Neurosurgery
Every diagnosis, procedure, medication, classification, and documentation framework a neurosurgery scribe needs to function from day one in clinic, OR, and ICU.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Neuroanatomy Essentials
Neurosurgery covers the entire central nervous system — brain, spinal cord, meninges, ventricular system — plus the peripheral nerves and the bony, vascular, and CSF compartments that surround them. You do not need to memorize a medical school neuroanatomy textbook, but you do need enough fluency to translate a surgeon saying "left frontotemporal craniotomy for a sphenoid wing meningioma abutting the MCA bifurcation" into an accurate, immediately legible note.
The Cerebrum and Lobes
The cerebrum is divided into two hemispheres connected by the corpus callosum. Each hemisphere contains four lobes. The frontal lobe controls voluntary movement (primary motor cortex, precentral gyrus), executive function, personality, and expressive language (Broca's area, usually left inferior frontal gyrus). The parietal lobe handles somatosensation (primary sensory cortex, postcentral gyrus) and spatial awareness. The temporal lobe handles hearing, memory (hippocampus), and receptive language (Wernicke's area, usually left superior temporal gyrus). The occipital lobe processes vision (primary visual cortex, calcarine fissure). Deep to the lobes lie the basal ganglia (caudate, putamen, globus pallidus, subthalamic nucleus, substantia nigra) and thalamus — the functional targets of deep brain stimulation.
Brainstem, Cerebellum, and Cranial Nerves
The brainstem (midbrain, pons, medulla) contains the reticular activating system (arousal), cranial nerve nuclei III–XII, and the motor/sensory long tracts. The cerebellum sits in the posterior fossa behind the brainstem and coordinates movement and balance. The twelve cranial nerves are numbered and must be documented when relevant: I olfactory, II optic, III oculomotor, IV trochlear, V trigeminal, VI abducens, VII facial, VIII vestibulocochlear, IX glossopharyngeal, X vagus, XI accessory, XII hypoglossal. Know the mnemonic and the function cold — a surgeon operating in the cerebellopontine angle for a vestibular schwannoma will rapid-fire comment on CN V, VII, and VIII function.
Meninges, Ventricles, and CSF
Three layers cover the brain: the tough outer dura mater, the thin middle arachnoid mater, and the delicate inner pia mater adherent to the cortex. Blood collections are named by the compartment they occupy: an epidural hematoma sits between skull and dura, a subdural hematoma between dura and arachnoid, and a subarachnoid hemorrhage in the CSF-filled space between arachnoid and pia. CSF is produced by the choroid plexus in the ventricles, flows from the lateral ventricles through the foramen of Monro into the third ventricle, through the cerebral aqueduct into the fourth ventricle, then out the foramina of Luschka and Magendie into the subarachnoid space before reabsorption at the arachnoid granulations. Normal adult CSF volume is roughly 150 mL with production near 500 mL/day.
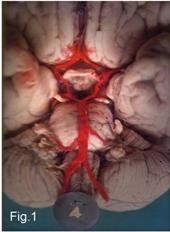
Cerebrovascular Anatomy
Arterial supply to the brain comes from two paired systems: the internal carotid arteries (anterior circulation) and the vertebral arteries (posterior circulation), which join at the circle of Willis at the base of the brain. Major branches include the anterior cerebral artery (ACA), middle cerebral artery (MCA), posterior cerebral artery (PCA), basilar artery, posterior inferior cerebellar artery (PICA), anterior inferior cerebellar artery (AICA), and superior cerebellar artery (SCA). Aneurysms most commonly occur at branch points in the circle of Willis — anterior communicating (ACom), posterior communicating (PCom), MCA bifurcation, and basilar tip.
Spinal Anatomy
The vertebral column has 7 cervical, 12 thoracic, 5 lumbar, 5 fused sacral, and 4 fused coccygeal vertebrae. The spinal cord runs from the medulla to the conus medullaris at about L1–L2 in adults; below this point the nerve roots continue as the cauda equina. Each vertebral level has a pair of nerve roots exiting through the neural foramina. The cord is surrounded by the same three meningeal layers as the brain, with the epidural space outside the dura containing fat and venous plexus (the epidural space targeted by epidural injections). Intervertebral discs sit between vertebral bodies and have a central nucleus pulposus surrounded by a fibrous annulus fibrosus.
Learn laterality. A neurosurgeon will almost never say "the patient has a tumor" without a side and a lobe. If you document "right frontal" when the surgeon said "left frontal," that is a wrong-site error waiting to happen. Always repeat laterality back to yourself as you type.
02 Scribe Documentation Framework
Neurosurgery encounters span clinic consults, pre-op evaluations, inpatient progress notes, ICU notes on EVD/ICP patients, emergency consults for TBI or SAH, and post-op follow-ups. The note format shifts with the venue, but the content backbone is consistent.
Chief Complaint: The anchor of the note — "worsening right leg weakness," "4 weeks of occipital headache," "post-op day 2 s/p right frontal craniotomy for GBM."
HPI: Use OLDCARTS, but always add neurosurgery-specific anchors: onset (sudden vs gradual), associated neurological symptoms (weakness, numbness, vision change, speech, bowel/bladder), headache characteristics ("worst headache of life" language is a red flag for SAH), trauma mechanism, loss of consciousness, post-traumatic amnesia, seizure history, and any prior imaging. For spine patients, always document radicular distribution, walking tolerance, neurogenic claudication, bowel/bladder function, and saddle anesthesia.
ROS: Problem-focused. Neurological ROS is mandatory; also ask about constitutional symptoms in tumor patients, fevers/IV drug use in possible abscess, and bleeding/clotting history before any craniotomy.
PMHx/PSHx/Meds/Allergies/Social/FHx: Capture anticoagulants and antiplatelets on every patient, seizure history, prior neurosurgical procedures and dates, prior radiation, smoking, alcohol, IV drug use, and occupational neurological exposures.
Vitals: BP (target varies by diagnosis: <140 systolic for ICH/SAH, MAP >80–90 for SCI), HR, SpO2, and for ICU patients the ICP, CPP, and temperature.
Neuro exam: Documented in a standardized sequence — GCS or level of arousal, orientation, cranial nerves II–XII, motor strength (MRC 0–5) in all four extremities, sensation, reflexes, coordination, gait. For spine patients, dermatomal sensation, myotomal strength, reflexes, and long-tract signs (Hoffman, Babinski, clonus).
Data: Always document specific imaging findings the surgeon references. "CT head demonstrates a 14 mm right holohemispheric acute SDH with 6 mm midline shift and effacement of the right lateral ventricle" is far more useful than "head CT abnormal."
List each active problem with the current working diagnosis, then the plan. Neurosurgical plans often include operative booking, EVD management, antiepileptic prophylaxis, DVT prophylaxis timing (high-stakes decision after intracranial bleeds), blood pressure targets, osmotic therapy, steroid dosing for tumor edema, and follow-up imaging schedule. Always document laterality and specific procedure name for any planned surgery.
Never chart "neuro intact." That phrase is meaningless and unsafe in neurosurgery. Chart the exam: "A&Ox3, follows commands, PERRL, EOMI, face symmetric, tongue midline, MAE 5/5, sensation intact to light touch, gait steady." If you did not see it, do not chart it.
03 Traumatic Brain Injury (TBI) Cranial
TBI is one of the most common reasons neurosurgery is consulted in any hospital. Scribes are expected to document the mechanism, initial GCS, pupil exam, imaging findings, and the neurosurgical disposition (observation, ICU, OR) with precision. Severity is graded by initial GCS: mild (13–15), moderate (9–12), and severe (3–8). The Brain Trauma Foundation Guidelines for the Management of Severe TBI (4th edition) are the reference standard for ICP, CPP, and craniectomy decision-making.
Concussion (Mild TBI)
A transient alteration in brain function after head trauma without structural injury on conventional imaging. Patients present with headache, dizziness, nausea, amnesia, and photophobia. The diagnosis is clinical. CT is used to rule out structural bleed when clinical decision rules (Canadian CT Head Rule, New Orleans Criteria) indicate imaging. Management is symptomatic; supervised return to play and cognitive rest.
Cerebral Contusion
Bruising of the brain parenchyma, classically at the frontal and temporal poles from coup/contrecoup injury. Contusions "blossom" — they often enlarge in the first 24–72 hours, so repeat imaging is standard. Large or expanding contusions with mass effect may require evacuation.
Epidural Hematoma (EDH)
Arterial bleed (typically middle meningeal artery) between skull and dura. Classically follows a temporal bone fracture. Presentation: transient loss of consciousness followed by a lucid interval, then rapid deterioration as the hematoma expands. CT shows a biconvex (lens-shaped) hyperdensity that does not cross suture lines. Surgical emergency — craniotomy for evacuation is life-saving.
Subdural Hematoma (SDH)
Venous bleed (bridging veins) between dura and arachnoid. CT shows a crescent-shaped hyperdensity that crosses sutures but not the midline. Acute SDH is hyperdense, subacute (3–21 days) is isodense, and chronic SDH is hypodense and may have layered or membrane-contained blood of mixed density. Management ranges from observation (small, stable), burr hole evacuation (chronic or subacute liquefied), to craniotomy (acute with significant mass effect). Chronic SDH in the elderly is graded by the Markwalder grade (see Section 23).
Traumatic Intracerebral Hemorrhage (tICH)
Bleeding within the brain parenchyma from trauma. Often multifocal, often in the frontal/temporal poles. Surgical evacuation considered for lobar hematomas with mass effect and neurological decline.
Diffuse Axonal Injury (DAI)
Shearing of axons from rotational acceleration/deceleration forces. CT may show small petechial hemorrhages at the gray–white junction, corpus callosum, and dorsolateral brainstem. MRI (especially SWI/GRE) is more sensitive. There is no surgical treatment — management is supportive with ICP control.
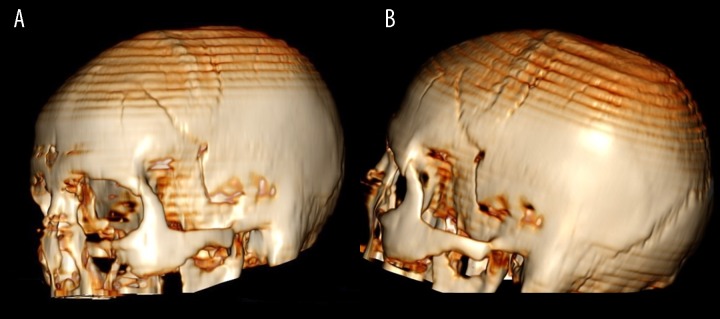
Skull Fractures
Types include linear (most common), depressed (surgery if >1 cm depression, compound, or overlying a dural venous sinus), basilar (racoon eyes, Battle sign, CSF rhinorrhea or otorrhea, hemotympanum), and diastatic (through a suture). Basilar skull fractures raise concern for CSF leak and carotid canal involvement requiring CTA.
GCS drop by ≥2 points, new pupillary asymmetry, Cushing response (hypertension, bradycardia, irregular respirations), posturing (decorticate or decerebrate), or seizure in an unmonitored patient all mandate immediate physician notification and repeat imaging. Document the time of the change and the time of notification.
Always document the initial GCS broken out into its three components (E, V, M) rather than just a total. "GCS 9 (E2 V3 M4)" tells the next team far more than "GCS 9." The motor score is the strongest predictor of outcome.
04 Intracranial Pressure, Edema & Herniation Cranial
The skull is a closed box. Any increase in the volume of brain, blood, or CSF must be offset by a decrease in another compartment (Monro–Kellie doctrine) or ICP will rise. Normal adult ICP is 7–15 mmHg. Sustained ICP >22 mmHg in severe TBI is associated with worse outcomes and is the standard threshold for escalation per BTF guidelines. Cerebral perfusion pressure (CPP) = MAP − ICP; target CPP in severe TBI is typically 60–70 mmHg.
Cerebral Edema
Two main types: vasogenic (breakdown of the blood–brain barrier, responds to steroids, classic around tumors and abscesses) and cytotoxic (cellular swelling from ischemia, does not respond to steroids). Recognize which type the surgeon is treating because it drives the plan.
Herniation Syndromes
| Syndrome | Mechanism | Clinical |
|---|---|---|
| Subfalcine | Cingulate gyrus under falx | Contralateral leg weakness if ACA compressed |
| Uncal (transtentorial) | Medial temporal (uncus) over tentorium | Ipsilateral CN III palsy (blown pupil), contralateral hemiparesis |
| Central transtentorial | Diencephalon downward through tentorium | Progressive decreased arousal, small reactive pupils, then posturing |
| Tonsillar | Cerebellar tonsils through foramen magnum | Brainstem compression, apnea, cardiovascular collapse |
| Upward (reverse) transtentorial | Posterior fossa mass pushes up through tentorium | Can be precipitated by over-aggressive EVD drainage with posterior fossa mass |
ICP Management Tiered Approach
Tier 1: head of bed 30°, head midline, normothermia, sedation/analgesia, normocapnia, adequate MAP for CPP target. Tier 2: osmotic therapy with mannitol (0.25–1 g/kg) or hypertonic saline (3% infusion or 23.4% bolus), ventricular CSF drainage via EVD, mild hyperventilation (PaCO2 30–35 mmHg, short term). Tier 3: decompressive craniectomy, barbiturate coma, therapeutic hypothermia (rescue). See Seattle International Severe TBI Consensus Conference (SIBICC) for the modern tier framework.
Whenever the attending says "bolt" or "the bolt is reading," they mean the parenchymal ICP monitor. When they say "drain" or "EVD," they mean the external ventricular drain, which can both measure and drain CSF. Do not confuse the two in the note — they are different devices with different complications.
05 Subarachnoid Hemorrhage & Cerebral Aneurysms Cranial
Nontraumatic SAH is most commonly caused by rupture of a saccular (berry) aneurysm at the circle of Willis. Classic presentation: sudden onset "worst headache of life" (thunderclap headache), meningismus, photophobia, possible LOC. Mortality approaches 30% at 30 days. 2023 AHA/ASA Guidelines for the Management of Aneurysmal SAH are the current reference.
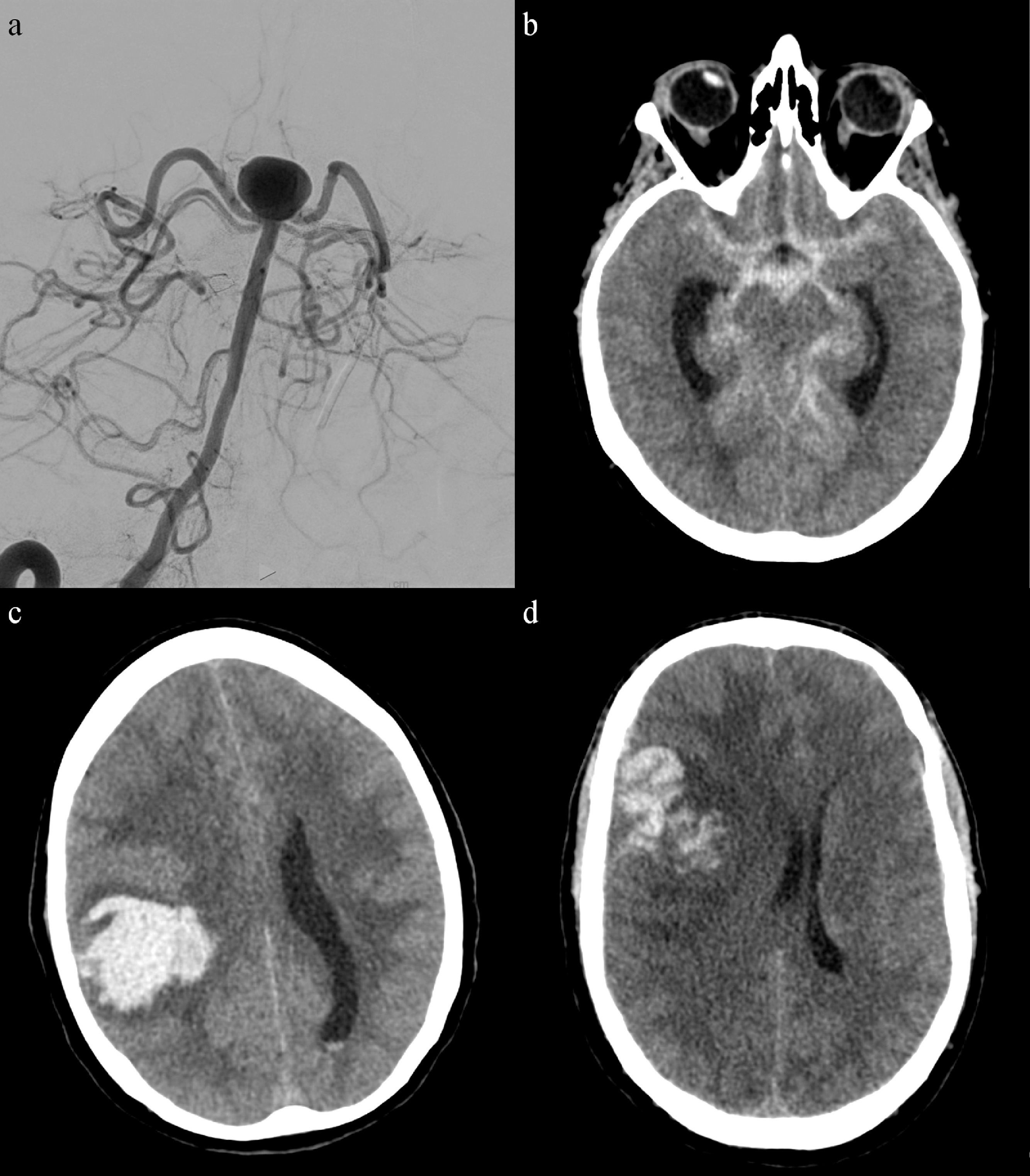
Aneurysm Locations
Approximately 85% of aneurysms arise from the anterior circulation: anterior communicating artery (ACom, most common), posterior communicating artery (PCom, can cause ipsilateral CN III palsy), MCA bifurcation, ICA terminus. Posterior circulation aneurysms (basilar tip, PICA, vertebral) are less common but technically demanding.
Other SAH Causes
Perimesencephalic (nonaneurysmal) SAH — bleeding around the midbrain with a benign course and normal CTA/DSA. Also: AVM rupture, dural AV fistula, mycotic aneurysm, vasculitis, trauma.
Workup
Noncontrast CT head is first-line — sensitivity >95% within 6 hours. If CT is negative but clinical suspicion is high, lumbar puncture for xanthochromia. CTA demonstrates the aneurysm in most cases; DSA (catheter angiography) is the gold standard and also allows treatment.
Grading
SAH severity is described by three scales (fully enumerated in Section 23): Hunt and Hess (clinical grade 1–5), WFNS (based on GCS and focal deficit), and modified Fisher (radiographic, predicts vasospasm risk).
Management
Secure the aneurysm within 24–72 hours by either microsurgical clipping (craniotomy, pterional approach most common) or endovascular coiling (catheter-based, often with stent or flow diverter assistance). Post-securing care focuses on preventing and treating complications: rebleeding, hydrocephalus (EVD), vasospasm and delayed cerebral ischemia (nimodipine 60 mg PO q4h for 21 days, euvolemia, permissive hypertension once aneurysm secured), hyponatremia (cerebral salt wasting vs SIADH), and seizures.
Delayed cerebral ischemia typically emerges days 4–14 after SAH. New focal deficit, altered mental status, or decrease in GCS should trigger repeat imaging and transcranial Doppler or CTA/CT perfusion. Document the TCD mean flow velocities and any new deficits time-stamped precisely.
06 Vascular Malformations Cranial
Arteriovenous Malformation (AVM)
Abnormal tangle (nidus) of vessels in which arteries connect directly to veins without an intervening capillary bed. Presentation: hemorrhage (most common), seizure, headache, or focal deficit. Graded by Spetzler–Martin scale (size, eloquence, venous drainage — fully enumerated in Section 23). Treatment options: microsurgical resection, stereotactic radiosurgery (Gamma Knife, CyberKnife), endovascular embolization (usually adjunctive), or multimodal combination. The ARUBA trial informed the debate on treating unruptured AVMs.
Cavernous Malformation (Cavernoma)
Cluster of abnormal thin-walled vessels without intervening brain parenchyma. "Popcorn" appearance on MRI with blood products of different ages and a hemosiderin ring on GRE/SWI. Often found incidentally; may present with seizure or small hemorrhage. Surgical resection for accessible symptomatic lesions.
Dural Arteriovenous Fistula (dAVF)
Abnormal connection between dural arteries and dural venous sinuses or cortical veins. Risk stratified by the Borden (I–III) and Cognard classifications; lesions with cortical venous drainage are high risk for hemorrhage. Treatment is typically endovascular embolization, occasionally microsurgical disconnection.
Moyamoya Disease
Progressive stenosis of the distal ICAs with compensatory abnormal collateral vessels (the "puff of smoke" on angiography). Presents with ischemic strokes in children and hemorrhagic strokes in adults. Treated with surgical revascularization: STA–MCA bypass (direct), encephaloduroarteriosynangiosis (EDAS), or encephalomyosynangiosis (EMS) (indirect).
07 ICH & Surgical Ischemic Stroke Cranial
Spontaneous Intracerebral Hemorrhage
Most commonly from hypertensive vasculopathy (deep locations: basal ganglia, thalamus, pons, cerebellum) or cerebral amyloid angiopathy (lobar locations in the elderly). Other causes: AVM, tumor, coagulopathy, drug use. Document location, volume (ABC/2 formula: A×B×C/2 in cm), intraventricular extension, and midline shift. Management per 2022 AHA/ASA Guideline for Spontaneous ICH: BP control (target SBP 130–150), reversal of coagulopathy, surgical evacuation selectively. Cerebellar ICH >3 cm or with brainstem compression or hydrocephalus is a surgical emergency — suboccipital craniectomy for evacuation.
Ischemic Stroke — Surgical Considerations
Large hemispheric ("malignant") MCA infarcts can develop cytotoxic edema that peaks at 2–5 days and causes fatal herniation. Decompressive hemicraniectomy within 48 hours in selected patients <60 years reduces mortality (DECIMAL, DESTINY, HAMLET trials). Large cerebellar infarcts may likewise require suboccipital craniectomy. Endovascular thrombectomy is managed by neurointerventionalists, but neurosurgery is often consulted for post-thrombectomy hemorrhagic transformation.
08 Hydrocephalus & Shunt Management Cranial
Hydrocephalus is an abnormal accumulation of CSF in the ventricles. Three broad categories: communicating (CSF flows freely out of the ventricles but is not reabsorbed, e.g. post-SAH), non-communicating (obstructive) (blockage within the ventricular system, e.g. aqueductal stenosis, colloid cyst, posterior fossa mass), and normal pressure hydrocephalus (NPH) (classic triad of gait disturbance, urinary incontinence, and cognitive decline — "wet, wobbly, wacky" — in older adults, often shunt-responsive).
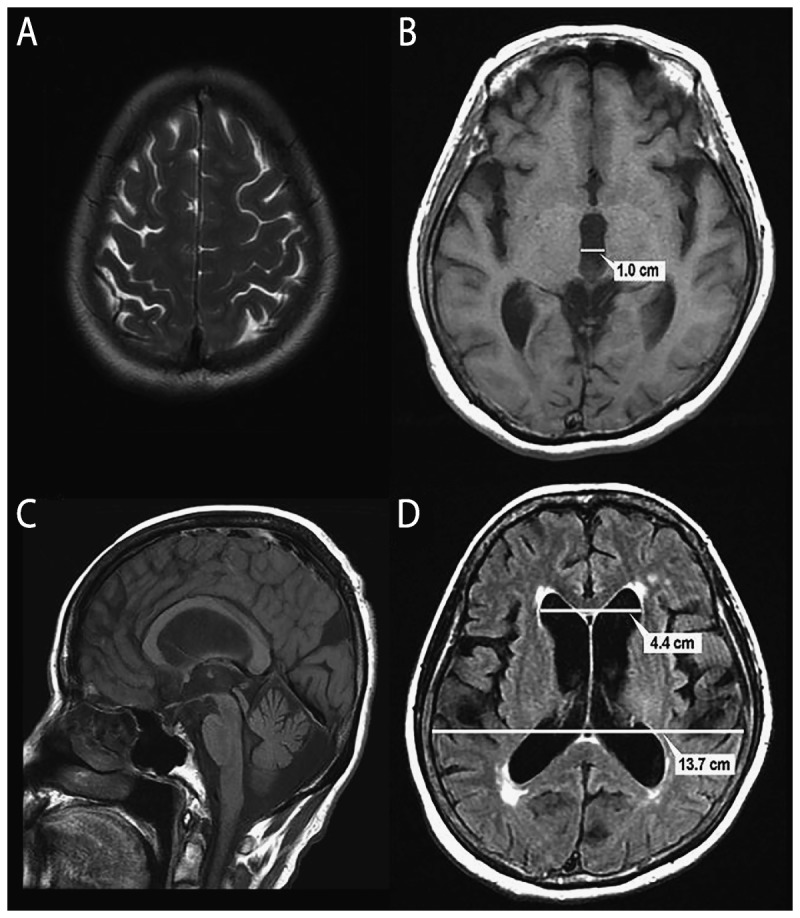
Clinical Features
Acute hydrocephalus causes headache, vomiting, papilledema, upward gaze palsy (Parinaud), and declining level of consciousness. In infants, look for bulging fontanelle, sunset sign, and accelerating head circumference.
Treatment
Emergent CSF diversion with an external ventricular drain (EVD) for acute symptomatic hydrocephalus. Permanent diversion options: ventriculoperitoneal (VP) shunt (most common), ventriculoatrial (VA) shunt, lumboperitoneal shunt, or endoscopic third ventriculostomy (ETV) for obstructive hydrocephalus.
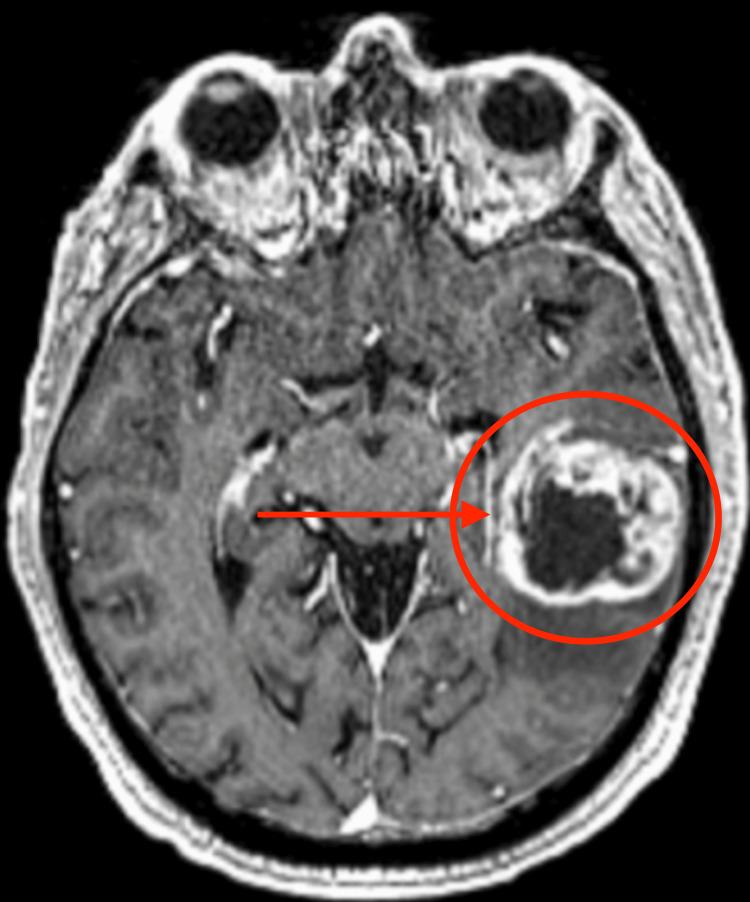
Shunt Complications
Any shunted patient presenting with headache, vomiting, altered mental status, or seizure must be worked up for shunt malfunction. Workup: head CT (ventricle size compared to prior), shunt series X-rays (looking for disconnection or migration), and shunt tap if indicated. Infection risk is highest within 6 months of placement and requires hardware removal plus IV antibiotics plus EVD as a bridge.
09 Brain Tumors Cranial
Brain tumors are classified as primary (arising from brain tissue) or metastatic (from extracranial cancer). The WHO Classification of Tumors of the CNS, 5th edition (2021) and NCCN Guidelines for CNS Cancers are the reference standards.
Gliomas
Tumors arising from glial cells. Graded WHO I–IV by histology and molecular features (IDH mutation, 1p/19q codeletion, MGMT methylation, H3 K27 status, etc.).
- WHO grade 1: Pilocytic astrocytoma — typically pediatric, often cerebellar, curable by gross total resection.
- WHO grade 2: Diffuse low-grade glioma (IDH-mutant astrocytoma or oligodendroglioma) — slow growing but infiltrative; typically progresses.
- WHO grade 3: Anaplastic astrocytoma or anaplastic oligodendroglioma — higher mitotic activity, shorter survival.
- WHO grade 4: Glioblastoma (GBM, IDH-wildtype) — the most aggressive and most common primary malignant brain tumor in adults. Ring-enhancing with central necrosis and peritumoral edema. Median survival ~15 months with maximal safe resection + concurrent temozolomide + radiation (Stupp protocol).
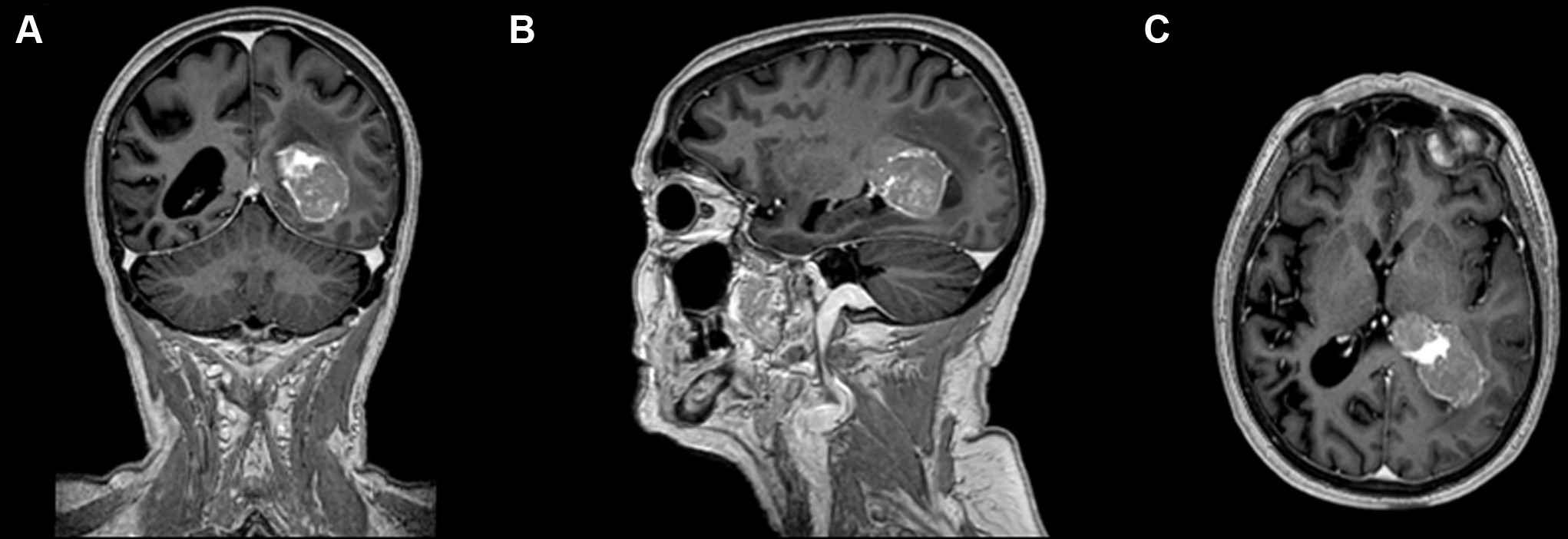
Meningioma
Arises from arachnoid cap cells. Usually dural-based, extra-axial, well-demarcated, homogeneously enhancing, often with a dural tail. WHO grade I (benign, ~80%), grade II (atypical), grade III (anaplastic). Extent of resection is graded by the Simpson grade (see Section 23).
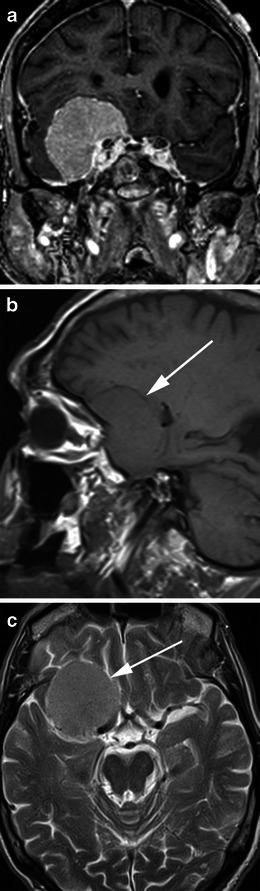
Vestibular Schwannoma (Acoustic Neuroma)
Benign tumor of the vestibular portion of CN VIII in the cerebellopontine angle. Presents with asymmetric sensorineural hearing loss, tinnitus, and imbalance. Options: observation with serial MRI, stereotactic radiosurgery, or microsurgical resection via retrosigmoid, middle fossa, or translabyrinthine approach. Facial nerve function documented with the House–Brackmann grade (see Section 23).
Other Primary Tumors
Craniopharyngioma (sellar/suprasellar, bimodal age distribution, Rathke pouch origin), medulloblastoma (posterior fossa, pediatric), ependymoma (fourth ventricle or spinal cord), primary CNS lymphoma (typically periventricular, enhances avidly, diagnosed by biopsy and treated with high-dose methotrexate rather than resection), pineal region tumors, and intraventricular tumors (colloid cyst, central neurocytoma).
Brain Metastases
More common than primary tumors. Most frequent primaries: lung, breast, melanoma, renal cell, GI. Typically at the gray–white junction with significant surrounding edema. Treatment: stereotactic radiosurgery for limited (1–4 or up to 10) metastases, whole brain radiation for diffuse disease, surgical resection for large symptomatic lesions or when diagnosis is needed.
10 Pituitary & Sellar Lesions Cranial
Pituitary adenomas are classified by size (microadenoma <10 mm, macroadenoma ≥10 mm) and by hormone secretion (prolactinoma, GH-secreting causing acromegaly, ACTH-secreting causing Cushing disease, TSH-secreting, gonadotroph, nonfunctioning). Presentation: visual field loss (bitemporal hemianopia from optic chiasm compression), headache, hormonal syndrome, or pituitary apoplexy (sudden hemorrhage/infarction, surgical emergency).
Management
Prolactinomas are usually treated medically with dopamine agonists (cabergoline, bromocriptine). Other pituitary adenomas with visual compromise or hormone excess typically require surgical resection via the endoscopic endonasal transsphenoidal approach — performed jointly with ENT, through the nose and sphenoid sinus into the sella. Document pre-op visual fields, cortisol level, and thyroid function, and post-op watch for diabetes insipidus, SIADH, CSF leak, and hypopituitarism.
Other Sellar Lesions
Craniopharyngioma, Rathke cleft cyst, meningioma, chordoma, and metastasis can all present in the sellar/parasellar region.
11 Functional & Epilepsy Neurosurgery Cranial
Deep Brain Stimulation (DBS)
Implanted electrodes in deep nuclei connected to a pulse generator. Indications: medically refractory Parkinson disease (STN or GPi), essential tremor (VIM thalamus), dystonia (GPi), and selectively OCD and treatment-resistant depression (anterior limb of internal capsule, subcallosal cingulate). Surgery done awake or asleep with stereotactic frame and microelectrode recording.
Epilepsy Surgery
For medically refractory epilepsy (failed ≥2 appropriate antiseizure drugs). Workup: video EEG, MRI, PET/SPECT, neuropsychological testing, Wada test, invasive monitoring (subdural grids or stereoEEG) to localize the seizure focus. Resective procedures: anterior temporal lobectomy with amygdalohippocampectomy (most common and most effective), lesionectomy, frontal/parietal resection. Disconnective: corpus callosotomy (for drop attacks), hemispherectomy/hemispherotomy (for hemispheric disease in children). Neuromodulation: vagal nerve stimulator (VNS), responsive neurostimulation (RNS), and anterior thalamic DBS.
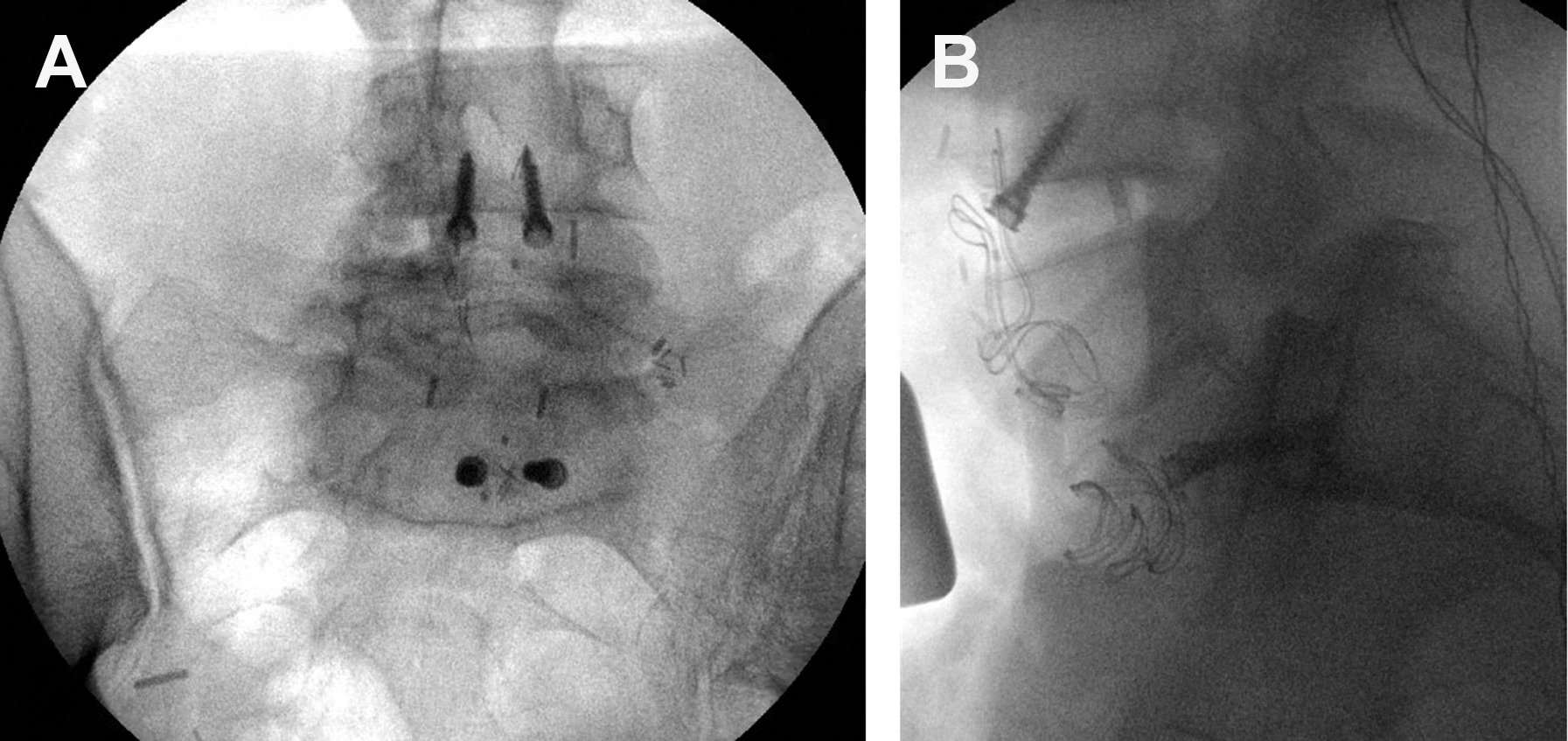
Trigeminal Neuralgia
Severe, lancinating facial pain in a CN V distribution, usually from vascular compression of the trigeminal nerve at the root entry zone. First-line medical therapy is carbamazepine. Surgical options: microvascular decompression (MVD) via retrosigmoid craniotomy (most durable), percutaneous rhizotomy (glycerol, balloon, radiofrequency), or stereotactic radiosurgery.
Hemifacial Spasm
Involuntary unilateral facial twitching from vascular compression of CN VII. Treated by MVD of the facial nerve at the root exit zone, or by botulinum toxin injections.
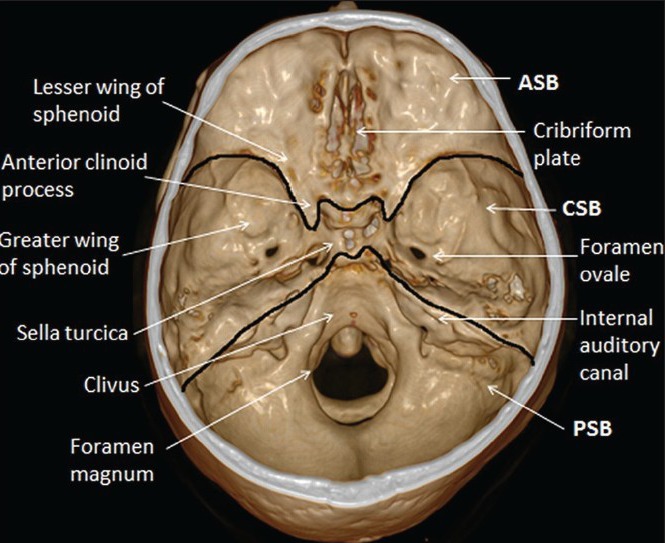
12 Cranial Nerve & Skull Base Disorders Cranial
Skull base surgery is a technically demanding subspecialty targeting lesions at the base of the brain adjacent to critical neurovascular structures. Tumors include meningiomas (sphenoid wing, olfactory groove, petroclival, tuberculum sellae), schwannomas (trigeminal, jugular foramen), chordoma, chondrosarcoma, esthesioneuroblastoma, and paragangliomas. Approaches include pterional, orbitozygomatic, subfrontal, transpetrosal, far lateral, and endoscopic endonasal — the choice driven by lesion location and goals of resection.
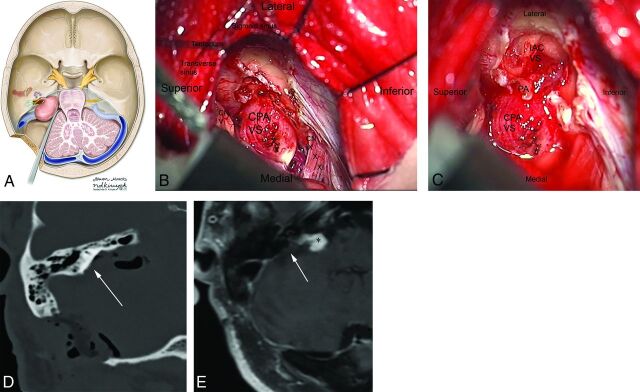
Chiari malformations involve downward displacement of the cerebellar tonsils through the foramen magnum. Type I (tonsils >5 mm below foramen magnum) often presents in young adults with Valsalva-induced occipital headache, neck pain, and possible syringomyelia. Type II is associated with myelomeningocele. Symptomatic cases treated with posterior fossa decompression ± duraplasty.
Syringomyelia is a fluid-filled cavity within the spinal cord, often associated with Chiari I. Presents with a cape-like distribution of pain and temperature loss (dissociated sensory loss) and progressive weakness. Treatment targets the underlying cause (Chiari decompression, tumor resection, adhesion lysis).
13 CNS Infections Cranial
Brain abscess: Focal collection of pus within the brain parenchyma. Routes: hematogenous (endocarditis, pulmonary), contiguous (sinusitis, otitis, dental), direct inoculation (trauma, surgery). MRI with contrast shows a ring-enhancing lesion with restricted diffusion centrally (a useful distinction from necrotic tumor, which does not restrict). Management: stereotactic aspiration for diagnosis and source control, prolonged IV antibiotics (often 6–8 weeks), treatment of the source, and occasional craniotomy for excision.
Subdural empyema: Pus in the subdural space, typically from sinusitis or mastoiditis. A neurosurgical emergency — progresses to seizures, focal deficits, and rapid decline. Treatment is urgent surgical drainage (craniotomy or burr hole) plus IV antibiotics.
Spinal epidural abscess: Pus in the spinal epidural space, classically from S. aureus. Presents with back pain, fever, and progressive neurologic deficit. Emergent MRI, IV antibiotics, and usually decompressive surgical drainage. Any delay in diagnosis allows irreversible cord injury.
14 Degenerative Cervical Spine Spine
Cervical disc herniation: Nucleus pulposus herniates laterally or centrally, compressing a nerve root or the cord. Lateral herniation causes radiculopathy in a dermatomal pattern (C5: shoulder/deltoid weakness; C6: biceps, radial forearm, thumb; C7: triceps, middle finger; C8: intrinsic hand, small finger). Central herniation can cause myelopathy.
Cervical spondylotic myelopathy (CSM): Progressive cord compression from age-related disc and facet degeneration, osteophytes, and ligamentum flavum hypertrophy. Signs: gait instability, hand clumsiness, loss of fine motor control, Hoffman sign, hyperreflexia, Babinski, clonus, sensory changes. Graded by the modified Japanese Orthopaedic Association (mJOA) score. Surgical decompression indicated for moderate/severe myelopathy.
OPLL (ossification of the posterior longitudinal ligament): Progressive ossification of the PLL causing cord compression, especially in Asian populations. Treated with anterior decompression/fusion or posterior laminoplasty/laminectomy depending on extent and cervical alignment.
Surgical options: ACDF (anterior cervical discectomy and fusion), cervical disc arthroplasty, posterior cervical laminectomy and fusion, or laminoplasty. AO Spine CSM guidelines offer an evidence-based framework.
15 Degenerative Thoracolumbar Spine Spine
Lumbar disc herniation: The classic cause of sciatica. A paracentral L4–L5 herniation typically affects the L5 root (foot drop, dorsiflexion weakness, lateral leg and dorsum of foot sensory loss), and an L5–S1 herniation affects the S1 root (plantarflexion weakness, loss of ankle reflex, lateral foot sensation). Positive straight leg raise correlates with nerve root tension. Most improve with conservative care; surgery (microdiscectomy) for refractory or progressive deficit.
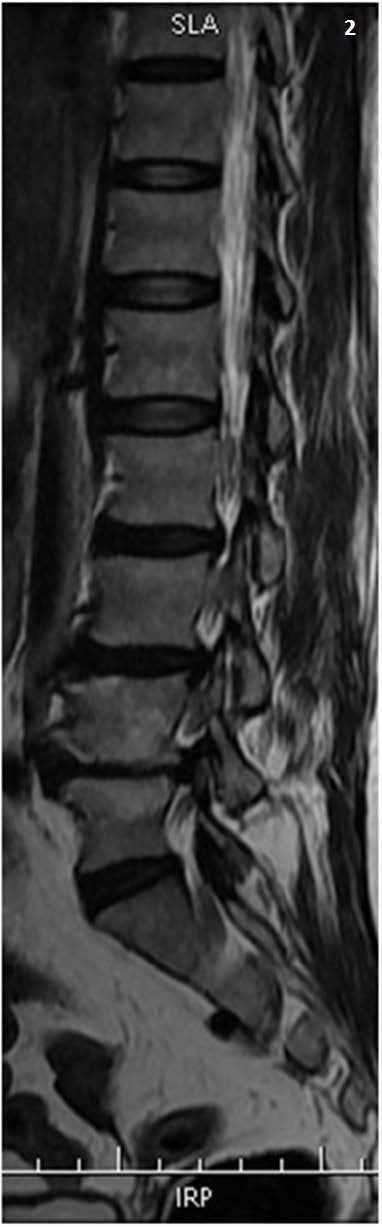
Lumbar spinal stenosis: Narrowing of the central canal or neural foramina from ligamentum hypertrophy, facet arthropathy, and disc bulging. Classic presentation: neurogenic claudication — bilateral leg pain/heaviness with walking and prolonged standing, relieved by sitting or leaning forward (e.g., on a shopping cart). Contrast this with vascular claudication, which is relieved by rest alone. Surgical treatment: lumbar laminectomy ± fusion.
Spondylolisthesis: Forward slippage of one vertebral body on another. Degenerative (most common in older adults at L4–L5, from facet arthritis) or isthmic (from pars interarticularis defect, classically at L5–S1, often in younger patients). Graded by the Meyerding system (I–V based on percentage of slip). Surgery: decompression ± fusion when unstable or symptomatic.
Vertebral compression fracture: Osteoporotic fractures common in elderly patients. Painful kyphotic deformity. Treat with analgesia, bracing, osteoporosis management, and for persistent pain kyphoplasty or vertebroplasty (percutaneous cement augmentation). NASS Clinical Guidelines for Lumbar Stenosis and Disc Herniation frame nonoperative and surgical decision-making.
Lumbar fusion techniques: PLIF (posterior lumbar interbody fusion), TLIF (transforaminal), ALIF (anterior), XLIF/LLIF (extreme/lateral), with pedicle screw–rod constructs providing posterior stabilization.
16 Spinal Deformity Spine
Scoliosis: Three-dimensional deformity of the spine with lateral curvature (Cobb angle >10°) and rotation. Adolescent idiopathic scoliosis is most common in pediatrics. Surgery (posterior spinal fusion with pedicle screw instrumentation) is indicated for curves >40–50° in skeletally immature patients or for progressive adult deformity with pain and neurologic deficit. Adult degenerative scoliosis is managed jointly with orthopedic spine, often with long-construct fusions.
Kyphosis: Abnormal forward curvature. Scheuermann kyphosis (adolescent), post-traumatic kyphosis, and iatrogenic flat-back are common indications for deformity correction with osteotomies and instrumented fusion. Sagittal alignment parameters (pelvic incidence, pelvic tilt, sacral slope, lumbar lordosis, sagittal vertical axis) are documented meticulously in deformity cases.
17 Spinal Trauma & SCI Spine
Spinal trauma is classified by level (cervical, thoracic, thoracolumbar, lumbar) and by fracture pattern. The AO Spine classification categorizes injuries as A (compression), B (distraction), or C (translation), with modifiers for neurologic status. Unstable injuries require stabilization.
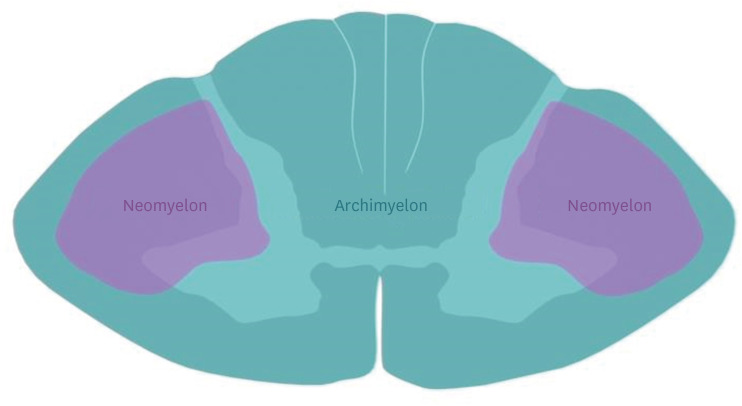
Spinal Cord Injury (SCI)
Complete (no motor or sensory function below the level, including S4–S5) versus incomplete (some preservation). The ASIA Impairment Scale (A–E) is the standard functional grade (fully enumerated in Section 23). Classic incomplete syndromes: central cord (upper > lower extremity weakness, classic in elderly with cervical spondylosis and hyperextension injury), Brown-Séquard (hemicord injury: ipsilateral motor and proprioception loss, contralateral pain/temperature loss), anterior cord (motor and pain/temperature loss, preserved dorsal columns), and posterior cord (proprioception loss, rare).
Cauda Equina & Conus Medullaris Syndromes
Cauda equina syndrome (CES): compression of the lumbosacral nerve roots, classically from a massive central disc herniation. Features: saddle anesthesia, urinary retention with overflow incontinence, fecal incontinence, bilateral lower extremity weakness, loss of anal tone. A surgical emergency — decompression within 24–48 hours. Conus medullaris syndrome presents with mixed UMN/LMN features from injury at the tip of the cord (T12–L1).
Acute Management
Immobilization, ATLS survey, MAP goal 85–90 mmHg for 5–7 days (avoid secondary injury), and urgent decompression/stabilization when indicated. High-dose methylscorticosteroids are not routinely recommended per current AANS/CNS guidelines.
18 Spinal Tumors & Vascular Lesions Spine
Spinal tumors are classified by compartment: extradural (~60%, mostly metastases from lung, breast, prostate, renal, and myeloma), intradural-extramedullary (meningioma, schwannoma, neurofibroma), and intramedullary (ependymoma, astrocytoma, hemangioblastoma). Presentation varies from progressive back pain and radiculopathy to frank myelopathy or CES. Metastatic epidural spinal cord compression is evaluated with the NOMS framework (Neurologic, Oncologic, Mechanical, Systemic) to integrate radiation, surgery, and systemic therapy. Separation surgery followed by SBRT is a modern paradigm for radioresistant tumors.
Spinal AVM/dAVF: Progressive myelopathy (Foix–Alajouanine syndrome) from venous congestion of a dural AV fistula. Treated with endovascular embolization or microsurgical disconnection.
19 Peripheral Nerve Surgery Peripheral
Carpal tunnel syndrome: Median nerve compression at the wrist. Numbness and tingling in the thumb, index, middle, and radial half of the ring finger; nocturnal paresthesias; thenar atrophy in severe cases. Positive Tinel and Phalen tests. Confirmed by EMG/NCS. Treatment: splinting, steroid injection, then carpal tunnel release (open or endoscopic).
Ulnar neuropathy at the elbow (cubital tunnel): Tingling in the small and ring fingers, intrinsic hand weakness, Froment sign. Treated with activity modification, night splinting, and cubital tunnel release with or without anterior transposition.
Peroneal palsy: Common peroneal compression at the fibular head causes foot drop. Treat the cause; decompression if persistent.
Brachial plexus injury: Traction injuries (motorcycle, birth trauma) cause varied deficits from upper (Erb) to lower (Klumpke) trunk patterns. Management ranges from observation to nerve grafting and nerve transfers (e.g., spinal accessory to suprascapular, Oberlin transfer).
Nerve sheath tumors: Schwannoma, neurofibroma, malignant peripheral nerve sheath tumor (MPNST). Excision for symptomatic or enlarging lesions; MPNST requires oncologic resection.
20 Neurosurgical Procedures — A to Z Procedures
| Procedure | What it is | Typical indication |
|---|---|---|
| Craniotomy | Removal and replacement of a bone flap to access the brain | Tumor, aneurysm, SDH/EDH, AVM, abscess |
| Decompressive craniectomy | Craniotomy in which the bone flap is not replaced | Refractory ICP, malignant MCA infarct |
| Burr hole | Single drill hole through skull | Chronic SDH, ICP monitor, EVD, biopsy |
| Twist drill | Small bedside drill hole at bedside | Emergent ICP bolt, bedside EVD |
| EVD placement | Catheter into the lateral ventricle via Kocher point | Hydrocephalus, ICP monitoring with drainage |
| ICP monitor | Parenchymal (Camino, Codman) or subdural bolt | Severe TBI with inability to examine |
| VP shunt | Ventricular catheter → valve → peritoneum | Chronic hydrocephalus, NPH |
| VA shunt | Ventricular catheter → valve → right atrium | When abdomen is not suitable |
| Lumboperitoneal shunt | Lumbar subarachnoid → peritoneum | Communicating hydrocephalus, pseudotumor cerebri |
| ETV | Endoscopic third ventriculostomy through floor of third ventricle | Obstructive hydrocephalus (e.g., aqueductal stenosis) |
| Aneurysm clipping | Metal clip across aneurysm neck via craniotomy | Ruptured or unruptured aneurysm |
| Endovascular coiling / flow diversion | Catheter-based occlusion (neuroIR) | Aneurysm, primarily posterior circulation and wide-neck |
| AVM resection | Microsurgical excision of nidus | Spetzler–Martin I–III AVM |
| Awake craniotomy | Craniotomy with intraoperative cortical mapping while patient awake | Tumor in eloquent cortex (Broca/Wernicke/motor) |
| Transsphenoidal surgery | Endoscopic endonasal approach to sella | Pituitary adenoma, craniopharyngioma, skull base |
| MVD | Microvascular decompression at root entry zone | Trigeminal neuralgia, hemifacial spasm |
| Stereotactic radiosurgery | Gamma Knife or CyberKnife focused radiation | AVM, schwannoma, brain metastases, TN |
| DBS | Stereotactic implantation of deep electrodes + IPG | Parkinson, essential tremor, dystonia, OCD |
| VNS / RNS | Neuromodulation implants for epilepsy | Refractory epilepsy |
| ACDF | Anterior cervical discectomy and fusion | Cervical disc/myelopathy |
| Posterior cervical fusion / laminoplasty | Posterior decompression ± instrumented fusion | Multilevel CSM, OPLL |
| Lumbar microdiscectomy | Minimally invasive nerve root decompression | Lumbar disc herniation with radiculopathy |
| Lumbar laminectomy | Removal of lamina to decompress central canal | Lumbar spinal stenosis |
| Lumbar fusion (PLIF/TLIF/ALIF/XLIF) | Interbody + pedicle screw construct | Instability, spondylolisthesis, deformity |
| Kyphoplasty / vertebroplasty | Percutaneous cement augmentation of vertebral body | Osteoporotic or pathologic compression fracture |
| Scoliosis correction | Long-construct instrumented fusion, osteotomies | Adolescent or adult deformity |
| Carpal/cubital tunnel release | Decompression of compressed nerve | Median/ulnar entrapment |
| Nerve graft / nerve transfer | Bridging a nerve gap with donor graft or functional transfer | Brachial plexus injury, peripheral nerve reconstruction |
| Intrathecal baclofen pump | Implanted catheter into lumbar thecal sac with SC pump | Severe spasticity (CP, MS, SCI) |
Procedure names in neurosurgery are long but consistent. Always document approach + side + level + indication. "Right pterional craniotomy for clipping of ruptured right MCA bifurcation aneurysm" is the bare minimum detail — anything less is incomplete.
21 Neuroimaging & Diagnostics Diagnostics
Noncontrast CT head: First-line for acute trauma and suspected intracranial hemorrhage. Acute blood is hyperdense (~60–80 HU). Document midline shift in mm, ventricular size, cisternal effacement, and any mass effect.
CT angiography (CTA): Demonstrates vascular anatomy, aneurysms, vasospasm, dissection, and occlusion in stroke. A CTA head + neck is routine in new stroke or SAH.
MRI brain: The workhorse for tumor, infection, demyelination, and subacute/chronic pathology. Sequences and what they show: T1 (anatomy, fat bright), T1+contrast (enhancement = BBB breakdown, tumor, infection), T2 (fluid bright, edema), FLAIR (fluid-attenuated, shows white matter lesions), DWI/ADC (restricted diffusion = acute stroke, abscess pus, hypercellular tumor), SWI/GRE (blood products, cavernomas, microbleeds), MRS (metabolite peaks for tumor differentiation).
MRI spine: Standard for disc disease, stenosis, cord pathology, tumor, infection. T2 sagittal shows the canal at a glance.
Digital subtraction angiography (DSA): Gold standard for cerebrovascular imaging. Performed by neurosurgery or neurointerventional radiology.
EEG, TCD, SSEP/MEP: EEG for seizure detection; TCD for vasospasm surveillance after SAH; intraoperative SSEP/MEP for spinal cord and cortical monitoring during complex cases.
22 Medications You Must Know Meds
| Drug | Class / Use | Key notes |
|---|---|---|
| Mannitol | Osmotic diuretic for elevated ICP | 0.25–1 g/kg IV; watch serum osm (<320), Na, renal function |
| Hypertonic saline 3% / 23.4% | Osmotherapy for ICP / edema | 23.4% is a bolus via central line; 3% by infusion. Target Na 145–155. |
| Nimodipine | CCB for SAH vasospasm | 60 mg PO q4h × 21 days after aneurysmal SAH |
| Levetiracetam (Keppra) | AED / seizure prophylaxis | Standard for post-craniotomy, TBI with hemorrhage, tumor |
| Dexamethasone (Decadron) | Steroid for vasogenic edema | Tumor, abscess. Not for TBI (CRASH trial showed harm). |
| Cefazolin / vancomycin | Surgical antibiotic prophylaxis | Given within 60 min of incision, redose for long cases |
| Heparin / LMWH (enoxaparin) | DVT prophylaxis | Timing after intracranial bleed is a critical neurosurgical decision |
| Fentanyl / propofol | ICU sedation/analgesia | Titrated for ventilator synchrony and ICP control; allow neuro checks |
| Midazolam (Versed) | Benzodiazepine sedation | Shorter-acting alternative for neuro ICU |
| Dexmedetomidine (Precedex) | Alpha-2 sedation | Preserves neuro exam, useful for awake craniotomy weaning |
| Phenytoin (Dilantin) | AED (older alternative to Keppra) | Check free level; many interactions |
| Intrathecal baclofen | Spasticity | Delivered via implanted pump; overdose/withdrawal are emergencies |
| Carbamazepine | AED; first-line trigeminal neuralgia | Monitor CBC, Na, LFTs |
| Dopamine agonists (cabergoline) | Prolactinoma | First-line; often avoids surgery |
| DDAVP (desmopressin) | Central DI after pituitary surgery | Titrate by urine output and Na |
| Hydrocortisone | Peri-op steroid coverage for pituitary/adrenal insufficiency | Stress dose for transsphenoidal |
| TXA (tranexamic acid) | Antifibrinolytic | CRASH-3 supports early use in TBI; surgeon preference dependent |
23 Classification Systems Reference
| Component | Score | Criteria |
|---|---|---|
| Eye (E, 1–4) | 4 | Spontaneous |
| 3 | To voice | |
| 2 | To pain | |
| 1 | None | |
| Verbal (V, 1–5) | 5 | Oriented |
| 4 | Confused conversation | |
| 3 | Inappropriate words | |
| 2 | Incomprehensible sounds | |
| 1 | None | |
| Motor (M, 1–6) | 6 | Obeys commands |
| 5 | Localizes pain | |
| 4 | Withdraws from pain | |
| 3 | Decorticate (flexor) posturing | |
| 2 | Decerebrate (extensor) posturing | |
| 1 | None |
| Grade | Clinical |
|---|---|
| I | Asymptomatic or mild headache and slight nuchal rigidity |
| II | Moderate to severe headache, nuchal rigidity, no deficit except cranial nerve palsy |
| III | Drowsiness, confusion, or mild focal deficit |
| IV | Stupor, moderate to severe hemiparesis, early decerebrate rigidity |
| V | Deep coma, decerebrate rigidity, moribund |
| Grade | GCS | Motor deficit |
|---|---|---|
| I | 15 | Absent |
| II | 13–14 | Absent |
| III | 13–14 | Present |
| IV | 7–12 | Present or absent |
| V | 3–6 | Present or absent |
| Grade | CT Findings |
|---|---|
| 0 | No SAH or IVH |
| 1 | Thin SAH, no IVH |
| 2 | Thin SAH with IVH |
| 3 | Thick SAH, no IVH |
| 4 | Thick SAH with IVH |
| Grade | Clinical |
|---|---|
| 0 | Neurologically normal |
| 1 | Alert, mild symptoms (headache, reflex asymmetry) |
| 2 | Drowsy or disoriented with variable neurologic deficit |
| 3 | Stuporous, severe focal deficit |
| 4 | Comatose, decerebrate or decorticate posturing |
| Feature | Points |
|---|---|
| Size <3 cm / 3–6 cm / >6 cm | 1 / 2 / 3 |
| Eloquent brain adjacent | 0 = no, 1 = yes |
| Deep venous drainage | 0 = no, 1 = yes |
Total 1–5. Grades 1–2 favor surgery; grade 3 is borderline; grades 4–5 usually managed conservatively or with multimodal therapy.
| Grade | Extent |
|---|---|
| I | Gross total resection + excision of dural attachment and abnormal bone |
| II | Gross total resection + coagulation of dural attachment |
| III | Gross total resection without treatment of dural attachment |
| IV | Subtotal resection |
| V | Biopsy / decompression only |
| Grade | Description |
|---|---|
| A | Complete — no motor or sensory function in S4–S5 |
| B | Sensory incomplete — sensation below level including S4–S5, no motor |
| C | Motor incomplete — more than half of key muscles below level with strength <3/5 |
| D | Motor incomplete — at least half of key muscles below level with strength ≥3/5 |
| E | Normal motor and sensory function |
| Grade | Description |
|---|---|
| A | Complete motor and sensory loss |
| B | Sensory only preserved |
| C | Motor preserved but nonfunctional |
| D | Motor preserved and functional |
| E | Normal |
| Type | Pattern |
|---|---|
| A | Compression injury (A0 minor, A1 wedge, A2 split/pincer, A3 incomplete burst, A4 complete burst) |
| B | Tension band / distraction injury (B1 bony Chance, B2 posterior tension band disruption, B3 anterior tension) |
| C | Translation / dislocation (all three columns) |
Modifiers: N0–N4 neurologic status; M1 (indeterminate ligamentous injury) and M2 (patient-specific modifiers).
| Grade | Function |
|---|---|
| I | Normal |
| II | Mild dysfunction — slight weakness on close inspection |
| III | Moderate dysfunction — obvious weakness, complete eye closure with effort |
| IV | Moderately severe — obvious weakness, incomplete eye closure |
| V | Severe dysfunction — barely perceptible motion |
| VI | Total paralysis |
| Grade | % Slip |
|---|---|
| I | 0–25% |
| II | 26–50% |
| III | 51–75% |
| IV | 76–100% |
| V (spondyloptosis) | >100% |
100 normal, no complaints. 90 normal activity with minor symptoms. 80 normal activity with effort. 70 unable to work but self-care. 60 requires occasional assistance. 50 requires considerable assistance. 40 disabled, requires special care. 30 severely disabled, hospitalization indicated. 20 very sick, active supportive treatment. 10 moribund. 0 dead.
0 no symptoms. 1 no significant disability, able to carry out all usual activities. 2 slight disability, unable to do all previous activities but independent in ADLs. 3 moderate disability, requires some help but walks unassisted. 4 moderately severe, unable to walk without assistance. 5 severe disability, bedridden, requires constant care. 6 dead.
24 Physical & Neurologic Exam Exam
The neurologic exam is the core physical exam of neurosurgery and must be documented at every encounter. It is organized by system and graded with objective scales.
MS: A&Ox3, fluent speech, follows commands
CN: II PERRL 3→2 mm, visual fields full; III/IV/VI EOMI; V face sensation intact; VII face symmetric; VIII hearing intact; IX/X palate elevates; XI SCM/trap 5/5; XII tongue midline
Motor: deltoid 5/5, biceps 5/5, triceps 5/5, wrist ext 5/5, grip 5/5; IP 5/5, quad 5/5, TA 5/5, EHL 5/5, gastroc 5/5 — bilateral
Sensory: intact to LT/PP in all dermatomes
Reflexes: 2+ biceps/triceps/brachioradialis/patellar/Achilles; toes downgoing; Hoffman neg; clonus neg
Coordination: FTN, HKS intact; no dysmetria
Gait: narrow-based, steady, tandem intact
Motor grading (MRC 0–5): 0 no contraction; 1 flicker of contraction; 2 movement with gravity eliminated; 3 movement against gravity; 4 movement against some resistance; 5 normal strength. Intermediate grades (4−, 4, 4+) are commonly used.
Reflex grading (0–4+): 0 absent; 1+ diminished; 2+ normal; 3+ brisk; 4+ clonus. Hyperreflexia, Hoffman, Babinski, and clonus suggest an upper motor neuron lesion — critical in suspected myelopathy.
Dermatomes to know cold: C5 lateral arm/deltoid; C6 thumb; C7 middle finger; C8 small finger; T4 nipple; T10 umbilicus; L1 inguinal; L2 anterior thigh; L3 knee; L4 medial leg; L5 dorsum foot/great toe; S1 lateral foot/small toe; S2–S4 perianal.
25 Abbreviations Master List Reference
Anatomy & Diagnoses
Procedures & Devices
Exam & Scales
26 Sample HPI Templates Templates
"Mr. [Name] is a 78-year-old male on apixaban for atrial fibrillation who presents after an unwitnessed fall at home. He was found confused by his wife approximately 2 hours ago. EMS documented GCS 13 (E3 V4 M6) in the field, improving to 14 on arrival. He reports a dull bifrontal headache 6/10 and mild nausea. He denies vomiting, seizure, weakness, or sensory change. Last apixaban dose this morning. Noncontrast CT head demonstrates a 12 mm right-sided acute-on-chronic subdural hematoma with 4 mm leftward midline shift and effacement of the right lateral ventricle. Neurosurgery consulted for evaluation and apixaban reversal."
"Mrs. [Name] is a 62-year-old right-handed female with no significant past medical history who presents for evaluation of a newly diagnosed right frontal mass. Over the last 6 weeks she has had progressive left-sided weakness and two episodes of focal left arm twitching concerning for focal seizures. MRI brain with and without contrast demonstrates a 4.2 × 3.8 cm heterogeneously enhancing right frontal mass with central necrosis and significant surrounding FLAIR hyperintensity, with 5 mm leftward midline shift — concerning for high-grade glioma. She is currently on dexamethasone 4 mg q6h and levetiracetam 1000 mg BID. She is here to discuss surgical options, including awake craniotomy with cortical mapping given proximity to motor cortex."
"Ms. [Name] is a 54-year-old female with hypertension and 20-pack-year smoking history who presents with sudden-onset "worst headache of life" that began abruptly 4 hours ago while gardening. She reports photophobia, nausea, and one episode of emesis. Denies focal weakness or seizure. On exam, GCS 14 (E4 V4 M6) with meningismus but no focal deficit (Hunt–Hess grade 2, WFNS grade 2). Noncontrast CT head demonstrates diffuse basal cistern SAH with bifrontal interhemispheric extension and small early hydrocephalus (modified Fisher 3). CTA demonstrates a 6 mm ACom aneurysm with broad neck. Admitted to neuro ICU; nimodipine started; plan for aneurysm securing within 24 hours."
"Mr. [Name] is a 41-year-old male presenting with 8 weeks of progressive right-sided low back and leg pain. The pain radiates from the right buttock down the posterolateral thigh and calf to the lateral foot and small toe, consistent with an S1 radicular distribution. Pain is 8/10 at worst, worsened by sitting and Valsalva. He has associated numbness in the lateral foot and subjective right calf weakness. Denies bowel/bladder dysfunction or saddle anesthesia. Has failed 6 weeks of PT, NSAIDs, and one epidural steroid injection with transient relief. Exam: positive right straight leg raise at 40°, right plantarflexion 4/5, diminished right Achilles reflex. MRI lumbar spine shows a right paracentral L5–S1 disc extrusion compressing the traversing right S1 nerve root. Presenting to discuss right L5–S1 microdiscectomy."
"Mr. [Name] is a 34-year-old male with a history of congenital aqueductal stenosis s/p right VP shunt placement in childhood who presents with 3 days of progressive headache, nausea, and vomiting. The headache is constant, worse when lying flat, and associated with blurred vision. Denies fever, neck stiffness, or focal weakness. On exam, he is drowsy but arousable, GCS 14, PERRL, with mild upward gaze limitation. CT head shows moderate ventriculomegaly compared to baseline imaging 1 year ago. Shunt series shows the distal catheter intact. Shunt tap yields opening pressure of 28 cm H2O without growth on Gram stain. Concern for proximal shunt obstruction. Plan: emergent shunt revision."
"Mr. [Name] is a 67-year-old male with a history of hypertension and hyperlipidemia presenting with progressive gait instability and hand clumsiness over the past 8 months. He reports difficulty buttoning his shirt and handling small objects. He has fallen twice in the last month. Denies frank weakness, sensory loss, or bowel/bladder dysfunction. Exam: wide-based gait, positive Hoffman bilaterally, hyperreflexia at the biceps and patellar reflexes, sustained ankle clonus on the right, positive Babinski bilaterally, intrinsic hand wasting. mJOA 12/18. MRI cervical spine demonstrates multilevel cervical spondylosis worst at C5–C6 and C6–C7 with cord compression and T2 cord signal change at C5–C6. Presenting to discuss C5–C7 ACDF versus posterior decompression and fusion."
The best neurosurgery scribes are operationally invisible. The attending never has to slow down to correct laterality, spell a procedure name, or re-dictate an exam. They know that "right MCA M1 occlusion" is different from "right ICA terminus occlusion" and chart accordingly. They understand that a GCS drop from 14 to 11 is an emergency and flag it rather than bury it in the note. They pre-pull imaging reports before the patient is seen. They know a House–Brackmann grade without looking it up.
Be obsessive about laterality, time stamps, and exam detail. Learn the surgeon's preferred phrasing. Read the MRI report before the clinic visit so you can pre-populate the note. Own the classification scales. Understand the why behind each plan, not just the what. A scribe who understands pathophysiology writes notes that save the next team time, protect the patient, and make the attending trust them.
Welcome to neurosurgery. Few specialties ask more precision from a scribe — and few specialties reward it more.
27 References & Sources
Clinical Practice Guidelines
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: The Whole Brain. OpenStax College. CC BY 3.0.
Figure 2: Cerebrum. OpenStax College. CC BY 3.0.
Figure 3: Brain Lobes. Blausen Medical. CC BY 3.0.
Figure 4: Brain Stem. OpenStax College. CC BY 3.0.
Figure 5: Brain and Spinal Cord. OpenStax College. CC BY 3.0.
Figure 6: Lumbar Spine. Blausen Medical. CC BY 3.0.
Figure 7: Subdural Hematoma with Herniation. James Heilman MD. CC BY-SA 3.0.