Ophthalmology
Every diagnosis, procedure, surgical technique, classification, complication, medication, and management algorithm across the full scope of ophthalmology in one place.
01 Ocular Anatomy
Globe Layers
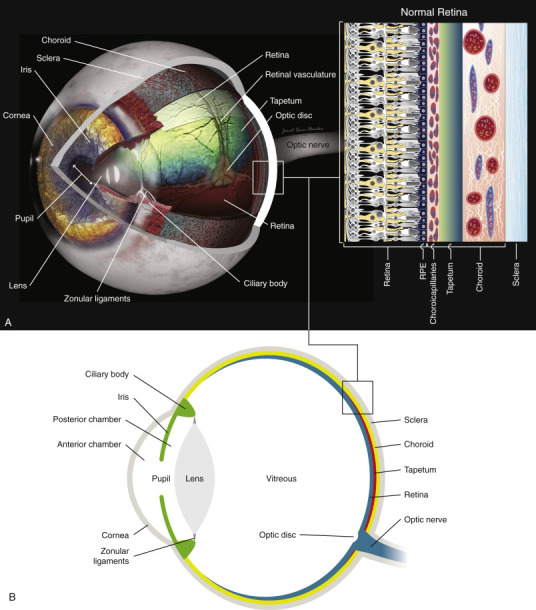
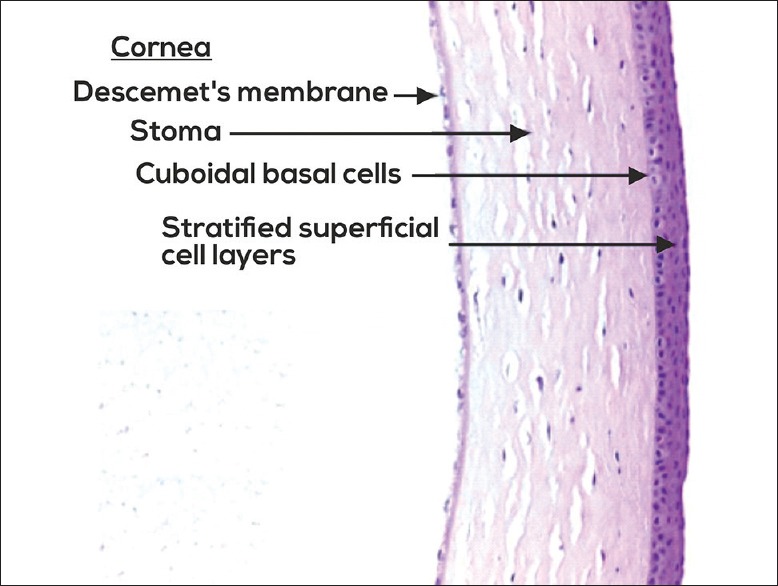
The eye is a ~24 mm sphere composed of three concentric coats. The outer fibrous coat consists of the sclera (opaque, collagen-rich posterior five-sixths) and the cornea (transparent anterior one-sixth). The cornea provides approximately two-thirds (~43 D) of the eye's total refractive power. It has five layers from anterior to posterior: epithelium, Bowman layer, stroma (90% of corneal thickness), Descemet membrane, and endothelium. The corneal endothelium is a single-cell layer of non-regenerating cells that maintains corneal clarity via its pump function; a cell count below ~500 cells/mm² leads to corneal edema and decompensation.
The middle vascular coat (uvea) comprises the iris (controls pupil size, divides anterior and posterior chambers), the ciliary body (produces aqueous humor, controls accommodation via the zonular fibers attached to the lens), and the choroid (highly vascularized layer supplying the outer retina and RPE). The choroid receives the greatest blood flow per gram of tissue of any organ in the body.
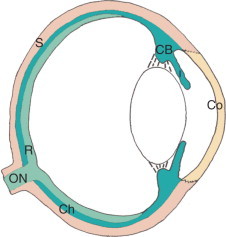
The inner neural coat (retina) is a 10-layer neurosensory tissue. Key structures include the macula (5.5 mm diameter area centered temporal to the optic disc responsible for central vision), the fovea (1.5 mm pit at the center of the macula, composed exclusively of cone photoreceptors, the region of highest visual acuity), and the optic disc (1.5 mm diameter, where retinal ganglion cell axons exit as the optic nerve; the physiologic blind spot). The retinal pigment epithelium (RPE) lies between the neurosensory retina and Bruch membrane, maintaining photoreceptor health through phagocytosis of outer segments, vitamin A metabolism, and the outer blood-retinal barrier.
Chambers, Lens & Vitreous
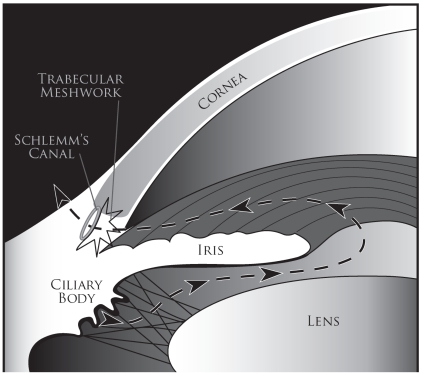
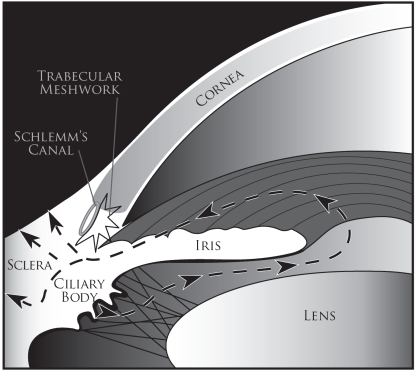
The anterior chamber lies between the cornea and the iris/pupil. The posterior chamber lies between the posterior iris and the lens. Both contain aqueous humor produced by the ciliary body at a rate of ~2.5 μL/min. Aqueous flows from the posterior chamber through the pupil into the anterior chamber and exits via two pathways: the trabecular meshwork (conventional pathway, ~90%) draining into Schlemm canal and then aqueous veins, and the uveoscleral (unconventional) pathway (~10%) through the ciliary muscle into the suprachoroidal space. Prostaglandin analogs increase uveoscleral outflow; this is why they are first-line glaucoma therapy.
The crystalline lens is a biconvex, avascular transparent structure suspended by zonular fibers from the ciliary body. It contributes ~20 D of refractive power and can change shape for accommodation (near focus) by contraction of the ciliary muscle (relaxing zonules). With age, the lens becomes progressively less flexible (presbyopia, beginning ~age 40) and less transparent (cataract). The vitreous humor fills the posterior segment (~4 mL, 80% of the globe volume), composed of 99% water with a collagen-hyaluronic acid framework. Posterior vitreous detachment (PVD) occurs when the vitreous separates from the retina — common after age 60; usually benign but can cause retinal tears at points of vitreoretinal adhesion.
Extraocular Muscles
Six extraocular muscles (EOMs) control eye movement, all originating from the annulus of Zinn at the orbital apex except the inferior oblique (which originates from the maxillary bone near the lacrimal fossa). The lateral rectus is innervated by CN VI (abducens), the superior oblique by CN IV (trochlear), and the remaining four muscles (medial rectus, superior rectus, inferior rectus, inferior oblique) by CN III (oculomotor). Mnemonic: LR6 SO4 — all the Rest 3.
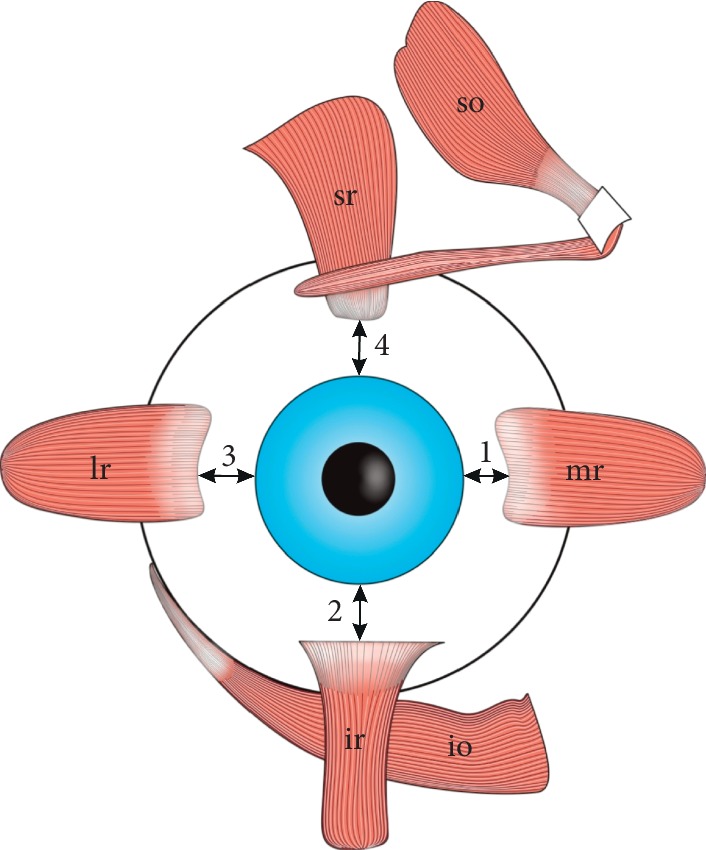
Actions: the medial rectus adducts; the lateral rectus abducts; the superior rectus primarily elevates (also intorts and adducts); the inferior rectus primarily depresses (also extorts and adducts); the superior oblique primarily intorts (also depresses and abducts); the inferior oblique primarily extorts (also elevates and abducts). The oblique muscles have their greatest vertical action when the eye is adducted, and the vertical recti have their greatest vertical action when the eye is abducted.
Lacrimal System
The lacrimal gland (located superolaterally in the lacrimal fossa of the frontal bone) produces the aqueous component of the tear film. Tears flow across the ocular surface and drain via the upper and lower puncta (medial lid margin) into the canaliculi, then the lacrimal sac (in the lacrimal fossa of the lacrimal bone), and finally through the nasolacrimal duct into the inferior nasal meatus beneath the inferior turbinate. Obstruction at any point causes epiphora (tearing). Congenital nasolacrimal duct obstruction occurs in ~6% of newborns, usually at the valve of Hasner; most resolve spontaneously by age 12 months. Dacryocystorhinostomy (DCR) is the definitive surgical treatment for adult nasolacrimal duct obstruction.
Orbit
The orbit is a pyramidal bony cavity formed by seven bones: frontal, zygomatic, maxillary, lacrimal, ethmoid, sphenoid (greater and lesser wings), and palatine. The optic canal (lesser wing of sphenoid) transmits the optic nerve and ophthalmic artery. The superior orbital fissure transmits CN III, CN IV, CN VI, V1 (ophthalmic division of trigeminal), and the superior ophthalmic vein. The inferior orbital fissure transmits V2 (maxillary nerve) and the inferior ophthalmic vein. The medial wall (lamina papyracea of ethmoid) is paper-thin and easily fractured — ethmoid sinusitis can spread to cause orbital cellulitis.
02 Optics & Refraction
Refractive Errors
Emmetropia is the state in which parallel light rays focus precisely on the retina without accommodation. Refractive errors (ametropia) are the most common cause of correctable visual impairment worldwide.
Myopia (nearsightedness): The eye is too long or the cornea too steep — light focuses anterior to the retina. Corrected with minus (concave/diverging) lenses. High myopia (> -6 D) increases risk for retinal detachment, glaucoma, posterior staphyloma, and myopic macular degeneration. Prevalence is rising globally, approaching 50% in young adults in East Asian populations.
Hyperopia (farsightedness): The eye is too short or the cornea too flat — light focuses posterior to the retina. Corrected with plus (convex/converging) lenses. Mild hyperopia may be compensated by accommodation in young patients but becomes symptomatic with age. Hyperopic eyes have shorter axial length and shallower anterior chambers, predisposing to angle-closure glaucoma.
Astigmatism: The cornea (or lens) has different curvatures in different meridians — light forms two focal lines rather than a single focal point. Regular astigmatism is corrected with cylindrical lenses; irregular astigmatism (e.g., keratoconus) requires rigid gas-permeable contact lenses or surgical correction. Classified as with-the-rule (steeper vertical meridian, common in youth), against-the-rule (steeper horizontal meridian, common in elderly), or oblique.
Presbyopia: Age-related loss of accommodative amplitude due to progressive lens sclerosis and loss of elasticity. Typically becomes clinically significant around age 40 when near point of focus recedes beyond comfortable reading distance. Accommodative amplitude declines from ~14 D at age 10 to ~1 D by age 60 (Duane's age-expected norms). Corrected with reading glasses, bifocals, progressive lenses, or multifocal contact lenses/IOLs.
| Condition | Mechanism | Correction | Associations |
|---|---|---|---|
| Myopia | Eye too long / cornea too steep → focus anterior to retina | Minus (concave) lens | Retinal detachment, glaucoma, posterior staphyloma, myopic maculopathy |
| Hyperopia | Eye too short / cornea too flat → focus posterior to retina | Plus (convex) lens | Angle-closure glaucoma, accommodative esotropia, amblyopia |
| Astigmatism | Unequal corneal curvature → two focal lines | Cylindrical lens / toric IOL | Irregular: keratoconus, corneal scar, post-surgical |
| Presbyopia | Age-related lens sclerosis → loss of accommodation | Plus add for near (reading glasses, bifocal, progressive) | Universal after age 40-45 |
Basic Optics Principles
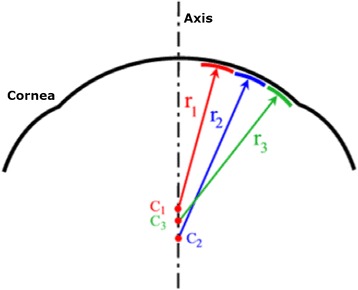
The total refractive power of the eye is approximately 60 D: the cornea contributes ~43 D (front surface ~48 D minus back surface ~-5 D) and the lens contributes ~20 D. Snell's law governs refraction: n1 sin θ1 = n2 sin θ2. The vergence formula U + P = V (where U = incoming vergence, P = lens power in diopters, V = outgoing vergence) is the fundamental equation for ophthalmic optics. A diopter is the reciprocal of the focal length in meters (D = 1/f).
Refractive Surgery Overview
Modern refractive procedures reshape the cornea or implant intraocular lenses to correct ametropia. LASIK (laser in situ keratomileusis) creates a corneal flap then uses an excimer laser (193 nm) to ablate the stromal bed. PRK (photorefractive keratectomy) ablates directly on the corneal surface after removing the epithelium — no flap, slower recovery but avoids flap complications. SMILE (small incision lenticule extraction) uses a femtosecond laser to create and remove an intrastromal lenticule through a small incision — no flap, less dry eye, but currently limited to myopic correction. IOL-based correction includes phakic IOLs (e.g., Visian ICL placed in the posterior chamber) for high myopia/hyperopia beyond the range of laser correction, and refractive lens exchange (clear lens extraction with premium IOL) for presbyopia.
IOL Power Calculation
Accurate IOL power calculation is essential for achieving the desired refractive outcome after cataract surgery. Key measurements: axial length (AL) — measured by optical biometry (IOLMaster, Lenstar — gold standard; uses partial coherence interferometry) or A-scan ultrasound (contact method — used when optical biometry fails, e.g., dense cataract), keratometry (K values) — corneal curvature measured by the biometer or topographer, and anterior chamber depth (ACD). IOL calculation formulas have evolved through generations:
| Generation | Formulas | Best For |
|---|---|---|
| 3rd generation (theoretical) | SRK/T, Holladay 1, Hoffer Q | SRK/T: long eyes (> 26 mm); Hoffer Q: short eyes (< 22 mm); Holladay 1: average eyes |
| 4th generation | Haigis, Holladay 2, Olsen | Improved accuracy across all axial lengths; use more biometric variables |
| AI/modern | Barrett Universal II, Hill-RBF, Kane, EVO | Best overall accuracy across all eye sizes; Barrett Universal II most widely used |
Special situations: post-refractive surgery eyes (post-LASIK/PRK) — standard formulas overestimate corneal power, leading to hyperopic surprises. Special methods required: clinical history method, Haigis-L, Barrett True-K, Shammas, or ASCRS online calculator. Silicone oil-filled eyes — AL measurement is inaccurate due to altered sound velocity; formulas with adjusted constants are needed. Target refraction: most patients are targeted for emmetropia (plano) or slight myopia (-0.25 to -0.50 D) for distance focus. Monovision: dominant eye set for distance, non-dominant eye for -1.25 to -2.0 D (near).
Monofocal: Single focal point — most common; patient typically set for distance and uses reading glasses for near. Can be used for monovision (dominant eye set for distance, non-dominant for near).
Multifocal: Concentric rings create multiple focal points for distance and near. Trade-off: increased halos and glare, reduced contrast sensitivity. Not ideal for patients with macular disease or high expectations.
Toric: Cylindrical correction built into the IOL to correct astigmatism (≥ 1.0 D). Requires precise rotational alignment during surgery.
Extended depth of focus (EDOF): Creates a single elongated focal zone rather than multiple discrete foci — better intermediate vision with fewer halos than multifocal. Example: Vivity (AcrySof).
Accommodating: Designed to shift position with ciliary muscle contraction (limited real-world efficacy). Example: Crystalens.
03 The Ophthalmic Examination
Visual Acuity
Visual acuity (VA) is the most important single measurement in ophthalmology. Measured using a Snellen chart at 20 feet (6 meters) or an ETDRS chart at 4 meters (preferred in clinical trials for its logarithmic design). Notation: 20/20 means the patient reads at 20 feet what a normal eye reads at 20 feet. 20/200 or worse in the better eye with best correction defines legal blindness. When the patient cannot read letters: count fingers (CF), hand motions (HM), light perception (LP), or no light perception (NLP). Pinhole acuity improves vision that is limited by refractive error (by reducing the effective aperture and increasing depth of focus) but does not improve vision limited by retinal or optic nerve disease.
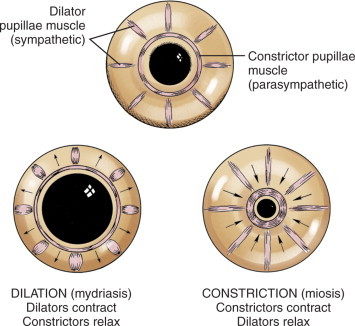
Pupillary Examination
Pupil evaluation is critical in neuro-ophthalmology. The relative afferent pupillary defect (RAPD, Marcus Gunn pupil) is detected by the swinging flashlight test: when light is swung from the normal eye to the affected eye, the affected pupil paradoxically dilates because its afferent input is weaker. An RAPD indicates asymmetric optic nerve disease (optic neuritis, ischemic optic neuropathy, compressive lesion, severe unilateral retinal disease). An RAPD is never caused by cataract, refractive error, or amblyopia.
Slit-Lamp Biomicroscopy
The slit lamp provides magnified, illuminated examination of the anterior segment. Key findings: corneal pathology (ulcers, dystrophies, edema, staining with fluorescein), anterior chamber cells and flare (graded 0 to 4+ for uveitis activity — SUN criteria), lens opacity (cataract type and grade), iris abnormalities (neovascularization, synechiae, nodules), and angle evaluation (with gonioscopy lens). With accessory lenses (78 D, 90 D, or Volk lens), the slit lamp provides a stereoscopic view of the fundus.
Intraocular Pressure Measurement
Goldmann applanation tonometry (GAT) is the gold standard for IOP measurement. Based on the Imbert-Fick principle: the force needed to flatten a 3.06 mm diameter area of the cornea equals the IOP. Normal IOP range: 10-21 mmHg (mean ~16 mmHg). Central corneal thickness (CCT) affects GAT readings: thin corneas (< 520 μm) underestimate and thick corneas (> 580 μm) overestimate IOP. Other methods: non-contact tonometry (NCT, "air puff") for screening; iCare rebound tonometry (no topical anesthesia needed, useful in children); Tono-Pen (portable, useful for irregular corneas); pneumatonometry (measures through bandage contact lenses).
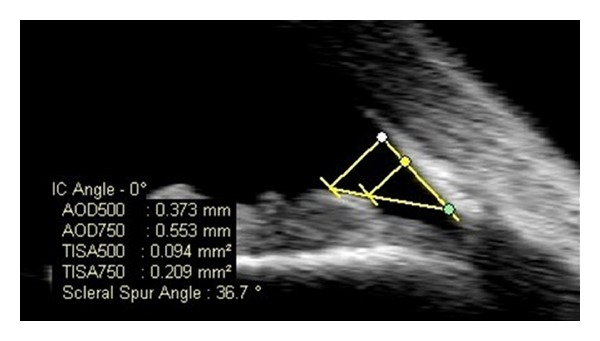
Gonioscopy
Gonioscopy is the examination of the anterior chamber angle using a contact lens with mirrors (Goldmann, Zeiss, Posner). The angle structures visible from anterior to posterior: Schwalbe line (termination of Descemet membrane), trabecular meshwork (pigmented and non-pigmented portions), scleral spur, ciliary body band, and iris root. The Shaffer grading system classifies angle width: Grade 4 (wide open, 35-45 degrees), Grade 3 (open, 25-35 degrees), Grade 2 (narrow, 20 degrees), Grade 1 (very narrow, 10 degrees — potential for closure), Grade 0 (closed). Indentation gonioscopy differentiates appositional from synechial angle closure.
Fundoscopy & Imaging
Direct ophthalmoscopy provides an upright, magnified (15x) view of the retina with a narrow field of view (~5 degrees). Indirect ophthalmoscopy (BIO) with a condensing lens (20 D or 28 D) provides an inverted, wider field (~40-60 degrees), stereoscopic, dimmer image — preferred for retinal examination, especially in the periphery. Optical coherence tomography (OCT) provides cross-sectional imaging of the retina and optic nerve at near-histologic resolution (~5 μm axial) — essential for diagnosing and monitoring macular diseases (DME, AMD, epiretinal membrane, macular hole) and glaucoma (RNFL thickness). Fluorescein angiography (FA) uses intravenous sodium fluorescein to image retinal and choroidal vasculature — identifies areas of leakage (edema), ischemia (capillary non-perfusion), and neovascularization. OCT angiography (OCTA) is a non-invasive, dye-free alternative that maps retinal vasculature using motion contrast.
Visual Field Testing
Humphrey visual field (HVF) automated perimetry is the standard for detecting and monitoring glaucomatous and neurological visual field loss. Key programs: 24-2 (tests central 24 degrees, standard for glaucoma), 10-2 (tests central 10 degrees, for advanced glaucoma and macular disease), 30-2 (tests central 30 degrees, for neurological fields). Reliability indices: fixation losses (< 20%), false positives (< 15%), false negatives (< 33%). Classic patterns: arcuate scotoma (glaucoma — follows the nerve fiber layer), bitemporal hemianopia (chiasmal compression), homonymous hemianopia (post-chiasmal lesion), central scotoma (optic neuritis, macular disease), altitudinal defect (ischemic optic neuropathy, branch retinal artery occlusion).
04 Cataracts & Lens Surgery
Cataract Types & Pathophysiology
A cataract is any opacity of the crystalline lens. Cataracts are the leading cause of reversible blindness worldwide. Risk factors include age (most common), UV exposure, diabetes, corticosteroid use, smoking, trauma, and radiation.
Nuclear sclerotic cataract: The most common age-related type. Progressive yellowing and hardening of the lens nucleus. Causes a "myopic shift" (second sight of the elderly) as the refractive index of the nucleus increases. Progresses slowly; eventual brunescence impairs red reflex.
Cortical cataract: Spoke-like (cuneiform) opacities radiating from the periphery toward the center in the lens cortex. Associated with diabetes. May cause significant glare, especially with oncoming headlights. The cortex hydrates and swells.
Posterior subcapsular cataract (PSC): Granular plaque-like opacity on the posterior capsule just anterior to the posterior capsule. Causes disproportionate visual symptoms relative to size because of its location in the nodal point of the eye. Associated with corticosteroid use (topical or systemic), diabetes, uveitis, radiation, and younger age. Patients complain of glare and difficulty reading.
LOCS III Grading
The Lens Opacities Classification System III (LOCS III) is the standard grading system for cataracts, using slit-lamp comparison photographs. Grades nuclear color (NC1-NC6), nuclear opalescence (NO1-NO6), cortical cataract (C1-C5), and posterior subcapsular cataract (P1-P5). Used in clinical trials and for surgical planning.
Phacoemulsification Technique
Phacoemulsification ("phaco") is the modern standard cataract surgery, performed through a small (~2.2-2.8 mm) clear corneal incision or scleral tunnel. Steps: (1) paracentesis and injection of viscoelastic, (2) main incision, (3) continuous curvilinear capsulorhexis (CCC — circular opening in the anterior capsule, typically 5-5.5 mm), (4) hydrodissection (separating cortex from capsule), (5) phacoemulsification of the nucleus using ultrasound energy (divide-and-conquer or chopping techniques), (6) irrigation/aspiration of residual cortex, (7) capsular bag polishing, (8) IOL implantation into the capsular bag, (9) removal of viscoelastic, (10) wound hydration to seal the incision. Femtosecond laser-assisted cataract surgery (FLACS) automates steps of the capsulorhexis, lens fragmentation, and corneal incision.
Cataract Surgery Complications
Posterior capsule rupture (PCR): The most feared intraoperative complication (~1-2% of cases). Risk is higher with dense cataracts, small pupils, pseudoexfoliation, and floppy iris syndrome (tamsulosin use — IFIS). Management depends on timing: if the capsular bag is unstable, vitrectomy must be performed to clear vitreous from the wound, and the IOL may need to be placed in the sulcus (with optic capture through the capsulorhexis) or sutured/glued to the sclera. IOL power calculation for sulcus placement: reduce power by 0.5-1.0 D compared to bag placement.
Dropped nucleus: Nuclear fragments falling into the vitreous cavity — requires pars plana vitrectomy by a retinal surgeon. Do NOT attempt to retrieve with the phaco handpiece.
Zonular dialysis: Weakness or loss of zonular support (pseudoexfoliation, Marfan syndrome, homocystinuria, trauma). May require capsular tension rings (CTR) or iris/scleral fixation of the IOL.
Postoperative endophthalmitis: The most devastating complication (~0.04-0.1% incidence). Typically presents 1-7 days post-op with pain, decreased vision, hypopyon, and vitritis. Most common organism: Staphylococcus epidermidis (coagulase-negative staph). Most virulent: Staphylococcus aureus and Streptococcus species. The Endophthalmitis Vitrectomy Study (EVS) showed: patients with VA of hand motions or better had equal outcomes with tap/inject vs. immediate vitrectomy; patients with LP-only vision benefited from immediate vitrectomy. Intravitreal antibiotics: vancomycin 1 mg/0.1 mL + ceftazidime 2.25 mg/0.1 mL (or amikacin if cephalosporin allergic).
Cystoid macular edema (CME, Irvine-Gass syndrome): Peaks 4-6 weeks post-op. Clinical CME in ~1-2% of uncomplicated cases. Treat with topical NSAIDs (ketorolac, nepafenac) and corticosteroids; refractory cases may require sub-Tenon or intravitreal triamcinolone or intravitreal anti-VEGF.
Posterior capsule opacification (PCO): The most common late complication ("secondary cataract"), occurring in up to 20-40% at 5 years. Due to proliferation and migration of residual lens epithelial cells. Treated with Nd:YAG laser posterior capsulotomy — highly effective office procedure.
IOL dislocation: Early (within days — from zonular weakness or capsular tear) or late (years — progressive zonular dehiscence, especially with pseudoexfoliation). Management: repositioning, IOL exchange, or scleral fixation.
Toxic anterior segment syndrome (TASS): Sterile postoperative inflammation within 12-48 hours caused by contaminants (residual detergent on instruments, preservatives, endotoxins). Distinguished from endophthalmitis by earlier onset and absence of vitreous involvement. Treated with intensive topical steroids.
05 Corneal Diseases & Transplantation
Keratoconus
Keratoconus is a progressive, bilateral (often asymmetric), non-inflammatory corneal ectasia characterized by central or paracentral stromal thinning and conical protrusion. Onset typically in puberty, progresses until the third or fourth decade. Risk factors: eye rubbing (strongest modifiable risk factor), atopy, Down syndrome, connective tissue disorders. Signs: Munson sign (V-shaped deformation of the lower lid on downgaze), Vogt striae (fine vertical stress lines in the deep stroma), Fleischer ring (iron deposition at the base of the cone), scissors reflex on retinoscopy.
Stage I: Eccentric corneal steepening, induced astigmatism < 5 D, corneal radii ≤ 48 D, no scarring.
Stage II: Induced astigmatism 5-8 D, corneal radii ≤ 53 D, no scarring, minimum corneal thickness ≥ 400 μm.
Stage III: Induced astigmatism 8-10 D, corneal radii > 53 D, no scarring, minimum corneal thickness 200-400 μm.
Stage IV: Refraction not measurable, corneal radii > 55 D, central scarring, minimum corneal thickness < 200 μm.
Management: spectacles (mild), rigid gas-permeable (RGP) contact lenses (moderate — provide regular anterior surface), scleral lenses, corneal collagen cross-linking (CXL) with riboflavin and UVA light (halts progression — indicated for documented progression, especially in young patients), INTACS (intrastromal corneal ring segments — flatten the cone), and corneal transplantation (deep anterior lamellar keratoplasty — DALK, preferred as it preserves the patient's endothelium; or penetrating keratoplasty — PK) for advanced disease with scarring.
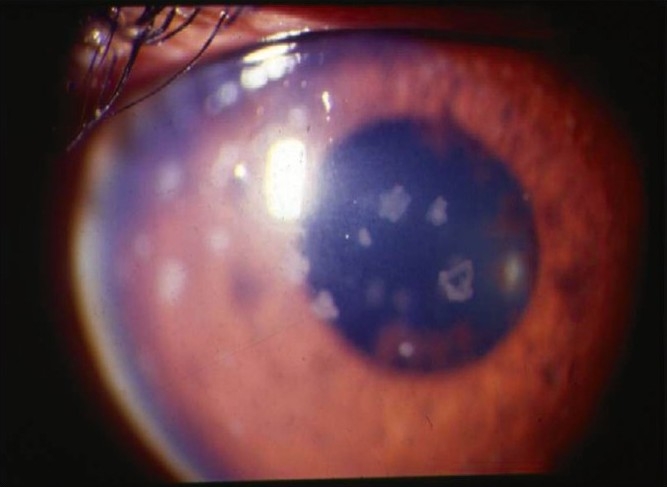
Fuchs Endothelial Dystrophy
Fuchs dystrophy is the most common corneal dystrophy requiring surgery. Autosomal dominant with variable penetrance, more common in women, onset 4th-5th decade. Characterized by progressive loss of corneal endothelial cells and formation of guttae (excrescences on Descemet membrane) → corneal edema → decreased vision, especially in the morning (edema worsens overnight with lid closure). Progresses from central guttae only to corneal edema, epithelial bullae (bullous keratopathy), and subepithelial scarring.
Surgical treatment: Descemet membrane endothelial keratoplasty (DMEK) — transplants only the Descemet membrane and endothelium; fastest visual recovery, lowest rejection rate (~1%), technically more challenging (graft detachment rate ~10-15%). Descemet stripping automated endothelial keratoplasty (DSAEK) — transplants the posterior stroma, Descemet membrane, and endothelium; slightly slower visual recovery but easier to perform. Both are vastly superior to full-thickness PK for endothelial disease.
Corneal Ulcer / Infectious Keratitis
Microbial keratitis is an ophthalmic emergency. Contact lens wear (especially overnight/extended wear) is the leading risk factor in developed countries.
Bacterial: Most common. Pseudomonas aeruginosa (contact lens wearers — rapidly progressive, green discharge), Staphylococcus aureus (most common overall), Streptococcus pneumoniae (aggressive, hypopyon). Treat with empiric fortified antibiotics (cefazolin + tobramycin or fortified vancomycin + fortified tobramycin) for sight-threatening ulcers, or fluoroquinolone monotherapy (moxifloxacin) for smaller peripheral ulcers.
Fungal: Suspect with vegetative/organic matter trauma (branch, thorn), agricultural workers, tropical climates. Filamentary fungi (Fusarium, Aspergillus) show feathery infiltrate with satellite lesions and elevated edges. Yeast (Candida) in immunocompromised patients. Diagnosis: corneal scraping with KOH prep, culture on Sabouraud agar. Treatment: natamycin 5% (first-line for filamentary), voriconazole (oral or topical) for resistant cases. Intrastromal voriconazole injection for deep infections.
Acanthamoeba: Almost exclusively in contact lens wearers (especially those who swim/shower with lenses or use tap water). Severe pain disproportionate to examination findings. Ring infiltrate (pathognomonic but late). Radial keratoneuritis (perineural infiltrate) is an early and characteristic sign. Diagnosis: confocal microscopy, culture on non-nutrient agar with E. coli overlay. Treatment: polyhexamethylene biguanide (PHMB) or chlorhexidine + propamidine (Brolene), for months. High recurrence risk.
HSV keratitis: Most common infectious cause of corneal blindness in developed countries. Dendritic ulcer (branching pattern with terminal bulbs, stains with fluorescein and rose bengal) is pathognomonic. Stromal keratitis (immune-mediated) causes scarring. Treatment: topical ganciclovir or trifluridine for epithelial disease; oral acyclovir/valacyclovir for stromal disease + topical corticosteroids (never steroids alone). The HEDS trials established oral acyclovir for prophylaxis of recurrent HSV keratitis.
Corneal Transplantation
Penetrating keratoplasty (PK): Full-thickness corneal replacement. Indicated for conditions affecting all corneal layers (advanced keratoconus with scarring, failed previous grafts, severe corneal scarring). Rejection rate ~20% at 10 years. Rejection signs: redness, decreased vision, corneal edema, keratic precipitates on the graft endothelium. Khodadoust rejection line (advancing line of endothelial rejection) is a classic finding. Treatment: intensive topical steroids.
DALK (deep anterior lamellar keratoplasty): Replaces all layers except Descemet membrane and endothelium. Preferred for keratoconus and stromal dystrophies — preserves recipient endothelium, lower rejection risk, no risk of endothelial rejection.
Endothelial keratoplasty (DMEK/DSAEK): As described in Fuchs section. Now accounts for the majority of corneal transplants in developed countries.
06 Refractive Surgery
LASIK
Laser in situ keratomileusis (LASIK) is the most commonly performed refractive surgery worldwide. A flap (typically 90-120 μm) is created with a femtosecond laser (or microkeratome), reflected, and the excimer laser (193 nm, argon fluoride) ablates the exposed stromal bed. The flap is repositioned without sutures. Corrections: myopia up to ~-10 D (tissue-dependent), hyperopia up to +4 D, astigmatism up to 5 D. Requires minimum residual stromal bed thickness of ≥ 250 μm to prevent ectasia.
Candidacy criteria: age ≥ 18 (ideally ≥ 21 for stable refraction), stable refraction for ≥ 1 year, adequate corneal thickness (≥ 480 μm typically), no keratoconus or ectasia risk (screening with corneal topography/tomography), no severe dry eye, no autoimmune disease, no pregnancy/nursing.
Complications: dry eye (most common — due to corneal nerve transection by the flap; usually transient), flap complications (free cap, buttonhole, wrinkles/striae, epithelial ingrowth), diffuse lamellar keratitis (DLK — "Sands of the Sahara," sterile inflammation at the flap interface, treated with steroids), ectasia (rare but devastating — progressive corneal thinning and protrusion post-LASIK, treated like keratoconus), over/undercorrection, halos/glare, and regression.
PRK & SMILE
PRK removes the corneal epithelium (mechanically, with alcohol, or with excimer laser — "transepithelial PRK"), then ablates the surface stroma directly. Avoids flap risks, better biomechanical strength. Longer recovery (3-5 days of pain, 1-3 months for vision stabilization). Mitomycin C (0.02%) applied briefly to prevent haze. Preferred for patients with thin corneas, high-risk occupations (military, contact sports), or ectasia risk.
SMILE (small incision lenticule extraction): Femtosecond laser creates an intrastromal lenticule that is dissected and removed through a 2-4 mm peripheral incision. No flap, no excimer laser. Advantages: less corneal nerve disruption (less dry eye), better biomechanics. Currently FDA-approved for myopia and astigmatism correction.
Refractive Surgery Complications — Comparison
| Complication | LASIK | PRK | SMILE |
|---|---|---|---|
| Dry eye | +++ (corneal nerve transection by flap) | ++ (surface nerve damage) | + (less nerve disruption) |
| Flap complications | Yes (striae, DLK, epithelial ingrowth, traumatic flap dislocation) | N/A (no flap) | N/A (no flap) |
| Corneal haze | Rare | Risk if no MMC applied | Rare |
| Ectasia risk | Highest (biomechanical weakening from flap) | Lower | Lowest (strongest residual cornea) |
| Pain/recovery | Minimal pain, rapid recovery (24 hrs) | Significant pain (3-5 days), slow visual recovery (1-3 months) | Mild discomfort, intermediate recovery |
| Enhancement options | Flap re-lift or surface ablation | Repeat surface ablation | Surface ablation (no re-treatment through same incision) |
Phakic IOLs
The Visian ICL (Implantable Collamer Lens) is a posterior-chamber phakic IOL placed between the iris and the crystalline lens. Indicated for high myopia (-3 to -20 D) or hyperopia where LASIK/PRK are not feasible. Advantages: reversible, excellent optical quality, no corneal tissue removal. Risks: cataract formation (if ICL vaults too low and touches the lens), pigment dispersion, IOP elevation, endophthalmitis. Modern ICLs (V4c) have a central port (KS-AquaPORT) eliminating the need for a peripheral iridotomy.
07 Anterior Uveitis
Classification & Etiology
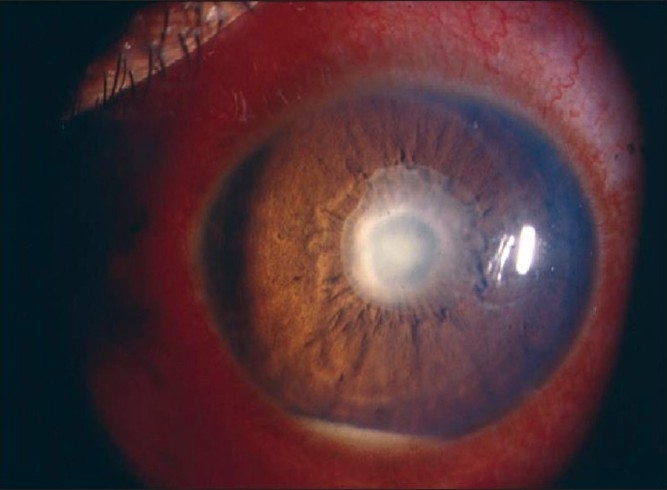
Anterior uveitis (iritis/iridocyclitis) is the most common form of uveitis (~50-60% of all uveitis). Classified by the SUN Working Group criteria based on onset (sudden vs. insidious), duration (limited ≤ 3 months vs. persistent > 3 months), and course (acute, recurrent, chronic).
Non-granulomatous: Fine, white keratic precipitates (KPs) on the corneal endothelium, diffuse anterior chamber cells and flare, no iris nodules. Associated with HLA-B27 (ankylosing spondylitis, reactive arthritis, inflammatory bowel disease, psoriatic arthritis), JIA (juvenile idiopathic arthritis — in girls, ANA-positive), and idiopathic causes. HLA-B27 uveitis is typically acute, unilateral, recurrent, and fibrinous (fibrin in the anterior chamber).
Granulomatous: Large, greasy "mutton-fat" KPs, iris nodules (Koeppe nodules at the pupillary margin, Busacca nodules in the iris stroma), posterior synechiae (iris adhered to the lens). Associated with sarcoidosis (most common), tuberculosis, syphilis, Vogt-Koyanagi-Harada syndrome, and sympathetic ophthalmia (bilateral granulomatous panuveitis after penetrating trauma to the fellow eye).
Workup
First episode of mild, unilateral, acute anterior uveitis in a young adult with HLA-B27 history: no workup needed. Recurrent, bilateral, granulomatous, or chronic anterior uveitis requires investigation: HLA-B27, chest X-ray (sarcoidosis, TB), ACE level and lysozyme (sarcoidosis), RPR/VDRL and FTA-ABS (syphilis — the "great masquerader"), QuantiFERON-TB Gold or PPD (tuberculosis), and CBC, CMP. In children: ANA (JIA-associated uveitis).
Treatment
First-line: topical corticosteroids (prednisolone acetate 1% — most commonly used, start hourly and taper gradually) + cycloplegics (cyclopentolate 1% or atropine 1%) to reduce pain from ciliary spasm, prevent/break posterior synechiae, and stabilize the blood-aqueous barrier. Chronic or steroid-dependent uveitis may require steroid-sparing immunosuppression: methotrexate, mycophenolate, azathioprine, or biologic agents (adalimumab — the only FDA-approved biologic for non-infectious intermediate, posterior, and panuveitis). Periocular or intravitreal steroid injections for refractory cases.
08 Dry Eye Disease & Ocular Surface
Pathophysiology & Classification
Dry eye disease (DED) is a multifactorial disease of the ocular surface characterized by loss of homeostasis of the tear film, causing ocular symptoms and visual disturbance, with tear film instability, hyperosmolarity, inflammation, and neurosensory abnormalities. The DEWS II classification (TFOS International Dry Eye Workshop) categorizes DED into: aqueous-deficient (reduced tear production — Sjogren syndrome, age-related lacrimal gland atrophy, medications such as antihistamines/anticholinergics), evaporative (increased tear evaporation — meibomian gland dysfunction is the leading cause, blepharitis, incomplete lid closure, reduced blink rate with screen use), and mixed (both mechanisms, most common). Tear film breakup time (TBUT) < 10 seconds is abnormal. Schirmer test < 5 mm in 5 minutes suggests aqueous deficiency.
Meibomian Gland Dysfunction
Meibomian gland dysfunction (MGD) is the most common cause of evaporative dry eye and affects up to 70% of DED patients. The meibomian glands (20-30 per lid in the tarsal plate) secrete meibum, the lipid component of the tear film that reduces evaporation. MGD causes thickened, turbid secretions that obstruct gland orifices, leading to gland dropout (visible on meibography), tear film instability, and ocular surface inflammation.
Treatment Ladder
Step 1 (Mild): Patient education (blink exercises, screen breaks — 20-20-20 rule), environmental modification (humidifier, avoid direct fan/air), lid hygiene (warm compresses, lid scrubs for MGD), artificial tears (preservative-free for > 4x/day use), dietary omega-3 fatty acid supplementation.
Step 2 (Moderate): Topical anti-inflammatory agents — cyclosporine 0.05% (Restasis) or cyclosporine 0.09% (Cequa) or lifitegrast 5% (Xiidra) — both take 4-12 weeks for full effect. Short pulse of topical corticosteroid (loteprednol 0.5%) to bridge. Punctal plugs (collagen temporary or silicone permanent) to retain tears. Moisture chamber spectacles.
Step 3 (Severe): Autologous serum tears (20-50%), varenicline nasal spray (Tyrvaya) — stimulates trigeminal nerve to increase tear production. Therapeutic contact lenses (bandage or scleral lenses — vault over the cornea creating a fluid reservoir, highly effective for severe DED). In-office MGD treatments: thermal pulsation (LipiFlow), intense pulsed light (IPL).
Step 4 (Refractory): Topical tacrolimus, systemic immunosuppression (for Sjogren syndrome), tarsorrhaphy (partial lid closure), amniotic membrane transplantation, salivary gland transplantation (Boston KPro in extreme cases).
09 Ocular Surface Tumors
Allergic Eye Disease
Seasonal/perennial allergic conjunctivitis: The most common form — IgE-mediated type I hypersensitivity. Bilateral itching (hallmark symptom), watery discharge, chemosis, papillary reaction. Treatment: avoidance of allergens, artificial tears, topical antihistamine/mast cell stabilizer (olopatadine 0.1-0.7%, alcaftadine 0.25% — dual-action agents are first-line), cool compresses. Oral antihistamines help but can worsen dry eye.
Vernal keratoconjunctivitis (VKC): Chronic, bilateral, severe allergic inflammation affecting young males (5-15 years) in warm climates. Giant cobblestone papillae on the upper tarsal conjunctiva (pathognomonic), Horner-Trantas dots (limbal collections of eosinophils), shield ulcer (sterile corneal ulcer from mechanical abrasion by giant papillae and toxic mediators — can scar and cause amblyopia). Treatment: topical mast cell stabilizers (cromolyn, lodoxamide), topical cyclosporine 0.05-2% (steroid-sparing), short courses of topical steroids for acute flares (monitor IOP — children are especially susceptible to steroid-induced glaucoma), and tacrolimus ointment 0.03% for refractory cases.
Atopic keratoconjunctivitis (AKC): Chronic bilateral ocular inflammation associated with atopic dermatitis. Unlike VKC, affects adults (20-50 years) and is perennial. Cicatrizing (scarring) conjunctivitis — can cause symblepharon (adhesion of palpebral to bulbar conjunctiva), fornix shortening, and keratinization. Associated with keratoconus, PSC cataract, and herpes simplex keratitis. Treatment: lid hygiene, topical antihistamine/mast cell stabilizer, topical cyclosporine/tacrolimus, systemic immunomodulation for severe disease (dupilumab — anti-IL-4/13 biologic approved for atopic dermatitis — may also benefit AKC).
Pterygium
Pterygium is a fibrovascular growth of degenerative conjunctival tissue that extends onto the cornea, typically from the nasal side. Associated with UV exposure, dry/windy climates ("surfer's eye"). Indications for excision: encroachment on the visual axis, significant induced astigmatism, motility restriction, or cosmetic concern. Recurrence rate with bare sclera excision is ~50-80%; conjunctival autograft reduces recurrence to ~5-10%. Mitomycin C (0.02%) or amniotic membrane graft may also reduce recurrence.
Conjunctivitis — Differential Diagnosis
Conjunctivitis is the most common cause of a red eye. Key differentiating features:
| Feature | Viral | Bacterial | Allergic | Chlamydial |
|---|---|---|---|---|
| Discharge | Watery, serous | Mucopurulent, thick | Watery, stringy mucus | Mucopurulent |
| Itching | Minimal | Minimal | Prominent (hallmark) | Minimal |
| Laterality | Starts unilateral, becomes bilateral | Often unilateral initially | Bilateral | Unilateral or bilateral |
| Lymph nodes | Preauricular LAD common | Uncommon (except gonococcal) | None | Preauricular LAD |
| Conjunctiva | Follicular reaction | Papillary reaction | Papillary (giant papillae) | Follicular (large) |
| Key organism(s) | Adenovirus (most common) | S. aureus, S. pneumoniae, H. influenzae | IgE-mediated; allergens | C. trachomatis D-K |
| Treatment | Supportive (cold compresses, artificial tears); self-limited 7-14 days | Topical antibiotics (fluoroquinolone or erythromycin ointment) | Antihistamine/mast cell stabilizer drops (olopatadine); cool compresses | Oral azithromycin 1g single dose or doxycycline x 7 days; treat sexual partners |
Hyperacute bacterial conjunctivitis (Neisseria gonorrhoeae) is an ophthalmic emergency: copious purulent discharge, severe chemosis, rapid corneal involvement → perforation if untreated. Treatment: systemic ceftriaxone 1g IM (+ oral azithromycin for possible co-infection with chlamydia), topical saline irrigation, and frequent topical antibiotics. Screen for other STIs. Ophthalmia neonatorum: conjunctivitis in the first 28 days of life. Causes by timing: chemical (erythromycin prophylaxis, day 1), gonococcal (days 2-5, hyperacute), chlamydial (days 5-14, most common infectious cause), and other bacteria (variable). Neonatal gonococcal conjunctivitis is a sight-threatening emergency requiring IV/IM ceftriaxone.
Ocular Surface Squamous Neoplasia (OSSN)
OSSN encompasses the spectrum from conjunctival intraepithelial neoplasia (CIN) to invasive squamous cell carcinoma. Risk factors: UV exposure, HPV (types 16, 18), HIV/immunosuppression, xeroderma pigmentosum. Presents as a leukoplakic, gelatinous, or papillomatous lesion, most commonly at the limbus (interpalpebral zone). Diagnosis: incisional or excisional biopsy. Management: excisional biopsy with 3-4 mm margins and cryotherapy to the bed ("no-touch technique"), or topical chemotherapy (interferon alpha-2b — first-line for diffuse/recurrent OSSN, 5-fluorouracil, mitomycin C). Invasive SCC may require orbital exenteration in advanced cases.
Conjunctival Melanoma
Conjunctival melanoma is a rare but life-threatening tumor, comprising ~2% of all ocular malignancies but with significant mortality. Arises from primary acquired melanosis with atypia (PAM, ~75%), de novo (~20%), or pre-existing nevi (~5%). Presents as a pigmented, vascularized, elevated lesion — most commonly at the limbus in the interpalpebral zone. Unlike uveal melanoma (which spreads hematogenously to the liver), conjunctival melanoma spreads via lymphatics. Treatment: wide excisional biopsy with cryotherapy to the margins and base ("no-touch technique" — avoid manipulating the tumor to prevent seeding), adjunctive topical mitomycin C 0.04% or interferon alpha-2b, and/or plaque brachytherapy for recurrent disease. Sentinel lymph node biopsy (preauricular and submandibular nodes) is increasingly used for staging. Local recurrence rate: ~50% at 10 years. Metastasis rate: ~25% at 10 years. Mortality: ~30% at 10 years. BRAF mutations present in ~50% of cases (potential target for systemic therapy with BRAF/MEK inhibitors in metastatic disease, unlike uveal melanoma which is typically GNAQ/GNA11 driven).
10 Open-Angle Glaucoma
Pathophysiology & Diagnosis
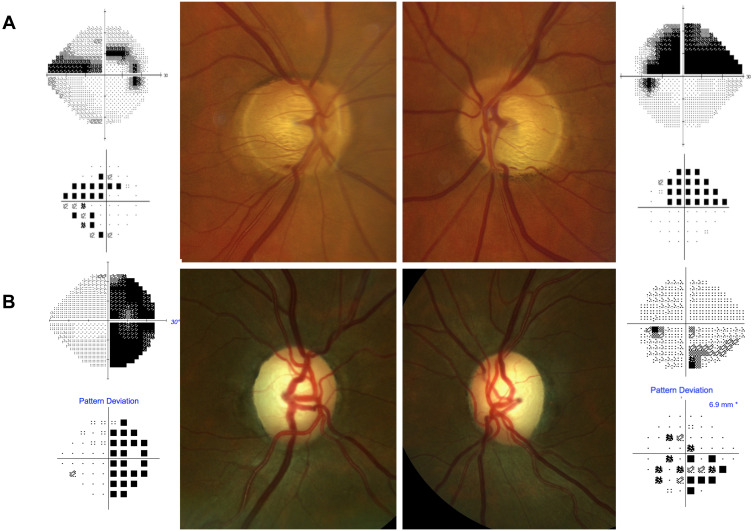
Primary open-angle glaucoma (POAG) is the most common form of glaucoma worldwide and a leading cause of irreversible blindness. It is a chronic, progressive optic neuropathy characterized by retinal ganglion cell death, optic disc cupping, and characteristic visual field loss, with an open and normal-appearing anterior chamber angle. IOP is the only modifiable risk factor. Normal-tension glaucoma (NTG) is a variant where optic neuropathy progresses with IOP consistently ≤ 21 mmHg — more common in Asian populations, may have a vascular component.
Risk factors: elevated IOP (strongest), older age, African ancestry (earlier onset, more aggressive, 6-8x higher prevalence than Caucasians), family history (first-degree relative increases risk 4-9x), myopia, thin central corneal thickness (< 520 μm — OHTS trial showed CCT is an independent risk factor), disc hemorrhages (strong predictor of progression — Drance hemorrhages, seen on the disc rim, often herald focal RNFL loss), and diabetes. Diagnosis is based on the triad of: (1) optic disc changes (increased cup-to-disc ratio — C/D > 0.6 or ≥ 0.2 asymmetry between eyes is especially suspicious, notching/thinning of the neuroretinal rim following the ISNT rule — normal rim thickness: Inferior ≥ Superior ≥ Nasal ≥ Temporal — violation suggests glaucoma, disc hemorrhages, RNFL loss, peripapillary atrophy), (2) characteristic visual field defects (nasal step, arcuate scotoma, paracentral scotoma — following the retinal nerve fiber layer pattern, respecting the horizontal midline), and (3) open angle on gonioscopy.
Ocular hypertension (OHT): IOP > 21 mmHg without optic disc damage or visual field loss. The Ocular Hypertension Treatment Study (OHTS) showed that treating OHT with topical medications reduced the 5-year risk of developing POAG from 9.5% to 4.4%. Risk factors for conversion: higher IOP, thinner CCT, older age, larger C/D ratio, higher PSD on HVF. The OHTS risk calculator helps guide treatment decisions. Not all OHT patients require treatment — many are observed with periodic monitoring.
IOP Target & Medical Therapy
The target IOP is individualized based on baseline IOP, severity of damage, and rate of progression. General guidelines: 20-30% reduction from baseline for early glaucoma; lower targets for advanced disease or rapid progression. The AGIS, CIGTS, and EMGT trials established that IOP lowering reduces progression.
Prostaglandin analogs (PGAs): First-line. Latanoprost, travoprost, bimatoprost, tafluprost (preservative-free). Increase uveoscleral outflow. IOP reduction: 25-33%. Dosed once daily at bedtime. Side effects: conjunctival hyperemia, iris darkening (increased melanin), eyelash growth, periorbital fat atrophy, prostaglandin-associated periorbitopathy (PAPS). Rarely: CME, reactivation of HSV keratitis.
Beta-blockers: Timolol (most common), betaxolol (cardioselective). Decrease aqueous production. IOP reduction: 20-25%. Dosed BID (or once daily for gel-forming timolol). Contraindications: asthma/COPD (non-selective), bradycardia, heart block, depression. Systemic absorption can cause significant side effects — teach nasolacrimal occlusion (punctal occlusion for 2 minutes after drop instillation).
Alpha-2 agonists: Brimonidine. Decrease aqueous production and increase uveoscleral outflow. IOP reduction: 20-25%. Side effects: allergic follicular conjunctivitis (~12-15% at 12 months — most common reason for discontinuation), fatigue/somnolence. Contraindicated in children < 2 years (crosses blood-brain barrier — apnea, hypotension, CNS depression).
Carbonic anhydrase inhibitors (CAIs): Topical: dorzolamide, brinzolamide. Oral: acetazolamide, methazolamide. Decrease aqueous production. Topical IOP reduction: 15-20%. Oral CAIs are more effective but have significant side effects (paresthesias, metallic taste, metabolic acidosis, aplastic anemia — rare, kidney stones). Contraindicated with sulfa allergy (cross-reactivity risk — debated).
Rho-kinase inhibitors: Netarsudil (Rhopressa). Increase trabecular outflow, decrease aqueous production, decrease episcleral venous pressure (the only class to do all three). IOP reduction: ~20%. Side effects: conjunctival hyperemia (most common), cornea verticillata (whorl-like deposits). Fixed combination: netarsudil/latanoprost (Rocklatan) — most potent single-bottle IOP lowering (~30-36%).
Cholinergic (miotic) agents: Pilocarpine. Contracts ciliary muscle, opens trabecular meshwork. Rarely used as chronic therapy (miosis, brow ache, myopic shift, retinal detachment risk). Used acutely for angle-closure attack and in some MIGS procedures. Low-dose pilocarpine (Vuity 1.25%) is FDA-approved for presbyopia — constricts the pupil to create a pinhole effect, improving near vision for 6 hours.
Latanoprostene bunod (Vyzulta): A nitric oxide-donating PGA — combines PGA mechanism (uveoscleral outflow) with nitric oxide-mediated relaxation of TM cells, offering dual outflow enhancement. IOP reduction: ~30-33%. Once-daily dosing. Represents a newer approach to dual-mechanism IOP lowering.
Laser Treatment
Selective laser trabeculoplasty (SLT): 532 nm Q-switched Nd:YAG laser applied to the trabecular meshwork. Stimulates cellular remodeling and improved aqueous outflow without thermal damage. IOP reduction: ~20-25%. Can be first-line treatment (LiGHT trial showed SLT as initial therapy was superior to eye drops at 3 years for maintaining target IOP). Repeatable. Side effects: transient IOP spike, anterior chamber inflammation. Argon laser trabeculoplasty (ALT): Older technique with thermal burns to the TM; not repeatable; largely replaced by SLT.
Fixed-Combination Glaucoma Drops
| Combination | Brand Name | Components | Dosing |
|---|---|---|---|
| Timolol + dorzolamide | Cosopt | Beta-blocker + CAI | BID |
| Timolol + brimonidine | Combigan | Beta-blocker + alpha agonist | BID |
| Timolol + latanoprost | Xalacom | Beta-blocker + PGA | Daily |
| Brimonidine + brinzolamide | Simbrinza | Alpha agonist + CAI | TID |
| Netarsudil + latanoprost | Rocklatan | Rho-kinase inhibitor + PGA | Daily (PM) |
Fixed-combination drops improve adherence by reducing the number of bottles and total drops per day (fewer drops means less exposure to preservatives, particularly benzalkonium chloride — BAK — which is toxic to the corneal surface and contributes to dry eye and ocular surface disease in glaucoma patients on chronic multi-drop therapy). Preservative-free formulations are increasingly available and preferred for patients on long-term therapy.
MIGS (Micro-Invasive Glaucoma Surgery)
MIGS encompasses a group of procedures that lower IOP with minimal tissue disruption, performed through a clear corneal incision (often combined with cataract surgery). IOP reduction is generally modest (~20-30%), making MIGS appropriate for mild-moderate glaucoma.
Trabecular bypass: iStent inject W (two titanium stents placed into Schlemm canal), Hydrus Microstent (8 mm scaffold in Schlemm canal spanning 90 degrees). Goniotomy/trabeculotomy: Kahook Dual Blade, gonioscopy-assisted transluminal trabeculotomy (GATT) — excises or incises trabecular meshwork. Subconjunctival filtration: XEN gel stent (6 mm gelatin tube creating a subconjunctival bleb — essentially a mini-trabeculectomy). Suprachoroidal: iStent Supra (currently limited availability).
Trabeculectomy & Tube Shunts
Trabeculectomy remains the gold standard filtration surgery for glaucoma. A partial-thickness scleral flap is created, a fistula is made into the anterior chamber, and aqueous drains under the conjunctiva forming a filtering bleb. Antimetabolites (mitomycin C 0.2-0.4 mg/mL or 5-FU) are applied to prevent scarring. IOP reduction: 30-50%. Complications: hypotony (over-filtration), bleb leak, bleb infection (blebitis → endophthalmitis), choroidal detachment/effusion, suprachoroidal hemorrhage, cataract progression, and failure due to scarring.
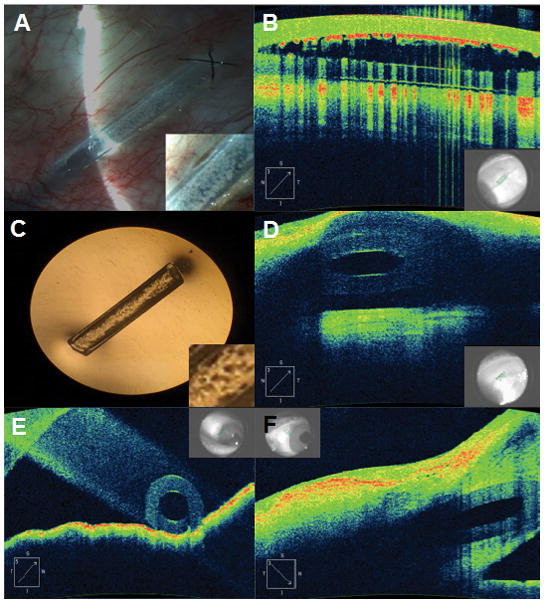
Glaucoma drainage devices (tube shunts): Used for refractory or high-risk glaucoma (failed trabeculectomy, neovascular glaucoma, uveitic glaucoma). Ahmed valve (valved — restricts flow to prevent early hypotony; plate area 184 mm²), Baerveldt implant (non-valved — larger plate 250-350 mm², requires temporary occlusion with a suture to prevent hypotony until the capsule forms ~4-6 weeks). The TVT trial showed Baerveldt tubes had a higher success rate than trabeculectomy with MMC at 5 years in eyes with prior failed trabeculectomy or cataract surgery.
11 Angle-Closure Glaucoma
Acute Angle-Closure Attack (EMERGENCY)
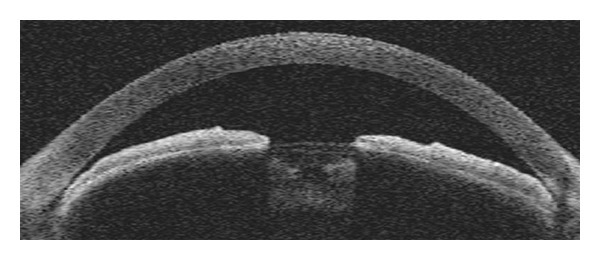
Acute primary angle closure (APAC) is an ophthalmic emergency. The iris apposes the trabecular meshwork, blocking aqueous outflow and causing a rapid, dramatic IOP rise (often 40-70+ mmHg). Presentation: severe eye pain, headache, nausea/vomiting, blurred vision, halos around lights, fixed mid-dilated pupil, corneal edema, shallow anterior chamber, conjunctival injection. Risk factors: hyperopia (short eye, shallow AC), female sex, older age, Asian or Inuit ethnicity, family history, dim lighting or pharmacologic mydriasis (precipitants).
Step 1 — Lower IOP immediately: Topical timolol 0.5% (one drop), topical brimonidine 0.2% (one drop), topical pilocarpine 2% (constricts pupil — but ONLY effective once IOP starts decreasing; a sphincter paralyzed by extreme IOP will not respond). Oral or IV acetazolamide 500 mg. IV mannitol 1-2 g/kg if IOP remains critically elevated.
Step 2 — Topical prednisolone acetate 1% every 15-30 minutes to reduce intraocular inflammation.
Step 3 — Definitive treatment: Laser peripheral iridotomy (LPI) once corneal edema clears (may require topical glycerin to temporarily clear cornea). LPI creates a full-thickness hole in the peripheral iris, allowing aqueous to bypass the pupillary block. Treat both eyes (the fellow eye has a ~50% risk of acute attack without prophylactic LPI).
Step 4 — Consider lens extraction if LPI fails or as primary treatment. The EAGLE trial showed that clear lens extraction was superior to LPI for primary angle closure and primary angle-closure glaucoma in terms of IOP control and quality of life at 3 years.
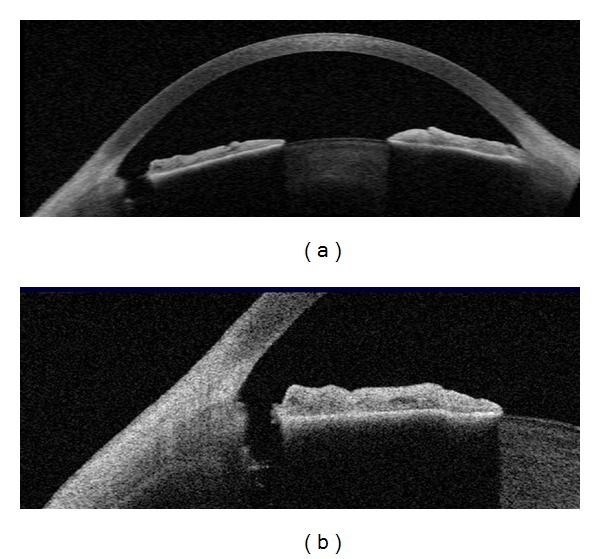
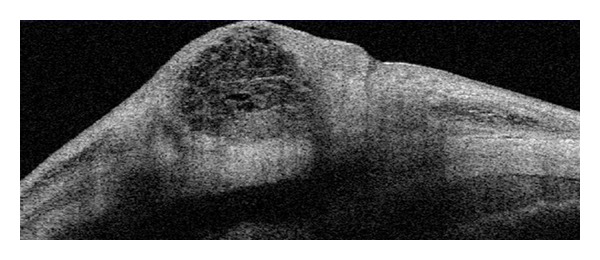
Plateau Iris Syndrome
Plateau iris describes an anatomic configuration where the ciliary body is anteriorly positioned, pushing the iris root forward and causing appositional angle closure despite a patent iridotomy. Diagnosed by ultrasound biomicroscopy (UBM) or anterior segment OCT. Presents in younger patients (30s-40s) with a flat iris plane (unlike the convex iris of pupillary block). Treatment: chronic pilocarpine, argon laser peripheral iridoplasty (ALPI — shrinks the peripheral iris stroma with contraction burns, opening the angle), or lens extraction.
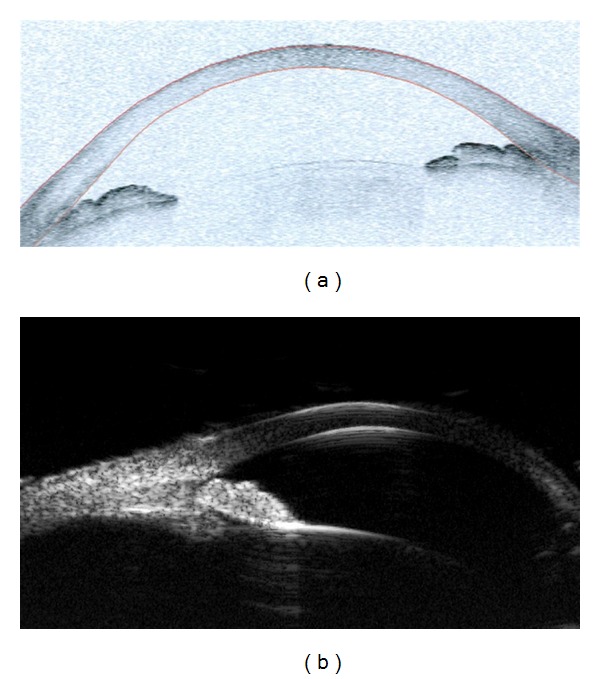
12 Secondary & Pediatric Glaucomas
Neovascular Glaucoma (NVG)
Neovascular glaucoma results from retinal ischemia driving VEGF-mediated neovascularization of the iris (rubeosis iridis) and angle, forming a fibrovascular membrane that obstructs the trabecular meshwork. Causes: proliferative diabetic retinopathy (most common, ~33%), central retinal vein occlusion (~33%), ocular ischemic syndrome (~13%), and CRAO. Management: (1) treat the underlying ischemia — panretinal photocoagulation (PRP) is essential, intravitreal anti-VEGF causes rapid regression of neovascularization (bridge to PRP); (2) lower IOP — medical therapy first, glaucoma drainage device (Ahmed valve preferred for its immediate flow restriction) if needed; trabeculectomy has poor results due to scarring; (3) cycloablation (cyclophotocoagulation) for refractory cases or eyes with poor visual potential.
Pseudoexfoliation Glaucoma
Pseudoexfoliation (PXF) syndrome is characterized by deposition of white, fibrillar extracellular material on the anterior lens capsule (classic "target" or "bull's-eye" pattern with a central disc and peripheral ring separated by a clear zone created by pupillary rubbing), iris, ciliary body, and trabecular meshwork. Associated with progressive trabecular meshwork obstruction → elevated IOP. PXF also causes zonular weakness (higher risk of zonular dialysis and lens subluxation during cataract surgery, IOL dislocation post-operatively). More common in Scandinavian populations, associated with increased risk of CRVO and cardiovascular disease. Often presents with higher IOP and more advanced disease than POAG at diagnosis. Responds well to SLT.
Pigmentary Glaucoma
Pigment dispersion syndrome occurs when the posterior iris surface rubs against the zonular fibers, liberating pigment granules that deposit in the trabecular meshwork (heavily pigmented TM on gonioscopy), on the corneal endothelium (Krukenberg spindle — vertical pigment deposition), and on the iris (transillumination defects in a spoke-like, mid-peripheral pattern). More common in young myopic males. Exercise and pupillary dilation can precipitate pigment showers with acute IOP spikes. Laser peripheral iridotomy may reduce pigment liberation by eliminating reverse pupillary block.
Steroid-Induced Glaucoma
Corticosteroids (topical, periocular, intravitreal, systemic, and even inhaled/intranasal) can cause IOP elevation by increasing resistance in the trabecular meshwork (altered glycosaminoglycan deposition, decreased phagocytic activity of TM cells). Steroid "responders" (~30-40% of the general population with topical steroids, higher with dexamethasone/prednisolone than with fluorometholone/loteprednol) show IOP rise within 2-6 weeks. Management: discontinue steroid if possible, switch to a "soft" steroid (loteprednol — metabolized quickly in the anterior chamber), add IOP-lowering drops. IOP usually normalizes after steroid discontinuation but can become permanent with prolonged use.
Pediatric Glaucoma
Primary congenital glaucoma (PCG) presents at birth or within the first 3 years. Caused by maldevelopment of the trabecular meshwork (trabeculodysgenesis). Classic triad: epiphora (tearing), photophobia, and blepharospasm. Signs: buphthalmos (enlarged globe — corneal diameter > 12 mm in a newborn is suspicious), Haab striae (horizontal breaks in Descemet membrane from globe stretching — pathognomonic), corneal edema, and elevated IOP. Bilateral in ~65-80%.
Treatment is surgical: goniotomy (incision of the trabecular meshwork under direct gonioscopic visualization — requires a clear cornea) or trabeculotomy (ab-externo approach — can be performed even with a cloudy cornea). Success rate ~80-90% with one or two procedures. If goniotomy/trabeculotomy fails: trabeculectomy with MMC, glaucoma drainage device, or cyclophotocoagulation.
13 Diabetic Retinopathy
Pathophysiology & Staging
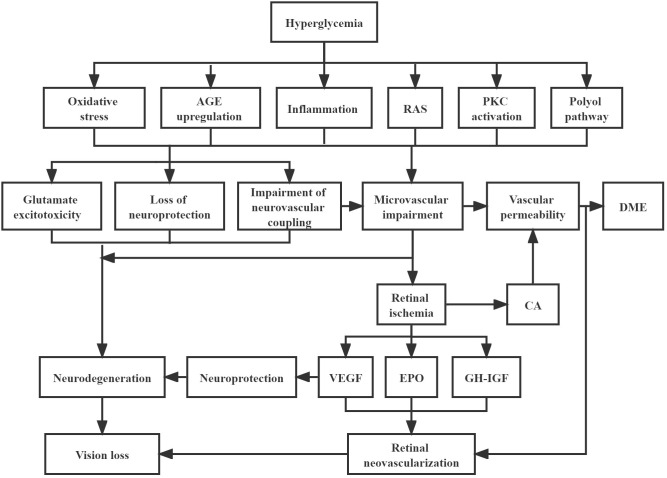
Diabetic retinopathy (DR) is the leading cause of blindness in working-age adults (20-65 years). Results from chronic hyperglycemia-induced damage to retinal capillaries: pericyte loss → basement membrane thickening → endothelial dysfunction → microaneurysm formation → capillary non-perfusion → ischemia → VEGF-driven neovascularization.
No DR: No abnormalities.
Mild NPDR: Microaneurysms only.
Moderate NPDR: Microaneurysms plus any combination of dot-blot hemorrhages, hard exudates, cotton-wool spots, and/or venous beading in fewer than 2 quadrants.
Severe NPDR (4-2-1 rule): Any one of: (1) severe hemorrhages/microaneurysms in all 4 quadrants, (2) venous beading in ≥ 2 quadrants, (3) prominent IRMA (intraretinal microvascular abnormalities) in ≥ 1 quadrant. Approximately 50% progress to PDR within 1 year.
Very severe NPDR: Two or more of the 4-2-1 criteria. ~75% progress to PDR within 1 year.
Proliferative DR (PDR): Neovascularization of the disc (NVD) or elsewhere (NVE), or vitreous/preretinal hemorrhage. High-risk characteristics (DRS): NVD ≥ 1/3 disc area, or any NVD with vitreous hemorrhage, or NVE ≥ 1/2 disc area with vitreous hemorrhage.
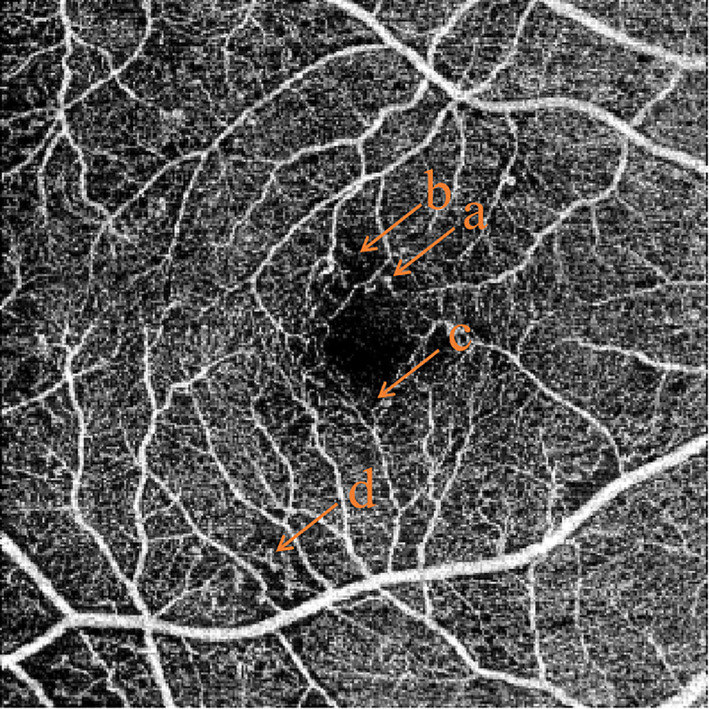
Diabetic Macular Edema (DME)
DME is the most common cause of vision loss in diabetic retinopathy (can occur at any DR stage). Defined as retinal thickening or hard exudates within 1 disc diameter of the foveal center. Center-involving DME (CI-DME) — OCT central subfield thickness > 300 μm with foveal involvement — is the threshold for treatment in most trials. Non-center-involving DME may be observed.
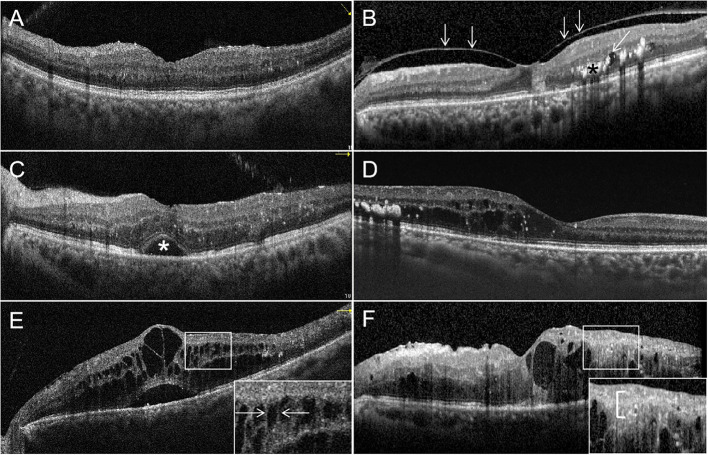
Treatment
Anti-VEGF therapy is the first-line treatment for CI-DME (DRCR.net Protocol T, Protocol I). Agents: aflibercept (Eylea, 2 mg) — most effective at lower baseline VA per Protocol T; ranibizumab (Lucentis, 0.3 mg); bevacizumab (Avastin, 1.25 mg) — off-label, most cost-effective; faricimab (Vabysmo) — bispecific antibody targeting both VEGF-A and Ang-2, longer durability (up to 16-week intervals). Treatment protocol: monthly loading injections (5-6 doses) then treat-and-extend or PRN based on OCT response.
Panretinal photocoagulation (PRP) remains a standard treatment for PDR — 1,200-1,600 spots of laser applied to the peripheral retina to reduce the ischemic stimulus for VEGF production. The DRCR.net Protocol S showed ranibizumab was non-inferior to PRP for PDR at 2 years, with better visual outcomes and less visual field loss, but requires ongoing injections and close follow-up. PRP remains preferred when patient compliance/follow-up is uncertain.
Vitrectomy: Indicated for non-clearing vitreous hemorrhage (typically wait 1-3 months for spontaneous clearing unless NVI/NVG develops), tractional retinal detachment involving or threatening the macula, and combined tractional-rhegmatogenous detachment.
14 Age-Related Macular Degeneration
Classification & Pathogenesis
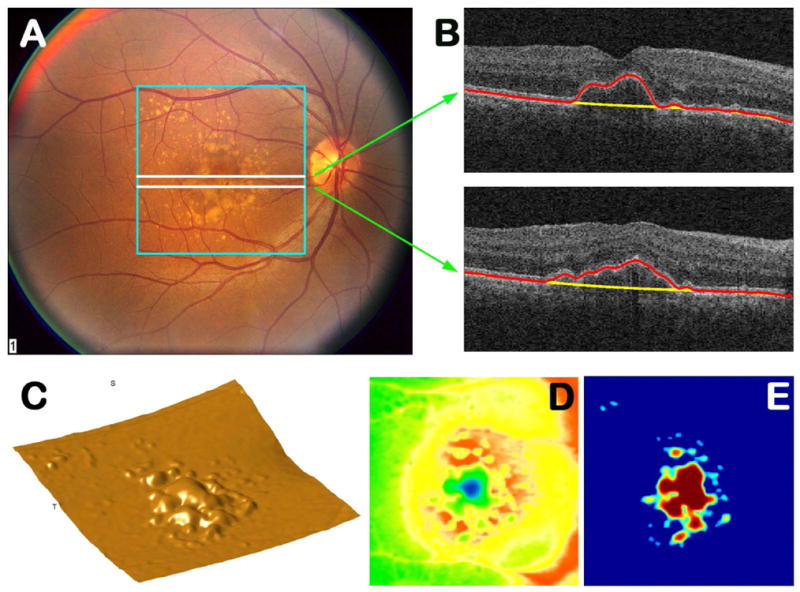
Age-related macular degeneration (AMD) is the leading cause of irreversible central vision loss in the developed world in patients over 50. It involves degeneration of the macula — RPE, Bruch membrane, and photoreceptors.
Dry (non-exudative, atrophic) AMD (~85-90%): Characterized by drusen (yellow deposits of extracellular material between the RPE and Bruch membrane), RPE changes (hyper/hypopigmentation), and in advanced stages, geographic atrophy (GA) — well-demarcated areas of RPE and photoreceptor loss that gradually enlarge. GA causes a slowly progressive, irreversible scotoma. Two recently approved complement inhibitors for GA: pegcetacoplan (Syfovre) — intravitreal anti-C3, and avacincaptad pegol (Izervay) — intravitreal anti-C5. Both slow GA growth by ~20-35% but do not reverse existing atrophy.
Wet (neovascular, exudative) AMD (~10-15%): Characterized by choroidal neovascularization (CNV) — abnormal blood vessels growing from the choroid through Bruch membrane into the sub-RPE or subretinal space. These new vessels are leaky and fragile, causing subretinal fluid, intraretinal fluid, hemorrhage, and ultimately a disciform scar with severe central vision loss if untreated. OCT findings: subretinal fluid (SRF), intraretinal fluid (IRF), sub-RPE fluid (PED — pigment epithelial detachment). Fluorescein angiography shows classic CNV (early hyperfluorescence with late leakage) vs. occult CNV (stippled hyperfluorescence, fibrovascular PED).
AREDS2 Supplementation
The Age-Related Eye Disease Study 2 (AREDS2) formula reduced the risk of progression to advanced AMD by ~25% in patients with intermediate AMD (extensive medium drusen or any large drusen) or advanced AMD in one eye. Formulation: vitamin C (500 mg), vitamin E (400 IU), lutein (10 mg), zeaxanthin (2 mg), zinc oxide (80 mg), and cupric oxide (2 mg). Beta-carotene was removed from AREDS2 due to increased lung cancer risk in smokers.
Anti-VEGF for Wet AMD
Anti-VEGF intravitreal injections are the standard of care for wet AMD and have revolutionized outcomes — prior to anti-VEGF, wet AMD caused rapid, severe, irreversible vision loss; now ~30-40% of treated patients gain ≥ 3 lines of vision. Major trials: ANCHOR/MARINA (ranibizumab — showed vision improvement for the first time in wet AMD), VIEW 1/2 (aflibercept non-inferior to ranibizumab), CATT (ranibizumab vs. bevacizumab — equivalent outcomes, establishing bevacizumab as a cost-effective alternative), TENAYA/LUCERNE (faricimab — comparable efficacy with up to 16-week intervals). Treatment regimens: monthly (clinical trial gold standard), treat-and-extend (most common in practice — extend intervals by 2 weeks if stable, shorten if worsening; reduces visit burden), or PRN (inject only when disease activity detected — risk of undertreatment). Most patients require ongoing treatment for years — stopping injections risks recurrence. Aflibercept 8 mg (Eylea HD) is a higher-dose formulation allowing longer intervals between injections (up to 16 weeks in some patients). Emerging therapies: port delivery system (PDS, Susvimo) — a refillable intraocular implant that continuously delivers ranibizumab (refilled every 6 months); gene therapy approaches to enable the retina to produce its own anti-VEGF protein are in clinical trials.
15 Retinal Detachment
Types
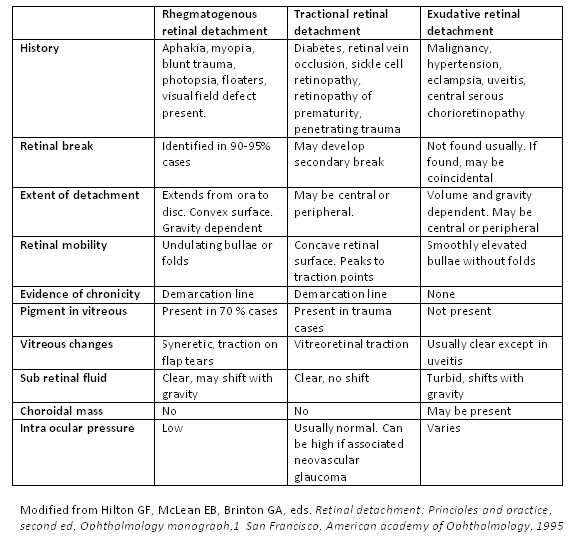
Rhegmatogenous retinal detachment (RRD): Most common type. A full-thickness retinal break (tear or hole) allows liquefied vitreous to pass through and separate the neurosensory retina from the RPE. Risk factors: posterior vitreous detachment (immediate cause — vitreous traction on the retina creates the tear), myopia (longer eye with thinner retina and more vitreous degeneration), lattice degeneration (present in ~8% of the population), prior cataract surgery (aphakia/pseudophakia — 1% lifetime risk), trauma, family history. Symptoms: flashes (photopsia — from vitreoretinal traction), floaters (vitreous hemorrhage or pigment cells — "Shafer sign" or "tobacco dust" on slit lamp), and a progressive curtain or shadow over the visual field.
Tractional retinal detachment (TRD): Fibrovascular proliferation on the retinal surface (from PDR, ROP, PVR, retinal vein occlusion, sickle cell retinopathy) mechanically pulls the retina away from the RPE. No retinal break (unless it becomes a combined tractional-rhegmatogenous detachment, which is a surgical emergency). Typically concave (tented-up) and immobile, unlike the convex, mobile contour of RRD. On OCT: taut, thickened retina with traction peaks. In diabetic TRD, progression is often slow; surgical treatment (pars plana vitrectomy with membrane peeling using delamination, segmentation, or en-bloc techniques) is indicated when the macula is threatened or involved, or if a combined tractional-rhegmatogenous component develops. Preoperative anti-VEGF injection (bevacizumab 1-5 days before surgery) can reduce intraoperative bleeding but must be used cautiously — it can accelerate fibrosis and traction, potentially converting a TRD to a combined detachment, so surgery should not be delayed more than 5-7 days after injection.
Exudative (serous) retinal detachment: Fluid accumulates in the subretinal space without a break or traction. Causes: choroidal tumors (melanoma, metastasis), inflammatory conditions (VKH, posterior scleritis), central serous chorioretinopathy, severe preeclampsia/eclampsia, and hypotony. Shifting fluid: the subretinal fluid shifts with position changes (gravity-dependent), unlike RRD. Treatment targets the underlying cause.
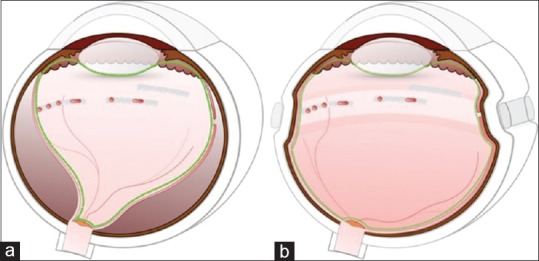
Surgical Management of RRD
Pneumatic retinopexy (PR): Office-based procedure for selected RRDs — superior breaks in the upper 8 clock hours, single or closely grouped breaks, no PVR. Intravitreal gas injection (SF6 or C3F8) + cryotherapy or laser retinopexy to the break. Patient must maintain head positioning to tamponade the break with the gas bubble. Single-operation success rate ~75-80%, but may need additional procedures. PIVOT trial: PR had equivalent visual outcomes to PPV at 1 year for eligible RRDs.
Pars plana vitrectomy (PPV): Most versatile and widely used method. Three-port (23-27 gauge) vitrectomy with removal of vitreous, drainage of subretinal fluid (through the break or retinotomy), endolaser around breaks, and gas or silicone oil tamponade. Indication: most RRDs, especially those with PVR, inferior breaks, giant retinal tears, combined TRD-RRD. Single-operation success rate ~85-90%.
Scleral buckle (SB): External silicone band or sponge sutured to the sclera to indent the wall toward the detached retina, closing the break. Combined with cryotherapy and often a drainage procedure. Particularly effective for young, phakic patients with inferior breaks or round holes, and dialyses. Preserves accommodation (avoids cataract formation that vitrectomy accelerates). Single-operation success rate ~85%.
Tamponade Agents
SF6 (sulfur hexafluoride): Expands to 2x volume, lasts ~2 weeks. Used for simple superior RRDs. C3F8 (perfluoropropane): Expands to 4x volume, lasts ~6-8 weeks. Used for complex RRDs requiring longer tamponade. Silicone oil (1000 or 5000 cSt): Long-term tamponade, does not expand, requires a second surgery for removal. Indications: complex RRD with PVR, giant retinal tears, inferior RRDs requiring prolonged tamponade, patients unable to position, single-eyed patients. Complications of silicone oil: cataracts, glaucoma, band keratopathy, emulsification.
16 Retinal Vascular Disease
Central Retinal Vein Occlusion (CRVO)
CRVO is occlusion of the central retinal vein at or posterior to the lamina cribrosa. Presents with sudden painless monocular vision loss. Fundus: widespread retinal hemorrhages in all four quadrants ("blood and thunder" appearance), dilated tortuous veins, disc edema, cotton-wool spots. Risk factors: hypertension (most common), diabetes, glaucoma (increased lamina cribrosa pressure on the vein), hypercoagulable states (especially in patients < 50). Classified as: non-ischemic (perfused) — milder, better prognosis, ~20% convert to ischemic over 3 years; ischemic (non-perfused) — ≥ 10 disc areas of capillary non-perfusion on FA, poor visual prognosis, high risk of neovascularization (~60%) → NVG.
Treatment: anti-VEGF injections for macular edema (CRUISE trial — ranibizumab; COPERNICUS/GALILEO — aflibercept). Intravitreal dexamethasone implant (Ozurdex) is an alternative for patients intolerant of anti-VEGF or with concurrent inflammation. PRP for neovascularization. Monthly monitoring for conversion from non-ischemic to ischemic.
Branch Retinal Vein Occlusion (BRVO)
BRVO is the most common retinal vascular occlusion. Occurs at arteriovenous crossings where the artery and vein share a common adventitial sheath — the artery compresses the vein. Fundus: flame-shaped hemorrhages confined to the distribution of the affected vein (sectoral, not diffuse like CRVO). Treatment of macular edema: anti-VEGF (BRAVO trial) or intravitreal steroid (SCORE trial showed dexamethasone implant comparable to ranibizumab at 6 months). Grid laser photocoagulation (BVOS study) for persistent macular edema not involving the center.
Central Retinal Artery Occlusion (CRAO) — EMERGENCY
CRAO is the ocular equivalent of an acute ischemic stroke — a "retinal stroke." Irreversible retinal damage occurs within 90-100 minutes of complete occlusion. Presents with sudden, painless, profound monocular vision loss (typically CF or worse). Fundus: diffuse retinal pallor/whitening with a cherry-red spot (the fovea appears red because the thin fovea allows underlying choroidal blood to show through while the surrounding edematous retina is opaque), attenuated arterioles, box-carring of blood columns, and possible visible embolus (Hollenhorst plaque = cholesterol; calcific = white; platelet-fibrin = gray).
Acute stroke protocol: CRAO is now classified as an acute ischemic stroke equivalent. AHA/ASA guidelines recommend emergent evaluation including neuroimaging, carotid imaging, and cardiac evaluation (echocardiography, telemetry) — similar workup to cerebral stroke, as the etiology (carotid atherosclerosis, cardioembolism, giant cell arteritis) is the same.
Time-sensitive treatment: If presentation is within 4.5 hours (and retinal artery is confirmed occluded): intra-arterial thrombolysis or IV tPA may be considered at stroke centers, though evidence is limited (EAGLE study). Traditional measures (ocular massage, anterior chamber paracentesis, carbogen inhalation) have limited evidence but may be attempted if immediate stroke center access is unavailable.
Critical rule-out: Giant cell arteritis (GCA) must be excluded in all CRAO patients ≥ 50 years old — check ESR, CRP, CBC (thrombocytosis). If GCA is suspected, start high-dose IV methylprednisolone immediately (before biopsy). GCA-related CRAO carries a high risk of fellow eye involvement and bilateral blindness if untreated.
Central Serous Chorioretinopathy (CSC)
CSC is a condition characterized by serous detachment of the neurosensory retina at the macula due to focal RPE dysfunction allowing choroidal fluid to leak into the subretinal space. Typical patient: young to middle-aged male (male:female ratio 6:1), type-A personality, under psychological stress. Associated with corticosteroid use (systemic, inhaled, topical, or even epidural), pregnancy, Cushing syndrome, and obstructive sleep apnea. Symptoms: blurred or dim vision, micropsia (objects appear smaller), metamorphopsia, central scotoma, and mild hyperopic shift.
Diagnosis: OCT shows well-defined dome-shaped serous neurosensory detachment at the macula, often with a small PED (pigment epithelial detachment). FA shows the classic "ink-blot" or "smokestack" pattern of leakage at the RPE leak point. ICG shows choroidal hyperpermeability. Most cases (~80-90%) resolve spontaneously within 3-4 months with resolution of the subretinal fluid. Treatment for chronic or recurrent CSC (> 4 months, recurrent episodes, or with RPE atrophy): photodynamic therapy (PDT) with verteporfin (half-dose or half-fluence — reduces choroidal hyperpermeability, most effective treatment for chronic CSC), mineralocorticoid receptor antagonists (eplerenone or spironolactone — off-label, mixed evidence), subthreshold micropulse laser. Discontinue any exogenous corticosteroids if possible.
Ocular Ischemic Syndrome (OIS)
OIS results from chronic hypoperfusion of the eye due to severe (≥ 90%) ipsilateral carotid artery stenosis. Presents with dull aching orbital pain, gradual vision loss, prolonged visual recovery after bright light exposure. Findings: dilated but non-tortuous retinal veins (unlike CRVO which has tortuous veins), dot-blot hemorrhages in the mid-periphery, asymmetric diabetic retinopathy (worse in the eye with the carotid stenosis), low IOP (reduced aqueous production from ciliary body ischemia), anterior segment neovascularization (NVI, NVG). Carotid Doppler ultrasound confirms the diagnosis. Treatment: carotid endarterectomy or stenting + PRP for neovascularization. PRP should be performed AFTER revascularization if possible (restoring blood flow to the ischemic retina post-PRP can cause NVG to worsen temporarily).
17 Vitreoretinal Surgery
Pars Plana Vitrectomy (PPV) — Technique & Indications
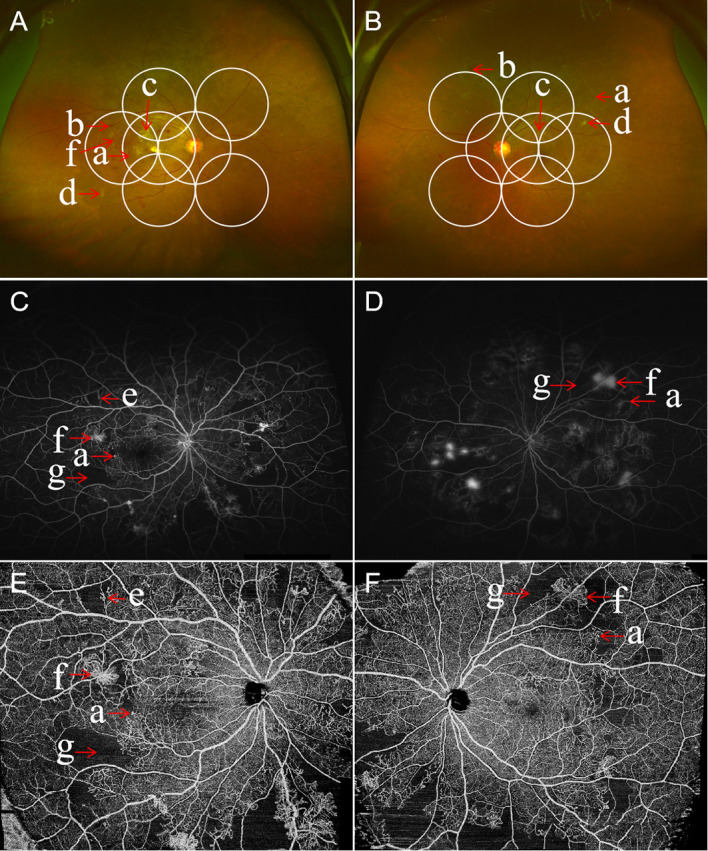
Pars plana vitrectomy is the core vitreoretinal surgical procedure. Three-port (or 2-port valved) entry through the pars plana (~3.5-4 mm posterior to the limbus) using 23-, 25-, or 27-gauge trocars: one for infusion, one for the light source (endoilluminator), and one for the vitrectomy cutter and other instruments. Small-gauge surgery (25G, 27G) allows sutureless wound closure in most cases.
Retinal detachment: RRD repair (most common indication), combined TRD-RRD in PDR.
Vitreous hemorrhage: Non-clearing VH (typically > 1-3 months, sooner if the cause is unknown or NVG develops). Diagnostic vitrectomy if the cause is uncertain.
Epiretinal membrane (ERM): Membranectomy and internal limiting membrane (ILM) peeling for symptomatic ERM causing metamorphopsia or decreased VA.
Macular hole: Vitrectomy with ILM peeling and gas tamponade. Staging (Gass): Stage 1 — impending hole (foveal cyst without full-thickness defect); Stage 2 — small full-thickness hole (≤ 400 μm); Stage 3 — full-thickness hole (> 400 μm) with operculum; Stage 4 — full-thickness hole with complete PVD. Success rate for hole closure: > 90% with primary surgery.
Retained lens fragments: Nuclear material dropped into the vitreous during complicated cataract surgery.
Endophthalmitis: Vitreous tap/biopsy and intravitreal antibiotics; full vitrectomy for severe cases (LP-only vision per EVS).
Diabetic tractional retinal detachment: Membrane peeling/delamination/segmentation to relieve traction.
Proliferative Vitreoretinopathy (PVR)
PVR is the most common cause of failure in retinal detachment surgery. It results from proliferation and contraction of membranes on both surfaces of the detached retina, formed by RPE cells, glial cells, fibroblasts, and macrophages that undergo epithelial-mesenchymal transition. These membranes contract and create fixed retinal folds (starfold configuration is characteristic), re-detaching the retina. Risk factors for PVR: duration and extent of initial RD (most important), vitreous hemorrhage, choroidal detachment, multiple retinal breaks, giant retinal tear, previous failed RD surgery, excessive cryotherapy, and ocular inflammation. Grading (Retina Society classification): Grade A (vitreous haze, pigment clumps), Grade B (wrinkling of inner retinal surface, rolled edges of retinal break), Grade C (full-thickness retinal folds — subclassified by location: anterior or posterior, and extent in clock hours). Treatment: repeat vitrectomy with membrane peeling, perfluorocarbon liquid to flatten the retina, endolaser, and silicone oil tamponade. Pharmacologic adjuncts under investigation: 5-FU, daunorubicin, anti-inflammatory agents.
Complications of Vitrectomy
Cataract formation (most common long-term complication — nuclear sclerosis develops in ~80% of phakic eyes within 2 years of PPV due to increased oxygen exposure to the lens after vitreous removal), retinal detachment (~2-5%), endophthalmitis (~0.03-0.05%), recurrent vitreous hemorrhage, elevated IOP (from expanding gas, silicone oil, or residual viscoelastic), hypotony, suprachoroidal hemorrhage (rare but devastating), phototoxicity from the endoilluminator, and sympathetic ophthalmia (extremely rare — bilateral granulomatous panuveitis after surgical violation of the uveal tract).
18 Posterior Uveitis & Endophthalmitis
Infectious Posterior Uveitis
Toxoplasma retinochoroiditis: The most common cause of infectious posterior uveitis worldwide. Caused by Toxoplasma gondii. Classic presentation: focal necrotizing retinitis appearing as a white lesion adjacent to an old, pigmented chorioretinal scar ("headlight in the fog" — active white lesion with overlying vitritis obscuring the view). Treatment: pyrimethamine + sulfadiazine + folinic acid (to prevent bone marrow suppression from pyrimethamine); or trimethoprim-sulfamethoxazole as an alternative. Corticosteroids added 24-48 hours after starting antimicrobials if there is significant vitritis or macular threat.
CMV retinitis: Occurs almost exclusively in severely immunocompromised patients (CD4 < 50 in HIV/AIDS, solid organ transplant recipients on heavy immunosuppression). "Pizza pie" or "ketchup and cottage cheese" fundus appearance — areas of hemorrhage and necrosis along vascular arcades, often with a granular white border. Without treatment, progresses relentlessly to total retinal destruction. Treatment: IV ganciclovir or valganciclovir (oral) induction then maintenance, intravitreal ganciclovir/foscarnet for sight-threatening lesions. Immune recovery uveitis (IRU) can occur when CD4 count recovers with ART — vitritis causing CME.
Ocular tuberculosis: Can cause choroidal tubercles (small, yellow-white granulomas — classic finding in miliary TB), serpiginous-like choroiditis, retinal vasculitis (occlusive periphlebitis), and optic disc granuloma. Diagnosis often presumptive (positive QuantiFERON or PPD + compatible ocular findings, response to anti-TB therapy). Treatment: standard anti-TB therapy (RIPE) + systemic corticosteroids for intraocular inflammation.
Acute retinal necrosis (ARN): A rapidly progressive viral necrotizing retinitis caused by varicella-zoster virus (most common in older patients), herpes simplex virus (more common in younger patients), or rarely CMV — typically in immunocompetent patients (unlike CMV retinitis, which affects the immunocompromised). Diagnostic criteria (ARA): (1) one or more foci of peripheral retinal necrosis with discrete borders, (2) rapid circumferential progression without antiviral therapy, (3) occlusive vasculopathy with arteriolar involvement, (4) prominent inflammatory reaction (vitritis and anterior chamber inflammation). High risk (~75%) of retinal detachment from necrotic retinal holes. Treatment: IV acyclovir (10 mg/kg q8h for 7-10 days) then oral valacyclovir (1g TID) for 6 weeks to 3 months, intravitreal foscarnet (2.4 mg/0.1 mL) or ganciclovir (2 mg/0.1 mL) for severe cases, prophylactic barrier laser posterior to the active necrosis to reduce RD risk, and low-dose aspirin (antiplatelet for vasculitis). The fellow eye is at risk (~36% without treatment, reduced with oral antivirals).
Behcet disease: A systemic vasculitis characterized by recurrent oral ulcers, genital ulcers, and ocular inflammation. Ocular involvement (~70%) includes bilateral, recurrent, explosive panuveitis with hypopyon (classically shifting, mobile hypopyon), retinal vasculitis (both arteries and veins), and retinal necrosis. Most common cause of uveitis in Turkey, Iran, Japan, and along the Silk Road. HLA-B51 associated. Sight-threatening — requires aggressive immunosuppression: cyclosporine, azathioprine, infliximab, or adalimumab. Interferon-alpha is an alternative. Without treatment, up to 25% of patients become legally blind within 10 years.
Non-infectious Posterior Uveitis
Sarcoidosis: The most common systemic cause of uveitis worldwide. Can cause any pattern of uveitis (anterior, intermediate, posterior, panuveitis) and affects ~25-50% of sarcoidosis patients. International Workshop on Ocular Sarcoidosis (IWOS) criteria define seven intraocular signs suggestive of ocular sarcoidosis: (1) mutton-fat KPs and/or iris nodules, (2) trabecular meshwork nodules and/or tent-shaped peripheral anterior synechiae, (3) snowball/string-of-pearls vitreous opacities, (4) multiple chorioretinal peripheral lesions (active or atrophic), (5) nodular and/or segmental periphlebitis (± candlewax drippings) and/or macroaneurysm, (6) optic disc nodule/granuloma and/or solitary choroidal nodule, (7) bilaterality. Laboratory support: elevated ACE and lysozyme (neither sensitive nor specific alone), elevated serum calcium, chest CT (bilateral hilar lymphadenopathy in ~90% of pulmonary sarcoidosis), gallium-67 scan or FDG-PET (increased uptake in lacrimal glands — "panda sign"; parotid glands — "lambda sign"), and biopsy showing non-caseating granulomas (conjunctival biopsy has ~30% yield even without visible lesions). Treatment: systemic corticosteroids for sight-threatening disease, steroid-sparing agents (methotrexate — first-line steroid-sparing, mycophenolate, adalimumab, infliximab — most effective biologic for refractory ocular sarcoidosis).
Vogt-Koyanagi-Harada (VKH) syndrome: Bilateral granulomatous panuveitis with exudative retinal detachments, affecting melanocyte-containing tissues. Associated findings: poliosis (whitening of eyelashes/eyebrows), vitiligo, alopecia, meningismus, tinnitus, and hearing loss. More common in darkly pigmented individuals (Asian, Hispanic, Native American). Treatment: aggressive systemic corticosteroids (IV pulse then oral taper over ≥ 6 months) to prevent "sunset glow" depigmented fundus and chronic recurrences.
Postoperative Endophthalmitis (EMERGENCY)
Acute postoperative endophthalmitis (typically within 1-7 days of cataract surgery) is an ophthalmic emergency. As detailed in section 4, the EVS guides management. Chronic (delayed) postoperative endophthalmitis presents weeks to months after surgery with mild granulomatous inflammation, often caused by Cutibacterium acnes (trapped in the capsular bag) — a white intracapsular plaque is characteristic. Treatment: intravitreal antibiotics (vancomycin), capsulectomy (removal of the capsular bag and IOL) may be required for refractory cases.
Post-intravitreal injection endophthalmitis: The most dreaded complication of anti-VEGF injections (~0.02-0.05% per injection). Streptococcus species from the oropharynx are a significant cause — hence the recommendation that patients and physicians avoid talking during the injection procedure. Topical povidone-iodine (5%) applied to the conjunctival surface before injection is the single most important prophylactic measure.
19 Optic Neuritis & Papilledema
Optic Neuritis
Optic neuritis is inflammation of the optic nerve, most commonly demyelinating. Typical presentation: acute, unilateral painful vision loss in a young woman (20-40 years), pain on eye movement (92% of cases in the ONTT), RAPD, dyschromatopsia (color vision loss, often red desaturation — out of proportion to VA loss), central or cecocentral scotoma on visual field. The optic disc appears normal in retrobulbar optic neuritis (~65% of cases — "the patient sees nothing and the doctor sees nothing"); disc edema is present in papillitis (~35%).
IV methylprednisolone (250 mg q6h x 3 days) followed by oral prednisone taper: Accelerated visual recovery but did NOT improve final visual outcome at 1 year. Also reduced the 2-year rate of MS development.
Oral prednisone alone: Increased the recurrence rate of optic neuritis — should NOT be used as monotherapy (one of the most famous results in ophthalmology).
Observation: Most patients (95%) recovered to 20/40 or better at 1 year regardless of treatment.
MS risk: Patients with ≥ 1 white matter lesion on baseline brain MRI had a 56% risk of developing MS at 10 years; those with a normal MRI had only a 22% risk. MRI is the most important prognostic test.
Association with multiple sclerosis: optic neuritis is the presenting symptom of MS in ~20% of cases and occurs at some point in ~50% of MS patients. All patients with optic neuritis should have a brain MRI to assess for demyelinating lesions and guide decisions about disease-modifying therapy.
Papilledema & Idiopathic Intracranial Hypertension
Papilledema is optic disc swelling specifically due to elevated intracranial pressure (ICP). Bilateral disc edema with preserved VA early on (unless acute or severe), enlarged blind spots on visual field, transient visual obscurations (brief seconds-long episodes of vision loss with posture changes), headache worse in the morning, pulsatile tinnitus, and CN VI palsy (false localizing sign — from stretching of the long intracranial course). Causes: space-occupying lesion (must be ruled out first with neuroimaging), cerebral venous sinus thrombosis, meningitis, and idiopathic intracranial hypertension (IIH).
Idiopathic intracranial hypertension (IIH, pseudotumor cerebri): Elevated ICP (> 25 cm H2O in adults) with normal brain parenchyma and CSF composition. Classic patient: obese woman of childbearing age. Modified Dandy criteria: symptoms/signs of elevated ICP, papilledema, normal neuroimaging (except empty sella, optic nerve sheath distension, transverse sinus stenosis), elevated opening pressure on LP, normal CSF composition. Treatment: weight loss (5-10% body weight can resolve IIH), acetazolamide (IIHTT trial showed acetazolamide + diet improved visual field, papilledema grade, and quality of life), topiramate (alternative — also promotes weight loss), furosemide. Surgical options for refractory or fulminant cases: optic nerve sheath fenestration (ONSF) — windows cut in the optic nerve sheath to allow CSF drainage, primarily protects vision; CSF diversion — ventriculoperitoneal or lumboperitoneal shunt for headache-predominant symptoms; venous sinus stenting for cases with documented transverse sinus stenosis with a pressure gradient.
20 Cranial Nerve Palsies
Third Nerve Palsy (CN III)
CN III innervates the medial rectus, superior rectus, inferior rectus, inferior oblique, and levator palpebrae superioris, and carries parasympathetic fibers to the pupillary sphincter (constriction) and ciliary muscle (accommodation). Complete CN III palsy: ptosis (complete, as the levator is paralyzed), eye is "down and out" (lateral rectus and superior oblique are unopposed), dilated fixed pupil.
Fourth Nerve Palsy (CN IV)
CN IV (trochlear) innervates the superior oblique muscle. The most common cause of isolated vertical diplopia. Congenital CN IV palsy is the most common congenital cranial nerve palsy (patients develop a head tilt to the opposite side to compensate — look for an old photograph showing the head tilt). Acquired causes: trauma (most common — the trochlear nerve has the longest intracranial course and is vulnerable to head trauma), microvascular (diabetes, hypertension), and rarely tumor or aneurysm. The Parks-Bielschowsky three-step test localizes the paretic muscle: (1) which eye is hypertropic? (2) does the hypertropia worsen in left or right gaze? (3) does it worsen with head tilt to the left or right? A right SO palsy: right hypertropia, worse in left gaze, worse with right head tilt.
Sixth Nerve Palsy (CN VI)
CN VI (abducens) innervates the lateral rectus muscle. Presents with esotropia (inward turning) and horizontal diplopia worse at distance and in the direction of the affected muscle. CN VI has the longest intracranial subarachnoid course, making it vulnerable to elevated ICP (false localizing sign in papilledema/IIH). Causes: microvascular (most common in adults > 50 — resolves in 3-6 months), elevated ICP, MS, trauma, tumor (nasopharyngeal carcinoma, pontine glioma — especially in children; a CN VI palsy in a child requires neuroimaging), Gradenigo syndrome (petrous apicitis from otitis media — CN VI palsy + ipsilateral facial pain from CN V involvement + otorrhea).
Evaluation of Cranial Nerve Palsies
All patients with acute CN palsies require a targeted history and examination. Neuroimaging (MRI brain with contrast + MRA) is indicated for: pupil-involving CN III palsy (emergent), any cranial nerve palsy in a patient < 50 without vascular risk factors, multiple cranial nerve palsies, progressive or non-resolving palsy, associated neurological signs, and pediatric patients. Isolated CN III (pupil-sparing), IV, or VI palsy in a patient > 50 with vasculopathic risk factors may be observed with close follow-up — expected to resolve within 3 months; if not, imaging is required.
Other Neuro-Ophthalmic Conditions
Ischemic optic neuropathy: Anterior ischemic optic neuropathy (AION) presents with sudden, painless, monocular vision loss with an altitudinal (usually inferior) visual field defect and a swollen, pale optic disc (often segmental pallor). Two types: non-arteritic (NAION) — most common; associated with a "disc at risk" (small, crowded optic disc with minimal cupping), nocturnal hypotension (especially in patients on aggressive antihypertensive therapy at bedtime), diabetes, hypertension, sleep apnea. No proven treatment; avoid further IOP-lowering if the disc is at risk. Fellow eye involvement risk: ~15% over 5 years. Arteritic AION (A-AION) — caused by giant cell arteritis (GCA); more severe vision loss, chalky-white disc swelling, associated with headache, jaw claudication, scalp tenderness, polymyalgia rheumatica, elevated ESR (> 50) and CRP. MUST treat immediately with high-dose IV methylprednisolone (1 g/day x 3 days then oral prednisone taper) to prevent bilateral blindness. Temporal artery biopsy confirms diagnosis (skip lesions may cause false negative — biopsy specimen should be ≥ 2 cm; biopsy remains positive for up to 2 weeks after starting steroids).
Nystagmus: Involuntary, rhythmic oscillation of the eyes. Infantile (congenital) nystagmus: typically horizontal, conjugate, present from early infancy; often associated with sensory visual deprivation (albinism, aniridia, optic nerve hypoplasia, Leber congenital amaurosis). Acquired nystagmus requires neurological evaluation — causes include MS (internuclear ophthalmoplegia — INO), brainstem/cerebellar lesions, medications (anticonvulsants, lithium), and vestibular disease. Downbeat nystagmus suggests a craniocervical junction lesion (Chiari malformation). See-saw nystagmus suggests a parasellar mass (craniopharyngioma).
21 Thyroid Eye Disease
Pathophysiology & Clinical Features
Thyroid eye disease (TED, Graves ophthalmopathy) is an autoimmune inflammatory disorder of the orbit, most commonly associated with Graves disease but can occur in hypothyroidism or euthyroid patients. TSH receptor antibodies cross-react with orbital fibroblasts, stimulating glycosaminoglycan production, adipogenesis, and orbital tissue expansion. Bilateral in ~85-95% (may be asymmetric). Features: lid retraction (most common sign — Dalrymple sign), proptosis/exophthalmos (measured by Hertel exophthalmometry; > 20 mm or ≥ 2 mm asymmetry is abnormal), restrictive strabismus (inferior rectus most commonly affected → limited upgaze), periorbital edema, conjunctival injection and chemosis, exposure keratopathy, and in severe cases, compressive optic neuropathy (DON — the most vision-threatening complication, caused by enlarged EOMs at the orbital apex compressing the optic nerve).
Clinical Activity Score (CAS)
One point each for: (1) spontaneous retrobulbar pain, (2) pain on attempted up or downgaze, (3) redness of the eyelids, (4) redness of the conjunctiva, (5) swelling of the eyelids, (6) inflammation of the caruncle and/or plica, (7) chemosis. CAS ≥ 3/7 indicates active disease likely to respond to immunosuppression. CAS < 3 suggests inactive (fibrotic) disease — may require surgical rehabilitation but unlikely to respond to medical therapy.
Management
General measures: Smoking cessation (strongest modifiable risk factor — smokers have 7-8x increased risk of TED, 2x risk of progression after radioactive iodine treatment, and poorer treatment response), thyroid function optimization (avoid both hyper- and hypothyroidism — TSH fluctuations worsen TED), selenium supplementation (100 μg BID for 6 months — EUGOGO trial showed benefit in mild active TED: improved quality of life, reduced CAS, slowed progression), lubricants for exposure keratopathy, prism glasses for diplopia, head-of-bed elevation for periorbital edema, and taping lids at night if lagophthalmos (incomplete lid closure) is present.
Active moderate-severe TED: IV methylprednisolone pulse therapy (EUGOGO protocol: 500 mg/week x 6 weeks, then 250 mg/week x 6 weeks; cumulative dose ≤ 8 g) is first-line for active moderate-severe TED. Teprotumumab (Tepezza) — an IGF-1 receptor inhibitor — is now FDA-approved for TED; it dramatically reduces proptosis (mean 3 mm reduction), diplopia, and CAS. Given as IV infusion every 3 weeks for 8 infusions. Side effects: hearing loss (in 10-20%), hyperglycemia, muscle spasms, infusion reactions. Tocilizumab (anti-IL-6) and mycophenolate are alternatives.
Dysthyroid optic neuropathy (DON): An emergency — treat with high-dose IV methylprednisolone. If no improvement within 1-2 weeks: urgent orbital decompression surgery. Orbital radiation (20 Gy in 10 fractions) is an adjunctive treatment for active TED and DON.
Surgical rehabilitation (for inactive/burned-out disease, in this order): (1) orbital decompression (medial and/or lateral wall removal to allow orbital tissue to expand into the sinuses — reduces proptosis), (2) strabismus surgery (large recession of fibrotic EOMs), (3) eyelid surgery (lid retraction repair, blepharoplasty). This sequence is critical because each stage can affect the next.
22 Orbital Tumors & Trauma
Orbital Cellulitis
Orbital infection is an ophthalmic emergency with risk of vision loss and life-threatening intracranial extension.
Group I — Preseptal (periorbital) cellulitis: Infection anterior to the orbital septum. Lid edema, erythema, warmth. Normal vision, normal motility, no proptosis, no pain with eye movement. Most common in children. Treated with oral antibiotics (amoxicillin-clavulanate). Does NOT threaten vision.
Group II — Orbital cellulitis: Infection posterior to the orbital septum. Proptosis, restricted/painful EOM, chemosis, decreased vision possible. CT shows fat stranding, edema of orbital soft tissues. IV antibiotics (ampicillin-sulbactam or piperacillin-tazobactam or vancomycin + ceftriaxone + metronidazole) and close monitoring.
Group III — Subperiosteal abscess: Collection between the periorbita and orbital wall (most commonly medial wall, from ethmoid sinusitis). Proptosis with displacement of the globe away from the abscess. CT shows well-defined collection. IV antibiotics; surgical drainage if: age > 9, large abscess (> 10 mm), no improvement in 48 hours, suspicion of anaerobic infection, or vision compromise.
Group IV — Orbital abscess: Collection within the orbital soft tissues. Marked proptosis, ophthalmoplegia, decreased vision. Requires urgent surgical drainage + IV antibiotics.
Group V — Cavernous sinus thrombosis: Life-threatening extension into the cavernous sinus. Bilateral orbital signs, CN III/IV/V1/V2/VI involvement, meningeal signs, prostration. IV antibiotics, anticoagulation (controversial), ICU care. Mortality ~30% even with treatment.
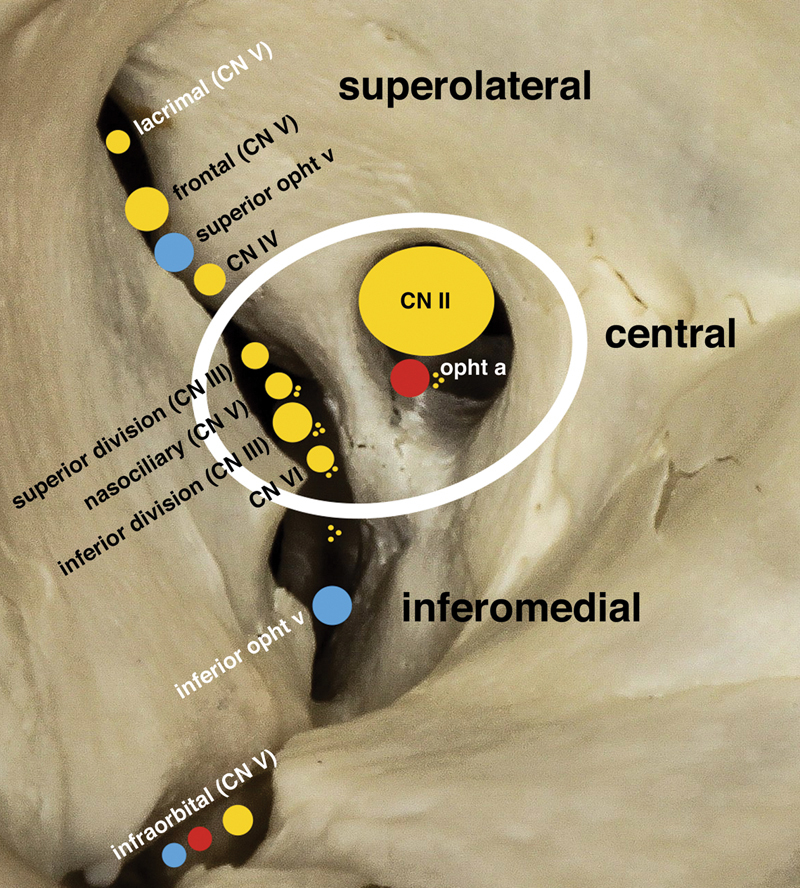
Orbital Fractures
Orbital floor (blow-out) fracture: Most common orbital fracture. Caused by blunt trauma (fist, ball) — hydraulic mechanism (globe transmits force to the thinnest orbital wall). Findings: infraorbital hypoesthesia (V2 nerve runs in the orbital floor), enophthalmos (sunken eye from orbital volume expansion), restricted upgaze (entrapment of the inferior rectus or periorbital tissue in the fracture), and periorbital ecchymosis. CT of the orbits (coronal views) shows the fracture with soft tissue herniation into the maxillary sinus, "teardrop sign." Indications for surgical repair (within 2 weeks): (1) symptomatic diplopia with positive forced duction test (mechanical entrapment), (2) significant enophthalmos (≥ 2 mm), (3) large fracture (> 50% of orbital floor). White-eyed blow-out fracture in children is a surgical emergency — a "trapdoor" fracture entraps the inferior rectus with minimal external signs but causes pain, nausea/vomiting, and restriction; the muscle can become ischemic if not released promptly.
Medial wall fracture: Often accompanies floor fracture. Subcutaneous emphysema (crepitus) from air entering from the ethmoid sinus — instruct the patient not to blow their nose. Rarely requires surgical repair unless symptomatic.
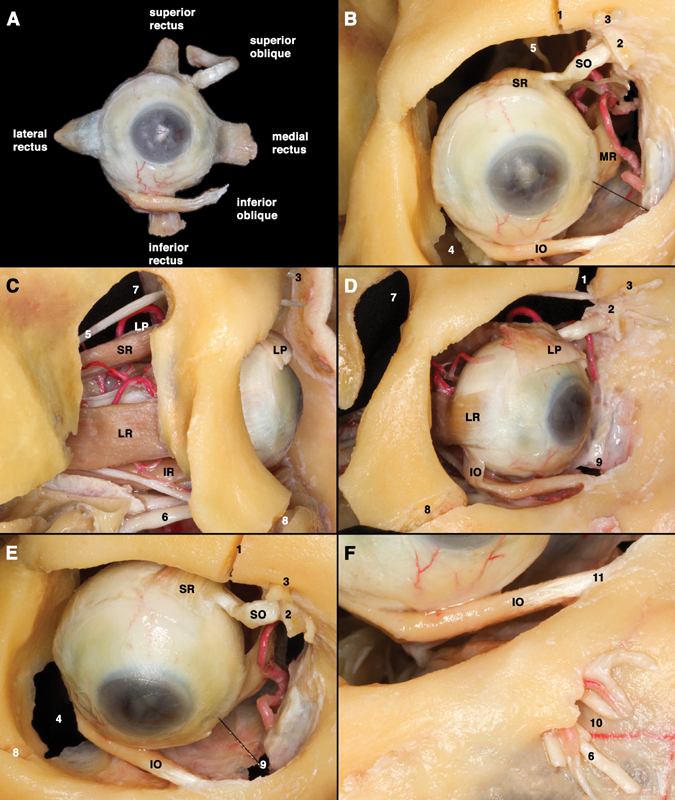
Orbital Tumors
Cavernous hemangioma (cavernous venous malformation): The most common benign orbital tumor in adults. Well-encapsulated, intraconal, slowly progressive proptosis. CT/MRI shows a well-defined, oval, intraconal mass with progressive contrast enhancement. Treatment: observation (if asymptomatic) or surgical excision for compressive optic neuropathy or significant proptosis.
Lymphoma: The most common malignant orbital tumor in adults. Most are low-grade B-cell lymphomas (extranodal marginal zone lymphoma — MALT lymphoma). Presents as a painless, palpable mass, often in the superior orbit or lacrimal gland. "Salmon-pink" appearance on biopsy. Diagnosis requires biopsy with flow cytometry and immunohistochemistry. Treatment: external beam radiation for localized disease; chemotherapy (R-CHOP) if systemic involvement. Systemic workup (CT chest/abdomen/pelvis, bone marrow biopsy) is mandatory because orbital lymphoma can be the presenting sign of systemic disease.
Dermoid cyst: The most common orbital tumor in children. A choristoma (congenitally displaced tissue) containing keratin, hair, and sebaceous material. Most commonly located superotemporally at the frontozygomatic suture. Painless, firm, non-tender mass present from birth. Surgical excision with intact capsule (rupture causes intense granulomatous inflammation).
Ocular Trauma
Open globe injury: A full-thickness wound of the eye wall (cornea or sclera). Types: laceration (caused by a sharp object — includes penetrating injury [single wound], perforating injury [entry + exit wound], and intraocular foreign body) or rupture (blunt force causing the globe to split at the weakest point — often at prior surgical sites, or at the limbus). Signs suggesting open globe: irregular pupil, shallow anterior chamber, positive Seidel test (fluorescein streaming from a wound site under cobalt blue light), low IOP, lens dislocation, vitreous prolapse, and subconjunctival hemorrhage 360 degrees. Management: shield the eye (rigid Fox shield — do NOT patch or apply pressure), administer IV broad-spectrum antibiotics (vancomycin + ceftazidime or levofloxacin), update tetanus, obtain CT orbits (thin-cut axial and coronal — to identify foreign bodies; NEVER MRI if metallic IOFB is suspected), and arrange emergent surgical repair. Primary repair: close the wound in layers (corneal with 10-0 nylon, scleral with 8-0 nylon or vicryl) within 24 hours. Secondary procedures (vitrectomy, IOFB removal, retinal detachment repair, IOL) are performed days to weeks later as needed. Endophthalmitis risk is ~2-7% in open globe injuries (higher with IOFB, especially organic material — Bacillus cereus is a feared organism in post-traumatic endophthalmitis, causing rapid destruction).
Chemical burns: An emergency requiring immediate irrigation — do NOT wait for examination. Alkali burns (lye, ammonia, cement) are worse than acid burns because alkali penetrates deeper (saponification of cell membranes, liquefactive necrosis). Treatment: copious irrigation with normal saline or balanced salt solution for at least 30 minutes; check pH every 15 minutes until normalized (7.0-7.4). The Roper-Hall classification grades severity based on corneal clarity and limbal ischemia (blanching): Grade I (clear cornea, no ischemia — excellent prognosis), Grade II (hazy cornea but iris visible, < 1/3 limbal ischemia — good prognosis), Grade III (corneal epithelial loss, stromal haze obscuring iris, 1/3-1/2 limbal ischemia — guarded), Grade IV (opaque cornea, > 1/2 limbal ischemia — poor prognosis). Ongoing management: topical antibiotics, cycloplegia, citric acid or ascorbic acid (to promote collagen repair), and avoidance of topical steroids after the first 7-10 days (increased risk of corneal perforation from collagenase inhibition). Severe cases may require amniotic membrane transplantation, limbal stem cell transplantation, and ultimately keratoprosthesis (Boston KPro).
Hyphema: Blood in the anterior chamber, usually from trauma (iris root or ciliary body vessel tear). Graded: Grade 1 (< 1/3 AC fill), Grade 2 (1/3-1/2), Grade 3 (> 1/2), Grade 4 (total hyphema — "eight-ball hyphema" when clotted and dark). Management: bed rest with head of bed elevated 30 degrees, rigid eye shield, topical cycloplegia (atropine), topical corticosteroid, avoid aspirin/NSAIDs and anticoagulants. Rebleeding risk is ~5-10% (peaks at days 3-5). Complications: IOP elevation (hyphema blocks TM), corneal blood staining (in prolonged total hyphema with elevated IOP), and optic atrophy from sustained pressure. All patients with hyphema must be tested for sickle cell trait/disease (sickle cells can obstruct the trabecular meshwork at lower IOP thresholds; avoid CAIs which worsen sickling via acidosis; surgical evacuation threshold is lower: IOP > 24 mmHg for > 24 hours in sickle cell patients vs. IOP > 50 for 5 days in others).
23 Oculoplastics
Ptosis
Ptosis (drooping of the upper eyelid) is classified by etiology:
Aponeurotic (involutional): Most common type in adults. Due to dehiscence or disinsertion of the levator aponeurosis from the tarsal plate, often accelerated by contact lens wear, eye rubbing, or previous intraocular surgery. Presents with good levator function (≥ 12 mm), high or absent lid crease, and thinning of the upper lid with a deep superior sulcus. The MRD1 (margin-to-reflex distance 1 — distance from the upper lid margin to the corneal light reflex) is the key measurement: normal ≥ 4 mm; ptosis < 2 mm. Treatment: levator advancement or external levator resection — surgical approach depends on the degree of ptosis and levator function. For ptosis with levator function ≥ 5 mm: external levator resection or Muller muscle-conjunctival resection (MMCR — for ptosis responsive to phenylephrine test). For poor levator function (< 4 mm): frontalis suspension using silicone rod, Gore-Tex, or autogenous fascia lata.
Myogenic: Caused by myopathy affecting the levator muscle. Myasthenia gravis (fatigable ptosis — worse with sustained upgaze, improves with ice test or edrophonium test; anti-AChR antibodies; treatment: pyridostigmine, immunosuppression), myotonic dystrophy, chronic progressive external ophthalmoplegia (CPEO — mitochondrial, bilateral, symmetric).
Neurogenic: CN III palsy (ptosis with motility deficit — "down and out"), Horner syndrome (mild ptosis 1-2 mm + miosis + anhidrosis; preganglionic lesions → lung apex tumor/Pancoast, carotid dissection; postganglionic → cavernous sinus lesion, cluster headache). Pharmacologic testing: cocaine drops (fail to dilate Horner pupil), hydroxyamphetamine (Paredrine) drops (dilate in preganglionic but not postganglionic lesions). Apraclonidine test (reversal of anisocoria — the Horner pupil dilates while the normal pupil constricts — due to denervation supersensitivity) is now the most commonly used clinical test.
Mechanical: Lid mass (chalazion, tumor), edema, scarring. Congenital ptosis is usually due to levator muscle maldevelopment (myogenic); treatment depends on levator function — frontalis sling if levator function < 4 mm.
Ectropion & Entropion
Ectropion (outward turning of the eyelid margin): most commonly involutional (age-related laxity of the lower lid), also cicatricial (skin scarring pulls lid outward), paralytic (CN VII palsy), or mechanical. Symptoms: tearing (exposure of punctum and conjunctiva), exposure keratopathy. Treatment: lateral tarsal strip procedure for involutional ectropion; skin graft for cicatricial.
Entropion (inward turning of the eyelid margin): most commonly involutional (lower lid laxity + overriding of preseptal orbicularis), also cicatricial (trachoma — the leading infectious cause of blindness worldwide, Stevens-Johnson syndrome, OCP), spastic. Symptoms: foreign body sensation, corneal abrasion/ulceration from misdirected lashes (trichiasis). Treatment: quickert sutures (temporary), lateral tarsal strip with retractor reinsertion (definitive for involutional); mucous membrane graft for cicatricial.
Blepharospasm & Hemifacial Spasm
Benign essential blepharospasm (BEB): Bilateral, involuntary, spasmodic contraction of the orbicularis oculi muscles causing forceful, involuntary eye closure. Typically affects women > 50 years. Distinguished from hemifacial spasm (unilateral, involves other facial muscles, caused by CN VII compression — usually by an ectatic blood vessel at the CPA). BEB can be functionally blinding despite normal visual acuity. Treatment: botulinum toxin type A (Botox, Dysport, Xeomin) injection into the orbicularis oculi is first-line and highly effective — repeated every 3-4 months. Side effects: ptosis, lagophthalmos, diplopia (from diffusion to adjacent muscles). Refractory cases: limited myectomy of the orbicularis muscle.
Dacryocystorhinostomy (DCR)
DCR is the definitive treatment for acquired nasolacrimal duct obstruction. Creates a direct communication between the lacrimal sac and the nasal cavity, bypassing the obstructed duct. External DCR (through a skin incision) has a 90-95% success rate. Endoscopic (endonasal) DCR avoids a skin incision with comparable success. A silicone stent is typically placed for 2-3 months. Acute dacryocystitis (infection of the lacrimal sac — painful, erythematous swelling inferior and medial to the medial canthus) requires antibiotics and, once resolved, definitive DCR to prevent recurrence.
Eyelid Malignancies
Basal cell carcinoma (BCC): The most common eyelid malignancy (~90%). Most common on the lower lid (> inner canthus > upper lid > lateral canthus). Pearly, nodular, telangiectatic lesion with central ulceration ("rodent ulcer"); may also be morpheaform (sclerosing — more aggressive, poorly defined borders). Rarely metastasizes but locally destructive. Treatment: excision with frozen section margin control (Mohs micrographic surgery ideal for periocular BCC), oculoplastic reconstruction.
Squamous cell carcinoma (SCC): Second most common (~5%). More aggressive than BCC with potential for perineural invasion and regional metastasis (~2-5%). Scaly, keratinized, erythematous lesion with irregular margins. Risk factors: UV exposure, immunosuppression (organ transplant — 65-250x increased risk), xeroderma pigmentosum, actinic keratosis. Treatment: wide excision with margin control (Mohs); consider sentinel lymph node biopsy for high-risk lesions; adjuvant radiation for perineural invasion.
Sebaceous cell carcinoma: Arises from meibomian glands (most common) or glands of Zeis. The "great masquerader" — mimics chalazion, blepharitis, or chronic unilateral conjunctivitis. Suspect in any recurrent chalazion or chronic unilateral blepharoconjunctivitis unresponsive to treatment. Pagetoid spread (intraepithelial invasion) through the conjunctiva is characteristic. Map biopsies of the conjunctiva are performed to assess extent. Mortality rate ~5-10%. Treatment: wide excision with margin control; exenteration for extensive orbital invasion.
24 Pediatric Ophthalmology & Strabismus
Amblyopia
Amblyopia ("lazy eye") is reduced best-corrected visual acuity in one or both eyes due to abnormal visual development during the critical period (birth to ~7-8 years, with some plasticity up to age 12-13). It is the most common cause of monocular vision loss in children (2-4%). Types: strabismic (misaligned eye is suppressed), refractive (anisometropic — large difference in refractive error between eyes; or bilateral ametropic — bilateral high uncorrected refractive error), and deprivation (obstructed visual axis — congenital cataract, ptosis — most severe form, requires urgent treatment). Treatment: correct the underlying cause (glasses, cataract surgery) + penalization of the better eye (patching 2-6 hours/day or atropine 1% penalization of the better eye on weekends — ATS studies showed equivalent efficacy for moderate amblyopia).
Strabismus
Esotropia (inward deviation): Infantile esotropia — onset before 6 months, large angle (> 30 prism diopters), alternating, cross-fixation pattern (uses right eye to look left and vice versa — may mimic bilateral CN VI palsy). Associated: dissociated vertical deviation (DVD), latent nystagmus, inferior oblique overaction. Treatment: bilateral medial rectus recession at ~6-12 months of age to allow binocular development. Accommodative esotropia — onset 1-3 years, associated with moderate hyperopia (+3 to +6 D); the child accommodates to see clearly and the associated convergence (AC/A ratio) drives the esotropia. Treatment: full hyperopic correction with glasses (full cycloplegic refraction — atropine or cyclopentolate refraction is essential in children). If residual esotropia persists with full correction → high AC/A ratio → consider bifocals (progressive or executive) or surgery. Sensory esotropia — secondary to poor vision in one eye (e.g., cataract, optic nerve hypoplasia) — treat the underlying cause.
Exotropia (outward deviation): Intermittent exotropia — the most common childhood exotropia, presenting typically between ages 2-5 years. The deviation is initially controlled by fusional convergence but manifests with fatigue, inattention, or bright sunlight (patients squint or close one eye in bright light — photophobia reflex disrupts fusional control). Control assessed by the Newcastle control score or Mayo classification. Treatment: observation if well-controlled (especially < 5 years), over-minus lens therapy (stimulates convergence), orthoptic exercises (convergence exercises), prism adaptation, or surgery — lateral rectus recession + medial rectus resection (R&R procedure) for poor control, large deviation, or progressive worsening. Bilateral lateral rectus recession is an alternative approach. Recurrence rate after surgery: 20-40% over 10 years.
Special Considerations in Strabismus
Duane retraction syndrome: A congenital cranial dysinnervation disorder caused by absence of the abducens nucleus with aberrant innervation of the lateral rectus by CN III. Type I (most common, 80%): limited abduction with globe retraction and narrowing of the palpebral fissure on adduction. Type II: limited adduction. Type III: limited both. Usually unilateral (left eye more common), more common in females. Most patients develop a compensatory face turn and do not require surgery unless significant deviation in primary position.
Brown syndrome: Restriction of elevation in adduction due to a tight or inelastic superior oblique tendon (congenital) or inflammation of the SO tendon sheath (acquired — trochleitis, associated with RA, JIA). The eye cannot elevate when adducted but elevates normally in abduction. May be confused with inferior oblique palsy. Congenital Brown syndrome often improves spontaneously. Acquired: treat inflammation with NSAIDs or local steroid injection. Surgical options: SO tenotomy or silicone expander if persistent.
Retinopathy of Prematurity (ROP)
ROP is a vasoproliferative disorder of the retina affecting premature infants. Incomplete retinal vascularization at birth (retinal vessels grow from the optic disc to the ora serrata, completing temporally by ~40 weeks gestational age) leads to peripheral retinal ischemia, VEGF production, and abnormal neovascularization. Screening criteria: birth weight ≤ 1500 g or gestational age ≤ 30 weeks (or select infants 30-36 weeks with unstable course). First exam at 31 weeks postmenstrual age or 4 weeks postnatal age, whichever is later.
Congenital cataract: Lens opacity present at birth or developing in infancy. Causes: idiopathic (most common), hereditary (autosomal dominant most common pattern), intrauterine infection (TORCH — rubella is the classic cause, also CMV, toxoplasmosis, HSV, syphilis), metabolic (galactosemia — oil-droplet nuclear cataract; Lowe syndrome; hypocalcemia — lamellar cataract), and chromosomal (Down syndrome, Turner syndrome). Visually significant unilateral congenital cataracts require urgent surgery (ideally within 4-6 weeks of life) to prevent severe deprivation amblyopia — the most challenging form of amblyopia to treat. Bilateral cataracts can be deferred slightly longer (6-10 weeks) but still require early intervention. Post-surgical management: contact lens correction in infants (IOL implantation is typically deferred until age 1-2 years due to difficulties with IOL power calculation in a rapidly growing eye), aggressive amblyopia treatment with patching.
Classification: Zone (I — posterior, II — midperipheral, III — peripheral), Stage (1 — demarcation line, 2 — ridge, 3 — ridge with extraretinal neovascularization, 4 — partial retinal detachment [4A — fovea attached, 4B — fovea detached], 5 — total retinal detachment), Plus disease (dilated and tortuous retinal vessels — indicates severity). Treatment threshold (ETROP): Type 1 ROP (zone I any stage with plus, zone I stage 3, zone II stage 2 or 3 with plus) → treat. Type 2 ROP → observe with frequent exams. Treatment: laser photocoagulation to avascular retina (gold standard) or intravitreal anti-VEGF (bevacizumab or ranibizumab — especially for zone I disease, but requires prolonged follow-up for late recurrence). Stage 4-5 ROP requires vitrectomy with or without scleral buckle.
Retinoblastoma
Retinoblastoma is the most common intraocular malignancy of childhood, affecting ~1 in 15,000-20,000 live births. Presents most commonly with leukocoria (white pupil reflex — the single most important sign, detected in ~60%) and strabismus (~20%). Mean age at diagnosis: 18 months (unilateral) and 12 months (bilateral). Bilateral in ~30% (germline RB1 mutation on chromosome 13q14 — autosomal dominant with ~95% penetrance — these patients also have increased lifetime risk of secondary malignancies, especially osteosarcoma and soft tissue sarcomas; require lifelong surveillance). Trilateral retinoblastoma refers to bilateral RB with a pinealoblastoma (primitive neuroectodermal tumor of the pineal gland).
Group A: Small tumor (≤ 3 mm) confined to the retina, away from fovea and disc.
Group B: Larger tumor (> 3 mm) or tumor closer to fovea/disc, still confined to retina, no vitreous or subretinal seeding.
Group C: Discrete focal vitreous or subretinal seeding (fine, localized).
Group D: Diffuse vitreous or subretinal seeding (massive "snow" of vitreous seeds or subretinal fluid with seeding).
Group E: Extensive tumor with one or more high-risk features (neovascular glaucoma, vitreous hemorrhage, tumor in anterior chamber, phthisis). Enucleation typically indicated.
Treatment: depends on staging — chemotherapy (systemic intravenous carboplatin/etoposide/vincristine, or intra-arterial — ophthalmic artery chemosurgery with melphalan, a transformative technique that has dramatically increased eye salvage rates), focal consolidation (transpupillary thermotherapy, laser, cryotherapy), intravitreal melphalan for vitreous seeds; enucleation for advanced unilateral disease (Group E); external beam radiation largely abandoned due to secondary malignancy risk in germline RB1 patients. Genetic counseling and RB1 testing are essential for all patients and families. Survival > 95% in developed countries with appropriate treatment.
25 Classification Systems
Cataract Grading — LOCS III
| Feature | Grades | Assessment Method |
|---|---|---|
| Nuclear color (NC) | NC1 (clear) — NC6 (brunescent) | Slit-lamp comparison photographs |
| Nuclear opalescence (NO) | NO1 — NO6 | Slit-lamp retroillumination |
| Cortical cataract (C) | C1 — C5 | Retroillumination, percent area affected |
| Posterior subcapsular (P) | P1 — P5 | Retroillumination, percent area affected |
Diabetic Retinopathy — ETDRS Severity Scale (Summary)
| Level | Findings | Action |
|---|---|---|
| No DR | No abnormalities | Annual screening |
| Mild NPDR | Microaneurysms only | Annual follow-up |
| Moderate NPDR | MAs + HMAs, HEs, CWS, VB in < 2 quadrants | 6-month follow-up |
| Severe NPDR | 4-2-1 rule (any one) | 3-4 month follow-up; consider anti-VEGF or PRP |
| Very severe NPDR | 4-2-1 rule (any two) | Consider PRP or anti-VEGF |
| PDR | NVD, NVE, VH, preretinal hemorrhage | Anti-VEGF and/or PRP; vitrectomy if indicated |
AMD Staging — AREDS Classification
| Category | Findings | Management |
|---|---|---|
| Category 1 (No AMD) | Few or no small drusen (< 63 μm) | Routine eye exam |
| Category 2 (Early AMD) | Multiple small drusen, few medium drusen (63-124 μm), or mild RPE changes | AREDS2 not indicated; observe |
| Category 3 (Intermediate AMD) | Extensive medium drusen, ≥ 1 large druse (≥ 125 μm), or non-central GA | AREDS2 supplements indicated |
| Category 4 (Advanced AMD) | Central GA or neovascular AMD in one eye | AREDS2 for fellow eye; anti-VEGF for wet AMD |
Shaffer Angle Grading
| Grade | Angle Width | Structures Visible | Clinical Significance |
|---|---|---|---|
| Grade 4 | 35-45° | Ciliary body band | Wide open, no risk of closure |
| Grade 3 | 25-35° | Scleral spur | Open, no risk of closure |
| Grade 2 | ~20° | Trabecular meshwork | Narrow, possible closure |
| Grade 1 | ~10° | Only Schwalbe line | Very narrow, high risk of closure |
| Grade 0 | 0° | None (iris against cornea) | Closed |
Chandler Classification of Orbital Cellulitis
| Group | Description | Treatment |
|---|---|---|
| I | Preseptal cellulitis | Oral antibiotics |
| II | Orbital cellulitis | IV antibiotics, monitoring |
| III | Subperiosteal abscess | IV antibiotics ± surgical drainage |
| IV | Orbital abscess | Urgent surgical drainage + IV antibiotics |
| V | Cavernous sinus thrombosis | IV antibiotics, anticoagulation, ICU |
Hodapp-Parrish-Anderson (HPA) Visual Field Classification for Glaucoma
| Severity | Mean Deviation (MD) | Pattern Deviation | Clinical Significance |
|---|---|---|---|
| Early | Better than -6 dB | < 25% of points depressed below 5% level, < 10% below 1% level; no point within 5° of fixation < 15 dB | Mild functional loss; monitor closely |
| Moderate | -6 to -12 dB | 25-50% of points depressed below 5% level, < 20% below 1% level; no point within 5° of fixation at 0 dB; only one hemifield with sensitivity < 15 dB within 5° of fixation | Significant loss; more aggressive IOP targets needed |
| Severe (Advanced) | Worse than -12 dB | > 50% of points depressed below 5% level, > 20% below 1% level; any point within central 5° at 0 dB; points within central 5° < 15 dB in both hemifields | Severe loss; risk of fixation split; consider surgical intervention |
Uveitis — SUN Anterior Chamber Cell Grading
| Grade | Cells per Field (1 mm x 1 mm slit beam) |
|---|---|
| 0 | < 1 |
| 0.5+ | 1-5 |
| 1+ | 6-15 |
| 2+ | 16-25 |
| 3+ | 26-50 |
| 4+ | > 50 |
Roper-Hall Classification of Chemical Burns
| Grade | Corneal Involvement | Limbal Ischemia | Prognosis |
|---|---|---|---|
| I | Corneal epithelial damage, no stromal haze | None | Excellent |
| II | Corneal haze, iris details visible | < 1/3 (120°) | Good |
| III | Total epithelial loss, stromal haze obscuring iris | 1/3-1/2 (120-180°) | Guarded |
| IV | Cornea opaque, iris/pupil not visible | > 1/2 (> 180°) | Poor |
Macular Hole Staging (Gass)
| Stage | Description | Management |
|---|---|---|
| Stage 1A | Yellow foveal spot (foveolar detachment) | Observation (50% resolve spontaneously) |
| Stage 1B | Yellow ring (foveolar detachment with dehiscence) | Observation |
| Stage 2 | Small full-thickness hole (≤ 400 μm) | Vitrectomy + ILM peel + gas tamponade |
| Stage 3 | Full-thickness hole (> 400 μm) + partial PVD | Vitrectomy + ILM peel + gas tamponade |
| Stage 4 | Full-thickness hole + complete PVD | Vitrectomy + ILM peel + gas tamponade |
ROP Classification
| Parameter | Classification |
|---|---|
| Zone | I (posterior — centered on disc, radius 2x disc-to-fovea distance), II (extends to nasal ora), III (remaining temporal crescent) |
| Stage | 1 (demarcation line), 2 (ridge), 3 (extraretinal NV), 4A (partial RD, fovea on), 4B (partial RD, fovea off), 5 (total RD) |
| Plus disease | Dilated and tortuous posterior pole vessels (≥ 2 quadrants) |
| Aggressive ROP (A-ROP) | Rapidly progressive, flat neovascularization, posterior location, prominent plus disease |
Choroidal Melanoma — TNM Staging (AJCC 8th Edition, Simplified)
| Category | Size Criteria | Clinical Features |
|---|---|---|
| T1 (Small) | Thickness ≤ 3 mm and basal diameter ≤ 16 mm | May be observed if no growth (watchful waiting with serial imaging) |
| T2 (Medium) | Thickness 3.1-8 mm or basal diameter > 16 mm | Standard treatment: plaque brachytherapy (I-125 or Ru-106) or proton beam radiation; COMS showed equivalent survival to enucleation |
| T3 (Large) | Thickness 8.1-15 mm | Enucleation usually indicated; plaque brachytherapy or proton beam for selected cases |
| T4 (Very large) | Thickness > 15 mm or extraocular extension | Enucleation ± orbital exenteration if extensive extraocular extension |
The COMS (Collaborative Ocular Melanoma Study) demonstrated that for medium choroidal melanomas, I-125 plaque brachytherapy had equivalent 5-year and 12-year mortality to enucleation, establishing eye-sparing radiation as the standard of care. Metastatic risk: ~30% at 5 years for medium-large tumors, primarily to the liver (> 90% of metastases). Prognostic factors: tumor size (most important), chromosome 3 monosomy (worst prognostic finding), gene expression profiling (Class 1A — low risk; Class 1B — intermediate; Class 2 — high metastatic risk), epithelioid cell type (worse than spindle cell), and extraocular extension. All patients require systemic surveillance with hepatic imaging (abdominal MRI or ultrasound every 6 months).
26 Medications Master Table
Anti-VEGF Agents
| Agent | Target | Dose (Intravitreal) | Key Indications | Notes |
|---|---|---|---|---|
| Bevacizumab (Avastin) | VEGF-A | 1.25 mg/0.05 mL | Wet AMD, DME, RVO, NV | Off-label; most cost-effective (~$50/dose) |
| Ranibizumab (Lucentis) | VEGF-A | 0.5 mg (AMD), 0.3 mg (DME, RVO) | Wet AMD, DME, DR, RVO | Fab fragment; FDA-approved |
| Aflibercept (Eylea) | VEGF-A, VEGF-B, PlGF | 2 mg/0.05 mL | Wet AMD, DME, DR, RVO | Decoy receptor (VEGF-Trap) |
| Aflibercept 8 mg (Eylea HD) | VEGF-A, VEGF-B, PlGF | 8 mg/0.07 mL | Wet AMD, DME | Higher dose for extended intervals (up to 16 weeks) |
| Faricimab (Vabysmo) | VEGF-A + Ang-2 | 6 mg/0.05 mL | Wet AMD, DME | Bispecific; up to q16-week dosing |
| Brolucizumab (Beovu) | VEGF-A | 6 mg/0.05 mL | Wet AMD | Risk of intraocular inflammation/vasculitis; limited use |
IOP-Lowering Drops
| Class | Agents | Mechanism | IOP Reduction | Key Side Effects |
|---|---|---|---|---|
| Prostaglandin analog | Latanoprost, travoprost, bimatoprost, tafluprost | ↑ Uveoscleral outflow | 25-33% | Hyperemia, iris darkening, lash growth, periorbital fat atrophy |
| Beta-blocker | Timolol, betaxolol, levobunolol | ↓ Aqueous production | 20-25% | Bronchospasm, bradycardia, depression, fatigue |
| Alpha-2 agonist | Brimonidine, apraclonidine | ↓ Aqueous production, ↑ uveoscleral outflow | 20-25% | Allergic conjunctivitis, somnolence (esp. children) |
| CAI (topical) | Dorzolamide, brinzolamide | ↓ Aqueous production | 15-20% | Stinging, metallic taste, corneal endothelial toxicity |
| CAI (oral) | Acetazolamide, methazolamide | ↓ Aqueous production | 25-30% | Paresthesias, metabolic acidosis, kidney stones, aplastic anemia |
| Rho-kinase inhibitor | Netarsudil | ↑ Trabecular outflow, ↓ EVP | ~20% | Conjunctival hyperemia, cornea verticillata |
| Miotic | Pilocarpine | Contracts ciliary muscle, opens TM | 20-25% | Miosis, brow ache, myopic shift, RD risk |
Ophthalmic Corticosteroids
| Route | Agent | Common Uses | Notes |
|---|---|---|---|
| Topical | Prednisolone acetate 1% | Anterior uveitis, post-op inflammation | Gold standard topical steroid; potent |
| Topical | Loteprednol 0.5% | Allergic conjunctivitis, mild inflammation | "Soft" steroid — less IOP rise |
| Topical | Difluprednate 0.05% | Post-op, severe uveitis | Most potent topical; penetrates well; highest IOP risk |
| Intravitreal | Triamcinolone acetonide (4 mg) | DME, uveitic CME, vitrectomy adjunct | Duration: 3-4 months; IOP rise, cataract |
| Intravitreal implant | Dexamethasone (Ozurdex, 0.7 mg) | RVO macular edema, uveitis, DME | Biodegradable; duration ~4-6 months |
| Intravitreal implant | Fluocinolone (Retisert, 0.59 mg) | Chronic non-infectious posterior uveitis | Non-biodegradable; lasts ~30 months; high cataract/glaucoma risk |
| Periocular | Triamcinolone (sub-Tenon, 40 mg) | CME, intermediate uveitis | Avoids intravitreal risk; depot effect |
Ophthalmic Antibiotics
| Agent | Route | Common Uses | Notes |
|---|---|---|---|
| Moxifloxacin 0.5% | Topical | Corneal ulcer (non-severe), surgical prophylaxis | 4th-gen fluoroquinolone; excellent corneal penetration |
| Fortified vancomycin (50 mg/mL) | Topical | Severe bacterial keratitis (Gram-positive) | Compounded; needs refrigeration |
| Fortified tobramycin (14 mg/mL) | Topical | Severe bacterial keratitis (Gram-negative) | Compounded; used in combination with vancomycin |
| Natamycin 5% | Topical | Fungal keratitis (filamentary) | Only commercial topical antifungal in the US |
| Voriconazole 1% | Topical / intrastromal | Fungal keratitis (resistant) | Compounded; broad-spectrum antifungal |
| Vancomycin 1 mg/0.1 mL | Intravitreal | Endophthalmitis (Gram-positive) | Standard intravitreal dose |
| Ceftazidime 2.25 mg/0.1 mL | Intravitreal | Endophthalmitis (Gram-negative) | Standard intravitreal dose; amikacin alternative |
Cycloplegics & Mydriatics
| Agent | Onset | Duration of Cycloplegia | Common Uses |
|---|---|---|---|
| Tropicamide 1% | 20-30 min | 2-6 hours | Routine dilation for fundus exam |
| Phenylephrine 2.5% or 10% | 15-30 min | 3-5 hours (mydriasis only, no cycloplegia) | Mydriasis; combined with tropicamide for full dilation |
| Cyclopentolate 1% | 30-60 min | 6-24 hours | Cycloplegic refraction in children, uveitis |
| Homatropine 2% | 30-60 min | 1-3 days | Uveitis, post-op |
| Atropine 1% | 30-60 min | 7-14 days | Severe uveitis, penalization for amblyopia |
Immunosuppressants in Ophthalmology
| Agent | Class | Indications | Key Monitoring |
|---|---|---|---|
| Methotrexate | Antimetabolite | Uveitis, OCP, scleritis | CBC, LFTs, renal function; folic acid supplementation |
| Mycophenolate mofetil | Antimetabolite | Uveitis, scleritis, OCP | CBC, LFTs; GI side effects common |
| Azathioprine | Antimetabolite | Uveitis (less commonly used) | TPMT testing before starting; CBC, LFTs |
| Adalimumab (Humira) | Anti-TNF-alpha | Non-infectious intermediate/posterior/panuveitis; JIA uveitis | TB screening; hepatitis B screening; infection risk |
| Infliximab (Remicade) | Anti-TNF-alpha | Refractory uveitis, Behcet disease | IV infusion; TB/hepatitis screening |
| Cyclosporine (systemic) | Calcineurin inhibitor | Refractory uveitis, Behcet, VKH | Blood pressure, renal function, drug levels |
| Teprotumumab (Tepezza) | Anti-IGF-1R | Thyroid eye disease | Blood glucose; audiometry (hearing loss risk) |
27 Imaging & Diagnostics
Optical Coherence Tomography (OCT)
OCT is the single most transformative imaging modality in ophthalmology. Spectral-domain (SD-OCT) and swept-source (SS-OCT) provide cross-sectional, near-histologic images (~5 μm axial resolution) of the retina, optic nerve, and anterior segment. Key applications: macular disease (AMD — subretinal/intraretinal fluid, PED, GA; DME — central subfield thickness for treatment decisions; ERM — traction on the fovea; macular hole staging), glaucoma (RNFL thickness maps — inferior and superior thinning earliest; ganglion cell analysis — GCC/GCL-IPL), and anterior segment OCT (corneal thickness, angle assessment). OCT angiography (OCTA) uses motion contrast to map retinal and choroidal vasculature without dye injection — detects CNV, capillary non-perfusion, and microaneurysms.
Fluorescein Angiography (FA)
FA involves IV injection of sodium fluorescein (10% or 25%) followed by rapid sequential fundus photography with blue excitation (490 nm) and yellow-green barrier (530 nm) filters. Phases: choroidal flush (8-12 sec), arterial (10-12 sec), arteriovenous (13-15 sec), venous (15-20 sec), recirculation (20-30 sec), and late (5-10 min). Patterns of abnormality: hyperfluorescence from leakage (edema, CNV, NV — increases in intensity and size), staining (scar tissue, drusen — increases in intensity but not size), pooling (PED, serous RD — fills a space), and window defects (RPE atrophy — early, does not change size). Hypofluorescence from blocking (hemorrhage, pigment) or non-perfusion (capillary dropout, vascular occlusion). Risks: nausea (most common, ~5%), skin yellowing and fluorescent urine (24 hours), rare anaphylaxis (~1 in 200,000). Contraindicated in pregnancy (relative).
Indocyanine Green Angiography (ICG)
ICG uses indocyanine green dye, which is highly protein-bound (98%) and therefore remains predominantly intravascular, providing better imaging of the choroidal vasculature (ICG fluoresces in the near-infrared range — 835 nm excitation, 800 nm — which penetrates the RPE, blood, and lipid better than fluorescein). Key indications: polypoidal choroidal vasculopathy (PCV — branching vascular network with polyps, common in Asian patients with wet AMD phenotype), central serous chorioretinopathy (CSC — identifies areas of choroidal hyperpermeability), and occult CNV. Contraindicated in iodine/shellfish allergy (ICG contains iodide).
B-Scan Ultrasonography
B-scan ultrasound (10 MHz) provides real-time, 2D cross-sectional imaging of the globe and orbit. Essential when the fundus cannot be visualized (dense cataract, vitreous hemorrhage, corneal opacity). Key uses: retinal detachment detection (attached or detached retina beneath VH — critical surgical planning), intraocular tumor evaluation (choroidal melanoma — acoustic hollowing, mushroom shape; retinoblastoma — calcification; metastasis), posterior vitreous detachment confirmation, orbital masses, and axial length measurement (A-scan). Ultrasound biomicroscopy (UBM, 35-50 MHz) provides high-resolution imaging of the anterior segment — ciliary body, angle, and posterior chamber. Key for plateau iris diagnosis, ciliary body tumors, and sulcus IOL position assessment.
Visual Field Interpretation
Key Humphrey visual field metrics: mean deviation (MD) — overall depression of the field (normal > -2 dB; mild loss -2 to -6 dB; moderate -6 to -12 dB; severe < -12 dB); pattern standard deviation (PSD) — degree of localized deviation from normal (elevated in early glaucoma with focal loss); visual field index (VFI) — percentage of normal field (100% = normal, 0% = complete loss); and the glaucoma hemifield test (GHT) — compares corresponding superior and inferior zones (result: within normal limits, borderline, or outside normal limits). Progression analysis: guided progression analysis (GPA) — event-based; VFI trend analysis — rate-based.
Ophthalmic Emergencies — Summary
| Emergency | Key Features | Immediate Action |
|---|---|---|
| Chemical burn | History of chemical exposure, pain, blepharospasm | Immediate irrigation (30+ min), pH check |
| CRAO | Sudden painless vision loss, cherry-red spot, pale retina | Acute stroke protocol; rule out GCA if ≥ 50 |
| Acute angle closure | Severe pain, nausea, fixed mid-dilated pupil, IOP > 40 | Topical timolol/brimonidine, IV acetazolamide, LPI |
| Open globe | Penetrating trauma, irregular pupil, low IOP, positive Seidel | Shield (NO patch), IV antibiotics, NPO, emergent repair |
| Endophthalmitis | Pain + vision loss 1-7 days post-op, hypopyon, vitritis | Vitreous tap/inject (vancomycin + ceftazidime); PPV if LP-only |
| Orbital cellulitis | Proptosis, restricted EOM, pain, fever, eyelid edema | CT orbits, IV antibiotics; surgical drainage if abscess |
| Retinal detachment | Flashes, floaters, curtain/shadow, Shafer sign | Same-day ophthalmology evaluation; urgent surgery |
| Pupil-involving CN III palsy | Ptosis, "down and out," dilated pupil | Emergent CTA/MRA — PComm aneurysm until proven otherwise |
| GCA (arteritic AION) | Vision loss + headache + jaw claudication, elevated ESR/CRP | IV methylprednisolone immediately; temporal artery biopsy |
| White-eyed blowout fracture (child) | Nausea/vomiting, restricted upgaze, minimal external signs | Emergent surgical release of entrapped muscle |
Electrophysiology
Electroretinography (ERG): Measures the electrical response of the retina to light stimulation. Full-field (flash) ERG evaluates generalized retinal function — essential for diagnosing retinal dystrophies (retinitis pigmentosa shows reduced/absent scotopic and photopic responses), evaluating retinal function when the fundus cannot be visualized (dense VH or media opacity), and preoperative assessment. Components: a-wave (photoreceptor response) and b-wave (bipolar/Muller cell response). Pattern ERG (PERG) evaluates ganglion cell function — useful in early glaucoma and optic neuropathies. Multifocal ERG (mfERG) maps localized retinal function — useful for hydroxychloroquine (Plaquenil) toxicity screening (early parafoveal depression). Visual evoked potential (VEP): Measures the cortical electrical response to visual stimulation — evaluates the entire visual pathway from retina to visual cortex. Used for optic nerve disease (prolonged latency in demyelinating optic neuritis), malingering detection, and pediatric visual acuity estimation.
Hydroxychloroquine retinal toxicity screening: The AAO recommends baseline eye exam within the first year of starting HCQ, then annual screening after 5 years (or earlier with risk factors: dose > 5 mg/kg real body weight, renal impairment, concomitant tamoxifen use, macular disease). Screening tests: SD-OCT (parafoveal thinning of the outer retinal layers — earliest detectable change), HVF 10-2 (paracentral scotomas), and mfERG (parafoveal amplitude depression). The classic "bull's eye maculopathy" on fundoscopy is a late finding indicating irreversible damage — screening aims to detect toxicity before this stage.
CT & MRI of the Orbits
CT orbits (with contrast): First-line for orbital fractures (thin-cut coronal and sagittal reformats), orbital cellulitis/abscess, metallic foreign body detection, acute proptosis. Advantages: fast, excellent bony detail, widely available. MRI orbits (with fat suppression and gadolinium): Superior for optic nerve pathology (optic neuritis — post-gadolinium enhancement of the optic nerve; optic nerve glioma, meningioma), soft tissue characterization of orbital tumors, cavernous sinus lesions, and evaluation of extraocular muscle inflammation (TED — T2 hyperintensity of muscles indicates active inflammation). Short-tau inversion recovery (STIR) sequence is particularly useful for detecting optic nerve and orbital inflammation.
| Clinical Scenario | First-Line Imaging | Key Findings |
|---|---|---|
| Orbital fracture | CT orbits (thin-cut, coronal + sagittal) | Bony defect, soft tissue herniation, trapped muscle |
| Orbital cellulitis / abscess | CT orbits with contrast | Fat stranding, rim-enhancing abscess, sinus opacification |
| Metallic intraocular foreign body | CT orbits (NO MRI) | Hyperdense foreign body, track through eye wall |
| Optic neuritis / MS | MRI brain + orbits with gadolinium | Optic nerve enhancement, white matter lesions |
| Thyroid eye disease (active) | MRI orbits (STIR + gadolinium) | T2 hyperintense / enhancing EOMs (inflammation vs. fibrosis) |
| Optic nerve tumor | MRI orbits with fat suppression | Optic nerve enlargement, railroad track sign (meningioma) |
| Choroidal melanoma / retinal detachment behind VH | B-scan ultrasound | Mushroom shape, acoustic hollowing (melanoma); RD membrane |
| Plateau iris / angle anatomy | UBM (35-50 MHz ultrasound) | Anteriorly positioned ciliary body, angle closure |
| Wet AMD / DME / CNV activity | OCT ± OCTA | Subretinal fluid, intraretinal cysts, PED, CNV flow |
| Retinal ischemia / neovascularization | Fluorescein angiography | Capillary non-perfusion, NV leakage, microaneurysms |
28 Abbreviations Master List
| Abbreviation | Definition |
|---|---|
| AC | Anterior chamber |
| ACG | Angle-closure glaucoma |
| AMD | Age-related macular degeneration |
| AREDS | Age-Related Eye Disease Study |
| BCC | Basal cell carcinoma |
| BIO | Binocular indirect ophthalmoscope |
| BRVO | Branch retinal vein occlusion |
| CAI | Carbonic anhydrase inhibitor |
| CAS | Clinical Activity Score |
| CCT | Central corneal thickness |
| CF | Count fingers |
| CI-DME | Center-involving diabetic macular edema |
| CME | Cystoid macular edema |
| CNV | Choroidal neovascularization |
| CRAO | Central retinal artery occlusion |
| CRVO | Central retinal vein occlusion |
| CSC | Central serous chorioretinopathy |
| CXL | Corneal collagen cross-linking |
| DALK | Deep anterior lamellar keratoplasty |
| DCR | Dacryocystorhinostomy |
| DED | Dry eye disease |
| DEWS | Dry Eye Workshop |
| DLK | Diffuse lamellar keratitis |
| DMEK | Descemet membrane endothelial keratoplasty |
| DME | Diabetic macular edema |
| DON | Dysthyroid optic neuropathy |
| DR | Diabetic retinopathy |
| DSAEK | Descemet stripping automated endothelial keratoplasty |
| EDOF | Extended depth of focus |
| EOM | Extraocular muscle |
| ERM | Epiretinal membrane |
| ETDRS | Early Treatment Diabetic Retinopathy Study |
| EVS | Endophthalmitis Vitrectomy Study |
| FA | Fluorescein angiography |
| FLACS | Femtosecond laser-assisted cataract surgery |
| GA | Geographic atrophy |
| GAT | Goldmann applanation tonometry |
| GCA | Giant cell arteritis |
| HM | Hand motions |
| HVF | Humphrey visual field |
| ICG | Indocyanine green angiography |
| ICL | Implantable Collamer lens |
| IFIS | Intraoperative floppy iris syndrome |
| IIH | Idiopathic intracranial hypertension |
| ILM | Internal limiting membrane |
| IOL | Intraocular lens |
| IOP | Intraocular pressure |
| IRMA | Intraretinal microvascular abnormality |
| KP | Keratic precipitate |
| LASIK | Laser in situ keratomileusis |
| LOCS | Lens Opacities Classification System |
| LP | Light perception |
| LPI | Laser peripheral iridotomy |
| MGD | Meibomian gland dysfunction |
| MIGS | Micro-invasive glaucoma surgery |
| NLP | No light perception |
| NPDR | Non-proliferative diabetic retinopathy |
| NVD | Neovascularization of the disc |
| NVE | Neovascularization elsewhere |
| NVG | Neovascular glaucoma |
| NVI | Neovascularization of the iris (rubeosis) |
| OCT | Optical coherence tomography |
| OCTA | OCT angiography |
| OIS | Ocular ischemic syndrome |
| ONTT | Optic Neuritis Treatment Trial |
| OSSN | Ocular surface squamous neoplasia |
| PAM | Primary acquired melanosis |
| PCO | Posterior capsule opacification |
| PCR | Posterior capsule rupture |
| PCV | Polypoidal choroidal vasculopathy |
| PDR | Proliferative diabetic retinopathy |
| PED | Pigment epithelial detachment |
| PGA | Prostaglandin analog |
| PK | Penetrating keratoplasty |
| POAG | Primary open-angle glaucoma |
| PPV | Pars plana vitrectomy |
| PRK | Photorefractive keratectomy |
| PRP | Panretinal photocoagulation |
| PSC | Posterior subcapsular cataract |
| PVD | Posterior vitreous detachment |
| PVR | Proliferative vitreoretinopathy |
| PXF | Pseudoexfoliation |
| RAPD | Relative afferent pupillary defect |
| RGP | Rigid gas-permeable (contact lens) |
| RNFL | Retinal nerve fiber layer |
| ROP | Retinopathy of prematurity |
| RPE | Retinal pigment epithelium |
| RRD | Rhegmatogenous retinal detachment |
| SB | Scleral buckle |
| SCC | Squamous cell carcinoma |
| SLT | Selective laser trabeculoplasty |
| SMILE | Small incision lenticule extraction |
| SUN | Standardization of Uveitis Nomenclature |
| TASS | Toxic anterior segment syndrome |
| TED | Thyroid eye disease |
| TRD | Tractional retinal detachment |
| UBM | Ultrasound biomicroscopy |
| VA | Visual acuity |
| VEGF | Vascular endothelial growth factor |
| VH | Vitreous hemorrhage |
| VKH | Vogt-Koyanagi-Harada (syndrome) |
| VF | Visual field |
| ARN | Acute retinal necrosis |
| BIO | Binocular indirect ophthalmoscopy |
| BSS | Balanced salt solution |
| CCC | Continuous curvilinear capsulorhexis |
| CN | Cranial nerve |
| CPEO | Chronic progressive external ophthalmoplegia |
| CTR | Capsular tension ring |
| CWS | Cotton-wool spot |
| DVD | Dissociated vertical deviation |
| EFT | Essential Frailty Toolset |
| ERG | Electroretinography |
| GATT | Gonioscopy-assisted transluminal trabeculotomy |
| HCQ | Hydroxychloroquine |
| ICRB | International Classification of Retinoblastoma |
| IOFB | Intraocular foreign body |
| IRU | Immune recovery uveitis |
| JIA | Juvenile idiopathic arthritis |
| MMC | Mitomycin C |
| MR | Medial rectus |
| LR | Lateral rectus |
| SO | Superior oblique |
| OCP | Ocular cicatricial pemphigoid |
| OHT | Ocular hypertension |
| OHTS | Ocular Hypertension Treatment Study |
| PD | Prism diopter |
| PDS | Port delivery system / pigment dispersion syndrome |
| SR | Superior rectus |
| IR | Inferior rectus |
| IO | Inferior oblique |
| STIR | Short-tau inversion recovery (MRI sequence) |
| TM | Trabecular meshwork |
| VEP | Visual evoked potential |
Phacoemulsification handpiece: Ultrasonic device that emulsifies the cataract nucleus for aspiration through a small incision.
Vitrectomy cutter: Guillotine-style cutting and suction instrument (up to 15,000 cuts/min in modern systems) for vitreous removal.
Endolaser: Intraocular laser probe (532 nm or 810 nm) for retinal photocoagulation during vitrectomy.
Excimer laser (193 nm): Argon fluoride laser used for corneal ablation in refractive surgery (LASIK, PRK). Breaks intermolecular bonds without thermal damage.
Nd:YAG laser (1064 nm): Used for posterior capsulotomy (PCO), laser peripheral iridotomy (LPI), and vitreolysis.
Argon/frequency-doubled Nd:YAG (532 nm): Used for PRP, focal/grid laser for macular edema, SLT, and retinopexy.
Femtosecond laser: Ultrashort-pulse infrared laser used for LASIK flap creation, FLACS (capsulorhexis, lens fragmentation), corneal incisions, and SMILE lenticule creation.
ONTT — Optic Neuritis Treatment Trial: IV steroids accelerate recovery but do not change final outcome; oral prednisone alone increases recurrence. PubMed 18057340
EVS — Endophthalmitis Vitrectomy Study: For post-cataract endophthalmitis, LP-only vision patients benefit from immediate PPV. PubMed 7629165
DRCR.net Protocol T — Comparative effectiveness of anti-VEGF agents for DME: aflibercept best at lower VA, equivalent at higher VA. PubMed 25692915
CATT — Comparison of AMD Treatments Trial: ranibizumab and bevacizumab equivalent for wet AMD. PubMed 21558220
AREDS / AREDS2 — Antioxidant supplementation reduces progression of intermediate AMD to advanced AMD by ~25%. PubMed 23644932
EAGLE — Early clear lens extraction was superior to LPI for PAC/PACG. PubMed 27720356
LiGHT — SLT as initial treatment was superior to eye drops for POAG at 3 years. PubMed 30862378
TVT — Tube vs. Trabeculectomy: Baerveldt tube had higher success rate than trabeculectomy with MMC at 5 years in previously operated eyes. PubMed 22828386
HEDS — Herpetic Eye Disease Study: oral acyclovir reduces recurrence of HSV keratitis. PubMed 9684095
IIHTT — IIH Treatment Trial: acetazolamide + diet improved visual field function in IIH with mild visual loss. PubMed 24718150
PIVOT — Pneumatic retinopexy vs. vitrectomy for RRD: equivalent VA outcomes at 1 year. PubMed 34037711
Protocol S (DRCR.net) — Ranibizumab non-inferior to PRP for PDR at 2 years. PubMed 26567078
ETROP — Early Treatment for ROP: early treatment of Type 1 ROP reduces unfavorable outcomes. PubMed 14662593
SCORE — Standard Care vs Corticosteroid for Retinal Vein Occlusion: intravitreal triamcinolone effective for CRVO macular edema; no benefit over grid laser for BRVO. PubMed 19520537
OHTS — Ocular Hypertension Treatment Study: topical IOP-lowering medications reduced 5-year risk of POAG development in OHT patients. PubMed 12049574
DRS — Diabetic Retinopathy Study: PRP reduced risk of severe vision loss in high-risk PDR by > 50%. PubMed 6993984
EUGOGO — Selenium supplementation improved outcomes in mild active TED. IV methylprednisolone pulse therapy superior to oral steroids for moderate-severe TED. PubMed 21591944
ATS — Amblyopia Treatment Studies: patching and atropine penalization are both effective for moderate amblyopia; 2 hours of patching as effective as 6 hours for moderate amblyopia. PubMed 12714610