Ophthalmology
Every diagnosis, condition, procedure, medication, abbreviation, and documentation framework you need to succeed on day one in an eye clinic or OR.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Ocular Anatomy Essentials
Ophthalmology is an optical system grafted onto a nervous system. Light travels through a precisely ordered series of structures — tear film, cornea, aqueous, pupil, lens, vitreous, retina — and is transduced into neural signals that exit the eye via the optic nerve. Every diagnosis in the specialty is ultimately a breakdown somewhere along that pathway, and every abbreviation the clinician uses refers to a specific anatomical structure. Know this section cold.
The Globe and Its Layers
The eyeball has three concentric coats. The outer fibrous tunic is composed of the transparent cornea anteriorly and the opaque white sclera posteriorly; they meet at the limbus. The middle uveal tract is a pigmented, vascular layer with three regions: the iris (the colored diaphragm that controls pupil size), the ciliary body (which produces aqueous humor and contains the muscles of accommodation), and the choroid (the highly vascular posterior layer between sclera and retina). The innermost neural tunic is the retina, whose photoreceptors (rods and cones) convert light into electrical signals.
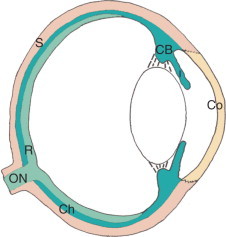
Anterior Segment
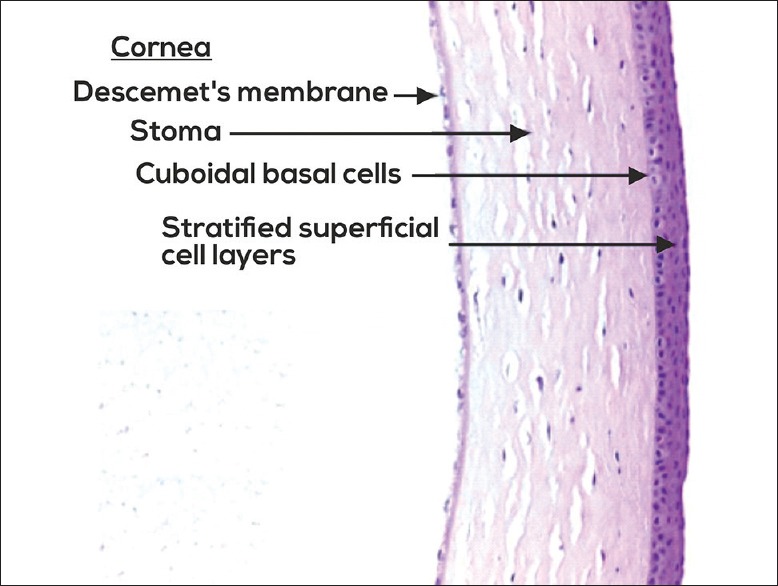
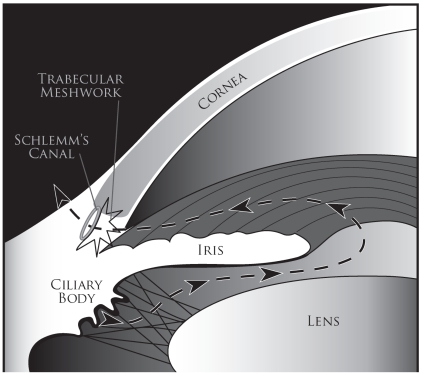
The anterior segment includes everything in front of the vitreous: cornea, anterior chamber, iris, posterior chamber, ciliary body, and lens. The cornea is a five-layer avascular window (epithelium, Bowman layer, stroma, Descemet membrane, endothelium) responsible for about two-thirds of the eye's refractive power. The anterior chamber (AC) is the fluid-filled space between the cornea and the iris; it is filled with aqueous humor produced by the ciliary body, which flows through the pupil into the anterior chamber and drains out through the trabecular meshwork at the anterior chamber angle into Schlemm's canal. Any disruption of this inflow/outflow balance raises intraocular pressure (IOP) and causes glaucoma. The crystalline lens sits behind the iris, suspended by zonular fibers from the ciliary body, and changes shape during accommodation to focus on near objects.
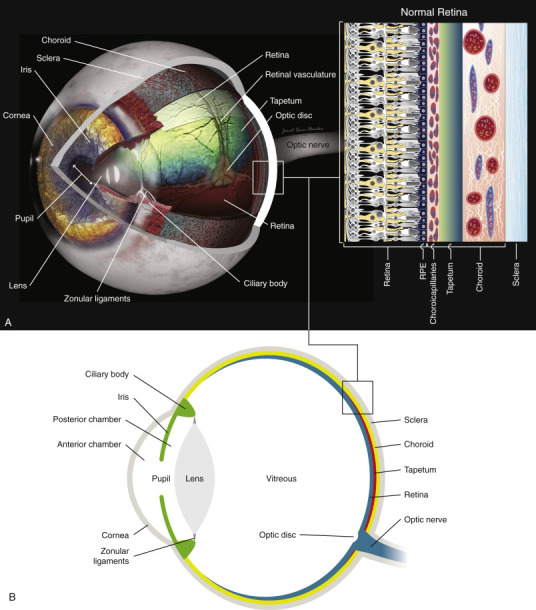
Posterior Segment
Behind the lens lies the vitreous cavity filled with clear gel-like vitreous humor. The retina lines the posterior two-thirds of the globe. Key retinal landmarks: the optic disc (ONH, optic nerve head) where retinal ganglion cell axons exit the eye, the macula (the central specialized retina responsible for high-acuity vision), and within the macula the fovea (a cone-rich pit responsible for 20/20 vision) with the foveal center called the foveola. The retina has a dual blood supply: the inner layers from the central retinal artery (a branch of the ophthalmic artery), and the outer layers (including photoreceptors) from the choriocapillaris. Beneath the retina is the retinal pigment epithelium (RPE), a single-cell layer crucial for photoreceptor support and sitting on Bruch's membrane.
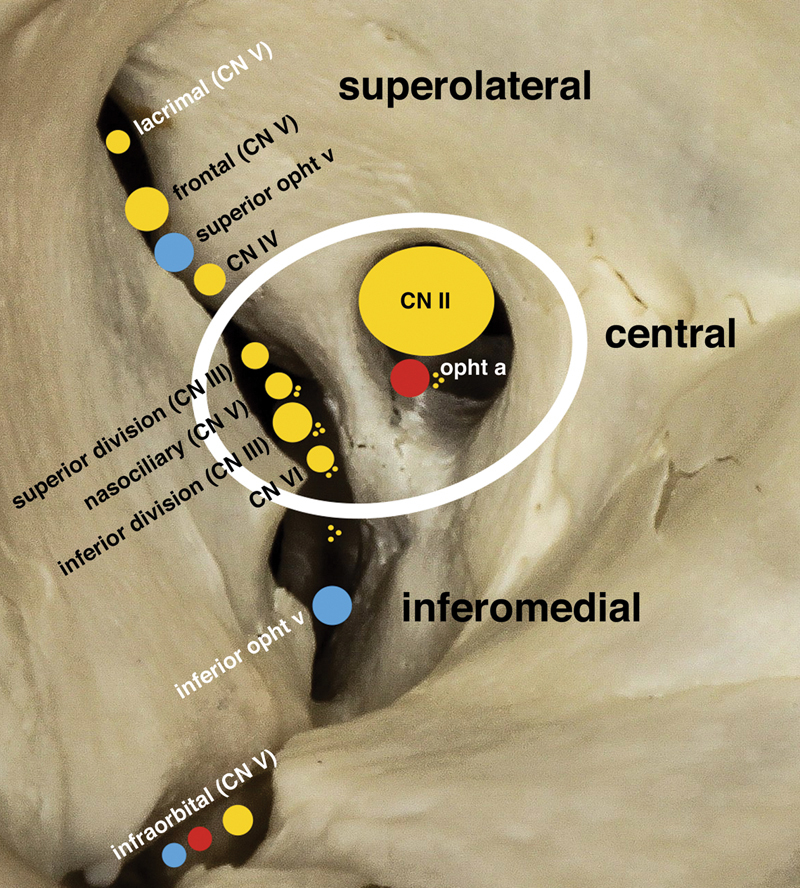
Extraocular Muscles and Orbit
Six extraocular muscles (EOMs) move each eye: four recti (superior, inferior, medial, lateral) and two obliques (superior, inferior). Innervation follows the mnemonic LR6 SO4 3 — the lateral rectus is CN VI (abducens), the superior oblique is CN IV (trochlear), and everything else is CN III (oculomotor). CN III also carries parasympathetic fibers to the pupillary sphincter (miosis) and levator palpebrae (eyelid elevation), so a complete CN III palsy causes a "down and out" eye, ptosis, and a blown pupil.
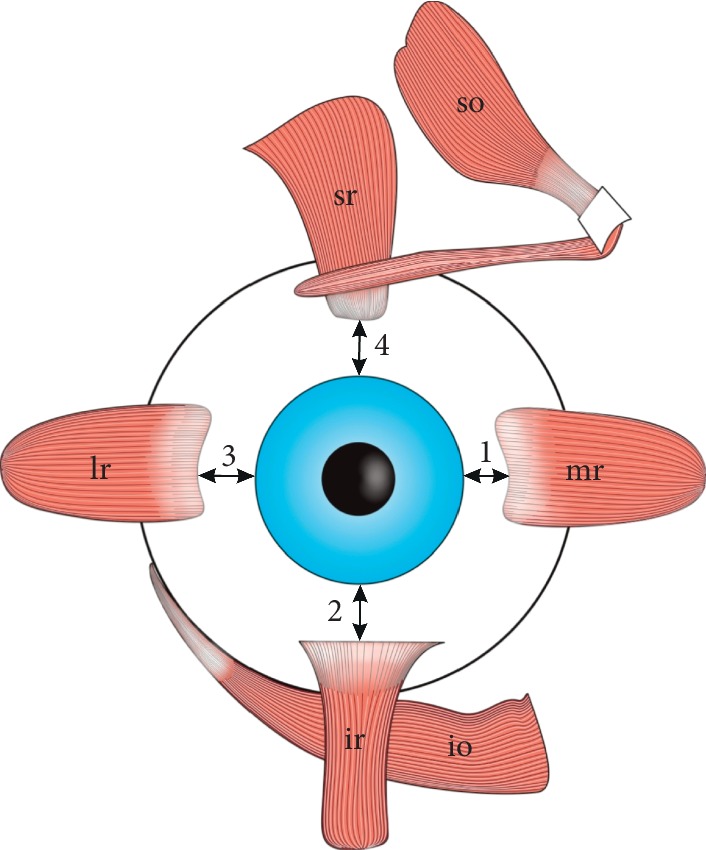
Visual Pathway
Light signals travel from the retinal photoreceptors through bipolar cells to retinal ganglion cells, whose axons form the optic nerve. At the optic chiasm, nasal fibers decussate; the optic tract then carries fibers to the lateral geniculate nucleus (LGN), where second-order neurons project via the optic radiations to the primary visual cortex (V1) in the occipital lobe. Understanding the pathway explains visual field defects: a lesion at the chiasm produces bitemporal hemianopsia, a lesion posterior to the chiasm produces a homonymous (congruous) hemianopsia, and a temporal lobe lesion produces a superior "pie-in-the-sky" defect via Meyer's loop.
Eyelids and Lacrimal System
The eyelids are composed (anterior to posterior) of skin, orbicularis oculi muscle (CN VII, closes the eye), orbital septum, tarsal plate (contains the meibomian glands), and palpebral conjunctiva. Tears are produced by the lacrimal gland in the superolateral orbit, spread across the ocular surface by blinking, and drain through the upper and lower puncta into the canaliculi, then the lacrimal sac, and finally the nasolacrimal duct into the inferior meatus of the nose. A blocked nasolacrimal duct causes epiphora (watering) and predisposes to dacryocystitis.
Abbreviations move fast in eye clinic. OD = right eye (oculus dexter), OS = left eye (oculus sinister), OU = both eyes (oculus uterque). You will chart these hundreds of times per day. Get them wrong once and you can put a wrong-eye surgery on the schedule. Double-check every laterality.
02 Scribe Documentation Framework (Eye-Specific)
Ophthalmology notes do not look like general medical notes. The structure is heavily templated around a repeatable data set: visual acuity, IOP, pupils, motility, confrontation fields, slit-lamp exam, and dilated fundus exam. The HPI is usually short, the exam is long, and the assessment/plan is organized by anatomic region.
HPIs are usually chief-complaint driven. The most common eye CCs are "blurry vision," "red eye," "eye pain," "flashes/floaters," "double vision," and "vision loss." For each, document: which eye (OD/OS/OU), onset (sudden vs gradual), duration, character, constant vs intermittent, associated symptoms (photophobia, halos, metamorphopsia, curtain in vision, scotoma), prior similar episodes, and what the patient has tried. For follow-up visits, also document interval change since last visit and current drop regimen compliance.
Every comprehensive eye exam chart has the same skeleton:
VAsc / VAcc: OD 20/__ OS 20/__ (with/without correction)
Manifest refraction: OD ___ OS ___
IOP (Goldmann / iCare / Tonopen): OD __ OS __ mmHg @ time
Pupils: PERRL, no APD OU
EOM: full OU, no pain/diplopia
CVF: full to finger counting OU
External / Lids / Lashes: WNL OU
Slit lamp: L/L, C/S, K, AC, I, L (each segment graded)
DFE: Disc, C/D ratio, Macula, Vessels, Periphery
Ophthalmology A/Ps are organized per diagnosis, each with its own mini-plan: the diagnosis (with laterality and stage), the plan, and the follow-up interval. A typical note might read: "1) Nuclear sclerotic cataract, OD, visually significant — recommend phacoemulsification with IOL; biometry today; scheduled for surgery. 2) POAG OU, controlled on latanoprost qhs — continue, RTC 4 months with VF and OCT RNFL. 3) NPDR, moderate, OU without DME — continue diabetes optimization, RTC 4 months for dilated exam."
Documentation Pearls Unique to Ophthalmology
Always write the laterality (OD, OS, OU) with every measurement, diagnosis, and intervention. Always include units with IOP (mmHg). Visual acuity is always fractional: the numerator is the test distance (usually 20 feet), the denominator is the smallest line the patient could read. "20/40" means the patient sees at 20 feet what a normal person sees at 40 feet. Document whether the acuity was without correction (VAsc, "sc"), with correction (VAcc, "cc"), or with pinhole (PH). A pinhole that improves vision suggests refractive error rather than organic disease.
When a patient cannot read the Snellen chart, acuity is progressively recorded as CF (counting fingers — at what distance), HM (hand motion), LP (light perception), or NLP (no light perception). NLP is the worst possible outcome — a functionally blind eye — and must never be documented without specific confirmation.
03 Refractive Error Optical
Refractive error is the single most common reason people see an eye care provider. It is an optical mismatch between the length of the eye and the focusing power of the cornea and lens. Refractive error is not a disease — it is a property that glasses, contact lenses, or surgery can correct.
Myopia (nearsightedness): The eye is too long (or the cornea too steep) relative to focal length, so light focuses in front of the retina. Patients see near objects clearly but distance is blurry. Corrected with a minus (concave) lens. High myopia (> −6.00 D) increases risk of retinal detachment, myopic maculopathy, and early cataract.
Hyperopia (farsightedness): The eye is too short, so light would focus behind the retina. Young hyperopes often see well at distance and near due to accommodation; adults develop blurry near vision first, then distance. Corrected with a plus (convex) lens. High hyperopia predisposes to narrow anterior chamber angles and angle-closure glaucoma.
Astigmatism: The cornea (or lens) has unequal curvature in different meridians, producing two focal points instead of one. Vision is distorted at all distances. Corrected with cylinder lenses.
Presbyopia: Age-related loss of accommodative amplitude as the crystalline lens stiffens (typically starting in the mid-40s). Patients lose the ability to focus on near objects. Corrected with reading glasses, bifocals, progressives, monovision contacts, or multifocal IOLs.
How the Refraction Is Documented
A manifest refraction is written as three numbers: sphere × cylinder × axis. For example, "−3.25 −1.50 × 180" means 3.25 diopters of myopia with 1.50 diopters of astigmatism at axis 180. Add-power for presbyopia is written after the distance refraction as "Add +2.50." Diopters are the unit of refractive power.
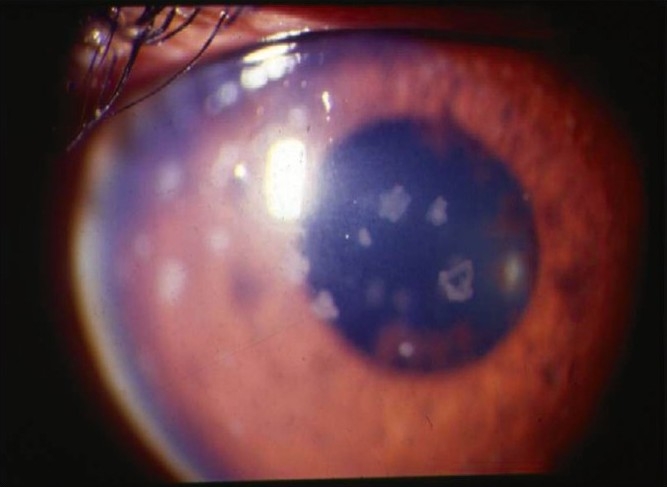
04 Cataracts Lens
Cataract is a clouding of the crystalline lens and is the leading cause of reversible blindness worldwide. It is the most common surgical diagnosis in ophthalmology: cataract surgery is the highest-volume surgical procedure performed in the United States. Every comprehensive ophthalmology clinic sees dozens of cataract patients weekly.
Morphology — The Three Main Types
| Type | Location | Clinical Features |
|---|---|---|
| Nuclear sclerotic (NS) | Central lens nucleus | Most common; age-related; yellow/brown discoloration; causes myopic shift ("second sight") |
| Cortical | Lens cortex, spoke-like opacities | Associated with diabetes and UV exposure; glare and halos especially at night |
| Posterior subcapsular (PSC) | Posterior lens capsule | Associated with steroid use, diabetes, radiation; disproportionately affects near vision and reading |
Clinical Presentation & Workup
Cataract patients complain of gradually worsening blurry vision, glare and halos around lights (especially at night when driving), difficulty reading, and loss of contrast sensitivity. Visual acuity measured with glare testing (BAT) often drops several lines. Workup includes BCVA, refraction, IOP, slit-lamp grading of the cataract, dilated fundus exam to rule out concomitant macular disease, and — preoperatively — biometry (IOL master or immersion A-scan) to calculate intraocular lens power.
The Lens Opacities Classification System III standardizes cataract grading at the slit lamp using reference photographs. Nuclear opalescence (NO) and nuclear color (NC) are graded 0.1–6.9; cortical (C) and posterior subcapsular (P) are graded 0.1–5.9. In everyday documentation, most providers use simple "1+ to 4+" severity (trace, mild, moderate, dense/mature) rather than formal LOCS grading.
Management
The only treatment is surgical removal of the cloudy lens and implantation of an intraocular lens (IOL). The AAO Cataract Preferred Practice Pattern is the reference guideline. Standard technique is phacoemulsification: a small (2.2–2.8 mm) clear corneal incision, capsulorhexis, hydrodissection, ultrasonic lens fragmentation and aspiration, and implantation of a foldable IOL in the capsular bag. IOL choices include monofocal (sets one focal distance), toric (corrects astigmatism), multifocal/EDOF (provides range of vision), and light-adjustable lenses.
The cataract consent discussion is a high-density documentation moment: surgeon will quote risks (endophthalmitis ∼1/2000, retinal detachment, cystoid macular edema, posterior capsule rupture, dropped nucleus, corneal edema), discuss IOL options, target refraction, and anesthesia plan. Capture it all.
05 Glaucoma Neuro-Optic
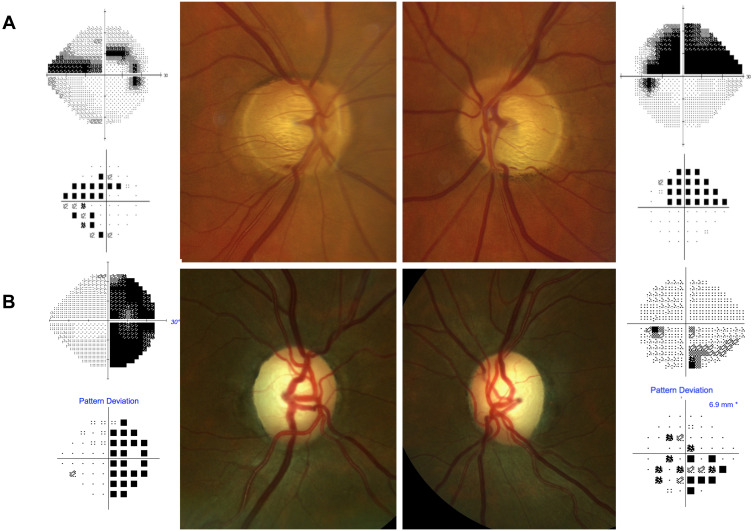
Glaucoma is an optic neuropathy characterized by progressive retinal ganglion cell loss, optic nerve head cupping, and corresponding visual field defects. It is the leading cause of irreversible blindness worldwide. Elevated IOP is the most important modifiable risk factor, but glaucoma can occur at any IOP. Management is lifelong and centers on IOP reduction.
Types of Glaucoma
The most common type. Open iridocorneal angle on gonioscopy but impaired trabecular outflow. Painless, bilateral, and asymptomatic in early stages — patients are often diagnosed on routine exam when optic nerve cupping or an elevated IOP is detected. Risk factors: age, family history, African ancestry, elevated IOP, thin central cornea, high myopia. Diagnosis requires characteristic optic nerve damage and corresponding visual field defect. The AAO POAG Preferred Practice Pattern governs evaluation and target IOP setting.
Ophthalmologic emergency. Sudden mechanical blockage of the trabecular meshwork by the peripheral iris causes IOP to rise to 40–80+ mmHg. Classic presentation: severe unilateral eye pain, nausea and vomiting, blurred vision with halos, fixed mid-dilated pupil, and a red, steamy cornea. Treat immediately with topical and oral IOP-lowering medications, then laser peripheral iridotomy.
Glaucomatous optic nerve damage and field loss despite IOPs in the "normal" range (≤ 21 mmHg). Associated with vasospasm, migraines, and obstructive sleep apnea. Managed similarly to POAG with further IOP lowering.
Pseudoexfoliation (PXF): flaky deposits on lens and pupillary margin, trabecular meshwork clogging. Pigmentary: iris pigment dispersion, Krukenberg spindle. Neovascular: new vessel growth on iris from retinal ischemia (diabetes, CRVO). Steroid-induced: chronic topical or systemic steroids. Uveitic: inflammation blocks outflow. Traumatic (angle recession): blunt ocular injury.
Congenital Glaucoma
Rare but vision-threatening. Infants present with the classic triad of epiphora, photophobia, and blepharospasm, plus corneal enlargement (buphthalmos) and clouding. Treatment is surgical (goniotomy or trabeculotomy).
Workup
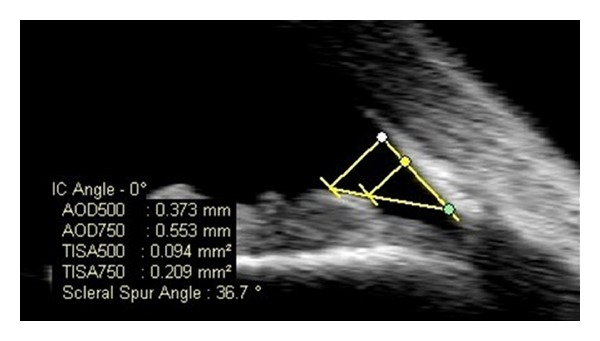
IOP (Goldmann applanation is gold standard), gonioscopy (to determine angle open vs closed), pachymetry (central corneal thickness influences IOP measurement), optic nerve imaging (OCT RNFL and ganglion cell complex), and automated visual field testing (24-2 or 10-2 Humphrey). Progression is tracked by comparing serial OCTs and visual fields.
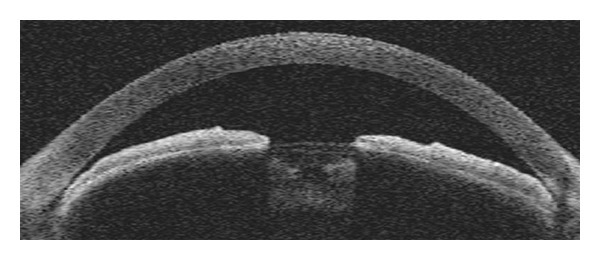
Management
First-line is usually topical medication. Drop classes and mechanisms are enumerated in Section 18. If drops fail or compliance is poor, selective laser trabeculoplasty (SLT), minimally invasive glaucoma surgery (MIGS), trabeculectomy, or tube shunts (Ahmed, Baerveldt) follow a stepwise escalation.
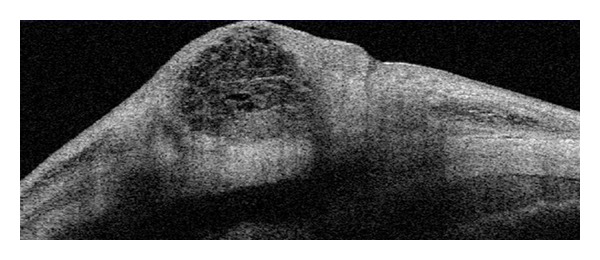
06 Diabetic Retinopathy & DME Retina
Diabetic retinopathy (DR) is a microvascular complication of diabetes and a leading cause of blindness in working-age adults. Every diabetic patient needs a yearly dilated fundus exam. Duration of diabetes and glycemic control are the dominant risk factors.
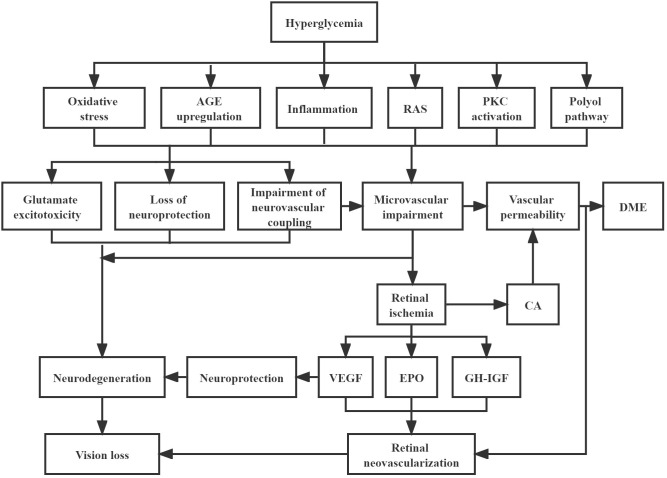
Pathophysiology
Chronic hyperglycemia damages retinal capillary endothelium, causing microaneurysm formation, capillary non-perfusion, and breakdown of the blood-retinal barrier. As ischemia worsens, VEGF release drives neovascularization — the hallmark of proliferative disease. Leakage from abnormal vessels or microaneurysms causes macular edema.
ETDRS-based Severity Staging
| Stage | Clinical Findings |
|---|---|
| No DR | No abnormalities |
| Mild NPDR | Microaneurysms only |
| Moderate NPDR | More than "mild" but less than "severe" — microaneurysms, dot-blot hemorrhages, cotton-wool spots, hard exudates |
| Severe NPDR | 4-2-1 rule: diffuse intraretinal hemorrhages in all 4 quadrants, OR venous beading in 2+ quadrants, OR prominent IRMA in 1+ quadrant |
| PDR | Neovascularization of disc (NVD), elsewhere (NVE), iris (NVI), or angle (NVA); preretinal or vitreous hemorrhage; tractional retinal detachment |
Diabetic macular edema (DME) can occur at any stage of DR and is independently graded. Center-involving DME threatens central vision and is the main indication for anti-VEGF therapy.
Management
Systemic glycemic, BP, and lipid control. Anti-VEGF intravitreal injections (bevacizumab, ranibizumab, aflibercept, faricimab) are first-line for center-involving DME and for PDR per the DRCR Protocol T and AAO DR Preferred Practice Pattern. Panretinal photocoagulation (PRP) remains the backbone treatment for high-risk PDR. Focal/grid laser is used for non-center-involving DME. Vitrectomy is used for non-clearing vitreous hemorrhage or tractional detachment.
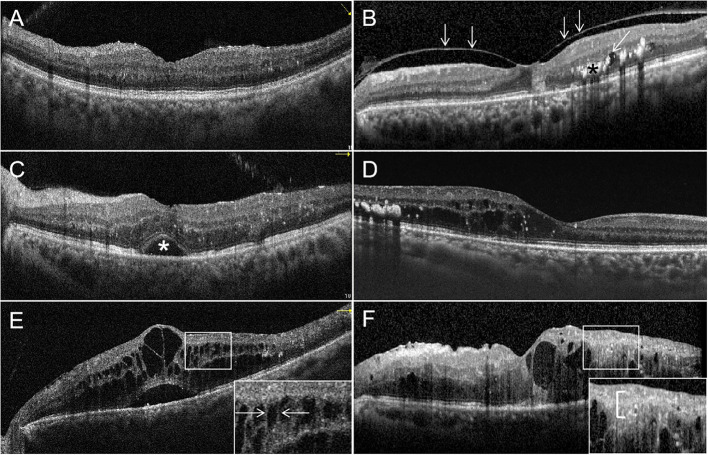
07 Age-Related Macular Degeneration Retina
AMD is the leading cause of severe central vision loss in adults over 60 in developed countries. It affects the macula — the cone-rich central retina — producing central scotomas and metamorphopsia while sparing peripheral vision. The two forms are dry (non-neovascular) and wet (neovascular).
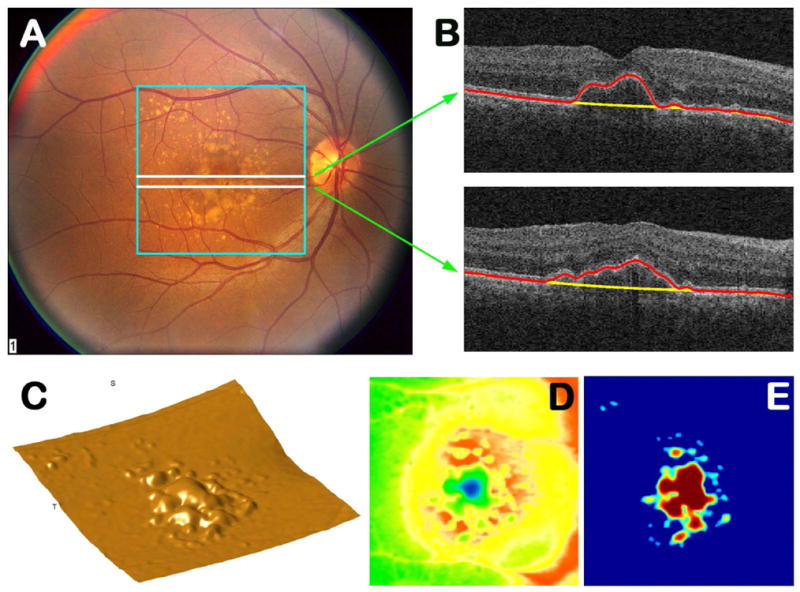
Dry vs Wet
Dry AMD accounts for ∼85–90% of cases. Hallmarks are drusen (yellow lipid/protein deposits beneath the RPE), RPE pigmentary changes, and in advanced disease geographic atrophy (GA). Vision loss is typically gradual. Wet AMD is defined by choroidal neovascularization (CNV) — abnormal vessels from the choroid break through Bruch's membrane and leak fluid or blood under or within the retina, causing rapid central vision loss and metamorphopsia.
AREDS Categories
| Category | Definition |
|---|---|
| 1 | No/few small drusen (< 63 μm); no AMD |
| 2 (Early AMD) | Multiple small drusen or few intermediate drusen (63–125 μm), or RPE abnormalities |
| 3 (Intermediate AMD) | Extensive intermediate drusen, at least one large druse (≥ 125 μm), or non-central GA |
| 4 (Advanced AMD) | GA involving the foveal center OR any neovascular AMD |
Management
Dry AMD: AREDS2 vitamin formulation (vitamin C, E, lutein, zeaxanthin, zinc, copper) for intermediate and unilateral advanced disease. Newer complement inhibitors (pegcetacoplan, avacincaptad pegol) slow geographic atrophy progression. Wet AMD: intravitreal anti-VEGF therapy (bevacizumab/Avastin, ranibizumab/Lucentis, aflibercept/Eylea, brolucizumab/Beovu, faricimab/Vabysmo), typically every 4–8 weeks initially, supported by landmark ANCHOR and MARINA trials. Home Amsler grid monitoring is critical.
08 Retinal Detachment & Vitreoretinal Interface Retina
Retinal detachment is a surgical emergency — the neurosensory retina separates from the RPE, cutting off outer retinal nutrition. Untreated, it leads to permanent vision loss.
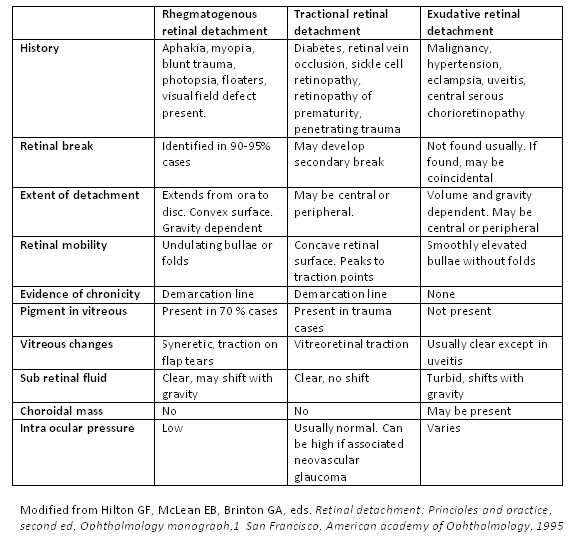
Rhegmatogenous (RRD): the most common type. A break/tear in the retina (often from PVD or lattice degeneration) allows liquefied vitreous to pass under and lift the retina. Risk factors: high myopia, prior cataract surgery, trauma, lattice degeneration. Classic symptoms: flashes, new floaters, and a "curtain" coming over vision.
Tractional (TRD): fibrovascular proliferation (most often from PDR, also sickle cell and ROP) pulls the retina off mechanically. Presentation is slower.
Exudative (serous): fluid accumulates under the retina from inflammatory or neoplastic processes (choroidal tumor, posterior scleritis, VKH, central serous), without a break.
Related Vitreoretinal Conditions
Posterior vitreous detachment (PVD): age-related separation of the vitreous from the retina — presents with flashes and floaters; retinal examination is essential to rule out a tear. Vitreous hemorrhage: blood in the vitreous cavity from torn retinal vessels, PDR, or trauma — patient reports sudden dense floaters or red/black shower. Macular hole: a full-thickness break in the foveal center from vitreomacular traction; causes central scotoma and metamorphopsia; treated with vitrectomy + ILM peel + gas tamponade. Epiretinal membrane (ERM): a contractile cellular sheet on the inner retina causing macular wrinkling and distortion; treated with vitrectomy and membrane peeling if symptomatic.
Management
Per the AAO Retinal Detachment Preferred Practice Pattern: rhegmatogenous detachments are repaired by pneumatic retinopexy (gas bubble + laser/cryo), scleral buckle, pars plana vitrectomy (PPV), or a combination, depending on break location, extent, and surgeon preference. Retinal tears without detachment are treated prophylactically with laser retinopexy or cryotherapy.
09 Vascular Occlusions (RVO, RAO) Retina
Sudden, painless vision loss in an older patient with vasculopathic risk factors should immediately raise concern for a retinal vascular occlusion.
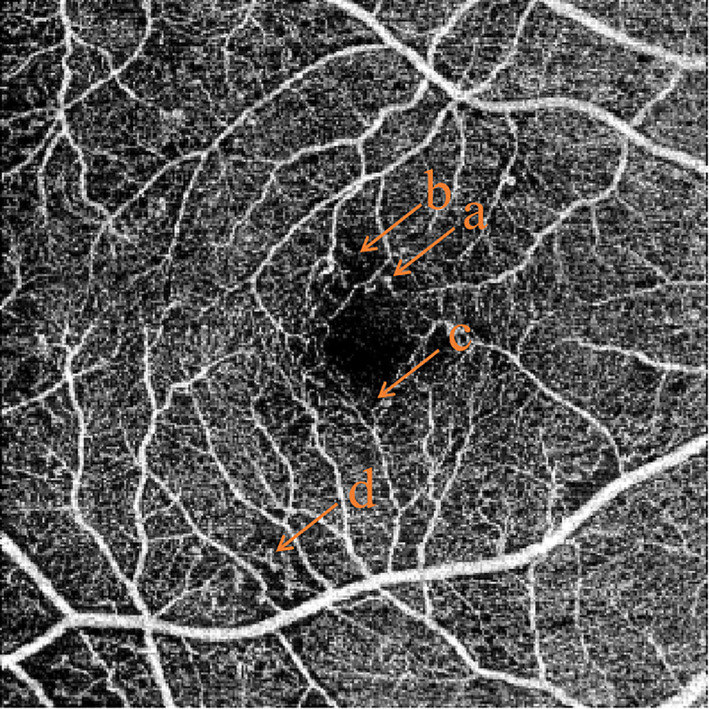
Retinal Vein Occlusions
CRVO (central retinal vein occlusion): thrombosis at or posterior to the lamina cribrosa. Fundus shows "blood and thunder" appearance — dilated tortuous veins, diffuse intraretinal hemorrhages in all four quadrants, cotton-wool spots, disc edema. Ischemic CRVO (> 10 disc areas of non-perfusion) carries high risk of 90-day neovascular glaucoma ("100-day glaucoma"). BRVO (branch): occlusion of a tributary vein, typically at an AV crossing; sector-shaped hemorrhages. Primary treatment of associated macular edema is anti-VEGF injections.
Retinal Artery Occlusions
Central retinal artery occlusion is an acute ischemic stroke equivalent. Sudden painless monocular vision loss with a pale retina and a cherry-red spot at the fovea (the thin foveal retina allows the underlying choroid to show through). Workup is the same as stroke: urgent carotid imaging, cardiac evaluation, GCA workup (ESR, CRP, temporal artery biopsy) in patients over 50. Refer to stroke service for acute management. BRAO (branch) causes a sectoral field defect.
10 Uveitis Inflammatory
Uveitis is inflammation of the uveal tract. It is classified anatomically by the primary site of inflammation — a schema that drives both workup and treatment.
| Type | Primary Site | Classic Features |
|---|---|---|
| Anterior uveitis (iritis, iridocyclitis) | Iris ± ciliary body | Pain, photophobia, red eye, ciliary flush, cell/flare in AC, KPs, miotic pupil, synechiae |
| Intermediate uveitis (pars planitis) | Vitreous, pars plana | Floaters, blurred vision, vitritis, snowballs, snowbanking on pars plana |
| Posterior uveitis | Retina and/or choroid | Floaters, blurred vision, chorioretinal lesions, vasculitis |
| Panuveitis | All segments | Combined features; think Behçet, VKH, sarcoidosis, sympathetic ophthalmia |
Specific named entities you will encounter: HSV iritis (often unilateral with elevated IOP and sectoral iris atrophy), Posner-Schlossman syndrome (glaucomatocyclitic crisis, markedly elevated IOP with minimal inflammation), Fuchs heterochromic iridocyclitis (chronic low-grade unilateral iritis, heterochromia, cataract, no synechiae), toxoplasma chorioretinitis ("headlight in the fog" over an old chorioretinal scar), CMV retinitis (in immunocompromised, "pizza-pie" retinitis), acute retinal necrosis (ARN) from HSV/VZV (emergency, requires IV acyclovir), Vogt-Koyanagi-Harada (bilateral panuveitis with serous RDs, vitiligo, poliosis, dysacusis), sympathetic ophthalmia (bilateral panuveitis after penetrating trauma to one eye).
Systemic associations include HLA-B27 (ankylosing spondylitis, reactive arthritis, IBD), sarcoidosis, syphilis, tuberculosis, toxoplasmosis, herpes viruses, juvenile idiopathic arthritis, VKH, and Behçet. Workup for recurrent or bilateral uveitis typically includes CBC, CMP, ACE, lysozyme, RPR/FTA-ABS, QuantiFERON-TB, HLA-B27, chest imaging, and targeted serologies. Management includes topical cyclopentolate (cycloplegic) and topical corticosteroids for anterior disease, periocular/intravitreal or oral steroids for posterior/intermediate disease, and steroid-sparing immunomodulators (methotrexate, mycophenolate, azathioprine, TNF-alpha inhibitors) for chronic cases, per the AAO Uveitis Preferred Practice Pattern.
11 Cornea, Conjunctiva & External Disease Anterior
Conjunctivitis
| Type | Discharge | Key Features |
|---|---|---|
| Viral (adenovirus) | Watery | Often bilateral, preauricular lymphadenopathy, starts unilateral; highly contagious; follicular reaction |
| Bacterial | Purulent | Crusting lids, mucopurulent discharge; hyperacute discharge suggests gonococcal |
| Allergic | Stringy mucus | Itching is the cardinal symptom; bilateral; chemosis; papillae |
| Giant papillary (GPC) | Mucus | Contact lens wearers; upper tarsal giant papillae |
Refer to the AAO Conjunctivitis Preferred Practice Pattern for management.
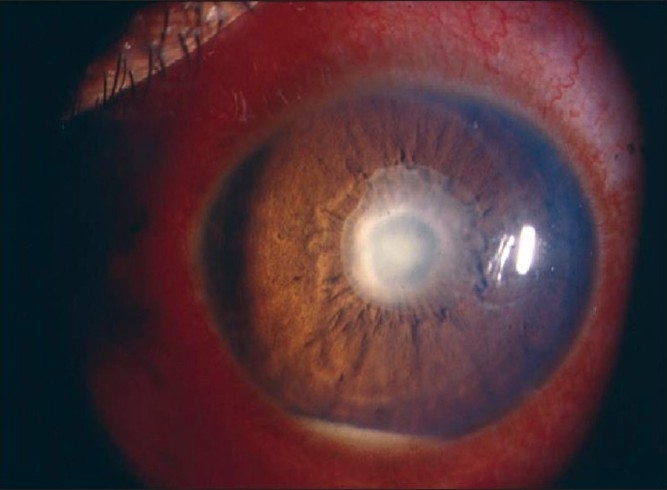
Keratitis and Corneal Disease
Bacterial keratitis: painful red eye with a stromal infiltrate, often in contact lens wearers (Pseudomonas). Treat with fortified antibiotic drops hourly. HSV keratitis: dendritic epithelial ulcer (fluorescein reveals branching dendrites); treat with topical ganciclovir gel or oral acyclovir/valacyclovir. Avoid topical steroids in epithelial HSV. Fungal keratitis: after trauma with vegetable matter or in contact lens wearers; feathery stromal infiltrate with satellite lesions; natamycin or voriconazole. Acanthamoeba keratitis: in contact lens wearers using tap water; severe pain out of proportion, ring infiltrate.
Corneal abrasion: epithelial defect stained by fluorescein; treat with lubricating or antibiotic drops and pain control. Recurrent corneal erosion: sharp morning pain at lid opening; managed with lubricants, hypertonic saline, bandage contact lens, or anterior stromal puncture. Dry eye disease is an enormous category — evaporative (MGD-driven) and aqueous-deficient subtypes — staged by the International Dry Eye Workshop (DEWS II) framework (see Section 19). Management per the AAO Dry Eye Preferred Practice Pattern: artificial tears, warm compresses/lid hygiene, cyclosporine (Restasis/Cequa), lifitegrast (Xiidra), varenicline nasal spray, punctal plugs, serum tears.
Other Anterior Segment Conditions
Blepharitis: eyelid margin inflammation, anterior (staphylococcal, seborrheic) or posterior (MGD); lid hygiene cornerstone. Pterygium: fibrovascular triangular growth from conjunctiva onto cornea, typically nasal, sun-related. Pinguecula: yellowish conjunctival nodule, cosmetic. Chalazion: chronic, non-infectious, sterile lipogranuloma of meibomian gland; warm compresses, intralesional steroid, or incision and curettage. Hordeolum (stye): acute, painful, infected meibomian or Zeis gland lesion.
12 Eyelid, Lacrimal & Orbital Disease Adnexa
Ptosis: drooping of the upper eyelid. Aponeurotic (age-related stretching of levator aponeurosis) is most common; other causes include myogenic (myasthenia gravis, CPEO), neurogenic (CN III palsy, Horner syndrome), mechanical, and congenital. Measure MRD1 (margin reflex distance) and levator function. Entropion: lid turned inward, lashes rub cornea. Ectropion: lid turned outward, exposure keratopathy and epiphora. Both typically age-related; surgical repair is definitive.
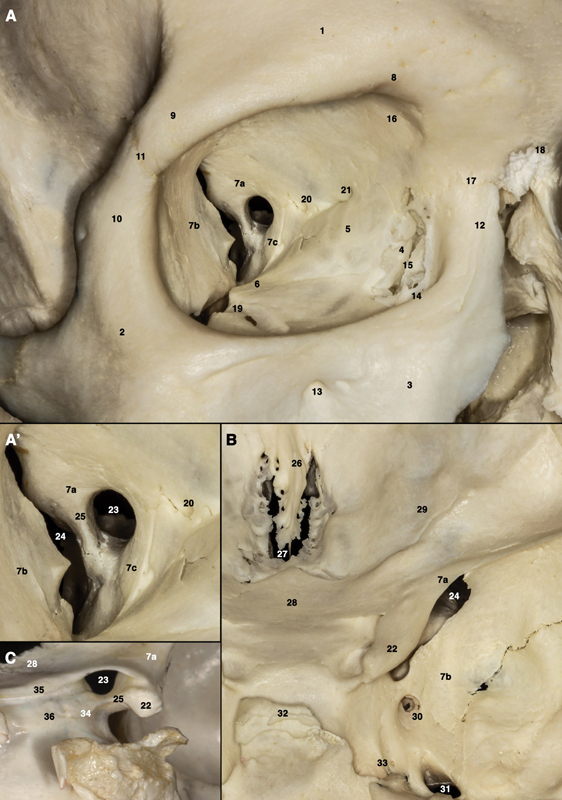
Dacryocystitis: infection of the lacrimal sac due to nasolacrimal duct obstruction; painful, red, swollen mass at the medial canthus. Treat with warm compresses, oral antibiotics (amoxicillin-clavulanate), and definitive dacryocystorhinostomy (DCR) to bypass the obstruction.
Preseptal cellulitis: infection anterior to the orbital septum — eyelid erythema and swelling without proptosis, vision loss, or painful EOM restriction. Treat with oral antibiotics and close follow-up.
Orbital cellulitis: infection posterior to the orbital septum, usually from ethmoid sinusitis. Emergency. Presents with proptosis, painful limitation of EOMs, decreased vision, chemosis, and fever. Requires urgent CT sinus/orbit, IV antibiotics, and ENT/ophthalmology co-management. Complications include subperiosteal abscess, cavernous sinus thrombosis, meningitis, and vision loss.
Orbital fractures: blowout fractures (usually inferior or medial wall) from blunt trauma. Look for enophthalmos, restricted upgaze (inferior rectus entrapment), diplopia, and infraorbital hypoesthesia. CT orbit without contrast. Pediatric "white-eyed" blowouts with trapped muscle are surgical emergencies due to oculocardiac reflex risk.
Thyroid eye disease (Graves orbitopathy): autoimmune orbitopathy with EOM and fat enlargement; lid retraction, proptosis, restrictive strabismus, and rarely compressive optic neuropathy.
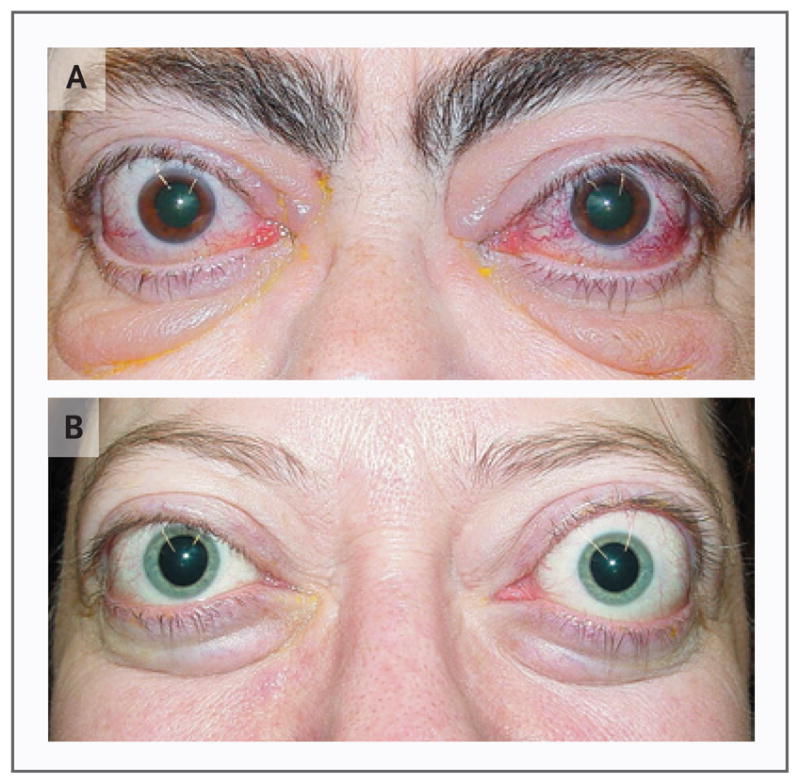
13 Neuro-Ophthalmology Neuro-Optic
Optic neuritis: inflammation of the optic nerve, most commonly demyelinating (multiple sclerosis, NMO-SD, MOG-AD). Young patients with unilateral subacute vision loss, eye pain worse with movement, dyschromatopsia (especially red desaturation), and a relative afferent pupillary defect (RAPD). Fundus may be normal (retrobulbar) or show disc edema. MRI brain and orbit with contrast is mandatory.
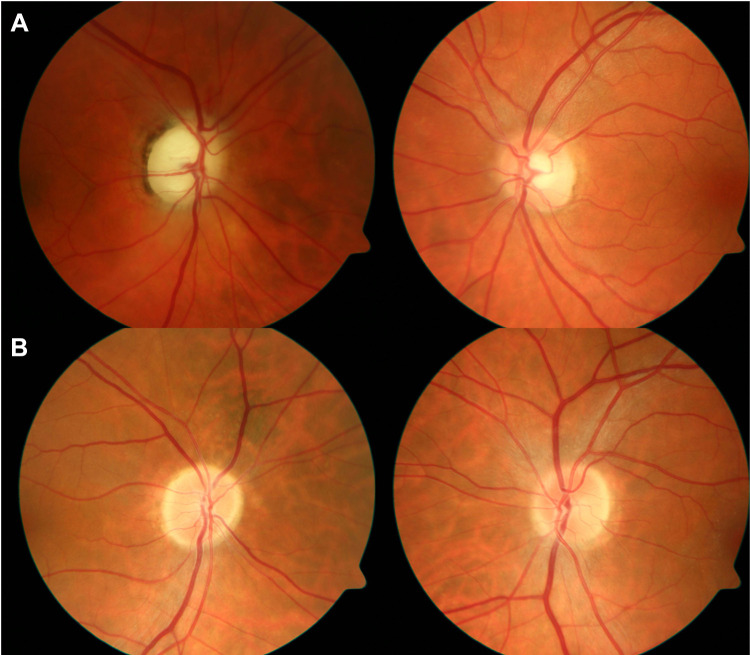
Papilledema: bilateral optic disc edema from elevated intracranial pressure. Presents with transient visual obscurations, headaches, and enlarged blind spots. Workup with urgent neuroimaging (rule out mass/venous sinus thrombosis) then lumbar puncture for opening pressure. Idiopathic intracranial hypertension (IIH) is the classic diagnosis in young, overweight women — managed with weight loss and acetazolamide, occasionally ONSF or CSF shunting.
Ischemic optic neuropathy: NAION (non-arteritic) — sudden painless unilateral vision loss with altitudinal field defect in a patient > 50 with small cup-to-disc ratio ("disc at risk") and vasculopathic risk factors. AION (arteritic) — giant cell arteritis; mandatory to rule out in any patient > 50 with vision loss. Check ESR, CRP, platelets immediately; start empiric high-dose IV methylprednisolone and obtain temporal artery biopsy.
Cranial nerve palsies: CN III: "down and out" eye, ptosis, ± pupil involvement; pupil-involving is aneurysm until proven otherwise (PCOM aneurysm). CN IV: vertical diplopia, head tilt away from affected side; most common cause of acquired vertical diplopia. CN VI: horizontal diplopia worse at distance; abduction deficit; non-localizing sign in elevated ICP.
Horner syndrome: miosis, ptosis, and anhidrosis from sympathetic chain disruption. Pharmacologic confirmation with apraclonidine (reverses anisocoria in the affected pupil). Localization by lesion level: first-order (brainstem, spinal cord), second-order (Pancoast tumor, apical lung), third-order (carotid dissection — painful Horner is a carotid dissection until proven otherwise, a neurologic emergency).
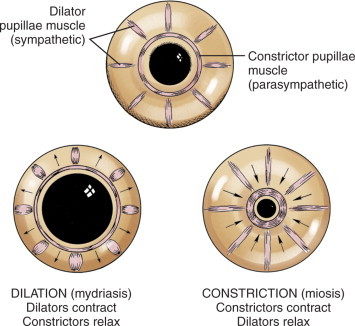
Internuclear ophthalmoplegia (INO): lesion of the medial longitudinal fasciculus (MLF). On attempted lateral gaze, the adducting eye lags or fails to cross the midline while the abducting eye shows nystagmus. In young patients consider multiple sclerosis; in older patients consider brainstem stroke.
14 Pediatric Ophthalmology & Strabismus Pediatric
Amblyopia ("lazy eye"): reduced best-corrected vision in an otherwise structurally normal eye from abnormal visual development in childhood. Causes: strabismic, refractive (anisometropic or isoametropic), and deprivational (congenital cataract, ptosis). Treated with refractive correction, patching, or atropine penalization of the better eye before cortical plasticity closes (usually by age 8–10).
Strabismus: ocular misalignment. Eso- = inward, exo- = outward, hyper- = upward, hypo- = downward. -tropia is a manifest deviation; -phoria is latent. Document deviation in prism diopters at distance and near. Treatment ranges from glasses (accommodative esotropia) and prisms to extraocular muscle surgery (recession, resection).
Retinopathy of prematurity (ROP): abnormal retinal vascularization in premature infants exposed to oxygen. Screen per birth weight and gestational age guidelines. Treat threshold/Type 1 disease with laser ablation of avascular retina or intravitreal anti-VEGF.
Congenital cataract: must be identified and removed early to prevent deprivation amblyopia. Check red reflex on every newborn exam. Retinoblastoma: most common intraocular malignancy of childhood; leukocoria (white pupil reflex) and strabismus are the two most common presentations. Urgent referral.
15 Ocular Trauma & Emergencies Trauma
Alkali burns are worse than acid (saponification and deeper penetration). Irrigate first, ask questions later: copious irrigation with saline or LR until pH normalizes (7.0–7.4). Check pH with litmus paper every 30 minutes. Classify severity using the Roper-Hall or Dua grading (limbal ischemia + corneal clarity). Topical steroids early, cycloplegics, doxycycline, ascorbate.
Corneal/conjunctival foreign body: topical anesthetic, slit-lamp evaluation, fluorescein staining, removal with spud or 25-gauge needle, rust ring removal, topical antibiotic. Evert the upper lid to check for subtarsal FBs.
Hyphema: blood in the anterior chamber from blunt trauma. Graded I (< 1/3), II (1/3–1/2), III (1/2–total), IV (8-ball, total). Elevate head of bed, shield eye, cycloplegic, topical steroid, monitor IOP. Sickle cell patients are at high risk of rebleed and optic neuropathy at lower pressures. Rebleeding peaks at days 2–5.
Traumatic iritis: cell and flare after blunt trauma; treat with cycloplegic and topical steroid. Globe rupture / open globe: teardrop pupil, positive Seidel sign, 360° subconjunctival hemorrhage, low IOP, prolapsed uveal tissue. NPO, shield the eye (no pressure patching), IV antibiotics, tetanus, antiemetics, and to the OR.
Endophthalmitis: intraocular infection, usually post-op (especially 3–7 days after cataract surgery) or post-injection. Severe pain, hypopyon, vitritis, dramatic vision loss. Emergent vitreous tap and intravitreal antibiotics (vancomycin + ceftazidime). Vitrectomy per EVS for LP-only vision.
16 Ophthalmic Procedures — A to Z
| Procedure | Description | Typical Indication |
|---|---|---|
| Phacoemulsification + IOL | Ultrasonic lens removal through clear corneal incision with foldable IOL implantation | Visually significant cataract |
| YAG capsulotomy | Nd:YAG laser opening of opacified posterior capsule | Posterior capsular opacification after cataract surgery |
| LASIK | Femtosecond/microkeratome flap + excimer stromal ablation | Myopia, hyperopia, astigmatism |
| PRK | Surface excimer ablation after epithelial removal | Refractive error in thin corneas or contact sport athletes |
| SMILE | Femtosecond-extracted lenticule through small incision | Myopic correction |
| Corneal cross-linking (CXL) | Riboflavin + UV-A to stiffen cornea | Progressive keratoconus |
| PKP | Full-thickness penetrating keratoplasty | Severe corneal scarring or decompensation |
| DSAEK / DMEK | Endothelial keratoplasty (selective back layer transplant) | Fuchs dystrophy, pseudophakic bullous keratopathy |
| Trabeculectomy | Scleral flap creates filtering bleb | Uncontrolled glaucoma |
| Tube shunt (Ahmed, Baerveldt) | Silicone tube drains aqueous to equatorial plate | Failed trab or complex glaucoma |
| MIGS (iStent, Hydrus, Xen, Kahook) | Minimally invasive Schlemm's canal or subconjunctival devices | Mild-moderate POAG, often combined with cataract |
| Laser peripheral iridotomy (LPI) | YAG laser iris opening | Angle closure / narrow angles |
| Selective laser trabeculoplasty (SLT) | Nd:YAG laser to trabecular meshwork | POAG first-line or adjunct |
| Intravitreal injection | Anti-VEGF or steroid injection into vitreous cavity via pars plana | Wet AMD, DME, RVO, PDR |
| Panretinal photocoagulation (PRP) | Scatter laser burns to peripheral retina | High-risk PDR, neovascular glaucoma |
| Focal / grid laser | Targeted macular laser | Non-center-involving DME |
| Pars plana vitrectomy (PPV) | Removal of vitreous via 23/25/27-gauge trocars | RD, vitreous hemorrhage, macular hole, ERM, endophthalmitis |
| Scleral buckle | Silicone band indents sclera to support retinal break | Rhegmatogenous RD |
| Pneumatic retinopexy | Intravitreal gas bubble + cryo/laser | Select superior RRDs |
| Blepharoplasty | Excess skin/fat removal from upper or lower lids | Dermatochalasis, cosmetic |
| Ptosis repair | Levator advancement, Müller muscle resection, frontalis sling | Aponeurotic, myogenic, or congenital ptosis |
| Strabismus surgery | EOM recession / resection / transposition | Misalignment refractory to prisms |
| Dacryocystorhinostomy (DCR) | External or endonasal creation of lacrimal sac-to-nasal anastomosis | Nasolacrimal duct obstruction, chronic dacryocystitis |
| Enucleation / Evisceration | Removal of globe ± intraocular contents with implant placement | Blind painful eye, intraocular malignancy |
17 Ophthalmic Imaging & Diagnostics
Optical coherence tomography (OCT): non-contact cross-sectional imaging of the retina and optic nerve. Macular OCT: detects DME, AMD (drusen, SRF/IRF, PED), ERM, macular hole. Optic nerve OCT (RNFL, ganglion cell complex): tracks glaucoma progression.
Fluorescein angiography (FA): IV fluorescein then serial fundus photography; shows vascular leakage, non-perfusion, neovascularization.
Indocyanine green angiography (ICG): better visualizes choroidal circulation; used in CNV, central serous, polypoidal choroidal vasculopathy.
OCT-angiography (OCTA): dye-free imaging of retinal capillary plexuses.
Humphrey visual field (HVF): automated perimetry. 24-2 for glaucoma; 10-2 for advanced disease or macular involvement. Report terms: MD (mean deviation), PSD (pattern standard deviation), VFI (visual field index), GHT (glaucoma hemifield test).
Goldmann visual field: kinetic perimetry with moving test targets; used for neuro-ophthalmic and advanced glaucoma assessment.
B-scan ultrasonography: used when view is obscured (dense cataract, vitreous hemorrhage) to evaluate retina, choroid, posterior segment masses.
Corneal topography / tomography: Placido disc (topography) or Scheimpflug (Pentacam) mapping of corneal curvature and thickness; keratoconus, refractive surgery planning.
Specular microscopy: endothelial cell count and morphology; important before cataract or intraocular surgery.
Biometry (IOLMaster, Lenstar, immersion A-scan): measures axial length, keratometry, anterior chamber depth for IOL power calculation.
18 Medications You Must Know
Glaucoma Drops by Class
| Class | Examples (Generic / Brand) | Mechanism | Key Notes |
|---|---|---|---|
| Prostaglandin analogs (PGA) | Latanoprost (Xalatan), travoprost (Travatan Z), bimatoprost (Lumigan), tafluprost (Zioptan), latanoprostene bunod (Vyzulta) | ↑ uveoscleral outflow | First-line; qhs dosing; lash growth, iris darkening, periorbital fat atrophy (PAP) |
| Beta-blockers | Timolol (Timoptic, Istalol), betaxolol, levobunolol | ↓ aqueous production | BID; avoid in asthma, COPD, bradycardia, heart block |
| Alpha-2 agonists | Brimonidine (Alphagan P) | ↓ production, ↑ uveoscleral outflow | TID; allergic conjunctivitis; avoid in infants (CNS depression) |
| Topical CAIs | Dorzolamide (Trusopt), brinzolamide (Azopt) | ↓ aqueous production | BID-TID; sulfa allergy caution; stinging |
| Rho kinase inhibitors | Netarsudil (Rhopressa), netarsudil-latanoprost (Rocklatan) | ↑ trabecular outflow | qhs; conjunctival hyperemia, cornea verticillata |
| Miotics | Pilocarpine | ↑ trabecular outflow | Rarely used chronically; used acutely in angle closure |
| Oral CAI | Acetazolamide (Diamox), methazolamide | Systemic aqueous suppression | Acute IOP reduction; paresthesias, metabolic acidosis, nephrolithiasis |
| Fixed combinations | Cosopt (timolol-dorzolamide), Combigan (timolol-brimonidine), Simbrinza (brinzolamide-brimonidine) | Combined | Improve compliance |
Anti-VEGF Injections
| Agent | Brand | Notes |
|---|---|---|
| Bevacizumab | Avastin | Off-label, inexpensive; most-used anti-VEGF in the US |
| Ranibizumab | Lucentis / Byooviz | Fab fragment, FDA-approved for AMD, DME, RVO |
| Aflibercept | Eylea / Eylea HD | VEGF-trap; extended interval dosing |
| Brolucizumab | Beovu | Small Fv; intraocular inflammation signal |
| Faricimab | Vabysmo | Bispecific anti-VEGF-A + Ang-2; extended dosing |
Anti-infective and Anti-inflammatory Drops
Antibiotics: moxifloxacin (Vigamox), gatifloxacin (Zymaxid), besifloxacin (Besivance), tobramycin, erythromycin ointment, polymyxin-trimethoprim (Polytrim), fortified vancomycin/tobramycin for severe keratitis. Topical steroids: prednisolone acetate 1% (Pred Forte), difluprednate (Durezol), loteprednol (Lotemax), dexamethasone, fluorometholone. Watch IOP on long-term steroid use (steroid-response glaucoma). Topical NSAIDs: ketorolac (Acular), bromfenac (Prolensa), nepafenac (Ilevro), diclofenac. Used for post-op CME prevention and allergic eye disease. Antivirals: trifluridine, ganciclovir gel (Zirgan) for HSV epithelial keratitis; oral acyclovir 400 mg 5x/day or valacyclovir 500 mg TID for HSV keratitis/uveitis and zoster ophthalmicus (1 g TID). Antifungals: natamycin 5% (filamentous), voriconazole, amphotericin B (yeast). Immunomodulators: cyclosporine 0.05/0.09% (Restasis, Cequa), lifitegrast 5% (Xiidra), perfluorohexyloctane (Miebo), varenicline (Tyrvaya) for dry eye. Mydriatics/cycloplegics: tropicamide 1%, phenylephrine 2.5%, cyclopentolate 1% (exam dilation); atropine 1%, homatropine 5% (uveitis/postop). Artificial tears: Systane, Refresh, Retaine, TheraTears, Oasis.
Systemic Medications
Oral prednisone for severe uveitis, scleritis, or thyroid eye disease. Steroid-sparing immunomodulators: methotrexate, mycophenolate mofetil, azathioprine, adalimumab (FDA-approved for non-infectious uveitis), infliximab. Teprotumumab (Tepezza) for active thyroid eye disease. Acetazolamide for IIH. Tetracyclines (doxycycline 50–100 mg) for MGD, blepharitis, and recurrent erosion.
19 Classification Systems
Snellen Visual Acuity & ETDRS
The Snellen chart uses letters of decreasing size at 20 feet; acuity is recorded as a fraction (20/20 is normal). The ETDRS chart (Early Treatment Diabetic Retinopathy Study) is the research standard: 5 letters per line, logMAR spacing, used in clinical trials. Below 20/400, acuity is recorded as CF (count fingers) / HM (hand motion) / LP (light perception) / NLP. Legal blindness in the US is BCVA ≤ 20/200 in the better eye or VF ≤ 20°.
AAO POAG Severity (ICD-10 based)
| Stage | Criteria |
|---|---|
| Mild / Early | Optic nerve changes consistent with glaucoma; no visual field defect on white-on-white perimetry |
| Moderate | Optic nerve changes + visual field defect in one hemifield not within 5° of fixation |
| Severe / Advanced | Optic nerve changes + visual field defect in both hemifields and/or loss within 5° of fixation in at least one hemifield |
| Indeterminate | Unable to stage |
ETDRS Diabetic Retinopathy Severity
| Level | Findings |
|---|---|
| No DR | Normal |
| Mild NPDR | Microaneurysms only |
| Moderate NPDR | More than mild, less than severe; hemorrhages, CWS, hard exudates |
| Severe NPDR | 4-2-1 rule (hemorrhages in 4 quadrants, VB in 2, IRMA in 1) |
| Very Severe NPDR | Any two of the 4-2-1 criteria |
| PDR | Neovascularization; vitreous/preretinal hemorrhage |
| High-risk PDR | NVD ≥ 1/4–1/3 DA; any NVD with VH; NVE ≥ 1/2 DA with VH |
AREDS AMD Category (see Section 7)
Roper-Hall Chemical Injury Grading
| Grade | Cornea | Limbal Ischemia | Prognosis |
|---|---|---|---|
| I | Epithelial damage only | None | Good |
| II | Haze but iris detail visible | < 1/3 | Good |
| III | Total epithelial loss, stromal haze obscures iris | 1/3–1/2 | Guarded |
| IV | Cornea opaque, iris and pupil obscured | > 1/2 | Poor |
Hughes Classification (modified)
An alternative severity scale for ocular chemical burns emphasizing limbal stem cell damage and conjunctival epithelial loss, grouping injuries into mild, moderate-severe, and very severe based on corneal clarity and limbal blanching extent.
Dua Classification (Chemical Injury)
Grades 0–VI based on clock hours of limbal involvement and percentage of conjunctival involvement; better prognostic resolution than Roper-Hall for severe burns.
DEWS II Dry Eye Severity
| Level | Features | Management Step |
|---|---|---|
| 1 | Mild/episodic discomfort; minimal staining | Education, environmental modification, artificial tears, lid hygiene |
| 2 | Moderate symptoms; mild-moderate staining; TBUT < 10 s | Preservative-free tears, cyclosporine/lifitegrast, omega-3, warm compresses, tetracyclines |
| 3 | Severe/frequent symptoms; marked staining; filamentary keratitis | Serum tears, punctal plugs, therapeutic contact lenses, short-term topical steroids |
| 4 | Severe disabling symptoms; sterile ulceration | Systemic immunosuppression, tarsorrhaphy, amniotic membrane, scleral lenses |
RAPD Grading
Graded in log units (0.3, 0.6, 0.9, 1.2, 1.5+) based on neutral density filter required to equalize the swinging flashlight response. Any RAPD is abnormal and suggests asymmetric optic nerve or extensive retinal disease.
Fontana Spaces & Schlemm's Canal Reference
Anatomic reference for the trabecular outflow: aqueous passes through the uveal and corneoscleral meshwork, into the spaces of Fontana, across the juxtacanalicular meshwork, into Schlemm's canal, and then to collector channels and episcleral venous system. Most resistance is at the juxtacanalicular layer, the target of MIGS devices.
Shaffer & Schwartz-Matsuo / Schwalbe
Gonioscopy grading of the anterior chamber angle: Shaffer grade 4 (35–45°, wide open) to grade 0 (closed). The Schwartz-Matsuo phenomenon describes pigmented cells in the AC from chronic retinal detachment with photoreceptor outer segments reaching the AC — a recognized but rare entity.
20 The Eye Exam
The eye exam is the most specialized part of ophthalmology documentation. The provider moves fast; you must keep up. Here is the standard order and what each component captures.
1. Visual acuity (distance VAsc / VAcc, pinhole if reduced; near if indicated).
2. Pupillary exam: size in light and dark, reactivity, check for RAPD with swinging flashlight. Document as "PERRL, no APD" when normal.
3. Extraocular motility (EOMs): "full OU" when normal; note any restriction, pain, or diplopia.
4. Confrontation visual fields (CVF): "full to finger counting OU."
5. IOP: Goldmann applanation (gold standard), iCare, or Tonopen; document with units, eye, and time.
6. External exam: lids/lashes, orbit, periocular skin, proptosis.
7. Slit-lamp exam: Lids/Lashes (L/L), Conjunctiva/Sclera (C/S), Cornea (K), Anterior Chamber (AC — graded cell/flare 0 to 4+), Iris (I), Lens (L — grade NS/CC/PSC severity).
8. Dilated fundus exam (DFE): Disc (C/D ratio, margins, color), Macula (foveal light reflex, drusen, edema), Vessels (caliber, crossings, attenuation), Periphery (breaks, lattice, holes, tumors).
Cell and flare in the anterior chamber are graded on a 0 to 4+ scale per SUN criteria. "Trace" = 1–5 cells per 1×1 mm high-mag slit beam; 1+ = 6–15; 2+ = 16–25; 3+ = 26–50; 4+ = > 50. You will chart these numbers verbatim in uveitis follow-ups.
21 Abbreviations Master List
Laterality & Basic Terms
Anatomy & Exam
Diagnoses
Procedures & Medications
22 Sample HPI Templates
The following HPIs show the density and style expected in real ophthalmology clinic notes. Pattern-match off these during your first weeks.
"Mrs. [Name] is a 72-year-old female with a history of hypertension and type 2 diabetes (last A1C 6.8%) who presents for cataract evaluation. She reports 18 months of gradually worsening blurry vision OU, worse OD than OS, with significant glare and halos while driving at night. She has difficulty reading small print despite updated readers. No flashes, floaters, curtains, or sudden vision changes. No eye pain or redness. Last eye exam 14 months ago with her optometrist, who referred her for cataract surgery consultation. Current VAcc 20/60 OD and 20/40 OS. She is pseudophakic in neither eye. No prior ocular surgery. Currently on latanoprost qhs OU for ocular hypertension. Allergies: NKDA. She is motivated for surgery OD first."
"Mr. [Name] is a 74-year-old male with hypertension, hyperlipidemia, atrial fibrillation on apixaban, and 50-pack-year smoking history presenting with sudden painless vision loss OD that began this morning upon waking. He reports the vision in the right eye is "like a curtain from the bottom." No eye pain, headache, jaw claudication, scalp tenderness, or weight loss. No history of similar episodes, transient monocular vision loss, TIAs, or stroke. VAsc OD CF at 3 ft, OS 20/25. RAPD present OD. IOP 16/15. DFE OD shows a pale retina with a cherry-red spot at the fovea, consistent with CRAO. Urgent stroke workup initiated; ESR/CRP/platelets sent to rule out GCA; stroke service consulted."
"Ms. [Name] is a 28-year-old soft contact lens wearer presenting with 2 days of severe left eye pain, photophobia, redness, and foreign body sensation. She sleeps in her lenses and frequently swims in them. No prior episodes. No systemic symptoms. VAcc 20/20 OD, 20/80 OS with pinhole to 20/60. OS slit lamp: 2+ conjunctival injection, 2 mm central paracentral corneal infiltrate with overlying epithelial defect staining with fluorescein, 1+ AC cell, no hypopyon. Concerning for bacterial (possible Pseudomonas) keratitis. Corneal scraping for Gram stain and culture performed; fortified vancomycin and tobramycin q1h initiated; contact lenses cultured; daily follow-up planned."
"Mr. [Name] is a 66-year-old African American male with moderate POAG OU (diagnosed 2019) presenting for 4-month follow-up. He reports good compliance with latanoprost qhs OU and brimonidine BID OU, no missed doses. No eye pain, redness, or vision changes. Current IOP 15/14 (target ≤ 16 bilateral). VAcc 20/25 OD, 20/20 OS. C/D 0.75 OD, 0.7 OS, stable compared to prior. Repeat HVF 24-2 today shows stable inferior arcuate defect OD (MD −4.8, PSD 6.2) and early superior nasal step OS (MD −2.1). OCT RNFL stable. Plan: continue current regimen, RTC 4 months with repeat HVF/OCT."
"Mrs. [Name] is a 58-year-old female with type 2 diabetes for 15 years (last A1C 8.2%), hypertension, and CKD stage 3 presenting for annual diabetic eye exam. She denies vision changes, floaters, or flashes. No prior laser or injections. VAcc 20/30 OD, 20/25 OS. DFE: OD — diffuse dot-blot hemorrhages in all 4 quadrants, venous beading in 2 quadrants, small IRMA, consistent with severe NPDR; mild center-involving DME on OCT (CFT 340 μm). OS — moderate NPDR without DME (CFT 272 μm). Plan: initiate intravitreal aflibercept OD today, discussed risks/benefits; optimize A1C and BP with PCP; RTC 4 weeks for repeat injection OD."
"Mr. [Name] is a 34-year-old male presenting with left eye foreign body sensation after metal grinding without safety glasses at work 3 hours ago. He reports tearing, mild pain, and blurred vision OS. No penetrating trauma history, no double vision, no loss of vision. VAcc 20/20 OD, 20/30 OS. OS slit lamp: small metallic foreign body at 4 o'clock on the mid-peripheral cornea with surrounding rust ring, no Seidel sign, AC deep and quiet, lens clear. No red flags for open globe. FB and rust ring removed with spud and burr under topical anesthetic; fluorescein confirms small residual epithelial defect; moxifloxacin QID OS and cyclopentolate for comfort; RTC 24–48 hours for reassessment."
Ophthalmology is unique in medicine because it is both a medical and a surgical specialty, and because nearly all of its documentation revolves around precise, numeric data rather than narrative prose. The best ophthalmology scribes internalize the exam flow so completely that they anticipate the next data point before the provider speaks. They know that after a cataract complaint will come a discussion of biometry and IOL choice; that after a flash-and-floater presentation will come a detailed peripheral retinal exam and the phrase "no holes, no tears"; that after a glaucoma suspect visit will come an OCT RNFL order and a comparison to prior visual fields. Learn the providers' shorthand. Pre-populate notes from the imaging and prior visits. Know every abbreviation in Section 21 without hesitation. Always confirm laterality.
Ophthalmology rewards precision. A single wrong letter (OD vs OS) can cascade into a wrong-eye surgery. Take the responsibility seriously, move fast, but never at the expense of accuracy. Welcome to the eye clinic.
23 References & Sources
Clinical Practice Guidelines
AAO Primary Open-Angle Glaucoma Preferred Practice Pattern. Ophthalmology. 2021.
AAO Cataract in the Adult Eye Preferred Practice Pattern. Ophthalmology. 2022.
AAO Diabetic Retinopathy Preferred Practice Pattern. Ophthalmology. 2020.
AAO Uveitis Preferred Practice Pattern. Ophthalmology. 2018.
AAO Conjunctivitis Preferred Practice Pattern. Ophthalmology. 2019.
AAO Dry Eye Syndrome Preferred Practice Pattern. Ophthalmology. 2019.
AAO Age-Related Macular Degeneration Preferred Practice Pattern. Ophthalmology. 2020.
TFOS DEWS II Definition and Classification Report. Ocular Surface. 2017.
Landmark Clinical Trials
AREDS2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for AMD. JAMA. 2013.
Brown DM et al. ANCHOR: Ranibizumab versus PDT for neovascular AMD. NEJM. 2006.
Rosenfeld PJ et al. MARINA: Ranibizumab for neovascular AMD. NEJM. 2006.
DRCR.net Protocol T: Aflibercept, bevacizumab, or ranibizumab for DME. NEJM. 2015.
ETDRS Report Number 9: Early photocoagulation for diabetic retinopathy. Ophthalmology. 1991.
OHTS: Ocular Hypertension Treatment Study. Arch Ophthalmol. 2002.
CIGTS: Collaborative Initial Glaucoma Treatment Study. Ophthalmology. 2001.
Diagram & Figure Sources
Figure 1: Eye in the Orbit. OpenStax College. CC BY 3.0.
Figure 2: Schematic diagram of the human eye. Rhcastilhos & Jmarchn. Public domain.
Figure 3: Extraocular Muscles. OpenStax College. CC BY 3.0.
Figure 4: Cataract in human eye. Rakesh Ahuja, MD. CC BY-SA 3.0.
Figure 5: Glaucoma visual field loss. National Eye Institute / NIH. Public domain.