or the development of automaticity in cells that do not normally exhibit it (injured atrial or ventricular myocytes — often from ischemia, electrolyte abnormalities, or catecholamine excess). Automatic arrhythmias show a "warm-up" and "cool-down" pattern, cannot be initiated or terminated by programmed stimulation, and are suppressed by overdrive pacing. Examples: accelerated idioventricular rhythm, multifocal atrial tachycardia, some focal atrial tachycardias.
Early afterdepolarizations (EADs): Occur during Phase 2 or Phase 3 when repolarization is prolonged. The classic example is torsades de pointes in the setting of QT prolongation. EADs are bradycardia-dependent and exacerbated by hypokalemia and hypomagnesemia. Treatment: magnesium, isoproterenol, temporary overdrive pacing (to shorten the QT).
Delayed afterdepolarizations (DADs): Occur after full repolarization (during Phase 4) and are caused by intracellular calcium overload. DADs are exacerbated by rapid heart rates and catecholamines. Classic examples: digitalis toxicity (bidirectional VT), catecholaminergic polymorphic VT (CPVT), right ventricular outflow tract (RVOT) VT. Treatment: address the underlying cause; for digitalis toxicity — DigiFab, correct K+/Mg2+.
03 ECG Interpretation for EP
Key Intervals
| Interval | Normal Value | Clinical Significance |
|---|---|---|
| PR interval | 120-200 ms | Short PR (< 120 ms): pre-excitation (WPW), enhanced AV conduction, ectopic atrial rhythm. Long PR (> 200 ms): first-degree AV block |
| QRS duration | < 120 ms | 120-149 ms: incomplete BBB or IVCD. ≥ 150 ms: complete BBB. Wide QRS tachycardia: VT vs SVT with aberrancy |
| QT interval | 350-450 ms (men), 350-460 ms (women) | QTc (Bazett) = QT / √RR. QTc > 500 ms: high risk for torsades. QTc < 340 ms: short QT syndrome |
| JT interval | Variable | More accurate assessment of repolarization when QRS is prolonged (BBB, pacing) |
Axis Determination
Normal axis: -30° to +90°. Left axis deviation (LAD): -30° to -90° — suggests left anterior fascicular block (LAFB), inferior MI, or LVH. Right axis deviation (RAD): +90° to +180° — suggests left posterior fascicular block (LPFB), RVH, lateral MI, or PE. Extreme/northwest axis: -90° to ±180° — ventricular tachycardia until proven otherwise.
Quick axis assessment: Look at leads I and aVF. Both positive = normal axis. Lead I positive, aVF negative = left axis deviation (further check lead II — if positive, still normal). Lead I negative, aVF positive = right axis deviation. Both negative = extreme axis deviation ("no man's land"). In the EP lab, the axis of a wide-complex tachycardia helps identify the VT origin: LBBB + inferior axis = RVOT origin; LBBB + superior axis = RV inferior wall or inferoseptal LV; RBBB + left axis = LV posteroseptal (fascicular VT); RBBB + right axis = LV anterolateral.
Bundle Branch Block Patterns
RBBB: QRS ≥ 120 ms, rsR' ("rabbit ears") in V1-V2, wide slurred S in I and V6. The initial forces are normal (septal activation is unchanged). RBBB can be a normal variant or indicate RV strain (PE), ASD, or conduction disease. LBBB: QRS ≥ 120 ms, broad monophasic R in I and V6, QS or rS in V1, absence of q in I and V6. LBBB always alters initial forces, making it impossible to assess for prior MI by Q waves. New LBBB in the setting of chest pain was previously considered a STEMI equivalent (now guidelines focus on ischemic symptoms regardless of LBBB status).
Wide-Complex Tachycardia: VT vs SVT with Aberrancy
Differentiating VT from SVT with aberrancy is one of the most important ECG skills in EP. Features favoring VT: AV dissociation (independent P waves — most specific), fusion beats, capture beats, extreme axis deviation ("northwest axis"), concordance (all precordial leads positive or negative), very wide QRS (> 160 ms), Brugada criteria (onset to nadir > 100 ms in any precordial lead), Vereckei aVR criteria (initial R wave in aVR, initial r or q > 40 ms, notch on descending limb of negative QRS in aVR, Vi/Vt ratio ≤ 1). Features favoring SVT with aberrancy: Typical RBBB or LBBB morphology, prior BBB on baseline ECG, prior history of SVT. Clinical pearl: When in doubt, treat as VT — this is safer than assuming SVT and giving a calcium channel blocker or adenosine (which can be fatal in VT or pre-excited AF).
Pre-excitation (WPW Pattern)
The ECG hallmarks of ventricular pre-excitation are: (1) short PR interval (< 120 ms), (2) delta wave (slurred initial QRS upstroke), and (3) widened QRS (> 120 ms). The delta wave polarity in the 12-lead ECG can localize the accessory pathway location (delta wave algorithms — Arruda, Fitzpatrick, Milstein). Left free wall pathways produce positive delta waves in V1; right-sided/septal pathways produce negative delta waves in V1.
Brugada ECG Pattern
Type 1 (coved): ST elevation ≥ 2 mm with a coved morphology in V1-V3, followed by T-wave inversion — the only diagnostic pattern. Type 2 (saddleback): ST elevation ≥ 2 mm with saddleback morphology — suggestive but not diagnostic; requires provocation with sodium channel blockers (ajmaline, procainamide, flecainide) to convert to Type 1 for diagnosis. High precordial leads (V1-V2 placed in the 2nd or 3rd intercostal space) increase sensitivity.
Long QT Morphology Patterns
LQT1 (KCNQ1): Broad-based T waves. LQT2 (KCNH2/hERG): Low-amplitude, bifid/notched T waves. LQT3 (SCN5A): Long isoelectric ST segment followed by a peaked, narrow T wave. These morphologic patterns can suggest the genotype before genetic testing is available and guide initial therapy.
Other EP-Relevant ECG Findings
Epsilon waves: Small positive deflections at the end of the QRS in V1-V3 — pathognomonic for arrhythmogenic right ventricular cardiomyopathy (ARVC). Early repolarization pattern: J-point elevation ≥ 1 mm in inferior or lateral leads. Once considered benign, now recognized as a potential marker for VF risk (early repolarization syndrome), especially when associated with horizontal or descending ST segments. J waves (Osborn waves): Prominent in hypothermia; also seen in Brugada syndrome and early repolarization syndrome.
04 The Electrophysiology Study
Intracardiac Catheters
Standard diagnostic EP study uses four multipolar catheters placed via the femoral veins (and sometimes the internal jugular vein):
| Catheter | Position | Records |
|---|---|---|
| High right atrium (HRA) | Right atrial appendage or high lateral RA | Atrial electrograms; used for atrial pacing |
| His bundle catheter | Across the tricuspid valve at the His bundle region | A (atrial), H (His), and V (ventricular) electrograms; measures AH and HV intervals |
| Coronary sinus (CS) catheter | Coronary sinus (decapolar catheter) | Left atrial activation sequence; essential for SVT differential diagnosis |
| Right ventricular (RV) catheter | RV apex | Ventricular electrograms; used for ventricular pacing and programmed stimulation |
Intracardiac Electrograms & Intervals
The His bundle electrogram records three deflections: A (atrial activation), H (His bundle depolarization), and V (ventricular activation). Two critical intervals are measured:
AH interval (60-125 ms): Represents conduction time through the AV node. Prolonged by vagal maneuvers, adenosine, beta-blockers, calcium channel blockers. Prolonged AH suggests AV nodal disease. HV interval (35-55 ms): Represents conduction time from the His bundle through the bundle branches to the ventricular myocardium. HV > 70 ms is markedly prolonged and indicates infranodal conduction disease — a potential indication for pacing. HV < 35 ms suggests pre-excitation (the ventricle is activated early via an accessory pathway). The HV interval is not affected by vagal maneuvers or adenosine (His-Purkinje tissue is not influenced by the autonomic nervous system).
Programmed Stimulation Protocols
Atrial programmed stimulation: Decremental atrial pacing (progressively shorter cycle lengths) to determine the AV node Wenckebach cycle length and the atrial ERP. Atrial extra-stimulus testing (S1-S2, S1-S2-S3) to induce SVT. A "jump" in the AH interval (> 50 ms increase with a 10 ms decrement in coupling interval) during atrial extra-stimulus testing indicates dual AV node physiology (fast pathway blocks, slow pathway conducts) and is the hallmark of AVNRT substrate.
Ventricular programmed stimulation: Used for VT induction — the standard protocol involves pacing from the RV apex and RVOT at two drive cycle lengths (600 ms and 400 ms) with up to three extra-stimuli (S1-S2, S1-S2-S3, S1-S2-S3-S4) decremented to the ventricular ERP (or 200 ms). Induction of sustained monomorphic VT (> 30 seconds or requiring termination due to hemodynamic compromise) is considered a positive study. Induction of polymorphic VT or VF with aggressive protocols (triple extra-stimuli at short coupling intervals) is considered a non-specific finding.
Mapping Techniques During EP Study
Activation mapping: Records the timing of local electrograms relative to a reference; the earliest activation site is the target for ablation (focal tachycardias, accessory pathways). Entrainment mapping: Pacing during tachycardia to assess whether a site is within the reentrant circuit (post-pacing interval minus tachycardia cycle length < 30 ms suggests the site is within the circuit). Pace mapping: Pacing from a site and comparing the QRS morphology to the clinical VT — a 12/12 lead match suggests the site is at or near the VT origin. Substrate mapping: Voltage mapping to identify scar (bipolar voltage < 0.5 mV in ventricle, < 0.5 mV in atrium) and border zone tissue for scar-based ablation strategies.
Transseptal Puncture
Transseptal access is required for left atrial procedures (AF ablation, left-sided AP ablation, mitral isthmus ablation). The technique involves: advancing a transseptal needle and sheath from the femoral vein, through the IVC, into the right atrium, and puncturing the fossa ovalis (the thinnest portion of the interatrial septum). Guidance: fluoroscopy (RAO and LAO views), intracardiac echocardiography (ICE — provides real-time visualization of the septum, tenting sign), and pressure waveform monitoring. Complications: cardiac tamponade (most feared — puncture into the aorta or posterior LA wall), air embolism, and pericardial effusion. A patent foramen ovale (present in ~25% of the population) may allow access without puncture.
Intracardiac Echocardiography (ICE)
ICE is increasingly used during EP procedures. A phased-array ultrasound catheter (AcuNav, Siemens) is placed in the right atrium (or rarely the LA) and provides real-time imaging of cardiac structures. Uses: guide transseptal puncture (visualize septum tenting), monitor for complications (pericardial effusion, tamponade), confirm catheter-tissue contact, visualize PV anatomy, detect thrombus, and assess lesion formation. ICE has reduced the need for general anesthesia and TEE during AF ablation at many centers.
05 Atrial Fibrillation
Classification
| Type | Definition |
|---|---|
| Paroxysmal AF | Terminates spontaneously or with intervention within 7 days of onset |
| Persistent AF | Continuous AF lasting > 7 days, including episodes terminated by cardioversion after ≥ 7 days |
| Long-standing persistent AF | Continuous AF lasting > 12 months when a rhythm control strategy is pursued |
| Permanent AF | AF accepted by the patient and physician; no further rhythm control attempts (a joint clinical decision, not a pathophysiologic classification) |
Pathophysiology
AF requires a trigger and a substrate. The landmark work by Haïssaguerre et al. (1998) demonstrated that ectopic foci in the pulmonary veins (PVs) are the primary triggers for AF initiation in most patients — this discovery is the foundation of PV isolation ablation. The substrate involves progressive atrial remodeling: electrical remodeling (shortened atrial ERP — "AF begets AF"), structural remodeling (fibrosis, dilation), and autonomic remodeling (ganglionated plexi). Risk factors include: hypertension, obesity, OSA, valvular disease (especially mitral), heart failure, thyroid disease, and heavy alcohol use ("holiday heart syndrome").
Haïssaguerre M et al. N Engl J Med. 1998;339:659-666. PMID: 9533160
Rate vs Rhythm Control
AFFIRM trial (2002): No mortality difference between rate control and rhythm control (with antiarrhythmic drugs) in older patients with AF and risk factors for stroke. Anticoagulation should be continued regardless of strategy. EAST-AFNET 4 trial (2020): Early rhythm control (within 1 year of AF diagnosis) using antiarrhythmic drugs and/or ablation reduced the composite of cardiovascular death, stroke, and hospitalization for HF or ACS compared with rate control. This paradigm-shifting trial has pushed the field toward earlier rhythm control, especially in patients with recently diagnosed AF.
AFFIRM Investigators. N Engl J Med. 2002;347:1825-1833. PMID: 12466507 Kirchhof P et al. N Engl J Med. 2020;383:1305-1316. PMID: 32865375Anticoagulation
Stroke risk is assessed using the CHA2DS2-VASc score: Congestive heart failure (1), Hypertension (1), Age ≥ 75 (2), Diabetes (1), Stroke/TIA/thromboembolism (2), Vascular disease (1), Age 65-74 (1), Sex category — female (1). Anticoagulation is recommended for men with score ≥ 2 and women with score ≥ 3. Score of 1 (men) or 2 (women) — consider anticoagulation. Score of 0 (men) or 1 (women with sex as only point) — no anticoagulation needed.
Bleeding risk is assessed with the HAS-BLED score: Hypertension (uncontrolled, SBP > 160), Abnormal renal/liver function (1 each), Stroke, Bleeding (prior major), Labile INR (< 60% time in therapeutic range), Elderly (> 65), Drugs (antiplatelets, NSAIDs) or alcohol (1 each). Score ≥ 3 = high bleeding risk — this should prompt modification of reversible risk factors, not withholding anticoagulation if it is indicated.
DOACs (dabigatran, rivaroxaban, apixaban, edoxaban) are preferred over warfarin for non-valvular AF (2019 AHA/ACC/HRS guidelines, Class I). Exceptions requiring warfarin: mechanical heart valves, moderate-to-severe mitral stenosis (rheumatic). Among DOACs, apixaban has the best bleeding profile (ARISTOTLE trial).
Cardioversion
For AF of unknown duration or ≥ 48 hours, either (1) anticoagulate for ≥ 3 weeks before cardioversion, or (2) perform TEE-guided cardioversion (exclude left atrial appendage thrombus, then cardiovert, then anticoagulate for ≥ 4 weeks after). For AF clearly < 48 hours, cardioversion can proceed without prior TEE or prolonged anticoagulation (but anticoagulation should be considered based on CHA2DS2-VASc). Electrical cardioversion: Synchronized, biphasic shock 200 J (anterior-posterior pad placement preferred). Pharmacologic cardioversion: Flecainide (300 mg PO "pill-in-pocket" for patients without structural heart disease), ibutilide (IV, risk of torsades), amiodarone (IV or PO, slower conversion).
Left Atrial Appendage Closure
For AF patients who cannot tolerate long-term anticoagulation (e.g., prior life-threatening hemorrhage), left atrial appendage (LAA) occlusion is an alternative stroke prevention strategy. The Watchman device (Boston Scientific) is the most studied percutaneous LAA occluder. The PROTECT AF and PREVAIL trials demonstrated non-inferiority to warfarin for stroke prevention. The device is delivered via transseptal access and deployed at the LAA ostium, sealing it off. Post-implant: dual antiplatelet therapy (DAPT) for 6 months, then aspirin alone (or DOAC for 45 days then DAPT in newer protocols). The LARIAT system uses epicardial suture ligation of the LAA. Surgical LAA exclusion (clipping or stapling) is performed during concurrent cardiac surgery.
AF Ablation Overview
Pulmonary vein isolation (PVI) is the cornerstone of AF ablation. All four PVs are electrically isolated from the left atrium. Techniques include: point-by-point radiofrequency ablation (wide-area circumferential ablation — WACA), cryoballoon ablation (FIRE AND ICE trial showed non-inferiority to RF for paroxysmal AF), and pulsed field ablation (PFA) — the newest modality with tissue selectivity for myocardium (spares esophagus, phrenic nerve). Additional targets beyond PVI for persistent AF: posterior wall isolation, CTI line, mitral isthmus line, complex fractionated atrial electrograms (CFAE — controversial), rotor ablation (CONFIRM trial — results not replicated). CABANA trial (2019): Ablation vs drug therapy — no significant difference in the primary ITT endpoint, but ablation significantly reduced AF recurrence and improved quality of life.
06 Atrial Flutter
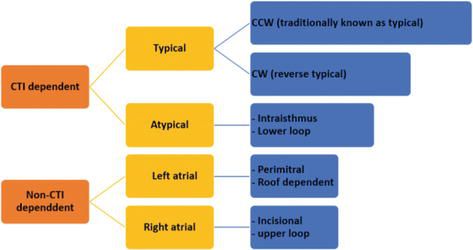
Typical (CTI-Dependent) Atrial Flutter
Typical atrial flutter is a macro-reentrant circuit that travels around the tricuspid valve annulus, with the cavotricuspid isthmus (CTI) — the region between the IVC, Eustachian ridge, and tricuspid annulus — as the critical isthmus. In counterclockwise (typical) flutter, the circuit goes up the septum, across the roof, down the lateral wall, and across the CTI. This produces the classic "sawtooth" pattern of flutter waves (inverted in inferior leads II, III, aVF; upright in V1) at a rate of ~300 bpm with 2:1 conduction giving a ventricular rate of ~150 bpm. Clockwise (reverse typical) flutter uses the same circuit in the opposite direction — upright flutter waves in the inferior leads.
Atypical Atrial Flutter
Atypical flutter refers to any macro-reentrant atrial tachycardia that does not use the CTI. Common substrates include: scar-related flutter (post-cardiac surgery, post-AF ablation — particularly around gaps in prior ablation lines), perimitral flutter (circuit around the mitral annulus), and left atrial roof-dependent flutter. The ECG does not show the classic sawtooth pattern, and the rate may be faster or slower than typical flutter. 3D electroanatomical mapping is required for diagnosis and ablation.
Treatment
CTI ablation for typical flutter has a success rate > 95% with a recurrence rate of ~5-10%. It is curative and is a Class I recommendation for recurrent or symptomatic typical flutter. Ablation involves creating a line of lesions across the CTI (from the tricuspid annulus to the IVC) and confirming bidirectional block. Bidirectional block is confirmed by pacing on each side of the ablation line and demonstrating that the activation wavefront must travel the long way around the tricuspid annulus (rather than crossing the CTI).
Rate control is often difficult in flutter (unlike AF) because the flutter circuit is stable and the atrial rate is relatively slow (~300 bpm), making it easy for the AV node to conduct 2:1 (rate ~150 bpm). Increasing AV nodal blockade may convert 2:1 to 4:1 (rate ~75 bpm) but this is unreliable. Class Ic drugs (flecainide, propafenone) should be avoided in flutter without a rate-control agent because they can slow the atrial flutter rate to ~200 bpm, at which point the AV node may conduct 1:1, producing a ventricular rate of ~200 bpm with hemodynamic collapse.
Note: ~30-50% of patients with typical flutter will develop AF in long-term follow-up — patients should be counseled about this risk and monitored. Anticoagulation follows the same CHA2DS2-VASc criteria as AF.
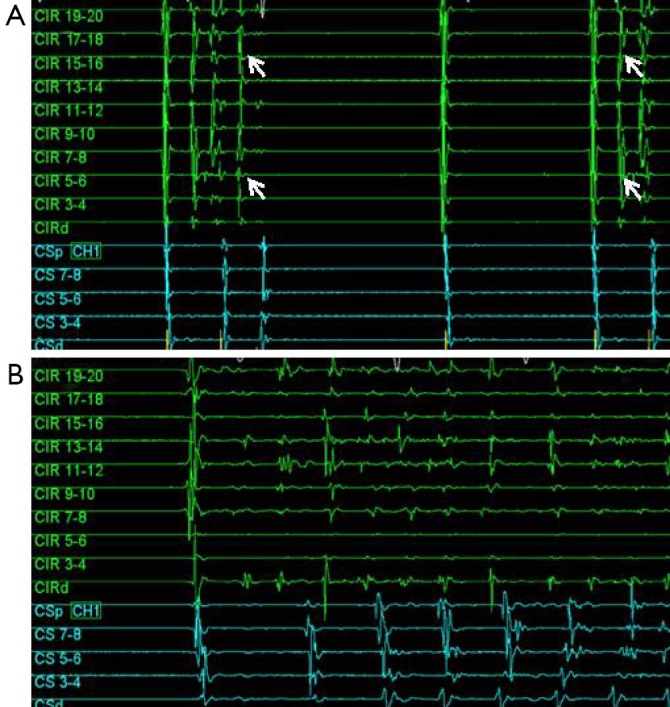
07 AVNRT
Mechanism & Dual AV Node Physiology
Atrioventricular nodal reentrant tachycardia (AVNRT) is the most common regular SVT (~60% of all SVTs). It utilizes dual AV node pathways: a fast pathway (located superiorly/anteriorly, with a long refractory period) and a slow pathway (located inferiorly/posteriorly, near the CS ostium, with a short refractory period). In typical (slow-fast) AVNRT (~90%), a PAC blocks in the fast pathway (which is still refractory), conducts down the slow pathway, and then returns up the now-recovered fast pathway — creating a reentrant circuit. Because the atria and ventricles are activated nearly simultaneously, the P wave is buried in or just after the QRS (pseudo-S in inferior leads, pseudo-r' in V1).
Atypical AVNRT
Fast-slow (atypical) AVNRT: Conducts antegradely through the fast pathway and retrogradely through the slow pathway — produces a long RP tachycardia (P wave well after the QRS, inverted in inferior leads). Must be differentiated from atypical AVRT (PJRT) and atrial tachycardia. Slow-slow AVNRT: Uses two slow pathways — another cause of long RP tachycardia. These atypical forms account for ~10% of AVNRT cases and can be more challenging to diagnose and ablate.
EP clues to AVNRT diagnosis: VA interval < 70 ms (near-simultaneous A and V — highly specific for typical AVNRT), AH jump during atrial extra-stimulus testing (confirming dual pathway physiology), inability to advance atrial activation with ventricular extra-stimuli delivered during His refractoriness (excludes an accessory pathway), and a "V-A-A-V" response to ventricular overdrive pacing (suggests AVNRT rather than AVRT). The His-refractory PVC maneuver is particularly useful: a PVC delivered during tachycardia when the His bundle is refractory cannot reach the atrium via the AV node — if it still advances the next atrial activation, an accessory pathway must be present (AVRT, not AVNRT).
Acute Management
Step 1 — Vagal maneuvers: Modified Valsalva (blowing against resistance while supine, then leg elevation — REVERT trial showed 43% termination rate, superior to standard Valsalva), carotid sinus massage (avoid in patients with carotid bruits or known carotid stenosis — risk of stroke), diving reflex (cold water to face). Step 2 — Adenosine: 6 mg rapid IV push via antecubital vein followed by 20 mL saline flush and arm elevation. If ineffective, 12 mg (may repeat x1). Give through a large-bore IV as proximal as possible; rapid push is critical due to the ultra-short half-life (~10 seconds). Warn the patient about brief chest tightness and "sense of doom." Step 3 — If adenosine fails: IV verapamil (5-10 mg over 2-3 min) or IV diltiazem (0.25 mg/kg over 2 min); or IV metoprolol (5 mg q5min x3). Avoid combining CCBs and beta-blockers IV (risk of severe bradycardia/asystole).
Definitive Treatment — Slow Pathway Ablation
Slow pathway ablation is the definitive treatment for recurrent AVNRT. RF energy is delivered to the posteroinferior AV nodal inputs (slow pathway region) in the triangle of Koch, guided by fluoroscopy (catheter positioned in the posteroseptal tricuspid annulus) and electrogram characteristics (low-amplitude atrial signal, large ventricular signal, and absence of a His deflection). The appearance of junctional rhythm during ablation indicates proximity to the slow pathway and effective lesion delivery. Ablation is continued until junctional rhythm appears and dual AV node physiology is eliminated (no jump on repeat atrial extra-stimulus testing). Success rate: > 95%. Risk of inadvertent complete heart block: < 1% (the fast pathway and compact AV node are preserved). Cryoablation can be used in the slow pathway region, particularly in younger patients, to minimize AV block risk — ice mapping allows testing before permanent lesion creation (reversible at -30°C, permanent at -80°C).
08 AVRT & Wolff-Parkinson-White Syndrome
Definitions
WPW pattern: Pre-excitation on ECG (delta wave, short PR, wide QRS) without symptoms. WPW syndrome: Pre-excitation plus symptomatic tachyarrhythmias. AVRT: A reentrant tachycardia using the AV node as one limb and an accessory pathway as the other limb.
Types of AVRT
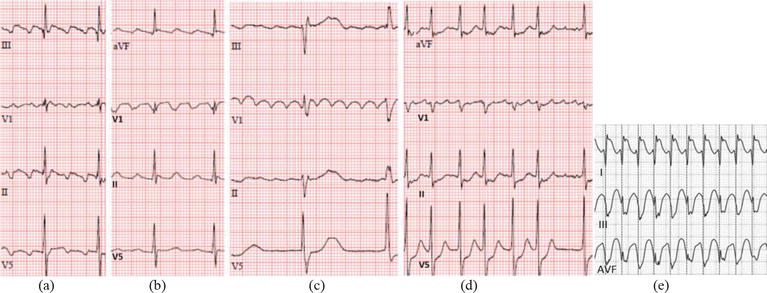
Orthodromic AVRT (~95% of AVRTs): Conducts antegradely through the AV node and retrogradely through the accessory pathway. Produces a narrow-complex tachycardia (because ventricular activation occurs normally through the His-Purkinje system). The P wave follows the QRS (short RP tachycardia). Antidromic AVRT (~5%): Conducts antegradely through the accessory pathway and retrogradely through the AV node. Produces a wide-complex tachycardia (maximal pre-excitation, fully pre-excited QRS) that may mimic VT.
Pre-excited Atrial Fibrillation Emergency
Pre-excited AF (AF conducting over an accessory pathway with a short refractory period) is a life-threatening emergency. The ECG shows a rapid, irregularly irregular wide-complex tachycardia with varying QRS morphology. Ventricular rates can exceed 250-300 bpm and degenerate into VF. CRITICAL: AV nodal blocking agents (adenosine, beta-blockers, calcium channel blockers, digoxin) are absolutely contraindicated — they block the AV node, forcing all conduction down the accessory pathway and potentially causing VF and death. Treatment: IV procainamide (slows AP conduction) or synchronized electrical cardioversion if hemodynamically unstable.
Risk Stratification in WPW
The primary concern in asymptomatic WPW is the risk of sudden cardiac death from pre-excited AF degenerating into VF. Risk assessment includes: non-invasive: intermittent pre-excitation on ECG or abrupt loss of pre-excitation during exercise testing suggests a long AP ERP (low risk). Invasive (EP study): shortest pre-excited RR interval (SPERRI) during induced AF — SPERRI ≤ 250 ms indicates a high-risk pathway. Accessory pathway ERP ≤ 250 ms is also high risk. Guidelines recommend EP study and ablation for symptomatic patients (Class I) and suggest considering EP study in asymptomatic patients, especially in high-risk occupations (first responders, competitive athletes).
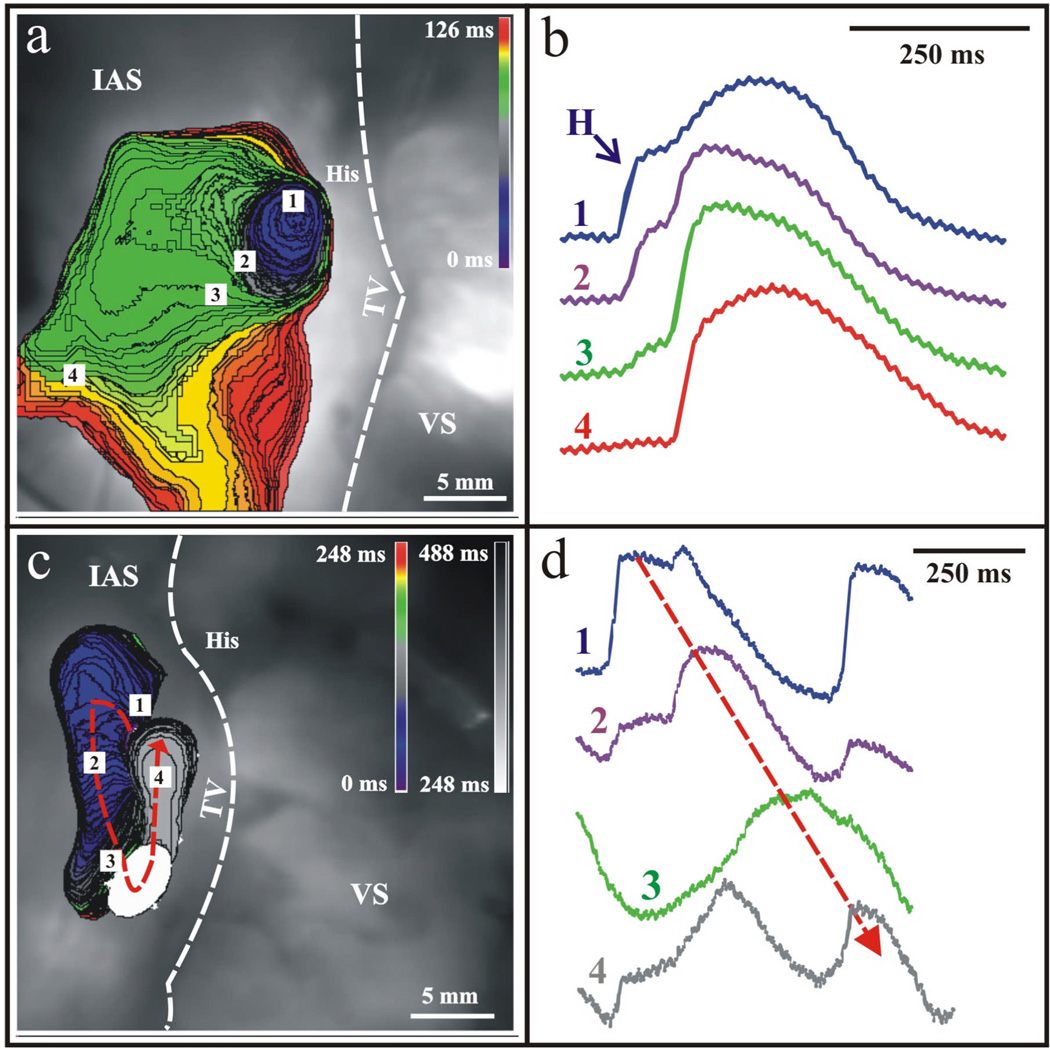
Accessory Pathway Ablation
Ablation success rate is > 95% for left-sided pathways (approached via retrograde aortic or transseptal approach) and ~90% for right-sided pathways. Septal pathways carry higher risk of AV block (particularly anteroseptal/parahisian pathways — cryoablation is preferred for these locations). Recurrence rate: ~5% overall. EP study and ablation are curative and are the definitive treatment for WPW syndrome.
Left free wall pathways (~50-60%): Accessed via retrograde aortic approach (femoral artery, across the aortic valve) or transseptal approach (femoral vein, through the interatrial septum). Both approaches have excellent success rates. The retrograde approach provides good catheter stability; the transseptal approach avoids arterial access and is preferred by many operators.
Right free wall pathways (~10%): Accessed directly via the femoral vein. Lower success rates (~85-90%) due to catheter instability and thinner tissue. May require steerable sheaths for stability.
Posteroseptal pathways (~20-30%): Can be epicardial (within the coronary sinus or middle cardiac vein) or endocardial. May require ablation inside the CS or from multiple approaches. CS diverticula are a substrate for some posteroseptal pathways.
Anteroseptal/parahisian pathways (~5%): Highest risk of AV block due to proximity to the His bundle. Cryoablation is preferred (allows ice mapping — test at -30°C before committing to a permanent lesion at -80°C). If cryoablation fails, RF with low power and close monitoring of the His bundle electrogram may be attempted.
09 Atrial Tachycardia
Focal Atrial Tachycardia
Arises from a single atrial focus with centrifugal spread of activation. Mechanism can be automaticity (most common — warm-up/cool-down behavior), triggered activity (DAD-mediated, sensitive to adenosine), or micro-reentry. Common sites of origin: crista terminalis, tricuspid annulus, coronary sinus ostium, pulmonary veins, mitral annulus, and left atrial appendage. ECG shows a distinct P-wave morphology different from sinus P waves; the P-wave axis and morphology can localize the focus.
Right atrial origins: P-wave positive in lead I, negative or isoelectric in aVL. Crista terminalis: P-wave morphology similar to sinus P wave (positive in II, III, aVF with slight variations). Tricuspid annulus: Low-amplitude P waves, negative in V1.
Left atrial origins: P-wave negative in lead I, positive in aVL. Pulmonary veins: Positive in V1 (due to posterior-to-anterior activation). Left atrial appendage: Positive in V1, notched P waves in inferior leads.
Superior origins (SVC, RSPV, LSPV): P-wave axis inferior (positive in II, III, aVF). Inferior origins (CS ostium, inferior PVs): P-wave axis superior (negative in II, III, aVF).
Isoelectric baseline between P waves distinguishes AT from flutter (continuous atrial activity). Adenosine may terminate (triggered) or transiently suppress (automatic) AT, or have no effect (micro-reentrant). Ablation success rate for focal AT is ~85-95%, depending on location.
Macro-Reentrant Atrial Tachycardia
Organized reentrant circuits in the atria, often related to prior surgery or ablation scars. Includes atypical atrial flutter (discussed in Section 06) and incisional tachycardia (e.g., following repair of congenital heart disease — Fontan, Mustard/Senning, ASD closure). 3D electroanatomical mapping is essential for identifying the circuit and critical isthmus. Ablation targets the narrowest isthmus of the circuit, often between two scars or between a scar and an anatomic barrier (e.g., between a surgical scar and the mitral annulus, or between two prior ablation lines).
Post-AF ablation macro-reentrant AT is increasingly common as more patients undergo AF ablation. These tachycardias often arise from gaps in prior PVI lines (reconnection-related), or new circuits formed around linear lesion sets (roof line, mitral isthmus line). They can be faster or slower than the original AF and are often highly symptomatic. Repeat ablation with high-density mapping to identify and close the gaps or critical isthmus has a high success rate (~80-90%).
Multifocal Atrial Tachycardia (MAT)
Defined by: rate > 100 bpm, ≥ 3 different P-wave morphologies, varying PP intervals, and varying PR intervals. Most commonly seen in patients with severe pulmonary disease (COPD), hypoxia, or critical illness. NOT a reentrant rhythm — caused by abnormal automaticity, often triggered by theophylline, hypoxia, hypomagnesemia. Treatment: correct the underlying cause, IV magnesium, rate control with non-dihydropyridine CCBs. Avoid beta-blockers in severe COPD. Cardioversion and antiarrhythmic drugs are ineffective.
Inappropriate Sinus Tachycardia (IST)
IST is a diagnosis of exclusion characterized by a resting heart rate > 100 bpm (or average 24-hour rate > 90 bpm) with normal P-wave morphology and appropriate 1:1 AV conduction, in the absence of secondary causes (anemia, hyperthyroidism, deconditioning, pain, anxiety, pheochromocytoma). Predominantly affects young women. Mechanism: enhanced automaticity of the SA node or inappropriate autonomic regulation. Treatment: ivabradine (If channel blocker — most effective, reduces HR without affecting BP), beta-blockers (may be poorly tolerated due to hypotension), and physical reconditioning. SA node modification by catheter ablation is a last resort (risk of sinus node destruction requiring pacemaker).
Postural Orthostatic Tachycardia Syndrome (POTS)
POTS is defined by a sustained increase in heart rate ≥ 30 bpm (or absolute HR ≥ 120 bpm) within 10 minutes of standing, without orthostatic hypotension, associated with chronic symptoms (≥ 6 months). Not a primary arrhythmia, but frequently referred to EP for evaluation. Subtypes: neuropathic POTS (peripheral autonomic denervation, especially lower extremity), hyperadrenergic POTS (elevated norepinephrine levels), and hypovolemic POTS. Treatment: non-pharmacologic measures first (increased fluid/salt intake, compression garments, exercise reconditioning), then medications (low-dose beta-blockers, midodrine, fludrocortisone, ivabradine). POTS is not treated with ablation or devices.
10 SVT Differential & Management Algorithm
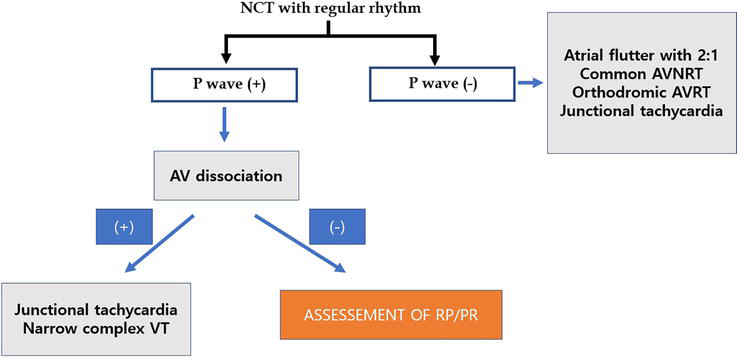
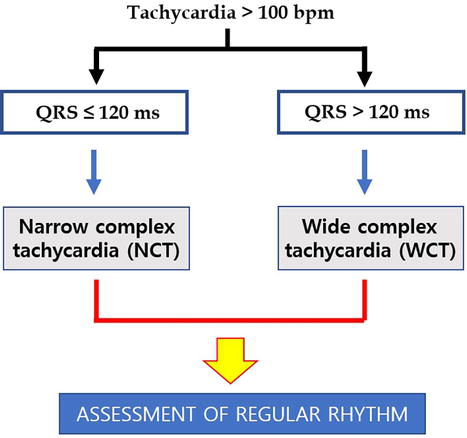
Regular Narrow-Complex Tachycardia Algorithm
When confronted with a regular narrow-complex tachycardia (QRS < 120 ms, rate > 100 bpm), the differential diagnosis and approach are:
Is the atrial rate faster than the ventricular rate (A > V)? If so, this is likely atrial tachycardia or atrial flutter with variable block. If A = V, proceed to the next step.
Short RP tachycardia (RP < PR): The P wave is close to the preceding QRS. Differential: typical (slow-fast) AVNRT (P wave buried in or just after QRS — pseudo-r' in V1, pseudo-S in inferior leads), orthodromic AVRT (P wave visible in the ST segment, ~70 ms after QRS), and atrial tachycardia with long PR. Long RP tachycardia (RP > PR): The P wave is closer to the next QRS. Differential: atypical (fast-slow) AVNRT, permanent junctional reciprocating tachycardia (PJRT — a form of AVRT using a slowly conducting posteroseptal AP), and atrial tachycardia.
Adenosine Response
| Response to Adenosine | Likely Diagnosis |
|---|---|
| Terminates with a retrograde P wave (terminates in the AV node) | AVNRT or AVRT |
| Reveals flutter waves / atrial activity during AV block | Atrial flutter or atrial tachycardia |
| Terminates with a P wave (terminates in the atrial focus) | Triggered atrial tachycardia (adenosine-sensitive) |
| No effect or transient slowing | Automatic atrial tachycardia, inappropriate sinus tachycardia |
| Transient AV block without termination | Atrial tachycardia or flutter (the arrhythmia continues despite AV block) |
VA Relationship During EP Study
The relationship between ventricular (V) and atrial (A) electrograms on the His catheter during tachycardia is key: VA interval < 70 ms (simultaneous V and A): typical AVNRT. VA interval 70-150 ms: AVRT using a septal accessory pathway, or atypical AVNRT. VA interval > 150 ms: AVRT using a free-wall accessory pathway, or atrial tachycardia. Additional maneuvers: para-Hisian pacing differentiates AVNRT from AVRT; VOP (ventricular overdrive pacing) during tachycardia — "VAV" response suggests AVRT or AVNRT, "VAAV" response suggests atrial tachycardia.
11 Ventricular Tachycardia
Monomorphic VT
Monomorphic VT has a uniform QRS morphology, implying a single activation wavefront or exit site. The most common cause is scar-related reentry — typically from prior myocardial infarction. The reentrant circuit travels through surviving myocardial bundles within or at the border of scar tissue. Other causes of monomorphic VT:
Bundle branch reentrant VT: The reentrant circuit uses the His-Purkinje system (down the RBB, across the septum, up the LBB, then retrograde through the His bundle). Seen in dilated cardiomyopathy with His-Purkinje disease. ECG: LBBB morphology (because the ventricles are activated via the RBB). Treatment: ablation of the RBB (curative).
Idiopathic VT (structurally normal heart): (1) RVOT VT — the most common idiopathic VT; LBBB morphology with inferior axis; triggered activity (cAMP-mediated DADs); responds to adenosine, beta-blockers, verapamil; ablation success > 90%. (2) Fascicular (verapamil-sensitive) VT — reentry involving the left posterior fascicle (most common); RBBB + left axis (superior axis); responds to IV verapamil; ablation targets the Purkinje potential along the midseptum. (3) Papillary muscle VT — from the LV papillary muscles; can be challenging to ablate due to the complex geometry.
Polymorphic VT
Polymorphic VT has a continuously varying QRS morphology. The critical first step is to measure the QT interval on a baseline ECG (before the arrhythmia onset, if available):
QT prolonged — Torsades de pointes: Characteristic "twisting of the points" morphology with a short-long-short initiation sequence. Caused by drug-induced QT prolongation (most common), congenital LQTS, or electrolyte abnormalities (hypokalemia, hypomagnesemia). Treatment: IV magnesium (2 g bolus — first-line even if Mg2+ is normal), isoproterenol (increases heart rate, shortens QT), temporary overdrive pacing at 90-110 bpm (shortens QT and eliminates the pause-dependent initiation). Withdraw offending drugs. Correct K+ to > 4.0 mEq/L and Mg2+ to > 2.0 mEq/L. Do NOT give amiodarone (prolongs QT and worsens torsades).
QT normal — Ischemic or idiopathic polymorphic VT: The most common cause is acute myocardial ischemia — treat with urgent coronary angiography and revascularization. Amiodarone and lidocaine are appropriate antiarrhythmics in this setting. Beta-blockers reduce recurrence. If no ischemia: consider Brugada syndrome, early repolarization syndrome, or idiopathic VF.
Other causes of polymorphic VT: Catecholaminergic polymorphic VT (CPVT) — exercise-induced, structurally normal heart; discussed in Section 13. Short QT syndrome — very rare. Commotio cordis — mechanical trigger on a normal heart.
VT Storm Emergency
VT storm is defined as ≥ 3 sustained VT episodes or appropriate ICD shocks within 24 hours. This is a medical emergency requiring aggressive management:
1. Hemodynamic stabilization: Electrical cardioversion/defibrillation for each episode as needed.
2. IV amiodarone: 150 mg bolus over 10 min, then 1 mg/min for 6 hours, then 0.5 mg/min. Amiodarone is the first-line antiarrhythmic.
3. Deep sedation: Intubation and propofol/midazolam to suppress sympathetic drive.
4. Sympathetic blockade: IV esmolol (beta-blocker), IV lidocaine (suppresses VT trigger). For refractory cases: stellate ganglion block (left-sided) or thoracic epidural — both dramatically reduce sympathetic input to the heart and can break the VT storm cycle.
5. Treat reversible causes: Correct electrolytes (K+ > 4.0, Mg2+ > 2.0), assess for ischemia (catheterization if appropriate), evaluate device malfunction.
6. Catheter ablation: Urgent/emergent VT ablation for refractory VT storm that fails medical therapy. Associated with reduced recurrence and improved survival when performed early.
12 Ventricular Fibrillation & Cardiac Arrest
Ventricular Fibrillation
VF is chaotic, disorganized electrical activity without coordinated ventricular contraction, producing hemodynamic collapse and cardiac arrest. Primary VF occurs in the setting of acute MI (most commonly in the first hour) and is not associated with long-term increased mortality if the patient survives. Secondary VF occurs in the context of chronic heart disease, cardiomyopathy, or channelopathies and carries significant prognostic implications. VF is the most common initial rhythm in out-of-hospital cardiac arrest (~25-35% in witnessed arrests) and the most "treatable" — early defibrillation is the definitive therapy.
ACLS Protocol for VF/Pulseless VT Emergency
1. Begin high-quality CPR (100-120 compressions/min, depth 5-6 cm, full recoil, minimize interruptions). 2. Defibrillation as soon as possible (biphasic 120-200 J, manufacturer-specific, or 200 J default). 3. Resume CPR immediately for 2 minutes after each shock. 4. Epinephrine 1 mg IV/IO every 3-5 minutes. 5. After 2nd shock: amiodarone 300 mg IV push (then 150 mg for subsequent dose), OR lidocaine 1-1.5 mg/kg IV push (then 0.5-0.75 mg/kg). 6. Consider reversible causes — H's and T's: Hypovolemia, Hypoxia, Hydrogen ion (acidosis), Hypo/Hyperkalemia, Hypothermia; Tension pneumothorax, Tamponade, Toxins, Thrombosis (coronary and pulmonary).
Post-Cardiac Arrest Care
Targeted temperature management (TTM): The TTM2 trial (2021) found no benefit of hypothermia at 33°C compared to normothermia (targeting ≤ 37.5°C with active fever prevention) in comatose survivors of out-of-hospital cardiac arrest. Current practice focuses on avoiding hyperthermia (fever > 37.7°C) rather than inducing active hypothermia to 33°C, though some centers still use 33°C or 36°C protocols. Additional post-arrest care: early coronary angiography for STEMI or high suspicion of ACS, hemodynamic support, neuroprognostication (delayed ≥ 72 hours), and assessment for ICD implantation once reversible causes are excluded.
Dankiewicz J et al. N Engl J Med. 2021;384:2283-2294 (TTM2). PMID: 34133859Neuroprognostication After Cardiac Arrest
Neuroprognostication should be delayed at least 72 hours after return of spontaneous circulation (ROSC), and longer if confounders are present (sedation, hypothermia, organ dysfunction). A multimodal approach is recommended — no single test should be used alone to predict poor outcome:
| Modality | Poor Prognostic Sign | Timing |
|---|---|---|
| Clinical exam | Bilateral absence of pupillary and corneal reflexes, myoclonus status epilepticus, GCS motor score 1-2 | ≥ 72h after normothermia |
| Somatosensory evoked potentials (SSEP) | Bilateral absence of N20 cortical response | ≥ 24h (most reliable single predictor) |
| EEG | Suppression, burst-suppression, or status epilepticus | ≥ 72h |
| Brain CT/MRI | Diffuse cerebral edema (loss of gray-white differentiation on CT), diffuse DWI restriction on MRI | CT: 24-72h; MRI: 2-5 days |
| Neuron-specific enolase (NSE) | High values (> 60 μg/L at 48-72h, though thresholds vary by lab) | 48-72h |
AED Programs and Public Access Defibrillation
Automated external defibrillators (AEDs) are critical for out-of-hospital cardiac arrest survival. For every minute without defibrillation, survival from VF decreases by ~7-10%. Public access defibrillation programs (placing AEDs in airports, schools, sports venues, public buildings) have dramatically improved survival. Modern AEDs analyze the rhythm and deliver a shock only for VF or pulseless VT — they are safe for lay rescuers. The combination of bystander CPR and early AED use increases survival from out-of-hospital VF from ~5% to > 50% in many communities.
13 Channelopathies
Long QT Syndrome (LQTS)
A group of inherited (or acquired) disorders characterized by prolonged ventricular repolarization (QTc prolongation) and risk of torsades de pointes and SCD. Prevalence ~1:2,000. Diagnosed when QTc ≥ 480 ms (or ≥ 460 ms with unexplained syncope), or when a pathogenic mutation is identified. The Schwartz score combines ECG findings, clinical history, and family history for diagnosis.
| Type | Gene | Channel | ECG | Triggers | Treatment |
|---|---|---|---|---|---|
| LQT1 | KCNQ1 | IKs (loss of function) | Broad-based T waves | Exercise (especially swimming) | Beta-blockers (most effective in LQT1); avoid competitive sports; ICD for cardiac arrest survivors |
| LQT2 | KCNH2 (hERG) | IKr (loss of function) | Low-amplitude, bifid/notched T waves | Auditory stimuli (alarm clocks), emotional stress, postpartum | Beta-blockers; maintain K+ > 4.0; ICD for high risk; avoid QT-prolonging drugs |
| LQT3 | SCN5A | INa (gain of function) | Long isoelectric ST, narrow peaked T wave | Rest/sleep (bradycardia-dependent) | Mexiletine (Na+ channel blocker — shortens QT); ICD often needed; beta-blockers less effective |
Brugada Syndrome
Brugada syndrome is an inherited channelopathy (most commonly SCN5A mutation, but gene-negative in ~70%) characterized by a distinctive ECG pattern and risk of VF and SCD, typically occurring during rest or sleep. Prevalence: ~1:5,000 to 1:2,000, more common in males and in Southeast Asian populations. The Type 1 (coved) Brugada ECG pattern is required for diagnosis: ≥ 2 mm J-point elevation with coved ST segment and T-wave inversion in ≥ 1 right precordial lead (V1-V2). This pattern may be spontaneous or unmasked by sodium channel blockers (ajmaline, procainamide, flecainide) or fever (a critical trigger — patients must aggressively treat fever with antipyretics).
Risk stratification: Spontaneous Type 1 pattern carries higher risk than drug-induced Type 1 alone. Syncope in the setting of spontaneous Type 1 carries ~5% annual risk of cardiac events. Asymptomatic patients with drug-induced Type 1 only have a much lower risk. The role of programmed ventricular stimulation (EP study) for risk stratification in asymptomatic Brugada patients remains controversial — some centers use VF inducibility to guide ICD decisions, while others do not.
Treatment: ICD for survivors of cardiac arrest (Class I) or with spontaneous Type 1 pattern and syncope (Class IIa). Quinidine (Ito blocker) — used in patients with recurrent ICD shocks, VT/VF storm, or as an alternative when ICD is not feasible (e.g., children, patient refusal). Quinidine reduces the Ito current that is responsible for the Phase 1 notch and the transmural voltage gradient driving the Brugada ECG pattern and arrhythmogenesis. Epicardial ablation of the RVOT substrate is an emerging therapy for refractory cases — targeting the arrhythmogenic epicardial substrate in the RVOT anterior wall (which shows prolonged, fractionated electrograms). Studies by Nademanee and Pappone have demonstrated reduction in VF episodes and even normalization of the ECG pattern after extensive epicardial ablation. Lifestyle modifications: Aggressive treatment of fever (fever can unmask or exacerbate the Brugada pattern), avoid Class IC drugs (flecainide, propafenone), excessive alcohol, cocaine, and large carbohydrate meals. A complete drug avoidance list is maintained at BrugadaDrugs.org.
Catecholaminergic Polymorphic VT (CPVT)
CPVT is caused by mutations in RyR2 (ryanodine receptor, ~60% — autosomal dominant) or CASQ2 (calsequestrin, ~5% — autosomal recessive), leading to abnormal calcium release from the sarcoplasmic reticulum during adrenergic stimulation. Typical presentation: exercise- or emotion-induced syncope or cardiac arrest in children/young adults with a structurally normal heart and normal resting ECG. The hallmark on exercise testing or catecholamine infusion is bidirectional VT (beat-to-beat alternation of the QRS axis) progressing to polymorphic VT and VF.
Treatment: Non-selective beta-blockers (nadolol) — first-line; dramatically reduce events. Flecainide — add-on therapy; directly inhibits RyR2 (the only antiarrhythmic that addresses the molecular defect). ICD for cardiac arrest survivors (but ICD shocks can trigger catecholamine surges that worsen the arrhythmia — program long detection intervals and maximize ATP). Exercise restriction (no competitive sports). Left cardiac sympathetic denervation (LCSD) for refractory cases — removes the left stellate ganglion and T1-T4 sympathetic ganglia, reducing sympathetic input to the heart.
Short QT Syndrome
Short QT syndrome (SQTS) is a rare channelopathy characterized by abnormally short QT interval (QTc < 340 ms) and risk of AF and VF. Caused by gain-of-function mutations in potassium channels (KCNH2, KCNQ1, KCNJ2) or loss-of-function mutations in calcium channels (CACNA1C, CACNB2). ECG shows short QT with tall, peaked T waves. Treatment: ICD for survivors of cardiac arrest; quinidine may prolong the QT interval. Very rare (< 200 cases reported worldwide).
14 Inherited Cardiomyopathies & Arrhythmia
Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC)
ARVC is characterized by fibrofatty replacement of the RV myocardium, predisposing to VT (typically with LBBB morphology — arising from the RV) and SCD, particularly during exercise. Prevalence: ~1:2,000-5,000. Most commonly caused by mutations in desmosomal proteins (PKP2 — plakophilin-2 is the most common). The 2010 Revised Task Force Criteria for diagnosis include:
| Category | Major Criteria | Minor Criteria |
|---|---|---|
| RV dysfunction (echo/MRI) | Regional RV akinesia/dyskinesia + RV dilation (RVOT ≥ 32 mm PLAX) or reduced EF (≤ 40%) | Regional akinesia/dyskinesia + mild RV dilation or mildly reduced EF (40-45%) |
| Tissue characterization | Residual myocytes < 60% with fibrofatty replacement on endomyocardial biopsy | Residual myocytes 60-75% with fibrofatty replacement |
| ECG depolarization | Epsilon waves in V1-V3 | Late potentials on SAECG; TAD ≥ 55 ms in V1-V3 (in absence of RBBB) |
| ECG repolarization | T-wave inversion V1-V3 (age > 14, no RBBB) | T-wave inversion V1-V2 (age > 14, no RBBB) or V4-V6 |
| Arrhythmias | Sustained or non-sustained VT with LBBB morphology and superior axis | Sustained or non-sustained VT with LBBB morphology and inferior axis, or > 500 PVCs/24h |
| Family history | ARVC confirmed in first-degree relative, or pathogenic mutation identified | Suspected ARVC in first-degree relative, or premature SCD (< 35) with suspected ARVC |
Definite ARVC: 2 major, or 1 major + 2 minor, or 4 minor criteria from different categories. Management: Exercise restriction (critical — exercise accelerates disease progression), beta-blockers, antiarrhythmics (sotalol, amiodarone), ICD for SCD prevention, and catheter ablation for recurrent VT (epicardial approach often needed).
Hypertrophic Cardiomyopathy (HCM) — SCD Risk
HCM is the most common inherited cardiomyopathy (~1:500) and the most common cause of SCD in young athletes. Major SCD risk factors in HCM include: prior cardiac arrest or sustained VT, family history of SCD from HCM, unexplained syncope, LV wall thickness ≥ 30 mm, non-sustained VT on Holter, and abnormal blood pressure response to exercise. The AHA/ACC model uses these conventional risk factors to guide ICD decisions (Class I for secondary prevention; Class IIa for ≥ 1 major risk factor). The ESC HCM Risk-SCD calculator provides a 5-year SCD risk estimate incorporating age, LV wall thickness, LA size, LVOT gradient, family history of SCD, NSVT, and unexplained syncope — ICD recommended if 5-year risk ≥ 6%; considered if 4-6%.
Late gadolinium enhancement (LGE) on cardiac MRI quantifies myocardial fibrosis and is an emerging risk marker. LGE ≥ 15% of LV mass is associated with higher SCD risk and may tip the decision toward ICD in borderline cases. Mavacamten (a cardiac myosin inhibitor) reduces LVOT obstruction and is FDA-approved for symptomatic obstructive HCM; its effect on arrhythmia risk is being studied.
Dilated Cardiomyopathy with Arrhythmia
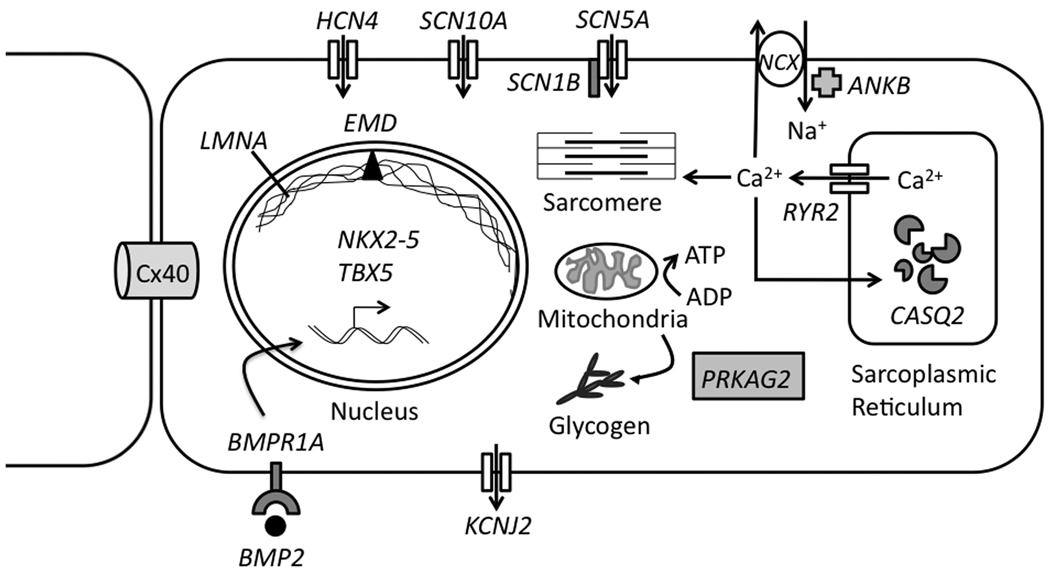
Dilated cardiomyopathy (DCM) is the most common indication for heart transplantation and a major substrate for VT and SCD. Arrhythmias in DCM include: monomorphic VT (scar-related or bundle branch reentry), polymorphic VT/VF, and AF (very common, ~25-50%). Specific genetic subtypes with high arrhythmia risk: LMNA mutations (lamin A/C — high risk of conduction disease and VT, ICD indicated even with LVEF > 35%), SCN5A mutations, FLNC mutations (filamin C), and DES mutations (desmin). Genetic testing is increasingly important for risk stratification in DCM.
Lamin A/C (LMNA) mutations deserve particular attention because they cause an aggressive form of cardiomyopathy with disproportionately high risk of conduction disease (AV block requiring pacing), atrial arrhythmias, and ventricular arrhythmias (VT/VF). Conduction disease often precedes ventricular dysfunction by years. The LMNA risk calculator uses four independent risk factors to predict 5-year SCD risk: non-sustained VT, LVEF < 45%, male sex, and non-missense mutations (insertions, deletions, truncations, splice-site). ICD is recommended when ≥ 2 risk factors are present, regardless of LVEF — this is one of the few conditions where ICD is recommended with LVEF > 35%. All first-degree relatives should undergo genetic screening.
Cardiac Sarcoidosis
Cardiac sarcoidosis is an infiltrative cardiomyopathy that is increasingly recognized as a cause of conduction disease, ventricular arrhythmias, and SCD. It can present with AV block (often in a young patient without traditional risk factors), VT, heart failure, or SCD. Diagnosis: cardiac MRI (patchy LGE in a non-coronary distribution, especially the basal septum) and FDG-PET (active inflammation appearing as focal FDG uptake after 18+ hour fast to suppress normal myocardial glucose uptake). Endomyocardial biopsy has low sensitivity (~25%) due to patchy involvement. Treatment: immunosuppression (corticosteroids first-line), ICD for patients with sustained VT or significant conduction disease, and catheter ablation for refractory VT. Any young patient (< 60) presenting with unexplained AV block (especially Mobitz II or complete heart block) should be evaluated for cardiac sarcoidosis.
15 Sudden Cardiac Death Risk Stratification
Primary Prevention ICD Criteria
Primary prevention ICDs are implanted in patients who have not yet had a sustained VT/VF episode but are at elevated risk:
MADIT II (2002): ICD reduced all-cause mortality by 31% in patients with prior MI and LVEF ≤ 30% (no EP testing required). SCD-HeFT (2005): ICD reduced mortality by 23% in patients with NYHA II-III heart failure and LVEF ≤ 35% (ischemic or non-ischemic). Amiodarone showed no benefit. DANISH (2016): ICD did not significantly reduce all-cause mortality in non-ischemic cardiomyopathy (LVEF ≤ 35%) — though it reduced SCD. This trial raised questions about the universal benefit of primary prevention ICD in non-ischemic DCM, though subgroup analysis suggested benefit in younger patients (< 68 years).
Current Indications (ACC/AHA/HRS 2017 Guidelines)
| Indication | Class |
|---|---|
| Survivors of VF or hemodynamically unstable sustained VT (secondary prevention) | I |
| Ischemic cardiomyopathy, LVEF ≤ 35%, NYHA II-III, ≥ 40 days post-MI, ≥ 90 days post-revascularization, on GDMT | I |
| Ischemic cardiomyopathy, LVEF ≤ 30%, NYHA I, ≥ 40 days post-MI, on GDMT | I |
| Non-ischemic DCM, LVEF ≤ 35%, NYHA II-III, on GDMT for ≥ 3 months | I |
| HCM with ≥ 1 major risk factor for SCD | IIa |
| ARVC with ≥ 1 risk factor (sustained VT, severe RV/LV dysfunction, extensive disease) | IIa |
| LMNA mutation with ≥ 2 risk factors (NSVT, LVEF < 45%, male sex, non-missense mutation) | IIa |
Electrophysiology Testing for SCD Risk
The role of EP testing (programmed ventricular stimulation) for SCD risk stratification has evolved significantly. In the MADIT I era, EPS with VT inducibility was required. MADIT II eliminated the need for EPS, relying solely on LVEF ≤ 30%. Currently, EP testing for risk stratification is used in: (1) post-MI patients with LVEF 30-40% — inducible VT may tip the decision toward ICD (Class IIa); (2) unexplained syncope with structural heart disease — inducible VT confirms a ventricular arrhythmia etiology; (3) Brugada syndrome — the role of EPS is debated (some use VF inducibility for risk stratification in asymptomatic patients with Type 1 pattern); (4) HCM — EPS is not recommended for routine risk stratification; (5) Bifascicular block with syncope — prolonged HV interval indicates infranodal disease (discussed in Section 18).
SCD in Athletes
Sudden cardiac death during sports is rare (~1:50,000 to 1:80,000 per year) but devastating. The most common causes by age group: age < 35: HCM (most common in US), ARVC (most common in northern Italy due to Veneto founder mutations), anomalous coronary arteries, myocarditis, LQTS, WPW, commotio cordis (blunt chest impact causing VF). Age > 35: Atherosclerotic coronary artery disease (overwhelming majority). Pre-participation screening remains controversial: the US approach uses history and physical exam alone; the Italian/ESC approach includes a 12-lead ECG, which has been shown to detect HCM, LQTS, WPW, Brugada, and ARVC. The Seattle Criteria and International Criteria for ECG interpretation in athletes help distinguish physiologic cardiac adaptation from pathologic findings.
Commotio Cordis
Commotio cordis is VF triggered by a non-penetrating blow to the chest (typically a baseball, hockey puck, or lacrosse ball) at a specific vulnerable window of the cardiac cycle (upstroke of the T wave — 15-30 ms window). Occurs in structurally normal hearts, predominantly in young male athletes. Treatment: immediate CPR and defibrillation. Survival has improved from ~15% to ~60% with increased availability of AEDs at athletic events. Prevention: age-appropriate softened baseballs, chest protectors (limited efficacy).
16 Sinus Node Dysfunction
Sick Sinus Syndrome
Sick sinus syndrome (SSS) encompasses a spectrum of SA node dysfunction: sinus bradycardia (inappropriate for physiologic needs), sinus arrest/pauses (failure of impulse generation — pauses > 3 seconds are significant), sinoatrial exit block (impulse generated but fails to conduct to the atrium), and tachy-brady syndrome (alternating tachyarrhythmias, usually AF, with symptomatic bradycardia — the most common form of SSS). SSS is the most common indication for permanent pacemaker implantation (~50% of all PPM implants). Causes: idiopathic fibrosis (aging), ischemia (SA nodal artery disease), infiltrative disease, post-surgical, medications (beta-blockers, CCBs, digoxin, amiodarone).
Chronotropic Incompetence
Chronotropic incompetence is the inability to achieve an adequate heart rate response to exercise. Defined as failure to reach 80% of age-predicted maximum heart rate (220 - age) or a chronotropic index < 0.8 during exercise testing. Clinically significant when it limits functional capacity. Treatment: rate-responsive pacemaker (DDDR or AAIR mode — accelerometer or minute ventilation sensor detects activity and increases pacing rate).
Indications for Pacing in SND
Pacemaker is indicated (Class I) when SND is documented with symptom-rhythm correlation: syncope, presyncope, exercise intolerance, or heart failure attributable to bradycardia. The key is establishing that symptoms are due to the bradycardia — ambulatory monitoring (Holter, event recorder, implantable loop recorder) is essential. Pacing is NOT indicated for asymptomatic sinus bradycardia, even if the rate is < 40 bpm. For tachy-brady syndrome, pacing (to prevent bradycardia) allows use of rate/rhythm control drugs that would otherwise be contraindicated.
Ambulatory Monitoring for Bradycardia
The choice of monitoring device depends on the frequency of symptoms: 24-48 hour Holter monitor — for daily symptoms. External loop recorder (event monitor) — for symptoms occurring weekly; patient-activated with auto-triggered detection for asymptomatic events; 2-4 week monitoring. Mobile cardiac telemetry (MCT) — continuous real-time monitoring for 2-4 weeks; auto-detection of bradycardia and pauses. Implantable loop recorder (ILR) — subcutaneous device (e.g., Medtronic Reveal LINQ, Abbott Confirm Rx) implanted in the left parasternal area; battery life 3-4 years; gold standard for infrequent symptoms (monthly or less). The ILR auto-detects pauses, bradycardia, AF, and tachycardia. The ISSUE-3 trial used ILR to document asystole during vasovagal syncope and guide pacing decisions.
17 Atrioventricular Block
Classification
| Type | ECG Features | Level of Block | Prognosis & Treatment |
|---|---|---|---|
| First degree | PR > 200 ms, all P waves conducted | Usually AV nodal | Benign; no treatment unless markedly prolonged (PR > 300 ms with symptoms — "pacemaker syndrome" from AV dyssynchrony) |
| Second degree — Mobitz I (Wenckebach) | Progressive PR prolongation until a dropped QRS; grouped beating | AV nodal (narrow QRS) | Usually benign; common in athletes, during sleep. Pacing only if symptomatic |
| Second degree — Mobitz II | Constant PR interval with sudden dropped QRS (no preceding PR prolongation) | Infranodal (His-Purkinje) | High risk of progression to complete heart block; Class I indication for pacing even if asymptomatic |
| 2:1 AV block | Every other P wave is not conducted | Cannot differentiate Mobitz I vs II from 2:1 pattern alone | Narrow QRS favors AV nodal (Mobitz I); wide QRS favors infranodal (Mobitz II). Atropine response helps: improved conduction = AV nodal; worsened = infranodal |
| High-grade AV block | ≥ 2 consecutive non-conducted P waves | Usually infranodal | Class I indication for pacing |
| Third degree (complete) | No AV conduction; P waves and QRS are independent (AV dissociation) | Nodal or infranodal | Narrow escape rhythm (40-60 bpm, junctional) = AV nodal. Wide escape rhythm (20-40 bpm) = infranodal (His-Purkinje). Class I pacing indication |
Infranodal vs Nodal Block
Distinguishing AV nodal from infranodal (His-Purkinje) block is critical because infranodal block has a worse prognosis and almost always requires pacing. Clinical clues: AV nodal block: narrow QRS escape rhythm, vagal maneuvers and atropine improve conduction, Wenckebach pattern, often associated with inferior MI. Infranodal block: wide QRS escape rhythm, atropine may worsen block (by increasing atrial rate beyond the capacity of the diseased His-Purkinje system), Mobitz II pattern, associated with anterior MI. EP study definitively localizes the block by measuring AH and HV intervals: prolonged AH = AV nodal block; prolonged HV or infra-Hisian block = infranodal block.
Reversible Causes of AV Block
Before committing to permanent pacing, evaluate and treat reversible causes: medications (beta-blockers, CCBs, digoxin, amiodarone), acute MI (inferior MI commonly causes transient AV block due to AV nodal ischemia — often resolves within 5-7 days; anterior MI causing AV block implies extensive His-Purkinje damage and has worse prognosis), hyperkalemia, Lyme disease (may present with high-degree AV block in young patients — treat with antibiotics, pacing usually temporary), myocarditis, post-cardiac surgery (wait 5-7 days for recovery before implanting PPM), post-TAVR (conduction abnormalities occur in 10-20%, most resolve within 48 hours), and hypothyroidism.
18 Bundle Branch Block & Fascicular Block
Left Bundle Branch Block (LBBB)
LBBB (QRS ≥ 120 ms, broad monophasic R in I, aVL, V5-V6; QS or rS in V1-V2) represents delayed activation of the left ventricle. LBBB is rarely a normal variant — it usually indicates underlying cardiac disease (hypertension, cardiomyopathy, aortic valve disease, CAD). New LBBB in the setting of acute symptoms requires urgent evaluation. LBBB causes electrical dyssynchrony that worsens heart failure — this is the rationale for CRT (discussed in Section 22). The Strauss criteria for strict LBBB (associated with the best CRT response) include: QRS ≥ 140 ms (men) / ≥ 130 ms (women), QS or rS in V1-V2, and mid-QRS notching or slurring in ≥ 2 of I, aVL, V1, V2, V5, V6.
Right Bundle Branch Block (RBBB)
RBBB (QRS ≥ 120 ms, rsR' in V1-V2, wide S in I and V6) represents delayed RV activation. RBBB can be a normal variant in healthy individuals. Pathologic causes: RV strain (PE, pulmonary hypertension), ASD, RV cardiomyopathy, and conduction system disease. Isolated RBBB has a good prognosis and does not require pacing.
Fascicular Blocks
Left anterior fascicular block (LAFB): Left axis deviation (-45° to -90°), qR in I and aVL, rS in II, III, aVF, normal QRS duration. The most common fascicular block (the LAF is thin and has a single blood supply). Left posterior fascicular block (LPFB): Right axis deviation (> +90°), rS in I, qR in III — must exclude other causes of RAD (RVH, lateral MI, PE). LPFB is uncommon because the LPF is thick with dual blood supply.
Bifascicular and Trifascicular Block
Bifascicular block: RBBB + LAFB (most common), or RBBB + LPFB. Indicates disease in two of three fascicles. Trifascicular block: Bifascicular block + first-degree AV block (implies slow conduction through the remaining fascicle). Alternating bundle branch block (RBBB alternating with LBBB on ECG) is the strongest indicator of trifascicular disease and is a Class I indication for pacing even without symptoms. Bifascicular block with syncope warrants EP study to measure the HV interval — HV ≥ 70 ms or infra-Hisian block during atrial pacing is an indication for pacing.
Rate-Related BBB (Aberrant Conduction)
Rate-related (functional) BBB occurs when one bundle branch has a longer refractory period than the other. At a critical heart rate, the impulse arrives during the refractory period of that bundle, producing a BBB pattern. This is the mechanism of Ashman phenomenon: a wide QRS complex follows a short-long-short cycle length sequence (the long cycle lengthens the refractory period of the bundle branch, and the subsequent short cycle finds the bundle still refractory). This is most commonly seen in AF — a wide QRS beat following a long R-R interval then a short R-R interval is likely aberrant conduction, not a PVC. Distinguishing features of aberrancy: typical BBB morphology (usually RBBB), Ashman phenomenon, identical initial QRS forces to normal beats.
19 Neurocardiogenic Syncope
Vasovagal Syncope
Vasovagal syncope (VVS) is the most common cause of syncope (~60% of cases). It results from an inappropriate reflex causing vasodilation (hypotension) and/or bradycardia (cardioinhibition) in response to a trigger (prolonged standing, emotional stress, pain, venipuncture). The Bezold-Jarisch reflex is the proposed mechanism: vigorous contraction of an underfilled ventricle activates mechanoreceptors, triggering paradoxical vagal activation. Classification: cardioinhibitory (predominant bradycardia/asystole), vasodepressor (predominant hypotension), or mixed. Treatment: patient education, physical counterpressure maneuvers, adequate hydration and salt intake, avoiding triggers. Medications: midodrine (alpha-agonist), fludrocortisone (volume expansion). Pacemaker: Considered for recurrent cardioinhibitory VVS with documented asystole > 3 seconds during syncope (captured on implantable loop recorder) in patients > 40 years — Class IIb recommendation; the BIOSync CLS trial showed benefit of dual-chamber pacing with closed-loop stimulation.
Carotid Sinus Hypersensitivity
Carotid sinus hypersensitivity (CSH) causes syncope when the carotid sinus is stimulated (head turning, tight collars, shaving). Most common in elderly males. Diagnosed by carotid sinus massage (CSM) — performed with the patient supine and then upright (more sensitive), with continuous ECG and blood pressure monitoring. A positive response is defined as:
| Type | Response to CSM | Treatment |
|---|---|---|
| Cardioinhibitory (most common, ~70%) | Asystole or ventricular pause > 3 seconds | Dual-chamber pacemaker (DDD) — Class I if associated with syncope |
| Vasodepressor (~10%) | Drop in SBP > 50 mmHg without significant bradycardia | Midodrine, fludrocortisone, physical counterpressure maneuvers. Pacing does not help |
| Mixed (~20%) | Both significant bradycardia and hypotension | Pacing addresses the cardioinhibitory component; additional medical therapy for the vasodepressor component |
Contraindications to CSM: Carotid bruit, known carotid stenosis > 70%, recent stroke/TIA (< 3 months), history of VT/VF. Complications of CSM are rare (< 0.5%) but include stroke (from carotid plaque embolization).
Tilt Table Testing
The head-up tilt table test (HUTT) is used to evaluate suspected neurocardiogenic syncope. The patient is tilted to 60-70° for 20-45 minutes with continuous blood pressure and heart rate monitoring. A positive test reproduces the patient's typical symptoms with hypotension and/or bradycardia. Isoproterenol or sublingual nitroglycerin may be used as provocative agents. Sensitivity is ~60-70% with specificity ~85-90%. The HUTT is most useful when the diagnosis is uncertain and symptom-rhythm correlation cannot be obtained by monitoring.
Syncope Evaluation Algorithm
Step 1 — Initial evaluation: History (triggers, prodrome, position, recovery time, witnesses), physical exam (orthostatic vitals — measure at 0, 1, and 3 minutes standing; carotid sinus massage if > 40 years; cardiac exam), 12-lead ECG. This initial evaluation establishes the diagnosis in ~50% of patients.
Step 2 — Risk stratification: High-risk features requiring admission and urgent workup: abnormal ECG (BBB, long QT, Brugada, pre-excitation, Q waves), structural heart disease, syncope during exertion, family history of SCD, severe injury from syncope. Low-risk: typical vasovagal features (prodrome, triggers, young age, normal heart).
Step 3 — Cardiac evaluation (if indicated): Echocardiography (structural heart disease, LV function), ambulatory monitoring (Holter, event recorder, or ILR based on frequency of events), exercise testing (exertional syncope), and EP study (suspected arrhythmic syncope with structural heart disease).
Step 4 — Autonomic evaluation (if cardiac workup negative): Tilt table testing, autonomic reflex testing, carotid sinus massage.
20 Permanent Pacemakers
Indications (ACC/AHA/HRS Guidelines)
Class I indications for permanent pacing include: symptomatic sinus bradycardia (symptom-rhythm correlation), Mobitz II second-degree AV block, high-grade AV block, third-degree (complete) AV block, alternating BBB, symptomatic chronotropic incompetence, and post-ablation AV block. The critical requirement is that bradycardia is documented and correlated with symptoms (except for Mobitz II and complete heart block, where pacing is indicated regardless of symptoms).
Pacing Modes — NBG Code
| Position | Meaning | Options |
|---|---|---|
| I — Chamber paced | Which chamber receives pacing impulses | A (atrium), V (ventricle), D (dual — both) |
| II — Chamber sensed | Which chamber is monitored for intrinsic activity | A, V, D, O (none) |
| III — Response to sensing | What the device does when it senses | I (inhibit), T (trigger), D (dual — inhibit + trigger), O (none) |
| IV — Rate modulation | Rate-responsive capability | R (rate-responsive), O (none) |
AAI/AAIR: Single-lead atrial pacing. Paces and senses the atrium. Used for isolated SND with intact AV conduction. Rarely used alone due to risk of future AV block. VVI/VVIR: Single-lead ventricular pacing. Paces and senses the ventricle. Used for chronic AF with slow ventricular response. DDD/DDDR: Dual-chamber pacing. The most versatile and commonly used mode. Paces and senses both chambers. Maintains AV synchrony. Tracks atrial activity and delivers ventricular pacing after a programmed AV delay (mimics normal conduction). VDD: Single-lead with atrial sensing and ventricular pacing — tracks native atrial rhythm and paces the ventricle (used for AV block with normal sinus node function).
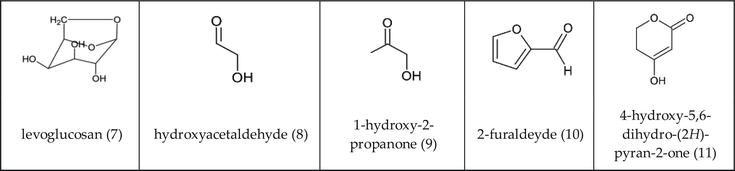
Battery and Longevity Considerations
Modern pacemaker batteries (lithium iodide) last 8-15 years depending on pacing burden, output settings, and features used. ICD batteries are larger but last only 5-10 years due to the high energy required for charging the defibrillation capacitor. Battery status is monitored at each follow-up: BOL (beginning of life), ERI (elective replacement indicator) — indicates approximately 3-6 months of remaining battery life, triggering planning for generator change, and EOL (end of life) — minimal remaining energy, urgent replacement needed. At generator change, the existing leads are tested; if functioning normally, they are connected to a new generator. Lead replacement is only performed if the existing leads are malfunctioning.
Conduction System Pacing
His bundle pacing (HBP): The lead is placed directly on the His bundle, capturing the native conduction system and producing a narrow QRS (physiologic activation). Advantages: avoids pacing-induced dyssynchrony. Limitations: higher capture thresholds (shorter battery life), technically challenging lead placement, sensing issues, and risk of lead dislodgement.
Left bundle branch area pacing (LBBAP): The lead is screwed deep into the interventricular septum from the RV side to capture the left bundle branch or its fascicles. Produces a narrow(er) QRS with physiologic LV activation. LBBAP has rapidly gained popularity as it offers advantages over HBP: lower and more stable thresholds, easier implantation, better sensing parameters, and similar physiologic activation. Confirmation of LBB capture is made by observing: (1) a paced RBBB morphology in V1 (indicating LBB capture with delayed RBB activation), (2) short stimulus-to-LVAT (left ventricular activation time) of ≤ 75-80 ms, and (3) selective vs non-selective LBB capture (selective = pure LBB capture with an isoelectric interval after the stimulus; non-selective = LBB + septal myocardial capture with no isoelectric interval). LBBAP is emerging as an alternative to both traditional RV pacing and CRT in appropriate patients, with mounting evidence from observational studies and registries.
Leadless Pacemakers
Pacemaker Implantation Technique
Standard transvenous pacemaker implantation involves: (1) vascular access via the subclavian vein (subclavian puncture or cephalic vein cutdown — the cephalic approach has lower pneumothorax risk), (2) lead advancement under fluoroscopy to the right atrium (RA appendage) and/or right ventricle (RV apex or RV septum), (3) lead fixation (active fixation with a screw-in helix or passive fixation with tines), (4) testing of pacing thresholds (target < 1.0V at 0.5 ms), sensing amplitudes (P wave > 1.5 mV, R wave > 5 mV), and lead impedance (400-1500 ohms), (5) connection of leads to the pulse generator, and (6) creation of a subcutaneous or submuscular pocket in the pectoral region. Post-procedure: CXR to confirm lead position and exclude pneumothorax; wound care; arm movement restriction for 4-6 weeks (to prevent lead dislodgement); device check before discharge and at 2-4 weeks.
Micra (Medtronic): A self-contained, leadless pacemaker implanted directly in the RV via femoral venous access. Available as VVI (Micra VR) and recently AV synchronous (Micra AV — uses an accelerometer to sense atrial contraction and provide AV-synchronous pacing). Advantages: no pocket, no lead (eliminates lead-related complications and pocket infection). Limitations: single-chamber (VVI) or limited AV synchrony, cannot provide atrial pacing, limited battery life (~12 years), and extraction is challenging (usually left in place with a new device implanted adjacent). Indicated for patients with AF and bradycardia, or those at high risk for lead/pocket complications (hemodialysis patients, prior device infection).
21 Implantable Cardioverter-Defibrillators (ICDs)
Primary vs Secondary Prevention
Secondary prevention: ICD for survivors of cardiac arrest due to VF or hemodynamically significant VT, or sustained VT with structural heart disease (Class I). Primary prevention: ICD for patients at elevated risk of SCD who have not yet had an event — see Section 15 for detailed criteria (MADIT II, SCD-HeFT). The primary prevention population accounts for the majority of ICD implants.
ICD Programming
Modern ICDs have tiered therapy:
Detection zones: Typically a VT zone (e.g., 150-200 bpm), a fast VT zone (200-250 bpm), and a VF zone (> 250 bpm). Detection requires a sustained number of intervals meeting the rate criterion (e.g., 30 of 40 intervals). SVT discriminators are programmed in the VT zone to differentiate SVT from VT: these include onset criteria (sudden vs gradual rate acceleration), stability (regular vs irregular RR intervals — AF is irregular), morphology analysis (comparing the EGM to stored sinus rhythm templates), and V > A criteria (more ventricular than atrial events suggests VT).
Anti-tachycardia pacing (ATP): Burst pacing slightly faster (~85-90% of VT cycle length) than the VT rate to penetrate and terminate the reentrant circuit — effective for monomorphic VT in ~80-90% of episodes and is painless for the patient. ATP is tried first in the VT zone and sometimes the fast VT zone before shock delivery. Ramp pacing (progressively faster pacing) is an alternative to burst pacing.
Shock therapy: High-energy defibrillation (up to 35-40 J biphasic) for VF or VT that fails ATP. At least 10 J safety margin above the defibrillation threshold (DFT) is desired (though routine DFT testing at implant is no longer required per the SIMPLE trial). ICD shocks are painful and associated with decreased quality of life and increased anxiety/depression — phantom shocks (perception of shock when none was delivered) are also common and require psychological support.
Contemporary programming trends: Higher rate detection cutoffs (e.g., ≥ 200 bpm for single zone), longer detection durations (30/40 intervals), and aggressive use of ATP before shocks — all shown to reduce inappropriate and unnecessary shocks without compromising safety (MADIT-RIT, ADVANCE III, PROVIDE trials).
Subcutaneous ICD (S-ICD)
The S-ICD (Boston Scientific) has no transvenous leads — the pulse generator is placed in the left lateral chest wall, and a subcutaneous lead runs along the sternum. Advantages: eliminates all lead-related intravascular complications (infection, endocarditis, vascular obstruction, lead failure). Limitations: cannot provide anti-tachycardia pacing (ATP), cannot provide bradycardia pacing (except brief post-shock pacing), and cannot deliver CRT. Therefore, S-ICD is appropriate for patients who do not need pacing and have VT likely to respond to defibrillation. Pre-implant ECG screening is required to ensure appropriate sensing. The PRAETORIAN trial demonstrated non-inferiority of S-ICD compared to transvenous ICD.
Knops RE et al. N Engl J Med. 2020;383:526-536 (PRAETORIAN). PMID: 32757521Wearable Cardioverter-Defibrillator (WCD)
The LifeVest (Zoll) is a wearable external defibrillator vest used as a bridge to ICD implantation or recovery. Indications: post-MI with LVEF ≤ 35% (within the 40-day waiting period), newly diagnosed cardiomyopathy on GDMT (within the 3-month optimization period), post-ICD explant for infection. The VEST trial did not show a significant reduction in the primary endpoint of arrhythmic death, though compliance was suboptimal. The WCD provides real-time monitoring and defibrillation capability and is commonly used in clinical practice despite the ambiguous trial data.
22 Cardiac Resynchronization Therapy (CRT)
Indications
Class I indication: LVEF ≤ 35%, NYHA II-IV (ambulatory) on GDMT, sinus rhythm, LBBB with QRS ≥ 150 ms. This group has the strongest evidence and highest response rate (~70%). Class IIa: LBBB with QRS 120-149 ms; non-LBBB with QRS ≥ 150 ms. Class IIb: Non-LBBB with QRS 120-149 ms (weakest evidence — low response rate). Key trials: COMPANION (CRT-D reduced all-cause mortality in NYHA III-IV HF), MADIT-CRT (CRT-D reduced HF events in NYHA I-II with LBBB), RAFT (CRT-D reduced mortality and HF hospitalization in NYHA II-III).
Bristow MR et al. N Engl J Med. 2004;350:2140-2150 (COMPANION). PMID: 15152059CRT-P vs CRT-D
CRT-P: Biventricular pacemaker without defibrillation capability. Appropriate for patients whose primary indication is heart failure symptom improvement (e.g., older patients, NYHA IV, limited life expectancy). CRT-D: Biventricular pacemaker with ICD capability. Preferred for patients who also meet ICD criteria. The decision between CRT-P and CRT-D should consider overall prognosis, patient preferences, and comorbidities.
LV Lead Placement & CS Anatomy
The LV lead is placed via the coronary sinus into a tributary vein to pace the LV epicardium. Target position: lateral or posterolateral LV wall, at a site of maximal electrical delay (latest activation). The CS is accessed from the right atrium through the CS ostium. Key tributaries: great cardiac vein (anterior interventricular groove — too anterior, usually not targeted), lateral vein (lateral wall — ideal target), posterolateral vein (posterolateral wall — good target), and middle cardiac vein (posterior interventricular groove — pacing here can worsen dyssynchrony). CS venography is performed to map the venous anatomy before lead placement.
CRT Response Predictors & Non-Responder Management
Predictors of CRT response: strict LBBB morphology (strongest predictor), QRS ≥ 150 ms, female sex, non-ischemic etiology, presence of viable myocardium at the LV lead site, absence of scar at the pacing site (scar reduces response). Approximately 30% of CRT patients are "non-responders." A systematic evaluation of non-response should include:
1. Confirm biventricular pacing > 98%: Check device interrogation. Common causes of reduced BiV pacing: frequent PVCs (consider ablation if PVC burden > 10%), AF with rapid ventricular response (may need AV node ablation), and loss of LV capture (elevated thresholds).
2. Optimize AV delay: Echocardiographic AV optimization (iterative method) or empiric algorithms (e.g., SmartDelay).
3. Optimize VV delay: Adjust the timing offset between RV and LV pacing to achieve the narrowest QRS or best hemodynamics.
4. Assess LV lead position: Is the lead in a lateral/posterolateral vein? Is there phrenic nerve stimulation limiting output? Consider lead repositioning.
5. Consider upgrade to conduction system pacing: His bundle pacing or LBBAP combined with LV pacing (His-optimized CRT, LOT-CRT) is an emerging strategy for CRT non-responders.
6. Ensure optimal medical therapy: GDMT (ACEi/ARB/ARNI, beta-blocker, MRA, SGLT2 inhibitor) must be maximized alongside device therapy.
AV Node Ablation and Pacing ("Ablate and Pace")
For patients with permanent AF and uncontrolled ventricular rate despite maximal medical therapy, AV node ablation with permanent pacing is a definitive rate control strategy. The AV node is ablated (creating complete heart block), and the patient depends entirely on pacing. This should be combined with BiV pacing (CRT) or LBBAP to avoid pacing-induced cardiomyopathy from chronic RV pacing. The APAF-CRT trial demonstrated that AV node ablation with CRT was superior to pharmacologic rate control in patients with permanent AF and narrow QRS who had been hospitalized for HF.
23 Lead Management & Extraction
Lead Complications
Lead dislodgement: Most common early complication (~2-3% of leads). Presents with failure to pace/sense, diaphragmatic stimulation (RV lead near the diaphragm), or intercostal muscle stimulation. Diagnosed by CXR (lead tip position change) and device interrogation (elevated thresholds, abnormal sensing). Lead perforation: Acute (within days) or late (chronic). Can cause pericardial effusion or tamponade. Risk factors: thin RV wall, steroid use, active fixation leads. Lead fracture: Break in the lead conductor, causing intermittent or complete loss of function. Often occurs at stress points (subclavian crush between the clavicle and first rib). Diagnosed by impedance changes (high impedance = conductor fracture, low impedance = insulation failure) and fluoroscopy. Insulation failure: Breakdown of the lead insulation material — causes inappropriate sensing (noise), oversensing, and potential inappropriate shocks (in ICDs). Twiddler syndrome: Conscious or unconscious manipulation of the pulse generator by the patient, causing lead dislodgement by coiling around the generator.
Lead Extraction
Indications: Device infection (the most common indication — mandatory extraction for CIED-associated endocarditis), lead malfunction when the lead poses a safety risk (e.g., ICD lead with inappropriate shocks from noise), venous occlusion preventing new lead implantation, and abandoned lead burden (relative indication).
Techniques: Simple traction: Direct pull on the lead — only effective for recently implanted leads (< 1 year) before significant fibrotic adhesions form. Laser sheath extraction: A powered sheath with excimer laser at the tip that vaporizes fibrous tissue binding the lead to the vein/myocardium. Mechanical (rotational) sheath: A sheath with a mechanical cutting tip. Femoral approach: Snare-based retrieval from below when superior approaches fail. Lead extraction carries significant risks (cardiac avulsion, SVC tear, tamponade — mortality ~0.2-0.5%) and should be performed in experienced centers with cardiac surgery backup.
24 Device Complications
Device Infection
Pocket infection: Presents with erythema, warmth, swelling, pain, and/or erosion of the generator through the skin. Most commonly caused by Staphylococcus aureus or coagulase-negative staphylococci (S. epidermidis). CIED-associated infective endocarditis (CIED-IE): Lead vegetations (diagnosed by TEE — sensitivity ~90%), bacteremia, and systemic infection. All CIED infections (pocket or systemic) require complete device and lead extraction plus prolonged antibiotic therapy (4-6 weeks for endocarditis). Infection rates: ~1-2% for initial implants, higher for generator changes and system revisions. Prevention: antibiotic prophylaxis (cefazolin IV pre-procedure), TYRX antibacterial envelope (mesh impregnated with minocycline and rifampin — WRAP-IT trial showed reduction in infections in high-risk patients).
Tarakji KG et al. N Engl J Med. 2019;380:1895-1905 (WRAP-IT). PMID: 30786013Inappropriate Shocks
ICD shocks delivered for rhythms other than VT/VF. Causes include: AF with rapid ventricular response (most common cause), sinus tachycardia, SVT, T-wave oversensing (the ICD counts the T wave as a QRS, "double-counting" the rate), lead noise (from fracture or insulation failure), and electromagnetic interference. Management: reprogram detection criteria (higher rate cutoffs, longer detection, discriminators for SVT), treat the underlying cause (rate control for AF, lead revision for fracture), and beta-blockers. Inappropriate shocks are associated with increased mortality and severe psychological distress.
Electromagnetic Interference (EMI)
MRI: MRI-conditional devices are now standard — patients can safely undergo MRI if device and leads meet the conditions-of-use (1.5T or 3T depending on system, specific programming before and after scan). Non-MRI-conditional devices require careful risk-benefit assessment. Surgical electrocautery: Use bipolar cautery when possible; if monopolar is needed, keep the grounding pad away from the device, use short bursts, and program the ICD to monitor-only mode (disable tachycardia therapies) during surgery. Other sources: Theft detectors (walk through at normal pace, do not linger), induction cooktops (may affect pacemakers if directly over the device), industrial welding equipment, and strong magnets.
Magnet Behavior
Pacemaker + magnet: Activates asynchronous pacing (DOO or VOO) — useful if oversensing causes inappropriate inhibition. The magnet rate may indicate battery status (e.g., change from 85 bpm to 65 bpm indicates elective replacement). ICD + magnet: Disables tachycardia detection and therapy (no shocks) but does NOT affect bradycardia pacing. Used during surgery to prevent inappropriate shocks from electrocautery. Important: Remove the magnet as soon as it is no longer needed — the ICD will not protect the patient from VT/VF while the magnet is applied.
Perioperative Device Management
Systematic approach for patients with CIEDs undergoing surgery:
| Step | Action |
|---|---|
| Pre-procedure | Identify device type (pacemaker vs ICD vs CRT), manufacturer, and indication. Determine if patient is pacemaker-dependent (no underlying rhythm when pacing is inhibited). Review procedure type and location of electrocautery relative to the device |
| Pacemaker-dependent + surgery above umbilicus | Reprogram to asynchronous mode (DOO/VOO) or place magnet over device during electrocautery. Have backup transcutaneous pacing available |
| ICD + any surgery with electrocautery | Disable tachycardia therapies (reprogram or magnet). Have external defibrillator immediately available. Use bipolar cautery when possible; if monopolar, place grounding pad on contralateral side, away from device |
| Post-procedure | Interrogate device after surgery. Restore all pre-procedure settings. Verify sensing, pacing thresholds, and lead impedances |
End-of-Life Device Management
Deactivation of ICDs at end of life is an important ethical and clinical consideration. ICD shocks in a dying patient cause unnecessary suffering. All major medical societies (HRS, ACC, AHA) affirm that device deactivation at end of life is ethically appropriate and does not constitute euthanasia or assisted suicide. The decision should be discussed proactively during advance care planning. Deactivation can be performed at the bedside by placing a magnet (for ICD) or reprogramming. Pacemaker deactivation in pacemaker-dependent patients requires more nuanced discussion, as it will lead to death from bradycardia — some ethicists consider this equivalent to withdrawing life-sustaining therapy.
25 Remote Monitoring
Alert-Based Follow-Up
Remote monitoring (RM) allows CIEDs to transmit data wirelessly to a secure server for review by the EP team, without the patient visiting the clinic. All major manufacturers have RM platforms (Medtronic CareLink, Abbott Merlin.net, Boston Scientific Latitude, Biotronik Home Monitoring). Standard RM includes: scheduled transmissions (every 1-3 months) and alert transmissions (triggered by specific events).
Clinically important alerts: VT/VF episodes and therapy delivery (shocks, ATP), AF detection and burden (duration, rate), lead impedance out of range (suggests fracture or insulation failure), pacing threshold elevation (suggests dislodgement or exit block), battery voltage at ERI (elective replacement indicator), and device reset events.
The HRS Expert Consensus (2015) and ESC Guidelines recommend RM for all CIED patients as a Class I recommendation. RM has been shown to detect actionable events earlier than in-office follow-up alone (TRUST, CONNECT, ECOST trials), reduce inappropriate shocks through early reprogramming, detect lead failure earlier, reduce time to clinical decision, and is associated with improved survival in some analyses. In-office visits are still recommended at least annually (or when remote findings require hands-on evaluation).
Device-Based Diagnostics
Beyond arrhythmia detection, modern CIEDs provide additional diagnostic data that aids clinical management:
| Diagnostic | Clinical Utility |
|---|---|
| Intrathoracic impedance (OptiVol, Medtronic) | Decreased impedance correlates with increasing pulmonary fluid — early warning of HF decompensation. The PARTNERS HF trial showed that combined impedance and device diagnostics predicted HF hospitalization. However, specificity is limited (false alerts common) |
| Heart rate variability (HRV) | Decreased HRV is a marker of autonomic dysfunction and predicts adverse outcomes in HF. Low HRV trends may prompt medication adjustment |
| Patient activity levels | Built-in accelerometers track daily activity; declining activity levels may indicate worsening HF or overall clinical decline |
| Nocturnal heart rate | Rising nocturnal HR may signal worsening HF, infection, or anemia |
| Percent ventricular pacing | High RV pacing burden (> 40%) may cause pacing-induced cardiomyopathy — should trigger consideration for upgrade to CRT or conduction system pacing |
| Stored EGMs | Intracardiac electrograms stored during detected episodes allow review of arrhythmia morphology and classification (appropriate vs inappropriate therapy) |
AF Burden Detection
CIEDs with atrial leads can detect and quantify AF episodes (AF burden = total duration of AF over a monitoring period). Device-detected atrial high-rate episodes (AHRE) are common and carry an increased stroke risk — though lower than clinical AF. The NOAH-AFNET 6 trial (2023) found that edoxaban did not significantly reduce the composite of stroke, systemic embolism, or cardiovascular death compared to placebo in patients with AHRE detected by devices but without clinical AF. The ARTESiA trial (2023) showed apixaban reduced stroke risk compared to aspirin in patients with subclinical AF detected by devices, but increased major bleeding. Clinical management of device-detected AF remains an evolving area.
Kirchhof P et al. N Engl J Med. 2023;389:1167-1179 (NOAH-AFNET 6). PMID: 37622677 Healey JS et al. N Engl J Med. 2024;390:107-117 (ARTESiA). PMID: 3762267626 Ablation Technology
Radiofrequency (RF) Ablation
RF ablation uses alternating current (300-750 kHz) delivered via a catheter electrode to create thermal lesions in the myocardium. Tissue is heated to > 50°C to cause irreversible cell death. Modern RF ablation uses irrigated-tip catheters — saline irrigation through the catheter tip cools the electrode-tissue interface, preventing char and thrombus formation while allowing deeper lesion formation. Contact force (CF) sensing: Catheters equipped with CF sensors (TactiCath, ThermoCool SmartTouch) measure the force applied to tissue, allowing the operator to optimize lesion quality. The TOCCASTAR trial showed that CF-guided ablation improved 12-month freedom from AF compared to standard ablation. The EFFICAS studies showed that a force-time integral (FTI) > 400 g·s correlated with durable PV isolation. Target CF: 10-20 grams for most applications.
Cryoablation
Cryoablation uses extreme cold (nitrous oxide refrigerant, reaching -40°C to -80°C) to create lesions. The cryoballoon (Arctic Front Advance, Medtronic) is designed specifically for PVI — the balloon is inflated at the PV ostium to achieve circumferential occlusion and ablation in a single application. The FIRE AND ICE trial demonstrated non-inferiority of cryoballoon PVI compared to RF point-by-point PVI for paroxysmal AF. Advantages of cryoablation: single-shot PVI (faster procedure), reproducible lesions, lower risk of esophageal injury (compared to posterior wall RF). Disadvantage: phrenic nerve injury (~2-5% with right superior PV ablation — monitor diaphragmatic excursion during ablation by pacing the phrenic nerve).
Kuck KH et al. N Engl J Med. 2016;374:2235-2245 (FIRE AND ICE). PMID: 27554722Pulsed Field Ablation (PFA)
Pulsed field ablation is the newest ablation energy source, using high-voltage, ultra-short electrical pulses to cause irreversible electroporation — creating nanopores in cell membranes that lead to cell death. The critical advantage of PFA is tissue selectivity: cardiac myocytes are preferentially destroyed while adjacent tissues (esophagus, phrenic nerve, coronary arteries) are relatively spared. This dramatically reduces the risk of collateral damage, including the feared atrial-esophageal fistula. The ADVENT trial (2023) demonstrated non-inferiority of PFA to conventional thermal ablation for drug-refractory paroxysmal AF, with a superior safety profile. The MANIFEST-PF registry confirmed low complication rates across large, real-world cohorts. PFA is rapidly becoming the preferred energy source for AF ablation at many centers.
Reddy VY et al. N Engl J Med. 2023;389:1660-1671 (ADVENT). PMID: 376341483D Mapping Systems
CARTO (Biosense Webster): Uses magnetic field technology (a magnetic emitter under the patient table and sensors in the catheter tip) to create 3D geometry and activation maps. Also incorporates impedance-based location. EnSite (Abbott): Uses impedance-based localization (electrical field between skin patches) and optional magnetic hybrid. Both systems allow: creation of detailed 3D chamber geometry, activation mapping (color-coded propagation), voltage mapping (identifying scar — low voltage < 0.5 mV in ventricle, < 0.5 mV in atrium), lesion tagging (marking ablation points), and integration with pre-acquired CT or MRI images.
Substrate Mapping
Voltage mapping identifies scar (dense scar < 0.5 mV bipolar), border zone (0.5-1.5 mV), and normal tissue (> 1.5 mV) in the ventricle. Local abnormal ventricular activities (LAVA): Fractionated, late, or split electrograms within or at the border of scar that represent surviving myocardial bundles — potential channels for VT circuits. Elimination of all LAVA is a substrate-based ablation strategy (LAVA elimination approach). Scar homogenization: Ablation of all abnormal electrograms within the scar to eliminate potential VT substrates, regardless of whether VT is inducible — a more aggressive approach used in VT storm and recurrent VT.
Ultra-High-Density Mapping
Modern mapping catheters with multiple mini-electrodes provide dramatically higher resolution than traditional mapping. Pentaray (Biosense Webster): 5-spline, 20-electrode catheter for rapid high-density activation and voltage mapping. Orion/HD Grid (Boston Scientific): 8-spline, 64-electrode basket catheter that provides omnipolar (direction-independent) mapping. Octaray (Biosense Webster): 8-spline, 48-electrode catheter for ultra-high-density mapping. These catheters can acquire thousands of electrogram points within minutes, creating detailed maps of activation, voltage, and scar that were previously impossible. Ultra-high-density mapping has improved the ability to identify conducting channels, isthmuses, and critical sites for ablation, particularly in complex scar-related arrhythmias.
Stereotactic Body Radiation Therapy (SBRT) for VT
Cardiac SBRT (radioablation) is an emerging non-invasive therapy for refractory VT. A single fraction of 25 Gy radiation is delivered to the VT substrate (identified by pre-procedure mapping and cardiac imaging) using a linear accelerator, similar to the technique used for stereotactic radiosurgery of brain tumors. The ENCORE-VT trial and other studies have shown promising results in patients with refractory VT who have failed catheter ablation. This technology is still investigational and limited to specialized centers, but represents a paradigm shift — the possibility of treating cardiac arrhythmias entirely non-invasively.
27 AF Ablation — Detailed
PVI Strategies
The goal of PVI is to electrically disconnect all four pulmonary veins from the left atrium. Wide-area circumferential ablation (WACA): RF lesions delivered 1-2 cm outside the PV ostia in a circumferential fashion, typically isolating the PVs in ipsilateral pairs (left superior + left inferior; right superior + right inferior). This creates a larger area of isolation and reduces PV stenosis risk (compared to ostial ablation). Cryoballoon PVI: The balloon is positioned at each PV ostium, inflated, and cooled for 180-240 seconds per vein. Optimal occlusion (confirmed by contrast injection showing complete occlusion — "leaks" reduce efficacy) is critical. PFA: Pentaspline or focal catheter designs deliver pulsed fields at each PV, achieving rapid isolation.
Additional Targets Beyond PVI
For persistent AF, PVI alone may be insufficient. Additional targets include: posterior wall isolation (the posterior LA wall has embryologic connection to the PVs and harbors AF-sustaining substrates), CTI ablation (to prevent typical flutter, which commonly coexists with AF), mitral isthmus line (from the left inferior PV to the mitral annulus — to prevent perimitral flutter), LA roof line (connecting left and right superior PVs), SVC isolation (the SVC can harbor AF triggers), and left atrial appendage (LAA) isolation (last resort — requires ongoing anticoagulation). However, the STAR AF II trial found that adding CFAE ablation or linear lesions to PVI did not improve outcomes for persistent AF. The optimal strategy beyond PVI remains debated.
First vs Repeat Procedures
Freedom from AF at 1 year after a first PVI ablation is approximately 60-80% for paroxysmal AF and 40-60% for persistent AF (off antiarrhythmic drugs). PV reconnection is the dominant cause of recurrence (~70% of repeat procedures show reconnection of previously isolated veins). Repeat ablation addresses PV reconnection and any additional substrates identified. After two procedures, long-term freedom from AF exceeds 80% for paroxysmal AF. A 3-month "blanking period" after ablation is standard — early arrhythmia recurrences during this period do not necessarily indicate procedural failure (inflammatory response, transient irritability).
Periprocedural Anticoagulation for AF Ablation
Current guidelines recommend uninterrupted anticoagulation throughout the AF ablation procedure. The BRUISE CONTROL trial demonstrated that continued warfarin during device implantation was superior to heparin bridging (fewer pocket hematomas). For AF ablation specifically: DOACs are continued without interruption (skip only the morning dose the day of the procedure in some protocols) or held for one dose. Intraprocedural heparin is administered after transseptal puncture, targeting an ACT > 300-350 seconds (checked every 15-30 minutes). Post-procedure: resume DOAC the evening of the procedure or the next morning. Anticoagulation is continued for a minimum of 2 months after ablation; long-term anticoagulation decisions are based on CHA2DS2-VASc score, NOT on the perceived success of the ablation (AF can recur silently).
Complications of AF Ablation
| Complication | Incidence | Mechanism & Management |
|---|---|---|
| Cardiac tamponade | 1-2% | Perforation from transseptal puncture or ablation. Pericardiocentesis; surgical repair if needed. Most feared acute complication. |
| Pulmonary vein stenosis | < 1% (with WACA) | Thermal injury to the PV; presents weeks-months later with dyspnea, cough, hemoptysis. CT/MRI for diagnosis. PV angioplasty/stenting. |
| Atrial-esophageal fistula | ~0.04% (rare but lethal) | Thermal injury to the esophagus from posterior wall ablation. Presents 2-4 weeks post-ablation with fever, sepsis, stroke, GI bleeding. Mortality > 50%. Prevention: esophageal temperature monitoring, reduced power on posterior wall, PFA (tissue-selective). Emergency |
| Phrenic nerve injury | 2-5% (cryoballoon) | Right phrenic nerve injury during right superior PV ablation. Monitor with phrenic nerve pacing during ablation. Usually transient (resolves within months). |
| Stroke/TIA | < 1% | Thromboembolism. Prevention: uninterrupted anticoagulation, intraprocedural heparin (ACT > 300-350 sec), bubble management. |
| Vascular access complications | 2-4% | Femoral hematoma, AV fistula, pseudoaneurysm. Manual compression, ultrasound-guided repair. |
28 VT Ablation
Substrate-Based Approach
Most scar-related VT ablation is performed using a substrate-based approach rather than relying solely on activation mapping during VT (which may be hemodynamically unstable). The procedure involves: (1) creating a detailed 3D voltage map of the LV (or RV) in sinus rhythm, (2) identifying scar (< 0.5 mV), border zone (0.5-1.5 mV), and conducting channels within the scar, (3) ablating abnormal electrograms (LAVAs, late potentials, fractionated signals) to eliminate the VT substrate. The VISTA trial demonstrated that substrate ablation (scar homogenization) during first ICD implant in post-MI patients reduced VT recurrence.
Epicardial Access & Ablation
Some VTs have an epicardial origin or circuit (especially in non-ischemic cardiomyopathy, ARVC, Chagas disease, and some idiopathic VTs). Epicardial access is obtained via subxiphoid percutaneous puncture (Sosa technique): under fluoroscopic guidance, a needle is advanced into the pericardial space, and a catheter is introduced for mapping and ablation. Complications include: coronary artery injury (must perform coronary angiography before ablating near epicardial vessels), phrenic nerve injury, hepatic or abdominal organ injury, and pericarditis. The pericardial fat pad can insulate the epicardium and reduce RF efficacy — irrigated ablation and higher power may be needed.
STAR Mapping & Scar Dechanneling
STAR mapping (Scar-related Tachycardia Activation Recording): A technique for mapping VT circuits within scar using ultra-high-density mapping catheters (Pentaray, Orion) during short runs of induced or spontaneous VT. Scar dechanneling: Identification and ablation of conducting channels within the scar by targeting the entrance (earliest LAVA) to eliminate the channels through which VT circuits propagate. This focused approach aims to eliminate VT while minimizing the total ablation volume. The Vergara et al. approach combines activation and substrate mapping for comprehensive VT ablation.
Outcomes of VT Ablation
Acute success (non-inducibility of clinical VT at the end of the procedure): ~70-80%. Freedom from VT recurrence at 1 year: ~50-70% (varies by substrate). Complications: ~5-10% (vascular access, tamponade, stroke, death ~0.5-1%). Timing: The VANISH trial (2016) showed that catheter ablation was superior to escalated antiarrhythmic drug therapy for patients with ischemic cardiomyopathy, prior ICD, and VT on amiodarone. The PARTITA trial (2022) demonstrated that early VT ablation after a first ICD shock (rather than waiting for VT storm) reduced the composite of VT storm, HF hospitalization, and death.
Sapp JL et al. N Engl J Med. 2016;375:111-121 (VANISH). PMID: 27197853Idiopathic VT Ablation
RVOT VT/PVC ablation: The most common idiopathic VT arises from the RVOT. Activation mapping identifies the earliest site of activation during VT or PVC. Pace mapping confirms the target (12/12 QRS match). RF ablation success rate is > 90% with low complication rates. Alternative origins: LVOT (aortic cusps — especially the left coronary cusp and the aortomitral continuity), papillary muscles (more challenging), and the epicardial summit (near the LV summit — may be inaccessible due to proximity to coronary arteries). PVC-induced cardiomyopathy: Frequent PVCs (> 10-15% PVC burden on 24-hour Holter) can cause a reversible dilated cardiomyopathy. PVC ablation eliminates the PVCs and allows LV recovery, typically within 3-6 months. This is an increasingly recognized and treatable cause of heart failure.
29 Classification Systems
Vaughan-Williams Antiarrhythmic Classification
| Class | Mechanism | Subclass | Agents | Key Uses |
|---|---|---|---|---|
| Class I | Sodium channel blockers | Ia — moderate Na+ block, prolong AP | Procainamide, quinidine, disopyramide | Procainamide: WCT, Brugada provocation. Quinidine: Brugada (Ito block) |
| Ib — weak Na+ block, shorten AP | Lidocaine, mexiletine | Lidocaine: VT/VF (ACLS). Mexiletine: LQT3, VT adjunct | ||
| Ic — strong Na+ block, no AP change | Flecainide, propafenone | Flecainide: AF/SVT (no structural heart disease), CPVT adjunct. Propafenone: AF/SVT | ||
| Class II | Beta-adrenergic blockers | — | Metoprolol, atenolol, nadolol, propranolol, esmolol | Rate control, LQTS, CPVT, post-MI VT prophylaxis |
| Class III | Potassium channel blockers (prolong repolarization) | — | Amiodarone, sotalol, dofetilide, ibutilide, dronedarone | Amiodarone: VT/VF, AF. Sotalol: AF, VT. Dofetilide: AF (in-hospital initiation). Ibutilide: acute AF/flutter conversion |
| Class IV | Calcium channel blockers (L-type) | — | Verapamil, diltiazem | AV nodal arrhythmias, rate control, fascicular VT (verapamil) |
| Class V (others) | Miscellaneous mechanisms | — | Adenosine (A1 receptor), digoxin (Na+/K+ ATPase), ivabradine (If), magnesium | Adenosine: SVT diagnosis/termination. Digoxin: rate control. Ivabradine: inappropriate sinus tachycardia |
Lown Grading of Ventricular Premature Beats
| Grade | Description |
|---|---|
| 0 | No VPBs |
| 1 | < 30 VPBs per hour |
| 2 | > 30 VPBs per hour |
| 3 | Multiform VPBs |
| 4a | Couplets (2 consecutive VPBs) |
| 4b | Non-sustained VT (≥ 3 consecutive VPBs) |
| 5 | R-on-T phenomenon (VPB falling on the T wave) |
CHA2DS2-VASc Score
| Risk Factor | Points |
|---|---|
| Congestive heart failure / LV dysfunction | 1 |
| Hypertension | 1 |
| Age ≥ 75 years | 2 |
| Diabetes mellitus | 1 |
| Stroke / TIA / thromboembolism | 2 |
| Vascular disease (prior MI, PAD, aortic plaque) | 1 |
| Age 65-74 years | 1 |
| Sex category (female) | 1 |
| Maximum score: 9. Anticoagulation: men ≥ 2, women ≥ 3. | |
HAS-BLED Score
| Risk Factor | Points |
|---|---|
| Hypertension (uncontrolled, SBP > 160 mmHg) | 1 |
| Abnormal renal function (dialysis, transplant, Cr > 2.26 mg/dL) | 1 |
| Abnormal liver function (cirrhosis, bilirubin > 2x ULN, AST/ALT > 3x ULN) | 1 |
| Stroke (prior history) | 1 |
| Bleeding (prior major bleeding or predisposition) | 1 |
| Labile INR (TTR < 60%) | 1 |
| Elderly (age > 65) | 1 |
| Drugs (antiplatelets, NSAIDs) | 1 |
| Alcohol (≥ 8 drinks/week) | 1 |
| Maximum score: 9. Score ≥ 3 = high bleeding risk — modify risk factors, do not withhold anticoagulation. | |
EHRA AF Symptom Score
| Score | Symptoms |
|---|---|
| EHRA I | No symptoms |
| EHRA IIa | Mild symptoms — normal daily activity not affected |
| EHRA IIb | Moderate symptoms — normal daily activity not affected but patient troubled by symptoms |
| EHRA III | Severe symptoms — normal daily activity affected |
| EHRA IV | Disabling symptoms — normal daily activity discontinued |
Killip Classification (Acute MI)
| Class | Findings | Mortality |
|---|---|---|
| I | No heart failure | ~6% |
| II | Rales, S3, JVD | ~17% |
| III | Pulmonary edema | ~38% |
| IV | Cardiogenic shock | ~81% |
NYHA Functional Classification
| Class | Description |
|---|---|
| I | No limitation of physical activity |
| II | Slight limitation — comfortable at rest, ordinary activity causes symptoms |
| III | Marked limitation — comfortable at rest, less than ordinary activity causes symptoms |
| IV | Unable to carry on any physical activity without discomfort — symptoms at rest |
Wells Score for Pulmonary Embolism (EP-Relevant)
Pulmonary embolism is a common cause of new-onset atrial arrhythmias (AF, atrial flutter, MAT), sinus tachycardia, and RBBB. The Wells score stratifies PE probability: clinical signs of DVT (3 points), PE most likely diagnosis (3), heart rate > 100 (1.5), immobilization/surgery in prior 4 weeks (1.5), previous DVT/PE (1.5), hemoptysis (1), malignancy (1). Score > 4 = PE likely, warranting CT pulmonary angiography. Acute RV strain from PE can also precipitate VT.
Schwartz Score for LQTS Diagnosis
| Criterion | Points |
|---|---|
| QTc ≥ 480 ms | 3 |
| QTc 460-479 ms | 2 |
| QTc 450-459 ms (males) | 1 |
| Torsades de pointes | 2 |
| T-wave alternans | 1 |
| Notched T waves in ≥ 3 leads | 1 |
| Low heart rate for age | 0.5 |
| Syncope with stress | 2 |
| Syncope without stress | 1 |
| Congenital deafness | 0.5 |
| Family member with definite LQTS | 1 |
| Unexplained SCD in family < 30 years old | 0.5 |
| ≤ 1 point: low probability. 1.5-3 points: intermediate. ≥ 3.5 points: high probability of LQTS. | |
30 Medications Master Table
Antiarrhythmic Drugs
| Drug | Class | Route | Key Indications | Major Side Effects / Monitoring |
|---|---|---|---|---|
| Amiodarone | III (also I, II, IV) | IV / PO | VT/VF (ACLS), AF (rate + rhythm), VT storm | Pulmonary toxicity (fibrosis), thyroid (hypo/hyper — contains iodine), hepatotoxicity, corneal microdeposits, photosensitivity, peripheral neuropathy, blue-gray skin. Monitor: PFTs, TFTs, LFTs, eye exam every 6-12 months. Very long half-life (~40-55 days) |
| Sotalol | III + II (beta-blocker) | PO | AF maintenance, VT prevention | QT prolongation (dose-dependent, must initiate in hospital with telemetry for 3 days), torsades de pointes, bradycardia, bronchospasm. Renally cleared — reduce dose for CKD. Contraindicated: CrCl < 40 (80 mg dose), QTc > 500 ms |
| Dofetilide | III (pure IKr blocker) | PO | AF/flutter maintenance | QT prolongation, torsades (3% risk). MUST be initiated in hospital with 3-day telemetry and strict CrCl-based dosing. Multiple drug interactions (verapamil, HCTZ, ketoconazole — contraindicated). Monitor QTc every dose; hold if QTc > 500 ms or increases > 15% |
| Flecainide | Ic | PO | AF/SVT (no structural heart disease), "pill-in-pocket", CPVT adjunct | Proarrhythmic (CAST trial — avoid in structural/ischemic heart disease), may organize AF into slow flutter with 1:1 conduction (give with AV nodal blocker), negative inotropy. Monitor: ECG (QRS widening > 25% = excessive) |
| Propafenone | Ic (+ mild II) | PO | AF/SVT (no structural heart disease) | Similar to flecainide. Mild beta-blocking activity. Metallic taste. Hepatic metabolism (CYP2D6 — poor metabolizers have higher levels). Avoid in structural heart disease |
| Procainamide | Ia | IV / PO | WCT (preferred agent for stable WCT), pre-excited AF, Brugada provocation test | Hypotension (IV infusion — monitor BP), QT prolongation, lupus-like syndrome (chronic PO use — anti-histone antibodies), agranulocytosis (rare). IV: load 15-18 mg/kg over 30-60 min, then 1-4 mg/min |
| Lidocaine | Ib | IV | VT/VF (ACLS, second-line after amiodarone), VT storm adjunct | CNS toxicity (drowsiness, seizures, confusion), bradycardia. Hepatically metabolized — reduce dose in liver failure and HF (reduced hepatic blood flow). Load 1-1.5 mg/kg, then 1-4 mg/min |
| Mexiletine | Ib | PO | VT adjunct (with amiodarone or sotalol), LQT3 (shortens QT) | GI intolerance (nausea — take with food), tremor, dizziness. Oral lidocaine analog |
| Ibutilide | III | IV only | Acute cardioversion of AF/flutter | QT prolongation, torsades (4-8% risk — must monitor for ≥ 4 hours after infusion). More effective for flutter than AF. Dose: 1 mg over 10 min, may repeat x1 |
| Dronedarone | III (amiodarone analog without iodine) | PO | AF maintenance (non-permanent AF, no HF) | Hepatotoxicity (monitor LFTs), increased mortality in permanent AF (PALLAS trial) and severe HF (ANDROMEDA trial) — contraindicated in both. Less toxic than amiodarone but less effective |
| Adenosine | V (A1 receptor agonist) | IV (rapid push) | SVT diagnosis/termination | Transient asystole (warn patient), chest tightness, flushing, bronchospasm. Ultra-short half-life (~10 seconds). Enhanced by dipyridamole, antagonized by caffeine and theophylline. Dose: 6 mg → 12 mg → 12 mg |
Anticoagulants for AF
| Drug | Mechanism | Dose for AF | Key Considerations |
|---|---|---|---|
| Apixaban | Direct factor Xa inhibitor | 5 mg BID (2.5 mg BID if ≥ 2 of: age ≥ 80, weight ≤ 60 kg, Cr ≥ 1.5) | Best bleeding profile (ARISTOTLE). No routine monitoring. Reversal: andexanet alfa |
| Rivaroxaban | Direct factor Xa inhibitor | 20 mg daily with dinner (15 mg if CrCl 15-50) | Once daily dosing. ROCKET AF. Reversal: andexanet alfa |
| Dabigatran | Direct thrombin inhibitor | 150 mg BID (75 mg BID if CrCl 15-30) | RE-LY trial. Dyspepsia common. Reversal: idarucizumab (Praxbind) |
| Edoxaban | Direct factor Xa inhibitor | 60 mg daily (30 mg if CrCl 15-50, weight ≤ 60 kg, or P-gp inhibitor) | ENGAGE AF-TIMI 48. Less effective at high CrCl (> 95 — avoid). Reversal: andexanet alfa |
| Warfarin | Vitamin K antagonist | Dose adjusted to INR 2.0-3.0 | Required for mechanical valves (INR 2.5-3.5) and moderate-severe mitral stenosis. Multiple food/drug interactions. Monitor INR regularly. Reversal: vitamin K, 4-factor PCC, FFP |
Rate Control Agents
| Drug | Class | Route | Dose | Notes |
|---|---|---|---|---|
| Metoprolol | Beta-1 selective blocker | IV / PO | IV: 5 mg q5min x3. PO: 25-200 mg BID (tartrate) or 25-400 mg daily (succinate) | First-line for rate control. Avoid in acute decompensated HF, severe bronchospasm |
| Diltiazem | Non-DHP CCB | IV / PO | IV: 0.25 mg/kg bolus, then 5-15 mg/hr. PO: 120-360 mg daily (ER) | Effective rate control. Avoid in HFrEF (negative inotropy). Avoid with beta-blockers (risk of severe bradycardia) |
| Verapamil | Non-DHP CCB | IV / PO | IV: 2.5-5 mg over 2 min. PO: 120-480 mg daily | Rate control, terminates AVNRT, treats fascicular VT. Avoid in HFrEF. CONTRAINDICATED in pre-excited AF (can cause VF) |
| Digoxin | Cardiac glycoside | IV / PO | PO: 0.125-0.25 mg daily (target level 0.5-0.9 ng/mL) | Rate control at rest (not during exercise). Useful in HFrEF (mild positive inotrope). Narrow therapeutic window. Toxicity enhanced by hypokalemia |
Drug-Induced QT Prolongation — High-Risk Agents
Beyond antiarrhythmic drugs, many non-cardiac medications prolong the QT interval and can cause torsades de pointes. The most important categories include:
| Category | High-Risk Drugs |
|---|---|
| Antibiotics | Fluoroquinolones (moxifloxacin > levofloxacin > ciprofloxacin), macrolides (erythromycin, azithromycin), trimethoprim-sulfamethoxazole |
| Antifungals | Fluconazole, voriconazole, ketoconazole |
| Antipsychotics | Haloperidol, droperidol, chlorpromazine, ziprasidone, thioridazine |
| Antidepressants | Citalopram (> 40 mg), escitalopram, tricyclics (amitriptyline) |
| Antiemetics | Ondansetron (IV > PO), domperidone |
| Opioids | Methadone (dose-dependent, significant risk at > 100 mg/day) |
Risk factors for drug-induced torsades: Female sex (women have a longer baseline QTc), hypokalemia, hypomagnesemia, bradycardia, baseline QT prolongation, concomitant QT-prolonging drugs, heart failure, hepatic/renal impairment (reducing drug clearance), and genetic susceptibility (subclinical LQTS mutations). The CredibleMeds.org database provides regularly updated lists of QT-prolonging drugs categorized by risk level.
31 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| AAD | Antiarrhythmic drug |
| ACT | Activated clotting time |
| AF | Atrial fibrillation |
| AFL | Atrial flutter |
| AHRE | Atrial high-rate episode |
| AP | Accessory pathway |
| ARVC | Arrhythmogenic right ventricular cardiomyopathy |
| AT | Atrial tachycardia |
| ATP | Anti-tachycardia pacing |
| AV | Atrioventricular |
| AVNRT | Atrioventricular nodal reentrant tachycardia |
| AVRT | Atrioventricular reciprocating tachycardia |
| BBB | Bundle branch block |
| CF | Contact force |
| CFAE | Complex fractionated atrial electrograms |
| CIED | Cardiac implantable electronic device |
| CPVT | Catecholaminergic polymorphic ventricular tachycardia |
| CRT | Cardiac resynchronization therapy |
| CS | Coronary sinus |
| CTI | Cavotricuspid isthmus |
| DAD | Delayed afterdepolarization |
| DCM | Dilated cardiomyopathy |
| DOAC | Direct oral anticoagulant |
| EAD | Early afterdepolarization |
| EGM | Electrogram |
| EP | Electrophysiology |
| EPS | Electrophysiology study |
| ERI | Elective replacement indicator |
| ERP | Effective refractory period |
| GDMT | Guideline-directed medical therapy |
| HBP | His bundle pacing |
| HCM | Hypertrophic cardiomyopathy |
| HRA | High right atrium |
| ICD | Implantable cardioverter-defibrillator |
| ILR | Implantable loop recorder |
| IVCD | Intraventricular conduction delay |
| LAA | Left atrial appendage |
| LAFB | Left anterior fascicular block |
| LAVA | Local abnormal ventricular activity |
| LBBB | Left bundle branch block |
| LBBAP | Left bundle branch area pacing |
| LCSD | Left cardiac sympathetic denervation |
| LGE | Late gadolinium enhancement |
| LMNA | Lamin A/C (gene) |
| LPFB | Left posterior fascicular block |
| LQTS | Long QT syndrome |
| LVEF | Left ventricular ejection fraction |
| MAT | Multifocal atrial tachycardia |
| NSVT | Non-sustained ventricular tachycardia |
| PFA | Pulsed field ablation |
| PPM | Permanent pacemaker |
| PV | Pulmonary vein |
| PVC | Premature ventricular complex |
| PVI | Pulmonary vein isolation |
| RBBB | Right bundle branch block |
| RF | Radiofrequency |
| RM | Remote monitoring |
| RVOT | Right ventricular outflow tract |
| SA | Sinoatrial |
| SCD | Sudden cardiac death |
| S-ICD | Subcutaneous implantable cardioverter-defibrillator |
| SPERRI | Shortest pre-excited R-R interval |
| SSS | Sick sinus syndrome |
| SVT | Supraventricular tachycardia |
| TEE | Transesophageal echocardiography |
| TTM | Targeted temperature management |
| VF | Ventricular fibrillation |
| VT | Ventricular tachycardia |
| WACA | Wide-area circumferential ablation |
| WCD | Wearable cardioverter-defibrillator |
| WPW | Wolff-Parkinson-White |
| ARP | Absolute refractory period |
| BiV | Biventricular |
| CL | Cycle length |
| DCCV | Direct current cardioversion |
| DFT | Defibrillation threshold |
| EAM | Electroanatomical mapping |
| EOL | End of life (battery) |
| FTI | Force-time integral |
| ICE | Intracardiac echocardiography |
| IST | Inappropriate sinus tachycardia |
| LAO | Left anterior oblique |
| LV | Left ventricle / left ventricular |
| NBG | NASPE/BPEG Generic (pacemaker code) |
| PAC | Premature atrial complex |
| PJRT | Permanent junctional reciprocating tachycardia |
| PROM | Predicted risk of mortality |
| RAO | Right anterior oblique |
| RV | Right ventricle / right ventricular |
| SVC | Superior vena cava |
| TdP | Torsades de pointes |
| VPB | Ventricular premature beat |