Endocrinology
Every diagnosis, hormonal axis, dynamic test, medication, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 The Endocrine System — Anatomy & Axes
Endocrinology is the study of hormones — chemical messengers secreted by glands into the bloodstream that regulate metabolism, growth, reproduction, stress response, fluid balance, and mineral homeostasis. Unlike most specialties, endocrinology is organized around feedback axes rather than a single organ. A scribe who understands the axes can predict lab ordering, anticipate the diagnosis, and document dynamic testing without constantly asking for clarification.
The Major Endocrine Glands
The hypothalamus sits at the base of the brain and is the master controller. It releases releasing hormones into the pituitary portal system that act on the anterior pituitary. The pituitary gland sits in the sella turcica at the skull base and has two lobes: the anterior pituitary (adenohypophysis) secretes six classical hormones — TSH, ACTH, LH, FSH, GH, and prolactin — while the posterior pituitary (neurohypophysis) stores and releases ADH (vasopressin) and oxytocin produced by hypothalamic neurons.
The thyroid gland is a butterfly-shaped gland in the anterior neck that produces T4 (thyroxine) and T3 (triiodothyronine) under TSH control. Parafollicular (C) cells produce calcitonin. The four parathyroid glands sit posterior to the thyroid and secrete PTH to regulate calcium. The adrenal glands are paired retroperitoneal organs capping each kidney, with an outer cortex (producing cortisol, aldosterone, and adrenal androgens in three zones: glomerulosa, fasciculata, reticularis) and an inner medulla (producing catecholamines — epinephrine, norepinephrine). The pancreas contains the islets of Langerhans: beta cells (insulin), alpha cells (glucagon), delta cells (somatostatin), and PP cells. Gonadal endocrine function is handled by the ovaries (estradiol, progesterone) and testes (testosterone).
The Three Core Feedback Axes
Almost every endocrine diagnosis is a disruption of one of these axes. Memorize them cold.
Hypothalamus releases CRH → anterior pituitary releases ACTH → adrenal cortex (zona fasciculata) releases cortisol. Cortisol feeds back negatively on both the hypothalamus and pituitary. Disruptions: Cushing syndrome (excess cortisol), Addison disease (primary adrenal insufficiency), secondary/tertiary adrenal insufficiency (pituitary/hypothalamic failure).
Hypothalamus releases TRH → anterior pituitary releases TSH → thyroid releases T4 and T3. T4/T3 feed back negatively. Disruptions: primary hypothyroidism (high TSH, low T4), hyperthyroidism (low TSH, high T4/T3), central hypothyroidism (low or inappropriately normal TSH with low T4).
Hypothalamus releases pulsatile GnRH → anterior pituitary releases LH and FSH → gonads release testosterone (testes) or estradiol/progesterone (ovaries). Disruptions: primary hypogonadism (high LH/FSH, low sex steroid), secondary/central hypogonadism (low LH/FSH, low sex steroid), PCOS (elevated LH:FSH ratio).
Know the axes cold. When the endocrinologist says "this looks like central hypothyroidism," you should immediately know that means low T4 with an inappropriately low or normal TSH, and the workup will include a pituitary MRI and evaluation of the other anterior pituitary hormones. Pattern recognition saves you constant clarification.
02 Scribe Documentation Framework
Endocrinology notes are lab-heavy, trend-heavy, and medication-titration-heavy. Unlike procedural specialties, the action of the visit is often interpreting numbers and adjusting dosages. Your job is to capture the numbers, the trend, and the dose changes accurately.
Chief Complaint: Usually a follow-up for a known condition ("diabetes follow-up," "thyroid nodule," "post-thyroidectomy follow-up"). New consults will be referral-driven ("abnormal TSH," "incidental adrenal mass," "osteoporosis on DEXA").
HPI: In diabetes, document fingerstick trends (fasting, post-prandial, nocturnal), hypoglycemia episodes and severity, current insulin or oral regimen and adherence, CGM time-in-range, diet and exercise patterns, and any sick days or steroid use that affected glucose. In thyroid, document symptoms of hyper- or hypothyroidism, medication adherence, neck symptoms, and prior biopsies. In bone disease, document prior fractures, falls, calcium/vitamin D intake, and bisphosphonate/denosumab history.
PMHx, Medications, Family History: Always capture full diabetes history (type, duration, complications: retinopathy, nephropathy, neuropathy, CAD, PAD), thyroid history, autoimmune diseases, malignancies, and reproductive history. Family history is critical for MEN syndromes, autoimmune thyroid disease, familial hypocalciuric hypercalcemia, and type 1 diabetes.
Vitals: BP, HR, weight (trended — important for thyroid, diabetes, adrenal disorders), BMI, orthostatics if adrenal insufficiency suspected.
Physical Exam: Thyroid palpation (size, nodules, tenderness), neck lymphadenopathy, skin (hyperpigmentation for Addison, striae for Cushing, vitiligo, acanthosis nigricans, diabetic dermopathy), proximal muscle strength, reflexes (delayed relaxation in hypothyroidism, hyperreflexia in hyperthyroidism), eye exam for proptosis/lid lag, diabetic foot exam with monofilament testing.
Results: Document all labs with specific numbers and reference ranges if abnormal — A1C, fasting glucose, TSH, free T4, free T3, cortisol, ACTH, PTH, calcium (corrected for albumin), vitamin D (25-OH), aldosterone, renin, IGF-1, prolactin, etc. Always trend labs against prior values.
Document each problem discretely with its current status (controlled, uncontrolled, worsening, improving), the specific medication change (dose up, dose down, add agent, hold agent), target for next visit, and follow-up interval. Endocrinology plans are almost always numeric: "Increase glargine from 24 to 28 units qHS. Target fasting 80–130. Recheck A1C in 3 months."
Always document the trend, not just the current value. "TSH 8.2 (prior 5.1 six months ago)" is far more useful than "TSH 8.2." Endocrinologists make decisions based on trajectory, and your note should support that reasoning.
03 Type 2 Diabetes Mellitus (T2DM) Metabolic
T2DM is by far the most common diagnosis in an endocrinology clinic, accounting for >90% of all diabetes. It is a disorder of insulin resistance with progressive beta-cell failure. Early in disease, the pancreas compensates by producing more insulin; eventually beta-cell mass declines and hyperglycemia ensues. T2DM is driven by obesity, sedentary lifestyle, genetic predisposition, and aging, and is strongly associated with metabolic syndrome (central obesity, hypertension, dyslipidemia, hyperglycemia).
Diagnostic Criteria
Per the ADA Standards of Care 2024, diabetes is diagnosed by any of: A1C ≥ 6.5%, fasting plasma glucose ≥ 126 mg/dL, 2-hour plasma glucose ≥ 200 mg/dL during OGTT, or random glucose ≥ 200 mg/dL with classic symptoms. In the absence of unequivocal hyperglycemia, two abnormal tests are required.
| Test | Normal | Prediabetes | Diabetes |
|---|---|---|---|
| A1C | < 5.7% | 5.7–6.4% | ≥ 6.5% |
| Fasting Plasma Glucose | < 100 mg/dL | 100–125 mg/dL (IFG) | ≥ 126 mg/dL |
| 2-hr OGTT Glucose | < 140 mg/dL | 140–199 mg/dL (IGT) | ≥ 200 mg/dL |
| Random Glucose | — | — | ≥ 200 mg/dL with symptoms |
ADA Glycemic Targets
A1C < 7.0% for most non-pregnant adults. A1C < 6.5% may be appropriate for younger patients with short diabetes duration and low hypoglycemia risk. A1C < 8.0% is acceptable for older adults, limited life expectancy, or history of severe hypoglycemia. Fasting / preprandial glucose 80–130 mg/dL. Peak postprandial glucose < 180 mg/dL. CGM time-in-range (70–180 mg/dL) > 70% with < 4% time below 70 and < 1% time below 54.
Management Framework
Per the ADA 2024 pharmacologic approach, first-line therapy combines lifestyle modification with metformin and, increasingly, agents with cardiovascular or renal benefit regardless of A1C. For patients with established ASCVD, heart failure, or CKD, GLP-1 receptor agonists (especially semaglutide, liraglutide, dulaglutide) and SGLT2 inhibitors (empagliflozin, dapagliflozin, canagliflozin) are preferred as second agents. For patients where weight loss is the dominant goal, GLP-1 RAs and dual GIP/GLP-1 agonists (tirzepatide) are preferred. Sulfonylureas (glipizide, glimepiride) and TZDs (pioglitazone) remain lower-cost options. Insulin is added when oral/injectable non-insulin therapy fails to achieve targets or when A1C > 10% at presentation with catabolic symptoms.
Screening for Complications
Every T2DM follow-up should screen for: retinopathy (annual dilated eye exam), nephropathy (annual urine albumin-to-creatinine ratio and eGFR), neuropathy (annual monofilament foot exam), cardiovascular disease, and foot ulcers. Document lipid panel (statin for most), blood pressure, and ACE-I/ARB if albuminuria.
Always capture the A1C trend (at least last 3 values), the specific medication regimen with doses and frequency, any hypoglycemic episodes in the past 3 months, and CGM/fingerstick patterns. "A1C 8.1 (was 7.6 and 7.2)" tells the next provider the trajectory is wrong and triggers intensification.
04 Type 1 Diabetes Mellitus (T1DM) Metabolic
T1DM is an autoimmune destruction of pancreatic beta cells, leading to absolute insulin deficiency. It accounts for 5–10% of diabetes. Peak onset is in childhood and adolescence, but latent autoimmune diabetes of adults (LADA) can present at any age. Patients require exogenous insulin for survival — stopping insulin leads to DKA within hours to days.

Pathophysiology & Diagnosis
Autoimmune destruction is mediated by T cells targeting islet antigens. Antibodies against GAD65, IA-2, insulin, and ZnT8 confirm autoimmune etiology. C-peptide (a byproduct of endogenous insulin synthesis) is low or undetectable. Initial presentation is often DKA or a weeks-long history of polyuria, polydipsia, polyphagia, and weight loss.
Insulin Regimens
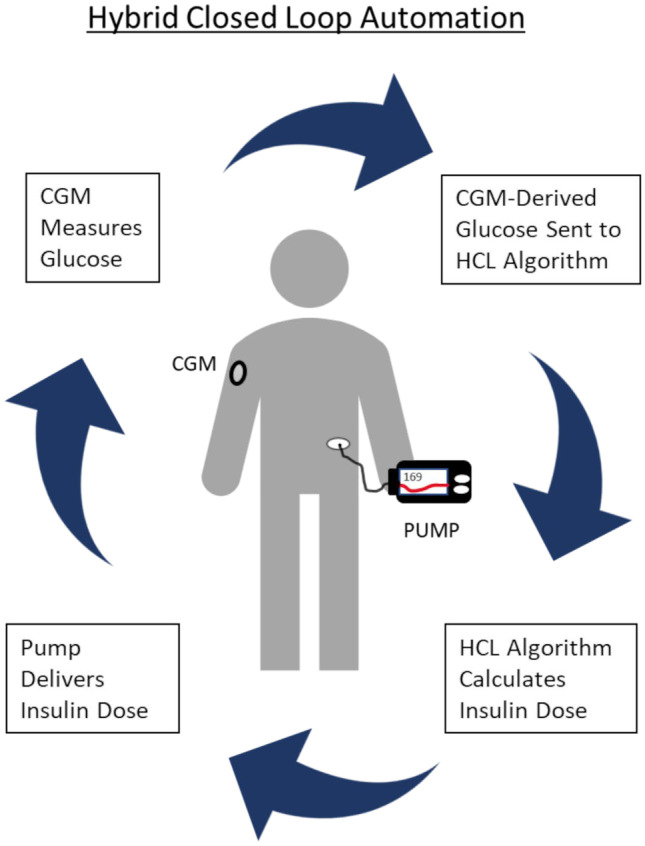
Standard T1DM management is a basal-bolus regimen: long-acting basal insulin (glargine U-100, glargine U-300, degludec, detemir) once or twice daily, plus rapid-acting bolus insulin (lispro, aspart, glulisine, faster aspart) before each meal, dosed using a carb ratio and correction factor. Insulin pumps (CSII — continuous subcutaneous insulin infusion) deliver basal as a continuous micro-infusion with user-initiated boluses. Hybrid closed-loop systems (Tandem Control-IQ, Medtronic 780G, Omnipod 5) use CGM data to automate basal and correction dosing.
Carb ratio (I:C): Units of insulin per gram of carbohydrate (e.g., 1:10 = 1 unit per 10 g carbs). Correction factor (insulin sensitivity factor, ISF): mg/dL drop in glucose per unit of insulin (e.g., 1:50). Total daily dose (TDD): Sum of all insulin over 24 hours; typically 0.5–1.0 units/kg/day in T1DM, split roughly 50% basal / 50% bolus.
CGM & Technology
Continuous glucose monitors (Dexcom G6/G7, Freestyle Libre 2/3, Medtronic Guardian) report glucose every 1–5 minutes. Key metrics: time in range (TIR) 70–180 mg/dL (goal > 70%), time below range (TBR) < 70 (goal < 4%), glucose management indicator (GMI) — CGM-derived A1C estimate, and coefficient of variation (CV) (goal < 36%).
In T1DM visits, always document the exact pump settings if the patient is on CSII: basal rate segments (e.g., "midnight–6am 0.8 U/hr, 6am–10pm 1.0 U/hr"), carb ratios by time block, correction factor, active insulin time, and target glucose. When the endocrinologist adjusts these, you must capture the new values precisely.
05 Diabetic Ketoacidosis (DKA) & HHS Emergency
Two life-threatening hyperglycemic emergencies. DKA predominates in T1DM (but occurs in T2DM, especially with SGLT2i use — "euglycemic DKA"). HHS (hyperosmolar hyperglycemic state) predominates in T2DM, especially elderly patients.
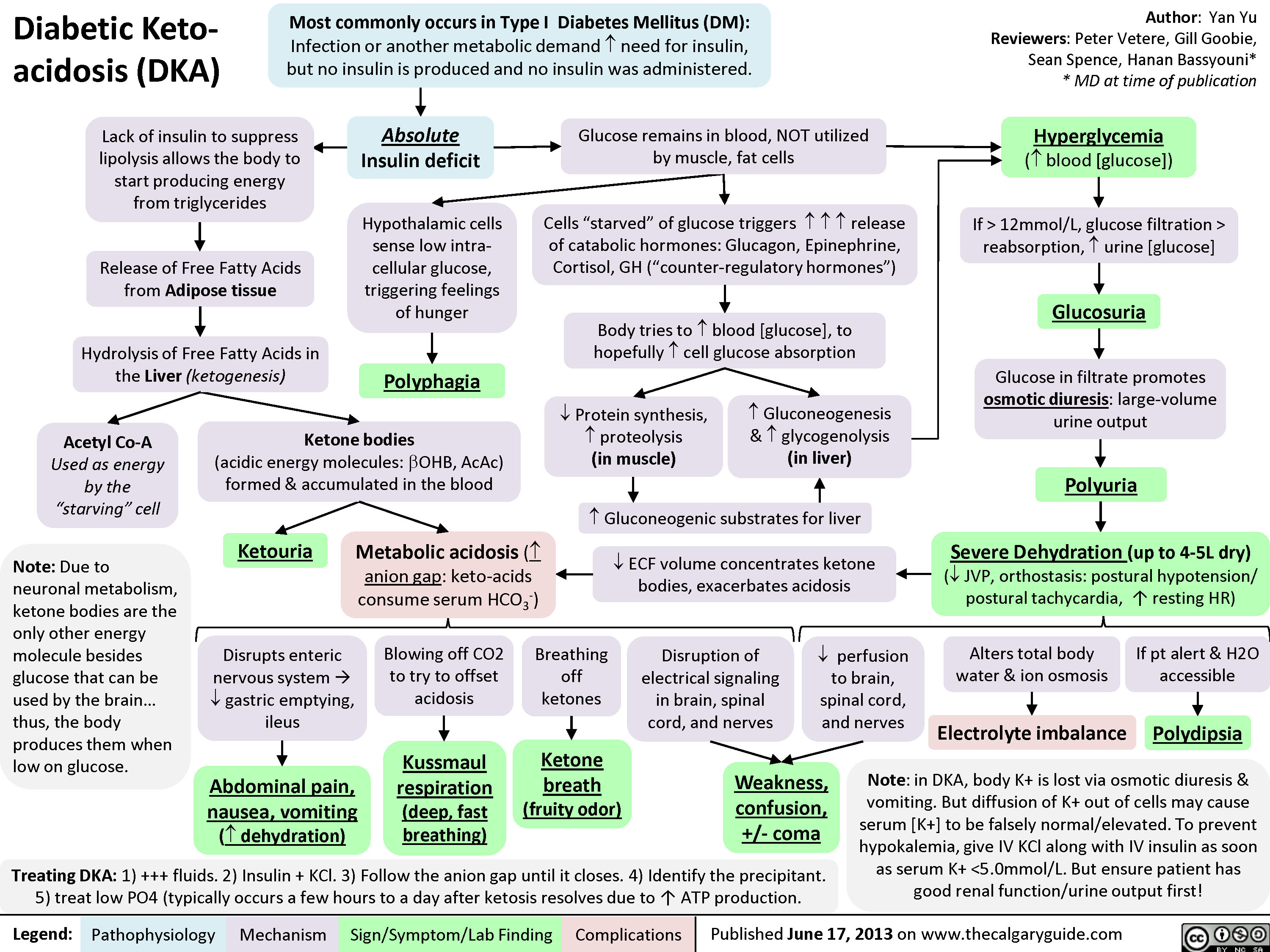
Glucose > 250 mg/dL (can be lower in euglycemic DKA), anion gap > 10–12, bicarbonate < 18 mEq/L, pH < 7.30, positive serum or urine ketones (beta-hydroxybutyrate preferred > 3 mmol/L). Severity: mild (pH 7.25–7.30), moderate (7.00–7.24), severe (< 7.00).
Glucose > 600 mg/dL, serum osmolality > 320 mOsm/kg, pH > 7.30, bicarbonate > 18, minimal or no ketosis, altered mental status proportional to osmolality.
Management
Both conditions require aggressive IV fluid resuscitation (normal saline, transitioning to 0.45% NS once volume replete), IV insulin infusion (typically 0.1 U/kg/hr after initial bolus or without bolus), potassium replacement (hold insulin if K < 3.3), and identification of the precipitating event (infection, MI, missed insulin, new-onset diabetes, pump failure). In DKA, continue IV insulin until the anion gap closes, not just until glucose normalizes — adding D5 to IVF when glucose < 200–250. Transition to subcutaneous insulin with 1–2 hour overlap.
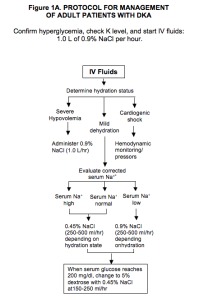
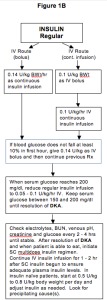
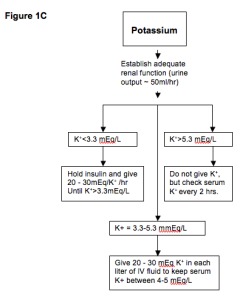
Scribes rarely document DKA from an endocrine clinic standpoint (inpatient service handles it), but endocrinology is often consulted. In the consult note, capture the precipitant, serial labs (gap, pH, glucose, K), current insulin drip rate, and the transition plan off the drip.
06 Hypoglycemia Metabolic
Hypoglycemia is defined by the Whipple triad: low plasma glucose, symptoms consistent with hypoglycemia, and resolution of symptoms with glucose administration. In diabetes, the ADA defines: Level 1 glucose < 70 mg/dL (alert value), Level 2 < 54 mg/dL (clinically significant), Level 3 severe hypoglycemia requiring assistance from another person regardless of the number.
Symptoms are adrenergic (tremor, palpitations, sweating, anxiety) and neuroglycopenic (confusion, difficulty concentrating, slurred speech, seizure, coma). Hypoglycemia unawareness — loss of autonomic warning symptoms — develops after recurrent hypoglycemia and is a major indication for CGM and insulin regimen reassessment. In non-diabetic hypoglycemia, workup includes insulinoma evaluation (72-hour fast with paired glucose, insulin, C-peptide, proinsulin, beta-hydroxybutyrate, and sulfonylurea screen).
For conscious hypoglycemia: give 15 g fast-acting carbohydrate (4 oz juice, 3–4 glucose tablets), recheck in 15 minutes, repeat until glucose > 70. For severe hypoglycemia: glucagon (injection or nasal 3 mg Baqsimi), or IV dextrose (D50 in hospital).
07 Gestational Diabetes & Prediabetes Metabolic
Gestational diabetes (GDM) is diabetes first recognized in pregnancy, typically diagnosed at 24–28 weeks. Two-step screening: 50 g 1-hour glucose challenge test (GCT); if > 130–140 mg/dL, proceed to 100 g 3-hour OGTT (Carpenter-Coustan criteria: fasting ≥ 95, 1-hr ≥ 180, 2-hr ≥ 155, 3-hr ≥ 140 — two abnormal values diagnose GDM). One-step alternative: 75 g 2-hour OGTT (fasting ≥ 92, 1-hr ≥ 180, 2-hr ≥ 153 — one abnormal value diagnoses).
Management: medical nutrition therapy first, insulin if targets not met (fasting < 95, 1-hr postprandial < 140, 2-hr < 120). Metformin and glyburide cross the placenta and are less preferred per ACOG/ADA. Postpartum 75-g OGTT at 6–12 weeks to detect persistent diabetes.
Prediabetes (A1C 5.7–6.4%, IFG 100–125, IGT 140–199) affects >90 million US adults. The Diabetes Prevention Program showed intensive lifestyle intervention (7% weight loss, 150 min/week exercise) reduces progression to diabetes by 58%. Metformin 850 mg BID is second-line, especially in younger patients with BMI > 35 or prior GDM.
08 Hypothyroidism Thyroid
Hypothyroidism is deficiency of thyroid hormone. Primary hypothyroidism (thyroid gland failure) accounts for >95% of cases, most commonly from Hashimoto thyroiditis (chronic autoimmune lymphocytic thyroiditis with anti-TPO and anti-thyroglobulin antibodies). Other causes: post-ablation (radioactive iodine, thyroidectomy), drug-induced (amiodarone, lithium, checkpoint inhibitors), iodine deficiency, and congenital.
Clinical Presentation
Fatigue, cold intolerance, weight gain, constipation, dry skin, hair loss, menorrhagia, bradycardia, delayed deep tendon reflex relaxation, myxedematous facies, hoarseness, depression, cognitive slowing. Severe untreated hypothyroidism can progress to myxedema coma — hypothermia, hypoventilation, hyponatremia, altered mental status — a rare but lethal endocrine emergency.
Diagnosis
| Pattern | TSH | Free T4 | Interpretation |
|---|---|---|---|
| Overt primary hypothyroidism | ↑↑ (> 10) | ↓ | Treat with levothyroxine |
| Subclinical hypothyroidism | ↑ (4.5–10) | Normal | Treat if symptomatic, pregnant, TPO+, or TSH > 10 |
| Central hypothyroidism | Low or inappropriately normal | ↓ | Workup for pituitary/hypothalamic disease |
| Euthyroid sick syndrome | Low | Low or normal | Nonthyroidal illness; do not treat acutely |
Management
Per the ATA hypothyroidism guidelines, levothyroxine (L-T4) is the standard. Starting dose is approximately 1.6 mcg/kg/day for overt primary hypothyroidism in healthy adults; lower starting doses (25–50 mcg) in the elderly or those with CAD. Recheck TSH in 6–8 weeks after any dose change. Take on an empty stomach, 30–60 min before food, and separate from calcium, iron, and PPIs. Goal TSH is within the normal reference range for most patients; 0.5–2.5 in pregnancy (trimester-specific).
Always document the current levothyroxine dose, whether the patient is taking it correctly (empty stomach, timing), and the specific TSH value with prior comparison. Dose changes are nearly always 12.5–25 mcg increments, and the scribe captures the new dose and the recheck interval.
09 Hyperthyroidism & Thyrotoxicosis Thyroid
Thyrotoxicosis is the clinical state of excess thyroid hormone. Hyperthyroidism specifically refers to excess thyroid hormone production (as opposed to thyroiditis or exogenous intake). Major causes:
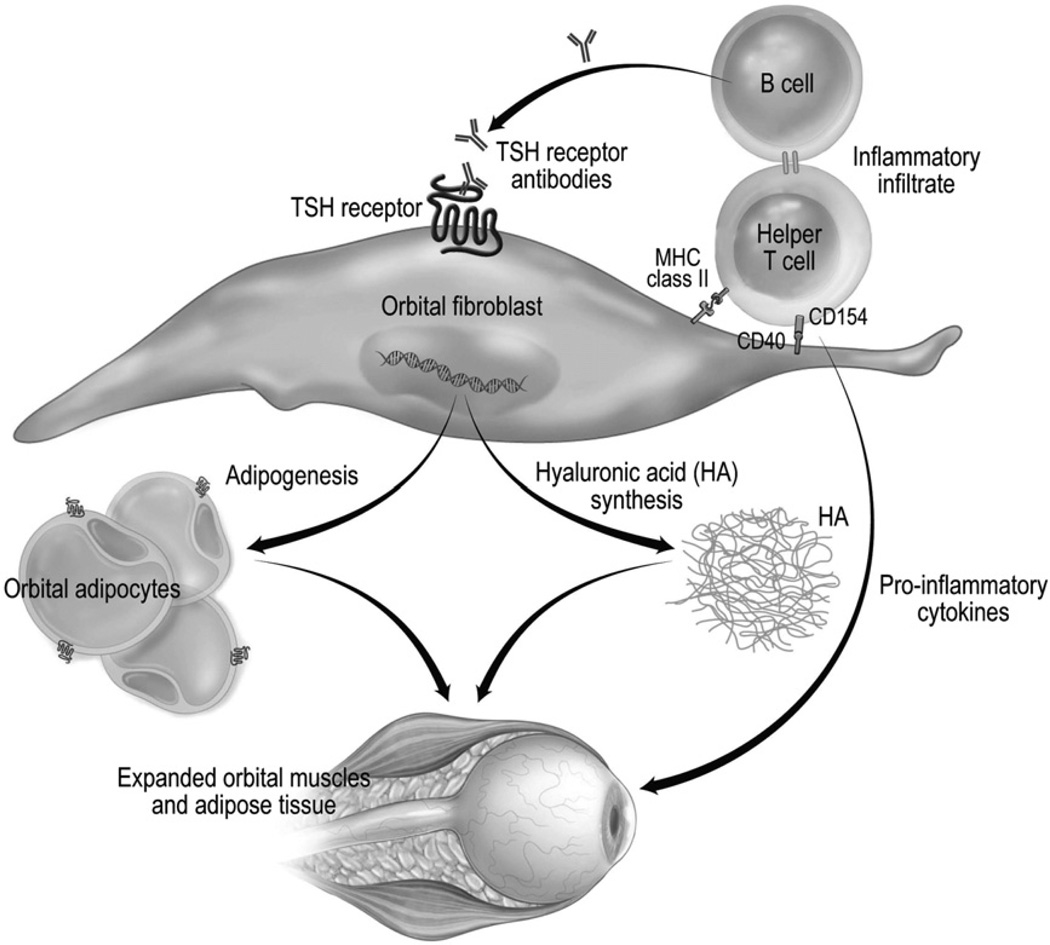
Autoimmune hyperthyroidism caused by TSH receptor-stimulating antibodies (TRAb / TSI). Presents with diffuse goiter, orbitopathy (proptosis, lid lag, diplopia), and occasionally pretibial myxedema. Most common cause of hyperthyroidism in young adults.
Autonomous hormone production from one or more thyroid nodules. More common in older adults and iodine-deficient regions. Radioactive iodine uptake scan shows focal "hot" nodules with suppression of surrounding thyroid (toxic adenoma) or patchy uptake (MNG).
Destructive release of preformed hormone — not true hyperthyroidism. Includes subacute (de Quervain, painful, post-viral), silent/postpartum (painless, lymphocytic), and drug-induced (amiodarone type 2). Radioactive iodine uptake is low, distinguishing it from Graves and toxic nodular disease.
Clinical Presentation
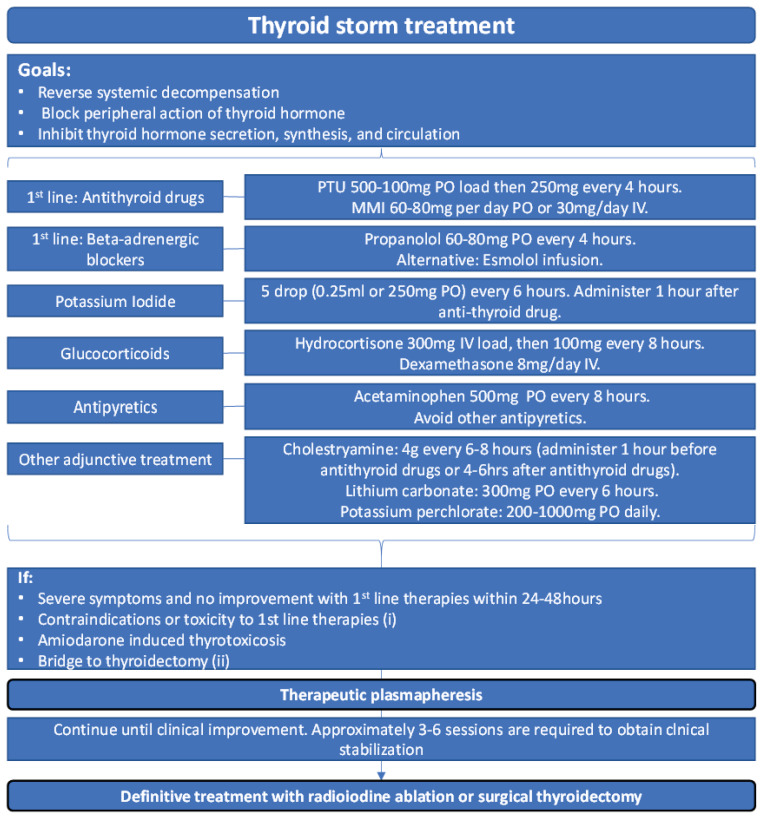
Weight loss despite increased appetite, heat intolerance, sweating, palpitations, tremor, anxiety, insomnia, diarrhea, oligomenorrhea, hyperreflexia, warm moist skin, fine hair, lid lag, proptosis (Graves), goiter. Thyroid storm is the emergency: fever, tachycardia, AMS, cardiovascular collapse.
Diagnosis & Workup
Initial: low TSH, elevated free T4 and/or free T3. Next step: TRAb (positive in Graves) and/or radioactive iodine uptake (RAIU) and scan. Diffuse increased uptake = Graves; focal increased uptake = toxic adenoma or MNG; low uptake = thyroiditis or exogenous intake.
Management
Per the ATA hyperthyroidism guidelines, three definitive options for Graves: antithyroid drugs (ATDs), radioactive iodine (RAI), or thyroidectomy. Methimazole is first-line ATD (5–40 mg daily); PTU is reserved for first-trimester pregnancy, thyroid storm, or methimazole intolerance due to hepatotoxicity risk. Beta-blockers (propranolol, atenolol) control adrenergic symptoms. RAI is a single oral dose of I-131 that ablates the thyroid; most patients become hypothyroid and require lifelong levothyroxine. Thyroidectomy is indicated for large goiters, concurrent nodules suspicious for cancer, severe ophthalmopathy, or patient preference.
Always clarify whether the patient has Graves (autoimmune) or nodular disease — management differs significantly. Document TRAb status, RAIU scan result, current ATD dose, and beta-blocker. Note any ophthalmopathy symptoms (diplopia, pain, pressure, vision changes) because severe eye disease may alter RAI decision.
10 Thyroid Nodules & Thyroid Cancer Thyroid
Thyroid nodules are extremely common (up to 60% of adults on ultrasound) but only ~5% are malignant. The scribe's role is to document the size, sonographic features, TI-RADS category, FNA cytology (Bethesda), and management decisions.
Workup
Every nodule gets a TSH and a neck ultrasound. If TSH is low, obtain a RAIU scan — "hot" nodules are almost never malignant and do not need FNA. If TSH is normal/high, proceed to ultrasound-based risk stratification with ACR TI-RADS and decide on FNA by size thresholds.
ACR TI-RADS
Points assigned for composition, echogenicity, shape, margin, and echogenic foci. Total points determine TR category:
TR1 — Benign (0 points): No FNA. TR2 — Not suspicious (2 points): No FNA. TR3 — Mildly suspicious (3 points): FNA if ≥ 2.5 cm; follow if ≥ 1.5 cm. TR4 — Moderately suspicious (4–6 points): FNA if ≥ 1.5 cm; follow if ≥ 1.0 cm. TR5 — Highly suspicious (≥ 7 points): FNA if ≥ 1.0 cm; follow if ≥ 0.5 cm.
Bethesda System for Thyroid Cytopathology
I — Nondiagnostic/Unsatisfactory: 5–10% malignancy risk. Repeat FNA. II — Benign: 0–3% risk. Clinical/US follow-up. III — AUS/FLUS (atypia of undetermined significance): 10–30% risk. Repeat FNA or molecular testing. IV — Follicular neoplasm / suspicious for follicular neoplasm: 25–40% risk. Lobectomy or molecular testing. V — Suspicious for malignancy: 50–75% risk. Surgery (lobectomy or total thyroidectomy). VI — Malignant: 97–99% risk. Total thyroidectomy.
Thyroid Cancer Types
Per the 2015 ATA thyroid nodule and cancer guidelines:
- Papillary thyroid cancer (PTC): ~85% of thyroid cancers. Excellent prognosis. Lymphatic spread common but rarely affects survival.
- Follicular thyroid cancer (FTC): ~10%. Hematogenous spread (bone, lung).
- Medullary thyroid cancer (MTC): 2–4%. Arises from parafollicular C cells; produces calcitonin. Associated with MEN2.
- Anaplastic thyroid cancer: < 2%. Extremely aggressive, median survival months.
Management depends on size, extrathyroidal extension, and lymph node involvement. Options range from lobectomy (low-risk micro-PTC) to total thyroidectomy with central/lateral neck dissection and post-op RAI for higher-risk disease. TSH suppression with levothyroxine is standard post-op to reduce recurrence, titrated to risk category.
11 Primary Hyperparathyroidism & Calcium Disorders Bone/Mineral
Primary hyperparathyroidism (PHPT) is autonomous PTH overproduction, usually from a single parathyroid adenoma (85%), four-gland hyperplasia (15%), or rarely carcinoma (< 1%). Most cases are discovered incidentally on routine chemistry showing hypercalcemia. Classic symptoms ("stones, bones, groans, psychiatric overtones"): nephrolithiasis, osteoporosis/fractures, abdominal pain/constipation, fatigue/depression.
Diagnosis
Hypercalcemia with inappropriately normal or elevated PTH establishes the diagnosis. Rule out familial hypocalciuric hypercalcemia (FHH) with 24-hour urine calcium (low in FHH, < 100 mg/24h; calcium-to-creatinine clearance ratio < 0.01). Check 25-OH vitamin D (often low and must be replaced). Additional workup: DEXA (all three sites including distal radius), renal US for stones, 24-hour urine calcium for stone risk.
Management
Per the Fourth International Workshop guidelines, parathyroidectomy is indicated for: symptomatic disease, serum calcium > 1.0 mg/dL above ULN, eGFR < 60, 24-hr urine calcium > 400 mg/24h with increased stone risk, nephrolithiasis/nephrocalcinosis, T-score ≤ −2.5 at any site, vertebral fracture, or age < 50. Pre-op localization with sestamibi scan and/or 4D-CT. Intraoperative PTH monitoring confirms cure (> 50% drop and into normal range).
Non-surgical management: cinacalcet (calcimimetic) for medical control, bisphosphonates for bone protection, adequate hydration, vitamin D repletion.
12 Osteoporosis & Metabolic Bone Disease Bone/Mineral
Osteoporosis is a systemic skeletal disease characterized by low bone mass and microarchitectural deterioration, leading to increased fracture risk. The diagnosis is made by: (1) T-score ≤ −2.5 on DEXA at the lumbar spine, femoral neck, total hip, or 33% radius; OR (2) a fragility fracture (hip or vertebral) regardless of T-score; OR (3) osteopenia with elevated FRAX 10-year risk (≥ 20% major osteoporotic or ≥ 3% hip).
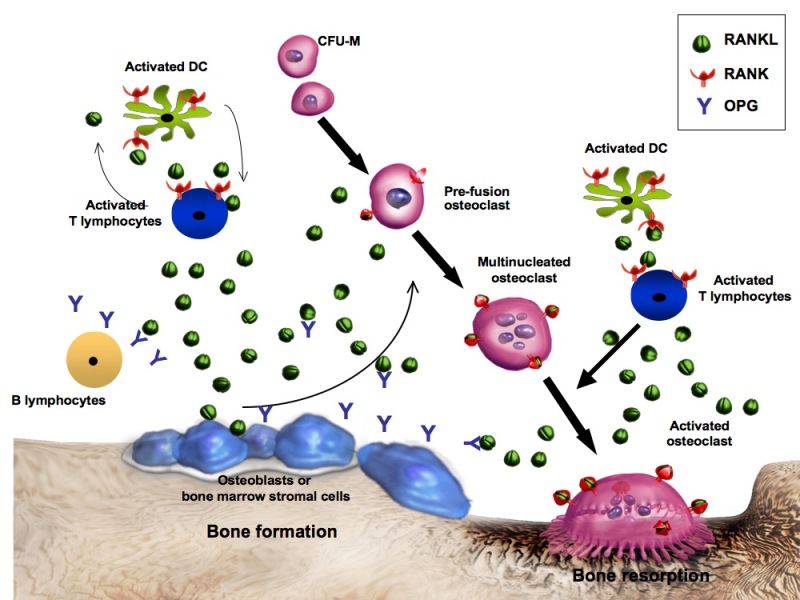
T-Score Classification
Normal: T-score ≥ −1.0. Osteopenia (low bone mass): T-score between −1.0 and −2.5. Osteoporosis: T-score ≤ −2.5. Severe (established) osteoporosis: T-score ≤ −2.5 with one or more fragility fractures.
T-score compares to a young adult reference population; Z-score compares to age/sex-matched peers and is used in premenopausal women, men < 50, and children.
FRAX
The FRAX calculator estimates 10-year probability of major osteoporotic fracture (clinical vertebral, hip, forearm, humerus) and hip fracture using: age, sex, weight, height, prior fracture, parental hip fracture, current smoking, glucocorticoid use, rheumatoid arthritis, secondary osteoporosis, ≥ 3 alcohol drinks/day, and femoral neck BMD. Treatment thresholds (US): 10-year major osteoporotic fracture risk ≥ 20% or hip fracture risk ≥ 3%.
Management
Per the AACE/ACE osteoporosis guidelines: ensure adequate calcium (1000–1200 mg/day) and vitamin D (800–1000 IU/day or higher to 25-OH D > 30). Pharmacologic options:
- Bisphosphonates (alendronate, risedronate, ibandronate, zoledronic acid): first-line antiresorptives. Oral weekly/monthly or IV annually.
- Denosumab (Prolia): RANK-ligand inhibitor, 60 mg SC q6 months. Must not be interrupted — rebound vertebral fractures.
- Teriparatide / Abaloparatide: PTH analogs, anabolic (build bone). Daily SC injection, max 2 years. For severe osteoporosis or multiple fractures.
- Romosozumab (Evenity): sclerostin inhibitor, dual anabolic/antiresorptive. Monthly SC × 12 months. Cardiovascular warning.
- Raloxifene: SERM, for postmenopausal women also seeking breast cancer risk reduction.
On osteoporosis follow-up, always capture: date and results of last DEXA (T-scores at each site), interim fractures, calcium/vitamin D supplementation, current antiresorptive/anabolic with start date and duration, and any side effects (GI with oral bisphosphonates, ONJ, atypical femur fracture).
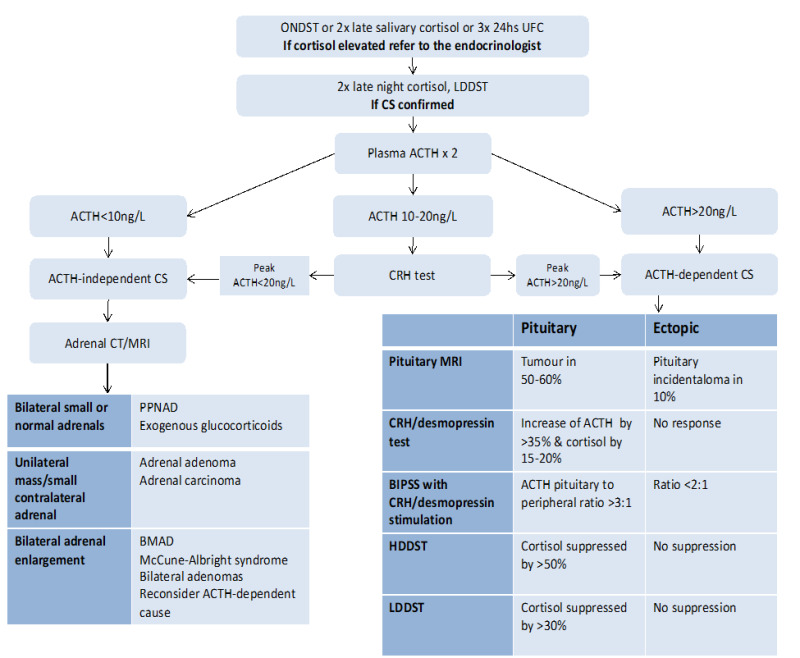
13 Cushing Syndrome Adrenal
Cushing syndrome is the clinical state of chronic cortisol excess. The most common cause is exogenous (iatrogenic glucocorticoid use). Endogenous causes are divided into ACTH-dependent (80%) and ACTH-independent (20%): Cushing disease (pituitary ACTH-secreting adenoma) — most common endogenous cause, ectopic ACTH (small cell lung cancer, bronchial carcinoid), adrenal adenoma/carcinoma, adrenal hyperplasia.
Clinical Presentation
Central obesity with thin extremities, "moon facies," dorsocervical fat pad ("buffalo hump"), supraclavicular fullness, wide (> 1 cm) purple striae, easy bruising, proximal muscle weakness, hypertension, hyperglycemia/diabetes, osteoporosis, hirsutism, menstrual irregularities, depression/psychosis, facial plethora.
Diagnostic Workup
Per the Endocrine Society Cushing guidelines, after excluding exogenous steroid use, screen with any two of:
- 24-hour urine free cortisol (> 3× ULN = positive)
- Late-night salivary cortisol (elevated = positive)
- 1 mg overnight dexamethasone suppression test (morning cortisol > 1.8 mcg/dL = positive)
After confirming hypercortisolism, measure ACTH to localize: suppressed ACTH = adrenal source (CT adrenals); normal/elevated ACTH = pituitary or ectopic. Further testing includes high-dose dexamethasone suppression, CRH stimulation, pituitary MRI, and inferior petrosal sinus sampling (IPSS) if pituitary MRI is negative or equivocal.
Management
Transsphenoidal surgery for Cushing disease. Adrenalectomy for adrenal adenoma. Tumor resection for ectopic ACTH. Medical therapy (ketoconazole, metyrapone, osilodrostat, mifepristone, pasireotide) is used when surgery is impossible, as a bridge, or for residual disease.
14 Adrenal Insufficiency Adrenal
Adrenal insufficiency is deficient adrenal cortex hormone production. Primary (Addison disease) is adrenal gland failure — usually autoimmune adrenalitis in the developed world, TB globally — causing deficiency of cortisol, aldosterone, and adrenal androgens. Secondary is pituitary ACTH deficiency (pituitary tumor, surgery, Sheehan syndrome); aldosterone is preserved because it's controlled by renin-angiotensin. Tertiary is hypothalamic CRH deficiency, most commonly from chronic exogenous glucocorticoid suppression.
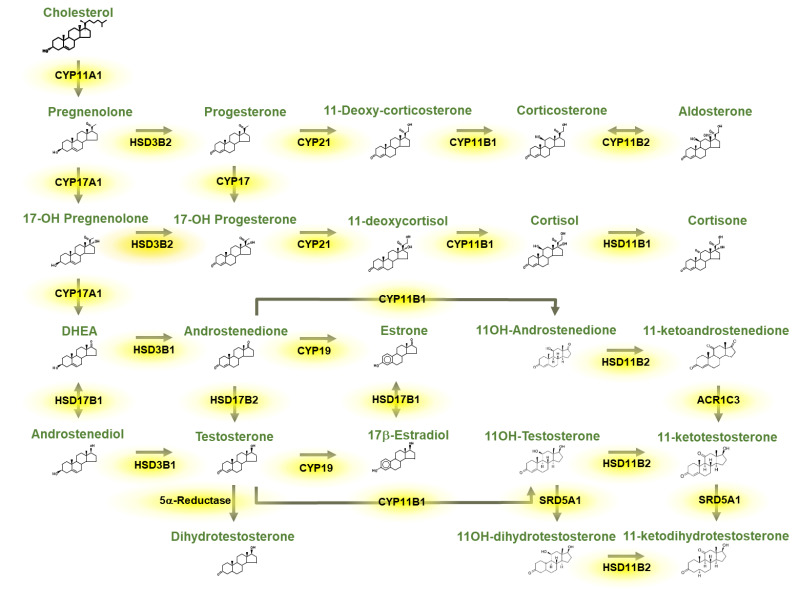
Clinical Presentation
Fatigue, weight loss, anorexia, nausea, orthostatic hypotension, salt craving, hyperpigmentation (primary only, due to high ACTH/MSH activity — palmar creases, buccal mucosa, scars), hyponatremia, hyperkalemia (primary only), hypoglycemia. Adrenal crisis: hypotension, shock, abdominal pain, vomiting — an endocrine emergency requiring immediate IV hydrocortisone 100 mg and fluid resuscitation.
Diagnosis
Per the Endocrine Society adrenal insufficiency guidelines: 8 AM cortisol < 3 mcg/dL is diagnostic; > 18 excludes. Intermediate values warrant cosyntropin (ACTH) stimulation test: baseline cortisol, inject 250 mcg cosyntropin IV/IM, recheck cortisol at 30 and 60 min. Normal response: peak ≥ 18 mcg/dL. After confirmation, measure ACTH (high = primary, low/normal = central), plus renin/aldosterone and adrenal antibodies (21-hydroxylase).
Management
Physiologic glucocorticoid replacement: hydrocortisone 15–25 mg/day divided (e.g., 10–5–5), or prednisone 3–5 mg daily. Primary also requires fludrocortisone 0.05–0.2 mg daily (mineralocorticoid). Stress-dose steroid education is critical: double or triple oral dose for minor illness, IM hydrocortisone 100 mg if vomiting or severely ill, emergency MedicAlert bracelet. Perioperative stress dosing protocols depend on surgical stress level.
15 Pheochromocytoma & Hyperaldosteronism Adrenal
Pheochromocytoma
Catecholamine-secreting tumor of adrenal medullary chromaffin cells (extra-adrenal paragangliomas arise from sympathetic chain). Classic triad: episodic headache, palpitations, diaphoresis — with paroxysmal or sustained hypertension. Rule of 10s (historical): 10% bilateral, 10% extra-adrenal, 10% malignant, 10% familial, 10% in children, 10% normotensive. Modern data: up to 40% have hereditary basis (MEN2, VHL, NF1, SDHx mutations).
Diagnosis: plasma-free metanephrines (most sensitive) or 24-hour urine metanephrines and catecholamines. Localization with CT or MRI adrenal; MIBG scan or DOTATATE PET for metastatic or extra-adrenal.
Management: Alpha-blockade first (phenoxybenzamine, doxazosin) for 10–14 days before adding beta-blockade — never beta-block first (unopposed alpha causes hypertensive crisis). Salt/volume loading, then laparoscopic adrenalectomy. Per the Endocrine Society pheochromocytoma guidelines, genetic testing is recommended for all patients.
Primary Hyperaldosteronism (Conn Syndrome)
Autonomous aldosterone secretion causing hypertension and hypokalemia (though ~50% are normokalemic). Causes: aldosterone-producing adenoma (APA), bilateral adrenal hyperplasia, glucocorticoid-remediable aldosteronism. Screen hypertensive patients with hypokalemia, resistant HTN, HTN before age 40, or incidental adrenal mass.
Screening: aldosterone-to-renin ratio (ARR). Elevated ARR (> 20–30) with aldosterone > 15 ng/dL warrants confirmation with saline suppression test or oral salt load. Adrenal CT then adrenal venous sampling (AVS) to distinguish unilateral (adrenalectomy) from bilateral (spironolactone or eplerenone).
16 Pituitary Disorders Pituitary
Prolactinoma
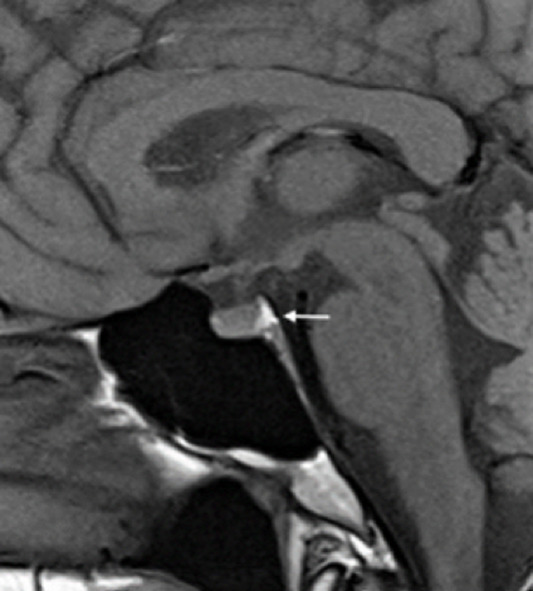
Most common functional pituitary adenoma. Microadenoma < 10 mm, macroadenoma ≥ 10 mm. Symptoms: women — galactorrhea, oligomenorrhea/amenorrhea, infertility; men — hypogonadism, decreased libido, ED, visual field defects if macroadenoma compresses the optic chiasm (classically bitemporal hemianopsia). Workup: prolactin level (> 200 ng/mL is highly specific for macroprolactinoma), pituitary MRI, evaluation of other pituitary hormones. Rule out hook effect (dilute sample for very large adenomas). Treatment: dopamine agonists — cabergoline (preferred, twice weekly) or bromocriptine — shrink tumor and normalize prolactin in >80%. Surgery reserved for drug resistance or intolerance.

Acromegaly
GH-secreting pituitary adenoma causing excess IGF-1. Insidious onset: enlarging hands/feet/jaw, coarse facial features, macroglossia, skin tags, sweating, OSA, carpal tunnel, hypertension, diabetes, cardiomyopathy. Gigantism if onset before epiphyseal closure. Diagnosis: IGF-1 (age/sex adjusted), then OGTT with failure to suppress GH < 1 ng/mL, pituitary MRI. Treatment: transsphenoidal surgery first-line; medical therapy with somatostatin analogs (octreotide, lanreotide, pasireotide), GH receptor antagonist (pegvisomant), or dopamine agonists (cabergoline). Radiation for residual.
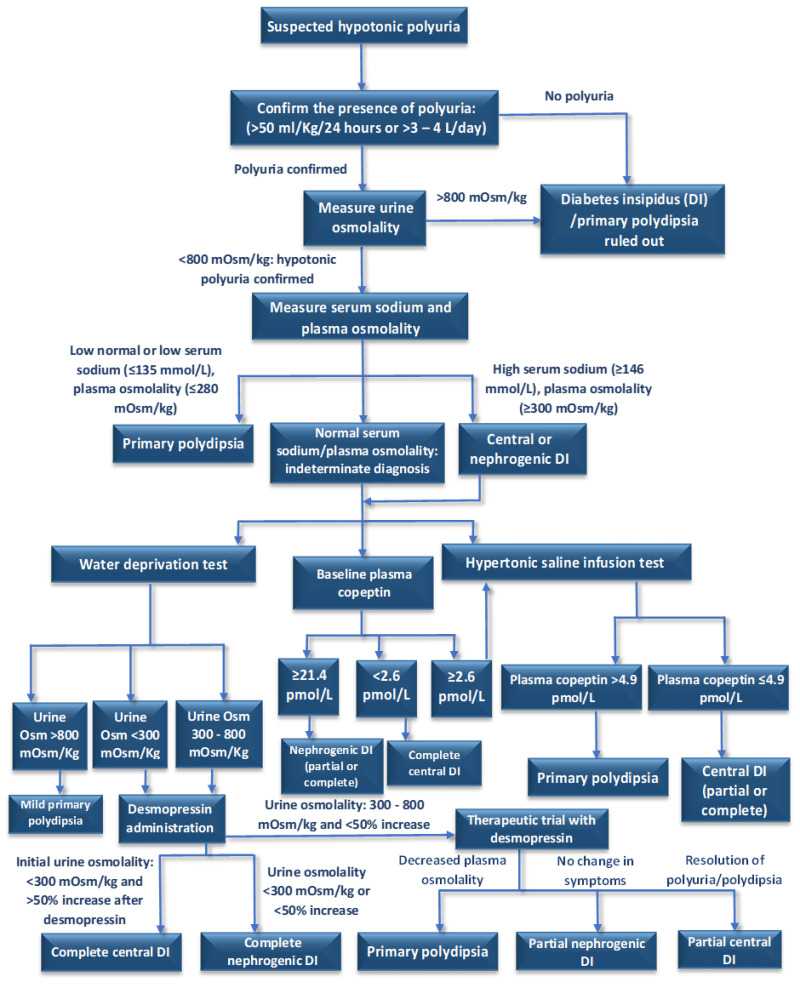
Diabetes Insipidus (Arginine Vasopressin Deficiency)
Now called AVP deficiency (central DI) or AVP resistance (nephrogenic DI). Presents with polyuria (> 3 L/day), polydipsia, dilute urine (osmolality < 300), hypernatremia if access to water is restricted. Workup: water deprivation test with desmopressin challenge — central DI responds (urine concentrates); nephrogenic does not. Treatment: desmopressin (DDAVP) for central; thiazides, low-salt diet, NSAIDs for nephrogenic.
SIADH
Syndrome of inappropriate antidiuretic hormone: euvolemic hyponatremia with concentrated urine (osm > 100), urine sodium > 40, low serum uric acid. Causes: malignancy (small cell lung), CNS disease, pulmonary disease, drugs (SSRIs, carbamazepine, cyclophosphamide). Treatment: fluid restriction, salt tablets, loop diuretics, tolvaptan for severe/refractory.
17 Reproductive Endocrinology Reproductive
Polycystic Ovary Syndrome (PCOS)
Most common endocrine disorder in reproductive-age women. Rotterdam criteria — at least 2 of 3: (1) oligo- or anovulation, (2) clinical or biochemical hyperandrogenism (hirsutism, acne, elevated free testosterone), (3) polycystic ovaries on ultrasound (≥ 20 follicles per ovary or ovarian volume > 10 mL). Rule out other causes: non-classical CAH (17-OHP), Cushing, hyperprolactinemia, thyroid dysfunction, androgen-secreting tumor. Associated: insulin resistance, obesity, T2DM, OSA, endometrial hyperplasia, subfertility.
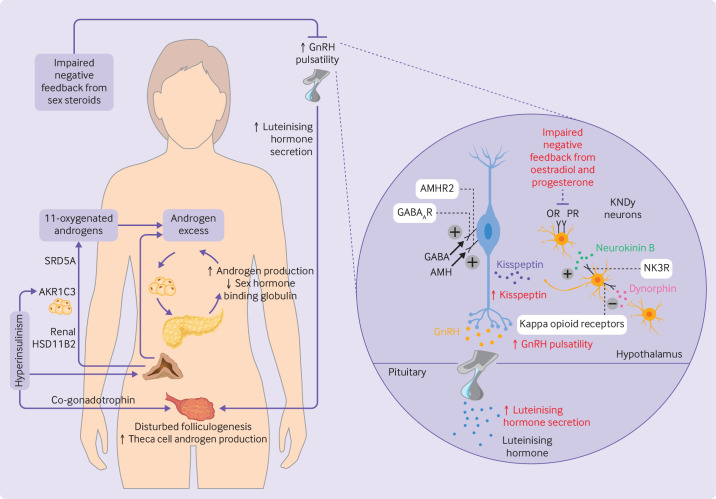
Management: lifestyle modification (5–10% weight loss), combined oral contraceptives (regulate menses, reduce androgens, protect endometrium), metformin for metabolic dysfunction, spironolactone for hirsutism/acne, letrozole for ovulation induction in women seeking pregnancy.
Male Hypogonadism
Testosterone deficiency confirmed by two separate morning (8–10 AM) total testosterone levels < 300 ng/dL with symptoms (low libido, ED, fatigue, reduced muscle mass, mood changes). Measure LH/FSH to differentiate primary (high gonadotropins) from secondary (low/normal). Workup for secondary includes prolactin, iron studies (hemochromatosis), pituitary imaging if severely low or other hormone abnormalities. Treatment: testosterone replacement (IM, gel, pellet, nasal, oral) after ruling out contraindications (untreated prostate cancer, severe LUTS, erythrocytosis, untreated OSA, desire for fertility — exogenous T suppresses spermatogenesis). Per the Endocrine Society testosterone guidelines, monitor testosterone, hematocrit, PSA, and symptoms.
18 Endocrine Procedures & Dynamic Testing Procedure
Unlike surgical specialties, endocrinology procedures are almost entirely diagnostic (dynamic hormonal testing) or office-based. You will chart these procedures in detail.
Thyroid Fine Needle Aspiration (FNA)
Ultrasound-guided FNA is the definitive test for thyroid nodules meeting TI-RADS size criteria. The endocrinologist or radiologist inserts a 25–27 gauge needle under US guidance, making multiple passes through the nodule to aspirate cells. Document: informed consent, nodule(s) sampled with size and location, number of passes, rapid on-site evaluation (ROSE) adequacy if performed, specimen handling (slides, ThinPrep, molecular testing), complications (hematoma, vasovagal), and post-procedure instructions.
Dynamic Testing — The Core of Endocrine Diagnostics
Indication: Suspected adrenal insufficiency. Protocol: Baseline cortisol (and ACTH), inject 250 mcg cosyntropin IV or IM, repeat cortisol at 30 and 60 minutes. Normal: peak cortisol ≥ 18 mcg/dL. Documentation: time of injection, baseline and post-stim values, interpretation.
Indications: Diagnosis of diabetes/prediabetes, GDM screening (50 g 1-hr or 75 g 2-hr or 100 g 3-hr), acromegaly (75 g with GH measurement — failure to suppress GH < 1 ng/mL diagnoses acromegaly). Protocol: fasting specimen, ingest glucose load, serial measurements.
1 mg overnight (low-dose): Cushing screen. Dexamethasone 1 mg at 11 PM, cortisol at 8 AM. Normal: cortisol < 1.8 mcg/dL. 2 mg × 2 days (Liddle): Alternative low-dose. 8 mg overnight (high-dose): Suppresses pituitary Cushing but not ectopic ACTH.
Indication: Suspected diabetes insipidus. Protocol: Supervised fluid restriction with hourly weights, urine volume/osm, serum osm/sodium. Continue until urine osm plateaus or weight loss 3–5%. Then administer desmopressin and measure urine osm response. Interpretation: Central DI — urine concentrates > 50% with DDAVP; nephrogenic — minimal response.
Indication: Confirm primary hyperaldosteronism. Saline infusion (2 L NS over 4 hrs) should suppress aldosterone < 5 ng/dL in normal subjects; failure to suppress confirms autonomous production.
Insulin Pump / CGM Management
Endocrinologists download and analyze pump and CGM data every visit. You may assist with data uploads (Dexcom Clarity, LibreView, Tandem t:connect, Omnipod View, Medtronic CareLink). Document pump settings (basal rates, I:C ratios, ISF, active insulin time, target), time in range, average glucose, GMI, coefficient of variation, hypoglycemia events, and any setting changes made today.
Radioactive Iodine (RAI) Therapy
Oral I-131 used for hyperthyroidism (Graves, toxic nodular disease) and post-thyroidectomy remnant ablation in thyroid cancer. Hyperthyroidism dosing typically 10–15 mCi; thyroid cancer ablation 30–150+ mCi based on risk. Pre-treatment: pregnancy test, thyroid function optimization, withdrawal of antithyroid drugs, low-iodine diet. Post-treatment: radiation safety precautions for days to weeks depending on dose, expected progression to hypothyroidism, levothyroxine initiation.
19 Labs, Imaging & Diagnostics Imaging
Key Labs with Reference Ranges
| Lab | Reference Range | Use |
|---|---|---|
| TSH | 0.4–4.5 mIU/L | Thyroid function screening |
| Free T4 | 0.8–1.8 ng/dL | Confirmation of thyroid dysfunction |
| Free T3 | 2.3–4.2 pg/mL | Hyperthyroidism confirmation, T3 toxicosis |
| A1C | < 5.7% normal | Diabetes diagnosis and monitoring |
| Fasting glucose | 70–99 mg/dL | Diabetes screening |
| Calcium (total) | 8.5–10.5 mg/dL | Bone/parathyroid; correct for albumin |
| Ionized calcium | 1.12–1.30 mmol/L | True free calcium |
| PTH (intact) | 15–65 pg/mL | Parathyroid evaluation |
| 25-OH Vitamin D | > 30 ng/mL sufficient | Vitamin D status |
| Cortisol (8 AM) | 5–25 mcg/dL | HPA axis |
| ACTH (8 AM) | 10–60 pg/mL | Distinguish primary vs central adrenal disease |
| Aldosterone | < 21 ng/dL upright | Mineralocorticoid assessment |
| Renin activity | 0.5–4 ng/mL/hr | Denominator of ARR |
| Prolactin | < 25 ng/mL (F), < 20 (M) | Pituitary evaluation |
| IGF-1 | Age/sex adjusted | GH axis |
| Total testosterone (M) | 264–916 ng/dL | Hypogonadism |
CKD-EPI eGFR Stages
G1: eGFR ≥ 90 — normal/high (requires albuminuria or other evidence of kidney damage). G2: 60–89 — mildly decreased. G3a: 45–59 — mildly-moderately decreased. G3b: 30–44 — moderately-severely decreased. G4: 15–29 — severely decreased. G5: < 15 — kidney failure.
Metformin is contraindicated at eGFR < 30 and dose-reduced at 30–45. SGLT2 inhibitors have demonstrated renoprotection at eGFR ≥ 20 per the DAPA-CKD trial.
Imaging Modalities
- Thyroid ultrasound: Size, nodules, TI-RADS features, vascularity, lymph nodes.
- RAIU scan (I-123): Differentiates Graves (diffuse uptake), toxic nodular disease (focal), and thyroiditis (low uptake).
- Pituitary MRI with/without contrast, thin-cut sellar protocol: Microadenoma vs macroadenoma, chiasm compression, cavernous sinus invasion.
- Adrenal CT: Characterize incidentaloma (size, attenuation in HU, washout). Benign adenoma: < 10 HU on non-contrast or absolute washout > 60%.
- Sestamibi parathyroid scan & 4D-CT: Localize parathyroid adenoma pre-op.
- DEXA (DXA): Lumbar spine (L1–L4), femoral neck, total hip, 33% radius. Report includes T-score, Z-score, BMD (g/cm²), and TBS.
- MIBG / DOTATATE PET: Pheochromocytoma/paraganglioma, neuroendocrine tumors.
20 Medications You Must Know Meds
Diabetes Medications
| Class | Generic (Brand) | Mechanism | Key Notes |
|---|---|---|---|
| Biguanide | Metformin (Glucophage) | Decreases hepatic glucose production, improves insulin sensitivity | First-line T2DM. Hold if eGFR < 30. GI side effects, B12 deficiency. No hypoglycemia. |
| GLP-1 RA | Semaglutide (Ozempic, Wegovy), Liraglutide (Victoza, Saxenda), Dulaglutide (Trulicity), Exenatide, Lixisenatide | Incretin mimetic; increases glucose-dependent insulin, decreases glucagon, slows gastric emptying, suppresses appetite | Weight loss, CV and renal benefit. GI side effects. Contraindicated in MTC/MEN2. Pancreatitis warning. |
| Dual GIP/GLP-1 | Tirzepatide (Mounjaro, Zepbound) | Dual incretin agonist | Most potent weight loss and A1C reduction. Weekly SC. |
| SGLT2i | Empagliflozin (Jardiance), Dapagliflozin (Farxiga), Canagliflozin (Invokana), Ertugliflozin | Blocks renal glucose reabsorption | CV, HF, and renal benefit. Risk: genital mycotic infection, DKA (including euglycemic), volume depletion, Fournier gangrene (rare). |
| DPP-4i | Sitagliptin (Januvia), Linagliptin (Tradjenta), Saxagliptin, Alogliptin | Inhibits DPP-4, increasing endogenous incretin | Weight neutral. Minimal hypoglycemia. Pancreatitis signal. Avoid with GLP-1 RA. |
| Sulfonylurea | Glipizide, Glimepiride, Glyburide | Stimulates pancreatic insulin release | Hypoglycemia risk (especially glyburide in elderly), weight gain. Cheap. |
| TZD | Pioglitazone (Actos) | PPAR-gamma agonist; improves insulin sensitivity | Weight gain, fluid retention, heart failure, fracture, bladder cancer signal. |
| Meglitinide | Repaglinide, Nateglinide | Short-acting insulin secretagogue | Preprandial dosing. Hypoglycemia risk. |
| Alpha-glucosidase inhibitor | Acarbose, Miglitol | Delays carbohydrate absorption | GI side effects; rarely used. |
| Insulin — Rapid | Lispro (Humalog), Aspart (Novolog), Glulisine (Apidra), Fast Aspart (Fiasp), Lispro-aabc (Lyumjev) | Bolus / mealtime | Onset 10–15 min, duration 3–5 hr. |
| Insulin — Short | Regular (Humulin R, Novolin R, U-500) | Bolus; IV use in DKA/HHS | Onset 30 min, duration 5–8 hr. |
| Insulin — Intermediate | NPH (Humulin N, Novolin N) | Basal (outdated for T1DM) | Peaks 4–10 hr; hypoglycemia risk. |
| Insulin — Long | Glargine U-100 (Lantus, Basaglar), Glargine U-300 (Toujeo), Detemir (Levemir), Degludec (Tresiba) | Basal | Degludec/glargine U-300 have longest, flattest profile. |
Thyroid Medications
| Class | Generic (Brand) | Use | Notes |
|---|---|---|---|
| Thyroid hormone | Levothyroxine (Synthroid, Levoxyl, Tirosint) | Hypothyroidism replacement | Empty stomach, separate from calcium/iron/PPI. |
| Liothyronine | Cytomel (T3) | Combination therapy, myxedema coma | Short half-life; rarely monotherapy. |
| Desiccated thyroid | Armour Thyroid, NP Thyroid | Patient preference | Variable T3/T4 ratio; not guideline-preferred. |
| Thionamide | Methimazole (Tapazole) | Hyperthyroidism first-line | Agranulocytosis, hepatotoxicity, teratogenic 1st trimester. |
| Thionamide | Propylthiouracil (PTU) | First trimester pregnancy, thyroid storm | Hepatotoxicity (black box), blocks T4→T3 conversion. |
Bone/Mineral Medications
| Class | Generic (Brand) | Mechanism | Notes |
|---|---|---|---|
| Bisphosphonate | Alendronate (Fosamax), Risedronate (Actonel), Ibandronate (Boniva), Zoledronic acid (Reclast) | Inhibits osteoclast-mediated resorption | Upright after oral dose, esophageal irritation, ONJ, atypical femur fracture, flu-like with IV. |
| RANKL inhibitor | Denosumab (Prolia) | Blocks RANK-ligand | SC q6mo. Do NOT interrupt — rebound fracture risk. Hypocalcemia risk. |
| Anabolic (PTH analog) | Teriparatide (Forteo), Abaloparatide (Tymlos) | Intermittent PTH analog stimulates osteoblasts | Daily SC, max 2 years lifetime. |
| Sclerostin inhibitor | Romosozumab (Evenity) | Dual anabolic/antiresorptive | Monthly SC × 12. CV warning. |
| SERM | Raloxifene (Evista) | Selective estrogen receptor modulator | VTE risk. Breast cancer risk reduction. |
| Calcimimetic | Cinacalcet (Sensipar) | Activates CaSR, lowers PTH | Secondary HPT in dialysis, parathyroid carcinoma, medical PHPT. |
Adrenal & Pituitary Medications
| Class | Generic (Brand) | Use | Notes |
|---|---|---|---|
| Glucocorticoid | Hydrocortisone, Prednisone, Dexamethasone | Adrenal insufficiency, stress dosing, immunosuppression | Stress dose education critical. |
| Mineralocorticoid | Fludrocortisone (Florinef) | Primary adrenal insufficiency | Monitor BP, K, edema. |
| Alpha blocker | Phenoxybenzamine, Doxazosin | Pheochromocytoma pre-op | Always before beta-blockade. |
| Aldosterone antagonist | Spironolactone, Eplerenone | Hyperaldosteronism (bilateral), HF | Hyperkalemia, gynecomastia. |
| Cortisol synthesis inhibitor | Ketoconazole, Metyrapone, Osilodrostat, Mitotane | Cushing syndrome medical therapy | Hepatotoxicity (keto); mitotane is adrenolytic. |
| Dopamine agonist | Cabergoline, Bromocriptine | Prolactinoma | Cabergoline preferred; cardiac valve concern at high doses. |
| Somatostatin analog | Octreotide, Lanreotide, Pasireotide | Acromegaly, NETs | Gallstones, hyperglycemia (pasireotide). |
| GH receptor antagonist | Pegvisomant (Somavert) | Acromegaly | Monitor LFTs. |
| Vasopressin analog | Desmopressin (DDAVP) | Central DI | Hyponatremia risk. |
21 Classification Systems
Bethesda — Thyroid Cytopathology
| Category | Description | Malignancy Risk | Management |
|---|---|---|---|
| I | Nondiagnostic / Unsatisfactory | 5–10% | Repeat FNA |
| II | Benign | 0–3% | Clinical follow-up |
| III | AUS/FLUS | 10–30% | Repeat FNA or molecular testing |
| IV | Follicular neoplasm / suspicious | 25–40% | Lobectomy or molecular testing |
| V | Suspicious for malignancy | 50–75% | Surgery |
| VI | Malignant | 97–99% | Total thyroidectomy |
TI-RADS — Thyroid US Risk
| Category | Points | Risk | FNA Threshold |
|---|---|---|---|
| TR1 | 0 | Benign | No FNA |
| TR2 | 2 | Not suspicious | No FNA |
| TR3 | 3 | Mildly suspicious | FNA ≥ 2.5 cm |
| TR4 | 4–6 | Moderately suspicious | FNA ≥ 1.5 cm |
| TR5 | ≥ 7 | Highly suspicious | FNA ≥ 1.0 cm |
WHO DEXA T-Score
| Category | T-Score |
|---|---|
| Normal | ≥ −1.0 |
| Osteopenia | −1.0 to −2.5 |
| Osteoporosis | ≤ −2.5 |
| Severe (established) | ≤ −2.5 + fragility fracture |
ADA Diabetes A1C Targets
| Population | A1C Target |
|---|---|
| Most non-pregnant adults | < 7.0% |
| Young, short duration, low hypoglycemia risk | < 6.5% |
| Older, limited life expectancy, comorbidities | < 8.0% |
| Pregnancy (pre-existing diabetes) | < 6.0–6.5% |
CKD-EPI eGFR Stages
| Stage | eGFR (mL/min/1.73m²) | Description |
|---|---|---|
| G1 | ≥ 90 | Normal/high (with kidney damage) |
| G2 | 60–89 | Mildly decreased |
| G3a | 45–59 | Mild–moderate decrease |
| G3b | 30–44 | Moderate–severe decrease |
| G4 | 15–29 | Severe decrease |
| G5 | < 15 | Kidney failure |
22 Abbreviations Master List
Diagnoses & Axes
Hormones & Labs
Medications & Procedures
23 Physical Exam — The Endocrine Exam
The endocrine exam is subtle and patterned. Unlike vascular or ortho, there are few maneuvers — but the inspection findings are diagnostic when you know what to look for.
Inspection: Neck contour, visible goiter, surgical scars. Palpation: Thyroid size (normal ~15–20 g), symmetry, consistency (soft, firm, rubbery), tenderness, nodules (size, number, mobility). Auscultation: Thyroid bruit (Graves). Lymphadenopathy: Cervical, supraclavicular. Document: "Thyroid: non-enlarged, no palpable nodules, no tenderness, no bruit. No cervical lymphadenopathy."
Inspection: Skin integrity, calluses, fissures, ulcers (location, size, depth, base, surrounding skin), deformities (Charcot, hammertoes), hair, nails. Neuro: 10-g monofilament at 10 sites, vibration (128 Hz tuning fork), ankle reflexes. Vascular: DP and PT pulses, ABI if indicated, capillary refill. Document: "Diabetic foot exam: skin intact without ulceration, no deformity, 10/10 monofilament bilaterally, DP/PT pulses 2+ bilaterally."
Other Key Exam Findings by Diagnosis
- Hypothyroidism: Bradycardia, cool/dry skin, coarse hair, delayed DTR relaxation, periorbital edema, goiter.
- Hyperthyroidism: Tachycardia, warm moist skin, fine tremor, hyperreflexia, lid lag, proptosis (Graves), pretibial myxedema.
- Cushing: Moon facies, buffalo hump, central obesity, purple striae, thin skin with ecchymoses, proximal muscle weakness.
- Addison: Hyperpigmentation (palmar creases, buccal mucosa, scars, areolae), orthostatic hypotension, thin/wasted.
- Acromegaly: Enlarged hands/feet, frontal bossing, prognathism, macroglossia, wide interdental spaces, deep voice, skin tags.
- PCOS: Hirsutism (Ferriman-Gallwey score), acne, acanthosis nigricans, central obesity.
- Hypogonadism (male): Decreased body hair, gynecomastia, small soft testes, loss of muscle mass.
24 Sample HPI Templates
These templates show the kind of documentation expected in an endocrinology clinic. Each is a complete HPI of the type you'll be expected to pattern-match in your first week.
"Mr. [Name] is a 54-year-old male with hypertension, hyperlipidemia, obesity (BMI 34), and newly diagnosed type 2 diabetes mellitus referred for initiation of care. Diabetes was identified 3 weeks ago when A1C returned at 9.2% on routine screening; random glucose at that visit was 278 mg/dL. He reports polyuria, nocturia × 3, polydipsia, and an unintentional 8-lb weight loss over 2 months. He denies blurred vision, numbness, foot ulcers, chest pain, or prior hypoglycemia. His PCP started metformin 500 mg BID with plans to uptitrate. Home fingerstick glucoses over the past week have ranged 180–240 fasting, 250–320 post-prandial. He has a family history of T2DM (mother, on insulin). He does not smoke, drinks 2 beers per weekend, sedentary work. Current medications: lisinopril 20 mg daily, atorvastatin 40 mg daily, metformin 500 mg BID."
"Mrs. [Name] is a 46-year-old female with Hashimoto hypothyroidism (TPO Ab positive, diagnosed 2018) presenting for 6-month follow-up on levothyroxine 88 mcg daily. She reports overall feeling well with resolution of prior cold intolerance and fatigue after the most recent dose increase 6 months ago (from 75 to 88 mcg). Denies palpitations, tremor, anxiety, or insomnia to suggest over-replacement. She takes levothyroxine on an empty stomach 30 minutes before breakfast and separates from her calcium supplement. Today's labs: TSH 2.1 (prior 4.8 six months ago, 7.2 one year ago), free T4 1.3. No interval hospitalizations, new medications, or pregnancies. Menses regular."
"Ms. [Name] is a 38-year-old female referred for evaluation of a right thyroid nodule discovered on carotid ultrasound performed for unrelated dizziness workup. She is asymptomatic — denies neck pain, dysphagia, dysphonia, compressive symptoms, hyper- or hypothyroid symptoms. No personal or family history of thyroid cancer, no history of childhood head/neck radiation. On palpation, a 1.5 cm firm right lower pole nodule is appreciated, without cervical lymphadenopathy. Dedicated thyroid ultrasound obtained last week demonstrates a 1.6 × 1.3 × 1.4 cm solid hypoechoic nodule in the right lower pole with irregular margins and punctate echogenic foci, ACR TI-RADS 5 (7 points). Left lobe and isthmus are unremarkable. TSH today is 1.4 (normal). We will proceed with ultrasound-guided FNA today given TR5 nodule ≥ 1.0 cm."
"Endocrine consult requested for a 22-year-old male with type 1 diabetes admitted overnight with DKA. Patient has had T1DM since age 11, managed on insulin pump (Tandem Control-IQ, Humalog). Presented to the ED with 2 days of nausea, vomiting, and abdominal pain after running out of pump infusion set supplies and missing basal insulin for approximately 36 hours. Admission labs: glucose 512, bicarbonate 8, anion gap 24, pH 7.15, beta-hydroxybutyrate 6.2, potassium 5.1. Currently on insulin drip at 6 units/hr, D5 0.45NS at 150 mL/hr, aggressive potassium replacement. Most recent labs (4 hours ago): glucose 198, gap 14, bicarbonate 14, K 3.9. Plan to transition to SC insulin when gap closes — will resume pump with prior settings (basal 0.8–1.0 U/hr segments, I:C 1:8 breakfast/1:10 lunch and dinner, ISF 1:45, target 110) with 1-hour overlap with drip termination."
"Mrs. [Name] is a 68-year-old postmenopausal female with osteoporosis (T-score −2.8 at femoral neck, −3.1 at lumbar spine on DEXA 2022) presenting for follow-up. She has been on alendronate 70 mg weekly for 3 years. She takes calcium carbonate 600 mg BID and vitamin D3 2000 IU daily. Denies new fractures, back pain, jaw problems, or atypical thigh pain. She tolerates alendronate well when taken per instructions (upright, empty stomach, 30 min before breakfast). No family history of hip fracture. No glucocorticoid use. 25-OH vitamin D today is 38 ng/mL. Repeat DEXA obtained last week: femoral neck T-score −2.5 (improved from −2.8), lumbar spine −2.8 (improved from −3.1). FRAX 10-year major osteoporotic risk 18%, hip 4.2%."
"Ms. [Name] is a 41-year-old female referred by her PCP for evaluation of possible Cushing syndrome. Over the past 12–18 months she has developed a 25-lb weight gain concentrated in the abdomen and face, new-onset hypertension (BP 148/92, started on lisinopril), easy bruising, wide purple abdominal striae, proximal muscle weakness (difficulty rising from a chair), and new prediabetes (A1C 6.2%). She denies exogenous glucocorticoid use (no oral steroids, inhalers, joint injections, or topical steroids). Menses have become irregular. No prior imaging. Initial screening labs obtained by PCP: 24-hour urine free cortisol 187 mcg/24h (ULN 45), late-night salivary cortisol elevated. Today we will obtain a 1 mg overnight dexamethasone suppression test, confirmatory 24-hour UFC, ACTH level, and begin to localize the source."
Endocrinology is a lab-driven, trend-driven specialty. The best scribes learn to read the lab panel before the patient is seen and can anticipate the conversation: an A1C that jumped from 7.2 to 8.6 means the endocrinologist will intensify therapy; a TSH of 8.2 on a levothyroxine-treated patient means a dose increase is coming; a calcium of 11.3 with an intact PTH of 95 means the conversation will pivot toward surgical referral.
Know the medications by both generic and brand name. Know the axes. Understand that "titrate glargine up by 2 units every 3 days until fasting < 130" is a standard algorithm and document it in full. Pay attention to follow-up intervals — they are not arbitrary (6–8 weeks for thyroid, 3 months for diabetes, 6 months for osteoporosis are standard because they match the physiology of the test).
Welcome to endocrinology. The specialty rewards precision. Master the numbers, and the rest follows.
25 References & Sources
Clinical Practice Guidelines
American Diabetes Association. Standards of Care in Diabetes — 2024. Diabetes Care. 2024.
USPSTF. Screening for Prediabetes and Type 2 Diabetes: Recommendation Statement. 2021.
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: The Endocrine System. OpenStax College. CC BY 3.0.
Figure 2: Hypothalamus-Pituitary Complex. OpenStax College. CC BY 3.0.
Figure 3: Thyroid & Parathyroid Glands. OpenStax College. CC BY 3.0.
Figure 4: The Adrenal Glands. OpenStax College. CC BY 3.0.
Figure 5: Diabetes Mechanism. Blausen Medical. CC BY 3.0.
Figure 6: Glucose & Insulin Across the Day. Suckale & Solimena. CC BY 2.0.