Gastroenterology
Every diagnosis, condition, procedure, medication, abbreviation, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 The GI System — Anatomy Essentials
Gastroenterology covers the entire alimentary canal from the upper esophageal sphincter to the anus, plus the accessory digestive organs (liver, gallbladder, biliary tree, and pancreas). A gastroenterologist's clinical domain spans luminal disease (reflux, ulcers, IBD, tumors, bleeding), hepatology (viral hepatitis, cirrhosis, HCC), pancreatobiliary disease (pancreatitis, stones, strictures, cancer), and a heavy procedural workload centered on endoscopy. A scribe has to understand this anatomy because the attending will routinely dictate findings like "pan-ulcerative colitis to the hepatic flexure," "LA grade C esophagitis at the GE junction," or "2 cm sessile polyp in the sigmoid" — and you are expected to chart each of those accurately.
The Upper GI Tract
The esophagus is a 25 cm muscular tube running from the pharynx, behind the trachea and heart, through the diaphragmatic hiatus, and into the stomach at the gastroesophageal (GE) junction, typically 38–40 cm from the incisors. The upper third is skeletal muscle, the middle third mixed, and the lower third smooth muscle. Two functional sphincters matter: the upper esophageal sphincter (UES), which is the cricopharyngeus muscle, and the lower esophageal sphincter (LES), a high-pressure zone at the GE junction that prevents reflux. The Z-line is the squamocolumnar junction between pale esophageal squamous mucosa and salmon-colored gastric columnar mucosa — displacement of the Z-line is how Barrett esophagus is recognized endoscopically.
The stomach is divided into the cardia (just below the GE junction), fundus (the dome above the cardia), body (the main central portion with rugal folds), antrum (the distal portion leading to the pylorus), and pylorus (the muscular outlet into the duodenum). The incisura angularis is the notch on the lesser curvature that marks the junction of body and antrum — a common site for ulcers and gastric cancer. Parietal cells (body/fundus) secrete acid and intrinsic factor; chief cells secrete pepsinogen; G cells (antrum) secrete gastrin.
The Small Intestine
The small intestine is roughly 6 meters long and divided into three segments: the duodenum (C-shaped, ~25 cm, retroperitoneal after the first portion, divided into D1 bulb, D2 descending, D3 horizontal, D4 ascending; the ampulla of Vater drains pancreatic and bile ducts into D2), the jejunum (~2.5 m, proximal, tall villi, prominent plicae circulares), and the ileum (~3.5 m, distal, Peyer patches, terminal ileum absorbs B12 and bile salts and is the classic site of Crohn disease). The ligament of Treitz (at the duodenojejunal flexure) is the anatomic boundary that divides "upper" from "lower" GI bleeding.
The Colon, Rectum & Anus
The colon runs from the ileocecal valve through the cecum (with the appendix), ascending colon, hepatic flexure, transverse colon, splenic flexure, descending colon, sigmoid colon, and into the rectum and anal canal. The ileocecal valve, appendiceal orifice, and triradiate fold are the landmarks the endoscopist uses to confirm a complete colonoscopy to the cecum. The rectosigmoid junction lies ~15 cm from the anal verge; the dentate (pectinate) line divides the rectal columnar mucosa from squamous anoderm and is the boundary between internal (above) and external (below) hemorrhoids.
Liver, Biliary Tree & Pancreas
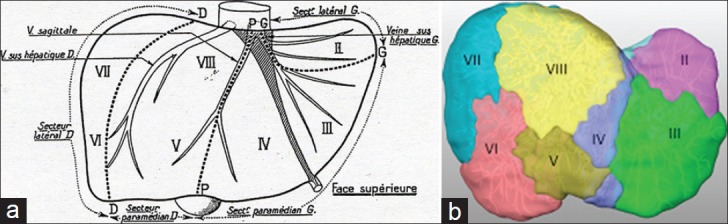
The liver has functional segments I–VIII (Couinaud) based on portal and hepatic venous anatomy. It receives a dual blood supply: ~75% from the portal vein (draining the splanchnic circulation) and ~25% from the hepatic artery. The biliary tree begins as intrahepatic ducts that merge into the right and left hepatic ducts, join as the common hepatic duct, and after the cystic duct joins from the gallbladder, become the common bile duct (CBD), which passes posterior to the duodenum and enters the ampulla of Vater at the major duodenal papilla. The pancreas lies retroperitoneally across L1–L2 with a head (cradled by the duodenum), uncinate process, neck, body, and tail (extending to the splenic hilum). The main pancreatic duct (duct of Wirsung) joins the CBD at the ampulla.
Know these distances cold: incisors to GE junction ~40 cm, pylorus ~55 cm, second part of duodenum ~65–70 cm. During colonoscopy the scope depths you will hear are approximate: rectum 0–15 cm, sigmoid 15–50 cm, descending 50–60 cm, splenic flexure ~60 cm, transverse 60–80 cm, hepatic flexure ~80 cm, ascending 80–90 cm, cecum ~90 cm. When the attending dictates "polyp at 35 cm," that is the sigmoid.
02 Scribe Documentation Framework
GI encounters span clinic new patients, follow-ups, procedure visits, and inpatient consults (GIB, pancreatitis, decompensated cirrhosis). Each has a distinct documentation rhythm, but all use the standard SOAP structure. Your goal as a scribe is to capture the attending's reasoning in real time so the note writes itself by the end of the encounter.
Chief Complaint: Typical GI CCs — "heartburn," "abdominal pain," "diarrhea," "blood in stool," "abnormal LFTs," "screening colonoscopy," "IBD follow-up," "jaundice."
HPI: Use OLDCARTS plus GI-specific elements. For abdominal pain always capture location (RUQ, epigastric, periumbilical, LLQ, diffuse), radiation (to back? shoulder?), relation to meals (postprandial? worse on empty stomach?), nocturnal symptoms, and relief with antacids or bowel movements. For diarrhea document frequency per day, stool consistency (use the Bristol scale), blood/mucus, nocturnal stools (a red flag pointing away from IBS), tenesmus, urgency, and weight loss. For dysphagia distinguish solids only (suggests mechanical obstruction) from solids+liquids (suggests motility disorder), and progressive vs intermittent.
ROS: A targeted ROS should always cover constitutional (weight loss, fever, fatigue), GI (nausea, vomiting, hematemesis, melena, hematochezia, jaundice, pruritus, abdominal pain, bloating, change in bowel habits, heartburn), and extraintestinal features when IBD is on the differential (joint pain, rashes, eye symptoms).
PMHx/PSHx/Meds/Allergies/SHx/FHx: Always capture alcohol use (quantify drinks per day and years), IV drug use and tattoos (hepatitis risk), NSAID use (PUD risk), travel history (infectious diarrhea, hepatitis A), family history of colon cancer or polyps (first-degree relative before 60 alters screening age), IBD family history, and any known liver disease.
Vitals: Pay particular attention to tachycardia and orthostatic BP in bleeding or dehydrated patients, fever in cholangitis or IBD flares, and BMI (relevant for MASLD/GERD/bariatric).
Abdominal Exam: Inspection (distension, scars, caput medusae, spider angiomas), auscultation (bowel sounds — hypoactive vs hyperactive), percussion (tympany, shifting dullness for ascites, Castell sign for splenomegaly), palpation (tenderness by quadrant, guarding, rebound, Murphy sign, McBurney point, hepatomegaly, splenomegaly, masses), and a DRE when appropriate (tone, masses, stool color, occult blood).
Results: CBC, CMP with LFTs, lipase, INR, stool studies, imaging, endoscopy reports. Always document specific values, not "normal."
Organized by problem. A classic GI A/P might look like: "1. GERD — LA Grade B esophagitis on EGD; start PPI BID x 8 weeks, lifestyle modifications, repeat EGD to confirm healing. 2. H. pylori positive — start clarithromycin triple therapy x 14 days, confirm eradication with stool antigen 4 weeks after completion. 3. Iron deficiency anemia — likely multifactorial; start oral iron, recheck in 6 weeks."
When charting a GI HPI, anchor every symptom to a timeline and severity. "Epigastric burning 2–3x weekly after large meals" beats "reflux." "Loose stools 6–8x daily with visible blood and nocturnal awakening" beats "diarrhea." The specificity directly drives the differential and the billing level.
03 GERD & Esophagitis Luminal
Gastroesophageal reflux disease (GERD) is the retrograde movement of gastric contents into the esophagus causing troublesome symptoms or mucosal injury. It is the single most common diagnosis in a GI clinic. The mechanism is failure of the anti-reflux barrier — a hypotensive LES, transient LES relaxations, hiatal hernia, delayed gastric emptying, or increased intra-abdominal pressure (obesity, pregnancy).
Clinical Presentation
Typical symptoms are heartburn (retrosternal burning, often postprandial and worse when supine) and regurgitation. Atypical symptoms include chronic cough, hoarseness, throat clearing, globus, asthma exacerbation, non-cardiac chest pain, and dental erosions. Dysphagia, odynophagia, weight loss, anemia, hematemesis, or melena are alarm features and mandate prompt EGD.
New dysphagia, odynophagia, unintentional weight loss, iron-deficiency anemia, overt GI bleeding (hematemesis, melena), persistent vomiting, family history of upper GI cancer, or age ≥ 60 with new-onset symptoms. These findings override empiric PPI therapy and push straight to endoscopy.
Diagnosis & LA Classification
In patients with typical symptoms and no alarm features, empiric PPI therapy is both diagnostic and therapeutic. Endoscopy is indicated for alarm features, refractory symptoms, or Barrett screening. When esophagitis is seen on EGD, it is graded using the Los Angeles (LA) classification.
| LA Grade | Mucosal Break Description | Clinical Correlation |
|---|---|---|
| A | One or more mucosal breaks ≤ 5 mm, not extending between tops of mucosal folds | Mild; most common finding |
| B | One or more mucosal breaks > 5 mm, not extending between tops of mucosal folds | Moderate |
| C | Mucosal breaks extending between ≥ 2 mucosal folds, involving < 75% of circumference | Severe; higher Barrett risk |
| D | Mucosal breaks involving ≥ 75% of circumference | Severe; high stricture and Barrett risk |
Ambulatory pH monitoring (24-hour pH-impedance or Bravo capsule) is reserved for refractory symptoms, atypical presentations, or pre-anti-reflux surgery evaluation. High-resolution esophageal manometry is performed before fundoplication to exclude achalasia.
Management
ACG clinical guidelines on GERD (2022) are the standard reference. First-line therapy is lifestyle modification (weight loss in overweight patients, head-of-bed elevation, avoiding late meals, trigger food avoidance) plus an 8-week course of once-daily PPI taken 30–60 minutes before breakfast. Step-up to BID dosing or step-down to the lowest effective dose is common. Erosive esophagitis (LA C/D) or Barrett esophagus warrants indefinite PPI therapy. Refractory GERD may warrant anti-reflux surgery (Nissen fundoplication, LINX magnetic sphincter augmentation) or endoscopic therapies (TIF, Stretta).
Always document PPI name, dose, timing (before meals), and duration. "Omeprazole 20 mg daily" is incomplete — "omeprazole 20 mg PO daily 30 minutes before breakfast x 8 weeks" is what belongs in the note. PPI timing matters because they must be taken before meals to bind actively secreting proton pumps.
04 Barrett Esophagus & Esophageal Cancer Oncology
Barrett esophagus (BE) is the replacement of normal squamous esophageal epithelium with specialized intestinal metaplasia (columnar epithelium with goblet cells) in response to chronic acid reflux. It is the only known precursor to esophageal adenocarcinoma (EAC). Risk factors include chronic GERD ≥ 5 years, male sex, white race, age > 50, central obesity, smoking, and family history of BE or EAC.
Diagnosis & Surveillance
Diagnosis requires both endoscopic salmon-colored columnar mucosa extending ≥ 1 cm above the GE junction and histologic confirmation of intestinal metaplasia with goblet cells. Extent is described using the Prague C & M criteria (C = circumferential length, M = maximal length). Dysplasia is graded as: no dysplasia, indefinite for dysplasia, low-grade dysplasia (LGD), or high-grade dysplasia (HGD).
ACG Barrett esophagus guidelines (2022) recommend surveillance EGD every 3–5 years for non-dysplastic BE, every 6–12 months for LGD (or endoscopic eradication), and endoscopic eradication therapy for HGD or intramucosal cancer using radiofrequency ablation (RFA), endoscopic mucosal resection (EMR), or endoscopic submucosal dissection (ESD).
Esophageal Cancer
Two histologies dominate: adenocarcinoma (distal esophagus/GE junction, arises from Barrett, rising incidence in the West, linked to GERD/obesity) and squamous cell carcinoma (mid-esophagus, linked to smoking, alcohol, hot beverages, achalasia, tylosis; higher incidence globally). Presentation is classic: progressive solid-food dysphagia with weight loss, often late in disease. Diagnosis is by EGD with biopsy; staging uses CT chest/abdomen, PET-CT, and endoscopic ultrasound (EUS) for T and N staging. Management depends on stage: endoscopic resection for T1a, esophagectomy for localized disease, and chemoradiation +/- surgery for locally advanced disease.
05 Eosinophilic Esophagitis & Motility Disorders Luminal
Eosinophilic Esophagitis (EoE)
EoE is a chronic immune/antigen-mediated disease characterized by symptoms of esophageal dysfunction and histologic evidence of eosinophil-predominant inflammation (≥ 15 eosinophils per high-power field on biopsy). It is increasingly common, classically in young males with atopy (asthma, allergic rhinitis, food allergies, eczema). Presentation: solid-food dysphagia, food impaction, chest pain, and heartburn unresponsive to PPI. Endoscopy shows rings ("feline esophagus" or trachealization), linear furrows, white exudates, edema, and strictures — captured in the EREFS score. Diagnosis requires multiple biopsies from proximal and distal esophagus. Treatment: PPI, swallowed topical steroids (budesonide or fluticasone), dietary elimination (6-food, 4-food, or targeted), dupilumab (IL-4Rα mAb, FDA-approved for EoE), and dilation for strictures.
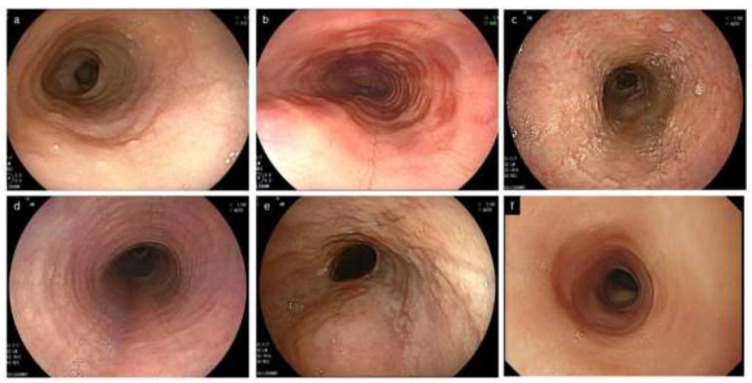
Achalasia
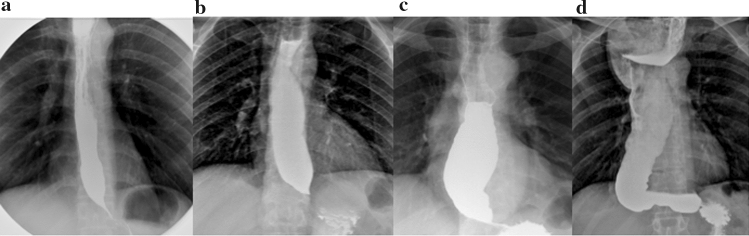
Achalasia is a primary motility disorder characterized by failure of LES relaxation and absent esophageal peristalsis due to loss of inhibitory neurons in the myenteric plexus. Presentation: progressive dysphagia to both solids and liquids, regurgitation of undigested food, weight loss, and nocturnal cough. Barium swallow shows the classic "bird's beak" tapering at the GE junction. High-resolution manometry is the gold standard and subtypes achalasia using the Chicago Classification (Type I = classic, Type II = with panesophageal pressurization, best prognosis, Type III = spastic). Treatment options include pneumatic dilation, peroral endoscopic myotomy (POEM), surgical Heller myotomy with partial fundoplication, or botulinum toxin injection for poor surgical candidates.
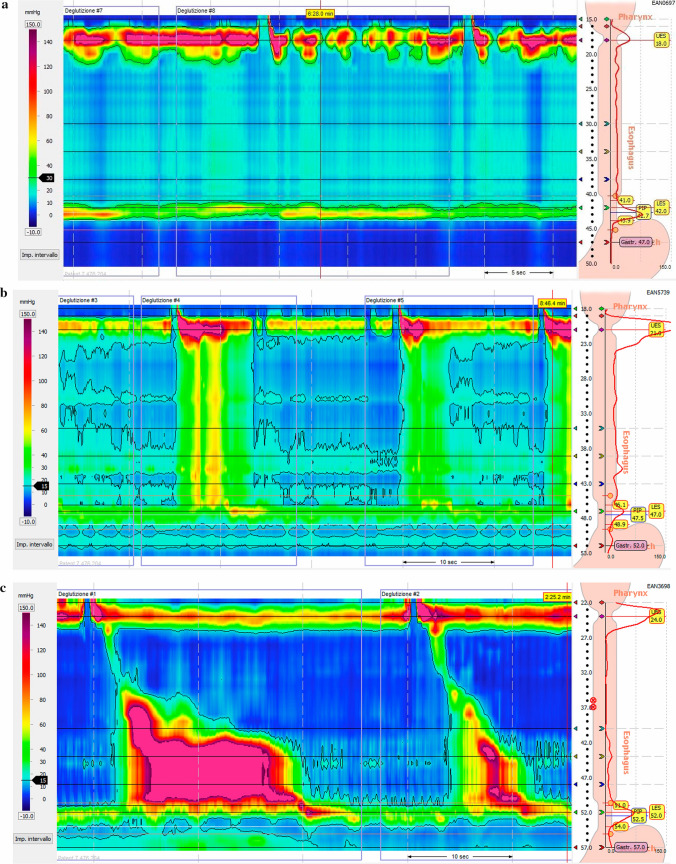
Other Motility Disorders
Distal esophageal spasm, jackhammer esophagus, ineffective esophageal motility, gastroparesis (documented with gastric emptying scintigraphy — > 10% retention at 4 hours is diagnostic), and functional dysphagia round out the motility differential. Gastroparesis is classically seen in long-standing diabetes and presents with early satiety, postprandial fullness, nausea, vomiting of undigested food, and bloating.
06 Peptic Ulcer Disease, H. pylori & Gastritis Luminal
Peptic ulcer disease (PUD) encompasses gastric and duodenal ulcers — mucosal defects extending through the muscularis mucosae. The two dominant causes are Helicobacter pylori infection and NSAID use. Other causes include Zollinger-Ellison syndrome, stress ulcers (in the critically ill), corticosteroids (synergistic with NSAIDs), smoking, and alcohol.
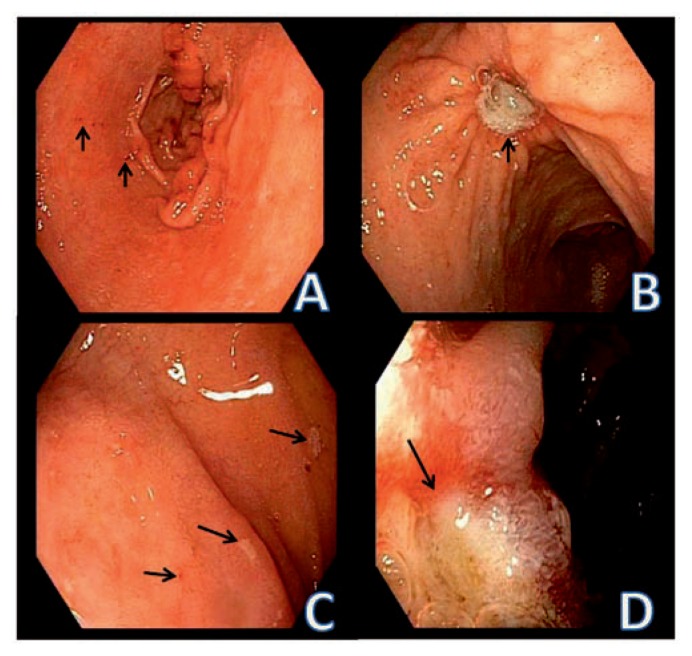
Clinical Presentation
Classic but inconsistent patterns: duodenal ulcers produce epigastric burning that is relieved by food and recurs 2–3 hours postprandial or at night; gastric ulcers produce pain that is worsened by food. Many ulcers are silent until they bleed or perforate. Complications include GI bleeding (most common), perforation (acute severe pain, peritonitis, free air on imaging), penetration (into the pancreas, causing pancreatitis-like pain), and gastric outlet obstruction from pyloric channel scarring.
H. pylori
H. pylori is a gram-negative spiral bacterium that colonizes the gastric mucosa, causing chronic gastritis, PUD, MALT lymphoma, and gastric adenocarcinoma. Testing options include stool antigen (sensitive, specific, cheap, confirms eradication), urea breath test (similar accuracy, requires fasting and PPI hold), serology (cannot distinguish active from prior infection, rarely used now), and histology with rapid urease (CLO) test on EGD biopsies. PPI must be stopped 2 weeks before non-serology testing to avoid false negatives.
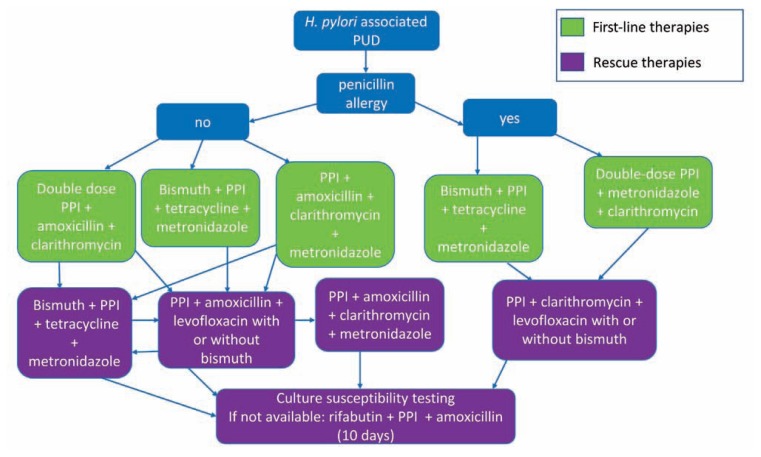
ACG H. pylori treatment guidelines (2024) now favor bismuth quadruple therapy (PPI + bismuth + tetracycline + metronidazole x 14 days) as first-line in most regions because of rising clarithromycin resistance. Clarithromycin-based triple therapy (PPI + clarithromycin + amoxicillin) is acceptable only where local resistance is < 15%. Vonoprazan-based dual or triple therapy is an FDA-approved alternative. Eradication must be confirmed with stool antigen or urea breath test ≥ 4 weeks after completing therapy and after holding PPI for 2 weeks.
Gastritis
Gastritis is histologic inflammation of the gastric mucosa. Patterns include H. pylori gastritis (most common), autoimmune atrophic gastritis (body-predominant, anti-parietal cell antibodies, pernicious anemia, increased risk of type 1 gastric NETs and cancer), reactive/chemical gastropathy (NSAIDs, bile reflux), and stress gastritis (ICU patients). Erosive gastritis differs from ulcer by depth — erosions do not penetrate the muscularis mucosae.
07 Gastric Cancer & Gastric Neoplasms Oncology
Gastric adenocarcinoma accounts for ~95% of gastric cancers. Two histologic types: intestinal type (well-differentiated, glandular, linked to H. pylori chronic atrophic gastritis, follows Correa cascade: normal → atrophic gastritis → intestinal metaplasia → dysplasia → carcinoma) and diffuse type (poorly differentiated, signet-ring cells, younger patients, linked to CDH1 mutations, includes the infiltrative linitis plastica presentation). Risk factors: H. pylori, smoking, diet high in salted/smoked foods, pernicious anemia, family history, blood type A. Symptoms are vague (dyspepsia, early satiety, weight loss) until advanced. Diagnosis is by EGD with biopsy; staging uses CT, EUS, and diagnostic laparoscopy. Other gastric neoplasms include MALT lymphoma (often H. pylori-driven, may regress with eradication), GIST (gastrointestinal stromal tumor, c-KIT/CD117 positive, treated with imatinib), and neuroendocrine tumors (NETs).
08 Functional GI: IBS, Functional Dyspepsia, Constipation Functional
Functional GI disorders are now called disorders of gut-brain interaction (DGBIs). They are defined by the Rome IV criteria and diagnosed on symptoms after exclusion of organic disease. They are real, common, and often debilitating — the "functional" label does not mean the symptoms are imagined.
Irritable Bowel Syndrome (IBS)
ACG IBS monograph (2021) is the standard citation. Rome IV criteria for IBS: recurrent abdominal pain on average ≥ 1 day/week in the last 3 months, with symptom onset ≥ 6 months earlier, associated with ≥ 2 of the following: related to defecation, associated with change in stool frequency, associated with change in stool form.
| IBS Subtype | Criteria | Typical Therapies |
|---|---|---|
| IBS-C (constipation-predominant) | > 25% hard stools (Bristol 1–2), < 25% loose | Soluble fiber, PEG, linaclotide, plecanatide, lubiprostone, tenapanor |
| IBS-D (diarrhea-predominant) | > 25% loose stools (Bristol 6–7), < 25% hard | Loperamide, rifaximin, eluxadoline, bile acid sequestrants, low-FODMAP diet |
| IBS-M (mixed) | > 25% hard AND > 25% loose | Symptom-directed; antispasmodics, TCAs, SSRIs |
| IBS-U (unclassified) | Does not fit other subtypes | Symptom-directed |
Red flags that mandate workup beyond IBS include age > 50 at onset, weight loss, nocturnal symptoms, rectal bleeding, anemia, family history of colon cancer or IBD, and short symptom duration. Basic workup includes CBC, CRP, celiac serology (tTG-IgA), and in IBS-D a fecal calprotectin to exclude IBD.
Functional Dyspepsia
Defined by bothersome postprandial fullness, early satiety, epigastric pain, or epigastric burning, for ≥ 3 months with onset ≥ 6 months earlier, in the absence of structural disease that would explain the symptoms. Subtypes: postprandial distress syndrome (PDS) and epigastric pain syndrome (EPS). Management: H. pylori test and treat, PPI trial, TCAs, prokinetics, and psychological therapies.
Chronic Constipation
Types: normal-transit (most common), slow-transit, outlet dysfunction (dyssynergic defecation), and IBS-C. Workup: history, DRE (for outlet dysfunction — paradoxical contraction, anismus), thyroid, calcium, colonoscopy if alarm features, anorectal manometry and balloon expulsion for suspected outlet dysfunction, colonic transit study (Sitzmark) for slow-transit. Treatment ladder: fiber → osmotic laxatives (PEG) → stimulant laxatives → secretagogues (linaclotide, plecanatide, lubiprostone) → prokinetics (prucalopride) → biofeedback for dyssynergia.
Type 1: Separate hard lumps, like nuts (very constipated)
Type 2: Sausage-shaped but lumpy (slightly constipated)
Type 3: Like a sausage with cracks on the surface (normal)
Type 4: Like a smooth, soft sausage or snake (ideal)
Type 5: Soft blobs with clear-cut edges (lacking fiber)
Type 6: Mushy with ragged edges (mild diarrhea)
Type 7: Watery, entirely liquid, no solid pieces (severe diarrhea)
09 Inflammatory Bowel Disease (UC & Crohn) Inflammatory
Inflammatory bowel disease (IBD) encompasses ulcerative colitis (UC) and Crohn disease (CD). Both are chronic relapsing immune-mediated conditions with bimodal age distribution (peak 15–35, second peak 50–70), increased risk with family history, smoking (protective in UC, worsens CD), and NSAID use. ACG UC guideline (2019) and ACG Crohn disease guideline (2018) are the core management references.
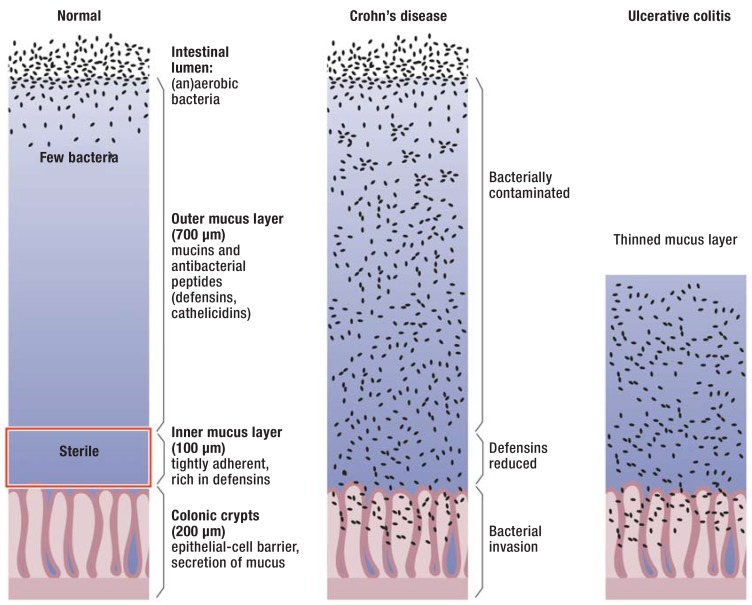
Ulcerative Colitis
UC involves continuous inflammation limited to the colonic mucosa, beginning in the rectum and extending proximally in a contiguous pattern. Extent is classified by the Montreal classification: E1 proctitis, E2 left-sided (up to splenic flexure), E3 extensive (beyond splenic flexure, including pancolitis). Presentation: bloody diarrhea, urgency, tenesmus, abdominal cramping, and in severe cases fever, tachycardia, anemia, weight loss. Labs: elevated CRP, ESR, fecal calprotectin; anemia; hypoalbuminemia. Colonoscopy shows continuous, circumferential erythema, loss of vascular pattern, friability, erosions, ulcerations.
Crohn Disease
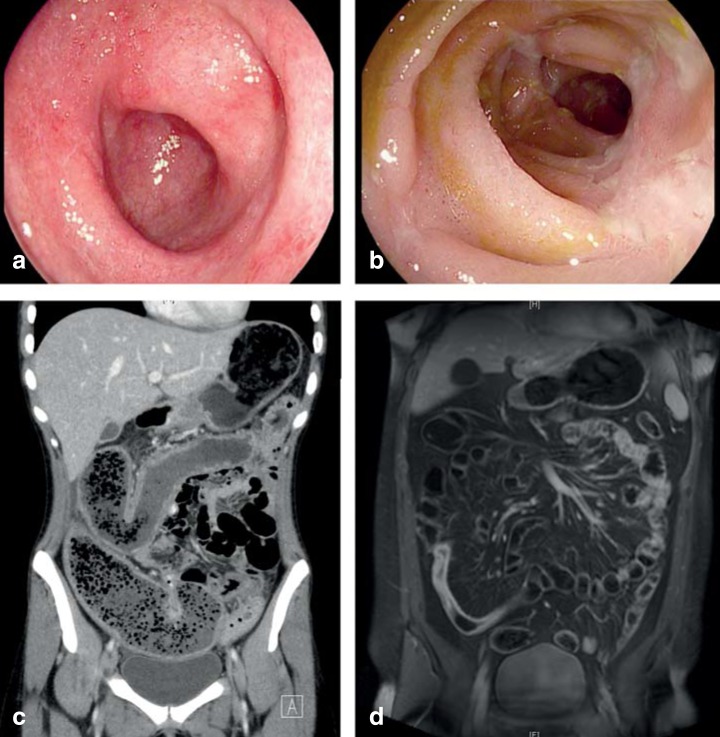
CD can involve any part of the GI tract from mouth to anus, classically the terminal ileum. Inflammation is transmural, discontinuous (skip lesions), and often includes granulomas histologically. Montreal classification for CD captures age at diagnosis (A), location (L1 ileal, L2 colonic, L3 ileocolonic, L4 upper GI, +/- perianal), and behavior (B1 non-stricturing non-penetrating, B2 stricturing, B3 penetrating/fistulizing). Presentation: abdominal pain (often RLQ), chronic diarrhea (may or may not be bloody), weight loss, fatigue, fevers, perianal disease (fistulas, abscesses, fissures), extraintestinal manifestations (peripheral arthritis, sacroiliitis, ankylosing spondylitis, erythema nodosum, pyoderma gangrenosum, uveitis, episcleritis, primary sclerosing cholangitis).
Severity Scoring
Four components, each 0–3: (1) stool frequency, (2) rectal bleeding, (3) physician global assessment, (4) endoscopic findings (0 normal, 1 mild — erythema/decreased vascular pattern, 2 moderate — marked erythema/absent vascular pattern/friability/erosions, 3 severe — spontaneous bleeding/ulceration).
Remission < 3, mild 3–5, moderate 6–10, severe 11–12. The partial Mayo (without endoscopy) is used clinically between scopes.
Five components summed: (1) general well-being 0–4, (2) abdominal pain 0–3, (3) number of liquid stools per day (count), (4) abdominal mass 0–3, (5) complications (each = 1: arthralgia, uveitis, erythema nodosum, aphthous ulcers, pyoderma gangrenosum, anal fissure, new fistula, abscess).
Remission < 5, mild 5–7, moderate 8–16, severe > 16.
Management Overview
Therapy is now stratified by severity and disease phenotype, with an emphasis on early effective therapy and treat-to-target strategies (clinical remission, biochemical normalization, endoscopic healing). Classes used:
| Class | Examples | Primary Use |
|---|---|---|
| 5-ASA (aminosalicylates) | Mesalamine (Asacol, Lialda, Pentasa, Apriso), sulfasalazine | Mild-moderate UC induction & maintenance; little role in CD |
| Corticosteroids | Prednisone, IV methylpred, budesonide (Entocort, Uceris) | Induction only — never maintenance |
| Immunomodulators | Azathioprine, 6-MP, methotrexate | Steroid-sparing maintenance; check TPMT before thiopurines |
| Anti-TNF biologics | Infliximab (Remicade), adalimumab (Humira), certolizumab (Cimzia), golimumab (Simponi) | Moderate-severe UC and CD, fistulizing CD |
| Anti-integrin | Vedolizumab (Entyvio) | Gut-selective, favorable safety; UC and CD |
| Anti-IL-12/23 | Ustekinumab (Stelara) | UC and CD |
| Anti-IL-23 (p19) | Risankizumab (Skyrizi), mirikizumab (Omvoh) | Moderate-severe CD (risankizumab) and UC (mirikizumab) |
| JAK inhibitors | Tofacitinib (Xeljanz), upadacitinib (Rinvoq) | Moderate-severe UC (both) and CD (upadacitinib) |
| S1P modulators | Ozanimod (Zeposia), etrasimod (Velsipity) | Moderate-severe UC |
Surveillance colonoscopy for dysplasia begins 8 years after diagnosis of UC or Crohn colitis, with chromoendoscopy or high-definition white-light and targeted biopsies. Acute severe UC (Truelove-Witts criteria: ≥ 6 bloody stools/day plus any of HR > 90, temp > 37.8, Hb < 10.5, ESR > 30) is a medical emergency requiring hospitalization, IV corticosteroids, VTE prophylaxis, and early surgical consultation.
When charting IBD visits always document: disease type and Montreal classification, current medications with doses and duration, last colonoscopy date and findings, fecal calprotectin trend, CRP, hemoglobin, albumin, steroid exposure in the last year, any extraintestinal manifestations, vaccination status (live vaccines are contraindicated on biologics), and TB/hepatitis B screening before biologics.
10 Celiac Disease & Microscopic Colitis Inflammatory
Celiac Disease
Celiac disease is an autoimmune enteropathy triggered by gluten (wheat, barley, rye) in genetically susceptible individuals (HLA-DQ2 or HLA-DQ8). It produces villous atrophy in the small intestine, malabsorption, and a wide spectrum of intestinal and extraintestinal manifestations. Classic presentation is chronic diarrhea, steatorrhea, weight loss, bloating, and iron-deficiency anemia, but many patients present atypically with only anemia, osteoporosis, transaminitis, infertility, dermatitis herpetiformis, or neurologic symptoms.
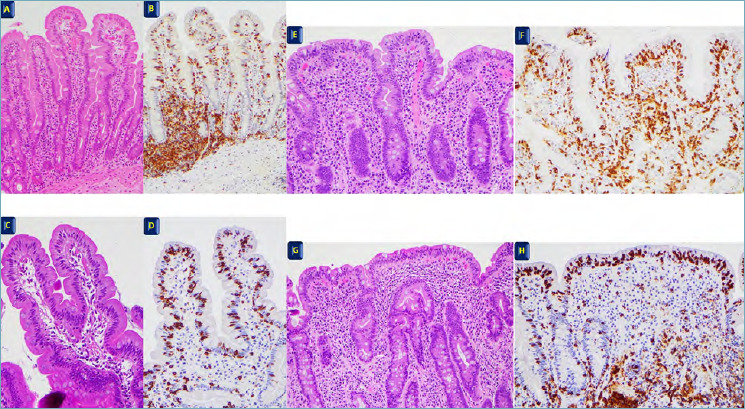
Diagnosis requires serology and confirmatory biopsy while on a gluten-containing diet. First-line serology is tTG-IgA (tissue transglutaminase IgA) with total IgA level (to exclude selective IgA deficiency, which is 10x more common in celiac patients). Confirmatory EGD with 4–6 duodenal biopsies (including bulb) shows intraepithelial lymphocytosis, crypt hyperplasia, and villous atrophy (graded by the Marsh classification, 0–3c). Treatment is a strict lifelong gluten-free diet; follow-up includes tTG trend, DEXA, and iron/B12/folate/vitamin D monitoring.
Microscopic Colitis
Microscopic colitis (collagenous and lymphocytic subtypes) presents with chronic watery non-bloody diarrhea, often in middle-aged to older women. Colonoscopy is macroscopically normal; diagnosis is made on random biopsies showing either a thickened subepithelial collagen band (collagenous) or increased intraepithelial lymphocytes (lymphocytic). Associations include NSAIDs, PPIs, SSRIs, celiac disease, and autoimmune disease. Treatment: stop offending medications and use budesonide 9 mg daily as first-line.
11 Diverticular Disease & Diverticulitis Luminal
Diverticulosis is the presence of diverticula (outpouchings of mucosa and submucosa through the muscularis propria at sites of vasa recta penetration), overwhelmingly in the sigmoid colon in Western populations. It is common (> 50% of adults over 60) and usually asymptomatic, but complications include diverticular bleeding (painless hematochezia, most common cause of lower GI bleeding in older adults) and diverticulitis.
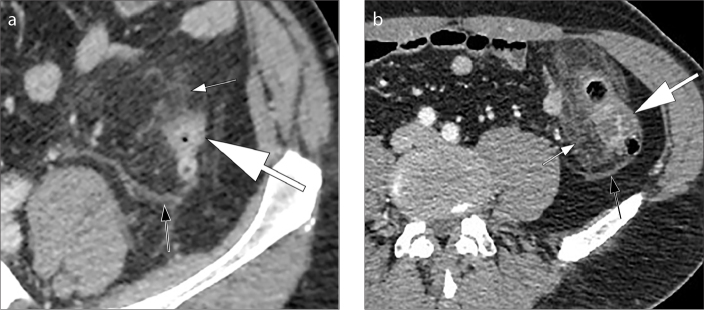
Acute diverticulitis is inflammation/micro-perforation of a diverticulum producing LLQ pain, fever, leukocytosis, and sometimes a palpable mass. CT with IV/PO contrast is the imaging of choice and classifies severity using the Hinchey classification:
| Hinchey Stage | Finding | Management |
|---|---|---|
| Ia | Phlegmon (pericolic inflammation) | Outpatient or inpatient antibiotics; many uncomplicated cases no longer require antibiotics |
| Ib | Pericolic abscess < 4 cm | IV antibiotics +/- percutaneous drainage |
| II | Pelvic/retroperitoneal abscess ≥ 4 cm | IV antibiotics + percutaneous drainage |
| III | Purulent peritonitis | Urgent surgery (laparoscopic lavage or resection) |
| IV | Feculent peritonitis | Emergency Hartmann procedure |
Outpatient management of uncomplicated disease is appropriate for reliable patients without high-risk features; recent guidelines support selective rather than routine antibiotic use in uncomplicated diverticulitis. Colonoscopy is recommended 6–8 weeks after resolution of a first episode if the patient has not had a recent high-quality colonoscopy, to exclude underlying malignancy.
12 Colorectal Cancer, Polyps & Screening Oncology
Colorectal cancer (CRC) is the third most common cancer and second leading cause of cancer death in the US. Most CRC arises from adenomatous polyps via the adenoma-carcinoma sequence (APC → KRAS → DCC → p53) or from sessile serrated lesions via the serrated pathway. Risk factors: age, family history (first-degree relative < 60), hereditary syndromes (Lynch, FAP, MUTYH), IBD, obesity, smoking, red/processed meat, alcohol.
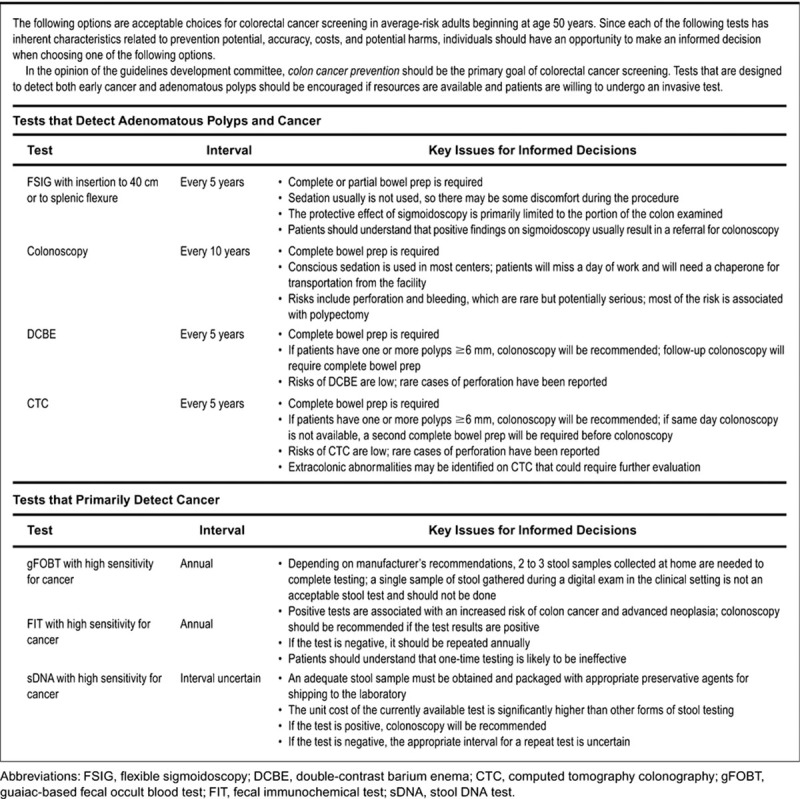
Screening
USPSTF colorectal cancer screening (2021) now recommends starting screening at age 45 for average-risk adults and continuing through age 75; ages 76–85 are individualized. ACG CRC screening guidelines (2021) align with the 45 start age. Options include:
| Test | Interval | Notes |
|---|---|---|
| Colonoscopy | Every 10 years | Gold standard; diagnostic and therapeutic |
| FIT (fecal immunochemical test) | Annually | Stool-based, inexpensive, high compliance |
| Multi-target stool DNA (Cologuard) | Every 3 years | FIT + DNA markers; higher sensitivity, more false positives |
| CT colonography | Every 5 years | Positive finding → colonoscopy for removal |
| Flexible sigmoidoscopy | Every 5–10 years | Uncommon in the US now |
Polyps
Polyp types and significance: tubular adenoma (most common, premalignant, risk increases with size > 10 mm, villous architecture, high-grade dysplasia), tubulovillous and villous adenomas (higher malignant potential), sessile serrated lesion (SSL) (right-sided, flat, easily missed, premalignant via serrated pathway), traditional serrated adenoma, hyperplastic (usually benign, distal), and hamartomatous (juvenile, Peutz-Jeghers).
Post-polypectomy surveillance intervals follow USMSTF 2020 recommendations: 10 years for 1–2 small (< 10 mm) tubular adenomas, 7–10 years for 3–4 small adenomas, 3–5 years for 5–10 adenomas, 3 years for advanced adenomas (≥ 10 mm, high-grade dysplasia, villous), and 1 year for > 10 adenomas or piecemeal resection of a large lesion.
Hereditary CRC Syndromes
Lynch syndrome (HNPCC) — mismatch repair gene mutations (MLH1, MSH2, MSH6, PMS2, EPCAM), associated with early-onset CRC, endometrial, ovarian, urothelial, and small bowel cancer. Familial adenomatous polyposis (FAP) — APC mutation, hundreds to thousands of adenomas, 100% CRC by age 40 without colectomy. MUTYH-associated polyposis — autosomal recessive, attenuated polyposis. Peutz-Jeghers, juvenile polyposis, and Cowden are hamartomatous polyposis syndromes.
13 Hemorrhoids & Anorectal Disease Luminal
Hemorrhoids are dilated submucosal vascular cushions in the anal canal. Internal hemorrhoids arise above the dentate line, are covered by columnar/transitional epithelium, are usually painless, and present with bright red blood per rectum (BRBPR) on the tissue or coating the stool and with prolapse. They are graded:
| Grade | Description |
|---|---|
| I | Protrude into the lumen, may bleed, do not prolapse |
| II | Prolapse with straining, reduce spontaneously |
| III | Prolapse with straining, require manual reduction |
| IV | Permanently prolapsed, cannot be reduced |
External hemorrhoids arise below the dentate line, are covered by anoderm (squamous, innervated), and are painful when thrombosed. Other anorectal diagnoses: anal fissure (painful tear, often posterior midline, from hard stool; treatment is fiber, sitz baths, topical nifedipine or nitroglycerin, botulinum toxin, lateral internal sphincterotomy for refractory cases), perianal abscess/fistula (often requiring I&D; Crohn-related fistulas managed with biologics and seton placement), and pruritus ani.
14 GI Bleeding (UGIB & LGIB) Emergency
Upper GI bleeding (UGIB) is bleeding from a source proximal to the ligament of Treitz; lower GI bleeding (LGIB) is from distal. Presentation varies: hematemesis and coffee-ground emesis are UGIB; melena (black tarry stool from digested blood) is usually UGIB but can occur with right colon bleeding; hematochezia (bright red blood) is usually LGIB but massive UGIB can present with hematochezia and hemodynamic instability.
UGIB Causes & Management
Most common causes: peptic ulcer disease (most common), esophageal/gastric varices, Mallory-Weiss tear, erosive esophagitis/gastritis, Dieulafoy lesion, angiodysplasia, malignancy. Initial management is resuscitation first: two large-bore IVs, type and cross, restrictive transfusion (Hb target 7 g/dL in most, 8 in cardiac patients), IV PPI (pantoprazole 80 mg bolus then 8 mg/hr or intermittent BID), correct coagulopathy, IV octreotide and ceftriaxone if variceal bleeding suspected (cirrhotic patient), and EGD within 24 hours for most stable patients, sooner for hemodynamic instability.
| Class | Description | Rebleed Risk | Intervention |
|---|---|---|---|
| Ia | Active arterial (spurting) bleeding | ~55% | Endoscopic therapy (epi + thermal or clip) |
| Ib | Active oozing bleeding | ~55% | Endoscopic therapy |
| IIa | Non-bleeding visible vessel | ~43% | Endoscopic therapy |
| IIb | Adherent clot | ~22% | Remove clot, treat underlying lesion |
| IIc | Flat pigmented spot | ~10% | Medical therapy alone |
| III | Clean ulcer base | ~5% | Medical therapy alone |
Variceal bleeding in cirrhosis carries a mortality up to 20% per episode. Endoscopic management is band ligation for esophageal varices and cyanoacrylate glue injection for gastric varices. Rescue therapies for uncontrollable bleeding include balloon tamponade (Sengstaken-Blakemore, Minnesota tube), self-expanding esophageal stents, and transjugular intrahepatic portosystemic shunt (TIPS). Secondary prophylaxis after a variceal bleed is non-selective beta-blocker (nadolol, propranolol, or carvedilol) plus serial band ligation.
LGIB Causes & Management
Most common causes: diverticular bleeding (most common, painless, often self-limited), angiodysplasia, ischemic colitis, hemorrhoids, neoplasia, post-polypectomy bleeding, IBD, radiation proctitis. Initial workup: resuscitate, rule out UGIB (NG lavage or upper endoscopy if brisk), then colonoscopy after rapid bowel prep. CT angiography is increasingly used for brisk bleeding to localize before intervention, and interventional radiology embolization is an option for unstable patients. Tagged RBC scan can detect slow bleeding but localization is imprecise.
15 Acute & Chronic Pancreatitis Panc/Bil/Hep
Acute Pancreatitis
Acute pancreatitis diagnosis requires 2 of 3: (1) characteristic abdominal pain (epigastric, radiating to the back, constant), (2) lipase or amylase > 3x upper limit of normal, (3) characteristic findings on imaging. Most common etiologies: gallstones and alcohol (together ~80%).
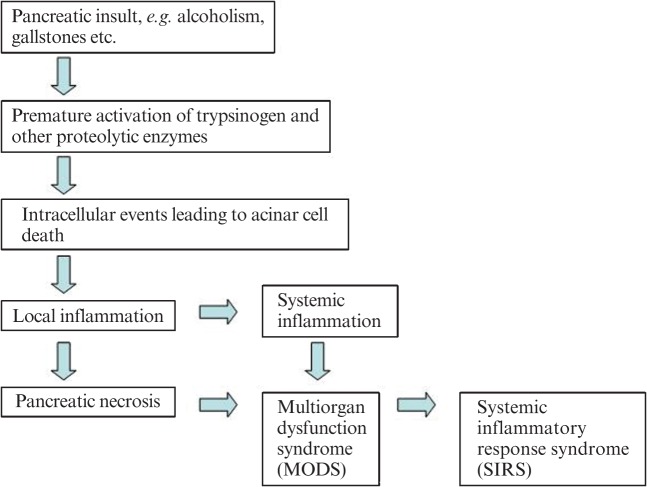
Other causes: hypertriglyceridemia (> 1000 mg/dL), hypercalcemia, post-ERCP, medications (azathioprine, thiazides, valproate, GLP-1 agonists rarely), autoimmune, trauma, viral (mumps, coxsackie), genetic (PRSS1, CFTR, SPINK1), and idiopathic. The mnemonic I GET SMASHED captures most etiologies.
AGA acute pancreatitis guidelines emphasize early aggressive (but not excessive) fluid resuscitation with lactated Ringer's, early enteral nutrition within 24–72 hours when tolerated, avoidance of prophylactic antibiotics in sterile necrosis, and urgent ERCP only for concurrent cholangitis or persistent biliary obstruction. Same-admission cholecystectomy is recommended for mild gallstone pancreatitis.
Mild: No organ failure, no local or systemic complications.
Moderately severe: Transient organ failure (< 48 hours) and/or local complications (peripancreatic fluid collection, pseudocyst, necrosis, walled-off necrosis) or exacerbation of comorbid disease.
Severe: Persistent organ failure (> 48 hours, using modified Marshall score for respiratory, cardiovascular, renal).
On admission: Age > 70, WBC > 18,000, glucose > 220, AST > 250, LDH > 400.
At 48 hours: Hct drop > 10%, BUN rise > 2, Ca < 8, base deficit > 5, fluid sequestration > 4 L, pO2 (not in gallstone set).
Mortality: 0–2 criteria ~1%, 3–4 ~15%, 5–6 ~40%, ≥ 7 ~100%. Non-gallstone Ranson uses slightly different thresholds and includes pO2 < 60.
Other severity tools include BISAP (Bedside Index for Severity in Acute Pancreatitis, 0–5 based on BUN, altered mental status, SIRS, age, pleural effusion) and APACHE II. Complications: peripancreatic fluid collection, pseudocyst, acute necrotic collection, walled-off necrosis (WON > 4 weeks), infected necrosis (requires step-up drainage), pancreatic ascites, splenic vein thrombosis, and disconnected pancreatic duct syndrome.
Chronic Pancreatitis
Chronic pancreatitis is irreversible fibrosis of the pancreas causing chronic abdominal pain, exocrine insufficiency (steatorrhea, fat-soluble vitamin deficiency), and endocrine insufficiency (diabetes, "type 3c"). Alcohol is the most common cause in adults; other causes include smoking, genetic (PRSS1 hereditary pancreatitis, CFTR), autoimmune (IgG4-related), obstructive, and idiopathic. Imaging shows calcifications, ductal dilation with strictures and stones (chain-of-lakes). Management: alcohol cessation and smoking cessation (critical), pancreatic enzyme replacement therapy (PERT) for steatorrhea, fat-soluble vitamins, pain management (avoiding chronic opioids), celiac plexus block, ERCP for stones/strictures, and surgery (Frey, Puestow, Whipple) in selected patients.
16 Pancreatic Cancer & Biliary Disease Panc/Bil/Hep
Pancreatic Adenocarcinoma
Pancreatic ductal adenocarcinoma (PDAC) is the most lethal common cancer in the US. Risk factors: smoking, family history, hereditary syndromes (BRCA1/2, Lynch, PRSS1, Peutz-Jeghers), chronic pancreatitis, long-standing diabetes, obesity.
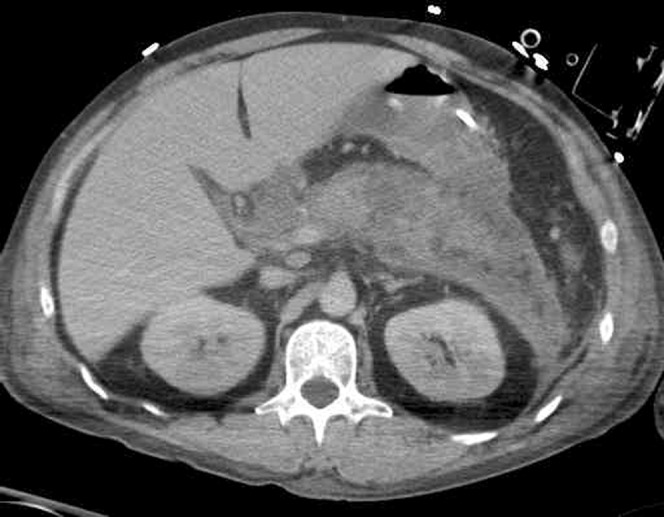
Presentation depends on location: head tumors — painless obstructive jaundice, weight loss, steatorrhea, and pruritus (Courvoisier sign: palpable, non-tender gallbladder with painless jaundice); body/tail tumors — vague abdominal/back pain and late presentation. Diagnosis by contrast-enhanced pancreatic protocol CT and EUS with FNA biopsy. CA 19-9 is used as a tumor marker (not for screening; can be falsely elevated by cholestasis). Management: Whipple (pancreaticoduodenectomy) for resectable head tumors, distal pancreatectomy for body/tail, neoadjuvant chemotherapy (FOLFIRINOX or gemcitabine/nab-paclitaxel) for borderline resectable/locally advanced, palliative chemotherapy for metastatic.
Pancreatic Cystic Lesions
Common cystic lesions include IPMN (intraductal papillary mucinous neoplasm, main duct or branch duct, premalignant), MCN (mucinous cystic neoplasm, women, body/tail, premalignant), SCA (serous cystadenoma, benign microcystic), and pseudocysts. The Fukuoka guidelines stratify IPMN risk using "worrisome features" and "high-risk stigmata" to decide between surveillance and resection.
Gallstones & Biliary Disease
Cholelithiasis is common and usually asymptomatic. Symptomatic disease includes:
| Diagnosis | Presentation | Key Finding | Treatment |
|---|---|---|---|
| Biliary colic | Postprandial RUQ pain, < 6 hours, no fever | Stones on US, normal LFTs, WBC | Elective cholecystectomy |
| Acute cholecystitis | RUQ pain > 6 hours, fever, Murphy sign | Gallbladder wall thickening, pericholecystic fluid, sonographic Murphy | Antibiotics, cholecystectomy within 72 hr |
| Choledocholithiasis | Jaundice, RUQ pain, +/- pancreatitis | Elevated bilirubin/ALP, dilated CBD on US | ERCP with stone extraction, then cholecystectomy |
| Ascending cholangitis | Charcot triad (fever, jaundice, RUQ pain); Reynolds pentad adds hypotension, AMS | Leukocytosis, cholestatic LFTs, dilated ducts | IV antibiotics, urgent ERCP for drainage |
| Gallstone pancreatitis | Epigastric pain radiating to back, nausea | Lipase > 3x ULN, stones on US | Supportive; ERCP if cholangitis; same-admission cholecystectomy |
17 NAFLD/MASLD & Viral Hepatitis Panc/Bil/Hep
MASLD (formerly NAFLD)
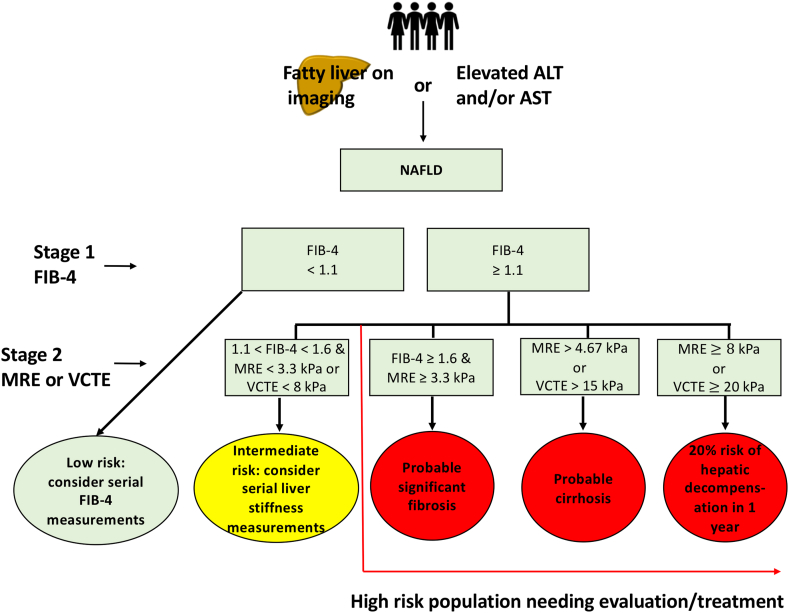
The field renamed NAFLD to MASLD (metabolic dysfunction-associated steatotic liver disease) in 2023, with MASH replacing NASH. MASLD is defined as hepatic steatosis plus at least one cardiometabolic risk factor (overweight/obesity, T2DM, dyslipidemia, hypertension) in the absence of other causes. It is the most common liver disease in the world. Workup: rule out other causes (hepatitis serologies, iron studies, ceruloplasmin, AMA, ANA), assess fibrosis non-invasively with FIB-4, NFS, vibration-controlled transient elastography (FibroScan), or MR elastography. Biopsy is reserved for uncertain cases. Management: weight loss (≥ 7–10% of body weight improves steatosis and inflammation), GLP-1 agonists, treatment of comorbidities, and the first FDA-approved MASH drug resmetirom (Rezdiffra), a selective thyroid hormone receptor-β agonist approved in 2024.
Viral Hepatitis
| Virus | Transmission | Acute/Chronic | Diagnosis | Treatment |
|---|---|---|---|---|
| Hepatitis A | Fecal-oral | Acute only | Anti-HAV IgM | Supportive; vaccine for prevention |
| Hepatitis B | Blood, sexual, perinatal | Acute or chronic | HBsAg, anti-HBc, HBeAg/anti-HBe, HBV DNA | Entecavir or tenofovir for eligible chronic HBV |
| Hepatitis C | Blood, IV drug use | ~75% chronic | Anti-HCV screen → HCV RNA confirmation | 8–12 weeks DAAs (sofosbuvir/velpatasvir, glecaprevir/pibrentasvir) |
| Hepatitis D | Blood (requires HBV) | Chronic | Anti-HDV, HDV RNA in HBsAg+ patients | Bulevirtide (EU), PEG-interferon |
| Hepatitis E | Fecal-oral, zoonotic | Usually acute | Anti-HEV IgM, HEV RNA | Supportive; ribavirin if immunocompromised |
Hepatitis B Serology
HBsAg: Surface antigen — active infection (acute or chronic if > 6 months).
Anti-HBs: Immunity (post-vaccination or recovered infection).
Anti-HBc IgM: Acute infection (also window period).
Anti-HBc total: Prior exposure (past or current).
HBeAg / anti-HBe: High replication / seroconversion.
HBV DNA: Viral load — guides treatment decisions.
AASLD HBV guidance informs when to treat chronic HBV (HBeAg status, ALT, HBV DNA, fibrosis, age). AASLD/IDSA HCV guidance is the continually updated source for pan-genotypic DAA regimens; nearly all HCV is now curable (> 95% SVR) with 8–12 weeks of oral therapy.
18 Autoimmune Liver Disease (AIH, PBC, PSC) Panc/Bil/Hep
Autoimmune Hepatitis (AIH)
AIH is an immune-mediated hepatitis classically in women, presenting with fatigue, jaundice, or asymptomatic transaminitis. Labs: elevated ALT/AST (hepatocellular pattern), hypergammaglobulinemia (IgG), positive ANA, anti-smooth muscle (SMA) or anti-LKM1. Biopsy shows interface hepatitis, plasma cell infiltrate, rosetting. Treatment: prednisone +/- azathioprine; most patients require long-term maintenance.
Primary Biliary Cholangitis (PBC)
PBC (formerly "primary biliary cirrhosis") is a chronic cholestatic autoimmune disease destroying small intrahepatic bile ducts, classically in middle-aged women with fatigue and pruritus. Labs: elevated ALP and GGT (cholestatic pattern), positive AMA (anti-mitochondrial antibody) in > 95%. Treatment: ursodeoxycholic acid (UDCA, ursodiol) 13–15 mg/kg/day is first-line, with obeticholic acid or fibrates added for inadequate response.
Primary Sclerosing Cholangitis (PSC)
PSC is a chronic cholestatic disease causing multifocal strictures of intra- and extrahepatic bile ducts, classically in young men, strongly associated with IBD (especially UC). MRCP shows "beaded" ducts. No medical therapy slows progression; management is surveillance for cholangiocarcinoma (MRCP + CA 19-9 annually), ERCP for dominant strictures, and liver transplant for end-stage disease. PSC-IBD patients need annual surveillance colonoscopy from the time of PSC diagnosis due to high CRC risk.
19 Cirrhosis, Portal HTN & HCC Panc/Bil/Hep
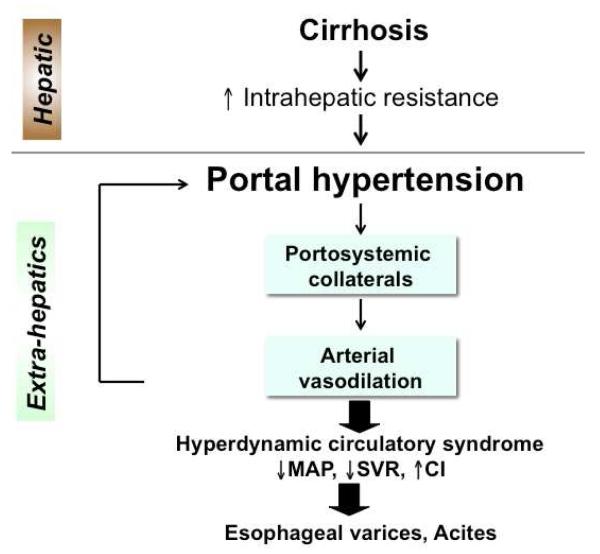
Cirrhosis is the end-stage of chronic liver injury, characterized by fibrosis and nodular regeneration. Causes: MASLD/MASH, alcohol, chronic HCV, chronic HBV, autoimmune (AIH, PBC, PSC), hemochromatosis, Wilson disease, A1AT deficiency, cryptogenic. Clinically divided into compensated (no complications, often asymptomatic) and decompensated (variceal bleeding, ascites, hepatic encephalopathy, jaundice). AASLD practice guidance on cirrhosis is the core reference.
Child-Pugh Score
| Parameter | 1 point | 2 points | 3 points |
|---|---|---|---|
| Total bilirubin (mg/dL) | < 2 | 2–3 | > 3 |
| Albumin (g/dL) | > 3.5 | 2.8–3.5 | < 2.8 |
| INR | < 1.7 | 1.7–2.3 | > 2.3 |
| Ascites | None | Mild/controlled | Moderate/refractory |
| Encephalopathy | None | Grade I–II | Grade III–IV |
Class A: 5–6 points (1-yr survival ~100%). Class B: 7–9 (~80%). Class C: 10–15 (~45%).
MELD Score
MELD (Model for End-Stage Liver Disease) is the objective score used for liver transplant allocation. Current MELD 3.0 (adopted by UNOS in 2023) includes bilirubin, creatinine, INR, sodium, albumin, and sex. Higher scores indicate worse prognosis; MELD ≥ 15 is typically the threshold for transplant listing. Scribes will see MELD trended at every hepatology visit.
Portal Hypertension Complications
Ascites is the most common decompensation. Workup includes diagnostic paracentesis with cell count, culture, albumin (for SAAG), and total protein. SAAG (serum-ascites albumin gradient) ≥ 1.1 = portal hypertensive, < 1.1 = non-portal. Management: sodium restriction (< 2 g/day), diuretics (spironolactone + furosemide in 100:40 ratio), large-volume paracentesis for tense ascites with albumin replacement (8 g per L removed above 5 L), and TIPS for refractory ascites.
Spontaneous bacterial peritonitis (SBP) is diagnosed when ascitic fluid PMN count ≥ 250 cells/mm³. Treatment is IV ceftriaxone 2 g daily x 5 days plus IV albumin (1.5 g/kg day 1, 1 g/kg day 3). Secondary prophylaxis with daily oral ciprofloxacin or TMP-SMX is indicated after a first episode.
Hepatic encephalopathy (HE) is a neuropsychiatric syndrome from accumulation of gut-derived toxins (ammonia and others). Graded by West Haven criteria: Grade 0 minimal (subclinical), I mild confusion, euphoria or anxiety, shortened attention, II lethargy, disorientation to time, personality change, asterixis, III somnolence, gross disorientation, bizarre behavior, IV coma. Precipitants: infection (especially SBP), GI bleeding, constipation, dehydration, electrolyte abnormalities (hypokalemia, alkalosis), medications (sedatives), TIPS, dietary protein load. Treatment: identify and treat precipitant, lactulose titrated to 2–3 soft stools/day, rifaximin 550 mg BID added for recurrent HE.
Hepatocellular Carcinoma (HCC)
HCC arises in cirrhotic livers and in chronic HBV even without cirrhosis. AASLD HCC guidance recommends screening every 6 months with ultrasound +/- AFP in all cirrhotic patients and high-risk HBV. Diagnosis can be made noninvasively by characteristic imaging features on multiphase CT or MRI using LI-RADS. Staging uses the BCLC (Barcelona Clinic Liver Cancer) system:
| BCLC Stage | Features | Treatment |
|---|---|---|
| 0 (very early) | Single ≤ 2 cm, PS 0, Child-Pugh A | Resection or ablation |
| A (early) | Single or up to 3 nodules ≤ 3 cm, PS 0 | Resection, ablation, or transplant |
| B (intermediate) | Multinodular, no vascular invasion, PS 0 | TACE (transarterial chemoembolization) |
| C (advanced) | Portal invasion or extrahepatic spread, PS 1–2 | Systemic therapy: atezolizumab + bevacizumab first-line |
| D (terminal) | PS > 2 or Child-Pugh C | Best supportive care |
On every cirrhosis visit, document: etiology, Child-Pugh class, MELD score, presence/absence of ascites and grade, presence/absence of HE and grade, last EGD for variceal screening and findings, HCC surveillance (last US date, AFP), vaccination status (HAV, HBV, pneumococcal, influenza, COVID), and alcohol/substance use.
20 Endoscopic & GI Procedures Procedures
Endoscopy is the defining procedural activity of gastroenterology. Scribes working in endoscopy suites will pre-populate procedure notes and chart findings, interventions, and specimens. Know each procedure cold.
| Procedure | What It Is | Key Indications |
|---|---|---|
| EGD (upper endoscopy) | Flexible endoscope from mouth through esophagus, stomach, duodenum | Dysphagia, dyspepsia, GIB, alarm features, Barrett surveillance, varices |
| Colonoscopy | Flexible endoscope from rectum to cecum/TI | Screening, surveillance, hematochezia, IBD, chronic diarrhea |
| Flexible sigmoidoscopy | Limited colonoscopy to splenic flexure | Hematochezia evaluation in young, UC flare assessment |
| Capsule endoscopy | Swallowed camera capsule images entire small bowel | Obscure GI bleeding, small bowel Crohn, Peutz-Jeghers |
| Device-assisted (double/single-balloon) enteroscopy | Deep small bowel endoscopy with therapeutic capability | Targeting lesions found on capsule study |
| EUS (endoscopic ultrasound) | Ultrasound probe on an endoscope, often with FNA | Pancreatic masses, subepithelial lesions, staging, cyst sampling, celiac block |
| ERCP | Side-viewing scope to duodenum; cannulate ampulla and access biliary/pancreatic ducts | Choledocholithiasis, cholangitis, strictures, pancreatic duct disorders, stent placement |
| EMR (endoscopic mucosal resection) | Submucosal injection followed by snare resection | Flat polyps, early cancers, Barrett dysplasia |
| ESD (endoscopic submucosal dissection) | En bloc dissection in the submucosal plane | Larger or deeper early neoplasia |
| POEM | Submucosal tunneling and myotomy via endoscope | Achalasia, spastic motility disorders |
| Variceal band ligation (EVL) | Rubber-band ligation of esophageal varices | Acute variceal bleeding, primary/secondary prophylaxis |
| Dilation | Balloon or bougie dilation of a stricture | Peptic stricture, Schatzki ring, anastomotic stricture, EoE |
| PEG placement | Percutaneous endoscopic gastrostomy tube | Long-term enteral nutrition |
| Paracentesis | Needle drainage of ascitic fluid | Diagnostic (new ascites, r/o SBP), therapeutic (tense ascites) |
| Liver biopsy | Percutaneous, transjugular, or EUS-guided | Unexplained liver disease, staging fibrosis, rejection surveillance |
| TIPS | Transjugular intrahepatic portosystemic shunt (IR procedure) | Refractory ascites, refractory variceal bleeding, hepatic hydrothorax |
Pre-Procedure Documentation
Every endoscopy note begins with indication, consent, sedation plan (moderate sedation with fentanyl/midazolam, MAC with propofol, or general anesthesia), Mallampati and ASA class, aspiration risk, and anticoagulation status. ASGE guidelines stratify procedures as low- or high-risk for bleeding to decide on holding antiplatelets and anticoagulants.
Post-Procedure & Complications
Post-procedure recovery includes monitoring vital signs, pain, and sedation recovery (Aldrete score). Findings are reviewed with the patient/family after the patient is alert. Common complications to know: perforation (abdominal pain, tachycardia, subcutaneous emphysema, free air on imaging), bleeding (post-polypectomy bleeding can be immediate or delayed up to 2 weeks), post-ERCP pancreatitis (most common ERCP complication, 3–15% depending on risk; rectal indomethacin and pancreatic duct stenting reduce risk), post-polypectomy syndrome (transmural thermal injury without frank perforation), aspiration pneumonia, and sedation-related events (hypoxia, hypotension).
21 GI Imaging, Labs & Diagnostics Special
Key Labs
| Lab | What It Reflects | Scribe Notes |
|---|---|---|
| ALT / AST | Hepatocellular injury | ALT is more liver-specific; AST:ALT > 2 suggests alcoholic hepatitis |
| ALP | Cholestasis or bone | GGT confirms hepatic origin |
| GGT | Biliary epithelial injury | Also elevated with alcohol, medications |
| Total/direct bilirubin | Cholestasis, hemolysis, synthetic dysfunction | Conjugated > 50% = cholestatic or hepatocellular |
| Albumin | Liver synthetic function (chronic) | Long half-life (~20 days); reflects chronic disease |
| INR / PT | Liver synthetic function (acute) | Rapidly responsive; used in MELD |
| Ammonia | Hepatic encephalopathy (limited value) | Clinical diagnosis; ammonia level not required |
| Lipase | Pancreatic injury | More specific than amylase; > 3x ULN diagnostic for pancreatitis |
| Fecal calprotectin | Colonic inflammation | Distinguishes IBD from IBS; monitors IBD activity |
| Fecal elastase | Pancreatic exocrine function | < 200 suggests insufficiency; < 100 severe |
| Tissue transglutaminase IgA | Celiac screen | Must include total IgA to rule out deficiency |
| Alpha fetoprotein (AFP) | HCC tumor marker | Used with ultrasound for HCC surveillance |
| CA 19-9 | Pancreatic cancer, cholangiocarcinoma | Elevated in cholestasis too; not screening |
Imaging Modalities
| Modality | Best For | Scribe Notes |
|---|---|---|
| Abdominal US | Gallstones, biliary dilation, ascites, cirrhosis screen, HCC surveillance | No radiation; operator-dependent; limited by gas |
| CT abdomen/pelvis (IV contrast) | Diverticulitis, appendicitis, obstruction, masses, pancreatitis | Check creatinine before contrast |
| CT enterography | Crohn disease small bowel assessment | Oral neutral contrast distends small bowel |
| MR enterography | Crohn disease, especially pediatric/young | No radiation; longer exam time |
| MRCP | Biliary and pancreatic duct anatomy | Non-invasive alternative to diagnostic ERCP |
| Elastography (FibroScan, MRE) | Liver fibrosis staging | kPa score; report LSM and CAP (steatosis) |
| HIDA scan | Acute cholecystitis, biliary dyskinesia | Non-visualization of gallbladder = obstructed cystic duct |
| Gastric emptying scintigraphy | Gastroparesis | > 10% retention at 4 hours diagnostic |
| Small bowel follow-through | Motility, fistula, partial obstruction | Declining use with cross-sectional imaging |
| Barium swallow/esophagram | Dysphagia, motility, strictures, achalasia | "Bird's beak" in achalasia |
22 Medications You Must Know Meds
Acid Suppression & Motility
| Drug | Brand | Class | Use |
|---|---|---|---|
| Omeprazole | Prilosec | PPI | GERD, PUD, H. pylori |
| Pantoprazole | Protonix | PPI | IV option for GIB; fewer drug interactions |
| Esomeprazole | Nexium | PPI | GERD, erosive esophagitis |
| Lansoprazole / Dexlansoprazole | Prevacid / Dexilant | PPI | GERD |
| Rabeprazole | Aciphex | PPI | GERD |
| Vonoprazan | Voquezna | P-CAB | H. pylori, erosive esophagitis; newer class |
| Famotidine | Pepcid | H2 blocker | Mild GERD, stress ulcer prophylaxis |
| Sucralfate | Carafate | Mucosal protectant | Ulcer healing, bile reflux |
| Metoclopramide | Reglan | Prokinetic (D2 antagonist) | Gastroparesis, refractory GERD; black-box for tardive dyskinesia |
| Erythromycin | — | Motilin agonist | Gastroparesis, pre-EGD for GIB to clear stomach |
| Prucalopride | Motegrity | 5-HT4 agonist | Chronic idiopathic constipation |
Antiemetics
| Drug | Class | Key Notes |
|---|---|---|
| Ondansetron (Zofran) | 5-HT3 antagonist | QT prolongation |
| Promethazine (Phenergan) | H1/D2 antagonist | Sedating; IV extravasation risk |
| Prochlorperazine (Compazine) | D2 antagonist | EPS risk |
| Aprepitant | NK1 antagonist | Chemotherapy-induced emesis |
IBD Medications
See the IBD management table in Section 09 for the complete class breakdown (5-ASA, corticosteroids, immunomodulators, anti-TNF, anti-integrin, anti-IL-12/23, anti-IL-23, JAK inhibitors, S1P modulators). Pre-biologic screening mandates: TB (IGRA or PPD), HBV (HBsAg, anti-HBc, anti-HBs), varicella, and updated vaccines.
Hepatology Medications
| Drug | Class | Use |
|---|---|---|
| Lactulose | Non-absorbable disaccharide | Hepatic encephalopathy, titrate to 2–3 soft stools/day |
| Rifaximin (Xifaxan) | Non-absorbable antibiotic | HE (add to lactulose), hepatic encephalopathy prevention, IBS-D, SIBO |
| Spironolactone | Aldosterone antagonist | Ascites (100 mg base, 100:40 with furosemide) |
| Furosemide (Lasix) | Loop diuretic | Ascites (paired with spironolactone) |
| Octreotide (Sandostatin) | Somatostatin analogue | Variceal bleeding, HRS, carcinoid |
| Terlipressin (Terlivaz) | Vasopressin analogue | Hepatorenal syndrome type 1 |
| Albumin | Volume expander | LVP, SBP, HRS |
| Entecavir (Baraclude) | Nucleoside analogue | Chronic HBV |
| Tenofovir (Viread, Vemlidy) | Nucleotide analogue | Chronic HBV (TAF preferred for renal/bone) |
| Sofosbuvir/velpatasvir (Epclusa) | DAA (NS5B + NS5A) | Pan-genotypic HCV, 12 weeks |
| Glecaprevir/pibrentasvir (Mavyret) | DAA (NS3/4A + NS5A) | Pan-genotypic HCV, 8 weeks treatment-naive |
| Ribavirin | Guanosine analogue | Retreatment HCV, HEV in immunocompromised |
| UDCA (ursodiol, Actigall) | Bile acid | PBC first-line, gallstone dissolution |
| Obeticholic acid (Ocaliva) | FXR agonist | PBC second-line |
| Resmetirom (Rezdiffra) | Thyroid hormone receptor-β agonist | MASH with significant fibrosis |
Pancreatic Enzymes & Other
| Drug | Category | Use |
|---|---|---|
| Pancrelipase (Creon, Zenpep, Pertzye) | PERT | Chronic pancreatitis, pancreatic cancer, CF |
| Linaclotide (Linzess) | GC-C agonist | IBS-C, CIC |
| Plecanatide (Trulance) | GC-C agonist | IBS-C, CIC |
| Lubiprostone (Amitiza) | Chloride channel activator | IBS-C, CIC, opioid-induced constipation |
| Tenapanor (Ibsrela) | NHE3 inhibitor | IBS-C |
| Eluxadoline (Viberzi) | Mixed opioid | IBS-D; avoid in cholecystectomy patients |
| Loperamide (Imodium) | Opioid receptor agonist | Non-infectious diarrhea |
| Cholestyramine | Bile acid sequestrant | Bile acid diarrhea, pruritus in cholestasis |
23 Classification Systems — Consolidated Special
| System | What It Classifies | Section |
|---|---|---|
| LA classification | Erosive esophagitis (Grade A–D) | 03 |
| Prague C&M | Barrett esophagus extent | 04 |
| Chicago Classification | Esophageal motility disorders by HRM | 05 |
| EREFS | EoE endoscopic features | 05 |
| Forrest classification | UGIB ulcer stigmata (Ia–III) | 14 |
| Montreal classification | IBD extent and behavior | 09 |
| Mayo Score | UC activity | 09 |
| Harvey-Bradshaw Index | Crohn activity | 09 |
| Truelove-Witts | Acute severe UC | 09 |
| Marsh | Celiac histology | 10 |
| Rome IV | Functional GI disorders | 08 |
| Bristol Stool Scale | Stool form (Type 1–7) | 08 |
| Hinchey | Diverticulitis severity | 11 |
| Revised Atlanta (2012) | Acute pancreatitis severity and collections | 15 |
| Ranson criteria | Pancreatitis mortality prediction | 15 |
| BISAP | Early pancreatitis severity | 15 |
| Child-Pugh | Cirrhosis severity (A/B/C) | 19 |
| MELD / MELD 3.0 | Transplant allocation, mortality | 19 |
| West Haven | Hepatic encephalopathy grade | 19 |
| BCLC | HCC staging (0/A/B/C/D) | 19 |
| LI-RADS | HCC imaging category | 19 |
| Internal hemorrhoid grade | I–IV by prolapse | 13 |
24 Physical Exam, Abbreviations & Sample HPIs
The GI & Hepatology Physical Exam
General: Alert, no jaundice/scleral icterus, no asterixis, no cachexia.
Inspection: Abdomen flat/distended, no scars, no caput medusae, no spider angiomas, no gynecomastia.
Auscultation: Normoactive bowel sounds, no bruits.
Percussion: Tympanic throughout, no shifting dullness, liver span normal, no splenomegaly on Castell sign.
Palpation: Soft, non-tender, non-distended, no rebound or guarding, no palpable masses, no hepatosplenomegaly, negative Murphy sign, negative McBurney tenderness.
Specific signs to know and document when present: Murphy sign (cessation of inspiration with RUQ palpation — cholecystitis), McBurney point (1/3 from ASIS to umbilicus — appendicitis), Rovsing, Psoas, and Obturator signs (appendicitis/pelvic inflammation), Cullen sign (periumbilical ecchymosis — severe pancreatitis, hemoperitoneum), Grey Turner sign (flank ecchymosis — retroperitoneal hemorrhage, severe pancreatitis), Courvoisier sign (palpable non-tender gallbladder with jaundice — pancreatic head cancer), shifting dullness and fluid wave (ascites), Terry nails and Muehrcke lines (hypoalbuminemia), asterixis (hepatic encephalopathy, uremia), caput medusae, spider angiomas, palmar erythema, gynecomastia, testicular atrophy (chronic liver disease).
Abbreviations Master List
Anatomy
Diagnoses
Procedures & Imaging
Labs & Scores
Sample HPI Templates
"Mr. [Name] is a 52-year-old male with a BMI of 33, hypertension, and hyperlipidemia who presents with a 6-month history of worsening heartburn. He describes a retrosternal burning sensation occurring 4–5 nights per week, worse after large evening meals and when lying flat, relieved partially by OTC famotidine. He endorses intermittent regurgitation of sour fluid but denies dysphagia, odynophagia, weight loss, hematemesis, or melena. No nocturnal cough or hoarseness. He has tried elevating the head of his bed and avoiding late meals with partial improvement. No prior EGD. He drinks 1–2 beers nightly and is a never-smoker."
"Ms. [Name] is a 64-year-old female with osteoarthritis on daily ibuprofen 600 mg TID for the past year who presents with a 3-week history of epigastric burning pain. She describes the pain as 6/10, localized to the epigastrium, without radiation, occurring 1–2 hours after meals and occasionally waking her at night, partially relieved by antacids. She endorses mild nausea but denies vomiting, hematemesis, melena, dysphagia, weight loss, or early satiety. No prior H. pylori testing or EGD. No family history of gastric cancer. She continues to take her NSAIDs as before."
"Mr. [Name] is a 71-year-old male with atrial fibrillation on apixaban 5 mg BID, hypertension, and CAD status post PCI on aspirin 81 mg daily who presents to the ED with 2 days of black tarry stools and lightheadedness. He reports 3 episodes of melena, most recently this morning, without frank hematemesis or bright red blood per rectum. He endorses mild epigastric discomfort and one syncopal episode when standing this morning. Vital signs on arrival: HR 108, BP 96/58, SpO2 97% on room air. Labs notable for Hb 7.8 g/dL (baseline 13.5), INR 1.4, BUN/Cr 48/1.2. He has received 2 units of PRBCs, pantoprazole 80 mg IV bolus followed by infusion, and apixaban has been held. GI consulted for urgent EGD."
"Ms. [Name] is a 28-year-old female with Montreal E3 pancolitis diagnosed in 2022, currently on infliximab 5 mg/kg every 8 weeks (last infusion 3 weeks ago) and mesalamine 1.5 g BID, presenting for routine follow-up. She reports she is doing well overall with 1–2 formed bowel movements daily, no blood, no urgency or tenesmus, no nocturnal stools. Partial Mayo score 0. She denies abdominal pain, weight loss, fever, arthralgias, skin, or eye symptoms. Most recent labs 2 weeks ago: CRP < 0.3, fecal calprotectin 42, Hb 13.1, albumin 4.2. Last colonoscopy 9 months ago showed Mayo endoscopic subscore 1 in the sigmoid and rectum with loss of vascular pattern, without ulceration; biopsies showed chronic inactive colitis. TB screen, HBV, and HAV vaccination up to date."
"Mr. [Name] is a 58-year-old male with type 2 diabetes, obesity (BMI 35), and hypertension referred for evaluation of painless jaundice and new-onset dark urine over the past 2 weeks. He has noted 15 lb unintentional weight loss over 3 months, progressive fatigue, and new-onset pruritus. He denies abdominal pain, fevers, chills, or prior liver disease. He drinks 2–3 glasses of wine weekly, no IV drug use, no tattoos, no recent travel. Labs: Tbili 8.2 (Dbili 6.4), ALP 540, AST 110, ALT 145, lipase WNL, CA 19-9 pending. Abdominal ultrasound outside shows intra- and extrahepatic biliary ductal dilation with a dilated CBD to 12 mm and a suspicious 2.8 cm hypoechoic mass in the pancreatic head. CT pancreas protocol and EUS with FNA are planned."
"Ms. [Name] is a 46-year-old female presenting for an initial average-risk screening colonoscopy. She is asymptomatic and denies abdominal pain, change in bowel habits, rectal bleeding, melena, weight loss, anemia, or unintentional weight loss. Bowel habits are regular, Bristol 3–4, once daily. No personal history of polyps or IBD. Family history is negative for colorectal cancer, polyps, or hereditary cancer syndromes. No prior colonoscopy. She has no personal history of anesthesia complications. Last FIT was not performed. Plan for split-dose PEG prep and screening colonoscopy with moderate sedation."
"Mr. [Name] is a 61-year-old male with Child-Pugh B (8), MELD 18, alcohol-associated cirrhosis complicated by ascites and grade 2 esophageal varices (on nadolol 40 mg daily for primary prophylaxis), presenting with 1 week of worsening abdominal distension and confusion. Family reports sleep-wake reversal and one episode of disorientation to time. He has been non-adherent with lactulose for the past 5 days due to diarrhea, and his furosemide was recently doubled by an urgent care. No fevers, hematemesis, or melena. Exam reveals scleral icterus, mild asterixis, tense ascites, and West Haven Grade II encephalopathy. Labs: Na 128, Cr 1.6 (baseline 1.0), Tbili 4.8, INR 1.9, albumin 2.7, Hb 10.2, Plt 72. Diagnostic and therapeutic paracentesis planned; ceftriaxone started empirically pending fluid cell count and culture for suspected SBP."
The best GI scribes understand that the specialty is equal parts clinic, procedure suite, and hospital consult service. When the attending says "LA Grade C esophagitis with Prague C2M4 Barrett and no dysplasia on biopsy," you should recognize a moderate-severe reflux disease with a 4 cm Barrett segment requiring indefinite PPI and surveillance in 3–5 years. When you hear "Child-Pugh B, MELD 22, tense ascites, AMS," you should already be preparing the encephalopathy workup, diagnostic paracentesis plan, and hepatology consult language.
Be precise. Distances, grades, classifications, and medication doses are not optional details — they are the documentation. Pre-read the imaging and endoscopy reports before the patient is seen, know the medications by generic and brand name, and memorize the classification systems in Section 23. GI medicine rewards the scribe who can hear a dictated finding and immediately chart it with the right anatomic location, the right classification stage, and the right plan.
Welcome to gastroenterology. The volume is high and the vocabulary is dense, but the workflows are repeatable — and once you have them, you will move faster than you thought possible.
25 References & Sources
Clinical Practice Guidelines
Rubin DT, et al. ACG Clinical Guideline: Ulcerative Colitis in Adults. Am J Gastroenterol. 2019.
USPSTF. Screening for Colorectal Cancer: Recommendation Statement. JAMA. 2021.
Diagram & Figure Sources
Figure 1: Components of the Digestive System. OpenStax College. CC BY 3.0.
Figure 2: Stomach Anatomy. OpenStax College. CC BY 3.0.
Figure 3: Anatomy of the Small Intestine. OpenStax College. CC BY 3.0.
Figure 4: Anatomy of the Large Intestine. OpenStax College. CC BY 3.0.
Figure 5: Accessory Digestive Organs. OpenStax College. CC BY 3.0.
Figure 6: Gallbladder Anatomy. Blausen Medical. CC BY 3.0.
Figure 7: Peptic Ulcer. Blausen Medical. CC BY 3.0.