Nephrology
Every diagnosis, syndrome, electrolyte and acid-base disturbance, dialysis modality, transplant concept, medication, and documentation pattern a nephrology scribe needs.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Renal Anatomy & Nephron Physiology
Nephrology is the medical specialty devoted to the kidneys — a pair of retroperitoneal, bean-shaped organs that filter roughly 180 liters of plasma per day, adjust salt and water balance, excrete nitrogenous waste, regulate acid-base status, control blood pressure through the renin-angiotensin-aldosterone system (RAAS), activate vitamin D, and stimulate red-cell production via erythropoietin. A scribe who does not understand the nephron will never write a convincing nephrology note, because virtually every diagnosis in this field is a story about one or more segments of the nephron failing to do their job.
Gross Anatomy
Each kidney sits at the T12–L3 level, surrounded by Gerota's fascia and perirenal fat. The outer cortex contains glomeruli, proximal and distal convoluted tubules, and cortical collecting ducts. The inner medulla is organized into pyramids whose apices (papillae) empty into minor calyces, which merge into major calyces, then the renal pelvis, then the ureter. Blood enters via the renal artery (a direct branch of the abdominal aorta just below the SMA at L1–L2), which divides into segmental, interlobar, arcuate, interlobular, and finally afferent arterioles supplying each glomerulus. Blood leaves the glomerulus through the efferent arteriole, then forms the peritubular capillaries and vasa recta that wrap the tubules before draining into the renal vein and IVC.
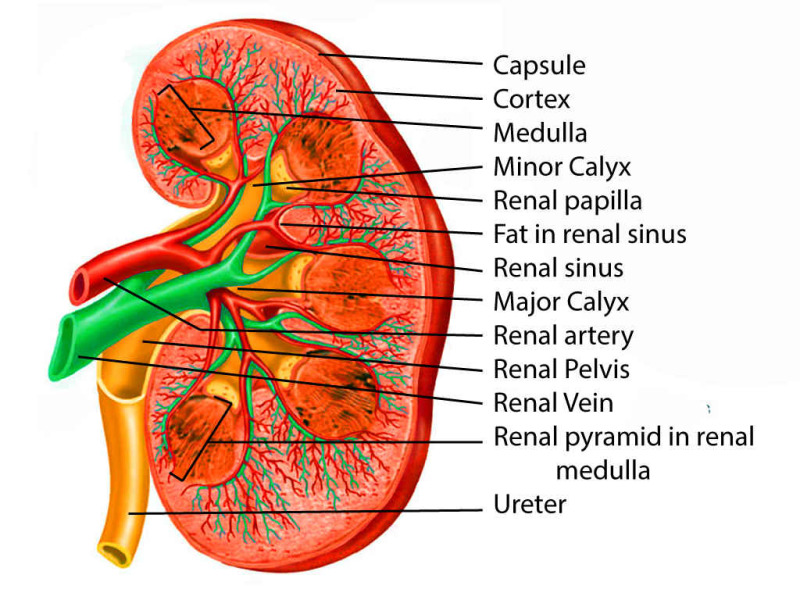
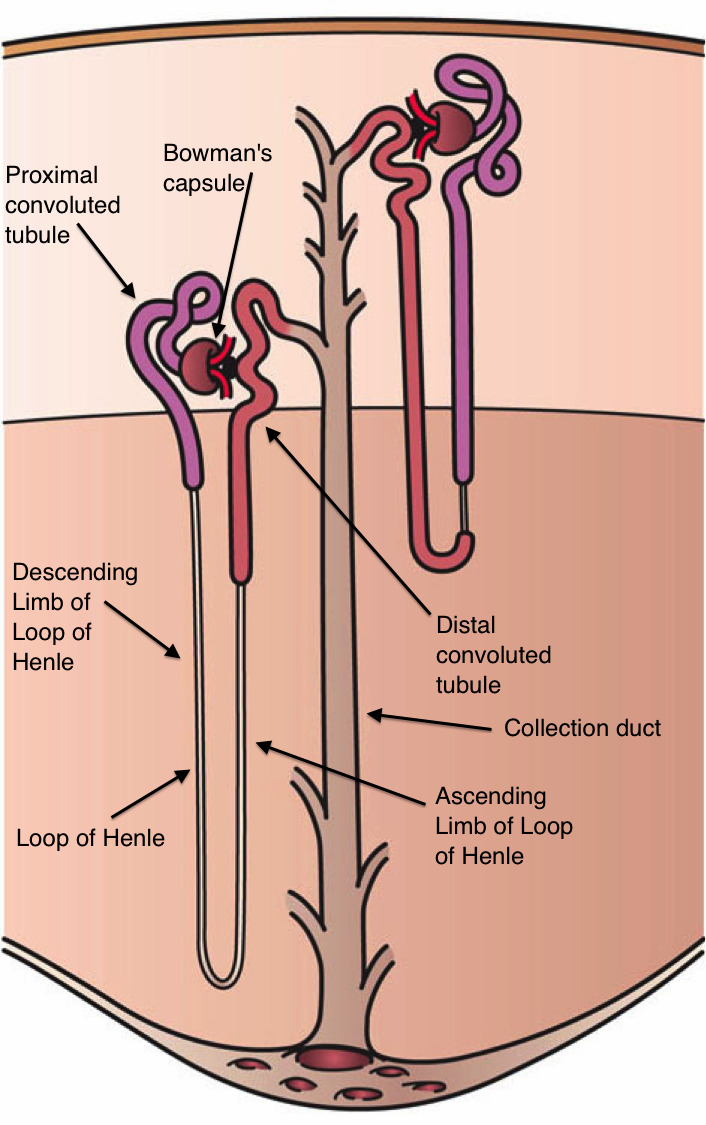
The Nephron — The Functional Unit
Each kidney contains roughly one million nephrons. Every nephron begins with a glomerulus (a tuft of fenestrated capillaries surrounded by Bowman's capsule) that ultrafilters plasma under the driving force of afferent arteriolar pressure. The filtrate then traverses the proximal convoluted tubule (PCT), where roughly 65% of filtered Na+, water, glucose, amino acids, phosphate, and bicarbonate are reabsorbed; the thin descending and thick ascending limbs of the loop of Henle, which generate the medullary concentration gradient via countercurrent multiplication and reabsorb another 25% of Na+ through the NKCC2 transporter (the target of loop diuretics); the distal convoluted tubule (DCT), where the NCC cotransporter (target of thiazides) fine-tunes Na+ reabsorption and calcium handling is regulated; and finally the collecting duct, where principal cells respond to aldosterone (ENaC channel — target of amiloride/spironolactone) and antidiuretic hormone (ADH/vasopressin opens aquaporin-2 channels), and intercalated cells handle final acid-base trimming.
Glomerular Filtration & RAAS
The glomerular filtration rate (GFR) is the volume of plasma filtered per minute (normal ~ 100–120 mL/min/1.73 m2) and is the single most important number in nephrology. It depends on renal plasma flow, the hydrostatic pressure across the glomerular capillary wall, and the integrity of the filtration barrier (endothelium → basement membrane → podocyte slit diaphragms). The juxtaglomerular apparatus — where the macula densa of the thick ascending limb contacts the afferent arteriole — senses distal Na+/Cl− delivery and releases renin when perfusion falls. Renin cleaves angiotensinogen to angiotensin I; ACE converts it to angiotensin II, which constricts the efferent arteriole (preserving GFR when flow is low), stimulates aldosterone, promotes thirst and ADH release, and drives Na+ reabsorption in the PCT. This axis is why ACE inhibitors, ARBs, and mineralocorticoid antagonists sit at the center of nephrology therapeutics.
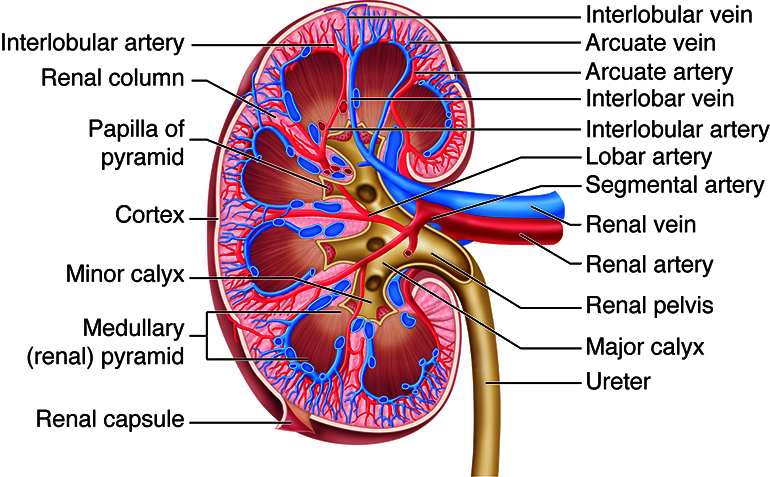
When a nephrologist says "the afferent is constricted" or "the efferent is dilated," they are describing why GFR fell. NSAIDs constrict the afferent (lose prostaglandin vasodilation); ACEi/ARBs dilate the efferent. Both drop GFR, but the mechanism matters for the note and for the plan.
Endocrine Functions of the Kidney
Beyond filtration, the kidney is a major endocrine organ. The juxtaglomerular cells produce renin in response to low perfusion, sympathetic tone, or low distal Na delivery. The peritubular interstitial fibroblasts of the cortex produce erythropoietin (EPO) in response to hypoxia; loss of EPO production is the primary driver of anemia of CKD. The proximal tubule hydroxylates 25-hydroxyvitamin D to the active form 1,25-dihydroxyvitamin D (calcitriol) via 1-alpha-hydroxylase — a step progressively lost in CKD, producing the hypocalcemia, secondary hyperparathyroidism, and renal osteodystrophy that define CKD-MBD. The kidney also handles phosphate excretion under the control of FGF-23, which rises early in CKD and is increasingly recognized as a cardiovascular risk marker.
How GFR Is Estimated
Serum creatinine alone is a poor surrogate because it is affected by muscle mass, diet, and tubular secretion. eGFR is calculated from creatinine using the CKD-EPI 2021 equation, which removed the race coefficient and is now the standard in the US. Cystatin C (less muscle-dependent) is used as a confirmatory test when creatinine-based eGFR is unreliable (amputees, malnourished, cirrhotic, body-builders). Measured GFR using iothalamate, inulin, or iohexol clearance is reserved for research or borderline transplant donor evaluation.
02 Scribe Documentation Framework
Nephrology notes live at the intersection of internal medicine, critical care, and outpatient chronic disease management. A consult note on a hospitalized AKI patient, an outpatient CKD follow-up, a dialysis progress note, and a transplant clinic visit all have different rhythms but share one feature: every note is built around numbers. Creatinine trend, eGFR, potassium, bicarbonate, urine output, weight, and blood pressure show up in every assessment. Capture them exactly.
CC: "Elevated creatinine," "follow-up CKD stage 4," "hyperkalemia," "dialysis access evaluation," "post-transplant day 30."
HPI: Document baseline creatinine/eGFR, the trajectory of current renal function, urine output (oliguria < 400 mL/day, anuria < 100 mL/day), volume symptoms (edema, orthopnea, PND, weight gain), uremic symptoms (fatigue, anorexia, nausea, pruritus, altered mental status, metallic taste), exposures (contrast, NSAIDs, aminoglycosides, vancomycin, PPIs, herbal supplements), and prior kidney history (stones, transplant, dialysis vintage).
ROS: Constitutional, CV (chest pain, DOE), GI (nausea, vomiting, diarrhea — volume loss), GU (dysuria, hematuria, foamy urine — proteinuria, flank pain), MSK (cramps, bone pain), skin (rash — AIN or vasculitis), neuro (asterixis, encephalopathy).
PMHx: Diabetes (type, A1C, duration), hypertension, CAD, CHF, cirrhosis, SLE, HIV, HCV/HBV, prior AKI, nephrolithiasis, BPH, malignancy, prior kidney biopsy results, family history of PKD or Alport.
Vitals: BP (seated and standing if orthostasis suspected), HR, weight (compare to dry weight for dialysis patients), I/Os, net fluid balance.
Exam: Volume status (JVP, lung crackles, S3, peripheral/sacral edema, mucous membranes, skin turgor), AV fistula/graft (thrill, bruit, signs of infection, steal), PD catheter exit site, transplant graft tenderness over the iliac fossa.
Data: BMP (Na, K, Cl, HCO3, BUN, Cr, glucose, anion gap), CMP (add Ca, Mg, Phos, albumin, LFTs), CBC, UA with microscopy, urine Na/Cr/protein, spot UPCR or UACR, ABG/VBG, renal ultrasound findings. Always record numbers, not adjectives.
Nephrology A&P is classically organized by problem: (1) AKI/CKD with stage and etiology, (2) each electrolyte and acid-base derangement, (3) volume status, (4) blood pressure, (5) anemia/mineral-bone disease (CKD), (6) dialysis modality/access, (7) medication reconciliation (renally dosed), and (8) dietary plan. Every problem gets its own mini-plan.
Always chart the baseline creatinine. "Cr 2.1" means nothing without context; "Cr 2.1 from baseline 0.9 two weeks ago" is a diagnosis in the making. Pull the trend from prior labs before the attending walks in the room.
03 Acute Kidney Injury (AKI) Acute
Acute kidney injury is the abrupt decline in kidney function over hours to days. It is the bread-and-butter inpatient nephrology consult and is defined by the KDIGO 2012 AKI guidelines using serum creatinine and urine output criteria. AKI is classified mechanistically into three buckets: prerenal (reduced perfusion), intrinsic (parenchymal damage to glomeruli, tubules, interstitium, or vessels), and postrenal (obstruction).
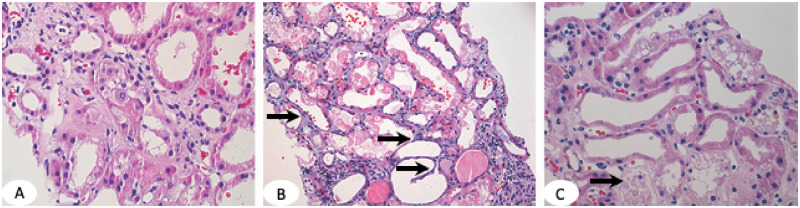
KDIGO Definition & Staging
AKI is present when any one of the following occurs: increase in serum creatinine by ≥ 0.3 mg/dL within 48 hours, increase in serum creatinine to ≥ 1.5 times baseline within the prior 7 days, or urine volume < 0.5 mL/kg/hr for 6 hours.
| Stage | Serum Creatinine | Urine Output |
|---|---|---|
| 1 | 1.5–1.9× baseline OR ≥ 0.3 mg/dL increase | < 0.5 mL/kg/hr for 6–12 hrs |
| 2 | 2.0–2.9× baseline | < 0.5 mL/kg/hr for ≥ 12 hrs |
| 3 | 3.0× baseline OR Cr ≥ 4.0 OR initiation of RRT OR (in < 18 y) eGFR drop < 35 | < 0.3 mL/kg/hr for ≥ 24 hrs OR anuria ≥ 12 hrs |
Historical Criteria: RIFLE & AKIN
Before KDIGO unified the field, two older systems were used; you will still see them in older notes and ICU charts.
| System | Risk / Stage 1 | Injury / Stage 2 | Failure / Stage 3 | Loss | ESRD |
|---|---|---|---|---|---|
| RIFLE | Cr 1.5× or GFR ↓ 25% | Cr 2× or GFR ↓ 50% | Cr 3× or GFR ↓ 75% or Cr ≥ 4 | Need RRT > 4 wk | Need RRT > 3 mo |
| AKIN | Cr ≥ 0.3 or 1.5–2× | Cr 2–3× | Cr > 3× or ≥ 4 + acute 0.5 rise | — | — |
Prerenal AKI
Prerenal AKI is a functional, reversible decline from reduced renal perfusion. The kidneys are structurally intact and simply under-pumped. Causes include true hypovolemia (GI losses, hemorrhage, diuretics, burns), effective hypovolemia (heart failure, cirrhosis, nephrotic syndrome), hypotension/sepsis, renal artery stenosis, and medications that impair autoregulation (NSAIDs constrict the afferent, ACEi/ARBs dilate the efferent). The laboratory signature is avid sodium reabsorption: FENa < 1%, urine Na < 20 mEq/L, urine osmolality > 500, and a BUN:Cr ratio > 20:1. Treatment is restoration of perfusion — crystalloid in true volume depletion, diuretics in congestive heart failure, albumin in cirrhosis, and holding offending drugs.
Postrenal AKI
Obstruction anywhere from the renal pelvis to the urethral meatus. In men, the most common cause is BPH; in women, pelvic malignancy. Bilateral obstruction (or unilateral with a single functioning kidney) is required to raise the creatinine. Bedside bladder scan and renal ultrasound looking for hydronephrosis are the diagnostic mainstays. Treatment is relief of obstruction: Foley catheter for bladder outlet obstruction, percutaneous nephrostomy or ureteral stent for upper tract obstruction. Watch for post-obstructive diuresis after relief.
Intrinsic AKI
Parenchymal damage. Subdivided by the injured compartment: tubular (ATN), interstitial (AIN), glomerular (glomerulonephritis), or vascular (atheroemboli, TMA, scleroderma renal crisis). Urinalysis with microscopy is the single most useful test — muddy brown granular casts suggest ATN, WBC casts with eosinophiluria suggest AIN, RBC casts with dysmorphic RBCs suggest glomerulonephritis.
Acidosis (refractory metabolic), Electrolytes (refractory hyperkalemia), Ingestions (lithium, toxic alcohols, salicylates, metformin-associated lactic acidosis), Overload (refractory volume overload / pulmonary edema), Uremia (pericarditis, encephalopathy, bleeding). Document which criterion is present when the note says "will initiate HD."
04 ATN, AIN & Contrast Nephropathy Acute
Acute Tubular Necrosis (ATN)
ATN is the most common cause of inpatient intrinsic AKI. Prolonged ischemia (essentially prerenal that was not reversed) or nephrotoxins (aminoglycosides, vancomycin, amphotericin, cisplatin, heme pigment from rhabdomyolysis, myeloma light chains) damage the tubular epithelium, which sloughs into the lumen and forms the pathognomonic muddy brown granular casts. Laboratory signature: FENa > 2%, urine Na > 40, urine osmolality ~ 300 (isosthenuric because tubules cannot concentrate), BUN:Cr < 15. ATN follows a classic three-phase course: initiation (insult), maintenance (oliguric, days to weeks), and recovery (often polyuric — watch for hypokalemia and hypomagnesemia during this phase). Management is supportive: remove the offending insult, avoid further nephrotoxins, dose-adjust all medications, maintain euvolemia, and provide RRT if AEIOU criteria are met.
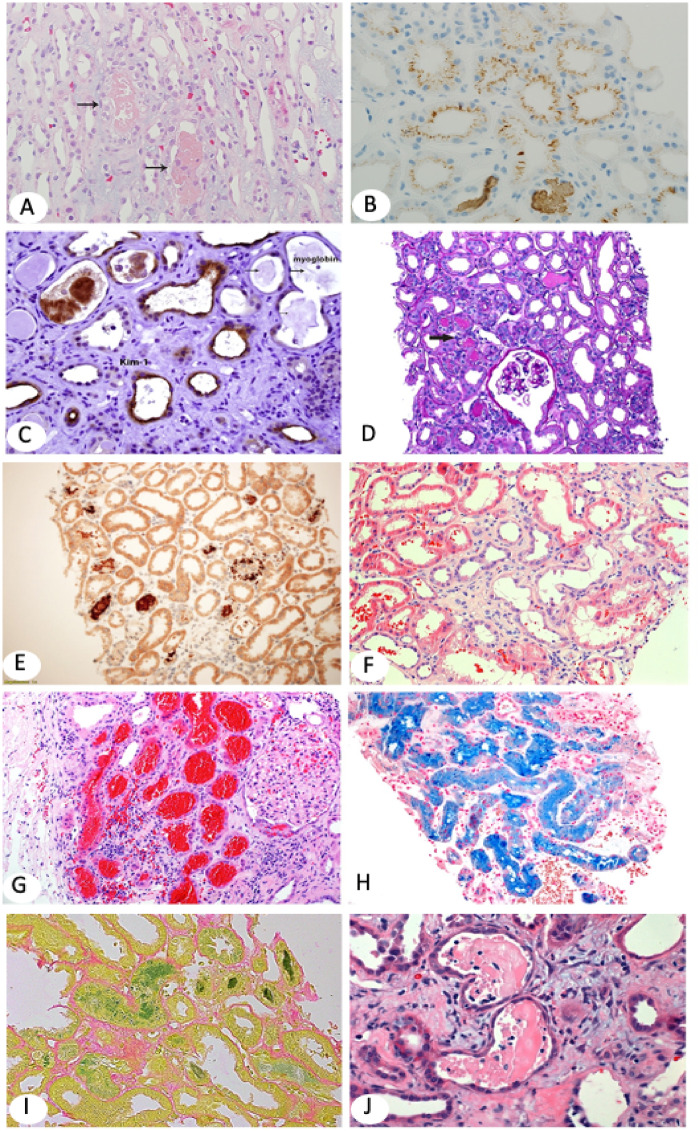
Acute Interstitial Nephritis (AIN)
AIN is a hypersensitivity inflammation of the renal interstitium, classically triggered by drugs: penicillins/cephalosporins, NSAIDs, PPIs, sulfonamides, rifampin, allopurinol, 5-ASA, and more recently immune checkpoint inhibitors. Infections (legionella, leptospira, CMV) and autoimmune disease (sarcoidosis, Sjögren) also cause AIN. The classic triad of fever, rash, and eosinophilia appears in < 10% of cases, so absence does not rule it out. Look for sterile pyuria, white-cell casts, subnephrotic proteinuria, and peripheral eosinophilia. Urine eosinophils are no longer considered sensitive or specific. Kidney biopsy is the gold standard when the diagnosis is not obvious. Treatment is prompt withdrawal of the offending drug; corticosteroids are often given for severe or non-recovering cases.
Contrast-Associated AKI (CA-AKI)
Iodinated contrast can cause AKI via direct tubular toxicity and medullary vasoconstriction, typically presenting as a creatinine rise within 24–48 hours of exposure that peaks at 3–5 days and resolves within 7–10 days. Recent evidence suggests the effect is smaller than historically feared; the current ACR/NKF consensus statement distinguishes contrast-associated AKI (temporal association) from contrast-induced AKI (causally proven) and supports isotonic crystalloid prophylaxis for high-risk patients (eGFR < 30). N-acetylcysteine is no longer recommended.
05 Rhabdomyolysis & Hepatorenal Syndrome Acute
Rhabdomyolysis
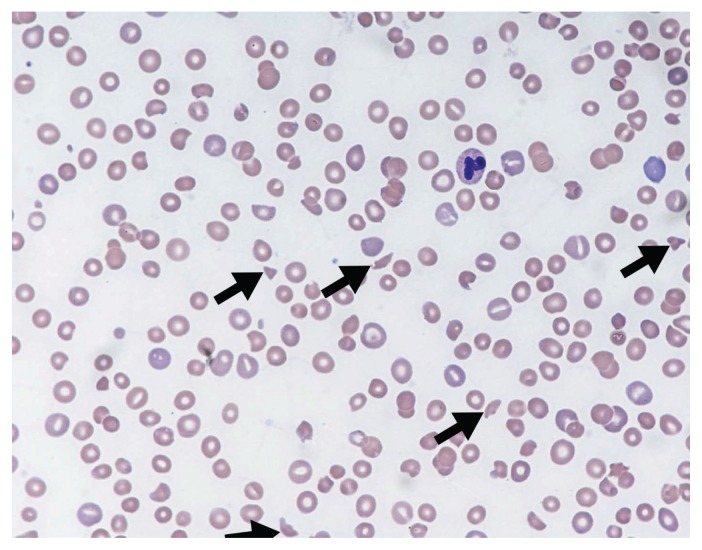
Skeletal muscle breakdown releases myoglobin, which precipitates in the tubules and causes a pigment nephropathy form of ATN. Triggers include crush injury, prolonged immobilization ("long lie"), seizures, extreme exertion, statins/fibrates, cocaine/stimulants, and inherited metabolic myopathies. Presentation is muscle pain, weakness, and tea-colored urine. Laboratory: massively elevated CK (often > 5,000 and frequently > 50,000), hyperkalemia, hyperphosphatemia, hypocalcemia (calcium deposits in damaged muscle), elevated uric acid, elevated LDH, elevated AST, and a UA that is "blood positive" on dipstick but shows no RBCs on microscopy (myoglobin cross-reacts). Treatment is aggressive isotonic fluid resuscitation (target urine output 200–300 mL/hr), correction of electrolytes, and monitoring for compartment syndrome.
Hepatorenal Syndrome (HRS)
HRS is AKI in the setting of advanced cirrhosis driven by splanchnic vasodilation, effective hypovolemia, and intense renal vasoconstriction. The kidneys are structurally normal. Diagnosis requires cirrhosis with ascites, AKI, no improvement after 48 hours of albumin and diuretic withdrawal, absence of shock, no recent nephrotoxins, and no structural kidney disease (bland urine, no or minimal proteinuria). HRS-AKI (formerly Type 1) is rapid and carries a dismal prognosis; HRS-NAKI (formerly Type 2) is slower. Treatment is albumin 1 g/kg/day plus a splanchnic vasoconstrictor: terlipressin (FDA-approved 2022), or norepinephrine (ICU), or midodrine + octreotide (floor). Definitive therapy is liver transplant.
06 Chronic Kidney Disease (CKD) Chronic
CKD is defined by KDIGO 2024 CKD guidelines as abnormalities of kidney structure or function present for > 3 months with implications for health. The diagnosis requires either GFR < 60 mL/min/1.73 m2 OR markers of kidney damage (albuminuria UACR ≥ 30, urine sediment abnormalities, electrolyte abnormalities, histologic abnormalities, structural abnormalities by imaging, or kidney transplant). CKD is staged by both GFR (G1–G5) and albuminuria (A1–A3) — the so-called "heat map" — because albuminuria is an independent driver of progression and cardiovascular risk. USPSTF screening guidance and KDIGO recommend screening high-risk patients (diabetes, hypertension, cardiovascular disease, family history) with eGFR and UACR.
| Stage | eGFR (mL/min/1.73 m2) | Description |
|---|---|---|
| G1 | ≥ 90 | Normal or high (kidney damage required) |
| G2 | 60–89 | Mildly decreased (kidney damage required) |
| G3a | 45–59 | Mildly-to-moderately decreased |
| G3b | 30–44 | Moderately-to-severely decreased |
| G4 | 15–29 | Severely decreased — prepare for RRT |
| G5 | < 15 | Kidney failure (add D if on dialysis, T if transplanted) |
| Stage | UACR (mg/g) | Description |
|---|---|---|
| A1 | < 30 | Normal to mildly increased |
| A2 | 30–300 | Moderately increased (formerly "microalbuminuria") |
| A3 | > 300 | Severely increased (formerly "macroalbuminuria") |
CKD Complications — The "CKD Bundle"
Every CKD progress note addresses the same cluster of problems:
- Hypertension: Target < 120/80 by 2021 KDIGO BP in CKD guideline when tolerated. ACEi or ARB is first line if albuminuria is present.
- Proteinuria: RAAS blockade + SGLT2 inhibitor (dapagliflozin or empagliflozin) independent of diabetes, per the DAPA-CKD and EMPA-KIDNEY trials.
- Anemia of CKD: Decreased EPO production; target Hgb 10–11.5 with ESAs (epoetin alfa, darbepoetin) and iron repletion (IV ferric gluconate, sucrose, or carboxymaltose). Never exceed Hgb 11.5 (increased CV risk).
- CKD-Mineral and Bone Disorder (CKD-MBD): Phosphate retention → FGF-23 rise → calcitriol suppression → secondary hyperparathyroidism → renal osteodystrophy. Manage with dietary phosphate restriction, phosphate binders (sevelamer, lanthanum, calcium acetate, ferric citrate), active vitamin D analogs (calcitriol, paricalcitol), and calcimimetics (cinacalcet, etelcalcetide) when PTH remains high.
- Metabolic acidosis: Treat when HCO3 < 22 with oral sodium bicarbonate.
- Hyperkalemia: Dietary restriction, loop diuretic, potassium binders (patiromer, sodium zirconium cyclosilicate) to enable RAAS blockade continuation.
- Dyslipidemia: Statin for most CKD patients > 50 years.
- Vaccinations: Hepatitis B (double dose), pneumococcal, influenza, COVID-19.
When the attending discusses "RRT preparation" in G4 CKD, document: access planning (AV fistula referral ≥ 6 months before anticipated start), modality education (HD vs PD vs transplant), transplant referral (pre-emptive listing at eGFR < 20), hepatitis B vaccination, and advance care planning.
07 Diabetic & Hypertensive Kidney Disease Chronic
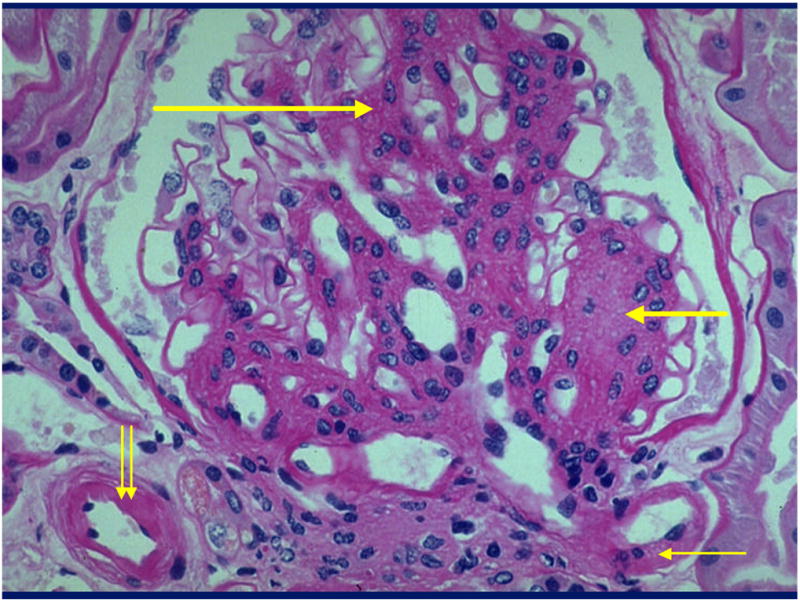
Diabetic Kidney Disease (DKD)
Diabetes is the leading cause of CKD and ESRD in the United States. DKD progresses through predictable stages: hyperfiltration → microalbuminuria (A2) → overt proteinuria (A3) → declining GFR → ESRD, typically over 10–20 years. Pathology shows mesangial expansion, glomerular basement membrane thickening, and in advanced cases nodular glomerulosclerosis (Kimmelstiel-Wilson nodules). Annual screening with UACR and eGFR is standard of care for all diabetic patients. Management is built on the four pillars: (1) glycemic control (A1C individualized 6.5–8%), (2) ADA SGLT2i recommendation for eGFR ≥ 20 regardless of A1C, (3) RAAS blockade (ACEi or ARB) titrated to maximum tolerated dose, and (4) non-steroidal MRA finerenone (Kerendia) for residual albuminuria, based on the FIDELIO-DKD and FIGARO-DKD trials. GLP-1 agonists (semaglutide, tirzepatide) provide additional cardiorenal benefit.
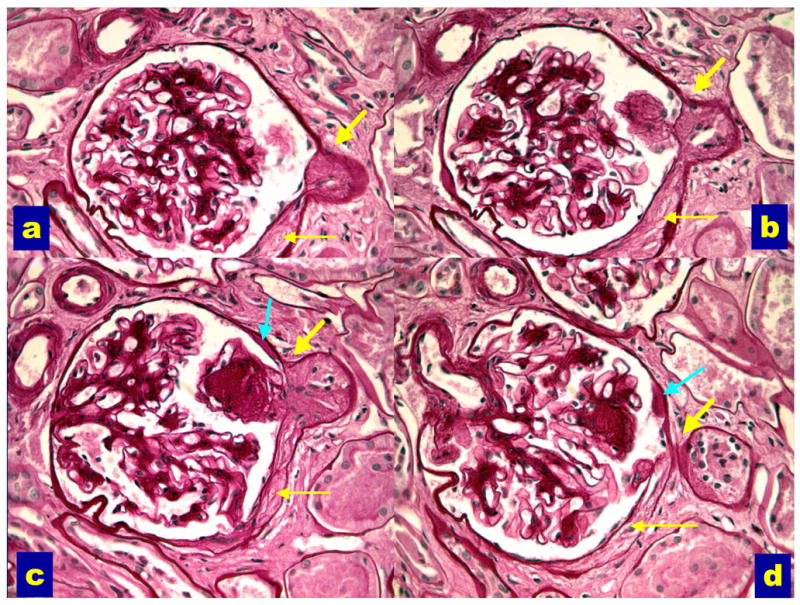
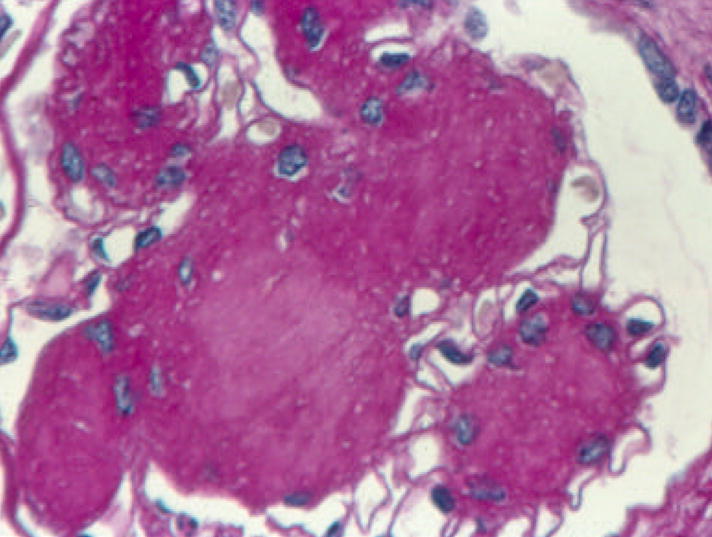
Hypertensive Nephrosclerosis
Long-standing hypertension produces arteriolar hyalinosis and ischemic glomerulosclerosis. It is the second leading cause of ESRD and disproportionately affects Black Americans (associated with APOL1 risk variants). Presentation is slowly progressive CKD with mild proteinuria (usually < 1 g/day), bland urinary sediment, and small echogenic kidneys on ultrasound. Management is aggressive BP control (target < 120/80 per KDIGO) with ACEi/ARB and additional agents as needed. Biopsy is rarely performed unless atypical features are present.
08 Nephrotic Syndrome Glomerular
Nephrotic syndrome is the clinical pentad of heavy proteinuria (> 3.5 g/day), hypoalbuminemia (< 3.0 g/dL), edema, hyperlipidemia, and hypercoagulability (loss of antithrombin III, protein C/S). Patients present with frothy urine, peripheral and periorbital edema, and sometimes acute DVT, renal vein thrombosis, or PE. Kidney biopsy is usually required for definitive diagnosis in adults. The KDIGO 2021 glomerular disease guidelines standardize management.

Minimal Change Disease (MCD): The most common nephrotic syndrome in children. Light microscopy is normal; electron microscopy shows diffuse podocyte foot-process effacement. Highly steroid-responsive. Associated with NSAIDs and Hodgkin lymphoma.
Focal Segmental Glomerulosclerosis (FSGS): The leading cause of primary nephrotic syndrome in adult Black Americans. Segmental sclerosis of some glomeruli. Primary (podocyte disease) vs secondary (HIV, heroin, obesity, reflux, APOL1). Variable steroid response.
Membranous Nephropathy (MN): The leading cause of primary nephrotic syndrome in older White adults. Subepithelial immune deposits produce a thickened GBM with "spike and dome" appearance. ~70% are primary (anti-PLA2R or anti-THSD7A antibodies); the rest are secondary to malignancy, hepatitis B, SLE, or drugs (gold, penicillamine, NSAIDs). High risk of venous thrombosis. Treatment: rituximab or cyclophosphamide + steroids for high-risk disease.
Membranoproliferative GN (MPGN): Now reclassified by pathogenesis into immune-complex-mediated (hepatitis C, SLE, cryoglobulinemia) and complement-mediated (C3 glomerulopathy, dense deposit disease). Often presents with mixed nephrotic/nephritic features.
Secondary Causes
Nephrotic syndrome is frequently a manifestation of a systemic disease. Diabetes is by far the most common cause in adults and usually does not require biopsy when the clinical picture fits. Amyloidosis (AL from plasma cell dyscrasia, AA from chronic inflammation) presents with nephrotic-range proteinuria, apple-green birefringence on Congo red staining, and often cardiac, GI, or neurologic involvement. Other secondary causes include HIV (collapsing FSGS), hepatitis B (membranous), hepatitis C (cryoglobulinemic MPGN), SLE (any class), solid and hematologic malignancies, and drugs (NSAIDs, pamidronate, heroin, interferon).
Management Principles
Every nephrotic patient receives RAAS blockade for proteinuria reduction, loop diuretics (often high-dose, sometimes IV) for edema, statin for hyperlipidemia, dietary sodium restriction, and prophylactic anticoagulation when albumin is profoundly low (< 2.0–2.5) or membranous nephropathy is present. Disease-specific immunosuppression depends on the biopsy diagnosis and risk stratification. Document response by serial UPCR/UACR, serum albumin, weight, and eGFR.
09 Nephritic Syndrome & Glomerulonephritis Glomerular
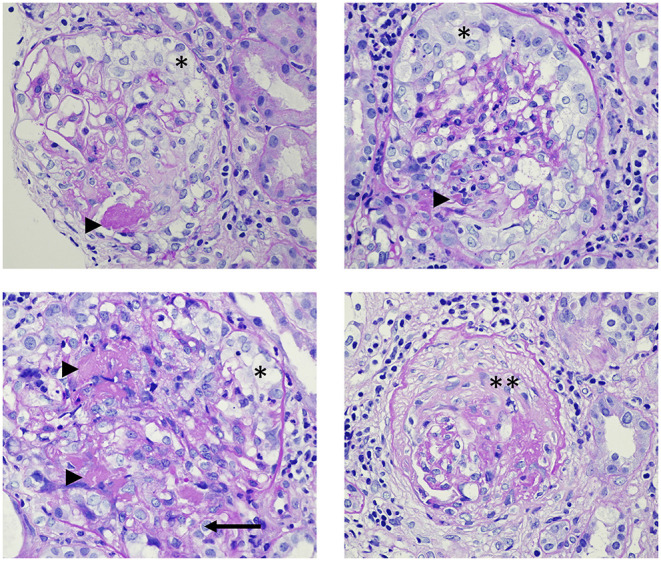
Nephritic syndrome is the classic "active urine" presentation: hematuria with dysmorphic RBCs and RBC casts, subnephrotic proteinuria (usually < 3.5 g/day), hypertension, edema, and rising creatinine. Rapidly progressive glomerulonephritis (RPGN) is nephritic syndrome with doubling of creatinine over days to weeks and crescents on biopsy — a medical emergency requiring urgent biopsy and immunosuppression.
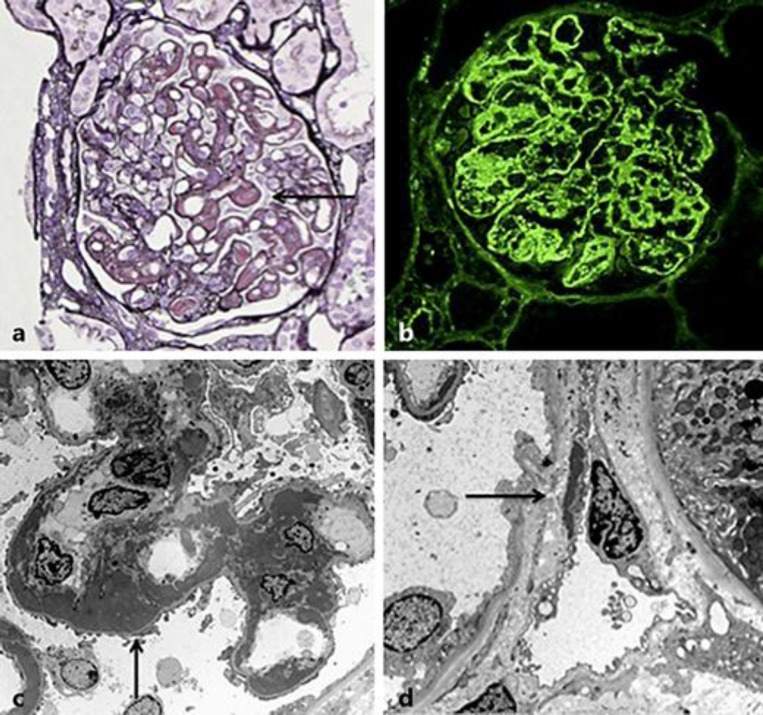
IgA Nephropathy (Berger disease): The most common primary GN worldwide. Classically presents as synpharyngitic gross hematuria (hematuria within 24–48 hours of a URI, in contrast to post-streptococcal GN which occurs 1–3 weeks later). Biopsy shows mesangial IgA deposits. Treatment: RAAS blockade and SGLT2i for all; targeted-release budesonide (Tarpeyo) and steroids/immunosuppression for high-risk disease.
Post-infectious (Post-streptococcal) GN: Follows group A strep pharyngitis or impetigo by 1–3 weeks. Presents with hematuria, edema, hypertension, and AKI. Labs: low C3, normal C4, elevated ASO/anti-DNAse B. Biopsy (rarely done) shows subepithelial "hump" deposits. Usually self-limited; supportive care only.
Anti-GBM Disease (Goodpasture): Autoantibodies against the alpha-3 chain of type IV collagen in the GBM. Linear IgG staining on biopsy. Presents as RPGN ± alveolar hemorrhage. Treatment: plasmapheresis, high-dose steroids, and cyclophosphamide.
ANCA-associated Vasculitis: Granulomatosis with polyangiitis (GPA, c-ANCA/PR3), microscopic polyangiitis (MPA, p-ANCA/MPO), and eosinophilic granulomatosis (EGPA). Pauci-immune crescentic GN on biopsy. Treatment: rituximab or cyclophosphamide + steroids; avacopan is now an approved steroid-sparing adjunct.
Lupus Nephritis: SLE-related GN, classified by ISN/RPS into six classes (I minimal mesangial, II mesangial proliferative, III focal, IV diffuse, V membranous, VI advanced sclerosing). Classes III and IV are the most aggressive and require induction with mycophenolate mofetil or cyclophosphamide + steroids, with voclosporin or belimumab now added per current guidelines.
10 Polycystic Kidney Disease & Inherited Tubulopathies Genetic
Autosomal Dominant PKD (ADPKD)
ADPKD is the most common inherited kidney disease, caused by mutations in PKD1 (~85%, chromosome 16, earlier and more severe) or PKD2 (~15%, chromosome 4, milder). Progressive cyst formation leads to massive kidney enlargement, hypertension by the second to third decade, flank pain, hematuria, stones, UTIs, and ESRD by the fifth to seventh decade. Extra-renal manifestations include hepatic cysts, intracranial berry aneurysms (screen if family history), mitral valve prolapse, and colonic diverticula. Diagnosis is by ultrasound (age-adjusted Ravine criteria) or by genetic testing. Tolvaptan (a vasopressin V2 antagonist) slows progression in rapidly progressive ADPKD but requires liver function monitoring. Autosomal recessive PKD (ARPKD) is a childhood disease with congenital hepatic fibrosis.
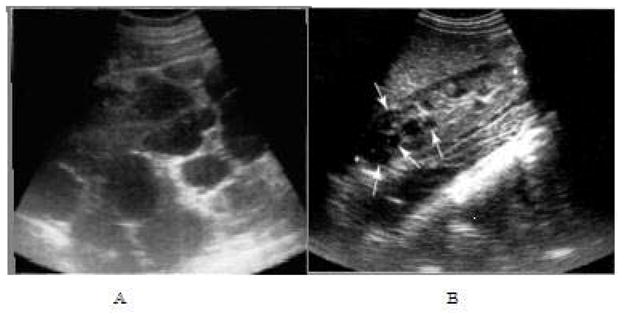
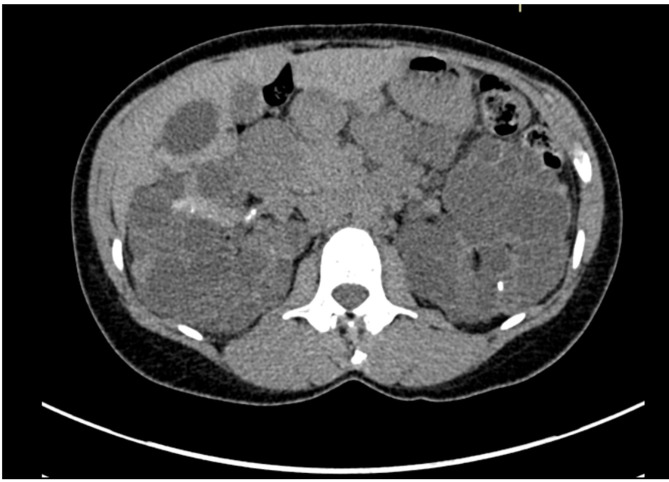
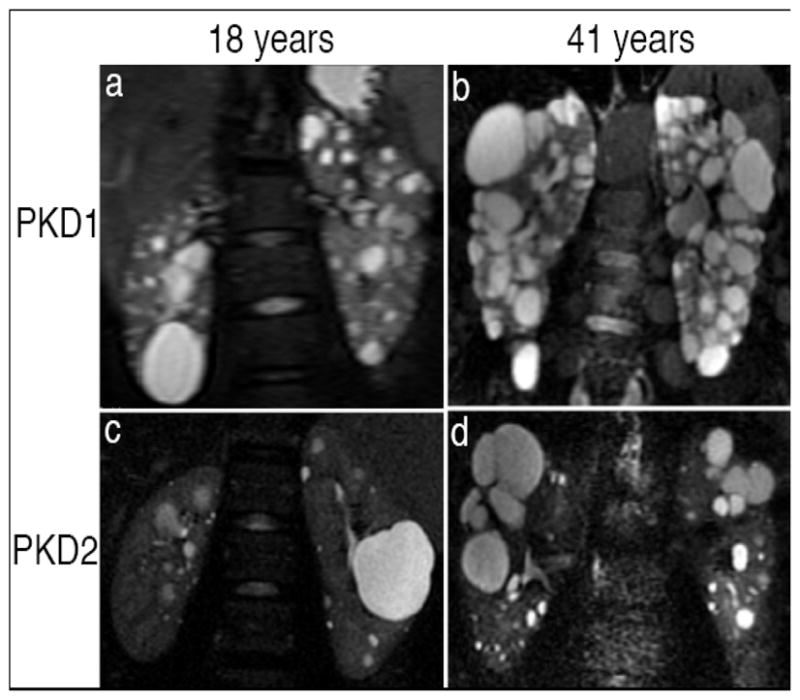
Tuberous Sclerosis & Von Hippel-Lindau
Tuberous sclerosis complex produces renal angiomyolipomas (AMLs) that can bleed catastrophically above 4 cm — managed with mTOR inhibitors (everolimus) or embolization. Von Hippel-Lindau disease carries a lifetime risk of clear-cell renal cell carcinoma and requires surveillance imaging.
Other Hereditary Diseases
Alport syndrome (type IV collagen mutation) causes hereditary nephritis, sensorineural hearing loss, and anterior lenticonus. Fabry disease (X-linked alpha-galactosidase deficiency) causes proteinuric CKD, angiokeratomas, and neuropathic pain. Gitelman (DCT, NCC loss — mimics thiazide) and Bartter (TAL, NKCC2 loss — mimics loop diuretic) syndromes present with hypokalemic metabolic alkalosis.
11 Renal Tubular Acidosis (RTA) Tubular
RTA is a non-anion-gap metabolic acidosis caused by impaired renal acidification. Three classic types plus a fourth, each with a distinct mechanism and signature.
| Type | Defect | Urine pH | Serum K+ | Classic Causes |
|---|---|---|---|---|
| Type 1 (Distal) | α-intercalated cell H+ secretion defect | > 5.5 | Low | Sjögren, SLE, amphotericin, ifosfamide; stones (calcium phosphate) |
| Type 2 (Proximal) | PCT HCO3− reabsorption defect | < 5.5 (initially) | Low | Fanconi syndrome, multiple myeloma, tenofovir, acetazolamide, ifosfamide |
| Type 4 (Hyperkalemic) | Aldosterone deficiency/resistance | < 5.5 | High | Diabetic nephropathy, ACEi/ARB, NSAIDs, heparin, calcineurin inhibitors, Addison disease |
(Type 3 was a mixed distal/proximal form in infants and is no longer used clinically.) Treatment is oral bicarbonate/citrate replacement for Types 1 and 2 (with potassium replacement in Type 1), and fludrocortisone or loop diuretics plus dietary K restriction for Type 4.
Generalized proximal tubule dysfunction producing type 2 RTA plus phosphaturia, glucosuria (with normal serum glucose), aminoaciduria, uricosuria, and bicarbonaturia. Classic causes in adults: multiple myeloma (light-chain proximal tubulopathy), tenofovir, ifosfamide, heavy metals, and Wilson disease. Inherited forms (cystinosis) present in childhood. Workup includes SPEP/UPEP with free light chains and drug history.
12 Sodium & Water Disorders Electrolyte
Hyponatremia (Na < 135)
Hyponatremia is the most common electrolyte disorder in hospitalized patients and is almost always a problem of too much water, not too little sodium. Workup is stepwise: (1) check serum osmolality to confirm hypotonicity (and exclude pseudohyponatremia from hyperlipidemia/hyperproteinemia or translocational hyponatremia from hyperglycemia — correction: add 1.6–2.4 to Na for every 100 mg/dL glucose > 100); (2) assess volume status; (3) check urine osmolality and urine sodium.
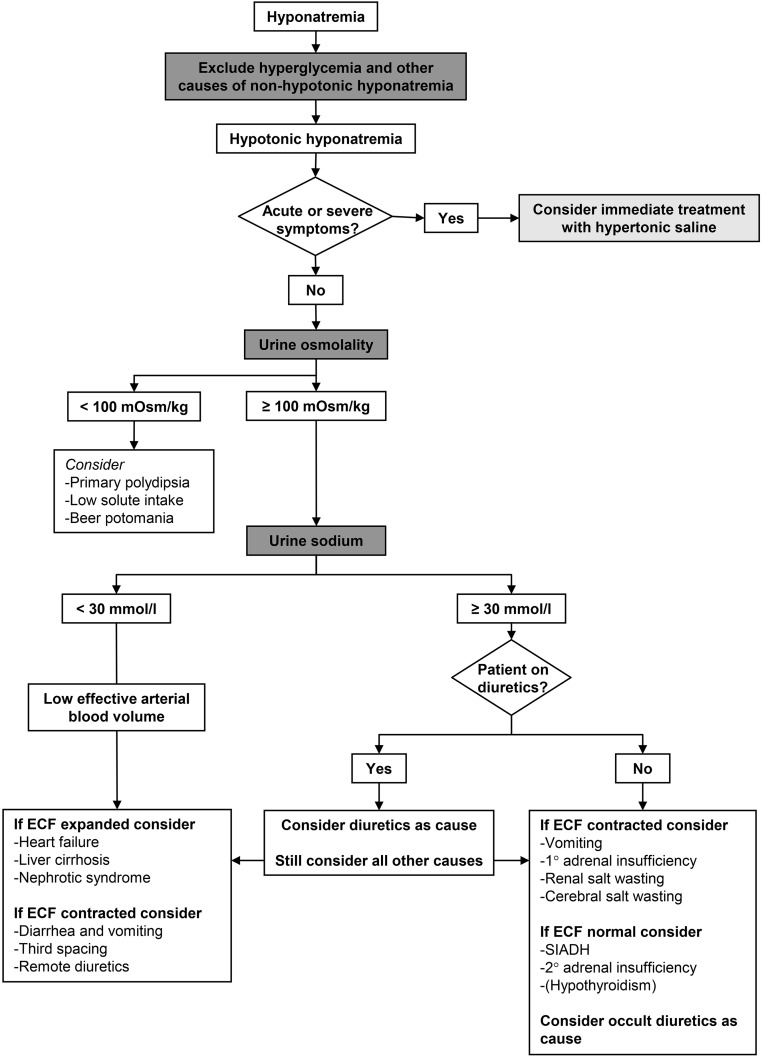
| Volume Status | UNa | Etiologies |
|---|---|---|
| Hypovolemic | < 20 (extrarenal) | GI losses, burns, pancreatitis, third-spacing |
| Hypovolemic | > 20 (renal) | Diuretics (thiazides classic), salt-wasting, adrenal insufficiency, cerebral salt wasting |
| Euvolemic | > 20, Uosm > 100 | SIADH, hypothyroidism, glucocorticoid deficiency |
| Euvolemic | Uosm < 100 | Primary polydipsia, beer potomania, tea-and-toast |
| Hypervolemic | < 20 | CHF, cirrhosis, nephrotic syndrome |
| Hypervolemic | > 20 | AKI/CKD (renal failure) |
Chronic hyponatremia (> 48 hours) must be corrected slowly: ≤ 8–10 mEq/L in 24 hours and ≤ 18 mEq/L in 48 hours. Over-rapid correction causes osmotic demyelination syndrome (ODS) — central pontine myelinolysis. For acute symptomatic hyponatremia (seizures, coma), give 3% hypertonic saline 100 mL bolus × up to 3 with close monitoring. DDAVP clamp + D5W is used to re-lower Na when correction overshoots.
Hypernatremia (Na > 145)
Hypernatremia is a water deficit. Causes include inadequate intake (bedbound elderly, altered mental status), insensible losses (fever, burns), GI losses (osmotic diarrhea), and diabetes insipidus (central vs nephrogenic — distinguished by response to DDAVP). Central DI responds to lithium discontinuation, trauma, or surgery; nephrogenic DI is caused by lithium, hypercalcemia, hypokalemia, or genetic V2 receptor defects. Treat with free water replacement (oral or D5W), correcting ≤ 10–12 mEq/L per 24 hours to avoid cerebral edema. Calculate the free water deficit: TBW × ((Na/140) − 1).
13 Potassium, Calcium, Magnesium & Phosphate Electrolyte
Hyperkalemia (K > 5.0)
Causes: pseudohyperkalemia (hemolyzed sample, fist clenching), transcellular shifts (acidosis, insulin deficiency, beta-blockers, succinylcholine, tumor lysis, rhabdomyolysis), decreased renal excretion (AKI, CKD, ACEi/ARB, MRA, NSAIDs, trimethoprim, heparin, calcineurin inhibitors, type 4 RTA), and increased intake. EKG progression: peaked T waves → PR prolongation → loss of P wave → widened QRS → sine wave → VF/asystole. Treatment sequence: (1) Stabilize the membrane with IV calcium gluconate or calcium chloride; (2) Shift K into cells with insulin + D50, beta-2 agonist (albuterol), and IV sodium bicarbonate if acidotic; (3) Remove K from the body with loop diuretics, potassium binders (patiromer, sodium zirconium cyclosilicate, SPS), and emergent hemodialysis if refractory or in ESRD.
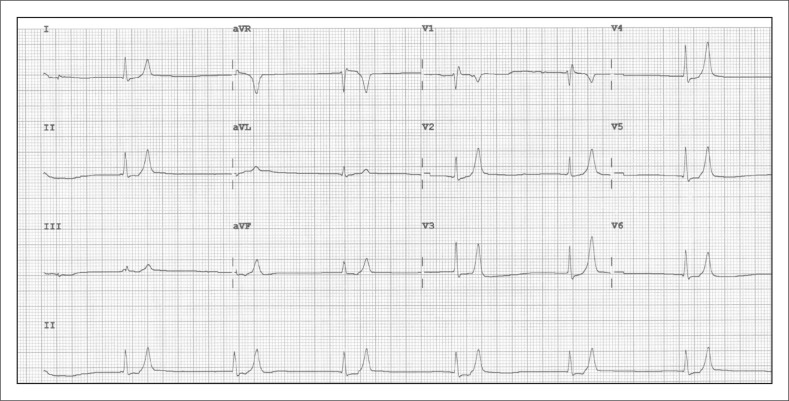
Hypokalemia (K < 3.5)
Causes: GI losses (diarrhea, laxative abuse), renal losses (loop and thiazide diuretics, hyperaldosteronism, Bartter/Gitelman, RTA 1 and 2, hypomagnesemia), transcellular shift (insulin, beta-2 agonists, alkalosis, refeeding). EKG: U waves, flattened T waves, ST depression, arrhythmias. Replace IV at ≤ 10–20 mEq/hr peripherally (higher via central line with monitoring). Always replace magnesium simultaneously — you cannot fix hypokalemia in the setting of hypomagnesemia.
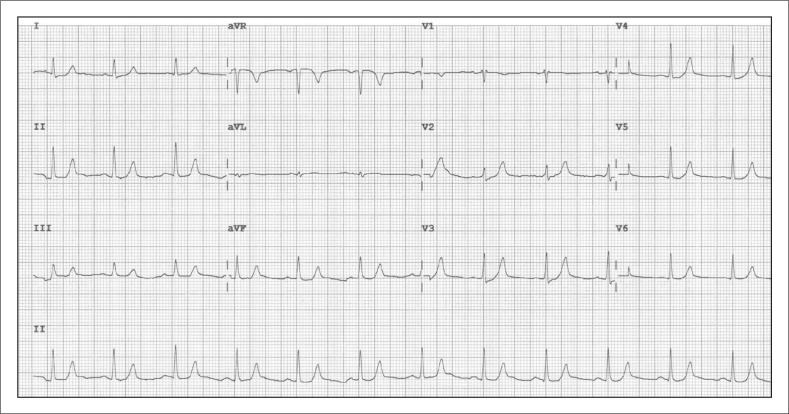
Calcium Disorders
Hypercalcemia: The two dominant causes are primary hyperparathyroidism (outpatient, mild, chronic) and malignancy (inpatient, severe, acute — PTHrP in squamous cell carcinoma, bone metastases, multiple myeloma, activated vitamin D in lymphoma). Other causes: granulomatous disease (sarcoid, TB), thiazides, vitamin D toxicity, milk-alkali syndrome, lithium, hyperthyroidism, immobilization. Symptoms: "stones, bones, groans, and psychiatric overtones" — nephrolithiasis, osteopenia, abdominal pain and constipation, altered mental status. Treatment: aggressive IV saline, loop diuretic once euvolemic, IV bisphosphonate (zoledronic acid) or denosumab, calcitonin (fast but tachyphylaxis), and hemodialysis for refractory cases. Always correct for albumin (add 0.8 for every 1 g/dL below 4.0) or measure ionized calcium.
Hypocalcemia: Causes include hypoparathyroidism (post-thyroidectomy, autoimmune), vitamin D deficiency, CKD, pancreatitis, hungry bone syndrome, hyperphosphatemia (tumor lysis, rhabdo), and sepsis. Symptoms: perioral numbness, tetany, Chvostek sign, Trousseau sign, QT prolongation, seizures. Treatment: oral or IV calcium (calcium gluconate IV) plus vitamin D.
Magnesium & Phosphate
Hypomagnesemia: Caused by GI and renal losses (PPIs are a major under-recognized cause, along with loop and thiazide diuretics, aminoglycosides, cisplatin, amphotericin, alcoholism). Produces refractory hypokalemia and hypocalcemia. Replace with IV MgSO4.
Hypophosphatemia: Refeeding syndrome, DKA recovery, alcoholism, Fanconi syndrome. Severe (< 1.0) can cause respiratory muscle weakness and rhabdomyolysis. Replace with oral or IV potassium/sodium phosphate.
Hyperphosphatemia: CKD/ESRD, tumor lysis, rhabdo. Treated with dietary restriction and phosphate binders.
14 Acid-Base Disorders Electrolyte
Every nephrology consult includes an acid-base assessment. Use a stepwise approach:
Step 1 — pH: Acidemia (< 7.35) or alkalemia (> 7.45)?
Step 2 — Primary disorder: If HCO3− moves in the same direction as the pH, it is metabolic; if PaCO2 moves opposite the pH, it is respiratory.
Step 3 — Anion gap (Na − Cl − HCO3; normal 8–12). Correct for albumin: add 2.5 to AG for every 1 g/dL below 4.0.
Step 4 — Expected compensation:
- Metabolic acidosis (Winter's): PaCO2 = 1.5(HCO3) + 8 ± 2
- Metabolic alkalosis: PaCO2 rises 0.7 mmHg per 1 mEq HCO3 rise
- Acute respiratory acidosis: HCO3 rises 1 per 10 PaCO2 rise
- Chronic respiratory acidosis: HCO3 rises 4 per 10 PaCO2 rise
- Acute respiratory alkalosis: HCO3 falls 2 per 10 PaCO2 fall
- Chronic respiratory alkalosis: HCO3 falls 4–5 per 10 PaCO2 fall
Step 5 — Delta-delta (ΔAG / ΔHCO3): < 1 suggests mixed non-gap acidosis; > 2 suggests concurrent metabolic alkalosis.
High Anion Gap Metabolic Acidosis (HAGMA)
Classic mnemonic GOLD MARK: Glycols (ethylene, propylene), Oxoproline (chronic acetaminophen), L-lactate, D-lactate, Methanol, Aspirin, Renal failure (uremia), Ketoacidosis (DKA, AKA, starvation).
Non-Gap (Hyperchloremic) Metabolic Acidosis
Mnemonic HARDUPS: Hyperalimentation, Addison disease, Renal tubular acidosis, Diarrhea, Ureteroenteric fistula, Pancreatic fistula, Saline (large-volume NS resuscitation). The urine anion gap (UNa + UK − UCl) helps distinguish: negative → GI losses (NH4+ excretion intact); positive → RTA (impaired NH4+ excretion).
Metabolic Alkalosis
Divided by chloride-responsiveness. Chloride-responsive (UCl < 20): vomiting, NG suction, diuretics (after the acute effect), post-hypercapnia, cystic fibrosis. Treated with volume and chloride repletion (normal saline, KCl). Chloride-resistant (UCl > 20): primary hyperaldosteronism, Cushing syndrome, Liddle syndrome, Bartter, Gitelman, severe hypokalemia, and exogenous mineralocorticoid excess (licorice). Treated by addressing the underlying driver.
Respiratory Disorders — Mentioned for Completeness
Respiratory acidosis results from hypoventilation (COPD exacerbation, neuromuscular disease, opioid overdose, OSA/obesity hypoventilation). Respiratory alkalosis results from hyperventilation (anxiety, pain, fever, sepsis, pulmonary embolism, salicylate toxicity — which classically produces a mixed respiratory alkalosis and anion-gap metabolic acidosis, a high-yield pattern). Always confirm acute vs chronic using the compensation formulas above — the chronicity dictates whether you are seeing full compensation or a true mixed disorder.
15 Nephrolithiasis (Kidney Stones) Urologic
Kidney stones affect ~10% of the US population. Presentation is sudden-onset unilateral flank pain radiating to the groin ("renal colic"), often with hematuria, nausea, and vomiting. Non-contrast CT of the abdomen/pelvis is the gold standard imaging; ultrasound is used in pregnancy.
| Stone Type | Frequency | Radiolucency | Urine pH | Causes / Risk | Prevention |
|---|---|---|---|---|---|
| Calcium oxalate | ~75% | Radiopaque | Any | Hypercalciuria, hyperoxaluria, hypocitraturia, low fluid intake | Hydration, thiazide, citrate, low-sodium/normal-calcium diet |
| Calcium phosphate | ~10% | Radiopaque | Alkaline (> 6.5) | Distal RTA (Type 1), primary hyperparathyroidism | Thiazide, citrate, treat underlying cause |
| Uric acid | ~10% | Radiolucent (seen only on CT) | Acidic (< 5.5) | Gout, tumor lysis, chronic diarrhea, diabetes/metabolic syndrome | Alkalinize urine with K-citrate, allopurinol, hydration |
| Struvite (Mg-NH4-phosphate) | ~5% | Radiopaque | Alkaline (> 7) | Urease-producing organisms (Proteus, Klebsiella, Pseudomonas); staghorn calculi | Complete stone removal + antibiotics |
| Cystine | < 1% | Faintly opaque | Acidic | Cystinuria (autosomal recessive) | High fluid intake, alkalinization, tiopronin |
Management of acute colic: IV fluids, NSAIDs (ketorolac, if renal function allows) ± opioids, antiemetics, and alpha-blocker (tamsulosin) for distal ureteral stones ≤ 10 mm. Stones < 5 mm usually pass spontaneously; 5–10 mm have variable passage rates; > 10 mm almost always require intervention (ureteroscopy with laser lithotripsy, ESWL, or PCNL for staghorns). Red flags requiring urgent urology: obstruction with infection (emergent decompression), AKI, solitary kidney, or intractable symptoms.
16 UTI, Pyelonephritis & Renal Artery Stenosis Mixed
UTI & Pyelonephritis
Cystitis presents with dysuria, frequency, urgency, and suprapubic discomfort without fever. Pyelonephritis adds fever, chills, flank pain, costovertebral angle tenderness, and often nausea/vomiting. E. coli causes ~80% of community-acquired cases. Complicated UTI (men, pregnancy, CKD, stones, catheters, immunosuppression, transplant) requires broader coverage and longer duration. Workup: UA with culture, CBC, BMP, and CT in complicated cases to assess for abscess, emphysematous pyelonephritis, or obstruction. Outpatient pyelonephritis is treated with fluoroquinolone or TMP-SMX (local resistance permitting) for 7–14 days; inpatient cases receive IV ceftriaxone, piperacillin-tazobactam, or a carbapenem.
Emphysematous & Complicated Pyelonephritis
Emphysematous pyelonephritis is a necrotizing infection with gas within the renal parenchyma, seen almost exclusively in poorly controlled diabetics and urinary obstruction; gas on CT is pathognomonic. Management is emergency broad-spectrum antibiotics, relief of obstruction, and often percutaneous drainage or nephrectomy. Renal and perinephric abscess requires drainage plus prolonged antibiotics. Xanthogranulomatous pyelonephritis is a chronic destructive form associated with obstruction and Proteus infection — surgical nephrectomy is usually required.
Renal Artery Stenosis (RAS)
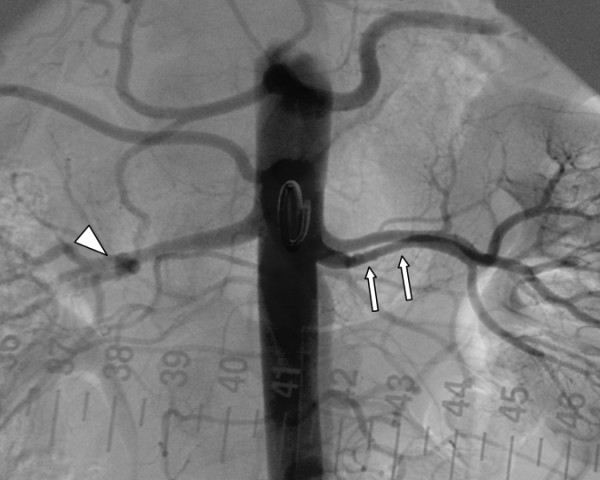
RAS is unilateral or bilateral narrowing of the renal artery, most commonly from atherosclerosis (older patients, CV disease) or fibromuscular dysplasia (young women, "string-of-beads" appearance). Presentation: resistant hypertension, recurrent flash pulmonary edema (bilateral RAS), sudden increase in creatinine after starting ACEi/ARB, unilateral small kidney, abdominal bruit. Diagnosis: duplex ultrasound (peak systolic velocity > 200 cm/s), CTA, or MRA. The CORAL trial showed no benefit of stenting over medical therapy for atherosclerotic RAS in most patients; intervention is reserved for refractory cases, flash pulmonary edema, or FMD (which responds well to angioplasty).
17 Dialysis — HD, PD & Access Dialysis
Hemodialysis (HD)
Standard in-center HD runs 3× per week for 3–4 hours per session. Blood is pumped from the patient through a dialyzer (semi-permeable membrane) where solute diffuses down its gradient into dialysate flowing counter-current, and excess water is removed by ultrafiltration (hydrostatic pressure gradient). Prescription elements documented in every HD note: dialyzer type and surface area, blood flow rate (Qb, typically 300–450 mL/min), dialysate flow rate (Qd, typically 500–800 mL/min), dialysate composition (Na, K, Ca, bicarbonate baths), treatment duration, ultrafiltration goal (target weight), heparin dose, and access used.
Adequacy is assessed by Kt/V (target ≥ 1.2 per session) and urea reduction ratio (URR ≥ 65%). Common intra-dialytic complications: hypotension (most common — excessive UF, cardiac dysfunction), cramps, nausea, dialysis disequilibrium syndrome (rapid urea shifts in new patients), first-use reactions, air embolism, and arrhythmia. Home HD and nocturnal HD are increasingly offered options.
The dialysate potassium concentration is chosen to match the patient's physiology. A 2K bath is standard for most stable HD patients; 3K or 4K baths are used when pre-dialysis K is near-normal or when arrhythmia risk from rapid K shifts is a concern (CAD, digoxin, long inter-dialytic interval on Monday); 1K or 0K baths are used only for severe hyperkalemia under close monitoring and are associated with sudden cardiac death risk.
Peritoneal Dialysis (PD)
PD uses the patient's peritoneal membrane as the dialysis barrier. Dialysate is instilled through a Tenckhoff catheter, dwells for hours, then drains. Two modalities: CAPD (continuous ambulatory, 4–5 manual exchanges per day) and APD (automated, cycler overnight). Advantages: home-based, continuous, preserves residual kidney function, hemodynamically gentle. Disadvantages: peritonitis (the big one — classic presentation is cloudy effluent with WBC > 100 and > 50% neutrophils), exit-site/tunnel infection, mechanical catheter problems, loss of ultrafiltration over time, and encapsulating peritoneal sclerosis (rare, devastating).
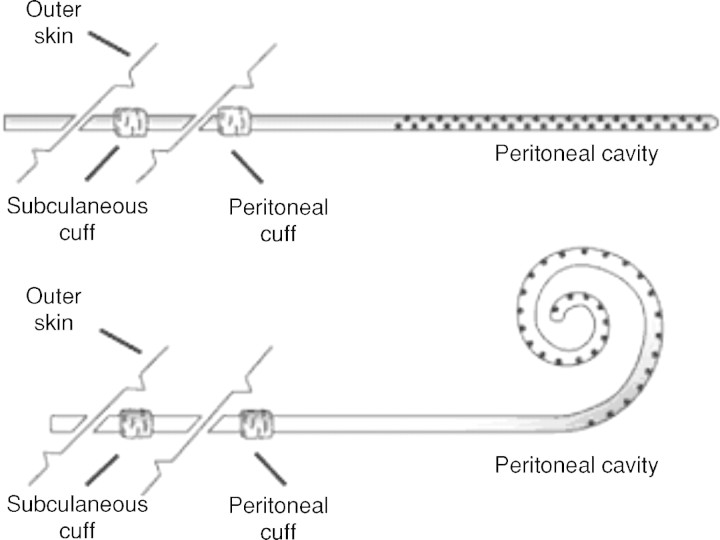
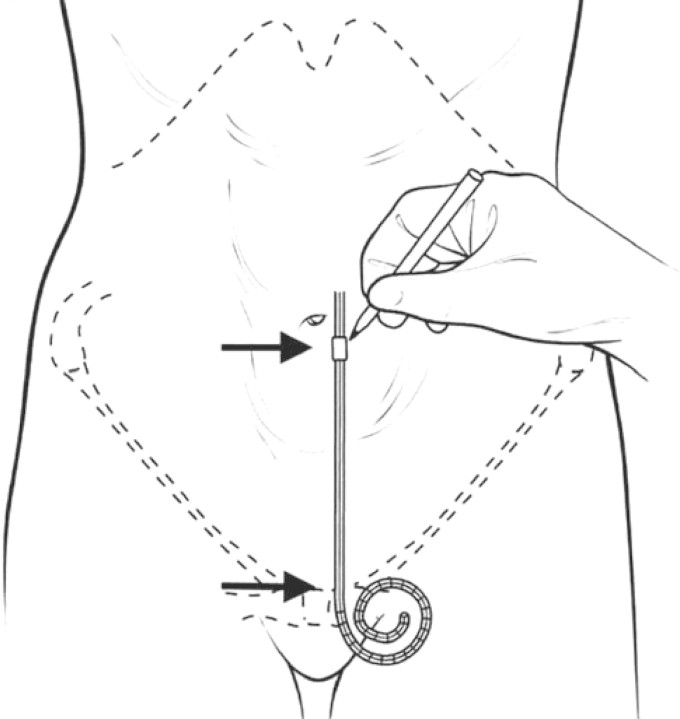
Vascular Access
The KDOQI access hierarchy prioritizes AV fistula (native vein-to-artery, preferred, best patency and lowest infection) > AV graft (PTFE or biologic conduit when veins inadequate) > tunneled dialysis catheter (last resort due to infection and central stenosis risk). Common fistula configurations: radiocephalic (Brescia-Cimino, wrist), brachiocephalic, and brachiobasilic transposition. Document: side, configuration, maturity, thrill/bruit, signs of stenosis (pulsatile, high venous pressures), steal syndrome (distal ischemia), aneurysm, infection, and last intervention (angioplasty, thrombectomy).
18 Kidney Transplant Management Transplant
Kidney transplant is the definitive treatment for ESRD, offering superior survival and quality of life compared with dialysis. Sources: deceased donor (standard criteria, expanded criteria, donation after cardiac death) and living donor (related or unrelated). The graft is typically placed extraperitoneally in the iliac fossa with anastomosis to the external iliac vessels and the ureter to the bladder. The native kidneys are almost always left in place.
Induction & Maintenance Immunosuppression
Induction: basiliximab (IL-2R antagonist) for low-risk or rabbit antithymocyte globulin (rATG) for high-risk recipients. Maintenance "triple therapy": calcineurin inhibitor (tacrolimus >> cyclosporine) + antimetabolite (mycophenolate mofetil or mycophenolic acid) + corticosteroid (prednisone, often tapered or withdrawn). mTOR inhibitors (sirolimus, everolimus) are alternatives in patients with CNI toxicity or certain malignancies. KDIGO transplant guidelines guide management.
Rejection
Hyperacute (minutes, preformed antibodies, now rare due to crossmatching), acute cellular (days to months, T-cell mediated, treated with steroid pulse ± rATG), acute antibody-mediated (donor-specific antibody, C4d deposition on biopsy, treated with plasmapheresis, IVIG, rituximab), and chronic (months to years, interstitial fibrosis/tubular atrophy, transplant glomerulopathy). Any rise in creatinine triggers workup: tacrolimus level, BK virus PCR, CMV PCR, urine studies, renal ultrasound with Dopplers, and often allograft biopsy.
Early Postoperative Course
Immediate graft function is defined by a brisk urine output and rapid creatinine fall. Delayed graft function (DGF) — requirement for dialysis within the first week — is more common with deceased-donor and expanded-criteria grafts and usually represents ischemia-reperfusion injury (ATN of the graft). Slow graft function is a partial form. Surgical complications to document include lymphocele, urine leak or ureteral stenosis, renal artery/vein thrombosis (emergency), and wound issues.
Transplant Complications & Monitoring
Infectious: CMV, BK polyomavirus (BK viremia → BK nephropathy), EBV/PTLD, pneumocystis (PJP prophylaxis with TMP-SMX), fungal, community infections. Malignancy: increased risk of skin cancer, PTLD, Kaposi, cervical. Cardiovascular disease remains the leading cause of death with a functioning graft. Recurrence of primary disease (FSGS, MPGN, IgA) is possible. Every transplant clinic note captures: days post-transplant, current immunosuppression (drug, dose, trough level), creatinine trend, BK/CMV status, infection/malignancy surveillance, BP, glycemic control (new-onset diabetes after transplant), and adherence.
19 Nephrology Procedures Procedural
| Procedure | Purpose | Key Documentation |
|---|---|---|
| Kidney biopsy (native) | Diagnose GN, AKI of unclear etiology, proteinuria | Indication, coagulation clearance, pre-procedure imaging, side, needle gauge, number of cores, light microscopy/IF/EM cores, post-procedure monitoring for bleeding |
| Kidney biopsy (allograft) | Evaluate graft dysfunction, rejection, BK, recurrence | Same as above plus indication (protocol vs for-cause) and immunosuppression regimen |
| AV fistula creation | Permanent HD access | Side, configuration (RC, BC, BB), vein mapping, expected maturation 6–12 weeks |
| AV fistula/graft angioplasty (fistulogram) | Treat access stenosis | Location and severity of stenosis, balloon size, result, residual stenosis, complications |
| Tunneled hemodialysis catheter | Temporary or bridge HD access | Side, vein (right IJ preferred), tip position at cavoatrial junction, cuff location, dressing |
| Temporary (non-tunneled) HD catheter | Urgent inpatient HD | Access site, length, confirmation of placement |
| Peritoneal dialysis catheter placement | Initiate PD | Surgical vs laparoscopic, tunnel and exit site location, break-in period 2 weeks |
| Plasmapheresis (TPE) | Anti-GBM, ANCA with severe renal disease or DAH, TTP, antibody-mediated rejection | Number of sessions, replacement fluid (albumin vs FFP), vascular access |
| CRRT (CVVH, CVVHD, CVVHDF) | Slow continuous RRT in hemodynamically unstable ICU patients | Modality, blood flow, dialysate/replacement rate, anticoagulation (citrate vs heparin), net UF/hr |
| Renal ultrasound with Dopplers | Size, echogenicity, hydronephrosis, RI, transplant perfusion | Kidney size, cortical thickness, hydro grade, RI values, stone/cyst presence |
Dialysis Prescription Elements
Every inpatient HD order specifies: treatment duration, target ultrafiltration (by dry weight), dialyzer type, Qb, Qd, dialysate K bath (1, 2, 3, or 4 mEq/L), dialysate Ca bath (2.5 or 3.0 mEq/L), dialysate Na (often 138–140), bicarbonate bath, heparin dose (loading and maintenance, or "tight heparin" / heparin-free), access, and any medications to give on dialysis (IV iron, ESA, vitamin D analog, antibiotics).
20 Labs, Urinalysis & Imaging Diagnostics
Urinalysis & Sediment
The urinalysis is the nephrologist's EKG. Dipstick captures pH, specific gravity, blood, protein, glucose, ketones, leukocyte esterase, nitrite, bilirubin, and urobilinogen. Microscopy captures RBCs (dysmorphic suggests glomerular origin), WBCs, epithelial cells, and the critical casts:
| Cast | Meaning |
|---|---|
| Hyaline | Normal, dehydration, exercise |
| Muddy brown / granular | ATN (pathognomonic) |
| RBC casts | Glomerulonephritis |
| WBC casts | AIN, pyelonephritis |
| Fatty / oval fat bodies (Maltese cross) | Nephrotic syndrome |
| Waxy / broad | Advanced CKD |
Proteinuria Quantification
24-hour urine collection is the historical gold standard but is prone to collection error. The spot urine protein-to-creatinine ratio (UPCR) and urine albumin-to-creatinine ratio (UACR) are more practical and correlate well with 24-hour values. UACR is preferred for CKD staging and monitoring. Nephrotic-range proteinuria is UPCR > 3.5 g/g or UACR > 2,200 mg/g.
FENa & FEUrea
FENa = (UNa × PCr) / (PNa × UCr) × 100. < 1% suggests prerenal or early glomerulonephritis; > 2% suggests ATN. FENa is unreliable in patients on diuretics — use FEUrea instead (< 35% is prerenal).
Serum Chemistries — The Basic Metabolic Panel
Every nephrology note is built on the BMP. Normal reference ranges vary slightly by lab, but the values a scribe must know cold are: Na 135–145, K 3.5–5.0, Cl 98–108, HCO3 22–28, BUN 8–20, Cr 0.6–1.2 (varies by sex and muscle mass), glucose 70–99 fasting, Ca 8.5–10.5, Mg 1.7–2.2, Phos 2.5–4.5, albumin 3.5–5.0. Critical values that trigger immediate action: K < 3.0 or > 6.0, Na < 120 or > 160, Ca < 7.0 or > 12.0, Mg < 1.0, Phos < 1.0, HCO3 < 15.
Imaging
Renal ultrasound is the first-line imaging for AKI/CKD: assesses size (small < 9 cm suggests chronic disease), echogenicity (increased in CKD), hydronephrosis (obstruction), cysts, masses, and stones. CT without contrast is gold standard for stones. CT with contrast or CTA evaluates masses and vascular anatomy (use cautiously in CKD). MRA evaluates renal arteries; avoid gadolinium in eGFR < 30 (nephrogenic systemic fibrosis risk with older agents; newer group II agents are considered safer). Nuclear renal scan (MAG3, DTPA) assesses differential function, obstruction, and transplant perfusion.
21 Medications You Must Know Medications
| Class | Generic (Brand) | Mechanism / Use | Key Notes |
|---|---|---|---|
| ACE inhibitors | lisinopril, enalapril, ramipril, benazepril | Block ACE → reduce AT-II → dilate efferent → reduce proteinuria | Cough, hyperkalemia, AKI, angioedema; hold in AKI or K > 5.5 |
| ARBs | losartan, valsartan, irbesartan, olmesartan | AT-II receptor blockade; same indications | No cough; same metabolic risks |
| Non-steroidal MRA | finerenone (Kerendia) | DKD with albuminuria on max ACEi/ARB | Monitor K; FIDELIO/FIGARO trials |
| SGLT2 inhibitors | dapagliflozin (Farxiga), empagliflozin (Jardiance), canagliflozin | Block proximal glucose/Na reabsorption; cardiorenal protection | Use regardless of DM per DAPA-CKD/EMPA-KIDNEY; euglycemic DKA risk |
| Loop diuretics | furosemide (Lasix), torsemide, bumetanide, ethacrynic acid | Block NKCC2 in TAL; potent diuresis, work at low GFR | Hypokalemia, hypomagnesemia, ototoxicity; torsemide has better PO bioavailability |
| Thiazides | hydrochlorothiazide, chlorthalidone, metolazone, indapamide | Block NCC in DCT | Hyponatremia, hypokalemia, hypercalcemia, hyperuricemia; weak at eGFR < 30 except metolazone |
| K-sparing / MRA | spironolactone, eplerenone, amiloride, triamterene | Aldosterone antagonism or ENaC blockade | Hyperkalemia; spironolactone causes gynecomastia |
| Phosphate binders | sevelamer (Renvela), lanthanum (Fosrenol), calcium acetate (PhosLo), ferric citrate (Auryxia), sucroferric oxyhydroxide (Velphoro) | Bind dietary phosphate in gut | Take with meals; Ca-based in low PTH only |
| Active vitamin D | calcitriol, paricalcitol, doxercalciferol | Suppress PTH in CKD-MBD | Monitor Ca and phosphate |
| Calcimimetics | cinacalcet (Sensipar, PO), etelcalcetide (Parsabiv, IV) | CaSR activation → suppress PTH | Hypocalcemia, nausea |
| ESAs | epoetin alfa (Epogen, Procrit), darbepoetin alfa (Aranesp) | Anemia of CKD | Target Hgb 10–11.5; CV risk if higher |
| IV iron | iron sucrose (Venofer), ferric gluconate (Ferrlecit), ferumoxytol, ferric carboxymaltose (Injectafer), ferric derisomaltose | Iron repletion in CKD/HD | Infusion reactions |
| K binders | patiromer (Veltassa), sodium zirconium cyclosilicate (Lokelma), SPS (Kayexalate) | Chronic hyperkalemia | Enables RAAS blockade; Kayexalate colonic necrosis risk |
| Vasopressin antagonist | tolvaptan (Samsca/Jynarque) | Euvolemic/hypervolemic hyponatremia; ADPKD progression | LFT monitoring in ADPKD; over-rapid Na rise risk |
| Alkali therapy | sodium bicarbonate, potassium citrate (Urocit-K) | Metabolic acidosis; stone prevention | Dose by HCO3 target |
| Calcineurin inhibitors | tacrolimus (Prograf), cyclosporine (Neoral) | Transplant maintenance | Nephrotoxic, neurotoxic, hyperkalemia, glucose intolerance, hypomagnesemia; trough monitoring |
| Antimetabolites | mycophenolate mofetil (CellCept), mycophenolic acid (Myfortic), azathioprine | Transplant maintenance; lupus nephritis | GI side effects, cytopenias, teratogenic (MMF) |
| mTOR inhibitors | sirolimus (Rapamune), everolimus (Zortress) | Transplant alternative | Impaired wound healing, proteinuria, mouth ulcers, hyperlipidemia |
| Induction biologics | basiliximab (Simulect), rabbit ATG (Thymoglobulin), alemtuzumab | Transplant induction | Infusion reactions, profound lymphocyte depletion with rATG |
| Steroids | prednisone, methylprednisolone (Solu-Medrol) | GN, rejection, AIN | Metabolic/infectious toxicity |
22 Physical Exam — The Renal Exam
The nephrology exam is dominated by volume assessment. Every patient, every visit, every note.
General: Alert, oriented; asterixis absent/present; mental status; uremic fetor.
Vitals/Weight: BP seated, orthostatics if indicated; weight vs dry weight (dialysis) or vs prior visit.
HEENT: Mucous membranes moist/dry; JVD at ___ cm above sternal angle; carotid upstroke.
Cardiac: Rate, rhythm, S1/S2, S3 (volume overload), S4, murmurs, pericardial rub (uremic pericarditis).
Lungs: Clear or crackles (location); decreased breath sounds (effusion).
Abdomen: Soft/distended, ascites (shifting dullness, fluid wave), bruits, CVA tenderness, palpable kidneys (PKD), transplant graft in iliac fossa (tenderness?).
Extremities: Edema (pitting 1–4+, distribution), peripheral pulses, skin changes, calciphylaxis lesions in advanced CKD.
Access exam (if applicable): AVF/AVG side and configuration, thrill, bruit, aneurysmal changes, signs of infection, arm edema, steal. PD catheter exit site: clean/dry, erythema, drainage, tunnel tenderness. Central catheter: site, dressing, signs of infection.
Neuro: Asterixis, myoclonus, Chvostek/Trousseau in hypocalcemia, peripheral neuropathy in diabetic CKD.
"Euvolemic" is not an acceptable one-word exam. Chart the evidence: "JVP 8 cm, lungs clear, no S3, trace bilateral ankle edema, mucous membranes moist." Volume status drives the plan — nail it every time.
23 Abbreviations Master List
Diagnoses & Syndromes
Labs, Imaging & Calculations
Treatment & Access
24 Sample HPI Templates
These templates show the rhythm of a well-written nephrology HPI. Use them as patterns when you sit down to chart.
"Mr. [Name] is a 74-year-old male with a history of HTN, HFrEF (EF 30%), type 2 DM, and CKD G3a (baseline Cr 1.3, eGFR 55) admitted 3 days ago with community-acquired pneumonia, for whom nephrology is consulted for AKI. Creatinine on admission was 1.4; today is 2.8 with urine output 0.3 mL/kg/hr over the last 8 hours (KDIGO Stage 2 AKI by both criteria). He has received ceftriaxone, azithromycin, and 3 L of NS over 48 hours; home furosemide 40 mg daily was held. Exam is notable for BP 98/58, lungs with bibasilar crackles, JVP 10 cm, 2+ lower extremity edema, no rash, no flank tenderness. Labs: Na 134, K 5.3, HCO3 19, BUN 52, Cr 2.8, Mg 2.0, Phos 5.2. UA: SG 1.012, 2+ blood, 100 protein, granular casts, 10–20 WBC. FENa 2.4% (off diuretics). Renal ultrasound: 10.5 cm kidneys bilaterally, no hydronephrosis. Assessment: AKI KDIGO 2 likely ATN in the setting of sepsis-related hypoperfusion and contrast exposure from yesterday's CTA, superimposed on G3a CKD."
"Ms. [Name] is a 62-year-old female with type 2 DM (A1C 7.4), HTN, hyperlipidemia, and diabetic kidney disease CKD G4A3 (eGFR 22, UACR 1,450 mg/g) returning for routine follow-up. She feels well without dyspnea, orthopnea, or edema; appetite and energy are stable. Home weight 78 kg (up 1 kg). Compliant with lisinopril 20 mg, empagliflozin 10 mg, furosemide 40 mg, atorvastatin 40 mg, insulin glargine, and sevelamer 800 mg TID. BP at home averaging 128/72. Recent labs: Cr 2.8 (prior 2.7), K 4.8, HCO3 20, Hgb 10.2, iPTH 185, phos 5.4, Ca 9.0, 25-OH-vit D 22. Plan: continue RAAS and SGLT2i, start oral sodium bicarbonate 650 mg BID for acidosis, increase cholecalciferol, initiate ESA given Hgb < 10 with iron saturation 28%, discuss AVF referral and hepatitis B vaccination as part of RRT preparation."
"Mr. [Name] is a 68-year-old male with CKD G4 (baseline Cr 2.6), HFrEF, and type 2 DM who presents to the ED with fatigue and new muscle weakness. Home medications include lisinopril 40 mg, spironolactone 25 mg, carvedilol, and metformin. He recently started TMP-SMX for a skin infection. ED labs notable for K 7.2, Cr 3.4, HCO3 18, glucose 210. EKG shows peaked T waves without QRS widening. He received IV calcium gluconate 2 g, 10 units regular insulin with D50, albuterol nebulizer, and IV sodium bicarbonate. Repeat K 5.9 one hour later. Lisinopril, spironolactone, and TMP-SMX held. Nephrology consulted for ongoing management and dialysis access evaluation."
"Mrs. [Name] is a 59-year-old female with ESRD secondary to DKD on in-center HD (MWF, 3.5 hrs/session, Qb 400, 2K/2.5Ca bath, dry weight 68 kg) via a left brachiocephalic AVF created 14 months ago, presenting for routine monthly clinic. Over the past 2 weeks she has noted prolonged bleeding after needle removal and increased venous pressures during HD (alarms at 280). Exam: fistula with strong thrill at the anastomosis but diminished thrill and pulsatile character in the outflow segment. No erythema, no drainage, no aneurysmal dilation. Kt/V last month 1.38, URR 70%. Most recent labs: Hgb 10.8, ferritin 420, TSAT 32%, K 4.9, phos 5.8, iPTH 320, Ca 9.1, albumin 3.9. Plan: fistulogram for suspected outflow stenosis, continue current ESA and IV iron, increase sevelamer to 1,600 mg TID for phos."
"Mr. [Name] is a 47-year-old male 6 months s/p living-related-donor kidney transplant for IgA nephropathy, presenting for routine clinic. He is well without fever, dysuria, graft pain, or respiratory symptoms. Baseline post-transplant creatinine 1.2; today 1.3. Immunosuppression: tacrolimus 3 mg BID (last trough 7.2), mycophenolate 1,000 mg BID, prednisone 5 mg daily. On TMP-SMX for PJP prophylaxis and valganciclovir for CMV. Most recent BK PCR negative, CMV PCR negative. BP 128/78 on amlodipine 5 mg. A1C 6.1, no new-onset diabetes. UACR 25 mg/g. Plan: continue current immunosuppression, obtain protocol labs including tacrolimus trough, BK and CMV PCR, CBC, CMP, and UACR; continue prophylaxis through month 6."
"Ms. [Name] is a 29-year-old previously healthy female presenting with 2 episodes of gross hematuria, each occurring 1–2 days after an upper respiratory infection over the past 6 months. She denies dysuria, flank pain, rash, joint pain, or hemoptysis. BP in clinic 142/88. Exam otherwise unremarkable. Labs: Cr 1.0 (baseline 0.8), UA with 50–100 dysmorphic RBCs/hpf, RBC casts present, 2+ protein. UPCR 1.1 g/g. ANA negative, ANCA negative, anti-GBM negative, C3/C4 normal, hepatitis B/C negative, IgA slightly elevated. Renal ultrasound normal. Findings most consistent with IgA nephropathy; kidney biopsy scheduled for next week. Started on lisinopril 10 mg daily for BP and proteinuria."
25 References & Sources
Clinical Practice Guidelines
KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int Suppl. 2012.
KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of CKD. Kidney Int. 2024.
KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021.
ADA Standards of Care in Diabetes — Chronic Kidney Disease and Risk Management. Diabetes Care. 2023.
USPSTF: Screening for Chronic Kidney Disease. Ann Intern Med.
KDOQI Clinical Practice Guidelines (Vascular Access, Nutrition, Anemia, Diabetes).
American Society of Nephrology (ASN) educational resources and position statements.
Landmark Clinical Trials
DAPA-CKD: Dapagliflozin in Patients with Chronic Kidney Disease. NEJM. 2020.
EMPA-KIDNEY: Empagliflozin in Patients with Chronic Kidney Disease. NEJM. 2023.
FIDELIO-DKD: Finerenone in Diabetic Kidney Disease. NEJM. 2020.
CORAL: Stenting and Medical Therapy for Atherosclerotic Renal-Artery Stenosis. NEJM. 2014.
TEMPO 3:4: Tolvaptan in Autosomal Dominant Polycystic Kidney Disease. NEJM. 2012.
Diagram & Figure Sources
Figure 1: Gross kidney anatomy. OpenStax College. CC BY 3.0.
Figure 2: Labeled kidney anatomy. Blausen Medical. CC BY 3.0.
Figure 3: Renal blood flow. OpenStax College. CC BY 3.0.
Figure 4: Nephron secretion and absorption. OpenStax College. CC BY 3.0.
Figure 5: Nephron in action. OpenStax College. CC BY 3.0.
Nephrology is the specialty of numbers, trends, and mechanism. A great scribe never writes "creatinine is up" — they write "Cr 2.8, up from baseline 1.3 two weeks ago, with FENa 2.4% and muddy brown casts on UA." They know that "hyperkalemia" means why (acidosis? ACEi? AKI? hemolysis?) and how bad (EKG changes? membrane stabilized?). They pull the prior biopsy report before the new patient walks in, pre-populate the problem list with the CKD bundle, and already have the tacrolimus trough ready when the transplant attending sits down.
Learn the mechanism, memorize the staging, track the trend, and document the evidence. The attending will notice.
Welcome to nephrology.