Hematology / Oncology
Every diagnosis, condition, procedure, medication, classification, and documentation framework you need to survive and thrive in a heme/onc clinic.
All diagrams on this page are sourced from published educational or institutional materials (OpenStax, Wikimedia Commons, Blausen Medical, NCI) rather than AI generation. Each figure caption links to the original source, and all diagram and guideline citations are collected in the references section at the bottom.
01 Hematopoiesis & Bone Marrow Essentials
Hematology and oncology are two overlapping but distinct practices joined administratively because both depend on blood, bone marrow, and the cellular mechanics of proliferation. Hematology manages diseases of red cells, white cells, platelets, and coagulation. Oncology manages solid tumors and, in heme-onc clinics, liquid malignancies (leukemias, lymphomas, myeloma). A scribe in this specialty will document everything from a simple iron-deficiency follow-up to a newly diagnosed stage IV lung cancer starting immunotherapy, often in the same morning.
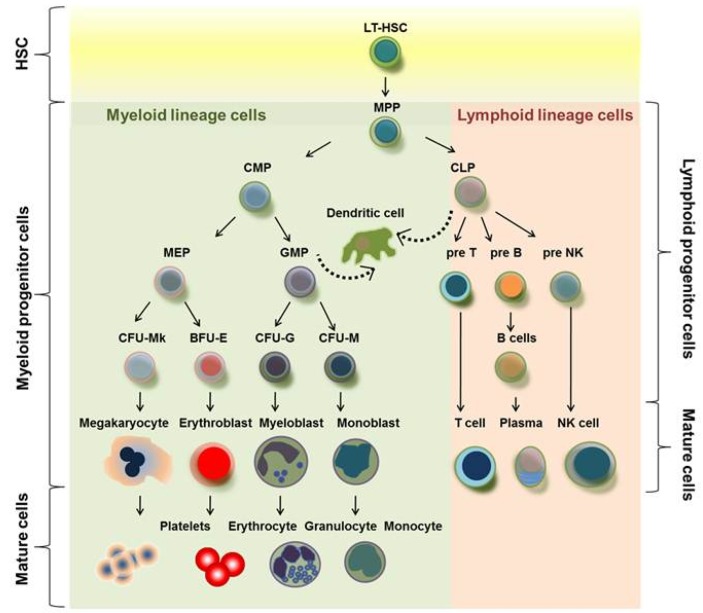
The Bone Marrow — Where It All Starts
All circulating blood cells originate from hematopoietic stem cells (HSCs) in the bone marrow. In adults, active (red) marrow is concentrated in the axial skeleton — vertebrae, sternum, ribs, pelvis, skull, and proximal long bones. HSCs differentiate along two major lineages: the myeloid line (erythrocytes, granulocytes, monocytes, platelets) and the lymphoid line (B cells, T cells, NK cells). Nearly every hematologic malignancy can be traced to a clonal expansion at one of these branches.
The Cells You Will Chart Every Day
Red blood cells (RBCs) carry oxygen via hemoglobin. A normal hemoglobin is roughly 13.5–17.5 g/dL in men and 12.0–15.5 g/dL in women. Mean corpuscular volume (MCV) classifies anemia as microcytic (< 80 fL), normocytic (80–100 fL), or macrocytic (> 100 fL). White blood cells (WBCs) include neutrophils (infection fighters), lymphocytes (adaptive immunity), monocytes, eosinophils, and basophils; the absolute neutrophil count (ANC) drives nearly every chemotherapy decision. Platelets are fragments of megakaryocytes that form primary hemostatic plugs; normal is 150–450 × 10³/µL.
Learn to read a CBC at a glance. The line you will hear most is “H/H is 8.2 and 24.6, platelets 72, ANC 0.4” — translate instantly: severe anemia, thrombocytopenia, and severe neutropenia. That combination triggers transfusion discussions, chemo dose holds, and isolation precautions within the same sentence.
02 Scribe Documentation Framework
Heme/onc notes are problem-oriented and cycle-aware. Every note references where the patient is in their treatment journey: new consult, pre-treatment workup, on active therapy (cycle X day Y), surveillance, progression, or end-of-life care.
Chief Complaint: “Cycle 3 day 1 FOLFOX,” “new anemia, Hgb 7.8,” “follow-up DLBCL s/p R-CHOP × 6.”
HPI: Always capture disease status, date of diagnosis, histology/stage, all prior lines of therapy (with start/stop dates and reason stopped), current regimen cycle/day, and interval symptoms. Do not forget B symptoms (fevers, drenching night sweats, unintentional weight loss > 10% over 6 months) in every lymphoma/leukemia note.
Toxicity Review: Review chemo side effects by system — nausea/vomiting, mucositis, diarrhea/constipation, neuropathy (tingling, numbness, buttoning shirts), fatigue, rash, infections, bleeding. Grade each using CTCAE when the clinician mentions a grade.
Performance Status: ECOG and Karnofsky should appear in every oncology note. Performance status drives treatment eligibility as much as stage does.
Vitals & Weight: Weight is critical because body surface area (BSA) determines chemo dosing. A 5 kg change triggers dose recalculation.
Exam: Lymph node survey (cervical, supraclavicular, axillary, inguinal), abdominal exam for hepatosplenomegaly, port site inspection, skin (rash, bruising, petechiae), mucositis, neuropathy testing.
Labs: CBC with differential, CMP, LDH, uric acid, tumor markers when applicable. Always document ANC, platelets, creatinine, and bilirubin — these are the four numbers that determine whether chemo can be given today.
Organized by problem: (1) primary malignancy with stage, current line of therapy, response; (2) treatment toxicities; (3) supportive care needs (antiemetics, growth factors, transfusions); (4) comorbidities affecting treatment. End with disposition: proceed with cycle, dose-reduce, hold, or change regimen.
Learn the shorthand “C#D#” (Cycle 3 Day 1 = C3D1) and always chart it. Oncologists speak in cycles, not calendar dates. Also learn “s/p” (status post) because every onc HPI will contain a string like “s/p R-CHOP × 6 completed 03/2025, in CR by PET.”
03 Iron Deficiency & Anemia of Chronic Disease Benign Heme
Iron deficiency anemia (IDA) is the most common anemia worldwide. Causes include GI blood loss (colon cancer must be ruled out in any adult male or postmenopausal female with new IDA), menstrual loss, poor absorption (celiac, post-bariatric surgery, PPI use), and dietary deficiency. Patients present with fatigue, pallor, pica, restless legs, koilonychia, and exercise intolerance.
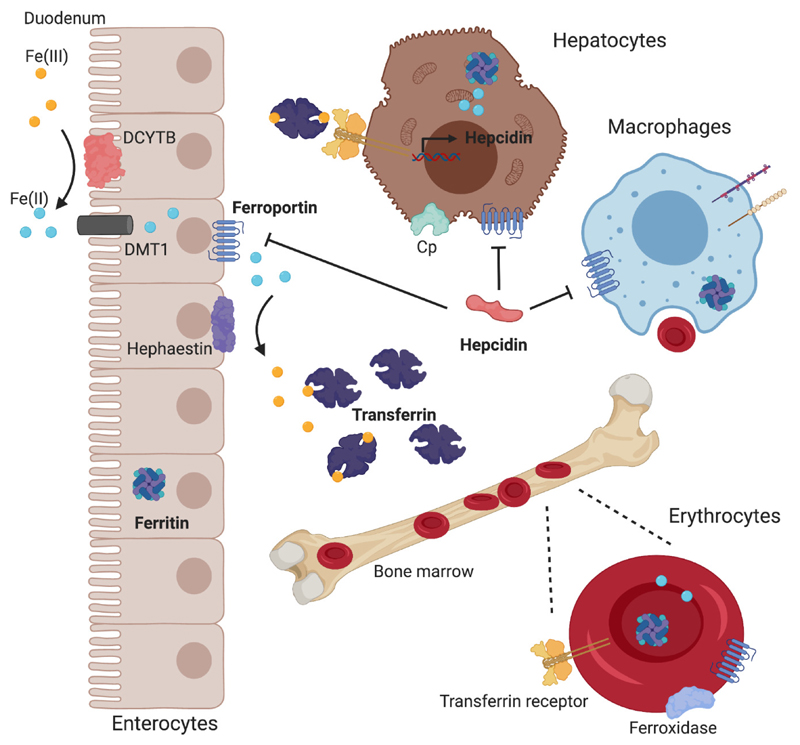
Labs show microcytic (MCV < 80), hypochromic anemia with low ferritin (the single most useful test), low serum iron, high TIBC, and low transferrin saturation. A ferritin < 30 ng/mL is diagnostic; < 100 ng/mL in the setting of inflammation still suggests iron deficiency. The peripheral smear shows small, pale (hypochromic) red cells, often with anisocytosis (elevated RDW) and occasional pencil cells. Reticulocyte count is inappropriately low for the degree of anemia. Management is oral iron (ferrous sulfate 325 mg every other day has better absorption than daily dosing) or IV iron (iron sucrose, ferric carboxymaltose, ferric derisomaltose) for intolerance, malabsorption, severe deficiency, or inflammatory bowel disease. Expect hemoglobin to rise 1–2 g/dL per month on oral therapy. New-onset IDA in any adult without an obvious source mandates GI workup (EGD + colonoscopy). In premenopausal women, gynecologic evaluation for heavy menstrual bleeding is equally important.
Start with MCV. Microcytic: iron deficiency, thalassemia, anemia of chronic disease (sometimes), sideroblastic anemia, lead toxicity. Normocytic: acute blood loss, ACD, early iron deficiency, hemolysis, marrow failure, renal disease. Macrocytic: B12/folate deficiency, hypothyroidism, liver disease, alcohol, myelodysplasia, drugs (methotrexate, hydroxyurea, zidovudine). Always order reticulocyte count: hyporegenerative (low retic) suggests production failure; hyperregenerative (high retic) suggests hemolysis or blood loss.
Anemia of chronic disease (ACD) — also called anemia of inflammation — is a normocytic (sometimes mildly microcytic) anemia driven by hepcidin-mediated iron sequestration. Ferritin is normal or elevated, TIBC is low, and transferrin saturation is low-normal. It accompanies chronic infection, autoimmune disease, CKD, and malignancy. Treatment is directed at the underlying disease; erythropoiesis-stimulating agents (ESAs) are used in CKD and select cancer patients (ASCO/ASH ESA guidelines).
Any adult with new IDA gets a colonoscopy referral documented in the plan — this is the diagnosis that most commonly uncovers an occult right-sided colon cancer.
04 B12 / Folate Deficiency & Megaloblastic Anemia Benign Heme
Macrocytic anemia (MCV > 100) with hypersegmented neutrophils on smear suggests a megaloblastic process. Vitamin B12 (cobalamin) deficiency is most commonly due to pernicious anemia (autoimmune loss of intrinsic factor), terminal ileum disease (Crohn’s, resection), strict vegan diet, or metformin/PPI use. Unlike folate deficiency, B12 deficiency causes neurologic symptoms: paresthesias, loss of proprioception/vibration, subacute combined degeneration of the cord, and cognitive changes. Diagnosis: low B12, elevated methylmalonic acid (MMA) and homocysteine. Treatment: IM cyanocobalamin 1000 mcg weekly × 4 then monthly, or high-dose oral 1000–2000 mcg daily.
Folate deficiency mimics B12 deficiency in labs but does not cause neurologic findings. Causes include alcoholism, pregnancy, methotrexate, and poor diet. Homocysteine is elevated but MMA is normal. Always replete B12 before folate — giving folate alone in B12 deficiency can mask the anemia while neurologic damage progresses.
05 Hemolytic Anemias Benign Heme
Hemolysis is premature RBC destruction. The labs are stereotyped: low haptoglobin, elevated LDH, elevated indirect bilirubin, and elevated reticulocyte count. A peripheral smear often clinches the diagnosis (spherocytes, schistocytes, bite cells).
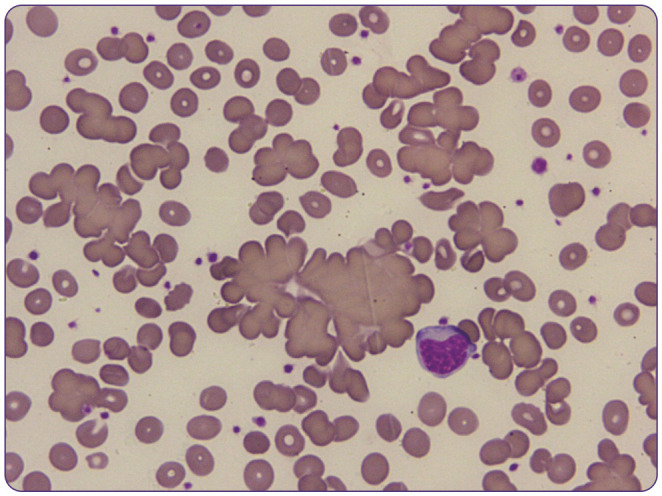
X-linked enzyme defect leaving RBCs vulnerable to oxidative stress. Hemolysis is triggered by fava beans, sulfa drugs, dapsone, primaquine, nitrofurantoin, and infection. Smear shows bite cells and Heinz bodies. Management: avoid triggers, supportive care during crises.
Warm AIHA (IgG, extravascular, spleen): associated with CLL, lupus, lymphoma, drugs (methyldopa). Treatment: corticosteroids, rituximab, splenectomy. Cold agglutinin disease (IgM, intravascular): associated with Mycoplasma, EBV, Waldenström’s. Treat underlying cause, avoid cold, rituximab ± sutimlimab. Both test positive on direct antiglobulin test (DAT / Coombs).
Membrane defect (ankyrin, spectrin). Presents with chronic hemolysis, splenomegaly, pigmented gallstones, aplastic crisis with parvovirus B19. Smear shows spherocytes, increased MCHC. Treatment: folate supplementation, splenectomy in severe cases.
06 Sickle Cell Disease & Thalassemia Benign Heme
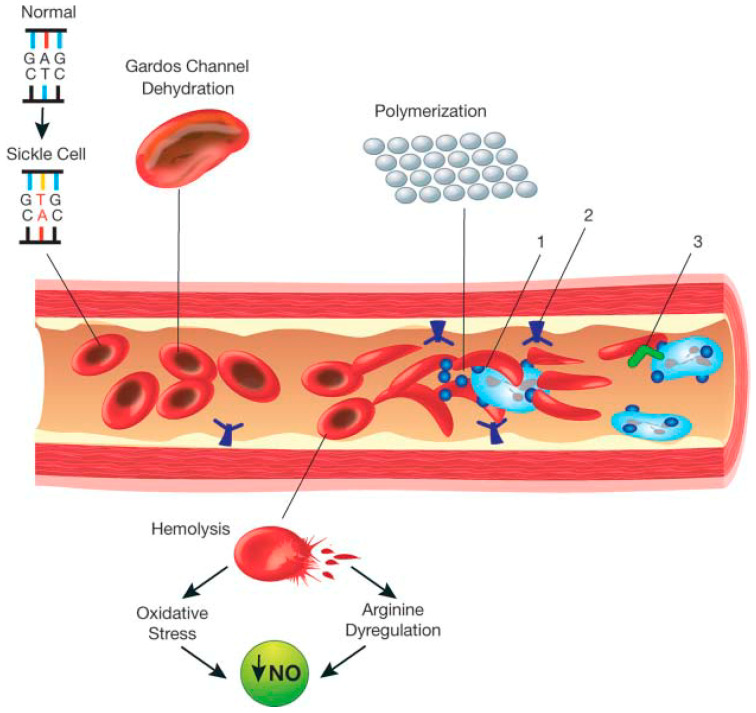
Sickle cell disease (SCD) is an autosomal recessive point mutation (Glu→Val at position 6 of beta-globin) producing HbS, which polymerizes in deoxygenated states, deforming RBCs into rigid sickles that occlude microvasculature and hemolyze. Patients present with vaso-occlusive crises (bone pain, chest, abdomen), acute chest syndrome (new infiltrate + fever/respiratory symptoms — a leading cause of mortality), splenic sequestration, aplastic crisis (parvovirus B19), stroke, priapism, avascular necrosis of the femoral head, and chronic kidney disease.
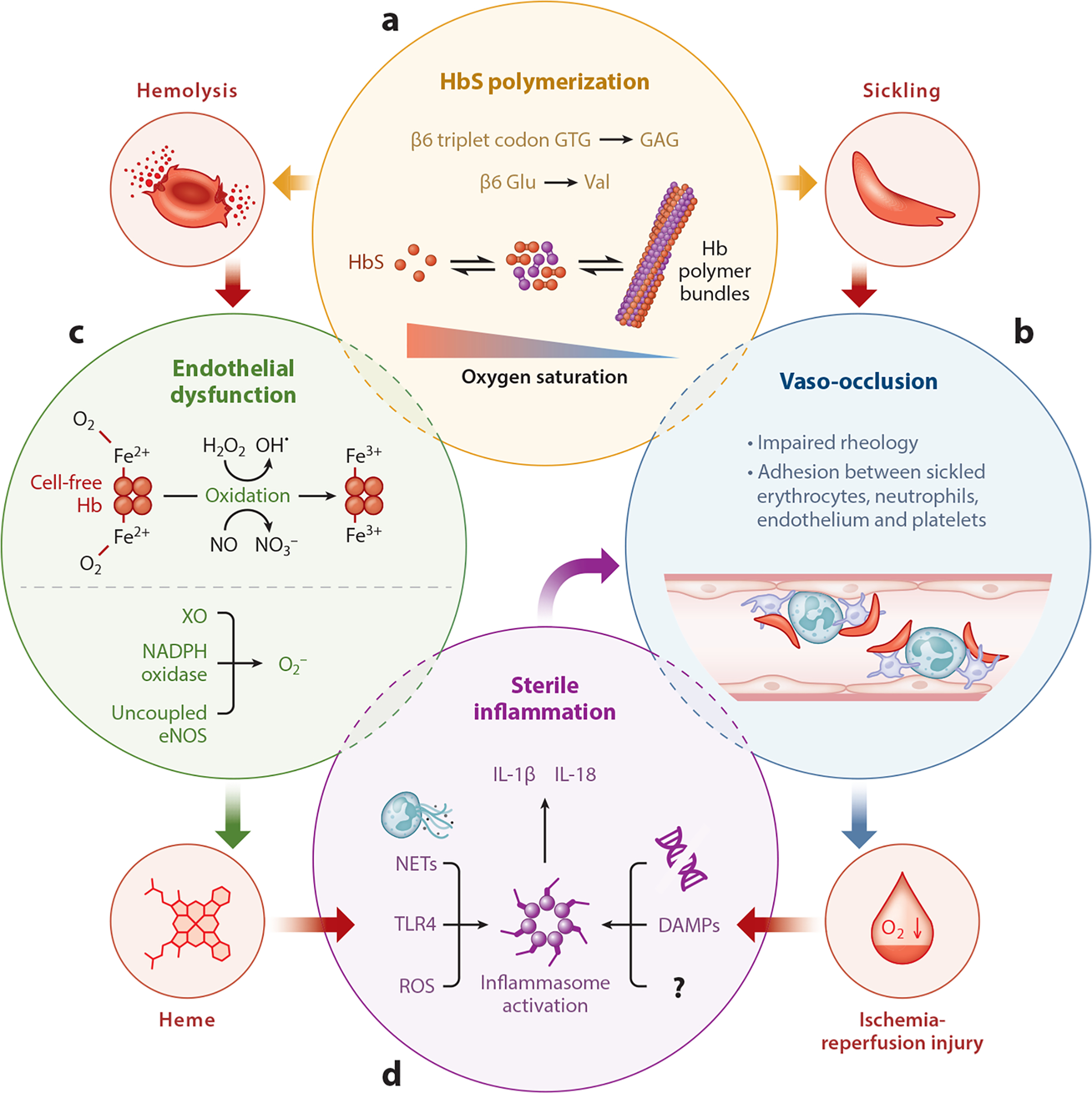
Diagnosis: hemoglobin electrophoresis shows HbSS (disease) or HbAS (trait). Management includes hydroxyurea (increases fetal hemoglobin, reduces crisis frequency — the cornerstone disease-modifying drug), L-glutamine, crizanlizumab, voxelotor, simple or exchange transfusion, and allogeneic stem cell transplant (curative). Pain crises: IV fluids, opioids, oxygen, incentive spirometry. Acute chest syndrome requires aggressive transfusion/exchange. Prophylaxis: penicillin in children, pneumococcal and meningococcal vaccines, folic acid.
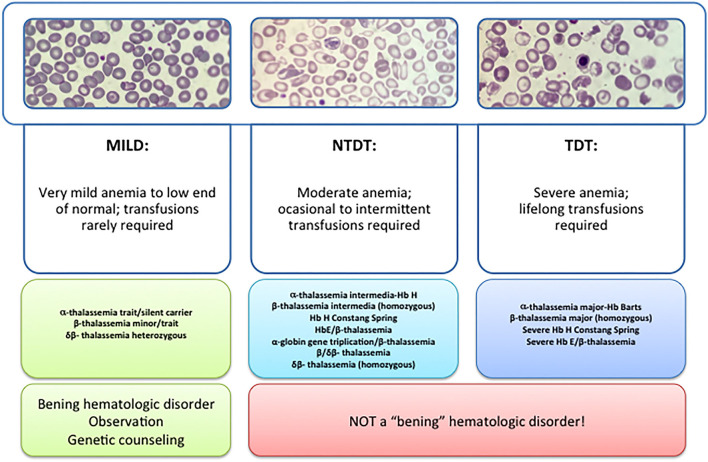
Thalassemias are inherited defects in alpha or beta globin chain production, producing microcytic anemia with normal iron studies. Beta-thalassemia major (Cooley anemia) causes transfusion dependence, iron overload, and hepatosplenomegaly. Alpha thalassemia (1–4 gene deletions) ranges from silent carrier to Hb Barts (hydrops fetalis). Diagnosis: elevated HbA2 in beta-thal, electrophoresis/genetic testing. Management: transfusion support, iron chelation (deferasirox, deferoxamine), luspatercept, stem cell transplant.
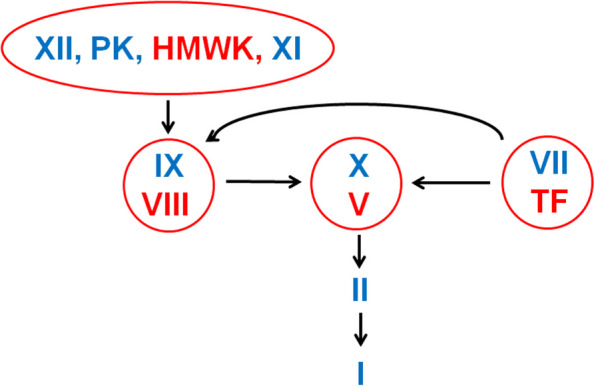
07 Bleeding Disorders Benign Heme
Isolated thrombocytopenia from antiplatelet autoantibodies. Diagnosis of exclusion. Treat if platelets < 30 or bleeding: corticosteroids, IVIG, anti-D, rituximab, TPO receptor agonists (romiplostim, eltrombopag), splenectomy. Chronic ITP in adults; often self-limited in children.
ADAMTS13 deficiency (< 10%) causes unrestrained vWF multimers → platelet microthrombi. Classic pentad: MAHA (schistocytes), thrombocytopenia, fever, renal dysfunction, neurologic changes. Treatment is urgent plasma exchange (PLEX), steroids, caplacizumab, rituximab. Do not transfuse platelets — can worsen thrombosis.
Shiga toxin (E. coli O157:H7) in children; atypical HUS is complement-mediated. Triad: MAHA, thrombocytopenia, AKI. Treatment: supportive; eculizumab for atypical HUS.
Systemic activation of coagulation consuming clotting factors and platelets. Triggers: sepsis, malignancy (APL especially), trauma, obstetric catastrophe. Labs: thrombocytopenia, prolonged PT/PTT, low fibrinogen, elevated D-dimer, schistocytes. Treatment: address cause, transfuse platelets/FFP/cryoprecipitate as needed.
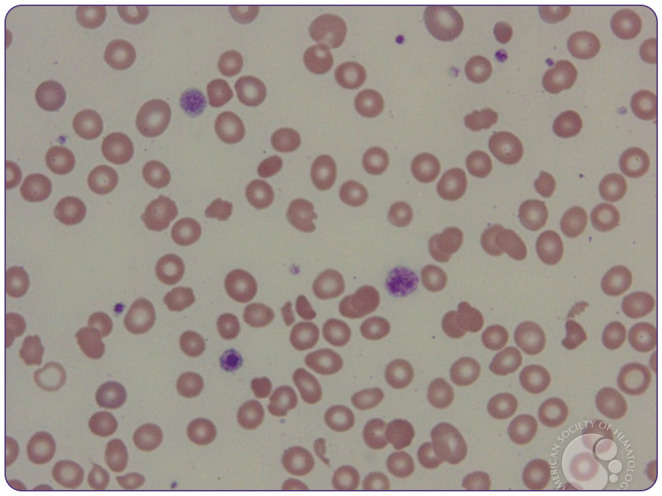
Hemophilia A (factor VIII deficiency) and hemophilia B (factor IX deficiency) are X-linked bleeding disorders presenting with hemarthroses, muscle bleeds, and post-surgical bleeding. Prolonged aPTT with normal PT. Treatment: factor replacement, emicizumab (bispecific antibody for hemophilia A), gene therapy. Von Willebrand disease (vWD) is the most common inherited bleeding disorder; types 1 (quantitative), 2 (qualitative), 3 (severe). Treatment: DDAVP (type 1), vWF/factor VIII concentrates, aminocaproic acid.
08 VTE & Anticoagulation Benign Heme
Venous thromboembolism — DVT and PE — is one of the most common heme consults. Virchow triad: stasis, endothelial injury, hypercoagulability. Provoked VTE occurs in the setting of surgery, hospitalization, trauma, OCPs, pregnancy, or immobilization. Unprovoked VTE raises suspicion for occult cancer (up to 10% incidence within a year) and inherited thrombophilia (Factor V Leiden, prothrombin gene mutation, protein C/S or antithrombin deficiency, antiphospholipid syndrome).
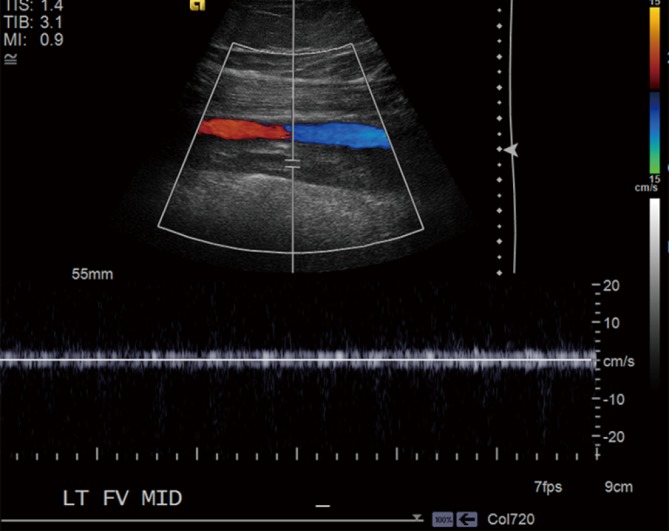
Diagnosis: D-dimer (high sensitivity, low specificity — best as a rule-out), compression duplex for DVT, CT pulmonary angiography for PE. V/Q scan is an alternative in pregnancy or contrast allergy. Treatment per ASH VTE guidelines: DOAC (apixaban, rivaroxaban, edoxaban, dabigatran) for most; LMWH preferred in cancer-associated thrombosis (or apixaban/rivaroxaban with caveats for luminal GI cancers); warfarin for mechanical valves and antiphospholipid syndrome. Duration: 3 months for provoked, indefinite for unprovoked or cancer-associated.
Heparin-induced thrombocytopenia (HIT) is an IgG antibody reaction to heparin-PF4 complexes causing paradoxical thrombosis (arterial or venous) with a 50% platelet drop typically 5–10 days after heparin exposure. Use the 4T score to estimate pretest probability; confirm with PF4 ELISA and functional serotonin release assay. Stop all heparin (including flushes) and switch to argatroban, bivalirudin, or fondaparinux; do not give warfarin alone until platelets recover and thrombosis is treated.
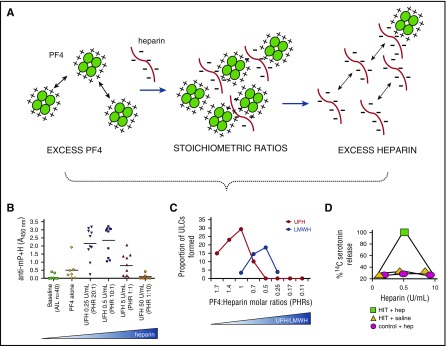
Warfarin: vitamin K + 4-factor prothrombin complex concentrate (Kcentra) for major bleeding. Dabigatran: idarucizumab. Apixaban/rivaroxaban: andexanet alfa or 4F-PCC. Heparin: protamine sulfate. LMWH: protamine (partial reversal).
Always document whether VTE is provoked or unprovoked, and whether the patient has active cancer — these determine duration and drug choice. “Cancer-associated thrombosis” is its own clinical category.
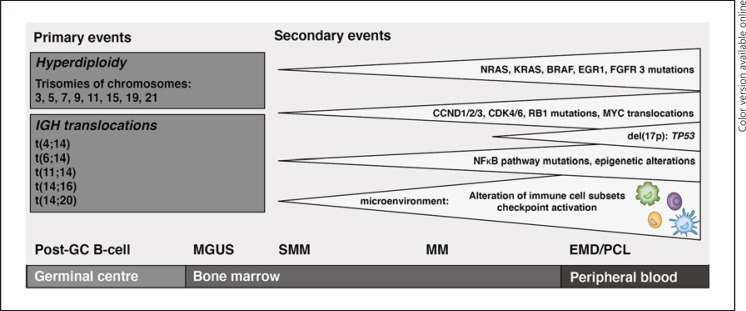
09 Plasma Cell Disorders Heme Malignancy
MGUS (monoclonal gammopathy of undetermined significance) is the asymptomatic precursor — M-protein < 3 g/dL, < 10% plasma cells in marrow, no CRAB features. Progresses to myeloma at ~1%/year. Smoldering multiple myeloma sits between MGUS and active disease.
Multiple myeloma is a clonal plasma cell malignancy defined by the CRAB criteria: Calcium elevation, Renal insufficiency, Anemia, Bone lesions (lytic). Additional myeloma-defining events: ≥ 60% clonal plasma cells, involved/uninvolved free light chain ratio ≥ 100, or > 1 focal MRI lesion. Workup: SPEP, UPEP, serum free light chains, beta-2 microglobulin, albumin, skeletal survey or whole-body low-dose CT or PET-CT, bone marrow biopsy with cytogenetics (FISH for del(17p), t(4;14), t(14;16), gain(1q)).
Staging uses ISS and R-ISS:
| R-ISS Stage | Criteria | Median OS |
|---|---|---|
| I | ISS I (β2M < 3.5, albumin ≥ 3.5) + normal LDH + standard-risk FISH | Not reached |
| II | Not stage I or III | ~83 months |
| III | ISS III (β2M ≥ 5.5) + high LDH or high-risk FISH (del17p, t(4;14), t(14;16)) | ~43 months |
Management per NCCN myeloma guidelines: induction with quadruplet (daratumumab + bortezomib + lenalidomide + dexamethasone, Dara-VRd), autologous stem cell transplant in transplant-eligible patients, lenalidomide maintenance, and multiple salvage options including CAR-T (ide-cel, cilta-cel), bispecifics (teclistamab), and carfilzomib/pomalidomide/selinexor combinations.
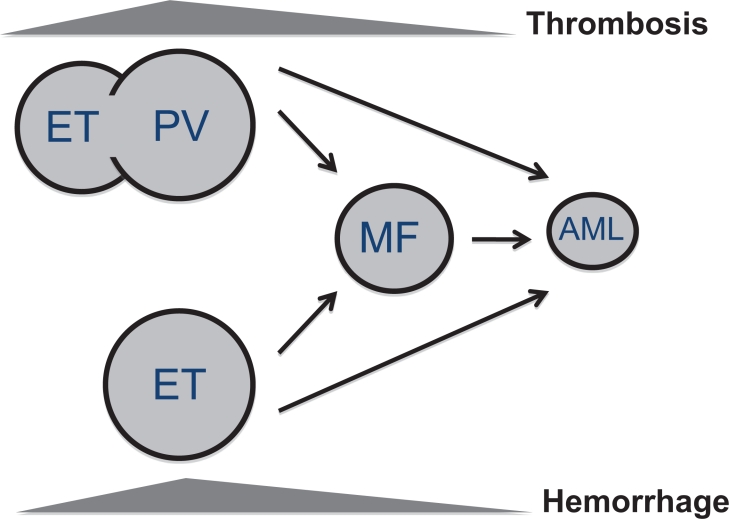
10 MDS & Myeloproliferative Neoplasms Heme Malignancy
Myelodysplastic syndromes (MDS) are clonal stem cell disorders with ineffective hematopoiesis — cytopenias with a hypercellular, dysplastic marrow. Risk is stratified by IPSS-R using cytogenetics, blast %, hemoglobin, platelets, and ANC (very low, low, intermediate, high, very high). Low-risk: supportive care, ESAs, luspatercept, lenalidomide (del5q). High-risk: hypomethylating agents (azacitidine, decitabine), venetoclax combinations, allogeneic stem cell transplant.
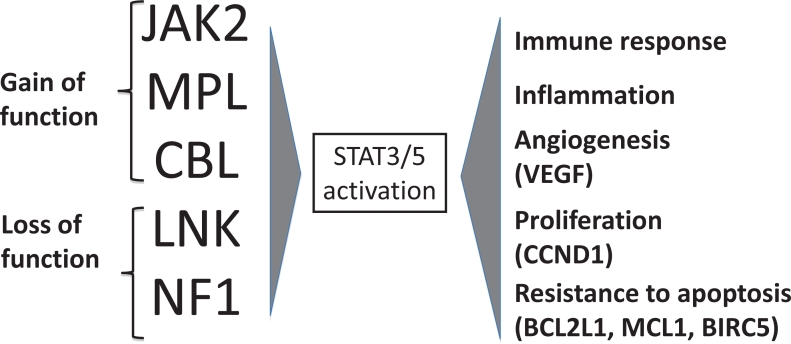
Myeloproliferative neoplasms (MPNs):
| MPN | Driver | Key Features | Treatment |
|---|---|---|---|
| Polycythemia vera (PV) | JAK2 V617F (> 95%) | Elevated Hgb/Hct, splenomegaly, pruritus (aquagenic), thrombosis risk | Phlebotomy (Hct < 45), low-dose ASA, hydroxyurea, ropeginterferon, ruxolitinib |
| Essential thrombocythemia (ET) | JAK2, CALR, MPL | Platelets > 450, thrombosis/bleeding risk | ASA, hydroxyurea or anagrelide if high-risk |
| Primary myelofibrosis (MF) | JAK2, CALR, MPL | Marrow fibrosis, massive splenomegaly, cytopenias, B symptoms | JAK inhibitors (ruxolitinib, fedratinib, pacritinib, momelotinib), allo-SCT |
| Chronic myeloid leukemia (CML) | t(9;22) BCR-ABL1 (Philadelphia) | Leukocytosis with left shift, splenomegaly, basophilia | TKIs: imatinib, dasatinib, nilotinib, bosutinib, ponatinib, asciminib |
CML response is monitored by BCR-ABL1 quantitative PCR, reported on the international scale (IS). Major molecular response = BCR-ABL1 ≤ 0.1% IS.
11 Acute Leukemias & CLL Heme Malignancy
Tobacco: pack-year history and quit date. Alcohol: drinks/week. Family cancer history: first- and second-degree relatives with age at diagnosis — triggers hereditary cancer panels (BRCA1/2, Lynch, Li-Fraumeni, Cowden). Prior cancers and treatments: prior RT fields, prior chemo (anthracycline cumulative dose matters for cardiotoxicity), prior transplant, HPV vaccination. Infections: HIV, HBV, HCV, HPV, H. pylori, EBV, HTLV-1. Occupational/environmental: asbestos, radon, benzene, aniline dyes, ionizing radiation.
AML (acute myeloid leukemia) presents with fatigue, bleeding, infection, and pancytopenia with circulating blasts. Diagnosis requires ≥ 20% blasts in marrow or blood. Classified by WHO/ICC by genetics: core-binding factor (t(8;21), inv(16)), NPM1-mutated, FLT3-ITD, APL (t(15;17) PML-RARA — a medical emergency treated with ATRA + arsenic trioxide). Treatment: 7+3 induction (cytarabine + anthracycline) in fit patients, azacitidine + venetoclax in unfit or older, targeted agents (midostaurin/gilteritinib for FLT3, ivosidenib/enasidenib for IDH1/2, gemtuzumab ozogamicin for CD33+), allogeneic stem cell transplant for intermediate/adverse risk.
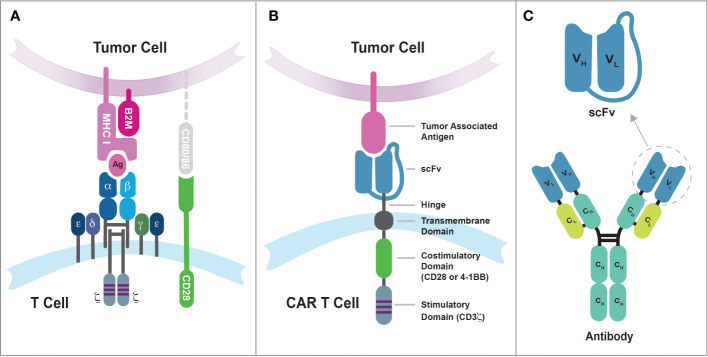
ALL (acute lymphoblastic leukemia) is most common in children but deadly in adults. B-cell or T-cell lineage. Philadelphia chromosome (Ph+) ALL is treated with TKI-containing regimens. CNS prophylaxis is mandatory (intrathecal methotrexate, cytarabine, hydrocortisone). Blinatumomab (CD19 BiTE) and inotuzumab ozogamicin (CD22 ADC) are key salvage therapies; CAR-T (tisagenlecleucel, brexucabtagene) is approved for relapsed/refractory disease.
CLL (chronic lymphocytic leukemia) is the most common adult leukemia. Asymptomatic lymphocytosis of mature B cells (CD5+, CD19+, CD23+). Staging:
| Rai Stage | Criteria | Risk |
|---|---|---|
| 0 | Lymphocytosis only | Low |
| I | Lymphocytosis + lymphadenopathy | Intermediate |
| II | Lymphocytosis + hepatosplenomegaly | Intermediate |
| III | Lymphocytosis + anemia (Hgb < 11) | High |
| IV | Lymphocytosis + thrombocytopenia (< 100) | High |
| Binet Stage | Criteria |
|---|---|
| A | < 3 lymphoid areas, no anemia/thrombocytopenia |
| B | ≥ 3 lymphoid areas, no anemia/thrombocytopenia |
| C | Anemia or thrombocytopenia |
Treatment is indication-driven: no therapy for asymptomatic early disease. Indications include symptomatic adenopathy/organomegaly, cytopenias, B symptoms, autoimmune cytopenias. First-line per NCCN CLL guidelines: BTK inhibitors (acalabrutinib, zanubrutinib, ibrutinib) or venetoclax + obinutuzumab.
12 Lymphomas Heme Malignancy
Hodgkin lymphoma (HL) features Reed-Sternberg cells (CD15+, CD30+). Bimodal age distribution. Classic HL subtypes: nodular sclerosing (most common), mixed cellularity, lymphocyte-rich, lymphocyte-depleted; plus nodular lymphocyte predominant HL. Presents with painless cervical/supraclavicular/mediastinal lymphadenopathy, B symptoms, alcohol-induced pain. Treatment: ABVD or BV-AVD (brentuximab vedotin + AVD) for advanced stage; PET-adapted therapy; checkpoint inhibitors (nivolumab, pembrolizumab) for relapsed.
Non-Hodgkin lymphomas (NHL) are a heterogeneous group. Aggressive: DLBCL (most common NHL; R-CHOP or Pola-R-CHP first line, CAR-T for relapsed), mantle cell (t(11;14), cyclin D1+; bendamustine-rituximab, BTK inhibitors), Burkitt (MYC translocation, extremely rapid — TLS risk). Indolent: follicular lymphoma (t(14;18), BCL2; watch and wait or bendamustine-rituximab or lenalidomide-rituximab), marginal zone (MALT, splenic, nodal; often H. pylori-driven gastric MALT responds to eradication), Waldenström macroglobulinemia (IgM, MYD88, BTK inhibitors).
Staging uses Lugano/Ann Arbor:
| Stage | Criteria |
|---|---|
| I | Single nodal region (or single extranodal site IE) |
| II | Two or more nodal regions, same side of diaphragm |
| III | Nodal regions on both sides of the diaphragm |
| IV | Disseminated extranodal involvement (bone marrow, liver, lung) |
Modifiers: A (no B symptoms), B (fevers, night sweats, > 10% weight loss), E (extranodal extension), X (bulky disease > 10 cm). Response is reported by Lugano response criteria using PET: complete metabolic response (Deauville 1–3), partial, no response, progressive disease. See Lugano classification.
13 Breast Cancer Solid Tumor
Breast cancer is the most common non-skin cancer in women. Presents as a screen-detected abnormality, a palpable mass, or locally advanced disease with skin/nipple changes. Workup: diagnostic mammogram + ultrasound, core needle biopsy, staging imaging for high-risk or node-positive disease. Biomarkers drive every treatment decision: ER, PR, HER2, Ki-67.
Subtypes: hormone receptor positive (ER+ and/or PR+, HER2−), HER2 positive (HER2 IHC 3+ or ISH amplified), triple-negative (ER−, PR−, HER2−). Staging is AJCC TNM with biomarker incorporation. USPSTF breast cancer screening recommends biennial mammography starting at age 40.
Treatment: surgery (lumpectomy with whole-breast RT vs mastectomy), sentinel lymph node biopsy or axillary dissection, adjuvant chemo (AC-T, TC, TCHP for HER2+), endocrine therapy (tamoxifen in premenopausal, aromatase inhibitors — anastrozole, letrozole, exemestane — in postmenopausal) for 5–10 years in HR+ disease, CDK4/6 inhibitors (palbociclib, ribociclib, abemaciclib) in metastatic and high-risk early HR+, trastuzumab/pertuzumab in HER2+, trastuzumab deruxtecan (T-DXd) for HER2+ and HER2-low metastatic, PARP inhibitors (olaparib, talazoparib) for BRCA-mutated, sacituzumab govitecan for TNBC. See NCCN breast cancer guidelines.
Every new breast cancer note captures: laterality, tumor size (cm), histology, grade, ER/PR percentages, HER2 status (IHC 0/1+/2+/3+; ISH ratio if 2+), Ki-67, cN status (clinical node stage), imaging staging workup, clinical TNM, family history, and BRCA/genetic testing plan. Oncotype DX recurrence score (0–100) drives adjuvant chemo decisions in HR+/HER2−/node-negative disease.
14 Lung Cancer (NSCLC & SCLC) Solid Tumor
Leading cause of cancer death in the US. Non-small cell lung cancer (NSCLC) accounts for ~85% and includes adenocarcinoma (most common), squamous cell, and large cell. Small cell lung cancer (SCLC) is a neuroendocrine malignancy almost always smoking-related with rapid growth and early metastasis. Screening: USPSTF recommends annual low-dose CT in adults 50–80 with ≥ 20 pack-year history who currently smoke or quit within 15 years.
NSCLC workup: biopsy with molecular profiling (EGFR, ALK, ROS1, BRAF, MET exon 14, RET, NTRK, KRAS G12C, HER2) and PD-L1 TPS. Staging by AJCC 8th edition TNM, brain MRI, PET-CT, mediastinal evaluation (EBUS). Treatment: early stage — surgical resection ± adjuvant chemo ± osimertinib (EGFR+) or atezolizumab; locally advanced — concurrent chemoradiation + durvalumab consolidation; metastatic — targeted therapy for driver mutations (osimertinib for EGFR, alectinib/lorlatinib for ALK), immunotherapy (pembrolizumab, nivolumab/ipilimumab, atezolizumab) ± chemotherapy. See NCCN NSCLC guidelines.
SCLC: SIADH, Cushing (ectopic ACTH), Lambert-Eaton myasthenic syndrome, paraneoplastic cerebellar degeneration. Squamous NSCLC: hypercalcemia from PTHrP, hypertrophic pulmonary osteoarthropathy. RCC: polycythemia (EPO), hypercalcemia, Stauffer syndrome. HCC: erythrocytosis, hypoglycemia. Thymoma: myasthenia gravis, pure red cell aplasia.
SCLC is staged as limited (confined to one radiation port) or extensive. Treatment: platinum/etoposide + atezolizumab or durvalumab, thoracic RT, prophylactic cranial irradiation in responders. Lurbinectedin, topotecan, and tarlatamab for relapse.
15 GI Malignancies Solid Tumor
Colorectal cancer (CRC): second leading cause of cancer death in the US. USPSTF recommends screening from age 45–75. Workup includes colonoscopy with biopsy, CEA, CT chest/abdomen/pelvis, MMR/MSI testing (all new CRC), RAS/BRAF/HER2 testing for metastatic disease. Stage-based treatment: surgery for I–III + adjuvant FOLFOX/CAPOX; neoadjuvant total neoadjuvant therapy for locally advanced rectal; metastatic CRC uses FOLFOX/FOLFIRI/FOLFIRINOX + bevacizumab or anti-EGFR (cetuximab, panitumumab — only in RAS wild-type), pembrolizumab for MSI-H, BRAF V600E combinations (encorafenib + cetuximab), HER2-targeted (trastuzumab + tucatinib).
Gastric cancer: associated with H. pylori, chronic gastritis, smoking, EBV. HER2 and PD-L1 testing mandatory. First-line metastatic: FOLFOX/CAPOX + nivolumab; add trastuzumab if HER2+; zolbetuximab for CLDN18.2+.
Pancreatic cancer: poor prognosis, most present with locally advanced or metastatic disease. CA 19-9 tumor marker. Treatment: surgical resection (Whipple) only in resectable disease, followed by modified FOLFIRINOX or gemcitabine + capecitabine; metastatic uses FOLFIRINOX, gemcitabine/nab-paclitaxel, olaparib (BRCA), NALIRIFOX.
Hepatocellular carcinoma (HCC): arises in cirrhosis (HCV, HBV, alcohol, MASH). Diagnosed radiographically by LI-RADS criteria without biopsy in cirrhosis. Treatment: resection, transplant (Milan criteria), ablation, TACE, Y-90, systemic therapy (atezolizumab + bevacizumab first-line, durvalumab + tremelimumab, lenvatinib, sorafenib, regorafenib, cabozantinib, ramucirumab).
16 GU Malignancies Solid Tumor
Prostate cancer: Screening is a shared decision in men 55–69 (USPSTF). Diagnosis: elevated PSA → MRI → targeted biopsy. Grading uses Gleason score (3+3=6, 3+4=7, 4+3=7, 4+4=8, up to 5+5=10) translated into Grade Groups 1–5. Management: active surveillance for low-risk; radical prostatectomy or radiation for localized; androgen deprivation therapy (leuprolide, degarelix, relugolix) ± androgen receptor pathway inhibitors (abiraterone, enzalutamide, apalutamide, darolutamide) for advanced; docetaxel, cabazitaxel, radium-223, lutetium-177 PSMA for metastatic castration-resistant.
Renal cell carcinoma (RCC): clear cell (most common), papillary, chromophobe. Presents with hematuria, flank pain, palpable mass (classic triad, rare), or incidentally on imaging. Treatment: nephrectomy (partial or radical), adjuvant pembrolizumab for high-risk; first-line metastatic: IO/IO (nivolumab + ipilimumab) or IO/TKI (pembrolizumab + axitinib, pembrolizumab + lenvatinib, nivolumab + cabozantinib).
Bladder cancer: non-muscle invasive (NMIBC) vs muscle-invasive (MIBC). NMIBC: TURBT + intravesical BCG or mitomycin. MIBC: neoadjuvant cisplatin-based chemo + radical cystectomy, or bladder-preservation with chemoradiation. Metastatic: pembrolizumab, enfortumab vedotin + pembrolizumab, erdafitinib (FGFR), sacituzumab govitecan.
17 Gynecologic, Head & Neck, Skin, CNS & Sarcoma Solid Tumor
Ovarian cancer: presents late with vague abdominal symptoms, ascites, pelvic mass. CA-125 tumor marker. Debulking surgery + carboplatin/paclitaxel ± bevacizumab. PARP inhibitors (olaparib, niraparib, rucaparib) for BRCA-mutant and HRD+ maintenance. Mirvetuximab soravtansine for FRα+ platinum-resistant.
Cervical cancer: HPV-driven. Screening with Pap + HPV testing. Locally advanced: concurrent cisplatin + RT + brachytherapy. Metastatic: cisplatin/paclitaxel + bevacizumab + pembrolizumab; tisotumab vedotin.
Endometrial cancer: postmenopausal bleeding is the cardinal symptom. Molecular classification: POLE-ultramutated, MSI-H, copy-number low, copy-number high (p53-abnormal). TAH-BSO + staging; adjuvant RT/chemo risk-adapted; advanced: carboplatin/paclitaxel + dostarlimab or pembrolizumab, lenvatinib + pembrolizumab.
Head and neck squamous cell carcinoma (HNSCC): tobacco, alcohol, HPV (especially oropharyngeal). Treatment: surgery, concurrent chemoradiation with cisplatin, cetuximab; pembrolizumab or nivolumab for recurrent/metastatic.
Melanoma: staged by Breslow depth, ulceration, mitotic rate, LN involvement. BRAF V600 testing. Adjuvant and metastatic: immunotherapy (pembrolizumab, nivolumab, nivolumab + ipilimumab, nivolumab + relatlimab), BRAF/MEK inhibitors (dabrafenib + trametinib, encorafenib + binimetinib), T-VEC, tebentafusp (uveal).
Glioblastoma (GBM): WHO grade 4 astrocytoma, IDH wild-type. Treatment: maximal safe resection + Stupp protocol (temozolomide + radiation, then adjuvant TMZ), tumor-treating fields, bevacizumab at recurrence. MGMT methylation predicts TMZ response.
Sarcomas: heterogeneous mesenchymal tumors. Soft-tissue sarcomas managed with wide resection ± RT ± anthracycline-based chemo. GIST: imatinib (KIT/PDGFRA). Osteosarcoma and Ewing sarcoma follow pediatric/AYA protocols with MAP and VDC/IE chemotherapy.
18 Oncologic Emergencies Emergency
ANC < 500 (or expected to fall < 500) with single temp ≥ 38.3°C or sustained ≥ 38.0°C. Empiric broad-spectrum antibiotics (cefepime, piperacillin-tazobactam, or meropenem) within 1 hour. Add vancomycin for catheter infection, skin/soft tissue, pneumonia, or hemodynamic instability. G-CSF support.
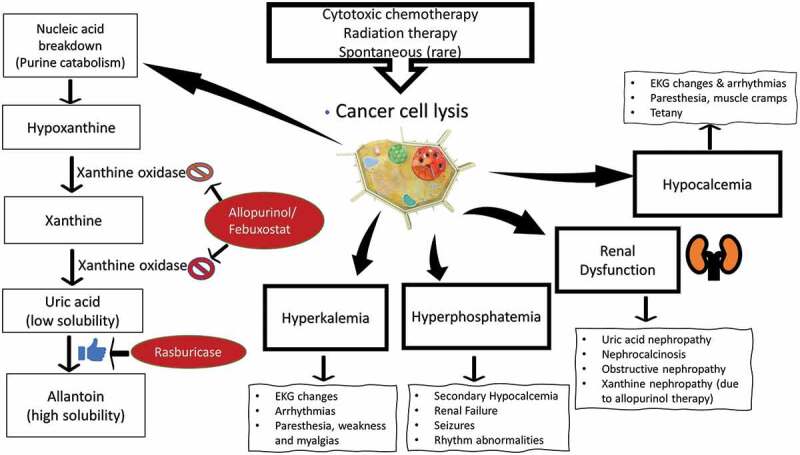
Massive cell lysis → hyperuricemia, hyperkalemia, hyperphosphatemia, hypocalcemia, AKI. Classic in Burkitt, ALL, bulky DLBCL, AML. Prevention: aggressive IV hydration, allopurinol (prophylaxis) or rasburicase (treatment/high-risk). Cairo-Bishop criteria define laboratory and clinical TLS.
PTHrP (squamous, breast, renal), lytic bone lesions (myeloma, breast), 1,25-OH vitamin D (lymphoma). Treatment: aggressive IV fluids, IV bisphosphonates (zoledronic acid, pamidronate), denosumab, calcitonin for rapid temporization.
Facial/arm swelling, distended neck veins, headache, dyspnea. Causes: lung cancer, lymphoma, thymoma, thrombosis. Diagnosis: CT chest. Treatment: address malignancy (chemo, RT), endovascular stent, steroids if lymphoma suspected.
Back pain (first symptom), weakness, sensory level, bladder/bowel dysfunction. MRI total spine. Immediate high-dose dexamethasone (often 10 mg IV load, then 4 mg q6h), neurosurgery/radiation oncology consult. Time-sensitive — motor function rarely recovers if lost > 24 hours.
Post-CAR-T or bispecific antibody complications. CRS: fever, hypotension, hypoxia, capillary leak; graded 1–4 by ASTCT criteria; treatment with tocilizumab ± steroids. ICANS: confusion, aphasia, seizures, cerebral edema; ICE score for grading; treatment with steroids (dexamethasone or high-dose methylprednisolone for grade ≥ 3).
WBC typically > 100,000 in AML or ALL with symptoms of respiratory distress, altered mental status, visual changes from sludging in the microvasculature. Treatment: immediate cytoreduction with hydroxyurea, leukapheresis, and definitive chemotherapy; avoid RBC transfusion if possible (worsens viscosity). Concurrent TLS prophylaxis is mandatory.
19 Procedures in Heme/Onc
| Procedure | What It Is | Key Documentation |
|---|---|---|
| Bone marrow biopsy / aspirate | Posterior iliac crest aspirate + core biopsy with Jamshidi needle under local ± conscious sedation | Site, consent, specimen sent (cytogenetics, flow, FISH, NGS), complications |
| Lumbar puncture (LP) | Intrathecal chemo delivery, CSF evaluation for leptomeningeal disease | Opening pressure, fluid appearance, volumes, intrathecal agent/dose |
| Port / Mediport placement | Implanted central venous access under IR for chemo delivery | Laterality, access vein, fluoro confirmation, de-accessed/accessed |
| PICC line | Peripherally inserted central catheter for prolonged IV therapy | Arm, vein, tip position at cavoatrial junction |
| Apheresis / plasma exchange | Blood separation to remove plasma (TTP), cells (hyperleukocytosis), collect stem cells or T cells | Volume processed, access type, product collected, adverse reactions |
| Transfusion (PRBC, platelets, FFP, cryo) | Blood product administration | Product, volume, indication, pre/post counts, premedication, reactions |
| IVIG infusion | Immunoglobulin replacement or immunomodulation (ITP, CLL hypogammaglobulinemia) | Dose (g/kg), rate, premedication, reactions |
| Autologous stem cell transplant | High-dose chemo + infusion of patient’s own stem cells (myeloma, relapsed lymphoma) | Conditioning regimen, CD34 dose, engraftment day |
| Allogeneic stem cell transplant | Donor stem cell infusion (sibling, MUD, haploidentical); risk of GVHD | Donor match, conditioning (MAC vs RIC), GVHD prophylaxis, chimerism |
| CAR-T cell therapy | Lymphodepleting chemo + engineered autologous T cells | Product (axi-cel, tisa-cel, liso-cel, brexu-cel, ide-cel, cilta-cel), CRS/ICANS grade |
| Paracentesis / thoracentesis | Drainage of malignant ascites / pleural effusion | Volume removed, appearance, cytology sent |
Bone Marrow Biopsy in Detail
The bone marrow biopsy is the single most important diagnostic procedure in hematology. It is performed at the posterior superior iliac spine with the patient in lateral decubitus or prone. After skin prep, local anesthetic (1% lidocaine) is infiltrated to the periosteum. A Jamshidi needle is advanced into the cortex and a liquid aspirate (“marrow tap”) is drawn for smears, flow cytometry, cytogenetics (karyotype, FISH), and molecular testing (NGS, PCR). A core biopsy — a solid cylinder of marrow — is then obtained for architecture, cellularity, and reticulin staining. Dry taps (packed marrow, fibrosis, or “hypocellular” disease) may require an imprint from the core. Document the site, laterality, consent, timeout, anesthetic volume, specimens sent, estimated blood loss, and post-procedure pressure dressing.
Transfusion Thresholds
| Product | Typical Trigger | Dose |
|---|---|---|
| Packed red blood cells | Hgb < 7 (general), < 8 cardiac disease, symptomatic | 1 unit = 1 g/dL rise |
| Platelets | < 10 prophylactic, < 20 with fever/sepsis, < 50 procedure, < 100 CNS/ophthalmic surgery | 1 apheresis unit raises by ~30k |
| Fresh frozen plasma | Active bleeding with INR > 1.7 or multiple factor deficiency | 10–15 mL/kg |
| Cryoprecipitate | Fibrinogen < 100–150 | 1 unit per 10 kg raises by ~50 mg/dL |
Bone marrow biopsy, lumbar puncture, and central venous access are the procedures scribes will document most. Always capture informed consent, timeout, site marking, anesthetic, needle type, number of passes, specimens sent, estimated blood loss, and post-procedure plan (imaging confirmation, hold pressure, patient discharged ambulating).
20 Chemotherapy & Targeted / Immuno / Endocrine Agents
Chemotherapy is organized by mechanism class. Each class has signature toxicities you must know because they drive the review of systems.
| Class | Examples | Mechanism | Key Toxicities |
|---|---|---|---|
| Alkylators | Cyclophosphamide, ifosfamide, bendamustine, melphalan, temozolomide | Cross-link DNA | Hemorrhagic cystitis (cyclo/ifos — mesna), secondary leukemia, infertility |
| Antimetabolites | Methotrexate, 5-FU, capecitabine, gemcitabine, cytarabine, pemetrexed | Inhibit DNA synthesis | Mucositis, diarrhea, myelosuppression, hand-foot syndrome (cape/5-FU) |
| Anthracyclines | Doxorubicin, daunorubicin, idarubicin, epirubicin | Topoisomerase II, free radicals | Cardiotoxicity (cumulative dose), red urine, vesicant |
| Taxanes | Paclitaxel, docetaxel, nab-paclitaxel | Stabilize microtubules | Peripheral neuropathy, myalgias, hypersensitivity |
| Platinums | Cisplatin, carboplatin, oxaliplatin | Crosslink DNA | Nephrotoxicity (cis), ototoxicity, neuropathy, N/V, cold dysesthesias (oxali) |
| Vinca alkaloids | Vincristine, vinblastine, vinorelbine | Inhibit microtubule assembly | Peripheral neuropathy (vincristine), constipation, NEVER intrathecal |
| Topoisomerase inhibitors | Etoposide, irinotecan, topotecan | Topo II (etop), Topo I (irino/topo) | Irinotecan: early & late diarrhea; secondary AML (etoposide) |
| TKIs | Imatinib, erlotinib, osimertinib, sunitinib, sorafenib, ibrutinib, etc. | Oral small-molecule kinase inhibitors | Rash, diarrhea, hypertension (VEGF), QT, hepatotoxicity |
| CDK4/6 inhibitors | Palbociclib, ribociclib, abemaciclib | Cell cycle arrest G1/S | Neutropenia, diarrhea (abema), QT (ribo) |
| PARP inhibitors | Olaparib, niraparib, rucaparib, talazoparib | Synthetic lethality in HRD/BRCA | Myelosuppression, MDS/AML, fatigue |
| Monoclonal antibodies | Rituximab, trastuzumab, cetuximab, bevacizumab, daratumumab | Target-specific | Infusion reactions; bev: HTN, proteinuria, bleeding, GI perforation |
| Antibody-drug conjugates | T-DM1, T-DXd, enfortumab vedotin, sacituzumab govitecan, brentuximab | Targeted delivery of cytotoxic payload | ILD (T-DXd), neuropathy, cytopenias |
| Checkpoint inhibitors | Pembrolizumab, nivolumab, atezolizumab, durvalumab, ipilimumab, cemiplimab | Block PD-1/PD-L1/CTLA-4 | irAEs: colitis, pneumonitis, hepatitis, endocrinopathies (thyroid, adrenal, hypophysitis), dermatitis |
| CAR-T cells | Axi-cel, tisa-cel, liso-cel, brexu-cel, ide-cel, cilta-cel | Engineered autologous T cells (CD19 or BCMA) | CRS, ICANS, cytopenias, B-cell aplasia |
| Endocrine (breast) | Tamoxifen, anastrozole, letrozole, exemestane, fulvestrant | SERM, AI, SERD | Hot flashes, VTE (tamoxifen), bone loss (AI), arthralgia |
| Endocrine (prostate) | Leuprolide, degarelix, relugolix, abiraterone, enzalutamide | GnRH agonist/antagonist, ARSI | Hot flashes, osteoporosis, metabolic syndrome, fatigue |
| Supportive | Filgrastim / pegfilgrastim (G-CSF), epoetin / darbepoetin (ESA), ondansetron, aprepitant, olanzapine, dexamethasone | Growth factors, antiemetics | Bone pain (G-CSF), thrombosis (ESA), QT, constipation |
Immune-related adverse events (irAEs) can hit any organ. When an immunotherapy patient reports new diarrhea, cough, rash, or fatigue, document it explicitly and flag it. “Grade 2 immune colitis” and “Grade 3 pneumonitis” are treatment-changing diagnoses.
21 Imaging, Labs & Tumor Markers
Imaging: CT chest/abdomen/pelvis with contrast is the workhorse staging and restaging study. PET-CT (FDG) is central to lymphoma response (Deauville score 1–5) and many solid tumors. MRI brain for CNS staging, MRI breast for occult disease in dense tissue or BRCA, MRI prostate (PI-RADS). Bone scan for prostate, breast; replaced by PSMA PET for prostate. Echocardiogram before and during anthracycline or HER2-targeted therapy to monitor LVEF.
Labs: CBC with differential, CMP, LDH (tumor burden marker), uric acid, magnesium, phosphate, coagulation panel. Absolute neutrophil count (ANC) = WBC × (% neutrophils + % bands). Severe neutropenia < 500; profound < 100.
Critical lab patterns: Tumor lysis shows high uric acid, high potassium, high phosphate, low calcium, high LDH, rising creatinine. Hemolysis shows low haptoglobin, high LDH, high indirect bilirubin, high retic count. DIC shows low fibrinogen, high D-dimer, low platelets, prolonged PT/PTT. Myeloma shows high total protein with low albumin (high gap), high calcium, high creatinine, anemia. Always compare today’s values to the trend — oncologists think in deltas.
Tumor markers:
| Marker | Primary Use |
|---|---|
| PSA | Prostate cancer screening, surveillance |
| CEA | Colorectal surveillance (not screening) |
| CA 19-9 | Pancreatic, biliary |
| CA 125 | Ovarian |
| CA 15-3 / CA 27-29 | Breast (metastatic monitoring) |
| AFP | HCC, germ cell |
| β-hCG | Germ cell, gestational trophoblastic |
| LDH | Germ cell, lymphoma, melanoma — tumor burden |
| β2 microglobulin | Myeloma staging, CLL prognosis |
| Thyroglobulin | Post-thyroidectomy thyroid cancer |
| Calcitonin | Medullary thyroid cancer |
| Chromogranin A | Neuroendocrine tumors |
22 Classification & Response Systems
| Grade | Description |
|---|---|
| 0 | Fully active, no restrictions |
| 1 | Restricted in strenuous activity; ambulatory, light work |
| 2 | Ambulatory, self-care, unable to work, up > 50% waking hours |
| 3 | Limited self-care, confined to bed/chair > 50% waking hours |
| 4 | Completely disabled, no self-care, totally confined |
| 5 | Dead |
100 = normal, no complaints; 90 = normal activity, minor symptoms; 80 = normal activity with effort; 70 = cares for self, cannot work; 60 = occasional assistance; 50 = considerable assistance, frequent care; 40 = disabled, special care; 30 = severely disabled, hospitalized; 20 = very sick, active supportive care; 10 = moribund; 0 = dead.
T = primary tumor size/extent (T1–T4), N = regional node involvement (N0–N3), M = distant metastasis (M0 or M1). Combined into stage groups I–IV. Always cite the edition (AJCC 8th). Many tumors have additional descriptors (Tis carcinoma in situ, Tx unable to assess).
| Response | Criteria |
|---|---|
| Complete response (CR) | Disappearance of all target lesions; any nodes < 10 mm |
| Partial response (PR) | ≥ 30% decrease in sum of target lesion diameters |
| Stable disease (SD) | Neither PR nor PD |
| Progressive disease (PD) | ≥ 20% increase in sum (and ≥ 5 mm absolute) or new lesions |
1 = no uptake above background; 2 = uptake ≤ mediastinum; 3 = uptake > mediastinum, ≤ liver; 4 = moderately > liver; 5 = markedly > liver or new lesions. Scores 1–3 considered complete metabolic response in most protocols.
Grade 1 = mild; 2 = moderate, limiting instrumental ADLs; 3 = severe, hospitalization indicated, limiting self-care ADLs; 4 = life-threatening; 5 = death. Every chemo toxicity documentation should carry a grade.
Risk is calculated from cytogenetics, marrow blast percent, hemoglobin, platelets, and ANC. Very low (≤ 1.5 points, median OS ~8.8 years), Low (> 1.5–3, ~5.3 years), Intermediate (> 3–4.5, ~3 years), High (> 4.5–6, ~1.6 years), Very high (> 6, ~0.8 years). Higher risk drives consideration of hypomethylating agents and allogeneic transplant. See IPSS-R reference.
Grade 1: ANC 1,500–1,999 (mild); Grade 2: 1,000–1,499 (moderate); Grade 3: 500–999 (severe — consider G-CSF); Grade 4: < 500 (life-threatening, infection precautions). Febrile neutropenia is always graded 3 at minimum.
23 Physical Exam & Abbreviations
Dx: Stage IIIB HER2+ invasive ductal carcinoma, right breast
Dx date: 02/2026
Biomarkers: ER 90% / PR 70% / HER2 3+ / Ki-67 35%
Current regimen: TCHP, C4D1
Prior lines: none
ECOG: 1 KPS: 80
Last imaging: CT 03/2026 — partial response
Allergies: NKDA Port: R chest, accessed today
The Heme/Onc-Focused Exam
General appearance and ECOG. HEENT: pallor of conjunctivae, icterus, oral mucositis, thrush, gum hypertrophy (AML M4/M5). Lymph node survey: cervical, supraclavicular (Virchow/Troisier on the left for GI malignancy), axillary, epitrochlear, inguinal — document size (in cm), consistency (rubbery vs firm vs matted), tenderness, mobility. Cardiovascular: rate, rhythm, murmurs (flow murmurs in anemia), JVD. Respiratory: dullness, crackles, decreased breath sounds (effusion). Abdomen: hepatomegaly, splenomegaly (estimate cm below costal margin), masses, ascites. Skin: petechiae, ecchymoses, purpura, chloromas, leukemia cutis, rashes (irAE), port site, radiation changes. Neuro: baseline mental status, cranial nerves, peripheral neuropathy (vibration, monofilament), motor/sensory in patients on neurotoxic chemo.
Abbreviations Master List
24 Sample HPI Templates & References
These templates show the cadence of a real heme/onc clinic note. Every HPI in this specialty references diagnosis date, histology, stage, line of therapy, cycle/day, and toxicities.
“Ms. [Name] is a 54-year-old postmenopausal female referred for newly diagnosed right breast cancer. She was found to have a palpable 2.1 cm mass in the right upper outer quadrant on self-exam. Diagnostic mammogram and ultrasound confirmed a BI-RADS 5 lesion; core biopsy on 03/14/2026 revealed invasive ductal carcinoma, grade 2, ER 95%, PR 80%, HER2 0 by IHC, Ki-67 15%. Right axillary ultrasound showed a 1.2 cm level I node with cortical thickening; FNA positive. Staging CT chest/abdomen/pelvis and bone scan negative. Clinical stage cT2N1M0 (IIB). She has no family history of breast or ovarian cancer; BRCA testing pending. PMH significant for hypertension and osteopenia. ECOG 0.”
“Mr. [Name] is a 68-year-old male referred for evaluation of microcytic anemia found on routine labs (Hgb 9.4, MCV 76, ferritin 8, TSAT 6%). He reports 3 months of fatigue and exertional dyspnea but denies melena, hematochezia, hematemesis, or overt bleeding. No NSAID use. Colonoscopy 8 years ago unremarkable. PMH: hypertension, GERD on pantoprazole. Physical exam reveals pale conjunctivae and a 2/6 systolic flow murmur. Workup today: iron panel, reticulocyte count, hemolysis labs, and GI referral for EGD/colonoscopy. Planning IV ferric carboxymaltose after imaging.”
“Mr. [Name] is a 62-year-old male with AML (NPM1-mutated, FLT3-ITD negative) diagnosed 11/2025, s/p 7+3 induction achieving CR on day 28 marrow, now C2D1 of HiDAC consolidation. He reports fatigue, mild nausea controlled with ondansetron, no fevers, no bleeding. ANC 1.8, platelets 95, Hgb 10.2. No infectious symptoms. Indwelling mediport without erythema or drainage. ECOG 1. Plans to proceed with cycle 2 today.”
“Ms. [Name] is a 46-year-old female with stage III DLBCL on R-CHOP, C3D10, who presents with temperature 38.6°C at home. She reports fatigue, mild rhinorrhea, no cough, dyspnea, diarrhea, dysuria, or rash. ANC 0.2, Hgb 9.1, platelets 88. Blood cultures × 2 drawn, UA, chest X-ray obtained. Empiric cefepime 2 g IV initiated within 45 minutes of presentation. Admitted for neutropenic fever management; ID consulted.”
“Mr. [Name] is a 28-year-old male presenting with newly diagnosed classical Hodgkin lymphoma, nodular sclerosing subtype, via excisional biopsy of a 4 cm left supraclavicular node (03/2026). He reports 4 months of drenching night sweats, 6 kg unintentional weight loss, and intermittent fevers to 38.5°C. PET-CT shows FDG-avid cervical, mediastinal (bulky, 11 cm), and para-aortic lymphadenopathy without extranodal sites. Bone marrow biopsy negative. Stage IIIB bulky. Echo LVEF 65%. PFT DLCO 92%. Planning BV-AVD × 6 cycles.”
“Mrs. [Name] is a 67-year-old female with metastatic pancreatic adenocarcinoma on FOLFIRINOX (C5D1 today) presenting with 2 days of left lower extremity swelling and calf pain. Venous duplex confirms occlusive thrombus in the left popliteal and femoral veins. No chest pain, dyspnea, or hemoptysis. Platelets 142, creatinine 0.9. No active bleeding. Initiated on apixaban 10 mg BID × 7 days then 5 mg BID, with plan for indefinite anticoagulation given active malignancy per ASH CAT guidance.”
References & Sources
Clinical Practice Guidelines
NCCN Non-Small Cell Lung Cancer Guidelines
NCCN Multiple Myeloma Guidelines
ASCO/ASH ESA in Cancer Guidelines
USPSTF Colorectal Cancer Screening 2021
USPSTF Breast Cancer Screening
USPSTF Lung Cancer Screening 2021
USPSTF Prostate Cancer Screening
Lugano Classification for Lymphoma (Cheson et al., JCO 2014)
Revised International Staging System (R-ISS) for Multiple Myeloma
IPSS-R for Myelodysplastic Syndromes
ESMO Clinical Practice Guidelines (master portal)
Diagram & Figure Sources
Figure 1: Bone Marrow Anatomy, OpenStax College. CC BY 3.0.
Figure 2: Hematopoiesis, OpenStax College. CC BY 3.0.
Figure 3: Lymphatic System (Female), Blausen Medical. CC BY 3.0.
Figure 4: T Lymphocyte, Blausen Medical. CC BY 3.0.
Figure 5: Sickled Red Cells, CDC/Janice Haney Carr. Public domain.
Figure 6: Multiple Myeloma Marrow Aspirate. CC BY-SA 3.0.
Heme/onc moves fast and forgives nothing. Your note is the legal record of dose decisions that determine whether a patient gets chemo today or is held for a week. Know the regimen names and cycle numbers cold. Document ANC, platelets, creatinine, and bilirubin in every note because those four numbers decide the day’s plan. Understand the difference between a complete response and stable disease; they sound similar but mean opposite things prognostically. Learn the vocabulary of immunotherapy toxicity, because an irAE caught early saves lives.
Most of all, remember who is on the other side of the note: a patient and family navigating the most frightening period of their lives. Accurate, compassionate documentation is not a clerical task. It is how the care team speaks to itself about a person. Welcome to heme/onc — the learning curve is real, but so is the impact.
Build a mental checklist for every chemo day note: ANC, platelets, creatinine, bilirubin, weight, performance status, toxicity review by system, and cycle/day. If one of those is missing from your note, go back and fill it in before the clinician signs.