Bariatric / Metabolic Surgery
Every diagnosis, comorbidity, procedure, medication, classification, abbreviation, and documentation framework you need to succeed in a bariatric clinic from day one.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Obesity & Metabolic Disease — Foundations
Bariatric and metabolic surgery is the branch of general surgery that treats obesity and its metabolic consequences with operations on the stomach and small intestine. Unlike most surgical fields, bariatrics is as much about disease remission as it is about anatomical repair: a successful Roux-en-Y gastric bypass is judged not only by whether the anastomoses heal but by whether the patient's type 2 diabetes, hypertension, obstructive sleep apnea, and fatty liver disease improve or resolve. This framing shapes every encounter — the scribe is documenting a chronic metabolic disease, not a simple weight problem.
Obesity is defined by the World Health Organization using body mass index (BMI), the ratio of weight in kilograms to height in meters squared. BMI is imperfect (it ignores lean mass, fat distribution, and ethnicity) but it remains the universal entry criterion for surgical candidacy. The metabolic consequences of chronic adiposity — insulin resistance, chronic inflammation, adipokine dysregulation, ectopic fat deposition in liver and pancreas — drive almost every comorbidity the scribe will see in clinic.
Why the Term "Metabolic" Matters
The 2022 ASMBS/IFSO Indications for Metabolic and Bariatric Surgery reframed bariatric surgery as metabolic surgery and formally lowered BMI thresholds for surgical candidacy. The practical implication: a patient with BMI 32 and poorly controlled type 2 diabetes is now a surgical candidate, whereas older NIH 1991 criteria required BMI ≥ 35 with comorbidity or ≥ 40 without. You will see both frameworks referenced in charts.
02 GI & Foregut Anatomy for Bariatrics
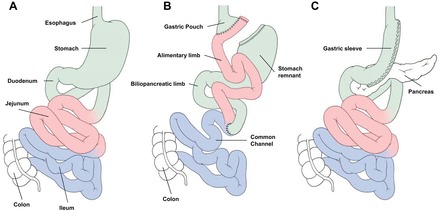
Every bariatric operation reshapes or reroutes the stomach and small intestine. You cannot chart a sleeve gastrectomy, gastric bypass, or duodenal switch accurately without a working map of the foregut.
Stomach Regions
The stomach is divided into the cardia (the junction with the esophagus at the gastroesophageal junction, or GEJ), the fundus (the dome above the cardia, where ghrelin-secreting cells are concentrated), the body (the largest central portion), the antrum (the distal funnel leading toward the pylorus), and the pylorus (the muscular sphincter controlling gastric emptying into the duodenum). The lesser curvature is the medial, right-sided border; the greater curvature is the lateral, left-sided border. A sleeve gastrectomy removes roughly 70–80% of the stomach along the greater curvature, preserving a narrow tube along the lesser curvature.
The angle of His is the acute angle where the esophagus meets the fundus, an important landmark for sleeve dissection and hiatal hernia repair. The crura of the diaphragm form the hiatal opening; a hiatal hernia exists when the GEJ or fundus migrates cephalad through this opening into the chest.
Small Intestine Segments
The small bowel runs from the pylorus to the ileocecal valve: duodenum (~25 cm, the first segment receiving bile and pancreatic secretions at the ampulla of Vater), jejunum (~2.5 m, the main site of nutrient absorption including iron, calcium, and folate), and ileum (~3.5 m, terminal segment absorbing vitamin B12 and bile salts). The ligament of Treitz marks the duodenojejunal junction and is the reference point for measuring bowel lengths in bypass operations.
Limb Terminology in Gastric Bypass
Alimentary (Roux) limb: The jejunal limb brought up to the gastric pouch; food travels through this limb. Typically 100–150 cm.
Biliopancreatic limb: The limb carrying bile and pancreatic juice from the excluded stomach and duodenum; no food passes through it. Typically 50–100 cm.
Common channel: The remaining small bowel distal to the jejunojejunostomy where food and biliopancreatic secretions mix and digestion/absorption occurs.
Gastric remnant: The bypassed stomach below the proximal pouch — still producing acid and endoscopically difficult to access.
You will hear "150/50" or "150/100" cited during RYGB cases — these are the Roux limb length followed by the biliopancreatic limb length. Longer Roux limbs mean more malabsorption but also more nutrient deficiency risk. Always document the exact limb lengths from the op note.
03 Scribe Documentation Framework
The Bariatric SOAP Note
Bariatric notes are longer than a typical surgical note because they must capture the entire metabolic picture. Every visit documents weight, BMI, comorbidity status, nutritional compliance, and (post-op) operative anatomy and limb lengths.
Weight history: Highest lifetime weight, current weight, starting weight at first consult, percent excess weight loss (%EWL) and percent total weight loss (%TWL) if post-op.
Diet history: Meal pattern, portion size, protein intake (grams/day), fluid intake, snacking, grazing, emotional eating, alcohol, sugar-sweetened beverages.
Prior weight-loss attempts: Commercial programs, medications (phentermine, GLP-1 agonists, orlistat), prior bariatric surgery, supervised diets.
Comorbidity status: Diabetes (on what medications? A1C trend?), hypertension, OSA (on CPAP? adherent?), hyperlipidemia, GERD, joint pain, depression, infertility.
Post-op specific: Nausea, vomiting, dysphagia, dumping symptoms, bowel habits, supplement adherence, exercise, support-group attendance.
Vitals & anthropometrics: Weight, BMI, waist circumference, blood pressure. Always document both absolute weight and the delta from prior visit.
Labs: CBC, CMP, lipid panel, A1C, TSH, vitamin B12, folate, ferritin, iron studies, 25-OH vitamin D, PTH, calcium, magnesium, zinc, copper, thiamine, vitamin A, prealbumin. Order frequency is typically at 3, 6, 12 months post-op then annually.
Physical exam: Focused cardiopulmonary, abdominal (scars, hernias, tenderness), skin (striae, intertrigo, panniculus), lower extremity edema.
Document each active problem with its status: (1) Obesity (class, BMI, %TWL); (2) Comorbidity updates (e.g., "T2DM — A1C improved from 8.4 to 6.1, stopped glipizide"); (3) Nutritional status and supplementation; (4) Surgical plan or post-op milestone; (5) Behavioral/psychological support; (6) Follow-up interval.
Percent excess weight loss (%EWL) uses ideal body weight at BMI 25 as the reference; percent total weight loss (%TWL) uses starting weight. Modern literature favors %TWL because it is less dependent on height. Always document both if the attending is tracking outcomes.
04 Obesity Classification & Metabolic Syndrome Metabolic
Precise classification matters because it determines surgical candidacy, insurance coverage, and the expected magnitude of weight loss. Memorize the BMI categories cold — you will use them every encounter.
| Category | BMI (kg/m²) | Clinical Notes |
|---|---|---|
| Underweight | < 18.5 | Evaluate for malnutrition/eating disorder |
| Normal | 18.5 – 24.9 | Reference range |
| Overweight | 25.0 – 29.9 | Lifestyle + risk factor control |
| Obesity Class I | 30.0 – 34.9 | Surgical candidate with metabolic disease (2022 ASMBS/IFSO) |
| Obesity Class II | 35.0 – 39.9 | Surgical candidate regardless of comorbidity |
| Obesity Class III (severe/morbid) | 40.0 – 49.9 | Standard surgical candidate |
| Super obesity | 50.0 – 59.9 | Higher perioperative risk; consider staged approach |
| Super-super obesity | ≥ 60.0 | Highest risk; often sleeve-first then second-stage DS |
Metabolic Syndrome
Metabolic syndrome is a cluster diagnosis — the presence of three or more of the following: central obesity (waist > 40 in men, > 35 in women), fasting glucose ≥ 100, triglycerides ≥ 150, HDL < 40 in men or < 50 in women, and blood pressure ≥ 130/85. It identifies patients at dramatically elevated cardiovascular and diabetes risk and is present in the majority of bariatric surgical candidates.
EOSS grades the clinical burden of obesity independent of BMI. It predicts mortality better than BMI alone and is increasingly cited in pre-op assessments.
| Stage | Description |
|---|---|
| 0 | No apparent obesity-related risk factors, physical symptoms, or functional limitations |
| 1 | Subclinical risk factors (borderline BP, mild dyslipidemia), mild symptoms, no functional limitation |
| 2 | Established obesity-related chronic disease (HTN, T2DM, OSA, PCOS) with moderate functional limitation |
| 3 | End-organ damage (MI, heart failure, diabetic complications, severe osteoarthritis) with significant functional limitation |
| 4 | Severe, potentially end-stage obesity-related disease with severe disability |
05 Type 2 Diabetes & Insulin Resistance Metabolic
T2DM is the single most important metabolic comorbidity driving bariatric referrals. Bariatric surgery — particularly RYGB and duodenal switch — produces diabetes remission that is rapid (within days, before significant weight loss), durable (years), and largely independent of the weight-loss effect. This is the mechanistic basis for calling these operations "metabolic."
Pathophysiology
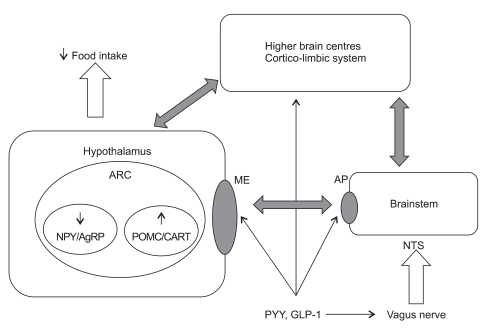
Visceral adiposity drives insulin resistance through chronic low-grade inflammation, free fatty acid flux, and altered adipokine signaling (decreased adiponectin, increased leptin and resistin). Hepatic insulin resistance raises fasting glucose; skeletal muscle insulin resistance blunts post-prandial glucose uptake; pancreatic β-cell failure eventually supervenes. Bariatric surgery improves diabetes through both foregut exclusion (bypassing the duodenum alters incretin signaling — the "foregut hypothesis") and accelerated hindgut nutrient delivery (increased GLP-1 and PYY — the "hindgut hypothesis"), alongside caloric restriction and weight loss.
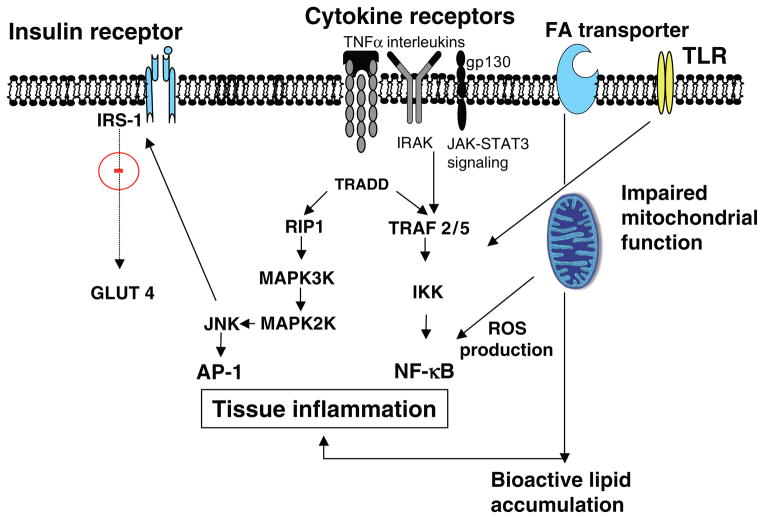
Diagnostic Thresholds
Diabetes is defined by fasting glucose ≥ 126, HbA1C ≥ 6.5%, 2-hour OGTT ≥ 200, or random glucose ≥ 200 with symptoms. Pre-diabetes: A1C 5.7–6.4%, fasting glucose 100–125. Document the current medication regimen, insulin total daily dose, and the most recent A1C at every bariatric visit.
Landmark Evidence
The STAMPEDE trial established 5-year superiority of bariatric surgery over intensive medical therapy for T2DM remission. Surgery patients had dramatically higher rates of A1C < 6.0% off all medications and better quality-of-life scores. Glycemic benefit is greatest for patients with shorter diabetes duration and preserved β-cell function.
After RYGB, insulin and sulfonylurea doses should be held or markedly reduced on the morning of surgery and often discontinued entirely within the first week post-op. Document any hypoglycemia, the exact medication changes, and communication with the managing endocrinologist or PCP.
06 OSA & Obesity Hypoventilation Syndrome Respiratory
Obstructive sleep apnea (OSA) is present in 60–90% of bariatric surgical candidates. Untreated OSA increases perioperative pulmonary complications, arrhythmias, and mortality. Pre-operative polysomnography or home sleep testing is standard in most bariatric programs.
OSA Definitions
OSA severity is graded by the apnea-hypopnea index (AHI): mild 5–14 events/hour, moderate 15–29, severe ≥ 30. Patients with moderate-to-severe OSA should be on CPAP pre-op; adherence (typically measured as ≥ 4 hours/night for ≥ 70% of nights) must be documented. The STOP-BANG questionnaire (Snoring, Tiredness, Observed apnea, Pressure, BMI > 35, Age > 50, Neck > 40 cm, Gender male) is the screening tool of choice — ≥ 3 positive responses = high OSA risk.
Obesity Hypoventilation Syndrome (OHS)
Defined as BMI ≥ 30 with daytime hypercapnia (PaCO2 ≥ 45) not attributable to another cause. Often coexists with severe OSA. Patients develop compensated respiratory acidosis, pulmonary hypertension, and right heart strain. OHS carries a markedly elevated perioperative risk and typically requires BiPAP (not just CPAP) and optimization before surgery. Always document serum bicarbonate — an HCO3 > 27 in an obese patient is a clue to undiagnosed OHS.
Pittsburgh Sleep Quality Index (PSQI)
The PSQI is a 19-item self-report tool scoring sleep quality across seven domains; a global score > 5 indicates poor sleep quality. It is often administered as part of the pre-op psychological evaluation.
07 NAFLD / MASLD & Hepatic Disease Metabolic
The condition formerly called non-alcoholic fatty liver disease (NAFLD) was renamed metabolic dysfunction-associated steatotic liver disease (MASLD) in 2023 to emphasize its metabolic etiology and reduce stigma. It is present in the majority of bariatric surgical candidates and ranges from simple steatosis to steatohepatitis (MASH, formerly NASH), fibrosis, and cirrhosis.
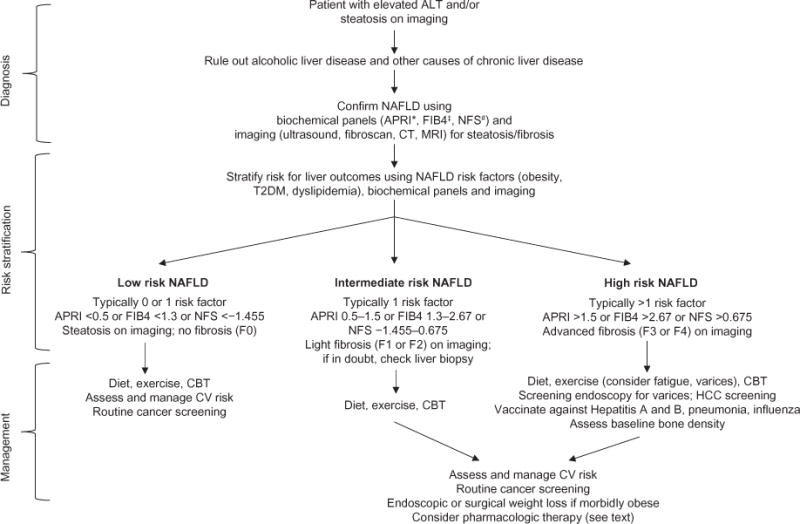
Diagnostic Workup
Scribes will document ALT, AST, alkaline phosphatase, platelet count, and non-invasive fibrosis scores (FIB-4, NAFLD fibrosis score). Liver elastography (FibroScan) gives a kPa value — < 8 kPa is low-risk for advanced fibrosis, > 12 kPa suggests advanced fibrosis/cirrhosis. Intraoperative liver biopsy is frequently performed during bariatric cases because the surgeon has direct visualization and palpation of the liver.
Implications for Surgery
Compensated cirrhosis (Child-Pugh A, MELD < 10) is not an absolute contraindication to bariatric surgery, but decompensated cirrhosis with portal hypertension, ascites, or varices dramatically increases operative risk and typically excludes patients from elective bariatric procedures. A very enlarged fatty liver can make sleeve gastrectomy technically difficult — pre-operative "liver shrinkage diets" (2-week very-low-calorie diet) are used to reduce hepatic volume before surgery.
08 GERD & Esophageal Disease in Obesity GI
Obesity is an independent risk factor for GERD because increased intra-abdominal pressure promotes reflux across the gastroesophageal junction. GERD status is critical in bariatric surgical planning because sleeve gastrectomy can worsen reflux while Roux-en-Y gastric bypass generally improves it.
Pre-Operative GERD Evaluation
Most bariatric programs obtain an upper endoscopy (EGD) before sleeve gastrectomy to rule out Barrett's esophagus, large hiatal hernia, esophagitis (graded by the Los Angeles A–D classification), and H. pylori infection. Patients with significant GERD symptoms, Barrett's esophagus, or LA grade C/D esophagitis are typically steered toward RYGB rather than sleeve. Hiatal hernias identified pre-op are repaired concurrently with the bariatric procedure.
| Grade | Endoscopic Finding |
|---|---|
| A | One or more mucosal breaks ≤ 5 mm, not extending between folds |
| B | One or more mucosal breaks > 5 mm, not continuous between folds |
| C | Mucosal breaks continuous between folds, < 75% of circumference |
| D | Mucosal breaks involving ≥ 75% of esophageal circumference |
09 Cardiovascular, Pulmonary HTN & Venous Disease Cardiovascular
Obesity-related cardiovascular burden extends beyond hypertension and coronary disease. Scribes should document the full constellation of cardiopulmonary comorbidities on every patient.
Hypertension & Heart Failure
Obesity causes eccentric left ventricular hypertrophy through chronic volume overload, eventually progressing to heart failure with preserved ejection fraction (HFpEF). Bariatric surgery improves both systolic and diastolic function; blood pressure medications are frequently reduced or stopped within weeks.
Pulmonary Hypertension
Chronic OSA, OHS, and left heart failure can all drive secondary pulmonary hypertension. A right heart catheterization or echocardiogram demonstrating elevated PA pressures (mean PA pressure ≥ 20 mmHg) is a significant risk modifier; severe pulmonary hypertension (PASP > 60) may preclude surgery or require pre-operative optimization.
Venous Thromboembolism Risk
Obese patients are at markedly elevated VTE risk because of venous stasis, chronic inflammation, and hypercoagulability. The Caprini score stratifies this risk; bariatric surgery patients are typically Caprini 5+ (high risk) and receive aggressive prophylaxis. Chronic lower extremity venous insufficiency, stasis dermatitis, and venous ulcers are common in severe obesity and should be documented pre-operatively.
Age, BMI > 25, BMI > 40, major surgery ≥ 45 min, prior VTE, varicose veins, OCP/HRT, pregnancy/postpartum, immobility, malignancy, inherited thrombophilia. Score ≥ 5 = highest risk category; bariatric patients almost always land here.
10 MSK, Reproductive & Psychiatric Comorbidities Multisystem
Musculoskeletal
Weight-bearing joint osteoarthritis (especially knees and hips), chronic low back pain, and reduced mobility are near-universal in severe obesity. Many patients are referred for bariatric surgery specifically to qualify for joint replacement (most orthopedic surgeons require BMI < 40 for elective arthroplasty). Document baseline functional status: can the patient walk one block? Climb stairs? Get out of a low chair?
Reproductive Health
Obesity causes anovulation, infertility, polycystic ovary syndrome (PCOS), and menstrual irregularity in women, and hypogonadism/low testosterone in men. Fertility typically improves rapidly after surgery — patients are counseled to avoid pregnancy for 12–18 months post-op to allow weight stabilization and reduce teratogenicity risk from nutritional deficiencies. Long-acting reversible contraception is frequently discussed. Pregnancy after bariatric surgery carries specific risks (internal hernia, nutrient deficiency in the fetus) that must be tracked closely.
Depression, Binge Eating, and Psychiatric Screening
Depression, anxiety, PTSD, and eating disorders (particularly binge eating disorder and night eating syndrome) are overrepresented in bariatric candidates. Every program requires a pre-operative psychological evaluation. Uncontrolled major depression, active substance use, untreated eating disorders, or psychosis are typical contraindications to surgery. Post-operatively, untreated depression predicts weight regain.
Bariatric surgery patients have an elevated risk of suicide and self-harm compared to the general population, with highest risk in the second and third post-operative years. Contributing factors include altered pharmacokinetics of antidepressants (particularly after RYGB), unresolved body image issues, marital stress from weight loss, and unmasking of underlying psychiatric disease. Always document mood, ideation screening, and therapy engagement at follow-ups.
11 Pre-Operative Evaluation & Patient Selection Workflow
Bariatric programs follow a multidisciplinary evaluation pathway that spans 3–6 months before surgery. Scribes will chart pieces of this process at multiple encounters.
ASMBS / IFSO 2022 Indications
Per the 2022 ASMBS/IFSO joint statement:
- BMI ≥ 35 regardless of presence, absence, or severity of comorbidities
- BMI 30–34.9 with metabolic disease (particularly T2DM)
- BMI ≥ 27.5 in appropriately selected Asian populations with comorbidities
- Children and adolescents with BMI ≥ 120% of the 95th percentile plus major comorbidity, or ≥ 140% of the 95th percentile
The older 1991 NIH Consensus Statement (BMI ≥ 40 or ≥ 35 with comorbidity) is still cited by some payors and old chart notes.
Pre-Op Workup Components
- Medical clearance: H&P, labs (CBC, CMP, A1C, lipids, TSH, vitamin D, iron studies, B12, PT/PTT/INR), ECG, chest X-ray, pregnancy test
- Cardiac clearance: Echocardiogram for patients with cardiac disease, stress testing or cardiology consultation as indicated
- Pulmonary: Sleep study (polysomnography or home sleep test); PFTs for select patients
- GI: Upper endoscopy (standard for sleeve; selective for bypass); H. pylori testing and treatment
- Nutrition: Registered dietitian evaluation and pre-op education; documentation of a supervised weight-loss attempt if insurance requires it
- Behavioral health: Psychological evaluation to screen for eating disorders, substance use, depression, and readiness for lifestyle change
- Pre-op weight loss: 2-week very-low-calorie "liver shrinkage" diet immediately before surgery
ASA Physical Status Classification
| Class | Description |
|---|---|
| I | Normal healthy patient |
| II | Mild systemic disease (e.g., controlled HTN, BMI 30–40) |
| III | Severe systemic disease (e.g., poorly controlled DM, BMI ≥ 40, OSA on CPAP) |
| IV | Severe systemic disease that is a constant threat to life |
| V | Moribund patient not expected to survive without the operation |
| VI | Declared brain-dead; organs being removed for donor |
12 Core Bariatric Procedures Surgical
Four operations dominate modern bariatric practice. Learn their anatomy, expected weight loss, comorbidity impact, and complication profile cold.
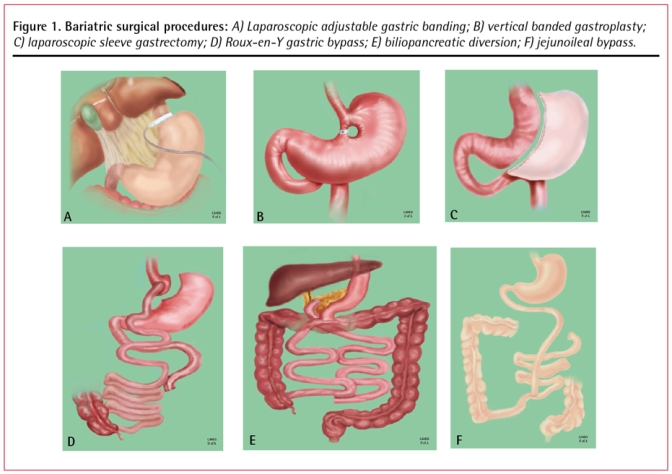
Roux-en-Y Gastric Bypass (RYGB)
Historically the "gold standard" of bariatric surgery. A small (15–30 mL) gastric pouch is created at the proximal stomach and divided from the remainder of the stomach. The jejunum is divided approximately 50–100 cm distal to the ligament of Treitz, and the distal end (the Roux limb) is brought up and anastomosed to the gastric pouch (gastrojejunostomy). The proximal end (the biliopancreatic limb) is then anastomosed to the Roux limb 100–150 cm distally (jejunojejunostomy), creating the common channel. Weight loss mechanism is a combination of restriction, malabsorption, altered gut hormones (increased GLP-1, PYY; decreased ghrelin), and food aversion via dumping physiology. Expected weight loss: 60–80% EWL at 1–2 years; excellent T2DM remission; improves GERD.
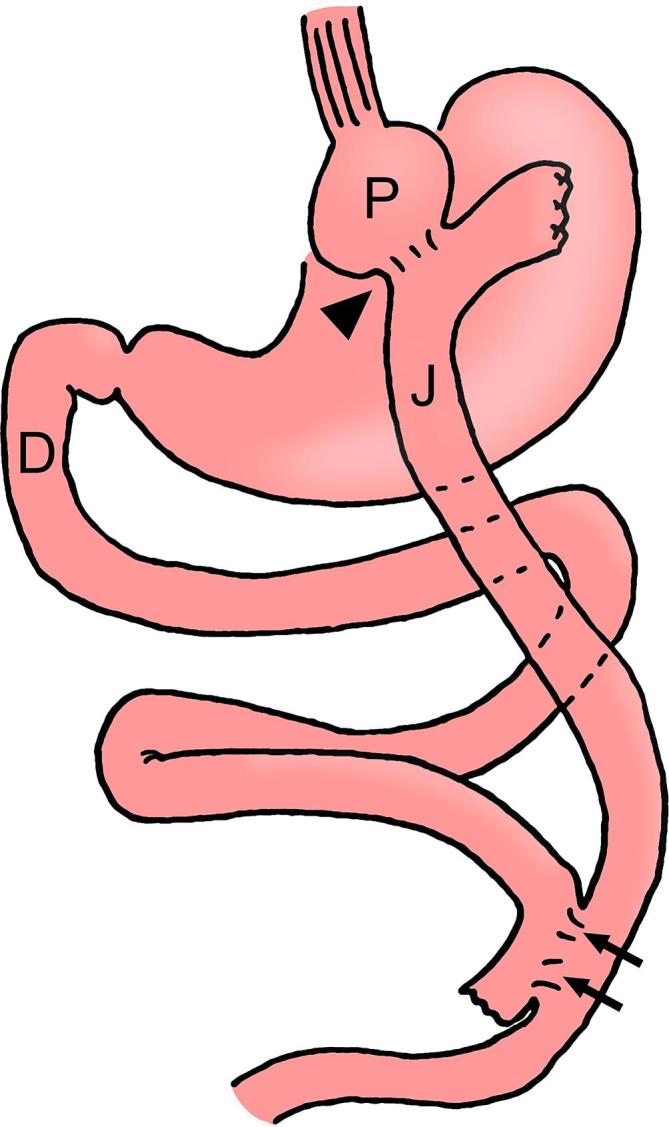
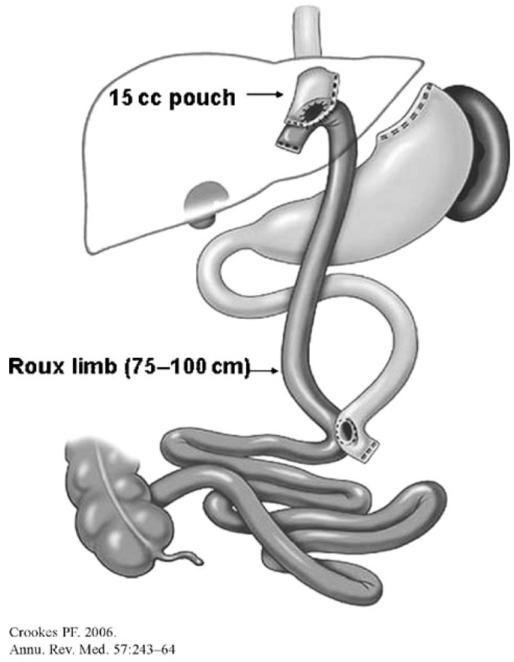
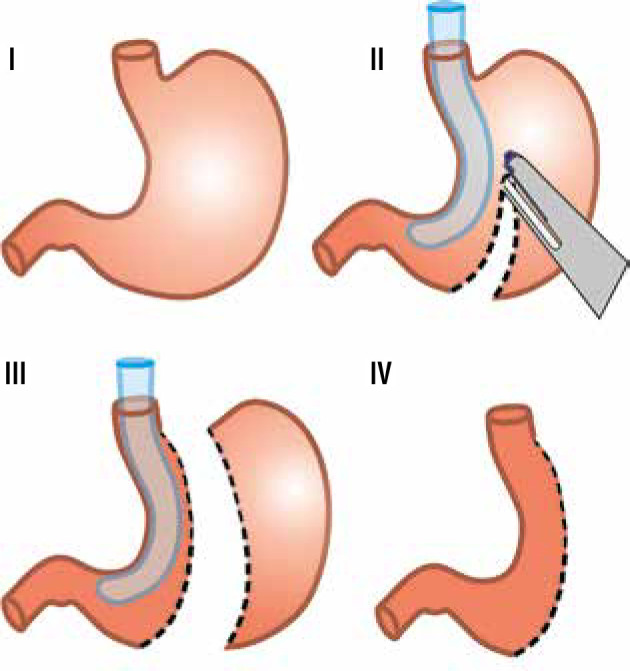
Sleeve Gastrectomy (SG)
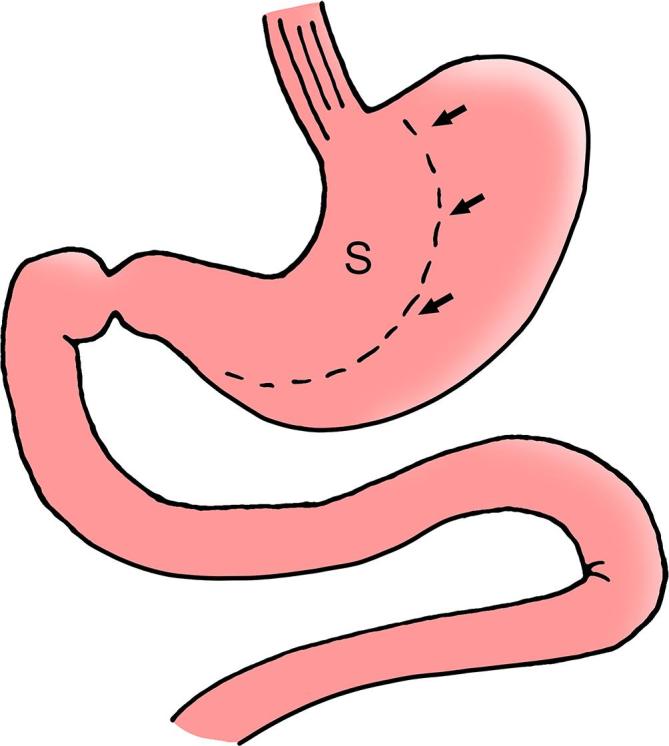
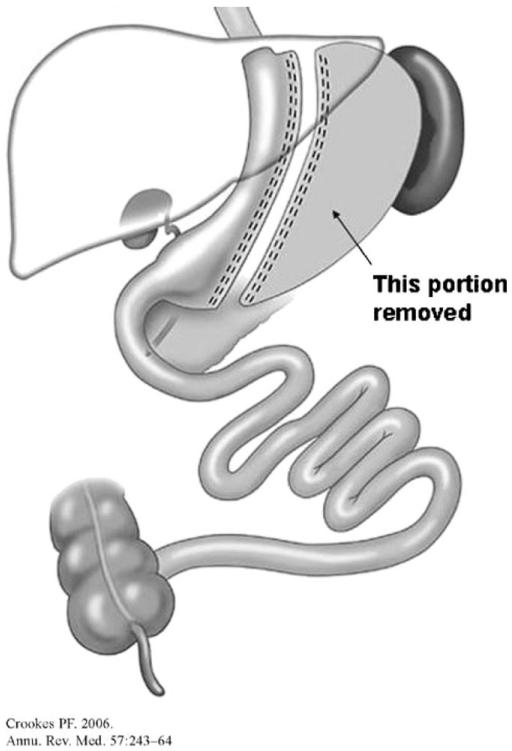
Now the most commonly performed bariatric operation worldwide. The greater curvature of the stomach is resected along a bougie (typically 36–40 Fr sizing catheter) from the antrum to the angle of His, removing roughly 70–80% of the stomach and leaving a narrow tubular gastric remnant. No anastomosis, no intestinal rerouting. Weight loss mechanism is restriction plus removal of ghrelin-producing fundus. Expected weight loss: 50–70% EWL at 1–2 years; good diabetes effect; may worsen or cause GERD. Landmark trials: SLEEVEPASS and SM-BOSS both demonstrated similar short- to medium-term weight-loss and comorbidity outcomes between SG and RYGB, with higher reflux rates after sleeve.
One-Anastomosis Gastric Bypass (OAGB / Mini-Gastric Bypass)
A long, narrow gastric pouch (15–20 cm) is created along the lesser curvature and anastomosed in an antecolic fashion to a loop of jejunum 150–200 cm distal to the ligament of Treitz, creating a single gastrojejunal anastomosis with a long biliopancreatic limb. Technically simpler than RYGB with one fewer anastomosis. Produces excellent weight loss and metabolic outcomes. Main concern is bile reflux into the pouch.
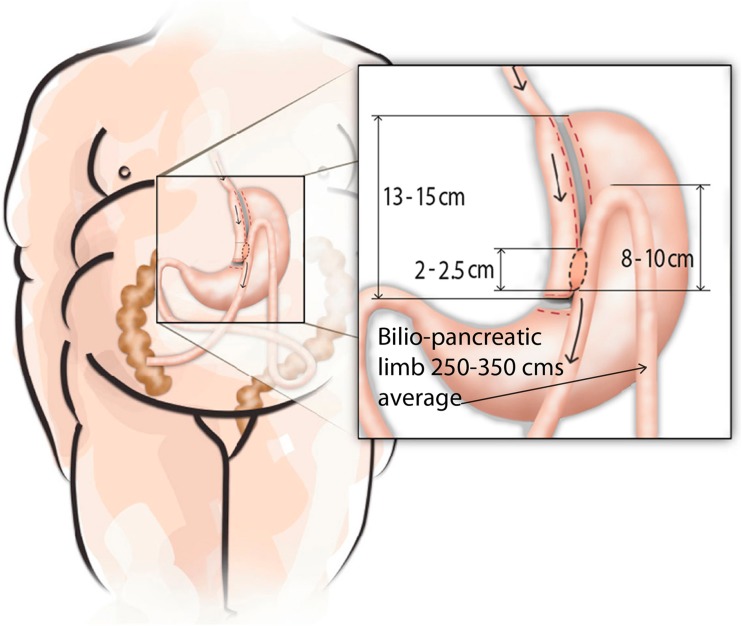
Duodenal Switch (BPD-DS) and SADI-S
The most powerful bariatric operations. Biliopancreatic diversion with duodenal switch (BPD-DS) combines a sleeve gastrectomy with division of the duodenum just distal to the pylorus, followed by a duodeno-ileostomy (alimentary limb ~150 cm) and an ileoileostomy (common channel ~100 cm). Produces 70–80%+ EWL and the highest rate of T2DM remission but with the highest nutrient deficiency risk. SADI-S (single anastomosis duodeno-ileal bypass with sleeve) is the simplified, single-anastomosis version with a duodeno-ileal loop ~250–300 cm from the ileocecal valve.
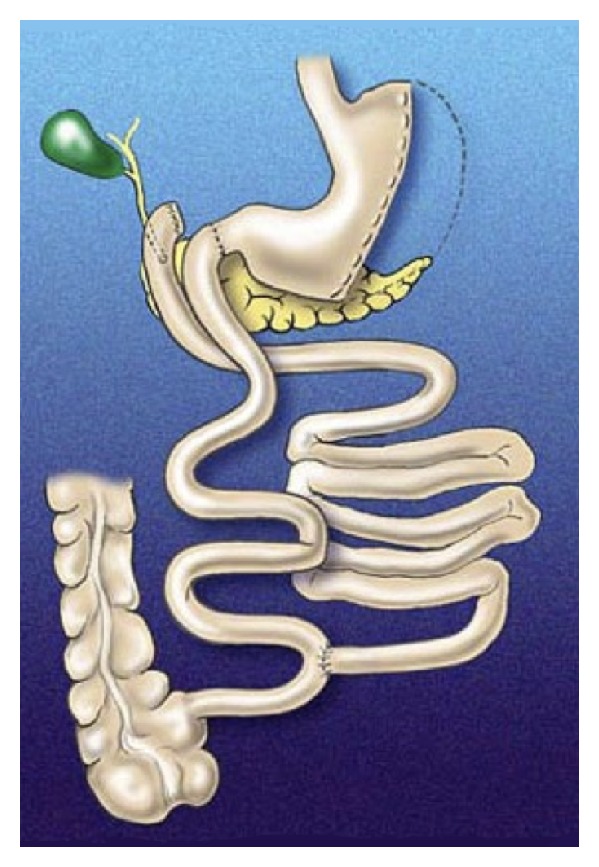
| Procedure | Mechanism | %EWL (1–2 yr) | T2DM Remission | Key Concern |
|---|---|---|---|---|
| RYGB | Restriction + malabsorption + hormonal | 60–80% | High | Internal hernia, marginal ulcer |
| Sleeve Gastrectomy | Restriction + ghrelin reduction | 50–70% | Moderate-high | GERD, staple line leak |
| OAGB | Restriction + malabsorption + hormonal | 65–80% | High | Bile reflux |
| BPD-DS / SADI-S | Restriction + high malabsorption + hormonal | 70–85% | Highest | Protein/fat-soluble vitamin deficiency |
| LAGB (historical) | Pure restriction | 40–50% | Low | Band slippage, erosion; mostly removed now |
IFSO grades weight loss success by %EWL: < 25% = failure, 25–50% = poor, 50–75% = good, > 75% = excellent. Targets at 1 year are typically ≥ 50% EWL for sleeve and ≥ 60% for RYGB; by 5 years most programs expect maintenance of at least 50% EWL.
13 Revisional & Endoscopic Bariatric Therapies Surgical
Revisional Surgery
Common conversion operations reflect the evolution of the field:
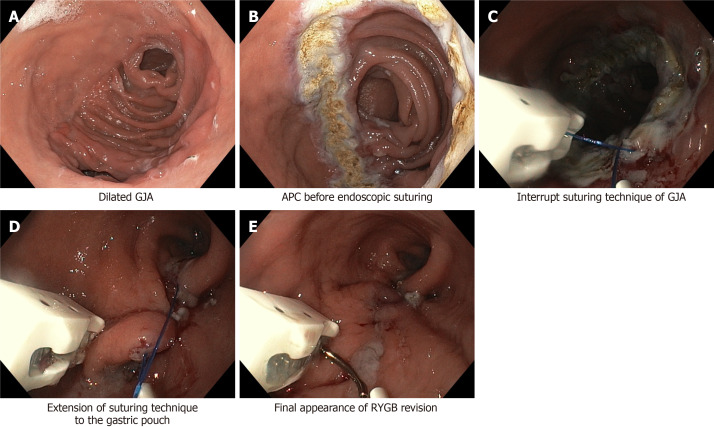
- LAGB → RYGB or SG: Adjustable gastric bands have largely been abandoned due to high complication and reoperation rates; most existing bands are being removed and converted.
- SG → RYGB: Typically for refractory GERD, Barrett's esophagus, or insufficient weight loss / weight regain.
- SG → BPD-DS or SADI-S: For inadequate weight loss in super-obese patients who had a planned staged approach.
- RYGB pouch revision: For weight regain, the gastric pouch can be reduced (either surgically or endoscopically) and the gastrojejunal anastomosis resized.
Endoscopic Bariatric Therapies
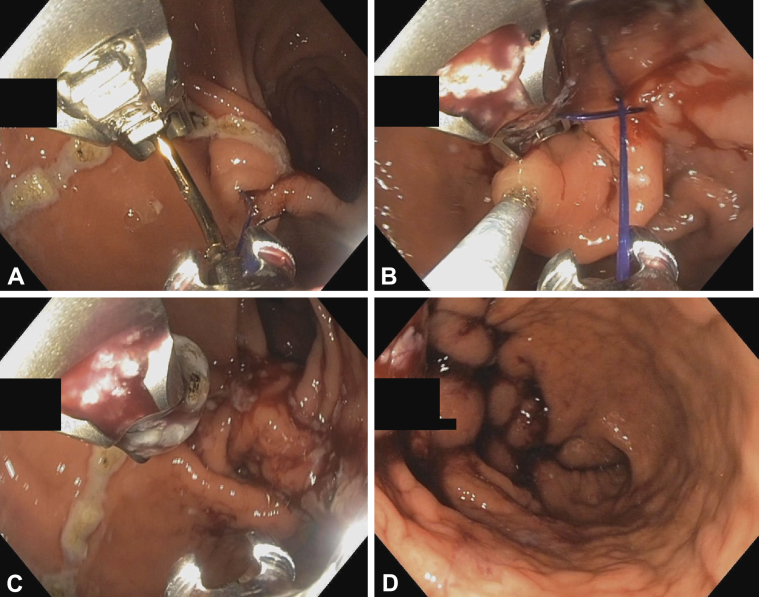
Endoscopic sleeve gastroplasty (ESG): Full-thickness endoscopic suturing plicates the stomach into a sleeve-like tube without resection. Typically produces 15–20% TWL.
Intragastric balloon: A saline- or gas-filled balloon placed endoscopically for 6 months; temporary restriction producing ~10–15% TWL.
Transoral outlet reduction (TORe): Endoscopic suturing to reduce the size of a dilated gastrojejunal anastomosis after RYGB, for weight regain.
Vagal blockade (vBloc/Maestro): Implanted device delivering high-frequency electrical signals to block vagal nerve activity. Rarely used today.
Endoscopic leak/stricture management: Stenting, clipping, or suturing of post-op staple-line leaks and anastomotic strictures.
14 Post-Operative Course & Nutrition Post-Op
Standard Post-Op Timeline
- POD 0–1: Clear liquids, ambulation, VTE prophylaxis, PPI start, SCDs, incentive spirometry. Most sleeve/bypass patients are discharged on POD 1–2.
- Week 1–2: Full liquids; protein shakes; chewable vitamins; wound checks.
- Week 2–4: Pureed diet; slow fluid progression; small frequent meals.
- Week 4–6: Soft diet; reintroduction of proteins.
- Week 6+: Regular bariatric diet — small portions, protein-first eating, no carbonated beverages, no drinking with meals.
Protein and Fluid Targets
Protein targets are typically 60–80 g/day (higher for BPD-DS, up to 100 g). Fluid targets are ≥ 64 oz/day, sipped between meals to avoid early satiety and dumping. Patients are instructed not to drink 30 minutes before, during, or 30 minutes after meals.
Follow-Up Schedule
Most programs see patients at 2 weeks, 6 weeks, 3 months, 6 months, 9 months, 12 months, and annually thereafter. Labs are checked at 3, 6, and 12 months then annually. Support group attendance is encouraged (or required) long-term.
15 Nutrient Deficiencies After Bariatric Surgery Post-Op
Every bariatric patient is at lifelong risk of micronutrient deficiency. The risk profile depends on the operation (malabsorptive procedures > restrictive) and on supplement adherence. The 2019 ASMBS nutritional guidelines codify screening and supplementation protocols.
| Nutrient | Mechanism | Clinical Finding | Supplementation |
|---|---|---|---|
| Iron | Reduced acid, bypassed duodenum | Microcytic anemia, fatigue, pica | Ferrous sulfate 65 mg elemental BID–TID; IV iron if malabsorption |
| Vitamin B12 | Loss of intrinsic factor production, bypassed ileum exposure | Macrocytic anemia, neuropathy, glossitis | Sublingual 500 mcg daily or IM 1000 mcg monthly |
| Folate | Inadequate intake, proximal jejunum bypass | Macrocytic anemia, elevated homocysteine | 400–800 mcg daily in MVI |
| Calcium | Reduced acid absorption, bypassed duodenum | Secondary hyperparathyroidism, bone loss | Calcium citrate 1200–1500 mg/day in divided doses |
| Vitamin D | Fat malabsorption, low baseline levels | Bone pain, osteomalacia | 3000 IU daily, titrated to 25-OH > 30 |
| Thiamine (B1) | Poor intake, vomiting, rapid weight loss | Wernicke's encephalopathy, beriberi, peripheral neuropathy | IV/IM in emesis or rapid weight loss; 12 mg/day in MVI |
| Zinc | Malabsorption | Hair loss, dysgeusia, delayed wound healing | 8–22 mg/day in MVI |
| Copper | Malabsorption, zinc over-supplementation | Myelopathy, anemia, neutropenia | 1–2 mg/day; monitor with zinc |
| Vitamin A | Fat malabsorption (DS/BPD) | Night blindness, xerophthalmia | 10,000 IU/day for DS; 5,000 IU/day for others |
| Vitamin K | Fat malabsorption | Coagulopathy, elevated INR | 90–120 mcg/day in MVI |
| Protein | Restriction, intolerance, malabsorption | Hypoalbuminemia, edema, sarcopenia, alopecia | 60–80 g/day (100+ for DS); protein shakes |
Thiamine deficiency is the most dangerous early nutritional emergency. The classic triad is ophthalmoplegia, ataxia, and confusion. It occurs most commonly in the first weeks to months after surgery, especially in patients with persistent vomiting or rapid weight loss. Any post-op bariatric patient with persistent vomiting needs IV thiamine BEFORE IV glucose — glucose without thiamine can precipitate or worsen Wernicke's. Document persistent emesis, thiamine administration, and neurologic findings carefully.
Refeeding Syndrome
In patients who have been severely nutritionally deprived — either from prolonged vomiting or very aggressive pre-op VLCDs — rapid nutritional reintroduction can trigger refeeding syndrome: hypophosphatemia, hypokalemia, hypomagnesemia, and thiamine deficiency with cardiovascular and neurologic decompensation. Gradual caloric reintroduction with pre-emptive electrolyte repletion is the prevention.
16 Surgical Complications Post-Op
The most feared early complication. Occurs in 1–3% of sleeves (typically at the angle of His) and 1–2% of gastric bypasses (at the gastrojejunostomy). Presents within 3–7 days post-op with tachycardia (> 120 often the first and only sign), fever, left-sided chest or shoulder pain, hypotension, and abdominal pain. Tachycardia in a fresh post-op bariatric patient is a leak until proven otherwise. Workup: CT with oral contrast, upper GI study, or diagnostic laparoscopy. Treatment: drainage, antibiotics, endoscopic stenting, or reoperation depending on severity.
Anastomotic Stricture / Stenosis
Narrowing of the gastrojejunostomy after RYGB (5–10%) or narrowing of the gastric sleeve at the incisura angularis (1–4%). Presents 4–12 weeks post-op with progressive intolerance to solids, regurgitation, and vomiting. Treatment is endoscopic balloon dilation (usually 15–18 mm); rarely requires surgical revision.
Marginal Ulcer
Ulceration at the gastrojejunal anastomosis after RYGB, occurring in 1–16% of patients. Risk factors: smoking, NSAID use, H. pylori, larger gastric pouch producing more acid, diabetes. Presents with epigastric pain, bleeding (melena, hematemesis), or perforation. Diagnosis: EGD. Treatment: high-dose PPI, sucralfate, smoking cessation, NSAID avoidance. All RYGB patients receive prophylactic PPI for the first 3–6 months.
Internal Hernia
Bowel herniates through defects created during the bypass — the Petersen's space (behind the Roux limb), the jejunojejunostomy mesenteric defect, or the transverse mesocolon defect (if retrocolic). Presents late (months to years post-op) with intermittent or acute crampy abdominal pain, nausea, and vomiting, often after significant weight loss. CT may be subtly abnormal or even normal — a "swirl sign" in the mesentery is classic. Diagnostic laparoscopy is often required because clinical suspicion alone justifies exploration. Untreated internal hernia can progress to bowel ischemia and necrosis. Always document abdominal pain in any post-RYGB patient with meticulous detail.
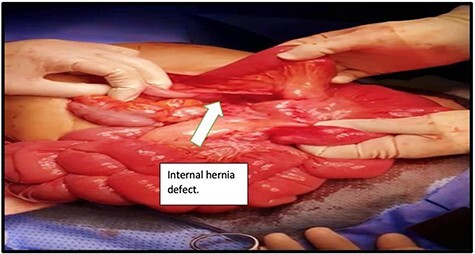
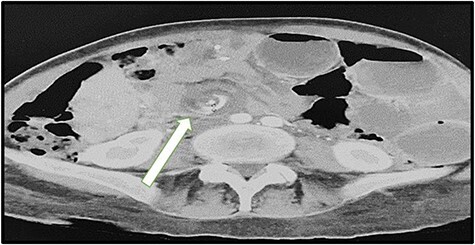
Gallstones
Rapid weight loss causes gallstone formation in up to 30% of bariatric patients during the first year. Many programs prescribe prophylactic ursodiol (UDCA) 300 mg BID for 6 months to reduce this risk. Symptomatic cholelithiasis is treated with laparoscopic cholecystectomy; after RYGB, ERCP is technically challenging because the biliary tree cannot be reached orally, and laparoscopic transgastric ERCP or EUS-directed transgastric ERCP (EDGE) is used instead.
Other Surgical Complications
- VTE (DVT/PE): Despite aggressive prophylaxis, PE remains a leading cause of 30-day mortality.
- Wound infection / incisional hernia: Lower with laparoscopic/robotic vs open approach.
- Small bowel obstruction: From adhesions, internal hernia, or intussusception at the jejunojejunostomy.
- Sleeve stenosis at incisura or twist: Classic presentation is "I can't keep liquids down."
17 Late Metabolic & Behavioral Complications Post-Op
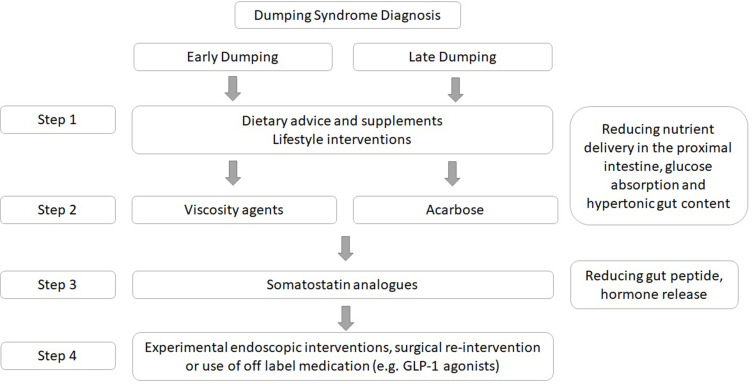
Dumping Syndrome
Early dumping (15–30 min after meals): Rapid delivery of hyperosmolar food into the small bowel triggers fluid shifts and GI hormone release. Symptoms: nausea, cramping, diarrhea, palpitations, diaphoresis, lightheadedness. Common after RYGB with high-sugar meals. Managed with dietary changes (avoid simple sugars, separate liquids from solids, small frequent meals).
Late dumping (1–3 hours after meals): Reactive hypoglycemia from exaggerated insulin response to rapid glucose absorption. Symptoms: sweating, tremor, weakness, confusion — classic hypoglycemic picture. Managed with complex carbs, protein at every meal, acarbose, sometimes octreotide.
Post-Bypass Hypoglycemia / Nesidioblastosis
A severe form of late dumping involving β-cell hypertrophy and inappropriate insulin release ("post-bariatric hypoglycemia"). Symptoms occur months to years post-op and include documented hypoglycemia with neuroglycopenic features. Workup includes mixed-meal tolerance test, continuous glucose monitoring, and insulin/C-peptide/proinsulin during symptomatic events. Treatment escalates from dietary modification to acarbose, diazoxide, octreotide, and — rarely — partial pancreatectomy or reversal of bypass.
Alcohol Use Disorder After RYGB
Roux-en-Y gastric bypass alters alcohol pharmacokinetics — peak blood alcohol levels are higher and reached faster. Combined with addiction transfer (substituting alcohol for food as a coping mechanism), this produces a demonstrably elevated rate of new-onset alcohol use disorder after RYGB. Screen at every visit using AUDIT-C and document counseling.
Bone Loss and Osteoporosis
Calcium and vitamin D malabsorption combined with reduced mechanical loading leads to progressive bone density loss, particularly after RYGB and DS. DEXA scans are commonly obtained at baseline and then every 1–2 years post-op; bisphosphonates are used cautiously because oral absorption is impaired.
18 Weight Regain & Body Contouring Post-Op
Weight Regain
Weight regain after bariatric surgery is common: most patients regain 5–10% of their lost weight after nadir (typically at 12–18 months post-op), and a subset regain significantly more. Contributing factors include dietary non-adherence, pouch dilation, gastrojejunal stoma dilation, inadequate follow-up, and underlying mental health or binge eating disorders. Management is a stepwise escalation: behavioral/dietary reinforcement → anti-obesity pharmacotherapy (GLP-1 agonists are highly effective post-op) → endoscopic revision (TORe, ESG of the pouch) → surgical revision or conversion.
Body Contouring After Massive Weight Loss
Massive weight loss (MWL) leaves redundant skin that often requires plastic surgery for functional and cosmetic reasons. Common procedures include panniculectomy, abdominoplasty, brachioplasty (arm lift), thighplasty, mastopexy, and lower body lift. Insurance coverage is inconsistent — most plans cover panniculectomy only when there is documented intertriginous infection, ulceration, or functional impairment. Patients should be at weight stability (typically > 12 months of stable weight) before contouring surgery.
19 Medications in Bariatric Care Medications
Anti-Obesity Medications (AOMs)
| Drug (Brand) | Class | Mechanism | Notes |
|---|---|---|---|
| Semaglutide (Wegovy, Ozempic) | GLP-1 agonist | Delayed gastric emptying, appetite suppression, incretin | ~15% TWL; weekly SC; GI side effects |
| Liraglutide (Saxenda, Victoza) | GLP-1 agonist | Same class | Daily SC; ~8% TWL |
| Tirzepatide (Mounjaro, Zepbound) | GIP/GLP-1 dual agonist | Dual incretin receptor agonism | ~20% TWL; weekly SC; highest efficacy |
| Phentermine | Sympathomimetic | Appetite suppression via NE release | Short-term (12 wk) per label; controlled substance |
| Phentermine / topiramate (Qsymia) | Combination | Appetite + satiety | Teratogenic — pregnancy test required |
| Naltrexone / bupropion (Contrave) | Combination | Reward pathway modulation | Avoid with seizure d/o, opioid use |
| Orlistat (Xenical, Alli) | Lipase inhibitor | Blocks fat absorption | Steatorrhea; fat-soluble vitamin malabsorption |
| Setmelanotide (Imcivree) | MC4R agonist | Melanocortin pathway | Rare genetic obesity syndromes only |
Peri-Operative Medications
- VTE prophylaxis: Enoxaparin 40 mg SC BID (higher than standard 40 mg daily because of weight-based dosing) or weight-based UFH; mechanical prophylaxis with SCDs. Extended prophylaxis (1–4 weeks post-discharge) for high-risk patients.
- Antibiotics: Cefazolin weight-adjusted (3 g for BMI ≥ 30 or > 120 kg) within 60 min of incision.
- PPI: Omeprazole, pantoprazole, or esomeprazole 20–40 mg daily for 3–6 months after RYGB for marginal ulcer prevention, and often after sleeve for GERD.
- Ursodiol (UDCA): 300 mg BID for 6 months post-op for gallstone prophylaxis.
- Antiemetics: Ondansetron 4 mg IV/ODT for routine post-op nausea; persistent emesis should prompt thiamine repletion before glucose-containing fluids.
- Supplementation: Chewable bariatric multivitamin BID, calcium citrate 1200–1500 mg/day, vitamin D 3000 IU/day, vitamin B12 (sublingual 500 mcg daily or IM 1000 mcg monthly), iron (in premenopausal women).
After RYGB and DS, extended-release medications are often poorly absorbed — switch to immediate-release formulations. NSAIDs are essentially contraindicated after RYGB because of marginal ulcer risk. Always document medication reconciliation with these rules in mind.
20 Classification & Risk Systems Reference
| %EWL | Outcome |
|---|---|
| > 75% | Excellent |
| 50–75% | Good |
| 25–50% | Poor |
| < 25% | Failure |
%EWL (Excess Weight Loss): ((Starting weight − current weight) / (Starting weight − ideal body weight at BMI 25)) × 100
%TWL (Total Weight Loss): ((Starting weight − current weight) / Starting weight) × 100
%EBMIL (Excess BMI Loss): ((Starting BMI − current BMI) / (Starting BMI − 25)) × 100
| Grade | Description |
|---|---|
| I | Any deviation from normal course; no pharmacologic (beyond antiemetics, analgesics, diuretics) or surgical intervention |
| II | Requires pharmacologic treatment including blood transfusion or TPN |
| IIIa | Requires intervention, not under general anesthesia |
| IIIb | Requires intervention under general anesthesia |
| IVa | Single organ dysfunction (including dialysis) |
| IVb | Multiorgan dysfunction |
| V | Death |
Target %EWL milestones typically documented at follow-up: 30% at 3 months, 50% at 6 months, 60–70% at 1 year, sustained ≥ 50% at 5 years.
21 Physical Exam in the Bariatric Patient Exam
The bariatric exam is focused but thorough. Key elements to document on every visit:
General: Appearance, mobility, distress level, body habitus.
Vitals: Weight (to nearest 0.1 kg), BMI, blood pressure (use appropriate large cuff), heart rate, oxygen saturation, waist circumference.
HEENT / Neck: Neck circumference (OSA screening), thyroid palpation, dentition (pre-op assessment).
Cardiovascular: Heart sounds, jugular venous distension, peripheral edema, peripheral pulses.
Respiratory: Air entry, effort, signs of obesity hypoventilation.
Abdomen: Scars (prior surgeries), hernias, panniculus assessment, tenderness, bowel sounds, masses. Document the number and location of laparoscopic port scars after prior bariatric surgery.
Skin: Intertrigo, stasis dermatitis, pretibial edema, venous ulcers, acanthosis nigricans (insulin resistance marker), striae, pressure injuries.
Extremities: Edema grading (1+ to 4+), pulses, joint mobility, gait.
Psychiatric: Mood, affect, engagement, insight.
22 Abbreviations Master List Reference
Anatomy & Procedures
Diagnoses & Comorbidities
Outcomes & Labs
Medications
23 Sample HPI Templates Reference
These templates show the level of detail and rhythm expected in a bariatric clinic note. Use them as patterns to internalize specialty-specific documentation.
"Ms. [Name] is a 42-year-old female with a BMI of 44 (weight 115 kg, height 162 cm) who presents for initial bariatric surgical consultation. Her highest lifetime weight is 118 kg. Comorbidities include poorly controlled type 2 diabetes (A1C 8.9% on metformin 1000 mg BID and glipizide 10 mg daily), hypertension on lisinopril 20 mg daily, dyslipidemia on atorvastatin 40 mg, moderate OSA on CPAP 9 cm H2O (compliant > 6 hours/night), and bilateral knee osteoarthritis limiting ambulation. She denies GERD, prior bariatric surgery, or eating disorder. Prior weight-loss attempts include Weight Watchers (lost 15 lb, regained), phentermine for 3 months in 2019 (lost 20 lb, regained), and a 6-month supervised diet through her PCP in 2024. She does not currently use alcohol or tobacco. She meets 2022 ASMBS/IFSO criteria for metabolic surgery. Today she is being educated about RYGB vs sleeve gastrectomy and will begin the multidisciplinary evaluation pathway."
"Mr. [Name] is a 51-year-old male, BMI 47, scheduled for laparoscopic sleeve gastrectomy in 3 weeks, presenting for final pre-op evaluation. Since his initial consult 5 months ago, he has completed dietitian education (5 visits), psychological evaluation (cleared, no contraindications), sleep study (moderate OSA, AHI 22, started on CPAP with good adherence), and screening EGD (small sliding hiatal hernia, LA grade A esophagitis, H. pylori negative). Labs today: A1C 6.8 (down from 7.5), 25-OH vitamin D 22 (started repletion), ferritin 35, B12 within normal limits, normal CBC, CMP, and coagulation studies. He has started the 2-week pre-op VLCD (Optifast) with good compliance. Weight today 138.4 kg, down 4 kg from his initial visit. He understands the operation, risks, expected course, and lifelong supplementation requirements. Consent reviewed and signed."
"Ms. [Name] is a 36-year-old female now 6 months s/p laparoscopic Roux-en-Y gastric bypass (150 cm Roux limb / 50 cm BPL). Pre-op weight 128 kg (BMI 46); current weight 94 kg (BMI 34), representing 26.6% TWL and 55% EWL. She is meeting protein targets (~70 g/day), hydrating well, and tolerating a regular bariatric diet. She takes her chewable bariatric MVI BID, calcium citrate 1500 mg/day in divided doses, vitamin D 3000 IU daily, sublingual B12 500 mcg daily, and iron (ferrous sulfate 325 mg daily). Comorbidity status: T2DM in remission (A1C 5.4, off all medications since POD #3), HTN resolved (off lisinopril), OSA — patient reports no snoring, repeat sleep study planned. Denies dumping symptoms, nausea, vomiting, abdominal pain, dysphagia, hair loss, or dysgeusia. Mood stable, attending monthly support group, walking 30 minutes daily."
"Mr. [Name] is a 48-year-old male 14 months s/p RYGB presenting with 3 weeks of progressive epigastric burning pain worse after meals, intermittent nausea, and one episode of dark stools yesterday. He admits to resuming smoking 5 months ago (now ½ PPD) and taking ibuprofen 400 mg several times per week for knee pain despite prior counseling. He discontinued his post-op PPI at 6 months. No hematemesis, fever, or weight loss. Vital signs stable; mild epigastric tenderness without peritonitis. Hemoglobin 10.2 (down from baseline 13.4). EGD scheduled urgently to evaluate for marginal ulcer; started on pantoprazole 40 mg BID, sucralfate 1 g QID, smoking cessation counseling reinforced, NSAIDs strictly prohibited. If ulcer confirmed, will repeat EGD in 8 weeks for healing verification."
"Ms. [Name] is a 54-year-old female 6 years s/p laparoscopic sleeve gastrectomy presenting with weight regain concerns. Pre-op weight 132 kg (BMI 49); nadir weight 78 kg (BMI 29) at 18 months post-op (41% TWL); current weight 102 kg (BMI 38), representing a 24 kg regain from nadir. She describes grazing throughout the day, increased carbohydrate intake, emotional eating related to a recent divorce, and loss of gym routine. She denies vomiting, dumping, or reflux. Comorbidities that had resolved (HTN) have recurred (now back on amlodipine 5 mg); A1C up to 6.1 from 5.4. Discussed re-engagement with the dietitian and behavioral health, initiation of GLP-1 agonist therapy (semaglutide), and evaluation for possible conversion to RYGB vs endoscopic revision if medical and behavioral approaches fail."
"Mr. [Name] is a 39-year-old male with a BMI of 38, type 2 diabetes (A1C 7.6 on metformin), and hyperlipidemia, presenting to discuss anti-obesity pharmacotherapy. He is not currently pursuing bariatric surgery but wants structured weight-loss treatment. He has no personal or family history of medullary thyroid cancer or MEN2, no history of pancreatitis, and normal lipase. Discussed class effect of GLP-1 receptor agonists including dose titration, expected GI side effects (nausea, constipation, early satiety), injection technique, and monitoring plan. Plan: initiate semaglutide 0.25 mg SC weekly, titrate monthly per label to a target of 2.4 mg weekly (Wegovy dosing). Will monitor A1C, weight, and tolerability at 1, 3, and 6 months. Dietitian referral placed. Counseled on pregnancy avoidance and contraception planning (not directly relevant to this patient but standard documentation)."
24 References & Sources
Clinical Practice Guidelines
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: Body Mass Index Chart. Wikimedia Commons. Public domain.
Figure 2: Bariatric Surgery Comparison Diagram. Wikimedia Commons. Public domain.
Figure 3: Stomach Anatomy (OpenStax Anatomy & Physiology, Fig. 24.21). Wikimedia Commons, OpenStax College. CC BY 3.0.
Figure 4: Roux-en-Y Gastric Bypass. Wikimedia Commons. Public domain.
Figure 5: Sleeve Gastrectomy. Wikimedia Commons. Public domain.
The best bariatric scribes understand that every note is telling two stories at once: a surgical story (pouch size, limb lengths, staple line, complications) and a metabolic story (A1C trajectory, blood pressure control, OSA resolution, nutrient status). When the surgeon says "sleeve at 36 Fr bougie, oversewn staple line, no leak on methylene blue test," you are writing that down verbatim — but you also know why it matters. When a 6-month post-op patient reports cramping after meals with palpitations and diaphoresis, you know to document "likely early dumping" and anticipate dietary counseling.
Be protective of your patients. Tachycardia in a fresh post-op is a leak until proven otherwise. Confusion with vomiting is Wernicke's until proven otherwise. Crampy abdominal pain months after RYGB is an internal hernia until proven otherwise. Your documentation of these red flags — timestamps, vital trends, specific symptom descriptions — is what drives the team's response.
Welcome to bariatric and metabolic surgery. Patients here are trusting the team with a chronic disease that has shaped their entire lives, and your notes are part of how that care becomes real.