Maternal-Fetal Medicine
Every high-risk obstetric condition, fetal diagnosis, ultrasound finding, genetic screening, procedure, classification, complication, medication, and management algorithm across the full scope of maternal-fetal medicine in one place.
01 Placental & Fetal Physiology
Placental Development
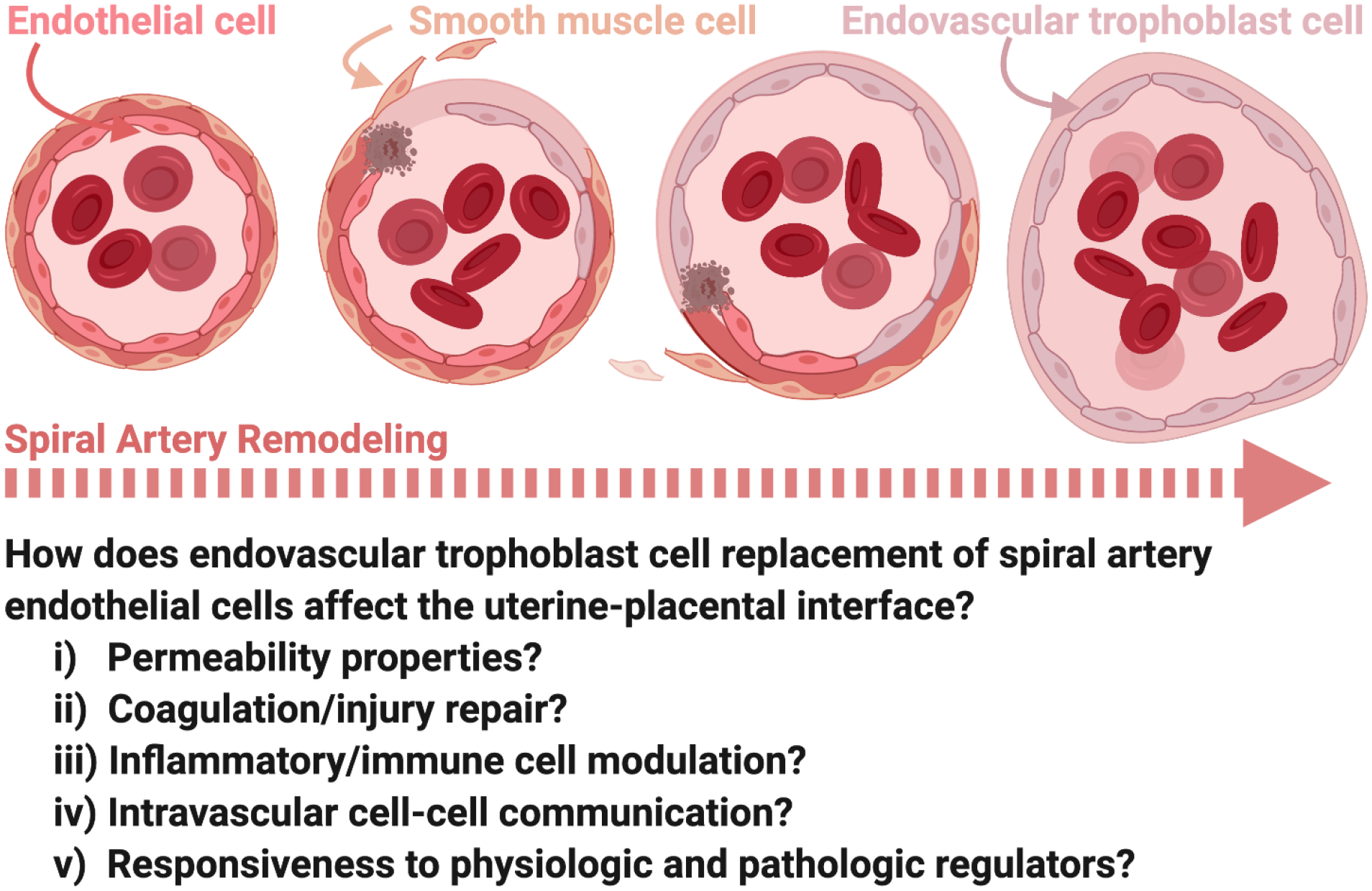
The placenta develops from the trophoblast, which differentiates into the outer syncytiotrophoblast (multinucleated, hormone-producing, in direct contact with maternal blood) and the inner cytotrophoblast (proliferative stem cells). Placentation involves two waves of trophoblast invasion: the first wave (6-10 weeks) invades the decidua, and the critical second wave (16-20 weeks) invades the myometrial segments of the spiral arteries, transforming them from high-resistance muscular vessels into low-resistance, high-capacitance channels. Failure of this second wave of trophoblast invasion is the central pathophysiology of preeclampsia and fetal growth restriction.
The mature placenta weighs ~500 g at term and contains approximately 150 mL of maternal blood in the intervillous space at any time. Maternal blood enters via 80-100 spiral arteries and bathes the chorionic villi (the functional units of gas and nutrient exchange). The placental barrier consists of syncytiotrophoblast, cytotrophoblast (diminishes after 20 weeks), villous connective tissue, and fetal capillary endothelium. By the third trimester, the barrier thins to ~3.5 μm, facilitating efficient diffusion.
Placental Gas Exchange
Oxygen transfer across the placenta occurs by simple diffusion down a partial pressure gradient. Maternal arterial PaO2 is ~100 mmHg, intervillous space PO2 is ~30-35 mmHg, and umbilical vein PO2 is ~30 mmHg. Despite this low PO2, the fetus maintains adequate oxygen delivery through several compensatory mechanisms: fetal hemoglobin (HbF) has a left-shifted oxygen-hemoglobin dissociation curve (higher oxygen affinity, P50 ~19 mmHg vs adult ~27 mmHg), fetal hemoglobin concentration is higher (16-18 g/dL), fetal cardiac output per kg is higher than adult, and the double Bohr effect (CO2 transfer from fetal to maternal blood shifts the fetal curve leftward and maternal curve rightward, facilitating bidirectional exchange).
Carbon dioxide crosses the placenta ~20 times faster than oxygen (high diffusibility), also by simple diffusion. Glucose crosses via facilitated diffusion (GLUT transporters). Amino acids are transported by active transport. IgG crosses by receptor-mediated endocytosis (pinocytosis), primarily in the third trimester — this provides passive immunity to the neonate. IgM and IgA do not cross the placenta (if IgM is detected in neonatal blood, it indicates fetal infection).
Fetal Circulation
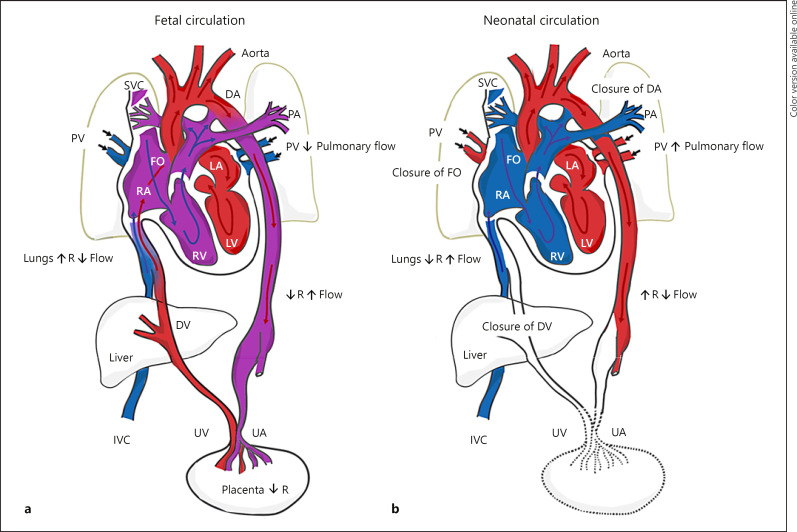
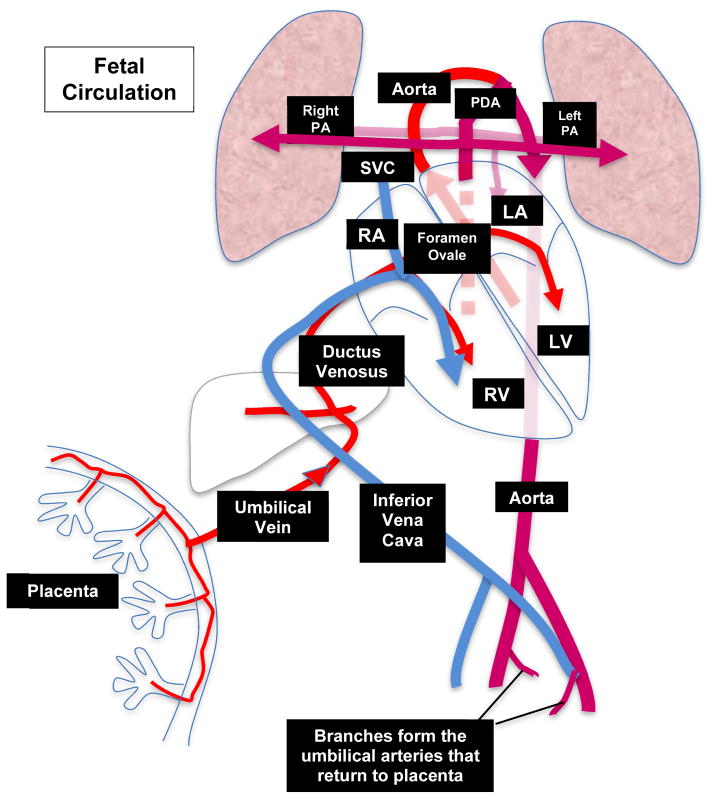
The fetal circulation contains three critical shunts that bypass the non-functioning lungs and liver:
Ductus venosus: Connects the umbilical vein to the IVC, bypassing the hepatic sinusoids. Carries well-oxygenated blood directly from the placenta to the right atrium. Constitutes ~50% of umbilical venous return. Abnormal Doppler waveforms (absent or reversed a-wave) indicate cardiac dysfunction and are an ominous sign in fetal growth restriction.
Foramen ovale: A right-to-left interatrial shunt that directs well-oxygenated blood (streaming from the IVC via the ductus venosus) from the right atrium to the left atrium and then to the left ventricle, ascending aorta, and the coronary and cerebral circulations. Preferential streaming of oxygenated blood through the foramen ovale ensures the brain and heart receive the best-oxygenated blood.
Ductus arteriosus: Connects the main pulmonary artery to the descending aorta, diverting ~90% of right ventricular output away from the high-resistance pulmonary vascular bed to the systemic circulation and placenta. Patency is maintained by low fetal PO2 and prostaglandins (PGE2). NSAID use in the third trimester can cause premature closure, resulting in fetal pulmonary hypertension and hydrops.
At birth, the first breath causes a dramatic decrease in pulmonary vascular resistance and increase in systemic resistance (clamping the umbilical cord). Increased left atrial pressure functionally closes the foramen ovale. Rising PaO2 and declining prostaglandins cause the ductus arteriosus to constrict (functional closure by 12-24 hours; anatomic closure by 2-3 weeks). The ductus venosus closes and becomes the ligamentum venosum.
Amniotic Fluid Dynamics
Amniotic fluid volume increases progressively through pregnancy, peaking at ~800-1000 mL at 36-37 weeks, then declining slightly toward term. Production: Early pregnancy — transudate across fetal skin and amnion; after 16 weeks — predominantly fetal urine (by term, fetal urine output is ~800-1200 mL/day) with a smaller contribution from fetal lung fluid (~250 mL/day). Removal: Fetal swallowing (~500-1000 mL/day) and intramembranous absorption. This balance explains why oligohydramnios (AFI < 5 cm or MVP < 2 cm) occurs with renal agenesis, posterior urethral valves, and uteroplacental insufficiency (decreased fetal renal perfusion), while polyhydramnios (AFI > 24 cm or MVP > 8 cm) occurs with esophageal atresia, duodenal atresia, and fetal anemia (conditions that impair fetal swallowing or increase fetal urine output).
02 Obstetric Ultrasound
First Trimester Ultrasound
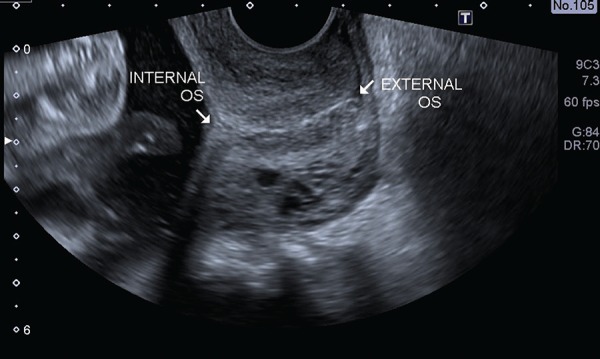
Dating: Crown-rump length (CRL) measured between 6-13+6 weeks is the most accurate parameter for determining gestational age (±5-7 days). If the CRL-based dating differs from LMP dating by > 5 days (in the first trimester) or > 7 days (second trimester), the ultrasound date should be used. A gestational sac is visible by transvaginal ultrasound (TVUS) at ~4.5-5 weeks, a yolk sac at ~5.5 weeks, and cardiac activity at 6-6.5 weeks (CRL ≥ 7 mm). Absence of cardiac activity with CRL ≥ 7 mm confirms nonviable pregnancy.
Nuchal translucency (NT): Measured between 11+0 and 13+6 weeks (CRL 45-84 mm). The NT is the sonolucent space between the skin and soft tissue overlying the fetal cervical spine. Normal NT is < 3.0 mm at the 95th percentile. Increased NT (≥ 3.0 mm) is associated with trisomy 21, trisomy 18, trisomy 13, Turner syndrome, congenital heart defects, skeletal dysplasias, and other structural anomalies. NT combined with maternal age, free β-hCG, and PAPP-A constitutes the first trimester combined screen (detection rate ~85-90% for trisomy 21, false positive rate ~5%).
Anatomy Scan — Systematic Approach
The standard detailed anatomy scan is performed at 18-22 weeks. A systematic survey includes:
Head: Biparietal diameter (BPD), head circumference (HC), lateral ventricles (< 10 mm — ventriculomegaly if ≥ 10 mm), choroid plexus, cavum septum pellucidum, cisterna magna (2-10 mm), cerebellum (mm ≈ gestational age in weeks), nuchal fold (< 6 mm at 15-20 weeks).
Face: Orbits, nasal bone, upper lip (cleft lip screening), mandible.
Spine: Sagittal and axial views through entire length, skin covering, sacrum.
Heart: Four-chamber view, left and right ventricular outflow tracts (LVOT/RVOT), three-vessel view, three-vessel trachea view. Heart rate and rhythm. Axis (normal: 45 ± 20 degrees, leftward).
Abdomen: Stomach (left-sided), kidneys (bilateral), bladder, abdominal wall integrity, umbilical cord insertion (three vessels: two arteries, one vein), abdominal circumference (AC).
Extremities: Femur length (FL), humerus, hands, feet. Long bone morphology.
Placenta: Location, cord insertion, number of vessels.
Cervix: Length (normal ≥ 25 mm at midtrimester).
Amniotic fluid: Amniotic fluid index (AFI) or maximum vertical pocket (MVP).
Fetal Growth Parameters
Four standard biometric measurements are used to calculate estimated fetal weight (EFW): BPD (biparietal diameter — outer edge to inner edge), HC (head circumference), AC (abdominal circumference — at the level of the stomach and intrahepatic umbilical vein), and FL (femur length — ossified diaphysis only). The Hadlock formula (using BPD, HC, AC, FL) is the most widely used EFW formula, with an accuracy of ±15-20%. The AC is the single most sensitive parameter for detecting growth abnormalities — a small AC reflects decreased liver glycogen stores in growth-restricted fetuses.
Cervical Length Assessment
Transvaginal cervical length measurement is a key screening tool for preterm birth. Normal cervical length at 18-24 weeks is ≥ 25 mm. A short cervix (< 25 mm) in a singleton pregnancy at 16-24 weeks is an indication for vaginal progesterone. In women with prior spontaneous preterm birth, cervical length screening begins at 16 weeks and is repeated every 1-2 weeks until 24 weeks. Cervical length < 25 mm in a patient with a prior preterm birth is an indication for cerclage placement (ultrasound-indicated cerclage).
Doppler Ultrasonography
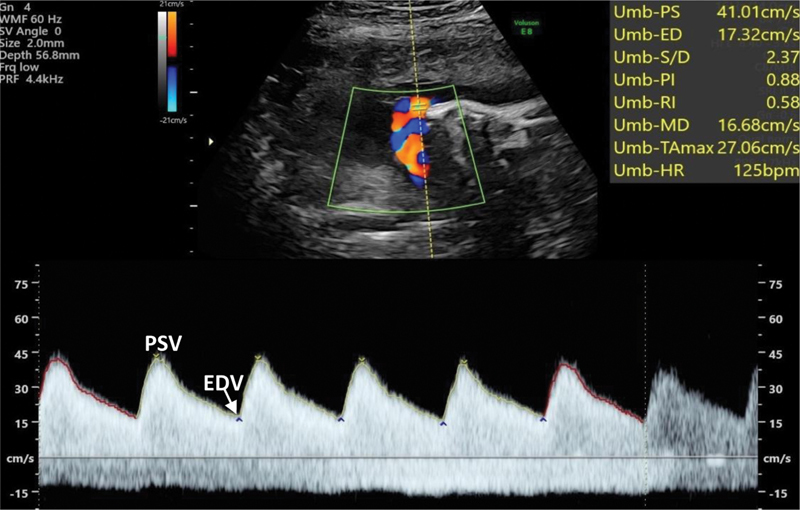
Doppler assessment is essential for surveillance of high-risk pregnancies, particularly fetal growth restriction:
Umbilical artery (UA) Doppler: Reflects placental vascular resistance. Normal: low-resistance forward flow throughout the cardiac cycle. Abnormal progression: elevated S/D ratio → absent end-diastolic flow (AEDF) → reversed end-diastolic flow (REDF). AEDF indicates loss of > 60% of placental vascular bed. REDF indicates loss of > 70% and carries a high risk of perinatal mortality. Delivery is generally recommended for REDF at ≥ 32 weeks (or earlier with abnormal venous Doppler).
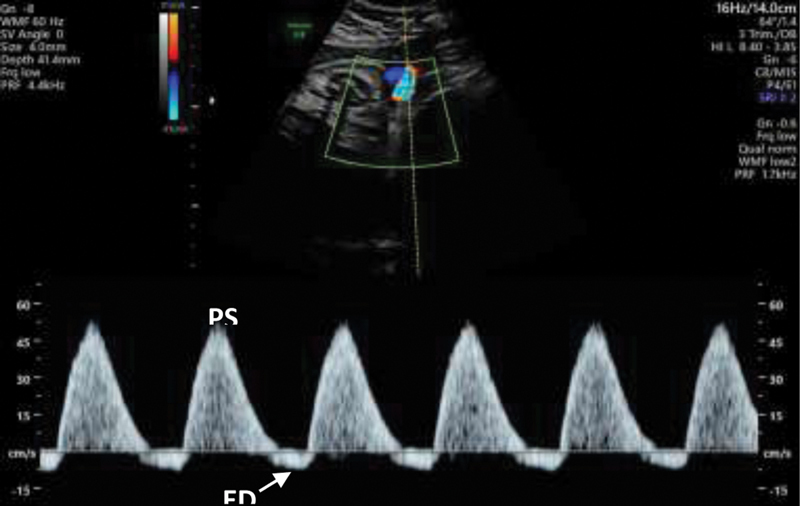
Middle cerebral artery (MCA) Doppler: Reflects fetal cerebral vascular resistance. Normally a high-resistance vessel. A low MCA PI (pulsatility index) indicates "brain sparing" — cerebral vasodilation in response to hypoxemia. MCA peak systolic velocity (MCA-PSV) is used to detect fetal anemia (elevated MCA-PSV > 1.5 MoM corresponds to moderate-severe anemia — this has largely replaced amniocentesis for Rh alloimmunization monitoring).
Cerebroplacental ratio (CPR): MCA PI / UA PI. Normal > 1.0. CPR < 1.0 indicates redistribution of blood flow to the brain and is associated with adverse perinatal outcomes even when individual values are normal.
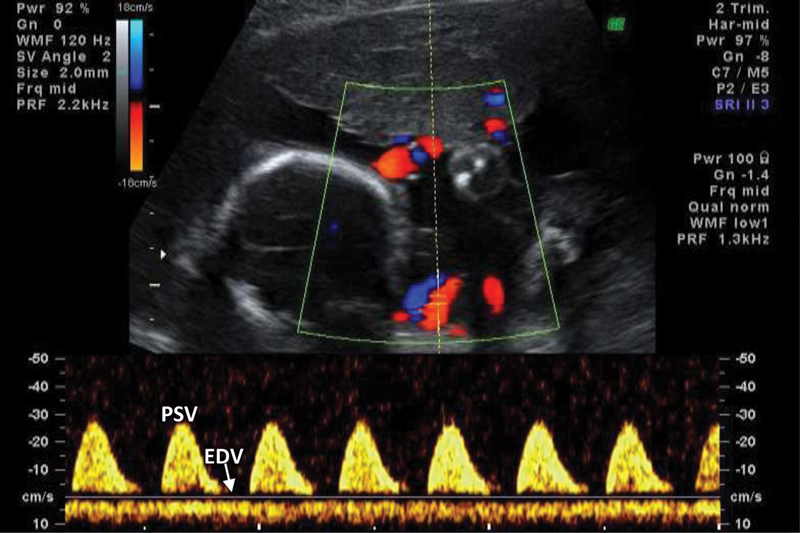
Ductus venosus (DV) Doppler: Reflects fetal cardiac function. Normal: forward flow throughout the cardiac cycle, including during atrial contraction (a-wave). Absent or reversed a-wave indicates increased central venous pressure and impending cardiac decompensation — is the most immediate predictor of fetal death in early-onset FGR and typically prompts delivery.
03 Genetic Screening & Diagnosis
Cell-Free DNA / Non-Invasive Prenatal Testing (NIPT)
Cell-free DNA (cfDNA) screening analyzes placental-origin cell-free DNA fragments in the maternal circulation. Available from 10 weeks gestation. Performance characteristics:
| Condition | Detection Rate | False Positive Rate | PPV (age 35) |
|---|---|---|---|
| Trisomy 21 (Down syndrome) | > 99% | 0.04% | ~80% |
| Trisomy 18 (Edwards syndrome) | 97-99% | 0.05% | ~65% |
| Trisomy 13 (Patau syndrome) | 92-99% | 0.04% | ~40% |
| Monosomy X (Turner syndrome) | 90-95% | 0.2% | ~30-50% |
| Sex chromosome aneuploidies | 85-95% | 0.2-1% | Variable |
Key limitations: cfDNA is a screening test, not a diagnostic test — all positive results require confirmation with diagnostic testing (amniocentesis or CVS). Factors that reduce accuracy: low fetal fraction (< 4%), early gestational age, maternal obesity, confined placental mosaicism, vanishing twin, maternal malignancy, maternal copy number variants. The cfDNA actually originates from the placenta (trophoblast), not the fetus — confined placental mosaicism is the most common cause of discordant results.
Traditional Serum Screening
First trimester combined screen (11-13+6 weeks): NT measurement + free β-hCG + PAPP-A. Detection rate for trisomy 21: 82-87%, FPR: 5%. Trisomy 21 pattern: increased NT, increased β-hCG, decreased PAPP-A. Trisomy 18 pattern: increased NT, decreased β-hCG, decreased PAPP-A.
Quad screen (15-22 weeks): AFP, hCG, unconjugated estriol (uE3), and inhibin A. Detection rate for trisomy 21: 81%, FPR: 5%. Trisomy 21 pattern: low AFP, high hCG, low uE3, high inhibin A ("ALAM" — AFP Low, All Markers abnormal). Open neural tube defects: elevated MSAFP (> 2.5 MoM). Trisomy 18: all four markers low.
Diagnostic Procedures
Chorionic villus sampling (CVS): Performed at 10-13 weeks. Obtains placental (trophoblast) tissue via transcervical or transabdominal approach. Advantage: earlier results allow earlier decision-making. Limitation: ~1-2% rate of confined placental mosaicism (CPM), which may necessitate follow-up amniocentesis. Procedure-related loss rate: ~1/455 (0.22%).
Amniocentesis: Performed at ≥ 15 weeks (typically 15-20 weeks). Obtains amniotic fluid containing fetal cells (primarily desquamated fetal skin and urogenital cells). Gold standard for karyotype. Procedure-related loss rate: ~1/900 (0.11%). Also used for fetal lung maturity testing, fetal infection workup, and ΔOD450 in alloimmunization.
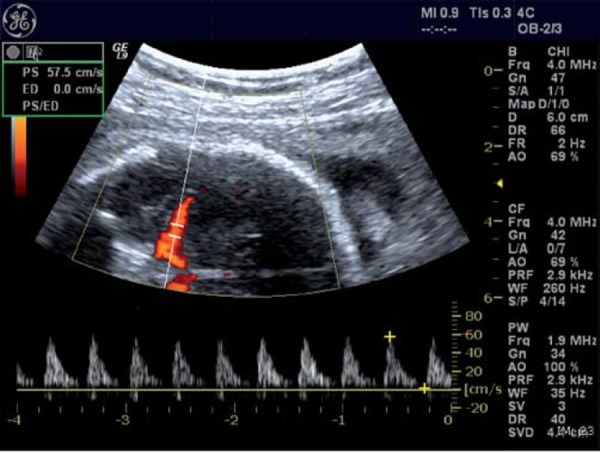
Cordocentesis (percutaneous umbilical blood sampling / PUBS): Obtains fetal blood directly from the umbilical vein at the cord insertion into the placenta. Performed at ≥ 18 weeks. Indications: rapid karyotype (results in 48-72 hours), evaluation of fetal anemia (hematocrit), fetal infection (IgM), thrombocytopenia (platelet count). Highest risk procedure — loss rate ~1-2%. Also used therapeutically for intrauterine transfusion.
Genetic Testing Modalities
Karyotype: Standard cytogenetic analysis. Detects aneuploidies, large structural rearrangements (translocations, inversions, large deletions/duplications > 5-10 Mb). Turnaround time: 10-14 days (requires cell culture). Resolution: 5-10 Mb.
FISH (fluorescence in situ hybridization): Rapid (< 24-48 hours), targeted — typically probes for chromosomes 13, 18, 21, X, Y. Does not require cell culture. Used for rapid preliminary results while awaiting full karyotype.
Chromosomal microarray analysis (CMA): Detects copy number variants (CNVs) — microdeletions and microduplications — at a resolution of ~50-100 kb (far exceeding karyotype). Recommended as a first-line test when structural anomalies are detected on ultrasound. Detects an additional ~6% of clinically significant abnormalities beyond karyotype when anomalies are present and ~1.7% when the anatomy scan is normal. Does not detect balanced translocations or inversions. Does not require cell culture (10-14 day turnaround on DNA extraction).
Exome/genome sequencing: Used when microarray is normal but a genetic etiology is suspected (multiple anomalies, skeletal dysplasia, etc.). Detects single nucleotide variants in coding regions. Diagnostic yield: ~10-30% additional diagnoses beyond microarray. Considerations: variants of uncertain significance (VUS), incidental findings, turnaround time (4-6 weeks).
04 Risk Assessment & Antepartum Surveillance
Biophysical Profile (BPP)
The BPP is a composite assessment of fetal well-being that evaluates five parameters, each scored 0 or 2 (total 0-10):
| Component | Normal (Score 2) | Abnormal (Score 0) |
|---|---|---|
| Fetal breathing movements | ≥ 1 episode ≥ 30 seconds in 30 minutes | Absent or < 30 seconds |
| Fetal movement | ≥ 3 discrete body/limb movements in 30 min | < 3 movements |
| Fetal tone | ≥ 1 episode of active extension/flexion of limbs or hand | Absent or slow extension/flexion |
| Non-stress test (NST) | Reactive (≥ 2 accelerations in 20 min) | Non-reactive |
| Amniotic fluid volume | MVP ≥ 2 cm (or AFI ≥ 5) | MVP < 2 cm (or AFI < 5) |
Interpretation:
| BPP Score | Interpretation | Management |
|---|---|---|
| 8-10 (normal AFV) | Normal — reassuring | Routine surveillance per protocol |
| 8 (oligohydramnios) | Chronic compromise suspected | Deliver if ≥ 37 weeks; evaluate if preterm |
| 6 (normal AFV) | Equivocal — possible asphyxia | Repeat in 4-6 hours; deliver if ≥ 37 weeks or persistent |
| 6 (oligohydramnios) | Probable chronic compromise | Deliver if ≥ 32 weeks |
| 4 | Abnormal — high probability of asphyxia | Deliver if ≥ 32 weeks |
| 0-2 | Strongly abnormal — almost certain asphyxia | Deliver regardless of gestational age |
The amniotic fluid component reflects long-term placental function, while the other four reflect acute CNS function. In progressive hypoxemia, the components are lost in reverse order of their developmental appearance: NST reactivity (last to develop, first to be lost) → breathing movements → body movements → tone (first to develop, last to be lost). This pattern reflects the maturation of CNS centers — the more recently developed centers (FHR reactivity, medulla) are more sensitive to hypoxemia than the earlier-developing centers (tone, cortex/subcortex).
Non-Stress Test (NST)
The NST assesses fetal heart rate (FHR) patterns by external cardiotocography. A reactive NST requires ≥ 2 accelerations of ≥ 15 bpm above baseline lasting ≥ 15 seconds within a 20-minute window (for fetuses ≥ 32 weeks). Before 32 weeks, the criteria are ≥ 10 bpm for ≥ 10 seconds. A non-reactive NST lacks these accelerations and may indicate fetal sleep cycle (extend to 40 minutes), fetal compromise, or medication effects (opioids, magnesium, betamethasone). Variable decelerations on NST suggest cord compression; late decelerations suggest uteroplacental insufficiency.
Contraction Stress Test (CST)
The CST evaluates FHR response to uterine contractions (endogenous or oxytocin-induced). Requires ≥ 3 contractions in 10 minutes. Negative CST: No late decelerations — highly reassuring (negative predictive value > 99.8% for fetal death within 1 week). Positive CST: Late decelerations with ≥ 50% of contractions — indicates uteroplacental insufficiency. Contraindications: preterm labor risk, PPROM, placenta previa, prior classical uterine incision.
Modified BPP
The modified BPP combines NST (acute marker) with amniotic fluid index (chronic marker). If both are normal (reactive NST + AFI ≥ 5), the result is reassuring. This is the most commonly used surveillance method due to ease of performance and comparable predictive value to the full BPP.
Surveillance Frequency by Condition
| Condition | Initiate Testing | Frequency | Method |
|---|---|---|---|
| Chronic hypertension (no meds) | 32 weeks | 1-2x/week | NST + AFI |
| Preeclampsia without severe features | At diagnosis | 2x/week | NST + AFI or BPP |
| Pregestational diabetes (well-controlled) | 32 weeks | 1-2x/week | NST + AFI |
| Gestational diabetes (insulin-treated) | 32 weeks | 1-2x/week | NST + AFI |
| FGR (normal Doppler) | At diagnosis | 1-2x/week | BPP + Doppler |
| FGR (abnormal Doppler) | At diagnosis | 2-3x/week or daily | BPP + Doppler |
| Decreased fetal movement | At presentation | Single evaluation | NST ± BPP |
| Post-dates (≥ 41 weeks) | 41 weeks | 2x/week | NST + AFI |
| Monochorionic twins | 16 weeks (for TTTS) | Every 2 weeks | US for TTTS signs |
05 Hypertensive Disorders of Pregnancy
Classification
Hypertensive disorders complicate 5-10% of pregnancies and are a leading cause of maternal morbidity and mortality. The ACOG classification divides them into four categories:
Chronic hypertension: HTN present before pregnancy or diagnosed before 20 weeks gestation (SBP ≥ 140 mmHg and/or DBP ≥ 90 mmHg). Persists > 12 weeks postpartum.
Gestational hypertension: New-onset HTN at ≥ 20 weeks without proteinuria or other features of preeclampsia. Up to 50% will progress to preeclampsia (especially if diagnosed before 32 weeks).
Preeclampsia-eclampsia: New-onset HTN at ≥ 20 weeks with proteinuria (≥ 300 mg/24 hours or protein/creatinine ratio ≥ 0.3) OR HTN with any of the following severe features even without proteinuria: thrombocytopenia, renal insufficiency, liver dysfunction, cerebral symptoms, or pulmonary edema.
Chronic HTN with superimposed preeclampsia: Worsening HTN or new proteinuria/end-organ dysfunction after 20 weeks in a woman with chronic HTN.
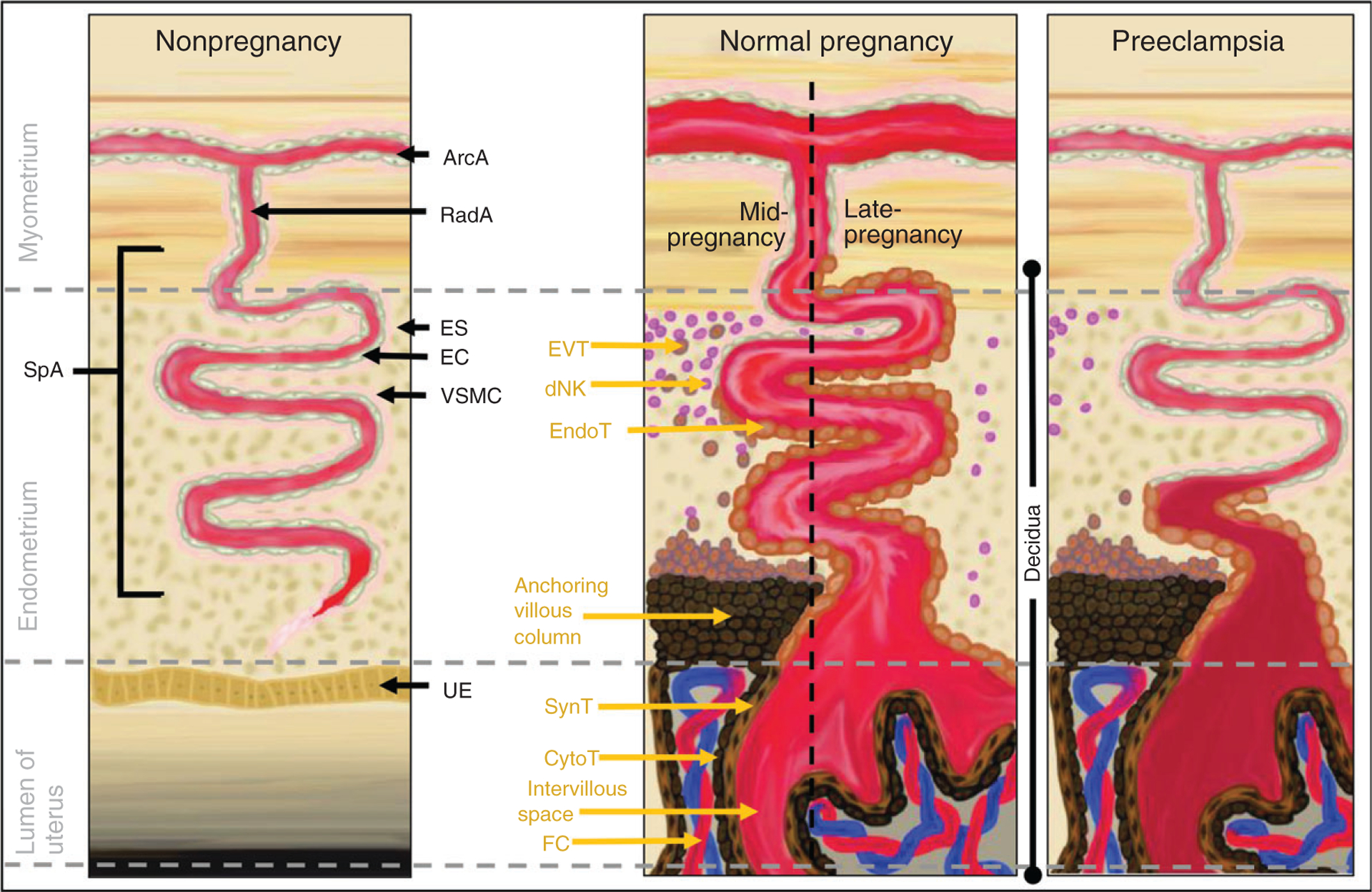
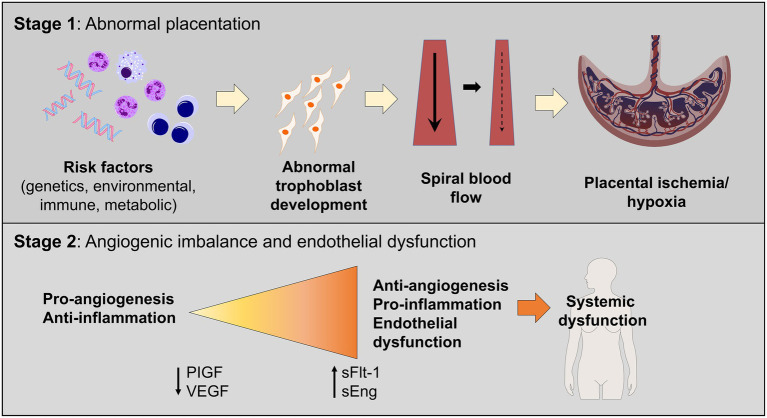
Preeclampsia — Diagnostic Criteria
Preeclampsia without severe features: BP ≥ 140/90 mmHg on two occasions at least 4 hours apart after 20 weeks + proteinuria (≥ 300 mg/24h, P:C ≥ 0.3, or dipstick ≥ 2+).
Preeclampsia with severe features (any one of the following):
- SBP ≥ 160 mmHg or DBP ≥ 110 mmHg on two occasions (minutes apart, allowing treatment)
- Platelet count < 100,000/μL
- Serum creatinine > 1.1 mg/dL or doubling of baseline
- Liver transaminases ≥ 2× upper limit of normal
- Pulmonary edema
- New-onset headache unresponsive to medication or visual disturbances
HELLP Syndrome
HELLP — Hemolysis, Elevated Liver enzymes, Low Platelets — is a severe variant of preeclampsia. It may occur without significant hypertension or proteinuria (15-20% of cases). Peak incidence: 28-36 weeks; 30% present postpartum.
Mississippi (Martin) Classification — by platelet nadir:
Class 1: Platelets ≤ 50,000/μL (most severe, highest complication rate)
Class 2: Platelets > 50,000 to ≤ 100,000/μL
Class 3: Platelets > 100,000 to ≤ 150,000/μL (+ LDH ≥ 600, AST/ALT ≥ 40)
Tennessee Classification — full vs partial:
Complete HELLP: All three criteria — hemolysis (LDH ≥ 600 U/L, abnormal smear), AST ≥ 70 U/L, platelets < 100,000/μL
Partial HELLP: Only one or two of the three criteria present
Eclampsia
Eclampsia is the occurrence of generalized tonic-clonic seizures in a patient with preeclampsia that cannot be attributed to other causes. It occurs antepartum (38-53%), intrapartum (18-36%), or postpartum (11-44%). It can occur without preceding severe hypertension or proteinuria. Management: left lateral position, protect airway, magnesium sulfate (4-6 g IV loading dose over 15-20 minutes, then 1-2 g/hour maintenance), stabilize BP, deliver after stabilization.
Aspirin Prophylaxis for Preeclampsia Prevention
ACOG/USPSTF recommendation: Low-dose aspirin (81 mg/day) starting at 12-16 weeks (optimally before 16 weeks) through 36 weeks for women at high risk. High-risk criteria (one or more): prior preeclampsia, multifetal gestation, chronic hypertension, type 1 or type 2 diabetes, renal disease, autoimmune disease (SLE, APS). Moderate-risk criteria (two or more): nulliparity, obesity (BMI > 30), family history of preeclampsia, African American race, low socioeconomic status, age ≥ 35, prior adverse outcome (SGA, abruption, stillbirth). The ASPRE trial demonstrated that aspirin 150 mg nightly (starting 11-14 weeks, based on first-trimester combined screening) reduced preterm preeclampsia by 62%. Nighttime administration is preferred (targets the nocturnal platelet activation peak).
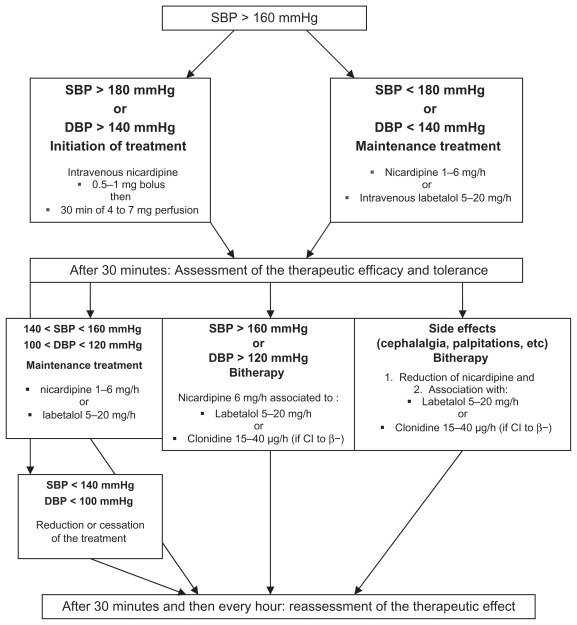
Acute Management of Severe HTN
Acute-onset severe hypertension (SBP ≥ 160 and/or DBP ≥ 110) requires urgent treatment within 30-60 minutes to prevent stroke, the leading cause of death in preeclampsia. First-line agents:
| Medication | Dose | Route | Onset | Notes |
|---|---|---|---|---|
| Labetalol | 20 mg IV, then 40 mg, then 80 mg (escalating q10 min; max 300 mg) | IV push | 5 minutes | Avoid in asthma, heart block, heart failure |
| Hydralazine | 5-10 mg IV q20 min (max 20 mg) | IV push | 10-20 minutes | Less predictable response, reflex tachycardia |
| Nifedipine | 10-20 mg PO q20-30 min (max 50 mg) | Oral (immediate release) | 10-20 minutes | Use only oral; avoid sublingual route |
Magnesium Sulfate Protocol
Indication: Seizure prophylaxis in preeclampsia with severe features and treatment of eclampsia. Loading dose: 4-6 g IV over 15-20 minutes, followed by maintenance infusion of 1-2 g/hour. Continue for 24-48 hours postpartum. Monitor: deep tendon reflexes (lost at 7-10 mEq/L), respiratory rate (≥ 12/min), urine output (≥ 30 mL/hour). Therapeutic level: 4-7 mEq/L. Toxicity: respiratory depression (10-12 mEq/L), cardiac arrest (15-20 mEq/L). Antidote: calcium gluconate 1 g IV over 3 minutes.
Delivery Timing
Gestational hypertension or preeclampsia without severe features: deliver at 37+0 weeks (per HYPITAT/PHOENIX trials). Preeclampsia with severe features: deliver at 34+0 weeks (or at diagnosis if ≥ 34 weeks). Before 34 weeks with severe features: individualize — administer corticosteroids and deliver within 48 hours unless expectant management criteria are met (stable maternal/fetal status, inpatient setting). HELLP: deliver after maternal stabilization, regardless of gestational age if < 34 weeks (after steroids if feasible). Eclampsia: deliver after seizure control and maternal stabilization.
06 Diabetes in Pregnancy
Pregestational Diabetes (Type 1 & Type 2)
Pregestational diabetes complicates ~1% of pregnancies and is associated with significantly increased risks of congenital anomalies (3-5× baseline, especially cardiac — VSD, transposition; NTDs; caudal regression syndrome), macrosomia, preeclampsia, stillbirth, and neonatal hypoglycemia. Risk is directly related to periconceptional glycemic control — HbA1c < 6.5% before conception is the target (anomaly risk is near baseline at this level; risk doubles with HbA1c > 8% and increases 5-10× with HbA1c > 10%).
Periconceptional counseling: Folic acid 4 mg/day (not 0.4 mg — the higher dose for NTD prevention in high-risk patients), optimize HbA1c to < 6.5%, discontinue teratogenic medications (ACE inhibitors, ARBs, statins), assess for retinopathy, nephropathy, and cardiac disease before pregnancy.
Glucose targets in pregnancy (ACOG recommendations): fasting < 95 mg/dL, 1-hour postprandial < 140 mg/dL, 2-hour postprandial < 120 mg/dL. Insulin is the preferred pharmacotherapy — it does not cross the placenta. Insulin requirements typically increase by 50-100% during pregnancy due to progressive insulin resistance (mediated by human placental lactogen, cortisol, TNF-α). A drop in insulin requirements in the third trimester may indicate placental insufficiency and warrants increased surveillance.
White Classification of Diabetes in Pregnancy
| Class | Description | Onset Age | Duration | Vascular Disease |
|---|---|---|---|---|
| A1 | Gestational — diet controlled | Any | Any | None |
| A2 | Gestational — medication required | Any | Any | None |
| B | Pregestational | ≥ 20 years | < 10 years | None |
| C | Pregestational | 10-19 years | 10-19 years | None |
| D | Pregestational | < 10 years | ≥ 20 years | Benign retinopathy |
| F | Pregestational | Any | Any | Nephropathy |
| R | Pregestational | Any | Any | Proliferative retinopathy |
| RF | Pregestational | Any | Any | Both retinopathy & nephropathy |
| H | Pregestational | Any | Any | CAD / cardiomyopathy |
| T | Pregestational | Any | Any | Prior renal transplant |
Gestational Diabetes (GDM)
Gestational diabetes affects 6-9% of pregnancies. Universal screening is recommended at 24-28 weeks. Two approaches:
Two-step approach (most common in the US): Step 1 — 50 g glucose challenge test (GCT), 1-hour glucose ≥ 130-140 mg/dL is positive screening. Step 2 — 100 g 3-hour oral glucose tolerance test (OGTT). Diagnosis requires ≥ 2 abnormal values: fasting ≥ 95 mg/dL, 1-hour ≥ 180, 2-hour ≥ 155, 3-hour ≥ 140 (Carpenter-Coustan criteria).
One-step approach (IADPSG/WHO): 75 g 2-hour OGTT. Diagnosis requires ≥ 1 abnormal value: fasting ≥ 92 mg/dL, 1-hour ≥ 180, 2-hour ≥ 153.
Management: Nutritional counseling (carbohydrate-controlled diet: 3 meals + 2-3 snacks, ~33-40% carbohydrate calories) and glucose monitoring (fasting + 1-hour or 2-hour postprandial, 4 times/day minimum). If targets are not met within 1-2 weeks, initiate pharmacotherapy. Insulin is first-line and the only FDA-approved agent for diabetes in pregnancy. Common regimens: basal insulin (NPH or detemir) for fasting hyperglycemia + rapid-acting insulin (lispro, aspart) for postprandial hyperglycemia. Metformin and glyburide are used off-label — metformin crosses the placenta (long-term effects on offspring under study, may have benefits for maternal weight and insulin requirements); glyburide is associated with higher rates of neonatal hypoglycemia and macrosomia compared to insulin and is increasingly falling out of favor after the MiTy and GEMS trials.
Fetal surveillance: A1 GDM (diet-controlled): no routine antepartum testing beyond growth scans (growth US at 28-32 weeks to assess for macrosomia). A2 GDM (medication-treated): initiate antepartum testing at 32 weeks (NST 1-2x/week). Pregestational diabetes: antepartum testing at 32 weeks (or earlier if poorly controlled or with vasculopathy); growth US every 3-4 weeks starting at 28 weeks. Delivery timing: A1 GDM: deliver at 39+0-40+6 weeks (expectant management to 40+6). A2 GDM: deliver at 39+0-39+6 weeks. Pregestational diabetes (well-controlled without vasculopathy): deliver at 37+0-39+6 weeks. Pregestational diabetes (poorly controlled or with vasculopathy): individualize delivery at 34+0-37+6 weeks. Early third-trimester fetal echocardiography is recommended for pregestational diabetes given the 3-5× increased risk of congenital heart defects.
07 Cardiac Disease in Pregnancy
Physiologic Cardiovascular Changes in Pregnancy
Normal pregnancy produces dramatic hemodynamic changes: blood volume increases by 40-50% (peaking at 32-34 weeks), cardiac output increases by 30-50% (increased HR by 10-20 bpm and increased stroke volume), systemic vascular resistance decreases by 20-30% (progesterone-mediated vasodilation), and blood pressure normally decreases in the first and second trimesters (nadir at 24-28 weeks) before returning to baseline at term. These changes make pregnancy a cardiovascular stress test — women with limited cardiac reserve may decompensate, particularly at 28-34 weeks (maximum volume loading) and during labor/delivery (autotransfusion of 300-500 mL with each contraction).
Modified WHO (mWHO) Risk Classification
mWHO Class I — No detectable increased risk: Small PDA, repaired simple lesions (ASD, VSD, PDA), mitral valve prolapse without significant regurgitation, isolated PVCs/PACs.
mWHO Class II — Small increased risk: Unoperated ASD or VSD, repaired tetralogy of Fallot, most arrhythmias, Turner syndrome without aortic dilation.
mWHO Class II-III — Intermediate risk: Mild LV dysfunction (EF 45-54%), hypertrophic cardiomyopathy, native or tissue valve disease not mWHO IV, Marfan without aortic dilation, bicuspid aortic valve with aorta < 45 mm.
mWHO Class III — Significantly increased risk: Mechanical valve, systemic RV (corrected TGA), Fontan circulation, unrepaired cyanotic heart disease, Marfan with aorta 40-45 mm, bicuspid aortic valve with aorta 45-50 mm.
mWHO Class IV — Extremely high risk (pregnancy contraindicated): Pulmonary arterial hypertension, severe systemic ventricular dysfunction (EF < 30%, NYHA III-IV), peripartum cardiomyopathy with residual LV dysfunction, severe mitral stenosis, severe aortic stenosis, Marfan with aorta > 45 mm, bicuspid aortic valve with aorta > 50 mm, severe coarctation, Eisenmenger syndrome. Maternal mortality in Eisenmenger = 20-50%.
Specific Cardiac Conditions
Mechanical heart valves: Require continuous anticoagulation throughout pregnancy. Options: dose-adjusted LMWH (anti-Xa 0.8-1.2 U/mL 4-6h post-dose) throughout pregnancy, warfarin throughout pregnancy (lowest fetal risk if dose ≤ 5 mg/day, but warfarin embryopathy risk 4-10% in first trimester at higher doses), or sequential regimen (LMWH in first trimester, warfarin second/early third trimester, switch to heparin near delivery). Warfarin offers the lowest thrombotic risk to the mother; LMWH offers the lowest teratogenic risk to the fetus. There is no perfect solution — shared decision-making is essential.
Peripartum cardiomyopathy (PPCM): New-onset HF with EF < 45% presenting in the last month of pregnancy through 5 months postpartum, with no other identifiable cause. Incidence ~1 in 1,000-4,000. Risk factors: African American race, multiparity, advanced maternal age, preeclampsia, tocolytic use. Treatment: standard HF therapy (diuretics, hydralazine/nitrates — avoid ACE inhibitors until postpartum, beta-blockers). Approximately 50% recover normal EF within 6 months. Bromocriptine shows promise. Subsequent pregnancy carries recurrence risk of 30-50% (higher if EF has not normalized).
Marfan syndrome: Autosomal dominant FBN1 mutation. Risk of aortic root dissection increases during pregnancy, particularly in the third trimester and peripartum (hemodynamic stress, hormonal effects on connective tissue). Aortic root < 40 mm: pregnancy generally tolerated with close monitoring (echocardiography every 4-8 weeks). Aortic root 40-45 mm: higher risk, individualized counseling advised, consider genetic counseling (50% transmission to offspring). Aortic root > 45 mm: pregnancy contraindicated — prophylactic aortic root repair before pregnancy recommended. Beta-blockers (metoprolol, atenolol) should be continued throughout pregnancy to reduce the rate of aortic root dilation.
Eisenmenger syndrome: Pulmonary hypertension with right-to-left shunting. Maternal mortality 20-50%. Pregnancy is absolutely contraindicated. The physiologic decrease in systemic vascular resistance during pregnancy worsens right-to-left shunting, leading to progressive cyanosis, syncope, and sudden death. If pregnancy occurs, termination should be offered. If continued, intensive monitoring, pulmonary vasodilators (sildenafil, inhaled nitric oxide), and early delivery are needed.
Aortic stenosis: Mild-moderate AS is generally well tolerated. Severe symptomatic AS (valve area < 1.0 cm²) is high risk (mWHO III-IV). The inability to increase cardiac output across a fixed obstruction during the hemodynamic changes of pregnancy leads to heart failure, syncope, and arrhythmia. Ideally, severe AS should be corrected before pregnancy. If diagnosed during pregnancy: medical management with beta-blockers, avoid dehydration and vasodilation, consider percutaneous balloon valvuloplasty for critical cases.
08 Renal Disease in Pregnancy
Chronic Kidney Disease (CKD) in Pregnancy
Normal pregnancy increases GFR by ~50% — serum creatinine normally decreases to 0.4-0.6 mg/dL (a "normal" non-pregnant creatinine of 1.0 mg/dL represents significant renal impairment in pregnancy). The degree of baseline renal dysfunction predicts outcomes: mild CKD (creatinine < 1.4): generally favorable outcomes, 20% risk of preeclampsia; moderate CKD (creatinine 1.4-2.5): 40% risk of preeclampsia, 35% risk of preterm delivery, 20% risk of accelerated decline in renal function; severe CKD (creatinine > 2.5): poor fetal outcomes, high risk of preeclampsia (60-80%), often requires dialysis during pregnancy. Pregnancy is generally advised against with creatinine > 2.5 mg/dL.
Pregnancy After Renal Transplant
Pregnancy is reasonable ≥ 1-2 years post-transplant if: stable graft function (creatinine < 1.5), no recent rejection episodes, no active infections, minimal proteinuria, controlled BP, and immunosuppression at maintenance levels. Safe immunosuppressants include: azathioprine, cyclosporine, tacrolimus, and prednisone. Mycophenolate mofetil (CellCept) is absolutely contraindicated — teratogenic (cleft lip/palate, ear anomalies, heart defects) — must be stopped ≥ 6 weeks before conception and replaced with azathioprine.
Acute Kidney Injury in Pregnancy
The differential diagnosis of AKI in pregnancy is broad and includes pregnancy-specific etiologies:
HELLP syndrome: Most common. Occurs in preeclampsia context. Treatment = delivery. LDH elevated, haptoglobin low, schistocytes present.
TTP (thrombotic thrombocytopenic purpura): ADAMTS13 activity < 10%. Pentad: thrombocytopenia, MAHA, neurologic symptoms, renal dysfunction, fever. Treatment: plasma exchange. Does NOT improve with delivery alone.
aHUS (atypical hemolytic uremic syndrome): Complement-mediated. Often postpartum onset. AKI predominates. Normal ADAMTS13. Treatment: eculizumab (C5 inhibitor). Does NOT improve with delivery alone.
AFLP (acute fatty liver of pregnancy): Microvesicular steatosis. Occurs in third trimester. Swansea criteria. Hypoglycemia and coagulopathy (low fibrinogen) are distinguishing features. Treatment = delivery. Associated with LCHAD deficiency (screen infant).
09 Thrombophilia & Venous Thromboembolism
Inherited Thrombophilias
Pregnancy is a hypercoagulable state due to increased clotting factors (I, VII, VIII, X, vWF), decreased protein S, increased PAI-1 and PAI-2, and venous stasis. The risk of VTE is 4-5× higher in pregnancy and 20-80× higher in the postpartum period.
| Thrombophilia | Prevalence | VTE Risk (Pregnancy) | Pregnancy Loss Risk |
|---|---|---|---|
| Factor V Leiden (heterozygous) | 5% (Caucasian) | 3-8× | Mild increase (late loss) |
| Factor V Leiden (homozygous) | 0.02% | 25-50× | Increased |
| Prothrombin G20210A (heterozygous) | 2-3% | 3-5× | Mild increase |
| Antithrombin III deficiency | 0.02-0.2% | 25-50× | Increased |
| Protein C deficiency | 0.2-0.5% | 3-10× | Conflicting data |
| Protein S deficiency | 0.1-1% | 2-6× | Conflicting data |
Antiphospholipid Syndrome (APS)
Sapporo (revised Sydney) criteria require at least one clinical AND one laboratory criterion:
Clinical criteria (at least one):
1. Vascular thrombosis: ≥ 1 episode of arterial, venous, or small vessel thrombosis
2. Pregnancy morbidity: (a) ≥ 1 unexplained death of morphologically normal fetus at ≥ 10 weeks, OR (b) ≥ 1 preterm birth < 34 weeks due to preeclampsia or placental insufficiency, OR (c) ≥ 3 consecutive unexplained spontaneous losses < 10 weeks
Laboratory criteria (at least one, on ≥ 2 occasions ≥ 12 weeks apart):
1. Lupus anticoagulant (LA) — strongest predictor of thrombosis
2. Anticardiolipin antibodies (aCL) — IgG or IgM, medium-high titer (> 40 GPL/MPL or > 99th percentile)
3. Anti-β2 glycoprotein I antibodies — IgG or IgM (> 99th percentile)
Treatment in obstetric APS: Low-dose aspirin (81-162 mg, starting before 16 weeks) PLUS prophylactic-dose heparin (unfractionated or LMWH) throughout pregnancy and 6-12 weeks postpartum. For patients with prior thrombosis: therapeutic-dose anticoagulation. For triple-positive APS (all three antibodies positive): highest risk — therapeutic anticoagulation is recommended even without prior thrombosis by some experts.
Thromboprophylaxis Protocols
| Risk Category | Antepartum | Postpartum |
|---|---|---|
| Low-risk thrombophilia (FVL het, PG20210A het) — no prior VTE | Surveillance only | Surveillance or prophylactic LMWH × 6 weeks (if additional risk factors) |
| Low-risk thrombophilia — prior VTE | Prophylactic or intermediate-dose LMWH | Therapeutic LMWH or warfarin × 6 weeks |
| High-risk thrombophilia (AT-III deficiency, FVL homozygous, compound heterozygote) — no prior VTE | Prophylactic LMWH | Prophylactic LMWH × 6 weeks |
| High-risk thrombophilia — prior VTE | Therapeutic LMWH | Therapeutic LMWH or warfarin × 6 weeks |
| APS — obstetric criteria only | Prophylactic LMWH + aspirin 81 mg | Prophylactic LMWH × 6-12 weeks |
| APS — prior thrombosis | Therapeutic LMWH + aspirin 81 mg | Therapeutic LMWH/warfarin (lifelong) |
| Prior unprovoked VTE — no thrombophilia | Prophylactic or intermediate-dose LMWH | Therapeutic LMWH or warfarin × 6 weeks |
| Prior provoked VTE (transient risk factor) — no thrombophilia | Surveillance | Prophylactic LMWH × 6 weeks |
VTE Treatment in Pregnancy
LMWH is the drug of choice for treatment of VTE in pregnancy (enoxaparin 1 mg/kg q12h, adjusted-dose dalteparin, or tinzaparin). LMWH does not cross the placenta and has a more predictable pharmacokinetic profile than UFH. Anti-Xa levels should be monitored (target peak 0.6-1.0 U/mL 4-6 hours post-dose for therapeutic dosing). Dose adjustments are often needed as pregnancy progresses due to increased volume of distribution and renal clearance. Warfarin is contraindicated for VTE treatment in pregnancy — crosses the placenta, causes warfarin embryopathy (nasal hypoplasia, stippled epiphyses) in weeks 6-12 and CNS anomalies throughout pregnancy. DOACs (rivaroxaban, apixaban, etc.) are also contraindicated — they cross the placenta and animal studies show reproductive toxicity. IVC filters are reserved for patients with contraindications to anticoagulation or recurrent PE despite therapeutic anticoagulation.
Peripartum management: Hold LMWH 24 hours before planned delivery (12 hours for prophylactic dose). Neuraxial anesthesia requires ≥ 12 hours after prophylactic dose, ≥ 24 hours after therapeutic dose. Resume anticoagulation 6-12 hours after vaginal delivery, 12-24 hours after cesarean. Continue for ≥ 6 weeks postpartum (minimum total duration: 3 months for provoked VTE). Warfarin and LMWH are both safe with breastfeeding. Postpartum transition to warfarin (INR 2-3) is an option for patients who prefer oral therapy.
10 Autoimmune Disease in Pregnancy
Systemic Lupus Erythematosus (SLE)
SLE flares occur in ~40-60% of pregnancies (risk higher if disease is active at conception — ideally defer pregnancy until 6 months of quiescence). Key complications: preeclampsia (20-30%), fetal growth restriction, preterm delivery, and pregnancy loss. Distinguishing lupus nephritis flare from preeclampsia is critical and often challenging — complement levels (C3/C4 — decrease in lupus flare but are normally increased in pregnancy), anti-dsDNA titers (rise in flare), and urine sediment (active sediment with casts in lupus) help differentiate. Hydroxychloroquine should be continued throughout pregnancy — it reduces flares, preeclampsia risk, and neonatal lupus incidence.
Neonatal lupus: Caused by transplacental passage of maternal anti-Ro (SSA) and anti-La (SSB) antibodies. Manifestations: cutaneous lupus rash (photosensitive, resolves by 6-9 months as maternal antibodies clear), hematologic (cytopenias), hepatic, and congenital heart block (CHB — the most serious complication, incidence ~2% with positive antibodies, 18% recurrence with prior affected child). Complete CHB is irreversible and may require fetal/neonatal pacemaker. Surveillance: serial fetal echocardiography every 1-2 weeks from 16-26 weeks to detect PR prolongation before progression to complete block. Dexamethasone (crosses the placenta) may be used for incomplete block, though efficacy for preventing progression is debated.
Rheumatoid Arthritis
RA typically improves during pregnancy (70% of patients) due to the immunosuppressive effect of pregnancy, but flares are common postpartum (90% within 3 months). Safe medications: hydroxychloroquine, sulfasalazine, low-dose prednisone, azathioprine, TNF-α inhibitors (certolizumab preferred — does not cross placenta due to pegylated Fab fragment structure). Contraindicated: methotrexate (teratogenic — stop ≥ 3 months before conception), leflunomide (requires cholestyramine washout), JAK inhibitors, cyclophosphamide.
Inflammatory Bowel Disease (IBD)
Active IBD at conception is the strongest predictor of poor pregnancy outcomes. Maintain disease remission through pregnancy — most IBD medications are safe (5-ASA, azathioprine/6-MP, anti-TNF agents, corticosteroids). Methotrexate must be stopped before conception. Timing of biologic dosing is adjusted so the last dose is given 6-10 weeks before the expected delivery to minimize neonatal immunosuppression. Live vaccines are delayed in infants exposed to biologics in utero until 6-12 months of age.
11 Hematologic Disorders
Sickle Cell Disease
Sickle cell disease (HbSS, HbSC, HbS-β-thalassemia) in pregnancy is associated with increased risk of pain crises, acute chest syndrome, preeclampsia (2-4×), FGR, preterm delivery, VTE, and maternal mortality (0.5-2%). Management: continued folic acid supplementation (4 mg/day), serial growth ultrasounds, low-dose aspirin for preeclampsia prevention, prophylactic transfusion is controversial (TAPS study suggests benefit). Pain crises are managed with IV hydration, oxygen, and opioid analgesia (avoid NSAIDs after 20 weeks). Hydroxyurea is teratogenic and must be stopped before conception (limited data on inadvertent first-trimester exposure). Iron overload should be assessed and treated before pregnancy.
Immune Thrombocytopenic Purpura (ITP)
ITP results from autoantibodies (IgG) against platelet glycoproteins. These antibodies cross the placenta and can cause fetal/neonatal thrombocytopenia (but severe neonatal bleeding is rare — < 1% intracranial hemorrhage). Treatment: first-line is corticosteroids (prednisone 1 mg/kg) or IVIG (1 g/kg × 2 days) for platelet counts < 30,000 or in the third trimester to prepare for delivery. Target for vaginal delivery: platelets ≥ 50,000. Target for neuraxial anesthesia: platelets ≥ 70,000-80,000. Mode of delivery is based on obstetric indications — ITP alone is not an indication for cesarean delivery.
Von Willebrand Disease
The most common inherited bleeding disorder. VWF levels normally increase during pregnancy (may reach normal levels in type 1 by the third trimester). Management varies by type: Type 1 (most common — quantitative deficiency): VWF levels typically normalize by delivery, DDAVP may be used for mild bleeding. Type 2: qualitative defects — VWF factor concentrates preferred over DDAVP. Type 3: severe quantitative deficiency — VWF/FVIII concentrates for delivery and postpartum (risk of delayed postpartum hemorrhage as VWF levels decline rapidly after delivery). Levels should be checked at 28-34 weeks and targeted to ≥ 50 IU/dL for delivery.
Iron Deficiency Anemia
The most common cause of anemia in pregnancy. Physiologic anemia of pregnancy (dilutional, hemoglobin nadir ~10.5 g/dL at 28-34 weeks) must be distinguished from true iron deficiency (ferritin < 30 ng/mL, low serum iron, elevated TIBC). Iron requirements increase from 1 mg/day prepregnancy to 6-7 mg/day in the third trimester. Oral iron (ferrous sulfate 325 mg 1-2x/day, between meals with vitamin C) is first-line. IV iron (ferric carboxymaltose, iron sucrose) is indicated for intolerance of oral iron, severe anemia (Hb < 7-8 g/dL), or second/third trimester when rapid repletion is needed.
12 Infections in Pregnancy
TORCH Infections
The TORCH acronym encompasses the major perinatal infections: Toxoplasmosis, Other (syphilis, varicella, parvovirus B19), Rubella, Cytomegalovirus, Herpes simplex virus. Shared clinical features of congenital TORCH infection include: IUGR, hepatosplenomegaly, jaundice, thrombocytopenia, rash, and intracranial calcifications (pattern varies by pathogen).
Cytomegalovirus (CMV): Most common congenital infection (0.2-2% of live births). Primary infection has 30-40% vertical transmission rate (reactivation: 1-2%). Congenital CMV: sensorineural hearing loss (most common sequela), periventricular calcifications, microcephaly, hepatosplenomegaly, petechiae. Treatment: valacyclovir in maternal primary infection may reduce transmission; ganciclovir/valganciclovir for symptomatic neonates.
Toxoplasmosis: Transmission risk increases with gestational age (15% first trimester, 70% third trimester), but severity is inversely related. Classic triad: hydrocephalus, intracranial calcifications (diffuse/scattered), chorioretinitis. Treatment: spiramycin (to reduce vertical transmission if maternal seroconversion before 18 weeks) or pyrimethamine + sulfadiazine + leucovorin (if fetal infection confirmed).
Parvovirus B19: Vertical transmission ~25-30%. Causes aplastic crisis in fetus (tropism for erythroid progenitors) leading to severe anemia, hydrops fetalis, and fetal death (highest risk: infection at 13-20 weeks). Monitor with serial MCA-PSV Doppler for anemia. Intrauterine transfusion for severe anemia. No antiviral treatment; IVIG does not prevent vertical transmission.
Syphilis: Vertical transmission occurs at any stage — highest with primary/secondary (60-100%). Congenital syphilis: hepatosplenomegaly, rash, snuffles, osteochondritis, periostitis. Screening at first prenatal visit (and third trimester in high-risk populations). Treatment: penicillin G (benzathine penicillin 2.4 million units IM for early syphilis — penicillin is the ONLY proven treatment in pregnancy; penicillin-allergic patients must undergo desensitization).
Herpes simplex virus (HSV): Neonatal herpes risk is highest with primary maternal infection near delivery (30-50% transmission) vs recurrent infection (< 3%). Management: acyclovir/valacyclovir suppression from 36 weeks in women with history of genital HSV. Cesarean delivery for active genital lesions or prodromal symptoms at time of labor. Avoid artificial rupture of membranes and fetal scalp electrode in HSV-positive patients.
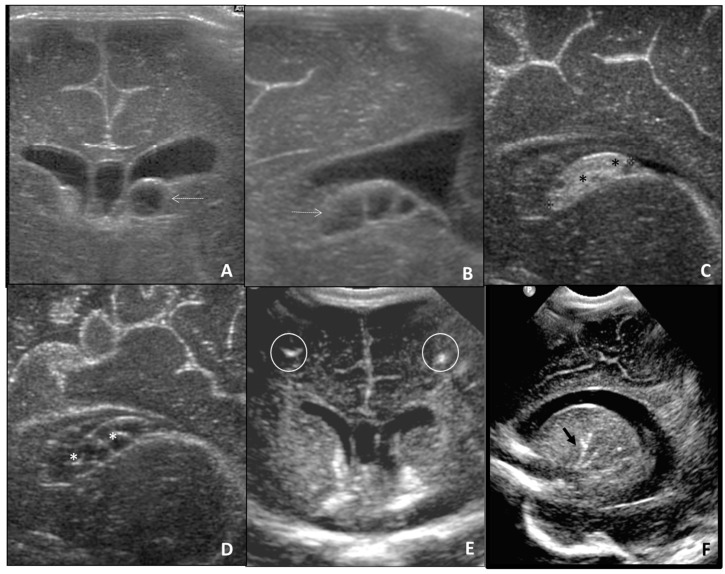
Zika Virus
Zika virus is an arbovirus (Flaviviridae family) transmitted by Aedes mosquitoes and sexually. Maternal infection in pregnancy (especially first/second trimester) is associated with congenital Zika syndrome: severe microcephaly, intracranial calcifications (subcortical, distinct from periventricular pattern of CMV), macular scarring/focal pigmentary retinal mottling, congenital contractures (arthrogryposis), and marked early hypertonia. Most maternal infections are asymptomatic or mild (rash, conjunctivitis, arthralgia, low-grade fever). Vertical transmission rate: ~5-15% (risk of microcephaly ~5% with first-trimester infection). Diagnosis: serum and urine Zika RNA PCR, Zika IgM (cross-reactivity with dengue is common). No specific antiviral treatment or vaccine. Prevention: avoid travel to endemic areas, mosquito precautions, condom use for partners with Zika exposure for ≥ 3 months.
HIV in Pregnancy
Without intervention, vertical transmission is 25-30%. With combined antiretroviral therapy (cART), elective cesarean for viral load > 1,000 copies/mL, and avoidance of breastfeeding, transmission is reduced to < 1-2%. Universal screening at first prenatal visit. ART should be initiated or continued regardless of CD4 count. Intrapartum: IV zidovudine for women with viral load > 1,000. Scheduled cesarean at 38 weeks if viral load > 1,000 near delivery. If viral load < 1,000 and on cART: vaginal delivery is appropriate. Avoid invasive procedures (amniotomy, fetal scalp electrode, operative vaginal delivery) when possible.
Hepatitis B & C
Hepatitis B: Universal screening with HBsAg at first prenatal visit. Vertical transmission with HBeAg-positive mother: 70-90% without prophylaxis. Prevention: neonatal hepatitis B immunoglobulin (HBIG) + HBV vaccine within 12 hours of birth (reduces transmission to < 5%). Tenofovir in the third trimester for mothers with viral load > 200,000 IU/mL further reduces transmission. Breastfeeding is safe after neonatal immunoprophylaxis.
Hepatitis C: Vertical transmission 5-6% (higher with HIV coinfection — 10-20%, and with higher viral loads). No interventions are currently proven to reduce vertical transmission (no vaccine, no immunoglobulin). Direct-acting antivirals (DAAs — sofosbuvir/ledipasvir, glecaprevir/pibrentasvir) are not yet approved in pregnancy, although studies are ongoing (STORC trial). Mode of delivery does not affect transmission — cesarean is not protective. Avoid prolonged rupture of membranes (> 6 hours may increase risk) and fetal scalp electrode when possible. Breastfeeding is not contraindicated unless nipples are cracked or bleeding. Test infant with HCV RNA at ≥ 2 months (earliest) or anti-HCV at ≥ 18 months (maternal antibodies persist before this).
Varicella-Zoster Virus (VZV): Primary varicella (chickenpox) in pregnancy carries risk of maternal complications (varicella pneumonia — 10-20% mortality if untreated in pregnancy) and fetal effects: congenital varicella syndrome (maternal infection at 8-20 weeks — 0.4-2% risk — limb hypoplasia, cicatricial skin scarring, eye anomalies, cortical atrophy, microcephaly) and neonatal varicella (maternal rash onset 5 days before to 2 days after delivery — 17-30% attack rate, 7% mortality). Management: varicella-zoster immune globulin (VariZIG) within 10 days of exposure for susceptible pregnant women; acyclovir/valacyclovir for active infection. Vaccination is a live vaccine — contraindicated in pregnancy, but inadvertent administration has not shown teratogenicity. Ensure immunity before conception (two doses recommended).
Group B Streptococcus (GBS)
GBS colonizes 10-30% of pregnant women. Universal screening with vaginal-rectal culture at 36+0-37+6 weeks. Intrapartum antibiotic prophylaxis (IAP) indications: positive GBS culture, GBS bacteriuria in current pregnancy, prior infant with GBS disease, or unknown GBS status with risk factors (preterm labor < 37 weeks, ROM ≥ 18 hours, intrapartum fever ≥ 38.0°C). Preferred agent: penicillin G 5 million units IV loading then 2.5-3 million units q4h until delivery. Adequate prophylaxis requires ≥ 4 hours of antibiotic administration before delivery.
13 Fetal Growth Abnormalities
Fetal Growth Restriction (FGR)
Fetal growth restriction is defined as estimated fetal weight (EFW) < 10th percentile for gestational age. It affects 5-10% of pregnancies and is a leading cause of perinatal morbidity and mortality. Distinction from constitutionally small fetus is important — a growth-restricted fetus shows declining growth velocity and/or abnormal Doppler findings.
Early-onset FGR (< 32 weeks): Strongly associated with defective placentation and preeclampsia. Severe placental insufficiency with progressive Doppler deterioration: elevated UA S/D → AEDF → REDF → abnormal ductus venosus → fetal demise. Sequential monitoring allows optimization of delivery timing. Management is dictated by Doppler staging and gestational age.
Late-onset FGR (≥ 32 weeks): More common (70-80% of FGR). Milder placental dysfunction. UA Doppler is often normal (lower sensitivity). CPR < 1.0 is more sensitive for detecting redistribution. Higher risk of sudden fetal death without preceding Doppler deterioration. The TRUFFLE and PORTO studies guide management.
Doppler-Based Staging & Delivery Timing for Early-Onset FGR
| Doppler Finding | Stage | Monitoring Frequency | Delivery Timing |
|---|---|---|---|
| Elevated UA PI (> 95th percentile), normal DV | I | Weekly | 37 weeks |
| Absent end-diastolic flow (AEDF) in UA | II | 2-3x/week | 34 weeks (after steroids) |
| Reversed end-diastolic flow (REDF) in UA OR DV PI > 95th percentile | III | 1-2x/day | 30-32 weeks (after steroids) |
| Reversed a-wave in DV OR spontaneous decelerations | IV | Continuous | 26-28 weeks (individualize) |
Macrosomia
Macrosomia is defined as birth weight ≥ 4,000 g (some use ≥ 4,500 g). Risk factors: maternal diabetes (most important modifiable risk factor), obesity, excessive weight gain, post-dates, multiparity, male fetus. Complications: shoulder dystocia (risk ~1-3% at 4,000-4,499 g, 5-9% at 4,500-4,999 g, 20-50% at ≥ 5,000 g), brachial plexus injury, clavicle fracture, maternal perineal lacerations, postpartum hemorrhage. Planned cesarean is recommended for EFW ≥ 5,000 g in non-diabetic women and ≥ 4,500 g in diabetic women. Ultrasound estimation of fetal weight has an inherent error of ±15-20%, which limits the accuracy of macrosomia prediction.
14 Fetal Structural Anomalies
Central Nervous System Anomalies
Anencephaly: Failure of closure of the rostral neural tube. Absence of the cranial vault and cerebral hemispheres. Universally lethal. Diagnosed by first trimester (absent calvarium). Elevated MSAFP. Folic acid supplementation (0.4-4 mg/day) reduces NTD risk by 50-70%.
Spina bifida (myelomeningocele): Failure of closure of the caudal neural tube. Open spina bifida: meninges and neural tissue protrude through vertebral defect. Associated with Arnold-Chiari II malformation (cerebellar tonsillar herniation), hydrocephalus (90%), and lower extremity motor/sensory deficits. US signs: "lemon sign" (scalloping of frontal bones) and "banana sign" (cerebellar compression). Elevated MSAFP. Candidate for fetal surgery (see MOMS trial, Section 25).
Ventriculomegaly: Lateral ventricular atrial width ≥ 10 mm. Mild (10-12 mm): isolated mild ventriculomegaly has 85-90% normal outcome — follow serially and offer genetic testing. Moderate (13-15 mm): higher association with genetic abnormalities and structural defects. Severe (≥ 15 mm, hydrocephalus): significant risk of neurodevelopmental impairment, often associated with other anomalies. Workup: detailed anatomy scan, MRI (fetal MRI superior to US for CNS evaluation), microarray, CMV testing (TORCH).
Dandy-Walker malformation: Complete or partial agenesis of the cerebellar vermis with cystic dilation of the fourth ventricle and enlarged posterior fossa. Associated with hydrocephalus (80%), other CNS anomalies, cardiac defects, and chromosomal abnormalities (trisomy 18, trisomy 13). US: cystic posterior fossa mass with absent or hypoplastic vermis. Prognosis depends on associated anomalies — isolated Dandy-Walker has better prognosis than previously thought.
Holoprosencephaly: Failure of the prosencephalon (forebrain) to divide into two hemispheres. Spectrum: alobar (most severe — single midline ventricle, fused thalami, absent falx), semilobar, lobar. Strongly associated with trisomy 13, trisomy 18, and midline facial defects (cyclopia, proboscis, cleft lip/palate). "The face predicts the brain." Alobar form is lethal.
Cardiac Anomalies
Congenital heart defects are the most common structural anomalies (~8 per 1,000 live births). The four-chamber view on screening US detects only ~50% of CHD; adding outflow tract views and the three-vessel view increases detection to ~80-90%. Fetal echocardiography is indicated for: increased NT, family history of CHD, pregestational diabetes, fetal arrhythmia, extracardiac anomaly, abnormal screening views, and teratogen exposure.
Ventricular septal defect (VSD): Most common CHD. Membranous (70%), muscular (20%), inlet, or outlet. Small muscular VSDs often close spontaneously. Large VSDs cause heart failure if unrepaired. Hypoplastic left heart syndrome (HLHS): Underdevelopment of the left ventricle, mitral valve, aortic valve, and ascending aorta. Fatal without intervention. Requires staged surgical palliation (Norwood, Glenn, Fontan) or cardiac transplantation. The four-chamber view will show a small left ventricle.
Abdominal Wall Defects
Omphalocele: Midline defect at the umbilical ring with herniated viscera (liver, bowel) COVERED by a membrane (peritoneum + amnion). Cord inserts into the sac. Associated with chromosomal abnormalities (trisomy 18, trisomy 13 — up to 50%), cardiac defects, Beckwith-Wiedemann syndrome. Offer karyotype/microarray.
Gastroschisis: Right-sided paraumbilical defect with eviscerated bowel WITHOUT a covering membrane. Cord insertion is normal (to the left of the defect). Rarely associated with chromosomal anomalies (< 1%). Risk factor: young maternal age. Complications: bowel atresia, volvulus, perforation. Delivery at 37 weeks (or earlier for complications). Surgical closure within hours of birth.
Renal Anomalies
Hydronephrosis: The most common prenatal urinary anomaly (1-5% of pregnancies). Grading (SFU): mild (renal pelvis dilation only), moderate (calyceal dilation), severe (parenchymal thinning). Most mild-moderate cases resolve postnatally. Severe bilateral hydronephrosis with oligohydramnios: consider obstructive uropathy (posterior urethral valves in males). Bilateral renal agenesis (Potter sequence): Lethal — absent kidneys, severe oligohydramnios, pulmonary hypoplasia, limb/facial compression deformities. Posterior urethral valves: Most common cause of lower urinary tract obstruction in males. US: bilateral hydronephrosis, distended bladder, "keyhole sign." May be candidate for fetal vesicoamniotic shunting if early and progressive.
Renal Anomalies — Additional Detail
Multicystic dysplastic kidney (MCDK): Non-functioning kidney with multiple non-communicating cysts of varying sizes (largest at periphery). The most common cystic renal disease in the fetus. Unilateral MCDK has excellent prognosis (contralateral kidney compensates). Bilateral MCDK is lethal (no functioning renal tissue). Must be distinguished from hydronephrosis (which has communicating dilated calyces centered on a dilated pelvis) and autosomal recessive polycystic kidney disease (ARPKD — bilateral enlarged echogenic kidneys with oligohydramnios).
Congenital diaphragmatic hernia (CDH): While classified as a thoracic anomaly, CDH is a critical fetal surgical consideration. Incidence: 1 in 2,500. Left-sided in 80-85%. US: herniated abdominal viscera (stomach, bowel, liver) in the thorax with mediastinal shift. The key prognostic factor is pulmonary hypoplasia, quantified by lung-to-head ratio (LHR) and observed/expected LHR. O/E LHR < 25% with liver herniation: severe (survival ~15-20% with standard postnatal care). Fetoscopic endoluminal tracheal occlusion (FETO) via balloon placement may improve lung growth in severe cases — the TOTAL trial showed benefit in severe left CDH. Delivery should be at a center with ECMO capability.
Skeletal Dysplasias
Skeletal dysplasias are a heterogeneous group of > 450 conditions. Key US findings: shortened long bones (rhizomelic — proximal, mesomelic — middle, or acromelic — distal), fractures, bowing, absent mineralization, and abnormal thoracic dimensions. The femur length/abdominal circumference (FL/AC) ratio and femur length/biparietal diameter (FL/BPD) ratio help identify short-limbed conditions. A narrow thorax with a thoracic circumference < 5th percentile for GA predicts lethal pulmonary hypoplasia.
Thanatophoric dysplasia (most common lethal skeletal dysplasia): severe micromelia, narrow thorax, cloverleaf skull (type II), platyspondyly, FGFR3 mutation. Lethal due to pulmonary hypoplasia. Achondroplasia (most common non-lethal skeletal dysplasia): rhizomelic shortening, frontal bossing, trident hands, FGFR3 mutation (different from thanatophoric). Often not detectable until late second/third trimester because shortening becomes more pronounced as growth progresses. Osteogenesis imperfecta type II (lethal): multiple fractures in utero, decreased mineralization, crumpled/accordion-like long bones, blue sclerae. Type I and IV are non-lethal with variable severity.
15 Fetal Chromosomal & Genetic Conditions
Trisomy 21 (Down Syndrome)
Most common viable autosomal trisomy (1 in 700 live births). Risk increases with maternal age (1 in 1,250 at age 25; 1 in 350 at age 35; 1 in 100 at age 40). US markers: increased NT, absent or hypoplastic nasal bone, shortened humerus and femur, echogenic bowel, echogenic intracardiac focus, mild ventriculomegaly, mild pyelectasis, sandal gap, clinodactyly. Cardiac defects in 40-50% (AVSD most characteristic, also VSD, ASD, tetralogy of Fallot). Duodenal atresia (double bubble sign).
Trisomy 18 (Edwards Syndrome)
Incidence: 1 in 5,000-8,000. Median survival: 5-15 days. US findings: IUGR, clenched hands with overlapping fingers (index over third, fifth over fourth), strawberry-shaped skull, choroid plexus cysts, omphalocele, cardiac defects (VSD, ASD, DORV), single umbilical artery, rocker-bottom feet. Screening: all serum markers low on quad screen; cfDNA detection rate 97-99%.
Trisomy 13 (Patau Syndrome)
Incidence: 1 in 10,000-16,000. Median survival: 7-10 days. US findings: holoprosencephaly, midline facial defects (cleft lip/palate, cyclopia, proboscis), polydactyly, cardiac defects (80%), echogenic kidneys, omphalocele. Screening: cfDNA detection rate 92-99%.
Turner Syndrome (45,X)
Incidence: 1 in 2,500 female births (but ~99% of 45,X conceptions end in miscarriage). Prenatal US findings: cystic hygroma (large, septated, posterior cervical), hydrops, coarctation of the aorta, horseshoe kidney, short femur. Postnatal features: short stature, webbed neck, widely spaced nipples, ovarian failure, cardiac defects (bicuspid aortic valve 30%, coarctation 10%). cfDNA detection rate 90-95%.
Microarray Findings & Single Gene Disorders
Chromosomal microarray detects clinically significant copy number variants (CNVs) in ~6% of fetuses with structural anomalies and ~1.7% of structurally normal fetuses beyond what karyotype identifies. Common pathogenic CNVs: 22q11.2 deletion (DiGeorge — conotruncal cardiac defects, thymic hypoplasia, cleft palate), 1p36 deletion, 15q11-q13 deletion (Prader-Willi/Angelman), 7q11.23 deletion (Williams syndrome), 5p deletion (Cri-du-chat). For fetuses with anomalies and normal microarray, exome sequencing has a diagnostic yield of ~10-30%, particularly for skeletal dysplasias, multiple congenital anomalies, and hydrops of unknown etiology.
Consanguinity
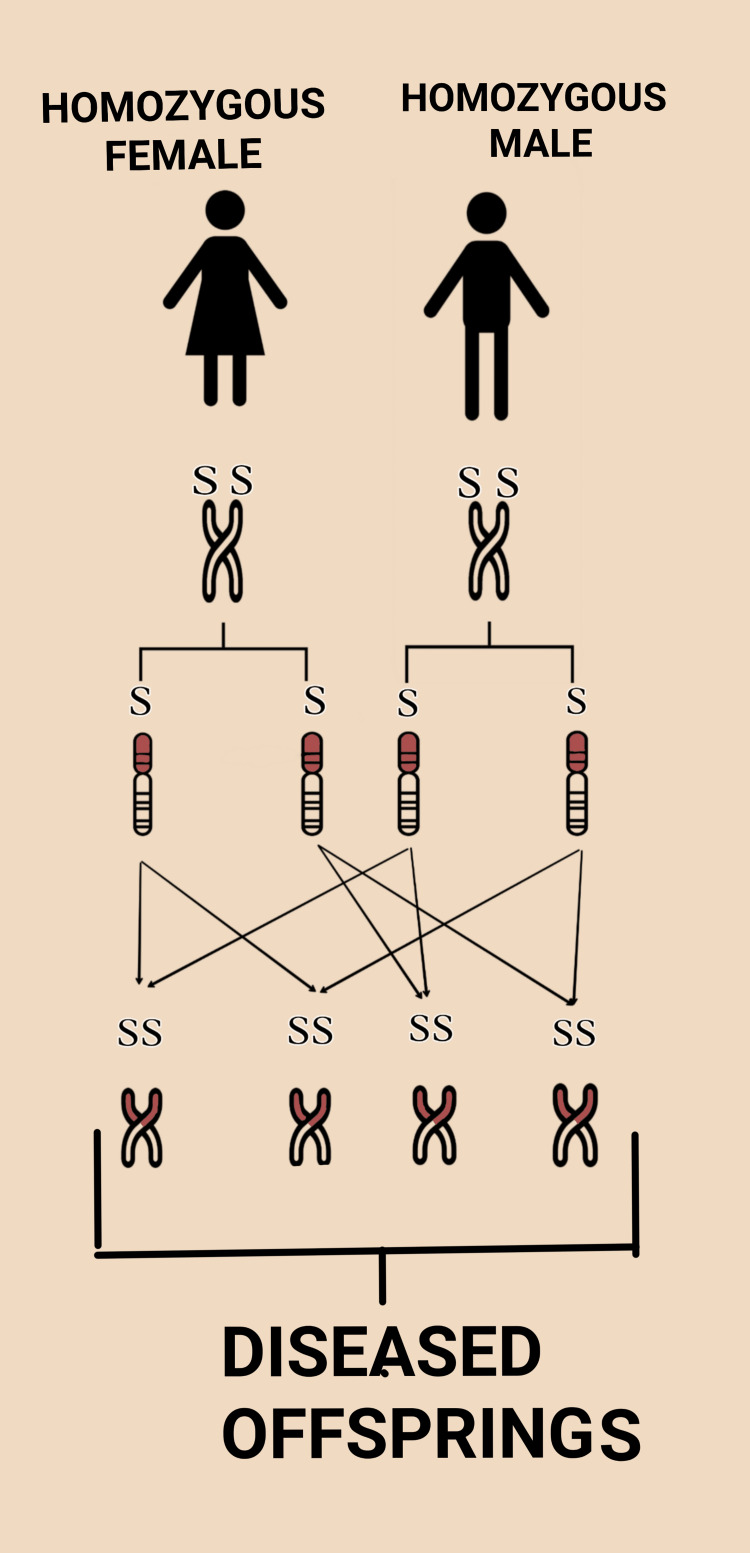
Consanguineous unions (first cousins: coefficient of inbreeding = 1/16) increase the risk of autosomal recessive conditions by increasing the likelihood of homozygosity for deleterious alleles. Genetic counseling should include: expanded carrier screening, detailed anatomy scan, consideration of exome/genome sequencing if anomalies are detected. The absolute risk of birth defects in first-cousin unions is approximately 4-7% (vs 2-3% in the general population).
Carrier Screening
ACOG recommendations: All patients should be offered carrier screening. Options include: ethnicity-based screening (CF for Caucasians, sickle cell for African Americans, Tay-Sachs for Ashkenazi Jewish patients, thalassemia for Mediterranean/Southeast Asian descent) or expanded carrier screening (ECS) panels — which simultaneously screen for > 100-300 autosomal recessive conditions regardless of ethnicity. ECS has largely replaced ethnicity-based screening in many practices. If both partners are carriers for the same autosomal recessive condition, there is a 25% chance of an affected offspring per pregnancy. Genetic counseling, prenatal diagnosis (CVS/amniocentesis), and preimplantation genetic testing (PGT-M with IVF) should be offered.
Cystic fibrosis: Most common lethal autosomal recessive condition in Caucasians. Carrier frequency: 1 in 25 (Caucasian). > 2,000 known CFTR mutations; ΔF508 most common (~70% of CF alleles). If one partner is a carrier and the other tests negative, there is a residual carrier risk that depends on ethnicity and the number of mutations screened (for Caucasians, ~1 in 240 with a negative expanded panel).
Spinal muscular atrophy (SMA): Carrier frequency: ~1 in 40-60 across ethnicities. Caused by homozygous deletion of SMN1 gene. ~2% of carriers have two copies on one chromosome (duplication, making detection difficult). Universal carrier screening is now recommended (ACOG 2017). Treatment of affected neonates with nusinersen or onasemnogene has dramatically improved outcomes, making early detection particularly important.
16 Multiple Gestations
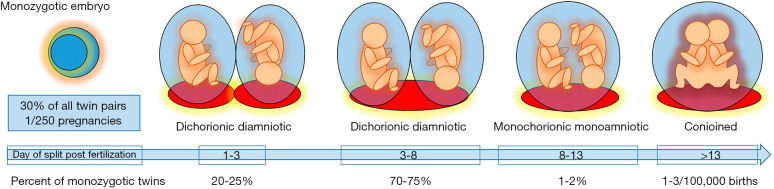
Chorionicity Determination
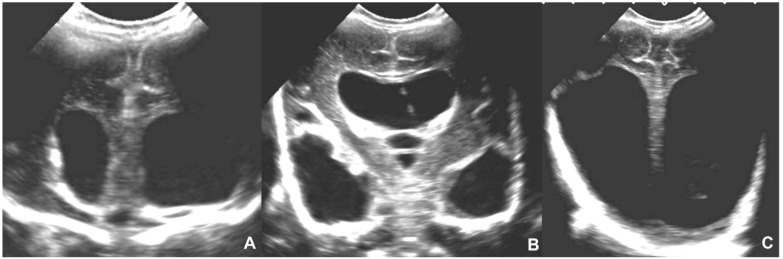
Determining chorionicity is the single most important step in managing twin pregnancies, ideally in the first trimester. Dichorionic-diamniotic (DCDA): "Lambda sign" (twin peak sign) — triangular projection of placental tissue extending between the membranes at the placental surface. Two separate placentas or thick dividing membrane. Monochorionic-diamniotic (MCDA): "T sign" — thin membrane inserts directly into the placental surface without an intervening wedge of tissue. Shared placenta. Monochorionic-monoamniotic (MCMA): No dividing membrane. All monochorionic twins share vascular anastomoses in the placenta, which are the basis for TTTS and other monochorionic-specific complications.
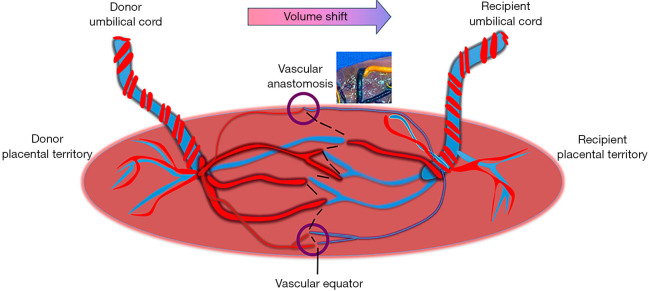
Monochorionic Twin Complications
TTTS results from unbalanced blood flow through placental arteriovenous anastomoses — the donor twin becomes hypovolemic (oligo, small bladder) and the recipient becomes hypervolemic (polyhydramnios, large bladder, eventual hydrops). Complicates 10-15% of MCDA twins.
Stage I: Polyhydramnios (MVP > 8 cm) in recipient + oligohydramnios (MVP < 2 cm) in donor. Donor bladder still visible.
Stage II: Stage I + absent bladder in donor twin (critically volume-depleted).
Stage III: Stage II + critically abnormal Doppler: absent/reversed AEDF in UA of either twin, reversed flow in DV, or pulsatile umbilical venous flow.
Stage IV: Hydrops in one or both twins.
Stage V: Demise of one or both twins.
Treatment: Stage I — serial monitoring (some resolve spontaneously). Stage II-IV — fetoscopic laser photocoagulation of placental anastomoses (Solomon technique — continuous line of coagulation connecting the anastomoses). Amnioreduction is a temporizing measure. The EUROFETUS trial demonstrated laser superiority over amnioreduction (survival and neurologic outcomes).
Twin anemia-polycythemia sequence (TAPS): Chronic, slow transfusion through tiny (< 1 mm) anastomoses causing large inter-twin hemoglobin difference (> 8 g/dL at birth) without amniotic fluid discordance. Diagnosed prenatally by MCA-PSV discordance (donor > 1.5 MoM, recipient < 1.0 MoM). Can occur spontaneously (3-5% of MCDA) or post-laser (13-16%).
Twin reversed arterial perfusion (TRAP) sequence: One twin (the "pump twin") perfuses a nonviable acardiac twin through reversed arterial flow. The acardiac mass acts as a parasitic shunt. Risk to the pump twin: high-output cardiac failure and hydrops. Treatment: radiofrequency ablation or bipolar coagulation of the cord to the acardiac twin when the mass approaches the size of the pump twin or signs of cardiac failure develop.
Selective FGR in monochorionic twins: Growth discordance > 25% (larger twin as reference). Classified by UA Doppler in the smaller twin: Type 1 (positive end-diastolic flow — favorable), Type 2 (persistent AEDF/REDF — unfavorable), Type 3 (intermittent AEDF/REDF — unpredictable). Management ranges from surveillance (Type 1) to laser/selective reduction/delivery (Type 2/3 depending on gestational age).
Dichorionic Twin Management
DCDA twins have lower risks than MCDA twins but still have increased rates of preterm birth (50% deliver < 37 weeks), preeclampsia (13% vs 5% in singletons), FGR (especially discordant growth), and gestational diabetes compared to singletons. Surveillance: growth US every 4 weeks. Growth discordance > 20% (calculated as [larger EFW - smaller EFW] / larger EFW × 100) warrants increased surveillance and evaluation for FGR in the smaller twin.
| Twin Type | Surveillance | Delivery Timing | Special Considerations |
|---|---|---|---|
| DCDA (uncomplicated) | Growth US q4 weeks; NST at 36 weeks | 38+0 weeks | Lower risk; manage as two independent singletons sharing a uterus |
| MCDA (uncomplicated) | US q2 weeks from 16 weeks (TTTS screening); growth q4 weeks; NST at 32 weeks | 36+0 weeks | Risk of TTTS, TAPS, selective FGR; lower threshold for delivery |
| MCMA | Inpatient monitoring from 24-28 weeks; daily NST; US q2 weeks | 32-34 weeks (after steroids) | Risk of cord entanglement (cord intertwining); elective cesarean delivery recommended |
Mode of delivery: Vaginal delivery is appropriate for DCDA and MCDA twins if the presenting twin (Twin A) is vertex, regardless of Twin B presentation, at centers experienced in breech extraction of the second twin. Cesarean is recommended for MCMA twins (cord entanglement risk during labor) and when Twin A is non-vertex.
17 Hydrops Fetalis
Definition & Classification
Hydrops fetalis is the pathologic accumulation of fluid in ≥ 2 fetal body compartments (ascites, pleural effusion, pericardial effusion, skin edema > 5 mm). Immune hydrops (10-15% of cases): caused by maternal red cell alloimmunization (anti-D, anti-Kell, anti-c) leading to fetal hemolytic anemia. Non-immune hydrops (85-90%): caused by a wide variety of etiologies — the differential is extensive.
Causes of Non-Immune Hydrops
Cardiovascular (20-25%): Structural heart defects (HLHS, Ebstein anomaly, AV canal), arrhythmias (SVT, complete heart block), cardiomyopathy, high-output failure (sacrococcygeal teratoma, vein of Galen malformation, TTTS recipient, chorioangioma, AV malformation).
Chromosomal/genetic (10-15%): Turner syndrome (45,X — most common chromosomal cause), trisomy 21, trisomy 18, Noonan syndrome, storage diseases (Gaucher, Niemann-Pick).
Hematologic (10-15%): Alpha-thalassemia (homozygous — Hb Bart's), parvovirus B19 infection (aplastic crisis), fetomaternal hemorrhage, G6PD deficiency.
Infection (5-10%): Parvovirus B19 (most common infectious cause), CMV, toxoplasmosis, syphilis.
Thoracic (5-10%): CCAM/CPAM, pulmonary sequestration, congenital diaphragmatic hernia, chylothorax.
Twin-related: TTTS (recipient), TAPS, TRAP.
Idiopathic: 15-25% remain unexplained despite extensive workup.
Workup
Systematic evaluation: Maternal — blood type and antibody screen (Coombs), Kleihauer-Betke (fetomaternal hemorrhage), TORCH serologies (parvovirus B19 IgM/IgG, CMV, toxoplasmosis, syphilis), hemoglobin electrophoresis (alpha-thalassemia carrier status), and metabolic screening. Fetal — detailed anatomy US and fetal echocardiography, MCA-PSV Doppler (fetal anemia), karyotype/microarray (± exome), and if indicated, cordocentesis (CBC, reticulocyte count, direct Coombs, viral PCR). MRI may be helpful for thoracic and CNS anomalies.
Management
Directed at the underlying cause when possible:
| Etiology | Treatment | Prognosis |
|---|---|---|
| Fetal anemia (parvovirus, alloimmunization) | Intrauterine transfusion | Good if treated early (80-90% survival for IUT in alloimmunization) |
| Fetal SVT/tachyarrhythmia | Transplacental antiarrhythmics: digoxin (first-line), flecainide, sotalol, or amiodarone | Good (85-95% conversion rate with treatment) |
| Large pleural effusion (chylothorax) | Thoracoamniotic shunt | 70% resolution if primary chylothorax |
| TTTS (recipient) | Fetoscopic laser photocoagulation | 70-80% survival at least one twin |
| LUTO with oligohydramnios | Vesicoamniotic shunt | Variable — depends on residual renal function |
| Chromosomal/structural anomaly | Counseling, comfort care vs intervention based on specific diagnosis | Poor for lethal anomalies |
| Congenital CMV | Valacyclovir (maternal); limited fetal treatment options | Poor with hydrops; better with mild disease |
Prognosis depends heavily on etiology — chromosomal causes and severe structural defects have a poor prognosis (mortality > 90%). Treatable causes (anemia, arrhythmia) have much better outcomes. Idiopathic hydrops has an overall survival of ~50-60%. The gestational age at presentation also matters: hydrops before 20 weeks carries worse prognosis than later onset. Overall perinatal mortality for hydrops is 50-90% depending on the cause and gestational age at diagnosis.
18 Fetal Demise
Definition & Evaluation
Intrauterine fetal demise (IUFD) is defined as fetal death at ≥ 20 weeks gestation (or birth weight ≥ 350 g if gestational age is unknown). Incidence: ~6 per 1,000 births. Diagnosis: absent fetal cardiac activity on ultrasound. Common causes: placental insufficiency (abruption, FGR, preeclampsia), umbilical cord abnormalities, fetal anomalies/aneuploidy, infection, maternal medical disease, and unexplained (25-60%).
Fetal evaluation: Karyotype and/or microarray (tissue from placenta or amniotic fluid is preferred — fetal blood may not grow in culture), external examination, and autopsy (highest diagnostic yield — identifies cause in 30-40% of otherwise unexplained cases). Fetal photographs.
Placental evaluation: Gross and histopathologic examination (identifies cause in 40-65% when abnormalities are found — chronic villitis, massive perivillous fibrin deposition, fetal vascular malperfusion, maternal vascular malperfusion).
Maternal evaluation: CBC, blood type and antibody screen, Kleihauer-Betke test (fetomaternal hemorrhage), thrombophilia panel (lupus anticoagulant, anticardiolipin, anti-β2 glycoprotein I, factor V Leiden, prothrombin mutation), TSH, HbA1c, toxicology screen, TORCH serologies, bile acids, antinuclear antibody. RPR/VDRL.
Management of IUFD
Delivery is recommended after diagnosis, though timing is individualized (most women prefer prompt delivery). Methods: induction of labor is preferred (vaginal delivery is appropriate at any gestational age). Cervical ripening with misoprostol: 200-400 μg vaginally q4-6h (< 28 weeks — higher doses may be used); 25-50 μg vaginally q4h (≥ 28 weeks, lower doses if prior uterine scar). Mifepristone 200 mg followed by misoprostol increases efficacy for second-trimester fetal demise. Mechanical dilation (balloon catheter) is an alternative. DIC may develop if IUFD is retained for > 3-4 weeks (consumptive coagulopathy from tissue thromboplastin release) — check fibrinogen, PT/INR, platelet count weekly if expectant management is chosen.
Recurrence Counseling
Recurrence risk of stillbirth in a subsequent pregnancy is approximately 2-10× the baseline risk (depending on cause). Modifiable risk factors should be addressed (optimize diabetes control, treat hypertension, aspirin for preeclampsia prevention, smoking cessation, weight management). Subsequent pregnancy management: early and serial growth ultrasounds (starting at 24-28 weeks, every 3-4 weeks), antepartum testing beginning at 32 weeks (or 1-2 weeks before the gestational age of the prior loss), low-dose aspirin if indicated, and delivery at 37-39 weeks (balancing prematurity risk against stillbirth risk). Psychosocial support is essential — subsequent pregnancies after stillbirth are associated with significant maternal anxiety, PTSD, and depression.
19 Preterm Birth & Cervical Insufficiency
Preterm Birth — Epidemiology & Risk Factors
Preterm birth (< 37 weeks) complicates ~10% of pregnancies and is the leading cause of neonatal morbidity and mortality. Risk factors: prior spontaneous preterm birth (strongest predictor — recurrence risk 15-50%), short cervical length, multiple gestation, uterine anomalies, prior cervical surgery (LEEP, cone biopsy), infection/inflammation, African American race, low BMI, tobacco use, short interpregnancy interval.
Cervical Length Screening
Universal transvaginal cervical length screening at 18-24 weeks is recommended by many professional organizations. Short cervix (< 25 mm) in a singleton without prior PTB: vaginal progesterone (200 mg suppository or 90 mg gel nightly until 36+6 weeks). Short cervix in a patient with prior PTB: ultrasound-indicated cerclage (if < 24 weeks) and progesterone.
Cerclage Types
History-indicated (prophylactic) cerclage: Placed at 12-14 weeks in women with ≥ 3 prior second-trimester losses or prior cerclage for cervical insufficiency. No cervical length monitoring needed (cerclage placed regardless).
Ultrasound-indicated cerclage: Placed when cervical length < 25 mm before 24 weeks in a woman with prior spontaneous preterm birth < 34 weeks. Serial CL monitoring from 16-24 weeks in at-risk women.
Physical exam-indicated (rescue/emergency) cerclage: Placed when cervical dilation is discovered on exam (typically 1-4 cm) with exposed membranes, in the absence of labor, infection, or bleeding, at 16-23+6 weeks. Higher complication rate but still beneficial.
McDonald cerclage: Most common. Purse-string suture around the cervix at the cervicovaginal junction. Removed at 36-37 weeks.
Shirodkar cerclage: Submucosal placement — bladder reflected superiorly and rectum inferiorly. May be left in place for cesarean delivery.
Transabdominal cerclage: Placed at the level of the internal os via laparotomy or laparoscopy. Reserved for patients who have failed transvaginal cerclage or have very short or absent cervix (prior trachelectomy). Requires cesarean delivery.
Progesterone for Preterm Birth Prevention
Vaginal progesterone (200 mg suppository or 90 mg gel nightly): Indicated for short cervix (< 25 mm) at 18-24 weeks in singleton pregnancies regardless of prior PTB history. Reduces preterm birth by ~40% (Fonseca, Hassan, OPPTIMUM trials). 17-alpha hydroxyprogesterone caproate (17-OHPC, Makena): Previously given IM weekly from 16-36 weeks for prior spontaneous PTB. The PROLONG trial (2019) failed to confirm benefit, leading to FDA withdrawal of accelerated approval. Current practice is evolving — vaginal progesterone is increasingly preferred.
Periviable Counseling
Periviable birth (20-25+6 weeks) requires multidisciplinary counseling (MFM, neonatology, social work). Survival and intact survival by gestational age (approximate, varies by center): 22 weeks: 10-30% survival (significant morbidity); 23 weeks: 30-50%; 24 weeks: 50-70%; 25 weeks: 70-80%. Key considerations: antenatal corticosteroids (typically offered from 23-24 weeks, though some centers offer at 22 weeks), mode of delivery (cesarean for fetal indication at periviable GA is controversial), comfort care vs active resuscitation. Family wishes are paramount.
20 Preterm Premature Rupture of Membranes
Definition & Epidemiology
PPROM occurs in ~3% of pregnancies and accounts for approximately 30-40% of all preterm births. It is distinct from preterm labor (which presents with contractions and cervical change with intact membranes). Risk factors: prior PPROM (recurrence 13-32%), genital tract infection (BV, GBS, Chlamydia), short cervical length, smoking, polyhydramnios, multiple gestation, prior cervical surgery, amniocentesis, and cerclage.
Diagnosis
PPROM is rupture of membranes before 37 weeks and before the onset of labor. Diagnosis is confirmed by the classic triad: pooling of amniotic fluid in the posterior vaginal vault on sterile speculum exam, positive nitrazine test (amniotic fluid is alkaline, pH 7.1-7.3, turns nitrazine paper blue — false positives with blood, semen, BV, cervical mucus), and ferning of dried vaginal fluid on microscopy (arborization pattern of amniotic fluid crystallized on a glass slide). Newer immunoassay tests include AmniSure (placental alpha-macroglobulin-1, PAMG-1 — sensitivity 98.9%, specificity 99.1%) and ROM Plus (IGFBP-1 + AFP). These are especially useful in equivocal presentations. Avoid digital cervical exams — use sterile speculum exam only (digital exams decrease latency and increase infection risk). Assess for cord prolapse, collect GBS culture, and obtain cervical length by ultrasound if indicated.
Management by Gestational Age
| GA | Management | Key Considerations |
|---|---|---|
| < 23 weeks (previable) | Counseling: expectant management vs induction. If expectant: antibiotics controversial, serial surveillance | High risk of infection, pulmonary hypoplasia, limb contractures. ~30-40% reach viability; of those, ~30-50% survive |
| 23+0-33+6 weeks | Expectant management: latency antibiotics, antenatal corticosteroids, GBS prophylaxis, MgSO4 for neuroprotection if < 32 weeks | Deliver for: chorioamnionitis, non-reassuring fetal status, cord prolapse, abruption, advanced labor |
| 34+0-36+6 weeks | Delivery recommended (PPROMEXIL-2 and similar trials) | Administer GBS prophylaxis. Consider late preterm steroids if not previously given (ALPS trial). Avoid MgSO4 for neuroprotection at this GA. |
Latency Antibiotics
Antibiotic prophylaxis extends the latency period and reduces neonatal infection. The ORACLE I regimen: ampicillin 2 g IV q6h + erythromycin 250 mg IV q6h for 48 hours, followed by amoxicillin 250 mg PO q8h + erythromycin 250 mg PO q8h for 5 days (total 7-day course). Alternative: ampicillin + azithromycin 1 g PO × 1 (increasingly used, simpler). Avoid amoxicillin-clavulanate (augmentin) — associated with increased risk of necrotizing enterocolitis in the neonate (ORACLE I trial finding).
GBS Prophylaxis in PPROM
If GBS status is positive or unknown: administer intrapartum GBS prophylaxis (penicillin G). If GBS culture is negative within the past 5 weeks and no other indication exists, GBS prophylaxis may be withheld. Re-culture at presentation with PPROM if status is unknown and results are pending.
21 Placenta Accreta Spectrum
Classification
Placenta accreta spectrum (PAS) describes abnormally adherent or invasive placentation resulting from defective decidua basalis. Risk increases dramatically with the number of prior cesarean deliveries, particularly with concurrent placenta previa.
Placenta accreta: Villi attach directly to the myometrium without intervening decidua (most common, ~75% of PAS).
Placenta increta: Villi invade into the myometrium (~15% of PAS).
Placenta percreta: Villi penetrate through the myometrium to the serosa or adjacent organs (bladder, bowel) (~10% of PAS). Highest morbidity.
Risk factors: Prior cesarean delivery (strongest risk factor), placenta previa, prior uterine surgery (myomectomy, D&C), advanced maternal age, multiparity, Asherman syndrome, IVF. Risk of PAS with placenta previa: no prior CD — 3%; 1 prior CD — 11%; 2 prior CDs — 40%; 3 prior CDs — 61%; ≥ 4 prior CDs — 67%.
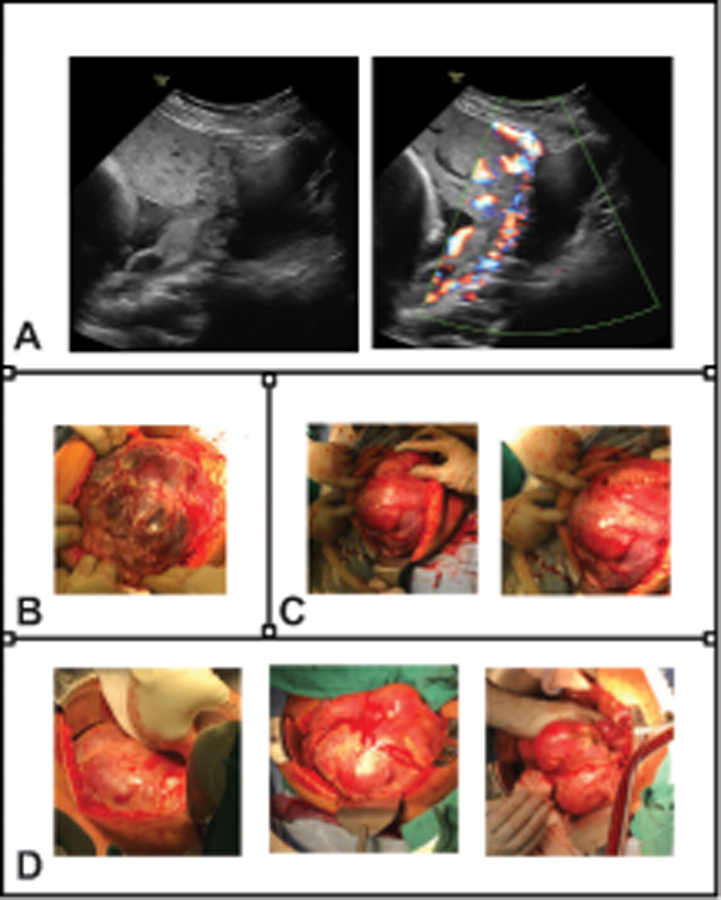
Diagnosis
Ultrasound findings: Loss of the retroplacental clear zone, irregular placental lacunae ("Swiss cheese" appearance — most predictive US finding), myometrial thinning, disruption of the bladder-uterine interface, bridging vessels crossing the uterine-bladder interface. Color Doppler: turbulent flow in lacunae, hypervascularity at the bladder interface. MRI is complementary — most useful for posterior placentas and to assess depth/extent of invasion (especially percreta involving the bladder or parametrium). Sensitivity of US: 77-97%; MRI adds value primarily in equivocal cases.
Management
Planned cesarean hysterectomy at 34-36 weeks (after antenatal corticosteroids) at a center of excellence with a multidisciplinary team (MFM, gynecologic oncology, anesthesia, urology, interventional radiology, blood bank). No attempt at manual placental removal (dramatically increases hemorrhage). Massive transfusion protocol should be activated proactively. Average blood loss: 3,000-5,000 mL. Conservative management (leaving the placenta in situ) is reserved for select cases with patient desire for future fertility — requires close follow-up and carries risk of delayed hemorrhage, infection, DIC, and sepsis.
22 Obstetric Hemorrhage
Antepartum Hemorrhage
Placental abruption: Premature separation of a normally implanted placenta from the uterine wall. Risk factors: chronic HTN (most important), prior abruption (recurrence 5-17%), cocaine use, trauma, PPROM, advanced maternal age, smoking. Presentation: vaginal bleeding (80%), abdominal/back pain, uterine tenderness, contractions, non-reassuring fetal heart tracing. Concealed hemorrhage (20%) can present with hemodynamic instability without visible bleeding. US sensitivity for abruption is only 25-50% (a negative US does NOT rule out abruption). Severe abruption can trigger DIC (release of tissue thromboplastin). Management: stabilize, transfuse, deliver. If mild and preterm (< 34 weeks): consider expectant management with close surveillance after steroids.
Placenta previa: Placenta covering or within 2 cm of the internal cervical os. Incidence: 0.3-0.5%. Risk factors: prior cesarean, prior previa, multiparity, multiple gestation, advanced maternal age, smoking, IVF. Presents with painless vaginal bleeding (typically second/third trimester). Management: pelvic rest, serial US (many low-lying placentas "migrate" as the lower uterine segment develops — previa diagnosed at 20 weeks resolves in 90% by term). Persistent previa: cesarean delivery at 36-37 weeks (after steroids if < 37 weeks). Emergent delivery for life-threatening hemorrhage regardless of GA.
Vasa previa: Fetal vessels crossing or within 2 cm of the internal os, unprotected by the umbilical cord or placental tissue. Types: Type 1 — velamentous cord insertion with vessels traversing the lower segment; Type 2 — vessels connecting between lobes of a succenturiate or bilobed placenta. Risk of fetal exsanguination with rupture of membranes (fetal mortality > 50% if undiagnosed). Prenatal diagnosis is life-saving — US with color Doppler identifies the vessels. Management: antenatal corticosteroids, inpatient admission at 30-34 weeks, and planned cesarean delivery at 34-37 weeks (before labor/ROM). Sinusoidal FHR pattern with ROM and vaginal bleeding = vasa previa rupture until proven otherwise.
Postpartum Hemorrhage & DIC in Pregnancy
Postpartum hemorrhage (PPH) is defined as blood loss ≥ 1,000 mL or bleeding with hemodynamic instability regardless of mode of delivery (2017 ACOG revision). Causes follow the "4 T's": Tone (uterine atony — 70-80% of PPH), Trauma (lacerations, uterine rupture), Tissue (retained placenta), Thrombin (coagulopathy). Management escalation: bimanual uterine massage, uterotonics (oxytocin, methylergonovine, carboprost, misoprostol), intrauterine tamponade (balloon), uterine compression sutures (B-Lynch), uterine artery embolization, and hysterectomy as last resort.
Oxytocin: 10-40 units in 1 L NS, run at 150-500 mL/h IV. First-line for uterine atony. Avoid IV bolus (can cause hypotension).
Methylergonovine (Methergine): 0.2 mg IM q2-4h. Causes sustained uterine contraction. CONTRAINDICATED in hypertension (causes severe vasoconstriction/hypertensive crisis).
Carboprost (Hemabate, 15-methyl PGF2α): 0.25 mg IM q15-90 min (max 8 doses). Potent uterotonic. CONTRAINDICATED in asthma (bronchospasm). Side effects: diarrhea, fever, bronchospasm.
Misoprostol (PGE1): 600-1000 μg rectally, sublingually, or buccally. Advantage: no contraindications (can be used in hypertension and asthma). Slower onset than other agents.
Tranexamic acid (TXA): 1 g IV over 10 minutes within 3 hours of delivery. The WOMAN trial demonstrated 20% reduction in death from bleeding (NNT 267 overall, NNT 67 when given within 1 hour). Now recommended by WHO as adjunctive therapy for PPH.
DIC in pregnancy: Causes include massive abruption, amniotic fluid embolism, HELLP, AFLP, sepsis, and retained dead fetus. Amniotic fluid embolism (AFE) is a rare (1 in 40,000) catastrophic event: sudden cardiovascular collapse + DIC + respiratory failure during labor/delivery or immediately postpartum. Mortality: 20-60%. Treatment is supportive (aggressive resuscitation, blood products, vasopressors, immediate delivery if antepartum). No specific therapy exists. Lab findings in DIC: low fibrinogen (most sensitive marker in pregnancy — normal pregnancy fibrinogen is 400-600 mg/dL; a level < 200 mg/dL is highly abnormal), elevated D-dimer, prolonged PT/aPTT, low platelets, schistocytes on smear.
23 Alloimmunization
Rh Alloimmunization
Rh(D) alloimmunization occurs when an Rh-negative mother produces anti-D antibodies after exposure to Rh-positive fetal red blood cells (via fetomaternal hemorrhage at delivery, trauma, procedures, or spontaneous events). Anti-D antibodies cross the placenta and cause fetal hemolytic anemia (hemolytic disease of the fetus and newborn — HDFN). Anti-D prophylaxis (RhoGAM): 300 μg anti-D immunoglobulin IM at 28 weeks and within 72 hours of delivery (if neonate is Rh-positive). Additional doses for sensitizing events (amniocentesis, CVS, external cephalic version, abdominal trauma, vaginal bleeding). Kleihauer-Betke test to quantify fetomaternal hemorrhage — additional doses for large hemorrhages (> 30 mL fetal blood requires additional vials).
Monitoring the Alloimmunized Pregnancy
If anti-D antibody titer is ≥ 1:16 (critical titer, varies by lab), initiate serial MCA peak systolic velocity (MCA-PSV) monitoring every 1-2 weeks. MCA-PSV > 1.5 MoM for gestational age indicates moderate-severe fetal anemia with a sensitivity of ~100% and false positive rate of ~12%. This has replaced serial amniocentesis for ΔOD450 (Liley curve) as the primary surveillance method. If MCA-PSV is elevated, proceed to cordocentesis for fetal hematocrit — if anemic, perform intrauterine transfusion.
Kell Alloimmunization
Anti-Kell antibodies are the second most common cause of severe HDFN. Unlike anti-D (which causes hemolysis), anti-Kell antibodies suppress fetal erythropoiesis directly, leading to reticulocytopenia and anemia without proportionate hyperbilirubinemia. The critical titer is lower than for anti-D (often ≥ 1:8). MCA-PSV monitoring is the same. Maternal anti-Kell titers correlate poorly with disease severity — paternal/fetal Kell genotyping is critical (90% of Caucasians are Kell-negative, so the fetus may be unaffected).
Intrauterine Transfusion (IUT)
Intravascular IUT via cordocentesis (needle into the umbilical vein at the cord insertion under US guidance). Target hematocrit: 40-50%. Transfuse type O, Rh-negative, Kell-negative, CMV-negative, irradiated, leukocyte-reduced packed red blood cells. Typically needed every 2-4 weeks (interval based on expected decline in fetal hematocrit — approximately 1%/day). Delivery is usually planned at 34-37 weeks depending on the severity and need for ongoing transfusions. Complications: bradycardia (most common), bleeding, PPROM, fetal death (1-3% per procedure).
24 Amniocentesis & Chorionic Villus Sampling
Amniocentesis — Technique & Indications
Timing: ≥ 15 weeks (early amniocentesis at < 15 weeks is associated with increased talipes equinovarus and is not recommended). Technique: Under continuous ultrasound guidance, a 20-22 gauge spinal needle is inserted transabdominally into a pocket of amniotic fluid, avoiding the placenta and fetus. Approximately 20-30 mL of fluid is aspirated (1 mL per week of gestation as a guideline). The first 1-2 mL may be discarded to reduce maternal cell contamination.
Indications: Diagnostic genetic testing (after positive screening test, advanced maternal age, abnormal US findings, known carrier status), fetal lung maturity assessment (lamellar body count, L/S ratio — rarely needed in modern practice), evaluation of fetal infection (CMV, toxoplasmosis PCR), assessment of fetal hemolytic disease (ΔOD450), and therapeutic amnio-reduction (polyhydramnios, TTTS).
Risks: Procedure-related pregnancy loss: ~1 in 900 (0.11%) — this is above and beyond the background loss rate at the gestational age of the procedure. Other complications: transient vaginal bleeding or amniotic fluid leakage (1-2%, usually self-limited), chorioamnionitis (< 0.1%), needle injury to fetus (extremely rare with continuous US guidance), and Rh sensitization (administer RhoGAM 300 μg IM to all Rh-negative, unsensitized women after the procedure). Patients should be counseled about rest for 24 hours after the procedure and to report fever, persistent fluid leakage, vaginal bleeding, or contractions.
Amniocentesis for fetal lung maturity (FLM): Historically used at 32-39 weeks to guide delivery timing, but with modern neonatal care and evidence-based delivery timing guidelines, FLM testing is now rarely indicated. The lamellar body count (≥ 50,000/μL = mature), lecithin-sphingomyelin (L/S) ratio (≥ 2.0 = mature), and phosphatidylglycerol (PG present = mature) are the traditional markers. ACOG recommends against FLM testing to guide delivery timing when a clear clinical indication for delivery exists.
Chorionic Villus Sampling — Technique & Indications
Timing: 10-13+6 weeks. Technique: Transcervical (catheter through cervix to placenta under US guidance — preferred when placenta is posterior/low) or transabdominal (needle through abdominal wall — preferred when placenta is anterior/fundal). Approximately 15-30 mg of chorionic villi are obtained.
Advantages over amniocentesis: Earlier results (first trimester), allows earlier decision-making, DNA analysis does not require cell culture (direct preparation results in 24-48 hours for FISH). Limitations: 1-2% rate of confined placental mosaicism (CPM — abnormal cell line present in placenta but not fetus; if mosaicism is found, amniocentesis at ≥ 15 weeks is recommended to clarify). Cannot assess for neural tube defects (no AFP). Procedure-related loss: ~1 in 455 (0.22%). Do NOT perform before 10 weeks — risk of limb reduction defects.
25 Fetal Procedures & Surgery
Intrauterine Transfusion (IUT)
Detailed in Section 23 (Alloimmunization). Key points: intravascular approach (umbilical vein at cord insertion) under US guidance; peritoneal approach as backup if intravascular access is not possible. Type O, Rh-negative, Kell-negative, CMV-negative, irradiated, leukocyte-reduced pRBCs. Fetal paralysis (pancuronium) is often administered to facilitate the procedure.
Fetal Shunt Placement
Thoracoamniotic shunt: Indicated for large, isolated pleural effusions (chylothorax) causing mediastinal shift, hydrops, or pulmonary hypoplasia. A pigtail catheter is placed percutaneously under US guidance from the fetal pleural space into the amniotic cavity. Success rate: ~70% resolution of hydrops if etiology is primary chylothorax (vs secondary to other causes).
Vesicoamniotic shunt: Indicated for lower urinary tract obstruction (LUTO — typically posterior urethral valves) with progressive bilateral hydronephrosis, megacystis, and oligohydramnios threatening pulmonary development. A pigtail catheter is placed from the fetal bladder into the amniotic fluid. Patient selection is critical — vesicocentesis with fetal urine electrolytes and β2-microglobulin can help predict residual renal function (favorable: Na < 100, Cl < 90, osmolality < 200, β2-microglobulin < 6). The PLUTO trial was underpowered but suggested possible benefit.
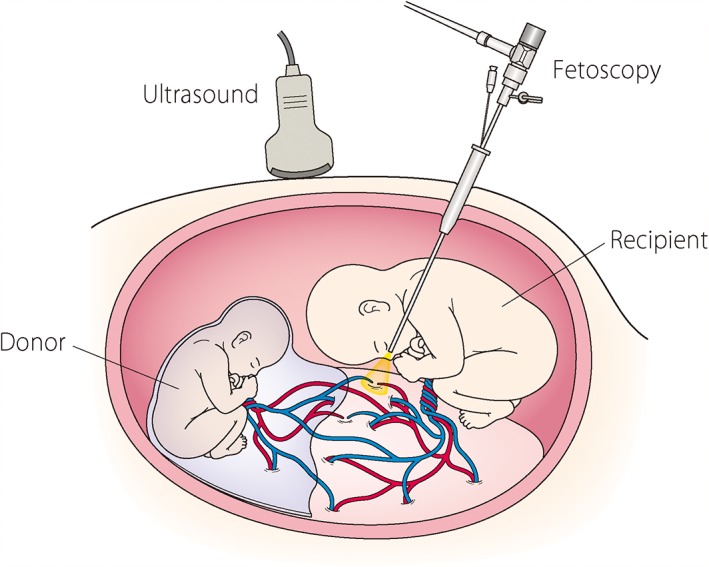
Fetoscopic Laser Photocoagulation for TTTS
The Solomon technique is the current standard: after identifying and coagulating all visible arteriovenous anastomoses on the placental surface, a continuous line of coagulation is drawn along the vascular equator connecting all the ablated vessels (creating a complete "dichorionization"). This reduces the rate of TAPS and twin anemia recurrence compared to selective coagulation alone. Performed under fetoscopic visualization with a diode or Nd:YAG laser. Overall survival: ~70-80% at least one twin, ~50-60% both twins. Major complication: PPROM (30%), preterm delivery.
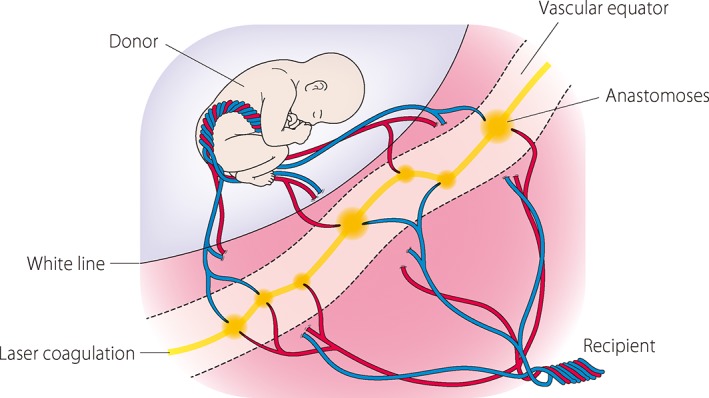
EXIT Procedure (Ex Utero Intrapartum Treatment)
The EXIT procedure maintains uteroplacental circulation during a partially delivered fetus to secure the airway before cord clamping. Indications: giant cervical teratoma, congenital high airway obstruction syndrome (CHAOS), large cervical lymphatic malformation, or any condition expected to cause complete airway obstruction at birth. The fetus is partially delivered via hysterotomy, the airway is secured (direct laryngoscopy, bronchoscopy, or tracheostomy) while placental gas exchange continues, and then the cord is clamped. Requires deep maternal general anesthesia (high-dose volatile agents for uterine relaxation).
Fetal Endoscopic Tracheal Occlusion (FETO) for CDH
For severe congenital diaphragmatic hernia (observed/expected LHR < 25% with liver herniation), FETO involves fetoscopic placement of a balloon in the fetal trachea at 27-29 weeks to occlude outflow, trapping lung fluid and promoting pulmonary growth. The balloon is removed at 34 weeks (or at the time of delivery) to allow resumption of fetal breathing movements. The TOTAL trial (NEJM 2021) demonstrated improved survival in severe left CDH (40% vs 15%) but increased the rate of preterm delivery. FETO is offered at specialized fetal therapy centers and remains a consideration for the most severe cases where expected survival with postnatal management alone is < 20%.
Fetal Surgery — Myelomeningocele Repair
The MOMS trial (Management of Myelomeningocele Study, 2011, NEJM) was the landmark RCT comparing prenatal repair (open hysterotomy at 19-25+6 weeks) vs standard postnatal repair. Results: prenatal repair reduced the need for VP shunt (40% vs 82%), improved motor function (42% vs 21% walking independently at 30 months), and reversed hindbrain herniation (Chiari II). Risks of prenatal repair: preterm delivery (79% before 37 weeks, mean 34 weeks), uterine dehiscence/thinning (35%), oligohydramnios, and maternal morbidity from hysterotomy. All subsequent pregnancies require cesarean delivery. Fetoscopic repair is under investigation and may reduce maternal morbidity.
26 Cervical Ripening & Induction in High-Risk Pregnancy
Cervical Ripening Methods
Prostaglandins: Misoprostol (PGE1 analog) — 25 μg intravaginally q3-6h (most effective ripening agent; contraindicated with prior uterine scar due to uterine rupture risk ~1-5%). Dinoprostone (PGE2) — 10 mg vaginal insert (Cervidil, retrievable) or 0.5 mg intracervical gel (Prepidil). Mechanical methods: Foley catheter (single or double balloon, inflated to 60-80 mL, placed transcervically through the internal os) — safe in prior cesarean (does not increase uterine rupture risk). Mechanical + pharmacologic combination may reduce time to delivery.
Oxytocin Induction
Oxytocin is initiated after cervical ripening or with a favorable cervix (Bishop score ≥ 6). Low-dose protocols: start 1-2 mU/min, increase by 1-2 mU/min every 30-40 minutes (maximum typically 20-40 mU/min). High-dose protocols: start 4-6 mU/min, increase by 4-6 mU/min every 15-30 minutes. Target: adequate contractions (3 in 10 minutes lasting 60-90 seconds). Tachysystole (> 5 contractions in 10 minutes averaged over 30 minutes) requires dose reduction or discontinuation, repositioning, and consideration of tocolysis (terbutaline 0.25 mg SC) if accompanied by FHR abnormalities.
Special considerations in high-risk induction: Prior cesarean with low transverse incision: cervical ripening with Foley catheter is acceptable (mechanical methods do not increase uterine rupture risk); misoprostol is contraindicated (uterine rupture risk ~1-5%); oxytocin augmentation is acceptable with close monitoring (rupture risk ~0.5-0.8% with TOLAC/VBAC). Fetal growth restriction: higher sensitivity to oxytocin; start at lowest dose. Preeclampsia with severe features: magnesium sulfate should be running concurrently; monitor fluid balance closely to avoid pulmonary edema (restrict total IV fluids to 80-100 mL/hour).
Delivery Timing by Condition
| Condition | Recommended Delivery GA |
|---|---|
| Chronic HTN (no medications) | 38+0-39+6 weeks |
| Chronic HTN (on medications) | 37+0-39+6 weeks |
| Gestational HTN | 37+0 weeks (per HYPITAT) |
| Preeclampsia without severe features | 37+0 weeks (per HYPITAT/PHOENIX) |
| Preeclampsia with severe features | 34+0 weeks (or at diagnosis if ≥ 34 wks) |
| GDM — diet-controlled (A1) | 39+0-40+6 weeks |
| GDM — medication-treated (A2) | 39+0-39+6 weeks |
| Pregestational diabetes — well controlled | 37+0-39+6 weeks |
| Pregestational diabetes — poorly controlled / vasculopathy | 34+0-37+6 weeks |
| FGR — normal Doppler, EFW ≥ 3rd percentile | 37+0-38+6 weeks |
| FGR — EFW < 3rd percentile | 34+0-37+0 weeks |
| DCDA twins — uncomplicated | 38+0 weeks |
| MCDA twins — uncomplicated | 36+0 weeks |
| MCMA twins | 32+0-34+0 weeks |
| Placenta previa | 36+0-37+6 weeks |
| Placenta accreta spectrum | 34+0-35+6 weeks |
| Prior classical cesarean | 36+0-37+0 weeks |
| Uncomplicated singleton (elective) | ≥ 39+0 weeks (ARRIVE: 39-wk induction reduces cesarean in nulliparas) |
27 Antenatal Corticosteroids & Magnesium Neuroprotection
Antenatal Corticosteroids
Indication: Women at risk of preterm delivery within 7 days between 24+0 and 33+6 weeks of gestation (some centers extend to 23 weeks). Regimen: Betamethasone 12 mg IM q24h × 2 doses (preferred — shown in the 1994 NIH Consensus and Crowley meta-analysis to reduce RDS, IVH, NEC, and neonatal death), OR dexamethasone 6 mg IM q12h × 4 doses.
Rescue course: A single rescue course of betamethasone may be administered if ≥ 14 days have elapsed since the first course, the patient remains at risk of delivery within 7 days, and GA < 34 weeks. Repeated courses beyond the rescue course are not recommended (associated with decreased birth weight and head circumference).
Late Preterm Steroids (ALPS Trial)
The ALPS trial (Antenatal Late Preterm Steroids, 2016, NEJM) demonstrated that betamethasone administered at 34+0-36+5 weeks to women at high risk of preterm delivery reduced neonatal respiratory morbidity (RDS, TTN, need for surfactant or respiratory support: 11.6% vs 14.4%, NNT ~36). However, it also increased neonatal hypoglycemia (24% vs 15%). Current practice: consider late preterm steroids for patients at high risk of delivery within 7 days at 34+0-36+5 weeks, particularly if no prior corticosteroid course. The A-PLUS trial is evaluating whether restricting late preterm steroids to those at highest risk improves the risk-benefit profile.
Magnesium Sulfate for Fetal Neuroprotection
Indication: Imminent preterm delivery at < 32 weeks of gestation. Mechanism: Reduces risk of cerebral palsy by ~30-40% (NNT ~63 to prevent one case of CP). Evidence from three major trials: MagNET, ACTOMgSO4, BEAM. Dose: 4-6 g IV loading over 20-30 minutes, followed by 1-2 g/hour maintenance until delivery (or for up to 12 hours if delivery does not occur). The same dosing is used for neuroprotection as for seizure prophylaxis, but the indications are different (neuroprotection is for fetal benefit at < 32 weeks; seizure prophylaxis is for maternal benefit in preeclampsia at any GA).
28 Classification Systems
ACOG Preeclampsia Criteria
See Section 5 for complete diagnostic criteria. Key threshold: BP ≥ 140/90 + proteinuria (≥ 300 mg/24h or P:C ≥ 0.3) or BP ≥ 140/90 + any severe feature (without proteinuria). Severe features: SBP ≥ 160/DBP ≥ 110, platelets < 100K, creatinine > 1.1, liver transaminases ≥ 2× ULN, pulmonary edema, cerebral/visual symptoms.
Mississippi Classification of HELLP Syndrome
Class 1: Platelets ≤ 50,000 + LDH ≥ 600 + AST/ALT ≥ 70. Class 2: Platelets 50,001-100,000 + LDH ≥ 600 + AST/ALT ≥ 70. Class 3: Platelets 100,001-150,000 + LDH ≥ 600 + AST/ALT ≥ 40. Class 1 carries the highest risk of maternal morbidity (DIC, renal failure, hepatic rupture).
White Classification of Diabetes in Pregnancy
See Section 6 for complete table. A1/A2 = gestational; B through T = pregestational with increasing severity of vascular disease.
Modified WHO Cardiac Risk Classification
See Section 7 for complete table. mWHO I: no increased risk. mWHO II: small increase. mWHO II-III: moderate. mWHO III: significant. mWHO IV: pregnancy contraindicated.
Quintero Staging of TTTS
See Section 16 for complete staging. Stage I: polyhydramnios/oligohydramnios only. Stage II: absent donor bladder. Stage III: abnormal Doppler. Stage IV: hydrops. Stage V: demise.
Biophysical Profile Scoring
See Section 4. Five components, each scored 0 or 2 (total 0-10): NST, fetal breathing, fetal movement, fetal tone, amniotic fluid. Score ≥ 8 = reassuring; ≤ 4 = abnormal.
Doppler Staging for Fetal Growth Restriction
See Section 13 for complete staging. Stage I: elevated UA PI. Stage II: AEDF. Stage III: REDF or elevated DV PI. Stage IV: reversed DV a-wave or spontaneous decelerations.
Bishop Score
| Parameter | 0 | 1 | 2 | 3 |
|---|---|---|---|---|
| Dilation (cm) | Closed | 1-2 | 3-4 | ≥ 5 |
| Effacement (%) | 0-30 | 40-50 | 60-70 | ≥ 80 |
| Station | -3 | -2 | -1/0 | +1/+2 |
| Consistency | Firm | Medium | Soft | — |
| Position | Posterior | Mid | Anterior | — |
Bishop score ≥ 6: favorable cervix — proceed directly to oxytocin. Bishop score < 6: unfavorable — cervical ripening required before induction. A modified Bishop score (omitting effacement) is sometimes used with similar predictive value. The Bishop score's primary utility is predicting the likelihood of successful induction and the duration of labor — it does not determine whether induction is indicated.
Swansea Criteria for Acute Fatty Liver of Pregnancy
AFLP is diagnosed when ≥ 6 of the following are present (in the absence of another explanation): vomiting, abdominal pain, polydipsia/polyuria, encephalopathy, elevated bilirubin (> 0.8 mg/dL), hypoglycemia (< 72 mg/dL), elevated uric acid (> 5.7 mg/dL), leukocytosis (> 11,000), ascites or bright liver on US, elevated transaminases (AST/ALT > 42), elevated ammonia (> 47 μmol/L), renal impairment (creatinine > 1.7 mg/dL), coagulopathy (PT > 14 s or aPTT > 34 s), and microvesicular steatosis on liver biopsy. Key distinguishing features from HELLP: hypoglycemia (most specific), coagulopathy (low fibrinogen, prolonged PT — more prominent than in HELLP), and encephalopathy. Treatment is delivery. Screen neonate for LCHAD deficiency (long-chain 3-hydroxyacyl-CoA dehydrogenase — recurrence risk 25% if present, as AFLP is associated with fetal LCHAD deficiency through accumulation of toxic fatty acid intermediates).
29 Medications Master Table
Antihypertensives
| Drug | Class | Dose | Route | Notes |
|---|---|---|---|---|
| Labetalol | α/β-blocker | Acute: 20-80 mg IV; Chronic: 200-2400 mg/day PO | IV/PO | First-line for acute severe HTN. Avoid in asthma, heart block. |
| Hydralazine | Vasodilator | 5-10 mg IV q20 min | IV | Second-line acute. Reflex tachycardia. |
| Nifedipine | CCB (dihydropyridine) | Acute: 10-20 mg PO; Chronic: 30-120 mg/day XL | PO | Oral only. First-line chronic HTN in pregnancy. |
| Methyldopa | Central α-agonist | 250-1000 mg PO BID-TID | PO | Long safety track record. Sedation, depression. |
| ACE inhibitors / ARBs | RAAS inhibitors | — | — | CONTRAINDICATED in pregnancy. Fetal renal failure, oligohydramnios, hypocalvaria. |
Magnesium Sulfate
| Indication | Loading Dose | Maintenance | Therapeutic Level | Toxicity/Antidote |
|---|---|---|---|---|
| Seizure prophylaxis (preeclampsia) | 4-6 g IV over 15-20 min | 1-2 g/h IV | 4-7 mEq/L | Loss of reflexes 7-10; resp arrest 10-12; cardiac arrest 15-20. Antidote: Ca gluconate 1 g IV. |
| Fetal neuroprotection | 4-6 g IV over 20-30 min | 1-2 g/h IV (up to 12h) | 4-7 mEq/L | Same toxicity profile. |
Tocolytics
| Drug | Class | Dose | Duration | Key Side Effects |
|---|---|---|---|---|
| Nifedipine | CCB | 20 mg PO loading, then 10-20 mg q4-6h | 48 hours (to allow steroid effect) | Hypotension, headache, flushing |
| Indomethacin | NSAID (COX inhibitor) | 50-100 mg loading, then 25-50 mg q6h | ≤ 48 hours; avoid after 32 weeks | Premature closure of ductus arteriosus, oligohydramnios, neonatal renal dysfunction |
| Terbutaline | β2-agonist | 0.25 mg SC q20 min × 3 | Short-term (< 48-72 hours) | Tachycardia, pulmonary edema, hyperglycemia. FDA black box: not for prolonged tocolysis. |
| Atosiban | Oxytocin antagonist | 6.75 mg IV bolus, then infusion | 48 hours | Not FDA-approved in US. Fewer side effects than β-agonists. |
Corticosteroids
| Drug | Dose | Regimen | Indication |
|---|---|---|---|
| Betamethasone | 12 mg IM | 2 doses, 24 hours apart | Fetal lung maturity (24-33+6 weeks); ALPS (34-36+5 weeks) |
| Dexamethasone | 6 mg IM | 4 doses, 12 hours apart | Alternative to betamethasone |
Anticoagulants
| Drug | Prophylactic Dose | Therapeutic Dose | Notes |
|---|---|---|---|
| Enoxaparin (LMWH) | 40 mg SC daily | 1 mg/kg SC q12h | Drug of choice in pregnancy. Does not cross placenta. Monitor anti-Xa levels. |
| UFH | 5,000 U SC q12h | Adjusted to aPTT 1.5-2.5× | Alternative to LMWH. Higher HIT risk. Short half-life (advantage near delivery). |
| Warfarin | — | INR-adjusted | CONTRAINDICATED (except mechanical valves — shared decision). Embryopathy weeks 6-12. |
| DOACs | — | — | CONTRAINDICATED in pregnancy. Insufficient safety data. Cross placenta. |
RhoGAM (Anti-D Immunoglobulin)
| Indication | Dose | Timing |
|---|---|---|
| Routine prophylaxis | 300 μg IM | 28 weeks |
| Postpartum (Rh+ infant) | 300 μg IM | Within 72 hours of delivery |
| First trimester event (< 12 weeks) | 50-120 μg IM (MICRhoGAM) | At event |
| Sensitizing event (≥ 12 weeks) | 300 μg IM | At event |
| Large fetomaternal hemorrhage | Additional 300 μg per 30 mL fetal blood | Per Kleihauer-Betke result |
30 Imaging & Diagnostics Reference
Ultrasound Parameters by Gestational Age
| GA (weeks) | BPD (mm) | HC (mm) | AC (mm) | FL (mm) | EFW (g) |
|---|---|---|---|---|---|
| 20 | 47 | 175 | 149 | 33 | 300 |
| 24 | 59 | 219 | 195 | 44 | 600 |
| 28 | 72 | 262 | 240 | 54 | 1,100 |
| 32 | 82 | 296 | 281 | 63 | 1,800 |
| 36 | 89 | 322 | 316 | 70 | 2,600 |
| 40 | 94 | 341 | 350 | 76 | 3,400 |
Doppler Indices — Normal Values
| Vessel | Parameter | Normal | Abnormal |
|---|---|---|---|
| Umbilical artery | S/D ratio (28 wks) | < 3.0 | > 3.0 (elevated); AEDF; REDF |
| MCA | PSV | Varies by GA | > 1.5 MoM = moderate-severe anemia |
| MCA | PI (pulsatility index) | High resistance | Low PI = brain sparing (cerebral vasodilation) |
| CPR (MCA PI/UA PI) | Ratio | > 1.0 | < 1.0 = redistribution |
| Ductus venosus | a-wave | Positive (forward flow) | Absent or reversed = cardiac decompensation |
| Uterine artery | Notching | No notching after 24 weeks | Bilateral notching = increased preeclampsia/FGR risk |
MRI in Pregnancy
Fetal MRI (without gadolinium) is safe at any gestational age (no evidence of teratogenicity at 1.5 or 3.0 Tesla) and is most useful for:
CNS anomalies: Superior to US for cortical malformations (lissencephaly, polymicrogyria, heterotopia), posterior fossa abnormalities (Dandy-Walker spectrum vs mega cisterna magna vs Blake pouch cyst), corpus callosum agenesis (US sensitivity ~50% vs MRI ~90%), ventriculomegaly etiology, and hemorrhage.
Thoracic assessment: Lung volume measurement (MRI-based fetal lung volume) for CDH prognostication and severe oligohydramnios — more accurate than US-based methods.
Placenta accreta spectrum: Complementary to US, particularly for posterior placentas and to assess parametrial/bladder invasion depth.
Complex fetal masses: Characterization of teratomas, lymphatic malformations, and other masses before fetal surgery or EXIT planning.
Optimal timing: After 20 weeks (myelination patterns visible; smaller fetus limits resolution). T2-weighted single-shot fast spin echo (SSFSE/HASTE) is the primary sequence. Diffusion-weighted imaging (DWI) can identify areas of ischemia or restricted diffusion in brain anomalies.
Gadolinium is contraindicated in pregnancy — it crosses the placenta, is excreted into amniotic fluid by the fetal kidneys, and has been associated with increased risk of rheumatologic/inflammatory conditions and stillbirth in one large retrospective cohort study (Ray et al., JAMA 2016). Non-contrast MRI is the standard for fetal imaging.
Genetic Testing Comparison
| Test | Timing | Resolution | Turnaround | Key Advantage | Key Limitation |
|---|---|---|---|---|---|
| cfDNA/NIPT | ≥ 10 weeks | Whole chromosome | 5-10 days | Highest sensitivity for trisomy 21 | Screening only; PPV varies |
| FISH | Any (on CVS/amnio sample) | Targeted probes | 24-48 hours | Rapid preliminary result | Limited targets (13, 18, 21, X, Y) |
| Karyotype | Any (CVS or amnio) | 5-10 Mb | 10-14 days | Gold standard; detects balanced rearrangements | Low resolution; requires culture |
| Microarray (CMA) | Any (CVS or amnio) | 50-100 kb | 10-14 days | Detects CNVs missed by karyotype | VUS (1.5%); misses balanced changes |
| Exome sequencing | Any (CVS or amnio) | Single nucleotide | 4-6 weeks | 10-30% additional yield | VUS; cost; incidental findings |
31 Abbreviations Master List
AC — Abdominal circumference
AEDF — Absent end-diastolic flow
AFE — Amniotic fluid embolism
AFI — Amniotic fluid index
AFLP — Acute fatty liver of pregnancy
aHUS — Atypical hemolytic uremic syndrome
APS — Antiphospholipid syndrome
BPD — Biparietal diameter
BPP — Biophysical profile
cART — Combined antiretroviral therapy
cfDNA — Cell-free DNA
CHB — Congenital heart block
CHD — Congenital heart defect
CKD — Chronic kidney disease
CMA — Chromosomal microarray analysis
CMV — Cytomegalovirus
CNV — Copy number variant
CPM — Confined placental mosaicism
CPR — Cerebroplacental ratio
CRL — Crown-rump length
CST — Contraction stress test
CVS — Chorionic villus sampling
DCDA — Dichorionic-diamniotic
DIC — Disseminated intravascular coagulation
DV — Ductus venosus
EFW — Estimated fetal weight
EXIT — Ex utero intrapartum treatment
FGR — Fetal growth restriction
FHR — Fetal heart rate
FL — Femur length
GA — Gestational age
GBS — Group B Streptococcus
GCT — Glucose challenge test
GDM — Gestational diabetes mellitus
HC — Head circumference
HDFN — Hemolytic disease of the fetus and newborn
HELLP — Hemolysis, Elevated Liver enzymes, Low Platelets
HLHS — Hypoplastic left heart syndrome
HSV — Herpes simplex virus
HTN — Hypertension
IAP — Intrapartum antibiotic prophylaxis
IUFD — Intrauterine fetal demise
IUGR — Intrauterine growth restriction (older term; FGR preferred)
IUT — Intrauterine transfusion
LMWH — Low-molecular-weight heparin
LUTO — Lower urinary tract obstruction
MCA — Middle cerebral artery
MCA-PSV — Middle cerebral artery peak systolic velocity
MCDA — Monochorionic-diamniotic
MCMA — Monochorionic-monoamniotic
MFM — Maternal-fetal medicine
MoM — Multiples of the median
MSAFP — Maternal serum alpha-fetoprotein
mWHO — Modified World Health Organization (cardiac risk)
MVP — Maximum vertical pocket
NIPT — Non-invasive prenatal testing
NST — Non-stress test
NT — Nuchal translucency
NTD — Neural tube defect
OGTT — Oral glucose tolerance test
PAPP-A — Pregnancy-associated plasma protein A
PAS — Placenta accreta spectrum
PPCM — Peripartum cardiomyopathy
PPH — Postpartum hemorrhage
PPROM — Preterm premature rupture of membranes
PTB — Preterm birth
PUBS — Percutaneous umbilical blood sampling (cordocentesis)
REDF — Reversed end-diastolic flow
SGA — Small for gestational age
SLE — Systemic lupus erythematosus
TAPS — Twin anemia-polycythemia sequence
TORCH — Toxoplasmosis, Other, Rubella, CMV, Herpes
TRAP — Twin reversed arterial perfusion
TTTS — Twin-to-twin transfusion syndrome
TTP — Thrombotic thrombocytopenic purpura
TVUS — Transvaginal ultrasound
UA — Umbilical artery
UFH — Unfractionated heparin
VSD — Ventricular septal defect
VTE — Venous thromboembolism
VUS — Variant of uncertain significance
32 Landmark Trials
MAGPIE Trial (Lancet 2002) — Magnesium sulphate for preeclampsia. 10,141 women with preeclampsia. MgSO4 reduced eclampsia by 58% (RR 0.42, 95% CI 0.29-0.60) with no significant effect on maternal mortality. Established MgSO4 as the standard of care for seizure prophylaxis in preeclampsia. PubMed 12049563
HYPITAT Trial (Lancet 2009) — Induction of labour versus expectant monitoring for gestational hypertension or mild preeclampsia after 36 weeks. Induction at 37 weeks reduced severe maternal hypertensive complications (RR 0.71) without increasing cesarean rate. Supported earlier delivery in gestational HTN/mild preeclampsia. PubMed 19394535
PHOENIX Trial (Lancet 2019) — Planned delivery vs expectant management for late preterm preeclampsia. 901 women at 34-37 weeks with preeclampsia. Planned delivery reduced adverse maternal outcomes (composite endpoint: 289 fewer per 1,000, absolute risk 32% vs 48%) with acceptable neonatal outcomes. Supports delivery at 37 weeks for preeclampsia. PubMed 31178098
MOMS Trial (NEJM 2011) — Prenatal vs postnatal repair of myelomeningocele. 183 patients randomized. Prenatal repair reduced VP shunt (40% vs 82%) and improved motor outcomes (42% vs 21% walking independently at 30 months). Increased preterm delivery and maternal morbidity. Landmark trial establishing fetal surgery for spina bifida. PubMed 21306277
ALPS Trial (NEJM 2016) — Antenatal late preterm steroids. 2,831 women at 34+0-36+5 weeks at risk of preterm delivery. Betamethasone reduced respiratory morbidity (RR 0.80) but increased neonatal hypoglycemia (24% vs 15%). Supports selective use of late preterm steroids. PubMed 26842679
ARRIVE Trial (NEJM 2018) — Elective induction of labor at 39 weeks vs expectant management in low-risk nulliparous women. 6,106 women. Induction at 39 weeks reduced cesarean delivery (18.6% vs 22.2%, RR 0.84) and hypertensive disorders (9.1% vs 14.1%) without increasing adverse perinatal outcomes. Challenged the assumption that elective induction increases cesarean rate. PubMed 30089070
BEAM Trial (NEJM 2008) — MgSO4 for neuroprotection before anticipated preterm delivery at 24-31 weeks. 2,241 women. MgSO4 reduced moderate-severe cerebral palsy (1.9% vs 3.5%, RR 0.55). No significant reduction in perinatal death. Established MgSO4 for fetal neuroprotection at < 32 weeks. PubMed 18184952
EUROFETUS Trial (NEJM 2004) — Endoscopic laser surgery vs serial amnioreduction for TTTS. 142 pregnancies with severe TTTS (Quintero stage I-IV). Laser was superior: at least one survivor at 6 months (76% vs 56%, p = 0.009) and fewer neurologic complications. Established fetoscopic laser as the standard treatment for TTTS ≥ stage II. PubMed 15254534
PROLONG Trial (AJOG 2020) — 17-OHPC for recurrent preterm birth prevention. 1,708 women with prior spontaneous PTB. 17-OHPC did NOT significantly reduce preterm birth < 35 weeks (11.0% vs 11.5%, p = 0.87). Led to FDA advisory committee recommendation to withdraw Makena. A pivotal reassessment of a previously established therapy. PubMed 31794434
TRUFFLE Trial (Lancet 2015) — Timing of delivery in early-onset FGR. 503 pregnancies at 26-32 weeks with FGR and abnormal UA Doppler. Compared three delivery triggers: reduced FHR short-term variability (cCTG), early DV changes, and late DV changes. Late DV changes (absent/reversed a-wave) provided the best balance of survival without neuroimpairment. Informs Doppler-based delivery timing in early-onset FGR. PubMed 25601357
ASPRE Trial (NEJM 2017) — Aspirin for evidence-based preeclampsia prevention. 1,776 women at high risk (first-trimester screening algorithm). Aspirin 150 mg nightly from 11-14 weeks to 36 weeks reduced preterm preeclampsia (< 37 weeks) by 62% (RR 0.38). Supports first-trimester screening-based aspirin prophylaxis. PubMed 28657417
ORACLE I Trial (Lancet 2001) — Antibiotics for preterm prelabour rupture of membranes. 4,826 women. Erythromycin reduced composite neonatal morbidity and prolonged pregnancy. Amoxicillin-clavulanate increased necrotizing enterocolitis. Established the standard latency antibiotic regimen for PPROM. PubMed 11297958
WOMAN Trial (Lancet 2017) — Tranexamic acid for postpartum hemorrhage. 20,060 women with PPH. TXA 1 g IV reduced death due to bleeding (1.5% vs 1.9%, RR 0.81) and the need for laparotomy. Greatest benefit when given within 3 hours (ideally within 1 hour). Established TXA as adjunctive therapy for PPH. PubMed 28456509
TOTAL Trial (NEJM 2021) — Fetoscopic endoluminal tracheal occlusion for severe congenital diaphragmatic hernia. FETO at 27-29 weeks improved survival to discharge in severe left CDH (40% vs 15%, p < 0.001). Increased preterm delivery. Established FETO as a viable fetal therapy for severe CDH. PubMed 34106558
FIGO Safe Motherhood / Crowley Meta-analysis (1994 NIH Consensus) — The landmark meta-analysis confirming that a single course of antenatal corticosteroids reduces RDS by 44%, IVH by 46%, and neonatal death by 31% when delivered at 24-34 weeks. One of the most impactful interventions in perinatal medicine. Led to universal adoption of antenatal steroids for threatened preterm delivery.
Solomon Trial (Lancet 2014) — Solomon technique vs standard selective laser for TTTS. 274 MCDA twin pregnancies with TTTS. Solomon technique (drawing a line connecting ablated anastomoses along the vascular equator) reduced recurrence of TTTS (1% vs 7%) and TAPS (3% vs 16%) compared to selective coagulation alone. No difference in overall survival. Established Solomon as the preferred laser technique for TTTS. PubMed 24613566
HAPO Study (NEJM 2008) — Hyperglycemia and Adverse Pregnancy Outcomes. 23,316 pregnancies. Demonstrated a continuous, graded relationship between maternal glucose levels (below previous diagnostic thresholds for GDM) and adverse outcomes (macrosomia, primary cesarean, neonatal hypoglycemia, cord C-peptide). Led to the IADPSG one-step diagnostic criteria for GDM. PubMed 18463375