Interventional Radiology
Every procedure, access technique, device, embolization agent, drainage protocol, classification, complication, medication, and management algorithm across the full scope of interventional radiology in one place.
01 Vascular Anatomy for IR
Aorta & Major Branches
The abdominal aorta enters the abdomen through the aortic hiatus of the diaphragm at T12 and bifurcates into the common iliac arteries at the level of L4. Major anterior visceral branches (in descending order): celiac trunk (T12) — left gastric, splenic, and common hepatic arteries; superior mesenteric artery (SMA) (L1) — supplies jejunum, ileum, ascending and transverse colon; inferior mesenteric artery (IMA) (L3) — supplies descending colon, sigmoid, and superior rectum. Lateral branches: renal arteries (L1-L2, right crosses behind the IVC), gonadal arteries, and paired lumbar arteries.
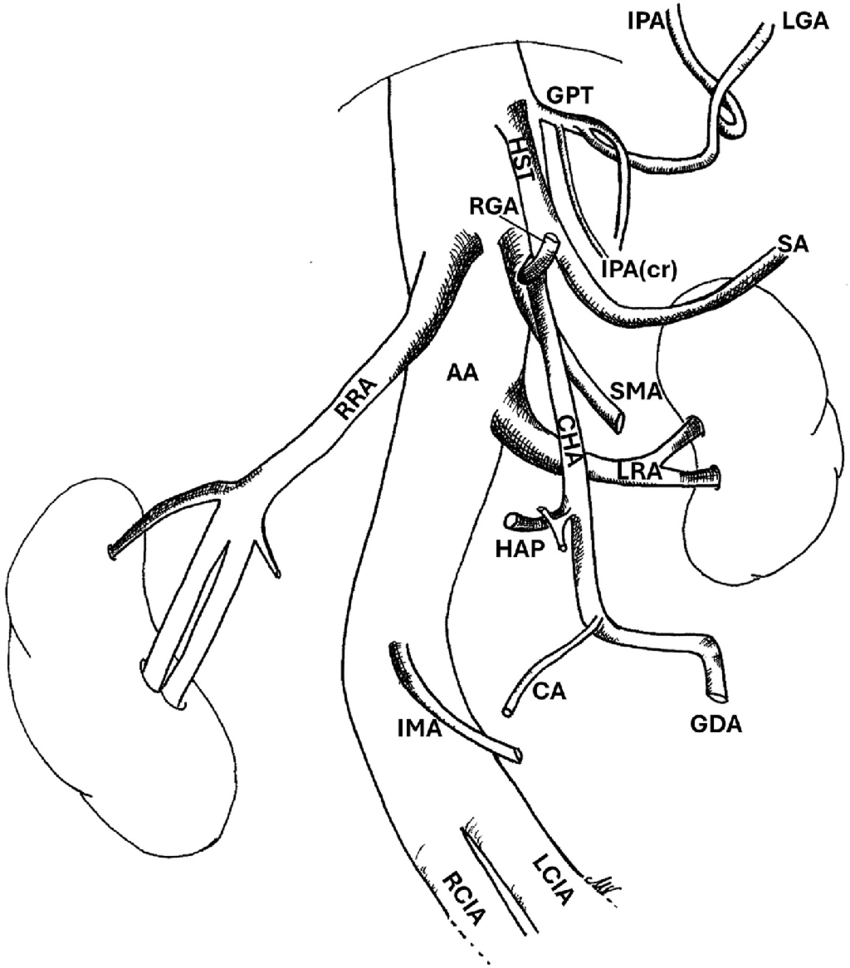
The celiac trunk has classic branching in ~55-75% of patients. Important variants: the replaced right hepatic artery (arises from the SMA, ~15-20% of patients) — must be identified on pre-procedural imaging before hepatic embolization or chemoembolization; the replaced left hepatic artery (arises from the left gastric artery, ~10-15%); and the hepatomesenteric trunk (common hepatic artery arises from the SMA). The gastroduodenal artery (GDA) is a key landmark — it is the first branch of the common hepatic artery and is the most common source of bleeding from duodenal ulcers.
Hepatic Arterial Anatomy & the Michel Classification
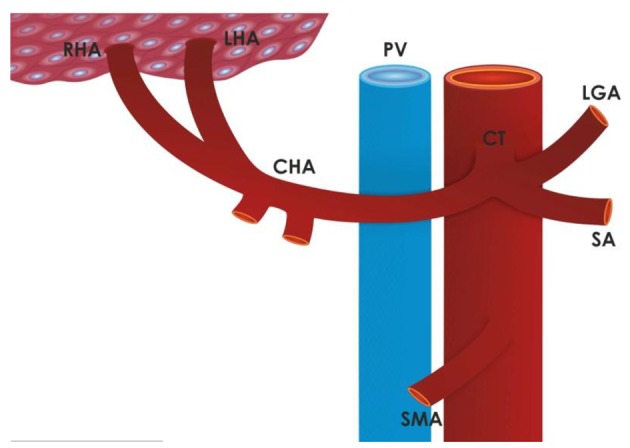
The standard hepatic arterial anatomy (Michel type I, ~55%) consists of the common hepatic artery arising from the celiac trunk, giving off the GDA, and continuing as the proper hepatic artery, which bifurcates into the right and left hepatic arteries. The Michel classification describes 10 variants:
| Type | Description | Frequency |
|---|---|---|
| I | Standard anatomy (CHA from celiac trunk) | ~55% |
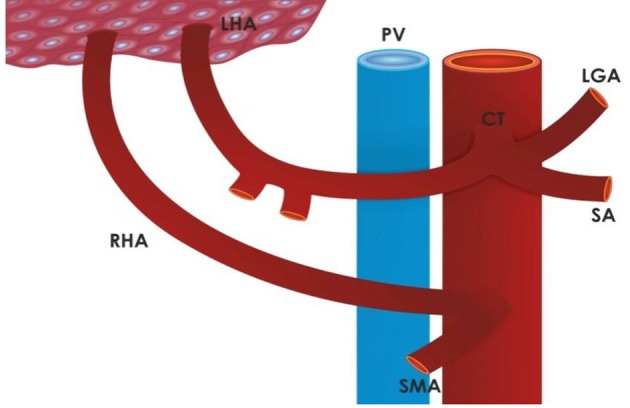
| II | Replaced LHA from left gastric artery | ~10% |
| III | Replaced RHA from SMA | ~11% |
| IV | Replaced RHA from SMA + replaced LHA from left gastric | ~1% |
| V | Accessory LHA from left gastric artery | ~8% |
| VI | Accessory RHA from SMA | ~7% |
| VII | Accessory RHA from SMA + accessory LHA from left gastric | ~1% |
| VIII | Replaced RHA from SMA + accessory LHA from left gastric (or vice versa) | ~2% |
| IX | CHA from SMA (hepatomesenteric trunk) | ~4.5% |
| X | CHA from left gastric artery | ~0.5% |
Portal Venous System
The portal vein is formed by the confluence of the splenic vein and the superior mesenteric vein (SMV) behind the neck of the pancreas. It carries ~75% of hepatic blood flow (but only ~50% of hepatic oxygen supply). The portal vein divides into right and left portal vein branches at the hepatic hilum. The inferior mesenteric vein (IMV) typically drains into the splenic vein. Normal portal venous pressure is 5-10 mmHg; portal hypertension is defined as a hepatic venous pressure gradient (HVPG) ≥ 6 mmHg, with clinically significant portal hypertension at HVPG ≥ 10 mmHg.
Important portosystemic collateral pathways in portal hypertension: left gastric (coronary) vein → esophageal varices; umbilical vein (recanalized) → caput medusae; superior rectal → middle/inferior rectal veins → rectal varices; retroperitoneal (of Retzius) veins; and splenorenal shunts. Understanding these pathways is critical for TIPS planning and variceal embolization.
Inferior Vena Cava & Hepatic Veins
The IVC is formed by the confluence of the common iliac veins at L5. It receives the renal veins (the left renal vein crosses anterior to the aorta and posterior to the SMA — the "nutcracker" position), the hepatic veins (right, middle, left — draining into the IVC just below the diaphragm), and the lumbar veins. The right renal vein is short and enters the IVC directly; the left renal vein is longer and receives the left gonadal vein and left adrenal vein. The infrarenal IVC is the standard location for IVC filter placement — a diameter > 28 mm may require a suprarenal filter or a bird's nest filter.
Iliac & Lower Extremity Arterial Anatomy
Each common iliac artery bifurcates into the external iliac artery (becomes the common femoral artery at the inguinal ligament) and the internal iliac (hypogastric) artery (supplies pelvic organs — critical in pelvic trauma embolization). The internal iliac artery divides into an anterior division (obturator, inferior vesical, middle rectal, internal pudendal, inferior gluteal, uterine arteries) and a posterior division (iliolumbar, lateral sacral, superior gluteal arteries). Knowledge of internal iliac branches is essential for pelvic embolization — the superior gluteal artery is the largest branch and passes through the greater sciatic foramen; non-target embolization can cause buttock claudication or necrosis.
The common femoral artery (CFA) is the primary access point for most IR procedures. It lies medial to the femoral nerve and lateral to the femoral vein (mnemonic: NAVEL — Nerve, Artery, Vein, Empty space, Lymphatics, from lateral to medial). The CFA bifurcates into the superficial femoral artery (SFA) and the profunda femoris (deep femoral artery). The SFA courses through the adductor canal and becomes the popliteal artery at the adductor hiatus. Below the knee, the popliteal artery trifurcates into the anterior tibial artery (becomes the dorsalis pedis), the posterior tibial artery (courses behind the medial malleolus), and the peroneal (fibular) artery.
Upper Extremity & Cerebrovascular Anatomy
The aortic arch gives rise to three great vessels (most common configuration, ~70%): the brachiocephalic (innominate) artery (divides into the right common carotid and right subclavian arteries), the left common carotid artery, and the left subclavian artery. Common variant: a bovine arch (~15-25%) where the left common carotid arises from the brachiocephalic artery. The vertebral arteries arise from the subclavian arteries and ascend through the transverse foramina of C6-C1 to join at the pontomedullary junction, forming the basilar artery. Left vertebral dominance is present in ~50% of patients; right dominance in ~25%; codominance in ~25%. The circle of Willis connects the anterior (carotid) and posterior (vertebrobasilar) circulations via the anterior communicating artery and posterior communicating arteries — providing collateral pathways, though a complete circle is present in only ~20-25% of the population.
02 Image Guidance & Radiation Safety
Fluoroscopy
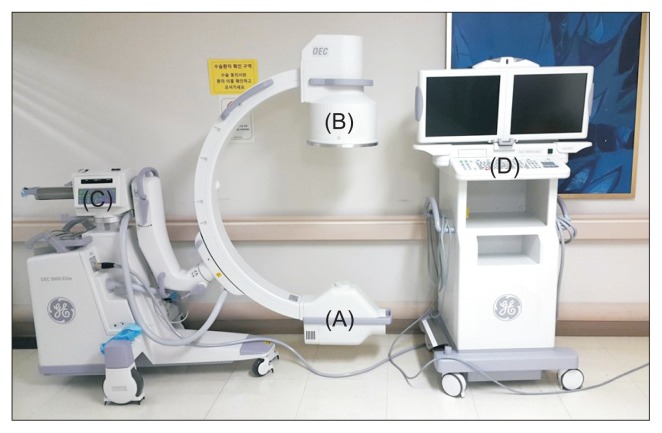
Fluoroscopy is the workhorse imaging modality for vascular IR procedures. It provides real-time X-ray imaging using a C-arm with an image intensifier (II, older technology) or flat-panel detector (FPD, modern — provides superior image quality, wider dynamic range, and lower dose than II systems). The C-arm can be rotated in multiple planes (anteroposterior, lateral, oblique) to provide different projections of the anatomy. Key concepts:
Pulse fluoroscopy (reduces radiation dose by pulsing the X-ray beam at intervals rather than continuous exposure — standard rates are 7.5 or 15 pulses/second; lower rates reduce dose proportionally but decrease temporal resolution); last-image hold (captures and displays the last fluoroscopic frame without additional radiation — use this to study anatomy rather than maintaining live fluoroscopy); magnification (electronic zoom that increases spatial resolution for detailed work but significantly increases radiation dose — doubling magnification can quadruple the dose due to increased mA required to maintain image quality in the smaller FOV; use only when necessary and return to normal magnification when possible); and digital subtraction angiography (DSA) (subtracts a pre-contrast mask image from contrast-enhanced images to display only the opacified vessels without overlying bone or soft tissue — the gold standard for vascular imaging in the angiography suite).
Cone-Beam CT (CBCT)
Cone-beam CT (also called DynaCT, XperCT, or Innova CT depending on the manufacturer) uses the C-arm to acquire a rotational dataset (typically 200-degree rotation over 5-20 seconds) that is reconstructed into cross-sectional images. Advantages: can be performed in the angiography suite without moving the patient, provides 3D localization during embolization or ablation procedures, identifies contrast extravasation or embolization endpoint, and enables CT-like needle guidance within the angio suite (needle guidance software overlays a planned needle trajectory on live fluoroscopy).
Clinical applications: during TACE/TARE, CBCT with intra-arterial contrast injection confirms catheter position within the tumor-feeding territory, identifies non-target supply that could lead to complications, and maps the perfused treatment volume in 3D. During ablation, CBCT can assess ablation zone coverage relative to the tumor margins. During complex embolization (Type II endoleak, pelvic hemorrhage), CBCT helps localize the bleeding source when conventional 2D DSA is inconclusive. Limitation: lower soft-tissue contrast resolution compared to diagnostic CT; motion artifacts are more pronounced due to the longer acquisition time.
Ultrasound Guidance
Ultrasound provides real-time, radiation-free guidance and is the first-line modality for vascular access (CFA, IJV, radial artery), abscess drainage, biopsy of superficial structures, paracentesis, thoracentesis, and nephrostomy. Key techniques: in-plane (needle parallel to the transducer — visualizes the entire needle shaft) vs out-of-plane (needle perpendicular to the transducer — visualizes only the needle cross-section). Doppler modes: color Doppler (shows flow direction — red toward, blue away by convention), spectral Doppler (waveform analysis — arterial vs venous), and power Doppler (sensitive to slow flow but does not show direction).
CT Guidance
CT-guided procedures include deep biopsy (lung, retroperitoneal, bone), abscess drainage, nerve blocks (celiac plexus, sympathetic chain, pudendal), and tumor ablation. Advantages: excellent spatial resolution (sub-millimeter in modern MDCT), precise visualization of deep structures and their relationship to the needle, reproducible needle trajectories using gantry angle and laser alignment, and the ability to confirm needle position before committing to biopsy/ablation. Disadvantages: non-real-time (intermittent scanning — "step and shoot" technique), ionizing radiation to both patient and operator, and needle approach traditionally limited to the axial plane (though angulated gantry and multiplanar reformats allow oblique trajectories).
CT fluoroscopy (continuous or near-continuous CT imaging during needle advancement) provides near-real-time visualization of the needle and target but significantly increases operator radiation exposure to the hands — require protective measures (lead gloves, needle holders that distance the hands from the scan plane). Newer technologies: CT-guided robotic systems (e.g., MAXIO by Perfint) plan and orient a needle guide robotically based on CT images, reducing operator radiation exposure and improving targeting accuracy. Electromagnetic navigation systems (SuperDimension/Medtronic) use EM field-guided catheters for bronchoscopic navigation to peripheral lung nodules, combining endobronchial and percutaneous approaches. Augmented reality / navigation platforms overlay planned needle trajectories on the patient using optical tracking cameras, enabling freehand needle guidance with real-time trajectory feedback.
Radiation Safety — ALARA Principle
The ALARA (As Low As Reasonably Achievable) principle governs radiation protection in IR. Three pillars of dose reduction: time (minimize fluoroscopy time), distance (inverse square law — doubling distance reduces dose by 75%), and shielding (lead aprons, thyroid shields, leaded glasses, table-mounted and ceiling-mounted lead shields).
Air kerma (Ka): Cumulative radiation dose at the interventional reference point (IRP) — the reference for skin dose. Measured in Gy. Threshold for deterministic skin injury: ~2 Gy (transient erythema), ~5 Gy (main erythema), ~15 Gy (dermal necrosis).
Dose-area product (DAP / KAP): Air kerma multiplied by the irradiated area — correlates with stochastic risk (cancer). Measured in Gy·cm².
Fluoroscopy time: Recorded for every procedure but is a poor surrogate for actual dose (does not account for magnification, patient size, or DSA runs).
Substantial radiation dose level (SRDL): Joint Commission requires notification when reference air kerma exceeds 5 Gy for a single procedure.
03 Access, Wires, Catheters & Closure Devices
The Seldinger Technique
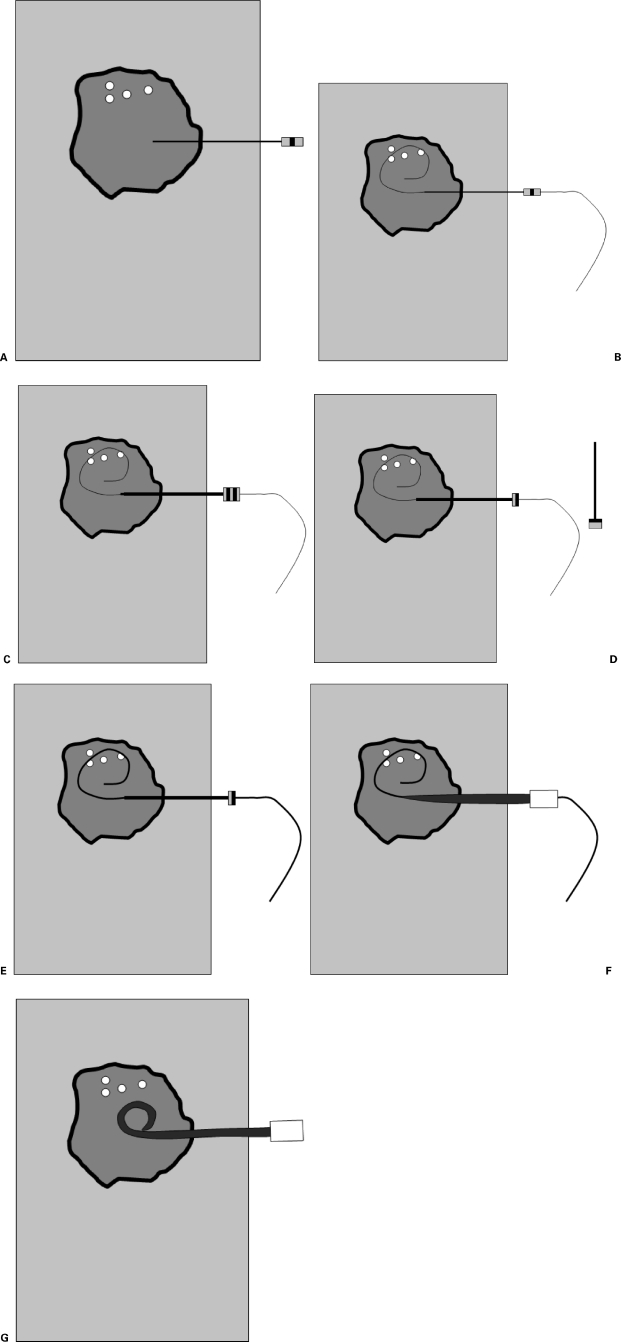
The Seldinger technique (described by Swedish radiologist Sven-Ivar Seldinger in 1953, published in Acta Radiologica) is the foundation of all percutaneous vascular and non-vascular access in IR and is one of the most important innovations in the history of medicine. Before this technique, vascular access required surgical cutdown. Steps: (1) puncture the target vessel/structure with a hollow needle under image guidance (US, fluoroscopy, or CT); (2) advance a guidewire through the needle into the lumen (confirm intraluminal position by observing the wire course under fluoroscopy or by aspirating blood/fluid); (3) remove the needle over the wire while maintaining wire position; (4) advance a catheter, dilator, or sheath over the wire into the desired position (critical: always maintain control of the wire — the proximal end of the wire must be visible and secured at all times to prevent wire embolization); (5) remove the wire.
The modified Seldinger technique uses a micropuncture set (21-gauge needle, 0.018-inch wire, coaxial transitional dilator that accepts the 0.018" wire and converts to a 0.035" system) to minimize access trauma. The micropuncture technique is standard for most IR procedures because the smaller needle and wire produce less vessel injury, and if the wrong structure is inadvertently punctured (e.g., the femoral artery during intended venous access), the small puncture is more easily managed. The micropuncture kit is particularly valuable for: radial artery access (small vessel), pediatric access, transplant renal biopsy (minimize parenchymal injury), and any access where precision is critical.
Access Sites
Common femoral artery (CFA): The most common arterial access site. Access over the medial third of the femoral head on fluoroscopy (between the inguinal ligament superiorly and the femoral bifurcation inferiorly). The CFA is preferred because it is compressible against the femoral head for hemostasis.
Radial artery: Increasingly used for neurointervention and coronary/peripheral interventions. Advantages: lower access-site bleeding complications, earlier ambulation. Requires a positive Allen test or Barbeau test (confirming dual blood supply via the ulnar artery). Uses 4-6 Fr sheaths.
Internal jugular vein (IJV): Primary venous access for TIPS, IVC filter placement, central venous catheter placement. Accessed under ultrasound guidance. Right IJV preferred for a straight path to the SVC/RA.
Popliteal artery: Retrograde access for SFA/iliac interventions when antegrade CFA access is not feasible. Accessed prone under US guidance.
Pedal access (dorsalis pedis, posterior tibial): Used for retrograde recanalization of tibial occlusions in CLI.
Guidewires
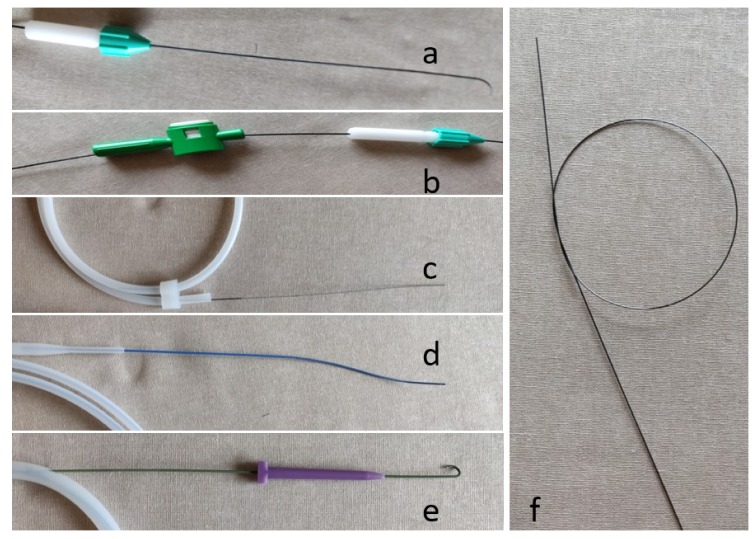
Bentson wire (0.035", 145-180 cm): Soft, floppy J-tip wire. Used for initial access and atraumatic advancement through normal vasculature. The standard "first wire."
Glidewire (Terumo, 0.035"): Hydrophilic-coated, angled tip. Used to cross stenoses and occlusions, navigate tortuous vessels. Essential for subintimal recanalization. Caution: can dissect or perforate if advanced without caution due to its slippery coating.
Amplatz Super Stiff (0.035", 260 cm): Stiff wire providing excellent rail support for large sheath/device delivery. Used for stent graft deployment, TIPS, and any procedure requiring a rigid platform.
Rosen wire (0.035", 180 cm): Curved floppy tip with a stiff shaft. Used for catheter exchanges requiring support while maintaining a safe, atraumatic tip position.
V-18 ControlWire (0.018"): Used through microcatheters for subselective embolization, crossing tight stenoses via micropuncture systems.
Lunderquist wire (0.035", 260 cm): Extra-stiff wire for EVAR, TEVAR, and transjugular procedures requiring maximum support.
Catheter Shapes
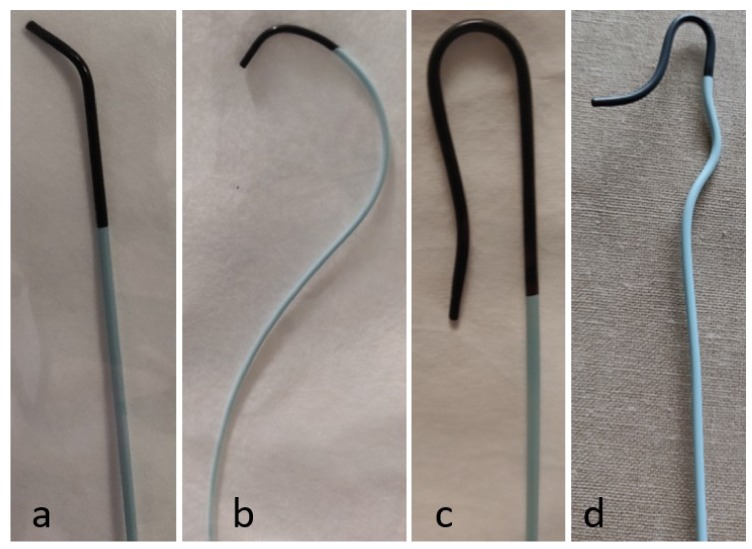
Cobra (C1, C2, C3): The C2 is the workhorse selective catheter. Used for catheterization of downward-oriented branches (renal arteries, celiac, SMA) from a femoral approach. The larger the Cobra number, the larger the primary curve.
SOS Omni / Sos: Reverse-curve catheter for selecting branches that arise at acute angles (e.g., celiac trunk, mesenteric arteries from a femoral approach in patients with a steep aortic bifurcation). Requires reformation in the aorta.
Simmons (Sim 1, 2, 3): Reverse-curve catheter commonly used for arch vessel catheterization from a femoral approach (carotids, subclavians). Also reformed in the aortic arch. The Simmons 2 is most commonly used.
Pigtail: Multi-side-hole catheter for high-flow injections (aortography, ventriculography). The pigtail prevents recoil and provides uniform contrast delivery.
Microcatheters (2.0-3.0 Fr): Coaxial catheters advanced through standard diagnostic catheters for superselective catheterization. Examples: Renegade (Boston Scientific), Progreat (Terumo). Essential for embolization to minimize non-target injury.
Sheaths
Sheaths (introducer sheaths) provide a working channel that protects the vessel wall from repeated catheter exchanges, maintain access to the vessel, incorporate a hemostatic valve that prevents blood loss around the catheter, and have a sidearm port for continuous heparinized saline flushing (prevents thrombus formation within the sheath). Sheath size is described by the inner diameter in French (Fr; 1 Fr = 0.33 mm) — a 7 Fr sheath accepts catheters up to 7 Fr outer diameter. However, the sheath's outer diameter is typically 1.5-2 Fr larger than its inner diameter (e.g., a 7 Fr sheath has an ~8.5 Fr outer diameter), which determines the actual vessel puncture size.
Sizes range from 4 Fr (diagnostic angiography) to 20+ Fr (EVAR, mechanical thrombectomy for PE). A 7 Fr sheath is standard for most interventional procedures (accepts 5 Fr catheters with room for contrast injection). Common configurations: short sheaths (10-15 cm — standard for CFA access), long sheaths (45-90 cm — provide additional support for catheter advancement through tortuous anatomy, allow working closer to the target lesion, and enable contrast injection at the lesion level without separate catheter exchanges; examples include Destination/Raabe sheaths for crossover iliac work, Shuttle sheaths for renal/mesenteric interventions). Guiding sheaths/catheters (6-8 Fr, 80-100 cm, with preformed curves) combine the functions of a sheath and a selective catheter, providing both access and directional guidance for neurointervention and complex peripheral procedures.
Vascular Closure Devices
Manual compression: Standard for ≤ 6 Fr access sites. Apply direct pressure over the arteriotomy (not the skin puncture, which may be lower) for 15-20 minutes, followed by bed rest (1-2 hours for 4 Fr, 4-6 hours for 6 Fr). Compression must be firm enough to achieve hemostasis but not occlusive to distal flow — check distal pulse during compression.
Angio-Seal (Terumo): Collagen plug + intravascular anchor + suture. Deploys a bioabsorbable anchor inside the vessel and a collagen plug outside the arteriotomy. Available in 6 Fr and 8 Fr sizes. Anchor and collagen resorb within 60-90 days. Contraindicated in severely calcified or small (< 4 mm) CFAs. Re-access at the same site should be avoided for 90 days.
Mynx (Cardinal Health): Extravascular polyethylene glycol (PEG) sealant. Deploys a water-soluble plug outside the arteriotomy. Dissolves within 30 days. Available for 5-7 Fr sheaths. Advantage: no intravascular component, so does not restrict re-access timing.
Perclose ProGlide (Abbott): Suture-mediated closure. Deploys a monofilament polypropylene suture through the arteriotomy in a figure-of-eight pattern. Can be used in a "preclose" technique for large-bore access: deploy two Perclose devices at 10 and 2 o'clock positions before upsizing the sheath, perform the procedure, then tighten the sutures to close the arteriotomy after device/sheath removal. This technique is standard for EVAR (12-20 Fr), TAVR, and other large-sheath procedures. Success rate ~90-95% with experienced operators.
StarClose SE (Abbott): Nitinol clip that apposes the edges of the arteriotomy extravascularly. Available for 5-6 Fr sheaths. Rapid hemostasis with minimal vessel intrusion.
MANTA (Teleflex): Collagen-based closure device designed specifically for large-bore access (12-25 Fr). Uses an intravascular toggle and extravascular collagen pad with a locking mechanism. Increasingly used for TAVR and EVAR access closure as an alternative to the preclose technique.
04 Contrast Agents & Procedural Sedation
Iodinated Contrast Media
Iodinated contrast agents are classified by osmolality and ionicity. High-osmolar contrast media (HOCM): Ionic monomers (e.g., diatrizoate/Hypaque) — osmolality ~1,500-2,000 mOsm/kg, highest rate of adverse reactions. Rarely used today. Low-osmolar contrast media (LOCM): Non-ionic monomers (e.g., iohexol/Omnipaque, iopamidol/Isovue, ioversol/Optiray) — osmolality ~600-800 mOsm/kg. Standard of care for nearly all IR procedures. Iso-osmolar contrast media (IOCM): Non-ionic dimers (e.g., iodixanol/Visipaque) — osmolality ~290 mOsm/kg (equal to blood). May have a lower risk of contrast-induced nephropathy in high-risk patients, though data are mixed.
CO2 Angiography
Carbon dioxide (CO2) can be used as a negative contrast agent when iodinated contrast is contraindicated (severe allergy, renal insufficiency). CO2 is buoyant (rises to the non-dependent surface of the vessel, which affects image acquisition — the patient may need to be rotated to opacify dependent vessel segments), non-nephrotoxic, non-allergenic, and is rapidly absorbed from the bloodstream (30-40 times more soluble in blood than nitrogen/air). These properties make CO2 an excellent alternative contrast agent for patients with CKD or severe iodinated contrast allergy undergoing procedures below the diaphragm.
Technique: CO2 is hand-injected (using a dedicated closed delivery system with a one-way valve to prevent air contamination) or delivered via an automated CO2 injector system (AngioDynamics, Leland Gas). It displaces blood and appears as a negative (dark) filling defect on DSA — specialized DSA processing with "stacking" of multiple images improves vessel visualization. CO2 DSA images are typically presented as reversed (black vessels on white background) for easier interpretation. Allow 2-3 minutes between injections for CO2 to be absorbed before re-injection. Maximum injection volume: 100-150 mL per injection; total procedure limit is not well defined but should be minimized.
Limitations and safety: CO2 is absolutely contraindicated above the diaphragm (risk of cerebral air/CO2 embolism causing stroke; even small volumes of CO2 in the cerebral circulation can cause neurological injury); provides lower image quality than iodinated contrast (motion artifact, incomplete vessel filling, inability to assess contrast blush or parenchymal enhancement); can cause mesenteric ischemia with repeated large-volume injections or trapped gas in mesenteric vessels (vapor lock phenomenon — gas bubbles obstruct small arterioles; treated by trendelenburg positioning and IV vasodilators); may cause transient abdominal pain during injection. An explosive delivery system (plastic bag method) must never be used — contamination of CO2 with room air introduces nitrogen, which is far less soluble in blood and increases the risk of embolic complications. Only dedicated closed systems with medical-grade CO2 should be used.
Gadolinium-Based Contrast
Gadolinium chelates are primarily MRI contrast agents but can be used off-label for fluoroscopic angiography in patients with iodinated contrast allergy who cannot receive CO2 (above the diaphragm) or when CO2 provides insufficient image quality. Common agents: gadopentetate dimeglumine (Magnevist — linear, ionic), gadobutrol (Gadavist — macrocyclic, highest concentration at 1 mmol/mL), gadoterate meglumine (Dotarem — macrocyclic, ionic). However, gadolinium provides lower vascular opacification than iodinated contrast at safe doses (limited to 0.3-0.4 mmol/kg) and should be diluted with saline to increase injectable volume.
Gadolinium carries the risk of nephrogenic systemic fibrosis (NSF) in patients with severe renal impairment (eGFR < 30), a debilitating and potentially fatal condition characterized by progressive fibrosis of the skin, joints, and internal organs. NSF is caused by free gadolinium ions released from unstable chelate molecules. ACR risk classification: Group I (high-risk) agents (gadodiamide/Omniscan, gadopentetate dimeglumine/Magnevist, gadoversetamide/OptiMARK — all linear chelates) should be avoided entirely in patients with eGFR < 30 or acute kidney injury. Group II (low-risk) agents (gadobutrol/Gadavist, gadoterate/Dotarem, gadoteridol/ProHance — all macrocyclic chelates) are considered safer and can be used with caution in patients with eGFR < 30 when the clinical benefit outweighs the risk. Since the adoption of Group II agents and avoidance of Group I agents in renal failure, new cases of NSF have become extremely rare. Gadolinium brain deposition (retention in the dentate nucleus and globus pallidus, visible on T1-weighted MRI) has been described even in patients with normal renal function after multiple gadolinium doses, particularly with linear agents; the clinical significance remains uncertain.
Contrast Reactions — ACR Classification & Management
Mild reactions: Limited urticaria, pruritus, nausea, single episode of emesis, mild bronchospasm. Management: Observation; diphenhydramine 25-50 mg IV for urticaria.
Moderate reactions: Diffuse urticaria/erythema, facial edema without dyspnea, bronchospasm with mild hypoxia, isolated chest tightness. Management: Diphenhydramine 25-50 mg IV; albuterol nebulizer 2.5 mg for bronchospasm; epinephrine 0.1-0.3 mg IM (1:1,000) for progressive symptoms.
Severe reactions: Diffuse edema or facial edema with dyspnea, severe bronchospasm/hypoxia, laryngeal edema, hypotension, anaphylactic shock. Management: Epinephrine IM 0.3 mg (1:1,000) — cornerstone of anaphylaxis management. IV fluid bolus for hypotension. IV epinephrine infusion (1:10,000) for refractory shock. Secure airway. Call for help.
Contrast Premedication Protocol
For patients with prior moderate/severe contrast reactions requiring iodinated contrast: ACR premedication protocol (elective, 13-hour regimen) — prednisone 50 mg PO at 13 hours, 7 hours, and 1 hour before the procedure + diphenhydramine 50 mg PO/IV/IM 1 hour before. Alternative for patients who cannot take oral medications: methylprednisolone 40 mg IV at 13, 7, and 1 hour before + diphenhydramine 50 mg IV 1 hour before.
For emergent/urgent procedures when the full 13-hour protocol is not feasible: hydrocortisone 200 mg IV immediately and every 4 hours until the procedure + diphenhydramine 50 mg IV 1 hour before. An accelerated 5-hour regimen (methylprednisolone 40 mg IV at 5 hours and 1 hour before + diphenhydramine 50 mg IV 1 hour before) has been described but has less evidence than the 13-hour protocol. Premedication reduces the incidence of repeat reactions from ~20-60% to ~5-10% but does not eliminate risk entirely — breakthrough reactions can occur despite adequate premedication. All premedicated patients should receive non-ionic, low-osmolar contrast (LOCM) rather than high-osmolar agents. A crash cart with epinephrine must be immediately available. If the procedure is elective and the patient had a prior severe/life-threatening reaction, consider alternative imaging modalities (CO2, gadolinium, MRA) or non-contrast approaches before committing to iodinated contrast with premedication.
Contrast-Induced Nephropathy (CIN) Prevention
CIN (also called post-contrast acute kidney injury, PC-AKI) is defined as a rise in serum creatinine ≥ 0.3 mg/dL or ≥ 50% above baseline within 48-72 hours of contrast administration. Risk factors: pre-existing CKD (eGFR < 30 highest risk), diabetes, dehydration, large contrast volumes, heart failure, concurrent nephrotoxic medications, and age > 70. There is ongoing debate about the true incidence of PC-AKI, as studies controlling for baseline AKI risk suggest that intravenous contrast may contribute less to AKI than previously believed.
Prevention: IV hydration (normal saline 1 mL/kg/hr for 6-12 hours before and 6-12 hours after) is the cornerstone prevention strategy. For outpatient/urgent procedures: rapid IV hydration with sodium bicarbonate (150 mEq in 1 L D5W, bolus 3 mL/kg over 1 hour before, then 1 mL/kg/hr for 6 hours after). Minimize contrast volume (aim for < 3.7 × eGFR mL or a contrast-to-creatinine-clearance ratio < 3.7). Hold nephrotoxic medications (NSAIDs, aminoglycosides) and metformin (for 48 hours post-contrast in patients with eGFR < 30 due to lactic acidosis risk if AKI develops). N-acetylcysteine (NAC) was widely used but the PRESERVE trial (PMID: 29130810, NEJM 2018) definitively showed no benefit over placebo. Consider CO2 angiography or gadolinium-based contrast as alternatives in very high-risk patients. Withholding contrast for a clinically necessary procedure (e.g., emergent angiography for hemorrhage) to avoid CIN is generally not appropriate — the risk of the untreated condition outweighs the CIN risk.
Moderate Sedation in IR
Most IR procedures are performed under moderate (conscious) sedation. The patient maintains protective airway reflexes and responds purposefully to verbal or light tactile stimulation. Standard regimen: midazolam (0.5-2 mg IV, anxiolytic/amnestic) + fentanyl (25-100 mcg IV, analgesic). Titrate to effect. Reversal agents: flumazenil (0.2 mg IV for benzodiazepine reversal) and naloxone (0.04-0.4 mg IV for opioid reversal). Monitoring requirements: continuous pulse oximetry, cardiac monitoring, blood pressure every 5 minutes, and end-tidal CO2 (capnography) per current standards. A pre-sedation assessment including NPO status (ASA guidelines: 2 hours clear liquids, 6 hours light meal, 8 hours full meal) and Mallampati score is mandatory.
Minimal sedation (anxiolysis): Normal response to verbal stimulation. Airway, ventilation, and cardiovascular function unaffected. Example: single dose of midazolam.
Moderate sedation (conscious sedation): Purposeful response to verbal or light tactile stimulation. No airway intervention required. Spontaneous ventilation adequate. Cardiovascular function usually maintained. Standard for most IR procedures.
Deep sedation: Purposeful response only to repeated or painful stimulation. Airway intervention may be required. Spontaneous ventilation may be inadequate. Cardiovascular function usually maintained. Requires an anesthesia provider in many institutions.
General anesthesia: Not arousable even with painful stimulation. Airway intervention usually required (ETT/LMA). Ventilatory function frequently inadequate. Cardiovascular function may be impaired. Used for complex procedures (EVAR/TEVAR, TIPS, pediatric cases).
Prophylactic Antibiotics in IR
Not all IR procedures require prophylactic antibiotics. SIR guidelines recommend antibiotics for: biliary procedures (PTC, biliary drain placement/exchange — cefazolin 1 g IV or ciprofloxacin if penicillin-allergic), genitourinary procedures (nephrostomy, ureteral stent — cefazolin 1 g IV or gentamicin), procedures in patients with bilioenteric anastomosis undergoing hepatic embolization (piperacillin-tazobactam or fluoroquinolone + metronidazole), tunneled CVC and port placement (cefazolin 1 g IV — recommended by some, though data are limited), and procedures involving prosthetic material implantation (stent grafts). TACE/TARE: routine prophylactic antibiotics are not indicated unless bilioenteric anastomosis is present (high abscess risk). Drainage of infected collections: antibiotics should be empiric and broad-spectrum until culture results are available.
05 Angioplasty & Stenting
Percutaneous Transluminal Angioplasty (PTA)

PTA, introduced by Andreas Gruentzig in 1974, uses a balloon catheter to dilate a stenotic or occluded vessel segment. The balloon is inflated to a specified pressure (rated burst pressure in atmospheres, typically 6-20 atm depending on balloon type) for 30-120 seconds (longer inflations may improve outcomes in tibial arteries — 3-5 minutes is common for infrapopliteal PTA). The mechanism of action is controlled intimal dissection (cracking the plaque), medial stretching, and adventitial expansion — the vessel lumen is enlarged by displacing and fracturing the obstructing plaque rather than by compressing it.
Balloon selection: diameter should match the reference vessel diameter (1:1 ratio for arteries) or slightly oversize (1.1:1 for veins, which are more compliant). Undersizing leads to inadequate dilation and early elastic recoil; oversizing risks vessel rupture or excessive dissection. Balloon types: standard (semi-compliant) — the balloon diameter increases with increasing pressure, providing gradual dilation; non-compliant (high-pressure) — maintains a fixed diameter even at high pressures (20-30 atm), used for resistant or fibrotic stenoses (dialysis access, biliary strictures); cutting balloon — has 3-4 microsurgical blades (atherotomes) on the balloon surface that score the plaque during inflation, reducing elastic recoil; used for resistant stenoses and in-stent restenosis; and scoring balloon (AngioSculpt, Chocolate) — has external elements (nitinol cage or scoring wires) that concentrate dilation force on the plaque, reducing vessel wall barotrauma. Balloon length should cover the entire lesion with a few millimeters of margin on each side. For long lesions, overlapping balloon inflations are performed rather than using excessively long balloons.
Drug-Coated Balloons (DCB)
Drug-coated balloons deliver paclitaxel (an antiproliferative agent that stabilizes microtubules and inhibits cell division) to the vessel wall during a standard-duration balloon inflation (typically 60-180 seconds), inhibiting neointimal hyperplasia and restenosis without leaving a permanent implant. The drug is embedded in an excipient matrix on the balloon surface that facilitates rapid transfer to the vessel wall upon inflation. Drug dose: typically 3.0 μg/mm² paclitaxel. Key advantage over bare PTA: significantly reduced restenosis rates by inhibiting smooth muscle cell proliferation; key advantage over drug-eluting stents: no permanent metallic scaffold left in the vessel (preserving vessel compliance and future treatment options).
The IN.PACT Admiral (Medtronic) and Lutonix (BD/Bard) DCBs have demonstrated superior patency over standard PTA in the femoropopliteal segment. Key evidence: IN.PACT SFA trial showed 82.2% primary patency at 12 months vs 52.4% for PTA (PMID: 25774601); Lutonix SFA trial showed 73.5% vs 56.8% (PMID: 25706499). DCBs are now first-line for femoropopliteal disease of moderate length (5-15 cm) in many centers and are increasingly used for in-stent restenosis, AV dialysis access stenosis, and below-the-knee disease. The paclitaxel safety signal: a 2018 patient-level meta-analysis by Katsanos et al. (PMID: 30516812) raised concerns about increased all-cause mortality with paclitaxel-coated devices (DCB and DES) at 2+ years. This led to an FDA advisory panel review in 2019, which concluded that the evidence was insufficient to establish a causal relationship. Subsequent larger analyses (SWEDEPAD trial, long-term IN.PACT data) and the FDA review found no confirmed safety signal, and DCBs remain in widespread clinical use with informed consent regarding the ongoing investigation.
Stents — Bare Metal vs Covered
Bare metal stents (BMS): Two main types based on deployment mechanism: Self-expanding stents (nitinol shape-memory alloy, e.g., SMART, Zilver, Complete SE, Innova — compressed in a delivery sheath and expand to their predetermined diameter upon release; provide continuous outward radial force; preferred in the femoropopliteal segment where the vessel is subject to flexion, compression, and torsion during leg movement). Balloon-expandable stents (stainless steel or cobalt-chromium, e.g., Palmaz, Express LD, Omnilink, Valeo — mounted on a balloon and expanded to the desired diameter by balloon inflation; provide precise deployment, high radial force, and the ability to flare the stent; preferred at the aortic bifurcation, iliac artery ostia, and renal artery ostia where precision and radial strength are paramount). Key principle: self-expanding for mobility zones; balloon-expandable for precision and radial force.
Covered stents (stent grafts): A stent skeleton covered with PTFE (polytetrafluoroethylene), Dacron (polyester), or other biocompatible material. The covering creates a barrier between the bloodstream and the vessel wall, sealing off perforations, excluding aneurysms, and preventing tissue ingrowth. Applications: vessel rupture/perforation (emergent deployment to seal the defect), pseudoaneurysm exclusion, AV fistula closure, and treatment of long-segment iliac occlusions. Key devices: Viabahn (Gore) — self-expanding nitinol skeleton with ePTFE covering, available with heparin bioactive surface; used in the femoropopliteal segment and as a dialysis access stent graft. iCast/Atrium (Maquet) — balloon-expandable covered stent; used for iliac rupture, pseudoaneurysm, and transplant renal artery stenosis. The COBEST trial showed superiority of covered stents (Viabahn) over BMS for long (> 15 cm) femoropopliteal lesions (PMID: 23182126). Drug-eluting stents (Zilver PTX — paclitaxel-coated self-expanding nitinol stent) combine the scaffold function with antiproliferative drug delivery; the ZILVER PTX trial showed superior patency over PTA with provisional bare stenting.
Iliac Artery Interventions
Iliac artery stenosis/occlusion is classified by the TASC II classification (see Section 26). TASC A/B lesions: endovascular first. TASC C/D lesions: surgery historically preferred, but endovascular approaches are increasingly successful even for complex iliac disease, with primary patency rates of 80-90% at 5 years for stented iliac arteries. Technique: typically retrograde CFA access (ipsilateral or contralateral crossover via aortic bifurcation using a crossover sheath — Balkin or Raabe sheath). Kissing balloon angioplasty / kissing stents for bilateral common iliac stenoses involving the aortic bifurcation to prevent plaque shift and contralateral iliac compromise — both stents are deployed simultaneously so neither obstructs the contralateral ostium.
Stent selection: balloon-expandable stents (e.g., Palmaz, Express, Omnilink) are preferred for CIA ostial lesions (precise deployment, high radial force at the aortic bifurcation). Self-expanding stents (e.g., Wallstent, Protege, Zilver) are preferred for long EIA lesions (flexibility, conformability to vessel tortuosity). Covered stents (iCast, Viabahn) are used for iliac artery rupture/perforation, aneurysmal disease, and may provide improved patency for long iliac occlusions. Common iliac artery stenting has the highest primary patency of any peripheral stent location (~90% at 5 years). External iliac results are slightly inferior due to smaller vessel caliber and increased tortuosity.
Femoropopliteal Interventions
The SFA is the most commonly treated peripheral artery. The adductor canal (Hunter canal) is a region of high mechanical stress (flexion, compression, torsion) and a common site for restenosis and stent fracture. Treatment algorithm:
Short lesions (< 5 cm): PTA ± bail-out stenting (stent only for flow-limiting dissection or > 30% residual stenosis).
Moderate lesions (5-15 cm): DCB preferred (IN.PACT SFA, Lutonix data); atherectomy + DCB for calcified lesions.
Long lesions (> 15 cm) or CTO: Atherectomy + DCB, or covered stent (Viabahn — VIASTAR trial showed superiority over BMS for long lesions). For in-stent restenosis: DCB (first-line) or atherectomy + DCB.
Popliteal artery: Interwoven stent (Supera) preferred for lesions crossing the knee joint (superior fracture resistance due to its helical interlocking wire design). Avoid rigid stents in the popliteal artery — high fracture rate with traditional nitinol stents.
Chronic total occlusion (CTO) crossing techniques: intraluminal (wire navigated through the true lumen of the occlusion — preferred when possible), subintimal (wire advanced into the subintimal space and re-enters the true lumen distally — the BOLIA technique), and retrograde access (pedal or popliteal access for retrograde recanalization when antegrade crossing fails). Re-entry devices (Outback, Pioneer) assist with controlled re-entry from the subintimal space into the true lumen.
Infrapopliteal & Critical Limb Ischemia
Infrapopliteal (tibial) disease is the hallmark of chronic limb-threatening ischemia (CLTI), formerly critical limb ischemia (CLI). Defined as rest pain, non-healing ulcer, or gangrene with hemodynamic compromise (ankle pressure < 70 mmHg, toe pressure < 50 mmHg, or TcPO2 < 40 mmHg). The angiosome concept guides revascularization: direct in-line flow to the angiosome supplying the wound provides the best healing rates. Tibial angioplasty with long balloon inflations (3-5 minutes) is the primary endovascular approach; stenting is limited by small vessel caliber and high restenosis rates.
The WIfI classification (Wound, Ischemia, foot Infection) stratifies CLTI patients by limb threat level and guides the need for revascularization. Each component is graded 0-3. The GLASS (Global Limb Anatomic Staging System) classifies infrapopliteal lesion complexity (from grade I to III) to help predict endovascular vs surgical outcomes and guide treatment strategy.
Atherectomy
Atherectomy removes atherosclerotic plaque from the vessel wall before angioplasty/stenting. Types: directional atherectomy (HawkOne/SilverHawk — rotating blade within a housing that excises plaque into a nosecone); rotational atherectomy (Rotablator — diamond-tipped burr for heavily calcified coronary lesions; Phoenix — rotating blade for peripheral vessels); orbital atherectomy (Diamondback 360 — orbiting diamond-coated crown for calcified lesions); and laser atherectomy (Spectranetics Turbo-Elite — excimer laser that photoablates tissue). Atherectomy is often combined with DCB ("athero-DCB" strategy) for calcified femoropopliteal disease. Distal embolization protection devices should be used during atherectomy when feasible.
Renal Artery Interventions
Renal artery stenosis (RAS) is most commonly atherosclerotic (ostial, ~90% — the stenosis involves the aortic ostium/proximal renal artery, caused by an extension of aortic plaque) or due to fibromuscular dysplasia (FMD, mid-to-distal renal artery, ~10% — classic "string of beads" appearance on angiography, most common in young women). Other causes: Takayasu arteritis, neurofibromatosis, radiation arteritis, and transplant renal artery stenosis.
The landmark ASTRAL (2009, PMID: 19907042) and CORAL (2014, PMID: 24245566) trials showed no benefit of renal artery stenting over medical therapy alone for atherosclerotic RAS in terms of blood pressure control, renal events, or cardiovascular outcomes. These trials significantly reduced the volume of renal artery stenting procedures performed worldwide. However, critics note that both trials included patients with mild-to-moderate stenosis and excluded some high-risk patients who might benefit most.
Current indications for renal artery intervention: FMD (angioplasty alone without stenting — excellent results with cure or improvement of hypertension in ~60-80%; stenting is reserved for dissection or recoil after PTA), resistant hypertension refractory to maximal medical therapy (≥ 3 drugs including a diuretic) with hemodynamically significant bilateral RAS or RAS to a solitary functioning kidney, recurrent flash pulmonary edema (Pickering syndrome — bilateral RAS causing rapid fluid overload that is out of proportion to LV dysfunction; stenting can be curative), and progressive decline in renal function with bilateral severe RAS (especially if renal function worsens with RAAS inhibitor therapy). For atherosclerotic RAS, balloon-expandable stent placement at the ostium is standard if intervention is pursued (precise deployment at the aortic ostium, high radial force to resist elastic recoil; typically 5-7 mm diameter stent, slightly protruding 1-2 mm into the aortic lumen).
06 Thrombolysis & Thrombectomy
Catheter-Directed Thrombolysis (CDT)
CDT delivers a thrombolytic agent directly into the thrombus via a multi-sidehole catheter (e.g., Unifuse, CraggMcNamara). The standard agent is alteplase (tPA), infused at 0.5-1.0 mg/hr (typical total dose 10-20 mg over 12-24 hours). Alternative: reteplase (0.25-0.5 units/hr). Concurrent IV heparin is administered through the sheath (typically 500 units/hr, target aPTT 40-60 seconds — supratherapeutic aPTT increases hemorrhagic risk). Monitoring: check fibrinogen q6h (hold thrombolysis if fibrinogen < 200 mg/dL, stop if < 100 mg/dL), CBC, aPTT. The patient is monitored in an ICU or step-down unit during infusion. Follow-up angiography every 8-24 hours to assess lysis progress and identify underlying lesions for definitive treatment.
Absolute: Active internal bleeding (excluding access site), recent (< 3 months) cerebrovascular event (hemorrhagic stroke at any time, ischemic stroke < 3 months), intracranial/spinal neoplasm or AVM, known bleeding diathesis.
Relative: Major surgery or trauma within 10 days, uncontrolled hypertension (SBP > 180 mmHg), recent GI bleeding (within 10 days), pregnancy, hepatic failure with coagulopathy, bacterial endocarditis, diabetic hemorrhagic retinopathy.
Acute Limb Ischemia (ALI)
ALI is classified by the Rutherford classification: Class I (viable): No sensory loss, no muscle weakness, audible arterial and venous Doppler — not immediately threatened. Class IIa (marginally threatened): Minimal sensory loss (toes), no muscle weakness, inaudible arterial Doppler — salvageable with prompt treatment. Class IIb (immediately threatened): Sensory loss beyond toes, mild-moderate muscle weakness, inaudible arterial Doppler — salvageable with immediate revascularization. Class III (irreversible): Profound sensory loss, paralysis, no audible Doppler — major tissue loss inevitable, amputation required. CDT is preferred for Class I and IIa ALI; surgical thromboembolectomy or hybrid approaches for IIb; primary amputation for III.
Pharmacomechanical Thrombolysis
Pharmacomechanical thrombolysis combines mechanical thrombus disruption with thrombolytic agents to accelerate clot removal and reduce lytic infusion time. The AngioJet (Boston Scientific) uses the Bernoulli effect (rheolytic thrombectomy) to create a vacuum that fragments and aspirates thrombus; it can also deliver a "power pulse spray" of tPA into the clot followed by aspiration. The EKOS (Boston Scientific) EndoWave system uses ultrasound energy to enhance tPA penetration into the thrombus, allowing lower drug doses. The ATTRACT trial (DVT) showed pharmacomechanical CDT reduced post-thrombotic syndrome severity but not overall PTS rates at 24 months compared to anticoagulation alone (PMID: 29247646).
Mechanical Thrombectomy Devices
Indigo System (Penumbra): Continuous aspiration thrombectomy. Uses a large-bore catheter connected to a vacuum pump (Penumbra ENGINE). Available in 6-8 Fr sizes for peripheral and 16-20 Fr for PE. Also used for stroke thrombectomy (Jet 7 and ACE catheters).
FlowTriever (Inari Medical): Large-bore aspiration catheter (20-24 Fr) with three self-expanding nitinol mesh discs that capture and extract thrombus in a single pass. Specifically designed for massive and submassive PE. FLARE trial showed rapid reduction in RV/LV ratio without thrombolytics (PMID: 31072507).
ClotTriever (Inari Medical): Designed for iliofemoral DVT. Uses a coring element and collection bag to mechanically remove thrombus in a single session without thrombolytics. CLOUT registry showed excellent thrombus removal with low bleeding rates.
AngioJet (Boston Scientific): Rheolytic thrombectomy using high-velocity saline jets that create a Bernoulli effect. Used in arterial and venous thrombosis. Caution: can cause hemolysis, hyperkalemia, bradycardia (especially in PE — relative contraindication for right heart use).
Pulmonary Embolism Interventions
Catheter-directed therapy for PE is indicated for massive PE (systolic BP < 90 mmHg sustained for ≥ 15 minutes, cardiac arrest, or need for vasopressors/mechanical circulatory support) when systemic thrombolysis fails or is contraindicated, and increasingly for submassive PE (hemodynamically stable with RV dysfunction on echocardiography or CT [RV/LV ratio > 0.9] and/or elevated troponin/BNP indicating myocardial strain). The PE severity index (PESI) or simplified PESI (sPESI) score aids risk stratification. Pulmonary Embolism Response Teams (PERTs) — multidisciplinary teams including IR, cardiology, pulmonology, hematology, and CT surgery — are increasingly standard for triaging and managing intermediate- and high-risk PE patients in real time.
Catheter-directed options: CDT with tPA (typically 1 mg/hr per catheter × bilateral PA catheters × 12-24 hours — the ULTIMA and SEATTLE II trials validated this approach; total tPA dose ~20-24 mg, significantly less than systemic thrombolysis dose of 100 mg), EKOS ultrasound-accelerated CDT (high-frequency, low-power ultrasound enhances fibrin permeability to tPA; 10-24 mg tPA over 12-24 hours), suction thrombectomy (FlowTriever — FLASH registry showing > 98% technical success with no intracranial hemorrhage; does not require thrombolytics), and Indigo aspiration system. The choice depends on institutional expertise, device availability, bleeding risk profile, and patient factors. Surgical embolectomy remains an option for massive PE refractory to catheter-based therapies, particularly in institutions without 24/7 IR capability or when contraindications to all catheter-based approaches exist (e.g., left atrial thrombus in transit).
07 Venous Disease & IVC Filters
IVC Filter Indications
Absolute indication: Acute VTE (DVT or PE) with an absolute contraindication to anticoagulation (active hemorrhage, recent major surgery or trauma, hemorrhagic stroke). Relative indications: Failure of anticoagulation (recurrent PE despite therapeutic anticoagulation), massive PE with hemodynamic compromise (as an adjunct to thrombolysis), free-floating iliocaval thrombus, poor cardiopulmonary reserve where even a small recurrent PE would be fatal, and prophylactic placement in very high-risk trauma patients.
IVC Filter Types
Nearly all filters placed today are retrievable (optional) filters, which can be removed when the indication resolves. Examples: Gunther Tulip, Celect, Denali, Option ELITE, ALN. These filters have a hook at the apex for snare retrieval via a jugular approach. Permanent filters (e.g., Bird's Nest, Greenfield, TrapEase) are used when lifelong protection is needed or when the IVC diameter exceeds the size range of retrievable filters (typically > 28 mm).
Placement: Standard via right IJV (preferred — allows straight-line deployment) or right CFA. An IVC-gram is performed to measure IVC diameter (normal: 20-28 mm; > 28 mm may be too large for standard filters), confirm patency (filter should not be placed in a thrombosed IVC), identify the renal vein positions (the lowest renal vein is the landmark for infrarenal deployment), and exclude IVC anomalies (duplicated IVC present in ~3% — requires bilateral filters or a suprarenal filter; left-sided IVC present in ~0.5%). The filter is deployed in the infrarenal IVC, just below the lowest renal vein. Suprarenal placement is used when the thrombus extends to or above the renal veins, in pregnancy (to avoid compression by the gravid uterus in the infrarenal segment), in the setting of renal vein or ovarian vein thrombosis, or when there is a duplicated IVC with infrarenal filter already in place.
IVC Filter Retrieval
The FDA issued a safety communication in 2010 and 2014 recommending retrieval of IVC filters as soon as the indication resolves (typically within 30-60 days), and no later than when the risk-benefit profile no longer favors having the filter in place. Prolonged dwell time increases the risk of filter-related complications: IVC thrombosis (~10-15%), filter fracture (strut breakage with embolization to the heart or lungs), migration, perforation of IVC wall (tilt/strut penetration extending through the caval wall into adjacent structures — aorta, duodenum, vertebral body), and post-thrombotic syndrome.
Standard retrieval: jugular venous access, snare the hook at the filter apex, collapse the filter into a sheath, and remove. Success rate for uncomplicated retrieval within 30-60 days: > 98%. Advanced retrieval techniques for embedded, tilted, or fractured filters include: rigid forceps (e.g., Whitehorse, Cook endobronchial forceps — grasp the embedded hook or struts directly), laser sheath (excimer laser to lyse tissue ingrowth), wire loop technique (loop a wire between the filter struts and the caval wall to create countertraction), hangman technique (stiff wire used to displace a tilted filter into the retrieval sheath), and combined femoral-jugular approaches (wire snared from below to provide traction). Advanced techniques have achieved retrieval success rates > 95% even for filters with prolonged dwell times (> 1 year). A dedicated IVC filter clinic with tracking systems to ensure timely retrieval is considered best practice.
May-Thurner Syndrome & Iliac Vein Stenting
May-Thurner syndrome (iliac vein compression syndrome) results from compression of the left common iliac vein by the overlying right common iliac artery against the L5 vertebral body. This causes intimal hyperplasia ("spurs"), venous stasis, and predisposes to left iliofemoral DVT. May-Thurner is present anatomically in ~20-30% of the population but is clinically significant in a much smaller percentage. It should be suspected in young women presenting with left-sided iliofemoral DVT without an obvious provoking factor.
Treatment: catheter-directed thrombolysis or pharmacomechanical thrombectomy (for acute DVT) followed by iliac vein stenting (typically large-diameter self-expanding stents, 14-16 mm, e.g., Wallstent, Venovo, ABRE). Intravascular ultrasound (IVUS) is essential for accurate assessment of venous stenosis — venography significantly underestimates the degree of compression because the vein appears to "fill" around the compression when contrast is injected under pressure. IVUS criteria for significant stenosis: > 50% cross-sectional area reduction compared to a normal adjacent reference segment. Dedicated venous stents (Venovo, ABRE, Zilver Vena) are designed with higher radial force and larger diameters than arterial stents to resist external compression. Stenting is also indicated for chronic post-thrombotic iliac vein stenosis causing symptomatic PTS (Villalta score ≥ 10). Post-stenting anticoagulation: typically 3-6 months of anticoagulation (longer if there is a hypercoagulable state).
08 Dialysis Access Interventions
Fistulogram & Diagnostic Assessment
A fistulogram is the standard angiographic evaluation of a dysfunctional arteriovenous fistula (AVF) or arteriovenous graft (AVG). Indications: elevated venous pressures during dialysis, decreased flow rates, inadequate clearance, prolonged bleeding after needle removal, arm edema (suggesting central venous stenosis), or failure to mature. Technique: puncture the AVF/AVG with a needle or micropuncture set; perform angiography of the entire circuit — arterial anastomosis, body of the fistula/graft, draining veins, central veins (subclavian, brachiocephalic, SVC). A hemodynamically significant stenosis is defined as > 50% diameter narrowing with associated clinical or physiological abnormality (NKF-KDOQI guidelines).
Angioplasty of AVF/AVG Stenosis
The most common cause of AVF/AVG dysfunction is venous outflow stenosis (typically at the venous anastomosis for AVG, or at the cephalic arch/swing point for AVF). Treatment: high-pressure balloon angioplasty (using non-compliant balloons inflated to 20-30 atm, e.g., Conquest, Dorado). Balloon diameter should match the reference vessel. If stenosis is refractory to angioplasty (elastic recoil), options include cutting balloon, drug-coated balloon, or stent graft placement. Cephalic arch stenosis is particularly challenging with high recurrence rates — stent grafts (Flair, Viabahn) have shown improved patency over PTA alone.
Thrombectomy of Clotted Access
Thrombosed AVG: Can usually be declotted with a high success rate (> 90%) using one of several techniques: pharmacomechanical thrombectomy (inject 2-5 mg tPA into the graft, dwell for 15-30 minutes, then balloon maceration of the clot using "sweeping" angioplasty + aspiration of thrombus debris through a sheath) or mechanical thrombectomy (AngioJet rheolytic thrombectomy, Arrow-Trerotola percutaneous thrombolytic device — a rotating basket that macerates clot). Key technical points: (1) always access the graft in both the arterial and venous limbs to treat the entire circuit; (2) retrieve or macerate the "arterial plug" (organized thrombus at the arterial anastomosis that is resistant to lysis); (3) always identify and treat the underlying stenosis after declotting — the graft thrombosed for a reason, and without treating the causative stenosis, rethrombosis is rapid.
Thrombosed AVF: More challenging than AVG declotting because AVF thrombus tends to be older, more organized, and extends over a longer length. May require prolonged tPA infusion (0.5-1 mg/hr for 6-24 hours) or aggressive pharmacomechanical approaches. The pulse-spray technique (forceful injection of tPA through a multi-sidehole catheter laced through the thrombus, creating a "Swiss cheese" pattern that increases lytic surface area) can accelerate lysis. Always evaluate for juxta-anastomotic stenosis (the most common cause of AVF thrombosis — stenosis at or near the arterial anastomosis) and treat with angioplasty. For mature AVFs that thrombose, outcomes of percutaneous declotting are better than for immature or newly thrombosed AVFs.
Central Venous Stenosis

Central venous stenosis (subclavian, brachiocephalic, or SVC) occurs in patients with a history of central venous catheters (up to 50% of patients with prior subclavian dialysis catheters develop stenosis) and is a significant cause of dialysis access dysfunction and arm edema. Treatment: angioplasty (first-line), with stenting reserved for elastic recoil or rapid recurrence (< 3 months). Wallstent (flexible, self-expanding) is commonly used for central venous stenosis. Avoid subclavian vein catheter placement on the ipsilateral side of a functioning AVF/AVG to minimize the risk of central stenosis — this is a key principle of the NKF-KDOQI vascular access guidelines.
AVF Maturation Failure
The "rule of 6s" defines a mature AVF: vein diameter ≥ 6 mm, flow rate ≥ 600 mL/min, depth ≤ 6 mm from the skin surface, and assessed at ≥ 6 weeks after creation. If an AVF fails to mature, duplex US identifies the problem: juxta-anastomotic stenosis (most common), accessory veins (stealing flow from the main outflow), central venous stenosis, or inadequate arterial inflow. IR treatment: angioplasty of the stenosis, coil embolization of accessory veins, and re-evaluation at 4-6 weeks. Early intervention for AVF maturation failure has improved fistula utilization rates significantly.
09 Aortic Interventions (EVAR & TEVAR)
Endovascular Aneurysm Repair (EVAR)
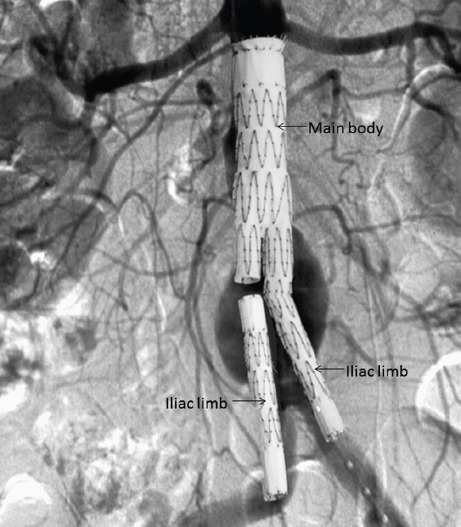
EVAR is the endovascular treatment for infrarenal abdominal aortic aneurysms (AAA). Indications for AAA repair: diameter ≥ 5.5 cm in men or ≥ 5.0 cm in women (SVS guidelines), rapid growth (> 0.5 cm in 6 months or > 1 cm/year), or symptomatic aneurysm (pain, tenderness, evidence of impending rupture). A modular bifurcated stent graft is deployed from bilateral CFA access (or iliac conduit if access vessels are too small/tortuous) to exclude the aneurysm sac from systemic pressure.
Anatomic requirements for standard EVAR: infrarenal neck ≥ 15 mm in length (10 mm with suprarenal fixation devices), ≤ 60 degrees angulation (≤ 45 degrees for some devices), diameter 18-32 mm (device-dependent), absence of significant thrombus or calcification at the seal zone, and absence of conical neck morphology. Iliac access vessels must accommodate 14-20 Fr delivery systems and have adequate diameter without excessive tortuosity. Pre-operative planning: thin-cut CTA (≤ 1 mm slices) with 3D reconstruction for accurate measurements. Centerline measurements (using workstations like TeraRecon, 3mensio, or Horos) are essential — axial measurements overestimate diameter in angulated anatomy.
Major device families: Gore Excluder (ePTFE fabric, low profile — 12-16 Fr delivery system, modular bifurcated design), Medtronic Endurant II / Aorfix (polyester fabric, suture-line fixation; Aorfix designed for highly angulated necks up to 90 degrees), Cook Zenith (suprarenal bare-stent fixation with barbs — provides additional fixation in hostile necks), and Medtronic AFX2 (unibody design — eliminates the risk of modular disconnection but offers less flexibility in sizing). Newer low-profile devices (e.g., Ovation Alto) use a polymer-filled sealing ring that conforms to irregular aortic necks.
Landmark trials: EVAR-1 (2004, PMID: 15157573) and DREAM (2004) showed lower 30-day mortality for EVAR vs open repair (~1.5% vs ~4.5%), but long-term survival was similar (EVAR-1 at 15 years showed no survival advantage for EVAR). OVER trial (VA population, PMID: 22989717) showed early survival advantage for EVAR that diminished by 3 years and was absent by 9 years. EVAR-2 (unfit patients) showed no survival benefit of EVAR over conservative management. These trials emphasize that EVAR requires lifelong imaging surveillance and reintervention (~20-30% reintervention rate at 10 years), whereas open repair is a durable, definitive treatment. Current practice: EVAR is the preferred approach for anatomically suitable patients with reasonable life expectancy; open repair is reserved for patients with hostile anatomy for EVAR, young patients (where long-term durability matters most), and those who cannot comply with imaging surveillance.
Endoleak Classification
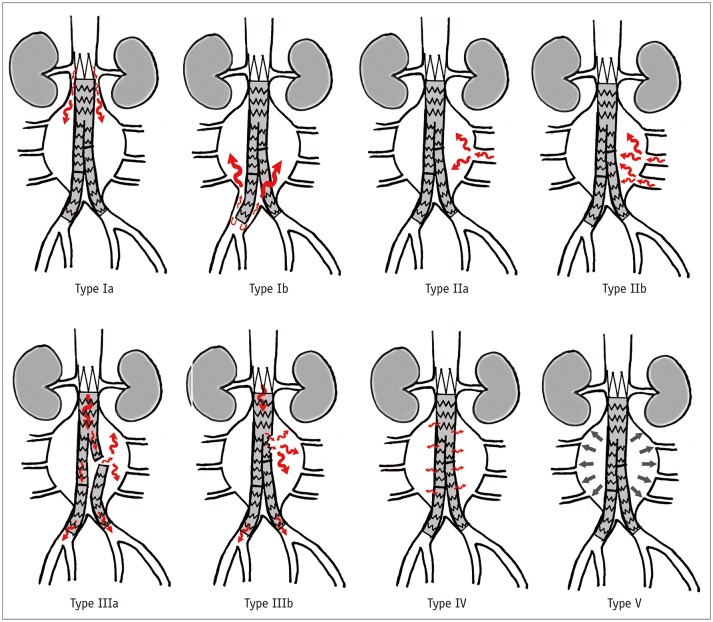
Type I — Attachment site leak: Ia (proximal seal zone), Ib (distal seal zone). High pressure, requires urgent treatment (extension cuff, relining, conversion to open). Risk of sac expansion and rupture.
Type II — Branch vessel backflow: Most common endoleak (~20-30%). Retrograde flow from lumbar arteries or the IMA into the sac. Usually benign. Treatment only if sac expansion > 5 mm (transarterial or translumbar embolization with coils/glue).
Type III — Graft defect: IIIa (modular disconnection between components), IIIb (fabric tear/hole). High pressure, requires relining or extension.
Type IV — Graft porosity: Contrast blush through intact graft fabric. Seen only on completion angiogram; self-limited.
Type V — Endotension: Sac expansion without identifiable endoleak on imaging. Controversial entity. May represent undetected low-flow endoleak.
Thoracic Endovascular Aortic Repair (TEVAR)
TEVAR is indicated for descending thoracic aortic aneurysms, acute type B dissections (complicated), traumatic aortic transection, penetrating aortic ulcers, and intramural hematoma. Landing zone classification (Ishimaru zones): Zone 0 — ascending aorta (requires arch debranching or total arch replacement); Zone 1 — between innominate and left carotid (requires left carotid bypass/chimney); Zone 2 — between left carotid and left subclavian (may require left subclavian revascularization); Zone 3 — proximal descending aorta (standard TEVAR landing zone); Zone 4 — mid-descending aorta. Coverage of the left subclavian artery without revascularization increases the risk of left arm ischemia, stroke (dominant left vertebral artery), and spinal cord ischemia.
For the treatment of complicated acute type B aortic dissection, TEVAR covers the primary entry tear and promotes thrombosis of the false lumen. The INSTEAD-XL trial showed long-term benefit of TEVAR for uncomplicated type B dissection at 5 years (PMID: 23425526). Spinal cord ischemia (paraplegia) is the most feared complication of TEVAR, occurring in 2-8% of cases. Risk factors: long segment coverage (> 20 cm), prior or concurrent infrarenal aortic repair, hypotension, and coverage of the left subclavian artery. Prevention: CSF drainage (target CSF pressure < 10 mmHg), maintain MAP > 80 mmHg, staged procedures when possible.
Fenestrated & Branched Endografts (F/BEVAR)
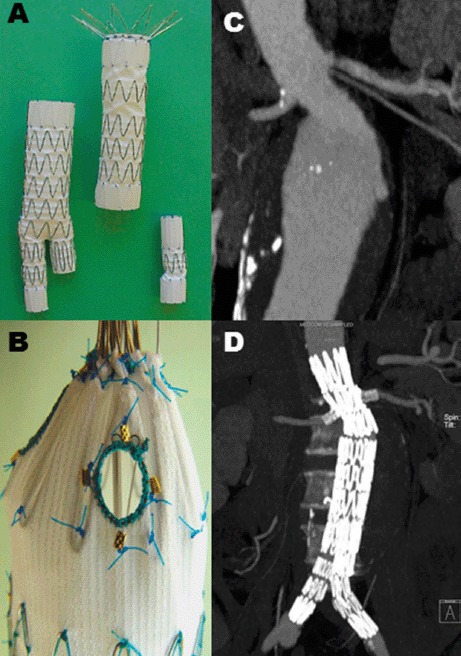
Fenestrated EVAR (FEVAR) extends endovascular repair to juxtarenal and pararenal AAAs by incorporating fenestrations (holes) or scallops in the stent graft fabric that align with the renal and visceral artery ostia, which are then stented to maintain perfusion. Branched EVAR (BEVAR) uses directional side branches for visceral vessels. These are custom-manufactured devices requiring 6-8 weeks of lead time. The Zenith Fenestrated (Cook) is FDA-approved. Physician-modified endografts (PMEGs) are created on-table for urgent/emergent cases. Outcomes: 30-day mortality ~2-3%, target vessel patency > 90% at 3 years.
10 Hemorrhage Control & Embolization
Principles of Embolization
Transcatheter embolization is the deliberate occlusion of a blood vessel to stop hemorrhage, devascularize a tumor, or eliminate a vascular malformation. Catheter position determines the level of occlusion: proximal embolization (occludes the main feeding artery — risk of collateral reconstitution), distal/superselective embolization (occludes at or near the bleeding point — preferred to preserve collateral flow and minimize ischemia), or sandwich technique (embolization proximal and distal to the bleeding point to prevent backflow via collaterals, e.g., GDA embolization for duodenal ulcer bleed).
Embolization Agents
Metallic coils: Detachable or pushable; sizes from 2-20 mm. Provide mechanical occlusion with subsequent thrombosis. Controlled deployment. Used for large-vessel occlusion, pseudoaneurysms, and selective organ devascularization. Platinum (MRI-compatible) or stainless steel. Fibered coils promote faster thrombosis.
Gelatin sponge (Gelfoam): Temporary embolic agent (resorbs in 2-6 weeks). Hand-cut into pledgets or made into a "torpedo" (slurry). Used for trauma embolization where temporary occlusion is desired (allows vessel recanalization). Also used for uterine fibroid embolization adjunct and GI bleeding.
Polyvinyl alcohol (PVA) particles: Permanent, irregularly shaped particles (45-1000 μm). Cause small-vessel occlusion. Used for tumor devascularization, UAE, and GI bleeding. Non-resorbable.
Calibrated microspheres (Embosphere, BeadBlock): Spherical, calibrated particles (40-1200 μm). Provide more predictable and uniform embolization compared to PVA. Can be loaded with chemotherapy drugs (drug-eluting beads for DEB-TACE).
N-butyl cyanoacrylate (NBCA / "glue"): Liquid embolic that polymerizes on contact with ionic media (blood). Mixed with ethiodized oil (Lipiodol) to control polymerization time (more oil = slower). Permanent. Used for high-flow AVMs, emergent hemorrhage, and varicocele embolization. Requires experience to avoid catheter entrapment.
Ethylene vinyl alcohol copolymer (Onyx): Liquid embolic dissolved in DMSO. Precipitates on contact with blood; forms a spongy, non-adhesive cast. Allows slow, controlled injection. Used for AVMs and dural AVFs. The DMSO solvent is toxic to vessel walls and requires a DMSO-compatible microcatheter.
Amplatzer Vascular Plug (AVP): Self-expanding nitinol mesh device for controlled occlusion of large vessels (5-22 mm). Deployed through a catheter. Used for internal iliac artery embolization before EVAR, pulmonary AVM embolization, and portal vein embolization.
Trauma Embolization
Splenic embolization: Indicated for blunt splenic injury (AAST grade III-V) in hemodynamically stable patients. Two approaches: proximal splenic artery embolization (coils placed in the main splenic artery to reduce perfusion pressure while maintaining splenic viability via short gastric and pancreatic collaterals — the spleen survives because it receives enough blood through collateral pathways to prevent infarction but at lower pressure, allowing hemostasis) vs distal superselective embolization (coils or gelfoam placed directly at the site of vascular injury for focal bleeding, pseudoaneurysm, or active extravasation seen on angiography). Proximal embolization has a higher splenic salvage rate and is the more commonly used technique. Some operators perform combined proximal and distal embolization for high-grade injuries. Post-embolization: vaccinate for encapsulated organisms (Haemophilus influenzae type b, Streptococcus pneumoniae, and Neisseria meningitidis) if there is significant splenic infarction or expected functional asplenia — ideally before discharge or within 14 days.
Hepatic embolization: For hepatic arterial bleeding (trauma, post-surgical, iatrogenic post-biopsy/post-biliary procedure, or tumor rupture — notably spontaneous rupture of HCC, which occurs in ~3-15% of HCC patients and carries a high mortality). The liver's dual blood supply (hepatic artery ~25% of blood flow but ~50% of oxygen delivery, portal vein ~75% of blood flow) allows aggressive arterial embolization with a low risk of hepatic infarction as long as portal vein flow is intact. In patients with portal vein thrombosis, hepatic artery embolization carries a significantly higher risk of hepatic infarction and should be performed as selectively as possible. Superselective embolization with coils, Gelfoam, or a combination is preferred to preserve hepatic parenchyma. For post-biopsy or post-ablation hemorrhage, the bleeding vessel is typically a small peripheral hepatic artery branch — coil embolization proximal and distal to the injury is definitive. The liver's regenerative capacity means that even extensive embolization is usually tolerated in patients with normal hepatic function.
Pelvic embolization: For pelvic fracture-related hemorrhage. The pelvic vasculature is supplied by branches of both internal iliac arteries, with extensive collateral networks that make definitive hemorrhage control challenging. In hemodynamically unstable patients with confirmed pelvic arterial bleeding (active extravasation on CTA or clinical suspicion after pelvic fracture), angiography and embolization should proceed emergently — delay for additional imaging is not warranted. Technique: bilateral internal iliac artery embolization with Gelfoam (temporary — allows vessel recanalization in 2-6 weeks, important for preserving pelvic blood supply). Non-selective embolization of both internal iliac arteries may be performed rapidly in unstable patients using a "damage control" approach; superselective embolization is preferred in stable patients to minimize non-target ischemia (bladder necrosis, buttock claudication, sexual dysfunction).
The decision to embolize is based on clinical context: pelvic angiography is indicated after initial resuscitative measures (massive transfusion, pelvic binder/external fixation, REBOA if available) if hemodynamic instability persists and other bleeding sources have been excluded (FAST exam negative for intraperitoneal hemorrhage) or addressed. The WSES (World Society of Emergency Surgery) guidelines recommend an integrated approach: hemodynamically unstable patients with pelvic fracture and negative FAST should proceed directly to angioembolization; those with positive FAST may require laparotomy first with subsequent pelvic angiography if hemodynamic instability persists. Hybrid operating rooms that combine surgical and IR capabilities allow simultaneous or sequential surgical and endovascular treatment.
Postpartum hemorrhage (PPH) embolization: For severe PPH refractory to medical management (uterotonics — oxytocin, methylergonovine, carboprost, misoprostol) and intrauterine balloon tamponade. Typically bilateral uterine artery embolization with Gelfoam pledgets or slurry. Success rate > 90% for PPH from uterine atony. Advantages over hysterectomy: preserves the uterus and future fertility. The uterine arteries are catheterized from a single CFA approach using a Waltman loop or Roberts catheter. Bilateral internal iliac artery embolization may be performed if uterine artery catheterization is technically difficult. Future pregnancies are possible after uterine artery embolization for PPH, though these patients should be monitored as high-risk.
GI Hemorrhage Embolization
Upper GI bleeding: Angiography is indicated when endoscopy fails to control hemorrhage (typically after two failed endoscopic attempts) or when bleeding is too brisk for endoscopic visualization. Active extravasation on angiography requires a bleeding rate of ~0.5-1.0 mL/min for detection by conventional angiography. CT angiography can detect as little as 0.3 mL/min and should be performed before angiography when feasible (it localizes the bleeding source and guides selective catheterization, reducing angiographic time and contrast volume). A negative CTA has a very high negative predictive value for active arterial bleeding.
Treatment: superselective embolization with coils, Gelfoam, or a combination. The rich collateral blood supply of the stomach and duodenum (from celiac and SMA territories via the pancreaticoduodenal arcades, left and right gastroepiploic arteries, and short gastric arteries) allows aggressive embolization with low ischemia risk. The classic teaching is to embolize the GDA both proximal and distal to the bleeding point ("sandwich technique") using coils to prevent backflow through the rich pancreaticoduodenal collateral network. If the bleeding artery is clearly identified, coil embolization immediately proximal and distal to the pseudoaneurysm or bleeding point is definitive. For diffuse mucosal bleeding without a discrete arterial source, Gelfoam embolization of the left gastric artery territory may temporize gastric hemorrhage while allowing vascular recanalization.
Lower GI bleeding: Typically from the SMA (right colon, ileocolic, middle colic) or IMA (left colon, sigmoid, superior rectal) territory. Lower GI bleeding is often intermittent, making localization challenging. CT angiography should be the first-line imaging study — it has sensitivity of ~85-90% for active arterial extravasation and provides a roadmap for subsequent angiography. If CTA shows active extravasation, proceed directly to selective mesenteric angiography of the bleeding territory. If CTA is negative, options include: observation with repeat CTA if rebleeding occurs, tagged RBC scan (nuclear medicine — can detect bleeding as slow as 0.1 mL/min over several hours of imaging, but with limited spatial localization), or provocative angiography (controversial).
Mesenteric angiography with provocative maneuvers (intra-arterial vasodilators such as tolazoline or nitroglycerin, or intra-arterial anticoagulants such as heparin 5,000 units) may be used to unmask intermittent bleeding that is not active at the time of angiography — this technique increases diagnostic yield but carries risk of uncontrolled hemorrhage and is used selectively. Empiric embolization of a suspected bleeding source identified on CTA but not confirmed on angiography is controversial due to the higher risk of bowel ischemia in the colon. The colon has a less robust collateral arterial network than the upper GI tract — the "watershed" areas at the splenic flexure (Griffiths point — junction of SMA and IMA territories via the marginal artery of Drummond) and rectosigmoid (Sudeck point — junction of the last sigmoidal branch and the superior rectal artery) are particularly vulnerable to ischemia after embolization. Superselective embolization with microcoils at the bleeding vasa recta (the straight arteries supplying the bowel wall) minimizes ischemia risk by preserving the marginal artery and other collateral pathways. Ischemic bowel complication rate after superselective colonic embolization: ~5-10%.
Uterine Artery Embolization (UAE)
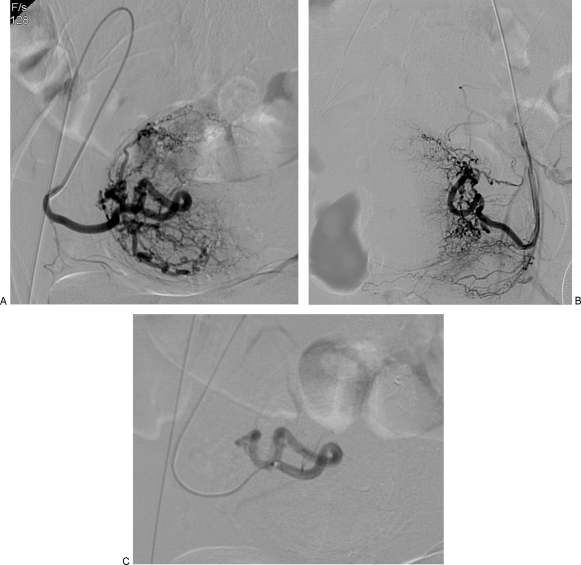
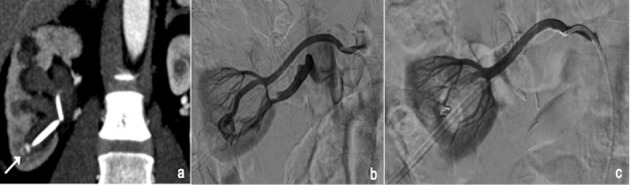
UAE is a minimally invasive treatment for symptomatic uterine fibroids (menorrhagia, bulk symptoms, pelvic pain). Both uterine arteries are catheterized (typically from a single CFA access using a Waltman loop or Roberts uterine catheter) and embolized with calibrated microspheres (500-700 μm or 700-900 μm). The embolization endpoint is near-stasis or "pruned tree" appearance of the uterine artery. Post-embolization syndrome (pain, nausea, low-grade fever) is expected and managed with IV PCA, NSAIDs, and antiemetics for 24-48 hours. Most patients are discharged within 23 hours. The REST trial and EMMY trial showed comparable symptom improvement between UAE and surgery (hysterectomy/myomectomy), with faster recovery for UAE. Complications: fibroid expulsion (2-5%), amenorrhea (~3%, higher in women > 45), endometritis, and rarely ovarian failure. UAE is a relative contraindication in women actively pursuing pregnancy, though successful pregnancies have been reported post-UAE.
Bronchial Artery Embolization
BAE is the first-line treatment for massive hemoptysis (> 300 mL/24 hours or life-threatening). Bronchial arteries typically arise from the descending thoracic aorta at the T5-T6 level. The normal configuration is two left and one right bronchial artery, but there is significant anatomic variability. Non-bronchial systemic arteries may also supply the lungs (subclavian, internal mammary, inferior phrenic, intercostal, thyrocervical) — these must be identified and embolized if contributing to hemorrhage. The most dangerous variant is a bronchial artery that gives off the anterior spinal artery (artery of Adamkiewicz) — embolization risks spinal cord infarction and paraplegia. Distal embolization with PVA particles (300-500 μm) is preferred; avoid proximal coil embolization alone (prevents future re-access if hemoptysis recurs, which occurs in 10-20% of patients). Angiographic identification of the spinal artery is mandatory before embolization. Common etiologies: bronchiectasis (most common worldwide), tuberculosis (most common in endemic areas), lung cancer, aspergilloma, and cystic fibrosis.
Varicocele Embolization
Varicocele embolization is an alternative to surgical ligation for symptomatic varicoceles or varicoceles associated with infertility. Approach: right IJV or right CFA access; catheterize the left renal vein (or left internal spermatic/gonadal vein), advance a microcatheter into the pampiniform plexus, and embolize with coils, sclerosant (sodium tetradecyl sulfate), or NBCA from distal to proximal. Right-sided varicoceles require catheterization of the right gonadal vein directly from the IVC. Technical success rate > 95%; clinical success (symptom improvement or semen parameter improvement) ~80-90%. Recurrence rate: ~5-10%, comparable to surgical ligation.
11 Venous Access — PICC, CVC & Ports
Peripherally Inserted Central Catheters (PICC)
A PICC is a central venous catheter inserted through a peripheral arm vein (basilic or brachial vein preferred — the basilic vein is larger and has a straighter course to the central veins than the cephalic vein, which has a sharp angulation at the deltopectoral groove that frequently causes difficulty with advancement). The cephalic vein should be used only as a last resort for PICC placement due to its tortuosity and the ~90-degree turn at the cephalic arch where it joins the axillary vein — PICCs placed via the cephalic vein have higher malposition and thrombosis rates.
Tip position: the caval-atrial junction (lower third of the SVC or upper RA) is the optimal tip location — this position minimizes the risk of thrombosis (higher flow rates dilute the infusate and reduce endothelial injury) and catheter malfunction. Tip confirmation: fluoroscopy (gold standard), chest X-ray (most common), or ECG-based tip confirmation systems (e.g., Sherlock 3CG — detects the P-wave amplitude increase as the catheter approaches the SA node, allowing bedside placement without fluoroscopy). Inserted under ultrasound guidance using modified Seldinger (micropuncture) technique. Available as single, double, or triple lumen; 3-6 Fr. Power-injectable PICCs (rated for power injector flow rates of 2-5 mL/s) are marked with a purple hub or specific product identifiers.
Indications: long-term IV antibiotics (> 7 days), TPN (requires dedicated lumen), chemotherapy (power-injectable PICC), and frequent blood draws. Complications: upper extremity DVT (~3-5% — risk factors include larger catheter diameter, triple lumen, ICU admission, malignancy, and catheter placed in the smaller basilic vein where the catheter-to-vein ratio is unfavorable), line-related bloodstream infection (CLABSI ~0.4-2.0 per 1,000 catheter-days), tip malposition (most commonly into the internal jugular vein or contralateral brachiocephalic vein), and fibrin sheath formation (~30-50% of PICCs develop a fibrin sheath within 1-2 weeks; presents as inability to withdraw blood despite easy flushing; treated with intraluminal tPA instillation — CathFlo Activase 2 mg in 2 mL, dwell 30-120 minutes, or percutaneous fibrin sheath stripping via a femoral venous approach). The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC) provides evidence-based criteria for selecting the most appropriate vascular access device based on therapy duration, nature of infusate, and patient factors.
Tunneled Central Venous Catheters
Tunneled CVCs have a subcutaneous tunnel (typically 5-10 cm) between the venous entry site (IJV) and the skin exit site (anterior chest wall), with a Dacron cuff positioned in the tunnel approximately 2 cm from the skin exit. The cuff promotes tissue ingrowth over 2-3 weeks, which anchors the catheter (preventing dislodgement) and provides a barrier against ascending bacterial migration (reducing infection risk compared to non-tunneled catheters). Tunneled catheters should not be removed until the cuff has been freed from the surrounding tissue — forceful extraction without cuff dissection can cause subcutaneous tissue injury.
Types: Hickman catheter (large bore, 9.6-12.5 Fr, single/double/triple lumen; used for bone marrow transplant, apheresis, long-term infusion); Broviac catheter (smaller caliber, 2.7-6.6 Fr, used in pediatrics); tunneled dialysis catheter (dual lumen, 14.5 Fr, step-tip or split-tip design — step-tip catheters have the arterial lumen opening more proximal than the venous lumen to prevent recirculation; split-tip catheters have two separate limbs that splay in the atrium to maximize flow and minimize recirculation). Dialysis catheter tip should be positioned in the mid-to-lower RA (for right IJV access) or at the caval-atrial junction to optimize flow rates (≥ 350 mL/min during dialysis). Preferred access: right IJV (shortest, straightest path to the SVC-RA junction, lowest malposition rate, lowest central stenosis rate compared to subclavian access). Hierarchy of access preference (NKF-KDOQI): right IJV > left IJV > right EJV > left EJV > right subclavian > left subclavian > femoral (last resort). Placement under ultrasound guidance for venous puncture and fluoroscopy for wire/catheter advancement and tip positioning.
Implantable Ports
A port (port-a-cath) is a totally implantable venous access device consisting of a titanium or plastic reservoir implanted in a subcutaneous pocket (typically on the anterior chest wall, over the pectoralis muscle) connected to a catheter in the SVC. The port is accessed by puncturing the port septum with a non-coring (Huber) needle — the septum is rated for 1,000-2,000 punctures. Advantages: lowest infection rate of all central venous devices (~0.1 infections per 1,000 catheter-days), cosmetically acceptable (completely under the skin when not in use), allows swimming/bathing, and requires flushing only every 4-6 weeks when not in use. Used for intermittent chemotherapy, long-term intermittent IV access, and patients who prefer minimal external hardware. Power-injectable ports (e.g., PowerPort, identified by palpable bumps on the reservoir or CT identification of port markings) are rated for CT contrast injection at power injector flow rates (up to 5 mL/s). Non-power-injectable ports may rupture under power injection pressures.
Port placement technique: IJV or subclavian vein access under US guidance; catheter advanced to the caval-atrial junction under fluoroscopy; subcutaneous pocket created on the chest wall (2-3 cm below the clavicle, lateral to the mid-clavicular line); catheter tunneled from the venous entry site to the pocket; port connected to the catheter and secured in the pocket with sutures. Complications: catheter-related infection (port removal often required for infection or bacteremia that persists despite 48-72 hours of systemic antibiotics), catheter fracture (pinch-off syndrome — catheter compressed between the clavicle and first rib, risk with subclavian access; diagnosed by narrowing of the catheter on CXR), port flipping in the pocket (requires revision), and fibrin sheath formation (treated with intraluminal tPA instillation or percutaneous stripping).
Difficult Venous Access Strategies
When standard IJV, subclavian, and upper extremity venous access is exhausted (bilateral central venous occlusion), alternative access options include: translumbar IVC access (direct puncture of the IVC via a posterior approach), transhepatic IVC access (through the hepatic veins), femoral vein tunneled catheter (higher infection and thrombosis rates), and recanalization of occluded central veins (sharp recanalization with a needle through a chronic occlusion, supported by stenting). Fibrin sheath stripping: performed by snaring the catheter tip from a femoral venous approach and stripping the fibrin sheath by pulling a snare along the catheter length — restores catheter function in ~90% of cases.
12 Image-Guided Biopsy
Principles
Image-guided percutaneous biopsy provides tissue diagnosis while minimizing surgical morbidity. The choice of imaging modality depends on target location and visibility: ultrasound for superficial structures (liver, kidney, thyroid, lymph nodes — advantages: real-time visualization, no radiation, portable), CT for deep or small targets (lung, retroperitoneal, bone, deep lymph nodes — advantages: excellent spatial resolution, visualization of adjacent critical structures), and MRI for lesions visible only on MRI (some prostate lesions, breast lesions, musculoskeletal tumors — requires MRI-compatible needles and real-time MR sequences). Fusion imaging (combining real-time US with a previously acquired CT or MRI dataset) is increasingly used for lesions visible on CT/MRI but not on US, allowing US-guided biopsy of otherwise CT-only targets.
The coaxial technique (introducing a larger outer needle — typically 17-19 gauge — to the margin of the lesion, through which multiple passes are made with a smaller inner biopsy needle — 18-20 gauge) reduces the number of pleural/capsular punctures and minimizes complications. Advantages of coaxial technique: (1) multiple samples through a single access, (2) ability to perform a blood patch or gelatin sponge tract embolization through the outer needle upon withdrawal (reducing pneumothorax in lung biopsy), and (3) reduced risk of tumor seeding along the needle tract (the outer needle shields the tract). For potentially resectable tumors (HCC, cholangiocarcinoma), the biopsy tract should be planned to be within the future surgical resection field to allow en bloc removal of the tract.
Needle Types
Fine-needle aspiration (FNA): 20-25 gauge needles. Aspirates individual cells for cytologic analysis. Higher sensitivity for carcinoma, lower for lymphoma (which requires tissue architecture). Core needle biopsy (CNB): 14-20 gauge spring-loaded cutting needles (e.g., Tru-Cut, Temno). Obtains a tissue core preserving histologic architecture. Essential for lymphoma, sarcoma, and molecular/genetic analysis. In practice, most IR biopsies use core needles, often supplemented by FNA for rapid on-site evaluation (ROSE) by cytopathology.
Organ-Specific Considerations
Liver biopsy: Diffuse disease (cirrhosis staging) — random biopsy via subcostal or transjugular approach (transjugular preferred when coagulopathy or ascites is present, as the biopsy track is within the hepatic vein, reducing bleeding risk). Focal lesions — CT or US-guided core biopsy. Major complication rate: ~1% (hemorrhage, bile peritonitis). In patients with suspected HCC, biopsy is often unnecessary when imaging meets LI-RADS 5 criteria (contrast-enhanced CT or MRI showing arterial hyperenhancement and washout).
Lung biopsy: CT-guided, typically with a coaxial system. Pneumothorax rate: 15-25% (chest tube required in ~5-7%). Risk factors for pneumothorax: small lesion size, deep location, crossing fissures, COPD/emphysema, multiple needle passes. Post-procedure management: upright chest X-ray at 2-4 hours; small (< 2 cm), stable pneumothoraces without symptoms can be observed.
Kidney biopsy: US-guided (native kidneys — lower pole; transplant kidneys — upper or lower pole). For medical renal disease (glomerulonephritis), 16-18 gauge automated core needle with at least two passes for adequate glomerular sampling (≥ 10 glomeruli for light microscopy, tissue for immunofluorescence and electron microscopy). Hemorrhage risk: ~1-2% requiring intervention.
Bone biopsy: CT-guided with a bone biopsy needle system (Bonopty, OnControl powered system). Cortical bone requires a trephine or drill-powered needle. Always biopsy the soft tissue component of a bone lesion when present (more representative tissue and easier access). For suspected primary bone tumors, coordinate with orthopedic oncology to plan the biopsy tract within the anticipated surgical resection field — the biopsy tract will be resected en bloc with the tumor during definitive surgery.
Lymph node biopsy: Performed under US guidance for accessible nodes (cervical, axillary, inguinal) and CT guidance for deep nodes (retroperitoneal, mesenteric, pelvic). Core biopsy is essential for lymphoma diagnosis (FNA alone is insufficient because tissue architecture is required for classification). For suspected lymphoma, fresh tissue should be sent in saline (not formalin) for flow cytometry in addition to standard histologic analysis.
Adrenal biopsy: Performed under CT guidance for indeterminate adrenal masses when imaging characteristics are insufficient for characterization and the result would change management. Important: rule out pheochromocytoma before biopsy (24-hour urine metanephrines or plasma free metanephrines) — biopsy of an unsuspected pheochromocytoma can trigger a hypertensive crisis. The right adrenal is approached posteriorly (trans-hepatic approach is also used); the left adrenal is approached from the left posterolateral direction, avoiding the spleen, pancreatic tail, and aorta.
13 Abscess & Fluid Drainage
Principles of Percutaneous Drainage
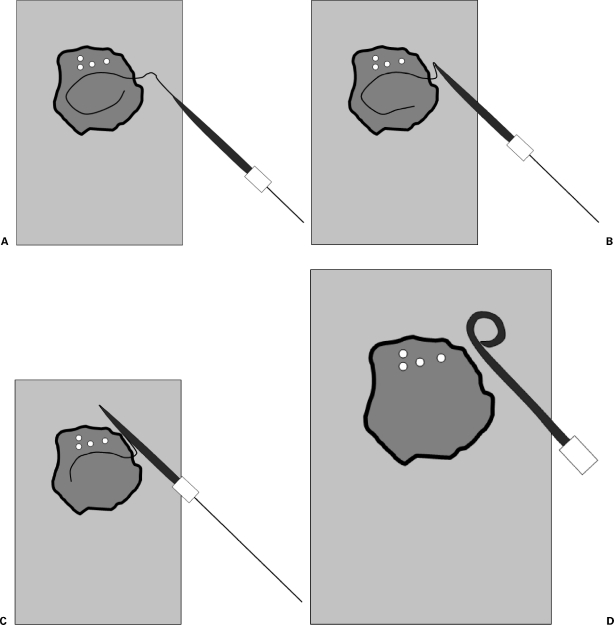
Image-guided percutaneous drainage is first-line treatment for most abdominal, pelvic, and thoracic fluid collections. Success rates exceed 80-90% for simple, unilocular abscesses. Indications: infected fluid collection (abscess), symptomatic non-infected collection (seroma, lymphocele, biloma, urinoma), and biliary obstruction. The decision to drain versus aspirate depends on the collection: simple abscesses < 3-4 cm and non-infected collections may be adequately treated with needle aspiration alone; larger, complex, or persistent collections require catheter drainage.
Drainage can be performed by the Seldinger technique (needle → wire → serial dilation → catheter; preferred for deep collections where precise needle positioning is critical) or the trocar technique (catheter mounted on a sharp stylet, advanced directly into the collection in a single step — faster but requires a confident target, a safe window, and an adequately large collection to avoid injury to adjacent structures). Route planning: choose the shortest, safest path avoiding bowel, major vessels, and solid organs. Transperitoneal routes are acceptable; transpleural routes should be avoided if possible (risk of empyema). For complex collections with septations, a large-bore catheter with aggressive flushing or fibrinolytic instillation (tPA 4 mg in 20 mL saline, dwell 30-60 minutes) may be needed to break down loculations.
Catheter Selection
Pigtail catheters (8-14 Fr) are standard for most abscess and fluid drainage. The pigtail curl provides self-retention within the collection. Larger catheters (14-16 Fr) are used for viscous fluid (e.g., hematoma, pancreatic necrosis). Sump drains (double-lumen, e.g., van Sonnenberg) have an air vent that prevents suction collapse of the drain against tissue — used for large, persistent collections. Drains are placed to gravity initially; wall suction may be applied for persistent collections.
Drain Management
After placement: document output volume, color, and character daily. Flush drains with 10 mL normal saline q8-12h to maintain patency. Criteria for drain removal: resolution of symptoms (pain, fever), drain output < 10 mL/day, imaging confirms cavity collapse, and resolution of the underlying etiology. Drains can be upsized if output is thick or inadequate despite a persistent collection. If a drain becomes occluded, attempt flushing with tPA (2-4 mg in 10-20 mL saline, dwell for 30-60 minutes) before catheter exchange.
Biliary Drainage
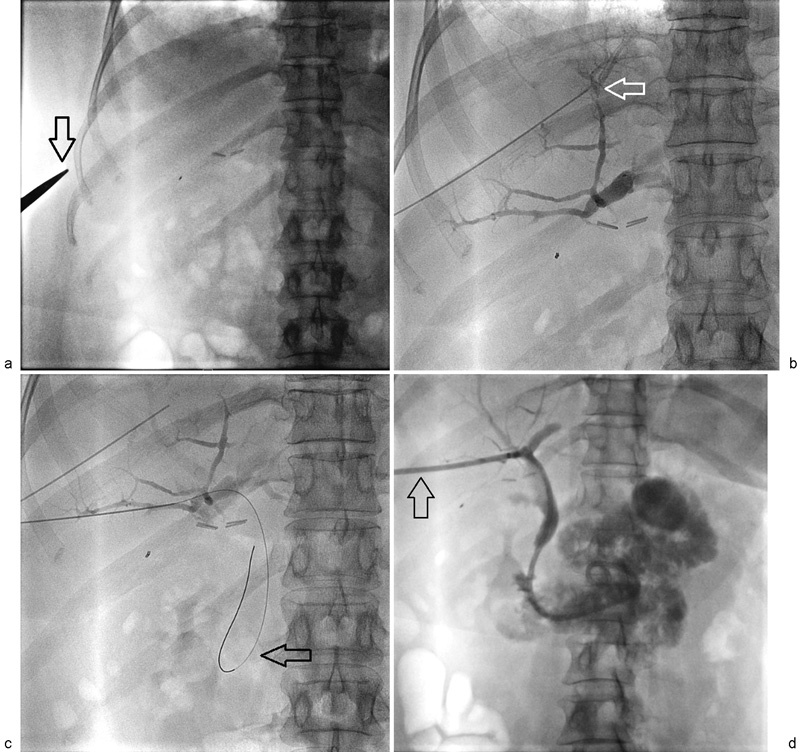
Percutaneous transhepatic cholangiography (PTC) and percutaneous biliary drainage (PBD) are performed when endoscopic retrograde cholangiopancreatography (ERCP) fails or is not feasible (e.g., surgically altered anatomy such as Roux-en-Y gastric bypass or hepaticojejunostomy, duodenal obstruction from pancreatic head mass, or failed ERCP cannulation after two attempts).
Technique: using a 21-gauge Chiba needle, access a peripheral intrahepatic bile duct under fluoroscopic guidance (aiming laterally from a right subcostal approach, targeting ducts at the level of the right portal vein) or US guidance (direct visualization of a dilated duct). Bile is aspirated to confirm intraductal position, and contrast is injected to opacify the biliary tree (cholangiogram). A 0.018" wire is then advanced through the needle and converted to a 0.035" system via coaxial dilators. From here, three drainage options: external drain (catheter tip proximal to the obstruction, all bile drains externally — temporary, used when the obstruction cannot be crossed); external-internal drain (catheter crosses the obstruction with sideholes above and below, allowing bile to drain both internally into the duodenum and externally into a bag — the most common initial configuration); and internal stent (metallic or plastic stent placed across the obstruction for permanent internal drainage).
A right-sided approach (through the right lobe of the liver, typically mid-axillary line) provides better catheter stability due to the liver's supporting parenchyma and a shorter, more direct tract to the common bile duct; this is the standard approach. A left-sided approach (through the left hepatic lobe via an epigastric/subxiphoid access) is used when right-sided access is not possible (right hepatectomy, right hepatic duct disconnected), for isolated left-sided ductal obstruction (Bismuth type III hilar cholangiocarcinoma), or when the patient will be self-managing the drain (anterior chest wall exit site is more accessible than a right lateral site). Complications: hemorrhage (~2-5% — minimize by traversing hepatic parenchyma which tamponades the tract), bile peritonitis (~1-2%), sepsis/cholangitis (~2-5% — always administer prophylactic antibiotics 30-60 minutes before PTC/PBD, typically cefazolin 1 g IV or ciprofloxacin 400 mg IV; do not over-inject contrast into an obstructed system as this introduces bacteria into the bloodstream), and pleural transgression (if the access is too cephalad).
Biliary stenting: After establishing biliary access, a metallic stent (self-expanding, typically 8-10 mm diameter, e.g., Wallstent, Viabil) is deployed across the stricture for permanent internal drainage in malignant obstruction (pancreatic head cancer, cholangiocarcinoma, metastatic hilar nodes). Plastic stents (8-12 Fr) are used for benign strictures as a temporizing measure. Covered metallic stents resist tumor ingrowth but may migrate; uncovered stents embed in the bile duct wall and have a higher rate of tumor ingrowth occlusion.
14 Urinary Interventions
Percutaneous Nephrostomy (PCN)
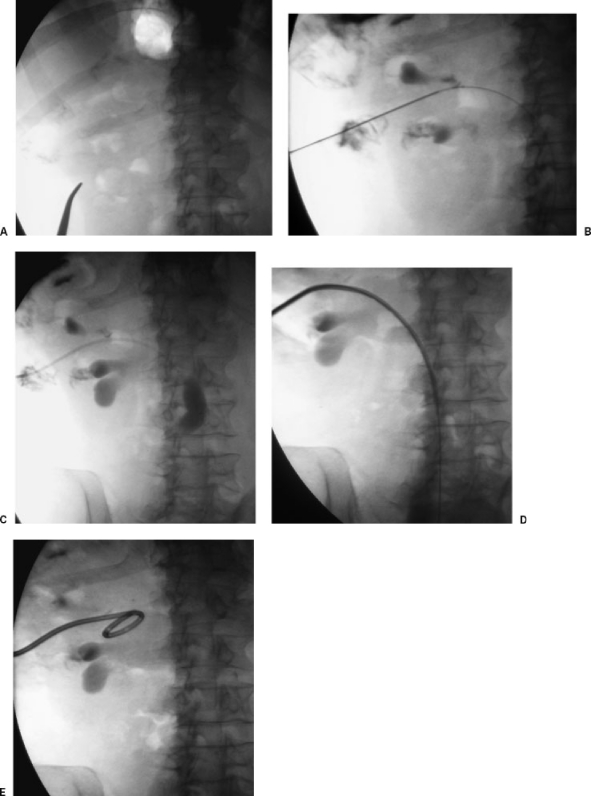
PCN is the placement of a drainage catheter directly into the renal collecting system through a posterior flank approach. Indications: urinary obstruction (ureteral calculi, malignant obstruction) when retrograde stenting is not possible, pyonephrosis (infected obstructed kidney — urologic emergency), urinary diversion prior to fistula repair, and access for percutaneous nephrolithotomy (PCNL). Technique: US or fluoroscopy-guided access into a posterior lower pole calyx (provides the safest, most direct access to the collecting system, avoiding major renal vessels which course anteriorly — Brodel's bloodless line of incision runs along the posterior lateral aspect of the kidney between the anterior and posterior segmental arterial distributions). A 10-12 Fr locking pigtail catheter is standard for drainage.
Anatomic considerations: the kidney has five vascular segments (apical, upper, middle, lower, and posterior) — the posterior segment is supplied by a single artery that courses behind the renal pelvis, and access through the posterior calyx avoids these vessels. The kidney is retroperitoneal, and the posterior approach traverses the paraspinal muscles (erector spinae, quadratus lumborum) but avoids the peritoneal cavity. Access should be below the 12th rib to minimize the risk of pleural transgression (the pleural reflection extends to the level of the 12th rib posteriorly).
Contraindications: uncorrectable coagulopathy (relative). Complications: hemorrhage (~1-4% requiring transfusion), sepsis (always administer prophylactic antibiotics), injury to adjacent organs (colon, pleura), catheter dislodgement. For pyonephrosis, initial drainage should be to gravity only (do not irrigate or inject contrast aggressively, which can cause bacteremia and septic shock).
Antegrade Ureteral Stenting
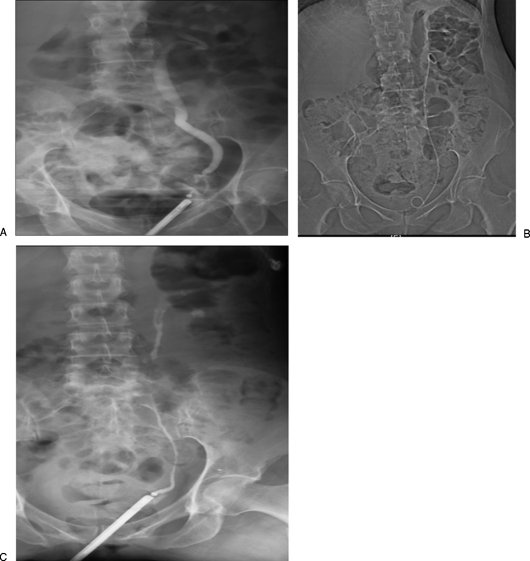
After establishing PCN access, a guidewire can be advanced antegrade through the ureter into the bladder. A loopogram/nephrostogram should be performed first to map the collecting system anatomy and identify the level and cause of obstruction. An internal ureteral stent (double-J stent, 6-8 Fr, typically 22-28 cm length depending on patient height) is then deployed over a stiff wire, with the proximal pigtail in the renal pelvis and the distal pigtail in the bladder. Proper positioning is confirmed fluoroscopically (proximal curl in the renal pelvis, distal curl in the bladder). This provides internal drainage and allows nephrostomy removal after a brief observation period (typically 24-48 hours capped trial).
Antegrade stenting is performed when retrograde (cystoscopic) stenting has failed, typically due to extrinsic malignant ureteral obstruction (cervical cancer, colorectal cancer, retroperitoneal lymphadenopathy), distal ureteral stricture, or urinary diversion anatomy (ileal conduit). Metallic ureteral stents are used for malignant obstruction refractory to standard polymer stents: Resonance stent (Stryker/Cook — tightly coiled alloy stent with no internal lumen, drains through interstices; resists compression by extrinsic tumor), Memokath stent (shape-memory nickel-titanium alloy; thermally expandable). Metallic ureteral stents have longer patency than polymer stents in malignant obstruction (~12 months vs ~3-4 months) and are the preferred option for patients with limited life expectancy who benefit from reduced need for stent exchanges.
Percutaneous Nephrolithotomy Access
The IR role in PCNL is to establish renal access for the urologist. This is a collaborative procedure where the IR provides optimal access and the urologist performs the stone removal. Under fluoroscopic and/or US guidance, a needle is placed into the target calyx (chosen based on stone location and the angle needed for the urologist to reach all stone-bearing calyces — ideally providing a straight-line trajectory to the maximum stone burden). The tract is then dilated to 24-30 Fr over an Amplatz super-stiff wire using sequential fascial dilators (telescoping Amplatz dilators from 8 Fr to 30 Fr) or a one-step balloon dilator (faster but more expensive).
Key landmarks: access through the posterior calyx along the axis of the infundibulum (provides the safest tract through Brodel's relatively avascular plane of the kidney). The ideal access point on the skin is at the posterior axillary line, below the 12th rib, lateral to the paraspinal muscles. The needle should enter the renal parenchyma through the fornix of the calyx (not through the infundibulum or renal pelvis, which risks vascular injury). Supracostal access (above the 12th rib, typically through the 11th intercostal space) may be required for upper pole stones, staghorn calculi, or ureteral stones best accessed through an upper pole calyx, but carries a higher risk of pleural injury (~5-15% pneumothorax or hydrothorax). A post-procedure chest X-ray is mandatory after supracostal access.
Multiple accesses (2-3 tracts) may be required for complex staghorn calculi to achieve a stone-free state. Mini-PCNL (using smaller tracts, 14-20 Fr) reduces bleeding and morbidity but limits the size of stone fragments that can be extracted; it is suitable for smaller stones (< 2 cm). Post-PCNL, a nephrostomy tube is typically left in place for 24-48 hours for hemostasis and drainage; tubeless PCNL (placement of an internal ureteral stent without a nephrostomy tube) is increasingly practiced in uncomplicated cases.
15 Musculoskeletal Interventions
Vertebroplasty & Kyphoplasty
Vertebroplasty: Percutaneous injection of polymethylmethacrylate (PMMA) bone cement directly into a fractured vertebral body under fluoroscopic or CT guidance. Indicated for painful osteoporotic vertebral compression fractures (VCFs) refractory to conservative management (4-6 weeks of analgesics, bracing, physical therapy) and pathologic fractures from metastatic disease or myeloma. Technique: uni- or bipedicular approach with 11-13 gauge bone biopsy needles into the anterior two-thirds of the vertebral body. Cement is injected under continuous fluoroscopy to monitor for extravasation.
Kyphoplasty (balloon-assisted vertebroplasty): A balloon tamp is inflated within the vertebral body to restore height and create a cavity before cement injection. Advantages over vertebroplasty: partial height restoration, lower cement extravasation rate (contained within the created cavity). The VERTOS II trial (2010) showed superiority of vertebroplasty over conservative treatment for acute VCFs (PMID: 20801742). Complications: cement extravasation (into the spinal canal — can cause cord compression requiring emergent decompression; into the neural foramina — radiculopathy; into the venous system — can cause pulmonary cement embolism), adjacent level fracture (~12-20% within 1-2 years), infection (< 1%), and rib fracture (from prone positioning). Absolute contraindications: asymptomatic VCF, active osteomyelitis at the target level, uncorrectable coagulopathy, and allergy to PMMA components. The VERTOS IV trial (2018) showed that vertebroplasty was not superior to sham procedure for acute VCFs < 9 weeks old (PMID: 29954840), generating ongoing debate about optimal patient selection.
Bone Tumor Ablation — Cryoablation
Percutaneous cryoablation is increasingly used for bone metastases (renal cell carcinoma, lung, breast) for pain palliation and local tumor control. Cryoprobes are placed under CT guidance; the ice ball is visible on CT (advantage over thermal ablation where the ablation zone is less visible). Typical protocol: two freeze-thaw cycles (10-minute freeze, 8-minute passive thaw, 10-minute freeze). The ice ball should extend ≥ 1 cm beyond the tumor margin. Cryoablation adjacent to neural structures requires careful monitoring (thermocouple needles) or nerve-protective measures (warm saline injection to insulate). Post-cryoablation augmentation with cement (cryoplasty) can be performed for weight-bearing bones at risk of pathologic fracture.
Joint Injections & Aspirations
Image-guided joint injections (US, fluoroscopy, or CT) provide precise needle placement, especially for deep joints (hip, sacroiliac) or joints with complex anatomy. Common indications: diagnostic/therapeutic aspiration (septic arthritis, crystal arthropathy), corticosteroid injection (osteoarthritis, adhesive capsulitis), and viscosupplementation (hyaluronic acid). Arthrography (injection of contrast into a joint) is combined with MRI (MR arthrography) for evaluation of labral tears, cartilage defects, and ligamentous injury. Fluoroscopic-guided facet joint injections, medial branch blocks, and sacroiliac joint injections are performed for spinal pain management.
Percutaneous Sclerotherapy for Venous & Lymphatic Malformations
Venous malformations (VMs) are low-flow vascular malformations composed of dysplastic venous channels. Treatment: percutaneous sclerotherapy under US and fluoroscopic guidance. Sclerosants: sodium tetradecyl sulfate (STS, 1-3%) mixed with air to create foam (Tessari method — improves contact with the endothelium), absolute ethanol (most potent sclerosant but highest complication rate — skin necrosis, nerve injury, cardiac arrest if > 0.5 mL/kg enters the systemic circulation), bleomycin (antibiotic/antineoplastic with sclerosing properties, well-suited for superficial VMs and pediatric patients, lower pain than ethanol; cumulative dose limit: 400 mg total to avoid pulmonary fibrosis), and doxycycline (for macrocystic lymphatic malformations — instill into the cyst, dwell for 30-60 minutes, aspirate). Multiple treatment sessions are typically required (3-5 sessions at 6-8 week intervals). VMs recur frequently; sclerotherapy provides volume reduction and symptom control rather than cure.
Lymphatic malformations (LMs) are classified as macrocystic (cysts > 2 cm — respond well to sclerotherapy), microcystic (cysts < 2 cm — poor response to sclerotherapy, may require surgical debulking), or mixed. Sclerosants for macrocystic LMs: doxycycline, OK-432 (picibanil, unavailable in the US), bleomycin, and STS foam. Technique: aspirate cyst fluid, inject sclerosant, and allow dwell time. Success rates for macrocystic LMs: ~80-90% with sclerotherapy.
16 Thoracic Interventions
Image-Guided Chest Tube Placement
IR-guided chest tube placement (pigtail catheter, 8-14 Fr) is performed for pleural effusion, empyema, or pneumothorax when standard bedside thoracostomy is not feasible or has failed. Advantages of image guidance: precise catheter positioning (dependent collection, loculated fluid), smaller caliber catheter (less pain), and reduced complication rate. Technique: US or CT identifies the collection; the safe access window is confirmed (avoiding the intercostal neurovascular bundle by entering over the superior border of the rib); Seldinger technique for catheter placement. Catheter tip should be positioned in the most dependent portion of the collection for effusion and at the apex for pneumothorax.
For empyema with loculations, intrapleural fibrinolytic therapy (tPA 10 mg + DNase 5 mg in 30-100 mL NS, instilled through the drain, clamped for 1-2 hours, then opened to suction, q12h for 3-6 doses — per the MIST2 trial protocol, PMID: 21131259) breaks down septations and improves drainage. The MIST2 trial demonstrated that the combination of tPA + DNase was superior to either agent alone or placebo in reducing the need for surgery. Drain output, temperature, and inflammatory markers (WBC, CRP) guide treatment response. Surgical referral (VATS decortication) is indicated if fibrinolytic therapy fails to achieve adequate drainage within 3-5 days.
Lung Biopsy
CT-guided percutaneous lung biopsy is the standard for peripheral pulmonary nodules not accessible by bronchoscopy. Core biopsy (18-20 gauge) has a diagnostic yield of ~90-95% for malignancy. The major complication is pneumothorax (15-25%), with chest tube required in ~5-7%. Blood-patch technique (injecting autologous blood or gelatin sponge through the coaxial needle as it is withdrawn from the lung) can reduce pneumothorax rates. Post-procedure: biopsy-side down positioning for 1-2 hours, chest X-ray at 2-4 hours.
Thoracic Duct Embolization
Thoracic duct embolization (TDE) is indicated for chylothorax or chylous ascites refractory to conservative management (low-fat diet, TPN, octreotide — typically a trial of 1-2 weeks) and thoracic duct ligation. Common etiologies of chylothorax: iatrogenic (post-esophagectomy, post-thoracic/cervical spine surgery, post-left neck dissection), trauma, malignancy (lymphoma), and idiopathic.
Technique: (1) intranodal lymphangiography — a peripheral inguinal lymph node is accessed under US guidance with a 25-gauge spinal needle, and ethiodized oil (Lipiodol) is slowly injected (1 mL over 15-20 minutes) to opacify the lymphatic channels and cisterna chyli; (2) the cisterna chyli (typically located at the L1-L2 level, anterior to the vertebral body, right of midline) is identified on fluoroscopy as it fills with Lipiodol; (3) the cisterna is accessed percutaneously via a transabdominal approach with a 21-gauge needle under fluoroscopic guidance; (4) a microwire and microcatheter are advanced into the thoracic duct; (5) the thoracic duct is embolized with coils and NBCA glue from distal to proximal. Technical success ~70-80%; clinical success in reducing chylous output ~70-90%. If the cisterna cannot be accessed (in ~20% of cases), therapeutic lymphangiography with Lipiodol alone can occlude the leak (the Lipiodol itself acts as an embolic agent within the lymphatic channels).
Tunneled Pleural Catheter (PleurX)
Tunneled pleural catheters are placed for recurrent malignant pleural effusions when pleurodesis has failed or is not feasible. The PleurX catheter is a 15.5 Fr silicone catheter with a polyester cuff in the subcutaneous tunnel (similar to a tunneled CVC). Patients or caregivers drain the effusion at home using vacuum bottles (typically 500-1000 mL every other day). Spontaneous pleurodesis occurs in ~45-70% of patients over several weeks to months due to the continuous drainage creating apposition of the visceral and parietal pleura. Complications: infection/empyema (~3-5%), catheter occlusion, tract metastasis (~1%), and pneumothorax.
17 GI & Hepatobiliary Interventions
Percutaneous Cholecystostomy
Percutaneous cholecystostomy is the placement of a drainage catheter into the gallbladder for acute cholecystitis in patients who are poor surgical candidates (critically ill, septic, high surgical risk). The Tokyo Guidelines (TG18/TG13) stratify acute cholecystitis severity: Grade I (mild) — does not meet Grade II/III criteria; Grade II (moderate) — elevated WBC, palpable mass, symptom duration > 72 hours, or marked local inflammation; Grade III (severe) — organ dysfunction (cardiovascular, neurologic, respiratory, renal, hepatic, hematologic). Percutaneous cholecystostomy is recommended for Grade III cholecystitis when emergent cholecystectomy carries prohibitive risk.
Performed under US guidance via a transhepatic approach (preferred — the catheter traverses hepatic parenchyma, which reduces bile peritonitis risk if the catheter dislodges and creates a more stable tract) or transperitoneal approach (shorter path, used when the gallbladder fundus is adherent to the anterior abdominal wall). An 8-10 Fr locking pigtail catheter is standard. Clinical improvement typically occurs within 24-48 hours. A cholecystogram (contrast injection through the tube) is performed at 2-3 weeks to assess cystic duct patency — if the cystic duct is patent and the patient is an operative candidate, the tube is capped and removed before or during interval cholecystectomy; if the cystic duct is obstructed, the tube remains in place for continued drainage.
Gastrostomy & Gastrojejunostomy Tubes
Percutaneous radiologic gastrostomy (PRG) is placed under fluoroscopic guidance when endoscopic gastrostomy (PEG) is not feasible (head and neck cancer with complete oropharyngeal obstruction, prior gastric surgery, altered upper GI anatomy, or inability to perform transillumination for PEG). Technique: (1) gastric insufflation via a nasogastric tube (if present) or percutaneous needle insufflation; (2) gastropexy using T-fasteners (typically 3-4 fasteners deployed around the planned gastrostomy site to anchor the stomach to the anterior abdominal wall — this critical step prevents peritonitis from gastric leak); (3) Seldinger access into the stomach through the center of the T-fastener array; (4) tract dilation to 12-16 Fr; and (5) deployment of a balloon-retention or mushroom-tip gastrostomy tube. T-fasteners are typically cut at the skin surface and released at 10-14 days once the tract has matured.
Gastrojejunostomy (GJ) tubes have a gastric port for decompression/medication administration and a jejunal extension for feeding — indicated for patients with gastroparesis, severe gastroesophageal reflux, or high aspiration risk. The jejunal limb is advanced through the pylorus and duodenum into the proximal jejunum (beyond the ligament of Treitz) using a combination of guidewire, catheter, and fluoroscopic guidance. Securing the jejunal limb in position can be challenging; some operators use the Draeger technique (weighted suture through the jejunal limb to prevent retraction). Tube exchanges: balloon-retention gastrostomy tubes are exchanged every 3-6 months; GJ tubes are exchanged when the jejunal limb migrates back into the stomach (a common problem). First-time tube exchanges should not be performed until the tract has matured (minimum 4 weeks after initial placement, ideally 6-8 weeks).
Cecostomy
Percutaneous cecostomy is placed for sigmoid volvulus decompression in patients not amenable to endoscopic decompression or surgery, and in pediatric patients for antegrade continence enemas (Malone-equivalent). Placed under CT or fluoroscopic guidance with T-fastener fixation of the cecum to the anterior abdominal wall. Catheter size: 10-14 Fr locking pigtail. The mature cecostomy tract forms over 2-4 weeks and allows tube exchanges as needed.
Abscess Drainage — Specific Locations
Hepatic abscess: Most commonly pyogenic (E. coli, Klebsiella, polymicrobial); less commonly amebic (Entamoeba histolytica — treat medically with metronidazole first, drain only if large > 5 cm, no response, or risk of rupture). Percutaneous catheter drainage is first-line for pyogenic liver abscesses > 3 cm. Smaller abscesses may respond to needle aspiration alone. Send fluid for aerobic, anaerobic, fungal, and AFB cultures. Duration of IV antibiotics: typically 2-4 weeks, followed by oral antibiotics to complete 4-6 weeks total.
Pelvic abscess: May be drained via a transgluteal (posterior), transrectal, or transvaginal approach depending on location. The transgluteal approach passes through the greater sciatic foramen, avoiding the sciatic nerve and internal iliac vessels (the catheter path should be inferior to the piriformis muscle and medial to the sciatic nerve). Transrectal drainage using endocavitary US guidance provides a direct route to deep pelvic collections without traversing the peritoneum.
Pancreatic collections: Revised Atlanta classification defines: acute peripancreatic fluid collection (APFC — < 4 weeks, no wall), pancreatic pseudocyst (> 4 weeks, encapsulated, no solid debris), acute necrotic collection (ANC — < 4 weeks, with necrotic debris), and walled-off necrosis (WON — > 4 weeks, encapsulated necrosis). Infected necrosis requires drainage: percutaneous catheter drainage (step-up approach per the PANTER trial — start with percutaneous drainage, escalate to minimally invasive retroperitoneal necrosectomy only if needed; PMID: 20410514). Large-bore drains (14-24 Fr) are often required for thick necrotic material. Catheter upsizing and multiple drains may be necessary.
Transjugular Intrahepatic Portosystemic Shunt (TIPS)
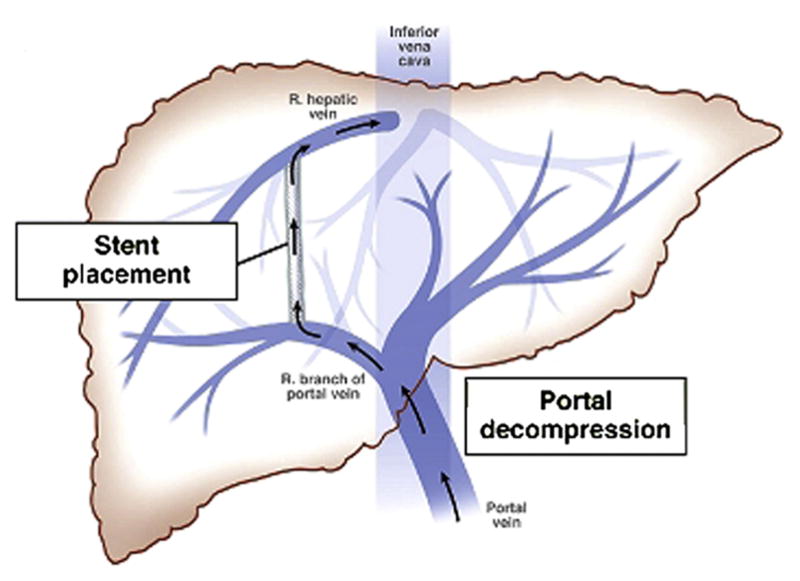
TIPS creates a shunt between the hepatic vein and the portal vein through the hepatic parenchyma, decompressing the portal system. Indications: refractory variceal bleeding (acute or secondary prophylaxis), refractory ascites, Budd-Chiari syndrome, hepatic hydrothorax, hepatorenal syndrome (bridge to transplant). Technique: via right IJV access, a catheter is advanced into a hepatic vein (typically right or middle); a long needle (TIPS set, e.g., Rosch-Uchida) is passed through the hepatic parenchyma into a portal vein branch; the tract is dilated and a covered stent (Viatorr, 8-10 mm) is deployed. Target portosystemic gradient: < 12 mmHg (below the threshold for variceal bleeding).
Complications: hepatic encephalopathy (~30-40% — the most common complication; occurs because the shunt diverts portal blood carrying gut-derived ammonia and other toxins directly into the systemic circulation, bypassing hepatic detoxification; managed with lactulose 15-30 mL PO q6h titrated to 2-4 bowel movements/day and rifaximin 550 mg PO BID; refractory encephalopathy may require TIPS reduction using a flow-restricting device placed within the stent); stent stenosis/occlusion (reduced dramatically with covered stents — Viatorr has ~80% primary patency at 1 year vs ~30-50% for bare metal stents; surveillance with Doppler US every 3-6 months to detect stenosis early; stent revision with angioplasty and/or relining for hemodynamically significant stenosis); hepatic failure (especially in patients with high MELD score — TIPS is generally contraindicated if MELD > 18-20 or Child-Pugh > 13; risk of precipitating liver failure by diverting portal perfusion from already compromised hepatocytes); right heart failure (TIPS increases preload to the right heart by ~15-20%; can unmask subclinical RV dysfunction; pre-TIPS echocardiography is recommended to assess RV function and estimate RVSP); and intraperitoneal hemorrhage (from portal vein capsular puncture during the procedure — this life-threatening complication occurs in ~1-2% and requires emergent embolization of the hepatic parenchymal tract).
Pre-TIPS assessment should include: Doppler US (confirm portal vein patency and direction of flow — TIPS cannot be performed if the portal vein is completely thrombosed without a targetable patent segment), echocardiography (assess RV function, rule out pulmonary hypertension — RVSP > 45 mmHg is a relative contraindication), MELD score calculation (stratify procedural risk), and cross-sectional imaging (CT or MRI to map portal and hepatic venous anatomy, identify portal vein thrombus, and plan the transhepatic puncture trajectory). Portal vein thrombosis (partial) can actually be an indication for TIPS if it is performed to re-establish portal flow and allow anticoagulation.
18 Transarterial Chemoembolization (TACE)
Principles & Indications
TACE exploits the dual blood supply of the liver: hepatocellular carcinoma (HCC) derives > 90% of its blood supply from the hepatic artery, while normal hepatocytes receive ~75% from the portal vein. By selectively delivering chemotherapy and embolic material into the tumor-feeding arteries, TACE achieves high intratumoral drug concentrations and ischemic necrosis while relatively sparing normal parenchyma. Primary indication: intermediate-stage HCC (BCLC stage B) — multinodular, preserved liver function (Child-Pugh A or B7), no portal vein invasion, no extrahepatic spread. TACE is also used for liver-dominant metastases (neuroendocrine, melanoma, colorectal — with DEB-TACE) and as a bridge to liver transplant or downstaging.
Conventional TACE (cTACE)
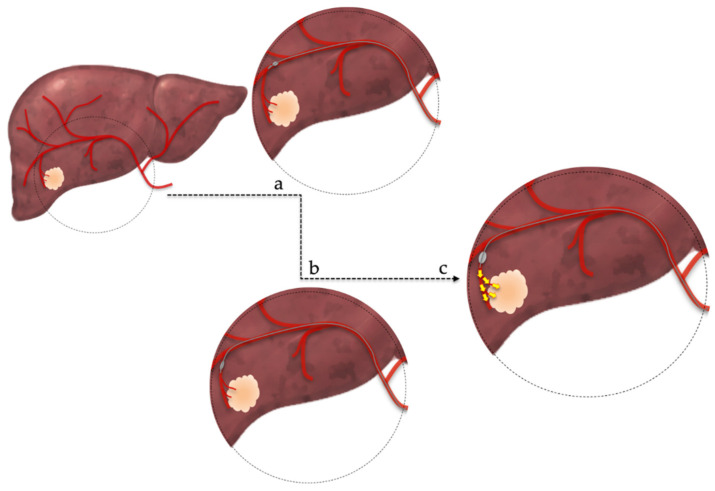
Conventional TACE involves injection of a chemotherapeutic agent (typically doxorubicin 50-75 mg or cisplatin 50-100 mg) mixed with ethiodized oil (Lipiodol) into the tumor-feeding artery, followed by particle embolization (Gelfoam pledgets, PVA 100-300 μm, or calibrated microspheres). The Lipiodol serves three functions: (1) drug carrier (creates a water-in-oil emulsion that concentrates chemotherapy within the tumor), (2) radiopaque marker (CT retention at 4-6 weeks correlates with tumor response — compact Lipiodol uptake indicates complete necrosis), and (3) microembolic agent (Lipiodol itself causes distal arteriolar embolization). Embolization endpoint: near-stasis ("tree-in-winter" appearance) in the tumor-feeding artery with dense Lipiodol filling of the tumor on fluoroscopy.
Technical considerations: catheter position should be as selective as possible (lobar at minimum; segmental or subsegmental preferred) to maximize drug delivery to the tumor and minimize injury to normal parenchyma. The Lipiodol-drug emulsion is prepared by vigorously mixing Lipiodol (5-20 mL) with the chemotherapy agent using two connected syringes (pumping method) until a uniform emulsion is achieved. The emulsion is injected slowly under fluoroscopy. Over-embolization risks non-target hepatic infarction; under-embolization results in insufficient treatment. Superselective TACE targeting a single tumor's feeding artery provides the best tumor kill with the least collateral damage.
DEB-TACE (Drug-Eluting Bead TACE)
DEB-TACE uses calibrated microspheres loaded with doxorubicin (DC Bead/Oncozene, HepaSphere). The beads provide sustained local drug release over 7-14 days and permanent vascular occlusion. Advantages over cTACE: more standardized procedure, lower systemic drug exposure, potentially fewer systemic side effects. The PRECISION V trial showed similar tumor response rates for DEB-TACE and cTACE, with fewer drug-related side effects in the DEB-TACE group (PMID: 19717544). Bead size selection: smaller beads (70-150 μm) penetrate more distally into the tumor vasculature; larger beads (300-500 μm) provide more proximal occlusion.
Post-Embolization Syndrome
Post-embolization syndrome (PES) occurs in 60-80% of TACE patients. Triad: right upper quadrant pain, nausea/vomiting, and fever (low-grade, typically < 38.5°C). Caused by tumor ischemia and necrosis. Onset: within hours; duration: 2-7 days. Management: IV hydration, antiemetics (ondansetron), analgesics (ketorolac, opioids), and monitoring. Distinguish PES from hepatic abscess (fever > 38.5°C persisting beyond 7 days, leukocytosis, imaging findings — requires aspiration/drainage). Patients with bilioenteric anastomosis (e.g., prior Whipple procedure) are at very high risk for hepatic abscess post-TACE due to enteric bacteria in the biliary system — prophylactic antibiotics are mandatory.
Assessment of Response
Response after TACE is assessed using mRECIST (modified Response Evaluation Criteria in Solid Tumors), which measures viable (enhancing) tumor rather than total tumor size:
| Response Category | mRECIST Definition |
|---|---|
| Complete response (CR) | Disappearance of any intratumoral arterial enhancement in all target lesions |
| Partial response (PR) | ≥ 30% decrease in the sum of diameters of viable (enhancing) target lesions |
| Stable disease (SD) | Any cases that do not qualify for either PR or PD |
| Progressive disease (PD) | ≥ 20% increase in the sum of diameters of viable target lesions, or new lesion(s) |
Follow-up imaging: contrast-enhanced CT or MRI at 4-6 weeks post-TACE. MRI with hepatocyte-specific contrast (gadoxetate/Eovist) is preferred for superior characterization of viable vs necrotic tumor. Repeat TACE sessions are performed for residual viable tumor ("on-demand" approach) until complete response, progression to a contraindicated stage (vascular invasion, extrahepatic spread), or liver function deterioration (progression to Child-Pugh C). Most patients require 2-4 TACE sessions to achieve maximal response. The concept of "TACE refractoriness" (failure to achieve objective response after ≥ 2 consecutive TACE sessions, or deterioration of liver function to Child-Pugh C) should prompt transition to systemic therapy or transplant evaluation.
Bland Embolization (TAE)
Transarterial bland embolization (TAE) uses embolic particles (microspheres, PVA) without chemotherapy agents. Some studies (including a Japanese RCT by Maeda et al.) have shown similar outcomes for TAE vs cTACE for HCC, suggesting that the embolic (ischemic) effect may be the dominant therapeutic mechanism. TAE is simpler, avoids chemotherapy-related toxicity, and may be preferred for patients with compromised liver function. However, TACE remains the standard of care in most guidelines (AASLD, EASL) based on the available evidence, including the landmark Llovet et al. 2002 RCT that established TACE as superior to best supportive care for intermediate HCC (PMID: 12091880).
19 Transarterial Radioembolization (TARE / Y-90)
Mechanism & Microsphere Types
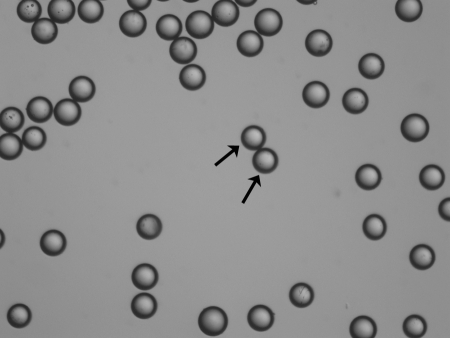
TARE (Y-90 radioembolization) delivers yttrium-90-laden microspheres into the hepatic artery. Y-90 is a pure beta emitter with a mean tissue penetration of 2.5 mm and a half-life of 64.2 hours. Two FDA-approved devices: TheraSphere (glass microspheres, Boston Scientific) — higher activity per sphere (~2,500 Bq), smaller number of spheres (1-8 million), less embolic effect, can be used with portal vein thrombosis; and SIR-Spheres (resin microspheres, Sirtex) — lower activity per sphere (~50 Bq), larger number of spheres (40-80 million), more embolic effect. TheraSphere is FDA-approved under an HDE for HCC; SIR-Spheres has full PMA for colorectal liver metastases.
Mapping Angiogram & Treatment Planning
A mapping (planning) angiogram is performed 1-2 weeks before Y-90 treatment. Goals: (1) map the hepatic arterial anatomy and identify all tumor-feeding vessels; (2) identify and embolize (coil) extrahepatic vessels that could cause non-target radiation (GDA, right gastric artery, cystic artery, falciform artery, left/right inferior phrenic arteries); (3) perform Tc-99m MAA injection (macroaggregated albumin) through the treatment catheter to simulate microsphere distribution and calculate the lung shunt fraction (LSF).
LSF < 10%: Proceed with full prescribed dose.
LSF 10-15%: Proceed with dose reduction (typically 20-40% reduction) or segmental treatment.
LSF 15-20%: Dose reduction required; consider limiting to a single lobe treatment.
LSF > 20%: Relative contraindication due to risk of radiation pneumonitis. Absolute cumulative lung dose > 30 Gy per session or > 50 Gy total is the threshold for pneumonitis.
Dosimetry
TheraSphere dosimetry (medical internal radiation dose — MIRD model): Activity (GBq) = Desired dose (Gy) × Liver mass (kg) / 50. Standard target dose: 80-150 Gy for whole-lobe treatment; 200-400+ Gy for radiation segmentectomy. SIR-Spheres dosimetry (BSA model or partition model): The BSA model calculates activity based on body surface area and tumor involvement; the partition model (more accurate) uses MAA SPECT to determine the tumor-to-normal liver ratio and calculates doses to tumor and normal liver separately.
Radiation Segmentectomy
Radiation segmentectomy involves superselective Y-90 delivery to 1-2 hepatic segments, delivering very high doses (> 190 Gy) to small treatment volumes. This approach achieves pathologic complete necrosis rates of ~80-90% and is increasingly used as a curative-intent therapy for early-stage HCC (BCLC 0/A, single lesion ≤ 5 cm). The LEGACY trial showed 72.2% complete pathologic necrosis and was the basis for TheraSphere approval for HCC (PMID: 34935560).
Y-90 Complications & Side Effects
Post-radioembolization syndrome: Fatigue (most common, ~60%), nausea, mild abdominal pain, and low-grade fever. Milder than post-TACE syndrome because Y-90 causes radiation-induced necrosis rather than ischemic necrosis. Duration: 1-2 weeks. Radiation-induced liver disease (RILD): Rare but potentially fatal; presents 4-8 weeks post-treatment with jaundice, ascites, and elevated liver enzymes in a pattern disproportionate to tumor burden. Risk increases with whole-lobe treatment of an already compromised liver. Prevention: accurate dosimetry ensuring normal liver dose < 70 Gy (TheraSphere) or < 40-50 Gy (SIR-Spheres). GI ulceration: From non-target microsphere deposition in GI arteries; prevented by coil embolization of extrahepatic vessels during the mapping angiogram. Radiation pneumonitis: From excessive lung shunting; prevented by adhering to LSF dose thresholds (cumulative lung dose < 30 Gy per session). Biliary complications: Biliary necrosis and biloma can occur when biliary structures are within the radiation field, especially with high-dose radiation segmentectomy.
Y-90 vs TACE — Evidence Summary
Several randomized trials have compared Y-90 (TARE) to TACE for HCC. The TRACE trial (2022) showed non-inferiority of Y-90 to TACE for intermediate-stage HCC with better quality of life. The DOSISPHERE trial demonstrated superior OS with personalized dosimetry (≥ 205 Gy to the tumor) compared to standard dosimetry. Y-90 is increasingly preferred over TACE for: large solitary tumors (> 5 cm), tumors with portal vein thrombosis (TARE is not contraindicated by portal vein invasion, unlike TACE), and as a bridge to transplant or downstaging strategy. Radiation segmentectomy for early-stage HCC is being studied head-to-head against ablation in the RACER trial.
20 Thermal Ablation (RFA, MWA, Cryoablation)
General Principles of Thermal Ablation
Thermal ablation destroys tumor tissue through temperature-mediated cell death. Hyperthermic ablation (RFA, MWA) achieves cell death at temperatures > 60°C (instantaneous coagulative necrosis) through protein denaturation and membrane disruption. Between 42-60°C, cell death requires prolonged exposure (minutes to hours). Hypothermic ablation (cryoablation) causes cell death through intracellular ice crystal formation at temperatures < -20°C, followed by osmotic injury during thawing. All ablation techniques aim for a circumferential ablation margin of ≥ 5-10 mm beyond the visible tumor boundary to account for microscopic tumor extension. Pre-ablation planning: careful review of cross-sectional imaging (CT/MRI) to determine tumor size, location, relationship to critical structures (bile ducts, vessels, bowel, diaphragm, nerves), and the planned approach (percutaneous vs laparoscopic vs open).
Radiofrequency Ablation (RFA)
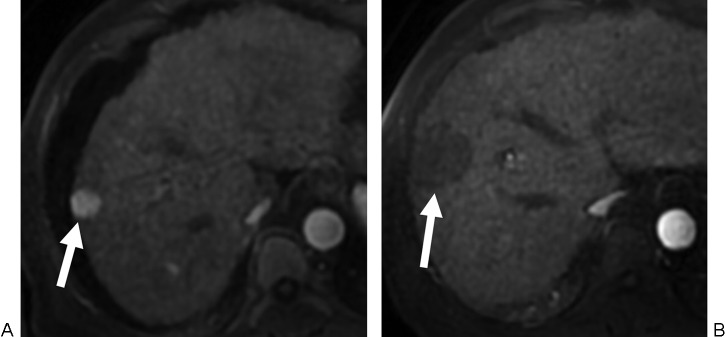
RFA delivers alternating electrical current (375-500 kHz) through a needle electrode into the tumor, causing ionic agitation and frictional heat generation (target tissue temperature: 60-100°C, causing immediate coagulative necrosis). The circuit is completed through grounding pads placed on the patient's thighs. Electrode types: single straight needle, expandable (LeVeen, Starburst — deploy tines in an umbrella configuration to increase ablation zone from 2 cm to 4-5 cm), and internally cooled (Cool-tip — circulating chilled saline prevents tissue charring near the electrode, which acts as an insulator reducing current flow). Ablation zone: typically 2-5 cm depending on electrode size, power, and duration (typically 12-20 minutes per ablation cycle). Limitation: the "heat sink" effect — adjacent large blood vessels (≥ 3 mm) dissipate heat via flowing blood, reducing ablation efficacy at the tumor margin. Hepatic tumors adjacent to major portal or hepatic veins have higher local recurrence rates after RFA. Strategies to mitigate the heat-sink effect include temporary balloon occlusion of the draining vein (Pringle maneuver equivalent) and combining embolization with ablation.
Microwave Ablation (MWA)
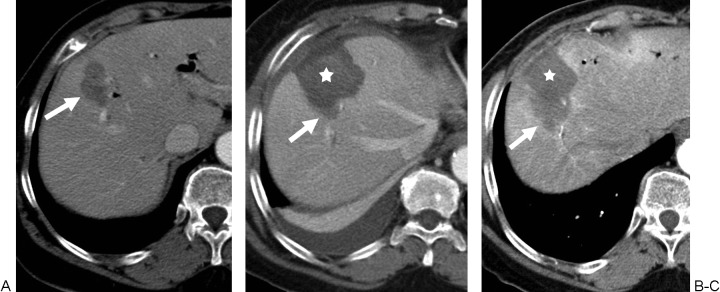
MWA uses electromagnetic microwaves (915 MHz or 2.45 GHz) to agitate water molecules in tissue, generating heat through dielectric hysteresis (the molecular rotation of polar water molecules attempting to align with the oscillating electromagnetic field). Unlike RFA (which relies on electrical current conduction and is therefore limited by tissue impedance), MWA actively generates heat in any tissue containing water, regardless of impedance. This fundamental difference accounts for most of MWA's advantages over RFA.
Advantages over RFA: (1) faster heating (temperatures > 100°C reached within seconds vs minutes), (2) larger ablation zones (up to 5-6 cm in a single antenna, vs 3-4 cm for RFA), (3) significantly less susceptibility to the heat-sink effect (microwave energy continues to propagate regardless of blood flow, whereas RFA's convective heating is directly impaired by adjacent blood flow), (4) no grounding pads required (microwave energy is emitted from the antenna directly into tissue, unlike RFA which requires a completed electrical circuit), (5) effective in tissues with high impedance such as lung and bone (which limit RFA's electrical current), and (6) the ability to use multiple antennas simultaneously (creating synergistic heating zones that merge to form a single large ablation). MWA has become the preferred thermal ablation modality for liver tumors in many centers.
Modern MWA systems: Emprint (Medtronic) — 2.45 GHz, thermosphere technology for spherical ablation zones, power up to 100W; NeuWave (Ethicon/J&J) — 2.45 GHz, certus 140 system with real-time thermal profiling; Solero (AngioDynamics) — 915 MHz, triaxial antenna design. Multiple antennas (2-4) can be placed simultaneously with 1-2 cm spacing for large or irregularly shaped tumors — the overlapping microwave fields create synergistic heating that produces a confluent ablation zone larger than the sum of individual antenna zones. Ablation time: typically 5-10 minutes per ablation cycle (significantly shorter than RFA's 12-20 minutes or cryoablation's 30-60 minutes). The primary disadvantage of MWA compared to cryoablation is that the ablation zone is less visible on intraprocedural CT (transient periablational ground-glass opacity is seen but does not clearly delineate the lethal margin) — contrast-enhanced imaging (CEUS or CBCT) is needed to assess completeness.
Cryoablation
Cryoablation uses argon gas (Joule-Thomson effect — rapid expansion of compressed gas through a small orifice causes dramatic cooling) delivered through cryoprobes to rapidly freeze tissue to -20 to -40°C, causing intracellular ice crystal formation, osmotic dehydration, and microvascular thrombosis. Helium gas is used for the active thaw cycle (Joule-Thomson effect in reverse — helium heats upon expansion). Modern cryoablation systems (Galil Medical/BTG, IceCure) allow placement of multiple cryoprobes (2-8) simultaneously to create large, conformal ablation zones for irregularly shaped tumors.
The ice ball is directly visible on CT and MRI (appears as a well-defined, low-density/low-signal region) but not on US (acoustic shadowing limits posterior visualization), allowing real-time monitoring of the ablation zone. Protocol: typically two freeze-thaw cycles (10 min freeze, 8 min passive thaw, 10 min freeze; some protocols use active thaw with helium). The lethal margin of the ice ball is ~3-5 mm inside the visible edge (tissue at the ice ball margin is at 0°C, which is not sufficient for cell kill; lethal temperature of -20°C to -40°C is achieved several millimeters inside the visible boundary). Therefore, the ice ball should extend at least 5-10 mm beyond the tumor margin to ensure the lethal zone encompasses the entire tumor.
Advantages: well-defined ablation zone on imaging (best intraprocedural monitoring of any ablation modality), less pain than heat-based ablation (ice provides cryoanalgesia), can treat lesions near critical structures with better real-time monitoring, no grounding pads needed, and multiple probes can be used simultaneously. Disadvantages: longer procedure time (30-60 minutes per ablation cycle vs 10-15 minutes for MWA), risk of cryoshock (rare but life-threatening systemic release of cytokines and cellular debris from large-volume ablation — presents like SIRS with hypotension, DIC, multiorgan failure; risk increases with ablation volumes > 200-300 mL), higher hemorrhagic risk than heat-based ablation (no cautery effect — the ablation zone does not seal blood vessels as effectively as thermal coagulation), and higher cost per procedure (multiple cryoprobes, argon/helium gas). Cryoablation is preferred over RFA/MWA for renal tumors (better visualization, preservation of collecting system), bone metastases (ice ball visibility on CT, cryoanalgesia), and tumors adjacent to critical structures where real-time monitoring is essential.
Indications & Target Organs
Liver (HCC): BCLC 0/A — single tumor ≤ 3 cm (ablation is equivalent to resection per multiple RCTs including SURF trial); up to 3 tumors each ≤ 3 cm (Milan criteria). Ablation margin: aim for ≥ 5-10 mm circumferential margin beyond the tumor boundary. RFA and MWA have comparable efficacy for tumors ≤ 3 cm; MWA is preferred for tumors 3-5 cm. (PMID: 31304473)
Liver (metastases): Colorectal liver metastases not amenable to resection — ablation combined with systemic chemotherapy (CLOCC trial showed improved OS with RFA + chemo vs chemo alone for unresectable CRLM; PMID: 28128024).
Kidney (RCC): T1a tumors (≤ 4 cm) in patients who are poor surgical candidates or prefer nephron-sparing approaches. Cryoablation is most commonly used for renal tumors due to excellent CT visibility of the ice ball and less injury to the collecting system. Biopsy before or concurrent with ablation is recommended.
Lung: Stage I NSCLC in non-surgical candidates, pulmonary metastases (≤ 3 cm). Cryoablation and MWA are increasingly used. Pneumothorax is the most common complication (~30-40%).
Bone metastases: Cryoablation for pain palliation and local control. Often combined with cementoplasty for weight-bearing bones.
21 Portal Vein Embolization
Rationale & Indications
Portal vein embolization (PVE) is performed before major hepatectomy to induce hypertrophy of the future liver remnant (FLR). By occluding portal venous flow to the liver segments planned for resection, hepatotrophic factors are redirected to the FLR, stimulating compensatory hypertrophy over 3-6 weeks. Indicated when the FLR is insufficient: < 20% of total liver volume in patients with normal liver, < 30% with chemotherapy-damaged liver (steatosis, steatohepatitis), or < 40% with cirrhosis. Without PVE, major hepatectomy with an inadequate FLR carries a high risk of post-hepatectomy liver failure (PHLF), which has a mortality rate of 50-75%.
Technique
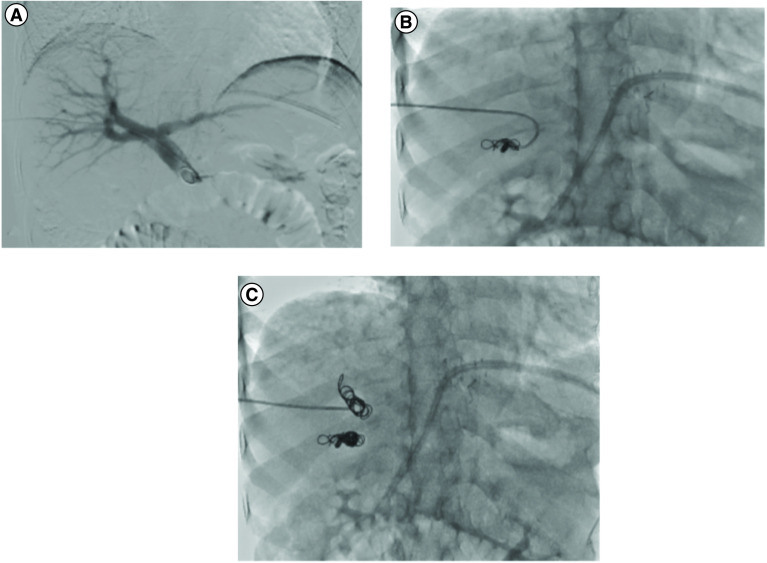
Approach: ipsilateral (percutaneous transhepatic) — puncture a portal vein branch in the lobe to be resected under US/fluoroscopic guidance and embolize antegrade. This is the most common approach and is preferred because: the puncture site will be within the resected specimen (no risk of damaging the FLR), and antegrade access provides direct catheterization of the target portal branches. Alternative: contralateral approach — puncture a portal vein branch in the FLR and catheterize across to the targeted side. This carries the disadvantage of traversing the FLR (potential bleeding, parenchymal injury to the remnant) but may be necessary if ipsilateral access is not feasible.
Embolic agents: NBCA (glue) mixed with Lipiodol (1:1 to 1:4 ratio depending on desired flow characteristics) is the most common agent worldwide — provides rapid, permanent occlusion and achieves more distal penetration than particles alone. Alternatives include PVA particles (300-500 μm), Gelfoam pledgets, coils (for main portal vein branch occlusion), and Amplatzer vascular plugs (for large-caliber proximal portal branches). Segment 4 embolization (in addition to right portal vein embolization for planned right or extended right hepatectomy) may be performed to maximize left lateral segment (segments 2-3) hypertrophy — this is especially important for extended right hepatectomy where the FLR consists only of segments 2-3.
Assessment of Hypertrophy
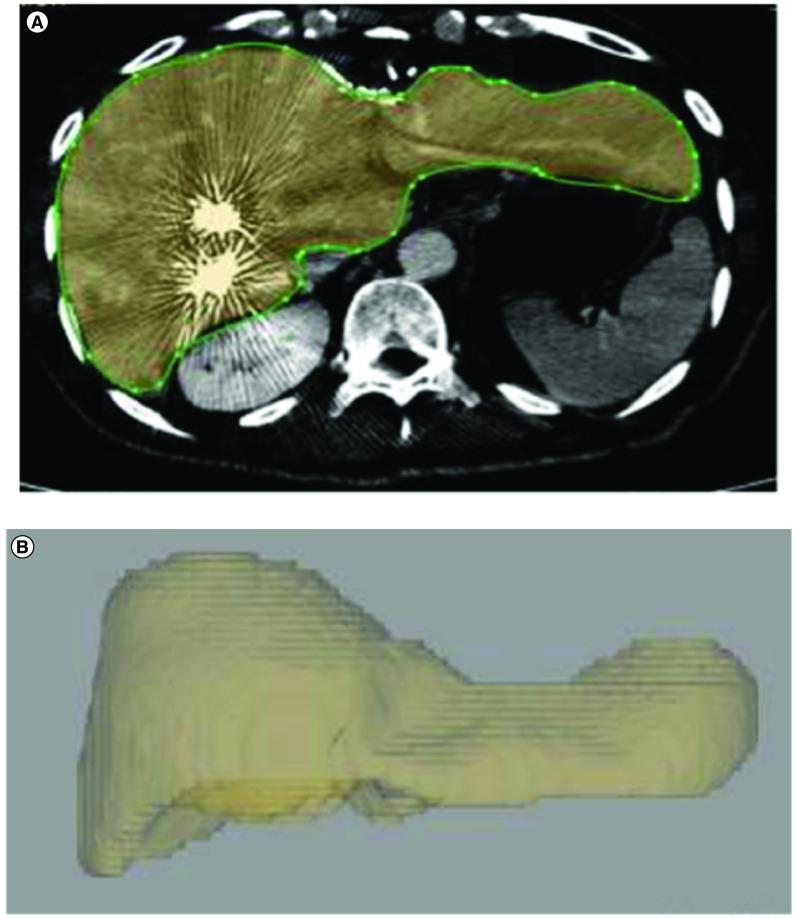
Volumetric CT or MRI is performed 3-6 weeks after PVE to measure FLR hypertrophy. Standardized FLR calculation: sFLR = FLR volume / (total liver volume - tumor volume) × 100%. The total liver volume can be measured on CT or estimated by formula (Vauthey): TLV = -794.41 + 1267.28 × BSA. Target sFLR thresholds: ≥ 20% for normal liver, ≥ 30% for chemotherapy-damaged liver, ≥ 40% for cirrhotic liver.
A kinetic growth rate (KGR) > 2% per week (KGR = [sFLR post-PVE - sFLR pre-PVE] / weeks since PVE) is a positive predictor of adequate post-operative liver function. KGR < 2%/week predicts higher risk of PHLF. Degree of hypertrophy (DH): typically 30-60% volume increase at 4-6 weeks with PVE alone. If the FLR has not reached the target volume after 4-6 weeks, options include: additional waiting time (repeat volumetry at 6-8 weeks), ipsilateral hepatic vein embolization to augment the effect (liver venous deprivation, see below), or ALPPS (Associating Liver Partition and Portal vein ligation for Staged hepatectomy) — a two-stage surgical procedure where the liver parenchyma is transected and the portal vein is ligated in the first stage, inducing rapid FLR hypertrophy (typically 47-93% volume increase sufficient by 7-14 days), followed by completion hepatectomy in the second stage. ALPPS achieves faster and more robust hypertrophy than PVE but carries higher morbidity (30-40%) and mortality (9-12% in early series, improving with patient selection and experience to ~5% in more recent reports). ALPPS is reserved for cases where PVE hypertrophy is inadequate or where tumor progression during the waiting period is a concern (e.g., colorectal liver metastases on chemotherapy hold).
Liver Venous Deprivation (LVD)
LVD is an emerging technique combining PVE with simultaneous hepatic vein embolization (HVE) of the future resection side. By occluding both portal inflow and venous outflow, LVD achieves greater and faster FLR hypertrophy than PVE alone. Early studies show ~50% FLR volume increase at 3 weeks with LVD versus ~30% with PVE alone. The venous outflow occlusion is performed using vascular plugs (Amplatzer) deployed in the right or middle hepatic vein via a transjugular approach. This technique may reduce the need for ALPPS and decrease the time to surgery. LVD is gaining adoption as a pre-hepatectomy optimization strategy, particularly in patients with borderline FLR who need to proceed to surgery rapidly (e.g., colorectal liver metastases on active chemotherapy).
22 Stroke Thrombectomy & Cerebrovascular Interventions
Mechanical Thrombectomy for Acute Ischemic Stroke
Endovascular thrombectomy (EVT) is the standard of care for acute ischemic stroke due to large vessel occlusion (LVO) of the anterior circulation (internal carotid artery, M1/proximal M2 segment of the middle cerebral artery). Five landmark trials in 2015 (MR CLEAN, ESCAPE, EXTEND-IA, SWIFT PRIME, REVASCAT) established EVT as superior to IV tPA alone for LVO stroke, with a number needed to treat (NNT) of ~2.6 for reduced disability. Key eligibility: NIHSS ≥ 6, small infarct core on imaging (CT perfusion or DWI-MRI with ASPECTS ≥ 6), and treatment within 6 hours from symptom onset (extended to 24 hours by DAWN and DEFUSE 3 trials for patients with favorable perfusion mismatch — small core, large penumbra).
Technique: CFA access (typically right), 8 Fr guide catheter or balloon guide catheter (BGC) in the ICA (BGC provides flow arrest during retrieval, improving reperfusion rates and reducing distal embolization), microcatheter/microwire navigation past the clot, deployment of a stent retriever (Solitaire, Trevo) or aspiration catheter (Penumbra ACE/JET). The stent retriever is deployed across the clot, allowed to integrate for 3-5 minutes, and then withdrawn into the guide catheter while applying aspiration. ADAPT (A Direct Aspiration First Pass Technique) uses a large-bore aspiration catheter advanced directly to the face of the clot. Combined techniques (stent retriever + aspiration, "Solumbra") are commonly used. The target outcome is TICI (Thrombolysis in Cerebral Infarction) grade 2b/3 reperfusion (successful reperfusion of > 50% of the affected territory), achieved in ~80-90% of cases at high-volume centers.
Grade 0: No perfusion — no antegrade flow beyond the point of occlusion.
Grade 1: Penetration with minimal perfusion — contrast passes beyond the obstruction but fails to opacify the entire distal bed.
Grade 2a: Partial filling (< 50%) of the entire vascular territory.
Grade 2b: Complete filling of the expected vascular territory, but filling is slower than normal.
Grade 2c: Near-complete perfusion except for slow flow in a few distal cortical vessels.
Grade 3: Complete perfusion — normal antegrade flow in the entire vascular territory with normal filling in all distal branches.
Posterior Circulation Stroke Thrombectomy
EVT for basilar artery occlusion (BAO) has been an area of active investigation. The BEST trial (China, 2020) was neutral, but the BASICS trial (2021) and ATTENTION trial (2022) showed benefit of EVT over medical management for acute BAO. Current practice: EVT is offered for BAO in patients with significant neurological deficit (NIHSS ≥ 10 or coma) and limited established infarction on imaging, within 24 hours of symptom onset. Technique is similar to anterior circulation thrombectomy but involves catheterization of the vertebral artery (typically the dominant side or the side with the most favorable anatomy) and navigation to the basilar artery.
Intracranial Aneurysm Coiling
Endovascular coiling is an alternative to surgical clipping for intracranial aneurysms. Detachable platinum coils (originally Guglielmi detachable coils; modern coils include bioactive, hydrogel-coated, and shape-modified designs) are packed into the aneurysm sac via a microcatheter, promoting thrombosis within the aneurysm and excluding it from the circulation. The goal is dense packing (> 25% packing density) to prevent recanalization. For ruptured aneurysms, treatment should occur within 24 hours to prevent rebleeding (rebleeding risk ~4% in the first 24 hours, ~20% in the first 2 weeks).
Adjunctive techniques for wide-necked aneurysms (dome-to-neck ratio < 2:1 or neck > 4 mm): balloon-assisted coiling (a compliant balloon is temporarily inflated across the aneurysm neck during coil deployment to prevent coil herniation into the parent artery; the balloon is deflated between coils to assess flow), stent-assisted coiling (a low-profile intracranial stent — LVIS, Neuroform Atlas, Enterprise — is deployed across the neck to serve as a scaffold, requiring dual antiplatelet therapy for 3-6 months; generally avoided in acute SAH due to antiplatelet requirement), and flow diversion (Pipeline Embolization Device, FRED, Surpass Streamline — a densely braided stent placed across the aneurysm neck that redirects flow and promotes progressive thrombosis over weeks to months; best for large/giant, wide-necked, or fusiform aneurysms; requires DAPT for 6-12 months). The ISAT trial showed better outcomes with coiling vs clipping for ruptured aneurysms suitable for either treatment at 1 year (PMID: 12414200). Long-term follow-up showed higher retreatment rates with coiling (~17% vs ~4% for clipping) due to coil compaction and recanalization.
Cerebral AVM Embolization
Cerebral arteriovenous malformations (AVMs) are classified by the Spetzler-Martin grading system (based on size, eloquence of adjacent brain, and venous drainage pattern; Grade I-V). Embolization is used as: (1) a standalone treatment for small, deep AVMs not amenable to surgery or radiosurgery; (2) a pre-surgical adjunct to reduce AVM size, obliterate deep feeders, and reduce intraoperative blood loss; or (3) a pre-radiosurgery treatment to reduce AVM volume. Embolic agents: Onyx (EVOH — currently the most commonly used; allows slow, controlled injection) and NBCA glue (faster polymerization, used for high-flow fistulous components). The ARUBA trial (2014, PMID: 24523846) showed superiority of medical management over any interventional therapy for unruptured AVMs at 33 months, but this remains controversial due to short follow-up and methodological concerns.
Carotid Artery Stenting (CAS)
CAS is an alternative to carotid endarterectomy (CEA) for extracranial carotid stenosis. Current guidelines favor CAS in: high surgical risk patients (neck radiation, prior CEA, contralateral occlusion, high cervical or intrathoracic lesion), symptomatic stenosis ≥ 50%, and asymptomatic stenosis ≥ 60-80% in selected patients. The CREST trial showed equivalent outcomes for CAS and CEA in combined stroke/MI/death endpoint, though CAS had a higher periprocedural stroke rate and CEA a higher periprocedural MI rate (PMID: 20505173). Embolic protection devices (EPD) are used in nearly all CAS procedures: distal filter (FilterWire, SpiderFX) or proximal flow reversal (GORE NPS, MoMa — may be superior for reducing periprocedural embolic events). Self-expanding nitinol stents (Precise, Acculink, Wallstent) are standard. Post-stenting dual antiplatelet therapy (aspirin + clopidogrel) for ≥ 30 days. Carotid body manipulation during CAS can cause hemodynamic depression (bradycardia, hypotension) — atropine 0.5-1 mg IV should be readily available.
Intracranial Atherosclerotic Disease
Intracranial stenosis accounts for ~8-10% of all ischemic strokes. The SAMMPRIS trial (2011) showed superiority of aggressive medical management (dual antiplatelet therapy + statin + lifestyle modification) over intracranial stenting (Wingspan stent) for symptomatic intracranial stenosis ≥ 70% (PMID: 21899409). As a result, intracranial stenting is reserved for patients who fail maximal medical therapy with recurrent strokes. Balloon-mounted stents and drug-eluting stents are used off-label in select cases.
23 Vascular & Access Complications
Access-Site Complications
Hematoma: Most common access complication (~3-5% for diagnostic, up to 10% for interventional). Management: manual compression, bed rest, serial hemoglobin monitoring. Large or expanding hematomas may require surgical evacuation or covered stent placement.
Pseudoaneurysm: Contained rupture through the arteriotomy with a communicating neck to the vessel. Identified by US: characteristic "yin-yang" swirling flow pattern with a "to-and-fro" spectral Doppler waveform in the neck. Management: US-guided compression (success rate ~60-90% for small pseudoaneurysms, lower if on anticoagulation); US-guided thrombin injection (0.5-1 mL of 1,000 IU/mL thrombin injected into the pseudoaneurysm sac under US visualization — success rate > 95%, first-line treatment for most pseudoaneurysms); covered stent for large or complex pseudoaneurysms or those involving the artery itself; and surgical repair for failed percutaneous treatment.
Arteriovenous fistula (AVF): Communication between the artery and adjacent vein at the puncture site. Continuous low-pitched bruit and thrill. Small AVFs may close spontaneously; persistent or hemodynamically significant AVFs require covered stent or surgical repair.
Retroperitoneal hemorrhage: Occurs with high puncture above the inguinal ligament (external iliac artery). Presents with flank/back pain, tachycardia, hypotension, and falling hemoglobin. CT confirms the diagnosis. Management: resuscitation, reversal of anticoagulation, covered stent or balloon tamponade of the arteriotomy, and surgical repair if endovascular management fails.
Intraprocedural Vascular Complications
Arterial dissection: Intimal flap caused by wire or catheter manipulation. Classification: flow-limiting vs non-flow-limiting. Management: non-flow-limiting dissections are often observed; flow-limiting dissections require angioplasty or stenting to restore flow. Prevention: advance wires and catheters under fluoroscopy, never force advancement.
Vessel perforation/rupture: Penetration of the vessel wall by a wire, catheter, or device. Signs: extravasation of contrast on angiography. Management: immediate balloon tamponade (inflate a balloon across the perforation to achieve temporary hemostasis), covered stent placement for arterial perforations, coil embolization for branch vessel perforations, and emergent surgery if endovascular management fails.
Distal embolization: Migration of thrombus or atherosclerotic debris to distal vessels during angioplasty/stenting. Prevention: embolic protection devices, careful technique. Treatment: aspiration thrombectomy, catheter-directed thrombolysis, mechanical thrombectomy. The "trash foot" (blue toe syndrome / atheroembolism) presents with painful, cyanotic toes with palpable pedal pulses — it is caused by cholesterol crystal embolization from proximal atherosclerotic plaque and may occur spontaneously, after catheter manipulation, or after anticoagulation initiation.
Air embolism: Inadvertent introduction of air through catheters or sheaths. Arterial air embolism can cause stroke or limb ischemia; venous air embolism can cause cardiovascular collapse if large volume (> 3-5 mL/kg). Prevention: careful catheter/wire flushing, use of hemostatic valves on sheaths, aspiration before injection. Treatment: Trendelenburg (left lateral decubitus for venous air embolism), 100% oxygen (reduces nitrogen content of air bubble), aspiration of air through catheter if possible, hyperbaric oxygen for severe cases.
Catheter/wire fracture: Retained foreign body in the vascular system. Can cause thrombosis, perforation, or arrhythmia. Retrieval: percutaneous snare retrieval (goose-neck snare or En Snare) via femoral or jugular access. Multiple retrieval techniques may be needed (snare, basket, forceps). Prevention: never advance a kinked wire through a catheter; never withdraw a wire through a needle (the needle tip can shear the wire).
Contrast-Induced Nephropathy
Rise in serum creatinine ≥ 0.3 mg/dL or ≥ 50% within 48-72 hours post-contrast. Risk factors: CKD (eGFR < 30 is highest risk), diabetes, heart failure, dehydration, large contrast volume. Prevention: pre- and post-procedure IV hydration (NS 1 mL/kg/hr × 12 hours), minimize contrast volume, use iso-osmolar contrast in high-risk patients, hold metformin for 48 hours post-contrast in patients with eGFR < 30 (risk of lactic acidosis if AKI develops). See Section 4 for detailed prevention protocols.
24 Procedure-Specific Complications
Post-Embolization Syndrome
Post-embolization syndrome (PES) occurs after virtually all embolization procedures (TACE, UAE, splenic embolization, renal embolization) and is the expected physiologic response to tissue ischemia and necrosis. The classic triad: pain (at the embolized organ — RUQ for liver, pelvic/lower abdominal for uterine, LUQ for splenic), fever (low-grade, typically < 38.5°C — caused by cytokine release from necrotic tissue, NOT infection in the acute setting), and nausea/vomiting. Onset: within hours of the procedure. Duration: 3-7 days for most procedures, up to 2 weeks for large-volume embolizations.
Management: supportive care with IV hydration (1-2 L NS over 12-24 hours), analgesics (ketorolac 15-30 mg IV q6h or PCA with morphine/hydromorphone for severe pain, transitioning to oral analgesics), antiemetics (ondansetron 4 mg IV q6-8h), and antipyretics (acetaminophen 650-1000 mg PO/IV q6h). Most patients with PES after TACE can be managed with 23-hour observation and discharged the following day. Distinguish PES from infection: persistent fever > 7 days, fever > 38.5°C or rising, leukocytosis (WBC > 15,000) that does not trend down, and imaging findings suggesting abscess (gas in the collection, rim enhancement, increasing size) all warrant investigation with aspiration and culture. Patients with bilioenteric anastomosis (prior Whipple, hepaticojejunostomy) have a very high rate of hepatic abscess post-embolization (> 50%) due to reflux of enteric bacteria into the biliary system — these patients require extended prophylactic antibiotic courses and close surveillance.
Pneumothorax
Complication of lung biopsy (15-25%), thoracentesis, chest tube placement, subclavian vein access, and vertebroplasty (rare). Management: small (< 2 cm), asymptomatic, stable pneumothorax — observation with serial CXR; symptomatic or large pneumothorax — aspiration catheter (8-14 Fr pigtail) placement, connected to a Heimlich valve or water seal. Tension pneumothorax: emergent decompression (large-bore needle at 2nd intercostal space, midclavicular line, followed by chest tube).
Bile Leak & Biloma
Complication of hepatic biopsy, percutaneous liver ablation (especially MWA of central tumors adjacent to major bile ducts), biliary drainage/manipulation, cholecystectomy, and TACE. Presents with RUQ pain, fever, leukocytosis, and sometimes bilious drainage from a percutaneous drain. Diagnosis: CT showing perihepatic or subhepatic fluid collection (bilomas are typically well-defined, low-density collections; may have a thin enhancing rim); HIDA (hepatobiliary iminodiacetic acid) scan confirms active bile leak by showing radiotracer accumulation outside the biliary tree; aspiration of the fluid with elevated bilirubin level confirms bilious nature. Management: (1) percutaneous catheter drainage of the biloma to control sepsis and prevent bile peritonitis; (2) reduction of intrabiliary pressure to promote leak closure — achieved by biliary drainage (PTC/internal-external drain to decompress the proximal biliary system) or ERCP with sphincterotomy and/or biliary stent placement (which reduces the pressure gradient driving bile through the leak); (3) small bile leaks often seal within 1-2 weeks with adequate decompression; persistent leaks may require covered metallic stent placement across the defect or surgical repair.
Non-Target Embolization
Inadvertent embolization of normal tissue is one of the most important preventable complications in IR. Examples: non-target liver parenchyma embolization during TACE (causes hepatic infarction, bile duct necrosis, or liver abscess), non-target gastric embolization during TACE (causes gastric ulceration, which can be severe and require surgical repair — from embolization through the right gastric artery, left gastric artery, or falciform artery), gallbladder infarction (from cystic artery embolization during hepatic embolization — usually self-limited but can cause acute cholecystitis requiring cholecystostomy or cholecystectomy), skin necrosis after uterine fibroid embolization (from anastomoses between uterine and cutaneous/vulvar arterial supply), and pulmonary embolism from passage of embolic material through intrahepatic or intrapulmonary AV shunts (particularly with liquid embolics or small particles).
Prevention of non-target embolization: (1) superselective catheter positioning (use microcatheters to access the tumor-feeding artery as distally as possible, beyond the takeoff of non-target branches); (2) thorough mapping angiography before treatment (DSA and cone-beam CT to identify all feeding vessels and potential collateral pathways to non-target territories); (3) protective coil embolization of non-target vessels before delivering embolic agents (e.g., coiling the GDA, right gastric artery, cystic artery, falciform artery, and left/right inferior phrenic arteries before TACE/TARE); (4) use of appropriately sized embolic agents (particles that are too small may pass through the capillary bed into the venous system; particles that are too large may reflux into non-target territories); (5) careful injection technique (slow, controlled injection under continuous fluoroscopy, stopping immediately if flow stasis is observed or if reflux of embolic material is seen); and (6) use of anti-reflux catheters (Surefire, Occlusafe) that have a deployable tip element that collapses under forward flow but expands under reflux, preventing retrograde escape of embolic material.
Spinal Cord Ischemia
A feared complication of aortic interventions (EVAR, TEVAR), bronchial artery embolization, and spinal procedures. The artery of Adamkiewicz (arteria radicularis magna) arises from the aorta between T8 and L2 (left side in ~75%) and is the major blood supply to the anterior spinal cord via the anterior spinal artery. Coverage or embolization of this artery can cause anterior spinal artery syndrome (paraplegia, loss of pain/temperature sensation, preserved proprioception/vibration — because the posterior columns are supplied by the posterior spinal arteries). The spinal cord blood supply has three sources: the anterior spinal artery (single, supplies the anterior 2/3 of the cord), two posterior spinal arteries (supply the posterior 1/3), and segmental radiculomedullary arteries (from the aorta, including the artery of Adamkiewicz).
Prevention of spinal cord ischemia: preoperative identification of the artery of Adamkiewicz on CTA/MRA, prophylactic CSF drainage (lumbar drain placed preoperatively, maintain CSF pressure < 10 mmHg, drain up to 15 mL/hr), maintain MAP > 80-90 mmHg (augment with vasopressors if needed), staged aortic coverage when possible (> 20 cm of aortic coverage significantly increases risk), avoid simultaneous coverage of the left subclavian artery and hypogastric arteries (both contribute collateral supply). Delayed spinal cord ischemia (hours to days post-procedure) may respond to aggressive MAP augmentation and CSF drainage.
Contrast Extravasation & Allergoid Reactions to Embolic Agents
Non-target contrast extravasation: During high-pressure injections (power injections through small catheters or into vessels with downstream occlusion), contrast can extravasate into the perivascular tissues. Usually self-limited; large-volume extravasation can cause compartment syndrome (especially in the extremities). Warm compresses and elevation are first-line treatment; surgical consultation for compartment syndrome. DMSO reactions (Onyx): DMSO solvent causes a garlic-like taste and odor; in rare cases, vasospasm, chest tightness, or pulmonary edema can occur. Inject Onyx slowly (< 0.3 mL/min initially) and monitor closely during the first few minutes of injection.
25 Post-Procedure Management
Anticoagulation Bridging for IR Procedures
The SIR classifies procedures by bleeding risk for anticoagulation management:
| Category | Examples | INR Target | Platelet Target |
|---|---|---|---|
| Category 1 (low risk) | Venous access (PICC, port, tunneled CVC), IVC filter, thoracentesis, paracentesis, superficial biopsy | ≤ 2.0-3.0 (usually no hold) | ≥ 20,000 |
| Category 2 (moderate risk) | Intra-abdominal/pelvic drainage, lung biopsy, liver biopsy, angioplasty/stenting | ≤ 1.5 | ≥ 50,000 |
| Category 3 (high risk) | Renal biopsy, TIPS, liver ablation, complex embolization, EVAR/TEVAR, nephrostomy | ≤ 1.5 | ≥ 50,000 |
For patients on warfarin undergoing Category 2/3 procedures: hold warfarin 5 days prior; check INR on the day of the procedure (≤ 1.5 for Category 2/3). Bridging with LMWH (enoxaparin 1 mg/kg SC q12h) or UFH infusion is reserved for high thromboembolic risk patients (mechanical heart valve — especially mitral; recent VTE within 3 months; high-risk atrial fibrillation with prior stroke/TIA). The BRIDGE trial (PMID: 26559317) showed that forgoing bridging was non-inferior to bridging for perioperative arterial thromboembolism and was associated with less major bleeding in AF patients undergoing elective procedures.
DOACs: hold for 2-3 half-lives — rivaroxaban (Xarelto): hold 48 hours; apixaban (Eliquis): hold 48 hours; dabigatran (Pradaxa): hold 48-72 hours (hold ≥ 96 hours if eGFR < 50 due to renal clearance). Dabigatran has a specific reversal agent: idarucizumab (Praxbind) 5 g IV. Factor Xa inhibitor reversal: andexanet alfa (Andexxa) for life-threatening bleeding associated with rivaroxaban or apixaban; 4-factor PCC (Kcentra) 25-50 IU/kg is an alternative when andexanet is unavailable. Antiplatelet agents: hold clopidogrel 5 days, prasugrel 7 days; aspirin is generally continued for most procedures (SIR guidelines). For patients with recent coronary stents (< 6 months BMS, < 12 months DES), consult cardiology before holding any antiplatelet — premature discontinuation risks stent thrombosis.
Post-Procedure Observation
Arterial access: Bed rest duration depends on sheath size and closure method: manual compression of 4-6 Fr access — 4-6 hours bed rest; closure device for 6-8 Fr access — 1-2 hours bed rest. Monitor access site (hematoma, expanding mass, bruit) and distal pulses (dorsalis pedis, posterior tibial — compare to pre-procedure baseline) q15min × 1 hour, q30min × 2 hours, then q1h until ambulation. Any new pulse deficit, expanding hematoma, or hemodynamic instability requires immediate evaluation (ultrasound of access site, CTA if retroperitoneal bleed suspected).
Sedation recovery: Monitor level of consciousness (Ramsay or Aldrete score), vital signs, oxygen saturation per post-anesthesia care standards. Discharge criteria (Aldrete score ≥ 9): stable vital signs for 30 minutes, ambulatory without assistance (or at pre-procedure baseline), tolerating oral fluids, pain controlled with oral medications, no active bleeding from access site, adequate oxygen saturation on room air, and a responsible adult to accompany the patient home. Written discharge instructions should include: signs of access-site complications (swelling, numbness, bleeding, fever), activity restrictions (no heavy lifting > 10 lbs for 48-72 hours, no driving for 24 hours after sedation), and contact information for emergencies.
Drain Management Protocols
Biliary drains: Record output (color, volume) daily. Flush with 10 mL sterile NS q8-12h to maintain patency. External drains should be capped and transitioned to internal-external drainage as soon as the obstruction can be crossed. Cholangiogram at 4-6 weeks to assess duct patency and plan stent placement or drain removal. Normal biliary output: 200-800 mL/day; sudden increase suggests obstruction of the internal limb; sudden decrease suggests catheter migration or occlusion.
Nephrostomy: Record output daily (normally matches contralateral kidney output, ~500-1500 mL/day for a single kidney). Flush 5 mL sterile NS q12h gently (never force flush — risk of pyelovenous backflow and sepsis). Nephrostogram at 1-2 weeks to assess obstruction relief and plan ureteral stenting. Urine should progressively clear; persistent gross hematuria beyond 48 hours warrants evaluation.
Abscess drains: Record output (volume, color, character) daily. Flush 10 mL NS q8-12h. Follow-up imaging (CT preferred) when output decreases to < 10-20 mL/day to confirm cavity collapse. Persistent high output despite imaging showing a small residual cavity suggests an ongoing communication (fistula to bowel, biliary system, or pancreatic duct).
General principle: Do not remove a drain until output is minimal (< 10 mL/day for abscesses, < 50 mL/day for biliary drains before capping trial), follow-up imaging shows resolution, the patient is clinically improved (afebrile, WBC normalizing), and the underlying etiology has been addressed.
IR Clinic Follow-Up
Standard follow-up: 1-2 weeks post-procedure for wound check, drain assessment, and review of pathology (biopsy). TACE/TARE: contrast-enhanced imaging at 4-6 weeks (MRI preferred for mRECIST assessment). EVAR/TEVAR: CTA at 1 month, 6 months, 12 months, then annually (or contrast-enhanced US as an alternative to reduce cumulative radiation). IVC filter: schedule retrieval within 30-60 days if indication resolves. Ablation: contrast-enhanced imaging at 4-6 weeks to assess completeness (MRI preferred for liver, CT for lung/renal). Maintain a formal IR clinic with structured follow-up protocols for all post-procedure patients.
Informed Consent & Pre-Procedure Checklist
Every IR procedure requires documented informed consent addressing: indication, alternatives (including no treatment), procedure description, expected outcomes, risks (including procedure-specific complications), and sedation plan. Pre-procedure checklist: verify patient identity, correct procedure and side (laterality), relevant imaging available, labs reviewed (INR, platelets, creatinine/eGFR), anticoagulation management, allergy status (contrast premedication if needed), NPO status, IV access, pregnancy test (women of childbearing age for radiation-exposure procedures), and prophylactic antibiotics administered if indicated. A pre-procedure "time-out" is mandatory per Joint Commission Universal Protocol.
Radiation Dose Tracking
All IR operators should maintain cumulative radiation dose records for patients undergoing multiple fluoroscopic procedures. The National Council on Radiation Protection (NCRP) recommends: occupational whole-body effective dose limit of 50 mSv/year (with 10-year average ≤ 20 mSv/year); lens of the eye 150 mSv/year; extremities 500 mSv/year. Pregnant operators: fetal dose limit of 0.5 mSv/month. Patients who receive > 5 Gy reference air kerma should be notified and have clinical follow-up for skin injury. Dose-reduction strategies: use lowest acceptable frame rate, collimate to the region of interest, minimize magnification, use last-image hold and stored fluoroscopy, position the image receptor close to the patient, and use copper filtration.
26 Classification Systems
SIR Adverse Event Classification
Minor Complications:
A: No therapy, no consequence.
B: Nominal therapy, no consequence; includes overnight admission for observation only.
Major Complications:
C: Requires therapy, minor hospitalization (< 48 hours).
D: Requires major therapy, unplanned increase in level of care, prolonged hospitalization (> 48 hours).
E: Permanent adverse sequelae.
F: Death.
Endoleak Classification
See Section 9 for the full endoleak classification (Types I-V). Type I and III are high-pressure leaks requiring urgent treatment. Type II is the most common and is usually benign unless associated with sac expansion > 5 mm.
BCLC Staging for HCC
Stage 0 (Very early): Single ≤ 2 cm, preserved liver function (Child-Pugh A), PS 0. Treatment: resection, ablation, or transplant.
Stage A (Early): Single or 2-3 nodules ≤ 3 cm, Child-Pugh A-B, PS 0. Treatment: resection, ablation, or transplant (within Milan criteria).
Stage B (Intermediate): Multinodular, Child-Pugh A-B, PS 0, no vascular invasion or extrahepatic spread. Treatment: TACE.
Stage C (Advanced): Portal vein invasion, extrahepatic spread, or PS 1-2. Treatment: systemic therapy (atezolizumab + bevacizumab first-line, lenvatinib, sorafenib).
Stage D (Terminal): Child-Pugh C (not transplant candidate) or PS 3-4. Treatment: best supportive care.
Child-Pugh Classification
| Parameter | 1 Point | 2 Points | 3 Points |
|---|---|---|---|
| Bilirubin (mg/dL) | < 2 | 2-3 | > 3 |
| Albumin (g/dL) | > 3.5 | 2.8-3.5 | < 2.8 |
| INR | < 1.7 | 1.7-2.3 | > 2.3 |
| Ascites | None | Mild/controlled | Moderate-severe |
| Encephalopathy | None | Grade 1-2 | Grade 3-4 |
Class A: 5-6 points. Class B: 7-9 points. Class C: 10-15 points. Child-Pugh class is critical for determining candidacy for liver-directed therapy — TACE and TARE are generally limited to Child-Pugh A and select B7 patients.
MELD Score
Model for End-Stage Liver Disease (MELD) = 3.78 × ln(bilirubin) + 11.2 × ln(INR) + 9.57 × ln(creatinine) + 6.43. Range: 6-40. Used for liver transplant allocation and predicting 90-day mortality in cirrhosis. MELD-Na (incorporating serum sodium) replaced MELD for transplant allocation in 2016. TIPS is relatively contraindicated with MELD > 18 (30-day mortality increases significantly).
NASCET Carotid Stenosis Measurement
NASCET method: % stenosis = (1 - [minimum residual lumen diameter / normal distal ICA diameter]) × 100. The distal ICA (beyond the bulb, where the walls become parallel) is used as the reference. ECST method: Uses the estimated original bulb diameter as the reference — produces higher stenosis percentages than NASCET for the same lesion. All major North American trials (NASCET, ACAS, CREST) use the NASCET method. Conversion: approximately ECST% = 0.6 × NASCET% + 40%. A 70% NASCET stenosis corresponds to approximately an 82% ECST stenosis.
Rutherford Classification of Chronic Limb Ischemia
| Category | Grade | Clinical Description |
|---|---|---|
| 0 | 0 | Asymptomatic — no hemodynamically significant disease |
| 1 | I | Mild claudication |
| 2 | I | Moderate claudication |
| 3 | I | Severe claudication |
| 4 | II | Ischemic rest pain |
| 5 | III | Minor tissue loss — nonhealing ulcer, focal gangrene with diffuse pedal ischemia |
| 6 | III | Major tissue loss — extending above the transmetatarsal level, functional foot no longer salvageable |
Spleen Injury Scale (AAST)
| Grade | Description | IR Relevance |
|---|---|---|
| I | Subcapsular hematoma < 10% surface area; capsular laceration < 1 cm depth | Observation |
| II | Subcapsular hematoma 10-50%; intraparenchymal hematoma < 5 cm; laceration 1-3 cm depth | Observation; consider angiography if active contrast extravasation on CT |
| III | Subcapsular hematoma > 50% or expanding; ruptured subcapsular/parenchymal hematoma; laceration > 3 cm depth | Embolization (proximal or selective) in hemodynamically stable patients |
| IV | Laceration involving segmental or hilar vessels with major devascularization (> 25%) | Embolization or surgery based on hemodynamic status |
| V | Completely shattered spleen; hilar vascular injury devascularizing the spleen | Surgery (splenectomy); embolization as adjunct in select cases |
Pulmonary AVM — Hereditary Hemorrhagic Telangiectasia
Pulmonary arteriovenous malformations (PAVMs) are abnormal communications between pulmonary arteries and veins, bypassing the capillary bed. Most (> 80%) are associated with hereditary hemorrhagic telangiectasia (HHT / Osler-Weber-Rendu syndrome), an autosomal dominant disorder. Risks of untreated PAVMs: paradoxical embolism (stroke, brain abscess — occur in ~30% of untreated patients), hemoptysis, and hypoxemia. Treatment threshold: feeding artery diameter ≥ 3 mm (some advocate treating all PAVMs identified on CT regardless of size). Technique: pulmonary angiography via femoral vein, catheterize the feeding artery, embolize with Amplatzer vascular plugs or detachable coils. Avoid using particles (risk of paradoxical embolism through the AVM). Follow-up CT at 6-12 months to confirm involution and screen for new or enlarging PAVMs. HHT patients require lifetime surveillance.
TASC II Classification (Aortoiliac)
| Class | Lesion Description | Preferred Treatment |
|---|---|---|
| A | Unilateral or bilateral CIA stenosis ≤ 3 cm; unilateral or bilateral EIA stenosis ≤ 3 cm | Endovascular |
| B | CIA stenosis 3-10 cm not extending into CFA; unilateral EIA occlusion not involving CFA or IIA | Endovascular |
| C | Bilateral EIA stenosis 5-10 cm not extending into CFA; unilateral EIA occlusion extending into CFA/IIA; heavily calcified unilateral EIA occlusion | Individual decision |
| D | Infrarenal aortoiliac occlusion; diffuse disease involving the aorta and both iliac arteries; bilateral EIA occlusions; iliac stenosis associated with AAA or other aortic pathology | Open surgical |
TASC II Classification (Femoropopliteal)
| Class | Lesion Description | Preferred Treatment |
|---|---|---|
| A | Single stenosis ≤ 10 cm; single occlusion ≤ 5 cm | Endovascular |
| B | Multiple stenoses/occlusions each ≤ 5 cm; single stenosis/occlusion ≤ 15 cm not involving the popliteal artery; heavily calcified single occlusion ≤ 5 cm | Endovascular |
| C | Multiple stenoses/occlusions > 15 cm total (with or without heavy calcification); recurrent stenoses/occlusions after two endovascular interventions | Individual decision |
| D | CTO of CFA or SFA > 20 cm involving the popliteal artery; CTO of popliteal artery involving the trifurcation | Open surgical |
Stanford Aortic Dissection Classification
Type A: Any dissection involving the ascending aorta (regardless of site of origin). Treatment: emergent open surgical repair. Mortality without surgery: ~1-2% per hour for the first 48 hours.
Type B: Dissection confined to the descending aorta (distal to the left subclavian artery). Treatment of uncomplicated type B: medical management (blood pressure control, target SBP 100-120 mmHg with IV beta-blockers). Treatment of complicated type B (malperfusion, rupture, refractory pain, rapid aortic expansion): TEVAR. The INSTEAD-XL trial showed improved 5-year outcomes with TEVAR for subacute uncomplicated type B dissection (PMID: 23425526).
DeBakey Aortic Dissection Classification
Type I: Originates in the ascending aorta, extends to at least the aortic arch and often to the descending aorta. Type II: Originates in and is confined to the ascending aorta. Type III: Originates in the descending aorta; IIIa extends only in the thoracic aorta, IIIb extends below the diaphragm. Stanford A = DeBakey I or II. Stanford B = DeBakey III.
27 Medications Master Table
Contrast Agents
| Agent | Type | Osmolality | Common Use |
|---|---|---|---|
| Iohexol (Omnipaque) | Non-ionic monomer (LOCM) | ~670-840 mOsm/kg | Standard IR angiography |
| Iopamidol (Isovue) | Non-ionic monomer (LOCM) | ~616-796 mOsm/kg | Angiography, CT enhancement |
| Iodixanol (Visipaque) | Non-ionic dimer (IOCM) | ~290 mOsm/kg | High-risk patients (CKD) |
| CO2 | Negative contrast | N/A | Contrast allergy, CKD (below diaphragm only) |
| Ethiodized oil (Lipiodol) | Iodinated oil | N/A | TACE (drug carrier), lymphangiography |
Thrombolytic Agents
| Agent | Mechanism | CDT Dose | Notes |
|---|---|---|---|
| Alteplase (tPA) | Plasminogen activator | 0.5-1.0 mg/hr (arterial), 1 mg/hr (venous/PE) | Most commonly used; half-life ~4 min |
| Reteplase | Recombinant plasminogen activator | 0.25-0.5 units/hr | Longer half-life (~15 min), less fibrin specificity |
| Tenecteplase | Modified tPA | Single bolus dosing (IV for stroke/PE) | Not typically used for CDT; used IV for stroke and PE |
Sedation & Analgesia
| Agent | Class | Dose (IV) | Reversal Agent |
|---|---|---|---|
| Midazolam | Benzodiazepine (anxiolytic/amnestic) | 0.5-2 mg, titrate | Flumazenil 0.2 mg q1min (max 1 mg) |
| Fentanyl | Opioid (analgesic) | 25-100 mcg, titrate | Naloxone 0.04-0.4 mg q2-3min |
| Propofol | General anesthetic (MAC) | 25-75 mcg/kg/min infusion | None specific (supportive care) |
| Dexmedetomidine | Alpha-2 agonist (sedative/analgesic) | 0.2-0.7 mcg/kg/hr | None specific |
Anticoagulants Used in IR
| Agent | Mechanism | IR Dose / Indication | Monitoring / Reversal |
|---|---|---|---|
| Unfractionated heparin (UFH) | Antithrombin III activation | Intraprocedural: 3,000-5,000 IU bolus; CDT: 500 IU/hr | aPTT; reversal: protamine 1 mg per 100 IU heparin |
| Enoxaparin (LMWH) | Factor Xa > IIa inhibition | Bridging: 1 mg/kg q12h SC | Anti-Xa level; partial reversal with protamine |
| Bivalirudin | Direct thrombin inhibitor | 0.75 mg/kg bolus + 1.75 mg/kg/hr (HIT patients) | ACT; no specific reversal (short half-life 25 min) |
Embolic Agents Summary
| Agent | Type | Duration | Key Applications |
|---|---|---|---|
| Metallic coils | Mechanical | Permanent | Vessel occlusion, pseudoaneurysm, organ devascularization |
| Gelfoam | Particulate (gelatin) | Temporary (2-6 weeks) | Trauma, GI bleed, preop devascularization |
| PVA particles | Particulate (polymer) | Permanent | UAE, tumor devascularization |
| Microspheres (Embosphere) | Calibrated particles | Permanent | UAE, TACE (drug-eluting), HCC |
| NBCA (glue) | Liquid embolic | Permanent | AVMs, emergent bleed, varicocele |
| Onyx (EVOH) | Liquid embolic | Permanent | Cerebral AVMs, dural AVFs |
| Amplatzer Vascular Plug | Mechanical device | Permanent | Large-vessel occlusion (IIA, PVE, PAVM) |
| Ethanol (absolute) | Sclerosant/liquid embolic | Permanent | Venous malformations, renal ablation |
28 Imaging Protocols
CT Angiography (CTA)
CTA of the aorta and runoff: Multidetector CT with iodinated contrast (100-120 mL at 4-5 mL/s), bolus tracking with ROI in the abdominal aorta (trigger at 150-200 HU). Arterial phase acquisition from the diaphragm to the feet (1-2 mm slice thickness). Delayed phase may be added for endoleak detection (EVAR surveillance). CTA of the chest for PE: ROI in the main pulmonary artery (trigger at 100-120 HU), acquisition from the aortic arch to below the diaphragm. ECG-gated CTA for aortic root evaluation (TAVR planning, aortic dissection).
MR Angiography (MRA)
Contrast-enhanced MRA: Gadolinium-based contrast agent (0.1-0.2 mmol/kg) with high-temporal-resolution acquisition. Provides excellent vascular imaging without radiation. Time-resolved MRA (e.g., TWIST, TRICKS) allows sequential imaging of arterial, parenchymal, and venous phases — useful for hepatic tumor evaluation and AVM characterization. Non-contrast MRA techniques: time-of-flight (TOF) for intracranial vessels, phase-contrast for flow quantification, and quiescent-interval single-shot (QISS) for peripheral arteries in patients with CKD.
Hepatic MRI for IR Planning
Liver MRI with hepatocyte-specific contrast (gadoxetate/Eovist) is the gold standard for pre-procedural evaluation of hepatic tumors. The LI-RADS (Liver Imaging Reporting and Data System) classification standardizes reporting of liver observations in patients at risk for HCC: LR-1 (definitely benign), LR-2 (probably benign), LR-3 (intermediate probability), LR-4 (probably HCC), LR-5 (definitely HCC — requires arterial phase hyperenhancement AND nonperipheral washout AND/OR enhancing capsule), and LR-M (probably or definitely malignant but not specific for HCC). LR-5 observations can be treated without biopsy. Pre-TACE/TARE MRI should also include assessment of tumor vascularity, portal vein patency, biliary anatomy, and the relationship of the tumor to major hepatic veins.
Digital Subtraction Angiography (DSA)
DSA remains the gold standard for vascular imaging in IR. A mask image is acquired before contrast injection; subsequent contrast-enhanced images are subtracted from the mask, eliminating bone and soft tissue to display only the contrast-filled vessels. Patient motion between the mask and contrast images causes misregistration artifacts — minimize by instructing the patient to hold still and suspend breathing during runs. Frame rates: 2-6 frames/second for most applications, higher (7.5-15 fps) for cerebrovascular imaging. Power injection rates and contrast volumes depend on the vessel:
| Vessel | Injection Rate (mL/s) | Total Volume (mL) | Catheter Position |
|---|---|---|---|
| Abdominal aorta | 15-25 | 30-40 | Pigtail at L1 |
| Pelvic aortogram | 10-15 | 20-30 | Pigtail at L4 bifurcation |
| Celiac trunk | 4-6 | 12-18 | Selective celiac |
| SMA | 4-6 | 12-18 | Selective SMA |
| Renal artery | 4-6 | 8-12 | Selective renal |
| Hepatic artery (selective) | 3-4 | 9-12 | Proper or right/left hepatic |
| Iliac artery | 4-6 | 10-15 | Common or external iliac |
| Femoral artery runoff | 3-5 | 10-15 | CFA or SFA |
| Pulmonary artery | 15-25 | 30-40 | Pigtail in main PA |
| Cerebral (ICA) | 4-6 | 8-10 | Cervical ICA |
Road mapping: a stored fluoroscopic image with contrast-filled vessels is overlaid on live fluoroscopy to guide catheter navigation in real time. This technique is essential for superselective catheterization and reduces the total contrast volume and radiation dose needed for navigation. Rotational angiography (3D spin) acquires images during C-arm rotation to provide 3D vascular reconstruction — useful for complex vascular anatomy (cerebral aneurysms, hepatic arterial variants).
Ultrasound Protocols for IR
Doppler evaluation of dialysis access: Gray-scale assessment of fistula/graft morphology (diameter, wall thickness, perivascular fluid/hematoma); color Doppler to identify stenosis (focal color aliasing — the "aliasing jet" sign indicates high-velocity flow through a stenosis); spectral Doppler to measure velocities — peak systolic velocity (PSV) ratio > 2:1 (PSV at the stenosis site compared to an adjacent normal segment) indicates > 50% stenosis. Inflow volume measured at the brachial artery (flow volume = vessel cross-sectional area × time-averaged mean velocity × 60 seconds) < 500 mL/min suggests access dysfunction. Normal flow: 600-1200 mL/min for a functioning AVF/AVG.
Hepatic Doppler for TIPS: Surveillance protocol performed at 2 weeks, then every 3-6 months. Key measurements: PSV in the TIPS stent at proximal, mid, and distal segments (normal 90-190 cm/s); PSV < 90 cm/s suggests stenosis (decreased flow through the stent); PSV > 190 cm/s also suggests stenosis (accelerated flow through a narrowed segment); direction of main portal vein flow (should convert from hepatopetal to hepatofugal or mixed after TIPS, depending on the pre-TIPS portal hemodynamics — reversal to hepatofugal flow through the stent confirms adequate decompression). Draining hepatic vein velocity should show continuous, non-pulsatile flow. A change in velocity of > 50 cm/s from baseline on serial exams suggests developing stenosis.
Duplex US for DVT: Non-compressibility of a venous segment is the primary diagnostic criterion (sensitivity ~95% for proximal DVT, lower for calf vein DVT). The exam systematically evaluates the common femoral vein, femoral vein (formerly superficial femoral vein), popliteal vein, and when indicated, the calf veins (posterior tibial and peroneal). Additional findings: echogenic intraluminal thrombus (chronic DVT shows hyperechoic, retracted thrombus with wall thickening; acute DVT is often hypoechoic or anechoic with a distended vein), loss of normal respiratory phasicity (suggests proximal obstruction — iliac vein or IVC thrombosis), and diminished augmentation response to calf compression (suggests intervening thrombus between the compression point and the probe). For upper extremity DVT: evaluate the subclavian, axillary, basilic, brachial, and internal jugular veins.
Contrast-enhanced ultrasound (CEUS): Uses microbubble contrast agents (sulfur hexafluoride — Lumason/SonoVue; perflutren — Definity) to provide real-time assessment of tissue perfusion without ionizing radiation or nephrotoxic contrast. Applications in IR: intraprocedural assessment of ablation completeness (residual enhancing tumor indicates incomplete ablation), evaluation of liver lesions (LI-RADS criteria now include CEUS), assessment of endoleak after EVAR, and characterization of indeterminate renal lesions. CEUS is gaining acceptance as a radiation-sparing alternative for surveillance after liver-directed therapy.
29 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| AAA | Abdominal aortic aneurysm |
| AVF | Arteriovenous fistula |
| AVG | Arteriovenous graft |
| AVM | Arteriovenous malformation |
| BAE | Bronchial artery embolization |
| BCLC | Barcelona Clinic Liver Cancer |
| BMS | Bare metal stent |
| BSA | Body surface area |
| CBCT | Cone-beam computed tomography |
| CDT | Catheter-directed thrombolysis |
| CFA | Common femoral artery |
| CHA | Common hepatic artery |
| CIA | Common iliac artery |
| CIN | Contrast-induced nephropathy |
| CLTI | Chronic limb-threatening ischemia |
| CO2 | Carbon dioxide (angiography) |
| CRLM | Colorectal liver metastases |
| CSF | Cerebrospinal fluid |
| CTA | Computed tomography angiography |
| CTO | Chronic total occlusion |
| CVC | Central venous catheter |
| DAP | Dose-area product |
| DCB | Drug-coated balloon |
| DEB | Drug-eluting bead |
| DMSO | Dimethyl sulfoxide |
| DOAC | Direct oral anticoagulant |
| DSA | Digital subtraction angiography |
| DVT | Deep vein thrombosis |
| EVAR | Endovascular aneurysm repair |
| EVT | Endovascular thrombectomy |
| FEVAR | Fenestrated endovascular aneurysm repair |
| FLR | Future liver remnant |
| FMD | Fibromuscular dysplasia |
| FNA | Fine-needle aspiration |
| Fr | French (catheter size; 1 Fr = 0.33 mm) |
| GDA | Gastroduodenal artery |
| GJ | Gastrojejunostomy |
| HCC | Hepatocellular carcinoma |
| HDE | Humanitarian device exemption |
| HVPG | Hepatic venous pressure gradient |
| IJV | Internal jugular vein |
| IMA | Inferior mesenteric artery |
| IMV | Inferior mesenteric vein |
| IOCM | Iso-osmolar contrast media |
| IVC | Inferior vena cava |
| IVUS | Intravascular ultrasound |
| KGR | Kinetic growth rate |
| LOCM | Low-osmolar contrast media |
| LSF | Lung shunt fraction |
| LVO | Large vessel occlusion |
| MAA | Macroaggregated albumin |
| MAP | Mean arterial pressure |
| MELD | Model for end-stage liver disease |
| MRA | Magnetic resonance angiography |
| mRECIST | Modified RECIST (response evaluation criteria) |
| MWA | Microwave ablation |
| NASCET | North American Symptomatic Carotid Endarterectomy Trial |
| NBCA | N-butyl cyanoacrylate |
| NSF | Nephrogenic systemic fibrosis |
| PAD | Peripheral arterial disease |
| PCN | Percutaneous nephrostomy |
| PCNL | Percutaneous nephrolithotomy |
| PE | Pulmonary embolism |
| PEG | Percutaneous endoscopic gastrostomy |
| PERT | Pulmonary embolism response team |
| PHLF | Post-hepatectomy liver failure |
| PICC | Peripherally inserted central catheter |
| PMMA | Polymethylmethacrylate |
| PRG | Percutaneous radiologic gastrostomy |
| PTA | Percutaneous transluminal angioplasty |
| PTC | Percutaneous transhepatic cholangiography |
| PVA | Polyvinyl alcohol |
| PVE | Portal vein embolization |
| RAS | Renal artery stenosis |
| RFA | Radiofrequency ablation |
| SFA | Superficial femoral artery |
| SIR | Society of Interventional Radiology |
| SMA | Superior mesenteric artery |
| SMV | Superior mesenteric vein |
| SRDL | Substantial radiation dose level |
| SVR | Systemic vascular resistance |
| TACE | Transarterial chemoembolization |
| TARE | Transarterial radioembolization |
| TASC | TransAtlantic Inter-Society Consensus |
| TDE | Thoracic duct embolization |
| TEVAR | Thoracic endovascular aortic repair |
| TIPS | Transjugular intrahepatic portosystemic shunt |
| tPA | Tissue plasminogen activator |
| UAE | Uterine artery embolization |
| UFH | Unfractionated heparin |
| VTE | Venous thromboembolism |
| Y-90 | Yttrium-90 |