Interventional Cardiology
Every procedure, device, access technique, hemodynamic assessment, classification, complication, medication, and management algorithm across the full scope of interventional cardiology in one place.
01 Coronary Anatomy & Variants
Coronary Arterial System
The coronary circulation arises from two ostia in the sinuses of Valsalva of the aortic root. The left main coronary artery (LMCA) arises from the left coronary sinus and typically courses 1-2 cm before bifurcating into the left anterior descending artery (LAD) and the left circumflex artery (LCx). In approximately 15-20% of patients, a ramus intermedius arises as a trifurcation branch between the LAD and LCx.
Left Anterior Descending (LAD): Courses in the anterior interventricular groove toward the apex. Gives off septal perforators (supply anterior two-thirds of the interventricular septum) and diagonal branches (D1, D2 — supply the anterolateral wall). The LAD supplies approximately 45-55% of the LV myocardium and is the most commonly diseased coronary artery. The first septal perforator is a landmark separating the left main from the proximal LAD.
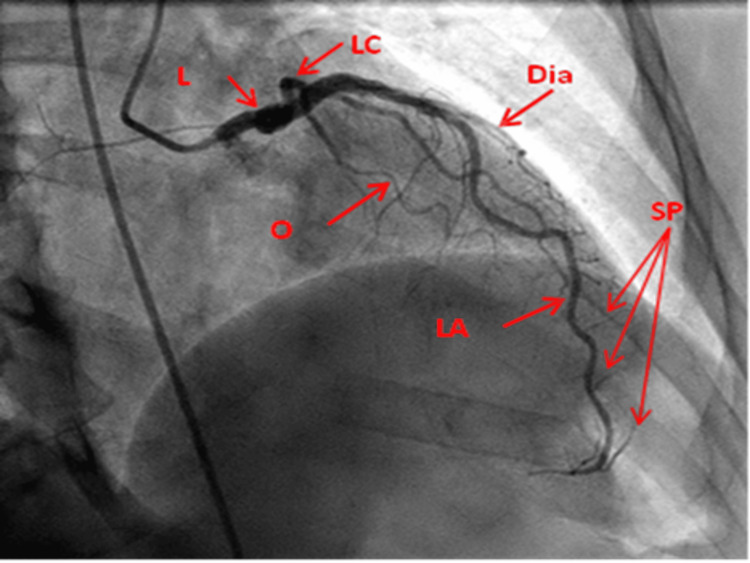
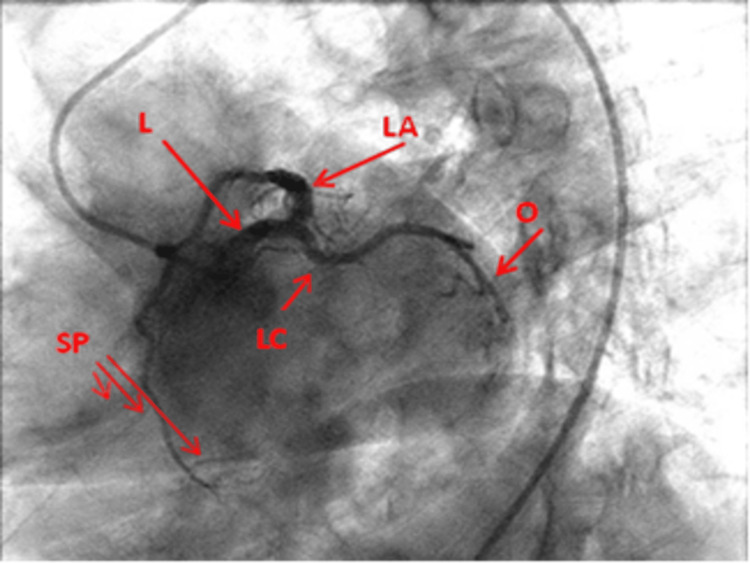
Left Circumflex (LCx): Courses in the left atrioventricular groove, giving off obtuse marginal branches (OM1, OM2) that supply the lateral and posterolateral LV wall. In approximately 15% of patients the LCx is dominant (gives rise to the PDA). The LCx also gives off the left atrial circumflex branch and, in some patients, the SA nodal artery.
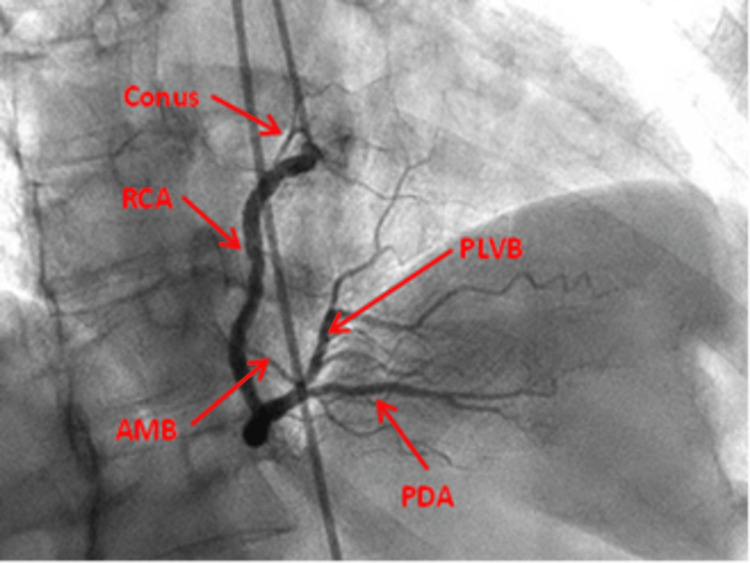
Right Coronary Artery (RCA): Arises from the right coronary sinus and courses in the right atrioventricular groove. Proximal branches include the conus branch (supplies the RV outflow tract), SA nodal artery (in ~55% of patients), and acute marginal branches (supply the RV free wall). The distal RCA gives rise to the posterior descending artery (PDA) in right-dominant circulation (~85% of the population) and the posterolateral branches.
Coronary Dominance
| Dominance | Prevalence | Definition | PDA Origin |
|---|---|---|---|
| Right dominant | ~85% | PDA arises from RCA | Distal RCA at the crux |
| Left dominant | ~8% | PDA arises from LCx | Distal LCx |
| Co-dominant | ~7% | PDA from both RCA and LCx | Shared supply |
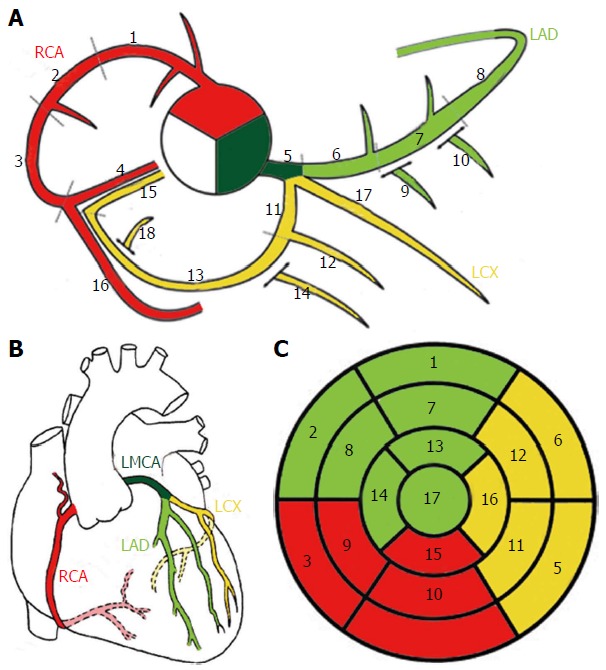
Coronary Segments (Modified AHA 15-Segment Model)
The coronary tree is divided into 15 segments for standardized reporting: RCA — proximal (1), mid (2), distal (3), PDA (4); Left Main (5); LAD — proximal (6), mid (7), distal (8), D1 (9), D2 (10); LCx — proximal (11), distal (12), OM1 (12a), OM2 (12b); Ramus intermedius (16 — if present). This segmental model is the basis of the SYNTAX score calculation.
Coronary Collaterals
Collateral circulation develops in response to chronic ischemia and high-grade stenoses. The Rentrop classification grades collateral filling: Grade 0 — no visible filling; Grade 1 — filling of side branches only; Grade 2 — partial filling of the epicardial artery; Grade 3 — complete filling of the epicardial artery. Well-developed collaterals (Rentrop 2-3) reduce infarct size and improve outcomes in CTO PCI.
Coronary Anomalies
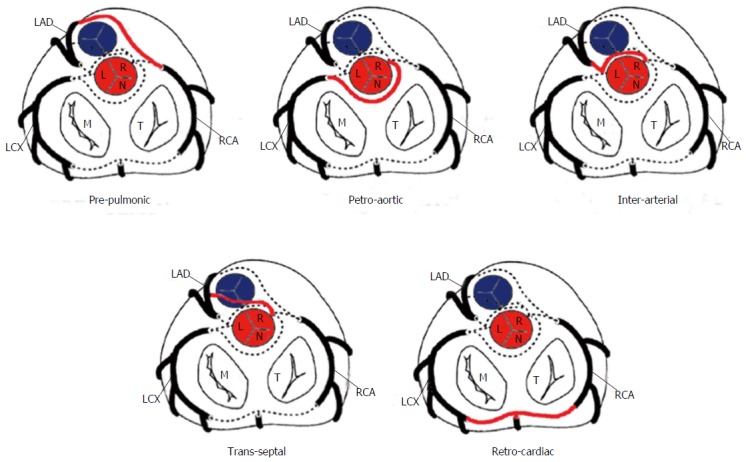
Congenital coronary anomalies occur in ~1% of the general population. Anomalous origin from the opposite sinus (ACAOS) is the most clinically significant. An anomalous left main or LAD arising from the right coronary sinus with an interarterial course (between the aorta and pulmonary artery) carries risk of sudden cardiac death due to compression during exercise. An anomalous RCA from the left sinus is more common but less often clinically significant. Other anomalies include myocardial bridging (most commonly mid-LAD, prevalence 5-25% at angiography), coronary fistulae, and anomalous origin from the pulmonary artery (ALCAPA/ARCAPA).
| Anomaly | Prevalence | Clinical Significance | Management |
|---|---|---|---|
| Anomalous LCA from right sinus (interarterial) | 0.02-0.05% | Highest risk — SCD during exertion | Surgical reimplantation or unroofing; restrict competitive sports |
| Anomalous RCA from left sinus (interarterial) | 0.1-0.3% | Lower risk; rarely causes SCD | Observation vs surgery based on symptoms/ischemia testing |
| Myocardial bridging | 5-25% (angiographic) | Systolic compression of mid-LAD; rarely causes ischemia | Beta-blockers first-line; avoid nitrates (worsen compression); myotomy if refractory |
| Coronary AV fistula | 0.1-0.2% | Left-to-right shunt; steal phenomenon; endocarditis risk | Transcatheter coil/plug embolization if symptomatic or Qp/Qs >1.5 |
| ALCAPA (Bland-White-Garland) | 1 in 300,000 | LCA from PA; myocardial ischemia in infancy | Surgical reimplantation of LCA to aorta |
Coronary Artery Territory & ECG Correlation
| Coronary Artery | Myocardial Territory | ECG Leads | Complications if Occluded |
|---|---|---|---|
| Proximal LAD | Anterior wall, septum, apex | V1-V4 (anterior); I, aVL if diagonal | Large anterior MI, LV dysfunction, CHB (septal necrosis) |
| LAD (mid/distal) | Apical anterior wall | V3-V6 | Apical aneurysm, mural thrombus |
| LCx | Lateral, posterolateral wall | I, aVL, V5-V6; reciprocal V1-V3 depression | Lateral MI; papillary muscle rupture (posteromedial) |
| RCA (proximal) | Inferior wall, RV, conduction system | II, III, aVF; V1, V4R (RV infarct) | Inferior MI, RV infarct, bradycardia (AV block) |
| PDA | Inferior septum, inferior wall | II, III, aVF | Inferior MI; AV nodal dysfunction |
02 The Cardiac Catheterization Lab
Radiation Safety
The cath lab uses fluoroscopy and cineangiography (digital acquisition) to visualize coronary arteries and cardiac structures. Radiation exposure is measured in air kerma (Gy) — cumulative dose at the interventional reference point — and dose-area product (DAP, Gy·cm²). Key principles include: maintaining maximum distance from the source (inverse square law), using collimation to limit field size, minimizing fluoroscopy time, reducing frame rate (7.5-15 fps vs 30 fps), avoiding steep angulations (which increase tissue penetration depth), and using lead shielding. The sentinel event threshold for skin dose is 5 Gy (risk of deterministic skin injury). Operators should monitor cumulative air kerma in real time during complex cases (CTO, structural procedures).
Operator dose limits: The annual effective dose limit for radiation workers is 50 mSv/year (ALARA principle — As Low As Reasonably Achievable). Cumulative lifetime limit: 10 mSv × age in years. Occupational hazards include cataract formation (lens dose threshold ~500 mGy), thyroid cancer (wear thyroid shield), and left-sided brain tumors (reported in interventional cardiologists — wear lead cap). Recommended protective equipment: lead apron (0.5 mm Pb equivalent), thyroid shield, lead glasses (0.75 mm Pb), ceiling-mounted lead glass shield, under-table lead drape, and radiation dosimeter badges (collar and waist levels).
Vascular Access — Radial vs Femoral
| Feature | Transradial Access (TRA) | Transfemoral Access (TFA) |
|---|---|---|
| Preferred for | ACS, diagnostic cath, standard PCI | Complex PCI (some), structural, MCS |
| Sheath size | 5-7 Fr (slender sheathless 7.5 Fr) | 5-8 Fr (up to 14-24 Fr for structural) |
| Bleeding risk | Lower (MATRIX, RIVAL trials) | Higher — retroperitoneal bleed risk |
| Mortality benefit | Yes in STEMI (MATRIX) | — |
| Crossover rate | 2-7% | <1% |
| Complications | Radial artery occlusion (1-5%), spasm | Pseudoaneurysm, AV fistula, RP bleed |
| Allen test | Recommended (dual palmar arch patency) | Not applicable |
Distal radial (snuffbox) access: Emerging technique with access at the anatomical snuffbox. Advantages include preserved antegrade radial flow, lower radial artery occlusion rates, and patient comfort (no wrist immobilization).
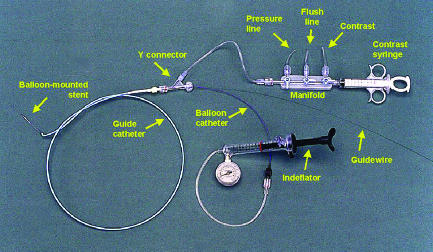
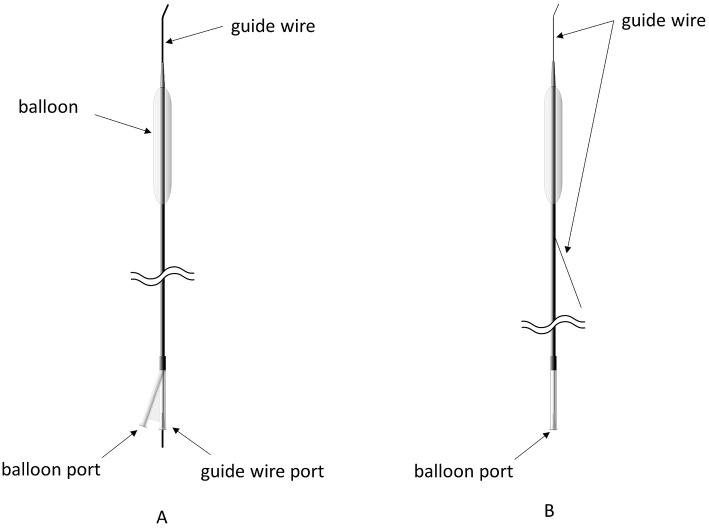
Catheters & Guidewires
Diagnostic catheters: Judkins Left (JL) — most common for LCA (JL 3.5 standard, JL 4.0 for dilated aortic root); Judkins Right (JR) — for RCA (JR 4.0 standard); Amplatz Left (AL) — alternative for LCA or anomalous RCA; Amplatz Right (AR) — for anomalous vessels; Multipurpose — for bypasses and anomalous coronaries. From radial access, Tiger, Jacky, or Ikari catheters can engage both left and right coronary arteries.
Guide catheters: 6 Fr is standard for PCI. EBU (Extra Backup) and XB (Xtra Backup) provide strong support for left coronary interventions. AL1/AL2 provide backup for RCA and anomalous vessels. Guide catheter selection is critical for adequate support in complex PCI — passive support (catheter shape), active support (deep seating), and guide extension catheters (GuideLiner, Trapliner) augment deliverability.
Coronary guidewires: 0.014-inch diameter is standard. Classified by tip stiffness — workhorse (Runthrough, BMW — 1 g tip load), intermediate (Pilot 50 — moderate steerability), stiff/CTO (Confianza Pro 9/12, Gaia 2nd/3rd — 9-12 g tip), and polymer-jacketed (Fielder XT, Gladius — for microchannel tracking). Wire tip shape, torque response, and prolapse resistance determine performance in different lesion types.
Vascular Closure Devices
Used after femoral access to achieve hemostasis and allow early ambulation. Types include: Collagen plug (Angio-Seal — deploys anchor and collagen sandwich), suture-mediated (Perclose ProGlide — pre-close technique for large-bore access up to 24 Fr), clip-based (StarClose), and extravascular sealant (Mynx). The pre-close technique with two Perclose devices deployed at 10 and 2 o'clock positions before sheath upsizing is standard for large-bore femoral access in TAVR and MCS.
03 Hemodynamic Assessment & Right Heart Catheterization
Right Heart Catheterization (Swan-Ganz)
A balloon-tipped pulmonary artery catheter (PAC) is advanced from venous access (internal jugular or femoral vein) through the RA → RV → PA and into the wedge position. Continuous monitoring provides: RA pressure, RV pressure, PA pressure, PCWP (pulmonary capillary wedge pressure), cardiac output, and mixed venous oxygen saturation (SvO2).
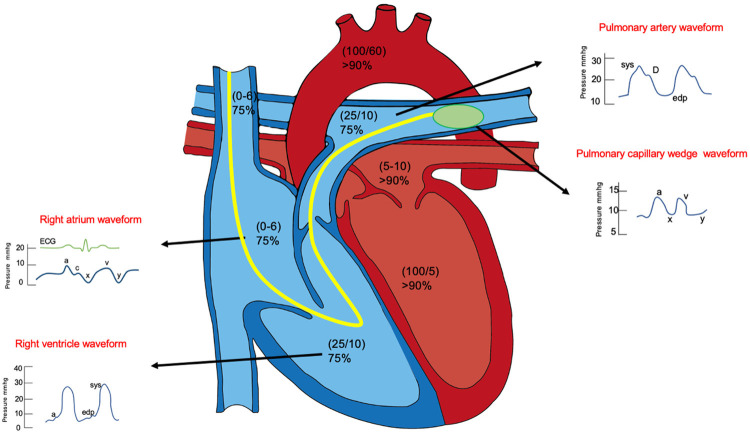
Normal Hemodynamic Values
| Parameter | Normal Range | Units |
|---|---|---|
| RA pressure (mean) | 0-8 | mmHg |
| RV pressure (systolic/diastolic) | 15-30 / 0-8 | mmHg |
| PA pressure (systolic/diastolic/mean) | 15-30 / 4-12 / 9-18 | mmHg |
| PCWP (mean) | 4-12 | mmHg |
| Cardiac output (CO) | 4.0-8.0 | L/min |
| Cardiac index (CI) | 2.5-4.0 | L/min/m² |
| Systemic vascular resistance (SVR) | 800-1200 | dynes·s/cm&sup5; |
| Pulmonary vascular resistance (PVR) | 20-120 (<3 Wood units) | dynes·s/cm&sup5; |
| Mixed venous O2 saturation (SvO2) | 60-80% | — |
Cardiac Output Measurement
Fick method: CO = VO2 / (CaO2 - CvO2), where VO2 is oxygen consumption (ideally measured, often assumed 125 mL/min/m² × BSA), CaO2 is arterial oxygen content, and CvO2 is mixed venous oxygen content. Most accurate when VO2 is directly measured. Thermodilution: Cold saline bolus injected via proximal port; temperature change detected at the PA thermistor generates a curve — CO is inversely proportional to the area under the curve. Less accurate in severe TR, low-output states, and intracardiac shunts.
Shunt Calculations
An intracardiac shunt is detected by an oxygen saturation step-up at the level of the shunt. A step-up of ≥7% at the atrial level (ASD), ≥5% at the ventricular level (VSD), or ≥5% at the PA level (PDA) is considered significant. Qp/Qs ratio = (SaO2 - SvO2) / (SpvO2 - SpaO2). A Qp/Qs ≥ 1.5 generally indicates a hemodynamically significant shunt warranting closure.
Valve Gradient Assessment
Aortic stenosis: Simultaneous measurement of LV and aortic pressures yields the peak-to-peak gradient (catheter gradient) and mean gradient. The Gorlin formula calculates valve area: AVA = CO / (44.3 × SEP × HR × √mean gradient). A Gorlin AVA ≤ 1.0 cm² with mean gradient ≥ 40 mmHg confirms severe AS. In low-flow, low-gradient AS, dobutamine stress hemodynamics help distinguish true-severe from pseudo-severe AS.
Mitral stenosis: The PCWP-to-LV diastolic gradient defines mitral valve gradient. MVA is calculated using the Gorlin formula or estimated by the pressure half-time method on echo. MVA ≤ 1.5 cm² is moderate; ≤ 1.0 cm² is severe.
Pulmonary Hypertension Assessment
Right heart catheterization is the gold standard for diagnosing pulmonary hypertension. Pre-capillary PH: mPAP >20 mmHg, PCWP ≤15 mmHg, PVR ≥3 Wood units. Post-capillary PH (group 2): mPAP >20 mmHg, PCWP >15 mmHg. Combined pre- and post-capillary PH: mPAP >20 mmHg, PCWP >15 mmHg, PVR ≥3 Wood units. Vasoreactivity testing with inhaled nitric oxide (10-20 ppm), IV epoprostenol, or IV adenosine is performed in idiopathic PAH — a positive response (≥10 mmHg decrease in mPAP to ≤40 mmHg with maintained or increased CO) identifies candidates for calcium channel blocker therapy.
PVR calculation: PVR = (mPAP - PCWP) / CO. Normal PVR is <3 Wood units (240 dynes·s/cm&sup5;). Conversion: 1 Wood unit = 80 dynes·s/cm&sup5;. In pre-TAVR assessment, PVR >5 Wood units raises concern for prohibitive surgical risk and may warrant vasodilator challenge to assess reversibility.
Intracardiac Shunt Detection — Oximetry Run
A complete oximetry run involves sampling oxygen saturations from the SVC, IVC, RA, RV, PA, LA (if accessible), LV, and aorta. Significant step-ups indicate left-to-right shunting at the corresponding level:
| Shunt Level | Significant Step-Up | Diagnosis |
|---|---|---|
| SVC → RA | ≥7% O2 sat increase | ASD, partial anomalous pulmonary venous return (PAPVR) |
| RA → RV | ≥5% O2 sat increase | VSD |
| RV → PA | ≥5% O2 sat increase | PDA, aortopulmonary window |
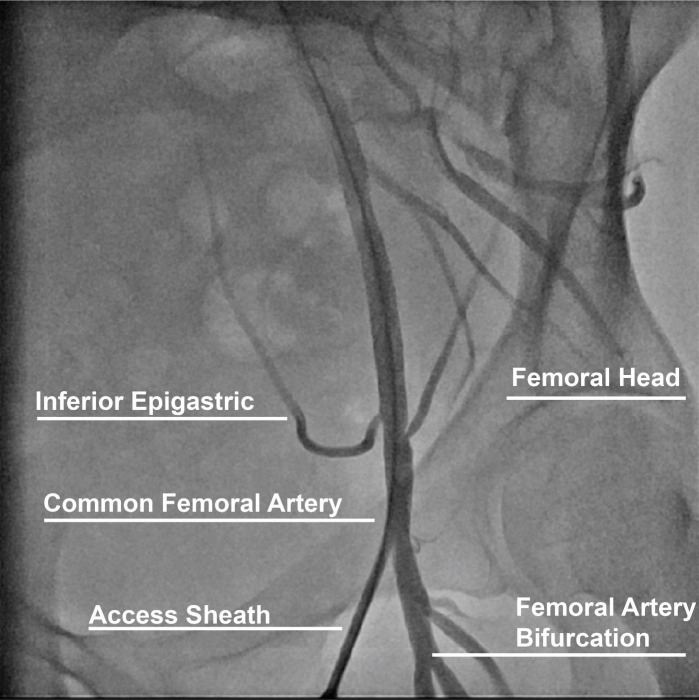
04 Diagnostic Coronary Angiography
Standard Angiographic Views
| View | Angulation | Best Visualizes |
|---|---|---|
| RAO caudal | RAO 20-30°, caudal 20-30° | Left main bifurcation, proximal LAD/LCx |
| RAO cranial | RAO 20-30°, cranial 20-30° | Mid/distal LAD, diagonal branches |
| LAO cranial (spider view) | LAO 40-60°, cranial 20-30° | Left main, LAD/LCx bifurcation, proximal LAD |
| LAO caudal | LAO 40-50°, caudal 20-30° | LCx, obtuse marginals, ramus |
| AP cranial | AP, cranial 30-40° | Mid LAD, diagonal branches, LAD/septal perforators |
| AP caudal | AP, caudal 20-30° | Left main, proximal LCx, ramus |
| LAO straight | LAO 30-45° | RCA (mid), crux, PDA |
| RAO straight | RAO 30° | RCA (proximal/mid), PDA bifurcation |
Stenosis Grading & Quantitative Coronary Angiography (QCA)
Visual estimation of diameter stenosis remains standard but has significant interobserver variability. QCA provides objective measurements: reference vessel diameter (RVD), minimum lumen diameter (MLD), percent diameter stenosis (%DS), and lesion length. The threshold for "significant" stenosis is generally ≥70% diameter stenosis for epicardial coronary arteries and ≥50% for the left main.
TIMI Flow Grade
| Grade | Definition |
|---|---|
| TIMI 0 | No perfusion — no antegrade flow beyond the point of occlusion |
| TIMI 1 | Penetration without perfusion — contrast passes beyond obstruction but fails to opacify the distal bed |
| TIMI 2 | Partial perfusion — contrast opacifies the distal bed but with delayed filling or clearance |
| TIMI 3 | Complete perfusion — antegrade flow fills the distal bed completely with normal clearance |
TIMI Myocardial Perfusion Grade (TMPG)
Assesses microvascular perfusion independent of epicardial flow: TMPG 0 — no myocardial blush; TMPG 1 — slow entry, staining persists on next injection; TMPG 2 — slow entry and exit; TMPG 3 — normal entry and exit. TMPG 3 after primary PCI is associated with the lowest mortality.
SYNTAX Score
The SYNTAX (Synergy Between PCI with Taxus and Cardiac Surgery) score is an angiographic tool that quantifies the complexity of coronary artery disease based on lesion location, severity, bifurcation involvement, calcification, tortuosity, thrombus, and vessel characteristics across all segments. Low SYNTAX (≤22): PCI and CABG equivalent; Intermediate (23-32): discuss with Heart Team; High (≥33): CABG preferred. The SYNTAX II score incorporates clinical variables (age, LVEF, CrCl, COPD, PVD) to improve prognostic accuracy.
05 Percutaneous Coronary Intervention (PCI)
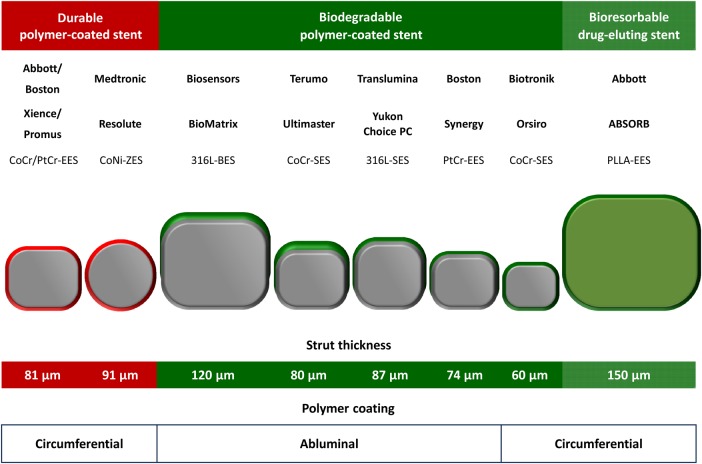
Evolution of Coronary Stents
| Generation | Examples | Drug / Polymer | Key Features |
|---|---|---|---|
| POBA | Plain old balloon angioplasty | None | 30-40% restenosis rate; acute vessel closure risk |
| BMS | Palmaz-Schatz, Liberté | None / bare metal | Reduced acute closure vs POBA; 20-30% ISR |
| 1st gen DES | Cypher (sirolimus), Taxus (paclitaxel) | Sirolimus/paclitaxel; durable polymer | Reduced ISR to ~5-8%; concerns for VLST |
| 2nd gen DES | Xience (everolimus), Resolute (zotarolimus) | Everolimus/zotarolimus; thinner polymer | Current standard; thinner struts (81 μm); lower VLST |
| 3rd gen DES | Orsiro (sirolimus, biodegradable polymer), Synergy (everolimus, abluminal) | Ultrathin struts; bioabsorbable polymer | 60 μm struts; rapid endothelialization |
| BVS | Absorb (everolimus, PLLA scaffold) | Everolimus; bioresorbable scaffold | Withdrawn — increased scaffold thrombosis (ABSORB III) |

PCI Technique — Step-by-Step
1. Guide catheter engagement — Coaxial alignment with the coronary ostium; adequate backup support. 2. Wiring the lesion — Advance a 0.014" guidewire across the stenosis into the distal vessel. 3. Predilation — Compliant balloon (sized 1:1 to reference vessel or slightly undersized) inflated across the lesion to create a channel for stent delivery. 4. Stent deployment — Select stent diameter to match reference vessel (by QCA or intravascular imaging); stent length should cover the lesion with 2-3 mm margin on each side. Inflate at nominal pressure (8-12 atm) for deployment. 5. Post-dilation — Non-compliant balloon (NC) inflated at high pressure (14-20 atm) to ensure full stent expansion and apposition; sized 1:1 to the reference vessel. 6. Final angiography — Confirm TIMI 3 flow, no dissection, no residual stenosis, good distal runoff.
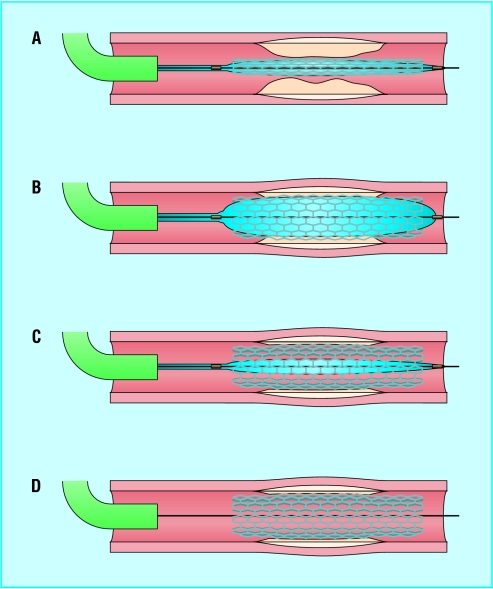
Stent Sizing Principles
Stent diameter should match the distal reference vessel diameter by angiography (or the media-to-media diameter by IVUS/OCT). Stent undersizing leads to malapposition (risk of stent thrombosis); oversizing leads to edge dissection and vessel injury. IVUS-guided PCI targets minimum stent area (MSA) ≥ 5.5 mm² in non-left-main vessels and ≥ 8.0 mm² in the left main. OCT criteria include: stent expansion ≥80% of reference lumen area, no edge dissection >200 μm, and complete stent apposition.
Kissing Balloon Inflation
Simultaneous inflation of two balloons in the main vessel and side branch at bifurcation stenting. Purpose: correct carina shift, maintain side branch patency, optimize stent apposition at the bifurcation. POT-side-POT (proximal optimization technique) is an alternative strategy: short NC balloon inflated in the proximal main vessel to flare the stent, followed by wire rewiring and kissing balloon or POT.
Specialty Balloons
| Balloon Type | Properties | Use |
|---|---|---|
| Compliant | Grows with increasing pressure; conforms to vessel shape | Predilation; lesion assessment |
| Semi-compliant | Moderate growth with pressure; used on stent delivery systems | Stent deployment (nominal pressure) |
| Non-compliant (NC) | Minimal growth beyond rated burst pressure; predictable diameter | Post-dilation; high-pressure expansion of underexpanded stents |
| Scoring/cutting balloon | Atherotome blades or wires on surface; concentrate force on plaque | Fibrotic/resistant lesions; ISR; prevent balloon slippage |
| Drug-coated balloon (DCB/DEB) | Paclitaxel or sirolimus coating; local drug delivery without permanent implant | ISR (AGENT IDE); small vessels; de novo lesions (BASKET-SMALL 2) |
| Intravascular lithotripsy (IVL) | Sonic pressure waves fracture calcium via Shockwave emitters | Severely calcified lesions; superficial and deep calcium |
Guide Extension Catheters
GuideLiner (Teleflex) and Guidezilla (Boston Scientific) are rapid-exchange coaxial guide extension catheters that telescope within the guide catheter and extend ~25 cm into the coronary artery. They provide enhanced backup support for device delivery in tortuous anatomy, calcified lesions, and distal lesions. The "trapping" technique (inflating a balloon in the guide to trap a wire) allows wire exchange through the extension catheter without losing position. Guide extensions also serve as conduits for delivering thrombus aspiration catheters, coils, and covered stents to distal locations.
06 STEMI Intervention & Cardiogenic Shock
Primary PCI for STEMI
Primary PCI is the preferred reperfusion strategy for STEMI when it can be performed within 120 minutes of first medical contact (door-to-balloon time target <90 minutes at PCI-capable hospitals; <120 minutes with transfer). Emergency Primary PCI achieves TIMI 3 flow in >90% of cases, compared to ~50-60% with fibrinolysis. Radial access is preferred (MATRIX trial — reduced bleeding and mortality in STEMI).
Culprit Lesion PCI vs Complete Revascularization
In STEMI with multivessel disease, culprit-lesion-only PCI is the standard acute approach. The COMPLETE trial demonstrated that staged complete revascularization of non-culprit lesions (during index hospitalization or within 45 days) reduced the composite of cardiovascular death and MI compared to culprit-only PCI. FFR-guided non-culprit PCI is supported by the DANAMI-3-PRIMULTI and Compare-Acute trials. Non-culprit PCI should generally not be performed at the time of primary PCI unless there is hemodynamic instability, ongoing ischemia, or cardiogenic shock.
Thrombus Management
Large thrombus burden is common in STEMI. Routine aspiration thrombectomy is no longer recommended (TOTAL, TASTE trials — no mortality benefit, increased stroke risk). Aspiration may be considered as a bailout strategy for massive thrombus burden with TIMI 0-1 flow despite wire crossing. GP IIb/IIIa inhibitors (abciximab, eptifibatide) may be used as bailout for large thrombus burden or no-reflow.
Cardiogenic Shock — STEMI
Emergency Cardiogenic shock complicates 5-10% of STEMI cases and carries 40-50% in-hospital mortality. The SHOCK trial established early revascularization (PCI or CABG within 6 hours) as the standard of care — 6-month mortality reduction from 63% to 50%. Key management principles:
Hemodynamic support: Vasopressors (norepinephrine preferred over dopamine — SOAP II trial), inotropes (dobutamine, milrinone), and mechanical circulatory support (MCS) — see Section 16. The SCAI shock classification (stages A-E) guides escalation of support. Stage C (classic shock: hypotension, CI <2.2, PCWP >15, lactate rising) should prompt MCS consideration. Stage D (deteriorating) and Stage E (extremis — refractory cardiac arrest) require emergent MCS or ECMO.
Mechanical Complications of STEMI
Mechanical complications typically occur 2-7 days post-MI (earlier with reperfusion). All are surgical emergencies:
| Complication | Timing | Presentation | Diagnosis | Management |
|---|---|---|---|---|
| Free wall rupture | Days 3-7 | Sudden PEA, tamponade | Echo: pericardial effusion, tamponade | Emergency Pericardiocentesis → emergent surgery |
| Ventricular septal rupture (VSR) | Days 3-5 | New harsh holosystolic murmur, acute HF | Echo with color Doppler; PA sat step-up | MCS (IABP/Impella) → surgical repair (delayed if possible) |
| Papillary muscle rupture | Days 2-7 | Acute severe MR, pulmonary edema, shock | Echo: flail mitral leaflet, severe MR | Emergency MCS → emergent MVR/repair |
Papillary muscle rupture most commonly involves the posteromedial papillary muscle (single blood supply from PDA — affected in inferior MI) rather than the anterolateral papillary muscle (dual supply from LAD and LCx). Complete rupture is rapidly fatal without surgical intervention. Partial rupture may present subacutely.
07 Complex PCI — CTO, Bifurcation & Left Main
Chronic Total Occlusion (CTO) PCI
A CTO is defined as a coronary occlusion with TIMI 0 flow for ≥3 months. CTO PCI is among the most technically demanding coronary interventions. The hybrid algorithm guides strategy selection based on four angiographic features: (1) ambiguity of the proximal cap, (2) lesion length (>20 mm), (3) presence of an interventional collateral for retrograde access, and (4) quality of the distal vessel target.
Antegrade wire escalation (AWE): First-line strategy for short CTOs (<20 mm) with a clear proximal cap. Escalation from soft-tipped wires through intermediate to stiff, tapered CTO wires (Confianza Pro 12, Gaia 3rd). Antegrade dissection re-entry (ADR): For long CTOs (>20 mm). A knuckled polymer wire is advanced into the subintimal space, and a re-entry device (Stingray balloon) redirects back into the true lumen distally. Retrograde approach: A wire is passed through a collateral channel (septal or epicardial) to the distal cap of the CTO. Techniques include reverse CART (Controlled Antegrade and Retrograde subintimal Tracking) — the most common retrograde strategy.
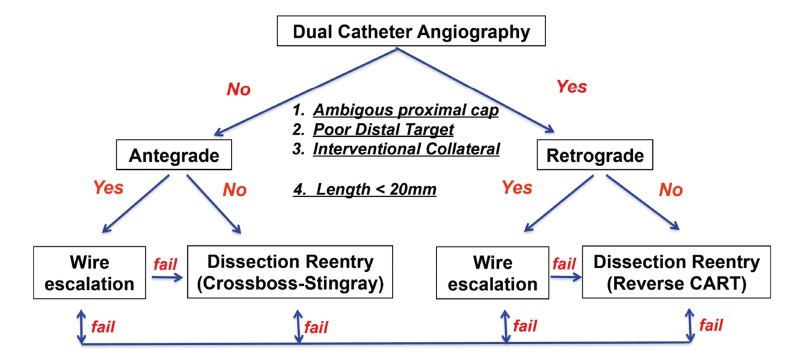
CTO PCI success rates at experienced centers exceed 85-90%. The J-CTO score predicts procedural difficulty: 0 = easy, 1 = intermediate, 2 = difficult, ≥3 = very difficult. Variables scored: blunt proximal cap, calcification, bending >45°, occlusion length >20 mm, and prior failed attempt.
CTO Crossing Strategies — Detailed
| Strategy | When to Use | Key Steps | Success Rate |
|---|---|---|---|
| Antegrade Wire Escalation (AWE) | Short CTO (<20 mm), clear proximal cap, no suitable collaterals | Escalate wire tip stiffness: workhorse → polymer-jacketed → tapered stiff (Gaia 2nd, Confianza Pro 12) | 60-70% as primary strategy |
| Antegrade Dissection Re-entry (ADR) | Long CTO (>20 mm), clear proximal cap, no collaterals | Knuckle wire into subintima → Stingray balloon for re-entry → confirm true lumen with IVUS | 70-80% |
| Retrograde via septal collateral | Ambiguous proximal cap; good septal collateral; prior failed antegrade | Corsair/Caravel microcatheter through septal channel → reverse CART → externalize wire | 75-85% |
| Retrograde via epicardial collateral | No septal collaterals available | Higher risk of donor vessel injury; Sion wire preferred for tortuosity | Lower; higher complication rate |
Reverse CART (Controlled Antegrade and Retrograde subintimal Tracking): The most common retrograde crossing technique. An antegrade knuckle wire creates a subintimal space; the retrograde wire punctures into this space, connecting the retrograde and antegrade paths. A balloon is inflated over the antegrade wire to enlarge the connection point, allowing the retrograde wire to cross into the antegrade guide catheter. The retrograde wire is then externalized (pulled through the antegrade guide) and used as a rail for stent delivery.
Bifurcation PCI
Bifurcation lesions account for 15-20% of PCI. The Medina classification describes involvement of the proximal main vessel, distal main vessel, and side branch (1 = ≥50% stenosis, 0 = <50%): e.g., Medina 1,1,1 = all three segments diseased; Medina 1,1,0 = main vessel only.
Provisional stenting is the default strategy: stent the main vessel, then assess the side branch. If the side branch has ≥75% stenosis, TIMI <3 flow, or threatened territory >10% of myocardium, perform side branch intervention (wire, balloon, or T-stent). Two-stent strategies are reserved for true bifurcations with large side branches:
| Technique | Description | Best For |
|---|---|---|
| Crush (DK-crush) | Side branch stent crushed by main vessel stent; sequential kissing and POT | True bifurcations; best evidence (DKCRUSH-V) |
| Culotte | Both stents protrude into the main vessel; full coverage of the carina | Y-shaped bifurcations with similar vessel sizes |
| TAP (T and Protrusion) | Side branch stent with minimal protrusion into main vessel | Wide-angle bifurcations |
| V-stenting / Simultaneous kissing stents | Two stents deployed simultaneously in a V configuration | Distal left main bifurcation |
Left Main PCI
Left main PCI is an alternative to CABG in selected patients. The EXCEL trial (5-year follow-up) showed no significant difference in the composite of death, MI, or stroke between PCI and CABG for left main disease with low-intermediate SYNTAX scores (≤32), though CABG had lower rates of repeat revascularization. The NOBLE trial favored CABG at 5 years, driven by higher non-procedural MI and repeat revascularization with PCI. Current guidelines recommend Heart Team discussion for all left main decisions.
Technical considerations: Distal left main bifurcation (most common location) is treated based on LCx ostial involvement — provisional stenting if LCx not significantly diseased, DK-crush or culotte if both LAD and LCx require stenting. Intravascular imaging is strongly recommended (class IIa). IVUS MSA targets: ≥8 mm² in the left main body, ≥7 mm² at the polygon of confluence, ≥6 mm² in the LAD ostium, ≥5 mm² in the LCx ostium.
Calcified Lesion Modification
Severe calcification impairs stent delivery and expansion. Calcium modification techniques:
| Technique | Mechanism | Key Considerations |
|---|---|---|
| Rotational atherectomy (RA) | Diamond-coated burr (1.25-2.5 mm) ablates calcium at 135,000-180,000 rpm | Burr:artery ratio ≤0.7; avoid decelerations >5,000 rpm; risk of slow-flow/no-reflow |
| Orbital atherectomy (OA) | Diamond-coated crown sands calcium at 80,000-120,000 rpm via centrifugal force | Single crown size treats varying lumens; bidirectional; reduced no-reflow risk |
| Intravascular lithotripsy (IVL) | Shockwave balloon emits pulsatile sonic waves that fracture calcium | Balloon sized 1:1 to vessel; treats deep (medial) calcium; easiest to use; DISRUPT CAD III data |
| Laser atherectomy | Excimer laser ablates tissue via photochemical, photothermal, and photomechanical effects | Used for ISR with underexpanded stents; also for lead extraction |
08 Intravascular Imaging & Coronary Physiology
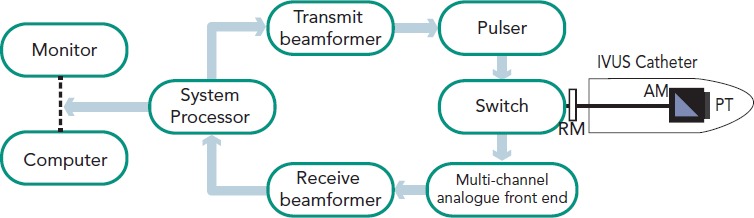
Intravascular Ultrasound (IVUS)
IVUS uses a miniaturized ultrasound transducer (20-60 MHz) on a catheter to produce cross-sectional images of the vessel wall. Resolution: axial ~100 μm, lateral ~200 μm. IVUS penetrates through calcium (unlike OCT) and visualizes the full vessel wall thickness. Grayscale IVUS identifies plaque burden, lumen area, external elastic membrane (EEM) area, and stent expansion. Virtual histology (VH-IVUS) uses radiofrequency backscatter analysis to characterize plaque composition (fibrous, fibrofatty, necrotic core, dense calcium).
IVUS-guided PCI targets: MSA ≥5.5 mm² (non-left main), MSA ≥8.0 mm² (left main body), no edge dissection involving the media, stent expansion ≥80% of reference lumen, and absence of geographic miss (lesion extending beyond stent edges). The ULTIMATE trial and ADAPT-DES study confirmed IVUS guidance reduces MACE compared to angiography alone.
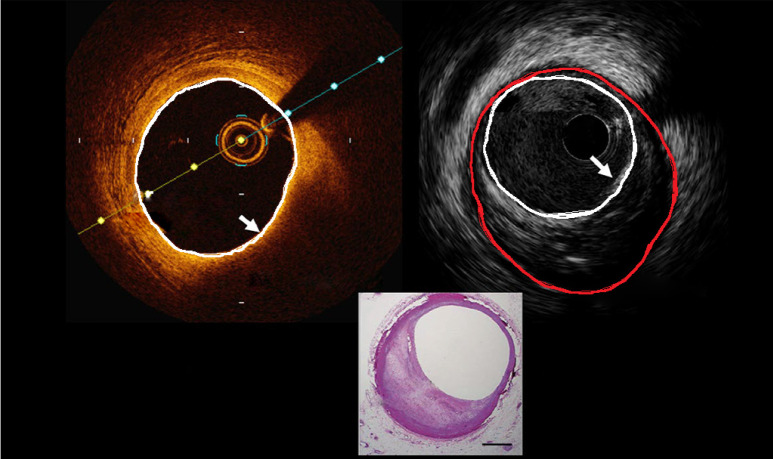
Optical Coherence Tomography (OCT)
OCT uses near-infrared light (1,300 nm wavelength) to produce ultra-high-resolution images of the vessel wall. Resolution: axial ~10-15 μm (10x better than IVUS), lateral ~20-40 μm. Limitation: poor tissue penetration (~1-2 mm) — cannot see through calcium or deep plaque; requires blood clearance (contrast flush). OCT excels at identifying: thin-cap fibroatheroma (TCFA) — cap thickness <65 μm overlying a lipid-rich core (vulnerable plaque); stent malapposition (strut-to-intima distance >strut thickness + polymer); stent underexpansion; edge dissection (flap >200 μm or involving the media); thrombus (red vs white); and neoatherosclerosis (lipid-laden intima within stents).
The ILUMIEN IV trial demonstrated that OCT-guided PCI achieved larger MSA and lower rates of stent underexpansion compared to angiography-guided PCI, though the primary clinical endpoint was not significantly different at 2 years.
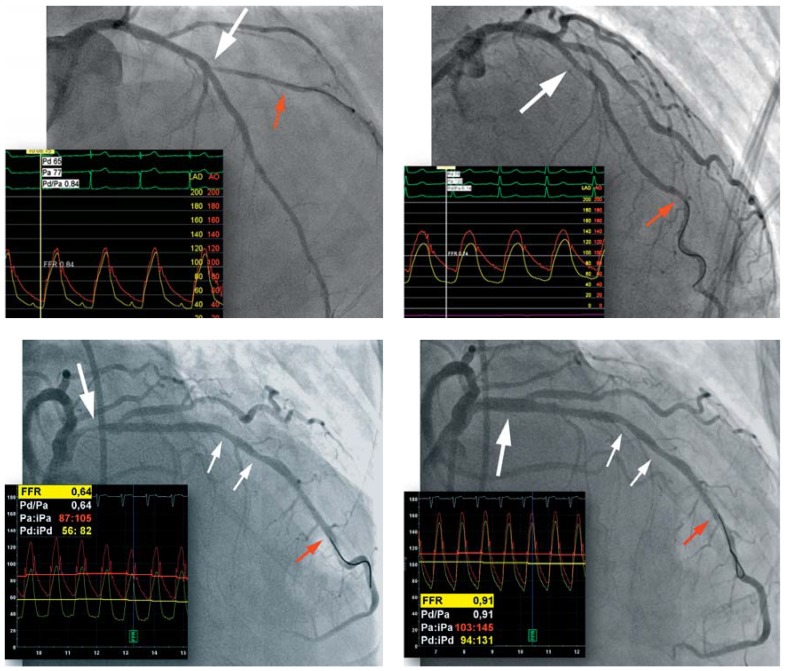
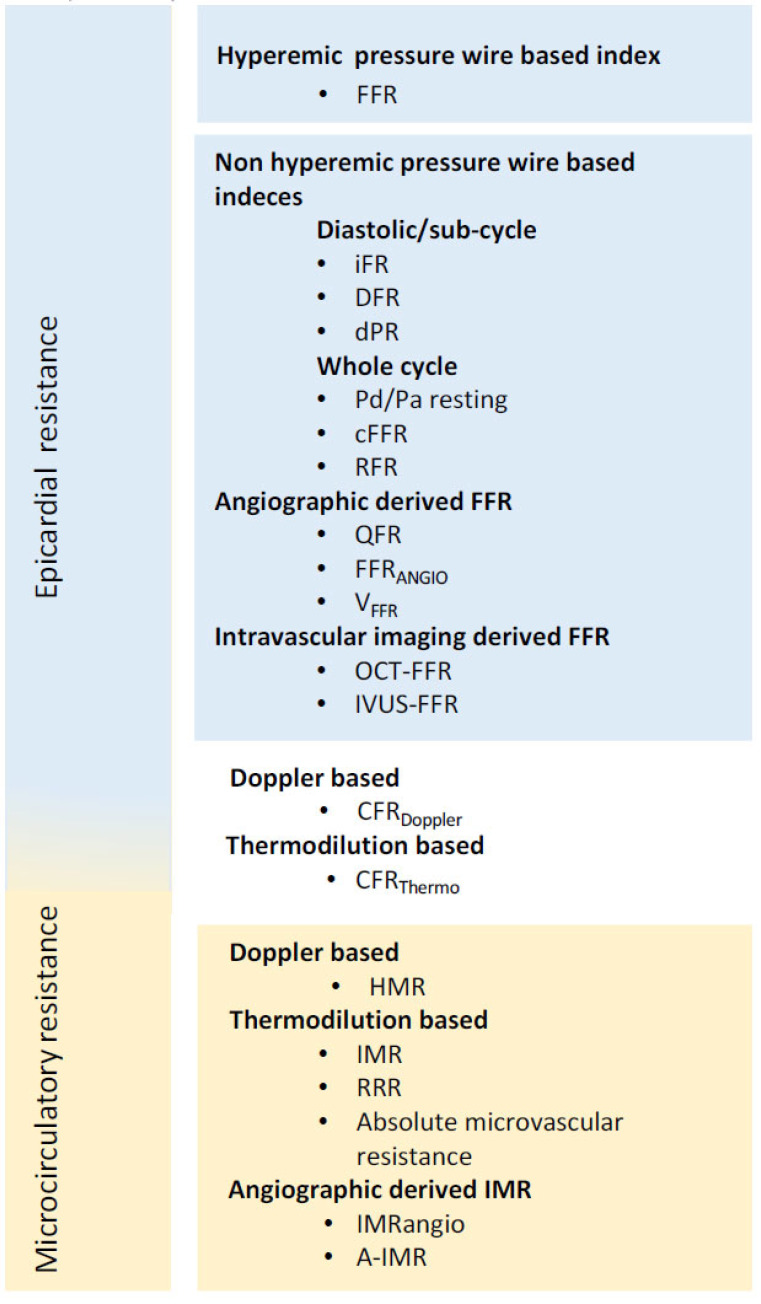
Fractional Flow Reserve (FFR)
FFR is the ratio of distal coronary pressure (Pd) to aortic pressure (Pa) at maximal hyperemia (adenosine 140 μg/kg/min IV or IC bolus). FFR = Pd/Pa. An FFR ≤0.80 indicates a hemodynamically significant stenosis warranting revascularization. Landmark trials:
FAME — FFR-guided PCI vs angiography-guided PCI in multivessel disease: FFR guidance reduced death, MI, and repeat revascularization at 1 year. FAME 2 — FFR-guided PCI + OMT vs OMT alone for FFR ≤0.80: PCI reduced urgent revascularization (trial stopped early); no mortality difference at 5 years. DEFER — Deferring PCI when FFR >0.75 was safe at 15-year follow-up.
Non-Hyperemic Pressure Ratios (NHPR)
iFR (instantaneous wave-free ratio) and RFR (resting full-cycle ratio) measure the Pd/Pa ratio during a specific diastolic window (iFR) or the lowest Pd/Pa during the entire cardiac cycle (RFR), without requiring hyperemia. An iFR/RFR ≤0.89 indicates hemodynamic significance. The DEFINE-FLAIR and iFR-SWEDEHEART trials demonstrated non-inferiority of iFR-guided revascularization compared to FFR-guided revascularization at 1 year.
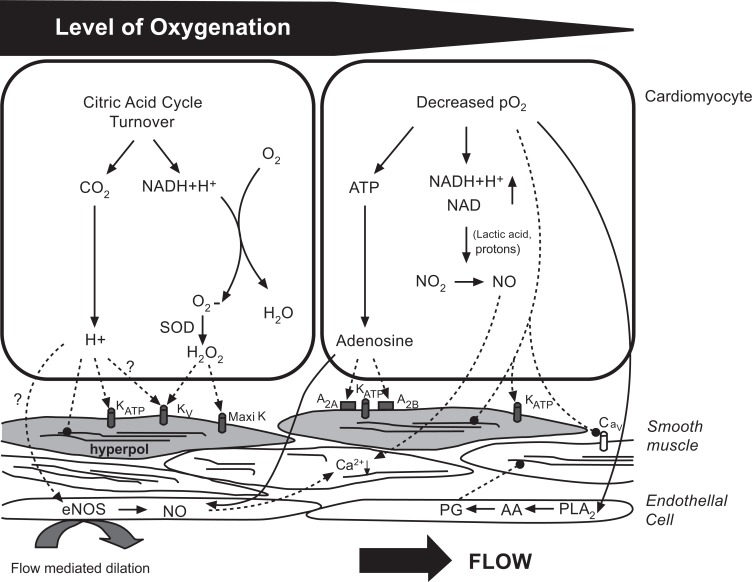
Coronary Flow Reserve (CFR) & Index of Microcirculatory Resistance (IMR)
CFR is the ratio of hyperemic to resting coronary flow velocity (measured by Doppler wire) or transit time (thermodilution). CFR <2.0 indicates impaired flow reserve, which may reflect epicardial stenosis, microvascular disease, or both. IMR = Pd × transit time at hyperemia; IMR ≥25 indicates microvascular dysfunction. In STEMI, elevated IMR post-PCI predicts larger infarct size and worse LV recovery. Combined assessment of FFR + CFR + IMR allows differentiation between epicardial disease, microvascular disease, and normal hemodynamics — the endotype classification guides targeted therapy.
Angioscopy & Near-Infrared Spectroscopy (NIRS)
NIRS (TVC Imaging System, Infraredx/Lipid-Rich Plaque) identifies lipid-core plaques within the vessel wall by measuring the absorption of near-infrared light. A lipid core burden index (LCBI) at the maximal 4 mm segment (maxLCBI4mm) ≥400 is associated with increased risk of periprocedural MI during PCI and future MACE (LRP study). NIRS is often combined with IVUS on a single catheter platform (Makoto, TVC) for simultaneous structural and compositional assessment.
09 Acute Coronary Syndromes — Antithrombotic Management
NSTEMI — Timing of Catheterization
Risk stratification guides timing: Immediate (<2 hours) — hemodynamic instability, ongoing chest pain despite medical therapy, life-threatening arrhythmias, mechanical complications. Early (<24 hours) — GRACE score >140, dynamic ST/T changes, rise/fall in troponin (TIMACS trial — early invasive strategy reduced the composite of death, MI, stroke in high-risk patients). Delayed invasive (24-72 hours) — lower-risk patients without high-risk features. Selective invasive (ischemia-guided) — low-risk patients with no recurrent symptoms and negative stress test.
Antiplatelet Therapy
| Agent | Mechanism | Loading / Maintenance | Key Trial Data |
|---|---|---|---|
| Aspirin | Irreversible COX-1 inhibition | 325 mg load / 81 mg daily | Cornerstone of antiplatelet therapy; lifelong after ACS |
| Clopidogrel | Irreversible P2Y12 inhibition (prodrug) | 600 mg load / 75 mg daily | CURE, CURRENT-OASIS 7; 3-15% poor metabolizers (CYP2C19) |
| Prasugrel | Irreversible P2Y12 inhibition (prodrug) | 60 mg load / 10 mg daily | TRITON-TIMI 38; contraindicated if prior stroke/TIA; avoid if ≥75 years or <60 kg |
| Ticagrelor | Reversible P2Y12 inhibition (direct-acting) | 180 mg load / 90 mg BID | PLATO; side effects: dyspnea, bradycardia; avoid with strong CYP3A4 inhibitors |
| Cangrelor | IV reversible P2Y12 inhibition | 30 μg/kg bolus + 4 μg/kg/min infusion | CHAMPION PHOENIX; onset 2 min, offset 60 min; bridging agent |
DAPT Duration
Standard: 12 months of DAPT (aspirin + P2Y12 inhibitor) after ACS regardless of stent type. Extended (>12 months): Consider in high ischemic risk / low bleeding risk (PEGASUS-TIMI 54 — ticagrelor 60 mg BID beyond 12 months reduced CV events). Shortened (3-6 months): Consider in high bleeding risk (TWILIGHT — ticagrelor monotherapy after 3 months of DAPT reduced bleeding without increasing ischemic events; STOPDAPT-2 — 1 month DAPT followed by clopidogrel monotherapy). The DAPT score (≥2 favors prolonged DAPT; <2 favors shortened) incorporates: age, diabetes, smoking, prior MI/PCI, stent diameter <3 mm, CHF/LVEF <30%, paclitaxel-eluting stent, and vein graft PCI.
Anticoagulation in ACS
Unfractionated heparin (UFH): Standard during PCI — 70-100 U/kg bolus (50-70 U/kg if GP IIb/IIIa used); target ACT 250-350 s (200-250 s with GP IIb/IIIa). Bivalirudin: Direct thrombin inhibitor — 0.75 mg/kg bolus + 1.75 mg/kg/h infusion; alternative to UFH, especially in high bleeding risk patients (HORIZONS-AMI, EUROMAX). No need for ACT monitoring. Enoxaparin: LMWH — 0.5-1 mg/kg SC or IV; if used pre-cath, additional 0.3 mg/kg IV bolus if last dose >8 hours ago. Fondaparinux: Factor Xa inhibitor — 2.5 mg SC daily; if used pre-cath, must supplement with UFH during PCI (OASIS-5 — catheter thrombosis risk without supplemental UFH).
GP IIb/IIIa Inhibitors
Routine upstream use is no longer recommended. Bailout use is appropriate for: large thrombus burden, no-reflow, threatened vessel closure, or slow flow. Agents: Abciximab (0.25 mg/kg bolus + 0.125 μg/kg/min × 12 h), eptifibatide (180 μg/kg double bolus + 2 μg/kg/min × 18-24 h), tirofiban (25 μg/kg bolus + 0.15 μg/kg/min × 18-24 h). Eptifibatide and tirofiban require dose adjustment in renal impairment.
10 Stent Complications & No-Reflow
In-Stent Restenosis (ISR)
ISR is defined as ≥50% diameter stenosis within the stent or within 5 mm of the stent edges. It is caused by neointimal hyperplasia (smooth muscle cell proliferation and extracellular matrix deposition) and, in late ISR, neoatherosclerosis. ISR rates: BMS 20-30% at 1 year; first-gen DES 5-10%; second/third-gen DES 3-5%. The Mehran classification describes ISR patterns: Type I (focal, ≤10 mm), Type II (intra-stent, >10 mm), Type III (proliferative, extending beyond stent edges), Type IV (total occlusion).
Treatment algorithm for ISR: Step 1 — Perform IVUS or OCT to identify mechanical causes (stent underexpansion is the most common correctable cause). Step 2 — If underexpanded, high-pressure NC balloon inflation (≥20 atm) or IVL to crack calcified plaque restricting expansion. Step 3 — If adequately expanded, treat the neointimal tissue: Drug-eluting balloon (DEB) — preferred for focal ISR (AGENT IDE trial — sirolimus DEB showed 76% freedom from TLR at 12 months); DES (different drug) — preferred for diffuse ISR (RIBS IV — everolimus DES superior to DEB for diffuse ISR); Laser atherectomy or rotational atherectomy — for severely underexpanded stents with circumferential calcium that cannot be dilated with balloons.
Stent fracture is an underrecognized cause of ISR and stent thrombosis. Risk factors: long stents, hinge points (RCA), overlapping stents, and right coronary artery location. Diagnosis: fluoroscopic "gap" in stent struts, confirmed by IVUS/OCT showing strut discontinuity. Treatment: additional DES deployment across the fracture site.
Stent Thrombosis
Stent thrombosis is a catastrophic event presenting as acute MI with high mortality (20-45%). The Academic Research Consortium (ARC) classification defines certainty (definite, probable, possible) and timing:
| Timing | Interval | Primary Mechanism |
|---|---|---|
| Acute | 0-24 hours | Procedural factors (dissection, malapposition, underexpansion) |
| Subacute | 1-30 days | DAPT non-compliance, platelet reactivity |
| Late | 30 days - 1 year | Delayed endothelialization, polymer hypersensitivity |
| Very late (VLST) | >1 year | Neoatherosclerosis, late malapposition, polymer degradation |
Risk factors: premature DAPT discontinuation (most important modifiable factor), stent underexpansion, edge dissection, residual stenosis, bifurcation stenting, long stents, small vessel diameter, diabetes, renal failure, and low LVEF. Treatment: emergent PCI with IVUS/OCT to identify and correct the mechanical cause.
Saphenous Vein Graft (SVG) Intervention
SVG PCI carries higher risk of distal embolization and no-reflow due to the friable, degenerated plaque within vein grafts. The SAFER trial demonstrated that a distal embolic protection device (EPD) — FilterWire or SpiderFX — reduces 30-day MACE by 42% during SVG PCI. Key principles: Always use embolic protection (class I recommendation); avoid direct stenting without predilation (risk of distal embolization from bulky plaque); use DES over BMS (lower restenosis rates); keep stent margins away from the aortic anastomosis; and avoid GP IIb/IIIa inhibitors as first-line (no proven benefit in SVG PCI; increase bleeding). For severely degenerated SVGs with diffuse disease, consider native vessel PCI (including CTO PCI of the native vessel) as an alternative to SVG intervention.
Coronary Perforation
The Ellis classification grades perforation severity:
| Type | Description | Management |
|---|---|---|
| Type I | Extraluminal crater without extravasation | Observation; usually self-limited |
| Type II | Myocardial or pericardial blush without contrast jet | Prolonged balloon inflation; reversal of anticoagulation; observation |
| Type III | Contrast streaming through a frank perforation (≥1 mm) | Emergency Covered stent (PK Papyrus, Graftmaster); pericardiocentesis if tamponade; surgical backup |
| Cavity spilling | Perforation into a cardiac chamber | Covered stent or coil embolization; surgical repair if refractory |
No-Reflow Phenomenon
No-reflow is defined as TIMI ≤2 flow in the absence of epicardial obstruction (dissection, thrombus, spasm, or residual stenosis). It is caused by distal embolization of thrombus/plaque debris, microvascular spasm, and reperfusion injury. Occurs in 2-5% of PCI and up to 10-30% of primary PCI for STEMI. Treatment: Intracoronary adenosine (100-200 μg boluses), nitroprusside (100-200 μg IC), verapamil (100-200 μg IC), nicardipine (200 μg IC), and epinephrine (50-200 μg IC for refractory cases). GP IIb/IIIa inhibitors may be used if thrombus burden is suspected. Distal protection devices (FilterWire, SpiderFX) are recommended for SVG interventions (SAFER trial) but not for native coronary PCI.
11 Transcatheter Aortic Valve Replacement (TAVR)
Indications & Evidence
TAVR is approved for symptomatic severe aortic stenosis across the surgical risk spectrum. Landmark trials by risk category:
| Risk Category | Key Trials | Findings |
|---|---|---|
| Inoperable / extreme risk | PARTNER 1B (Edwards SAPIEN) | TAVR reduced mortality by 20% at 1 year vs medical therapy |
| High risk | PARTNER 1A (SAPIEN), CoreValve US Pivotal | TAVR non-inferior (PARTNER 1A) or superior (CoreValve) to SAVR at 1 year |
| Intermediate risk | PARTNER 2A (SAPIEN 3), SURTAVI (Evolut R) | TAVR non-inferior to SAVR; lower stroke/bleeding with TAVR |
| Low risk | PARTNER 3 (SAPIEN 3), Evolut Low Risk | TAVR non-inferior to SAVR at 1 year; PARTNER 3 showed superiority of TAVR at 1 year (composite of death, stroke, rehospitalization) |
Device Platforms
Edwards SAPIEN 3 / SAPIEN 3 Ultra: Balloon-expandable bovine pericardial valve; cobalt-chromium frame; sizes 20, 23, 26, 29 mm; outer skirt to reduce paravalvular leak; 14-16 Fr eSheath (expandable). Medtronic CoreValve Evolut R / Evolut PRO / Evolut FX: Self-expanding porcine pericardial valve; nitinol frame; sizes 23, 26, 29, 34 mm; supra-annular leaflet position; recapturable and repositionable; 14 Fr InLine sheath.
CT Planning for TAVR
Multidetector CT is the gold standard for pre-procedural planning: Annular sizing — perimeter-derived diameter (most accurate), area-derived diameter, and mean diameter determine valve size; Coronary heights — distance from annulus to left and right coronary ostia (risk of coronary obstruction if <12 mm with SAPIEN or <10 mm with self-expanding); Sinus of Valsalva dimensions — small sinuses increase coronary obstruction risk; Calcium scoring — valve and LVOT calcium distribution predict paravalvular leak, conduction disturbance, and annular rupture risk; Iliofemoral access — minimum diameter ≥5.5 mm for current-generation devices; tortuosity and calcification assessment.
TAVR Access Routes
Transfemoral (TF): Preferred approach (>95% of cases); lowest morbidity. Alternative routes (when TF not feasible): transaxillary/subclavian (surgical cutdown or percutaneous), transaortic (mini-sternotomy), transapical (left thoracotomy — largely abandoned due to higher mortality), transcaval (IVC-to-aorta puncture — emerging), transcarotid.
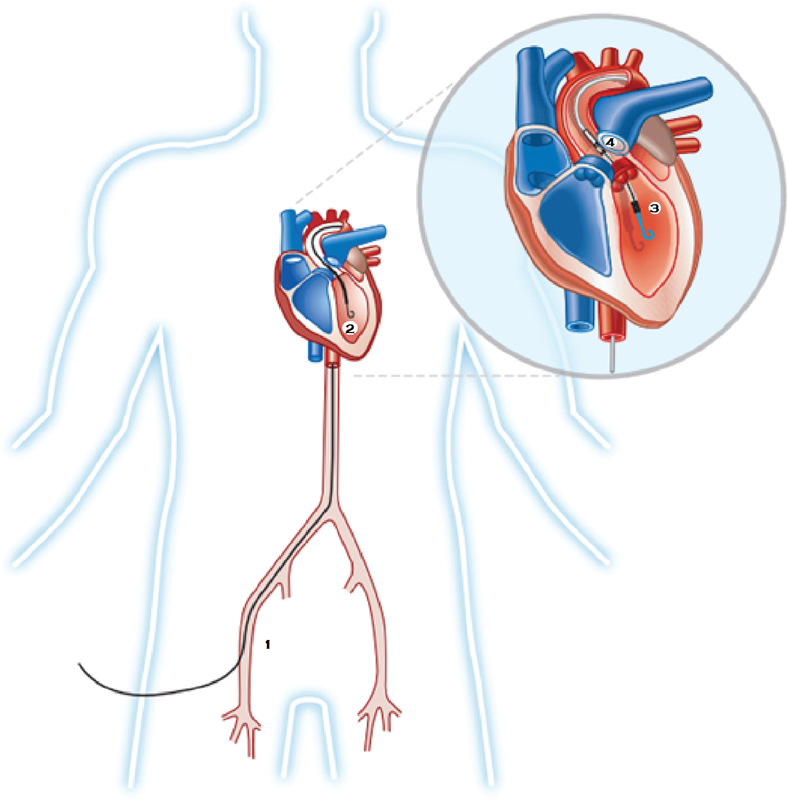
TAVR Procedural Steps
1. Access and sheath placement: Percutaneous femoral access with pre-close (2 Perclose ProGlide devices); eSheath (Edwards) or InLine sheath (Medtronic) advanced to the descending aorta. 2. Crossing the aortic valve: A straight-tipped wire crosses the stenotic aortic valve; exchanged for an extra-stiff guidewire (Safari, Lunderquist, Confida) positioned in the LV apex. 3. Balloon aortic valvuloplasty (BAV): Optional predilation with a 20-25 mm balloon under rapid ventricular pacing (180-220 bpm) to create a passage for the TAVR device. Increasingly, BAV is deferred ("direct implantation"). 4. Valve deployment: Balloon-expandable (SAPIEN) — positioned at the annular level under fluoroscopy and rapid pacing, then inflated with precise volume to deploy the valve. Self-expanding (Evolut) — unsheathed gradually; repositionable; no rapid pacing required. 5. Assessment: Aortography and TEE/TTE to assess paravalvular leak, valve position, coronary patency, and hemodynamics. Post-dilation with a larger balloon if significant PVL. 6. Closure: Tighten Perclose sutures; angiography to confirm hemostasis.
Valve-in-Valve TAVR
TAVR within a previously implanted surgical bioprosthetic valve that has degenerated (stenosis or regurgitation). Sizing is based on the true internal diameter (ID) of the surgical valve (available in manufacturer charts). Key considerations: risk of patient-prosthesis mismatch (PPM) — small surgical valves (≤21 mm) may result in high residual gradients after VIV; coronary obstruction risk is higher with VIV (especially with externally mounted leaflets that can cover the coronary ostia upon TAVR deployment); BASILICA technique (Bioprosthetic or native Aortic Scallop Intentional Laceration to prevent Iatrogenic Coronary Artery obstruction) — intentional electrosurgical laceration of a bioprosthetic leaflet before TAVR to maintain coronary flow.
TAVR Complications
| Complication | Incidence | Management |
|---|---|---|
| Paravalvular leak (PVL) | Mild 20-30%; moderate-severe 3-5% | Post-dilation; valve-in-valve; surgical conversion for severe |
| Conduction disturbance / new LBBB | LBBB 15-30%; PPM 10-20% | Higher with self-expanding; monitor 24-48 h; PPM if high-degree AVB |
| Coronary obstruction | <1% | Emergency PCI of obstructed ostium; BASILICA technique (laceration of leaflet to prevent obstruction) |
| Stroke | 2-4% | Cerebral embolic protection devices (Sentinel); anticoagulation |
| Vascular complications | 5-15% | Covered stent for iliac rupture; surgical repair |
| Annular rupture | <1% | Emergency Contained — observation; uncontained — emergent surgery; almost always fatal if free rupture |
12 Mitral & Tricuspid Interventions
Transcatheter Edge-to-Edge Repair (TEER) — MitraClip / PASCAL
TEER creates a tissue bridge between the anterior and posterior mitral leaflets, mimicking the surgical Alfieri edge-to-edge repair. The MitraClip (Abbott) is delivered via transseptal puncture from the femoral vein, guided by TEE and fluoroscopy.
COAPT trial: In patients with heart failure (LVEF 20-50%) and severe secondary (functional) mitral regurgitation on maximally tolerated GDMT, TEER reduced heart failure hospitalizations by 47% and all-cause mortality by 29% at 2 years compared to GDMT alone. Key enrollment criterion: effective regurgitant orifice area (EROA) ≥30 mm² and LVEDV ≤200 mL (disproportionate MR concept). MITRA-FR trial: Did not show benefit of TEER in secondary MR — different patient population (proportionate MR, larger ventricles, lower EROA threshold).
Primary (degenerative) MR: TEER is indicated for patients with symptomatic severe primary MR who are at prohibitive or high surgical risk. The EVEREST II trial showed MitraClip was less effective than surgery for reducing MR but had superior safety.
PASCAL device: The PASCAL system (Edwards Lifesciences) is a newer TEER platform with a central spacer that fills the regurgitant orifice and broad paddles for leaflet grasping, plus independent clasping of each leaflet. It may be advantageous for wide flail gaps and complex anatomy where MitraClip grasping is difficult. The CLASP IID/IIF trials evaluated PASCAL in both degenerative and functional MR.
Mitral Annuloplasty Devices
Cardioband (Edwards): Percutaneous direct annuloplasty — anchors deployed along the posterior mitral annulus via transseptal approach under TEE guidance; a contraction wire reduces annular dimensions, similar to surgical annuloplasty. Approved for functional MR in Europe. Carillon (Cardiac Dimensions): Indirect annuloplasty via the coronary sinus — a nitinol device placed in the coronary sinus compresses the posterior mitral annulus to reduce annular dimensions and MR. Limitation: may impinge on the LCx artery.
Transcatheter Mitral Valve Replacement (TMVR)
Multiple devices in development or early clinical use (Tendyne, Intrepid/APOLLO, Evoque) for patients not amenable to TEER. Challenges include: large device profile, LVOT obstruction risk, leaflet interaction, paravalvular leak, and need for transseptal or transapical access. Valve-in-valve TMVR for failed surgical bioprostheses (using SAPIEN 3) is established with good outcomes.
Percutaneous Balloon Mitral Valvuloplasty (PBMV)
Indicated for symptomatic moderate-severe rheumatic mitral stenosis with favorable valve morphology. The Wilkins score (0-16) assesses leaflet mobility, thickening, calcification, and subvalvular disease — a score ≤8 predicts good results. Contraindications: moderate-severe MR, left atrial thrombus, severe calcification. Performed via transseptal puncture using an Inoue balloon. Success defined as MVA >1.5 cm² and ≤1+ MR increase.
Tricuspid Interventions
Severe tricuspid regurgitation (TR) is increasingly recognized as an important contributor to morbidity and mortality. Transcatheter options are expanding:
TriClip (TEER for tricuspid): The TRILUMINATE Pivotal trial demonstrated TEER reduced TR severity at 1 year compared to medical therapy, though the primary endpoint of composite of death, tricuspid surgery, heart failure hospitalization, or improvement in quality of life narrowly missed superiority. PASCAL tricuspid TEER: Similar edge-to-edge concept with a broader clasping mechanism. Transcatheter tricuspid valve replacement (TTVR): Devices in trial include EVOQUE (heterotopic — placed in the IVC/SVC to reduce TR regurgitant volume) and orthotopic replacement systems. The TRISCEND II trial (EVOQUE) showed significant TR reduction. Cardioband: Percutaneous annuloplasty device delivering anchors along the tricuspid annulus; provides annular reduction similar to surgical ring annuloplasty.
13 Left Atrial Appendage Closure
Rationale
The left atrial appendage (LAA) is the source of >90% of thrombi in non-valvular atrial fibrillation. LAA closure (LAAC) provides an alternative to long-term oral anticoagulation (OAC) for stroke prevention in patients with AF who have contraindications to OAC or high bleeding risk.
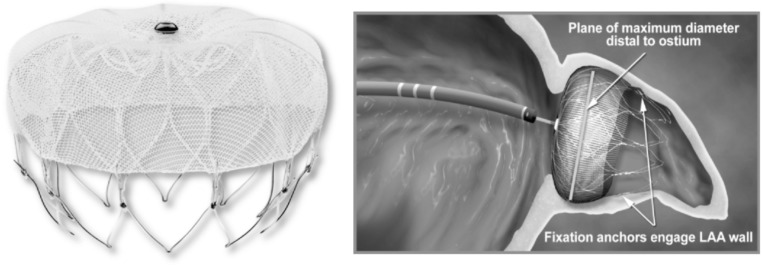
WATCHMAN Device
The WATCHMAN (Boston Scientific) is a nitinol frame with a permeable polyester fabric cap that is delivered via transseptal puncture and deployed in the LAA ostium. The PROTECT-AF trial demonstrated non-inferiority of WATCHMAN to warfarin for the composite of stroke, systemic embolism, and cardiovascular death at 5-year follow-up, with a significant reduction in hemorrhagic stroke and all-cause mortality. The PREVAIL trial confirmed non-inferiority for ischemic stroke at 5 years. The WATCHMAN FLX (next generation) has improved sealing, lower device-related thrombus, and higher procedural success (>98%).
Amulet Device
The Amulet (Abbott) is a dual-seal device with a lobe and disc that provides two levels of LAA sealing. The Amulet IDE trial demonstrated non-inferiority to WATCHMAN for safety and efficacy at 18 months, with numerically lower rates of peri-device leak.
Lariat Device (Epicardial LAA Ligation)
The Lariat system uses a combined epicardial and endocardial approach to deliver a suture loop around the LAA. A magnet-tipped wire is placed in the LAA via transseptal puncture, and a corresponding epicardial wire is advanced through a subxiphoid pericardial puncture. The Lariat snare is then delivered over the epicardial wire to lasso and ligate the LAA base. Advantages: no implanted device; complete LAA exclusion. Limitations: requires pericardial access (not feasible with prior cardiac surgery or pericardial adhesions), risk of pericarditis, pericardial effusion, and incomplete closure. The device has fallen out of favor relative to endocardial devices (WATCHMAN, Amulet).
Post-Implant Management
Standard protocol: DAPT (aspirin + clopidogrel) or short-term anticoagulation (45 days to 6 months) post-implant until device endothelialization, confirmed by TEE showing complete LAA seal and absence of device-related thrombus. Then transition to aspirin monotherapy or discontinuation of all antithrombotic therapy based on individual risk.
Patient Selection for LAAC
LAAC is most appropriate for AF patients with: CHA2DS2-VASc ≥2 (stroke risk warrants OAC), HAS-BLED ≥3 (high bleeding risk), and specific contraindications to long-term OAC (prior life-threatening bleed, intracranial hemorrhage, end-stage renal disease on dialysis, recurrent falls, or labile INR). Current guidelines give LAAC a class IIb recommendation for patients with AF unsuitable for long-term anticoagulation.
14 ASD/PFO Closure & Septal Ablation
Patent Foramen Ovale (PFO) Closure
PFO is present in ~25% of the general population. The association between PFO and cryptogenic stroke led to pivotal closure trials:
| Trial | Device | Key Finding |
|---|---|---|
| RESPECT (extended follow-up) | Amplatzer PFO Occluder | 45% relative risk reduction in recurrent stroke vs medical therapy |
| CLOSE | Various devices | 97% relative risk reduction in recurrent stroke; NNT = 20 at 5 years |
| REDUCE | GORE Helex / Cardioform | 77% relative risk reduction in recurrent ischemic stroke |
| DEFENSE-PFO | Amplatzer PFO Occluder | Significant reduction in composite of stroke, vascular death, TIMI major bleeding |
Current indications for PFO closure: age 18-60, cryptogenic stroke (thorough workup excluding other causes), PFO with moderate-large shunt or atrial septal aneurysm (ASA), and high RoPE score (Risk of Paradoxical Embolism — higher score indicates greater probability that PFO is causal). Post-closure: DAPT for 1-6 months, then aspirin alone for 5 years.
Atrial Septal Defect (ASD) Closure
Transcatheter closure is standard for secundum ASD with adequate rims (≥5 mm on all borders except the aortic rim). Devices: Amplatzer Septal Occluder (most widely used), GORE Cardioform. Indications: Qp/Qs ≥1.5, right heart enlargement, or paradoxical embolism. Primum, sinus venosus, and coronary sinus ASDs require surgical closure. Sizing is guided by TEE and/or intracardiac echocardiography (ICE). Complications: device embolization (1-2%), erosion (<0.1%), residual shunt, AF, air embolism.
Alcohol Septal Ablation (ASA)
Percutaneous treatment for hypertrophic obstructive cardiomyopathy (HOCM) with symptomatic LVOT obstruction (gradient ≥50 mmHg at rest or with provocation) refractory to medical therapy. Technique: identify the first or second septal perforator branch supplying the basal septum; confirm territory with myocardial contrast echocardiography; inject 1-3 mL of absolute (96-100%) ethanol to create a controlled septal infarction. Result: septal thinning and regression of LVOT gradient over 3-6 months. Success rate: 85-95% gradient reduction.
Complications: complete heart block requiring PPM (10-20%), LAD dissection, coronary spasm, VSD (rare), and RV perforation. ASA is an alternative to surgical septal myectomy (gold standard), particularly in patients at high surgical risk, elderly patients, or those with comorbidities. The long-term scar from ASA may serve as a substrate for ventricular arrhythmias, though large registries have not shown excess sudden death.
ASA vs Septal Myectomy — Comparison
| Feature | Alcohol Septal Ablation | Surgical Septal Myectomy |
|---|---|---|
| Approach | Percutaneous (cath lab) | Open heart surgery (sternotomy) |
| Gradient reduction | 75-85% reduction | 90-95% reduction |
| Mortality | 1-2% | 0.5-1% (experienced centers) |
| Pacemaker rate | 10-20% | 2-5% |
| Concomitant MV repair | Not possible | Can address SAM and MR simultaneously |
| Preferred for | Elderly, high surgical risk, comorbidities | Young patients, very thick septum (>30 mm), concomitant MV disease |
| Recovery | 1-3 days | 5-7 days; full recovery 6-8 weeks |
Paravalvular Leak (PVL) Closure
Paravalvular leaks occur around surgically implanted prosthetic valves, causing hemolysis (mechanical shear stress), heart failure (volume overload), or both. Transcatheter closure uses Amplatzer Vascular Plug (AVP II, AVP IV) or other occluder devices delivered via retrograde aortic (for aortic PVL) or transseptal (for mitral PVL) approach. Success rates: 75-90%. Complications include device embolization, valve impingement, and residual leak.
15 Peripheral Vascular Interventions
Carotid Artery Stenting (CAS) & TCAR
CAS is an alternative to carotid endarterectomy (CEA) for significant carotid stenosis. Indications: symptomatic carotid stenosis ≥50% or asymptomatic stenosis ≥70% with high surgical risk. Embolic protection devices (EPDs) — distal filter (FilterWire, SpiderFX) or proximal occlusion (Mo.Ma) — are mandatory during CAS to reduce stroke risk. TCAR (transcarotid artery revascularization) uses direct carotid access with flow reversal via an arteriovenous circuit, providing superior neuroprotection. The ROADSTER trials demonstrated 30-day stroke/death rates <2% with TCAR.
Renal Artery Interventions
Renal artery stenting for atherosclerotic renal artery stenosis was not shown to improve outcomes over medical therapy in the CORAL and ASTRAL trials. However, intervention may be considered for: flash pulmonary edema (Pickering syndrome), refractory hypertension on ≥3 medications, progressive renal deterioration with bilateral disease, and fibromuscular dysplasia (balloon angioplasty without stenting — good results). For FMD, the "string of beads" appearance on angiography is characteristic of the medial fibroplasia subtype.
Renal denervation (RDN): Catheter-based ablation of the renal sympathetic nerves for resistant hypertension. Radiofrequency (Symplicity Spyral) or ultrasound-based (Paradise) energy is delivered to the renal artery adventitia. The SPYRAL HTN-ON MED and RADIANCE-HTN SOLO trials showed modest but significant BP reductions (5-10 mmHg systolic) vs sham. RDN is approved in Europe and under FDA review. Patient selection is critical — best candidates are those with true resistant hypertension after confirming medication adherence.
Lower Extremity Peripheral Arterial Disease (PAD)
Endovascular revascularization is indicated for lifestyle-limiting claudication refractory to exercise therapy and pharmacotherapy, and for chronic limb-threatening ischemia (CLTI) with rest pain, ulceration, or gangrene. The Global Vascular Guidelines (GVG) use the WIfI classification (Wound, Ischemia, foot Infection) to stage CLTI severity and guide management.
| Anatomy | Preferred Approach | Techniques |
|---|---|---|
| Aorto-iliac (TASC A/B) | Endovascular | Balloon-expandable stent (kissing stents for aortic bifurcation); covered stents for long lesions |
| Femoropopliteal (SFA) | Endovascular (short/intermediate) or surgical bypass (long occlusions) | DCB (drug-coated balloon), self-expanding stent (Supera interwoven), atherectomy + DCB |
| Infrapopliteal (tibial) | Endovascular for CLTI | POBA, DCB, atherectomy; angiosome-directed revascularization |
Aortic Interventions
Thoracic Endovascular Aortic Repair (TEVAR): Endograft placement for descending thoracic aortic aneurysms (≥5.5 cm or rapid growth >0.5 cm/year) and complicated type B aortic dissections (malperfusion, rupture, refractory pain/hypertension). Landing zone classification (Ishimaru zones 0-4) determines feasibility and need for supra-aortic debranching. Zone 2 (distal to left common carotid) is the minimum proximal landing zone without left subclavian revascularization (though LSA coverage should be revascularized if possible — CSRA, left carotid-subclavian bypass).
Endovascular Aneurysm Repair (EVAR): For infrarenal abdominal aortic aneurysms (≥5.5 cm in men, ≥5.0 cm in women, or growth >0.5 cm/6 months). Anatomic requirements: infrarenal neck ≥10-15 mm, neck angle <60°, iliac diameter ≥7 mm for device delivery. Complications: endoleak (Type I — proximal/distal seal failure; Type II — branch vessel back-bleeding [most common]; Type III — graft fabric/junction failure; Type IV — graft porosity; Type V — endotension), limb occlusion, and post-implantation syndrome.
Pulmonary Embolism Interventions
Catheter-directed therapy (CDT) for massive and submassive PE includes: catheter-directed thrombolysis (CDT) — ultrasound-facilitated low-dose tPA delivery (EKOS/EkoSonic — ULTIMA, SEATTLE-II trials), aspiration thrombectomy (FlowTriever — FLARE trial; Indigo — EXTRACT-PE trial), and rheolytic thrombectomy. The FlowTriever device uses large-bore aspiration and self-expanding nitinol discs to mechanically remove thrombus without fibrinolytic agents, reducing bleeding risk. CDT for submassive PE (RV dysfunction without hemodynamic instability) remains an area of active investigation (HI-PEITHO trial).
16 Mechanical Circulatory Support
Intra-Aortic Balloon Pump (IABP)
The IABP is a counterpulsation device inserted via the femoral artery and positioned in the descending aorta just distal to the left subclavian artery. Inflation during diastole augments coronary perfusion pressure and diastolic blood pressure. Deflation during systole reduces afterload, LV wall stress, and myocardial oxygen demand. Timing: inflation at the dicrotic notch; deflation just before systole. The IABP-SHOCK II trial showed no mortality benefit of IABP in cardiogenic shock complicating MI, leading to a downgrade in guidelines. IABP remains useful as an adjunct for mechanical complications (acute MR, VSD) and as a bridge in selected patients.

Impella Devices
| Device | Flow (L/min) | Access | Mechanism | Indications |
|---|---|---|---|---|
| Impella CP | Up to 4.3 | 14 Fr femoral artery | Axial flow pump across aortic valve (LV → aorta) | High-risk PCI, cardiogenic shock (SCAI C-D) |
| Impella 5.0 | Up to 5.0 | 21 Fr axillary artery (surgical cutdown) | Same axial flow | Severe cardiogenic shock, bridge to recovery/transplant/LVAD |
| Impella 5.5 | Up to 5.5 | 21 Fr axillary artery | Same, with purge-free motor | Extended MCS support; ambulatory support possible |
| Impella RP | Up to 4.4 | 22 Fr femoral vein | Axial flow (RA → PA) | RV failure after LVAD implant, cardiac surgery, or MI |
The PROTECT III trial demonstrated that Impella-supported high-risk PCI reduced major adverse events at 90 days compared to IABP support. Complications of Impella include: hemolysis (monitor plasma-free hemoglobin), limb ischemia, aortic valve injury, device migration, and ventricular perforation.
Venoarterial ECMO (VA-ECMO)
VA-ECMO provides full cardiopulmonary support by draining deoxygenated blood from the venous system (femoral vein or RA), passing it through an oxygenator and heat exchanger, and returning oxygenated blood to the arterial system (femoral artery or aorta). VA-ECMO can provide flows of 3-7 L/min independent of cardiac function. Emergency Indications include: refractory cardiogenic shock (SCAI D-E), cardiac arrest (ECPR — extracorporeal CPR), post-cardiotomy shock, massive pulmonary embolism, and bridge to transplant/LVAD decision.
Key consideration: VA-ECMO increases LV afterload (retrograde arterial flow) and may worsen LV distension, pulmonary edema, and LV thrombus formation. LV venting strategies — Impella, IABP, atrial septostomy, or direct LA/LV vent — are often required to unload the LV during VA-ECMO support.
TandemHeart
The TandemHeart is a percutaneous left atrial-to-femoral artery bypass system. A 21 Fr transseptal inflow cannula is placed in the LA; oxygenated blood is withdrawn and pumped to the femoral artery via an extracorporeal centrifugal pump, providing up to 5 L/min of flow. Advantages: effective LV unloading (bypasses the LV entirely). Disadvantages: requires transseptal puncture, large-bore venous and arterial access, residual ASD after decannulation, and risk of limb ischemia. Largely supplanted by Impella in current practice.
Device Selection by Shock Severity
| SCAI Stage | First-Line MCS | Escalation | Notes |
|---|---|---|---|
| B (Beginning) | Vasopressors alone | IABP or Impella CP if deteriorating | Monitor lactate trend, urine output |
| C (Classic) | Impella CP | Impella 5.0/5.5 or VA-ECMO | PA catheter placement; goal CI >2.2, CPO >0.6 |
| D (Deteriorating) | VA-ECMO ± Impella (ECPELLA) | Surgical MCS (LVAD) | Bridge to decision; multidisciplinary shock team |
| E (Extremis) | VA-ECMO (ECPR) | — | Consider futility; neurologic assessment after ROSC |
SCAI Shock Classification
| Stage | Description | Hemodynamics | Clinical |
|---|---|---|---|
| A — At risk | Not in shock; at risk | Normal BP, HR | No signs of hypoperfusion; large MI, severe HF |
| B — Beginning | Hypotension or tachycardia without hypoperfusion | SBP <90 or MAP <60; HR >100 | Warm extremities, normal mentation, normal lactate |
| C — Classic | Hypoperfusion requiring intervention | CI <2.2, PCWP >15 | Cool extremities, altered mentation, rising lactate, oliguria |
| D — Deteriorating | Failing to stabilize despite initial interventions | Worsening CI despite vasopressors/MCS | Escalating lactate, multiorgan dysfunction |
| E — Extremis | Cardiac arrest or refractory hemodynamic collapse | Near-pulseless; PEA/VF/asystole | CPR, refractory VT/VF; pH <7.2; consider ECPR |
17 Advanced Hemodynamic Profiles & Waveforms
Hemodynamic Profiles in Shock
| Condition | RA | PCWP | CO/CI | SVR | Distinguishing Feature |
|---|---|---|---|---|---|
| LV cardiogenic shock | ↑ | ↑↑ | ↓↓ | ↑↑ | PCWP > 18; CI < 1.8; RA < PCWP |
| RV failure / RV infarct | ↑↑ | Normal/↓ | ↓ | ↑ | RA ≥ PCWP; RA/PCWP ratio >0.8; Kussmaul sign |
| Cardiac tamponade | ↑↑ | ↑↑ | ↓ | ↑↑ | Equalization of diastolic pressures; blunted Y descent in RA |
| Constrictive pericarditis | ↑↑ | ↑↑ | ↓/Normal | ↑ | Dip-plateau; prominent Y descent; discordance of RV/LV systolic pressures with respiration |
| Massive PE | ↑↑ | Normal/↓ | ↓↓ | ↑↑ | PA systolic ↑ (usually <60 mmHg in acute PE); RA > PCWP |
| Septic shock | ↓/Normal | ↓/Normal | ↑ (early) | ↓↓ | High CO, low SVR (warm shock); low CO in late/refractory |
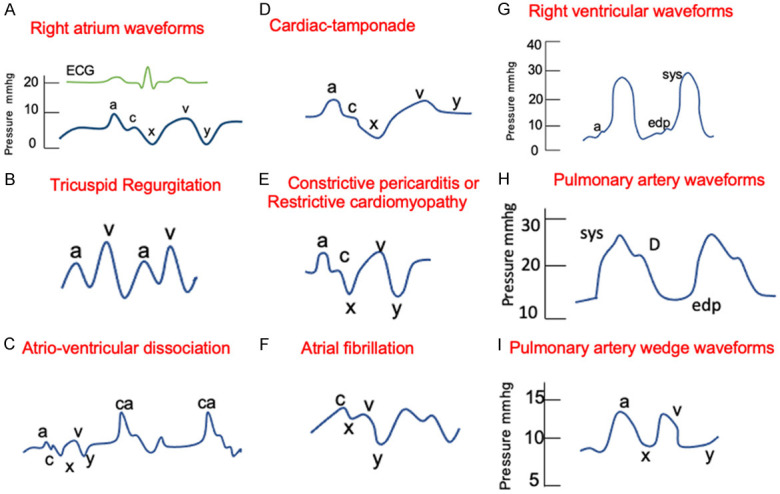
RA Waveform Analysis
The RA waveform consists of: a wave (atrial contraction), c wave (tricuspid closure/bulging), x descent (atrial relaxation/tricuspid annular descent), v wave (passive atrial filling against a closed tricuspid valve), and y descent (tricuspid opening and passive ventricular filling). Key abnormalities: Giant a waves — tricuspid stenosis, pulmonary hypertension, RV hypertrophy; Cannon a waves — AV dissociation, complete heart block, V-pacing without AV synchrony; Large v waves — severe TR; Absent a waves — atrial fibrillation; Prominent x descent — tamponade (exaggerated); Blunted y descent — tamponade; Steep y descent — constriction, severe TR.
PCWP Waveform
The PCWP tracing mirrors the LA pressure waveform with a phase delay of 50-100 ms. Key findings: Large v waves in the PCWP suggest severe mitral regurgitation (v wave amplitude ≥2x mean PCWP is highly suggestive). However, large v waves can also occur with increased LA compliance (chronic MR) or with VSD (left-to-right shunt increasing pulmonary venous return). The a wave in PCWP corresponds to LA contraction — absent in AF; giant a waves in mitral stenosis.
Provocative Maneuvers
Rapid volume loading (500-1000 mL saline): Used to unmask constrictive physiology — RVEDP and LVEDP will rise and equalize with characteristic dip-plateau pattern. Dynamic respiratory assessment: In constriction, RV and LV systolic pressures show discordance with respiration (RV pressure rises while LV pressure falls during inspiration) — this is the most sensitive and specific hemodynamic finding for constriction. In tamponade, RV and LV systolic pressures show concordance (both fall during inspiration). Exercise hemodynamics: Supine bicycle exercise in the cath lab unmasks heart failure with preserved ejection fraction (HFpEF) — a PCWP ≥25 mmHg with exercise is diagnostic.
PA Pulsatility Index (PAPi)
PAPi = (PA systolic - PA diastolic) / RA mean pressure. PAPi is a marker of RV function. A PAPi <0.9 predicts RV failure requiring RVAD support after LVAD implantation. In the setting of acute inferior MI, a PAPi <1.0 predicts hemodynamically significant RV infarction and the need for RV mechanical support. PAPi is increasingly used to assess RV reserve before MCS implantation and to guide the need for biventricular support (BiVAD or TAH) versus isolated LVAD.
Hemodynamic Assessment During Intervention
LVEDP measurement: Obtained by pigtail catheter in the LV; elevated LVEDP (>18 mmHg) correlates with HF and increased periprocedural risk. Used in the POSEIDON protocol for CIN prevention (hydration rate adjusted to LVEDP). Aortic regurgitation assessment: Aortography graded by Sellers classification (0-4+) based on density and washout of contrast from the LV. Simultaneous pressure measurement across the aortic valve (dual-lumen catheter or femoral artery + LV) provides peak-to-peak gradient and mean gradient for AS severity assessment.
18 Access Site Complications
Femoral Access Complications
Retroperitoneal hemorrhage: Emergency Occurs with high femoral arterial puncture (above the inguinal ligament) or through-and-through puncture. Presents with hemodynamic instability, back/flank pain, ipsilateral abdominal distension, and unexplained Hgb drop. Diagnosis: CT abdomen/pelvis with contrast. Management: volume resuscitation, reversal of anticoagulation, blood transfusion, and covered stent or surgical repair for active extravasation. Maintain a low threshold for diagnosis — mortality is 4-12%.
Pseudoaneurysm: Pulsatile mass at the access site with a "to-and-fro" signal on duplex ultrasound. Small (<2 cm) pseudoaneurysms may thrombose spontaneously. Treatment: ultrasound-guided thrombin injection (first-line — 95% success rate), ultrasound-guided compression, or surgical repair for large or failed cases.
Arteriovenous (AV) fistula: Communication between the femoral artery and vein, detected by continuous bruit/thrill and confirmed by duplex showing arterialized venous flow. Most small fistulae (<5 mm) close spontaneously. Large or symptomatic fistulae require covered stent placement or surgical repair.
Femoral artery dissection / occlusion: Can result from catheter manipulation, closure device deployment, or guide wire trauma. Presents with limb ischemia (acute pain, pallor, pulselessness). Management: angiography with stenting or surgical repair.
Radial Access Complications
Radial artery occlusion (RAO): Occurs in 1-10% of cases; usually asymptomatic due to dual palmar arch supply. Prevention: use smallest effective sheath, patent hemostasis (maintain antegrade flow during compression with pulse oximetry/plethysmography), and adequate anticoagulation (UFH ≥50 U/kg). Radial artery spasm: Prevented with intra-arterial verapamil/nitroglycerin cocktail and hydrophilic-coated sheaths. Managed with additional vasodilators and gentle catheter manipulation — never force catheter withdrawal against spasm (risk of avulsion).
Compartment syndrome: Rare but devastating complication from perforation of a small radial branch. Presents with forearm swelling, pain, and neurovascular compromise. Emergency Treatment: emergent fasciotomy.
Large-Bore Access Complications
TAVR, MCS, and ECMO require large-bore femoral access (14-24 Fr). Specific complications include:
Iliac rupture / dissection: Occurs during sheath insertion in calcified, tortuous, or small iliofemoral arteries. Presents with sudden hypotension and contrast extravasation on iliac angiography. Management: balloon tamponade followed by covered stent deployment (iCAST, Viabahn). Surgical repair if endovascular approach fails.
Acute limb ischemia: Large sheaths can obstruct distal flow, especially if sheath diameter exceeds the vessel lumen. Prevention: place a distal perfusion catheter (DPC) for VA-ECMO and prolonged large-bore access. Treatment: thrombectomy, fasciotomy if compartment syndrome develops.
Lymphocele / lymphatic injury: Disruption of inguinal lymphatics during access, presenting as a fluctuant non-pulsatile groin mass days to weeks after the procedure. Most resolve spontaneously; aspiration or sclerotherapy for large or symptomatic collections.
BARC Bleeding Classification
| Type | Definition |
|---|---|
| Type 0 | No bleeding |
| Type 1 | Bleeding not actionable; does not cause unscheduled evaluation or treatment |
| Type 2 | Any overt, actionable bleeding not meeting criteria for Type 3-5 |
| Type 3a | Overt bleeding + Hgb drop 3-5 g/dL (or HCT drop 9-15%); any transfusion |
| Type 3b | Overt bleeding + Hgb drop ≥5 g/dL; cardiac tamponade; requiring surgical or percutaneous intervention; vasoactive agents |
| Type 3c | Intracranial hemorrhage (confirmed by imaging) |
| Type 4 | CABG-related bleeding (perioperative intracranial bleeding, reoperation for bleeding, transfusion ≥5 units, chest tube output ≥2 L/24 h) |
| Type 5a | Probable fatal bleeding |
| Type 5b | Definite fatal bleeding |
19 Procedural Complications
Contrast-Induced Nephropathy (CIN)
CIN is defined as a rise in serum creatinine of ≥0.5 mg/dL or ≥25% from baseline within 48-72 hours of contrast exposure. Risk factors: pre-existing CKD (eGFR <60), diabetes, volume depletion, contrast volume, hemodynamic instability, and nephrotoxic agents. Prevention: adequate hydration (IV NS 1 mL/kg/h for 3-12 h pre- and 6-12 h post-procedure — POSEIDON protocol targets LVEDP-guided hydration), minimize contrast volume (target contrast/eGFR ratio <3.7), use iso-osmolar or low-osmolar contrast, avoid nephrotoxic agents (NSAIDs, aminoglycosides), and hold metformin for 48 hours post-contrast. N-acetylcysteine has not shown consistent benefit (PRESERVE trial — no benefit of NAC or IV sodium bicarbonate over IV saline).
Coronary Perforation
See Section 10 for Ellis classification and management. Additional considerations: Wire perforation (most common cause) — typically Type I-II; managed by balloon inflation proximal to the perforation, reversal of anticoagulation with protamine, and observation. Balloon/stent perforation — oversized balloon or stent in a diseased vessel; managed with covered stent (PK Papyrus). Atherectomy perforation — rotational or orbital atherectomy in a tortuous or thin-walled segment; high risk of Type III perforation.
Stroke During Cardiac Catheterization
Periprocedural stroke occurs in 0.1-0.4% of diagnostic catheterizations and 0.2-0.6% of PCI. Risk factors: aortic arch atheroma, prolonged procedure, catheter manipulation in the aortic root, atrial fibrillation, and left heart catheterization via femoral approach. Management: immediate neurology consultation, CT head to exclude hemorrhagic stroke, catheter-directed thrombectomy or IV tPA if within the appropriate time window, and supportive care.
Air Embolism
Coronary air embolism presents with acute ST elevation, hemodynamic collapse, and chest pain. Emergency Management: vigorous aspiration through the guide catheter, forceful injection of saline and blood (to fragment air bubbles), 100% oxygen (accelerates nitrogen reabsorption), and intracoronary vasodilators for associated spasm. Prevention: meticulous catheter aspiration and flushing, avoiding back-bleeding of guide catheters, and careful attention to manifold connections. Large-volume air embolism (manifold disconnection or sheath flushing error) can cause immediate cardiovascular collapse requiring CPR and potentially emergent percutaneous aspiration from the coronary arteries or aortic root.
Coronary Spasm (Iatrogenic)
Catheter-induced spasm occurs at the site of guide catheter engagement or wire manipulation. More common with radial access (spasm of the radial artery itself is distinct from coronary spasm). Treatment: intracoronary nitroglycerin (100-200 μg bolus, may repeat), intracoronary verapamil (100-200 μg if NTG ineffective), or intracoronary nicardipine (100-200 μg). Refractory spasm may mimic acute occlusion — always consider spasm before escalating to emergent intervention. Patients with known variant (Prinzmetal) angina may have heightened vasoreactivity; pretreat with IC NTG before diagnostic injection.
Radiation Skin Injury
Prolonged fluoroscopy during complex PCI (CTO, chronic total occlusions) can cause radiation dermatitis. Effects are dose-dependent: 2-5 Gy — transient erythema; 5-10 Gy — prolonged erythema, dry desquamation; 10-15 Gy — moist desquamation, pain; >15 Gy — dermal necrosis, ulceration. The patient's back is the most commonly affected site (entrance skin dose). Monitoring: real-time air kerma display; alert at 5 Gy (sentinel event). Management: dermatology referral for skin injuries >5 Gy; wound care for ulceration. Prevention: limit fluoroscopy time, use low-dose protocols, change angulation to distribute dose across skin surface.
Catheter-Induced Coronary Dissection
Occurs during guide catheter engagement or aggressive manipulation, particularly with Amplatz-type catheters in the RCA. The NHLBI dissection classification grades severity:
| Type | Description | Management |
|---|---|---|
| A | Minor radiolucency within the lumen; minimal or no persistence | Conservative; observation |
| B | Parallel tracts or double lumen; may persist after contrast clearance | Conservative if flow maintained; stent if flow-limiting |
| C | Extraluminal cap with contrast persistence (dissection pocket) | Stent to seal flap |
| D | Spiral dissection; contrast staining of vessel wall | Stent; may require multiple stents |
| E | New persistent filling defect (intimal flap) | Stent if flow-limiting |
| F | Dissection leading to total occlusion (TIMI 0) | Emergency Immediate stenting; wire past dissection flap |
Left main dissection is a catastrophic complication requiring immediate stenting to restore flow. If left main dissection extends to the aortic root, emergent surgical consultation is required.
Cholesterol Embolization Syndrome
Shower of cholesterol crystals from disrupted aortic atherosclerotic plaques during catheter manipulation. Presents days to weeks after the procedure with: blue/purple toes (livedo reticularis) with palpable pedal pulses, renal failure (progressive, may require dialysis), eosinophilia, elevated ESR, low complement, and Hollenhorst plaques on fundoscopy. Diagnosis is clinical; skin or renal biopsy shows biconvex "cleft" spaces (cholesterol crystals dissolved during processing). There is no specific treatment — supportive care, statin therapy, and avoidance of further catheterization if possible. Mortality rate is 70-90% when systemic involvement is severe.
20 Antithrombotic Protocols & Bleeding Management
DAPT Protocols by Indication
| Indication | Standard DAPT Duration | Preferred P2Y12 | Notes |
|---|---|---|---|
| ACS + DES | 12 months | Ticagrelor or prasugrel | Consider extended (>12 mo) if high ischemic risk, low bleed risk |
| Stable CAD + DES | 6 months (may shorten to 3 mo) | Clopidogrel | TWILIGHT/STOPDAPT support shortened DAPT |
| Left main PCI | 12 months | Ticagrelor or clopidogrel | Imaging-guided optimization recommended |
| CTO PCI | 12 months | Ticagrelor or clopidogrel | Long stented segments; consider extended |
| TAVR | 3-6 months DAPT or anticoagulation | Clopidogrel | Single antiplatelet (aspirin) may suffice per POPular TAVI |
| LAAC | 45 days DAPT → ASA monotherapy | Clopidogrel | Some centers use short-term anticoagulation |
Triple Therapy — AF + PCI
Patients with AF requiring OAC who undergo PCI face competing risks of thromboembolism (stent thrombosis, stroke) and bleeding (triple therapy). Current evidence favors minimizing triple therapy duration:
WOEST — Dual therapy (OAC + clopidogrel, no aspirin) reduced bleeding without increasing ischemic events. RE-DUAL PCI — Dabigatran + P2Y12 inhibitor vs triple therapy: non-inferior for ischemic events, lower bleeding. AUGUSTUS — Apixaban + P2Y12 inhibitor (without aspirin) was the safest strategy; aspirin should be dropped by 1-4 weeks. ENTRUST-AF PCI — Edoxaban + P2Y12 inhibitor was non-inferior to triple therapy.
Current approach: Triple therapy (OAC + DAPT) for 1 week to 1 month maximum → dual therapy (DOAC + P2Y12 inhibitor, drop aspirin) for up to 12 months → OAC monotherapy lifelong. Use a DOAC (not warfarin) and clopidogrel (not prasugrel or ticagrelor) to minimize bleeding.
Special Considerations — Mechanical Heart Valves
Patients with mechanical prosthetic valves requiring PCI present a unique challenge — warfarin (not DOACs) is mandatory for valve thrombosis prevention. The INR target is 2.5-3.5 for mechanical mitral valves and 2.0-3.0 for mechanical aortic valves. After PCI: triple therapy (warfarin + ASA + clopidogrel) for 1 month → warfarin + single antiplatelet for 12 months → warfarin alone. Radial access and BMS (to shorten DAPT) or second-gen DES with shortened DAPT should be considered. DOACs are contraindicated with mechanical valves (RE-ALIGN trial — dabigatran caused thromboembolic and bleeding complications).
CKD & Dialysis Patients
Patients with CKD (eGFR <30) and dialysis patients have higher rates of bleeding, stent thrombosis, and mortality after PCI. Key considerations: minimize contrast volume (consider IVUS-guided "zero-contrast" PCI for severe CKD); iso-osmolar contrast (iodixanol) may reduce CIN risk; BMS vs DES decision is individualized (DES preferred if life expectancy >1 year); prasugrel and ticagrelor lack strong safety data in dialysis — clopidogrel is generally preferred; bivalirudin may be preferred over UFH (predictable pharmacokinetics); fondaparinux is contraindicated if CrCl <30. Dialysis timing: schedule dialysis 24-48 hours post-procedure if contrast was used.
Perioperative Bridging of DAPT
Elective non-cardiac surgery should ideally be deferred for 6 months after DES implantation (minimum 3 months with newer-gen DES) and 1 month after BMS. If surgery cannot be delayed, aspirin should be continued perioperatively in most cases. P2Y12 inhibitors should be held for 5 days (clopidogrel, ticagrelor) or 7 days (prasugrel) before surgery. Bridging with cangrelor or GP IIb/IIIa inhibitors is reserved for very high-risk patients (recent stent <30 days) who require urgent surgery. There is no evidence supporting routine bridging with IV heparin for DAPT cessation.
Timing of Surgery After Stent Implantation
| Stent Type | Elective Surgery Timing | Semi-Urgent Surgery | Minimum DAPT Before Surgery |
|---|---|---|---|
| Balloon angioplasty | ≥14 days | Proceed with aspirin | 14 days |
| BMS | ≥30 days | Proceed after 14 days with aspirin | 1 month |
| DES (older generation) | ≥6 months | Proceed after 3 months with aspirin | 6 months |
| DES (newer generation) | ≥3-6 months | Proceed after 1-3 months with aspirin | 3 months |
| Complex PCI (LM, CTO, bifurcation) | ≥12 months | Proceed after 6 months with aspirin | 12 months |
Bleeding Management & Reversal Agents
| Agent | Reversal | Notes |
|---|---|---|
| Unfractionated heparin | Protamine sulfate (1 mg per 100 U UFH) | Maximum dose 50 mg; risk of anaphylaxis (fish allergy, prior protamine exposure) |
| Enoxaparin | Protamine (1 mg per 1 mg enoxaparin, within 8 h) | ~60% reversal of anti-Xa activity |
| Bivalirudin | No specific reversal; short half-life (25 min) | Stop infusion; supportive care; consider hemodialysis if renal failure |
| Aspirin / P2Y12 | Platelet transfusion | Transfuse only for life-threatening bleeding; ticagrelor inhibits transfused platelets (reversible) |
| Dabigatran | Idarucizumab (Praxbind, 5 g IV) | Complete reversal within minutes |
| Rivaroxaban / Apixaban / Edoxaban | Andexanet alfa (Andexxa) | Factor Xa decoy; high dose for rivaroxaban, low dose for apixaban |
| Warfarin | 4-factor PCC (KCentra), vitamin K | PCC for immediate reversal; vitamin K (10 mg IV) for sustained effect |
21 Classification Systems
SYNTAX Score
Angiographic scoring system for coronary disease complexity. Low (≤22): PCI equivalent to CABG; Intermediate (23-32): Heart Team discussion; High (≥33): CABG preferred. Variables: number of lesions, segments involved, total occlusions, trifurcation/bifurcation, aorto-ostial lesions, severe tortuosity, calcification, thrombus, vessel length >20 mm, small vessels, and diffuse disease.
TIMI Risk Score — STEMI
Predicts 30-day mortality in STEMI (0-14 points): Age (65-74 = 2, ≥75 = 3), diabetes/HTN/angina (1), SBP <100 (3), HR >100 (2), Killip II-IV (2), weight <67 kg (1), anterior STEMI or LBBB (1), time to treatment >4 h (1).
TIMI Risk Score — UA/NSTEMI
Predicts 14-day risk of death, MI, or urgent revascularization (0-7 points, 1 each): Age ≥65, ≥3 CAD risk factors, known CAD (≥50% stenosis), ASA use in past 7 days, ≥2 anginal events in 24 h, ST deviation ≥0.5 mm, elevated cardiac biomarkers.
GRACE Score
Most validated risk score for in-hospital and 6-month mortality in ACS. Variables: age, heart rate, SBP, creatinine, Killip class, cardiac arrest, ST deviation, elevated biomarkers. Low risk (<109): <1% mortality; Intermediate (109-140): 1-3%; High (>140): >3% — early invasive strategy recommended.
CRUSADE Bleeding Score
Predicts in-hospital major bleeding risk in NSTEMI patients. Variables (0-100 points): baseline hematocrit, creatinine clearance, heart rate, SBP, sex, diabetes, prior vascular disease, and signs of CHF. Very low (≤20): 3% bleed risk; Low (21-30): 6%; Moderate (31-40): 9%; High (41-50): 12%; Very high (>50): 19%. Helps guide selection of antithrombotic intensity and vascular access strategy.
HEART Score
Used for chest pain risk stratification in the emergency department (0-10 points): History (0-2), ECG (0-2), Age (0-2), Risk factors (0-2), Troponin (0-2). Score 0-3: low risk (safe for discharge); 4-6: moderate risk (observation, further testing); 7-10: high risk (early invasive strategy). Validated in the HEART Pathway trial for safe early discharge of low-risk chest pain patients.
CCS Angina Classification
| Class | Description |
|---|---|
| I | Angina with strenuous or prolonged exertion; ordinary activity does not cause angina |
| II | Slight limitation of ordinary activity — angina with walking >2 blocks on level ground or climbing >1 flight of stairs |
| III | Marked limitation — angina with walking 1-2 blocks or climbing 1 flight of stairs |
| IV | Inability to perform any physical activity without angina; may be present at rest |
Killip Classification — Acute MI
| Class | Clinical Findings | Mortality |
|---|---|---|
| I | No heart failure signs | ~6% |
| II | Rales, S3, JVD | ~17% |
| III | Pulmonary edema | ~38% |
| IV | Cardiogenic shock | ~80% (without revascularization) |
NYHA Functional Classification
| Class | Description |
|---|---|
| I | No limitation of physical activity; ordinary activity does not cause symptoms |
| II | Slight limitation; comfortable at rest; ordinary activity causes fatigue, dyspnea, palpitations, or angina |
| III | Marked limitation; comfortable at rest; less than ordinary activity causes symptoms |
| IV | Unable to carry out any physical activity without discomfort; symptoms at rest |
ARC Stent Thrombosis Classification
Definite: Angiographic or pathological confirmation of thrombus within the stent. Probable: Unexplained death within 30 days of stent implantation, or any MI in the stented territory without angiographic confirmation. Possible: Unexplained death >30 days after stent implantation. Timing: Acute (0-24 h), subacute (1-30 d), late (30 d-1 y), very late (>1 y).
Ellis Coronary Perforation Classification
See Section 10 for full details. Type I: extraluminal crater; Type II: myocardial/pericardial blush; Type III: frank perforation with streaming; Cavity spilling: into cardiac chamber.
Mehran In-Stent Restenosis Classification
Type I: focal (≤10 mm); Type II: diffuse intra-stent (>10 mm); Type III: proliferative (extending beyond stent margins); Type IV: total occlusion.
Medina Bifurcation Classification
Three-digit code describing involvement (1 = ≥50% stenosis, 0 = <50%) of the proximal main branch, distal main branch, and side branch: 1,1,1 = all three segments; 1,1,0 = main vessel only; 0,0,1 = side branch only; 1,0,1 = proximal main and side branch (Medina classification is essential for bifurcation PCI planning).
Rentrop Collateral Classification
Grade 0: No filling of collateral channels. Grade 1: Filling of side branches of the occluded vessel via collateral channels, without visualization of the epicardial segment. Grade 2: Partial filling of the epicardial artery via collateral channels. Grade 3: Complete filling of the epicardial artery by collateral channels.
Wilkins Score (Mitral Balloon Valvuloplasty)
Each parameter scored 1-4 (total 4-16): Leaflet mobility (1 = mobile, 4 = immobile); Leaflet thickening (1 = near-normal, 4 = severe); Subvalvular thickening (1 = minimal, 4 = extensive); Calcification (1 = none, 4 = extensive). Score ≤8: favorable for PBMV; >8: consider surgical MVR.
RoPE Score (PFO-Associated Stroke)
Risk of Paradoxical Embolism score (0-10 points): age (0-5 points based on age 18-≥70), no hypertension (1), no diabetes (1), no stroke/TIA (1), nonsmoker (1), cortical infarct on imaging (1). Higher score = greater probability that PFO is causally related to the stroke (and greater benefit from closure). RoPE ≥7: ~84% probability PFO is pathogenic.
J-CTO Score (CTO Difficulty)
Predicts guidewire crossing within 30 minutes (0-5 points, 1 each): Blunt proximal cap, Calcification, Bending >45°, Occlusion length >20 mm, Prior failed attempt. Score 0: easy (success ~90%); 1: intermediate (~75%); 2: difficult (~55%); ≥3: very difficult (<40% wire crossing in 30 min).
PROGRESS-CTO Score
Predicts technical success of CTO PCI using the hybrid algorithm (0-6 points): proximal cap ambiguity (1), absence of interventional collateral (1), moderate-severe tortuosity (1), circumferential calcification (1), CTO length ≥20 mm (1), prior failed attempt (1). Score 0-1: >90% success; 2: ~85%; 3: ~75%; ≥4: <65% success. Unlike the J-CTO score, PROGRESS-CTO accounts for collateral quality, which is essential for retrograde strategy selection.
Sellers Classification (Aortic Regurgitation on Aortography)
| Grade | Description |
|---|---|
| 1+ | Contrast enters the LV but clears with each systole; no opacification of the entire LV |
| 2+ | Faint, incomplete opacification of the entire LV cavity |
| 3+ | Complete opacification of the LV; equal density to the aorta |
| 4+ | Complete opacification of the LV on the first beat; LV density greater than aorta; LV dilated |
WIfI Classification (Chronic Limb-Threatening Ischemia)
The Wound, Ischemia, and foot Infection (WIfI) classification stages CLTI severity on a 0-3 scale for each domain: Wound (0 = no ulcer, 3 = extensive gangrene); Ischemia (0 = ABI ≥0.80, 3 = ABI ≤0.39 or absolute toe pressure <30 mmHg); foot Infection (0 = none, 3 = SIRS/sepsis). Combined staging predicts amputation risk and benefit from revascularization. WIfI stages 3-4 (high ischemia, high wound burden) benefit most from revascularization.
DAPT Score
Predicts benefit vs risk of prolonged DAPT after PCI (range -2 to 10): Positive score (≥2) — benefit from extended DAPT (ischemic risk reduction outweighs bleeding risk). Negative score (<2) — bleeding risk outweighs ischemic benefit; favor shorter DAPT. Variables: age (≥75 = -2, 65-74 = -1), smoking (+1), diabetes (+1), MI at presentation (+1), prior PCI or MI (+1), paclitaxel stent (+1), stent diameter <3 mm (+1), CHF/LVEF <30% (+2), vein graft PCI (+2).
22 Medications Master Table
Antiplatelet Agents
| Drug | Mechanism | Dose | Key Considerations |
|---|---|---|---|
| Aspirin | Irreversible COX-1 inhibitor | Load 325 mg; maintenance 81 mg daily | Lifelong after ACS/PCI; GI protection with PPI |
| Clopidogrel | Irreversible P2Y12 (prodrug, CYP2C19) | Load 600 mg; maintenance 75 mg daily | 3-15% poor metabolizers; consider PFT or CYP2C19 genotyping |
| Prasugrel | Irreversible P2Y12 (prodrug) | Load 60 mg; maintenance 10 mg daily | Contraindicated: prior stroke/TIA; caution: age ≥75, wt <60 kg (5 mg dose) |
| Ticagrelor | Reversible P2Y12 (direct-acting) | Load 180 mg; maintenance 90 mg BID | Dyspnea (14%), bradycardia; avoid with strong CYP3A4 inhibitors; 60 mg BID for extended DAPT |
| Cangrelor | IV reversible P2Y12 | 30 μg/kg bolus + 4 μg/kg/min | Onset 2 min; offset 60 min; bridging for surgery |
Anticoagulants
| Drug | Mechanism | Dose (PCI) | Monitoring / Reversal |
|---|---|---|---|
| Unfractionated heparin | Antithrombin III potentiator (anti-IIa, anti-Xa) | 70-100 U/kg bolus; target ACT 250-350 s | ACT; reverse with protamine |
| Bivalirudin | Direct thrombin inhibitor | 0.75 mg/kg bolus + 1.75 mg/kg/h infusion | ACT; no specific reversal (t½ 25 min) |
| Enoxaparin | LMWH (primarily anti-Xa) | 0.5 mg/kg IV bolus during PCI | Anti-Xa level; partial reversal with protamine |
| Fondaparinux | Selective factor Xa inhibitor | 2.5 mg SC daily (pre-PCI); add UFH for PCI | Anti-Xa level; no effective reversal |
GP IIb/IIIa Inhibitors
| Drug | Dose | Duration | Renal Adjustment |
|---|---|---|---|
| Abciximab | 0.25 mg/kg bolus + 0.125 μg/kg/min | 12 h post-PCI | None; cleared by RES (not renal) |
| Eptifibatide | 180 μg/kg ×2 bolus (10 min apart) + 2 μg/kg/min | 18-24 h | 1 μg/kg/min if CrCl <50; contraindicated if dialysis |
| Tirofiban | 25 μg/kg bolus + 0.15 μg/kg/min | 18-24 h | 50% dose reduction if CrCl <30 |
Vasodilators & Vasopressors
| Drug | Use in Cath Lab | Dose |
|---|---|---|
| Nitroglycerin | IC for spasm assessment; IC for no-reflow | 100-200 μg IC bolus |
| Adenosine | FFR hyperemia; IC for no-reflow | IV: 140 μg/kg/min; IC: 100-200 μg (RCA), 200-300 μg (LCA) |
| Nitroprusside | IC for no-reflow / spasm | 100-200 μg IC |
| Verapamil | IC for no-reflow / radial spasm | 100-200 μg IC (caution if LV dysfunction) |
| Norepinephrine | Vasopressor of choice in cardiogenic shock | 0.05-0.4 μg/kg/min IV |
| Phenylephrine | Pure alpha — vasodilation with adequate CO | 100-200 μg IV bolus; 0.5-5 μg/kg/min |
| Dopamine | Inotrope/vasopressor (dose-dependent) | 2-20 μg/kg/min IV |
| Dobutamine | Inotrope for low CO states | 2-20 μg/kg/min IV |
| Milrinone | Inodilator (PDE3 inhibitor) | 0.375-0.75 μg/kg/min IV (no bolus in shock) |
| Atropine | Symptomatic bradycardia; RCA injection vagal response | 0.5-1 mg IV; may repeat |
23 Imaging & Diagnostics Reference
Coronary Angiography Views — Quick Reference
| Vessel / Segment | Optimal Views |
|---|---|
| Left main | AP caudal, LAO cranial (spider), RAO caudal |
| Proximal/mid LAD | RAO cranial, AP cranial, LAO cranial |
| Distal LAD | RAO cranial, lateral |
| Diagonal branches | RAO cranial, AP cranial, LAO cranial |
| LCx / OM | RAO caudal, LAO caudal, AP caudal |
| Proximal/mid RCA | LAO straight, RAO straight |
| Distal RCA / PDA | LAO cranial, RAO cranial |
IVUS vs OCT Comparison
| Feature | IVUS | OCT |
|---|---|---|
| Energy source | Ultrasound (20-60 MHz) | Near-infrared light (1,300 nm) |
| Axial resolution | ~100 μm | ~10-15 μm |
| Penetration depth | 4-8 mm (full vessel wall) | 1-2 mm |
| Blood clearance | Not required | Required (contrast flush) |
| Calcium assessment | Identifies but cannot measure thickness | Measures calcium thickness and arc |
| Stent apposition | Good | Superior (resolution advantage) |
| Thrombus detection | Limited | Excellent (red vs white thrombus) |
| Vulnerable plaque | Limited (VH-IVUS) | Superior — identifies TCFA (cap <65 μm) |
| Aorto-ostial lesions | Preferred (no flush needed) | Difficult (flush inadequacy) |
FFR / iFR Decision Matrix
| Value | FFR | iFR / RFR | Decision |
|---|---|---|---|
| Not significant | >0.80 | >0.89 | Defer PCI; optimal medical therapy |
| Significant | ≤0.80 | ≤0.89 | Revascularization recommended |
| Grey zone (FFR) | 0.75-0.80 | — | Consider clinical context; IVUS/OCT may help |
Coronary CTA (CCTA)
Coronary CT angiography is a non-invasive alternative to invasive angiography for evaluating coronary anatomy and stenosis. Key metrics: CT-FFR (FFRCT) — computational fluid dynamics applied to CCTA images to estimate FFR non-invasively (PLATFORM, ADVANCE trials); Stenosis severity — <50% (non-obstructive), 50-69% (moderate), ≥70% (severe), ≥50% left main; High-risk plaque features — positive remodeling, low-attenuation plaque (<30 HU), napkin-ring sign, spotty calcification. CCTA is endorsed as a first-line test for chest pain evaluation in low-intermediate risk patients (NICE guidelines, PROMISE, SCOT-HEART trials).
CT Coronary Calcium Scoring (Agatston Score)
| Score | Interpretation | Risk |
|---|---|---|
| 0 | No identifiable calcium | Very low risk; CAD unlikely |
| 1-99 | Mild calcification | Low risk |
| 100-399 | Moderate calcification | Moderate risk; indicates established CAD |
| ≥400 | Severe calcification | High risk; advanced CAD |
Echocardiography for Structural Procedures
TEE for TAVR: Confirms annular sizing, guides valve positioning, assesses paravalvular leak, detects pericardial effusion, and evaluates LV function. TEE for TEER: Guides transseptal puncture, visualizes leaflet grasping, assesses residual MR, and monitors for complications (pericardial effusion, single-leaflet device attachment). ICE (intracardiac echo): Increasingly used for ASD/PFO closure, LAA closure, and transseptal puncture guidance — eliminates need for general anesthesia required with TEE.
Transseptal Puncture — Technical Considerations
Transseptal access is the gateway to all left-sided structural interventions (TEER, LAAC, TMVR, PBMV, PFO/ASD closure). The ideal puncture site varies by procedure: TEER — posterior and superior (4-4.5 cm above the mitral annulus); LAAC — posterior and inferior (directed toward the LAA ostium); PBMV — posterior and inferior. Equipment: Brockenbrough needle (BRK or BRK-1), Mullins sheath or deflectable sheath (Agilis). Puncture site is confirmed by tenting of the fossa ovalis on TEE or ICE. Complications: cardiac perforation/tamponade (0.5-1%), aortic root puncture, air embolism, and residual ASD (usually closes spontaneously; rarely hemodynamically significant).
24 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ACT | Activated clotting time |
| ADR | Antegrade dissection re-entry |
| ARC | Academic Research Consortium |
| ASD | Atrial septal defect |
| ASA | Alcohol septal ablation (or atrial septal aneurysm, per context) |
| AVA | Aortic valve area |
| AWE | Antegrade wire escalation |
| BARC | Bleeding Academic Research Consortium |
| BMS | Bare metal stent |
| BVS | Bioresorbable vascular scaffold |
| CABG | Coronary artery bypass grafting |
| CAS | Carotid artery stenting |
| CI | Cardiac index |
| CIN | Contrast-induced nephropathy |
| CLTI | Chronic limb-threatening ischemia |
| CO | Cardiac output |
| CTO | Chronic total occlusion |
| DAPT | Dual antiplatelet therapy |
| DCB | Drug-coated balloon |
| DEB | Drug-eluting balloon |
| DES | Drug-eluting stent |
| DOAC | Direct oral anticoagulant |
| EBU | Extra backup (guide catheter) |
| ECMO | Extracorporeal membrane oxygenation |
| ECPR | Extracorporeal cardiopulmonary resuscitation |
| EROA | Effective regurgitant orifice area |
| FFR | Fractional flow reserve |
| GDMT | Guideline-directed medical therapy |
| HOCM | Hypertrophic obstructive cardiomyopathy |
| IABP | Intra-aortic balloon pump |
| ICE | Intracardiac echocardiography |
| iFR | Instantaneous wave-free ratio |
| ISR | In-stent restenosis |
| IVL | Intravascular lithotripsy |
| IVUS | Intravascular ultrasound |
| LAA | Left atrial appendage |
| LAAC | Left atrial appendage closure |
| LAD | Left anterior descending artery |
| LCx | Left circumflex artery |
| LMCA | Left main coronary artery |
| LVEF | Left ventricular ejection fraction |
| LVOT | Left ventricular outflow tract |
| MACE | Major adverse cardiovascular events |
| MCS | Mechanical circulatory support |
| MLD | Minimum lumen diameter |
| MR | Mitral regurgitation |
| MSA | Minimum stent area |
| MVA | Mitral valve area |
| NC | Non-compliant (balloon) |
| NHPR | Non-hyperemic pressure ratio |
| NSTEMI | Non-ST-elevation myocardial infarction |
| OA | Orbital atherectomy |
| OCT | Optical coherence tomography |
| OMT | Optimal medical therapy |
| PA | Pulmonary artery |
| PAC | Pulmonary artery catheter |
| PAD | Peripheral arterial disease |
| PBMV | Percutaneous balloon mitral valvuloplasty |
| PCI | Percutaneous coronary intervention |
| PCWP | Pulmonary capillary wedge pressure |
| PDA | Posterior descending artery |
| PFO | Patent foramen ovale |
| POBA | Plain old balloon angioplasty |
| POT | Proximal optimization technique |
| PPM | Permanent pacemaker |
| PVL | Paravalvular leak |
| PVR | Pulmonary vascular resistance |
| QCA | Quantitative coronary angiography |
| RA | Rotational atherectomy (or right atrium, per context) |
| RAO | Right anterior oblique |
| RCA | Right coronary artery |
| RFR | Resting full-cycle ratio |
| RVD | Reference vessel diameter |
| SAVR | Surgical aortic valve replacement |
| SCAI | Society for Cardiovascular Angiography & Interventions |
| STEMI | ST-elevation myocardial infarction |
| SVG | Saphenous vein graft |
| SVR | Systemic vascular resistance |
| TAVR | Transcatheter aortic valve replacement |
| TCAR | Transcarotid artery revascularization |
| TCFA | Thin-cap fibroatheroma |
| TEER | Transcatheter edge-to-edge repair |
| TEE | Transesophageal echocardiography |
| TF | Transfemoral |
| TIMI | Thrombolysis in Myocardial Infarction |
| TMPG | TIMI myocardial perfusion grade |
| TMVR | Transcatheter mitral valve replacement |
| TR | Tricuspid regurgitation |
| TRA | Transradial access |
| TTVR | Transcatheter tricuspid valve replacement |
| UFH | Unfractionated heparin |
| VA-ECMO | Venoarterial extracorporeal membrane oxygenation |
| VH-IVUS | Virtual histology intravascular ultrasound |
| VLST | Very late stent thrombosis |
| VSD | Ventricular septal defect |
25 Landmark Trials
Coronary Revascularization Strategy
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| COURAGE | 2007 | PCI + OMT did not reduce death or MI vs OMT alone in stable CAD | PMID 17387127 |
| ISCHEMIA | 2020 | Invasive strategy did not reduce death or MI vs conservative strategy in stable CAD with moderate-severe ischemia | PMID 32227755 |
| SYNTAX | 2009 | CABG superior to PCI in 3-vessel/left main disease; SYNTAX score predicts PCI outcomes | PMID 19228612 |
| EXCEL | 2019 | PCI non-inferior to CABG for left main (low-intermediate SYNTAX) at 5 years; higher repeat revascularization with PCI | PMID 31562134 |
| NOBLE | 2020 | CABG superior to PCI for left main at 5 years (non-procedural MI, repeat revascularization) | PMID 31813566 |
Coronary Physiology & Imaging
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| FAME | 2009 | FFR-guided PCI reduced MACE vs angiography-guided PCI in multivessel disease | PMID 19144937 |
| FAME 2 | 2012 | FFR-guided PCI + OMT reduced urgent revascularization vs OMT alone when FFR ≤0.80 | PMID 22998338 |
| DEFINE-FLAIR | 2017 | iFR-guided revascularization non-inferior to FFR-guided at 1 year | PMID 28317438 |
| iFR-SWEDEHEART | 2017 | iFR-guided revascularization non-inferior to FFR-guided at 1 year | PMID 28317439 |
| ULTIMATE | 2021 | IVUS-guided PCI reduced MACE vs angiography-guided PCI at 3 years | PMID 33454528 |
STEMI & ACS
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| SHOCK | 1999 | Early revascularization reduced 6-month mortality in cardiogenic shock complicating MI | PMID 10471456 |
| COMPLETE | 2019 | Staged complete revascularization after primary PCI reduced CV death and MI vs culprit-only | PMID 31475799 |
| MATRIX | 2015 | Radial access reduced net adverse clinical events (including bleeding and mortality) vs femoral in ACS | PMID 26466021 |
| TOTAL | 2015 | Routine aspiration thrombectomy did not reduce CV death, MI, cardiogenic shock, or NYHA IV HF in STEMI | PMID 25638659 |
| TIMACS | 2009 | Early invasive strategy (<24 h) reduced composite endpoint vs delayed (>36 h) in high-risk NSTEMI | PMID 19458363 |
Antiplatelet Therapy
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| PLATO | 2009 | Ticagrelor reduced CV death, MI, stroke vs clopidogrel in ACS | PMID 19717846 |
| TRITON-TIMI 38 | 2007 | Prasugrel reduced ischemic events vs clopidogrel in ACS-PCI; more bleeding | PMID 17982182 |
| TWILIGHT | 2019 | Ticagrelor monotherapy after 3 months DAPT reduced BARC 2-5 bleeding without increasing ischemic events | PMID 31556372 |
| AUGUSTUS | 2019 | Apixaban + P2Y12 (without ASA) safest antithrombotic strategy in AF + PCI patients | PMID 30883055 |
Structural Heart
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| PARTNER 1A/1B | 2010-11 | TAVR superior to medical Rx (inoperable) and non-inferior to SAVR (high risk) | PMID 20961243 |
| PARTNER 3 | 2019 | TAVR superior to SAVR in low-risk patients at 1 year (composite of death, stroke, rehospitalization) | PMID 30883058 |
| COAPT | 2018 | TEER (MitraClip) reduced HF hospitalization and death in HFrEF with severe secondary MR on GDMT | PMID 30280640 |
| MITRA-FR | 2018 | TEER did not reduce death or HF hospitalization in secondary MR (different patient selection vs COAPT) | PMID 30280674 |
| PROTECT-AF | 2014 | WATCHMAN non-inferior to warfarin for stroke prevention in AF; reduced hemorrhagic stroke and mortality at 3.8 yr | PMID 25065991 |
| RESPECT | 2017 | PFO closure reduced recurrent stroke vs medical therapy in cryptogenic stroke patients | PMID 28902580 |
Mechanical Circulatory Support
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| IABP-SHOCK II | 2012 | IABP did not reduce 30-day mortality in cardiogenic shock complicating MI | PMID 23013624 |
| PROTECT III | 2024 | Impella-supported high-risk PCI reduced 90-day major adverse events vs IABP | PMID 38587237 |
Bifurcation & CTO Trials
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| DKCRUSH-V | 2017 | DK-crush technique superior to provisional stenting for distal left main bifurcation (true bifurcations) | PMID 28622955 |
| EBC MAIN | 2023 | Systematic two-stent strategy non-inferior to provisional stenting for left main bifurcation with complex anatomy | PMID 37634136 |
| EURO-CTO | 2018 | CTO PCI improved angina, quality of life, and functional status vs medical therapy | PMID 30366740 |
| DISRUPT CAD III | 2021 | IVL safe and effective for calcium modification before stenting; 92.4% procedural success | PMID 33465776 |
Triple Therapy Trials
| Trial | Year | Key Finding | Reference |
|---|---|---|---|
| WOEST | 2013 | OAC + clopidogrel (no aspirin) reduced bleeding vs triple therapy in AF + PCI | PMID 23415013 |
| RE-DUAL PCI | 2017 | Dabigatran dual therapy reduced bleeding vs triple therapy in AF + PCI | PMID 28844200 |
| ENTRUST-AF PCI | 2019 | Edoxaban + P2Y12 non-inferior to VKA-based triple therapy for bleeding/ischemic events | PMID 31556371 |