Geriatrics
Every diagnosis, syndrome, assessment tool, medication, abbreviation, and documentation framework you need to succeed on day one in a geriatrics practice.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Aging Physiology — What Changes with Age
Geriatrics is the care of older adults, typically 65 and older, with particular focus on those with multiple chronic conditions, functional decline, cognitive impairment, and complex social circumstances. Unlike most specialties, geriatrics is defined not by an organ system but by the interaction between aging physiology, multimorbidity, and function. A scribe in a geriatrics clinic must understand that the same disease in an 80-year-old looks, presents, and is treated differently than in a 50-year-old.
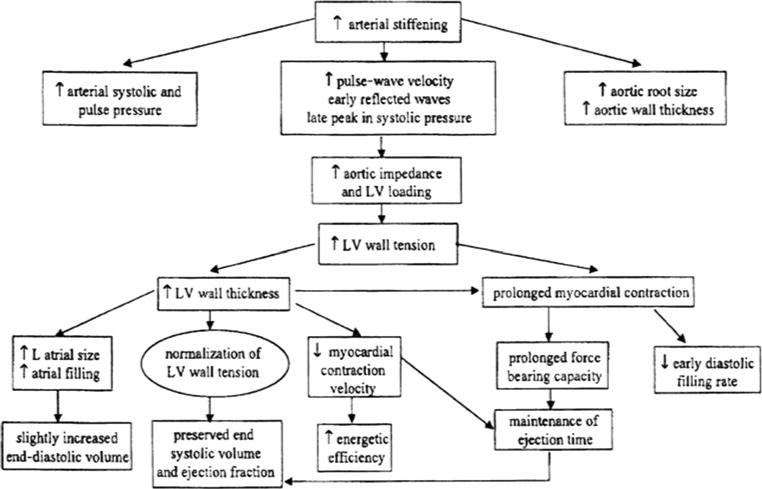
Physiologic Reserve and Homeostenosis
With age, nearly every organ system loses functional reserve — the capacity to respond to stress. This is called homeostenosis. A young adult can lose 30–40% of renal function and remain asymptomatic; an older adult with the same loss may develop acute kidney injury from a single NSAID dose. Baseline is usually adequate; it is the response to an insult (infection, dehydration, new medication, hospitalization) that fails.
System-by-System Changes
The cardiovascular system shows arterial stiffening, left ventricular hypertrophy, decreased beta-adrenergic responsiveness, and reduced maximum heart rate. Isolated systolic hypertension is the rule. Orthostatic hypotension becomes common because of impaired baroreflex sensitivity.
The renal system loses approximately 0.75–1 mL/min/year of GFR after age 40. Serum creatinine can remain falsely "normal" because muscle mass also declines, which is why eGFR (CKD-EPI or Cockcroft-Gault) is the correct measure. Drug clearance of renally excreted medications (digoxin, many antibiotics, DOACs) must be dose-adjusted.
The hepatic system has reduced first-pass metabolism and phase I (CYP450) oxidation capacity, prolonging half-lives of benzodiazepines, opioids, and many psychotropics. Phase II conjugation is relatively preserved, which is why lorazepam, oxazepam, and temazepam (LOT) are the "safer" benzodiazepines in the elderly when one must be used.
The central nervous system shows cortical atrophy, loss of cholinergic neurons (the substrate for dementia and delirium), reduced dopamine, and disrupted sleep architecture with less deep sleep. Anticholinergic medications are particularly damaging.
Body composition shifts toward increased fat and decreased lean body mass and total body water. This changes drug pharmacokinetics: lipophilic drugs (diazepam) have a larger volume of distribution and prolonged effect; hydrophilic drugs (lithium, digoxin) have a smaller volume of distribution and higher peak levels at a given dose.
Whenever the attending says "watch the creatinine" or "that dose is too high for her kidneys," you should be looking at the eGFR, not the serum creatinine. An 85-year-old woman with a creatinine of 1.1 mg/dL often has a GFR below 45. Always record eGFR and weight in the note when renally-cleared drugs are being started or changed.
02 Scribe Documentation Framework
The Geriatrics SOAP Note
Geriatrics notes use the standard SOAP structure but with extra emphasis on function, cognition, mood, mobility, and the medication list. A note that documents only diagnoses and labs will miss the core of a geriatric encounter.
Chief Complaint: Often a caregiver concern ("she's more forgetful," "he fell twice last month"), a follow-up of a chronic problem, or a scheduled annual wellness visit. Document who is providing history — the patient, a spouse, an adult child, or a facility nurse.
HPI: Classic OLDCARTS applies, but geriatric HPIs always include function before and after the problem started (what could they do, what can they do now), cognition (any new confusion, word-finding difficulty, getting lost), and mood changes. For falls, document circumstances, loss of consciousness, injuries, and fear of falling.
Medications: A full reconciliation every visit. Document every prescription, OTC, supplement, and herbal product, with dose, frequency, and who is administering it. Note adherence.
Social History: Living situation (alone, with spouse, assisted living, skilled nursing), caregiver support, stairs in the home, driving status, alcohol/tobacco, advance directives (POLST, DPOA, code status), and any financial or safety concerns.
Vitals: Include orthostatic vital signs (supine, then standing at 1 and 3 minutes) in any patient with falls, dizziness, or syncope. Document weight at every visit — unintentional loss of 5% in 6 months is a red flag.
Functional status: ADLs (bathing, dressing, toileting, transferring, continence, feeding), IADLs (finances, medications, shopping, cooking, transportation, telephone, housework, laundry), and mobility (cane, walker, wheelchair, gait speed if measured).
Cognitive/mood screens: Mini-Cog, MoCA, MMSE, or SLUMS scores with the date, and PHQ-9 or GDS-15 if performed.
Problem-oriented, with each chronic problem addressed (not just the acute complaint). Every geriatric A&P should also address goals of care, code status, advance directives, and any deprescribing decisions. Caregiver education and social work involvement are frequently part of the plan.
In a geriatrics note, always capture the source of history ("History obtained from patient and daughter who lives with her") and the reliability ("patient a limited historian due to moderate dementia; collateral from daughter"). This is a medicolegal expectation and also tells the next reader how much weight to give the narrative.
03 Comprehensive Geriatric Assessment (CGA) Assessment
The Comprehensive Geriatric Assessment is the signature tool of geriatric medicine — a multidimensional, multidisciplinary evaluation that covers medical, functional, cognitive, psychological, social, and environmental domains. A CGA is the default framework for a new geriatrics consultation and underpins most documentation.
1. Medical: Complete problem list, medication review, nutrition, continence, pain, sensory function (vision, hearing, dentition).
2. Functional: Basic ADLs (Katz), instrumental ADLs (Lawton), mobility, gait and balance, fall history.
3. Cognitive: Formal cognitive screening (Mini-Cog, MoCA, MMSE, SLUMS), history of memory complaints, collateral.
4. Psychological: Depression screening (GDS-15 or PHQ-9), anxiety, sleep, substance use, grief.
5. Social: Caregiver availability and strain, living situation, finances, elder abuse screen, social engagement.
6. Environmental: Home safety, stairs, rugs, bathroom grab bars, transportation, access to food and pharmacy.
CGA has been shown in multiple randomized trials to reduce functional decline and nursing home admission when applied to frail hospitalized older adults, and it is endorsed as a core framework by the Assessing Care of Vulnerable Elders (ACOVE) quality indicators.
04 Dementia — Alzheimer, Vascular, DLB, FTD, Mixed, MCI Cognitive
Dementia (now formally called major neurocognitive disorder in DSM-5) is an acquired decline in one or more cognitive domains that interferes with independence in daily activities. It is not a single disease but a syndrome with many causes. Roughly 6 million Americans have Alzheimer disease, and dementia is the leading cause of institutionalization in the elderly.
Mild Cognitive Impairment (MCI)
MCI is cognitive decline greater than expected for age but not severe enough to impair function. Approximately 10–15% of MCI patients progress to dementia per year. Documenting whether the patient is MCI vs early dementia hinges on functional impairment, not just test scores.
Alzheimer Disease (AD)
The most common cause of dementia (60–70%). Pathologically characterized by amyloid-beta plaques and neurofibrillary tau tangles, with hippocampal atrophy on MRI. Clinically presents with insidious, gradually progressive short-term memory loss, word-finding difficulty, and eventually apraxia, agnosia, and loss of executive function. Diagnosis is now guided by the NIA-AA criteria, which incorporate biomarkers (CSF amyloid/tau, amyloid PET).
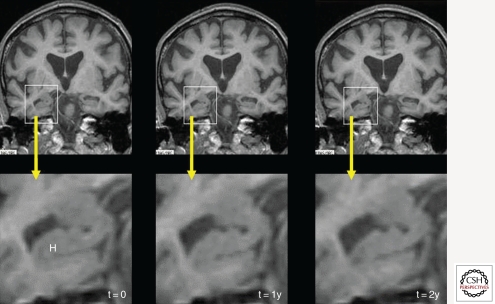
Vascular Dementia
Second most common cause. Caused by cumulative large- or small-vessel cerebrovascular disease. Classically presents with stepwise decline, executive dysfunction, gait disturbance (often early), and focal neurologic signs. MRI shows white matter disease, lacunar infarcts, or cortical strokes.
Dementia with Lewy Bodies (DLB)
Characterized by the triad of fluctuating cognition, visual hallucinations, and spontaneous parkinsonism. Patients also have REM sleep behavior disorder and severe sensitivity to antipsychotics (neuroleptic sensitivity — avoid typical antipsychotics and most atypicals; quetiapine or pimavanserin are safer if needed).
Frontotemporal Dementia (FTD)
Typically younger onset (50s–60s). Behavioral variant FTD presents with disinhibition, apathy, compulsive behaviors, and loss of empathy, with relative sparing of memory early on. Primary progressive aphasia variants present with language impairment as the dominant feature.
Mixed Dementia
Coexistence of AD pathology with vascular disease, very common in the oldest-old. Documented when imaging shows significant small-vessel disease in a patient meeting clinical AD criteria.
Diagnostic Workup
Every new cognitive complaint should trigger: formal cognitive testing (MoCA preferred over MMSE for sensitivity to MCI), TSH, B12, CMP, CBC, and an MRI (or CT if MRI contraindicated) to rule out reversible or structural causes (NPH, subdural, tumor, stroke). Depression screening is mandatory to rule out "pseudodementia." Amyloid PET and CSF biomarkers are reserved for diagnostic uncertainty or disease-modifying therapy candidacy.
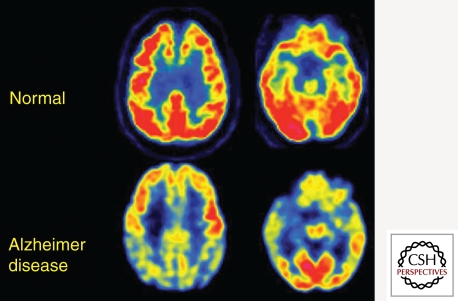
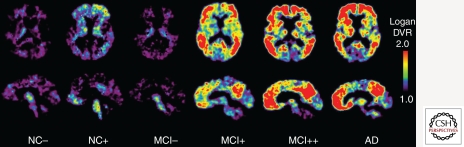
Management
Cholinesterase inhibitors (donepezil, rivastigmine, galantamine) are first-line for mild to moderate AD and DLB. Memantine, an NMDA antagonist, is added for moderate to severe AD. The anti-amyloid monoclonal antibodies lecanemab (Leqembi) and donanemab (Kisunla) are FDA-approved for early symptomatic AD with confirmed amyloid pathology; they modestly slow decline but carry a risk of ARIA (amyloid-related imaging abnormalities — edema and microhemorrhage) and require serial MRI monitoring. The AAN practice guideline on MCI is the standard citation for cognitive screening recommendations. Non-pharmacologic management is equally important: caregiver education, safety assessment, driving evaluation, advance care planning, and, eventually, hospice referral.
Agitation, aggression, wandering, sundowning, psychosis, and resistance to care. First-line management is non-pharmacologic: identify unmet needs (pain, toileting, hunger, fatigue), reduce environmental triggers, maintain routine. Antipsychotics carry a black-box warning for increased mortality in dementia and should only be used for severe, refractory symptoms with explicit informed consent documented.
When the attending diagnoses dementia, you should be documenting: (1) the specific subtype or "unspecified," (2) severity (mild/moderate/severe, often anchored to MoCA score), (3) functional impairment (which ADLs and IADLs are lost), (4) whether BPSD is present, (5) caregiver identity and capacity, and (6) code status / advance directive status. Missing any of these is an incomplete dementia note.
05 Delirium Cognitive
Delirium is an acute, fluctuating disturbance of attention and awareness, with altered cognition or perception, caused by an underlying medical condition, substance, or medication. It is not a psychiatric diagnosis — it is a medical emergency. Delirium affects up to 50% of hospitalized older adults and is associated with increased mortality, prolonged hospitalization, and long-term cognitive decline.
Subtypes
Hyperactive delirium — agitated, hypervigilant, combative (the stereotypic presentation). Hypoactive delirium — quiet, withdrawn, somnolent (the most common and most missed). Mixed — fluctuating between the two.
Diagnosis: CAM Criteria
Delirium is present when features 1 AND 2 AND (3 OR 4) are present:
1. Acute onset and fluctuating course — Is there evidence of an acute change in mental status from baseline? Did the abnormal behavior come and go or fluctuate during the day?
2. Inattention — Did the patient have difficulty focusing attention, being easily distractible, or having difficulty keeping track of what was said? (Tested with digit span, days of the week backward, or months of the year backward.)
3. Disorganized thinking — Was the patient's thinking disorganized or incoherent, with rambling or irrelevant conversation, unclear or illogical flow of ideas?
4. Altered level of consciousness — Was the patient anything other than alert: vigilant (hyperalert), lethargic, stuporous, or comatose?
Workup — Find the Cause
Delirium is always caused by something. The mnemonic "DIMES" (Drugs, Infection, Metabolic, Environment/Endocrine, Stroke/Structural) or "I WATCH DEATH" is a standard framework. Core workup: medication review (especially new anticholinergics, benzodiazepines, opioids), UA and urine culture, CBC, CMP, TSH, calcium, B12, chest X-ray, EKG, O2 saturation, finger-stick glucose, and neurologic exam. Head CT is reserved for focal findings, trauma, or anticoagulation. LP and EEG are occasional second-line tests.
Management
Treat the underlying cause. Non-pharmacologic bundle: reorientation, adequate lighting, glasses and hearing aids in place, family at bedside, uninterrupted sleep, early mobilization, adequate hydration, minimizing lines and tethers. Avoid restraints. Pharmacologic treatment is reserved for severe agitation threatening patient or staff safety; low-dose haloperidol or an atypical antipsychotic (quetiapine) may be used briefly, but never in DLB or Parkinson disease dementia.
If an older patient is "not acting right," it is delirium until proven otherwise. Document the time course (hours to days), the fluctuation (which is pathognomonic), and the inattention. A patient who cannot recite the months of the year backward has failed an attention screen — write that in the exam.
06 Depression & Anxiety in the Elderly Psych
Depression in older adults is underdiagnosed because it often presents somatically (fatigue, pain, weight loss, insomnia) rather than with classic sadness. Late-life depression is a major risk factor for suicide (white men over 85 have the highest suicide rate of any demographic in the United States) and for progression to dementia.
Screening
The Geriatric Depression Scale (GDS-15) is the preferred screen in cognitively intact elderly. The PHQ-9 is also used and gives a severity score. A positive screen triggers a full diagnostic interview (DSM-5 criteria: 5 of 9 symptoms, ≥ 2 weeks, including depressed mood or anhedonia).
Treatment
First-line pharmacotherapy is an SSRI — sertraline, escitalopram, and citalopram are the geriatric favorites (citalopram capped at 20 mg/day due to QT prolongation). Avoid paroxetine (anticholinergic) and fluoxetine (long half-life, drug interactions). SNRIs (duloxetine, venlafaxine) are useful when there is comorbid pain. Mirtazapine is an excellent choice in patients with insomnia and poor appetite because it promotes sleep and weight gain. Avoid tricyclic antidepressants (amitriptyline, imipramine) — highly anticholinergic and on the Beers list. Non-pharmacologic: behavioral activation, CBT, problem-solving therapy, exercise, and treatment of sensory deficits. ECT remains the most effective treatment for severe, psychotic, or refractory late-life depression.
Anxiety
Generalized anxiety disorder is also common. SSRIs and SNRIs are first-line. Benzodiazepines are on the Beers list for nearly all older adults because of falls, fractures, delirium, and cognitive impairment — use is limited to short courses or specific indications (alcohol withdrawal, severe acute anxiety, end-of-life symptoms).
07 Falls & Gait Disorders Mobility
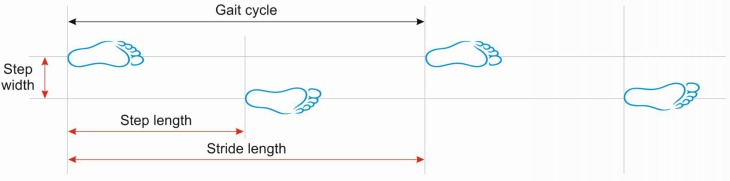
One in three community-dwelling adults over 65 falls each year; one in ten falls results in a serious injury. Falls are a sentinel event and a mandatory documentation item at every geriatric visit. The USPSTF recommendation on fall prevention endorses exercise interventions in community-dwelling adults 65+ at increased risk, and the AGS/BGS falls prevention guideline is the standard framework for multifactorial assessment.
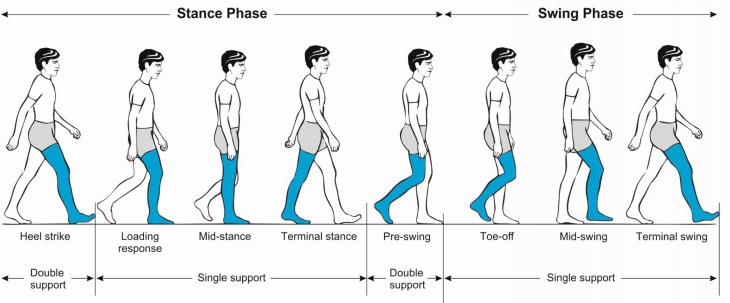
Fall Screening and Assessment
Every older adult is asked annually: (1) Have you fallen in the past year? (2) Do you feel unsteady when standing or walking? (3) Are you worried about falling? A "yes" to any triggers a multifactorial falls assessment: medication review (focus on psychotropics, antihypertensives, hypoglycemics, anticholinergics), orthostatic vital signs, vision screen, foot and footwear exam, neurologic and musculoskeletal exam, and a performance measure (Timed Up and Go, or Tinetti POMA).
Gait and Balance Assessment Tools
Patient rises from a standard armchair, walks 3 meters at normal pace, turns, walks back, and sits down. Timed in seconds. < 10 sec — normal mobility. 10–20 sec — good mobility, usually independent. > 12 sec — increased fall risk in community-dwelling elderly. > 20 sec — significant mobility impairment, needs further assessment. > 30 sec — dependent in most ADLs and mobility.
Balance score (max 16): sitting balance, rising from chair, attempts to rise, immediate standing balance, standing balance, nudge test, eyes closed, turning 360°, sitting down.
Gait score (max 12): initiation of gait, step length and height, step symmetry, step continuity, path, trunk sway, walking stance.
Total /28: ≥ 25 low fall risk; 19–24 moderate fall risk; < 19 high fall risk.
History of falling (25), secondary diagnosis (15), ambulatory aid (0/15/30), IV/heparin lock (20), gait (0/10/20), mental status (0/15). 0–24 no risk; 25–50 low risk; ≥ 51 high risk.
Gait Disorders

Common patterns: antalgic (painful — OA, fracture), parkinsonian (shuffling, stooped, reduced arm swing, festination), apraxic/magnetic (feet stick to the floor — NPH), ataxic (wide-based, unsteady — cerebellar, sensory, vestibular), hemiparetic (circumduction — stroke), and neuropathic/steppage (foot drop — peripheral neuropathy).
After every fall, chart the mechanism (mechanical, syncopal, unknown), location, injuries sustained, head strike and LOC, current anticoagulation, and any medication or environmental contributor the attending identifies. A fall on warfarin with head strike almost always triggers a head CT.
08 Syncope in the Older Adult Cardiovascular
Syncope is transient loss of consciousness from global cerebral hypoperfusion with spontaneous complete recovery. In older adults it is frequently multifactorial and often difficult to distinguish from an unwitnessed mechanical fall. The key categories are reflex/vasovagal, orthostatic, and cardiac. Cardiac syncope carries the highest mortality and includes arrhythmias (sinus node dysfunction, AV block, VT) and structural disease (aortic stenosis, HCM, PE). Orthostatic causes include volume depletion, autonomic failure (diabetes, Parkinson, MSA), and medications (alpha-blockers, diuretics, nitrates, tricyclics).
Workup
Every syncope workup in the elderly includes orthostatic vitals, EKG, CBC, BMP, troponin if cardiac concern, and often echocardiogram. Inpatient telemetry is indicated for suspected cardiac syncope (abnormal EKG, structural disease, exertional syncope, syncope without prodrome). Tilt-table testing is reserved for recurrent unexplained cases.
09 Urinary Incontinence GU
Urinary incontinence affects up to half of community-dwelling older women and a significant minority of men. It is a major driver of caregiver burden and nursing home placement. It is not a normal part of aging and almost always deserves evaluation.
Types
| Type | Mechanism | Classic Presentation | First-Line Management |
|---|---|---|---|
| Urge (overactive bladder) | Detrusor overactivity | Sudden, uncontrollable urge; leakage on the way to bathroom; nocturia | Bladder training, pelvic floor exercises, antimuscarinics (caution), mirabegron |
| Stress | Urethral sphincter weakness | Leakage with cough, laugh, sneeze, lifting; typically women post-childbirth | Pelvic floor (Kegel) exercises, pessary, sling surgery |
| Mixed | Urge + stress | Features of both | Treat the dominant component first |
| Overflow | Impaired contractility or outlet obstruction (BPH, stricture, neurogenic) | Dribbling, weak stream, incomplete emptying, elevated PVR | Treat obstruction (tamsulosin, TURP), intermittent catheterization |
| Functional | Cognitive or mobility impairment preventing timely toileting | Continent bladder but cannot reach toilet in time | Scheduled toileting, environmental modification |
Reversible Causes — DIAPPERS
Delirium, Infection (UTI), Atrophic vaginitis, Pharmaceuticals (diuretics, anticholinergics, opioids, alpha-blockers), Psychological (depression), Excessive urine output (hyperglycemia, CHF, hypercalcemia), Restricted mobility, Stool impaction.
Workup
History, voiding diary, exam (pelvic and prostate), urinalysis, post-void residual (ultrasound or catheter). Urodynamics reserved for refractory or diagnostic uncertainty.
Antimuscarinic bladder medications (oxybutynin, tolterodine, solifenacin) are anticholinergic and on the Beers list — they worsen cognition and contribute to delirium. If the attending prescribes one, document the cognitive baseline and the plan to reassess. Mirabegron (a beta-3 agonist) is the preferred alternative in cognitively vulnerable patients.
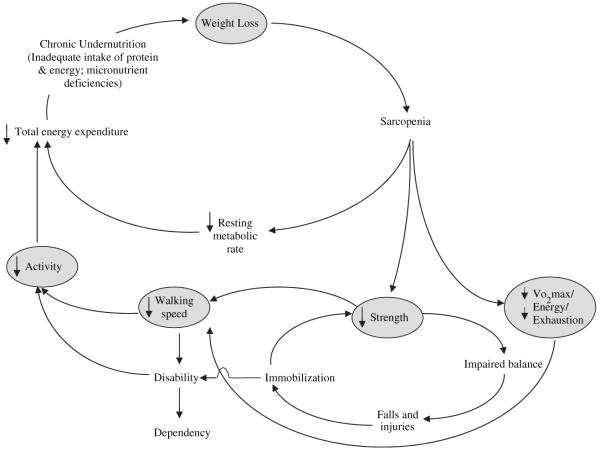
10 Frailty, Sarcopenia & Unintentional Weight Loss Syndromes
Frailty is a state of increased vulnerability to stressors due to decline across multiple physiologic systems. It predicts disability, falls, hospitalization, and death independently of comorbidity and disability. Two dominant operational definitions exist.
Frail = 3 or more of: (1) unintentional weight loss ≥ 10 lb in the past year; (2) self-reported exhaustion; (3) weakness (grip strength in the lowest quintile by sex and BMI); (4) slow walking speed (> 6–7 sec to walk 15 ft); (5) low physical activity. 1–2 criteria = pre-frail. 0 = robust.
Five-item self-report screen: Fatigue, Resistance (inability to climb one flight of stairs), Ambulation (inability to walk one block), Illnesses (> 5 comorbidities), and Loss of weight (> 5%). 0 = robust; 1–2 = pre-frail; 3–5 = frail.
Sarcopenia
Age-related loss of skeletal muscle mass, strength, and function. Diagnosed by reduced grip strength plus low muscle mass (DEXA or BIA) or slow gait speed. Treatment is resistance exercise and adequate protein intake (1.0–1.2 g/kg/day or more).
Unintentional Weight Loss
Loss of ≥ 5% body weight in 6–12 months is a red flag and warrants evaluation. Differential: malignancy (one-third), GI disease, depression, dementia, hyperthyroidism, medication side effects, poor dentition, social/financial barriers (food insecurity), and cognitive inability to prepare food. Workup: CBC, CMP, TSH, age-appropriate cancer screening, chest X-ray, and targeted imaging based on history.
Malnutrition
Screened with the Mini Nutritional Assessment (MNA) or MUST tool. Management includes dietitian referral, small frequent meals, fortification, oral nutritional supplements (Ensure, Boost), and treatment of reversible contributors.
11 Polypharmacy & Deprescribing Meds
Polypharmacy is commonly defined as the use of 5 or more regular medications. It is the rule, not the exception, in older adults and is strongly associated with adverse drug events, falls, hospitalization, and nonadherence. Deprescribing — the structured process of identifying and stopping medications where harms outweigh benefits — is a core geriatric intervention.
The Beers Criteria
The AGS Beers Criteria (2023 update) is the most widely used list of potentially inappropriate medications (PIMs) in adults 65+. It is organized into categories: medications to avoid in most older adults, medications to avoid in specific conditions, medications to use with caution, drug-drug interactions to avoid, and dose adjustments for renal function.
First-generation antihistamines (diphenhydramine, hydroxyzine, chlorpheniramine): anticholinergic, sedating, fall risk.
Benzodiazepines & Z-drugs (zolpidem, eszopiclone): falls, fractures, delirium, cognitive impairment.
Anticholinergics broadly: tricyclic antidepressants, oxybutynin, tolterodine, cyclobenzaprine, scopolamine, hyoscyamine, paroxetine.
Skeletal muscle relaxants (cyclobenzaprine, methocarbamol, carisoprodol): anticholinergic, sedating.
NSAIDs chronically: GI bleeding, AKI, HTN, CHF exacerbation.
Sliding-scale insulin as sole regimen: hypoglycemia risk.
Digoxin > 0.125 mg/day: toxicity.
Alpha-1 blockers (doxazosin, terazosin, prazosin) as antihypertensives: orthostatic hypotension.
Sulfonylureas (long-acting) (glyburide, chlorpropamide): prolonged hypoglycemia.
PPIs > 8 weeks without clear indication: C. diff, fractures, B12 deficiency.
Meperidine: neurotoxicity, seizures, delirium.
STOPP/START Criteria
STOPP/START version 3 is a European explicit-criteria tool. STOPP (Screening Tool of Older Persons' Prescriptions) lists medications to stop. START (Screening Tool to Alert to Right Treatment) lists medications that should be considered but are often omitted (e.g., statin after stroke, ACEI after MI, calcium/vitamin D in osteoporosis).
Anticholinergic Burden
The total anticholinergic load of a medication regimen is calculated with tools like the Anticholinergic Cognitive Burden (ACB) scale. A cumulative ACB ≥ 3 is associated with cognitive impairment, delirium, and falls. Common contributors: diphenhydramine, oxybutynin, tricyclics, paroxetine, cyclobenzaprine, and many antipsychotics.
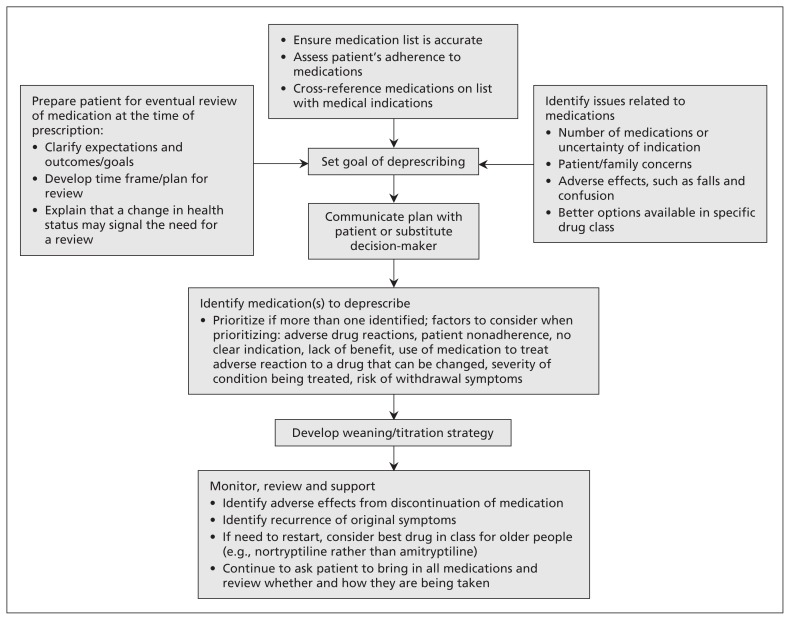
When the attending says "let's taper the Ambien" or "let's stop the oxybutynin," that is a deprescribing decision and should be charted with the rationale (fall risk, cognitive risk, Beers list), the taper plan, and the follow-up. Do not just remove the drug from the list without narrative.
12 Osteoporosis & Fragility Fractures Bone
Osteoporosis is a skeletal disorder of compromised bone strength leading to increased fracture risk. Hip, vertebral, and wrist fractures are the dominant morbidities. A fragility fracture is a fracture from a fall from standing height or less and is itself diagnostic of osteoporosis regardless of DXA.
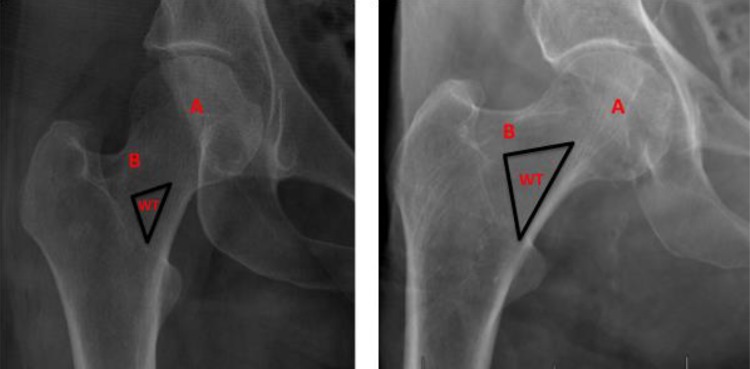
Screening
DXA scan is the standard test. USPSTF recommends DXA in all women ≥ 65 and in postmenopausal women younger than 65 with elevated risk. Men are screened based on risk (prior fragility fracture, chronic steroids, hypogonadism, multiple risk factors). Endocrine Society guidelines on postmenopausal osteoporosis are the standard citation for pharmacologic management thresholds.
T-Score Categories (WHO)
| T-Score | Category | Clinical Meaning |
|---|---|---|
| ≥ −1.0 | Normal | Within 1 SD of young adult mean |
| −1.0 to −2.5 | Osteopenia (low bone mass) | Consider FRAX to guide treatment |
| ≤ −2.5 | Osteoporosis | Pharmacologic treatment indicated |
| ≤ −2.5 + fragility fx | Severe (established) osteoporosis | Definite treatment; consider anabolic |
FRAX
The WHO Fracture Risk Assessment Tool calculates 10-year risk of major osteoporotic fracture and hip fracture. Treatment thresholds (US): 10-year hip fracture risk ≥ 3% or major osteoporotic fracture risk ≥ 20%.
Medications
| Drug | Class | Key Notes |
|---|---|---|
| Alendronate (Fosamax) | Oral bisphosphonate | Weekly; upright 30 min; avoid in severe GERD, CKD stage 4–5 |
| Risedronate (Actonel) | Oral bisphosphonate | Similar to alendronate |
| Zoledronic acid (Reclast) | IV bisphosphonate | Annual infusion; acute phase flu-like reaction; avoid if CrCl < 35 |
| Denosumab (Prolia) | RANKL monoclonal antibody | Q6 months SC; must not be discontinued abruptly (rebound vertebral fractures) |
| Teriparatide (Forteo) | PTH 1–34 analog (anabolic) | Daily SC; 2-year lifetime max; severe or refractory disease |
| Abaloparatide (Tymlos) | PTHrP analog (anabolic) | Similar to teriparatide |
| Romosozumab (Evenity) | Sclerostin inhibitor | Monthly SC × 12; black-box MI/stroke risk |
| Raloxifene | SERM | Vertebral fracture only; VTE risk |
All patients need adequate calcium (1200 mg/day, preferably dietary) and vitamin D (800–1000 IU/day), weight-bearing exercise, fall prevention, and avoidance of tobacco and excess alcohol. All patients on bisphosphonates should be counseled about osteonecrosis of the jaw (ONJ) and atypical femoral fractures, though both are rare.
13 Pressure Injuries, Constipation, Sleep & Sensory Decline Syndromes
Pressure Injuries
Staged by the NPUAP/NPIAP system. Risk assessed with the Braden scale (sensory perception, moisture, activity, mobility, nutrition, friction/shear; total 6–23; ≤ 18 = at risk). Prevention: turning q2h, pressure-redistributing surfaces, nutrition optimization, moisture management.
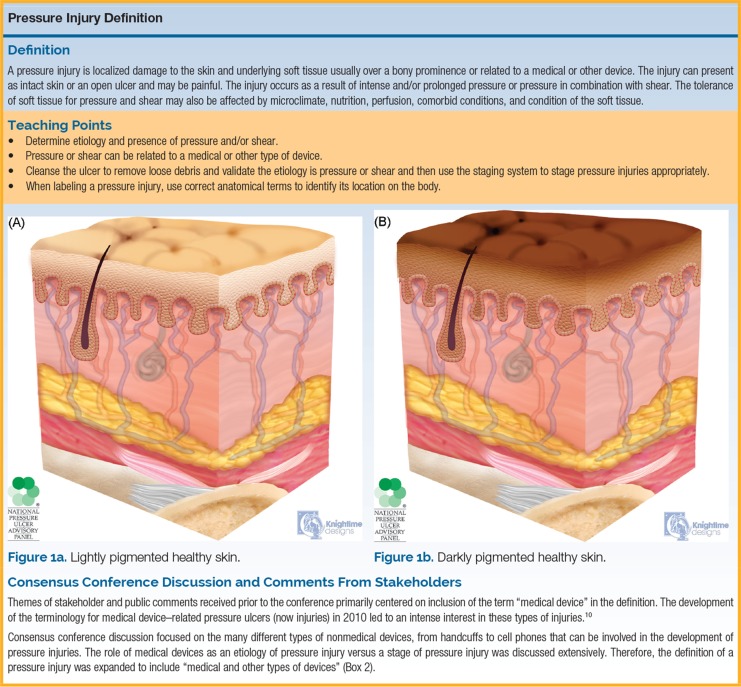
Stage 1: Intact skin with non-blanchable erythema.
Stage 2: Partial-thickness skin loss with exposed dermis (may look like a shallow open ulcer or intact/ruptured blister).
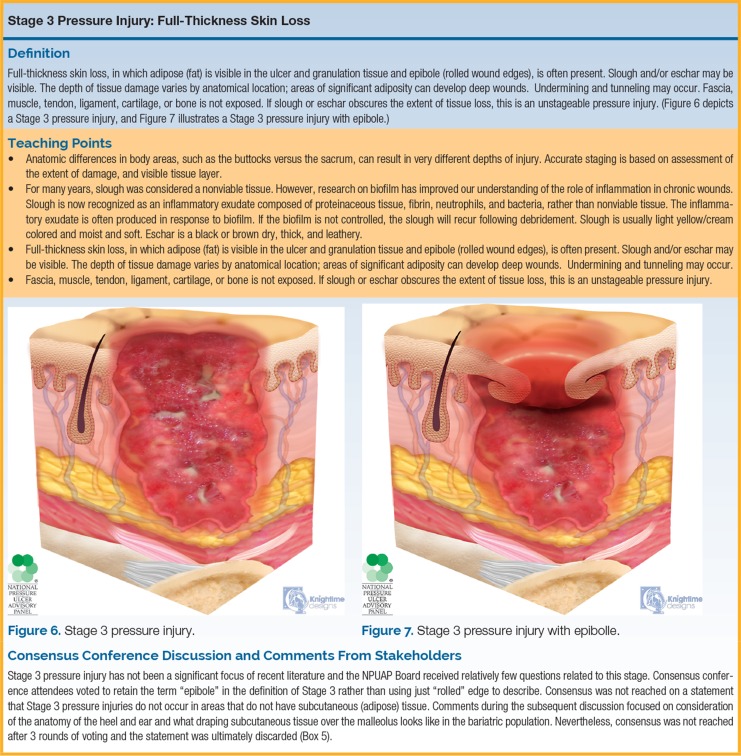
Stage 3: Full-thickness skin loss; subcutaneous fat visible; no exposed bone, tendon, or muscle.
Stage 4: Full-thickness skin and tissue loss with exposed or palpable fascia, muscle, tendon, ligament, cartilage, or bone.
Unstageable: Full-thickness loss obscured by slough or eschar.
Deep Tissue Pressure Injury (DTPI): Persistent non-blanchable deep red, maroon, or purple discoloration of intact or nonintact skin.
Constipation
Very common. Evaluate for alarm features (weight loss, blood, new onset in a patient > 50 without prior history) and secondary causes (hypothyroidism, hypercalcemia, opioids, anticholinergics, iron, calcium channel blockers). Stepwise management: fiber and fluids, osmotic laxatives (PEG 3350, lactulose), stimulants (senna, bisacodyl), and prescription agents (lubiprostone, linaclotide) for refractory cases. Opioid-induced constipation may require peripherally acting mu-opioid antagonists (methylnaltrexone, naloxegol).
Sleep Disorders
Sleep architecture changes with age (less deep sleep, more awakenings). Insomnia evaluation includes sleep hygiene, caffeine and alcohol, medication review, mood, and screening for sleep apnea (very common and underdiagnosed) and restless legs syndrome. CBT-I (cognitive behavioral therapy for insomnia) is first-line. Avoid benzodiazepines and Z-drugs — both Beers-listed. Low-dose trazodone or mirtazapine are more commonly used in practice, with melatonin as a gentler option.
Vision and Hearing
Age-related macular degeneration (dry and wet), cataracts, glaucoma, and diabetic retinopathy are the leading causes of vision loss. Annual eye exams are standard. Presbycusis (age-related sensorineural hearing loss) affects most older adults and strongly contributes to social isolation, depression, and accelerated cognitive decline. Document hearing aid use (or non-use and why). Whisper test and handheld audioscope are office screens.
14 Decisional Capacity, Advance Care Planning & Elder Abuse Ethics
Decisional Capacity
Capacity is the clinical determination that a patient can make a specific medical decision. It has four elements (Appelbaum): (1) understand relevant information; (2) appreciate the situation and its consequences; (3) reason about treatment options; (4) express a choice. Capacity is decision-specific, time-specific, and exists on a continuum — a patient may have capacity for simple decisions but not complex ones. It is distinct from competency, which is a legal determination.
Advance Care Planning
The process of helping patients articulate and document their values and preferences for future care. Documents include advance directive/living will, durable power of attorney for health care (DPOAHC/healthcare proxy), and POLST/MOLST (Physician/Medical Orders for Life-Sustaining Treatment) — actionable medical orders that travel with the patient. Every geriatric note should reference current code status and whether a surrogate decision-maker has been identified.
Elder Abuse & Neglect
Affects approximately 1 in 10 community-dwelling older adults. Types: physical, sexual, emotional/psychological, financial exploitation, neglect, and self-neglect. Red flags: unexplained injuries at various stages of healing, pressure injuries in a patient with an engaged caregiver, poor hygiene, missing assistive devices, caregiver who dominates the visit or refuses to leave, weight loss, dehydration, medication mismanagement, unpaid bills, sudden financial changes. Reporting to Adult Protective Services (APS) is mandatory in most jurisdictions when abuse is suspected. Document the findings objectively, who was in the room, and the APS report if filed.
15 Palliative, Hospice & End-of-Life Care EOL
Palliative care is specialized medical care focused on symptom relief and quality of life at any stage of serious illness, alongside curative or life-prolonging therapy. Hospice is a Medicare benefit for patients with a prognosis of ≤ 6 months if the disease runs its natural course, who agree to forgo curative treatment of the hospice diagnosis.
Hospice Eligibility
The attending and hospice medical director must both certify prognosis. Common non-cancer hospice diagnoses use NHPCO/LCD guidance: advanced dementia (FAST stage 7), end-stage heart failure (NYHA IV with optimal therapy), end-stage COPD (dyspnea at rest, cor pulmonale, ≥ 88% SpO2 on room air or recent admissions), ESRD without dialysis, end-stage liver disease.
Symptom Management at End of Life
Pain: opioids, titrated to effect; bowel regimen mandatory. Dyspnea: low-dose opioids (morphine), oxygen if hypoxic, fan to face. Terminal secretions ("death rattle"): repositioning, glycopyrrolate, scopolamine patch. Agitation/terminal delirium: haloperidol, low-dose midazolam. Nausea: haloperidol, ondansetron, metoclopramide.
Communication Frameworks
Standardized approaches: SPIKES (breaking bad news), NURSE (responding to emotion), REMAP (goals-of-care discussion). Document the content of the conversation, who was present, and the resulting plan.
Decreased intake, decreased responsiveness, mottling of extremities, cool extremities, irregular (Cheyne-Stokes) respirations, terminal secretions, urinary retention or incontinence, and eventual apnea. Family education and support are central.
16 Assessment Tools & Office Procedures Procedures
Geriatrics is a cognitively-demanding specialty with relatively few bedside procedures. The "procedures" are the structured assessments themselves.
Cognitive Screens
3-item recall (apple, table, penny) + clock drawing test. Scoring: recall 0 = impaired; recall 3 = not impaired; recall 1–2 with normal clock = not impaired; recall 1–2 with abnormal clock = impaired. Fast (< 3 min), widely used as an initial screen.
Total 30 points. Domains: visuospatial/executive (5), naming (3), attention (6), language (3), abstraction (2), delayed recall (5), orientation (6). Add 1 point if ≤ 12 years of education. ≥ 26 normal; 18–25 mild cognitive impairment; 10–17 moderate; < 10 severe.
More sensitive than MMSE for MCI and mild AD, especially executive dysfunction. Requires certification to administer as of 2020.
Total 30 points. Orientation to time (5), orientation to place (5), registration (3), attention/calculation (5 — serial 7s or WORLD backward), recall (3), language and praxis (9 — naming, repetition, 3-step command, reading, writing, copying).
≥ 24 normal; 19–23 mild impairment; 10–18 moderate; < 10 severe. Less sensitive than MoCA for MCI; copyright-protected.
11 items, 30 points. Adjusted for education level. Validated against MCI. Freely available alternative to MMSE.
Mood Screens
15 yes/no questions oriented to mood rather than somatic symptoms. 0–4 normal; 5–8 mild; 9–11 moderate; 12–15 severe depression. Validated in cognitively intact older adults; less reliable in moderate-severe dementia.
9 items over the past 2 weeks, scored 0–3 each. 0–4 none; 5–9 mild; 10–14 moderate; 15–19 moderately severe; 20–27 severe. Includes item 9 on suicidal ideation — any positive triggers a safety assessment.
Functional Scales
Six basic ADLs: Bathing, Dressing, Toileting, Transferring, Continence, Feeding. Scored 1 (independent) or 0 (dependent) each; total 0–6.
Eight instrumental ADLs: telephone, shopping, food preparation, housekeeping, laundry, transportation, medication management, finances. Total 0–8 (women); 0–5 is often used for men excluding traditionally gendered items.
The Geriatric Annual Wellness Visit bundles cognitive screening, depression screening, fall risk assessment, functional assessment, advance care planning, and medication review — all billable components under Medicare. Scribes must document the specific screens performed, the tool used, the score, and the resulting plan.
17 Treatment Monitoring & Adverse Drug Events Safety
Adverse drug events (ADEs) are among the most common reasons for geriatric hospitalization. High-risk drug classes are warfarin, insulins, oral antiplatelets/anticoagulants, and oral hypoglycemics — together responsible for nearly two-thirds of ADE-related hospitalizations in older adults.
Monitoring Essentials
- Cholinesterase inhibitors: GI side effects (nausea, diarrhea, weight loss), bradycardia, syncope; check HR and weight.
- Memantine: renal dose adjustment if CrCl < 30.
- Lecanemab / donanemab: serial MRI for ARIA; APOE genotyping before initiation.
- SSRIs: hyponatremia (SIADH), QT prolongation (citalopram), GI bleeding when combined with antiplatelets/NSAIDs, falls.
- Antipsychotics in dementia: black-box mortality warning; minimum effective dose, shortest duration, documented informed consent.
- Warfarin: INR goal typically 2–3 (2.5–3.5 for mechanical mitral valve); frequent monitoring; many food/drug interactions.
- DOACs: renal dose adjustment (apixaban, rivaroxaban, dabigatran, edoxaban); no routine monitoring but check CrCl at least annually.
- Bisphosphonates: dental exam before initiation if risk factors; avoid if CrCl < 30–35 depending on agent.
- Denosumab: hypocalcemia risk; never discontinue abruptly without transition to bisphosphonate.
- Diuretics: electrolytes, renal function, orthostatic vitals.
- Opioids: bowel regimen, sedation, respiratory depression, fall risk; avoid combination with benzodiazepines or gabapentinoids.
Anytime a new medication is started or dose changed, document the indication, starting dose, titration plan, monitoring plan (what labs, how often), and the specific adverse effects the patient and caregiver were counseled about. Missing these is the most common chart deficiency in geriatrics.
18 Labs, Imaging & Diagnostics Diagnostics
Core Labs
CBC, CMP (creatinine, eGFR, glucose, electrolytes, liver panel), TSH, B12, vitamin D (25-OH), A1C (diabetics), lipid panel, PSA (shared decision-making after 70), UA, INR if on warfarin, ferritin/iron studies for anemia. Any cognitive decline, weight loss, or new fatigue triggers TSH and B12 at minimum.
Imaging
MRI brain is preferred for new cognitive complaints; CT is acceptable if MRI contraindicated. DXA for bone density. Echocardiography for new murmurs, heart failure, syncope. Chest X-ray for new cough, weight loss, or suspected infection. Bladder ultrasound (bedside) for post-void residual. Imaging in the frail elderly is always weighed against the risk of false positives, contrast nephropathy, and downstream invasive workup.
Interpreting Vital Signs in the Elderly
Fever blunted or absent — a temperature ≥ 100°F (37.8°C) or ≥ 2°F above baseline is clinically significant. Tachycardia may be absent due to beta-blockers or autonomic dysfunction. Hypotension may represent sepsis at "normal" numbers for younger patients. Always anchor to baseline.
19 Medications — High-Yield Geriatric Drug Classes Meds
Dementia
| Drug | Brand | Class / Mechanism | Key Notes |
|---|---|---|---|
| Donepezil | Aricept | Cholinesterase inhibitor | 5–10 mg QHS; GI side effects; bradycardia |
| Rivastigmine | Exelon | Cholinesterase inhibitor | Patch preferred (fewer GI side effects); also approved in PDD |
| Galantamine | Razadyne | Cholinesterase inhibitor | BID dosing; ER available |
| Memantine | Namenda | NMDA antagonist | Moderate-severe AD; renal dose; well tolerated |
| Lecanemab | Leqembi | Anti-amyloid mAb | IV Q2wk; early AD; ARIA monitoring; APOE testing |
| Donanemab | Kisunla | Anti-amyloid mAb | IV Q4wk; early AD; ARIA monitoring |
Antidepressants Safe in Elderly
| Drug | Class | Notes |
|---|---|---|
| Sertraline | SSRI | First-line; minimal drug interactions |
| Escitalopram | SSRI | First-line; up to 10 mg in elderly |
| Citalopram | SSRI | Cap 20 mg/day (QT prolongation) |
| Mirtazapine | NaSSA | Useful with insomnia and poor appetite; sedation |
| Duloxetine | SNRI | Good for comorbid neuropathic pain |
| Bupropion | NDRI | Activating; avoid if seizures or eating disorder |
| Avoid: paroxetine, amitriptyline, imipramine | Anticholinergic — Beers list |
Anticoagulants in the Elderly
| Drug | Class | Notes |
|---|---|---|
| Apixaban (Eliquis) | DOAC, Xa inhibitor | Dose reduction (2.5 mg BID) if 2 of: age ≥ 80, weight ≤ 60 kg, creatinine ≥ 1.5 |
| Rivaroxaban (Xarelto) | DOAC, Xa inhibitor | Once daily with food; CrCl-dependent dosing |
| Dabigatran (Pradaxa) | DOAC, direct thrombin | GI side effects; avoid if CrCl < 30 |
| Edoxaban (Savaysa) | DOAC, Xa inhibitor | Not for CrCl > 95; renal-dose dependent |
| Warfarin | Vitamin K antagonist | Used when DOAC contraindicated (mechanical valve, severe CKD) |
Other High-Yield Classes
Antihypertensives: thiazides, ACEI/ARB, calcium channel blockers are mainstays; beta-blockers if specific indication (HF, CAD, AF). Statins: moderate-intensity is usually appropriate; continuation in very frail/hospice often discontinued. Diabetes: metformin first-line if eGFR ≥ 30; avoid long-acting sulfonylureas; individualize A1C target (< 7.5–8% in most older adults; < 8.5% in frail with limited life expectancy). Pain: acetaminophen first-line (up to 3 g/day); topical NSAIDs; short-course oral NSAIDs only with GI protection and careful renal monitoring; opioids as a last resort.
Immunization Schedule for Older Adults
Annual influenza (high-dose or adjuvanted preferred for ≥ 65). COVID-19 per current CDC schedule. Pneumococcal: PCV15 or PCV20 for adults ≥ 65, followed by PPSV23 if PCV15 was used. Shingles: Shingrix 2-dose series for adults ≥ 50. Tdap once, then Td or Tdap every 10 years. RSV for adults ≥ 75 and 60–74 at increased risk, per current ACIP guidance.
20 Classification Systems — Full Enumerations Reference
Clinical Frailty Scale (Rockwood, CFS)
| Level | Label | Description |
|---|---|---|
| 1 | Very fit | Robust, active, energetic, exercise regularly |
| 2 | Well | No active disease; less fit than category 1 |
| 3 | Managing well | Medical problems well controlled; not regularly active |
| 4 | Vulnerable | Symptoms limit activities; "slowed up" |
| 5 | Mildly frail | More evident slowing; needs help with IADLs |
| 6 | Moderately frail | Needs help with all outside activities and some ADLs (bathing, dressing) |
| 7 | Severely frail | Completely dependent for personal care; stable |
| 8 | Very severely frail | Completely dependent, approaching end of life |
| 9 | Terminally ill | Life expectancy < 6 months, not otherwise severely frail |
FAST Scale (Functional Assessment Staging Tool, for AD)
| Stage | Clinical |
|---|---|
| 1 | Normal adult |
| 2 | Subjective cognitive complaints |
| 3 | Mild decline, difficulty with demanding work |
| 4 | Moderate decline, difficulty with complex tasks (finances, travel) |
| 5 | Moderately severe — needs help choosing clothing |
| 6a–e | Severe — needs help dressing, bathing, toileting; incontinence |
| 7a–f | Very severe — limited speech (≤ 6 words), ambulation lost, unable to sit up, smile, or hold head up. FAST 7 is the hospice-eligibility cutoff for advanced dementia. |
Braden Scale (Pressure Injury Risk)
Six subscales (sensory perception, moisture, activity, mobility, nutrition — each 1–4; friction/shear 1–3). Total 6–23. ≤ 9 severe risk; 10–12 high risk; 13–14 moderate; 15–18 mild; > 18 minimal risk.
21 Physical Exam — The Geriatric Exam Exam
The geriatric physical exam is longer and more function-focused than the standard adult exam. Core components:
General: well-appearing, cachectic, frail, groomed, appropriate for age; cognitive engagement.
Vitals: BP supine and standing (orthostatic drop defined as ≥ 20 mmHg systolic or ≥ 10 mmHg diastolic within 3 minutes of standing), HR, RR, SpO2, temperature, weight, BMI.
Sensory: vision (near and distance), hearing (whisper test), dentition.
HEENT: oral mucosa, dentition, thyroid.
Cardiopulmonary: rate, rhythm, murmurs, S3/S4, JVD, edema, lungs.
Abdomen: bowel sounds, tenderness, masses, organomegaly, palpable bladder.
Musculoskeletal: joint swelling, deformity, ROM, kyphosis, muscle bulk (temporalis, thenar wasting).
Skin: turgor, bruising, ulcers, pressure points, lesions.
Neurologic: mental status, cranial nerves, motor, sensation, reflexes, coordination (finger-to-nose, heel-to-shin).
Gait: Timed Up and Go; observe initiation, stride, symmetry, turning, use of assistive device.
Cognitive: formal screen (Mini-Cog/MoCA/MMSE/SLUMS) with score and date.
22 Abbreviations Master List Reference
Syndromes & Diagnoses
Assessments & Scales
Care Planning & Systems
23 Sample HPI Templates Templates
These templates show the kind of documentation expected in a geriatrics clinic. Use them as frameworks to internalize the content and rhythm of a geriatric HPI.
"Mrs. [Name] is an 82-year-old right-handed widowed female with a history of HTN, hyperlipidemia, and osteoarthritis who presents with her daughter for evaluation of progressive forgetfulness over the past 12–18 months. The daughter reports that her mother repeats questions within the same conversation, has missed two physician appointments, has difficulty managing her checkbook, and once left the stove on. The patient acknowledges "some forgetfulness" but feels she is "fine." No focal neurologic symptoms, no hallucinations, no parkinsonian features, no stepwise decline. Independent in basic ADLs; requires assistance with finances, medications, and transportation (IADLs 5/8). No new medications. No recent infection or hospitalization. PHQ-9 today 4/27. Mini-Cog in office: recall 1/3, abnormal clock — positive screen. MoCA 19/30 (lost points on delayed recall, executive function, visuospatial). Plan: MRI brain, TSH, B12, CMP, CBC, vitamin D; dementia workup; discuss driving, advance care planning, and caregiver support at next visit."
"Mr. [Name] is a 79-year-old male with a history of PAD, T2DM, HTN, and BPH who presents for follow-up after a mechanical fall at home 5 days ago. He tripped on a rug in the hallway while walking to the bathroom at night; no syncope, no prodrome, no chest pain or palpitations. He struck his right hip but not his head; denies LOC. No head strike. Evaluated in the ED; X-rays negative; discharged home. Since then he has been fearful of walking and is using a cane his son purchased at a pharmacy (unfitted). No new medications. Home meds include tamsulosin, glipizide, lisinopril, atorvastatin, aspirin, and as-needed zolpidem for sleep. Orthostatic vitals today: supine 134/78 HR 72, standing (1 min) 108/64 HR 82 — positive. TUG today 16 sec. Vision screen reveals uncorrected presbyopia; last eye exam 3 years ago. Plan: deprescribe zolpidem and glipizide (switch to glipiZIDE-alternative without hypoglycemia risk), reduce tamsulosin trial, PT referral for gait training and proper walker fitting, home safety evaluation, vitamin D 1000 IU daily, optometry referral."
"Mrs. [Name] is an 84-year-old female referred for comprehensive medication review. She takes 14 medications including lisinopril, amlodipine, metoprolol, HCTZ, atorvastatin, metformin, glipizide, omeprazole, diphenhydramine QHS, oxybutynin, gabapentin, sertraline, alendronate, and tramadol. Recently she has noted increased confusion in the evenings, two near-falls, constipation, and dry mouth. She lives alone in a first-floor apartment. Daughter checks in daily. Mini-Cog 2/3 with normal clock. Orthostatic positive (drop 24 mmHg systolic). Current Beers-listed drugs identified: diphenhydramine, oxybutynin, glipizide (long-acting sulfonylurea), PPI > 8 weeks without clear indication, and tramadol with SSRI (serotonin syndrome risk). Deprescribing plan discussed with patient and daughter: stop diphenhydramine (melatonin substitute), taper oxybutynin and trial mirabegron, transition glipizide to low-dose metformin adjustment, taper omeprazole to PRN, discontinue tramadol with scheduled acetaminophen substitution. Follow up in 4 weeks."
"Mr. [Name] is an 88-year-old male with moderate Alzheimer disease (MoCA 14/30, FAST 5), NYHA III heart failure, CKD stage 4, and remote prostate cancer who presents with his son (DPOAHC) for advance care planning discussion. Patient is oriented to self only; son provides primary history. Currently lives in assisted living. Recent hospitalization for CHF exacerbation 6 weeks ago. Discussion covered disease trajectory, goals of care, prognosis in the context of advanced HF and progressive dementia. Patient expresses he would not want "to be on machines." Son reports prior conversations consistent with comfort-focused approach. Capacity for this decision assessed as limited; decision made by son as DPOAHC, consistent with patient's prior expressed wishes. POLST completed: DNR/DNI, no hospitalization for comfort-manageable issues, oral medications only for symptom relief, no artificial nutrition. Hospice referral initiated given NYHA IV trajectory, cachexia, and cognitive decline."
"Mrs. [Name] is an 86-year-old female (baseline MoCA 23/30 at PCP visit 2 months ago) admitted 2 days ago for UTI and treated with ceftriaxone. Geriatrics consulted for acute confusion that began the evening of admission. Nursing reports fluctuating attention, pulling at IV, nocturnal agitation, and daytime somnolence. Patient is disoriented to time and place, unable to recite months of year backward, rambling speech. CAM positive (acute onset, inattention, disorganized thinking, fluctuating level of consciousness). Review of medication administration record reveals scheduled diphenhydramine, PRN lorazepam (received twice overnight), and opioid for mild pain. Workup: UA improving, CMP with mild hyponatremia (Na 131), no new imaging indicated. Plan: discontinue diphenhydramine and lorazepam, switch PRN pain to scheduled acetaminophen, reorientation bundle, glasses and hearing aids at bedside, family at bedside, mobilize with PT, restore sleep-wake cycle, correct hyponatremia gently, avoid antipsychotics unless severe agitation."
"Mrs. [Name] is a 77-year-old female with HTN and osteoarthritis who presents with 4 months of progressive urinary leakage. She reports sudden urges with inability to reach the bathroom in time, estimates 3–4 episodes daily, and wears pads continuously. She also leaks small amounts with coughing and sneezing. No dysuria, hematuria, or fever. No constipation. On HCTZ for 5 years. Fluid intake approximately 2 L/day, including 3 cups of coffee. Lives at home, independent in ADLs. Exam: abdomen benign, no suprapubic tenderness, pelvic exam reveals mild atrophic changes without prolapse. UA negative. Bedside bladder scan PVR 40 mL. Assessment: mixed urinary incontinence, urge-predominant. Plan: bladder diary, caffeine reduction, timed voiding, pelvic floor exercises, topical vaginal estrogen, consider mirabegron (avoid antimuscarinics given mild cognitive concerns); reassess in 6 weeks."
24 References & Sources
Clinical Practice Guidelines
Landmark Evidence
van Dyck CH, et al. Lecanemab in Early Alzheimer's Disease (CLARITY AD). N Engl J Med. 2023.
Sims JR, et al. Donanemab in Early Symptomatic Alzheimer Disease (TRAILBLAZER-ALZ 2). JAMA. 2023.
Inouye SK, et al. Clarifying confusion: the Confusion Assessment Method. Ann Intern Med. 1990.
Diagram & Figure Sources
Figure 1: Dementia — Normal vs Atrophic Brain. BruceBlaus / Blausen Medical, Wikimedia Commons. CC BY 3.0.
Figure 2: Older Adult Using a Walker. Wikimedia Commons. Public domain / CC BY-SA.
The best geriatrics scribes understand that the note is not a list of diagnoses — it is a story about a whole person. When the attending says "MoCA 22, IADLs 5/8, one fall in the past year, living with daughter," a great scribe already knows this is mild cognitive impairment or early dementia, early functional decline, increased fall risk, and a supportive caregiver, and organizes the note accordingly. When the attending says "let's stop the diphenhydramine and the oxybutynin," the scribe understands both are on the Beers list and charts the rationale without being told.
Geriatrics moves slowly in clinic and quickly in the hospital. Learn the assessment tools cold. Memorize the Beers high-yield categories. Know the difference between delirium and dementia in one sentence. Always capture function, cognition, mood, mobility, and goals of care. When in doubt, ask the family who is in the room — the collateral history is often more accurate than what the patient reports.
Welcome to geriatrics. It is the specialty of whole-person medicine, and it is the specialty of the future.