Psychiatry
Mood disorders, psychotic disorders, anxiety, trauma, psychopharmacology, neuromodulation, risk assessment, and every classification system, diagnostic threshold, and management algorithm across the full scope of adult psychiatry.
01 Neuroanatomy & Neurophysiology for Psychiatry
Psychiatric illness arises from dysfunction in specific neural circuits rather than single brain regions. Understanding the anatomy and neurochemistry of these circuits is essential for rational psychopharmacology and for interpreting neuroimaging findings in clinical practice.
The Limbic System
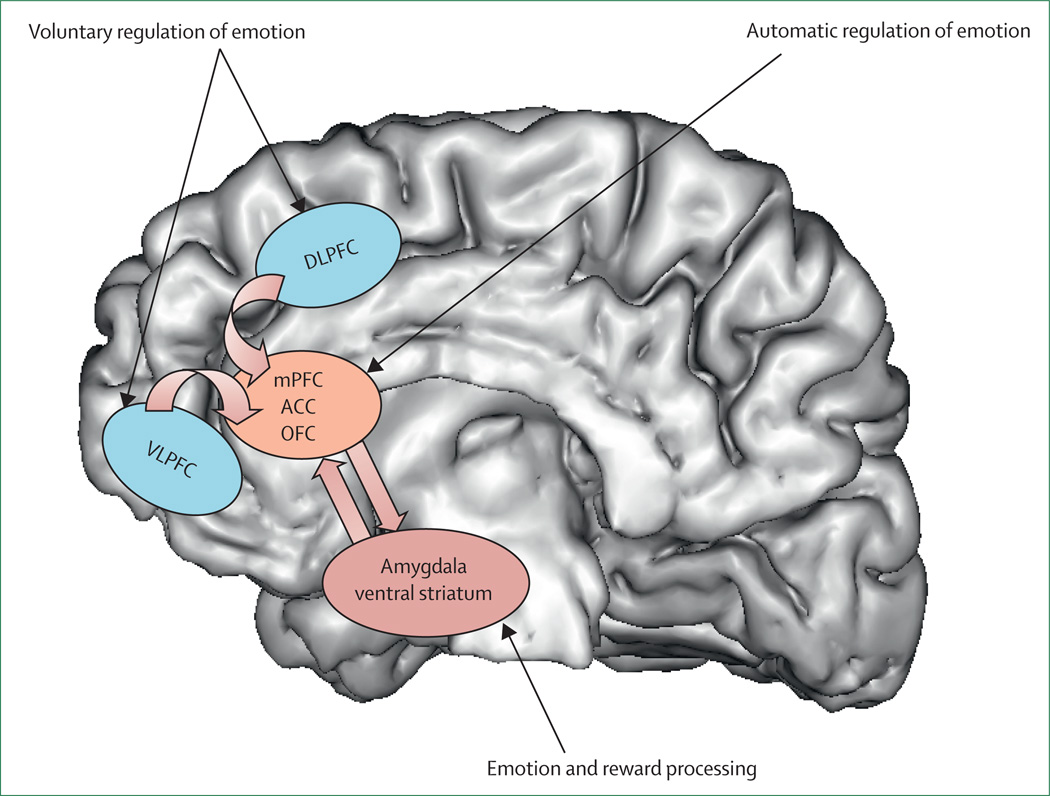
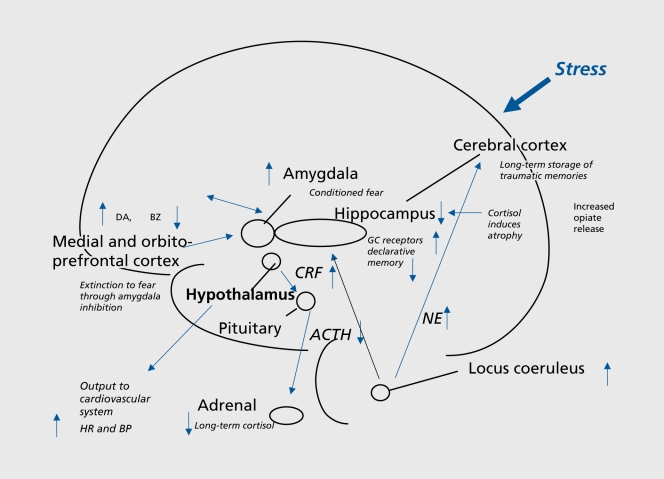
The amygdala (basolateral and central nuclei) is the hub for fear conditioning, threat detection, and emotional memory. Hyperactivity of the amygdala is consistently found in PTSD and social anxiety disorder on fMRI. The hippocampus (CA1–CA4 subfields, dentate gyrus) is critical for declarative memory consolidation and contextual fear learning; hippocampal volume reduction of 5–12% is a replicated finding in chronic depression and PTSD. The cingulate cortex divides functionally: the anterior cingulate cortex (ACC) subgenual portion (Brodmann area 25) is a key target for deep brain stimulation in treatment-resistant depression, while the dorsal ACC mediates error monitoring and conflict detection (implicated in OCD). The hypothalamus orchestrates the HPA axis via corticotropin-releasing hormone (CRH) release to the anterior pituitary, driving cortisol secretion—chronically elevated in melancholic depression and blunted in atypical depression and PTSD.
Prefrontal Cortex
The dorsolateral prefrontal cortex (dlPFC) (Brodmann areas 9, 46) mediates working memory, executive function, and cognitive flexibility. Hypoactivation of the left dlPFC is a consistent finding in depression—the rationale for targeting this region with high-frequency rTMS. The ventromedial PFC (vmPFC) integrates emotional valence with decision-making and is critical for fear extinction; reduced vmPFC activity is observed in PTSD. The orbitofrontal cortex (OFC) (areas 11, 47) evaluates reward value and mediates response inhibition; OFC hyperactivity in the caudate-thalamo-cortical loop is the leading neuroanatomical model for OCD.
Basal Ganglia & Thalamus
The caudate nucleus and putamen (together forming the striatum) receive dopaminergic input from the midbrain. The ventral striatum (nucleus accumbens) is the core reward-processing structure; blunted ventral striatal response to reward is characteristic of anhedonia in depression. The thalamus serves as the relay station for cortical-subcortical circuits; thalamic overactivity is seen in the hyperarousal of PTSD, while thalamic underactivity characterizes the negative symptoms of schizophrenia.
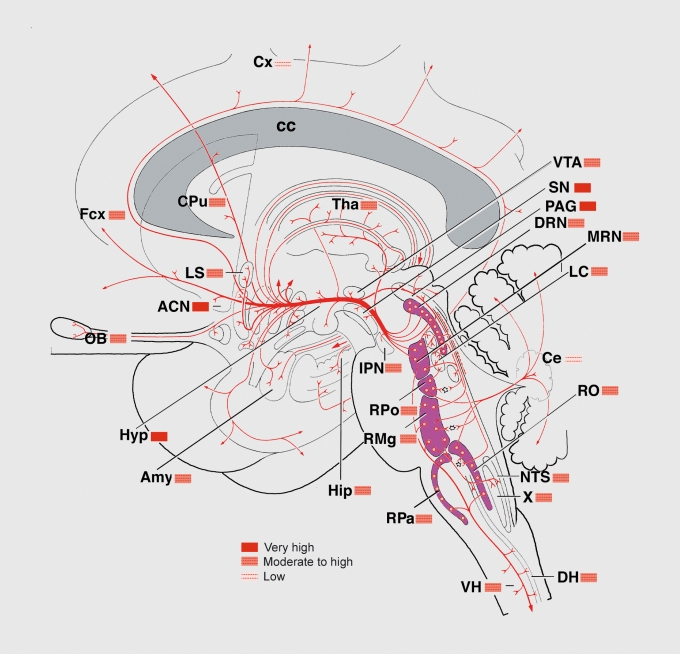
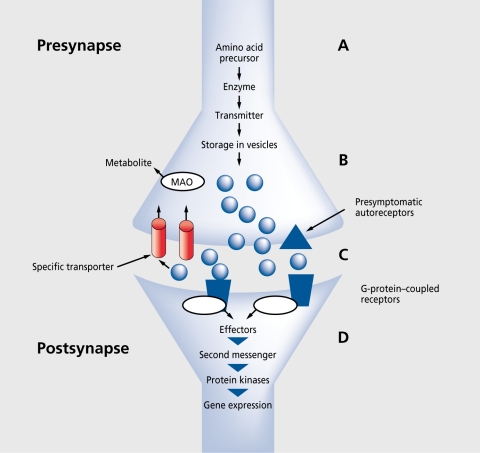
Major Neurotransmitter Systems
Origin: Raphe nuclei (dorsal and median) in the brainstem pons/midbrain. Projections: Widespread cortical, limbic, hypothalamic, and spinal cord projections. Key receptors: 5-HT1A (autoreceptor on raphe neurons; postsynaptic in hippocampus—anxiolytic when activated; buspirone is a partial agonist), 5-HT2A (cortical—mediates hallucinogenic effects; blocked by atypical antipsychotics), 5-HT2C (appetite/weight regulation; blocked by olanzapine and mirtazapine causing weight gain), 5-HT3 (area postrema—nausea; antagonists like ondansetron are antiemetics). Clinical relevance: SSRIs block SERT (serotonin transporter), increasing synaptic 5-HT. The 3–4 week delay in antidepressant response correlates with desensitization of 5-HT1A autoreceptors, which initially limit serotonin release.
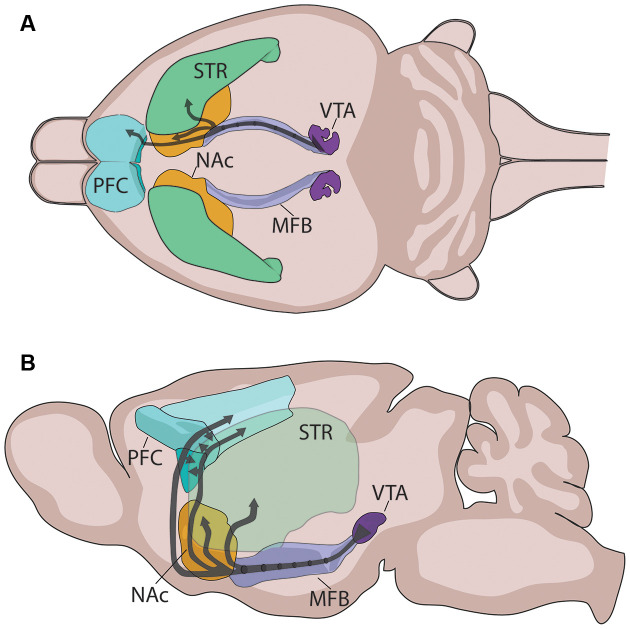
Four major pathways: (1) Mesolimbic — VTA to nucleus accumbens; mediates reward, motivation, and positive symptoms of psychosis (hyperactivity). (2) Mesocortical — VTA to prefrontal cortex; mediates cognition and executive function; hypoactivity produces negative/cognitive symptoms of schizophrenia. (3) Nigrostriatal — substantia nigra pars compacta to dorsal striatum; motor control; blockade causes EPS and tardive dyskinesia. (4) Tuberoinfundibular — hypothalamic arcuate nucleus to anterior pituitary; tonically inhibits prolactin release; D2 blockade causes hyperprolactinemia. Key receptors: D1-like (D1, D5)—excitatory, Gs-coupled; D2-like (D2, D3, D4)—inhibitory, Gi-coupled. Antipsychotic efficacy correlates with D2 receptor occupancy: 60–80% occupancy = therapeutic effect; >80% occupancy = EPS.
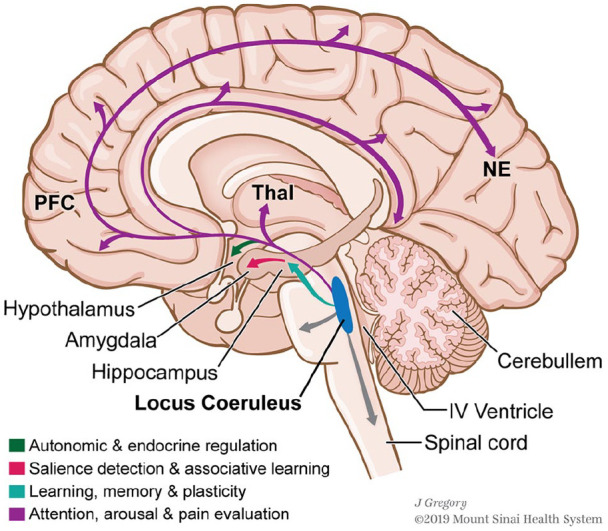
Origin: Locus coeruleus (LC) in the dorsal pons—contains ~50% of all NE neurons in the brain. Projections: Diffuse cortical, limbic, cerebellar, and spinal cord projections. Function: Arousal, attention, vigilance, stress response. LC hyperactivity drives hyperarousal, insomnia, and exaggerated startle in PTSD and panic disorder. Receptors: Alpha-1 (postsynaptic, excitatory), alpha-2 (presynaptic autoreceptor—inhibitory; clonidine and guanfacine are agonists used for hyperarousal in PTSD), beta-1/2 (postsynaptic; propranolol blocks peripheral and central beta receptors—used for performance anxiety). SNRIs (venlafaxine, duloxetine) and TCAs block NE reuptake via NET (norepinephrine transporter).
GABA (gamma-aminobutyric acid) is the primary inhibitory neurotransmitter. GABA-A receptors are ligand-gated chloride channels; benzodiazepines bind the alpha subunit allosteric site and increase chloride channel opening frequency (vs. barbiturates which increase duration). GABA-B receptors are G-protein coupled (baclofen is an agonist). Reduced GABAergic tone in the amygdala and PFC is implicated in anxiety disorders. Glutamate is the primary excitatory neurotransmitter. NMDA receptors (require glycine co-agonist, voltage-dependent Mg2+ block) are central to synaptic plasticity and long-term potentiation. The NMDA receptor hypofunction hypothesis of schizophrenia posits that reduced NMDA signaling on cortical interneurons produces disinhibition mimicking both positive and negative symptoms—supported by the psychotomimetic effects of PCP and ketamine. Ketamine's rapid antidepressant effect (hours vs. weeks for SSRIs) involves NMDA blockade leading to AMPA receptor upregulation and increased BDNF/mTOR signaling.
Acetylcholine & Histamine in Psychiatry
The cholinergic system (nucleus basalis of Meynert projecting to cortex; pedunculopontine and laterodorsal tegmental nuclei projecting to thalamus) is critical for attention and memory. Degeneration of cholinergic neurons is the hallmark of Alzheimer dementia—basis for cholinesterase inhibitor therapy (donepezil, rivastigmine, galantamine). Anticholinergic medication burden in psychiatry (TCAs, low-potency FGAs, benztropine, diphenhydramine) causes cognitive impairment, delirium (especially in elderly), urinary retention, constipation, dry mouth, and blurred vision. The histaminergic system (tuberomammillary nucleus of the posterior hypothalamus) promotes wakefulness. H1 receptor blockade by sedating psychotropics (quetiapine, olanzapine, mirtazapine, doxepin, hydroxyzine) produces sedation and weight gain via appetite stimulation.
The HPA Axis in Psychiatric Illness
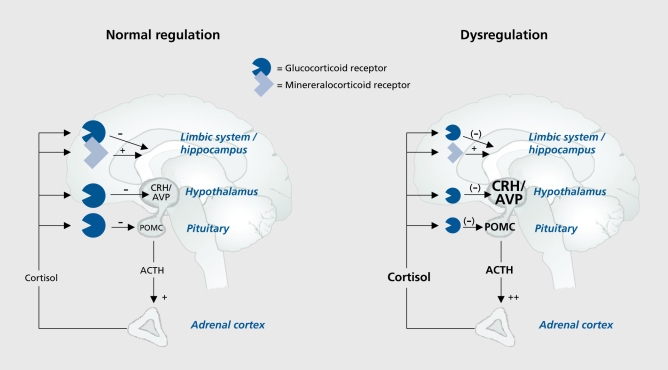
The hypothalamic-pituitary-adrenal axis is the primary stress response system. CRH from the paraventricular nucleus stimulates ACTH from the anterior pituitary, which drives cortisol release from the adrenal cortex. Cortisol exerts negative feedback at the hypothalamus and pituitary. In melancholic depression, this feedback loop is impaired—dexamethasone non-suppression test (DST) is positive in ~50% of patients (cortisol remains >5 mcg/dL after 1 mg dexamethasone at 11 PM). In PTSD, cortisol levels are paradoxically LOW with enhanced negative feedback sensitivity (enhanced dexamethasone suppression), suggesting a distinct HPA axis dysregulation from depression. Chronic cortisol elevation is neurotoxic to hippocampal neurons, contributing to the hippocampal volume loss observed in depression and PTSD.
02 Psychiatric Assessment & Mental Status Examination
The psychiatric interview combines history-taking with systematic behavioral observation. The Mental Status Examination (MSE) is the psychiatric equivalent of the physical exam—it documents the patient's psychological state at the time of assessment and is organized into standardized domains.
Components of the Psychiatric History
Chief complaint (CC) — in the patient's own words. History of present illness (HPI) — onset, duration, severity, precipitants, associated symptoms, functional impact, prior episodes, prior treatments and responses. Past psychiatric history — all previous diagnoses, hospitalizations (voluntary/involuntary), medication trials with doses and reasons for discontinuation, ECT or other neuromodulation, psychotherapy types and duration. Substance use history — each substance with age of first use, route, frequency, quantity, last use, withdrawal history, treatment history (detox, rehab, MAT). Family psychiatric history — first-degree relatives with psychiatric illness, suicide, substance use, hospitalizations. Social history — housing, employment, education, legal history, relationships, trauma/abuse history, access to firearms. Medical history — chronic conditions, medications, allergies, head injuries, seizure history.
Mental Status Examination — Complete Framework
| Domain | What to Assess | Documentation Examples |
|---|---|---|
| Appearance | Age, grooming, hygiene, dress, body habitus, distinguishing features, level of distress | "Appears stated age, disheveled, malodorous, wearing hospital gown, thin habitus, no acute distress" |
| Behavior/Psychomotor | Eye contact, cooperation, agitation, retardation, abnormal movements (tremor, tics, stereotypies, catatonic signs), gait | "Poor eye contact, psychomotor retardation with slowed movements, cooperative but guarded" |
| Speech | Rate, rhythm, volume, tone, spontaneity, articulation, latency | "Slow rate, low volume, monotonous tone, increased latency to response" |
| Mood | Patient's subjective emotional state (in their own words) | "I feel empty" / "Terrible" / "On top of the world" |
| Affect | Clinician-observed emotional expression: range (full, restricted, blunted, flat), quality (euthymic, dysphoric, euphoric, anxious, irritable), congruence with mood, reactivity, lability, appropriateness to content | "Flat affect with minimal facial expression, incongruent with stated mood of 'fine,' non-reactive" |
| Thought process | Organization and flow: linear/goal-directed, circumstantial, tangential, loose associations, flight of ideas, word salad, thought blocking, perseveration, clang associations, neologisms | "Tangential thought process with loose associations and occasional neologisms" |
| Thought content | Suicidal ideation (passive/active, plan, intent, means), homicidal ideation, delusions (type: persecutory, grandiose, referential, erotomanic, somatic, nihilistic, bizarre), obsessions, phobias, ideas of reference, overvalued ideas | "Denies SI/HI. Endorses persecutory delusion that FBI is monitoring him through his TV." |
| Perception | Hallucinations (auditory, visual, tactile, olfactory, gustatory), illusions, depersonalization, derealization | "Endorses command auditory hallucinations telling him to hurt himself. Denies visual hallucinations." |
| Cognition | Alertness, orientation (person, place, time, situation), attention (serial 7s, spell WORLD backward), memory (registration, recall), fund of knowledge, abstraction (proverb interpretation, similarities), executive function | "Alert, oriented x4. Attention impaired—unable to do serial 7s. Recall 1/3 at 5 min." |
| Insight | Patient's understanding of their illness and need for treatment (good, fair, poor, absent) | "Poor insight—does not believe he has a mental illness." |
| Judgment | Quality of decision-making and impulse control, assessed from history and hypothetical scenarios | "Impaired judgment as evidenced by recent reckless spending." |
Full/broad — normal range of emotional expression. Restricted/constricted — reduced range but some variation present. Blunted — severely reduced emotional expression. Flat — complete absence of emotional expression (characteristic of schizophrenia negative symptoms). Labile — rapid, unpredictable shifts in affect (seen in mania, pseudobulbar affect, personality disorders).
03 Diagnostic Frameworks — DSM-5-TR & ICD-11
The DSM-5-TR (Diagnostic and Statistical Manual of Mental Disorders, 5th edition, Text Revision, 2022) is the primary classification system for psychiatric diagnosis in the United States. The ICD-11 (International Classification of Diseases, 11th revision, WHO, 2022) is used internationally and for insurance coding.
Key Changes from DSM-IV to DSM-5
The multiaxial system was eliminated. Previously: Axis I (clinical disorders), Axis II (personality disorders and intellectual disability), Axis III (general medical conditions), Axis IV (psychosocial/environmental stressors), Axis V (GAF score 0–100). DSM-5 integrates all into a single diagnostic listing with separate notation of psychosocial stressors and disability (using WHODAS 2.0 instead of GAF). The bereavement exclusion for MDD was removed—grief and depression can co-occur, and treatment should not be withheld solely because symptoms began after a loss. Asperger syndrome was subsumed into autism spectrum disorder. Substance abuse and dependence were combined into a single substance use disorder with severity specifiers (mild: 2–3 criteria; moderate: 4–5; severe: 6+). The obsessive-compulsive chapter was separated from anxiety disorders. Trauma- and stressor-related disorders became a separate chapter.
Categorical vs. Dimensional Approaches
DSM-5 remains primarily categorical (disorder is present or absent based on threshold criteria), but increasingly incorporates dimensional elements: severity specifiers (mild/moderate/severe), cross-cutting symptom measures, and the Alternative Model for Personality Disorders (AMPD) in Section III, which rates personality functioning on a 0–4 scale and assesses maladaptive trait domains (negative affectivity, detachment, antagonism, disinhibition, psychoticism). ICD-11 has adopted a fully dimensional model for personality disorders, rating severity (mild, moderate, severe) and trait domains rather than categorical PD types.
Neurodevelopmental disorders → Schizophrenia spectrum → Bipolar → Depressive → Anxiety → OCD → Trauma/stressor-related → Dissociative → Somatic symptom → Feeding/eating → Elimination → Sleep-wake → Sexual dysfunctions → Gender dysphoria → Disruptive/impulse-control → Substance use → Neurocognitive → Personality disorders → Paraphilic disorders. The ordering reflects a "lifespan" and neurodevelopmental framework, with chapters organized by shared pathophysiology and comorbidity patterns rather than superficial symptom similarity.
04 Key Terminology & Abbreviations
| Term | Definition |
|---|---|
| Euthymic | Normal, non-depressed, non-manic mood state |
| Anhedonia | Inability to experience pleasure—cardinal symptom of MDD |
| Avolition | Lack of motivation/initiative—negative symptom of schizophrenia |
| Alogia | Poverty of speech—negative symptom of schizophrenia |
| Anosognosia | Lack of awareness of one's own illness; common in schizophrenia and mania |
| Akathisia | Subjective restlessness with inability to sit still—antipsychotic side effect |
| Dystonia | Sustained involuntary muscle contraction—acute EPS, treat with benztropine or diphenhydramine |
| Catatonia | Syndrome of motor and behavioral abnormalities including stupor, mutism, waxy flexibility, posturing, negativism |
| Tangentiality | Responses drift away from the question and never return to the point |
| Circumstantiality | Excessive detail before eventually reaching the point |
| Loose associations | Ideas shift between unrelated topics without logical connection |
| Flight of ideas | Rapid shifting between topics with discernible (if tenuous) associations—seen in mania |
| Word salad | Incoherent, unintelligible mixture of words—severe thought disorganization |
| Thought blocking | Sudden interruption of speech mid-sentence; patient unable to recall what they were saying |
| Ideas of reference | Belief that neutral events have personal significance (e.g., news anchor is speaking to them) |
| Echolalia | Repetition of another's words—seen in catatonia and autism |
| Echopraxia | Imitation of another's movements—seen in catatonia |
| La belle indifférence | Lack of concern about a serious symptom—classically associated with conversion disorder (though not diagnostically reliable) |
| Ego-dystonic | Thoughts/behaviors experienced as distressing and inconsistent with self-image (OCD obsessions) |
| Ego-syntonic | Thoughts/behaviors experienced as consistent with self-image (personality disorder traits, OCPD) |
05 Major Depressive Disorder
DSM-5-TR Diagnostic Criteria
Five or more of the following symptoms present during the same 2-week period, with at least one being (1) depressed mood or (2) anhedonia, representing a change from previous functioning:
Sleep disturbance (insomnia or hypersomnia nearly every day). Interest loss (anhedonia—markedly diminished interest or pleasure in almost all activities). Guilt (excessive or inappropriate guilt, may be delusional). Energy loss (fatigue nearly every day). Concentration impairment (diminished ability to think, concentrate, or indecisiveness). Appetite change (decrease or increase, with ≥5% weight change in a month). Psychomotor agitation or retardation (observable, not merely subjective). Suicidal ideation (recurrent thoughts of death, SI with or without plan). Plus: depressed mood most of the day, nearly every day.
Symptoms must cause clinically significant distress or impairment in social, occupational, or other functioning. Not attributable to substance use or another medical condition. Never met criteria for manic or hypomanic episode (which would reclassify as bipolar).
PHQ-9 Scoring
| Score | Severity | Clinical Action |
|---|---|---|
| 0–4 | Minimal/none | Monitor; no treatment indicated |
| 5–9 | Mild | Watchful waiting; consider counseling |
| 10–14 | Moderate | Treatment plan: therapy and/or medication |
| 15–19 | Moderately severe | Active treatment with pharmacotherapy + therapy |
| 20–27 | Severe | Immediate pharmacotherapy; consider specialist referral |
Severity Specifiers & Subtypes
Mild — few symptoms beyond minimum, minor functional impairment. Moderate — symptom count and functional impairment between mild and severe. Severe — most symptoms present, marked functional impairment, significant distress. With anxious distress — feeling tense, restless, difficulty concentrating due to worry, fear that something awful may happen, feeling of loss of control; rated mild (2 symptoms) to severe (4–5 symptoms); predicts poorer treatment response and higher suicide risk.
Melancholic features — loss of pleasure in all activities (not just reduced), lack of reactivity to positive stimuli, distinct quality of depressed mood (different from grief), worse in morning, early morning awakening (≥2 hours before usual), psychomotor retardation or agitation, significant anorexia/weight loss, excessive guilt. Responds better to TCAs, SNRIs, and ECT than to SSRIs alone.
Atypical features — mood reactivity (mood brightens to positive events), increased appetite/weight gain, hypersomnia, leaden paralysis (heavy feeling in limbs), rejection sensitivity (longstanding pattern). MAOIs historically most effective; SSRIs are first-line in practice.
Psychotic features — delusions or hallucinations present during the depressive episode. Mood-congruent (themes of guilt, worthlessness, disease, death, deserved punishment) vs. mood-incongruent (persecutory, thought insertion—worse prognosis). Treatment requires antidepressant PLUS antipsychotic; ECT is highly effective.
Treatment Algorithm
Step 1 (mild-moderate): Psychotherapy alone (CBT or interpersonal therapy) or SSRI monotherapy. Step 2 (moderate-severe or failed Step 1): SSRI at adequate dose (e.g., sertraline 100–200 mg, escitalopram 10–20 mg) for adequate duration (4–6 weeks at therapeutic dose). Step 3 (partial response): Augmentation with aripiprazole 2–10 mg, lithium 600–900 mg (level 0.6–0.8), bupropion 150–300 mg, or T3 (liothyronine) 25–50 mcg. Step 4 (non-response): Switch antidepressant class (SSRI to SNRI, or to bupropion, mirtazapine). Step 5 (treatment-resistant depression—TRD): Defined as failure of ≥2 adequate antidepressant trials. Options include esketamine (Spravato) nasal spray, IV ketamine, ECT, TMS, or MAOI.
Treatment-Resistant Depression (TRD)
Defined as failure to achieve remission after ≥2 adequate antidepressant trials (adequate dose for adequate duration of ≥6–8 weeks with confirmed adherence). Affects approximately 30% of MDD patients. Re-evaluate diagnosis (rule out bipolar disorder, substance use, medical causes, personality disorders). Confirm treatment adequacy—check serum drug levels when available (e.g., nortriptyline, imipramine). Strategies include switching class, augmentation (lithium, aripiprazole, triiodothyronine), combination antidepressants (SSRI + bupropion, SSRI + mirtazapine), esketamine nasal spray (Spravato), IV ketamine, ECT, TMS, and MAOI trial.
Maintenance & Discontinuation
First episode of MDD: continue antidepressant for ≥6–9 months after remission. Two or more episodes, chronic course, or severe index episode: consider long-term (possibly indefinite) maintenance. When discontinuing, taper over ≥4 weeks (longer for paroxetine and venlafaxine) to avoid antidepressant discontinuation syndrome (dizziness, nausea, headache, paresthesias/"electric shocks," irritability, insomnia, vivid dreams—onset within 2–4 days of abrupt cessation; typically resolves within 1–2 weeks; NOT a sign of addiction). Fluoxetine has the lowest discontinuation risk due to its long half-life.
06 Bipolar I & II Disorder
Bipolar I Disorder
Defined by the occurrence of at least one manic episode. A manic episode requires a distinct period of abnormally and persistently elevated, expansive, or irritable mood AND increased goal-directed activity or energy, lasting ≥7 days (or any duration if hospitalization is required).
Three or more of the following (four if mood is only irritable): Distractibility. Indiscretion (excessive involvement in pleasurable activities with high potential for painful consequences—spending sprees, sexual indiscretions, foolish investments). Grandiosity (inflated self-esteem, may be delusional). Flight of ideas or racing thoughts. Activity increase (goal-directed or psychomotor agitation). Sleep decreased (decreased need for sleep—feels rested after 3 hours, distinct from insomnia). Talkativeness (pressured speech, difficult to interrupt). The episode must cause marked functional impairment, necessitate hospitalization, or include psychotic features.
Bipolar II Disorder
Defined by at least one hypomanic episode AND at least one major depressive episode, with no history of a full manic episode. A hypomanic episode has the same symptom criteria as mania but lasts ≥4 days (not 7), does NOT cause marked functional impairment, does NOT require hospitalization, and has NO psychotic features. The change in functioning is uncharacteristic and observable by others but not severe enough to cause marked impairment. Bipolar II is NOT "milder" bipolar I—depressive burden is often greater, and suicide risk is comparable or higher.
Additional Specifiers
Mixed features — full criteria for one pole with ≥3 symptoms of the opposite pole simultaneously (e.g., manic episode with depressive symptoms). Carries higher suicide risk. Rapid cycling — ≥4 mood episodes in 12 months (any combination of manic, hypomanic, depressive). More common in women, associated with hypothyroidism and antidepressant use. Lithium may be less effective; valproate and lamotrigine preferred. With psychotic features — seen in severe mania or depression; mood-congruent (grandiose in mania, nihilistic in depression) vs. mood-incongruent.
Pharmacotherapy
| Agent | Acute Mania | Acute Depression | Maintenance | Key Monitoring |
|---|---|---|---|---|
| Lithium | Yes (level 0.8–1.2) | Moderate evidence | Yes (level 0.6–1.0)—anti-suicide | Levels, Cr, TSH, Ca q6mo; ECG |
| Valproate (divalproex) | Yes (level 80–120 mcg/mL) | Limited | Yes | Levels, LFTs, CBC, ammonia; teratogen (neural tube defects) |
| Lamotrigine | No | Yes (first-line bipolar depression) | Yes (depression prevention) | Rash monitoring—SJS risk; titrate slowly (25 mg x2 wk → 50 mg x2 wk → 100 mg → 200 mg target) |
| Quetiapine | Yes | Yes (300 mg for bipolar depression) | Yes | Metabolic panel, fasting glucose, lipids, weight |
| Olanzapine | Yes (high efficacy) | Yes (olanzapine-fluoxetine combo) | Yes (weight limiting) | Metabolic monitoring; highest weight gain risk among SGAs |
| Aripiprazole | Yes | No | Yes | Akathisia monitoring; weight-neutral |
| Carbamazepine | Yes | Limited | Yes | Levels 4–12 mcg/mL, CBC (agranulocytosis), LFTs, Na; CYP inducer—many drug interactions; HLA-B*1502 testing in Asian patients |
| Lurasidone | No | Yes (FDA-approved bipolar depression) | Limited | Take with ≥350 kcal food; metabolic monitoring |
07 Persistent Depressive Disorder & Cyclothymia
Peripartum & Seasonal Specifiers
With peripartum onset — mood episode (depressive or manic) with onset during pregnancy or within 4 weeks postpartum (clinically, symptoms may begin anytime in the first year). Peripartum depression affects ~10–15% of women; screening with Edinburgh Postnatal Depression Scale (EPDS, cutoff ≥10). Distinguish from "baby blues" (mood lability, tearfulness in first 2 weeks postpartum, self-limited, affects 50–80% of women). Postpartum psychosis is a psychiatric emergency (1–2/1000 births)—typically onset within first 2 weeks; confusion, disorientation, mood lability, hallucinations, paranoia, disorganized behavior; 5% infanticide rate; requires hospitalization; strongly associated with bipolar disorder. Treatment: mood stabilizer + antipsychotic; ECT for severe/refractory cases. Brexanolone (Zulresso) IV is FDA-approved for postpartum depression (neurosteroid GABA-A modulator; 60-hour infusion; requires REMS).
With seasonal pattern (seasonal affective disorder, SAD) — regular temporal relationship between onset and remission and a particular time of year (typically fall/winter onset, spring/summer remission). Characterized by atypical features (hypersomnia, increased appetite, carbohydrate craving, weight gain, leaden paralysis). Treatment: bright light therapy (10,000 lux, 30 minutes each morning, within first hour of waking)—first-line; SSRIs; bupropion XL is FDA-approved for prevention of seasonal depressive episodes.
Persistent Depressive Disorder (Dysthymia)
Depressed mood for most of the day, for more days than not, for ≥2 years (1 year in children/adolescents). During the 2-year period, never symptom-free for >2 months. While depressed, two or more of: poor appetite or overeating, insomnia or hypersomnia, low energy/fatigue, low self-esteem, poor concentration/difficulty making decisions, feelings of hopelessness. May have concurrent major depressive episodes superimposed ("double depression"). Treatment: SSRIs/SNRIs are first-line; combination with psychotherapy (CBT or CBASP—Cognitive Behavioral Analysis System of Psychotherapy) shows best outcomes. CBASP was specifically developed for chronic depression and focuses on interpersonal problem-solving.
Cyclothymic Disorder
Chronic, fluctuating mood disturbance for ≥2 years involving numerous periods of hypomanic symptoms (not meeting full hypomanic criteria) and depressive symptoms (not meeting MDD criteria). Never symptom-free for >2 months. Symptoms never severe enough to meet full criteria for manic, hypomanic, or major depressive episode. Approximately 15–50% eventually develop bipolar I or II disorder. Management: mood stabilizers (lithium, valproate) if symptoms are functionally impairing; psychoeducation about risk of progression; avoid antidepressant monotherapy (risk of hypomanic switch).
08 Suicide Risk Assessment & Management
Suicide is the 10th leading cause of death in the US (~48,000 deaths/year) and the 2nd leading cause in ages 10–34. Approximately 90% of completed suicides have a diagnosable psychiatric disorder at the time of death. Risk assessment is a core psychiatric competency.
Risk Factors
Male sex (complete suicide 3.5x more common in men; women attempt 3x more often). Age (>65 years highest completion rate; 15–24 years highest attempt rate). White and Native American/Alaska Native ethnicity. Family history of suicide (2–6x increased risk). Prior suicide attempt (single strongest predictor—20–30x increased risk). History of childhood abuse/trauma. Military/veteran status.
Active psychiatric illness (depression, bipolar, schizophrenia, borderline PD, substance use). Current substance intoxication (alcohol involved in ~25% of suicides). Access to lethal means (firearms account for ~50% of suicide deaths in US). Social isolation, recent loss, unemployment. Hopelessness (Beck Hopelessness Scale score ≥9 is strongly predictive). Chronic pain, terminal illness. Recent psychiatric hospitalization (highest risk in first week after discharge).
Protective Factors
Strong social support and connectedness. Children in the home (especially for women). Religious/spiritual beliefs. Reasons for living. Engaged in treatment. Problem-solving skills. Restricted access to means.
Columbia Suicide Severity Rating Scale (C-SSRS)
Standardized tool for assessing suicidal ideation and behavior. Assesses five types of ideation on increasing severity: (1) wish to be dead, (2) non-specific active suicidal thoughts, (3) active suicidal ideation with any methods (not plan), (4) active suicidal ideation with some intent to act, (5) active suicidal ideation with specific plan and intent. Also assesses suicidal behavior: actual attempt, interrupted attempt, aborted attempt, preparatory acts/behavior. Widely adopted in ED triage protocols.
SAD PERSONS Scale
| Letter | Factor | Points |
|---|---|---|
| S | Sex (male) | 1 |
| A | Age (<19 or >45) | 1 |
| D | Depression | 2 |
| P | Previous attempt | 1 |
| E | Ethanol/substance abuse | 1 |
| R | Rational thinking loss (psychosis) | 2 |
| S | Social supports lacking | 1 |
| O | Organized plan | 2 |
| N | No spouse/partner | 1 |
| S | Sickness (chronic/debilitating) | 1 |
Scoring: 0–5 may be safe to discharge with outpatient follow-up; 6–8 consider hospitalization; ≥9 strong indication for admission.
Safety Planning
The Stanley-Brown Safety Plan is a brief, structured intervention: (1) Warning signs that a crisis may be developing. (2) Internal coping strategies (self-soothing, distraction). (3) Social contacts and settings that provide distraction. (4) People to call for help. (5) Professionals/agencies to contact in crisis (988 Suicide and Crisis Lifeline, local crisis services, ED). (6) Making the environment safe—means restriction. Lethal means counseling: Directly ask about firearms, medications, and other means; counsel on safe storage, removal, or transfer to a trusted person. Means restriction is one of the most evidence-based suicide prevention strategies.
High-Risk Periods & Warning Signs
Acute warning signs (requiring immediate assessment): threatening to hurt or kill self, looking for means (access to firearms, pills, rope), talking or writing about death/dying/suicide, hopelessness, rage/anger/seeking revenge, recklessness, feeling trapped, increasing substance use, withdrawing from friends/family, anxiety/agitation, dramatic mood changes, no sense of purpose. High-risk transition periods: First week after psychiatric hospital discharge (40x population risk), first month after discharge, transition from inpatient to outpatient care, early antidepressant treatment (activation before mood improvement, particularly in adolescents/young adults—basis for FDA black box warning on antidepressants in ages <25), loss of key relationship or support, anniversary dates of traumatic events.
Documentation of Suicide Risk Assessment
Every suicide risk assessment should document: (1) Risk factors identified (static and dynamic). (2) Protective factors identified. (3) Current ideation (passive vs. active, plan, intent, means, timeline). (4) Prior attempts (number, lethality, circumstances). (5) Overall risk level determination (low, moderate, high, imminent). (6) Clinical rationale for risk level. (7) Interventions implemented (safety plan, means restriction counseling, medication adjustments, disposition decision). (8) Follow-up plan. This documentation serves both clinical and medicolegal purposes.
09 Schizophrenia
Schizophrenia is a chronic psychotic disorder with a lifetime prevalence of ~1%, equal across sexes. Onset typically late adolescence to early adulthood (males 18–25, females 25–35, with a second peak in women at ~45). Heritability is ~80%; concordance in monozygotic twins is ~48% (not 100%, indicating environmental contributions).
Schneider First-Rank Symptoms
Kurt Schneider identified symptoms considered highly suggestive (though not pathognomonic) of schizophrenia: auditory hallucinations (voices commenting on actions, two or more voices conversing, thought echo/audible thoughts), thought withdrawal (thoughts removed from one's mind by external force), thought insertion (alien thoughts placed in one's mind), thought broadcasting (one's thoughts are accessible to others), delusions of control/passivity (actions, feelings, or impulses controlled by external force), delusional perception (normal perception given private, delusional significance), somatic passivity (bodily sensations imposed by external agency).
Symptom Domains
| Domain | Symptoms | Neurobiology | Treatment Response |
|---|---|---|---|
| Positive | Hallucinations (auditory most common ~70%), delusions (persecutory, referential, grandiose, bizarre), disorganized speech | Mesolimbic DA excess | Good response to antipsychotics |
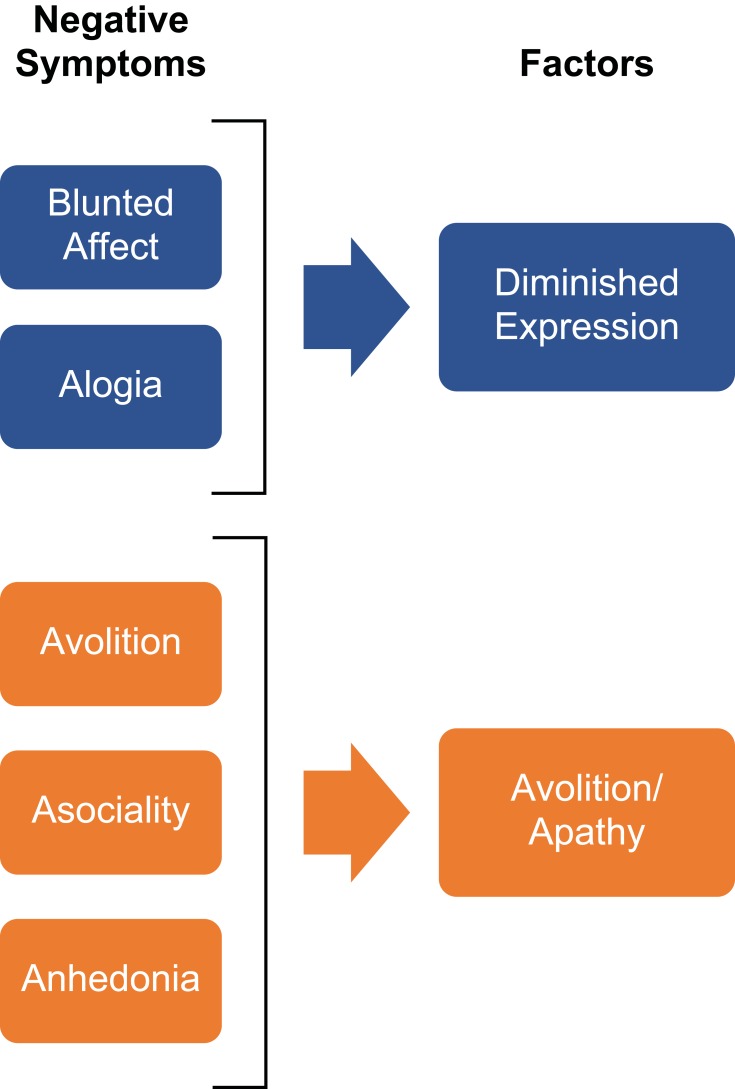
| Negative | Affective flattening, alogia, avolition, anhedonia, asociality (the "5 A's") | Mesocortical DA deficit; frontal hypometabolism | Poor response to typical antipsychotics; clozapine, cariprazine may help |
| Cognitive | Impaired working memory, attention, processing speed, executive function | dlPFC dysfunction | Largely unresponsive to current medications; cognitive remediation therapy |
| Disorganized | Disorganized speech (tangentiality, loose associations, word salad), disorganized behavior (bizarre dress, inappropriate affect, catatonia) | Frontal-subcortical circuit disruption | Moderate response to antipsychotics |
| Affective | Depression, anxiety, suicidality (~5% lifetime suicide rate), hostility | Multifactorial | May need adjunctive antidepressant |
DSM-5 Diagnostic Criteria
Two or more of the following, each present for a significant portion of time during a 1-month period (at least one must be 1, 2, or 3): (1) Delusions, (2) Hallucinations, (3) Disorganized speech, (4) Grossly disorganized or catatonic behavior, (5) Negative symptoms. Level of functioning in work, interpersonal relations, or self-care is markedly below premorbid level. Continuous signs of the disturbance persist for ≥6 months (including at least 1 month of active-phase symptoms). Schizoaffective and mood disorders excluded. Not attributable to substance or medical condition.
Phases of Illness
Prodromal — attenuated positive symptoms (unusual thought content, suspiciousness, perceptual disturbances), social withdrawal, declining function, typically 1–5 years before first episode; identified by structured assessment (SIPS/SOPS). Active (acute) — full psychotic symptoms meeting diagnostic criteria. Residual — between acute episodes; may have persistent negative symptoms, attenuated positive symptoms, continued functional impairment.
Antipsychotic Selection
All antipsychotics (except clozapine) have similar efficacy for positive symptoms. Selection is guided by side effect profile. D2 receptor occupancy of 60–80% is the therapeutic window; >80% increases EPS risk. First-generation (typical) antipsychotics (high-potency: haloperidol, fluphenazine—more EPS, less sedation/anticholinergic; low-potency: chlorpromazine—more sedation, orthostasis, anticholinergic, less EPS). Second-generation (atypical) antipsychotics — lower EPS risk (due to 5-HT2A antagonism) but higher metabolic risk (weight gain, diabetes, dyslipidemia—especially clozapine and olanzapine).

10 Schizoaffective, Brief Psychotic & Delusional Disorder
Schizoaffective Disorder
An uninterrupted period of illness during which there is a major mood episode (depressive or manic) concurrent with Criterion A symptoms of schizophrenia. Key distinguishing criterion: Delusions or hallucinations must be present for ≥2 weeks in the absence of a major mood episode during the lifetime duration of the illness. This separates schizoaffective disorder from mood disorders with psychotic features (where psychosis occurs only during mood episodes). Two subtypes: bipolar type (manic episode included) and depressive type (only depressive episodes). Treatment combines antipsychotic + mood stabilizer or antidepressant as appropriate for subtype.
Brief Psychotic Disorder
At least one of: delusions, hallucinations, disorganized speech, grossly disorganized or catatonic behavior. Duration: ≥1 day but <1 month, with full return to premorbid functioning. Often triggered by an identifiable stressor (specify "with marked stressor" vs. "without"). Postpartum onset is a specifier. Treatment: short-term antipsychotic; benzodiazepine for agitation; most patients recover fully, but ~50% eventually receive a longer-term psychotic disorder diagnosis.
Delusional Disorder
One or more delusions lasting ≥1 month. Criteria A for schizophrenia has never been met (hallucinations, if present, are not prominent and are related to the delusional theme). Apart from the impact of the delusion(s), functioning is not markedly impaired and behavior is not obviously bizarre.
| Subtype | Core Delusion | Key Features |
|---|---|---|
| Persecutory | Being conspired against, spied on, poisoned | Most common subtype; may involve litigiousness |
| Grandiose | Inflated self-worth, special power, identity, or relationship | May believe they have a great unrecognized talent or discovery |
| Erotomanic | Another person (often famous) is in love with them | De Clérambault syndrome; may lead to stalking |
| Jealous | Partner is unfaithful | Othello syndrome; can be dangerous |
| Somatic | Bodily function or sensation (e.g., infestation, body odor, physical deformity) | Morgellons delusion, delusional parasitosis |
| Mixed | More than one delusional theme | No single theme predominates |
11 First-Episode Psychosis — Workup & Management
First-episode psychosis (FEP) warrants a thorough medical workup to rule out organic causes before attributing symptoms to a primary psychotic disorder. Duration of untreated psychosis (DUP) is the single strongest predictor of long-term outcome—every effort should target early intervention.
Medical Mimics to Rule Out
Substances: Stimulants (amphetamine, cocaine, methamphetamine), cannabis (especially high-potency THC), PCP, LSD, synthetic cannabinoids, alcohol withdrawal (delirium tremens), benzodiazepine withdrawal, anticholinergics, corticosteroids. Neurological: Temporal lobe epilepsy, brain tumor (especially temporal/frontal lobe), dementia with Lewy bodies, Huntington disease, Wilson disease, multiple sclerosis, neurosyphilis, HIV encephalopathy, NMDA receptor encephalitis (anti-NMDAR—especially in young women with ovarian teratoma). Endocrine/Metabolic: Thyroid storm, myxedema, Cushing disease, Addison disease, acute intermittent porphyria, hepatic encephalopathy, uremia, hypo/hypernatremia, hypo/hyperglycemia, B12 deficiency. Autoimmune: SLE cerebritis, anti-NMDAR encephalitis, Hashimoto encephalopathy.
Recommended Workup
| Category | Tests | Rationale |
|---|---|---|
| Baseline labs | CBC with differential, CMP (glucose, electrolytes, renal/hepatic function), TSH, urinalysis | Screen for metabolic derangements, thyroid disease, infection |
| Toxicology | Urine drug screen (UDS), blood alcohol level | Substance-induced psychosis is the most common mimic |
| Infectious | RPR/VDRL (syphilis), HIV | Neurosyphilis and HIV encephalopathy present with psychiatric symptoms |
| Nutritional | Vitamin B12, folate | B12 deficiency can cause psychosis |
| Autoimmune | ANA, ESR/CRP; NMDA receptor antibodies if clinical suspicion | SLE cerebritis, anti-NMDAR encephalitis |
| Metabolic | Ceruloplasmin (if age <40) | Wilson disease |
| Neuroimaging | Brain MRI (preferred) or CT | Rule out tumor, demyelination, structural lesion |
| Other | EEG (if seizure suspected), lumbar puncture (if encephalitis suspected), pregnancy test | Temporal lobe epilepsy, autoimmune encephalitis |
Early Intervention Principles
Coordinated specialty care (CSC) programs like NAVIGATE and OnTrackNY improve outcomes in FEP. Components: low-dose antipsychotic (start low, go slow—risperidone 1–3 mg or aripiprazole 5–15 mg), individual CBT for psychosis (CBTp), family psychoeducation, supported employment/education, and case management. The RAISE trial demonstrated that CSC improved quality of life, symptoms, and engagement compared to usual care. Antipsychotic dose in FEP should be ~50% of the dose used for chronic schizophrenia—FEP patients are more sensitive to both efficacy and side effects.
Prognostic Factors in First-Episode Psychosis
Good prognosis: Acute onset, clear precipitant, late onset (>30 years), female sex, good premorbid functioning, married/stable relationships, prominent mood symptoms, family history of mood disorders, absence of negative symptoms, early treatment initiation (short DUP). Poor prognosis: Insidious onset, early onset, male sex, poor premorbid functioning, social isolation, prominent negative symptoms, family history of schizophrenia, structural brain abnormalities (enlarged ventricles, reduced gray matter), substance use comorbidity, long DUP (>1 year), poor treatment adherence.
The "rule of thirds" for schizophrenia prognosis is a rough approximation: approximately one-third achieve significant improvement and good functional outcomes, one-third have intermittent episodes with some residual impairment, and one-third have a chronic, unremitting course with persistent disability. Early intervention programs aim to shift these proportions toward better outcomes.
12 Generalized Anxiety Disorder
Generalized Anxiety Disorder (GAD) is characterized by excessive anxiety and worry (apprehensive expectation) about a number of events or activities, occurring more days than not for ≥6 months. The individual finds it difficult to control the worry. Three or more of (only one required in children): (1) Restlessness or feeling keyed up/on edge, (2) Being easily fatigued, (3) Difficulty concentrating or mind going blank, (4) Irritability, (5) Muscle tension, (6) Sleep disturbance. Lifetime prevalence ~5.7%; F:M ratio 2:1.
GAD-7 Screening Tool
| Score | Severity | Action |
|---|---|---|
| 0–4 | Minimal | Monitor |
| 5–9 | Mild | Watchful waiting; reassess |
| 10–14 | Moderate | Consider treatment (therapy and/or medication) |
| 15–21 | Severe | Active treatment warranted |
Sensitivity 89%, specificity 82% for GAD at cutoff ≥10.
Treatment
First-line pharmacotherapy: SSRIs (sertraline 50–200 mg, escitalopram 10–20 mg, paroxetine 20–50 mg) or SNRIs (venlafaxine XR 75–225 mg, duloxetine 60–120 mg). Allow 4–6 weeks at adequate dose before judging efficacy. Second-line: Buspirone 15–60 mg/day in divided doses (5-HT1A partial agonist; no sedation, no dependence, no interaction with alcohol; takes 2–4 weeks for effect; ineffective PRN). Adjunctive: Hydroxyzine 25–50 mg TID (antihistamine anxiolytic; no dependence risk). Pregabalin 150–600 mg/day (approved for GAD in Europe, not US). Short-term/bridging: Benzodiazepines (lorazepam 0.5–1 mg BID-TID, clonazepam 0.25–0.5 mg BID) for acute symptom relief while waiting for SSRI/SNRI onset; taper within 2–4 weeks to avoid dependence. Psychotherapy: CBT is the gold standard—comparable efficacy to medication, more durable response. Key CBT components: psychoeducation, cognitive restructuring, relaxation training, worry time scheduling, behavioral experiments.
Differential Diagnosis of Anxiety
| Medical Condition | Distinguishing Clues | Workup |
|---|---|---|
| Hyperthyroidism | Weight loss, heat intolerance, tremor, tachycardia, exophthalmos | TSH (low), free T4 (high) |
| Pheochromocytoma | Episodic hypertension, headache, diaphoresis, pallor | Plasma free metanephrines or 24-hr urine catecholamines |
| Cardiac arrhythmia | Palpitations with syncope, irregular pulse, known cardiac history | ECG, Holter monitor, event recorder |
| Caffeinism | >400 mg caffeine/day; temporal relationship with intake | History; caffeine diary; symptoms resolve with reduction |
| Substance use/withdrawal | Stimulants (intoxication), alcohol/BZD (withdrawal), cannabis | UDS; timeline of substance use vs. symptom onset |
| Temporal lobe epilepsy | Episodic fear with automatisms, aura, altered consciousness | EEG; brain MRI |
| COPD/Asthma | Dyspnea, wheezing, smoking history | PFTs; CXR |
13 Panic Disorder & Agoraphobia
Panic Attack Criteria
An abrupt surge of intense fear or discomfort reaching a peak within minutes, with four or more of 13 symptoms: (1) Palpitations or accelerated heart rate, (2) Sweating, (3) Trembling or shaking, (4) Shortness of breath or smothering, (5) Feelings of choking, (6) Chest pain or discomfort, (7) Nausea or abdominal distress, (8) Dizziness, lightheadedness, or faintness, (9) Chills or heat sensations, (10) Paresthesias (numbness or tingling), (11) Derealization or depersonalization, (12) Fear of losing control or "going crazy," (13) Fear of dying. Panic attacks can occur in any anxiety disorder but are not themselves a diagnosis.
Panic Disorder
Recurrent unexpected panic attacks (at least two) with ≥1 month of persistent concern about additional attacks, worry about consequences ("going crazy," "having a heart attack"), or maladaptive behavioral change (avoidance). Lifetime prevalence ~2–3%; F:M 2:1.
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Acute coronary syndrome | ECG changes, troponin elevation, risk factors; chest pain not confined to attacks |
| Pheochromocytoma | Episodic hypertension, headache, diaphoresis, pallor; elevated metanephrines |
| Hyperthyroidism | Weight loss, heat intolerance, tremor, goiter; abnormal TSH/T4 |
| Cardiac arrhythmia | Irregular pulse, ECG/Holter abnormalities, may have syncope |
| Asthma/COPD exacerbation | Wheezing, known pulmonary disease, abnormal PFTs |
| Substance-induced | Caffeine, stimulants, cannabis; temporal relationship to use |
Treatment
First-line: SSRIs (start at half typical dose due to initial anxiogenic effect—sertraline 25 mg, escitalopram 5 mg, then titrate). SNRIs also effective. Acute/bridging: Benzodiazepines (alprazolam 0.25–0.5 mg TID, clonazepam 0.5 mg BID) while waiting for SSRI onset. Psychotherapy: CBT with interoceptive exposure (deliberately inducing panic symptoms in a controlled setting) and cognitive restructuring of catastrophic misinterpretations of bodily sensations. Combination CBT + SSRI may be superior to either alone. Avoid: Long-term benzodiazepine monotherapy, caffeine, stimulant medications.
Agoraphobia
Marked fear or anxiety about two or more of five situations: (1) Using public transportation, (2) Being in open spaces, (3) Being in enclosed places, (4) Standing in line or being in a crowd, (5) Being outside of the home alone. Feared situations are avoided or endured with intense distress because escape might be difficult or help unavailable if panic-like symptoms occur. Duration ≥6 months. In DSM-5, agoraphobia is diagnosed independently of panic disorder (unlike DSM-IV). Treatment: SSRI/SNRI plus CBT with graded exposure to feared situations.
14 PTSD & Acute Stress Disorder
DSM-5 PTSD Criteria
Exposure to actual or threatened death, serious injury, or sexual violence via: direct experience, witnessing in person, learning it occurred to a close family member/friend (must be violent or accidental), or repeated/extreme exposure to aversive details (e.g., first responders). Then four symptom clusters, each required:
Recurrent, involuntary, intrusive distressing memories. Recurrent distressing dreams related to the event. Dissociative reactions (flashbacks) in which the individual feels or acts as if the event were recurring. Intense psychological distress at exposure to cues. Marked physiological reactions to cues resembling the event.
Avoidance of distressing memories, thoughts, or feelings associated with the traumatic event. Avoidance of external reminders (people, places, activities, situations) that arouse distressing memories/thoughts/feelings.
Inability to remember an important aspect of the event (dissociative amnesia). Persistent negative beliefs about oneself, others, or the world ("I am bad," "No one can be trusted," "The world is completely dangerous"). Persistent distorted cognitions about the cause or consequences of the event leading to self-blame. Persistent negative emotional state (fear, horror, anger, guilt, shame). Markedly diminished interest in significant activities. Feelings of detachment or estrangement from others. Persistent inability to experience positive emotions (emotional numbing).
Irritable behavior and angry outbursts (with little or no provocation). Reckless or self-destructive behavior. Hypervigilance. Exaggerated startle response. Problems with concentration. Sleep disturbance (difficulty falling/staying asleep).
Duration >1 month (if <1 month, consider acute stress disorder). Specifiers: with dissociative symptoms (depersonalization or derealization), with delayed expression (full criteria not met until ≥6 months after event).
PCL-5 (PTSD Checklist for DSM-5)
20-item self-report measure corresponding to DSM-5 PTSD symptoms. Each item scored 0–4 (not at all to extremely). Total score range 0–80. Provisional diagnosis cutoff: ≥31–33. Can also score by cluster to identify predominant symptom domains.
Evidence-Based Treatments
Psychotherapy (first-line): Prolonged Exposure (PE) — repeated in-vivo and imaginal exposure to trauma memories and avoided situations; 8–15 sessions. Cognitive Processing Therapy (CPT) — identifies and challenges "stuck points" (maladaptive beliefs about the trauma); 12 sessions. EMDR (Eye Movement Desensitization and Reprocessing) — bilateral stimulation during trauma memory processing; 6–12 sessions. All three have strong Level 1 evidence.
Pharmacotherapy: SSRIs are first-line (sertraline and paroxetine are FDA-approved for PTSD; typical doses sertraline 50–200 mg, paroxetine 20–50 mg). SNRIs (venlafaxine) also effective. Prazosin 1–15 mg at bedtime for trauma-related nightmares (alpha-1 antagonist that reduces noradrenergic hyperarousal during sleep—titrate from 1 mg, increase by 1–2 mg every 3–7 days; monitor for orthostatic hypotension). Benzodiazepines are NOT recommended in PTSD—they may worsen outcomes and interfere with fear extinction learning.

Acute Stress Disorder
Similar symptom profile to PTSD but occurs 3 days to 1 month after trauma. Requires ≥9 of 14 symptoms across any of the five categories (intrusion, negative mood, dissociation, avoidance, arousal). Approximately 50% of individuals with acute stress disorder progress to PTSD. Early CBT with trauma-focused exposure reduces progression to PTSD.
15 OCD & Related Disorders
Obsessive-Compulsive Disorder
Presence of obsessions (recurrent, persistent, intrusive, unwanted thoughts, urges, or images causing marked anxiety) and/or compulsions (repetitive behaviors or mental acts performed to reduce anxiety or prevent a dreaded event). Time-consuming (≥1 hour/day) or cause clinically significant distress/impairment. Lifetime prevalence ~2.3%. Equal sex ratio in adults (male predominance in childhood-onset). Mean age of onset ~20 years.
Common Obsession-Compulsion Pairs
| Obsession Theme | Typical Compulsions |
|---|---|
| Contamination (dirt, germs, bodily fluids) | Handwashing, cleaning, avoiding "contaminated" objects |
| Harm (fear of harming self or others) | Checking locks, stoves, appliances; seeking reassurance |
| Symmetry/exactness | Ordering, arranging, counting, repeating until "just right" |
| Forbidden/taboo thoughts (sexual, religious, violent) | Mental rituals (praying, counting, neutralizing), avoidance |
| Doubt/incompleteness | Checking, rereading, rewriting |
| Hoarding (if primary obsession about discarding) | Saving, difficulty discarding items |
Yale-Brown Obsessive Compulsive Scale (Y-BOCS)
10 items (5 for obsessions, 5 for compulsions), each scored 0–4. Total score range 0–40. Severity thresholds: 0–7 subclinical, 8–15 mild, 16–23 moderate, 24–31 severe, 32–40 extreme. A ≥35% reduction in Y-BOCS score is considered treatment response.
Treatment
First-line: SRI therapy (SSRIs at HIGHER doses than used for depression—this is critical). Typical OCD doses: fluoxetine 40–80 mg, fluvoxamine 100–300 mg, sertraline 100–200 mg, paroxetine 40–60 mg, escitalopram 20–40 mg, citalopram 40–60 mg. Clomipramine (TCA with potent SRI activity) 150–250 mg is the most effective single agent but limited by side effects (anticholinergic, cardiac). Adequate trial duration: 8–12 weeks (longer than for depression). Second-line/augmentation: Low-dose antipsychotic augmentation (aripiprazole 2–10 mg, risperidone 0.5–2 mg) for SRI partial responders—about 1/3 of SRI non-responders improve with augmentation. Psychotherapy: Exposure and Response Prevention (ERP) is the gold-standard psychotherapy—systematic, graduated exposure to obsessional triggers while preventing the compulsive response. Combination ERP + SRI is superior to either alone.
OCD-Related Disorders
Body Dysmorphic Disorder (BDD) — preoccupation with perceived defects in physical appearance not observable to others; repetitive behaviors (mirror checking, skin picking, reassurance seeking, camouflaging). Treatment: SSRI (high dose, as for OCD) + CBT with ERP. Cosmetic procedures generally do NOT improve BDD and may worsen it. Hoarding Disorder — persistent difficulty discarding possessions regardless of value, resulting in accumulation that clutters living spaces and compromises their intended use. Treatment: specialized CBT for hoarding (not standard ERP); SRIs less effective than in OCD. Trichotillomania (Hair-Pulling Disorder) — recurrent pulling out of one's hair resulting in hair loss. Treatment: habit reversal training (HRT); N-acetylcysteine 1200–2400 mg/day has some evidence. Excoriation (Skin-Picking) Disorder — recurrent skin picking resulting in skin lesions. Treatment: HRT; SSRIs may help.
OCD Pharmacotherapy Dosing Comparison
| Drug | MDD Dose | OCD Dose | FDA Max |
|---|---|---|---|
| Fluoxetine | 20–40 mg | 40–80 mg | 80 mg |
| Fluvoxamine | N/A (not FDA-approved for MDD) | 100–300 mg | 300 mg |
| Sertraline | 50–150 mg | 100–200 mg | 200 mg |
| Paroxetine | 20–40 mg | 40–60 mg | 60 mg |
| Escitalopram | 10–20 mg | 20–40 mg | 40 mg (off-label for OCD) |
| Clomipramine | 100–200 mg | 150–250 mg | 250 mg |
OCD requires higher SRI doses and longer trial duration (8–12 weeks) than depression. Approximately 40–60% of patients respond to initial SRI trial. If inadequate response, switch SRI, augment with low-dose atypical antipsychotic, or add CBT/ERP.
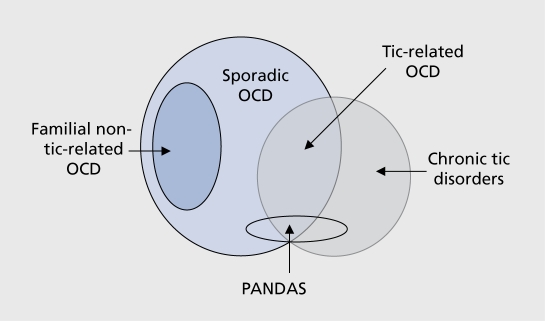
PANDAS (Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal infections): Acute-onset OCD or tic disorder in prepubertal children temporally associated with group A streptococcal infection. Thought to involve molecular mimicry with anti-basal ganglia antibodies. PANS (Pediatric Acute-onset Neuropsychiatric Syndrome): Broader category including non-streptococcal infectious and non-infectious triggers. Treatment: treat underlying infection (antibiotics); immunomodulatory therapy (IVIG, plasmapheresis) for severe cases; standard OCD treatments (SRI + CBT) for persistent symptoms.
16 Personality Disorders — Clusters A, B, C
Personality disorders are enduring, pervasive patterns of inner experience and behavior that deviate markedly from cultural expectations, are inflexible, onset in adolescence/early adulthood, are stable over time, and lead to distress or impairment. They manifest in ≥2 of: cognition, affectivity, interpersonal functioning, impulse control.
Cluster A — "Odd/Eccentric"
| Disorder | Key Features | Distinguishing Points |
|---|---|---|
| Paranoid PD | Pervasive distrust and suspiciousness; interprets motives as malevolent | Does not have persistent delusions (vs. delusional disorder); suspicious but reality-testing intact |
| Schizoid PD | Detachment from social relationships; restricted emotional expression; preference for solitary activities; indifference to praise/criticism | Does NOT desire relationships (vs. avoidant PD, who desires them but fears rejection) |
| Schizotypal PD | Acute discomfort in close relationships, cognitive/perceptual distortions, eccentric behavior; ideas of reference, magical thinking, unusual perceptual experiences, odd speech, suspiciousness, constricted affect | Genetically related to schizophrenia; may progress to schizophrenia in ~10% |
Cluster B — "Dramatic/Emotional/Erratic"
| Disorder | Key Features | Treatment Highlights |
|---|---|---|
| Antisocial PD | Disregard for and violation of others' rights; deceitfulness, impulsivity, irritability/aggressiveness, reckless disregard for safety, irresponsibility, lack of remorse; must be ≥18 with history of conduct disorder before 15 | No established pharmacotherapy; CBT programs in forensic settings; comorbid substance use treatment critical |
| Borderline PD | Instability in relationships, self-image, affects; marked impulsivity; frantic efforts to avoid abandonment; unstable/intense relationships (idealization/devaluation—"splitting"); identity disturbance; chronic emptiness; affective instability; transient stress-related paranoid ideation or dissociation; recurrent suicidal behavior/self-harm | DBT (dialectical behavior therapy) is the gold standard—reduces self-harm, hospitalizations, and suicidality; mentalization-based therapy (MBT) also effective; transference-focused psychotherapy (TFP); no FDA-approved medications, but mood stabilizers, SSRIs, and low-dose antipsychotics used symptomatically |
| Histrionic PD | Excessive emotionality and attention-seeking; discomfort when not the center of attention; inappropriate sexually seductive behavior; rapidly shifting/shallow emotions; uses physical appearance to draw attention; impressionistic, vague speech; considers relationships more intimate than they are | Psychodynamic psychotherapy; group therapy helpful |
| Narcissistic PD | Grandiosity, need for admiration, lack of empathy; entitled; exploitative; envious; arrogant; believes self special/unique; requires excessive admiration; preoccupied with fantasies of unlimited success, power, brilliance | Psychotherapy (but engagement is difficult due to poor insight); address narcissistic injury/vulnerability |
Cluster C — "Anxious/Fearful"
| Disorder | Key Features | Treatment |
|---|---|---|
| Avoidant PD | Social inhibition, feelings of inadequacy, hypersensitivity to negative evaluation; avoids activities involving interpersonal contact due to fear of criticism/rejection; views self as socially inept, unappealing | CBT; social skills training; SSRIs for comorbid anxiety. Key distinction: avoidant PD WANTS relationships (vs. schizoid who does not) |
| Dependent PD | Excessive need to be taken care of; submissive, clinging behavior; difficulty making decisions without reassurance; difficulty expressing disagreement; difficulty initiating projects independently; goes to excessive lengths to obtain nurturance | CBT focusing on assertiveness training and self-efficacy; psychodynamic therapy |
| Obsessive-Compulsive PD (OCPD) | Preoccupation with orderliness, perfectionism, and control; rigid, inflexible; devoted to work to exclusion of leisure; unable to delegate; miserly spending; hoards worthless objects; overconscientious about morality/ethics | CBT; psychodynamic therapy. OCPD is ego-syntonic (vs. OCD which is ego-dystonic). OCPD does NOT have true obsessions or compulsions |
General Diagnostic Approach to Personality Disorders
DSM-5 requires all of the following: (A) Enduring pattern of inner experience and behavior deviating from cultural expectations in ≥2 of: cognition, affectivity, interpersonal functioning, impulse control. (B) Pattern is inflexible and pervasive across personal and social situations. (C) Leads to clinically significant distress or impairment. (D) Pattern is stable and of long duration, traceable to adolescence or early adulthood. (E) Not better explained by another mental disorder. (F) Not attributable to substance or medical condition. Personality disorders are highly comorbid with each other and with Axis I disorders (MDD, anxiety, substance use). Prevalence: ~10–15% of the general population; much higher in clinical settings (~40–60%).
Dialectical Behavior Therapy (DBT) developed by Marsha Linehan. Four modules: (1) Mindfulness—core skill; observing and describing present experience non-judgmentally. (2) Distress tolerance—crisis survival skills (TIPP: Temperature change, Intense exercise, Paced breathing, Progressive relaxation). (3) Emotion regulation—identifying and labeling emotions, reducing vulnerability (PLEASE skills: treat PhysicaL illness, balanced Eating, avoid mood-Altering substances, balanced Sleep, Exercise). (4) Interpersonal effectiveness—assertiveness and relationship skills (DEAR MAN: Describe, Express, Assert, Reinforce, Mindful, Appear confident, Negotiate). Standard DBT includes weekly individual therapy, weekly skills group, phone coaching, and therapist consultation team. RCTs demonstrate 50% reduction in self-harm, 73% reduction in psychiatric hospitalizations, and improved global functioning.
17 Somatic Symptom & Related Disorders
Somatic Symptom Disorder
One or more somatic symptoms that are distressing or result in significant disruption of daily life. Excessive thoughts, feelings, or behaviors related to the symptoms: (1) disproportionate and persistent thoughts about severity, (2) persistently high level of anxiety about health or symptoms, (3) excessive time and energy devoted to the symptoms. Although any one symptom may not be continuously present, the state of being symptomatic is persistent (≥6 months). Replaced DSM-IV somatization disorder and removed the requirement that symptoms be "medically unexplained"—the focus is on the excessive response to symptoms.
Illness Anxiety Disorder
Preoccupation with having or acquiring a serious illness. Somatic symptoms are NOT present or are only mild. High level of anxiety about health, and the individual is easily alarmed about personal health status. Performs excessive health-related behaviors (repeatedly checking body, frequent doctor visits) OR exhibits maladaptive avoidance (avoids appointments, hospitals). Specify: care-seeking type vs. care-avoidant type. Duration ≥6 months. Replaced DSM-IV hypochondriasis.
Conversion Disorder (Functional Neurological Symptom Disorder)
One or more symptoms of altered voluntary motor or sensory function. Clinical findings provide evidence of incompatibility between the symptom and recognized neurological/medical conditions. Positive neurological signs: Hoover sign (involuntary hip extension of the "weak" leg when flexing the contralateral hip against resistance), tremor entrainment (functional tremor changes frequency when contralateral hand performs rhythmic movements), give-way weakness (sudden collapse of resistance during exam), tubular visual field defects (non-physiologic—true field defects expand with distance). Renamed "functional neurological symptom disorder" to reduce stigma and emphasize positive diagnostic signs rather than diagnosis of exclusion.
| Symptom Type | Positive Diagnostic Signs |
|---|---|
| Functional weakness | Hoover sign, hip abductor sign, drift without pronation, collapsing/give-way pattern |
| Functional tremor | Entrainment (changes frequency to match examiner's rhythm), variability in frequency/amplitude, distractibility |
| Functional gait | Dragging monoplegic gait, astasia-abasia (wildly swaying without falling), excessive slowness inconsistent with neurological deficit |
| Functional seizures (PNES) | Preserved awareness with bilateral convulsions, eyes closed during event (epileptic seizures: eyes open), pelvic thrusting, side-to-side head movements, prolonged duration (>2 min), waxing/waning intensity, normal postictal EEG, no postictal confusion |
| Functional sensory | Midline splitting (hemisensory loss exactly at midline—anatomically impossible), non-dermatomal distribution, splitting of vibration sense on skull |
Treatment: validate symptoms as real and distressing, explain diagnosis positively (brain "software" problem, not "hardware"), physiotherapy for motor symptoms, CBT for psychological factors. Prognosis: ~50% improve within 1 year, better if acute onset, identifiable precipitant, and early diagnosis.
Factitious Disorder
Factitious disorder imposed on self (Munchausen syndrome) — falsification of physical or psychological signs/symptoms or induction of illness; presents self to others as ill/injured; deceptive behavior evident even in absence of obvious external rewards; motivation is to assume the sick role. Factitious disorder imposed on another (Munchausen by proxy) — falsification of symptoms in another person (typically a child); the perpetrator, not the victim, receives the diagnosis; constitutes child abuse and mandates reporting. Distinguished from malingering: malingering has clear external incentive (financial gain, avoiding work/legal consequences, obtaining medications) and is NOT a psychiatric diagnosis—it is a V-code/Z-code.
Distinguishing Somatic Spectrum Conditions
| Condition | Symptoms Real? | Intentional Production? | Motivation |
|---|---|---|---|
| Somatic symptom disorder | Yes (but excessive response) | No | Unconscious |
| Illness anxiety disorder | Absent or minimal | No | Unconscious (fear of disease) |
| Conversion disorder | Real neurological symptoms | No | Unconscious (psychological conflict) |
| Factitious disorder | Fabricated or induced | Yes | Internal (sick role) |
| Malingering | Fabricated or exaggerated | Yes | External (financial, legal gain) |
18 Dissociative Disorders
Dissociative Identity Disorder (DID)
Disruption of identity characterized by two or more distinct personality states (in some cultures described as an experience of possession). Marked discontinuity in sense of self and agency, accompanied by alterations in affect, behavior, consciousness, memory, perception, cognition, and/or sensory-motor functioning. Recurrent gaps in recall of everyday events, important personal information, and/or traumatic events inconsistent with ordinary forgetting. Strongly associated with severe childhood trauma (physical/sexual abuse in ~90% of cases). Treatment: phase-oriented therapy—(1) stabilization and symptom reduction, (2) trauma processing, (3) integration/rehabilitation. No FDA-approved medications; treat comorbid depression, anxiety, PTSD symptomatically.
Dissociative Amnesia
Inability to recall important autobiographical information, usually of a traumatic or stressful nature, inconsistent with ordinary forgetting. Types: localized (inability to recall events during a specific time period—most common), selective (inability to recall some but not all events during a period), generalized (complete loss of identity and life history—rare). Specify: with dissociative fugue — apparently purposeful travel or bewildered wandering associated with amnesia for identity or other important autobiographical information.
Depersonalization/Derealization Disorder
Persistent or recurrent experiences of depersonalization (feeling detached from one's mental processes or body, as if one is an outside observer of oneself) and/or derealization (experiencing surroundings as unreal, dreamlike, distant, or distorted). Reality testing remains intact during the experience (distinguishes from psychosis). Onset often late adolescence; can be triggered by severe stress, cannabis, hallucinogens. Treatment: CBT for dissociation, grounding techniques; SSRIs for comorbid anxiety/depression.
Dissociative symptoms can mimic psychotic presentations. Key distinctions: (1) Auditory hallucinations in DID are often experienced as internal voices (inside the head), whereas in schizophrenia they are typically external (from outside). (2) DID patients often have awareness that the voices are not "normal" (preserved insight vs. psychosis). (3) Dissociative patients typically have a trauma history and may exhibit sudden shifts in identity, behavior, and handwriting. (4) Depersonalization/derealization may superficially resemble negative symptoms of schizophrenia but insight is preserved. (5) Thought disorder (formal thought disorder with loose associations) is characteristic of schizophrenia and is not a feature of dissociative disorders. (6) The Dissociative Experiences Scale (DES) is a screening tool; scores ≥30 are highly suggestive of a dissociative disorder.
19 Antidepressants — Complete Drug Tables
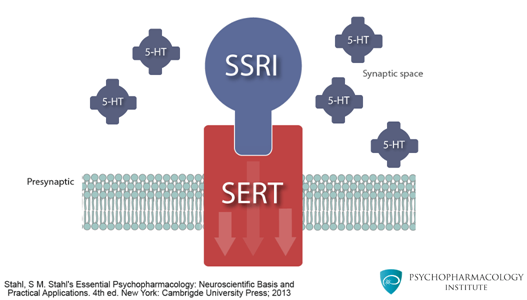
Selective Serotonin Reuptake Inhibitors (SSRIs)
| Drug | Brand | Start Dose | Target Dose | Key Pearls |
|---|---|---|---|---|
| Fluoxetine | Prozac | 20 mg | 20–80 mg | Longest half-life (2–6 days; norfluoxetine 4–16 days)—self-tapering, least discontinuation syndrome; CYP2D6 inhibitor; FDA-approved for OCD, bulimia |
| Sertraline | Zoloft | 50 mg | 50–200 mg | Mild dopamine reuptake inhibition; safest in cardiac patients (SADHART trial); FDA-approved for PTSD, OCD, panic, social anxiety |
| Paroxetine | Paxil | 20 mg | 20–50 mg | Most anticholinergic SSRI; most weight gain; worst discontinuation syndrome (short half-life ~21 hr); strong CYP2D6 inhibitor; avoid in pregnancy (cardiac defects) |
| Citalopram | Celexa | 20 mg | 20–40 mg | FDA max 40 mg due to dose-dependent QTc prolongation; max 20 mg in age >60, hepatic impairment, or CYP2C19 poor metabolizers |
| Escitalopram | Lexapro | 10 mg | 10–20 mg | S-enantiomer of citalopram; fewest drug interactions among SSRIs; generally well tolerated |
| Fluvoxamine | Luvox | 50 mg | 100–300 mg | FDA-approved only for OCD; potent CYP1A2 inhibitor (raises clozapine, theophylline, caffeine levels); sigma-1 receptor agonist |
Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs)
| Drug | Brand | Start Dose | Target Dose | Key Pearls |
|---|---|---|---|---|
| Venlafaxine | Effexor XR | 37.5–75 mg | 75–225 mg | Primarily serotonergic at low doses; NE reuptake inhibition begins at ≥150 mg; dose-dependent hypertension; significant discontinuation syndrome |
| Duloxetine | Cymbalta | 30 mg | 60–120 mg | FDA-approved for MDD, GAD, fibromyalgia, diabetic neuropathy, chronic musculoskeletal pain; avoid in hepatic impairment and heavy alcohol use |
| Desvenlafaxine | Pristiq | 50 mg | 50 mg | Active metabolite of venlafaxine; fewer drug interactions (minimal CYP metabolism); flat dose-response (no benefit >50 mg in trials) |
| Levomilnacipran | Fetzima | 20 mg | 40–120 mg | Greater NE:5-HT selectivity (~2:1); may help fatigue and psychomotor retardation |
Atypical Antidepressants
| Drug | Mechanism | Dose | Key Pearls |
|---|---|---|---|
| Bupropion (Wellbutrin) | NDRI (NE and DA reuptake inhibitor) | 150–450 mg | No sexual dysfunction; no weight gain (may aid weight loss); lowers seizure threshold (contraindicated in seizure disorder, bulimia, anorexia); used for smoking cessation (Zyban); activating—insomnia, anxiety possible |
| Mirtazapine (Remeron) | Alpha-2 antagonist, 5-HT2A/2C/3 antagonist, H1 antagonist | 15–45 mg | Sedating and appetite-stimulating (useful in underweight/insomnia); paradoxically MORE sedating at 15 mg than 30–45 mg (H1 antagonism predominates at low dose; NE activation increases at higher doses); "California rocket fuel" = mirtazapine + venlafaxine |
| Trazodone (Desyrel) | SARI (serotonin antagonist/reuptake inhibitor) | 50–400 mg | Commonly used at low dose (25–100 mg) for insomnia rather than as antidepressant; priapism risk (~1/10,000); orthostatic hypotension |
| Vilazodone (Viibryd) | SSRI + 5-HT1A partial agonist | 10–40 mg | Take with food for absorption; may have less sexual dysfunction than pure SSRIs |
| Vortioxetine (Trintellix) | Multimodal: SERT inhibitor, 5-HT3/7 antagonist, 5-HT1A agonist, 5-HT1B partial agonist | 5–20 mg | Procognitive effects (improves processing speed, executive function); nausea is most common side effect |
Tricyclic Antidepressants (TCAs)
| Drug | Start Dose | Target Dose | Relative Selectivity | Key Pearls |
|---|---|---|---|---|
| Amitriptyline | 25 mg | 150–300 mg | 5-HT > NE | Most anticholinergic; used for migraine prophylaxis, neuropathic pain, fibromyalgia |
| Nortriptyline | 25 mg | 75–150 mg | NE > 5-HT | Therapeutic window 50–150 ng/mL; less orthostasis than other TCAs; preferred TCA in elderly |
| Imipramine | 25 mg | 150–300 mg | 5-HT = NE | First TCA; used for enuresis in children; desmethylated to desipramine |
| Desipramine | 25 mg | 150–300 mg | NE >> 5-HT | Most noradrenergic TCA; least sedating/anticholinergic; highest risk of cardiac arrhythmia in overdose |
| Clomipramine | 25 mg | 150–250 mg | 5-HT >> NE | Most serotonergic TCA; most effective single agent for OCD; seizure risk at higher doses |
All TCAs share a side effect profile: anticholinergic effects (dry mouth, constipation, urinary retention, blurred vision), orthostatic hypotension (alpha-1 blockade), sedation (H1 blockade), weight gain, cardiac conduction delay (QRS widening—lethal in overdose via ventricular arrhythmia). Therapeutic drug monitoring available for nortriptyline (50–150 ng/mL) and desipramine (>115 ng/mL). ECG before initiation if cardiac risk factors.
Monoamine Oxidase Inhibitors (MAOIs)
| Drug | Type | Dose | Key Pearls |
|---|---|---|---|
| Phenelzine (Nardil) | Non-selective, irreversible | 45–90 mg | Most studied MAOI; highly effective for atypical depression, treatment-resistant depression, social anxiety |
| Tranylcypromine (Parnate) | Non-selective, irreversible | 30–60 mg | More activating than phenelzine; structurally related to amphetamine |
| Selegiline patch (Emsam) | Selective MAO-B at low dose; non-selective at higher doses | 6–12 mg/24hr patch | At 6 mg/24hr: no dietary restrictions needed (selective MAO-B); at 9–12 mg: tyramine diet required |
Tyramine reaction ("hypertensive crisis"): Foods high in tyramine (aged cheese, cured meats, tap/draft beer, fermented soy, red wine) combined with MAOIs cause massive NE release—severe hypertension, headache, stroke risk. Treatment: phentolamine IV or nifedipine SL. Washout periods: 2 weeks when switching from MAOI to serotonergic drug (or vice versa); 5 weeks for fluoxetine to MAOI (due to long half-life of norfluoxetine).
Cause: Excess serotonergic activity, usually from drug combinations (SSRI + MAOI, SSRI + tramadol, SSRI + linezolid, SSRI + triptans, MDMA). Hunter Criteria (most sensitive/specific): In the presence of a serotonergic agent, ONE of: (1) spontaneous clonus, (2) inducible clonus + agitation or diaphoresis, (3) ocular clonus + agitation or diaphoresis, (4) tremor + hyperreflexia, (5) hypertonia + temperature >38°C + ocular or inducible clonus. Treatment: Discontinue offending agents; benzodiazepines for agitation; cyproheptadine (5-HT2A antagonist) 12 mg initial then 4 mg q2h; cooling measures for hyperthermia; avoid dantrolene. Distinguish from NMS: Serotonin syndrome has clonus/hyperreflexia/myoclonus; NMS has rigidity ("lead pipe")/hyporeflexia/bradykinesia.
20 Antipsychotics — FGAs & SGAs
First-Generation Antipsychotics (FGAs / Typicals)
| Drug | Potency | Dose Range | EPS Risk | Sedation | Key Pearls |
|---|---|---|---|---|---|
| Haloperidol (Haldol) | High | 2–20 mg | High | Low | IM/IV available; QTc prolongation (especially IV); most commonly used FGA; decanoate (LAI) available q4wk |
| Fluphenazine | High | 2.5–20 mg | High | Low | Decanoate (LAI) available q2–4wk |
| Perphenazine | Mid | 4–64 mg | Moderate | Moderate | CATIE trial showed comparable efficacy to SGAs |
| Chlorpromazine (Thorazine) | Low | 200–800 mg | Low | High | First antipsychotic (1952); high anticholinergic, orthostatic hypotension; photosensitivity; "chlorpromazine equivalent" used for dose comparison |
| Thioridazine | Low | 200–800 mg | Low | High | QTc prolongation risk highest among FGAs; retinitis pigmentosa at high doses (>800 mg); restricted use |
Second-Generation Antipsychotics (SGAs / Atypicals)
| Drug (Brand) | Dose Range | Weight Gain | Metabolic Risk | EPS | QTc | Key Pearls |
|---|---|---|---|---|---|---|
| Risperidone (Risperdal) | 1–6 mg | Moderate | Moderate | Dose-dependent (higher at >6 mg) | Low | Highest prolactin elevation among SGAs; LAI (Risperdal Consta) q2wk; paliperidone is active metabolite |
| Olanzapine (Zyprexa) | 5–20 mg | High | High | Low | Low | Highest weight gain and metabolic risk; highly effective for acute mania; Zyprexa Relprevv (LAI) requires 3-hr post-injection monitoring (PDSS risk) |
| Quetiapine (Seroquel) | 150–800 mg | Moderate | Moderate | Very low | Low | Very sedating (H1 antagonism); used off-label for insomnia at 25–100 mg; FDA-approved for bipolar depression (300 mg) and MDD augmentation |
| Aripiprazole (Abilify) | 5–30 mg | Low | Low | Akathisia common | Low | D2 partial agonist—stabilizes dopamine ("Goldilocks" effect); LAI (Abilify Maintena) monthly or Aristada q4–8wk |
| Ziprasidone (Geodon) | 40–160 mg | Low | Low | Low-moderate | Moderate | Must take with ≥500 kcal food for absorption; IM formulation for acute agitation; weight-neutral |
| Lurasidone (Latuda) | 20–160 mg | Low | Low | Moderate | Low | Must take with ≥350 kcal food; FDA-approved for bipolar depression; weight-neutral |
| Cariprazine (Vraylar) | 1.5–6 mg | Low | Low | Moderate (akathisia) | Low | D3-preferring partial agonist; very long half-life (active metabolites 2–4 weeks); may improve negative symptoms |
| Brexpiprazole (Rexulti) | 1–4 mg | Low | Low | Low | Low | D2 partial agonist like aripiprazole but less akathisia; FDA-approved for MDD augmentation and schizophrenia |
| Clozapine (Clozaril) | 150–900 mg | High | High | Very low | Moderate | Gold standard for treatment-resistant schizophrenia; anti-suicide; REMS monitoring for agranulocytosis; seizure risk (dose-dependent, >600 mg); myocarditis risk (first month); sialorrhea, sedation, constipation (can cause ileus) |
| Pimozide | 1–10 mg | Low | Low | Moderate | High | FDA-approved for Tourette syndrome; CYP3A4/2D6 substrate; significant QTc risk—requires baseline ECG |

Extrapyramidal Symptoms (EPS)
| Type | Onset | Features | Treatment |
|---|---|---|---|
| Acute dystonia | Hours to days | Sustained muscle contraction (torticollis, oculogyric crisis, opisthotonus, laryngospasm) | Benztropine 1–2 mg IM/IV or diphenhydramine 50 mg IM/IV—rapid relief |
| Akathisia | Days to weeks | Subjective restlessness, inability to sit still, pacing | Reduce dose; propranolol 20–80 mg/day; benzodiazepine; mirtazapine; switch antipsychotic |
| Parkinsonism (drug-induced) | Weeks to months | Bradykinesia, rigidity, tremor, masked facies, shuffling gait | Benztropine 1–2 mg BID; trihexyphenidyl; amantadine 100 mg BID |
| Tardive dyskinesia | Months to years | Involuntary choreoathetoid movements of face/tongue/lips (lip smacking, tongue protrusion, chewing), extremities, trunk | Valbenazine (Ingrezza) 40–80 mg/day or deutetrabenazine (Austedo) 6–48 mg/day (VMAT2 inhibitors); consider switching to clozapine or quetiapine (lowest TD risk) |
21 Mood Stabilizers
Lithium
The oldest and best-studied mood stabilizer. Mechanism: inhibits inositol monophosphatase (depleting IP3 signaling), inhibits GSK-3beta, enhances serotonergic transmission, neuroprotective (increases gray matter volume). Uniquely reduces suicide risk.
Therapeutic levels: Acute mania 0.8–1.2 mEq/L; maintenance 0.6–1.0 mEq/L; elderly 0.4–0.8 mEq/L. Starting dose: 300 mg BID-TID; adjust based on levels drawn 12 hours post-dose (trough), steady state at ~5 days. Side effects: Fine tremor (treat with propranolol 20–40 mg), polyuria/polydipsia (nephrogenic diabetes insipidus—40% of long-term users; amiloride can help), hypothyroidism (20–30%—treat with levothyroxine, do not discontinue lithium), weight gain, GI upset, cognitive dulling, acne, psoriasis exacerbation. Toxicity signs by level: 1.5–2.0: coarse tremor, nausea, vomiting, diarrhea, ataxia, blurred vision; 2.0–2.5: confusion, stupor, fasciculations, hyperreflexia; >2.5: seizures, coma, renal failure, cardiac arrhythmia, death. Monitoring: Lithium level, BUN/Cr, TSH, calcium q6 months; ECG at baseline (can cause T-wave flattening, Brugada-like pattern). Drug interactions that increase levels: NSAIDs, ACE inhibitors, ARBs, thiazide diuretics, dehydration, metronidazole. Pregnancy: Ebstein anomaly risk (tricuspid valve malformation)—0.05–0.1% (lower than historically stated, but still above baseline).
Valproate (Divalproex Sodium / Valproic Acid)
Mechanism: Enhances GABA (inhibits GABA transaminase), blocks voltage-gated sodium channels, inhibits histone deacetylase. Therapeutic level: 50–125 mcg/mL (acute mania target 80–120). Loading strategy for acute mania: 20–30 mg/kg/day in divided doses. Side effects: GI upset, weight gain, tremor, hair loss (supplemental zinc and selenium may help), thrombocytopenia (dose-dependent), elevated ammonia (can cause encephalopathy even with normal levels—check ammonia if altered mental status), hepatotoxicity (LFT monitoring, contraindicated in hepatic disease), pancreatitis (rare but serious). Teratogenicity: Neural tube defects (1–2% risk, highest of any mood stabilizer), craniofacial anomalies, reduced IQ in exposed children. Absolutely contraindicated in pregnancy and women of childbearing potential without reliable contraception. Monitoring: VPA level, CBC, LFTs q6 months; ammonia if symptomatic.
Lithium Toxicity Management
Acute ingestion vs. chronic accumulation: chronic toxicity occurs at lower levels due to higher brain concentrations. Management: (1) Stop lithium immediately. (2) Aggressive IV normal saline hydration (200–300 mL/hr) to enhance renal clearance. (3) Avoid diuretics (loop diuretics are acceptable; thiazides are contraindicated). (4) Monitor serum lithium levels every 2–4 hours. (5) Hemodialysis indications: level >4.0 mEq/L, level >2.5 with severe symptoms (AMS, seizures), level >2.5 with renal failure, or clinical deterioration despite supportive care. Post-dialysis rebound can occur due to redistribution—repeat level 6 hours after dialysis and consider repeat dialysis if level rises. (6) Whole bowel irrigation with polyethylene glycol for acute ingestion of sustained-release formulations. Prevention: patient education about maintaining hydration, avoiding NSAIDs and new medications without checking interactions, and recognizing early toxicity symptoms.
Carbamazepine
Mechanism: Voltage-gated sodium channel blocker. Therapeutic level: 4–12 mcg/mL. Side effects: Dizziness, ataxia, diplopia, hyponatremia (SIADH), aplastic anemia (1/200,000), agranulocytosis, hepatotoxicity, Stevens-Johnson syndrome (HLA-B*1502 testing mandatory before initiation in patients of Asian ancestry). Drug interactions: Potent CYP3A4 inducer—reduces levels of oral contraceptives, warfarin, other anticonvulsants, many medications. Also auto-induces its own metabolism (levels drop after 3–4 weeks; recheck and adjust). Monitoring: CBZ level, CBC, LFTs, sodium q6 months; HLA-B*1502 before starting.
Lamotrigine
Mechanism: Voltage-gated sodium channel blocker; inhibits glutamate release. Primary role: Bipolar depression (prevention and acute) and maintenance. NOT effective for acute mania. Titration (critical): Must be slow to avoid SJS/TEN. Without valproate: 25 mg/day x 2 weeks → 50 mg/day x 2 weeks → 100 mg/day x 1 week → target 200 mg/day. WITH valproate (which doubles lamotrigine levels): 25 mg every other day x 2 weeks → 25 mg daily x 2 weeks → 50 mg/day x 1 week → target 100 mg/day. With carbamazepine (which halves lamotrigine levels): faster titration, target 300–400 mg/day. SJS/TEN risk: ~0.08% (higher with rapid titration, valproate co-administration, and pediatric use). Discontinue immediately at first sign of rash—though most rashes are benign, cannot distinguish clinically. If discontinued >5 days, must restart from beginning of titration.
22 Anxiolytics, Hypnotics & Stimulants
Benzodiazepine Equivalency Table
| Drug | Equivalent Dose | Half-Life | Onset | Primary Use |
|---|---|---|---|---|
| Alprazolam (Xanax) | 0.5 mg | 6–12 hr | Fast | Panic, GAD |
| Lorazepam (Ativan) | 1 mg | 10–20 hr | Intermediate | Anxiety, catatonia, status epilepticus, alcohol withdrawal |
| Clonazepam (Klonopin) | 0.25–0.5 mg | 18–50 hr | Intermediate | Panic, seizure, anxiety |
| Diazepam (Valium) | 5 mg | 20–100 hr (active metabolites) | Fast | Anxiety, muscle spasm, alcohol withdrawal, seizure |
| Chlordiazepoxide (Librium) | 25 mg | 24–48 hr | Intermediate | Alcohol withdrawal (CIWA protocol) |
| Oxazepam (Serax) | 15 mg | 4–15 hr | Slow | Anxiety; preferred in hepatic impairment (no active metabolites, glucuronidation only—"LOT" = Lorazepam, Oxazepam, Temazepam) |
| Temazepam (Restoril) | 15 mg | 8–22 hr | Intermediate | Insomnia |
| Midazolam (Versed) | 1.5–2 mg | 1.5–2.5 hr | Very fast | Procedural sedation, status epilepticus (intranasal/IM) |
LOT drugs (Lorazepam, Oxazepam, Temazepam) are preferred in hepatic impairment because they undergo only glucuronidation (Phase II metabolism) without Phase I oxidation—no active metabolites and no CYP dependence.
Z-Drugs (Non-Benzodiazepine Hypnotics)
| Drug | Dose | Half-Life | Key Pearls |
|---|---|---|---|
| Zolpidem (Ambien) | 5–10 mg (5 mg in women per FDA) | 2.5 hr | Selective BZ1/GABA-A alpha-1 agonist; parasomnias (sleepwalking, sleep-driving, sleep-eating); lower dose in women due to slower metabolism |
| Zaleplon (Sonata) | 5–10 mg | 1 hr | Ultra-short acting; good for sleep-onset insomnia; can redose if ≥4 hr before wake time |
| Eszopiclone (Lunesta) | 1–3 mg | 6 hr | Longest Z-drug half-life; can help with sleep maintenance; metallic taste |
Stimulants for ADHD
| Drug | Dose Range | Duration | Key Pearls |
|---|---|---|---|
| Methylphenidate IR (Ritalin) | 5–60 mg/day in divided doses | 3–4 hr | Blocks DA and NE reuptake; first-line for ADHD |
| Methylphenidate ER (Concerta) | 18–72 mg/day | 10–12 hr | OROS delivery system; non-crushable |
| Mixed amphetamine salts (Adderall) | 5–40 mg/day in divided doses | 4–6 hr | Blocks reuptake AND promotes DA/NE release; Schedule II |
| Lisdexamfetamine (Vyvanse) | 20–70 mg/day | 10–14 hr | Prodrug (requires enzymatic cleavage to active d-amphetamine); reduced abuse potential; also FDA-approved for binge eating disorder |
| Atomoxetine (Strattera) | 40–100 mg/day | 24 hr | NRI (not a stimulant); non-controlled substance; takes 4–6 weeks for full effect; black box warning for suicidal ideation in children |
| Guanfacine ER (Intuniv) | 1–4 mg/day | 24 hr | Alpha-2A agonist; non-stimulant; also used for PTSD hyperarousal and tics |
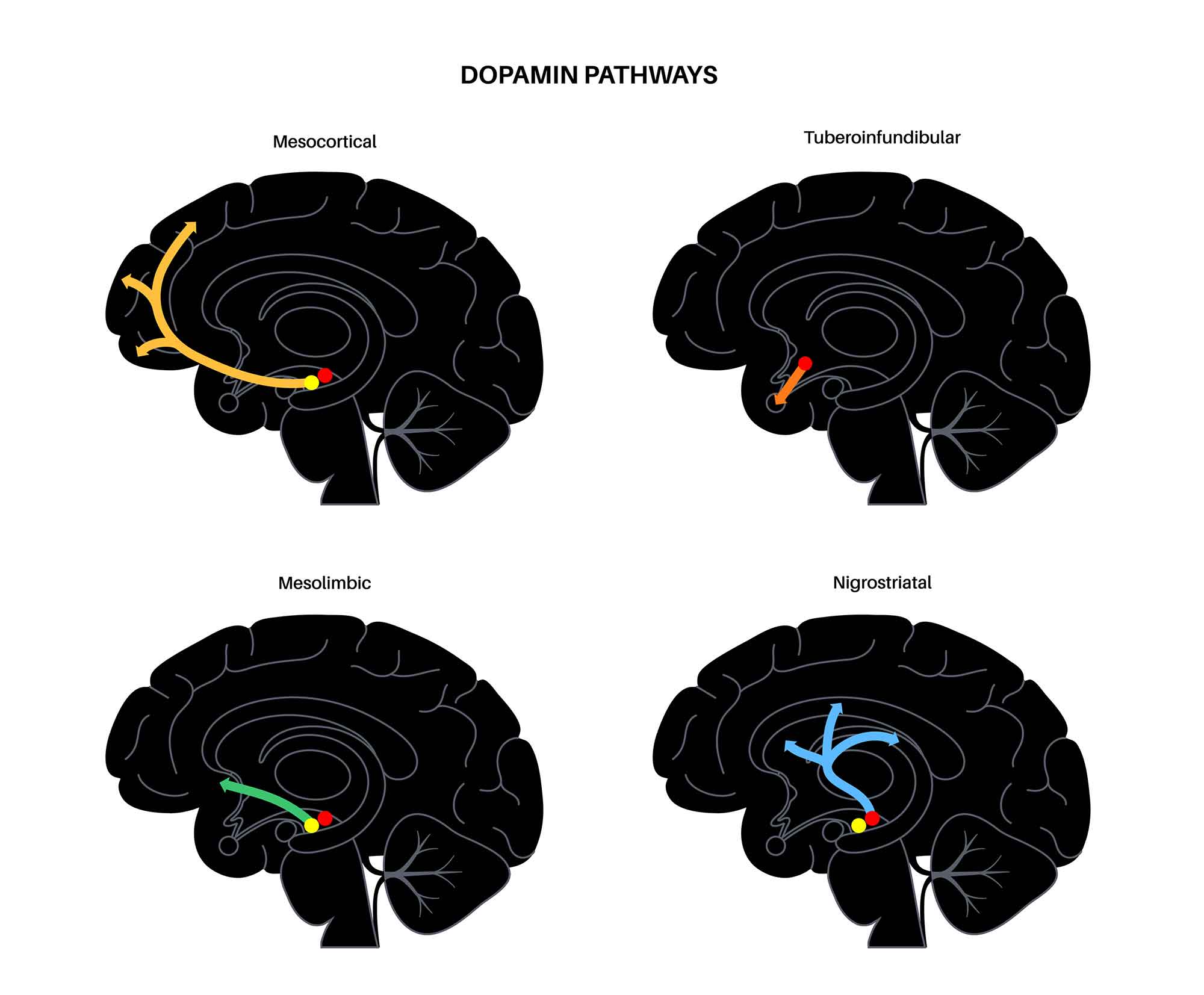
23 Psychopharmacology in Pregnancy & Lactation
The FDA replaced pregnancy letter categories (A/B/C/D/X) with the Pregnancy and Lactation Labeling Rule (PLLR) in 2015, requiring descriptive summaries of human and animal data. Clinical decisions require balancing teratogenic risk against the risk of untreated psychiatric illness (which itself carries risks: poor prenatal care, substance use, preeclampsia, preterm birth, suicide).
Medications to Avoid in Pregnancy
| Drug | Risk | Timing of Concern |
|---|---|---|
| Valproate | Neural tube defects (1–2%), craniofacial anomalies, reduced IQ (~8–9 points), autism risk | First trimester (organogenesis); cognitive effects across pregnancy |
| Carbamazepine | Neural tube defects (0.5–1%), craniofacial anomalies (less than valproate) | First trimester |
| Paroxetine | Cardiac malformations (OR ~1.5–2.0, particularly VSD and ASD) | First trimester |
| Lithium | Ebstein anomaly (0.05–0.1%—lower than previously estimated); neonatal toxicity, floppy infant | First trimester (cardiac); delivery (toxicity) |
| Benzodiazepines | Possible cleft palate risk (controversial, OR ~1.1); neonatal sedation, withdrawal, floppy infant | First trimester; third trimester/delivery |
Relatively Safer Options
Depression: Sertraline (most safety data in pregnancy), fluoxetine, citalopram. All SSRIs carry a small risk of persistent pulmonary hypertension of the newborn (PPHN) and neonatal adaptation syndrome (jitteriness, irritability, feeding difficulty) if used in third trimester. Bipolar disorder: Lamotrigine (no clear teratogenic signal; levels decrease 40–60% during pregnancy—monitor and increase dose); second-generation antipsychotics (quetiapine, olanzapine—metabolic monitoring). Psychosis: Haloperidol (extensive safety data), olanzapine, quetiapine. Anxiety: SSRIs preferred; hydroxyzine for acute anxiety; minimize benzodiazepine use. Insomnia: Diphenhydramine, doxepin (low dose).
Lactation Considerations
Sertraline and paroxetine have the lowest breast milk concentrations among SSRIs. Lamotrigine levels in breast milk are significant (~50% of maternal serum)—monitor infant for rash and sedation. Lithium is generally avoided in breastfeeding (infant levels can reach 50% of maternal levels; risk of dehydration-induced toxicity). Valproate and carbamazepine are considered relatively compatible with breastfeeding. All benzodiazepines transfer into breast milk—short-acting agents preferred if necessary.
24 ECT, TMS & Other Neuromodulation
Electroconvulsive Therapy (ECT)
Indications: Treatment-resistant depression (failed ≥2 adequate medication trials), psychotic depression (remission rate ~90%), catatonia (when lorazepam fails), severe suicidality requiring rapid response, bipolar depression/mania (refractory), schizoaffective disorder. Also used in pregnancy when medications pose unacceptable risk. Mechanism: Deliberate induction of a generalized seizure under general anesthesia (methohexital or propofol) with succinylcholine for muscle relaxation.
Electrode placement: Right unilateral (RUL, d'Elia) — fewer cognitive side effects, requires 6x seizure threshold dosing; bilateral (BL, bitemporal) — faster response, more cognitive effects; bifrontal — may balance efficacy and side effects. Pulse width: Brief pulse (0.5–2 ms) standard; ultrabrief pulse (0.3 ms) — significantly less cognitive impairment, preferred for RUL placement but may require more sessions. Seizure adequacy: EEG seizure duration ≥15–25 seconds (motor seizure ≥20 seconds); postictal suppression on EEG indicates adequate seizure. Treatment course: Typically 6–12 sessions, 2–3x/week. Maintenance ECT (weekly to monthly) to prevent relapse. Cognitive side effects: Acute postictal confusion (resolves minutes to hours); anterograde amnesia (usually resolves within weeks); retrograde amnesia (autobiographical events around treatment period—may be permanent); not associated with structural brain damage.
Absolute contraindication: Raised intracranial pressure (pheochromocytoma is a relative contraindication). There are NO absolute psychiatric contraindications. ECT is one of the safest procedures performed under general anesthesia—mortality rate ~2 per 100,000 treatments. Pre-ECT workup: Medical history and physical exam, anesthesia evaluation, CBC, BMP, ECG; brain imaging only if neurological signs suggest intracranial pathology. Consent must be obtained from the patient (or guardian if legally incompetent). Hold lithium on night before and morning of ECT (increased risk of postictal confusion and prolonged seizure). Benzodiazepines and anticonvulsants raise seizure threshold—taper or hold before ECT when possible. Caffeine may be used to augment seizure duration if seizures are inadequate.
Transcranial Magnetic Stimulation (TMS)
Repetitive TMS (rTMS): Electromagnetic coil placed over the scalp generates focused magnetic pulses that induce electrical currents in underlying cortex. FDA-cleared for: MDD (failed ≥1 antidepressant), OCD, smoking cessation, anxious depression. Standard protocol: High-frequency (10 Hz) stimulation of the left dlPFC for depression (excitatory); low-frequency (1 Hz) stimulation of the right dlPFC is an alternative. Treatment course: 5 days/week for 4–6 weeks (20–30 sessions), each session ~20–40 minutes. Theta burst stimulation (TBS): Intermittent TBS (iTBS) to left dlPFC achieves comparable results in ~3 minutes per session (Stanford SAINT protocol uses accelerated iTBS—10 sessions/day for 5 days with 90% remission in TRD). Side effects: Scalp discomfort at stimulation site (most common), headache; seizure risk very low (~0.01%); no systemic side effects (no anesthesia required). Contraindications: Ferromagnetic implants near the coil, cochlear implants, metallic hardware in the skull.
Other Neuromodulation
Vagus Nerve Stimulation (VNS): Implanted pulse generator in the chest stimulates the left vagus nerve via an electrode wrapped around the nerve in the neck. FDA-approved for TRD (failed ≥4 treatments). Slow onset (months to years); used as adjunct to medications. Deep Brain Stimulation (DBS): Investigational for TRD and severe OCD. Targets: subcallosal cingulate (Brodmann area 25), ventral capsule/ventral striatum, nucleus accumbens. Ketamine/Esketamine: Ketamine IV infusion (0.5 mg/kg over 40 minutes) produces rapid antidepressant effect (hours) lasting ~1 week; serial infusions for sustained response. Esketamine (Spravato) nasal spray—FDA-approved for TRD and MDD with acute suicidal ideation; administered under REMS (2-hour observation post-dose); given with oral antidepressant. Common side effects: dissociation, nausea, dizziness, headache, elevated BP.
25 Psychiatric Emergencies — Agitation, Catatonia, NMS, Serotonin Syndrome
Acute Agitation Management
De-escalation first: Verbal techniques—calm tone, non-threatening posture, offer choices ("Would you like to sit down?"), identify and address needs (pain, hunger, fear), maintain safe distance, avoid confrontation. Pharmacologic management (when de-escalation fails):
| Scenario | First-Line | Alternative |
|---|---|---|
| Undifferentiated agitation | Olanzapine 10 mg IM (onset 15–30 min) OR haloperidol 5 mg IM + lorazepam 2 mg IM + diphenhydramine 50 mg IM ("B-52": Benadryl 50 + haloperidol 5 + Ativan 2) | Ziprasidone 10–20 mg IM; midazolam 5 mg IM |
| Psychosis-related | Haloperidol 5 mg IM or olanzapine 10 mg IM | Droperidol 5 mg IM (very rapid onset) |
| Alcohol/sedative withdrawal | Lorazepam 2–4 mg IM/IV | Diazepam 10–20 mg IV; phenobarbital if refractory |
| Stimulant intoxication | Lorazepam 2–4 mg IM/IV (avoid antipsychotics—lower seizure threshold, worsen hyperthermia) | Midazolam 5 mg IM |
| Known bipolar mania | Olanzapine 10 mg IM or aripiprazole 9.75 mg IM | Haloperidol 5 mg IM + lorazepam 2 mg IM |
| Delirium (non-alcohol related) | Haloperidol 0.5–2 mg IV/IM (lowest effective dose; avoid in Lewy body dementia) | Quetiapine 25–50 mg PO; treat underlying cause |
| Dementia-related agitation | Non-pharmacologic interventions first; if severe: risperidone 0.25–0.5 mg or quetiapine 12.5–25 mg | FDA black box: antipsychotics increase mortality in elderly dementia patients (~1.6–1.7x) |
Physical restraint is used only as a last resort when less restrictive interventions have failed and there is imminent risk of harm. Requires physician order specifying type and duration (typically reassessed every 1–4 hours). Monitor vital signs, circulation, and mental status every 15 minutes. Restraint-associated risks include positional asphyxia, aspiration, DVT, and psychological trauma. Always document indication, alternatives attempted, monitoring plan, and patient's response. Release as soon as safely possible.
Catatonia
Catatonia is a psychomotor syndrome occurring in mood disorders, schizophrenia, medical conditions, and substance use. The Bush-Francis Catatonia Rating Scale (BFCRS) assesses 23 items. Key signs (3+ for diagnosis): stupor, catalepsy, waxy flexibility, mutism, negativism, posturing, mannerisms, stereotypy, agitation (not influenced by external stimuli), grimacing, echolalia, echopraxia, staring, autonomic instability (in malignant catatonia).
Lorazepam challenge: Administer lorazepam 1–2 mg IV; assess at 5–10 minutes. Marked improvement (resolution of ≥50% of catatonic signs) confirms the diagnosis and guides treatment. Treatment: lorazepam 1–2 mg IV/IM every 4–8 hours, titrate up to 8–24 mg/day. If lorazepam fails after 3–5 days, proceed to ECT (the most effective treatment for catatonia). Avoid antipsychotics in catatonia—may worsen symptoms and precipitate neuroleptic malignant syndrome (NMS), especially with high-potency FGAs.
NMS vs. Serotonin Syndrome Differential
| Feature | Neuroleptic Malignant Syndrome (NMS) | Serotonin Syndrome |
|---|---|---|
| Cause | Dopamine D2 blockade (antipsychotics, metoclopramide, withdrawal of dopaminergic agents) | Serotonergic excess (drug combinations: SSRI+MAOI, SSRI+tramadol) |
| Onset | Days to weeks (slow, insidious) | Hours (rapid, within 24 hours of drug change) |
| Temperature | Hyperthermia (>40°C possible) | Hyperthermia (usually <40°C unless severe) |
| Muscle tone | "Lead-pipe" rigidity (generalized) | Clonus, hyperreflexia, myoclonus |
| Reflexes | Hyporeflexia, bradykinesia | Hyperreflexia (lower extremity > upper) |
| Pupils | Normal | Dilated (mydriasis) |
| GI | Incontinence, ileus | Diarrhea, hyperactive bowel sounds |
| Labs | Elevated CK (often >1000 U/L), leukocytosis, elevated LFTs, myoglobinuria | CK may be mildly elevated |
| Treatment | Stop offending agent; dantrolene 1–2.5 mg/kg IV q6h (muscle relaxant); bromocriptine 2.5–10 mg TID (DA agonist); supportive care; ICU admission | Stop offending agent; cyproheptadine 12 mg load then 4 mg q2h; benzodiazepines; cooling measures |
| Mortality | 5–10% (if recognized early) | <1% (if recognized early) |
26 Forensic Psychiatry & Involuntary Commitment
Capacity vs. Competency
Capacity is a clinical determination made by any physician at the bedside. A patient has decision-making capacity if they can: (1) Understand the relevant information about the proposed treatment, (2) Appreciate how the information applies to their situation, (3) Reason about the options (weigh risks/benefits), (4) Communicate a choice consistently. Capacity is decision-specific (a patient may have capacity for one decision but not another) and can fluctuate. Competency is a legal determination made by a judge in court. An incompetent patient requires a court-appointed guardian to make decisions.
Involuntary Commitment
Criteria vary by jurisdiction but generally require: (1) Presence of a mental illness, AND (2) Danger to self (suicidal), danger to others (homicidal), or grave disability (unable to provide for basic needs—food, shelter, medical care). Most states allow 72-hour emergency psychiatric holds (e.g., "5150" in California, "Baker Act" in Florida, "302" in Pennsylvania) by designated professionals (physicians, police officers, psychiatric nurses depending on jurisdiction). Continued involuntary hospitalization beyond the emergency hold requires judicial review. Patients retain the right to legal counsel and hearing.
Right to Refuse Treatment
Involuntary commitment does NOT automatically override the right to refuse medication. In most jurisdictions, a separate judicial proceeding (often called a "medication over objection" hearing or "Rogers order") is required to administer medication involuntarily to a non-emergency patient. Exceptions: (1) Emergency exception—imminent danger to self or others permits short-term involuntary medication without court order. (2) Substituted judgment (some jurisdictions)—court determines what the patient would have chosen if competent. (3) Best interest standard (some jurisdictions)—court determines what treatment is in the patient's best interest. Informed consent for psychiatric treatment requires disclosure of diagnosis, proposed treatment, risks/benefits, alternatives, and consequences of refusal. Patient must demonstrate decision-making capacity (understanding, appreciation, reasoning, communication of a choice).
Tarasoff Duty
Tarasoff v. Regents of University of California (1976): When a therapist determines that a patient presents a serious danger of violence against a reasonably identifiable victim, the therapist has a duty to protect the intended victim. This may include warning the victim, notifying police, or taking other reasonable steps. Not adopted in all jurisdictions—some states require warning, others permit but do not mandate it, some have no Tarasoff-type statute. Does NOT apply to general threats without an identifiable victim.
Insanity Defense Standards
| Standard | Criteria | Jurisdiction |
|---|---|---|
| M'Naghten Rule (1843) | At the time of the act, by reason of mental disease or defect, the defendant did not know the nature and quality of the act, OR did not know that what they were doing was wrong | Most US states; UK; "cognitive" test only |
| ALI/Model Penal Code (1962) | As a result of mental disease or defect, the defendant lacked substantial capacity to appreciate the criminality (wrongfulness) of their conduct OR to conform their conduct to the requirements of law | Some US states; adds "volitional" component (irresistible impulse) |
| Durham Rule (1954) | The act was the "product of" mental disease or defect | Largely abandoned; only New Hampshire |
| Federal (Insanity Defense Reform Act, 1984) | At the time of the offense, as a result of severe mental disease or defect, unable to appreciate the nature and quality or wrongfulness of their acts | Federal courts; enacted after Hinckley verdict; eliminated volitional prong |
Additional Forensic Concepts
Competency to stand trial (CST) is the most commonly requested forensic evaluation. Based on Dusky v. United States (1960): defendant must have (1) a rational and factual understanding of the proceedings and (2) the ability to assist counsel in their own defense. CST is about current mental state (at time of trial), not mental state at time of offense (which is the insanity defense). If found incompetent, the defendant is treated (usually involuntarily) to restore competency; if unrestorable, charges may be dismissed or civil commitment pursued. Fitness for duty evaluations assess whether an individual can safely perform job functions. Psychiatric malpractice most commonly involves: failure to assess/prevent suicide, improper prescribing, sexual boundary violations, breach of confidentiality, and failure to obtain informed consent.
27 Sleep Disorders in Psychiatry
Insomnia Disorder
Difficulty initiating sleep, maintaining sleep, or early morning awakening with inability to return to sleep, despite adequate opportunity. ≥3 nights/week for ≥3 months, causing daytime impairment. Prevalence 6–10% (chronic insomnia). Comorbid with depression (75% of MDD patients have insomnia), anxiety, substance use, and chronic pain. First-line treatment is CBT for Insomnia (CBT-I)—more effective than medication long-term.
Sleep restriction: Limit time in bed to match actual sleep time (e.g., if sleeping 5 of 8 hours in bed, restrict to 5 hours); increase by 15–30 minutes when sleep efficiency exceeds 85%. Stimulus control: Bed is for sleep and sex only; go to bed only when sleepy; get out of bed if awake >20 minutes; wake at the same time daily regardless of sleep quality. Sleep hygiene: Regular sleep-wake schedule, avoid caffeine after noon, limit alcohol (disrupts sleep architecture), cool/dark/quiet environment, avoid screens 1 hour before bed, exercise regularly (but not within 3 hours of bedtime). Cognitive restructuring: Challenge catastrophic thoughts about sleep. Relaxation training: Progressive muscle relaxation, breathing techniques, mindfulness.
Circadian Rhythm Disorders
Delayed sleep-wake phase disorder: Habitual sleep-wake timing delayed ≥2 hours relative to desired or socially acceptable times. Common in adolescents. Treatment: morning bright light therapy (10,000 lux, 20–30 minutes upon awakening), low-dose melatonin 0.5–3 mg administered 5–7 hours before habitual sleep onset (not at bedtime), chronotherapy. Advanced sleep-wake phase disorder: Habitual sleep-wake timing advanced; more common in elderly. Treatment: evening bright light therapy, melatonin in the morning (less studied). Shift work disorder: Insomnia and/or excessive sleepiness associated with recurring work hours during the habitual sleep period. Treatment: strategic napping, bright light during work hours, modafinil 200 mg before shift.
Polysomnography Basics
Sleep stages: N1 (light sleep, theta waves, 4–5% of sleep), N2 (sleep spindles and K-complexes, 45–55%), N3 (slow-wave/deep sleep, delta waves, 15–25%), REM (rapid eye movements, muscle atonia, dreaming, 20–25%). Normal sleep latency: 10–20 minutes. Sleep efficiency (time asleep/time in bed): normal ≥85%. REM latency: normally 90 minutes; shortened REM latency seen in depression, narcolepsy, and alcohol/BZD withdrawal.
Sleep Disorders with Psychiatric Overlap
Obstructive sleep apnea (OSA) frequently complicates psychiatric illness. Prevalence is elevated in patients on SGAs (weight gain increases OSA risk). OSA causes daytime somnolence, cognitive impairment, and depressive symptoms that may be misattributed to psychiatric illness. Screen with STOP-BANG questionnaire (score ≥3 = high risk). Polysomnography confirms diagnosis; CPAP is first-line treatment. Treating OSA can significantly improve depressive and cognitive symptoms.
Restless legs syndrome (RLS) is exacerbated by SSRIs, SNRIs, mirtazapine, and antipsychotics (particularly due to dopamine blockade). Consider switching to bupropion (least likely to worsen RLS). Treat with iron supplementation if ferritin <75, gabapentin, or low-dose dopamine agonists. Nightmare disorder is highly comorbid with PTSD; prazosin 1–15 mg at bedtime is the primary pharmacotherapy; imagery rehearsal therapy (IRT) is the evidence-based psychotherapy.
Medication Effects on Sleep Architecture
| Drug Class | Effect on Sleep |
|---|---|
| SSRIs | Increase sleep latency; suppress REM sleep; may cause insomnia or vivid dreams; increase periodic limb movements |
| TCAs | Sedating (H1 blockade); potently suppress REM; may improve sleep continuity |
| Benzodiazepines | Decrease sleep latency; increase N2; decrease N3 (slow-wave) and REM; reduce sleep-related arousals |
| Alcohol | Initially sedating; disrupts sleep in second half of night; suppresses REM early, REM rebound later; worsens OSA |
| Mirtazapine | Markedly sedating; increases total sleep time; increases slow-wave sleep |
| Trazodone | Mildly increases slow-wave sleep; minimal REM suppression; used primarily as a hypnotic |
| Stimulants | Increase sleep latency; decrease total sleep time; suppress REM |
28 Comorbid Medical & Psychiatric Conditions
Delirium vs. Dementia vs. Depression
| Feature | Delirium | Dementia | Depression ("Pseudodementia") |
|---|---|---|---|
| Onset | Acute (hours to days) | Insidious (months to years) | Weeks to months |
| Course | Fluctuating (worse at night—"sundowning") | Progressive, stable day-to-day | Diurnal variation (worse in morning) |
| Attention | Impaired (cardinal feature) | Relatively preserved until late | Preserved (but effort-dependent) |
| Consciousness | Altered (clouded) | Clear until late stages | Clear |
| Memory | Impaired registration | Impaired recall, then recognition | "I don't know" responses; inconsistent performance |
| Psychomotor | Hyperactive, hypoactive, or mixed | Normal until late | Retardation or agitation |
| Hallucinations | Visual (common), tactile | Uncommon until late (Lewy body: visual) | Rare (unless psychotic depression) |
| Reversibility | Usually reversible if cause treated | Usually irreversible (progressive) | Reversible with treatment |
Metabolic Syndrome Monitoring on Antipsychotics
Second-generation antipsychotics (especially olanzapine and clozapine) carry significant metabolic risk. Follow the ADA/APA monitoring consensus guidelines:
| Parameter | Baseline | 4 wk | 8 wk | 12 wk | Quarterly | Annually |
|---|---|---|---|---|---|---|
| Weight/BMI | X | X | X | X | X | |
| Waist circumference | X | X | ||||
| Blood pressure | X | X | X | |||
| Fasting glucose | X | X | X | |||
| Fasting lipid panel | X | X | X (q5yr if normal) |
Threshold for concern: weight gain ≥5% from baseline warrants intervention (dietary counseling, exercise, consider switching to weight-neutral agent like aripiprazole or ziprasidone, or adding metformin 500–1000 mg BID). HbA1c should be obtained if fasting glucose is elevated or in patients with risk factors.
Hepatic Considerations
Most psychotropics are hepatically metabolized. In hepatic impairment: prefer medications with renal clearance or glucuronidation (LOT benzodiazepines, gabapentin, lithium, paliperidone). Avoid valproate (hepatotoxic), duloxetine (contraindicated in hepatic impairment), nefazodone (withdrawn in many countries due to hepatotoxicity). Reduce doses of most SSRIs, antipsychotics, and TCAs by 50% in significant hepatic disease. Check LFTs at baseline and periodically for valproate, carbamazepine, and duloxetine.
Renal Considerations
Lithium is renally cleared and requires dose adjustment in renal impairment (contraindicated in severe CKD). Monitor Cr and eGFR. Gabapentin and pregabalin require renal dose adjustment. Paliperidone (extended-release) requires dose reduction in renal impairment (CrCl <80). Most SSRIs and antipsychotics do not require renal dose adjustment as they are hepatically metabolized, but accumulation of active metabolites may occur.
Cardiac Considerations
QTc prolongation is a concern with several psychotropics. Agents with highest QTc risk: thioridazine, IV haloperidol, ziprasidone, citalopram (>40 mg), droperidol, methadone. Obtain baseline ECG when initiating high-risk agents. QTc >500 ms or increase >60 ms from baseline warrants drug discontinuation or switch. Risk factors for torsades de pointes: female sex, hypokalemia, hypomagnesemia, bradycardia, congenital long QT syndrome, heart failure, concomitant QTc-prolonging drugs. TCAs cause dose-dependent QRS prolongation—lethal in overdose (sodium channel blockade leading to ventricular arrhythmia); treat TCA overdose with IV sodium bicarbonate if QRS >100 ms.
Endocrine Considerations
Antipsychotic-induced hyperprolactinemia (highest with risperidone/paliperidone, FGAs) causes amenorrhea, galactorrhea, sexual dysfunction, gynecomastia, and may increase osteoporosis risk with long-term exposure. Management: reduce dose, switch to prolactin-sparing agent (aripiprazole, quetiapine, clozapine), or add low-dose aripiprazole (2–5 mg) as a partial D2 agonist to reduce prolactin. Lithium-induced hypothyroidism (20–30% of patients; more common in women) is treated with levothyroxine supplementation—lithium should generally NOT be discontinued solely due to hypothyroidism. SIADH and hyponatremia risk with SSRIs/SNRIs is highest in the first 2–4 weeks of treatment, in elderly patients, and in those on thiazide diuretics.
29 Classification Systems & Rating Scales
PHQ-9 (Patient Health Questionnaire-9)
9 items each scored 0–3 (not at all, several days, more than half the days, nearly every day) over the past 2 weeks. Items correspond to DSM-5 MDD criteria: (1) little interest/pleasure, (2) feeling down/depressed/hopeless, (3) sleep problems, (4) tired/little energy, (5) appetite changes, (6) feeling bad about self, (7) trouble concentrating, (8) psychomotor changes, (9) thoughts of self-harm. Scores: 0–4 minimal, 5–9 mild, 10–14 moderate, 15–19 moderately severe, 20–27 severe. Item 9 score ≥1 warrants direct suicide assessment.
GAD-7 (Generalized Anxiety Disorder-7)
7 items scored 0–3 over the past 2 weeks: (1) feeling nervous/anxious/on edge, (2) not being able to stop/control worrying, (3) worrying too much about different things, (4) trouble relaxing, (5) being so restless it is hard to sit still, (6) becoming easily annoyed/irritable, (7) feeling afraid as if something awful might happen. Scores: 0–4 minimal, 5–9 mild, 10–14 moderate, 15–21 severe.
MADRS (Montgomery-Åsberg Depression Rating Scale)
10 items, each scored 0–6, clinician-rated: (1) apparent sadness, (2) reported sadness, (3) inner tension, (4) reduced sleep, (5) reduced appetite, (6) concentration difficulties, (7) lassitude, (8) inability to feel, (9) pessimistic thoughts, (10) suicidal thoughts. Total 0–60. Thresholds: 0–6 normal, 7–19 mild, 20–34 moderate, ≥35 severe. A ≥50% reduction = response; score ≤10 = remission. Preferred over HAM-D in many clinical trials due to greater sensitivity to change.
HAM-D (Hamilton Depression Rating Scale)
17-item clinician-rated scale (HAM-D-17). Scores: 0–7 normal, 8–13 mild, 14–18 moderate, 19–22 severe, ≥23 very severe. Items include depressed mood, guilt, suicide, insomnia (early/middle/late), work/activities, retardation, agitation, anxiety (psychic/somatic), somatic symptoms, genital symptoms, hypochondriasis, weight loss, insight. Extended 21-item version adds diurnal variation, derealization, paranoia, obsessive symptoms.
YMRS (Young Mania Rating Scale)
11 items, clinician-rated, assessing manic symptoms: (1) elevated mood, (2) increased motor activity/energy, (3) sexual interest, (4) sleep, (5) irritability, (6) speech (rate and amount), (7) language-thought disorder, (8) content (grandiosity), (9) disruptive-aggressive behavior, (10) appearance, (11) insight. Four items scored 0–8, seven scored 0–4. Total 0–60. Thresholds: <12 remission, 12–19 mild mania, 20–25 moderate, ≥26 severe.
PANSS (Positive and Negative Syndrome Scale)
30 items, each scored 1–7, in three subscales: Positive (7 items: delusions, conceptual disorganization, hallucinatory behavior, excitement, grandiosity, suspiciousness/persecution, hostility); Negative (7 items: blunted affect, emotional withdrawal, poor rapport, passive/apathetic social withdrawal, difficulty in abstract thinking, lack of spontaneity/flow of conversation, stereotyped thinking); General Psychopathology (16 items). Total range 30–210. A ≥20% reduction from baseline = response; ≥50% = marked response.
AIMS (Abnormal Involuntary Movement Scale)
12 items assessing tardive dyskinesia. Items 1–7 (facial/oral movements: muscles of facial expression, lips and perioral area, jaw, tongue; extremity movements: upper, lower; trunk movements) scored 0–4 (none to severe). Items 8–10 assess global judgment (severity, incapacitation, patient awareness). Items 11–12 assess dental status. Should be performed at baseline and periodically during antipsychotic treatment.
CGI (Clinical Global Impression Scale)
Two single-item scales: CGI-S (Severity) — rated 1 (normal/not ill) to 7 (among the most extremely ill patients). CGI-I (Improvement) — rated 1 (very much improved) to 7 (very much worse). Widely used as a global outcome measure in clinical trials. Simple but validated and highly correlated with more detailed scales.
CIWA-Ar (Clinical Institute Withdrawal Assessment for Alcohol, Revised)
10-item clinician-rated scale for alcohol withdrawal severity: nausea/vomiting, tremor, paroxysmal sweats, anxiety, agitation, tactile disturbances, auditory disturbances, visual disturbances, headache/fullness in head, orientation/clouding of sensorium. Each item scored 0–7 (orientation 0–4). Maximum score 67. Scoring thresholds: <10 mild withdrawal (may not require pharmacotherapy), 10–18 moderate (consider treatment), >20 severe (high risk for complications—seizures, delirium tremens; requires aggressive benzodiazepine treatment). Used for symptom-triggered benzodiazepine dosing protocols, which reduce total benzodiazepine use and ICU length of stay compared to fixed-schedule dosing.
MoCA (Montreal Cognitive Assessment)
30-point screening tool for mild cognitive impairment: visuospatial/executive (5 points), naming (3), memory (5—delayed recall, no points at registration), attention (6), language (3), abstraction (2), orientation (6). Score ≥26 is normal; <26 suggests cognitive impairment (sensitivity ~90% for MCI). Add 1 point if education ≤12 years. More sensitive than MMSE for detecting mild cognitive impairment and frontal/executive deficits.
30 Medications Master Table
| Class | Drug | Typical Dose | Mechanism | Key Indication | Major Side Effects / Pearls |
|---|---|---|---|---|---|
| SSRIs | Fluoxetine | 20–80 mg | SERT inhibitor | MDD, OCD, bulimia, panic | Longest t1/2; CYP2D6 inhibitor |
| Sertraline | 50–200 mg | SERT inhibitor | MDD, PTSD, OCD, panic, SAD | Safest in cardiac; mild DRI | |
| Paroxetine | 20–50 mg | SERT inhibitor | MDD, GAD, panic, PTSD, OCD, SAD | Most anticholinergic; worst discontinuation; avoid in pregnancy | |
| Citalopram | 20–40 mg | SERT inhibitor | MDD | QTc risk; max 20 mg if >60 yr | |
| Escitalopram | 10–20 mg | SERT inhibitor | MDD, GAD | Fewest interactions; well tolerated | |
| Fluvoxamine | 100–300 mg | SERT inhibitor, sigma-1 agonist | OCD | CYP1A2 inhibitor; raises clozapine levels | |
| SNRIs | Venlafaxine XR | 75–225 mg | SERT + NET inhibitor | MDD, GAD, panic, SAD | HTN at high doses; discontinuation syndrome |
| Duloxetine | 60–120 mg | SERT + NET inhibitor | MDD, GAD, fibromyalgia, neuropathy | Avoid in hepatic disease; nausea | |
| Desvenlafaxine | 50 mg | SERT + NET inhibitor | MDD | Active metabolite of venlafaxine; fewer interactions | |
| Atypical AD | Bupropion | 150–450 mg | NDRI | MDD, smoking cessation, ADHD | No sexual dysfunction; seizure risk; avoid in bulimia/anorexia |
| Mirtazapine | 15–45 mg | Alpha-2 antagonist, 5-HT2/3 antagonist | MDD (especially with insomnia, poor appetite) | Sedation, weight gain; more sedating at 15 mg | |
| Trazodone | 50–400 mg | SARI | Insomnia (off-label), MDD | Priapism; orthostasis; usually used at low dose for sleep | |
| Mood Stabilizers | Lithium | 600–1800 mg | Multiple (IP3, GSK-3beta) | Bipolar (mania, maintenance), anti-suicide | Narrow TI; renal, thyroid toxicity; teratogenic (Ebstein) |
| Valproate | 750–2000 mg | GABA enhancer, Na+ channel | Bipolar mania, maintenance | Hepatotoxic; teratogenic (NTD); weight gain; thrombocytopenia | |
| Lamotrigine | 100–200 mg | Na+ channel blocker, glutamate inhibitor | Bipolar depression, maintenance | SJS risk; slow titration mandatory | |
| FGAs | Haloperidol | 2–20 mg | D2 antagonist | Schizophrenia, acute psychosis, agitation | High EPS; QTc (IV); LAI available |
| Chlorpromazine | 200–800 mg | D2 antagonist + multi-receptor | Schizophrenia, psychosis | Sedation; orthostasis; photosensitivity | |
| Fluphenazine | 2.5–20 mg | D2 antagonist | Schizophrenia | High EPS; LAI decanoate available | |
| SGAs | Risperidone | 1–6 mg | D2/5-HT2A antagonist | Schizophrenia, bipolar mania, irritability in autism | Highest prolactin; dose-dependent EPS |
| Olanzapine | 5–20 mg | D2/5-HT2A + multi-receptor | Schizophrenia, bipolar, agitation | Highest metabolic risk; high efficacy for mania | |
| Quetiapine | 150–800 mg | D2/5-HT2A/H1 antagonist | Schizophrenia, bipolar, MDD augmentation | Very sedating; metabolic risk; low EPS | |
| Clozapine | 150–900 mg | D4/5-HT2A > D2 | Treatment-resistant schizophrenia, anti-suicide | Agranulocytosis (REMS); seizures; metabolic; myocarditis |
31 Laboratory Monitoring Guide
| Drug Class / Agent | Baseline Labs | Ongoing Monitoring | Frequency |
|---|---|---|---|
| Lithium | BMP (Cr, BUN, electrolytes), TSH, calcium, CBC, urinalysis, ECG, pregnancy test | Lithium level (trough, 12hr post-dose), Cr, TSH, calcium | Level: 5–7 days after dose change until stable, then q3–6 months. Cr/TSH/Ca: q6 months |
| Valproate | CBC with differential, LFTs, pregnancy test, lipase | VPA level, CBC, LFTs; ammonia if AMS | Level: 3–5 days after dose change; CBC/LFTs q6 months |
| Carbamazepine | CBC, LFTs, BMP (sodium), HLA-B*1502 (in Asian patients), pregnancy test | CBZ level, CBC, LFTs, sodium | Level: 1 week after dose change; auto-induction check at 3–4 weeks; CBC/LFTs/Na q6 months |
| Lamotrigine | LFTs, renal function | Monitor for rash; lamotrigine level if needed (target 3–14 mcg/mL) | Rash assessment at each titration step; level not routine |
| Clozapine | ANC, fasting glucose, fasting lipids, weight/BMI, ECG | ANC (mandatory REMS), metabolic panel, weight | ANC: weekly x 6 mo, biweekly x 6 mo, then monthly. Metabolic: see SGA schedule |
| All SGAs | Fasting glucose, fasting lipid panel, weight/BMI, waist circumference, BP | Weight, fasting glucose, lipids, BP | Weight: monthly x 3 mo, then quarterly. Glucose/lipids: 12 weeks, then annually. BP: 12 weeks, then annually |
| All FGAs | AIMS score, ECG (if using IV haloperidol or thioridazine) | AIMS score; QTc if indicated | AIMS: q3 months for FGAs; q6 months for SGAs |
| TCAs | ECG (especially age >40 or cardiac risk), drug level for nortriptyline/desipramine | Drug level if toxicity suspected; ECG if dose changed | As clinically indicated; nortriptyline therapeutic window 50–150 ng/mL |
| MAOIs | BP, hepatic function | BP monitoring (orthostatic and hypertensive crisis) | BP at each visit; LFTs periodically |
| SSRIs/SNRIs | Sodium (elderly, diuretic users—SIADH risk) | Sodium if symptomatic; sexual function assessment | Sodium: 2–4 weeks after initiation in at-risk patients |
| Stimulants | Weight, height (children), BP, HR, cardiac history screening | Weight, BP, HR, growth parameters (children) | BP/HR at each visit; growth: q3–6 months in children |
32 Abbreviations Master List
General Psychiatry
| Abbreviation | Meaning |
|---|---|
| MDD | Major depressive disorder |
| GAD | Generalized anxiety disorder |
| OCD | Obsessive-compulsive disorder |
| PTSD | Post-traumatic stress disorder |
| ASD | Acute stress disorder (or autism spectrum disorder—context dependent) |
| BPD | Borderline personality disorder |
| ASPD | Antisocial personality disorder |
| DID | Dissociative identity disorder |
| SSD | Somatic symptom disorder |
| PDD | Persistent depressive disorder (dysthymia) |
| SAD | Social anxiety disorder (or seasonal affective disorder) |
| FEP | First-episode psychosis |
| TRD | Treatment-resistant depression |
| SUD | Substance use disorder |
| AUD | Alcohol use disorder |
| OUD | Opioid use disorder |
| DUP | Duration of untreated psychosis |
Clinical Assessment
| Abbreviation | Meaning |
|---|---|
| MSE | Mental status examination |
| SI / HI | Suicidal ideation / homicidal ideation |
| AVH | Auditory-verbal hallucinations |
| VH | Visual hallucinations |
| AH | Auditory hallucinations |
| IDA | Ideas of reference |
| LOC | Level of consciousness |
| OxP | Oriented to person, place, time (Ox3) or Ox4 (+ situation) |
| GAF | Global assessment of functioning (DSM-IV Axis V; now retired) |
| WHODAS | WHO Disability Assessment Schedule (DSM-5 replacement for GAF) |
| C-SSRS | Columbia Suicide Severity Rating Scale |
| PHQ-9 | Patient Health Questionnaire-9 (depression) |
| GAD-7 | Generalized Anxiety Disorder 7-item scale |
Rating Scales
| Abbreviation | Meaning |
|---|---|
| MADRS | Montgomery-Åsberg Depression Rating Scale |
| HAM-D | Hamilton Depression Rating Scale |
| YMRS | Young Mania Rating Scale |
| PANSS | Positive and Negative Syndrome Scale |
| Y-BOCS | Yale-Brown Obsessive Compulsive Scale |
| BFCRS | Bush-Francis Catatonia Rating Scale |
| AIMS | Abnormal Involuntary Movement Scale |
| CGI-S / CGI-I | Clinical Global Impression—Severity / Improvement |
| PCL-5 | PTSD Checklist for DSM-5 |
| CIWA-Ar | Clinical Institute Withdrawal Assessment for Alcohol (revised) |
| COWS | Clinical Opiate Withdrawal Scale |
| AUDIT | Alcohol Use Disorders Identification Test |
| CAGE | Cut down, Annoyed, Guilty, Eye-opener (alcohol screening) |
| MMSE | Mini-Mental State Examination |
| MoCA | Montreal Cognitive Assessment |
Pharmacology
| Abbreviation | Meaning |
|---|---|
| SSRI | Selective serotonin reuptake inhibitor |
| SNRI | Serotonin-norepinephrine reuptake inhibitor |
| TCA | Tricyclic antidepressant |
| MAOI | Monoamine oxidase inhibitor |
| NDRI | Norepinephrine-dopamine reuptake inhibitor (bupropion) |
| SARI | Serotonin antagonist and reuptake inhibitor (trazodone) |
| SRI | Serotonin reuptake inhibitor (umbrella term for SSRIs + clomipramine) |
| FGA / SGA | First-generation antipsychotic / second-generation antipsychotic |
| LAI | Long-acting injectable (antipsychotic) |
| REMS | Risk Evaluation and Mitigation Strategy |
| ANC | Absolute neutrophil count (clozapine monitoring) |
| VMAT2 | Vesicular monoamine transporter 2 (target of valbenazine, deutetrabenazine) |
| SERT / NET / DAT | Serotonin / norepinephrine / dopamine transporter |
Neuroscience & Anatomy
| Abbreviation | Meaning |
|---|---|
| DA | Dopamine |
| 5-HT | Serotonin (5-hydroxytryptamine) |
| NE | Norepinephrine |
| GABA | Gamma-aminobutyric acid |
| NMDA | N-methyl-D-aspartate (glutamate receptor subtype) |
| AMPA | Alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (glutamate receptor) |
| BDNF | Brain-derived neurotrophic factor |
| HPA axis | Hypothalamic-pituitary-adrenal axis |
| VTA | Ventral tegmental area |
| dlPFC | Dorsolateral prefrontal cortex |
| vmPFC | Ventromedial prefrontal cortex |
| OFC | Orbitofrontal cortex |
| ACC | Anterior cingulate cortex |
| LC | Locus coeruleus |
Procedures & Treatment Modalities
| Abbreviation | Meaning |
|---|---|
| ECT | Electroconvulsive therapy |
| TMS / rTMS | Transcranial magnetic stimulation / repetitive TMS |
| iTBS | Intermittent theta burst stimulation |
| VNS | Vagus nerve stimulation |
| DBS | Deep brain stimulation |
| CBT | Cognitive behavioral therapy |
| DBT | Dialectical behavior therapy |
| IPT | Interpersonal therapy |
| CPT | Cognitive processing therapy |
| PE | Prolonged exposure therapy |
| EMDR | Eye movement desensitization and reprocessing |
| ERP | Exposure and response prevention |
| CBT-I | Cognitive behavioral therapy for insomnia |
| MBT | Mentalization-based therapy |
| HRT | Habit reversal training |
| MAT | Medication-assisted treatment (for SUDs) |
| CSC | Coordinated specialty care (for FEP) |
Legal & Administrative
| Abbreviation | Meaning |
|---|---|
| AMA | Against medical advice |
| IVC | Involuntary commitment |
| LPS | Lanterman-Petris-Short Act (California mental health law) |
| M'Naghten | Insanity defense standard (cognitive test) |
| ALI | American Law Institute (insanity standard with volitional prong) |
| NGRI | Not guilty by reason of insanity |
| GBMI | Guilty but mentally ill |
| NMS | Neuroleptic malignant syndrome |
| EPS | Extrapyramidal symptoms |
| TD | Tardive dyskinesia |
| SJS / TEN | Stevens-Johnson syndrome / toxic epidermal necrolysis |
| PDSS | Post-injection delirium/sedation syndrome (olanzapine LAI) |
| BEN | Benign ethnic neutropenia (relevant to clozapine ANC thresholds) |