Infectious Disease
Every syndrome, pathogen, antimicrobial, procedure, and documentation framework you need to survive the ID consult service and the inpatient ward.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Microbiology & Immunology Essentials
Infectious disease practice revolves around a simple question asked hundreds of times a day: which bug, which drug, which host, which site? As a scribe on the ID consult service you must learn to hear a clinician say "gram-positive cocci in clusters from two bottles" and immediately understand the working diagnosis is Staphylococcus aureus bacteremia, not just transcribe the phrase. The foundation for that pattern recognition is a basic command of microbiology taxonomy, host immunity, and the anatomic reservoirs in which pathogens live.
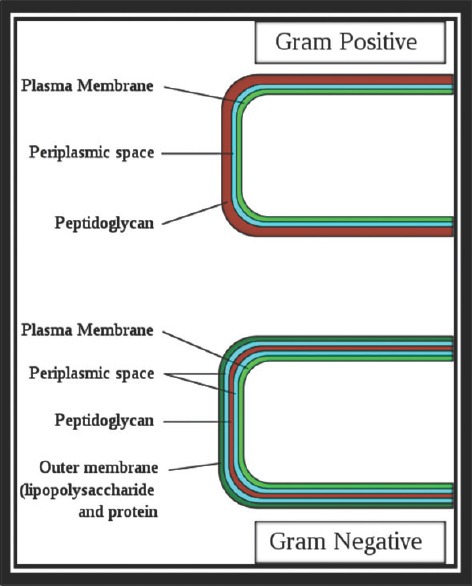
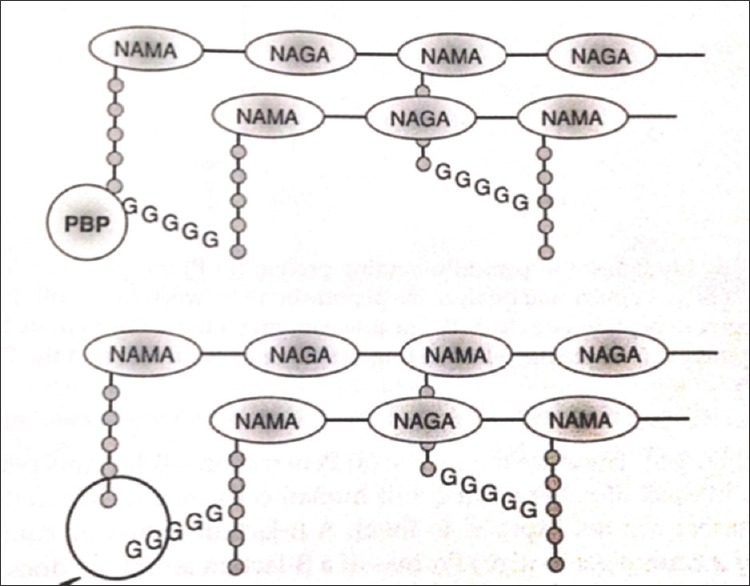
Bacteria — The Gram Stain Framework
The Gram stain divides bacteria into two large groups based on cell wall structure. Gram-positive organisms retain crystal violet and appear purple; gram-negative organisms do not retain it and counter-stain pink with safranin. Morphology adds a second axis — cocci (spheres), bacilli (rods), or coccobacilli. Arrangement matters too: clusters suggest staphylococci, chains suggest streptococci and enterococci, diplococci suggest pneumococcus or Neisseria.
The short list you will hear on rounds daily: Gram-positive cocci — Staphylococcus aureus (MSSA, MRSA), coagulase-negative staph (CoNS) including S. epidermidis, Streptococcus pneumoniae, group A strep (S. pyogenes), group B strep (S. agalactiae), viridans streptococci, Enterococcus faecalis and E. faecium (including VRE). Gram-positive bacilli — Listeria monocytogenes, Clostridium difficile, Clostridium perfringens, Bacillus, Corynebacterium. Gram-negative rods — Escherichia coli, Klebsiella pneumoniae, Proteus, Enterobacter, Serratia, Pseudomonas aeruginosa, Acinetobacter baumannii, Stenotrophomonas maltophilia. Gram-negative cocci / coccobacilli — Neisseria meningitidis, Neisseria gonorrhoeae, Moraxella catarrhalis, Haemophilus influenzae. Atypicals (no cell wall or poor Gram staining) — Mycoplasma pneumoniae, Chlamydia/Chlamydophila, Legionella pneumophila.
Mycobacteria — Mycobacterium tuberculosis, M. avium complex (MAC), M. kansasii, M. abscessus, M. leprae — have a waxy mycolic-acid cell wall and require an acid-fast bacilli (AFB) stain (Ziehl-Neelsen or auramine-rhodamine). Spirochetes include Treponema pallidum (syphilis), Borrelia burgdorferi (Lyme), and Leptospira.
Viruses, Fungi, and Parasites
Viruses are categorized by genome (DNA vs RNA), envelope, and family. The ones you will chart constantly are HIV-1, hepatitis A/B/C/D/E, influenza A/B, SARS-CoV-2, RSV, the human herpesviruses (HSV-1, HSV-2, VZV, EBV, CMV, HHV-6, HHV-8), and adenovirus. Fungi split into yeasts (Candida, Cryptococcus), molds (Aspergillus, Mucorales), dimorphic endemic fungi (Histoplasma, Blastomyces, Coccidioides), and Pneumocystis jirovecii. Parasites include protozoa (Plasmodium, Giardia, Entamoeba histolytica, Toxoplasma gondii, Trypanosoma, Leishmania), helminths (Strongyloides, schistosomes, Ascaris), and ectoparasites.
Host Immunity — The Second Half of the Equation
The same pathogen behaves very differently depending on the host's immune status. Key defects to document on every ID consult: neutropenia (ANC < 500) — high risk for bacterial and mold infections; lymphopenia / cell-mediated defect (HIV, steroids, transplant, biologics) — risk for intracellular pathogens, PJP, fungi, reactivation of latent herpes and TB; humoral / splenic defects — risk for encapsulated organisms (pneumococcus, meningococcus, Hib); barrier breaches — central lines, urinary catheters, surgical wounds, and burns break the skin/mucosal barrier and invite hospital flora.
Learn to hear the Gram stain call-back the way surgeons hear imaging reads. "Gram-positive cocci in clusters" → staph, start vancomycin. "Gram-positive cocci in chains or pairs" → strep/enterococcus. "Gram-negative rods" in a sick patient → pseudomonal coverage until speciated. "Yeast on Gram stain" → candidemia, call ophthalmology for fundoscopy. These reflexes save you from having to stop and ask the attending every time.
02 Scribe Documentation Framework
ID notes have a specific rhythm that differs from general internal medicine. They are consult-heavy, data-dense, and revolve around a structured "bug-drug-source" assessment. Whether you are charting a new inpatient consult, a daily follow-up, or an outpatient OPAT clinic visit, your job is to make the pathogen, site, severity, and treatment plan unmistakable in a single glance.
Reason for consult: The single clinical question, usually one sentence — "Please evaluate fever and gram-positive bacteremia," "Assist with antimicrobial selection in complicated pyelonephritis," "Evaluate for osteomyelitis of the first metatarsal."
HPI: Time course of the current illness, fevers/chills/rigors, prior antibiotic exposure (critical — always document dose, duration, and stop date), exposures (travel, animals, sick contacts, sexual history, IV drug use, tick/mosquito), immunizations, and relevant prior cultures.
Past medical history: Focus on immunocompromising conditions — HIV (with last CD4 and VL), diabetes, malignancy/chemotherapy, transplant, biologics, splenectomy, prior MDR organisms, indwelling hardware, and prior surgeries.
Objective: Vitals trend (especially Tmax, lowest MAP, highest lactate, pressor requirement), antibiotic-day counter ("hospital day 4, vancomycin day 3, cefepime day 2"), culture data in chronological order, imaging, and the focused exam.
Assessment & Plan: Written as a problem list: the suspected syndrome, the likely pathogen (or confirmed organism and susceptibilities), the source, and the antimicrobial plan with day-counted duration.
The Bug-Drug-Source-Duration Framework
Every ID plan answers four questions. Bug: what organism, confirmed or suspected? Drug: which antimicrobial, what dose, adjusted for renal function, and for how long? Source: where is the infection anatomically, and does it need source control (drainage, debridement, line removal, hardware removal)? Duration: what is the planned stop date, counted from day 1 of effective therapy or from source control?
Day 1 of antibiotics is the first calendar day a patient receives an effective agent. ID attendings will reference antibiotic days on every round — "she's on vancomycin day 6, cefepime day 4, planned 14-day total from first negative blood culture." Always capture the start date, current day, and planned stop date for every antimicrobial. The "clock" sometimes resets from day 1 of source control (e.g., bacteremia day 1 = first negative culture after drainage).
Never write "on antibiotics" without specifying the drug, dose, route, indication, and day number. ID attendings will correct you immediately. Also: prior outpatient antibiotic exposures in the last 90 days dramatically change empiric coverage and must be in the HPI, not buried in the PMH.
03 Sepsis, Septic Shock & Bacteremia Syndrome
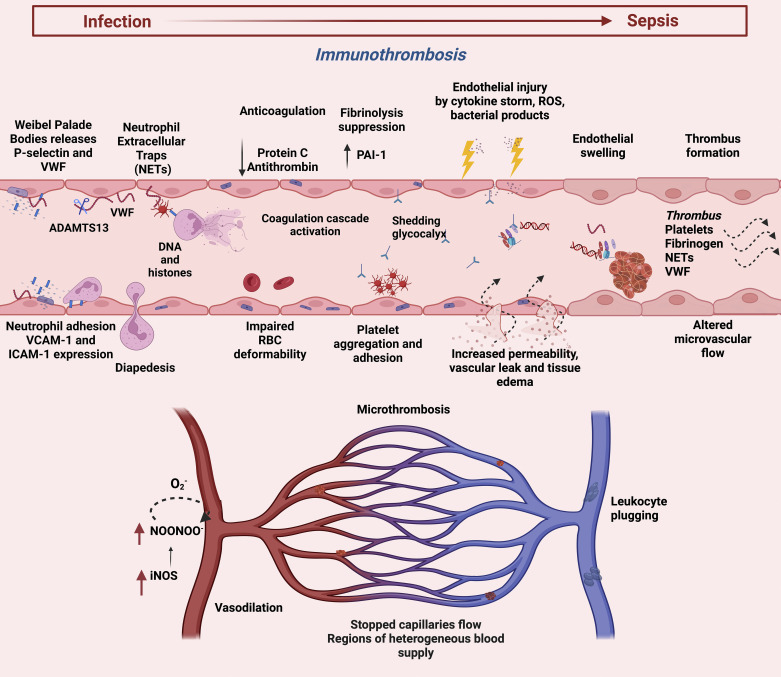
Sepsis is life-threatening organ dysfunction caused by a dysregulated host response to infection. It is the single most common reason ID is consulted in the hospital, and it is the clinical scenario in which the speed and precision of your note matters most. The modern definition (Sepsis-3) emphasizes organ dysfunction rather than the older SIRS criteria alone, though you will still see both referenced.
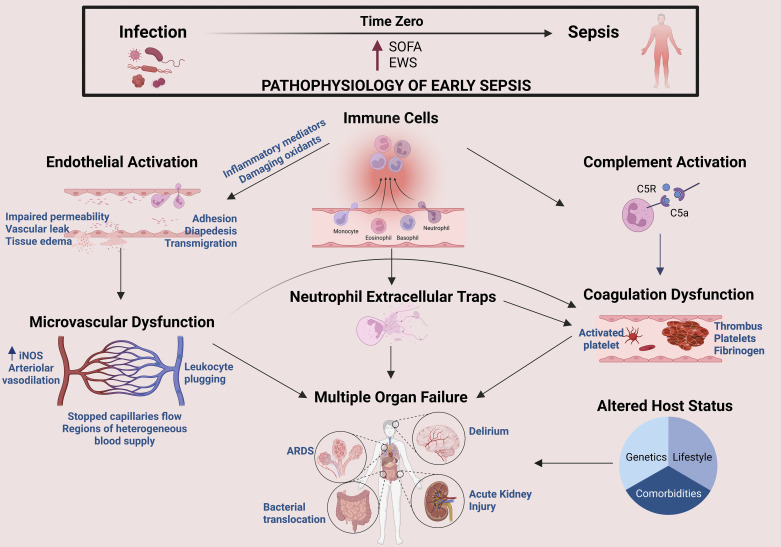
Temperature > 38°C or < 36°C · Heart rate > 90 · Respiratory rate > 20 or PaCO2 < 32 · WBC > 12K, < 4K, or > 10% bands.
Altered mental status (GCS < 15) · Respiratory rate ≥ 22 · Systolic BP ≤ 100. A score ≥ 2 warrants full SOFA workup and escalation.
Six organ systems each scored 0–4: respiration (PaO2/FiO2), coagulation (platelets), liver (bilirubin), cardiovascular (MAP, pressors), CNS (GCS), and renal (creatinine, urine output). A rise of ≥ 2 points from baseline defines sepsis in a patient with suspected infection.
Sepsis with persistent hypotension requiring vasopressors to maintain MAP ≥ 65 mmHg and serum lactate > 2 mmol/L despite adequate fluid resuscitation. Hospital mortality exceeds 40%.
The Hour-1 Bundle
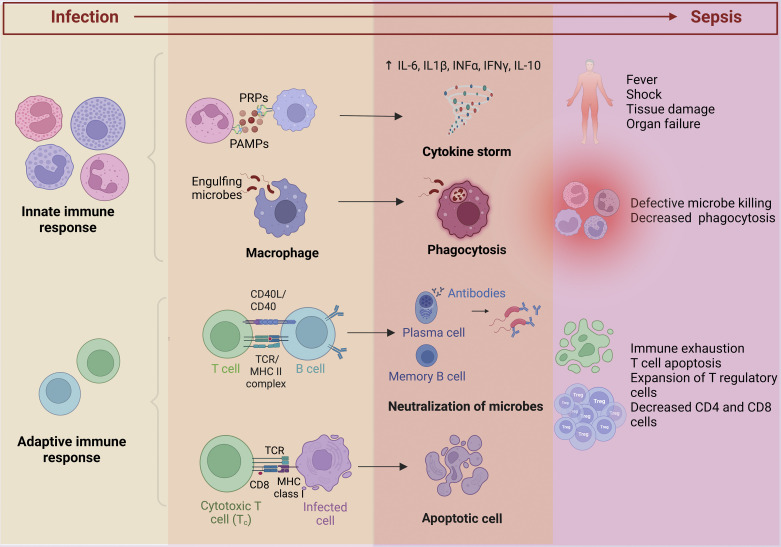
The Surviving Sepsis Campaign 2021 guidelines specify a one-hour bundle: measure lactate, obtain blood cultures before antibiotics, administer broad-spectrum antibiotics, begin 30 mL/kg crystalloid for hypotension or lactate ≥ 4, and start vasopressors (norepinephrine first-line) if MAP remains < 65 after fluids. Scribes must document exact timestamps for lactate, cultures, and first antibiotic dose — these are quality measures reported publicly.
Bacteremia
Bacteremia is bacteria in the blood, confirmed by blood culture. Document the organism, number of positive bottles and sets, time to positivity, and source. Staphylococcus aureus bacteremia (SAB) always requires ID consult, repeat blood cultures every 48–72h until clearance, transthoracic or transesophageal echocardiogram to rule out endocarditis, and a minimum 14-day course for uncomplicated SAB or 4–6 weeks for complicated disease. Gram-negative bacteremia typically needs 7–14 days of an effective agent after source control.
Unknown source, community: ceftriaxone + vancomycin.
Unknown source, hospital or MDR risk: cefepime (or piperacillin-tazobactam or meropenem) + vancomycin.
Neutropenic fever: cefepime or piperacillin-tazobactam; add vancomycin if line infection, SSTI, or hemodynamic instability.
Intra-abdominal: piperacillin-tazobactam or meropenem; add antifungal if Candida risk.
Necrotizing fasciitis: piperacillin-tazobactam + vancomycin + clindamycin (for toxin suppression).
The most common scribe error in sepsis: writing "blood cultures pending" without documenting when they were drawn. Always chart the exact time of blood culture collection and the exact time the first antibiotic dose was given. The time gap between them is a publicly reported quality measure (SEP-1).
04 Infective Endocarditis & Line Infections Cardiac
Infective endocarditis (IE) is infection of the endocardial surface, usually a valve, producing a vegetation that embolizes and seeds distal organs. Left-sided IE (aortic, mitral) is the classic; right-sided IE (tricuspid) is associated with injection drug use. Native valve, prosthetic valve, and cardiac device IE are separate entities with different pathogens and durations.
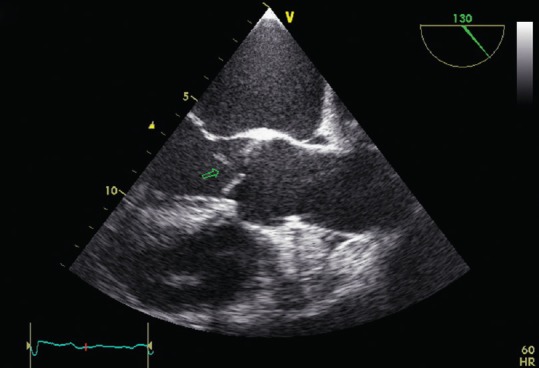
Duke Criteria
Diagnosis relies on the modified Duke criteria. Definite IE requires either 2 major, 1 major + 3 minor, or 5 minor criteria.
Blood culture positive for IE: typical organisms (viridans strep, S. bovis/S. gallolyticus, HACEK, S. aureus, community-acquired enterococci without primary focus) from two separate cultures, or persistently positive cultures, or a single positive blood culture for Coxiella burnetii.
Evidence of endocardial involvement: echocardiogram with vegetation, abscess, or new valve dehiscence; or new valvular regurgitation.
Predisposition (IVDU, predisposing heart condition) · Fever > 38°C · Vascular phenomena (septic emboli, mycotic aneurysm, Janeway lesions, conjunctival hemorrhage) · Immunologic phenomena (Osler nodes, Roth spots, glomerulonephritis, positive RF) · Microbiologic evidence not meeting a major criterion.
Management
The AHA Scientific Statement on Infective Endocarditis is the primary U.S. reference. Empiric coverage for native valve IE typically includes vancomycin plus ceftriaxone; prosthetic valve IE adds gentamicin and rifampin. Duration is 4–6 weeks of parenteral therapy from the first negative blood culture. Surgical consultation is indicated for heart failure, uncontrolled infection, large vegetations, or embolic complications.
Central Line-Associated Bloodstream Infection (CLABSI)
Any central venous catheter, PICC, tunneled catheter, or port can seed the bloodstream. Suspect line infection when blood cultures grow staphylococci, enterococci, candida, or gram-negative rods in a patient with a line > 48 hours in place. Differential time to positivity > 2 hours (line culture positive at least 2 hours earlier than peripheral) strongly suggests catheter source. Management: remove the line for S. aureus, candida, pseudomonas, or persistent bacteremia; consider line salvage with antibiotic lock therapy for selected coag-negative staph or enterococcal infections.
05 Pneumonia (CAP, HAP, VAP, Aspiration) Respiratory
Pneumonia is categorized by where it was acquired because the pathogens and empiric therapy differ dramatically. Community-acquired pneumonia (CAP) develops outside the hospital. Hospital-acquired pneumonia (HAP) develops ≥ 48 hours after admission. Ventilator-associated pneumonia (VAP) develops ≥ 48 hours after intubation. Aspiration pneumonia occurs after macroaspiration of oropharyngeal or gastric contents.
CAP — Pathogens & Severity
The dominant CAP pathogens are S. pneumoniae, H. influenzae, atypicals (Mycoplasma, Chlamydia pneumoniae, Legionella), and respiratory viruses. S. aureus (especially post-influenza) and gram-negatives appear in severe or structural lung disease.
Confusion · Urea (BUN) > 19 mg/dL · Respiratory rate ≥ 30 · Blood pressure (SBP < 90 or DBP ≤ 60) · Age ≥ 65.
0–1 = outpatient; 2 = short admission or observation; 3–5 = inpatient, consider ICU. PSI/PORT is the alternative and more granular tool.
Outpatient, no comorbidities: amoxicillin or doxycycline or a macrolide (if local pneumococcal resistance < 25%).
Outpatient with comorbidities: amoxicillin-clavulanate or cephalosporin + macrolide or doxycycline; or a respiratory fluoroquinolone (levofloxacin, moxifloxacin).
Inpatient, non-severe: ceftriaxone + azithromycin, or a respiratory fluoroquinolone.
Inpatient, severe (ICU): beta-lactam + azithromycin or beta-lactam + respiratory fluoroquinolone. Add MRSA coverage (vancomycin or linezolid) and antipseudomonal coverage only if risk factors present.
HAP / VAP
Empiric therapy per the 2016 IDSA/ATS HAP/VAP guideline is driven by unit-specific antibiograms and individual MDR risk factors. A typical regimen is piperacillin-tazobactam or cefepime + vancomycin, escalating to dual antipseudomonal coverage (adding a fluoroquinolone or aminoglycoside) for septic shock or high MDR risk. De-escalate at 48–72 hours based on cultures.
Aspiration Pneumonia vs Pneumonitis
Aspiration of acidic gastric contents causes chemical pneumonitis, which often improves without antibiotics. Bacterial aspiration pneumonia follows macroaspiration in a patient with impaired consciousness, dysphagia, or poor dentition, and involves oral anaerobes and streptococci; treat with ampicillin-sulbactam or ceftriaxone (routine anaerobic coverage is no longer recommended for most cases).
06 Tuberculosis & Nontuberculous Mycobacteria Respiratory
Tuberculosis remains a top global infectious killer. As a scribe you will chart latent TB infection (LTBI), active pulmonary TB, extrapulmonary TB, and NTM disease — and you must understand the isolation implications of each.
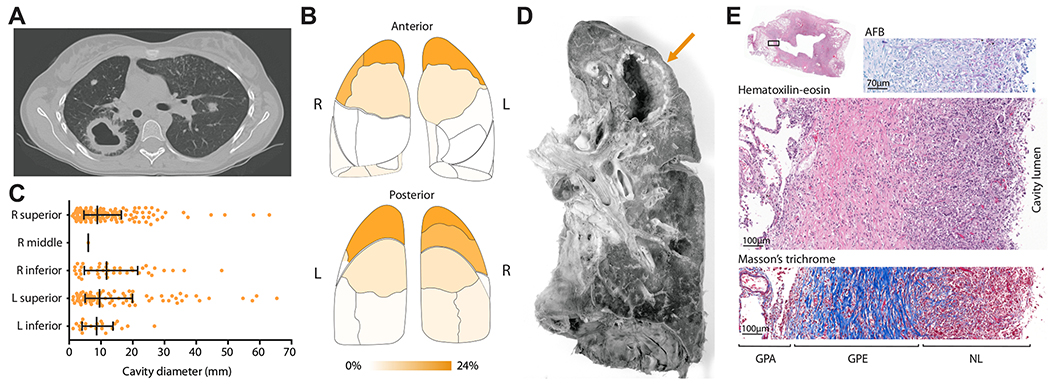
Latent vs Active TB
Latent TB infection (LTBI) is immune-contained infection with no symptoms, negative chest X-ray, and positive screening test (TST or IGRA). Active pulmonary TB presents with chronic cough, fever, night sweats, weight loss, hemoptysis, and upper-lobe cavitation on imaging. Extrapulmonary sites include lymph nodes (scrofula), pleural space, bone (Pott disease), meninges, GU tract, peritoneum, and miliary dissemination.
Diagnostics
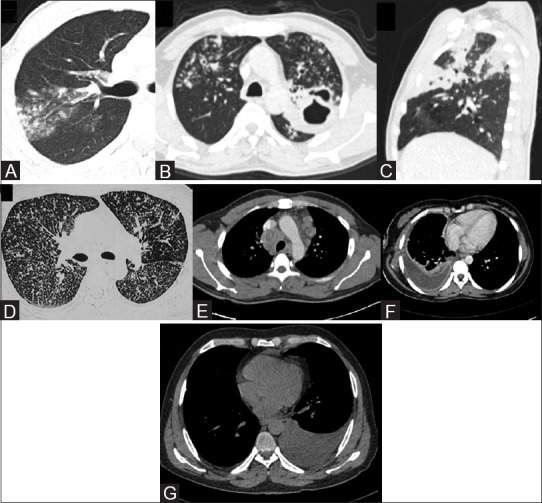
Screening uses the tuberculin skin test (TST/PPD) with stratified cutoffs (5 mm for HIV, close contacts, organ transplant, immunosuppressed, fibrotic CXR; 10 mm for recent immigrants, IVDU, healthcare workers, prisoners, diabetics; 15 mm for low-risk), or interferon-gamma release assays (IGRAs) such as QuantiFERON-TB Gold or T-SPOT. Active disease requires three sputum samples for AFB smear and culture, nucleic acid amplification testing (Xpert MTB/RIF), and chest imaging.
Standard regimen is 2 months of RIPE — Rifampin, Isoniazid, Pyrazinamide, Ethambutol — followed by 4 months of rifampin + isoniazid. Directly observed therapy (DOT) is standard. Drug-resistant TB (MDR-TB, XDR-TB) requires specialist regimens of 9–24 months.
Suspected active pulmonary TB requires immediate negative-pressure airborne isolation with N95 respirators until three separate AFB smears are negative. Never wait for culture results to initiate isolation — document time of first smear and isolation status in every progress note.
LTBI treatment options include 3 months of weekly rifapentine + isoniazid (3HP), 4 months of daily rifampin, or 9 months of daily isoniazid. NTM disease (MAC, M. abscessus, M. kansasii) typically requires multi-drug therapy for 12–18 months and is far less contagious than M. tuberculosis.
07 Viral Respiratory Infections Respiratory
Influenza, SARS-CoV-2, and RSV are the big three viral respiratory pathogens in adults. Rhinovirus, parainfluenza, adenovirus, and human metapneumovirus round out the viral panel. Multiplex PCR respiratory panels report all of these simultaneously.
Influenza
Seasonal influenza A (H1N1, H3N2) and B present with abrupt fever, myalgia, cough, sore throat, and headache. Rapid antigen and PCR confirm diagnosis. Treatment with oseltamivir 75 mg BID for 5 days is most effective within 48 hours of symptom onset but is recommended for any hospitalized patient or high-risk outpatient regardless of duration. Baloxavir is a single-dose alternative.
COVID-19
SARS-CoV-2 infection ranges from asymptomatic to severe pneumonia with ARDS. Severity is stratified as mild, moderate (hypoxia but no supplemental O2), severe (SpO2 < 94%, requires O2), or critical (mechanical ventilation, shock, multi-organ failure). NIH treatment guidelines recommend nirmatrelvir-ritonavir (Paxlovid) for non-hospitalized high-risk patients within 5 days of symptom onset; remdesivir, dexamethasone, and immunomodulators (baricitinib, tocilizumab) for hospitalized patients by oxygen requirement tier.
RSV
Respiratory syncytial virus is a major cause of bronchiolitis in infants and severe lower respiratory infection in older adults and immunocompromised hosts. Treatment is supportive; ribavirin is occasionally used in transplant recipients. RSV vaccines are now recommended for adults ≥ 60 and for pregnancy to protect newborns.
08 Herpesviruses (HSV, VZV, EBV, CMV) Viral
All eight human herpesviruses establish lifelong latency after primary infection and can reactivate in immunocompromised hosts. The four you will chart most on ID consults:
HSV-1 / HSV-2: orolabial and genital herpes, herpetic whitlow, eczema herpeticum, HSV encephalitis (temporal lobe, treat with IV acyclovir), neonatal HSV. Confirm with PCR from lesion swab or CSF.
VZV (varicella-zoster): primary infection causes chickenpox; reactivation causes shingles (zoster) in a dermatomal distribution. Disseminated zoster, zoster ophthalmicus (V1), and Ramsay Hunt syndrome (facial nerve) require systemic acyclovir, valacyclovir, or famciclovir. The recombinant zoster vaccine (Shingrix) is recommended for adults ≥ 50 and immunocompromised adults ≥ 19.
EBV: causes infectious mononucleosis (fever, pharyngitis, lymphadenopathy, splenomegaly, atypical lymphocytes, positive monospot), post-transplant lymphoproliferative disorder (PTLD), and is associated with Burkitt lymphoma, nasopharyngeal carcinoma, and Hodgkin lymphoma. Usually self-limited; avoid amoxicillin (morbilliform rash).
CMV: major pathogen in transplant and HIV. Causes CMV retinitis, colitis, esophagitis, pneumonitis, and hepatitis. Diagnosed by quantitative PCR viral load, biopsy with owl-eye inclusions, or retinal exam. Treat with ganciclovir/valganciclovir; alternatives include foscarnet, cidofovir, letermovir (prophylaxis), and maribavir (resistant disease).
09 HIV & AIDS-Defining Infections Viral
HIV care is an enormous component of outpatient ID. You will chart routine follow-ups, new diagnoses, PrEP/PEP visits, opportunistic infections, and ART regimen changes. The DHHS Adult and Adolescent ARV Guidelines are the definitive U.S. reference.
CDC HIV Staging
Stage 0: Early HIV infection (negative or indeterminate test within 180 days of a positive test).
Stage 1: CD4 ≥ 500 cells/μL (or ≥ 26%), no AIDS-defining illness.
Stage 2: CD4 200–499 (or 14–25%), no AIDS-defining illness.
Stage 3 (AIDS): CD4 < 200 (or < 14%) or documented AIDS-defining condition.
Stage U: Unknown (insufficient information).
AIDS-Defining Opportunistic Infections
CD4 < 200: Pneumocystis jirovecii pneumonia (PJP), esophageal candidiasis, Kaposi sarcoma. CD4 < 100: toxoplasmosis encephalitis, cryptococcal meningitis, chronic cryptosporidiosis. CD4 < 50: disseminated MAC, CMV retinitis and colitis, CNS lymphoma, progressive multifocal leukoencephalopathy (PML). Prophylaxis thresholds: TMP-SMX for PJP at CD4 < 200; TMP-SMX for toxo at CD4 < 100 with positive IgG; azithromycin for MAC at CD4 < 50 (no longer routine if starting effective ART).
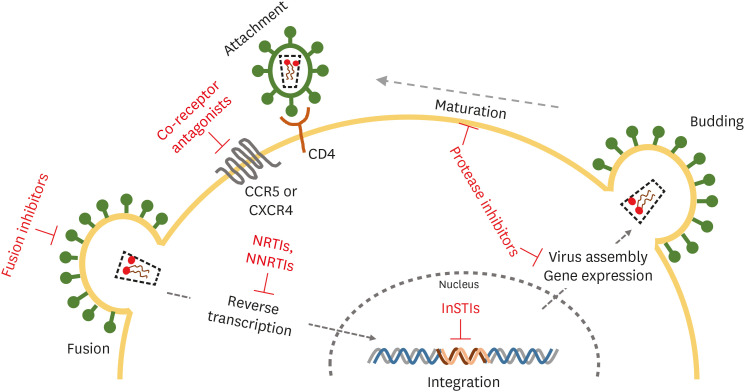
ART and Screening
Modern first-line ART is almost always an integrase strand-transfer inhibitor (INSTI)-based regimen: bictegravir/emtricitabine/tenofovir alafenamide (Biktarvy) or dolutegravir + emtricitabine/TAF or TDF. The USPSTF 2019 HIV screening recommendation calls for one-time screening in adolescents and adults 15–65 and in pregnancy. Pre-exposure prophylaxis (PrEP) options include daily tenofovir/emtricitabine, tenofovir alafenamide/emtricitabine, or injectable cabotegravir every 2 months. Post-exposure prophylaxis (PEP) must begin within 72 hours of exposure and runs for 28 days.
On every HIV follow-up note, document the most recent CD4 count and HIV viral load with dates. "VL undetectable" is not enough — write "VL < 20 copies/mL (Feb 2026)" so the trajectory is clear.
10 Viral Hepatitis (A, B, C, D, E) Viral
Hepatitis A is fecal-oral, self-limited, and prevented by vaccine. Hepatitis E is similarly transmitted and can cause fulminant disease in pregnancy. These rarely need ID management beyond supportive care.
Hepatitis B (HBV) is a DNA virus transmitted via blood, sexual contact, and perinatally. Serology is layered: HBsAg (current infection), anti-HBs (immunity from vaccine or prior infection), anti-HBc IgM (acute), anti-HBc IgG (past exposure), HBeAg and HBV DNA (active replication). Treat chronic HBV with entecavir or tenofovir in patients meeting guideline criteria. The USPSTF HBV screening recommendation supports screening adolescents and adults at increased risk; the CDC now recommends universal adult HBV screening at least once. Hepatitis D (delta) only occurs as co- or superinfection with HBV and worsens prognosis.
Hepatitis C (HCV) is an RNA virus primarily transmitted parenterally. Chronic HCV causes cirrhosis and hepatocellular carcinoma. Direct-acting antivirals (DAAs) such as glecaprevir/pibrentasvir (Mavyret) or sofosbuvir/velpatasvir (Epclusa) produce sustained virologic response (cure) > 95% in 8–12 weeks. The USPSTF 2020 HCV screening recommendation calls for universal one-time screening in all adults 18–79.
11 Sexually Transmitted Infections GU
STIs account for a large share of outpatient ID visits. The core set: gonorrhea, chlamydia, syphilis, trichomoniasis, HSV, HPV, and HIV (covered separately). Document exposure history frankly: number of partners in the last 12 months, gender of partners, anatomic sites of exposure, condom use, and last known negative test.
Gonorrhea & Chlamydia
Neisseria gonorrhoeae and Chlamydia trachomatis cause urethritis, cervicitis, proctitis, pharyngitis, and pelvic inflammatory disease (PID). Diagnose by NAAT from the appropriate anatomic site. Per the CDC 2021 STI Treatment Guidelines, treat gonorrhea with ceftriaxone 500 mg IM (1 g if ≥ 150 kg) and chlamydia with doxycycline 100 mg BID × 7 days.
Syphilis
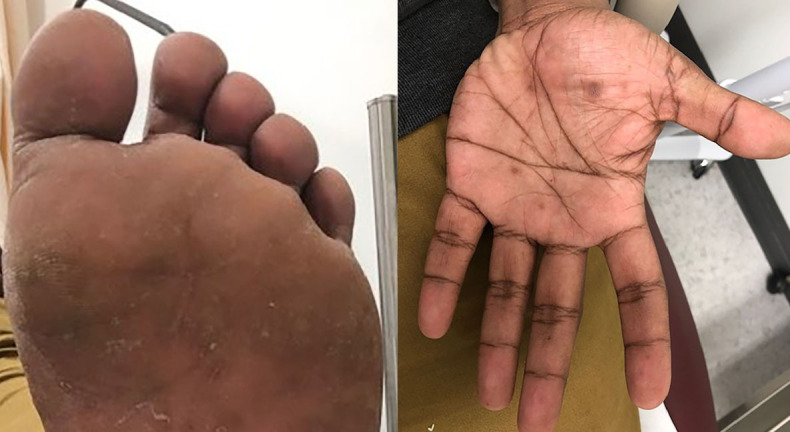
Treponema pallidum progresses through stages: primary (painless chancre 3 weeks after exposure), secondary (diffuse maculopapular rash including palms and soles, condyloma lata, mucous patches, 6–12 weeks later), latent (asymptomatic; early latent < 1 year, late latent > 1 year), and tertiary (gummas, cardiovascular syphilis, neurosyphilis). Screening uses non-treponemal tests (RPR, VDRL) confirmed by treponemal tests (FTA-ABS, TP-PA, EIA). Treat primary, secondary, and early latent with benzathine penicillin G 2.4 million units IM × 1; late latent and tertiary with 2.4 million units weekly × 3; neurosyphilis with IV penicillin G 18–24 million units/day for 10–14 days.
Other STIs to recognize: trichomoniasis (metronidazole), genital HSV (valacyclovir), genital warts / HPV (cryotherapy, imiquimod), chancroid, lymphogranuloma venereum (LGV), and Mycoplasma genitalium.
12 Urinary Tract Infections & Pyelonephritis GU
UTIs span uncomplicated cystitis, complicated UTI, pyelonephritis, catheter-associated UTI, and urosepsis. E. coli causes > 80% of community UTIs; Klebsiella, Proteus, enterococci, staphylococci, and pseudomonas appear in complicated and catheter-associated disease.
Uncomplicated cystitis: nitrofurantoin 100 mg BID × 5 days, TMP-SMX DS BID × 3 days, or fosfomycin 3 g × 1.
Pyelonephritis, outpatient: ciprofloxacin 500 mg BID × 7 days or levofloxacin 750 mg daily × 5 days.
Pyelonephritis, inpatient: ceftriaxone 1 g IV daily, piperacillin-tazobactam, or carbapenem if MDR risk.
Asymptomatic bacteriuria should not be treated except in pregnancy or before urologic procedures with anticipated mucosal bleeding. Treating bacteriuria in catheterized, nursing home, or elderly patients without symptoms is a major stewardship error.
13 Skin & Soft Tissue Infections SSTI
SSTIs range from superficial impetigo and erysipelas to limb-threatening necrotizing fasciitis. Scribes must capture the exact borders (outline marked on skin with date/time), drainage, crepitus, and systemic signs.
Cellulitis & Erysipelas
Cellulitis is infection of the dermis and subcutaneous tissue presenting with erythema, warmth, tenderness, and ill-defined borders. Erysipelas is the more superficial cousin with sharply demarcated borders ("raised, advancing edge"). Streptococci and S. aureus dominate; treat per the IDSA 2014 SSTI Guideline with cephalexin or dicloxacillin for non-purulent, adding MRSA coverage (TMP-SMX, doxycycline, clindamycin, or vancomycin for severe) for purulent or abscess-associated disease.
Abscess
Incision and drainage is the primary therapy; antibiotics are added for > 2 cm lesions, systemic signs, immunocompromise, or failure to respond to I&D alone.
Pain out of proportion, rapidly advancing erythema, crepitus, bullae, gray "dishwater" drainage, systemic toxicity. LRINEC score aids risk stratification but never delays surgery. Management: immediate broad-spectrum antibiotics (vancomycin + piperacillin-tazobactam + clindamycin) and emergent surgical debridement. Mortality 20–40%.
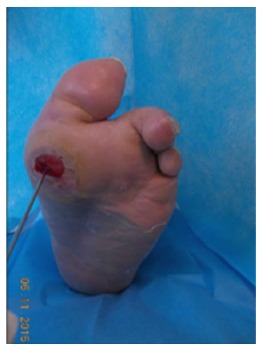
Diabetic Foot Infection (DFI)
Classified by IDSA as mild (superficial, < 2 cm erythema), moderate (deeper or > 2 cm), or severe (SIRS). Assess always for underlying osteomyelitis with probe-to-bone test and MRI. Empiric therapy depends on severity, prior exposure, and MRSA/pseudomonal risk.
14 Bone & Joint Infections MSK
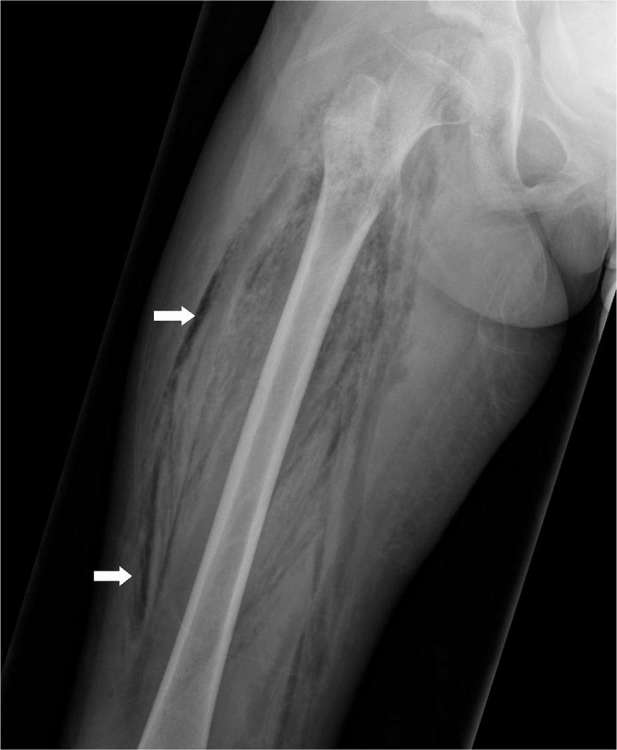
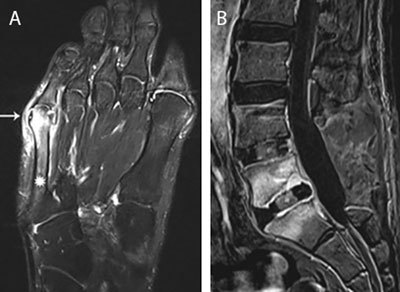
Osteomyelitis is infection of bone from hematogenous seeding, contiguous spread, or direct inoculation. Vertebral osteomyelitis presents with progressive back pain, fever, and elevated inflammatory markers; diagnosis requires MRI and CT-guided biopsy for culture before antibiotics when possible. Most common organisms: S. aureus, coagulase-negative staph, streptococci, gram-negatives, and (in IVDU) Pseudomonas. Treatment is typically 6 weeks of pathogen-directed IV or highly bioavailable oral therapy.
Septic arthritis presents with acute monoarthritis, fever, and a painful, hot, swollen joint with restricted range of motion. Arthrocentesis is mandatory: synovial fluid WBC > 50,000 (typically > 75% neutrophils), Gram stain, culture, and crystal analysis. Empiric therapy (vancomycin + ceftriaxone) is started after joint fluid is drawn, with urgent orthopedic washout. Gonococcal arthritis is a separate entity with migratory polyarthralgias, tenosynovitis, and dermatitis.
Prosthetic joint infection (PJI) is diagnosed by periprosthetic tissue or fluid culture, imaging, and inflammatory markers. Management is surgical (DAIR, one-stage or two-stage exchange) combined with prolonged antibiotics per the IDSA PJI Guideline.
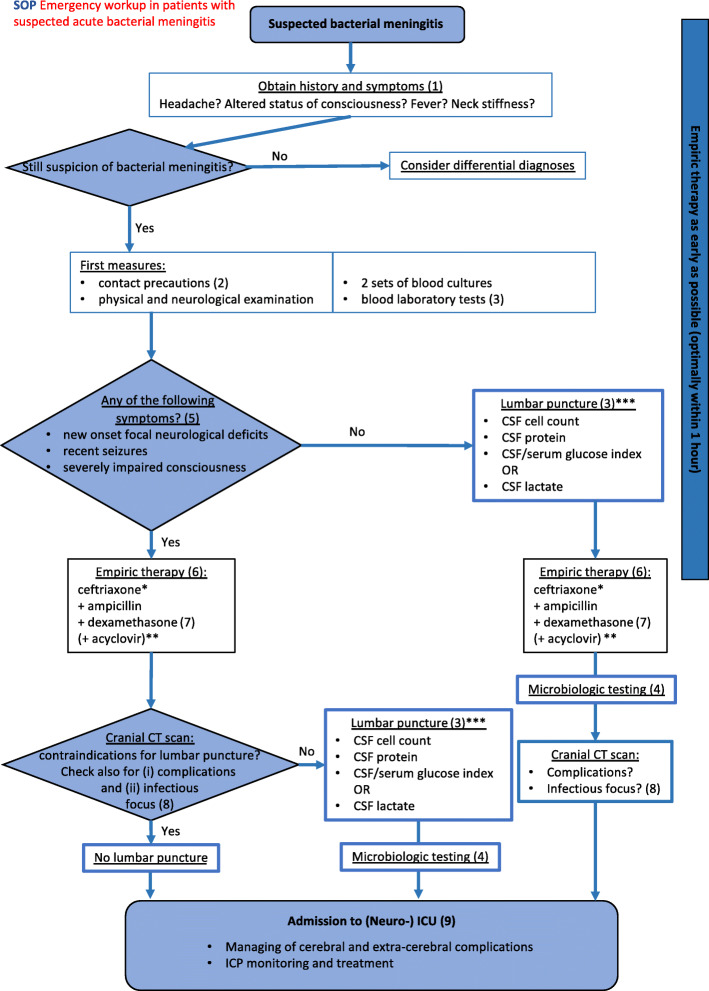
15 CNS Infections — Meningitis, Encephalitis, Abscess CNS
Bacterial Meningitis
Acute bacterial meningitis presents with fever, headache, neck stiffness, photophobia, and altered mental status. The classic triad is present in only about half. Dominant pathogens by age: neonates — group B strep, E. coli, Listeria; children/adults — S. pneumoniae, N. meningitidis; > 50 or immunocompromised — add Listeria. Empiric therapy: vancomycin + ceftriaxone, adding ampicillin for Listeria coverage in the young, old, pregnant, or immunocompromised, and adjunctive dexamethasone 10 mg IV every 6 hours for suspected pneumococcal meningitis (start before or with the first antibiotic dose).
| Parameter | Bacterial | Viral | Fungal/TB |
|---|---|---|---|
| Opening pressure | Elevated | Normal | Very high |
| WBC | 1000–5000, PMN | 50–1000, lymph | 100–500, lymph |
| Protein | > 100 | < 100 | 100–500 |
| Glucose | < 40 (low) | Normal | Low |
| Gram stain | Positive ~60% | Negative | Negative |
Viral Meningitis & Encephalitis
Enteroviruses cause most viral meningitis (self-limited). HSV encephalitis presents with fever, altered mental status, seizures, and focal temporal lobe findings; start empiric acyclovir 10 mg/kg IV every 8 hours while awaiting HSV PCR. Other viral encephalitides include VZV, West Nile, arboviruses, and (in children) measles and mumps.
Brain Abscess
Usually polymicrobial from contiguous (otitis, sinusitis, dental) or hematogenous spread. Management combines surgical drainage and prolonged antibiotics (typically 6–8 weeks of a beta-lactam + metronidazole, with vancomycin for staph coverage when indicated).
16 GI Infections — C. difficile & Intra-abdominal GI
Clostridioides difficile Infection (CDI)
Risk factors: recent antibiotics (especially clindamycin, fluoroquinolones, cephalosporins), hospitalization, advanced age, PPIs. Diagnosis is by stool NAAT or GDH + toxin EIA in a symptomatic patient (≥ 3 unformed stools in 24 hours). Severity: non-severe (WBC < 15,000, Cr < 1.5), severe (WBC ≥ 15,000 or Cr ≥ 1.5), fulminant (hypotension, ileus, megacolon). Per the IDSA/SHEA 2021 CDI Focused Update, first-line therapy is oral fidaxomicin 200 mg BID × 10 days (preferred) or vancomycin 125 mg PO QID × 10 days. Fulminant CDI adds IV metronidazole and may require colectomy.
Intra-Abdominal Infections
Peritonitis, intra-abdominal abscess, cholangitis, and diverticulitis. Community-acquired: piperacillin-tazobactam, ceftriaxone + metronidazole, or a carbapenem for severe disease. Hospital-acquired or high-risk: carbapenem ± antifungal. Source control — drainage or surgery — is mandatory. Spontaneous bacterial peritonitis (SBP) in cirrhotics is diagnosed by ascitic PMN ≥ 250; treat with ceftriaxone or cefotaxime and add IV albumin in selected patients.
17 Neutropenic Fever & Immunocompromised Host Host
Neutropenic fever is defined as a single oral temperature ≥ 38.3°C (or ≥ 38.0°C sustained over an hour) in a patient with ANC < 500 (or expected to drop below 500 within 48 hours). It is a medical emergency. Per the IDSA 2010 Fever in Neutropenia Guideline, empiric antipseudomonal coverage (cefepime, piperacillin-tazobactam, or meropenem) should be administered within 60 minutes of presentation. Add vancomycin for hemodynamic instability, line infection, SSTI, or known MRSA colonization. Add antifungal coverage (echinocandin or voriconazole) for persistent fever after 4–7 days despite broad-spectrum antibiotics.
Other compromised hosts: solid organ and stem cell transplant recipients (watch for CMV, EBV, BK virus, invasive fungi, PJP), patients on biologics (TNF inhibitors reactivate TB, histoplasmosis; rituximab reactivates HBV), and patients on high-dose steroids (PJP, strongyloides hyperinfection).
18 Travel & Tropical Medicine Travel
The "fever in the returning traveler" is a classic ID consult. Your HPI must capture: countries visited, rural vs urban, dates of travel, accommodations, food and water exposures, insect bites, freshwater exposure, animal contact, sexual exposures, pre-travel vaccines, and malaria chemoprophylaxis (drug, compliance, stop date).
Malaria
Plasmodium falciparum causes the most severe disease and can kill within 24 hours. Other species: P. vivax, P. ovale (both cause relapses from hepatic hypnozoites and require primaquine), P. malariae, and P. knowlesi. Diagnose by thick and thin blood smears (for parasite density and species) or rapid antigen test. Treat uncomplicated falciparum with artemether-lumefantrine (Coartem) or atovaquone-proguanil; severe falciparum with IV artesunate.
Other Travel Infections
Typhoid (Salmonella Typhi), dengue, chikungunya, Zika, leptospirosis, amebic liver abscess (E. histolytica), schistosomiasis, and traveler's diarrhea (ETEC, Campylobacter, Shigella, Giardia). Fever in a traveler returning from sub-Saharan Africa is malaria until proven otherwise.
19 Tick-Borne & Zoonotic Infections Zoonotic
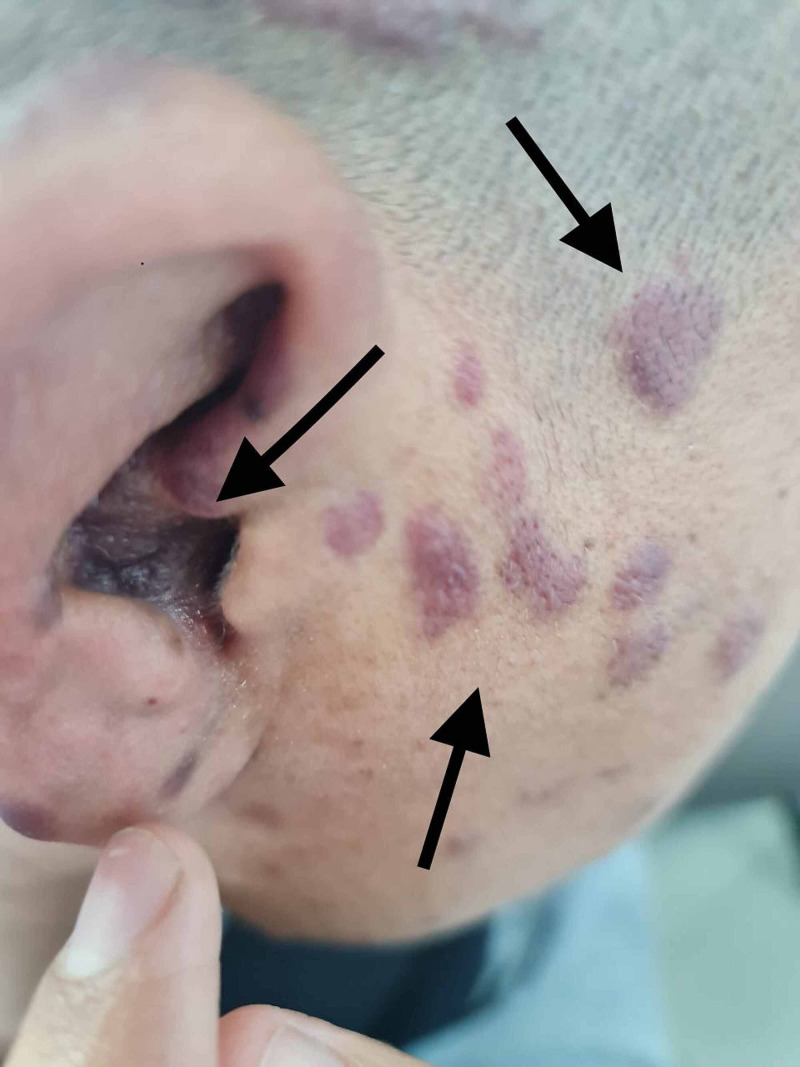
Lyme disease (Borrelia burgdorferi, Ixodes tick) progresses through early localized (erythema migrans target rash), early disseminated (multiple EM, carditis with AV block, facial palsy, meningitis), and late (arthritis, neuroborreliosis) stages. Treat early disease with doxycycline 100 mg BID × 10–21 days; IV ceftriaxone for neuro or cardiac involvement.
Rocky Mountain spotted fever (RMSF) (Rickettsia rickettsii, Dermacentor) causes fever, headache, and a centripetal petechial rash that begins on the wrists and ankles. Mortality is high without early doxycycline (start empirically on suspicion — do not wait for serology).
Human monocytic ehrlichiosis (HME), human granulocytic anaplasmosis (HGA), and babesiosis (Babesia microti, malaria-like intraerythrocytic parasites, Maltese cross form) are additional tick-borne infections in the U.S. Northeast and Upper Midwest. Doxycycline covers most; babesiosis is treated with atovaquone + azithromycin or clindamycin + quinine.
Other zoonoses: brucellosis (unpasteurized dairy, livestock), tularemia, leptospirosis (freshwater exposure), cat-scratch disease (Bartonella henselae), Q fever (Coxiella burnetii), and rabies.
20 Fungal Infections Fungal
Candida
Invasive candidiasis, candidemia, and deep-seated candida infections are managed per the IDSA 2016 Candidiasis Guideline. First-line therapy for candidemia is an echinocandin (caspofungin, micafungin, or anidulafungin), with step-down to fluconazole for susceptible isolates. Mandatory workup includes repeat blood cultures until clearance, ophthalmologic exam for endophthalmitis, central line removal, and TTE/TEE if bacteremia is persistent.
Aspergillus
Invasive aspergillosis occurs primarily in neutropenic, transplant, and prolonged-steroid patients. Presents with pulmonary nodules, halo sign on CT, hemoptysis, or disseminated disease. Diagnose with serum galactomannan, BAL galactomannan, beta-D-glucan, and tissue biopsy. Treat with voriconazole or isavuconazole.
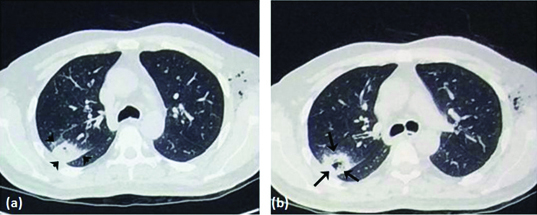
Endemic Mycoses
Histoplasmosis (Ohio and Mississippi River valleys; bat and bird droppings; intracellular yeast in macrophages). Blastomycosis (Midwest, Great Lakes; broad-based budding yeast; pulmonary and cutaneous disease). Coccidioidomycosis (Southwest US, "Valley fever"; spherules with endospores; pulmonary disease with possible dissemination to meninges, skin, bone). Treat with itraconazole for mild-moderate disease, amphotericin B for severe or disseminated disease.
Pneumocystis jirovecii Pneumonia (PJP)
Opportunistic pneumonia in HIV (CD4 < 200), transplant, and steroid-treated patients. Presents with subacute dyspnea, dry cough, and hypoxia with diffuse bilateral infiltrates and elevated LDH and beta-D-glucan. Diagnose by induced sputum or BAL with silver stain or PCR. Treat with TMP-SMX 15–20 mg/kg/day divided TID-QID for 21 days; add adjunctive steroids for room air PaO2 < 70 or A-a gradient > 35.
Cryptococcus & Toxoplasma
Cryptococcal meningitis (C. neoformans) is a classic HIV opportunistic infection at CD4 < 100; diagnose by CSF cryptococcal antigen, India ink, or culture; treat with induction amphotericin B + flucytosine, then consolidation fluconazole. Toxoplasmosis encephalitis presents with ring-enhancing brain lesions and positive IgG; treat with pyrimethamine + sulfadiazine + leucovorin.
21 Procedures — Cultures, LP, Paracentesis, I&D Procedure
ID is a largely cognitive specialty, but a handful of bedside procedures are essential for diagnosis and source control. You will document these on consult notes, procedure notes, and sometimes assist with timeout documentation.
| Procedure | Indication | Scribe Documentation |
|---|---|---|
| Blood cultures | Sepsis, bacteremia workup, endocarditis, neutropenic fever | Two sets from two separate sites before antibiotics; document draw time, site, number of bottles |
| Lumbar puncture | Meningitis, encephalitis, subarachnoid hemorrhage, CNS malignancy | Consent, position, level, opening pressure, appearance, tube distribution, closing pressure |
| Paracentesis | Ascites diagnosis, SBP workup, therapeutic drainage | Volume removed, appearance, albumin replacement if > 5 L |
| Thoracentesis | Pleural effusion, empyema evaluation | Volume, appearance, Light criteria, cultures |
| Arthrocentesis | Septic arthritis, crystal arthropathy | Joint, volume, appearance, cell count, crystals, Gram stain, culture |
| Abscess I&D | SSTI abscess, wound drainage | Location, size, anesthesia, purulent drainage volume, packing, culture |
| Skin/tissue biopsy | Rash of unclear etiology, fungal/mycobacterial infection, vasculitis | Site, size, technique (punch vs excision), specimens sent (histology, cultures) |
| Bronchoscopy with BAL | Pneumonia in immunocompromised, PJP, TB, invasive fungal | Lobe sampled, volume instilled/recovered, cell differential, stains and cultures sent |
| Bone biopsy | Osteomyelitis culture and histology | Site, imaging guidance, specimens (culture, histology, AFB, fungal) |
Lumbar Puncture in Detail
Performed at L3-L4 or L4-L5 interspace (below the conus medullaris). Position lateral decubitus or seated; the lateral position is preferred for measuring an accurate opening pressure. Collect four tubes typically: tube 1 for cell count and differential, tube 2 for protein and glucose, tube 3 for Gram stain and culture (with targeted studies like HSV PCR, cryptococcal antigen, VDRL, AFB as indicated), tube 4 for repeat cell count (to differentiate traumatic tap from true bleeding). Document opening pressure (normal 6–25 cm H2O in adults), appearance (clear, turbid, bloody, xanthochromic), any complications, and post-procedure neurologic status.
Blood Culture Technique
Two sets (each set = 1 aerobic + 1 anaerobic bottle, 10 mL each) from two separate venipunctures. Never draw both sets from the same site or from an existing line without also drawing a peripheral set. Contamination is suggested when only one of multiple bottles grows skin flora (coag-negative staph, Cutibacterium, Bacillus, Corynebacterium). Document the draw site and time for each set — the differential time to positivity matters for line infection workups.
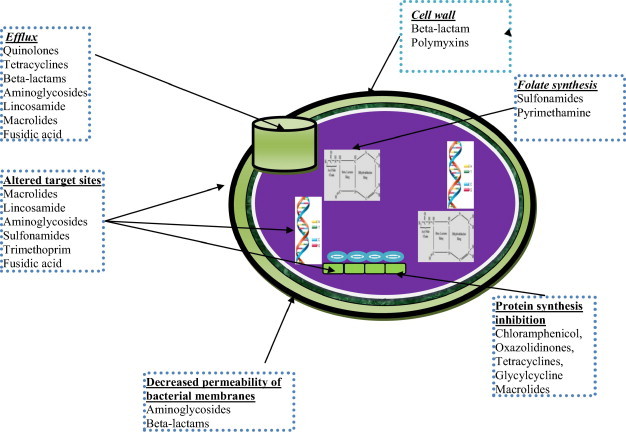
22 Antimicrobial Stewardship & Medications Drugs
You will chart dozens of antimicrobials every day. Organize them by class and know the generic, brand, mechanism, primary use, and red-flag notes.
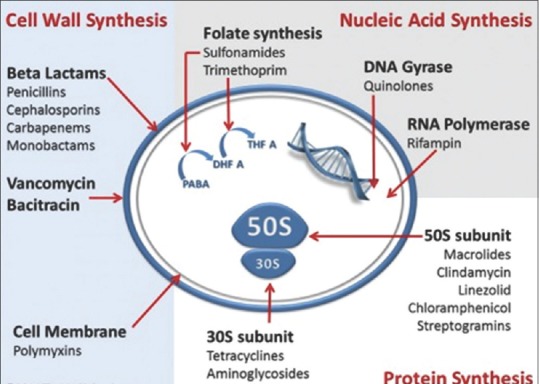
| Class | Examples (generic / brand) | Key Coverage | Critical Notes |
|---|---|---|---|
| Penicillins | Penicillin G, ampicillin, amoxicillin, nafcillin, oxacillin, dicloxacillin | Strep, Listeria (amp), MSSA (naf/ox/diclox), oral anaerobes | Allergy history essential; nafcillin = MSSA bacteremia/endocarditis workhorse |
| β-lactam/β-lactamase inhibitors | Amoxicillin-clavulanate (Augmentin), ampicillin-sulbactam (Unasyn), piperacillin-tazobactam (Zosyn) | Broad gram-positive, gram-negative, anaerobic; Zosyn adds Pseudomonas | Zosyn is the workhorse for hospital-acquired infection empiric coverage |
| Cephalosporins 1st gen | Cefazolin (Ancef), cephalexin (Keflex) | MSSA, strep, some gram-negatives | Surgical prophylaxis standard; MSSA bacteremia alternative to nafcillin |
| Cephalosporins 2nd gen | Cefuroxime, cefoxitin, cefotetan | Extended gram-negative; cefoxitin/cefotetan cover anaerobes | Obstetric and intra-abdominal prophylaxis |
| Cephalosporins 3rd gen | Ceftriaxone (Rocephin), cefotaxime, ceftazidime | Broad gram-negative; ceftazidime covers Pseudomonas | Ceftriaxone = meningitis, CAP, pyelo, gonorrhea workhorse |
| Cephalosporins 4th/5th gen | Cefepime (Maxipime), ceftaroline (Teflaro) | Cefepime adds Pseudomonas; ceftaroline is the only MRSA-active cephalosporin | Cefepime: neurotoxicity in CKD; ceftaroline for MRSA pneumonia/bacteremia |
| Carbapenems | Meropenem, imipenem, ertapenem, doripenem | Very broad including ESBL; meropenem covers Pseudomonas (ertapenem does not) | Reserve for MDR organisms, severe intra-abdominal and neutropenic infection |
| Monobactams | Aztreonam | Gram-negative only (including Pseudomonas) | Safe in severe beta-lactam allergy |
| Fluoroquinolones | Ciprofloxacin, levofloxacin, moxifloxacin | Gram-negatives, atypicals; respiratory FQs for CAP | Black box: tendinopathy, QTc, aortic dissection, C. diff, dysglycemia |
| Macrolides | Azithromycin, clarithromycin, erythromycin | Atypicals, strep, MAC, pertussis | QTc prolongation; azithromycin = CAP, STI, MAC |
| Glycopeptides | Vancomycin, telavancin, dalbavancin, oritavancin | MRSA, CoNS, ampicillin-resistant enterococci | Vanco: AUC monitoring preferred; nephrotoxicity, red man syndrome |
| Aminoglycosides | Gentamicin, tobramycin, amikacin | Aerobic gram-negatives, synergy for endocarditis | Nephrotoxicity and ototoxicity; trough monitoring |
| Oxazolidinones | Linezolid (Zyvox), tedizolid | MRSA, VRE | Thrombocytopenia, serotonin syndrome with SSRIs, peripheral/optic neuropathy |
| Lipopeptides | Daptomycin (Cubicin) | MRSA, VRE, bacteremia, RHS endocarditis | Inactivated by surfactant — do NOT use for pneumonia; monitor CK |
| Tetracyclines | Doxycycline, minocycline, tigecycline, omadacycline | Atypicals, Lyme, RMSF, CA-MRSA, MDR gram-negatives | Doxycycline = tick-borne empiric standard |
| Sulfonamides | TMP-SMX (Bactrim) | CA-MRSA, UTI, PJP, nocardia, toxoplasmosis | Hyperkalemia, rash, AKI, hematologic toxicity |
| Metronidazole | Flagyl | Anaerobes, C. diff, Giardia, trichomonas, amebiasis | Disulfiram reaction with alcohol |
| Antivirals | Acyclovir, valacyclovir, ganciclovir, valganciclovir, oseltamivir, baloxavir, nirmatrelvir-ritonavir, remdesivir | HSV/VZV; CMV; influenza; SARS-CoV-2 | Acyclovir nephrotoxicity; ganciclovir myelosuppression; Paxlovid drug interactions |
| Antifungals — azoles | Fluconazole, itraconazole, voriconazole, posaconazole, isavuconazole | Candida, endemic mycoses, aspergillus (vori/isa/posa) | Many drug interactions; voriconazole visual changes and hepatotoxicity |
| Antifungals — echinocandins | Caspofungin, micafungin, anidulafungin | Candidemia first-line | Well tolerated; no CSF penetration |
| Antifungals — polyenes | Amphotericin B (deoxycholate, lipid complex, liposomal) | Severe invasive mycoses, cryptococcal meningitis induction | Infusion reactions, nephrotoxicity, electrolyte wasting |
| Antiretrovirals | Bictegravir, dolutegravir, raltegravir (INSTIs); emtricitabine, tenofovir (NRTIs); efavirenz, rilpivirine (NNRTIs); darunavir (PI) | HIV-1 | First-line regimens are INSTI-based; check renal function and HBV serology before tenofovir |
Start broad when the patient is sick; de-escalate as soon as cultures return; choose the narrowest effective agent; use the shortest evidence-based duration; document the indication, day of therapy, and planned stop date on every antibiotic, every day. Stewardship teams review orders for these elements — a note that captures them cleanly makes rounds faster.
23 Labs, Cultures & Imaging Data
ID notes are data-dense. Learn the normal ranges and how to interpret trends.
Inflammatory Markers
CBC with differential: leukocytosis with left shift or bandemia supports bacterial infection; leukopenia or neutropenia is ominous. CRP rises within 6–8 hours of inflammation and falls with effective therapy. Procalcitonin is more specific for bacterial infection and is used to guide antibiotic de-escalation in respiratory infection and sepsis. ESR is slow to rise and fall; useful for osteomyelitis and endocarditis monitoring. Lactate reflects tissue hypoperfusion in sepsis.
Cultures
Report structure includes the specimen source, Gram stain result, preliminary growth, final identification, and susceptibility testing (MIC values by antibiotic with S/I/R interpretation). Document organism, source, and resistance pattern on every note.
Serology & Molecular
IgM indicates recent infection; IgG indicates past exposure or chronic infection. Paired acute and convalescent titers can diagnose infections for which direct testing is difficult. PCR (qualitative and quantitative) is standard for HIV, HCV, HBV, CMV, EBV, BK virus, HSV, respiratory viral panels, and multiplex GI panels. Antigen tests: urinary legionella and pneumococcal antigen, histoplasma urine antigen, cryptococcal antigen, galactomannan, beta-D-glucan.
Imaging
Chest X-ray and CT for pulmonary infection. CT abdomen/pelvis with contrast for intra-abdominal source. MRI for osteomyelitis, brain abscess, spinal epidural abscess, and septic joint evaluation. Echocardiogram (TTE then TEE) for endocarditis. Tagged WBC scan and PET-CT are occasionally used for occult infection.
24 Classification Systems Reference
Infection: Suspected or documented infection.
Sepsis: Life-threatening organ dysfunction (SOFA rise ≥ 2) due to a dysregulated host response to infection.
Septic shock: Sepsis + vasopressor requirement to maintain MAP ≥ 65 + lactate > 2 despite adequate volume resuscitation.
Points assigned for temperature, blood pressure, mechanical ventilation, cardiac arrest, and mental status. A score ≥ 4 predicts high mortality in gram-negative bacteremia.
CRP, WBC, hemoglobin, sodium, creatinine, glucose. Score ≥ 6 suggests nec fasc; ≥ 8 is high probability. Never delays surgery on clinical suspicion.
Stage 0 (early HIV, < 180 days from negative test) · Stage 1 (CD4 ≥ 500 or ≥ 26%) · Stage 2 (CD4 200–499 or 14–25%) · Stage 3 (CD4 < 200 or < 14% or AIDS-defining illness) · Stage U (unknown).
0 — outpatient, low risk; 1 — outpatient, low risk; 2 — consider short admission; 3 — inpatient, moderate risk; 4–5 — ICU consideration, high mortality.
25 Physical Exam in ID Exam
The ID exam is a head-to-toe search for an occult source.
General: Ill-appearing vs well; rigors; diaphoresis; cachexia.
Vitals: Tmax, Tcurrent, heart rate, respiratory rate, blood pressure, MAP, oxygen saturation, FiO2, pressor doses.
HEENT: Oral thrush, dental abscess, pharyngeal exudate, sinus tenderness, cervical and supraclavicular lymphadenopathy, meningismus, fundoscopy for Roth spots or endophthalmitis.
Cardiac: New or changed murmur (endocarditis), pericardial rub, peripheral stigmata (Janeway lesions, Osler nodes, splinter hemorrhages, conjunctival petechiae).
Pulmonary: Crackles, rhonchi, egophony, dullness to percussion, bronchial breath sounds.
Abdomen: Tenderness, guarding, rebound, hepatosplenomegaly, Murphy sign, CVA tenderness.
GU/Pelvis/Rectal: Prostate tenderness, perirectal abscess, genital ulcers, vaginal discharge, cervical motion tenderness.
Extremities: Line sites, joint swelling, IVDU tracks, peripheral pulses, skin breakdown, diabetic foot exam with probe-to-bone.
Skin: Rash character (macular, papular, petechial, vesicular, pustular), distribution, mucosal involvement, desquamation, purpura, ecthyma gangrenosum, targetoid lesions.
Neurologic: GCS, orientation, meningeal signs (Kernig, Brudzinski), focal deficits, cranial nerves, asterixis.
Document the skin exam with the same precision a dermatologist uses. "Diffuse rash" is useless. "Blanching erythematous macular rash on trunk sparing palms and soles without mucosal involvement" is what the attending needs.
26 Abbreviations Master List Reference
Syndromes & Diagnoses
Pathogens & Resistance
Labs, Procedures & Meds
27 Sample HPI Templates Templates
These templates reflect the density, structure, and tone of real ID consult notes. Use them as scaffolding during your first weeks.
"Mr. [Name] is a 68-year-old male with a history of type 2 diabetes (A1C 9.1), CKD stage 3, and BPH, admitted from the ED with fever, confusion, and hypotension concerning for urosepsis. Symptoms began 48 hours ago with dysuria, flank pain, and rigors. He was brought in by family after altered mental status developed this morning. ED vitals: T 39.2, HR 122, BP 82/48 (MAP 59), RR 24, SpO2 94% on 2 L. Lactate 4.2, WBC 18.4 with 14% bands, Cr 2.3 (baseline 1.4), UA with > 100 WBC and nitrite positive. Two sets of blood cultures and urine culture were drawn at 0815; ceftriaxone 2 g IV was administered at 0840; he received 30 mL/kg crystalloid with MAP improving to 68. Currently on norepinephrine 0.05 mcg/kg/min. No prior MDR organisms; no recent antibiotics; no indwelling hardware."
"Ms. [Name] is a 42-year-old female with obesity and poorly controlled type 2 diabetes (A1C 10.3) presenting with 4 days of progressive right lower extremity erythema, warmth, and pain. She denies trauma but does report a small blister on the dorsum of her right foot one week ago. She has had low-grade fevers (Tmax 38.4) and chills for 48 hours. On exam, the right lower extremity demonstrates confluent non-purulent erythema from the dorsum of the foot extending to the mid-calf with ill-defined borders (marked at 0930 today), warmth, and 2+ pitting edema. No crepitus, no fluctuance, no skin necrosis, no drainage. Distal pulses intact. She has never had MRSA previously. No recent hospitalizations or antibiotics."
"Ms. [Name] is a 27-year-old otherwise healthy female presenting with 3 days of dysuria, urinary frequency, and suprapubic pain progressing over the last 24 hours to right-sided flank pain, fever to 38.9, nausea, and one episode of non-bloody vomiting. She denies vaginal discharge, recent antibiotic use, pregnancy, or prior UTIs requiring hospitalization. She is sexually active with a single male partner and uses barrier contraception. On exam, she has right CVA tenderness and suprapubic tenderness without peritoneal signs. UA demonstrates > 100 WBC, large leukocyte esterase, and positive nitrites. Urine culture pending. CBC with WBC 14.2 and neutrophil predominance; BMP unremarkable. She received ceftriaxone 1 g IV in the ED at 1120."
"Mr. [Name] is a 38-year-old male with HIV-1 (diagnosed 2018, CDC Stage 2, nadir CD4 320) presenting for routine 3-month follow-up. He has been on bictegravir/emtricitabine/tenofovir alafenamide (Biktarvy) one tablet daily since 2019 with excellent tolerability and adherence. Most recent labs (January 2026): CD4 680 (35%), HIV VL < 20 copies/mL (undetectable for 18 consecutive months). He denies new symptoms, opportunistic infection symptoms, medication side effects, or missed doses. Immunizations are up to date including HPV, hepatitis A and B, pneumococcal, meningococcal, and annual influenza. He declines Tdap booster today (received 2 years ago). STI screen at last visit was negative. Up-to-date on colorectal and anal cancer screening."
"Ms. [Name] is a 64-year-old female POD #5 from open sigmoid colectomy for perforated diverticulitis with primary anastomosis and diverting loop ileostomy, with ID consulted for new fever and leukocytosis. Post-operative course was initially uncomplicated; she received piperacillin-tazobactam for 4 days postoperatively with planned stop today. Last evening she developed Tmax 38.8 with rigors, WBC rose from 9.8 to 18.2, and she reports new worsening left lower quadrant pain. Exam reveals new LLQ tenderness with voluntary guarding, clean dry wound, functioning ileostomy. CT abdomen/pelvis with contrast obtained this morning shows a 5.3 × 4.8 cm rim-enhancing fluid collection in the pelvis concerning for deep organ space SSI. Blood cultures drawn at 0400 are pending. IR has been consulted for percutaneous drainage. Question: antibiotic management and duration."
"Mr. [Name] is a 29-year-old male presenting with 5 days of fever (Tmax 40.1), rigors, frontal headache, myalgias, and one episode of non-bloody diarrhea. He returned 12 days ago from a 3-week trip to rural western Kenya where he participated in a volunteer construction project. He slept in open-air accommodations without bed nets. He took atovaquone-proguanil for malaria prophylaxis but admits to missing several doses toward the end of the trip and stopped entirely upon return. He received pre-travel hepatitis A, typhoid, and yellow fever vaccines. He denies freshwater swimming, sexual exposures, animal bites, or tick attachments. On exam: ill-appearing, T 39.6, HR 118, BP 108/66; no rash, no eschar, no lymphadenopathy, mild hepatosplenomegaly. Initial labs: platelets 78K, hemoglobin 11.2, LDH elevated, indirect bilirubin 2.1. Thick and thin blood smears ordered STAT; rapid malaria antigen pending."
ID is the most narrative specialty in internal medicine. Every note tells a story: this patient, this exposure, this organism, this source, this drug, for this duration. The best ID scribes listen for the story and help the attending tell it cleanly. When the fellow says "gram-negative rods from two bottles, time to positivity 8 hours, source likely urinary given pyuria and hydronephrosis," a great scribe has already mentally built the assessment: community-acquired complicated UTI with gram-negative bacteremia, empiric ceftriaxone, awaiting susceptibilities, urology consulted for obstruction.
Memorize the common bugs at common sites. Learn to pronounce every drug the first time you hear it. Track every antibiotic day. Read the culture and susceptibility reports before rounds. Recognize when sepsis is brewing and when a note needs to move faster. Document exposures in detail — the HPI is often the single most important diagnostic tool in ID.
Welcome to infectious disease. The differentials are wide, the stakes are high, and the pattern recognition you build in your first months will serve you for the rest of your medical career.
28 References & Sources References
Clinical Practice Guidelines
Screening Recommendations
Diagram & Figure Sources
Figure 1: Streptococcus pneumoniae Gram stain. CDC Public Health Image Library. Public domain.
Figure 2: MRSA scanning electron micrograph. NIAID/NIH. Public domain.
Figure 3: Mycobacterium tuberculosis Ziehl-Neelsen stain. CDC. Public domain.
Figure 4: Anatomy of the Lymphatic System. OpenStax College. CC BY 3.0.
Figure 5: Immune System Cell Migration. OpenStax College. CC BY 3.0.
Figure 6: HIV budding from lymphocyte. CDC/C. Goldsmith. Public domain.
Figure 7: Plasmodium falciparum blood smear. CDC Public Health Image Library. Public domain.