Cardiothoracic Surgery
Every diagnosis, condition, procedure, medication, abbreviation, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Cardiothoracic Anatomy Essentials
Cardiothoracic surgery (CT surgery) covers the heart, great vessels, lungs, mediastinum, esophagus, chest wall, diaphragm, and pleural space. In most US centers, the field splits into adult cardiac (coronary, valvular, aortic root and arch, arrhythmia surgery, mechanical circulatory support, transplant) and general thoracic (lung, esophagus, mediastinum, chest wall, trachea). Many surgeons still cover both. Congenital cardiac surgery is a separate subspecialty but adult congenital patients are commonly seen on adult services.
Cardiac Anatomy You Must Know
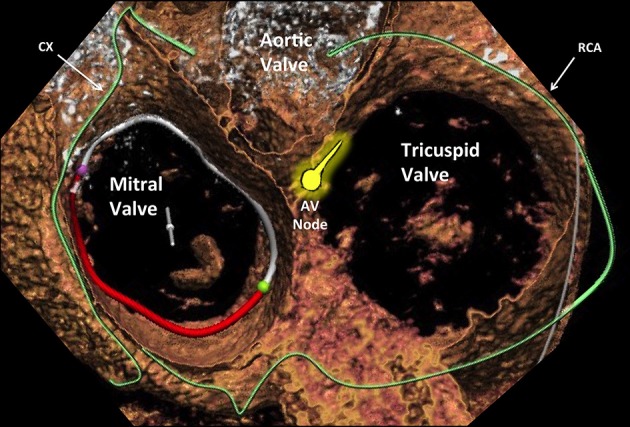
The heart sits in the middle mediastinum within the pericardial sac. Four chambers: right atrium (RA), right ventricle (RV), left atrium (LA), and left ventricle (LV). Deoxygenated blood returns to the RA via the superior and inferior vena cavae (SVC/IVC), crosses the tricuspid valve into the RV, is ejected through the pulmonic valve into the main pulmonary artery, oxygenates in the lungs, returns via four pulmonary veins to the LA, crosses the mitral valve into the LV, and is ejected across the aortic valve into the ascending aorta.
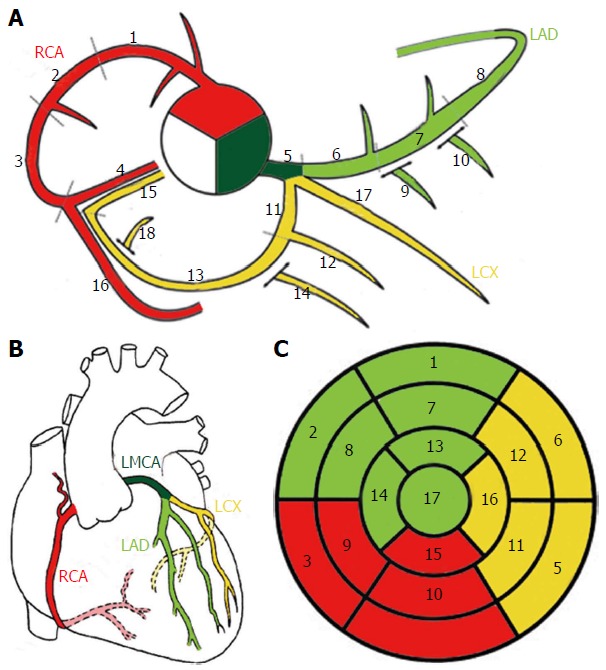
The coronary arteries arise from the sinuses of Valsalva just above the aortic valve leaflets. The left main (LM) divides into the left anterior descending (LAD) — supplying the anterior LV and anterior two-thirds of the interventricular septum via diagonal branches — and the left circumflex (LCx) supplying the lateral LV via obtuse marginal (OM) branches. The right coronary artery (RCA) supplies the RV, inferior LV, and in most patients (right-dominant circulation, ~85%) the posterior descending artery (PDA) and posterolateral branches. A left-dominant circulation means the PDA comes off the LCx. The left internal mammary artery (LIMA) graft to the LAD is the single most durable bypass conduit in cardiac surgery.
Thoracic Anatomy You Must Know
The thoracic cavity is divided into the right and left pleural spaces separated by the mediastinum. The right lung has three lobes (upper, middle, lower) separated by the major (oblique) and minor (horizontal) fissures. The left lung has two lobes (upper, lower) with the lingula functioning as the middle lobe equivalent. Each lung has bronchopulmonary segments — the right has 10 and the left has 8–10 — which are the anatomic units of segmentectomy. The trachea bifurcates at the carina (approximately T4–T5) into the right and left mainstem bronchi; the right mainstem is shorter, wider, and more vertical, which is why aspirated foreign bodies and malpositioned ET tubes favor the right.
The mediastinum is divided into compartments: anterior (thymus, lymph nodes, internal mammary vessels — the "4 T's" for masses: thymoma, teratoma, thyroid, "terrible" lymphoma), middle (heart, pericardium, great vessels, trachea, main bronchi, lymph nodes), and posterior (esophagus, descending aorta, azygos system, thoracic duct, sympathetic chain, neurogenic structures). The esophagus runs from the cricopharyngeus (C6) through the posterior mediastinum, crosses the diaphragm at T10, and joins the stomach at the gastroesophageal junction (GEJ).
Know the coronary targets cold. When the surgeon dictates "LIMA to LAD, SVG to OM1, SVG to PDA," you need to instantly write "3-vessel CABG with LIMA-LAD, SVG-OM1, SVG-PDA." If you hesitate, the operative note rhythm breaks. Also memorize the mediastinal compartments — the compartment predicts the differential for any mass you will document.
02 Scribe Documentation Framework
Cardiothoracic surgery documentation straddles the operating room, the ICU, the stepdown floor, and the outpatient clinic. As a scribe, you will shift between pre-op clinic notes, post-op day notes, weaning and extubation notes, and discharge summaries. Each has its own rhythm.
Chief complaint: Most common: chest pain, dyspnea on exertion (DOE), syncope, palpitations, lung nodule, mass for biopsy, valvular murmur for surgical evaluation, or a transfer from cardiology/pulmonology for operative consideration.
Cardiac HPI: Chest pain characterization (CCS class), NYHA functional class, syncope/pre-syncope, orthopnea, paroxysmal nocturnal dyspnea (PND), lower-extremity edema, palpitations, prior MI, prior PCI/stents, prior cardiac surgery (redo sternotomy is a major risk), prior radiation, most recent echo (EF, valve gradients, chamber sizes), catheterization findings (which vessels, percent stenosis, SYNTAX score), and stress test results.
Thoracic HPI: Smoking history in pack-years with quit date, occupational exposures (asbestos, silica), cough, hemoptysis, weight loss, dysphagia (solids vs liquids, progression), reflux symptoms, hoarseness (recurrent laryngeal nerve involvement), Horner syndrome (apical tumor), pulmonary function tests (FEV1, DLCO), and any prior lung resection or radiation.
Vitals: BP in both arms (arm-to-arm difference for subclavian disease or dissection), HR and rhythm, SpO2 on room air, BMI.
Cardiac exam: JVP estimation, carotid upstroke and bruits, apical impulse (displaced, sustained, hyperdynamic), heart sounds (S1, S2 intensity and splitting, S3, S4), murmurs (systolic vs diastolic, location, radiation, grade I–VI/VI), lower extremity edema, peripheral pulses.
Thoracic exam: Respiratory rate and effort, chest wall inspection (pectus, prior scars), tracheal position, percussion (dullness over effusion), auscultation (breath sounds, rales, rhonchi, wheeze, egophony), palpation of supraclavicular nodes.
Cardiothoracic A&P is problem-based and always includes: (1) primary operative diagnosis with staging or severity, (2) estimated surgical risk (STS risk score, EuroSCORE II, frailty), (3) planned operation with approach (sternotomy vs thoracotomy vs VATS vs robotic), (4) pre-op workup still needed (carotid duplex, dental clearance, PFTs, stress test, cath, echo, CT), (5) medication adjustments (hold anticoagulation, bridge if mechanical valve, continue beta blocker and statin), and (6) multidisciplinary coordination (heart team, tumor board, anesthesia, cardiology, pulmonology).
Post-Op ICU Note Structure
Post-op cardiac ICU notes are highly structured, organ-system based, and change dramatically by post-op day (POD). A typical POD 1 note moves through Neuro (sedation level, RASS, extubation readiness), CV (rhythm, rate, BP, pressors, inotropes, CI, CVP, SvO2, pacing wires, chest tube output), Resp (ventilator settings, ABG, weaning trial), GI (diet, bowel function), GU (urine output, creatinine trend), Heme (Hgb, platelets, coagulation, drain output), ID (temperature, antibiotics, cultures), Endo (glucose, insulin drip), and Lines/drains (central line, arterial line, chest tubes, pacing wires, Foley).
In CT ICU the phrases "on epi at 4, milrinone at 0.375, norepi at 8" will be said fast. Write the drug and the dose in that exact order every time. Do not convert units on the fly — capture what the intensivist says verbatim and let the pharmacist reconcile if needed.
03 Coronary Artery Disease & CABG Indications Cardiac
Coronary artery disease (CAD) is the most common reason patients enter a cardiac surgeon's clinic. Atherosclerotic plaque in the epicardial coronaries reduces myocardial perfusion, producing angina, infarction, ischemic cardiomyopathy, and sudden death. The cardiac surgeon is consulted when the lesion distribution, complexity, comorbidities, or ventricular function make surgical revascularization (CABG) preferable to percutaneous coronary intervention (PCI).
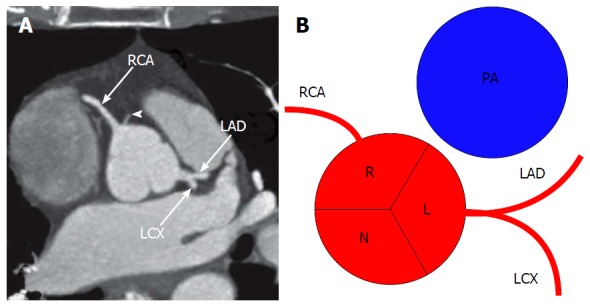
Indications for CABG over PCI
The 2021 ACC/AHA/SCAI coronary revascularization guideline and heart-team consensus favor surgical revascularization in: significant left main disease (≥ 50% stenosis), triple-vessel disease especially with reduced EF or diabetes, two-vessel disease with proximal LAD involvement, complex anatomy with a high SYNTAX score (> 32), failed PCI, and mechanical complications of MI (VSD, acute MR from papillary muscle rupture, free-wall rupture). The SYNTAX trial established CABG as superior to PCI in high-complexity three-vessel and left-main disease.
The CABG Operation
Standard CABG is performed via median sternotomy, typically on cardiopulmonary bypass (CPB) with cardioplegic arrest (on-pump). The surgeon harvests the LIMA pedicled from the chest wall, and additional conduits as needed: right internal mammary artery (RIMA), radial artery (RA), and saphenous vein grafts (SVG) from the leg (open or endoscopic vein harvest). After heparinization (ACT > 400), CPB is initiated via aortic and right atrial (or bicaval) cannulation; the aorta is cross-clamped and cardioplegia is delivered to arrest the heart. Distal anastomoses are constructed onto the target coronaries, then proximal anastomoses onto the ascending aorta (except the LIMA, whose proximal end stays attached to the subclavian). The cross-clamp is removed, the heart is reperfused, weaned from bypass, and heparin is reversed with protamine. Off-pump CABG (OPCAB) and beating-heart CABG avoid CPB using stabilizer devices; fewer centers do these routinely but the technique is important when CPB is contraindicated (porcelain aorta, severe coagulopathy).
LIMA to LAD: The gold standard. 10-year patency > 90%. Used for the LAD in virtually every CABG when possible.
RIMA: Excellent durability but increases deep sternal wound infection risk in diabetics when both mammaries are harvested (bilateral IMA / BIMA).
Radial artery: Arterial conduit, good patency when grafted to a target with > 70% stenosis; Allen test or Doppler needed pre-op.
Saphenous vein graft (SVG): Most common workhorse conduit. 10-year patency ~50–60%. Can be harvested open or endoscopically.
"Three-vessel CABG" tells you nothing useful by itself. Always document which grafts went where: LIMA to LAD, SVG to OM1, SVG to PDA. Operative notes and discharge summaries depend on this detail.
04 Aortic Valve Disease (AS, AR) Valvular
Aortic Stenosis (AS)
Aortic stenosis is narrowing of the aortic valve, most commonly from calcific degeneration in elderly patients, bicuspid aortic valve (BAV) in younger adults (presenting in the 50s–60s), or rheumatic disease. The classic triad is angina, syncope, and heart failure; once symptoms appear, untreated severe AS has a median survival of 2–3 years.
Severity is graded by echocardiography using peak aortic jet velocity, mean transvalvular gradient, and aortic valve area (AVA):
| Stage | Peak Velocity | Mean Gradient | AVA |
|---|---|---|---|
| Mild | 2.6–2.9 m/s | < 20 mmHg | > 1.5 cm2 |
| Moderate | 3.0–3.9 m/s | 20–39 mmHg | 1.0–1.5 cm2 |
| Severe | ≥ 4.0 m/s | ≥ 40 mmHg | < 1.0 cm2 |
| Very severe | ≥ 5.0 m/s | ≥ 60 mmHg | < 0.8 cm2 |
Management follows the 2020 ACC/AHA valvular heart disease guideline. Surgical or transcatheter AVR is indicated in symptomatic severe AS, asymptomatic severe AS with LV dysfunction (EF < 50%), asymptomatic very severe AS, or AS with another indication for cardiac surgery. SAVR (surgical AVR) with a mechanical or bioprosthetic valve is performed via sternotomy on CPB. TAVR (transcatheter AVR) is a catheter-delivered valve (typically transfemoral) and has become the default for most intermediate- and high-risk patients and many low-risk elderly patients per PARTNER 3.
Aortic Regurgitation (AR)
AR is incompetence of the aortic valve producing diastolic backflow into the LV. Etiologies include bicuspid valve, aortic root dilation, endocarditis, rheumatic disease, and aortic dissection. Chronic AR produces eccentric LV hypertrophy and dilation (the "volume overload" ventricle); acute AR (from endocarditis or dissection) produces flash pulmonary edema and cardiogenic shock because the LV cannot dilate fast enough. Severity is graded by regurgitant jet width, vena contracta, regurgitant volume/fraction, and effective regurgitant orifice area (EROA). AVR is indicated for symptomatic severe AR or asymptomatic severe AR with LV dysfunction (EF ≤ 55%) or LV dilation (LVESD > 50 mm).
05 Mitral Valve Disease (MR, MS) Valvular
Mitral Regurgitation (MR)
MR is divided into primary (degenerative) — a leaflet or chordal problem, most often myxomatous degeneration with prolapse (Barlow disease, fibroelastic deficiency) or flail leaflet from ruptured chordae — and secondary (functional) — a ventricular problem (ischemic cardiomyopathy, dilated cardiomyopathy) in which annular dilation and papillary muscle displacement prevent leaflet coaptation despite normal leaflets.
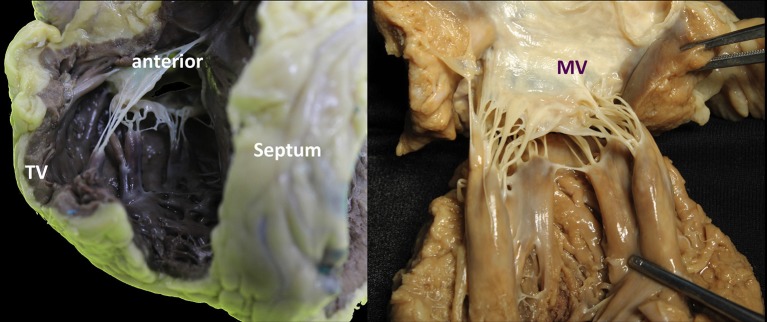
Severity grading uses vena contracta, regurgitant volume, regurgitant fraction, and EROA:
| Grade | Vena Contracta | Reg Volume | EROA |
|---|---|---|---|
| Mild | < 0.3 cm | < 30 mL | < 0.20 cm2 |
| Moderate | 0.3–0.69 cm | 30–59 mL | 0.20–0.39 cm2 |
| Severe | ≥ 0.7 cm | ≥ 60 mL | ≥ 0.40 cm2 |
The mantra for degenerative MR is "repair, don't replace." Mitral valve repair (annuloplasty ring, leaflet resection, chordal replacement with neochords, Alfieri edge-to-edge) preserves native valve tissue and the subvalvular apparatus. For non-repairable valves, mitral valve replacement (MVR) is performed. The transcatheter MitraClip is FDA-approved for selected prohibitive-risk degenerative MR and for functional MR in selected HF patients per COAPT.
Mitral Stenosis (MS)
Mitral stenosis is overwhelmingly rheumatic in the modern era (outside of rare congenital or degenerative forms). The stenotic orifice produces a pressure gradient from LA to LV, causing LA enlargement, atrial fibrillation, pulmonary hypertension, and right heart failure. MS severity by mitral valve area (MVA): mild > 1.5 cm2, moderate 1.0–1.5 cm2, severe < 1.0 cm2, very severe < 0.8 cm2. Treatment is percutaneous balloon mitral valvuloplasty (PBMV) in favorable anatomy (Wilkins score ≤ 8) or surgical MVR for unfavorable valves or concomitant disease.
06 Tricuspid & Pulmonic Valve Disease Valvular
Tricuspid regurgitation (TR) is most often functional, driven by RV dilation from left-sided disease, pulmonary hypertension, or atrial fibrillation with annular dilation. Primary causes include endocarditis (IVDU), carcinoid, Ebstein anomaly, and pacemaker lead impingement.
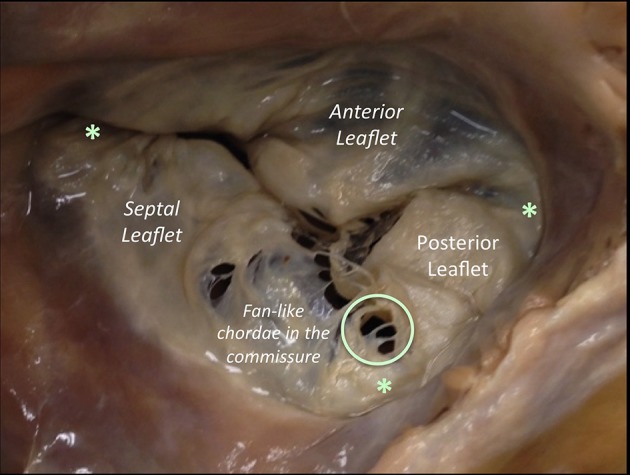
Severity: mild, moderate, severe, and massive/torrential (newer grades). Isolated severe TR causes peripheral edema, ascites, hepatic congestion, and the classic "pulsatile liver." Repair via annuloplasty ring (DeVega, rigid, or semi-rigid) is typically performed at the time of left-sided surgery. Isolated late TR after left-sided surgery is a morbid entity; transcatheter options (TriClip, EVOQUE) are emerging.
Pulmonic valve disease is uncommon in adult acquired disease and is mostly seen in congenital (Tetralogy of Fallot, pulmonary stenosis) or carcinoid contexts. The Ross procedure uses the patient's own pulmonic valve to replace the aortic valve, with a pulmonary homograft in the pulmonic position — mostly reserved for younger patients.
07 Infective Endocarditis (Surgical) Valvular
Infective endocarditis (IE) is infection of the endocardial surface, most commonly the valves, producing vegetations of fibrin, platelets, and organisms. The cardiac surgeon is consulted when medical therapy alone will not clear infection or when mechanical complications arise.
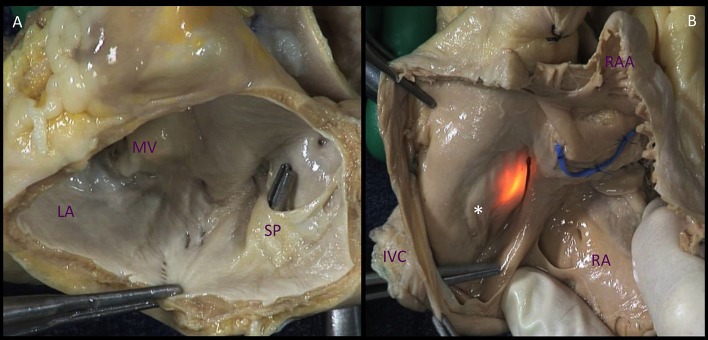
Per the AHA IE scientific statement and 2023 ESC endocarditis guideline, surgery is indicated for:
Heart failure: Valve destruction causing acute severe regurgitation with pulmonary edema or cardiogenic shock — emergency surgery.
Uncontrolled infection: Perivalvular abscess, fistula, persistent bacteremia despite appropriate antibiotics, fungal or highly resistant organisms.
Embolic prevention: Large vegetations (> 10 mm) especially after one embolic event, or vegetations on the anterior mitral leaflet.
Surgery involves debridement of infected tissue, valve replacement (usually bioprosthetic in the acute setting), and reconstruction of annular defects with pericardial patches. Root abscess may require full aortic root replacement (homograft is often preferred for prosthetic valve endocarditis with annular destruction). A full 6-week course of IV antibiotics typically continues post-op.
08 Thoracic Aortic Aneurysm & Dissection Aortic
The thoracic aorta is divided into the root (sinuses of Valsalva, sinotubular junction), ascending aorta, arch, and descending thoracic aorta. Aneurysmal dilation (≥ 1.5× normal) and acute dissection are the two dominant surgical diseases. Connective tissue disorders (Marfan, Loeys-Dietz, Ehlers-Danlos vascular type), bicuspid aortic valve, hypertension, and family history are the major risk drivers.
Thoracic Aortic Aneurysm (TAA)
Asymptomatic TAAs are typically found incidentally on chest imaging. Surgical thresholds per the 2022 ACC/AHA aortic disease guideline: ascending aorta ≥ 5.5 cm in most patients, ≥ 5.0 cm in bicuspid valve patients or Marfan with additional risk features, ≥ 4.5 cm in Marfan with family history of dissection, or growth > 0.5 cm/year. Descending TAA surgical threshold is typically ≥ 5.5 cm, with TEVAR as the default for anatomically suitable patients.
Acute Aortic Dissection
A dissection begins with an intimal tear allowing blood to enter the aortic media, creating a false lumen that propagates antegrade or retrograde. The Stanford classification is operationally dominant:
| Stanford | Definition | Management |
|---|---|---|
| Type A | Involves the ascending aorta (regardless of entry site) | Emergency surgery — open repair |
| Type B | Does not involve the ascending aorta (distal to left subclavian) | Medical management first; TEVAR for complications |
The older DeBakey classification is still dictated: Type I involves ascending + arch + descending, Type II is confined to the ascending aorta, Type IIIa is descending proximal to the diaphragm, Type IIIb extends below the diaphragm.
Type A dissection is a surgical emergency with an untreated mortality of roughly 1% per hour for the first 48 hours. Operative repair involves replacement of the ascending aorta with a Dacron graft, with or without concomitant hemiarch or total arch replacement, aortic root reconstruction (supracoronary graft, Bentall composite valved graft, or valve-sparing root replacement / David procedure), and occasionally a frozen elephant trunk (FET) for extensive arch and descending disease. The procedure usually requires deep hypothermic circulatory arrest (DHCA) with antegrade or retrograde cerebral perfusion.
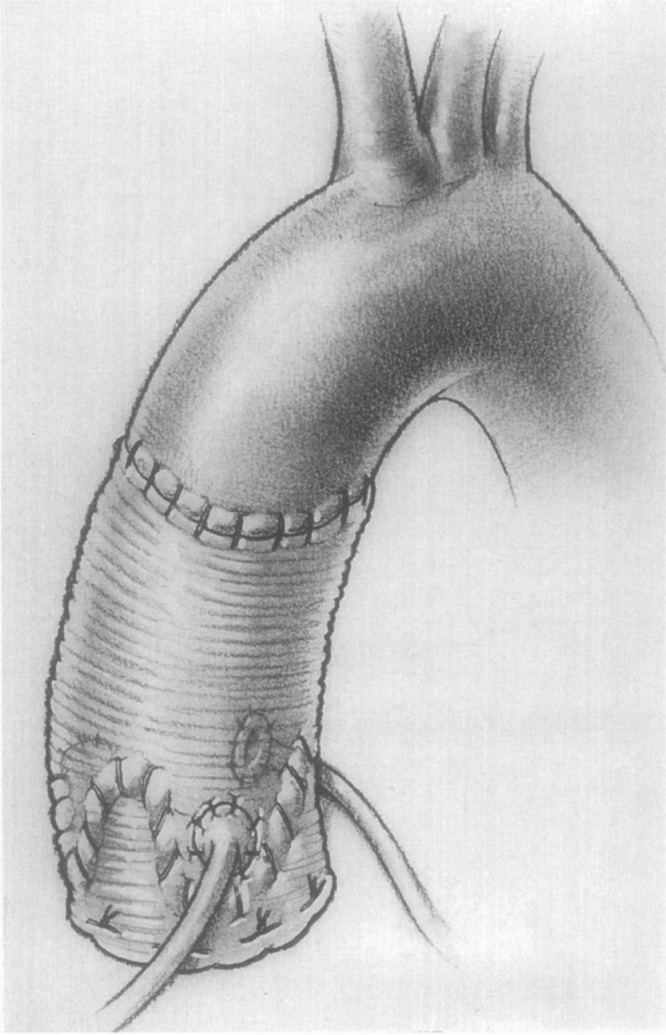
Type B dissection is managed medically with anti-impulse therapy (IV beta blockade targeting HR < 60, then afterload reduction) unless complicated by rupture, malperfusion (visceral, renal, spinal, limb), refractory pain, or rapid expansion — at which point TEVAR is the default per INSTEAD-XL.
Traumatic aortic transection (blunt thoracic trauma, classically at the ligamentum arteriosum just distal to the left subclavian) is repaired urgently with TEVAR in almost all modern centers.
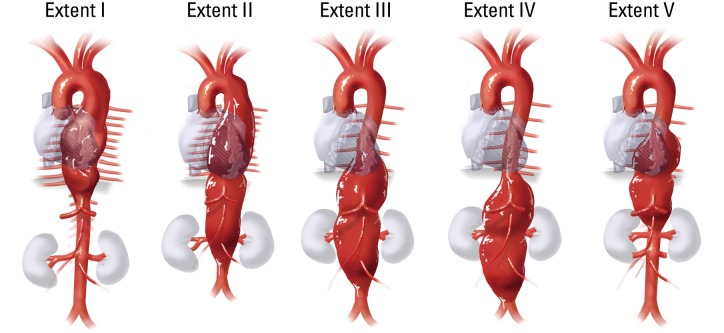
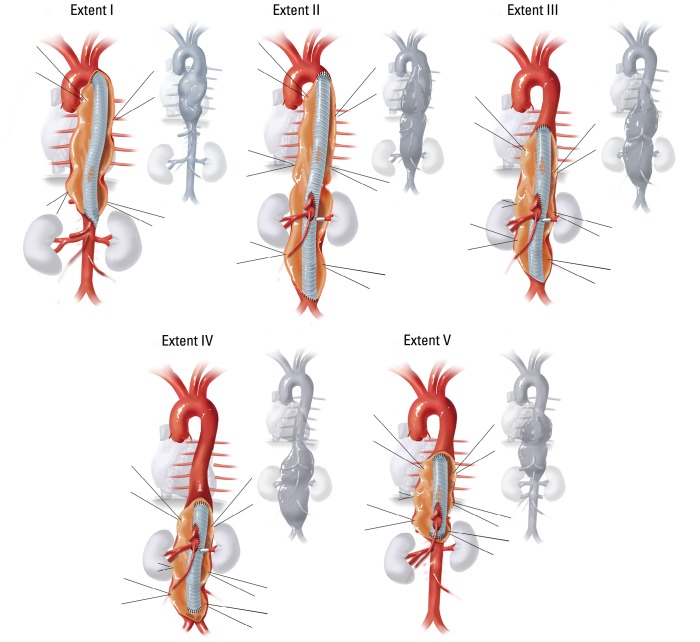
Type I: Distal to left subclavian to above renal arteries.
Type II: Distal to left subclavian to below renal arteries — most extensive.
Type III: Distal descending thoracic (below T6) to below renal arteries.
Type IV: Entire abdominal aorta from diaphragm to aortic bifurcation.
Type V: Distal descending thoracic (below T6) to just above renal arteries (modified).
09 Atrial Fibrillation & Maze Procedure Cardiac
Atrial fibrillation (AF) is the most common arrhythmia and is managed surgically when a patient is undergoing another cardiac operation or rarely as a standalone procedure for refractory drug-failed AF. Most AF originates from ectopic foci near the pulmonary vein ostia. The Cox-Maze IV procedure creates a series of ablation lesions (radiofrequency or cryothermy) in the left and right atria to isolate the pulmonary veins and interrupt reentrant circuits. Left atrial appendage (LAA) exclusion via stapler or clip (AtriClip) is routinely added to reduce embolic stroke risk. Concomitant surgical ablation should be considered in patients with AF undergoing valve or CABG surgery per the 2017 STS surgical ablation guideline.
10 Heart Failure, LVAD & Transplant Cardiac
Advanced heart failure refractory to maximal medical therapy (INTERMACS profiles 1–4) is evaluated for mechanical circulatory support (MCS) and heart transplant. Cardiothoracic surgeons lead both programs.
LVAD
The left ventricular assist device (LVAD) is an implantable continuous-flow centrifugal pump (HeartMate 3 is the current dominant device) that draws blood from the LV apex via an inflow cannula and returns it to the ascending aorta via an outflow graft, with a percutaneous driveline exiting through the abdominal wall to a controller and battery pack.
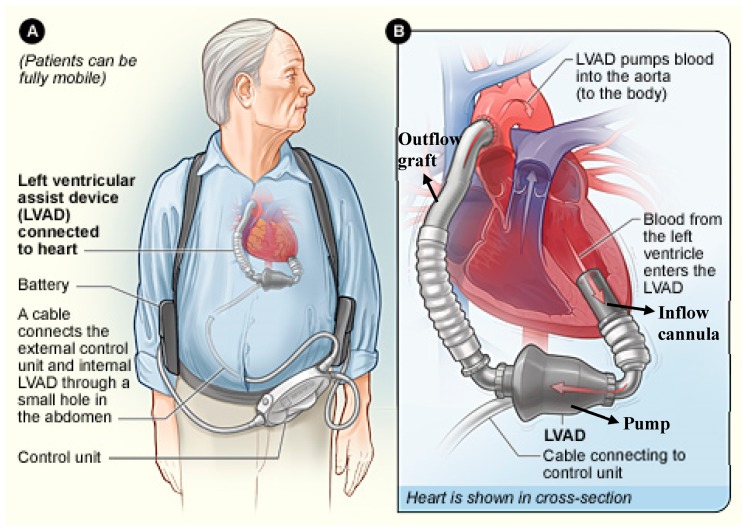
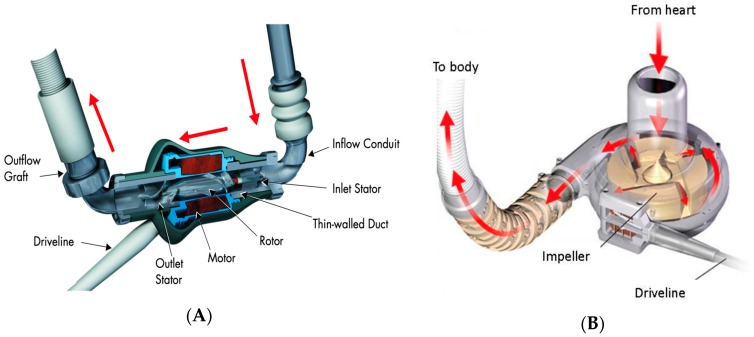
Indications are bridge-to-transplant (BTT), bridge-to-candidacy, and destination therapy (DT) for non-transplant candidates. MOMENTUM 3 established HeartMate 3 as the standard of care. Complications scribes will document: driveline infection, GI bleeding (from acquired von Willebrand deficiency and AV malformations), pump thrombosis, stroke, and right heart failure.
Heart Transplant
Orthotopic heart transplantation remains the gold standard for end-stage HF. Candidates are listed per UNOS criteria (the 2018 allocation system uses 6 status tiers; Status 1–2 are sickest, often on ECMO or temporary MCS). The operation removes the recipient heart leaving atrial or bicaval cuffs and implants the donor heart in the orthotopic position.
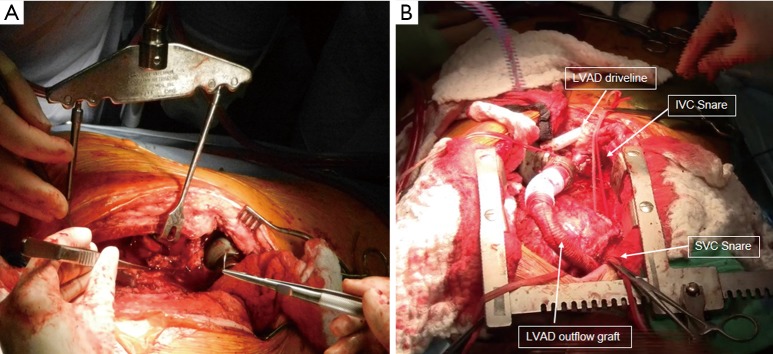
Post-op management is driven by the ISHLT guidelines: induction immunosuppression (basiliximab, ATG), maintenance (tacrolimus + MMF + steroids), and surveillance endomyocardial biopsies for rejection.
11 Cardiac Tumors, Pericardial & Adult Congenital Disease Cardiac
Cardiac myxoma is the most common primary cardiac tumor, most often a pedunculated left atrial mass arising from the fossa ovalis. Patients present with embolic events, positional obstructive symptoms (classic "tumor plop"), and constitutional symptoms. Treatment is surgical excision with a rim of atrial septum to prevent recurrence.
Constrictive pericarditis is a rigid, fibrotic, often calcified pericardium that restricts diastolic filling. Causes include prior cardiac surgery, radiation, tuberculosis, and idiopathic. Presents with right-sided heart failure, Kussmaul sign, pericardial knock, and equalization of diastolic pressures on catheterization. Treatment is pericardiectomy — complete stripping of the pericardium off both ventricles — via sternotomy or left anterolateral thoracotomy.
Adult congenital heart disease (ACHD) seen on cardiac surgery services includes atrial septal defect (ASD) closure (percutaneous or surgical for primum, sinus venosus, and large secundum defects), ventricular septal defect closure, patent ductus arteriosus ligation, coarctation repair (often endovascular in adults), and reoperations for previously repaired Tetralogy of Fallot (pulmonary valve replacement for chronic PR), Ebstein anomaly, and Fontan patients with failing circulation.
12 Lung Cancer & Pulmonary Nodules Thoracic
Lung cancer is the leading cause of cancer death in the US and the dominant diagnosis on a general thoracic service. The pathologic split: non-small cell lung cancer (NSCLC, ~85%) — adenocarcinoma, squamous cell, large cell — and small cell lung cancer (SCLC, ~15%), which is rarely surgical (managed with chemoradiation except for very early stage T1N0).

Pulmonary Nodules
The workup of a solitary pulmonary nodule is guided by the Fleischner Society 2017 guidelines: size, solid vs subsolid vs ground-glass, and patient risk category drive follow-up intervals. High-risk nodules (> 8 mm solid in a smoker) prompt PET/CT, biopsy, or up-front resection.
NSCLC Staging & Management
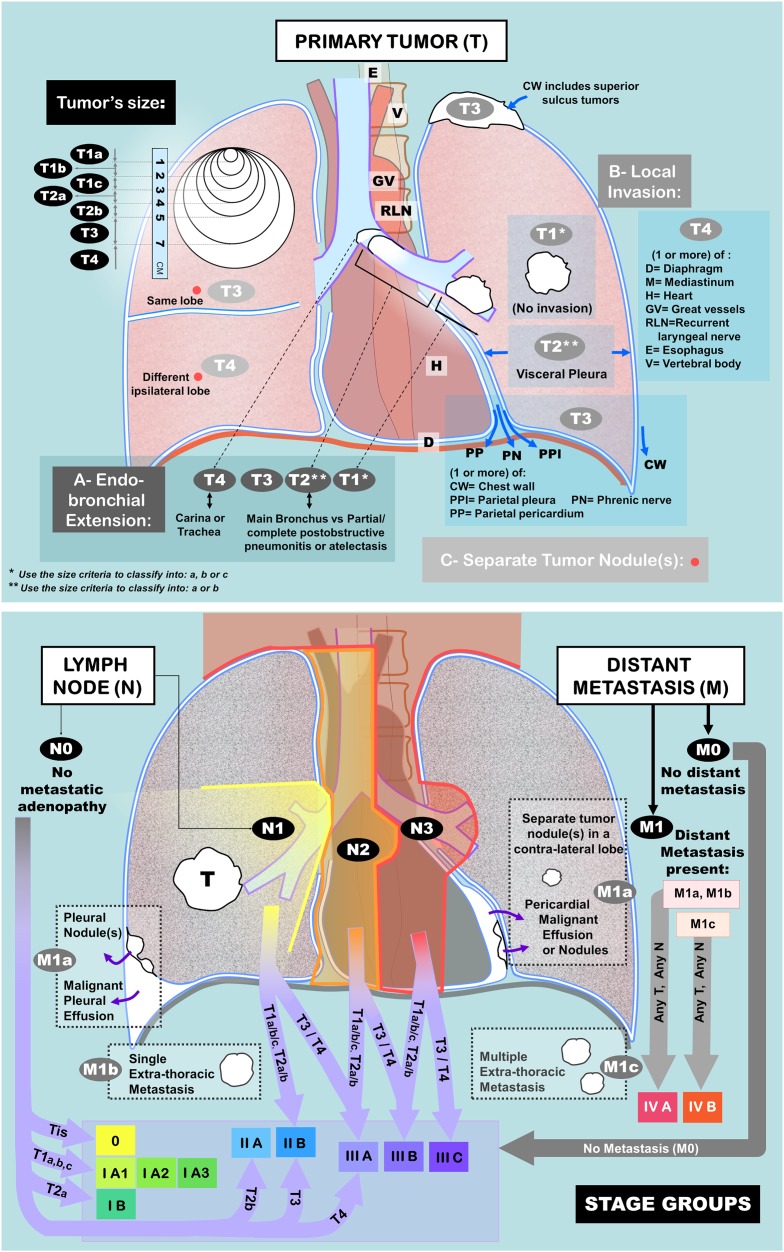
Staging follows the IASLC 8th edition TNM and is the basis of all treatment decisions. Complete pretreatment workup includes CT chest/abdomen/pelvis, PET/CT, brain MRI (stage II+ or symptomatic), PFTs (FEV1 and DLCO for resection candidacy), and mediastinal nodal sampling via EBUS-TBNA or mediastinoscopy.
| T Stage | Criteria |
|---|---|
| T1a | ≤ 1 cm |
| T1b | > 1–2 cm |
| T1c | > 2–3 cm |
| T2a | > 3–4 cm or visceral pleura / main bronchus involvement |
| T2b | > 4–5 cm |
| T3 | > 5–7 cm, chest wall, phrenic nerve, or separate nodule same lobe |
| T4 | > 7 cm, diaphragm, mediastinum, heart, great vessels, carina, or nodule different ipsilateral lobe |
| N Stage | Criteria |
|---|---|
| N0 | No regional node involvement |
| N1 | Ipsilateral hilar, intrapulmonary |
| N2 | Ipsilateral mediastinal or subcarinal |
| N3 | Contralateral mediastinal/hilar or supraclavicular |
Stage I and II NSCLC are surgical. The standard operation is anatomic lobectomy with mediastinal lymph node dissection, most often via VATS or robotic approach. JCOG0802/CALGB 140503 established that sublobar resection (segmentectomy) is non-inferior to lobectomy for small (≤ 2 cm) peripheral NSCLC. Stage IIIA is often treated with neoadjuvant chemo-immunotherapy (CheckMate 816) followed by resection per NCCN NSCLC guidelines. Stage IIIB and IV are non-surgical.
13 Pleural Disease Thoracic
Pleural effusion: Fluid in the pleural space, classified as transudative (heart failure, cirrhosis) or exudative (infection, malignancy, PE) by Light's criteria. Thoracic surgery is involved when drainage is required: thoracentesis (bedside), chest tube, indwelling pleural catheter (PleurX) for recurrent malignant effusions, or VATS pleurodesis.
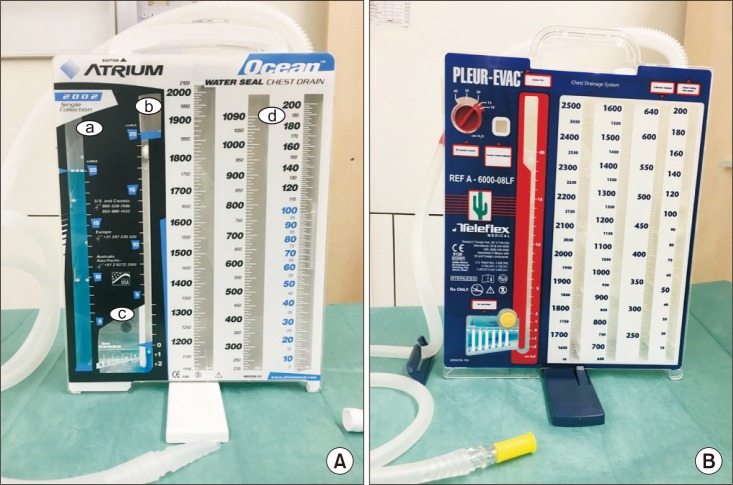
Empyema: Infected pleural fluid. Evolves through three stages: exudative (stage I, free-flowing, drain with chest tube), fibrinopurulent (stage II, loculated, needs fibrinolytics or VATS decortication), and organized (stage III, thick peel, requires VATS or open decortication via thoracotomy).
Pneumothorax: Primary spontaneous (tall thin young males with apical blebs), secondary (COPD, cystic fibrosis), traumatic, and iatrogenic. Small asymptomatic PTX can be observed; larger or symptomatic PTX is treated with needle decompression (if tension) followed by chest tube. Recurrent or persistent air leak goes to VATS bleb resection with pleurodesis (mechanical abrasion or talc). Tension pneumothorax is a clinical diagnosis and a surgical emergency.
Malignant pleural mesothelioma: Asbestos-related mesothelial malignancy with poor prognosis. Surgical options are extrapleural pneumonectomy (EPP) — en bloc resection of lung, pleura, pericardium, and hemidiaphragm — or the lung-preserving pleurectomy/decortication (P/D), usually combined with chemotherapy and sometimes radiation.
14 Esophageal Cancer & Benign Esophageal Disease Esophageal
Esophageal Cancer
Two dominant histologies: adenocarcinoma (distal esophagus and GEJ, associated with reflux and Barrett esophagus, rising in incidence in Western populations) and squamous cell carcinoma (proximal and mid esophagus, associated with smoking and alcohol). Staging workup includes endoscopy with biopsy, EUS (for T and N staging), PET/CT, and diagnostic laparoscopy for GEJ tumors. Neoadjuvant chemoradiation (CROSS protocol) followed by esophagectomy is standard for locally advanced disease.
Esophagectomy approaches:
- Ivor Lewis (transthoracic): Laparotomy or laparoscopy for gastric mobilization plus right thoracotomy (or thoracoscopy/robotic) for esophageal mobilization and intrathoracic anastomosis. The workhorse for distal and GEJ tumors.
- McKeown (three-field): Right chest, abdomen, and left neck, with a cervical anastomosis. Used for mid and upper esophageal tumors.
- Transhiatal: Laparotomy and left neck incision; the esophagus is bluntly dissected through the hiatus. Avoids thoracotomy but sacrifices full mediastinal lymphadenectomy.
Benign Esophageal Disease
GERD: Refractory reflux despite PPI therapy is treated with laparoscopic Nissen fundoplication (360° wrap) or partial wraps (Toupet 270°, Dor 180°) for patients with impaired motility. Hiatal hernia is classified: Type I sliding (GEJ displaced), Type II paraesophageal (fundus herniates, GEJ fixed), Type III mixed, Type IV with additional organ herniation. Large paraesophageal hernias are repaired laparoscopically with sac excision, crural closure, and fundoplication.
Achalasia: Loss of esophageal peristalsis and failure of LES relaxation. Diagnosis by manometry (aperistalsis, elevated LES pressure with failed relaxation) and barium swallow ("bird's beak"). Treatment is laparoscopic Heller myotomy with partial fundoplication or POEM (peroral endoscopic myotomy). Pneumatic dilation is an endoscopic alternative.
15 Mediastinal & Thymic Disease Mediastinal
Mediastinal masses are classified by compartment. Anterior compartment masses dominate thoracic surgery referrals: the differential is the "4 T's" — thymoma, teratoma (and other germ cell tumors), thyroid (substernal goiter), and "terrible lymphoma."
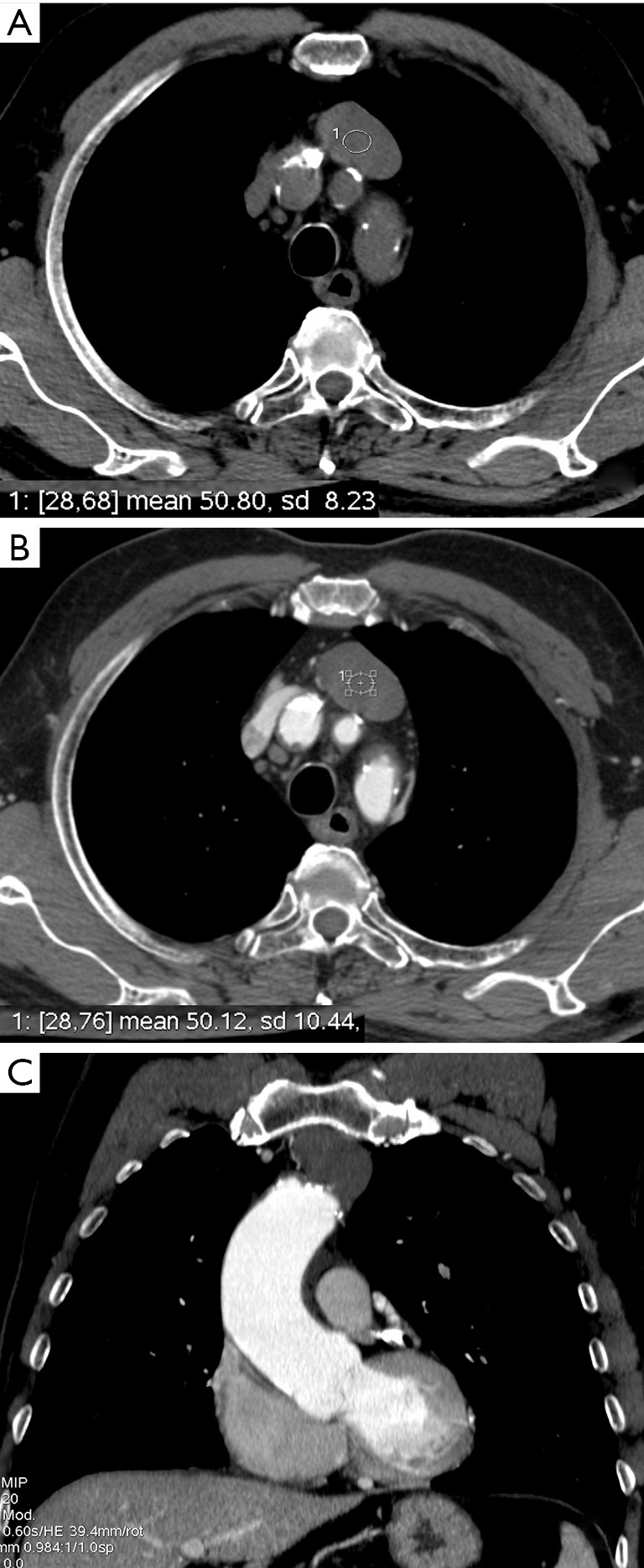
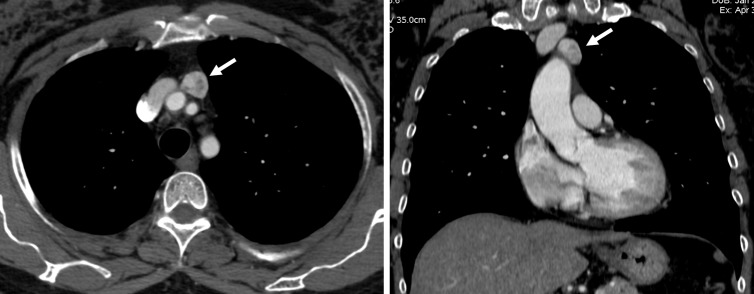
Thymoma & Thymic Carcinoma
Thymomas are the most common anterior mediastinal tumor in adults. About 30–50% of thymoma patients have myasthenia gravis (MG). Staging uses the Masaoka-Koga system:
| Stage | Description |
|---|---|
| I | Completely encapsulated |
| IIA | Microscopic transcapsular invasion |
| IIB | Macroscopic invasion into thymic or mediastinal fat |
| III | Invasion into neighboring organs (pericardium, great vessels, lung) |
| IVA | Pleural or pericardial dissemination |
| IVB | Lymphogenous or hematogenous metastasis |
Treatment is complete surgical resection — thymectomy via sternotomy, VATS, or robotic approach — with adjuvant radiation for stage II and above.
Myasthenia Gravis (Thymectomy)
Thymectomy is indicated in generalized MG even without thymoma for patients age 18–65 with positive AChR antibodies, per MGTX trial. Pre-op optimization of MG with plasmapheresis or IVIG is standard to reduce post-op respiratory failure.
16 Chest Wall, Tracheal & Diaphragm Disease Chest Wall
Pectus excavatum is a posterior displacement of the sternum producing a "funnel chest." Severity is graded by the Haller index on CT (transverse chest diameter divided by sternum-to-vertebra distance); > 3.25 is considered severe. Repair options are the minimally invasive Nuss procedure (a curved metal bar placed behind the sternum via small bilateral incisions) and the open Ravitch procedure (costal cartilage resection and sternal osteotomy). Pectus carinatum ("pigeon chest") is anterior protrusion, usually managed with bracing but occasionally Ravitch-style repair.
Tracheal stenosis from prolonged intubation, tracheostomy, or idiopathic inflammation may require tracheal resection and reconstruction. Up to 50% of the trachea can be resected and primary anastomosis performed via a cervical approach, with head flexion maintained post-op to avoid anastomotic tension. Tracheal tumors (squamous, adenoid cystic) are also managed with resection.
Diaphragmatic paralysis/eventration is addressed with plication (VATS or open) when symptomatic. Thoracic outlet syndrome is covered in the vascular surgery guide and is typically managed jointly.
17 Cardiac Procedures A to Z
| Procedure | Description / Indication |
|---|---|
| Median sternotomy | Vertical division of the sternum with a sternal saw; standard access for CABG, valve, aortic, and transplant operations. |
| Cardiopulmonary bypass (CPB) | Extracorporeal circuit diverting blood from the RA/vena cavae to an oxygenator/reservoir and returning it to the ascending aorta. Requires full heparinization and protamine reversal. |
| CABG (on-pump) | Bypass of stenotic coronary arteries using LIMA, RIMA, radial, or SVG conduits with cardioplegic arrest. |
| OPCAB / beating heart CABG | CABG without CPB using myocardial stabilizer devices. |
| SAVR | Surgical aortic valve replacement, mechanical or bioprosthetic. |
| TAVR | Transcatheter aortic valve replacement (transfemoral is default). |
| Aortic root replacement (Bentall) | Composite valved graft replacing aortic valve + root with coronary reimplantation. |
| Valve-sparing root replacement (David) | Root replacement preserving the native aortic valve; reimplantation of leaflets into a Dacron graft. |
| Hemiarch replacement | Beveled replacement of the proximal aortic arch, typically during Type A dissection repair. |
| Total arch replacement | Complete arch replacement with reimplantation of the brachiocephalic, left common carotid, and left subclavian arteries. |
| Frozen elephant trunk (FET) | Hybrid graft with sewn proximal and stented distal portion for extensive arch + descending disease. |
| TEVAR | Thoracic endovascular aortic repair for descending TAA, complicated type B dissection, transection. |
| Mitral valve repair | Annuloplasty ring, leaflet resection, chordal replacement (neochords), Alfieri edge-to-edge stitch. |
| Mitral valve replacement (MVR) | Mechanical or bioprosthetic replacement when repair is not feasible. |
| MitraClip | Transcatheter edge-to-edge repair for prohibitive-risk MR. |
| Tricuspid repair/replacement | Annuloplasty ring typically, rarely replacement. |
| Cox-Maze IV | RF or cryoablation lesion set for atrial fibrillation; LAA exclusion routinely added. |
| LAA exclusion (AtriClip) | Epicardial clip across the LAA for stroke prevention. |
| Pericardiectomy | Stripping of pericardium for constrictive pericarditis or chronic effusion. |
| ASD closure | Surgical or percutaneous closure of atrial septal defect. |
| LVAD implantation | HeartMate 3 (or prior HVAD) placement with apical inflow cannula and outflow graft to ascending aorta. |
| Heart transplant | Orthotopic donor heart implantation via bicaval or biatrial technique. |
| IABP | Intra-aortic balloon pump for cardiogenic shock or low-output states. |
| ECMO (VA / VV) | Extracorporeal membrane oxygenation; VA for cardiogenic shock, VV for respiratory failure. |
18 Thoracic Procedures A to Z
| Procedure | Description / Indication |
|---|---|
| Posterolateral thoracotomy | Incision along the 5th or 6th interspace; classic open thoracic access. |
| Axillary (muscle-sparing) thoracotomy | Smaller access sparing the latissimus dorsi. |
| VATS (video-assisted thoracoscopic surgery) | Port-based minimally invasive thoracic access using a camera and instruments. |
| Robotic thoracic surgery | Da Vinci platform for lobectomy, segmentectomy, thymectomy, and esophagectomy. |
| Lobectomy | Anatomic resection of a lobe with lymph node dissection — the standard for early NSCLC. |
| Segmentectomy | Anatomic resection of a bronchopulmonary segment; acceptable for small peripheral NSCLC. |
| Wedge resection | Non-anatomic stapled resection for diagnosis or metastasectomy. |
| Pneumonectomy | Removal of an entire lung; reserved when lobectomy cannot achieve R0. |
| Sleeve lobectomy | Lobectomy with bronchial sleeve resection and reanastomosis to spare lung parenchyma. |
| Decortication | Removal of thickened pleural peel for empyema or trapped lung. |
| Pleurodesis | Obliteration of the pleural space with talc, mechanical abrasion, or chemical agents. |
| Chest tube placement | Tube thoracostomy for pneumothorax, hemothorax, effusion, or empyema. |
| Indwelling pleural catheter (PleurX) | Tunneled catheter for home drainage of recurrent malignant effusions. |
| Mediastinoscopy | Cervical approach for biopsy of paratracheal and subcarinal nodes. |
| EBUS-TBNA | Endobronchial ultrasound with transbronchial needle aspiration for mediastinal staging. |
| Lung volume reduction surgery (LVRS) | Resection of hyperinflated emphysematous lung in selected COPD patients. |
| Lung transplant (single or bilateral) | For end-stage lung disease (IPF, COPD, CF, pulmonary hypertension). |
| Esophagectomy (Ivor Lewis) | Abdomen + right thorax with intrathoracic anastomosis. |
| Esophagectomy (McKeown) | Three-field: right thorax, abdomen, left neck anastomosis. |
| Transhiatal esophagectomy | Abdomen + left neck with blunt mediastinal dissection. |
| Heller myotomy + Dor/Toupet | Laparoscopic LES myotomy with partial fundoplication for achalasia. |
| Nissen fundoplication | 360° laparoscopic wrap for GERD. |
| Paraesophageal hernia repair | Sac excision, crural closure (± mesh), fundoplication. |
| Nuss procedure | Minimally invasive pectus excavatum repair with retrosternal bar. |
| Ravitch procedure | Open pectus repair with costal cartilage resection and sternal osteotomy. |
| Tracheal resection | Segmental resection of stenotic or tumor-bearing trachea with primary anastomosis. |
| Thymectomy | Resection of thymus via sternotomy, VATS, or robotic approach. |
| Diaphragm plication | VATS or open plication for symptomatic diaphragmatic eventration/paralysis. |
19 Post-Operative ICU Care & Complications
Early Post-Cardiac Surgery
Post-CABG/valve patients arrive in the CVICU intubated, with invasive monitoring (arterial line, central venous catheter, often PA catheter or continuous cardiac output device), mediastinal and pleural chest tubes, temporary epicardial pacing wires (atrial and ventricular), a Foley, and infusions.
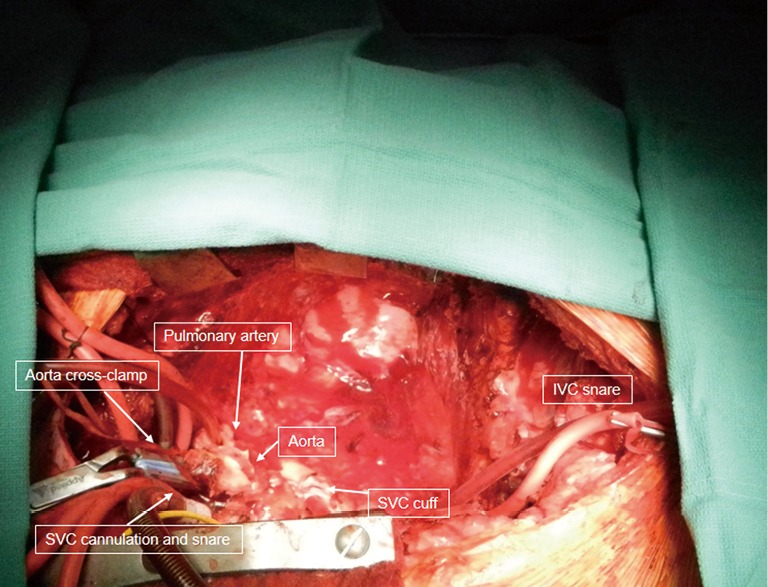
Scribes will document: hemodynamics (MAP, CVP, PA pressures, CI, SvO2), rhythm (sinus, paced, AF), chest tube output (report if > 200 mL/hr — concerning for surgical bleeding, may need re-exploration), rewarming, coagulation correction, extubation readiness (RSBI, ABG, mental status), and inotrope weaning.
Bleeding/tamponade: Chest tube output > 200 mL/hr, falling MAP, rising CVP with equalization, widened mediastinum, or sudden cessation of output (clot). Take-back to OR.
Post-op atrial fibrillation: Occurs in 20–40% of cardiac surgery patients on POD 2–4. Manage with amiodarone, beta blockade, anticoagulation if persistent.
Low cardiac output syndrome: CI < 2.0 L/min/m2; inotropes (epi, milrinone), IABP, or VA ECMO.
Acute kidney injury: Contrast, CPB time, hypotension.
Stroke: Atheroembolic, especially with porcelain aorta or epi-aortic scanning findings.
Sternal wound infection: Superficial (skin/subcutaneous) or deep (mediastinitis) — high mortality, requires debridement and often pectoralis or omental flap reconstruction.
Heparin-induced thrombocytopenia (HIT): Platelet drop > 50% POD 5–10; switch to argatroban or bivalirudin.
Early Post-Thoracic Surgery
After lung resection, patients have one or more chest tubes on water seal or suction monitored for air leak (persistent bubbling on cough/exhalation; quantified by digital drainage system or the bubble column). Prolonged air leak > 5 days is the most common complication of lobectomy. Other key complications: atrial fibrillation (post-thoracotomy), pneumonia, atelectasis (managed with incentive spirometry, bronchoscopy for mucus plugging), chylothorax (thoracic duct injury, milky output > 1000 mL/day that rises with enteral fat intake), bronchopleural fistula, pulmonary embolism, and recurrent laryngeal nerve injury (hoarseness, aspiration risk). Esophagectomy carries the additional risks of anastomotic leak (intrathoracic leaks are highly morbid), conduit ischemia, and delayed gastric emptying.
Always document the chest tube status in three parts: output volume (since last check), character (serous, serosanguinous, sanguinous, chylous, purulent), and air leak status (none, expiratory, continuous). These three elements drive the chest tube removal decision.
20 Imaging, Labs & Diagnostics
Cardiac Imaging & Studies
Transthoracic echocardiography (TTE): First-line for valve disease, EF, chamber sizes, pericardial effusion. Document EF, valve gradients and areas, chamber dimensions, RVSP (estimated PA pressure).
Transesophageal echo (TEE): Higher resolution for mitral valve, LA appendage thrombus, endocarditis vegetations, dissection. Routinely used intraoperatively for all valve and complex cardiac surgery.
Left heart catheterization: Coronary angiography grading stenoses and defining the roadmap for CABG. Document each vessel: LM, LAD (and diagonals), LCx (and OMs), RCA (and PDA, PLB), with percent stenosis.
Right heart catheterization: PA pressures, PCWP, CO/CI, PVR for HF/transplant/LVAD evaluation.
CT angiogram (gated): Coronary CTA, aortic CTA (dissection protocol includes arch through iliacs, with and without contrast).
Cardiac MRI: Myocardial viability, infiltrative disease, complex congenital assessment.
EKG: Rate, rhythm, axis, intervals (PR, QRS, QTc), ischemia, hypertrophy, bundle branch block.
Thoracic Imaging & Studies
CT chest (with contrast): Primary imaging for lung nodules, masses, mediastinal disease, staging. Document size, location by segment/lobe, relation to pleura and fissures, nodal stations.
PET/CT: SUV max for primary and any FDG-avid nodes or distant sites; essential for NSCLC staging.
Pulmonary function tests: Document FEV1 (absolute and percent predicted), DLCO, and predicted post-op FEV1 and DLCO (ppoFEV1, ppoDLCO). Values < 40% predicted raise major concerns for lobectomy tolerance.
Bronchoscopy / EBUS: Airway inspection and mediastinal nodal sampling.
Esophagogastroduodenoscopy (EGD): Diagnosis and biopsy of esophageal tumors, hernias, Barrett esophagus.
Manometry / pH study: Achalasia and GERD evaluation.
Barium esophagram: Anatomic survey for hernia, motility, and leak assessment.
Key Labs
Pre-op: CBC, BMP, coagulation (PT/INR, aPTT, fibrinogen), type and cross, troponin, BNP/NT-proBNP, lipid panel, HbA1c, LFTs, TSH. On CPB: ACT every 20–30 minutes (target > 400 sec). Post-op: serial CBC, BMP, coagulation, lactate, ABG, troponin trends, and platelet trend to detect HIT.
21 Medications You Must Know
Intraoperative & Early Post-Op
| Drug | Class / Use | Notes |
|---|---|---|
| Heparin (UFH) | Anticoagulation for CPB | Bolus 300–400 units/kg; target ACT > 400 sec |
| Protamine | Heparin reversal | 1 mg per 100 units heparin; risk of hypotension / anaphylaxis |
| Tranexamic acid (TXA) | Antifibrinolytic | Reduces bleeding in cardiac surgery |
| ε-Aminocaproic acid | Antifibrinolytic | Alternative to TXA |
| Aprotinin | Antifibrinolytic | Largely withdrawn due to renal/mortality safety concerns |
| Cardioplegia (del Nido, Buckberg) | Cold, high-K arrest solution | Delivered antegrade / retrograde to arrest heart |
Inotropes & Vasoactives
| Drug | Mechanism | Use |
|---|---|---|
| Epinephrine | α + β1 + β2 | Post-pump low output, arrest |
| Norepinephrine | α > β | First-line vasopressor for vasoplegia |
| Dobutamine | β1 > β2 | Inotropic support in HF |
| Milrinone | PDE-3 inhibitor (inodilator) | RV failure, pulmonary hypertension |
| Dopamine | Dose-dependent DA/β/α | Less commonly used today |
| Vasopressin | V1 receptor | Vasoplegia refractory to norepi |
| Phenylephrine | Pure α1 | SAM, hypotension with preserved CO |
| Nitroprusside | Arterial + venous dilator | Afterload reduction, dissection BP control |
| Nitroglycerin | Venodilator | Ischemia, preload reduction |
| Inhaled nitric oxide (iNO) | Pulmonary vasodilator | RV failure, post-transplant PH |
Antiarrhythmics & Cardiac Meds
| Drug | Class / Use |
|---|---|
| Amiodarone | Class III; first-line for post-op AF and VT |
| Lidocaine | Class IB; VT/VF |
| Metoprolol / esmolol | Beta blocker for rate control, HTN, aortic anti-impulse |
| Diltiazem | Non-DHP CCB for rate control |
| Lisinopril / losartan | ACEi/ARB for HF, HTN |
| Atorvastatin / rosuvastatin | High-intensity statin post-CABG |
| Aspirin | Antiplatelet — start POD 1 after CABG |
| Clopidogrel / ticagrelor | P2Y12 inhibitors; held pre-CABG (5 days / 3 days) |
| Warfarin | For mechanical valves and AF |
| DOACs (apixaban, rivaroxaban) | For nonvalvular AF; NOT for mechanical valves |
Mechanical AVR (bileaflet On-X or newer): INR 1.5–2.0 (with aspirin) for low-risk patients per FDA.
Mechanical AVR (standard bileaflet): INR 2.0–3.0.
Mechanical MVR: INR 2.5–3.5.
Any mechanical valve with AF, prior thromboembolism, or hypercoagulability: INR 2.5–3.5 with aspirin 75–100 mg.
Bioprosthetic valves: Aspirin alone long-term; warfarin 3–6 months post-op is reasonable.
Bridging: patients on warfarin for mechanical valves require bridging with IV heparin or therapeutic LMWH around invasive procedures; DOACs are simply held (typically 2–3 days pre-op based on renal function).
22 Classification Systems
Class I: No limitation of activity; ordinary activity does not cause symptoms.
Class II: Slight limitation; comfortable at rest, ordinary activity causes symptoms.
Class III: Marked limitation; less than ordinary activity causes symptoms.
Class IV: Unable to carry out any activity without symptoms; symptoms at rest.
Class I: Angina only with strenuous exertion.
Class II: Slight limitation with ordinary activity (e.g., walking > 2 blocks).
Class III: Marked limitation (walking 1–2 blocks).
Class IV: Angina with any activity or at rest.
The STS Adult Cardiac Surgery Risk Score estimates operative mortality and morbidity (stroke, renal failure, prolonged ventilation, deep sternal infection, reoperation, and composite) for CABG, valve, and combined procedures, stratified as low < 4%, intermediate 4–8%, and high > 8%. EuroSCORE II is the European equivalent; both are required for heart team decision-making between SAVR and TAVR.
ASA I: Normal healthy patient.
ASA II: Mild systemic disease (well-controlled HTN/DM).
ASA III: Severe systemic disease (limits activity).
ASA IV: Severe systemic disease that is a constant threat to life.
ASA V: Moribund patient not expected to survive without the operation.
ASA VI: Declared brain-dead, organs being harvested.
E suffix: Emergency.
Unintentional weight loss > 10 lb/year, exhaustion, weakness (grip strength), slow walking speed, and low physical activity. 0 criteria = robust, 1–2 = pre-frail, ≥ 3 = frail. Frailty independently predicts post-cardiac-surgery mortality and is required documentation for TAVR heart team evaluations.
Profile 1: Critical cardiogenic shock ("crash and burn").
Profile 2: Progressive decline on inotropes.
Profile 3: Stable but inotrope-dependent.
Profile 4: Resting symptoms, home on oral therapy.
Profile 5: Exertion intolerant.
Profile 6: Exertion limited.
Profile 7: Advanced NYHA III.
23 Physical Exam & Abbreviations Master List
The Cardiothoracic Physical Exam
General: [NAD / ill-appearing], [A&O x3]
Neck: JVP [cm above sternal angle]; carotid upstrokes [normal/delayed]; no bruits
Chest: clear to auscultation bilaterally; no dullness, no egophony
Cardiac: RRR / irregular; S1, S2 [normal/fixed split]; [grade __/VI systolic/diastolic murmur at __ radiating to __]; no S3/S4; no rubs
Abdomen: soft, nontender, no HSM
Extremities: no clubbing, cyanosis, or edema; pulses 2+ throughout
Skin: healed prior sternotomy / thoracotomy scars noted
Neuro: grossly intact, no focal deficits
Key murmur patterns: AS — crescendo-decrescendo systolic at RUSB radiating to carotids, with paradoxically split S2 in severe disease; AR — early diastolic decrescendo at LLSB, best with patient leaning forward in end expiration; MR — holosystolic at apex radiating to axilla; MS — opening snap followed by low-pitched diastolic rumble at apex in left lateral decubitus; TR — holosystolic at LLSB increasing with inspiration (Carvallo sign).
Abbreviations — Anatomy & Diagnoses
Abbreviations — Procedures
Abbreviations — Meds, Labs, Hemodynamics
24 Sample HPI Templates & References
These templates show the kind of documentation expected in a cardiothoracic clinic or consult. Use them as frameworks for the rhythm and content of a good CT HPI.
"Mr. [Name] is a 67-year-old male with hypertension, hyperlipidemia, type 2 diabetes (A1C 8.1), 35-pack-year smoking history (quit 2021), and newly diagnosed multivessel CAD referred for surgical revascularization. He presented 2 weeks ago to his PCP with CCS class III exertional angina after 1 block of walking. EKG showed lateral ST-T changes. Troponin was negative. Stress echo demonstrated a large area of inducible ischemia in the anterolateral and inferior walls. Cath revealed LM 60% stenosis, proximal LAD 85% stenosis, LCx 75% with OM1 90%, and dominant RCA with 90% mid-vessel stenosis. SYNTAX score 28. LVEF preserved at 55%. He denies rest pain, orthopnea, PND, or syncope. Current medications: aspirin 81, atorvastatin 80, metoprolol succinate 50, lisinopril 20, metformin 1000 BID. Plan: 4-vessel CABG (LIMA-LAD, SVG-diagonal, SVG-OM1, SVG-PDA) pending carotid duplex and dental clearance."
"Mrs. [Name] is a 78-year-old female with hypertension, CKD stage 3 (baseline Cr 1.5), and known bicuspid aortic valve referred for evaluation of severe symptomatic aortic stenosis. She reports progressive exertional dyspnea (NYHA class III), two episodes of presyncope while climbing stairs in the past month, and new atypical chest pressure with exertion. TTE: severely calcified bicuspid aortic valve with peak velocity 4.8 m/s, mean gradient 58 mmHg, AVA 0.7 cm2, preserved LVEF 60%, mild concentric LVH, mild-moderate MR, normal RV function, RVSP 38 mmHg. Cath: no significant CAD. STS risk for SAVR 3.2%. Frailty assessment: pre-frail (2/5). Heart team discussion planned for SAVR vs TAVR; anatomy on gated CT appears favorable for transfemoral TAVR."
"Mr. [Name] is a 54-year-old male with poorly controlled hypertension transferred from an outside ED with acute onset severe 'tearing' interscapular and anterior chest pain that began 3 hours ago while mowing the lawn. On arrival BP 210/115 right arm, 165/90 left arm, HR 98, diaphoretic. Neuro intact. Exam: new 2/6 early diastolic murmur at LLSB, no pericardial rub, symmetric peripheral pulses, bilateral femoral pulses palpable. CT aorta demonstrates a Stanford type A (DeBakey I) dissection with intimal flap from the sinotubular junction extending through the arch into the descending thoracic and abdominal aorta to the level of the left common iliac, with a small pericardial effusion, moderate aortic regurgitation, and patent visceral vessels off the true lumen. Started on esmolol and nicardipine targeting HR < 60 and SBP < 120. To OR emergently for ascending + hemiarch replacement; AVR vs valve-sparing root to be determined intraoperatively."
"Ms. [Name] is a 66-year-old female with 40-pack-year smoking history (quit 6 months ago), COPD (FEV1 68% predicted, DLCO 62% predicted), and newly diagnosed right upper lobe NSCLC referred for surgical resection. Nodule was incidentally detected on low-dose CT lung cancer screening: a 2.3 cm spiculated solid mass in the RUL posterior segment. PET/CT showed SUV max 9.2 in the primary, no FDG-avid mediastinal nodes or distant disease. Brain MRI negative. EBUS-TBNA of 4R, 7, and 11R stations all negative for malignancy. CT-guided biopsy confirmed invasive adenocarcinoma, PD-L1 40%, EGFR and ALK negative. Clinical stage cT1cN0M0, IA3. PPO FEV1 estimated at 54%, PPO DLCO 48%. Plan: right VATS RUL lobectomy with mediastinal lymph node dissection."
"Post-op day 2 status post right VATS RUL lobectomy for cT1cN0 adenocarcinoma. Patient feels well overnight, ambulating with PT, pain controlled on scheduled acetaminophen + oxycodone PRN. Incentive spirometer use 10×/hr. Apical chest tube: 120 mL serosanguinous over 24 hours, continuous small expiratory air leak (grade 2 on digital drainage system) unchanged from yesterday. Basilar chest tube: 80 mL serosanguinous, no air leak — candidate for removal today. Vitals stable, afebrile, O2 sat 94% on 2L NC. Chest X-ray: RUL post-op changes, no pneumothorax, small residual apical space, no effusion. Plan: pull basilar chest tube, continue apical tube on water seal, advance activity, home O2 assessment."
"Mr. [Name] is a 42-year-old male with no prior cardiopulmonary history referred for evaluation of an anterior mediastinal mass discovered on chest X-ray obtained for a persistent cough. CT chest: 5.8 × 4.2 cm well-circumscribed anterior mediastinal mass without local invasion, no pleural effusion, no pericardial effusion, no lymphadenopathy. Labs: AFP and beta-hCG negative (germ cell screen), TSH normal, AChR antibodies pending. No symptoms of myasthenia gravis (no ptosis, diplopia, fatigable weakness, dysphagia, or dyspnea). PFTs within normal limits. Differential: thymoma (most likely), teratoma, lymphoma, thyroid. Plan: robotic-assisted thymectomy with en bloc resection; frozen section to guide extent; pre-op MG evaluation with neurology given association."
References & Sources
Delgado V et al. 2023 ESC Guidelines for the Management of Endocarditis. Eur Heart J. 2023.
NCCN Clinical Practice Guidelines in Oncology — Non-Small Cell Lung Cancer.
Goldstraw P et al. The IASLC Lung Cancer Staging Project: 8th Edition TNM. J Thorac Oncol. 2016.
SYNTAX: Serruys PW et al. PCI vs CABG for Severe CAD. NEJM. 2009.
PARTNER 3: Mack MJ et al. TAVR vs SAVR in Low-Risk Patients. NEJM. 2019.
COAPT: Stone GW et al. Transcatheter Mitral-Valve Repair in Heart Failure. NEJM. 2018.
MOMENTUM 3: Mehra MR et al. A Fully Magnetically Levitated LVAD — Final Report. NEJM. 2019.
MGTX: Wolfe GI et al. Randomized Trial of Thymectomy in Myasthenia Gravis. NEJM. 2016.
Figure 1: Diagram of the Human Heart. Wikimedia Commons / Wapcaplet. CC BY-SA 3.0.
Figure 2: Coronary Vessels Anterior View. OpenStax. CC BY 3.0.
Figure 3: Conduction System of Heart. OpenStax. CC BY 3.0.
Figure 4: Major Respiratory Organs. OpenStax. CC BY 3.0.
Figure 5: Bronchial Tree. OpenStax. CC BY 3.0.
Figure 6: Aortic Dissection Classification. Wikimedia Commons. Public domain.
Cardiothoracic surgery is one of the steepest learning curves in medicine. The vocabulary is dense, the decisions are high-stakes, and the documentation touches every organ system. The best CT scribes learn the logic of the specialty: why LIMA-to-LAD is non-negotiable, why a type A dissection is to the OR within minutes, why the chest tube air leak matters more than the fluid output on POD 3, and why every valve patient needs a heart team discussion.
Pre-read every cath, every echo, every CT, and every PFT before the patient is seen. Know the surgeon's conduit preferences and dictation style. Memorize the medications by generic and brand name. Anticipate the plan from the diagnosis. Ask questions in the downtime between cases. Keep this guide open as a reference until the content is second nature.
Welcome to cardiothoracic surgery.