Cardiology
Every diagnosis, arrhythmia, valve lesion, procedure, medication, classification, and documentation framework you need to succeed in a cardiology clinic, CCU, or cath lab on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Cardiac Anatomy & Physiology Essentials Foundations
Cardiology is built on a tight coupling of anatomy, electrical conduction, and hemodynamic physiology. Every diagnosis in this guide — from a subtle ECG change to a failing left ventricle — becomes understandable once you can picture where the problem lives inside the chest and what job that structure is supposed to do.
Chambers, Valves, and Great Vessels
The heart is a four-chambered pump divided into a right side (receives deoxygenated blood and sends it to the lungs) and a left side (receives oxygenated blood and pumps it to the body). Blood returning from the body enters the right atrium (RA) through the superior and inferior vena cavae, crosses the tricuspid valve into the right ventricle (RV), is ejected through the pulmonic valve into the main pulmonary artery, and returns oxygenated from the lungs via the four pulmonary veins into the left atrium (LA). From the LA it crosses the mitral valve into the left ventricle (LV), then out the aortic valve into the aorta and the systemic circulation.
The aortic and mitral valves guard the high-pressure left heart and take the brunt of degenerative disease; the tricuspid and pulmonic valves sit in the low-pressure right heart and are more often affected by endocarditis, congenital disease, or secondary dilation.
Coronary Arteries
The heart muscle itself is fed by the coronary arteries, which arise from the aortic root just above the aortic valve. The left main coronary artery (LM) splits into the left anterior descending (LAD) — supplying the anterior wall, anterior septum, and apex of the LV — and the left circumflex (LCx), which wraps laterally and feeds the lateral LV wall. The right coronary artery (RCA) feeds the RV, the inferior LV wall in most patients, and critically supplies the SA node (in ~60%) and AV node (in ~90%). Coronary dominance refers to whichever artery gives off the posterior descending artery (PDA) — right-dominant in about 85% of people.
Conduction System
The electrical impulse that drives every heartbeat originates in the sinoatrial (SA) node at the junction of the RA and superior vena cava. It spreads across both atria (producing the P wave), reaches the atrioventricular (AV) node in the interatrial septum (PR interval — the physiological delay that lets the atria finish emptying), then races down the bundle of His, splits into the right and left bundle branches, and terminates in the Purkinje fibers that depolarize the ventricles (QRS complex). Ventricular repolarization produces the T wave.
The Cardiac Cycle & Hemodynamics
A single heartbeat is divided into systole (ventricular contraction and ejection) and diastole (ventricular relaxation and filling). Stroke volume (SV) is the blood ejected per beat, cardiac output (CO) = SV × heart rate, and ejection fraction (EF) = SV ÷ end-diastolic volume — the single most important number in clinical cardiology. Normal LVEF is 52–72% in men and 54–74% in women by echocardiographic consensus.
Three levers determine how much blood the heart moves: preload (the stretch on the ventricle at end-diastole, approximated by central venous or wedge pressure), afterload (the resistance the ventricle must eject against, approximated by systemic vascular resistance and blood pressure), and contractility (the intrinsic force of myocardial shortening). Every cardiology drug you will chart manipulates one or more of these three variables.
Memorize the EF categories cold: HFrEF (reduced) is EF ≤ 40%, HFmrEF (mildly reduced) is 41–49%, and HFpEF (preserved) is ≥ 50%. The entire GDMT (guideline-directed medical therapy) decision tree branches off this single number, and you will hear it in almost every heart failure note.
02 Scribe Documentation Framework Foundations
Cardiology notes have their own rhythm. A good cardiology HPI is not just "chest pain for 2 days" — it quantifies exertion tolerance, maps symptoms to hemodynamic categories, and anchors every complaint to objective data (ECG, troponin, BNP, echo EF).
Chief Complaint (CC): Usually one of: chest pain, shortness of breath, palpitations, syncope/presyncope, lower extremity edema, or "follow-up" of a known diagnosis (CAD, HF, AFib, valve disease).
HPI — OLDCARTS plus cardiac layers: Onset (sudden vs gradual), Location (substernal, radiating to jaw/arm), Duration, Character (pressure, tearing, sharp, burning), Aggravating factors (exertion, deep breath, lying flat), Relieving factors (rest, nitroglycerin, sitting up), Timing, Severity (0–10). Always capture: functional capacity in METs or NYHA class, orthopnea, PND, exertional dyspnea quantified in blocks/flights of stairs, lightheadedness, and any witnessed loss of consciousness.
ROS highlights: CV (chest pain, palpitations, DOE, PND, orthopnea, edema, claudication), pulmonary (cough, hemoptysis, wheeze), neuro (syncope, TIA, focal deficits), GI (early satiety, RUQ fullness from hepatic congestion).
PMHx/PSHx: HTN, DM, HLD, CKD, tobacco use (pack-years), prior MI, stents (vessel, date), CABG, valve replacements, pacemaker/ICD, AFib with current rhythm/rate strategy and CHA₂DS₂-VASc.
Vitals: BP in both arms if coarctation or dissection suspected, HR, RR, SpO₂, weight (critical for HF patients — the trend is more useful than any single value).
Exam: JVP in cm above the sternal angle, carotid upstroke, PMI location, S1/S2, added sounds (S3 = volume overload/HFrEF, S4 = stiff ventricle/HTN/HCM), murmurs (grade, location, radiation, maneuvers), lungs (crackles, effusion), abdomen (hepatomegaly, hepatojugular reflux, ascites), extremities (edema, pulses, perfusion).
Data: Most recent ECG (rate, rhythm, axis, intervals, ischemic changes), troponin trend, BNP/NT-proBNP, BMP (esp. K+, Cr, eGFR), CBC, INR or DOAC, lipid panel, HbA1C, TSH (new AFib), most recent echo EF and valve assessments, stress test results, cath report.
Cardiology A&P is almost always organized by problem: (1) CAD s/p DES to LAD 2022 — continue DAPT, high-intensity statin, ACEi; (2) HFrEF EF 30% — optimize quad therapy (BB, ARNI, MRA, SGLT2i), daily weights, 2 g Na diet; (3) AFib CHA₂DS₂-VASc 4 — continue apixaban, rate control with metoprolol; (4) HTN — add amlodipine; (5) HLD — atorvastatin 80 mg.
For every chest pain HPI, document the five features that determine pre-test probability of ACS: character (pressure vs sharp), radiation, exertional trigger, duration, and associated symptoms (diaphoresis, nausea, dyspnea). Cardiologists will scan your HPI looking for these — miss them and the note is incomplete.
03 Hypertension (HTN) Vascular
Hypertension is the most common diagnosis in any cardiology clinic and the most important modifiable risk factor for stroke, MI, heart failure, and chronic kidney disease. Under the 2017 ACC/AHA hypertension guideline, stage 1 HTN begins at 130/80 mmHg rather than the older 140/90 threshold.
Classification
| Category | Systolic (mmHg) | Diastolic (mmHg) |
|---|---|---|
| Normal | < 120 | and < 80 |
| Elevated | 120–129 | and < 80 |
| Stage 1 HTN | 130–139 | or 80–89 |
| Stage 2 HTN | ≥ 140 | or ≥ 90 |
| Hypertensive urgency | ≥ 180 | or ≥ 120 (no end-organ damage) |
| Hypertensive emergency | ≥ 180 | or ≥ 120 + acute end-organ damage |
Primary (essential) HTN accounts for ~90% of cases and has no single identifiable cause. Secondary HTN should be considered when onset is < 30 or > 55, BP is resistant to 3+ drugs, or there are clues on exam/labs. Causes to know: renal artery stenosis (abdominal bruit, flash pulmonary edema after ACEi), primary aldosteronism (hypokalemia, suppressed renin), pheochromocytoma (paroxysmal HTN, headache, palpitations, sweating), Cushing syndrome, obstructive sleep apnea, coarctation of the aorta (arm-leg BP gradient), and thyroid disease.
Management
First-line agents are thiazide diuretics (chlorthalidone, HCTZ), ACE inhibitors, ARBs, and dihydropyridine calcium channel blockers (amlodipine). In Black adults without HF or CKD, thiazides and CCBs are preferred as initial monotherapy. BP goal is generally < 130/80 for most adults with cardiovascular disease or elevated ASCVD risk. Resistant HTN (uncontrolled on 3 agents including a diuretic) warrants evaluation for secondary causes and consideration of spironolactone as the fourth agent.
Requires IV therapy (nicardipine, labetalol, clevidipine, nitroprusside) in a monitored setting. Lower MAP by no more than 20–25% in the first hour to avoid cerebral, cardiac, or renal hypoperfusion — except in aortic dissection (target SBP < 120 and HR < 60 within minutes) or acute ischemic stroke (permissive HTN unless eligible for tPA).
Document whether the BP reading was in-office, ambulatory, or home — and always note if the patient was recently caffeinated, anxious, or in pain. "White-coat HTN" is real and often changes the plan.
04 Coronary Artery Disease & Stable Angina Ischemic
CAD is atherosclerotic narrowing of one or more epicardial coronary arteries. When a stenosis becomes flow-limiting (generally ≥ 70% angiographic or fractional flow reserve ≤ 0.80), the myocardium distal to the lesion cannot meet oxygen demand during exertion, producing stable angina — predictable, reproducible chest pressure on exertion or emotional stress, relieved within minutes by rest or sublingual nitroglycerin.
Canadian Cardiovascular Society (CCS) Angina Classification
| CCS Class | Angina Threshold | Functional Impact |
|---|---|---|
| I | Only with strenuous, rapid, or prolonged exertion | Ordinary activity does not cause angina |
| II | Slight limitation of ordinary activity (walking > 2 blocks, climbing > 1 flight) | Mild limitation |
| III | Marked limitation — angina walking 1–2 blocks or climbing 1 flight at normal pace | Moderate limitation |
| IV | Inability to carry out any physical activity without discomfort; angina at rest | Severe, disabling |
Diagnostic Workup
Resting ECG (often normal between episodes), high-sensitivity troponin (should be negative in stable angina), lipid panel, HbA1C, and functional or anatomic testing. Functional options include exercise treadmill ECG, stress echocardiography, and nuclear myocardial perfusion imaging (SPECT or PET). Anatomic testing is increasingly dominated by coronary CT angiography (CCTA), which carries a class I recommendation for intermediate-risk patients in the 2023 ACC/AHA chronic coronary disease guideline. Invasive coronary angiography is reserved for high-risk findings or persistent symptoms despite medical therapy.
Antiplatelet: aspirin 81 mg daily (clopidogrel if ASA intolerant). Statin: high-intensity (atorvastatin 40–80, rosuvastatin 20–40) targeting LDL < 70, often < 55 in very high-risk patients. Anti-anginal: beta-blocker first-line, add long-acting nitrate or CCB if symptoms persist, ranolazine as add-on. ACEi/ARB: indicated if HTN, DM, CKD, or LVEF < 40%. Lifestyle: smoking cessation, Mediterranean diet, 150 min/week exercise, weight control.
The ISCHEMIA trial showed that in stable CAD with moderate-to-severe ischemia, an initial invasive strategy did not reduce death or MI compared with optimal medical therapy — reinforcing that revascularization is for symptoms refractory to GDMT or for specific anatomy (left main, proximal LAD, multivessel disease with reduced EF).
05 Acute Coronary Syndromes (UA, NSTEMI, STEMI) Ischemic
ACS is the spectrum of clinical presentations caused by acute reduction in coronary blood flow, almost always from atherosclerotic plaque rupture and thrombus formation. It includes three overlapping entities: unstable angina (UA), NSTEMI, and STEMI.
Unstable angina: new-onset angina, angina at rest, or accelerating pattern — WITHOUT troponin elevation and without ST elevation. Rare in the high-sensitivity troponin era.
NSTEMI: ischemic symptoms + troponin rise/fall + no persistent ST elevation. ECG may show ST depression, T-wave inversion, or be normal.
STEMI: ischemic symptoms + persistent ST elevation in two contiguous leads (≥ 1 mm in limb leads, ≥ 2 mm in V2–V3 for men ≥ 40), or new LBBB with ischemic presentation. Represents complete occlusion of an epicardial vessel.
STEMI Territories
| ECG Leads | Territory | Culprit Vessel |
|---|---|---|
| II, III, aVF | Inferior | RCA (85%) or LCx |
| V1–V2 | Septal | LAD (septal branches) |
| V3–V4 | Anterior | LAD |
| V5–V6, I, aVL | Lateral | LCx or LAD diagonal |
| V1–V6 + I, aVL | Extensive anterior | Proximal LAD / left main |
| ST depression V1–V3 with tall R | Posterior | RCA or LCx |
| V3R, V4R ST elevation | Right ventricle | Proximal RCA |
Management
For STEMI, the clock starts at first medical contact. The goal is door-to-balloon time < 90 minutes for primary PCI or < 120 min for transfer to a PCI-capable center, per the 2013 ACC/AHA STEMI guideline. If PCI is not achievable within 120 minutes, fibrinolytic therapy (tenecteplase, alteplase) is given within 30 minutes of arrival.
For NSTEMI, risk stratification drives timing: very high-risk features (hemodynamic instability, refractory angina, mechanical complications, life-threatening arrhythmia) warrant immediate angiography; high-risk features (GRACE > 140, dynamic ST changes, troponin rise) get an early invasive strategy within 24 hours per the 2014 ACC/AHA NSTEMI guideline.
Antiplatelet: aspirin 325 mg chewed + P2Y₁₂ inhibitor (ticagrelor 180 mg, prasugrel 60 mg, or clopidogrel 600 mg). Anticoagulation: UFH or enoxaparin. Statin: high-intensity (atorvastatin 80 mg) as early as possible. Beta-blocker: oral within 24 h if hemodynamically stable (avoid in HF/cardiogenic shock). Nitroglycerin: SL or IV for ongoing chest pain/HTN (avoid in RV infarct, recent PDE5 inhibitor use, SBP < 90). Oxygen: only if SpO₂ < 90%. ACEi/ARB: within 24 h if LVEF < 40%, HF, HTN, or DM.
Killip Classification (STEMI hemodynamic severity)
| Class | Clinical Findings | In-Hospital Mortality |
|---|---|---|
| I | No signs of heart failure | ~6% |
| II | S3 gallop, bibasilar crackles, JVD | ~17% |
| III | Acute pulmonary edema | ~38% |
| IV | Cardiogenic shock (SBP < 90, end-organ hypoperfusion) | ~81% |
GRACE Risk Score
The GRACE score estimates in-hospital and 6-month mortality in ACS using age, HR, SBP, creatinine, Killip class, cardiac arrest at admission, ST deviation, and elevated cardiac enzymes. Scores > 140 identify high-risk NSTEMI patients who benefit from early invasive strategy; scores < 109 are low risk.
For any STEMI activation, document the exact times: symptom onset, EMS arrival, first ECG, cath lab activation, and first device (balloon) inflation. These timestamps drive quality metrics and reimbursement; missing them is a common scribe failure.
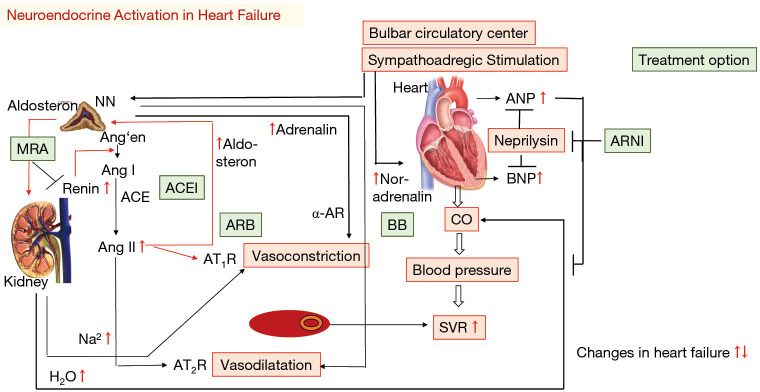
06 Heart Failure (HFrEF, HFpEF, HFmrEF) Failure
Heart failure is a clinical syndrome in which structural or functional cardiac abnormality impairs the heart's ability to fill with or eject blood to meet metabolic demands. It is the final common pathway of almost every form of cardiac disease and the leading cause of hospital readmission in the United States.
Classification by Ejection Fraction
| Type | LVEF | Typical Pathophysiology |
|---|---|---|
| HFrEF | ≤ 40% | Reduced systolic function — ischemic cardiomyopathy, dilated CMP, post-MI |
| HFmrEF | 41–49% | Mid-range; may recover or progress |
| HFpEF | ≥ 50% | Diastolic dysfunction — HTN, obesity, DM, aging, HCM |
| HFimpEF | Baseline ≤ 40% now > 40% | Recovered EF — continue GDMT |
ACC/AHA Stages and NYHA Functional Class
Stage A: At risk (HTN, DM, CAD, cardiotoxic drugs, family history) — no structural disease, no symptoms.
Stage B: Structural heart disease (LVH, low EF, prior MI, valve disease) — no current or prior symptoms.
Stage C: Structural disease + current or prior symptoms of HF.
Stage D: Refractory HF requiring specialized interventions (inotropes, LVAD, transplant, hospice).
Class I: No limitation of physical activity. Ordinary activity does not cause symptoms.
Class II: Slight limitation. Comfortable at rest; ordinary activity causes symptoms.
Class III: Marked limitation. Comfortable at rest; less than ordinary activity causes symptoms.
Class IV: Symptoms at rest; any activity increases discomfort.
Diagnostic Workup
BNP or NT-proBNP (BNP > 100 or NT-proBNP > 300 supports HF; levels are lower in obesity, higher in AFib and CKD), 12-lead ECG, CXR (cardiomegaly, Kerley B lines, pulmonary edema, effusions), transthoracic echo (EF, wall motion, valves, diastolic function, RV function, estimated PA pressure), BMP (Na, K, Cr), TSH, HbA1C, iron studies (iron deficiency is common and treatable), and consideration of ischemic workup.
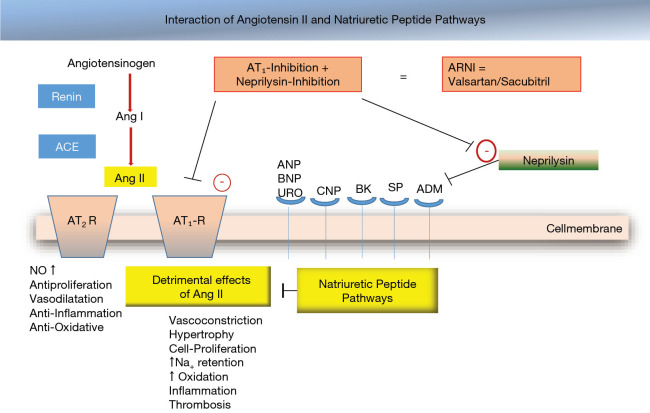
Guideline-Directed Medical Therapy (HFrEF — Quadruple Therapy)
The 2022 AHA/ACC/HFSA heart failure guideline established "the four pillars" of HFrEF therapy, all four of which should be initiated and titrated as tolerated:

1. ARNI / ACEi / ARB: Sacubitril/valsartan (Entresto) is preferred based on PARADIGM-HF showing superior mortality vs enalapril. Use ACEi (lisinopril, enalapril) or ARB (valsartan, losartan) if ARNI not feasible.
2. Beta-blocker: Only three are proven in HFrEF — carvedilol, metoprolol succinate (Toprol XL), and bisoprolol.
3. Mineralocorticoid receptor antagonist (MRA): Spironolactone or eplerenone. Monitor K+ and Cr.
4. SGLT2 inhibitor: Dapagliflozin or empagliflozin, regardless of diabetes status. EMPEROR-Reduced and DAPA-HF both showed reduction in HF hospitalization and CV death.
Additional agents when appropriate: loop diuretics (furosemide, torsemide, bumetanide) for volume overload, hydralazine + isosorbide dinitrate (especially in Black patients or ACEi intolerance), ivabradine for HR > 70 in sinus rhythm despite maximal BB, and digoxin for symptom control. Device therapy: ICD for primary prevention if EF ≤ 35% despite 3 months of GDMT, and CRT if EF ≤ 35% with LBBB and QRS ≥ 150 ms.
HFpEF Management
Historically frustrating, HFpEF now has its first evidence-based therapy: SGLT2 inhibitors have a Class I recommendation based on EMPEROR-Preserved and DELIVER. MRAs may be considered. The rest of management targets comorbidities: aggressive BP control, diuretics for congestion, AFib rate/rhythm control, and weight loss.
In any HF note, document: (1) EF and date of echo, (2) NYHA class TODAY, (3) ACC/AHA stage, (4) current weight vs "dry weight," (5) each GDMT agent with dose, and (6) any GDMT agents NOT on board with reason (e.g., "MRA held for K+ 5.2"). This is the structure attendings expect.
07 Atrial Fibrillation & Atrial Flutter Arrhythmia
AFib is the most common sustained arrhythmia, characterized by disorganized atrial electrical activity, loss of effective atrial contraction, and an irregularly irregular ventricular response. It affects 1 in 4 adults over their lifetime and is the leading cardiac cause of embolic stroke.
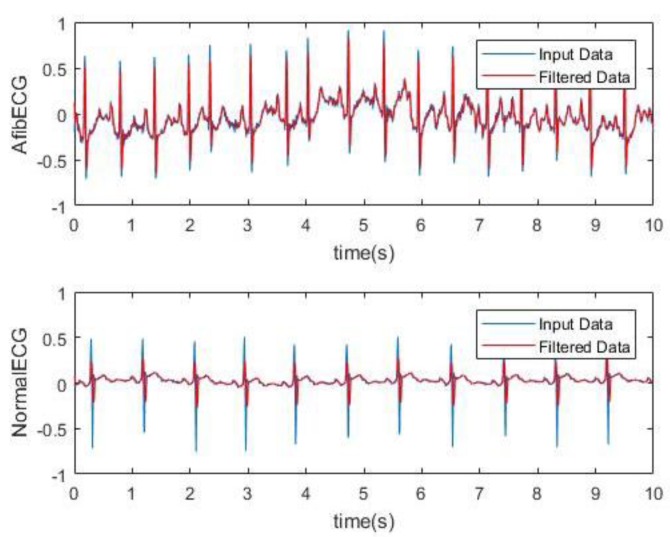
Classification
| Type | Duration |
|---|---|
| Paroxysmal | Self-terminating < 7 days (usually < 48 h) |
| Persistent | > 7 days; requires cardioversion to terminate |
| Long-standing persistent | > 12 months continuous |
| Permanent | Accepted; no further rhythm control attempts |
Stroke Risk — CHA₂DS₂-VASc
C — CHF (1) | H — HTN (1) | A₂ — Age ≥ 75 (2) | D — DM (1) | S₂ — prior Stroke/TIA/TE (2) | V — Vascular disease: prior MI, PAD, aortic plaque (1) | A — Age 65–74 (1) | Sc — Sex category female (1)
Anticoagulation thresholds: Score 0 (male) or 1 (female) — no therapy. Score 1 (male) — consider. Score ≥ 2 (male) or ≥ 3 (female) — anticoagulation recommended.
Bleeding Risk — HAS-BLED
HTN (SBP >160), Abnormal renal/liver function (1 each), Stroke, Bleeding history/predisposition, Labile INR, Elderly (> 65), Drugs (antiplatelets, NSAIDs) or alcohol (1 each). Score ≥ 3 indicates high bleeding risk — not a contraindication to AC, but a trigger to address modifiable factors.
Management Framework — Rate vs Rhythm, Anticoagulation, Upstream
The 2023 ACC/AHA/ACCP/HRS AFib guideline restructured AFib management around four pillars: Stroke risk and Stroke prevention, Better symptom control (rate/rhythm), Cardiovascular and comorbidity optimization, and Evaluation with risk factor modification.
Rate control: beta-blockers (metoprolol, carvedilol), non-dihydropyridine CCBs (diltiazem, verapamil — avoid in HFrEF), and digoxin as add-on. Target resting HR < 110 (lenient) or < 80 (strict, in symptomatic patients).
Rhythm control: Direct current cardioversion, antiarrhythmic drugs (flecainide/propafenone in structurally normal hearts; amiodarone, dofetilide, sotalol, dronedarone in structural disease), and catheter ablation (pulmonary vein isolation). The EAST-AFNET 4 trial and newer ablation trials support early rhythm control in selected patients.
Anticoagulation: DOACs (apixaban, rivaroxaban, dabigatran, edoxaban) are preferred over warfarin in non-valvular AFib. Warfarin remains indicated in mechanical valves and moderate-to-severe mitral stenosis. Left atrial appendage occlusion (Watchman) is an alternative in patients with long-term contraindications to anticoagulation.
Atrial Flutter
A macro-reentrant atrial tachycardia, classically with "sawtooth" flutter waves (negative in II, III, aVF for typical counterclockwise flutter) and an atrial rate of ~300 with 2:1 AV conduction (ventricular rate ~150). Stroke risk and anticoagulation mirror AFib. Cavotricuspid isthmus ablation is highly effective (> 95% success) and often curative for typical flutter.
Always document the CHA₂DS₂-VASc score numerically in the note (not just "moderate risk"). Also document time of AFib onset if known — the 48-hour window matters for cardioversion without prior TEE or 3 weeks of anticoagulation.
08 Supraventricular Tachycardias (SVT) Arrhythmia
SVT is any tachyarrhythmia (HR > 100) originating at or above the AV node. In common usage the term usually refers to the paroxysmal regular narrow-complex tachycardias: AV nodal reentrant tachycardia (AVNRT), AV reentrant tachycardia (AVRT) involving an accessory pathway (e.g., WPW), and atrial tachycardia.
AVNRT is the most common, typically presenting in young women with sudden-onset palpitations, sometimes neck pounding (from atrial contraction against closed AV valves), at rates of 150–220. AVRT involves a macro-reentrant circuit through an accessory pathway; WPW syndrome includes a short PR, delta wave, and widened QRS during sinus rhythm, with risk of pre-excited AFib (can degenerate to VF).
Stable: Vagal maneuvers (Valsalva, modified Valsalva with leg elevation, carotid sinus massage) first, then IV adenosine 6 mg rapid push, followed by 12 mg if no response. IV beta-blocker or diltiazem as alternatives.
Unstable: Synchronized cardioversion 50–100 J. In pre-excited AFib, AVOID AV-nodal blockers (adenosine, BB, CCB, digoxin) — they can accelerate conduction down the accessory pathway and precipitate VF. Use procainamide, ibutilide, or cardioversion.
Long-term management for recurrent SVT is catheter ablation — success rates > 95% with very low complication rates. Beta-blockers or CCBs can be used as bridge therapy.
09 Ventricular Arrhythmias (VT/VF) Arrhythmia
Ventricular tachycardia is a wide-complex (> 120 ms) tachyarrhythmia originating below the bundle of His. It is classified by duration (sustained ≥ 30 sec or causing hemodynamic compromise, vs non-sustained) and morphology (monomorphic vs polymorphic).
Monomorphic VT: Usually reentry around scar, most commonly post-MI. Also arises in nonischemic cardiomyopathies, sarcoidosis, and arrhythmogenic right ventricular cardiomyopathy (ARVC).
Polymorphic VT: Beat-to-beat QRS morphology change. Causes: acute ischemia, electrolyte disturbance.
Torsades de pointes: Polymorphic VT in the setting of a prolonged QT. Causes include QT-prolonging drugs (methadone, ondansetron, many antibiotics, antipsychotics, class IA and III antiarrhythmics), hypokalemia, hypomagnesemia, and congenital long QT syndrome. Treat with IV magnesium, correction of electrolytes, and overdrive pacing.
Management: Pulseless VT and VF are treated with immediate defibrillation per ACLS. Stable sustained VT: IV amiodarone, lidocaine, or procainamide; unstable: synchronized cardioversion. Long-term management of recurrent sustained VT involves ICD implantation, antiarrhythmic drug therapy (amiodarone, sotalol, mexiletine), and catheter ablation of the VT circuit. ICDs are indicated for secondary prevention after any sustained VT/VF not due to reversible causes, and for primary prevention in HFrEF with EF ≤ 35% on GDMT.
10 Bradyarrhythmias & Conduction Disease Arrhythmia
Bradyarrhythmias involve either failure of impulse generation (sinus node dysfunction) or failure of impulse conduction (AV block). Common causes: aging, ischemia (especially inferior MI affecting the AV node), medications (BB, CCB, digoxin, amiodarone), Lyme disease, sarcoidosis, and post-cardiac-surgery.
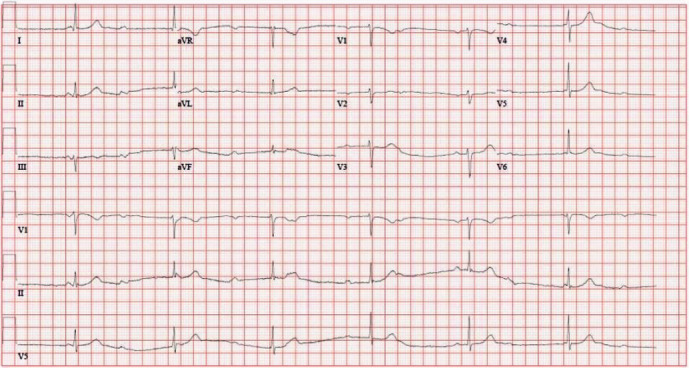
AV Block Grades
| Degree | ECG Finding | Significance |
|---|---|---|
| First-degree | PR > 200 ms, every P conducts | Usually benign |
| Second-degree Mobitz I (Wenckebach) | Progressive PR lengthening until a dropped QRS | Usually AV nodal; often benign |
| Second-degree Mobitz II | Fixed PR with intermittently dropped QRS | Infranodal; high risk of progression — pacemaker |
| High-grade AV block | Two or more consecutive non-conducted P waves | Pacemaker |
| Third-degree (complete) | Complete AV dissociation; escape rhythm | Pacemaker (unless reversible) |
Sinus node dysfunction (SND/SSS): Inappropriate sinus bradycardia, sinus pauses > 3 sec, sinoatrial exit block, or tachy-brady syndrome (often alternating AFib with symptomatic bradycardia). Symptomatic SND is the most common indication for pacemaker implantation.
Bundle branch blocks: RBBB (rsR' in V1, wide S in I/V6) is often benign in structurally normal hearts; new LBBB (broad monophasic R in I/V6, QS in V1) is always pathologic and in the right clinical context is treated as a STEMI equivalent (Sgarbossa criteria).
11 Valvular Heart Disease Valvular
The 2020 ACC/AHA valvular heart disease guideline governs staging (A at-risk, B progressive, C asymptomatic severe, D symptomatic severe), imaging surveillance, and intervention thresholds. Every valve lesion should be documented by echo parameters (gradient, area, regurgitation grade), symptom status, LV size/function, and PA pressure.
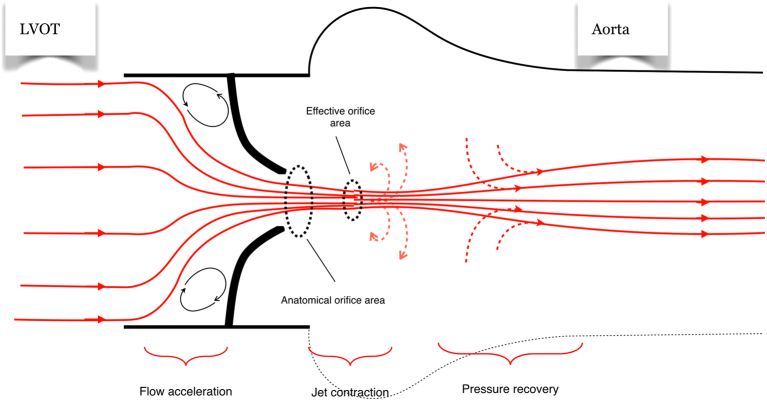
Aortic Stenosis (AS)
Progressive calcific narrowing of the aortic valve, typically in adults > 65 (calcific degeneration) or younger patients with bicuspid aortic valves. Classic triad when severe: angina, syncope, heart failure. Exam: crescendo-decrescendo systolic ejection murmur at the right upper sternal border radiating to carotids, delayed and diminished carotid upstroke (pulsus parvus et tardus), soft or absent S2.
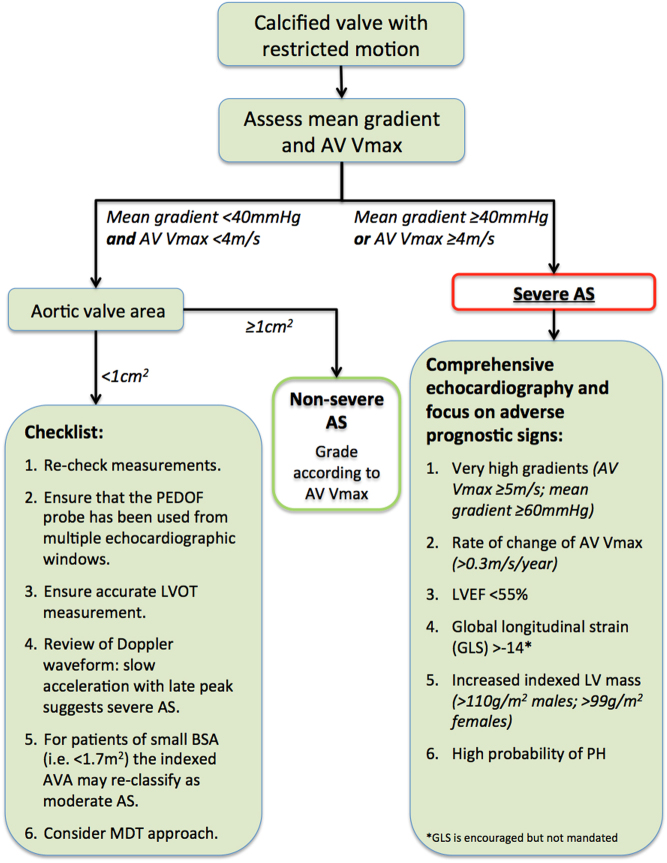
| AS Severity | Peak Velocity | Mean Gradient | AVA |
|---|---|---|---|
| Mild | 2.0–2.9 m/s | < 20 mmHg | > 1.5 cm² |
| Moderate | 3.0–3.9 m/s | 20–39 mmHg | 1.0–1.5 cm² |
| Severe | ≥ 4.0 m/s | ≥ 40 mmHg | ≤ 1.0 cm² |
| Very severe | ≥ 5.0 m/s | ≥ 60 mmHg | — |
Intervention: Symptomatic severe AS is a Class I indication for AVR — surgical (SAVR) or transcatheter (TAVR). TAVR is now approved across the full surgical risk spectrum based on PARTNER and Evolut trials; SAVR remains preferred in younger patients, bicuspid valves, or when concomitant cardiac surgery is needed.
Aortic Regurgitation (AR)
Causes: bicuspid valve, rheumatic disease, endocarditis, aortic root dilation (HTN, Marfan, dissection). Presents with wide pulse pressure, "water-hammer" pulse, and an early diastolic decrescendo murmur at the left sternal border. Severe chronic AR produces LV dilation and eccentric hypertrophy. Intervention thresholds: symptomatic severe AR, or asymptomatic severe AR with LVEF ≤ 55% or LVESD > 50 mm.
Mitral Regurgitation (MR)
Divided into primary (degenerative — mitral valve prolapse, flail leaflet, endocarditis) and secondary/functional (from LV dilation and leaflet tethering in HFrEF, or LA dilation in AFib). Murmur: holosystolic at the apex, radiating to the axilla; may have S3 in severe cases. Intervention for severe primary MR when symptomatic or with LV dysfunction (EF < 60% or LVESD ≥ 40 mm). For secondary MR in HFrEF despite GDMT, the COAPT trial supports transcatheter edge-to-edge repair (MitraClip).
Mitral Stenosis (MS)
Almost always rheumatic in origin globally. Pathognomonic exam: opening snap followed by a low-pitched diastolic rumble at the apex, best heard in left lateral decubitus. Complications: atrial fibrillation, systemic embolization, pulmonary hypertension. Treatment: percutaneous mitral balloon valvuloplasty (PMBV) for favorable valve anatomy (Wilkins score ≤ 8) or surgical valve replacement. AFib in moderate-to-severe MS is a clear indication for warfarin (DOACs are contraindicated).
Tricuspid and Pulmonic Disease
Tricuspid regurgitation is most often secondary to RV dilation (from pulmonary HTN or left-sided disease) or lead-related (pacemaker/ICD leads). Primary TR causes include endocarditis (IVDU), carcinoid, Ebstein anomaly. Pulmonic stenosis is usually congenital; pulmonic regurgitation is often post-surgical (tetralogy repair).
12 Pericardial Disease & Tamponade Pericardial
Acute Pericarditis
Inflammation of the pericardium, most commonly idiopathic/viral. Classic clinical features: pleuritic chest pain that is worse lying flat and improved sitting forward, pericardial friction rub on exam, widespread concave ST elevation with PR depression on ECG, and pericardial effusion on echo. Diagnosis requires 2 of 4 (pain, rub, ECG changes, effusion). Treatment: NSAIDs (ibuprofen or ASA) plus colchicine × 3 months (reduces recurrence per the ICAP/CORP trials). Corticosteroids are reserved for refractory, autoimmune, or uremic cases — they increase recurrence.
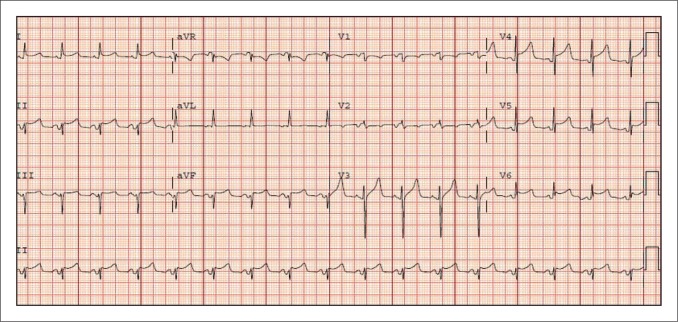
Pericardial Effusion and Tamponade
Fluid accumulation in the pericardial space compresses cardiac chambers. The rate of accumulation matters more than the volume: a slowly accumulating 1-liter effusion may be asymptomatic, while a rapidly accumulating 200 mL can tamponade.
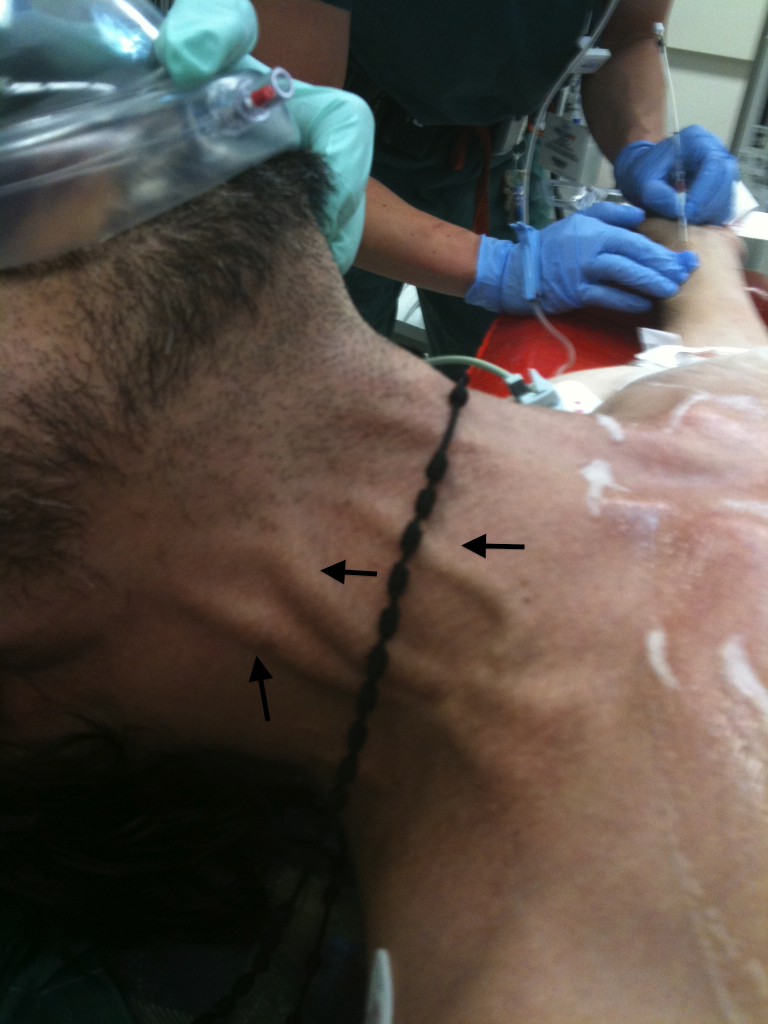
Beck's triad: hypotension, muffled heart sounds, JVD.
Pulsus paradoxus: > 10 mmHg drop in SBP with inspiration.
ECG: low voltage, electrical alternans (beat-to-beat QRS amplitude variation).
Echo: RA and RV diastolic collapse, plethoric IVC, respiratory variation of mitral/tricuspid inflow. Treatment is emergent pericardiocentesis or pericardial window.
Constrictive Pericarditis
Scarred, rigid pericardium restricts diastolic filling. Causes: prior cardiac surgery, radiation, TB (globally), idiopathic. Presents as right-sided heart failure with preserved EF: elevated JVP, Kussmaul's sign, ascites, edema, pericardial knock on auscultation. Distinguished from restrictive cardiomyopathy by echo, cardiac MRI, and invasive hemodynamics. Definitive treatment is pericardiectomy.
13 Cardiomyopathies Myocardial
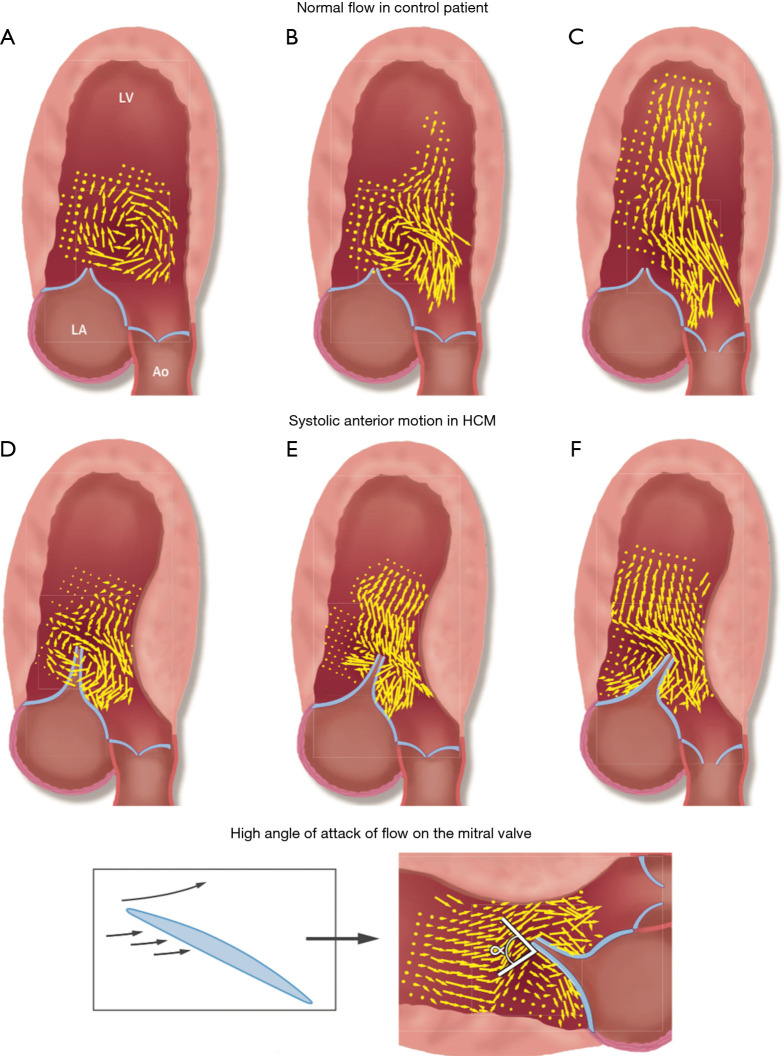
Hypertrophic Cardiomyopathy (HCM)
Autosomal dominant disorder of sarcomeric protein genes producing asymmetric LVH, often with septal predominance. A subset has left ventricular outflow tract (LVOT) obstruction from systolic anterior motion (SAM) of the anterior mitral leaflet. Clinical features: exertional dyspnea, angina, syncope, and risk of sudden cardiac death (especially in young athletes). Exam: harsh systolic murmur at LLSB that increases with Valsalva and standing (opposite of AS). Management: beta-blockers or non-dihydropyridine CCBs, disopyramide, and the cardiac myosin inhibitor mavacamten. Septal reduction (myectomy or alcohol septal ablation) for refractory symptomatic LVOTO. ICD for high-risk features (prior cardiac arrest, family history of SCD, massive LVH ≥ 30 mm, unexplained syncope, non-sustained VT, abnormal BP response to exercise).
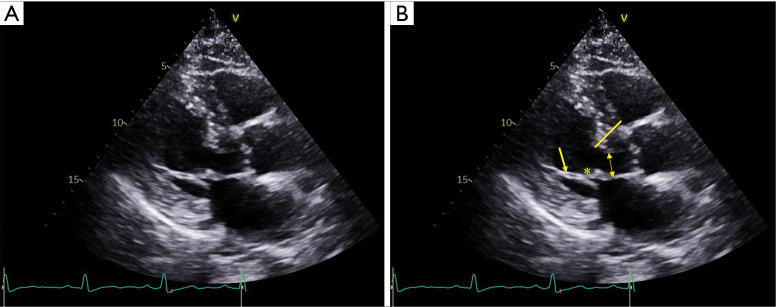
Dilated Cardiomyopathy (DCM)
LV dilation and systolic dysfunction in the absence of ischemic, valvular, or hypertensive cause sufficient to explain the findings. Causes: genetic (30–40%), viral myocarditis, alcohol, cocaine/methamphetamine, peripartum, thyroid disease, tachycardia-mediated, chemotherapy (anthracyclines, trastuzumab), and idiopathic. Treatment is HFrEF GDMT plus treatment of the underlying cause.
Restrictive Cardiomyopathy
Stiff non-compliant ventricles with impaired diastolic filling but preserved systolic function. Causes: cardiac amyloidosis (transthyretin or AL type), sarcoidosis, hemochromatosis, endomyocardial fibrosis, radiation. Cardiac amyloidosis has become increasingly recognized and treatable with tafamidis for TTR-CM.
Stress (Takotsubo) Cardiomyopathy
Transient LV apical ballooning in the setting of emotional or physical stress, classically in post-menopausal women. Presents like ACS (chest pain, ECG changes, modest troponin elevation) but angiogram shows no obstructive CAD. Echo shows apical akinesis with basal hyperkinesis. Usually recovers within weeks with supportive care.
14 Pulmonary Hypertension Vascular
PH is defined by mean PA pressure > 20 mmHg at rest by right heart catheterization. The WHO classifies PH into five groups:
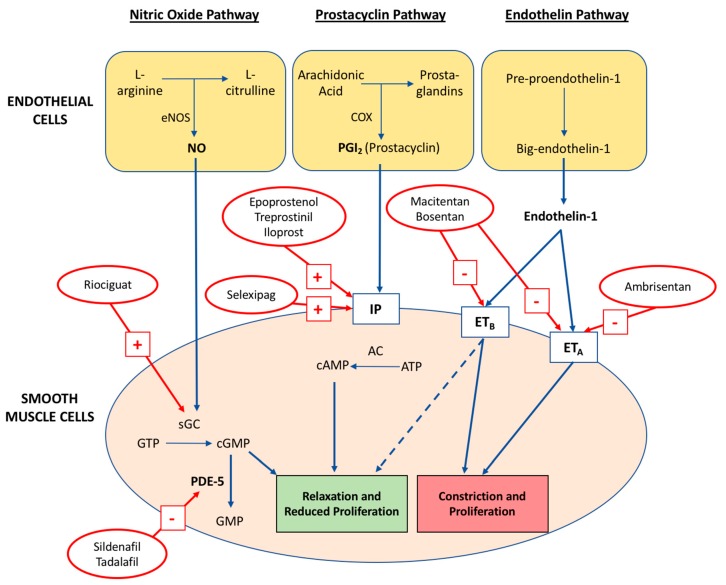
Group 1 — Pulmonary arterial hypertension (PAH): idiopathic, heritable, drug-induced (anorexigens, methamphetamine), connective tissue disease (scleroderma), HIV, portal HTN, congenital heart disease.
Group 2 — PH due to left heart disease: HFpEF, HFrEF, valvular. Most common cause overall.
Group 3 — PH due to lung disease/hypoxia: COPD, ILD, OSA.
Group 4 — Chronic thromboembolic (CTEPH): treatable with pulmonary endarterectomy or balloon pulmonary angioplasty.
Group 5 — Multifactorial/unclear: sarcoidosis, sickle cell, metabolic disorders.
PAH (Group 1) is treated at specialty centers with endothelin receptor antagonists (bosentan, ambrisentan, macitentan), PDE5 inhibitors (sildenafil, tadalafil), soluble guanylate cyclase stimulators (riociguat), and prostacyclins (epoprostenol, treprostinil). Group 2 PH is treated by optimizing the left-sided heart disease — PAH-targeted therapy is harmful.
15 Infective Endocarditis Infectious
Infection of the endocardial surface, most often the heart valves. Risk factors: prosthetic valves, prior endocarditis, IV drug use, structural heart disease, indwelling lines, immunosuppression. Organisms: Staphylococcus aureus (most common overall, especially acute and IVDU), viridans streptococci (subacute, dental), enterococci (GU/GI source), HACEK organisms, coagulase-negative staph (prosthetic valves).
The modified Duke criteria are used for diagnosis (definite endocarditis = 2 major, or 1 major + 3 minor, or 5 minor).
Major: (1) Positive blood cultures with typical organism (2 separate draws), or persistently positive; (2) Evidence of endocardial involvement (vegetation, abscess, new valvular regurgitation, or dehiscence of prosthetic valve on echo).
Minor: predisposing condition/IVDU, fever > 38°C, vascular phenomena (septic emboli, Janeway lesions, mycotic aneurysm), immunologic phenomena (Osler nodes, Roth spots, glomerulonephritis, RF), microbiologic evidence not meeting major criteria.
Workup: blood cultures × 3 sets from separate sites before antibiotics, TTE followed by TEE (higher sensitivity, mandatory for prosthetic valves or high suspicion), CBC, inflammatory markers. Empiric antibiotics depend on acuity and valve type; definitive therapy is typically 4–6 weeks of IV antibiotics. Surgery indications include heart failure from valve dysfunction, uncontrolled infection (abscess, persistent bacteremia), large vegetations (> 10 mm) with embolic events, and prosthetic valve involvement.
16 Syncope & Palpitations Symptom
Syncope is transient loss of consciousness from global cerebral hypoperfusion, followed by spontaneous complete recovery. The first task in evaluation is to separate benign causes (reflex/vasovagal) from dangerous ones (cardiac).
Reflex (neurally mediated): vasovagal (most common; prodrome of nausea, diaphoresis, tunnel vision), situational (cough, micturition), carotid sinus hypersensitivity.
Orthostatic: drug-induced, volume depletion, autonomic failure. SBP drop ≥ 20 or DBP drop ≥ 10 within 3 minutes of standing.
Cardiac (dangerous): arrhythmias (VT, AV block, SSS), structural (severe AS, HCM, MI, cardiac tamponade, PE, aortic dissection, myxoma). Syncope during exertion or supine, syncope without prodrome, and syncope with palpitations all suggest a cardiac cause.
Workup: ECG in every syncope patient, orthostatic vitals, BMP, troponin if suspicion of ACS. Echo if structural disease suspected. Ambulatory monitoring (Holter, event monitor, mobile cardiac telemetry, implantable loop recorder) for suspected arrhythmic syncope. Tilt-table testing for recurrent reflex syncope. Electrophysiology study for high-risk patients with structural disease.
Palpitations (awareness of one's own heartbeat) should be characterized by: onset/offset (sudden vs gradual), regular vs irregular, rate, duration, triggers, associated symptoms. Regular sustained palpitations suggest SVT or VT; irregularly irregular suggests AFib. "Skipped beats" or "flip-flops" are classic for PVCs or PACs.
17 Cardiology Procedures — A to Z Procedures
Diagnostic & Interventional Cath Lab
| Procedure | Description / Indication |
|---|---|
| Left heart cath / coronary angiography | Radial or femoral arterial access, selective coronary injection. For ACS, unexplained HF, pre-op evaluation, abnormal stress testing. |
| Right heart cath | Venous access (usually IJ or femoral) with Swan-Ganz catheter. Measures RA, RV, PA, PCWP, CO, SVR. Gold standard for PH diagnosis and HF hemodynamic profiling. |
| PCI (percutaneous coronary intervention) | Balloon angioplasty + drug-eluting stent (DES). Standard of care for STEMI, high-risk NSTEMI, and refractory stable CAD. |
| Fractional flow reserve (FFR) / iFR | Physiologic assessment of lesion significance. FFR ≤ 0.80 or iFR ≤ 0.89 indicates hemodynamically significant stenosis. |
| Intravascular ultrasound (IVUS) / OCT | Intracoronary imaging for lesion characterization and stent optimization. |
| Rotational atherectomy | Burr-based debulking of heavily calcified lesions before stenting. |
| Intra-aortic balloon pump (IABP) | Mechanical support in cardiogenic shock, refractory ischemia, high-risk PCI. |
| Impella / VA-ECMO | Temporary mechanical circulatory support in cardiogenic shock or high-risk PCI. |
Structural Heart
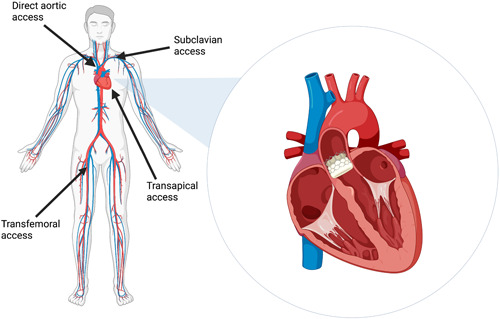
| Procedure | Description |
|---|---|
| TAVR | Transcatheter aortic valve replacement. Femoral (most common), transapical, subclavian, or transcaval access. Balloon-expandable (Edwards SAPIEN) or self-expanding (Medtronic Evolut). |
| MitraClip / TEER | Transcatheter edge-to-edge repair of mitral regurgitation. Primary degenerative MR in high surgical risk, or secondary MR per COAPT criteria. |
| PMBV | Percutaneous mitral balloon valvuloplasty for rheumatic MS with favorable anatomy. |
| LAA occlusion (Watchman) | Left atrial appendage closure device for AFib patients with contraindications to long-term anticoagulation. |
| PFO / ASD closure | Percutaneous device closure, used for cryptogenic stroke with PFO in selected patients and hemodynamically significant ASDs. |
| Alcohol septal ablation | Targeted septal infarction for symptomatic obstructive HCM refractory to medical therapy. |
Electrophysiology
| Procedure | Description |
|---|---|
| EP study | Intracardiac catheters characterize conduction, induce arrhythmias, and localize circuits. |
| Catheter ablation | RF or cryoablation of arrhythmia substrate. AFib → pulmonary vein isolation; flutter → CTI; AVNRT → slow pathway; VT → scar-based substrate modification. |
| Permanent pacemaker (PPM) | Single-chamber (VVI, AAI), dual-chamber (DDD), or biventricular (CRT). Indicated for symptomatic bradycardia, high-grade AV block, and sinus node dysfunction. |
| ICD | Primary prevention: HFrEF with EF ≤ 35% on GDMT, HCM high-risk. Secondary prevention: prior sustained VT/VF. |
| CRT / CRT-D | Biventricular pacing for HFrEF with EF ≤ 35%, LBBB, QRS ≥ 150 ms. |
| Leadless pacemaker | Self-contained device (e.g., Micra) implanted directly in the RV. For single-chamber indications. |
| Subcutaneous ICD (S-ICD) | No transvenous leads; for primary prevention without need for pacing or anti-tachy pacing. |
| Cardioversion (DCCV) | Synchronized shock for AFib, flutter, SVT, stable VT. |
Surgical (Cardiothoracic Referrals)
| Procedure | Indication |
|---|---|
| CABG | Left main disease, multivessel CAD with reduced EF or diabetes, failed PCI. |
| Surgical AVR / MVR | Symptomatic severe valve disease unsuitable for percutaneous approach. |
| Mitral valve repair | Primary degenerative MR (preferred over replacement when feasible). |
| Septal myectomy | Obstructive HCM with refractory symptoms. |
| LVAD | Advanced HF as destination therapy or bridge to transplant. |
| Orthotopic heart transplant | End-stage HF refractory to all other therapies. |
18 Post-Procedure Management & Complications Procedures
Post-PCI
Access site management is paramount. Radial access has largely replaced femoral because of lower bleeding risk. Monitor for: access site hematoma, retroperitoneal bleed (femoral), pseudoaneurysm, AV fistula, radial artery occlusion. Watch for contrast-induced nephropathy (rise in creatinine at 48–72 h). DAPT duration depends on stent type and clinical context: 6 months after DES for stable CAD, 12 months after DES for ACS, longer in selected high ischemic risk. Continue aspirin indefinitely.
Post-TAVR
Monitor for: conduction disturbances (new LBBB, complete heart block — ~10–15% require new PPM), paravalvular leak (assess on post-procedure TTE), vascular access complications, stroke. Antithrombotic: single antiplatelet therapy (aspirin) is now standard, not DAPT, based on the POPular TAVI trial.
Post-Device (PPM/ICD/CRT)
Early complications: pneumothorax, lead dislodgement, pocket hematoma, infection. Patients are instructed to restrict ipsilateral arm motion (no raising above shoulder) for 4–6 weeks. Device interrogation is done before discharge and at 2–4 weeks, then remotely every 3–12 months.
Post-Ablation
After AFib ablation: blanking period of 3 months (early recurrences do not count as failure), continue anticoagulation for at least 2–3 months regardless of CHA₂DS₂-VASc. Rare but serious complications: cardiac tamponade, atrioesophageal fistula (presents weeks later with fever, stroke, GI bleeding — ~100% mortality if missed), pulmonary vein stenosis, phrenic nerve injury.
Watch for mechanical complications 2–7 days after MI: free wall rupture (sudden tamponade, PEA arrest), VSD (new harsh holosystolic murmur, step-up in oxygen saturation from RA to RV), papillary muscle rupture with acute severe MR (flash pulmonary edema, often inferior MI affecting posteromedial papillary muscle). All are surgical emergencies.
19 Cardiac Imaging & Diagnostics Diagnostics
12-Lead ECG
The first test in almost every cardiology encounter. Read systematically: rate, rhythm, axis, intervals (PR, QRS, QT/QTc), chamber hypertrophy, ST/T changes, pathological Q waves.
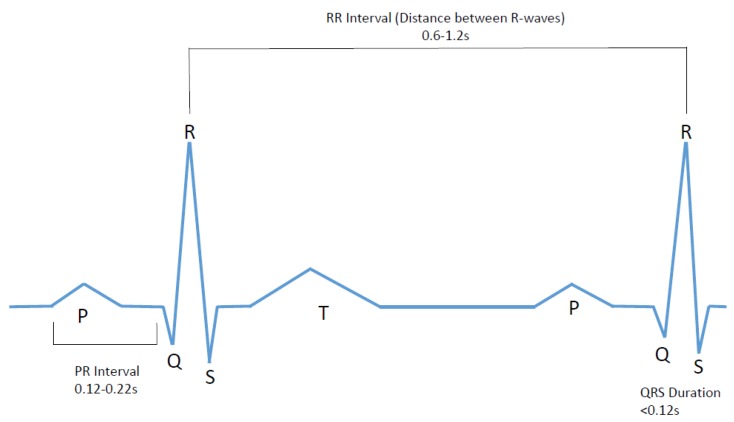
Rate: 60–100 (sinus). PR: 120–200 ms. QRS: < 120 ms. QTc: < 440 ms (men) / < 460 ms (women); prolonged if > 500 with arrhythmia risk. Axis: -30° to +90°.
Echocardiography
TTE: Non-invasive; assesses chamber sizes, LVEF, wall motion (segmental abnormalities suggest CAD), valves (gradient, area, regurgitation), pericardium, RV function, estimated PA systolic pressure from TR jet. TEE: Superior for mitral valve, LAA thrombus (before cardioversion), prosthetic valves, endocarditis, aortic dissection, and intracardiac shunts. Stress echo: assesses ischemia and valve behavior under stress (exercise or dobutamine).
Stress Testing
Exercise ECG (treadmill, Bruce protocol) for low-intermediate risk patients with an interpretable baseline ECG. Stress echo or nuclear (SPECT, PET) MPI if baseline ECG abnormal or exercise not feasible. Pharmacologic stress uses dobutamine (contractility) or vasodilators (regadenoson, adenosine, dipyridamole) for nuclear imaging.
Cardiac CT & MRI
CCTA: Excellent negative predictive value for obstructive CAD; increasingly first-line for intermediate-risk chest pain. Also used for TAVR planning. Coronary artery calcium score (CAC): non-contrast gated CT quantifying calcified plaque; risk-stratifies asymptomatic intermediate-risk patients. Cardiac MRI: gold standard for LV/RV volumes and function, tissue characterization (late gadolinium enhancement patterns diagnose infiltrative disease, myocarditis, ischemic vs non-ischemic scar).
Key Cath Lab Numbers
| Parameter | Normal |
|---|---|
| RA pressure | 2–6 mmHg |
| RV pressure | 25/4 mmHg |
| PA pressure | 25/10 mmHg (mean ≤ 20) |
| PCWP | 6–12 mmHg |
| Cardiac index | 2.5–4.0 L/min/m² |
| SVR | 800–1200 dyn·s/cm⁵ |
20 Cardiology Medications You Must Know Pharmacology
Antiplatelets
| Generic | Brand | Mechanism / Use |
|---|---|---|
| Aspirin | — | COX-1 inhibitor; secondary prevention, ACS, post-PCI |
| Clopidogrel | Plavix | P2Y₁₂ inhibitor; post-PCI, ASA intolerance |
| Ticagrelor | Brilinta | Reversible P2Y₁₂; ACS; dyspnea side effect |
| Prasugrel | Effient | P2Y₁₂; PCI in ACS; avoid in prior stroke/TIA, age ≥ 75, < 60 kg |
Anticoagulants
| Generic | Brand | Use / Notes |
|---|---|---|
| Warfarin | Coumadin | Vitamin K antagonist; mechanical valves, moderate-severe MS. INR goal 2–3 (mechanical AVR) or 2.5–3.5 (mechanical MVR) |
| Apixaban | Eliquis | Factor Xa inhibitor; non-valvular AFib, VTE. 5 mg BID (2.5 BID if 2 of: age ≥ 80, wt ≤ 60 kg, Cr ≥ 1.5) |
| Rivaroxaban | Xarelto | Factor Xa inhibitor; AFib, VTE, PAD/CAD (2.5 BID add-on per COMPASS) |
| Dabigatran | Pradaxa | Direct thrombin inhibitor; reversal with idarucizumab |
| Edoxaban | Savaysa | Factor Xa inhibitor; AFib, VTE |
| Enoxaparin | Lovenox | LMWH; ACS, VTE, bridging |
| UFH | — | IV; titrated to aPTT or anti-Xa. ACS, peri-procedure |
The COMPASS trial established rivaroxaban 2.5 mg BID + aspirin as a strategy in stable CAD/PAD with benefit in reducing MACE.
Beta-Blockers
| Generic | Brand | Selectivity / Use |
|---|---|---|
| Metoprolol tartrate | Lopressor | β1-selective; rate control, post-MI |
| Metoprolol succinate | Toprol XL | β1-selective; HFrEF GDMT |
| Carvedilol | Coreg | Non-selective β + α1; HFrEF GDMT |
| Bisoprolol | Zebeta | β1-selective; HFrEF GDMT |
| Atenolol | Tenormin | β1-selective; HTN, angina |
| Propranolol | Inderal | Non-selective; HCM, thyroid storm, migraine |
| Esmolol | Brevibloc | Ultra-short-acting IV; acute rate control |
| Labetalol | Normodyne | Combined α/β; HTN emergency, pregnancy |
ACEi / ARB / ARNI / MRA
| Generic | Brand | Class / Use |
|---|---|---|
| Lisinopril | Zestril | ACEi; HTN, HFrEF, post-MI |
| Enalapril | Vasotec | ACEi |
| Ramipril | Altace | ACEi; strong post-MI evidence (HOPE) |
| Losartan | Cozaar | ARB |
| Valsartan | Diovan | ARB |
| Sacubitril/valsartan | Entresto | ARNI; HFrEF first-line (per PARADIGM-HF) |
| Spironolactone | Aldactone | MRA; HFrEF, resistant HTN; gynecomastia |
| Eplerenone | Inspra | MRA; post-MI HF, fewer endocrine effects |
SGLT2 Inhibitors & Other HF Therapies
| Generic | Brand | Notes |
|---|---|---|
| Dapagliflozin | Farxiga | HFrEF, HFpEF, CKD — DAPA-HF, DELIVER |
| Empagliflozin | Jardiance | HFrEF, HFpEF, CKD — EMPEROR trials |
| Ivabradine | Corlanor | If channel inhibitor; HFrEF with sinus HR > 70 on max BB |
| Digoxin | Lanoxin | HFrEF symptom control; AFib rate control adjunct |
| Hydralazine + ISDN | BiDil | HFrEF, especially Black patients |
| Mavacamten | Camzyos | Cardiac myosin inhibitor; obstructive HCM |
| Tafamidis | Vyndaqel | TTR stabilizer; ATTR cardiac amyloidosis |
Antiarrhythmics (Vaughan Williams)
| Class | Mechanism | Examples |
|---|---|---|
| IA | Na channel block, intermediate | Quinidine, procainamide, disopyramide |
| IB | Na channel block, fast | Lidocaine, mexiletine |
| IC | Na channel block, slow | Flecainide, propafenone |
| II | Beta-blockers | Metoprolol, carvedilol, esmolol |
| III | K channel block | Amiodarone, sotalol, dofetilide, ibutilide, dronedarone |
| IV | Non-DHP CCB | Diltiazem, verapamil |
| Other | — | Adenosine, digoxin, magnesium |
Lipid-Lowering Therapy
| Generic | Brand | Intensity / Use |
|---|---|---|
| Atorvastatin 40–80 | Lipitor | High-intensity statin |
| Rosuvastatin 20–40 | Crestor | High-intensity statin |
| Pravastatin / Simvastatin / Lovastatin | — | Moderate-intensity |
| Ezetimibe | Zetia | NPC1L1 inhibitor; add-on for LDL |
| Alirocumab / Evolocumab | Praluent / Repatha | PCSK9 monoclonal antibodies; very high-risk ASCVD, FH |
| Inclisiran | Leqvio | PCSK9 siRNA; twice-yearly injection |
| Bempedoic acid | Nexletol | ATP-citrate lyase inhibitor; statin-intolerant |
| Icosapent ethyl | Vascepa | Pure EPA; TG lowering + CV benefit (REDUCE-IT) |
Diuretics
| Class | Examples | Notes |
|---|---|---|
| Loop | Furosemide (Lasix), torsemide (Demadex), bumetanide (Bumex) | HF volume overload; torsemide has better oral bioavailability |
| Thiazide | HCTZ, chlorthalidone, metolazone | HTN; metolazone potentiates loops |
| K-sparing / MRA | Spironolactone, eplerenone, amiloride, triamterene | HFrEF, resistant HTN |
| Carbonic anhydrase | Acetazolamide | Metabolic alkalosis from loops; ADVOR trial adjunct in acute HF |
Always chart medications by both generic and brand name on the first mention, along with the dose, frequency, and route. Cardiology patients often know their meds only by brand names (Eliquis, Entresto, Jardiance) — your note should bridge both.
21 Classification Systems Reference
Every cardiology note leans on a classification system somewhere. These are the ones you will chart by name — memorize the full enumeration, not just the acronym.
NYHA Functional Class
| Class | Description |
|---|---|
| I | No limitation; ordinary activity does not cause symptoms |
| II | Slight limitation; ordinary activity causes symptoms |
| III | Marked limitation; less than ordinary activity causes symptoms |
| IV | Symptoms at rest; any activity causes discomfort |
ACC/AHA HF Stages
| Stage | Definition |
|---|---|
| A | At risk (HTN, DM, CAD, cardiotoxic exposure, family history); no structural disease or symptoms |
| B | Structural heart disease without current or prior HF symptoms |
| C | Structural disease + current or prior HF symptoms |
| D | Advanced HF requiring specialized interventions |
CCS Angina Class
| Class | Description |
|---|---|
| I | Angina only with strenuous, prolonged, or rapid exertion |
| II | Slight limitation of ordinary activity |
| III | Marked limitation — angina with 1–2 blocks or 1 flight of stairs |
| IV | Inability to carry out any physical activity; may occur at rest |
Killip Classification (post-MI)
| Class | Findings | Mortality |
|---|---|---|
| I | No HF | ~6% |
| II | S3, crackles, JVD | ~17% |
| III | Pulmonary edema | ~38% |
| IV | Cardiogenic shock | ~81% |
CHA₂DS₂-VASc
CHF 1, HTN 1, Age ≥ 75 (2), DM 1, Stroke/TIA/TE (2), Vascular disease 1, Age 65–74 (1), Sex category female 1. Max 9. Anticoagulation recommended at ≥ 2 (men) or ≥ 3 (women).
HAS-BLED
HTN, Abnormal renal/liver, Stroke, Bleeding, Labile INR, Elderly, Drugs/alcohol. Max 9. Score ≥ 3 = high bleeding risk.
TIMI Risk Score (NSTEMI/UA)
Age ≥ 65, ≥ 3 CAD risk factors, known CAD (stenosis ≥ 50%), ASA use in past 7 days, severe angina (≥ 2 episodes in 24 h), ST deviation ≥ 0.5 mm, positive biomarker. 1 point each, 0–7. Higher score = higher 14-day risk of death/MI/urgent revascularization.
GRACE Score
Uses age, HR, SBP, Cr, Killip class, cardiac arrest at admission, ST deviation, elevated enzymes. Scores > 140 = high-risk NSTEMI benefiting from early invasive strategy; < 109 = low risk.
Aortic Stenosis Stages (AHA/ACC)
Stage A: at risk (bicuspid, sclerosis). Stage B: mild-moderate. Stage C1: asymptomatic severe with preserved LVEF. Stage C2: asymptomatic severe with LVEF < 50%. Stage D1: symptomatic high-gradient severe. D2: symptomatic low-flow/low-gradient with reduced EF. D3: symptomatic low-gradient severe with preserved EF (paradoxical).
22 The Cardiovascular Physical Exam Exam
The cardiology exam is precise and stylized. Document in the same order the cardiologist performs it.
General: NAD, well-appearing, no distress
Vitals: BP 128/76 (R arm, seated) HR 72 RR 14 SpO₂ 98% RA
Neck: JVP 6 cm above sternal angle; no carotid bruits; carotid upstroke normal bilaterally
Chest wall: no deformity; no prior sternotomy scar
Heart: PMI at 5th ICS midclavicular line, non-displaced; RRR; normal S1, physiologically split S2; no S3, no S4; no murmurs, rubs, or gallops
Lungs: CTAB, no crackles or wheezes
Abdomen: soft, non-tender, no hepatomegaly, no bruits
Extremities: no clubbing, cyanosis, or edema; distal pulses 2+/2+ throughout; warm, well-perfused
Murmur Grading
| Grade | Description |
|---|---|
| I/VI | Barely audible, requires concentration |
| II/VI | Soft but easily heard |
| III/VI | Moderately loud, no thrill |
| IV/VI | Loud with palpable thrill |
| V/VI | Very loud, heard with stethoscope partly off chest |
| VI/VI | Audible with stethoscope off the chest |
Characteristic Murmurs
| Lesion | Location | Character / Radiation |
|---|---|---|
| Aortic stenosis | RUSB (2nd R ICS) | Crescendo-decrescendo systolic, radiates to carotids |
| Aortic regurgitation | LLSB, sitting forward | Early diastolic decrescendo |
| Mitral regurgitation | Apex | Holosystolic, radiates to axilla |
| Mitral stenosis | Apex, left lateral decubitus | Opening snap + diastolic rumble |
| Tricuspid regurgitation | LLSB | Holosystolic, increases with inspiration (Carvallo sign) |
| HCM | LLSB | Systolic; increases with Valsalva/standing |
| VSD | LLSB | Harsh holosystolic |
| PDA | Left infraclavicular | Continuous "machinery" murmur |
When a murmur is detected, always document: timing (systolic vs diastolic vs continuous), location of maximal intensity, radiation, grade, shape (crescendo-decrescendo, holosystolic, decrescendo), and response to maneuvers (Valsalva, handgrip, standing, squatting).
23 Abbreviations Master List Reference
Diagnoses & Conditions
Procedures & Devices
Imaging & Diagnostics
Medications & Therapies
24 Sample HPI Templates Templates
These templates show the density, terminology, and structure expected in a cardiology clinic or consult note. Use them as scaffolding for your own documentation during your first weeks.
"Mr. [Name] is a 64-year-old male with HTN, T2DM (A1C 8.1), former 25-pack-year smoker (quit 2018), and newly diagnosed non-ischemic dilated cardiomyopathy who presents for initial cardiology evaluation following a 4-day hospitalization for acute decompensated heart failure. He reports progressive DOE over 6 weeks, now with 2-pillow orthopnea and PND 2–3 times weekly. He has gained 12 lb over the past month and developed bilateral lower extremity edema to the mid-shins. He denies chest pain, palpitations, or syncope. TTE from admission: LVEF 25%, global hypokinesis, LV 6.0 cm EDD, moderate functional MR, estimated PASP 45 mmHg. Left heart cath on day 2 showed non-obstructive CAD. Discharge meds: furosemide 40 mg PO BID, carvedilol 3.125 mg BID, sacubitril/valsartan 24/26 mg BID, spironolactone 25 mg daily, empagliflozin 10 mg daily, atorvastatin 40 mg. He is NYHA class III today. Currently adhering to 2 g Na diet. No active chest pain, fevers, hemoptysis, or leg pain."
"Mrs. [Name] is a 58-year-old female with HTN, HLD, DM2, and a 20-pack-year smoking history (active) presenting to the ED with 90 minutes of substernal chest pressure that began at rest while watching television. She describes the pain as 7/10, squeezing, radiating to the left arm and jaw, associated with diaphoresis, nausea, and shortness of breath. No prior history of CAD. She took 2 sublingual nitroglycerin tablets from a friend with partial relief. On arrival, vitals 148/92, HR 94, SpO₂ 96% RA. Initial ECG: 2 mm ST depression V4–V6 and I, aVL with biphasic T waves. Initial hs-troponin 124 ng/L (URL 14). She has received ASA 325 mg chewed, ticagrelor 180 mg loading, atorvastatin 80 mg, IV heparin bolus and infusion, and metoprolol 5 mg IV. Cardiology is activating the cath lab for early invasive strategy. GRACE score estimated 152, TIMI 4."
"Mr. [Name] is a 72-year-old male with HTN, DM2, and CKD stage 3 (baseline Cr 1.5) who presents with 3 days of palpitations, fatigue, and mild dyspnea on exertion. He denies chest pain, syncope, or lightheadedness. He first noted an irregular heartbeat on his home BP cuff. Clinic ECG today demonstrates atrial fibrillation with rapid ventricular response (HR 128). No prior history of arrhythmia. TSH, BMP, and CBC pending. TTE ordered. CHA₂DS₂-VASc score 4 (HTN 1, age 65–74 = 1, DM 1, vascular disease [prior MI 2019] 1). HAS-BLED score 2. Plan to initiate apixaban 5 mg BID for stroke prevention and metoprolol tartrate 25 mg BID for rate control, with cardiology follow-up in 1 week to discuss rhythm control strategy including possible cardioversion and/or catheter ablation."
"Mr. [Name] is a 67-year-old male with HTN, HLD, DM2, and CAD s/p DES to the mid-LAD (90% stenosis) 6 weeks ago for NSTEMI, presenting for routine post-PCI follow-up. He reports resolution of his exertional chest pressure and is now walking 1–2 miles daily without limitation. He denies recurrent chest pain, dyspnea, palpitations, bleeding, or bruising. He is adherent to DAPT (aspirin 81 mg daily + ticagrelor 90 mg BID), atorvastatin 80 mg, metoprolol succinate 50 mg daily, lisinopril 10 mg daily. He has enrolled in cardiac rehabilitation and attended 8 of 36 sessions. Recent fasting lipid panel: LDL 62 (down from 138 pre-admission). He has not smoked since hospitalization. Assessment: NYHA I, CCS 0. Plan: continue current regimen, DAPT for a total of 12 months post-ACS, continue cardiac rehab, repeat lipid panel in 3 months."
"Ms. [Name] is a 78-year-old female with HTN, HLD, and severe aortic stenosis (peak velocity 4.3 m/s, mean gradient 48 mmHg, AVA 0.8 cm² on TTE 2 months ago) referred for evaluation of exertional syncope. She reports two episodes in the past 3 weeks: the first while climbing the stairs at home, the second while walking from her car to a grocery store. Both were preceded by brief lightheadedness without nausea, diaphoresis, or palpitations, lasted < 30 seconds, and had spontaneous complete recovery. She denies chest pain but admits to increasing DOE over 2 months, now NYHA III from NYHA II. No history of arrhythmia or prior MI. ECG in clinic: sinus rhythm, LVH with strain pattern, no ischemic changes. Given the triad of exertional syncope in severe AS with progressive dyspnea, this meets criteria for symptomatic severe AS (Stage D1). Plan: expedited referral to structural heart team for TAVR evaluation, TTE repeat, CTA for TAVR sizing, coronary angiography."
"Ms. [Name] is a 32-year-old otherwise healthy female presenting with a 6-month history of recurrent palpitations. Episodes begin abruptly, are described as a 'racing heart' with rapid regular rhythm, last 5–30 minutes, and often terminate with bearing down or drinking cold water. Frequency is approximately twice per month. She denies chest pain, syncope, or dyspnea during episodes. No history of thyroid disease, caffeine excess, or stimulant use. Family history is negative for sudden cardiac death or arrhythmia. ECG in clinic: sinus rhythm, PR 140, QRS 88, QTc 420; no delta wave, no pre-excitation. TSH, BMP, CBC normal. Two-week ambulatory patch monitor ordered. Clinical picture consistent with paroxysmal SVT, most likely AVNRT. Discussed vagal maneuvers, trigger avoidance, and referral to electrophysiology if monitor confirms SVT for consideration of catheter ablation."
The best cardiology scribes think in numbers. When the attending says "EF is 30, BP 90/60, lactate 3.2, and she's on a nor-epi drip at 8," a great scribe already knows this is cardiogenic shock and anticipates a right heart cath and possibly temporary mechanical support. When a referral note says "new murmur, peak velocity 4.2, mean gradient 46," you hear "severe AS" before finishing the sentence and start framing a TAVR workup.
Listen for the GDMT pillars in every HF note. Listen for CHA₂DS₂-VASc in every AFib note. Listen for Killip class in every STEMI note. Document the EF and its date in every encounter that touches heart failure. Pre-populate troponin trends, BNP, and the most recent echo before the patient is seen. Learn each attending's phrasing preferences — some say "RRR, no murmurs," others dictate every heart sound individually. Anticipate orders based on the diagnosis.
Cardiology moves fast and its documentation is unforgiving. But once the patterns click, you will find yourself writing notes that genuinely help the team take better care of patients. Welcome to cardiology.
25 References & Sources References
Clinical Practice Guidelines
Landmark Clinical Trials
PARADIGM-HF: McMurray JJV, et al. Angiotensin-Neprilysin Inhibition vs Enalapril in HF. NEJM. 2014.
DAPA-HF: McMurray JJV, et al. Dapagliflozin in HFrEF. NEJM. 2019.
COMPASS: Eikelboom JW, et al. Rivaroxaban with or without Aspirin in Stable CV Disease. NEJM. 2017.
COAPT: Stone GW, et al. Transcatheter Mitral-Valve Repair in Patients with HF. NEJM. 2018.
EAST-AFNET 4: Kirchhof P, et al. Early Rhythm-Control Therapy in AFib. NEJM. 2020.
Diagram & Figure Sources
Figure 1: Diagram of the human heart. Wapcaplet / Yaddah, via Wikimedia Commons. Public domain.
Figure 2: Coronary Vessels Anterior View. OpenStax College, via Wikimedia Commons. CC BY 3.0.
Figure 3: Conduction System of the Heart. OpenStax College, via Wikimedia Commons. CC BY 3.0.
Figure 4: Cardiac Cycle vs Electrocardiogram. OpenStax College, via Wikimedia Commons. CC BY 3.0.
Figure 5: Heart Attack (Myocardial Infarction). Blausen Medical, via Wikimedia Commons. CC BY 3.0.
Figure 6: Left Ventricular Hypertrophy. Patho, via Wikimedia Commons. CC BY-SA 3.0.
Figure 7: Atrial Fibrillation ECG. J. Heuser, via Wikimedia Commons. CC BY-SA 3.0.
Figure 8: Normal Sinus Rhythm Labeled. Agateller (Anthony Atkielski), via Wikimedia Commons. Public domain.