OB/GYN
Every diagnosis, procedure, surgical technique, classification, complication, medication, and management algorithm across the full scope of obstetrics and gynecology in one place.
01 Pelvic Anatomy
Uterus, Cervix & Vagina
The uterus is a thick-walled, pear-shaped muscular organ measuring approximately 7-8 cm in length, 4-5 cm in width, and 2-3 cm in anteroposterior diameter in the nulliparous adult. It is divided into the fundus (superior dome above the tubal ostia), corpus (body), isthmus (lower uterine segment — the site of the low transverse cesarean incision), and cervix. The uterine wall consists of three layers: the endometrium (functionalis layer shed during menses + basalis layer that regenerates), the myometrium (thick smooth muscle arranged in interlacing bundles — critical for hemostasis after placental delivery via the "living ligature" of myometrial contraction), and the serosa (visceral peritoneum).
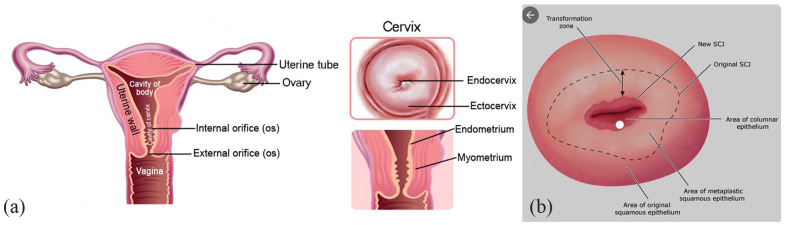
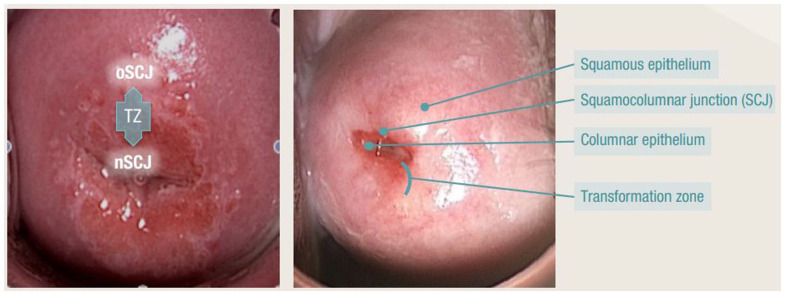
The cervix projects into the upper vagina and is divided into the supravaginal and vaginal portions. The endocervical canal is lined by columnar epithelium; the ectocervix is covered by squamous epithelium. The transformation zone (squamocolumnar junction) is the area where metaplastic squamous epithelium replaces columnar epithelium — this is the site where cervical dysplasia and cancer originate, and where Pap smears and colposcopic biopsies must be directed.
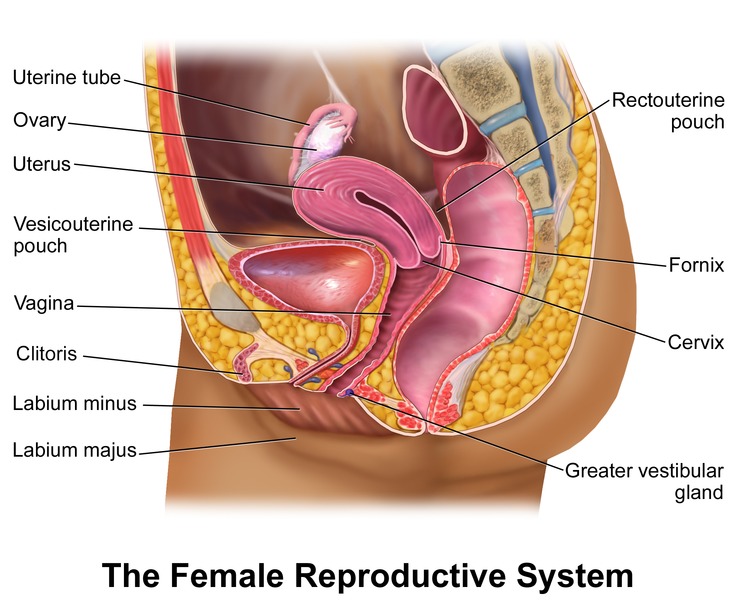
The vagina is a fibromuscular canal approximately 7-9 cm in length. The anterior wall is shorter than the posterior wall. The posterior fornix is the deepest part of the vaginal canal and lies directly anterior to the rectouterine pouch (pouch of Douglas) — the most dependent part of the peritoneal cavity in the upright position, where fluid collects in ectopic pregnancy rupture and can be accessed by culdocentesis.
Ovaries & Fallopian Tubes
The ovaries are paired, almond-shaped organs (~3 × 2 × 1 cm) located in the ovarian fossa on the lateral pelvic wall. Each ovary is attached to the broad ligament by the mesovarium, to the uterus by the utero-ovarian ligament (ovarian ligament), and to the pelvic sidewall by the infundibulopelvic (IP) ligament (suspensory ligament of the ovary) — which contains the ovarian artery and vein. Ligation of the IP ligament during oophorectomy or salpingo-oophorectomy must be done carefully because the ureter crosses just beneath it as it enters the pelvis.
The fallopian tubes (~10-12 cm long) have four segments: interstitial (intramural) — within the myometrium; isthmus — narrow, thick-walled segment (site of tubal ligation); ampulla — widest segment, where fertilization typically occurs, and the most common site of ectopic pregnancy (~70%); and infundibulum — the funnel-shaped terminal portion with finger-like fimbriae that sweep the ovulated oocyte into the tube.
Pelvic Ligaments & Supporting Structures
Broad ligament: A double layer of peritoneum extending from the lateral uterus to the pelvic sidewall. Contains the round ligament (anteriorly), fallopian tube (superiorly), utero-ovarian ligament, uterine artery, and ureter. The broad ligament is divided into the mesosalpinx (supports the tube), mesovarium (supports the ovary), and mesometrium (supports the uterus).
Cardinal ligaments (transverse cervical, Mackenrodt ligaments): The primary support of the uterus and upper vagina. Extend from the lateral cervix to the pelvic sidewall at the level of the ischial spines. Contain the uterine artery and veins. Clamped, cut, and ligated during hysterectomy — this is where the uterine artery is secured.
Uterosacral ligaments: Extend from the posterolateral cervix to the anterior sacrum (S2-S4). Important for uterine support and for suspension during prolapse surgery (McCall culdoplasty, uterosacral ligament suspension). The ureters course close to the uterosacral ligaments — another site of potential ureteral injury.
Round ligaments: Extend from the uterine cornua through the inguinal canal to the labia majora. Maintain uterine anteversion but provide minimal support. Analogous to the gubernaculum testis in males.
Blood Supply
The uterine artery (branch of the anterior division of the internal iliac artery) is the primary blood supply to the uterus. It courses medially along the base of the broad ligament, crosses over the ureter at the level of the internal cervical os ("water under the bridge"), and ascends along the lateral uterus in a tortuous course, anastomosing with the ovarian artery at the cornua. The ovarian artery arises directly from the aorta (below the renal arteries) and courses through the IP ligament to reach the ovary. The left ovarian vein drains into the left renal vein; the right ovarian vein drains directly into the IVC.
The vaginal artery (also from the internal iliac) supplies the vagina and lower cervix. The internal pudendal artery (terminal branch of the anterior division of the internal iliac) supplies the vulva, perineum, and clitoris via the pudendal canal (Alcock canal). During postpartum hemorrhage, uterine artery embolization (UAE) targets the uterine arteries via the internal iliac system.
Venous drainage: The uterine veins parallel the uterine arteries and drain into the internal iliac veins. The ovarian venous drainage is asymmetric: the right ovarian vein drains directly into the IVC, while the left ovarian vein drains into the left renal vein (analogous to the left testicular vein — this asymmetry explains why left-sided varicoceles are more common). The pampiniform plexus of veins around the ovary can become dilated (pelvic congestion syndrome — a cause of chronic pelvic pain).
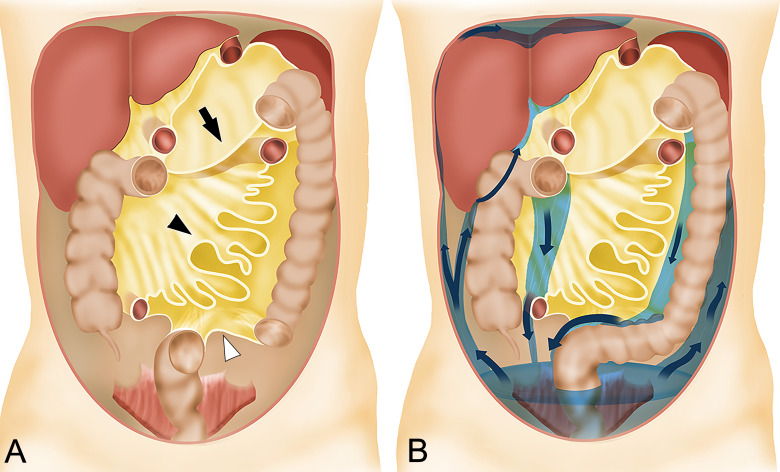
Lymphatic drainage: The fundus and upper body of the uterus drain primarily to the para-aortic lymph nodes (via the IP ligament, following the ovarian vessels). The lower uterine segment, cervix, and upper vagina drain to the obturator, internal iliac, and external iliac lymph nodes (pelvic nodes). The lower vagina and vulva drain to the inguinofemoral lymph nodes. Understanding lymphatic drainage is critical for surgical staging of gynecologic cancers and sentinel lymph node mapping.
Pelvic Floor Musculature
The pelvic diaphragm is the muscular floor of the pelvis, composed primarily of the levator ani muscle complex: pubococcygeus (including pubovaginalis and puboanalis), puborectalis (maintains the anorectal angle — critical for fecal continence), and iliococcygeus. The coccygeus (ischiococcygeus) muscle completes the pelvic floor posteriorly. These muscles are innervated by branches of the sacral plexus (S3-S4). Damage to the levator ani during vaginal delivery is a major risk factor for pelvic organ prolapse and urinary incontinence.
The urogenital diaphragm (perineal membrane) is a triangular fascial layer spanning the anterior pelvic outlet, containing the deep transverse perineal muscle and the external urethral sphincter. The perineal body is the central tendon of the perineum between the vaginal introitus and the anus — a critical support structure that is lacerated during obstetric perineal tears.
Ureter Course in the Pelvis
The ureter is at risk of injury at three locations during gynecologic surgery: (1) at the pelvic brim, where it crosses under the IP ligament and ovarian vessels; (2) at the base of the broad ligament, where the uterine artery crosses over it ("water under the bridge") approximately 1.5-2 cm lateral to the cervix; and (3) at the cardinal ligament/uterosacral ligament level near the vaginal fornix. Ureteral injury occurs in ~0.5-1% of hysterectomies and up to 2-3% when performed for advanced endometriosis or gynecologic malignancy.
02 Reproductive Physiology
Hypothalamic-Pituitary-Ovarian (HPO) Axis
The hypothalamus secretes gonadotropin-releasing hormone (GnRH) in a pulsatile fashion from the arcuate nucleus. The pulsatile pattern is critical: high-frequency GnRH pulses favor LH secretion, while low-frequency pulses favor FSH secretion. Continuous (non-pulsatile) GnRH administration paradoxically suppresses gonadotropin release — this is the principle behind GnRH agonist therapy (leuprolide) for endometriosis, fibroids, and precocious puberty (initial flare, then downregulation). GnRH antagonists (cetrorelix, ganirelix, elagolix) achieve immediate suppression without the initial flare.
The anterior pituitary secretes FSH (stimulates follicular growth, granulosa cell estrogen production) and LH (stimulates theca cell androgen production, triggers ovulation, supports the corpus luteum). The two-cell, two-gonadotropin model describes ovarian steroidogenesis: theca cells (LH-driven) produce androgens (androstenedione); granulosa cells (FSH-driven) convert androgens to estrogens via aromatase.
The Menstrual Cycle
The normal menstrual cycle averages 28 days (range 21-35) and is divided into ovarian and endometrial phases:
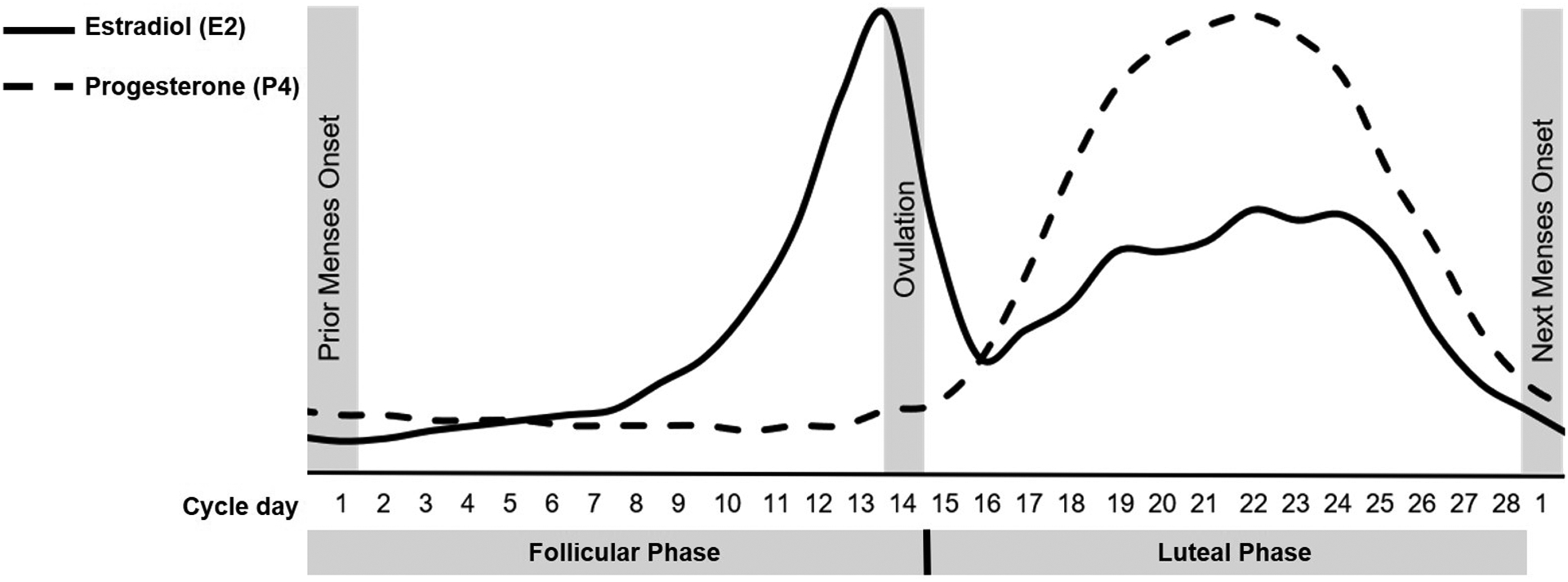
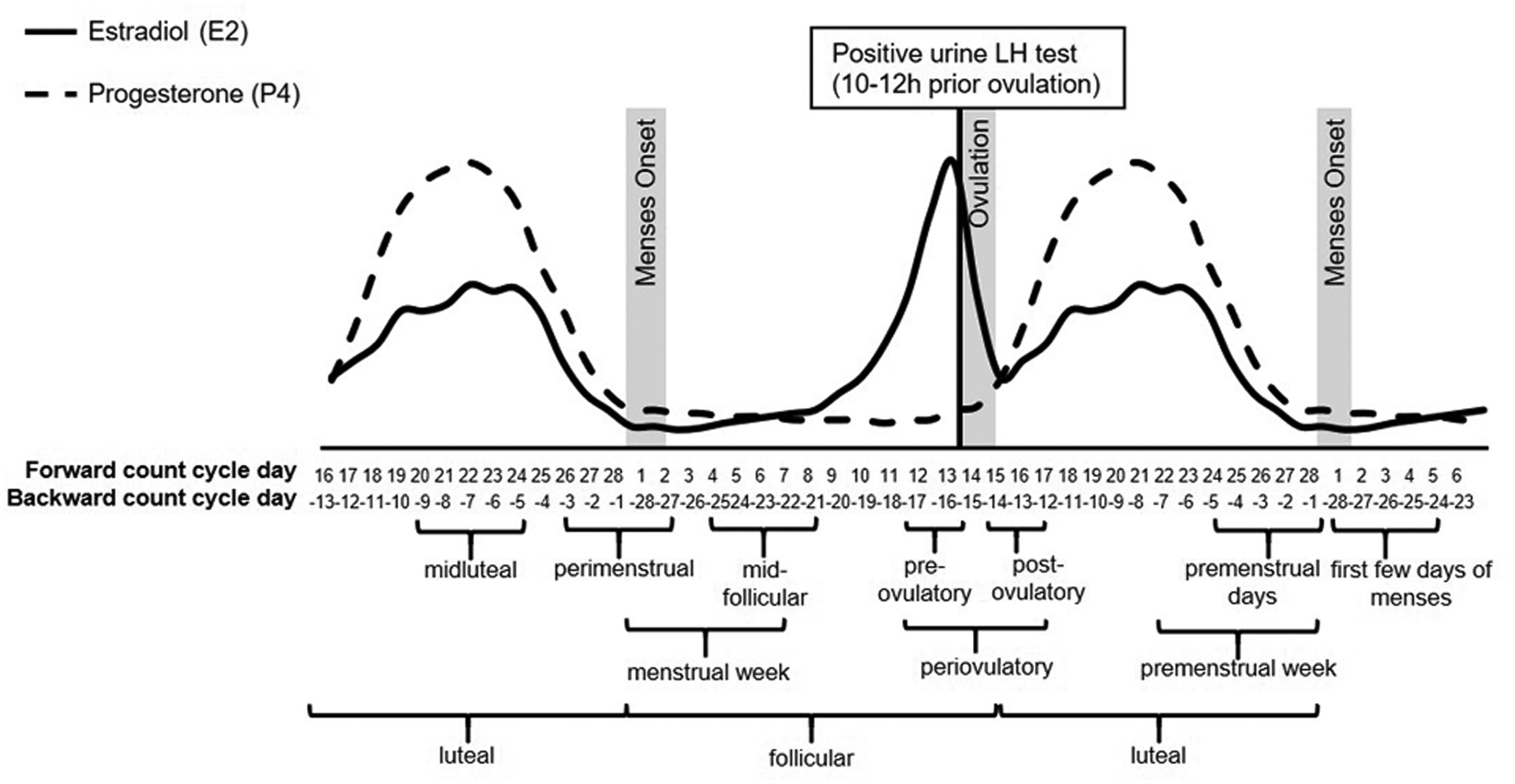
Follicular phase (Days 1-14, variable): FSH stimulates growth of a cohort of antral follicles. One dominant follicle is selected by day 5-7, producing rising estradiol levels. Estradiol exerts negative feedback on FSH (causing atresia of non-dominant follicles) but at high sustained levels triggers positive feedback on LH, leading to the LH surge.
Ovulation (Day 14): The LH surge triggers completion of meiosis I, luteinization of granulosa cells, prostaglandin/protease release causing follicular rupture, and oocyte release approximately 34-36 hours after LH surge onset. Ovulation prediction kits detect the urinary LH surge.
Luteal phase (Days 15-28, fixed ~14 days): The ruptured follicle becomes the corpus luteum, producing progesterone and estradiol. Progesterone is the dominant hormone of the luteal phase. If pregnancy does not occur, the corpus luteum degenerates (luteolysis) by ~day 24-26, progesterone and estradiol fall, and menstruation occurs. If implantation occurs, hCG from the trophoblast rescues the corpus luteum ("corpus luteum of pregnancy") until the placenta assumes progesterone production (~8-10 weeks).
Menstrual phase (Days 1-5): Shedding of the functionalis layer due to progesterone withdrawal, spiral arteriole vasoconstriction, and tissue ischemia.
Proliferative phase (Days 5-14): Estrogen-driven regeneration and thickening of the endometrium. Straight, tubular glands. Corresponds to the follicular phase.
Secretory phase (Days 15-28): Progesterone-driven transformation. Glands become tortuous and secrete glycogen. Spiral arterioles develop. Characteristic histologic features allow precise dating of the endometrium (subnuclear vacuoles by day 16-17; peak secretion days 19-20; stromal predecidual reaction days 23-24). Corresponds to the luteal phase.
Fertilization & Implantation
Fertilization & Implantation
Fertilization occurs in the ampulla of the fallopian tube within 12-24 hours of ovulation. Sperm undergo capacitation in the female reproductive tract and the acrosome reaction to penetrate the zona pellucida. The cortical reaction after sperm entry prevents polyspermy. The zygote undergoes cleavage divisions during transport through the tube, reaching the uterine cavity as a morula by day 3-4 and a blastocyst by day 5-6. Implantation occurs 6-7 days after fertilization (day 20-21 of the cycle), when the blastocyst hatches from the zona pellucida and the trophoblast invades the endometrium during the window of implantation (days 20-24, coinciding with peak progesterone-induced endometrial receptivity).
03 The OB/GYN Examination
Speculum Examination
The speculum exam is fundamental for visualizing the vagina and cervix. A Graves speculum (most common, wider blades) or Pedersen speculum (narrower blades, for nulliparous or atrophic patients) is used. The cervix is inspected for lesions, discharge, cervical motion, and os dilation. The Pap smear is obtained from the transformation zone using a spatula and endocervical brush (or a broom device). Cervical cultures (gonorrhea, chlamydia — NAAT) are collected during the speculum exam.
Bimanual Examination
The bimanual exam assesses the uterus (size, shape, position — anteverted/retroverted, consistency, tenderness, masses) and adnexa (ovarian enlargement, masses, tenderness). Cervical motion tenderness (chandelier sign) suggests peritoneal irritation and is classically associated with PID or ectopic pregnancy. Uterine size is described in gestational week equivalents (e.g., "12-week size" for a fibroid uterus approximately at the level of the pubic symphysis).
Obstetric Examination — Leopold Maneuvers
Four maneuvers to determine fetal lie, presentation, position, and engagement:
First maneuver (fundal grip): Palpate the fundus to determine which fetal pole (head vs breech) occupies the fundus. The head is hard, round, and ballottable; the breech is soft, irregular, and less mobile.
Second maneuver (umbilical grip): Palpate the lateral aspects of the uterus to locate the fetal back (smooth, continuous surface) versus the small parts (limbs — irregular, mobile).
Third maneuver (Pawlik grip): Grasp the presenting part above the symphysis pubis to determine presentation (cephalic vs breech) and whether it is engaged in the pelvis.
Fourth maneuver (pelvic grip): Face the patient's feet and palpate deeply toward the pelvic inlet to assess the degree of engagement and the attitude of the presenting part (flexion vs extension).
Bishop Score
The Bishop score predicts the likelihood of successful induction of labor. A score ≥ 8 is considered favorable (similar success rate to spontaneous labor). A score ≤ 5 suggests the cervix is unfavorable and cervical ripening agents (prostaglandins, mechanical dilation) should be used before oxytocin.
| Parameter | 0 | 1 | 2 | 3 |
|---|---|---|---|---|
| Dilation (cm) | Closed | 1-2 | 3-4 | ≥ 5 |
| Effacement (%) | 0-30 | 40-50 | 60-70 | ≥ 80 |
| Station | -3 | -2 | -1, 0 | +1, +2 |
| Consistency | Firm | Medium | Soft | — |
| Position | Posterior | Mid | Anterior | — |
Fetal Heart Rate Monitoring — Basics
Continuous electronic fetal monitoring (EFM) uses an external Doppler ultrasound transducer or an internal fetal scalp electrode (FSE) to assess fetal heart rate (FHR). A tocodynamometer (external) or intrauterine pressure catheter (IUPC) monitors uterine contractions. The baseline FHR, variability, presence of accelerations, and type of decelerations are systematically evaluated (detailed in Section 12).
04 Normal Pregnancy & Prenatal Care
Prenatal Visit Schedule
Standard visit frequency: every 4 weeks until 28 weeks, every 2 weeks from 28-36 weeks, and weekly from 36 weeks until delivery. The first visit (ideally 6-10 weeks) includes comprehensive history, physical exam, dating ultrasound (if uncertain LMP), and initial labs.
Routine Laboratory Testing
First trimester: CBC, blood type & Rh, antibody screen, rubella immunity, hepatitis B surface antigen (HBsAg), HIV, syphilis (RPR/VDRL), urine culture, gonorrhea/chlamydia NAAT, Pap smear (if due), varicella immunity (if unknown). TSH if clinically indicated.
15-20 weeks: Maternal serum alpha-fetoprotein (MSAFP) or quad screen (AFP, hCG, estriol, inhibin A) if not done with first-trimester screening.
24-28 weeks: One-hour glucose challenge test (50 g GCT), repeat CBC, repeat antibody screen if Rh-negative, RhoGAM administration at 28 weeks if Rh-negative.
35-37 weeks: Group B Streptococcus (GBS) vaginal-rectal culture. Repeat STI screening in high-risk patients.
Ultrasound Timing
First trimester (11-14 weeks): Dating, viability, number of fetuses, nuchal translucency measurement (NT — part of first-trimester combined screening). Second trimester (18-22 weeks): Anatomy scan — detailed fetal structural survey, placental location, cervical length, amniotic fluid volume. Third trimester: Growth ultrasound as clinically indicated (IUGR, macrosomia, multiple gestations).
Genetic Screening & Diagnostic Testing
First-trimester combined screening (11-14 weeks): NT measurement + serum PAPP-A + free β-hCG. Detection rate for trisomy 21 ~82-87%. Quad screen (15-20 weeks): AFP, hCG, unconjugated estriol (uE3), inhibin A. Detection rate for trisomy 21 ~81%.
Trisomy 21 (Down syndrome): ↓ AFP, ↑ hCG, ↓ estriol, ↑ inhibin A.
Trisomy 18 (Edwards syndrome): ↓ AFP, ↓ hCG, ↓ estriol, normal inhibin A.
Open neural tube defect: ↑↑ AFP (also elevated in abdominal wall defects, multiple gestation, placental abnormalities).
Cell-free DNA / NIPT (non-invasive prenatal testing): Screens fetal cell-free DNA fragments in maternal blood from ~10 weeks. Detection rate > 99% for trisomy 21, ~97% for trisomy 18, ~92% for trisomy 13. Also screens for sex chromosome aneuploidies. ACOG recommends offering NIPT to all pregnant patients regardless of age or risk. It is a screening test, not diagnostic — positive results require confirmatory amniocentesis (15-20 weeks, ~0.1-0.3% miscarriage risk) or chorionic villus sampling (CVS) (10-13 weeks, ~0.5-1% miscarriage risk).
Physiologic Changes of Pregnancy
Cardiovascular: Blood volume increases 40-50% (peaks at 32-34 weeks), CO increases 30-50%, SVR decreases, physiologic anemia of pregnancy (dilutional — plasma volume increase exceeds RBC mass increase). Respiratory: Tidal volume increases 30-40% (progesterone-driven), mild respiratory alkalosis (PaCO2 ~30 mmHg), FRC decreases (elevated diaphragm). Renal: GFR increases 50% (creatinine decreases to ~0.5-0.7 mg/dL), physiologic hydronephrosis (progesterone effect + uterine compression, more on right). Hematologic: Hypercoagulable state (increased factors VII, VIII, X, fibrinogen; decreased protein S) — risk of VTE is 5-10× higher in pregnancy.
05 Hypertensive Disorders of Pregnancy
Classification
Hypertensive disorders complicate 5-10% of all pregnancies and are a leading cause of maternal morbidity and mortality. The classification includes:
Chronic hypertension: BP ≥ 140/90 mmHg present before pregnancy or diagnosed before 20 weeks’ gestation.
Gestational hypertension: New-onset BP ≥ 140/90 mmHg after 20 weeks without proteinuria or other features of preeclampsia. Resolves by 12 weeks postpartum.
Preeclampsia: New-onset hypertension after 20 weeks PLUS proteinuria (≥ 300 mg/24 h, protein/creatinine ratio ≥ 0.3, or dipstick ≥ 2+) OR evidence of end-organ dysfunction (even without proteinuria).
Eclampsia: Preeclampsia complicated by new-onset generalized tonic-clonic seizures (not attributable to other causes).
Chronic hypertension with superimposed preeclampsia: Worsening HTN, new proteinuria, or new end-organ dysfunction after 20 weeks in a patient with chronic HTN.
Preeclampsia — With and Without Severe Features
Preeclampsia without severe features: BP ≥ 140/90 on two occasions at least 4 hours apart after 20 weeks, with proteinuria. Management: close outpatient surveillance, serial labs (CBC, CMP, LDH), fetal testing (NST, BPP), delivery at 37 weeks.
• Systolic BP ≥ 160 mmHg or diastolic BP ≥ 110 mmHg (on two occasions at least 4 hours apart, unless antihypertensives initiated sooner)
• Thrombocytopenia (platelets < 100,000/μL)
• Liver transaminases ≥ 2× upper limit of normal
• Serum creatinine > 1.1 mg/dL or doubling of creatinine
• Pulmonary edema
• New-onset headache unresponsive to medication (cerebral symptoms)
• Visual disturbances (scotomata, blurred vision)
HELLP Syndrome
HELLP = Hemolysis (microangiopathic hemolytic anemia — schistocytes, elevated LDH > 600, elevated indirect bilirubin), Elevated Liver enzymes (AST/ALT ≥ 2× ULN), Low Platelets (< 100,000/μL). Occurs in 10-20% of severe preeclampsia cases. Can present with RUQ or epigastric pain, nausea, malaise. Complications include hepatic rupture, DIC, placental abruption, renal failure, and maternal death. Management is delivery regardless of gestational age, with magnesium for seizure prophylaxis and treatment of severe hypertension.
Management
Magnesium sulfate: The drug of choice for seizure prophylaxis in severe preeclampsia and treatment of eclamptic seizures. Loading dose: 4-6 g IV over 15-20 minutes. Maintenance: 1-2 g/hour IV infusion. Therapeutic level: 4-7 mEq/L. Monitor for toxicity: loss of deep tendon reflexes (first sign, ~10 mEq/L), respiratory depression (> 12 mEq/L), cardiac arrest (> 25 mEq/L). Antidote: calcium gluconate 1 g IV.
Antihypertensives for acute severe hypertension: First-line agents are IV labetalol (20 mg initial bolus, then 40 mg, 80 mg escalating doses every 10 minutes, max 300 mg), IV hydralazine (5-10 mg every 20 minutes), or oral nifedipine (10-20 mg, may repeat in 20 minutes). Goal: reduce BP to < 160/110 within 30-60 minutes. Avoid precipitous drops that may compromise uteroplacental perfusion.
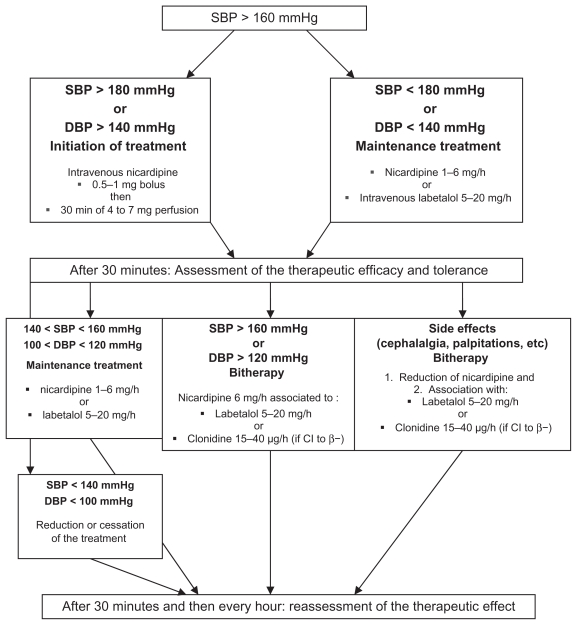
Delivery timing: Preeclampsia with severe features at ≥ 34 weeks — deliver after stabilization. At < 34 weeks, expectant management may be attempted at a tertiary center with antenatal corticosteroids, but delivery is indicated if maternal or fetal condition deteriorates. Eclampsia and HELLP — deliver regardless of gestational age after maternal stabilization.
Aspirin Prophylaxis
The USPSTF recommends low-dose aspirin (81 mg daily) starting at 12-16 weeks for women at high risk of preeclampsia. High-risk factors include: prior preeclampsia, multifetal gestation, chronic HTN, type 1 or 2 diabetes, renal disease, autoimmune disease (SLE, antiphospholipid syndrome). Moderate-risk factors (two or more needed): nulliparity, obesity (BMI > 30), family history of preeclampsia, age ≥ 35, prior adverse pregnancy outcome. The ASPRE trial demonstrated a 62% reduction in preterm preeclampsia with aspirin 150 mg nightly in high-risk women identified by first-trimester screening (UtA-PI + MAP + PAPP-A + PlGF).
Eclampsia Management
Eclamptic seizures are generalized tonic-clonic seizures occurring in the setting of preeclampsia. Can occur antepartum (38-53%), intrapartum (18-36%), or postpartum (up to 48 hours, occasionally up to 6 weeks). Management: (1) Protect airway and prevent aspiration (left lateral decubitus), (2) Magnesium sulfate 4-6 g IV bolus over 5-10 minutes if not already infusing, (3) If seizures persist, additional 2 g MgSO4 bolus. If refractory to magnesium, consider diazepam 5-10 mg IV or lorazepam 2-4 mg IV. (4) Stabilize maternal condition, (5) Monitor FHR (transient fetal bradycardia is common during/after seizure and usually resolves), (6) Plan for delivery once stabilized. Do NOT perform emergent cesarean during a seizure — stabilize the mother first.
06 Gestational Diabetes
Screening
Universal screening at 24-28 weeks is recommended. Two approaches exist:
Two-step approach (most common in the US): Step 1 — 50 g oral glucose challenge test (GCT), non-fasting. Threshold: ≥ 130 or ≥ 140 mg/dL at 1 hour (institution-dependent). If positive, proceed to Step 2 — 100 g, 3-hour oral glucose tolerance test (GTT), fasting. Two or more abnormal values = GDM diagnosis (Carpenter-Coustan criteria: fasting ≥ 95, 1 hr ≥ 180, 2 hr ≥ 155, 3 hr ≥ 140 mg/dL).
One-step approach (IADPSG criteria): 75 g, 2-hour OGTT, fasting. One or more abnormal values = GDM (fasting ≥ 92, 1 hr ≥ 180, 2 hr ≥ 153 mg/dL).
Management
Dietary modification and exercise are first-line therapy. Carbohydrate-controlled diet (~33-40% of calories from carbohydrates, distributed across 3 meals and 2-3 snacks). Glucose targets: fasting < 95 mg/dL, 1-hour postprandial < 140 mg/dL (or 2-hour < 120 mg/dL). If targets are not met within 1-2 weeks, insulin is the preferred pharmacologic agent (insulin does not cross the placenta). Metformin and glyburide are sometimes used but do cross the placenta, and recent evidence suggests insulin is superior in outcomes.
Fetal Complications
Macrosomia (birth weight > 4000 g or > 4500 g) is the hallmark complication — caused by fetal hyperinsulinemia in response to maternal hyperglycemia (Pedersen hypothesis). Macrosomia increases the risk of shoulder dystocia, brachial plexus injury, operative delivery, and birth trauma. Other fetal complications: neonatal hypoglycemia, hyperbilirubinemia, polycythemia, respiratory distress syndrome, and hypertrophic cardiomyopathy. Delivery planning: Well-controlled GDM — delivery by 39-40 weeks. GDM on medication — delivery by 39 weeks. Poorly controlled — earlier delivery based on clinical judgment. Estimated fetal weight ≥ 4500 g in a diabetic mother is a relative indication for cesarean delivery to avoid shoulder dystocia.
Shoulder Dystocia
Shoulder dystocia occurs when the fetal anterior shoulder becomes impacted behind the maternal pubic symphysis after delivery of the head. A true obstetric emergency with risk of neonatal brachial plexus injury (Erb palsy — C5-C6, most common; Klumpke palsy — C8-T1), clavicular/humeral fracture, hypoxic-ischemic encephalopathy, and maternal hemorrhage/lacerations.
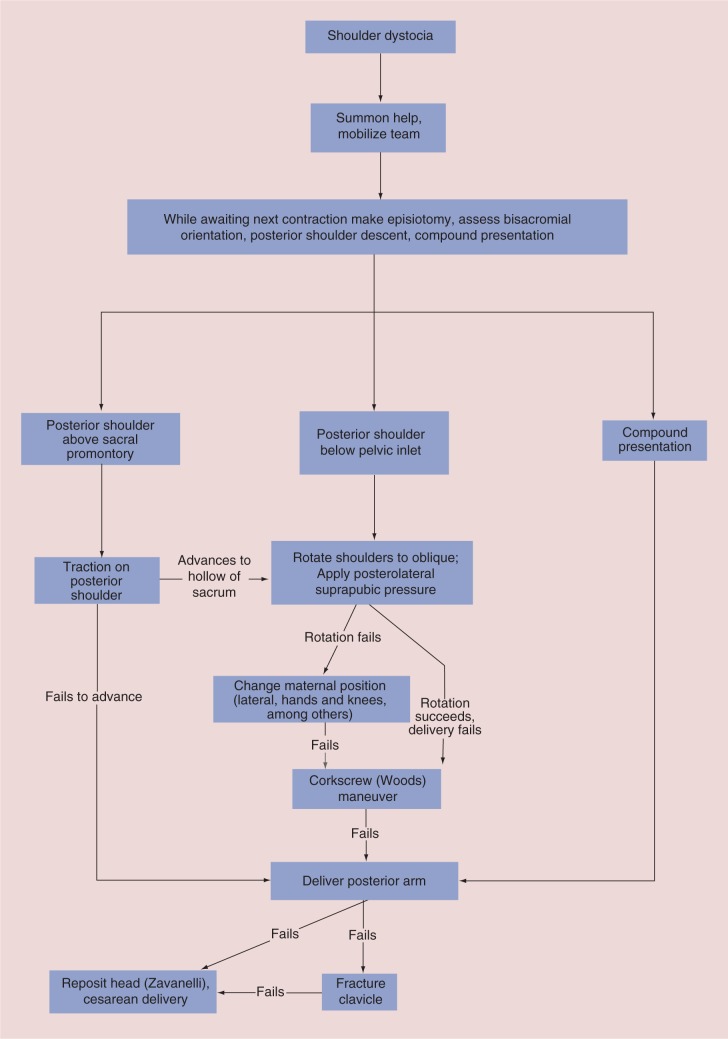
H — Call for Help (additional nurses, OB, anesthesia, NICU)
E — Evaluate for Episiotomy (provides more room for maneuvers, does not resolve bony impaction)
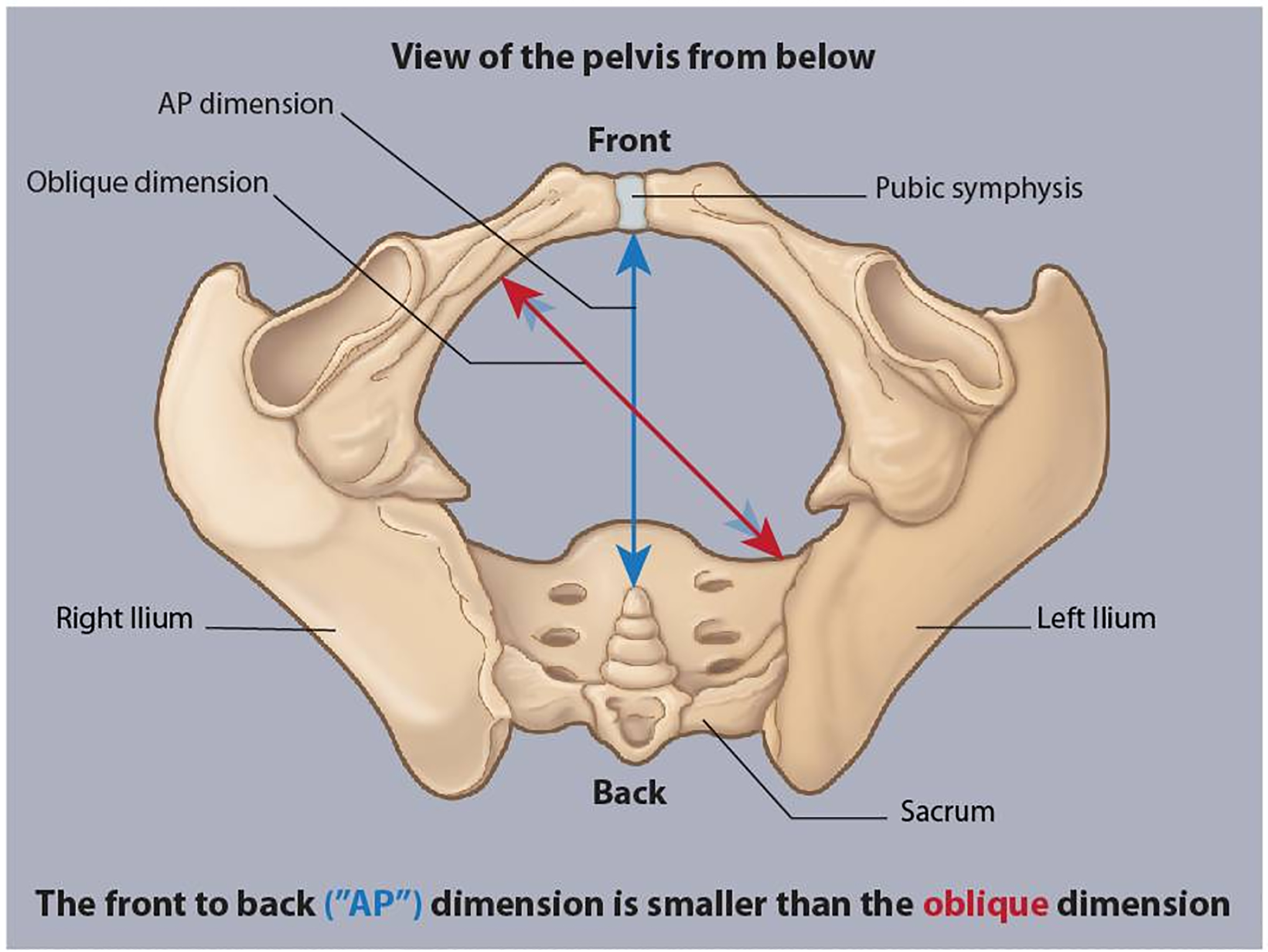
L — Legs (McRoberts maneuver: hyperflexion of maternal hips to flatten the sacrum and increase AP diameter of the pelvic outlet — first and most effective maneuver, resolves 42% of cases alone)
P — suprapubic Pressure (downward and lateral pressure on the anterior shoulder to push it under the symphysis; do NOT apply fundal pressure)
E — Enter maneuvers (internal rotation — Rubin II: pressure on the posterior aspect of the anterior shoulder to adduct and rotate; Woods corkscrew: rotation of the posterior shoulder 180°)
R — Remove the posterior arm (deliver the posterior arm by sweeping it across the fetal chest)
R — Roll the patient (Gaskin maneuver: all-fours position, uses gravity and changes pelvic diameters)
Last resort: Zavanelli maneuver (cephalic replacement — push the fetal head back into the vagina for emergent cesarean) or intentional clavicular fracture (rarely performed).
Pregestational Diabetes in Pregnancy
Women with preexisting type 1 or type 2 diabetes require preconception counseling and optimization. Hemoglobin A1c < 6.5% before conception is ideal (A1c > 10% carries significantly elevated risk of congenital anomalies). Key risks: congenital anomalies (cardiac defects, neural tube defects, caudal regression syndrome — most specific for maternal diabetes), macrosomia, IUGR (in vascular disease), preeclampsia, polyhydramnios, preterm birth, stillbirth, DKA. Management: insulin therapy (tight glycemic control), folic acid supplementation (4 mg/day for NTD prevention), first-trimester detailed US, fetal echocardiography (18-22 weeks), serial growth ultrasounds, antenatal fetal surveillance starting 32-34 weeks.
07 Placental Abnormalities
Placenta Previa
Placenta previa is defined as placental tissue covering or adjacent to the internal cervical os. Previously classified as complete, partial, marginal, and low-lying; current terminology distinguishes placenta previa (covering the os) from low-lying placenta (within 2 cm of the os but not covering it). Incidence ~0.5%. Risk factors: prior cesarean delivery, prior uterine surgery, multiparity, advanced maternal age, multiple gestation, smoking.
Clinical presentation: Painless, bright red vaginal bleeding, typically in the late second or third trimester. Diagnosis by transvaginal ultrasound (safe and more accurate than transabdominal). Management: Pelvic rest, no digital cervical exams, serial US for possible placental migration (many low-lying placentas resolve by third trimester as the lower uterine segment develops). Delivery by cesarean section at 36-37 weeks (or earlier if bleeding is severe or uncontrolled). Previa diagnosed in the second trimester should be rechecked at 32-36 weeks.
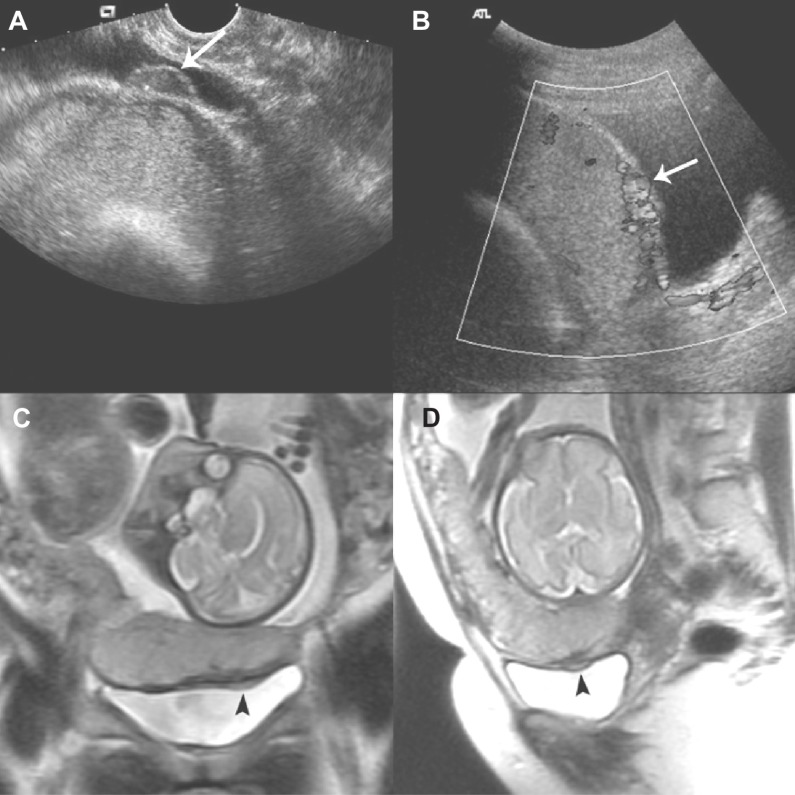
Placenta Accreta Spectrum (PAS)
Abnormal placental adherence/invasion into the myometrium. Incidence is rising with increasing cesarean delivery rates (~1 in 272 pregnancies).
Accreta: Placental villi attach directly to the myometrium (absent decidua basalis/Nitabuch layer). Most common form (~75%).
Increta: Placental villi invade into the myometrium (~18%).
Percreta: Placental villi penetrate through the myometrium to the serosa and potentially into adjacent organs (bladder, bowel) (~7%). Highest morbidity and mortality.
Key risk factor: Prior cesarean delivery + placenta previa. Risk increases exponentially with number of prior cesareans: 1 prior CD + previa = 3% risk; 2 prior = 11%; 3 prior = 40%; 4 prior = 61%. Management: planned cesarean hysterectomy at 34-36 weeks at a tertiary center with a multidisciplinary team (MFM, gynecologic oncology, interventional radiology, urology, blood bank). Do NOT attempt placental removal.
Placental Abruption
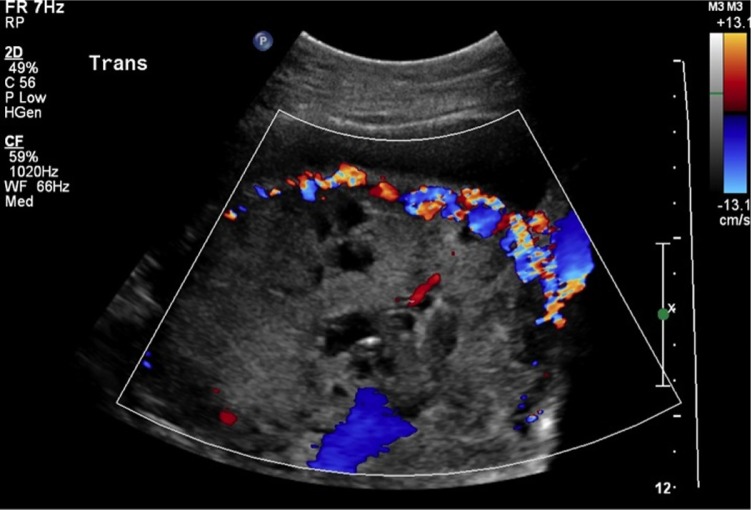
Placental Abruption
Placental abruption is premature separation of the normally implanted placenta from the uterine wall before delivery. Incidence ~1%. Risk factors: chronic hypertension (strongest), preeclampsia, trauma, cocaine use, prior abruption, PPROM, thrombophilia, short umbilical cord. Clinical presentation: Painful vaginal bleeding (in ~80% — concealed hemorrhage occurs in 20%), uterine tenderness, hypertonicity ("board-like" uterus), non-reassuring fetal heart rate tracing, and signs of maternal hemodynamic compromise. Diagnosis is clinical — ultrasound has poor sensitivity (~25%) for abruption (may show retroplacental clot).
Vasa Previa
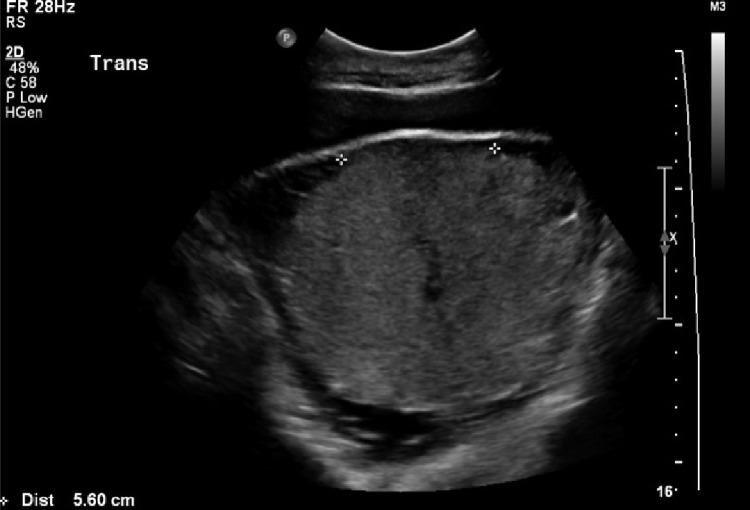
Vasa previa occurs when fetal blood vessels (from a velamentous cord insertion or succenturiate placental lobe) traverse the fetal membranes over the internal cervical os, unprotected by placental tissue or Wharton jelly. Rupture of membranes can cause fetal vessel laceration and rapid fetal exsanguination (fetal blood volume is only ~250 mL at term). Mortality if undiagnosed approaches 60%; with prenatal diagnosis and planned cesarean at 35-36 weeks, survival exceeds 95%. Diagnosed by transvaginal US with color Doppler. The Apt test (alkali denaturation test) differentiates fetal blood (resistant to denaturation, stays pink) from maternal blood (denatures to brown) in vaginal bleeding.
08 Preterm Labor & Cervical Insufficiency
Preterm Labor
Preterm labor is defined as regular uterine contractions with cervical change between 20 0/7 and 36 6/7 weeks. Preterm birth is the leading cause of neonatal morbidity and mortality worldwide. Risk factors: prior preterm birth (strongest predictor), short cervix, multiple gestation, uterine anomalies, infections (bacterial vaginosis, UTI, chorioamnionitis), PPROM, polyhydramnios, cervical surgery (LEEP, conization).
Tocolytic Agents
Nifedipine (calcium channel blocker): First-line tocolytic. 20-30 mg loading dose, then 10-20 mg every 4-6 hours. Side effects: hypotension, headache, flushing. Avoid with magnesium (severe hypotension).
Indomethacin (NSAID/COX inhibitor): 50-100 mg loading, then 25-50 mg every 6 hours. Very effective but limited to < 32 weeks and < 48-72 hours of use due to risk of premature closure of the ductus arteriosus, oligohydramnios, and neonatal renal impairment.
Magnesium sulfate: 4-6 g IV load, then 2-4 g/hour. Dual role: tocolysis and fetal neuroprotection (< 32 weeks). Side effects: flushing, nausea, muscle weakness, respiratory depression at high levels.
Terbutaline (β-agonist): 0.25 mg SQ. Limited to < 48-72 hours, not for maintenance. Black box warning for serious cardiovascular events. Rarely used as first-line.
Antenatal Corticosteroids
Betamethasone 12 mg IM × 2 doses, 24 hours apart (preferred), or dexamethasone 6 mg IM × 4 doses, 12 hours apart. Indicated at 24 0/7 to 33 6/7 weeks when preterm delivery is anticipated within 7 days. Reduces neonatal RDS by 50%, intraventricular hemorrhage, necrotizing enterocolitis, and neonatal death. A single rescue course may be given if > 14 days from the initial course and < 34 weeks. Maximum benefit 48 hours to 7 days after administration. Also recommended at 34 0/7 to 36 6/7 weeks (late preterm) if no prior course given.
Cervical Insufficiency & Cerclage
Cervical insufficiency is painless cervical dilation in the second trimester leading to pregnancy loss, typically without contractions. History-indicated cerclage (McDonald or Shirodkar): Placed at 12-14 weeks in patients with a history of ≥ 3 second-trimester losses or prior cerclage. Ultrasound-indicated cerclage: Placed when cervical length is < 25 mm before 24 weeks in a singleton pregnancy with a history of prior preterm birth. Rescue/emergent cerclage: Placed for advanced cervical dilation (membranes visible at os) — higher risk, variable success. Vaginal progesterone (200 mg nightly) is an alternative for short cervix (< 25 mm) without prior preterm birth.
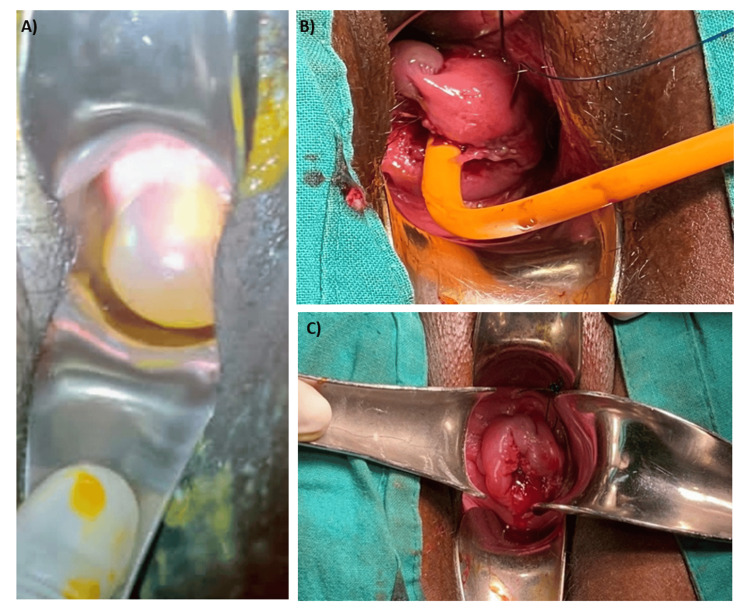
Preterm Premature Rupture of Membranes (PPROM)
PPROM is rupture of the fetal membranes before 37 weeks’ gestation in the absence of labor. Diagnosed by: (1) sterile speculum exam showing pooling of fluid in the posterior fornix, (2) Nitrazine test (amniotic fluid is alkaline, pH ≥ 7.1, turns paper blue), (3) fern test (amniotic fluid dries in a ferning pattern on a glass slide). If equivocal, AmniSure (placental alpha macroglobulin-1) or ROM Plus (PAMG-1 + AFP) are highly sensitive and specific bedside tests. Ultrasound may show oligohydramnios but is not diagnostic alone. Avoid digital cervical exam in PPROM (increases infection risk) unless delivery is imminent.
Management: Depends on gestational age. ≥ 34 weeks: Deliver (induction or expectant management of 12-24 hours for GBS prophylaxis). 24-33 6/7 weeks: Expectant management (latency antibiotics, antenatal corticosteroids, GBS prophylaxis, fetal monitoring). Latency antibiotics (ampicillin 2 g IV q6h × 48 hours + azithromycin 1 g PO × 1, then amoxicillin 250 mg PO TID × 5 days) prolong latency ~7 days and reduce chorioamnionitis and neonatal infection. Magnesium sulfate for neuroprotection if < 32 weeks. Tocolysis is NOT recommended in PPROM (may mask signs of infection). Indications for delivery regardless of GA: chorioamnionitis (maternal fever, uterine tenderness, fetal tachycardia, purulent discharge), non-reassuring fetal status, placental abruption, cord prolapse.
09 Labor & Delivery
Stages of Labor
First stage: Onset of regular contractions to complete cervical dilation (10 cm). Divided into: Latent phase (0-6 cm, slow irregular progress, may last hours to days) and Active phase (6-10 cm, more rapid dilation, expected rate ≥ 1 cm/hr for nulliparas historically, though modern data suggest 0.5-0.7 cm/hr is acceptable). Active phase arrest: no cervical change for ≥ 6 hours with adequate contractions (≥ 200 Montevideo units by IUPC) or ≥ 4 hours with adequate contractions and no change.
Second stage: Complete dilation to delivery of the infant. Pushing stage. Acceptable duration: nulliparas up to 3 hours (4 hours with epidural); multiparas up to 2 hours (3 hours with epidural). Arrest of descent: no progress for ≥ 2-3 hours of pushing in nulliparas, ≥ 1-2 hours in multiparas.
Third stage: Delivery of the infant to delivery of the placenta. Active management (gentle cord traction + uterotonic) reduces blood loss. Normal duration < 30 minutes; retained placenta if > 30 minutes — may require manual extraction or curettage.
Labor Curves & Modern Definitions
The classic Friedman curve (1955) defined active labor as beginning at 4 cm with expected dilation of 1.2 cm/hr (nulliparas) and 1.5 cm/hr (multiparas). The Contemporary Labor Curve (Zhang et al., Consortium on Safe Labor) demonstrated that active labor does not reliably begin until 6 cm, cervical dilation from 4-6 cm is slow and variable, and labor progress accelerates significantly only after 6 cm. This led to updated ACOG/SMFM guidelines: do not diagnose active-phase arrest before 6 cm, and allow more time before declaring arrest of labor.
Labor Augmentation
Oxytocin (Pitocin): The primary agent for labor augmentation and induction. Typically started at 1-2 mU/min and increased every 15-30 minutes. Goal: 3-5 contractions per 10 minutes. Complications: tachysystole (> 5 contractions/10 min), uterine hyperstimulation, fetal heart rate decelerations, uterine rupture (especially in prior CD), water intoxication (ADH-like effect at high doses). Amniotomy (artificial rupture of membranes) augments labor by increasing prostaglandin release and allowing the fetal head to directly apply pressure to the cervix.
Operative Vaginal Delivery
Forceps: Classified by station — outlet (fetal skull visible at introitus), low (station ≥ +2), and mid (station 0 to +2). Prerequisites: full dilation, ruptured membranes, vertex presentation, known position, adequate pelvis, empty bladder, adequate anesthesia. Types: Simpson (most common, for molded head), Tucker-McLane (solid blades), Kielland (for rotation), Piper (for aftercoming head in breech). Complications: maternal lacerations (3rd/4th degree), fetal facial nerve injury, cephalohematoma, intracranial hemorrhage.
Vacuum: Applied to the fetal vertex at the "flexion point" (3 cm anterior to the posterior fontanelle). Maximum 3 pop-offs or 20-30 minutes. Complications: cephalohematoma, subgaleal hemorrhage (most dangerous — can cause life-threatening neonatal hemorrhage), retinal hemorrhage. Contraindicated at < 34 weeks, face or breech presentation, and suspected fetal coagulopathy.
Cesarean Section
Indications: Non-reassuring fetal status, arrest of labor (first or second stage), malpresentation (breech, transverse lie), placenta previa, prior classical CD, cord prolapse, active genital herpes at delivery, certain fetal anomalies.
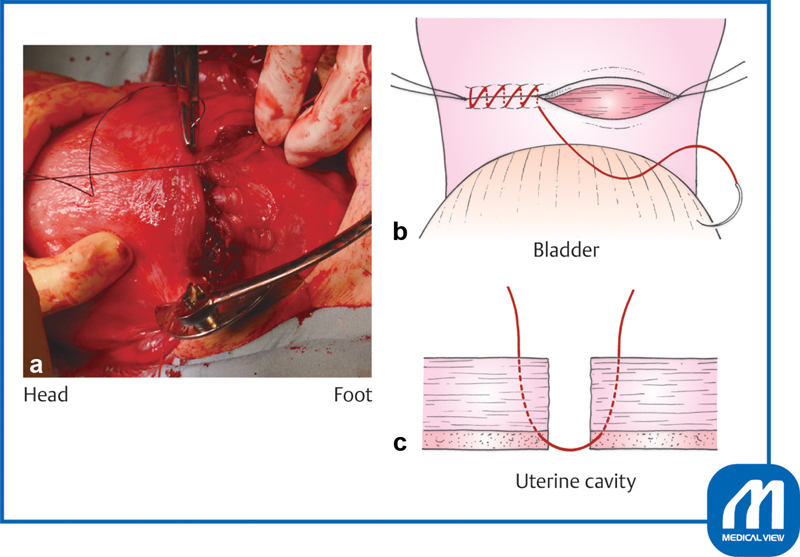
Technique (low transverse): Pfannenstiel or Joel-Cohen skin incision → fascial incision → rectus muscle separation → peritoneal entry → bladder flap creation → low transverse uterine incision (through the lower uterine segment) → delivery of the infant → placental delivery → uterine closure (single vs double layer) → fascial closure. A classical (vertical) uterine incision is reserved for specific situations: preterm breech with poorly formed lower segment, anterior placenta previa, transverse lie with back-down presentation. Classical CD mandates repeat cesarean for all future pregnancies (VBAC contraindicated due to 4-9% uterine rupture risk vs 0.5-0.7% for low transverse).
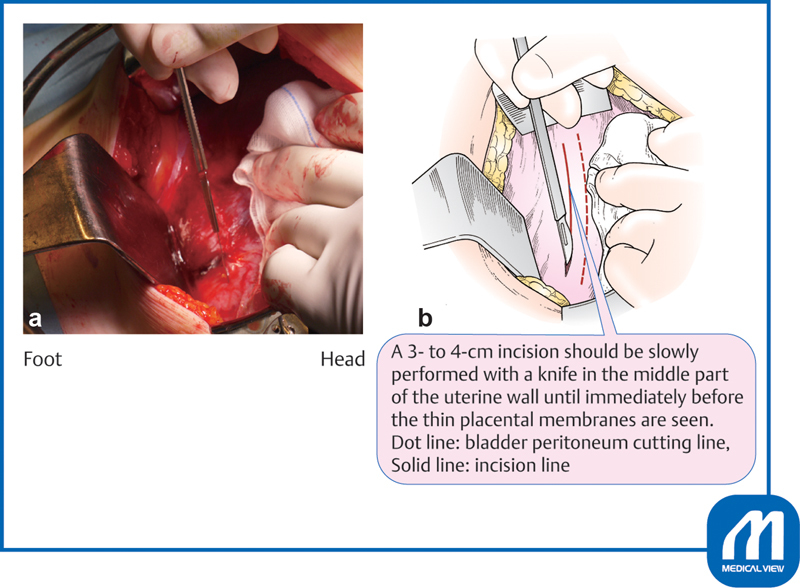
Trial of labor after cesarean (TOLAC): Candidates: one prior low transverse CD, no contraindications to vaginal delivery, adequate facility for emergent CD. VBAC success rate: 60-80%. Uterine rupture risk: 0.5-0.7%. The MFMU VBAC calculator incorporates maternal age, BMI, prior vaginal delivery, indication for prior CD, dilation, and effacement to predict success. Contraindications to TOLAC: prior classical or T-incision, prior uterine rupture, ≥ 2 prior CDs (relative), breech presentation.
Perineal Lacerations
Classified by depth of tissue involvement:
| Degree | Extent of Injury | Repair |
|---|---|---|
| 1st degree | Vaginal mucosa and perineal skin only | May not require repair; if repaired, simple running suture |
| 2nd degree | Extends into perineal body musculature (bulbocavernosus, transverse perineal) | Layered closure of muscle, vaginal mucosa, and skin |
| 3rd degree | Involves the anal sphincter (3a: < 50% external sphincter; 3b: > 50% external sphincter; 3c: internal sphincter also torn) | End-to-end or overlapping sphincter repair by trained provider; antibiotics |
| 4th degree | Extends through the anal sphincter into the rectal mucosa | Rectal mucosa closure first (absorbable suture), then sphincter repair, then perineal body. Antibiotics, stool softeners |
Obstetric Anesthesia
Neuraxial anesthesia (epidural, combined spinal-epidural) is the most effective form of labor analgesia. Epidural: Catheter placed in the epidural space (L3-L4 or L2-L3 interspace) provides continuous infusion of dilute local anesthetic (bupivacaine 0.0625-0.125%) + opioid (fentanyl). Can be titrated for labor and topped up for cesarean section. Combined spinal-epidural (CSE): Rapid onset from intrathecal injection + continuous epidural catheter. Spinal anesthesia: Single-shot intrathecal injection, typically for planned cesarean (bupivacaine 12 mg + fentanyl + morphine). Complications of neuraxial: hypotension (most common — treat with IV fluids, ephedrine, phenylephrine), post-dural puncture headache (PDPH — positional, worse upright; treat with blood patch if conservative measures fail), high/total spinal (respiratory arrest — emergency), epidural hematoma (rare, associated with coagulopathy). Contraindications: patient refusal, coagulopathy, local infection at insertion site, uncorrected hypovolemia, raised ICP.
10 Postpartum Hemorrhage
Definition & Epidemiology
Postpartum hemorrhage (PPH) is defined as cumulative blood loss ≥ 1000 mL or blood loss accompanied by signs/symptoms of hypovolemia within 24 hours of delivery (ACOG revised definition). PPH complicates 1-5% of deliveries and is the leading cause of maternal mortality worldwide. Primary PPH occurs within 24 hours; secondary PPH occurs from 24 hours to 12 weeks postpartum (usually retained products or endometritis).
Etiology — The 4 T's
Tone (70%): Uterine atony — the most common cause. Risk factors: overdistension (macrosomia, polyhydramnios, multiple gestation), prolonged labor, chorioamnionitis, uterine relaxants (magnesium, halogenated anesthetics), grand multiparity.
Trauma (20%): Genital tract lacerations (cervical, vaginal, perineal), uterine rupture, uterine inversion, episiotomy extension.
Tissue (10%): Retained placenta or placental fragments, abnormally adherent placenta (accreta spectrum), retained blood clots.
Thrombin (< 1%): Coagulopathy — preexisting (von Willebrand disease, thrombocytopenia) or acquired (DIC from abruption, amniotic fluid embolism, HELLP, massive transfusion dilutional coagulopathy).
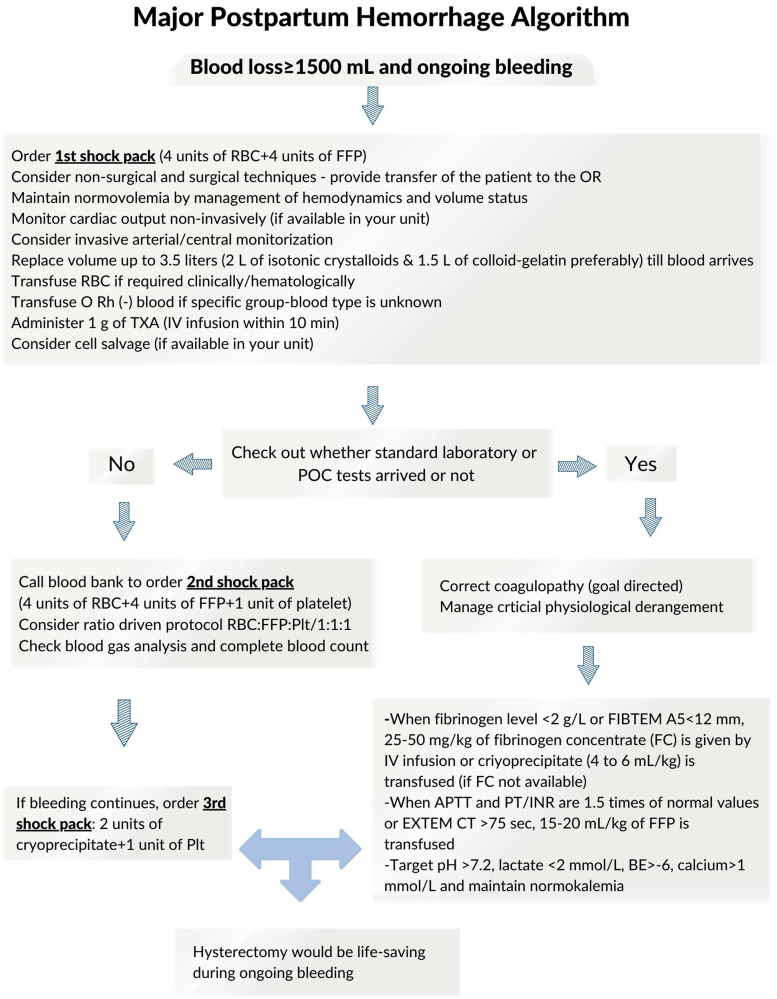
Management Algorithm
Stepwise escalation based on response to treatment:
Step 1 — Uterine massage & uterotonics: Bimanual uterine massage. Oxytocin 10-40 units in 1L NS IV infusion. Methylergonovine (Methergine) 0.2 mg IM (contraindicated in hypertension). Carboprost (Hemabate/15-methyl PGF2α) 0.25 mg IM every 15-90 min, max 8 doses (contraindicated in asthma). Misoprostol (Cytotec) 800-1000 mcg rectal or sublingual.
Step 2 — Intrauterine tamponade: Bakri balloon (filled with 300-500 mL saline) or other uterine tamponade balloon. Condom catheter tamponade is an alternative in low-resource settings.
Step 3 — Surgical interventions: B-Lynch compression suture (brace suture around the uterus), uterine artery ligation (O’Leary stitch), internal iliac artery ligation. Uterine artery embolization (UAE) by interventional radiology if patient is hemodynamically stable enough for transfer.
Step 4 — Hysterectomy: Peripartum hysterectomy is the definitive treatment when all other measures fail. Usually subtotal (supracervical) hysterectomy for speed.
Massive transfusion protocol: Activated when ongoing hemorrhage exceeds 1500 mL or hemodynamic instability persists. Target ratio of PRBCs:FFP:platelets is 1:1:1. Tranexamic acid (TXA) 1 g IV should be administered within 3 hours of delivery onset (WOMAN trial: reduced death from hemorrhage by 19%). Cryoprecipitate for fibrinogen < 200 mg/dL. Keep calcium normalized during massive transfusion (citrate in blood products chelates calcium).
11 Ectopic Pregnancy
Epidemiology & Risk Factors
Ectopic pregnancy occurs when a fertilized ovum implants outside the endometrial cavity. Incidence ~2% of all pregnancies. It is the leading cause of first-trimester maternal death. Sites: tubal (~95% — ampulla 70%, isthmus 12%, fimbrial 11%, interstitial/cornual 2%), ovarian (~3%), abdominal (~1%), cervical (< 1%), cesarean scar (< 1%). Risk factors: prior ectopic (strongest — 10-15% recurrence), PID/tubal damage, prior tubal surgery, IUD in situ (does not increase absolute risk but shifts proportion of pregnancies to ectopic), smoking, infertility treatment (especially IVF), endometriosis, DES exposure.
Diagnosis
Clinical presentation: Amenorrhea, vaginal bleeding, and lower abdominal/pelvic pain (classic triad, present in ~50%). Ruptured ectopic presents with acute abdomen, peritoneal signs, shoulder pain (diaphragmatic irritation from hemoperitoneum), and hemodynamic instability.
Diagnostic algorithm: Positive pregnancy test (urine or serum β-hCG) → transvaginal US. The discriminatory level of β-hCG (the level at which an intrauterine pregnancy should be visible on TVUS) is 1500-3500 mIU/mL. If β-hCG is above the discriminatory level and no IUP is seen, ectopic pregnancy is highly suspected. If β-hCG is below discriminatory level, serial measurements every 48 hours: normal IUP should have ≥ 53% rise in 48 hours. A plateau or abnormal rise suggests ectopic or nonviable IUP.
Medical Management — Methotrexate
Absolute criteria for medical management: Hemodynamically stable, no signs of rupture, able to comply with follow-up, no contraindications to methotrexate.
Optimal candidates: β-hCG < 5000 mIU/mL, no fetal cardiac activity on US, ectopic mass < 3.5 cm.
Contraindications: Hemodynamic instability, signs of rupture, breastfeeding, immunodeficiency, hepatic/renal disease, blood dyscrasias, active pulmonary disease, peptic ulcer disease, inability to follow up.
Pre-treatment labs: CBC, CMP (liver, renal function), blood type & Rh, β-hCG level.
Single-dose protocol: Methotrexate 50 mg/m² IM. Check β-hCG on days 4 and 7. Expect transient rise by day 4. Require ≥ 15% decline between days 4 and 7. If inadequate decline, repeat dose. Follow β-hCG weekly until undetectable. Success rate: 87-93% (single or multiple doses). Two-dose protocol: MTX on days 1 and 4, with β-hCG monitoring on days 4 and 7. Multi-dose protocol: MTX alternating with leucovorin on days 1, 3, 5, 7 with hCG monitoring — used for higher hCG levels.
Surgical Management
Salpingostomy: Linear incision over the ectopic, removal of products, tube preserved. Indicated when future fertility is desired and the contralateral tube is absent or damaged. Requires β-hCG follow-up (10-15% persistent trophoblastic tissue).
Salpingectomy: Removal of the affected tube. Preferred in most cases — eliminates risk of persistent ectopic, no significant difference in future fertility when the contralateral tube is normal. Indicated for ruptured ectopic, recurrent ectopic in the same tube, severely damaged tube. Laparoscopy is the preferred surgical approach (vs laparotomy) when the patient is hemodynamically stable.
12 Fetal Monitoring & Rh Isoimmunization
NICHD Nomenclature for Fetal Heart Rate Interpretation
Baseline FHR: Mean FHR during a 10-minute segment, rounded to increments of 5 bpm. Normal: 110-160 bpm. Tachycardia: > 160 bpm (fever, chorioamnionitis, fetal anemia, maternal medications). Bradycardia: < 110 bpm (late fetal compromise, cord compression, maternal hypothermia).
Baseline variability: Fluctuations in the FHR baseline of ≥ 2 cycles/min. Absent: amplitude undetectable (concerning). Minimal: amplitude 1-5 bpm. Moderate: amplitude 6-25 bpm (most reassuring feature on a tracing). Marked: amplitude > 25 bpm.
Accelerations: Abrupt increase in FHR ≥ 15 bpm above baseline lasting ≥ 15 seconds (before 32 weeks: ≥ 10 bpm for ≥ 10 seconds). Presence of accelerations is reassuring.
Decelerations: Early — gradual onset, mirrors contraction, nadir coincides with peak of contraction. Caused by fetal head compression. Benign. Late — gradual onset, onset after contraction begins, nadir after contraction peak. Caused by uteroplacental insufficiency. Concerning, especially if repetitive with absent variability. Variable — abrupt onset (< 30 sec to nadir), variable in shape and timing. Caused by umbilical cord compression. Mild variables are common; severe or persistent variables with absent variability are concerning. Prolonged — decrease ≥ 15 bpm lasting 2-10 minutes.
Three-Tier FHR Interpretation System
| Category | Features | Interpretation | Management |
|---|---|---|---|
| Category I (Normal) | Baseline 110-160, moderate variability, no late or variable decelerations, accelerations may/may not be present | Normal acid-base status, no intervention needed | Routine monitoring |
| Category II (Indeterminate) | All tracings not meeting Category I or III criteria (e.g., minimal variability without recurrent decels, absent accelerations, recurrent variable decels with moderate variability) | Not predictive of abnormal acid-base status but requires evaluation | Continued surveillance, intrauterine resuscitation, further evaluation |
| Category III (Abnormal) | Absent variability WITH recurrent late decelerations, recurrent variable decelerations, or bradycardia; OR sinusoidal pattern | Abnormal fetal acid-base status | Intrauterine resuscitation; if unresolved, expeditious delivery |
Intrauterine Resuscitation
For non-reassuring FHR patterns: (1) Maternal repositioning (left lateral decubitus or hands-and-knees), (2) IV fluid bolus (to improve uteroplacental perfusion), (3) Discontinue oxytocin if running, (4) Supplemental oxygen (limited evidence but commonly used), (5) Amnioinfusion for recurrent variable decelerations (infusion of normal saline into the uterus to relieve cord compression), (6) Terbutaline 0.25 mg SQ for tachysystole, (7) Address underlying cause (maternal hypotension, fever, cord prolapse).
Rh Isoimmunization
An Rh-negative mother carrying an Rh-positive fetus can develop anti-D antibodies after fetomaternal hemorrhage (delivery, miscarriage, trauma, invasive procedures). In subsequent Rh-positive pregnancies, these IgG antibodies cross the placenta and cause hemolytic disease of the fetus and newborn (HDFN) — fetal anemia, hydrops fetalis, kernicterus.
RhoGAM (anti-D immunoglobulin) prophylaxis: 300 mcg IM at 28 weeks’ gestation and within 72 hours of delivery of an Rh-positive infant. Also given after: miscarriage/abortion, ectopic pregnancy, amniocentesis/CVS, abdominal trauma, external cephalic version, antepartum bleeding. The standard 300 mcg dose covers up to 30 mL of fetal whole blood (15 mL of fetal RBCs). The Kleihauer-Betke test quantifies fetomaternal hemorrhage — if > 30 mL, additional RhoGAM doses are needed.
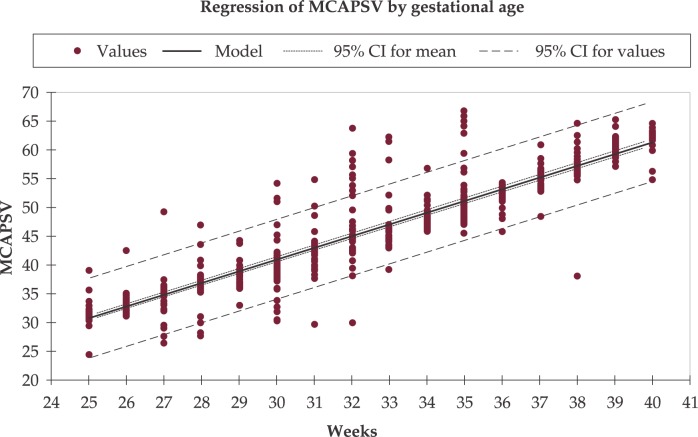
Monitoring sensitized pregnancies: Maternal anti-D antibody titers are followed. Once the critical titer is reached (typically 1:16 or 1:32), middle cerebral artery (MCA) Doppler peak systolic velocity is monitored serially. MCA-PSV > 1.5 multiples of the median (MoM) for gestational age indicates moderate-severe fetal anemia and is an indication for cordocentesis (percutaneous umbilical blood sampling) and intrauterine transfusion.
Antepartum Fetal Surveillance
Methods of assessing fetal wellbeing in the third trimester:
Non-stress test (NST): Monitoring FHR for accelerations over 20-40 minutes. Reactive: ≥ 2 accelerations of ≥ 15 bpm lasting ≥ 15 seconds in 20 minutes. Non-reactive: does not meet criteria — proceed to further testing (BPP, vibroacoustic stimulation).
Biophysical profile (BPP): Five parameters, each scored 0 or 2 (max 10): NST reactivity, fetal breathing movements (≥ 1 episode of ≥ 30 sec in 30 min), fetal movement (≥ 3 body/limb movements), fetal tone (≥ 1 episode of extension/flexion), amniotic fluid volume (single deepest pocket ≥ 2 cm). Score 8-10: reassuring. 6: equivocal. ≤ 4: abnormal — consider delivery.
Modified BPP: NST + amniotic fluid index (AFI). Most commonly used surveillance method (combines short-term and long-term fetal assessment).
Contraction stress test (CST): Monitor FHR during uterine contractions (spontaneous or oxytocin-induced). Negative: no late decelerations. Positive: late decelerations with ≥ 50% of contractions — concerning for uteroplacental insufficiency.
Umbilical artery Doppler: Used specifically for fetal growth restriction (FGR). Normal: low-resistance flow. Abnormal: elevated S/D ratio, absent end-diastolic flow (AEDF), or reversed end-diastolic flow (REDF — most ominous, associated with fetal acidemia and stillbirth).
13 Cervical Cancer
HPV & Cervical Carcinogenesis
Human papillomavirus (HPV) is the causative agent in > 99% of cervical cancers. High-risk types: HPV 16 (causes ~50% of cervical cancers) and HPV 18 (causes ~20%). Other high-risk types: 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68. Low-risk types (6, 11) cause condylomata (genital warts) but not cancer. The oncoproteins E6 (inactivates p53) and E7 (inactivates Rb) drive malignant transformation. The HPV vaccine (9-valent Gardasil 9) covers types 6, 11, 16, 18, 31, 33, 45, 52, 58 and is recommended for all individuals aged 9-26 (catch-up through 45).
Screening — Pap Smear & HPV Testing
ASCCP/ACS 2020 guidelines: Preferred strategy is primary HPV testing alone every 5 years starting at age 25. Acceptable alternatives: co-testing (Pap + HPV) every 5 years or Pap alone every 3 years from 21-65. Screening ends at age 65 if adequate prior screening and no history of CIN2+.
Bethesda Classification & ASCCP Management
NILM: Negative for intraepithelial lesion or malignancy. ASC-US: Atypical squamous cells of undetermined significance — reflex HPV testing; if HPV+, colposcopy. ASC-H: Atypical squamous cells, cannot exclude HSIL — colposcopy recommended. LSIL: Low-grade squamous intraepithelial lesion (corresponds to CIN 1/HPV effect) — colposcopy. HSIL: High-grade squamous intraepithelial lesion (corresponds to CIN 2-3) — colposcopy with immediate excision or biopsy. AGC: Atypical glandular cells — colposcopy + endocervical curettage + endometrial biopsy (if ≥ 35 or abnormal bleeding).
Colposcopy: Application of 3-5% acetic acid to the cervix under magnification. Abnormal findings: acetowhite epithelium, punctation, mosaic pattern, atypical vessels, irregular surface contour. Biopsies of suspicious areas and endocervical curettage (ECC) are performed. Histologic results (CIN 1, 2, 3 / AIS) guide further management per ASCCP algorithms.
FIGO Staging & Treatment
Cervical cancer is clinically staged (FIGO 2018 revision now allows imaging and pathologic findings to be incorporated):
| Stage | Description | Primary Treatment |
|---|---|---|
| IA1 | Stromal invasion ≤ 3 mm depth, ≤ 7 mm width | Cone biopsy (if margins clear, fertility desired) or simple hysterectomy |
| IA2 | Stromal invasion > 3 to ≤ 5 mm depth, ≤ 7 mm width | Modified radical hysterectomy + pelvic LND (or radical trachelectomy if fertility desired) |
| IB1 | Clinically visible lesion ≤ 2 cm | Radical hysterectomy + pelvic LND (± para-aortic LND) or chemoradiation |
| IB2 | Clinically visible lesion > 2 to ≤ 4 cm | Radical hysterectomy + LND or chemoradiation |
| IB3 | Clinically visible lesion > 4 cm | Chemoradiation (cisplatin-based) preferred |
| IIA | Upper 2/3 vagina involvement | Chemoradiation or radical hysterectomy (IIA1 ≤ 4 cm) |
| IIB | Parametrial invasion | Chemoradiation (cisplatin 40 mg/m² weekly + EBRT + brachytherapy) |
| III | Lower 1/3 vagina (IIIA), pelvic wall/hydronephrosis (IIIB), pelvic/para-aortic LN+ (IIIC) | Chemoradiation ± extended-field RT for para-aortic nodes |
| IV | Bladder/rectal mucosa (IVA), distant metastasis (IVB) | Chemoradiation (IVA) or systemic therapy (IVB) ± pembrolizumab (PD-L1+) |
LEEP (loop electrosurgical excision procedure): Excision of the transformation zone for CIN 2/3 with a thin wire loop and electrosurgical current. Can be performed in the office. Cold-knife conization: Preferred when AIS (adenocarcinoma in situ) is suspected or when LEEP margins are positive. Provides a specimen with intact margins for pathologic evaluation.
14 Endometrial Cancer
Epidemiology & Risk Factors
Endometrial cancer is the most common gynecologic malignancy in the United States. Mean age at diagnosis: 63 years. The primary risk factor is unopposed estrogen exposure: obesity (adipose tissue aromatization of androgens to estrogens), early menarche, late menopause, nulliparity, anovulation (PCOS), estrogen-only HRT without progestin, tamoxifen (acts as estrogen agonist in the endometrium). Protective factors: combined OCP use (50% risk reduction with ≥ 5 years of use), progestins, pregnancy, physical activity.
Type I (endometrioid, ~80%): Estrogen-driven, arise from atypical endometrial hyperplasia, generally low-grade, favorable prognosis. Type II (~20%): Non-estrogen-driven, high-grade histologies (serous, clear cell, carcinosarcoma), arise from atrophic endometrium, behave aggressively with early extrauterine spread.
Molecular Classification (TCGA/ProMisE)
POLE ultramutated: Excellent prognosis regardless of histologic grade. High mutation burden, robust immune response. May not need adjuvant therapy even with adverse features.
MSI-hypermutated (dMMR): Microsatellite instable, deficient mismatch repair. Associated with Lynch syndrome (germline MLH1, MSH2, MSH6, PMS2 mutations). Good prognosis. Responds to immune checkpoint inhibitors (pembrolizumab, dostarlimab).
Copy number low (p53 wild-type): Intermediate prognosis. Most common subtype. Typically low-grade endometrioid.
Copy number high (p53 mutant/abnormal): Worst prognosis. Includes most serous carcinomas. Aggressive behavior. Adjuvant chemotherapy indicated.
Staging & Surgical Treatment
Endometrial cancer is surgically staged. Standard surgical staging: total hysterectomy, bilateral salpingo-oophorectomy (TH/BSO), with sentinel lymph node (SLN) mapping (using indocyanine green or technetium-99m). Full pelvic and para-aortic lymphadenectomy is reserved for high-risk histologies or failed SLN mapping. Minimally invasive approach (laparoscopic or robotic) is the standard of care (GOG LAP2 trial).
| FIGO Stage | Description | Adjuvant Therapy |
|---|---|---|
| IA | Confined to endometrium or < 50% myometrial invasion | Observation (low-grade); vaginal cuff brachytherapy (intermediate risk) |
| IB | ≥ 50% myometrial invasion | Vaginal cuff brachytherapy ± EBRT; chemotherapy for high-grade |
| II | Cervical stromal invasion | EBRT + vaginal cuff brachytherapy ± chemotherapy |
| IIIA | Uterine serosa or adnexal involvement | Chemotherapy + radiation (sandwich or sequential) |
| IIIB | Vaginal or parametrial involvement | Chemotherapy + radiation |
| IIIC | Pelvic (IIIC1) or para-aortic (IIIC2) lymph node metastasis | Chemotherapy + extended-field radiation |
| IVA | Bladder/bowel mucosal invasion | Chemotherapy + individualized radiation |
| IVB | Distant metastasis | Systemic chemotherapy (carboplatin/paclitaxel) ± pembrolizumab (dMMR/MSI-H) or dostarlimab |
Landmark trial: GOG 258 showed chemotherapy + radiation did not improve recurrence-free survival over chemotherapy alone in stage III-IVA. PORTEC-3 showed benefit of adding chemotherapy to radiation in high-risk (stage III, serous/clear cell) disease. RUBY trial: Dostarlimab + carboplatin/paclitaxel significantly improved PFS in advanced/recurrent endometrial cancer, especially dMMR/MSI-H.
15 Ovarian Cancer
Epithelial Ovarian Cancer — Histologic Subtypes
Ovarian cancer is the most lethal gynecologic malignancy due to late-stage presentation (~70% diagnosed at stage III-IV). Epithelial ovarian cancer accounts for ~90% of cases.
High-grade serous carcinoma (HGSC, ~70%): Most common and most lethal. Associated with TP53 mutations (nearly universal), BRCA1/2 mutations (15-20%), homologous recombination deficiency (HRD ~50%). Most arise from the fallopian tube fimbria (serous tubal intraepithelial carcinoma, STIC). Responds well to platinum-based chemotherapy and PARP inhibitors.
Low-grade serous carcinoma (LGSC, ~5%): Slower growing, associated with KRAS/BRAF mutations, less responsive to chemotherapy. MEK inhibitors (trametinib) are active in recurrent disease.
Endometrioid (~10%): Associated with endometriosis. May co-occur with synchronous endometrial cancer. ARID1A, PIK3CA, PTEN mutations.
Clear cell (~5%): Associated with endometriosis. Platinum-resistant. Worse prognosis stage-for-stage. PI3K/AKT pathway mutations.
Mucinous (~3%): Often diagnosed at early stage. Must rule out GI primary (appendiceal, colorectal) with immunohistochemistry (CK7+/CK20-, PAX8+). KRAS mutations common.
FIGO Staging & Surgical Management
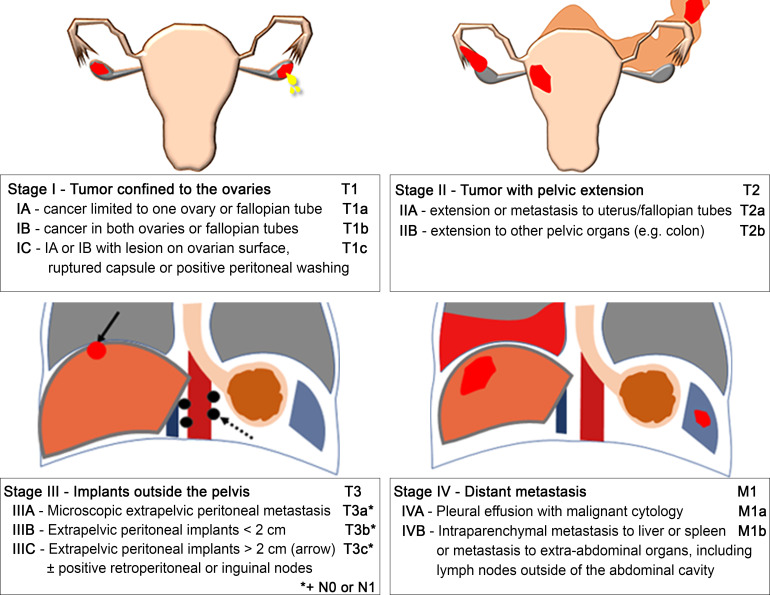
Ovarian cancer is surgically staged. The goal of primary surgery is optimal cytoreduction (debulking) — ideally R0 resection (no visible residual disease). Procedures may include: TH/BSO, omentectomy, pelvic and para-aortic lymphadenectomy, peritoneal biopsies, appendectomy (mucinous tumors), diaphragm stripping, bowel resection, splenectomy as needed. Patients who are poor surgical candidates undergo neoadjuvant chemotherapy (NACT) with 3-4 cycles of carboplatin/paclitaxel followed by interval debulking surgery (IDS) (CHORUS and LION trials).
Systemic Therapy
First-line: Carboplatin (AUC 5-6) + paclitaxel (175 mg/m²) every 3 weeks × 6 cycles. Addition of bevacizumab in advanced disease (GOG 218, ICON7). PARP inhibitors (olaparib, niraparib, rucaparib) as maintenance therapy after first-line platinum response — greatest benefit in BRCA-mutated and HRD-positive tumors (SOLO-1: 5-year PFS 48% vs 21% with olaparib in BRCA-mutated). Olaparib + bevacizumab maintenance for HRD-positive (PAOLA-1). CA-125 monitoring: Used for treatment response assessment and surveillance. Normal < 35 U/mL. Rising CA-125 suggests recurrence but routine monitoring in asymptomatic patients does not improve overall survival (OV05/EORTC 55955 trial).
BRCA and Genetic Testing
All patients with epithelial ovarian cancer should undergo germline and somatic BRCA1/2 testing and ideally comprehensive genomic profiling including HRD assessment. Germline BRCA1/2 mutations are found in 15-20% of ovarian cancer patients. Risk-reducing bilateral salpingo-oophorectomy (RRBSO) is recommended by age 35-40 for BRCA1 carriers and by 40-45 for BRCA2 carriers after completion of childbearing (reduces ovarian cancer risk by ~80%). Cascade genetic testing of family members is essential.
Non-Epithelial Ovarian Tumors
Germ cell tumors (~5%): Affect young women (10-30 years). Types: dysgerminoma (most common malignant germ cell tumor, elevated LDH, excellent prognosis, highly radiosensitive and chemosensitive), immature teratoma (graded I-III, AFP may be elevated in grade III), yolk sac tumor (endodermal sinus tumor) (elevated AFP, Schiller-Duval bodies on histology), embryonal carcinoma (elevated AFP and hCG), choriocarcinoma (elevated hCG). Treatment: fertility-sparing surgery (unilateral salpingo-oophorectomy + staging) followed by BEP chemotherapy (bleomycin, etoposide, cisplatin) for all stages except stage IA dysgerminoma and stage IA grade 1 immature teratoma.
Sex cord-stromal tumors (~5%): Granulosa cell tumor (most common — adult type produces estrogen, Call-Exner bodies on histology, elevated inhibin B; may cause precocious puberty in children or endometrial hyperplasia/cancer in adults; indolent but late recurrences possible). Sertoli-Leydig cell tumor (produces androgens → virilization; inhibin elevated). Fibroma (benign solid tumor; associated with Meigs syndrome: ovarian fibroma + ascites + right pleural effusion — resolves after tumor removal). Treatment: surgery; chemotherapy (BEP or carboplatin/paclitaxel) for advanced or recurrent disease.
16 Vulvar & Vaginal Cancer
Vulvar Cancer
Vulvar cancer accounts for ~5% of gynecologic malignancies. Two pathways: (1) HPV-related (younger patients, associated with VIN/HSIL, warty/basaloid histology, better prognosis) and (2) HPV-independent (older patients, associated with lichen sclerosus and differentiated VIN, keratinizing SCC, TP53 mutations, worse prognosis). Squamous cell carcinoma accounts for ~90% of vulvar cancers.
Staging (FIGO 2021): Stage I — confined to vulva/perineum (IA: ≤ 2 cm with stromal invasion ≤ 1 mm; IB: > 2 cm or invasion > 1 mm). Stage II — adjacent perineal structures (lower urethra, lower vagina, anus). Stage III — inguinofemoral lymph node metastasis. Stage IV — upper urethral, bladder/rectal mucosa (IVA), distant metastasis (IVB).
Treatment: IA — wide local excision (WLE), no lymph node assessment needed (risk of LN mets < 1%). IB — radical local excision with ≥ 1 cm margins + ipsilateral or bilateral inguinofemoral sentinel lymph node biopsy (GROningen INternational Study on Sentinel nodes in Vulvar cancer — GROINSS-V). Positive nodes → inguinofemoral lymphadenectomy or radiation. Advanced disease → chemoradiation (cisplatin-based) ± surgery.
Vulvar Dermatoses
Lichen sclerosus: Chronic inflammatory dermatosis causing white, thin, atrophic skin ("cigarette paper" or "parchment" appearance). Intense pruritus. Affects vulva and perianal area ("figure-of-eight" pattern). Biopsy to confirm and rule out VIN/cancer. Treatment: ultra-potent topical corticosteroid (clobetasol 0.05%). Long-term follow-up required — 4-5% lifetime risk of vulvar SCC. Lichen planus: Erosive variant causes painful vulvovaginal erosions, vaginal adhesions, and stenosis. Wickham striae (fine white lines). Treatment: topical corticosteroids, tacrolimus. May co-exist with oral and vaginal disease. Lichen simplex chronicus: Thickened, lichenified skin from chronic scratching. Break the itch-scratch cycle with topical steroids and antihistamines.
Vaginal Cancer
Vaginal cancer is rare (~1-2% of gynecologic malignancies). Most are SCC (~80%), often HPV-related. Must rule out extension from cervix, vulva, or other primary sites — by convention, a tumor involving both the cervix and vagina is classified as cervical cancer, and a tumor involving the vulva and vagina is classified as vulvar cancer. Clear cell adenocarcinoma of the vagina is associated with in utero DES exposure (DES daughters — risk highest age 15-25). Other vaginal lesions: vaginal melanoma (rare, poor prognosis), sarcoma botryoides (embryonal rhabdomyosarcoma — children, "grape-like" mass protruding from vagina). FIGO staging mirrors cervical cancer staging principles. Treatment is primarily radiation (EBRT + brachytherapy); surgery is limited to small early-stage lesions in the upper vagina (upper vaginectomy ± radical hysterectomy for stage I).
17 Gestational Trophoblastic Disease
Hydatidiform Mole
Complete mole: 46,XX or 46,XY (entirely paternal origin — empty ovum fertilized by one sperm that duplicates, or two sperm). No fetal tissue. "Snowstorm" pattern on ultrasound. Markedly elevated β-hCG (> 100,000 mIU/mL common). Bilateral theca lutein cysts (ovarian hyperstimulation from high hCG). Risk of GTN: 15-20%.
Partial mole: 69,XXX or 69,XXY (triploid — normal ovum + two sperm). Fetal tissue present (usually abnormal/nonviable). Moderately elevated hCG. "Swiss cheese" placenta on US. Risk of GTN: 1-5%.
Management: Suction curettage (uterine evacuation). RhoGAM if Rh-negative. Follow serial β-hCG weekly until undetectable for 3 consecutive weeks, then monthly for 6 months (complete) or 6 months (partial). Reliable contraception during monitoring (hCG monitoring would be confounded by pregnancy). Rise or plateau of hCG during surveillance suggests GTN.
Gestational Trophoblastic Neoplasia (GTN)
Includes invasive mole, choriocarcinoma, placental site trophoblastic tumor (PSTT), and epithelioid trophoblastic tumor (ETT). Diagnosed by: rising hCG (≥ 10% rise over 3 values over 2 weeks), plateau of hCG (4 values over 3 weeks), hCG still detectable at 6 months post-evacuation, or histologic diagnosis of choriocarcinoma.
FIGO/WHO Scoring System: Uses age, antecedent pregnancy, interval from index pregnancy, pre-treatment hCG, largest tumor size, site/number of metastases, prior failed chemotherapy. Score ≤ 6 = low risk; ≥ 7 = high risk.
Treatment: Low-risk GTN — single-agent methotrexate (8-day protocol with leucovorin rescue, or weekly IM) or actinomycin D. Cure rate > 90%. High-risk GTN — multi-agent chemotherapy: EMA-CO (etoposide, methotrexate, actinomycin D, cyclophosphamide, vincristine). Cure rate 80-90%. Ultra-high-risk (score ≥ 13 or liver/brain metastases) — induction low-dose EP (etoposide/cisplatin) before EMA-CO to avoid hemorrhage. hCG is followed to undetectable, then 3 additional cycles of consolidation chemotherapy. Fertility preservation is possible in most cases — GTN is one of the most curable malignancies in oncology.
FIGO/WHO GTN Scoring Table
| Variable | Score 0 | Score 1 | Score 2 | Score 4 |
|---|---|---|---|---|
| Age (years) | < 40 | ≥ 40 | — | — |
| Antecedent pregnancy | Mole | Abortion | Term | — |
| Interval from index (months) | < 4 | 4-6 | 7-12 | > 12 |
| Pre-treatment hCG (mIU/mL) | < 10³ | 10³-10&sup4; | 10&sup4;-10&sup5; | > 10&sup5; |
| Largest tumor size (cm) | < 3 | 3-4 | ≥ 5 | — |
| Site of metastases | Lung | Spleen, kidney | GI | Brain, liver |
| Number of metastases | 0 | 1-4 | 5-8 | > 8 |
| Prior failed chemotherapy | — | — | Single agent | ≥ 2 agents |
Score ≤ 6: Low-risk — single-agent chemotherapy. Score ≥ 7: High-risk — multi-agent chemotherapy (EMA-CO).
18 Uterine Fibroids
Overview & Classification
Uterine leiomyomas (fibroids) are the most common pelvic tumors in women, affecting up to 70-80% of women by age 50. They are benign smooth muscle tumors of the myometrium, estrogen- and progesterone-dependent (grow during reproductive years, shrink after menopause). Risk factors: African American race (3× higher prevalence, earlier onset, more symptomatic), early menarche, nulliparity, obesity, family history.
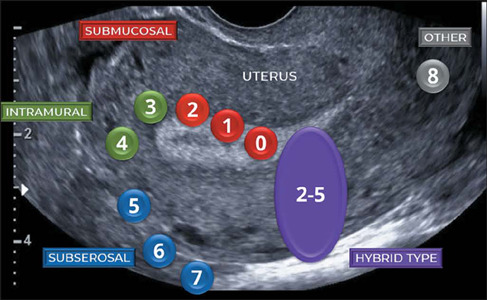
Type 0: Pedunculated intracavitary (entirely within the cavity). Type 1: < 50% intramural. Type 2: ≥ 50% intramural (submucosal types 0-2 are most associated with abnormal bleeding and infertility). Type 3: Contacts endometrium, 100% intramural. Type 4: Intramural (entirely within the myometrium). Type 5: Subserosal, ≥ 50% intramural. Type 6: Subserosal, < 50% intramural. Type 7: Subserosal, pedunculated. Type 8: Other (cervical, parasitic, broad ligament).
Symptoms
Heavy menstrual bleeding (HMB) — the most common symptom, especially with submucosal fibroids. Bulk symptoms: Pelvic pressure, urinary frequency/urgency (anterior fibroids compressing bladder), constipation (posterior fibroids), hydronephrosis (rare, large fibroids compressing ureters). Reproductive: Infertility (submucosal fibroids distort the cavity and impair implantation), recurrent pregnancy loss, preterm labor, malpresentation.
Medical Management
Hormonal contraceptives (OCPs, levonorgestrel IUD — Mirena) reduce bleeding but do not shrink fibroids. GnRH agonists (leuprolide): induce a hypoestrogenic state, shrink fibroids ~50% in 3-6 months. Used as preoperative adjunct to reduce fibroid volume and correct anemia. Limited to 6 months due to bone loss. Add-back therapy (low-dose estrogen/progestin) mitigates vasomotor symptoms and bone loss. GnRH antagonist (elagolix with add-back, relugolix combination) — oral agents, rapid onset, FDA-approved for fibroid-associated HMB. Tranexamic acid (antifibrinolytic) reduces menstrual blood loss by ~40%. Iron supplementation for anemia.
Surgical & Interventional Management
Myomectomy: Removal of fibroids preserving the uterus. Hysteroscopic myomectomy for submucosal types 0-2. Laparoscopic/robotic myomectomy for intramural/subserosal fibroids. Open (abdominal) myomectomy for very large or numerous fibroids. Risk of recurrence: 15-30% over 5 years. Morcellation controversy: power morcellation may disseminate occult leiomyosarcoma (FDA black box warning 2014). Contained morcellation with specimen bags is an alternative.
Hysterectomy: Definitive treatment. Approach depends on uterine size, pathology, and surgeon expertise: vaginal (preferred for benign disease when feasible), laparoscopic/robotic (standard minimally invasive approach), abdominal (reserved for very large uteri, suspected malignancy, extensive adhesions).
Uterine artery embolization (UAE): Interventional radiology procedure. Polyvinyl alcohol particles or microspheres are injected into both uterine arteries to infarct the fibroids. Reduces fibroid volume ~50% and improves symptoms in 85-90%. Not recommended for women desiring future fertility (theoretical concerns about uterine blood supply). Complications: postembolization syndrome (pain, fever, nausea), fibroid expulsion, amenorrhea (1-5% if > age 45).
19 Endometriosis
Pathogenesis & Clinical Features
Endometriosis is defined as the presence of endometrial-like tissue (glands and stroma) outside the uterine cavity. Affects ~10% of reproductive-age women. The leading theory is retrograde menstruation (Sampson theory) — reflux of menstrual tissue through the fallopian tubes into the peritoneal cavity, with subsequent implantation and growth. Other theories: coelomic metaplasia (transformation of peritoneal mesothelium), lymphatic/vascular dissemination (explains distant sites like lung, brain), stem cell theory, and immune dysfunction (failure to clear ectopic implants).
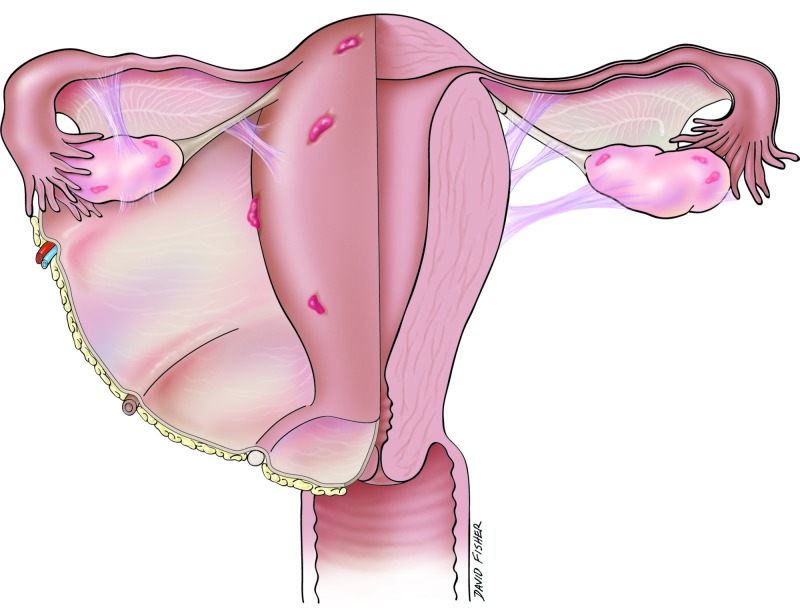
Symptoms: Dysmenorrhea (progressive, often severe), dyspareunia (especially deep dyspareunia), chronic pelvic pain, dyschezia (painful bowel movements, especially catamenial), infertility (30-50% of infertile women have endometriosis), and catamenial symptoms at distant sites (hemoptysis, seizures). Endometriosis is a chronic, estrogen-dependent inflammatory condition with significant impact on quality of life. Average diagnostic delay from symptom onset is 7-10 years. Physical exam may reveal uterosacral nodularity, fixed retroverted uterus, or adnexal mass (endometrioma). Laparoscopy with histologic confirmation remains the gold standard for definitive diagnosis, though empiric medical therapy is increasingly recommended without surgical confirmation.
rASRM Staging (Revised American Society for Reproductive Medicine)
Surgical staging system based on implant size, depth, location, and adhesion severity. Stage I (minimal): 1-5 points. Stage II (mild): 6-15 points. Stage III (moderate): 16-40 points (endometriomas, more adhesions). Stage IV (severe): > 40 points (large endometriomas, dense adhesions, cul-de-sac obliteration). Importantly, staging correlates poorly with pain severity — a patient with stage I disease can have severe pain, while stage IV may be incidentally discovered.
Medical Management
NSAIDs for pain relief. Combined oral contraceptives (continuous use preferred over cyclic to suppress menstruation). Progestins: Norethindrone acetate 5 mg daily, medroxyprogesterone acetate, depot medroxyprogesterone, levonorgestrel IUD, etonogestrel implant. Dienogest (2 mg daily) — a progestin with specific efficacy for endometriosis, FDA-approved in many countries. GnRH agonists (leuprolide with add-back therapy) for refractory cases. GnRH antagonists (elagolix — Orilissa, relugolix combination) — oral, dose-dependent suppression. Aromatase inhibitors (letrozole) — third-line for refractory disease (off-label).
Surgical Management
Laparoscopic excision (preferred) or ablation of endometriotic implants. Excision is superior to ablation for deep infiltrating endometriosis (DIE). Deep infiltrating endometriosis involves invasion > 5 mm beneath the peritoneal surface and commonly affects the rectovaginal septum, uterosacral ligaments, bladder, and rectosigmoid colon. Requires multidisciplinary surgical planning (colorectal surgery, urology if bowel or bladder involvement). Endometrioma excision (stripping the cyst wall) is preferred over drainage/ablation for recurrence reduction and histologic diagnosis, but repeated surgery can diminish ovarian reserve. Hysterectomy with bilateral salpingo-oophorectomy is reserved for severe refractory disease when fertility is no longer desired.
20 Abnormal Uterine Bleeding
PALM-COEIN Classification
The FIGO PALM-COEIN system classifies causes of abnormal uterine bleeding (AUB) in non-pregnant reproductive-age women:
Structural causes (PALM): Polyp — endometrial or endocervical. Adenomyosis — endometrial glands within the myometrium. Leiomyoma — submucosal (L-SM) or other (L-O). Malignancy and hyperplasia.
Non-structural causes (COEIN): Coagulopathy (von Willebrand disease, thrombocytopenia, anticoagulant use). Ovulatory dysfunction (PCOS, hypothalamic amenorrhea, thyroid disorders). Endometrial (primary endometrial disorder of hemostasis or inflammation). Iatrogenic (hormonal contraceptives, anticoagulants, IUDs). Not yet classified.
Workup
History and physical examination including speculum and bimanual exam. Pregnancy test (always rule out in reproductive-age women). CBC (assess anemia). TSH, prolactin (endocrine causes). Coagulation studies (if suspected coagulopathy, especially in adolescents with heavy menses at menarche — screen for von Willebrand disease). Pelvic ultrasound (transvaginal preferred — evaluate myometrium, endometrial thickness, ovaries). Saline infusion sonohysterography (SIS) for intracavitary lesions. Endometrial biopsy (EMB) — indicated in all women ≥ 45 with AUB, and in women < 45 with risk factors for endometrial hyperplasia/cancer (obesity, PCOS, chronic anovulation, tamoxifen, failed medical therapy). Hysteroscopy with directed biopsy for definitive evaluation.
Management by Cause
Ovulatory dysfunction: Hormonal regulation with combined OCPs, cyclic progestins (medroxyprogesterone 10 mg × 10-14 days/month), levonorgestrel IUD (most effective for HMB with AUB-O). Polyps: Hysteroscopic polypectomy. Fibroids: See Section 18. Adenomyosis: Levonorgestrel IUD (first-line), GnRH agonists/antagonists, definitive treatment is hysterectomy. Hyperplasia without atypia: Progestin therapy (levonorgestrel IUD preferred, or oral progestins). Atypical hyperplasia: Hysterectomy (standard); progestin therapy with close surveillance (levonorgestrel IUD + EMB every 3-6 months) if fertility preservation is desired.
Polycystic Ovary Syndrome (PCOS)
PCOS is the most common endocrine disorder in reproductive-age women (~6-12%). Diagnosis by Rotterdam criteria (2 of 3): oligo/anovulation, clinical or biochemical hyperandrogenism (hirsutism, acne, elevated total/free testosterone), polycystic ovarian morphology on US (≥ 12 follicles per ovary or ovarian volume > 10 mL) — after exclusion of other causes (thyroid disease, congenital adrenal hyperplasia, Cushing syndrome, hyperprolactinemia).
Metabolic implications: Insulin resistance (present in 50-70%), metabolic syndrome, increased risk of type 2 diabetes, dyslipidemia, NAFLD, obstructive sleep apnea, cardiovascular disease. Reproductive implications: Anovulatory infertility (most common cause), increased risk of endometrial hyperplasia/cancer (chronic unopposed estrogen), recurrent pregnancy loss, GDM, preeclampsia.
Management: Lifestyle modification (weight loss of 5-10% can restore ovulation). OCPs for menstrual regulation and hyperandrogenism. Spironolactone (anti-androgen) for hirsutism/acne. Metformin for insulin resistance and metabolic features. For infertility: letrozole (aromatase inhibitor) is first-line for ovulation induction in PCOS (NICHD trial: superior live birth rate vs clomiphene). Clomiphene citrate is second-line. Gonadotropins and laparoscopic ovarian drilling are third-line. IVF for refractory cases.
21 Ovarian Cysts & Adnexal Masses
Functional Cysts
Follicular cysts arise from unruptured dominant follicles, typically < 8 cm, thin-walled, unilocular, simple on US. Usually resolve spontaneously in 1-3 cycles. Corpus luteum cysts form from the hemorrhagic corpus luteum, may contain internal echoes (hemorrhage), thick wall with peripheral "ring of fire" vascularity on Doppler. Can cause pain if they rupture or hemorrhage. Management: observation and repeat US in 6-8 weeks. OCPs may prevent future functional cysts but do not hasten resolution of existing cysts.
Pathologic Ovarian Masses
Mature cystic teratoma (dermoid): Most common benign ovarian neoplasm in reproductive-age women. Contains tissue from all three germ layers (hair, teeth, sebaceous material). Characteristic US appearance: echogenic "tip of the iceberg" sign, fat-fluid level, Rokitansky nodule. Bilateral in 10-15%. Rarely undergoes malignant transformation (~1%, usually SCC, in postmenopausal women). Treatment: cystectomy (fertility-sparing) or oophorectomy.
Endometrioma ("chocolate cyst"): Endometriotic cyst of the ovary. Homogeneous low-level internal echoes ("ground glass") on US. Treatment: excision (stripping) for > 4 cm or symptomatic. Consider ovarian reserve impact (AMH) before surgery.
Cystadenoma: Serous (thin-walled, unilocular, clear fluid) or mucinous (multiloculated, thick mucinous fluid, can become very large). Benign but may require excision for size or symptoms.
IOTA Rules for Risk Stratification
The IOTA (International Ovarian Tumor Analysis) Simple Rules classify adnexal masses as benign, malignant, or indeterminate using five benign features (B-rules: unilocular, solid component < 7 mm, acoustic shadows, smooth multilocular < 100 mm, no blood flow) and five malignant features (M-rules: irregular solid tumor, ascites, ≥ 4 papillary projections, irregular multilocular-solid > 100 mm, very strong blood flow). If only B-rules apply → benign; only M-rules → malignant; both or neither → indeterminate (requires expert US or MRI).
Ovarian Torsion
Torsion of the ovary (and often the fallopian tube — adnexal torsion) around the IP ligament, compromising vascular supply. Risk factors: ovarian mass (dermoid most common, any mass > 5 cm), ovarian hyperstimulation, pregnancy, previous torsion. Presentation: Acute-onset severe unilateral lower abdominal/pelvic pain, nausea/vomiting (90%), low-grade fever. More common on the right side. Diagnosis: Clinical suspicion + pelvic US with Doppler. Absent or decreased ovarian arterial flow is suggestive but normal Doppler does not rule out torsion (dual blood supply). Enlarged ovary with peripheral follicles ("string of pearls") and free fluid are supportive. Treatment: Emergent laparoscopy with detorsion (untwist). The ovary should be preserved if viable (even if appears dusky — most recover). Oophorectomy only if clearly necrotic or if postmenopausal. Oophoropexy may be performed to prevent recurrence.
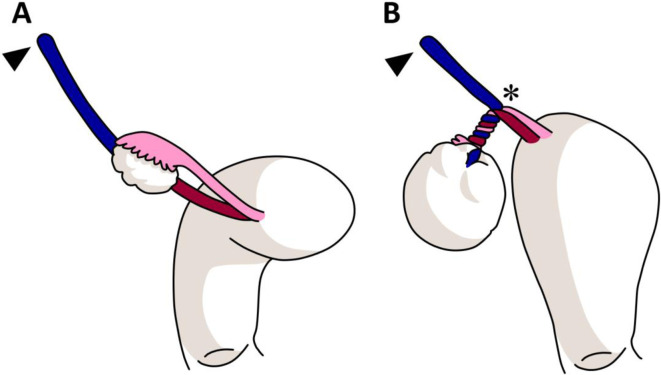
22 Pelvic Inflammatory Disease & STIs
Pelvic Inflammatory Disease (PID)
PID is an ascending infection of the upper genital tract (endometritis, salpingitis, tubo-ovarian abscess, peritonitis). Most commonly caused by Neisseria gonorrhoeae and Chlamydia trachomatis, but often polymicrobial (anaerobes, GNR, Mycoplasma genitalium). Risk factors: age 15-25, multiple sexual partners, prior STI/PID, IUD insertion within prior 3 weeks (risk window only).
Diagnosis (CDC minimal criteria): Uterine tenderness, adnexal tenderness, or cervical motion tenderness in a sexually active young woman with no other identifiable cause. Additional criteria supporting diagnosis: oral temperature > 38.3°C, mucopurulent cervical discharge, elevated ESR/CRP, laboratory confirmation of cervical GC/CT infection.
Outpatient (mild-moderate PID): Ceftriaxone 500 mg IM × 1 dose PLUS doxycycline 100 mg PO BID × 14 days PLUS metronidazole 500 mg PO BID × 14 days.
Inpatient (any of: surgical emergency not excluded, TOA, pregnant, no clinical improvement on oral therapy, unable to tolerate oral regimen, severe illness): Cefotetan 2 g IV Q12H + doxycycline 100 mg PO/IV Q12H; OR Cefoxitin 2 g IV Q6H + doxycycline 100 mg PO/IV Q12H. Transition to oral doxycycline + metronidazole to complete 14 days after 24-48 hours of clinical improvement.
Tubo-Ovarian Abscess (TOA)
A complex, inflammatory mass involving the tube and ovary, often with pus. Diagnosed by pelvic US (complex, multiloculated, thick-walled mass with internal echoes). Management: IV antibiotics (as above). If no improvement in 48-72 hours, US or CT-guided percutaneous drainage or surgical drainage (laparoscopy or laparotomy). Ruptured TOA is a surgical emergency — peritonitis, sepsis, and potential death if not treated surgically.
Fitz-Hugh-Curtis Syndrome
Perihepatitis — inflammation of the liver capsule (Glisson capsule) and adjacent peritoneum, causing right upper quadrant pain. Associated with PID (GC or CT ascending infection). "Violin string" adhesions between the anterior liver surface and the anterior abdominal wall seen at laparoscopy. Can mimic cholecystitis, hepatitis, or PE. Treatment is the same as PID.
Common STIs in Gynecologic Practice
Chlamydia (C. trachomatis): Most common bacterial STI. Often asymptomatic. Screen annually all sexually active women < 25 and those with risk factors. Treat: doxycycline 100 mg PO BID × 7 days (preferred) or azithromycin 1 g PO × 1. Gonorrhea (N. gonorrhoeae): Ceftriaxone 500 mg IM × 1 (if < 150 kg) — always co-treat for chlamydia if not tested. Trichomonas vaginalis: Metronidazole 500 mg PO BID × 7 days (women) or 2 g PO × 1 (men). Herpes simplex virus (HSV): Acyclovir 400 mg TID × 7-10 days (first episode); valacyclovir 500 mg daily for suppression. Syphilis (T. pallidum): Benzathine penicillin G 2.4 million units IM × 1 (primary, secondary, early latent); 3 weekly doses for late latent or unknown duration. In pregnancy, penicillin is the ONLY adequate treatment — desensitize penicillin-allergic patients. Congenital syphilis is preventable with adequate maternal treatment.
Vaginitis — Differential Diagnosis
| Condition | Discharge | pH | Diagnosis | Treatment |
|---|---|---|---|---|
| Bacterial vaginosis | Thin, gray-white, fishy odor | > 4.5 | Clue cells; Amsel criteria (3 of 4): homogeneous discharge, pH > 4.5, positive whiff test, clue cells | Metronidazole 500 mg PO BID × 7 d |
| Vulvovaginal candidiasis | Thick, white, cottage cheese | < 4.5 | Pseudohyphae on KOH prep | Fluconazole 150 mg PO × 1 |
| Trichomoniasis | Green-yellow, frothy | > 4.5 | Motile trichomonads; strawberry cervix | Metronidazole 500 mg PO BID × 7 d |
| Atrophic vaginitis | Thin, watery, blood-tinged | > 4.5 | Parabasal cells; pale, thin, friable mucosa | Vaginal estrogen |
23 Menopause & Hormone Replacement Therapy
Definitions & Physiology
Menopause is defined retrospectively as 12 consecutive months of amenorrhea in a woman ≥ 45 years (without other pathologic cause), reflecting permanent cessation of ovarian follicular activity. Mean age in the US: 51 years. Perimenopause (menopausal transition) begins years before menopause with fluctuating cycles, hormonal variability, and vasomotor symptoms. Characterized by rising FSH, declining inhibin B, and variable estradiol levels. Premature ovarian insufficiency (POI): Menopause before age 40 — warrants evaluation for karyotype (Turner syndrome mosaicism), FMR1 premutation, autoimmune causes, and iatrogenic factors.
Symptoms
Vasomotor symptoms (VMS): Hot flashes and night sweats, experienced by ~75% of women. Median duration 7.4 years. Most disruptive symptom. Genitourinary syndrome of menopause (GSM): Vaginal dryness, dyspareunia, urinary urgency, recurrent UTIs — due to estrogen-dependent atrophy of vaginal and urethral epithelium. Unlike VMS, GSM is progressive and does not resolve without treatment. Other: Sleep disturbance, mood changes, cognitive complaints, joint pain, decreased libido.
Menopausal Hormone Therapy (MHT)
Systemic estrogen is the most effective treatment for vasomotor symptoms. Women with an intact uterus require a progestogen (to protect the endometrium from estrogen-stimulated hyperplasia/cancer). Women without a uterus (post-hysterectomy) use estrogen alone.
Estrogen formulations: Conjugated equine estrogen (CEE 0.3-0.625 mg/day), estradiol (oral 0.5-2 mg/day, transdermal patch 0.025-0.1 mg/day, gel, spray). Transdermal estradiol is preferred for women with VTE risk factors, hypertriglyceridemia, or migraine with aura (avoids first-pass hepatic effect, lower VTE risk).
Progestogen options: Medroxyprogesterone acetate (MPA, 2.5-5 mg daily or 5-10 mg × 12-14 days/month), micronized progesterone (100-200 mg daily or cyclically — preferred for metabolic profile and breast risk), levonorgestrel IUD, norethindrone acetate.
Combined preparations: Estradiol/norethindrone, estradiol/drospirenone, CEE/bazedoxifene (TSEC — tissue-selective estrogen complex, no progestogen needed).
Local estrogen (for GSM only): Vaginal estrogen cream, tablet (Vagifem), ring (Estring), or DHEA insert (Intrarosa/prasterone). Minimal systemic absorption at standard doses; progestogen not generally required.
WHI Trial Implications
The Women's Health Initiative (WHI) trials fundamentally shaped MHT prescribing. Key findings: CEE + MPA arm (women with uterus): increased breast cancer, coronary events, stroke, VTE; decreased hip fractures and colorectal cancer. CEE-alone arm (women without uterus): no increased breast cancer risk (actually trended toward decrease), increased stroke and VTE, decreased hip fractures. The "timing hypothesis" emerged from subgroup analyses: women who initiated MHT within 10 years of menopause or before age 60 had a favorable cardiovascular risk profile, while those starting > 10 years post-menopause had increased risk. Current guidelines: MHT is appropriate for symptomatic women < 60 or within 10 years of menopause, at the lowest effective dose for the shortest duration needed. Absolute contraindications: unexplained vaginal bleeding, active breast cancer, active VTE/PE, active liver disease, known CHD/stroke.
Non-Hormonal Alternatives for VMS
Fezolinetant (Veozah) — oral neurokinin 3 (NK3) receptor antagonist, FDA-approved 2023 for moderate-severe VMS. SSRIs/SNRIs: Paroxetine 7.5 mg (Brisdelle — only FDA-approved SSRI for VMS), venlafaxine 37.5-75 mg, escitalopram. Gabapentin 300-900 mg at bedtime. Clonidine 0.1 mg patch weekly. Cognitive behavioral therapy (CBT) and clinical hypnosis have demonstrated efficacy. Oxybutynin 2.5 mg BID (anticholinergic) has shown VMS reduction.
Contraception Overview
Contraceptive counseling is a core component of gynecologic care. Methods ranked by effectiveness (typical use failure rate):
| Method | Typical Use Failure Rate (1 year) | Key Notes |
|---|---|---|
| Etonogestrel implant (Nexplanon) | 0.05% | Most effective reversible contraceptive; 3-year duration; irregular bleeding common |
| Levonorgestrel IUD (Mirena, Liletta) | 0.2% | 5-8 years; reduces menstrual bleeding; treats AUB, endometriosis, hyperplasia |
| Copper IUD (Paragard) | 0.8% | 10-12 years; non-hormonal; may increase menstrual bleeding/cramping |
| Depot medroxyprogesterone (Depo-Provera) | 6% | Every 3 months IM; bone density concerns with > 2 years of use; delayed return to fertility |
| Combined oral contraceptives | 9% | Daily pill; CI in smokers ≥ 35, migraine with aura, VTE history, active breast cancer |
| Patch / Vaginal ring | 9% | Weekly patch (Xulane) or monthly ring (NuvaRing); same CIs as COCs |
| Progestin-only pill (minipill) | 9% | Must take at same time daily (± 3 hours); safe in breastfeeding, HTN, migraine with aura |
| Male condom | 18% | Only method (with female condom) that also prevents STIs |
| Tubal ligation / salpingectomy | 0.5% | Permanent; salpingectomy preferred (reduces ovarian cancer risk) |
Emergency contraception: Copper IUD (most effective, up to 5 days post-coitus), ulipristal acetate (ella, up to 5 days, progesterone receptor modulator), levonorgestrel (Plan B, up to 3 days, less effective with BMI > 25). The US Medical Eligibility Criteria for Contraceptive Use (US MEC) guides safe prescribing based on patient comorbidities.
24 Pelvic Organ Prolapse
Types & Pathophysiology
Pelvic organ prolapse (POP) is the descent of pelvic organs through the vagina due to loss of support. Affects up to 50% of parous women to some degree. Types: cystocele (anterior vaginal wall prolapse, most common), rectocele (posterior vaginal wall), uterine prolapse (descent of the uterus/cervix), vaginal vault prolapse (post-hysterectomy, descent of the vaginal cuff), enterocele (herniation of small bowel into the rectovaginal space). Risk factors: vaginal delivery (strongest, especially forceps/prolonged second stage), advanced age, menopause (estrogen deficiency), chronic straining (constipation, heavy lifting, chronic cough), obesity, connective tissue disorders, prior pelvic surgery.
POP-Q Staging System
The Pelvic Organ Prolapse Quantification (POP-Q) system is the standardized, site-specific measurement system. Six points (Aa, Ba, C, D, Ap, Bp) are measured relative to the hymen (positive = beyond hymen, negative = above hymen). Additional measurements: genital hiatus (gh), perineal body (pb), total vaginal length (TVL).
| Stage | Description |
|---|---|
| Stage 0 | No prolapse |
| Stage I | Most distal portion > 1 cm above the hymen |
| Stage II | Most distal portion within 1 cm above or below the hymen |
| Stage III | Most distal portion > 1 cm below the hymen but not complete eversion |
| Stage IV | Complete eversion (procidentia) |
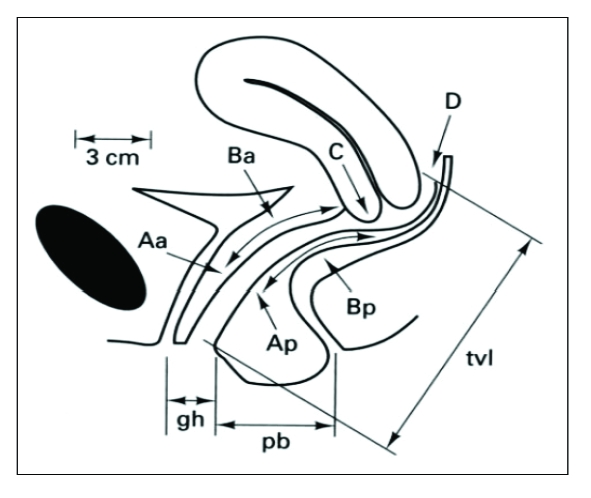
Symptoms & Evaluation
Many women with POP are asymptomatic — treatment is based on symptoms, not stage alone. Symptoms: Vaginal bulge sensation ("something falling out" — most specific symptom for prolapse), pelvic pressure/heaviness, difficulty with defecation (posterior prolapse — may require digital splinting/digitation), voiding dysfunction (incomplete emptying, hesitancy, positional voiding with anterior prolapse), sexual dysfunction, and low back pain. Symptoms typically worsen with prolonged standing and activity and improve with lying down.
Evaluation: POP-Q examination at rest and with maximum Valsalva maneuver, assessment of each compartment (anterior, apical, posterior independently), and evaluation for concomitant stress urinary incontinence. Occult SUI may be unmasked after prolapse reduction (the prolapsed tissue kinks the urethra, masking SUI) — perform a prolapse-reduced cough stress test before surgical planning to determine if an anti-incontinence procedure should be performed concurrently.
Management
Conservative: Pelvic floor muscle training (PFMT/Kegel exercises) — first-line for mild POP and urinary incontinence. Pessary fitting — ring pessary (most common initial choice), Gellhorn (for more advanced prolapse), cube, donut. Appropriate for women who decline surgery, have comorbidities precluding surgery, or desire future pregnancy. Require follow-up for cleaning every 3-6 months and monitoring for erosion/ulceration.
Surgical: Abdominal sacrocolpopexy (ASC) — polypropylene mesh suspension of the vaginal apex to the anterior longitudinal ligament of the sacrum. Gold standard for apical prolapse repair, typically performed robotically or laparoscopically. Success rate ~90% at 5 years. Native tissue repair: Uterosacral ligament suspension (USLS), sacrospinous ligament fixation (SSLF) — vaginal approaches. Higher recurrence rates than mesh sacrocolpopexy but avoid mesh-related complications. Anterior/posterior colporrhaphy: Plication of the pubocervical fascia (anterior) or rectovaginal fascia (posterior). Transvaginal mesh controversy: FDA ordered withdrawal of all transvaginal mesh for POP repair in 2019 due to high complication rates (mesh erosion/exposure, pain, dyspareunia). Sacrocolpopexy mesh (abdominal, not transvaginal) remains standard of care. Obliterative surgery (colpocleisis): For elderly women who are not sexually active — high success rate, lower morbidity.
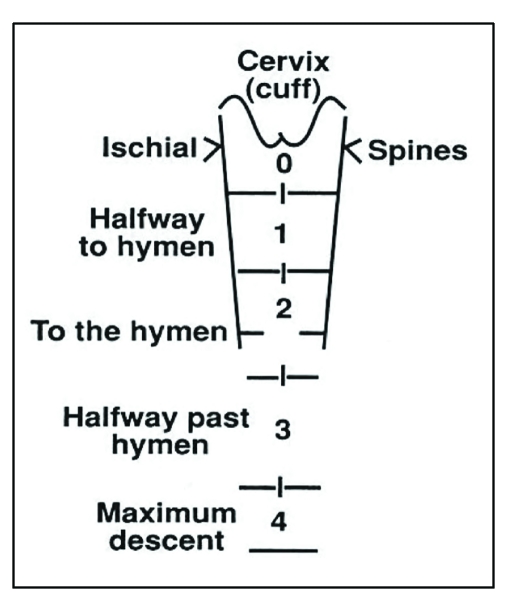
25 Urinary Incontinence
Types
Stress urinary incontinence (SUI): Involuntary urine leakage with increased abdominal pressure (coughing, sneezing, laughing, exercise). Due to urethral hypermobility or intrinsic sphincter deficiency. Most common type in younger women.
Urgency urinary incontinence (UUI): Involuntary leakage accompanied by or immediately preceded by urgency. Due to detrusor overactivity. Part of overactive bladder (OAB) syndrome (urgency, with or without incontinence, usually with frequency and nocturia).
Mixed urinary incontinence: Combination of stress and urgency components. Treat the predominant/most bothersome symptom first.
Evaluation
History: Bladder diary (frequency, volume, leakage episodes, triggers). Physical exam: Cough stress test (observe for leakage with cough at comfortably full bladder), POP-Q assessment, vaginal atrophy assessment. Urinalysis (rule out UTI, hematuria). Post-void residual (PVR) — elevated PVR (> 150-200 mL) suggests overflow incontinence or impaired detrusor contractility. Urodynamic studies (UDS): Multi-channel cystometry, pressure-flow studies, leak point pressure. Indicated before surgical intervention for SUI (especially if mixed symptoms, prior failed surgery, or neurologic conditions). Cystoscopy: If hematuria, suspected fistula, or prior mesh/surgery complications.
Management of SUI
Conservative: PFMT/Kegel exercises (with biofeedback), weight loss if overweight, vaginal estrogen for atrophic urethral tissue, continence pessary. Surgical: Midurethral sling (MUS) — retropubic (TVT) or transobturator (TOT) polypropylene mesh sling placed tension-free under the mid-urethra. Gold standard surgical treatment. Success rate 80-90%. Complications: urinary retention, mesh erosion, pain, bladder perforation (retropubic). Burch colposuspension — laparoscopic or open suturing of paravaginal tissue to Cooper (pectineal) ligament. Alternative to MUS, often performed concurrently with sacrocolpopexy. Urethral bulking agents — injection of bulking material around the urethra. Less invasive, lower success rate (~50-70%), may need repeat injections.
Management of UUI/OAB
First-line: Behavioral modifications — timed voiding, bladder training (progressive voiding intervals), fluid management, caffeine/alcohol reduction. PFMT. Second-line: Antimuscarinics — oxybutynin (IR/ER/patch/gel), tolterodine, solifenacin, darifenacin, fesoterodine, trospium. Side effects: dry mouth, constipation, cognitive impairment (especially in elderly — avoid in patients > 70 with cognitive concerns). Beta-3 agonist — mirabegron (25-50 mg), vibegron (75 mg). Fewer anticholinergic side effects. Can be combined with antimuscarinics. Third-line: OnabotulinumtoxinA (Botox) 100 units injected into the detrusor muscle cystoscopically (lasts 6-9 months, risk of urinary retention). Percutaneous tibial nerve stimulation (PTNS). Sacral neuromodulation (InterStim).
26 Gynecologic Procedures
Hysteroscopy
Hysteroscopy is the direct visualization of the uterine cavity using a hysteroscope inserted through the cervix. Diagnostic hysteroscopy: Office-based, using a small-diameter scope (3-4 mm), saline distension. For evaluation of AUB, polyps, submucosal fibroids, intrauterine adhesions, uterine anomalies, lost IUD. Operative hysteroscopy: OR-based, larger scope with working channel. Uses fluid distension media (normal saline for bipolar energy; glycine or sorbitol for monopolar). Procedures: polypectomy, myomectomy (types 0-2), adhesiolysis (Asherman syndrome), endometrial ablation, septum resection, removal of retained products. Complications: fluid overload (hyponatremia with hypotonic media — monitor deficit, stop at 1000-1500 mL for hypotonic or 2500 mL for isotonic), uterine perforation, air embolism, cervical injury.
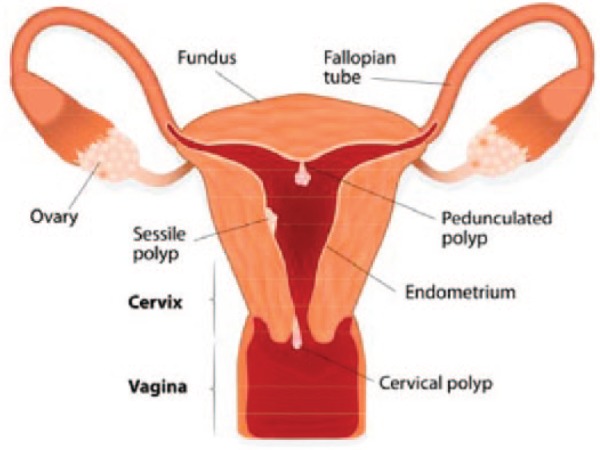
Laparoscopy & Robotic Surgery
Minimally invasive gynecologic surgery (MIGS) is the standard of care for most benign and many malignant gynecologic conditions. Advantages: Smaller incisions, less blood loss, shorter hospital stay, faster recovery, less adhesion formation. Entry techniques: Veress needle (closed entry at Palmer point or umbilicus), Hasson (open entry), direct trocar entry, optical trocar entry. Common procedures: Diagnostic laparoscopy, laparoscopic/robotic hysterectomy, myomectomy, adnexal surgery (cystectomy, oophorectomy, salpingectomy), endometriosis excision, tubal ligation, lymph node dissection (cancer staging).
D&C (Dilation & Curettage)
Suction D&C: Used for first-trimester miscarriage management (missed abortion, incomplete abortion), elective termination, and molar pregnancy evacuation. Sharp curettage: Rarely used alone; may be added after suction to ensure completeness. Complications: uterine perforation, cervical laceration, retained products, Asherman syndrome (intrauterine adhesions from aggressive curettage), infection.
Colposcopy & LEEP
Colposcopy: Examination of the cervix under magnification (6-40×) after application of 3-5% acetic acid. Indications: abnormal Pap smear (per ASCCP guidelines), abnormal cervical appearance, follow-up of CIN. Biopsies are taken of acetowhite lesions and ECC is performed. LEEP (loop electrosurgical excision procedure): Excision of the transformation zone with a thin wire loop under colposcopic guidance. Used for CIN 2/3, persistent CIN 1, and diagnostic excision when ECC is positive. Performed under local anesthesia. Specimen evaluated for margins and invasion. Complications: bleeding, infection, cervical stenosis, cervical insufficiency in future pregnancies (risk increases with depth of excision > 10 mm).
Hysterectomy — Routes & Considerations
Hysterectomy is the most common major gynecologic surgery. Routes of hysterectomy (in order of preference per ACOG):
Vaginal hysterectomy: Preferred when feasible (benign disease, mobile uterus, adequate vaginal access). Shortest recovery, lowest complication rate, most cost-effective. Limited by uterine size, prior surgery/adhesions, need for adnexectomy, and narrow vaginal access.
Laparoscopic/robotic-assisted hysterectomy: Standard minimally invasive approach. Total laparoscopic hysterectomy (TLH) or laparoscopic-assisted vaginal hysterectomy (LAVH). Robotic platform (da Vinci) offers wristed instruments and 3D visualization. Same recovery advantages as vaginal. Used for larger uteri, endometriosis, adnexal pathology, and oncologic indications.
Abdominal hysterectomy (TAH): Reserved for very large uteri, suspected malignancy requiring staging, dense pelvic adhesions, or cases requiring concomitant bowel/urologic procedures. Larger incision, longer recovery (4-6 weeks).
Types: Total hysterectomy (removal of uterine corpus and cervix), supracervical/subtotal hysterectomy (cervix preserved — faster procedure, debated role in preserving pelvic support and sexual function, requires ongoing Pap screening), radical hysterectomy (removal of uterus, cervix, parametria, upper vagina, pelvic LND — for cervical cancer). Opportunistic salpingectomy: Removal of fallopian tubes at the time of benign hysterectomy is now routinely recommended to reduce lifetime ovarian cancer risk (given STIC origin of HGSC).
Endometrial Ablation
Destruction of the endometrial lining for treatment of AUB in women who have completed childbearing. Methods: Radiofrequency (NovaSure — most common), thermal balloon (ThermaChoice), cryoablation, hydrothermal ablation, microwave. Prerequisites: negative endometrial biopsy (rule out malignancy/hyperplasia), completed childbearing, reliable contraception (pregnancy after ablation is dangerous). Amenorrhea rate: 30-50%; satisfaction rate: 85-90%. Not a sterilization method. Contraindicated with active uterine infection, prior classical CD, or desire for future fertility.
Cervical Ripening Methods
For induction of labor when the Bishop score is unfavorable (≤ 5):
| Method | Mechanism | Key Notes |
|---|---|---|
| Misoprostol (PGE1) | Prostaglandin → cervical remodeling + uterine contractions | 25 mcg PV q3-6h or 25-50 mcg PO q4h. Contraindicated in prior CD (uterine rupture risk) |
| Dinoprostone (PGE2, Cervidil) | Prostaglandin → cervical ripening | 10 mg vaginal insert (removable), or 0.5 mg gel intracervical. Can use with prior CD |
| Foley catheter (mechanical) | Mechanical dilation of the cervix | 30-80 mL balloon placed through the cervical os. Safe with prior CD. Can combine with misoprostol |
| Double-balloon catheter (Cook) | Mechanical dilation (intrauterine + vaginal balloons) | Similar efficacy to Foley; may reduce risk of ascending infection |
| Laminaria/osmotic dilators | Hygroscopic cervical dilation | Primarily used for second-trimester procedures rather than labor induction |
27 Classification Systems
FIGO Staging — Cervical Cancer (2018)
Stage IA: microscopic invasion (≤ 5 mm depth). IB: clinically visible or > 5 mm (IB1 ≤ 2 cm, IB2 2-4 cm, IB3 > 4 cm). IIA: upper 2/3 vagina (IIA1 ≤ 4 cm, IIA2 > 4 cm). IIB: parametrial invasion. IIIA: lower 1/3 vagina. IIIB: pelvic wall or hydronephrosis. IIIC: lymph node metastasis (IIIC1 pelvic, IIIC2 para-aortic). IVA: bladder/rectal mucosa. IVB: distant metastasis.
FIGO Staging — Endometrial Cancer (2023 Revised)
Stage IA: < 50% myometrial invasion. IB: ≥ 50% invasion. IC: aggressive histology confined to a polyp or endometrium. II: cervical stromal invasion. IIIA: serosa/adnexa. IIIB: vaginal/parametrial. IIIC: lymph node metastasis (IIIC1 pelvic, IIIC2 para-aortic). IVA: bladder/bowel mucosa. IVB: distant. The 2023 revision incorporates molecular classification (POLE, dMMR, p53abn, NSMP) into prognostic risk groups.
FIGO Staging — Ovarian Cancer
Stage I: confined to ovary/tube (IA one ovary, IB both, IC rupture/capsule surface/positive washings). II: pelvic extension (IIA uterus/tubes, IIB other pelvic tissue). III: peritoneal spread beyond pelvis or retroperitoneal nodes (IIIA microscopic peritoneal metastasis ± retroperitoneal nodes, IIIB macroscopic peritoneal ≤ 2 cm, IIIC > 2 cm or retroperitoneal nodes). IV: distant metastasis (IVA pleural effusion with positive cytology, IVB parenchymal liver/spleen metastasis or extra-abdominal).
Bethesda System for Pap Smear Reporting
NILM, ASC-US, ASC-H, LSIL, HSIL, SCC, AGC (NOS, favor neoplasia), AIS, adenocarcinoma. Specimen adequacy: satisfactory (with or without endocervical/transformation zone component) or unsatisfactory. See Section 13 for management algorithms.
Bishop Score
See Section 3 for the complete scoring table. Score ≥ 8 = favorable cervix. Score ≤ 5 = unfavorable, recommend cervical ripening.
POP-Q Staging
See Section 24 for the complete staging system description and table.
PALM-COEIN Classification of AUB
See Section 20 for the complete classification. Polyp, Adenomyosis, Leiomyoma, Malignancy & hyperplasia (structural/PALM); Coagulopathy, Ovulatory dysfunction, Endometrial, Iatrogenic, Not yet classified (non-structural/COEIN).
rASRM Endometriosis Staging
See Section 19. Stage I (1-5 pts), II (6-15), III (16-40), IV (> 40). Points assigned for: peritoneal implants (superficial vs deep, size), ovarian endometriomas (size, unilateral vs bilateral), and adhesions (filmy vs dense, extent of ovarian/tubal enclosure). Importantly, staging does NOT correlate well with pain severity or fertility prognosis — it was primarily designed to standardize surgical documentation.
Perineal Laceration Classification
See Section 9. First degree: vaginal mucosa/skin only. Second degree: perineal body musculature. Third degree: anal sphincter (3a < 50% EAS, 3b > 50% EAS, 3c includes IAS). Fourth degree: rectal mucosa. Third- and fourth-degree lacerations are collectively termed obstetric anal sphincter injuries (OASIS) and require subspecialist or experienced repair.
Cervical Cancer Screening Schedule Summary
Age 21-24: Pap alone every 3 years (HPV testing not recommended — high transient HPV prevalence). Age 25-65 (preferred): Primary HPV testing every 5 years. Age 25-65 (acceptable): Co-testing (HPV + Pap) every 5 years, or Pap alone every 3 years. Age > 65: Discontinue screening if adequate prior screening and no history of CIN2+ in the past 25 years. Post-hysterectomy: No screening if cervix removed and no history of CIN2+. Immunosuppressed (HIV+): Begin screening within 1 year of sexual activity (or by age 21), Pap annually, transition to co-testing at age 30.
Delivery Timing Guidelines by Condition
| Condition | Recommended Delivery Timing |
|---|---|
| Uncomplicated singleton pregnancy | 39 0/7 to 40 6/7 weeks |
| GDM, diet-controlled | 39 0/7 to 40 6/7 weeks |
| GDM, on medication (well-controlled) | 39 0/7 to 39 6/7 weeks |
| Pregestational DM (well-controlled) | 37 0/7 to 39 6/7 weeks |
| Chronic HTN (no meds) | 38 0/7 to 39 6/7 weeks |
| Chronic HTN (on meds) | 37 0/7 to 38 6/7 weeks |
| Gestational HTN | 37 0/7 weeks (upon diagnosis at ≥ 37 wk) |
| Preeclampsia without severe features | 37 0/7 weeks |
| Preeclampsia with severe features | 34 0/7 weeks (or earlier if unstable) |
| Placenta previa | 36 0/7 to 37 6/7 weeks (cesarean) |
| Prior classical cesarean | 36 0/7 to 37 0/7 weeks (cesarean) |
| Dichorionic-diamniotic twins | 38 0/7 to 38 6/7 weeks |
| Monochorionic-diamniotic twins | 36 0/7 to 37 6/7 weeks |
NICHD Three-Tier FHR Classification
See Section 12 for the complete classification table (Category I — normal, Category II — indeterminate, Category III — abnormal).
FIGO/WHO GTN Scoring System
See Section 17 for the complete scoring table. Score ≤ 6 = low-risk GTN (single-agent chemotherapy — methotrexate or actinomycin D). Score ≥ 7 = high-risk GTN (multi-agent chemotherapy — EMA-CO). Ultra-high-risk (score ≥ 13, brain/liver metastases) — induction EP before EMA-CO. Variables: age, antecedent pregnancy type, interval from index pregnancy, pre-treatment hCG, largest tumor size, number of metastases, site of metastases, prior failed chemotherapy. GTN has the highest cure rate of any solid malignancy with appropriate treatment.
28 Medications Master Table
Uterotonics
| Drug | Dose/Route | Mechanism | Key Contraindications |
|---|---|---|---|
| Oxytocin (Pitocin) | 10-40 units IV infusion | Oxytocin receptor agonist → myometrial contraction | Hypersensitivity (very few absolute CIs) |
| Methylergonovine (Methergine) | 0.2 mg IM | Ergot alkaloid → sustained uterine contraction | Hypertension, preeclampsia, CAD |
| Carboprost (Hemabate) | 0.25 mg IM q15-90 min (max 8 doses) | PGF2α analog → myometrial contraction | Asthma (causes bronchospasm) |
| Misoprostol (Cytotec) | 800-1000 mcg rectal/SL | PGE1 analog → myometrial contraction | Few absolute CIs; may cause fever, diarrhea |
Tocolytics
| Drug | Dose/Route | Key Side Effects/Notes |
|---|---|---|
| Nifedipine | 20-30 mg PO load, then 10-20 mg q4-6h | Hypotension, headache; avoid with MgSO4 |
| Indomethacin | 50-100 mg PO/PR load, then 25-50 mg q6h | Premature ductus closure (> 32 wks); oligohydramnios; limit to 48-72 hrs |
| Magnesium sulfate | 4-6 g IV load, 2-4 g/hr maintenance | Respiratory depression; also provides fetal neuroprotection < 32 wks |
| Terbutaline | 0.25 mg SQ | Tachycardia, pulmonary edema; FDA black box warning; limited use |
Hormonal Agents
| Drug | Indication | Key Notes |
|---|---|---|
| Combined OCPs | Contraception, AUB, endometriosis, PCOS | VTE risk increased 3-5×; CI in smokers ≥ 35 |
| Levonorgestrel IUD (Mirena) | Contraception, AUB, endometriosis, endometrial hyperplasia | Reduces menstrual bleeding 90-97%; can be therapeutic for AUB |
| Medroxyprogesterone (MPA) | AUB, endometrial hyperplasia, contraception (depot) | Depot: bone density concerns with prolonged use |
| Leuprolide (Lupron) | Fibroids, endometriosis, precocious puberty, prostate cancer | Initial flare then suppression; add-back therapy for > 6 months |
| Elagolix (Orilissa) | Endometriosis, uterine fibroids (with add-back) | Oral GnRH antagonist; dose-dependent estrogen suppression |
| Letrozole | Ovulation induction, breast cancer, off-label endometriosis | Aromatase inhibitor; first-line for ovulation induction in PCOS (vs clomiphene) |
| Clomiphene citrate | Ovulation induction | SERM; hot flashes, visual disturbances, multiple gestation risk |
Gynecologic Oncology Agents
| Drug | Cancer Type | Key Notes |
|---|---|---|
| Cisplatin | Cervical (concurrent with RT), ovarian | Nephrotoxic, ototoxic, neurotoxic; requires hydration |
| Carboplatin | Ovarian, endometrial | Less nephrotoxic than cisplatin; dosed by AUC (Calvert formula) |
| Paclitaxel | Ovarian, endometrial | Peripheral neuropathy, alopecia, hypersensitivity; premedicate |
| Bevacizumab (Avastin) | Ovarian (advanced), cervical | Anti-VEGF; risk of bowel perforation, HTN, wound complications |
| Olaparib (Lynparza) | Ovarian (BRCA/HRD+), maintenance | PARP inhibitor; myelosuppression, fatigue, small MDS/AML risk |
| Pembrolizumab (Keytruda) | Endometrial (dMMR/MSI-H), cervical (PD-L1+) | Immune checkpoint inhibitor; immune-related AEs |
| Dostarlimab (Jemperli) | Endometrial (dMMR/MSI-H) | Anti-PD-1; RUBY trial |
| Methotrexate | Ectopic pregnancy, GTN | Folate antagonist; hepatotoxic, myelosuppressive; leucovorin rescue for multi-dose |
Antibiotics (Gynecologic Infections)
| Condition | Regimen |
|---|---|
| Chlamydia | Doxycycline 100 mg PO BID × 7 days (preferred); azithromycin 1 g PO × 1 (alternative) |
| Gonorrhea | Ceftriaxone 500 mg IM × 1 (1 g if ≥ 150 kg) |
| PID (outpatient) | Ceftriaxone 500 mg IM + doxycycline 100 mg BID × 14 d + metronidazole 500 mg BID × 14 d |
| Bacterial vaginosis | Metronidazole 500 mg PO BID × 7 days; or metronidazole gel 0.75% intravaginal × 5 days |
| Vulvovaginal candidiasis | Fluconazole 150 mg PO × 1; or topical azole × 3-7 days |
| Trichomoniasis | Metronidazole 500 mg PO BID × 7 days (women); 2 g PO × 1 (men) |
| GBS prophylaxis (intrapartum) | Penicillin G 5 million units IV load, then 2.5-3 million units IV q4h until delivery |
29 Imaging & Diagnostics
Pelvic Ultrasound
Transvaginal ultrasound (TVUS) is the primary imaging modality in gynecology and early pregnancy. Higher resolution than transabdominal US for pelvic structures. Key measurements and findings:
Endometrial thickness: Postmenopausal woman with bleeding — ≤ 4 mm is reassuring (PPV for cancer very low). Premenopausal — varies with cycle (up to 16 mm in secretory phase).
Early pregnancy landmarks: Gestational sac (5 weeks), yolk sac (5.5 weeks), fetal pole with cardiac activity (6-6.5 weeks). Mean sac diameter (MSD) ≥ 25 mm without embryo = anembryonic pregnancy. Crown-rump length (CRL) ≥ 7 mm without cardiac activity = embryonic demise (ACOG/SRU criteria).
Ectopic pregnancy: Empty uterus + adnexal mass + free fluid. "Tubal ring sign" (hyperechoic ring around an extrauterine gestational sac). Pseudosac (fluid in endometrial cavity mimicking a gestational sac — lacks the double decidual sign).
Ovarian masses: Simple cyst (thin wall, anechoic, no solid component) = almost certainly benign. Complex features suggesting malignancy: solid components, thick septations, papillary projections, ascites, increased vascularity.
Fibroid mapping: Location (submucosal, intramural, subserosal), size, number, relationship to cavity (FIGO classification). Saline infusion sonohysterography (SIS) for submucosal fibroid characterization.
Hysterosalpingography (HSG)
Fluoroscopic evaluation of the uterine cavity and fallopian tube patency. Radiopaque contrast is injected through the cervix. Indications: Infertility workup — assesses tubal patency (bilateral spill of contrast into the peritoneal cavity = patent tubes), uterine cavity contour (filling defects = polyps, fibroids, synechiae; abnormal shape = uterine anomaly). Complications: Infection, contrast allergy, pain, vasovagal response.
MRI Pelvis
Superior soft-tissue contrast resolution. Indications: Fibroid mapping before myomectomy (especially for large or numerous fibroids), adenomyosis confirmation (junctional zone > 12 mm), deep infiltrating endometriosis evaluation, cervical cancer staging (parametrial invasion assessment), uterine anomaly classification (Mullerian anomalies), adnexal mass characterization (indeterminate on US). MRI is the most accurate imaging modality for placenta accreta spectrum (loss of retroplacental dark zone, uterine bulging, bladder invasion).
Fetal Heart Rate Monitoring Terminology
See Section 12 for complete NICHD nomenclature (baseline, variability, accelerations, early/late/variable/prolonged decelerations, sinusoidal pattern, Category I/II/III system).
Colposcopy Terminology
IFCPC 2011 nomenclature: Normal: Mature/immature squamous epithelium, columnar epithelium, transformation zone (types 1-3). Abnormal Grade 1 (minor): Thin acetowhite, fine punctation, fine mosaic. Abnormal Grade 2 (major): Dense acetowhite, coarse punctation, coarse mosaic, sharp border, rapid acetowhite onset, inner border sign, ridge sign. Suspicious for invasion: Atypical vessels, fragile tissue, irregular surface, exophytic lesion, necrosis/ulceration.
Amniocentesis & CVS
Amniocentesis: Performed at 15-20 weeks under continuous US guidance. A spinal needle is inserted transabdominally into the amniotic cavity, and 20-30 mL of amniotic fluid is aspirated. Provides fetal karyotype, microarray, molecular testing (FISH for rapid aneuploidy, full karyotype in 7-14 days). Also used for fetal lung maturity testing (lamellar body count, lecithin/sphingomyelin ratio — largely replaced by gestational age-based management), assessment of fetal infection (CMV PCR), and bilirubin measurement in Rh disease. Procedure-related pregnancy loss: ~0.1-0.3%.
Chorionic villus sampling (CVS): Performed at 10-13 weeks. Transcervical or transabdominal approach under US guidance. Samples placental chorionic villi for karyotype, microarray, molecular testing. Advantage: earlier diagnosis than amniocentesis. Disadvantage: cannot detect neural tube defects (no amniotic fluid AFP), risk of confined placental mosaicism (~1%). Procedure-related pregnancy loss: ~0.5-1%.
Key OB/GYN Landmark Trials
WOMAN Trial (2017): Tranexamic acid 1 g IV within 3 hours reduces death from PPH by 19%. Now standard of care.
ARRIVE Trial (2018): Elective induction at 39 weeks in low-risk nulliparas reduces cesarean rate (18.6% vs 22.2%) without increasing adverse neonatal outcomes. Changed practice regarding elective induction.
MAGPIE Trial (2002): MgSO4 reduces eclampsia risk by 58% in women with preeclampsia. Definitive trial for magnesium prophylaxis.
BEAM Trial (2008): MgSO4 before anticipated preterm delivery at < 32 weeks reduces moderate-severe cerebral palsy (RR 0.55). Basis for fetal neuroprotection indication.
SOLO-1 Trial (2018): Olaparib maintenance after first-line platinum response in BRCA-mutated ovarian cancer: 5-year PFS 48% vs 21%.
RUBY Trial (2023): Dostarlimab + chemotherapy improved PFS in advanced endometrial cancer, especially dMMR/MSI-H (PFS HR 0.28).
GOG LAP2 (2012): Laparoscopic surgical staging for endometrial cancer is non-inferior to laparotomy with fewer complications. Established MIS as standard.
WHI (2002): Defined risks and benefits of postmenopausal hormone therapy. Led to nuanced "timing hypothesis" for MHT initiation.
30 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AIS | Adenocarcinoma in situ |
| AMA | Advanced maternal age (≥ 35 at delivery) |
| AMH | Anti-Müllerian hormone |
| ART | Assisted reproductive technology |
| ASC-H | Atypical squamous cells, cannot exclude HSIL |
| ASC-US | Atypical squamous cells of undetermined significance |
| AUB | Abnormal uterine bleeding |
| BPP | Biophysical profile |
| BRCA | Breast cancer susceptibility gene |
| BSO | Bilateral salpingo-oophorectomy |
| CD | Cesarean delivery |
| CIN | Cervical intraepithelial neoplasia (1, 2, 3) |
| CRL | Crown-rump length |
| CVS | Chorionic villus sampling |
| D&C | Dilation and curettage |
| DES | Diethylstilbestrol |
| DIE | Deep infiltrating endometriosis |
| dMMR | Deficient mismatch repair |
| ECC | Endocervical curettage |
| EFM | Electronic fetal monitoring |
| EFW | Estimated fetal weight |
| EMB | Endometrial biopsy |
| FHR | Fetal heart rate |
| FSE | Fetal scalp electrode |
| GBS | Group B Streptococcus |
| GCT | Glucose challenge test |
| GDM | Gestational diabetes mellitus |
| GnRH | Gonadotropin-releasing hormone |
| GSM | Genitourinary syndrome of menopause |
| GTD | Gestational trophoblastic disease |
| GTN | Gestational trophoblastic neoplasia |
| GTT | Glucose tolerance test |
| hCG | Human chorionic gonadotropin |
| HELLP | Hemolysis, Elevated Liver enzymes, Low Platelets |
| HGSC | High-grade serous carcinoma |
| HPO | Hypothalamic-pituitary-ovarian (axis) |
| HPV | Human papillomavirus |
| HRD | Homologous recombination deficiency |
| HRT/MHT | Hormone replacement therapy / Menopausal hormone therapy |
| HSG | Hysterosalpingography |
| HSIL | High-grade squamous intraepithelial lesion |
| IOTA | International Ovarian Tumor Analysis |
| IP ligament | Infundibulopelvic ligament |
| IUP | Intrauterine pregnancy |
| IUPC | Intrauterine pressure catheter |
| IVF | In vitro fertilization |
| LEEP | Loop electrosurgical excision procedure |
| LGSC | Low-grade serous carcinoma |
| LSIL | Low-grade squamous intraepithelial lesion |
| MCA-PSV | Middle cerebral artery peak systolic velocity |
| MFM | Maternal-fetal medicine |
| MIGS | Minimally invasive gynecologic surgery |
| MSI-H | Microsatellite instability-high |
| NACT | Neoadjuvant chemotherapy |
| NIPT | Non-invasive prenatal testing (cell-free DNA) |
| NST | Non-stress test |
| NT | Nuchal translucency |
| OAB | Overactive bladder |
| OCP | Oral contraceptive pill |
| PALM-COEIN | FIGO classification of AUB causes |
| PARP | Poly(ADP-ribose) polymerase |
| PAS | Placenta accreta spectrum |
| PCOS | Polycystic ovary syndrome |
| PID | Pelvic inflammatory disease |
| POI | Premature ovarian insufficiency |
| POP | Pelvic organ prolapse |
| POP-Q | Pelvic Organ Prolapse Quantification |
| PPH | Postpartum hemorrhage |
| PPROM | Preterm premature rupture of membranes |
| PSTT | Placental site trophoblastic tumor |
| PVR | Post-void residual |
| QBL | Quantitative blood loss |
| rASRM | Revised American Society for Reproductive Medicine (endometriosis staging) |
| RRBSO | Risk-reducing bilateral salpingo-oophorectomy |
| SCC | Squamous cell carcinoma |
| SIS | Saline infusion sonohysterography |
| SLN | Sentinel lymph node |
| STIC | Serous tubal intraepithelial carcinoma |
| SUI | Stress urinary incontinence |
| TH/BSO | Total hysterectomy / bilateral salpingo-oophorectomy |
| TOA | Tubo-ovarian abscess |
| TOLAC | Trial of labor after cesarean |
| TVUS | Transvaginal ultrasound |
| TXA | Tranexamic acid |
| UAE | Uterine artery embolization |
| UDS | Urodynamic studies |
| UUI | Urgency urinary incontinence |
| VBAC | Vaginal birth after cesarean |
| VMS | Vasomotor symptoms |
| VTE | Venous thromboembolism |
| WHI | Women’s Health Initiative |
| WLE | Wide local excision |
Key PubMed References
• ACOG Practice Bulletin No. 222: Gestational Hypertension and Preeclampsia. Obstet Gynecol. 2020;135(6):e237-e260. PubMed 32443079
• WOMAN Trial Collaborators. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post-partum haemorrhage. Lancet. 2017;389(10084):2105-2116. PubMed 28456509
• Grobman WA, et al. Labor Induction versus Expectant Management in Low-Risk Nulliparous Women (ARRIVE Trial). N Engl J Med. 2018;379(6):513-523. PubMed 30089070
• Moore K, et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer (SOLO-1). N Engl J Med. 2018;379(26):2495-2505. PubMed 30345884
• Mirza MR, et al. Dostarlimab for Primary Advanced or Recurrent Endometrial Cancer (RUBY). N Engl J Med. 2023;388(23):2145-2158. PubMed 36972026
• Colombo N, et al. ESMO-ESGO-ESTRO Consensus Conference on Endometrial Cancer. Ann Oncol. 2016;27(1):16-41. PubMed 26634381
• ACOG Practice Bulletin No. 228: Management of Symptomatic Uterine Leiomyomas. Obstet Gynecol. 2021;137(6):e100-e115. PubMed 34011888
• Zhang J, et al. Reassessing the labor curve in nulliparous women (Consortium on Safe Labor). Am J Obstet Gynecol. 2010;203(4):326.e1-10. PubMed 20708166
• Rolnik DL, et al. Aspirin versus Placebo in Pregnancies at High Risk for Preterm Preeclampsia (ASPRE). N Engl J Med. 2017;377(7):613-622. PubMed 28657417
• The Endometriosis Guideline 2022 (ESHRE). PubMed 35024833
• ACOG/SMFM Obstetric Care Consensus No. 7: Placenta Accreta Spectrum. Obstet Gynecol. 2018;132(6):e259-e275. PubMed 30461695
• Persson M, et al. Updated ASCCP Risk-Based Management Consensus Guidelines for Abnormal Cervical Cancer Screening Tests and Cancer Precursors. J Low Genit Tract Dis. 2020;24(2):102-131. PubMed 32243307