Child & Adolescent Psychiatry
Neurodevelopmental disorders, pediatric mood and anxiety disorders, ADHD, autism spectrum, eating disorders, psychopharmacology in youth, evidence-based therapies, and every screening tool, dosing guideline, and developmental milestone across the full scope of child and adolescent psychiatry.
01 Neurodevelopment & Brain Maturation
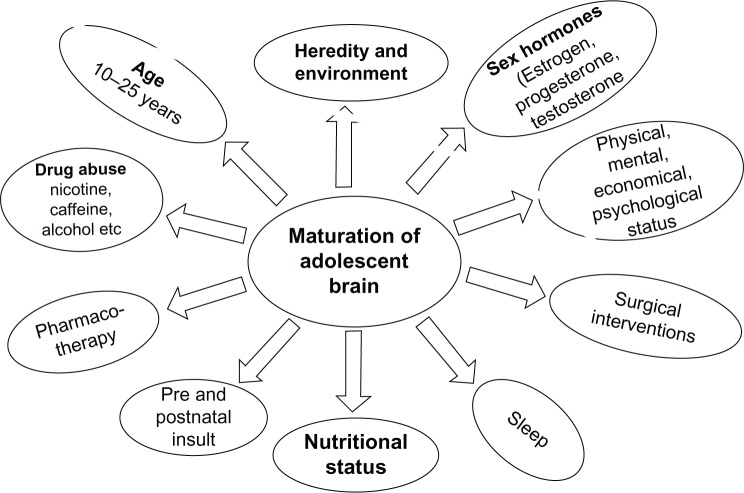
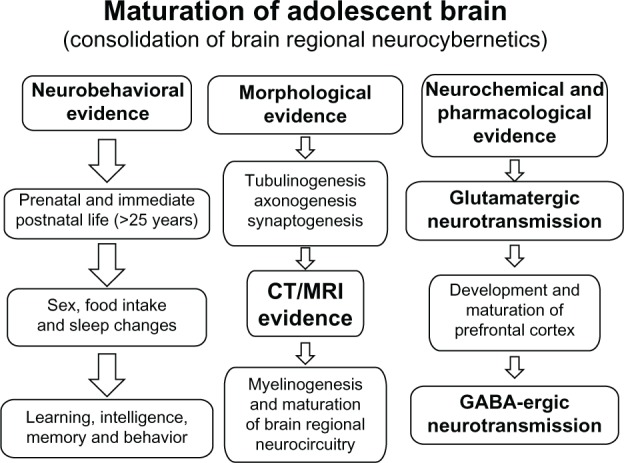
The developing brain undergoes a predictable sequence of structural and functional changes from gestation through early adulthood. Understanding these trajectories is essential for recognizing deviation from typical development and for appreciating why psychiatric disorders manifest at particular ages. Psychiatric symptoms in children often represent the interaction between normal neurodevelopmental processes and genetic, epigenetic, and environmental risk factors.
Neuronal Proliferation & Migration
During weeks 8–16 of gestation, neuronal proliferation in the ventricular zone and subventricular zone generates roughly 250,000 neurons per minute. Radial glial cells guide neuronal migration from the ventricular zone to the cortical plate in an "inside-out" pattern: deeper cortical layers (V–VI) form first, while superficial layers (II–III) form later. Disruption of migration during this critical period produces lissencephaly, heterotopias, or polymicrogyria — malformations frequently associated with intellectual disability and epilepsy.
Synaptogenesis & Synaptic Pruning
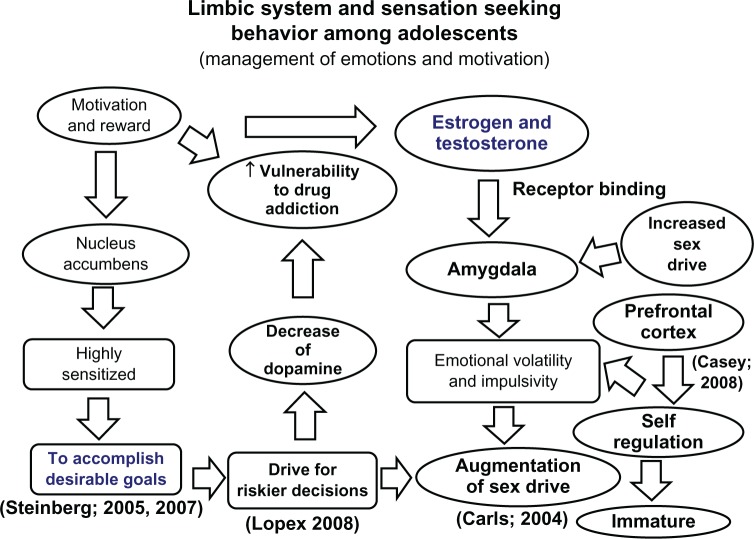
Synaptogenesis begins in the third trimester and peaks during the first 2–3 years of life, reaching densities 50% higher than adult levels in many cortical regions. The infant brain forms approximately 700–1,000 new synaptic connections per second during this period. Synaptic pruning then refines circuits through activity-dependent elimination of weaker synapses (Hebbian principle: "neurons that fire together wire together"). Sensory cortex pruning largely completes by middle childhood, while prefrontal cortex pruning continues through the mid-20s. This prolonged pruning window in prefrontal regions explains adolescent vulnerability to impulsive decision-making and risk-taking.
Myelination Timeline
Myelination follows a posterior-to-anterior, inferior-to-superior gradient. Major milestones include:
| Structure/Region | Myelination Window | Clinical Relevance |
|---|---|---|
| Brainstem & cerebellum | Prenatal – 1 year | Supports basic motor reflexes, vital functions |
| Internal capsule & motor tracts | Birth – 2 years | Gross and fine motor milestone acquisition |
| Sensory cortex (visual, auditory) | Birth – 5 years | Sensitive periods for vision/hearing; amblyopia risk |
| Language areas (Broca's, Wernicke's) | 1 – 10 years | Language acquisition windows; early intervention importance |
| Association cortex (parietal, temporal) | 5 – 15 years | Abstract reasoning, reading comprehension development |
| Prefrontal cortex (PFC) | Adolescence – age 25 | Executive function, impulse control, judgment maturation |
Prefrontal Cortex Development
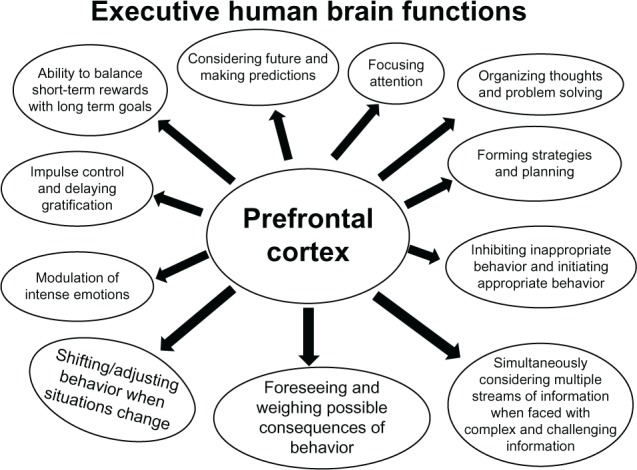
The dorsolateral PFC (dlPFC) subserves working memory, planning, and cognitive flexibility. The ventromedial PFC (vmPFC) integrates emotional valence into decision-making. The orbitofrontal cortex (OFC) processes reward and social cues. Gray matter volume in these regions peaks around age 11–12 and then declines as pruning eliminates excess synapses. White matter volume increases linearly through the mid-20s, reflecting ongoing myelination and improved connectivity between frontal and subcortical structures. Functional MRI studies show progressively increasing frontal-to-subcortical connectivity from childhood through adulthood, supporting improved emotional regulation and executive control.
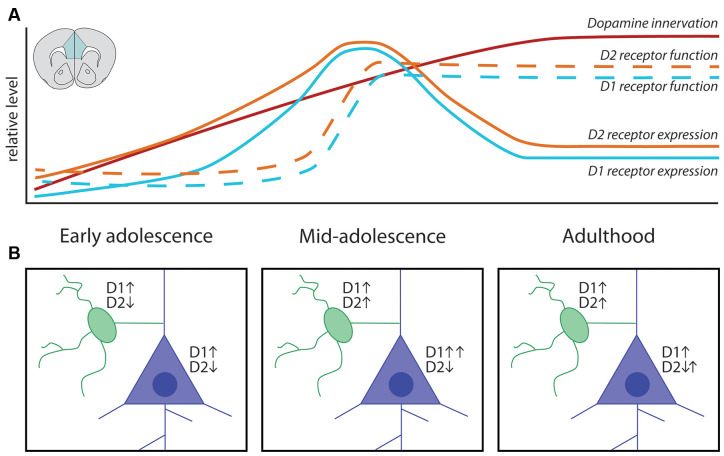
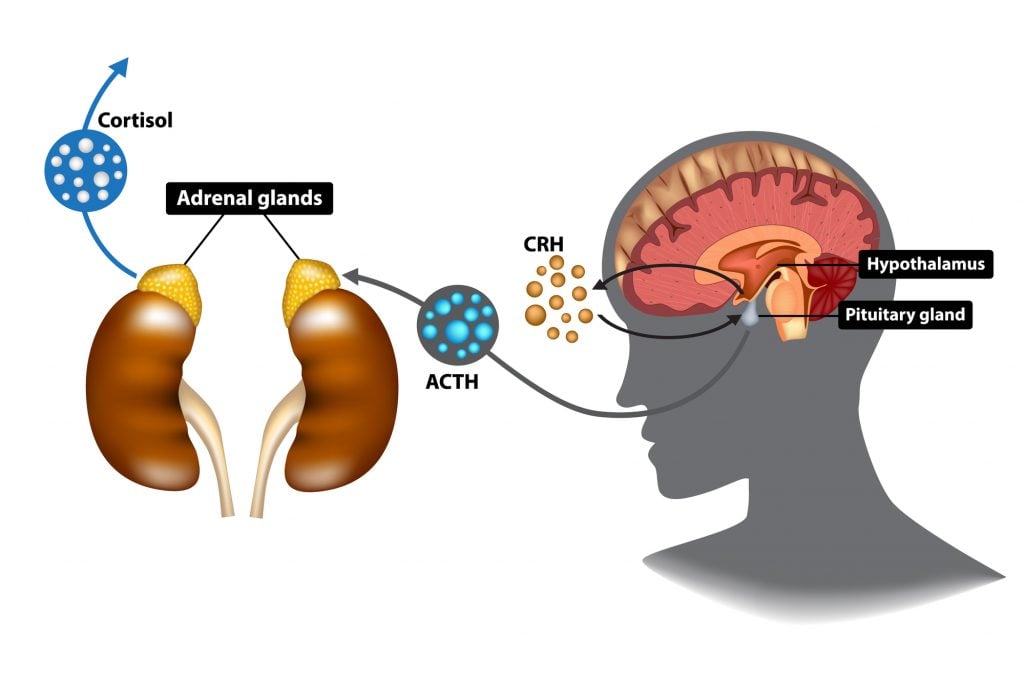
HPA Axis Development
The hypothalamic-pituitary-adrenal (HPA) axis is functional by the third trimester. Neonates exhibit a robust cortisol response to stress but also a hyporesponsive period from approximately 3–12 months during which cortisol responses are dampened when adequate caregiving is present. Secure attachment relationships serve as a biological buffer, regulating the infant's stress response system. Early adversity (neglect, abuse, institutional care) can dysregulate the HPA axis, producing either blunted cortisol responses (chronic stress adaptation) or hyperresponsive patterns. These alterations are associated with increased risk for anxiety, depression, and PTSD in later childhood and are a biological mechanism behind the effects of Adverse Childhood Experiences (ACEs).
Sensitive & Critical Periods
A critical period is a time window during which specific input is required for normal development (e.g., binocular vision by age 6–8). A sensitive period is a broader window during which the brain is particularly responsive to environmental input but can still develop capacity outside it. Language acquisition has a sensitive period peaking before age 5–7, after which acquisition of native-like proficiency becomes progressively harder. Attachment formation has a sensitive period in the first 2–3 years. Social cognition, emotional regulation, and executive function continue to have sensitive periods extending through adolescence, making intervention in these domains potentially effective well into the teenage years.
1. Brain development follows a predictable sequence; deviations signal pathology. 2. Synaptic pruning is experience-dependent — environmental enrichment strengthens adaptive circuits. 3. The prefrontal cortex develops last, explaining adolescent impulsivity. 4. Early adversity can permanently alter stress-response systems. 5. Sensitive periods exist for language, attachment, and emotional regulation — early intervention yields the greatest benefit.
02 Developmental Milestones & Assessment
Developmental surveillance is a core competency in child psychiatry. Milestones represent median ages of acquisition; significant delay in any domain warrants formal evaluation. The four primary developmental domains are gross motor, fine motor, language (receptive and expressive), and social-emotional/cognitive.
Motor Milestones
| Age | Gross Motor | Fine Motor |
|---|---|---|
| 2 months | Lifts head prone, some head control | Hands mostly fisted |
| 4 months | Rolls front to back, steady head control | Reaches for objects, hands open |
| 6 months | Sits with support, rolls both ways | Raking grasp, transfers objects hand-to-hand |
| 9 months | Sits without support, crawls | Pincer grasp developing, bangs objects |
| 12 months | Pulls to stand, cruises furniture, walks with one hand held | Mature pincer grasp, puts objects in container |
| 15 months | Walks independently | Scribbles, stacks 2 blocks |
| 18 months | Runs clumsily, walks up stairs with hand held | Stacks 3–4 blocks, removes clothing |
| 2 years | Runs well, kicks ball, walks up/down stairs | Stacks 6 blocks, turns pages, vertical line |
| 3 years | Pedals tricycle, walks up stairs alternating feet | Copies circle, stacks 9 blocks, uses scissors |
| 4 years | Hops on one foot, climbs well | Copies cross, draws person with 3 parts |
| 5 years | Skips, catches ball | Copies square, draws person with 6 parts, ties shoes |
Language Milestones
| Age | Receptive Language | Expressive Language |
|---|---|---|
| 2 months | Alerts to sound, social smile | Cooing |
| 6 months | Turns to voice, responds to name | Babbling (consonant-vowel combinations) |
| 9 months | Understands "no," responds to own name consistently | Babbling with inflection ("mama/dada" nonspecific) |
| 12 months | Follows simple commands with gesture | 1–3 specific words, "mama/dada" specific |
| 15 months | Points to body parts on request | 4–6 words, uses jargon with true words |
| 18 months | Identifies pictures when named | 10–25 words, vocabulary spurt begins |
| 2 years | Follows 2-step commands | 50+ words, 2-word phrases, 50% intelligible to strangers |
| 3 years | Understands prepositions (in, on, under) | 3-word sentences, 75% intelligible, uses pronouns |
| 4 years | Follows 3-step commands | 4–5 word sentences, 100% intelligible, tells stories |
| 5 years | Understands complex/conditional sentences | Full sentences, correct grammar mostly, names colors |
Social-Emotional & Cognitive Milestones
| Age | Social-Emotional | Cognitive |
|---|---|---|
| 2 months | Social smile | Tracks past midline, regards faces |
| 6 months | Stranger anxiety emerging, laughs | Object permanence developing |
| 9 months | Separation anxiety, joint attention emerging | Object permanence established, looks for dropped object |
| 12 months | Waves bye-bye, shows objects to others | Cause-and-effect play, explores objects |
| 18 months | Parallel play, early empathy | Symbolic play begins, uses objects as tools |
| 2 years | Parallel play, says "mine," emerging autonomy | Pretend play, sorts shapes, completes 3-piece puzzle |
| 3 years | Interactive play, takes turns, knows gender identity | Counts to 3, knows full name and age |
| 4 years | Cooperative play, has friends, imagination | Counts to 10, understands "same/different" |
| 5 years | Understanding of rules, follows game rules | Counts to 20+, beginning reading readiness |
Red Flags Requiring Immediate Evaluation
Any age: Loss of previously acquired skills (regression). 4 months: No social smile, no head control. 6 months: No reaching, no babbling, persistent fisting. 9 months: No sitting, no consonant babbling. 12 months: No words, no gestures (pointing, waving), no response to name. 15 months: Not walking. 18 months: Fewer than 6 words, no pointing, no pretend play. 24 months: Fewer than 50 words, no 2-word phrases, no imitation. 36 months: Speech unintelligible to strangers, no 3-word sentences, no interactive play.
Screening Instruments
The Denver Developmental Screening Test II (Denver II) evaluates four domains (gross motor, fine motor-adaptive, language, personal-social) in children 0–6 years using 125 items. Items are scored as "pass," "fail," "caution," or "delayed" based on age-normed percentiles. Two or more delays in any sector warrants referral. The Ages and Stages Questionnaire, 3rd Edition (ASQ-3) is a parent-completed screener for children 1–66 months covering communication, gross motor, fine motor, problem-solving, and personal-social domains; scores below cutoffs (2 SDs below the mean for the child's age) in any domain trigger referral. The Modified Checklist for Autism in Toddlers, Revised with Follow-Up (M-CHAT-R/F) is a 20-item parent-report screen recommended at 18 and 24 months: a score of 0–2 is low risk, 3–7 is medium risk (administer follow-up interview), and 8–20 is high risk (refer directly). The follow-up interview reduces the false-positive rate from ~50% to ~20%.
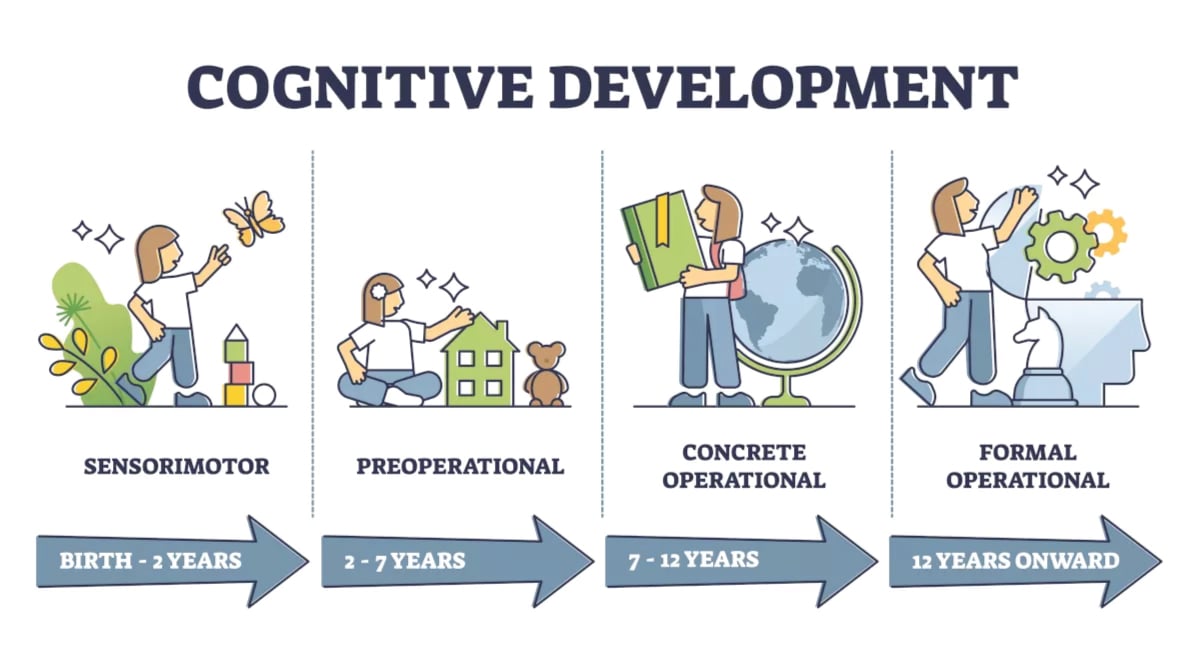
Piaget's Stages of Cognitive Development
| Stage | Age Range | Key Characteristics | Clinical Relevance |
|---|---|---|---|
| Sensorimotor | Birth – 2 years | Knowledge through sensory experiences and motor actions; object permanence develops (8–12 months); cause and effect understanding | Cannot participate in verbal therapy; treatment is caregiver-focused; object permanence underlies separation anxiety |
| Preoperational | 2 – 7 years | Symbolic thinking (pretend play, language explosion); egocentrism (inability to take another's perspective); magical thinking; animism; centration | Play therapy is primary assessment/treatment modality; children may believe they caused parents' divorce or illness (magical thinking); cannot yet do cognitive restructuring |
| Concrete Operational | 7 – 11 years | Logical thinking about concrete events; conservation; classification; seriation; reversibility; decentration | Can begin cognitive components of CBT with concrete examples; can understand cause-and-effect of behavior; can use rating scales |
| Formal Operational | 12+ years | Abstract reasoning; hypothetical-deductive reasoning; metacognition; idealism; imaginary audience; personal fable | Full CBT feasible; adolescent egocentrism (imaginary audience) contributes to social anxiety; personal fable ("it won't happen to me") contributes to risk-taking |

Erikson's Psychosocial Stages
| Stage | Age | Crisis | Favorable Outcome | Psychopathology of Failure |
|---|---|---|---|---|
| 1 | 0–1 year | Trust vs Mistrust | Hope; secure attachment | Attachment disorders; anxiety |
| 2 | 1–3 years | Autonomy vs Shame/Doubt | Will; self-control | Excessive shame; toileting issues; OCD features |
| 3 | 3–6 years | Initiative vs Guilt | Purpose; imagination | Inhibition; excessive guilt; phobias |
| 4 | 6–12 years | Industry vs Inferiority | Competence; mastery | Low self-esteem; school avoidance; depression |
| 5 | 12–18 years | Identity vs Role Confusion | Fidelity; coherent identity | Identity diffusion; oppositional behavior; substance use |
03 Psychiatric Assessment in Children & Adolescents
Psychiatric evaluation of children and adolescents differs fundamentally from adult assessment. Children are brought to evaluation by others (parents, teachers, courts), may lack insight into their symptoms, and present differently at varying developmental stages. A thorough assessment requires multiple informants, attention to developmental context, and integration of behavioral observations with collateral information.
Multi-Informant Approach
Children's behavior varies across settings. A child may be anxious at school but calm at home, or aggressive at home but compliant in clinic. Best practice requires gathering information from parents/caregivers (history, home behavior, developmental milestones, family psychiatric history), teachers (academic functioning, peer relationships, classroom behavior via standardized rating scales), and the child themselves (self-report of internalizing symptoms is often more reliable than parent report for anxiety and depression in children aged 8+). When informants disagree, the clinician must weigh the context: teacher reports are often better for ADHD and externalizing behaviors, while child self-report is better for internalizing symptoms.
Clinical Interview: Age-Based Adaptations
Preschool (3–5 years): Rely primarily on parent interview and direct behavioral observation. Use play-based assessment: the child's use of toys, engagement with the examiner, symbolic play capacity, attention span, and affective expression provide diagnostic information. School-age (6–11 years): Interview parent and child separately. Use structured drawings ("draw your family"), sentence completion tasks, and direct questioning adapted to developmental level. Children this age can report on feelings but may need concrete anchors ("happy like getting a present, sad like losing your dog"). Adolescents (12–17 years): Interview the teen first alone to build rapport and facilitate disclosure of sensitive topics (substance use, sexual activity, suicidality, abuse), then interview parents. Assure limited confidentiality (explain that safety concerns require disclosure).
Structured & Semi-Structured Interviews
| Instrument | Format | Ages | Key Features |
|---|---|---|---|
| K-SADS-PL (Schedule for Affective Disorders and Schizophrenia for School-Age Children — Present and Lifetime) | Semi-structured, clinician-administered | 6–18 years | Gold standard for DSM diagnoses in research; parent and child modules; takes 1–3 hours |
| DISC (Diagnostic Interview Schedule for Children) | Fully structured, can be lay-administered | 9–17 (child), 6–17 (parent) | Computer-assisted; yes/no format; used in epidemiologic studies (MECA, GSMS) |
| MINI-KID | Brief structured, clinician-administered | 6–17 years | Short (15–30 min); screens for 24 DSM disorders; useful in clinical settings |
| PAPA (Preschool Age Psychiatric Assessment) | Semi-structured, parent interview | 2–5 years | Adapted from CAPA; covers developmental and behavioral symptoms in preschoolers |
Parent & Teacher Rating Scales
The Child Behavior Checklist (CBCL) is a 113-item parent-report form (ages 1.5–18) yielding internalizing, externalizing, and total problem scores, plus DSM-oriented subscales. The Teacher Report Form (TRF) is the parallel teacher version. The Strengths and Difficulties Questionnaire (SDQ) is a brief (25-item) screening tool covering emotional symptoms, conduct problems, hyperactivity, peer problems, and prosocial behavior; it is free, widely translated, and validated. The Conners Rating Scales (Conners-4) are multimodal (parent, teacher, self-report) scales focused on ADHD symptoms and comorbid domains. The Vanderbilt Assessment Scales are ADHD-specific, free-to-use parent and teacher forms that also screen for ODD, conduct disorder, anxiety, and depression.
Mental Status Exam in Children
The pediatric mental status exam includes: Appearance (grooming, nutritional status, physical signs of abuse/neglect, dysmorphic features). Behavior and psychomotor activity (eye contact, activity level, restlessness, tics, stereotypies, aggression, cooperation). Speech (rate, volume, articulation errors, stuttering). Mood and affect (stated mood in child's own words, observed affect range and congruence). Thought process (tangentiality, perseveration, flight of ideas — rare before adolescence). Thought content (fears, worries, obsessions, hallucinations, suicidal/homicidal ideation — always ask directly in children aged 6+). Cognition (orientation, attention span, memory for developmental level). Insight and judgment (age-appropriate: a 5-year-old cannot be expected to have adult insight).
HEADSS/HEEADSSS Psychosocial Interview for Adolescents
The HEEADSSS framework is a structured approach to psychosocial screening in adolescents, moving from less threatening to more sensitive topics:
| Letter | Domain | Key Questions |
|---|---|---|
| H | Home environment | Who lives at home? Relationship with parents/siblings? Safety? Recent moves? Running away? |
| E | Education/Employment | School performance? Favorite/difficult subjects? Future plans? Employment? |
| E | Eating | Body image? Dieting? Binge/purge? Weight changes? |
| A | Activities | Peer relationships? Hobbies? Sports? Screen time? Social media? |
| D | Drugs | Tobacco, alcohol, marijuana, vaping, prescription drugs, other substances? Friends' use? CRAFFT screening. |
| S | Sexuality | Sexual orientation? Gender identity? Sexual activity? Contraception? STI history? Abuse? |
| S | Suicide/Depression | Mood? Sleep? Hopelessness? Self-harm? Suicidal ideation, plan, access to means? |
| S | Safety | Bullying? Interpersonal violence? Guns in home? Seatbelts? Helmet use? Online safety? |
04 Key Terminology & Abbreviations
Core terms and abbreviations encountered throughout child and adolescent psychiatry. A comprehensive abbreviation reference is in Section 30.
Neurodevelopmental disorder — A condition with onset in the developmental period that produces deficits in personal, social, academic, or occupational functioning; includes ADHD, ASD, ID, learning disorders, communication disorders, and motor disorders (DSM-5 category). Externalizing disorder — Conditions characterized by outwardly directed behaviors (aggression, defiance, hyperactivity): ADHD, ODD, conduct disorder. Internalizing disorder — Conditions characterized by inwardly directed distress (anxiety, depression, somatic complaints). Comorbidity — The co-occurrence of two or more disorders; extremely common in child psychiatry (e.g., 60–80% of children with ADHD have at least one comorbid condition). Dimensional vs categorical — Symptoms exist on a continuum (dimensional); DSM diagnoses impose categorical thresholds for clinical utility. Developmental psychopathology — The study of how disorders emerge, change, and resolve across development; emphasizes equifinality (multiple paths to same outcome) and multifinality (same risk factor leads to different outcomes).
| Abbreviation | Full Term |
|---|---|
| ADHD | Attention-Deficit/Hyperactivity Disorder |
| ASD | Autism Spectrum Disorder |
| ODD | Oppositional Defiant Disorder |
| CD | Conduct Disorder |
| DMDD | Disruptive Mood Dysregulation Disorder |
| GAD | Generalized Anxiety Disorder |
| SAD | Separation Anxiety Disorder |
| OCD | Obsessive-Compulsive Disorder |
| PTSD | Post-Traumatic Stress Disorder |
| RAD | Reactive Attachment Disorder |
| DSED | Disinhibited Social Engagement Disorder |
| AN | Anorexia Nervosa |
| BN | Bulimia Nervosa |
| ARFID | Avoidant/Restrictive Food Intake Disorder |
| ID | Intellectual Disability |
| SLD | Specific Learning Disorder |
| NSSI | Non-Suicidal Self-Injury |
| CBT | Cognitive Behavioral Therapy |
| DBT-A | Dialectical Behavior Therapy for Adolescents |
| TF-CBT | Trauma-Focused Cognitive Behavioral Therapy |
05 Attention-Deficit/Hyperactivity Disorder (ADHD)
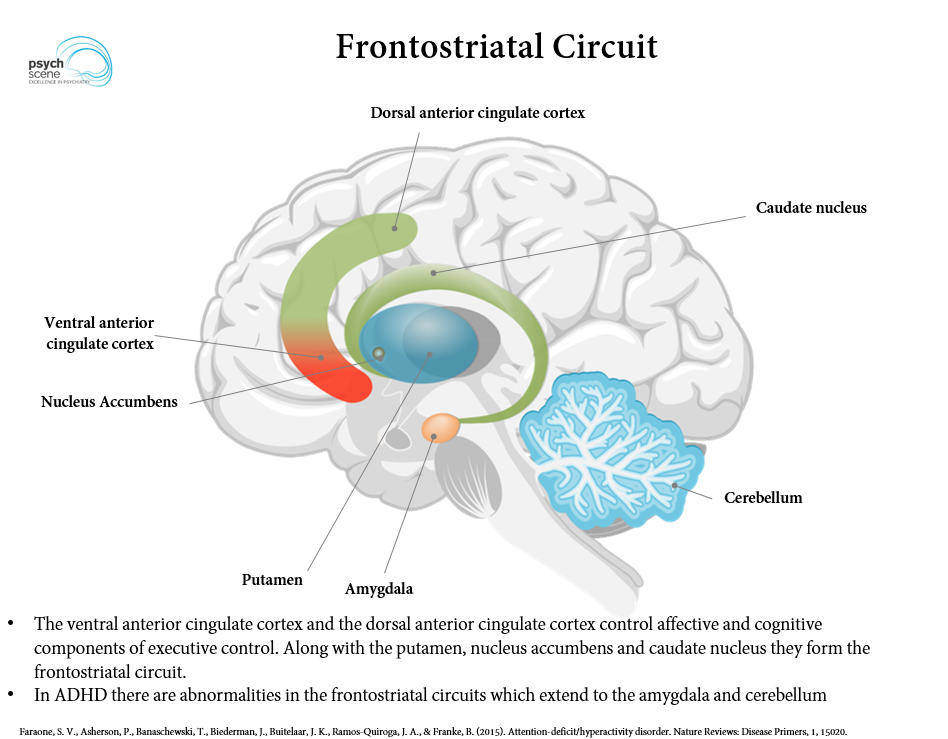
ADHD is the most common neurodevelopmental disorder, affecting 5–7% of children worldwide and 9.4% in the United States (2016 NSCH data). It is characterized by developmentally inappropriate levels of inattention, hyperactivity, and impulsivity that cause functional impairment across settings. ADHD has a strong genetic basis (heritability ~74%) with polygenic architecture involving dopaminergic, noradrenergic, and serotonergic gene variants. Neuroimaging consistently shows reduced volume and delayed maturation of prefrontal cortex, caudate, and cerebellar vermis.
DSM-5-TR Diagnostic Criteria
A. Persistent pattern of inattention and/or hyperactivity-impulsivity that interferes with functioning or development, as characterized by (1) and/or (2):
(1) Inattention: Six or more (five for age 17+) of the following for at least 6 months to a degree inconsistent with developmental level:
(a) Fails to give close attention to details; careless mistakes. (b) Difficulty sustaining attention in tasks or play. (c) Does not seem to listen when spoken to directly. (d) Does not follow through on instructions; fails to finish tasks. (e) Difficulty organizing tasks and activities. (f) Avoids tasks requiring sustained mental effort. (g) Loses things necessary for tasks. (h) Easily distracted by extraneous stimuli. (i) Forgetful in daily activities.
(2) Hyperactivity-Impulsivity: Six or more (five for age 17+) for at least 6 months:
(a) Fidgets or squirms. (b) Leaves seat when remaining seated is expected. (c) Runs/climbs in inappropriate situations. (d) Unable to play quietly. (e) "On the go" or "driven by a motor." (f) Talks excessively. (g) Blurts out answers before questions are completed. (h) Difficulty waiting turn. (i) Interrupts or intrudes on others.
B. Several symptoms present before age 12. C. Symptoms present in 2+ settings. D. Clear evidence of functional impairment. E. Not better explained by another mental disorder.
Presentations: Combined (meets both criteria), Predominantly Inattentive, Predominantly Hyperactive-Impulsive. Severity: Mild, Moderate, Severe. Specify if in partial remission.
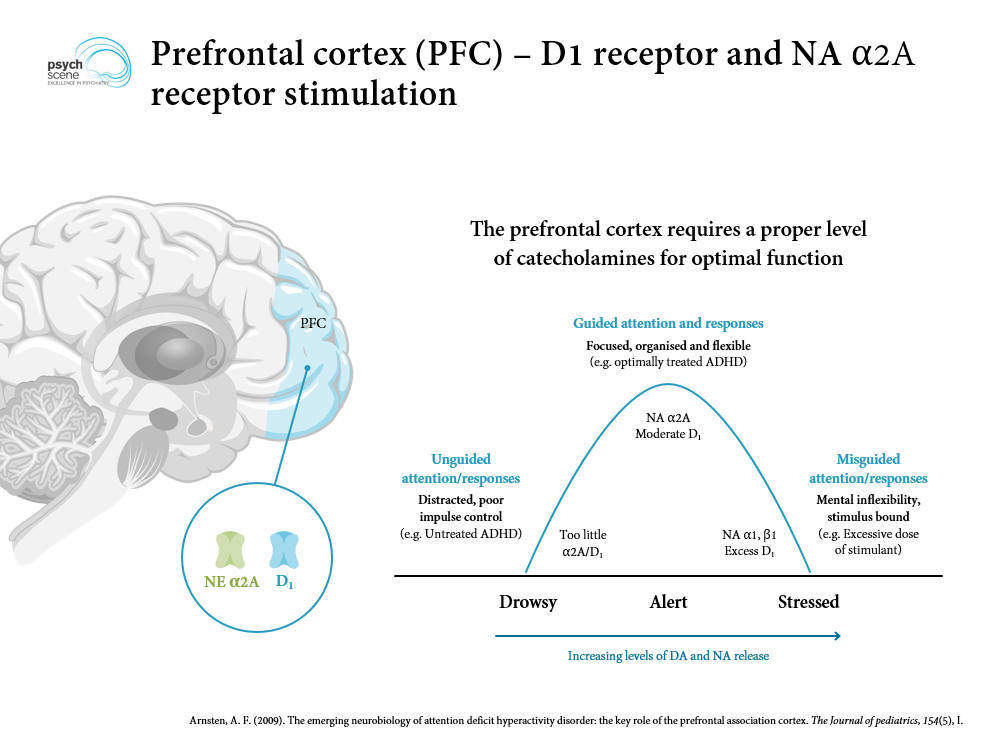
Rating Scales for ADHD
The Vanderbilt ADHD Diagnostic Rating Scale (parent and teacher forms) maps directly to DSM criteria and screens for comorbidities. Scoring: each item 0–3 (never to very often); 6+ items rated 2–3 in the inattention or hyperactivity-impulsivity domain constitutes a positive screen. Performance items (academic/behavioral functioning) rated 4–5 ("somewhat/very much of a problem") indicate impairment. The Conners Rating Scales (Conners-4) provide T-scores (mean 50, SD 10); T-scores above 65 are elevated and above 70 are very elevated. Include validity scales to detect inconsistent or overly positive/negative reporting.
ADHD Pharmacotherapy
Stimulants are first-line for ADHD aged 6+ (AAP, AACAP guidelines). For ages 4–5, parent training in behavior management is first-line; methylphenidate may be added if behavioral interventions are insufficient. Stimulants block dopamine and norepinephrine transporters in the PFC and striatum, increasing synaptic availability of these neurotransmitters. Response rates are 70–80% for any single stimulant and up to 90% when both methylphenidate and amphetamine classes are tried.
| Medication | Formulation | Starting Dose | Max Dose | Duration |
|---|---|---|---|---|
| Methylphenidate IR (Ritalin) | Tablet: 5, 10, 20 mg | 5 mg BID-TID | 60 mg/day | 3–4 hours |
| Methylphenidate ER (Concerta) | OROS tablet: 18, 27, 36, 54 mg | 18 mg QAM | 72 mg/day (54 mg for children) | 10–12 hours |
| Methylphenidate LA (Ritalin LA) | Capsule: 10, 20, 30, 40 mg | 10–20 mg QAM | 60 mg/day | 8–10 hours |
| Dexmethylphenidate ER (Focalin XR) | Capsule: 5, 10, 15, 20, 25, 30, 35, 40 mg | 5 mg QAM | 30 mg/day (children); 40 mg (adults) | 8–12 hours |
| Mixed amphetamine salts IR (Adderall) | Tablet: 5, 7.5, 10, 12.5, 15, 20, 30 mg | 5 mg QD-BID | 40 mg/day | 4–6 hours |
| Mixed amphetamine salts XR (Adderall XR) | Capsule: 5, 10, 15, 20, 25, 30 mg | 5–10 mg QAM | 30 mg/day (children); 40 mg (adults) | 10–12 hours |
| Lisdexamfetamine (Vyvanse) | Capsule/chewable: 10, 20, 30, 40, 50, 60, 70 mg | 20–30 mg QAM | 70 mg/day | 10–14 hours |
Non-Stimulant ADHD Medications
| Medication | Mechanism | Starting Dose | Target/Max Dose | Key Pearls |
|---|---|---|---|---|
| Atomoxetine (Strattera) | Selective NE reuptake inhibitor | 0.5 mg/kg/day × 3 days | 1.2 mg/kg/day (max 100 mg) | Not a controlled substance; takes 4–6 weeks for full effect; FDA black box for suicidal ideation; hepatotoxicity rare but serious |
| Guanfacine ER (Intuniv) | Alpha-2A adrenergic agonist | 1 mg QHS | 4 mg/day (ages 6–12); 7 mg (13–17) | Also helps tic disorders, aggression, insomnia; monitor for bradycardia, hypotension, sedation; taper to discontinue |
| Clonidine ER (Kapvay) | Alpha-2 adrenergic agonist (less selective) | 0.1 mg QHS | 0.4 mg/day (divided BID) | More sedating than guanfacine; useful for sleep initiation; rebound hypertension if stopped abruptly |
| Viloxazine ER (Qelbree) | NE reuptake inhibitor; 5-HT modulator | 100 mg QAM (6–11 yr); 200 mg QAM (12–17 yr) | 200 mg (6–11); 400 mg (12–17) | FDA-approved 2021; non-scheduled; may worsen suicidal ideation (black box); interacts with CYP1A2 substrates |
Before starting stimulants: Height, weight, BMI, BP, HR, cardiac history screening (personal and family history of sudden death, arrhythmia, cardiomyopathy). EKG is NOT routinely required (AAP/AHA). Ongoing monitoring: Height/weight every 3 months (plot on growth curve); BP/HR at each visit; assess for appetite suppression, sleep disturbance, mood changes, tics. Drug holidays: Consider during summer to assess ongoing need and allow growth catch-up; not routinely recommended as standard of care.
06 Autism Spectrum Disorder (ASD)
ASD is a neurodevelopmental disorder characterized by persistent deficits in social communication and social interaction across contexts, combined with restricted, repetitive patterns of behavior. DSM-5 collapsed the previous DSM-IV categories (autistic disorder, Asperger's disorder, PDD-NOS, childhood disintegrative disorder) into a single spectrum with severity levels. Prevalence is approximately 1 in 36 children (CDC, 2023 data from 2020 surveillance year), with a 4:1 male-to-female ratio (though females may be underdiagnosed due to camouflaging/masking).
DSM-5-TR Diagnostic Criteria
A. Persistent deficits in social communication and social interaction across multiple contexts, as manifested by ALL of the following (currently or by history):
(1) Deficits in social-emotional reciprocity (e.g., abnormal social approach, failure of back-and-forth conversation, reduced sharing of interests/emotions/affect, failure to initiate or respond to social interactions). (2) Deficits in nonverbal communicative behaviors (e.g., poorly integrated verbal and nonverbal communication, abnormal eye contact and body language, deficits in understanding/use of gestures, total lack of facial expressions). (3) Deficits in developing, maintaining, and understanding relationships (e.g., difficulty adjusting behavior to social contexts, difficulty sharing imaginative play or making friends, absence of interest in peers).
B. Restricted, repetitive patterns of behavior, interests, or activities, as manifested by at least 2 of the following:
(1) Stereotyped or repetitive motor movements, use of objects, or speech (e.g., hand flapping, lining up toys, echolalia, idiosyncratic phrases). (2) Insistence on sameness, inflexible adherence to routines, ritualized patterns (e.g., extreme distress at small changes, rigid thinking, greeting rituals, need to take same route). (3) Highly restricted, fixated interests abnormal in intensity or focus (e.g., strong attachment to unusual objects, excessively circumscribed interests). (4) Hyper- or hyporeactivity to sensory input or unusual interest in sensory aspects of environment (e.g., apparent indifference to pain/temperature, adverse response to specific sounds/textures, excessive smelling/touching, visual fascination with lights/movement).
C. Symptoms present in the early developmental period (may not fully manifest until social demands exceed capacity). D. Symptoms cause clinically significant impairment. E. Not better explained by ID or global developmental delay (though ASD and ID frequently co-occur).
ASD Severity Levels
| Level | Social Communication | Restricted/Repetitive Behaviors |
|---|---|---|
| Level 1 — "Requiring support" | Without supports, deficits cause noticeable impairments; difficulty initiating interactions; may appear to have decreased interest in social interactions; atypical/unsuccessful responses | Inflexibility causes significant interference in one or more contexts; difficulty switching between activities; problems with organization and planning hamper independence |
| Level 2 — "Requiring substantial support" | Marked deficits in verbal and nonverbal social communication; social impairments apparent even with supports; limited initiation; reduced or abnormal responses to social overtures | Inflexibility, difficulty coping with change, RRBs frequent enough to be obvious to casual observer; distress/difficulty changing focus or action |
| Level 3 — "Requiring very substantial support" | Severe deficits in verbal and nonverbal social communication causing severe impairments; very limited initiation; minimal response to social overtures | Inflexibility, extreme difficulty coping with change; RRBs markedly interfere with functioning in all spheres; great distress/difficulty changing focus or action |
Diagnostic Instruments
The Autism Diagnostic Observation Schedule, 2nd Edition (ADOS-2) is a semi-structured, standardized assessment of communication, social interaction, play, and restricted/repetitive behaviors. It uses five modules based on expressive language level: Toddler Module (12–30 months), Module 1 (31+ months, no phrase speech), Module 2 (phrase speech), Module 3 (fluent speech, child/adolescent), Module 4 (fluent speech, adolescent/adult). Scores are classified as "autism," "autism spectrum," or "non-spectrum." The Autism Diagnostic Interview-Revised (ADI-R) is a comprehensive parent interview (2–3 hours) covering early development and current functioning. Together, the ADOS-2 and ADI-R are considered the gold standard diagnostic battery for ASD research.
Early Signs of ASD
Signs observable by 12–18 months: reduced or absent joint attention (not following a point, not showing objects to share interest), reduced social referencing (not looking to caregiver for emotional cues), lack of protodeclarative pointing (pointing to share interest rather than to request), poor response to name, reduced social smile, lack of imitation, absence of pretend play by 18 months, and language delay or regression.
Comorbidities
Approximately 70% of individuals with ASD have at least one comorbid psychiatric condition, and 40% have two or more. Common comorbidities: intellectual disability (~33%), ADHD (30–50%; DSM-5 now allows dual diagnosis), anxiety disorders (40–50%), depression (12–70% depending on age/IQ), epilepsy (20–30%, bimodal onset in early childhood and adolescence), sleep disorders (50–80%), GI problems (constipation, functional abdominal pain in 30–70%), and feeding difficulties.
Treatment Approach
Applied Behavior Analysis (ABA) is the most extensively researched behavioral intervention, using principles of operant conditioning to teach skills and reduce challenging behaviors. Early intensive behavioral intervention (EIBI, 25–40 hours/week for 2+ years starting before age 4) produces significant gains in IQ, adaptive behavior, and language in some children. Speech-language therapy targets pragmatic language, social communication, and augmentative/alternative communication (AAC) for minimally verbal children. Occupational therapy addresses sensory processing differences, fine motor skills, and self-care. No medication treats core ASD symptoms. Risperidone (FDA-approved age 5–16) and aripiprazole (FDA-approved age 6–17) are approved for irritability associated with ASD (aggression, self-injury, severe tantrums).

Genetic & Neurobiological Findings
ASD has a heritability of approximately 80%. Twin studies show concordance of 60–90% in monozygotic twins vs 0–30% in dizygotic twins. Identified genetic causes account for 20–25% of cases: recurrent CNVs (16p11.2 deletion/duplication, 15q11-13 duplication, 22q11.2 deletion), single-gene disorders (fragile X in 2–5%, tuberous sclerosis in 1–4%, Rett syndrome, PTEN mutations), and de novo point mutations identified by whole exome sequencing. Neuroanatomical findings include early brain overgrowth (larger head circumference and brain volume in the first 2 years, normalizing by adolescence), abnormalities in minicolumnar organization, reduced long-range connectivity with increased local connectivity ("underconnectivity theory"), and reduced Purkinje cell counts in the cerebellum.
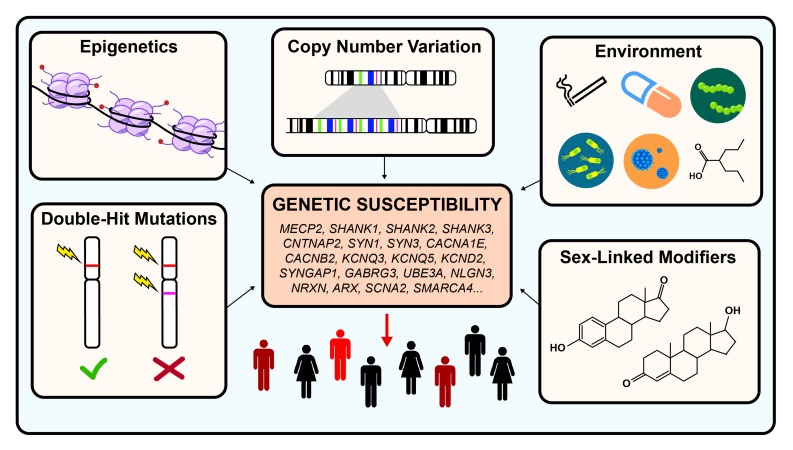
Pharmacotherapy for ASD-Associated Symptoms
| Target Symptom | First-Line Agent | Dose Range | Notes |
|---|---|---|---|
| Irritability, aggression, self-injury | Risperidone (FDA-approved 5–16); aripiprazole (FDA-approved 6–17) | Risp: 0.25–3 mg; Arip: 2–15 mg | Only FDA-approved agents for ASD irritability; monitor metabolic effects |
| Hyperactivity, inattention (comorbid ADHD) | Methylphenidate (lower doses) | Start 2.5–5 mg BID | RUPP study: methylphenidate effective but with higher side effect rate (irritability, appetite loss) in ASD vs ADHD alone |
| Anxiety | SSRIs (low dose) | Fluoxetine 5–20 mg | Limited RCT evidence in ASD; start low ("start low, go slow, but go"); monitor for activation |
| Repetitive behaviors | SSRIs (off-label) | Variable by agent | Adult RCT evidence mixed; pediatric trials largely negative for core RRBs; may help rigidity/anxiety driving RRBs |
| Sleep disturbance | Melatonin | 1–10 mg at bedtime | Strong evidence in ASD (multiple RCTs); improves sleep onset latency by 30–40 min on average |
07 Intellectual Disability & Global Developmental Delay
Intellectual disability (ID) is characterized by deficits in both intellectual functioning (IQ approximately 2 SDs below the mean, ~70 or below) and adaptive functioning (conceptual, social, and practical domains) with onset during the developmental period. Global developmental delay (GDD) is the diagnosis used for children under 5 years who fail to meet developmental milestones in 2+ domains but cannot yet be reliably assessed with standardized IQ testing. GDD has a prevalence of 1–3%; ID affects approximately 1% of the population.
DSM-5 Severity Levels
| Severity | IQ Range (Approx.) | % of ID | Conceptual Domain | Social Domain | Practical Domain |
|---|---|---|---|---|---|
| Mild | 50–70 | 85% | Learning difficulties in academic skills; in adults, abstract thinking, executive function, and short-term memory impaired | Immature social interactions; difficulty reading social cues; communication more concrete | May function age-appropriately in personal care; needs support with complex daily tasks, healthcare decisions, finances |
| Moderate | 35–49 | 10% | Academic skills develop slowly; in adults, elementary level; needs ongoing assistance for work and personal affairs | Spoken language primary social tool but much less complex; relationships limited; needs significant social support | Can attend to personal care with extended teaching; supervised employment; needs support for all household tasks |
| Severe | 20–34 | 3–4% | Limited attainment of conceptual skills; little understanding of written language or number concepts | Spoken language limited in vocabulary and grammar; communication focused on here and now | Requires support for all ADLs; supervision at all times; cannot make responsible decisions about well-being |
| Profound | <20 | 1–2% | Conceptual skills involve physical world rather than symbolic; may use objects for self-care, work, recreation | Very limited symbolic communication; may understand simple instructions/gestures; expresses desires through nonverbal communication | Dependent on others for all aspects of physical care, health, safety; may participate in some activities |
Adaptive Functioning Assessment
DSM-5 bases ID severity on adaptive functioning rather than IQ alone. The Vineland Adaptive Behavior Scales, 3rd Edition (Vineland-3) is the most widely used measure, assessing communication, daily living skills, socialization, and motor skills through a semi-structured caregiver interview or parent/teacher rating form. It yields standard scores (mean 100, SD 15) and age-equivalent scores. Scores below 70 (2+ SDs below mean) indicate significant adaptive limitations. The Adaptive Behavior Assessment System, 3rd Edition (ABAS-3) is an alternative parent/teacher rating form covering conceptual, social, and practical adaptive skill domains.
Etiologic Evaluation
Identifiable etiology is found in 40–60% of cases with comprehensive workup. The first-tier evaluation includes: chromosomal microarray (CMA) — detects copy number variants (CNVs) in 15–20% of unexplained ID; fragile X testing (trinucleotide repeat expansion in FMR1 gene) — the most common inherited cause of ID; metabolic screening (amino acids, organic acids, acylcarnitine profile, thyroid function). Second-tier evaluation if first-tier negative: whole exome sequencing (WES) identifies pathogenic variants in an additional 25–40% of cases; brain MRI (structural abnormalities in 30% of moderate-severe ID); EEG if seizures suspected. Common genetic syndromes: Down syndrome (trisomy 21, IQ 30–70), fragile X syndrome (FMRP deficiency, IQ 40–70 in males), Williams syndrome (7q11.23 deletion, mean IQ 55–70, hypersociality), Prader-Willi syndrome (15q11.2 paternal deletion, hyperphagia, IQ 60–70), Angelman syndrome (15q11.2 maternal deletion, severe ID, happy demeanor, seizures).
08 Specific Learning Disorders & Communication Disorders
Specific Learning Disorder (SLD) involves persistent difficulties in learning and using academic skills, with onset during school years, that are not explained by ID, sensory impairment, or inadequate instruction. Prevalence is 5–15% of school-age children. SLD is specified with impairment in reading (dyslexia), written expression (dysgraphia), or mathematics (dyscalculia). Severity is rated mild, moderate, or severe based on functional impact and support needs.
SLD Subtypes
| Subtype | Core Deficits | Prevalence | Key Features |
|---|---|---|---|
| Dyslexia (impairment in reading) | Phonological awareness, decoding, word recognition, reading fluency, comprehension | 5–10% | Most common SLD; familial; deficits in left temporoparietal region; responds to structured literacy (Orton-Gillingham approach); does NOT relate to IQ |
| Dysgraphia (impairment in written expression) | Spelling, grammar, punctuation, clarity/organization of writing, handwriting | 7–15% | Often co-occurs with dyslexia and ADHD; fine motor difficulties overlap with DCD |
| Dyscalculia (impairment in mathematics) | Number sense, math fact memorization, math reasoning, calculation fluency | 3–7% | Associated with deficits in intraparietal sulcus; comorbid with ADHD in 20–40% |
Communication Disorders
Language Disorder (previously expressive and mixed receptive-expressive language disorders) involves persistent difficulties in acquiring and using language across modalities (spoken, written, sign language) due to deficits in comprehension or production of vocabulary, sentence structure, and discourse. Affects 7–8% of kindergartners. Speech Sound Disorder involves persistent difficulty with speech sound production (articulation, phonological processes) interfering with intelligibility; affects 2–3% at age 6–7. Childhood-Onset Fluency Disorder (Stuttering) involves disruptions in speech fluency and time patterning (sound repetitions, prolongations, blocks, circumlocutions); onset typically 2–7 years; 80% resolve spontaneously by adolescence; 1% persist into adulthood. Social (Pragmatic) Communication Disorder involves persistent difficulties in the social use of verbal and nonverbal communication (understanding implied meaning, adjusting communication to context, conversational turn-taking, narrative coherence) without restricted/repetitive behaviors (which would indicate ASD instead).
Social Communication Disorder (SCD) shares Criterion A of ASD (social communication deficits) but LACKS Criterion B (restricted, repetitive behaviors/interests). If any RRBs are present, ASD should be diagnosed instead. SCD was introduced in DSM-5 partly to capture individuals previously diagnosed with Asperger's or PDD-NOS who did not meet full ASD criteria. Both conditions benefit from pragmatic language therapy and social skills groups.
09 Tic Disorders & Tourette Syndrome
Tic disorders exist on a spectrum defined by tic type (motor and/or vocal) and duration. Tourette syndrome (TS) is the most severe, requiring both motor and vocal tics for more than one year. Prevalence of TS is 0.3–1% of school-age children, with a male-to-female ratio of 3–4:1. Tics typically begin at age 5–7, peak in severity at 10–12, and improve in 60–80% of cases by late adolescence.
Tic Classification
| Type | Simple Tics | Complex Tics |
|---|---|---|
| Motor | Eye blinking, facial grimacing, head jerking, shoulder shrugging, nose twitching | Jumping, touching, squatting, twirling, echopraxia (imitating movements), copropraxia (obscene gestures) |
| Vocal (Phonic) | Throat clearing, sniffing, grunting, barking, coughing | Echolalia (repeating others' words), palilalia (repeating own words), coprolalia (obscene words — occurs in only 10–15% of TS) |
DSM-5 Tic Disorder Diagnoses
Tourette syndrome: Multiple motor tics AND at least one vocal tic present at some time during illness (not necessarily concurrently); tics present >1 year; onset before age 18. Persistent (Chronic) Motor or Vocal Tic Disorder: Single or multiple motor OR vocal tics (not both); >1 year duration; onset before age 18. Provisional Tic Disorder: Single or multiple motor and/or vocal tics; duration <1 year; onset before age 18.
Waxing and Waning Course
Tics characteristically wax and wane in frequency, intensity, type, and location over weeks to months. They are often preceded by a premonitory urge (a subjective uncomfortable sensation relieved by performing the tic), are temporarily suppressible (with effort), and worsen with stress, fatigue, excitement, and illness. Tics often improve during focused activity and sleep.
Assessment
The Yale Global Tic Severity Scale (YGTSS) is the most widely used clinician-rated measure. It rates number, frequency, intensity, complexity, and interference of motor and vocal tics separately on 0–5 scales, yielding a Total Tic Score (0–50) and an Impairment Score (0–50), for a Global Severity Score (0–100). Scores above 20 on the Total Tic Score suggest moderate severity.
Differential Diagnosis of Tics
| Condition | Distinguishing Features |
|---|---|
| Stereotypies (ASD, ID) | More rhythmic, fixed pattern (hand flapping, body rocking), usually bilateral, begin earlier (<3 years), no premonitory urge, less suppressible |
| Myoclonus | Involuntary, shock-like; no premonitory urge; not suppressible; may have EEG correlate (cortical myoclonus) |
| Chorea (Sydenham's) | Random, flowing movements; associated with rheumatic fever and elevated ASO titers; hypotonic "milkmaid's grip" |
| Compulsions (OCD) | Driven by anxiety/obsession; purposeful; aimed at reducing distress; ego-dystonic |
| Functional (psychogenic) movement disorder | Inconsistent, distractible, entrainable, incongruent with organic patterns; may be triggered by stress |
Common Comorbidities
Only 10–15% of individuals with TS have tics alone. Comorbidities include: ADHD (50–60%, often the greater source of impairment), OCD (30–50%, often with "just right" sensations and symmetry obsessions), anxiety disorders (30%), mood disorders (20%), learning disabilities (25%), rage attacks/emotional dysregulation (25–70%), and sleep disturbances.
Treatment
Comprehensive Behavioral Intervention for Tics (CBIT) is first-line for tics causing functional impairment. CBIT incorporates habit reversal training (awareness training + competing response training), function-based interventions, and relaxation techniques. The CBIT trial showed 52.5% response rate vs 18.5% for supportive therapy. Pharmacotherapy is reserved for tics that are moderate-severe and functionally impairing despite CBIT:
| Medication | Mechanism | Dose Range | Evidence/Pearls |
|---|---|---|---|
| Guanfacine ER | Alpha-2A agonist | 1–4 mg/day | Mild-moderate tic reduction; good for comorbid ADHD; sedation, hypotension |
| Clonidine | Alpha-2 agonist | 0.1–0.4 mg/day | Similar to guanfacine; more sedating; useful for insomnia |
| Fluphenazine | D2 antagonist (typical) | 0.5–5 mg/day | Effective for tics; EPS risk; weight gain |
| Aripiprazole | D2 partial agonist | 2.5–15 mg/day | Strong evidence for tics; less metabolic effects; akathisia risk |
| Pimozide | D2 antagonist (typical) | 1–6 mg/day | FDA-approved for TS; requires EKG for QTc monitoring; drug interactions via CYP3A4 |
| Haloperidol | D2 antagonist (typical) | 0.25–5 mg/day | FDA-approved for TS; effective but high EPS burden limits use |
10 Pediatric Depression
Major depressive disorder (MDD) affects approximately 2% of prepubertal children and 4–8% of adolescents, with a lifetime prevalence by age 18 of 11–14%. Before puberty, the sex ratio is approximately equal; after puberty, females are affected 2–3 times more often than males. Pediatric depression differs from adult depression in several important ways: irritability may be the predominant mood (rather than sadness), somatic complaints (headaches, stomachaches) are common, and behavioral changes (declining grades, social withdrawal, oppositional behavior) may be more prominent than verbalizing depressed mood.
DSM-5 Criteria Adaptation for Youth
The same DSM-5 MDD criteria apply to children and adults with two modifications: (1) irritable mood can substitute for depressed mood in children and adolescents, and (2) failure to make expected weight gains can substitute for weight loss. Duration requirement remains at least 2 weeks. Five of nine symptoms must be present (depressed/irritable mood, anhedonia, weight/appetite change, insomnia/hypersomnia, psychomotor agitation/retardation, fatigue, worthlessness/guilt, concentration difficulty, suicidal ideation). At least one must be depressed/irritable mood or anhedonia.
Screening & Assessment
The Children's Depression Rating Scale — Revised (CDRS-R) is a clinician-rated 17-item scale for ages 6–12 (often used up to 18); total score range 17–113, with scores above 40 suggesting clinically significant depression. The Patient Health Questionnaire for Adolescents (PHQ-A) is a self-report adaptation of the PHQ-9 for teens, recommended for universal screening at annual well visits for ages 12–21 (USPSTF recommendation). Scores: 0–4 minimal, 5–9 mild, 10–14 moderate, 15–19 moderately severe, 20–27 severe.
The TADS Study
The Treatment for Adolescents with Depression Study (TADS) was a landmark NIMH-funded RCT (n=439, ages 12–17) comparing fluoxetine, CBT, combined treatment, and placebo. At 12 weeks: combined treatment (71% response) was superior to fluoxetine alone (61%), CBT alone (43%), and placebo (35%). Fluoxetine was superior to placebo; CBT alone was not statistically superior to placebo at 12 weeks. By 36 weeks, all active treatments converged. Combined treatment also had the best safety profile — CBT appeared protective against suicidal ideation associated with fluoxetine alone.
Pharmacotherapy
Fluoxetine is the only SSRI with FDA approval for MDD in children aged 8+. Escitalopram is FDA-approved for MDD in adolescents aged 12+. Starting dose of fluoxetine is 10 mg/day, increasing to 20 mg after 1–2 weeks; maximum 60 mg/day. Escitalopram starts at 5–10 mg/day, target 10–20 mg. All antidepressants carry an FDA black box warning for increased suicidal thinking and behavior in children, adolescents, and young adults (ages <25) based on pooled analysis of 24 trials (4% risk vs 2% for placebo); no completed suicides occurred in these trials. The risk of untreated depression (including suicidality) generally outweighs antidepressant risk when properly monitored.
When starting or dose-adjusting an antidepressant in patients under 25: see patient weekly for the first 4 weeks, then biweekly for weeks 5–8, then at week 12, then as clinically indicated. Assess for worsening depression, agitation, irritability, suicidal ideation, and unusual behavior changes at every visit. Educate families to call immediately if concerning symptoms emerge between visits.
Other Considerations in Pediatric Depression
Persistent Depressive Disorder (Dysthymia) in youth requires depressed or irritable mood for most of the day, more days than not, for at least 1 year (vs 2 years in adults), plus 2+ of: appetite change, sleep disturbance, low energy, low self-esteem, poor concentration, hopelessness. Chronic low-grade depression is often missed in children because it becomes the child's baseline and parents may not recognize it as pathological. Double depression (MDD superimposed on dysthymia) carries a worse prognosis and higher relapse rate.
Psychotic features in pediatric depression are rare but serious and include mood-congruent hallucinations (voices saying the child is worthless, deserves to die) and delusions of guilt or worthlessness. Psychotic depression requires antidepressant + antipsychotic combination; ECT may be considered for treatment-refractory cases (rarely used in children but can be lifesaving). Seasonal affective disorder can present in adolescents, with winter onset of depressive symptoms; light therapy (10,000 lux for 30 min in the morning) is an evidence-based treatment.
The AAP Bright Futures guidelines and USPSTF (2016, reaffirmed 2022) recommend universal screening for MDD in adolescents aged 12–18 at well-child visits. The PHQ-A or PHQ-2 (2-item screen: depressed mood and anhedonia, with referral for full PHQ-A if either endorsed) are recommended instruments. Positive screens should prompt diagnostic evaluation, not automatic treatment. Screening without adequate follow-up systems in place is insufficient and may cause harm.
11 Pediatric Bipolar Disorder & DMDD
Pediatric bipolar disorder is among the most controversial diagnoses in child psychiatry. The core debate centers on whether chronic, non-episodic irritability in children represents bipolar disorder or a different condition. DSM-5 addressed this by introducing Disruptive Mood Dysregulation Disorder (DMDD) as an alternative diagnostic home for severely irritable children who were previously labeled with bipolar disorder.
Bipolar Disorder in Youth
The same DSM-5 criteria for bipolar I and II apply to children as adults, with no age-specific modifications. Episodicity is the key diagnostic requirement: manic episodes must have a clear onset and offset, representing a change from the child's usual state. Pediatric mania presents with elated or expansive mood (giddy, silly, grandiose) or irritable mood plus increased energy, lasting at least 7 days (or any duration if hospitalization required). Associated symptoms include decreased need for sleep (not just insomnia), grandiosity (believing they have superpowers, are above rules), pressured speech, flight of ideas, distractibility, increased goal-directed activity, and risk-taking. Mean age of onset is 15–19 years; prepubertal bipolar I is rare and should be diagnosed with extreme caution.
DMDD — DSM-5-TR Criteria
A. Severe recurrent temper outbursts (verbal rages and/or physical aggression toward people or property) that are grossly out of proportion in intensity or duration to the situation or provocation. B. Temper outbursts are inconsistent with developmental level. C. Temper outbursts occur, on average, 3+ times per week. D. Mood between outbursts is persistently irritable or angry most of the day, nearly every day, observable by others (parents, teachers, peers). E. Criteria A–D present for 12+ months without a symptom-free period of 3+ consecutive months. F. Criteria A and D present in at least 2 of 3 settings (home, school, peers) and severe in at least 1. G. Diagnosis should not be made for the first time before age 6 or after age 18. H. Age of onset of Criteria A–E before age 10. I. No distinct manic/hypomanic episode lasting more than 1 day. J. Behaviors do not occur exclusively during an MDD episode. K. Not better explained by another mental disorder or substance.
DMDD CANNOT be diagnosed with ODD, IED, or bipolar disorder. CAN be comorbid with MDD, ADHD, CD, and substance use disorders.
Pharmacotherapy for Pediatric Bipolar Disorder
| Medication | FDA Indication (Pediatric) | Dose Range | Key Considerations |
|---|---|---|---|
| Lithium | Bipolar I mania/maintenance, age 7+ | Start 300 mg BID-TID; target level 0.8–1.2 mEq/L (acute), 0.6–1.0 (maintenance) | Requires serum monitoring (levels, renal, thyroid); narrow therapeutic index; lithium toxicity: tremor, GI, confusion, seizures |
| Aripiprazole | Bipolar I mania, age 10–17 | 2–30 mg/day | Less metabolic burden; akathisia; EPS |
| Quetiapine | Bipolar I mania, age 10–17 | 400–600 mg/day | Sedation; significant metabolic effects; weight gain |
| Risperidone | Bipolar I mania, age 10–17 | 0.5–6 mg/day | Prolactin elevation; weight gain; EPS |
| Olanzapine | Bipolar I mania, age 13–17 | 2.5–20 mg/day | Highest metabolic risk; substantial weight gain; reserved for treatment-resistant cases |
| Valproate (divalproex) | Not FDA-approved for pediatric bipolar (used off-label) | 15–60 mg/kg/day; target level 50–125 mcg/mL | Teratogenic (contraindicated in females of reproductive potential without contraception); hepatotoxic risk <2 yr; pancreatitis; PCOS |
12 Anxiety Disorders in Children
Anxiety disorders are the most common psychiatric conditions in childhood, affecting 15–20% of children and adolescents. Unlike typical developmental fears (stranger anxiety at 8–12 months, fear of the dark at 3–5 years), pathological anxiety is excessive, persistent, and functionally impairing. Without treatment, childhood anxiety disorders are chronic and predict adult anxiety, depression, and substance use.
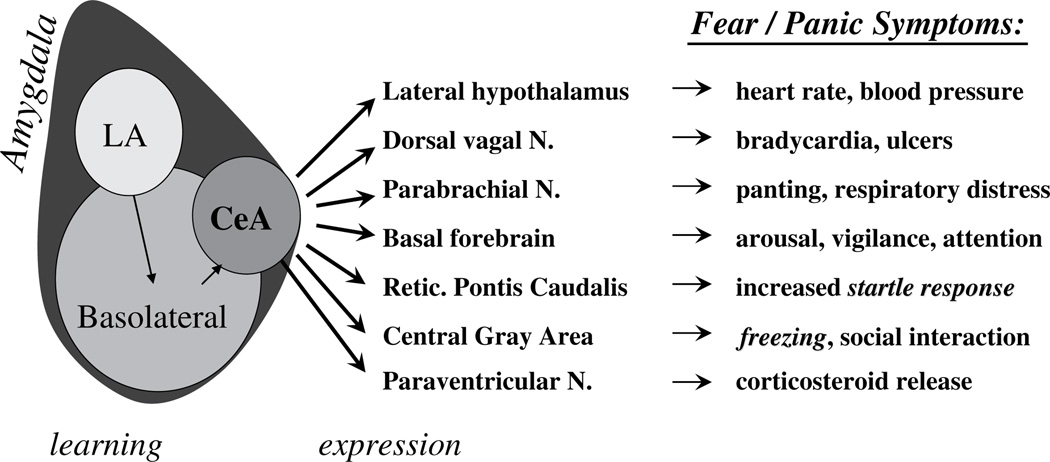
Separation Anxiety Disorder (SAD)
Developmentally inappropriate and excessive fear or anxiety concerning separation from attachment figures, with onset before age 18 (usually 7–9 years). Requires at least 3 of 8 symptoms for at least 4 weeks (children) or 6 months (adults): excessive distress when separation occurs or is anticipated, worry about losing attachment figures, worry about events causing separation (kidnapping, getting lost), reluctance/refusal to go out due to fear of separation, fear of being alone, reluctance to sleep away from home, nightmares about separation, physical complaints when separation occurs or is anticipated. SAD is the most common anxiety disorder in children under 12.
Selective Mutism
Consistent failure to speak in specific social situations where speech is expected (e.g., school) despite speaking in other situations (e.g., home). Duration at least 1 month (not limited to the first month of school). Most children with selective mutism have an underlying social anxiety disorder. Treatment: graduated exposure-based interventions, with the child gradually increasing verbal communication in feared settings; SSRIs (fluoxetine) for moderate-severe cases.
Social Anxiety Disorder (Social Phobia)
Marked fear or anxiety about one or more social situations involving possible scrutiny by others. In children, the anxiety must occur in peer settings (not just adult interactions) and may manifest as crying, tantrums, freezing, clinging, or shrinking from social situations. Duration at least 6 months. Cognitive distortions center on negative evaluation by others. CBT with social skills training is first-line; SSRIs for moderate-severe or treatment-resistant cases.
Generalized Anxiety Disorder (GAD)
Excessive anxiety and worry about a variety of topics (school performance, health, world events, family finances), occurring more days than not for at least 6 months. In children, only 1 of 6 associated symptoms is required (vs 3 for adults): restlessness, fatigue, difficulty concentrating, irritability, muscle tension, sleep disturbance. Children with GAD are often "little worriers" who seek excessive reassurance from parents and teachers. CBT targets cognitive restructuring and worry management skills.
Specific Phobias
Marked fear or anxiety about a specific object or situation (animals, natural environment, blood-injection-injury, situational, other). In children, the fear may be expressed by crying, tantrums, freezing, or clinging. Duration at least 6 months. Common childhood phobias: dogs, insects, darkness, thunderstorms, needles, vomiting (emetophobia). Treatment is exposure-based therapy (systematic desensitization, graduated exposure). Medication is generally not indicated.
SCARED Screening
The Screen for Child Anxiety Related Disorders (SCARED) is a 41-item child and parent-report questionnaire for ages 8–18. Total score of 25+ indicates an anxiety disorder (sensitivity 71%, specificity 67%). Subscales: panic/somatic (score ≥7), GAD (≥9), separation anxiety (≥5), social anxiety (≥8), school avoidance (≥3). Free to use, available in multiple languages.
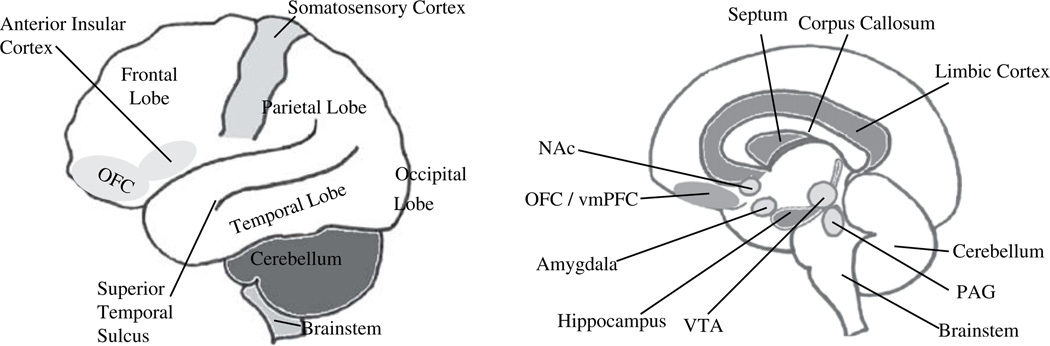
Panic Disorder in Adolescents
Panic disorder is uncommon before puberty but increases in prevalence during adolescence (1–3% by age 18). Adolescents experience recurrent unexpected panic attacks (discrete episodes of intense fear peaking within minutes) with at least 4 of 13 symptoms: palpitations, sweating, trembling, shortness of breath, choking sensation, chest pain, nausea, dizziness, derealization/depersonalization, fear of losing control, fear of dying, paresthesias, chills/hot flushes. At least 1 month of worry about additional attacks or maladaptive behavioral change (agoraphobic avoidance) is required. Adolescents frequently present to EDs with somatic complaints mimicking cardiac or respiratory emergencies. Treatment: CBT with interoceptive exposure (deliberately provoking physical sensations of panic to reduce catastrophic misinterpretation) plus SSRIs for moderate-severe cases.
The CAMS Study
The Child/Adolescent Anxiety Multimodal Study (CAMS) (n=488, ages 7–17) compared sertraline, CBT, combined treatment (sertraline + CBT), and placebo for childhood anxiety disorders (SAD, GAD, social anxiety). At 12 weeks: combined treatment (80.7% response) was superior to sertraline alone (54.9%), CBT alone (59.7%), and placebo (23.7%). Both monotherapies were superior to placebo. Combined treatment was the clear winner — one of the most robust combination therapy effects in child psychiatry.
13 OCD in Children & Adolescents
Obsessive-compulsive disorder affects 1–3% of children and adolescents, with bimodal onset: early childhood (mean age 10, more common in males) and late adolescence/early adulthood. Pediatric OCD is strongly familial, with a 4–8 times increased risk in first-degree relatives. Common obsession themes: contamination (38%), harm/aggressive (30%), symmetry/exactness (30%), sexual/religious (15–20%). Common compulsions: washing/cleaning (50%), checking (30%), repeating (35%), ordering/arranging (28%), counting (20%).
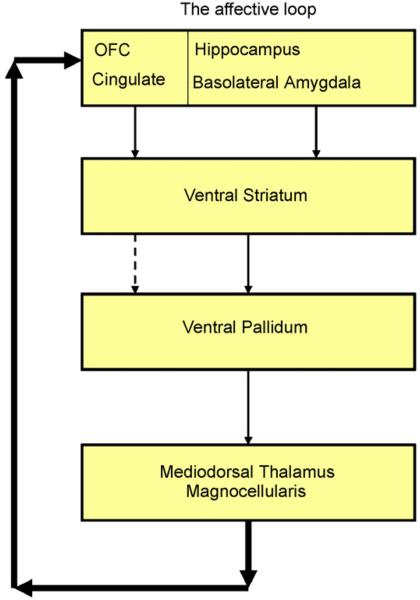
Assessment: CY-BOCS
The onset of OCD in childhood is often acute or subacute, and symptoms may initially be hidden from parents (children may perform rituals secretly or attribute their avoidance to other reasons). Common presentations by age: preschool (rigid routines, repetitive questioning, elaborate bedtime rituals), school-age (contamination fears, checking, symmetry, harm obsessions, homework perfectionism), adolescent (sexual/religious obsessions, mental rituals, existential obsessions). The PANDAS/PANS phenotype should be considered when onset is abrupt and dramatic (see below).
The Children's Yale-Brown Obsessive Compulsive Scale (CY-BOCS) is the gold standard clinician-rated measure for pediatric OCD severity. It separately rates obsessions and compulsions on five dimensions: time, interference, distress, resistance, and control (each 0–4). Total score ranges 0–40. Score interpretation: 0–7 subclinical, 8–15 mild, 16–23 moderate, 24–31 severe, 32–40 extreme. A 25–35% reduction in CY-BOCS score constitutes a clinically meaningful response to treatment.
Treatment
Exposure and Response Prevention (ERP) is the gold standard psychotherapy for pediatric OCD, with response rates of 50–70%. ERP involves constructing a fear hierarchy, then systematically exposing the child to anxiety-provoking stimuli while preventing the compulsive ritual. Adaptations for children include using developmentally appropriate language ("bossing back" OCD), involving parents as exposure coaches, gamifying the hierarchy, and externalizing OCD as the enemy ("what does OCD want you to do? Let's do the opposite"). ERP is typically delivered in 12–16 sessions.
SRI pharmacotherapy is indicated for moderate-severe OCD or when ERP alone is insufficient. Pediatric OCD requires higher SRI doses than depression and longer duration to response (8–12 weeks). FDA-approved SRIs for pediatric OCD:
| Medication | FDA-Approved Age | Starting Dose | Target OCD Dose | Max Dose |
|---|---|---|---|---|
| Fluoxetine | 7+ years | 10 mg/day | 20–60 mg/day | 80 mg/day |
| Fluvoxamine | 8+ years | 25 mg QHS | 100–200 mg/day | 200 mg (<12 yr); 300 mg (12–17) |
| Sertraline | 6+ years | 25 mg/day | 100–200 mg/day | 200 mg/day |
| Clomipramine | 10+ years | 25 mg QHS | 100–200 mg/day | 3 mg/kg/day or 200 mg (whichever is less) |
Augmentation strategies for SRI-resistant OCD: add low-dose aripiprazole (2.5–10 mg), risperidone (0.25–2 mg), or combine SRI with ERP intensification. The POTS II trial showed that adding 7 sessions of ERP to an adequate SRI trial produced significantly better outcomes than adding medication management or adding instructions in CBT alone.
OCD Subtypes & Related Conditions in Youth
Hoarding (now a separate DSM-5 diagnosis) can present in childhood; unlike adult hoarding, pediatric hoarding often involves collecting unusual items (broken toys, wrappers, rocks) and is frequently associated with ADHD. Body Dysmorphic Disorder (BDD) has typical onset in adolescence (mean age 16); adolescents obsess about perceived appearance defects (most commonly skin, hair, nose); BDD responds to the same treatments as OCD (SRIs + CBT/ERP targeting appearance-checking and mirror avoidance). Trichotillomania (hair-pulling disorder) and excoriation disorder (skin-picking) are classified with OCD in DSM-5; onset often in adolescence; habit reversal training (HRT) is the first-line behavioral treatment; SRIs are less effective than for OCD, but NAC has emerging evidence.
Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS) and its broader category Pediatric Acute-onset Neuropsychiatric Syndrome (PANS) describe acute, dramatic onset of OCD and/or tic symptoms temporally associated with Group A streptococcal infection (PANDAS) or other infectious/inflammatory triggers (PANS). Criteria: abrupt, dramatic onset of OCD or severely restricted food intake; concurrent additional neuropsychiatric symptoms (anxiety, emotional lability, irritability, aggression, behavioral regression, deterioration in school performance, sensory/motor abnormalities, somatic signs including sleep disturbance, enuresis, urinary frequency). Treatment of the underlying infection (antibiotics) plus standard OCD treatment. Anti-inflammatory therapies (NSAIDs, IVIG, plasmapheresis) are used in severe cases but remain controversial.
14 Disruptive Behavior Disorders — ODD & Conduct Disorder
Oppositional defiant disorder (ODD) and conduct disorder (CD) are the disruptive, impulse-control, and conduct disorders most frequently encountered in child psychiatry. ODD affects 3–6% of children; CD affects 2–10%. Both are more common in males (2–3:1 for ODD, up to 4:1 for CD). ODD is a risk factor for CD, but most children with ODD do NOT develop CD.
ODD — DSM-5-TR Criteria
A pattern of angry/irritable mood, argumentative/defiant behavior, or vindictiveness lasting at least 6 months, with at least 4 of the following symptoms exhibited during interaction with at least one individual who is NOT a sibling:
Angry/Irritable Mood: (1) Often loses temper. (2) Often touchy or easily annoyed. (3) Often angry and resentful.
Argumentative/Defiant Behavior: (4) Often argues with authority figures or adults. (5) Often actively defies or refuses to comply with rules or requests. (6) Often deliberately annoys others. (7) Often blames others for their mistakes or misbehavior.
Vindictiveness: (8) Has been spiteful or vindictive at least twice within past 6 months.
Frequency thresholds for children <5 years: most days for at least 6 months. For children ≥5 years: at least once per week for 6 months (except vindictiveness). Severity: Mild (symptoms confined to 1 setting), Moderate (2 settings), Severe (3+ settings).
Conduct Disorder — DSM-5-TR Criteria
A repetitive and persistent pattern of behavior violating the basic rights of others or major age-appropriate societal norms, with at least 3 of 15 criteria in the past 12 months and at least 1 in the past 6 months across four categories: Aggression to people and animals (bullies/threatens/intimidates; initiates physical fights; used a weapon; physically cruel to people; physically cruel to animals; stolen while confronting victim; forced sexual activity). Destruction of property (deliberately set fires to cause damage; deliberately destroyed others' property). Deceitfulness or theft (broken into house/building/car; lies for gain or to avoid obligations; stolen nontrivial items without confrontation). Serious violations of rules (stays out at night despite parental rules, beginning before age 13; runaway from home overnight at least twice; truant from school before age 13).
Subtypes by onset: Childhood-onset type (at least 1 criterion before age 10, worse prognosis), Adolescent-onset type (no criteria before age 10, better prognosis), Unspecified onset.
Callous-Unemotional (CU) Specifier: Limited Prosocial Emotions
DSM-5 added the specifier "with limited prosocial emotions" when at least 2 of the following are present persistently (over 12+ months, in multiple settings): (1) Lack of remorse or guilt. (2) Callous — lack of empathy. (3) Unconcerned about performance (school, work). (4) Shallow or deficient affect. The CU specifier identifies a subgroup (~25–30% of CD) with more severe aggression, poorer treatment response, distinct neurobiology (reduced amygdala reactivity to fear/distress cues), and higher risk of adult antisocial personality disorder and psychopathy.
Treatment Approaches
Parent Management Training (PMT) is the most evidence-based intervention for ODD and mild-moderate CD in children under 12. Programs include the Oregon model (PMTO), Triple P, and Incredible Years. Core principles: positive reinforcement of desired behaviors, consistent consequences, reducing coercive parent-child cycles, improving parent-child relationship quality. Multisystemic Therapy (MST) is an intensive, family- and community-based intervention for adolescents with severe CD and delinquent behavior; therapists work in the home, school, and community; reduces incarceration rates by 25–70%. No medication is FDA-approved for ODD or CD. Risperidone has the most evidence for reactive aggression in children with subaverage IQ and disruptive behavior (RUPP studies). Stimulants effectively reduce aggression in children with comorbid ADHD.
Aggression Subtypes & Management
Distinguishing aggression subtypes guides treatment selection. Reactive (impulsive, affective) aggression is provoked, emotionally driven, "hot-blooded," associated with threat perception, and linked to amygdala hyperreactivity. It is more common in ADHD, DMDD, and PTSD. Treatment: address underlying condition (stimulants for ADHD reduce reactive aggression by 50–60%), PMT, CBT anger management, risperidone for severe cases. Proactive (instrumental, predatory) aggression is planned, goal-directed, "cold-blooded," used to achieve a desired outcome, and associated with CU traits. It is more treatment-resistant and associated with worse long-term prognosis. PMT is less effective; treatment focuses on reward-based behavioral programs (as children with CU traits are less responsive to punishment but do respond to reward).
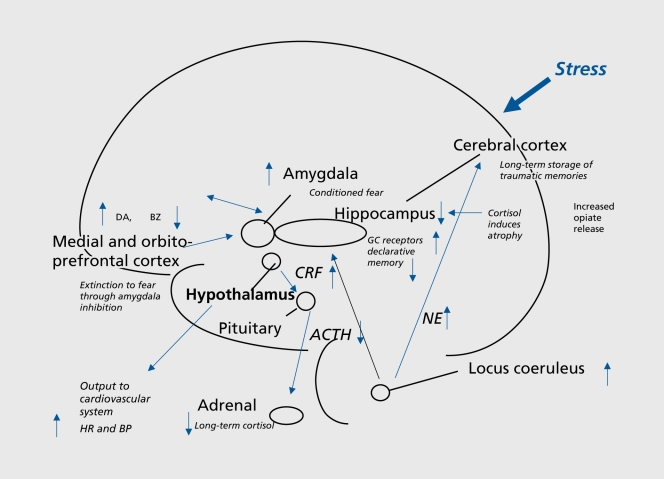
15 PTSD & Trauma in Children
Approximately two-thirds of children experience at least one traumatic event by age 16. Traumatic events include physical, sexual, or emotional abuse; neglect; witnessing domestic violence; community violence; natural disasters; motor vehicle accidents; sudden loss of a loved one; medical trauma (painful procedures, ICU stays, cancer diagnosis); refugee/war experiences; and human trafficking. The type, chronicity, and developmental timing of trauma exposure influence the clinical presentation and treatment approach.
DSM-5 includes a separate PTSD criterion set for children aged 6 and under, recognizing that young children may express re-experiencing symptoms through repetitive play that reenacts traumatic themes, frightening dreams without recognizable trauma content, and trauma-specific reenactment. The threshold is also lower: only 1 symptom (rather than 3) is required in the combined avoidance/negative cognitions cluster. Rates of PTSD following trauma vary by event type: 25–50% after sexual abuse, 25–35% after interpersonal violence, 10–25% after physical abuse, 10–15% after accidents/natural disasters. DSM-5 provides a specific PTSD criterion set for children 6 years and younger, reflecting developmental differences in symptom expression.
Developmental Variations in PTSD Presentation
| Age Group | Expression of PTSD |
|---|---|
| Preschool (0–5 years) | Separation anxiety, regression (loss of toileting, language skills), repetitive play reenacting trauma, new fears not directly related to trauma, sleep disturbances, somatic complaints, clinginess, aggression, constricted affect |
| School-age (6–12 years) | Intrusive thoughts, trauma-specific fears, reenactment in play, cognitive distortions (guilt, shame), concentration difficulties, school performance decline, psychosomatic symptoms, time skew (believing they can predict future trauma) |
| Adolescents (13–17 years) | Presentation more similar to adults: flashbacks, nightmares, emotional numbing, avoidance, hypervigilance, irritability, risk-taking behavior, substance use, self-harm, dissociation |
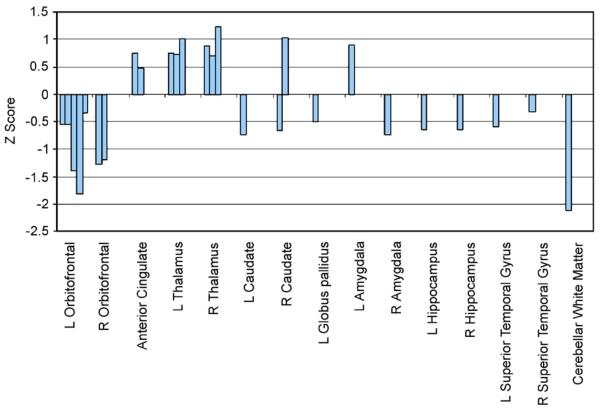
Adverse Childhood Experiences (ACEs)
The ACE study (Felitti et al., 1998) identified 10 categories of childhood adversity: physical abuse, emotional abuse, sexual abuse, physical neglect, emotional neglect, household substance abuse, household mental illness, parental separation/divorce, domestic violence against mother, incarcerated household member. ACE scores correlate with adult health outcomes in a dose-response relationship: ACE score of 4+ associated with 4–12 times increased risk for alcoholism, drug abuse, depression, and suicide attempt; 2–4 times increased risk for smoking, STIs, obesity, heart disease, cancer, and liver disease. Biological mechanisms include HPA axis dysregulation, chronic inflammation, epigenetic changes, and altered brain development (reduced hippocampal and PFC volume, amygdala hyperreactivity).
Trauma-Focused CBT (TF-CBT)
TF-CBT is the gold standard treatment for pediatric PTSD, with the largest evidence base of any trauma-specific therapy (15+ RCTs). It is a components-based model for children ages 3–18 and their non-offending caregivers, typically delivered in 12–25 sessions. The components are summarized by the acronym PRACTICE: Psychoeducation and parenting skills, Relaxation skills, Affective modulation, Cognitive coping and processing, Trauma narrative and processing, In-vivo mastery of trauma reminders, Conjoint child-parent sessions, Enhancing future safety and development. Response rates are 60–80% for significant symptom reduction.
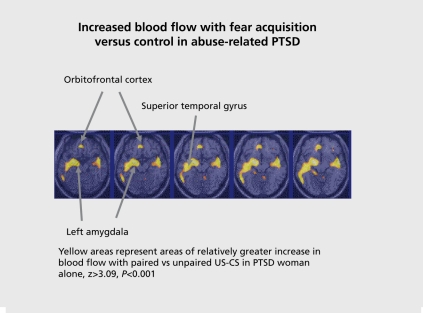
Complex Trauma
Complex trauma refers to exposure to multiple, chronic, interpersonal traumas (typically beginning in early childhood) within the caregiving system. Unlike single-incident PTSD, complex trauma produces pervasive developmental disruption: affect dysregulation, dissociation, negative self-concept, disturbed attachment patterns, behavioral problems, and somatic disturbances. Though not a formal DSM-5 diagnosis, "developmental trauma disorder" has been proposed (van der Kolk) to capture this presentation. Treatment requires a phased approach: (1) safety and stabilization, (2) trauma processing (TF-CBT or adapted models), (3) reintegration and future-oriented work.
Screening for Trauma
The UCLA PTSD Reaction Index is the most widely used trauma screening measure for children ages 7–18, available in child, adolescent, and parent versions. It maps to DSM-5 PTSD criteria and provides a total severity score. The Child PTSD Symptom Scale (CPSS-5) is a 27-item child self-report for ages 8–18 that also maps to DSM-5 criteria. The Trauma Symptom Checklist for Children (TSCC) assesses trauma-related symptoms including dissociation, anxiety, depression, anger, and sexual concerns; it has validity scales to detect under- and over-reporting. For commercial sexual exploitation of children (CSEC): the CSE-IT (Commercial Sexual Exploitation — Identification Tool) helps professionals identify youth at risk of or experiencing sex trafficking.
16 Attachment Disorders — RAD & DSED
Attachment disorders arise from severely inadequate caregiving in early childhood (before age 5). Both DSM-5 diagnoses — Reactive Attachment Disorder (RAD) and Disinhibited Social Engagement Disorder (DSED) — require a history of social neglect, frequent changes in primary caregivers, or rearing in institutions with high child-to-caregiver ratios.
Reactive Attachment Disorder (RAD)
RAD is an internalizing disorder characterized by a consistent pattern of inhibited, emotionally withdrawn behavior toward adult caregivers: the child rarely or minimally seeks comfort when distressed and rarely or minimally responds to comfort when offered. Additionally, the child shows persistent social and emotional disturbance with at least 2 of: minimal social/emotional responsiveness to others, limited positive affect, episodes of unexplained irritability, sadness, or fearfulness during nonthreatening interactions. Symptoms must be present before age 5. RAD should NOT be diagnosed in children who meet criteria for ASD.
Disinhibited Social Engagement Disorder (DSED)
DSED is an externalizing pattern in which the child actively approaches and interacts with unfamiliar adults, exhibiting at least 2 of: reduced/absent reticence in approaching unfamiliar adults, overly familiar verbal or physical behavior, diminished checking back with caregiver after venturing away (even in unfamiliar settings), willingness to go off with an unfamiliar adult with minimal/no hesitation. Unlike RAD, DSED can persist even after placement in a nurturing caregiving environment and can be diagnosed after age 5.
Key Differential: RAD vs ASD
| Feature | RAD | ASD |
|---|---|---|
| History of pathogenic care | Required | Not required |
| Social reciprocity | Absent due to inhibition; improves with adequate care | Qualitatively different; restricted interests and repetitive behaviors present |
| Restricted/repetitive behaviors | Absent | Core feature (Criterion B) |
| Response to nurturing environment | Significant improvement expected | Social deficits persist regardless of caregiving quality |
| Language | May be delayed due to deprivation but improves with enrichment | Qualitative abnormalities in communication |
Treatment
RAD prevalence in the general population is very low (<1–2%) but much higher in high-risk samples: 40–50% of children raised in institutional care show features of RAD or DSED. Both diagnoses require evidence of insufficient care as a presumptive cause. If a child has never experienced pathogenic care, neither diagnosis can be made regardless of symptom presentation.
The primary intervention is ensuring a stable, nurturing caregiving environment. No medication treats attachment disorders. Evidence-based approaches include: fostering a consistent attachment relationship, enhancing caregiver sensitivity and responsiveness (e.g., through Child-Parent Psychotherapy, Circle of Security), and addressing comorbid problems (ADHD, language delays, behavioral dysregulation). Attachment therapies involving physical restraint, holding, or coercion ("rebirthing therapy") are dangerous, unproven, and explicitly condemned by AACAP.
17 Anorexia Nervosa & Bulimia in Adolescents
Eating disorders have the highest mortality rate of any psychiatric illness. Anorexia nervosa (AN) has a lifetime prevalence of 0.5–2% in females and a mortality rate of 5–10% per decade (from medical complications and suicide). Bulimia nervosa (BN) affects 1–3% of adolescent females. Peak onset for both is 14–18 years. Males comprise approximately 10–25% of cases and are often underdiagnosed.
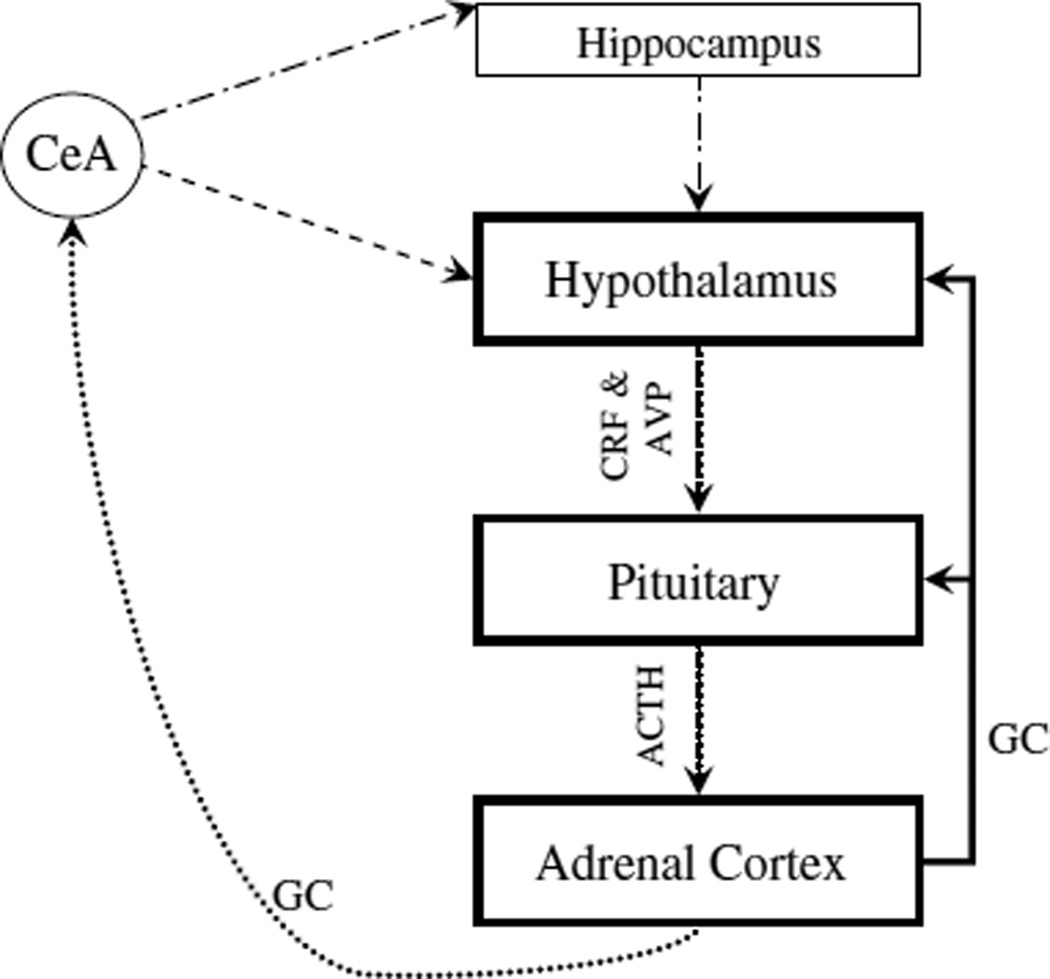
Medical Complications
| System | Anorexia Nervosa | Bulimia Nervosa |
|---|---|---|
| Cardiovascular | Bradycardia (HR <50), hypotension, QTc prolongation, mitral valve prolapse, pericardial effusion, heart failure (refeeding) | Arrhythmias from electrolyte abnormalities, ipecac-induced cardiomyopathy (rare) |
| Electrolyte | Hypokalemia, hyponatremia, hypophosphatemia (especially during refeeding), hypomagnesemia | Hypokalemia (vomiting, laxatives), metabolic alkalosis (vomiting), metabolic acidosis (laxative abuse) |
| Endocrine | Amenorrhea (hypothalamic, low LH/FSH/estrogen), low T3 syndrome (euthyroid sick), elevated cortisol, low IGF-1, growth retardation | Menstrual irregularities less common than AN |
| GI | Delayed gastric emptying, constipation, SMA syndrome, hepatic steatosis | Parotid gland enlargement (sialadenosis), dental erosion (perimolysis), esophageal tears (Mallory-Weiss), Russell's sign (calluses on knuckles) |
| Skeletal | Osteopenia/osteoporosis (irreversible if prolonged), stress fractures, bone age delay | Usually normal bone density |
| Hematologic | Leukopenia, anemia, thrombocytopenia, bone marrow hypoplasia | Usually normal |
| Dermatologic | Lanugo hair, hair loss, dry skin, acrocyanosis, carotenodermia | Russell's sign, perioral irritation |
Refeeding Syndrome
Refeeding syndrome is a potentially fatal metabolic complication occurring when nutrition is reintroduced to a severely malnourished patient. As insulin rises with carbohydrate intake, phosphate, potassium, and magnesium shift intracellularly, causing dangerous hypophosphatemia (the hallmark), hypokalemia, hypomagnesemia, and fluid retention. Complications include cardiac arrhythmias, heart failure, respiratory failure, rhabdomyolysis, seizures, and death. Prevention: start at 1,200–1,500 kcal/day (or lower in severely malnourished patients, per "start low, advance slow" protocol), advance by 200–400 kcal every 1–2 days, supplement phosphorus prophylactically, monitor electrolytes (phosphate, potassium, magnesium) daily for the first 5–7 days, check for peripheral edema and fluid overload, cardiac monitoring.
Inpatient Admission Criteria (AACAP/APA/SAHM Guidelines)
Vital signs: HR <50 bpm (daytime) or <45 (sleeping), systolic BP <90/45, temperature <96°F (35.6°C), orthostatic changes (ΔHR >20 bpm or ΔSBP >20 mmHg). Weight: <75% of median BMI for age/sex, acute weight loss with food refusal. Labs: K <3.2 mEq/L, phosphorus <2.5 mg/dL, glucose <60 mg/dL. EKG: QTc >460 ms, arrhythmia. Psychiatric: Suicidal ideation, comorbid condition interfering with outpatient treatment, failure of outpatient treatment. Other: Syncope, seizures, uncontrollable binging/purging, severe dehydration.
Family-Based Treatment (FBT/Maudsley Approach)
FBT is the most evidence-based outpatient treatment for adolescent AN and the AACAP first-line recommendation. It is a 3-phase model over approximately 6–12 months: Phase 1 (Weight Restoration): Parents take complete control of the adolescent's eating, supervised meals, prevent exercise and purging; the illness is externalized. Phase 2 (Returning Control): As weight normalizes, eating control is gradually returned to the adolescent with parental oversight. Phase 3 (Adolescent Issues): Address normal adolescent developmental tasks disrupted by the illness (identity, autonomy, peer relationships). Response rates at end of treatment: 40–50% remission; at 5-year follow-up: 75–90% have good or intermediate outcome.
For BN in adolescents, CBT adapted for adolescents (CBT-A) and FBT-BN are both effective. Fluoxetine (60 mg/day) is the only FDA-approved medication for BN (approved for adults; used off-label in adolescents). No medication has demonstrated efficacy for the core symptoms of AN.
Laboratory Workup for Eating Disorders
| Test | Expected Findings | Clinical Significance |
|---|---|---|
| BMP (Na, K, Cl, CO2, BUN, Cr, glucose) | Hypokalemia, hypochloremic metabolic alkalosis (purging), hypoglycemia (AN), elevated BUN (dehydration) | Electrolyte abnormalities are the most common cause of morbidity; hypokalemia causes arrhythmias |
| Phosphorus, magnesium, calcium | Hypophosphatemia (especially during refeeding), hypomagnesemia | Phosphorus is the hallmark of refeeding syndrome; monitor daily during initial refeeding |
| CBC | Leukopenia, anemia, thrombocytopenia (AN) | Bone marrow suppression from malnutrition; resolves with weight restoration |
| TSH, free T4, T3 | Low T3 syndrome (euthyroid sick), normal TSH (AN) | Do NOT treat with thyroid hormone; resolves with weight restoration |
| LFTs | Elevated transaminases (AN — starvation hepatitis or fatty liver during refeeding) | Transaminases >3x ULN warrant further evaluation; may indicate hepatic damage |
| EKG | Bradycardia, prolonged QTc, low voltage, ST/T wave changes | QTc >460 ms increases arrhythmia risk; bradycardia <50 bpm is admission criterion |
| DXA scan | Low bone mineral density (Z-score <−1.0) | If amenorrheic >6 months or low weight >6 months; osteopenia may be irreversible |
| Amylase | Elevated salivary amylase (purging) | Marker of purging frequency; parotid enlargement correlates |
18 ARFID & Other Feeding Disorders
Avoidant/Restrictive Food Intake Disorder (ARFID) is a DSM-5 diagnosis characterized by persistent failure to meet nutritional/energy needs (not due to body image disturbance or lack of food availability), manifested by significant weight loss or growth failure, nutritional deficiency, dependence on enteral feeding or oral supplements, or marked psychosocial interference. ARFID encompasses three phenotypes: sensory sensitivity (limited diet based on taste, texture, appearance, smell), lack of interest in eating (low appetite, poor interoceptive awareness), and fear of aversive consequences (choking phobia, fear of vomiting, pain). Prevalence in pediatric feeding disorder clinics is 5–14%.
ARFID vs AN
| Feature | ARFID | Anorexia Nervosa |
|---|---|---|
| Body image disturbance | Absent | Present (core feature) |
| Fear of weight gain | Absent | Present (core feature) |
| Typical onset | Early childhood (though can be any age) | Adolescence |
| Gender distribution | More equal; slight male predominance | Strong female predominance |
| Comorbidities | ASD, ADHD, anxiety | Depression, anxiety, OCD |
| Motivation for restriction | Sensory, lack of interest, fear of choking/vomiting | Drive for thinness, calorie restriction |
Pica
Pica is the persistent eating of nonnutritive, nonfood substances for at least 1 month, inappropriate to developmental level (not diagnosed before age 2), and not part of a culturally sanctioned practice. Common substances: dirt/clay (geophagy), ice (pagophagia — often associated with iron deficiency), paint chips (risk of lead poisoning), hair (trichotillophagia, which can cause trichobezoar/Rapunzel syndrome), starch (amylophagy). Pica is more common in children with intellectual disability (10–30%), ASD, and iron deficiency. Workup includes CBC, iron studies, lead level, zinc level. Treatment: address nutritional deficiencies, behavioral interventions, environmental modifications.
Rumination Disorder
Sensory-Based Feeding Difficulties
Many children — particularly those with ASD, sensory processing differences, or prematurity history — exhibit sensory-based feeding difficulties that do not meet full ARFID criteria but cause significant mealtime distress. Features include gagging or retching with new textures, extreme food selectivity (often limited to 5–10 "safe" foods, typically beige/crunchy items), inability to tolerate mixed textures, and aversion to specific colors or temperatures. Occupational therapy with a feeding specialist is the primary intervention, using systematic desensitization (food chaining, gradual texture advancement, sequential oral sensory approach — SOS). Parent coaching is essential: avoid force-feeding, reduce mealtime pressure, offer repeated low-pressure exposures (15–30 exposures may be needed before a child accepts a new food).
Rumination disorder involves repeated regurgitation of food for at least 1 month; food may be re-chewed, re-swallowed, or spit out. It is not attributable to a GI condition (e.g., GERD, pyloric stenosis) and does not occur exclusively during AN, BN, BED, or ARFID. Most common in infants and individuals with ID. In neurotypical individuals, rumination often begins as a pleasurable learned behavior. Treatment: diaphragmatic breathing training to prevent the abdominal pressure wave that initiates regurgitation; habit reversal training; in infants, increasing nurturing and stimulation during and after feeds.
19 Self-Harm & Suicidality in Youth
Suicide is the second leading cause of death among individuals aged 10–24 in the United States. Rates have increased approximately 60% since 2007. In the 2021 Youth Risk Behavior Survey (YRBS), 22% of high school students seriously considered suicide, 18% made a plan, and 10% made an attempt in the past year. Non-suicidal self-injury (NSSI) — deliberate, self-directed damage to body tissue without suicidal intent — affects 15–20% of adolescents and is distinct from suicidal behavior but is a significant risk factor for later suicide attempts.
NSSI vs Suicidal Self-Injury
| Feature | NSSI | Suicidal Self-Injury |
|---|---|---|
| Intent | Emotional regulation, self-punishment, communication; NOT to die | Intent to die |
| Methods | Cutting (most common), burning, hitting, scratching, biting | Overdose, hanging, firearms, jumping |
| Frequency | Often repetitive (dozens to hundreds of episodes) | Usually isolated or few events |
| Lethality | Low medical severity (superficial cuts/burns) | Higher medical severity |
| Functions | Affect regulation (70%), anti-dissociation, self-punishment, interpersonal influence | Escape from unbearable suffering |
Suicide Risk Assessment
The Ask Suicide-Screening Questions (ASQ) is a validated 4-question screening tool for ages 10–24 in medical settings: (1) In the past few weeks, have you wished you were dead? (2) In the past few weeks, have you felt that you or your family would be better off if you were dead? (3) In the past week, have you been having thoughts about killing yourself? (4) Have you ever tried to kill yourself? Any "yes" is a positive screen. If positive: (5) Are you having thoughts of killing yourself right now? (Yes = acute positive screen requiring immediate safety intervention.)
The Columbia Suicide Severity Rating Scale (C-SSRS) is a more comprehensive clinician-administered tool that classifies ideation intensity (wish to be dead, nonspecific active suicidal thoughts, active suicidal ideation with method, ideation with intent, ideation with plan and intent) and behavior (actual attempt, interrupted attempt, aborted attempt, preparatory behavior, NSSI). It provides a standardized framework for risk stratification and documentation.
Risk & Protective Factors
Individual: Previous attempt (strongest predictor), psychiatric disorder (depression, bipolar, SUD, conduct disorder), NSSI history, hopelessness, impulsivity, LGBTQ+ identity (3–4x higher attempt rate), access to lethal means (firearms — most common completed suicide method in youth). Family: Family history of suicide, parental psychiatric illness, family discord, history of abuse/neglect, recent loss. Social: Bullying (victim or perpetrator), social isolation, exposure to peer suicide (contagion), media portrayal of suicide. Protective: Family connectedness, school connectedness, problem-solving skills, mental health treatment, restricted access to means, reasons for living, cultural/religious beliefs against suicide.
Safety Planning for Adolescents
The Stanley-Brown Safety Plan adapted for adolescents includes: (1) Recognizing warning signs ("What tells you a crisis is building?"); (2) Internal coping strategies the teen can do alone (distraction, relaxation, physical activity); (3) People and social settings that provide distraction (friends, going to a public place); (4) People the teen can ask for help (trusted adults, friends who know); (5) Professionals and crisis contacts (therapist, crisis line 988, crisis text line, emergency department); (6) Making the environment safe (means restriction — specifically firearms, medications, sharps). Means restriction counseling is essential: storing firearms locked and unloaded with ammunition separate reduces youth firearm suicide by 73%. Securely store medications, particularly acetaminophen, tricyclics, and opioids.
Management After a Suicide Attempt
Following a suicide attempt, the priority sequence is: (1) Medical stabilization (overdose management, wound care). (2) Psychiatric evaluation (ideation, intent, lethality of attempt, ongoing plan, access to means). (3) Safety determination (can the patient be safely discharged with a safety plan, or does inpatient admission required?). (4) Disposition: inpatient psychiatric hospitalization is indicated when there is ongoing suicidal intent, highly lethal attempt, psychosis, intoxication, lack of protective factors, or inability to safety plan. Partial hospitalization (PHP) or intensive outpatient (IOP) may be appropriate for patients with suicidal ideation without active intent who have engaged caregivers and a safety plan. (5) Outpatient follow-up within 72 hours of discharge — the highest-risk period for re-attempt is the first week after psychiatric discharge. (6) Collaborative safety planning with the family, including lethal means restriction. (7) Initiate evidence-based treatment: DBT-A for chronic suicidality/NSSI, CBT-SP (CBT for Suicide Prevention), or SAFETY (Safe Alternatives for Teens and Youth).
20 Stimulants & ADHD Medications — Complete Drug Table
Stimulant medications are the most effective pharmacological treatment for ADHD, with effect sizes of 0.8–1.0 (large). They act by blocking the dopamine transporter (DAT) and norepinephrine transporter (NET) in the prefrontal cortex and striatum. There are two classes: methylphenidate-based (MPH) and amphetamine-based (AMPH). Amphetamines additionally promote vesicular release of dopamine and norepinephrine. All stimulants are DEA Schedule II controlled substances.
Methylphenidate Formulations
| Brand Name | Formulation | Release Mechanism | Onset | Duration | Starting Dose | Max Dose |
|---|---|---|---|---|---|---|
| Ritalin | IR tablet | Immediate release | 20–30 min | 3–4 hr | 5 mg BID | 60 mg/day |
| Ritalin LA | ER capsule (can sprinkle) | 50% IR / 50% DR beads | 30 min | 8–10 hr | 10–20 mg QAM | 60 mg/day |
| Concerta | OROS tablet (do NOT crush) | 22% IR overcoat / 78% osmotic push | 30–60 min | 10–12 hr | 18 mg QAM | 72 mg/day |
| Focalin | IR tablet (d-MPH) | Immediate release | 20–30 min | 4–5 hr | 2.5 mg BID | 20 mg/day |
| Focalin XR | ER capsule (d-MPH, can sprinkle) | 50% IR / 50% DR beads | 30 min | 8–12 hr | 5 mg QAM | 30 mg (child); 40 mg (adult) |
| Daytrana | Transdermal patch | Continuous delivery while worn | 2 hr | Wear time + 3 hr after removal | 10 mg/9 hr patch | 30 mg/9 hr patch |
| Quillivant XR | Extended-release liquid | 20% IR / 80% ER | 30–45 min | 12 hr | 20 mg QAM | 60 mg/day |
| Jornay PM | DR/ER capsule (evening dosing) | Delayed onset by 8–10 hr | Next morning | 12 hr from onset | 20 mg QPM | 100 mg/day |
Amphetamine Formulations
| Brand Name | Formulation | Release Mechanism | Onset | Duration | Starting Dose | Max Dose |
|---|---|---|---|---|---|---|
| Adderall | IR tablet (mixed amphetamine salts) | Immediate release | 20–30 min | 4–6 hr | 5 mg QD-BID | 40 mg/day |
| Adderall XR | ER capsule (can sprinkle) | 50% IR / 50% DR beads | 30 min | 10–12 hr | 5–10 mg QAM | 30 mg (child); 40 mg (adult) |
| Vyvanse | Capsule/chewable (lisdexamfetamine) | Prodrug — cleaved in blood by RBCs | 1–2 hr | 10–14 hr | 20–30 mg QAM | 70 mg/day |
| Dexedrine | IR tablet (dextroamphetamine) | Immediate release | 20–30 min | 4–5 hr | 5 mg QD-BID | 40 mg/day |
| Dexedrine Spansule | ER capsule | Sustained-release beads | 30–60 min | 6–10 hr | 5–10 mg QAM | 40 mg/day |
| Mydayis | Triple-bead ER capsule (MAS) | 33% IR / 33% DR / 33% delayed ER | 30 min | 16 hr | 12.5 mg QAM | 25 mg (<13 yr); 50 mg (13+) |
| Evekeo | IR tablet (amphetamine sulfate) | Immediate release | 20–30 min | 4–6 hr | 5 mg QD-BID | 40 mg/day |
Common Side Effects & Management
Appetite suppression (most common, 60–80%): give medication after breakfast, ensure adequate evening meal and snack. Insomnia: use shorter-acting formulations, dose earlier in the day, consider melatonin 1–5 mg at bedtime. Headache and stomachache: usually transient, take with food. Emotional blunting or rebound irritability: adjust dose or formulation. Growth suppression: average 1 cm/year height deficit and 1–2 kg weight deficit; monitor growth curves every 3 months; consider drug holidays. Cardiovascular: average HR increase 3–6 bpm, BP increase 2–4 mmHg; clinically significant in rare cases; screen for cardiac risk factors before initiation.
Contraindications & Drug Interactions
Absolute contraindications to stimulants: concurrent MAOI use (or within 14 days of MAOI discontinuation), known hypersensitivity, structural cardiac abnormalities associated with sudden death risk. Relative contraindications: moderate-severe hypertension, active psychosis (stimulants can exacerbate psychotic symptoms), current substance use disorder (consider lisdexamfetamine or non-stimulants as they have lower abuse potential), tic disorders (not an absolute contraindication per TICS study but monitor), and narrow-angle glaucoma (for amphetamines). Key drug interactions: Stimulants may increase plasma levels of TCAs, phenytoin, and warfarin. MAOIs are absolutely contraindicated. Acidifying agents (ascorbic acid, fruit juice) decrease amphetamine absorption; alkalinizing agents (sodium bicarbonate) increase it. Concerta's OROS mechanism is not affected by GI pH.
Stimulant Misuse & Diversion
Stimulant diversion (giving or selling prescribed medications to others) occurs in 5–35% of college students with ADHD prescriptions. Risk factors include: immediate-release formulations (higher abuse potential), older age, comorbid substance use, conduct disorder traits. Protective strategies: use long-acting formulations (preferred), use prodrug formulations (lisdexamfetamine has lowest abuse potential due to requiring enzymatic cleavage), educate patients and families about secure medication storage, monitor prescription refills and pill counts, and use state prescription drug monitoring programs (PDMPs). Evidence from longitudinal studies (Wilens et al., 2003) demonstrates that treating ADHD with stimulants actually reduces subsequent substance use disorder risk by approximately 35–40%, countering the common parental concern that stimulants lead to addiction.
21 SSRIs & Antidepressants in Pediatrics
SSRIs are the first-line pharmacotherapy for pediatric depression, anxiety disorders, and OCD. They work by selectively blocking the serotonin transporter (SERT), increasing serotonin availability in the synaptic cleft. Onset of therapeutic effect is typically 2–4 weeks for anxiety, 4–6 weeks for depression, and 6–12 weeks for OCD.
FDA-Approved Pediatric Indications
| Medication | MDD | OCD | Anxiety | Min Age | Starting Dose | Typical Target |
|---|---|---|---|---|---|---|
| Fluoxetine (Prozac) | Age 8+ | Age 7+ | Off-label | 7 | 10 mg/day | 20–60 mg |
| Escitalopram (Lexapro) | Age 12+ | Off-label | Off-label | 12 | 5–10 mg/day | 10–20 mg |
| Sertraline (Zoloft) | Off-label | Age 6+ | Off-label | 6 | 25 mg/day | 50–200 mg |
| Fluvoxamine (Luvox) | Off-label | Age 8+ | Off-label | 8 | 25 mg QHS | 100–300 mg |
| Duloxetine (Cymbalta) | Off-label | Off-label | GAD age 7+ | 7 | 30 mg/day | 60–120 mg |
Black Box Warning — Detailed Analysis
The FDA black box warning (2004) was based on pooled analysis of 24 short-term pediatric antidepressant trials (4,400 patients). Findings: antidepressant-treated patients had approximately 4% incidence of suicidal thinking/behavior vs 2% for placebo (NNH = 50). There were zero completed suicides in any trial. The risk was highest in the first 1–2 months and with dose changes. The warning applies to ALL antidepressants (not just SSRIs) in patients under 25. The Bridge II-TR study (2007) later showed that treatment benefits for depression (NNT = 10) substantially outweigh suicidality risk (NNH = 112) when analysis uses more stringent definitions of suicidal events.
Activation Syndrome
Activation syndrome is an adverse effect seen in 3–8% of children starting SSRIs, characterized by increased agitation, restlessness, insomnia, irritability, impulsivity, disinhibition, silliness, and hyperactivity occurring within the first 1–2 weeks. It is distinct from mania and typically resolves with dose reduction. Risk factors: younger age, higher starting dose, rapid titration. Management: reduce dose, slow titration, reassess. If activation is severe or includes suicidal ideation, discontinue the medication.
Serotonin Syndrome
Serotonin syndrome is a potentially life-threatening condition resulting from excess serotonergic activity. It presents with a triad of altered mental status (agitation, confusion, delirium), autonomic instability (hyperthermia, tachycardia, diaphoresis, BP lability, diarrhea), and neuromuscular excitability (clonus — especially lower extremity and ocular, hyperreflexia, myoclonus, tremor, rigidity). Risk is highest when combining serotonergic agents (SSRI + MAOI, SSRI + tramadol, SSRI + triptans, SSRI + dextromethorphan, SSRI + linezolid). The Hunter Criteria require a serotonergic agent plus one of: spontaneous clonus; inducible clonus + agitation or diaphoresis; ocular clonus + agitation or diaphoresis; tremor + hyperreflexia; or hypertonia + temperature >38°C + ocular or inducible clonus. Treatment: discontinue all serotonergic agents, benzodiazepines, cooling, cyproheptadine (serotonin antagonist) 4–8 mg PO initial dose.
22 Antipsychotics in Children
Antipsychotic use in children and adolescents has increased dramatically (5–7-fold since the 1990s), primarily second-generation (atypical) antipsychotics. FDA-approved pediatric indications include ASD-associated irritability, bipolar mania, schizophrenia (age 13+), and Tourette syndrome. Metabolic side effects are a major concern in youth, who are more vulnerable than adults to antipsychotic-induced weight gain and metabolic syndrome.

FDA-Approved Pediatric Indications by Agent
| Medication | Schizophrenia | Bipolar I Mania | ASD Irritability | Tourette | Other |
|---|---|---|---|---|---|
| Risperidone (Risperdal) | 13–17 yr | 10–17 yr | 5–16 yr | — | — |
| Aripiprazole (Abilify) | 13–17 yr | 10–17 yr | 6–17 yr | 6–18 yr | Adjunct MDD 13+ (off-label in peds) |
| Quetiapine (Seroquel) | 13–17 yr | 10–17 yr | — | — | — |
| Olanzapine (Zyprexa) | 13–17 yr | 13–17 yr | — | — | — |
| Paliperidone (Invega) | 12–17 yr | — | — | — | — |
| Lurasidone (Latuda) | 13–17 yr | 10–17 yr (depressive episodes) | — | — | Bipolar depression 10–17 yr |
| Pimozide (Orap) | — | — | — | 12+ yr | — |
| Haloperidol (Haldol) | — | — | — | 3+ yr | Severe behavioral problems 3+ yr |
Metabolic Monitoring Protocol
Baseline (before starting): Weight, height, BMI percentile, waist circumference, fasting glucose, fasting lipid panel, HbA1c, blood pressure, personal/family history of diabetes, obesity, dyslipidemia, cardiovascular disease. Month 1: Weight, BMI. Month 2: Weight, BMI. Month 3: Weight, BMI, fasting glucose, fasting lipids, BP. Every 3 months thereafter: Weight, BMI. Every 6 months: Fasting glucose, HbA1c, BP, waist circumference. Annually: Fasting lipid panel. Prolactin: Baseline and if symptoms develop (galactorrhea, gynecomastia, menstrual irregularity, sexual dysfunction); risperidone/paliperidone have highest prolactin elevation risk.
Metabolic Risk by Agent
| Agent | Weight Gain | Dyslipidemia | Diabetes Risk | Prolactin | Sedation | EPS |
|---|---|---|---|---|---|---|
| Olanzapine | ++++ | ++++ | +++ | ++ | +++ | + |
| Quetiapine | +++ | +++ | ++ | + | ++++ | + |
| Risperidone | +++ | ++ | ++ | ++++ | ++ | ++ |
| Aripiprazole | ++ | + | + | +/− | + | ++ (akathisia) |
| Lurasidone | + | + | + | ++ | ++ | ++ |
| Paliperidone | ++ | ++ | ++ | ++++ | ++ | ++ |
Extrapyramidal Symptoms (EPS)
EPS in children include: Acute dystonia (involuntary muscle contractions — torticollis, oculogyric crisis, laryngospasm; treat with diphenhydramine or benztropine IM). Akathisia (subjective inner restlessness, inability to sit still; often mistaken for worsening agitation; treat with dose reduction, beta-blocker, or benzodiazepine). Parkinsonism (bradykinesia, rigidity, tremor; treat with dose reduction or anticholinergic). Tardive dyskinesia (TD) (late-onset involuntary movements, especially orofacial; usually after months-years of exposure; may be irreversible; valbenazine and deutetrabenazine are FDA-approved for TD in adults). Children on antipsychotics should be assessed for abnormal movements at every visit using the AIMS (Abnormal Involuntary Movement Scale).
23 Other Agents — Mood Stabilizers, Alpha-Agonists, Melatonin
Lithium in Adolescents
Lithium is FDA-approved for bipolar I disorder in children aged 7+ (mania and maintenance). Mechanism: multifactorial — inhibits inositol monophosphatase, modulates GSK-3beta, neuroprotective effects. Starting dose: 300 mg BID or TID. Serum levels: 0.8–1.2 mEq/L for acute mania, 0.6–1.0 mEq/L for maintenance. Draw trough level 12 hours post-dose, after 5 days at a stable dose. Monitoring: serum lithium level, BUN/creatinine, TSH, calcium (q3–6 months); CBC with differential; pregnancy test in females; EKG if cardiac concerns. Side effects: GI (nausea, diarrhea), tremor, polyuria/polydipsia (nephrogenic diabetes insipidus), hypothyroidism (20–30%), weight gain, acne. Toxicity signs: coarse tremor, vomiting, ataxia, confusion, seizures — risk increases with dehydration, NSAIDs, ACE inhibitors, thiazide diuretics.
Valproate (Divalproex Sodium)
Not FDA-approved for pediatric bipolar disorder but widely used off-label. Starting dose: 10–15 mg/kg/day divided BID-TID; target serum level 50–125 mcg/mL. Black box warnings: hepatotoxicity (highest risk in children <2 years on polypharmacy), pancreatitis, teratogenicity (neural tube defects in 1–2% of exposed pregnancies; lower IQ in children exposed in utero; CONTRAINDICATED in females of reproductive potential without reliable contraception). Other side effects: weight gain, alopecia, tremor, GI upset, thrombocytopenia, polycystic ovarian syndrome. Monitoring: LFTs, CBC with platelets, valproate level, lipase if GI symptoms, pregnancy test.
Alpha-2 Agonists
| Medication | Primary Uses in Youth | Dosing | Key Points |
|---|---|---|---|
| Guanfacine ER (Intuniv) | ADHD (FDA-approved 6–17 yr), tics, aggression, insomnia | 1–4 mg QHS (6–12 yr), 1–7 mg (13–17 yr) | Selective alpha-2A; less sedating than clonidine; monitor BP, HR; taper over 3–7 days to discontinue |
| Clonidine ER (Kapvay) | ADHD (FDA-approved 6–17 yr), tics, insomnia, aggression | 0.1 mg QHS, increase by 0.1 mg weekly; max 0.4 mg/day divided BID | Non-selective alpha-2; more sedating; rebound hypertension if stopped abruptly; available as patch |
| Clonidine IR | Insomnia (off-label), ADHD adjunct, tics, aggression, opioid withdrawal | 0.05–0.1 mg QHS (start); max 0.3–0.4 mg/day | Short duration; useful for sleep onset; monitor for hypotension |
Melatonin
Melatonin is the most commonly used sleep aid in pediatrics, available over-the-counter. It is an endogenous hormone produced by the pineal gland that regulates circadian rhythm. Useful for sleep onset delay (the most common sleep problem in children with ADHD, ASD, and anxiety). Dosing: start low at 0.5–1 mg, given 30–60 minutes before desired bedtime; increase to 3–5 mg if needed (some experts suggest up to 10 mg for ASD). Higher doses are not necessarily more effective. Slow-release formulations may help with sleep maintenance. Melatonin has an excellent safety profile in short-to-medium term studies; long-term safety data in children are limited. Common side effects: morning grogginess, vivid dreams, headache. Caution: OTC melatonin supplements are unregulated and may contain varying actual doses (studies show 71–478% of labeled content).
N-Acetylcysteine (NAC)
NAC is a glutamate modulator and antioxidant used off-label as an adjunctive treatment in several pediatric psychiatric conditions. Evidence is emerging for: irritability in ASD (1,200–2,400 mg/day in divided doses; one RCT showed significant improvement in irritability vs placebo), trichotillomania (1,200–2,400 mg/day), OCD augmentation (limited evidence), and substance use disorders (cannabis use in adolescents). Side effects are generally mild: GI upset, nausea. NAC is available over-the-counter and is well-tolerated. Evidence is preliminary for most indications, and it should be considered adjunctive rather than primary treatment.
Benzodiazepines in Pediatric Psychiatry
Benzodiazepines have a very limited role in pediatric psychiatry due to risks of dependence, tolerance, disinhibition (paradoxical agitation — more common in children than adults), and cognitive impairment. Appropriate uses: acute agitation/catatonia (lorazepam 0.5–2 mg IM/PO), acute anxiety crisis (short-term bridge while waiting for SSRI to take effect), and procedural anxiety. Benzodiazepines are NOT recommended as ongoing treatment for anxiety disorders in youth — SSRIs and CBT are first-line. If used, prescribe for the shortest duration possible with a clear taper plan.
Before prescribing any sleep aid, ensure adequate sleep hygiene: consistent bedtime and wake time (even weekends), bedroom dark/cool/quiet, no screens 1 hour before bed (blue light suppresses endogenous melatonin), no caffeine after noon, regular exercise (not within 2 hours of bedtime), bedtime routine (bath, reading), no naps after 3 PM, bed used only for sleep. For stimulant-treated ADHD patients with insomnia: ensure the stimulant duration does not extend into evening, consider earlier dosing or shorter-acting formulation.
24 Evidence-Based Psychotherapies
Psychotherapy is a first-line or co-first-line treatment for most pediatric psychiatric disorders. The choice of modality depends on the diagnosis, child's age and cognitive development, family resources, and available providers. The following modalities have the strongest evidence bases in child and adolescent psychiatry.
Cognitive Behavioral Therapy (CBT)
CBT targets the relationship between thoughts, feelings, and behaviors. The cognitive model holds that maladaptive cognitions (automatic thoughts, intermediate beliefs, core beliefs) drive emotional distress and behavioral dysfunction. In children, common cognitive distortions include:
| Distortion | Description | Example in Youth |
|---|---|---|
| Catastrophizing | Expecting the worst-case scenario | "If I fail this test, I'll never get into college and my life is over" |
| All-or-nothing thinking | Seeing things as entirely good or bad | "If I'm not the best at soccer, I'm the worst" |
| Mind reading | Assuming you know what others think | "Everyone at lunch was thinking I'm weird" |
| Personalization | Blaming yourself for external events | "Mom and Dad are fighting because of me" |
| Fortune telling | Predicting negative outcomes | "I know I'm going to mess up my presentation" |
| Emotional reasoning | Treating feelings as facts | "I feel stupid, so I must be stupid" |
CBT It teaches children to identify cognitive distortions (catastrophizing, black-and-white thinking, personalization), challenge maladaptive thoughts, and develop coping skills. CBT is typically 12–16 sessions. Evidence level: Strong for depression (NNT 3–4), anxiety (NNT 2–3), OCD (NNT 2–3), PTSD, and behavioral problems. Adaptations for children include using concrete examples, visual aids, games, and stories to teach cognitive concepts. CBT requires a minimum cognitive developmental level (typically age 7–8+); for younger children, the behavioral component (exposure, behavioral activation) is emphasized over cognitive restructuring.
Dialectical Behavior Therapy for Adolescents (DBT-A)
DBT-A is adapted from Marsha Linehan's DBT for adults with borderline personality features. It targets adolescents with chronic suicidality, NSSI, and emotional dysregulation. The four skill modules are: mindfulness (present-moment awareness), distress tolerance (crisis survival skills without making things worse), emotion regulation (understanding and managing intense emotions), and interpersonal effectiveness (communicating needs while maintaining relationships). Added for adolescents: "walking the middle path" (dialectics of adolescent-parent conflict, validation strategies). Structure: weekly individual therapy + weekly multifamily skills group + phone coaching. Duration: typically 16–24 weeks. Evidence: RCTs show significant reductions in NSSI, suicidal ideation, and hospitalizations compared to treatment as usual.
Interpersonal Therapy for Adolescents (IPT-A)
IPT-A is a 12–16 session manualized treatment for adolescent depression (ages 12–18). It focuses on four interpersonal problem areas: grief, role disputes (conflicts with significant others), role transitions (life changes such as starting high school, parental divorce), and interpersonal deficits (loneliness, social isolation). IPT-A has the advantage of being effective for depression without requiring the cognitive sophistication needed for CBT. Evidence: Multiple RCTs show efficacy equal to CBT for adolescent depression; recommended by NICE and AACAP guidelines.
Parent-Child Interaction Therapy (PCIT)
PCIT is a dyadic behavioral parent training model for children ages 2–7 with disruptive behavior (ODD, aggression, tantrums). Two phases: Child-Directed Interaction (CDI) — the parent practices PRIDE skills (Praise, Reflect, Imitate, Describe, Enthusiasm) during child-led play, strengthening the parent-child relationship. Parent-Directed Interaction (PDI) — the parent gives effective commands and follows through with consistent consequences (time-out for noncompliance, labeled praise for compliance). A therapist coaches the parent in real-time via a one-way mirror and bug-in-the-ear device. Mastery criteria determine progression. Evidence: Over 150 studies; large effect sizes for disruptive behavior (d = 1.0–1.5); also effective for child physical abuse prevention.
Parent Management Training (PMT)
PMT programs (Incredible Years, Triple P, PMTO) teach parents to use positive reinforcement, consistent discipline, and effective communication to reduce coercive parent-child interactions and disruptive child behavior. Indicated for ODD and CD in children ages 3–12. Core components: positive attention and labeled praise, ignoring minor misbehavior, effective commands, logical consequences, token economies. Evidence: Robust across multiple programs; NNT 2–4 for clinically significant behavior improvement.
Trauma-Focused CBT (TF-CBT)
Described in detail in Section 15. Gold standard for pediatric PTSD, ages 3–18, with 15+ RCTs. The PRACTICE model involves parallel child and caregiver components.
Family Therapy
Family-Based Treatment (FBT) for eating disorders (Section 17). Functional Family Therapy (FFT) for adolescent behavioral problems and substance use (11–18 sessions targeting family communication patterns). Multisystemic Therapy (MST) for severe antisocial behavior (intensive, home-based, 3–5 months). All address the family system as a key lever for change. MST has the best evidence for reducing incarceration in delinquent youth.
Depression: CBT, IPT-A, behavioral activation. Anxiety: CBT with exposure (including SPACE). OCD: ERP (a form of CBT). PTSD: TF-CBT. Disruptive behavior (ages 2–7): PCIT. Disruptive behavior (ages 3–12): PMT. Severe antisocial behavior (adolescents): MST, FFT. NSSI/suicidality: DBT-A. Eating disorders: FBT (adolescent AN), CBT (BN). ADHD behavioral component: Behavioral parent training.
25 School-Based Interventions & IEP/504 Plans
Schools are the primary setting where child psychiatric conditions affect functioning. Understanding the educational framework for supporting students with psychiatric disabilities is essential for child psychiatrists, who frequently write letters supporting accommodations and participate in school meetings.
IDEA Disability Categories
The Individuals with Disabilities Education Act (IDEA) guarantees a Free Appropriate Public Education (FAPE) in the Least Restrictive Environment (LRE) for children ages 3–21 with qualifying disabilities. The 13 IDEA categories: (1) Autism, (2) Deaf-blindness, (3) Deafness, (4) Emotional disturbance, (5) Hearing impairment, (6) Intellectual disability, (7) Multiple disabilities, (8) Orthopedic impairment, (9) Other health impairment (includes ADHD), (10) Specific learning disability, (11) Speech or language impairment, (12) Traumatic brain injury, (13) Visual impairment. "Emotional disturbance" (ED) is the primary IDEA category for children with psychiatric conditions that adversely affect educational performance. "Other health impairment" (OHI) covers ADHD and medical conditions affecting alertness/vitality.
IEP vs 504 Plan
| Feature | IEP (Individualized Education Program) | 504 Plan |
|---|---|---|
| Governing law | IDEA (federal education law) | Section 504 of the Rehabilitation Act (civil rights law) |
| Eligibility | Must qualify under 1 of 13 IDEA categories AND require specially designed instruction | Must have a disability that substantially limits a major life activity (broader definition) |
| Evaluation | Full psychoeducational evaluation required; school-funded | Evaluation required but less formal; often based on existing records |
| Services | Specially designed instruction, related services (speech, OT, counseling), goals/objectives, progress monitoring | Accommodations and modifications only (no specialized instruction) |
| Legal protections | Annual review, triennial reevaluation, prior written notice, due process rights, stay-put provision | Periodic review (no mandated timeline), fewer procedural protections |
| Common examples | Child with ASD receiving social skills instruction; child with SLD receiving specialized reading intervention | Child with ADHD receiving extended test time, preferential seating; child with anxiety receiving access to school counselor |
Common Accommodations for Psychiatric Conditions
ADHD: Preferential seating (front of class, away from distractions), extended time on tests (typically 1.5x), breaks during work periods, reduced homework assignments, behavioral chart with daily teacher feedback, fidget tools, chunking of assignments. Anxiety: Pre-arranged signal to leave class for counselor visit, extended time, testing in a separate/quiet location, advance notice of schedule changes, permission to call parent during panic episodes, reduced oral presentation requirements. Depression: Flexible attendance policy, late assignment completion without penalty, access to school counselor, reduced workload during acute episodes, homebound instruction if unable to attend. ASD: Visual schedule, social skills group, sensory breaks, aide support, social narratives for transitions, modified PE requirements.
School Refusal
School refusal affects 1–5% of school-age children and is typically driven by anxiety (separation anxiety, social anxiety, specific phobia, GAD) or depression. It is distinct from truancy (non-anxiety-motivated absence, often associated with conduct disorder, delinquency). Management: rapid return to school is the primary goal; graduated exposure with planned check-ins, morning routine restructuring, parent coaching to avoid accommodation, school-based support (counselor meeting at arrival, safe space), CBT targeting underlying anxiety, SSRI if severe. Homebound instruction should be avoided if possible as it reinforces avoidance; if medically necessary, provide a concrete time-limited plan with school reintegration goals.
Telepsychiatry & Digital Interventions
Telepsychiatry has become an essential modality in child and adolescent psychiatry, particularly for underserved rural communities. Evidence shows equivalent diagnostic accuracy and treatment outcomes to in-person care for ADHD, depression, anxiety, and disruptive behaviors. Digital CBT platforms (e.g., MoodGYM, BRAVE Online) have RCT evidence for mild-moderate anxiety and depression in adolescents. Considerations specific to telepsychiatry with youth: ensure private space for the child (not in shared rooms), verify emergency contacts and local crisis resources, perform safety assessments at every session, and consider whether the child's developmental level allows meaningful engagement via video. Hybrid models (some in-person, some telehealth) may be optimal for complex cases requiring observation of motor behavior, tics, or parent-child interaction.
Crisis Intervention in Schools
School-based crisis intervention follows the PREPaRE model (developed by the National Association of School Psychologists): Prevent/prepare for crises, Reaffirm physical health, Evaluate psychological trauma risk, Provide interventions (triage, individual crisis intervention, classroom-based), Respond to psychological needs (psychoeducation, caregiver training), and Examine the effectiveness of the response. Tier 1 (universal) interventions include classroom psychoeducation about normal stress reactions. Tier 2 (targeted) includes small group crisis intervention for exposed students. Tier 3 (intensive) involves individual trauma-focused therapy referral for students with persistent symptoms.
26 Gender Diversity & Gender-Affirming Care
Gender dysphoria in children and adolescents refers to marked incongruence between experienced/expressed gender and assigned gender, associated with clinically significant distress or impairment. Gender diverse youth experience higher rates of depression (2–3x), anxiety (2–3x), suicidal ideation (5–6x), and suicide attempts (4x) compared to cisgender peers. Social support, family acceptance, and access to gender-affirming care are strongly associated with reduced psychiatric morbidity.
DSM-5-TR: Gender Dysphoria in Children
Requires marked incongruence between experienced/expressed gender and assigned gender lasting at least 6 months, manifested by at least 6 of the following: (1) Strong desire to be of the other gender or insistence that one is the other gender. (2) Strong preference for cross-dressing. (3) Strong preference for cross-gender roles in play. (4) Strong preference for toys/activities stereotypically used by other gender. (5) Strong preference for playmates of the other gender. (6) Strong rejection of typically gender-associated toys/activities. (7) Strong dislike of one's own sexual anatomy. (8) Strong desire for primary/secondary sex characteristics matching experienced gender. Must be associated with clinically significant distress or impairment. For adolescents/adults: at least 2 of 6 criteria for at least 6 months.
Tanner Staging Relevance
Medical interventions are guided by pubertal development as measured by Tanner staging. Puberty blockers (GnRH agonists) are initiated at Tanner stage 2 (breast budding in natal females, testicular volume 4+ mL in natal males), which represents the onset of puberty. This is the earliest that medical intervention is considered. Tanner stages: Stage 1 (prepubertal), Stage 2 (early puberty — breast buds/early testicular enlargement), Stage 3 (mid-puberty), Stage 4 (late puberty), Stage 5 (adult).
Medical Interventions
Puberty blockers (GnRH agonists) — leuprolide acetate (Lupron) or histrelin (Supprelin) — suppress the hypothalamic-pituitary-gonadal axis, pausing puberty. This is a fully reversible intervention that provides time for continued psychosocial assessment and reduces the distress of developing unwanted secondary sex characteristics. Effects reverse upon discontinuation. Side effects include decreased bone mineral density (monitored with DXA scans), potential effects on growth, and rare injection-site reactions. Gender-affirming hormones (estrogen or testosterone) are typically initiated at age 16 (some guidelines allow earlier, from age 14); effects are partially reversible (e.g., breast development, voice deepening are not reversible). Surgical interventions are generally deferred until adulthood (age 18+) for genital surgery; chest masculinization may be considered earlier in select cases.
Mental Health Assessment Role
The mental health professional's role is to: (1) Assess gender identity development comprehensively (not to serve as a "gatekeeper" but as a supportive evaluator). (2) Assess and treat co-occurring psychiatric conditions. (3) Evaluate the adolescent's capacity for informed consent/assent. (4) Support family adjustment and acceptance. (5) Provide ongoing mental health support throughout transition. Family acceptance is the single strongest protective factor against psychiatric morbidity in transgender youth: accepted transgender youth have depression and suicidality rates comparable to cisgender peers.
27 Legal & Ethical Issues in Child Psychiatry
Child and adolescent psychiatry operates at the intersection of clinical care, family dynamics, and legal systems. The child psychiatrist must navigate complex ethical terrain involving children who cannot legally consent, families who may disagree with treatment, mandatory reporting obligations, and the tension between adolescent autonomy and parental authority.
Informed Consent & Assent
Informed consent for treatment of a minor is provided by the parent or legal guardian. The consent must include: disclosure of the nature of the proposed treatment, risks and benefits, alternatives, and the right to refuse. Assent is the child's affirmative agreement to participate in treatment, appropriate to their developmental level. AACAP recommends seeking assent from children aged 7+ and treating dissent seriously even if parents consent. Exceptions where minors may consent for their own care (varies by state): emergency treatment, emancipated minors, mature minor doctrine (court determination of capacity), substance abuse treatment, STI/HIV testing and treatment, contraception, prenatal care, and in some states, mental health treatment for adolescents aged 14–16+.
Confidentiality with Minors
Adolescents require a degree of confidentiality to engage honestly in treatment. Best practice: at the first session, discuss with both the parent and teen that the therapist will keep the teen's disclosures private UNLESS there is a safety concern (suicidality, homicidality, abuse, significant substance use endangering health). The clinician provides parents with general updates on progress and themes without sharing session content verbatim. HIPAA provides additional protections: in states where minors can consent for their own mental health care, the minor controls access to those records.
Mandatory Reporting
All 50 states and DC have mandatory reporting laws requiring designated professionals (including all healthcare providers) to report suspected child abuse and neglect to child protective services (CPS). Key points: the threshold is reasonable suspicion (not certainty); reporters are protected from liability for good-faith reports; failure to report is a criminal offense in most states; the report is made to the CPS hotline in the state where the child resides; the clinician is not responsible for investigating — only for reporting. Reportable categories: physical abuse, sexual abuse, emotional/psychological abuse, neglect (physical, emotional, medical, educational), and exposure to domestic violence (in some states).
Custody Evaluations
Child psychiatrists may be asked to conduct custody evaluations or provide expert testimony in custody disputes. AACAP Practice Parameters: the evaluator should be impartial and appointed by the court (not retained by one party), interview both parents and the child (separately), obtain collateral information (school, pediatrician, other treaters), use validated instruments when appropriate, and base recommendations on the best interests of the child standard. Factors assessed: each parent's capacity to meet the child's developmental needs, the child's relationships with each parent, stability and safety of each home environment, the child's preference (given appropriate weight by age), and any history of abuse or domestic violence. A treating clinician should NOT serve as the custody evaluator (dual role conflict).
Involuntary Treatment
Parents generally have the authority to consent to psychiatric hospitalization for their minor child. However, Parham v. J.R. (1979, US Supreme Court) established that an independent review by a "neutral factfinder" (physician, not necessarily a judge) is required before a child can be committed to a psychiatric facility at the parent's request, to protect the child's liberty interest. For adolescents who refuse treatment but whose parents consent: in most states, the adolescent can be treated over their objection with parental consent if a clinician agrees treatment is necessary. In some states, adolescents aged 14–16+ can refuse psychiatric hospitalization. Emergency detention (involuntary hold) criteria for minors generally mirror adult criteria: danger to self, danger to others, or gravely disabled.
Forensic Considerations
Juvenile competency to stand trial: Minors can be evaluated for competency using instruments such as the Juvenile Adjudicative Competence Interview (JACI). Developmental immaturity (not just mental illness) can render a juvenile incompetent. Transfer/waiver to adult court: Forensic evaluators assess amenability to treatment, sophistication/maturity, risk of recidivism, and severity of offense. Miranda rights comprehension: Juveniles under 15 have poor understanding of Miranda rights; the Grisso instruments assess comprehension. Sex offense evaluations: Juvenile sex offenders differ from adult offenders — most do not become adult sex offenders; risk assessment tools (J-SOAP-II, ERASOR) assess dynamic and static risk factors; treatment focuses on CBT-based programs rather than punitive approaches.
Beneficence: Act in the child's best interest, which may differ from the parent's wishes. Autonomy: Respect the adolescent's developing autonomy proportional to their maturity. Non-maleficence: Minimize harm (consider medication side effects, labeling effects, family disruption from hospitalization). Justice: Address disparities in access to mental health care (racial/ethnic minorities, rural populations, uninsured). Fidelity: Maintain the therapeutic alliance with both the child and the family, even when they conflict. Veracity: Be honest with children at developmentally appropriate levels about diagnosis, treatment, and prognosis.
28 Rating Scales & Screening Tools
A compilation of the most commonly used assessment instruments in child and adolescent psychiatry, with age ranges, administration details, scoring guidelines, and clinical interpretation. Selection of instruments should be guided by the clinical question, the child's age and developmental level, and practical considerations (time, cost, availability). No single instrument replaces clinical judgment; rating scales are screening and monitoring tools, not diagnostic instruments. Multi-informant assessment (obtaining data from child, parent, AND teacher) is essential because agreement between informants is typically low (r = 0.2–0.3), and each informant provides unique, valid information about the child's functioning in different settings.
| Instrument | Domain | Ages | Format | Scoring & Interpretation |
|---|---|---|---|---|
| Vanderbilt ADHD Rating Scale | ADHD, ODD, CD, anxiety, depression | 6–12 | Parent and teacher forms; 43–55 items | Symptom items rated 0–3; 6+ items scored 2–3 in ADHD domain = positive screen; performance items 1–5 identify impairment |
| Conners Rating Scales (Conners-4) | ADHD, executive function, emotional dysregulation | 6–18 | Parent, teacher, self-report; multiple lengths | T-scores (mean 50, SD 10); elevated ≥65; very elevated ≥70; includes validity scales |
| SCARED | Anxiety disorders (panic, GAD, SAD, social, school avoidance) | 8–18 | 41-item child and parent report | Total ≥25 = anxiety disorder; subscale cutoffs: panic ≥7, GAD ≥9, SAD ≥5, social ≥8, school ≥3 |
| PHQ-A (Adolescent) | Depression | 11–17 | 9-item self-report (modified PHQ-9) | 0–4 minimal, 5–9 mild, 10–14 moderate, 15–19 mod-severe, 20–27 severe; item 9 screens suicidality |
| CDRS-R | Depression severity | 6–12 (used up to 18) | 17-item clinician-rated | Total 17–113; ≥40 clinically significant depression; used as outcome measure in treatment trials (e.g., TADS) |
| CY-BOCS | OCD severity | 6–17 | 10-item clinician-rated + symptom checklist | 0–40; 0–7 subclinical, 8–15 mild, 16–23 moderate, 24–31 severe, 32–40 extreme; 25–35% reduction = response |
| M-CHAT-R/F | Autism screening | 16–30 months | 20-item parent report + follow-up interview | 0–2 low risk, 3–7 medium risk (do follow-up), 8–20 high risk (refer). Follow-up score 2+ = refer for evaluation |
| ADOS-2 | ASD diagnostic assessment | 12 months – adult | Semi-structured clinician observation; 5 modules by language level | Algorithm scores classified as "autism," "autism spectrum," or "non-spectrum"; combined with ADI-R for gold standard |
| YGTSS | Tic severity | 6–17 | Clinician-rated; motor and vocal tics separately | Total Tic Score 0–50 + Impairment 0–50 = Global 0–100; ≥20 Total Tic Score = moderate |
| CRAFFT | Substance use screening | 12–21 | 6-item self-report (after Part A frequency screen) | Score ≥2 = positive screen for substance use disorder (sensitivity 76%, specificity 94%) |
| ASQ (Ask Suicide-Screening Questions) | Suicide risk | 10–24 | 4 yes/no questions + 1 acuity question | Any "yes" to items 1–4 = positive screen; "yes" to item 5 = acute positive (immediate safety intervention) |
| C-SSRS | Suicidal ideation and behavior | All ages (adapted versions) | Clinician-administered; structured interview | Classifies ideation (1–5 intensity) and behavior (actual attempt, interrupted, aborted, preparatory, NSSI) |
| CBCL (Child Behavior Checklist) | Broadband behavioral/emotional problems | 1.5–18 | 113-item parent report; also TRF (teacher) and YSR (youth 11–18) | T-scores: <65 normal, 65–69 borderline, ≥70 clinical; yields internalizing, externalizing, total problem, and DSM-oriented subscales |
| SDQ (Strengths and Difficulties Questionnaire) | Brief behavioral/emotional screen | 4–17 | 25-item parent, teacher, or self-report | 5 subscales (0–10 each): emotional, conduct, hyperactivity, peer, prosocial. Total difficulties 0–40; abnormal ≥17 (parent), ≥16 (teacher), ≥20 (self) |
| ACE Questionnaire | Adverse childhood experiences | Adult retrospective (used clinically with adolescents) | 10-item yes/no | Score 0–10; each point increases risk of negative health outcomes; ≥4 = high risk for chronic disease, mental illness, substance abuse |
| Pediatric Symptom Checklist (PSC-17) | Broadband psychosocial screening | 4–18 | 17-item parent report (PSC-35 also available) | Internalizing ≥5, externalizing ≥7, attention ≥7, or total ≥15 = positive screen; free; used in pediatric primary care |
| NICHQ Vanderbilt Follow-Up | ADHD treatment monitoring | 6–12 | Parent and teacher follow-up forms | Tracks symptom severity and side effects at each visit; essential for titration and monitoring |
| SNAP-IV | ADHD and ODD symptom severity | 6–18 | 26-item teacher and parent rating scale | Items rated 0–3; average per-item score >1.2 (teacher) or >1.8 (parent) for inattention/hyperactivity subscales = clinically significant |
| Aberrant Behavior Checklist (ABC) | Behavioral problems in developmental disabilities | 6–adult | 58-item caregiver-completed | 5 subscales: irritability, lethargy/social withdrawal, stereotypy, hyperactivity, inappropriate speech; primary outcome measure in ASD medication trials (RUPP) |
ADHD evaluation: Vanderbilt (free, broad) or Conners-4 (comprehensive, fee). Anxiety screening: SCARED (free). Depression screening: PHQ-A (free, quick). OCD severity: CY-BOCS (clinician-rated gold standard). ASD diagnostic: ADOS-2 + ADI-R (gold standard, specialist-administered). ASD screening: M-CHAT-R/F (16–30 months). Broadband screening: CBCL/TRF/YSR (comprehensive, fee) or SDQ/PSC-17 (free, brief). Suicide screening: ASQ (quick, free) then C-SSRS (comprehensive) if positive. Substance use: CRAFFT (free, validated for 12–21). Trauma: UCLA PTSD-RI or CPSS-5. Tics: YGTSS.
29 Medications Master Table
Comprehensive pediatric psychopharmacology reference organized by drug class. All doses are for children and adolescents unless otherwise specified. Always verify current prescribing information. General principles of pediatric psychopharmacology: (1) Start low, go slow, but go — initiate at the lowest available dose and titrate gradually to therapeutic effect. (2) Use monotherapy when possible; avoid polypharmacy unless clearly indicated. (3) Set clear target symptoms and use rating scales to monitor response. (4) Reassess the need for medication periodically; plan for discontinuation trials when appropriate. (5) Always combine medication with evidence-based psychotherapy and environmental interventions. (6) Informed consent from parents and assent from the child must be obtained, with discussion of off-label use, expected benefits, risks, and monitoring requirements.
Stimulants (Schedule II)
| Generic (Brand) | Class | Ages | Starting Dose | Target/Max | Key Pearls |
|---|---|---|---|---|---|
| Methylphenidate IR (Ritalin) | MPH | 6+ | 5 mg BID | 20 mg TID / 60 mg/day | Shortest acting; flexible dosing; tid dosing limits adherence |
| Methylphenidate ER (Concerta) | MPH | 6+ | 18 mg QAM | 54 mg (child) / 72 mg (adolescent) | OROS — do NOT crush; ghost tablet in stool (normal) |
| Dexmethylphenidate ER (Focalin XR) | d-MPH | 6+ | 5 mg QAM | 30 mg (child) / 40 mg (adult) | Half dose of racemic MPH; can sprinkle beads |
| Mixed amphetamine salts IR (Adderall) | AMPH | 3+ | 5 mg QD-BID | 40 mg/day | 75:25 d-amp:l-amp; rapid onset |
| Mixed amphetamine salts XR (Adderall XR) | AMPH | 6+ | 5–10 mg QAM | 30 mg (child) / 40 mg | 50/50 IR:DR beads; can sprinkle |
| Lisdexamfetamine (Vyvanse) | AMPH prodrug | 6+ | 20–30 mg QAM | 70 mg/day | Prodrug reduces abuse potential; also for BED (adults); longest acting stimulant |
Non-Stimulant ADHD Medications
| Generic (Brand) | Mechanism | Ages | Starting Dose | Target/Max | Key Pearls |
|---|---|---|---|---|---|
| Atomoxetine (Strattera) | NRI | 6+ | 0.5 mg/kg/day | 1.2 mg/kg/day; max 100 mg | Non-controlled; 4–6 wk to full effect; black box suicidality; rare hepatotoxicity; CYP2D6 poor metabolizers need lower dose |
| Guanfacine ER (Intuniv) | Alpha-2A agonist | 6–17 | 1 mg QHS | 4 mg (6–12); 7 mg (13–17) | Helps tics, aggression, insomnia; taper to stop; somnolence, hypotension |
| Clonidine ER (Kapvay) | Alpha-2 agonist | 6–17 | 0.1 mg QHS | 0.4 mg/day (divided BID) | More sedating; rebound hypertension; useful for sleep |
| Viloxazine ER (Qelbree) | NRI + 5-HT modulator | 6–17 | 100 mg QAM (6–11); 200 mg (12–17) | 200 mg (6–11); 400 mg (12–17) | Non-controlled; FDA 2021; CYP1A2 inhibitor; black box suicidality |
SSRIs
| Generic (Brand) | FDA Pediatric Indications | Starting Dose | Target/Max | Key Pearls |
|---|---|---|---|---|
| Fluoxetine (Prozac) | MDD 8+; OCD 7+ | 10 mg/day | 20–60 mg (MDD); 20–80 mg (OCD) | Longest half-life (1–3 days parent, 4–16 days norfluoxetine); least discontinuation syndrome; CYP2D6 inhibitor |
| Escitalopram (Lexapro) | MDD 12+ | 5–10 mg/day | 10–20 mg | S-enantiomer of citalopram; clean pharmacokinetics; well-tolerated |
| Sertraline (Zoloft) | OCD 6+ | 25 mg/day | 50–200 mg | Widely used off-label for anxiety/depression; GI side effects; highest SRI dose may be needed for OCD |
| Fluvoxamine (Luvox) | OCD 8+ | 25 mg QHS | 100–200 mg (<12); 100–300 mg (12–17) | Potent CYP1A2 and CYP2C19 inhibitor; many drug interactions; divide doses >100 mg |
| Citalopram (Celexa) | None (used off-label) | 10 mg/day | 20–40 mg | QTc prolongation risk at higher doses; FDA max 40 mg in adults; avoid with congenital long QT |
Atypical Antipsychotics
| Generic (Brand) | Pediatric Indications | Starting Dose | Typical Target | Key Concerns |
|---|---|---|---|---|
| Risperidone (Risperdal) | ASD irritability 5–16; bipolar mania 10–17; schizophrenia 13–17 | 0.25–0.5 mg QHS | 0.5–3 mg (ASD); 1–6 mg (bipolar/schizo) | Highest prolactin elevation; gynecomastia; weight gain; EPS |
| Aripiprazole (Abilify) | ASD irritability 6–17; bipolar mania 10–17; schizophrenia 13–17; Tourette 6–18 | 2 mg/day | 5–15 mg | D2 partial agonist; least metabolic; akathisia most common SE; may lower prolactin |
| Quetiapine (Seroquel) | Bipolar mania 10–17; schizophrenia 13–17 | 25–50 mg QHS | 400–600 mg (schizo/bipolar) | Very sedating; significant weight gain; metabolic effects; used off-label for insomnia/anxiety (not recommended) |
| Olanzapine (Zyprexa) | Bipolar mania 13–17; schizophrenia 13–17 | 2.5–5 mg QHS | 10–20 mg | Highest weight gain and metabolic risk; reserve for treatment-resistant; very effective |
| Lurasidone (Latuda) | Bipolar depression 10–17; schizophrenia 13–17 | 20 mg with food (350+ kcal) | 40–80 mg | Must take with food (absorption); favorable metabolic profile; EPS and akathisia; only atypical FDA-approved for pediatric bipolar depression |
Mood Stabilizers & Other Agents
| Generic (Brand) | Indication | Dose/Level | Key Monitoring |
|---|---|---|---|
| Lithium (Lithobid, Eskalith) | Bipolar I (age 7+) | Start 300 mg BID; level 0.6–1.2 mEq/L | Lithium level, BUN/Cr, TSH, Ca q3–6 mo; weight; EKG if indicated |
| Divalproex (Depakote) | Bipolar (off-label in peds) | 10–15 mg/kg/day; level 50–125 mcg/mL | LFTs, CBC/platelets, VPA level; pregnancy test; weight; lipase if GI symptoms |
| Lamotrigine (Lamictal) | Bipolar maintenance (off-label in peds) | Very slow titration: 25 mg/day × 2 wk, increase by 25 mg q2wk; target 100–200 mg | Rash monitoring (SJS risk 0.3–1% in children — higher than adults; risk increases with rapid titration and VPA co-administration) |
| Clomipramine (Anafranil) | OCD (age 10+) | 25 mg QHS, titrate to 3 mg/kg/day or 200 mg max | EKG (QTc); seizure risk at doses >200 mg; anticholinergic effects; TCA overdose risk |
| Melatonin | Sleep onset delay (OTC) | 0.5–5 mg 30–60 min before bed | None required; assess efficacy and side effects |
30 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AACAP | American Academy of Child and Adolescent Psychiatry |
| AAP | American Academy of Pediatrics |
| ABA | Applied Behavior Analysis |
| ACE | Adverse Childhood Experience |
| ADHD | Attention-Deficit/Hyperactivity Disorder |
| ADI-R | Autism Diagnostic Interview — Revised |
| ADOS-2 | Autism Diagnostic Observation Schedule, 2nd Edition |
| AIMS | Abnormal Involuntary Movement Scale |
| AN | Anorexia Nervosa |
| ARFID | Avoidant/Restrictive Food Intake Disorder |
| ASD | Autism Spectrum Disorder |
| ASQ | Ask Suicide-Screening Questions |
| ASQ-3 | Ages and Stages Questionnaire, 3rd Edition |
| BN | Bulimia Nervosa |
| BP | Blood Pressure |
| CAMS | Child/Adolescent Anxiety Multimodal Study |
| CBCL | Child Behavior Checklist |
| CBIT | Comprehensive Behavioral Intervention for Tics |
| CBT | Cognitive Behavioral Therapy |
| CD | Conduct Disorder |
| CDC | Centers for Disease Control and Prevention |
| CDRS-R | Children's Depression Rating Scale — Revised |
| CMA | Chromosomal Microarray Analysis |
| CNV | Copy Number Variant |
| CRAFFT | Car, Relax, Alone, Forget, Friends, Trouble (substance screening) |
| CPS | Child Protective Services |
| C-SSRS | Columbia Suicide Severity Rating Scale |
| CU | Callous-Unemotional (traits) |
| CY-BOCS | Children's Yale-Brown Obsessive Compulsive Scale |
| DAT | Dopamine Transporter |
| DBT-A | Dialectical Behavior Therapy for Adolescents |
| DCD | Developmental Coordination Disorder |
| DISC | Diagnostic Interview Schedule for Children |
| dlPFC | Dorsolateral Prefrontal Cortex |
| DMDD | Disruptive Mood Dysregulation Disorder |
| DSED | Disinhibited Social Engagement Disorder |
| DSM-5-TR | Diagnostic and Statistical Manual of Mental Disorders, 5th Ed., Text Revision |
| EEG | Electroencephalogram |
| EIBI | Early Intensive Behavioral Intervention |
| EKG/ECG | Electrocardiogram |
| EPS | Extrapyramidal Symptoms |
| ERP | Exposure and Response Prevention |
| FAPE | Free Appropriate Public Education |
| FBT | Family-Based Treatment (Maudsley method) |
| FFT | Functional Family Therapy |
| GAD | Generalized Anxiety Disorder |
| GDD | Global Developmental Delay |
| GnRH | Gonadotropin-Releasing Hormone |
| HPA | Hypothalamic-Pituitary-Adrenal (axis) |
| HR | Heart Rate |
| ID | Intellectual Disability |
| IDEA | Individuals with Disabilities Education Act |
| IEP | Individualized Education Program |
| IPT-A | Interpersonal Therapy for Adolescents |
| K-SADS-PL | Schedule for Affective Disorders and Schizophrenia for School-Age Children — Present and Lifetime |
| LRE | Least Restrictive Environment |
| M-CHAT-R/F | Modified Checklist for Autism in Toddlers, Revised with Follow-Up |
| MDD | Major Depressive Disorder |
| MPH | Methylphenidate |
| MST | Multisystemic Therapy |
| MTA | Multimodal Treatment Study of ADHD |
| NET | Norepinephrine Transporter |
| NNH | Number Needed to Harm |
| NNT | Number Needed to Treat |
| NRI | Norepinephrine Reuptake Inhibitor |
| NSSI | Non-Suicidal Self-Injury |
| OCD | Obsessive-Compulsive Disorder |
| ODD | Oppositional Defiant Disorder |
| OFC | Orbitofrontal Cortex |
| OHI | Other Health Impairment (IDEA category) |
| PANDAS | Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections |
| PANS | Pediatric Acute-onset Neuropsychiatric Syndrome |
| PCIT | Parent-Child Interaction Therapy |
| PFC | Prefrontal Cortex |
| PHQ-A | Patient Health Questionnaire for Adolescents |
| PMT | Parent Management Training |
| PTSD | Post-Traumatic Stress Disorder |
| QTc | Corrected QT Interval |
| RAD | Reactive Attachment Disorder |
| RCT | Randomized Controlled Trial |
| RRB | Restricted, Repetitive Behaviors |
| SAD | Separation Anxiety Disorder |
| SCARED | Screen for Child Anxiety Related Disorders |
| SCD | Social (Pragmatic) Communication Disorder |
| SDQ | Strengths and Difficulties Questionnaire |
| SERT | Serotonin Transporter |
| SJS | Stevens-Johnson Syndrome |
| SLD | Specific Learning Disorder |
| SPACE | Supportive Parenting for Anxious Childhood Emotions |
| SRI | Serotonin Reuptake Inhibitor |
| SSRI | Selective Serotonin Reuptake Inhibitor |
| TADS | Treatment for Adolescents with Depression Study |
| TD | Tardive Dyskinesia |
| TF-CBT | Trauma-Focused Cognitive Behavioral Therapy |
| TRF | Teacher Report Form |
| TS | Tourette Syndrome |
| vmPFC | Ventromedial Prefrontal Cortex |
| WPATH | World Professional Association for Transgender Health |
| WES | Whole Exome Sequencing |
| YGTSS | Yale Global Tic Severity Scale |
| YSR | Youth Self-Report |