Otolaryngology (ENT)
Every diagnosis, procedure, surgical technique, classification, complication, medication, and management algorithm across the full scope of otolaryngology — head and neck surgery in one place.
01 Head & Neck Anatomy
Temporal Bone
The temporal bone is one of the most complex bones in the body, housing the organs of hearing and balance. It consists of five parts: squamous (forms the lateral skull wall), mastoid (contains air cells communicating with the middle ear via the aditus ad antrum), petrous (contains the inner ear — cochlea and vestibular labyrinth — and the internal auditory canal), tympanic (forms the anterior, inferior, and posterior walls of the external auditory canal), and the styloid process.
The middle ear (tympanic cavity) is an air-filled space containing the three ossicles: malleus (handle attaches to the tympanic membrane), incus (body articulates with the malleus head; long process articulates with the stapes), and stapes (footplate occupies the oval window). The middle ear is bounded laterally by the tympanic membrane, medially by the promontory (overlying the basal turn of the cochlea), superiorly by the tegmen tympani (separates from the middle cranial fossa), and inferiorly by the jugular bulb. The chorda tympani nerve (branch of CN VII) traverses the middle ear between the malleus and incus, carrying taste from the anterior two-thirds of the tongue.
The inner ear consists of the bony labyrinth (cochlea, vestibule, three semicircular canals) containing the membranous labyrinth. The cochlea has 2.5 turns and contains three fluid-filled chambers: scala vestibuli (perilymph), scala media (endolymph — high K+, low Na+, produced by the stria vascularis), and scala tympani (perilymph). The organ of Corti sits on the basilar membrane within the scala media and contains inner and outer hair cells — the primary sensory transducers for hearing.
Paranasal Sinuses
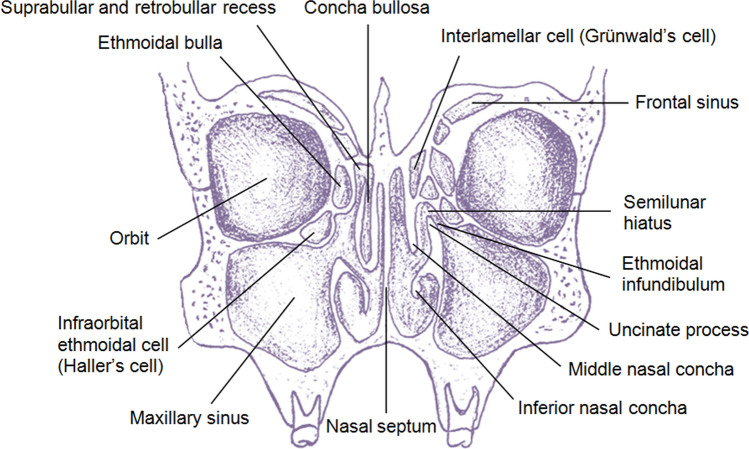
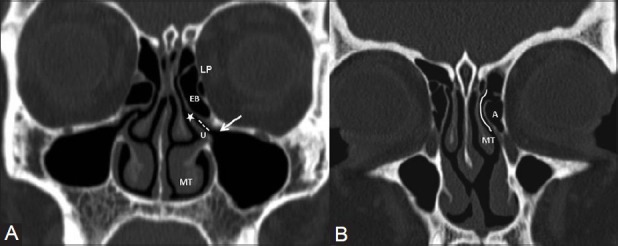
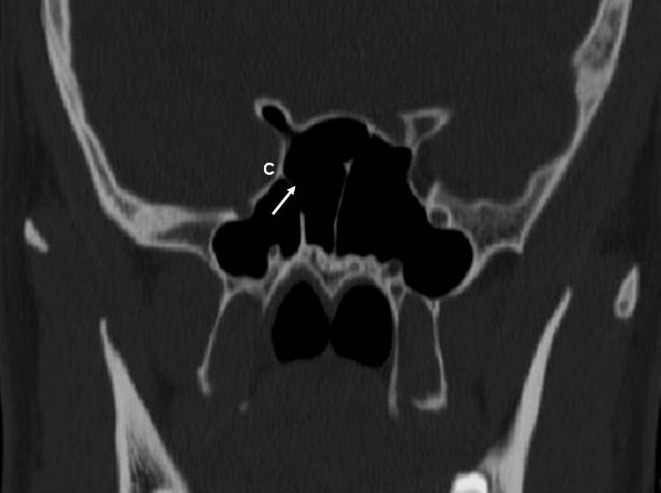
Four paired sinuses drain into the nasal cavity: maxillary (largest, drains via the natural ostium into the middle meatus through the hiatus semilunaris), ethmoid (anterior cells drain into the middle meatus; posterior cells drain into the superior meatus/sphenoethmoidal recess), frontal (drains via the frontal recess/nasofrontal duct into the middle meatus), and sphenoid (drains into the sphenoethmoidal recess). The ostiomeatal complex (OMC) is the common drainage pathway for the maxillary, anterior ethmoid, and frontal sinuses — its obstruction is the key event in the pathogenesis of sinusitis.
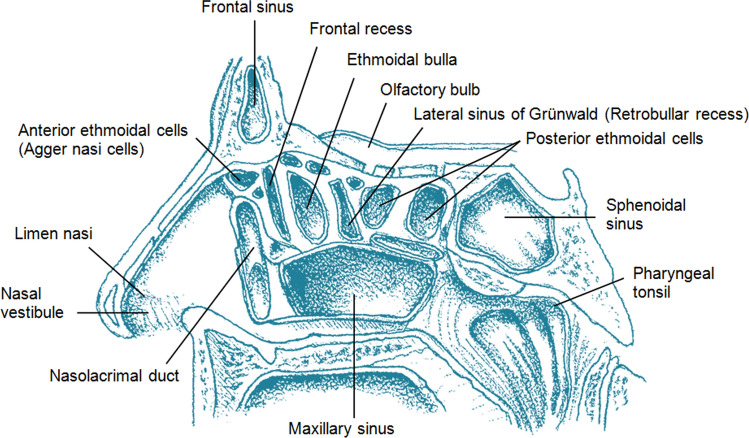
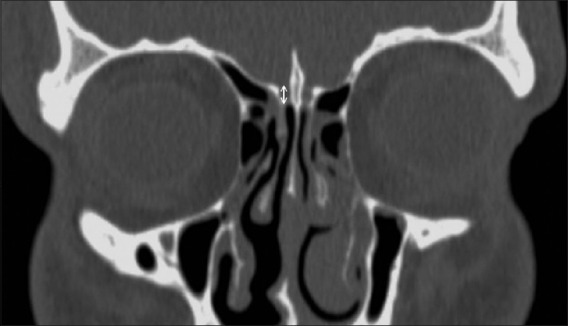
Critical relationships: the lamina papyracea (paper-thin medial wall of the orbit) separates the ethmoid sinuses from the orbit; the skull base (fovea ethmoidalis) forms the roof of the ethmoid sinuses; the optic nerve and internal carotid artery are intimately related to the sphenoid sinus (dehiscent bony walls in 4-8% and 8-25% of patients, respectively). The Keros classification grades the depth of the olfactory fossa (I: 1-3 mm, II: 4-7 mm, III: 8-16 mm) — higher grades have increased risk of skull base injury during FESS.
Larynx Framework
The larynx extends from the epiglottis (C3) to the inferior border of the cricoid cartilage (C6) and is composed of three unpaired cartilages — thyroid (shield-shaped, largest), cricoid (complete ring, only complete cartilaginous ring in the airway), and epiglottis (elastic cartilage) — and three paired cartilages — arytenoid (vocal process attaches the vocal ligament; muscular process receives the posterior cricoarytenoid and lateral cricoarytenoid muscles), corniculate, and cuneiform.
The larynx is divided into three regions: supraglottis (epiglottis, aryepiglottic folds, false vocal folds, ventricle — rich lymphatic drainage, early nodal metastasis), glottis (true vocal folds, anterior commissure, posterior commissure — sparse lymphatics, late nodal disease), and subglottis (from 1 cm below the free edge of the vocal fold to the inferior border of the cricoid — the narrowest portion of the adult airway is the glottis, but in children it is the subglottis at the level of the cricoid).
Pharynx
The pharynx extends from the skull base to the level of C6 and is divided into three regions: nasopharynx (skull base to soft palate — contains the adenoid pad/pharyngeal tonsil, torus tubarius, and Eustachian tube orifice; Rosenmüller's fossa is the most common site of nasopharyngeal carcinoma), oropharynx (soft palate to hyoid bone — contains the palatine tonsils, tongue base/lingual tonsils, soft palate, and posterior pharyngeal wall), and hypopharynx (hyoid to cricopharyngeus — contains the piriform sinuses, postcricoid area, and posterior pharyngeal wall; the piriform sinus is the most common site of hypopharyngeal carcinoma).
The pharyngeal constrictor muscles (superior, middle, inferior) form the muscular wall. The Killian's dehiscence (gap between the oblique fibers of the thyropharyngeus and the horizontal fibers of the cricopharyngeus, both parts of the inferior constrictor) is the weak point where Zenker's diverticulum herniates posteriorly.
02 Neurovascular Anatomy
Facial Nerve (CN VII)
The facial nerve has the longest intraosseous course of any cranial nerve. It exits the brainstem at the cerebellopontine angle (CPA), enters the internal auditory canal (IAC) in the anterosuperior quadrant, then courses through the temporal bone in three segments: labyrinthine (narrowest segment — most common site of entrapment in Bell's palsy; from fundus of IAC to geniculate ganglion), tympanic (from geniculate ganglion across the medial wall of the middle ear, above the oval window and below the lateral semicircular canal), and mastoid (descends from the second genu to the stylomastoid foramen).
Key branches: greater superficial petrosal nerve (at geniculate ganglion — parasympathetic to lacrimal gland), nerve to stapedius (mastoid segment — stapedial reflex), chorda tympani (distal mastoid segment — taste to anterior 2/3 of tongue, parasympathetic to submandibular and sublingual glands). After exiting the stylomastoid foramen, the nerve enters the parotid gland and divides at the pes anserinus into five terminal branches: temporal, zygomatic, buccal, marginal mandibular, and cervical.
Recurrent Laryngeal Nerve (RLN) & Superior Laryngeal Nerve (SLN)
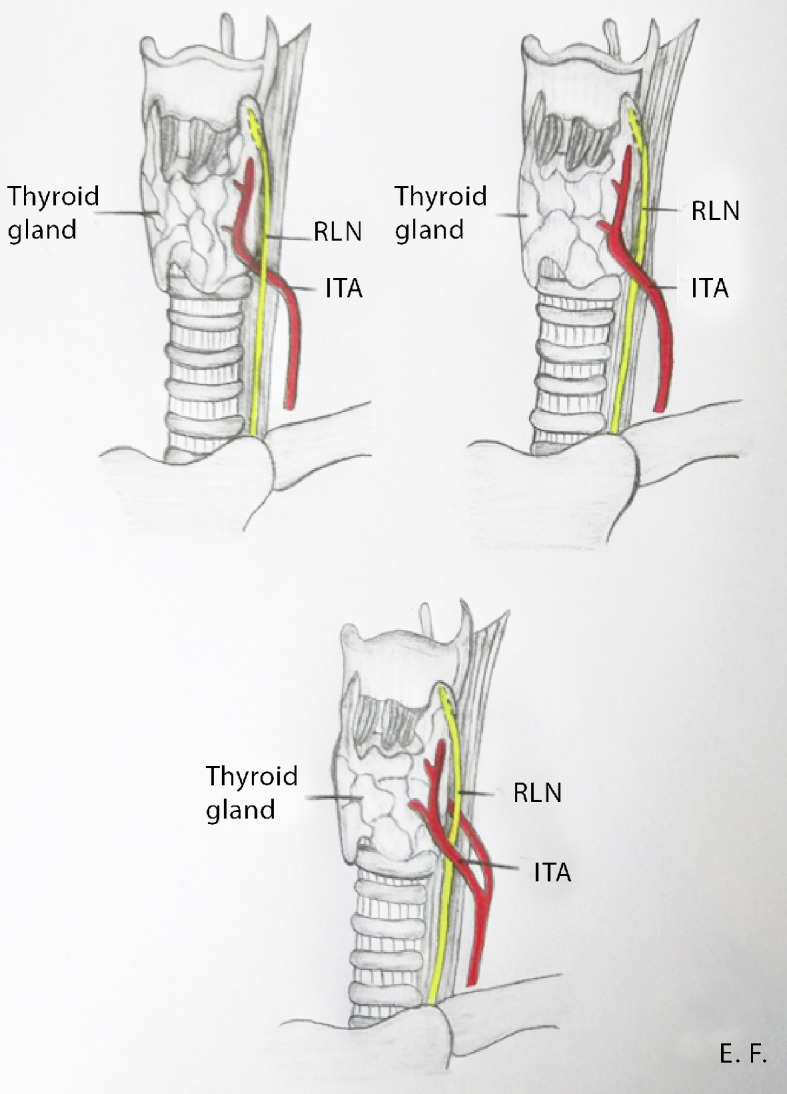
The recurrent laryngeal nerve branches from the vagus nerve (CN X). The left RLN loops around the aortic arch (ligamentum arteriosum) and ascends in the tracheoesophageal groove. The right RLN loops around the right subclavian artery. Both nerves enter the larynx posterior to the cricothyroid joint. The RLN innervates all intrinsic laryngeal muscles except the cricothyroid (which is innervated by the external branch of the SLN). A non-recurrent laryngeal nerve occurs in ~0.5-1% of patients on the right (associated with an aberrant right subclavian artery — arteria lusoria) and is extremely rare on the left.
The superior laryngeal nerve divides into the internal branch (sensory to the supraglottis — pierces the thyrohyoid membrane) and the external branch (motor to the cricothyroid muscle — a tensor of the vocal fold, affecting pitch). The external branch of the SLN (EBSLN) is at risk during superior pole ligation in thyroidectomy. The Cernea classification describes its relationship to the superior thyroid artery and superior pole.
Cranial Nerves VII-XII in the Head & Neck
| CN | Name | Key Function in H&N | Surgical Risk |
|---|---|---|---|
| VII | Facial | Motor to face; taste anterior 2/3 tongue; lacrimation | Parotidectomy, mastoidectomy, CPA surgery |
| VIII | Vestibulocochlear | Hearing & balance | CPA tumors, temporal bone surgery |
| IX | Glossopharyngeal | Sensation posterior 1/3 tongue; stylopharyngeus; parotid secretion | Tonsillectomy (tonsillar fossa), skull base surgery |
| X | Vagus | Laryngeal motor/sensory (via RLN, SLN); pharyngeal plexus | Thyroidectomy, carotid surgery, neck dissection |
| XI | Spinal accessory | Motor to SCM and trapezius | Posterior triangle neck dissection (Level V) |
| XII | Hypoglossal | Motor to tongue | Submandibular triangle surgery, tongue base surgery |
Carotid & Jugular Relationships
The carotid sheath contains the common carotid artery (medial), internal jugular vein (lateral), and vagus nerve (posterior). The internal carotid artery (ICA) courses posteromedial to the tonsillar fossa (1.5-2.5 cm lateral) — aberrant ICA is a surgical hazard during tonsillectomy and parapharyngeal space surgery. In the sphenoid sinus, the ICA courses along the lateral wall — dehiscence occurs in 8-25% of patients. The external carotid artery (ECA) gives off branches critical in ENT: superior thyroid artery (supplies upper thyroid pole), lingual artery, facial artery, ascending pharyngeal artery, occipital artery, posterior auricular artery, and terminal branches (maxillary and superficial temporal arteries). The sphenopalatine artery (terminal branch of the internal maxillary artery from the ECA) is the primary arterial supply to the posterior nasal cavity — its ligation is the definitive surgical treatment for posterior epistaxis.
03 The ENT Examination
Otoscopy
The otoscopic examination begins with inspection of the auricle and periauricular region (pre-auricular pits, post-auricular swelling in mastoiditis). The external auditory canal is examined for cerumen, otorrhea, masses, and exostoses. Pull the pinna posterosuperiorly in adults (posteroinferiorly in infants) to straighten the canal. The tympanic membrane (TM) is assessed for color (pearly gray = normal; erythematous, bulging = AOM; retracted = negative middle ear pressure/OME), integrity (perforation location and size), and mobility (pneumatic otoscopy — decreased mobility suggests effusion). Key TM landmarks: pars tensa, pars flaccida (Shrapnell's membrane — retraction pockets here suggest cholesteatoma), umbo, handle of malleus, light reflex (anteroinferior quadrant), and lateral process of the malleus.
Anterior Rhinoscopy & Nasal Endoscopy
Anterior rhinoscopy with a nasal speculum and headlight provides a limited view of the anterior nasal cavity — assess for septal deviation, turbinate hypertrophy, polyps, masses, and mucosal quality. Flexible nasolaryngoscopy (FNL) is the gold standard for comprehensive evaluation of the nasal cavity, nasopharynx, oropharynx, hypopharynx, and larynx. After topical decongestion (oxymetazoline 0.05%) and anesthesia (lidocaine 4%), the scope is passed along the floor of the nose. Three passes are standard: (1) floor of nose to nasopharynx (Eustachian tube orifice, adenoid pad), (2) middle meatus (uncinate, bulla ethmoidalis, polyps, purulence), (3) nasopharynx to larynx (vocal fold mobility, supraglottic structures, pooling of secretions).
Tuning Fork Tests
| Test | Technique | Conductive Loss | Sensorineural Loss |
|---|---|---|---|
| Weber (512 Hz) | Tuning fork on vertex of skull | Lateralizes to affected ear | Lateralizes to better (unaffected) ear |
| Rinne (512 Hz) | Compare air conduction (AC) at EAC vs bone conduction (BC) at mastoid | BC > AC (Rinne negative) in affected ear | AC > BC (Rinne positive) bilaterally |

Complete Head & Neck Exam
The head and neck examination includes systematic evaluation of: oral cavity (lips, buccal mucosa, hard palate, floor of mouth — bimanual palpation, tongue mobility, dentition), oropharynx (soft palate, tonsils, tongue base), neck (palpation of thyroid, lymph node levels I-VI, parotid and submandibular glands, laryngeal framework — cricothyroid membrane, thyroid notch), cranial nerve examination (CN V motor/sensory, VII — forehead wrinkling, eye closure, smile; IX/X — gag reflex, palate elevation; XI — shoulder shrug, head turn; XII — tongue protrusion). Flexible nasolaryngoscopy completes the exam to evaluate structures not visible on mirror or direct inspection.
Stroboscopy
Videolaryngostroboscopy uses a strobe light synchronized to the patient's fundamental frequency to create a slow-motion view of vocal fold vibration. It evaluates the mucosal wave (propagation of the mucosa over the vocal fold body — reduced or absent in scarring, sulcus vocalis, or carcinoma), glottic closure pattern (complete, incomplete, hourglass, spindle-shaped), amplitude (extent of lateral excursion), and symmetry/periodicity of vibration. Stroboscopy is the single most important tool for evaluating voice disorders and differentiating benign from malignant vocal fold lesions — a stiff, non-vibrating segment strongly suggests malignancy or deep invasion.
Otitis Externa
Acute otitis externa (AOE) ("swimmer's ear") is diffuse infection of the EAC skin, most commonly caused by Pseudomonas aeruginosa and S. aureus. Presents with otalgia (worsened by tragal pressure or pinna manipulation), otorrhea, canal edema, and debris. Treatment: aural toilet, topical antibiotic drops (fluoroquinolone ± steroid — ciprofloxacin-dexamethasone, ofloxacin), wick placement for severely edematous canals. Avoid systemic antibiotics unless cellulitis extends beyond the canal. Malignant (necrotizing) otitis externa is an aggressive, life-threatening infection of the skull base, almost exclusively in elderly diabetic or immunocompromised patients. Causative organism: P. aeruginosa (~95%). Hallmark finding: granulation tissue at the bony-cartilaginous junction of the EAC floor. Progression: osteomyelitis of the temporal bone, cranial nerve palsies (CN VII most common, then IX, X, XI, XII), sigmoid sinus thrombosis, meningitis. Diagnosis: CT (bone erosion), Tc-99m bone scan or Ga-67 scan (active infection), MRI (soft tissue extent). Treatment: prolonged IV antipseudomonal antibiotics (ciprofloxacin IV/PO, or piperacillin-tazobactam) for 6-8 weeks minimum; serial inflammatory markers (ESR, CRP) to guide duration; surgical debridement for refractory cases. Emergency
Bell's Palsy
Bell's palsy is the most common cause of acute unilateral facial paralysis. Presumed viral etiology (HSV-1 reactivation in the geniculate ganglion). Presents with rapid-onset (24-72 hours) unilateral facial weakness involving all branches (upper and lower face — distinguishes it from central/UMN lesions which spare the forehead). Associated symptoms: ear pain, hyperacusis (stapedial reflex loss), altered taste (chorda tympani), decreased lacrimation. Diagnosis is clinical — must exclude other causes (Ramsay Hunt syndrome/herpes zoster oticus — vesicles on pinna and EAC; Lyme disease; cholesteatoma; parotid tumor; stroke). Treatment: oral corticosteroids (prednisone 60-80 mg/day for 1 week with taper) started within 72 hours — strong recommendation per AAO-HNS CPG. Antivirals (valacyclovir) combined with steroids may offer modest additional benefit. Eye protection is critical: artificial tears, ointment at night, moisture chamber/tape for incomplete eye closure. Prognosis: 70% achieve complete recovery; 85% show some improvement within 3 weeks. ENoG and EMG at 2-3 weeks help predict prognosis. AAO-HNS CPG: Bell's Palsy (2013) PMID 24189771
04 Audiology Basics
Audiogram Interpretation
The pure tone audiogram measures hearing thresholds (in dB HL) at frequencies 250-8000 Hz. Air conduction (AC) is tested with headphones or insert earphones; bone conduction (BC) is tested with a bone oscillator on the mastoid. The air-bone gap (difference between AC and BC thresholds) indicates a conductive component. Normal hearing: thresholds ≤ 25 dB HL at all frequencies.
| Degree of Hearing Loss | Threshold (dB HL) | Impact |
|---|---|---|
| Normal | ≤ 25 | No significant difficulty |
| Mild | 26-40 | Difficulty with soft speech |
| Moderate | 41-55 | Difficulty with conversational speech |
| Moderately severe | 56-70 | Speech must be loud to be heard |
| Severe | 71-90 | Only very loud speech/sounds heard |
| Profound | > 90 | May rely on vibrotactile cues; CI candidate |
Tympanometry
Tympanometry measures middle ear compliance as a function of pressure changes in the ear canal. It generates a tympanogram classified by the Jerger classification:
| Type | Peak Compliance | Peak Pressure | Interpretation |
|---|---|---|---|
| A | Normal | 0 daPa (normal) | Normal middle ear function |
| As | Reduced | Normal | Stiffened system — otosclerosis, tympanosclerosis |
| Ad | Elevated | Normal | Hypermobile TM — ossicular discontinuity |
| B | Flat (no peak) | N/A | Middle ear effusion or TM perforation (distinguish by ear canal volume) |
| C | Normal | Negative (< −100 daPa) | Eustachian tube dysfunction, developing effusion |
Auditory Brainstem Response (ABR)
The ABR records neural activity from the auditory nerve to the brainstem via surface electrodes. Five waves (I-V) correspond to successive anatomical generators: Wave I (distal CN VIII), Wave II (proximal CN VIII), Wave III (cochlear nucleus), Wave IV (superior olivary complex), Wave V (lateral lemniscus/inferior colliculus). The I-V interpeak latency is normally ≤ 4.0 ms. Prolonged I-III interpeak latency suggests retrocochlear pathology (e.g., vestibular schwannoma). ABR is the gold standard for newborn hearing screening and for estimating hearing thresholds in patients who cannot cooperate with behavioral audiometry.
Otoacoustic Emissions (OAE)
OAEs are sounds generated by the active motility of outer hair cells (OHCs) in the cochlea. Two main types: transient-evoked OAE (TEOAE) and distortion product OAE (DPOAE). Present OAEs indicate functioning OHCs and a hearing threshold ≤ 30-35 dB HL. OAEs are absent with sensorineural hearing loss > 30-35 dB HL, middle ear pathology (conductive component attenuates the emission), or OHC damage. OAEs are present in auditory neuropathy spectrum disorder (ANSD) despite abnormal ABR — this distinguishes ANSD from cochlear hearing loss.
05 Hearing Loss
Conductive vs Sensorineural
Conductive hearing loss (CHL) results from impaired sound transmission through the external or middle ear (cerumen impaction, TM perforation, ossicular discontinuity, otosclerosis, middle ear effusion, cholesteatoma). On audiogram: air-bone gap ≥ 10 dB with normal bone conduction. Weber lateralizes to the affected ear; Rinne is negative (BC > AC).
Sensorineural hearing loss (SNHL) results from damage to the cochlea (sensory) or auditory nerve (neural/retrocochlear). On audiogram: air and bone conduction thresholds are equally elevated (no air-bone gap). Weber lateralizes to the unaffected ear; Rinne is positive (AC > BC) bilaterally.
Mixed hearing loss has both conductive and sensorineural components — elevated bone conduction with an additional air-bone gap.
Sudden Sensorineural Hearing Loss (SSNHL)
SSNHL is defined as ≥ 30 dB sensorineural hearing loss across three contiguous frequencies occurring within 72 hours. This is an Emergency — otologic emergency requiring urgent evaluation and treatment. Incidence: 5-20 per 100,000 per year. Most cases are idiopathic (presumed viral cochleitis or microvascular occlusion). Must rule out retrocochlear pathology (vestibular schwannoma — present in ~1-3% of SSNHL cases) with MRI of the IAC with gadolinium.
Treatment: High-dose oral corticosteroids (prednisone 1 mg/kg/day, max 60 mg, for 10-14 days with taper) initiated within 2 weeks of onset — earlier is better. Intratympanic dexamethasone (10-24 mg/mL, injected through the TM into the middle ear, 0.3-0.5 mL per injection, typically 3-4 sessions at weekly intervals) is used as primary therapy if systemic steroids are contraindicated (uncontrolled diabetes, active TB, immunocompromised) or as salvage therapy if oral steroids fail (salvage treatment initiated within 2-6 weeks of onset). Recovery rates: approximately one-third recover fully, one-third partially, and one-third have no recovery. Poor prognostic factors: profound initial hearing loss, downsloping audiometric pattern, delayed treatment, older age, presence of vertigo. AAO-HNS CPG: Sudden Hearing Loss (2012) PMID 22965903
Presbycusis
Presbycusis (age-related hearing loss) is the most common cause of SNHL in adults. It is bilateral, symmetric, and predominantly affects high frequencies. Audiometric pattern: sloping high-frequency SNHL. Speech discrimination declines, especially in noisy environments. Pathology involves loss of hair cells (particularly outer hair cells at the base of the cochlea — sensory presbycusis), strial atrophy (metabolic presbycusis — flat audiometric pattern), or loss of spiral ganglion neurons (neural presbycusis). Treatment: hearing aids; severe-to-profound cases may benefit from cochlear implantation.
Noise-Induced Hearing Loss (NIHL)
NIHL is caused by chronic exposure to hazardous noise levels (> 85 dB over 8 hours) or acute acoustic trauma (> 140 dB impulse noise). Audiometric hallmark: 4 kHz notch (dip at 4 kHz with recovery at 8 kHz). Pathology: damage to outer hair cells, beginning at the basal turn of the cochlea (high-frequency region). NIHL is preventable with hearing protection. There is no effective medical or surgical treatment for established NIHL — hearing aids are the mainstay.
Ototoxicity
Ototoxic medications cause sensorineural hearing loss, tinnitus, or vestibular dysfunction. Key ototoxic agents: Aminoglycosides (gentamicin — predominantly vestibulotoxic; tobramycin, amikacin — predominantly cochleotoxic; irreversible damage to hair cells; genetic susceptibility via mitochondrial DNA mutation m.1555A>G). Cisplatin (dose-dependent, cumulative, irreversible high-frequency SNHL; monitor with serial audiometry during treatment). Loop diuretics (furosemide, ethacrynic acid — usually reversible; potentiated by concurrent aminoglycosides). Salicylates (aspirin at high doses — reversible tinnitus and HL). Quinine, erythromycin (usually reversible). Baseline and serial audiometry is recommended for patients receiving cisplatin or aminoglycoside therapy.
Autoimmune Inner Ear Disease (AIED)
AIED presents with bilateral, rapidly progressive (over weeks to months) sensorineural hearing loss, often with vestibular symptoms. May occur in isolation or associated with systemic autoimmune diseases (rheumatoid arthritis, SLE, ulcerative colitis, Cogan syndrome — interstitial keratitis + audiovestibular dysfunction). Audiometric pattern: bilateral fluctuating or progressive SNHL; may show a "cookie-bite" mid-frequency pattern. Diagnosis is clinical (no definitive lab test); improvement with corticosteroids supports the diagnosis. Treatment: high-dose oral corticosteroids (prednisone 1 mg/kg/day); steroid-sparing agents for maintenance (methotrexate, azathioprine, cyclophosphamide). Cochlear implantation for refractory bilateral profound loss.
06 Otitis Media & Cholesteatoma
Acute Otitis Media (AOM)
AOM is an acute infection of the middle ear, most common in children aged 6-24 months. Risk factors: Eustachian tube dysfunction, daycare attendance, lack of breastfeeding, cleft palate. Most common pathogens: Streptococcus pneumoniae, Haemophilus influenzae (non-typable), Moraxella catarrhalis. Diagnosis requires (1) acute onset, (2) middle ear effusion (bulging TM, decreased mobility on pneumatic otoscopy, air-fluid level), and (3) signs of middle ear inflammation (erythema, otalgia, fever).
Treatment: High-dose amoxicillin (80-90 mg/kg/day) is first-line. Amoxicillin-clavulanate for treatment failure or beta-lactamase producers. Observation (watchful waiting) is appropriate for children ≥ 2 years with mild, unilateral AOM and no otorrhea. Tympanostomy tubes indicated for recurrent AOM (≥ 3 episodes in 6 months or ≥ 4 in 12 months with one in the past 6 months). AAO-HNS CPG: Tympanostomy Tubes (2013) PMID 23479559
Otitis Media with Effusion (OME)
OME is a middle ear effusion without signs of acute infection. Most common cause of conductive hearing loss in children. Most OME resolves spontaneously within 3 months. Tympanostomy tubes are indicated if bilateral OME persists for ≥ 3 months with documented hearing loss (≥ 20 dB) or if OME is associated with speech/language delay, learning difficulties, or structural abnormalities (cleft palate, Down syndrome).
Chronic Suppurative Otitis Media (CSOM)
CSOM is defined as persistent otorrhea through a TM perforation for > 6-12 weeks despite appropriate medical treatment. Common organisms: Pseudomonas aeruginosa, Staphylococcus aureus, anaerobes, and polymicrobial infections. Treatment: aural toilet, topical fluoroquinolone eardrops (ofloxacin or ciprofloxacin-dexamethasone). Surgical management: tympanoplasty (repair of TM perforation using temporalis fascia or perichondrium/cartilage graft) once infection is controlled.
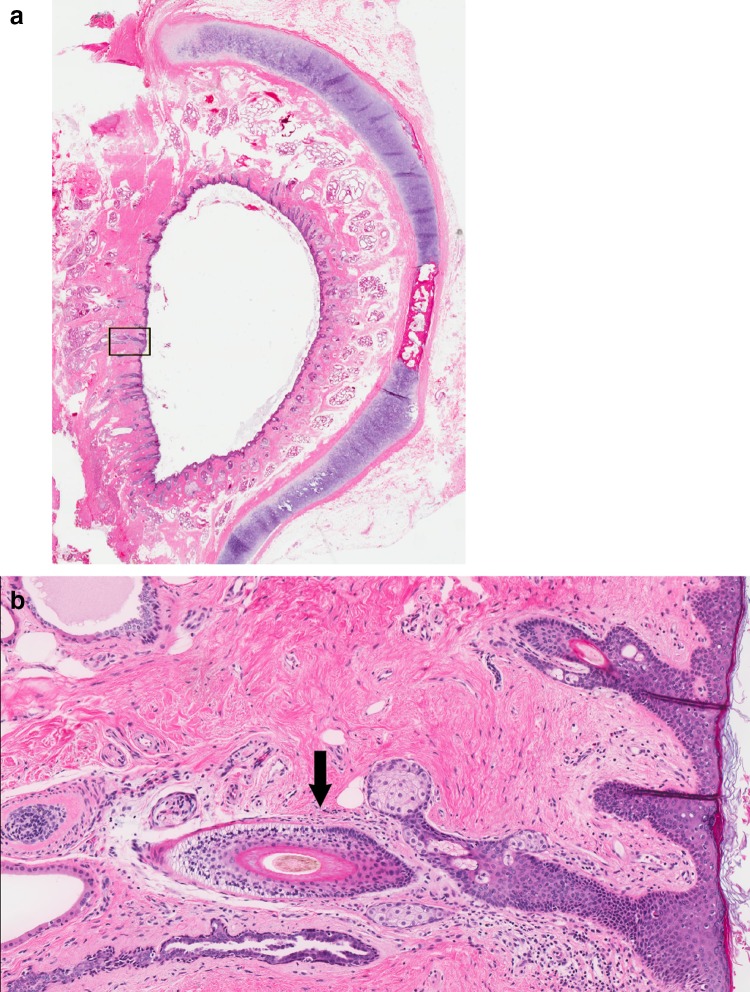
Cholesteatoma
Cholesteatoma is a keratinizing squamous epithelium in the middle ear or mastoid. It is not a true neoplasm but is locally destructive — erodes bone through enzymatic activity (collagenase, osteoclast activation) and pressure necrosis. Classified as congenital (white mass behind an intact TM, no history of infection or surgery — most common in the anterosuperior quadrant of the middle ear) or acquired (primary: pars flaccida retraction pocket with trapped keratinous debris; secondary: migration of squamous epithelium through a TM perforation).
Complications of cholesteatoma: ossicular erosion (conductive hearing loss — incus long process is the most commonly eroded ossicle), labyrinthine fistula (lateral SCC most common — sensorineural hearing loss, vertigo), facial nerve paralysis, tegmen erosion with intracranial extension (epidural abscess, meningitis, brain abscess, lateral sinus thrombosis). Emergency
Surgical management: Tympanomastoidectomy — either canal wall up (CWU, intact canal wall — lower recurrence visualization but requires second-look surgery in 6-12 months) or canal wall down (CWD — better disease clearance, lower recurrence but requires lifelong mastoid cavity care/water precautions). The goal is complete removal of cholesteatoma matrix and reconstruction of the sound-conducting mechanism (ossiculoplasty).
07 Otosclerosis
Otosclerosis is an autosomal dominant disorder with variable penetrance affecting the otic capsule. Abnormal remodeling of the bony labyrinth leads to fixation of the stapes footplate at the oval window, causing progressive conductive hearing loss. It is the most common cause of CHL in a young adult with a normal TM. Peak onset: 20s-30s. Female-to-male ratio: 2:1. Bilateral in ~80%. Pregnancy may accelerate progression (estrogen effect).
Audiometric Findings
Low-frequency conductive hearing loss (air-bone gap) that progresses. The Carhart notch is a characteristic dip in bone conduction at 2 kHz — an audiometric artifact caused by altered resonance of the ossicular chain, not true sensorineural loss. It disappears after successful stapes surgery. Tympanometry shows Type As (reduced compliance). Absent stapedial reflexes. In advanced cases, cochlear otosclerosis (otospongiosis) produces a mixed hearing loss with sensorineural component.
Treatment
Hearing aids are appropriate for patients declining surgery or with contraindications. Stapedectomy (complete removal of the stapes superstructure and footplate, placement of a prosthesis) or stapedotomy (small fenestra in the footplate with a piston prosthesis — currently preferred, lower complication rate) achieve closure of the air-bone gap to ≤ 10 dB in ~90-95% of cases. Complications: sensorineural hearing loss (1-3%), vertigo, taste disturbance (chorda tympani injury), perilymph fistula, prosthesis displacement. Sodium fluoride has been used to slow cochlear otosclerosis but evidence is limited.
08 Vestibular Disorders
Benign Paroxysmal Positional Vertigo (BPPV)
BPPV is the most common cause of vertigo. Caused by free-floating otoconia (calcium carbonate crystals) displaced from the utricle into a semicircular canal — most commonly the posterior canal (~80-90%). Patients experience brief episodes (< 60 seconds) of intense rotational vertigo triggered by changes in head position (rolling over in bed, looking up, bending down). No hearing loss or tinnitus.
Differential diagnosis of vertigo: Central vertigo (stroke, MS, posterior fossa tumor — associated with other neurologic signs: vertical nystagmus, direction-changing nystagmus, negative head impulse test, inability to walk, new headache) must be distinguished from peripheral vertigo (BPPV, vestibular neuritis, Ménière's — associated with horizontal/torsional nystagmus, positive head impulse test, no neurologic deficits). The HINTS exam (Head Impulse, Nystagmus, Test of Skew) in acute vestibular syndrome is more sensitive than MRI for stroke in the first 24-48 hours. A dangerous (central) pattern: normal head impulse, direction-changing nystagmus, and skew deviation — this patient needs urgent MRI and neurology consultation.
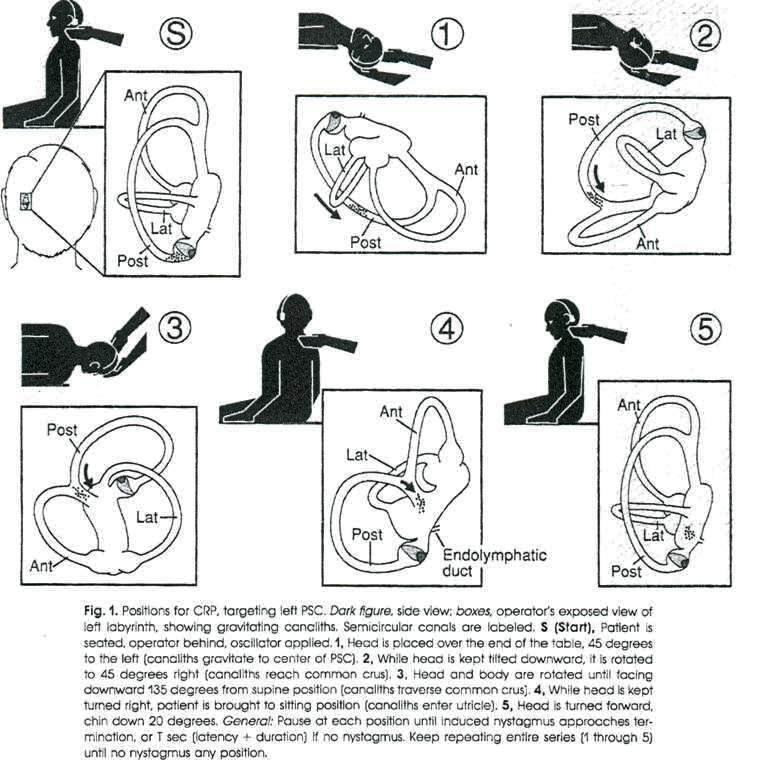
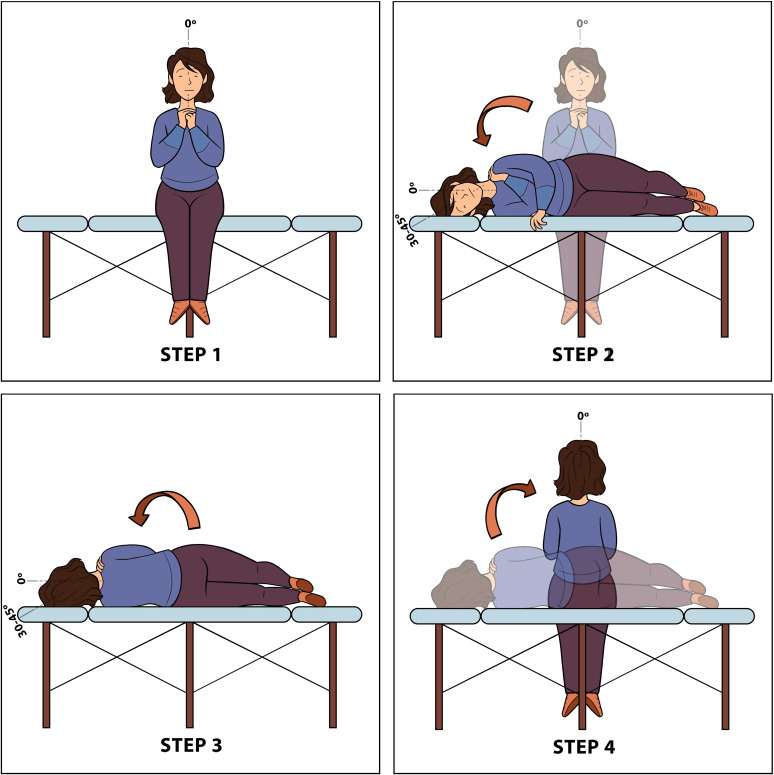
Diagnosis: The Dix-Hallpike test is diagnostic for posterior canal BPPV — the patient is moved from seated to supine with the head turned 45 degrees and extended 20 degrees below the table. A positive test produces upbeating, torsional (toward the affected ear) nystagmus with a latency of 1-5 seconds, lasting < 60 seconds, and fatigable with repeated testing. For horizontal canal BPPV, the supine roll test (Pagnini-McClure) is used — produces horizontal nystagmus.
Treatment: The Epley maneuver (canalith repositioning procedure) for posterior canal BPPV has a success rate of ~80% in a single session and > 90% with repeated maneuvers. The Lempert (BBQ roll) maneuver is used for horizontal canal BPPV. Surgical options for refractory cases: posterior canal occlusion or singular neurectomy.
Ménière's Disease
Ménière's disease is an idiopathic inner ear disorder characterized by endolymphatic hydrops. The classic triad/tetrad: (1) episodic vertigo lasting 20 minutes to 12 hours, (2) fluctuating low-frequency sensorineural hearing loss, (3) tinnitus (roaring), and (4) aural fullness. Diagnosis is clinical (AAO-HNS 1995 criteria: definite Ménière's requires two or more spontaneous vertigo episodes lasting 20 min to 12 hours, audiometrically documented low-to-mid frequency SNHL in the affected ear, and fluctuating aural symptoms — tinnitus, fullness, hearing loss).
Pathophysiology: Endolymphatic hydrops — distension of the endolymphatic compartment (scala media, saccule, utricle) due to either overproduction or impaired absorption of endolymph. Rupture of Reissner's membrane (membrane between scala media and scala vestibuli) allows mixing of potassium-rich endolymph with perilymph, causing toxic depolarization of hair cells and vestibular receptors — this is the proposed mechanism for the acute vertiginous episode.
Treatment ladder: (1) Dietary salt restriction (< 1500-2000 mg/day), avoidance of caffeine and alcohol; (2) Diuretics (hydrochlorothiazide, acetazolamide) — limited evidence; (3) Intratympanic dexamethasone for refractory vertigo; (4) Intratympanic gentamicin (chemical labyrinthectomy — ablates vestibular function, risk of hearing loss in 10-30%); (5) Surgical: endolymphatic sac decompression/shunt, vestibular nerve section, labyrinthectomy (destroys all residual hearing — only for non-serviceable hearing). AAO-HNS CPG: Ménière's Disease (2020) PMID 32267799
Vestibular Neuritis & Labyrinthitis
Vestibular neuritis is acute inflammation of the vestibular nerve (usually superior division), presumably viral. Presents with sudden, severe, continuous vertigo lasting days, with nausea/vomiting, spontaneous horizontal nystagmus beating away from the affected ear, positive head impulse test (catch-up saccade toward the affected side), and no hearing loss. Labyrinthitis has the same presentation but includes sensorineural hearing loss (inflammation involves both vestibular and cochlear portions). Treatment: short-term vestibular suppressants (meclizine, diazepam) for 24-48 hours only, followed by early vestibular rehabilitation. A short course of oral corticosteroids may accelerate recovery.
Superior Semicircular Canal Dehiscence (SSCD)
SSCD (Minor's syndrome) is a bony defect in the superior semicircular canal creating a third mobile window into the inner ear. Symptoms: sound-induced vertigo (Tullio phenomenon), pressure-induced vertigo (Hennebert sign), autophony (hearing one's own voice/heartbeat), pulsatile tinnitus, and conductive hyperacusis. Audiometry may show an air-bone gap at low frequencies (mimicking otosclerosis) with present acoustic reflexes (unlike otosclerosis). VEMP testing shows a low-threshold cervical VEMP. Diagnosis confirmed by high-resolution CT temporal bone (0.5 mm cuts, reformatted in Poschl and Stenvers planes). Treatment for symptomatic patients: middle fossa craniotomy with canal plugging or resurfacing; transmastoid approach is an alternative.
09 Temporal Bone Fractures
Temporal bone fractures occur in ~18-22% of skull fractures, usually from high-energy blunt trauma. Two classification systems are used:
Traditional Classification (Relative to Petrous Ridge)
| Type | Frequency | Fracture Line | Key Features |
|---|---|---|---|
| Longitudinal | 70-80% | Parallel to long axis of petrous bone | Conductive hearing loss (ossicular disruption, TM tear), EAC bleeding/step-off, hemotympanum. Facial nerve injury: 10-25% (usually at geniculate ganglion). CSF otorrhea in 10-20%. |
| Transverse | 10-20% | Perpendicular to long axis of petrous bone | Sensorineural hearing loss (otic capsule disrupted), facial nerve injury: 30-50% (labyrinthine/tympanic segment), vertigo. CSF otorrhea less common (CSF rhinorrhea via ET may occur). |
| Mixed/Oblique | ~10% | Combined pattern | Features of both |
Modern Classification (Otic Capsule Involvement)
This system better predicts clinical outcomes: Otic capsule-sparing (~95%) — fracture line does not cross the cochlea or vestibular labyrinth; typically CHL, lower rate of facial nerve injury and SNHL. Otic capsule-violating (~5%) — fracture line crosses the cochlea or labyrinth; associated with severe/profound SNHL, facial nerve injury in ~50%, and higher CSF leak rate.
Management Priorities
(1) Facial nerve: Immediate-onset complete paralysis may warrant surgical exploration/decompression (within 2-3 weeks); delayed-onset or incomplete paralysis is managed with observation and corticosteroids — prognosis is excellent (90-100% recovery). Electrophysiologic testing (ENoG) within 3-14 days: ≥ 90% degeneration is an indication for surgical decompression. (2) Hearing: CHL often resolves (hemotympanum/effusion) or is surgically correctable (ossiculoplasty). SNHL from otic capsule violation is usually permanent. (3) CSF leak: Most resolve with conservative measures (head elevation, avoidance of straining, lumbar drain if persistent). Surgical repair if persistent > 7-10 days or recurrent meningitis. (4) Cholesteatoma: Entrapped squamous epithelium can develop into a post-traumatic cholesteatoma — long-term follow-up required.
10 Cochlear Implants & Hearing Rehabilitation
Cochlear Implant Candidacy
Cochlear implants (CI) bypass damaged hair cells and directly stimulate the spiral ganglion neurons of the auditory nerve. FDA-approved candidacy criteria have expanded over time. General adult criteria: bilateral severe-to-profound SNHL with limited benefit from appropriately fitted hearing aids (sentence recognition scores ≤ 50% in the ear to be implanted and ≤ 60% in the contralateral or binaural condition). Pediatric criteria: age ≥ 9-12 months (FDA-approved at 9 months), bilateral severe-to-profound SNHL, limited benefit from hearing aids after adequate trial (3-6 months).
Device Components
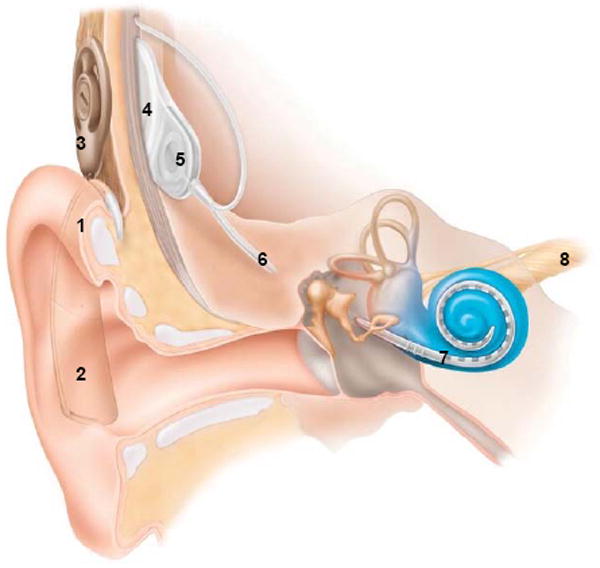
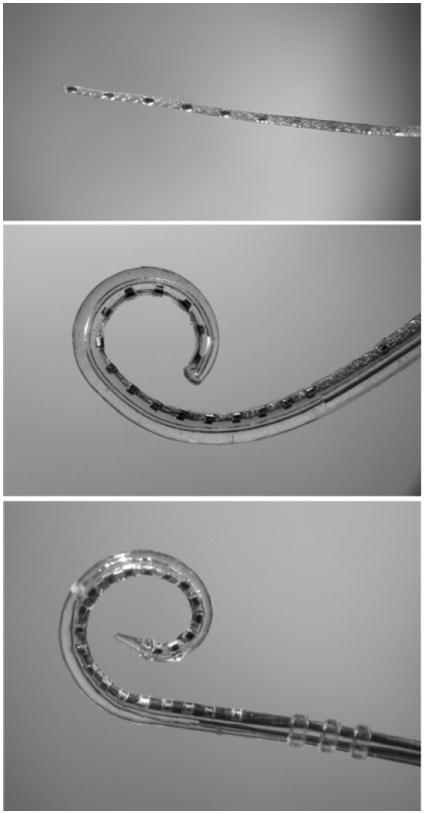
External: Microphone, speech processor, transmitting coil. Internal (surgically implanted): Receiver-stimulator (beneath the skin), electrode array (inserted into the scala tympani of the cochlea). Modern devices have 12-22 intracochlear electrodes. The speech processor converts sound into electrical signals transmitted transcutaneously via the coil to the internal device.
Surgical Considerations
Approach: cortical mastoidectomy followed by posterior tympanotomy (facial recess approach — bounded by the fossa incudis superiorly, facial nerve posteriorly, and chorda tympani anteriorly) to access the round window for electrode insertion. Alternative: round window niche direct approach or suprameatal approach. Atraumatic insertion techniques ("soft surgery") aim to preserve residual hearing for electric-acoustic stimulation (EAS/hybrid devices) — principles include: slow insertion speed, round window insertion (avoids drilling a cochleostomy), use of a thin flexible electrode array, perioperative systemic steroids, and avoidance of suctioning perilymph.
Preoperative assessment: CT temporal bone (cochlear patency, cochlear malformations, mastoid anatomy), MRI (cochlear nerve presence and size — aplasia is a contraindication, hypoplasia is a relative contraindication; cochlear fluid signal confirming patent lumen), comprehensive audiometric evaluation, speech perception testing, hearing aid trial documentation. Complications: Device failure/extrusion (1-5% over 10 years), facial nerve injury (< 1%), meningitis (mitigated by pre-CI and post-CI pneumococcal vaccination — both PCV13 and PPSV23), flap infection, electrode migration/tip fold-over, vertigo/vestibular dysfunction, taste disturbance (chorda tympani injury).
Outcomes & Special Populations
Most post-lingually deafened adults achieve open-set speech recognition scores of 60-80% within 6-12 months. Pre-lingually deafened children implanted early (< 2-3 years) develop age-appropriate speech and language skills. Duration of deafness is the strongest negative predictor of CI outcome. Bilateral implantation provides improved sound localization and speech recognition in noise compared to unilateral implantation.
Single-sided deafness (SSD): CI is now FDA-approved for SSD — restores bilateral hearing, improves speech in noise, and reduces tinnitus. Cochlear malformations: Common cavity deformity, incomplete partition types I-III, enlarged vestibular aqueduct (EVA) — may still receive CI but electrode choice and insertion technique are modified; risk of CSF gusher (perilymph flooding during cochleostomy). Cochlear ossification (post-meningitis): Fibrosis/ossification can occur as early as 2 weeks after bacterial meningitis; CT and MRI assess patency. Early implantation is critical; drill-out technique for partial ossification; split array or double-array for complete ossification.
Other Hearing Rehabilitation
Hearing aids: Behind-the-ear (BTE), in-the-ear (ITE), receiver-in-canal (RIC), completely-in-canal (CIC). Bone-anchored hearing devices (BAHD/BAHA): Utilize bone conduction; indicated for single-sided deafness, conductive/mixed hearing loss with contraindications to conventional aids (chronic ear drainage, aural atresia). Middle ear implants: Vibrant Soundbridge — for moderate-to-severe SNHL or mixed HL when conventional aids are inadequate or contraindicated.
11 Rhinosinusitis
Acute Rhinosinusitis (ARS)
ARS is inflammation of the nasal cavity and paranasal sinuses lasting < 4 weeks. Most cases are viral (common cold) — symptoms include nasal congestion, purulent rhinorrhea, facial pressure/pain, and hyposmia. Acute bacterial rhinosinusitis (ABRS) should be suspected when: (1) symptoms persist ≥ 10 days without improvement, (2) symptoms are severe (fever ≥ 39°C/102°F with purulent discharge or facial pain for ≥ 3 consecutive days), or (3) "double sickening" — worsening after initial improvement. Common pathogens: S. pneumoniae, H. influenzae, M. catarrhalis.
Treatment of ABRS: First-line: amoxicillin-clavulanate (500/125 mg TID or 875/125 mg BID for 5-7 days; high-dose 2 g BID for resistant organisms or recent antibiotic use). Alternatives for penicillin allergy: doxycycline, respiratory fluoroquinolone (levofloxacin, moxifloxacin). Adjunctive: intranasal saline irrigation, intranasal corticosteroids, analgesics. Avoid routine CT imaging for uncomplicated ARS. AAO-HNS CPG: Adult Sinusitis (2015) PMID 25832968
Complications of Acute Sinusitis
Complications are classified as orbital (Chandler classification) or intracranial:
| Chandler Stage | Description | Treatment |
|---|---|---|
| I — Preseptal cellulitis | Infection anterior to the orbital septum; eyelid edema, erythema; no proptosis, no vision changes | IV antibiotics (ampicillin-sulbactam or clindamycin); oral antibiotics if mild |
| II — Orbital cellulitis | Infection posterior to the septum; proptosis, chemosis, limited EOM, pain with eye movement | IV antibiotics; CT scan; ophthalmology consultation; close monitoring of visual acuity |
| III — Subperiosteal abscess | Pus collection between lamina papyracea and periorbita; proptosis, EOM limitation, may have visual compromise | IV antibiotics + surgical drainage (endoscopic approach via ethmoidectomy). Small abscesses in children may trial IV antibiotics first. |
| IV — Orbital abscess | Abscess within orbital fat; severe proptosis, ophthalmoplegia, visual loss | Emergency Urgent surgical drainage + IV antibiotics. Vision loss may be irreversible if not promptly treated. |
| V — Cavernous sinus thrombosis | Bilateral eye findings, CN III/IV/VI palsies, high fever, altered mental status, meningismus | Emergency IV antibiotics, anticoagulation (controversial), surgical drainage of source. High morbidity/mortality. |
Intracranial complications: epidural abscess, subdural empyema, brain abscess (frontal lobe — from frontal sinusitis via retrograde thrombophlebitis of diploic veins), and meningitis. The adolescent male with frontal sinusitis is at particular risk for Pott's puffy tumor — subperiosteal abscess of the frontal bone with overlying forehead swelling due to osteomyelitis of the frontal bone. Treatment requires IV antibiotics, surgical drainage of the intracranial collection, and treatment of the frontal sinus (craniotomy or endoscopic Draf III procedure).
Chronic Rhinosinusitis (CRS)
CRS is defined as sinonasal inflammation lasting ≥ 12 weeks with at least two of: mucopurulent drainage, nasal obstruction, facial pain/pressure, decreased smell. Objective evidence required (endoscopic findings of polyps, edema, mucopurulent discharge, or CT showing mucosal changes). Two major phenotypes:
CRS without nasal polyps (CRSsNP) — predominantly neutrophilic inflammation, Type 1/Type 3 immune response. Associated with anatomic obstruction, biofilm formation. Treatment: maximal medical therapy (intranasal corticosteroids, saline irrigations, culture-directed antibiotics, short courses of oral steroids) followed by FESS if refractory.
CRS with nasal polyps (CRSwNP) — predominantly eosinophilic inflammation, Type 2 immune response (elevated IL-4, IL-5, IL-13). Associated with asthma, aspirin-exacerbated respiratory disease (AERD/Samter's triad: asthma, nasal polyps, aspirin sensitivity), and allergic fungal rhinosinusitis. Higher recurrence rate after surgery. Treatment: intranasal corticosteroids (high-volume budesonide irrigations), short courses of oral steroids, biologic agents for refractory disease (dupilumab — anti-IL-4R; omalizumab — anti-IgE; mepolizumab — anti-IL-5). FESS for refractory cases.
Fungal Rhinosinusitis
| Type | Immune Status | Features | Treatment |
|---|---|---|---|
| Allergic fungal rhinosinusitis (AFRS) | Immunocompetent, atopic | Bent-Kuhn criteria: Type 1 hypersensitivity, nasal polyps, CT with heterogeneous sinus opacification with hyperintense foci, eosinophilic mucin with non-invasive fungal hyphae, positive fungal stain. "Allergic mucin" — thick, peanut butter-like mucus. | FESS (debulk polyps, open sinuses), systemic and topical steroids, immunotherapy. High recurrence. |
| Fungal ball (mycetoma) | Immunocompetent | Dense fungal concretion, usually in maxillary sinus. Unilateral. CT: metallic-density opacification. Non-invasive. | FESS with complete removal. No antifungals needed. |
| Acute invasive fungal sinusitis | Immunocompromised (neutropenic, DM with DKA, transplant) | Emergency Rapidly progressive tissue necrosis. Pale/black eschar on turbinate or palate. Angioinvasion by Aspergillus or Mucor/Rhizopus (mucormycosis). High mortality (50-80%). | Emergent surgical debridement (radical — may require orbital exenteration, maxillectomy), systemic antifungals (amphotericin B lipid formulation for Mucor; voriconazole for Aspergillus), reversal of immunosuppression (G-CSF, insulin for DKA). |
| Chronic invasive fungal sinusitis | Mildly immunosuppressed (diabetes, chronic steroids) | Indolent progression over ≥ 12 weeks. Granulomatous tissue. Orbital or intracranial extension. | Surgical debridement, prolonged antifungal therapy. |
12 Functional Endoscopic Sinus Surgery (FESS)
Indications
FESS is indicated for CRS refractory to maximal medical therapy, recurrent acute sinusitis, complications of sinusitis, mucoceles, fungal sinusitis, sinonasal tumors (biopsy and select resections), CSF leak repair, and orbital decompression. The principle is to restore sinus ventilation and mucociliary clearance by removing obstructing disease while preserving mucosa.
Messerklinger Technique
The Messerklinger approach (anterior-to-posterior dissection) is the standard technique: (1) Uncinectomy — medialize or resect the uncinate process to expose the natural maxillary ostium and ethmoid infundibulum; (2) Maxillary antrostomy — enlarge the natural ostium; (3) Anterior ethmoidectomy — open anterior ethmoid air cells, identify the basal lamella of the middle turbinate; (4) Posterior ethmoidectomy — enter posterior ethmoid cells (behind the basal lamella); (5) Sphenoidotomy — identify and widen the sphenoid ostium (medial to the superior turbinate); (6) Frontal sinusotomy (Draf procedures) — clear the frontal recess (Draf I: simple opening; Draf IIa: removal of cells between the middle turbinate and lamina papyracea; Draf IIb: removal of the frontal sinus floor from the lamina papyracea to the nasal septum; Draf III/modified Lothrop: bilateral frontal sinusotomy with removal of the interfrontal septum and superior nasal septum — creates a single large drainage pathway).
Key Surgical Landmarks
Maxillary ostium: Located in the posterior third of the ethmoid infundibulum — always identify the natural ostium before enlarging (creating a second ostium leads to mucus recirculation). Lamina papyracea: Lateral limit of dissection — breach causes orbital fat herniation, orbital hematoma, or extraocular muscle injury. Skull base (fovea ethmoidalis): Superior limit — identified by the anterior ethmoidal artery (runs in a mesentery at the junction of the lateral lamella and fovea ethmoidalis). Anterior ethmoidal artery: Key landmark — located at the posterior aspect of the frontal recess, marks the approximate level of the cribriform plate.
Complications
| Complication | Mechanism | Management |
|---|---|---|
| Orbital hematoma | Lamina papyracea breach, anterior ethmoidal artery injury | Emergency Lateral canthotomy/cantholysis within 60 minutes if vision threatened; decompression of orbit |
| Nasolacrimal duct injury | Antrostomy anterior to the natural ostium | Observation; DCR if epiphora develops |
| CSF leak | Skull base (fovea ethmoidalis/cribriform plate) breach | Intraoperative repair with mucosal graft, fascial overlay; lumbar drain if large defect |
| Meningitis | Intracranial entry with contamination | IV antibiotics, CSF leak repair |
| Optic nerve injury | Dissection in sphenoid sinus (Onodi cell) or posterior ethmoid | Immediate high-dose IV steroids; surgical decompression if edema |
| ICA injury | Lateral wall of sphenoid sinus breach | Emergency Pack with muscle/hemostatic agents; emergent angiography/embolization. Life-threatening. |
| Synechiae (scarring) | Mucosal apposition between septum and lateral wall | Debridement in clinic; spacers/stents at surgery |
13 Nasal Obstruction
Septal Deviation & Septoplasty
Nasal septal deviation is the most common structural cause of nasal obstruction. The septum consists of cartilage (quadrangular cartilage anteriorly) and bone (perpendicular plate of the ethmoid superiorly, vomer posteroinferiorly). Septoplasty involves submucosal resection/reshaping of the deviated cartilage and bone while preserving a dorsal and caudal L-strut of cartilage (≥ 1.0-1.5 cm width) to maintain nasal support. Complications: septal perforation (most common significant complication, 1-5%), saddle nose deformity (inadequate L-strut), septal hematoma (must be drained immediately to prevent cartilage necrosis and saddle nose — Emergency), CSF leak (rare — cribriform plate injury).
Turbinate Hypertrophy
Inferior turbinate hypertrophy (mucosal hypertrophy from allergic rhinitis, vasomotor rhinitis, or chronic inflammation) contributes to nasal obstruction. Medical treatment: intranasal corticosteroids, antihistamines, decongestants (short-term), allergen avoidance. Surgical options: submucosal reduction (radiofrequency ablation, microdebrider submucosal resection), outfracture (lateralize the turbinate), or partial turbinectomy. Avoid total inferior turbinectomy — risk of empty nose syndrome (paradoxical nasal obstruction despite wide-open nasal cavity, with dryness, crusting, and subjective dyspnea).
Nasal Valve Collapse
The internal nasal valve (angle between the upper lateral cartilage and septum, normally 10-15 degrees) is the narrowest portion of the nasal airway, accounting for approximately half of the total nasal resistance. Collapse or stenosis is a common and underdiagnosed cause of nasal obstruction. The external nasal valve (nasal ala, lateral crus of the lower lateral cartilage, columella) — weakness or collapse causes visible alar collapse during inspiration, especially during exercise.
Diagnosis: Cottle maneuver (lateral traction on the cheek to open the valve — relief of obstruction suggests valve pathology) and modified Cottle test (placement of a Cottle elevator or curette to support the valve internally — more specific than Cottle). Subjective and objective improvement confirm the diagnosis.
Treatment: Internal nasal valve: Spreader grafts (cartilage grafts placed between the upper lateral cartilage and septum to widen the valve angle — the gold standard), spreader flaps (autospreader technique using the upper lateral cartilage itself), butterfly graft, nasal valve suspension sutures, or absorbable implants (Latera). External nasal valve: Alar batten grafts (cartilage grafts placed lateral to and over the lateral crura to provide structural support), lateral crural strut grafts, alar rim grafts. Functional rhinoplasty combines aesthetic and functional goals when both are indicated.
Allergic Rhinitis
Allergic rhinitis is IgE-mediated nasal inflammation triggered by inhaled allergens (dust mites, pollen, mold, animal dander). Affects 10-30% of adults and up to 40% of children. Presents with nasal congestion, rhinorrhea (watery, clear), sneezing, nasal pruritus, and postnasal drip. Examination: pale, boggy (edematous) inferior turbinates, clear rhinorrhea, "allergic shiners" (infraorbital darkening), "allergic salute" (transverse nasal crease from repeated wiping). Diagnosis: clinical history and positive skin prick testing or specific IgE levels. Treatment: allergen avoidance, intranasal corticosteroids (most effective single agent — fluticasone, mometasone, budesonide), second-generation oral antihistamines (loratadine, cetirizine, fexofenadine), intranasal antihistamines (azelastine), leukotriene receptor antagonists (montelukast — less effective than intranasal steroids), allergen immunotherapy (subcutaneous — SCIT, or sublingual — SLIT) for patients refractory to pharmacotherapy.
14 Epistaxis
Anterior Epistaxis
Anterior epistaxis accounts for ~90% of nosebleeds and arises from Kiesselbach's plexus (Little's area) on the anterior nasal septum — an anastomotic network of the anterior ethmoidal artery, sphenopalatine artery, greater palatine artery, and superior labial artery (from the facial artery). Management: (1) Direct pressure (pinch the alar cartilages for 15-20 minutes, lean forward), (2) Topical vasoconstriction (oxymetazoline 0.05%), (3) Chemical cautery (silver nitrate), (4) Anterior nasal packing (Merocel sponge, Rapid Rhino, or ribbon gauze with petroleum) if cautery fails.
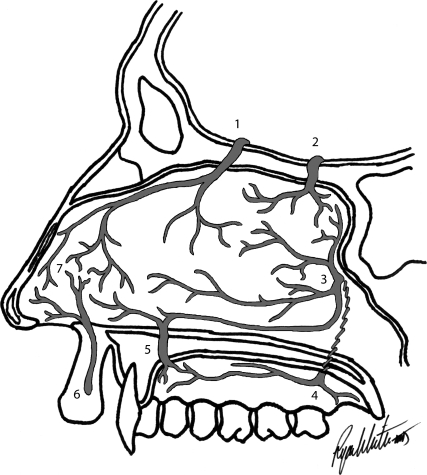
Posterior Epistaxis
Posterior epistaxis arises from branches of the sphenopalatine artery or Woodruff's plexus (posterolateral nasal wall, inferior to the posterior end of the middle turbinate). More common in elderly patients with hypertension and atherosclerosis. Higher risk of hemodynamic compromise. Management: (1) Posterior packing (Foley catheter balloon, epistaxis balloon catheter — double-balloon system), (2) Endoscopic sphenopalatine artery ligation (ESPAL) — definitive treatment, success rate > 90%, (3) Anterior ethmoidal artery ligation (via external approach if bleeding from the ethmoidal distribution), (4) Angiographic embolization of the internal maxillary artery (reserved for refractory cases or when surgical ligation is not feasible).
Epistaxis Management Algorithm
Step 1: Assess hemodynamic stability — resuscitate if needed (IV access, fluid, type and cross). Step 2: Attempt to localize the bleeding site (anterior rhinoscopy after clearing clot with suction/nose blowing, topical vasoconstriction). Step 3: If anterior source identified: silver nitrate cautery (one side of septum only per session to avoid septal perforation). Step 4: If cautery fails or source not seen: anterior nasal packing. Step 5: If anterior packing fails (continued bleeding posteriorly): posterior packing or epistaxis balloon + admit to monitored bed. Step 6: If packing fails: endoscopic sphenopalatine artery ligation (ESPAL) in the OR, or angiographic embolization. Always assess for anticoagulant/antiplatelet use, coagulopathy, hypertension, and hereditary hemorrhagic telangiectasia.
Special Considerations
Hereditary hemorrhagic telangiectasia (HHT/Osler-Weber-Rendu): Autosomal dominant disorder causing mucocutaneous telangiectasias and arteriovenous malformations (AVMs). Recurrent epistaxis is the most common presentation. Management of nasal bleeding: humidification, topical estrogen, laser cautery (KTP), septodermoplasty, bevacizumab (anti-VEGF, systemic or topical), Young's procedure (nasal closure — last resort). Screen for pulmonary and hepatic AVMs. Anticoagulant-associated epistaxis: Balance bleeding management with thrombotic risk — reversal agents only for life-threatening hemorrhage; consult the prescribing team regarding temporary cessation.
15 Sinonasal Tumors
Inverted Papilloma
Inverted papilloma is the most common benign sinonasal tumor. It arises from the lateral nasal wall (most commonly at the middle meatus/ethmoid region) and has a characteristic endophytic growth pattern (Schneiderian epithelium inverts into the underlying stroma). Key features: unilateral nasal obstruction and rhinorrhea; associated with SCC in 5-15% (must be thoroughly sampled histologically). HPV types 6, 11, 16, 18 have been implicated. Staging: Krouse classification (T1: limited to nasal cavity; T2: involving ostiomeatal complex and ethmoid; T3: extending to maxillary, frontal, or sphenoid sinus; T4: extending outside the sinuses or with malignancy). Treatment: complete surgical excision with attachment site identification and drilling — endoscopic medial maxillectomy is standard for lateral wall tumors. Recurrence rate: 5-15% with modern endoscopic techniques.
Sinonasal Squamous Cell Carcinoma
The most common sinonasal malignancy. Most arise in the maxillary sinus. Risk factors: occupational exposures (woodworking — strong association with adenocarcinoma of the ethmoids; nickel refining — SCC; leather tanning), smoking, HPV (types 16, 18 — associated with a subset of sinonasal SCC with better prognosis). Presentation: unilateral nasal obstruction, epistaxis, facial pain/swelling, cranial neuropathies (advanced — infraorbital nerve involvement causes V2 numbness/pain; orbital invasion causes diplopia, proptosis). Trotter's triad (conductive hearing loss, ipsilateral palatal fixation, and trigeminal neuralgia) suggests nasopharyngeal or infratemporal fossa involvement.
Treatment: Surgical resection (endoscopic for select tumors confined to the nasal cavity/ethmoid sinuses; open approaches — infrastructure maxillectomy, total maxillectomy, craniofacial resection for skull base involvement) with adjuvant radiotherapy for advanced disease (T3-T4, positive margins, perineural invasion, nodal disease). Elective neck treatment is generally not indicated for sinonasal tumors (nodal metastasis rate is low, ~10-15%). Prognosis depends on stage at presentation — 5-year survival ranges from 60-80% for early-stage to 20-40% for advanced disease. Often diagnosed late due to nonspecific symptoms mimicking chronic sinusitis.
Other Sinonasal Malignancies
Sinonasal undifferentiated carcinoma (SNUC): Highly aggressive, poorly differentiated tumor. Presents with rapid growth, early skull base and orbital invasion. Treatment: induction chemotherapy followed by surgery and radiation (trimodality). Poor prognosis. Sinonasal mucosal melanoma: Rare (< 1% of melanomas); aggressive biology with high local recurrence and distant metastasis rates. Treatment: wide excision (endoscopic for select cases) with adjuvant radiation. 5-year survival: ~20-30%. Adenoid cystic carcinoma: Most common sinonasal minor salivary gland malignancy. Perineural invasion is characteristic. Slow but relentless growth; late distant metastases.
Esthesioneuroblastoma (Olfactory Neuroblastoma)
Esthesioneuroblastoma arises from the olfactory neuroepithelium at the cribriform plate. Bimodal age distribution (2nd and 6th decades). Staging: Kadish classification (A: limited to nasal cavity; B: nasal cavity and paranasal sinuses; C: extension beyond nasal cavity and sinuses — orbit, skull base, intracranial, cervical nodes). Modified Kadish adds stage D for distant metastasis. Hyams histologic grading (I-IV) predicts prognosis. Treatment: craniofacial resection (combined neurosurgical-otolaryngologic approach, increasingly performed endoscopically) with adjuvant radiotherapy. Cervical metastases present in 5-20% at diagnosis.
Juvenile Nasopharyngeal Angiofibroma (JNA)
JNA is a benign but locally aggressive vascular tumor arising from the sphenopalatine foramen. Occurs almost exclusively in adolescent males. Presents with unilateral nasal obstruction, recurrent epistaxis, and a smooth, lobulated, vascular mass on examination (do NOT biopsy in clinic — risk of catastrophic hemorrhage). CT shows Holman-Miller sign (anterior bowing of the posterior wall of the maxillary sinus). MRI delineates intracranial extension. Treatment: preoperative embolization (24-72 hours before surgery) followed by surgical excision (endoscopic for smaller tumors; open approaches — midfacial degloving, lateral rhinotomy, infratemporal fossa — for extensive tumors). Radiotherapy reserved for unresectable disease.
16 Voice Disorders & Vocal Fold Paralysis
Vocal Fold Paralysis
Unilateral vocal fold paralysis (UVFP) results from injury to the recurrent laryngeal nerve. Most common causes: iatrogenic (thyroidectomy — most common surgical cause, followed by anterior cervical spine surgery, carotid endarterectomy, cardiothoracic surgery), malignancy (lung, thyroid, esophageal, skull base tumors), idiopathic (viral neuritis — 20-30%, often recovers), and neurologic (stroke, MS). The paralyzed fold assumes a paramedian position. Symptoms: breathy, weak voice; aspiration; ineffective cough.
Evaluation: Flexible laryngoscopy (assess fold position and mobility), CT from skull base to aortic arch (entire course of the vagus/RLN) to rule out compressive lesion, laryngeal EMG (at 4-6 weeks — helps predict recovery; fibrillation potentials and absent motor unit potentials indicate denervation; polyphasic motor units suggest reinervation).
Treatment: Observation for 6-12 months if etiology is expected to recover (idiopathic, post-viral). Injection laryngoplasty (office-based or operative) with temporary fillers (hyaluronic acid, carboxymethylcellulose — lasts 2-4 months) for early medialization while awaiting recovery, or permanent materials (calcium hydroxylapatite — Radiesse Voice). Medialization thyroplasty (Type I thyroplasty) — insertion of a silicone or Gore-Tex implant through a window in the thyroid cartilage to medialize the paralyzed fold; performed under local anesthesia with voice monitoring. Arytenoid adduction — rotates the arytenoid to close a posterior glottic gap; often combined with thyroplasty. Laryngeal reinervation (ansa cervicalis to RLN anastomosis) — restores tone and bulk to the thyroarytenoid muscle; increasingly used as primary treatment.
Bilateral Vocal Fold Paralysis
Bilateral vocal fold paralysis (BVFP) typically presents with airway compromise (stridor, dyspnea) rather than voice problems — the folds are in the median or paramedian position, maintaining a near-normal voice but a critically narrow airway. Emergency Most common cause: bilateral RLN injury during thyroidectomy. Management: emergent tracheotomy for acute airway compromise; definitive procedures include posterior cordotomy (endoscopic laser), arytenoidectomy, or posterior costal cartilage graft laryngoplasty — all trade voice quality for airway.
Other Voice Disorders
Vocal fold nodules: Bilateral, symmetric, at the junction of the anterior one-third and posterior two-thirds of the vocal fold (point of maximal vibration). Caused by vocal abuse/misuse. Treatment: voice therapy (first-line, curative in most cases). Vocal fold polyps: Usually unilateral, broader-based; may require surgical excision (microlaryngoscopy) if voice therapy fails. Reinke's edema: Bilateral, diffuse polypoid degeneration of the superficial lamina propria; strongly associated with smoking. Treatment: smoking cessation, voice therapy, microlaryngoscopy with debulking. Laryngeal papillomatosis: Recurrent, HPV 6/11-driven, warty growths on the vocal folds/larynx; most common benign laryngeal tumor in children; treated with repeated debulking (microdebrider, laser); adjuvant therapies for aggressive disease include bevacizumab (anti-VEGF — increasingly used as first-line adjuvant, reduces surgical frequency by 50-80%), intralesional cidofovir (antiviral — efficacy debated), and systemic interferon (historical). Malignant transformation to SCC occurs in 2-5%. HPV vaccination may reduce future incidence.
Spasmodic dysphonia: A focal laryngeal dystonia. Adductor type (most common — 85%): involuntary hyperadduction of the vocal folds during phonation producing a strained, strangled, effortful voice with phonatory breaks. Abductor type: Involuntary abduction during phonation producing a breathy voice. Diagnosis: perceptual voice assessment and laryngoscopic evaluation showing task-specific laryngeal spasm. Treatment: botulinum toxin injection into the thyroarytenoid muscle (adductor type) or posterior cricoarytenoid muscle (abductor type) — highly effective but requires repeat injections every 3-6 months. Selective laryngeal adductor denervation-reinervation (Berke procedure) is a surgical alternative offering long-term benefit.
Sulcus vocalis: A groove or invagination in the medial edge of the vocal fold where the epithelium is adherent to the vocal ligament, obliterating the superficial lamina propria (Reinke's space). Causes stiffness of the vocal fold with decreased mucosal wave on stroboscopy and a characteristic thin, breathy, high-pitched voice. Treatment is challenging: medialization procedures to improve glottic closure (injection laryngoplasty, type I thyroplasty), or surgical approaches to release the sulcus (incision, elevation, and grafting of the superficial lamina propria).
17 Laryngeal Cancer
Epidemiology & Risk Factors
Laryngeal squamous cell carcinoma accounts for ~1% of all malignancies. Major risk factors: tobacco smoking (most important), alcohol (synergistic with tobacco), HPV (less significant than in oropharynx). Male predominance (4:1). Three subsites with distinct behavior:
| Subsite | Frequency | Lymphatics | Presentation |
|---|---|---|---|
| Glottic | ~60% | Poor lymphatic drainage — nodal metastasis rare in early-stage | Early hoarseness (detected early); stridor in advanced disease |
| Supraglottic | ~30% | Rich bilateral lymphatic drainage — nodal metastasis common (40-60% at presentation) | Throat pain, dysphagia, otalgia (referred via CN X), late hoarseness |
| Subglottic | ~5% | Paratracheal/mediastinal nodes | Stridor, dyspnea; rare and usually diagnosed late |
TNM Staging (AJCC 8th Edition — Glottic)
| T Stage | Description |
|---|---|
| T1a | Limited to one vocal fold, normal mobility |
| T1b | Involves both vocal folds, normal mobility |
| T2 | Extends to supraglottis/subglottis, or impaired fold mobility |
| T3 | Limited to larynx with vocal fold fixation, or invasion of paraglottic space, or inner cortex of thyroid cartilage |
| T4a | Invasion through thyroid cartilage, or beyond larynx (trachea, esophagus, tongue base, thyroid, strap muscles) |
| T4b | Invasion of prevertebral space, mediastinal structures, or encases carotid artery |
Treatment
Early glottic cancer (T1-T2, N0): Equally effective treatment with either radiation therapy (cure rate 85-95% for T1, 70-80% for T2) or transoral laser microsurgery (TLM)/endoscopic cordectomy — voice quality may be better with radiation for T1a; TLM preferred by some for small T1 lesions. Early supraglottic cancer: Radiation or transoral robotic surgery (TORS)/supraglottic laryngectomy.
Advanced laryngeal cancer (T3-T4): Organ preservation with concurrent chemoradiation (cisplatin + radiation — based on the VA Laryngeal Cancer Study and RTOG 91-11 trial) is the standard for T3 and select T4a tumors. Total laryngectomy for very advanced T4a, failed chemoradiation (salvage laryngectomy), or recurrence. Total laryngectomy creates a permanent tracheostomy and separation of the airway from the pharynx — eliminates aspiration. Voice rehabilitation: tracheoesophageal puncture (TEP) with voice prosthesis (most common, best voice quality), esophageal speech, or electrolarynx. RTOG 91-11 Trial: Organ Preservation in Laryngeal Cancer PMID 14645423
Neck Management in Laryngeal Cancer
Glottic cancer: Early-stage (T1-T2, N0) — the neck is NOT electively treated due to the sparse lymphatics of the true vocal folds. Advanced glottic cancer with supraglottic or subglottic extension: selective neck dissection (levels II-IV) or irradiation of the neck. Supraglottic cancer: The rich bilateral lymphatic drainage means the neck must be addressed even in N0 disease — bilateral selective neck dissection (levels II-IV) or bilateral neck irradiation. N+ disease: Modified radical or selective neck dissection with adjuvant radiation ± chemotherapy (cisplatin added for extranodal extension or positive margins per EORTC 22931/RTOG 9501).
Conservation Laryngeal Surgery
Vertical partial laryngectomy (hemilaryngectomy): For select T1-T2 glottic tumors with limited subglottic extension and normal contralateral fold mobility. Supraglottic laryngectomy: For supraglottic tumors without vocal fold fixation or tongue base invasion — requires adequate pulmonary reserve (FEV1 > 50%) as temporary aspiration is expected. Supracricoid partial laryngectomy: Removes thyroid cartilage, both true and false folds, and the preepiglottic space while preserving at least one arytenoid, the cricoid, and the hyoid — allows decannulation and laryngeal voice.
18 Airway Management & Subglottic Stenosis
Tracheotomy
Tracheotomy is the creation of a surgical opening in the anterior tracheal wall for insertion of a tracheostomy tube. Indications: prolonged mechanical ventilation, upper airway obstruction, secretion management, pulmonary toilet, and as part of head and neck surgical procedures. Surgical tracheotomy: Horizontal skin incision, division of strap muscles in the midline, identification of the trachea, incision between tracheal rings 2-3 or 3-4 (avoid ring 1 — risk of subglottic stenosis; cricothyroid membrane is for emergency cricothyrotomy). Percutaneous dilatational tracheotomy (PDT): Modified Seldinger technique under bronchoscopic guidance; suitable for elective ICU patients. Complications: hemorrhage (innominate artery erosion — late, catastrophic), pneumothorax, tube displacement/false passage, tracheoesophageal fistula, subglottic/tracheal stenosis, stomal infection.
Cricothyrotomy
Cricothyrotomy is an emergency surgical airway created through the cricothyroid membrane. Emergency Indicated when endotracheal intubation fails or is impossible ("can't intubate, can't oxygenate" scenario). Technique: palpate the cricothyroid membrane (between thyroid and cricoid cartilages), horizontal stab incision, insert bougie/tube. Should be converted to a formal tracheotomy within 24-72 hours if continued surgical airway is needed. Contraindicated in children < 12 years (needle cricothyrotomy with jet ventilation preferred in pediatric emergencies due to small cricothyroid membrane).
Subglottic Stenosis
Subglottic stenosis (SGS) is narrowing of the airway from the vocal folds to the lower border of the cricoid. Most common cause in adults: prolonged endotracheal intubation (pressure necrosis of the mucosa, perichondritis, granulation, fibrosis). Other causes: granulomatosis with polyangiitis (GPA/Wegener's), idiopathic, post-tracheotomy, external trauma. Graded by the Cotton-Myer classification:
| Grade | % Obstruction | Management |
|---|---|---|
| I | 0-50% | Observation or endoscopic dilation |
| II | 51-70% | Endoscopic dilation ± laser, steroid injection |
| III | 71-99% | Laryngotracheal reconstruction (LTR) or cricotracheal resection (CTR) |
| IV | 100% (no lumen) | Cricotracheal resection |
Laryngotracheal reconstruction (LTR): Expands the airway lumen using autologous cartilage grafts (rib cartilage) placed anteriorly and/or posteriorly in the split cricoid lamina. Cricotracheal resection (CTR): Excision of the stenotic segment and primary reanastomosis; higher success rate (~95%) than LTR for Grade III-IV stenosis but greater surgical complexity. Endoscopic management: Balloon dilation, laser excision, steroid injection (triamcinolone 40 mg/mL), mitomycin-C application — used for mild stenosis or as temporizing measures.
19 Dysphagia & Esophageal Disorders
Evaluation of Dysphagia
Fiberoptic Endoscopic Evaluation of Swallowing (FEES): Flexible laryngoscopy performed during swallowing of dyed food/liquid boluses. Assesses laryngeal sensation, pharyngeal residue, penetration, and aspiration. Advantages: portable (bedside), no radiation. Modified Barium Swallow (MBS/VFSS): Videofluoroscopic swallowing study — the patient swallows barium-coated boluses of various consistencies under fluoroscopy. Assesses oral preparatory phase, oral transit, pharyngeal transit, laryngeal closure, UES opening, and aspiration/penetration. Penetration-Aspiration Scale (Rosenbek): 1 (material does not enter the airway) to 8 (silent aspiration — material enters below the vocal folds with no cough response).
Cricopharyngeal Dysfunction
The cricopharyngeus muscle (upper esophageal sphincter) may fail to relax adequately during swallowing, causing dysphagia for solids, globus sensation, and aspiration. Diagnosis: MBS showing a persistent posterior bar at the cricopharyngeus level, or esophageal manometry showing incomplete UES relaxation. Treatment: cricopharyngeal myotomy (endoscopic using laser or stapler, or open transcervical approach), Botox injection (temporary, diagnostic/therapeutic).
Zenker's Diverticulum
Zenker's diverticulum is a posterior pulsion diverticulum that herniates through Killian's dehiscence (between the thyropharyngeus and cricopharyngeus muscles). Most common in elderly males. Symptoms: progressive dysphagia, regurgitation of undigested food, halitosis, aspiration, gurgling voice. Diagnosis: barium swallow (definitive — shows a posterior midline pouch). Do NOT perform esophagoscopy as the primary diagnostic study — risk of perforation. Treatment: endoscopic stapler-assisted diverticulotomy (divides the common wall between the diverticulum and esophagus, simultaneously performing a cricopharyngeal myotomy) — preferred for smaller diverticula. Open transcervical diverticulectomy with cricopharyngeal myotomy for large diverticula (> 3-5 cm).
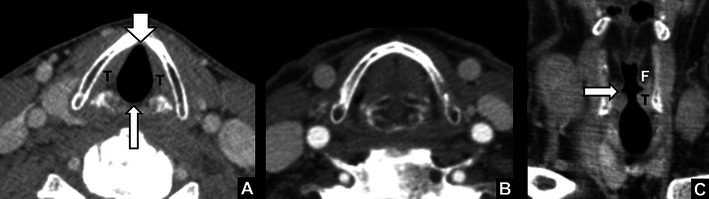
20 Pediatric Airway
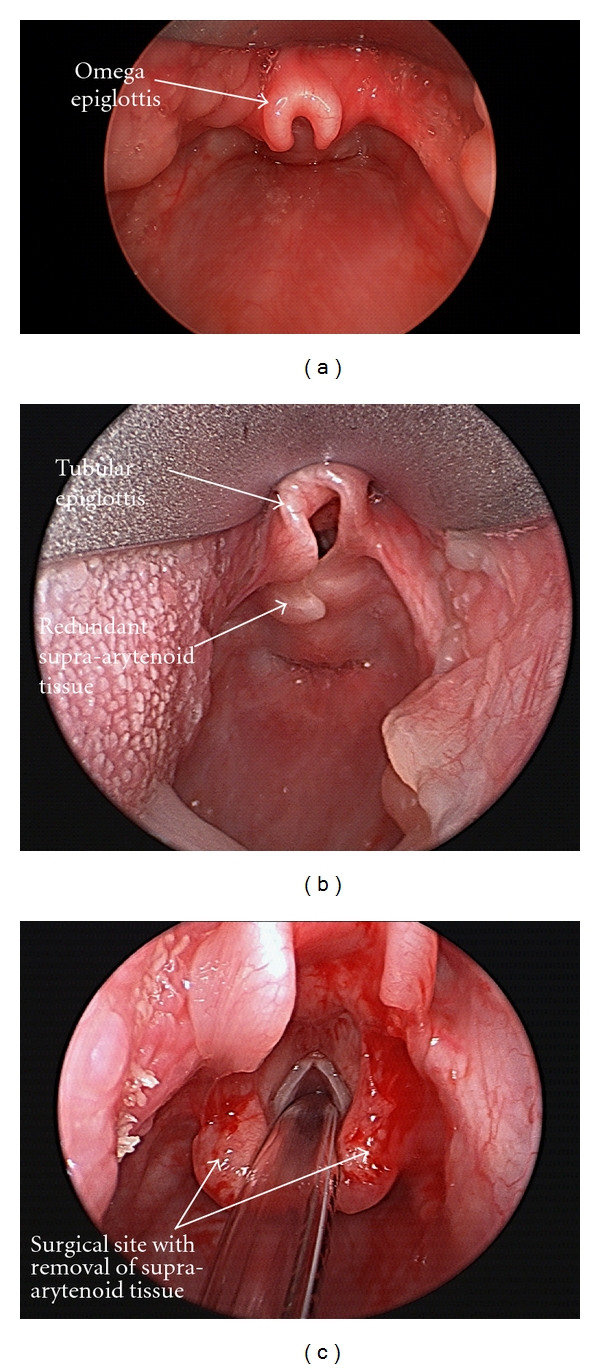
Laryngomalacia
Laryngomalacia is the most common cause of stridor in neonates. Caused by collapse of supraglottic structures (omega-shaped/tubular epiglottis, shortened aryepiglottic folds, redundant arytenoid mucosa) during inspiration. Presents with inspiratory stridor that worsens with feeding, crying, and supine position. Diagnosis: flexible laryngoscopy showing prolapse of supraglottic structures on inspiration. Most cases (~90%) are self-limiting, resolving by 12-18 months. Severe cases (failure to thrive, cyanotic episodes, cor pulmonale) require supraglottoplasty (endoscopic division of shortened aryepiglottic folds, trimming of redundant arytenoid mucosa).
Subglottic Hemangioma
Subglottic hemangioma is the most common subglottic tumor in infants. Presents at 4-6 weeks of age with biphasic stridor (with an expiratory component) that worsens as the hemangioma grows during the proliferative phase (peaks at 6-10 months). Associated with cutaneous hemangiomas in 50% (especially "beard distribution" — mandible, chin, neck). Diagnosis: flexible laryngoscopy/direct laryngoscopy showing a submucosal, compressible mass — do NOT biopsy (risk of hemorrhage). Treatment: propranolol (2-3 mg/kg/day) is first-line — dramatically effective, has transformed management. Alternatives for refractory cases: systemic corticosteroids, endoscopic laser ablation, open excision, tracheotomy (rarely needed now).
Croup (Laryngotracheobronchitis)
Croup is viral infection (parainfluenza types 1 and 3 most common) causing subglottic edema. Peak age: 6 months to 3 years. Presents with barking cough, inspiratory stridor, hoarse voice, and low-grade fever. Steeple sign on AP neck radiograph (subglottic narrowing). Treatment: single dose of dexamethasone (0.6 mg/kg PO/IM) for all severities; nebulized racemic epinephrine (0.5 mL of 2.25% solution) for moderate-severe stridor — observe for 2-4 hours after administration (rebound possible). Rarely requires intubation.
Epiglottitis
Epiglottitis (supraglottitis) is acute bacterial infection of the epiglottis and supraglottic structures. Emergency Previously caused by Haemophilus influenzae type b (now rare in vaccinated populations); current pathogens include Streptococcus species, Staphylococcus aureus. Children present with acute high fever, dysphagia, drooling, muffled voice, and toxic appearance — prefer sitting upright and leaning forward ("tripod" or "sniffing" position). Thumb sign on lateral neck radiograph (swollen epiglottis). Management: secure the airway FIRST (in the OR with ENT/anesthesia available for possible emergent tracheotomy); IV antibiotics (ceftriaxone or ampicillin-sulbactam); do NOT examine the throat or lay the child supine before the airway is secured.
Foreign Body Aspiration
Airway foreign body is most common in children 1-3 years (peanuts and other foods are the most common objects). Presents with sudden-onset choking, coughing, and wheezing. A classic triad of unilateral wheezing, coughing, and decreased breath sounds is present in only a minority of cases. The right mainstem bronchus is the most common site (shorter, wider, more vertical angle of takeoff). Radiologic findings: unilateral hyperinflation (air trapping due to ball-valve effect — best seen on expiratory or bilateral decubitus films), mediastinal shift away from the affected side on expiratory films, atelectasis if complete obstruction. Chest radiograph may be normal in up to 20-30% of cases — a high index of clinical suspicion warrants bronchoscopy even with negative imaging.
Diagnosis and treatment: Rigid bronchoscopy is both diagnostic and therapeutic (gold standard for removal). It provides the largest working channel, best airway control, and ability to ventilate while working. Optical grasping forceps are used for extraction. Flexible bronchoscopy may assist with diagnosis or be used in adults but rigid bronchoscopy provides superior instrument access for pediatric cases. Complications of retained foreign body: recurrent pneumonia, bronchiectasis, lung abscess, erosion into adjacent structures. Esophageal foreign body: Button batteries lodged in the esophagus are a surgical emergency (Emergency) — liquefaction necrosis occurs within 2 hours. Requires emergent removal by rigid esophagoscopy. Coins are the most common esophageal foreign body in children — coins in the esophagus orient in the coronal plane on AP radiograph (vs tracheal foreign bodies which orient in the sagittal plane).
21 Thyroid Surgery
Indications for Thyroidectomy
Indications for thyroid surgery include: malignancy or suspicious cytology (Bethesda V-VI), indeterminate cytology (Bethesda III-IV) for definitive diagnosis, compressive goiter (dyspnea, dysphagia), substernal goiter, hyperthyroidism refractory to medical therapy (Graves' disease, toxic multinodular goiter, toxic adenoma), and patient preference.
Bethesda System for Reporting Thyroid Cytopathology
| Category | Diagnosis | Risk of Malignancy | Recommended Action |
|---|---|---|---|
| I | Non-diagnostic/Unsatisfactory | 5-10% | Repeat FNA with US guidance |
| II | Benign | 0-3% | Follow-up; repeat US in 12-24 months |
| III | Atypia of undetermined significance (AUS) / Follicular lesion of undetermined significance (FLUS) | 10-30% | Repeat FNA, molecular testing, or diagnostic lobectomy |
| IV | Follicular neoplasm / Suspicious for follicular neoplasm | 25-40% | Diagnostic lobectomy (or molecular testing) |
| V | Suspicious for malignancy | 50-75% | Lobectomy or total thyroidectomy |
| VI | Malignant | 97-99% | Total thyroidectomy (for most > 1 cm PTC/FTC) or lobectomy (for < 1 cm, low-risk) |
Thyroid Cancer Types
| Type | Frequency | Features | Prognosis |
|---|---|---|---|
| Papillary thyroid carcinoma (PTC) | 80-85% | Most common thyroid malignancy. Orphan Annie eye nuclei, psammoma bodies, papillary architecture. Lymph node metastasis common (30-80%) but has minimal impact on survival in younger patients. Spread via lymphatics. | Excellent (10-year survival > 95%) |
| Follicular thyroid carcinoma (FTC) | 10-15% | Requires capsular/vascular invasion for diagnosis (cannot be diagnosed on FNA alone). Spread via hematogenous route (bone, lung). Less frequent nodal metastasis than PTC. | Good (10-year survival ~85-90%) |
| Medullary thyroid carcinoma (MTC) | 3-5% | Arises from parafollicular C-cells. Produces calcitonin (tumor marker) and CEA. 25% familial (MEN 2A: MTC + pheochromocytoma + hyperparathyroidism; MEN 2B: MTC + pheochromocytoma + marfanoid habitus + mucosal neuromas). RET proto-oncogene mutation. Total thyroidectomy with central +/- lateral neck dissection. NOT radioiodine responsive. | Intermediate (10-year survival ~75-85%) |
| Anaplastic thyroid carcinoma | < 2% | Most aggressive thyroid cancer. Typically elderly patients with rapidly enlarging neck mass, compressive symptoms. May arise from dedifferentiation of well-differentiated thyroid cancer. Locally invasive, distant metastases common. | Dismal (median survival 3-6 months) |
Surgical Technique & Key Principles
(1) RLN identification and preservation: The RLN is identified in the tracheoesophageal groove or at Berry's ligament (posterior suspensory ligament of the thyroid — highest risk area for RLN injury). Intraoperative nerve monitoring (IONM) is widely used as an adjunct (not a substitute) for direct visualization. Loss of signal during IONM on the first side may prompt aborting contralateral dissection to prevent bilateral RLN injury. (2) Parathyroid preservation: At least two parathyroid glands should be preserved with their blood supply. If a gland is devascularized or inadvertently removed, it should be confirmed histologically (touch-prep/frozen section) and autotransplanted (minced into 1 mm pieces and implanted into the SCM or forearm muscle). (3) External branch of the SLN: Identify and preserve during superior pole ligation by ligating individual branches of the superior thyroid artery close to the thyroid capsule. (4) Hemostasis: Energy devices (harmonic scalpel, LigaSure) have simplified vessel ligation.
Complications of Thyroidectomy
RLN injury: Temporary (3-5%), permanent (0.5-2%). Hypoparathyroidism: Temporary (20-30% after total thyroidectomy), permanent (1-4%) — monitor ionized calcium postoperatively; symptomatic hypocalcemia (perioral tingling, Chvostek/Trousseau signs, tetany) requires IV calcium gluconate. Hematoma: Post-thyroidectomy neck hematoma causing airway compromise is a surgical emergency (Emergency) — open the wound at the bedside immediately (remove skin clips/sutures, evacuate hematoma) before returning to the OR. Seroma, wound infection (uncommon). Hypothyroidism (expected after total thyroidectomy — initiate levothyroxine replacement at 1.6 mcg/kg/day; after lobectomy, ~20-30% of patients require supplementation — check TSH at 6-8 weeks). Chyle leak: Rare complication of lateral neck dissection (thoracic duct injury on the left); presents as milky drain output increasing with oral fat intake; management: low-fat/MCT diet, pressure dressing, octreotide; surgical exploration for persistent high-output leaks (> 600 mL/day). Horner syndrome: Rare (sympathetic chain injury during level IV dissection) — ipsilateral ptosis, miosis, anhidrosis.
22 Parathyroid Surgery
Primary Hyperparathyroidism (PHPT)
PHPT is the most common cause of hypercalcemia in the outpatient setting. Caused by a single adenoma (~85%), multigland hyperplasia (~10-15%), double adenoma (~4%), or rarely carcinoma (< 1%). Biochemistry: elevated serum calcium with inappropriately normal or elevated PTH. Symptoms: "stones, bones, moans, groans, and psychiatric overtones" (nephrolithiasis, osteoporosis, abdominal pain/constipation, neuromuscular weakness, depression/cognitive changes). Many patients are asymptomatic — discovered incidentally on routine labs.
Surgical Indications (NIH Guidelines for Asymptomatic PHPT)
Surgery recommended for asymptomatic patients meeting any one criterion: (1) serum calcium > 1.0 mg/dL above upper limit of normal, (2) T-score ≤ −2.5 at lumbar spine, hip, or distal radius, (3) vertebral fracture, (4) eGFR < 60 mL/min, (5) 24-hour urine calcium > 400 mg/day with increased stone risk, (6) nephrolithiasis/nephrocalcinosis, (7) age < 50. All symptomatic patients should undergo surgery.
Localization Studies
Sestamibi scan (Tc-99m sestamibi): Taken up by mitochondria-rich oxyphil cells in parathyroid adenomas; delayed washout compared to thyroid tissue. Sensitivity for single adenoma: 80-90%. SPECT/CT (single-photon emission CT fused with CT) improves localization accuracy, especially for ectopic glands. 4D-CT parathyroid: High spatial resolution with multiphasic contrast enhancement; identifies adenomas by their characteristic arterial-phase enhancement and rapid washout. Sensitivity: 85-94%; particularly useful for reoperative cases, discordant imaging, or multigland disease. The "4th dimension" refers to the temporal changes in contrast enhancement over time. Ultrasound: Operator-dependent; sensitivity 65-80%; useful for concurrent thyroid pathology assessment and surgeon-performed US for preoperative planning. Ectopic gland locations: Undescended (near the carotid bifurcation), intrathyroidal, retroesophageal, mediastinal (thymus — most common ectopic location for inferior glands; derived from the 3rd pharyngeal pouch along with the thymus), within the carotid sheath. Ectopic glands are the most common cause of failed initial parathyroidectomy.
Surgical Approaches
Minimally invasive parathyroidectomy (MIP): Targeted removal of a localized adenoma through a small (~2-3 cm) incision, guided by concordant preoperative imaging. Confirmed by intraoperative PTH monitoring (IOPTH) — the Miami criterion: PTH drops ≥ 50% from the highest pre-excision level at 10 minutes post-excision AND falls into the normal range. If the Miami criterion is not met, multigland disease should be suspected and bilateral neck exploration performed. Bilateral neck exploration (4-gland exploration): Standard approach when localization is negative, discordant, or IOPTH suggests multigland disease. All four glands are identified; abnormal glands are excised. For multigland hyperplasia: subtotal parathyroidectomy (3.5 glands removed, leaving a ~50 mg remnant of the most normal-appearing gland) or total parathyroidectomy with forearm autotransplant.
Complications
RLN injury, hungry bone syndrome (severe hypocalcemia postoperatively — especially in patients with significant preoperative bone disease; calcium and calcitriol supplementation required), persistent/recurrent hyperparathyroidism (failure to achieve normocalcemia or recurrence — most commonly due to missed multigland disease, ectopic gland, or supernumerary gland).
23 Head & Neck Squamous Cell Carcinoma
Risk Factors & HPV-Positive Oropharyngeal Cancer
Head and neck SCC (HNSCC) arises from the mucosal epithelium of the upper aerodigestive tract. Traditional risk factors: tobacco and alcohol (synergistic — 15-fold increase). HPV-positive oropharyngeal SCC (p16+) is now the dominant etiology of oropharyngeal cancer in developed countries. HPV type 16 accounts for ~90% of HPV+ cases. HPV+ OPC has a distinct epidemiology (younger, non-smoking males, higher socioeconomic status), biology (favorable prognosis, better response to treatment), and staging (AJCC 8th edition created a separate staging system for p16+ OPC). Immunohistochemistry for p16 overexpression is a surrogate marker for transcriptionally active HPV — p16+ oropharyngeal cancers have significantly better survival (70-90% 5-year OS) compared to HPV-negative tumors (40-50%).
Staging & Workup
AJCC 8th edition staging. Workup: complete head and neck exam with FNL, biopsy (tissue diagnosis), CT neck with contrast (primary tumor and nodal assessment), MRI (superior for soft tissue — tongue base, parapharyngeal space), PET/CT (nodal and distant metastasis staging, especially for unknown primary), panendoscopy (direct laryngoscopy, esophagoscopy, bronchoscopy) if indicated for second primary evaluation (field cancerization).
Neck Dissection
Cervical lymph node levels: Level I (submental/submandibular), Level II (upper jugular — IIa anterior to SAN, IIb posterior), Level III (middle jugular), Level IV (lower jugular), Level V (posterior triangle — Va above cricoid, Vb below), Level VI (anterior/central — pre-tracheal, paratracheal), Level VII (superior mediastinal).
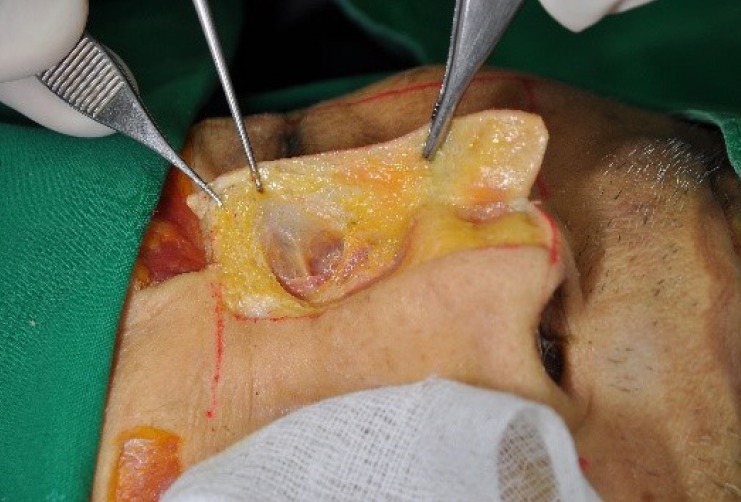
| Type | Structures Removed | Indication |
|---|---|---|
| Radical neck dissection (RND) | Levels I-V + SCM + IJV + CN XI | Bulky N2-N3 disease with involvement of these structures; historical gold standard |
| Modified radical ND (MRND) | Levels I-V, preserving ≥ 1 of: SCM, IJV, CN XI | Clinically positive nodes without invasion of preserved structures |
| Selective ND | Selected levels only (e.g., I-III for oral cavity; II-IV for oropharynx/larynx) | Clinically N0 neck (elective) or limited nodal disease |
Free Flap Reconstruction
Microvascular free tissue transfer is essential for reconstructing large surgical defects. Common flaps: radial forearm free flap (RFFF) — thin, pliable; oral cavity/oropharynx reconstruction. Anterolateral thigh flap (ALT) — versatile, bulkier; oropharynx, hypopharynx, skull base. Fibular free flap — bone + skin; mandibular reconstruction. Scapular/parascapular flap — bone and soft tissue for complex composite defects. Free flap success rate: > 95% in experienced centers. Postoperative monitoring: external Doppler checks of the vascular pedicle every 1-2 hours for 48-72 hours; venous congestion (flap appears purple/dark) is the most common cause of flap compromise — urgent return to OR for thrombectomy and reanastomosis within 6-8 hours.
Unknown Primary of the Head & Neck
Approximately 2-5% of patients with HNSCC present with cervical lymphadenopathy (usually level II) and no identifiable primary site on physical examination. Workup: FNL with directed biopsies of high-yield sites (tongue base, tonsils, nasopharynx), FDG-PET/CT (identifies the primary in 25-35% of cases), panendoscopy with bilateral tonsillectomy (uncovers the primary in an additional 20-30% — palatine and lingual tonsils harbor occult HPV+ tumors). p16 positivity in the node directs attention to the oropharynx. Management: if primary found — site-specific treatment; if primary not found — bilateral neck irradiation covering the mucosal axis (nasopharynx, oropharynx, hypopharynx) with concurrent chemotherapy for advanced nodal disease.
24 Salivary Gland Surgery
Salivary Gland Tumors
The parotid gland accounts for ~80% of salivary gland tumors, of which ~80% are benign. The submandibular gland has a roughly 50:50 benign-to-malignant ratio. Minor salivary glands (palate is the most common site) have a higher proportion of malignancy (~50-80%).
| Tumor | Benign/Malignant | Key Features |
|---|---|---|
| Pleomorphic adenoma | Benign (most common salivary tumor, 60-70%) | Slow-growing, painless, firm mass; incomplete capsule — local recurrence if inadequately excised (enucleation is insufficient); risk of malignant transformation to carcinoma ex pleomorphic adenoma (5-15% over 15-20 years) |
| Warthin's tumor | Benign (5-10%) | Almost exclusively in parotid; bilateral in 10-15%; associated with smoking; cystic, well-encapsulated; Tc-99m pertechnetate avid |
| Mucoepidermoid carcinoma | Malignant (most common salivary malignancy overall) | Low-grade: cystic, slow-growing, excellent prognosis. High-grade: solid, aggressive, poor prognosis. Most common malignant tumor in children and in the parotid. |
| Adenoid cystic carcinoma | Malignant | Perineural invasion (hallmark — painful, facial nerve involvement). Three patterns: tubular (best prognosis), cribriform (intermediate), solid (worst prognosis). Slow-growing but relentless; high rate of late distant metastasis (lung); prolonged clinical course even with metastases. |
| Acinic cell carcinoma | Malignant (low-grade) | Second most common parotid malignancy; bilateral in 3%. Generally favorable prognosis. |
Parotidectomy
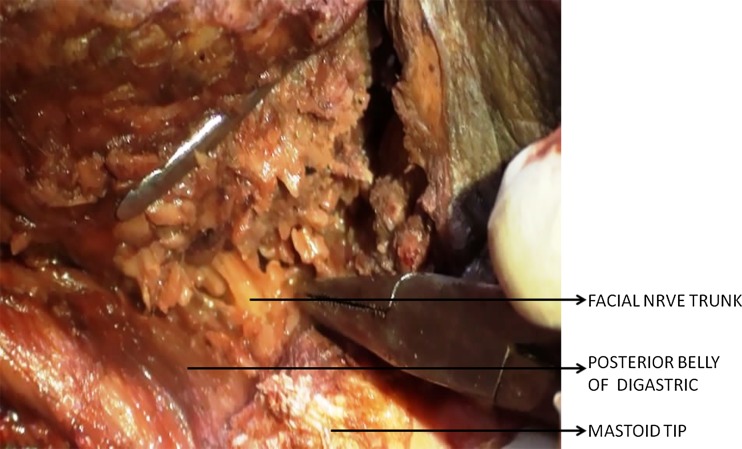
Facial nerve identification is the critical step. The nerve trunk is found using landmarks: (1) tragal pointer (1 cm medial and inferior), (2) tympanomastoid suture line, (3) posterior belly of digastric muscle (nerve crosses superficial to this at the stylomastoid foramen). The nerve is dissected from trunk to branches (antegrade) or from a distal branch to trunk (retrograde) — the nerve is preserved unless directly invaded by tumor. Superficial parotidectomy: Removal of tissue lateral to the facial nerve plane; sufficient for most benign tumors of the superficial lobe. Total parotidectomy: Removal of superficial and deep lobes with facial nerve preservation; indicated for deep-lobe tumors and malignancies. Radical parotidectomy: Total parotidectomy with sacrifice of the facial nerve (for direct nerve invasion by malignancy); nerve reconstruction with cable grafting (sural nerve, great auricular nerve).
Submandibular Gland Excision
Performed through a transcervical incision 2 cm below the mandible. Key structures at risk: marginal mandibular nerve (CN VII — identify and protect with the Hayes-Martin maneuver), lingual nerve (runs deep to the gland and loops around Wharton's duct — must be identified and preserved), hypoglossal nerve (deep to the gland). Indications: sialolithiasis refractory to conservative management, chronic sialadenitis, tumor.
Sialolithiasis
Sialolithiasis (salivary gland stones) is the most common cause of salivary gland swelling. The submandibular gland accounts for ~80% of salivary stones — its duct (Wharton's duct) courses upward against gravity, the saliva is more alkaline and viscous, and the duct has a narrower orifice. Presentation: recurrent, meal-related swelling and pain of the affected gland. Diagnosis: bimanual palpation of the floor of mouth (stones often palpable in the duct), CT (calcium-containing stones — 80% of submandibular stones are radiopaque vs only 20% of parotid stones), ultrasound, sialendoscopy. Treatment: small stones (< 5 mm) — sialendoscopy with basket retrieval/lithotripsy; larger stones or stones near the hilum — intraoral duct incision (for distal Wharton's duct stones), sialendoscopy-assisted transoral surgery, or gland excision for recurrent/hilar stones refractory to minimally invasive techniques.
Complications of Parotidectomy
Facial nerve injury (temporary: 20-30%, permanent: 1-4% for benign tumors), Frey's syndrome (gustatory sweating — aberrant reinervation of parasympathetic fibers to cutaneous sweat glands; caused by postganglionic parasympathetic fibers from the auriculotemporal nerve innervating sweat glands instead of the parotid gland after surgery; diagnosis confirmed by Minor starch-iodine test; treatment: topical antiperspirant, botulinum toxin injection into the affected skin), great auricular nerve injury (numbness of the ear lobule — most commonly injured nerve in parotid surgery; transected in up to 7% of parotidectomies), salivary fistula/sialocele (managed with pressure dressings, aspiration, botulinum toxin injection into the gland remnant), first bite syndrome (pain with first bite of each meal — sympathetic denervation of myoepithelial cells, associated with deep-lobe surgery or parapharyngeal space surgery; management: botulinum toxin, gabapentin, carbamazepine).
25 Facial Plastics & Reconstruction
Rhinoplasty Overview
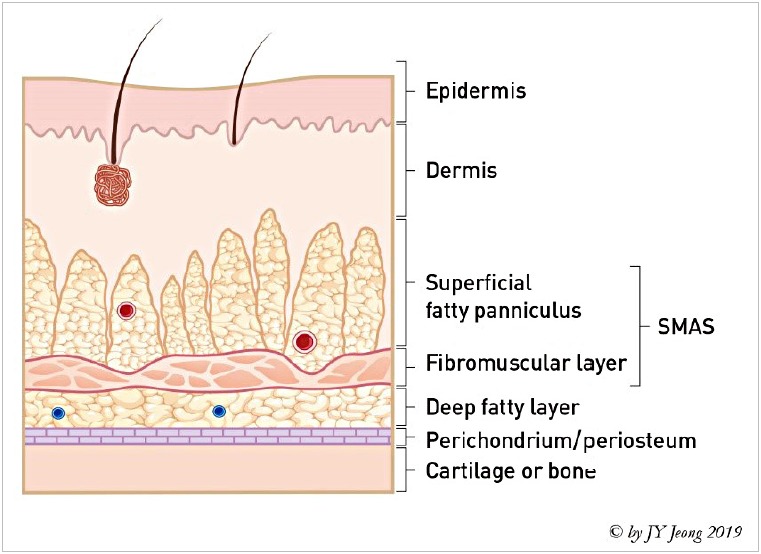
Rhinoplasty is performed for both functional (nasal obstruction, valve collapse) and aesthetic indications. It is one of the most complex facial plastic procedures. Approaches: closed (endonasal) — all incisions inside the nose; limited visualization; suitable for straightforward cases. Open (external) — transcolumellar incision (inverted-V) connecting bilateral marginal incisions; provides full exposure of the nasal framework; preferred for complex revisions, tip work, and asymmetric deformities. Key concepts: the nasal tripod (two lateral crura and the paired medial crura), tip projection and rotation, dorsal profile alignment, nasal valve integrity, and skin envelope characteristics (thick vs thin). Grafting materials: septal cartilage (preferred), auricular cartilage, costal cartilage (rib — for revision cases with depleted septal cartilage).
Facial Nerve Repair
Options after facial nerve injury: primary neurorrhaphy (tension-free end-to-end repair, ideal if performed within 72 hours of injury and gap < 1 cm). Cable grafting (interposition nerve graft — sural nerve or great auricular nerve — for gaps > 1 cm). Cross-facial nerve grafting (from contralateral normal facial nerve — for proximal injuries with unavailable proximal stump). Nerve transfer (masseteric nerve to facial nerve branch — fastest recovery, excellent smile restoration). Static procedures (gold weight upper eyelid implant for lagophthalmos, lower lid tightening, fascia lata sling for oral commissure).
Otoplasty & Auricular Reconstruction
Otoplasty for prominent ears (typically performed after age 5-6 when the auricle has reached ~85% of adult size): techniques include cartilage scoring/suturing (Mustardé, Furnas) to create or accentuate the antihelical fold and reduce conchal excess. Microtia reconstruction: Congenital absence/hypoplasia of the external ear. Grading: Grade I (small ear with identifiable structures), Grade II (some structures present), Grade III (peanut ear — small skin/cartilage remnant), Grade IV (anotia — complete absence). Reconstruction options: autologous rib cartilage framework (Nagata/Brent technique — staged, beginning at age 6-10), porous polyethylene implant (Medpor — single stage, higher extrusion rate), or prosthetic ear (osseointegrated implants). Associated with conductive hearing loss (aural atresia) — BAHA for hearing rehabilitation.
Cutaneous Malignancies
Basal cell carcinoma (BCC): Most common skin cancer; head and neck is the most common site; locally destructive but metastasis is exceedingly rare. Treatment: excision (4 mm margin) or Mohs micrographic surgery (MMS — preferred for high-risk areas: nose, periorbital, ears, lips — margin-controlled excision with highest cure rate > 99%). Squamous cell carcinoma (SCC): Second most common; higher metastatic potential than BCC (especially in immunosuppressed patients, perineural invasion, recurrent tumors). Melanoma: Excision margins based on Breslow thickness (in situ: 0.5-1 cm; ≤ 1 mm: 1 cm; 1.01-2 mm: 1-2 cm; > 2 mm: 2 cm). Sentinel lymph node biopsy for tumors > 0.8 mm or with ulceration. Head and neck melanoma has more complex lymphatic drainage (sentinel nodes may be in the parotid, neck, or even contralateral neck) — preoperative lymphoscintigraphy is essential. Lentigo maligna (melanoma in situ) of the face may be managed with staged excision with margin control (slow Mohs) given the need for tissue conservation.
Local and Regional Flaps
For smaller defects where free tissue transfer is not required, the head and neck has excellent blood supply supporting numerous local and regional flap options. Local flaps: advancement (V-Y, A-to-T), rotation, transposition (rhombic/Limberg flap, bilobed flap — especially useful for nasal defects), interpolation flaps (paramedian forehead flap — the workhorse for large nasal defects, based on the supratrochlear artery; requires a pedicle division at 3 weeks; nasolabial flap for alar/upper lip defects). Regional pedicled flaps: pectoralis major myocutaneous flap (based on thoracoacromial artery — reliable backup for head and neck reconstruction, especially pharyngeal defects; does not require microvascular anastomosis), submental flap (based on the submental artery — excellent for floor of mouth and lower face), supraclavicular flap, temporalis muscle flap (for orbital or lateral skull base defects). Flap selection depends on the defect size, location, depth (skin only vs composite), donor site morbidity, and patient comorbidities.
26 Pediatric ENT
Tonsillectomy & Adenoidectomy
Tonsillectomy is one of the most commonly performed surgical procedures in children. Indications: (1) Obstructive sleep apnea (OSA) — the most common indication in children (adenotonsillar hypertrophy is the primary cause of pediatric OSA; polysomnography is the gold standard for diagnosis). (2) Recurrent tonsillitis — Paradise criteria: ≥ 7 episodes in 1 year, or ≥ 5 per year for 2 years, or ≥ 3 per year for 3 years (each episode documented with fever > 38.3°C, cervical lymphadenopathy, tonsillar exudate, or positive group A strep test). (3) Peritonsillar abscess (recurrent or non-drainable), (4) Suspected malignancy (asymmetric tonsillar enlargement).
Complications: Post-tonsillectomy hemorrhage (~3-5% overall; primary: within 24 hours; secondary: 5-10 days postop, most common timing) — Emergency may require return to OR for hemostasis. Dehydration (pain limiting oral intake). Velopharyngeal insufficiency (VPI — especially in patients with occult submucous cleft palate). Atlantoaxial subluxation (Grisel syndrome — rare). Eagle syndrome (elongated styloid process causing pain — rare late complication).
Pediatric Obstructive Sleep Apnea
Adenotonsillar hypertrophy is the primary cause of pediatric OSA. Unlike adult OSA, obesity is not the primary driver in most children (though it is increasingly important). Consequences of untreated pediatric OSA: failure to thrive, behavioral problems (ADHD-like symptoms), neurocognitive impairment, enuresis, pulmonary hypertension, cor pulmonale. First-line treatment: Adenotonsillectomy (cure rate ~80% in non-obese children). CPAP/high-flow oxygen for persistent OSA after surgery or in children who are not surgical candidates.
Obstructive Sleep Apnea — Adult Considerations
While pediatric OSA is discussed above, adult OSA is managed differently. ENT evaluation is critical for identifying the site(s) of obstruction: velum/palate (Friedman tonsil grading, modified Mallampati), oropharyngeal lateral walls, tongue base, and epiglottis. Drug-induced sleep endoscopy (DISE) allows direct visualization of the airway under sedation mimicking sleep — identifies the level and pattern of collapse (anteroposterior, lateral, concentric) to guide surgical planning. Surgical options for adults who fail CPAP: uvulopalatopharyngoplasty (UPPP) — addresses palatal and tonsillar obstruction (cure rate only ~40-50% as monotherapy), tongue base reduction (transoral robotic surgery — TORS, radiofrequency ablation), genioglossus advancement, hyoid suspension, and maxillomandibular advancement (MMA) — the most effective single surgical intervention for adult OSA (cure rate > 85%), reserved for severe or multilevel obstruction. Hypoglossal nerve stimulation (Inspire) — implantable device that stimulates the hypoglossal nerve to protrude the tongue during sleep, preventing tongue base collapse; indicated for moderate-severe OSA with BMI < 35 and CPAP failure/intolerance; no complete concentric collapse at the palate on DISE.
Newborn Hearing Screening
Universal newborn hearing screening (UNHS) uses OAE and/or ABR. Failed screening requires diagnostic ABR within 3 months; amplification (hearing aids) by 6 months. Early identification and intervention (< 6 months) is critical for speech and language development. Risk factors for hearing loss: NICU stay > 5 days, family history, ototoxic medications (aminoglycosides, loop diuretics), hyperbilirubinemia requiring exchange transfusion, CMV infection, craniofacial anomalies.
Congenital Neck Masses
| Mass | Location | Key Features | Treatment |
|---|---|---|---|
| Thyroglossal duct cyst | Midline, at or below hyoid | Most common congenital midline neck mass. Elevates with tongue protrusion and swallowing. May contain ectopic thyroid tissue (confirm normal thyroid position with US before excision). | Sistrunk procedure: excision of the cyst, the central body of the hyoid bone, and the tract up to the foramen cecum. Recurrence ~3% with Sistrunk (vs 50% with simple excision). |
| Branchial cleft anomaly (2nd) | Anterior border of SCM | Most common branchial anomaly (95%). Second arch: cyst/sinus/fistula. Fistula runs from tonsillar fossa (internal opening) to anterior SCM border (external opening), passing between the internal and external carotid arteries, over CN XII, and deep to CN IX. | Complete surgical excision including the entire tract/fistula. |
| First branchial cleft anomaly | Periauricular | Intimate relationship with the facial nerve (Work classification: Type I — ectodermal duplication near EAC; Type II — involves EAC and cartilage, courses lateral, medial, or through the facial nerve). | Excision with facial nerve identification and superficial parotidectomy. |
| Dermoid cyst | Midline, submental or nasal | Contains skin appendages (hair follicles, sebaceous glands). Nasal dermoid: must evaluate for intracranial extension (dumbbell-shaped) with MRI before excision. | Complete excision including any intracranial component. |
27 Classification Systems
House-Brackmann Facial Nerve Grading System
| Grade | Description | Characteristics |
|---|---|---|
| I | Normal | Normal facial function in all areas |
| II | Mild dysfunction | Slight weakness on close inspection; complete eye closure with minimal effort; slight mouth asymmetry |
| III | Moderate dysfunction | Obvious weakness but not disfiguring; complete eye closure with effort; slight mouth movement with maximum effort |
| IV | Moderately severe | Obvious disfiguring weakness; incomplete eye closure; asymmetric mouth movement with maximum effort |
| V | Severe dysfunction | Barely perceptible motion; incomplete eye closure; slight mouth movement |
| VI | Total paralysis | No movement |
Cotton-Myer Subglottic Stenosis Classification
| Grade | % Obstruction |
|---|---|
| I | 0-50% |
| II | 51-70% |
| III | 71-99% |
| IV | 100% (no detectable lumen) |
AAO-HNS Ménière's Disease Staging
| Stage | Pure Tone Average (0.5, 1, 2, 3 kHz) |
|---|---|
| 1 | ≤ 25 dB |
| 2 | 26-40 dB |
| 3 | 41-70 dB |
| 4 | > 70 dB |
Krouse Classification — Inverted Papilloma
| Stage | Extent |
|---|---|
| T1 | Confined to nasal cavity (no extension into sinuses) |
| T2 | Involving ostiomeatal complex, ethmoid sinuses, and/or medial maxillary sinus |
| T3 | Involving lateral, inferior, superior, or posterior walls of maxillary sinus, or frontal or sphenoid sinus |
| T4 | Extension outside the nose/sinuses (orbit, intracranial) or associated malignancy |
Kadish Staging — Esthesioneuroblastoma
| Stage | Extent |
|---|---|
| A | Confined to the nasal cavity |
| B | Nasal cavity and one or more paranasal sinuses |
| C | Extension beyond nasal cavity and sinuses (orbit, skull base, intracranial, cervical nodes) |
| D (Modified) | Distant metastasis |
Bethesda System for Thyroid FNA
| Category | Diagnosis | Risk of Malignancy |
|---|---|---|
| I | Non-diagnostic / Unsatisfactory | 5-10% |
| II | Benign | 0-3% |
| III | AUS / FLUS | 10-30% |
| IV | Follicular neoplasm / Suspicious for FN | 25-40% |
| V | Suspicious for malignancy | 50-75% |
| VI | Malignant | 97-99% |
Keros Classification — Olfactory Fossa Depth
| Type | Depth of Olfactory Fossa | Surgical Risk |
|---|---|---|
| I | 1-3 mm | Low — lateral lamella is short and thick |
| II | 4-7 mm | Moderate — most common configuration |
| III | 8-16 mm | High — lateral lamella is long and thin; highest risk of CSF leak during FESS |
Lund-Mackay CT Scoring System
| Sinus (Each Side) | Score |
|---|---|
| Maxillary | 0 (clear), 1 (partial opacification), 2 (complete opacification) |
| Anterior ethmoid | 0, 1, 2 |
| Posterior ethmoid | 0, 1, 2 |
| Sphenoid | 0, 1, 2 |
| Frontal | 0, 1, 2 |
| Ostiomeatal complex | 0 (not occluded), 2 (occluded) |
Total score range: 0-24 (12 per side). A Lund-Mackay score of 0 has a high negative predictive value for CRS. Scores do not correlate linearly with symptom severity.
AJCC 8th Edition — Key H&N Staging Highlights
p16+ oropharyngeal cancer: Separate staging — clinical T staging uses tumor size (T1 ≤ 2 cm, T2 2-4 cm, T3 > 4 cm, T4 invasion of adjacent structures); N staging by number and laterality (N1: ipsilateral ≤ 6 cm, N2: contralateral/bilateral ≤ 6 cm, N3: > 6 cm). Stage groupings are more favorable than p16-negative — even patients with extensive nodal disease may be Stage I-II. p16-negative HNSCC: Standard staging with ENE (extranodal extension) now a critical factor — ENE+ automatically upstages to N3b regardless of node size. Larynx: Staging varies by subsite (glottic, supraglottic, subglottic) as described in Section 17.
Cernea Classification — External Branch of the SLN
| Type | EBSLN Position Relative to Superior Thyroid Pole | Surgical Risk |
|---|---|---|
| 1 | Crosses superior thyroid vessels > 1 cm above the upper pole | Low |
| 2a | Crosses within 1 cm above the upper pole | Moderate |
| 2b | Crosses below the upper pole (within the surgical field) | High (20% of patients) |
28 Medications Master Table
Antibiotics
| Drug | Indication | Dose (Adult) | Key Notes |
|---|---|---|---|
| Amoxicillin | AOM (first-line), ABRS | 875 mg BID or 80-90 mg/kg/day (pediatric) | High-dose for resistant S. pneumoniae |
| Amoxicillin-clavulanate | ABRS (first-line), AOM (second-line/treatment failure) | 875/125 mg BID or 2 g/125 mg BID (high-dose) | Covers beta-lactamase producers (H. influenzae, M. catarrhalis) |
| Ceftriaxone | AOM (IM), severe infections, epiglottitis | 50 mg/kg IM (AOM); 1-2 g IV daily | Single IM dose for AOM treatment failure or vomiting child |
| Ciprofloxacin-dexamethasone otic | Otitis externa, CSOM, post-tympanostomy tube otorrhea | 4 drops BID x 7 days | Ototoxicity-safe fluoroquinolone for middle ear use |
| Ofloxacin otic | CSOM, tympanostomy tube otorrhea | 5-10 drops BID x 14 days | FDA-approved for middle ear; no ototoxicity |
| Clindamycin | Peritonsillar abscess, deep neck space infections | 600-900 mg IV q8h; 300-450 mg PO QID | Excellent anaerobic coverage; use with metronidazole or alone |
| Ampicillin-sulbactam | Deep neck space infections, epiglottitis | 3 g IV q6h | Broad spectrum including anaerobes |
Corticosteroids
| Drug | Indication | Dose | Key Notes |
|---|---|---|---|
| Dexamethasone (systemic) | Croup, peritonsillar abscess, SSNHL, post-extubation stridor | 0.6 mg/kg PO/IM (croup); 10 mg IV (adult loading) | Single dose highly effective for croup |
| Prednisone | SSNHL, CRSwNP, Bell's palsy, Ménière's | 1 mg/kg/day (max 60 mg) x 10-14 days with taper | Start within 72 hours for SSNHL; 2 weeks for Bell's palsy |
| Intratympanic dexamethasone | SSNHL (primary or salvage), Ménière's | 10-24 mg/mL, 0.3-0.5 mL per injection x 3-4 sessions | Direct inner ear delivery; avoids systemic side effects |
| Fluticasone propionate nasal spray | Allergic rhinitis, CRS maintenance | 1-2 sprays per nostril daily | First-line for allergic rhinitis; safe for long-term use |
| Budesonide nasal irrigations | CRSwNP, post-FESS | 0.5 mg in 240 mL saline BID | High-volume irrigation delivers steroid to sinus mucosa |
Biologics for CRSwNP
| Drug | Mechanism | Dose | Key Notes |
|---|---|---|---|
| Dupilumab | Anti-IL-4Rα (blocks IL-4 and IL-13) | 300 mg SC q2 weeks | FDA-approved for CRSwNP; reduces polyp score, improves smell, reduces need for surgery and oral steroids |
| Omalizumab | Anti-IgE | 75-600 mg SC q2-4 weeks (weight/IgE-based) | FDA-approved for CRSwNP; particularly useful with comorbid asthma |
| Mepolizumab | Anti-IL-5 | 100 mg SC q4 weeks | FDA-approved for CRSwNP; targets eosinophilic inflammation |
Other Key Medications
| Drug | Indication | Dose | Key Notes |
|---|---|---|---|
| Propranolol | Infantile subglottic hemangioma | 2-3 mg/kg/day PO divided TID | First-line; monitor for bradycardia, hypoglycemia, bronchospasm |
| Intratympanic gentamicin | Ménière's disease (refractory vertigo) | 26.7-40 mg/mL, 0.3-0.5 mL per injection | Chemical labyrinthectomy; 10-30% risk of hearing loss |
| Oxymetazoline 0.05% | Epistaxis, nasal decongestion (pre-procedure) | 2-3 sprays per nostril | Limit to 3-5 days to prevent rhinitis medicamentosa |
| Cisplatin | H&N SCC (concurrent chemoradiation) | 100 mg/m² IV q3 weeks x 3 cycles (or 40 mg/m² weekly) | Standard radiosensitizer; ototoxic, nephrotoxic, neurotoxic |
| Cetuximab | H&N SCC (with radiation; platinum-ineligible) | 400 mg/m² loading then 250 mg/m² weekly | Anti-EGFR; used when cisplatin contraindicated |
| Pembrolizumab/Nivolumab | Recurrent/metastatic H&N SCC | 200 mg IV q3 weeks (pembro); 240 mg IV q2 weeks (nivo) | Anti-PD-1 immunotherapy; first-line for recurrent/metastatic disease |
29 Imaging & Diagnostics
CT Temporal Bone
High-resolution CT temporal bone (0.5-0.625 mm axial cuts, bone algorithm) is the primary imaging modality for: cholesteatoma (soft tissue in the middle ear/mastoid with bony erosion), temporal bone fractures (otic capsule-sparing vs -violating), otosclerosis (lucent foci at the fissula ante fenestram), SSCD (dehiscence on Poschl/Stenvers reformats), congenital aural atresia (Jahrsdoerfer grading), and cochlear implant evaluation (cochlear patency, cochlear anomalies). CT uses bone detail superbly but is limited for soft tissue differentiation.
MRI Internal Auditory Canal (IAC)
MRI with gadolinium is the gold standard for: vestibular schwannoma (enhancing mass in IAC/CPA — most common CPA tumor; SSNHL workup), other CPA tumors (meningioma — dural tail sign; epidermoid — DWI restriction), facial nerve pathology (Bell's palsy — enhancement of labyrinthine/meatal segments), inner ear anomalies (cochlear nerve hypoplasia/aplasia in congenital SNHL), and perineural spread of malignancy along cranial nerves. 3D FIESTA/CISS sequences provide high-resolution images of the fluid-filled inner ear and cranial nerves without contrast — excellent for cochlear patency assessment and small intracanalicular schwannomas.
CT Sinuses
CT sinuses (non-contrast, coronal and axial) is the imaging standard for sinusitis and pre-FESS planning. Lund-Mackay scoring: each of 6 sinus groups (maxillary, anterior ethmoid, posterior ethmoid, sphenoid, frontal — each side) scored 0 (no opacification), 1 (partial), or 2 (complete); ostiomeatal complex scored 0 (not occluded) or 2 (occluded). Total score range: 0-24. A score of 0 effectively rules out CRS. Key pre-FESS review: Keros classification, Onodi cells, lamina papyracea integrity, ICA/optic nerve dehiscence in sphenoid, frontal recess anatomy.
PET/CT for Head & Neck Cancer
FDG-PET/CT is used in H&N oncology for: staging (detecting occult nodal and distant metastases), unknown primary workup (FDG-avid mucosal lesion directs biopsy), treatment response assessment (3 months post-chemoradiation), and surveillance for recurrence. Negative PET/CT at 12 weeks post-treatment has a high negative predictive value (> 95%) for persistent nodal disease — may obviate the need for planned neck dissection. False positives: inflammation, post-radiation changes, infection. False negatives: small lesions (< 1 cm), well-differentiated tumors with low metabolic activity.
Vestibular Schwannoma (Acoustic Neuroma)
Vestibular schwannoma is the most common tumor of the cerebellopontine angle (~80% of CPA tumors) and the most common tumor of the temporal bone. Arises from the Schwann cells of the vestibular division of CN VIII, usually at the transition zone between central and peripheral myelin (Obersteiner-Redlich zone) within the IAC. Bilateral vestibular schwannomas are pathognomonic for neurofibromatosis type 2 (NF2). Presentation: progressive unilateral SNHL (most common), tinnitus, disequilibrium. Large tumors: CN V numbness, CN VII weakness, cerebellar signs, hydrocephalus. Diagnosis: MRI with gadolinium — enhancing mass in IAC ± CPA. Treatment options: observation with serial MRI (for small, slow-growing tumors in elderly patients), stereotactic radiosurgery (Gamma Knife — for tumors < 2.5-3 cm, > 90% tumor control rate), or microsurgical excision (translabyrinthine — sacrifices hearing; retrosigmoid or middle fossa approach — hearing preservation possible for small tumors with good preoperative hearing). Treatment choice depends on tumor size, hearing status, patient age, and comorbidities.
Deep Neck Space Infections
Peritonsillar abscess (PTA/quinsy) is the most common deep neck space infection. Presents with severe sore throat, odynophagia, "hot potato" voice, trismus, uvular deviation, and unilateral tonsillar bulging. Treatment: needle aspiration or incision and drainage (I&D) + antibiotics (amoxicillin-clavulanate or clindamycin). Interval tonsillectomy for recurrent PTA. Parapharyngeal space abscess presents with trismus, medial displacement of the tonsil/lateral pharyngeal wall, and neck swelling below the angle of the mandible. Retropharyngeal abscess occurs predominantly in children < 6 years (retropharyngeal nodes — nodes of Rouviere — suppurate after URI). Lateral neck radiograph: widening of the prevertebral soft tissue (> 7 mm at C2, > 14 mm in children / > 22 mm in adults at C6). CT with contrast is the imaging standard. Treatment: IV antibiotics; surgical drainage (transoral for uncomplicated retropharyngeal abscess; transcervical approach for parapharyngeal or complex infections). Ludwig's angina is a rapidly spreading cellulitis/phlegmon of the submandibular, sublingual, and submental spaces, usually from a dental infection (mandibular molar). Presents with floor-of-mouth swelling, tongue elevation and protrusion, drooling, and airway compromise. Emergency Management: secure the airway (awake fiberoptic intubation or tracheotomy; avoid blind nasal intubation), IV antibiotics (ampicillin-sulbactam, clindamycin), and surgical drainage. Lemierre syndrome: Internal jugular vein septic thrombophlebitis following oropharyngeal infection, most commonly caused by Fusobacterium necrophorum. Septic pulmonary emboli are characteristic. Treatment: prolonged IV antibiotics; anticoagulation is controversial.
Audiogram Pattern Recognition
| Pattern | Diagnosis |
|---|---|
| Low-frequency air-bone gap | Otosclerosis, SSCD, middle ear effusion |
| Flat CHL with Type B tympanogram | Middle ear effusion |
| High-frequency sloping SNHL | Presbycusis, ototoxicity |
| 4 kHz notch | Noise-induced hearing loss |
| Low-frequency SNHL (fluctuating) | Ménière's disease |
| Asymmetric SNHL | Vestibular schwannoma (until proven otherwise) |
| Carhart notch at 2 kHz | Otosclerosis |
| Cookie-bite (mid-frequency loss) | Genetic SNHL, autoimmune inner ear disease |
| Profound bilateral SNHL (congenital) | Connexin 26 (GJB2) mutation, CMV, inner ear malformation |
| Progressive mixed HL with normal TM | Advanced otosclerosis with cochlear involvement |
Key Imaging Protocols
| Study | Protocol | Primary Indications |
|---|---|---|
| CT temporal bone | 0.5-0.625 mm axial, bone algorithm, coronal reformats | Cholesteatoma, temporal bone fracture, otosclerosis, SSCD, aural atresia, CI evaluation |
| MRI IAC with gadolinium | T1 pre/post-contrast, T2 (3D FIESTA/CISS) | Vestibular schwannoma, SSNHL workup, facial nerve pathology, inner ear anomalies |
| CT sinuses | Non-contrast, 1-2 mm, coronal + axial + sagittal reformats | CRS, FESS planning, sinonasal tumors, complications of sinusitis |
| CT neck with contrast | 3 mm axial, soft tissue algorithm | Deep neck space infections, H&N cancer staging, nodal disease |
| MRI neck with contrast | T1, T2, post-contrast fat-saturated sequences | Soft tissue tumor delineation (tongue base, parapharyngeal space), perineural spread |
| FDG-PET/CT | Whole body with dedicated H&N images | H&N cancer staging, unknown primary, post-treatment surveillance |
| Barium swallow | AP and lateral fluoroscopy with barium | Zenker's diverticulum, esophageal stricture, pharyngeal motility |
| CT angiography | Contrast-enhanced, vascular protocol | Vascular tumors (JNA, carotid body tumor), preoperative vascular mapping |
30 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| ABR | Auditory brainstem response |
| ABRS | Acute bacterial rhinosinusitis |
| AC | Air conduction |
| AERD | Aspirin-exacerbated respiratory disease |
| AFRS | Allergic fungal rhinosinusitis |
| AIFS | Acute invasive fungal sinusitis |
| ALT | Anterolateral thigh (flap) |
| ANSD | Auditory neuropathy spectrum disorder |
| AOM | Acute otitis media |
| BC | Bone conduction |
| BCC | Basal cell carcinoma |
| BPPV | Benign paroxysmal positional vertigo |
| BVFP | Bilateral vocal fold paralysis |
| CHL | Conductive hearing loss |
| CI | Cochlear implant |
| CN | Cranial nerve |
| CPA | Cerebellopontine angle |
| CRS | Chronic rhinosinusitis |
| CRSsNP | CRS without nasal polyps |
| CRSwNP | CRS with nasal polyps |
| CSOM | Chronic suppurative otitis media |
| CSF | Cerebrospinal fluid |
| CT | Computed tomography |
| CTR | Cricotracheal resection |
| CWD | Canal wall down (mastoidectomy) |
| CWU | Canal wall up (mastoidectomy) |
| DPOAE | Distortion product otoacoustic emissions |
| EAC | External auditory canal |
| EBL | Estimated blood loss |
| EBSLN | External branch of the superior laryngeal nerve |
| ECA | External carotid artery |
| ENE | Extranodal extension |
| ESPAL | Endoscopic sphenopalatine artery ligation |
| FEES | Fiberoptic endoscopic evaluation of swallowing |
| FESS | Functional endoscopic sinus surgery |
| FNA | Fine-needle aspiration |
| FNL | Flexible nasolaryngoscopy |
| H&N | Head and neck |
| HHT | Hereditary hemorrhagic telangiectasia |
| HNSCC | Head and neck squamous cell carcinoma |
| HPV | Human papillomavirus |
| IAC | Internal auditory canal |
| ICA | Internal carotid artery |
| IJV | Internal jugular vein |
| IONM | Intraoperative nerve monitoring |
| IOPTH | Intraoperative parathyroid hormone monitoring |
| JNA | Juvenile nasopharyngeal angiofibroma |
| LTR | Laryngotracheal reconstruction |
| MBS | Modified barium swallow |
| MIP | Minimally invasive parathyroidectomy |
| MRI | Magnetic resonance imaging |
| MRND | Modified radical neck dissection |
| NIHL | Noise-induced hearing loss |
| OAE | Otoacoustic emissions |
| OMC | Ostiomeatal complex |
| OME | Otitis media with effusion |
| OSA | Obstructive sleep apnea |
| PET | Positron emission tomography |
| PHPT | Primary hyperparathyroidism |
| PTH | Parathyroid hormone |
| RFFF | Radial forearm free flap |
| RLN | Recurrent laryngeal nerve |
| RND | Radical neck dissection |
| SCC | Squamous cell carcinoma |
| SCM | Sternocleidomastoid muscle |
| SGS | Subglottic stenosis |
| SLN | Superior laryngeal nerve |
| SNHL | Sensorineural hearing loss |
| SPA | Sphenopalatine artery |
| SSCD | Superior semicircular canal dehiscence |
| SSNHL | Sudden sensorineural hearing loss |
| TEP | Tracheoesophageal puncture |
| TLM | Transoral laser microsurgery |
| TM | Tympanic membrane |
| TORS | Transoral robotic surgery |
| UES | Upper esophageal sphincter |
| UNHS | Universal newborn hearing screening |
| UVFP | Unilateral vocal fold paralysis |
| VEMP | Vestibular evoked myogenic potential |
| VPI | Velopharyngeal insufficiency |
31 Landmark Guidelines & Trials
| Trial / Guideline | Year | Key Finding / Recommendation |
|---|---|---|
| VA Laryngeal Cancer Study | 1991 | Induction chemotherapy (cisplatin + 5-FU) followed by radiation achieved laryngeal preservation in 64% of patients with advanced laryngeal cancer, with equivalent survival to total laryngectomy. Established organ preservation as a viable strategy. PMID 1978225 |
| RTOG 91-11 | 2003 | Concurrent cisplatin + radiation was superior to induction chemo then radiation for laryngeal preservation in T3-T4 laryngeal cancer. Established concurrent chemoradiation as the standard organ-preservation regimen. PMID 14645423 |
| RTOG 0129 / Ang et al. | 2010 | HPV status (p16+) is the strongest prognostic factor in oropharyngeal SCC. HPV+ tumors had 58% reduction in risk of death vs HPV-negative tumors, independent of treatment. PMID 20530316 |
| KEYNOTE-048 | 2019 | Pembrolizumab (alone or with chemotherapy) improved overall survival vs cetuximab + chemotherapy in first-line recurrent/metastatic HNSCC. Established immunotherapy as a new standard. PMID 31679945 |
| AAO-HNS CPG: Sudden Hearing Loss | 2012 | Defined SSNHL, recommended audiometric confirmation, MRI to rule out retrocochlear pathology, systemic or intratympanic corticosteroids within 2 weeks of onset. Against routine CT, antivirals, or routine blood work. PMID 22965903 |
| AAO-HNS CPG: Adult Sinusitis | 2015 | Defined ARS and CRS diagnostic criteria. Recommended against radiographic imaging for uncomplicated ARS. Amoxicillin-clavulanate as first-line for ABRS. Watchful waiting acceptable for mild uncomplicated ABRS. PMID 25832968 |
| AAO-HNS CPG: Tympanostomy Tubes | 2013 | Tubes recommended for bilateral OME ≥ 3 months with documented hearing loss; for recurrent AOM (≥ 3 in 6 months or ≥ 4 in 12 months). Against prophylactic antibiotics as first-line for recurrent AOM. PMID 23479559 |
| AAO-HNS CPG: Ménière's Disease | 2020 | Defined diagnostic criteria. Recommended dietary modification, diuretics, and betahistine (outside US). Intratympanic steroids before gentamicin. Surgery reserved for refractory cases. PMID 32267799 |
| AAO-HNS CPG: Tonsillectomy in Children | 2019 | Paradise criteria for recurrent tonsillitis. Polysomnography recommended before tonsillectomy for OSA in children at risk (obesity, Down syndrome, craniofacial anomalies). Single IV dexamethasone reduces post-tonsillectomy nausea/vomiting. PMID 30921525 |
| AAO-HNS CPG: Bell's Palsy | 2013 | Oral steroids (prednisone) within 72 hours of onset recommended (strong recommendation). Antivirals alone not recommended; combination antivirals + steroids is an option. Eye protection critical. PMID 24189771 |
| LIBERTY NP (SINUS) Trials | 2019 | Dupilumab significantly reduced nasal polyp score, improved nasal congestion, smell (Lund-Mackay score), and reduced need for systemic steroids and surgery in refractory CRSwNP. Led to FDA approval. PMID 31112386 |
| Léauté-Labrèze et al. (Propranolol for Hemangiomas) | 2008/2015 | Propranolol dramatically effective for infantile hemangiomas — transformed management from corticosteroids/surgery to medical therapy. PMID 18550876 |
| NIH Consensus on Asymptomatic PHPT | 2014 | Updated surgical indications for asymptomatic primary hyperparathyroidism: calcium > 1.0 mg/dL above normal, T-score ≤ −2.5, eGFR < 60, nephrolithiasis, age < 50. |
Key Emergency Summary
The following ENT conditions require emergent intervention:
| Emergency | Key Action | Time-Critical Window |
|---|---|---|
| Post-thyroidectomy hematoma | Open wound at bedside to evacuate hematoma | Minutes — before complete airway obstruction |
| Orbital hematoma (post-FESS) | Lateral canthotomy and inferior cantholysis | 60-90 minutes before irreversible vision loss |
| Acute invasive fungal sinusitis | Emergent surgical debridement + systemic antifungals | Hours — mortality rises with delay |
| Epiglottitis with airway compromise | Secure airway in OR (intubation or tracheotomy) | Immediate — unpredictable total obstruction |
| Bilateral vocal fold paralysis | Tracheotomy or intubation | Immediate for acute presentation |
| Button battery in esophagus | Emergent rigid esophagoscopy for removal | < 2 hours — liquefaction necrosis begins |
| Sudden sensorineural hearing loss | Same-day audiogram and corticosteroids | Within 2 weeks; best outcomes < 72 hours |
| Cricothyrotomy (failed airway) | Surgical airway through cricothyroid membrane | Immediate — "can't intubate, can't oxygenate" |
| Posterior epistaxis with hemodynamic instability | Posterior packing, resuscitation, ESPAL or embolization | Minutes to hours depending on blood loss |
| Septal hematoma | Incision and drainage | Within hours — cartilage necrosis/saddle nose |