Reproductive Endocrinology
Every diagnosis, hormonal pathway, fertility treatment, assisted reproduction technique, classification, complication, medication, and management algorithm across the full scope of reproductive endocrinology and infertility in one place.
01 Reproductive Anatomy & Physiology
Hypothalamic-Pituitary-Ovarian (HPO) Axis
The HPO axis is the master regulatory circuit for female reproduction. The hypothalamus releases gonadotropin-releasing hormone (GnRH) in a pulsatile fashion from the arcuate nucleus into the hypophyseal portal system. GnRH pulsatility is critical: high-frequency pulses (~every 60 minutes) preferentially stimulate LH secretion, while low-frequency pulses (~every 90-120 minutes) favor FSH secretion. Continuous (non-pulsatile) GnRH administration paradoxically suppresses gonadotropin release through receptor downregulation — the pharmacologic basis of GnRH agonist protocols.
The anterior pituitary gonadotrophs secrete FSH and LH in response to GnRH. FSH stimulates follicular recruitment, granulosa cell proliferation, and aromatase activity. LH stimulates theca cell androgen production and triggers ovulation at mid-cycle. Negative feedback is exerted by estradiol (low levels suppress FSH/LH), progesterone, and inhibin B (selectively suppresses FSH). Positive feedback occurs when estradiol exceeds ~200 pg/mL for ≥50 hours, triggering the LH surge that initiates ovulation approximately 36 hours later.
Folliculogenesis
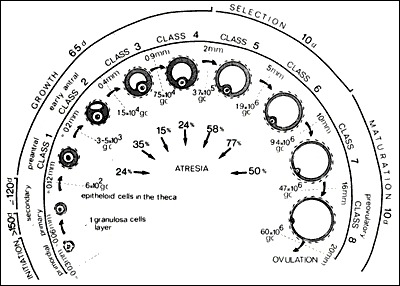
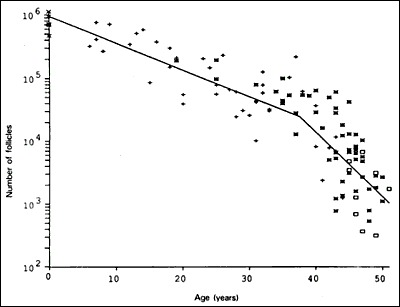
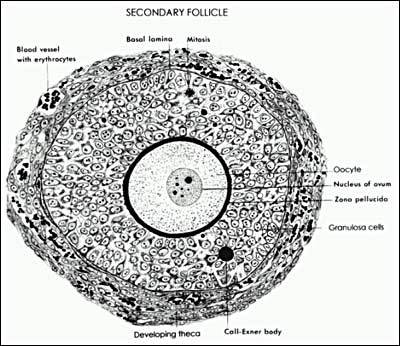
A woman is born with approximately 1-2 million oocytes; by menarche ~300,000-400,000 remain. Follicular development proceeds through defined stages: primordial follicle (quiescent, single layer of flat granulosa cells) → primary follicle (cuboidal granulosa, zona pellucida formation) → secondary (preantral) follicle (multiple granulosa layers, theca cell recruitment) → antral follicle (fluid-filled cavity, FSH-responsive from ~2 mm) → Graafian (preovulatory) follicle (~18-25 mm, dominant follicle ready for ovulation).
The transition from primordial to secondary follicle takes approximately 290 days and is largely gonadotropin-independent (regulated by local paracrine factors including AMH, Kit ligand, BMPs). The gonadotropin-dependent phase begins at the antral stage and spans approximately 70 days. Each menstrual cycle, a cohort of antral follicles is recruited by rising FSH; through follicular selection, a single dominant follicle emerges (by cycle day 6-7) while remaining cohort follicles undergo atresia due to declining FSH.
Two-Cell, Two-Gonadotropin Theory
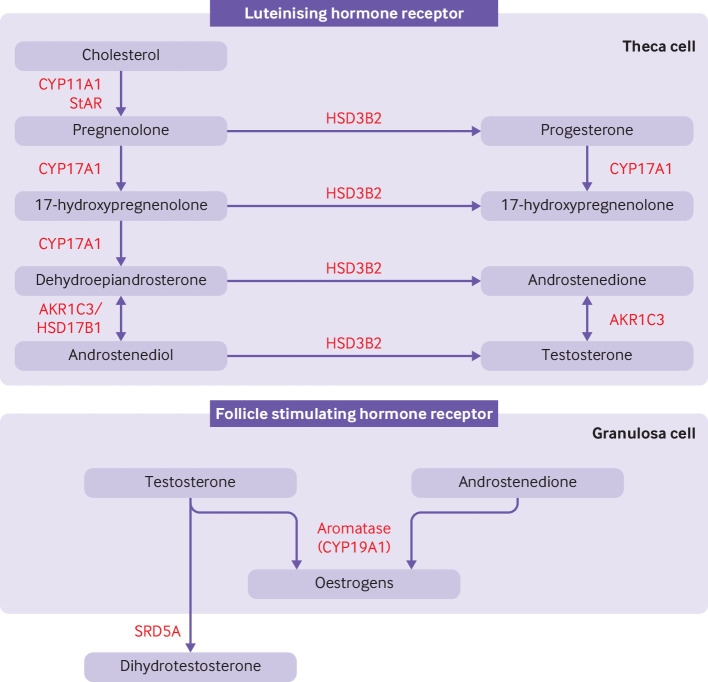
Estrogen biosynthesis requires cooperation between two ovarian cell types. Theca cells (outer layer) express LH receptors and convert cholesterol to androstenedione (via CYP17/17α-hydroxylase/17,20-lyase). Granulosa cells (inner layer) express FSH receptors and contain aromatase (CYP19), which converts thecal androgens to estradiol. Neither cell type alone can efficiently produce estrogen — this is the two-cell, two-gonadotropin model.
Corpus Luteum & Luteal Phase
After ovulation, the collapsed follicle transforms into the corpus luteum, a transient endocrine organ that produces progesterone (from luteinized granulosa cells) and estradiol. Progesterone secretion peaks 7-8 days after ovulation (mid-luteal), reaching levels of 10-20 ng/mL. The corpus luteum has a fixed lifespan of ~14 days; without hCG rescue from an implanting embryo, it undergoes luteolysis, progesterone falls, and menstruation ensues. In early pregnancy, hCG maintains the corpus luteum until the luteal-placental shift occurs at approximately 7-9 weeks of gestation.
Oocyte Maturation & Ovulation
The oocyte within the dominant follicle is arrested in prophase I of meiosis (dictyate stage) since fetal life. The LH surge triggers resumption of meiosis I, extrusion of the first polar body, and arrest in metaphase II (MII) — this is the mature, fertilizable oocyte. Ovulation occurs approximately 36 hours after the LH surge onset: proteolytic enzymes (MMPs, plasmin) and prostaglandins weaken the follicular wall, while smooth muscle contraction and local inflammation facilitate follicular rupture. The cumulus-oocyte complex (COC) is expelled and captured by the fimbriae of the fallopian tube. NSAIDs (which inhibit prostaglandin synthesis) can impair ovulation — patients undergoing fertility treatment should avoid them.
After ovulation, the oocyte remains viable for approximately 12-24 hours. Sperm survive in the female reproductive tract for up to 3-5 days. Fertilization occurs in the ampullary region of the fallopian tube. Capacitation (removal of cholesterol from the sperm membrane in the female tract) is required for the acrosome reaction that enables zona pellucida penetration. Upon sperm entry, the oocyte completes meiosis II, extrudes the second polar body, and forms the female pronucleus. The zona reaction (cortical granule exocytosis) provides the block to polyspermy.
The Endometrial Cycle
The endometrium undergoes cyclic changes driven by ovarian steroid hormones: (1) Proliferative phase — estradiol stimulates endometrial growth, glandular proliferation, and spiral arteriole elongation. Endometrial thickness increases from ~2 mm (early proliferative) to 8-14 mm. The endometrium has two layers: the functionalis (superficial, shed during menstruation) and the basalis (deep, regenerates the functionalis). (2) Secretory phase — progesterone from the corpus luteum induces glandular secretory transformation (subnuclear vacuolization by day 17, then supranuclear secretion), stromal decidualization (predecidual reaction begins periarteriolarly), and the window of implantation (approximately days 20-24 of a 28-day cycle, corresponding to 6-10 days post-ovulation). Histologic dating uses the Noyes criteria. Endometrial receptivity markers include pinopodes (small surface projections), integrins (especially αvβ3), and leukemia inhibitory factor (LIF). (3) Menstrual phase — withdrawal of estrogen and progesterone causes vasoconstriction of spiral arterioles, ischemic necrosis of the functional layer, and shedding. Prostaglandins (PGF2α) mediate myometrial contractions and vasoconstriction.
Implantation
The blastocyst reaches the uterine cavity approximately 4-5 days after fertilization. Implantation involves three stages: (1) Apposition — initial contact of the blastocyst trophectoderm with the endometrial surface epithelium; (2) Adhesion — firm attachment mediated by integrins, selectins, and cadherins; (3) Invasion — trophoblast cells penetrate the endometrial epithelium and invade the stroma, eroding spiral arterioles to establish uteroplacental circulation. Progesterone is essential for maintaining the decidualized endometrium and immune tolerance (via regulatory T cells and uterine NK cells). Defects in any stage may contribute to implantation failure or early pregnancy loss.
02 Reproductive Hormones & Assays
Estradiol (E2)
Estradiol is the principal circulating estrogen in premenopausal women, produced primarily by the dominant follicle's granulosa cells. Early follicular phase levels: 20-60 pg/mL. Each mature follicle contributes approximately 200-300 pg/mL in stimulated cycles. The mid-cycle peak (200-400 pg/mL in natural cycles) triggers the LH surge via positive feedback. In IVF monitoring, E2 levels guide dose adjustments: rapidly rising E2 (>3,000-4,000 pg/mL) signals OHSS risk.
Progesterone
Secreted by the corpus luteum after ovulation. Mid-luteal progesterone >3 ng/mL confirms ovulation (levels of 10-20 ng/mL are typical in a normal cycle). In the follicular phase, progesterone should be <1.5 ng/mL — premature luteinization (progesterone >1.5 ng/mL on trigger day) during IVF may impair endometrial receptivity and is a rationale for freeze-all strategy.
Anti-Müllerian Hormone (AMH)
AMH is produced by granulosa cells of preantral and small antral follicles (2-6 mm). It reflects the size of the remaining primordial follicle pool and is the most reliable serum marker of ovarian reserve. AMH can be drawn on any cycle day (minimal cycle variation). Age-specific norms: age 25 — median ~3.0 ng/mL; age 30 — ~2.5 ng/mL; age 35 — ~1.5 ng/mL; age 40 — ~1.0 ng/mL; age 45 — ~0.5 ng/mL. Low AMH (<1.0 ng/mL) suggests diminished ovarian reserve (DOR). High AMH (>3.5 ng/mL) is associated with PCOS and increased OHSS risk.
FSH & LH
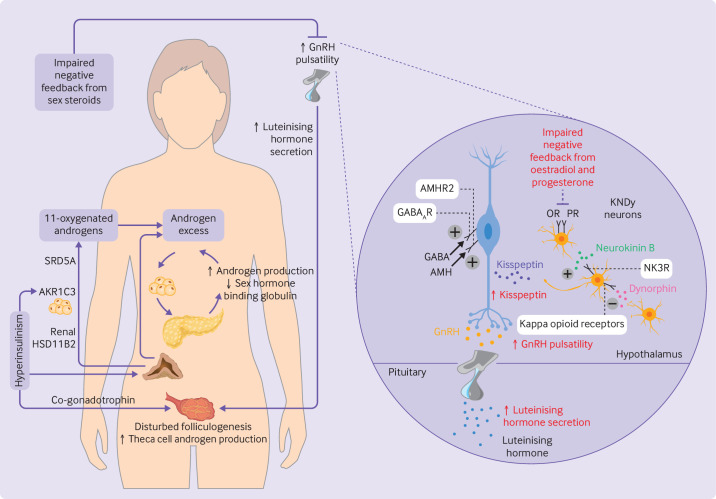
Cycle day 3 FSH is a cornerstone of ovarian reserve testing. Normal: <10 mIU/mL. Elevated FSH (>10-15 mIU/mL) indicates diminished ovarian reserve, with levels >20 mIU/mL suggesting poor prognosis. A single elevated FSH value has prognostic significance even if subsequent values are normal. The FSH:LH ratio >3:1 may suggest poor ovarian reserve; LH:FSH ratio >2:1 is classically associated with PCOS (though not a diagnostic criterion). An elevated LH to FSH ratio disrupts follicular development and may contribute to anovulation in PCOS through premature luteinization.
Inhibin B
Produced by granulosa cells of small antral follicles, inhibin B selectively suppresses pituitary FSH secretion via negative feedback. Low cycle day 3 inhibin B (<45 pg/mL) suggests diminished ovarian reserve. As the follicle pool declines, reduced inhibin B leads to loss of FSH suppression — hence the rise in FSH seen with reproductive aging.
Androgens
Total testosterone, free testosterone, DHEA-S, and androstenedione are measured in the evaluation of hyperandrogenism (PCOS, CAH, androgen-secreting tumors). In PCOS, testosterone is mildly elevated (typically 50-150 ng/dL). Total testosterone >200 ng/dL or DHEA-S >700 μg/dL should prompt evaluation for an androgen-secreting tumor or adrenal pathology. 17-hydroxyprogesterone (17-OHP) is the screening test for non-classic congenital adrenal hyperplasia (21-hydroxylase deficiency) — fasting early morning level >200 ng/dL warrants ACTH stimulation testing.
Prolactin & Thyroid Hormones
Prolactin elevation (>25 ng/mL) causes anovulation by suppressing GnRH pulsatility. Mild elevations (25-100 ng/mL) may be drug-induced (antipsychotics, metoclopramide, SSRIs), stress-related, or due to microprolactinoma. Levels >200 ng/mL are highly suggestive of macroprolactinoma. The "hook effect" can falsely normalize very high prolactin levels in giant prolactinomas — request serial dilutions if a macroadenoma is present with only mildly elevated prolactin. Screen for macroprolactin (big-big prolactin, biologically inactive) if prolactin is elevated with no clinical symptoms.
TSH should be checked in all infertility evaluations: subclinical hypothyroidism (TSH 2.5-4.0 mIU/L) may impair fertility and increase miscarriage risk. ASRM recommends a TSH target of <2.5 mIU/L in women attempting conception. Overt hypothyroidism (TSH >4.0 mIU/L) is associated with anovulation, hyperprolactinemia (TRH stimulates prolactin), and increased miscarriage risk. Thyroid autoimmunity (positive TPO antibodies) is present in ~10-15% of reproductive-age women and is independently associated with miscarriage and IVF failure, even with normal TSH — levothyroxine supplementation to maintain TSH <2.5 is recommended. Hyperthyroidism (Graves disease) can cause menstrual irregularity and should be treated before conception.
FSH <10 mIU/mL: Normal reserve. FSH 10-15 mIU/mL: Possible diminished reserve, correlate with AMH and AFC. FSH >15 mIU/mL: Diminished reserve; >20 suggests poor prognosis.
E2 <80 pg/mL on day 3: Normal (not masking elevated FSH). E2 >80 pg/mL on day 3: May falsely suppress FSH; indicates advanced follicular recruitment and possible DOR.
AMH >1.0 ng/mL: Adequate reserve for age. AMH <1.0 ng/mL: Diminished reserve. AMH >3.5 ng/mL: Consider PCOS; high OHSS risk with stimulation.
03 Ovarian Reserve Assessment
Anti-Müllerian Hormone (AMH) — Clinical Application
AMH is the single best endocrine marker of ovarian reserve, offering several clinical advantages: (1) Can be drawn on any cycle day (minimal intra-cycle variation — though small fluctuations exist, they are not clinically significant); (2) Not affected by oral contraceptive use (unlike FSH and AFC, which are suppressed by OCPs); (3) Provides a continuous quantitative measure rather than a threshold value; (4) Highly reproducible across assay platforms since standardization (picoAMH Elisa, Elecsys AMH — the Elecsys assay is now the most widely used and provides values ~15-20% higher than the Gen II ELISA). AMH predicts quantitative ovarian response to stimulation (number of oocytes retrieved) but does not reliably predict oocyte quality or live birth rate independent of age — this is a critical counseling point: a woman with low AMH at age 30 has reduced oocyte quantity but likely normal quality for her age. AMH declines steadily with age (approximately halving every 3-4 years) and becomes undetectable approximately 5 years before menopause, potentially serving as a predictor of time to menopause. However, AMH values should always be interpreted in clinical context alongside AFC, age, and clinical history. AMH is NOT a fertility test — women with low AMH can and do conceive naturally; conversely, normal AMH does not guarantee fertility.
Antral Follicle Count (AFC)
The AFC is the total number of follicles measuring 2-10 mm on both ovaries combined on transvaginal ultrasound during the early follicular phase (cycle days 2-4). The count should include all visible follicles in both transverse and longitudinal planes of each ovary, ideally using a standardized systematic approach (automated follicle counting software is available but manual counting remains the standard). Normal AFC: 10-20 (age-dependent: ~15-30 at age 25-30, ~10-15 at age 35, ~5-10 at age 40). AFC <5-7 suggests diminished reserve and predicts poor response to stimulation. AFC >24 is associated with PCOS and OHSS risk.
AFC correlates well with AMH (r = 0.6-0.8) and predicts quantitative stimulation response (number of oocytes retrieved). Like AMH, AFC does not reliably predict oocyte quality or pregnancy probability independent of age. Limitations: inter-observer variability (~10-20% — operator experience and ultrasound quality matter), cycle-to-cycle variability (less than AMH but present), dependence on equipment quality (higher-frequency transducers detect more small follicles), and difficulty counting in patients with elevated BMI or PCOS (overlapping, difficult-to-distinguish follicles). Despite these limitations, AFC remains a cornerstone of ovarian reserve assessment and stimulation protocol planning.
Bologna Criteria for Poor Ovarian Response (2011)
The ESHRE Bologna criteria define poor ovarian response (POR) when at least two of three criteria are met: (1) Advanced maternal age (≥40 years) or other risk factor for POR; (2) Previous POR (≤3 oocytes with conventional stimulation); (3) Abnormal ovarian reserve test (AFC <5-7 or AMH <0.5-1.1 ng/mL). Two episodes of POR after maximal stimulation are sufficient for diagnosis regardless of age or reserve markers.
POSEIDON Classification (2016)
The POSEIDON (Patient-Oriented Strategies Encompassing IndividualizeD Oocyte Number) classification provides a more nuanced approach to low prognosis patients than the Bologna criteria:
Group 1: Age <35, adequate reserve (AFC ≥5, AMH ≥1.2 ng/mL), unexpected poor or suboptimal response (<4 oocytes in prior cycle, or 4-9 oocytes).
Group 2: Age ≥35, adequate reserve, unexpected poor or suboptimal response.
Group 3: Age <35, diminished reserve (AFC <5, AMH <1.2 ng/mL) — expected poor response but good egg quality potential.
Group 4: Age ≥35, diminished reserve — expected poor response and compromised egg quality. Worst prognosis subgroup.
Clomiphene Citrate Challenge Test (CCCT)
Historical test of ovarian reserve that provided additional prognostic information beyond baseline FSH: cycle day 3 FSH is measured; clomiphene 100 mg is given on days 5-9; cycle day 10 FSH is measured. An elevated day 10 FSH (>10 mIU/mL) suggests diminished reserve (clomiphene blocks estrogen feedback at the hypothalamus/pituitary; a healthy follicle pool should produce enough inhibin B and estradiol to suppress FSH by day 10 despite clomiphene blockade — failure to suppress indicates inadequate follicular response). The CCCT was more sensitive than baseline day 3 FSH alone, detecting some women with DOR who had normal basal FSH. However, it has been largely replaced by AMH and AFC in current practice due to the following: AMH is more sensitive and specific, does not require medication administration, can be drawn on any cycle day, and provides a continuous (rather than binary) measure of reserve. The CCCT adds minimal information beyond what AMH and AFC provide.
Responder Categorization for Stimulation Planning
Poor responder: AMH <1.0 ng/mL, AFC <5-7, age >38 — expect ≤3 oocytes; consider high-dose gonadotropins (300-450 IU/day), growth hormone co-treatment (experimental), mini-IVF (clomiphene + low-dose gonadotropins), dual stimulation (two retrievals in one cycle), accumulation of oocytes/embryos over multiple cycles, or donor oocytes if prognosis is very poor. Normal responder: AMH 1.0-3.5 ng/mL, AFC 7-20 — standard protocol (150-300 IU gonadotropins/day), expect 8-15 oocytes. High responder: AMH >3.5 ng/mL, AFC >20, often PCOS — OHSS risk, use low-dose gonadotropins (100-150 IU/day), antagonist protocol, GnRH agonist trigger, consider freeze-all. The OPTIMIST trial (van Tilborg, Lancet 2017) showed individualized dosing based on AMH reduced extreme responses (OHSS and cancellation) but did not improve overall live birth rates.
04 The REI Evaluation
Hysterosalpingography (HSG)
HSG is a first-line radiographic test for simultaneous assessment of tubal patency and uterine cavity morphology. Under fluoroscopy, radiopaque contrast is injected through the cervix via a cannula while serial spot images are obtained. Two types of contrast: water-soluble contrast (Omnipaque/iohexol, Conray/iothalmate — most commonly used; quickly absorbed from peritoneal cavity; provides good detail) and oil-based contrast (Lipiodol/ethiodized oil — may have a therapeutic fertility-enhancing effect; the H2Oil trial, Dreyer et al., NEJM 2017, showed higher ongoing pregnancy rates in the oil-based group: 39.7% vs 29.1%; possible mechanisms include tubal flushing of mucus plugs and debris, immunomodulatory effects on peritoneal macrophages, and improved tubal ciliary function). Bilateral free spill of contrast into the peritoneal cavity confirms tubal patency. HSG sensitivity for tubal occlusion: ~65% (significant false-positive rate); specificity: ~83%. False-positive tubal occlusion occurs in ~15-20% of cases, most commonly from cornual spasm at the tubal ostium. Pre-medication with ibuprofen 600 mg and/or a small dose of anxiolytic may reduce spasm and improve patient comfort. Procedure-related pain is common (cramping during and after injection) but usually self-limited.
Saline Infusion Sonohysterography (SIS/SHG)
Sterile saline (10-20 mL) is instilled into the uterine cavity through a thin catheter (e.g., Goldstein catheter, insemination catheter with balloon) during transvaginal ultrasound to distend the cavity and delineate intracavitary pathology. SIS is superior to standard transvaginal ultrasound and HSG for detecting submucosal fibroids (sensitivity ~90% vs ~50% for TVS), endometrial polyps, intrauterine adhesions, and Müllerian anomalies. 3D SIS provides additional information on uterine external contour and allows volume rendering. SIS does not assess tubal patency (unless combined with air/saline contrast — HyCoSy/hysterosalpingo-contrast-sonography, using agitated saline or commercial contrast agents like ExEm Foam, which allow visualization of tubal spill).
Timing: performed in the early proliferative phase (days 5-12) after cessation of menses but before ovulation (to avoid disrupting a potential early pregnancy and to minimize false-positive findings from thick secretory endometrium). Contraindications: active pelvic infection, positive pregnancy test. Complications: mild cramping (common, self-limited), vasovagal reaction (rare), and very rarely ascending infection (<1%). No routine antibiotic prophylaxis is required. SIS is increasingly used as the primary uterine cavity assessment in the infertility workup (replacing HSG for cavity evaluation when tubal patency is being assessed by other means).
Hysteroscopy
Hysteroscopy is the gold standard for evaluating and treating intracavitary pathology. Office (diagnostic) hysteroscopy uses a narrow-caliber rigid or flexible scope (2.7-3.5 mm outer diameter) with saline distension medium. Can be performed without anesthesia in most patients (paracervical block optional). Allows direct visualization of the endocervical canal, internal os, uterine cavity, tubal ostia, and any pathology. "See and treat" approach: small polyps, thin adhesions, and small Type 0 fibroids can be removed with miniature grasping or cutting instruments through the office hysteroscope.
Surgical (operative) hysteroscopy requires larger-caliber instruments: resectoscope (8-10 mm, monopolar or bipolar loop for fibroid and septum resection, roller-ball for endometrial ablation) or mechanical tissue removal systems (MyoSure, TruClear — morcellate polyps and fibroids with suction). Distension media: normal saline (with bipolar systems — preferred) or glycine/sorbitol/mannitol (with monopolar systems — risk of hyponatremia from fluid absorption — strict fluid deficit monitoring required: max 1,000-1,500 mL deficit for hypotonic media, 2,500 mL for isotonic saline). Indications: abnormal SIS findings requiring treatment, recurrent implantation failure (cavity assessment and possible endometrial scratch), Asherman syndrome adhesiolysis, uterine septum resection (scissors or loop), submucosal fibroid resection (FIGO type 0-2), and endometrial polypectomy. Complications: uterine perforation (~1%, higher with septum resection and adhesiolysis — concurrent ultrasound or laparoscopic guidance reduces risk), fluid overload, hemorrhage, cervical laceration, and infection.
Laparoscopy
Diagnostic and operative laparoscopy allows direct assessment of the entire pelvis, including tubal status, ovarian surface, peritoneal surfaces (for endometriosis), pelvic sidewalls, uterosacral ligaments, and adhesive disease. Chromopertubation (injection of dilute indigo carmine or methylene blue through the cervix via a uterine manipulator during laparoscopy) provides the most reliable assessment of tubal patency — dye is observed flowing from the fimbriated ends of the tubes into the peritoneal cavity.
Laparoscopy is no longer routine in all infertility workups (replaced by HSG and ultrasound for initial assessment) but is indicated when: (1) endometriosis is suspected (pelvic pain, dysmenorrhea, normal HSG, failed initial treatment); (2) HSG suggests significant tubal or peritoneal disease; (3) unexplained infertility not responding to empiric treatment (to identify occult endometriosis or adhesions); (4) treatment of known pathology (endometriosis excision, adhesiolysis, salpingectomy for hydrosalpinx, ovarian cystectomy, tubal reanastomosis). Important: if diagnostic laparoscopy is performed and endometriosis is found, treatment at the same surgery is recommended (do not just diagnose and leave disease untreated).
Semen Analysis — WHO 6th Edition (2021) Reference Values
Semen analysis should be among the first tests in any infertility evaluation, given male factor contributes to approximately 50% of cases. The WHO 6th edition provides updated lower reference limits (5th percentile of fertile men):
Volume: ≥1.4 mL (5th percentile). Sperm concentration: ≥16 million/mL. Total sperm count: ≥39 million/ejaculate. Total motility: ≥42%. Progressive motility: ≥30%. Normal morphology: ≥4% (Kruger strict criteria). Vitality: ≥54% live. pH: ≥7.2. White blood cells: <1.0 million/mL (leukocytospermia if exceeded).
At least two semen analyses, obtained 2-4 weeks apart after 2-5 days of abstinence, are recommended before diagnosing abnormalities, given significant intra-individual variability. Key terminology: oligozoospermia (concentration <16 M/mL), severe oligozoospermia (<5 M/mL), cryptozoospermia (rare sperm found only on centrifuged pellet), asthenozoospermia (progressive motility <30%), teratozoospermia (normal forms <4%), oligoasthenoteratozoospermia (OAT) (all three parameters abnormal — the most common semen analysis diagnosis in male infertility), azoospermia (no sperm in ejaculate after centrifugation of the entire specimen — must be confirmed on at least two analyses), necrozoospermia (all sperm dead/immotile), aspermia (absence of ejaculate).
Other Diagnostic Tests
Post-coital test (PCT/Sims-Hühner test): Examines cervical mucus 2-8 hours after intercourse during the periovulatory period; presence of motile sperm indicates adequate mucus-sperm interaction. Largely abandoned due to poor standardization and lack of prognostic value. Endometrial biopsy: Formerly used for luteal phase dating (Noyes criteria); no longer recommended for routine infertility evaluation as endometrial dating does not discriminate fertile from infertile women (Coutifaris et al., NEJM 2004). Now reserved for evaluating chronic endometritis (CD138 staining for plasma cells — prevalence 10-15% in infertile women, treated with doxycycline 100 mg BID for 14-28 days) or endometrial receptivity testing (ERA — gene expression array to time the window of implantation for personalized embryo transfer).
Infertility Evaluation Timeline
Standard initial evaluation is recommended after 12 months of unprotected intercourse in women <35, or after 6 months in women ≥35. Earlier evaluation is indicated for known risk factors: irregular cycles, history of PID, endometriosis, tubal surgery, known male factor, DES exposure, or prior cancer treatment. The basic workup can be completed in one menstrual cycle: cycle day 3 labs (FSH, E2, AMH, TSH, prolactin), mid-luteal progesterone (cycle day 21), HSG (cycle days 5-12), and semen analysis. Advanced testing (laparoscopy, hysteroscopy, genetic testing) is added based on initial findings.
The Fertility Evaluation — Male Partner
Beyond semen analysis, the male evaluation includes: detailed history (prior paternity, childhood illnesses including cryptorchidism and mumps orchitis, prior surgery, medications, environmental exposures, tobacco/alcohol/marijuana use), physical examination (testicular size and consistency, varicocele assessment with Valsalva, vas deferens palpation, secondary sexual characteristics), and hormonal evaluation when semen parameters are abnormal (FSH, testosterone, and prolactin). Referral to a reproductive urologist is indicated for azoospermia, severe oligozoospermia (<5 M/mL), clinical varicocele with abnormal parameters, or abnormal hormonal findings. Genetic evaluation (karyotype, Y-microdeletion, CFTR) is indicated for severe oligozoospermia or azoospermia.
05 Ovulatory Disorders
WHO Group I — Hypogonadotropic Hypogonadism
WHO Group I anovulation is characterized by low FSH, low LH, and low estradiol (<20 pg/mL) — reflecting hypothalamic or pituitary failure. This accounts for ~5-10% of anovulatory infertility.
Kallmann syndrome: Congenital GnRH deficiency due to failure of GnRH neuron migration from the olfactory placode. Associated with anosmia/hyposmia. Genetics: KAL1 (X-linked, anosmin-1), FGFR1, PROKR2, and others. Diagnosed by absent puberty, low gonadotropins, and impaired smell testing. Treatment: pulsatile GnRH via pump (physiologic) or exogenous gonadotropins (FSH + LH/hCG) to induce ovulation.
Functional hypothalamic amenorrhea (FHA): Suppression of GnRH pulsatility due to energy deficit (excessive exercise, eating disorders, weight loss), psychological stress, or a combination. The female athlete triad (low energy availability, menstrual dysfunction, low bone mineral density) is a common presentation. Pathophysiology: energy deficit reduces leptin and increases ghrelin and cortisol, which suppress the GnRH pulse generator via kisspeptin neurons. Diagnostic criteria: secondary amenorrhea, low/normal FSH and LH (typically both <5 mIU/mL), low estradiol (<20 pg/mL), exclusion of organic causes (MRI to rule out pituitary lesion, TSH, prolactin, karyotype if primary amenorrhea). Treatment: address underlying cause (nutritional rehabilitation — target weight gain of 2-5% of body weight, stress reduction, CBT — proven effective in the Berga et al. study); if fertility is desired and lifestyle modification fails, pulsatile GnRH via subcutaneous pump (most physiologic, restores entire HPO axis) or exogenous gonadotropin therapy (FSH + LH). Important: these patients should not receive clomiphene (requires functional HPO axis for effect) and are at risk for OHSS with gonadotropins (multiple follicles due to accumulated antral follicles from prolonged anovulation).
Sheehan syndrome: Postpartum pituitary necrosis due to severe hemorrhage and hypovolemia during delivery. Presents with failure of lactation, amenorrhea, fatigue, and hypotension. Panhypopituitarism may develop. Treatment: hormone replacement (thyroid, cortisol, estrogen/progesterone); fertility with gonadotropin therapy.
WHO Group II — Normogonadotropic Anovulation (PCOS)
WHO Group II accounts for ~75-85% of anovulatory infertility. FSH and LH are within normal range, and estrogen levels are present (estrogenized). Polycystic ovary syndrome (PCOS) is by far the most common cause — see Section 21 for detailed coverage.
Treatment ladder for ovulation induction in WHO Group II: (1) Lifestyle modification (weight loss of 5-10% restores ovulation in ~55-80% of overweight PCOS patients). (2) Letrozole 2.5-7.5 mg on cycle days 3-7 — now first-line per PPCOS II trial (Legro et al., NEJM 2014), superior to clomiphene for live birth rate. (3) Clomiphene citrate 50-150 mg on cycle days 3-7 or 5-9 — second-line. (4) Gonadotropins (low-dose step-up protocol — FSH starting at 37.5-75 IU daily, increasing every 7-14 days) — higher risk of multiples and OHSS. (5) Laparoscopic ovarian drilling (LOD) — alternative to gonadotropins in clomiphene-resistant PCOS. (6) IVF — if above measures fail.
WHO Group III — Premature Ovarian Insufficiency (POI)
POI (formerly premature ovarian failure) is defined as loss of ovarian function before age 40, characterized by amenorrhea/oligomenorrhea for ≥4 months and two FSH levels >25 mIU/mL measured ≥4 weeks apart (ESHRE 2016 criteria). Affects ~1% of women under 40. Etiologies:
Genetic (10-15%): Turner syndrome (45,X) and mosaic variants (45,X/46,XX), FMR1 premutations (55-200 CGG repeats — 13-26% lifetime risk of POI; women with premutations should be counseled about both POI risk and fragile X syndrome risk for offspring — PGT-M is available), other X chromosome abnormalities (deletions, translocations involving Xq13-q26 — the "critical region" for ovarian function), galactosemia (galactose-1-phosphate uridylyltransferase deficiency — >80% develop POI), BMP15 and FOXL2 mutations (rare). All women with POI <30 should have karyotype; all should be screened for FMR1 premutation.
Autoimmune (4-30%): Associated with autoimmune thyroiditis (~25% of POI patients have TPO antibodies), Addison disease (~4% develop adrenal insufficiency — screen with morning cortisol and ACTH or 21-hydroxylase antibodies), type 1 diabetes, celiac disease, myasthenia gravis, and autoimmune polyglandular syndromes (APS-1 and APS-2). Anti-ovarian antibodies are not standardized and have limited clinical utility. Iatrogenic (10-15%): Chemotherapy (especially alkylating agents — cyclophosphamide dose-response: cumulative dose >20 g causes POI in >90% of women >30), pelvic radiation (>6 Gy to ovaries is sterilizing in adults; dose threshold is lower in younger patients), bilateral oophorectomy. Infectious: Mumps oophoritis (rare in vaccinated populations), tuberculosis (in endemic areas). Idiopathic: Accounts for the majority (~50-60%) of cases even after thorough evaluation.
Fertility options: spontaneous conception occurs in ~5-10% of POI patients (intermittent ovarian function). Donor oocytes are the primary fertility treatment with high success rates (~50-55% live birth per transfer). In vitro activation (IVA) of residual follicles is investigational — involves disruption of the Hippo signaling pathway in ovarian cortex fragments followed by autotransplantation; early case reports are promising but the technique remains experimental. All POI patients need long-term management: HRT until the natural age of menopause (~50-51), bone density monitoring, cardiovascular risk assessment, and psychological support.
06 Tubal Factor Infertility
Etiology & Prevalence
Tubal factor accounts for ~25-35% of female infertility. The most common cause is pelvic inflammatory disease (PID) from ascending Chlamydia trachomatis or Neisseria gonorrhoeae infection. Risk of tubal infertility by number of PID episodes: 1 episode = ~8%; 2 episodes = ~20%; 3+ episodes = ~40% (Westrom et al., classic Swedish study). The damage is often subclinical — up to 50% of women with tubal factor infertility have no history of clinical PID (silent salpingitis, especially from Chlamydia). Chlamydia antibody testing (CAT) can serve as a screening test for tubal damage in the absence of clinical PID history. Other causes: previous ectopic pregnancy (especially if treated with salpingectomy), prior tubal surgery (including tubal sterilization), endometriosis-related adhesions, peritoneal adhesions from previous abdominal/pelvic surgery (appendectomy with perforation is a classic cause of right-sided tubal adhesions), and tuberculous salpingitis (in endemic regions — typically causes bilateral distal tubal occlusion with calcification).
Hydrosalpinx
A hydrosalpinx is a distally occluded, fluid-filled, dilated fallopian tube. The fimbriae are destroyed or agglutinated, and the tubal epithelium is often denuded. Hydrosalpinx fluid is embryotoxic: it contains cytokines, prostaglandins, and reactive oxygen species that impair endometrial receptivity, embryo development, and implantation. The mechanical reflux of fluid into the uterine cavity may also physically dislodge the embryo. Hydrosalpinx reduces IVF implantation rates by ~50% and live birth rates by ~50% when left in situ (Zeyneloglu et al., multiple meta-analyses).
Management before IVF: Laparoscopic salpingectomy (removal of the affected tube) before IVF is the gold standard — significantly improves IVF implantation and pregnancy rates (Johnson et al., Cochrane 2010; NNT = 5 for one additional clinical pregnancy). Salpingectomy is preferred over proximal tubal ligation (salpingostomy/clip/coagulation) because it completely removes the source of toxic fluid. Proximal tubal occlusion (Essure insert or laparoscopic clip/coagulation of the isthmic segment) is an alternative when salpingectomy is technically difficult (dense adhesions, concern for ovarian blood supply compromise). Ultrasound-guided aspiration of hydrosalpinx fluid at the time of oocyte retrieval is a temporary measure with high recurrence and is inferior to definitive surgical treatment.
Key consideration: the ovarian blood supply partially derives from a tubal branch of the uterine artery. Salpingectomy should be performed with meticulous technique to preserve the mesosalpinx vasculature and avoid compromising ovarian blood flow (which could reduce ovarian reserve and IVF response). Some data suggest modest reduction in ipsilateral AFC after salpingectomy, though this is debated. Ultrasound-visible hydrosalpinx (i.e., visible on baseline TVS without stimulation) carries the worst prognosis and is the strongest indication for surgical intervention before IVF.
Tubal Surgery vs. IVF
Decision depends on disease severity, patient age, additional infertility factors, and patient preference for natural conception vs. ART.
Mild distal tubal disease (phimosis, thin adhesions): Fimbrioplasty or neosalpingostomy; cumulative pregnancy rate ~40-60% over 12-24 months in women <35. May be offered as an alternative to IVF.
Moderate distal tubal disease (complete distal occlusion without hydrosalpinx): Neosalpingostomy with ~20-30% pregnancy rate; IVF may be more efficient depending on age.
Severe distal disease / hydrosalpinx: IVF is clearly superior. Salpingectomy before IVF (do not attempt tubal repair).
Proximal tubal occlusion: Selective tubal catheterization (fluoroscopic or hysteroscopic) first — resolves ~80% of proximal occlusions (many are due to spasm/debris). If true occlusion, consider microsurgical tubocornual anastomosis or IVF.
Tubal reanastomosis after sterilization: Success depends on remaining tubal length (≥4 cm optimal), method of original ligation (clip/ring better prognosis than Pomeroy/cautery), patient age (<35 ideal), and absence of other infertility factors. Pregnancy rates: 50-80% in well-selected patients <35. In women ≥38 or with additional factors (male factor, DOR), IVF is generally more efficient (per-cycle success higher, faster time to pregnancy).
General principle: IVF success rates are not affected by tubal factor (assuming hydrosalpinx is treated), so IVF is always an option. Tubal surgery offers the possibility of multiple natural pregnancies without further intervention but requires months of healing and carries ectopic pregnancy risk (~5-15%).
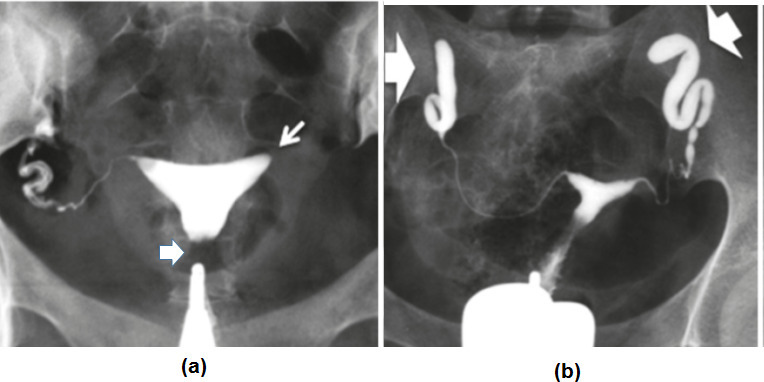
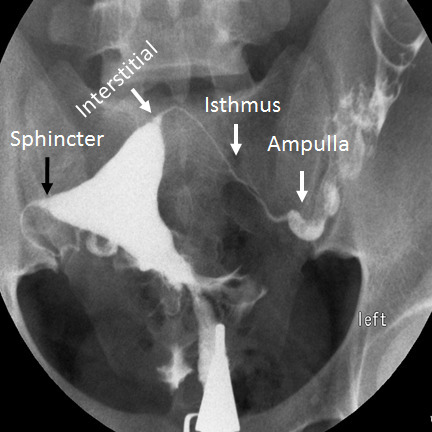
HSG Interpretation
HSG is performed in a fluoroscopy suite on cycle days 5-12 (after menstruation, before ovulation). A tenaculum is placed on the cervix and a cannula (acorn tip or balloon catheter) is inserted into the cervical canal. Water-soluble (Omnipaque, Conray) or oil-based (Lipiodol) contrast is injected under fluoroscopic guidance with serial spot images. Findings:
Normal: Smooth triangular uterine cavity, bilateral tubal fill (thin, serpentine tubes), and free peritoneal spill (contrast disperses widely into the peritoneal cavity).
Proximal tubal occlusion: Contrast stops at the cornual region (no tubal fill). Differential: true obstruction (salpingitis isthmica nodosa, endometriosis, prior ectopic), spasm (most common cause of false-positive proximal block — ~15%), or mucus/debris plug. Next step: repeat HSG with antispasmodic premedication (glucagon or buscopan), or selective tubal catheterization under fluoroscopy (resolves ~80% of proximal occlusions).
Distal tubal occlusion: Tube fills and distends without spill; clubbed or sausage-shaped appearance suggests hydrosalpinx. Thin-walled dilation with preserved mucosal folds has better prognosis than thick-walled, smooth-lumen hydrosalpinx (Mage score).
Loculated spill: Contrast pools in a restricted area adjacent to the tube rather than dispersing freely — suggests peritubal adhesions even if the tube is technically patent. Associated with reduced fecundity.
Intracavitary filling defects: Smooth, round = polyp; irregular = submucosal fibroid; linear/irregular with cavity distortion = synechiae (Asherman). Confirmed with SIS or hysteroscopy.
Salpingitis isthmica nodosa (SIN): Multiple small contrast-filled diverticula projecting from the isthmic portion of the tube. Associated with ectopic pregnancy risk and tubal infertility.
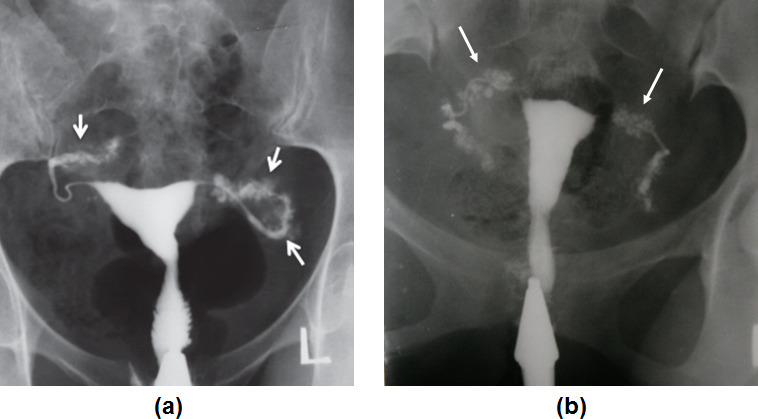
Endometriosis-Related Tubal Disease
Endometriosis can cause tubal factor through peritubal and periovarian adhesions, tubal distortion, and impaired fimbrial ovum pickup. The Endometriosis Fertility Index (EFI) predicts pregnancy rates after surgical treatment: factors include age, infertility duration, prior pregnancies, AFS/rASRM scores, and least-function scores for tubes, fimbriae, and ovaries. EFI ≥9: good prognosis for spontaneous conception post-surgery (~55% 3-year pregnancy rate); EFI ≤4: proceed to IVF (~15% 3-year rate with expectant management).
07 Uterine Factors
Fibroids (Leiomyomas)
Uterine fibroids (leiomyomas) affect ~70-80% of women by age 50, with higher prevalence in Black women (earlier onset, larger size, more symptomatic). Most fibroids are asymptomatic and do not impair fertility. The impact on reproduction depends primarily on location relative to the endometrial cavity:
Submucosal fibroids (FIGO type 0-2): Clearly impair implantation and increase miscarriage risk. Mechanisms: distortion of the endometrial cavity, altered endometrial vascularity, local inflammation, and impaired uterine contractility. Hysteroscopic myomectomy is recommended before fertility treatment (Pritts et al., meta-analysis 2009 — submucosal fibroids decrease pregnancy rates by ~70%; removal restores rates to baseline). Type 0 (pedunculated, entirely intracavitary) and Type 1 (>50% intracavitary) are straightforward hysteroscopic resections. Type 2 (≥50% intramural) may require two-stage resection or a combined hysteroscopic-laparoscopic approach.
Intramural fibroids (FIGO type 3-5): Evidence is conflicting. Fibroids >4-5 cm that distort the cavity (confirmed on SIS or MRI) may warrant myomectomy (abdominal or laparoscopic) before IVF. Non-cavity-distorting intramural fibroids <4 cm probably do not significantly affect IVF outcomes, but fibroids >5 cm (even non-distorting) may reduce pregnancy rates by ~10-15% in some studies. The decision to operate must weigh the benefit of fibroid removal against surgical risks (blood loss, adhesion formation, and the need for cesarean delivery if the myometrium is entered deeply).
Subserosal fibroids (FIGO type 6-7): Do not appear to impact fertility and generally do not require treatment in the infertility setting. Exception: very large subserosal fibroids that compress other pelvic structures or cause pain. Post-myomectomy considerations: Recommended waiting period of 3-6 months before conception to allow adequate scar healing. Cesarean delivery is generally recommended if the myomectrium was entered deeply during myomectomy (to avoid uterine rupture risk during labor, though evidence for this is limited to case reports).
Endometrial Polyps
Endometrial polyps are found in 6-8% of infertile women (higher with advancing age, tamoxifen use, and chronic anovulation). Polyps may impair fertility through mechanical interference with sperm transport or embryo implantation, local inflammatory changes, and altered endometrial receptivity. Polypectomy before IUI improves pregnancy rates (Perez-Medina et al., Hum Reprod 2005 — clinical pregnancy rate 63% vs 28% after polypectomy vs no treatment in the subsequent IUI cycle). The impact of small polyps (<1 cm) on IVF outcomes is less clear, but most REI practitioners remove polyps before embryo transfer. Hysteroscopic polypectomy is the standard approach — performed in the office or operating room, typically in the proliferative phase. Recurrence rate: ~10-15% within 1 year. Malignancy risk in reproductive-age polyps: <1% (higher in postmenopausal women).
Asherman Syndrome (Intrauterine Adhesions)
Asherman syndrome results from damage to the basalis layer of the endometrium, most commonly following post-pregnancy curettage (~90% of cases — particularly curettage for retained products of conception, missed abortion, or postpartum hemorrhage). Risk is highest when curettage is performed 2-4 weeks postpartum (when the endometrium is most vulnerable). Other causes: post-endometritis curettage, hysteroscopic myomectomy or septum resection (thermal injury), genital tuberculosis (in endemic regions — a common cause in India, Africa), and uterine artery embolization.
Symptoms: hypomenorrhea or amenorrhea (despite normal hormonal function), cyclic pelvic pain (if hematometra from outflow obstruction), infertility, and recurrent pregnancy loss. Diagnosed by HSG (irregular filling defects, "moth-eaten" appearance), SIS (adhesion bands seen within the distended cavity), or hysteroscopy (gold standard — directly visualizes adhesions). Classification systems: ESGE/AFS classification grades adhesions as mild (thin, filmy adhesions involving <1/4 of the cavity), moderate (thick adhesions ± partial occlusion, <3/4 of cavity), or severe (thick, dense adhesions obliterating ≥3/4 of the cavity, with agglutination of the walls).
Treatment: hysteroscopic adhesiolysis under ultrasound or concurrent laparoscopic guidance (to monitor depth and prevent perforation). Technique: sharp dissection with hysteroscopic scissors is preferred over electrosurgery or laser (less thermal injury to the basalis). Post-adhesiolysis measures to prevent re-formation: (1) Intrauterine balloon catheter (e.g., Foley catheter with 3-5 mL or Cook balloon stent) left in place for 5-14 days; (2) Estrogen therapy (estradiol 2-4 mg daily or conjugated estrogens 2.5 mg daily for 4-6 weeks, with medroxyprogesterone added for the last 10 days to induce withdrawal bleed); (3) Prophylactic antibiotics during stent placement; (4) Repeat "second-look" hysteroscopy at 4-8 weeks to assess healing and lyse any re-formed adhesions. For severe cases, multiple operative sessions may be required. Emerging therapies: platelet-rich plasma (PRP) intrauterine instillation, stem cell therapy, and amnion membrane grafts to promote endometrial regeneration — all investigational.
Prognosis: mild Asherman — menstrual function restores in ~90%, subsequent pregnancy rate ~60-70%. Moderate — ~70% menstrual restoration, ~30-40% pregnancy. Severe — <50% menstrual restoration, ~15-25% pregnancy rate. Gestational carrier may be needed for refractory cases. Pregnancies after adhesiolysis carry increased risk of placenta accreta spectrum (due to damaged endometrium).
Müllerian Anomalies
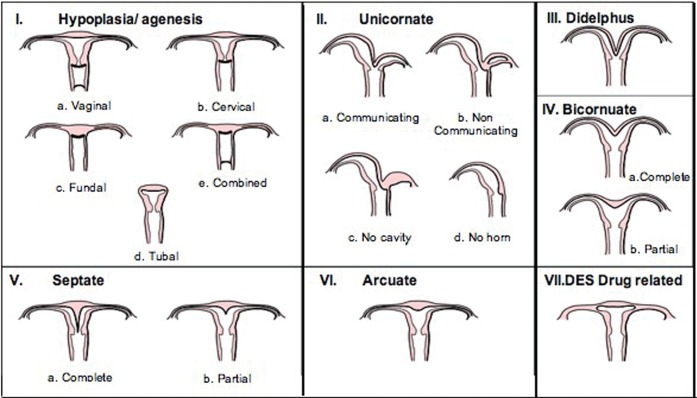
Congenital uterine anomalies result from abnormal development, fusion, or resorption of the Müllerian (paramesonephric) ducts. Prevalence: ~5-7% of the general population, ~8% in infertile women, ~13-25% in women with recurrent pregnancy loss. See Section 23 for detailed ASRM classification. Impact on fertility:
Septate uterus: Most common anomaly associated with adverse reproductive outcomes. The septum has poor vascularity. Hysteroscopic septum resection is recommended for recurrent pregnancy loss and may benefit patients with infertility/implantation failure (though RCT evidence is debated — TRUST trial, NEJM 2021, showed no benefit for septum resection in preventing miscarriage). Bicornuate uterus: Associated with second-trimester loss, preterm delivery, malpresentation; cerclage may be needed. Unicornuate uterus: Associated with increased ectopic, miscarriage, preterm delivery; a rudimentary horn may contain endometrium (risk of ectopic pregnancy in the horn). Uterine didelphys: Generally best reproductive outcomes among major anomalies; may carry pregnancies to term.
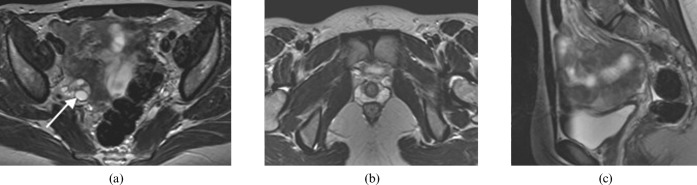
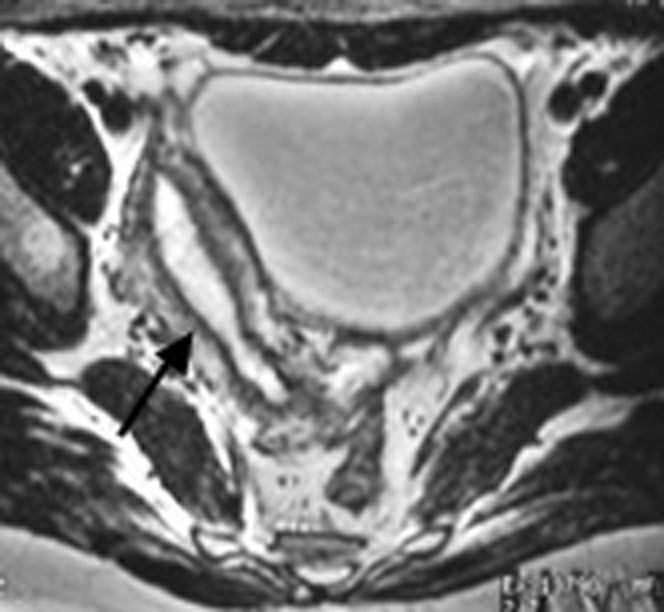
Adenomyosis
Adenomyosis (invasion of endometrial glands and stroma into the myometrium) is increasingly recognized as a cause of infertility and IVF failure. Prevalence: estimated 20-35% of reproductive-age women (likely underdiagnosed). Pathophysiology: disruption of the junctional zone impairs uterine peristalsis and sperm transport, and may alter endometrial receptivity. MRI is the gold standard for diagnosis (junctional zone thickness >12 mm; features include diffuse thickening, myometrial cysts, and heterogeneous signal). Transvaginal ultrasound features include heterogeneous myometrium, myometrial cysts, asymmetric wall thickening, and poor endomyometrial border definition. A meta-analysis by Vercellini et al. (2014) showed adenomyosis reduced IVF clinical pregnancy rates by ~30%. Treatment strategies for fertility: (1) GnRH agonist suppression for 2-3 months before frozen embryo transfer may improve outcomes (Niu et al., 2013); (2) adenomyomectomy for focal adenomyomas is technically challenging and not widely performed for fertility; (3) high-dose progestins (dienogest) before FET is under investigation.
08 Endometriosis & Infertility
Pathophysiology of Endometriosis-Associated Infertility
Endometriosis — the presence of endometrial-like tissue outside the uterine cavity — affects ~6-10% of reproductive-age women and is found in up to 25-50% of infertile women. The leading theories of pathogenesis include: retrograde menstruation (Sampson theory) — reflux of menstrual endometrial fragments through the fallopian tubes into the peritoneal cavity; coelomic metaplasia — transformation of peritoneal mesothelium into endometrial tissue; and lymphovascular dissemination — explaining distant sites (pleura, brain). Likely a combination of retrograde menstruation (universal phenomenon) with impaired immune clearance and genetic predisposition.
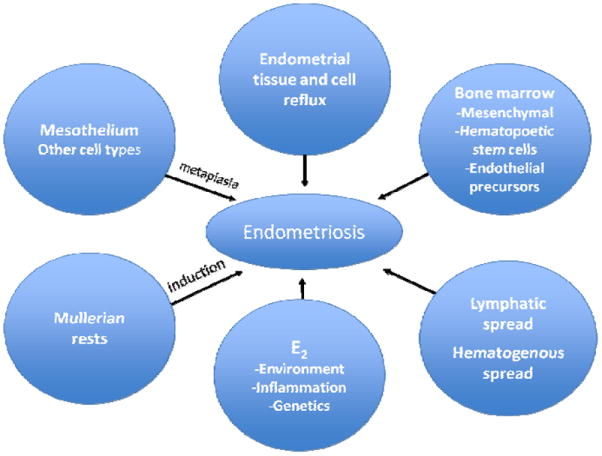
Multiple mechanisms impair fertility: peritoneal inflammation (elevated IL-1, IL-6, TNF-α, prostaglandins in peritoneal fluid; activated macrophages that phagocytose sperm), distorted pelvic anatomy (adhesions affecting tuboovarian relationships, tubal damage), impaired oocyte quality (oxidative stress, altered follicular fluid cytokine milieu), altered endometrial receptivity (progesterone resistance — downregulation of progesterone receptors, impaired decidualization, altered integrin expression during the implantation window), and disrupted sperm function (decreased motility in inflammatory peritoneal fluid).
rASRM Staging (Revised American Society for Reproductive Medicine)
The rASRM classification assigns point scores based on location, size, and depth of implants, as well as adhesion density and extent:
Stage I (Minimal): 1-5 points. Isolated implants, no significant adhesions.
Stage II (Mild): 6-15 points. Superficial implants <5 cm, no significant adhesions.
Stage III (Moderate): 16-40 points. Deep implants, endometriomas, filmy adhesions.
Stage IV (Severe): >40 points. Deep implants, large endometriomas, dense adhesions, cul-de-sac obliteration.
Limitation: rASRM staging poorly correlates with fertility outcomes — a patient with stage I endometriosis may have significant infertility while one with stage III may conceive spontaneously. The Endometriosis Fertility Index (EFI) is better at predicting fertility after surgical treatment.
Medical Suppression vs. Surgery for Fertility
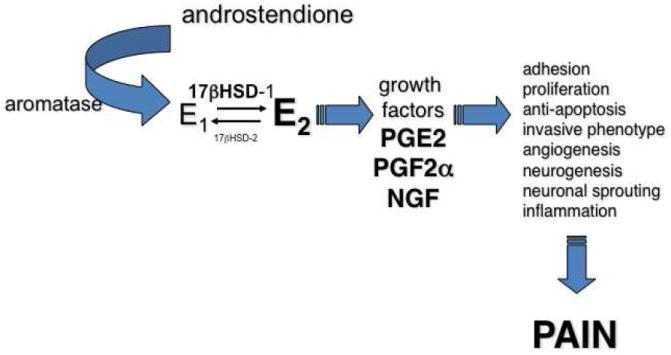
Medical suppression of endometriosis (GnRH agonists, progestins, OCPs, aromatase inhibitors) does not improve fertility and should not be used as a fertility treatment — these medications are all contraceptive by design. There is no evidence that preoperative or postoperative medical suppression improves fertility outcomes (it merely delays conception attempts). Exception: prolonged GnRH agonist downregulation (3-6 months) before IVF may improve implantation in moderate-severe endometriosis (Sallam et al., Cochrane 2006 — increased live birth rate).
Surgical treatment for fertility in endometriosis: the evidence depends on disease severity. Minimal-mild (Stage I-II): Laparoscopic ablation or excision of visible implants improves pregnancy rates vs diagnostic laparoscopy alone (Marcoux et al., NEJM 1997 — RCT, 30.7% vs 17.7% cumulative pregnancy at 36 weeks; NNT = 8). However, a subsequent Italian RCT by Parazzini et al. (1999) showed no significant benefit, leaving the question somewhat unresolved. ESHRE guidelines recommend surgical treatment of endometriosis at the time of diagnostic laparoscopy. Moderate-severe (Stage III-IV): No RCT data exist. Observational studies show crude pregnancy rates of 35-65% after complete excision in experienced centers. Surgery before IVF is debated — potential benefits (improved anatomy, reduced inflammation) must be weighed against risks (adhesion formation, reduced ovarian reserve from cystectomy). The decision should be individualized based on age, symptoms, reserve markers, and surgical complexity.
Endometriomas
Endometriomas (endometriotic ovarian cysts, "chocolate cysts") are found in 17-44% of women with endometriosis. They arise from invagination and progressive accumulation of menstrual-like debris within the ovarian cortex. Management (≥3-4 cm) is a major clinical dilemma:
Surgical excision (cystectomy): The stripping technique (identifying the plane between the cyst wall/pseudocapsule and healthy cortex, then stripping with traction/countertraction) removes the cyst wall more completely, reducing recurrence (~10-20% at 2 years vs ~40-60% with drainage/ablation alone). However, cystectomy unavoidably removes a rim of healthy ovarian cortex containing primordial follicles: post-cystectomy AMH declines by ~30-50% (Raffi et al., meta-analysis 2012), and the effect is worse with bilateral cystectomy or repeat surgery. The "fire and ice" technique (bipolar coagulation at the hilum + laser/hemostatics elsewhere) minimizes thermal damage to surrounding follicles. New techniques: sclerotherapy (ethanol or tetracycline injection after aspiration) and laser ablation of the cyst wall with CO2 or plasma energy may preserve more ovarian tissue.
ASRM and ESHRE guidelines: cystectomy for endometriomas ≥4 cm is indicated primarily for diagnostic confirmation (to exclude malignancy — rare but possible, especially in women >40), symptom relief, and to improve access to follicles during oocyte retrieval. Cystectomy is NOT routinely recommended before IVF if the endometrioma is <4 cm, the patient has no pain, and follicle access is adequate. IVF outcomes with endometriomas <4 cm in situ are similar to those without endometriomas. Aspiration of endometriomas before or during oocyte retrieval is associated with high recurrence and infection risk and is generally not recommended. If the endometrioma is traversed during retrieval, prophylactic antibiotics and monitoring for abscess formation are essential.
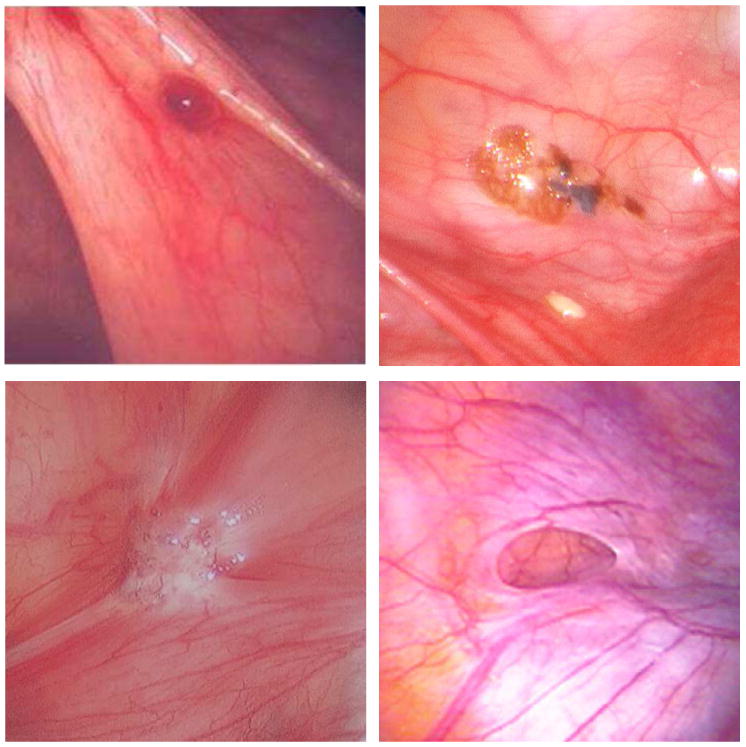
Deep Infiltrating Endometriosis (DIE)
DIE involves invasion >5 mm below the peritoneal surface, affecting the rectovaginal septum (most common location), uterosacral ligaments, bladder (wall and detrusor), bowel (rectosigmoid in ~90% of bowel DIE; appendix, cecum, small bowel less commonly), and ureters (extrinsic compression or intrinsic invasion — can cause silent hydronephrosis). Symptoms: severe dysmenorrhea, deep dyspareunia, dyschezia (painful bowel movements, especially catamenial), hematuria, and infertility.
Diagnosis: MRI with specific protocols (sagittal T2-weighted sequences, gel-filled vagina for rectovaginal assessment) and transvaginal ultrasound by experienced sonographer (with bowel preparation for bowel assessment — "sliding sign" negative indicates cul-de-sac obliteration). Both modalities have sensitivity >90% for DIE when performed by experts. Renal ultrasound should be performed to exclude ureteral involvement and hydronephrosis. Surgery for DIE is complex (may require colorectal surgery for shaving, disc excision, or segmental resection; urology for ureteral reimplantation) and should be performed in multidisciplinary centers of expertise. Complication rates: anastomotic leak 1-3%, neurogenic bladder dysfunction 5-10% after deep pelvic dissection.
The impact of DIE surgery on fertility is uncertain: retrospective data suggest improved natural conception rates post-surgery, but no RCTs exist comparing surgery to direct IVF. IVF may be preferred when fertility is the primary goal (particularly in women ≥35, those with additional infertility factors, or when surgery carries high complication risk). If surgery is performed, an EFI score should be calculated to guide post-operative management.
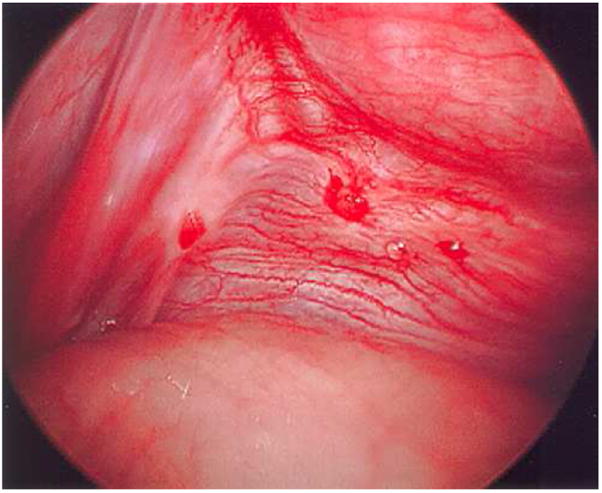
IVF Outcomes with Endometriosis
Stage III-IV endometriosis is associated with ~10-20% lower IVF pregnancy rates compared to tubal factor infertility. Long GnRH agonist downregulation (3-6 months of suppression before IVF stimulation) may improve outcomes in moderate-severe endometriosis (Sallam et al., Cochrane 2006). Oocyte quality rather than endometrial receptivity appears to be the primary issue, as donor oocyte recipients with endometriosis have similar outcomes to those without. Fewer oocytes are typically retrieved in endometriosis patients, and fertilization rates may be lower. Management considerations: avoid traversing endometriomas during retrieval if possible (infection risk), pre-treat with GnRH agonist if severe disease, and consider freeze-all if endometrioma aspiration occurs.
09 Unexplained Infertility
Definition & Prevalence
Unexplained infertility is a diagnosis of exclusion, made when standard evaluation reveals no identifiable cause: (1) documented regular ovulation (regular cycles, mid-luteal progesterone >3 ng/mL, or positive ovulation predictor kit); (2) patent fallopian tubes (by HSG or laparoscopy); (3) normal uterine cavity (by HSG, SIS, or hysteroscopy); (4) normal semen analysis (per WHO 6th edition criteria); and (5) adequate ovarian reserve (AMH >1.0, AFC ≥7, FSH <10). It accounts for ~15-30% of infertile couples — the proportion varies depending on how thorough the evaluation is (more testing = fewer unexplained cases).
Possible occult mechanisms that explain "unexplained" infertility: subtle oocyte or sperm dysfunction not detected by standard testing (sperm DNA fragmentation, impaired acrosome reaction, zona binding defects), impaired fertilization (only detectable with IVF), subclinical/minimal endometriosis (not diagnosed without laparoscopy), altered endometrial receptivity (window of implantation displacement — only detectable with ERA testing), cervical factor (immunologic or mucus quality issues), impaired tubal function (intact tubes but impaired peristalsis or ciliary function), and peritoneal factors (mild adhesions, altered peritoneal fluid environment).
Empiric Treatment
Treatment is typically escalated in a stepwise fashion based on age and duration of infertility:
Step 1 — Ovarian stimulation + IUI: Clomiphene citrate or letrozole + IUI for 3-4 cycles. Per cycle pregnancy rate: ~8-10%. The AMIGOS trial (Fertility and Sterility, 2015) found clomiphene + IUI, letrozole + IUI, and gonadotropins + IUI had similar live birth rates per cycle (~8-10%), but gonadotropins had higher multiple pregnancy rates.
Step 2 — Gonadotropins + IUI: If oral medications fail after 3-4 cycles. Per cycle pregnancy rate: ~10-15%. Higher multiple pregnancy risk (20-30%). Careful monitoring with follicular tracking is essential; cancel cycle if >3 dominant follicles.
Step 3 — IVF: After 3-6 cycles of failed IUI, or sooner based on age/duration. IVF per cycle pregnancy rate: ~40-50% in women <35. The FASTT trial (Fertility and Sterility, 2010) demonstrated that accelerated treatment to IVF (after 3 clomiphene/IUI cycles) was more cost-effective than extended superovulation/IUI in couples with unexplained infertility, particularly for women ≥38.
Prognosis by Age
Age is the single most important prognostic factor. In women <35 with unexplained infertility of <3 years duration, cumulative live birth rates with expectant management alone approach ~30-50% over 12 months. In women ≥38, treatment escalation should be accelerated (consider IVF after 3 IUI cycles or immediately for women ≥40). The monthly fecundability of untreated couples with unexplained infertility is approximately 2-4%, compared to ~15-20% in normally fertile couples.
IUI Technique & Considerations
Intrauterine insemination (IUI) involves processing the semen sample (density gradient centrifugation or swim-up technique to select motile sperm), then depositing the washed sample directly into the uterine cavity via a thin catheter. Timing: performed 24-36 hours after hCG trigger or LH surge detection. Total motile sperm count (TMSC) after processing: ≥5 million motile sperm is optimal for IUI; <1-2 million is associated with very low success rates — proceed to IVF/ICSI. Per-cycle success rates: ~8-15% (depending on age, diagnosis, and stimulation protocol). Common side effects: mild cramping. Serious complications rare (infection <0.5%). Maximum recommended IUI cycles before IVF: 3-6 (age-dependent — fewer in older patients).
10 Recurrent Pregnancy Loss
Definition & Epidemiology
Recurrent pregnancy loss (RPL) is defined by ASRM as two or more clinical pregnancy losses (documented by ultrasound or histopathology — this includes missed abortions and documented gestational sacs, not chemical pregnancies). ESHRE defines RPL as three or more pregnancy losses (not necessarily consecutive). The ASRM definition (two losses) lowers the threshold for evaluation because the risk of a subsequent loss after two losses (~25-30%) is similar to the risk after three losses (~30-35%), and early evaluation may identify treatable conditions sooner.
RPL affects 1-2% of couples trying to conceive. Important distinction: sporadic miscarriage (the most common complication of pregnancy, affecting ~15-25% of clinically recognized pregnancies; primarily caused by embryonic aneuploidy, especially trisomy) vs. recurrent pregnancy loss (implies a potentially recurrent underlying cause, though recurrent embryonic aneuploidy remains the most common etiology even in RPL). The probability of a live birth after RPL is generally favorable: even after 3 consecutive losses with no identified cause, the chance of a successful next pregnancy is ~60-75% (depending on maternal age and prior obstetric history).
Etiologic Workup
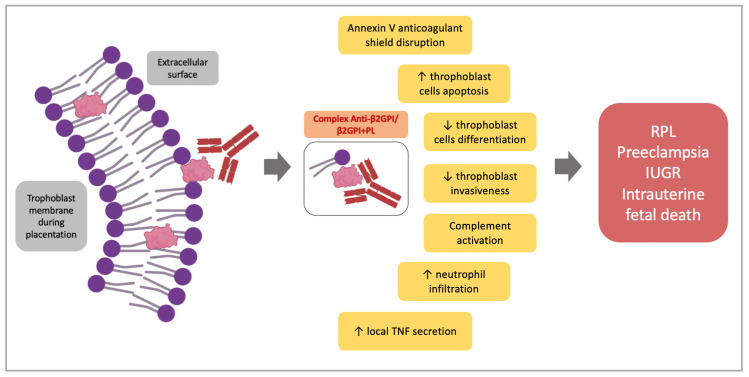
Genetic (3-5%): Parental karyotyping identifies balanced translocations (robertsonian, reciprocal) or inversions in 3-5% of couples with RPL; genetic counseling and PGT-SR with IVF are offered to reduce the risk of future unbalanced conceptions. Products of conception (POC) karyotype or chromosomal microarray on the loss tissue identifies the cause in up to 50-60% of losses — aneuploidy is the single most common finding (especially trisomy 16, 22, and monosomy X). POC testing helps direct subsequent evaluation: if the loss was aneuploid, parental factors are less likely causative; if the loss was euploid, maternal factors (APS, uterine, endocrine) should be investigated more aggressively.
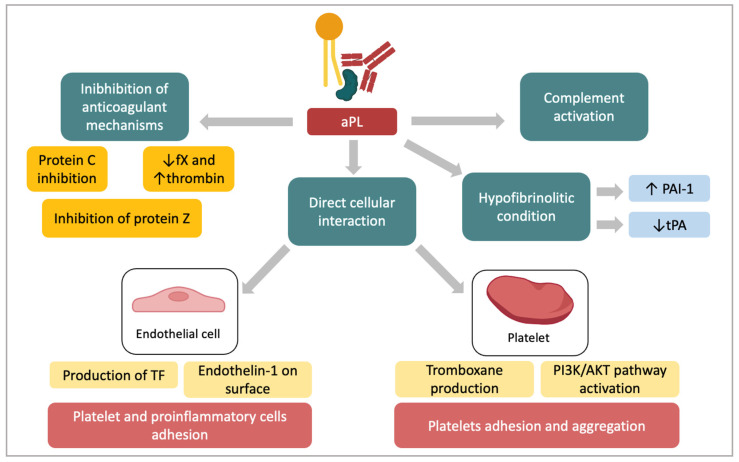
Antiphospholipid syndrome (5-15% of RPL): The most important treatable cause of RPL. Sapporo/Sydney criteria require at least one clinical event plus persistent positivity (≥12 weeks apart) of lupus anticoagulant, anticardiolipin antibodies (IgG/IgM, medium-high titer), and/or anti-β2-glycoprotein I antibodies. Treatment: low-dose aspirin (81 mg, started preconception) + prophylactic heparin (unfractionated heparin 5,000 IU SC BID or enoxaparin 40 mg SC daily, started at positive pregnancy test) — improves live birth rate from ~10-15% to ~70-80%. Continue heparin until 6 weeks postpartum (VTE risk). Triple-positive APS (LA + aCL + anti-β2GP1) carries the highest risk and may benefit from higher-dose anticoagulation.
Uterine anatomy (10-15%): Evaluate with SIS, hysteroscopy, or 3D ultrasound/MRI for septate uterus, intrauterine adhesions, submucosal fibroids. Septum resection is traditionally recommended for RPL (though TRUST trial challenged this); adhesiolysis for Asherman syndrome; myomectomy for cavity-distorting submucosal fibroids.
Thrombophilia (inherited): Factor V Leiden (most common, ~5% Caucasian prevalence), prothrombin gene mutation (G20210A), protein C/S deficiency, antithrombin III deficiency — evidence for treatment (LMWH) in inherited thrombophilia is weak; ALIFE2 trial (NEJM 2023) showed no benefit of LMWH for RPL with inherited thrombophilia. Routine thrombophilia screening is controversial and not universally recommended.
Endocrine: TSH and TPO antibodies (subclinical hypothyroidism and thyroid autoimmunity each increase miscarriage risk; treat with levothyroxine to maintain TSH <2.5). Uncontrolled diabetes (HbA1c >8%) increases miscarriage risk — optimize glycemic control preconception. Hyperprolactinemia should be treated.
Progesterone: The PRISM trial (Coomarasamy et al., NEJM 2019) showed vaginal micronized progesterone (400 mg BID) in women with early pregnancy bleeding significantly increased live birth rates in the subgroup with ≥3 prior miscarriages (72% vs 57%). Progesterone support from time of positive pregnancy test through 16 weeks is now widely recommended for women with RPL.
All couples: Parental karyotypes, APS panel (LA, aCL, anti-β2-GP1), uterine anatomy assessment (SIS/hysteroscopy), TSH, TPO antibodies.
Consider: POC genetic analysis, thrombophilia panel (Factor V Leiden, prothrombin G20210A, protein C/S, antithrombin III), HbA1c (uncontrolled diabetes), prolactin.
Not recommended routinely: NK cell testing, HLA typing, paternal leukocyte immunization, IVIg — insufficient evidence of benefit.
11 Semen Analysis & Interpretation
Collection & Processing
Semen specimens should be collected by masturbation into a sterile, non-toxic, wide-mouthed container after 2-5 days of abstinence. Longer abstinence (>5 days) increases volume and concentration but decreases motility and increases DNA fragmentation; shorter abstinence (<2 days) may reduce volume and concentration. Specimen must be kept at body temperature (20-37°C) and delivered to the laboratory within 60 minutes (ideally 30 minutes). Liquefaction (normally complete within 20-30 minutes; if not liquefied by 60 minutes, mechanical mixing or bromelain treatment is used) must occur before analysis. A minimum of two analyses, 2-4 weeks apart, are needed to account for the 70-day spermatogenesis cycle and significant intra-individual variability (up to 100% variation in sperm count between samples from the same individual).
Semen Processing for ART
Raw semen cannot be inseminated directly into the uterus (contains prostaglandins causing severe uterine cramping, and potentially infectious agents). Processing techniques: Density gradient centrifugation: Layered over colloidal silica gradient (e.g., 40/80% PureSperm); centrifuged to separate motile sperm from seminal plasma, debris, and non-motile sperm. Pellet is resuspended in culture medium. Best for normal to moderately abnormal samples. Swim-up technique: Washed sperm pellet is overlaid with culture medium; motile sperm "swim up" into the medium over 30-60 minutes and are collected. Selects highly motile sperm but lower yield. Best for normal samples. Magnetic-activated cell sorting (MACS): Uses annexin V-conjugated magnetic beads to remove apoptotic sperm. Emerging technique for selecting sperm with lower DNA fragmentation. Total motile sperm count (TMSC) after processing is the key parameter for treatment decisions: TMSC ≥10 million — suitable for IUI; TMSC 5-10 million — borderline (IUI may still be attempted); TMSC <5 million — IVF/ICSI recommended; TMSC <1 million — ICSI only.
Parameters & Interpretation
Refer to WHO 6th edition reference values in Section 4. Key interpretive points: Severe oligozoospermia (<5 million/mL) warrants genetic evaluation (karyotype, Y-microdeletion, CFTR). Azoospermia requires determination of obstructive vs non-obstructive etiology through history, physical exam (testicular size, vas deferens presence), hormonal evaluation (FSH, testosterone), and potentially testicular biopsy. Isolated teratozoospermia with strict criteria <4% has limited predictive value for IUI but ICSI should be considered if morphology is <1%. Pyospermia (WBC >1 million/mL) may indicate genital tract infection — culture and antibiotics considered.
Advanced Sperm Testing
DNA fragmentation testing (TUNEL, SCD/Halosperm, SCSA/DFI): Elevated fragmentation (>30% by SCSA, or >36% DFI) is associated with reduced natural fertility, lower IUI success, and possibly lower IVF/ICSI outcomes. Causes: varicocele, infection, oxidative stress, advanced paternal age, environmental toxins, smoking. Interventions that may reduce fragmentation: varicocelectomy, antioxidants, lifestyle modification (smoking cessation, weight loss), shorter abstinence intervals (daily ejaculation for 2-4 days before sample), or use of testicular sperm (which has lower fragmentation than ejaculated sperm because DNA damage accumulates during epididymal transit). Reactive oxygen species (ROS) testing: Oxidative stress impairs sperm motility and membrane integrity; measured by chemiluminescence assays or the MiOXSYS system. Hypo-osmotic swelling test (HOS): Assesses membrane integrity of immotile sperm by exposing them to hypotonic solution; tail swelling indicates a live cell with an intact membrane — useful in selecting viable immotile sperm for ICSI in cases of complete asthenozoospermia or immotile cilia syndrome.
12 Male Factor Conditions
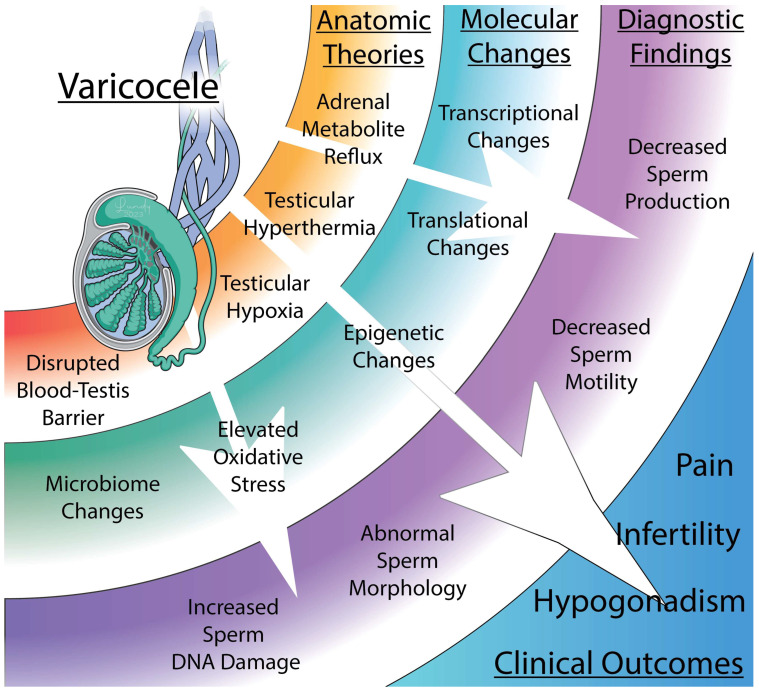
Varicocele
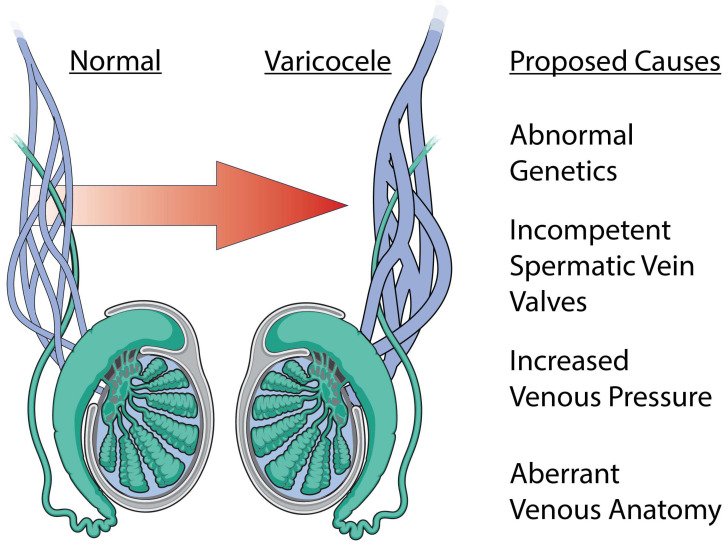
Varicocele (dilation of the pampiniform plexus of the spermatic cord veins) is found in ~15% of the general male population, ~35-40% of men with primary infertility, and ~75-80% of men with secondary infertility. Left-sided predominance (~80-90%) due to the left testicular (internal spermatic) vein draining into the left renal vein at a perpendicular angle (compared to the right testicular vein which drains obliquely into the IVC). Bilateral varicoceles are found in ~30-40% of affected men.
Grading: Grade I — palpable only with Valsalva maneuver; Grade II — palpable without Valsalva; Grade III — visible as a "bag of worms" through scrotal skin. Pathophysiology of infertility: venous stasis leads to increased scrotal temperature (testes require 2-3°C below core body temperature for optimal spermatogenesis), reflux of adrenal and renal metabolites, hypoxia, and oxidative stress.
Repair indications (AUA/ASRM guidelines): (1) Palpable varicocele (subclinical varicocele repair is NOT indicated), (2) Abnormal semen parameters on at least one analysis, (3) Female partner with normal fertility or a correctable infertility factor, (4) Couple is attempting to conceive. Surgical approaches: microsurgical subinguinal varicocelectomy (gold standard — operating microscope allows identification and preservation of testicular artery and lymphatics; recurrence rate <1%, hydrocele rate <1%); inguinal approach (with or without microscope); laparoscopic (higher recurrence rate); percutaneous embolization (interventional radiology — alternative for patients who prefer non-surgical approach or have recurrent varicocele). Post-repair improvement: ~60-70% show improved semen parameters by 3-6 months; ~30-40% achieve spontaneous pregnancy within 1-2 years. Varicocelectomy may also improve testosterone levels by ~100 ng/dL.
Obstructive Azoospermia (OA)
Characterized by normal spermatogenesis but blocked outflow. Key distinguishing features from NOA: FSH is usually normal (<7.6 mIU/mL), testicular volume is normal (≥15 mL), and testicular biopsy (if performed) shows full spermatogenesis. Causes:
Congenital bilateral absence of the vas deferens (CBAVD): Present in ~1-2% of infertile men and ≥97% of males with cystic fibrosis. Physical exam reveals absent vas deferens bilaterally on palpation. Semen analysis shows low volume (<1 mL), acidic pH, azoospermia, and absent fructose (seminal vesicles are often absent or atrophied). CFTR mutation screening is mandatory for the patient AND partner (if both carry CFTR mutations, offspring may have classic CF — PGT-M with IVF or donor sperm should be discussed). Renal ultrasound recommended (unilateral renal agenesis in ~10-15% of CBAVD without classic CF mutations). Fertility achieved by MESA or TESE + ICSI (vas reconstruction is not possible).
Vasectomy: The most common cause of OA worldwide. Options: reversal or sperm retrieval + IVF/ICSI. Vasectomy reversal: vasovasostomy (end-to-end reanastomosis of the vas) when fluid from the testicular end contains sperm or is clear; vasoepididymostomy (bypass — vas anastomosed to epididymis) when fluid is thick/pasty or absent, suggesting secondary epididymal obstruction. Success depends on: time since vasectomy (<3 years: ~97% patency, ~76% pregnancy rate; 3-8 years: ~88% patency, ~53% pregnancy; 9-14 years: ~79%, ~44%; >15 years: ~71%, ~30% — from Vasectomy Reversal Study Group data), surgeon microsurgical expertise, and need for vasoepididymostomy (lower success than vasovasostomy). Cost-effectiveness: reversal may be more cost-effective than IVF when interval is short and female partner is young.
Ejaculatory duct obstruction: Diagnosed by low-volume (<1.5 mL), azoospermic, acidic ejaculate with dilated seminal vesicles (>1.5 cm) on TRUS. May be caused by Müllerian duct cysts (midline prostatic cysts), Wolffian duct cysts, calcification, or post-inflammatory stenosis. Treatment: transurethral resection of the ejaculatory ducts (TURED) — success rate ~50-75% for sperm return to ejaculate. Complications: retrograde ejaculation, recurrent epididymitis, and watery ejaculate.
Non-Obstructive Azoospermia (NOA)
Characterized by impaired spermatogenesis due to intrinsic testicular failure. Distinguishing features: FSH is typically elevated (>7.6 mIU/mL — reflecting loss of inhibin B negative feedback from Sertoli cells), testicular volume is often reduced (<15 mL bilaterally), and testosterone may be low-normal or low. NOA accounts for ~60% of all azoospermia cases.
Genetic causes (15-30% of NOA):
Klinefelter syndrome (47,XXY): Most common chromosomal abnormality in men (1:600 live male births) and the most common genetic cause of NOA (~10-15% of azoospermic men). Classic features: tall stature, small firm testes (typically <6 mL bilateral), gynecomastia, long limbs, learning difficulties (particularly verbal). Testosterone is low-normal to low; FSH and LH are elevated. Spermatogenesis: most patients are azoospermic but ~8% have rare sperm in the ejaculate (especially in mosaic 46,XY/47,XXY). Micro-TESE retrieves sperm in ~40-70% of Klinefelter patients (higher success rates in younger patients and those with higher testosterone levels). Retrieved sperm are used for ICSI; PGT-A is recommended as Klinefelter-derived sperm have higher aneuploidy rates. Timing: there is emerging evidence that testicular function declines progressively in Klinefelter patients, leading to recommendations for earlier sperm banking or TESE (even in adolescents in some protocols).
Y chromosome microdeletions: Found in ~10-15% of azoospermic men and ~5-10% of severely oligozoospermic men. Must be tested before TESE (results determine prognosis). Three critical regions on the long arm of the Y chromosome (Yq11): AZFa (proximal) — complete deletion = Sertoli cell-only syndrome (no germ cells present); sperm retrieval rate ~0% with TESE; TESE is NOT recommended; AZFb (middle) — complete deletion = maturation arrest (spermatogenesis halts before mature sperm are formed); sperm retrieval rate ~0-5%; TESE generally NOT recommended for complete AZFb deletions; AZFc (distal) — the most common deletion (~60% of Y microdeletions); variable phenotype from severe oligozoospermia to azoospermia; sperm retrieval rate ~50-70% with micro-TESE. Critical counseling point: AZFc deletions will be transmitted to ALL male offspring conceived via ICSI, who will also have impaired spermatogenesis. Partial AZFc deletions (e.g., gr/gr deletion) are associated with a 2-3x increased risk of impaired spermatogenesis but are considered a risk factor rather than a definitive cause.
Histologic patterns on testicular biopsy: (1) Sertoli cell-only (SCO) — complete absence of germ cells; only Sertoli cells lining the seminiferous tubules; worst prognosis but micro-TESE may find focal spermatogenesis in ~20-30% (due to heterogeneous distribution). (2) Maturation arrest (early = arrest at spermatogonia/spermatocyte stage; late = arrest at spermatid stage); micro-TESE success: ~30-50% for late arrest, lower for early arrest. (3) Hypospermatogenesis — all stages of spermatogenesis present but in reduced quantity; best prognosis among NOA patterns; micro-TESE success ~70-80%.
Ejaculatory Disorders
Retrograde ejaculation: Semen enters the bladder due to failure of bladder neck closure (post-orgasm urine shows sperm — obtain post-ejaculatory urine for analysis). Causes: diabetes mellitus (autonomic neuropathy), medications (alpha-blockers, antidepressants), post-surgical (TURP, retroperitoneal lymph node dissection for testicular cancer). Treatment: sympathomimetic agents (pseudoephedrine 60 mg QID, imipramine 25-50 mg TID) taken 3-5 days before planned collection to promote bladder neck closure; or alkalinized urine sperm recovery (patient alkalinizes urine with sodium bicarbonate, urine is collected post-ejaculation, sperm are processed for IUI/IVF). Anejaculation: Complete inability to ejaculate. Causes include spinal cord injury (most common), multiple sclerosis, psychogenic. Treatment: penile vibratory stimulation (PVS — first-line for spinal cord injury above T10; success ~80%) or electroejaculation (EEJ — rectal probe stimulation under anesthesia; success ~90% for spinal cord injury). Sperm quality from EEJ is typically poor; ICSI may be required.
Oxidative Stress & Male Infertility
Excessive reactive oxygen species (ROS) damage sperm DNA, lipid membranes, and proteins. Sources: leukocytospermia, varicocele, environmental toxins, smoking, obesity. The balance between ROS production and antioxidant defense (SOD, catalase, glutathione peroxidase in seminal plasma) determines oxidative stress. Treatment: lifestyle modification (smoking cessation, weight loss, exercise), antioxidant supplementation (vitamins C and E, CoQ10, L-carnitine, selenium, zinc — evidence is moderate quality but widely used), treatment of varicocele, and antibiotics for infection-related leukocytospermia.
13 Male Hormonal Evaluation & Treatment
Hormonal Assessment
Initial male hormonal panel (drawn in the morning, 8-10 AM, fasting — testosterone exhibits diurnal variation with peak in early morning): total testosterone (low if <264 ng/dL per AUA 2018 guidelines; must be confirmed on two separate morning draws), FSH, and LH. Interpretation:
Elevated FSH + low testosterone + small testes: Primary testicular failure (hypergonadotropic hypogonadism). Klinefelter, Y-microdeletion, prior orchitis, cryptorchidism, radiation/chemotherapy damage.
Low FSH + low LH + low testosterone: Hypogonadotropic hypogonadism (secondary). Pituitary/hypothalamic cause — MRI, prolactin, check for exogenous testosterone/anabolic steroid use, opioids, hemochromatosis.
Elevated FSH + normal testosterone + normal-to-small testes: Isolated spermatogenic failure with preserved Leydig cell function. Common in NOA with focal spermatogenesis.
Normal FSH + normal testosterone + azoospermia: Suggestive of obstructive azoospermia (spermatogenesis intact, inhibin B feedback preserved).
Additional tests as indicated: prolactin (if testosterone low — rule out prolactinoma), estradiol (if gynecomastia — elevated estrogen may indicate aromatase excess or liver disease), DHEA-S (if adrenal pathology suspected), free testosterone and SHBG (if total T borderline — SHBG elevations from liver disease, hyperthyroidism, or aging may mask true hypogonadism), inhibin B (marker of Sertoli cell function; low levels correlate with impaired spermatogenesis but not routinely measured).
Hypogonadotropic Hypogonadism
In men, hypogonadotropic hypogonadism (HH) causes azoospermia or severe oligozoospermia with low gonadotropins. Congenital HH includes Kallmann syndrome (anosmia, GnRH deficiency — KAL1, FGFR1 genes) and idiopathic HH (normal olfaction). Acquired causes: pituitary tumor (prolactinoma, non-functioning adenoma), hemochromatosis (iron deposition in pituitary and testes), opioid-induced hypogonadism (chronic opioid use suppresses GnRH — increasingly common), anabolic steroid/exogenous testosterone use, obesity (aromatization of testosterone to estrogen suppresses HPG axis), and head trauma. Treatment for fertility: hCG (1,000-2,000 IU SC 2-3 times per week — stimulates Leydig cell testosterone production, which raises intratesticular testosterone to support spermatogenesis) + recombinant FSH (75-150 IU SC 3 times per week — directly stimulates Sertoli cells to support spermatogenesis) if spermatogenesis does not initiate with hCG alone after 6-12 months. Recovery of spermatogenesis takes 6-24 months (longer if pre-pubertal onset HH). Monitoring: serum testosterone (target mid-normal range), semen analysis every 3 months. Prior testosterone use must be discontinued (exogenous testosterone at supraphysiologic or even replacement doses suppresses FSH/LH and creates an intratesticular testosterone deficit that halts spermatogenesis — recovery may take 3-12+ months, and ~5-10% may not fully recover). Alternative for hypogonadal men desiring fertility while awaiting recovery: clomiphene citrate 25-50 mg daily (off-label — stimulates endogenous gonadotropin secretion via hypothalamic estrogen receptor blockade).
Hyperprolactinemia in Males
Elevated prolactin (>20 ng/mL) suppresses GnRH pulsatility and can cause hypogonadism, decreased libido, and impaired spermatogenesis. Causes: prolactinoma (usually macroadenoma in men, as diagnosis is often delayed), medications (antipsychotics, metoclopramide). Treatment: cabergoline (0.25-1 mg twice weekly) is first-line for prolactinoma, superior to bromocriptine in efficacy and tolerability. MRI pituitary is indicated.
14 Surgical Sperm Retrieval
Techniques & Indications
TESA (Testicular Sperm Aspiration): Percutaneous needle aspiration of testicular tissue. Simple, office-based. Best for obstructive azoospermia. Sperm yield may be limited. TESE (Testicular Sperm Extraction): Open biopsy with extraction of testicular tissue. Provides more tissue than TESA. Used in both OA and NOA. Micro-TESE (Microsurgical TESE): Uses operating microscope to identify dilated seminiferous tubules (more likely to contain sperm) under 15-25x magnification. Gold standard for NOA — higher sperm retrieval rates (40-60%) compared to conventional TESE (25-30%) with less tissue removed and less vascular injury. MESA (Microsurgical Epididymal Sperm Aspiration): Aspiration of sperm from the epididymis under microscopic guidance. Provides high-quality motile sperm; ideal for OA (especially CBAVD). Yields large quantities sufficient for cryopreservation.
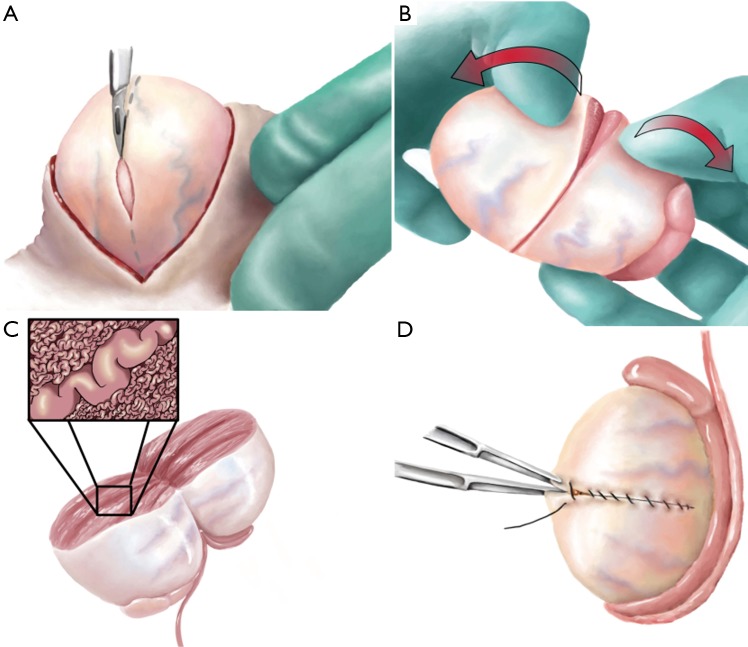
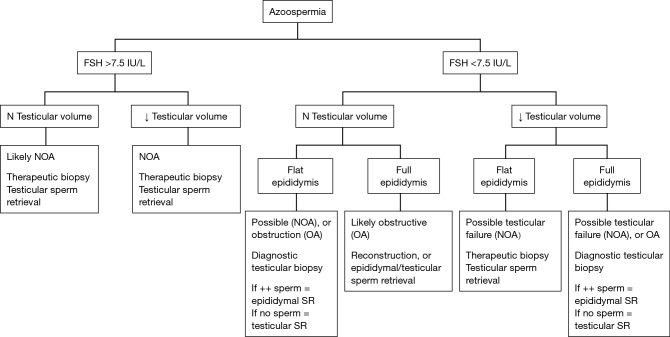
Obstructive azoospermia (CBAVD, post-vasectomy): MESA preferred (high yield, motile sperm, can cryopreserve). TESA/TESE as alternatives.
Non-obstructive azoospermia: Micro-TESE is the gold standard. Timing: coordinate with partner's IVF/ICSI cycle (fresh sperm preferred) or perform in advance with cryopreservation of any sperm found.
All retrieved sperm are used with ICSI (intracytoplasmic sperm injection), as surgically retrieved sperm cannot undergo conventional IVF.
15 Ovarian Stimulation Protocols
GnRH Agonist Long Protocol
The traditional "long protocol" was the gold standard for IVF for decades and remains widely used: GnRH agonist (leuprolide acetate 0.5-1.0 mg SC daily, or nafarelin nasal spray 200 μg BID) is started in the mid-luteal phase of the preceding cycle (~day 21, or following OCP priming). The agonist initially causes a "flare" (transient rise in FSH/LH for 2-3 days) followed by pituitary downregulation through GnRH receptor desensitization and internalization. After 10-14 days of downregulation (confirmed by E2 <50 pg/mL, thin endometrium, no follicles >10 mm on ultrasound, and absence of menses), gonadotropin stimulation begins with leuprolide dose reduced to 0.25 mg daily (maintained throughout stimulation to prevent LH recovery).
Advantages: (1) Nearly eliminates premature LH surge (<1% risk vs ~5-8% with antagonist); (2) Synchronizes follicular cohort for more uniform development; (3) Allows flexible scheduling (stimulation start can be adjusted ±1-2 days once downregulated). Disadvantages: (1) Longer overall treatment duration (~4-5 weeks including downregulation); (2) More total gonadotropin usage (due to suppressed endogenous FSH/LH); (3) Higher OHSS risk because GnRH agonist trigger CANNOT be used (pituitary already desensitized — hCG trigger is the only option); (4) Menopausal symptoms during downregulation phase (hot flashes, headache, mood changes); (5) Higher cost. The long agonist protocol remains preferred by some practitioners for: patients with endometriosis (prolonged downregulation may benefit implantation), patients with prior premature LH surge, and when scheduling flexibility is needed.
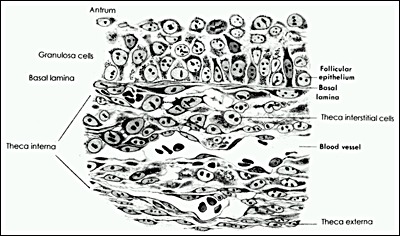
GnRH Antagonist Protocol
Now the most commonly used protocol worldwide. Gonadotropin stimulation begins on cycle day 2-3 (after baseline ultrasound confirms no ovarian cysts >15 mm and thin endometrium). GnRH antagonist (ganirelix 0.25 mg or cetrorelix 0.25 mg SC daily) is started when the lead follicle reaches ~13-14 mm (flexible start — most common) or on a fixed day (stimulation day 5-6). The antagonist is continued daily until and including trigger day.
Advantages: shorter overall treatment duration (~10-12 days total vs. ~4 weeks with long agonist), less total gonadotropin usage, allows GnRH agonist trigger (the single most effective OHSS prevention strategy — impossible in agonist protocols because the pituitary is already desensitized), no menopausal side effects during downregulation, and comparable pregnancy rates to long agonist protocol (Cochrane review: no significant difference in live birth rates between antagonist and agonist protocols). Disadvantages: requires more precise timing (premature LH surge can occur if antagonist start is delayed), slightly lower oocyte yield in some studies (though this may reflect less over-stimulation). This is the preferred protocol for high responders, PCOS patients, OHSS-risk patients, oncofertility (random-start compatible), and in most modern IVF practices as the default protocol.
Other Protocols
Mild/Mini-stimulation (mini-IVF): Low-dose gonadotropins (75-150 IU/day) or clomiphene/letrozole + low-dose gonadotropins. Aim: 2-7 oocytes. Used in poor responders (where high doses do not improve yield), older patients, patients with low AMH, or those seeking a gentler/lower-cost approach. Lower per-cycle cost but fewer embryos — may require accumulation over multiple cycles. Per-cycle pregnancy rates are lower but cumulative rates over multiple mini-IVF cycles may approach those of conventional IVF in certain populations.
Natural cycle IVF: No gonadotropin stimulation; retrieves the single dominant follicle with a GnRH antagonist added to prevent premature ovulation and hCG or GnRH agonist trigger. Very low yield (one oocyte per cycle, ~70% retrieval rate) but avoids OHSS entirely and has minimal medication burden. Per-cycle live birth rate: ~7-10%. Requires precise timing and frequent monitoring. May require multiple cycles. Best suited for patients who cannot tolerate stimulation or as an accumulation strategy.
Flare/microflare protocol: Microdose leuprolide (20-40 μg BID, prepared by specialty pharmacy as a diluted formulation) started on cycle day 2-3 (often preceded by OCP priming to synchronize follicles and prevent functional cyst formation), exploiting the initial "flare" effect (2-3 days of endogenous FSH/LH surge before desensitization) to boost ovarian response. Gonadotropins begin on cycle day 3-4. Used primarily in poor responders as an alternative to the antagonist protocol. Cannot use GnRH agonist trigger (pituitary desensitized).
Dual stimulation (DuoStim/Shanghai protocol): Two stimulation cycles within the same menstrual cycle — first stimulation in the follicular phase (standard start), oocyte retrieval, then a second stimulation begun in the luteal phase (5 days after first retrieval), followed by a second retrieval. Takes advantage of the discovery that follicular waves occur in both phases of the cycle. Maximizes oocyte yield in poor responders or for urgent fertility preservation (e.g., cancer patients who need maximum oocytes in minimum time). All embryos are vitrified (no fresh transfer possible).
Progestin-primed ovarian stimulation (PPOS): Medroxyprogesterone acetate (MPA 10 mg daily) or dydrogesterone is given alongside gonadotropins to prevent premature LH surge (instead of GnRH antagonist). Pioneered in China. Advantages: oral medication, lower cost than injectable antagonist, effective LH suppression. Disadvantage: fresh transfer not possible (progestin impairs endometrial receptivity) — freeze-all mandatory. Growing evidence for comparable outcomes to antagonist protocols.
Gonadotropin Medications
Recombinant FSH: Follitropin alfa (Gonal-F), follitropin beta (Follistim/Puregon), follitropin delta (Rekovelle — dosed by AMH and body weight). Human menopausal gonadotropin (hMG): Menopur (contains both FSH and LH activity from highly purified urinary source). Recombinant LH: Lutropin alfa (Luveris) — used in combination with FSH for patients with hypogonadotropic hypogonadism or advanced age where LH supplementation may benefit. Corifollitropin alfa (Elonva): Long-acting recombinant FSH (single injection replaces 7 days of daily FSH).
Dose Adjustments & Individualization
Gonadotropin starting dose is individualized based on age, BMI, AMH, AFC, and prior response. General guidelines: Poor responders (AMH <1.0): 300-450 IU/day, consider LH supplementation, growth hormone adjunct (experimental). Normal responders (AMH 1.0-3.5): 150-300 IU/day. High responders/PCOS (AMH >3.5): 100-150 IU/day. Dose adjustments during stimulation: based on follicular growth rate (target 1-2 mm/day) and E2 rise. If response is slow, increase dose by 75-150 IU; if over-responding (E2 rising too fast, >20 follicles developing), reduce dose or consider coasting (holding gonadotropins while continuing antagonist). The Rekovelle (follitropin delta) dosing algorithm automatically individualizes dose by AMH and body weight.
Monitoring & Trigger
Ovarian stimulation is monitored with serial transvaginal ultrasound (follicle measurement — number and mean diameter of each follicle) and serum estradiol (and often progesterone on trigger day). Monitoring visits occur every 1-3 days depending on response. Stimulation typically lasts 9-12 days. Trigger criteria: ≥2-3 follicles ≥17-18 mm (programs vary; some use ≥3 follicles ≥17 mm). Trigger options: hCG trigger (recombinant hCG — Ovidrel 250 μg; or urinary hCG 5,000-10,000 IU) — surrogates for the LH surge, triggers final oocyte maturation; oocyte retrieval is performed 34-36 hours after trigger. GnRH agonist trigger (leuprolide 1-4 mg) — induces endogenous LH/FSH surge from the pituitary; only works in antagonist protocols (pituitary not already desensitized); dramatically reduces OHSS risk but may compromise luteal phase (freeze-all strategy recommended unless aggressive luteal support given). Dual trigger (GnRH agonist + low-dose hCG 1,000-1,500 IU) — optimizes maturation while reducing OHSS risk; allows fresh transfer with appropriate luteal support.
16 Oocyte Retrieval & IVF Laboratory
Transvaginal Oocyte Retrieval
Performed 34-36 hours after trigger under IV sedation (propofol ± fentanyl) or less commonly under general anesthesia. The timing is critical: retrieval must occur before spontaneous ovulation (which occurs ~38-40 hours after trigger). A transvaginal ultrasound probe with an attached needle guide (typically 16-17 gauge single-lumen needle) is used to puncture each follicle sequentially. Continuous suction (100-200 mmHg) aspirates follicular fluid into test tubes maintained at 37°C. Each aspirate is immediately examined by the embryologist under the stereomicroscope for cumulus-oocyte complexes (COC). After aspiration, each follicle is flushed with warmed media if the oocyte was not found in the initial aspirate (flushing is controversial — the FLUSH trial showed no benefit in normal responders but may help in poor responders with few follicles).

Expected oocyte recovery rate: ~80% of follicles ≥14 mm yield an oocyte. Maturity assessment: MII (mature) — extruded first polar body visible; ready for ICSI. MI (immature) — no polar body, germinal vesicle broken down; may mature in vitro (in vitro maturation, IVM) over 24-48 hours. GV (germinal vesicle) — immature with intact GV; can be matured in vitro but lower quality. Typically 70-85% of retrieved oocytes are MII. Complications are rare: vaginal bleeding (~1% — usually controlled with pressure), pelvic infection (<0.5%), ovarian torsion (<0.1%), hemoperitoneum from ovarian capsule bleeding (rare, <0.1% requiring intervention), and very rarely bowel or ureteral injury. Prophylactic antibiotics (doxycycline 100 mg BID starting on retrieval day or azithromycin 1g prior) are routinely given.
Conventional IVF vs. ICSI
Conventional IVF (insemination): Oocytes are incubated with 50,000-100,000 motile sperm per oocyte in culture medium; fertilization occurs through natural sperm capacitation, acrosome reaction, zona pellucida binding, and oocyte penetration. Indicated when semen parameters are normal and no PGT is planned. Fertilization rate: ~60-70%. Advantages: less manipulation of the oocyte, lower cost. Disadvantage: risk of total fertilization failure (~5-10% of cycles).
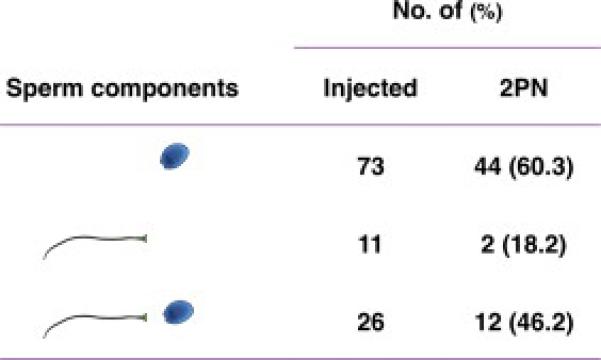
ICSI (Intracytoplasmic Sperm Injection): A single sperm is immobilized (tail crushed with injection pipette to breach the membrane and release activating factors), then injected directly into the oocyte cytoplasm through a glass micropipette using a micromanipulation system. The cumulus cells are first stripped from the oocyte using hyaluronidase to assess maturity and facilitate injection. ICSI fertilization rate: ~70-80%. Indications: severe male factor (OAT), surgically retrieved sperm (always requires ICSI), prior complete fertilization failure with conventional IVF, low oocyte yield (<4 oocytes), PGT planned (avoids contamination from cumulus cell DNA), in vitro matured oocytes, previously cryopreserved oocytes (zona hardened from vitrification). Normal fertilization is confirmed at 16-18 hours post-insemination by the presence of two pronuclei (2PN) and two polar bodies. Abnormal fertilization: 1PN (may be haploid or diploid — generally discarded), 3PN (triploidy from failure of polar body extrusion or dispermy — discarded), 0PN (may have failed activation — check for late-appearing pronuclei).
Split ICSI: In cases of unexplained infertility or borderline semen parameters, half the oocytes are inseminated conventionally and half by ICSI — this provides insurance against total fertilization failure while testing the sperm's fertilizing ability. Rescue ICSI: If conventional insemination results in no fertilization at 6-8 hours (assessed by polar body check or early pronuclear assessment), ICSI is performed on unfertilized oocytes. Success rates of rescue ICSI are lower than primary ICSI.
Embryo Culture
Embryos are cultured in specialized sequential media at 37°C with 5-6% CO2 and reduced O2 (5% — mimics the ~2-8% oxygen tension in the fallopian tube and uterus). Low oxygen culture improves blastocyst development rates and pregnancy outcomes compared to atmospheric O2 (20%). Culture systems: individual droplets under oil overlay (mineral oil or paraffin prevents evaporation and pH changes) in either benchtop incubators or conventional large-format incubators with separate chambers.
Cleavage stage development: Day 1 (zygote, 2PN); Day 2 (2-4 cells, grade 1-4 based on fragmentation and symmetry); Day 3 (6-8 cells, <10% fragmentation ideal). Compaction: Day 4 (morula, cells begin to compact and form tight junctions). Blastocyst stage: Day 5-6 (cavitation occurs, forming the blastocoel; ICM and TE differentiate; expanded blastocyst hatches from zona pellucida). Day 7 blastocysts: some laboratories now culture to day 7 for slow-developing embryos; pregnancy rates are lower than day 5-6 but not negligible (~25-35% if euploid on PGT-A).
Extended culture to blastocyst allows self-selection of higher-quality embryos (embryos with significant developmental arrest fail to reach blastocyst) but results in fewer embryos available for transfer/cryopreservation compared to day 3. Blastocyst transfer has higher implantation rates per embryo (~50-60% vs ~25-30% for cleavage stage) and allows single embryo transfer (SET), reducing multiple pregnancy rates. Indications for day 3 transfer (rather than blastocyst): very few embryos (≤2), concern about in vitro culture conditions, patient preference. The decision to culture to blastocyst vs. transfer at cleavage stage should be individualized.
Gardner Blastocyst Grading System
Expansion grade (1-6): 1 = early blastocyst (cavity <50% of embryo); 2 = blastocyst (cavity ≥50%); 3 = full blastocyst; 4 = expanded blastocyst (cavity larger than embryo, thinning zona); 5 = hatching blastocyst; 6 = hatched blastocyst.
Inner Cell Mass (ICM) grade: A = tightly packed, many cells; B = loosely grouped, several cells; C = very few cells.
Trophectoderm (TE) grade: A = many cells forming a cohesive layer; B = few cells forming a loose epithelium; C = very few large cells.
Example: "4AA" = expanded blastocyst with excellent ICM and trophectoderm. A grade of ≥3BB is generally considered good quality for transfer.
In Vitro Maturation (IVM)
In vitro maturation involves retrieval of immature oocytes (GV or MI stage) from small antral follicles with minimal or no gonadotropin stimulation, followed by in vitro maturation to MII over 24-48 hours in specialized IVM media. Indications: PCOS patients at very high OHSS risk (avoids stimulation entirely), oncofertility when there is insufficient time for full stimulation, or patients with prior recurrent OHSS. Maturation rates: ~50-70% of retrieved oocytes reach MII. Clinical outcomes: historically lower pregnancy rates than standard IVF, but modern IVM protocols (including priming with FSH for 2-3 days and hCG trigger) have improved results substantially. IVM-derived embryos have normal developmental potential when matured under optimized conditions. Capacitation IVM (CAPA-IVM): A newer protocol using a two-step culture system that improves oocyte competence by allowing nuclear and cytoplasmic maturation to synchronize.
Assisted Hatching
The zona pellucida is thinned or breached (laser — most common, precise; mechanical — partial zona dissection with microneedle; or chemical — acidified Tyrode's solution) before embryo transfer to facilitate blastocyst hatching and implantation. Rationale: age-related zona hardening or cryo-induced zona changes may impair natural hatching. May benefit: women ≥38, repeated implantation failure (≥2 failed cycles with good-quality embryos), morphologically thick zona pellucida (>13 μm), frozen-thawed embryos. Evidence of overall benefit for routine use is limited (Cochrane 2020 showed small increase in clinical pregnancy rate but no clear increase in live birth rate). Risk: monozygotic twinning rate may be slightly increased with assisted hatching.
Time-Lapse Imaging
Continuous imaging systems (EmbryoScope, GERI) allow morphokinetic assessment without removing embryos from the incubator. Parameters such as time to first cleavage (tPNf — pronuclear fading), duration of cell cycles (cc2 = time from 2-cell to 3-cell), synchrony of division, and direct cleavage detection are used with AI-assisted algorithms (e.g., iDAScore, KIDScore) for embryo selection. May improve selection beyond static morphology grading, but definitive evidence for improved live birth rates from RCTs is still emerging. Advantages include uninterrupted culture conditions and detailed developmental documentation.
Artificial Intelligence in Embryo Selection
AI-based algorithms trained on large datasets of embryo images and outcomes are increasingly used to predict blastocyst development, implantation potential, and ploidy status. Non-invasive PGT-A (niPGT-A) using spent culture media cell-free DNA analysis is an emerging alternative to trophectoderm biopsy, though accuracy is not yet sufficient to replace standard PGT-A. Other emerging technologies include hyperspectral imaging, metabolomic profiling of culture media, and OCT (optical coherence tomography) for non-invasive embryo assessment.
17 Preimplantation Genetic Testing
PGT-A (Aneuploidy)
PGT-A screens embryos for whole chromosome aneuploidy (monosomy, trisomy, segmental gains/losses). The evolution of PGT: first generation used FISH on single blastomeres (PGS 1.0 — limited chromosomes tested, now obsolete); second generation used aCGH or SNP arrays on blastomere or TE biopsy (PGS 2.0); current standard uses trophectoderm (TE) biopsy at the blastocyst stage (5-10 cells from day 5-6 blastocyst) analyzed by next-generation sequencing (NGS) — this is PGT-A 3.0. TE biopsy is preferred over blastomere biopsy because it samples cells destined for the placenta (not the embryo proper), removes more cells for higher accuracy, and causes less embryo damage.
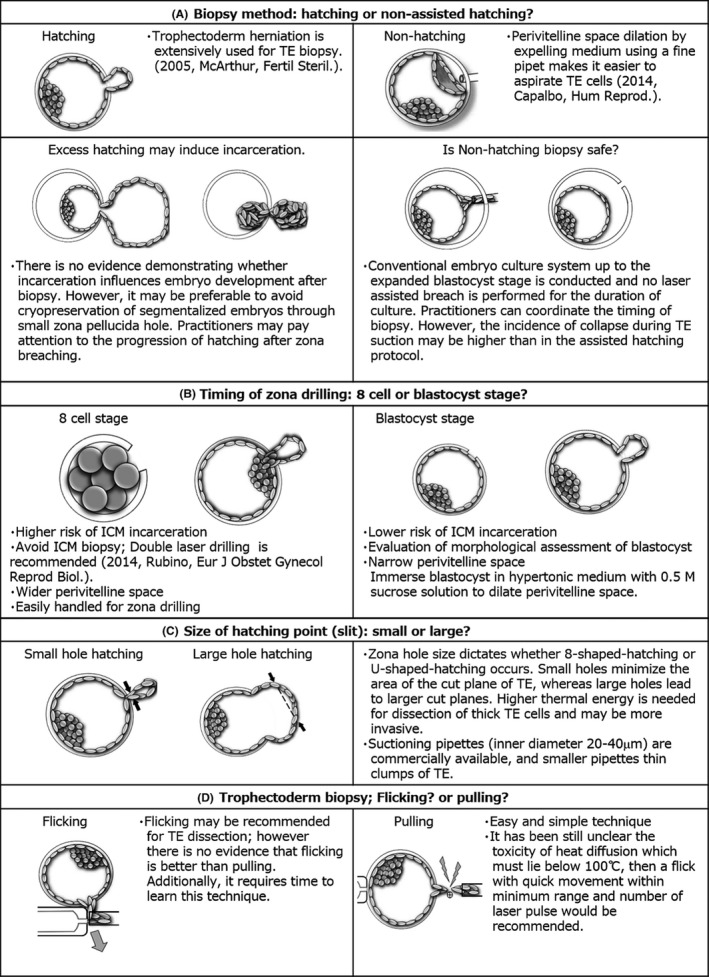
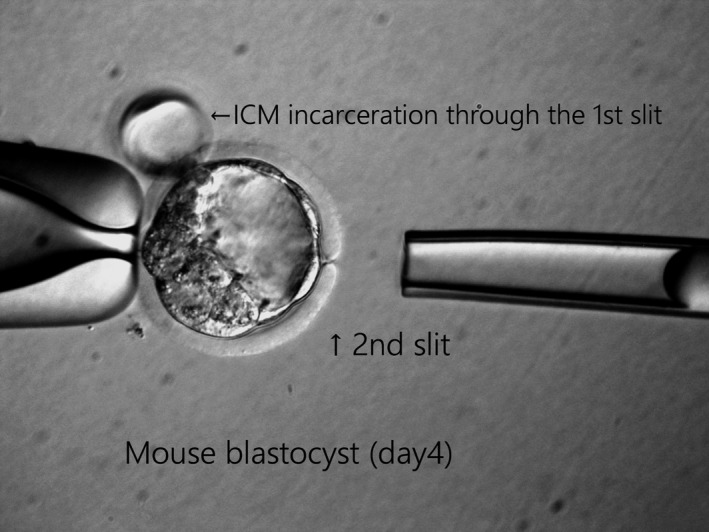
Results are reported as: euploid (46 chromosomes, all pairs balanced), aneuploid (whole chromosome gain or loss, or segmental imbalance ≥10-20 Mb — not recommended for transfer), or mosaic (mixture of euploid and aneuploid cells — see below). The primary clinical benefits of PGT-A: (1) Improved implantation rate per transfer (~50-60% for euploid vs ~30-35% for unscreened); (2) Reduced miscarriage rate (~8-10% for euploid vs ~15-25% for unscreened in women ≥35); (3) Reduced time to pregnancy (avoidance of transferring embryos destined to fail); (4) Supports single embryo transfer policy (confidence in the selected embryo).
The STAR trial (Munne et al., Fertil Steril 2019) showed PGT-A improved ongoing pregnancy rates per transfer in women 25-40. However, controversy persists regarding universal PGT-A: the ESHRE PETE trial (Cornelisse et al., Hum Reprod 2020) suggested no improvement in cumulative live birth rate per started IVF cycle when accounting for all embryos (including those not transferred due to aneuploidy). The debate centers on whether PGT-A improves efficiency (fewer transfers needed) without improving the total baby-per-cycle rate. PGT-A is most clearly beneficial for: women ≥35 (higher aneuploidy rate), RPL history, prior failed implantation, single embryo transfer programs, and reducing multiple pregnancy risk.
Mosaic Embryos
Mosaicism (20-80% aneuploid cells in the biopsy) is found in ~10-20% of biopsied blastocysts. Important: mosaicism in the TE biopsy may not reflect the ICM (future baby) — the TE biopsy samples the future placenta. Low-level mosaicism (<40% aneuploid cells) carries higher implantation potential than high-level mosaicism (>60%). Reported live birth rates from mosaic embryo transfers: ~30-50% (vs. ~50-60% for euploid), with generally normal neonatal outcomes but limited long-term data. Transfer of mosaic embryos may be considered when no euploid embryos are available, with appropriate genetic counseling.
PGDIS (Preimplantation Genetic Diagnosis International Society) guidelines rank mosaic embryos for transfer priority: (1) Low-level monosomic mosaics are preferred over trisomic mosaics. (2) Avoid chromosomes associated with viable trisomies (13, 18, 21) and chromosomes associated with uniparental disomy syndromes (6, 7, 11, 14, 15, 20). (3) Complex mosaicism (involving multiple chromosomes) is lowest priority. Amniocentesis or CVS is recommended for pregnancies resulting from mosaic embryo transfers, though the vast majority show normal fetal karyotypes.
PGT-M (Monogenic/Single Gene Disorders)
PGT-M tests for specific inherited single-gene disorders (cystic fibrosis, sickle cell disease, Huntington disease, BRCA mutations, spinal muscular atrophy, fragile X, etc.). Requires a custom probe/linkage analysis designed from the couple's DNA and often from affected family members. Turnaround for probe development: 4-8 weeks. Biopsy technique and processing similar to PGT-A. Can be combined with PGT-A on the same biopsy.
PGT-SR (Structural Rearrangement)
PGT-SR is used for carriers of balanced translocations (robertsonian or reciprocal) or inversions. These carriers produce high rates of unbalanced gametes, leading to recurrent miscarriage or affected offspring. Robertsonian translocation carriers (e.g., rob(13;14)) produce ~15% unbalanced gametes; reciprocal translocation carriers may produce 50-70% unbalanced gametes depending on the specific breakpoints. PGT-SR identifies embryos with balanced/normal chromosomal complement. NGS-based platforms can detect segmental imbalances (≥5-10 Mb). Important limitation: PGT-SR cannot distinguish between a balanced carrier and a completely normal embryo in most cases (requires specialized methods like haplarithm analysis or karyomapping). Genetic counseling is essential to discuss the implications of transferring potentially carrier embryos and the small chance of uniparental disomy in robertsonian translocation cases.
Emerging PGT Technologies
Non-invasive PGT-A (niPGT-A): Analysis of cell-free DNA in spent blastocyst culture media (blastocentesis or spent media analysis). Avoids TE biopsy and its potential risks. Concordance with standard PGT-A: ~70-80% — insufficient for standalone clinical use currently. Polygenic risk scoring (PRS): Controversial application of PGT to screen embryos for polygenic traits (height, IQ, disease risk). ASRM and ESHRE have expressed significant ethical concerns. Mitochondrial DNA (mtDNA) copy number: Initially proposed as a viability biomarker (MitoScore), but clinical utility has not been confirmed. Whole genome sequencing of biopsied cells provides comprehensive chromosomal and potentially sequence-level analysis but at higher cost and complexity.
18 Embryo Transfer & Luteal Support
Fresh vs. Frozen Embryo Transfer
The trend in ART has shifted toward freeze-all strategies (vitrifying all embryos, performing FET in a subsequent cycle). Advantages: avoids transfer in a supraphysiologic hormonal environment, mandatory when using GnRH agonist trigger, allows time for PGT results, and eliminates late-onset OHSS. The E-Freeze trial (Vuong et al., NEJM 2018) showed frozen embryo transfer had higher ongoing pregnancy rates than fresh transfer in PCOS patients. However, for normal responders the evidence is mixed — some studies show equivalent outcomes for fresh vs frozen (Shi et al., NEJM 2018). FET is associated with slightly higher risk of pre-eclampsia and large-for-gestational-age infants in programmed (HRT) cycles compared to fresh transfers.
Endometrial Preparation for FET
Three main approaches exist for preparing the endometrium before frozen embryo transfer:
Natural cycle FET: Relies on endogenous follicular development and ovulation; transfer is timed to the endogenous progesterone rise detected by serial LH monitoring (urine OPK or serum LH) and ultrasound confirmation of dominant follicle ≥17 mm and subsequent ovulation. Blastocyst transfer is scheduled ~7 days after LH surge (or 5 days after confirmed ovulation). Advantages: no exogenous hormones, physiologic endocrine environment with intact corpus luteum (provides endogenous progesterone), lower pre-eclampsia risk (emerging evidence that natural cycle FET has lower pre-eclampsia rates than programmed cycles — the corpus luteum may produce vasoactive factors including relaxin that are protective). Disadvantages: requires regular ovulatory cycles, less scheduling flexibility, cycle cancellation if ovulation is missed or does not occur.
Programmed (HRT/artificial) cycle: The most commonly used protocol. Oral estradiol valerate (escalating doses: 2 mg → 4 mg → 6 mg daily, or fixed 6 mg from the start) or transdermal estradiol patches (100-200 μg) to grow the endometrium. GnRH agonist downregulation or antagonist may be added to prevent spontaneous ovulation. Once endometrial thickness ≥7-8 mm on ultrasound (typically after 10-14 days of estrogen), progesterone is added. The day of progesterone initiation determines the transfer date: day 6 of progesterone for blastocyst transfer (5 full days of progesterone exposure before transfer). Advantages: precise scheduling, no need for ovulatory cycles (useful for anovulatory patients, donor oocyte recipients, and scheduling convenience). Disadvantages: exogenous hormones for 10-12+ weeks, no corpus luteum (all hormonal support is exogenous — abrupt discontinuation causes miscarriage), possibly higher pre-eclampsia risk, and slightly higher large-for-gestational-age birth weight.
Modified natural cycle: Natural follicular development is monitored, and an hCG trigger (5,000-10,000 IU or Ovidrel 250 μg) is given when the dominant follicle reaches ≥17-18 mm to precisely time ovulation. Transfer is scheduled 7 days after trigger for blastocyst. Progesterone supplementation (vaginal) may be added for additional luteal support. Combines the advantages of a natural endocrine environment (functioning corpus luteum) with more precise timing than a purely natural cycle.
Progesterone for Luteal Support
Progesterone supplementation is mandatory after IVF because ovarian stimulation and oocyte retrieval disrupt normal corpus luteum function: (1) Aspiration of follicles removes granulosa cells that produce progesterone; (2) GnRH agonist trigger (in antagonist cycles) produces a shorter LH surge that poorly supports the corpus luteum; (3) Multiple corpora lutea in stimulated cycles may paradoxically produce less progesterone than a single CL in a natural cycle due to premature luteolysis. Without luteal support, implantation and early pregnancy rates are significantly lower.
Formulations: Vaginal micronized progesterone (Endometrin 100 mg BID or TID; Crinone 8% gel daily or BID; Utrogestan 200 mg BID or TID) — most commonly used worldwide due to ease of administration and the "first uterine pass effect" (vaginal progesterone achieves high endometrial concentrations despite relatively low serum levels — serum progesterone measurement does NOT reliably reflect endometrial levels with vaginal route). Intramuscular progesterone in oil (50-100 mg daily) — reliable absorption, higher and more consistent serum levels, but painful daily injections (large-gauge needle, oil vehicle), risk of sterile abscess, local reactions, and injection site nodules. Subcutaneous progesterone (Prolutex 25 mg daily) — aqueous formulation, alternative to IM with less injection pain. Oral dydrogesterone (Duphaston 10 mg TID) — the LOTUS I and II trials (Tournaye et al., NEJM 2017; Griesinger et al., Fertil Steril 2018) demonstrated non-inferiority to vaginal micronized progesterone for luteal support; provides a well-tolerated oral option. Combined regimens: Some clinicians use vaginal + IM or vaginal + oral for patients with suboptimal early pregnancy hCG rise or prior luteal phase deficiency.
Duration: progesterone is typically continued until 8-10 weeks of gestation (through the luteal-placental shift, when the placenta assumes progesterone production at ~7-9 weeks). Some practitioners taper rather than abruptly discontinue. For programmed FET cycles (no corpus luteum), exogenous estrogen and progesterone must be continued for the full 10-12 weeks or until placental function is confirmed (abrupt withdrawal can cause miscarriage).
Single Embryo Transfer (SET) Policy
Elective single embryo transfer (eSET) is the most important measure for reducing multiple pregnancy rates in IVF. Key evidence: cumulative live birth rate (from sequential fresh + frozen transfers from a single stimulation cycle) with eSET is equivalent to double embryo transfer (DET) while dramatically reducing twin rates (from ~25-35% with DET to ~1-2% with eSET).
Age <35: Transfer 1 blastocyst (or 1-2 cleavage-stage). Strongly recommend eSET.
Age 35-37: Transfer 1-2 blastocysts (or 2 cleavage-stage). eSET encouraged if good prognosis.
Age 38-40: Transfer 2-3 cleavage-stage or 2 blastocysts.
Age 41-42: Transfer up to 3 cleavage-stage or 2-3 blastocysts.
Age >42: Transfer up to 3-4 cleavage-stage or 3 blastocysts.
Euploid embryos (PGT-A tested): Transfer 1 euploid embryo at a time regardless of age.
Donor oocyte cycles: Follow the donor's age group for transfer guidelines.
Many countries (Belgium, Sweden, Finland, Australia) have mandated single embryo transfer through legislation or insurance requirements, achieving national twin rates of <5% from IVF. Patients should be counseled that twins are not a desirable IVF outcome but rather a complication that significantly increases risks to mother and babies.

Transfer Technique
Ultrasound-guided embryo transfer is standard (abdominal US with full bladder for acoustic window). A soft catheter (Cook, Wallace, Guardia) is loaded with the embryo(s) in a small volume of culture medium (~10-20 μL) flanked by air bubbles for ultrasound visualization, and advanced through the cervix. The catheter tip is positioned 1.5-2 cm from the fundus in the mid-cavity. Key factors affecting success: (1) atraumatic passage (avoid cervical trauma, use outer sheath if cervical stenosis; mock transfer beforehand is recommended), (2) absence of blood or mucus on catheter (correlates with lower pregnancy rates if present), (3) ultrasound confirmation of embryo deposition (air bubble visible in the cavity), (4) slow, gentle catheter withdrawal, (5) immediate microscopic catheter check to confirm no retained embryos. If the embryo is retained in the catheter, it is reloaded and transfer is repeated immediately.
Adjunctive measures: bed rest after transfer has no proven benefit (Purcell et al., Fertil Steril 2007) and is no longer recommended. Patients can resume normal (non-strenuous) activities. Acupuncture at the time of transfer has been studied (mixed results; not routinely recommended but not harmful). Atropine (0.5 mg IM) may reduce uterine contractility and is sometimes given empirically. The embryo transfer is widely considered the single most important and technique-dependent step in IVF — provider experience significantly affects success rates.
19 Cryopreservation & IVF Outcomes
Vitrification
Vitrification is an ultrarapid freezing technique (>15,000°C/min cooling rate) that prevents intracellular ice crystal formation by transitioning the cell directly from liquid to a glass-like (vitreous) solid state. The process requires high concentrations of cryoprotectants (DMSO + ethylene glycol, typically 15% each) and very small volumes (microdrops on open carrier devices: Cryotop, Cryolock, Rapid-i; or closed systems: CryoTip, RapidVit). It has largely replaced slow-freezing protocols (which cooled at ~0.3°C/min and were associated with ice crystal damage) due to dramatically superior survival rates.
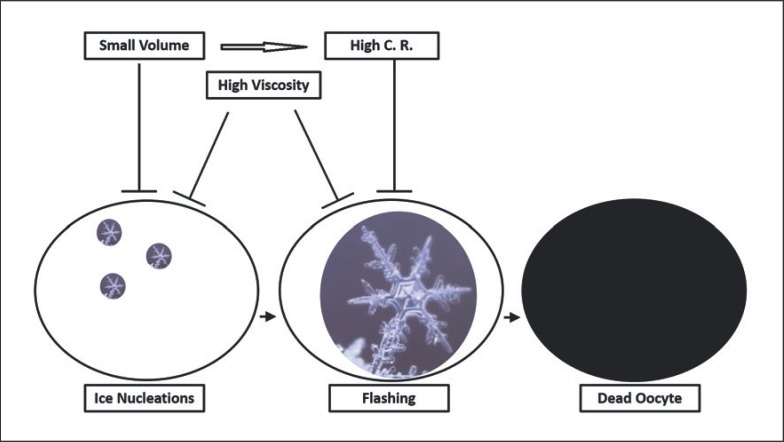
Survival rates by cell type: Oocyte vitrification: 85-95% survival for mature MII oocytes (post-thaw oocytes have comparable fertilization rates to fresh oocytes). Oocyte vitrification was declared "no longer experimental" by ASRM in 2012. Embryo vitrification (blastocyst): >95-99% survival; frozen-thaw blastocysts have pregnancy rates equivalent to or exceeding fresh transfer in many programs. Cleavage-stage embryo vitrification: ~90-95% survival (lower than blastocyst due to the compact morula/early blastocyst stage being more challenging). Sperm cryopreservation: conventional slow freeze with glycerol-based cryoprotectant remains the standard; post-thaw motility recovery: ~40-60% (depending on initial quality). Vitrification of sperm in microvolumes is an emerging technique (particularly useful for very small samples from TESE) but not yet standard practice.
Long-Term Storage
Vitrified oocytes and embryos stored in liquid nitrogen (-196°C) show no deterioration over time — pregnancies have been reported from embryos stored for >25 years. At -196°C, all biological processes are effectively halted. Storage considerations: secure cryobank facilities with 24/7 monitoring and backup systems, unique patient identifiers with dual witness verification at every step, electronic witnessing systems (barcode or RFID) to prevent sample mix-ups, informed consent for disposition (discard, donate to research, donate to another patient, continued storage), and clear policies for storage in cases of death, divorce, or abandonment. Regulatory frameworks: many countries have storage time limits (e.g., 10 years in the UK, extendable); in the US, no federal storage limits exist but individual clinic policies vary.
Sperm Cryopreservation
Conventional slow-freeze technique: sperm are mixed with cryoprotectant (glycerol-based medium), cooled at a controlled rate, and stored in liquid nitrogen. Post-thaw motility recovery: typically ~40-60% of the pre-freeze value. Severely oligozoospermic samples may have very few surviving sperm post-thaw, necessitating ICSI. Sperm can also be cryopreserved in very small volumes using specialized devices (Cryolock, SpermVD) for surgically retrieved specimens. Donor sperm banks use quarantine protocols: samples are stored for 6 months, and the donor is retested for infectious diseases before releasing samples for use.
IVF Outcomes — Age-Specific Success Rates
SART (Society for Assisted Reproductive Technology) national reporting data (US, 2021) for live birth rate per intended egg retrieval:
Age <35: Live birth rate ~46% per retrieval (cumulative with fresh + frozen transfers: ~55-65%).
Age 35-37: Live birth rate ~36% per retrieval.
Age 38-40: Live birth rate ~23% per retrieval.
Age 41-42: Live birth rate ~12% per retrieval.
Age >42: Live birth rate ~4% per retrieval with own oocytes.
Donor oocyte cycles: Live birth rate ~50-55% per transfer regardless of recipient age.
Prognostic Factors
Key factors predicting IVF success:
1. Female age — The dominant factor, reflecting oocyte quality and aneuploidy rate. Aneuploidy rates by age: <35 = ~30-40%, 38-40 = ~50-60%, 41-42 = ~60-75%, >42 = ~80-90%. Age cannot be overcome by increasing oocyte numbers.
2. Embryo quality — Gardner grade, morphokinetics, PGT-A status (euploid embryo transfer: ~50-60% implantation regardless of age up to 42-43).
3. Ovarian reserve (AMH, AFC) — Predicts quantitative response (number of oocytes) but does not independently predict quality after controlling for age.
4. Number of oocytes retrieved — Optimal range: 10-15 (Sunkara et al., Hum Reprod 2011). Below this, fewer embryos for selection. Above ~20, diminishing returns with increased OHSS risk.
5. Diagnosis — Tubal factor and male factor generally have better IVF outcomes than DOR or severe endometriosis.
6. BMI — Obesity (BMI >30) reduces success by ~10-20%, impairs response to gonadotropins, increases miscarriage risk, and complicates oocyte retrieval and embryo transfer. Many programs set BMI limits for treatment.
7. Prior IVF success — Previous live birth from IVF is a positive prognostic indicator.
8. Lifestyle factors — Smoking reduces IVF success by ~50% (accelerated follicle loss, zona pellucida hardening, impaired implantation); alcohol (>4 drinks/week) may reduce success; caffeine (>200 mg/day) is a possible risk factor for miscarriage.
When to Stop Treatment
A difficult but important counseling discussion. Factors to consider: cumulative prognosis diminishes with each failed cycle; after 3-4 complete IVF cycles without pregnancy in women ≥40 or with DOR, live birth rates per additional cycle are very low (<5% per cycle). Data suggests that ~95% of IVF successes occur within the first 6 cycles. Transition to donor oocytes, surrogacy, adoption, or child-free living should be discussed sensitively. Psychological support (counseling, support groups, mind-body programs) should be offered throughout treatment and at discontinuation. Factors favoring continued treatment: previously achieved pregnancy/embryos, euploid embryos remaining, identifiable and correctable factor in prior failure (e.g., thin endometrium, transfer difficulty). Factors favoring discontinuation: no embryos despite maximal stimulation, all embryos aneuploid across multiple cycles, no implantation of euploid embryos, significant emotional/financial/physical burden.
IVF Laboratory Quality Control
IVF laboratory performance is critical to outcomes and requires rigorous quality control: (1) Air quality — HEPA-filtered positive-pressure environment with VOC (volatile organic compound) filtration; carbon-activated air filters. (2) Temperature control — Heated stages on all microscopes, 37°C incubators with continuous monitoring and alarms. (3) pH and gas — CO2 levels maintained at 5-6% (bicarbonate-buffered media) or 0% with HEPES/MOPS-buffered media for bench work. (4) Witnessing — Electronic witnessing systems (RFID) at every step to prevent patient mix-ups; double witness check for critical steps (insemination, transfer loading). (5) KPIs — Laboratory key performance indicators: fertilization rate (normal ≥65% for conventional IVF, ≥70% for ICSI), blastocyst development rate (≥40% of 2PN), cryosurvival rate (≥90% for vitrified blastocysts), implantation rate. (6) Accreditation — CAP/CLIA certification required in the US; regular proficiency testing and inspections.
20 ART Complications
Ovarian Hyperstimulation Syndrome (OHSS)
OHSS is the most serious iatrogenic complication of ovarian stimulation. Pathophysiology: hCG triggers massive vascular endothelial growth factor (VEGF) release from hyperstimulated ovarian granulosa-lutein cells, causing increased capillary permeability, third-space fluid shifts, hemoconcentration, and potential end-organ damage.
Mild OHSS: Abdominal distension, mild pain, ovaries <8 cm. Management: outpatient, oral hydration, analgesics, monitoring.
Moderate OHSS: Nausea/vomiting, ultrasonographic ascites, ovaries 8-12 cm. Management: close outpatient monitoring, daily weights, I/O tracking, serial labs (Hct, renal function, electrolytes).
Severe OHSS: Clinical ascites, hydrothorax, Hct >45%, WBC >15,000, oliguria, creatinine 1.0-1.5 mg/dL, liver dysfunction, ovaries >12 cm. Management: hospitalization, IV fluids (NS/LR to maintain urine output >20-30 mL/hr), paracentesis for tense ascites/respiratory compromise, VTE prophylaxis (LMWH — OHSS is a prothrombotic state), albumin infusion.
Critical OHSS: Renal failure, ARDS, tense ascites, thromboembolism, pericardial effusion. Management: ICU, aggressive fluid management, thromboprophylaxis, consider cycle cancellation and pregnancy termination in extreme cases.
Prevention strategies: (1) Identify risk factors early (PCOS, high AMH, young age, low BMI, prior OHSS). (2) Antagonist protocol. (3) GnRH agonist trigger (instead of hCG) — most effective single prevention measure; combined with freeze-all. (4) Cabergoline 0.5 mg daily for 8 days from trigger day (reduces VEGF receptor expression). (5) Freeze-all (prevents late OHSS from pregnancy-derived hCG). (6) Lower gonadotropin starting doses. (7) Coasting (withholding gonadotropins while continuing antagonist until E2 falls).
Multiple Pregnancy
Multiple pregnancy remains the most common and most significant complication of ART, with substantial maternal and neonatal consequences. Specific risks of twin pregnancy: preterm birth ~60% (mean gestational age at delivery: ~35-36 weeks vs ~39 weeks for singletons), preeclampsia 2-3x increased, gestational diabetes 1.5x, placenta previa 2x, postpartum hemorrhage, cesarean delivery (~75%), and neonatal morbidity (NICU admission, respiratory distress, intraventricular hemorrhage, cerebral palsy). The shift to elective single embryo transfer (eSET) has dramatically reduced ART-related twin rates from ~30% to <5-10% in centers with strong SET policies (including Nordic countries where twin rates from IVF are now <5%). The economic cost of a twin ART pregnancy is approximately 3-5x that of a singleton ART pregnancy.
Higher-order multiples (≥3) are now rare from IVF (due to transfer guidelines) but remain a complication of gonadotropin/IUI cycles with inadequate follicular monitoring. Cycle cancellation should be enforced when >3 dominant follicles (≥14 mm) develop during IUI stimulation. Multifetal pregnancy reduction (transabdominal or transvaginal intracardiac KCl injection, typically performed at 11-14 weeks) to twins or singleton may be offered for triplets or higher-order multiples, with discussion of the ~5-7% procedure-related pregnancy loss risk per fetus reduced. Monozygotic twinning (identical twins from a single embryo) is slightly increased with IVF (~2% vs ~0.4% naturally), possibly related to zona pellucida manipulation, extended culture, and assisted hatching.
Ectopic & Heterotopic Pregnancy
Ectopic pregnancy after IVF occurs in ~2-5% of clinical pregnancies (higher than the ~1-2% natural rate), especially with tubal factor infertility. Heterotopic pregnancy (simultaneous intrauterine and ectopic pregnancy) occurs in ~1% of IVF pregnancies (vs. ~1/30,000 naturally) — requires high clinical suspicion. An intrauterine pregnancy on ultrasound does NOT exclude ectopic in IVF patients. Treatment of ectopic component: laparoscopic salpingectomy (methotrexate is contraindicated when a viable intrauterine pregnancy coexists). Cesarean scar pregnancy and interstitial (cornual) ectopic may also occur after embryo transfer and require specialized management (methotrexate injection, UAE, or surgical excision).
Other ART-Related Risks
Ovarian torsion: Enlarged, stimulated ovaries are at increased risk of torsion. Presents with acute unilateral pelvic pain ± nausea/vomiting. Diagnosis: Doppler ultrasound (absent or reduced ovarian blood flow, though normal Doppler does not exclude torsion). Treatment: laparoscopic detorsion (preserve ovary if viable — even if it appears dark/congested, most recover with detorsion). Bleeding from oocyte retrieval: Vaginal wall bleeding (usually self-limited; apply pressure or vaginal packing), intraperitoneal bleeding from ovarian capsule (rarely requires laparoscopy). Pelvic abscess: Rare (<0.5%), risk increased if endometrioma is traversed during retrieval. Present with fever, pelvic pain days after retrieval. Treatment: IV antibiotics ± drainage. Long-term safety: Large population studies have not demonstrated an increased risk of breast or ovarian cancer with fertility medications, though data for >6 cycles of clomiphene and borderline ovarian tumors remain under surveillance.
21 Polycystic Ovary Syndrome (PCOS)
Rotterdam Criteria (2003)
PCOS is diagnosed when two of three criteria are met (after exclusion of other etiologies): (1) Oligo-anovulation (cycles >35 days, <8 cycles/year, or amenorrhea); (2) Clinical and/or biochemical hyperandrogenism (hirsutism by modified Ferriman-Gallwey score ≥4-6, acne, alopecia, and/or elevated total/free testosterone, androstenedione, or DHEA-S); (3) Polycystic ovarian morphology on ultrasound (≥20 follicles per ovary of 2-9 mm and/or ovarian volume ≥10 mL — 2018 updated threshold with modern transducers). Not required in adolescents (unreliable).

PCOS Phenotypes
Phenotype A (classic/complete): Hyperandrogenism + oligo-anovulation + PCO morphology. All three criteria present. Highest metabolic risk (insulin resistance, dyslipidemia, T2DM). Most severe reproductive phenotype. ~50% of PCOS patients.
Phenotype B (classic without PCO): Hyperandrogenism + oligo-anovulation (no PCO morphology on ultrasound). Similar metabolic risk to phenotype A. ~15% of PCOS patients.
Phenotype C (ovulatory PCOS): Hyperandrogenism + PCO morphology (regular ovulatory cycles). Intermediate metabolic risk. May present with hirsutism/acne as primary complaint. ~20% of PCOS patients.
Phenotype D (non-hyperandrogenic): Oligo-anovulation + PCO morphology (no clinical or biochemical hyperandrogenism). Lowest metabolic risk. Mildest phenotype. Some experts debate whether this truly represents PCOS. ~15% of PCOS patients.
The 2018 International Evidence-Based Guideline for PCOS endorses the Rotterdam criteria and emphasizes that not all PCOS is the same — management should be tailored to the individual phenotype and presenting concerns (fertility, metabolic risk, cosmetic/dermatologic, psychological).
Metabolic Implications
PCOS is a metabolic syndrome with lifelong health implications beyond reproduction:
Insulin resistance: Present in ~50-80% of PCOS patients regardless of BMI (though obesity worsens it). Mechanism: post-receptor signaling defect in the insulin pathway, affecting both metabolic and reproductive tissues. Compensatory hyperinsulinemia stimulates ovarian androgen production (via theca cell LH receptor augmentation) and reduces hepatic SHBG (increasing free testosterone). Screen with 2-hour oral glucose tolerance test (OGTT) at diagnosis and every 1-3 years (fasting glucose alone misses ~35% of impaired glucose tolerance in PCOS).
Type 2 diabetes: 5-10x increased risk; up to 30-40% of PCOS women will develop impaired glucose tolerance or T2DM by age 40. Dyslipidemia: Elevated TG, low HDL, elevated LDL — present in up to 70% of PCOS patients. Cardiovascular risk: Increased subclinical atherosclerosis markers (carotid intima-media thickness, coronary artery calcium), though definitive cardiovascular outcome data are limited. Non-alcoholic fatty liver disease (NAFLD): 2-3x increased prevalence. Endometrial cancer: 2-6x increased risk due to chronic unopposed estrogen from anovulation — prescribe cyclic progestin (medroxyprogesterone 10 mg for 10-14 days every 1-3 months) or combined OCPs in non-fertility-seeking patients to protect the endometrium. Obstructive sleep apnea: 5-30x increased prevalence in PCOS; screen with Epworth Sleepiness Scale. Depression/anxiety: 2-3x increased prevalence; screen routinely.
Treatment for Ovulation Induction in PCOS
(1) Lifestyle modification — 5-10% weight loss restores ovulation in ~55-80% of overweight patients. First-line for overweight/obese PCOS. (2) Letrozole 2.5-7.5 mg days 3-7 — first-line pharmacologic treatment per international evidence-based guideline (2018) and PPCOS II trial; higher live birth rate, lower multiple rate, and no anti-estrogenic endometrial effects compared to clomiphene. (3) Clomiphene citrate 50-150 mg days 3-7 or 5-9 — second-line; ~80% will ovulate but only ~40-45% achieve pregnancy (discordance due to anti-estrogenic effects on cervical mucus and endometrium). (4) Metformin 1,500-2,550 mg daily — not first-line for ovulation induction alone but useful adjunct; may enhance letrozole/clomiphene response; primary role in metabolic management. (5) Gonadotropins (low-dose step-up protocol: start FSH 37.5-75 IU/day, increase by 37.5 IU every 7-14 days until single dominant follicle) — third-line; higher multiple and OHSS risk. (6) Laparoscopic ovarian drilling (LOD) — electrocautery or laser to ovarian cortex (4-10 punctures per ovary); induces ovulation in ~50%; alternative to gonadotropins for clomiphene-resistant PCOS. Risk of adhesions and reduced reserve. (7) IVF — last resort; PCOS patients are high responders (OHSS risk — antagonist protocol, GnRH agonist trigger, freeze-all).
22 Amenorrhea & Hyperprolactinemia
Primary Amenorrhea
Defined as absence of menarche by age 15 with normal secondary sexual characteristics (breast development present, indicating estrogen production), or by age 13 without any secondary sexual characteristics. Prevalence: ~0.1%.
Workup algorithm: (1) Physical examination (secondary sexual characteristics, external genitalia, vaginal patency, height/growth chart); (2) Pelvic ultrasound (confirm presence or absence of uterus and ovaries); (3) If uterus absent: karyotype — 46,XX = Müllerian agenesis (MRKH); 46,XY = androgen insensitivity syndrome (CAIS). (4) If uterus present: FSH level — elevated FSH = gonadal dysgenesis (karyotype to diagnose Turner syndrome 45,X vs 46,XX gonadal dysgenesis vs 46,XY gonadal dysgenesis/Swyer); low/normal FSH = hypothalamic/pituitary cause (MRI pituitary, assess for Kallmann with smell testing). (5) If normal anatomy and hormones: consider outflow tract obstruction (imperforate hymen — bulging introitus with hematocolpos; transverse vaginal septum).
Most common causes by frequency: gonadal dysgenesis (Turner syndrome — 45,X — short stature, web neck, shield chest, streak gonads, ~30% of primary amenorrhea), Müllerian agenesis (Mayer-Rokitansky-Küster-Hauser syndrome — absent uterus and upper two-thirds of vagina, normal ovaries and secondary sexual characteristics, 46,XX — ~15%), constitutional delay of puberty (diagnosis of exclusion — ~10%), hypothalamic/pituitary dysfunction (Kallmann, craniopharyngioma, hyperprolactinemia), complete androgen insensitivity (46,XY, female phenotype, absent uterus), and outflow obstruction (imperforate hymen, transverse vaginal septum).
Secondary Amenorrhea
Absence of menses for ≥3 months in women with previously regular cycles or ≥6 months with previously irregular cycles. More common than primary amenorrhea, affecting up to 3-5% of reproductive-age women. After pregnancy is excluded (always the first step — serum β-hCG), workup includes: TSH, prolactin, FSH, estradiol, and a progesterone withdrawal test (medroxyprogesterone acetate 10 mg daily for 10 days or micronized progesterone 200 mg daily for 10 days). Interpretation: withdrawal bleeding within 2-7 days after completing progestin indicates adequate estrogen production and a patent outflow tract (the endometrium was primed by endogenous estrogen); this pattern is most consistent with anovulation (PCOS is the most common cause). No withdrawal bleeding suggests either insufficient estrogen to prime the endometrium (hypoestrogenic state) or outflow tract obstruction (Asherman syndrome).
Step 1: Exclude pregnancy (β-hCG).
Step 2: TSH (hypothyroidism), prolactin (hyperprolactinemia).
Step 3: Progesterone withdrawal test. Bleeding = anovulation with adequate estrogen (most commonly PCOS). No bleeding = proceed to step 4.
Step 4: Estrogen + progesterone challenge (conjugated estrogen 1.25 mg × 21 days + medroxyprogesterone last 10 days). Bleeding = hypoestrogenic state (proceed to step 5). No bleeding = outflow tract obstruction (Asherman syndrome).
Step 5: FSH level. High FSH = ovarian failure (POI). Low/normal FSH = hypothalamic/pituitary dysfunction (MRI pituitary, consider hypothalamic amenorrhea, Sheehan syndrome).
Hyperprolactinemia
Prolactin elevation suppresses GnRH pulsatility, causing anovulation and amenorrhea. Microprolactinoma (<10 mm): Most common pituitary tumor in women of reproductive age; prolactin typically 25-200 ng/mL. Macroprolactinoma (≥10 mm): Prolactin usually >200 ng/mL; may cause visual field defects (bitemporal hemianopia from optic chiasm compression). Important: the "stalk effect" from non-functioning pituitary macroadenomas compressing the stalk causes mild prolactin elevation (usually <100 ng/mL) — this should not be confused with prolactinoma.
Treatment: Cabergoline (starting 0.25 mg twice weekly, titrated up to 1-2 mg twice weekly based on prolactin levels) is first-line — superior efficacy (normalizes prolactin in >90%) and tolerability (fewer GI side effects) compared to bromocriptine. Cabergoline also shrinks tumors in ~80% of cases (tumor response is assessed with MRI at 6-12 months). Bromocriptine (1.25-5 mg BID, titrated slowly to minimize nausea/orthostatic hypotension) is preferred by some practitioners in pregnancy due to its longer safety record, though increasing data support cabergoline safety in early pregnancy. Once prolactin normalizes and menses resume, ovulation typically returns and fertility treatment may proceed. In microprolactinomas, medication may be discontinued after 2 years if prolactin normalizes and tumor is not visible on MRI (recurrence rate ~25-50% after withdrawal). Important: macroprolactinomas carry a risk of tumor expansion during pregnancy (due to loss of dopamine agonist suppression and estrogen-mediated stimulation) — visual field monitoring is recommended during pregnancy in women with macroprolactinomas.
23 Müllerian Anomalies
Embryology
The Müllerian (paramesonephric) ducts develop at ~6 weeks of gestation alongside the mesonephric (Wolffian) ducts. In the female fetus (absence of AMH from Sertoli cells and absence of testosterone from Leydig cells), the Wolffian ducts regress and the Müllerian ducts develop. Müllerian duct development involves three stages: (1) Elongation — ducts grow caudally; (2) Fusion — the caudal portions fuse in the midline to form the uterovaginal primordium (uterus and upper vagina); (3) Resorption — the midline septum between the fused ducts is resorbed. Failure at each stage produces different anomalies.
ASRM Classification (2021 Update)
Class I — Agenesis/Hypoplasia: Müllerian agenesis (MRKH syndrome — absent uterus and upper vagina, normal ovaries, 46,XX, ~1:4,500 females), vaginal agenesis, cervical agenesis, uterine hypoplasia. May have associated renal anomalies (~30%).
Class II — Unicornuate Uterus: Failure of one Müllerian duct to develop. Subtypes: with communicating rudimentary horn, with non-communicating horn (risk of hematometra, ectopic pregnancy — recommend excision), without horn, without cavity. Associated with ipsilateral renal agenesis (~40%). Increased risk of preterm delivery, malpresentation, IUGR.
Class III — Uterus Didelphys: Complete failure of fusion — two separate uterine horns, two cervices, often a longitudinal vaginal septum. Generally best reproductive outcomes among major anomalies; may need cervical cerclage for preterm birth prevention.
Class IV — Bicornuate Uterus: Incomplete fusion of the Müllerian ducts, resulting in two uterine horns with a single cervix (bicornuate unicollis) or two cervices (bicornuate bicollis). Fundal indentation >1 cm on imaging. Associated with second-trimester loss and preterm delivery. Strassman metroplasty (unification surgery) is rarely performed; cerclage may be more appropriate.
Class V — Septate Uterus: Complete or partial failure of resorption of the midline septum. Most common anomaly associated with RPL. The septum is poorly vascularized — embryo implantation on the septum leads to miscarriage. Normal external uterine contour (distinguishes from bicornuate on MRI or 3D US). Hysteroscopic septum resection is performed in cases of RPL or possibly infertility (though the TRUST trial showed uncertain benefit for septum resection in preventing miscarriage in a general population).
Class VI — Arcuate Uterus: Minimal fundal indentation (<1 cm). Generally considered a normal variant without significant reproductive impact.
Class VII — DES-Related: T-shaped uterine cavity, hypoplastic uterus in women exposed to diethylstilbestrol in utero. Rare in current practice (DES was banned in 1971).
Diagnosis: 3D transvaginal ultrasound and MRI are the gold standard imaging modalities for classification. HSG shows the internal cavity but cannot assess external contour (cannot differentiate septate from bicornuate — the most important clinical distinction). The key differentiating feature: a septate uterus has a normal or minimally indented external fundal contour with an internal cavity division, while a bicornuate uterus has a significant external fundal cleft (>1 cm). On MRI, the zonal anatomy (junctional zone, myometrium) helps classify the anomaly. Laparoscopy + hysteroscopy provides definitive assessment (external contour + internal cavity) but is invasive and usually unnecessary given modern 3D imaging.
All patients with Müllerian anomalies should undergo renal imaging (ultrasound or CT) due to the embryologic association between Müllerian and mesonephric (Wolffian) duct development — renal agenesis is found in ~30-40% of unicornuate uterus patients and is ipsilateral to the absent horn. Skeletal anomalies may also be associated (vertebral anomalies, as in MURCS association: Müllerian duct aplasia, Renal aplasia, Cervicothoracic Somite dysplasia).
24 Menopause, POI & Perimenopause
STRAW+10 Staging System
The Stages of Reproductive Aging Workshop +10 (STRAW+10) classification divides reproductive aging into stages: Reproductive (early, peak, late — characterized by regular cycles, variable AMH); Menopausal transition (early — cycle length variability ≥7 days; late — skipped cycles, amenorrhea 60+ days); Postmenopause (early — within 6 years of final menstrual period; late — >6 years). Key biomarker changes: AMH becomes undetectable in the late menopausal transition, FSH rises progressively (>25 mIU/mL in late transition), and inhibin B becomes undetectable.
Menopause
Menopause is diagnosed retrospectively after 12 consecutive months of amenorrhea without other cause. Median age: 51 years (range 45-55). Pathophysiology: depletion of the ovarian follicle pool leads to loss of estrogen and progesterone production, loss of inhibin feedback, and markedly elevated FSH (>30 mIU/mL). Symptoms: vasomotor symptoms (hot flashes in ~75%, caused by thermoregulatory dysfunction from estrogen withdrawal acting on the hypothalamic KNDy neurons — kisspeptin, neurokinin B, dynorphin), genitourinary syndrome of menopause (GSM — vaginal dryness, dyspareunia, urinary urgency/frequency), sleep disturbance, mood changes, and cognitive changes. Long-term consequences: accelerated bone loss (most rapid in first 5 years, ~2-3% per year), increased cardiovascular disease risk, and increased fracture risk.
Premature Ovarian Insufficiency (POI)
POI is cessation of ovarian function before age 40 (see also Section 5, WHO Group III). Long-term health implications of prolonged hypoestrogenism: accelerated bone loss (osteoporosis risk), cardiovascular disease risk, cognitive decline, genitourinary symptoms. Management: HRT (estrogen + cyclic progesterone if uterus present) until the natural age of menopause (~50-51) for bone and cardiovascular protection. Fertility: donor oocytes are the primary option; spontaneous conception ~5-10%.
HRT in the REI Context
Hormone replacement therapy in POI and early menopause serves both symptom management and long-term health protection. Preferred regimen: transdermal estradiol (50-100 μg patch) + micronized progesterone (200 mg oral, 12-14 days per month if uterus present). Transdermal estrogen is preferred over oral because it avoids first-pass hepatic effects (lower VTE risk, less effect on SHBG/clotting factors, more physiologic steady-state levels). Combined oral contraceptives are an alternative in younger women who also desire contraception, though physiologic HRT is generally preferred for POI. Duration: continue until at least age 50-51. Key differences from postmenopausal HRT: POI patients are younger, have more years of estrogen deficiency, and the WHI findings (which studied women aged 50-79) do not apply to young women with POI — the benefits of HRT in POI clearly outweigh the risks.
Early Menopause vs. POI
Early menopause is defined as cessation of ovarian function between ages 40-45 (affects ~5% of women). While less severe than POI (<40), early menopause still carries increased cardiovascular and skeletal risks compared to menopause at the median age. HRT should be considered for these patients as well, particularly if symptomatic or with risk factors for osteoporosis or cardiovascular disease. Fertility implications are similar to POI — donor oocytes are the primary option, though ovarian reserve may be sufficient for IVF if detected early enough (AMH and AFC monitoring in women with family history of early menopause).
25 Disorders of Sexual Development
46,XX DSD
Congenital adrenal hyperplasia (CAH): The most common cause of 46,XX DSD and the most common autosomal recessive disorder affecting fertility. 21-hydroxylase deficiency (CYP21A2 mutations) accounts for ~95% of CAH. The enzyme block prevents cortisol synthesis, leading to ACTH elevation and shunting of steroid precursors into the androgen pathway.
Classic CAH (incidence ~1:15,000): two forms — salt-wasting (~75%, severe enzyme deficiency with both cortisol and aldosterone deficiency, presenting with adrenal crisis in neonatal period) and simple virilizing (~25%, cortisol deficiency without salt-wasting). Both cause ambiguous genitalia at birth in 46,XX individuals (clitoromegaly, labial fusion, urogenital sinus) from excess adrenal androgens. Elevated 17-OHP is diagnostic (often >10,000 ng/dL in classic CAH). Treatment: glucocorticoid replacement (hydrocortisone preferred in children, dexamethasone in adults) + mineralocorticoid (fludrocortisone) if salt-wasting. Fertility in classic CAH women: ~50-80% can achieve pregnancy with glucocorticoid optimization (suppresses excess androgens, restoring ovulation). Associated with PCOS-like phenotype, vaginal stenosis requiring surgical correction, and psychosexual considerations.
Non-classic (late-onset) CAH: Much more common (~1:100 to 1:1,000 depending on ethnicity — highest in Ashkenazi Jewish, Hispanic, Italian populations). Presents in adolescence/adulthood with hirsutism, acne, menstrual irregularity, and infertility (phenotypically mimics PCOS). No genital ambiguity at birth. Screening: fasting morning 17-OHP drawn in the early follicular phase. A level >200 ng/dL is suspicious; confirm with ACTH stimulation test (cosyntropin 250 μg IV, measure 17-OHP at 0 and 60 minutes) — stimulated 17-OHP >1,000 ng/dL is diagnostic. CYP21A2 genotyping confirms diagnosis and is essential for partner carrier screening (carrier frequency ~1:25 — if both partners carry mutations, offspring may have classic CAH). Fertility management: low-dose glucocorticoid (dexamethasone 0.25-0.5 mg nightly or prednisone 5-7.5 mg daily) suppresses adrenal androgens and restores ovulatory function in most cases. Many women with non-classic CAH conceive without treatment.
46,XY DSD
Complete androgen insensitivity syndrome (CAIS): 46,XY individuals with female external phenotype due to complete resistance to androgens (AR gene mutation, X-linked). Presents as primary amenorrhea with normal female external genitalia, absent uterus, short blind-ending vagina, and inguinal/abdominal testes. Testes produce AMH (no Müllerian structures) and testosterone (which cannot act). Gonadectomy recommended after puberty (allows spontaneous feminization) due to germ cell tumor risk (~2-5% in CAIS, higher in partial AIS). HRT after gonadectomy. No fertility with own gametes.
Partial androgen insensitivity syndrome (PAIS): Variable phenotype from predominantly female to ambiguous to predominantly male with undervirilization (hypospadias, micropenis, gynecomastia). Higher germ cell tumor risk than CAIS (15-20%). Fertility rarely possible. Gender assignment and management are complex and require multidisciplinary DSD team involvement.
5-alpha reductase deficiency: 46,XY with impaired conversion of testosterone to dihydrotestosterone (DHT) due to SRD5A2 gene mutations (autosomal recessive). Female or ambiguous genitalia at birth but virilization at puberty (testosterone rises, partial masculinization — voice deepening, phallic growth, muscular development). Classic description of "guevedoces" in Dominican Republic and "turnims" in Papua New Guinea. If raised male and testes present, fertility may be possible with assisted reproduction. Diagnosis: elevated testosterone-to-DHT ratio (>20:1) basally or after hCG stimulation.
Swyer syndrome (46,XY complete gonadal dysgenesis): Phenotypically female with streak gonads, normal Müllerian structures (uterus present, as gonads do not produce AMH), absent puberty. Gonadectomy is urgently indicated due to high gonadoblastoma/dysgerminoma risk (~30%). After gonadectomy, HRT for pubertal development. Fertility is possible via donor oocyte IVF and pregnancy in the patient's own uterus (with HRT support).
Turner Syndrome (45,X)
Turner syndrome affects ~1:2,500 live female births. Karyotype: 45,X (monosomy X) in ~50%; mosaicism (45,X/46,XX, 45,X/46,XY) in ~30%; structural X abnormalities (isochromosome Xq, ring X, partial deletion) in ~20%. Clinical features: short stature (average adult height ~4'8" without treatment), gonadal dysgenesis (streak gonads — fibrotic ovarian stroma without follicles), primary amenorrhea (though mosaic forms 45,X/46,XX may have some ovarian function and spontaneous puberty in ~10-20%), shield chest with widely spaced nipples, webbed neck, low posterior hairline, lymphedema, cubitus valgus. Associated anomalies: coarctation of the aorta (~10%), bicuspid aortic valve (~15-30%), horseshoe kidney, hypothyroidism, hearing loss, learning differences. Fertility: spontaneous pregnancy in ~2-5% (mainly mosaics); donor oocyte IVF is the primary option. Cardiac evaluation is mandatory before pregnancy — risk of aortic dissection is 2-6% during pregnancy (MRI of aorta required; aortic size index >2.0 cm/m² is a contraindication to pregnancy). Pregnancy in Turner syndrome is considered high-risk and requires multidisciplinary management.
Klinefelter Syndrome (47,XXY) — Fertility Aspects
Affects ~1:600 live male births; most common sex chromosome aneuploidy. The vast majority (~90%) are undiagnosed. Diagnosis is often made during infertility evaluation. Hormonal profile: FSH markedly elevated (>15-40 mIU/mL), LH elevated, testosterone low (150-300 ng/dL typically), inhibin B low or undetectable, estradiol may be relatively elevated (E2:T ratio increased due to increased aromatase activity in excess adipose tissue and Leydig cell dysfunction). Progressive testicular failure occurs from puberty onwards — Leydig cells become hyalinized and seminiferous tubules undergo fibrosis. Despite severe testicular dysfunction, focal islands of spermatogenesis may persist.
Fertility management: (1) Micro-TESE is the gold standard — retrieves sperm in ~40-70% of cases (higher success in younger patients, mosaic 46,XY/47,XXY, and those with higher pre-operative testosterone). The vast majority of sperm from Klinefelter patients are chromosomally normal (24,X or 24,Y) due to elimination of aneuploid germ cells during meiotic checkpoints, though rates of sex chromosome aneuploidy are slightly elevated. PGT-A is recommended for embryos created from Klinefelter sperm. (2) Adolescent fertility preservation: Emerging practice to offer sperm banking or micro-TESE to adolescent Klinefelter patients (ages 15-18) before progressive testicular deterioration, especially before testosterone replacement is initiated. (3) Testosterone replacement for hypogonadism (weakness, reduced bone density, metabolic syndrome) — but NOT if fertility is currently desired as it will further suppress residual spermatogenesis. Alternatives: clomiphene citrate (off-label) or hCG to boost endogenous testosterone while preserving spermatogenesis.
26 Oncofertility & Fertility Preservation
Oocyte & Embryo Cryopreservation
Oocyte vitrification is the standard for fertility preservation in post-pubertal females facing gonadotoxic therapy (chemotherapy, pelvic radiation) or elective social fertility preservation. A standard ovarian stimulation cycle (random-start protocol allows initiation regardless of cycle day — important when treatment cannot be delayed) yields 10-20 oocytes in ~2 weeks. Expected live birth rate: approximately 8-10% per oocyte vitrified in women <35 (so ~15-20 oocytes needed for a reasonable chance of at least one live birth). Embryo cryopreservation is an alternative for patients with a partner or willing to use donor sperm — similar stimulation process, followed by fertilization and blastocyst vitrification.
GnRH Agonist Co-Treatment
Administration of a GnRH agonist during chemotherapy (starting 1 week before and continuing throughout) may provide some ovarian protection by suppressing gonadotropin stimulation of follicles. The POEMS trial (Moore et al., NEJM 2015) showed improved rates of ovarian function recovery and pregnancy in breast cancer patients receiving GnRH agonist during chemotherapy. Should be used as an adjunct to, not a replacement for, oocyte/embryo cryopreservation.
Ovarian Tissue Cryopreservation (OTC)
Ovarian cortical tissue is removed laparoscopically (unilateral oophorectomy or cortical biopsy), cut into thin strips (1-2 mm thick to allow cryoprotectant penetration), and cryopreserved using slow-freeze or vitrification protocols. After cancer treatment and confirmation of remission, tissue is autotransplanted: orthotopic — to the remaining ovary, ovarian fossa, or pelvic peritoneum (allows natural conception in some cases); or heterotopic — to the forearm, abdominal wall, or subcutaneous tissue (restores endocrine function but requires oocyte retrieval for IVF). Transplanted tissue typically resumes function within 3-6 months (return of FSH suppression, E2 production, and menses). Graft longevity: typically 3-5 years per transplant; repeated transplantation of remaining tissue is possible.
Over 200 live births reported worldwide from OTC-transplant. The only established option for prepubertal girls (who cannot undergo ovarian stimulation for oocyte cryopreservation). Important concern: risk of reintroducing malignant cells with transplanted tissue — highest risk with hematologic malignancies (leukemia, lymphoma — ovarian tissue may harbor occult malignant cells); lowest risk with breast cancer, sarcoma. Histologic and molecular screening of tissue before transplant is performed but cannot guarantee absence of micrometastases. No longer considered experimental (ASRM 2019). Emerging alternatives: in vitro follicle growth from cryopreserved tissue (activating primordial follicles in vitro to produce mature oocytes without transplantation — still experimental in humans).
Male Fertility Preservation
Sperm banking (cryopreservation) is the standard for post-pubertal males facing gonadotoxic therapy (chemotherapy, radiation, surgery). It is simple, non-invasive, inexpensive, and effective. At least 2-3 samples should be collected before treatment initiation, ideally with 24-48 hour intervals between collections. Even severely oligozoospermic samples can be cryopreserved and used later for ICSI. Post-thaw motility is lower than fresh (~40-60% recovery), but even a few viable sperm are sufficient for ICSI.
For pre-pubertal males who cannot produce a semen sample: testicular tissue cryopreservation is the only option and remains experimental. The tissue contains spermatogonial stem cells (SSCs) that may potentially be used in the future for in vitro spermatogenesis, SSC transplantation, or testicular tissue grafting. Several research centers offer this under IRB-approved protocols. For adolescents with Tanner stage 2+ who may be able to produce sperm but cannot ejaculate by masturbation: electroejaculation under anesthesia, penile vibratory stimulation, or testicular sperm extraction (TESE) are options. Adolescent Klinefelter syndrome patients should be offered sperm banking or micro-TESE before testosterone replacement is initiated.
Timeline Considerations
Oncology-REI consultation should occur as soon as cancer diagnosis is made, before treatment initiation. A random-start stimulation cycle requires only ~2 weeks (can begin regardless of menstrual cycle day). For patients who cannot delay treatment even 2 weeks, GnRH agonist co-treatment or OTC may be the only options. ASCO and ASRM guidelines recommend discussing fertility preservation with all reproductive-age cancer patients before gonadotoxic therapy. Documentation of the fertility preservation discussion should be part of the oncology treatment plan.
Elective (Social) Fertility Preservation
Oocyte cryopreservation for age-related fertility decline (without a current medical indication) is increasingly common. ASRM has removed the "experimental" label from oocyte cryopreservation since 2012. Optimal age for elective freezing: <35 years (oocyte quality and quantity are highest). Studies suggest that the cost-effectiveness of elective freezing is optimal when performed at age 30-34. Counseling should address realistic expectations: the number of oocytes needed (a commonly cited benchmark is 15-20 mature oocytes for a reasonable chance of at least one live birth in women <35), that not all oocytes will survive thaw or produce a viable pregnancy, and that there is no guarantee of a live birth. Multiple stimulation cycles may be needed for adequate oocyte numbers.
High risk (>80% amenorrhea): Alkylating agents (cyclophosphamide — especially cumulative dose; busulfan, melphalan, chlorambucil), total body irradiation, pelvic radiation.
Intermediate risk: Cisplatin, doxorubicin (adriamycin).
Low risk: Taxanes (paclitaxel, docetaxel), vincristine, bleomycin, 5-FU, methotrexate.
Unknown/minimal risk: Targeted therapies (trastuzumab, imatinib), immunotherapy.
27 Third-Party Reproduction
Donor Oocytes
Indications: POI/premature menopause, diminished ovarian reserve with poor IVF prognosis after multiple cycles, repeated IVF failure with own oocytes, genetic disease in the female (when PGT-M is not desired or feasible), gonadal dysgenesis (Turner syndrome, Swyer syndrome), bilateral oophorectomy, advanced maternal age with very low success rates using autologous oocytes.
Donors are typically young women (21-34 years) who undergo standard ovarian stimulation and oocyte retrieval. Two models: fresh donor cycles (synchronization of donor stimulation and recipient endometrial preparation; oocytes fertilized on retrieval day) and frozen donor oocyte banks (oocytes vitrified and shipped to recipient's clinic; more convenient scheduling and wider donor selection but slightly lower per-oocyte survival). Donor oocytes are fertilized with the recipient's partner's (or donor) sperm, and embryos are transferred to the recipient's hormonally prepared uterus (programmed cycle with estrogen + progesterone). Success rates: ~50-55% live birth per transfer, largely independent of the recipient's age up to ~50 (because oocyte quality reflects the donor's age). After age 45-50, obstetric risks increase substantially (hypertension, preeclampsia, gestational diabetes).
Screening requirements (ASRM and FDA): FDA-mandated infectious disease testing (HIV, hepatitis B/C, syphilis, CMV, gonorrhea, chlamydia), expanded genetic carrier screening (≥100 conditions including CF, SMA, fragile X, hemoglobinopathies), psychological evaluation by a mental health professional, detailed family history (3 generations), physical examination, drug screening, and AMH/AFC to ensure adequate response potential. Known donors (friend or family member) undergo the same screening. Recipient counseling should address the unique psychological aspects of donor oocyte parenting, disclosure to offspring, and potential for later contact with the donor (open-identity programs).
Donor Sperm
Indications: Azoospermia with failed TESE or when TESE is not desired, severe male factor not amenable to ICSI, genetic disease in the male partner (when PGT-M is not feasible or desired), single women desiring pregnancy, and same-sex female couples. Commercial sperm banks provide cryopreserved donor sperm with extensive screening: FDA-mandated infectious disease testing (HIV, hepatitis B/C, syphilis, HTLV, CMV, gonorrhea, chlamydia), expanded genetic carrier screening, family medical history (3 generations), personal history, physical characteristics, educational background, and semen quality assessment. Donors are typically 18-39 years old with total motile count >40 million pre-freeze.
Processing and use: donor sperm undergoes a 6-month quarantine period (the donor is tested for infectious diseases at donation and retested 6 months later; sperm is released for use only after both screens are negative). Frozen donor sperm can be used for IUI (intracervical insemination is much less effective and rarely used) or IVF/ICSI. Post-thaw motility may be lower (~40-60% of pre-freeze), but is generally adequate for IUI if TMSC ≥5 million. Known/directed donors (friend or family member): undergo the same FDA-mandated screening; quarantine is recommended but can be waived in some states with physician documentation. Legal considerations: known donors may have parental rights claims depending on jurisdiction — legal counsel is advised before proceeding. Directed donation from a male partner (e.g., for fertility preservation before cancer treatment) follows standard sperm cryopreservation protocols without FDA donor screening requirements.
Gestational Carrier (Surrogacy)
Gestational carrier (GC) (the carrier has no genetic relationship to the child — embryo is created from intended parents' or donors' gametes) is distinguished from traditional surrogacy (carrier provides the oocyte — rarely practiced in medical settings due to legal and emotional complexities). Medical indications: absent uterus (MRKH syndrome, post-hysterectomy), severe uterine pathology (Asherman syndrome refractory to treatment, severe adenomyosis, multiple failed embryo transfers despite euploid embryos), medical contraindication to pregnancy (severe cardiac disease, Marfan syndrome with aortic root dilation, pulmonary hypertension, renal transplant with complications), recurrent pregnancy loss without identified treatable cause, and same-sex male couples.
GC screening: medical evaluation (confirmed prior uncomplicated pregnancy and delivery, age 21-45, BMI <35, non-smoker), psychological evaluation by a licensed mental health professional experienced in third-party reproduction, infectious disease screening, uterine cavity assessment. Obstetric outcomes with GC: generally comparable to age-matched spontaneous pregnancies, though slightly higher preeclampsia rates are reported (possibly due to immune mechanisms with a genetically unrelated pregnancy). Legal contracts between intended parents and carrier must be executed before embryo transfer; pre-birth parentage orders are available in some jurisdictions.
Legal & Ethical Considerations
Third-party reproduction raises complex legal and ethical issues: informed consent, anonymity vs. open-identity donors (trend toward open-identity globally), compensation (distinguishing reasonable payment from commodification — ASRM Ethics Committee guidelines cap oocyte donor compensation), legal parentage determination (pre-birth orders vs. post-birth adoption depending on jurisdiction), disposition of unused embryos (storage, donation, research, disposal — advance directives required), international surrogacy arrangements (concerns about exploitation), and the welfare of the child. ASRM Ethics Committee provides comprehensive guidance documents on these topics. Legal frameworks vary dramatically between states and countries — some jurisdictions prohibit surrogacy entirely, others permit only altruistic surrogacy, and some allow commercial surrogacy.
Reproductive Options for LGBTQ+ Individuals
REI plays a central role in family-building for LGBTQ+ individuals and couples. Same-sex female couples: Donor sperm insemination (IUI or IVF); reciprocal IVF (one partner provides oocytes, the other carries the pregnancy). Same-sex male couples: Donor oocytes + gestational carrier. Transgender individuals: Fertility preservation (sperm banking before feminizing hormones/orchiectomy; oocyte cryopreservation before masculinizing hormones/oophorectomy) should be discussed before gender-affirming therapy. Testosterone therapy causes amenorrhea and impaired folliculogenesis, but effects are often reversible upon discontinuation. ASRM Ethics Committee affirms that fertility services should be provided without discrimination based on sexual orientation or gender identity.
28 Classification Systems
WHO Classification of Anovulation
Group I — Hypogonadotropic Hypogonadism: Low FSH, low LH, low estrogen. Hypothalamic/pituitary failure (Kallmann, FHA, Sheehan). ~5-10% of anovulation.
Group II — Normogonadotropic Anovulation: Normal FSH/LH, normal estrogen. PCOS is the prototype. ~75-85% of anovulation.
Group III — Hypergonadotropic Hypogonadism: High FSH, low estrogen. Ovarian failure (POI). ~5-10% of anovulation.
Group IV — Hyperprolactinemic Anovulation: Elevated prolactin suppresses GnRH. Prolactinoma, medications.
Rotterdam PCOS Criteria (2003, Updated 2018)
Two of three criteria (after exclusion of other etiologies): (1) Oligo-anovulation (cycles >35 days, <8 cycles/year, or amenorrhea); (2) Clinical and/or biochemical hyperandrogenism (hirsutism — modified Ferriman-Gallwey score ≥4-6 depending on ethnicity, acne, androgenetic alopecia; or elevated total/free testosterone, androstenedione); (3) Polycystic ovarian morphology on ultrasound (≥20 follicles per ovary of 2-9 mm diameter and/or ovarian volume ≥10 mL — 2018 updated thresholds reflecting modern high-frequency transducers; the original 2003 threshold was ≥12 follicles, which is no longer valid with current ultrasound technology). Ultrasound criterion should NOT be used for diagnosis in adolescents <8 years post-menarche (ovarian morphology is unreliable in this group). Exclusions required before diagnosis: thyroid disease (TSH), non-classic congenital adrenal hyperplasia (17-OHP), Cushing syndrome (24-hour urine cortisol or overnight dexamethasone suppression test if clinical suspicion), androgen-secreting tumors (total testosterone >200 ng/dL or DHEA-S >700 μg/dL), hyperprolactinemia (prolactin).
ASRM Müllerian Anomaly Classification
See Section 23 for full classification: Class I (agenesis/hypoplasia — includes MRKH syndrome), Class II (unicornuate — with or without rudimentary horn), Class III (didelphys — complete non-fusion with two cervices), Class IV (bicornuate — partial non-fusion with fundal cleft >1 cm), Class V (septate — most common anomaly associated with RPL; failure of septal resorption), Class VI (arcuate — minimal variant, generally normal), Class VII (DES-related — T-shaped cavity). Note: The ESHRE/ESGE classification (2013) uses a different system (U0-U6) based on uterine morphology and is also widely used internationally.
rASRM Endometriosis Staging
See Section 8 for full staging details. Point-based system: Stage I (minimal, 1-5 points — superficial peritoneal implants), Stage II (mild, 6-15 points — superficial implants with some adhesions), Stage III (moderate, 16-40 points — deep implants, endometriomas, filmy adhesions), Stage IV (severe, >40 points — large endometriomas, dense adhesions, cul-de-sac obliteration). Important limitation: staging poorly predicts fertility outcomes. The Endometriosis Fertility Index (EFI) is more useful for predicting pregnancy after surgical treatment.
Bologna Criteria for Poor Ovarian Response (ESHRE 2011)
See Section 3 for full criteria. At least two of three: (1) Advanced maternal age (≥40) or other POR risk factor; (2) Previous POR (≤3 oocytes with conventional stimulation); (3) Abnormal ovarian reserve test (AFC <5-7 or AMH <0.5-1.1 ng/mL). Two episodes of POR after maximal stimulation are sufficient regardless of other criteria. Criticism: too heterogeneous (groups together patients with different prognoses). Superseded for many purposes by the POSEIDON classification.
POSEIDON Classification (2016)
See Section 3 for full detail. A more refined approach to the "low prognosis" IVF patient: Group 1 (age <35, normal reserve, unexpected poor response — best prognosis for quality), Group 2 (age ≥35, normal reserve, unexpected poor response), Group 3 (age <35, low reserve — expected poor response, good quality potential), Group 4 (age ≥35, low reserve — worst prognosis, both quantity and quality compromised). The POSEIDON concept defines an "optimal" endpoint: the minimum number of oocytes needed to achieve at least one euploid blastocyst for transfer. Management strategies are tailored to each group.
OHSS Classification (Golan)
Mild (Grade 1-2): Abdominal distension and discomfort, mild nausea, diarrhea. Ovaries enlarged <8 cm. No significant lab abnormalities. Incidence: 20-33% of stimulated cycles.
Moderate (Grade 3): Moderate abdominal pain, nausea/vomiting, ultrasonographic ascites, ovaries 8-12 cm. Incidence: 3-6%.
Severe (Grade 4): Clinical ascites ± hydrothorax, hemoconcentration (Hct >45%), WBC >15,000, oliguria (<300 mL/24hr), creatinine 1.0-1.5 mg/dL, liver dysfunction (elevated transaminases), ovaries >12 cm. Incidence: 0.1-2%.
Critical (Grade 5): Tense ascites, massive hydrothorax, pericardial effusion, Hct >55%, WBC >25,000, renal failure (creatinine >1.6 mg/dL or anuria), venous thromboembolism (DVT, PE, stroke, arterial thrombosis), ARDS, sepsis. Incidence: <0.1%. Potentially fatal.
Early vs Late OHSS: Early OHSS occurs 3-7 days after trigger (response to exogenous hCG trigger). Late OHSS occurs 12-17 days after trigger (driven by endogenous hCG from implanting embryo). Late OHSS is prevented by freeze-all strategy (no pregnancy = no endogenous hCG).
Gardner Blastocyst Grading
See Section 16 for full grading system. Three-part score: Expansion (1-6, where 1 = early blastocyst and 6 = hatched), ICM quality (A = tightly packed many cells, B = loosely grouped several cells, C = few cells), TE quality (A = many cells cohesive layer, B = few cells loose epithelium, C = very few large cells). Good quality for transfer: ≥3BB. Excellent quality: 4AA, 5AA. Fair quality: 3BC, 4BC. Poor quality (low implantation potential): any C/C combination. The grading system correlates with implantation rates: 4AA ~60-70%, 4BB ~40-50%, 4BC ~20-30%. PGT-A ploidy status is a stronger predictor of implantation than morphology grade, but among euploid embryos, higher morphology grades still predict better outcomes.
WHO Semen Analysis Reference Values (6th Edition, 2021)
See Section 4 for full reference values. Key updates from the 5th edition (2010): Volume decreased from ≥1.5 mL to ≥1.4 mL; Concentration from ≥15 M/mL to ≥16 M/mL; Total sperm number from ≥39 M to ≥39 M (unchanged); Total motility from ≥40% to ≥42%; Progressive motility from ≥32% to ≥30%; Morphology unchanged at ≥4% (Kruger strict criteria); Vitality from ≥58% to ≥54%. These values represent the 5th percentile of recent fathers (not the minimum for conception). Men with values below these thresholds have reduced but not zero fertility potential — the relationship between semen parameters and fertility is a continuum, not a threshold.
Varicocele Grading
Grade I: palpable with Valsalva only. Grade II: palpable without Valsalva. Grade III: visible through scrotal skin. Subclinical varicocele: detectable only by ultrasound (testicular vein diameter >3 mm with retrograde flow on Valsalva) — surgical repair is NOT indicated for subclinical varicocele.
FIGO Fibroid Classification (PALM-COEIN System)
Submucosal (impacting the cavity):
Type 0: Pedunculated intracavitary (entirely within the cavity, attached by a stalk). Type 1: <50% intramural component (predominantly intracavitary). Type 2: ≥50% intramural component (predominantly intramural but distorting the cavity).
Other (not distorting the cavity):
Type 3: 100% intramural, contacts the endometrium but does not distort it. Type 4: Intramural (entirely within the myometrium, no endometrial or serosal contact). Type 5: Subserosal (≥50% intramural). Type 6: Subserosal (<50% intramural). Type 7: Subserosal pedunculated (attached to the serosal surface by a stalk). Type 8: Other/does not fit above categories (cervical, parasitic, broad ligament).
Hybrid fibroids (extending from endometrium to serosa) are described with two numbers: e.g., Type 2-5 = intramural fibroid with both submucosal and subserosal components.
Endometriosis Fertility Index (EFI)
Developed by Adamson and Pasta (2010). Combines historical factors (age 0-2 points, years of infertility 0-2, prior pregnancy history 0-2) with surgical factors (total AFS/rASRM endometriosis score 0-1, total AFS adhesion score 0-1, least function score for tubes 0-4, fimbriae 0-4, ovaries 0-4 — from which the lowest score is selected). Total EFI score range: 0-10. Interpretation: EFI ≥9: ~55% 3-year non-ART pregnancy rate; EFI 5-8: ~30-40% 3-year rate; EFI ≤4: ~15% 3-year rate — recommend proceeding directly to IVF rather than expectant management or further surgical intervention.
Sapporo/Sydney Criteria for Antiphospholipid Syndrome
Requires at least one clinical criterion AND one laboratory criterion. Clinical criteria: (1) Vascular thrombosis (arterial, venous, or small vessel); (2) Pregnancy morbidity (≥1 unexplained fetal death ≥10 weeks, or ≥1 premature birth <34 weeks due to preeclampsia/placental insufficiency, or ≥3 consecutive unexplained miscarriages <10 weeks). Laboratory criteria (must be positive on two occasions ≥12 weeks apart): (1) Lupus anticoagulant (LA), (2) Anticardiolipin antibodies (aCL IgG or IgM, medium or high titer), (3) Anti-β2-glycoprotein I antibodies (IgG or IgM). Triple positivity (all three markers positive) carries the highest thrombotic and obstetric risk.
29 Medications Master Table
GnRH Agonists
Leuprolide acetate (Lupron): SC injection. IVF long protocol: 0.5-1.0 mg daily (downregulation); reduced to 0.25 mg during stimulation. Microdose flare: 20-40 μg BID. Trigger: 1-4 mg single dose (antagonist protocol only). Depot: 3.75 mg IM monthly for endometriosis/fibroid suppression. Side effects: hot flashes, headache, bone loss with prolonged use.
Nafarelin (Synarel): Intranasal spray 200-400 μg BID. Used for endometriosis suppression and IVF downregulation.
Goserelin (Zoladex): 3.6 mg SC implant monthly. Endometriosis, fibroid shrinkage, oncofertility co-treatment.
GnRH Antagonists
Ganirelix (Orgalutran): 0.25 mg SC daily. Started when lead follicle ≥13-14 mm or fixed day 5-6. Immediate LH suppression, no flare. Continued until trigger day (inclusive).
Cetrorelix (Cetrotide): 0.25 mg SC daily (same protocol as ganirelix). Also available as 3 mg single dose (provides ~4 days of suppression).
Gonadotropins
Follitropin alfa (Gonal-F): Recombinant FSH. Multi-dose pen or vial. Stimulation: 150-450 IU/day SC. Ovulation induction: 75-150 IU/day (low-dose step-up).
Follitropin beta (Follistim/Puregon): Recombinant FSH. Pen device. Dosing similar to Gonal-F.
Follitropin delta (Rekovelle): Recombinant FSH dosed by body weight and AMH (individualized dosing algorithm).
Menotropins/hMG (Menopur): Highly purified urinary FSH + LH activity (75 IU each per vial). SC injection. May benefit patients who need LH supplementation.
Corifollitropin alfa (Elonva): Long-acting recombinant FSH. Single SC injection replaces 7 days of daily FSH. 100 μg (≤60 kg) or 150 μg (>60 kg).
Lutropin alfa (Luveris): Recombinant LH. 75 IU SC daily. Used with FSH in hypogonadotropic hypogonadism or advanced age.
Ovulation Induction Agents
Letrozole (Femara): Aromatase inhibitor. 2.5-7.5 mg PO daily, cycle days 3-7. First-line for PCOS ovulation induction (PPCOS II). No anti-estrogenic endometrial/cervical effects. Half-life: ~48 hours (cleared before implantation). Off-label for infertility.
Clomiphene citrate (Clomid, Serophene): Selective estrogen receptor modulator (SERM). 50-150 mg PO daily, cycle days 3-7 or 5-9. Blocks estrogen feedback at hypothalamus/pituitary, increasing FSH release. Anti-estrogenic effects: thin endometrium, hostile cervical mucus. Max 6 cycles. Side effects: hot flashes, visual disturbances, mood changes, ovarian cysts.
hCG Trigger & Luteal Support
Choriogonadotropin alfa (Ovidrel): Recombinant hCG 250 μg SC. Triggers ovulation 36 hours after injection. Also used for luteal support (1,500 IU every 3 days — increases OHSS risk).
Urinary hCG (Pregnyl, Novarel): 5,000-10,000 IU IM. Same indication as Ovidrel.
Micronized progesterone (Prometrium): 200 mg PO or vaginal BID-TID for luteal support. Vaginal route achieves higher endometrial concentrations.
Progesterone vaginal inserts (Endometrin): 100 mg vaginally BID-TID.
Progesterone in oil: 50-100 mg IM daily. Reliable absorption but painful.
Crinone 8% vaginal gel: 90 mg progesterone vaginally daily.
Dydrogesterone (Duphaston): 10 mg PO TID. Oral progestogen for luteal support (LOTUS I/II trials).
Other Medications
Cabergoline (Dostinex): Dopamine agonist. 0.5 mg PO daily for 8 days from trigger for OHSS prevention. 0.25-1 mg PO twice weekly for hyperprolactinemia.
Bromocriptine (Parlodel): Dopamine agonist. 1.25-5 mg PO BID. Hyperprolactinemia (preferred in pregnancy due to longer safety record).
Metformin (Glucophage): Insulin sensitizer. 1,500-2,550 mg PO daily (titrated). PCOS metabolic management. Adjunct to ovulation induction. GI side effects (use extended-release to minimize). Continue through first trimester if conceiving (may reduce miscarriage in PCOS).
Estradiol (Estrace, Vivelle, Climara): Oral 2-8 mg daily or transdermal patches (0.1-0.4 mg). Endometrial preparation for FET. Asherman syndrome post-adhesiolysis. HRT.
Doxycycline: 100 mg PO BID. Prophylactic antibiotic for oocyte retrieval, HSG, and hysteroscopy. Treatment of chronic endometritis (14-28 days).
Aspirin (low-dose, 81 mg): Used in APS treatment with heparin. Some evidence for thin endometrium. Routine use in IVF is not supported by strong evidence.
Enoxaparin (Lovenox): LMWH. 40 mg SC daily. APS treatment in pregnancy. VTE prophylaxis in OHSS.
30 Abbreviations Master List
AFC — Antral Follicle Count
AFS — American Fertility Society (now ASRM)
AIS — Androgen Insensitivity Syndrome
AMH — Anti-Müllerian Hormone
APS — Antiphospholipid Syndrome
ART — Assisted Reproductive Technology
ASRM — American Society for Reproductive Medicine
AZF — Azoospermia Factor (Y chromosome regions a, b, c)
BMI — Body Mass Index
CAH — Congenital Adrenal Hyperplasia
CBAVD — Congenital Bilateral Absence of the Vas Deferens
CCCT — Clomiphene Citrate Challenge Test
CF — Cystic Fibrosis
CFTR — Cystic Fibrosis Transmembrane Conductance Regulator
COH — Controlled Ovarian Hyperstimulation
DES — Diethylstilbestrol
DHEA-S — Dehydroepiandrosterone Sulfate
DIE — Deep Infiltrating Endometriosis
DOR — Diminished Ovarian Reserve
DSD — Disorders of Sexual Development
E2 — Estradiol
EFI — Endometriosis Fertility Index
ERA — Endometrial Receptivity Analysis
ESHRE — European Society of Human Reproduction and Embryology
eSET — Elective Single Embryo Transfer
FET — Frozen Embryo Transfer
FHA — Functional Hypothalamic Amenorrhea
FIGO — International Federation of Gynecology and Obstetrics
FMR1 — Fragile X Messenger Ribonucleoprotein 1
FSH — Follicle-Stimulating Hormone
GnRH — Gonadotropin-Releasing Hormone
hCG — Human Chorionic Gonadotropin
hMG — Human Menopausal Gonadotropin
HPO — Hypothalamic-Pituitary-Ovarian (axis)
HRT — Hormone Replacement Therapy
HSG — Hysterosalpingography
ICSI — Intracytoplasmic Sperm Injection
IUI — Intrauterine Insemination
IVA — In Vitro Activation
IVF — In Vitro Fertilization
LH — Luteinizing Hormone
LMWH — Low Molecular Weight Heparin
LOD — Laparoscopic Ovarian Drilling
MESA — Microsurgical Epididymal Sperm Aspiration
MRKH — Mayer-Rokitansky-Küster-Hauser (syndrome)
NGS — Next-Generation Sequencing
NOA — Non-Obstructive Azoospermia
OA — Obstructive Azoospermia
OHSS — Ovarian Hyperstimulation Syndrome
OTC — Ovarian Tissue Cryopreservation
PCOS — Polycystic Ovary Syndrome
PGT-A — Preimplantation Genetic Testing for Aneuploidy
PGT-M — Preimplantation Genetic Testing for Monogenic disorders
PGT-SR — Preimplantation Genetic Testing for Structural Rearrangements
PID — Pelvic Inflammatory Disease
POI — Premature Ovarian Insufficiency
POR — Poor Ovarian Response
rASRM — revised American Society for Reproductive Medicine (staging)
RPL — Recurrent Pregnancy Loss
SART — Society for Assisted Reproductive Technology
SET — Single Embryo Transfer
SIS/SHG — Saline Infusion Sonohysterography
STRAW — Stages of Reproductive Aging Workshop
TE — Trophectoderm
TESA — Testicular Sperm Aspiration
TESE — Testicular Sperm Extraction
TRUS — Transrectal Ultrasound
TURED — Transurethral Resection of the Ejaculatory Ducts
VEGF — Vascular Endothelial Growth Factor
WHO — World Health Organization
17-OHP — 17-Hydroxyprogesterone
31 Landmark Trials
PPCOS II (Legro et al., NEJM 2014): Letrozole vs. clomiphene for ovulation induction in PCOS. Letrozole: higher live birth rate (27.5% vs. 19.1%, p = 0.007), higher ovulation rate, lower multiple pregnancy rate. Established letrozole as first-line ovulation induction agent for PCOS. PMID: 25006718
AMIGOS (Diamond et al., Fertil Steril 2015): Assessment of Multiple Intrauterine Gestations from Ovarian Stimulation. Compared clomiphene/IUI, gonadotropin/IUI, and letrozole/IUI for unexplained infertility. Similar live birth rates per cycle (~8-10%); gonadotropins had highest multiple rate. Supported oral agents as first-line for unexplained infertility with IUI. PMID: 26597629
FASTT (Reindollar et al., Fertil Steril 2010): Fast Track and Standard Treatment trial. Accelerated treatment (3 cycles clomiphene/IUI then IVF) vs. conventional (3 cycles clomiphene/IUI, 3 cycles gonadotropin/IUI, then IVF) for unexplained infertility. Similar live birth rates but accelerated group reached pregnancy faster and more cost-effectively, especially in women ≥38. PMID: 20004371
OPTIMIST (van Tilborg et al., Lancet 2017): Individualized FSH dosing based on AMH in IVF. Tailored dosing did not improve live birth rates compared to standard dosing but reduced OHSS in high responders and reduced cancellation in poor responders. PMID: 27939963
E-Freeze (Vuong et al., NEJM 2018): Freeze-only vs. fresh embryo transfer in IVF for PCOS (non-IVF infertile women). Frozen transfer: higher ongoing pregnancy rate (49.3% vs. 42.0%), lower OHSS rate. Supported freeze-all in PCOS patients. PMID: 29320655
Fresh vs. Frozen in Normal Responders (Shi et al., NEJM 2018): In first IVF cycles for infertile women without PCOS, frozen-thaw transfer did not result in significantly higher live birth rates than fresh transfer. Supported continued use of fresh transfer as a valid strategy. PMID: 29320657
H2Oil Trial (Dreyer et al., NEJM 2017): Oil-based contrast (Lipiodol) vs. water-based contrast for HSG. Oil-based group: higher ongoing pregnancy rate (39.7% vs. 29.1%). Suggested a therapeutic fertility-enhancing effect of oil-based HSG contrast, possibly through tubal flushing or immunomodulation. PMID: 28557926
PRISM (Coomarasamy et al., NEJM 2019): Progesterone in Spontaneous Miscarriage. Vaginal micronized progesterone (400 mg BID) in women with early pregnancy bleeding. Subgroup analysis: significant live birth benefit in women with ≥3 prior miscarriages. Supported progesterone use for women with RPL and bleeding in early pregnancy. PMID: 31116916
TRUST (Rikken et al., NEJM 2021): Trial on Uterine Septum Transection. Hysteroscopic septum resection vs. expectant management in women with a septate uterus and RPL or infertility. No significant difference in live birth rate. Challenged the routine recommendation for septum resection. PMID: 34133864
ALIFE2 (Goddijn et al., NEJM 2023): Anticoagulants for Living Fetuses in Women With Recurrent Miscarriage and Inherited Thrombophilia. LMWH did not increase live birth rate compared to standard care in women with RPL and inherited thrombophilia. Changed practice by demonstrating lack of benefit. PMID: 36884325
POEMS (Moore et al., NEJM 2015): Prevention of Early Menopause Study. GnRH agonist (goserelin) during chemotherapy for breast cancer. GnRH agonist group: less ovarian failure (8% vs. 22%), more pregnancies. Supported GnRH agonist co-treatment for ovarian protection during chemotherapy. PMID: 25693012
LOTUS I & II (Tournaye et al., NEJM 2017; Griesinger et al., Fertil Steril 2018): Oral dydrogesterone vs. vaginal micronized progesterone for luteal support in IVF. Non-inferiority demonstrated. Provided an oral alternative for progesterone luteal support. PMID: 28586796
Marcoux et al. (NEJM 1997): Laparoscopic surgery vs. diagnostic laparoscopy for minimal-mild endometriosis-associated infertility. Surgery improved pregnancy rate (30.7% vs. 17.7%). Supported surgical treatment of mild endometriosis for fertility. PMID: 9211802
STAR Trial (Munne et al., Fertil Steril 2019): Single Thawed Euploid Embryo Transfer. PGT-A improved ongoing pregnancy rates per transfer (compared to morphology-based selection) in women 25-40. Supported PGT-A use for embryo selection, particularly in women ≥35. PMID: 31088710
Key References
Legro RS et al. Letrozole versus clomiphene for infertility in the polycystic ovary syndrome. N Engl J Med. 2014;371(2):119-129. PMID: 25006718
Vuong LN et al. IVF transfer of fresh or frozen embryos in women without polycystic ovaries. N Engl J Med. 2018;378(2):137-147. PMID: 29320655
Marcoux S et al. Laparoscopic surgery in infertile women with minimal or mild endometriosis. N Engl J Med. 1997;337(4):217-222. PMID: 9211802
Practice Committee of ASRM. Diagnostic evaluation of the infertile female: a committee opinion. Fertil Steril. 2015;103(6):e44-e50.
Teede HJ et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Hum Reprod. 2018;33(9):1602-1618.
Harlow SD et al. Executive summary of the Stages of Reproductive Aging Workshop + 10: addressing the unfinished agenda of staging reproductive aging. J Clin Endocrinol Metab. 2012;97(4):1159-1168.
WHO. WHO Laboratory Manual for the Examination and Processing of Human Semen, 6th edition. Geneva: World Health Organization; 2021.
ESHRE Guideline Group on POI. ESHRE Guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926-937.
Coomarasamy A et al. A randomized trial of progesterone in women with bleeding in early pregnancy. N Engl J Med. 2019;380(19):1815-1824. PMID: 31116916
Moore HCF et al. Goserelin for ovarian protection during breast-cancer adjuvant chemotherapy. N Engl J Med. 2015;372(10):923-932. PMID: 25693012