Colorectal Surgery
Every diagnosis, procedure, medication, classification, and documentation framework you need to succeed as a colorectal surgery scribe from day one.
All diagrams on this page are sourced from published educational or institutional materials (Wikimedia Commons, OpenStax, Blausen Medical, Gray's Anatomy) rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Colorectal Anatomy Essentials
Colorectal surgery encompasses the colon, rectum, anus, and pelvic floor, along with the small bowel when it overlaps with colorectal pathology. A scribe cannot chart effectively without a solid mental map of the large bowel, its vascular supply, and the anatomy of the anorectum. Every operative note, pathology report, and endoscopy document will reference these structures by exact anatomical name.
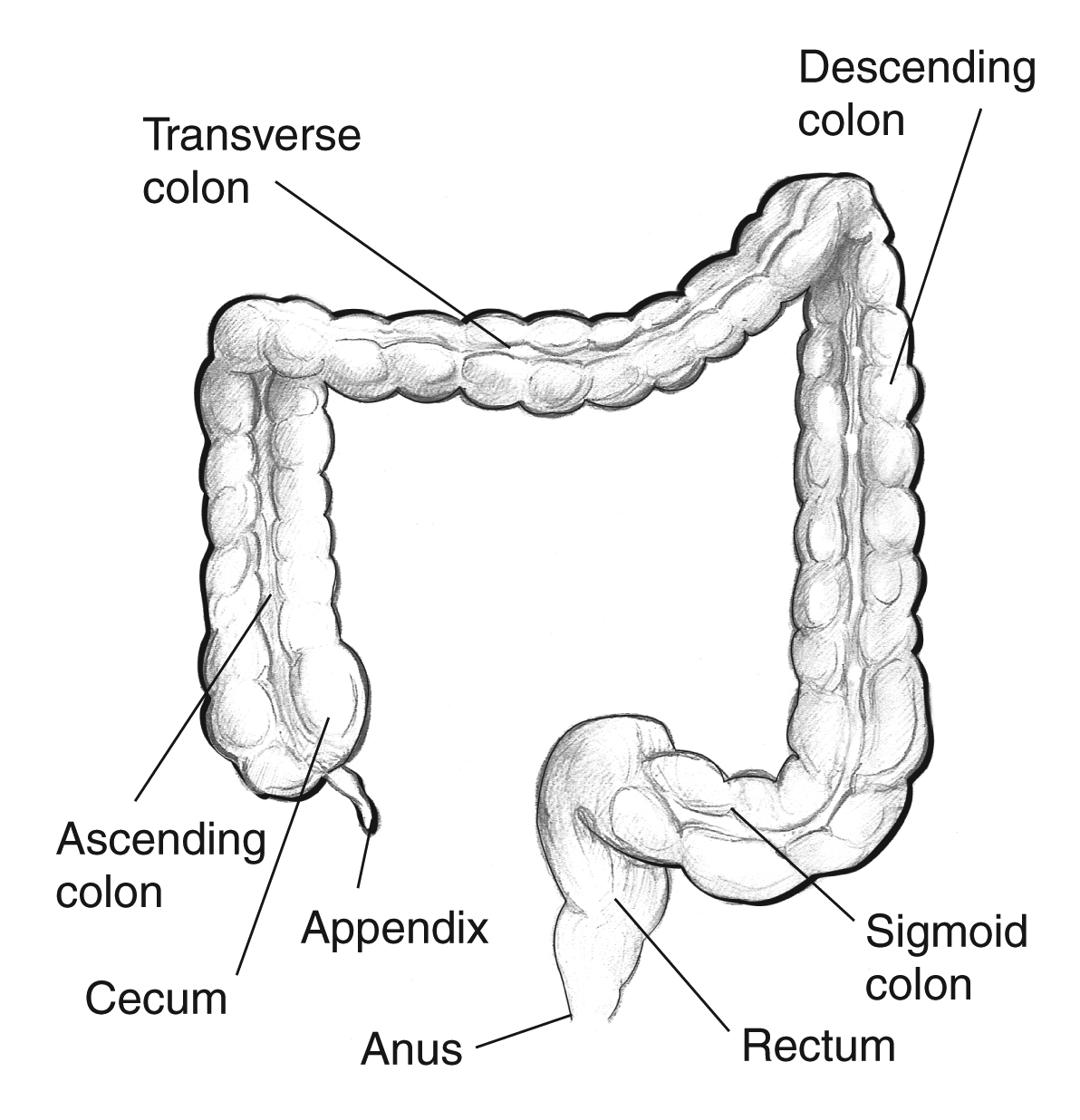
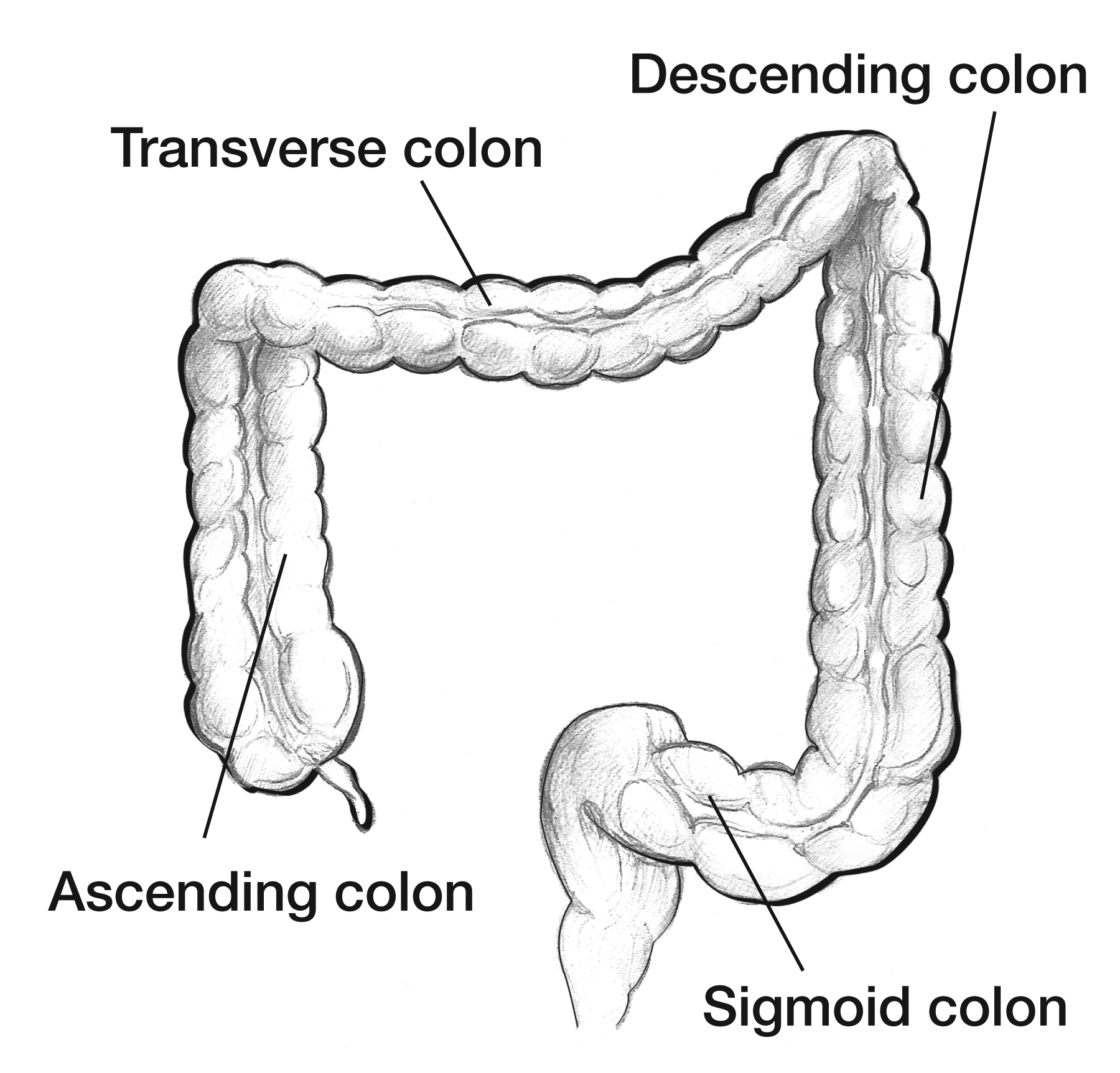
The Colon — Segments & Landmarks
The large intestine begins at the ileocecal valve and terminates at the anal verge. Moving distally, the segments are: cecum (with the appendix), ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon, and rectum. The rectum is approximately 12–15 cm long and is divided into upper, mid, and lower thirds. Below the rectum lies the anal canal (~4 cm), bounded superiorly by the dentate (pectinate) line — the critical anatomic and embryologic boundary between columnar rectal mucosa (visceral, insensate) and squamous anoderm (somatic, exquisitely sensitive).
Vascular Supply — Why Resections Are Named the Way They Are
The colon has a split blood supply that dictates every colectomy boundary. The superior mesenteric artery (SMA) supplies the right colon via the ileocolic, right colic, and middle colic branches — up to roughly the splenic flexure. The inferior mesenteric artery (IMA) supplies the left colon and rectum via the left colic, sigmoidal branches, and superior rectal artery. The rectum additionally receives middle rectal arteries (from the internal iliac) and inferior rectal arteries (from the internal pudendal). The marginal artery of Drummond runs along the mesenteric border and provides collateral flow between SMA and IMA territories. The watershed zones at the splenic flexure (Griffiths' point) and rectosigmoid junction (Sudeck's point) are vulnerable to ischemic colitis.
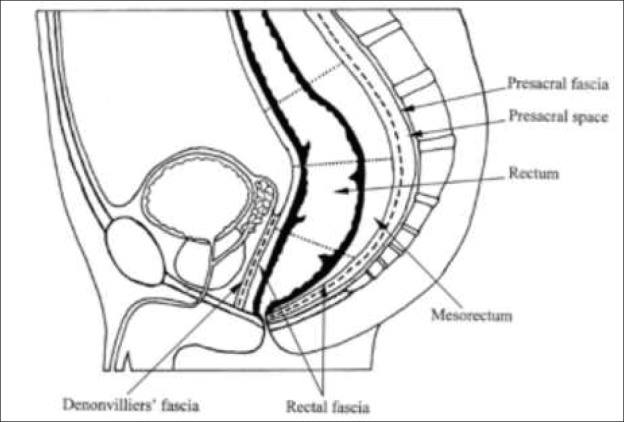
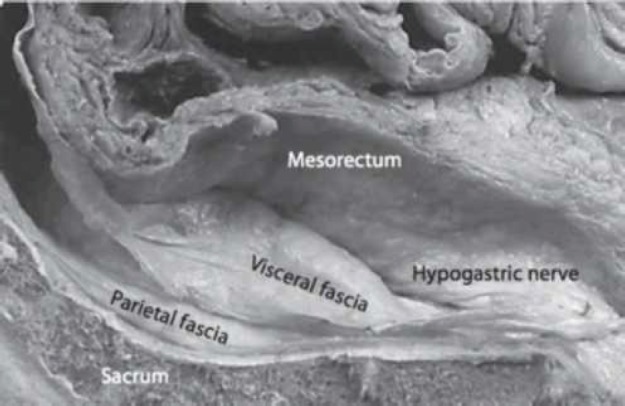
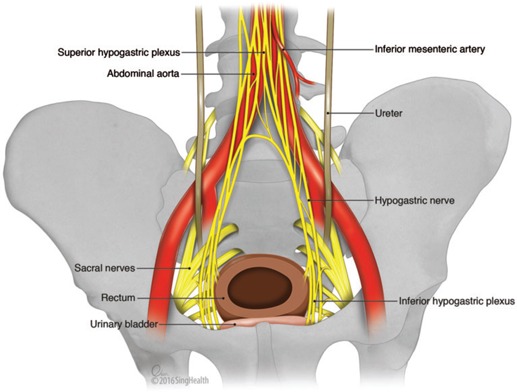
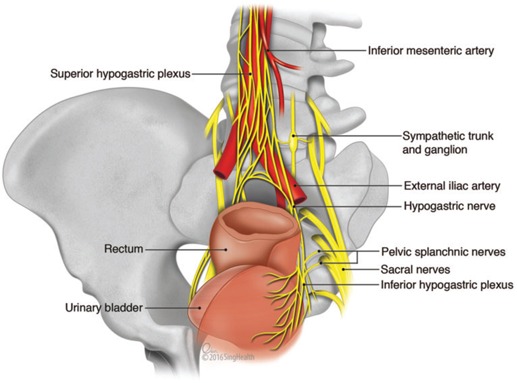
The Rectum, Mesorectum & Total Mesorectal Excision Plane
The rectum is surrounded by the mesorectum, a fatty envelope containing lymphatic drainage enclosed by the mesorectal fascia. Quality rectal cancer surgery requires total mesorectal excision (TME) — sharp dissection in the "holy plane" between mesorectal fascia and presacral (Waldeyer's) fascia. Key surrounding structures the surgeon will name in every rectal case: the presacral venous plexus, hypogastric nerves (sympathetic), nervi erigentes (parasympathetic, S2–S4), the peritoneal reflection (anterior), and for males the Denonvilliers' fascia separating the rectum from the prostate/seminal vesicles.
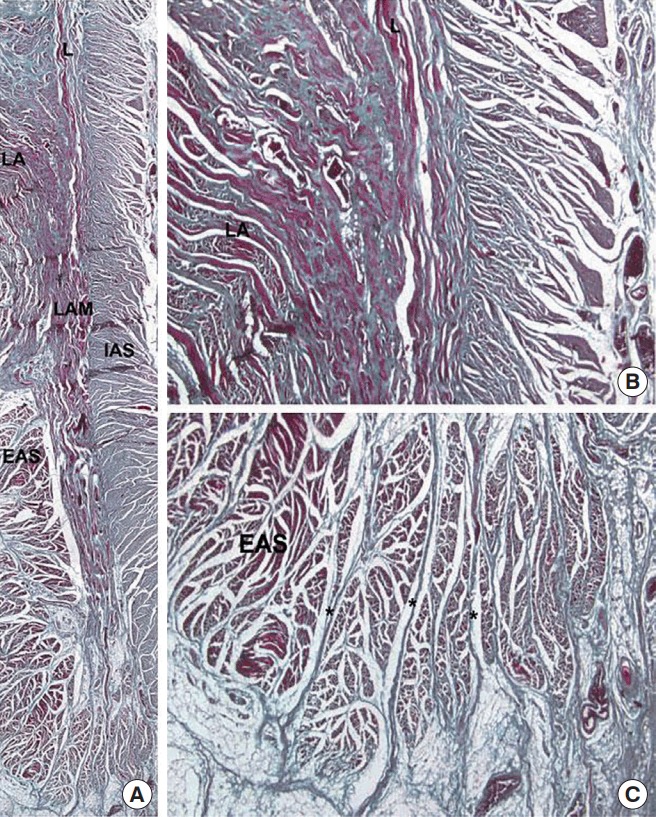
Anal Canal & Sphincter Complex
The anal sphincter has two components: the internal anal sphincter (IAS) — smooth muscle, involuntary, continuous with the rectal circular muscle — and the external anal sphincter (EAS) — skeletal muscle, voluntary, innervated by the inferior rectal branch of the pudendal nerve. Between them lies the intersphincteric plane. The puborectalis sling of the levator ani maintains the anorectal angle and is essential for continence. The anal columns of Morgagni terminate at the dentate line in anal crypts that drain the anal glands — the origin of cryptoglandular abscesses and fistulas.
Memorize the dentate line. It explains almost every anorectal exam finding you will chart. Above the dentate line: columnar epithelium, visceral innervation, internal hemorrhoids (painless bleeding), adenocarcinoma. Below: squamous epithelium, somatic innervation, external hemorrhoids and fissures (exquisitely painful), squamous cell carcinoma. The surgeon will constantly reference whether a lesion is "above" or "below" the dentate line.
02 Scribe Documentation Framework
Colorectal notes are structured around a few recurring encounter types: new consult for a colonoscopy finding (polyp, mass, bleeding), inflammatory bowel disease follow-up, diverticulitis presentation (clinic or ED), anorectal complaint, pre-op and post-op cancer clinic, and stoma clinic. Each has its own rhythm.
Chief Complaint: "Rectal bleeding," "abnormal colonoscopy," "anal pain," "follow-up colon cancer," "recurrent diverticulitis."
Bowel history is the HPI. You must capture: baseline bowel habits (frequency, consistency, Bristol stool score), any change in caliber, blood in/on stool vs mixed in, mucus, tenesmus, urgency, incomplete evacuation, nocturnal bowel movements (red flag in IBD), pain with defecation, pruritus ani, prolapse or bulge, incontinence (gas, liquid, solid), and current bowel regimen (fiber, laxatives, anti-diarrheals).
Screening history: Date and findings of most recent colonoscopy, prior polyps (number, size, histology), family history of colorectal cancer or polyps with age at diagnosis.
PMH/PSH: Prior abdominal surgery (adhesion risk), radiation, IBD history, diverticulitis episodes, ostomy status. Medications including anticoagulants, NSAIDs, opioids, immunosuppressants, biologics.
Abdomen: inspection (scars, distention, stoma), auscultation (bowel sounds), palpation (tenderness, masses, peritoneal signs, hernias), percussion (tympany vs dullness).
Anorectal exam: inspection (external hemorrhoids, skin tags, fissure, fistula opening, prolapse on Valsalva), digital rectal exam (tone, mass, tenderness, blood, prostate in males), anoscopy (internal hemorrhoids by quadrant and grade), rigid/flexible sigmoidoscopy findings when performed.
Stoma exam: location, type (end vs loop, ileostomy vs colostomy), stoma color (beefy red normal, dusky or black = ischemia), height above skin, peristomal skin, effluent character and volume, presence of parastomal hernia.
A/P is typically organized by problem: cancer staging and treatment plan, surveillance schedule, medical therapy (for IBD), procedural plan (OR date, bowel prep, consent), and return precautions. Always capture the staging (AJCC TNM), the MDT/tumor board recommendation if discussed, and which neoadjuvant/adjuvant therapy is anticipated.
The most frequently missed HPI element in colorectal clinic is quantifying bleeding. "Rectal bleeding" is not enough. Chart: bright red vs dark/maroon, on the paper vs in the bowl vs coating stool vs mixed in, teaspoon vs tablespoon vs "filled the bowl," frequency, and associated symptoms. This distinction drives the entire differential (hemorrhoid vs distal cancer vs proximal source).
03 Colorectal Cancer Neoplastic
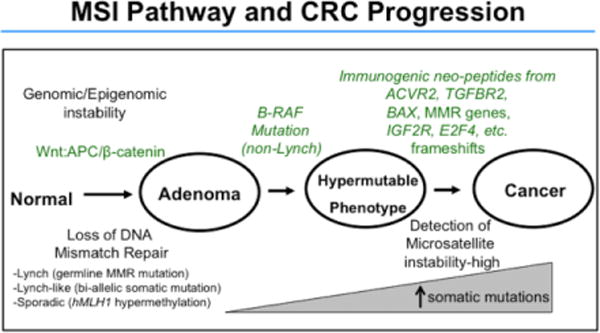
Colorectal cancer (CRC) is the third most common cancer in both sexes in the United States and the second leading cause of cancer death overall. It is the defining diagnosis of colorectal surgery clinic. CRC arises almost exclusively from the adenoma–carcinoma sequence or the serrated pathway, and is the reason screening colonoscopy exists.
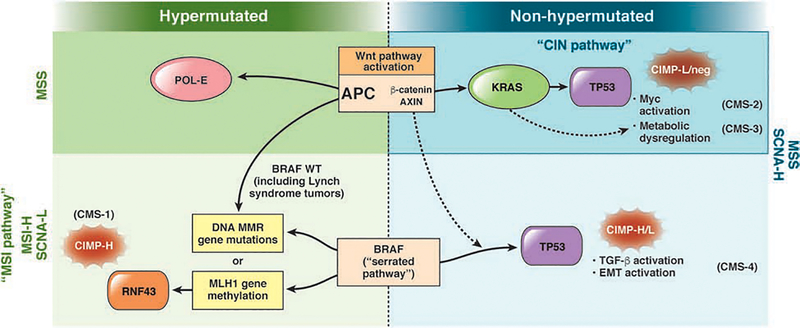
Pathophysiology & Location-Based Biology
Right-sided (cecum, ascending, proximal transverse) tumors tend to be larger, fungating, and present with iron-deficiency anemia and occult blood loss rather than obstruction — the bowel lumen is wider and stool more liquid. They are more often microsatellite-instability-high (MSI-H) and are linked to Lynch syndrome and the serrated pathway. Left-sided tumors (splenic flexure, descending, sigmoid) are more often annular "apple-core" lesions producing change in stool caliber, constipation, hematochezia, and large bowel obstruction. Rectal cancers present with hematochezia, tenesmus, urgency, and change in bowel habits and are staged and managed distinctly from colon cancer because of pelvic anatomy and the value of neoadjuvant therapy.
Presentation & Red Flags
Hematochezia or melena, unintentional weight loss, iron-deficiency anemia in a male or postmenopausal female, change in bowel habit > 6 weeks, tenesmus, pencil-thin stools, obstructive symptoms (distention, vomiting, obstipation), palpable abdominal or rectal mass, family history of CRC before age 50.
Workup
Every new CRC patient gets: full colonoscopy with biopsy (to confirm histology and screen for synchronous lesions — 2–5% incidence), CT chest/abdomen/pelvis with contrast for staging, CEA (carcinoembryonic antigen, baseline tumor marker), and a CBC, CMP, LFTs. For rectal cancer, additionally: pelvic MRI with rectal protocol to define T stage, mesorectal fascia involvement, and extramural vascular invasion, and endorectal ultrasound (ERUS) for early T stage assessment. Molecular testing includes MMR/MSI (mismatch repair, universal per NCCN), KRAS/NRAS/BRAF, and HER2 for metastatic disease.
AJCC 8th Edition TNM Staging
| Stage | TNM | Clinical Correlation |
|---|---|---|
| 0 | Tis N0 M0 | Carcinoma in situ (intramucosal); endoscopic excision often curative |
| I | T1–T2 N0 M0 | Invades submucosa (T1) or muscularis propria (T2); surgery alone usually curative |
| IIA | T3 N0 M0 | Through muscularis into pericolorectal tissues |
| IIB | T4a N0 M0 | Penetrates visceral peritoneum |
| IIC | T4b N0 M0 | Invades adjacent organ/structure |
| IIIA | T1–T2 N1/N1c or T1 N2a | Node positive, limited T |
| IIIB | T3–T4a N1; T2–T3 N2a; T1–T2 N2b | Node positive, advanced T or higher N |
| IIIC | T4a N2a; T3–T4a N2b; T4b N1–N2 | Heavy nodal burden or T4b with nodes |
| IVA | Any T Any N M1a | Distant mets to 1 site/organ (no peritoneum) |
| IVB | Any T Any N M1b | Mets to > 1 site/organ (no peritoneum) |
| IVC | Any T Any N M1c | Peritoneal metastases ± other sites |
Management Overview
Colon cancer management is primarily surgical for stages I–III, with adjuvant chemotherapy (FOLFOX or CAPOX) added for stage III and high-risk stage II disease, per the NCCN Colon Cancer Guidelines. Resection is tailored to the tumor location: right hemicolectomy for cecum through proximal transverse, extended right for distal transverse/splenic flexure, left hemicolectomy for descending, sigmoidectomy for sigmoid. Oncologic principles include high vascular ligation, adequate proximal/distal margins (5 cm typical), and harvest of at least 12 lymph nodes.
Rectal cancer management is more complex. Locally advanced rectal cancer (T3/T4 or node positive) is increasingly managed with total neoadjuvant therapy (TNT) — full-course chemotherapy plus chemoradiation before surgery — which improves pathologic complete response rates and enables watch-and-wait strategies in select patients (NCCN Rectal Cancer Guidelines). Surgical options include low anterior resection (LAR) for tumors that allow a sphincter-sparing anastomosis, and abdominoperineal resection (APR) with permanent colostomy for very low tumors that cannot obtain a negative distal margin. Transanal excision or TAMIS/TEM may be appropriate for carefully selected early T1 tumors.
Screening
USPSTF 2021 CRC screening recommends beginning average-risk screening at age 45 (lowered from 50) and continuing to age 75. Acceptable modalities include colonoscopy every 10 years, stool-based testing (FIT annual, FIT-DNA every 3 years), or flexible sigmoidoscopy. The ACG guideline explicitly endorses 45 as the start age. High-risk patients (family history, polyposis, IBD) start earlier and screen more frequently.
When charting a new cancer consult, always capture: tumor location measured from the anal verge in cm (for rectal), percent circumference, biopsy histology, clinical T/N/M from MRI/CT, CEA value, MMR status, and whether the tumor board has reviewed the case. Missing any of these will require the attending to edit the note.
04 Polyps & Hereditary Polyposis Syndromes Neoplastic
Most colorectal cancers begin as polyps, so polyp management and surveillance are central to colorectal practice. Polyps are classified histologically and by morphology (Paris classification: pedunculated, sessile, flat, depressed).
Polyp Histology
| Type | Malignant Potential | Notes |
|---|---|---|
| Hyperplastic | Low (except large proximal) | Usually distal, small; no surveillance change |
| Tubular adenoma | Low to moderate | Most common adenoma |
| Tubulovillous adenoma | Moderate | Mixed architecture |
| Villous adenoma | High | Often large, sessile; highest risk |
| Sessile serrated lesion (SSL) | Moderate to high | Serrated pathway; proximal colon; CIMP/BRAF |
| Traditional serrated adenoma | High | Rare, distal |
Hereditary Polyposis Syndromes
Autosomal dominant, APC gene mutation on 5q. Hundreds to thousands of adenomatous polyps begin in adolescence; CRC is essentially inevitable by age 40 untreated. Extraintestinal manifestations: desmoid tumors, duodenal/ampullary adenomas (second leading cause of death after CRC), osteomas, epidermoid cysts, congenital hypertrophy of the retinal pigment epithelium (CHRPE). Gardner syndrome = FAP plus osteomas and soft tissue tumors. Turcot syndrome = FAP (or Lynch) plus CNS tumors. Treatment: prophylactic total proctocolectomy with IPAA (ileal pouch–anal anastomosis) or total colectomy with IRA (ileorectal anastomosis), typically in late teens/early twenties.
Autosomal dominant germline mutation in mismatch repair genes (MLH1, MSH2, MSH6, PMS2, EPCAM). Most common hereditary CRC syndrome (~3% of all CRC). Tumors are right-sided, poorly differentiated, mucinous, and MSI-high. Associated cancers: endometrial (highest after CRC), ovarian, gastric, small bowel, urothelial, pancreas, brain (Turcot), sebaceous (Muir-Torre). Diagnosis triggered by universal MMR/MSI testing on all CRC specimens. Surveillance colonoscopy every 1–2 years starting age 20–25.
MUTYH-associated polyposis (MAP): autosomal recessive, biallelic MUTYH mutation; attenuated adenomatous polyposis phenotype.
Peutz-Jeghers syndrome (PJS): STK11/LKB1 mutation; hamartomatous polyps throughout GI tract, mucocutaneous pigmentation (lips, buccal mucosa), elevated risk of GI and breast/pancreatic/gynecologic cancers.
Juvenile polyposis syndrome (JPS): BMPR1A or SMAD4 mutation; multiple juvenile (hamartomatous) polyps, elevated CRC and gastric cancer risk.
Cowden syndrome: PTEN mutation; hamartomas, breast, thyroid, endometrial cancer risk.
05 Inflammatory Bowel Disease (Surgical) Inflammatory
Ulcerative colitis (UC) and Crohn disease are the two major inflammatory bowel diseases. Colorectal surgeons see them when medical therapy fails, when dysplasia or cancer is found, or when complications develop (stricture, fistula, perforation, toxic megacolon).
Ulcerative Colitis
UC is a continuous mucosal inflammation that starts at the rectum and extends proximally. It never skips, never reaches beyond the colon (ileal "backwash" aside), and is limited to the mucosa/submucosa. Extent is classified as proctitis, left-sided colitis (to splenic flexure), or extensive/pancolitis. Symptoms: bloody diarrhea, tenesmus, urgency, lower abdominal cramping, nocturnal bowel movements. Extraintestinal: primary sclerosing cholangitis (PSC), uveitis, erythema nodosum, pyoderma gangrenosum, arthritis. Surgical indications: medically refractory disease, dysplasia or cancer on surveillance, toxic megacolon, massive hemorrhage. The definitive operation is total proctocolectomy with ileal pouch-anal anastomosis (IPAA, J-pouch), typically in 2 or 3 stages. See the ASCRS UC Clinical Practice Guideline.
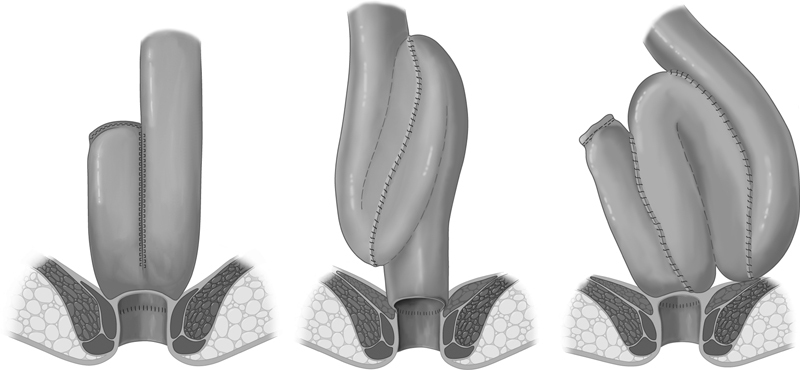
Crohn Disease
Crohn is a transmural, patchy, skip-lesion inflammation that can affect any site from mouth to anus. Ileocolic involvement is most common. The three behavioral phenotypes (Montreal classification) are inflammatory (B1), stricturing (B2), and penetrating/fistulizing (B3). Symptoms depend on location: RLQ pain and diarrhea with ileocolic disease, perianal fistulas and abscesses with colonic/perianal disease, obstructive symptoms with stricturing phenotype. Surgery is not curative — disease recurs, typically at the anastomosis — so the principle is bowel preservation: resect as little as possible, use strictureplasty for short strictures, and reserve resection for failure. Common operations: ileocolic resection for terminal ileal disease, strictureplasty, diverting loop ileostomy for severe perianal Crohn. See the ASCRS Crohn Disease Guideline.
Transverse colon diameter > 6 cm with systemic toxicity (fever, tachycardia, leukocytosis, anemia, hypotension). Occurs in severe UC or C. difficile colitis. Management: aggressive medical resuscitation, IV steroids, broad antibiotics, decompression. Failure to improve in 24–72 hours mandates total abdominal colectomy with end ileostomy and Hartmann closure of the rectal stump. The rectum is preserved for later completion proctectomy/IPAA.
06 Diverticular Disease Inflammatory
Diverticula are mucosal outpouchings through the colonic wall at points of weakness where the vasa recta penetrate. They are overwhelmingly sigmoid in Western populations (high-pressure segment, narrow caliber, low-fiber diets). Diverticulosis is the presence of diverticula; diverticulitis is inflammation/infection, typically from micro-perforation of a diverticulum. Diverticular bleeding is painless hematochezia from erosion of the vasa recta at the neck of a diverticulum.
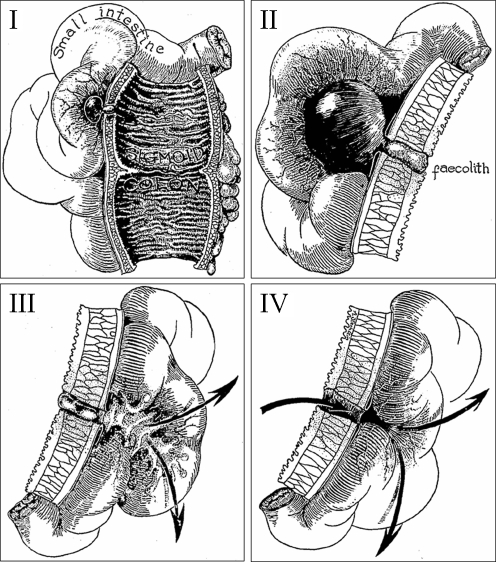
Uncomplicated vs Complicated Diverticulitis
Uncomplicated diverticulitis = localized inflammation without abscess, perforation, fistula, obstruction, or stricture. Complicated diverticulitis = any of the above. The historical teaching that all diverticulitis requires antibiotics has evolved; mild uncomplicated diverticulitis in selected patients can be managed without antibiotics per the ASCRS Diverticulitis Guideline.
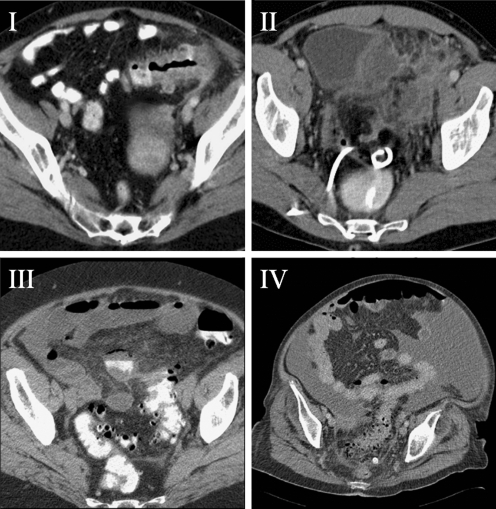
Hinchey Classification of Perforated Diverticulitis
| Stage | Finding | Management |
|---|---|---|
| I | Pericolic or mesenteric abscess (confined) | IV antibiotics ± percutaneous drainage |
| II | Walled-off pelvic abscess | Percutaneous drainage + antibiotics; elective resection consideration |
| III | Purulent peritonitis | Emergent surgery: Hartmann or resection with primary anastomosis ± diversion |
| IV | Feculent peritonitis | Emergent Hartmann procedure |
Recurrent Diverticulitis & Elective Resection
The old rule of "resect after two attacks" has been abandoned. Current guidance individualizes the decision based on severity, symptoms between episodes, immunocompromise, fistula, stricture, and patient preference. Complicated diverticulitis (perforation, abscess requiring drainage, fistula, obstruction) and chronic smoldering symptoms are relative indications for elective sigmoidectomy. Fistulas complicating diverticulitis include colovesical (most common, presents with pneumaturia/fecaluria/UTI), colovaginal, coloenteric, and colocutaneous.
07 Volvulus & Large Bowel Obstruction Mechanical
Volvulus is rotation of a bowel loop around its mesenteric axis, producing obstruction and potentially ischemia. Sigmoid volvulus (most common, 60–75%) affects elderly, debilitated, institutionalized patients with chronic constipation. Classic imaging: "coffee bean" sign on plain film, "bird's beak" on contrast enema, whirl sign on CT. First-line management is endoscopic detorsion (flexible sigmoidoscopy with decompression tube), followed by elective sigmoidectomy during the same admission because recurrence without resection is ~50%. Emergent sigmoidectomy (Hartmann) is required if there is peritonitis, suspected ischemia, or failure of endoscopic detorsion.
Cecal volvulus (~25%) occurs in younger patients with congenitally mobile cecum. Imaging: "comma" or displaced cecum in LUQ. Endoscopic detorsion is generally unsuccessful; treatment is right hemicolectomy (or cecopexy in very high-risk patients). Cecal bascule is anterior folding of the cecum without axial rotation.
Large Bowel Obstruction (LBO)
Causes: cancer (most common in adults), diverticular stricture, volvulus, Ogilvie syndrome (acute colonic pseudo-obstruction, non-mechanical). LBO with a competent ileocecal valve produces a closed-loop obstruction — the cecum dilates progressively and may perforate at > 12 cm diameter (Laplace's law). Workup: CT with contrast to identify transition point. Management depends on cause: emergent decompression (stent or resection) for obstructing cancer, Hartmann or primary resection for malignant LBO. Ogilvie syndrome is treated with IV neostigmine after mechanical obstruction is excluded.
08 Small Bowel Obstruction & Ileus Mechanical
Though technically general surgery territory, colorectal surgeons manage SBO in their post-op patients constantly. Causes of SBO in a colorectal patient: adhesions (far and away most common after any abdominal surgery), internal hernia, parastomal hernia, incisional hernia, anastomotic stricture, recurrent cancer, Crohn stricture, gallstone ileus, intussusception.
Presentation: crampy abdominal pain, distension, nausea/vomiting (feculent with distal obstruction), obstipation. CT findings: dilated small bowel loops (> 3 cm), transition point, collapsed distal bowel, pneumatosis or portal venous gas (ischemia). Management of uncomplicated partial SBO: NPO, NG decompression, IV fluids, serial exams. Water-soluble contrast challenge (Gastrografin) has both diagnostic and therapeutic value. Surgery is indicated for complete obstruction, closed-loop, ischemia, failure to resolve in 3–5 days, or peritonitis.
Post-op ileus is functional (non-mechanical) dysmotility after abdominal surgery. Expected up to 3–5 days. Prolonged ileus is defined as > 5 days. Management: NG decompression if vomiting, correct electrolytes (especially potassium and magnesium), limit opioids, ambulate. ERAS protocols reduce ileus duration significantly.
09 Hemorrhoidal Disease Anorectal
Hemorrhoids are normal vascular cushions in the submucosa of the anal canal; they become symptomatic when engorged, prolapsed, or thrombosed. Internal hemorrhoids arise above the dentate line, are covered by columnar mucosa, and are insensate — they bleed painlessly. External hemorrhoids arise below the dentate line, are covered by anoderm, and are exquisitely sensitive — they hurt when thrombosed. Classic positions: right anterior, right posterior, left lateral (3, 7, 11 o'clock in prone jackknife).
Goligher Grading of Internal Hemorrhoids
| Grade | Finding | Typical Management |
|---|---|---|
| I | Bleed but do not prolapse | Fiber, water, sitz baths, topical agents |
| II | Prolapse with straining, reduce spontaneously | Rubber band ligation, sclerotherapy, infrared coagulation |
| III | Prolapse requiring manual reduction | Banding (if feasible), stapled hemorrhoidopexy, or excisional hemorrhoidectomy |
| IV | Irreducibly prolapsed | Excisional hemorrhoidectomy (Ferguson or Milligan-Morgan) |
Management follows a stepwise escalation per the ASCRS Hemorrhoid Guideline. First-line is medical: fiber (25–35 g/day), increased water, sitz baths, topical hydrocortisone or lidocaine, avoidance of prolonged toilet sitting. Office procedures (band ligation, sclerotherapy, IRC) are highly effective for grades I–II. Grade III–IV and mixed internal-external disease usually need operative hemorrhoidectomy.
Thrombosed external hemorrhoid is an acute painful purple perianal lump from clotted external hemorrhoid. If within 48–72 hours of onset, excision under local anesthesia provides rapid relief. After 72 hours the pain is typically receding and conservative management suffices.
10 Anal Fissure Anorectal
An anal fissure is a linear tear in the anoderm distal to the dentate line. Posterior midline (90%) is classic; anterior midline is more common in women post-delivery. Fissures off the midline should raise suspicion for Crohn, HIV, tuberculosis, syphilis, or malignancy. Presentation: sharp pain with defecation ("passing glass"), small amount of bright red blood on paper, anal spasm, sometimes a sentinel tag. Pathophysiology: hypertonic internal anal sphincter produces ischemia of the posterior commissure, preventing healing.
Management per the ASCRS Anal Fissure Guideline: first-line is fiber/water/sitz baths plus topical chemical sphincterotomy — topical nitroglycerin 0.4% ointment BID or topical nifedipine/diltiazem — which relaxes the IAS and restores blood flow. Botulinum toxin injection is a second-line option. Lateral internal sphincterotomy (LIS) is the surgical gold standard for chronic refractory fissures; incontinence risk must be discussed, particularly in women with prior obstetric injury.
11 Perianal Abscess & Anal Fistula Anorectal
The cryptoglandular theory: infection of an anal gland at the dentate line spreads through the intersphincteric plane to form an abscess, and when the abscess drains (spontaneously or surgically), a chronic epithelialized tract can remain as a fistula-in-ano.
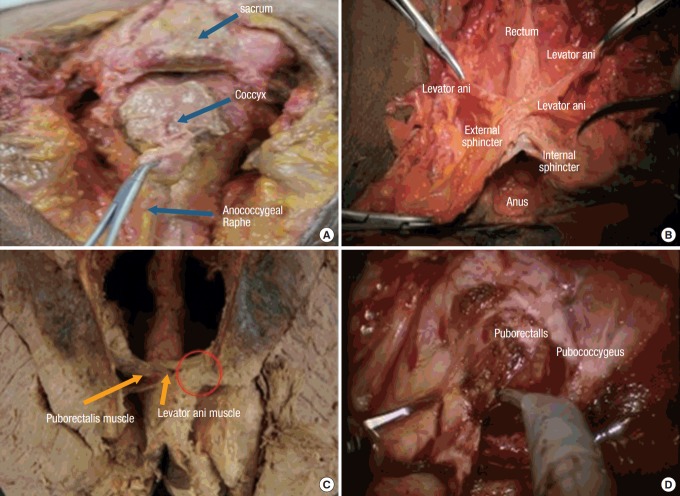
Abscess Classification by Location
Perianal (most common, superficial): drains at the anal verge. Ischiorectal: lateral, may be bilateral (horseshoe). Intersphincteric: between IAS and EAS, painful but no external swelling. Supralevator: above the levators, deep, may be from pelvic source. Management: incision and drainage, generally with a cruciate incision as close to the anal verge as possible to minimize future fistula tract length. Antibiotics alone are inadequate; drainage is required.
Park's Classification of Anal Fistulas
| Type | Tract Course | Notes |
|---|---|---|
| Intersphincteric | Through IAS only, tracks in intersphincteric plane to skin | Most common (~70%) |
| Transsphincteric | Through both IAS and EAS into ischiorectal fossa | ~25% |
| Suprasphincteric | Through IAS, up over puborectalis, down through ischiorectal fossa | Rare |
| Extrasphincteric | From perineal skin through levators into rectum, bypassing sphincter complex | Rare; usually iatrogenic, Crohn, or pelvic source |
Goodsall's rule predicts the internal opening: external openings anterior to a transverse line through the anus tract radially to the nearest anal crypt; posterior openings curve to the posterior midline.
Fistula Management
Treatment depends on tract depth and sphincter involvement per the ASCRS Anorectal Abscess/Fistula Guideline. Simple low fistulas (minimal sphincter involvement) can be treated with fistulotomy. Complex fistulas (high transsphincteric, anterior in women, Crohn, multiple tracts, recurrent) require sphincter-preserving options: draining seton (silastic loop to maintain drainage), cutting seton, LIFT (ligation of intersphincteric fistula tract), endorectal advancement flap, fibrin glue, or fistula plug. Rectovaginal fistulas are particularly challenging; options include advancement flap, bioprosthetic interposition, or diverting stoma for Crohn-related disease.
12 Pilonidal Disease Anorectal
Pilonidal disease is a chronic inflammatory condition of the natal cleft, caused by hair penetration and foreign-body reaction. Common in young males with thick body hair, obesity, prolonged sitting. Presents as an acute midline abscess or chronic draining sinus with lateral openings. Treatment: acute abscess requires off-midline I&D. Chronic disease is treated with pit-picking (Bascom), excision with primary closure, excision with healing by secondary intention, or flap procedures (Karydakis, Limberg rhomboid, cleft lift) for recurrent disease. See the ASCRS Pilonidal Guideline.
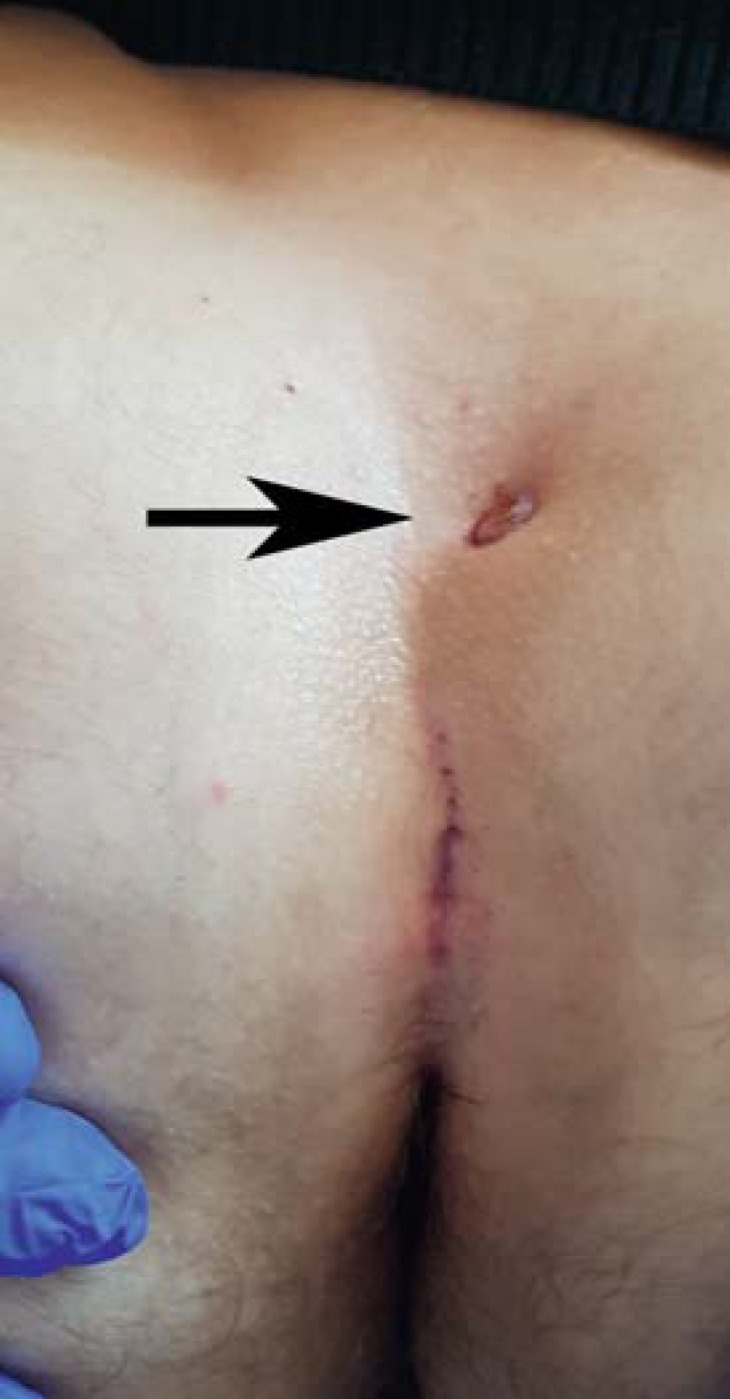
13 Anal Cancer & HPV-Related Disease Neoplastic
Anal cancer is overwhelmingly squamous cell carcinoma (SCC) of the anal canal, strongly linked to HPV (types 16, 18), HIV, immunosuppression, receptive anal intercourse, and smoking. Adenocarcinoma of the anal canal is rare and usually arises from anal glands or ducts; it behaves like rectal cancer.
Presentation: rectal bleeding, anal pain, palpable mass, pruritus, tenesmus, inguinal lymphadenopathy. Exam under anesthesia with biopsy confirms diagnosis. Staging: MRI pelvis, CT chest/abdomen, PET/CT, HIV testing.
Treatment is non-surgical: the Nigro protocol — concurrent chemoradiation with 5-FU + mitomycin C — is curative for most SCC of the anal canal. Surgery (APR) is reserved for persistent/recurrent disease (salvage) per the NCCN Anal Cancer Guidelines. Surveillance is clinical (DRE, anoscopy, inguinal palpation) with imaging for recurrence.
Anal intraepithelial neoplasia (AIN) is the precursor lesion (low-grade LSIL, high-grade HSIL). High-resolution anoscopy (HRA) with targeted ablation is used for surveillance, particularly in HIV+ men who have sex with men. Condyloma acuminatum (anogenital warts) is treated with topical agents (imiquimod, podophyllin, TCA), excision, or electrocautery. Buschke-Löwenstein tumor is a giant condyloma / verrucous carcinoma variant that behaves locally aggressively and requires wide excision.
Pruritus ani is chronic perianal itching. Primary (idiopathic) forms are most common; secondary causes include fecal soilage, hemorrhoids, fissure, dietary triggers (coffee, spicy food, chocolate, citrus), fungal infection, psoriasis, lichen sclerosus, and rarely neoplasia. Management: eliminate irritants, gentle cleansing (avoid scrubbing and perfumed wipes), dry the perianal skin, mild topical hydrocortisone for short courses, and a dietary trial. Biopsy any suspicious lesion.
14 Rectal Prolapse, Rectocele & Pelvic Floor Pelvic Floor
Full-thickness rectal prolapse (procidentia) is protrusion of all layers of the rectal wall through the anus. Classic in elderly multiparous women. Symptoms: visible prolapse with Valsalva, mucus, bleeding, incontinence, constipation. Differentiate from mucosal prolapse (only mucosa) and internal intussusception (not visible externally; diagnosed on defecography).
Surgical approaches per the ASCRS Rectal Prolapse Guideline:
- Abdominal (preferred for good surgical candidates): ventral rectopexy (laparoscopic or robotic), suture rectopexy, resection rectopexy (Frykman-Goldberg).
- Perineal (for frail/elderly): Altemeier perineal proctosigmoidectomy, Delorme mucosal sleeve resection.
Rectocele is herniation of the anterior rectal wall into the vagina, presenting with obstructed defecation and need for vaginal splinting. Enterocele is small bowel herniation into the rectovaginal septum. Sigmoidocele is redundant sigmoid in the pelvis. Workup includes defecography and anorectal manometry. Management is first conservative (fiber, biofeedback) and escalates to transvaginal or transanal repair.
Anismus (paradoxical puborectalis contraction) is a pelvic floor dyssynergia where the puborectalis fails to relax with straining. Diagnosed on defecography and manometry. Treatment is biofeedback.
Solitary rectal ulcer syndrome (SRUS) is associated with chronic straining and internal intussusception. Presents with bleeding, mucus, tenesmus, and characteristic anterior rectal ulceration. Treatment focuses on the underlying defecation disorder (biofeedback, fiber); surgery is reserved for refractory cases.
Retrorectal (presacral) masses are uncommon lesions occupying the space between the rectum and sacrum. The broad differential includes congenital (tailgut cyst, epidermoid cyst, teratoma, anterior meningocele), neurogenic (schwannoma, neurofibroma), osseous (chordoma, sacral tumors), and inflammatory lesions. MRI pelvis is the imaging of choice. Most benign lesions require excision to rule out malignancy and prevent complications.
15 Fecal Incontinence & Constipation Functional
Fecal incontinence is involuntary passage of gas, liquid, or solid stool. Causes: obstetric sphincter injury (most common in women), prior anorectal surgery, neurologic disease, diabetes, diarrhea, rectal prolapse, radiation. Workup includes anorectal manometry, endoanal ultrasound (sphincter integrity), pudendal nerve terminal motor latency, defecography. Severity is quantified with the Wexner (Cleveland Clinic) Incontinence Score (0 = continent, 20 = complete incontinence, scoring frequency of incontinence to gas/liquid/solid, pad use, lifestyle alteration).
Management ladder: dietary modification and bulking agents, loperamide, biofeedback, sacral nerve stimulation (SNS) (first-line for refractory incontinence), injectable anal bulking agents, sphincteroplasty (overlapping), and diverting colostomy for severe refractory disease.
Chronic constipation evaluated by a colorectal surgeon is typically either slow-transit constipation or obstructed defecation. Workup: colonoscopy (rule out mass/stricture), colonic transit study (Sitz marker), anorectal manometry, defecography. The Wexner Constipation Score quantifies severity. Slow-transit refractory to medical therapy may be treated with total abdominal colectomy with ileorectal anastomosis; outcomes are variable and patient selection critical.
16 Ischemic & Infectious Colitis Inflammatory
Ischemic colitis most commonly affects watershed zones (splenic flexure, rectosigmoid). Non-occlusive in elderly with atherosclerosis, hypotension, cardiac surgery, vasopressors, cocaine, hypercoagulable states. Presents with sudden cramping lower abdominal pain, bloody diarrhea. CT shows segmental wall thickening. Colonoscopy shows patchy erythema, hemorrhagic mucosa, single-stripe sign. Most cases resolve with bowel rest, IV fluids, and antibiotics; gangrenous or full-thickness ischemia requires resection.
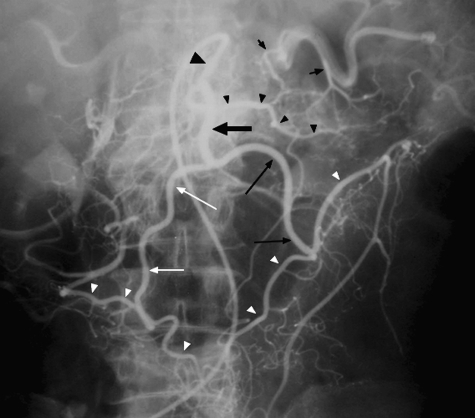
Pseudomembranous (C. difficile) colitis — the surgical urgency is fulminant C. difficile with megacolon, shock, lactic acidosis, or perforation. Surgical management is total abdominal colectomy with end ileostomy, or the diverting loop ileostomy with antegrade vancomycin lavage (Neal procedure) as a colon-sparing alternative. Medical management is PO vancomycin and IV metronidazole; fidaxomicin is increasingly first-line for recurrent disease.
Stercoral colitis/perforation is pressure necrosis from an impacted fecal bolus, classically at the antimesenteric border of the rectosigmoid. Elderly, constipated, bedbound patients. Emergent resection is required.
17 Ostomy Management & Complications Post-Op
Stomas are a central feature of colorectal surgery. A scribe must be able to describe a stoma accurately on exam.
Stoma Types
| Type | Configuration | Typical Output | Common Indication |
|---|---|---|---|
| End colostomy | Single barrel, usually LLQ | Formed stool | Hartmann, APR |
| Loop colostomy | Two limbs, transverse or sigmoid | Semi-formed | Temporary diversion |
| End ileostomy | Single barrel, RLQ, spouted (Brooke) | Liquid, 500–1200 mL/day | Total colectomy, IPAA |
| Loop ileostomy | Two limbs, RLQ | Liquid | Protection of distal anastomosis (LAR, IPAA) |
Stoma Siting & Care
Ideal stoma siting is performed pre-operatively by an ostomy nurse: within the rectus muscle, away from scars, skin folds, the beltline, and bony prominences, visible to the patient seated and standing. Poor siting is the single biggest driver of peristomal skin complications and appliance failure. A well-sited Brooke ileostomy should protrude 2–3 cm above the skin to allow effluent to drop cleanly into the bag. Colostomies can be flush or slightly raised.
Routine stoma care includes appliance changes every 3–5 days, skin barrier products for denuded peristomal skin, convexity appliances for retracted or flush stomas, and stoma paste or rings to seal irregular contours. Patients should be counseled to recognize warning signs: dusky color, bleeding beyond minor irritation, significant output changes, parastomal pain, or inability to pass flatus or stool.
Ostomy Complications
Ischemia/necrosis (dusky or black stoma), retraction (stoma pulled below skin), mucocutaneous separation, high output (> 1500–2000 mL/day, dehydration risk in ileostomy), peristomal skin breakdown, obstruction.
Parastomal hernia (very common, up to 50% at 5 years), prolapse, stenosis, skin irritation from effluent, psychosocial issues, dehydration/electrolyte imbalance in high-output ileostomy.
High-output ileostomy is a common post-op ED visit. Management: loperamide 4 mg QID, diphenoxylate/atropine, oral rehydration solutions, limit hypotonic fluids (water) which worsen sodium losses, codeine or tincture of opium for severe cases, PPI to reduce gastric secretions. Hospitalize for AKI or severe electrolyte derangement.
18 Anastomotic Leak & Post-Op Complications Post-Op
Anastomotic leak is the most feared complication of colorectal surgery. Incidence: 3–6% for colonic anastomosis, up to 15% for low pelvic (LAR) anastomosis. Risk factors: male sex, low anastomosis, neoadjuvant radiation, smoking, malnutrition, steroids, poor perfusion, tension. Classic presentation: post-op day 5–7 fever, tachycardia, leukocytosis, peritonitis, ileus, feculent drain output, or pelvic sepsis.
Workup: CT with rectal contrast (for low anastomoses) or oral/IV contrast. Management depends on severity: small contained leak → antibiotics + percutaneous drainage; large or uncontained leak → reoperation with takedown and diversion (Hartmann for left-sided, ileostomy + washout for protected anastomoses). ICG fluorescence angiography intraoperatively has been shown to reduce leak rates by confirming anastomotic perfusion.
Clavien-Dindo Classification of Surgical Complications
| Grade | Definition |
|---|---|
| I | Any deviation from normal course without need for pharmacologic (except antiemetics, analgesics, antipyretics, diuretics, electrolytes), surgical, endoscopic, or radiologic intervention |
| II | Requires pharmacologic treatment (including blood transfusion, TPN) |
| IIIa | Requires surgical, endoscopic, or radiologic intervention, not under general anesthesia |
| IIIb | Requires intervention under general anesthesia |
| IVa | Life-threatening, single-organ dysfunction requiring ICU |
| IVb | Life-threatening, multi-organ dysfunction |
| V | Death of the patient |
Other post-op complications to document: surgical site infection (superficial, deep, organ-space), ileus, SBO, urinary retention (especially after pelvic dissection from autonomic nerve injury), sexual dysfunction (anterograde ejaculation loss, erectile dysfunction), anastomotic stricture, incisional hernia, deep venous thrombosis/pulmonary embolism.
Low Anterior Resection Syndrome (LARS)
After sphincter-preserving rectal surgery, a significant proportion of patients develop LARS — a constellation of urgency, frequency, clustering, fragmentation, and incontinence related to loss of rectal reservoir function. Severity is graded by the LARS Score (0–42; 0–20 no LARS, 21–29 minor, 30–42 major). Management is multimodal: bulking agents, loperamide, biofeedback, pelvic floor physical therapy, sacral nerve stimulation, and dietary counseling. Always ask about and chart LARS symptoms on every post-LAR follow-up.
Pouchitis
Inflammation of the ileal pouch after IPAA occurs in up to 50% of UC patients over their lifetime. Presentation: increased stool frequency, urgency, cramping, bleeding, fever. Diagnosis by pouchoscopy with biopsy. First-line treatment is a two-week course of ciprofloxacin or metronidazole; chronic pouchitis may require budesonide, vedolizumab, or ustekinumab.
19 Colorectal Procedures — A to Z Operative
A colorectal scribe must recognize every procedure name. Operations can be performed open, laparoscopic, or robotic; robotic is increasingly favored for rectal cancer due to superior pelvic visualization. ICG fluorescence is used for perfusion assessment and sentinel node mapping.
Colonic Resections
| Procedure | Extent of Resection | Indication |
|---|---|---|
| Right hemicolectomy | Terminal ileum, cecum, ascending, hepatic flexure, proximal transverse | Right-sided cancer, cecal volvulus, Crohn ileocolic disease |
| Extended right hemicolectomy | Right plus transverse colon to splenic flexure | Hepatic flexure or transverse colon cancer |
| Transverse colectomy | Transverse colon only | Limited transverse lesion (uncommon) |
| Left hemicolectomy | Distal transverse, splenic flexure, descending, proximal sigmoid | Left-sided cancer |
| Sigmoidectomy | Sigmoid colon | Sigmoid cancer, diverticulitis, sigmoid volvulus |
| Subtotal colectomy | Ileum to sigmoid/rectum (preserves rectum) | Synchronous cancers, fulminant C. diff, toxic megacolon |
| Total abdominal colectomy | Ileum to rectum, end ileostomy | Fulminant colitis, HNPCC prophylaxis, slow transit constipation |
| Total proctocolectomy | Entire colon and rectum | UC, FAP; done with IPAA or end ileostomy |
Rectal Operations
| Procedure | Description | Indication |
|---|---|---|
| Low anterior resection (LAR) | Sigmoid + rectum resection with TME + colorectal or coloanal anastomosis ± diverting loop ileostomy | Mid and upper rectal cancer |
| Abdominoperineal resection (APR) | Rectum + anus resection with permanent end colostomy | Low rectal cancer without sphincter preservation option, recurrent anal cancer |
| Proctocolectomy + IPAA (J-pouch) | Total proctocolectomy with ileal pouch-anal anastomosis | UC, FAP |
| Hartmann procedure | Sigmoid/rectal resection with end colostomy and closed rectal stump (no anastomosis) | Emergent sigmoid resection (perforation, feculent peritonitis, unstable patient) |
| Transanal excision | Local excision of distal rectal lesion through the anus | T1 low rectal tumor, large villous adenoma |
| TAMIS / TEM | Transanal minimally invasive surgery / transanal endoscopic microsurgery — single-port platform for precise excision of mid/upper rectal lesions | Select early rectal tumors, large adenomas |
| Rectopexy | Mobilization and fixation of the rectum to the sacrum (ventral mesh, suture, resection rectopexy) | Rectal prolapse |
| Altemeier | Perineal proctosigmoidectomy | Rectal prolapse in frail/elderly |
| Delorme | Mucosal sleeve resection with muscular plication | Short-segment rectal prolapse |
Anorectal Procedures
| Procedure | Indication |
|---|---|
| Rubber band ligation | Internal hemorrhoids grade I–II |
| Sclerotherapy / infrared coagulation | Small internal hemorrhoids |
| Excisional hemorrhoidectomy (Ferguson closed, Milligan-Morgan open) | Grade III–IV, mixed, failed office treatment |
| Stapled hemorrhoidopexy (PPH) | Circumferential prolapsing hemorrhoids |
| Lateral internal sphincterotomy | Chronic anal fissure |
| Fistulotomy | Simple low fistula |
| Seton placement (draining or cutting) | Complex fistula, Crohn |
| LIFT procedure | Transsphincteric fistula |
| Endorectal advancement flap | Complex high fistula, rectovaginal |
| Pilonidal excision ± flap (Bascom, Karydakis, Limberg, cleft lift) | Chronic pilonidal disease |
| EUA + biopsy | Diagnostic, mass evaluation |
ERAS — Enhanced Recovery After Surgery
ERAS protocols have transformed colorectal recovery. Key elements per the ERAS Society Colorectal Guideline:
- Pre-op: patient education, no prolonged fasting (clear liquids up to 2 h pre-op), carbohydrate loading, mechanical bowel prep + oral antibiotics for colorectal resections
- Intra-op: multimodal analgesia (avoid opioids), regional anesthesia (TAP block, epidural), goal-directed fluids, normothermia, minimally invasive approach
- Post-op: early PO intake, early ambulation (POD 0), early Foley removal, multimodal pain control (acetaminophen, NSAIDs, gabapentin), alvimopan for accelerated ROBF, DVT prophylaxis, early drain removal
20 Imaging, Endoscopy & Labs Diagnostics
Endoscopy
Colonoscopy is the workhorse — diagnostic and therapeutic. Quality metrics: adequate prep (Boston Bowel Prep Scale ≥ 6, no segment < 2), cecal intubation rate > 95%, adenoma detection rate > 25% in screening. Document findings by location (cm from anal verge or anatomic segment), size, morphology (Paris classification), and histology. Flexible sigmoidoscopy reaches the splenic flexure and is useful for distal lesions, rectal cancer follow-up, and volvulus detortion. Anoscopy and rigid sigmoidoscopy are office procedures for anorectal complaints.
Imaging
| Modality | Use |
|---|---|
| CT abdomen/pelvis with IV contrast | Diverticulitis, obstruction, abscess, staging |
| CT with oral/rectal contrast | Anastomotic leak evaluation |
| CT colonography (virtual) | Screening alternative, incomplete colonoscopy |
| MRI pelvis (rectal protocol) | Rectal cancer T-staging, mesorectal fascia, EMVI |
| Endorectal ultrasound (ERUS) | Early rectal cancer T staging |
| Endoanal ultrasound | Sphincter integrity (incontinence), fistula mapping |
| Defecography (fluoroscopic or MR) | Pelvic floor, rectocele, intussusception, anismus |
| PET/CT | Recurrence evaluation, anal cancer staging, indeterminate lesions |
| Contrast enema | Volvulus, anastomotic integrity, stricture |
Labs
CEA baseline and surveillance in CRC (rising CEA suggests recurrence). CBC for anemia (right colon cancer), infection. CMP for electrolytes (high-output stoma), renal function, LFTs (metastasis). Lactate for suspected ischemia. Fecal calprotectin for IBD activity. C. difficile toxin/PCR for diarrhea. Stool studies for infectious etiologies. Type and screen/cross for major operations.
Surveillance After Curative CRC Resection
Per NCCN: history/physical + CEA every 3–6 months for 2 years then every 6 months to year 5; CT chest/abdomen/pelvis annually for 5 years (more frequent for high-risk disease); surveillance colonoscopy at 1 year post-op (or 3–6 months if not cleared pre-op), then 3 years, then every 5 years. Rising CEA prompts CT and, if negative, PET/CT.
Pathology Reports — What to Chart
Every colorectal pathology report contains specific elements the scribe should capture: tumor location and size, histologic type and grade, depth of invasion (T stage), lymphovascular invasion (LVI), perineural invasion (PNI), tumor budding, margin status (proximal, distal, radial/circumferential for rectum), number of lymph nodes examined and positive, MMR/MSI status, KRAS/NRAS/BRAF, and final pathologic TNM stage (pTNM; ypTNM if after neoadjuvant therapy). A circumferential margin < 1 mm is considered positive for rectal cancer and carries significant local recurrence risk.
21 Medications You Must Know Pharmacology
Bowel Preparation
| Agent | Type | Notes |
|---|---|---|
| Polyethylene glycol (GoLYTELY, MiraLAX) | Osmotic, isotonic | Large volume (4 L) split dose |
| Suprep (sodium sulfate) | Hypertonic saline | Low volume alternative |
| Magnesium citrate | Saline laxative | Adjunct; avoid in renal failure |
| Oral neomycin + metronidazole (or erythromycin) | Oral antibiotic bowel prep | Combined with mechanical prep reduces SSI |
Perioperative Antibiotics
Pre-op IV: cefoxitin, cefotetan, or ertapenem; alternative ampicillin-sulbactam or ciprofloxacin + metronidazole for beta-lactam allergy. Within 60 minutes of incision.
Post-op Pain & ERAS Adjuncts
Multimodal: scheduled acetaminophen, ketorolac/ibuprofen, gabapentin, IV/oral opioids for breakthrough, TAP block or epidural. Alvimopan (Entereg) is a peripheral mu-opioid antagonist used to accelerate GI recovery after bowel resection.
VTE Prophylaxis
Enoxaparin 40 mg SC daily (or BID in obese/high-risk), or unfractionated heparin 5000 U SC TID. Mechanical prophylaxis (SCDs) in all. Extended 28-day VTE prophylaxis for cancer surgery.
IBD Medications (Overview for Surgical Context)
| Class | Examples | Notes |
|---|---|---|
| 5-ASA | Mesalamine, sulfasalazine | First-line mild-moderate UC |
| Corticosteroids | Prednisone, IV methylprednisolone, budesonide | Induction; not maintenance; wound healing concern |
| Immunomodulators | Azathioprine, 6-MP, methotrexate | Maintenance |
| Anti-TNF biologics | Infliximab, adalimumab, certolizumab, golimumab | Must hold before surgery per protocol |
| Anti-integrin | Vedolizumab | Gut-selective |
| Anti-IL-12/23 | Ustekinumab | Crohn, UC |
| JAK inhibitors | Tofacitinib, upadacitinib | Moderate-severe UC |
Anorectal Topicals
Nitroglycerin 0.4% ointment (Rectiv) — anal fissure. Nifedipine 0.2–0.3% cream, diltiazem 2% gel — anal fissure alternatives with fewer headaches. Lidocaine 2–5% for symptomatic relief. Hydrocortisone (Anusol-HC, Proctofoam-HC) for hemorrhoids and pruritus ani. Sitz baths (warm water 10–15 minutes TID) are the cornerstone of anorectal comfort.
Bowel Regimen Medications
Fiber (psyllium, methylcellulose), docusate, polyethylene glycol (MiraLAX), bisacodyl, senna, lactulose, lubiprostone, linaclotide. Antidiarrheals: loperamide, diphenoxylate/atropine, codeine, tincture of opium. Antispasmodics: dicyclomine, hyoscyamine.
Chemotherapy Regimens (Know the Names)
| Regimen | Components | Use |
|---|---|---|
| FOLFOX | 5-FU + leucovorin + oxaliplatin | Adjuvant stage III colon, metastatic CRC |
| CAPOX (XELOX) | Capecitabine + oxaliplatin | Adjuvant, oral alternative to FOLFOX |
| FOLFIRI | 5-FU + leucovorin + irinotecan | Metastatic CRC (second-line or first-line) |
| FOLFOXIRI | 5-FU + leucovorin + oxaliplatin + irinotecan | Aggressive metastatic CRC in fit patients |
| Nigro protocol | 5-FU + mitomycin C + radiation | Anal SCC (definitive chemoradiation) |
Targeted agents: bevacizumab (anti-VEGF), cetuximab/panitumumab (anti-EGFR, for RAS wild-type left-sided disease), pembrolizumab (anti-PD-1, for MSI-H/dMMR tumors), encorafenib + cetuximab (BRAF V600E mutant), trastuzumab + pertuzumab/tucatinib (HER2 amplified).
22 Classification Systems Reference
Scores 0–30 summing eight items: frequency of BMs, difficult/painful evacuation, incomplete evacuation, abdominal pain, minutes per attempt, assistance required (laxatives/digital/enema), unsuccessful attempts per 24 h, duration of constipation. Higher = worse. Threshold > 15 typically considered significant.
Scores 0–20, five items (incontinence to solid, liquid, gas; pad use; lifestyle alteration) each rated 0 (never) to 4 (daily). 0 = perfect continence, 20 = complete incontinence.
Type 1 (separate hard lumps), Type 2 (lumpy sausage), Type 3 (cracked sausage), Type 4 (smooth sausage — ideal), Type 5 (soft blobs), Type 6 (mushy), Type 7 (watery). Scribe note: always chart Bristol type when available.
Hinchey (Section 06), Goligher (Section 09), Park's (Section 11), AJCC TNM (Section 03), and Clavien-Dindo (Section 18) are fully enumerated in their respective sections.
23 Physical Exam — Anorectal & Abdominal Exam
General: NAD, well-appearing
Abdomen: soft, non-distended, non-tender, no masses, no hernia, normal BS; [scar description]; [stoma description: site, type, color, output]
Anorectal:
Inspection: no external hemorrhoids, skin tags, fissures, fistula openings, or prolapse
DRE: normal tone, no mass, no tenderness, no blood on glove
Anoscopy: internal hemorrhoids grade [I/II/III/IV] at [position]; no masses; dentate line intact
Positioning: left lateral decubitus (Sims) or prone jackknife (best for anoscopy and office procedures). Always announce the exam in the note: "Anorectal exam performed in the prone jackknife position." Stoma exam detail: site, type, mucosa color (beefy red vs dusky vs black), height above skin, peristomal skin, presence of parastomal bulge with cough/Valsalva, effluent character and volume over 24 h.
A complete DRE always documents: sphincter tone at rest and with squeeze, presence of mass, tenderness, blood on glove, prostate in men, and any palpable fixed pelvic pathology. In rectal cancer follow-up, note the exact distance from the anal verge to any palpable anastomotic ridge or lesion.
24 Abbreviations, HPIs & References Reference
Abbreviations Master List
Sample HPI Templates
"Mr. [Name] is a 62-year-old male with HTN, hyperlipidemia, and 20-pack-year smoking history who presents for evaluation of biopsy-proven sigmoid adenocarcinoma. He initially presented to his PCP with 3 months of change in bowel habits (increasing constipation alternating with loose stools), intermittent bright red blood mixed in stool, and 8-pound unintentional weight loss. Screening colonoscopy (deferred since age 55) was performed and demonstrated a near-obstructing mass at 25 cm from the anal verge; biopsy returned moderately differentiated adenocarcinoma. Staging CT chest/abdomen/pelvis shows a sigmoid mass without evidence of distant metastases or significant lymphadenopathy. CEA 4.2. MMR proficient. Family history is notable for a paternal uncle with colon cancer at age 68. Patient is otherwise functional at baseline. Plan: laparoscopic sigmoidectomy with primary anastomosis following tumor board review."
"Ms. [Name] is a 58-year-old female with a history of two prior episodes of uncomplicated sigmoid diverticulitis who presents to the ED with 4 days of progressively worsening LLQ pain, subjective fevers, nausea, and decreased appetite. Exam notable for temperature 38.6 °C, HR 108, LLQ tenderness with localized guarding. WBC 17.4. CT with IV contrast demonstrates sigmoid diverticulitis with a 5.2 cm pelvic abscess (Hinchey II) without free perforation or pneumoperitoneum. Patient is hemodynamically stable. Plan: admit, IV ceftriaxone + metronidazole, IR-guided percutaneous drain placement, bowel rest; elective sigmoidectomy will be discussed at follow-up given complicated presentation in a third episode."
"Ms. [Name] is a 34-year-old female G2P2 (vaginal deliveries 2018, 2021) presenting with 6 weeks of severe pain with bowel movements and bright red blood on toilet paper. Pain is sharp, 'like passing glass,' lasting 30–60 minutes after defecation. She has been fearful of bowel movements and has developed worsening constipation. Denies prior episodes, no fevers, no systemic symptoms. No prior anorectal surgery. On exam in prone jackknife, inspection reveals a posterior midline anal fissure with a small sentinel tag; DRE was deferred due to pain. Plan: fiber 25 g/day, MiraLAX, sitz baths, topical nifedipine 0.2% cream BID × 6 weeks; return for consideration of botulinum toxin or LIS if refractory."
"Mr. [Name] is a 28-year-old male with pancolonic ulcerative colitis diagnosed in 2019, currently on infliximab and oral prednisone 20 mg daily, who presents for discussion of definitive surgical management. He has had three hospitalizations in the past year for severe flares requiring IV steroids, and his most recent colonoscopy demonstrated active pancolitis with low-grade dysplasia in a random biopsy from the transverse colon. He is steroid-dependent and has declined further escalation of biologic therapy. Nocturnal BMs 4–6 per night, bloody, urgency. Weight stable. Family history negative. Plan: three-stage total proctocolectomy with IPAA; first stage (total abdominal colectomy with end ileostomy) scheduled after steroid taper and nutrition optimization."
"Mrs. [Name] is an 82-year-old multiparous female with a 2-year history of progressive rectal prolapse, now requiring manual reduction several times per day. She reports mucus discharge, occasional bleeding, and worsening fecal incontinence to liquid stool and gas (Wexner score 12). She has tried a high-fiber diet without benefit. Past medical history includes well-controlled HTN and osteoarthritis; she lives independently with a walker. On exam, full-thickness rectal prolapse is demonstrated with Valsalva in the left lateral position, reducing approximately 6 cm of rectum with concentric rings (distinguishing from mucosal prolapse). Anal tone is diminished. Defecography and manometry have been ordered. Plan: discuss abdominal ventral rectopexy vs Altemeier perineal proctosigmoidectomy; patient leans toward perineal approach given age."
"Mr. [Name] is a 67-year-old male POD 6 from robotic low anterior resection with diverting loop ileostomy for a T3N1 mid-rectal adenocarcinoma (s/p neoadjuvant chemoradiation), who developed new fevers to 38.9 °C, tachycardia to 115, and increasing pelvic pain overnight. Exam notable for lower abdominal tenderness and cloudy effluent from the pelvic drain. Labs: WBC 18.2, lactate 2.4, CRP 22. CT with rectal contrast demonstrates extravasation at the colorectal anastomosis with a 4 cm presacral collection. Plan: NPO, IV piperacillin-tazobactam, IR drainage of collection; return to OR for washout and anastomosis assessment if hemodynamic compromise or failure to improve."
References & Sources
Clinical Practice Guidelines
NCCN Clinical Practice Guidelines in Oncology: Colon Cancer. National Comprehensive Cancer Network.
NCCN Clinical Practice Guidelines in Oncology: Rectal Cancer. National Comprehensive Cancer Network.
NCCN Clinical Practice Guidelines in Oncology: Anal Carcinoma. National Comprehensive Cancer Network.
USPSTF Recommendation: Screening for Colorectal Cancer. JAMA. 2021.
ACG Clinical Guidelines: Colorectal Cancer Screening 2021. American Journal of Gastroenterology.
ASCRS Clinical Practice Guidelines for the Management of Hemorrhoids. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guideline for the Management of Anal Fissures. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guidelines for the Management of Anorectal Abscess, Fistula-in-Ano, and Rectovaginal Fistula. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guidelines for the Treatment of Left-Sided Colonic Diverticulitis. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guidelines for the Surgical Management of Ulcerative Colitis. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guidelines for the Surgical Management of Crohn's Disease. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guidelines for the Treatment of Rectal Prolapse. Diseases of the Colon & Rectum.
ASCRS Clinical Practice Guidelines for the Management of Pilonidal Disease. Diseases of the Colon & Rectum.
Diagram & Figure Sources
Figure 1: Anatomy of the Large Intestine. OpenStax College. CC BY 3.0.
Figure 2: Large Intestine Anatomy. Blausen Medical. CC BY 3.0.
Figure 3: Anal Anatomy. Wikimedia Commons. Public domain.
Figure 4: Diverticulitis. Wikimedia Commons. Public domain.
Figure 5: Internal and External Hemorrhoids. Wikimedia Commons. Public domain.
Colorectal surgery is the most documentation-heavy of the general surgery subspecialties. You will chart cancer staging, polyp surveillance schedules, complex anorectal exams, stoma descriptions, and lengthy operative reports. The best colorectal scribes develop an instinct for the details that matter: exact distance from the anal verge, the dentate line relationship, MMR/MSI status, the TNM breakdown, and the Hinchey stage of every diverticulitis case.
Know the operations cold. When the surgeon says "LAR with diverting loop ileostomy" versus "APR with permanent end colostomy," you should immediately know the implications for the patient's long-term quality of life and follow-up. When you hear "Hinchey III," you should already be thinking Hartmann or primary anastomosis with washout. Pattern recognition is what makes scribes fast and accurate.
Welcome to colorectal surgery. The patients are often complex, the documentation is detailed, and the surgery is among the most elegant in medicine. Learn the anatomy, memorize the classifications, and you will thrive.