Urology
Every diagnosis, procedure, surgical technique, classification, complication, medication, and management algorithm across the full scope of urology in one place.
01 Upper Urinary Tract Anatomy
Kidneys
The kidneys are paired retroperitoneal organs lying at the T12-L3 vertebral level. The right kidney sits slightly lower than the left due to the liver. Each kidney measures approximately 10-12 cm in length, 5-7 cm in width, and 3 cm in anteroposterior diameter, weighing 120-170 g. The kidney is surrounded by Gerota fascia (renal fascia), within which lies perinephric fat. Deep to this is the true renal capsule.
The renal parenchyma consists of an outer cortex and an inner medulla. The medulla is organized into 8-18 renal pyramids, each with its apex (papilla) projecting into a minor calyx. Minor calyces merge into 2-3 major calyces, which join to form the renal pelvis. The functional unit is the nephron — each kidney contains approximately 1 million nephrons. The renal hilum transmits the renal artery, renal vein, renal pelvis, lymphatics, and autonomic nerves. The relationship from anterior to posterior at the hilum is: vein, artery, pelvis (mnemonic: VAP).
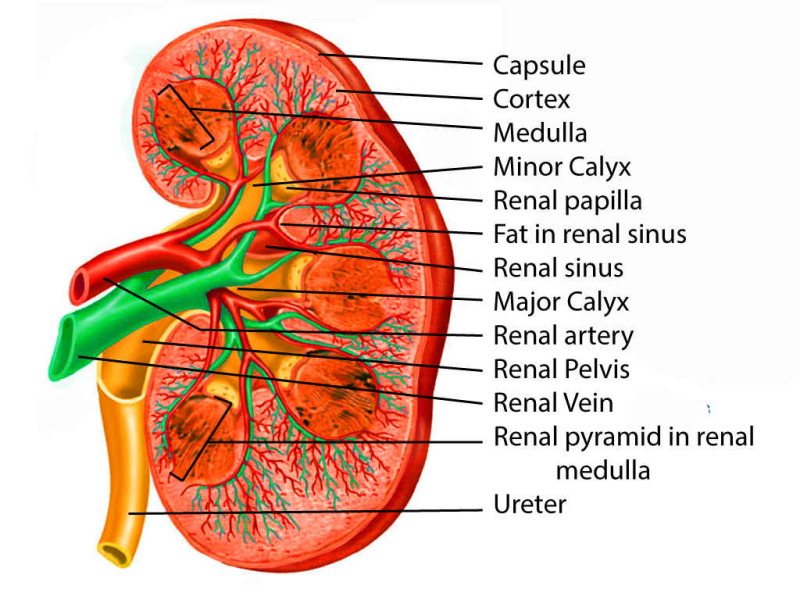
Renal Vascular Anatomy
The renal arteries arise from the aorta at the L1-L2 level. The right renal artery passes posterior to the IVC. Each renal artery divides into 5 segmental arteries (apical, upper, middle, lower, posterior) — these are end arteries with no collateral circulation, meaning occlusion causes segmental infarction. The segmental arteries further divide: segmental → interlobar → arcuate → interlobular → afferent arteriolar → glomerular capillaries → efferent arteriolar → peritubular capillaries/vasa recta.
The left renal vein is longer than the right, passing anterior to the aorta and posterior to the SMA (the "nutcracker" position). It receives the left gonadal vein, left adrenal vein, and lumbar veins — making left-sided donor nephrectomy preferred (longer vein for anastomosis). The right renal vein drains directly into the IVC with no major tributaries. The right gonadal vein drains directly into the IVC, while the left gonadal vein drains into the left renal vein — this explains why left varicoceles are far more common than right.
Ureters
The ureters are 25-30 cm muscular tubes that transport urine from the renal pelvis to the bladder via peristalsis. They cross the pelvic brim at the bifurcation of the common iliac artery. Three anatomic points of narrowing (sites where stones commonly impact): (1) the ureteropelvic junction (UPJ), (2) the crossing of the iliac vessels at the pelvic brim, and (3) the ureterovesical junction (UVJ) — the narrowest point.
The ureter has a three-layered wall: inner transitional epithelium (urothelium), middle smooth muscle (inner longitudinal, middle circular, outer longitudinal), and outer adventitia. Blood supply comes from multiple sources: renal artery superiorly, gonadal artery and aorta in the mid-portion, and iliac and superior vesical arteries inferiorly. The blood supply approaches medially in the upper ureter and laterally in the lower ureter — critical during surgical mobilization to avoid devascularization.
Adrenal Glands
The adrenal glands sit atop the kidneys within Gerota fascia. The right adrenal is pyramidal and sits behind the IVC; its venous drainage is a short vein directly into the IVC (injury here during right adrenalectomy or nephrectomy can cause significant hemorrhage). The left adrenal is crescent-shaped and drains via a longer vein into the left renal vein.
Lymphatic Drainage of the GU Tract
Knowledge of lymphatic drainage is critical for surgical oncology. The kidneys drain to para-aortic and paracaval lymph nodes (left kidney → left para-aortic; right kidney → right paracaval and interaortocaval). The testes drain to the retroperitoneal (para-aortic) lymph nodes at the level of the renal hilum — NOT the inguinal nodes (unless the scrotal skin has been violated). The bladder drains to the obturator and internal iliac nodes, then external iliac and common iliac nodes. The prostate drains primarily to the obturator lymph nodes (most common site of prostate cancer lymph node metastasis), followed by internal iliac and presacral nodes. The penis and scrotal skin drain to the superficial and deep inguinal lymph nodes.
Innervation of the Lower Urinary Tract
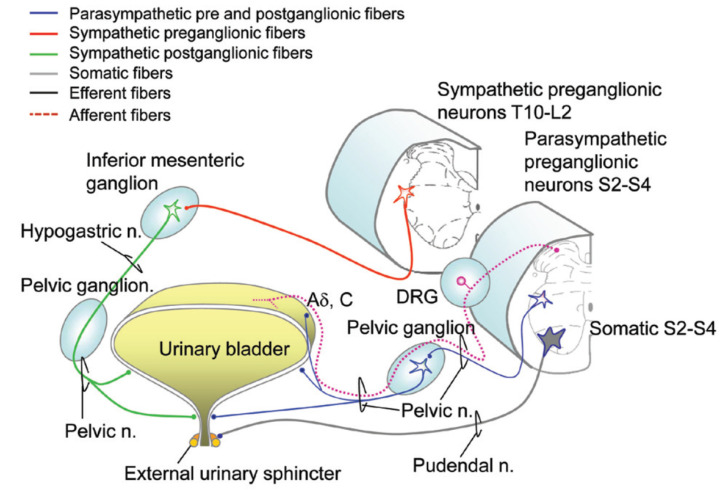
Three neural pathways control micturition: (1) Parasympathetic (S2-S4, pelvic nerve) — acetylcholine acts on M3 receptors on the detrusor, causing contraction (voiding). (2) Sympathetic (T10-L2, hypogastric nerve) — norepinephrine acts on beta-3 receptors in the detrusor body (relaxation/storage) and alpha-1 receptors at the bladder neck and prostatic urethra (contraction/continence). (3) Somatic (S2-S4, pudendal nerve) — voluntary control of the external urethral sphincter (striated muscle) via acetylcholine at nicotinic receptors. The pontine micturition center (Barrington nucleus) coordinates the voiding reflex — simultaneous detrusor contraction and sphincter relaxation. Understanding these pathways is essential for pharmacologic management of LUTS and neurogenic bladder.
02 Lower Urinary Tract & Male Genital Anatomy
Urinary Bladder
The bladder is a hollow muscular organ with a capacity of 400-600 mL. It lies in the retropubic space (space of Retzius) when empty. The bladder wall consists of: mucosa (transitional epithelium/urothelium), lamina propria, detrusor muscle (three layers of smooth muscle — inner longitudinal, middle circular, outer longitudinal), and serosa/adventitia. The trigone is the triangular area bounded by the two ureteral orifices and the internal urethral meatus — it is the most fixed and least distensible part of the bladder. The detrusor muscle is innervated by parasympathetic fibers (S2-S4) via the pelvic nerve; contraction causes voiding.
The bladder blood supply comes from the superior and inferior vesical arteries (branches of the internal iliac artery). Venous drainage is via the vesical venous plexus into the internal iliac veins. In males, the vesical plexus communicates with the prostatic venous plexus (Santorini plexus) — this valveless venous system (Batson plexus) communicates with the vertebral venous plexus, providing a route for hematogenous metastasis of prostate and bladder cancer to the spine without passing through the lungs.
Prostate
The prostate is a walnut-sized gland (~20 g in young men) that surrounds the prostatic urethra below the bladder neck. It is divided into zones (McNeal): the peripheral zone (70% of glandular tissue — site of ~70-80% of prostate cancers and where DRE palpates), the transition zone (5-10% of tissue — site of BPH), the central zone (25% — surrounds ejaculatory ducts), and the anterior fibromuscular stroma (nonglandular). The prostate is bounded posteriorly by Denonvilliers fascia (separating it from the rectum) and laterally by the neurovascular bundles (NVB) — containing the cavernous nerves responsible for erectile function. Preservation of the NVBs during radical prostatectomy is critical for postoperative potency.
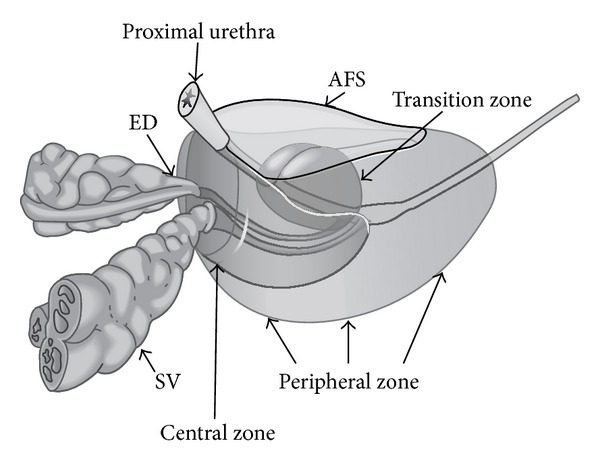
Male Urethra
The male urethra is approximately 18-22 cm and has four segments: (1) prostatic urethra (~3-4 cm, widest and most distensible — verumontanum marks the opening of ejaculatory ducts and prostatic utricle), (2) membranous urethra (~1-2 cm, passes through the external urethral sphincter and urogenital diaphragm — the most common site of traumatic urethral injury in pelvic fractures), (3) bulbar urethra (~3-4 cm, surrounded by bulbospongiosus muscle, most common site of stricture from instrumentation/infection), and (4) penile (pendulous) urethra (~15 cm, within the corpus spongiosum, terminating at the fossa navicularis and external meatus).
Female Urethra
The female urethra is approximately 3-5 cm in length, running from the bladder neck to the external meatus anterior to the vaginal opening. It is supported by the pubourethral ligaments and the anterior vaginal wall. The external urethral sphincter (rhabdosphincter) provides voluntary continence and is innervated by the pudendal nerve (S2-S4). The internal sphincter (smooth muscle at the bladder neck) provides involuntary continence and is innervated by sympathetic fibers (alpha-1 adrenergic). The shorter urethral length and proximity to the vaginal introitus and anus contribute to the higher incidence of urinary tract infections in women compared to men.
Support of the female urethra is provided by the pubourethral ligaments, endopelvic fascia, and the levator ani muscles (specifically the pubococcygeus component). Weakening of these support structures — from childbirth, aging, chronic straining, or connective tissue disorders — leads to urethral hypermobility, which is the most common mechanism of stress urinary incontinence in women.
Penis
The penis contains three erectile bodies: two dorsal corpora cavernosa (responsible for rigidity during erection) and one ventral corpus spongiosum (surrounds the urethra, expands distally into the glans penis). The corpora cavernosa are surrounded by the tunica albuginea — a tough, fibrous sheath. Arterial supply is from the internal pudendal artery, which gives rise to the cavernosal (deep) arteries, dorsal artery, and bulbourethral artery. Venous drainage occurs via the deep dorsal vein (beneath Buck fascia) draining into the prostatic venous plexus. Erection is a parasympathetic event (S2-S4, via NO-mediated smooth muscle relaxation); ejaculation is sympathetic (T10-L2).
Testes, Epididymis & Spermatic Cord
The testes are ovoid organs (~4 × 3 × 2.5 cm, ~20 mL volume) within the scrotum. Each testis is covered by the tunica vaginalis (a peritoneal remnant) and the tunica albuginea. The testicular parenchyma contains seminiferous tubules (site of spermatogenesis) and Leydig cells (produce testosterone in the interstitium). Spermatozoa pass from the seminiferous tubules through the rete testis into the epididymis (head/caput → body/corpus → tail/cauda). The cauda epididymis is continuous with the vas deferens.
The spermatic cord contains: vas deferens, testicular artery (from aorta), artery of the vas deferens (from inferior vesical artery), cremasteric artery (from inferior epigastric artery), pampiniform venous plexus, genital branch of the genitofemoral nerve, lymphatics, and the processus vaginalis remnant. Three fascial layers cover the cord (corresponding to abdominal wall layers encountered during inguinal descent): external spermatic fascia (from external oblique aponeurosis), cremasteric fascia and muscle (from internal oblique), and internal spermatic fascia (from transversalis fascia). The ilioinguinal nerve (L1) runs on the surface of the spermatic cord within the inguinal canal and provides sensation to the medial thigh, root of the penis, and anterior scrotum — it can be injured during inguinal surgery causing chronic groin pain.
The seminal vesicles are paired accessory glands posterior to the bladder base that contribute ~70% of ejaculate volume (fructose-rich alkaline fluid). They lie just superior to the prostate and lateral to the ampullae of the vas deferens. Each seminal vesicle duct joins with the ipsilateral vas deferens to form the ejaculatory duct, which traverses the prostate to empty at the verumontanum. Ejaculatory duct obstruction (EDO) is a treatable cause of male infertility — diagnosed by low ejaculate volume, azoospermia/oligospermia, and dilated seminal vesicles on TRUS; treated by transurethral resection of the ejaculatory ducts (TURED).
03 Renal Physiology & Fluid/Electrolyte Basics
Glomerular Filtration
The glomerular filtration rate (GFR) is approximately 120 mL/min (180 L/day) in a healthy adult. Filtration is driven by net filtration pressure — the balance of hydrostatic and oncotic pressures across the glomerular capillary. The GFR is regulated by: myogenic reflex (afferent arteriolar constriction in response to increased pressure), tubuloglomerular feedback (macula densa senses NaCl delivery and modulates afferent tone), and RAAS (angiotensin II preferentially constricts the efferent arteriole, maintaining GFR in low-flow states). ACE inhibitors and ARBs reduce efferent arteriolar tone, which lowers intraglomerular pressure — protective in diabetic nephropathy but can precipitate acute kidney injury in bilateral renal artery stenosis.
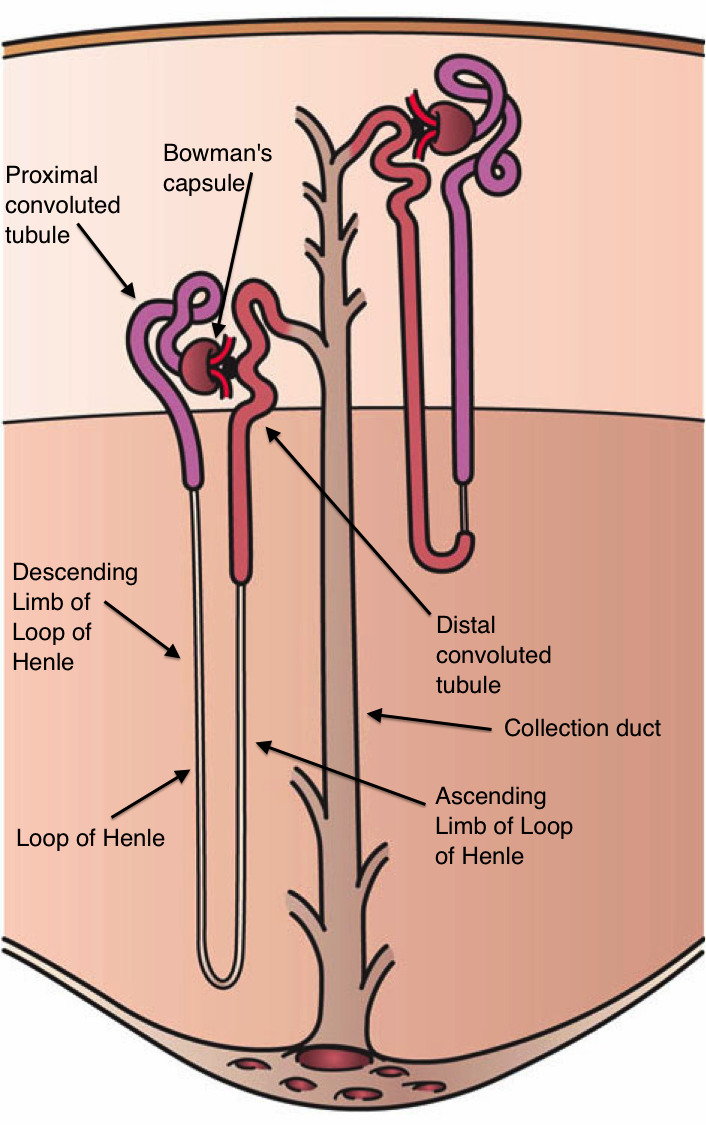
Tubular Function
The proximal convoluted tubule (PCT) reabsorbs ~65% of filtered Na+, water, glucose, amino acids, bicarbonate, phosphate, and uric acid. The loop of Henle establishes the medullary concentration gradient via countercurrent multiplication — the thick ascending limb actively reabsorbs NaCl via the Na-K-2Cl cotransporter (target of loop diuretics). The distal convoluted tubule (DCT) reabsorbs NaCl via the Na-Cl cotransporter (target of thiazides). The collecting duct is regulated by aldosterone (ENaC, Na reabsorption/K secretion) and ADH/vasopressin (aquaporin-2 insertion for water reabsorption).
Acid-Base Balance
The kidneys maintain acid-base homeostasis by: (1) reabsorbing filtered HCO3- (primarily in the PCT via carbonic anhydrase), (2) generating new HCO3- through ammonium (NH4+) excretion, and (3) excreting titratable acid. Renal tubular acidosis (RTA) is relevant to urology because Type I (distal) RTA causes alkaline urine → calcium phosphate stones, while Type II (proximal) RTA causes bicarbonate wasting. Uric acid stones form in persistently acidic urine (pH < 5.5).
Urologic Electrolyte Considerations
Post-TURP syndrome (TUR syndrome) occurs from absorption of hypotonic irrigation fluid (glycine or sorbitol) during monopolar TURP — causing dilutional hyponatremia, hypo-osmolality, volume overload, and glycine toxicity (visual disturbances, encephalopathy). Symptoms include confusion, nausea, bradycardia, and seizures when Na+ falls below 120 mEq/L. Treatment: stop the procedure, administer IV hypertonic saline (3% NaCl) carefully, and give loop diuretics. Bipolar TURP uses normal saline irrigation and has essentially eliminated this complication.
Hyperchloremic metabolic acidosis is the most common metabolic complication of urinary diversion using bowel segments. Ileal and colonic segments reabsorb urinary chloride (via Cl-/HCO3- exchange) and ammonium, leading to non-anion-gap metabolic acidosis. Treatment: oral sodium bicarbonate supplementation. Jejunal segments cause hyponatremic, hyperkalemic, metabolic acidosis (least commonly used for this reason).
Metabolic Complications by Bowel Segment in Urinary Diversion
| Bowel Segment | Metabolic Complication | Mechanism |
|---|---|---|
| Ileum / Colon | Hyperchloremic metabolic acidosis (non-anion gap) | Cl-/HCO3- exchange across bowel mucosa + NH4+ reabsorption |
| Jejunum | Hyponatremic, hyperkalemic metabolic acidosis | Na+/Cl- secretion with K+/H+ absorption; rarely used |
| Stomach | Hypochloremic metabolic alkalosis + hematuria-dysuria syndrome | HCl secretion into urine; rarely used except in patients with renal insufficiency |
Additional long-term complications of bowel-based urinary diversion: vitamin B12 deficiency (if > 60 cm of terminal ileum resected — monitor levels annually after 3-5 years), fat-soluble vitamin malabsorption (ileal resection impairs bile salt reabsorption), diarrhea (bile salt malabsorption), urinary tract stones (hyperoxaluria from fat malabsorption + mucus nidus), recurrent UTIs (mucus-producing bowel epithelium), and secondary malignancy at the bowel-urine interface (adenocarcinoma — rare but reported, typically > 10 years after diversion; recommend annual endoscopic surveillance after 10 years for continent diversions).
04 The Urologic Exam & Assessment
History & Physical Examination
The urologic history should address: voiding symptoms (LUTS — frequency, urgency, nocturia, hesitancy, weak stream, straining, incomplete emptying, terminal dribbling), storage symptoms (urgency, incontinence, frequency), hematuria (gross vs microscopic, timing — initial/terminal/total, painful vs painless), pain (flank, suprapubic, perineal, scrotal, penile), sexual function (erectile dysfunction, ejaculatory disorders), and fertility. The digital rectal exam (DRE) assesses prostate size, consistency (normal = firm, rubbery), nodularity, and tenderness. A hard, irregular, asymmetric prostate suggests malignancy. Rectal tone is assessed to evaluate sphincter function.
Urinalysis & Urine Studies
Urinalysis is the most fundamental urologic test. Key findings: hematuria (> 3 RBCs/HPF — requires evaluation for malignancy in adults ≥ 35 years per AUA guidelines), pyuria (> 5 WBCs/HPF — suggests infection), bacteriuria, proteinuria, crystals (calcium oxalate = envelope-shaped, uric acid = rhomboid/rosette, struvite = coffin-lid, cystine = hexagonal). Urine culture is indicated for suspected UTI, with ≥ 105 CFU/mL considered significant.
Urine cytology has high specificity but low sensitivity for urothelial carcinoma, particularly high-grade tumors. It is useful for surveillance of bladder cancer and CIS. 24-hour urine collection is the cornerstone of metabolic stone evaluation — measures volume, calcium, oxalate, citrate, uric acid, sodium, pH, and supersaturation indices.
PSA — Prostate-Specific Antigen
PSA is a serine protease produced by prostatic epithelium. Normal value is age-dependent but generally < 4.0 ng/mL. PSA is organ-specific but NOT cancer-specific — elevated in BPH, prostatitis, instrumentation, ejaculation, and cancer. PSA density = PSA / prostate volume (suspicious if > 0.15). PSA velocity = rate of change over time (suspicious if > 0.75 ng/mL/year). Free-to-total PSA ratio: lower percentage free PSA (< 10%) is more suspicious for cancer. PSA values 4-10 ng/mL represent a "diagnostic gray zone" with ~25% cancer detection rate on biopsy.
Uroflowmetry & Post-Void Residual
Uroflowmetry measures the rate of urine flow. The key parameter is Qmax (maximum flow rate): normal > 15 mL/s; values < 10 mL/s suggest obstruction. A voided volume of ≥ 150 mL is required for a reliable study. The flow pattern helps distinguish obstruction (prolonged, plateau-shaped) from detrusor weakness (low, extended curve). Post-void residual (PVR) is measured by catheterization or ultrasound immediately after voiding. PVR > 200 mL is generally significant; PVR > 300 mL suggests significant bladder dysfunction.
AUA Symptom Score (IPSS)
The International Prostate Symptom Score (IPSS) is a validated 7-question self-assessment tool scoring voiding symptoms from 0-35: mild (0-7), moderate (8-19), severe (20-35). An additional quality-of-life (QoL) question is scored 0-6. The IPSS is used for baseline assessment and monitoring treatment response in BPH. It is not diagnostic of any specific condition.
Hematuria Evaluation Algorithm
The AUA/SUFU guideline for microhematuria (updated 2020) stratifies patients by risk: Low risk: age < 40 (women) or < 25 (men), 3-10 RBC/HPF, no risk factors, never smoker → repeat UA in 6 months. Intermediate risk: age 40-59 (women) or 25-59 (men), 11-25 RBC/HPF, or 3-25 RBC/HPF with risk factors (smoking history, chemical exposure) → cystoscopy + renal US. High risk: age ≥ 60, > 25 RBC/HPF, or any history of gross hematuria, or prior urologic cancer → cystoscopy + CT urogram. All patients with gross hematuria require full urologic evaluation (cystoscopy + CT urogram) regardless of age.
05 Prostate Cancer
Epidemiology & Risk Factors
Prostate cancer is the most commonly diagnosed non-cutaneous malignancy in men and the second leading cause of cancer death in American men. Risk factors: age (rare before 50; median age at diagnosis ~66), race (African American men have ~60% higher incidence and 2-3× higher mortality), family history (first-degree relative → 2× risk; BRCA2 carriers have 3-5× risk), and diet (high animal fat).
PSA Screening
Current AUA/NCCN guidelines recommend shared decision-making for PSA screening starting at age 55-69 (or age 40-45 for high-risk men: African American, BRCA2, strong family history). Screening is not recommended for men with < 10-year life expectancy. A baseline PSA at age 40-45 can help stratify future risk: PSA > 0.7 ng/mL at age 40 or > 0.9 at age 50 confers significantly higher lifetime risk.
Gleason Grading & ISUP Grade Groups
The Gleason score is the sum of the two most prevalent architectural patterns (each graded 1-5). The primary pattern (most prevalent) is listed first. The ISUP Grade Group system (2014) simplifies reporting:
| ISUP Grade Group | Gleason Score | Prognosis |
|---|---|---|
| 1 | 3+3 = 6 | Favorable — candidate for active surveillance |
| 2 | 3+4 = 7 | Favorable intermediate |
| 3 | 4+3 = 7 | Unfavorable intermediate |
| 4 | 4+4 = 8, 3+5, 5+3 | High risk |
| 5 | 4+5, 5+4, 5+5 = 9-10 | Very high risk |
TNM Staging (Prostate Cancer, AJCC 8th Edition)
| Stage | Description |
|---|---|
| T1a | Incidental, ≤ 5% of resected tissue |
| T1b | Incidental, > 5% of resected tissue |
| T1c | Identified by needle biopsy (PSA elevation) |
| T2a | Involves ≤ one half of one lobe |
| T2b | Involves > one half of one lobe |
| T2c | Involves both lobes |
| T3a | Extracapsular extension (unilateral or bilateral) |
| T3b | Seminal vesicle invasion |
| T4 | Invasion of adjacent structures (bladder neck, external sphincter, rectum, levator muscles, pelvic wall) |
| N1 | Regional lymph node metastasis |
| M1a | Non-regional lymph node metastasis |
| M1b | Bone metastasis |
| M1c | Other distant metastasis |
Risk Stratification (NCCN/D'Amico)
Very low risk: T1c, Gleason 6, PSA < 10, fewer than 3 positive cores, ≤ 50% cancer in any core, PSA density < 0.15. Low risk: T1-T2a, Gleason 6, PSA < 10. Intermediate risk: T2b-T2c, or Gleason 7, or PSA 10-20. High risk: T3a, or Gleason 8-10, or PSA > 20. Very high risk: T3b-T4, primary Gleason pattern 5, or > 4 cores with Gleason 8-10.
Active Surveillance
Active surveillance (AS) is the preferred strategy for very-low-risk and low-risk prostate cancer in men with ≥ 10-year life expectancy. Protocol includes: PSA every 3-6 months, DRE every 12 months, confirmatory MRI-targeted biopsy within 6-12 months, then repeat biopsy every 1-2 years. Reclassification triggers intervention (Gleason upgrade to 4+3 or higher, increased tumor volume). The landmark ProtecT trial showed no difference in prostate-cancer-specific mortality at 15 years between AS, radical prostatectomy, and radiotherapy for localized disease.
Radical Prostatectomy
Radical prostatectomy (RP) involves removal of the entire prostate, seminal vesicles, and often pelvic lymph nodes. Approaches: robot-assisted laparoscopic (RARP) — now the most common approach in the US, offering improved visualization and nerve-sparing precision; open retropubic (RRP); and perineal (rarely used). Key steps include apical dissection, dorsal venous complex ligation, nerve-sparing (when oncologically appropriate), vesicourethral anastomosis, and lymph node dissection. Major complications: erectile dysfunction (30-70% depending on nerve-sparing status and age) and urinary incontinence (5-20% at 1 year). Positive surgical margins occur in 10-30% of cases.
Radiation Therapy
External beam radiation therapy (EBRT) — intensity-modulated radiation therapy (IMRT) or stereotactic body radiation therapy (SBRT) — is an alternative to surgery for localized disease. Brachytherapy (permanent seed implantation, typically I-125) is used for low-risk disease. For intermediate and high-risk disease, radiation is combined with androgen deprivation therapy (ADT) — 6 months for intermediate risk, 18-36 months for high risk (per DART/STAMPEDE data). Radiation side effects: proctitis, cystitis, erectile dysfunction (delayed onset), and secondary malignancies.
Androgen Deprivation Therapy (ADT)
ADT is the backbone of treatment for advanced and metastatic prostate cancer. Methods: (1) GnRH agonists (leuprolide, goserelin) — cause initial testosterone flare then suppression (co-administer anti-androgen for first 2-4 weeks to prevent flare); (2) GnRH antagonists (degarelix, relugolix) — immediate suppression without flare; (3) bilateral orchiectomy — surgical castration; (4) anti-androgens (bicalutamide, enzalutamide, apalutamide, darolutamide) — block androgen receptor. Target testosterone level: < 50 ng/dL (castrate). ADT side effects: hot flashes, sexual dysfunction, osteoporosis, metabolic syndrome, cardiovascular risk, cognitive changes, and anemia.
Castration-Resistant Prostate Cancer (CRPC)
CRPC is defined as disease progression despite castrate testosterone levels (< 50 ng/dL). Treatment options include: novel hormonal agents (enzalutamide, abiraterone + prednisone, apalutamide, darolutamide), chemotherapy (docetaxel, cabazitaxel), radiopharmaceuticals (radium-223 for bone metastases, Lu-177-PSMA-617 for PSMA-positive disease), PARP inhibitors (olaparib, rucaparib for BRCA/HRR-mutated tumors), immunotherapy (sipuleucel-T), and bone-protective agents (denosumab, zoledronic acid).
Salvage Therapy & Biochemical Recurrence
Biochemical recurrence (BCR) after radical prostatectomy is defined as a detectable and rising PSA ≥ 0.2 ng/mL (confirmed on two measurements). After radiation therapy, BCR is defined as a PSA nadir + 2.0 ng/mL (Phoenix definition). For BCR after RP: salvage radiation therapy (SRT) is most effective when PSA is < 0.5 ng/mL (ideally < 0.2 ng/mL) and when Gleason score, margin status, and PSA kinetics suggest local recurrence. GETUG-AFU 16 and RTOG 9601 trials demonstrated benefit of concurrent ADT with salvage radiation. For BCR after radiation: salvage prostatectomy, cryotherapy, HIFU, or ADT depending on risk profile. PSMA PET-CT has revolutionized BCR evaluation — it detects recurrence at PSA levels as low as 0.2-0.5 ng/mL with high sensitivity, enabling more precise targeting of salvage therapy.
Prostate Biopsy Techniques
The standard approach has evolved from TRUS-guided systematic biopsy (12-core template) to MRI-TRUS fusion targeted biopsy. In fusion biopsy, suspicious lesions identified on mpMRI (PI-RADS ≥ 3) are co-registered with real-time TRUS, and targeted cores are obtained from each lesion in addition to systematic cores. The transperineal approach is increasingly preferred over transrectal due to lower infection risk (near-zero sepsis rate without antibiotics vs ~1-4% with transrectal approach despite prophylaxis). MRI-targeted biopsy improves detection of clinically significant cancer (Gleason ≥ 7) and reduces detection of clinically insignificant cancer (Gleason 6).
06 Bladder Cancer
Epidemiology & Risk Factors
Bladder cancer is the 6th most common cancer overall and the most common malignancy of the urinary tract. Approximately 90% of bladder cancers in the Western world are urothelial carcinoma (transitional cell carcinoma, TCC). Other histologies: squamous cell carcinoma (associated with chronic inflammation, Schistosoma haematobium), adenocarcinoma (urachal remnant), and small cell carcinoma. Risk factors: cigarette smoking (strongest risk factor, 3-4× risk, accounts for ~50% of cases), occupational exposure to aromatic amines (2-naphthylamine, benzidine — rubber, dye, chemical industries), cyclophosphamide, pelvic radiation, and chronic irritation (catheters, stones, Schistosoma).
Presentation & Diagnosis
The hallmark presentation is painless gross hematuria — present in ~80-90% of patients. Irritative voiding symptoms (frequency, urgency, dysuria) may suggest carcinoma in situ (CIS). Diagnosis requires cystoscopy with biopsy/resection. Urine cytology has high specificity for high-grade tumors and CIS but low sensitivity for low-grade tumors. CT urogram evaluates the upper tracts for synchronous disease.
TNM Staging (Bladder Cancer)
| Stage | Description | Category |
|---|---|---|
| Ta | Noninvasive papillary carcinoma (confined to urothelium) | Non-muscle-invasive (NMIBC) |
| Tis (CIS) | Carcinoma in situ — flat, high-grade, confined to urothelium | |
| T1 | Invades lamina propria (subepithelial connective tissue) | |
| T2a | Invades superficial muscle (inner half of detrusor) | Muscle-invasive (MIBC) |
| T2b | Invades deep muscle (outer half of detrusor) | |
| T3 | Invades perivesical tissue (T3a microscopically, T3b macroscopically) | |
| T4 | Invades adjacent organs (T4a: prostate stroma/uterus/vagina; T4b: pelvic wall/abdominal wall) |
TURBT — Transurethral Resection of Bladder Tumor
TURBT is both diagnostic (staging) and therapeutic (for NMIBC). Critical technique: resection must include detrusor muscle in the specimen to accurately stage — absence of muscle requires re-TURBT. A re-TURBT is performed 2-6 weeks after initial resection for high-grade T1 tumors and when no muscle was in the specimen. Approximately 40-50% of re-TURBTs show residual disease, and up to 25% are upstaged.
Intravesical Therapy for NMIBC
BCG (Bacillus Calmette-Guérin) is the gold standard intravesical immunotherapy for intermediate-risk and high-risk NMIBC. Induction: 6 weekly instillations. Maintenance (SWOG protocol): 3 weekly instillations at 3, 6, 12, 18, 24, 30, and 36 months — proven to reduce recurrence and progression. BCG is the only intravesical agent shown to reduce progression to muscle-invasive disease. Contraindications: active UTI, traumatic catheterization, immunosuppression. Complications: BCG cystitis (common), BCG sepsis (rare but life-threatening — treat with isoniazid + rifampin + fluoroquinolone). Intravesical chemotherapy (mitomycin C, gemcitabine) is used for low-risk NMIBC or BCG failures.
Radical Cystectomy & Urinary Diversion
Radical cystectomy is the gold standard for muscle-invasive bladder cancer (T2-T4a, N0-Nx, M0) and BCG-unresponsive high-risk NMIBC. In men: removal of bladder, prostate, seminal vesicles, and pelvic lymph nodes. In women: removal of bladder, uterus, ovaries, anterior vaginal wall, and pelvic lymph nodes. Pelvic lymph node dissection (extended template to the aortic bifurcation) is critical — ~25% of patients are upstaged by LND.
Urinary diversion options: (1) Ileal conduit (Bricker) — the most common diversion; a segment of ileum serves as a urine conduit to an abdominal stoma (requires external appliance). (2) Orthotopic neobladder (Studer, Hautmann) — detubularized ileal segment fashioned into a spherical reservoir anastomosed to the urethra; allows volitional voiding (no stoma). Requires intact external sphincter and negative urethral margin. Patients void by Valsalva/abdominal straining. (3) Continent cutaneous diversion (Indiana pouch) — catheterizable stoma, no external appliance.
Neoadjuvant cisplatin-based chemotherapy (MVAC or dose-dense MVAC or gemcitabine/cisplatin) provides a 5-8% absolute overall survival benefit and should be offered to eligible patients before cystectomy. Adjuvant immunotherapy (nivolumab) is now standard for patients at high risk of recurrence post-cystectomy. Trimodal therapy (maximal TURBT + concurrent chemoradiation) is a bladder-preserving alternative for select patients.
Immunotherapy for Metastatic Urothelial Carcinoma
Immune checkpoint inhibitors have transformed treatment of advanced urothelial carcinoma. First-line: For cisplatin-eligible patients, pembrolizumab + enfortumab vedotin (EV-302/KEYNOTE-A39 trial) is the new standard; cisplatin-based chemotherapy followed by avelumab maintenance (JAVELIN Bladder 100) is an established option. For cisplatin-ineligible patients: pembrolizumab or atezolizumab (if PD-L1 positive) or carboplatin-based chemotherapy. Second-line: Enfortumab vedotin (antibody-drug conjugate targeting Nectin-4), erdafitinib (FGFR inhibitor for FGFR2/3-altered tumors), sacituzumab govitecan (Trop-2 ADC). The urothelial carcinoma treatment landscape is rapidly evolving with novel ADCs and targeted therapies.
07 Renal Cell Carcinoma
Epidemiology & Subtypes
Renal cell carcinoma (RCC) accounts for ~90% of renal malignancies and ~3% of all adult cancers. Peak incidence age 60-70; M:F ratio 2:1. Risk factors: smoking, obesity, hypertension, acquired cystic kidney disease (dialysis patients), von Hippel-Lindau (VHL) syndrome, and hereditary papillary RCC. Histologic subtypes:
| Subtype | Frequency | Key Features |
|---|---|---|
| Clear cell | 70-80% | VHL gene mutation (chromosome 3p); highly vascular; most responsive to targeted therapy and immunotherapy |
| Papillary (Type 1 & 2) | 10-15% | Type 1: MET mutation, better prognosis; Type 2: worse prognosis, associated with hereditary leiomyomatosis and RCC (HLRCC) |
| Chromophobe | 5% | Best prognosis of RCC subtypes; associated with Birt-Hogg-Dubé syndrome |
| Collecting duct (Bellini) | < 1% | Aggressive, poor prognosis |
| Medullary | Rare | Almost exclusively in patients with sickle cell trait; highly aggressive |
Presentation
The classic triad of flank pain, hematuria, and palpable abdominal mass is present in only ~10% of cases and usually indicates advanced disease. Most RCCs today are discovered incidentally on imaging. Paraneoplastic syndromes occur in ~20%: hypercalcemia (PTHrP), erythrocytosis (EPO), hypertension (renin), hepatic dysfunction without liver metastases (Stauffer syndrome — resolves after nephrectomy), and non-metastatic hepatic dysfunction.
Bosniak Classification for Renal Cysts
| Category | Features | Malignancy Risk | Management |
|---|---|---|---|
| I | Simple cyst: thin wall, no septa/calcification/enhancement | < 1% | No follow-up |
| II | Few thin septa, fine calcification, < 3 cm hyperdense cyst, no enhancement | < 5% | No follow-up |
| IIF | Multiple thin septa, minimal smooth thickening, thick calcification, ≥ 3 cm hyperdense, no enhancement | 5-15% | Follow-up imaging |
| III | Thick/irregular septa or wall, measurable enhancement | 40-60% | Surgery or active surveillance |
| IV | Enhancing soft tissue component | > 80% | Surgery |
Surgical Management
For localized RCC (T1-T2), surgery is the primary treatment. Partial nephrectomy (nephron-sparing surgery) is the gold standard for T1a tumors (≤ 4 cm) and preferred for T1b (≤ 7 cm) when technically feasible — equivalent oncologic outcomes with better renal function preservation. Approaches: robotic-assisted (most common), laparoscopic, or open. Radical nephrectomy is indicated for larger tumors (T2, > 7 cm), centrally located tumors not amenable to partial nephrectomy, and locally advanced disease. Includes removal of Gerota fascia and kidney en bloc. Ipsilateral adrenalectomy is only indicated if imaging suggests adrenal involvement.
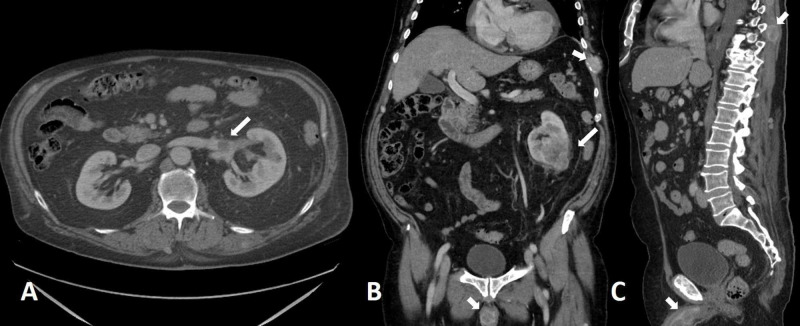
For tumors with IVC tumor thrombus (occurs in ~4-10% of RCC), surgical thrombectomy is performed with radical nephrectomy. Level of thrombus dictates approach: Level I (renal vein) — simple vein control; Level II (infrahepatic IVC) — IVC clamping; Level III (intrahepatic/retrohepatic IVC) — may require liver mobilization; Level IV (supradiaphragmatic/intra-atrial) — requires cardiopulmonary bypass with cardiothoracic surgery. Despite the IVC thrombus, if there is no distant metastatic disease, 5-year survival is 40-65% after complete resection.
IMDC Risk Stratification (Metastatic RCC)
The International Metastatic RCC Database Consortium (IMDC/Heng criteria) stratifies mRCC prognosis using 6 risk factors: (1) KPS < 80%, (2) time from diagnosis to treatment < 1 year, (3) hemoglobin < lower limit of normal, (4) corrected calcium > upper limit of normal, (5) neutrophils > upper limit of normal, (6) platelets > upper limit of normal. Favorable risk: 0 factors (median OS ~43 months). Intermediate risk: 1-2 factors (median OS ~23 months). Poor risk: 3-6 factors (median OS ~8 months). IMDC risk group guides treatment selection in mRCC.
Systemic Therapy for Advanced/Metastatic RCC
First-line treatment for clear cell mRCC has shifted to immune checkpoint inhibitor (ICI) combinations: ipilimumab + nivolumab (IMDC intermediate/poor risk — CheckMate 214), pembrolizumab + axitinib (KEYNOTE-426), nivolumab + cabozantinib (CheckMate 9ER), or pembrolizumab + lenvatinib (KEYNOTE-581/CLEAR). For favorable-risk patients, ICI combinations or single-agent TKI (cabozantinib, sunitinib, pazopanib) are options. Cytoreductive nephrectomy is selectively offered in the immunotherapy era (no longer standard for all patients — CARMENA and SURTIME trials).
Small Renal Masses & Active Surveillance
Small renal masses (SRM) are defined as enhancing renal tumors ≤ 4 cm. Approximately 20-30% of SRMs are benign (oncocytoma, angiomyolipoma). Active surveillance is appropriate for patients with significant comorbidities, limited life expectancy, or high surgical risk. Growth rate is typically slow (~0.3 cm/year). Renal mass biopsy can help guide management — high diagnostic accuracy for distinguishing benign from malignant lesions and determining histologic subtype. Thermal ablation (cryoablation, radiofrequency ablation, microwave ablation) is an option for T1a tumors in patients who are poor surgical candidates — lower cancer-specific survival than partial nephrectomy but acceptable for appropriately selected patients.
08 Testicular Cancer
Epidemiology & Risk Factors
Testicular cancer is the most common solid malignancy in men aged 15-35. Incidence has been increasing over recent decades. Risk factors: cryptorchidism (3-8× increased risk; orchiopexy before puberty may reduce risk but does not eliminate it), prior testicular cancer (contralateral risk ~2-5%), family history (brother with testicular cancer → 8-10× risk), disorders of sex development (gonadal dysgenesis), and infertility/testicular atrophy.
Classification — Germ Cell Tumors (95%)
| Category | Subtypes | Key Features |
|---|---|---|
| Seminoma (~55%) | Classical, spermatocytic | Peak age 30-40; homogeneous on US; exquisitely radiosensitive; AFP is NEVER elevated (if AFP elevated, treat as NSGCT regardless of histology) |
| Nonseminomatous GCT (NSGCT, ~45%) | Embryonal carcinoma, yolk sac tumor, choriocarcinoma, teratoma, mixed | Peak age 20-30; more aggressive; choriocarcinoma produces hCG; yolk sac tumor produces AFP; teratoma is chemoresistant — must be resected |
Tumor Markers
AFP (α-fetoprotein): Elevated in yolk sac tumors and some embryonal carcinomas. Half-life: 5-7 days. NEVER elevated in pure seminoma (if elevated, treat as NSGCT). hCG (β-human chorionic gonadotropin): Elevated in choriocarcinoma (very high levels) and ~10-20% of seminomas (modest elevation). Half-life: 24-36 hours. LDH: Nonspecific marker of tumor burden/cell turnover. Marker decline after orchiectomy follows predictable half-lives — failure to normalize or rising markers indicates residual/metastatic disease.
Staging & IGCCCG Risk Classification
Stage I: confined to testis (no lymph node or distant metastasis). Stage II: retroperitoneal lymph node involvement (IIA: ≤ 2 cm; IIB: 2-5 cm; IIC: > 5 cm). Stage III: distant metastatic disease (IIIA: lung/non-regional LN; IIIB-C: based on marker levels or non-pulmonary visceral metastasis). The International Germ Cell Cancer Collaborative Group (IGCCCG) classifies metastatic GCT into good, intermediate, and poor prognosis groups based on site of primary, presence of non-pulmonary visceral metastases, and marker levels — 5-year survival: good 90-92%, intermediate 72-80%, poor 48-54%.
Management
Radical inguinal orchiectomy is the initial treatment for all suspected testicular tumors. An inguinal approach is mandatory (NOT transscrotal) — a transscrotal biopsy or orchiectomy violates the scrotal skin, alters lymphatic drainage (scrotal skin drains to inguinal nodes, testis drains to retroperitoneal nodes), and may necessitate hemiscrotectomy. A testicular prosthesis can be placed at the time of orchiectomy.
Stage I seminoma: Options include surveillance (preferred for most — recurrence rate ~15-20%, nearly all curable at relapse), single-agent carboplatin (1-2 cycles), or adjuvant radiation (less used now due to long-term toxicity). Stage I NSGCT: Options include surveillance (if markers normalize and risk factors absent), retroperitoneal lymph node dissection (RPLND), or adjuvant BEP × 1 cycle.
BEP chemotherapy (bleomycin, etoposide, cisplatin) is the standard regimen for metastatic GCT: 3 cycles for good-prognosis, 4 cycles for intermediate/poor-prognosis. Cure rates for metastatic testicular cancer are among the highest of any solid tumor (>90% for good-prognosis disease). Post-chemotherapy RPLND is indicated for residual retroperitoneal masses > 1 cm in NSGCT (may contain residual viable tumor, teratoma, or necrosis). For seminoma, residual masses are observed with PET/CT — post-chemotherapy surgery only if PET-positive and ≥ 3 cm.
Late Effects of Testicular Cancer Treatment
Given the young age of most patients and high cure rates, long-term survivorship is critical. Cardiovascular disease: Cisplatin-based chemotherapy increases long-term cardiovascular risk (metabolic syndrome, coronary artery disease) — 2-3× risk of cardiovascular events; ongoing surveillance recommended. Secondary malignancies: Radiation and chemotherapy increase risk of solid tumors (GI, lung, thyroid, melanoma) and leukemia; risk rises with time. Fertility: Chemotherapy impairs spermatogenesis — recovery occurs in ~80% by 5 years for BEP; sperm banking before treatment is essential. Hypogonadism: ~10-20% of long-term survivors develop testosterone deficiency. Nephrotoxicity, ototoxicity, neurotoxicity: Cisplatin-related effects may persist or worsen over time (tinnitus, hearing loss, peripheral neuropathy). Retrograde ejaculation: After RPLND (even nerve-sparing); rates ~5-10% with nerve-sparing technique vs ~70-100% without. Long-term follow-up protocols should be individualized based on tumor stage, histology, and treatment received.
09 Upper Tract Urothelial Carcinoma
Overview
Upper tract urothelial carcinoma (UTUC) accounts for 5-10% of urothelial cancers. It involves the renal pelvis (~60%) or ureter (~40%). Risk factors mirror those of bladder cancer (smoking, aromatic amines) with additional associations: aristolochic acid (Balkan endemic nephropathy, Chinese herbal nephropathy) and Lynch syndrome (MSH2 mutations — lifetime UTUC risk ~8%). UTUC has a higher rate of invasive disease at presentation (~60% are invasive at diagnosis vs ~25% for bladder cancer).
Diagnosis
Presentation: hematuria (gross or microscopic) is the most common symptom (~70-80%); flank pain from obstruction (~20-30%). Diagnosis: CT urogram (sensitivity ~96% for detecting UTUC — filling defect or urothelial thickening in the excretory phase), urine cytology (higher sensitivity for high-grade UTUC), and diagnostic ureteroscopy with biopsy and barbotage. Selective upper tract washing cytology improves diagnostic accuracy. All patients with UTUC should undergo cystoscopy to evaluate for concurrent bladder cancer (~17% have synchronous bladder lesions).
Management
Radical nephroureterectomy (RNU) with excision of a bladder cuff is the gold standard for high-risk UTUC (high grade, invasive, large, multifocal). The bladder cuff must be removed because of high rates (~30-50%) of subsequent bladder recurrence if the distal ureter/intramural segment is left behind. Kidney-sparing endoscopic management (ureteroscopic laser ablation) is appropriate for low-risk UTUC (low grade, small, unifocal) in patients with a solitary kidney, bilateral disease, or significant CKD. Adjuvant instillation of mitomycin C within the bladder after RNU reduces intravesical recurrence (ODMIT-C trial). Neoadjuvant cisplatin-based chemotherapy is increasingly considered given the loss of one kidney after RNU.
10 Penile Cancer
Overview
Penile cancer is rare in developed countries (< 1% of male cancers) but more common in developing nations. The vast majority (~95%) are squamous cell carcinoma. Risk factors: lack of circumcision (phimosis and chronic balanitis), poor hygiene, HPV infection (types 16 and 18 — present in ~50% of cases), smoking, and immunosuppression. Precursor lesions include erythroplasia of Queyrat (CIS of the glans), Bowen disease (CIS of the shaft), and bowenoid papulosis (HPV-associated, multifocal).
Staging & Management
Treatment of the primary tumor depends on stage: Tis/Ta — topical therapy (5-FU, imiquimod) or laser ablation; T1 (low grade, no LVI) — wide local excision or glansectomy; T1 (high grade or LVI) through T2 — partial penectomy (2 cm margin historically, though recent evidence supports narrower margins for lower-stage tumors); T3-T4 — total penectomy with perineal urethrostomy. A minimum penile stump length of ~2 cm is desired for standing voiding after partial penectomy.
Inguinal lymph node management is critical — the most important prognostic factor is inguinal lymph node status. For clinically node-negative patients with intermediate/high-risk primary tumors (T1 high grade or higher): dynamic sentinel node biopsy (DSNB) or modified inguinal lymphadenectomy. For clinically positive nodes: fine-needle aspiration followed by inguinal lymphadenectomy if positive. Pelvic lymph node dissection is indicated if ≥ 2 positive inguinal nodes or extranodal extension.
Penile Cancer TNM Staging
| Stage | Description |
|---|---|
| Tis | Carcinoma in situ (penile intraepithelial neoplasia) |
| Ta | Noninvasive verrucous carcinoma |
| T1a | Invades subepithelial tissue, no LVI, grade 1-2 |
| T1b | Invades subepithelial tissue, with LVI or grade 3 |
| T2 | Invades corpus spongiosum or corpora cavernosa |
| T3 | Invades urethra |
| T4 | Invades adjacent structures (prostate, pubic bone) |
11 Nephrolithiasis — Evaluation & Medical Management
Epidemiology & Stone Types
Nephrolithiasis affects ~10% of the population, with a recurrence rate of ~50% within 5-10 years. Peak incidence age 30-60; M:F ratio ~2:1 (narrowing with rising female obesity rates). Stone composition:
| Stone Type | Frequency | Crystal Appearance | Key Features |
|---|---|---|---|
| Calcium oxalate | 70-80% | Envelope-shaped (monohydrate: dumbbell) | Most common; associated with hypercalciuria, hyperoxaluria, hypocitraturia; radiopaque on X-ray |
| Calcium phosphate | 10-15% | Amorphous | Associated with RTA Type I (alkaline urine pH > 7.0), hyperparathyroidism; radiopaque |
| Uric acid | 5-10% | Rhomboid/rosette | RADIOLUCENT on X-ray (visible on CT); formed in acidic urine (pH < 5.5); associated with gout, metabolic syndrome, myeloproliferative disorders; can be dissolved with urinary alkalinization |
| Struvite (MAP) | 5-10% | Coffin-lid | Magnesium ammonium phosphate; caused by urease-producing bacteria (Proteus, Klebsiella, Pseudomonas); form staghorn calculi; alkaline urine pH > 7.2 |
| Cystine | 1-3% | Hexagonal | Autosomal recessive cystinuria; recurrent stones starting in childhood/adolescence; semi-opaque on X-ray |
Acute Renal Colic — Presentation & Workup
Presentation: sudden-onset, severe colicky flank pain radiating to the groin/ipsilateral testicle or labium, often with nausea/vomiting, hematuria (present in ~85%), and restlessness (cannot find a comfortable position). Pain location correlates with stone position: upper ureteral stones → flank/costovertebral angle pain; mid-ureteral stones (crossing iliac vessels) → anterior abdominal pain mimicking appendicitis or diverticulitis; distal ureteral stones (near UVJ) → suprapubic pain, urinary urgency/frequency, pain radiating to the ipsilateral testicle/labium. The gold standard imaging is non-contrast CT abdomen/pelvis — sensitivity ~97%, specificity ~96% for ureteral stones. Also identifies stone size, location, number, and alternative diagnoses. Ultrasound is the initial study in pregnant patients and children. KUB X-ray is useful for follow-up of known radiopaque stones.
Indications for Urgent Intervention in Stone Disease
While most ureteral stones can be managed expectantly, certain situations require urgent decompression (ureteral stent or nephrostomy tube): (1) Emergency infected obstructed kidney (fever + obstructing stone = pyonephrosis until proven otherwise — can progress to urosepsis rapidly), (2) impending renal failure (bilateral obstruction, solitary kidney, pre-existing CKD), (3) intractable pain/vomiting despite adequate analgesia, and (4) high-grade obstruction with declining renal function on serial labs.
Medical Expulsive Therapy (MET)
For distal ureteral stones ≤ 10 mm, spontaneous passage is possible (likelihood decreases with size: < 5 mm ~68%, 5-10 mm ~47%). Tamsulosin 0.4 mg daily (alpha-blocker) is the most commonly used MET agent — relaxes ureteral smooth muscle, facilitating stone passage, particularly for distal ureteral stones 5-10 mm. Nifedipine (calcium channel blocker) is a second-line option. Pain management: NSAIDs (ketorolac) are first-line (also reduce ureteral edema and spasm), with opioids for breakthrough pain. MET trial duration: 4-6 weeks with close follow-up.
Metabolic Evaluation
Indicated for recurrent stone formers, first-time formers with risk factors, and all children. Includes: 24-hour urine collection (× 2 for reliability) measuring volume, calcium, oxalate, citrate, uric acid, sodium, pH, magnesium, phosphorus, creatinine, and cystine (if suspected). Serum studies: calcium, PTH, uric acid, bicarbonate, BUN/creatinine. Stone analysis (infrared spectroscopy or X-ray diffraction) should be performed on every recovered stone.
Prevention Strategies
Universal: fluid intake to achieve > 2.5 L/day urine output (most impactful intervention); dietary sodium restriction (< 2,300 mg/day — reduces calcium excretion); moderate protein intake; avoid excessive oxalate-rich foods; maintain normal calcium intake (1,000-1,200 mg/day — dietary calcium restriction INCREASES stone risk by reducing intestinal oxalate binding). Specific: thiazide diuretics (hydrochlorothiazide, chlorthalidone, indapamide) for hypercalciuria; potassium citrate for hypocitraturia and uric acid stones (alkalinizes urine to pH 6.5-7.0); allopurinol for hyperuricosuric calcium stones and uric acid stones; tiopronin or D-penicillamine for cystine stones (chelate cystine).
12 Surgical Stone Management
Indications for Surgical Intervention
Surgery is indicated when: stone fails to pass with MET after 4-6 weeks, stone > 10 mm (unlikely to pass spontaneously), intractable pain, persistent obstruction with infection (Emergency — sepsis risk), bilateral obstruction, solitary kidney with obstruction, and patient preference. Choice of surgical modality depends on stone size, location, composition, anatomy, and patient factors.
Shock Wave Lithotripsy (SWL)
SWL uses focused extracorporeal shock waves to fragment stones. Best for: renal and proximal ureteral stones ≤ 20 mm (ideally ≤ 10-15 mm), lower pole stones ≤ 10 mm. Success rate: ~80-85% for stones < 10 mm, decreasing with size. Contraindications: pregnancy, uncorrected coagulopathy, aortic or renal artery aneurysm, distal obstruction. Limitations: less effective for hard stones (calcium oxalate monohydrate, cystine, brushite), large stones, lower pole stones > 10 mm (poor clearance due to gravity-dependent calyx), and obese patients (skin-to-stone distance > 10 cm). Complications: steinstrasse (stone street — column of fragments obstructing the ureter), subcapsular hematoma, and perinephric hematoma.
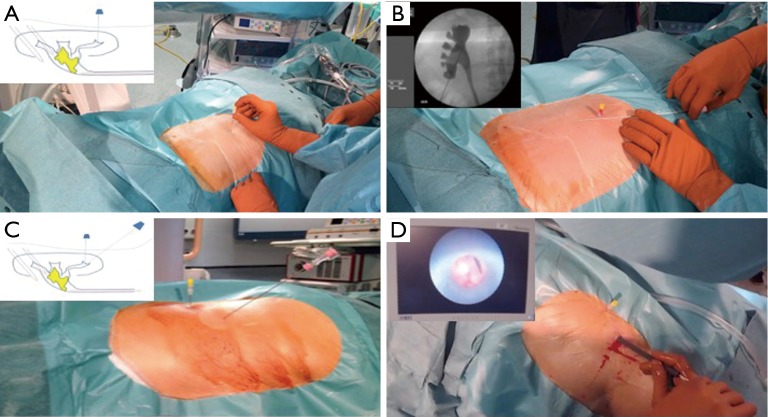
Ureteroscopy (URS)
Ureteroscopy uses a semirigid or flexible endoscope introduced retrograde through the urethra, bladder, and ureteral orifice. Stones are fragmented with a holmium:YAG laser (gold standard for intracorporeal lithotripsy) and fragments retrieved with baskets. URS is first-line for: mid and distal ureteral stones (any size), proximal ureteral stones ≤ 15-20 mm, renal stones ≤ 20 mm, and when SWL has failed. Stone-free rates: ~95% for distal ureteral stones, ~85-90% for proximal ureteral and renal stones ≤ 15 mm. A ureteral access sheath (UAS) facilitates multiple passes and reduces intrarenal pressure. A post-procedural double-J stent is often placed for 1-2 weeks. Complications: ureteral injury/perforation, stricture (long-term), UTI/sepsis, and stone migration.
Percutaneous Nephrolithotomy (PCNL)
PCNL is the procedure of choice for large renal stones (> 20 mm), staghorn calculi, lower pole stones > 10-15 mm, and stones resistant to SWL/URS. Technique: percutaneous renal access is obtained under fluoroscopic or ultrasound guidance (typically through a posterior calyx), the tract is dilated to 24-30 Fr, and a nephroscope is introduced. Stones are fragmented with ultrasonic, pneumatic, or laser lithotripsy and extracted. Stone-free rates: ~85-95% for large stones. Miniaturized PCNL (mini-PCNL, micro-PCNL, ultra-mini-PCNL) uses smaller tracts (11-20 Fr) with reduced morbidity. Complications: bleeding (most common significant complication — 2-5% require transfusion), pneumothorax (supracostal access above 12th rib), colonic injury, urosepsis, and AV fistula.
| Modality | Best Indication | Stone-Free Rate | Key Advantage |
|---|---|---|---|
| SWL | Renal/proximal ureteral ≤ 15 mm | 70-85% | Noninvasive, outpatient |
| URS | Any ureteral; renal ≤ 20 mm | 85-95% | High success, direct visualization |
| PCNL | Renal > 20 mm, staghorn | 85-95% | Best for large stone burden |
Staghorn Calculi
Staghorn calculi are branched stones that fill the renal pelvis and extend into at least two calyces. Most are composed of struvite (infection stones) or, less commonly, cystine or uric acid. Struvite staghorn calculi form in the setting of chronic infection with urease-producing organisms (Proteus mirabilis is the most common; also Klebsiella, Pseudomonas, Staphylococcus saprophyticus). Urease splits urea into ammonia and CO2, raising urine pH > 7.2 and promoting crystallization of magnesium ammonium phosphate (struvite) and calcium phosphate (apatite). Untreated staghorn calculi cause progressive renal damage, recurrent sepsis, and eventual loss of renal function. Treatment: PCNL (first-line) — may require staged procedures or multiple access tracts for complete clearance. SWL monotherapy has a low stone-free rate for large staghorn calculi (< 50%). Concurrent and prolonged antibiotic therapy is essential. Acetohydroxamic acid (urease inhibitor) may be used adjunctively to prevent recurrence.
13 Ureteral Obstruction & Stenting
Acute Ureteral Obstruction
Causes: nephrolithiasis (most common), malignant extrinsic compression (cervical, colorectal, ovarian cancer, retroperitoneal lymphadenopathy), retroperitoneal fibrosis (idiopathic/Ormond disease or drug-induced — methysergide, ergotamine), iatrogenic ureteral injury (pelvic surgery), UPJ obstruction, ureteral stricture, and blood clots. Bilateral obstruction or obstruction of a solitary kidney causes obstructive renal failure (post-renal AKI). Unilateral obstruction may be asymptomatic if the contralateral kidney is normal.
Pathophysiology of Obstruction
Acute unilateral obstruction produces a triphasic renal hemodynamic response: Phase 1 (0-1.5 hrs) — increased renal blood flow and ureteral pressure (prostaglandin-mediated vasodilation); Phase 2 (1.5-5 hrs) — decreased renal blood flow with continued elevated ureteral pressure; Phase 3 (> 5 hrs) — decreased renal blood flow AND decreased ureteral pressure. Prolonged obstruction causes progressive tubular damage, cortical thinning, and eventual loss of renal function. Recovery potential decreases with duration of obstruction: complete recovery is expected if obstruction is relieved within 1-2 weeks; after 6-8 weeks of complete obstruction, recovery is unlikely. Post-obstructive diuresis may occur after relief of bilateral obstruction or obstruction of a solitary kidney — characterized by massive polyuria (may exceed 500 mL/hr) requiring careful fluid and electrolyte monitoring. Replace urine output with 0.45% saline, monitoring electrolytes every 4-6 hours.
Ureteral Stents
The double-J (DJ) ureteral stent is the most commonly placed ureteral stent. It has coiled ends (J-shaped) to anchor in the renal pelvis proximally and bladder distally. Indications: relief of ureteral obstruction, post-URS (to prevent edema-related obstruction), after ureteral injury/repair, and pre-SWL for large stones (to prevent steinstrasse). Stents are typically 6 Fr in diameter and 22-28 cm in length (patient height-dependent). Stent-related symptoms (frequency, urgency, flank pain with voiding, hematuria) are common and can be managed with alpha-blockers (tamsulosin) and anticholinergics.
Percutaneous Nephrostomy
Percutaneous nephrostomy (PCN) provides external urinary drainage via a tube placed through the flank into the renal collecting system under ultrasound/fluoroscopic guidance. Preferred over retrograde stent placement in: infected obstructed system (pyonephrosis/sepsis — often technically easier and faster), patients too unstable for general anesthesia, failed retrograde stent placement, and severely ill patients. PCN can be converted to an antegrade ureteral stent once the patient stabilizes.
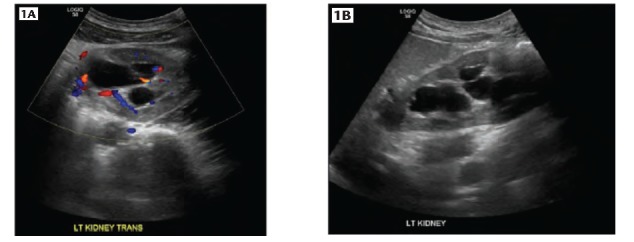
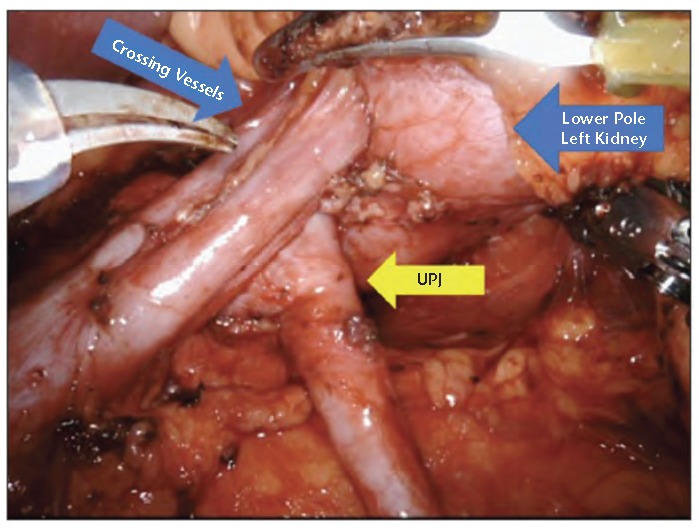
UPJ Obstruction
Ureteropelvic junction (UPJ) obstruction is the most common cause of hydronephrosis in children but also presents in adults. May be intrinsic (adynamic segment, high insertion of ureter) or extrinsic (crossing vessel — lower pole renal artery compressing the UPJ, present in ~30-50% of adult cases). Diagnosis: diuretic renography (MAG3 scan with furosemide) showing delayed drainage (T½ > 20 min). Definitive treatment: dismembered pyeloplasty (Anderson-Hynes) — excision of the obstructed segment and reanastomosis of the ureter to a spatulated renal pelvis. Success rate > 95%. Robotic-assisted pyeloplasty is now the most common approach. Endopyelotomy (endoscopic incision) is an alternative but has lower long-term success (~70-80%).
14 Benign Prostatic Hyperplasia (BPH)
Pathophysiology
BPH is a histologic diagnosis characterized by proliferation of stromal and epithelial cells in the prostatic transition zone, leading to glandular enlargement and potential bladder outlet obstruction (BOO). BPH affects ~50% of men aged 50-60 and ~90% of men > 80. The relationship between prostate size and symptom severity is not linear — a small prostate with median lobe enlargement can cause more obstruction than a very large prostate. Two components contribute to obstruction: static (tissue bulk) and dynamic (smooth muscle tone mediated by alpha-1 adrenergic receptors in the prostate and bladder neck).
Clinical Assessment
Lower urinary tract symptoms (LUTS) include storage symptoms (frequency, urgency, nocturia) and voiding symptoms (hesitancy, weak stream, intermittency, straining, incomplete emptying). Assessment: IPSS/AUA symptom score, DRE, urinalysis, PSA, uroflowmetry (Qmax), and post-void residual (PVR). Optional: transrectal or transabdominal ultrasound for prostate volume, pressure-flow urodynamics for equivocal cases.
Medical Therapy
| Drug Class | Examples | Mechanism | Key Effects/Side Effects |
|---|---|---|---|
| Alpha-1 blockers | Tamsulosin, alfuzosin, silodosin, doxazosin, terazosin | Relax prostatic smooth muscle (dynamic component) | Rapid symptom relief (days-weeks); side effects: orthostatic hypotension (less with tamsulosin/alfuzosin), retrograde ejaculation, intraoperative floppy iris syndrome (IFIS) |
| 5-alpha reductase inhibitors (5-ARIs) | Finasteride (type 2), dutasteride (type 1 & 2) | Block conversion of testosterone to DHT → reduce prostate volume by 20-30% | Full effect takes 6-12 months; reduce PSA by ~50% (must double PSA value for cancer screening); side effects: decreased libido, ED, gynecomastia |
| Combination therapy | Tamsulosin + dutasteride (Jalyn) | Both mechanisms | Superior to monotherapy for large prostates (> 30-40 mL) — MTOPS and CombAT trials |
| PDE5 inhibitors | Tadalafil 5 mg daily | Relax smooth muscle via NO/cGMP pathway | Improves both LUTS and erectile dysfunction; FDA-approved for BPH |
| Anticholinergics / Beta-3 agonists | Oxybutynin, mirabegron | Reduce detrusor overactivity (storage symptoms) | Used cautiously with BPH; check PVR (risk of retention) |
Surgical Management
Surgical indications: refractory to medical therapy, recurrent urinary retention, recurrent UTI, bladder stones secondary to BPH, renal insufficiency due to BOO, recurrent gross hematuria from BPH, and patient preference.
| Procedure | Prostate Size | Technique | Key Features |
|---|---|---|---|
| TURP | 30-80 mL | Transurethral resection with monopolar or bipolar electrocautery | Gold standard; bipolar eliminates TUR syndrome; retrograde ejaculation ~65-75% |
| HoLEP | Any size | Holmium laser enucleation — laser dissects adenoma from capsule, tissue morcellated in bladder | Size-independent; lowest retreatment rate; steep learning curve; equivalent to open prostatectomy for large glands |
| Greenlight PVP | 30-80 mL | 532 nm laser photoselective vaporization | Good for anticoagulated patients (minimal bleeding); may require retreatment |
| Rezum | 30-80 mL | Transurethral water vapor (steam) injection → thermal ablation of prostatic tissue | Office-based; preserves ejaculatory function; effect develops over weeks |
| UroLift (PUL) | 30-80 mL (no median lobe) | Implants retract obstructing lateral lobes | Preserves ejaculatory and erectile function; may need retreatment |
| Aquablation | 30-150 mL | Robotic waterjet ablation using real-time TRUS guidance | Automated resection; preserves ejaculation in some; emerging technology |
| Simple prostatectomy | > 80-100 mL | Enucleation of adenoma — robotic, laparoscopic, or open (suprapubic/retropubic) | For very large glands; robotic approach increasingly common |
Prostate Artery Embolization (PAE)
Prostate artery embolization is an interventional radiology procedure that selectively embolizes prostatic arteries to cause ischemic necrosis and shrinkage of the prostate. It is performed through a transfemoral or transradial approach. PAE is offered to men who are poor surgical candidates, those who decline surgery, or those who prioritize preservation of sexual function. Evidence shows modest improvement in IPSS and Qmax, but outcomes are generally inferior to TURP and HoLEP. PAE is not endorsed as first-line by AUA guidelines but is recognized as an option by some guidelines (NICE).
Acute Urinary Retention (AUR)
Acute urinary retention is the sudden inability to void, presenting with severe suprapubic pain and a distended, palpable bladder. Most common cause in men: BPH (precipitated by medications — anticholinergics, sympathomimetics, opioids — or constipation, UTI, anesthesia). Immediate management: urethral catheterization (Foley or intermittent). If urethral catheterization fails: coude-tip catheter (angled tip navigates the prostatic urethra), or if still unsuccessful, suprapubic catheter placement. After decompression, initiate alpha-blocker (tamsulosin) and attempt trial without catheter (TWOC) in 3-7 days. Success rate of TWOC: ~40-60% with alpha-blocker. Failed TWOC or recurrent AUR is an indication for surgical management of BPH.
15 Overactive Bladder & Urinary Incontinence
Types of Urinary Incontinence
| Type | Mechanism | Key Features |
|---|---|---|
| Stress urinary incontinence (SUI) | Urethral hypermobility or intrinsic sphincter deficiency (ISD) | Leakage with cough, sneeze, lifting, exertion; more common in women (after childbirth, menopause) and post-prostatectomy in men |
| Urgency urinary incontinence (UUI) | Detrusor overactivity (involuntary detrusor contractions) | Sudden, compelling urge to void with inability to defer; associated with OAB syndrome |
| Mixed incontinence | Combination of SUI + UUI | Very common in women; treat the predominant component first |
| Overflow incontinence | Chronic urinary retention (BOO or detrusor underactivity) | Continuous dribbling; high PVR; may be painless; causes: BPH, neurogenic bladder, medications |
| Functional incontinence | Cognitive/mobility impairment preventing timely toileting | Common in elderly/dementia; bladder function may be normal |
Overactive Bladder (OAB)
OAB is a symptom complex of urgency, with or without urgency incontinence, usually with frequency and nocturia, in the absence of infection or other pathology. Prevalence increases with age, affecting ~15-30% of adults. Diagnosis is clinical; urodynamics are not required for initial management but indicated for refractory or complex cases.
Treatment of OAB/UUI
First-line: Behavioral therapy — bladder training (scheduled voiding, progressively increasing intervals), pelvic floor muscle exercises (Kegels), fluid management, caffeine restriction, and weight loss. Second-line: Pharmacotherapy — antimuscarinics (oxybutynin, tolterodine, solifenacin, darifenacin, fesoterodine, trospium) block M3 receptors on the detrusor; side effects include dry mouth, constipation, blurred vision, and cognitive impairment (avoid in elderly — use trospium or darifenacin, which have less CNS penetration). Mirabegron (beta-3 agonist) relaxes the detrusor via a different mechanism — no anticholinergic side effects; can be combined with antimuscarinics. Vibegron is another beta-3 agonist with fewer drug interactions.
Third-line: OnabotulinumtoxinA (Botox) injection — 100-200 units injected cystoscopically into the detrusor; effective for 6-9 months; risk of urinary retention (must be willing to perform CIC). Sacral neuromodulation (SNM, InterStim) — implanted lead at S3 modulates sacral nerve function; indicated for refractory OAB and non-obstructive urinary retention. Percutaneous tibial nerve stimulation (PTNS) — office-based, weekly for 12 weeks then monthly maintenance.
Stress Urinary Incontinence — Female
Evaluation: cough stress test, voiding diary, PVR, and urodynamics for complex cases. Conservative management: pelvic floor muscle training (PFMT, Kegels) — first-line for all SUI; vaginal pessary. Surgical management: midurethral sling (MUS) — retropubic (TVT) or transobturator (TOT) polypropylene mesh tape placed tension-free at the midurethra; success rate ~80-90%. Burch colposuspension (open or laparoscopic) — sutures the periurethral tissue to Cooper ligament; durable results but more invasive. Urethral bulking agents (Bulkamid) — office-based injection of hydrogel at the bladder neck; less invasive, lower long-term success; good for ISD or patients unfit for surgery.
Stress Urinary Incontinence — Male (Post-Prostatectomy)
Post-radical-prostatectomy incontinence affects ~5-20% of men at 1 year. Conservative: PFMT (ideally started preoperatively). Surgical: Artificial urinary sphincter (AUS, AMS 800) — the gold standard for moderate-to-severe male SUI; an inflatable cuff placed around the bulbar urethra with a control pump in the scrotum; success rate ~75-90%. Male sling (AdVance, ATOMS) — for mild-to-moderate SUI; repositions/compresses the bulbar urethra.
16 Neurogenic Bladder
Overview
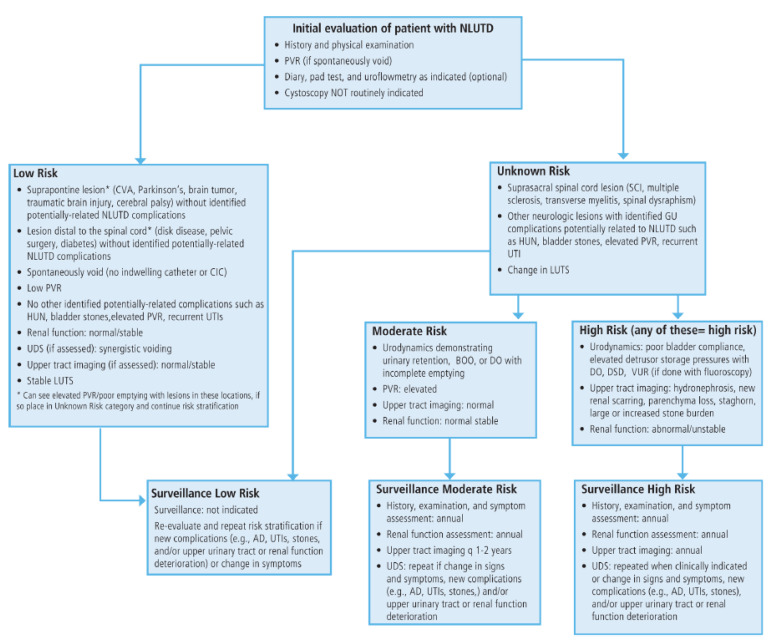
Neurogenic bladder refers to bladder dysfunction caused by neurologic disease or injury. The clinical presentation depends on the level of the neurologic lesion:
| Lesion Level | Type | Bladder Behavior | Examples |
|---|---|---|---|
| Suprapontine (above pons) | Upper motor neuron | Detrusor overactivity with coordinated sphincter; incontinence | Stroke, Parkinson disease, brain tumors, MS (can be any pattern) |
| Suprasacral spinal cord (above S2-S4) | Upper motor neuron | Detrusor overactivity with detrusor-sphincter dyssynergia (DSD) — the most dangerous pattern (high pressures → upper tract damage) | Spinal cord injury (above conus), MS, transverse myelitis |
| Sacral (S2-S4) / infrasacral | Lower motor neuron | Acontractile/areflexic detrusor; overflow incontinence; open/denervated sphincter | Cauda equina syndrome, conus medullaris injury, pelvic surgery, diabetic neuropathy |
Evaluation
Urodynamic study (UDS) is essential — measures detrusor pressure during filling and voiding, assesses compliance, detects involuntary contractions, and identifies DSD. Key parameters: detrusor leak point pressure (DLPP) > 40 cm H2O is associated with upper tract deterioration and requires intervention. Renal ultrasound is performed to assess for hydronephrosis. VCUG evaluates for vesicoureteral reflux. Serum creatinine and GFR monitor renal function.
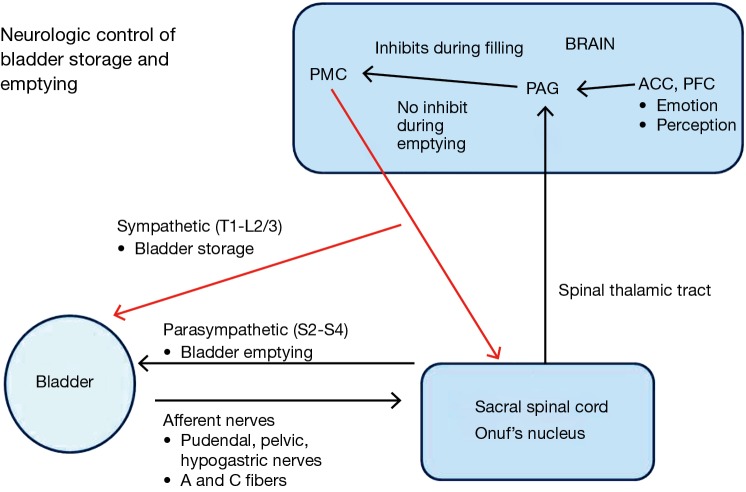
Management
Clean intermittent catheterization (CIC) is the gold standard for bladder emptying in neurogenic bladder — preferred over indwelling catheters (which increase UTI risk, stone formation, urethral erosion, and squamous cell carcinoma of the bladder with long-term use). CIC typically performed 4-6 times daily. Antimuscarinics or beta-3 agonists reduce detrusor overactivity and improve compliance. OnabotulinumtoxinA (200 units) injected into the detrusor is effective for neurogenic detrusor overactivity refractory to oral medications. Augmentation cystoplasty (enterocystoplasty) — using ileum or sigmoid colon to enlarge the bladder — is a last resort for refractory low-compliance bladders threatening upper tract function. Complications: mucus production, metabolic acidosis, stones, perforation, and theoretical malignancy risk.
Spinal Cord Injury — Acute Phase
During spinal shock (immediately after SCI, lasting days to weeks), the bladder is areflexic and acontractile. Management during this phase: indwelling urethral catheter initially, then transition to CIC as soon as feasible. After spinal shock resolves, patients with suprasacral injuries typically develop detrusor overactivity with DSD, while those with conus/cauda equina injuries develop areflexic bladder. Annual urologic surveillance (renal US, serum creatinine, urodynamics) is recommended for all SCI patients to monitor for upper tract deterioration.
Autonomic Dysreflexia
Emergency Autonomic dysreflexia (AD) is a potentially life-threatening emergency in patients with SCI at T6 or above. Triggered by noxious stimuli below the level of injury — the most common trigger is bladder distension (blocked catheter, urinary retention). Presentation: severe paroxysmal hypertension (SBP > 200 mmHg possible), pounding headache, flushing and sweating above the injury level, bradycardia, and piloerection. Management: sit the patient upright, remove tight clothing, identify and relieve the trigger (catheterize the bladder or relieve catheter obstruction), and if BP remains elevated, give fast-acting antihypertensive (nifedipine SL, nitrate paste, or IV hydralazine). Prevention: regular bladder emptying, bowel program, and avoidance of triggers.
17 Urethral Stricture Disease
Etiology
Urethral stricture is fibrotic narrowing of the urethra caused by spongiofibrosis (scar tissue in the corpus spongiosum). Etiologies: iatrogenic (most common in developed countries — urethral catheterization, instrumentation, hypospadias repair, TURP), idiopathic (~30-40%, likely related to unrecognized lichen sclerosus or trauma), traumatic (straddle injury → bulbar urethra; pelvic fracture → membranous/prostatic urethra — posterior urethral disruption injury), infectious (gonococcal urethritis — historically common; Chlamydia), and lichen sclerosus (balanitis xerotica obliterans/BXO — affects glans and fossa navicularis, may extend to penile urethra).
Diagnosis
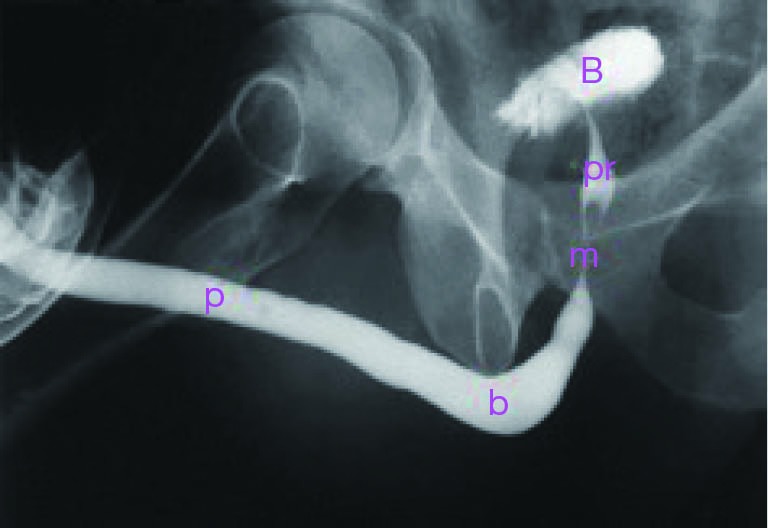
Retrograde urethrogram (RUG) is the gold standard imaging study — demonstrates the location, length, and degree of stricture. Combined with voiding cystourethrogram (VCUG) for posterior urethral evaluation. Uroflowmetry shows an obstructive pattern (low Qmax, prolonged curve). Cystourethroscopy provides direct visualization and is both diagnostic and used for planning repair.
Management
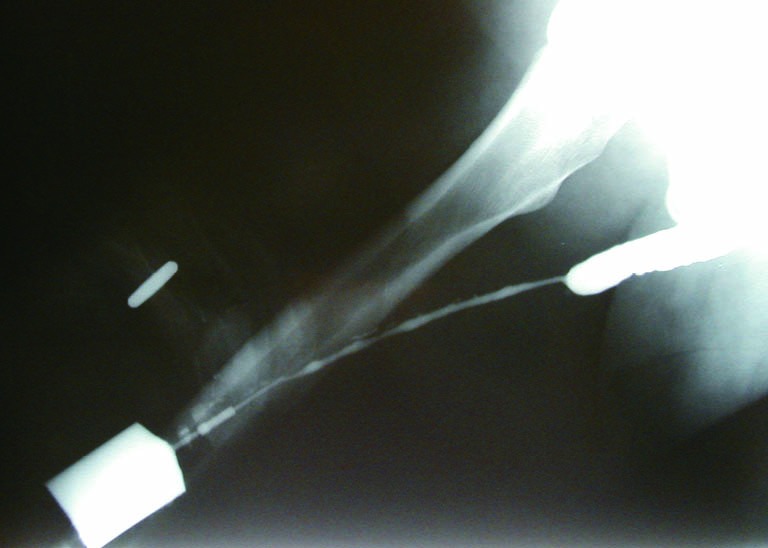
Urethral dilation — progressive passage of dilators (filiform and followers, balloon, or sounds); temporary relief but high recurrence rate (~50-60% at 1 year). Appropriate as initial treatment for short, non-recurrent strictures. Direct vision internal urethrotomy (DVIU) — endoscopic cold-knife incision of the stricture (typically at the 12 o'clock position); similar recurrence rate to dilation (~50-60%); best results for short (< 2 cm), single, bulbar strictures. Repeated DVIU has diminishing returns — after 2 failed DVIU/dilations, urethroplasty should be strongly considered.
Urethroplasty is the definitive treatment with the highest long-term success rates (85-95%). Types: (1) Excision and primary anastomosis (EPA) — for short (≤ 2 cm) bulbar strictures; strictured segment is excised, and the healthy urethral ends are spatulated and anastomosed; success rate > 95%. (2) Buccal mucosal graft (BMG) urethroplasty — for longer strictures (> 2 cm); a graft harvested from the inner cheek is placed dorsally (Barbagli dorsal onlay), ventrally, or as a dorsal inlay (Asopa) on the urethral plate; success rate ~85-90%. (3) Staged urethroplasty — for complex, long, or panurethral strictures (especially in lichen sclerosus); first stage opens the stricture and places BMG; second stage tubularizes the neourethra after 3-6 months.
For posterior urethral disruption injuries (pelvic fracture urethral injury — PFUI): initial management is suprapubic catheter placement; delayed posterior urethroplasty at 3-6 months after the pelvic fracture has healed. Immediate attempts at realignment are an option but carry risk of stricture, incontinence, and ED. The classic presentation of PFUI is blood at the urethral meatus, inability to void, and high-riding prostate on DRE in the setting of pelvic fracture. A retrograde urethrogram must be performed before any attempt at urethral catheterization.
Urethral Trauma Classification
| Type | Description | Management |
|---|---|---|
| Anterior (bulbar) | Straddle injury; direct blow to perineum; most common cause of anterior urethral injury | Urethral catheter if partial; suprapubic catheter if complete; delayed urethroplasty |
| Posterior (membranous/prostatic) | Pelvic fracture urethral injury; occurs in ~5-10% of pelvic fractures; disruption at prostatomembranous junction | Suprapubic catheter; delayed posterior urethroplasty at 3-6 months; primary realignment is an option but controversial |
Lichen Sclerosus (Balanitis Xerotica Obliterans)
Lichen sclerosus (LS), formerly BXO, is a chronic inflammatory dermatosis that affects the glans, prepuce, and urethral meatus. It causes progressive fibrosis with a characteristic white, sclerotic appearance. LS is the most common cause of acquired phimosis in adults and a major cause of meatal stenosis and fossa navicularis strictures. The etiology is autoimmune. LS is associated with long, panurethral strictures that are challenging to manage. Buccal mucosal grafts are preferred for LS-related strictures because BMG is resistant to LS recurrence (unlike genital skin grafts). LS carries a small risk (~5%) of squamous cell carcinoma — long-term surveillance is recommended.
18 Testicular Torsion & Scrotal Emergencies
Testicular Torsion
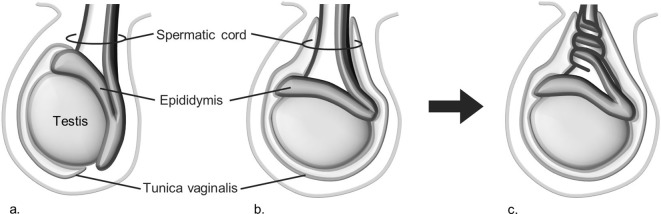
Emergency Testicular torsion is a surgical emergency caused by twisting of the spermatic cord with resultant ischemia of the testis. Peak incidence: bimodal — neonates and adolescents (12-18 years). The underlying anomaly is the "bell clapper" deformity — high attachment of the tunica vaginalis allowing the testis to rotate freely on the spermatic cord (present bilaterally in most cases).
Presentation: sudden-onset severe testicular pain (often during sleep or physical activity), nausea/vomiting, high-riding testis with a horizontal lie, absent cremasteric reflex (most sensitive physical exam finding — ~99% sensitivity in torsion), and a thickened/twisted spermatic cord. The cremasteric reflex is absent in testicular torsion.
Time to Salvage
| Duration of Torsion | Salvage Rate |
|---|---|
| < 6 hours | ~90-100% |
| 6-12 hours | ~50% |
| 12-24 hours | ~20% |
| > 24 hours | ~0-10% |
Diagnosis & Management
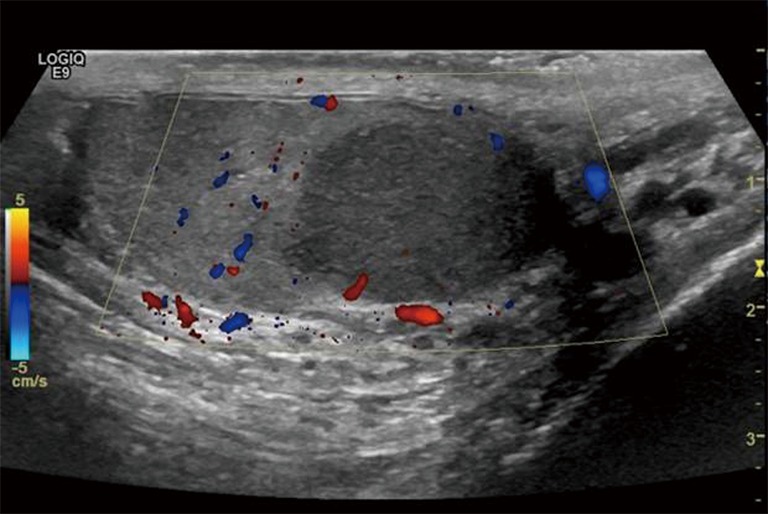
If clinical suspicion is high, do NOT delay surgery for imaging — proceed directly to scrotal exploration. Color Doppler ultrasound (decreased or absent blood flow to the affected testis) is obtained when the diagnosis is uncertain. Sensitivity ~88-100%, specificity ~90-100%.
Treatment: emergent scrotal exploration, detorsion, and bilateral orchiopexy. The affected testis is untwisted, assessed for viability (return of color/bleeding after warm-saline wraps), and fixed to the dartos fascia in 3-point fixation. The contralateral testis must also be fixed (bilateral orchiopexy) because the bell clapper deformity is usually bilateral. If the testis is nonviable, orchiectomy is performed. Manual detorsion (outward rotation — "open the book") can be attempted while preparing for surgery but should not delay definitive operative exploration.
Torsion of Appendages
Torsion of the appendix testis (Müllerian duct remnant) or appendix epididymis is the most common cause of acute scrotum in prepubertal boys. Presents with gradual-onset scrotal pain, often with a visible "blue dot sign" (infarcted appendage visible through the scrotal skin). Cremasteric reflex is typically present. Management is conservative: NSAIDs, rest, scrotal support. Surgery only if torsion of the testis itself cannot be excluded.
Epididymitis & Epididymo-Orchitis
Epididymitis presents with gradual-onset scrotal pain, swelling of the epididymis (starting posterolaterally), fever, and urinary symptoms. In young men (< 35): commonly caused by Chlamydia trachomatis or Neisseria gonorrhoeae — treat with ceftriaxone 500 mg IM + doxycycline 100 mg BID × 10 days. In older men or boys: commonly caused by coliforms (E. coli) — treat with fluoroquinolone or TMP-SMX. Prehn sign (relief of pain with testicular elevation) is classically positive in epididymitis but is unreliable for excluding torsion.
19 Cryptorchidism & Inguinal Disorders
Cryptorchidism
Cryptorchidism (undescended testis) is the most common congenital anomaly of the male genitalia, affecting ~3% of full-term and ~30% of premature male neonates. By 6 months of age, most undescended testes will spontaneously descend — prevalence stabilizes at ~1% after 1 year. The testis may be palpable (80% — in the inguinal canal, prescrotal, or ectopic) or nonpalpable (20% — intra-abdominal, absent/vanishing testis, or ectopic).
Complications of Cryptorchidism
Infertility: Bilateral cryptorchidism causes infertility in ~75% if untreated; even unilateral cryptorchidism reduces fertility. Elevated temperature impairs spermatogenesis. Malignancy: 3-8× increased risk of testicular cancer (most commonly seminoma) — the risk is highest for intra-abdominal testes. Orchiopexy before puberty may reduce cancer risk but does not eliminate it; orchiopexy facilitates examination and tumor detection. Torsion: Higher risk due to abnormal attachment. Inguinal hernia: Patent processus vaginalis present in ~90% of cryptorchid testes.
Management
Hormonal therapy (hCG or GnRH) is not recommended by AUA/EAU guidelines due to low success rates and lack of long-term benefit. Orchiopexy is the treatment of choice and should be performed between 6 and 18 months of age (AUA guideline: referral by 6 months, surgery by 18 months). For palpable testes: inguinal orchiopexy (inguinal incision, mobilization of testis and cord, closure of processus vaginalis, fixation of testis in a sub-dartos pouch in the scrotum). For nonpalpable testes: diagnostic laparoscopy is the first step — if testis is found intra-abdominally and viable, either single-stage or two-stage Fowler-Stephens orchiopexy (stage 1: ligate testicular vessels laparoscopically; stage 2: 6 months later, bring testis to scrotum on a pedicle of vas deferens and its artery + gubernacular collaterals). If testis is absent or nubbins found, orchiectomy of remnant.
Inguinal Hernia & Hydrocele in Children
An indirect inguinal hernia in children results from a patent processus vaginalis. It presents as a groin bulge that appears with crying/straining and is reducible. Unlike adult hernias, pediatric inguinal hernias do not resolve spontaneously and carry a risk of incarceration — all pediatric inguinal hernias require surgical repair. Repair: inguinal hernia repair with high ligation of the processus vaginalis (no mesh in children). A communicating hydrocele results from a narrow patent processus vaginalis that allows peritoneal fluid (but not abdominal contents) to surround the testis — size fluctuates with activity/position. Communicating hydroceles in children < 12-24 months may resolve spontaneously; if persistent beyond 12-24 months, surgical repair (same as hernia repair — ligation of processus vaginalis). A non-communicating (simple) hydrocele in adults is managed with observation if asymptomatic; aspiration is temporary; hydrocelectomy if bothersome.
20 Vesicoureteral Reflux
Overview
Vesicoureteral reflux (VUR) is the retrograde flow of urine from the bladder into the ureter and renal collecting system. It is the most common urologic abnormality in children, affecting ~1% of the general pediatric population and ~30-40% of children who present with UTI. VUR predisposes to recurrent pyelonephritis and reflux nephropathy (renal scarring), which can lead to hypertension and chronic kidney disease.
Classification
VUR is classified by voiding cystourethrogram (VCUG) into 5 grades (International Reflux Study):
| Grade | Description | Spontaneous Resolution Rate |
|---|---|---|
| I | Reflux into ureter only (does not reach renal pelvis) | ~80% |
| II | Reflux into renal pelvis and calyces without dilation | ~60-80% |
| III | Mild dilation of ureter and renal pelvis; mild calyceal blunting | ~50% |
| IV | Moderate dilation; tortuous ureter; moderate calyceal blunting | ~10-20% |
| V | Severe dilation and tortuosity; papillary impression lost; gross intrarenal reflux | < 5% |
Management
Medical (conservative) management: Low-grade VUR (I-III) in young children has high spontaneous resolution rates. Continuous antibiotic prophylaxis (CAP) with low-dose trimethoprim or nitrofurantoin reduces recurrent UTI. The RIVUR trial showed that CAP reduces UTI recurrence by ~50% in children with VUR but does not prevent renal scarring.
Surgical management is indicated for: high-grade VUR (IV-V), breakthrough UTIs on prophylaxis, persistent VUR, non-compliance with medical management, and renal scarring with ongoing reflux. Options: (1) Endoscopic injection (STING/Deflux) — injection of dextranomer/hyaluronic acid (Deflux) at the ureteral orifice; outpatient procedure; success rate ~70-80% for grades I-III, lower for higher grades; may require repeat injection. (2) Open ureteral reimplantation — the gold standard with ~95-98% success rate; the ureter is reimplanted into the bladder with a longer submucosal tunnel (5:1 tunnel length-to-diameter ratio) to create a flap-valve mechanism (Cohen cross-trigonal or Lich-Gregoir extravesical technique). (3) Robotic-assisted reimplantation is emerging with comparable success rates.
Renal Scarring & Reflux Nephropathy
Reflux nephropathy is renal scarring resulting from VUR-associated pyelonephritis, particularly in young children (< 5 years). It is the most common cause of hypertension in children and the leading urologic cause of end-stage renal disease in children. Risk factors for scarring: high-grade VUR (IV-V), recurrent febrile UTIs, delayed treatment, younger age at first UTI, and intrarenal reflux. DMSA (dimercaptosuccinic acid) renal scan is the gold standard for detecting renal scarring — more sensitive than ultrasound. Prevention of scarring requires prompt treatment of febrile UTIs (antibiotics within 72 hours of fever onset) and prevention of recurrent UTIs through antibiotic prophylaxis or surgical correction of VUR.
21 Hypospadias & Congenital Anomalies
Hypospadias
Hypospadias is a congenital anomaly in which the urethral meatus opens on the ventral surface of the penis, proximal to the normal glanular position. Incidence: ~1 in 200-300 live male births — one of the most common congenital anomalies. Classification by meatal position: distal/anterior (~70% — glanular, coronal, distal shaft), middle/midshaft (~15%), and proximal/posterior (~15% — penoscrotal, scrotal, perineal). Associated findings: ventral curvature (chordee) in ~10-50%, hooded/incomplete foreskin (dorsal hood), and undescended testis (especially in proximal forms — evaluate for DSD if bilateral UDT + hypospadias).
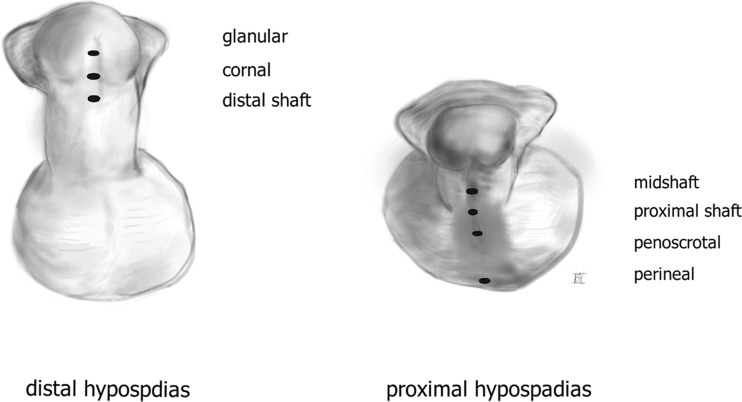
Repair Principles
Repair is recommended at 6-18 months of age. Goals: straight penis, slit-like meatus at the tip of the glans, conical glans shape, and functional urethra with a good stream. The most commonly used technique is the TIP (tubularized incised plate) repair (Snodgrass) — relaxing midline incision of the urethral plate allows tubularization into a neourethra; used for distal and many midshaft hypospadias with excellent cosmetic and functional results. For proximal hypospadias or those with severe chordee: two-stage repair (first stage: chordee correction and placement of buccal mucosal or preputial skin graft; second stage: tubularization of the neourethral plate at 6+ months). Circumcision must NOT be performed before hypospadias repair — the prepuce may be needed as tissue for reconstruction.
Posterior Urethral Valves (PUV)
Posterior urethral valves (PUV) are the most common cause of congenital lower urinary tract obstruction in males. Type I valves (most common, ~95%) are mucosal folds extending from the verumontanum to the membranous urethra. Presentation: prenatal hydronephrosis and oligohydramnios (severe cases → pulmonary hypoplasia), postnatal urinary retention, poor stream, UTI, renal insufficiency. Diagnosis: VCUG showing dilated posterior urethra with a "keyhole" sign. Treatment: primary valve ablation (endoscopic incision) once the infant is stable; vesicostomy if the infant is too small for endoscopic instruments. Despite treatment, ~30-40% develop end-stage renal disease due to renal dysplasia and the "valve bladder" (chronic high-pressure detrusor changes with poor compliance).
Epispadias & Bladder Exstrophy
Bladder exstrophy is a rare (1:30,000-50,000 births) midline closure defect where the bladder is open and exposed on the anterior abdominal wall. Associated with epispadias (dorsally displaced urethral meatus), pubic diastasis, and inguinal hernias. Management requires staged reconstruction: primary closure of the bladder and abdominal wall (ideally within 72 hours of birth), epispadias repair, and later bladder neck reconstruction for continence. The exstrophy-epispadias complex carries a lifelong risk of malignancy (adenocarcinoma).
Ureterocele & Ectopic Ureter
A ureterocele is a cystic dilation of the intravesical submucosal ureter, most commonly associated with the upper pole moiety of a duplex collecting system. Classified as intravesical (orthotopic) or ectopic (extends to the bladder neck or urethra). Presents in children with UTI, obstruction, or prenatal hydronephrosis. On ultrasound, appears as a thin-walled cystic structure within the bladder ("cobra head" sign on IVP). Treatment depends on size and obstruction: endoscopic puncture/incision, upper pole heminephroureterectomy for a non-functioning moiety, or ureterocele excision with reimplantation.
An ectopic ureter inserts at a location other than the normal trigonal position. In the Weigert-Meyer rule for duplex systems: the upper pole ureter inserts medially and inferiorly (more prone to obstruction/ureterocele), while the lower pole ureter inserts laterally and superiorly (more prone to reflux). In girls, an ectopic ureter may insert below the external sphincter (into the vagina or vestibule) — presenting with continuous urinary dribbling despite normal voiding. In boys, ectopic ureters always insert above the external sphincter (into the prostatic urethra, seminal vesicles, or vas deferens) and do not cause incontinence.
22 Fournier Gangrene, Priapism & Paraphimosis
Fournier Gangrene
Emergency Fournier gangrene is a life-threatening necrotizing fasciitis of the perineum and external genitalia. It is a polymicrobial infection (aerobic + anaerobic organisms: E. coli, Bacteroides, Streptococcus, Staphylococcus, Clostridium) involving the superficial fascia (Colles, dartos, Buck, Scarpa). Sources: perianal (30-50%), urogenital (20-40%), cutaneous (20%). Risk factors: diabetes mellitus (most common), immunosuppression, alcoholism, obesity, chronic kidney disease.
Presentation: rapidly progressive perineal/scrotal pain, swelling, erythema extending beyond the apparent margin, crepitus (subcutaneous gas), systemic toxicity (fever, tachycardia, sepsis). Management: (1) aggressive surgical debridement — the cornerstone of treatment; wide excision of all necrotic tissue; often requires multiple returns to the OR (planned "second look" at 24-48 hours); (2) broad-spectrum IV antibiotics (vancomycin + piperacillin-tazobactam + clindamycin or a carbapenem); (3) resuscitation and ICU care. Testes are typically spared (separate blood supply from the testicular artery) and can be placed in subcutaneous thigh pouches. Wound management: negative pressure wound therapy (wound VAC) followed by delayed primary closure, skin grafting, or flap reconstruction. Mortality: 20-40% despite treatment — higher with delayed debridement.
Priapism
Emergency Priapism is a persistent penile erection unrelated to sexual stimulation lasting > 4 hours. Two main types:
| Feature | Ischemic (Low-Flow) — EMERGENCY | Nonischemic (High-Flow) |
|---|---|---|
| Mechanism | Veno-occlusive: blood trapped in corpora cavernosa | Arterial: unregulated cavernosal arterial inflow (usually post-traumatic) |
| Pain | Painful, rigid | Painless, partially rigid |
| Blood gas | Dark blood; pO2 < 30, pCO2 > 60, pH < 7.25 (ischemic) | Bright red blood; arterial gas values |
| Causes | Sickle cell disease (most common in children), medications (PDE5 inhibitors, intracavernosal injections, trazodone, antipsychotics), idiopathic | Perineal/penile trauma (straddle injury) → cavernosal artery pseudoaneurysm/fistula |
| Management | URGENT: corporal aspiration with 16-18G needle → injection of phenylephrine (100-500 mcg every 3-5 min) → if refractory (> 72 hrs), surgical shunt (distal: Winter percutaneous, T-shunt; proximal: Quackels; last resort: penile prosthesis) | Non-urgent: observation (many resolve); selective arterial embolization if persistent |
| Sequelae | Smooth muscle necrosis & fibrosis → ED if untreated > 24-48 hrs | ED rare |
Paraphimosis
Emergency Paraphimosis is the inability to reduce a retracted foreskin back over the glans penis, causing a constricting band at the coronal sulcus. This leads to venous congestion, edema of the glans, and if untreated, arterial compromise and gangrene. Most common cause: failure to replace the foreskin after catheterization or examination. Management: (1) manual reduction — apply steady circumferential pressure to the edematous glans for 5-10 minutes (ice/osmotic agents like granulated sugar can reduce edema), then push the glans back through the phimotic ring while pulling the foreskin forward; (2) if manual reduction fails: dorsal slit (incision of the constricting band under local anesthesia) to release the constriction; (3) elective circumcision after edema resolves to prevent recurrence.
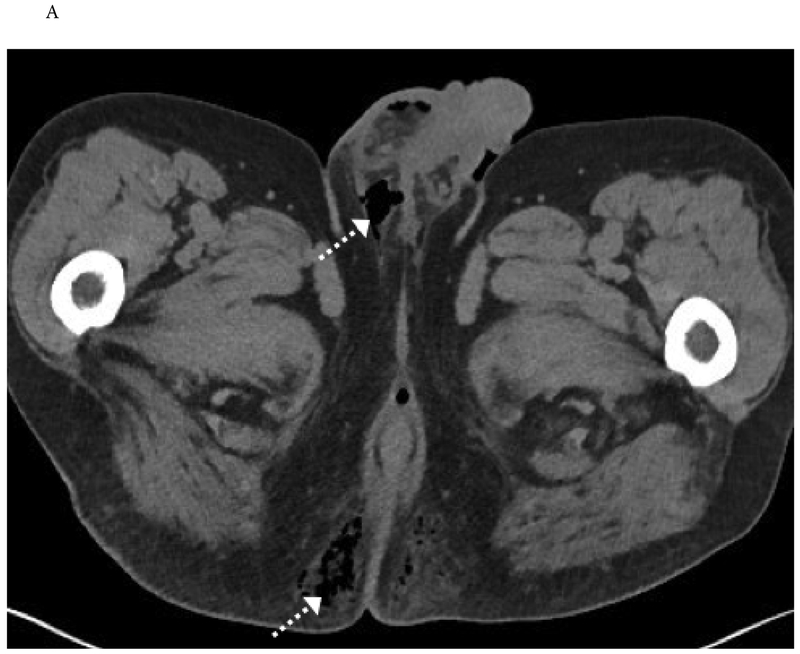
Erectile Dysfunction
Erectile dysfunction (ED) is the inability to achieve or maintain an erection sufficient for satisfactory sexual performance. Prevalence increases with age (~40% of men at age 40, ~70% at age 70). Etiology is often multifactorial: vascular (most common — atherosclerosis of penile arteries; shared risk factors with cardiovascular disease), neurogenic (diabetes, spinal cord injury, post-radical prostatectomy), hormonal (hypogonadism), medication-related (antihypertensives, SSRIs, anti-androgens), psychogenic, and structural (Peyronie disease).
Evaluation: sexual history, IIEF-5 questionnaire, focused physical exam, fasting glucose/HbA1c, lipid panel, morning testosterone. Treatment: First-line: PDE5 inhibitors — sildenafil (Viagra, 25-100 mg), tadalafil (Cialis, 5-20 mg — also daily 5 mg for BPH/ED), vardenafil (Levitra), avanafil (Stendra). Contraindicated with nitrates (severe hypotension). Second-line: Intracavernosal injection therapy (alprostadil, trimix — PGE1 + phentolamine + papaverine), vacuum erection devices, intraurethral alprostadil (MUSE). Third-line: Penile prosthesis implantation — inflatable 3-piece (AMS 700, Coloplast Titan) provides the most natural erection; malleable/semi-rigid rods are simpler. Patient satisfaction rates exceed 90% with inflatable prostheses.
Peyronie Disease
Peyronie disease is an acquired fibrotic disorder of the tunica albuginea causing penile curvature, pain, and potential ED. Prevalence ~3-9% of adult men. Two phases: acute/inflammatory phase (first 6-12 months — pain, evolving curvature, plaque formation) and chronic/stable phase (pain resolves, curvature stabilizes). Treatment during stable phase: intralesional collagenase (Xiaflex) for curvature 30-90 degrees (FDA-approved); surgical correction for refractory cases — plication (Nesbit, 16-dot) for curvature < 60-70 degrees with adequate length, or plaque incision/excision with grafting for severe curvature or short penis. Penile prosthesis for concurrent ED.
23 Classification Systems
Gleason / ISUP Grading (Prostate Cancer)
| ISUP Grade Group | Gleason Score | Pattern Description |
|---|---|---|
| 1 | 3+3 = 6 | Well-formed individual glands |
| 2 | 3+4 = 7 | Predominantly well-formed glands with a lesser component of poorly formed/fused/cribriform glands |
| 3 | 4+3 = 7 | Predominantly poorly formed/fused/cribriform glands with a lesser component of well-formed glands |
| 4 | 8 (4+4, 3+5, 5+3) | Only poorly formed/fused/cribriform glands, or lacking gland formation with or without well-formed glands |
| 5 | 9-10 (4+5, 5+4, 5+5) | Lacking gland formation (sheets, cords, single cells, comedonecrosis) with or without poorly formed glands |
TNM Staging — Kidney Cancer (AJCC 8th Edition)
| T Stage | Description |
|---|---|
| T1a | ≤ 4 cm, confined to kidney |
| T1b | > 4 cm but ≤ 7 cm, confined to kidney |
| T2a | > 7 cm but ≤ 10 cm, confined to kidney |
| T2b | > 10 cm, confined to kidney |
| T3a | Extends into renal vein or perinephric/renal sinus fat, not beyond Gerota fascia |
| T3b | Extends into IVC below diaphragm |
| T3c | Extends into IVC above diaphragm or invades IVC wall |
| T4 | Extends beyond Gerota fascia or into ipsilateral adrenal |
TNM Staging — Testicular Cancer (AJCC 8th Edition)
| T Stage | Description |
|---|---|
| pT1 (Seminoma) | Any size, limited to testis without lymphovascular invasion (LVI) |
| pT1 (NSGCT) | Limited to testis without LVI; may invade rete testis, epididymis, tunica albuginea |
| pT2 | LVI present, or involvement of hilar soft tissue, epididymis, or penetrates tunica vaginalis |
| pT3 | Invades spermatic cord |
| pT4 | Invades scrotum |
Bosniak Classification (Renal Cysts) — Summary
| Category | Management | Malignancy Risk |
|---|---|---|
| I — Simple cyst | Benign, no follow-up | < 1% |
| II — Minimally complex | Benign, no follow-up | < 5% |
| IIF — Mildly complex, needs follow-up | Follow-up imaging | 5-15% |
| III — Indeterminate | Surgery or active surveillance | 40-60% |
| IV — Clearly malignant | Surgery | > 80% |
AUA Symptom Score (IPSS)
| Score Range | Severity | Implication |
|---|---|---|
| 0-7 | Mild | Watchful waiting acceptable if not bothersome |
| 8-19 | Moderate | Consider medical therapy |
| 20-35 | Severe | Medical or surgical therapy recommended |
VUR Grading (International Reflux Study)
| Grade | Description |
|---|---|
| I | Reflux into non-dilated ureter only |
| II | Reflux into renal pelvis and calyces, no dilation, normal calyceal fornices |
| III | Mild to moderate dilation of ureter, renal pelvis, and calyces; minimal calyceal blunting |
| IV | Moderate dilation and tortuosity of ureter; moderate dilation of pelvis and calyces; complete obliteration of sharp calyceal angles |
| V | Gross dilation and tortuosity of ureter, pelvis, and calyces; papillary impressions no longer visible; intrarenal reflux |
Stone Composition & Crystal Morphology
| Stone Type | Crystal Shape | X-Ray Appearance | Urine pH |
|---|---|---|---|
| Calcium oxalate monohydrate | Dumbbell/oval | Radiopaque | Any |
| Calcium oxalate dihydrate | Bipyramidal envelope | Radiopaque | Any |
| Calcium phosphate (apatite) | Amorphous | Radiopaque | > 6.5 |
| Uric acid | Rhomboid/rosette | Radiolucent | < 5.5 |
| Struvite (MAP) | Coffin-lid | Radiopaque | > 7.2 |
| Cystine | Hexagonal | Semi-opaque | < 6.5 |
PI-RADS v2.1 (Prostate MRI)
| Score | Assessment | Likelihood of Clinically Significant Cancer |
|---|---|---|
| 1 | Very low | Clinically significant cancer highly unlikely |
| 2 | Low | Clinically significant cancer unlikely |
| 3 | Intermediate | Equivocal — clinically significant cancer possible |
| 4 | High | Clinically significant cancer likely |
| 5 | Very high | Clinically significant cancer highly likely |
Clavien-Dindo Classification of Surgical Complications
| Grade | Definition | Examples in Urology |
|---|---|---|
| I | Any deviation from normal postoperative course; no pharmacologic/surgical/endoscopic/radiologic intervention needed | Wound infection treated at bedside, ileus resolving with conservative measures |
| II | Requiring pharmacologic treatment, blood transfusion, or TPN | Blood transfusion after PCNL, antibiotics for UTI, DVT requiring anticoagulation |
| IIIa | Requiring surgical/endoscopic/radiologic intervention — NOT under general anesthesia | Ureteral stent placement for urinoma, percutaneous drainage of abscess |
| IIIb | Requiring intervention under general anesthesia | Reoperation for bleeding, repair of ureteral injury |
| IVa | Single organ dysfunction (including dialysis) | Acute renal failure requiring dialysis, respiratory failure requiring intubation |
| IVb | Multi-organ dysfunction | Septic shock with multi-organ failure |
| V | Death | Any cause |
R.E.N.A.L. Nephrometry Score (Renal Tumor Complexity)
The R.E.N.A.L. nephrometry score standardizes description of renal tumor complexity to guide surgical planning (partial vs radical nephrectomy):
| Component | 1 Point | 2 Points | 3 Points |
|---|---|---|---|
| R — Radius (maximal diameter) | ≤ 4 cm | > 4 to < 7 cm | ≥ 7 cm |
| E — Exophytic/endophytic | ≥ 50% exophytic | < 50% exophytic | Entirely endophytic |
| N — Nearness to collecting system/sinus (mm) | ≥ 7 mm | > 4 to < 7 mm | ≤ 4 mm |
| A — Anterior/Posterior | a = anterior, p = posterior, x = neither (suffix, not scored) | ||
| L — Location relative to polar lines | Entirely above/below polar line | Crosses a polar line | > 50% across polar line or crosses axial renal midline, or entirely between polar lines |
Total score: 4-6 = low complexity, 7-9 = moderate complexity, 10-12 = high complexity. Higher complexity scores correlate with longer ischemia times, higher complication rates, and greater likelihood of requiring radical nephrectomy.
Urinary Tract Infection — Classification & Management
| Type | Definition | Management |
|---|---|---|
| Uncomplicated cystitis | Lower UTI in a non-pregnant, pre-menopausal woman with normal urinary tract | Nitrofurantoin 5 days, TMP-SMX 3 days, or fosfomycin single dose |
| Uncomplicated pyelonephritis | Upper UTI without complicating factors | Oral fluoroquinolone 5-7 days or TMP-SMX 14 days; IV if unable to tolerate oral |
| Complicated UTI | UTI with structural/functional abnormality (stones, obstruction, catheter, neurogenic bladder, male sex, pregnancy, immunosuppression) | Broader spectrum antibiotics (fluoroquinolone, cephalosporin, or carbapenem), longer course (7-14 days); address underlying cause |
| Catheter-associated UTI (CAUTI) | UTI in patient with indwelling catheter or within 48h of removal | Remove or replace catheter; treat only if symptomatic; 7 days if symptoms resolve promptly |
| Recurrent UTI | ≥ 2 infections in 6 months or ≥ 3 in 12 months | Evaluate for structural cause; low-dose prophylaxis (nitrofurantoin, TMP-SMX); vaginal estrogen in postmenopausal women; D-mannose |
| Emphysematous pyelonephritis | Necrotizing infection with gas in renal parenchyma; associated with diabetes | Emergency — IV antibiotics + percutaneous drainage or nephrectomy for severe cases; mortality 20-40% |
24 Medications Master Table
Alpha-Adrenergic Blockers (BPH / MET)
| Drug | Selectivity | Dose | Key Notes |
|---|---|---|---|
| Tamsulosin | Alpha-1A selective | 0.4 mg daily | Most commonly prescribed; less orthostatic hypotension; IFIS risk; also used for MET |
| Alfuzosin | Alpha-1 (uroselective) | 10 mg daily (ER) | Once daily; contraindicated with CYP3A4 inhibitors |
| Silodosin | Alpha-1A (most selective) | 8 mg daily | Highest rate of retrograde ejaculation (~28%) |
| Doxazosin | Non-selective alpha-1 | 1-8 mg daily | More orthostatic hypotension; also treats hypertension |
| Terazosin | Non-selective alpha-1 | 1-10 mg daily | Requires dose titration; orthostatic hypotension |
5-Alpha Reductase Inhibitors
| Drug | Mechanism | Dose | Key Notes |
|---|---|---|---|
| Finasteride | Type 2 5-AR inhibitor | 5 mg daily | Reduces prostate volume ~20-30%; PSA reduced ~50%; effect in 6-12 months |
| Dutasteride | Type 1 & 2 5-AR inhibitor | 0.5 mg daily | Dual inhibition; similar clinical outcomes to finasteride; long half-life (5 weeks) |
Antimuscarinics (OAB)
| Drug | Selectivity | Dose | Key Notes |
|---|---|---|---|
| Oxybutynin | Non-selective M | 5 mg BID-TID (IR), patch, gel | Most anticholinergic side effects; transdermal form has fewer side effects; avoid in elderly |
| Tolterodine | Non-selective M | 2 mg BID (IR) or 4 mg daily (ER) | Better tolerated than oxybutynin; ER preferred |
| Solifenacin | M3 selective | 5-10 mg daily | Once daily; less dry mouth than oxybutynin |
| Darifenacin | M3 selective | 7.5-15 mg daily | Minimal CNS effects (less crosses BBB); good for elderly |
| Trospium | Non-selective M | 20 mg BID | Quaternary amine — does NOT cross BBB; safest for cognition in elderly |
| Fesoterodine | Non-selective M | 4-8 mg daily | Active metabolite of tolterodine |
Beta-3 Agonists (OAB)
| Drug | Dose | Key Notes |
|---|---|---|
| Mirabegron | 25-50 mg daily | No anticholinergic side effects; may raise BP slightly; can combine with antimuscarinics |
| Vibegron | 75 mg daily | No CYP2D6 inhibition (fewer drug interactions than mirabegron) |
Androgen Deprivation Therapy
| Drug | Class | Route | Key Notes |
|---|---|---|---|
| Leuprolide | GnRH agonist | IM/SC (1, 3, 4, 6 month depot) | Initial testosterone flare — co-administer anti-androgen × 2-4 wks |
| Goserelin | GnRH agonist | SC implant (1, 3 month) | Similar to leuprolide |
| Degarelix | GnRH antagonist | SC monthly | No testosterone flare; injection site reactions |
| Relugolix | Oral GnRH antagonist | 120 mg PO daily | No flare; lower CV risk vs leuprolide (HERO trial); oral convenience |
| Enzalutamide | AR antagonist (2nd gen) | 160 mg PO daily | For mCRPC and mHSPC; seizure risk; drug interactions |
| Abiraterone | CYP17 inhibitor | 1000 mg PO daily + prednisone 5 mg BID | Blocks adrenal/intratumoral androgen synthesis; monitor LFTs, electrolytes, BP |
| Apalutamide | AR antagonist | 240 mg PO daily | For nmCRPC and mHSPC; rash common |
| Darolutamide | AR antagonist | 600 mg PO BID | For nmCRPC and mHSPC; minimal CNS side effects; low drug interaction risk |
Chemotherapy (Urologic Malignancies)
| Regimen | Indication | Key Toxicities |
|---|---|---|
| BEP (bleomycin, etoposide, cisplatin) | Metastatic GCT (testicular cancer) | Bleomycin pulmonary toxicity (limit total dose; PFTs); cisplatin nephrotoxicity, neuropathy, ototoxicity |
| Gemcitabine/Cisplatin (GC) | Metastatic urothelial carcinoma, neoadjuvant bladder cancer | Myelosuppression, nephrotoxicity; similar efficacy to MVAC with less toxicity |
| MVAC (methotrexate, vinblastine, adriamycin, cisplatin) | Metastatic urothelial carcinoma | Significant myelosuppression, mucositis; dose-dense MVAC with GCSF preferred |
| Docetaxel | mCRPC (first-line chemo), mHSPC | Neutropenia, neuropathy, fluid retention |
| Cabazitaxel | mCRPC (post-docetaxel) | Myelosuppression (neutropenia) |
Medical Expulsive Therapy & Stone Prevention
| Drug | Indication | Mechanism/Notes |
|---|---|---|
| Tamsulosin 0.4 mg daily | Distal ureteral stones 5-10 mm | Alpha-blocker; relaxes ureteral smooth muscle |
| Potassium citrate | Hypocitraturia, uric acid stones, RTA stones | Alkalinizes urine; binds calcium; increases citrate excretion |
| Hydrochlorothiazide 25-50 mg | Hypercalciuria (calcium stones) | Enhances renal calcium reabsorption |
| Allopurinol 300 mg daily | Hyperuricosuria, uric acid stones | Xanthine oxidase inhibitor; reduces uric acid production |
| Tiopronin | Cystine stones | Thiol agent — chelates cystine, increases solubility |
| Acetohydroxamic acid (AHA) | Struvite stones (adjunct) | Urease inhibitor; prevents struvite crystallization |
Erectile Dysfunction Medications
| Drug | Onset | Duration | Key Features |
|---|---|---|---|
| Sildenafil (Viagra) | 30-60 min | 4-6 hrs | Take on empty stomach; avoid with high-fat meals; original PDE5i |
| Tadalafil (Cialis) | 30-60 min | 24-36 hrs | Longest duration; 5 mg daily dosing for BPH + ED; food-independent |
| Vardenafil (Levitra) | 30-60 min | 4-6 hrs | Avoid with QT-prolonging drugs; ODT form available |
| Avanafil (Stendra) | 15-30 min | 6 hrs | Fastest onset; most selective PDE5i; fewer visual side effects |
All PDE5 inhibitors are absolutely contraindicated with nitrates (nitroglycerin, isosorbide mononitrate/dinitrate) — risk of severe, refractory hypotension. Relative caution with alpha-blockers (orthostatic hypotension). Common side effects: headache, flushing, dyspepsia, nasal congestion, visual disturbances (blue tinge with sildenafil — PDE6 cross-reactivity), and myalgias (tadalafil — PDE11 cross-reactivity).
Intracavernosal Injection Therapy
| Agent | Mechanism | Notes |
|---|---|---|
| Alprostadil (PGE1) | Direct smooth muscle relaxation via cAMP | FDA-approved; injection or intraurethral (MUSE); penile pain in ~30% |
| Trimix (PGE1 + phentolamine + papaverine) | Triple mechanism: cAMP, alpha-blockade, PDE inhibition | Most effective injectable; compounded; lower pain than alprostadil alone |
| Papaverine | Non-selective PDE inhibitor | Rarely used alone; risk of priapism and fibrosis |
25 Imaging & Diagnostics
CT Urogram
The CT urogram is the gold standard for evaluating hematuria and the upper urinary tract. It consists of three phases: non-contrast (detects stones, calcifications), nephrographic (enhancing renal masses), and delayed/excretory (opacifies the collecting system, ureters, and bladder — detects urothelial lesions). Sensitivity for ureteral stones ~97%; sensitivity for renal masses ~95%; sensitivity for UTUC ~96%.
Renal Ultrasound
Renal ultrasound is the first-line imaging for hydronephrosis, renal cysts (Bosniak I-II), pediatric evaluation, pregnant patients with suspected stones, and transplant kidney assessment. Advantages: no radiation, no contrast, portable, real-time. Limitations: operator-dependent, poor sensitivity for small ureteral stones (< 5 mm), cannot reliably characterize solid renal masses. Scrotal ultrasound with color Doppler is the first-line imaging for testicular masses and acute scrotum.
MRI Prostate (mpMRI)
Multiparametric MRI (mpMRI) of the prostate uses T2-weighted, diffusion-weighted (DWI/ADC), and dynamic contrast-enhanced (DCE) sequences. The PI-RADS v2.1 scoring system (1-5) standardizes reporting. mpMRI is used for: (1) pre-biopsy risk stratification — PI-RADS 1-2 has a high negative predictive value for clinically significant cancer (may avoid biopsy); (2) targeted biopsy guidance (MRI-TRUS fusion biopsy) for PI-RADS ≥ 3 lesions; (3) active surveillance monitoring; (4) local staging (extracapsular extension, SV invasion). The PRECISION trial showed that MRI-targeted biopsy detected more clinically significant cancers and fewer insignificant cancers compared to standard systematic biopsy.
Voiding Cystourethrogram (VCUG)
VCUG is the gold standard for diagnosing and grading VUR. Fluoroscopic imaging during bladder filling and voiding with contrast. Also evaluates: posterior urethral valves (dilated posterior urethra), bladder diverticula, and urethral abnormalities. In pediatric patients, a nuclear cystogram (radionuclide cystography) can be used for VUR surveillance with lower radiation dose.
Cystoscopy
Cystoscopy (flexible or rigid) allows direct visualization of the urethra and bladder. Indications: hematuria evaluation, bladder tumor surveillance, foreign body removal, stent placement/removal, and therapeutic interventions (TURBT, Botox injection). Flexible cystoscopy is performed in the office under local anesthesia (lidocaine gel). Blue-light (photodynamic) cystoscopy using hexaminolevulinate (HAL) improves detection of CIS and flat urothelial lesions compared to white-light cystoscopy.
Urodynamic Studies (UDS)
Urodynamics is the functional study of the lower urinary tract. Components: (1) Uroflowmetry — non-invasive measurement of flow rate; (2) Filling cystometry — measures intravesical pressure, compliance, capacity, and detects involuntary detrusor contractions during filling; (3) Pressure-flow study (voiding cystometry) — measures detrusor pressure during voiding to differentiate BOO from detrusor underactivity; (4) EMG — assesses pelvic floor/sphincter activity; (5) Video-urodynamics — combines UDS with fluoroscopy for anatomic correlation (gold standard for neurogenic bladder). Key parameters: detrusor pressure at Qmax (PdetQmax) > 40 cm H2O with Qmax < 12 mL/s = obstructed (Abrams-Griffiths nomogram).
Diuretic Renography (MAG3 Scan)
MAG3 diuretic renography uses Tc-99m-MAG3 radionuclide with furosemide administration to assess differential renal function and drainage. Key parameters: differential function (< 40% indicates significant impairment of the affected kidney), T½ (half-time of radionuclide washout after furosemide) — T½ < 10 min = non-obstructed, 10-20 min = indeterminate, > 20 min = obstructed. Primary indication: evaluation of UPJ obstruction and hydronephrosis to differentiate true obstruction from non-obstructive dilation.
PSMA PET-CT
PSMA PET-CT (prostate-specific membrane antigen positron emission tomography) uses radiolabeled tracers (Ga-68 PSMA-11 or F-18 piflufolastat/DCFPyL) that bind to PSMA expressed on prostate cancer cells. It has dramatically improved the detection of recurrent and metastatic prostate cancer, with sensitivity far exceeding conventional imaging (CT, bone scan, MRI). Indications: biochemical recurrence after definitive therapy (detects recurrence at PSA levels as low as 0.2 ng/mL), initial staging of high-risk or unfavorable intermediate-risk prostate cancer, and identification of PSMA-expressing tumors eligible for Lu-177-PSMA-617 radioligand therapy. PSMA PET-CT has been shown to change management in > 50% of patients with BCR.
Angiomyolipoma (AML)
Angiomyolipoma is the most common benign renal tumor. It contains fat, smooth muscle, and blood vessels. Classic AMLs are identified by the presence of macroscopic fat on CT (pathognomonic — HU < -10). Most are sporadic (80%, typically solitary, in women 40-60). Twenty percent occur in association with tuberous sclerosis complex (TSC) — bilateral, multiple, and presenting at a younger age. AMLs > 4 cm or symptomatic (hemorrhage) may require treatment: selective arterial embolization (first-line for acute hemorrhage), partial nephrectomy, or mTOR inhibitors (everolimus — for TSC-associated AMLs). Fat-poor AMLs can mimic RCC on imaging and may require biopsy or excision.
Renal Trauma
Renal trauma is classified by the AAST (American Association for the Surgery of Trauma) grading system:
| Grade | Description | Management |
|---|---|---|
| I | Subcapsular hematoma, non-expanding; contusion without laceration | Conservative: bed rest, serial hematocrit, imaging follow-up |
| II | Perirenal hematoma confined to retroperitoneum; cortical laceration < 1 cm depth, no collecting system involvement | Conservative |
| III | Cortical laceration > 1 cm depth, no collecting system involvement or urinary extravasation | Conservative; selective angioembolization if active bleeding |
| IV | Laceration into collecting system with urinary extravasation; renal artery/vein injury with contained hemorrhage; segmental infarction | Conservative if hemodynamically stable; ureteral stent for persistent urinoma; angioembolization for vascular injury |
| V | Shattered kidney; renal hilar avulsion; devascularized kidney | Surgical exploration; nephrectomy often required for hilar avulsion; attempt repair if possible |
Key principle: >95% of blunt renal injuries (Grades I-IV) can be managed non-operatively with observation, bed rest, and serial imaging. Indications for surgical exploration: hemodynamic instability not responding to resuscitation, expanding/pulsatile perirenal hematoma found during laparotomy, and Grade V hilar injury. Always attempt to confirm two functioning kidneys before performing nephrectomy (one-shot IVP or contralateral kidney palpation at surgery).
Varicocele
Varicocele is abnormal dilation of the pampiniform venous plexus within the spermatic cord, present in ~15% of the general male population and ~35-40% of men with primary infertility. Left-sided varicoceles are far more common (~90%) due to the longer course and perpendicular drainage of the left gonadal vein into the left renal vein. On examination, a varicocele feels like a "bag of worms" that distends with Valsalva and decompresses in the supine position. Grading: Grade I (palpable only with Valsalva), Grade II (palpable without Valsalva), Grade III (visible through scrotal skin).
Indications for varicocele repair: palpable varicocele + abnormal semen parameters + couple trying to conceive, or adolescent with testicular growth arrest (> 2 mL or 20% asymmetry). Repair options: microsurgical subinguinal varicocelectomy (gold standard — lowest recurrence ~1%, lowest hydrocele rate ~0.5%), inguinal varicocelectomy, laparoscopic varicocelectomy, and percutaneous embolization (interventional radiology). Semen parameters improve in ~60-70% of men after repair; spontaneous pregnancy rates increase by ~30-40%.
Male Infertility — Evaluation Overview
Male factor contributes to ~50% of couple infertility. Evaluation: semen analysis (× 2, 2-7 days abstinence) — WHO 2021 reference values: volume ≥ 1.4 mL, concentration ≥ 16 million/mL, total motility ≥ 42%, progressive motility ≥ 30%, normal morphology ≥ 4% (strict Kruger criteria). Azoospermia (no sperm in ejaculate) is classified as obstructive (normal FSH, normal testis volume — causes: vasectomy, ejaculatory duct obstruction, congenital bilateral absence of vas deferens/CBAVD associated with CFTR mutations) or non-obstructive (elevated FSH, small testes — causes: Klinefelter syndrome 47,XXY, Y-chromosome microdeletions, cryptorchidism, chemotherapy/radiation). Hormonal evaluation: FSH, LH, testosterone, prolactin. Genetic testing: karyotype and Y-microdeletion analysis for severe oligospermia (< 5 million/mL) or non-obstructive azoospermia.
Microsurgical testicular sperm extraction (micro-TESE) is the gold standard for sperm retrieval in non-obstructive azoospermia — identifies enlarged seminiferous tubules (more likely to contain spermatogenesis) under the operating microscope; sperm retrieval rate ~50%. Retrieved sperm are used for intracytoplasmic sperm injection (ICSI). Vasectomy reversal (vasovasostomy or vasoepididymostomy) has patency rates of 85-97% for vasovasostomy and 60-85% for vasoepididymostomy; success decreases with obstructive interval > 15 years.
26 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| ADT | Androgen deprivation therapy |
| AFP | Alpha-fetoprotein |
| AR | Androgen receptor |
| AUA | American Urological Association |
| AUS | Artificial urinary sphincter |
| BCG | Bacillus Calmette-Guérin |
| BEP | Bleomycin, etoposide, cisplatin |
| BMG | Buccal mucosal graft |
| BOO | Bladder outlet obstruction |
| BPH | Benign prostatic hyperplasia |
| CIC | Clean intermittent catheterization |
| CIS | Carcinoma in situ |
| CKD | Chronic kidney disease |
| CRPC | Castration-resistant prostate cancer |
| CT | Computed tomography |
| DCE | Dynamic contrast-enhanced (MRI) |
| DJ | Double-J (ureteral stent) |
| DLPP | Detrusor leak point pressure |
| DRE | Digital rectal examination |
| DSD | Detrusor-sphincter dyssynergia |
| DVIU | Direct vision internal urethrotomy |
| DWI | Diffusion-weighted imaging |
| EBRT | External beam radiation therapy |
| ED | Erectile dysfunction |
| EPA | Excision and primary anastomosis |
| GCT | Germ cell tumor |
| GFR | Glomerular filtration rate |
| GnRH | Gonadotropin-releasing hormone |
| hCG | Human chorionic gonadotropin |
| HoLEP | Holmium laser enucleation of prostate |
| HPF | High-power field |
| ICI | Immune checkpoint inhibitor |
| IFIS | Intraoperative floppy iris syndrome |
| IGCCCG | International Germ Cell Cancer Collaborative Group |
| IMRT | Intensity-modulated radiation therapy |
| IPSS | International Prostate Symptom Score |
| ISD | Intrinsic sphincter deficiency |
| ISUP | International Society of Urological Pathology |
| IVC | Inferior vena cava |
| LDH | Lactate dehydrogenase |
| LUTS | Lower urinary tract symptoms |
| MAP | Magnesium ammonium phosphate (struvite) |
| MET | Medical expulsive therapy |
| MIBC | Muscle-invasive bladder cancer |
| mpMRI | Multiparametric MRI |
| mRCC | Metastatic renal cell carcinoma |
| MVAC | Methotrexate, vinblastine, adriamycin, cisplatin |
| NMIBC | Non-muscle-invasive bladder cancer |
| NSGCT | Nonseminomatous germ cell tumor |
| NVB | Neurovascular bundle |
| OAB | Overactive bladder |
| PCNL | Percutaneous nephrolithotomy |
| PCN | Percutaneous nephrostomy |
| PDE5 | Phosphodiesterase type 5 |
| PFMT | Pelvic floor muscle training |
| PI-RADS | Prostate Imaging — Reporting and Data System |
| PSA | Prostate-specific antigen |
| PTNS | Percutaneous tibial nerve stimulation |
| PUV | Posterior urethral valves |
| PVR | Post-void residual |
| RAAS | Renin-angiotensin-aldosterone system |
| RARP | Robot-assisted radical prostatectomy |
| RCC | Renal cell carcinoma |
| RNU | Radical nephroureterectomy |
| RP | Radical prostatectomy |
| RPLND | Retroperitoneal lymph node dissection |
| RTA | Renal tubular acidosis |
| RUG | Retrograde urethrogram |
| SBRT | Stereotactic body radiation therapy |
| SNM | Sacral neuromodulation |
| SUI | Stress urinary incontinence |
| SWL | Shock wave lithotripsy |
| TCC | Transitional cell carcinoma |
| TIP | Tubularized incised plate |
| TKI | Tyrosine kinase inhibitor |
| TNM | Tumor-Node-Metastasis (staging) |
| TURBT | Transurethral resection of bladder tumor |
| TURP | Transurethral resection of prostate |
| UDS | Urodynamic studies |
| UPJ | Ureteropelvic junction |
| URS | Ureteroscopy |
| UTUC | Upper tract urothelial carcinoma |
| UUI | Urgency urinary incontinence |
| UVJ | Ureterovesical junction |
| VCUG | Voiding cystourethrogram |
| VHL | Von Hippel-Lindau |
| VUR | Vesicoureteral reflux |
| 5-ARI | 5-alpha reductase inhibitor |
| AD | Autonomic dysreflexia |
| ADC | Apparent diffusion coefficient (MRI) |
| AML | Angiomyolipoma |
| BRCA | Breast cancer gene |
| CAUTI | Catheter-associated urinary tract infection |
| CBAVD | Congenital bilateral absence of vas deferens |
| CFTR | Cystic fibrosis transmembrane conductance regulator |
| CT | Computed tomography |
| DSD | Disorders of sex development / Detrusor-sphincter dyssynergia |
| GCNIS | Germ cell neoplasia in situ |
| HRR | Homologous recombination repair |
| ICSI | Intracytoplasmic sperm injection |
| IMDC | International Metastatic RCC Database Consortium |
| LVI | Lymphovascular invasion |
| MAG3 | Mercaptoacetyltriglycine (renal scan) |
| mHSPC | Metastatic hormone-sensitive prostate cancer |
| mTOR | Mechanistic target of rapamycin |
| MUS | Midurethral sling |
| PARP | Poly (ADP-ribose) polymerase |
| PDE5i | Phosphodiesterase type 5 inhibitor |
| PSMA | Prostate-specific membrane antigen |
| PUL | Prostatic urethral lift (UroLift) |
| PVP | Photoselective vaporization of prostate |
| RENAL | Radius, Exophytic, Nearness, Anterior/posterior, Location (nephrometry) |
| SCI | Spinal cord injury |
| SRM | Small renal mass |
| SRT | Salvage radiation therapy |
| TESE | Testicular sperm extraction |
| TSC | Tuberous sclerosis complex |
| TWOC | Trial without catheter |
| UAS | Ureteral access sheath |
| VAC | Vacuum-assisted closure (wound therapy) |
| VEGF | Vascular endothelial growth factor |
| WHO | World Health Organization |
Landmark Trials in Urology
| Trial | Topic | Key Finding |
|---|---|---|
| ProtecT (2016) | Localized prostate cancer | No difference in prostate-cancer-specific mortality at 10 years between active monitoring, radical prostatectomy, and EBRT |
| PRECISION (2018) | Prostate biopsy | MRI-targeted biopsy detected more clinically significant cancers and fewer insignificant cancers than systematic biopsy |
| CHAARTED (2015) | mHSPC | ADT + docetaxel improved OS by 13.6 months vs ADT alone in high-volume mHSPC |
| LATITUDE (2017) | mHSPC | ADT + abiraterone/prednisone significantly improved OS in newly diagnosed high-risk mHSPC |
| CheckMate 214 (2018) | Metastatic RCC | Ipilimumab + nivolumab improved OS vs sunitinib in intermediate/poor-risk mRCC |
| KEYNOTE-426 (2019) | Metastatic RCC | Pembrolizumab + axitinib improved OS and PFS vs sunitinib across all IMDC risk groups |
| MTOPS (2003) | BPH | Combination doxazosin + finasteride superior to monotherapy in reducing BPH clinical progression |
| CombAT (2010) | BPH | Dutasteride + tamsulosin superior to monotherapy in men with enlarged prostates (> 30 mL) |
| RIVUR (2014) | Pediatric VUR | Antibiotic prophylaxis reduced UTI recurrence by ~50% in children with VUR but did not prevent scarring |
| SWOG 8794 (2009) | Adjuvant RT after RP | Adjuvant RT after RP for pT3 prostate cancer improved biochemical recurrence-free survival |
| EV-302/KEYNOTE-A39 (2023) | Metastatic urothelial | Enfortumab vedotin + pembrolizumab became new standard first-line for metastatic urothelial carcinoma |
| HERO (2020) | ADT for prostate cancer | Oral relugolix achieved testosterone suppression with lower cardiovascular event rate vs leuprolide |
| CARMENA (2018) | Cytoreductive nephrectomy in mRCC | Sunitinib alone was non-inferior to cytoreductive nephrectomy + sunitinib in IMDC intermediate/poor-risk mRCC |
| JAVELIN Bladder 100 (2020) | Maintenance immunotherapy in urothelial carcinoma | Avelumab maintenance after platinum-based chemo improved OS in advanced urothelial carcinoma |
Urologic Emergencies — Quick Reference
| Emergency | Key Time Window | Immediate Action |
|---|---|---|
| Testicular torsion | Salvage within 6 hours | Emergent scrotal exploration, detorsion, bilateral orchiopexy |
| Fournier gangrene | Hours (mortality rises with delay) | Broad-spectrum IV antibiotics + emergent surgical debridement |
| Ischemic priapism | Intervene within 4-6 hours | Corporal aspiration + phenylephrine injection |
| Paraphimosis | Urgent (risk of ischemic necrosis) | Manual reduction; dorsal slit if unsuccessful |
| Infected obstructed kidney | Urgent (sepsis risk) | IV antibiotics + ureteral stent or percutaneous nephrostomy |
| Autonomic dysreflexia | Minutes (hypertensive crisis) | Sit up, identify trigger (usually bladder distension), catheterize, antihypertensives |
| Bilateral ureteral obstruction | Urgent (renal failure) | Emergent decompression (stents or nephrostomy tubes bilaterally) |
| Renal trauma Grade V | Immediate (hemorrhage) | Surgical exploration if hemodynamically unstable; angioembolization if stable |