Neurology
Every diagnosis, condition, procedure, medication, abbreviation, and documentation framework you need to succeed on day one and beyond in a neurology clinic, stroke service, or neurohospitalist setting.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Neuroanatomy & Neurophysiology Essentials
Neurology is the medical (non-surgical) specialty managing disorders of the brain, spinal cord, peripheral nerves, neuromuscular junction, and muscle. A strong scribe understands the anatomic substrate for every complaint because neurologists localize before they diagnose — the single most important question in a neurology note is "where is the lesion?"
The Central Nervous System
The cerebrum is divided into two hemispheres joined by the corpus callosum. Each hemisphere has four lobes: the frontal (executive function, motor planning, Broca's speech area on the dominant side, personality), parietal (primary somatosensory cortex, spatial processing, the dominant-hemisphere angular gyrus for reading/writing), temporal (hearing, memory via hippocampus, Wernicke's receptive language area on the dominant side), and occipital (primary visual cortex). The diencephalon contains the thalamus (sensory relay) and hypothalamus (autonomic/endocrine). The basal ganglia (caudate, putamen, globus pallidus, substantia nigra, subthalamic nucleus) orchestrate movement. The cerebellum coordinates smooth movement, balance, and gait. The brainstem — midbrain, pons, medulla — houses the cranial nerve nuclei, reticular activating system, and long tracts.
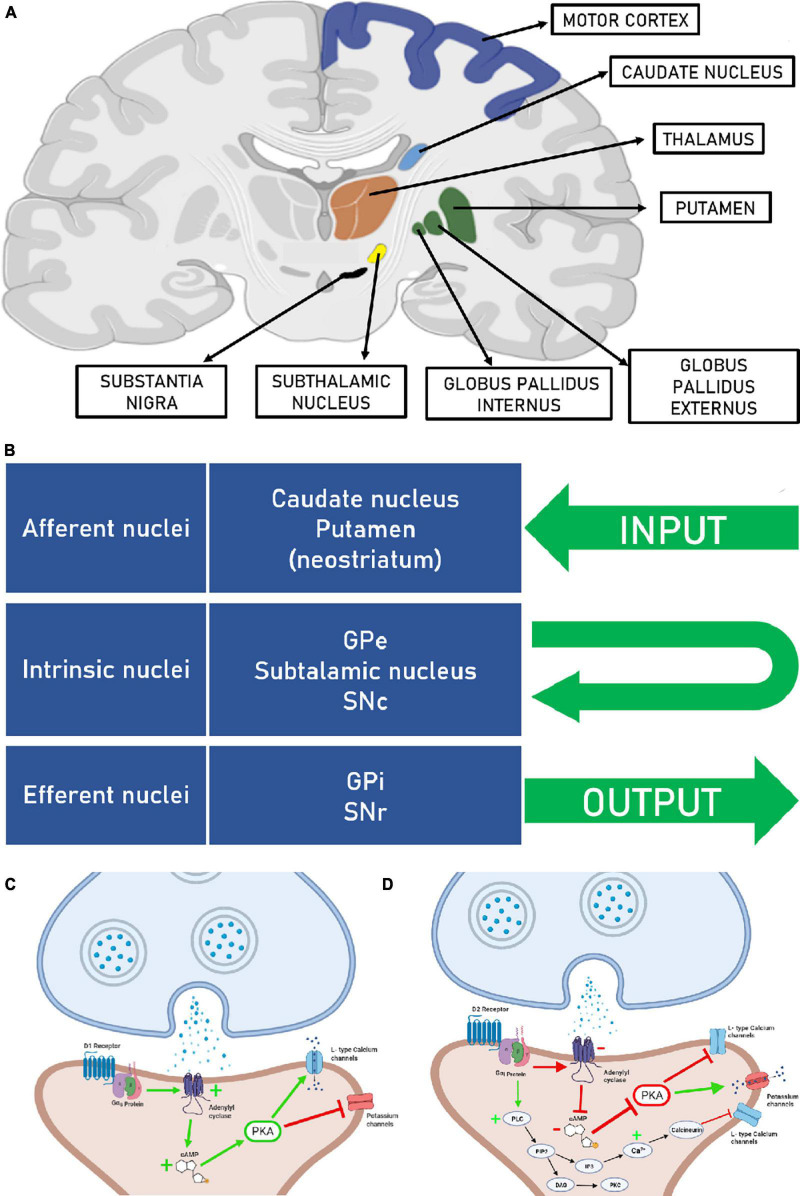
Vascular Supply
The brain is perfused by the anterior circulation (internal carotid → anterior and middle cerebral arteries) and the posterior circulation (vertebral arteries → basilar artery → posterior cerebral arteries, plus PICA, AICA, SCA to the cerebellum/brainstem). These connect at the circle of Willis via the anterior and posterior communicating arteries. Stroke syndromes are named for the occluded vessel: an MCA stroke produces contralateral arm-face weakness and, on the dominant side, aphasia; an ACA stroke produces contralateral leg weakness; a PCA stroke produces homonymous hemianopia; lacunar strokes in deep perforators produce pure motor, pure sensory, or ataxic-hemiparesis syndromes without cortical findings.
Cranial Nerves & Tracts
The 12 cranial nerves must be memorized: I olfactory, II optic, III oculomotor, IV trochlear, V trigeminal, VI abducens, VII facial, VIII vestibulocochlear, IX glossopharyngeal, X vagus, XI accessory, XII hypoglossal. The long tracts — corticospinal (motor), dorsal columns (proprioception/vibration), spinothalamic (pain/temperature) — run through the spinal cord with characteristic crossing patterns that let the neurologist localize spinal cord lesions to a specific level and side.
Neurologists speak in localizing language. When the attending says "right MCA superior division" or "left pontine lacune" or "C5–C6 radiculopathy," you need to chart it verbatim. Learn the vascular territories and dermatomes cold — they are used every shift.
02 Scribe Documentation Framework
Neurology notes are organized around localization and temporal profile. A well-written neurology HPI tells the reader, in order, what happened, when, how fast it came on, what systems are involved, and where in the nervous system the process lives. Neurology encounters fall into three buckets: acute (ED stroke, status epilepticus, ICH), outpatient subspecialty (headache, MS, epilepsy, movement, memory), and consultative (inpatient "altered mental status," "new weakness," "seizure?").
Chief Complaint: A single concise statement — "acute right-sided weakness," "follow-up MS on ocrelizumab," "memory loss," "first-time seizure."
HPI: Use OLDCARTS plus the neurology-specific modifiers: onset (hyperacute seconds, acute minutes, subacute days–weeks, chronic months–years), course (improving, worsening, stepwise, relapsing, progressive), triggers (posture, Valsalva, sleep deprivation, flashing lights, head movement), associated neurologic symptoms by system (cognition, vision, speech, weakness, sensation, balance, sphincter).
Last Known Well (LKW): In acute stroke, the time the patient was last seen at baseline — drives eligibility for thrombolysis and thrombectomy. Always document to the minute.
PMHx/PSHx/Meds/Allergies/SHx/FHx: Capture vascular risk factors (HTN, DM, HLD, AFib, smoking, OSA), prior strokes/TIAs, seizures (frequency and last event), head trauma, migraines, family history of neurodegenerative disease (Huntington, early-onset Alzheimer, Parkinson), alcohol and recreational drug use.
Vitals: BP (both arms for stroke, watch for permissive hypertension targets), HR and rhythm, glucose (always check in altered mental status), temperature.
Neurologic exam: Mental status, cranial nerves II–XII, motor (tone, bulk, power by MRC 0–5), reflexes (0–4+), sensation (light touch, pinprick, vibration, proprioception), coordination (finger-to-nose, heel-to-shin, rapid alternating movements), gait (casual, tandem, Romberg). Add the NIHSS for every stroke alert.
Results: Labs (CBC, BMP, coags, lipid, A1C, TSH, B12 for neuropathy/cognition), imaging (NCCT, CTA, MRI), EEG, LP findings, EMG/NCS.
Neurology A&P starts with a one-sentence summary that localizes: "65M with acute onset right face-arm weakness and expressive aphasia localizing to the left MCA territory, most consistent with acute ischemic stroke." Then list each problem with its workup and plan. A good neuro note always closes the loop between symptom, localization, and treatment.
In stroke alerts, the three times you must chart are last known well, symptom discovery, and arrival/door time. Thrombolysis windows (4.5 hours) and thrombectomy windows (up to 24 hours in selected patients per DAWN/DEFUSE-3) depend on these.
03 Ischemic Stroke & TIA Vascular
Ischemic stroke accounts for roughly 87% of all strokes. It results from occlusion of a cerebral artery, producing focal neurologic deficit from infarction of the territory it supplies. Transient ischemic attack (TIA) is a brief episode of focal neurologic dysfunction from ischemia without infarction on imaging — time-based definitions ("< 24 hours") have been replaced by tissue-based definitions.
Risk Factors & Comorbidities
Hypertension: the single most important modifiable stroke risk factor — document baseline BP, home regimen, and adherence.
Atrial fibrillation: document rhythm, prior rate/rhythm control, anticoagulation status and CHA2DS2-VASc score.
Diabetes: most recent A1C, diabetic meds.
Hyperlipidemia: LDL, current statin.
Smoking: pack-years, quit date.
Obstructive sleep apnea: strong link to stroke recurrence — document CPAP use.
Prior stroke/TIA, carotid disease, PFO in younger patients, sickle cell, hypercoagulable states, and family history of stroke round out the risk-factor section.
Subtypes (TOAST Classification)
- Large artery atherosclerosis — carotid or intracranial plaque causing artery-to-artery embolism or hemodynamic failure.
- Cardioembolic — most commonly atrial fibrillation; also LV thrombus, endocarditis, patent foramen ovale with paradoxical embolism.
- Small vessel (lacunar) — lipohyalinosis of deep penetrating arteries. Classic syndromes: pure motor hemiparesis (internal capsule), pure sensory (thalamus), ataxic hemiparesis, dysarthria-clumsy hand.
- Other determined etiology — dissection, vasculitis, hypercoagulable state, sickle cell.
- Cryptogenic / undetermined — workup negative or incomplete. Often pushed toward extended rhythm monitoring to look for occult AFib.
Balance (sudden loss), Eyes (vision change), Face (drooping), Arm (weakness), Speech (slurred or absent), Time (call 911, document LKW). Scribes should chart the time of each event with precision — every minute of delay loses approximately 1.9 million neurons.
Clinical Presentation by Territory
| Vessel | Classic Findings |
|---|---|
| MCA (superior division) | Contralateral face/arm weakness > leg, Broca aphasia (dominant) |
| MCA (inferior division) | Wernicke aphasia (dominant), neglect (non-dominant), superior quadrantanopia |
| MCA (deep / lenticulostriate) | Pure motor hemiparesis from internal capsule involvement |
| ACA | Contralateral leg > arm weakness, abulia, urinary incontinence |
| PCA | Contralateral homonymous hemianopia, alexia without agraphia (dominant) |
| Basilar / brainstem | Crossed findings, diplopia, dysarthria, vertigo, "locked-in" if severe |
| Cerebellar (PICA / AICA / SCA) | Ipsilateral ataxia, vertigo, nystagmus, nausea — can mimic peripheral vertigo |
Acute Management
The first step is NCCT of the head to exclude hemorrhage, followed by CTA head and neck to identify large vessel occlusion (LVO), and often CT perfusion to assess salvageable penumbra. 2019 AHA/ASA Guidelines for the Early Management of Acute Ischemic Stroke are the standard citation. IV thrombolysis with alteplase (tPA) 0.9 mg/kg is indicated within 4.5 hours of LKW in eligible patients; tenecteplase (TNK) 0.25 mg/kg single bolus is now widely used as a more practical alternative and is supported by multiple non-inferiority trials. Mechanical thrombectomy for LVO is standard up to 6 hours and up to 24 hours in selected patients with favorable imaging (DAWN, DEFUSE-3).
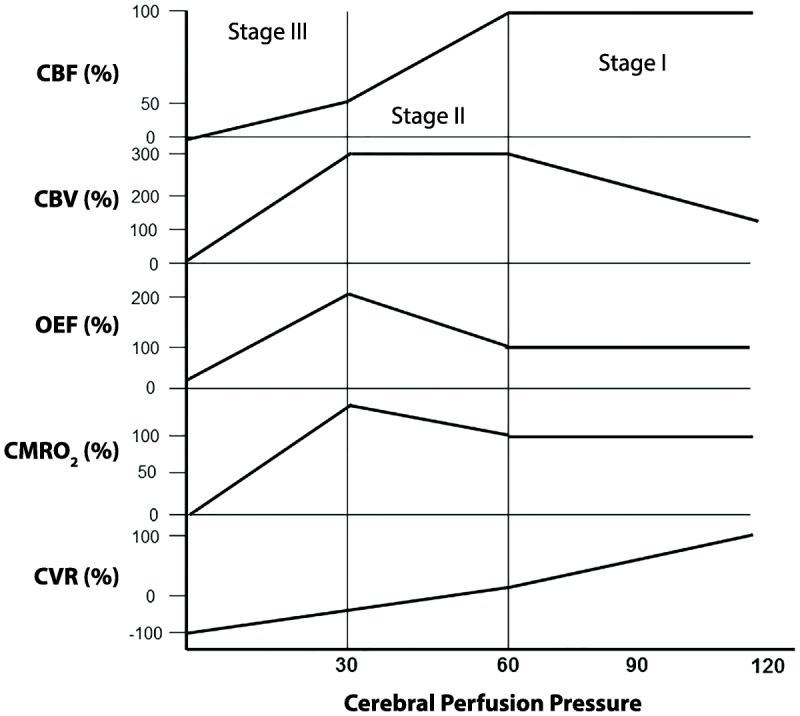
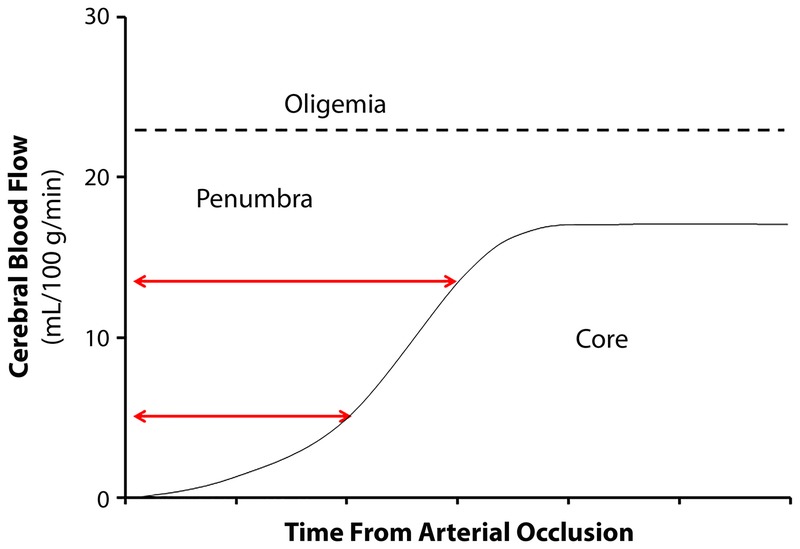
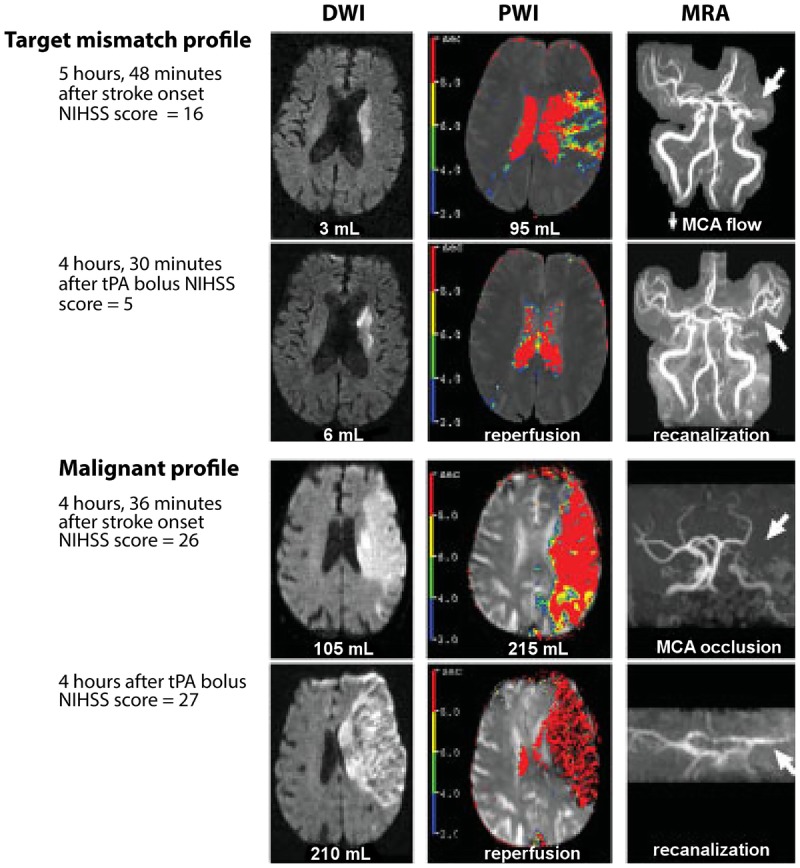
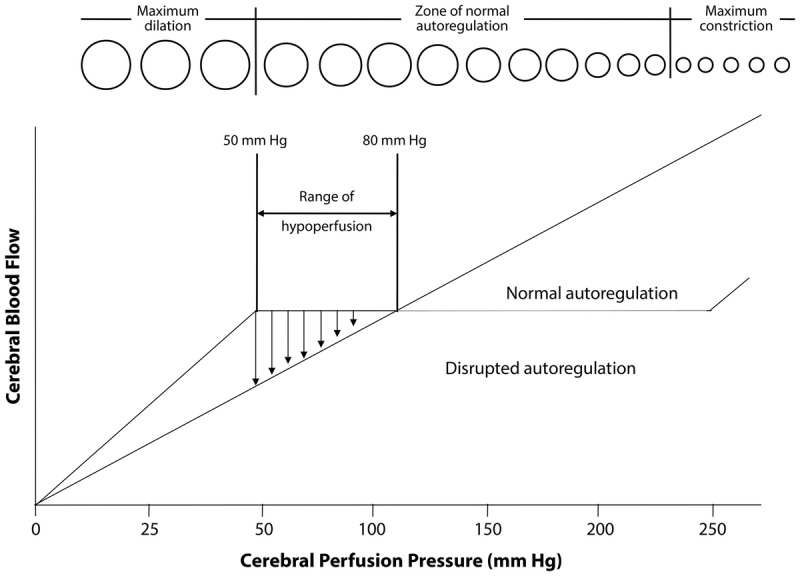
Blood pressure is managed with "permissive hypertension" (up to 220/120) in non-thrombolysis patients to maintain perfusion; targets are lowered to <185/110 before tPA and <180/105 for 24 hours after. Secondary prevention starts immediately: antiplatelet (aspirin 81 mg, or clopidogrel; dual antiplatelet with aspirin + clopidogrel for 21–90 days after minor stroke or high-risk TIA per CHANCE and POINT), high-intensity statin, BP control, glycemic control, smoking cessation, and anticoagulation for cardioembolic sources. The 2021 AHA/ASA Guideline for the Prevention of Stroke in Patients with Stroke and TIA is the foundational citation for secondary prevention.
For every stroke alert, chart the NIHSS score item by item, the LKW, the NCCT read ("no hemorrhage, no early ischemic changes" or "ASPECTS 8"), whether tPA/TNK was given and the time of bolus, and whether the patient went to the neuro-IR suite for thrombectomy. The attending will dictate these rapidly — miss nothing.
04 Intracerebral Hemorrhage (ICH) Vascular
Spontaneous ICH accounts for ~10–15% of strokes but carries the highest mortality of any stroke subtype — 30-day mortality ~40% and only about 20% of survivors regain functional independence.
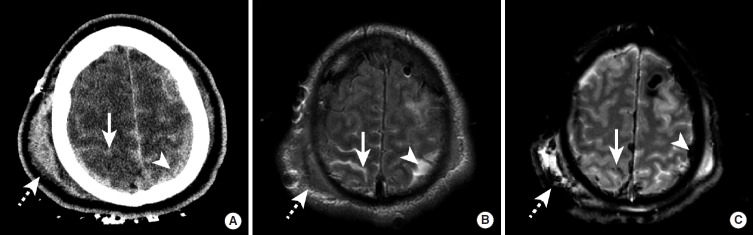
Etiologies: hypertensive arteriopathy (deep bleeds in basal ganglia, thalamus, pons, cerebellum from lipohyalinosis of perforating arteries), cerebral amyloid angiopathy (CAA) (lobar bleeds and microbleeds in elderly, from amyloid deposition in cortical/leptomeningeal vessels), vascular malformations (AVM, cavernoma, dural AV fistula), coagulopathy/anticoagulation, hemorrhagic conversion of ischemic stroke, tumor hemorrhage (especially melanoma, RCC, thyroid, choriocarcinoma, GBM), venous sinus thrombosis with venous infarct, sympathomimetic drug use (cocaine, methamphetamine), and reversible cerebral vasoconstriction syndrome (RCVS).
Presentation & Workup
Abrupt focal deficit often with headache, vomiting, and depressed level of consciousness — more likely than ischemic stroke to present with reduced GCS and early clinical decline. Deep bleeds produce hemiparesis out of proportion to cortical signs; cerebellar bleeds present with headache, vomiting, ataxia, and risk of brainstem compression; pontine bleeds produce coma, pinpoint pupils, and crossed findings. NCCT shows hyperdense (~60–80 HU) blood immediately and quantifies volume using the ABC/2 formula. CTA identifies a "spot sign" (active contrast extravasation, predicting hematoma expansion) and excludes an underlying vascular lesion. MRI with gradient echo or susceptibility-weighted imaging reveals microbleeds that support a CAA diagnosis (Boston criteria). Labs: CBC with platelets, PT/INR, PTT, anti-Xa, type and screen, basic chemistries, toxicology.
Management
Per 2022 AHA/ASA ICH Guideline: aggressive SBP lowering to ~140 mmHg is reasonable in mild-moderate ICH (INTERACT-2, ATACH-2), reversal of any anticoagulation is mandatory (4-factor PCC for warfarin with vitamin K, idarucizumab for dabigatran, andexanet alfa or 4F-PCC for factor Xa inhibitors, platelets are generally NOT indicated in antiplatelet-associated ICH per PATCH), seizure prophylaxis only if seizures occur or in lobar ICH with depressed consciousness, neurosurgical evacuation for cerebellar hemorrhage > 3 cm or with brainstem compression/hydrocephalus, external ventricular drainage for obstructive hydrocephalus or large IVH, and ICU-level monitoring including head-of-bed elevation, normothermia, and osmotherapy (mannitol, hypertonic saline) for elevated ICP. The ICH Score (0–6) predicts 30-day mortality based on GCS, ICH volume, IVH, infratentorial origin, and age ≥ 80. Minimally invasive surgical evacuation (MISTIE, ENRICH) is an evolving option for selected supratentorial ICH.
05 Subarachnoid Hemorrhage (SAH) Vascular
Non-traumatic SAH is most commonly due to rupture of a saccular (berry) aneurysm at the circle of Willis. The hallmark presentation is the thunderclap headache — "the worst headache of my life," reaching maximum intensity within seconds. Associated features include neck stiffness, photophobia, transient LOC, and vomiting. NCCT within 6 hours is >95% sensitive; if negative but suspicion remains high, lumbar puncture looking for xanthochromia is standard. CTA or catheter angiography identifies the aneurysm.
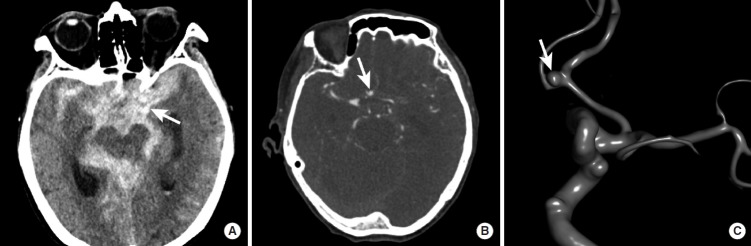
Grading & Management
Severity is graded by the Hunt and Hess scale (I asymptomatic/mild headache; II moderate-severe headache, nuchal rigidity, no deficit; III drowsy, mild deficit; IV stupor, moderate-severe hemiparesis; V deep coma, decerebrate posturing) and the modified Fisher scale based on CT blood burden. Management per 2023 AHA/ASA Aneurysmal SAH Guideline includes early aneurysm securing (clip or coil within 24–72 hours), nimodipine 60 mg PO q4h for 21 days to reduce delayed cerebral ischemia from vasospasm, BP control, and ICU monitoring with transcranial Doppler for vasospasm surveillance days 4–14.
06 Headache Disorders Medical
Headache is the most common neurology complaint. The scribe must distinguish primary headaches (migraine, tension, cluster) from secondary causes (SAH, meningitis, tumor, IIH, dissection, giant cell arteritis). The red-flag mnemonic SNOOP — Systemic symptoms, Neurologic deficits, Onset thunderclap, Older age new-onset, Pattern change — drives the decision to image.
Migraine
Episodic, recurrent, unilateral (often), pulsating, moderate-to-severe headache lasting 4–72 hours, worsened by activity, with nausea, photophobia, or phonophobia.
Migraine with aura includes transient visual (scintillating scotoma, fortification spectra), sensory, or language phenomena lasting 5–60 minutes. Abortive therapy: triptans (sumatriptan 100 mg PO or 6 mg SC, rizatriptan, eletriptan), NSAIDs, gepants (ubrogepant, rimegepant), lasmiditan. Preventive therapy (indicated at ≥4 headache days/month): beta blockers (propranolol, metoprolol), topiramate, valproate, amitriptyline, candesartan, and the CGRP monoclonal antibodies (erenumab, fremanezumab, galcanezumab, eptinezumab). OnabotulinumtoxinA (Botox) every 12 weeks is approved for chronic migraine (≥15 headache days/month). AHS 2021 Consensus Statement on Migraine Prevention is the standard citation.
Tension-Type Headache
Bilateral, pressing/tightening ("band-like"), mild-to-moderate, no nausea, no photo/phonophobia (or one but not both). Managed with NSAIDs, stress reduction, and TCA prophylaxis if frequent.
Cluster Headache
Severe unilateral orbital/periorbital pain in bouts of 15–180 minutes, occurring up to 8 times/day, with ipsilateral autonomic features (conjunctival injection, lacrimation, rhinorrhea, ptosis, miosis) and restlessness. Acute treatment: high-flow O2 12–15 L/min via non-rebreather and SC sumatriptan. Preventive: verapamil, short steroid bridge, galcanezumab.
Systemic symptoms (fever, weight loss) or Secondary risk factors (HIV, cancer); Neurologic symptoms or abnormal exam; Onset sudden/thunderclap; Older age (new onset >50); Pattern change from prior headaches, Positional, Precipitated by Valsalva/exertion, Papilledema. Any red flag prompts neuroimaging.
Secondary Headache — Trigeminal Neuralgia, IIH, GCA
Trigeminal neuralgia: lancinating, electric-shock facial pain in V2/V3 distribution triggered by light touch, chewing, brushing teeth. First-line therapy is carbamazepine or oxcarbazepine; refractory cases go for microvascular decompression or percutaneous rhizotomy. Idiopathic intracranial hypertension (IIH): typically obese young woman with daily headaches, pulsatile tinnitus, transient visual obscurations, and papilledema; LP opening pressure >25 cm H2O. Treat with weight loss and acetazolamide; VP shunt or optic nerve sheath fenestration for vision-threatening disease. Giant cell arteritis: >50 yo with new temporal headache, jaw claudication, scalp tenderness, PMR, ESR/CRP elevated — start high-dose steroids immediately and biopsy within 1–2 weeks.
07 Seizures & Epilepsy Paroxysmal
A seizure is a transient occurrence of signs/symptoms due to abnormal excessive or synchronous neuronal activity. Epilepsy is defined as ≥2 unprovoked seizures >24 hours apart, or one unprovoked seizure with high recurrence risk. The ILAE classification divides seizures into focal (with or without impaired awareness; with or without motor features), generalized (tonic-clonic, absence, myoclonic, atonic, tonic, clonic), and unknown onset.
Semiology the Scribe Will Hear
Focal aware (formerly "simple partial"): preserved consciousness with a localized sensation — déjà vu, rising epigastric sensation (temporal lobe), unilateral jerking (motor strip). Focal impaired awareness (formerly "complex partial"): blank staring, oral and manual automatisms, postictal confusion. Generalized tonic-clonic: sudden LOC, tonic stiffening, clonic jerking, tongue biting (lateral), incontinence, postictal confusion lasting minutes to an hour.
Workup
MRI brain to look for structural lesion, EEG (routine, prolonged, or inpatient video-EEG) to capture interictal epileptiform discharges or ictal events, labs including glucose, electrolytes, magnesium, toxicology, and LP if CNS infection is suspected.
Status Epilepticus
Defined as a seizure >5 minutes or recurrent seizures without return to baseline. Treatment algorithm per Neurocritical Care Society / AES Guideline: first-line IV lorazepam 4 mg (or IM midazolam 10 mg); second-line loading with IV levetiracetam 60 mg/kg, fosphenytoin 20 mg PE/kg, or valproate 40 mg/kg (ESETT trial showed equivalence); third-line is anesthetic-level sedation with midazolam, propofol, or pentobarbital infusions in the ICU with continuous EEG monitoring.
A provoked (acute symptomatic) seizure occurs in the setting of an acute insult — hypoglycemia, hyponatremia (<120), uremia, alcohol withdrawal (12–48 hours after last drink), drug toxicity, acute stroke, meningitis, head trauma, eclampsia. Correcting the trigger typically prevents recurrence, and most patients are NOT started on long-term ASM. An unprovoked seizure occurs without an acute trigger and drives the epilepsy workup. Always chart the presumed provoking factor if one exists.
Chronic Management
Antiseizure medications (ASMs) are chosen by seizure type, comorbidities, pregnancy potential, and side-effect profile. Broad-spectrum agents (effective for focal and generalized): lamotrigine, levetiracetam, valproate, topiramate, zonisamide. Narrow-spectrum (focal only): carbamazepine, oxcarbazepine, phenytoin, lacosamide. Absence seizures respond to ethosuximide or valproate. Valproate is teratogenic and contraindicated in pregnancy whenever possible. Lamotrigine requires slow titration due to Stevens-Johnson risk. AAN Guideline on Treatment of New-Onset Epilepsy is the standard citation.
08 Syncope & Transient Loss of Consciousness Paroxysmal
Neurologists are consulted to distinguish syncope (global cerebral hypoperfusion, brief, rapid return to baseline) from seizure (cortical electrical event, often with postictal state). Cardiogenic syncope (arrhythmia, structural heart disease) is managed by cardiology but overlaps with neurology work-up. Key distinguishing features: tongue biting (lateral → seizure, tip → nonspecific), incontinence (nonspecific), duration of LOC (seconds in syncope vs minutes in seizure), postictal confusion and myalgias (absent in syncope, typical in tonic-clonic), preceding aura or déjà vu (seizure), preceding warmth/nausea/tunnel vision (vasovagal), and position at onset (supine onset favors seizure or arrhythmia). Convulsive syncope — brief myoclonic jerks at the end of a vasovagal episode — is commonly mistaken for seizure but lacks a true postictal state.
Workup: ECG in every patient, orthostatic vitals, echo if structural disease is suspected, and Holter, 30-day event monitor, or implantable loop recorder for recurrent unexplained syncope. Tilt-table testing identifies neurocardiogenic and orthostatic causes. EEG and MRI brain are not routinely indicated in classic vasovagal syncope — they are reserved for cases with features suggestive of seizure or a focal deficit.
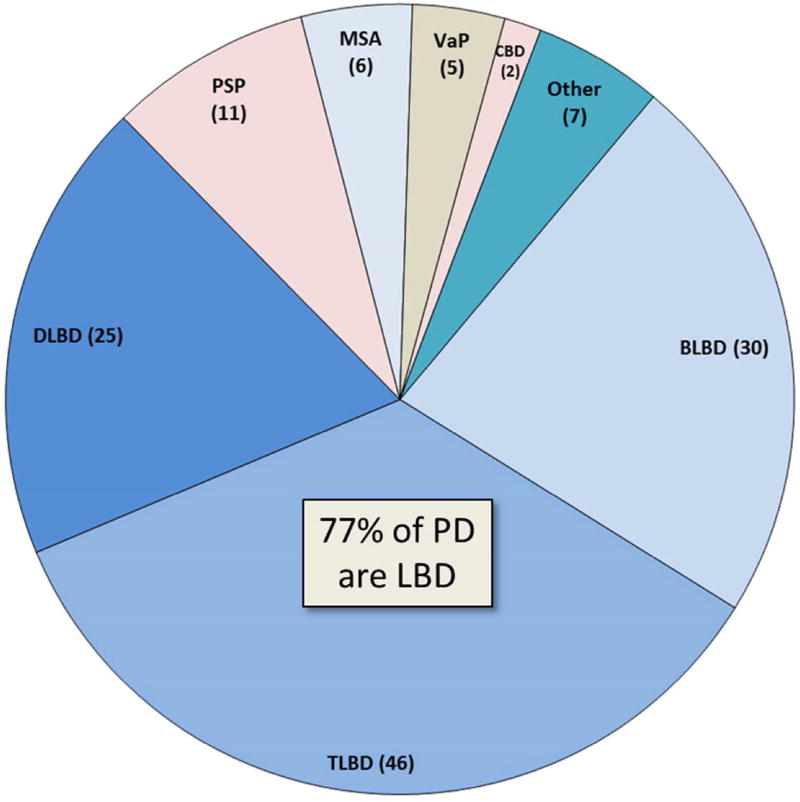
09 Multiple Sclerosis & Demyelinating Disease Demyelinating
Multiple sclerosis (MS) is an autoimmune demyelinating disease of the CNS characterized by lesions disseminated in space and time. Onset is typically age 20–40, female predominant. Phenotypes: clinically isolated syndrome (CIS), relapsing-remitting MS (RRMS) (~85% at onset), secondary progressive MS (SPMS), and primary progressive MS (PPMS).
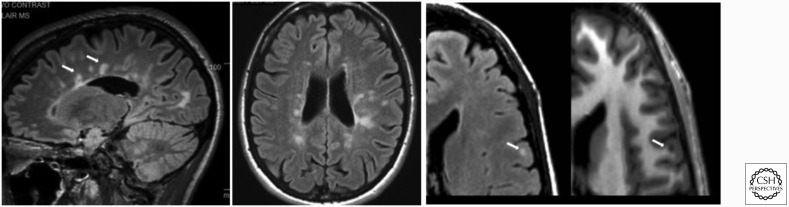
Clinical Presentations
- Optic neuritis — painful unilateral vision loss with afferent pupillary defect and central scotoma, typically recovering over weeks.
- Transverse myelitis — spinal cord syndrome with paraparesis, sensory level, bowel/bladder dysfunction. Lhermitte sign (electric shock with neck flexion).
- Brainstem/cerebellar — internuclear ophthalmoplegia (bilateral INO is essentially pathognomonic in a young adult), vertigo, ataxia.
- Cerebral — cognitive changes, hemisensory or hemimotor symptoms.
- Uhthoff phenomenon — symptom worsening with heat or exercise.
Diagnosis
McDonald criteria require dissemination in space (≥2 typical CNS regions on MRI) and time (simultaneous enhancing and non-enhancing lesions, or new lesions on follow-up MRI). CSF shows oligoclonal bands unique to CSF in ~85% of MS patients. Evoked potentials (visual, somatosensory) may show subclinical demyelination.
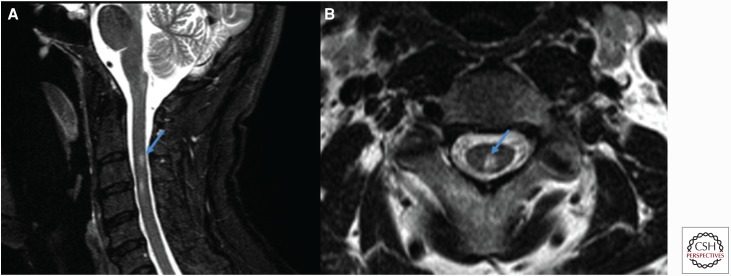
Management
Acute relapses: high-dose IV methylprednisolone 1 g daily x 3–5 days (PO equivalents acceptable per AAN evidence review); plasma exchange for steroid-refractory attacks. Disease-modifying therapies (DMTs): injectables (interferon beta-1a/1b, glatiramer acetate), oral agents (dimethyl fumarate, diroximel fumarate, fingolimod and other S1P modulators ozanimod/ponesimod/siponimod, teriflunomide, cladribine), and high-efficacy monoclonals (natalizumab — monitor JC virus for PML risk; ocrelizumab — anti-CD20, approved for RRMS and PPMS; ofatumumab, ublituximab; alemtuzumab). Treatment selection is driven by disease activity, patient preference, pregnancy plans, and safety monitoring needs. AAN 2018 Practice Guideline on DMTs for MS is the standard citation.
NMO and MOGAD
Neuromyelitis optica spectrum disorder (NMOSD) is a distinct astrocytopathy with aquaporin-4 IgG antibodies, classically producing severe optic neuritis and longitudinally extensive transverse myelitis (≥3 vertebral segments). Treated with rituximab, eculizumab, inebilizumab, or satralizumab — NOT standard MS DMTs, which can worsen it. MOGAD (myelin oligodendrocyte glycoprotein antibody disease) overlaps clinically and is managed with immunotherapy.
10 Amyotrophic Lateral Sclerosis Degenerative
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease affecting both upper motor neurons (corticospinal tract — spasticity, hyperreflexia, Babinski, pseudobulbar affect) and lower motor neurons (anterior horn cells — weakness, atrophy, fasciculations, cramps). The hallmark is mixed UMN and LMN findings in the same body segment. Median survival is 3–5 years, most commonly from respiratory failure, though ~10% live longer than 10 years. About 10% of cases are familial (most commonly C9orf72 hexanucleotide expansion, SOD1, FUS, TARDBP mutations). Up to 50% of ALS patients have measurable cognitive or behavioral involvement on the ALS-FTD spectrum.
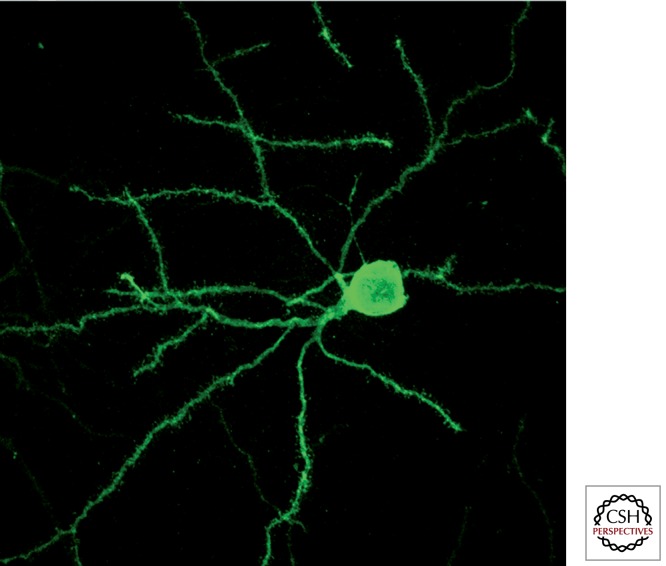
Clinical Phenotypes & Diagnosis
Presentations include limb-onset (asymmetric foot drop or hand weakness that spreads), bulbar-onset (dysarthria, dysphagia, tongue atrophy and fasciculations, sialorrhea, emotional lability), and respiratory-onset (orthopnea, dyspnea on exertion, morning headache from nocturnal hypoventilation). Progressive muscular atrophy (pure LMN) and primary lateral sclerosis (pure UMN) are related motor neuron disorders on the same continuum. Diagnosis is clinical with EMG confirmation — EMG shows widespread denervation with fasciculations, positive sharp waves, fibrillation potentials, and chronic reinnervation changes (large-amplitude long-duration motor unit potentials). MRI is typically used to exclude mimics such as cervical spondylotic myelopathy, multifocal motor neuropathy with conduction block (responds to IVIG), Kennedy disease, and paraneoplastic motor neuronopathy. Revised El Escorial / Awaji criteria guide diagnostic certainty.
Management
Care is delivered through multidisciplinary ALS clinics combining neurology, pulmonology, PT/OT, speech, nutrition, respiratory therapy, and palliative care. Disease-modifying therapies: riluzole 50 mg BID (glutamate antagonist, modestly prolongs survival ~3 months), edaravone (IV or PO, free-radical scavenger, slows functional decline in selected patients with preserved function), tofersen for SOD1-ALS (antisense oligonucleotide). Symptomatic care includes non-invasive ventilation (BiPAP) when FVC falls below ~50% predicted, PEG feeding when dysphagia and weight loss become unsafe, secretion management (glycopyrrolate, scopolamine, suction), spasticity treatment (baclofen, tizanidine), dextromethorphan/quinidine for pseudobulbar affect, and early hospice/palliative integration. Serial monitoring uses the ALSFRS-R functional scale and pulmonary function tests.
11 Parkinson Disease & Parkinsonism Movement
Parkinson disease (PD) is the second most common neurodegenerative disorder after Alzheimer. Pathology: loss of dopaminergic neurons in the substantia nigra pars compacta with alpha-synuclein-containing Lewy bodies. The MDS Clinical Diagnostic Criteria (Postuma et al., 2015) require bradykinesia plus rest tremor and/or rigidity, with supportive features and absence of absolute exclusion criteria.
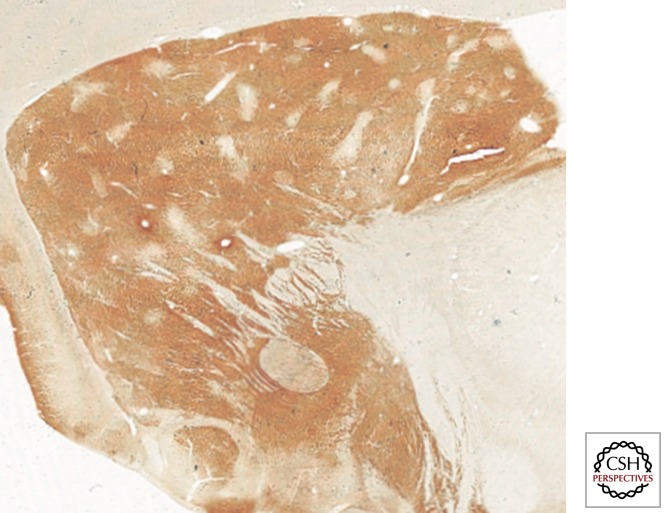
Cardinal Features — "TRAP"
- Tremor at rest, 4–6 Hz, pill-rolling, asymmetric onset.
- Rigidity — lead-pipe or cogwheel.
- Akinesia/bradykinesia — slowness, hypomimia, micrographia, decreased arm swing.
- Postural instability (later feature) — positive pull test, retropulsion, falls.
Non-motor features are now recognized as major drivers of disability: REM sleep behavior disorder (often premotor), hyposmia, constipation, orthostatic hypotension, urinary urgency, depression, anxiety, and cognitive impairment/PDD in later disease. Severity is staged with Hoehn & Yahr (I unilateral; II bilateral no balance impairment; III mild bilateral with postural instability, physically independent; IV severe disability, still able to walk/stand unassisted; V wheelchair/bedbound) and functional impact with MDS-UPDRS.
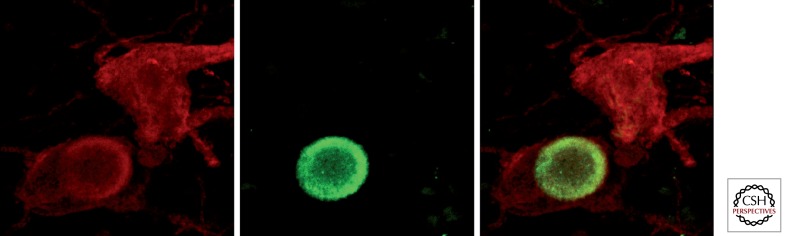
After 3–5 years of levodopa therapy, motor fluctuations emerge: wearing-off (return of parkinsonism before the next dose), on-off phenomenon (unpredictable transitions), and peak-dose dyskinesia (involuntary choreiform movements at maximum levodopa effect). Management includes more frequent dosing, long-acting formulations (Rytary), COMT inhibitor add-on, amantadine for dyskinesia, continuous infusion therapies (apomorphine, Duopa), or DBS. Scribes should chart each fluctuation pattern explicitly — "wearing off 30 minutes before each dose" or "peak-dose dyskinesia at 90 minutes post-dose."
Atypical Parkinsonism ("Parkinson-Plus")
| Syndrome | Distinguishing Features |
|---|---|
| Progressive supranuclear palsy (PSP) | Early falls, vertical gaze palsy, axial rigidity, poor levodopa response |
| Multiple system atrophy (MSA) | Autonomic failure, cerebellar or parkinsonian predominant, poor levodopa response |
| Corticobasal degeneration (CBD) | Asymmetric rigidity, apraxia, alien limb, cortical sensory loss |
| Dementia with Lewy bodies (DLB) | Dementia preceding or within 1 year of parkinsonism, fluctuating cognition, visual hallucinations, REM sleep behavior disorder, neuroleptic sensitivity |
Treatment
First-line for motor symptoms is carbidopa/levodopa, the most effective dopaminergic therapy. Dopamine agonists (pramipexole, ropinirole, rotigotine patch) are often used in younger patients to delay levodopa-induced dyskinesia but carry impulse-control disorder risk. MAO-B inhibitors (selegiline, rasagiline, safinamide) and COMT inhibitors (entacapone, opicapone) extend levodopa action. Amantadine reduces dyskinesia. Advanced therapies include deep brain stimulation (DBS) of the STN or GPi, continuous subcutaneous apomorphine, and levodopa-carbidopa intestinal gel (Duopa).
12 Other Movement Disorders Movement
Essential tremor (ET) is the most common movement disorder: bilateral, largely symmetric postural and kinetic hand tremor (worse with target-directed activity such as pouring or drinking from a cup), often with head ("no-no" or "yes-yes") and voice involvement, frequently familial (autosomal dominant in many pedigrees), classically improved by small amounts of alcohol, worsened by caffeine, fatigue, and stress. Distinguishing ET from PD hinges on tremor phenomenology (action in ET vs rest in PD), absence of bradykinesia and rigidity, symmetry, and DaTscan normality if needed. Treatment: first-line propranolol 60–320 mg/day or primidone titrated up to 250–750 mg/day; second-line topiramate, gabapentin, or benzodiazepines; refractory cases are excellent candidates for DBS of the ventral intermediate nucleus (VIM) of the thalamus or MR-guided focused ultrasound thalamotomy.
Huntington disease (HD) is an autosomal dominant CAG trinucleotide repeat expansion in the HTT gene on chromosome 4, with anticipation (earlier onset in successive generations, especially paternal inheritance). Typical age of onset 30–50. The classic triad is chorea (involuntary dance-like movements), psychiatric symptoms (depression, irritability, psychosis, suicidality), and progressive cognitive decline/dementia. Atrophy of the caudate nucleus produces the "boxcar" appearance on imaging. Genetic testing confirms diagnosis and guides family counseling. Symptomatic treatment: tetrabenazine, deutetrabenazine, or valbenazine (VMAT2 inhibitors) suppress chorea; SSRIs and atypical antipsychotics for mood and psychosis. No disease-modifying therapy is yet available.
Wilson disease is an autosomal recessive copper-transport disorder (ATP7B gene) presenting in adolescents and young adults with tremor (classically "wing-beating"), dystonia, parkinsonism, cerebellar signs, psychiatric symptoms, liver disease, and Kayser-Fleischer rings on slit-lamp exam. Screen with low serum ceruloplasmin, elevated 24-hour urinary copper, and slit-lamp exam; liver biopsy and genetic testing confirm. Treat with copper chelation (penicillamine, trientine) and zinc (which blocks intestinal copper absorption). Life-long therapy is required.
Dystonia is sustained or intermittent muscle contractions causing twisting, repetitive movements, or abnormal postures. It can be focal (cervical dystonia/spasmodic torticollis, blepharospasm, oromandibular dystonia, writer's cramp, laryngeal dystonia/spasmodic dysphonia), segmental, multifocal, or generalized (often genetic, including DYT1). Secondary dystonia can result from dopamine-blocking medications (tardive), stroke, or metabolic disease. Botulinum toxin injections every 12 weeks are first-line for focal dystonia; trihexyphenidyl, baclofen, and deep brain stimulation of the globus pallidus interna are options for generalized or refractory disease. Tourette syndrome (childhood-onset motor and vocal tics, often with OCD and ADHD) and tardive dyskinesia from chronic dopamine receptor blockade (treated with valbenazine or deutetrabenazine) are additional movement disorders the scribe will encounter.
13 Dementia & Cognitive Disorders Degenerative
Mild cognitive impairment (MCI) is cognitive decline beyond normal aging without functional impairment. Dementia (major neurocognitive disorder) requires cognitive decline sufficient to impair independent function in daily activities.
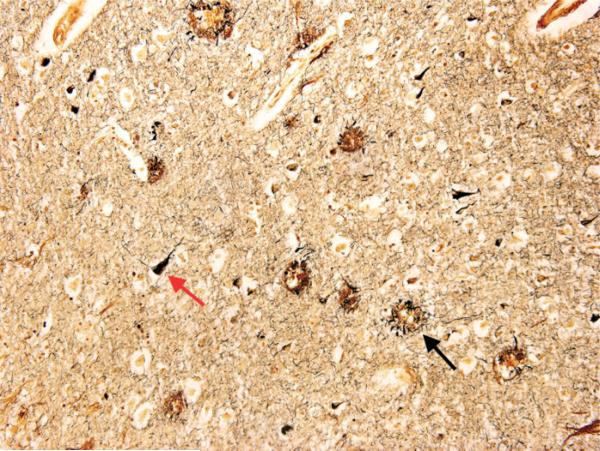
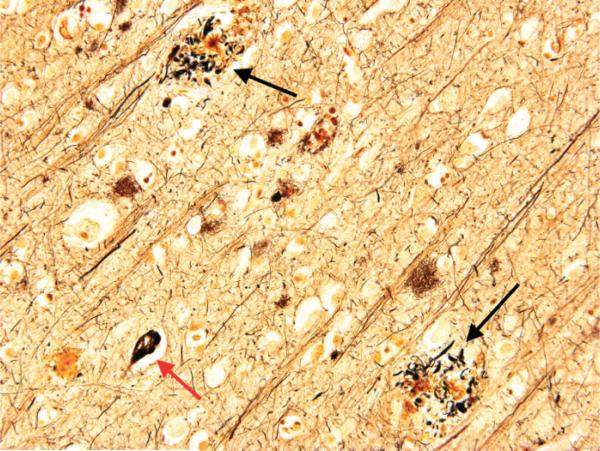
Subtypes
- Alzheimer disease — insidious amnestic (hippocampal) onset, with progressive involvement of language, visuospatial, and executive domains. Biomarkers: CSF Aβ42 low / tau-p high, amyloid PET positive, MRI with hippocampal and medial temporal atrophy.
- Vascular dementia — stepwise decline, focal deficits, executive dysfunction, white-matter disease or prior strokes on imaging.
- Frontotemporal dementia (FTD) — behavioral variant (disinhibition, apathy, compulsions, hyperorality) or primary progressive aphasia variants (semantic, nonfluent). Onset often in 50s–60s.
- Dementia with Lewy bodies (DLB) — see section 11. Neuroleptics can produce severe reactions.
- Normal pressure hydrocephalus — covered in section 17.
Assessment & Treatment
Cognitive screens: MMSE (max 30, <24 suggests dementia; insensitive to MCI and executive dysfunction), MoCA (max 30, ≥26 normal; more sensitive to MCI, executive, visuospatial). Workup includes TSH, B12, RPR, HIV when indicated, MRI brain, and specialty testing (LP amyloid/tau, amyloid/tau PET) when biomarkers will change management. Symptomatic pharmacologic treatment: cholinesterase inhibitors (donepezil, rivastigmine, galantamine) and memantine (NMDA receptor antagonist) for moderate-to-severe Alzheimer. Disease-modifying anti-amyloid monoclonals lecanemab and donanemab are approved for early Alzheimer and require MRI surveillance for ARIA (amyloid-related imaging abnormalities — edema and microhemorrhages). AAN Practice Guideline on Mild Cognitive Impairment is a standard citation.
14 Peripheral Neuropathy Peripheral
Peripheral neuropathy can be classified by distribution (symmetric length-dependent polyneuropathy vs mononeuropathy multiplex vs plexopathy vs radiculopathy vs polyradiculoneuropathy), by fiber type (large fiber — vibration/proprioception/motor; small fiber — pain/temperature/autonomic), and by pathology (axonal vs demyelinating). The most common etiology in North America is diabetes mellitus, producing a classic distal symmetric sensorimotor polyneuropathy in a stocking-glove distribution. Other causes: B12 deficiency (subacute combined degeneration with dorsal column and corticospinal involvement), alcohol, thyroid disease, HIV, chemotherapy (platinum compounds like cisplatin/oxaliplatin, taxanes, vincristine, bortezomib), hereditary (Charcot-Marie-Tooth — most commonly CMT1A from PMP22 duplication), paraneoplastic (anti-Hu, anti-CV2), vasculitic (painful mononeuropathy multiplex, nerve biopsy diagnostic), heavy metals, amyloid (TTR, AL), and idiopathic.
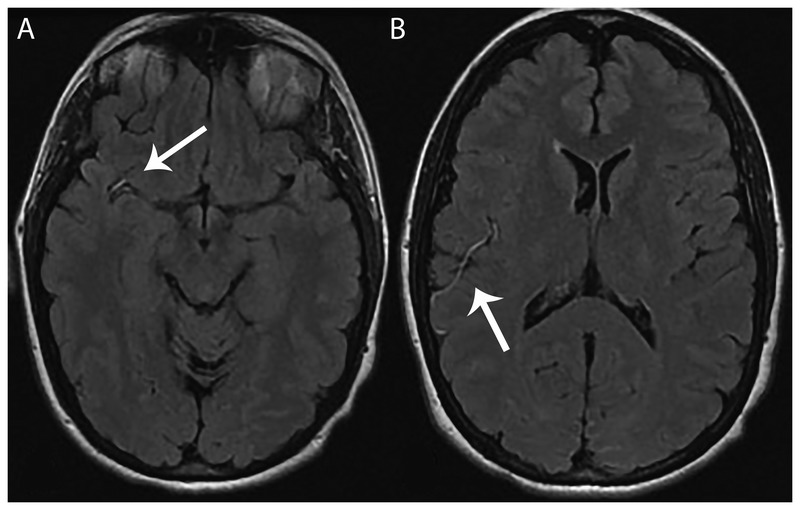
Workup
Labs: A1C, B12 with MMA, TSH, CMP, CBC, SPEP/IFE (monoclonal gammopathy can cause demyelinating neuropathy), HIV, RPR; genetic testing for CMT if hereditary picture. EMG/NCS localizes and characterizes (axonal vs demyelinating). LP for inflammatory demyelinating cases.
GBS and CIDP
Guillain-Barré syndrome (GBS): acute (<4 weeks) ascending symmetric weakness, areflexia, variable sensory loss, often post-infectious (Campylobacter jejuni classically — associated with the AMAN variant and worse prognosis; also CMV, EBV, Zika, SARS-CoV-2, Mycoplasma). Subtypes include acute inflammatory demyelinating polyradiculoneuropathy (AIDP, most common in West), acute motor axonal neuropathy (AMAN), acute motor and sensory axonal neuropathy (AMSAN), and Miller-Fisher syndrome (ophthalmoplegia, ataxia, areflexia, anti-GQ1b antibodies). CSF shows albuminocytologic dissociation (high protein, normal cells) after the first week. Treat with IVIG 2 g/kg over 5 days or plasma exchange (5 sessions); they are equally effective — steroids do NOT help. Monitor forced vital capacity, negative inspiratory force, and bulbar function closely because respiratory failure requiring intubation occurs in ~25%. Autonomic dysfunction (arrhythmias, BP lability, ileus) is a common cause of morbidity. Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) evolves over >8 weeks with symmetric proximal and distal weakness, areflexia, sensory involvement; NCS shows demyelinating features. Treated with steroids, IVIG, SC Ig, or plasma exchange as first-line therapy.
Painful Neuropathy Treatment
First-line neuropathic pain agents: gabapentin, pregabalin, duloxetine, amitriptyline/nortriptyline. Topicals (lidocaine 5% patch, capsaicin 8% patch) add benefit. Avoid chronic opioids.
15 Neuromuscular Junction & Myopathies Neuromuscular
Myasthenia gravis (MG) is an autoimmune disease directed against postsynaptic acetylcholine receptors (AChR Ab positive in ~85% of generalized MG, ~50% of ocular MG) or MuSK (muscle-specific kinase, ~5–10%). A smaller subset is "seronegative." Hallmark is fatigable weakness — ptosis, diplopia, bulbar symptoms (dysarthria, dysphagia, nasal regurgitation), neck flexion weakness, and proximal limb weakness that worsens with activity and improves with rest and cold. Diagnosis relies on antibody testing, bedside ice pack test for ptosis, EMG with repetitive nerve stimulation (>10% decremental response) or single-fiber EMG (jitter), and CT chest for thymoma (present in ~10–15%).
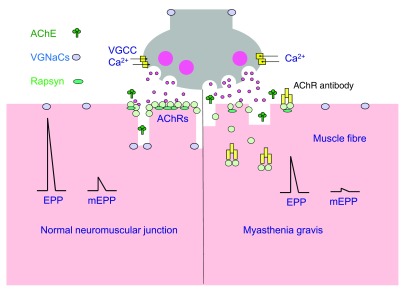
Myasthenic crisis is life-threatening respiratory failure from progressive bulbar and respiratory muscle weakness, treated with IVIG or plasma exchange plus pulse steroids, ICU monitoring, and early intubation before CO2 retention (watch NIF ≤−20 and FVC ≤15–20 mL/kg). Medications to avoid in MG: aminoglycosides, fluoroquinolones, macrolides, beta blockers, magnesium, neuromuscular blockers, and telithromycin — each can precipitate crisis. Chronic therapy: pyridostigmine 30–60 mg q4–6h (acetylcholinesterase inhibitor — symptomatic only), prednisone (start low and titrate up to avoid early worsening), steroid-sparing immunosuppressants (azathioprine, mycophenolate mofetil, tacrolimus, methotrexate, cyclosporine), thymectomy (especially if thymoma, also beneficial in non-thymomatous AChR+ generalized MG per MGTX trial), and newer targeted biologics: eculizumab and ravulizumab (C5 complement inhibitors), efgartigimod and rozanolixizumab (FcRn antagonists). MGFA clinical classification ranges from I (ocular only) through V (intubated).
Lambert-Eaton myasthenic syndrome (LEMS) is a presynaptic disorder (voltage-gated calcium channel antibodies), often paraneoplastic from small-cell lung cancer (~60% of cases), with proximal weakness that paradoxically improves with brief exercise (facilitation), autonomic features (dry mouth, constipation, erectile dysfunction), and reduced or absent reflexes that augment after sustained contraction. EMG shows incremental response with rapid repetitive stimulation. Treatment: evaluate for underlying malignancy, 3,4-diaminopyridine (amifampridine), pyridostigmine, steroids, and immunosuppression.
Myopathies: inflammatory myositis (polymyositis, dermatomyositis with characteristic skin findings — heliotrope rash, Gottron papules, shawl sign; inclusion body myositis with finger-flexor and quadriceps weakness in older men; immune-mediated necrotizing myopathy with SRP or HMGCR antibodies) presents with proximal weakness and elevated CK (sometimes >10,000); EMG shows myopathic units with irritability (fibrillations, sharp waves); muscle biopsy confirms pathology; treated with steroids, IVIG, and steroid-sparing immunosuppression. Dystrophies (Duchenne, Becker, myotonic types 1 and 2, limb-girdle, FSHD) are typically hereditary and diagnosed with genetic testing. Statin-associated myopathy ranges from mild myalgia to rhabdomyolysis to immune-mediated necrotizing myopathy with anti-HMGCR antibodies — the last does not resolve with statin discontinuation and requires immunotherapy. Metabolic myopathies (McArdle, Pompe, mitochondrial) can present with exercise intolerance.
16 CNS Tumors & Neuro-Oncology Oncologic
Primary CNS tumors are classified per WHO with both histology and molecular features. Gliomas include astrocytomas, oligodendrogliomas, and glioblastoma — graded I–IV by WHO with molecular classification integrating IDH mutation status, 1p/19q codeletion (defining oligodendroglioma), ATRX loss, TERT promoter mutation, and MGMT promoter methylation (which predicts temozolomide response). Glioblastoma (WHO grade 4) is the most aggressive, with median survival of ~15 months despite maximal therapy. Meningiomas are usually benign extra-axial tumors arising from arachnoid cap cells and are often incidental findings on imaging. Schwannomas, particularly vestibular schwannoma (acoustic neuroma) at the cerebellopontine angle, present with unilateral sensorineural hearing loss, tinnitus, and disequilibrium. Other primary tumors include ependymoma, medulloblastoma (pediatric), pituitary adenoma, craniopharyngioma, and CNS lymphoma.
Metastatic disease is more common than primary CNS tumors overall — top sources are lung, breast, melanoma, renal, and GI. Mets are typically multiple and at gray-white junction, with prominent vasogenic edema. Leptomeningeal carcinomatosis presents with cranial neuropathies, radiculopathies, and communicating hydrocephalus; CSF cytology and MRI with gadolinium are diagnostic.
Presentations include headache (worse in morning, with Valsalva, progressive), focal deficits, new-onset seizures (up to 30% of brain tumor patients), and cognitive/personality change. Management is multidisciplinary across neurology, neurosurgery, radiation oncology, and medical oncology: dexamethasone for vasogenic edema (typically 4–16 mg/day), antiseizure medications if seizures occur (levetiracetam is preferred because it does not induce hepatic metabolism of chemotherapy), surgical maximal safe resection or biopsy, radiation (whole-brain, stereotactic radiosurgery, or fractionated), temozolomide for glioblastoma following the Stupp protocol, tumor-treating fields (TTF), and increasingly targeted therapies (BRAF/MEK inhibitors, EGFR inhibitors, immune checkpoint inhibitors) selected by molecular profile.
17 CNS Infections & Normal Pressure Hydrocephalus Infectious/CSF
CNS infections overlap with infectious disease, but neurologists are frequently consulted for diagnosis and management. Bacterial meningitis: fever, headache, neck stiffness, altered mental status; classic triad is present in fewer than half of cases, so a single sign plus fever is enough to trigger workup. Empiric therapy in adults is ceftriaxone + vancomycin + dexamethasone; add ampicillin for Listeria coverage in >50 yo, immunocompromised, pregnant, or alcohol use disorder. CSF: high WBC with neutrophilic predominance, low glucose (<40 or CSF/serum <0.4), high protein, positive Gram stain/culture. CT before LP is indicated for immunocompromise, new seizures, focal deficits, papilledema, or reduced GCS to avoid herniation risk.
Viral meningitis (enterovirus commonly) is self-limited — CSF shows lymphocytic pleocytosis, normal glucose, mildly elevated protein. Viral encephalitis (HSV-1 classically) presents with fever, behavioral/personality change, seizures, and temporal lobe findings on MRI; treat empirically with IV acyclovir 10 mg/kg q8h while awaiting CSF HSV PCR. Delaying acyclovir worsens outcomes — start it on suspicion.
Other important CNS infections: tuberculous meningitis (basilar enhancement, cranial neuropathies, hydrocephalus), neurocysticercosis (seizures with ring-enhancing or calcified cystic lesions in immigrants from endemic regions), cryptococcal meningitis (AIDS and other immunocompromised, elevated OP, positive cryptococcal antigen), toxoplasmosis (ring-enhancing lesions in AIDS), Lyme neuroborreliosis (facial palsy, radiculitis, aseptic meningitis), neurosyphilis (RPR and CSF VDRL), and progressive multifocal leukoencephalopathy (PML) from JC virus reactivation in immunocompromised, particularly natalizumab patients — always screen JC virus serology before starting natalizumab and during therapy.
Autoimmune encephalitis (anti-NMDA receptor, anti-LGI1, anti-CASPR2, GAD65, etc.) is an increasingly recognized mimic presenting with psychiatric symptoms, seizures, memory loss, and movement abnormalities; treat with immunotherapy (steroids, IVIG, plasma exchange, rituximab) and evaluate for underlying tumor (ovarian teratoma in anti-NMDA).
Normal pressure hydrocephalus (NPH) is the classic triad of gait apraxia ("magnetic" or "glue-foot" gait), urinary incontinence, and cognitive decline ("wet, wacky, wobbly"). MRI shows ventriculomegaly out of proportion to cortical atrophy (Evans index > 0.3). Large-volume LP (~30–50 mL removal) or external lumbar drainage trial assesses whether ventriculoperitoneal shunting will help — gait typically responds best, cognition least reliably.
18 Vertigo & Dizziness Vestibular
First distinguish vertigo (illusion of motion) from presyncope, disequilibrium, and non-specific dizziness. Then separate peripheral (inner ear/vestibular nerve) from central (brainstem/cerebellum) causes. The HINTS exam (Head Impulse, Nystagmus, Test of Skew) in acute vestibular syndrome is more sensitive than early MRI for posterior fossa stroke when performed by an experienced examiner. A "benign" HINTS pattern is: abnormal (positive, corrective saccade) head impulse, unidirectional horizontal nystagmus, and no vertical skew — consistent with peripheral cause. Any finding of normal head impulse, direction-changing nystagmus, or skew deviation suggests central etiology ("dangerous HINTS") and warrants urgent MRI and stroke workup.
Peripheral Vertigo
- BPPV — brief (< 1 min) positional vertigo from otoconia in semicircular canals. Diagnosed with Dix-Hallpike; treated with Epley maneuver.
- Vestibular neuritis — acute prolonged vertigo lasting days, often post-viral, without hearing loss. Labyrinthitis adds hearing loss. Short vestibular suppressants + early ambulation + vestibular rehab.
- Ménière disease — episodic vertigo lasting 20 minutes to hours with fluctuating low-frequency hearing loss, tinnitus, aural fullness. Low-salt diet, diuretics, intratympanic steroids or gentamicin for refractory disease.
Central vertigo (cerebellar/brainstem stroke, MS, tumor) — any vertical or direction-changing nystagmus, gait ataxia out of proportion to vertigo, or other brainstem findings warrants urgent MRI.
19 Neurologic Procedures & Diagnostics Procedural
Neurology is a largely diagnostic specialty, but a scribe will document a defined set of procedures performed in clinic, at the bedside, or in specialized labs.
| Procedure | Description / Indication |
|---|---|
| Lumbar puncture (LP) | CSF sampling for meningitis, SAH workup, MS, GBS, idiopathic intracranial hypertension (measure opening pressure), and intrathecal drug delivery. Document opening pressure, cell counts, protein, glucose, Gram stain, cultures, and specialty studies ordered. |
| Routine EEG | 20-minute scalp recording with activation procedures (hyperventilation, photic stimulation). Used to characterize seizures, evaluate AMS. |
| Prolonged / ambulatory EEG | 24–72 hours of recording to capture infrequent events. |
| Video-EEG monitoring (vEEG) | Inpatient epilepsy monitoring unit for seizure characterization, medication adjustment, and pre-surgical evaluation. |
| Continuous EEG (cEEG) | ICU monitoring for non-convulsive status epilepticus and post-cardiac arrest prognostication. |
| EMG / NCS | Needle electromyography plus nerve conduction studies to diagnose radiculopathy, neuropathy, NMJ disorders, and myopathy. |
| Evoked potentials | Visual (VEP), brainstem auditory (BAEP), and somatosensory (SSEP) — used for MS and intraoperative monitoring. |
| Occipital nerve block | Local anesthetic ± steroid for occipital neuralgia and refractory migraine. |
| Sphenopalatine ganglion block | Transnasal for cluster and refractory migraine. |
| OnabotulinumtoxinA injections | Chronic migraine (PREEMPT protocol, 31-155 units across 31 sites), cervical dystonia, blepharospasm, hemifacial spasm, limb spasticity. |
| Intrathecal baclofen pump refill | Severe spasticity from MS, SCI, CP. |
| Nusinersen intrathecal dosing | SMA treatment. |
| Deep brain stimulation (DBS) | Implanted by neurosurgery; programming and management done by movement disorders neurologists for PD, essential tremor, dystonia. |
| Plasma exchange / IVIG | Ordered for GBS, myasthenic crisis, CIDP, MS relapse, NMO. |
For every LP, chart the opening pressure, the appearance of the CSF ("clear and colorless" vs "bloody" vs "xanthochromic"), the exact tube counts with cell differentials, and every send-out study ordered. Missing studies means another LP — patients hate that.
20 Neuroimaging & Electrodiagnostics Diagnostic
NCCT head: first-line for stroke alert to rule out hemorrhage, SAH (up to 6 hours), skull fracture, large mass effect. CTA head and neck: evaluates for LVO, aneurysm, dissection, vasculitis. CT perfusion: estimates ischemic core and salvageable penumbra using CBF/CBV/Tmax maps — supports late-window thrombectomy decisions. MRI brain: DWI is the most sensitive sequence for acute infarct within minutes of onset; FLAIR shows edema and demyelination; T2*/SWI detects microbleeds and hemorrhage; T1 with gadolinium identifies active inflammation, blood-brain barrier breakdown, and tumors. MR angiography/venography: non-contrast time-of-flight or contrast-enhanced vascular imaging. Catheter angiography (DSA): gold standard for vascular anatomy and therapeutic access.
EEG Waveforms & Terms
You will hear phrases like: alpha rhythm (normal posterior dominant rhythm ~8–13 Hz), beta, theta, delta, spike-and-wave (epileptiform), generalized periodic discharges (GPDs), lateralized periodic discharges (LPDs), triphasic waves (metabolic encephalopathy), burst suppression (deep sedation, severe encephalopathy).
EMG/NCS Findings
Neurogenic/axonal: reduced CMAP amplitude with relatively preserved velocities; EMG shows fibrillation potentials, positive sharp waves, and large long-duration polyphasic motor units. Demyelinating: slow conduction velocities, prolonged distal latencies, conduction block, temporal dispersion. Myopathic: small short-duration polyphasic motor units with early recruitment. NMJ disorders: decrement on repetitive stimulation (MG, > 10% drop) or increment with rapid stimulation/exercise (LEMS).
21 Medications You Must Know Pharmacology
| Class | Agents | Primary Use / Notes |
|---|---|---|
| Antiplatelets | Aspirin, clopidogrel, aspirin+dipyridamole, ticagrelor | Secondary stroke prevention; DAPT 21–90 days after minor stroke/high-risk TIA |
| Anticoagulants | Warfarin, apixaban, rivaroxaban, dabigatran, edoxaban, LMWH, UFH | Cardioembolic stroke, CVST; DOACs preferred in non-valvular AFib |
| Thrombolytics | Alteplase (tPA), tenecteplase (TNK) | Acute ischemic stroke within 4.5 h window |
| Statins | Atorvastatin 40–80 mg, rosuvastatin 20–40 mg | High-intensity for all ischemic stroke/TIA |
| Broad-spectrum ASMs | Lamotrigine, levetiracetam, valproate, topiramate, zonisamide, lacosamide | Focal and generalized epilepsy |
| Narrow-spectrum ASMs | Carbamazepine, oxcarbazepine, phenytoin, eslicarbazepine, gabapentin, pregabalin | Focal epilepsy; CBZ/OXC also trigeminal neuralgia |
| Acute migraine | Sumatriptan, rizatriptan, eletriptan, ubrogepant, rimegepant, lasmiditan, NSAIDs | Triptans contraindicated in CAD/uncontrolled HTN |
| Migraine prevention | Propranolol, topiramate, valproate, amitriptyline, candesartan, erenumab, fremanezumab, galcanezumab, eptinezumab, atogepant, rimegepant | CGRP mAbs and gepants are newer options |
| Dopaminergic | Carbidopa/levodopa, pramipexole, ropinirole, rotigotine, apomorphine | PD first-line for motor symptoms |
| MAO-B / COMT inhibitors | Selegiline, rasagiline, safinamide; entacapone, opicapone, tolcapone | Adjuncts to levodopa |
| Anticholinesterases (dementia) | Donepezil, rivastigmine, galantamine | Alzheimer, Parkinson disease dementia |
| NMDA antagonist | Memantine | Moderate-to-severe Alzheimer |
| Anti-amyloid mAbs | Lecanemab, donanemab | Early Alzheimer; monitor ARIA on MRI |
| MS DMTs — injectable | Interferon beta 1a/1b, glatiramer acetate | Lower efficacy, long safety record |
| MS DMTs — oral | Dimethyl/diroximel fumarate, teriflunomide, fingolimod, ozanimod, ponesimod, siponimod, cladribine | S1P modulators require first-dose cardiac monitoring |
| MS DMTs — monoclonals | Natalizumab, ocrelizumab, ofatumumab, ublituximab, alemtuzumab | Natalizumab — PML risk, check JC virus |
| NMOSD | Eculizumab, inebilizumab, satralizumab, rituximab | Do NOT use standard MS DMTs |
| ALS | Riluzole, edaravone, tofersen (SOD1) | Modest survival / functional benefit |
| Myasthenia gravis | Pyridostigmine, prednisone, azathioprine, mycophenolate, eculizumab, efgartigimod, rozanolixizumab | Avoid drugs that worsen MG (aminoglycosides, fluoroquinolones, beta blockers, magnesium) |
| Neuropathic pain | Gabapentin, pregabalin, duloxetine, amitriptyline, nortriptyline, lidocaine patch | First-line for diabetic neuropathy, PHN |
| Spasticity | Baclofen (PO or intrathecal), tizanidine, dantrolene, botulinum toxin | MS, SCI, post-stroke, CP |
22 Classification & Scoring Systems Reference
15-item scale (0–42). Items: 1a LOC, 1b LOC questions, 1c LOC commands, 2 best gaze, 3 visual fields, 4 facial palsy, 5a/5b motor arm L/R, 6a/6b motor leg L/R, 7 limb ataxia, 8 sensory, 9 best language, 10 dysarthria, 11 extinction/inattention. Severity: 0 no deficit; 1–4 minor; 5–15 moderate; 16–20 moderate-severe; 21–42 severe.
10-point topographic score on NCCT; one point subtracted for each MCA region with early ischemic change. 10 = normal; ≤7 correlates with worse outcome and larger infarct; historically ASPECTS ≥6 was often required for thrombectomy, though recent trials extend eligibility to lower scores.
I asymptomatic or mild headache; II moderate-severe headache, nuchal rigidity, no deficit except CN palsy; III drowsy, confused, mild focal deficit; IV stupor, moderate-severe hemiparesis; V deep coma, decerebrate posturing, moribund.
0 no SAH or IVH; 1 minimal/thin SAH without IVH; 2 minimal/thin SAH with IVH; 3 thick SAH without IVH; 4 thick SAH with IVH. Higher grades predict vasospasm.
0 no symptoms; 1 no significant disability; 2 slight disability (unable to carry out all previous activities but independent in ADLs); 3 moderate disability (requires some help, walks unassisted); 4 moderately severe (unable to walk or attend to own bodily needs without assistance); 5 severe (bedridden, incontinent); 6 dead. Favorable outcome is typically mRS 0–2.
GCS 3–4 = 2, 5–12 = 1, 13–15 = 0; ICH volume ≥30 mL = 1; IVH present = 1; infratentorial origin = 1; age ≥80 = 1. Total 0–6 predicts 30-day mortality (0 ~0%, 5–6 ~100%).
Eye (1–4): 1 none, 2 pain, 3 voice, 4 spontaneous. Verbal (1–5): 1 none, 2 incomprehensible, 3 inappropriate words, 4 confused, 5 oriented. Motor (1–6): 1 none, 2 extension, 3 flexion abnormal, 4 withdraws, 5 localizes, 6 obeys commands. Range 3–15; ≤8 typically prompts intubation.
MMSE 30-point: orientation (10), registration (3), attention (5), recall (3), language (8), construction (1). Thresholds: 24–30 normal, 18–23 mild, <18 severe. MoCA 30-point adds executive, visuospatial, abstraction; ≥26 normal, sensitive to MCI.
I unilateral involvement; II bilateral without balance impairment; III mild-to-moderate bilateral with postural instability, physically independent; IV severe disability, still able to walk or stand unassisted; V wheelchair-bound or bedridden unless aided.
0 normal neurologic exam; 1–3.5 minimal to mild disability with functional systems scores; 4 fully ambulatory without aid for 500 m; 6 intermittent or unilateral assistance to walk 100 m; 7 essentially restricted to wheelchair; 8 essentially bedridden/chair; 9 helpless bed patient; 10 death due to MS.
I ocular only; II mild generalized (IIa limb, IIb bulbar/respiratory); III moderate generalized (IIIa/IIIb); IV severe generalized (IVa/IVb); V requires intubation.
23 Neurologic Physical Exam Reference
The neurologic exam is the most specialty-defining part of the note. Master the components and the documentation rhythm.
Mental status: alert and oriented x 3 (or 4), attention (months backward, serial 7s), language (fluent, comprehension intact, naming, repetition), memory (3-item recall at 5 min), fund of knowledge, calculation, praxis, neglect.
Cranial nerves: II visual acuity and fields to confrontation, pupils equal round reactive to light and accommodation (PERRLA), fundoscopy if relevant; III/IV/VI extraocular movements full, no nystagmus, no ptosis; V facial sensation intact V1-V3, masseter strength; VII facial symmetry at rest and with activation; VIII hearing intact to finger rub; IX/X palate elevation symmetric, gag; XI SCM and trapezius; XII tongue midline.
Motor: bulk and tone normal, no drift. Strength by MRC 0–5 in deltoids, biceps, triceps, wrist ext/flex, grip, finger abduction, iliopsoas, quads, hamstrings, tibialis anterior, gastroc, EHL.
Reflexes: biceps, triceps, brachioradialis, patellar, Achilles (graded 0–4+); plantar responses (flexor = normal, extensor = Babinski sign).
Sensory: light touch, pinprick, vibration, proprioception, temperature. Document by dermatome when abnormal.
Coordination: finger-to-nose, heel-to-shin, rapid alternating movements, no dysmetria.
Gait: casual gait normal base and stride, tandem, Romberg, heel and toe walking.
MRC scale for motor strength: 0 no contraction, 1 flicker/trace, 2 active movement with gravity eliminated, 3 against gravity only, 4 against some resistance (can be 4-, 4, 4+), 5 normal. Reflex scale: 0 absent, 1+ diminished, 2+ normal, 3+ brisker than normal, 4+ clonus.
24 Abbreviations, Sample HPIs & References
Abbreviations — Diagnoses & Syndromes
Abbreviations — Procedures, Imaging & Exam
Abbreviations — Medications
Sample HPI Templates
"Mr. [Name] is a 72-year-old right-handed male with a history of hypertension, hyperlipidemia, type 2 diabetes, and paroxysmal atrial fibrillation (on apixaban 5 mg BID) who presents as a stroke alert. Last known well was 0745 this morning when his wife saw him reading the paper. At 0830 she found him with right facial droop, right arm and leg weakness, and slurred/non-fluent speech. EMS arrival 0848, ED arrival 0902. On arrival BP 182/96, HR 88 irregular, glucose 142. Exam: NIHSS 14 (1a-0, 1b-1, 1c-1, 2-0, 3-0, 4-2, 5a-3, 5b-2, 6a-2, 6b-1, 7-0, 8-1, 9-1, 10-0, 11-0). NCCT at 0918: no hemorrhage, ASPECTS 9. CTA: left M1 occlusion. Patient is outside the tPA window from LKW but within 6 hours — activating neuro-IR for mechanical thrombectomy."
"Ms. [Name] is a 34-year-old female with chronic migraine without aura (17 headache days/month at baseline) who returns for follow-up. Since starting erenumab 70 mg SC monthly 3 months ago, headache days have decreased to 9 per month. Attacks are still unilateral throbbing 7/10 with nausea, photophobia, and phonophobia, lasting 6–12 hours. She aborts with sumatriptan 100 mg PO with good response in 2/3 attacks; no triptan-limiting side effects. No medication overuse (triptan use <10 days/month). Denies new neurologic symptoms, weakness, vision change, or aura phenomena. Last Botox PREEMPT cycle was 10 weeks ago."
"Mr. [Name] is a 28-year-old man presenting for evaluation of a first-time witnessed generalized tonic-clonic seizure. Event occurred yesterday at 0600 after 3 hours of sleep following a night shift. Wife witnessed sudden stiffening followed by ~90 seconds of bilateral rhythmic limb jerking, eyes rolled up, tongue bitten on the left lateral aspect, with urinary incontinence. Post-event confusion lasted ~30 minutes. He denies prior seizures, head trauma, CNS infection, or family history of epilepsy. Social history notable for 4 beers nightly (not binge drinking), no illicit drugs. Exam today is non-focal. MRI brain without contrast and routine EEG are pending; labs including CBC, BMP, magnesium, and urine toxicology are unremarkable."
"Mrs. [Name] is a 73-year-old right-handed woman brought in by her daughter for evaluation of progressive memory decline over the past 2 years. She repeats questions within the same conversation, has forgotten recent family events, misplaces items, and has stopped managing her checkbook (her daughter took over 6 months ago). She has had two episodes of getting lost while driving in familiar areas. She denies hallucinations, REM sleep behavior disorder, or prominent motor symptoms. No stepwise decline. PMH: hypertension, hypothyroidism. Family history: mother had dementia diagnosed age 78. MoCA today 19/30 with deficits in delayed recall (0/5), visuospatial (cube and clock), and orientation. MRI brain shows bilateral hippocampal atrophy out of proportion to global volume loss. Presentation most consistent with probable Alzheimer disease, early stage."
"Ms. [Name] is a 56-year-old woman with hypertension and hyperlipidemia presenting with acute-onset continuous vertigo and imbalance since waking this morning (~6 hours ago). Describes constant spinning worsened by head movement, with nausea and one episode of vomiting. Denies hearing loss, tinnitus, diplopia, dysarthria, facial droop, limb weakness, or numbness. On exam, alert, spontaneous left-beating horizontal nystagmus with rotary component, positive head impulse test to the right (corrective saccade), no skew deviation — HINTS benign, consistent with peripheral vestibular neuritis rather than central cause. Gait wide-based but able to walk. Started on meclizine PRN, short steroid course considered, early vestibular rehabilitation to follow."
"Mr. [Name] is a 64-year-old man with poorly controlled type 2 diabetes (A1C 9.8) presenting with 18 months of progressive bilateral foot numbness and burning pain. Symptoms began in the toes and have ascended to the mid-calf in a stocking distribution. Pain is worst at night, described as burning with intermittent electric shocks, 6/10. He has no upper extremity symptoms (yet), no weakness, no bowel or bladder complaints. PMH: T2DM x 15 years, HLD, HTN. Medications: metformin, glipizide, lisinopril. Denies alcohol excess. Exam: reduced pinprick and vibration to mid-calf, absent ankle reflexes, preserved strength, normal gait. Monofilament testing abnormal at great toe bilaterally. Plan: confirm with EMG/NCS, screen for reversible causes (B12, TSH, SPEP), start duloxetine 30 mg daily for neuropathic pain, intensify diabetes management."
References & Sources
Clinical Practice Guidelines
Postuma RB et al. MDS Clinical Diagnostic Criteria for Parkinson's Disease. Mov Disord. 2015.
Petersen RC et al. Practice Guideline Update Summary: Mild Cognitive Impairment. Neurology. 2018.
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: Overview of the Brain. OpenStax College. CC BY 3.0.
Figure 2: The Cerebrum. OpenStax College. CC BY 3.0.
Figure 3: Brainstem. OpenStax College. CC BY 3.0.
Figure 4: Brain and Spinal Cord. OpenStax College. CC BY 3.0.
Figure 5: Sensory Homunculus. OpenStax College. CC BY 3.0.
Figure 6: Stroke. Blausen Medical. CC BY 3.0.
Figure 7: Dementia. Blausen Medical. CC BY 3.0.
Great neurology scribes think in terms of localization and temporal profile. When the attending says "hyperacute onset right face-arm weakness with aphasia," you should already be anticipating an MCA stroke assessment and pre-populating the NIHSS template. When she says "slowly progressive proximal weakness with elevated CK," you know myopathy, not neuropathy, and you pull up the dermatomyositis labs. When the patient describes "the worst headache of my life, started while lifting something," you know SAH is in play and the team will want NCCT followed by LP if negative.
The neurologic exam is long. Learn the attending's shorthand. Know the difference between "2+ symmetric reflexes with downgoing toes" (normal) and "3+ reflexes with sustained clonus and upgoing toes" (UMN lesion). Memorize the NIHSS items so you can chart them in real time during a stroke alert. Understand why the attending stopped levodopa in a DLB patient or why natalizumab requires JC virus testing before every cycle.
This guide gives you the scaffolding. The expertise comes from doing it — shift after shift, patient after patient — until the localization, the differential, and the documentation rhythm become automatic. Welcome to neurology.