Maternal-Fetal Medicine
Every high-risk obstetric diagnosis, procedure, medication, classification, and documentation framework a scribe needs in the MFM clinic, antepartum unit, and labor floor.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Maternal Physiology & Pregnancy Essentials
Maternal-fetal medicine (MFM), also called perinatology, is the obstetric subspecialty focused on high-risk pregnancy. MFM physicians manage pregnancies complicated by maternal medical disease (diabetes, hypertension, lupus, cardiac disease), obstetric complications (preeclampsia, preterm labor, placental disorders), fetal disorders (growth restriction, anomalies, isoimmunization), and multiple gestations. They also perform invasive fetal diagnostic and therapeutic procedures. Scribes in MFM clinics encounter a mix of consult visits, ultrasound-integrated encounters, antepartum testing visits, and inpatient antepartum/L&D documentation.
Gestational Dating & Trimesters
Every MFM note begins with gestational age, expressed as weeks + days (e.g., "32w4d"). Gestational age (GA) is calculated from the last menstrual period (LMP) and confirmed or revised by first-trimester ultrasound (most accurate dating). The estimated due date (EDD) is 40 weeks from LMP. Pregnancy is divided into the first trimester (0–13w6d), second trimester (14w0d–27w6d), and third trimester (28w0d–delivery). Term is 37–41w6d; early term is 37w0d–38w6d, full term is 39w0d–40w6d, late term is 41w0d–41w6d, post-term is ≥42w0d. Preterm is <37w0d (subdivided into late preterm 34–36w6d, moderate 32–33w6d, very preterm 28–31w6d, and extremely preterm <28w0d).
Gravidity & Parity (GTPAL)
Every obstetric note uses a GTPAL (or GPA) summary. G = gravidity (total pregnancies including current). T = term deliveries (≥37w). P = preterm deliveries (20–36w6d). A = abortions (spontaneous or elective losses <20w, plus ectopics). L = living children. A G3P1011 woman has had 3 pregnancies, 1 term delivery, 0 preterm, 1 loss, and 1 living child.
Maternal Physiologic Changes
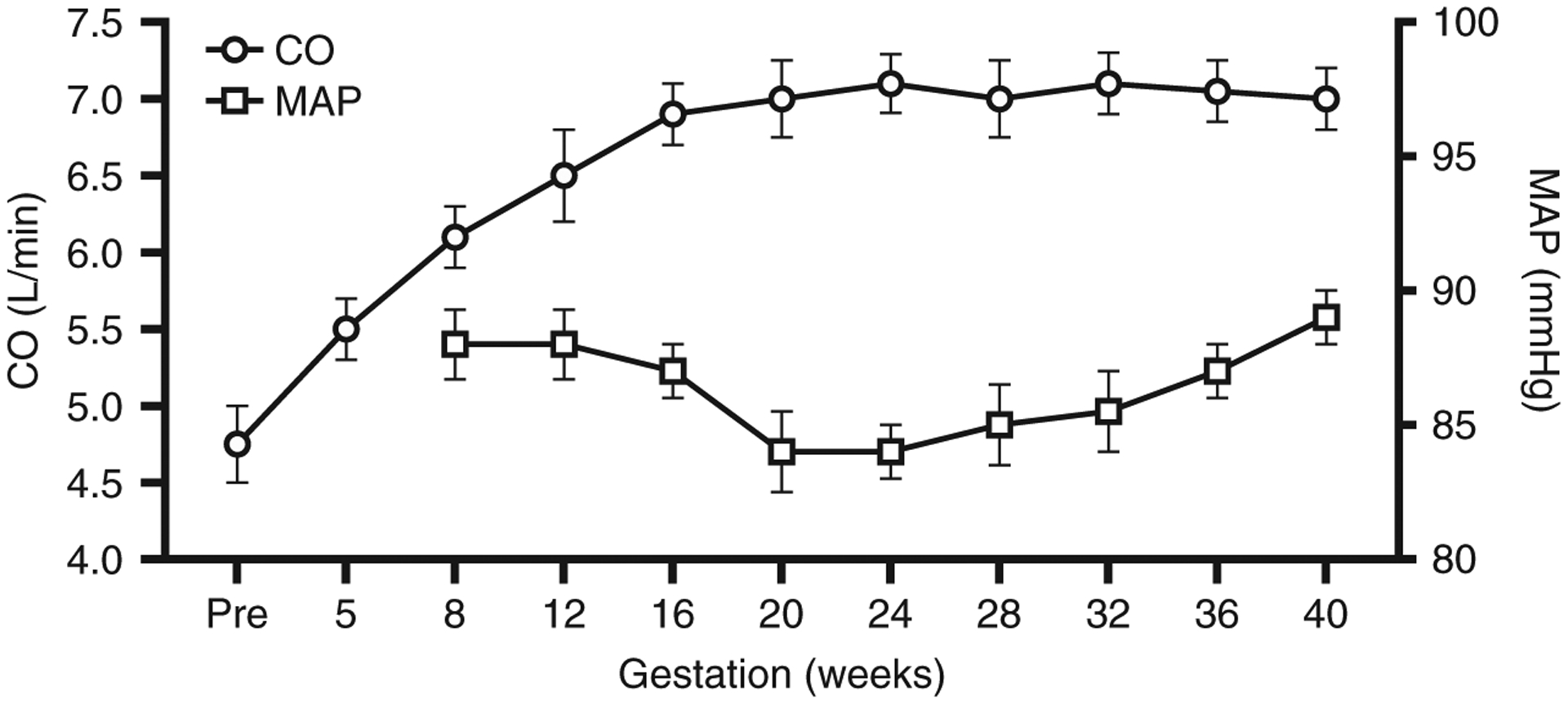
Pregnancy produces predictable physiologic changes the scribe will see quoted in notes and vitals. Cardiovascular: plasma volume increases 40–50%, cardiac output rises 30–50%, heart rate increases 10–20 bpm, systemic vascular resistance falls (BP drops in mid-pregnancy, returns to baseline by term). Hematologic: dilutional anemia (Hgb nadir ~10.5 g/dL in the second trimester); procoagulant shift with rising fibrinogen, factors VII, VIII, X, and von Willebrand factor, making pregnancy and the puerperium a high VTE-risk state. Respiratory: tidal volume and minute ventilation rise; compensated respiratory alkalosis is normal (pCO2 ~30 mm Hg). Renal: GFR rises ~50%, serum creatinine falls (normal pregnancy Cr <0.8 mg/dL; ≥1.0 is abnormal). Endocrine: insulin resistance rises progressively due to placental hormones (hPL, cortisol, progesterone), driving gestational diabetes. Thyroid-binding globulin rises; total T4 rises but free T4 remains normal.
Never confuse LMP dating with ultrasound dating. Once an EDD is "established" (usually by first-trimester CRL), it is locked in — subsequent ultrasounds do not change it. The MFM attending will correct you if you re-date a patient based on a third-trimester scan.
02 Scribe Documentation Framework in MFM
The MFM Consultation Note
MFM encounters are usually consultations from a general OB or primary care provider. The note structure is: reason for consult, pregnancy history (GTPAL, EDD, dating method), current pregnancy course, PMH/PSH, medications, allergies, family history, social history, ROS, vitals, fundal height, fetal heart tones (FHT), ultrasound findings, assessment, and plan. The assessment frames the problem in terms of both maternal risk and fetal risk, and the plan is typically broken into maternal surveillance, fetal surveillance, delivery planning, and patient counseling.
- Gestational age and EDD with dating method (LMP vs first-trimester US vs IVF)
- GTPAL and pertinent obstetric history (prior preterm birth, preeclampsia, stillbirth, cesarean)
- Current pregnancy course: labs (prenatal panel, aneuploidy screen, anatomy scan), complications, hospitalizations
- Reason for MFM referral: the specific high-risk feature
- Fetal movement, contractions, leakage of fluid, vaginal bleeding (every visit ROS)
- Maternal medical conditions and medication list with pregnancy-category considerations
- Social history: tobacco, alcohol, substance use, IPV screening, work, support
The Antepartum Testing (AFT) Visit
Many MFM visits are "AFT" visits dedicated to fetal surveillance. These are short, focused notes documenting nonstress test (NST) results, biophysical profile (BPP) findings, amniotic fluid assessment, fetal growth (if interval), and Doppler velocimetry when indicated. The note closes with a reactive/nonreactive or reassuring/nonreassuring determination and a plan for timing of delivery.
On every antepartum note you must document fetal movement ("FM present"), contractions ("no CTX"), leakage of fluid ("no LOF"), vaginal bleeding ("no VB"), and headache/visual changes/RUQ pain (preeclampsia symptoms). If any of these is positive, the note's plan changes dramatically. These five questions are asked every single visit.
03 Chronic Medical Conditions in Pregnancy
A large portion of MFM consults are for pregestational (chronic) medical disease. Pregnancy changes the pharmacokinetics, disease activity, and fetal risk profile of nearly every chronic condition.
Pregestational Diabetes (Type 1 & Type 2)
Pregestational DM carries markedly elevated risks of congenital anomalies (cardiac, neural tube, caudal regression), miscarriage, stillbirth, macrosomia, shoulder dystocia, neonatal hypoglycemia, and preeclampsia. Glycemic control before conception is the single strongest determinant of outcome; a first-visit HbA1c <6.5% is the target. Management centers on insulin (oral agents are second-line; metformin and glyburide cross the placenta). Goals: fasting <95, 1-hour postprandial <140, 2-hour <120 mg/dL. Delivery is typically at 39w0d for well-controlled patients and earlier (36–38w) for poorly controlled or vasculopathy. ACOG Practice Bulletin 201 (Pregestational Diabetes) is the standard reference.
Chronic Hypertension
Defined as BP ≥140/90 before pregnancy or before 20w. Chronic HTN doubles the risk of superimposed preeclampsia, abruption, FGR, and preterm birth. The CHAP trial established that treating BP to <140/90 in pregnancy reduces adverse perinatal outcomes without compromising fetal growth. First-line agents: labetalol, nifedipine ER, methyldopa. ACE inhibitors and ARBs are contraindicated (fetal renal agenesis, oligohydramnios).
Lupus & Antiphospholipid Syndrome (APS)
SLE increases risks of preeclampsia, FGR, preterm birth, stillbirth, and neonatal lupus (anti-Ro/SSA antibodies cause congenital heart block). Pregnancy should be planned during a ≥6-month quiescent period. Hydroxychloroquine is continued throughout. APS (positive lupus anticoagulant, anti-cardiolipin, or anti-β2 glycoprotein antibodies with clinical criteria) is treated with prophylactic heparin/LMWH plus low-dose aspirin. Secondary APS with prior thrombosis requires therapeutic anticoagulation.
Other Chronic Conditions
- RA/IBD: Often improve in pregnancy. Continue biologics through the second trimester (certolizumab crosses placenta minimally and can continue throughout).
- Asthma: Poor control is far riskier than medications. Continue ICS/LABA; albuterol as needed. Oral steroids are safe when needed.
- Epilepsy: Keep the fewest AEDs at lowest effective dose. Avoid valproate (neural tube defects, neurodevelopmental effects). Folic acid 4 mg daily. Levetiracetam and lamotrigine are first-line in pregnancy.
- Thyroid disease: Hypothyroidism — increase levothyroxine ~30% at pregnancy confirmation; TSH goal <2.5 first trimester. Hyperthyroidism — PTU in the first trimester, methimazole after.
- Cardiac disease: mWHO classification guides risk. Classes III–IV (severe MS, pulmonary HTN, Marfan with aortic dilation, severely reduced LVEF, Eisenmenger) carry high maternal mortality. Peripartum cardiomyopathy is a separate entity (see Section 17).
- CKD: Higher baseline Cr and proteinuria → higher preeclampsia and FGR risk. Cr >1.5 mg/dL pre-pregnancy is a marker of especially high risk.
- Sickle cell disease: Increased VOC, acute chest, VTE, preeclampsia, FGR. Simple transfusion for Hgb <8 or symptomatic; exchange for acute chest.
04 Gestational Diabetes Mellitus (GDM)
GDM is glucose intolerance first recognized in pregnancy, driven by placental insulin resistance. It affects roughly 6–10% of US pregnancies. Untreated GDM causes macrosomia, shoulder dystocia, birth trauma, neonatal hypoglycemia, polycythemia, hyperbilirubinemia, and an elevated lifetime risk of type 2 DM for the mother.
Screening & Diagnosis
Universal screening at 24–28 weeks using the two-step approach (preferred in the US): a 50-g 1-hour glucose challenge test (GCT) with a threshold of 130, 135, or 140 mg/dL, followed by a diagnostic 100-g 3-hour oral glucose tolerance test (OGTT) using Carpenter-Coustan criteria if screen-positive. Two or more abnormal values on the 3-hour OGTT confirm GDM.
| Timepoint | Threshold (mg/dL) |
|---|---|
| Fasting | ≥95 |
| 1 hour | ≥180 |
| 2 hours | ≥155 |
| 3 hours | ≥140 |
Two or more abnormal values → GDM. The alternative IADPSG one-step uses a 75-g 2-hour OGTT with thresholds 92 / 180 / 153 mg/dL; one abnormal value is diagnostic.
Classification & Management
White/modified classification: A1GDM — diet-controlled; A2GDM — requires pharmacologic therapy. First-line treatment is insulin (does not cross placenta). Metformin and glyburide are alternatives but cross the placenta. Fetal surveillance is intensified for A2GDM (weekly NSTs or BPPs from 32–34 weeks). Delivery: A1GDM — 39w0d–40w6d; A2GDM well-controlled — 39w0d; A2GDM poorly controlled — 36w0d–39w0d individualized. ACOG Practice Bulletin 190 (Gestational Diabetes) guides thresholds and delivery timing.
05 Hypertensive Disorders of Pregnancy
Hypertensive disorders complicate 10–15% of pregnancies and remain a leading cause of maternal mortality globally. The four categories:
| Category | Criteria |
|---|---|
| Chronic HTN | BP ≥140/90 before 20w or pre-pregnancy |
| Gestational HTN | New BP ≥140/90 after 20w, no proteinuria, no severe features |
| Preeclampsia | BP ≥140/90 after 20w + proteinuria OR end-organ dysfunction |
| Chronic HTN + Superimposed Preeclampsia | Preexisting HTN with new proteinuria or end-organ dysfunction after 20w |
Gestational Hypertension
New-onset BP ≥140/90 on two occasions ≥4 hours apart after 20 weeks, without proteinuria or severe features. Up to half progress to preeclampsia. Treatment: antihypertensives for BP ≥140/90 per CHAP; delivery at 37w0d.
06 Preeclampsia, Eclampsia & HELLP
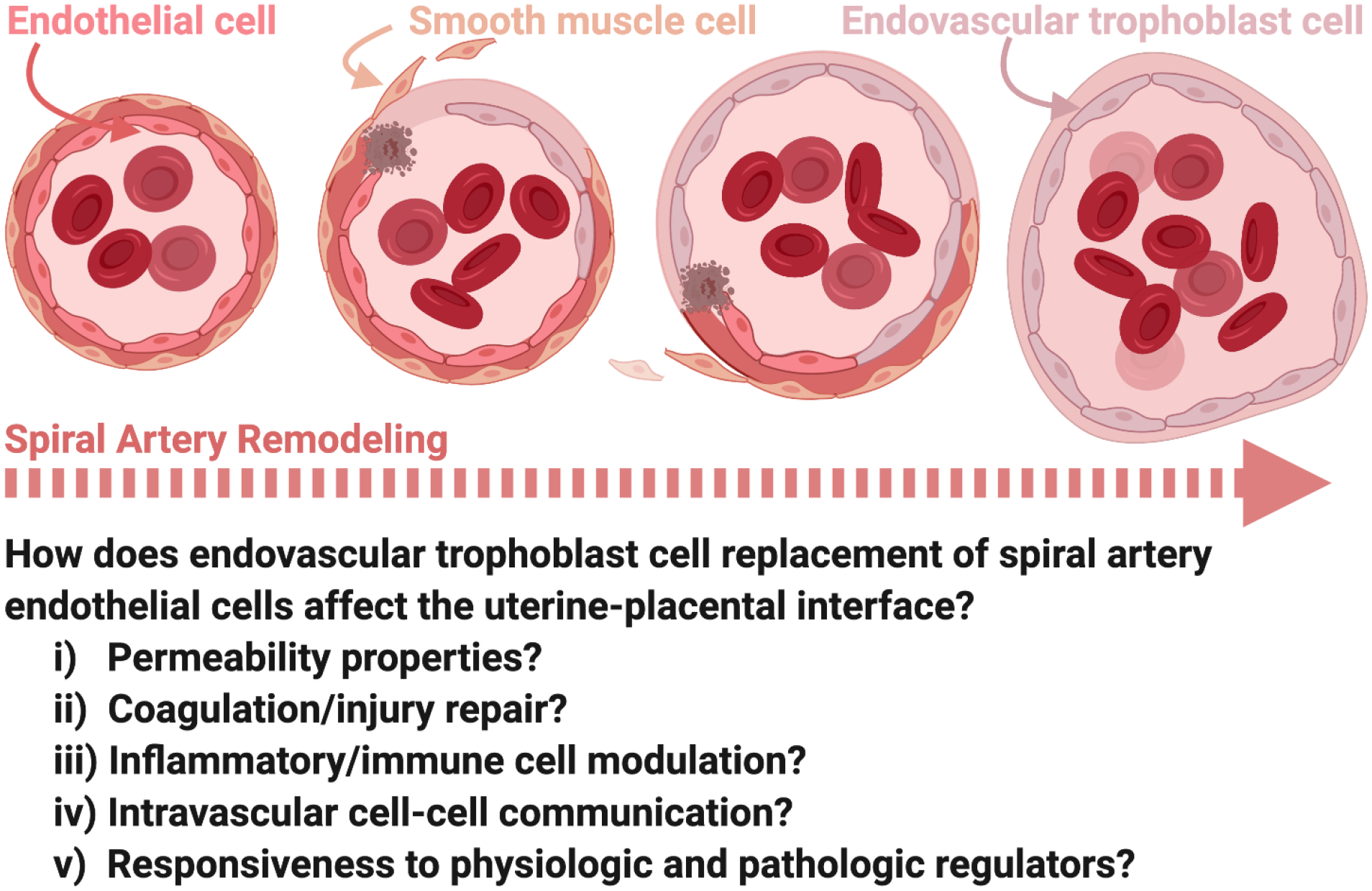
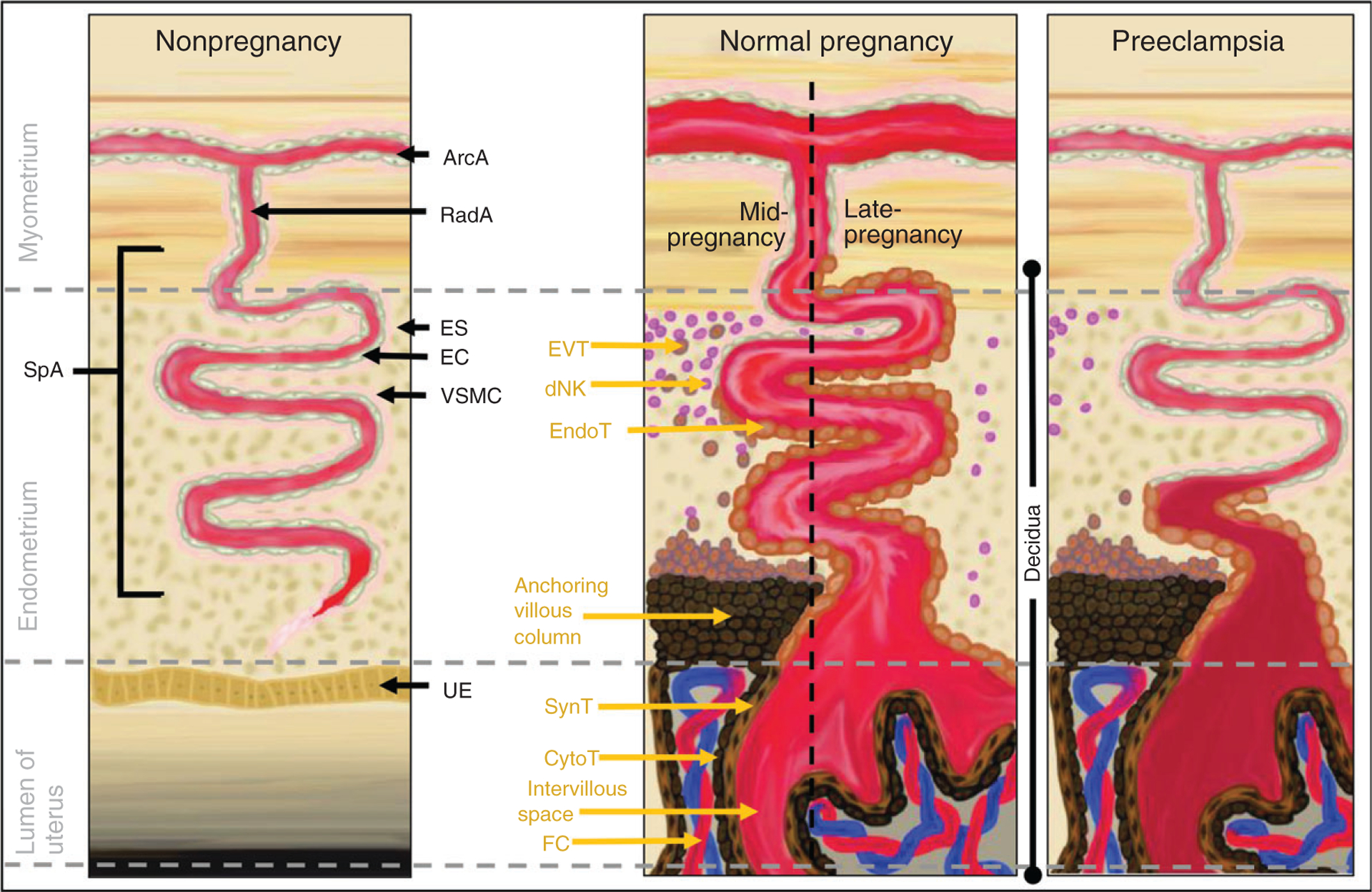
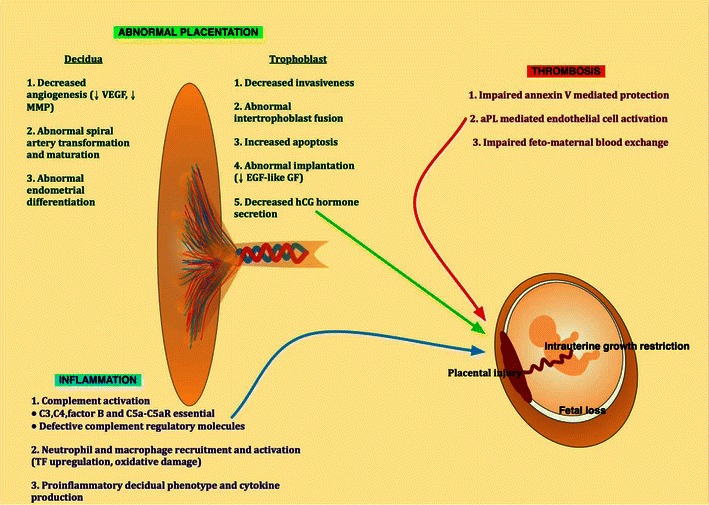
Preeclampsia is a multisystem disorder of pregnancy characterized by new-onset hypertension and end-organ dysfunction after 20 weeks. Pathophysiology centers on abnormal placentation: shallow trophoblastic invasion of the spiral arteries leads to placental hypoperfusion, release of antiangiogenic factors (sFlt-1, soluble endoglin) that antagonize VEGF and PlGF, and systemic endothelial dysfunction. The clinical manifestations — hypertension, proteinuria, hepatic injury, thrombocytopenia, cerebral edema — are all downstream of that endothelial injury.
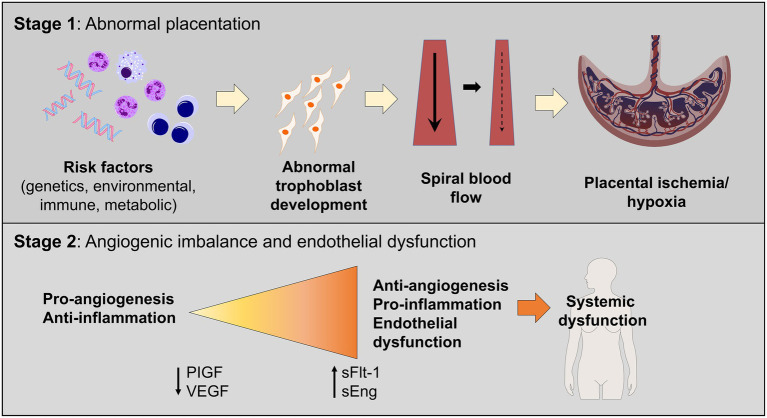
Diagnostic Criteria
BP ≥140/90 on two occasions ≥4 hours apart plus one of: proteinuria (≥300 mg/24h, P/Cr ≥0.3, or dipstick 2+), thrombocytopenia (<100k), serum creatinine >1.1 or doubling, transaminases ≥2× upper limit, pulmonary edema, or new-onset cerebral/visual symptoms.
- BP ≥160/110 on two occasions ≥4 hours apart (or sooner if antihypertensives started)
- Thrombocytopenia <100,000/µL
- Serum creatinine >1.1 mg/dL or doubling without other renal disease
- Transaminases ≥2× upper limit of normal
- Pulmonary edema
- New-onset persistent headache unresponsive to medication and not attributable to alternative cause
- Visual disturbances (scotomata, photopsia, cortical blindness)
- Severe persistent RUQ or epigastric pain
Proteinuria magnitude is not a severe feature. ACOG Gestational Hypertension & Preeclampsia (PB 222).
HELLP Syndrome
A severe variant with Hemolysis, Elevated Liver enzymes, and Low Platelets. Tennessee criteria: platelets <100k, AST ≥70, LDH ≥600, peripheral smear with schistocytes or total bilirubin ≥1.2. Mississippi criteria further grade by platelet nadir (Class 1 <50k, Class 2 50–100k, Class 3 100–150k). HELLP can occur without significant hypertension or proteinuria.
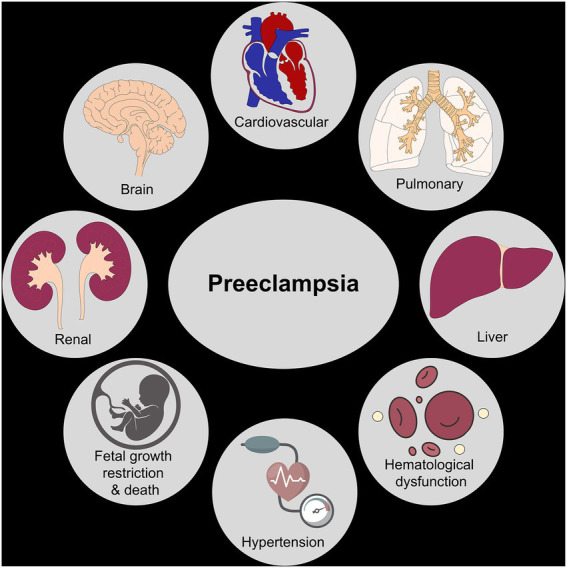
Eclampsia
New-onset generalized tonic-clonic seizure in a patient with preeclampsia (or as the presenting feature). Mortality is high; cerebral edema and stroke are the killers. Treatment: magnesium sulfate 4–6 g IV loading + 1–2 g/hr infusion, airway protection, BP control, and delivery.
Management
Definitive treatment is delivery. Timing depends on severity and gestational age:
- Gestational HTN / mild preeclampsia (no severe features): Deliver at 37w0d.
- Preeclampsia with severe features, stable: Deliver at 34w0d.
- Preeclampsia with severe features, unstable / HELLP / eclampsia / pulmonary edema / uncontrollable BP / nonreassuring fetal status / abruption: Deliver promptly regardless of GA.
- Expectant management <34w only in tertiary centers with maternal/fetal stability.
Seizure prophylaxis with magnesium sulfate for all patients with preeclampsia with severe features and during intrapartum/postpartum (24 hours). Acute severe hypertension (≥160/110 sustained >15 minutes) requires emergent treatment within 30–60 minutes using IV labetalol, IV hydralazine, or PO immediate-release nifedipine. Low-dose aspirin (81–162 mg) starting at 12–16 weeks reduces preeclampsia risk in high-risk patients per USPSTF preeclampsia prevention guidance.
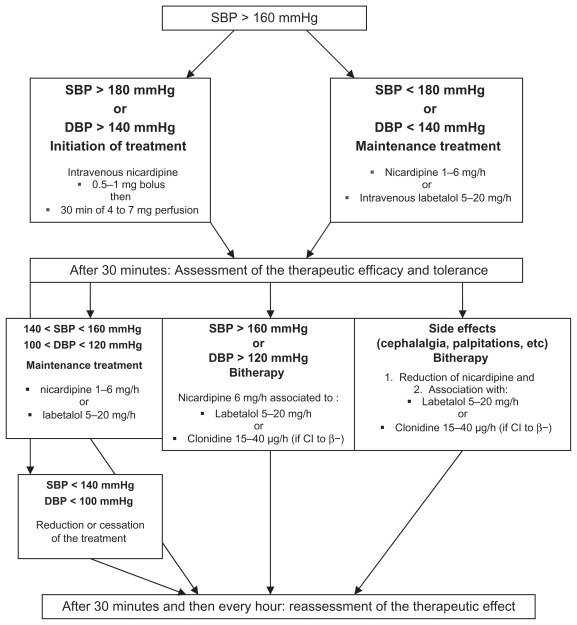
07 Intrahepatic Cholestasis & Hyperemesis Gravidarum
Intrahepatic Cholestasis of Pregnancy (ICP)
A third-trimester hepatic disorder presenting with generalized pruritus (especially palms and soles, worse at night) without a rash. Diagnosis: elevated total serum bile acids (≥10 µmol/L; severe ≥40; very severe ≥100). Transaminases are often mildly elevated. Fetal risks include preterm birth, meconium-stained fluid, and intrauterine fetal demise — risks rise sharply at bile acids ≥100. Treatment: ursodeoxycholic acid (UDCA) 300 mg TID improves pruritus. Delivery timing: 36w0d–37w0d for typical ICP; 34w0d–36w0d for severe (≥100 µmol/L) per SMFM Consult Series 53 on ICP.
Hyperemesis Gravidarum (HG)
Severe nausea and vomiting of pregnancy with weight loss >5% of pre-pregnancy weight, dehydration, and ketosis. Differential includes thyroid storm (beta-hCG cross-stimulates TSH receptor), molar pregnancy, and multiple gestation. Treatment ladder: vitamin B6 (pyridoxine) ± doxylamine → dimenhydrinate/diphenhydramine → metoclopramide/promethazine → ondansetron → IV fluids, thiamine (before dextrose to prevent Wernicke), and methylprednisolone for refractory cases.
08 Placental Disorders
Placenta Previa
The placenta covers or approaches the internal cervical os. Classic presentation: painless, bright-red vaginal bleeding in the second or third trimester. No digital exam — transvaginal ultrasound is the diagnostic modality. Many previas diagnosed at anatomy scan resolve by term due to lower uterine segment development; follow-up scan at 32w. Delivery: scheduled cesarean at 36w0d–37w6d for persistent previa.
Placental Abruption
Premature separation of the normally implanted placenta. Classic triad: abdominal pain, vaginal bleeding (may be concealed), and a tense, tender uterus. Risk factors: hypertension, prior abruption, trauma, cocaine use, PPROM, smoking, advanced maternal age. Ultrasound is insensitive — diagnosis is clinical. Complications: maternal hemorrhage, DIC, fetal distress, stillbirth. Management depends on severity and fetal status: expectant with close monitoring (stable, remote from term) or emergent delivery (unstable or nonreassuring fetal status).
Placenta Accreta Spectrum (PAS)
Abnormal trophoblastic invasion into the uterine wall. Three severities:
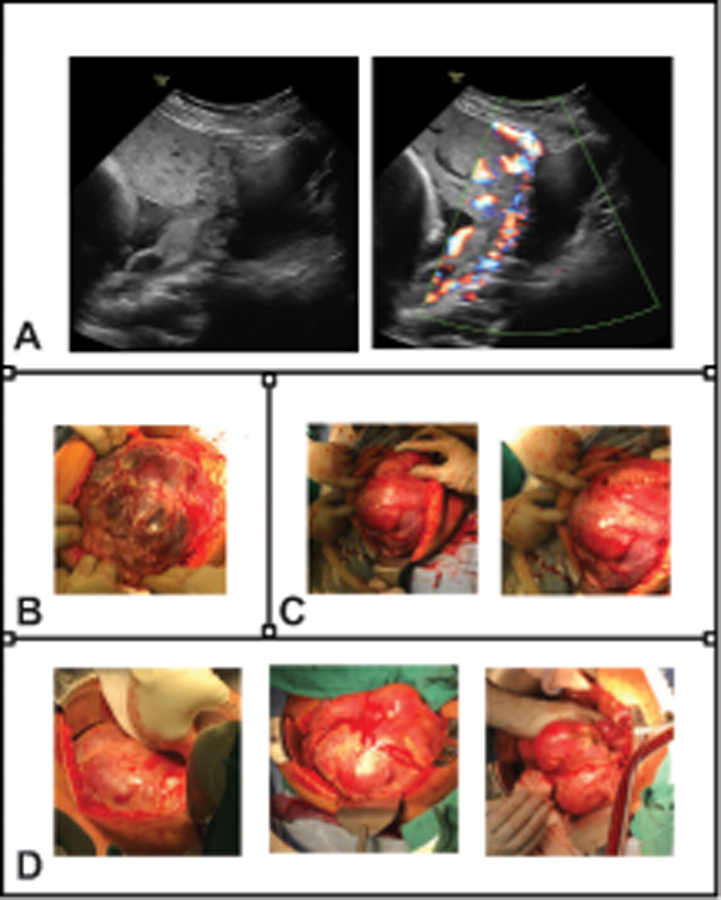
| Grade | Depth |
|---|---|
| Accreta | Placenta attaches directly to myometrium (no decidua) |
| Increta | Invasion into myometrium |
| Percreta | Invasion through myometrium, possibly into serosa or adjacent organs (bladder) |
Primary risk factors: prior cesarean delivery (risk rises with number of prior CDs) + placenta previa. Scheduled delivery at 34w0d–35w6d in a referral center with multidisciplinary team, cesarean hysterectomy planned, blood products available. ACOG/SMFM Placenta Accreta Spectrum Obstetric Care Consensus.
Vasa Previa
Fetal vessels (unprotected by cord or placenta) traverse the membranes over the cervical os. Rupture at ROM causes rapid fetal exsanguination. Screening transvaginal US with color Doppler in at-risk patients (velamentous cord, succenturiate lobe, low-lying placenta in second trimester). Scheduled cesarean at 34w0d–37w0d before ROM.
09 VTE in Pregnancy
Pregnancy is a hypercoagulable state; VTE risk is 4–5× baseline and highest in the postpartum period. PE remains a leading cause of maternal mortality. DVT in pregnancy preferentially involves the left iliofemoral veins (right iliac artery compresses the left iliac vein).
Diagnosis
Suspected DVT: compression ultrasound. Suspected PE: start with CXR; then CT-PA (preferred in most patients) or V/Q scan (lower maternal breast radiation). D-dimer is unreliable in pregnancy because it rises physiologically. Treatment: LMWH (enoxaparin) is first-line — it does not cross the placenta. Unfractionated heparin for patients with renal failure or when rapid reversibility is needed. Warfarin is contraindicated (teratogenic — warfarin embryopathy) except in mechanical heart valves. DOACs are not used in pregnancy.
Prophylactic dosing (enoxaparin 40 mg SC daily) for patients with prior VTE or high-risk thrombophilia; therapeutic dosing (1 mg/kg BID) for acute VTE. LMWH is held 24 hours before planned delivery/neuraxial anesthesia. Anticoagulation is continued throughout pregnancy and at least 6 weeks postpartum per ACOG Practice Bulletin 196 (Thromboembolism).
10 Fetal Growth Restriction & LGA
Fetal Growth Restriction (FGR / IUGR)
Defined as estimated fetal weight (EFW) or abdominal circumference (AC) <10th percentile for gestational age. Severe FGR is <3rd percentile. FGR is further categorized as early-onset (<32 weeks; usually placental insufficiency; high risk of preeclampsia and stillbirth) and late-onset (≥32 weeks; milder placental dysfunction). Causes: placental insufficiency, maternal vascular disease, infections (CMV, toxoplasmosis), aneuploidy, maternal substance use, multiple gestation.
Surveillance & Doppler
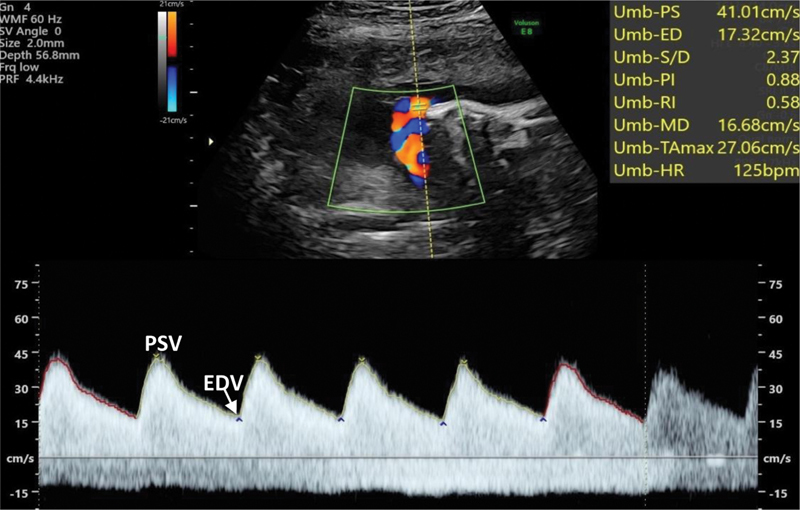
Serial growth every 3–4 weeks. Weekly NSTs and BPPs starting at diagnosis. Umbilical artery Doppler velocimetry is central: normal flow, elevated S/D, absent end-diastolic flow (AEDF), and reversed end-diastolic flow (REDF) represent progressive placental dysfunction. Middle cerebral artery (MCA) Doppler detects fetal brain-sparing (“cephalization”) and fetal anemia (peak systolic velocity). Ductus venosus (DV) Doppler is used in severe early-onset FGR for delivery timing.
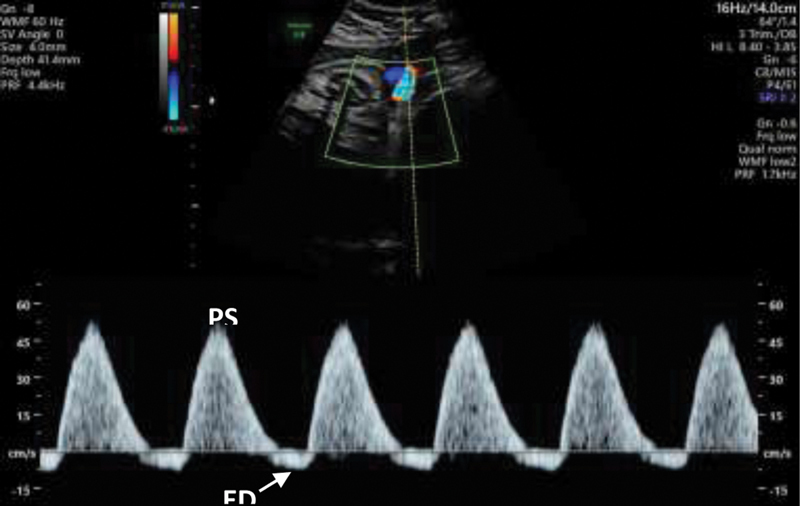
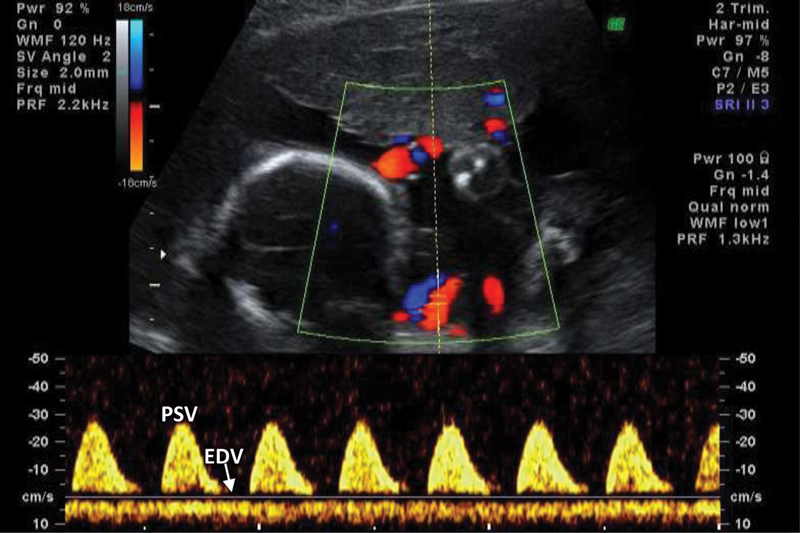
| Scenario | Delivery Timing |
|---|---|
| FGR with normal Dopplers | 36w0d–37w6d |
| FGR with elevated umbilical S/D | 37w0d |
| FGR with AEDF (absent end-diastolic flow) | 34w0d |
| FGR with REDF (reversed end-diastolic flow) | 30w0d |
| Severe FGR (<3rd percentile) | 37w0d |
Large for Gestational Age (LGA) & Macrosomia
LGA: EFW >90th percentile. Macrosomia: EFW ≥4,000 g (high risk) or ≥4,500 g (extreme, shoulder dystocia risk). Cesarean is offered for EFW ≥5,000 g in non-diabetics or ≥4,500 g in diabetics.
11 Amniotic Fluid Disorders
Amniotic fluid is measured on ultrasound by the amniotic fluid index (AFI) — the sum of the deepest vertical pocket in each of four uterine quadrants — or by the single deepest pocket (SDP / MVP).
| Condition | AFI | Single Deepest Pocket |
|---|---|---|
| Oligohydramnios | <5 cm | <2 cm |
| Borderline/Low | 5–8 cm | — |
| Normal | 8–24 cm | 2–8 cm |
| Polyhydramnios (mild) | 24–29.9 cm | 8–11.9 cm |
| Polyhydramnios (moderate) | 30–34.9 cm | 12–15.9 cm |
| Polyhydramnios (severe) | ≥35 cm | ≥16 cm |
| Anhydramnios | No measurable pocket | — |
Oligohydramnios causes: PPROM, placental insufficiency, post-term pregnancy, fetal renal anomalies (agenesis, posterior urethral valves), maternal ACE inhibitor/NSAID use. Polyhydramnios causes: fetal GI obstruction (esophageal/duodenal atresia), neurologic impairment (impaired swallowing), diabetes, fetal anemia, hydrops, TTTS (recipient twin). Severe polyhydramnios can be managed with amnioreduction for maternal respiratory distress.
12 Multiple Gestation
Multiple gestation is a core MFM domain. Outcomes depend on chorionicity (number of placentas) and amnionicity (number of sacs), which are determined by the timing of embryo splitting and must be assigned in the first trimester.
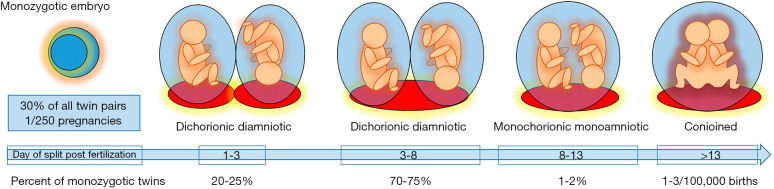
| Type | Placentas | Sacs | Notes |
|---|---|---|---|
| DCDA (dichorionic/diamniotic) | 2 | 2 | All dizygotic twins + ~30% of monozygotic; lowest risk |
| MCDA (monochorionic/diamniotic) | 1 | 2 | ~70% of monozygotic; TTTS/TAPS/sIUGR risk |
| MCMA (monochorionic/monoamniotic) | 1 | 1 | Rare; high cord entanglement mortality |
| Conjoined twins | 1 | 1 | Very rare; late splitting |
First-trimester ultrasound signs: “lambda sign” (thick dividing membrane) = DCDA; “T sign” (thin membrane insertion) = MCDA.
Twin-Twin Transfusion Syndrome (TTTS)
Unbalanced placental vascular anastomoses in monochorionic twins create a donor–recipient imbalance. Quintero staging:
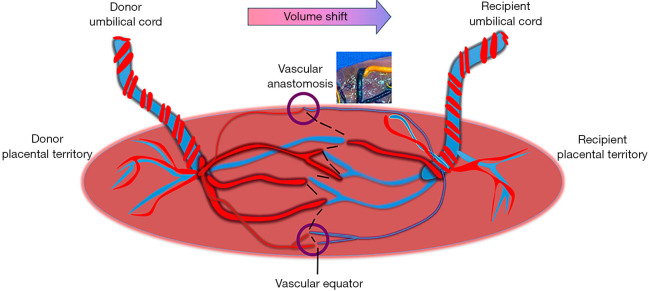
| Stage | Criteria |
|---|---|
| I | Oligohydramnios in donor (MVP <2) + polyhydramnios in recipient (MVP >8, or >10 after 20w); bladder still visible in donor |
| II | Donor bladder not visible |
| III | Abnormal Doppler (AEDF/REDF in UA, reversed DV a-wave, pulsatile umbilical vein) in either twin |
| IV | Hydrops in either twin |
| V | Demise of either twin |
Stage II+ is treated with fetoscopic laser photocoagulation of the placental anastomoses. SMFM Consult Series on Twin Complications.
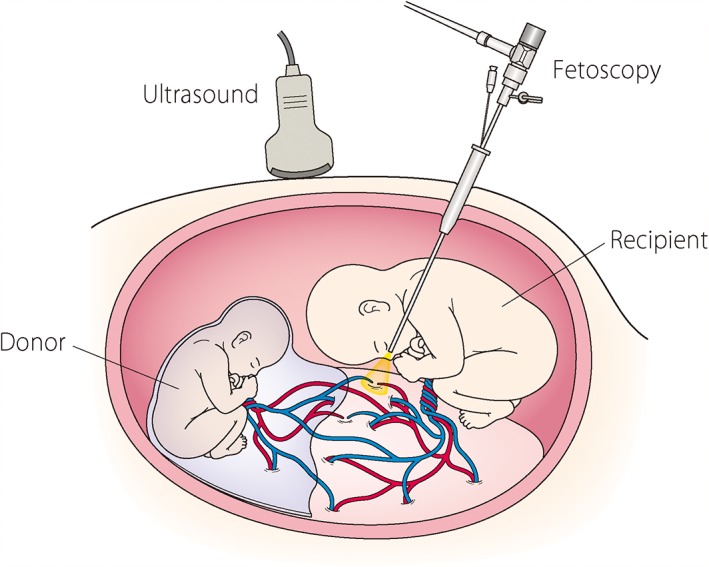
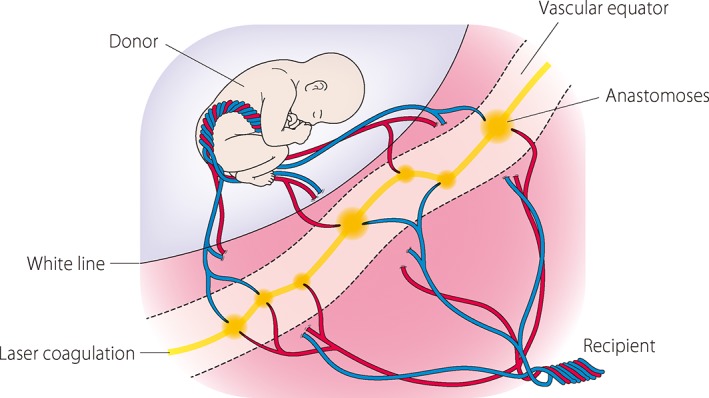
Other Monochorionic Complications
TAPS (twin anemia-polycythemia sequence): chronic slow transfusion through small anastomoses, without fluid discordance. Diagnosed by MCA Doppler (donor high PSV, recipient low). sIUGR (selective IUGR): unequal placental sharing; classified by umbilical artery Doppler pattern (Types I, II, III). TRAP sequence: twin reversed arterial perfusion — acardiac twin perfused by the pump twin.
Delivery Timing
- DCDA: 38w0d
- MCDA: 36w0d–37w6d
- MCMA: 32w0d–34w0d (with antenatal corticosteroids, usually cesarean after inpatient monitoring)
13 Cervical Insufficiency, Preterm Labor & PPROM
Cervical Insufficiency & Cerclage
Painless cervical dilation in the second trimester leading to recurrent pregnancy loss or extreme preterm birth. Indications for cerclage: history-indicated (≥1 prior second-trimester loss or preterm birth; placed 12–14w); ultrasound-indicated (short cervix <25 mm before 24w in a patient with prior preterm birth); exam-indicated / “rescue” (dilated cervix on exam in the second trimester without labor). McDonald and Shirodkar are the two common techniques. Cerclage is removed at 36–37w or with labor/PPROM.
Preterm Labor
Regular contractions with cervical change before 37 weeks. Evaluation: speculum exam (rule out PPROM), fetal fibronectin (fFN) in borderline cases, transvaginal cervical length (<20 mm is concerning). Management:
- Antenatal corticosteroids (betamethasone 12 mg IM q24h ×2 doses or dexamethasone 6 mg IM q12h ×4 doses) between 24w0d and 33w6d; rescue course possible 34w0d–36w6d for late preterm risk
- Magnesium sulfate for fetal neuroprotection <32 weeks (reduces cerebral palsy)
- Tocolysis (48 hours to allow steroid benefit): nifedipine (first-line), indomethacin (<32w), or terbutaline (short-term)
- GBS prophylaxis if GBS status unknown or positive
- Transfer to a center with appropriate NICU level
PPROM (Preterm Prelabor Rupture of Membranes)
Rupture of membranes before 37w without labor. Diagnosis: pooling on speculum exam, nitrazine test (pH >6.5, amniotic fluid is alkaline), ferning, and commercial immunoassays (AmniSure, ROM Plus). Management is gestational-age dependent: expectant with latency antibiotics (ampicillin + azithromycin + amoxicillin) and steroids before 34w, delivery at 34w0d if uncomplicated. ACOG Practice Bulletin 217 (Prelabor Rupture of Membranes).
14 Fetal Demise & Recurrent Pregnancy Loss
Intrauterine Fetal Demise (IUFD / Stillbirth)
Fetal death ≥20 weeks. Workup after delivery: fetal autopsy, placental pathology, karyotype/microarray, Kleihauer-Betke (fetomaternal hemorrhage), antiphospholipid antibodies, screening for hereditary thrombophilias (selectively), HbA1c, TSH, and toxicology. Delivery mode is typically induction of labor; dilation and evacuation remains an option in select cases. Counseling on future pregnancy recurrence risk is part of the MFM follow-up.
Recurrent Pregnancy Loss (RPL)
Two or more clinical pregnancy losses. Workup: parental karyotype, antiphospholipid antibody panel (LA, aCL, β2-GPI), uterine imaging (HSG or saline infusion sonography for septum/fibroids/synechiae), TSH, HbA1c. Most RPL is unexplained; reassurance and close early surveillance are the mainstay. Low-dose aspirin + LMWH if APS confirmed.
15 Fetal Anomalies, Isoimmunization & Hydrops
Aneuploidy Screening & Diagnostic Testing
First-trimester screening combines nuchal translucency (NT) ultrasound at 11w0d–13w6d with serum PAPP-A and free β-hCG. Cell-free DNA (cfDNA) / NIPT tests maternal blood for fetal chromosomal aneuploidy (trisomy 21, 18, 13, sex chromosome aneuploidy) from 10w onward with high sensitivity but requires confirmation for positives. Diagnostic testing: chorionic villus sampling (CVS) at 10w0d–13w6d or amniocentesis at ≥15w.
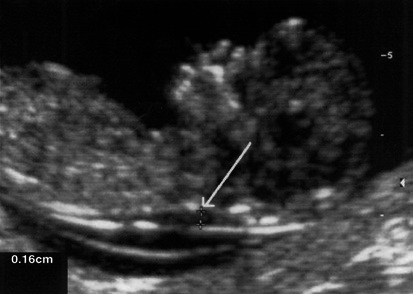
Anatomy Ultrasound
Detailed fetal anatomy scan at 18w0d–22w6d assesses cranium, face, heart, spine, abdomen, kidneys, limbs, and placenta. A detailed survey (SMFM definition) is performed in high-risk pregnancies (diabetes, teratogen exposure, abnormal screening, FGR, multiples).
Fetal Echocardiography
Specialized cardiac imaging at 20–22w for congenital heart disease screening in high-risk pregnancies (pregestational diabetes, maternal CHD, fetus with extracardiac anomaly, family history, NT ≥3.5 mm, monochorionic twins, teratogen exposure). Evaluates four-chamber view, outflow tracts, aortic arch, ductal arch, and atrial/ventricular septa.
Isoimmunization (Rh, Kell, Duffy)
Maternal alloantibodies against fetal red cell antigens cross the placenta and cause fetal hemolysis. Anti-D (Rh) is classic; anti-Kell (Kell kills) is especially severe because it suppresses erythropoiesis in addition to hemolysis. Workup: maternal antibody titer (critical titer ≥1:16 for most antibodies, any titer for Kell), paternal antigen status, fetal genotype (cfDNA or amniocentesis), and serial MCA peak systolic velocity (MCA-PSV) Doppler. MCA-PSV >1.5 multiples of the median (MoM) predicts moderate-severe fetal anemia. Treatment: intrauterine transfusion (IUT) via cordocentesis. Anti-D immunoglobulin (RhoGAM) 300 µg at 28 weeks and within 72 hours of delivery (and after any sensitizing event: bleeding, trauma, amniocentesis) prevents Rh isoimmunization.
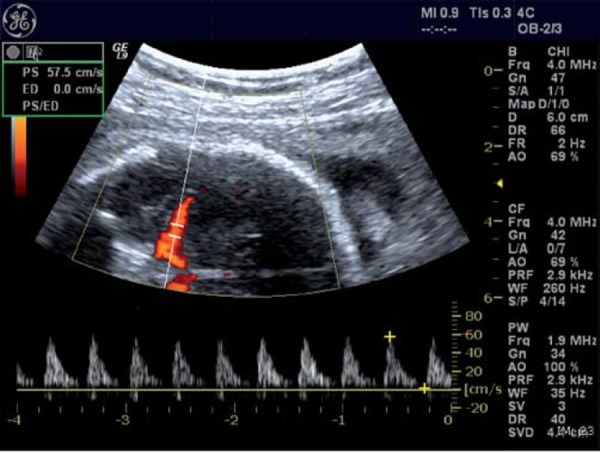
Fetal Hydrops
Abnormal fluid accumulation in two or more fetal compartments (ascites, pleural effusion, pericardial effusion, skin edema, polyhydramnios, placentomegaly). Immune hydrops: isoimmunization. Nonimmune hydrops: cardiac anomalies/arrhythmias, chromosomal, infections (parvovirus B19, CMV, toxo, syphilis), TTTS, chylothorax, α-thalassemia major (Bart hydrops), lymphatic anomalies. Workup is exhaustive; treatment is directed at the cause (IUT for parvovirus anemia, digoxin for SVT, laser for TTTS).
16 Congenital Infections (TORCH, GBS, HSV, HIV)
The TORCH acronym catches perinatal infections that cause fetal injury: Toxoplasmosis, Other (syphilis, parvovirus, varicella, Zika, listeria), Rubella, CMV, HSV.
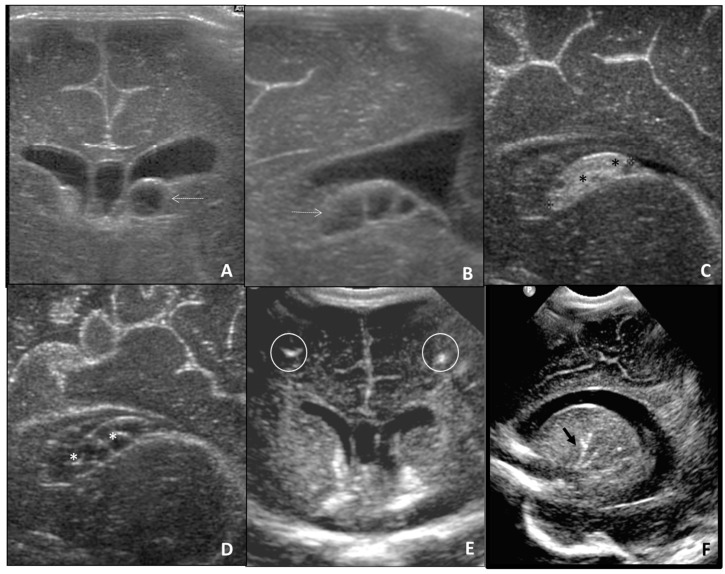
- CMV: Most common congenital infection. Primary maternal infection carries highest fetal risk (SNHL, microcephaly, periventricular calcifications, FGR). Suspect with mono-like illness or ultrasound findings. Maternal IgM/IgG; amniocentesis for fetal PCR.
- Parvovirus B19: Fifth disease; causes fetal anemia, hydrops, demise. Serial MCA-PSV surveillance for 8–12 weeks post-exposure; IUT if severe anemia.
- Toxoplasmosis: Cat feces, undercooked meat. Triad: chorioretinitis, hydrocephalus, intracranial calcifications. Spiramycin (maternal) + pyrimethamine/sulfadiazine (fetal infection confirmed).
- Syphilis: Screened at first visit and again third trimester in high-risk. Treatment: penicillin G (desensitize if allergic — no alternatives in pregnancy).
- Zika: Microcephaly, severe brain anomalies. Avoid travel to endemic areas.
- GBS: Universal vaginal/rectal culture at 36w0d–37w6d. Intrapartum prophylaxis with penicillin (cefazolin, clindamycin, vancomycin for allergies) for positive cultures, prior GBS-affected infant, or unknown status with risk factors.
- HSV: Active genital lesions or prodrome at labor = cesarean delivery. Suppressive acyclovir from 36w in women with history of genital HSV.
- HIV: ART throughout pregnancy; goal VL <1,000 copies/mL for vaginal delivery, otherwise cesarean at 38w. Intrapartum zidovudine if VL elevated. Avoid breastfeeding in US.
17 Substance Use & Severe Maternal Morbidity
Substance Use in Pregnancy
Universal screening with validated tools (4P's, NIDA, SURP-P). Opioid use disorder is treated with medication-assisted therapy — buprenorphine or methadone (both safer than withdrawal, which increases fetal loss). Neonatal abstinence syndrome (NAS) is anticipated. Alcohol is teratogenic (FASD). Tobacco cessation counseling every visit; nicotine replacement is preferred to continued smoking. Cocaine and methamphetamine increase abruption and preterm birth risk.
Peripartum Cardiomyopathy (PPCM)
New-onset heart failure with LVEF <45% in the last month of pregnancy or within 5 months postpartum, without other identifiable cause. Treatment per standard HFrEF protocols with pregnancy-safe drugs (no ACE/ARB antepartum; safe postpartum if not breastfeeding or with enalapril/captopril). Anticoagulation for LVEF <30% due to LV thrombus risk. Future pregnancies are high-risk, especially if LVEF does not recover.
Amniotic Fluid Embolism (AFE)
Catastrophic syndrome of cardiovascular collapse, hypoxia, and DIC during labor or immediately postpartum. Pathogenesis is an anaphylactoid reaction to fetal antigens. Mortality is high; management is supportive: ACLS, intubation, massive transfusion protocol, fibrinogen repletion.
Severe Maternal Morbidity (SMM)
CDC defines SMM by 21 indicators (ICU admission, massive transfusion, hysterectomy, eclampsia, acute MI, etc.). MFM teams lead SMM case review and quality improvement. Documentation of quantitative blood loss, maternal early warning system (MEWS) triggers, and time-stamped responses is emphasized.
18 MFM Procedures — A to Z
| Procedure | Description & Indications |
|---|---|
| Amniocentesis | Transabdominal needle aspiration of amniotic fluid under US guidance; ≥15w0d. Indications: aneuploidy diagnosis, fetal infection PCR, lung maturity (rare), fluid reduction. Loss rate ~1/900–1/1600. |
| Chorionic Villus Sampling (CVS) | Transabdominal or transcervical sampling of placental villi at 10w0d–13w6d for karyotype/microarray. Earlier diagnosis than amnio; cannot diagnose NTDs. |
| Cordocentesis / PUBS | Percutaneous umbilical blood sampling. Fetal hematocrit assessment and intrauterine transfusion access. |
| Intrauterine Transfusion (IUT) | O-negative, CMV-negative, irradiated, leukoreduced PRBC transfusion into umbilical vein for fetal anemia (isoimmunization, parvovirus). |
| Fetoscopic Laser Photocoagulation | Endoscopic laser ablation of placental anastomoses for TTTS stage II+. Referral procedure at fetal therapy centers. |
| Amnioreduction | Therapeutic removal of amniotic fluid for severe polyhydramnios with maternal respiratory distress or preterm labor. |
| Cervical Cerclage | Suture placed around the cervix (McDonald or Shirodkar technique) for cervical insufficiency. |
| Cervical Length Measurement | Transvaginal ultrasound; the cervix is measured from internal to external os; <25 mm before 24w is abnormal. |
| External Cephalic Version (ECV) | Manual rotation of a breech fetus to cephalic at 36–37w under ultrasound guidance, with tocolysis, in a delivery-ready setting. |
| Fetal Echocardiography | Specialized cardiac ultrasound at 20–22w in high-risk patients. |
| Biophysical Profile (BPP) | 30-minute ultrasound assessing fetal tone, movement, breathing, amniotic fluid, plus NST. |
For any procedure note, document: indication, consent, Rh status (and RhoGAM given if Rh-negative), ultrasound guidance used, fetal heart tones before and after, gauge of needle, amount of fluid withdrawn or transfused, maternal tolerance, complications, and disposition. Procedure notes are audited by hospital QA.
19 Fetal Assessment & Imaging
Nonstress Test (NST)
External fetal monitoring for at least 20 minutes. Reactive NST (≥32w): ≥2 accelerations of ≥15 bpm lasting ≥15 seconds in 20 minutes. Before 32 weeks: ≥2 accelerations of ≥10 bpm for ≥10 seconds. Nonreactive NSTs prompt BPP or further assessment.
Biophysical Profile (BPP)
| Component | Normal (2 points) |
|---|---|
| Fetal breathing movements | ≥1 episode of rhythmic breathing ≥30 sec in 30 min |
| Gross body movement | ≥3 discrete body/limb movements in 30 min |
| Fetal tone | ≥1 episode of active extension with return to flexion |
| Amniotic fluid volume | Single vertical pocket ≥2 cm |
| NST | Reactive |
Total: 10. Scores: 8–10 reassuring; 6 equivocal (repeat or deliver); <6 concerning (consider delivery). Modified BPP = NST + AFI.
Doppler Velocimetry
Umbilical artery (UA): S/D ratio, PI, RI. Normal flow → elevated S/D → AEDF → REDF is a progression of placental resistance. Middle cerebral artery (MCA): PSV for fetal anemia; PI for brain-sparing (low PI → cephalization). Ductus venosus (DV): reversed a-wave is an ominous sign in severe early FGR. Uterine artery: bilateral notching in the early second trimester predicts preeclampsia/FGR.
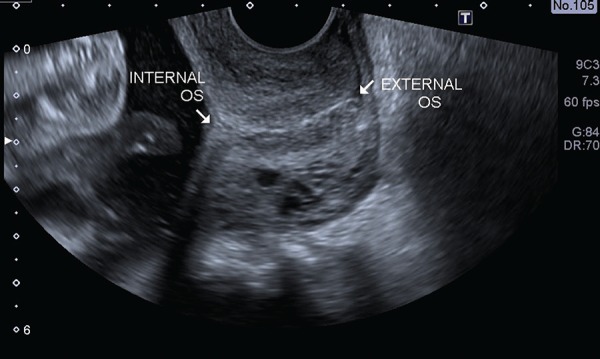
Cervical Length
Transvaginal measurement from internal to external os. A length <25 mm before 24 weeks is short; <15 mm is a threshold for especially high PTB risk. Serial measurements every 2 weeks in high-risk patients.
Imaging Modalities & Labs
Obstetric labs every scribe will document: prenatal panel (CBC, type & screen, rubella, syphilis, HIV, hepatitis B/C, urine culture), aneuploidy screen, 1-hour GCT at 24–28w, repeat CBC and type screen at 28w, GBS culture at 36w, and TSH/HbA1c for chronic conditions. Fetal MRI is used when ultrasound is inconclusive (CNS anomalies, placenta accreta spectrum) per ACR Appropriateness Criteria for obstetric imaging.
20 Medications You Must Know
Antihypertensives
| Drug | Use | Notes |
|---|---|---|
| Labetalol | Chronic + acute severe HTN | IV 10–20 mg q10 min up to 300 mg; oral 100–1200 mg/day divided |
| Nifedipine ER | Chronic HTN | 30–120 mg daily; immediate-release used for acute severe HTN |
| Hydralazine | Acute severe HTN | IV 5–10 mg q20 min; reflex tachycardia |
| Methyldopa | Chronic HTN (historical) | Safest long-term data; depression, sedation |
| ACEi/ARB | Contraindicated | Fetal renal agenesis, oligohydramnios, skull hypoplasia |
Diabetes
| Drug | Notes |
|---|---|
| Insulin (regular, lispro, aspart, NPH, detemir) | First-line in pregnancy; does not cross placenta |
| Metformin | Second-line for GDM; crosses placenta |
| Glyburide | Second-line; crosses placenta; neonatal hypoglycemia |
Preeclampsia & Preterm Labor
| Drug | Use | Dose |
|---|---|---|
| Magnesium sulfate | Seizure prophylaxis in preeclampsia; neuroprotection <32w | 4–6 g IV load, 1–2 g/hr maintenance |
| Betamethasone | Antenatal corticosteroids 24w0d–36w6d | 12 mg IM q24h ×2 |
| Dexamethasone | Alternative antenatal steroid | 6 mg IM q12h ×4 |
| Nifedipine (IR) | Tocolysis (first-line); acute severe HTN | 10–20 mg PO q15–30 min |
| Indomethacin | Tocolysis <32w (ductal constriction after) | 50 mg loading, 25 mg q6h ×48h |
| Terbutaline | Short-term tocolysis, uterine relaxation | 0.25 mg SC; black box <48–72h |
| Low-dose aspirin | Preeclampsia prevention from 12–16w in high-risk | 81–162 mg PO daily |
Anticoagulation
| Drug | Notes |
|---|---|
| Enoxaparin (LMWH) | First-line; prophylactic or therapeutic; doesn't cross placenta |
| Unfractionated heparin | Renal failure, peri-delivery (short half-life) |
| Warfarin | Contraindicated except mechanical valves (teratogen) |
| DOACs | Not used in pregnancy |
Miscellaneous Pregnancy Medications
- Levothyroxine: Hypothyroidism; increase dose ~30% at pregnancy confirmation.
- Prednisone: Autoimmune disease; minimally crosses placenta (11β-HSD2 inactivation).
- Hydroxychloroquine, azathioprine, certolizumab, tacrolimus: Immunosuppressants compatible with pregnancy.
- Antiretrovirals (HIV): Continue ART throughout; integrase inhibitor-based regimens preferred.
- Ursodeoxycholic acid: ICP treatment.
- Anti-D immunoglobulin (RhoGAM): 300 µg IM at 28w and postpartum (within 72h), after any sensitizing event.
- Penicillin G: Syphilis treatment; desensitize for allergy.
- Azithromycin + ampicillin + amoxicillin: PPROM latency regimen.
- Rh(D)-negative pregnancy kits, tranexamic acid (TXA): Postpartum hemorrhage management.
21 Classification Systems & Criteria
Bishop Score (Cervical Readiness for Induction)
| Factor | 0 | 1 | 2 | 3 |
|---|---|---|---|---|
| Dilation (cm) | Closed | 1–2 | 3–4 | ≥5 |
| Effacement (%) | 0–30 | 40–50 | 60–70 | ≥80 |
| Station | -3 | -2 | -1/0 | +1/+2 |
| Consistency | Firm | Medium | Soft | — |
| Position | Posterior | Mid | Anterior | — |
Score ≤6 = unfavorable (ripening needed); ≥8 = favorable.
Modified WHO Maternal Cardiovascular Risk (mWHO)
| Class | Risk | Examples |
|---|---|---|
| I | No detectable increase | Mild PS, small VSD/PDA, repaired lesion without residua |
| II | Small increase in mortality, moderate in morbidity | Unrepaired ASD/VSD, repaired TOF, most arrhythmias |
| II–III | Intermediate | Mild LV dysfunction, HCM, repaired coarctation |
| III | Significant increase in mortality and severe morbidity | Mechanical valve, systemic RV, Fontan, unrepaired cyanotic CHD |
| IV | Pregnancy contraindicated | Pulmonary HTN, severe systemic ventricular dysfunction (EF <30%, NYHA III/IV), severe MS, severe symptomatic AS, Marfan aorta >45 mm, Eisenmenger |
Other Systems Referenced Throughout
Quintero staging for TTTS (Section 12), Carpenter-Coustan and IADPSG for GDM (Section 4), BPP Manning criteria (Section 19), PAS accreta/increta/percreta (Section 8), HELLP Tennessee and Mississippi criteria (Section 6), White classification of diabetes in pregnancy (A1/A2 + pregestational B–T).
22 Obstetric Physical Exam
The MFM physical exam is focused and standardized. Every visit includes:
- Vitals: BP (document both arms in hypertensive patients), HR, RR, SpO2, weight, BMI
- General: Well-appearing, in NAD, comfortable
- HEENT: No facial edema, no scleral icterus, oropharynx clear
- Neck: No thyromegaly, no lymphadenopathy, JVP normal
- CV: RRR, no murmurs (physiologic systolic ejection murmur common in pregnancy)
- Pulm: CTA bilaterally, no respiratory distress
- Abdomen: Gravid, soft, nontender, fundal height [cm], FHT [bpm] on Doppler, no contractions
- Extremities: Trace/1+/2+/3+ pitting edema, DTRs 2+/4 (3+ or clonus in preeclampsia), calves nontender
- Skin: No rash, no excoriations (relevant for ICP), linea nigra, striae
- Neuro: Alert, oriented, no focal deficits, no visual disturbance
- Pelvic / sterile speculum (when indicated): cervix appearance, pooling, dilation, effacement, station, presentation
Fundal Height
From 20 weeks, the fundal height in centimeters from the pubic symphysis should approximate gestational age in weeks (±2 cm). Lag suggests FGR or oligohydramnios; excess suggests LGA, polyhydramnios, or multiples.
Leopold Maneuvers
Four palpations to determine fetal lie, presentation, and position: (1) fundal grip — which pole is in the fundus (head vs breech)? (2) umbilical grip — which side is the back? (3) suprapubic grip — is the presenting part engaged? (4) pelvic grip — attitude of the head.
Cervical Exam
Documented as dilation / effacement / station (e.g., “4/90%/−1”). Station is measured in cm above (−) or below (+) the ischial spines; −5 to +5. Position and consistency complete the Bishop score.
23 Abbreviations Master List
Pregnancy & Anatomy
Diagnoses
Procedures & Testing
Medications
24 Sample HPI Templates
34 y/o G2P1001 at 31w3d by first-trimester US with a dichorionic singleton pregnancy, presenting for MFM consultation for suspected preeclampsia. Prenatal course complicated by chronic hypertension on labetalol 200 mg BID and baseline proteinuria (300 mg/24h at 12w). BPs at home have been rising over the past 5 days, 150s–160s/90s–100s. Presented to L&D last night with BP 168/104, persistent frontal headache, and RUQ discomfort. Labs notable for AST 92, ALT 110, platelets 98k, creatinine 0.9 (baseline 0.7), urine P/Cr 1.8. Admitted for preeclampsia with severe features superimposed on chronic HTN. Currently on magnesium sulfate and IV labetalol. FM present, no VB, no LOF, no contractions. Plan: betamethasone completed at outside hospital, maternal stabilization, delivery planned within 24 hours given severe features at 31w3d. Fetal status reassuring on continuous monitoring, EFW 1,450 g (25th percentile), AFI 10, normal umbilical Dopplers.
29 y/o G1P0 at 28w2d presenting to MFM clinic for GDM management after failed 3-hour OGTT (fasting 98, 1h 195, 2h 168, 3h 148 — 3 abnormal values by Carpenter-Coustan). BMI 34, family history of T2DM. Has been on diabetic diet for 2 weeks; fingerstick log reviewed: fasting averaging 102–108, 1-hour postprandials 145–160 after dinner. Starting insulin today: NPH 10 units qHS and lispro 4 units with dinner. Fetal kick counts normal. No polyhydramnios on growth scan; EFW 70th percentile, AC 75th percentile. Plan: A2GDM, insulin initiation, weekly NSTs starting 32w, growth scan at 32w, delivery 39w0d if well-controlled.
36 y/o G3P2002 at 29w5d presents to MFM for growth follow-up. Anatomy scan at 20w normal. Growth scan at 28w showed EFW 8th percentile, AC 6th percentile. Repeat today: EFW 850 g (6th percentile), AC <5th percentile, HC and FL appropriate (asymmetric FGR). Umbilical artery Doppler with elevated S/D 4.2 (>95th percentile), end-diastolic flow preserved. MCA PI decreased (cerebroplacental ratio <1). Amniotic fluid low-normal (AFI 8). FM decreased per patient over last 2 days but NST today reactive. No BP elevation, no proteinuria. Plan: admit for antenatal corticosteroids (betamethasone #1 today), twice-weekly BPP, weekly Doppler, growth in 2 weeks, delivery at 34w0d if progression to AEDF, otherwise 37w0d.
27 y/o G1P0 at 22w0d with MCDA twin pregnancy (T sign on first-trimester US, single placenta). Referred from general OB for MFM co-management. Both twins with concordant growth at 20w anatomy (EFWs within 5%), no structural anomalies, normal cardiac views. Today: growth discordance now 18% (Twin A 480 g, Twin B 395 g); MVP Twin A 5.5 cm, Twin B 1.8 cm; Twin B bladder not visualized; umbilical artery Doppler Twin B with AEDF. Consistent with TTTS Stage III. No maternal symptoms; no contractions. Plan: urgent referral to fetal therapy center for fetoscopic laser photocoagulation evaluation; betamethasone deferred pending fetal therapy team input.
32 y/o G4P0030 presenting for preconception MFM consultation after three first-trimester losses (7w, 9w, 6w). All losses were spontaneous; no prior workup. PMH notable for mild hypothyroidism on levothyroxine 50 mcg (TSH 2.1 most recent). No known autoimmune disease, no thrombosis history. Family history: sister with two losses and antiphospholipid syndrome. RPL workup ordered: parental karyotypes, antiphospholipid panel (LA, aCL, anti-β2 GP1), TSH with reflex, HbA1c, saline infusion sonohysterogram for uterine cavity. Counseled on recurrence risk, benefit of early ultrasound, and role of low-dose aspirin + LMWH if APS confirmed. Will follow up with results in 3 weeks.
41 y/o G4P3003 at 14w2d with chronic hypertension, obesity (BMI 38), and prior GDM presenting for MFM new OB consult. Previously on lisinopril 20 mg (discontinued at positive pregnancy test); now on labetalol 200 mg BID with home BPs 130s–140s/80s. No headache, visual changes, RUQ pain, or swelling. Baseline workup: CBC normal, CMP Cr 0.6, urine P/Cr 0.15, HbA1c 5.7, TSH 1.8, EKG normal. First-trimester aneuploidy screen pending. Counseled on aspirin 81 mg daily starting today for preeclampsia prevention per USPSTF, goal BP <140/90 per CHAP, early GDM screen at 14–16w given risk factors, growth scans every 4 weeks from 28w, and delivery at 37w0d if pregnancy remains uncomplicated. Established care, returning in 4 weeks.
Closing Note
Maternal-fetal medicine sits at the intersection of two patients, two physiologies, and two sets of stakes. Great MFM scribes master both: they chart the mother's blood pressure trend and the fetus's Doppler waveform with equal care. They recognize that a single sentence — “headache”, “decreased fetal movement”, “leaking fluid” — can redirect a visit from a routine growth scan into an emergent admission. Know the numbers, know the thresholds, and when in doubt, write it down. In high-risk obstetrics, the documentation you create becomes the safety net both patients depend on.
25 References & Sources
Clinical Practice Guidelines
ACOG Practice Bulletin 222: Gestational Hypertension and Preeclampsia. Obstet Gynecol. 2020.
ACOG Practice Bulletin 190: Gestational Diabetes Mellitus. Obstet Gynecol. 2018.
ACOG Practice Bulletin 201: Pregestational Diabetes Mellitus. Obstet Gynecol. 2018.
ACOG Practice Bulletin 227: Fetal Growth Restriction. Obstet Gynecol. 2021.
ACOG Practice Bulletin 217: Prelabor Rupture of Membranes. Obstet Gynecol. 2020.
ACOG Practice Bulletin 196: Thromboembolism in Pregnancy. Obstet Gynecol. 2018.
ACOG/SMFM Obstetric Care Consensus: Placenta Accreta Spectrum. Obstet Gynecol. 2018.
SMFM Consult Series 53: Intrahepatic Cholestasis of Pregnancy. Am J Obstet Gynecol. 2021.
SMFM Consult Series: Twin-Twin Transfusion Syndrome. Am J Obstet Gynecol.
USPSTF: Aspirin Use to Prevent Preeclampsia and Related Morbidity and Mortality. JAMA. 2021.
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: Female Reproductive System. OpenStax College. CC BY 3.0.
Figure 2: The Embryo. OpenStax College. CC BY 3.0.
Figure 3: Pregnancy. Wikimedia Commons. Public domain.
Figure 4: First-Trimester CRL Ultrasound. Wolfgang Moroder. CC BY-SA 3.0.
Figure 5: Prenatal Ultrasound. Wikimedia Commons. Public domain.