Addiction Medicine
Every substance use disorder, withdrawal syndrome, medication, screening instrument, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Neurobiology of Addiction — Foundations
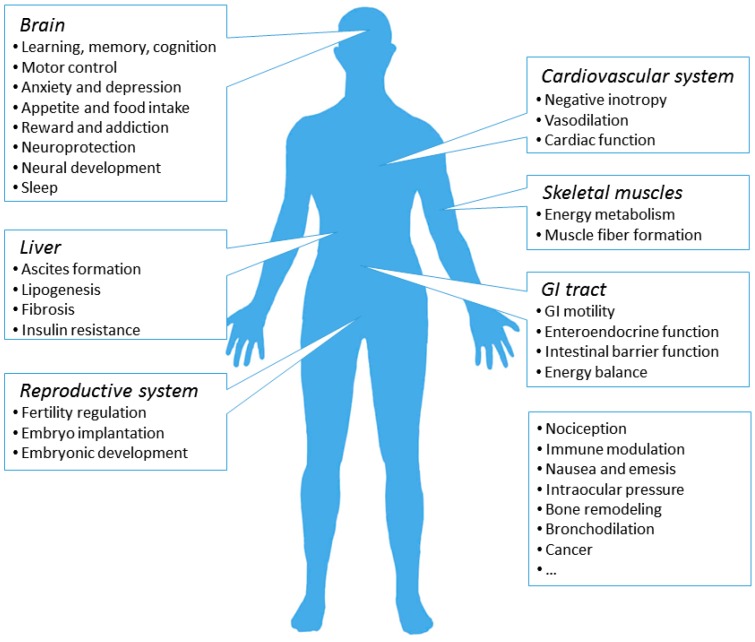
Addiction medicine treats a chronic, relapsing brain disorder characterized by compulsive substance use despite harmful consequences. Before you can document an encounter intelligently, you need to understand that addiction is not a moral failing or a lack of willpower — it is a disease of the brain's reward, motivation, memory, and executive control circuits. Every medication, every psychosocial intervention, and every relapse-prevention strategy in this guide targets one of these circuits.
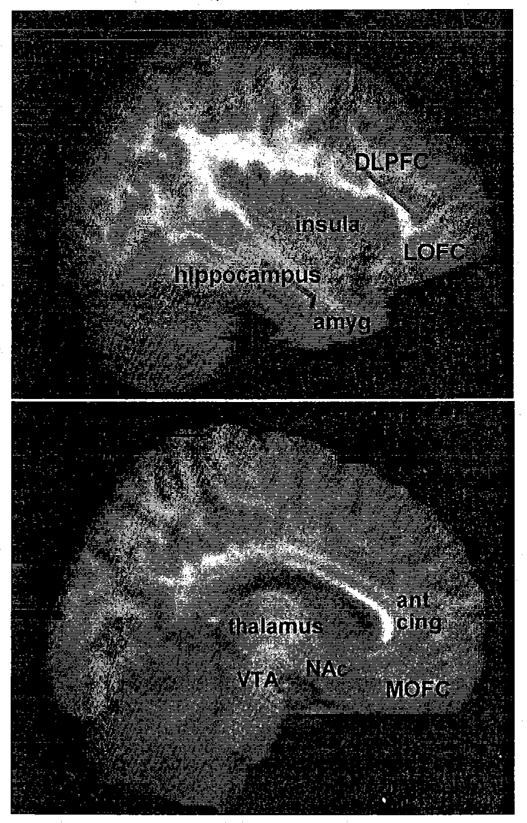
The Reward Pathway
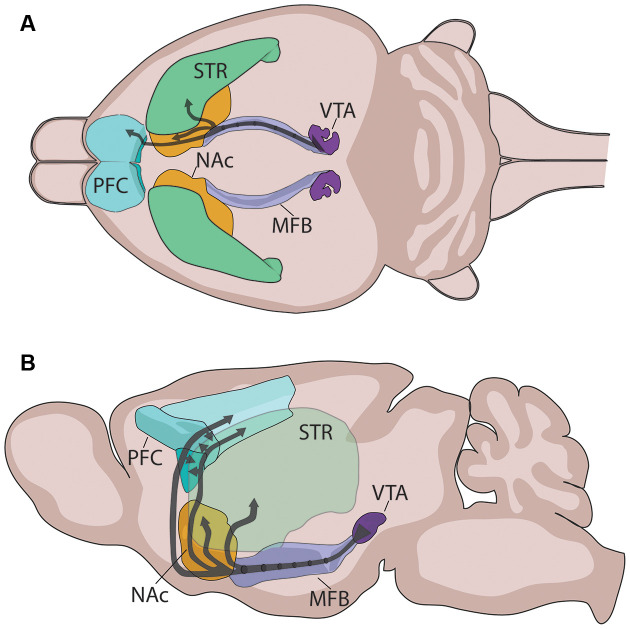
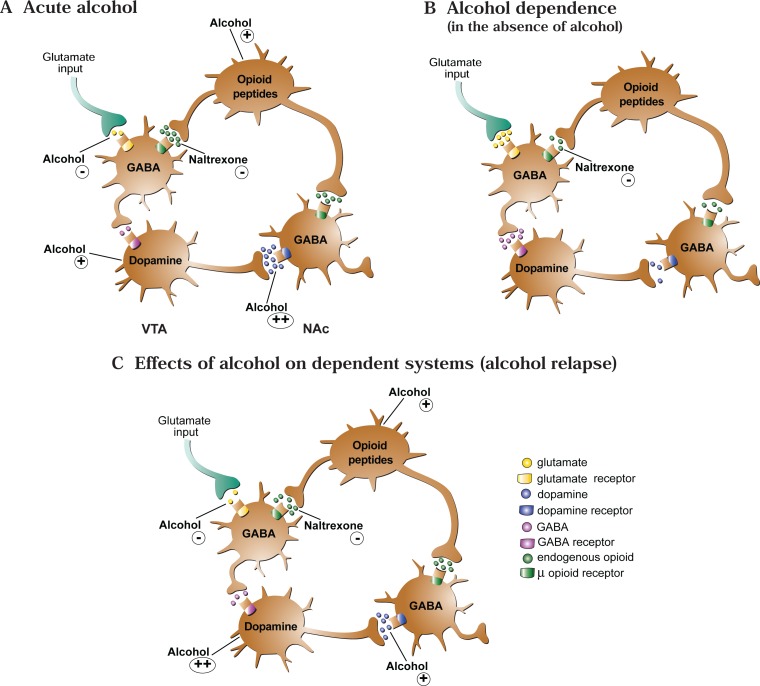
The mesolimbic dopamine pathway — also called the reward pathway — is the final common substrate for virtually every addictive substance. It originates in the ventral tegmental area (VTA) of the midbrain and projects to the nucleus accumbens (NAc) in the ventral striatum, with additional projections to the prefrontal cortex, amygdala, and hippocampus. Natural rewards (food, sex, social connection) cause modest, phasic dopamine release in the NAc. Drugs of abuse hijack this system, producing dopamine surges that are orders of magnitude larger and more reliable than any natural reward.
Three-Stage Cycle of Addiction
George Koob and Nora Volkow's model describes addiction as a three-stage cycle that gets progressively entrenched with each loop. Binge/intoxication is driven by dopaminergic reward in the basal ganglia. Withdrawal/negative affect is driven by recruitment of the extended amygdala (CRF, dynorphin, norepinephrine) and produces anxiety, dysphoria, and physical withdrawal symptoms. Preoccupation/anticipation (craving) is driven by executive-control deficits in the prefrontal cortex combined with cue-triggered memories from the hippocampus and amygdala. Chronic use blunts the reward response to natural rewards (anhedonia) while sensitizing the stress/withdrawal system, trapping the patient in a cycle where using the drug only relieves the dysphoria it created.
Substance Mechanisms at a Glance
| Substance | Primary Receptor/Target | Net Effect in Reward Pathway |
|---|---|---|
| Alcohol | GABA-A agonist, NMDA antagonist | Indirect dopamine release via GABA disinhibition |
| Opioids | Mu-opioid receptor agonist | Disinhibits VTA dopamine neurons |
| Cocaine | Monoamine reuptake inhibitor (DAT) | Blocks dopamine reuptake in NAc |
| Amphetamine/Meth | Reverses DAT; releases dopamine | Massive synaptic dopamine flooding |
| Nicotine | Nicotinic acetylcholine receptor (α4β2) | Stimulates VTA dopamine neurons |
| Cannabis (THC) | CB1 cannabinoid receptor agonist | Disinhibits VTA via GABA interneurons |
| Benzodiazepines | GABA-A positive allosteric modulator | Enhances GABA inhibition; mild reward |
| Hallucinogens (LSD, psilocybin) | 5-HT2A receptor agonist | Minimal direct dopamine; low addiction liability |
| PCP/Ketamine | NMDA antagonist | Dissociative; variable reward |
| MDMA | Serotonin & dopamine releaser | Empathogenic; moderate reward |
When documenting, never use stigmatizing language. Write "person with opioid use disorder" not "addict." Write "positive for fentanyl" not "dirty urine." Write "return to use" rather than "relapse" when that's the physician's framing. The ASAM and SAMHSA both publish language guidance — your physician will notice.
02 Scribe Documentation Framework
The Addiction Medicine SOAP Note
Addiction medicine encounters blend medical and psychiatric documentation. You will write a SOAP note, but the HPI is substance-focused, the exam includes a mental status exam (MSE), and the assessment frequently references DSM-5-TR criteria, ASAM level of care, and a medication-assisted treatment (MAT) plan. Confidentiality is more stringent than a typical medical note: federal regulation 42 CFR Part 2 governs the disclosure of substance use treatment records and is stricter than HIPAA.
Chief Complaint: The reason for today's visit in the patient's words. Examples: "I want to stop drinking," "here for my Suboxone," "court-ordered evaluation," "had an overdose last week."
HPI: A substance use HPI documents, for each substance of interest: age of first use, age of regular use, current quantity and frequency, route of administration (oral, intranasal, inhaled/smoked, IV), date and time of last use, maximum use, longest period of abstinence, prior treatment episodes, withdrawal history (including history of seizures or DTs for alcohol/benzos), overdose history, and triggers/context for use.
Past Psychiatric History: Prior diagnoses, hospitalizations, suicide attempts, self-harm, current mental health providers, current psychiatric medications, history of trauma.
Past Medical History: Document hepatitis C, hepatitis B, HIV, endocarditis, cellulitis/abscesses, tuberculosis, cirrhosis, pancreatitis, GI bleeding, seizures, and chronic pain syndromes — all common in SUD populations.
Social History: Housing stability (housed, unstably housed, unsheltered), employment, relationships, legal involvement (probation, parole, pending charges, drug court), insurance, custody issues, and peer use environment.
Review of Systems: Focus on withdrawal symptoms (nausea, sweating, tremor, anxiety, insomnia, piloerection, yawning, lacrimation, rhinorrhea, muscle aches), constitutional symptoms (weight loss, fatigue), GI (pain, bleeding), skin (injection sites, abscesses), and psychiatric (mood, sleep, appetite, SI/HI).
Vitals: Always document temperature, blood pressure, heart rate, respiratory rate, SpO2, and pain score. Tachycardia and hypertension suggest withdrawal or intoxication. Bradypnea suggests opioid toxicity.
Physical Exam: Focused head-to-toe with attention to pupils (pinpoint = opioids; dilated = stimulant/withdrawal), mucous membranes (dry in stimulant use), tremor, diaphoresis, gooseflesh, track marks, abscesses, cellulitis, stigmata of liver disease, heart murmur (endocarditis), and cognitive status.
Mental Status Exam: Appearance, behavior, speech, mood, affect, thought process, thought content (SI/HI, cravings), perception (hallucinations), cognition, insight, judgment.
Data: CIWA-Ar or COWS score if in withdrawal, UDS results, breath alcohol (BAL/BAC), labs (CBC, CMP, LFTs, lipase, PT/INR, ammonia, magnesium, phosphorus, HIV, hepatitis panel, pregnancy test, CK if stimulant).
Frame each diagnosis with DSM-5-TR severity (mild/moderate/severe), specify current use status (active, in early remission < 12 months, in sustained remission ≥ 12 months, in a controlled environment, on maintenance therapy), and document the ASAM level of care. The plan should address pharmacotherapy (MAT initiation or continuation), psychosocial (individual counseling, group, 12-step, IOP, residential), harm reduction (naloxone, sterile syringes, fentanyl test strips), labs, imaging, specialist referrals, safety planning, and follow-up.
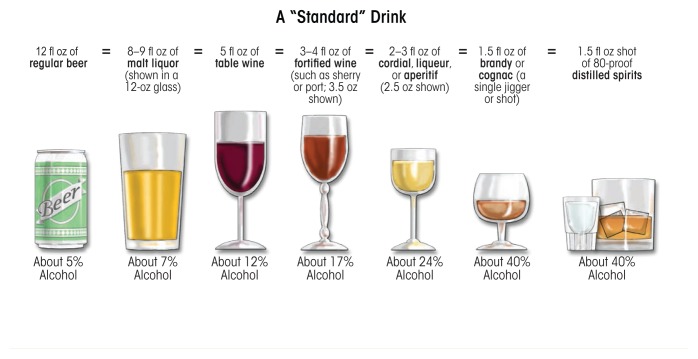
Quantify everything. "Drinks alcohol" is useless; "12 standard drinks daily (1.5 L vodka) for the last 3 years, last drink 6 hours ago" is a defensible note. One standard drink = 14 g ethanol = 12 oz beer = 5 oz wine = 1.5 oz spirits. Pack-years for tobacco. Grams/day for stimulants. Bags/day and MME (morphine milligram equivalents) for opioids.
03 DSM-5-TR Substance Use Disorder Criteria Framework
Every SUD in DSM-5-TR shares the same 11-item criterion set, grouped into four clusters. A diagnosis requires at least 2 criteria within a 12-month period. Severity is graded by count: mild (2–3), moderate (4–5), severe (6 or more). You will document these counts on virtually every new-patient note.
Impaired control (1–4):
1. Taken in larger amounts or over longer periods than intended.
2. Persistent desire or unsuccessful efforts to cut down or control use.
3. Great deal of time spent obtaining, using, or recovering from the substance.
4. Craving or a strong desire to use.
Social impairment (5–7):
5. Recurrent use resulting in failure to fulfill major role obligations at work, school, or home.
6. Continued use despite persistent social or interpersonal problems.
7. Important social, occupational, or recreational activities given up or reduced.
Risky use (8–9):
8. Recurrent use in physically hazardous situations.
9. Continued use despite knowledge of physical or psychological problems caused by the substance.
Pharmacologic (10–11):
10. Tolerance (need more to achieve the same effect, or diminished effect with the same amount).
11. Withdrawal (characteristic withdrawal syndrome, or use to avoid withdrawal).
Criteria 10 and 11 do not count when the substance is taken as prescribed under appropriate medical supervision.
| Severity | Criteria Met | Clinical Correlation |
|---|---|---|
| Mild | 2–3 | Outpatient counseling and brief intervention; consider pharmacotherapy |
| Moderate | 4–5 | Structured outpatient, pharmacotherapy, regular monitoring |
| Severe | 6 or more | Intensive outpatient, PHP, residential, or inpatient; full MAT offered |
04 Screening, Brief Intervention & Referral (SBIRT) Framework
Universal screening for unhealthy substance use is endorsed for adults in primary care settings by the USPSTF unhealthy alcohol use screening & brief counseling recommendation and, for drug use, by the USPSTF illicit drug use screening recommendation. You will see the physician administer or review several screening tools. Know each one.
C — Ever felt you should Cut down on drinking?
A — Have people Annoyed you by criticizing your drinking?
G — Have you felt Guilty about drinking?
E — Have you ever had a drink first thing in the morning (Eye-opener)?
Scoring: 2 or more "yes" answers is a positive screen.
The full AUDIT is 10 items, scored 0–40. The first 3 items (AUDIT-C) cover frequency, quantity, and heavy-drinking episodes and are scored 0–12.
AUDIT-C positive: ≥ 4 men, ≥ 3 women.
AUDIT total cutoffs: 8+ = hazardous/harmful use; 15+ (men) / 13+ (women) suggests likely alcohol dependence.
The 10 AUDIT items cover frequency of drinking, typical quantity, heavy-drinking frequency, inability to stop, failure to do what was expected, morning drinking, guilt, memory loss, injury from drinking, and concern from others.
Ten yes/no questions about non-alcohol drug use in the past 12 months. Scoring: 0 = no problems, 1–2 = low level, 3–5 = moderate, 6–8 = substantial, 9–10 = severe. A score of ≥ 3 is generally considered a positive screen warranting further assessment.
The NIDA Quick Screen asks about past-year use of alcohol, tobacco, prescription drugs used non-medically, and illegal drugs. Any "yes" triggers the full NIDA-Modified ASSIST, which generates a substance-specific involvement score guiding no-intervention / brief-intervention / brief-intervention-plus-referral pathways.
Ask — ask about use at every visit.
Advise — provide clear, personalized advice to change.
Assess — evaluate willingness to make a quit attempt.
Assist — offer counseling, pharmacotherapy, resources.
Arrange — schedule follow-up and referrals.
SBIRT is operationalized through the 5 A's in primary care and through the FRAMES model (Feedback, Responsibility, Advice, Menu, Empathy, Self-efficacy) in motivational interviewing.
Always document the score, not just "positive." "AUDIT-C 7, AUDIT 22" is meaningful; "CAGE positive" without a count is not. For motivational interviewing visits, document the stage of change (precontemplation, contemplation, preparation, action, maintenance) — the physician often uses these explicitly.
05 Alcohol Use Disorder (AUD) Depressant
AUD is the most common SUD in the United States and a top-five cause of preventable death. Alcohol is a CNS depressant that potentiates GABA-A receptors and antagonizes NMDA receptors. Chronic exposure downregulates GABA and upregulates glutamate, setting the stage for the hyperexcitable state of withdrawal. The 2020 ASAM Clinical Practice Guideline on Alcohol Withdrawal Management is the governing reference.
Clinical Presentation
Patients present across a spectrum: hazardous drinking without a disorder, AUD of varying severity, acute intoxication, withdrawal, or end-organ complications. A new-patient AUD HPI should quantify drinks per day, drinks per sitting, longest abstinence, consequences (DUI, blackouts, job loss, family conflict), and prior treatment. Ask specifically about history of seizures, delirium tremens, and prior detox admissions — these markedly raise the risk of a complicated withdrawal this time.
0.02–0.05% — relaxation, mild impairment.
0.08% — legal limit; impaired coordination and judgment.
0.15–0.25% — slurred speech, ataxia, vomiting.
0.25–0.35% — stupor, loss of consciousness.
> 0.40% — respiratory depression, coma, death in a non-tolerant drinker.
Tolerant drinkers can be alert and conversant at BACs that would kill an opioid-naive person.
End-Organ Complications
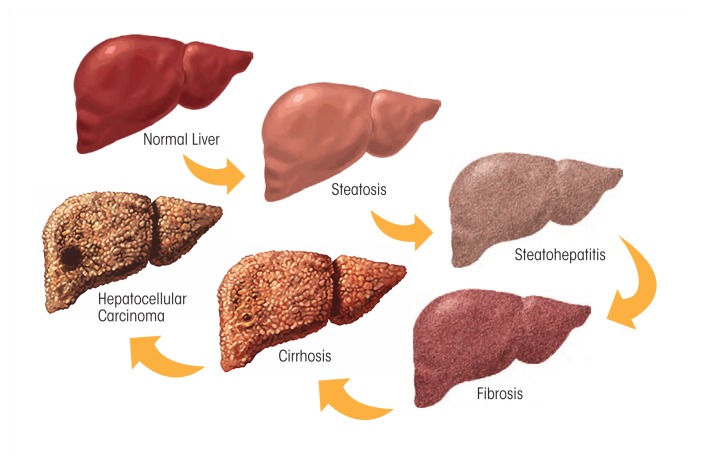
Document the full constellation when present: alcoholic hepatitis (jaundice, tender hepatomegaly, AST:ALT > 2 with AST usually < 500, leukocytosis), alcoholic cirrhosis (stigmata include spider angiomata, palmar erythema, gynecomastia, caput medusae, ascites, splenomegaly, hepatic encephalopathy) — cross-referenced in detail in the hepatology scribe guide — pancreatitis (acute and chronic), Wernicke encephalopathy (thiamine deficiency triad: encephalopathy, ophthalmoplegia, ataxia), and Korsakoff syndrome (dense anterograde amnesia with confabulation).
Always give IV thiamine before any glucose in a patient with suspected AUD. Giving glucose to a thiamine-deficient brain precipitates Wernicke encephalopathy. The typical prophylactic regimen is thiamine 100 mg IV/IM daily; treatment doses in suspected Wernicke are 500 mg IV TID for 2–3 days.
Management
FDA-approved pharmacotherapy for AUD includes naltrexone (oral 50 mg daily or long-acting injectable Vivitrol 380 mg IM monthly), acamprosate (666 mg PO TID), and disulfiram (250–500 mg PO daily). Off-label options supported by evidence include gabapentin, topiramate, and baclofen. All AUD patients should be offered pharmacotherapy — it is the standard of care, not an optional add-on. Psychosocial treatment (CBT, motivational enhancement, 12-step facilitation) is complementary, not alternative.
06 Alcohol Withdrawal & CIWA-Ar Withdrawal
Alcohol withdrawal is a potentially lethal syndrome driven by the unmasking of upregulated glutamatergic tone when alcohol (a GABA agonist) is removed. Symptoms begin 6–12 hours after the last drink and follow a predictable progression.
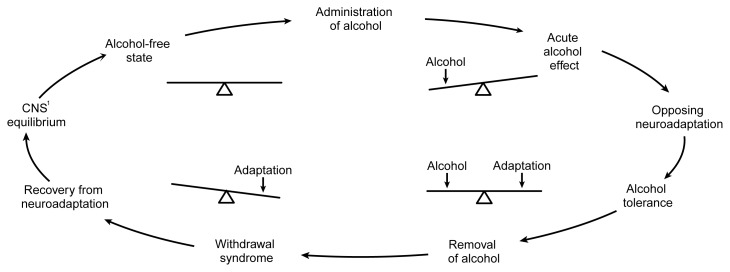
6–12 hours: tremor, anxiety, insomnia, GI upset, tachycardia, hypertension, diaphoresis.
12–24 hours: alcoholic hallucinosis (visual/auditory/tactile, usually with intact reality testing).
24–48 hours: withdrawal seizures (generalized tonic-clonic, typically a single or small cluster).
48–96 hours: delirium tremens (DTs) — autonomic instability, global confusion, severe tremor, hallucinations; mortality 1–5% with modern care, higher if untreated.
Any history of seizures or DTs predicts worse withdrawal and should push the team toward inpatient management.
The Clinical Institute Withdrawal Assessment for Alcohol, Revised. Nine items scored 0–7, one item (orientation) scored 0–4. Total score 0–67.
1. Nausea/vomiting (0–7)
2. Tremor (0–7)
3. Paroxysmal sweats (0–7)
4. Anxiety (0–7)
5. Agitation (0–7)
6. Tactile disturbances (0–7)
7. Auditory disturbances (0–7)
8. Visual disturbances (0–7)
9. Headache / fullness in head (0–7)
10. Orientation and clouding of sensorium (0–4)
Severity: < 10 mild (no pharmacotherapy usually needed), 10–18 moderate, > 18 severe (always treat, consider ICU).
Management
Benzodiazepines are the first-line treatment, administered in a symptom-triggered fashion guided by serial CIWA-Ar scores rather than fixed schedules when possible. Long-acting benzodiazepines (chlordiazepoxide, diazepam) provide a self-tapering effect and are preferred in patients with normal liver function; lorazepam is preferred in hepatic impairment and the elderly because it is not oxidatively metabolized. Phenobarbital is increasingly used (ED and ICU settings) for severe withdrawal or benzodiazepine-refractory DTs. Adjuncts include thiamine 100 mg, folate, multivitamins, IV fluids, magnesium replacement, and phosphorus replacement. Gabapentin is used for mild/moderate ambulatory withdrawal.
CIWA scores are only valid in a patient who can communicate. An intubated, sedated, or delirious patient cannot self-report nausea or hallucinations, and CIWA becomes meaningless. In those cases, the team switches to a symptom-based or scheduled approach (fixed benzodiazepine or phenobarbital protocols).
07 Opioid Use Disorder (OUD) Opioid
OUD is the driving force behind the overdose epidemic. Illicit opioids today are overwhelmingly fentanyl and its analogs, not heroin. Prescription OUD (oxycodone, hydrocodone, morphine, hydromorphone, methadone diverted) remains common. The ASAM National Practice Guideline for the Treatment of Opioid Use Disorder and SAMHSA TIP 63 are the two foundational references.
Intoxication & Overdose
Opioid intoxication presents with miosis (pinpoint pupils), sedation, slurred speech, and decreased respiratory rate. The overdose triad is miosis + unresponsiveness + respiratory depression. Death is usually from hypoxic respiratory failure. Treatment is naloxone 0.4–2 mg IV/IM/IN, titrated to restore respirations without precipitating full withdrawal. In fentanyl overdoses, repeat or higher doses may be needed due to potency and tissue redistribution.
Withdrawal
Opioid withdrawal is miserable but, unlike alcohol or benzodiazepine withdrawal, rarely directly lethal in adults. (The exception is in dehydrated patients where severe vomiting and diarrhea can cause fatal fluid/electrolyte derangements, and in neonates — see NAS below.) Onset depends on the half-life of the opioid: 6–12 hours for heroin/short-acting opioids, 24–48 hours for methadone.
Neonatal Abstinence Syndrome (NAS / NOWS)
Neonatal opioid withdrawal syndrome occurs in 55–94% of infants exposed to opioids in utero. Presents in the first 24–72 hours with tremor, high-pitched cry, hypertonia, poor feeding, diarrhea, sneezing, and sometimes seizures. Scored using the Finnegan tool or the newer ESC (Eat, Sleep, Console) approach. First-line treatment is non-pharmacologic (rooming-in, breastfeeding when appropriate, swaddling, low stimulation); pharmacologic treatment uses morphine or methadone.
Management
OUD is a chronic condition treated with medications for opioid use disorder (MOUD): buprenorphine, methadone, and extended-release naltrexone. MOUD reduces all-cause mortality by 50%+ and is the standard of care. Detox alone without maintenance MOUD has extraordinarily high return-to-use and overdose rates and is actively discouraged.
08 Opioid Withdrawal & COWS Withdrawal
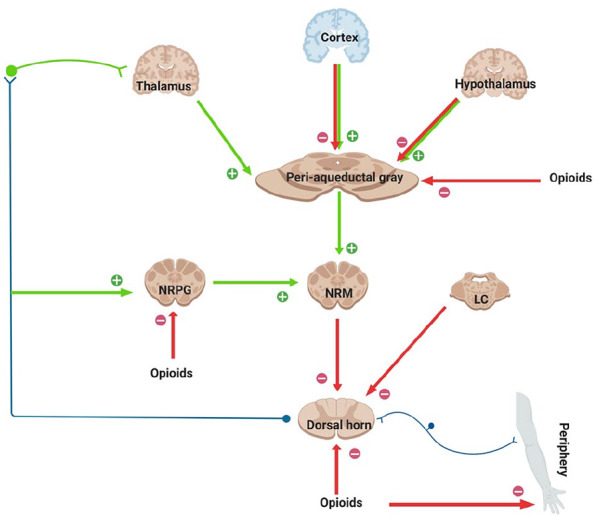
Clinical Opiate Withdrawal Scale. Eleven items, total score 0–48.
1. Resting pulse rate (0–4)
2. Sweating (0–4)
3. Restlessness (0–5)
4. Pupil size (0–5)
5. Bone or joint aches (0–4)
6. Runny nose or tearing (0–4)
7. GI upset (0–5)
8. Tremor (0–4)
9. Yawning (0–4)
10. Anxiety or irritability (0–4)
11. Gooseflesh skin (0–5)
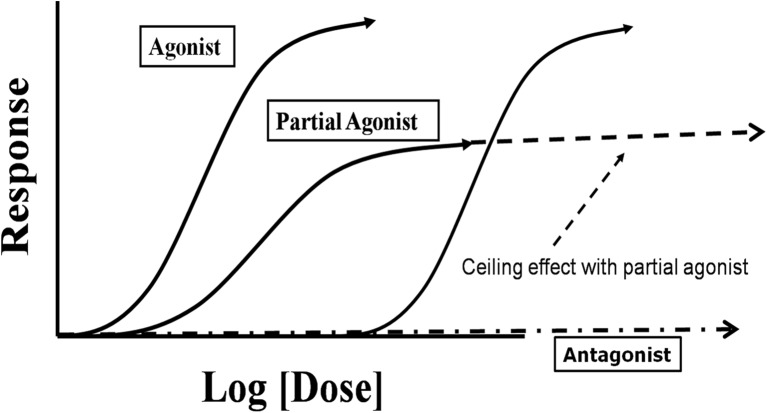
Severity: 5–12 mild, 13–24 moderate, 25–36 moderately severe, > 36 severe. A COWS ≥ 8 is the traditional threshold for standard buprenorphine induction; a COWS ≥ 11–13 is safer with fentanyl.
Induction Strategies
Standard (macro) buprenorphine induction: wait for objective withdrawal (COWS ≥ 8–13), then administer buprenorphine 2–4 mg SL and re-dose every 1–2 hours up to 8–16 mg on day 1. Low-dose induction (also called microinduction or the "Bernese method"): gives small, gradually escalating doses of buprenorphine while the patient remains on the full agonist, avoiding precipitated withdrawal. Commonly used for patients on high-dose chronic opioids, on fentanyl, or unable to tolerate traditional withdrawal. Methadone induction: done in an OTP; typical start 20–30 mg day 1, titrated up slowly (no more than 10 mg every few days) because of methadone's long half-life and delayed steady state.
Fentanyl changes everything. Chronic fentanyl use saturates fat tissue and causes prolonged, unpredictable withdrawal. Classic COWS ≥ 8 may precipitate a brutal withdrawal if you give buprenorphine too early. The current trend is either higher COWS thresholds or low-dose induction protocols — know your clinic's approach.
Adjunct Symptom Management
Clonidine 0.1–0.2 mg PO q4–6h for autonomic symptoms. Loperamide for diarrhea (warn about high-dose misuse). Ondansetron for nausea. Hydroxyzine or trazodone for insomnia. NSAIDs for body aches. Dicyclomine for abdominal cramping.
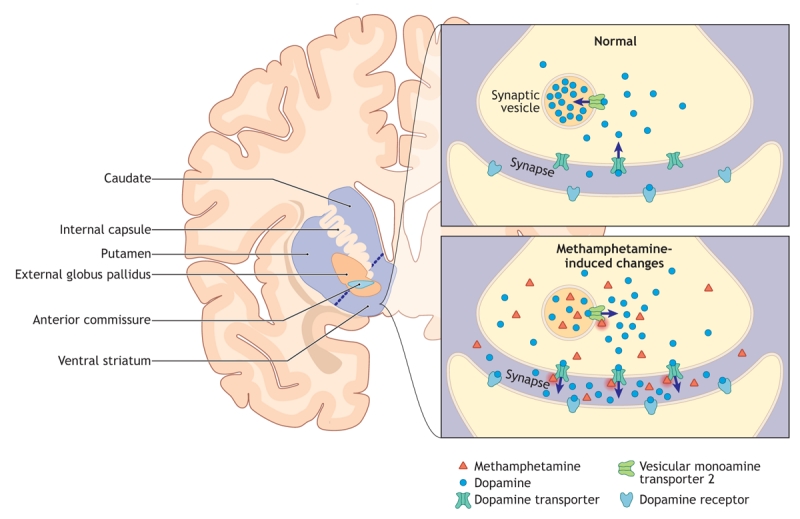
09 Stimulant Use Disorder Stimulant
Stimulant use disorder covers cocaine, amphetamines, and methamphetamine. All block or reverse monoamine transporters to flood the synapse with dopamine and norepinephrine. There is no FDA-approved pharmacotherapy for any stimulant use disorder — contingency management is the highest-evidence intervention.
Cocaine
Cocaine is typically snorted (intranasal), smoked (crack), or injected. Half-life is short (~1 hour), producing a rapid, brief high followed by craving. Presents with euphoria, hyperactivity, dilated pupils, tachycardia, hypertension, and hyperthermia. Complications: MI (cocaine causes coronary vasospasm — avoid beta-blockers in acute intoxication), aortic dissection, stroke, nasal septum perforation (chronic insufflation), levamisole-induced agranulocytosis and vasculitis (common adulterant), and seizures.
Amphetamines & Methamphetamine
Longer half-life than cocaine (8–12 hours). Presentations range from productive high-functioning use to severe stimulant-induced psychosis (paranoia, hallucinations, formication — "meth mites"). Chronic methamphetamine use causes marked dental destruction ("meth mouth"), skin excoriations, and substantial weight loss. Withdrawal is primarily psychiatric: dysphoria, hypersomnia, hyperphagia, anhedonia, and intense craving — not life-threatening but associated with high suicide risk in the first 1–2 weeks.
Management
No medication is FDA-approved. Off-label agents with modest evidence include mirtazapine (particularly for methamphetamine), bupropion, topiramate, modafinil, and the naltrexone + bupropion combination studied in recent trials. Contingency management (monetary or voucher incentives contingent on negative drug tests) is the single most effective intervention and is the standard of care where reimbursable.
10 Sedative-Hypnotic Use Disorder Depressant
Covers benzodiazepines, barbiturates, and Z-drugs (zolpidem, zaleplon, eszopiclone). All are GABA-A positive allosteric modulators. Patients often develop physiologic dependence on legitimately prescribed benzodiazepines; recognize this is not synonymous with SUD — it becomes a disorder when DSM-5-TR criteria are met (loss of control, craving, dose escalation beyond prescription, etc.).
Intoxication & Withdrawal
Intoxication resembles alcohol intoxication: sedation, ataxia, slurred speech, disinhibition, and in overdose, respiratory depression (much worse when combined with opioids). Reversal with flumazenil is generally avoided in chronic users because it precipitates seizures. Withdrawal mirrors alcohol withdrawal (tremor, anxiety, insomnia, autonomic instability, seizures, delirium) but with a longer, more protracted course because of longer half-lives. Withdrawal seizures and delirium can be fatal; abrupt discontinuation of a long-term benzodiazepine is never appropriate.
Management
Taper protocols: convert to a long-acting benzodiazepine (clonazepam, chlordiazepoxide, or diazepam) and reduce by 10–25% every 1–4 weeks. Faster tapers cause breakthrough withdrawal; slower tapers are often needed for long-duration or high-dose users. The phenobarbital protocol (a single loading dose followed by a predictable auto-taper via phenobarbital's 80-hour half-life) is used in inpatient and ED settings.
11 Cannabis Use Disorder Cannabinoid
Cannabis is the most commonly used illicit drug in the US; high-potency products (concentrates, edibles, vape cartridges) have dramatically increased THC exposure since the 1990s. Approximately 9% of users develop a cannabis use disorder; risk rises to ~17% for those starting in adolescence and ~25–50% for daily users.
Clinical Features
Intoxication: euphoria, conjunctival injection, increased appetite, dry mouth, tachycardia, impaired short-term memory, anxiety or panic (especially with edibles). Cannabis-induced psychosis can occur at high doses or in predisposed individuals. Cannabinoid hyperemesis syndrome (CHS) presents as cyclic vomiting in chronic users, classically relieved by hot showers — the only definitive treatment is abstinence. Withdrawal: irritability, anxiety, sleep disturbance, decreased appetite, and dysphoria peaking at 2–6 days and lasting up to 2 weeks.
Management
No FDA-approved pharmacotherapy. Limited evidence for N-acetylcysteine in adolescents, gabapentin, and off-label agents. Cognitive-behavioral therapy, motivational enhancement, and contingency management are the mainstays.
12 Hallucinogens, Dissociatives & MDMA Hallucinogen
Classic Hallucinogens (LSD, Psilocybin, DMT, Mescaline)
5-HT2A receptor agonists. Produce perceptual distortions, synesthesia, depersonalization, altered sense of time, and emotional intensification. Minimal reward pathway activation — low addiction liability, no withdrawal syndrome. Acute risks include panic reactions ("bad trips"), hallucinogen persisting perception disorder (HPPD), and trauma from impaired judgment. Managed with reassurance, a quiet environment, and a low-dose benzodiazepine if severe anxiety.
Dissociatives (Ketamine, PCP, Dextromethorphan)
NMDA antagonists. PCP (phencyclidine, "angel dust") causes vertical and horizontal nystagmus (pathognomonic), agitation, violent behavior, hyperthermia, rhabdomyolysis, and psychosis that can persist for days. Ketamine intoxication produces dissociation, analgesia, and amnesia; bladder toxicity (ketamine cystitis) is characteristic of chronic heavy use. Note that ketamine also has expanding legitimate clinical use for treatment-resistant depression.
MDMA (Ecstasy, Molly)
Serotonin and dopamine releaser with both empathogen and stimulant effects. Acute risks include serotonin syndrome, hyperthermia, hyponatremia (from excessive water intake during dance/rave settings), and — rarely — fatal heatstroke. Chronic use may cause persistent mood and cognitive changes.
13 Inhalants, Nicotine, Caffeine & Behavioral Addictions Other
Inhalants
Volatile solvents (glue, paint thinner, gasoline), aerosols, nitrites ("poppers"), and nitrous oxide. Rapid onset via lipid-soluble CNS penetration. Associated with sudden sniffing death syndrome (cardiac arrhythmia), hypoxia, and, for chronic use, leukoencephalopathy, peripheral neuropathy, and B12 deficiency (nitrous oxide inactivates B12). Most common in adolescents.
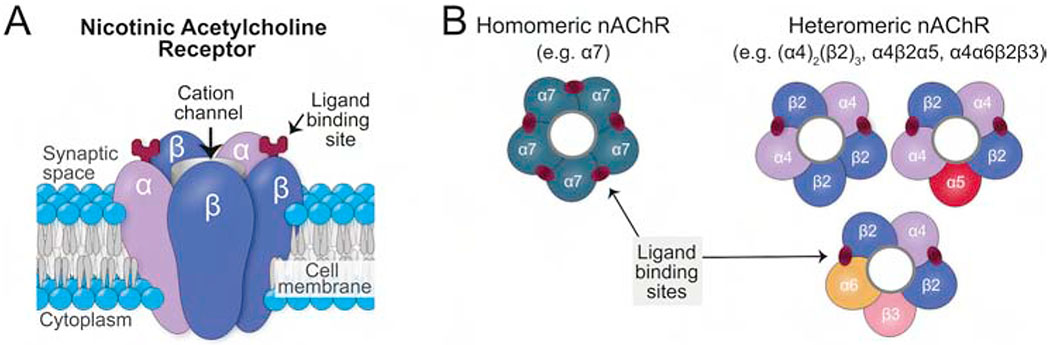
Tobacco / Nicotine Use Disorder
Nicotine activates α4β2 nicotinic receptors in the VTA. Withdrawal presents with irritability, anxiety, difficulty concentrating, increased appetite, and intense craving, peaking at 2–3 days and lasting 2–4 weeks. Screen every patient at every visit (ask, advise, assess, assist, arrange — the 5 A's). FDA-approved therapies include varenicline (most effective monotherapy; partial nicotinic agonist), bupropion SR, and nicotine replacement therapy — patch, gum, lozenge, inhaler, and nasal spray. Combination NRT (patch + short-acting) is more effective than either alone. Cite the 2021 USPSTF tobacco cessation recommendation when documenting counseling.
E-Cigarettes / Vaping
Deliver nicotine (often at very high concentrations — a single JUUL pod ~ 1 pack of cigarettes) in aerosolized form. Associated with EVALI (e-cigarette/vaping associated lung injury), predominantly linked to vitamin E acetate in illicit THC vape products. Effectiveness as a cessation tool is debated; never recommended as first-line.
Caffeine
DSM-5-TR recognizes caffeine intoxication, withdrawal, and "unspecified caffeine-related disorder" but not caffeine use disorder as a codable diagnosis. Document intake quantitatively when relevant (often relevant in anxiety, insomnia, and palpitation workups).
Gambling, Internet & Gaming
DSM-5-TR classifies gambling disorder under "Substance-Related and Addictive Disorders" — the only non-substance addiction currently recognized. Uses the same 11-criterion framework (adapted) with mild/moderate/severe grading. Internet gaming disorder is in the DSM-5-TR appendix as a condition for further study. ICD-11 includes gaming disorder as a formal diagnosis.
14 Dual Diagnosis & Substance-Induced Conditions Psychiatric
The co-occurrence of a substance use disorder with another psychiatric condition is the rule, not the exception. Roughly half of adults with any SUD have a co-occurring mental illness. Every note should explicitly address both. The distinction between a substance-induced disorder (symptoms that resolve with sustained abstinence) and an independent co-occurring disorder matters for treatment selection and prognosis — typically 30 days of abstinence is used as the minimum observation period before calling a mood or anxiety disorder "independent."
AUD + MDD: Up to 40% lifetime overlap. Treat both; SSRIs are first-line for MDD, naltrexone or acamprosate for AUD.
OUD + PTSD: High overlap, especially in veterans and trauma-exposed populations. Integrated trauma-focused care + MOUD is the standard.
SUD + Bipolar: Bipolar I with SUD is particularly high risk; mood stabilizers first, avoid chronic stimulant and benzodiazepine prescribing.
SUD + Schizophrenia: Tobacco and cannabis use are markedly elevated; long-acting injectable antipsychotics plus integrated SUD care.
SUD + Anxiety disorders: Treat with SSRIs, SNRIs, and non-benzodiazepine options (buspirone, hydroxyzine, gabapentin).
SUD + Borderline personality disorder: DBT-informed care integrated with SUD treatment.
Substance-Induced Psychiatric Conditions
DSM-5-TR recognizes substance-induced mood disorder, anxiety disorder, psychotic disorder, sleep disorder, sexual dysfunction, neurocognitive disorder, and delirium. Each requires the symptom to develop during or soon after intoxication or withdrawal and to be better explained by the substance than an independent disorder.
15 Medication-Assisted Treatment (MAT / MOUD) Treatment
MAT (the umbrella term) and MOUD (specifically medications for OUD) represent the single highest-impact intervention in addiction medicine. The FDA and ASAM both endorse buprenorphine, methadone, and naltrexone. Following the 2023 elimination of the X-waiver via the Mainstreaming Addiction Treatment (MAT) Act, any DEA-registered prescriber can now prescribe buprenorphine for OUD in the US.
Buprenorphine
Partial mu-agonist with a ceiling effect on respiratory depression and high mu affinity (displaces other opioids). Available as sublingual tablet/film (mono and combined with naloxone as Suboxone, Zubsolv, Bunavail), subcutaneous depot injection (Sublocade — monthly; Brixadi — weekly or monthly), and an older implant (Probuphine). The naloxone component is not absorbed sublingually and is included to deter injection misuse. Typical maintenance dose is 8–24 mg SL daily.
Methadone
Full mu-agonist with a long half-life. For OUD, methadone is dispensed exclusively through federally licensed Opioid Treatment Programs (OTPs). (It may be prescribed for chronic pain in any clinical setting — the OTP restriction applies only to its use for OUD.) Patients typically attend daily initially, earning "take-home" doses with sustained stability. Typical maintenance dose is 60–120 mg daily.
Naltrexone
Full mu-opioid antagonist. Available as oral 50 mg daily tablets and as extended-release IM injection (Vivitrol) 380 mg monthly. Requires 7–10 days of opioid abstinence before initiation to avoid precipitated withdrawal. Also FDA-approved for AUD. No dependence potential but adherence is the main challenge with oral formulation.
Buprenorphine: most patients, including primary care, telehealth, and pregnancy.
Methadone: high-tolerance patients, those who have failed buprenorphine, those needing the structure of daily OTP visits, chronic pain with OUD.
Naltrexone (XR): highly motivated patients, those who do not want an agonist, post-incarceration transitions.
Treatment Settings
MOUD can be initiated in office-based outpatient, telehealth (expanded post-COVID and now permanent for buprenorphine), hospital-based bridging (initiated on discharge from an ED or inpatient stay with warm handoff to outpatient care), and OTP settings (for methadone).
16 Psychosocial Treatments & Harm Reduction Treatment
Psychotherapies
Motivational interviewing (MI) is a collaborative, goal-oriented style of communication for strengthening motivation and commitment to change. Core principles are partnership, acceptance, compassion, and evocation. Cognitive-behavioral therapy (CBT) targets the thoughts and behaviors driving use. Contingency management (CM) provides tangible rewards (vouchers, prizes) for verified drug-negative urine tests — the strongest-evidence psychosocial treatment for stimulant use disorder. 12-step facilitation is a structured intervention designed to encourage engagement with AA, NA, or similar mutual-help groups. The Matrix Model is a structured 16-week outpatient treatment originally designed for stimulant use disorder.
Harm Reduction

Naloxone distribution: Every OUD patient and their household should leave with naloxone and training. Many states have standing orders allowing pharmacy dispensing without a prescription.
Fentanyl test strips: Allow users to test drugs for fentanyl contamination; legal status varies by state.
Syringe service programs (SSPs): Provide sterile syringes and other injection equipment to reduce HIV, hepatitis C, and soft-tissue infection transmission.
Supervised consumption sites (overdose prevention centers): Facilities where people can use pre-obtained drugs under medical supervision. Legal in a small number of US jurisdictions and widely operational internationally.
Safer-use education: Not using alone, rotating injection sites, avoiding combinations (especially opioids + benzodiazepines).
Levels of Care
See Section 20 for the full ASAM level-of-care enumeration. Levels range from early intervention (0.5) through outpatient (1), intensive outpatient/partial hospitalization (2.1/2.5), residential (3.1–3.7), and medically managed inpatient (4). Matching patients to the appropriate level is a core ASAM function and should be documented in every assessment.
17 Addiction Procedures & Office Workflows Procedure
Addiction medicine is less procedure-heavy than surgical specialties, but there are many office-based clinical workflows that function as "procedures" for documentation purposes.
| Workflow | Description | Scribe Documentation |
|---|---|---|
| SBIRT session | Screening + brief intervention + referral | Tool used, score, advice given, referral |
| Motivational interviewing session | Structured MI conversation | Stage of change, change talk, plan |
| Buprenorphine induction (standard) | In-office observed induction with COWS monitoring | COWS score q1–2h, cumulative dose, response |
| Buprenorphine induction (low-dose) | Microdose escalation while on full agonist | Daily dose, overlap with other opioid, COWS |
| Home (unobserved) induction | Patient self-initiates after in-office education | Instructions given, contact plan |
| Methadone induction | OTP-based; initial dose 20–30 mg | Dose, sedation/toxicity check, take-home status |
| Naltrexone XR (Vivitrol) injection | 380 mg IM gluteal every 4 weeks | Opioid-free interval, injection site, lot number |
| Sublocade administration | Monthly SQ buprenorphine depot | SL bup tolerated ≥ 7 days, abdominal SQ injection |
| Naloxone training | Overdose recognition + rescue instruction | Kit dispensed, who trained, recipient |
| Urine drug screen | Point-of-care or send-out | Observed vs unobserved, results, confirmatory needed |
| Breath alcohol (BrAC) | Handheld breathalyzer | Result in g/dL, device, time |
| Contingency management session | Verified-abstinence incentive | Sample verified, reward provided |
| Syringe exchange | Sterile equipment distribution | Items dispensed, education provided |
| PDMP query | State prescription monitoring database review | "PDMP reviewed" + any relevant findings |
| Treatment agreement | Controlled substance agreement signed | Document signing and key elements reviewed |
For every buprenorphine induction, document the initial COWS, the dose and time of each administration, the COWS score 60–90 minutes after each dose, total cumulative dose on day 1, and the plan for day 2 and beyond. A well-documented induction note is often auditable evidence of appropriate prescribing.
18 Drug Testing & Laboratory Interpretation Diagnostics
Urine Drug Screens (UDS)
Immunoassay-based point-of-care or lab UDS panels are the workhorse. Standard panels detect amphetamines, benzodiazepines, cocaine (detects the metabolite benzoylecgonine), opiates, PCP, and THC. Critical pitfalls: a standard "opiate" immunoassay detects morphine/codeine and often heroin but typically does not reliably detect oxycodone, hydrocodone, methadone, buprenorphine, or fentanyl — each of these requires a separate immunoassay or confirmatory test. Always confirm unexpected results (positive or negative) with GC-MS or LC-MS/MS before using them clinically.
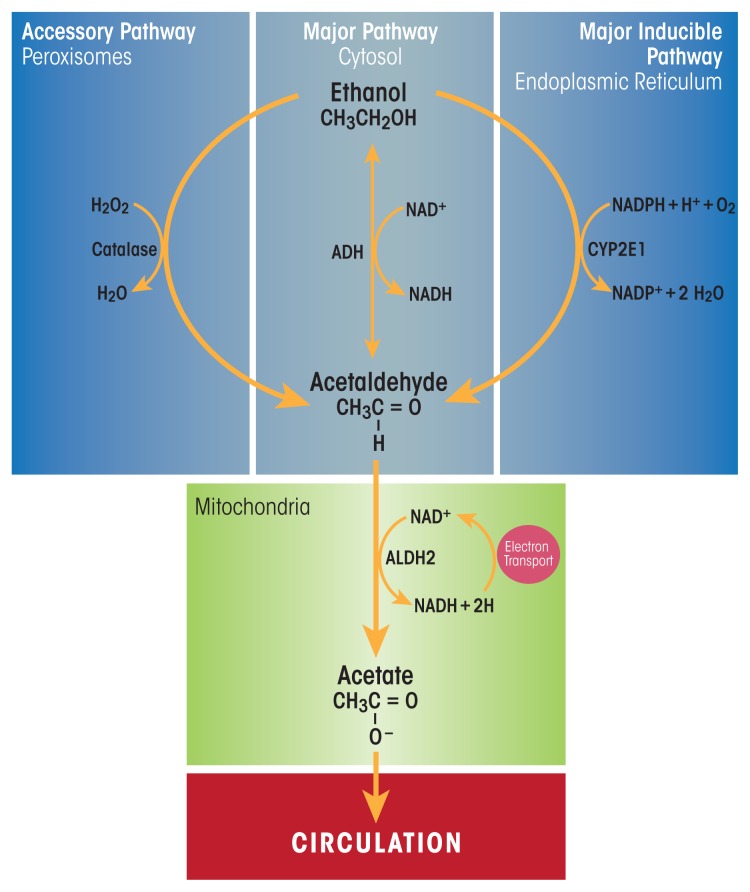
Detection Windows (Approximate)
| Substance | Urine Detection Window | Notes |
|---|---|---|
| Alcohol | 8–12 h (ethanol); up to 80 h (EtG/EtS) | EtG is highly sensitive; false positives from incidental exposure |
| Amphetamines | 2–3 days | False positives from pseudoephedrine, bupropion, trazodone |
| Benzodiazepines | 3–7 days; longer for long-acting | Many immunoassays miss clonazepam and lorazepam |
| Cocaine | 2–3 days | Metabolite: benzoylecgonine |
| Opiates (morphine/codeine) | 2–3 days | Poppy seeds can cause true positives |
| Oxycodone | 2–4 days | Requires separate assay |
| Methadone | 3–7 days | Separate assay |
| Buprenorphine | 3–7 days | Separate assay |
| Fentanyl | 1–3 days | Separate assay (critical) |
| THC (cannabis) | 3 days (single use) to 30+ days (chronic) | Lipid-soluble; prolonged with chronic use |
| PCP | 7–14 days |
Other Matrices
Breath alcohol (BrAC) — real-time alcohol measurement; reports in g/210 L or g/dL. Hair testing — 90-day retrospective window; used in custody and forensic settings, less useful clinically. Oral fluid — detects recent use (hours to 1–2 days). Phosphatidylethanol (PEth) — blood marker for alcohol use over the preceding 2–4 weeks; highly specific.
19 Medications You Must Know Medications
Alcohol Use Disorder
| Generic | Brand | Mechanism | Dose | Notes |
|---|---|---|---|---|
| Naltrexone (oral) | ReVia | Mu-opioid antagonist | 50 mg PO daily | Avoid with active opioid use; hepatotoxicity (rare) |
| Naltrexone XR | Vivitrol | Mu-opioid antagonist | 380 mg IM q4 weeks | Gluteal injection |
| Acamprosate | Campral | NMDA/GABA modulator | 666 mg PO TID | Renal dosing; safe in hepatic disease |
| Disulfiram | Antabuse | Aldehyde dehydrogenase inhibitor | 250–500 mg PO daily | Aversive reaction with alcohol; motivated patients only |
| Gabapentin | Neurontin | α2δ calcium channel | 600–1800 mg/day | Off-label; helpful with insomnia/anxiety |
| Topiramate | Topamax | GABA/glutamate modulator | 25–300 mg/day | Off-label; cognitive side effects |
| Baclofen | Lioresal | GABA-B agonist | 30–80 mg/day | Off-label; may help in cirrhosis |
Opioid Use Disorder
| Generic | Brand | Mechanism | Dose | Notes |
|---|---|---|---|---|
| Buprenorphine/naloxone | Suboxone, Zubsolv | Partial mu agonist | 8–24 mg SL daily | Ceiling effect; office-based |
| Buprenorphine mono | Subutex | Partial mu agonist | 8–24 mg SL daily | Preferred in pregnancy |
| Buprenorphine depot | Sublocade | Partial mu agonist | 300 mg SQ × 2, then 100 mg monthly | Requires ≥ 7 days of SL |
| Buprenorphine depot | Brixadi | Partial mu agonist | Weekly or monthly SQ | |
| Methadone | Dolophine | Full mu agonist | 60–120 mg PO daily | OTP only for OUD; QTc monitoring |
| Naltrexone XR | Vivitrol | Full mu antagonist | 380 mg IM q4 weeks | ≥ 7–10 days opioid-free |
| Naloxone | Narcan, Kloxxado | Mu antagonist | 4 mg intranasal q2–3 min PRN | Overdose reversal; distribute widely |
Tobacco Use Disorder
| Generic | Brand | Mechanism | Dose |
|---|---|---|---|
| Varenicline | Chantix | Partial nicotinic agonist | 0.5 mg → 1 mg BID over 1 week; 12 weeks |
| Bupropion SR | Zyban | NDRI | 150 mg daily → BID; 7–12 weeks |
| Nicotine patch | NicoDerm | NRT | 21 / 14 / 7 mg, step-down over 8 weeks |
| Nicotine gum | Nicorette | NRT | 2 or 4 mg q1–2h PRN |
| Nicotine lozenge | Commit | NRT | 2 or 4 mg q1–2h PRN |
| Nicotine inhaler | Nicotrol | NRT | 6–16 cartridges/day |
| Nicotine nasal spray | Nicotrol NS | NRT | 1–2 doses/h |
Withdrawal Management
| Indication | Drug | Role |
|---|---|---|
| Alcohol WD | Chlordiazepoxide, diazepam, lorazepam | First-line; symptom-triggered or fixed |
| Alcohol WD | Phenobarbital | Benzo-refractory; ICU protocols |
| Alcohol WD | Thiamine, folate, multivitamin | Prevent Wernicke; always |
| Opioid WD | Buprenorphine | Treatment of choice |
| Opioid WD | Clonidine, lofexidine | Autonomic symptom control |
| Opioid WD | Loperamide, ondansetron, hydroxyzine | Symptomatic adjuncts |
| Benzo WD | Long-acting benzo taper (clonazepam, chlordiazepoxide, diazepam) | Gradual taper over weeks |
| Benzo WD | Phenobarbital protocol | Inpatient rapid taper |
20 ASAM Criteria & Classification Systems Framework
The Six ASAM Dimensions
Every ASAM assessment evaluates the patient across six dimensions. Each is scored for severity, and the composite determines the recommended level of care.
Dimension 1: Acute intoxication and/or withdrawal potential.
Dimension 2: Biomedical conditions and complications.
Dimension 3: Emotional, behavioral, or cognitive conditions and complications.
Dimension 4: Readiness to change.
Dimension 5: Relapse, continued use, or continued problem potential.
Dimension 6: Recovery/living environment.
ASAM Levels of Care (Full Enumeration)
| Level | Name | Description |
|---|---|---|
| 0.5 | Early intervention | Education and brief intervention for at-risk individuals without a diagnosed SUD |
| 1 | Outpatient | < 9 hours/week of services; traditional office-based care |
| 2.1 | Intensive outpatient (IOP) | 9–19 hours/week; structured group/individual |
| 2.5 | Partial hospitalization (PHP) | ≥ 20 hours/week; day-hospital level |
| 3.1 | Clinically managed low-intensity residential | 24-hour supportive living |
| 3.3 | Clinically managed population-specific high-intensity residential | For patients with cognitive impairments |
| 3.5 | Clinically managed high-intensity residential | 24-hour intensive residential |
| 3.7 | Medically monitored intensive inpatient | 24-hour nursing with physician availability |
| 4 | Medically managed intensive inpatient | 24-hour medical and nursing; acute hospital setting |
When your physician places the patient at "ASAM 2.1" or "ASAM 3.5," document both the level and the dimensions driving the placement ("Dim 1 low, Dim 3 high due to active SI, Dim 6 unstable housing → recommend ASAM 3.5"). The payer may require dimensional justification for authorization.
21 Special Populations Population
MOUD — buprenorphine or methadone — is the standard of care for OUD in pregnancy. Detoxification is not recommended due to high relapse and fetal risk. Historically methadone was the default; buprenorphine (particularly the mono product) is now considered equivalent and is often preferred. Naltrexone is generally deferred until postpartum. Alcohol and tobacco cessation are priorities; benzodiazepines are avoided when possible (cleft palate risk, neonatal withdrawal). Document consumption in detail because of fetal alcohol spectrum disorder prevention.
Cannabis is the dominant substance. Screen with CRAFFT. Family-based therapies (MDFT, MST, FFT) have strong evidence. Buprenorphine and naltrexone are FDA-approved down to age 16; methadone requires special authorization.
Alcohol and prescription benzodiazepine/opioid use disorders dominate. Use the SMAST-G (geriatric MAST) for screening. Pharmacokinetics shift: reduced clearance, polypharmacy, and fall risk. Lower doses, slower tapers.
Post-release overdose risk is 10–40× baseline because tolerance falls during incarceration. MOUD continuation or initiation before release is a key intervention. Document probation/parole status, drug court participation, and mandated treatment.
Men who have sex with men (MSM) have elevated rates of methamphetamine and GHB use. Integrate HIV pre-exposure prophylaxis (PrEP) counseling. People who inject drugs (PWID) need routine screening for HIV, HCV, HBV, endocarditis, abscess, and cellulitis; hepatitis C treatment is now short-course and highly effective and should not be withheld because of ongoing use.
22 Physical Exam & Mental Status in Addiction
GEN: appearance, nutrition, hygiene, in/no distress
VS: T / BP / HR / RR / SpO2 / pain
HEENT: pupils (size, reactivity), nystagmus, nasal septum, oral mucosa, dentition
NECK: lymphadenopathy, thyromegaly, JVD
CV: rate, rhythm, murmurs (endocarditis)
LUNGS: air entry, rales, wheezes
ABD: tenderness, hepatomegaly, splenomegaly, ascites
SKIN: track marks, abscesses, cellulitis, jaundice, spider angiomata, diaphoresis, gooseflesh
EXT: edema, tremor, clubbing
NEURO: A&O, CN II–XII, motor, sensory, gait, coordination, asterixis
Appearance: grooming, dress, eye contact
Behavior: cooperative, agitated, psychomotor
Speech: rate, rhythm, volume, prosody
Mood: patient-reported
Affect: range, congruence, reactivity
Thought process: linear, tangential, circumstantial, loose
Thought content: SI/HI, cravings, delusions, obsessions
Perception: AH/VH, illusions
Cognition: alertness, orientation, attention, memory
Insight: good / fair / limited / poor
Judgment: good / fair / limited / poor
Addiction exams emphasize findings that localize to substance use: pupils (miosis with opioids, mydriasis with stimulants/withdrawal), nasal septum perforation from chronic cocaine insufflation, track marks and abscesses in IV drug users, stigmata of chronic liver disease in AUD, meth-associated dental caries, and the tremor, diaphoresis, and autonomic hyperactivity of withdrawal. A reliable CIWA or COWS score is documented as objective data.
23 Abbreviations Master List
Disorders & Syndromes
Treatment & Programs
Screening & Assessment
Medications, Labs & Testing
24 Sample HPI Templates
These sample HPIs reflect the rhythm and content of real addiction medicine clinic notes. Quantify everything, document the timeline, and always address the key diagnostic criteria.
"Mr. [Name] is a 54-year-old male self-referred for 'help with my drinking.' He reports daily alcohol use for approximately 20 years, escalating to 15–20 standard drinks per day (1.75 L of vodka) over the past 6 months. He drinks upon awakening to prevent morning tremor and anxiety. Longest period of abstinence was 2 weeks in 2023 during an involuntary hospital admission for a withdrawal seizure; he has never been through formal detox. He denies prior episodes of delirium tremens. Last drink was 4 hours ago (4 oz vodka). He meets criteria for severe AUD with 9 of 11 DSM-5-TR criteria endorsed. AUDIT-C 12, full AUDIT 34. He has lost his construction job due to drinking, is in the process of a divorce, and has been cited for DUI twice. Medical comorbidities include hypertension, elevated LFTs (AST 140, ALT 76), and thrombocytopenia concerning for early cirrhosis. He has no history of other SUD. He expresses strong motivation to quit and is open to medication-assisted treatment."
"Ms. [Name] is a 31-year-old female with severe OUD presenting for buprenorphine induction. She reports daily intranasal and IV fentanyl use for the past 2 years (approximately 1 gram per day of powder purchased as 'heroin'), escalating from prescription oxycodone following a knee surgery in 2019. Last use was approximately 18 hours ago. She reports severe withdrawal symptoms: body aches, diarrhea, sweating, yawning, restlessness, anxiety, and craving. COWS on arrival: 18 (moderate-severe). She has survived 2 overdoses (last 3 months ago, reversed with naloxone by a friend). Prior treatment: 1 prior buprenorphine attempt that ended after 2 weeks; 1 residential admission completed. No current methadone. Hepatitis C positive, untreated. She is unhoused, staying with friends. She has an 8-year-old child in her mother's custody. She has been out of jail for 3 weeks and is on probation. She is highly motivated and consents to buprenorphine induction today, with a plan to start at 4 mg SL, reassess at 60 minutes, and target 16 mg by end of day 1."
"Mr. [Name] is a 38-year-old male brought in by his partner for evaluation of paranoia and insomnia. He reports daily smoked methamphetamine use for 5 years, currently approximately 1 gram per day. Last use was 2 hours ago. He describes 'shadow people,' intrusive thoughts that his neighbors are monitoring him, and formication on his forearms (multiple excoriations present on exam). He has not slept in 4 days. He denies command hallucinations or acute suicidal ideation but acknowledges passive thoughts of 'not wanting to be alive.' Vitals: HR 118, BP 162/94, T 37.6, diaphoretic. Pupils 6 mm, reactive. He has lost 22 lb in 6 months and has significant dental decay. No IV use. No opioids. Intermittent alcohol use. Meets criteria for severe stimulant use disorder and stimulant-induced psychotic disorder. No prior treatment. He is open to residential (ASAM 3.5) placement."
"Mr. [Name] is a 62-year-old male with a 40-pack-year smoking history presenting for follow-up of his quit attempt. He set a quit date 3 weeks ago and started varenicline (0.5 mg titrated to 1 mg BID) and nicotine lozenges PRN. He has been completely abstinent from cigarettes for 14 days, though he reports moderate cravings in the morning and with his coffee, and has had one 'slip' (2 cigarettes at a party) that he did not consider a full relapse. He denies nausea, vivid dreams, or mood changes with varenicline. Exhaled CO today 4 ppm (consistent with nonsmoking). He plans to continue varenicline for a total of 12 weeks and will attend the hospital smoking cessation group weekly."
"Ms. [Name] is a 27-year-old female admitted 2 days ago after a witnessed opioid overdose; EMS administered intranasal naloxone 8 mg with restoration of ventilation, and she was intubated briefly in the ED for aspiration. She is now medically stable on the floor and being seen by the addiction medicine consult service. She reports daily fentanyl use (IV and intranasal) for 3 years, approximately 1.5 grams per day. This is her 4th known overdose. Prior MOUD history: brief methadone trial 2022, discontinued due to transportation barriers; no buprenorphine experience. She is hepatitis C positive (treatment-naïve), HIV negative, and has had one prior episode of cellulitis from injection at a left antecubital site (healed scar visible). She is motivated to start treatment after this overdose and is willing to begin in-hospital buprenorphine induction today with a warm handoff to our office-based clinic at discharge. Naloxone kit and education provided to her and her sister."
"Ms. [Name] is a 44-year-old female established patient with severe OUD on buprenorphine/naloxone 16 mg SL daily for 8 months and a history of major depressive disorder presenting for follow-up. She remains abstinent from illicit opioids (confirmed by UDS today: positive buprenorphine, negative fentanyl, negative other opioids). She reports worsening depressive symptoms over the past 3 weeks: anhedonia, early-morning awakening, decreased appetite with 6 lb weight loss, poor concentration, and a PHQ-9 of 18 today. She denies active SI but endorses passive death wishes. She is currently on sertraline 100 mg daily (stable for 6 months). She attends weekly individual therapy and bi-weekly SMART Recovery meetings. No return to use, no new stressors identified, though she notes the anniversary of her brother's overdose is next week. She is willing to increase sertraline and add a brief course of CBT for depression."
The best addiction medicine scribes are precise, non-judgmental, and fluent in both medical and psychiatric documentation. When the physician says "positive CIWA, start symptom-triggered chlordiazepoxide and thiamine IV," a great scribe already knows the CIWA components, recognizes the Wernicke prophylaxis, and charts the plan in the right structure. When the physician says "this patient needs a low-dose bup induction because of chronic fentanyl," the scribe anticipates the microdose schedule and the detailed COWS documentation required.
Learn to use non-stigmatizing language. Quantify every substance. Document the timeline meticulously — last use, longest abstinence, prior treatments, overdoses. Know the difference between dependence and a use disorder. Know the difference between a substance-induced condition and an independent comorbidity. Know the screening instruments and the withdrawal scales cold. Respect 42 CFR Part 2 confidentiality — SUD records have stricter protections than the rest of the chart.
Welcome to addiction medicine. This is the specialty where good documentation genuinely saves lives — every well-documented overdose, every accurate COWS score, every thorough MOUD note contributes to a patient's chance of recovery.
25 References & Sources
Clinical Practice Guidelines
ASAM. Clinical Practice Guideline on Alcohol Withdrawal Management. J Addict Med. 2020.
SAMHSA TIP 63: Medications for Opioid Use Disorder. 2021.
USPSTF. Screening for Unhealthy Drug Use. JAMA. 2020.
USPSTF. Interventions for Tobacco Smoking Cessation in Adults. JAMA. 2021.
CDC Clinical Practice Guideline for Prescribing Opioids for Pain — 2022.
FDA Approval and Clinical Use of Buprenorphine Formulations for Opioid Use Disorder.
ASAM Criteria (Fourth Edition): The Standard for Addiction Treatment Placement.
NIDA. Commonly Used Drugs Charts and Research Reports.
SAMHSA. Medications, Counseling, and Related Conditions for SUD.
Landmark Literature
Diagram & Figure Sources
Figure 1: Brain reward pathway diagram. NIDA / Wikimedia Commons. Public domain.
Figure 2: Limbic system diagram. Wikimedia Commons. Public domain.
Figure 3: OpenStax anatomy illustration of the human brain. OpenStax College / Wikimedia Commons. CC BY 3.0.
Figure 4: Naloxone nasal spray. Wikimedia Commons. Public domain.
Figure 5: Methadone treatment clinic. Wikimedia Commons. Public domain.