Addiction Medicine
Alcohol, opioid, stimulant, sedative, cannabis, and tobacco use disorders, withdrawal syndromes, medication-assisted treatment, motivational interviewing, harm reduction, drug screening, and every pharmacotherapy protocol, scoring system, and management algorithm across the full scope of addiction medicine.
01 Neurobiology of Addiction
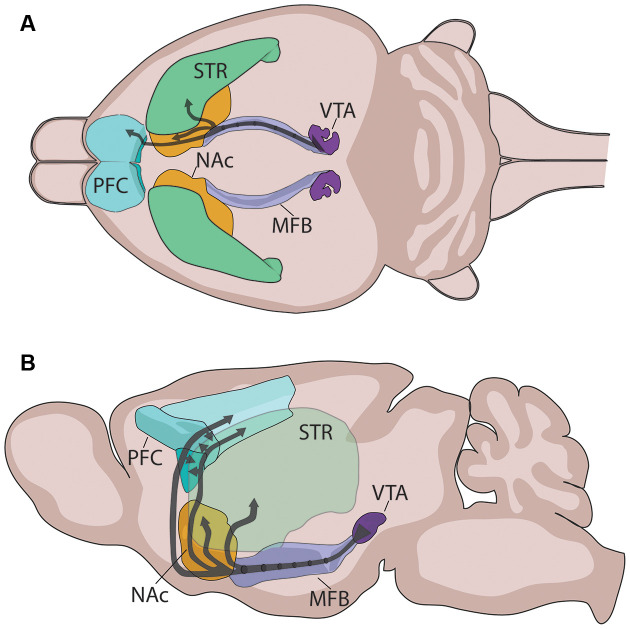
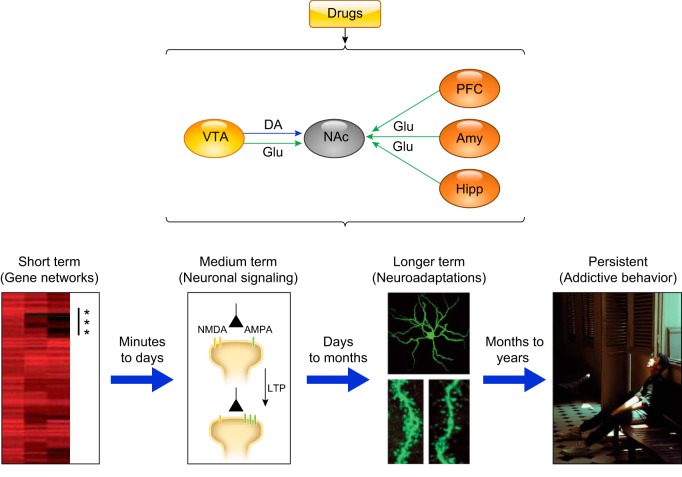
Addiction is a chronic, relapsing brain disorder characterized by compulsive substance seeking and use despite harmful consequences. The neurobiology centers on the mesolimbic dopamine pathway — a circuit linking the ventral tegmental area (VTA) in the midbrain to the nucleus accumbens (NAc) in the ventral striatum, with projections to the prefrontal cortex (PFC), amygdala, and hippocampus. Natural rewards (food, sex, social bonding) increase dopamine release in the NAc by 50–100%; drugs of abuse hijack this system with supraphysiologic dopamine surges — cocaine increases synaptic dopamine 300–400%, methamphetamine up to 1,200%.
Reward Circuitry & Key Structures
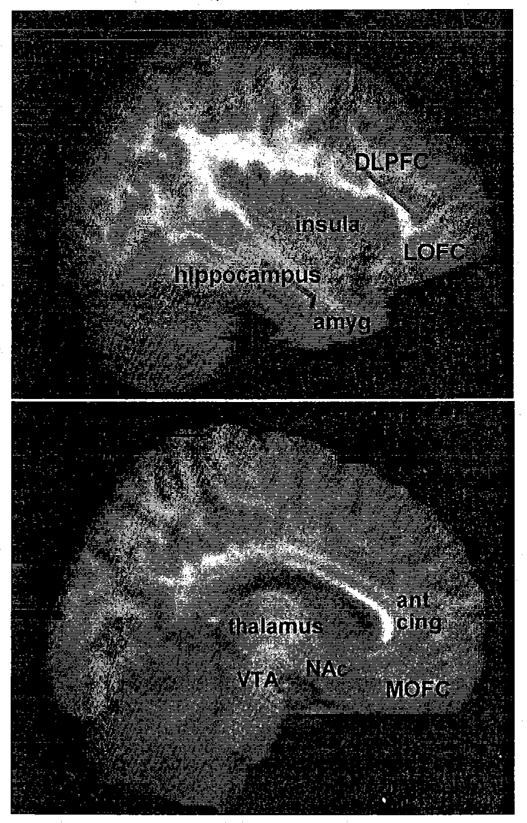
The VTA contains dopaminergic neurons that project to the NAc (mesolimbic pathway) and PFC (mesocortical pathway). The nucleus accumbens integrates motivational salience — it assigns "wanting" (incentive salience) to stimuli, distinct from "liking" (hedonic pleasure). The prefrontal cortex mediates executive function, impulse control, and decision-making; chronic substance use produces PFC hypofrontality, impairing the ability to inhibit compulsive drug-seeking. The amygdala (extended amygdala including the bed nucleus of the stria terminalis) encodes negative emotional states during withdrawal, driving the "dark side" of addiction. The hippocampus consolidates contextual memories linking environmental cues to drug reward, forming the neurobiological basis of triggers and cue-induced craving.
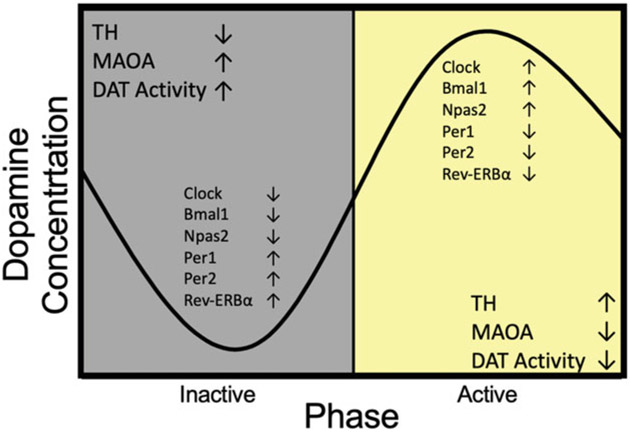
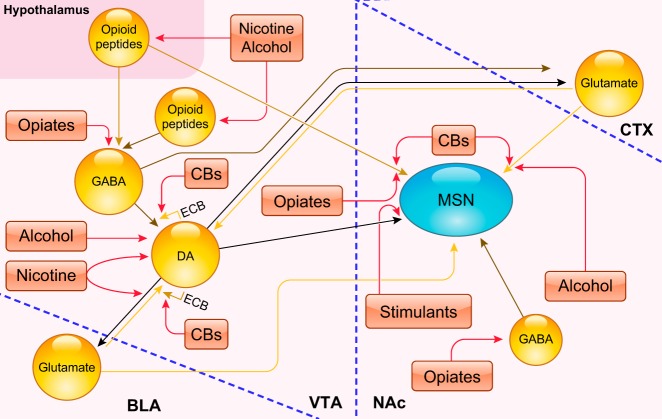
Neurotransmitter Systems
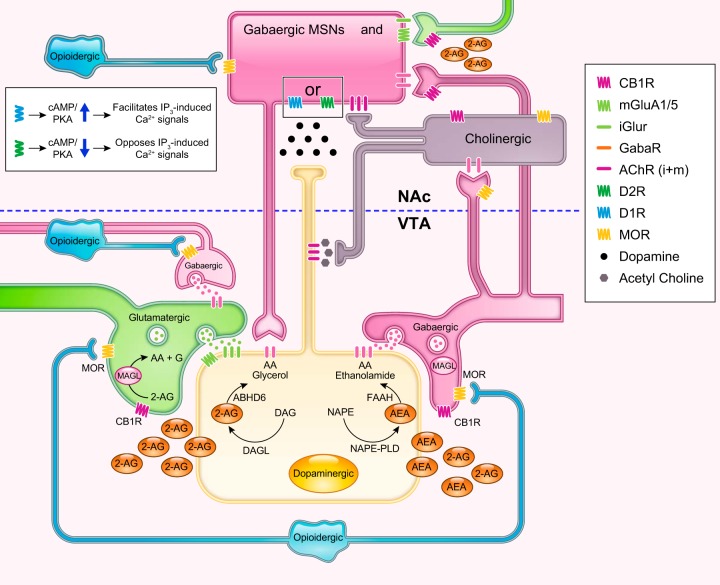
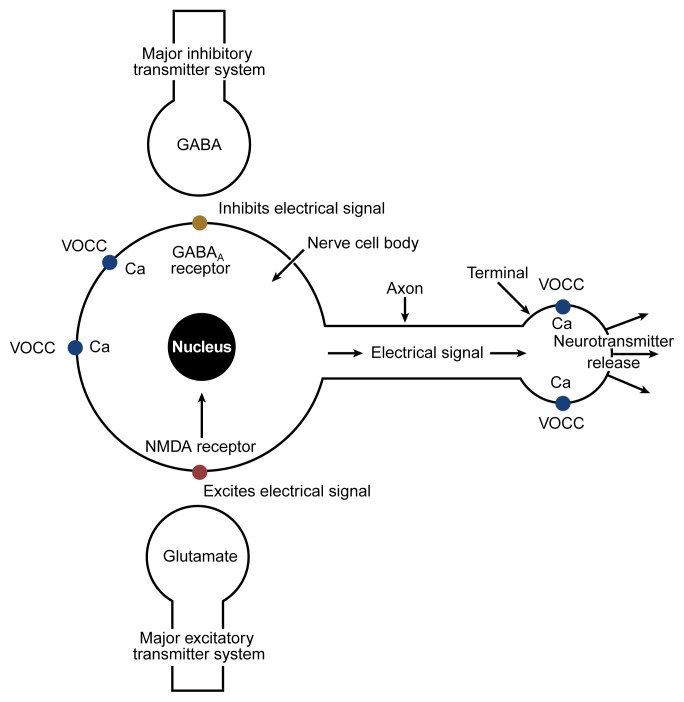
Dopamine is central but not the only neurotransmitter involved. GABA (the principal inhibitory neurotransmitter) is the target of alcohol, benzodiazepines, and barbiturates — they enhance GABA-A receptor chloride conductance, producing sedation and anxiolysis. Glutamate (the principal excitatory neurotransmitter, acting on NMDA and AMPA receptors) is suppressed by alcohol and upregulated during chronic use — withdrawal unmasks excitotoxicity, producing seizures and delirium tremens. Endogenous opioids (endorphins, enkephalins, dynorphins acting on mu, kappa, and delta receptors) modulate pain, reward, and stress. Endocannabinoids (anandamide, 2-AG) act as retrograde messengers at CB1 receptors throughout the reward circuitry. Serotonin (5-HT) modulates mood, impulsivity, and the subjective effects of MDMA, psychedelics, and alcohol.
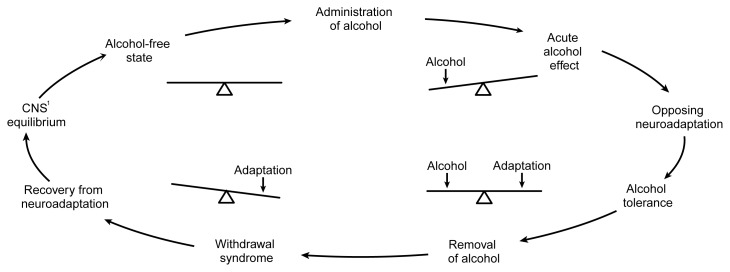
Neuroadaptation: Tolerance, Sensitization & Kindling
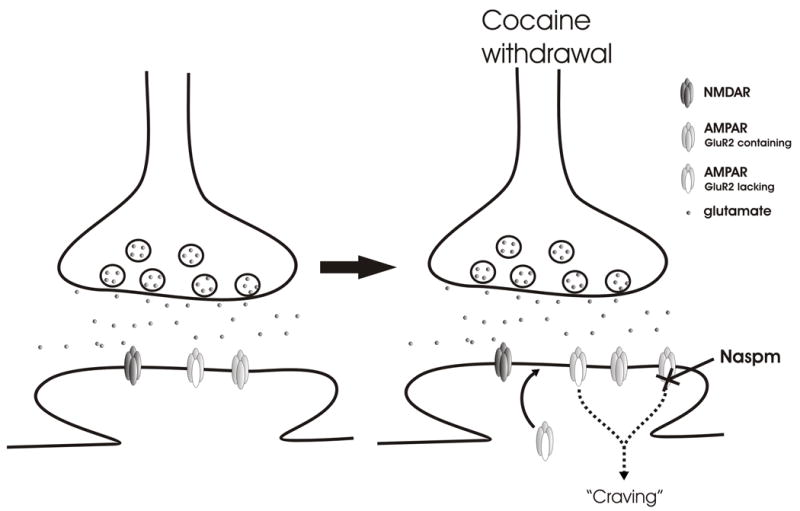
Tolerance is the need for increasing doses to achieve the same effect, resulting from receptor downregulation (pharmacodynamic tolerance) and increased hepatic metabolism (pharmacokinetic tolerance). Cross-tolerance occurs between drugs acting on the same receptor system (e.g., alcohol and benzodiazepines at GABA-A). Sensitization (reverse tolerance) is the progressive amplification of a drug response with repeated exposure — particularly relevant to stimulants, where repeated administration produces escalating locomotor activation and stereotyped behaviors. Kindling describes the phenomenon where repeated withdrawal episodes become progressively more severe; each alcohol withdrawal episode lowers the seizure threshold for subsequent episodes, making untreated withdrawal increasingly dangerous. This is mediated by NMDA receptor upregulation and glutamatergic excitotoxicity.
Genetic Factors
Heritability of substance use disorders is estimated at 40–60%. Key genetic variants include: OPRM1 A118G (mu-opioid receptor polymorphism) — the G allele is associated with greater opioid binding affinity, increased alcohol reward, and better response to naltrexone treatment. ADH1B*2 and ALDH2*2 — the ALDH2*2 variant (common in East Asian populations, ~30–50% prevalence) produces a nonfunctional aldehyde dehydrogenase, causing acetaldehyde accumulation after alcohol ingestion (flushing, nausea, tachycardia) and conferring a strong protective effect against AUD. CHRNA5-A3-B4 gene cluster on chromosome 15q25 encodes nicotinic acetylcholine receptor subunits and influences nicotine dependence severity and lung cancer risk. GABRA2 polymorphisms are associated with alcohol dependence and altered neural response to alcohol cues. DRD2 Taq1A (dopamine D2 receptor) — the A1 allele reduces D2 receptor density and has been inconsistently associated with SUD vulnerability.
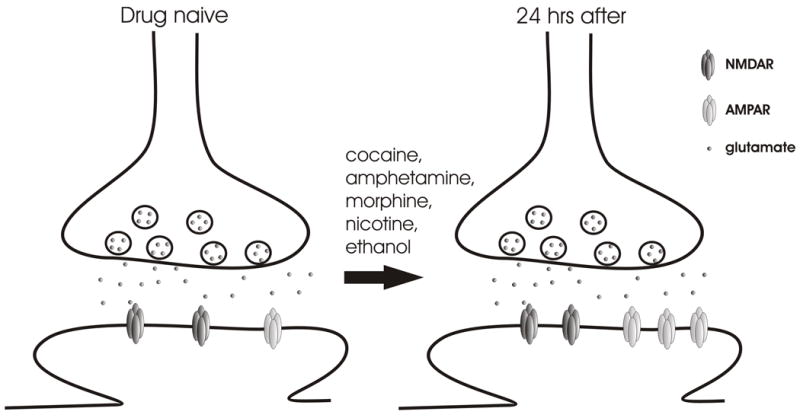
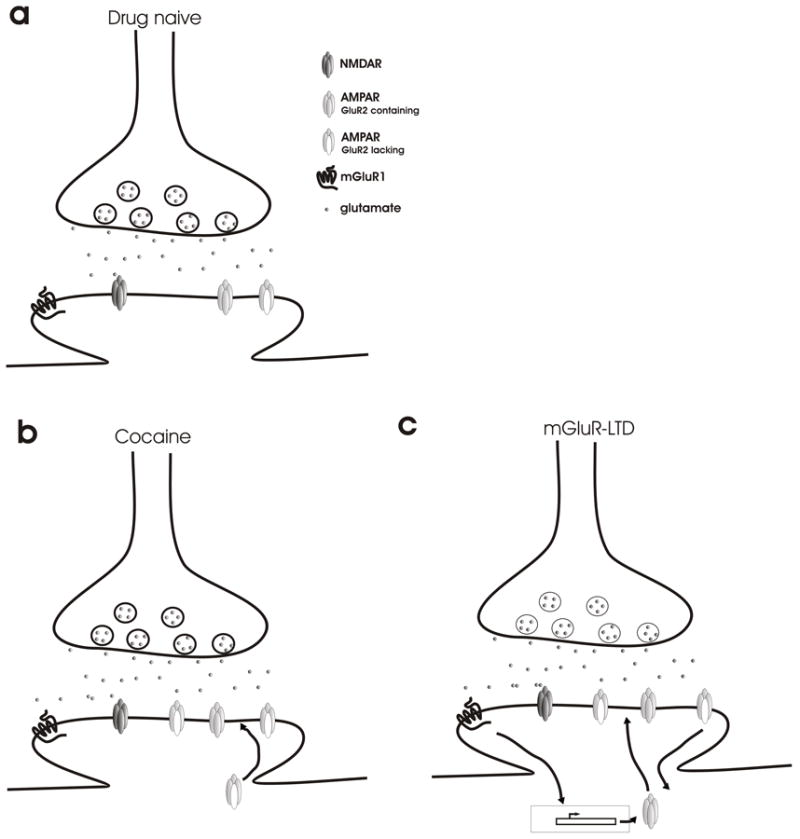
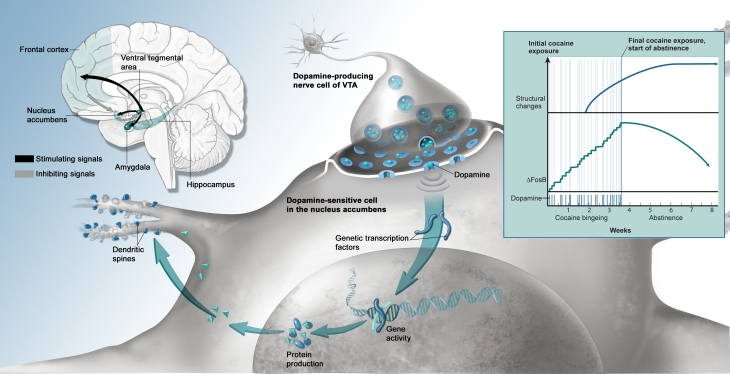
Chronic substance use induces long-term potentiation (LTP) at glutamatergic synapses onto VTA dopamine neurons, strengthening drug-associated memories. Simultaneously, dopamine receptor density decreases in the NAc (particularly D2 receptors), producing anhedonia and driving escalation of use. These synaptic changes can persist for months to years after cessation, providing the neurobiological substrate for relapse. Functional MRI studies demonstrate that drug cues activate the same reward circuitry in abstinent individuals, often more intensely than natural rewards.
02 Screening & Assessment
Universal screening for substance use disorders is recommended by USPSTF (Grade B for alcohol in adults; Grade I for drugs). The SBIRT framework (Screening, Brief Intervention, Referral to Treatment) is the evidence-based model for integrating screening into primary care, emergency departments, and hospital settings.
CAGE Questionnaire
A 4-item screening tool for alcohol use disorder. Each "yes" scores 1 point. Score ≥2 is considered positive (sensitivity 60–95%, specificity 40–95% depending on population). Limitations: poor sensitivity in women, minorities, and binge drinkers; does not quantify consumption.
C — Have you ever felt you should Cut down on your drinking?
A — Have people Annoyed you by criticizing your drinking?
G — Have you ever felt bad or Guilty about your drinking?
E — Have you ever had a drink first thing in the morning to steady your nerves or get rid of a hangover (Eye-opener)?
AUDIT (Alcohol Use Disorders Identification Test)
A 10-item WHO-developed instrument covering consumption (items 1–3), dependence (items 4–6), and consequences (items 7–10). Each item scored 0–4 (total range 0–40). Scoring thresholds: 0–7 = low risk; 8–15 = hazardous use (brief intervention); 16–19 = harmful use (brief intervention + continued monitoring); ≥20 = possible dependence (referral for specialist evaluation). Sensitivity 80–97%, specificity 78–95% for AUD at the ≥8 threshold.
AUDIT — All 10 Items
| # | Question | Scoring |
|---|---|---|
| 1 | How often do you have a drink containing alcohol? | 0 = Never; 1 = Monthly or less; 2 = 2–4×/month; 3 = 2–3×/week; 4 = ≥4×/week |
| 2 | How many standard drinks on a typical drinking day? | 0 = 1–2; 1 = 3–4; 2 = 5–6; 3 = 7–9; 4 = ≥10 |
| 3 | How often do you have ≥6 drinks on one occasion? | 0 = Never; 1 = Less than monthly; 2 = Monthly; 3 = Weekly; 4 = Daily/almost daily |
| 4 | How often in the past year have you found you could not stop drinking once started? | 0 = Never; 1 = Less than monthly; 2 = Monthly; 3 = Weekly; 4 = Daily/almost daily |
| 5 | How often have you failed to do what was expected because of drinking? | Same scale as item 4 |
| 6 | How often have you needed a drink in the morning to get yourself going? | Same scale as item 4 |
| 7 | How often have you had guilt or remorse after drinking? | Same scale as item 4 |
| 8 | How often have you been unable to remember what happened while drinking? | Same scale as item 4 |
| 9 | Have you or someone else been injured because of your drinking? | 0 = No; 2 = Yes, but not in the past year; 4 = Yes, during the past year |
| 10 | Has a relative, friend, doctor, or health worker been concerned about your drinking? | 0 = No; 2 = Yes, but not in the past year; 4 = Yes, during the past year |
AUDIT-C (Abbreviated)
First 3 items of the AUDIT (consumption questions only). Score range 0–12. Positive screen: ≥4 for men, ≥3 for women. Widely used in primary care for rapid screening; comparable sensitivity to full AUDIT for identifying hazardous drinking.
DAST-10 (Drug Abuse Screening Test)
A 10-item yes/no questionnaire assessing drug use (excluding alcohol and tobacco) over the past 12 months. Scoring: 0 = no problems; 1–2 = low level; 3–5 = moderate level; 6–8 = substantial level; 9–10 = severe level. A score ≥3 warrants further assessment.
CRAFFT (Adolescent Screening)
A 6-item screening tool for substance use in adolescents aged 12–21 years. Part A asks about alcohol, cannabis, and other drug use in the past 12 months. Part B (the CRAFFT questions) is administered if any Part A answers are positive:
C — Have you ever ridden in a Car driven by someone (including yourself) who was high or had been using alcohol or drugs?
R — Do you ever use alcohol or drugs to Relax, feel better about yourself, or fit in?
A — Do you ever use alcohol or drugs while you are by yourself, or Alone?
F — Do you ever Forget things you did while using alcohol or drugs?
F — Do your Family or friends ever tell you that you should cut down on your drinking or drug use?
T — Have you ever gotten into Trouble while you were using alcohol or drugs?
Score ≥2 is a positive screen (sensitivity ~80%, specificity ~80% for SUD).
NIDA Quick Screen
Single-question prescreening: "In the past year, how many times have you used [alcohol/tobacco/prescription drugs for non-medical reasons/illegal drugs]?" Any use triggers the NIDA-Modified ASSIST for substance-specific risk assessment.
DSM-5 Substance Use Disorder Criteria
The DSM-5 eliminated the distinction between "abuse" and "dependence," replacing them with a single substance use disorder diagnosis graded by severity. The diagnosis requires ≥2 of 11 criteria within a 12-month period:
Impaired Control (Criteria 1–4):
1. Substance taken in larger amounts or over a longer period than intended
2. Persistent desire or unsuccessful efforts to cut down or control use
3. Great deal of time spent obtaining, using, or recovering from the substance
4. Craving, or strong desire/urge to use the substance
Social Impairment (Criteria 5–7):
5. Recurrent use resulting in failure to fulfill major role obligations (work, school, home)
6. Continued use despite persistent or recurrent social or interpersonal problems caused or exacerbated by the substance
7. Important social, occupational, or recreational activities given up or reduced because of use
Risky Use (Criteria 8–9):
8. Recurrent use in situations in which it is physically hazardous
9. Continued use despite knowledge of having a persistent or recurrent physical or psychological problem likely caused or exacerbated by the substance
Pharmacological (Criteria 10–11):
10. Tolerance (need for increased amounts or diminished effect at same dose)
11. Withdrawal (characteristic withdrawal syndrome or substance taken to relieve/avoid withdrawal)
Severity: Mild = 2–3 criteria; Moderate = 4–5 criteria; Severe = ≥6 criteria.
03 Motivational Interviewing & Stages of Change
Transtheoretical Model (Prochaska & DiClemente)
The Stages of Change model describes readiness for behavior change as a dynamic, non-linear process. Patients may cycle through stages multiple times before achieving sustained change. Relapse is considered a normal part of the change process, not a failure.
| Stage | Definition | Patient Statement | Clinician Approach |
|---|---|---|---|
| Precontemplation | No intention to change within the next 6 months; unaware or in denial of the problem | "I don't have a problem" | Raise awareness, provide information about risks, express concern without confrontation |
| Contemplation | Aware of the problem; considering change within the next 6 months; ambivalent | "Maybe I drink too much, but I'm not sure I'm ready to stop" | Explore ambivalence, elicit pros/cons, develop discrepancy between values and behavior |
| Preparation | Intends to take action within the next 30 days; may have already taken small steps | "I want to quit — I'm thinking about calling that program" | Help develop a concrete plan, identify barriers, build self-efficacy, connect to resources |
| Action | Actively modifying behavior; has made observable changes within the past 6 months | "I stopped drinking 3 weeks ago and started going to meetings" | Support changes, reinforce self-efficacy, problem-solve barriers, prevent relapse |
| Maintenance | Sustaining change for >6 months; working to prevent relapse | "I've been sober for 8 months now" | Reinforce gains, identify high-risk situations, develop coping strategies, long-term planning |
| Relapse | Return to prior behavior; reentry into an earlier stage | "I started drinking again after my divorce" | Normalize relapse as part of the process, re-engage without judgment, reassess and restart |
Motivational Interviewing (MI)
Motivational interviewing is a collaborative, person-centered counseling method developed by Miller and Rollnick that strengthens a person's own motivation and commitment to change. MI is not a technique to "get people to change" but a way of communicating that evokes the patient's own reasons for change. The spirit of MI rests on four elements: partnership (collaboration, not authority), acceptance (absolute worth, autonomy, affirmation, accurate empathy), compassion (prioritizing the patient's welfare), and evocation (drawing out the patient's own ideas and motivations).
OARS — Core MI Skills
O — Open-ended questions: Invite elaboration rather than yes/no answers. "Tell me about your relationship with alcohol" rather than "Do you drink too much?"
A — Affirmations: Recognize the patient's strengths and efforts. "It took a lot of courage to come here today."
R — Reflections: Mirror the patient's statements to demonstrate understanding and deepen exploration. Simple reflections repeat or rephrase; complex reflections add meaning, feeling, or emphasis. "It sounds like you're worried about what drinking is doing to your family."
S — Summaries: Collect and link statements the patient has made, especially change talk. "So on one hand, drinking helps you relax, and on the other, it's affecting your marriage and your health."
Key MI Principles
Express empathy: Use reflective listening to convey understanding. Ambivalence is normal, not pathological. Develop discrepancy: Help the patient see the gap between current behavior and stated values or goals. "You've said being a good father is the most important thing to you — how does your drinking fit with that?" Roll with resistance: Avoid arguing or confronting directly. Resistance is a signal to change approach, not to push harder. Reframe statements, emphasize autonomy. Support self-efficacy: Belief in one's ability to change is a key predictor of outcome. Highlight past successes and strengths.
Brief Interventions
Brief interventions are time-limited (5–30 minutes), structured counseling sessions typically delivered in primary care or ED settings. The FRAMES model summarizes effective elements: Feedback (personalized information about risk), Responsibility (emphasize that change is the patient's choice), Advice (clear, specific recommendation to reduce or stop use), Menu (offer a range of options), Empathy (warm, reflective, non-judgmental style), Self-efficacy (support confidence in ability to change). Evidence shows that a single brief intervention in primary care can reduce heavy drinking by 10–30% over 6–12 months.
Change Talk & Sustain Talk
Change talk is any patient speech that favors movement toward change. It can be categorized using the mnemonic DARN-CAT: Desire ("I want to quit"), Ability ("I could cut back"), Reasons ("My health would improve"), Need ("I have to stop before I lose my job"), Commitment ("I will call the program tomorrow"), Activation ("I'm ready to make a change"), Taking steps ("I poured out the bottles last night"). Preparatory change talk (DARN) signals readiness; mobilizing change talk (CAT) predicts actual behavior change. The clinician's goal is to evoke and reinforce change talk while not amplifying sustain talk (arguments against change). When the patient makes a change talk statement, the clinician should reflect it, affirm it, and ask for elaboration.
Assessing Readiness: Readiness Rulers
A practical MI tool: ask the patient to rate on a scale of 1–10 "How important is it for you to make this change?" and "How confident are you that you could make this change if you decided to?" Then ask: "Why did you choose a [number] and not a lower number?" This evokes change talk by asking the patient to argue for the side of change. If the patient scores low (≤3), the focus is on importance building; if importance is high but confidence is low, the focus shifts to self-efficacy enhancement and practical planning.
04 Key Terminology & Abbreviations
Core definitions used throughout addiction medicine. These terms have specific clinical meanings that differ from colloquial usage.
| Term | Definition |
|---|---|
| Addiction | Chronic brain disorder characterized by compulsive substance use despite harm, loss of control, and craving; preferred term in clinical/research settings is "substance use disorder" |
| Physical dependence | Physiologic adaptation to a substance manifested by tolerance and/or withdrawal upon cessation; occurs with many prescribed medications and does NOT equate to addiction |
| Tolerance | Need for increasing doses to achieve the same effect, or diminished effect at the same dose |
| Withdrawal | Characteristic syndrome upon cessation or dose reduction of a substance to which physiologic dependence has developed |
| Craving | Intense urge or desire to use a substance; added as DSM-5 criterion (criterion 4) |
| Intoxication | Reversible syndrome of behavioral/psychological changes due to recent substance exposure |
| Remission | Early remission: 3–12 months without meeting any SUD criteria (except craving). Sustained remission: ≥12 months without criteria (except craving) |
| Harm reduction | Strategies that aim to reduce negative consequences of substance use without necessarily requiring abstinence |
| MOUD | Medications for opioid use disorder (preferred term over MAT because psychotherapy is the "treatment," not just the medication) |
| Agonist therapy | Treatment using a medication that activates the same receptor as the drug of abuse (e.g., methadone, buprenorphine for OUD; NRT for nicotine) |
| Antagonist therapy | Treatment using a medication that blocks the receptor, preventing the effect of the drug (e.g., naltrexone for OUD/AUD) |
| Polysubstance use | Concurrent use of multiple substances; the norm rather than the exception in most clinical populations |
| Drug-drug interaction | Pharmacokinetic or pharmacodynamic interaction between substances (e.g., opioids + benzodiazepines = synergistic respiratory depression) |
| Contingency management | Behavioral reinforcement strategy providing tangible incentives (vouchers, prizes) contingent on objectively verified positive behaviors |
| Precipitated withdrawal | Acute, severe withdrawal triggered by administration of an antagonist or partial agonist (e.g., naloxone or buprenorphine) to a physically dependent patient |
| Ceiling effect | Maximum pharmacologic effect achieved beyond which increasing dose produces no additional effect (e.g., buprenorphine respiratory depression plateaus at ~24–32 mg SL) |
| Cross-tolerance | Tolerance to one substance conferring tolerance to another acting on the same receptor system (e.g., alcohol and benzodiazepines at GABA-A) |
| Kindling | Progressive worsening of withdrawal severity with each successive withdrawal episode, mediated by neuroplastic changes |
| Protracted withdrawal / PAWS | Post-acute withdrawal syndrome: persistent sub-acute symptoms (dysphoria, insomnia, anxiety, cognitive impairment) lasting weeks to months after acute withdrawal resolves; a major driver of relapse |
Substances ranked by all-cause mortality risk: Tobacco > Alcohol > Opioids > Methamphetamine > Cocaine > Sedatives > Cannabis. Tobacco kills more people than all other substances combined, yet receives the least clinical attention. Opioids have the highest acute overdose mortality rate per user. Alcohol has the broadest organ system damage and the most widespread social harm. Cannabis has the lowest direct mortality risk but is not harmless (impaired driving, psychiatric effects).
05 AUD — Diagnosis & Medical Complications
Alcohol use disorder affects approximately 29 million Americans (10.5% of adults). It is the third leading preventable cause of death in the United States (~95,000 deaths/year). The DSM-5 SUD criteria (Section 2) are applied specifically to alcohol. Standard drink definition: 14 g pure ethanol = 12 oz beer (5%), 5 oz wine (12%), or 1.5 oz spirits (40%).
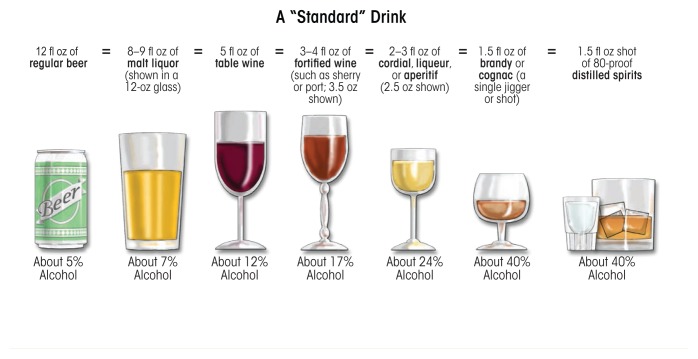
NIAAA Drinking Thresholds
| Category | Men | Women |
|---|---|---|
| Low-risk drinking | ≤14 drinks/week AND ≤4/occasion | ≤7 drinks/week AND ≤3/occasion |
| Binge drinking | ≥5 drinks within 2 hours | ≥4 drinks within 2 hours |
| Heavy drinking | ≥5 drinks/day on ≥5 days/month | ≥4 drinks/day on ≥5 days/month |
Biomarkers for Alcohol Use
GGT (gamma-glutamyl transferase): Elevated in 75% of heavy drinkers; non-specific (also elevated with hepatobiliary disease, medications, obesity). Half-life ~26 days; normalizes in 2–4 weeks of abstinence. CDT (carbohydrate-deficient transferrin): More specific than GGT for heavy alcohol use (≥60 g/day for ≥2 weeks); sensitivity 60–80%, specificity 80–95%. MCV (mean corpuscular volume): Macrocytosis (MCV >100 fL) is common in chronic AUD due to direct toxic effect of ethanol on erythropoiesis and folate deficiency; non-specific. AST:ALT ratio >2:1 is suggestive of alcoholic liver disease (alcohol induces AST via mitochondrial damage and depletes pyridoxal phosphate needed for ALT synthesis). EtG (ethyl glucuronide) and EtS (ethyl sulfate) are direct ethanol metabolites detectable in urine for 2–3 days after consumption; highly sensitive but can be positive from incidental alcohol exposure (hand sanitizer, mouthwash).
Medical Complications of Chronic Alcohol Use
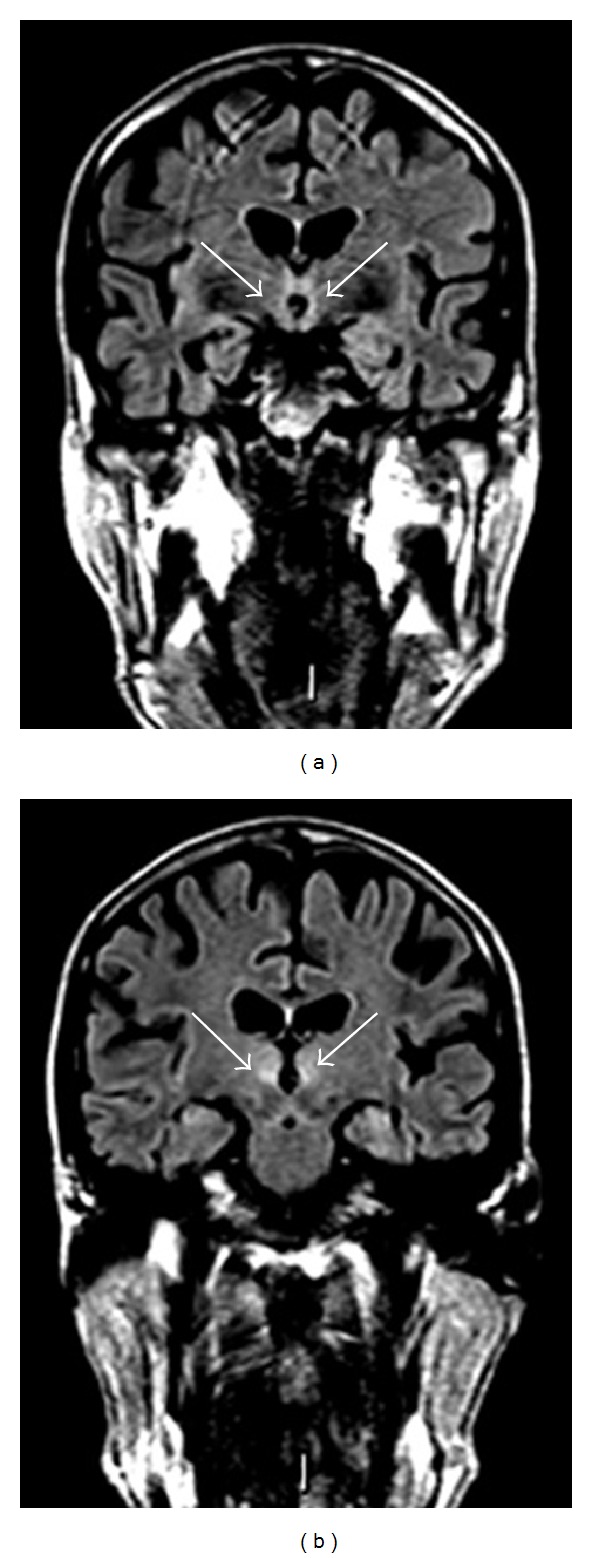
Wernicke encephalopathy: Acute thiamine (B1) deficiency — classic triad of confusion, ophthalmoplegia (typically lateral rectus palsy/nystagmus), and ataxia. Only 10–16% of patients present with all three findings. Treat emergently with IV thiamine 500 mg TID for 3–5 days before giving glucose (glucose metabolism consumes thiamine and can precipitate or worsen Wernicke). Korsakoff syndrome: Chronic/irreversible sequela of untreated Wernicke — anterograde amnesia, confabulation, apathy. Only ~20% fully recover with treatment.
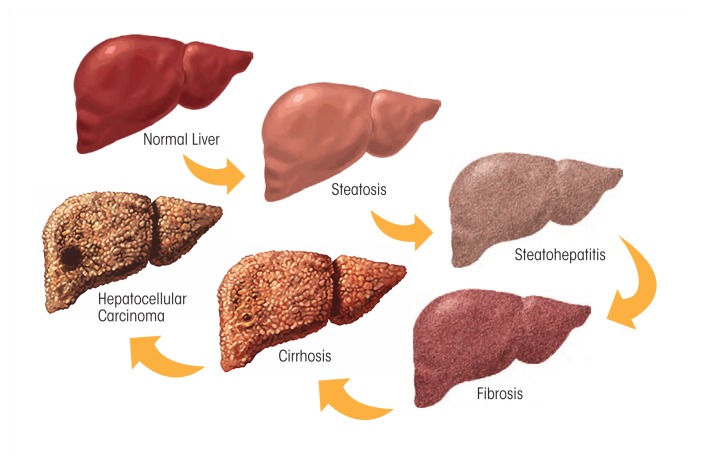
Alcoholic hepatitis: Presents with jaundice, hepatomegaly, fever, leukocytosis, AST >ALT (both typically <300). Maddrey discriminant function (MDF) = 4.6 × (patient PT − control PT) + total bilirubin; MDF ≥32 indicates severe disease (28-day mortality ~35%) and is an indication for prednisolone 40 mg/day × 28 days (if no contraindications: GI bleeding, sepsis, hepatorenal syndrome). MELD score ≥21 is an alternative severity threshold. Alcoholic cirrhosis: End-stage liver disease; portal hypertension leading to varices, ascites, hepatic encephalopathy, hepatorenal and hepatopulmonary syndromes. Abstinence is the single most important intervention and can reverse fibrosis if pre-cirrhotic.
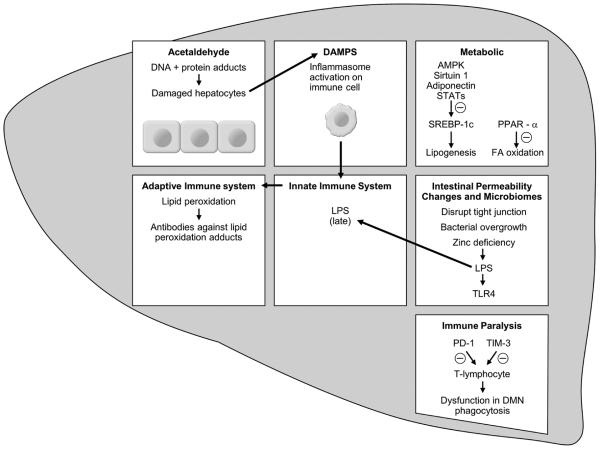
Alcoholic cardiomyopathy: Dilated cardiomyopathy with reduced EF from chronic heavy drinking (typically >7–8 drinks/day for ≥5 years); can improve significantly with abstinence. Cerebellar degeneration: Truncal ataxia, wide-based gait from Purkinje cell loss (superior vermis most affected). Peripheral neuropathy: Symmetric, distal, sensory-predominant "stocking-glove" pattern from direct toxic effect and thiamine/B12/folate deficiency. Pancreatitis: Alcohol is the most common cause of chronic pancreatitis and the second most common cause of acute pancreatitis (after gallstones). Macrocytosis: Direct toxic effect on erythroid precursors even without folate/B12 deficiency. Increased cancer risk: Oropharyngeal, esophageal (squamous), hepatocellular, breast, colorectal — alcohol is classified as a Group 1 carcinogen by IARC.
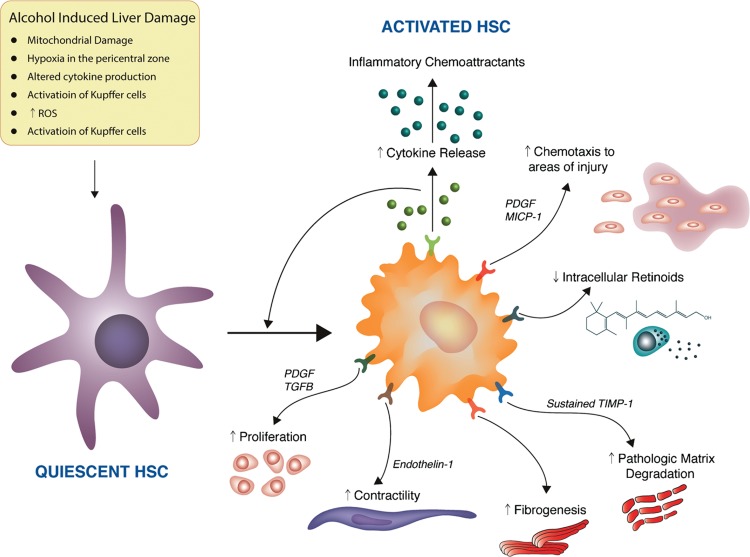
06 Alcohol Withdrawal — CIWA Protocol & Management
Alcohol withdrawal results from abrupt cessation or reduction of heavy, prolonged alcohol use. Chronic alcohol exposure upregulates excitatory NMDA glutamate receptors and downregulates inhibitory GABA-A receptors. Upon cessation, unopposed glutamatergic excitation produces the withdrawal syndrome. Onset is typically 6–24 hours after last drink, with peak severity at 24–72 hours.
Timeline of Alcohol Withdrawal
| Time After Last Drink | Syndrome | Features |
|---|---|---|
| 6–12 hours | Minor withdrawal / tremulousness | Anxiety, insomnia, tremor, diaphoresis, palpitations, GI upset, mild hypertension/tachycardia |
| 12–24 hours | Alcoholic hallucinosis | Visual, auditory, or tactile hallucinations with intact sensorium (patient knows hallucinations are not real); resolves in 24–48 hours |
| 12–48 hours | Withdrawal seizures | Generalized tonic-clonic seizures, typically brief and self-limited; may occur in clusters; 3% risk overall, 30% risk if prior withdrawal seizures |
| 48–96 hours | Delirium tremens (DT) | Severe agitation, global confusion, disorientation, hallucinations, profound autonomic instability (tachycardia, hypertension, hyperthermia, diaphoresis); mortality 5–15% untreated, ~1% with appropriate treatment |
CIWA-Ar Scale (Clinical Institute Withdrawal Assessment for Alcohol, Revised)
The CIWA-Ar is a 10-item assessment tool used to quantify the severity of alcohol withdrawal and guide pharmacotherapy. Each item is scored by the clinician based on observation and patient interview. Maximum score = 67.
1. Nausea/vomiting (0–7): 0 = no nausea; 1 = mild nausea, no vomiting; 4 = intermittent nausea with dry heaves; 7 = constant nausea, frequent dry heaves/vomiting
2. Tremor (0–7): Arms extended, fingers spread. 0 = no tremor; 1 = not visible but felt fingertip to fingertip; 4 = moderate, with arms extended; 7 = severe, even with arms not extended
3. Paroxysmal sweats (0–7): 0 = no sweat visible; 1 = barely perceptible sweating, palms moist; 4 = beads of sweat on forehead; 7 = drenching sweats
4. Anxiety (0–7): 0 = no anxiety, at ease; 1 = mildly anxious; 4 = moderately anxious or guarded; 7 = equivalent to acute panic states
5. Agitation (0–7): 0 = normal activity; 1 = somewhat more than normal activity; 4 = moderately fidgety, restless; 7 = paces back and forth, constant thrashing
6. Tactile disturbances (0–7): 0 = none; 1 = itching, pins and needles, burning, numbness; 2 = mild itching/burning/numbness; 3 = moderate; 4 = moderately severe hallucinations; 5 = severe; 6 = extremely severe; 7 = continuous hallucinations
7. Auditory disturbances (0–7): 0 = not present; 1 = very mild harshness or frightening quality; 2 = mild; 3 = moderate; 4 = moderately severe hallucinations; 5 = severe; 6 = extremely severe; 7 = continuous hallucinations
8. Visual disturbances (0–7): 0 = not present; 1 = very mild sensitivity; 2 = mild sensitivity; 3 = moderate sensitivity; 4 = moderately severe hallucinations; 5 = severe; 6 = extremely severe; 7 = continuous hallucinations
9. Headache, fullness in head (0–7): 0 = not present; 1 = very mild; 2 = mild; 3 = moderate; 4 = moderately severe; 5 = severe; 6 = very severe; 7 = extremely severe
10. Orientation/clouding of sensorium (0–4): 0 = oriented, can do serial additions; 1 = uncertain about date; 2 = date uncertain by >2 days; 3 = disoriented for date by >2 calendar days; 4 = disoriented for place and/or person
CIWA-Ar Scoring Interpretation & Management
| CIWA-Ar Score | Severity | Management |
|---|---|---|
| <10 | Minimal withdrawal | Supportive care, reassess every 4–8 hours, consider no benzodiazepine needed |
| 10–18 | Moderate withdrawal | Symptom-triggered benzodiazepine dosing; reassess every 1–2 hours |
| >18 | Severe withdrawal | Aggressive benzodiazepine dosing, consider ICU monitoring, reassess every 30–60 minutes |
| >35 | Very severe / impending DT | ICU admission, IV benzodiazepines, consider phenobarbital or adjuncts, 1:1 nursing |
Benzodiazepine Protocols
Symptom-triggered dosing (preferred): Administer benzodiazepine only when CIWA-Ar ≥10. Results in lower total benzodiazepine dose and shorter treatment duration compared to fixed-schedule dosing. Fixed-schedule dosing (for patients unable to participate in CIWA assessment, e.g., intubated, severe cognitive impairment): Chlordiazepoxide 50–100 mg q6h with taper over 3–5 days.
| Benzodiazepine | Dose | Half-life | Advantages | Preferred In |
|---|---|---|---|---|
| Chlordiazepoxide | 25–100 mg PO q4–6h PRN | Long (24–48 h, active metabolites >100 h) | Smooth tapering due to long half-life, less interdose withdrawal | General medical floor, outpatient detox |
| Diazepam | 5–20 mg IV/PO q5–15 min for loading, then PRN | Long (20–100 h) | Rapid onset (especially IV), self-tapering due to active metabolites | "Front-loading" protocol for severe withdrawal |
| Lorazepam | 1–4 mg IV/PO q1–2h PRN | Short-intermediate (10–20 h) | No active metabolites, no hepatic oxidation (glucuronidation only) | Liver disease, elderly, respiratory compromise |
Phenobarbital Protocol
Phenobarbital is increasingly used as a first-line or adjunctive agent for alcohol withdrawal, particularly in severe cases or benzodiazepine-refractory withdrawal. Protocol: Loading dose 10 mg/kg IV (typical: 650–1300 mg over 30 minutes, max rate 60 mg/min), then 130–260 mg IV q15–30 min PRN for ongoing severe symptoms. Phenobarbital has a very long half-life (80–120 hours), providing a built-in taper. Monitor respiratory status closely. Advantages: effective in benzodiazepine-resistant withdrawal, acts at a different site on the GABA-A receptor (prolongs chloride channel opening vs. benzodiazepine-mediated increased frequency), can directly open the channel at high doses without requiring endogenous GABA.
Delirium Tremens (DT)
DT occurs in ~5% of patients undergoing alcohol withdrawal, typically 48–96 hours after last drink. Risk factors: prior DT or withdrawal seizures, age >65, concurrent illness, CIWA >15 on admission, heart rate >100, heavy prolonged drinking, kindling from multiple prior withdrawals. Management requires ICU-level care with IV benzodiazepines (diazepam 10–20 mg IV q5–10 min or lorazepam 2–4 mg IV q15 min until calm but arousable), aggressive fluid and electrolyte replacement (hypomagnesemia and hypokalemia are common), thiamine, glucose monitoring, and hemodynamic monitoring. Refractory DT may require propofol or dexmedetomidine infusion.
Seizure Prophylaxis
Benzodiazepines are effective for seizure prevention. Phenytoin is NOT effective for alcohol withdrawal seizures and should not be used. If seizures occur, treat with lorazepam 2 mg IV; if recurrent, load with phenobarbital. Status epilepticus protocol applies if seizures are prolonged (>5 minutes).
07 Pharmacotherapy for AUD
Three medications have FDA approval for AUD: naltrexone, acamprosate, and disulfiram. Several off-label agents have supporting evidence. Despite strong efficacy data, fewer than 10% of patients with AUD receive any pharmacotherapy.
| Medication | Mechanism | Dosing | Key Evidence | Contraindications / Pearls |
|---|---|---|---|---|
| Naltrexone (oral) | Mu-opioid receptor antagonist; blocks alcohol-induced endorphin release, reducing reward and craving | 50 mg PO daily (start 25 mg × 2 days to minimize nausea) | COMBINE trial (2006): reduced heavy drinking days by 25%; NNT ~12 for return to any drinking | Contraindicated with current opioid use (precipitates withdrawal); check LFTs (rare hepatotoxicity at high doses); patients must be opioid-free ≥7–10 days |
| Naltrexone (IM extended-release) | Same as oral; monthly depot injection | 380 mg IM gluteal q4 weeks | Improves adherence; RCT showed reduced heavy drinking vs. placebo; useful when adherence is a concern | Same opioid contraindications; injection site reactions; expensive |
| Acamprosate | Modulates glutamate (NMDA antagonist) and GABA activity; reduces the hyperexcitable state of protracted withdrawal | 666 mg PO TID (two 333 mg tablets TID); dose-reduce to 333 mg TID if CrCl 30–50 | European trials show efficacy for maintaining abstinence; COMBINE trial did not show benefit over naltrexone + behavioral therapy alone | Contraindicated if CrCl <30; renally excreted (no hepatic metabolism — safe in liver disease); most common side effect is diarrhea; TID dosing reduces adherence |
| Disulfiram | Irreversibly inhibits aldehyde dehydrogenase → acetaldehyde accumulation after alcohol ingestion → aversive reaction (flushing, nausea, vomiting, headache, hypotension, tachycardia) | 250 mg PO daily (range 125–500 mg); must be alcohol-free ≥12 hours before starting | Best evidence when administered under direct observation (supervised dosing); limited efficacy in unsupervised settings due to non-adherence | Contraindicated with metronidazole, certain cephalosporins (disulfiram-like reaction); severe hepatic/cardiac disease; psychosis; pregnancy. Reaction can be severe/fatal. Patient must be fully informed and motivated |
| Gabapentin (off-label) | Modulates voltage-gated calcium channels (alpha-2-delta subunit); GABAergic effects; reduces craving and anxiety during early recovery | 900–1800 mg/day divided TID | RCTs show reduced heavy drinking and improved abstinence, especially at 1800 mg dose; also treats insomnia and anxiety common in early recovery | Dose-adjust for renal function; misuse potential (especially in patients with OUD); sedation; peripheral edema |
| Topiramate (off-label) | Multiple mechanisms: enhances GABA, inhibits glutamate (AMPA/kainate), blocks voltage-gated sodium channels | Start 25 mg daily, titrate to 200–300 mg/day divided BID over 8 weeks | Multiple RCTs show reduced heavy drinking days and improved abstinence; may be particularly effective for heavy drinkers not yet abstinent | Cognitive side effects ("brain fog"), paresthesias, weight loss, kidney stones, metabolic acidosis; teratogenic (cleft palate); contraindicated in pregnancy; slow titration essential |
Patients carrying the OPRM1 A118G polymorphism (Asp40 variant, ~15–25% of European-descent individuals) show enhanced response to naltrexone, with a greater reduction in heavy drinking days. While pharmacogenomic testing is not yet standard of care, this evidence supports the role of personalized medicine in addiction treatment.
08 Alcohol-Related Emergencies
Acute Alcohol Intoxication
BAC (blood alcohol concentration) correlates with clinical effects: 50–100 mg/dL = impaired coordination and judgment; 100–200 mg/dL = ataxia, slurred speech, nystagmus; 200–300 mg/dL = marked ataxia, vomiting, obtundation; 300–400 mg/dL = stupor, hypothermia, respiratory depression; >400 mg/dL = coma and risk of death (though chronic users may tolerate much higher levels). Management is supportive: airway protection, IV fluids, glucose check, thiamine, electrolyte monitoring, aspiration precautions. Consider co-ingestants in any intoxicated patient (acetaminophen, opioids).
Toxic Alcohols — Methanol & Ethylene Glycol
Both methanol (windshield washer fluid, moonshine) and ethylene glycol (antifreeze) are metabolized by alcohol dehydrogenase (ADH) to toxic metabolites. Methanol → formaldehyde → formic acid (causes retinal toxicity and blindness, basal ganglia hemorrhagic necrosis). Ethylene glycol → glycolaldehyde → glycolic acid → oxalic acid (causes renal tubular damage, calcium oxalate crystalluria, acute kidney injury).
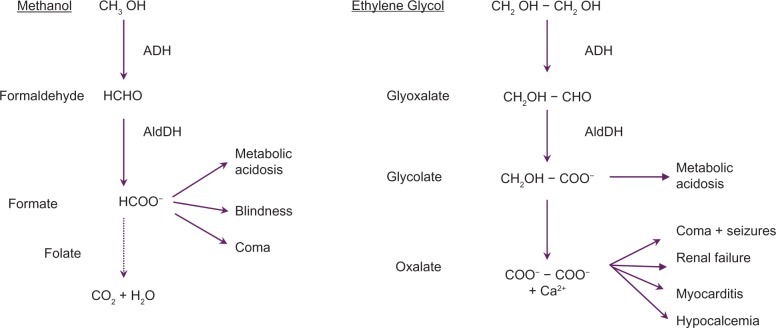
Suspect when: Anion gap metabolic acidosis (AG >12) + elevated osmolar gap (>10 mOsm/kg) + patient appears "drunk" but has low or undetectable ethanol level. Osmolar gap = measured osmolality − calculated osmolality (2×Na + BUN/2.8 + glucose/18 + EtOH/4.6). Early presentation: high osmolar gap before toxic metabolites form. Late presentation: high anion gap after conversion to acids (osmolar gap may normalize).
Treatment: (1) Fomepizole (4-methylpyrazole): competitive ADH inhibitor. Loading dose 15 mg/kg IV, then 10 mg/kg q12h × 4 doses, then 15 mg/kg q12h. First-line antidote. (2) Ethanol drip (alternative if fomepizole unavailable): target serum ethanol 100–150 mg/dL; difficult to titrate, causes intoxication. (3) Hemodialysis: indicated for severe acidosis (pH <7.25), renal failure, visual symptoms (methanol), serum methanol or ethylene glycol level >50 mg/dL, or end-organ damage. (4) Folinic acid (leucovorin) 50 mg IV q6h for methanol (enhances formate metabolism). Thiamine 100 mg IV + pyridoxine 50 mg IV q6h for ethylene glycol (shunts metabolism to less toxic pathways).
Wernicke Encephalopathy Prophylaxis & Treatment
Prophylaxis for at-risk patients (malnourished, chronic AUD, prolonged vomiting): thiamine 250–500 mg IV daily for 3–5 days. Always give before or concurrent with glucose-containing fluids. Treatment of established Wernicke: thiamine 500 mg IV TID for 2–3 days, then 250 mg IV daily for 3–5 days, then oral thiamine 100 mg TID indefinitely. Oral thiamine alone is inadequate for treatment — gut absorption is saturated at ~4.5 mg and is further impaired in alcoholism.
Alcoholic Ketoacidosis (AKA)
AKA develops in chronic alcohol users after a binge followed by cessation of intake (often with vomiting and poor oral intake). The combination of alcohol metabolism (high NADH:NAD+ ratio), glycogen depletion, volume depletion, and starvation shifts hepatic metabolism toward ketogenesis. Presentation: anion gap metabolic acidosis, ketonemia/ketonuria, nausea, vomiting, abdominal pain, and typically a low or normal blood glucose (distinguishes from DKA). Ethanol level is often low or undetectable by the time of presentation. Treatment: IV normal saline with dextrose (D5NS) — dextrose stimulates insulin release, which suppresses ketogenesis, and volume repletion restores renal acid excretion. Thiamine before glucose. Electrolyte repletion (K+, Mg2+, phosphate). Resolves rapidly with fluids and dextrose, typically within 12–24 hours.
Mallory-Weiss & Boerhaave Syndrome
Forceful vomiting or retching (common in alcohol intoxication) can cause a Mallory-Weiss tear (partial-thickness mucosal laceration at the gastroesophageal junction, presenting as hematemesis; usually self-limited, treat with PPI and endoscopy if persistent). Rarely, complete transmural esophageal rupture occurs (Boerhaave syndrome), a surgical emergency presenting with severe chest pain, subcutaneous emphysema, and pneumomediastinum.
09 OUD — Epidemiology, Pharmacology & Diagnosis
Opioid use disorder (OUD) affects approximately 2.7 million Americans. Opioid-involved overdose deaths reached ~82,000 in 2022, driven primarily by illicitly manufactured fentanyl. The opioid epidemic evolved through three waves: (1) prescription opioid deaths rising from the late 1990s; (2) heroin deaths increasing from 2010; (3) synthetic opioid (fentanyl) deaths surging from 2013.
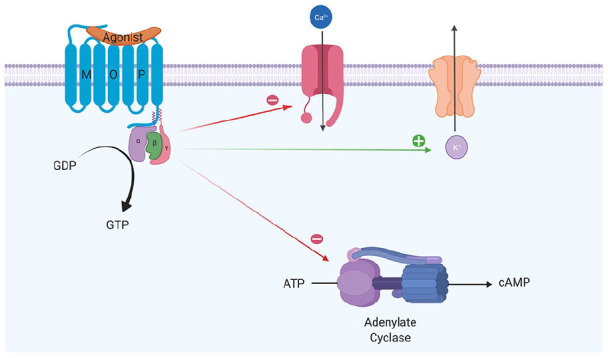
Opioid Receptor Pharmacology
| Receptor | Endogenous Ligand | Location | Effects When Activated |
|---|---|---|---|
| Mu (μ) / MOR | Beta-endorphin | PAG, thalamus, brainstem, spinal cord dorsal horn, GI tract, NAc | Analgesia (supraspinal and spinal), euphoria, respiratory depression, miosis, decreased GI motility, physical dependence, cough suppression |
| Kappa (κ) / KOR | Dynorphin | Spinal cord, hypothalamus, PAG, NAc | Analgesia (spinal), dysphoria, sedation, diuresis, miosis; modulates reward (anti-reward system) |
| Delta (δ) / DOR | Enkephalin | Cerebral cortex, olfactory bulb, pontine nuclei | Analgesia, anxiolysis, antidepressant effects; modulates mu receptor function |
Opioid Drug Classification
| Category | Examples | Clinical Significance |
|---|---|---|
| Full mu agonists | Morphine, oxycodone, hydromorphone, fentanyl, heroin, methadone | Dose-dependent analgesia and respiratory depression; no ceiling effect for respiratory depression |
| Partial mu agonist | Buprenorphine | Ceiling effect on respiratory depression at ~32 mg SL; high mu receptor affinity (displaces full agonists); can precipitate withdrawal if given to opioid-dependent patients |
| Mixed agonist-antagonist | Butorphanol, nalbuphine, pentazocine | Mu antagonist / kappa agonist; ceiling effect on respiratory depression; can precipitate withdrawal in opioid-dependent patients; dysphoria from kappa activation |
| Full antagonists | Naloxone, naltrexone, nalmefene | Competitive mu/kappa/delta antagonists; reverse opioid effects; no intrinsic activity; precipitate withdrawal in dependent patients |
Fentanyl Potency & Equivalency Comparisons
| Opioid | Approximate Potency (vs. Morphine) | Clinical Notes |
|---|---|---|
| Codeine | 0.15× | Prodrug metabolized by CYP2D6 to morphine; ultra-rapid metabolizers at risk for toxicity |
| Tramadol | 0.1× | Weak mu agonist + SNRI activity; seizure risk; serotonin syndrome risk |
| Hydrocodone | 1× | Equipotent to morphine; most commonly prescribed opioid in the US |
| Morphine | 1× (reference) | Reference standard for equianalgesic calculations |
| Oxycodone | 1.5× | Higher oral bioavailability than morphine |
| Hydromorphone | 4× | More potent; useful in renal impairment (fewer active metabolites than morphine) |
| Methadone | 4–10× (variable) | Non-linear potency; increases with chronic dosing; incomplete cross-tolerance; dangerous in rotation |
| Fentanyl | 50–100× | Highly lipophilic; rapid onset; illicitly manufactured fentanyl (IMF) is the leading cause of overdose deaths |
| Sufentanil | 500–1000× | Used in anesthesia; sublingual formulation available for acute pain |
| Carfentanil | 10,000× | Veterinary tranquilizer; detected in illicit drug supply; lethal in microgram quantities |
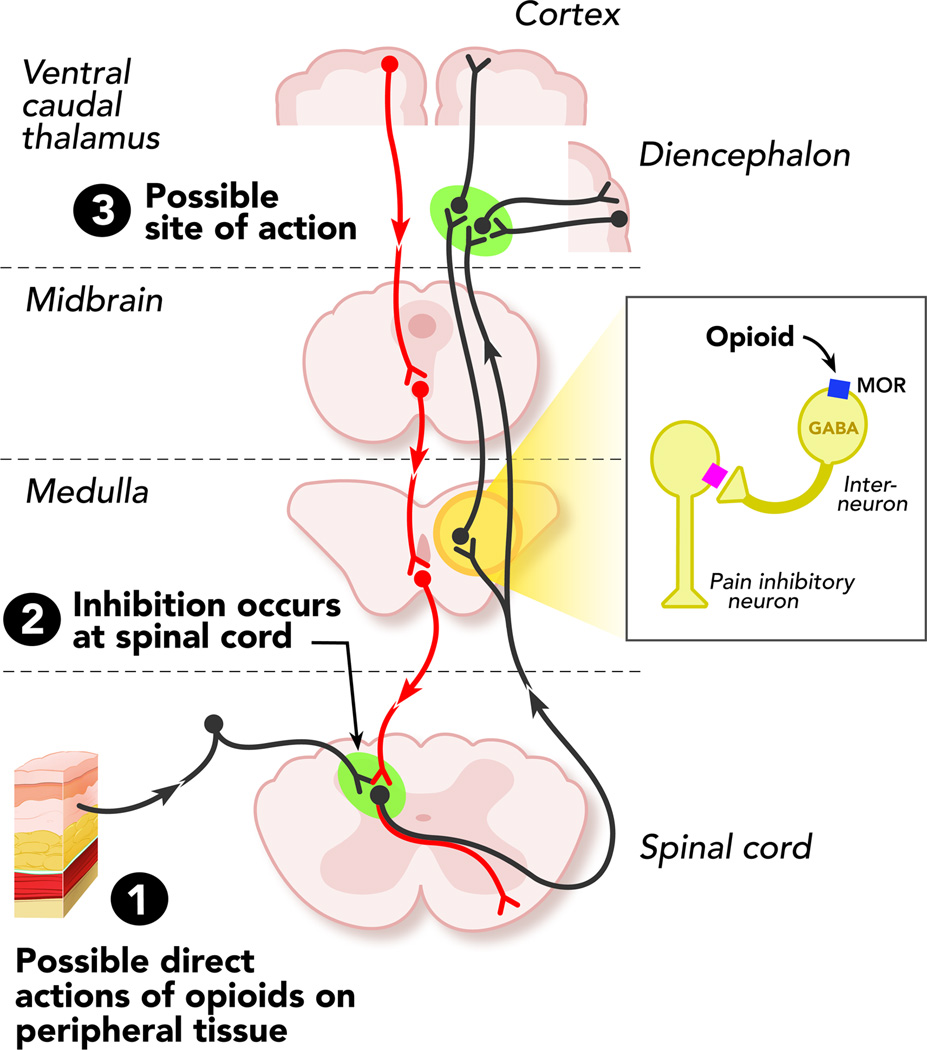
10 Opioid Withdrawal — COWS Protocol
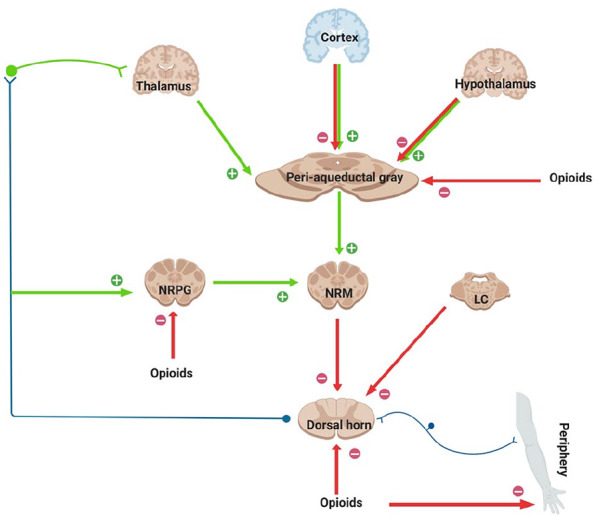
Opioid withdrawal is intensely uncomfortable but, unlike alcohol or benzodiazepine withdrawal, is not life-threatening in otherwise healthy adults. Symptoms resemble a severe flu: rhinorrhea, lacrimation, yawning, myalgias, diarrhea, abdominal cramps, piloerection ("gooseflesh" / "cold turkey"), anxiety, insomnia, restlessness, and dilated pupils. The timeline varies by drug half-life.
Withdrawal Timeline by Opioid
| Opioid | Onset | Peak | Duration |
|---|---|---|---|
| Heroin / short-acting opioids | 8–12 hours after last use | 36–72 hours | 5–7 days |
| Fentanyl (illicit) | 2–6 hours (rapid onset due to tissue redistribution) | 24–48 hours | 7–10 days (may be prolonged due to lipophilicity/tissue depot) |
| Methadone | 24–48 hours (long half-life) | 72–96 hours | 14–21 days |
| Buprenorphine | 24–72 hours | 3–5 days | 7–14 days |
COWS (Clinical Opiate Withdrawal Scale) — Fully Enumerated
The COWS is an 11-item clinician-rated instrument used to assess opioid withdrawal severity and guide buprenorphine induction. Score range: 0–48.
1. Resting pulse rate (measured after sitting/lying for 1 min): 0 = ≤80 bpm; 1 = 81–100; 2 = 101–120; 4 = >120
2. Sweating (over past 30 min, not from room temperature/activity): 0 = no report of chills or flushing; 1 = subjective report of chills/flushing; 2 = flushed or observable moistness on face; 3 = beads of sweat on brow or face; 4 = sweat streaming off face
3. Restlessness (observation during assessment): 0 = able to sit still; 1 = reports difficulty sitting still but able to; 3 = frequent shifting or extraneous movements; 5 = unable to sit still for more than a few seconds
4. Pupil size: 0 = pupils pinned or normal for room light; 1 = pupils possibly larger than normal; 2 = pupils moderately dilated; 5 = pupils so dilated only rim of iris visible
5. Bone or joint aches (if patient was having pain previously, assess only additional component): 0 = not present; 1 = mild diffuse discomfort; 2 = patient reports severe diffuse aching; 4 = patient is rubbing joints/muscles and unable to sit still due to discomfort
6. Runny nose or tearing (not from cold/allergies): 0 = not present; 1 = nasal stuffiness or unusually moist eyes; 2 = nose running or tearing; 4 = nose constantly running or tears streaming down cheeks
7. GI upset (over last 30 min): 0 = no GI symptoms; 1 = stomach cramps; 2 = nausea or loose stool; 3 = vomiting or diarrhea; 5 = multiple episodes of diarrhea or vomiting
8. Tremor (observation of outstretched hands): 0 = no tremor; 1 = tremor can be felt but not observed; 2 = slight tremor observable; 4 = gross tremor or muscle twitching
9. Yawning (observation during assessment): 0 = no yawning; 1 = yawning once or twice; 2 = yawning three or more times; 4 = yawning several times/minute
10. Anxiety or irritability: 0 = none; 1 = patient reports increasing irritability or anxiousness; 2 = patient obviously irritable/anxious; 4 = patient so irritable or anxious that participation in assessment is difficult
11. Gooseflesh skin: 0 = skin is smooth; 3 = piloerection of skin can be felt or hairs standing up on arms; 5 = prominent piloerection
COWS Scoring Interpretation
| Score | Severity | Clinical Implication |
|---|---|---|
| 5–12 | Mild | May begin buprenorphine induction in some protocols; symptomatic management may suffice |
| 13–24 | Moderate | Appropriate to initiate standard buprenorphine induction |
| 25–36 | Moderately severe | Initiate buprenorphine; consider more aggressive dosing |
| >36 | Severe | Urgent treatment required; buprenorphine should be initiated immediately |
Symptomatic Management of Opioid Withdrawal
| Symptom | Medication | Dose | Mechanism |
|---|---|---|---|
| Autonomic symptoms (anxiety, tachycardia, sweating, rhinorrhea) | Clonidine | 0.1–0.3 mg PO q6–8h (hold for SBP <90 or HR <60) | Alpha-2 agonist; reduces noradrenergic hyperactivity from locus coeruleus |
| Diarrhea | Loperamide | 4 mg initially, then 2 mg after each loose stool (max 16 mg/day) | Peripheral mu-opioid agonist (does not cross BBB at therapeutic doses) |
| Nausea/vomiting | Ondansetron | 4–8 mg PO/IV q8h PRN | 5-HT3 receptor antagonist |
| Abdominal cramps | Dicyclomine | 20 mg PO QID PRN | Anticholinergic/antispasmodic |
| Insomnia | Trazodone or hydroxyzine | Trazodone 50–100 mg HS; hydroxyzine 25–50 mg HS | Avoid benzodiazepines/Z-drugs in OUD patients |
| Myalgias | NSAIDs (ibuprofen) or acetaminophen | Ibuprofen 400–600 mg q6h; APAP 650 mg q4–6h | Non-opioid analgesics |
11 Buprenorphine — Induction, Stabilization & Maintenance
Buprenorphine is a partial mu-opioid agonist with very high receptor binding affinity (Ki ~1 nM, higher than morphine, methadone, or fentanyl). Its partial agonism produces a ceiling effect on respiratory depression (maximal at ~24–32 mg SL), making it significantly safer than full agonists in overdose. It also has kappa-opioid antagonist activity, which may contribute to antidepressant effects.
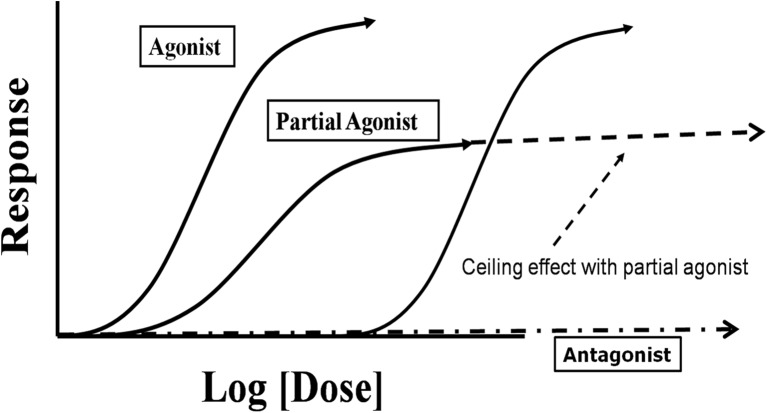
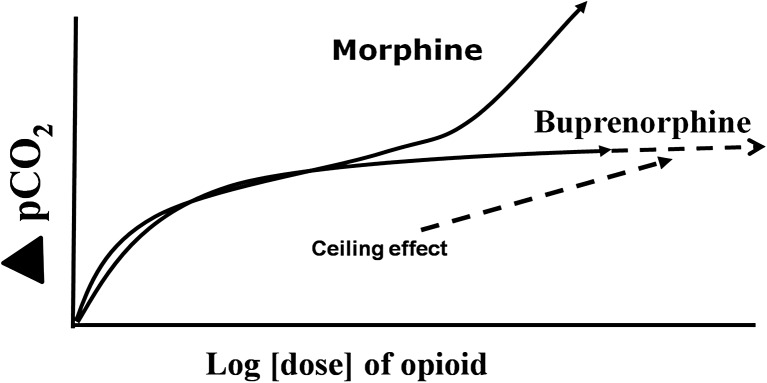
Regulatory History
The Drug Addiction Treatment Act (DATA) of 2000 allowed office-based buprenorphine prescribing via the "X-waiver." This requirement was eliminated in January 2023 by the Consolidated Appropriations Act — any DEA-licensed prescriber with a Schedule III authority can now prescribe buprenorphine for OUD without additional waiver, training, or patient caps.
Formulations
| Formulation | Brand Examples | Route | Notes |
|---|---|---|---|
| Buprenorphine/naloxone SL film | Suboxone | Sublingual | Most commonly prescribed; naloxone component deters IV misuse (naloxone has poor SL bioavailability but high IV bioavailability); available as 2/0.5 mg and 8/2 mg |
| Buprenorphine/naloxone SL tablet | Zubsolv | Sublingual | Higher bioavailability than film; 5.7/1.4 mg tablet ≈ 8/2 mg film |
| Buprenorphine SL tablet (mono) | Subutex (generic) | Sublingual | Used in pregnancy (naloxone component avoided due to theoretical risk, though likely safe); available 2 mg, 8 mg |
| Buprenorphine buccal film | Bunavail | Buccal | Applied to inner cheek; higher bioavailability than SL |
| Buprenorphine subdermal implant | Probuphine | Subdermal (inner upper arm) | 4 implants deliver ~8 mg SL equivalent/day for 6 months; for stable patients on ≤8 mg SL/day |
| Buprenorphine extended-release injection | Sublocade | SC abdominal injection monthly | 300 mg monthly × 2 months, then 100 mg or 300 mg monthly; eliminates daily adherence burden; depot provides steady-state levels |
Standard Induction Protocol
Because buprenorphine has higher receptor affinity but lower intrinsic activity than full agonists, it can displace full agonists from receptors and cause precipitated withdrawal if administered too early. Standard induction requires the patient to be in moderate withdrawal (COWS ≥8–12) before first dose.
Day 1: Wait until COWS ≥8–12. Administer buprenorphine/naloxone 2–4 mg SL. Reassess in 1–2 hours. If withdrawal persists or worsens, administer additional 2–4 mg SL. Maximum Day 1 dose: 8 mg.
Day 2: Administer total Day 1 dose plus an additional 2–4 mg if needed. Target dose: 8–16 mg. Maximum Day 2 dose: 16 mg.
Day 3 onward: Titrate to maintenance dose. Most patients stabilize on 12–24 mg/day. Maximum recommended dose: 24 mg/day (32 mg is the absolute maximum that provides additional receptor occupancy benefit).
Goal: ≥80% mu receptor occupancy is needed to suppress craving and block exogenous opioid effects; this typically requires ≥16 mg SL/day.
Micro-dosing / Bernese Method
The Bernese method involves gradual initiation of buprenorphine while the patient continues their full agonist opioid, avoiding the need for a withdrawal period. The rationale: very low doses of buprenorphine gradually accumulate on mu receptors over days, eventually displacing the full agonist without precipitating withdrawal. This approach is particularly useful for patients using fentanyl (which has unpredictable tissue depot effects making standard induction difficult) or patients unable to tolerate withdrawal.
Day 1: Buprenorphine 0.5 mg SL
Day 2: Buprenorphine 0.5 mg SL BID (1 mg total)
Day 3: Buprenorphine 1 mg SL BID (2 mg total)
Day 4: Buprenorphine 2 mg SL BID (4 mg total)
Day 5: Buprenorphine 3 mg SL BID (6 mg total)
Day 6: Buprenorphine 4 mg SL BID (8 mg total)
Day 7: Buprenorphine 12 mg SL (discontinue full agonist)
Day 8+: Titrate to therapeutic dose (16–24 mg/day)
Throughout Days 1–6: Patient continues usual opioid use (or prescribed full agonist). Adjunctive comfort medications (clonidine, ondansetron) as needed.
Precipitated Withdrawal
Precipitated withdrawal occurs when buprenorphine displaces a full agonist from mu receptors, producing acute, severe withdrawal within 30–60 minutes of administration. It is more intense than spontaneous withdrawal and can last 2–4 hours. Management: additional buprenorphine (paradoxically, providing more buprenorphine increases receptor occupancy and resolves symptoms), clonidine 0.1–0.3 mg, ondansetron, benzodiazepines for severe agitation. Prevention: ensure adequate COWS score before induction, use micro-dosing protocol for fentanyl users, wait longer for long-acting opioids (methadone: wait ≥72 hours).
12 Methadone Maintenance
Methadone is a full mu-opioid agonist with additional NMDA receptor antagonist activity and serotonin/norepinephrine reuptake inhibition. It has strong evidence for reducing illicit opioid use, criminal activity, HIV transmission, and all-cause mortality. However, its unique pharmacokinetics require careful management.
Regulatory Framework
In the US, methadone for OUD can only be dispensed through federally certified Opioid Treatment Programs (OTPs), regulated by SAMHSA, DEA, and state authorities. Patients must attend the OTP for observed daily dosing until they earn take-home privileges. Methadone for pain can be prescribed by any DEA-licensed practitioner, but methadone for OUD cannot be prescribed in an office setting (unlike buprenorphine). Take-home criteria are based on time in treatment, negative drug screens, attendance, and clinical stability.
Dosing & Titration
Initial dose: 20–30 mg PO on Day 1 (NEVER exceed 40 mg on Day 1, even if the patient reports high tolerance). Observe for 2–4 hours; an additional 5–10 mg may be given if withdrawal persists (max Day 1 total: 40 mg).
Titration: Increase by 5–10 mg every 3–7 days. Methadone has a long and variable half-life (8–59 hours, mean ~24 hours) with delayed peak effect — steady state is not reached for 3–5 days. Premature dose increases cause accumulation and respiratory depression, typically on days 3–5 (the most dangerous period).
Maintenance dose: Most patients stabilize at 60–120 mg/day. Doses <60 mg are associated with poorer retention and continued illicit use. Some patients require >120 mg for adequate receptor blockade.
Split dosing: Consider for patients with rapid metabolism (CYP3A4 inducers, pregnancy), breakthrough craving, or pain management needs — divide total daily dose BID or TID.
QTc Monitoring
Methadone prolongs the QTc interval in a dose-dependent manner by blocking the hERG (human ether-a-go-go-related gene) potassium channel. This creates a risk of torsades de pointes. Recommendations: obtain baseline ECG; repeat at 30 days and annually. If QTc >450 ms, discuss risks, monitor more frequently. If QTc >500 ms, consider dose reduction, switching to buprenorphine, or elimination of other QTc-prolonging medications. If patient develops syncope, palpitations, or seizure — obtain urgent ECG.
Drug Interactions — CYP3A4 Considerations
Methadone is primarily metabolized by CYP3A4, with contributions from CYP2B6, CYP2D6, and CYP2C19. CYP3A4 inducers decrease methadone levels and can precipitate withdrawal; CYP3A4 inhibitors increase levels and risk toxicity.
| CYP3A4 Inducers (decrease methadone levels) | CYP3A4 Inhibitors (increase methadone levels) |
|---|---|
| Rifampin (most potent — can reduce methadone levels by >50%), phenytoin, carbamazepine, phenobarbital, efavirenz, nevirapine, St. John's wort | Fluconazole, ketoconazole, erythromycin, clarithromycin, ciprofloxacin, ritonavir (complex — net effect varies), grapefruit juice, fluvoxamine |
Take-Home Criteria (Abridged)
SAMHSA regulations define a phased approach: Phase 1 (first 90 days) = no take-homes, daily observed dosing. After 90 days with demonstrated stability (negative UDS, regular attendance, no criminal activity, absence of recent substance use): 1 take-home per week. Gradually increasing to 2/week after 6 months, 3/week after 1 year, 6/week after 2 years, and up to 27 days of take-homes after 3 years of continuous demonstrated stability. COVID-era blanket exceptions (2020) expanded take-home access significantly.
13 Naloxone & Overdose Reversal
Naloxone is a competitive mu-opioid receptor antagonist that rapidly reverses opioid-induced respiratory depression. It is the cornerstone of opioid overdose emergency response and has been designated an over-the-counter (OTC) medication (Narcan nasal spray) since 2023.
Dosing & Routes
| Route | Formulation | Dose | Onset | Duration |
|---|---|---|---|---|
| Intranasal | Narcan nasal spray | 4 mg (1 spray in 1 nostril); Kloxxado: 8 mg | 2–5 minutes | 30–90 minutes |
| Intramuscular | Auto-injector or vial | 0.4–2 mg IM (deltoid or anterolateral thigh) | 2–5 minutes | 30–90 minutes |
| Intravenous | Vial | 0.04–0.4 mg IV; titrate q2–3 min (use lower doses in opioid-dependent patients to avoid precipitating severe withdrawal) | 1–2 minutes | 30–90 minutes |
Renarcotization
Naloxone's duration of action (30–90 minutes) is shorter than most opioids, especially long-acting agents (methadone: half-life 8–59 hours) and fentanyl (which, when used in large quantities or as tissue depots, can outlast naloxone by hours). Renarcotization occurs when naloxone wears off and residual opioid re-activates receptors, causing recurrent respiratory depression. Any patient receiving naloxone should be observed for a minimum of 2 hours after the last naloxone dose (4 hours or more for long-acting opioids or large fentanyl exposures). If repeated naloxone doses are required, consider a continuous naloxone infusion (two-thirds of the initial effective bolus dose per hour).
Community Distribution Programs
Naloxone distribution programs provide take-home naloxone kits to people who use opioids, their family members, and community organizations. Evidence shows these programs are associated with reduced opioid overdose mortality at the community level. All 50 US states have enacted naloxone access laws, and most allow pharmacist dispensing without a prescription. Training focuses on recognizing overdose (unresponsiveness, shallow/absent breathing, cyanosis, pinpoint pupils) and administering naloxone with rescue breathing.
Good Samaritan Laws
Most US states have enacted Good Samaritan laws that provide limited legal immunity to individuals who call 911 to report an overdose. These laws aim to reduce overdose deaths by removing the fear of criminal prosecution (for drug possession) that may otherwise deter bystanders from seeking emergency assistance. Coverage varies by state — some only protect the caller, others protect the overdose victim as well.
14 Stimulant Use Disorder — Cocaine & Methamphetamine
Stimulant use disorder encompasses cocaine and amphetamine-type stimulants (methamphetamine, prescription amphetamines). There are no FDA-approved pharmacotherapies for stimulant use disorder; contingency management is the most effective evidence-based behavioral treatment.
Pharmacology
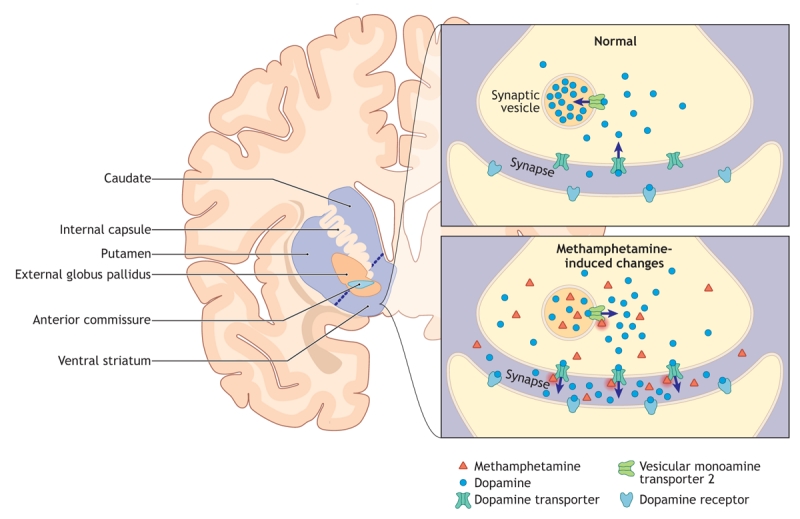
Cocaine blocks the dopamine transporter (DAT), norepinephrine transporter (NET), and serotonin transporter (SERT), preventing reuptake and increasing synaptic monoamine concentrations. It also blocks voltage-gated sodium channels (local anesthetic effect). Half-life: ~1 hour (intranasal); onset varies by route — smoked/IV: seconds, intranasal: 3–5 minutes. Methamphetamine enters the presynaptic terminal via DAT, reverses the vesicular monoamine transporter (VMAT2), and causes massive release of dopamine (and norepinephrine/serotonin) into the synapse. It also inhibits monoamine oxidase (MAO). The result is a much longer and more intense dopamine surge than cocaine (half-life 10–12 hours).
Intoxication — Sympathomimetic Toxidrome
Stimulant intoxication produces the sympathomimetic toxidrome: tachycardia, hypertension, hyperthermia, diaphoresis, mydriasis (dilated pupils), agitation, paranoia, psychosis, seizures, and potentially life-threatening cardiovascular events. Specific complications include: acute coronary syndrome (cocaine causes coronary vasospasm and thrombosis — treat with benzodiazepines, nitrates, aspirin; AVOID beta-blockers due to unopposed alpha stimulation), aortic dissection, stroke (hemorrhagic > ischemic), rhabdomyolysis, and hyperthermia (can be fatal; treat aggressively with external cooling and benzodiazepines).
Withdrawal / "Crash"
Stimulant withdrawal is characterized by the "crash" phase: profound fatigue, hypersomnia, increased appetite, dysphoria, anhedonia, vivid unpleasant dreams, psychomotor retardation, and intense craving. Withdrawal is not medically dangerous (no seizures, no autonomic instability) but the severe dysphoria and craving drive relapse. Symptoms peak at 2–4 days and may last 1–3 weeks, with protracted anhedonia for months.
Contingency Management
Contingency management (CM) provides tangible incentives (typically escalating monetary-value vouchers or prize draws) contingent on verified abstinence (negative UDS). CM produces the largest effect sizes of any behavioral treatment for stimulant use disorder (d = 0.42–0.62). The VA and some state Medicaid programs have implemented CM programs. Limitations: sustainability after incentives end, funding, and perceived ethical concerns about "paying people not to use drugs."
Cardiovascular Complications of Chronic Use
Cocaine cardiovascular effects: Accelerated atherosclerosis (even in young patients without traditional risk factors), coronary artery aneurysms, coronary artery dissection, dilated cardiomyopathy, aortic dissection, and infective endocarditis (particularly right-sided tricuspid valve endocarditis in IV cocaine users — often caused by Staphylococcus aureus). Cocaine-associated MI should be treated with benzodiazepines, aspirin, nitroglycerin, and cardiac catheterization if indicated; avoid beta-blockers acutely.
Methamphetamine cardiovascular effects: Dilated cardiomyopathy is the most common cardiovascular complication, often presenting in patients aged 30–50 with no other risk factors; may partially reverse with abstinence and standard HF therapy. Pulmonary arterial hypertension (associated with methamphetamine more than cocaine), accelerated hypertension, and aortic dissection. Methamphetamine-associated dental disease ("meth mouth"): Results from xerostomia (decreased salivary flow), bruxism, poor oral hygiene, high sugar intake, and direct vasoconstriction of oral blood supply; manifests as rampant caries, tooth fractures, and periodontal disease.
Dermatologic Complications
Methamphetamine use is associated with formication (tactile hallucination of insects crawling on or under the skin), leading to compulsive skin-picking and excoriations ("meth sores" or "crank bugs"). Cocaine use via nasal insufflation causes nasal septal perforation from chronic vasoconstriction-mediated ischemic necrosis of the septal cartilage. IV stimulant use carries the same injection-related complications as IV opioid use: abscess, cellulitis, endocarditis, HIV, HCV.
15 Cannabis Use Disorder
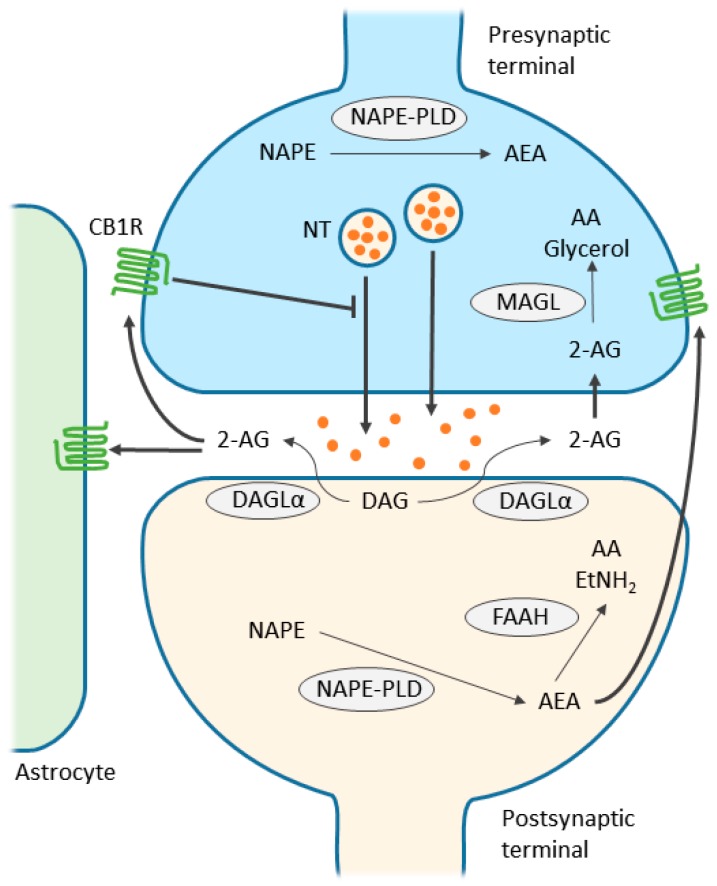
Cannabis is the most widely used illicit substance globally. Approximately 16 million Americans meet DSM-5 criteria for cannabis use disorder. The primary psychoactive compound is delta-9-tetrahydrocannabinol (THC), which acts as a partial agonist at CB1 receptors (densely expressed in the hippocampus, cerebellum, basal ganglia, and cortex) and CB2 receptors (primarily immune cells). Cannabidiol (CBD) is a non-intoxicating cannabinoid with complex pharmacology including 5-HT1A agonism, GPR55 antagonism, and allosteric modulation of mu-opioid receptors.
Intoxication
Acute effects of cannabis include euphoria, relaxation, altered time perception, impaired short-term memory, increased appetite, conjunctival injection, dry mouth, tachycardia, and impaired psychomotor coordination. High doses or high-potency products can produce anxiety, paranoia, and acute psychosis (especially in predisposed individuals). THC concentration in commercial cannabis products has increased from ~4% in 1995 to >15–20% currently, with concentrates reaching 60–90%.
Cannabis Withdrawal
Withdrawal is a recognized DSM-5 diagnosis: onset within 1 week of cessation, peak at 1–2 weeks, duration up to 4 weeks. Symptoms include irritability, anger, anxiety, insomnia, decreased appetite, restlessness, depressed mood, and at least one physical symptom (abdominal pain, tremor, sweating, fever, chills, headache). Withdrawal is not medically dangerous but can drive relapse.
Cannabinoid Hyperemesis Syndrome (CHS)
CHS is a paradoxical condition in chronic, heavy cannabis users characterized by cyclic episodes of severe nausea, intractable vomiting, and diffuse abdominal pain. The pathophysiology is poorly understood but may relate to CB1 receptor desensitization in the enteric nervous system or hypothalamic thermoregulation. Diagnostic features: history of regular cannabis use for ≥1 year, ≥3 stereotypical episodes, and resolution with cannabis cessation.
Acute relief: Hot water bathing/showering (compulsive, near-pathognomonic behavior — patients spend hours in hot showers); topical capsaicin cream (0.075%) applied to the abdomen (stimulates TRPV1 receptors, mimicking hot water); IV fluids; ondansetron (often ineffective); haloperidol 2.5–5 mg IV (evidence supports superior antiemetic effect vs. ondansetron in CHS).
Definitive treatment: Permanent cessation of all cannabis products is the only definitive cure. Symptoms resolve within 1–2 weeks of complete cessation and recur with resumed use.
Synthetic Cannabinoids (K2/Spice)
Synthetic cannabinoids are full CB1 agonists (unlike THC, which is a partial agonist), producing more intense and unpredictable effects: severe agitation, psychosis, seizures, tachycardia, hypertension, acute kidney injury, coagulopathy (some batches have been contaminated with brodifacoum, a long-acting anticoagulant). Not detected on standard urine drug screens. Management is supportive; benzodiazepines for agitation and seizures.
Medical Consequences of Chronic Cannabis Use
Pulmonary: Regular cannabis smoking is associated with chronic bronchitis symptoms (cough, sputum, wheeze); evidence for COPD and lung cancer is less clear than with tobacco due to confounding (most cannabis smokers also smoke tobacco). Cannabis smoke contains many of the same carcinogens as tobacco smoke. Psychiatric: Cannabis use during adolescence (especially heavy use of high-potency products) is associated with a 2–4 fold increased risk of developing psychotic disorders, including schizophrenia, particularly in individuals with genetic predisposition (COMT Val158Met polymorphism). Cognitive: Acute THC impairs attention, working memory, and executive function; chronic heavy use during adolescence may produce persistent cognitive deficits even after cessation, though evidence for permanent structural brain changes is debated. Reproductive: THC reduces sperm count and motility; cannabis use during pregnancy is associated with lower birth weight.
Treatment
No FDA-approved pharmacotherapy exists for cannabis use disorder. Motivational Enhancement Therapy (MET) combined with CBT is the most studied behavioral approach (MET/CBT-5 and MET/CBT-12 are manualized protocols). N-acetylcysteine (NAC) 1200 mg BID showed modest benefit in adolescents (but not adults) in one RCT. Gabapentin has shown preliminary evidence for reducing withdrawal symptoms and cannabis use. Abstinence-based contingency management also shows efficacy. Brief interventions using MI principles are effective for reducing cannabis use in young adults.
16 Sedative-Hypnotic Use Disorder — Benzodiazepines & Z-drugs
Benzodiazepines and Z-drugs (zolpidem, zaleplon, eszopiclone) act on the GABA-A receptor complex. Benzodiazepines bind the alpha subunit at the benzodiazepine binding site, increasing the frequency of chloride channel opening (requiring endogenous GABA). Physiologic dependence develops rapidly, often within 2–4 weeks of daily use, and withdrawal can be life-threatening (seizures, delirium).
Diazepam Equivalency Table
Converting all benzodiazepines to a diazepam equivalent facilitates standardized tapering. Diazepam is preferred for tapering because of its long half-life and availability in small-dose increments.
| Benzodiazepine | Approximate Dose Equivalent to Diazepam 10 mg | Half-life (hours) | Onset |
|---|---|---|---|
| Alprazolam (Xanax) | 0.5 mg | 6–12 | Intermediate |
| Chlordiazepoxide (Librium) | 25 mg | 24–48 (active metabolites >100) | Intermediate |
| Clonazepam (Klonopin) | 0.5 mg | 18–50 | Intermediate |
| Clorazepate (Tranxene) | 7.5 mg | 36–200 (active metabolite) | Rapid |
| Diazepam (Valium) | 10 mg (reference) | 20–100 (active metabolites) | Rapid |
| Lorazepam (Ativan) | 1 mg | 10–20 | Intermediate |
| Oxazepam (Serax) | 15 mg | 4–15 | Slow |
| Temazepam (Restoril) | 15 mg | 8–22 | Intermediate |
| Triazolam (Halcion) | 0.25 mg | 2–5 | Rapid |
| Midazolam (Versed) | 3 mg (IV) | 1.5–2.5 | Rapid |
Ashton Taper Method
The Ashton Manual (Professor C. Heather Ashton) provides detailed, individualized benzodiazepine tapering protocols. Key principles: (1) Convert to an equivalent dose of diazepam (long half-life, smoother plasma levels); (2) Taper by approximately 10% of the current dose every 1–2 weeks (some patients require slower reductions of 5% every 2–4 weeks); (3) Final stages of taper are the hardest — slow down further below 5 mg diazepam equivalent; (4) Total taper duration: 6 weeks to 12+ months depending on the dose, duration of use, and patient tolerance. Withdrawal symptoms: insomnia, anxiety, irritability, tremor, diaphoresis, perceptual disturbances, seizures (most dangerous), depersonalization, and rarely psychosis.
Benzodiazepine Withdrawal Symptom Phases
| Phase | Timeline | Symptoms |
|---|---|---|
| Acute withdrawal | 1–4 days after cessation (short-acting) or 2–7 days (long-acting) | Anxiety, insomnia, tremor, diaphoresis, tachycardia, hypertension; seizures (peak risk day 2–7); delirium (rare but life-threatening) |
| Protracted withdrawal | Weeks to months after acute phase | Anxiety, insomnia, depression, cognitive impairment, perceptual disturbances (tinnitus, paresthesias, visual distortions), depersonalization, GI symptoms; wax and wane; can last 6–18 months |
| Rebound | Days to weeks | Return of the original symptoms (anxiety, insomnia) for which the benzodiazepine was prescribed, often at greater intensity than baseline; self-limited (1–3 weeks) |
Carbamazepine: 200–400 mg/day — reduces withdrawal seizure risk and anxiety; useful adjunct during taper.
Gabapentin/Pregabalin: Reduces anxiety and insomnia during taper; pregabalin has some evidence of misuse in SUD populations.
Propranolol: 10–20 mg TID — reduces autonomic symptoms (tremor, tachycardia, sweating) but does NOT prevent seizures.
Trazodone: 50–100 mg HS for insomnia during and after taper.
SSRIs/SNRIs: Start early in the taper to provide long-term anxiety/depression treatment as benzodiazepine is withdrawn.
Barbiturate Withdrawal
Barbiturate withdrawal is similar to but potentially more severe than benzodiazepine withdrawal. Barbiturates directly open GABA-A chloride channels (do not require endogenous GABA at high doses), and withdrawal produces severe autonomic instability, seizures, and delirium. Phenobarbital taper is the standard treatment: convert to phenobarbital equivalents, stabilize for 2–3 days, then taper by 30 mg/day. The pentobarbital tolerance test can help estimate the degree of barbiturate dependence: administer 200 mg pentobarbital orally and observe after 1 hour — if the patient is asleep, minimal dependence; if awake and comfortable, moderate dependence; if no effect, severe dependence requiring higher stabilization doses.
Z-Drug Considerations
Zolpidem, zaleplon, and eszopiclone bind selectively to the alpha-1 subunit of the GABA-A receptor (unlike benzodiazepines, which are non-selective). Despite initial marketing as non-addictive, physical dependence and withdrawal occur with chronic use, and the withdrawal syndrome is similar to benzodiazepine withdrawal (though typically less severe). Cross-tolerance with benzodiazepines is partial. Zolpidem is associated with complex sleep-related behaviors (sleep-driving, sleep-eating) and is a controlled substance (Schedule IV). Taper by converting to an equivalent diazepam dose (zolpidem 10 mg ≈ diazepam 10 mg) and following the Ashton protocol.
17 Tobacco Use Disorder
Tobacco use is the leading preventable cause of death in the United States (~480,000 deaths/year, including ~41,000 from secondhand smoke). Nicotine acts on nicotinic acetylcholine receptors (nAChRs), particularly the alpha-4-beta-2 subtype in the VTA, stimulating dopamine release in the nucleus accumbens. Nicotine is one of the most addictive substances known, with a relapse rate of ~95% without treatment.
Fagerstrom Test for Nicotine Dependence (FTND)
| Question | Answer | Score |
|---|---|---|
| How soon after waking do you smoke your first cigarette? | Within 5 min / 6–30 min / 31–60 min / >60 min | 3 / 2 / 1 / 0 |
| Do you find it difficult to refrain from smoking in places where it is forbidden? | Yes / No | 1 / 0 |
| Which cigarette would you hate most to give up? | First in morning / Any other | 1 / 0 |
| How many cigarettes per day do you smoke? | ≤10 / 11–20 / 21–30 / ≥31 | 0 / 1 / 2 / 3 |
| Do you smoke more frequently in the first hours after waking? | Yes / No | 1 / 0 |
| Do you smoke even if you are ill enough to be in bed most of the day? | Yes / No | 1 / 0 |
Score range 0–10: 0–2 = very low dependence; 3–4 = low; 5 = medium; 6–7 = high; 8–10 = very high dependence.
Pharmacotherapy — Nicotine Replacement Therapy (NRT)
| NRT Formulation | Dosing | Notes |
|---|---|---|
| Transdermal patch | >10 cig/day: 21 mg/day × 6 weeks, then 14 mg/day × 2 weeks, then 7 mg/day × 2 weeks. ≤10 cig/day: Start at 14 mg | Provides steady-state nicotine; apply to clean, dry, hairless skin; rotate sites; remove at night if causing insomnia/vivid dreams |
| Gum | 2 mg (if smokes ≤25 cig/day) or 4 mg (if >25/day); 1 piece q1–2h for first 6 weeks, then taper; max 24 pieces/day | "Chew and park" technique: chew until peppery taste, park between cheek and gum; avoid acidic beverages (coffee, juice) which reduce buccal absorption |
| Lozenge | 2 mg or 4 mg (use 4 mg if first cigarette within 30 min of waking); 1 q1–2h; max 5/6 hours, 20/day | Dissolve in mouth over 20–30 minutes; do not chew or swallow; same acidic beverage precaution as gum |
| Nasal spray | 1 spray per nostril (1 mg total) q30–60 min PRN; max 40 sprays/day (5 per hour) | Fastest-acting NRT; most closely mimics the rapid nicotine delivery of smoking; initial nasal irritation is common |
| Oral inhaler | 6–16 cartridges/day; puff for 20 minutes per cartridge; taper over 6–12 weeks | Delivers nicotine vapor to oropharyngeal mucosa (not to lungs); mimics hand-to-mouth behavioral component of smoking |
Varenicline
Varenicline (Chantix) is a partial agonist at the alpha-4-beta-2 nAChR. It provides moderate dopaminergic stimulation (reducing craving and withdrawal) while blocking the rewarding effects of nicotine from cigarettes. Dosing: 0.5 mg daily × 3 days, then 0.5 mg BID × 4 days, then 1 mg BID for 12 weeks (may extend to 24 weeks). Set a quit date during the second week of treatment. Most effective single agent for smoking cessation (OR ~2.2–2.9 vs. placebo). The earlier FDA black box warning for neuropsychiatric events was removed in 2016 based on the EAGLES trial (no significant increase in neuropsychiatric adverse events vs. NRT or placebo). Common side effects: nausea (30%, mitigate by taking with food), vivid dreams, insomnia.
Bupropion
Bupropion SR (Zyban) is a norepinephrine-dopamine reuptake inhibitor (NDRI) that reduces craving and withdrawal symptoms. Dosing: 150 mg daily × 3 days, then 150 mg BID for 7–12 weeks. Start 1–2 weeks before quit date. Contraindicated in seizure disorders, eating disorders (bulimia/anorexia), and concurrent MAOIs. Lowers the seizure threshold. Also used for depression (Wellbutrin), providing dual benefit in patients with comorbid depression.
Combination Therapy
Combination NRT (patch + short-acting form such as gum or lozenge) is more effective than single NRT (OR ~1.9 vs. monotherapy). Varenicline + NRT patch may provide additional benefit. Combination pharmacotherapy + behavioral counseling is the most effective overall strategy, with 6-month quit rates of 25–35% (vs. ~5% for unaided quit attempts).
Electronic nicotine delivery systems (ENDS) deliver aerosolized nicotine without combustion, eliminating tar and most carcinogens. The relative harm reduction compared to combustible cigarettes is estimated at 90–95% less toxic, though long-term health effects remain uncertain. ENDS are not FDA-approved for smoking cessation, but some evidence (NEJM Hajek et al., 2019) suggests they may be more effective than NRT for smoking cessation. Concerns include: nicotine addiction initiation in youth, dual use (not fully switching), EVALI (E-cigarette or Vaping product use-Associated Lung Injury, primarily linked to vitamin E acetate in THC-containing products), and unknown long-term pulmonary effects.
18 Hallucinogens, Inhalants & Novel Psychoactive Substances
Classic Serotonergic Hallucinogens
LSD (lysergic acid diethylamide) and psilocybin (from Psilocybe mushrooms) are 5-HT2A receptor agonists that produce altered perception, visual hallucinations, synesthesia, mystical experiences, and altered sense of time. Duration: LSD 8–12 hours; psilocybin 4–6 hours. There is no physiologic dependence or withdrawal syndrome with classic psychedelics. Tolerance develops rapidly (tachyphylaxis within 1–3 days) and cross-tolerance exists between LSD and psilocybin. Acute adverse effects: "bad trip" (anxiety, paranoia, panic), rare persistent perceptual changes (HPPD — hallucinogen persisting perception disorder). Psilocybin has shown promising results in clinical trials for treatment-resistant depression and end-of-life distress; FDA granted "breakthrough therapy" designation in 2018.
MDMA (3,4-methylenedioxymethamphetamine)
MDMA is an amphetamine derivative ("entactogen") that causes massive serotonin release (with dopamine and norepinephrine release). Effects: empathy, emotional openness, euphoria, heightened sensory perception. Risks: hyperthermia, hyponatremia (SIADH from serotonin release + excessive water intake), serotonin syndrome (especially with SSRIs or MAOIs), hepatotoxicity, neurotoxicity (serotonergic axon damage with chronic use). MDMA-assisted psychotherapy received FDA review for PTSD treatment; initial results showed significant efficacy but the NDA was not approved in 2024 due to study design concerns.
Dissociative Agents — PCP & Ketamine
PCP (phencyclidine) is an NMDA receptor antagonist that also affects dopamine, sigma, and opioid receptors. Intoxication: violent agitation, vertical and horizontal nystagmus (unique to PCP), analgesia, psychosis, hyperthermia, rhabdomyolysis, seizures. Management: benzodiazepines for agitation (avoid antipsychotics if hyperthermia is present, as they impair thermoregulation); dark quiet room; monitor for rhabdomyolysis (CK, renal function). Ketamine is a shorter-acting NMDA antagonist; intoxication produces dissociative anesthesia, "K-hole" (deep dissociation at high doses). Esketamine (Spravato) nasal spray is FDA-approved for treatment-resistant depression. Chronic ketamine use causes ulcerative cystitis ("ketamine bladder").
Inhalants
Inhalants include volatile solvents (toluene, gasoline, paint thinner), aerosols, gases (nitrous oxide), and nitrites (amyl nitrite/"poppers"). Most prevalent in early adolescence (12–14 years). Mechanism: most are CNS depressants enhancing GABA and inhibiting glutamate. Sudden sniffing death syndrome is the leading cause of inhalant-related death — volatile hydrocarbons sensitize the myocardium to catecholamines, causing fatal ventricular fibrillation, often triggered by startle or exertion during or immediately after inhalant use. Chronic toluene exposure causes renal tubular acidosis (non-anion gap metabolic acidosis), cerebellar ataxia, and cognitive impairment. Chronic nitrous oxide use depletes vitamin B12, causing subacute combined degeneration of the spinal cord (posterior columns + lateral corticospinal tracts).
Novel Psychoactive Substances
Synthetic cathinones ("bath salts" — mephedrone, MDPV, alpha-PVP/"flakka") are synthetic analogs of cathinone (from the khat plant). They inhibit catecholamine reuptake and/or promote catecholamine release. Effects resemble methamphetamine or MDMA: severe sympathomimetic toxidrome with agitation, psychosis, hyperthermia, and rhabdomyolysis. MDPV is a particularly potent DAT inhibitor with high abuse potential. Treatment is supportive with benzodiazepines for agitation. Kratom (Mitragyna speciosa) contains mitragynine, a partial mu-opioid agonist; used for self-treatment of pain and opioid withdrawal, but carries risks of dependence, seizures, and hepatotoxicity.
19 Harm Reduction Principles & Programs
Harm reduction is a public health approach that aims to minimize the negative health, social, and economic consequences of substance use without necessarily requiring abstinence. It is grounded in the pragmatic recognition that some people cannot or will not stop using substances, and that reducing harm while maintaining engagement is preferable to an all-or-nothing approach.
Core Principles
(1) Pragmatism: Accept that drug use is part of the human experience and work to minimize harm rather than ignore or condemn use. (2) Spectrum of use: Understand drug use as a spectrum from abstinence to severe dependence — some use is clearly safer than other use, and any movement toward reduced harm is valuable. (3) Quality of life focus: Establish quality of individual and community life as criteria for evaluating interventions, not simply reduction in drug use. (4) Non-judgmental services: Provide services without judgment and without requiring abstinence as a precondition for receiving help. (5) Meaningful participation: Ensure people who use drugs have a voice in creating and evaluating programs that serve them. (6) Social determinants: Recognize that the root causes of problematic drug use — poverty, racism, trauma, adverse childhood experiences, social isolation, incarceration, homelessness — must be addressed alongside individual substance use.

Key Programs & Interventions
| Program | Description | Evidence |
|---|---|---|
| Needle/syringe exchange programs (SSPs) | Provide sterile injection equipment in exchange for used syringes; also offer wound care, testing for HIV/HCV, naloxone, referrals to treatment | Reduce HIV transmission by ~50%; do NOT increase drug use or crime in surrounding areas; cost-effective (each HIV infection prevented saves ~$380,000) |
| Supervised consumption sites (SCS) | Medically supervised facilities where people can use pre-obtained drugs under observation; staff can intervene for overdose | Zero overdose deaths have occurred at supervised consumption sites worldwide; associated with reduced public injecting, reduced discarded syringes, increased treatment entry. First US site opened in NYC in 2021 |
| Naloxone distribution | Provide naloxone kits to people who use opioids, family, community organizations | Communities with naloxone distribution programs have 27–46% lower overdose death rates |
| Fentanyl test strips | Immunoassay strips that detect fentanyl in drug samples before use | Users who test positive for fentanyl are 5× more likely to adopt risk-reduction behaviors (lower dose, use with someone, have naloxone present) |
| Housing First | Provide stable housing without requiring sobriety or treatment participation as a precondition | Reduces chronic homelessness by 80–90%; does not increase substance use; improves health outcomes and treatment engagement |
| Safer use education | Teach overdose prevention (never use alone, start low/go slow, avoid mixing substances), wound care for injection-related infections, safer smoking supplies | Empowers informed decision-making; reduces infectious complications and overdose risk |
20 Co-Occurring Disorders — Dual Diagnosis
Approximately 50% of individuals with a severe mental illness and 30–40% with any mental illness also have a substance use disorder. Conversely, people with SUD have high rates of comorbid depression (20–40%), anxiety disorders (20–30%), PTSD (30–60% in women with SUD), bipolar disorder, ADHD, and personality disorders (particularly antisocial and borderline).
Treatment Models
| Model | Approach | Evidence |
|---|---|---|
| Sequential | Treat one disorder first (typically SUD), then address the other | Outdated; patients fall through the cracks between systems; high dropout |
| Parallel | Treat both disorders simultaneously but in separate programs/providers | Better than sequential but fragmented; communication gaps between providers |
| Integrated | Treat both disorders simultaneously, within the same program, by the same treatment team using a unified treatment plan | Best evidence; recommended by SAMHSA; associated with superior outcomes for both conditions |
Medication Considerations
Avoid benzodiazepines whenever possible in patients with SUD (high abuse potential; cross-tolerance with alcohol; risk of respiratory depression with opioids). Preferred anxiolytics: SSRIs, SNRIs, buspirone, hydroxyzine, gabapentin (with caution regarding misuse potential). Antidepressants: SSRIs are first-line for comorbid depression/anxiety; bupropion is useful if comorbid tobacco dependence. Mood stabilizers: Valproate may reduce both alcohol and cannabis use in bipolar disorder. Lithium remains first-line for bipolar disorder but does not independently reduce substance use. Antipsychotics: Clozapine has the best evidence for reducing substance use in schizophrenia. Avoid prescribing medications with high abuse potential (stimulants for ADHD in active SUD — use extended-release formulations or non-stimulants like atomoxetine if needed).
21 Special Populations
Pregnancy — Opioid Use Disorder
MOUD is the standard of care for pregnant patients with OUD. Both methadone and buprenorphine are safe and recommended; medically supervised withdrawal (detoxification) during pregnancy is associated with high relapse rates and increased risk of fetal distress. Methadone has been used longest; buprenorphine is associated with shorter NICU stays and less severe neonatal withdrawal in the MOTHER trial (2012). Buprenorphine monoproduct (without naloxone) has traditionally been preferred in pregnancy, though evidence suggests the combination product is likely safe as naloxone has poor sublingual bioavailability. Dose requirements often increase during pregnancy (expanded volume of distribution, increased hepatic metabolism, especially in the third trimester).
Neonatal Abstinence Syndrome (NAS) / Neonatal Opioid Withdrawal Syndrome (NOWS)
NAS/NOWS occurs in 30–80% of neonates exposed to opioids in utero. Onset is typically 24–72 hours after birth (may be delayed up to 5–7 days with methadone or buprenorphine). Symptoms include high-pitched cry, tremors, irritability, poor feeding, vomiting, diarrhea, sneezing, yawning, nasal congestion, fever, and seizures (rare).
Scored q3–4h by nursing. Major categories and typical items:
CNS disturbances: Excessive high-pitched cry (2–3 points), continuous high-pitched cry (3), sleep <1 hour after feeding (3), sleep <2 hours (2), sleep <3 hours (1), hyperactive Moro reflex (2–3), tremors when disturbed/undisturbed (1–4), increased muscle tone (2), excoriation (1), myoclonic jerks (3), generalized seizures (5)
Metabolic/vasomotor/respiratory: Sweating (1), fever 37.2–38.3°C (1) or >38.4°C (2), frequent yawning (1), mottling (1), nasal stuffiness (1), sneezing (1), nasal flaring (2), respiratory rate >60 (1) or >60 with retractions (2)
GI disturbances: Excessive sucking (1), poor feeding (2), regurgitation (2), projectile vomiting (3), loose stools (2), watery stools (3)
Interpretation: Score ≥8 on three consecutive assessments → initiate pharmacotherapy. First-line: oral morphine (0.04–0.08 mg/kg q3–4h, titrate based on scores, wean by 10–20% every 24–48 hours once stable). Alternative: methadone. Non-pharmacologic care is critical: low-stimulation environment, swaddling, skin-to-skin contact, small frequent feedings, rooming-in with mother.
Breastfeeding Guidelines
Breastfeeding is encouraged for mothers on methadone or buprenorphine who are: (1) stable in treatment, (2) not using illicit drugs, (3) HIV-negative, and (4) not using other contraindicated medications. The small amount of methadone/buprenorphine transferred in breast milk is insufficient to treat NAS but may ease symptoms slightly. Breastfeeding should be discontinued if the mother resumes illicit drug use.
Adolescents
Screen with CRAFFT (Section 2). Adolescent brains are particularly vulnerable to substances due to ongoing prefrontal cortex development (not complete until age ~25). Earlier onset of use is associated with higher lifetime risk of SUD. Family-based therapies (Multidimensional Family Therapy, Functional Family Therapy) are first-line behavioral treatments. Buprenorphine is approved for adolescents ≥16 years with OUD; younger adolescents may receive it off-label with specialist consultation.
Older Adults
Prescription opioid and benzodiazepine misuse is the most common form of SUD in older adults. Screening is often missed due to atypical presentations (confusion attributed to dementia, falls attributed to aging). Lower doses of medications are needed due to decreased hepatic/renal function, reduced lean body mass, and increased sensitivity. AUDIT thresholds may need to be lower (≥5 instead of ≥8) in older adults.
Healthcare Professionals
Physician Health Programs (PHPs) provide confidential monitoring, treatment referral, and advocacy for healthcare professionals with SUD. PHPs have excellent outcomes: 5-year sobriety rates of 70–80%, far exceeding general population outcomes. This is attributed to high motivation (licensure at stake), structured monitoring (random drug testing, group meetings), and professional accountability. Anesthesiologists have the highest rate of opioid use disorder among physician specialties due to occupational exposure.
Healthcare Professionals — PHP Programs in Detail
Physician Health Programs operate in all 50 states and monitor healthcare professionals with SUD or psychiatric illness. The typical PHP contract includes: random urine drug testing (80–100+ tests/year for 5 years), mandatory 12-step or mutual support group attendance (typically 3–5 meetings/week), facilitated peer support groups, workplace monitoring by a colleague, complete abstinence from all intoxicants (including alcohol), restriction of prescribing controlled substances for a defined period, and regular reports to the licensing board. Non-compliance results in license suspension. The PHP model achieves 70–80% long-term sobriety rates because it combines high-intensity monitoring with significant consequences (loss of medical license and career). Anesthesiologists with fentanyl OUD pose a unique challenge: they have the highest relapse rates among physician specialties, and relapse is often fatal due to IV fentanyl access. Many programs recommend career change away from operating room environments for anesthesiologists with fentanyl OUD.
Criminal Justice System
Incarcerated individuals have SUD rates of 50–65%. MOUD should be offered in correctional settings — the first 2 weeks after release from incarceration is the highest-risk period for fatal overdose (tolerance loss during incarceration with return to prior use levels). Multiple court decisions (including Pesce v. Coppinger and Smith v. Aroostook County) have affirmed the right of incarcerated individuals to receive MOUD. Evidence shows that in-jail MOUD initiation reduces post-release overdose mortality by ~60%. Drug courts are specialized court programs that divert non-violent drug offenders from incarceration into supervised treatment; participants have lower recidivism rates (8–14%) compared to traditional adjudication (25–50%). The drug court model combines judicial supervision, mandatory treatment, graduated sanctions and incentives, and frequent drug testing.
Indigenous & Racial/Ethnic Minority Populations
American Indian/Alaska Native populations have the highest rates of SUD and substance-related deaths of any racial/ethnic group in the US, driven by historical trauma, poverty, limited treatment access on reservations, and inadequate IHS funding. Culturally adapted treatments (incorporating traditional healing practices, sweat lodge ceremonies, talking circles, and Indigenous identity strengthening) show improved engagement and outcomes. Black Americans with OUD are significantly less likely to receive buprenorphine compared to White Americans despite similar prevalence of OUD, reflecting structural racism in healthcare access. Hispanic/Latino populations face barriers including language, immigration-related fears, and cultural stigma around addiction treatment.
22 Urine Drug Screens & Confirmatory Testing
Immunoassay (Screening) vs. Confirmatory Testing
Standard urine drug screens (UDS) use immunoassay technology: antibodies recognize drug class metabolites. Advantages: rapid (results in minutes), inexpensive, broad panels available. Limitations: cross-reactivity produces false positives; cannot distinguish specific drugs within a class; cannot quantify levels; certain synthetic opioids are not detected by standard assays.
Confirmatory testing uses gas chromatography-mass spectrometry (GC-MS) or liquid chromatography-tandem mass spectrometry (LC-MS/MS). These identify specific drugs and metabolites with very high sensitivity and specificity. Confirmatory testing should be ordered when clinical decisions depend on the result, when a result is unexpected, or when a patient disputes a positive result.
Standard Panel & Detection Windows
| Substance | Immunoassay Cutoff | Detection Window (Urine) | Notes |
|---|---|---|---|
| Amphetamines | 500–1000 ng/mL | 1–3 days | Detects amphetamine and methamphetamine; may miss MDMA at standard cutoff |
| Benzodiazepines | 200–300 ng/mL | 3 days (short-acting) to 30+ days (chronic diazepam/clonazepam) | Detects oxazepam metabolite; may MISS clonazepam, lorazepam, alprazolam at standard cutoff |
| Cannabinoids (THC-COOH) | 50 ng/mL | Single use: 3–5 days; chronic use: 30+ days (lipophilic storage) | THC-COOH is the metabolite detected; positive result does not indicate current impairment |
| Cocaine (benzoylecgonine) | 150–300 ng/mL | 2–4 days (single use); up to 14 days (heavy use) | Very specific assay; few false positives. Metabolite benzoylecgonine is detected |
| Opiates (morphine/codeine) | 300–2000 ng/mL | 1–3 days | Detects morphine, codeine, heroin (via 6-MAM); does NOT detect synthetic opioids (fentanyl, methadone, tramadol, oxycodone) |
| PCP | 25 ng/mL | 7–14 days (chronic use up to 30 days) | Few false positives; lipophilic with prolonged detection |
Common False Positives
| Reported Positive | Potential Cause of False Positive |
|---|---|
| Amphetamines | Pseudoephedrine, phentermine, bupropion, labetalol, trazodone, ranitidine, selegiline, lisdexamfetamine/prescribed amphetamines |
| Benzodiazepines | Sertraline, efavirenz, oxaprozin |
| Cannabinoids (THC) | Dronabinol (prescribed), hemp-derived CBD products (trace THC), ibuprofen (historical, largely eliminated with modern assays), pantoprazole (rare) |
| Opiates | Poppy seeds (contain morphine/codeine; threshold raised to 2000 ng/mL by federal workplace testing to reduce this), dextromethorphan (can cross-react for PCP as well), rifampin, quinolones |
| PCP | Dextromethorphan, diphenhydramine, ketamine, tramadol, venlafaxine |
EtG/EtS Testing for Alcohol
Ethyl glucuronide (EtG) and ethyl sulfate (EtS) are direct, non-oxidative metabolites of ethanol detectable in urine for 2–3 days after consumption. EtG is highly sensitive but can be positive from incidental alcohol exposure: hand sanitizer (transdermally absorbed ethanol), mouthwash (Listerine), fermented foods, communion wine, or even overripe fruit. Cutoff recommendations: ≥500 ng/mL = recent intentional drinking (within 24 hours); 100–500 ng/mL = possible incidental exposure; <100 ng/mL = negative. EtS is less susceptible to bacterial production artifacts in vitro and is often tested alongside EtG for confirmation. Phosphatidylethanol (PEth) in whole blood is a newer biomarker with a detection window of 2–4 weeks, reflecting cumulative alcohol exposure.
Observed vs. Unobserved Collection
Observed collection (direct visual observation of voiding) is required in federal workplace testing following a positive test and in some treatment/legal settings. Temperature strips on collection cups (90–100°F within 4 minutes) detect substitution. Specimen validity testing includes creatinine (<2 mg/dL = substituted; <20 = dilute), specific gravity (<1.001 or >1.020 = abnormal), pH (normal 4.5–9.0; outside range suggests adulteration), and oxidant assays (detect bleach, nitrites, chromate additives).
Alternative Matrices for Drug Testing
| Matrix | Detection Window | Advantages | Limitations |
|---|---|---|---|
| Oral fluid (saliva) | 24–48 hours | Observed collection (no bathroom needed); detects recent use; difficult to adulterate; correlates with blood levels (impairment) | Short window; affected by oral contamination after recent use; lower sensitivity for cannabis |
| Hair | Up to 90 days (1 cm of hair ≈ 1 month) | Long detection window; difficult to adulterate; documents pattern of use over months | Expensive; cannot detect very recent use (<7–10 days); potential for environmental contamination; racial bias (darker/coarser hair may incorporate more drug) |
| Blood/serum | Hours to days (substance-dependent) | Correlates best with current impairment; definitive for acute intoxication | Invasive; short window; expensive; not practical for routine monitoring |
| Sweat patch | 1–2 weeks (continuous wear) | Monitors continuously over the wearing period; tamper-evident | Limited availability; potential for environmental contamination; cannot distinguish timing of use during wearing period |
| Meconium | Entire third trimester of pregnancy | Detects prenatal substance exposure over months | Available only at birth; legal/ethical implications for the mother |
23 Prescription Drug Monitoring & Safe Prescribing
Prescription Drug Monitoring Programs (PDMPs)
PDMPs are state-run electronic databases that track controlled substance prescriptions. All 50 US states have operational PDMPs. Most states mandate PDMP check before prescribing opioids or benzodiazepines. PDMPs help identify: patients receiving controlled substances from multiple prescribers ("doctor shopping"), escalating doses, dangerous combinations (opioids + benzodiazepines), and prescribers with aberrant patterns.
CDC Clinical Practice Guideline for Prescribing Opioids (2022 Update)
1. Nonopioid therapies are preferred for chronic pain; opioids should only be used when benefits for pain and function are expected to outweigh risks.
2. Before starting opioids, establish realistic treatment goals (functional improvement, not pain elimination) and discuss risks/benefits with the patient.
3. When opioids are started, prescribe the lowest effective dose. Use caution at any dosage; carefully reassess evidence of individual benefits and risks when considering increasing dosage to ≥50 MME/day; avoid increasing to ≥90 MME/day or carefully justify if doing so.
4. For acute pain, prescribe the lowest effective dose of immediate-release opioids; ≤3 days is usually sufficient; >7 days is rarely needed.
5. Prescribe naloxone for patients at risk for overdose: ≥50 MME/day, concurrent benzodiazepines, history of overdose, history of SUD, or per patient request.
6. Check the PDMP before every opioid prescription and periodically during ongoing therapy.
7. Use urine drug testing before initiating opioid therapy and at least annually during ongoing therapy to assess for prescribed medications and illicit substances.
8. Avoid concurrent opioid and benzodiazepine prescribing whenever possible.
9. Offer or arrange evidence-based treatment (usually MOUD) for patients with OUD.
10. Do not abruptly discontinue or rapidly taper opioids in patients who are physically dependent — this increases risk of withdrawal, uncontrolled pain, psychological distress, and illicit opioid use.
Opioid Risk Assessment Tools
| Tool | Items | Purpose | Scoring |
|---|---|---|---|
| ORT (Opioid Risk Tool) | 5 items: family history, personal history of substance abuse, age, history of preadolescent sexual abuse, psychological disease | Pre-prescribing risk stratification | 0–3 = low risk; 4–7 = moderate; ≥8 = high risk for opioid misuse |
| SOAPP-R (Screener & Opioid Assessment for Patients with Pain — Revised) | 24 items (self-report) | Predicts aberrant drug-related behavior in patients being considered for chronic opioid therapy | Score ≥18 = positive screen for increased risk |
| COMM (Current Opioid Misuse Measure) | 17 items (self-report) | Identifies misuse in patients already receiving opioids | Score ≥9 = positive for current misuse |
Naloxone Co-Prescribing
Co-prescribe naloxone when: opioid dose ≥50 MME/day, concurrent benzodiazepine prescribing, history of SUD or overdose, patient request, or other high-risk factors (sleep apnea, renal/hepatic impairment, elderly). Evidence from VA studies shows naloxone co-prescribing is associated with a 47% reduction in opioid-related ED visits.
Opioid Prescribing for Acute Pain — Best Practices
For acute pain (post-surgical, injury, dental), the CDC recommends the lowest effective dose of immediate-release opioids for the shortest expected duration. Specific guidance by condition: Dental pain: NSAIDs ± acetaminophen are first-line; opioids rarely indicated beyond 3 days. Post-surgical: Multimodal analgesia (acetaminophen + NSAID + regional anesthesia) reduces opioid requirements; most patients need ≤3–5 days of opioids after major surgery. Fracture/trauma: Short-acting opioids for 3–7 days with reassessment. Chronic non-cancer pain: Opioids should only be considered after failure of non-opioid pharmacotherapy (NSAIDs, duloxetine, gabapentin, physical therapy, cognitive behavioral therapy for chronic pain). Starting opioids for chronic pain commits the patient to a long-term medication that is difficult to discontinue and may not improve function.
Tapering Patients Off Chronic Opioids
The CDC emphasizes that abrupt discontinuation or rapid taper of opioids in physically dependent patients is dangerous and can lead to withdrawal, psychological crisis, illicit opioid use, and suicide. Recommended approach: reduce by 5–10% of the original dose per month. Slower tapers (5% per month) are better tolerated. Pause the taper if the patient reports increased pain, decreased function, or psychological distress. Adjunctive strategies: start or optimize non-opioid analgesics, initiate CBT for chronic pain, consider buprenorphine transition if the patient has OUD features, provide frequent follow-up and psychosocial support throughout the taper.
24 Levels of Care — ASAM Criteria
The ASAM Criteria (American Society of Addiction Medicine), formerly known as the "ASAM Patient Placement Criteria," is the most widely used and comprehensive set of guidelines for matching patients to appropriate levels and types of addiction treatment. Placement decisions are based on a multidimensional assessment across six dimensions.
Six Assessment Dimensions
| Dimension | Description |
|---|---|
| 1. Acute intoxication and/or withdrawal potential | Current intoxication, risk/severity of withdrawal, need for medical monitoring |
| 2. Biomedical conditions and complications | Co-occurring medical conditions that may complicate treatment (e.g., liver disease, HIV, pregnancy) |
| 3. Emotional, behavioral, or cognitive conditions | Co-occurring psychiatric disorders, cognitive impairment, emotional readiness |
| 4. Readiness to change | Stage of change, motivation, ambivalence, resistance |
| 5. Relapse, continued use, or continued problem potential | Relapse history, coping skills, triggers, relapse prevention plan |
| 6. Recovery/living environment | Social support, housing stability, family dynamics, peer influences, access to substances |
ASAM Levels of Care
| Level | Name | Description | Setting |
|---|---|---|---|
| 0.5 | Early Intervention | Assessment and education for at-risk individuals who do not yet meet diagnostic criteria for SUD; SBIRT | Any healthcare setting, schools, workplace |
| 1 | Outpatient Services | <9 hours/week of treatment services (adults); individual/group therapy, psychoeducation, MOUD | Office, clinic, telehealth |
| 2.1 | Intensive Outpatient (IOP) | 9–19 hours/week (adults); structured programming while patient lives at home or in supportive housing | Outpatient clinic, partial hospital |
| 2.5 | Partial Hospitalization | ≥20 hours/week; day treatment with significant medical/nursing monitoring; patient returns home at night | Freestanding or hospital-based day program |
| 3.1 | Clinically Managed Low-Intensity Residential | 24-hour living support with ≥5 hours of clinical services/week; "halfway house" model; reintegration focus | Residential facility |
| 3.3 | Clinically Managed Population-Specific High-Intensity Residential | 24-hour care for patients with cognitive or functional limitations requiring a slower pace and repetitive engagement | Residential facility with specialized programming |
| 3.5 | Clinically Managed High-Intensity Residential | 24-hour staffing with trained counselors providing a structured therapeutic community; "residential treatment" | Residential treatment center; therapeutic community |
| 3.7 | Medically Monitored Intensive Inpatient | 24-hour nursing care with physician availability; medical and psychiatric management of co-occurring conditions; "inpatient detox/rehab" | Freestanding or hospital-based inpatient unit |
| 4 | Medically Managed Intensive Inpatient | 24-hour medical/nursing care with daily physician management; for severe medical/psychiatric instability requiring acute hospital-level care (e.g., DT, severe withdrawal, acute psychosis with SUD) | Acute care hospital, ICU |
25 Evidence-Based Psychotherapies
| Therapy | Approach | Key Evidence | Best For |
|---|---|---|---|
| Cognitive Behavioral Therapy (CBT) | Identifies and modifies maladaptive thought patterns and behaviors; teaches coping skills, trigger management, refusal skills | Strong evidence across all substances; effects maintained after treatment ends (skill acquisition) | All SUDs; co-occurring anxiety and depression |
| Motivational Interviewing / Motivational Enhancement Therapy (MI/MET) | Resolves ambivalence, enhances intrinsic motivation for change using OARS and MI spirit | Effective as a brief intervention and as a treatment modality; especially effective early in treatment and for less severe SUD | Ambivalent patients; cannabis, alcohol, and multi-substance use |
| Contingency Management (CM) | Provides tangible reinforcement (vouchers, prizes) for verified positive behaviors (abstinence, treatment attendance) | Largest effect sizes for stimulant use disorder; effective for opioid, cannabis, tobacco, and alcohol use disorders | Stimulant use disorder (first-line); treatment adherence |
| 12-Step Facilitation (TSF) | Structured therapy encouraging active involvement in 12-step mutual support groups (AA, NA); acceptance of addiction, surrender, active involvement | Project MATCH: TSF was as effective as CBT and MET; Cochrane 2020 review: AA/TSF produces higher rates of continuous abstinence than other treatments at 12 months | Alcohol use disorder; patients open to group support and spiritual framework |
| Community Reinforcement Approach (CRA) | Rearranges environmental contingencies so that sober behavior is more rewarding than substance use; addresses employment, social, and recreational functioning | Strong evidence for AUD and cocaine use disorder, especially when combined with voucher-based CM (CRA + vouchers) | Cocaine use disorder; AUD; patients with significant functional impairment |
| Matrix Model | Intensive structured 16-week outpatient program combining CBT, MI, 12-step, family education, relapse prevention, and drug testing | Developed for methamphetamine use disorder; NIDA-funded RCTs show reduced use during treatment | Stimulant use disorder |
| Mindfulness-Based Relapse Prevention (MBRP) | Integrates mindfulness meditation with cognitive-behavioral relapse prevention; "urge surfing" — observe craving without acting on it | RCTs show reduced heavy drinking days, drug use days, and craving at 6–12 months; equivalent or superior to standard relapse prevention | Relapse prevention for patients in early recovery from any SUD |
Family-Based Therapies
Multidimensional Family Therapy (MDFT): Evidence-based for adolescent SUD; addresses individual, family, peer, and community systems simultaneously over 4–6 months. Functional Family Therapy (FFT): Short-term (12–14 sessions) for youth with behavioral problems and SUD; focuses on improving family communication and reducing negative interaction patterns. CRAFT (Community Reinforcement and Family Training): Trains concerned family members of treatment-resistant individuals in behavioral strategies to encourage their loved one to enter treatment; 64–74% treatment entry rate vs. 30% with Al-Anon and 13% with Johnson intervention. Behavioral Couples Therapy (BCT): For patients with SUD and a committed partner; incorporates a recovery contract with partner-observed medication taking and couples communication skills; strong evidence for reducing substance use and improving relationship functioning simultaneously.
26 Classification Systems & Scoring Tools
This section consolidates all clinical scoring instruments referenced throughout the page with scoring thresholds and clinical application. Each tool is validated for specific populations and clinical contexts; using the correct tool in the correct setting improves diagnostic accuracy and treatment decisions.
Primary care screening: AUDIT-C (alcohol), NIDA Quick Screen (drugs), FTND (tobacco), CRAFFT (adolescents 12–21).
Detailed assessment after positive screen: Full AUDIT (alcohol), DAST-10 (drugs), DSM-5 criteria (formal diagnosis).
Withdrawal management: CIWA-Ar (alcohol), COWS (opioid), Modified Finnegan (neonatal).
Opioid prescribing risk: ORT (pre-prescribing), SOAPP-R (pre-prescribing, more detailed), COMM (during opioid therapy).
Treatment placement: ASAM Criteria 6-dimensional assessment.
Nicotine dependence: FTND to guide pharmacotherapy intensity.
Liver disease severity: Maddrey DF, MELD for alcoholic hepatitis prognosis.
| Tool | Items | Score Range | Key Thresholds | Application |
|---|---|---|---|---|
| CIWA-Ar | 10 | 0–67 | <10 mild; 10–18 moderate; >18 severe; >35 very severe/DT risk | Alcohol withdrawal severity; guides benzodiazepine dosing |
| COWS | 11 | 0–48 | 5–12 mild; 13–24 moderate; 25–36 moderately severe; >36 severe | Opioid withdrawal severity; guides buprenorphine induction timing |
| Finnegan (Modified) | 31 items in 3 domains | Variable | ≥8 on 3 consecutive scores → pharmacotherapy | Neonatal opioid withdrawal scoring; guides NAS treatment |
| AUDIT | 10 | 0–40 | 0–7 low risk; 8–15 hazardous; 16–19 harmful; ≥20 possible dependence | Alcohol use screening; validated in diverse populations |
| AUDIT-C | 3 | 0–12 | ≥4 men / ≥3 women = positive | Abbreviated alcohol screening for primary care |
| CAGE | 4 | 0–4 | ≥2 = positive screen | Quick alcohol screening; low sensitivity in some populations |
| DAST-10 | 10 | 0–10 | 0 none; 1–2 low; 3–5 moderate; 6–8 substantial; 9–10 severe | Drug use screening (excluding alcohol/tobacco) |
| CRAFFT | 6 | 0–6 | ≥2 = positive screen | Adolescent substance use screening (ages 12–21) |
| FTND (Fagerstrom) | 6 | 0–10 | 0–2 very low; 3–4 low; 5 medium; 6–7 high; 8–10 very high | Nicotine dependence severity |
| ORT | 5 | 0–26 | 0–3 low; 4–7 moderate; ≥8 high risk | Pre-prescribing opioid misuse risk |
| SOAPP-R | 24 | 0–96 | ≥18 = positive screen | Predicts aberrant opioid-related behavior |
| COMM | 17 | 0–68 | ≥9 = current misuse | Identifies misuse in patients already on opioids |
| Maddrey DF | Formula | Continuous | ≥32 = severe alcoholic hepatitis (consider steroids) | Alcoholic hepatitis severity/prognosis |
| ASAM Criteria | 6 dimensions | Qualitative | Levels 0.5 through 4 | Treatment placement matching for SUD |
27 Medications Master Table
Medications for Alcohol Use Disorder
| Drug | Class | Mechanism | Dose | Key Side Effects | Monitoring |
|---|---|---|---|---|---|
| Naltrexone PO | Opioid antagonist | Mu-opioid receptor blockade | 50 mg daily | Nausea, headache, dizziness, hepatotoxicity (rare) | LFTs at baseline and periodically; ensure opioid-free ≥7 days |
| Naltrexone IM (Vivitrol) | Opioid antagonist | Same as oral; depot formulation | 380 mg IM q4 weeks | Injection site reactions, nausea, headache | Same as oral; verify no opioid use |
| Acamprosate | Glutamate modulator | NMDA antagonism, GABA agonism | 666 mg PO TID | Diarrhea, nausea | Renal function (contraindicated CrCl <30) |
| Disulfiram | Aldehyde dehydrogenase inhibitor | Blocks acetaldehyde metabolism | 250 mg PO daily | Disulfiram-ethanol reaction, hepatotoxicity, peripheral neuropathy | LFTs; patient education; supervised dosing preferred |
| Gabapentin | Calcium channel modulator | Alpha-2-delta subunit binding | 900–1800 mg/day divided TID | Sedation, dizziness, peripheral edema | Renal function; misuse potential assessment |
| Topiramate | Anticonvulsant | GABA enhancement, glutamate inhibition | 200–300 mg/day divided BID | Cognitive impairment, paresthesias, weight loss, kidney stones | Bicarbonate (metabolic acidosis); pregnancy test (teratogenic) |
Medications for Opioid Use Disorder
| Drug | Class | Mechanism | Dose | Key Side Effects | Monitoring |
|---|---|---|---|---|---|
| Buprenorphine/naloxone SL | Partial mu agonist + antagonist | High-affinity partial mu agonism; naloxone deters IV misuse | 8–24 mg/day SL | Headache, nausea, constipation, insomnia, diaphoresis | UDS, PDMP, pregnancy test, LFTs |
| Buprenorphine XR SC (Sublocade) | Partial mu agonist | Same; monthly depot injection | 300 mg SC monthly × 2, then 100–300 mg monthly | Injection site reactions, constipation, nausea | Same as SL; observe injection technique |
| Methadone | Full mu agonist | Mu-opioid agonism + NMDA antagonism | 60–120 mg/day PO (start 20–30 mg) | QTc prolongation, respiratory depression, constipation, sedation, diaphoresis | ECG (baseline, 30 days, annual); UDS; PDMP; CYP interactions |
| Naltrexone PO | Opioid antagonist | Competitive mu-opioid blockade | 50 mg daily | Nausea, hepatotoxicity (rare), injection site reactions (IM) | Must be opioid-free ≥7–10 days; LFTs; naloxone challenge if uncertain |
| Naltrexone IM (Vivitrol) | Opioid antagonist | Same; monthly depot | 380 mg IM q4 weeks | Injection site reactions, nausea, headache | Same as oral; gluteal injection only |
| Naloxone | Opioid antagonist | Competitive mu-opioid antagonism | 4 mg IN or 0.4–2 mg IM/IV q2–3 min PRN | Precipitated withdrawal, tachycardia, hypertension | Monitor for renarcotization; observe ≥2 hours after last dose |
Medications for Tobacco Use Disorder
| Drug | Class | Mechanism | Dose | Key Side Effects | Notes |
|---|---|---|---|---|---|
| NRT Patch | Nicotine replacement | Provides steady-state nicotine | 21/14/7 mg stepped taper over 8–12 weeks | Skin irritation, insomnia, vivid dreams | Combine with short-acting NRT for best results |
| NRT Gum/Lozenge | Nicotine replacement | Rapid oral nicotine absorption | 2–4 mg q1–2h PRN | Jaw soreness (gum), hiccups, nausea | "Chew and park" technique; avoid acidic beverages |
| Varenicline | Partial nAChR agonist | Partial alpha-4-beta-2 agonism | 0.5 mg daily → 1 mg BID over 1 week; maintain 12–24 weeks | Nausea (30%), insomnia, vivid dreams | Most effective single agent; FDA removed neuropsych black box (2016) |
| Bupropion SR | NDRI | NE/DA reuptake inhibition | 150 mg daily × 3 days, then 150 mg BID | Insomnia, dry mouth, seizure risk | Contraindicated in seizure disorders, eating disorders |
Adjunctive Medications for Withdrawal
| Drug | Indication | Dose | Notes |
|---|---|---|---|
| Clonidine | Opioid withdrawal (autonomic symptoms) | 0.1–0.3 mg PO q6–8h | Monitor for hypotension, bradycardia |
| Loperamide | Opioid withdrawal (diarrhea) | 4 mg initially, then 2 mg PRN (max 16 mg/day) | Peripheral mu agonist; QTc risk at supratherapeutic doses |
| Ondansetron | Nausea/vomiting in withdrawal | 4–8 mg PO/IV q8h PRN | 5-HT3 antagonist |
| Thiamine | Wernicke prophylaxis/treatment | Prophylaxis: 250–500 mg IV daily; Treatment: 500 mg IV TID | Always give before glucose in malnourished patients |
| Fomepizole | Methanol/ethylene glycol poisoning | 15 mg/kg IV load, then 10 mg/kg q12h | ADH inhibitor; first-line antidote for toxic alcohols |
28 MME Conversion Table
The morphine milligram equivalent (MME) is a standardized measure used to compare potency across different opioid medications. It represents the dose of a given opioid that is approximately equivalent to 1 mg of oral morphine. The 2022 CDC guideline uses MME thresholds for risk assessment: use caution at ≥50 MME/day and avoid or carefully justify ≥90 MME/day.
| Opioid | MME Conversion Factor | Example: 30 MME Equivalent Dose |
|---|---|---|
| Codeine | 0.15 | 200 mg |
| Tramadol | 0.1 | 300 mg |
| Hydrocodone | 1.0 | 30 mg |
| Morphine (oral) | 1.0 (reference) | 30 mg |
| Oxycodone | 1.5 | 20 mg |
| Oxymorphone | 3.0 | 10 mg |
| Hydromorphone | 4.0 | 7.5 mg |
| Tapentadol | 0.4 | 75 mg |
| Methadone (1–20 mg/day) | 4.0 | 7.5 mg |
| Methadone (21–40 mg/day) | 8.0 | 3.75 mg |
| Methadone (41–60 mg/day) | 10.0 | 3 mg |
| Methadone (>60 mg/day) | 12.0 | 2.5 mg |
| Fentanyl transdermal (mcg/hr) | 2.4 (per mcg/hr) | 12.5 mcg/hr patch |
Patient taking oxycodone 10 mg QID + hydrocodone 5 mg TID:
Oxycodone: 10 mg × 4 doses = 40 mg/day × 1.5 (conversion factor) = 60 MME
Hydrocodone: 5 mg × 3 doses = 15 mg/day × 1.0 = 15 MME
Total: 75 MME/day — above the 50 MME caution threshold; consider naloxone co-prescribing, reassess pain management plan, and explore non-opioid alternatives.
1. Calculate the current total daily MME.
2. Convert to the new opioid using published equianalgesic ratios.
3. Reduce the calculated dose by 25–50% to account for incomplete cross-tolerance (the patient is not equally tolerant to the new opioid). For methadone rotations, reduce by ≥75–90% given its unique pharmacology.
4. Provide breakthrough medication (5–15% of total daily dose q2–4h PRN) during the transition period.
5. Titrate cautiously over days to the effective dose, increasing by no more than 25% per day.
6. Consider clinical context: Reduce more aggressively in elderly patients, those with renal/hepatic impairment, or when converting from high-dose opioids. All equianalgesic tables are approximations; clinical judgment and close monitoring are essential.
29 Drug Interaction Reference
Methadone Drug Interactions
| Interacting Drug | Effect on Methadone | Clinical Consequence | Management |
|---|---|---|---|
| Rifampin | Potent CYP3A4 induction; reduces methadone levels by >50% | Precipitated withdrawal within days | Increase methadone dose; consider split dosing; monitor closely during and after rifampin course |
| Phenytoin / Carbamazepine / Phenobarbital | CYP3A4 induction | Reduced methadone levels, withdrawal symptoms | Increase methadone dose; consider alternative anticonvulsants (levetiracetam, valproate) |
| Efavirenz / Nevirapine | CYP3A4 induction | Reduced methadone levels; withdrawal symptoms | Increase methadone; consider alternative ART regimens |
| Fluconazole / Ketoconazole | CYP3A4 inhibition; increases methadone levels | Sedation, respiratory depression, QTc prolongation | Monitor for toxicity; may need to decrease methadone dose |
| Ciprofloxacin | CYP1A2 inhibition; increases methadone levels | Toxicity risk | Monitor; use alternative fluoroquinolone if possible |
| QTc-prolonging drugs (antipsychotics, macrolides, fluoroquinolones, TCAs) | Additive QTc prolongation | Risk of torsades de pointes | ECG monitoring; avoid combinations when possible; if QTc >500 ms, reduce/change medications |
| Benzodiazepines | Additive CNS/respiratory depression | Increased overdose risk | Avoid combination when possible; if required, start low, increase slowly, use short-acting agents, educate patient |
Buprenorphine Drug Interactions
| Interacting Drug | Effect | Management |
|---|---|---|
| Full mu agonists (heroin, fentanyl, oxycodone) | Buprenorphine blocks their effects due to higher receptor affinity; precipitates withdrawal if given during active opioid use | Ensure adequate COWS score before standard induction; use micro-dosing for fentanyl users |
| Benzodiazepines / alcohol | Increased sedation and respiratory depression (though less than with full agonists) | FDA black box warning; do NOT withhold buprenorphine solely because of benzodiazepine use; counsel patients on risk; consider taper of benzodiazepines |
| CYP3A4 inhibitors (ketoconazole, ritonavir) | Increased buprenorphine levels | Monitor for over-sedation; dose reduction may be needed |
| CYP3A4 inducers (rifampin, phenytoin, carbamazepine) | Decreased buprenorphine levels | May need dose increase; monitor for breakthrough withdrawal/craving |
| Naltrexone | Blocks buprenorphine effects; if given after naltrexone wears off, no precipitated withdrawal (buprenorphine is a partial agonist) | Wait ≥7 days after last naltrexone dose before starting buprenorphine (14 days for IM naltrexone) |
Disulfiram-Ethanol Reaction
Mechanism: Disulfiram irreversibly inhibits aldehyde dehydrogenase (ALDH). When ethanol is consumed, acetaldehyde accumulates to 5–10× normal levels, producing a toxic syndrome.
Onset: 10–30 minutes after alcohol ingestion (can occur up to 2 weeks after last disulfiram dose due to irreversible enzyme inhibition).
Symptoms: Facial flushing, throbbing headache, nausea, vomiting, diaphoresis, chest pain, palpitations, tachycardia, hypotension, dyspnea, confusion.
Severe reaction: Cardiovascular collapse, arrhythmias, seizures, respiratory depression, death (rare, typically with large alcohol intake or high disulfiram dose).
Management: Supportive — IV fluids, supine positioning, antiemetics, H1/H2 blockers for flushing. Severe hypotension: IV fluids, norepinephrine. Most reactions are self-limited (30–60 minutes).
Warning: Patients must avoid all alcohol sources: beverages, cooking wine, alcohol-containing medications (cough syrups, mouthwash), vinegar, aftershave (topical), and even hand sanitizer in sensitive individuals.
Serotonin Syndrome Risk Combinations
Several substances encountered in addiction medicine carry serotonin syndrome risk when combined with serotonergic medications:
| High-Risk Combination | Mechanism | Clinical Context |
|---|---|---|
| MDMA + MAOIs | Massive serotonin release + blocked metabolism | Potentially fatal; most dangerous combination |
| MDMA + SSRIs/SNRIs | Dual serotonin reuptake inhibition + release | Recreational MDMA use in patients on antidepressants |
| Tramadol + SSRIs | Tramadol is a weak SNRI + mu agonist | Common in pain patients on antidepressants |
| Fentanyl + SSRIs (high doses) | Fentanyl has weak serotonergic activity at high doses | Generally low risk at therapeutic doses; higher risk with illicit high-dose fentanyl |
| Dextromethorphan + SSRIs/MAOIs | DXM inhibits serotonin reuptake | OTC cough suppressant; often overlooked interaction |
| Methadone + SSRIs | Methadone has mild serotonergic activity | Very common co-prescription; usually low risk but monitor |
| Linezolid + serotonergic agent | Linezolid is a reversible MAO inhibitor | Antibiotic prescribed for MRSA; often missed interaction |
Recognition: The Hunter Serotonin Toxicity Criteria require a serotonergic agent plus any of: spontaneous clonus, inducible clonus + agitation/diaphoresis, ocular clonus + agitation/diaphoresis, tremor + hyperreflexia, or hypertonia + temperature >38°C + clonus. The classic triad is altered mental status, autonomic instability, and neuromuscular hyperactivity (clonus > rigidity, hyperreflexia). Treatment: Discontinue all serotonergic agents; benzodiazepines for agitation; cyproheptadine (5-HT2A antagonist) 12 mg PO loading, then 2 mg q2h PRN (max 32 mg/day); active cooling for hyperthermia; IV fluids; severe cases may require intubation, paralysis, and ICU-level care. Avoid dantrolene (only for NMS, not serotonin syndrome).
30 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| AA | Alcoholics Anonymous |
| ADH | Alcohol dehydrogenase |
| ALDH | Aldehyde dehydrogenase |
| ASAM | American Society of Addiction Medicine |
| AUD | Alcohol use disorder |
| AUDIT | Alcohol Use Disorders Identification Test |
| BAC | Blood alcohol concentration |
| BBB | Blood-brain barrier |
| BID | Twice daily (bis in die) |
| CAGE | Cut down, Annoyed, Guilty, Eye-opener |
| CB1/CB2 | Cannabinoid receptor type 1/2 |
| CBT | Cognitive behavioral therapy |
| CDC | Centers for Disease Control and Prevention |
| CDT | Carbohydrate-deficient transferrin |
| CHS | Cannabinoid hyperemesis syndrome |
| CIWA-Ar | Clinical Institute Withdrawal Assessment for Alcohol, Revised |
| CM | Contingency management |
| COMM | Current Opioid Misuse Measure |
| COWS | Clinical Opiate Withdrawal Scale |
| CRAFFT | Car, Relax, Alone, Forget, Family/Friends, Trouble |
| CrCl | Creatinine clearance |
| CYP | Cytochrome P450 |
| DAST | Drug Abuse Screening Test |
| DAT | Dopamine transporter |
| DEA | Drug Enforcement Administration |
| DOR | Delta opioid receptor |
| DSM-5 | Diagnostic and Statistical Manual of Mental Disorders, 5th edition |
| DT | Delirium tremens |
| ECG/EKG | Electrocardiogram |
| ED | Emergency department |
| ENDS | Electronic nicotine delivery system |
| EtG | Ethyl glucuronide |
| EtOH | Ethanol (alcohol) |
| EtS | Ethyl sulfate |
| FDA | Food and Drug Administration |
| FTND | Fagerstrom Test for Nicotine Dependence |
| GABA | Gamma-aminobutyric acid |
| GC-MS | Gas chromatography-mass spectrometry |
| GGT | Gamma-glutamyl transferase |
| HCV | Hepatitis C virus |
| hERG | Human ether-a-go-go-related gene (potassium channel) |
| HIV | Human immunodeficiency virus |
| HPPD | Hallucinogen persisting perception disorder |
| IOP | Intensive outpatient program |
| IV | Intravenous |
| KOR | Kappa opioid receptor |
| LC-MS/MS | Liquid chromatography-tandem mass spectrometry |
| LFT | Liver function test |
| LSD | Lysergic acid diethylamide |
| MAO | Monoamine oxidase |
| MAT | Medication-assisted treatment (older term; MOUD preferred) |
| MBRP | Mindfulness-based relapse prevention |
| MCV | Mean corpuscular volume |
| MDF | Maddrey discriminant function |
| MDMA | 3,4-methylenedioxymethamphetamine |
| MELD | Model for End-Stage Liver Disease |
| MET | Motivational enhancement therapy |
| MI | Motivational interviewing |
| MME | Morphine milligram equivalent |
| MOR | Mu opioid receptor |
| MOUD | Medications for opioid use disorder |
| NA | Narcotics Anonymous |
| NAc | Nucleus accumbens |
| nAChR | Nicotinic acetylcholine receptor |
| NAS | Neonatal abstinence syndrome |
| NET | Norepinephrine transporter |
| NIDA | National Institute on Drug Abuse |
| NMDA | N-methyl-D-aspartate |
| NOWS | Neonatal opioid withdrawal syndrome |
| NRT | Nicotine replacement therapy |
| OARS | Open questions, Affirmations, Reflections, Summaries |
| ORT | Opioid Risk Tool |
| OTP | Opioid Treatment Program |
| OUD | Opioid use disorder |
| PCP | Phencyclidine |
| PDMP | Prescription Drug Monitoring Program |
| PEth | Phosphatidylethanol |
| PFC | Prefrontal cortex |
| PHP | Physician Health Program |
| PO | By mouth (per os) |
| PRN | As needed (pro re nata) |
| QTc | Corrected QT interval |
| SAMHSA | Substance Abuse and Mental Health Services Administration |
| SBIRT | Screening, Brief Intervention, Referral to Treatment |
| SC | Subcutaneous |
| SERT | Serotonin transporter |
| SL | Sublingual |
| SOAPP-R | Screener and Opioid Assessment for Patients with Pain — Revised |
| SSP | Syringe services program |
| SUD | Substance use disorder |
| THC | Tetrahydrocannabinol |
| TID | Three times daily (ter in die) |
| TSF | Twelve-step facilitation |
| UDS | Urine drug screen |
| USPSTF | US Preventive Services Task Force |
| VMAT2 | Vesicular monoamine transporter 2 |
| VTA | Ventral tegmental area |